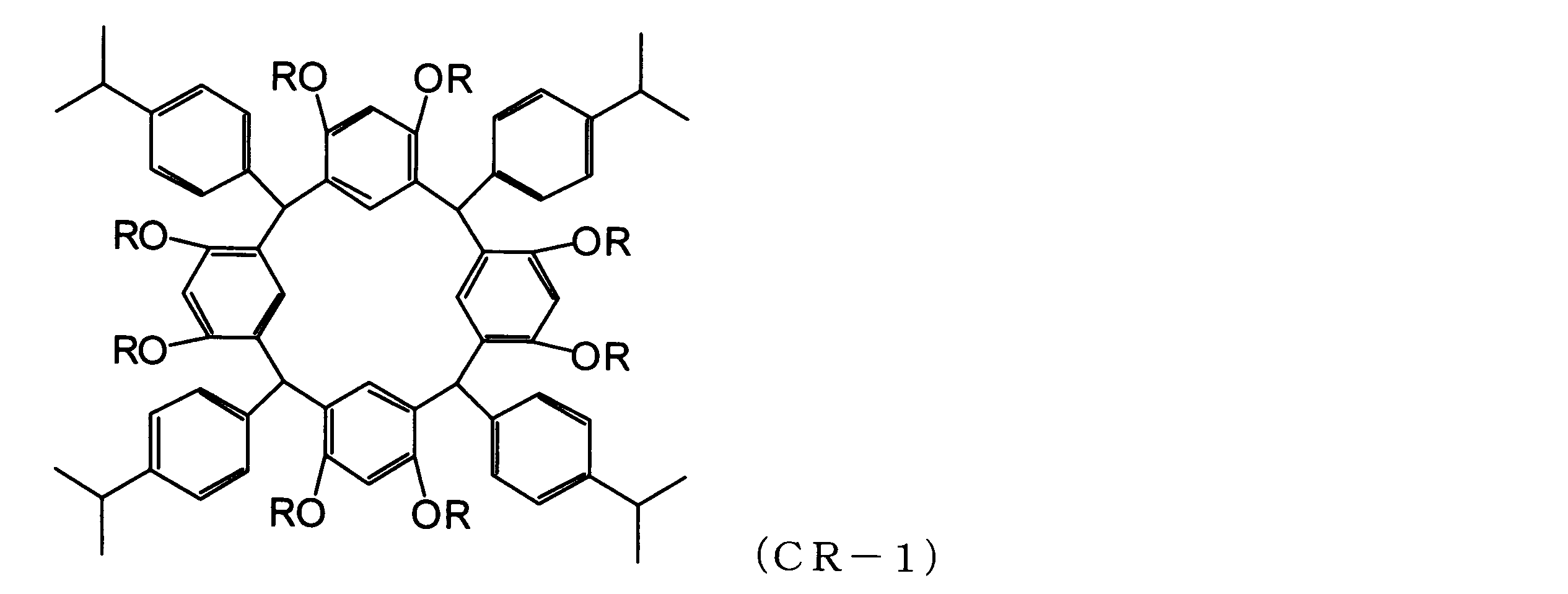
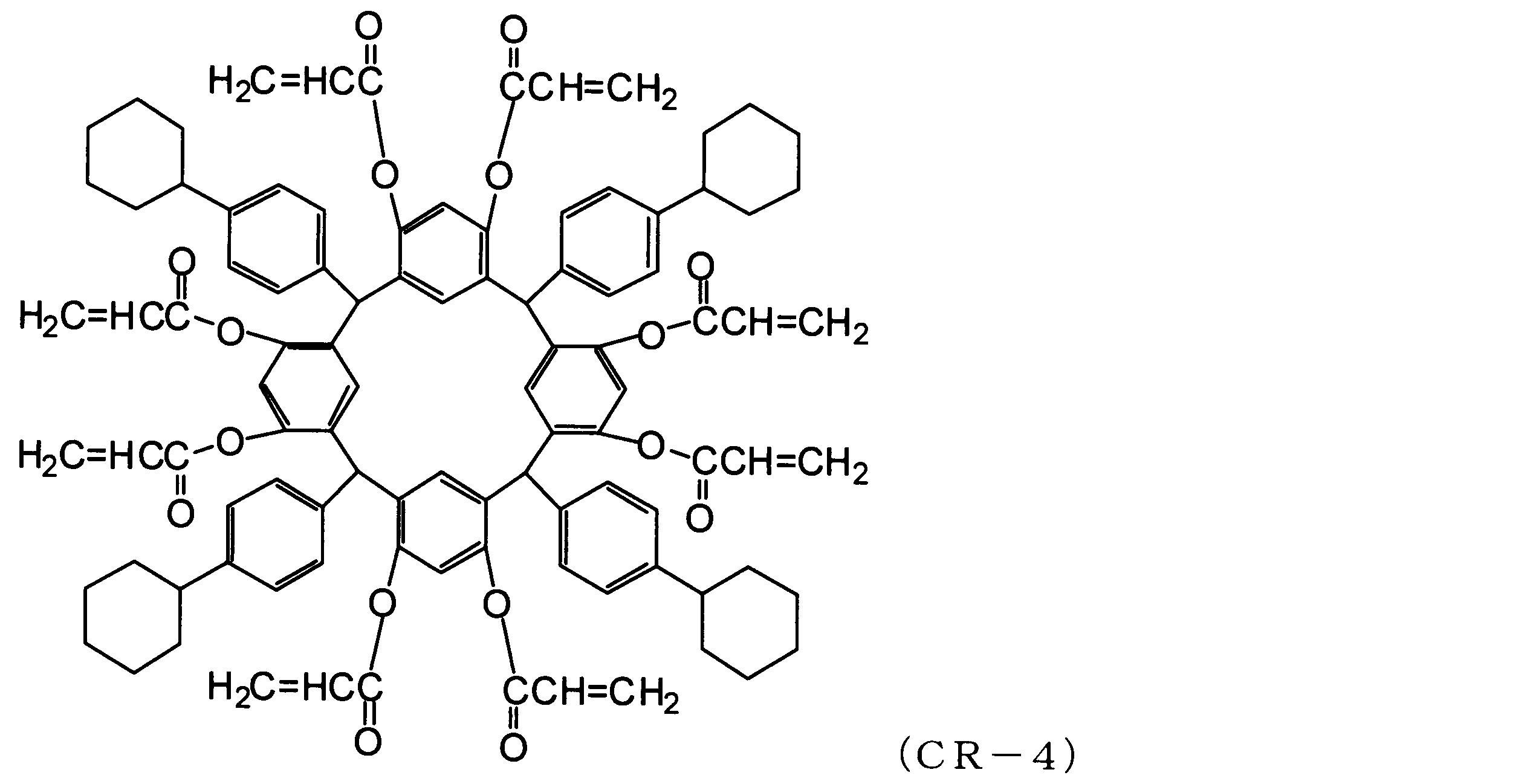
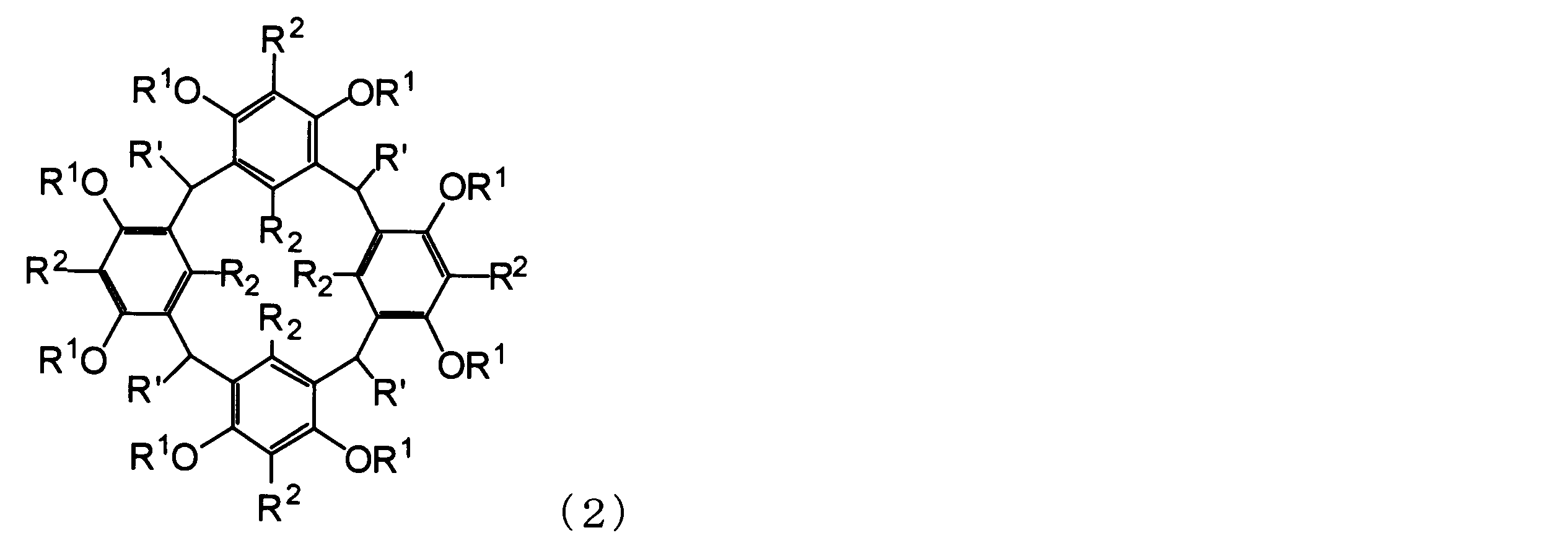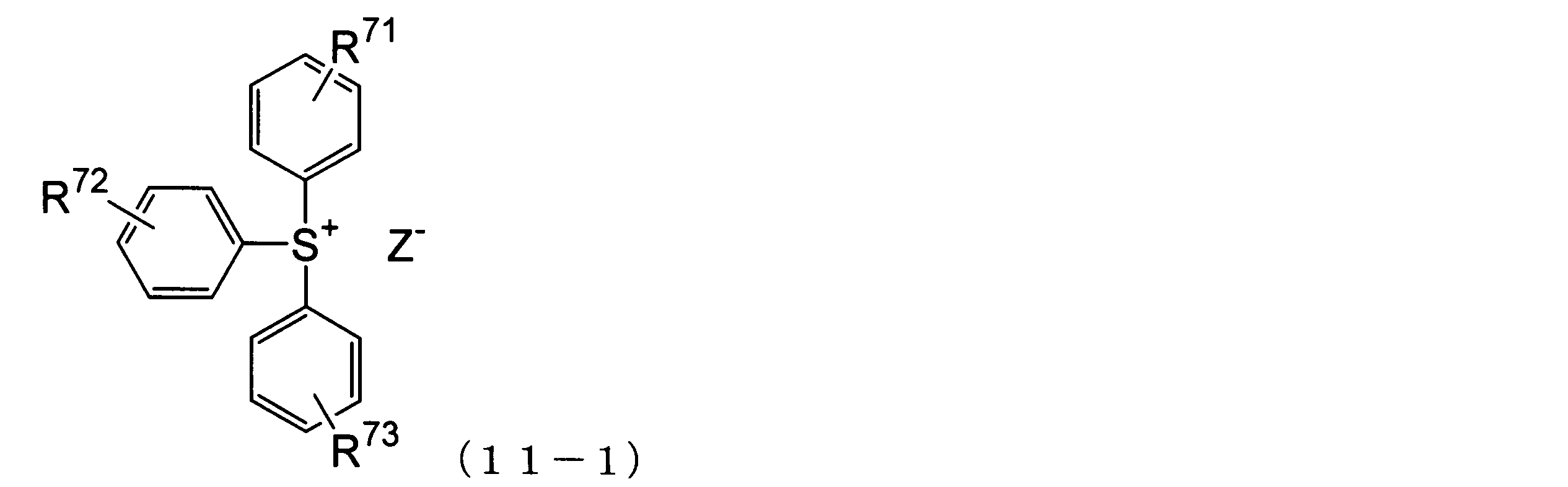WO2013073582A1 - 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 - Google Patents
環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 Download PDFInfo
- Publication number
- WO2013073582A1 WO2013073582A1 PCT/JP2012/079528 JP2012079528W WO2013073582A1 WO 2013073582 A1 WO2013073582 A1 WO 2013073582A1 JP 2012079528 W JP2012079528 W JP 2012079528W WO 2013073582 A1 WO2013073582 A1 WO 2013073582A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituted
- unsubstituted
- carbon atoms
- aliphatic hydrocarbon
- Prior art date
Links
- 150000001923 cyclic compounds Chemical class 0.000 title claims abstract description 127
- 230000005855 radiation Effects 0.000 title claims description 126
- 239000000203 mixture Substances 0.000 title claims description 123
- 238000000034 method Methods 0.000 title claims description 56
- 238000004519 manufacturing process Methods 0.000 title claims description 15
- -1 aldehyde compounds Chemical class 0.000 claims description 319
- 125000004432 carbon atom Chemical group C* 0.000 claims description 238
- 239000002253 acid Substances 0.000 claims description 93
- 125000001931 aliphatic group Chemical group 0.000 claims description 72
- 239000002904 solvent Substances 0.000 claims description 71
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 36
- 239000003431 cross linking reagent Substances 0.000 claims description 34
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 34
- 125000003118 aryl group Chemical group 0.000 claims description 33
- 239000007787 solid Substances 0.000 claims description 31
- 125000004122 cyclic group Chemical group 0.000 claims description 28
- 150000002989 phenols Chemical class 0.000 claims description 27
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 claims description 26
- 238000004090 dissolution Methods 0.000 claims description 26
- 239000000758 substrate Substances 0.000 claims description 26
- 125000003545 alkoxy group Chemical group 0.000 claims description 25
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 24
- 125000005843 halogen group Chemical group 0.000 claims description 23
- 125000005103 alkyl silyl group Chemical group 0.000 claims description 21
- 238000009792 diffusion process Methods 0.000 claims description 20
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 20
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 claims description 20
- 125000000623 heterocyclic group Chemical group 0.000 claims description 19
- 229910052799 carbon Inorganic materials 0.000 claims description 17
- 238000010894 electron beam technology Methods 0.000 claims description 15
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 14
- 125000003342 alkenyl group Chemical group 0.000 claims description 13
- 150000001721 carbon Chemical class 0.000 claims description 13
- 125000002252 acyl group Chemical group 0.000 claims description 11
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 10
- 238000004528 spin coating Methods 0.000 claims description 9
- 230000008569 process Effects 0.000 claims description 8
- 125000003277 amino group Chemical group 0.000 claims description 7
- 238000010884 ion-beam technique Methods 0.000 claims description 6
- 125000003107 substituted aryl group Chemical group 0.000 claims description 6
- 125000002947 alkylene group Chemical group 0.000 claims description 5
- 125000000732 arylene group Chemical group 0.000 claims description 4
- 238000006482 condensation reaction Methods 0.000 claims description 4
- 125000001033 ether group Chemical group 0.000 claims description 4
- NIBFJPXGNVPNHK-UHFFFAOYSA-N 2,2-difluoro-1,3-benzodioxole-4-carbaldehyde Chemical group C1=CC(C=O)=C2OC(F)(F)OC2=C1 NIBFJPXGNVPNHK-UHFFFAOYSA-N 0.000 claims description 3
- 125000001183 hydrocarbyl group Chemical group 0.000 claims 1
- 150000001875 compounds Chemical class 0.000 description 122
- 229910052757 nitrogen Inorganic materials 0.000 description 66
- 239000010408 film Substances 0.000 description 36
- 230000015572 biosynthetic process Effects 0.000 description 33
- 238000003786 synthesis reaction Methods 0.000 description 33
- 239000000243 solution Substances 0.000 description 29
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Natural products C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 27
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 26
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 25
- 125000000217 alkyl group Chemical group 0.000 description 22
- 235000013824 polyphenols Nutrition 0.000 description 22
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 20
- 235000019441 ethanol Nutrition 0.000 description 20
- 230000035945 sensitivity Effects 0.000 description 20
- 238000006243 chemical reaction Methods 0.000 description 19
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 17
- 239000004094 surface-active agent Substances 0.000 description 17
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 16
- 239000003795 chemical substances by application Substances 0.000 description 16
- 238000011161 development Methods 0.000 description 16
- 239000007983 Tris buffer Substances 0.000 description 15
- 230000000694 effects Effects 0.000 description 15
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 14
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 14
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 14
- LZCLXQDLBQLTDK-UHFFFAOYSA-N ethyl 2-hydroxypropanoate Chemical compound CCOC(=O)C(C)O LZCLXQDLBQLTDK-UHFFFAOYSA-N 0.000 description 14
- 239000000126 substance Substances 0.000 description 14
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 13
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 13
- 238000004132 cross linking Methods 0.000 description 13
- 239000000463 material Substances 0.000 description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 13
- 229920005989 resin Polymers 0.000 description 13
- 239000011347 resin Substances 0.000 description 13
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 12
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 12
- UHOVQNZJYSORNB-UHFFFAOYSA-N monobenzene Natural products C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 12
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 11
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 11
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 11
- CZZYITDELCSZES-UHFFFAOYSA-N diphenylmethane Chemical compound C=1C=CC=CC=1CC1=CC=CC=C1 CZZYITDELCSZES-UHFFFAOYSA-N 0.000 description 11
- 239000007788 liquid Substances 0.000 description 11
- 239000003960 organic solvent Substances 0.000 description 11
- LLHKCFNBLRBOGN-UHFFFAOYSA-N propylene glycol methyl ether acetate Chemical compound COCC(C)OC(C)=O LLHKCFNBLRBOGN-UHFFFAOYSA-N 0.000 description 11
- 238000005406 washing Methods 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 10
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 10
- 239000007864 aqueous solution Substances 0.000 description 10
- ZSIAUFGUXNUGDI-UHFFFAOYSA-N hexan-1-ol Chemical compound CCCCCCO ZSIAUFGUXNUGDI-UHFFFAOYSA-N 0.000 description 10
- 230000001771 impaired effect Effects 0.000 description 10
- 238000005160 1H NMR spectroscopy Methods 0.000 description 9
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- 239000000654 additive Substances 0.000 description 9
- BTANRVKWQNVYAZ-UHFFFAOYSA-N butan-2-ol Chemical compound CCC(C)O BTANRVKWQNVYAZ-UHFFFAOYSA-N 0.000 description 9
- 125000000524 functional group Chemical group 0.000 description 9
- VPVSTMAPERLKKM-UHFFFAOYSA-N glycoluril Chemical class N1C(=O)NC2NC(=O)NC21 VPVSTMAPERLKKM-UHFFFAOYSA-N 0.000 description 9
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 9
- 125000004433 nitrogen atom Chemical group N* 0.000 description 9
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 9
- 229920000642 polymer Polymers 0.000 description 9
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 8
- BBMCTIGTTCKYKF-UHFFFAOYSA-N 1-heptanol Chemical compound CCCCCCCO BBMCTIGTTCKYKF-UHFFFAOYSA-N 0.000 description 8
- HCFAJYNVAYBARA-UHFFFAOYSA-N 4-heptanone Chemical compound CCCC(=O)CCC HCFAJYNVAYBARA-UHFFFAOYSA-N 0.000 description 8
- 238000002425 crystallisation Methods 0.000 description 8
- 230000008025 crystallization Effects 0.000 description 8
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 8
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 8
- GHMLBKRAJCXXBS-UHFFFAOYSA-N resorcinol Chemical compound OC1=CC=CC(O)=C1 GHMLBKRAJCXXBS-UHFFFAOYSA-N 0.000 description 8
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 8
- AGJAUFUNZWHLKE-UHFFFAOYSA-N (2E,4E)-N-isobutyl-2,4-tetradecadienamide Natural products CCCCCCCCCC=CC=CC(=O)NCC(C)C AGJAUFUNZWHLKE-UHFFFAOYSA-N 0.000 description 7
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 7
- 150000001408 amides Chemical class 0.000 description 7
- 229940116333 ethyl lactate Drugs 0.000 description 7
- 238000001914 filtration Methods 0.000 description 7
- 230000009477 glass transition Effects 0.000 description 7
- 229910052736 halogen Inorganic materials 0.000 description 7
- 150000002367 halogens Chemical class 0.000 description 7
- UFWIBTONFRDIAS-UHFFFAOYSA-N naphthalene-acid Natural products C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 7
- NUJGJRNETVAIRJ-UHFFFAOYSA-N octanal Chemical compound CCCCCCCC=O NUJGJRNETVAIRJ-UHFFFAOYSA-N 0.000 description 7
- QQZOPKMRPOGIEB-UHFFFAOYSA-N 2-Oxohexane Chemical compound CCCCC(C)=O QQZOPKMRPOGIEB-UHFFFAOYSA-N 0.000 description 6
- ZPVFWPFBNIEHGJ-UHFFFAOYSA-N 2-octanone Chemical compound CCCCCCC(C)=O ZPVFWPFBNIEHGJ-UHFFFAOYSA-N 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 6
- 239000004593 Epoxy Substances 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Natural products NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 6
- HFBMWMNUJJDEQZ-UHFFFAOYSA-N acryloyl chloride Chemical compound ClC(=O)C=C HFBMWMNUJJDEQZ-UHFFFAOYSA-N 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- 150000007514 bases Chemical class 0.000 description 6
- 239000004305 biphenyl Substances 0.000 description 6
- 235000010290 biphenyl Nutrition 0.000 description 6
- 238000000576 coating method Methods 0.000 description 6
- 230000000052 comparative effect Effects 0.000 description 6
- WTWBUQJHJGUZCY-UHFFFAOYSA-N cuminaldehyde Chemical compound CC(C)C1=CC=C(C=O)C=C1 WTWBUQJHJGUZCY-UHFFFAOYSA-N 0.000 description 6
- 125000006165 cyclic alkyl group Chemical group 0.000 description 6
- BGTOWKSIORTVQH-UHFFFAOYSA-N cyclopentanone Chemical compound O=C1CCCC1 BGTOWKSIORTVQH-UHFFFAOYSA-N 0.000 description 6
- MWKFXSUHUHTGQN-UHFFFAOYSA-N decan-1-ol Chemical compound CCCCCCCCCCO MWKFXSUHUHTGQN-UHFFFAOYSA-N 0.000 description 6
- DIOQZVSQGTUSAI-UHFFFAOYSA-N decane Chemical compound CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 6
- HFJRKMMYBMWEAD-UHFFFAOYSA-N dodecanal Chemical compound CCCCCCCCCCCC=O HFJRKMMYBMWEAD-UHFFFAOYSA-N 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 150000002430 hydrocarbons Chemical group 0.000 description 6
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 6
- ZXEKIIBDNHEJCQ-UHFFFAOYSA-N isobutanol Chemical compound CC(C)CO ZXEKIIBDNHEJCQ-UHFFFAOYSA-N 0.000 description 6
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 6
- VKCYHJWLYTUGCC-UHFFFAOYSA-N nonan-2-one Chemical compound CCCCCCCC(C)=O VKCYHJWLYTUGCC-UHFFFAOYSA-N 0.000 description 6
- 229910052698 phosphorus Inorganic materials 0.000 description 6
- 239000011574 phosphorus Substances 0.000 description 6
- 238000007747 plating Methods 0.000 description 6
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 6
- 239000004065 semiconductor Substances 0.000 description 6
- 238000003860 storage Methods 0.000 description 6
- WGTYBPLFGIVFAS-UHFFFAOYSA-M tetramethylammonium hydroxide Chemical compound [OH-].C[N+](C)(C)C WGTYBPLFGIVFAS-UHFFFAOYSA-M 0.000 description 6
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 5
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 5
- 230000002378 acidificating effect Effects 0.000 description 5
- 150000001298 alcohols Chemical class 0.000 description 5
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzenecarboxaldehyde Natural products O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 5
- 239000004202 carbamide Substances 0.000 description 5
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 5
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 5
- 239000003054 catalyst Substances 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 5
- 239000005453 ketone based solvent Substances 0.000 description 5
- 239000002736 nonionic surfactant Substances 0.000 description 5
- PGMYKACGEOXYJE-UHFFFAOYSA-N pentyl acetate Chemical compound CCCCCOC(C)=O PGMYKACGEOXYJE-UHFFFAOYSA-N 0.000 description 5
- 238000000746 purification Methods 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- JOLQKTGDSGKSKJ-UHFFFAOYSA-N 1-ethoxypropan-2-ol Chemical compound CCOCC(C)O JOLQKTGDSGKSKJ-UHFFFAOYSA-N 0.000 description 4
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 4
- ZNQVEEAIQZEUHB-UHFFFAOYSA-N 2-ethoxyethanol Chemical compound CCOCCO ZNQVEEAIQZEUHB-UHFFFAOYSA-N 0.000 description 4
- SVONRAPFKPVNKG-UHFFFAOYSA-N 2-ethoxyethyl acetate Chemical compound CCOCCOC(C)=O SVONRAPFKPVNKG-UHFFFAOYSA-N 0.000 description 4
- QMYGFTJCQFEDST-UHFFFAOYSA-N 3-methoxybutyl acetate Chemical compound COC(C)CCOC(C)=O QMYGFTJCQFEDST-UHFFFAOYSA-N 0.000 description 4
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 4
- MRABAEUHTLLEML-UHFFFAOYSA-N Butyl lactate Chemical compound CCCCOC(=O)C(C)O MRABAEUHTLLEML-UHFFFAOYSA-N 0.000 description 4
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 4
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 4
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 4
- 239000003377 acid catalyst Substances 0.000 description 4
- 230000000996 additive effect Effects 0.000 description 4
- 239000005456 alcohol based solvent Substances 0.000 description 4
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 4
- 125000004849 alkoxymethyl group Chemical group 0.000 description 4
- NMJJFJNHVMGPGM-UHFFFAOYSA-N butyl formate Chemical compound CCCCOC=O NMJJFJNHVMGPGM-UHFFFAOYSA-N 0.000 description 4
- 238000004140 cleaning Methods 0.000 description 4
- 239000013078 crystal Substances 0.000 description 4
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 description 4
- KSMVZQYAVGTKIV-UHFFFAOYSA-N decanal Chemical compound CCCCCCCCCC=O KSMVZQYAVGTKIV-UHFFFAOYSA-N 0.000 description 4
- 239000003759 ester based solvent Substances 0.000 description 4
- 238000005530 etching Methods 0.000 description 4
- 239000004210 ether based solvent Substances 0.000 description 4
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 4
- BHXIWUJLHYHGSJ-UHFFFAOYSA-N ethyl 3-ethoxypropanoate Chemical compound CCOCCC(=O)OCC BHXIWUJLHYHGSJ-UHFFFAOYSA-N 0.000 description 4
- FKRCODPIKNYEAC-UHFFFAOYSA-N ethyl propionate Chemical compound CCOC(=O)CC FKRCODPIKNYEAC-UHFFFAOYSA-N 0.000 description 4
- CATSNJVOTSVZJV-UHFFFAOYSA-N heptan-2-one Chemical compound CCCCCC(C)=O CATSNJVOTSVZJV-UHFFFAOYSA-N 0.000 description 4
- QNVRIHYSUZMSGM-UHFFFAOYSA-N hexan-2-ol Chemical compound CCCCC(C)O QNVRIHYSUZMSGM-UHFFFAOYSA-N 0.000 description 4
- FUZZWVXGSFPDMH-UHFFFAOYSA-M hexanoate Chemical compound CCCCCC([O-])=O FUZZWVXGSFPDMH-UHFFFAOYSA-M 0.000 description 4
- PHTQWCKDNZKARW-UHFFFAOYSA-N isoamylol Chemical compound CC(C)CCO PHTQWCKDNZKARW-UHFFFAOYSA-N 0.000 description 4
- 150000002576 ketones Chemical class 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 4
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 4
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 4
- JYVLIDXNZAXMDK-UHFFFAOYSA-N pentan-2-ol Chemical compound CCCC(C)O JYVLIDXNZAXMDK-UHFFFAOYSA-N 0.000 description 4
- YFSUTJLHUFNCNZ-UHFFFAOYSA-N perfluorooctane-1-sulfonic acid Chemical compound OS(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F YFSUTJLHUFNCNZ-UHFFFAOYSA-N 0.000 description 4
- ILVGAIQLOCKNQA-UHFFFAOYSA-N propyl 2-hydroxypropanoate Chemical compound CCCOC(=O)C(C)O ILVGAIQLOCKNQA-UHFFFAOYSA-N 0.000 description 4
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 4
- 229910052710 silicon Inorganic materials 0.000 description 4
- 239000010703 silicon Substances 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 4
- 239000008096 xylene Substances 0.000 description 4
- RYNQKSJRFHJZTK-UHFFFAOYSA-N (3-methoxy-3-methylbutyl) acetate Chemical compound COC(C)(C)CCOC(C)=O RYNQKSJRFHJZTK-UHFFFAOYSA-N 0.000 description 3
- DYLIWHYUXAJDOJ-OWOJBTEDSA-N (e)-4-(6-aminopurin-9-yl)but-2-en-1-ol Chemical compound NC1=NC=NC2=C1N=CN2C\C=C\CO DYLIWHYUXAJDOJ-OWOJBTEDSA-N 0.000 description 3
- 0 *C(C(C(C(*)c(c(O)c1*)c(*C(*C(C2*)C(*)=C(*)C3*)C3C(*)c(c(*)c34)c(*)c(*)c3O)c2c1O*)C(*)=C1C4I)O)C1O Chemical compound *C(C(C(C(*)c(c(O)c1*)c(*C(*C(C2*)C(*)=C(*)C3*)C3C(*)c(c(*)c34)c(*)c(*)c3O)c2c1O*)C(*)=C1C4I)O)C1O 0.000 description 3
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 3
- VXQBJTKSVGFQOL-UHFFFAOYSA-N 2-(2-butoxyethoxy)ethyl acetate Chemical compound CCCCOCCOCCOC(C)=O VXQBJTKSVGFQOL-UHFFFAOYSA-N 0.000 description 3
- FPZWZCWUIYYYBU-UHFFFAOYSA-N 2-(2-ethoxyethoxy)ethyl acetate Chemical compound CCOCCOCCOC(C)=O FPZWZCWUIYYYBU-UHFFFAOYSA-N 0.000 description 3
- SBASXUCJHJRPEV-UHFFFAOYSA-N 2-(2-methoxyethoxy)ethanol Chemical compound COCCOCCO SBASXUCJHJRPEV-UHFFFAOYSA-N 0.000 description 3
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- PTTPXKJBFFKCEK-UHFFFAOYSA-N 2-Methyl-4-heptanone Chemical compound CC(C)CC(=O)CC(C)C PTTPXKJBFFKCEK-UHFFFAOYSA-N 0.000 description 3
- WFSMVVDJSNMRAR-UHFFFAOYSA-N 2-[2-(2-ethoxyethoxy)ethoxy]ethanol Chemical compound CCOCCOCCOCCO WFSMVVDJSNMRAR-UHFFFAOYSA-N 0.000 description 3
- SFNWXOPQLKYXHN-UHFFFAOYSA-N 2-cyclohexylbenzaldehyde Chemical compound O=CC1=CC=CC=C1C1CCCCC1 SFNWXOPQLKYXHN-UHFFFAOYSA-N 0.000 description 3
- MFKRHJVUCZRDTF-UHFFFAOYSA-N 3-methoxy-3-methylbutan-1-ol Chemical compound COC(C)(C)CCO MFKRHJVUCZRDTF-UHFFFAOYSA-N 0.000 description 3
- WVYWICLMDOOCFB-UHFFFAOYSA-N 4-methyl-2-pentanol Chemical compound CC(C)CC(C)O WVYWICLMDOOCFB-UHFFFAOYSA-N 0.000 description 3
- VGVHNLRUAMRIEW-UHFFFAOYSA-N 4-methylcyclohexan-1-one Chemical compound CC1CCC(=O)CC1 VGVHNLRUAMRIEW-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 3
- 239000005711 Benzoic acid Substances 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 239000004215 Carbon black (E152) Substances 0.000 description 3
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 3
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 3
- 150000003973 alkyl amines Chemical class 0.000 description 3
- 229910052782 aluminium Inorganic materials 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 125000003368 amide group Chemical group 0.000 description 3
- 229940072049 amyl acetate Drugs 0.000 description 3
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- 235000010233 benzoic acid Nutrition 0.000 description 3
- 239000001191 butyl (2R)-2-hydroxypropanoate Substances 0.000 description 3
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 3
- 150000001735 carboxylic acids Chemical class 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 229910052802 copper Inorganic materials 0.000 description 3
- 239000010949 copper Substances 0.000 description 3
- 125000000753 cycloalkyl group Chemical group 0.000 description 3
- KVFDZFBHBWTVID-UHFFFAOYSA-N cyclohexane-carboxaldehyde Natural products O=CC1CCCCC1 KVFDZFBHBWTVID-UHFFFAOYSA-N 0.000 description 3
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 229910001873 dinitrogen Inorganic materials 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 125000005745 ethoxymethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])* 0.000 description 3
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 3
- MNWFXJYAOYHMED-UHFFFAOYSA-M heptanoate Chemical compound CCCCCCC([O-])=O MNWFXJYAOYHMED-UHFFFAOYSA-M 0.000 description 3
- JARKCYVAAOWBJS-UHFFFAOYSA-N hexanal Chemical compound CCCCCC=O JARKCYVAAOWBJS-UHFFFAOYSA-N 0.000 description 3
- 229930195733 hydrocarbon Natural products 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 229940035429 isobutyl alcohol Drugs 0.000 description 3
- 238000001459 lithography Methods 0.000 description 3
- 150000007974 melamines Chemical class 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- JESXATFQYMPTNL-UHFFFAOYSA-N mono-hydroxyphenyl-ethylene Natural products OC1=CC=CC=C1C=C JESXATFQYMPTNL-UHFFFAOYSA-N 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 125000001624 naphthyl group Chemical group 0.000 description 3
- GYHFUZHODSMOHU-UHFFFAOYSA-N nonanal Chemical compound CCCCCCCCC=O GYHFUZHODSMOHU-UHFFFAOYSA-N 0.000 description 3
- 125000000962 organic group Chemical group 0.000 description 3
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 3
- 238000000059 patterning Methods 0.000 description 3
- QCCDLTOVEPVEJK-UHFFFAOYSA-N phenylacetone Chemical compound CC(=O)CC1=CC=CC=C1 QCCDLTOVEPVEJK-UHFFFAOYSA-N 0.000 description 3
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 3
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 239000012487 rinsing solution Substances 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 239000007921 spray Substances 0.000 description 3
- 238000000859 sublimation Methods 0.000 description 3
- 230000008022 sublimation Effects 0.000 description 3
- NAWXUBYGYWOOIX-SFHVURJKSA-N (2s)-2-[[4-[2-(2,4-diaminoquinazolin-6-yl)ethyl]benzoyl]amino]-4-methylidenepentanedioic acid Chemical compound C1=CC2=NC(N)=NC(N)=C2C=C1CCC1=CC=C(C(=O)N[C@@H](CC(=C)C(O)=O)C(O)=O)C=C1 NAWXUBYGYWOOIX-SFHVURJKSA-N 0.000 description 2
- RXYPXQSKLGGKOL-UHFFFAOYSA-N 1,4-dimethylpiperazine Chemical compound CN1CCN(C)CC1 RXYPXQSKLGGKOL-UHFFFAOYSA-N 0.000 description 2
- RUFPHBVGCFYCNW-UHFFFAOYSA-N 1-naphthylamine Chemical compound C1=CC=C2C(N)=CC=CC2=C1 RUFPHBVGCFYCNW-UHFFFAOYSA-N 0.000 description 2
- OJVAMHKKJGICOG-UHFFFAOYSA-N 2,5-hexanedione Chemical compound CC(=O)CCC(C)=O OJVAMHKKJGICOG-UHFFFAOYSA-N 0.000 description 2
- QNVRIHYSUZMSGM-LURJTMIESA-N 2-Hexanol Natural products CCCC[C@H](C)O QNVRIHYSUZMSGM-LURJTMIESA-N 0.000 description 2
- GSMHKHIXWAAXIP-UHFFFAOYSA-N 2-[5,8-bis(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound CC(O)(C)C1=CC=C(C(C)(C)O)C2=CC(C(C)(O)C)=CC=C21 GSMHKHIXWAAXIP-UHFFFAOYSA-N 0.000 description 2
- DRYBUHKBBRHEAE-UHFFFAOYSA-N 2-[diazo(propan-2-ylsulfonyl)methyl]sulfonylpropane Chemical compound CC(C)S(=O)(=O)C(=[N+]=[N-])S(=O)(=O)C(C)C DRYBUHKBBRHEAE-UHFFFAOYSA-N 0.000 description 2
- CETWDUZRCINIHU-UHFFFAOYSA-N 2-heptanol Chemical compound CCCCCC(C)O CETWDUZRCINIHU-UHFFFAOYSA-N 0.000 description 2
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 2
- BSKHPKMHTQYZBB-UHFFFAOYSA-N 2-methylpyridine Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 2
- LCRCBXLHWTVPEQ-UHFFFAOYSA-N 2-phenylbenzaldehyde Chemical compound O=CC1=CC=CC=C1C1=CC=CC=C1 LCRCBXLHWTVPEQ-UHFFFAOYSA-N 0.000 description 2
- VQGHOUODWALEFC-UHFFFAOYSA-N 2-phenylpyridine Chemical compound C1=CC=CC=C1C1=CC=CC=N1 VQGHOUODWALEFC-UHFFFAOYSA-N 0.000 description 2
- DTALCVXXATYTQJ-UHFFFAOYSA-N 2-propan-2-ylbenzaldehyde Chemical compound CC(C)C1=CC=CC=C1C=O DTALCVXXATYTQJ-UHFFFAOYSA-N 0.000 description 2
- MWZLEHUCHYHXOV-UHFFFAOYSA-N 2-propylbenzaldehyde Chemical compound CCCC1=CC=CC=C1C=O MWZLEHUCHYHXOV-UHFFFAOYSA-N 0.000 description 2
- JJYPMNFTHPTTDI-UHFFFAOYSA-N 3-methylaniline Chemical compound CC1=CC=CC(N)=C1 JJYPMNFTHPTTDI-UHFFFAOYSA-N 0.000 description 2
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 2
- JVZRCNQLWOELDU-UHFFFAOYSA-N 4-Phenylpyridine Chemical compound C1=CC=CC=C1C1=CC=NC=C1 JVZRCNQLWOELDU-UHFFFAOYSA-N 0.000 description 2
- YXZXRYDYTRYFAF-UHFFFAOYSA-M 4-methylbenzenesulfonate;triphenylsulfanium Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 YXZXRYDYTRYFAF-UHFFFAOYSA-M 0.000 description 2
- XLSZMDLNRCVEIJ-UHFFFAOYSA-N 4-methylimidazole Chemical compound CC1=CNC=N1 XLSZMDLNRCVEIJ-UHFFFAOYSA-N 0.000 description 2
- FKNQCJSGGFJEIZ-UHFFFAOYSA-N 4-methylpyridine Chemical compound CC1=CC=NC=C1 FKNQCJSGGFJEIZ-UHFFFAOYSA-N 0.000 description 2
- 125000004199 4-trifluoromethylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C(F)(F)F 0.000 description 2
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical compound N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 description 2
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 2
- DLFVBJFMPXGRIB-UHFFFAOYSA-N Acetamide Chemical compound CC(N)=O DLFVBJFMPXGRIB-UHFFFAOYSA-N 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 2
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 2
- RZKSECIXORKHQS-UHFFFAOYSA-N Heptan-3-ol Chemical compound CCCCC(O)CC RZKSECIXORKHQS-UHFFFAOYSA-N 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- AMIMRNSIRUDHCM-UHFFFAOYSA-N Isopropylaldehyde Chemical compound CC(C)C=O AMIMRNSIRUDHCM-UHFFFAOYSA-N 0.000 description 2
- WMFYOYKPJLRMJI-UHFFFAOYSA-N Lercanidipine hydrochloride Chemical compound Cl.COC(=O)C1=C(C)NC(C)=C(C(=O)OC(C)(C)CN(C)CCC(C=2C=CC=CC=2)C=2C=CC=CC=2)C1C1=CC=CC([N+]([O-])=O)=C1 WMFYOYKPJLRMJI-UHFFFAOYSA-N 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- 229920000877 Melamine resin Polymers 0.000 description 2
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 2
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 2
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 2
- OHLUUHNLEMFGTQ-UHFFFAOYSA-N N-methylacetamide Chemical compound CNC(C)=O OHLUUHNLEMFGTQ-UHFFFAOYSA-N 0.000 description 2
- AFBPFSWMIHJQDM-UHFFFAOYSA-N N-methylaniline Chemical compound CNC1=CC=CC=C1 AFBPFSWMIHJQDM-UHFFFAOYSA-N 0.000 description 2
- ATHHXGZTWNVVOU-UHFFFAOYSA-N N-methylformamide Chemical compound CNC=O ATHHXGZTWNVVOU-UHFFFAOYSA-N 0.000 description 2
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical compound OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- NBBJYMSMWIIQGU-UHFFFAOYSA-N Propionic aldehyde Chemical compound CCC=O NBBJYMSMWIIQGU-UHFFFAOYSA-N 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- POQWSGILBITVBD-UHFFFAOYSA-N [3-(2-hydroxypropan-2-yl)phenyl] benzoate Chemical compound CC(C)(O)C1=CC=CC(OC(=O)C=2C=CC=CC=2)=C1 POQWSGILBITVBD-UHFFFAOYSA-N 0.000 description 2
- CWRYPZZKDGJXCA-UHFFFAOYSA-N acenaphthene Chemical compound C1=CC(CC2)=C3C2=CC=CC3=C1 CWRYPZZKDGJXCA-UHFFFAOYSA-N 0.000 description 2
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 2
- YRKCREAYFQTBPV-UHFFFAOYSA-N acetylacetone Chemical compound CC(=O)CC(C)=O YRKCREAYFQTBPV-UHFFFAOYSA-N 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- DZBUGLKDJFMEHC-UHFFFAOYSA-N acridine Chemical compound C1=CC=CC2=CC3=CC=CC=C3N=C21 DZBUGLKDJFMEHC-UHFFFAOYSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 2
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 2
- 239000012965 benzophenone Substances 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- SZUCEMJQEUTBEW-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;acetate Chemical compound CC([O-])=O.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 SZUCEMJQEUTBEW-UHFFFAOYSA-M 0.000 description 2
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 2
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- YCIMNLLNPGFGHC-UHFFFAOYSA-N catechol Chemical compound OC1=CC=CC=C1O YCIMNLLNPGFGHC-UHFFFAOYSA-N 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 2
- INVYSLWXPIEDIQ-UHFFFAOYSA-N cyclobutanecarbaldehyde Chemical compound O=CC1CCC1 INVYSLWXPIEDIQ-UHFFFAOYSA-N 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- JMYVMOUINOAAPA-UHFFFAOYSA-N cyclopropanecarbaldehyde Chemical compound O=CC1CC1 JMYVMOUINOAAPA-UHFFFAOYSA-N 0.000 description 2
- 230000007547 defect Effects 0.000 description 2
- JQVDAXLFBXTEQA-UHFFFAOYSA-N dibutylamine Chemical compound CCCCNCCCC JQVDAXLFBXTEQA-UHFFFAOYSA-N 0.000 description 2
- 238000000113 differential scanning calorimetry Methods 0.000 description 2
- DMBHHRLKUKUOEG-UHFFFAOYSA-N diphenylamine Chemical compound C=1C=CC=CC=1NC1=CC=CC=C1 DMBHHRLKUKUOEG-UHFFFAOYSA-N 0.000 description 2
- 238000001312 dry etching Methods 0.000 description 2
- 239000000975 dye Substances 0.000 description 2
- 125000004185 ester group Chemical group 0.000 description 2
- XLLIQLLCWZCATF-UHFFFAOYSA-N ethylene glycol monomethyl ether acetate Natural products COCCOC(C)=O XLLIQLLCWZCATF-UHFFFAOYSA-N 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 235000019256 formaldehyde Nutrition 0.000 description 2
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 238000001879 gelation Methods 0.000 description 2
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 229910052737 gold Inorganic materials 0.000 description 2
- 239000010931 gold Substances 0.000 description 2
- NGAZZOYFWWSOGK-UHFFFAOYSA-N heptan-3-one Chemical compound CCCCC(=O)CC NGAZZOYFWWSOGK-UHFFFAOYSA-N 0.000 description 2
- 125000001072 heteroaryl group Chemical group 0.000 description 2
- ZOCHHNOQQHDWHG-UHFFFAOYSA-N hexan-3-ol Chemical compound CCCC(O)CC ZOCHHNOQQHDWHG-UHFFFAOYSA-N 0.000 description 2
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 description 2
- AOGQPLXWSUTHQB-UHFFFAOYSA-N hexyl acetate Chemical compound CCCCCCOC(C)=O AOGQPLXWSUTHQB-UHFFFAOYSA-N 0.000 description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 2
- 239000003456 ion exchange resin Substances 0.000 description 2
- 229920003303 ion-exchange polymer Polymers 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- HJOVHMDZYOCNQW-UHFFFAOYSA-N isophorone Chemical compound CC1=CC(=O)CC(C)(C)C1 HJOVHMDZYOCNQW-UHFFFAOYSA-N 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- VHRYZQNGTZXDNX-UHFFFAOYSA-N methacryloyl chloride Chemical compound CC(=C)C(Cl)=O VHRYZQNGTZXDNX-UHFFFAOYSA-N 0.000 description 2
- TZIHFWKZFHZASV-UHFFFAOYSA-N methyl formate Chemical compound COC=O TZIHFWKZFHZASV-UHFFFAOYSA-N 0.000 description 2
- 229940043265 methyl isobutyl ketone Drugs 0.000 description 2
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 2
- DNIAPMSPPWPWGF-UHFFFAOYSA-N monopropylene glycol Natural products CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 2
- XTAZYLNFDRKIHJ-UHFFFAOYSA-N n,n-dioctyloctan-1-amine Chemical compound CCCCCCCCN(CCCCCCCC)CCCCCCCC XTAZYLNFDRKIHJ-UHFFFAOYSA-N 0.000 description 2
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- GNWCSWUWMHQEMD-UHFFFAOYSA-N naphthalene-1,2-dione diazide Chemical group [N-]=[N+]=[N-].[N-]=[N+]=[N-].C1=CC=C2C(=O)C(=O)C=CC2=C1 GNWCSWUWMHQEMD-UHFFFAOYSA-N 0.000 description 2
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- 229920003986 novolac Polymers 0.000 description 2
- RNVCVTLRINQCPJ-UHFFFAOYSA-N o-toluidine Chemical compound CC1=CC=CC=C1N RNVCVTLRINQCPJ-UHFFFAOYSA-N 0.000 description 2
- SJWFXCIHNDVPSH-UHFFFAOYSA-N octan-2-ol Chemical compound CCCCCCC(C)O SJWFXCIHNDVPSH-UHFFFAOYSA-N 0.000 description 2
- NMRPBPVERJPACX-UHFFFAOYSA-N octan-3-ol Chemical compound CCCCCC(O)CC NMRPBPVERJPACX-UHFFFAOYSA-N 0.000 description 2
- 125000005447 octyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 2
- 125000004430 oxygen atom Chemical group O* 0.000 description 2
- ZRSNZINYAWTAHE-UHFFFAOYSA-N p-methoxybenzaldehyde Chemical compound COC1=CC=C(C=O)C=C1 ZRSNZINYAWTAHE-UHFFFAOYSA-N 0.000 description 2
- RZXMPPFPUUCRFN-UHFFFAOYSA-N p-toluidine Chemical compound CC1=CC=C(N)C=C1 RZXMPPFPUUCRFN-UHFFFAOYSA-N 0.000 description 2
- HGBOYTHUEUWSSQ-UHFFFAOYSA-N pentanal Chemical compound CCCCC=O HGBOYTHUEUWSSQ-UHFFFAOYSA-N 0.000 description 2
- YNPNZTXNASCQKK-UHFFFAOYSA-N phenanthrene Chemical compound C1=CC=C2C3=CC=CC=C3C=CC2=C1 YNPNZTXNASCQKK-UHFFFAOYSA-N 0.000 description 2
- ACVYVLVWPXVTIT-UHFFFAOYSA-N phosphinic acid Chemical compound O[PH2]=O ACVYVLVWPXVTIT-UHFFFAOYSA-N 0.000 description 2
- 229920002120 photoresistant polymer Polymers 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000001294 propane Substances 0.000 description 2
- KRIOVPPHQSLHCZ-UHFFFAOYSA-N propiophenone Chemical compound CCC(=O)C1=CC=CC=C1 KRIOVPPHQSLHCZ-UHFFFAOYSA-N 0.000 description 2
- YKYONYBAUNKHLG-UHFFFAOYSA-N propyl acetate Chemical compound CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 description 2
- KIDHWZJUCRJVML-UHFFFAOYSA-N putrescine Chemical compound NCCCCN KIDHWZJUCRJVML-UHFFFAOYSA-N 0.000 description 2
- 229940079877 pyrogallol Drugs 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- KZNICNPSHKQLFF-UHFFFAOYSA-N succinimide Chemical compound O=C1CCC(=O)N1 KZNICNPSHKQLFF-UHFFFAOYSA-N 0.000 description 2
- 230000008961 swelling Effects 0.000 description 2
- 239000010409 thin film Substances 0.000 description 2
- 125000003944 tolyl group Chemical group 0.000 description 2
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 description 2
- WLOQLWBIJZDHET-UHFFFAOYSA-N triphenylsulfonium Chemical compound C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 WLOQLWBIJZDHET-UHFFFAOYSA-N 0.000 description 2
- 239000012953 triphenylsulfonium Substances 0.000 description 2
- FAYMLNNRGCYLSR-UHFFFAOYSA-M triphenylsulfonium triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 FAYMLNNRGCYLSR-UHFFFAOYSA-M 0.000 description 2
- 150000003672 ureas Chemical class 0.000 description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 2
- SNICXCGAKADSCV-JTQLQIEISA-N (-)-Nicotine Chemical compound CN1CCC[C@H]1C1=CC=CN=C1 SNICXCGAKADSCV-JTQLQIEISA-N 0.000 description 1
- GYXAHUXQRATWDV-UHFFFAOYSA-N (1,3-dioxoisoindol-2-yl) trifluoromethanesulfonate Chemical compound C1=CC=C2C(=O)N(OS(=O)(=O)C(F)(F)F)C(=O)C2=C1 GYXAHUXQRATWDV-UHFFFAOYSA-N 0.000 description 1
- RLLFCCPTQOZGOL-UHFFFAOYSA-N (2,5-dioxo-3,4-diphenylpyrrol-1-yl) trifluoromethanesulfonate Chemical compound O=C1N(OS(=O)(=O)C(F)(F)F)C(=O)C(C=2C=CC=CC=2)=C1C1=CC=CC=C1 RLLFCCPTQOZGOL-UHFFFAOYSA-N 0.000 description 1
- OKRLWHAZMUFONP-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) trifluoromethanesulfonate Chemical compound FC(F)(F)S(=O)(=O)ON1C(=O)CCC1=O OKRLWHAZMUFONP-UHFFFAOYSA-N 0.000 description 1
- SRPYWHQMGQUUFF-UHFFFAOYSA-M (2-methylphenyl)-diphenylsulfanium;1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F.CC1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 SRPYWHQMGQUUFF-UHFFFAOYSA-M 0.000 description 1
- PNXSDOXXIOPXPY-UHFFFAOYSA-N (3,5-dioxo-4-azatricyclo[5.2.1.02,6]dec-8-en-4-yl) 1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound C1=CC2CC1C1C2C(=O)N(OS(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F)C1=O PNXSDOXXIOPXPY-UHFFFAOYSA-N 0.000 description 1
- YSLWIRRLTICCEK-UHFFFAOYSA-N (3,5-dioxo-4-azatricyclo[5.2.1.02,6]dec-8-en-4-yl) 2-(trifluoromethyl)benzenesulfonate Chemical compound FC(F)(F)C1=CC=CC=C1S(=O)(=O)ON1C(=O)C2C(C=C3)CC3C2C1=O YSLWIRRLTICCEK-UHFFFAOYSA-N 0.000 description 1
- YNTRIOUAKPQPDY-UHFFFAOYSA-N (3,5-dioxo-4-azatricyclo[5.2.1.02,6]dec-8-en-4-yl) naphthalene-1-sulfonate Chemical compound C1=CC=C2C(S(=O)(=O)ON3C(=O)C4C5CC(C=C5)C4C3=O)=CC=CC2=C1 YNTRIOUAKPQPDY-UHFFFAOYSA-N 0.000 description 1
- JHIGBMVXTRJDTO-UHFFFAOYSA-N (4-hydroxyphenyl)-diphenylsulfanium;1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F.C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 JHIGBMVXTRJDTO-UHFFFAOYSA-N 0.000 description 1
- JILJYTPDQRWDGS-UHFFFAOYSA-N (4-hydroxyphenyl)-diphenylsulfanium;4-methylbenzenesulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 JILJYTPDQRWDGS-UHFFFAOYSA-N 0.000 description 1
- KLBKAFGZQZNLTE-UHFFFAOYSA-N (4-hydroxyphenyl)-diphenylsulfanium;acetate Chemical compound CC([O-])=O.C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 KLBKAFGZQZNLTE-UHFFFAOYSA-N 0.000 description 1
- OKVVPOOEJLJCGE-UHFFFAOYSA-N (4-hydroxyphenyl)-diphenylsulfanium;hydroxide Chemical compound [OH-].C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 OKVVPOOEJLJCGE-UHFFFAOYSA-N 0.000 description 1
- ADOBQGXDCURFNZ-UHFFFAOYSA-N (4-hydroxyphenyl)-diphenylsulfanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 ADOBQGXDCURFNZ-UHFFFAOYSA-N 0.000 description 1
- AWOATHYNVXCSGP-UHFFFAOYSA-M (4-methylphenyl)-diphenylsulfanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(C)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 AWOATHYNVXCSGP-UHFFFAOYSA-M 0.000 description 1
- VERIUQVZMSUGKB-UHFFFAOYSA-N (4-tert-butylphenyl)-(4-hydroxyphenyl)iodanium;2-carboxyphenolate Chemical compound OC(=O)C1=CC=CC=C1[O-].C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(O)C=C1 VERIUQVZMSUGKB-UHFFFAOYSA-N 0.000 description 1
- PZAICYOSEYLSLW-UHFFFAOYSA-N (4-tert-butylphenyl)-(4-hydroxyphenyl)iodanium;acetate Chemical compound CC([O-])=O.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(O)C=C1 PZAICYOSEYLSLW-UHFFFAOYSA-N 0.000 description 1
- MTPFZIRDMCKEKY-UHFFFAOYSA-N (7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl)methanesulfonate;(4-hydroxyphenyl)-diphenylsulfanium Chemical compound C1CC2(CS([O-])(=O)=O)C(=O)CC1C2(C)C.C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 MTPFZIRDMCKEKY-UHFFFAOYSA-N 0.000 description 1
- HHYVKZVPYXHHCG-UHFFFAOYSA-M (7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl)methanesulfonate;diphenyliodanium Chemical compound C=1C=CC=CC=1[I+]C1=CC=CC=C1.C1CC2(CS([O-])(=O)=O)C(=O)CC1C2(C)C HHYVKZVPYXHHCG-UHFFFAOYSA-M 0.000 description 1
- FJALTVCJBKZXKY-UHFFFAOYSA-M (7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl)methanesulfonate;triphenylsulfanium Chemical compound C1CC2(CS([O-])(=O)=O)C(=O)CC1C2(C)C.C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 FJALTVCJBKZXKY-UHFFFAOYSA-M 0.000 description 1
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 description 1
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 description 1
- 125000006651 (C3-C20) cycloalkyl group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- FANQEIMTCNGAGK-IZZDOVSWSA-N (e)-3-(4-hydroxyphenyl)-1-(4-methylphenyl)prop-2-en-1-one Chemical compound C1=CC(C)=CC=C1C(=O)\C=C\C1=CC=C(O)C=C1 FANQEIMTCNGAGK-IZZDOVSWSA-N 0.000 description 1
- VLLPVDKADBYKLM-UHFFFAOYSA-M 1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate;triphenylsulfanium Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F.C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 VLLPVDKADBYKLM-UHFFFAOYSA-M 0.000 description 1
- AVQQQNCBBIEMEU-UHFFFAOYSA-N 1,1,3,3-tetramethylurea Chemical compound CN(C)C(=O)N(C)C AVQQQNCBBIEMEU-UHFFFAOYSA-N 0.000 description 1
- UDYXMTORTDACTG-UHFFFAOYSA-N 1,1,3-tributylthiourea Chemical compound CCCCNC(=S)N(CCCC)CCCC UDYXMTORTDACTG-UHFFFAOYSA-N 0.000 description 1
- YBBLOADPFWKNGS-UHFFFAOYSA-N 1,1-dimethylurea Chemical compound CN(C)C(N)=O YBBLOADPFWKNGS-UHFFFAOYSA-N 0.000 description 1
- FTTATHOUSOIFOQ-UHFFFAOYSA-N 1,2,3,4,6,7,8,8a-octahydropyrrolo[1,2-a]pyrazine Chemical compound C1NCCN2CCCC21 FTTATHOUSOIFOQ-UHFFFAOYSA-N 0.000 description 1
- OKIRBHVFJGXOIS-UHFFFAOYSA-N 1,2-di(propan-2-yl)benzene Chemical compound CC(C)C1=CC=CC=C1C(C)C OKIRBHVFJGXOIS-UHFFFAOYSA-N 0.000 description 1
- NZUPFZNVGSWLQC-UHFFFAOYSA-N 1,3,5-tris(2,3-dibromopropyl)-1,3,5-triazinane-2,4,6-trione Chemical compound BrCC(Br)CN1C(=O)N(CC(Br)CBr)C(=O)N(CC(Br)CBr)C1=O NZUPFZNVGSWLQC-UHFFFAOYSA-N 0.000 description 1
- CYSGHNMQYZDMIA-UHFFFAOYSA-N 1,3-Dimethyl-2-imidazolidinon Chemical compound CN1CCN(C)C1=O CYSGHNMQYZDMIA-UHFFFAOYSA-N 0.000 description 1
- 229940057054 1,3-dimethylurea Drugs 0.000 description 1
- GWEHVDNNLFDJLR-UHFFFAOYSA-N 1,3-diphenylurea Chemical compound C=1C=CC=CC=1NC(=O)NC1=CC=CC=C1 GWEHVDNNLFDJLR-UHFFFAOYSA-N 0.000 description 1
- FCRYMIBFQMLJPR-UHFFFAOYSA-N 1,4,5,5,6,6-hexafluorocyclohex-2-ene-1-sulfonic acid Chemical compound C1=CC(C(C(C1F)(F)F)(F)F)(F)S(=O)(=O)O FCRYMIBFQMLJPR-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- RSWGJHLUYNHPMX-UHFFFAOYSA-N 1,4a-dimethyl-7-propan-2-yl-2,3,4,4b,5,6,10,10a-octahydrophenanthrene-1-carboxylic acid Chemical compound C12CCC(C(C)C)=CC2=CCC2C1(C)CCCC2(C)C(O)=O RSWGJHLUYNHPMX-UHFFFAOYSA-N 0.000 description 1
- VCHOFVSNWYPAEF-UHFFFAOYSA-N 1-(3-acetylphenyl)ethanone Chemical compound CC(=O)C1=CC=CC(C(C)=O)=C1 VCHOFVSNWYPAEF-UHFFFAOYSA-N 0.000 description 1
- IHDLMQAVIMEALP-UHFFFAOYSA-N 1-(trifluoromethyl)-4-[4-(trifluoromethyl)phenyl]sulfonylsulfonylbenzene Chemical compound C1=CC(C(F)(F)F)=CC=C1S(=O)(=O)S(=O)(=O)C1=CC=C(C(F)(F)F)C=C1 IHDLMQAVIMEALP-UHFFFAOYSA-N 0.000 description 1
- PJLDAUREVSXMAM-UHFFFAOYSA-N 1-[2,4,6-tris(2-hydroxypropan-2-yl)phenyl]ethanone Chemical compound CC(=O)C1=C(C(C)(C)O)C=C(C(C)(C)O)C=C1C(C)(C)O PJLDAUREVSXMAM-UHFFFAOYSA-N 0.000 description 1
- MNEFLNSJCTVLTJ-UHFFFAOYSA-N 1-[3-(2-hydroxypropan-2-yl)phenyl]ethanone Chemical compound CC(=O)C1=CC=CC(C(C)(C)O)=C1 MNEFLNSJCTVLTJ-UHFFFAOYSA-N 0.000 description 1
- CAXHWIHQTUOQTF-UHFFFAOYSA-N 1-[4-(2-hydroxypropan-2-yl)phenyl]-2-methylpropan-1-one Chemical compound CC(C)C(=O)C1=CC=C(C(C)(C)O)C=C1 CAXHWIHQTUOQTF-UHFFFAOYSA-N 0.000 description 1
- WBXYSXOGWGBCLC-UHFFFAOYSA-N 1-[4-(2-hydroxypropan-2-yl)phenyl]butan-1-one Chemical compound CCCC(=O)C1=CC=C(C(C)(C)O)C=C1 WBXYSXOGWGBCLC-UHFFFAOYSA-N 0.000 description 1
- CRBMRKRXIIWEPD-UHFFFAOYSA-N 1-[4-(2-hydroxypropan-2-yl)phenyl]pentan-1-one Chemical compound CCCCC(=O)C1=CC=C(C(C)(C)O)C=C1 CRBMRKRXIIWEPD-UHFFFAOYSA-N 0.000 description 1
- QETBWXBJLGGPLS-UHFFFAOYSA-N 1-[4-(2-hydroxypropan-2-yl)phenyl]propan-1-one Chemical compound CCC(=O)C1=CC=C(C(C)(C)O)C=C1 QETBWXBJLGGPLS-UHFFFAOYSA-N 0.000 description 1
- DNIBBCUEPZXVNA-UHFFFAOYSA-N 1-[4-[2-[4-(1-hydroxypropyl)phenyl]propan-2-yl]phenyl]propan-1-ol Chemical compound C1=CC(C(O)CC)=CC=C1C(C)(C)C1=CC=C(C(O)CC)C=C1 DNIBBCUEPZXVNA-UHFFFAOYSA-N 0.000 description 1
- PMAAFKCNQQQBAA-UHFFFAOYSA-N 1-[4-[2-[4-(1-hydroxypropyl)phenyl]propyl]phenyl]propan-1-ol Chemical compound C1=CC(C(O)CC)=CC=C1CC(C)C1=CC=C(C(O)CC)C=C1 PMAAFKCNQQQBAA-UHFFFAOYSA-N 0.000 description 1
- GLYOFBNLYMTEPS-UHFFFAOYSA-N 1-[diazo(2-methylpropylsulfonyl)methyl]sulfonyl-2-methylpropane Chemical compound CC(C)CS(=O)(=O)C(=[N+]=[N-])S(=O)(=O)CC(C)C GLYOFBNLYMTEPS-UHFFFAOYSA-N 0.000 description 1
- WUYAQJZXAJBVFT-UHFFFAOYSA-N 1-[diazo(propylsulfonyl)methyl]sulfonylpropane Chemical compound CCCS(=O)(=O)C(=[N+]=[N-])S(=O)(=O)CCC WUYAQJZXAJBVFT-UHFFFAOYSA-N 0.000 description 1
- OESYNCIYSBWEQV-UHFFFAOYSA-N 1-[diazo-(2,4-dimethylphenyl)sulfonylmethyl]sulfonyl-2,4-dimethylbenzene Chemical compound CC1=CC(C)=CC=C1S(=O)(=O)C(=[N+]=[N-])S(=O)(=O)C1=CC=C(C)C=C1C OESYNCIYSBWEQV-UHFFFAOYSA-N 0.000 description 1
- GYQQFWWMZYBCIB-UHFFFAOYSA-N 1-[diazo-(4-methylphenyl)sulfonylmethyl]sulfonyl-4-methylbenzene Chemical compound C1=CC(C)=CC=C1S(=O)(=O)C(=[N+]=[N-])S(=O)(=O)C1=CC=C(C)C=C1 GYQQFWWMZYBCIB-UHFFFAOYSA-N 0.000 description 1
- LIPRQQHINVWJCH-UHFFFAOYSA-N 1-ethoxypropan-2-yl acetate Chemical compound CCOCC(C)OC(C)=O LIPRQQHINVWJCH-UHFFFAOYSA-N 0.000 description 1
- YMBZYYIZTDQCOY-UHFFFAOYSA-N 1-fluoro-2-(2-fluorophenyl)sulfonylsulfonylbenzene Chemical compound FC1=CC=CC=C1S(=O)(=O)S(=O)(=O)C1=CC=CC=C1F YMBZYYIZTDQCOY-UHFFFAOYSA-N 0.000 description 1
- AOXKCCRPCGPYLO-UHFFFAOYSA-N 1-fluoro-4-(4-fluorophenyl)sulfonylsulfonylbenzene Chemical compound C1=CC(F)=CC=C1S(=O)(=O)S(=O)(=O)C1=CC=C(F)C=C1 AOXKCCRPCGPYLO-UHFFFAOYSA-N 0.000 description 1
- BMVXCPBXGZKUPN-UHFFFAOYSA-N 1-hexanamine Chemical compound CCCCCCN BMVXCPBXGZKUPN-UHFFFAOYSA-N 0.000 description 1
- KUXDDVUXCGCYFX-UHFFFAOYSA-N 1-methyl-4-(4-methylphenyl)sulfonylsulfonylbenzene Chemical compound C1=CC(C)=CC=C1S(=O)(=O)S(=O)(=O)C1=CC=C(C)C=C1 KUXDDVUXCGCYFX-UHFFFAOYSA-N 0.000 description 1
- SQAINHDHICKHLX-UHFFFAOYSA-N 1-naphthaldehyde Chemical compound C1=CC=C2C(C=O)=CC=CC2=C1 SQAINHDHICKHLX-UHFFFAOYSA-N 0.000 description 1
- QQLIGMASAVJVON-UHFFFAOYSA-N 1-naphthalen-1-ylethanone Chemical compound C1=CC=C2C(C(=O)C)=CC=CC2=C1 QQLIGMASAVJVON-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- DMFAHCVITRDZQB-UHFFFAOYSA-N 1-propoxypropan-2-yl acetate Chemical compound CCCOCC(C)OC(C)=O DMFAHCVITRDZQB-UHFFFAOYSA-N 0.000 description 1
- SZAPBCMXGPYERU-UHFFFAOYSA-N 1-tert-butyl-4-(4-tert-butylphenyl)sulfonylsulfonylbenzene Chemical compound C1=CC(C(C)(C)C)=CC=C1S(=O)(=O)S(=O)(=O)C1=CC=C(C(C)(C)C)C=C1 SZAPBCMXGPYERU-UHFFFAOYSA-N 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- UIQGEWJEWJMQSL-UHFFFAOYSA-N 2,2,4,4-tetramethylpentan-3-one Chemical compound CC(C)(C)C(=O)C(C)(C)C UIQGEWJEWJMQSL-UHFFFAOYSA-N 0.000 description 1
- FJJYHTVHBVXEEQ-UHFFFAOYSA-N 2,2-dimethylpropanal Chemical compound CC(C)(C)C=O FJJYHTVHBVXEEQ-UHFFFAOYSA-N 0.000 description 1
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 1
- CRPNQSVBEWWHIJ-UHFFFAOYSA-N 2,3,4-trihydroxybenzaldehyde Chemical compound OC1=CC=C(C=O)C(O)=C1O CRPNQSVBEWWHIJ-UHFFFAOYSA-N 0.000 description 1
- GCFXUDDVXNOPLM-UHFFFAOYSA-N 2,3-diethylbenzaldehyde Chemical compound CCC1=CC=CC(C=O)=C1CC GCFXUDDVXNOPLM-UHFFFAOYSA-N 0.000 description 1
- IXWOUPGDGMCKGT-UHFFFAOYSA-N 2,3-dihydroxybenzaldehyde Chemical compound OC1=CC=CC(C=O)=C1O IXWOUPGDGMCKGT-UHFFFAOYSA-N 0.000 description 1
- UIFVCPMLQXKEEU-UHFFFAOYSA-N 2,3-dimethylbenzaldehyde Chemical compound CC1=CC=CC(C=O)=C1C UIFVCPMLQXKEEU-UHFFFAOYSA-N 0.000 description 1
- BVBUVHZVGAXBJX-UHFFFAOYSA-N 2,4,6-tris(2,3-dibromopropyl)-1,3,5-triazine Chemical compound BrCC(Br)CC1=NC(CC(Br)CBr)=NC(CC(Br)CBr)=N1 BVBUVHZVGAXBJX-UHFFFAOYSA-N 0.000 description 1
- VYAFSOWKHGTKOE-UHFFFAOYSA-N 2,4,6-tris(2-hydroxypropan-2-yl)phenol Chemical compound CC(C)(O)C1=CC(C(C)(C)O)=C(O)C(C(C)(C)O)=C1 VYAFSOWKHGTKOE-UHFFFAOYSA-N 0.000 description 1
- ZCIVTTVWBMTBTH-UHFFFAOYSA-M 2,4-difluorobenzenesulfonate;diphenyl-(2,4,6-trimethylphenyl)sulfanium Chemical compound [O-]S(=O)(=O)C1=CC=C(F)C=C1F.CC1=CC(C)=CC(C)=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 ZCIVTTVWBMTBTH-UHFFFAOYSA-M 0.000 description 1
- YIZDIDDKUUSNHM-UHFFFAOYSA-M 2,4-difluorobenzenesulfonate;diphenyliodanium Chemical compound [O-]S(=O)(=O)C1=CC=C(F)C=C1F.C=1C=CC=CC=1[I+]C1=CC=CC=C1 YIZDIDDKUUSNHM-UHFFFAOYSA-M 0.000 description 1
- WHNFHFXJESETFD-UHFFFAOYSA-N 2-(2,5-dimethylfuran-3-yl)propan-2-ol Chemical compound CC1=CC(C(C)(C)O)=C(C)O1 WHNFHFXJESETFD-UHFFFAOYSA-N 0.000 description 1
- ZWAFTLGUMWLXJD-UHFFFAOYSA-N 2-(2-hydroxypropan-2-yl)phenol Chemical compound CC(C)(O)C1=CC=CC=C1O ZWAFTLGUMWLXJD-UHFFFAOYSA-N 0.000 description 1
- IJVFFYRZMIELJB-UHFFFAOYSA-N 2-(2-hydroxypropanoyl)benzaldehyde Chemical compound CC(O)C(=O)C1=CC=CC=C1C=O IJVFFYRZMIELJB-UHFFFAOYSA-N 0.000 description 1
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 1
- VRLCJQCQBUKRDH-UHFFFAOYSA-N 2-(2-methylfuran-3-yl)propan-2-ol Chemical compound CC=1OC=CC=1C(C)(C)O VRLCJQCQBUKRDH-UHFFFAOYSA-N 0.000 description 1
- ZFUFIXXMJTYOSK-UHFFFAOYSA-N 2-(3-phenylphenyl)propan-2-ol Chemical group CC(C)(O)C1=CC=CC(C=2C=CC=CC=2)=C1 ZFUFIXXMJTYOSK-UHFFFAOYSA-N 0.000 description 1
- GOKGIYHIVSGXDM-UHFFFAOYSA-N 2-(4-phenylphenyl)propan-2-ol Chemical group C1=CC(C(C)(O)C)=CC=C1C1=CC=CC=C1 GOKGIYHIVSGXDM-UHFFFAOYSA-N 0.000 description 1
- WUZAVESBWDKBBS-UHFFFAOYSA-N 2-(5-butylfuran-3-yl)propan-2-ol Chemical compound CCCCC1=CC(C(C)(C)O)=CO1 WUZAVESBWDKBBS-UHFFFAOYSA-N 0.000 description 1
- UKLGAAHFEQCVEI-UHFFFAOYSA-N 2-(5-ethylfuran-3-yl)propan-2-ol Chemical compound CCC1=CC(C(C)(C)O)=CO1 UKLGAAHFEQCVEI-UHFFFAOYSA-N 0.000 description 1
- OMFVZARETAOHFK-UHFFFAOYSA-N 2-(5-methylfuran-3-yl)propan-2-ol Chemical compound CC1=CC(C(C)(C)O)=CO1 OMFVZARETAOHFK-UHFFFAOYSA-N 0.000 description 1
- XHPCCIGGHMTJFP-UHFFFAOYSA-N 2-(5-pentylfuran-3-yl)propan-2-ol Chemical compound CCCCCC1=CC(C(C)(C)O)=CO1 XHPCCIGGHMTJFP-UHFFFAOYSA-N 0.000 description 1
- NUXMZJVXDWBZJZ-UHFFFAOYSA-N 2-(5-propylfuran-3-yl)propan-2-ol Chemical compound CCCC1=CC(C(C)(C)O)=CO1 NUXMZJVXDWBZJZ-UHFFFAOYSA-N 0.000 description 1
- VTPCOJUXLMYPKF-UHFFFAOYSA-N 2-(5-tert-butylfuran-3-yl)propan-2-ol Chemical compound CC(C)(C)C1=CC(C(C)(C)O)=CO1 VTPCOJUXLMYPKF-UHFFFAOYSA-N 0.000 description 1
- DGPBVJWCIDNDPN-UHFFFAOYSA-N 2-(dimethylamino)benzaldehyde Chemical compound CN(C)C1=CC=CC=C1C=O DGPBVJWCIDNDPN-UHFFFAOYSA-N 0.000 description 1
- SAKBHTQAACRFAO-UHFFFAOYSA-N 2-(furan-3-yl)propan-2-ol Chemical compound CC(C)(O)C=1C=COC=1 SAKBHTQAACRFAO-UHFFFAOYSA-N 0.000 description 1
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- IZGZXJUFKVOAGF-UHFFFAOYSA-N 2-[2,5-diethyl-4-(2-hydroxypropan-2-yl)furan-3-yl]propan-2-ol Chemical compound CCC=1OC(CC)=C(C(C)(C)O)C=1C(C)(C)O IZGZXJUFKVOAGF-UHFFFAOYSA-N 0.000 description 1
- DRZOOMANMBUNIG-UHFFFAOYSA-N 2-[3,4-bis(2-hydroxypropan-2-yl)phenyl]propan-2-ol Chemical compound CC(C)(O)C1=CC=C(C(C)(C)O)C(C(C)(C)O)=C1 DRZOOMANMBUNIG-UHFFFAOYSA-N 0.000 description 1
- FMCWGKXGRNQNLD-UHFFFAOYSA-N 2-[3,5-bis(2-hydroxypropan-2-yl)phenyl]propan-2-ol Chemical compound CC(C)(O)C1=CC(C(C)(C)O)=CC(C(C)(C)O)=C1 FMCWGKXGRNQNLD-UHFFFAOYSA-N 0.000 description 1
- LIGFYCSWGHMRHH-UHFFFAOYSA-N 2-[3-(2-hydroxypropan-2-yl)-5-[2,4,6-tris(2-hydroxypropan-2-yl)phenyl]phenyl]propan-2-ol Chemical group CC(O)(C)C1=CC(C(C)(O)C)=CC(C=2C(=CC(=CC=2C(C)(C)O)C(C)(C)O)C(C)(C)O)=C1 LIGFYCSWGHMRHH-UHFFFAOYSA-N 0.000 description 1
- IJUBXXQWGRHQFZ-UHFFFAOYSA-N 2-[3-(2-hydroxypropan-2-yl)-5-phenylphenyl]propan-2-ol Chemical group CC(O)(C)C1=CC(C(C)(O)C)=CC(C=2C=CC=CC=2)=C1 IJUBXXQWGRHQFZ-UHFFFAOYSA-N 0.000 description 1
- UGPWRRVOLLMHSC-UHFFFAOYSA-N 2-[3-(2-hydroxypropan-2-yl)phenyl]propan-2-ol Chemical compound CC(C)(O)C1=CC=CC(C(C)(C)O)=C1 UGPWRRVOLLMHSC-UHFFFAOYSA-N 0.000 description 1
- JZINQZGJUFGIQP-UHFFFAOYSA-N 2-[3-[3,5-bis(2-hydroxypropan-2-yl)phenyl]-5-(2-hydroxypropan-2-yl)phenyl]propan-2-ol Chemical group CC(O)(C)C1=CC(C(C)(O)C)=CC(C=2C=C(C=C(C=2)C(C)(C)O)C(C)(C)O)=C1 JZINQZGJUFGIQP-UHFFFAOYSA-N 0.000 description 1
- GERCGKZLCZJRPZ-UHFFFAOYSA-N 2-[3-[3,5-bis(2-hydroxypropan-2-yl)phenyl]phenyl]propan-2-ol Chemical group CC(C)(O)C1=CC=CC(C=2C=C(C=C(C=2)C(C)(C)O)C(C)(C)O)=C1 GERCGKZLCZJRPZ-UHFFFAOYSA-N 0.000 description 1
- ZUMYWCDSXBCHES-UHFFFAOYSA-N 2-[3-[3-(2-hydroxypropan-2-yl)phenyl]phenyl]propan-2-ol Chemical group CC(C)(O)C1=CC=CC(C=2C=C(C=CC=2)C(C)(C)O)=C1 ZUMYWCDSXBCHES-UHFFFAOYSA-N 0.000 description 1
- JXKZSUBUHCAKRP-UHFFFAOYSA-N 2-[4,6,8-tris(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound C1=C(C(C)(C)O)C=C(C(C)(C)O)C2=CC(C(C)(O)C)=CC(C(C)(C)O)=C21 JXKZSUBUHCAKRP-UHFFFAOYSA-N 0.000 description 1
- DALPWGJPMCACKJ-UHFFFAOYSA-N 2-[4-(2-hydroxypropan-2-yl)-2,5-dimethylfuran-3-yl]propan-2-ol Chemical compound CC=1OC(C)=C(C(C)(C)O)C=1C(C)(C)O DALPWGJPMCACKJ-UHFFFAOYSA-N 0.000 description 1
- VESIXQCUYZXTSB-UHFFFAOYSA-N 2-[4-(2-hydroxypropan-2-yl)furan-3-yl]propan-2-ol Chemical compound CC(C)(O)C1=COC=C1C(C)(C)O VESIXQCUYZXTSB-UHFFFAOYSA-N 0.000 description 1
- ZYOHOMRVLXLMNO-UHFFFAOYSA-N 2-[4-(2-hydroxypropan-2-yl)naphthalen-1-yl]propan-2-ol Chemical compound C1=CC=C2C(C(C)(O)C)=CC=C(C(C)(C)O)C2=C1 ZYOHOMRVLXLMNO-UHFFFAOYSA-N 0.000 description 1
- KJJNSJHNWDNACJ-UHFFFAOYSA-N 2-[4-(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound C1=CC=CC2=CC(C(C)(O)C)=CC(C(C)(C)O)=C21 KJJNSJHNWDNACJ-UHFFFAOYSA-N 0.000 description 1
- DYNINZXZSZCVCL-UHFFFAOYSA-N 2-[4-(2-phenylpropan-2-yl)phenyl]propan-2-ol Chemical compound C1=CC(C(C)(O)C)=CC=C1C(C)(C)C1=CC=CC=C1 DYNINZXZSZCVCL-UHFFFAOYSA-N 0.000 description 1
- MGCSYWCXSNYWMP-UHFFFAOYSA-N 2-[4-(2-phenylpropyl)phenyl]propan-2-ol Chemical compound C=1C=CC=CC=1C(C)CC1=CC=C(C(C)(C)O)C=C1 MGCSYWCXSNYWMP-UHFFFAOYSA-N 0.000 description 1
- ZEORFNBANZTPNW-UHFFFAOYSA-N 2-[4-(4-phenylbutyl)phenyl]propan-2-ol Chemical compound C1=CC(C(C)(O)C)=CC=C1CCCCC1=CC=CC=C1 ZEORFNBANZTPNW-UHFFFAOYSA-N 0.000 description 1
- IMGRVVYGJFZPIW-UHFFFAOYSA-N 2-[4-(5-phenylpentyl)phenyl]propan-2-ol Chemical compound C1=CC(C(C)(O)C)=CC=C1CCCCCC1=CC=CC=C1 IMGRVVYGJFZPIW-UHFFFAOYSA-N 0.000 description 1
- CJLHYUMEBPPLJM-UHFFFAOYSA-N 2-[4-[2-[4-(2-hydroxypropan-2-yl)phenyl]ethyl]phenyl]propan-2-ol Chemical compound C1=CC(C(C)(O)C)=CC=C1CCC1=CC=C(C(C)(C)O)C=C1 CJLHYUMEBPPLJM-UHFFFAOYSA-N 0.000 description 1
- YLJNKKVNLJYTND-UHFFFAOYSA-N 2-[4-[3,5-bis(2-hydroxypropan-2-yl)phenyl]phenyl]propan-2-ol Chemical group C1=CC(C(C)(O)C)=CC=C1C1=CC(C(C)(C)O)=CC(C(C)(C)O)=C1 YLJNKKVNLJYTND-UHFFFAOYSA-N 0.000 description 1
- OINGAZBKDCNUHE-UHFFFAOYSA-N 2-[4-[3-(2-hydroxypropan-2-yl)phenyl]phenyl]propan-2-ol Chemical group C1=CC(C(C)(O)C)=CC=C1C1=CC=CC(C(C)(C)O)=C1 OINGAZBKDCNUHE-UHFFFAOYSA-N 0.000 description 1
- BALFTRWHVULYEO-UHFFFAOYSA-N 2-[4-[4-(2-hydroxypropan-2-yl)phenyl]phenyl]propan-2-ol Chemical group C1=CC(C(C)(O)C)=CC=C1C1=CC=C(C(C)(C)O)C=C1 BALFTRWHVULYEO-UHFFFAOYSA-N 0.000 description 1
- LPUYIAIROCCWHR-UHFFFAOYSA-N 2-[5,7-bis(2-hydroxypropan-2-yl)naphthalen-1-yl]propan-2-ol Chemical compound C1=CC=C(C(C)(C)O)C2=CC(C(C)(O)C)=CC(C(C)(C)O)=C21 LPUYIAIROCCWHR-UHFFFAOYSA-N 0.000 description 1
- YBSFNIINXGWDLP-UHFFFAOYSA-N 2-[5,7-bis(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound CC(O)(C)C1=CC(C(C)(C)O)=CC2=CC(C(C)(O)C)=CC=C21 YBSFNIINXGWDLP-UHFFFAOYSA-N 0.000 description 1
- QZIRPTFOJIXAPH-UHFFFAOYSA-N 2-[5-(2-hydroxypropan-2-yl)naphthalen-1-yl]propan-2-ol Chemical compound C1=CC=C2C(C(C)(O)C)=CC=CC2=C1C(C)(C)O QZIRPTFOJIXAPH-UHFFFAOYSA-N 0.000 description 1
- GJFIANXAVJSVMH-UHFFFAOYSA-N 2-[5-(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound CC(O)(C)C1=CC=CC2=CC(C(C)(O)C)=CC=C21 GJFIANXAVJSVMH-UHFFFAOYSA-N 0.000 description 1
- ZNUKBUMMPZZCLU-UHFFFAOYSA-N 2-[6,8-bis(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound C1=C(C(C)(C)O)C=C(C(C)(C)O)C2=CC(C(C)(O)C)=CC=C21 ZNUKBUMMPZZCLU-UHFFFAOYSA-N 0.000 description 1
- DXIUSVDANCIIAR-UHFFFAOYSA-N 2-[6-(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound C1=C(C(C)(C)O)C=CC2=CC(C(C)(O)C)=CC=C21 DXIUSVDANCIIAR-UHFFFAOYSA-N 0.000 description 1
- COVOHNBLFUXYNP-UHFFFAOYSA-N 2-[7-(2-hydroxypropan-2-yl)naphthalen-2-yl]propan-2-ol Chemical compound C1=CC(C(C)(C)O)=CC2=CC(C(C)(O)C)=CC=C21 COVOHNBLFUXYNP-UHFFFAOYSA-N 0.000 description 1
- LJBWJFWNFUKAGS-UHFFFAOYSA-N 2-[bis(2-hydroxyphenyl)methyl]phenol Chemical compound OC1=CC=CC=C1C(C=1C(=CC=CC=1)O)C1=CC=CC=C1O LJBWJFWNFUKAGS-UHFFFAOYSA-N 0.000 description 1
- SAFWZKVQMVOANB-UHFFFAOYSA-N 2-[tert-butylsulfonyl(diazo)methyl]sulfonyl-2-methylpropane Chemical compound CC(C)(C)S(=O)(=O)C(=[N+]=[N-])S(=O)(=O)C(C)(C)C SAFWZKVQMVOANB-UHFFFAOYSA-N 0.000 description 1
- NDOPHXWIAZIXPR-UHFFFAOYSA-N 2-bromobenzaldehyde Chemical compound BrC1=CC=CC=C1C=O NDOPHXWIAZIXPR-UHFFFAOYSA-N 0.000 description 1
- NQBXSWAWVZHKBZ-UHFFFAOYSA-N 2-butoxyethyl acetate Chemical compound CCCCOCCOC(C)=O NQBXSWAWVZHKBZ-UHFFFAOYSA-N 0.000 description 1
- WKTKKWVPHISOFP-UHFFFAOYSA-N 2-carboxyphenolate;(4-hydroxyphenyl)-diphenylsulfanium Chemical compound OC(=O)C1=CC=CC=C1[O-].C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 WKTKKWVPHISOFP-UHFFFAOYSA-N 0.000 description 1
- KKLIEUWPBXKNFS-UHFFFAOYSA-M 2-carboxyphenolate;triphenylsulfanium Chemical compound OC1=CC=CC=C1C([O-])=O.C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 KKLIEUWPBXKNFS-UHFFFAOYSA-M 0.000 description 1
- OXJFDUURJXWYBN-UHFFFAOYSA-N 2-cyclobutylbenzaldehyde Chemical compound O=CC1=CC=CC=C1C1CCC1 OXJFDUURJXWYBN-UHFFFAOYSA-N 0.000 description 1
- SKRSNLWVIPLNRR-UHFFFAOYSA-N 2-cyclopentylbenzaldehyde Chemical compound O=CC1=CC=CC=C1C1CCCC1 SKRSNLWVIPLNRR-UHFFFAOYSA-N 0.000 description 1
- ZVDXHSUCDWVQEY-UHFFFAOYSA-N 2-cyclopropylbenzaldehyde Chemical compound O=CC1=CC=CC=C1C1CC1 ZVDXHSUCDWVQEY-UHFFFAOYSA-N 0.000 description 1
- NTWBHJYRDKBGBR-UHFFFAOYSA-N 2-ethylbenzaldehyde Chemical compound CCC1=CC=CC=C1C=O NTWBHJYRDKBGBR-UHFFFAOYSA-N 0.000 description 1
- NRGGMCIBEHEAIL-UHFFFAOYSA-N 2-ethylpyridine Chemical compound CCC1=CC=CC=N1 NRGGMCIBEHEAIL-UHFFFAOYSA-N 0.000 description 1
- 125000004200 2-methoxyethyl group Chemical group [H]C([H])([H])OC([H])([H])C([H])([H])* 0.000 description 1
- IGTUUFWQCPWFCF-UHFFFAOYSA-N 2-methyl-1,1-diphenylpropan-2-ol Chemical compound C=1C=CC=CC=1C(C(C)(O)C)C1=CC=CC=C1 IGTUUFWQCPWFCF-UHFFFAOYSA-N 0.000 description 1
- ZOJKUHSSLKZYIO-UHFFFAOYSA-N 2-methyl-3-propan-2-ylbenzaldehyde Chemical compound CC(C)C1=CC=CC(C=O)=C1C ZOJKUHSSLKZYIO-UHFFFAOYSA-N 0.000 description 1
- CRWNQZTZTZWPOF-UHFFFAOYSA-N 2-methyl-4-phenylpyridine Chemical compound C1=NC(C)=CC(C=2C=CC=CC=2)=C1 CRWNQZTZTZWPOF-UHFFFAOYSA-N 0.000 description 1
- NXKOSHBFVWYVIH-UHFFFAOYSA-N 2-n-(butoxymethyl)-1,3,5-triazine-2,4,6-triamine Chemical compound CCCCOCNC1=NC(N)=NC(N)=N1 NXKOSHBFVWYVIH-UHFFFAOYSA-N 0.000 description 1
- XLPVLPWLUVFRGN-UHFFFAOYSA-N 2-n-(ethoxymethyl)-1,3,5-triazine-2,4,6-triamine Chemical compound CCOCNC1=NC(N)=NC(N)=N1 XLPVLPWLUVFRGN-UHFFFAOYSA-N 0.000 description 1
- LQRDVHKFQFSRFZ-UHFFFAOYSA-N 2-n-(propan-2-yloxymethyl)-1,3,5-triazine-2,4,6-triamine Chemical compound CC(C)OCNC1=NC(N)=NC(N)=N1 LQRDVHKFQFSRFZ-UHFFFAOYSA-N 0.000 description 1
- ZQAXBTFXHKKQHN-UHFFFAOYSA-N 2-n-(propoxymethyl)-1,3,5-triazine-2,4,6-triamine Chemical compound CCCOCNC1=NC(N)=NC(N)=N1 ZQAXBTFXHKKQHN-UHFFFAOYSA-N 0.000 description 1
- JSQYKOSZQFTNSN-UHFFFAOYSA-N 2-n-[(2-methylpropan-2-yl)oxymethyl]-1,3,5-triazine-2,4,6-triamine Chemical compound CC(C)(C)OCNC1=NC(N)=NC(N)=N1 JSQYKOSZQFTNSN-UHFFFAOYSA-N 0.000 description 1
- IBENYRAUOZMVRG-UHFFFAOYSA-N 2-naphthalen-1-ylpropan-2-ol Chemical compound C1=CC=C2C(C(C)(O)C)=CC=CC2=C1 IBENYRAUOZMVRG-UHFFFAOYSA-N 0.000 description 1
- CSONNHKIVVSLQQ-UHFFFAOYSA-N 2-naphthalen-2-ylpropan-2-ol Chemical compound C1=CC=CC2=CC(C(C)(O)C)=CC=C21 CSONNHKIVVSLQQ-UHFFFAOYSA-N 0.000 description 1
- DWYHDSLIWMUSOO-UHFFFAOYSA-N 2-phenyl-1h-benzimidazole Chemical compound C1=CC=CC=C1C1=NC2=CC=CC=C2N1 DWYHDSLIWMUSOO-UHFFFAOYSA-N 0.000 description 1
- XLLXMBCBJGATSP-UHFFFAOYSA-N 2-phenylethenol Chemical compound OC=CC1=CC=CC=C1 XLLXMBCBJGATSP-UHFFFAOYSA-N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- BDCFWIDZNLCTMF-UHFFFAOYSA-N 2-phenylpropan-2-ol Chemical compound CC(C)(O)C1=CC=CC=C1 BDCFWIDZNLCTMF-UHFFFAOYSA-N 0.000 description 1
- QMAQLCVJIYANPZ-UHFFFAOYSA-N 2-propoxyethyl acetate Chemical compound CCCOCCOC(C)=O QMAQLCVJIYANPZ-UHFFFAOYSA-N 0.000 description 1
- PANBRUWVURLWGY-UHFFFAOYSA-N 2-undecenal Chemical compound CCCCCCCCC=CC=O PANBRUWVURLWGY-UHFFFAOYSA-N 0.000 description 1
- WADCPEMKIBAJHH-UHFFFAOYSA-N 3,4-diphenylpyrrole-2,5-dione Chemical compound O=C1NC(=O)C(C=2C=CC=CC=2)=C1C1=CC=CC=C1 WADCPEMKIBAJHH-UHFFFAOYSA-N 0.000 description 1
- CQXZJTSSVQUFEB-UHFFFAOYSA-N 3,5-bis(2-hydroxypropan-2-yl)phenol Chemical compound CC(C)(O)C1=CC(O)=CC(C(C)(C)O)=C1 CQXZJTSSVQUFEB-UHFFFAOYSA-N 0.000 description 1
- ODJJXKURUDQRLO-UHFFFAOYSA-N 3-(2-hydroxypropan-2-yl)benzoic acid Chemical compound CC(C)(O)C1=CC=CC(C(O)=O)=C1 ODJJXKURUDQRLO-UHFFFAOYSA-N 0.000 description 1
- KNCICOZTHWGRMY-UHFFFAOYSA-N 3-(2-hydroxypropan-2-yl)phenol Chemical compound CC(C)(O)C1=CC=CC(O)=C1 KNCICOZTHWGRMY-UHFFFAOYSA-N 0.000 description 1
- PYSRRFNXTXNWCD-UHFFFAOYSA-N 3-(2-phenylethenyl)furan-2,5-dione Chemical compound O=C1OC(=O)C(C=CC=2C=CC=CC=2)=C1 PYSRRFNXTXNWCD-UHFFFAOYSA-N 0.000 description 1
- NMRPBPVERJPACX-QMMMGPOBSA-N 3-Octanol Natural products CCCCC[C@@H](O)CC NMRPBPVERJPACX-QMMMGPOBSA-N 0.000 description 1
- HIYRIYOUSQLJHP-UHFFFAOYSA-N 3-[2-(4-aminophenyl)propan-2-yl]aniline Chemical compound C=1C=CC(N)=CC=1C(C)(C)C1=CC=C(N)C=C1 HIYRIYOUSQLJHP-UHFFFAOYSA-N 0.000 description 1
- COPUOMGHQGSBQO-UHFFFAOYSA-N 3-[2-(4-aminophenyl)propan-2-yl]phenol Chemical compound C=1C=CC(O)=CC=1C(C)(C)C1=CC=C(N)C=C1 COPUOMGHQGSBQO-UHFFFAOYSA-N 0.000 description 1
- QHRSPPDKVWGPQP-UHFFFAOYSA-N 3-ethyl-2-methylbenzaldehyde Chemical compound CCC1=CC=CC(C=O)=C1C QHRSPPDKVWGPQP-UHFFFAOYSA-N 0.000 description 1
- NMUMFCGQLRQGCR-UHFFFAOYSA-N 3-methoxypentyl acetate Chemical compound CCC(OC)CCOC(C)=O NMUMFCGQLRQGCR-UHFFFAOYSA-N 0.000 description 1
- TWIHWYXRPSNKHJ-UHFFFAOYSA-N 3-methyl-2-pyridin-2-ylpyridine Chemical group CC1=CC=CN=C1C1=CC=CC=N1 TWIHWYXRPSNKHJ-UHFFFAOYSA-N 0.000 description 1
- YGHRJJRRZDOVPD-UHFFFAOYSA-N 3-methylbutanal Chemical compound CC(C)CC=O YGHRJJRRZDOVPD-UHFFFAOYSA-N 0.000 description 1
- GPIUUMROPXDNRH-UHFFFAOYSA-N 3647-74-3 Chemical compound C1C2C3C(=O)NC(=O)C3C1C=C2 GPIUUMROPXDNRH-UHFFFAOYSA-N 0.000 description 1
- YBRVSVVVWCFQMG-UHFFFAOYSA-N 4,4'-diaminodiphenylmethane Chemical compound C1=CC(N)=CC=C1CC1=CC=C(N)C=C1 YBRVSVVVWCFQMG-UHFFFAOYSA-N 0.000 description 1
- PMZXHWXCHFTBFZ-UHFFFAOYSA-N 4-(2-hydroxypropan-2-yl)phenol Chemical class CC(C)(O)C1=CC=C(O)C=C1 PMZXHWXCHFTBFZ-UHFFFAOYSA-N 0.000 description 1
- JISGCUGKPYJJID-UHFFFAOYSA-N 4-(3-hydroxynaphthalen-1-yl)sulfonylsulfonylnaphthalen-2-ol Chemical compound C1=CC=C2C(S(=O)(=O)S(=O)(=O)C=3C=C(C=C4C=CC=CC4=3)O)=CC(O)=CC2=C1 JISGCUGKPYJJID-UHFFFAOYSA-N 0.000 description 1
- FAZZFXWVHOPPPD-UHFFFAOYSA-N 4-(4-hydroxyphenyl)sulfonylsulfonylphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)S(=O)(=O)C1=CC=C(O)C=C1 FAZZFXWVHOPPPD-UHFFFAOYSA-N 0.000 description 1
- HLBLWEWZXPIGSM-UHFFFAOYSA-N 4-Aminophenyl ether Chemical compound C1=CC(N)=CC=C1OC1=CC=C(N)C=C1 HLBLWEWZXPIGSM-UHFFFAOYSA-N 0.000 description 1
- ZYEDGEXYGKWJPB-UHFFFAOYSA-N 4-[2-(4-aminophenyl)propan-2-yl]aniline Chemical compound C=1C=C(N)C=CC=1C(C)(C)C1=CC=C(N)C=C1 ZYEDGEXYGKWJPB-UHFFFAOYSA-N 0.000 description 1
- NFGPNZVXBBBZNF-UHFFFAOYSA-N 4-[2-(4-aminophenyl)propan-2-yl]phenol Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(N)C=C1 NFGPNZVXBBBZNF-UHFFFAOYSA-N 0.000 description 1
- HESXPOICBNWMPI-UHFFFAOYSA-N 4-[2-[4-[2-(4-aminophenyl)propan-2-yl]phenyl]propan-2-yl]aniline Chemical compound C=1C=C(C(C)(C)C=2C=CC(N)=CC=2)C=CC=1C(C)(C)C1=CC=C(N)C=C1 HESXPOICBNWMPI-UHFFFAOYSA-N 0.000 description 1
- DLJMSUPTAMKSOB-UHFFFAOYSA-N 4-[diphenyl-(2,4,6-trimethylphenyl)methyl]benzenesulfonic acid Chemical compound CC1=CC(C)=CC(C)=C1C(C=1C=CC(=CC=1)S(O)(=O)=O)(C=1C=CC=CC=1)C1=CC=CC=C1 DLJMSUPTAMKSOB-UHFFFAOYSA-N 0.000 description 1
- ARIREUPIXAKDAY-UHFFFAOYSA-N 4-butylbenzaldehyde Chemical compound CCCCC1=CC=C(C=O)C=C1 ARIREUPIXAKDAY-UHFFFAOYSA-N 0.000 description 1
- KUHNCPCUPOPMDY-UHFFFAOYSA-N 4-cyclohexylbenzaldehyde Chemical group C1=CC(C=O)=CC=C1C1CCCCC1 KUHNCPCUPOPMDY-UHFFFAOYSA-N 0.000 description 1
- VJXRKZJMGVSXPX-UHFFFAOYSA-N 4-ethylpyridine Chemical compound CCC1=CC=NC=C1 VJXRKZJMGVSXPX-UHFFFAOYSA-N 0.000 description 1
- RGHHSNMVTDWUBI-UHFFFAOYSA-N 4-hydroxybenzaldehyde Chemical compound OC1=CC=C(C=O)C=C1 RGHHSNMVTDWUBI-UHFFFAOYSA-N 0.000 description 1
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 1
- GVSNQMFKEPBIOY-UHFFFAOYSA-N 4-methyl-2h-triazole Chemical group CC=1C=NNN=1 GVSNQMFKEPBIOY-UHFFFAOYSA-N 0.000 description 1
- JGEGJYXHCFUMJF-UHFFFAOYSA-N 4-methylpentanal Chemical compound CC(C)CCC=O JGEGJYXHCFUMJF-UHFFFAOYSA-N 0.000 description 1
- 125000000590 4-methylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- QZHXKQKKEBXYRG-UHFFFAOYSA-N 4-n-(4-aminophenyl)benzene-1,4-diamine Chemical compound C1=CC(N)=CC=C1NC1=CC=C(N)C=C1 QZHXKQKKEBXYRG-UHFFFAOYSA-N 0.000 description 1
- TYOXIFXYEIILLY-UHFFFAOYSA-N 5-methyl-2-phenyl-1h-imidazole Chemical compound N1C(C)=CN=C1C1=CC=CC=C1 TYOXIFXYEIILLY-UHFFFAOYSA-N 0.000 description 1
- XZGLNCKSNVGDNX-UHFFFAOYSA-N 5-methyl-2h-tetrazole Chemical group CC=1N=NNN=1 XZGLNCKSNVGDNX-UHFFFAOYSA-N 0.000 description 1
- ZPQAKYPOZRXKFA-UHFFFAOYSA-N 6-Undecanone Chemical compound CCCCCC(=O)CCCCC ZPQAKYPOZRXKFA-UHFFFAOYSA-N 0.000 description 1
- GZVHEAJQGPRDLQ-UHFFFAOYSA-N 6-phenyl-1,3,5-triazine-2,4-diamine Chemical class NC1=NC(N)=NC(C=2C=CC=CC=2)=N1 GZVHEAJQGPRDLQ-UHFFFAOYSA-N 0.000 description 1
- LPEKGGXMPWTOCB-UHFFFAOYSA-N 8beta-(2,3-epoxy-2-methylbutyryloxy)-14-acetoxytithifolin Natural products COC(=O)C(C)O LPEKGGXMPWTOCB-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 102100033806 Alpha-protein kinase 3 Human genes 0.000 description 1
- 101710082399 Alpha-protein kinase 3 Proteins 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 229930185605 Bisphenol Natural products 0.000 description 1
- ZTQSAGDEMFDKMZ-UHFFFAOYSA-N Butyraldehyde Chemical compound CCCC=O ZTQSAGDEMFDKMZ-UHFFFAOYSA-N 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- XPUNRGMNFVNPPI-UHFFFAOYSA-N C1=CC2CC1C1C2C(=O)N(OS(=O)(=O)CCCCCCCC)C1=O Chemical compound C1=CC2CC1C1C2C(=O)N(OS(=O)(=O)CCCCCCCC)C1=O XPUNRGMNFVNPPI-UHFFFAOYSA-N 0.000 description 1
- HSUCTLMTZNDURI-UHFFFAOYSA-N CCCCCCCCCCS(CN=NS(C1CCCCC1)(=O)=O)(=O)=O Chemical compound CCCCCCCCCCS(CN=NS(C1CCCCC1)(=O)=O)(=O)=O HSUCTLMTZNDURI-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- XZMCDFZZKTWFGF-UHFFFAOYSA-N Cyanamide Chemical compound NC#N XZMCDFZZKTWFGF-UHFFFAOYSA-N 0.000 description 1
- 229920003270 Cymel® Polymers 0.000 description 1
- MHZGKXUYDGKKIU-UHFFFAOYSA-N Decylamine Chemical compound CCCCCCCCCCN MHZGKXUYDGKKIU-UHFFFAOYSA-N 0.000 description 1
- YXHKONLOYHBTNS-UHFFFAOYSA-N Diazomethane Chemical compound C=[N+]=[N-] YXHKONLOYHBTNS-UHFFFAOYSA-N 0.000 description 1
- JYFHYPJRHGVZDY-UHFFFAOYSA-N Dibutyl phosphate Chemical compound CCCCOP(O)(=O)OCCCC JYFHYPJRHGVZDY-UHFFFAOYSA-N 0.000 description 1
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical compound C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- ASMQGLCHMVWBQR-UHFFFAOYSA-N Diphenyl phosphate Chemical compound C=1C=CC=CC=1OP(=O)(O)OC1=CC=CC=C1 ASMQGLCHMVWBQR-UHFFFAOYSA-N 0.000 description 1
- IMROMDMJAWUWLK-UHFFFAOYSA-N Ethenol Chemical compound OC=C IMROMDMJAWUWLK-UHFFFAOYSA-N 0.000 description 1
- XXRCUYVCPSWGCC-UHFFFAOYSA-N Ethyl pyruvate Chemical compound CCOC(=O)C(C)=O XXRCUYVCPSWGCC-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- SKXXQLPBGBBRGM-UHFFFAOYSA-N FC1=C(F)C(F)=C(F)C(F)=C1S(=O)(=O)ON1C(=O)C2C(C=C3)CC3C2C1=O Chemical compound FC1=C(F)C(F)=C(F)C(F)=C1S(=O)(=O)ON1C(=O)C2C(C=C3)CC3C2C1=O SKXXQLPBGBBRGM-UHFFFAOYSA-N 0.000 description 1
- 239000007818 Grignard reagent Substances 0.000 description 1
- WJYIASZWHGOTOU-UHFFFAOYSA-N Heptylamine Chemical compound CCCCCCCN WJYIASZWHGOTOU-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 229910021578 Iron(III) chloride Inorganic materials 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- RJUFJBKOKNCXHH-UHFFFAOYSA-N Methyl propionate Chemical compound CCC(=O)OC RJUFJBKOKNCXHH-UHFFFAOYSA-N 0.000 description 1
- WRQNANDWMGAFTP-UHFFFAOYSA-N Methylacetoacetic acid Chemical compound COC(=O)CC(C)=O WRQNANDWMGAFTP-UHFFFAOYSA-N 0.000 description 1
- MGJKQDOBUOMPEZ-UHFFFAOYSA-N N,N'-dimethylurea Chemical compound CNC(=O)NC MGJKQDOBUOMPEZ-UHFFFAOYSA-N 0.000 description 1
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 description 1
- XTUVJUMINZSXGF-UHFFFAOYSA-N N-methylcyclohexylamine Chemical compound CNC1CCCCC1 XTUVJUMINZSXGF-UHFFFAOYSA-N 0.000 description 1
- XGEGHDBEHXKFPX-UHFFFAOYSA-N N-methylthiourea Natural products CNC(N)=O XGEGHDBEHXKFPX-UHFFFAOYSA-N 0.000 description 1
- VNMZCZVSKDHQNV-UHFFFAOYSA-N N1C(CCC1)=O.C(C1=CC=CC=C1)(=O)N Chemical compound N1C(CCC1)=O.C(C1=CC=CC=C1)(=O)N VNMZCZVSKDHQNV-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- NMNFDTCOWIDEAV-UHFFFAOYSA-N OP(O)=O.OP(O)=O Chemical compound OP(O)=O.OP(O)=O NMNFDTCOWIDEAV-UHFFFAOYSA-N 0.000 description 1
- HERWCMONDVEDDA-UHFFFAOYSA-N OS(C(C(C1(F)F)(F)F)(C=CC1F)F)(=O)=O.I Chemical compound OS(C(C(C1(F)F)(F)F)(C=CC1F)F)(=O)=O.I HERWCMONDVEDDA-UHFFFAOYSA-N 0.000 description 1
- ZPOPUCAXKMZPFQ-UHFFFAOYSA-N OS(C1=CC=CC=C1C(F)(F)F)(=O)=O.I Chemical compound OS(C1=CC=CC=C1C(F)(F)F)(=O)=O.I ZPOPUCAXKMZPFQ-UHFFFAOYSA-N 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- QLZHNIAADXEJJP-UHFFFAOYSA-N Phenylphosphonic acid Chemical compound OP(O)(=O)C1=CC=CC=C1 QLZHNIAADXEJJP-UHFFFAOYSA-N 0.000 description 1
- 229920001665 Poly-4-vinylphenol Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920002873 Polyethylenimine Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 229920000147 Styrene maleic anhydride Polymers 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- 239000004809 Teflon Substances 0.000 description 1
- 229920006362 Teflon® Polymers 0.000 description 1
- NSOXQYCFHDMMGV-UHFFFAOYSA-N Tetrakis(2-hydroxypropyl)ethylenediamine Chemical compound CC(O)CN(CC(C)O)CCN(CC(C)O)CC(C)O NSOXQYCFHDMMGV-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- KWVPHRLGZMLJEV-UHFFFAOYSA-N [4-(2-hydroxypropan-2-yl)phenyl] benzoate Chemical compound C1=CC(C(C)(O)C)=CC=C1OC(=O)C1=CC=CC=C1 KWVPHRLGZMLJEV-UHFFFAOYSA-N 0.000 description 1
- AMIUMHHQNFTNQO-UHFFFAOYSA-M [4-[(2-methylpropan-2-yl)oxy]phenyl]-diphenylsulfanium;1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F.C1=CC(OC(C)(C)C)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 AMIUMHHQNFTNQO-UHFFFAOYSA-M 0.000 description 1
- HKKMPPDCCCBZHM-UHFFFAOYSA-M [4-[(2-methylpropan-2-yl)oxy]phenyl]-diphenylsulfanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(OC(C)(C)C)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 HKKMPPDCCCBZHM-UHFFFAOYSA-M 0.000 description 1
- LFJLNVUGIWJQCX-UHFFFAOYSA-N [[(4-bromophenyl)-cyanomethylidene]amino] methanesulfonate Chemical compound CS(=O)(=O)ON=C(C#N)C1=CC=C(Br)C=C1 LFJLNVUGIWJQCX-UHFFFAOYSA-N 0.000 description 1
- XDHUWCBFSRIEEC-UHFFFAOYSA-N [[cyano(phenyl)methylidene]amino] methanesulfonate Chemical compound CS(=O)(=O)ON=C(C#N)C1=CC=CC=C1 XDHUWCBFSRIEEC-UHFFFAOYSA-N 0.000 description 1
- UPXATANHGSYTNL-UHFFFAOYSA-N [[cyano-(4-methoxyphenyl)methylidene]amino] ethanesulfonate Chemical compound CCS(=O)(=O)ON=C(C#N)C1=CC=C(OC)C=C1 UPXATANHGSYTNL-UHFFFAOYSA-N 0.000 description 1
- BADIYTSGNBSILF-UHFFFAOYSA-N [[cyano-(4-methoxyphenyl)methylidene]amino] methanesulfonate Chemical compound COC1=CC=C(C(=NOS(C)(=O)=O)C#N)C=C1 BADIYTSGNBSILF-UHFFFAOYSA-N 0.000 description 1
- ZMIGBWNVUSOGIL-UHFFFAOYSA-N [[cyano-(4-methoxyphenyl)methylidene]amino] trifluoromethanesulfonate Chemical compound COC1=CC=C(C(=NOS(=O)(=O)C(F)(F)F)C#N)C=C1 ZMIGBWNVUSOGIL-UHFFFAOYSA-N 0.000 description 1
- WVCAZSUCPKYBCC-UHFFFAOYSA-N [[cyano-(4-methylphenyl)methylidene]amino] propane-1-sulfonate Chemical compound CCCS(=O)(=O)ON=C(C#N)C1=CC=C(C)C=C1 WVCAZSUCPKYBCC-UHFFFAOYSA-N 0.000 description 1
- NVJPBZCLWGTJKD-UHFFFAOYSA-N [bis(4-tert-butylphenyl)-lambda3-iodanyl] trifluoromethanesulfonate Chemical compound CC(C)(C)c1ccc(cc1)[I](OS(=O)(=O)C(F)(F)F)c1ccc(cc1)C(C)(C)C NVJPBZCLWGTJKD-UHFFFAOYSA-N 0.000 description 1
- GLGXSTXZLFQYKJ-UHFFFAOYSA-N [cyclohexylsulfonyl(diazo)methyl]sulfonylcyclohexane Chemical compound C1CCCCC1S(=O)(=O)C(=[N+]=[N-])S(=O)(=O)C1CCCCC1 GLGXSTXZLFQYKJ-UHFFFAOYSA-N 0.000 description 1
- HXGDTGSAIMULJN-UHFFFAOYSA-N acetnaphthylene Natural products C1=CC(C=C2)=C3C2=CC=CC3=C1 HXGDTGSAIMULJN-UHFFFAOYSA-N 0.000 description 1
- 125000003668 acetyloxy group Chemical group [H]C([H])([H])C(=O)O[*] 0.000 description 1
- 150000008065 acid anhydrides Chemical class 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 150000007933 aliphatic carboxylic acids Chemical class 0.000 description 1
- 125000002877 alkyl aryl group Chemical group 0.000 description 1
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 1
- 150000005215 alkyl ethers Chemical class 0.000 description 1
- 150000001350 alkyl halides Chemical class 0.000 description 1
- 125000005037 alkyl phenyl group Chemical group 0.000 description 1
- 150000001343 alkyl silanes Chemical class 0.000 description 1
- 125000005336 allyloxy group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 238000010539 anionic addition polymerization reaction Methods 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- XJDFBLQCLSBCGQ-UHFFFAOYSA-N anthracene-1-carbaldehyde Chemical compound C1=CC=C2C=C3C(C=O)=CC=CC3=CC2=C1 XJDFBLQCLSBCGQ-UHFFFAOYSA-N 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 description 1
- 125000002102 aryl alkyloxo group Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- IVRMZWNICZWHMI-UHFFFAOYSA-N azide group Chemical group [N-]=[N+]=[N-] IVRMZWNICZWHMI-UHFFFAOYSA-N 0.000 description 1
- RQPZNWPYLFFXCP-UHFFFAOYSA-L barium dihydroxide Chemical compound [OH-].[OH-].[Ba+2] RQPZNWPYLFFXCP-UHFFFAOYSA-L 0.000 description 1
- 229910001863 barium hydroxide Inorganic materials 0.000 description 1
- CXJVMJWCNFOERL-UHFFFAOYSA-N benzenesulfonylsulfonylbenzene Chemical compound C=1C=CC=CC=1S(=O)(=O)S(=O)(=O)C1=CC=CC=C1 CXJVMJWCNFOERL-UHFFFAOYSA-N 0.000 description 1
- UDEWPOVQBGFNGE-UHFFFAOYSA-N benzoic acid n-propyl ester Natural products CCCOC(=O)C1=CC=CC=C1 UDEWPOVQBGFNGE-UHFFFAOYSA-N 0.000 description 1
- 150000008366 benzophenones Chemical class 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 125000001231 benzoyloxy group Chemical group C(C1=CC=CC=C1)(=O)O* 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- ZLSMCQSGRWNEGX-UHFFFAOYSA-N bis(4-aminophenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=C(N)C=C1 ZLSMCQSGRWNEGX-UHFFFAOYSA-N 0.000 description 1
- DGHWIWLPQJTBRO-UHFFFAOYSA-N bis(4-fluorophenyl)-(4-hydroxyphenyl)sulfanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(O)=CC=C1[S+](C=1C=CC(F)=CC=1)C1=CC=C(F)C=C1 DGHWIWLPQJTBRO-UHFFFAOYSA-N 0.000 description 1
- YRUQSKZJSITXPM-UHFFFAOYSA-N bis(4-hydroxyphenyl)-phenylsulfanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(O)=CC=C1[S+](C=1C=CC(O)=CC=1)C1=CC=CC=C1 YRUQSKZJSITXPM-UHFFFAOYSA-N 0.000 description 1
- DNFSNYQTQMVTOK-UHFFFAOYSA-N bis(4-tert-butylphenyl)iodanium Chemical compound C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 DNFSNYQTQMVTOK-UHFFFAOYSA-N 0.000 description 1
- MDUKBVGQQFOMPC-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;(7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl)methanesulfonate Chemical compound C1CC2(CS([O-])(=O)=O)C(=O)CC1C2(C)C.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 MDUKBVGQQFOMPC-UHFFFAOYSA-M 0.000 description 1
- DJBAOXYQCAKLPH-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 DJBAOXYQCAKLPH-UHFFFAOYSA-M 0.000 description 1
- SDAQQOKKCSSGPU-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;2,4-difluorobenzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=C(F)C=C1F.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 SDAQQOKKCSSGPU-UHFFFAOYSA-M 0.000 description 1
- DFWHUMPTRSVZOW-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;2-carboxyphenolate Chemical compound OC1=CC=CC=C1C([O-])=O.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 DFWHUMPTRSVZOW-UHFFFAOYSA-M 0.000 description 1
- IBWLSQDQZDVNSS-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;4-(trifluoromethyl)benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=C(C(F)(F)F)C=C1.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 IBWLSQDQZDVNSS-UHFFFAOYSA-M 0.000 description 1
- UEJFJTOGXLEPIV-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;4-methylbenzenesulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 UEJFJTOGXLEPIV-UHFFFAOYSA-M 0.000 description 1
- WMVCDPHJNPHEIB-UHFFFAOYSA-M bis(4-tert-butylphenyl)iodanium;hydroxide Chemical compound [OH-].C1=CC(C(C)(C)C)=CC=C1[I+]C1=CC=C(C(C)(C)C)C=C1 WMVCDPHJNPHEIB-UHFFFAOYSA-M 0.000 description 1
- DBAVIPITSXUODT-UHFFFAOYSA-N bis[4-(trifluoromethyl)phenyl]iodanium Chemical compound C1=CC(C(F)(F)F)=CC=C1[I+]C1=CC=C(C(F)(F)F)C=C1 DBAVIPITSXUODT-UHFFFAOYSA-N 0.000 description 1
- BMOAHCCGSGYJFA-UHFFFAOYSA-M bis[4-(trifluoromethyl)phenyl]iodanium;1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F.C1=CC(C(F)(F)F)=CC=C1[I+]C1=CC=C(C(F)(F)F)C=C1 BMOAHCCGSGYJFA-UHFFFAOYSA-M 0.000 description 1
- QFJRKCBZAPLGLD-UHFFFAOYSA-M bis[4-(trifluoromethyl)phenyl]iodanium;4-methylbenzenesulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.C1=CC(C(F)(F)F)=CC=C1[I+]C1=CC=C(C(F)(F)F)C=C1 QFJRKCBZAPLGLD-UHFFFAOYSA-M 0.000 description 1
- IPNMEMKDCWTRGK-UHFFFAOYSA-M bis[4-(trifluoromethyl)phenyl]iodanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(C(F)(F)F)=CC=C1[I+]C1=CC=C(C(F)(F)F)C=C1 IPNMEMKDCWTRGK-UHFFFAOYSA-M 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- 125000006014 bromoethoxy group Chemical group 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- GDLKDGFIUWNVSJ-UHFFFAOYSA-N butyl 3-methoxy-3-methylbutanoate Chemical compound CCCCOC(=O)CC(C)(C)OC GDLKDGFIUWNVSJ-UHFFFAOYSA-N 0.000 description 1
- XYKQGLNAVREJFA-UHFFFAOYSA-N butyl 3-methoxybutanoate Chemical compound CCCCOC(=O)CC(C)OC XYKQGLNAVREJFA-UHFFFAOYSA-N 0.000 description 1
- 229940043232 butyl acetate Drugs 0.000 description 1
- 125000004744 butyloxycarbonyl group Chemical group 0.000 description 1
- 125000000480 butynyl group Chemical group [*]C#CC([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- MIOPJNTWMNEORI-UHFFFAOYSA-N camphorsulfonic acid Chemical compound C1CC2(CS(O)(=O)=O)C(=O)CC1C2(C)C MIOPJNTWMNEORI-UHFFFAOYSA-N 0.000 description 1
- CREMABGTGYGIQB-UHFFFAOYSA-N carbon carbon Chemical compound C.C CREMABGTGYGIQB-UHFFFAOYSA-N 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000003729 cation exchange resin Substances 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 229920001429 chelating resin Polymers 0.000 description 1
- NFJPGAKRJKLOJK-UHFFFAOYSA-N chembl1901631 Chemical compound CCCCOP(=O)OCCCC NFJPGAKRJKLOJK-UHFFFAOYSA-N 0.000 description 1
- 239000007810 chemical reaction solvent Substances 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 125000002668 chloroacetyl group Chemical group ClCC(=O)* 0.000 description 1
- 125000004651 chloromethoxy group Chemical group ClCO* 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 239000003426 co-catalyst Substances 0.000 description 1
- 239000013065 commercial product Substances 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 125000002592 cumenyl group Chemical group C1(=C(C=CC=C1)*)C(C)C 0.000 description 1
- 229960003280 cupric chloride Drugs 0.000 description 1
- 125000002993 cycloalkylene group Chemical group 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- UKJLNMAFNRKWGR-UHFFFAOYSA-N cyclohexatrienamine Chemical group NC1=CC=C=C[CH]1 UKJLNMAFNRKWGR-UHFFFAOYSA-N 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- XCIXKGXIYUWCLL-UHFFFAOYSA-N cyclopentanol Chemical compound OC1CCCC1 XCIXKGXIYUWCLL-UHFFFAOYSA-N 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 125000003074 decanoyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C(*)=O 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000006612 decyloxy group Chemical group 0.000 description 1
- 125000005070 decynyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C#C* 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000005695 dehalogenation reaction Methods 0.000 description 1
- 238000006704 dehydrohalogenation reaction Methods 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 125000004915 dibutylamino group Chemical group C(CCC)N(CCCC)* 0.000 description 1
- ZFTFAPZRGNKQPU-UHFFFAOYSA-N dicarbonic acid Chemical class OC(=O)OC(O)=O ZFTFAPZRGNKQPU-UHFFFAOYSA-N 0.000 description 1
- QVQGTNFYPJQJNM-UHFFFAOYSA-N dicyclohexylmethanamine Chemical compound C1CCCCC1C(N)C1CCCCC1 QVQGTNFYPJQJNM-UHFFFAOYSA-N 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 125000001664 diethylamino group Chemical group [H]C([H])([H])C([H])([H])N(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006264 diethylaminomethyl group Chemical group [H]C([H])([H])C([H])([H])N(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical group C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 125000006222 dimethylaminomethyl group Chemical group [H]C([H])([H])N(C([H])([H])[H])C([H])([H])* 0.000 description 1
- LAWOZCWGWDVVSG-UHFFFAOYSA-N dioctylamine Chemical compound CCCCCCCCNCCCCCCCC LAWOZCWGWDVVSG-UHFFFAOYSA-N 0.000 description 1
- IJINQHYPVXDROA-UHFFFAOYSA-M diphenyl-(2,3,4-trimethylphenyl)sulfanium;4-methylbenzenesulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.CC1=C(C)C(C)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 IJINQHYPVXDROA-UHFFFAOYSA-M 0.000 description 1
- UIJMWEXVXVYFID-UHFFFAOYSA-M diphenyl-(2,4,6-trimethylphenyl)sulfanium 1,4,5,5,6,6-hexafluorocyclohex-2-ene-1-sulfonate Chemical compound FC1C(C(C(C=C1)(S(=O)(=O)[O-])F)(F)F)(F)F.C1(=CC=CC=C1)[S+](C1=C(C=C(C=C1C)C)C)C1=CC=CC=C1 UIJMWEXVXVYFID-UHFFFAOYSA-M 0.000 description 1
- CTWXRPQORLZRLG-UHFFFAOYSA-M diphenyl-(2,4,6-trimethylphenyl)sulfanium;4-(trifluoromethyl)benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=C(C(F)(F)F)C=C1.CC1=CC(C)=CC(C)=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 CTWXRPQORLZRLG-UHFFFAOYSA-M 0.000 description 1
- OZLBDYMWFAHSOQ-UHFFFAOYSA-N diphenyliodanium Chemical compound C=1C=CC=CC=1[I+]C1=CC=CC=C1 OZLBDYMWFAHSOQ-UHFFFAOYSA-N 0.000 description 1
- ORPDKMPYOLFUBA-UHFFFAOYSA-M diphenyliodanium;1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound C=1C=CC=CC=1[I+]C1=CC=CC=C1.[O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F ORPDKMPYOLFUBA-UHFFFAOYSA-M 0.000 description 1
- LRYJJIPJQVBUNA-UHFFFAOYSA-M diphenyliodanium;2-(trifluoromethyl)benzenesulfonate Chemical compound C=1C=CC=CC=1[I+]C1=CC=CC=C1.[O-]S(=O)(=O)C1=CC=CC=C1C(F)(F)F LRYJJIPJQVBUNA-UHFFFAOYSA-M 0.000 description 1
- AVTMKEVUEUHBNF-UHFFFAOYSA-M diphenyliodanium;4-(trifluoromethyl)benzenesulfonate Chemical compound C=1C=CC=CC=1[I+]C1=CC=CC=C1.[O-]S(=O)(=O)C1=CC=C(C(F)(F)F)C=C1 AVTMKEVUEUHBNF-UHFFFAOYSA-M 0.000 description 1
- UMIKAXKFQJWKCV-UHFFFAOYSA-M diphenyliodanium;4-methylbenzenesulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.C=1C=CC=CC=1[I+]C1=CC=CC=C1 UMIKAXKFQJWKCV-UHFFFAOYSA-M 0.000 description 1
- SBQIJPBUMNWUKN-UHFFFAOYSA-M diphenyliodanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C=1C=CC=CC=1[I+]C1=CC=CC=C1 SBQIJPBUMNWUKN-UHFFFAOYSA-M 0.000 description 1
- 125000004914 dipropylamino group Chemical group C(CC)N(CCC)* 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- JRBPAEWTRLWTQC-UHFFFAOYSA-N dodecylamine Chemical compound CCCCCCCCCCCCN JRBPAEWTRLWTQC-UHFFFAOYSA-N 0.000 description 1
- ODQWQRRAPPTVAG-GZTJUZNOSA-N doxepin Chemical compound C1OC2=CC=CC=C2C(=C/CCN(C)C)/C2=CC=CC=C21 ODQWQRRAPPTVAG-GZTJUZNOSA-N 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- CEIPQQODRKXDSB-UHFFFAOYSA-N ethyl 3-(6-hydroxynaphthalen-2-yl)-1H-indazole-5-carboximidate dihydrochloride Chemical compound Cl.Cl.C1=C(O)C=CC2=CC(C3=NNC4=CC=C(C=C43)C(=N)OCC)=CC=C21 CEIPQQODRKXDSB-UHFFFAOYSA-N 0.000 description 1
- IJUHLFUALMUWOM-UHFFFAOYSA-N ethyl 3-methoxypropanoate Chemical compound CCOC(=O)CCOC IJUHLFUALMUWOM-UHFFFAOYSA-N 0.000 description 1
- 229940117360 ethyl pyruvate Drugs 0.000 description 1
- 125000000031 ethylamino group Chemical group [H]C([H])([H])C([H])([H])N([H])[*] 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000001900 extreme ultraviolet lithography Methods 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 125000003983 fluorenyl group Chemical class C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- WBJINCZRORDGAQ-UHFFFAOYSA-N formic acid ethyl ester Natural products CCOC=O WBJINCZRORDGAQ-UHFFFAOYSA-N 0.000 description 1
- 150000002240 furans Chemical group 0.000 description 1
- 125000000457 gamma-lactone group Chemical group 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 150000004795 grignard reagents Chemical class 0.000 description 1
- 230000020169 heat generation Effects 0.000 description 1
- 125000004836 hexamethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 1
- 125000003104 hexanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000003707 hexyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 125000005935 hexyloxycarbonyl group Chemical group 0.000 description 1
- 125000005980 hexynyl group Chemical group 0.000 description 1
- 238000007602 hot air drying Methods 0.000 description 1
- UTSOXZIZVGUTCF-UHFFFAOYSA-N hydrate;hydroiodide Chemical compound O.I UTSOXZIZVGUTCF-UHFFFAOYSA-N 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000012433 hydrogen halide Substances 0.000 description 1
- 229910000039 hydrogen halide Inorganic materials 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical class I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 1
- XLSMFKSTNGKWQX-UHFFFAOYSA-N hydroxyacetone Chemical compound CC(=O)CO XLSMFKSTNGKWQX-UHFFFAOYSA-N 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 229930002839 ionone Natural products 0.000 description 1
- 150000002499 ionone derivatives Chemical class 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 description 1
- 230000001678 irradiating effect Effects 0.000 description 1
- JMMWKPVZQRWMSS-UHFFFAOYSA-N isopropanol acetate Natural products CC(C)OC(C)=O JMMWKPVZQRWMSS-UHFFFAOYSA-N 0.000 description 1
- 125000000555 isopropenyl group Chemical group [H]\C([H])=C(\*)C([H])([H])[H] 0.000 description 1
- 229940011051 isopropyl acetate Drugs 0.000 description 1
- GWYFCOCPABKNJV-UHFFFAOYSA-M isovalerate Chemical compound CC(C)CC([O-])=O GWYFCOCPABKNJV-UHFFFAOYSA-M 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 125000000400 lauroyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- JDSHMPZPIAZGSV-UHFFFAOYSA-N melamine Chemical compound NC1=NC(N)=NC(N)=N1 JDSHMPZPIAZGSV-UHFFFAOYSA-N 0.000 description 1
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- OJURWUUOVGOHJZ-UHFFFAOYSA-N methyl 2-[(2-acetyloxyphenyl)methyl-[2-[(2-acetyloxyphenyl)methyl-(2-methoxy-2-oxoethyl)amino]ethyl]amino]acetate Chemical compound C=1C=CC=C(OC(C)=O)C=1CN(CC(=O)OC)CCN(CC(=O)OC)CC1=CC=CC=C1OC(C)=O OJURWUUOVGOHJZ-UHFFFAOYSA-N 0.000 description 1
- HSDFKDZBJMDHFF-UHFFFAOYSA-N methyl 3-ethoxypropanoate Chemical compound CCOCCC(=O)OC HSDFKDZBJMDHFF-UHFFFAOYSA-N 0.000 description 1
- LYLUAHKXJUQFDG-UHFFFAOYSA-N methyl 3-methoxy-2-methylpropanoate Chemical compound COCC(C)C(=O)OC LYLUAHKXJUQFDG-UHFFFAOYSA-N 0.000 description 1
- 229940057867 methyl lactate Drugs 0.000 description 1
- 229940017219 methyl propionate Drugs 0.000 description 1
- CWKLZLBVOJRSOM-UHFFFAOYSA-N methyl pyruvate Chemical compound COC(=O)C(C)=O CWKLZLBVOJRSOM-UHFFFAOYSA-N 0.000 description 1
- 125000000250 methylamino group Chemical group [H]N(*)C([H])([H])[H] 0.000 description 1
- XGEGHDBEHXKFPX-NJFSPNSNSA-N methylurea Chemical compound [14CH3]NC(N)=O XGEGHDBEHXKFPX-NJFSPNSNSA-N 0.000 description 1
- IQTRBJRORQFYLN-UHFFFAOYSA-N molport-019-739-976 Chemical compound C1=CC(C)=CC=C1S(=O)(=O)ON1C(=O)C2C(C=C3)CC3C2C1=O IQTRBJRORQFYLN-UHFFFAOYSA-N 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 125000001064 morpholinomethyl group Chemical group [H]C([H])(*)N1C([H])([H])C([H])([H])OC([H])([H])C1([H])[H] 0.000 description 1
- YCMDNBGUNDHOOD-UHFFFAOYSA-N n -((trifluoromethylsulfonyl)oxy)-5-norbornene-2,3-dicarboximide Chemical compound C1=CC2CC1C1C2C(=O)N(OS(=O)(=O)C(F)(F)F)C1=O YCMDNBGUNDHOOD-UHFFFAOYSA-N 0.000 description 1
- ZQJAONQEOXOVNR-UHFFFAOYSA-N n,n-di(nonyl)nonan-1-amine Chemical compound CCCCCCCCCN(CCCCCCCCC)CCCCCCCCC ZQJAONQEOXOVNR-UHFFFAOYSA-N 0.000 description 1
- FRQONEWDWWHIPM-UHFFFAOYSA-N n,n-dicyclohexylcyclohexanamine Chemical compound C1CCCCC1N(C1CCCCC1)C1CCCCC1 FRQONEWDWWHIPM-UHFFFAOYSA-N 0.000 description 1
- COFKFSSWMQHKMD-UHFFFAOYSA-N n,n-didecyldecan-1-amine Chemical compound CCCCCCCCCCN(CCCCCCCCCC)CCCCCCCCCC COFKFSSWMQHKMD-UHFFFAOYSA-N 0.000 description 1
- DIAIBWNEUYXDNL-UHFFFAOYSA-N n,n-dihexylhexan-1-amine Chemical compound CCCCCCN(CCCCCC)CCCCCC DIAIBWNEUYXDNL-UHFFFAOYSA-N 0.000 description 1
- YWFWDNVOPHGWMX-UHFFFAOYSA-N n,n-dimethyldodecan-1-amine Chemical compound CCCCCCCCCCCCN(C)C YWFWDNVOPHGWMX-UHFFFAOYSA-N 0.000 description 1
- OOHAUGDGCWURIT-UHFFFAOYSA-N n,n-dipentylpentan-1-amine Chemical compound CCCCCN(CCCCC)CCCCC OOHAUGDGCWURIT-UHFFFAOYSA-N 0.000 description 1
- WDQKICIMIPUDBL-UHFFFAOYSA-N n-[2-(dimethylamino)ethyl]prop-2-enamide Chemical compound CN(C)CCNC(=O)C=C WDQKICIMIPUDBL-UHFFFAOYSA-N 0.000 description 1
- MXLKZNKNVRJFLQ-UHFFFAOYSA-N n-[4-[(benzenesulfonyldiazenyl)methylsulfonyl]butylsulfonylmethylimino]benzenesulfonamide Chemical compound C=1C=CC=CC=1S(=O)(=O)N=NCS(=O)(=O)CCCCS(=O)(=O)CN=NS(=O)(=O)C1=CC=CC=C1 MXLKZNKNVRJFLQ-UHFFFAOYSA-N 0.000 description 1
- SAOJTUFUKMIENZ-UHFFFAOYSA-N n-[6-[(benzenesulfonyldiazenyl)methylsulfonyl]hexylsulfonylmethylimino]benzenesulfonamide Chemical compound C=1C=CC=CC=1S(=O)(=O)N=NCS(=O)(=O)CCCCCCS(=O)(=O)CN=NS(=O)(=O)C1=CC=CC=C1 SAOJTUFUKMIENZ-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229940017144 n-butyl lactate Drugs 0.000 description 1
- 125000006126 n-butyl sulfonyl group Chemical group 0.000 description 1
- GMTCPFCMAHMEMT-UHFFFAOYSA-N n-decyldecan-1-amine Chemical compound CCCCCCCCCCNCCCCCCCCCC GMTCPFCMAHMEMT-UHFFFAOYSA-N 0.000 description 1
- UWHRNIXHZAWBMF-UHFFFAOYSA-N n-dodecyl-n-methyldodecan-1-amine Chemical compound CCCCCCCCCCCCN(C)CCCCCCCCCCCC UWHRNIXHZAWBMF-UHFFFAOYSA-N 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- NJWMENBYMFZACG-UHFFFAOYSA-N n-heptylheptan-1-amine Chemical compound CCCCCCCNCCCCCCC NJWMENBYMFZACG-UHFFFAOYSA-N 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- PXSXRABJBXYMFT-UHFFFAOYSA-N n-hexylhexan-1-amine Chemical compound CCCCCCNCCCCCC PXSXRABJBXYMFT-UHFFFAOYSA-N 0.000 description 1
- OMEMQVZNTDHENJ-UHFFFAOYSA-N n-methyldodecan-1-amine Chemical compound CCCCCCCCCCCCNC OMEMQVZNTDHENJ-UHFFFAOYSA-N 0.000 description 1
- MFHKEJIIHDNPQE-UHFFFAOYSA-N n-nonylnonan-1-amine Chemical compound CCCCCCCCCNCCCCCCCCC MFHKEJIIHDNPQE-UHFFFAOYSA-N 0.000 description 1
- JACMPVXHEARCBO-UHFFFAOYSA-N n-pentylpentan-1-amine Chemical compound CCCCCNCCCCC JACMPVXHEARCBO-UHFFFAOYSA-N 0.000 description 1
- VBEGHXKAFSLLGE-UHFFFAOYSA-N n-phenylnitramide Chemical compound [O-][N+](=O)NC1=CC=CC=C1 VBEGHXKAFSLLGE-UHFFFAOYSA-N 0.000 description 1
- OOYZLFZSZZFLJW-UHFFFAOYSA-M naphthalen-1-yl(diphenyl)sulfanium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC=CC=C1[S+](C=1C2=CC=CC=C2C=CC=1)C1=CC=CC=C1 OOYZLFZSZZFLJW-UHFFFAOYSA-M 0.000 description 1
- CXAYOCVHDCXPAI-UHFFFAOYSA-N naphthalen-1-yl(phenyl)methanone Chemical compound C=1C=CC2=CC=CC=C2C=1C(=O)C1=CC=CC=C1 CXAYOCVHDCXPAI-UHFFFAOYSA-N 0.000 description 1
- 150000002790 naphthalenes Chemical group 0.000 description 1
- 229960002715 nicotine Drugs 0.000 description 1
- SNICXCGAKADSCV-UHFFFAOYSA-N nicotine Natural products CN1CCCC1C1=CC=CN=C1 SNICXCGAKADSCV-UHFFFAOYSA-N 0.000 description 1
- 235000001968 nicotinic acid Nutrition 0.000 description 1
- 239000011664 nicotinic acid Substances 0.000 description 1
- 229960003512 nicotinic acid Drugs 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- FJDUDHYHRVPMJZ-UHFFFAOYSA-N nonan-1-amine Chemical compound CCCCCCCCCN FJDUDHYHRVPMJZ-UHFFFAOYSA-N 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- BTFQKIATRPGRBS-UHFFFAOYSA-N o-tolualdehyde Chemical compound CC1=CC=CC=C1C=O BTFQKIATRPGRBS-UHFFFAOYSA-N 0.000 description 1
- IOQPZZOEVPZRBK-UHFFFAOYSA-N octan-1-amine Chemical compound CCCCCCCCN IOQPZZOEVPZRBK-UHFFFAOYSA-N 0.000 description 1
- WOFPPJOZXUTRAU-UHFFFAOYSA-N octan-4-ol Chemical compound CCCCC(O)CCC WOFPPJOZXUTRAU-UHFFFAOYSA-N 0.000 description 1
- 125000002801 octanoyl group Chemical group C(CCCCCCC)(=O)* 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000005069 octynyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C#C* 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- CDXVUROVRIFQMV-UHFFFAOYSA-N oxo(diphenoxy)phosphanium Chemical compound C=1C=CC=CC=1O[P+](=O)OC1=CC=CC=C1 CDXVUROVRIFQMV-UHFFFAOYSA-N 0.000 description 1
- RQKYHDHLEMEVDR-UHFFFAOYSA-N oxo-bis(phenylmethoxy)phosphanium Chemical compound C=1C=CC=CC=1CO[P+](=O)OCC1=CC=CC=C1 RQKYHDHLEMEVDR-UHFFFAOYSA-N 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 125000001312 palmitoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 125000004115 pentoxy group Chemical group [*]OC([H])([H])C([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- RPXRNZKIARTPPW-UHFFFAOYSA-N pentyl 4-(2-hydroxypropan-2-yl)benzoate Chemical compound CCCCCOC(=O)C1=CC=C(C(C)(C)O)C=C1 RPXRNZKIARTPPW-UHFFFAOYSA-N 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 125000001148 pentyloxycarbonyl group Chemical group 0.000 description 1
- 125000005981 pentynyl group Chemical group 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 125000001484 phenothiazinyl group Chemical class C1(=CC=CC=2SC3=CC=CC=C3NC12)* 0.000 description 1
- ICNNGWUEZQCEGJ-UHFFFAOYSA-N phenyl 3-(2-hydroxypropan-2-yl)benzoate Chemical compound CC(C)(O)C1=CC=CC(C(=O)OC=2C=CC=CC=2)=C1 ICNNGWUEZQCEGJ-UHFFFAOYSA-N 0.000 description 1
- JPNCJWQEOLYHTA-UHFFFAOYSA-N phenyl 4-hydroxy-2-propan-2-ylbenzoate Chemical compound OC1=CC(=C(C(=O)OC2=CC=CC=C2)C=C1)C(C)C JPNCJWQEOLYHTA-UHFFFAOYSA-N 0.000 description 1
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 1
- MLCHBQKMVKNBOV-UHFFFAOYSA-N phenylphosphinic acid Chemical compound OP(=O)C1=CC=CC=C1 MLCHBQKMVKNBOV-UHFFFAOYSA-N 0.000 description 1
- 150000003007 phosphonic acid derivatives Chemical class 0.000 description 1
- XKJCHHZQLQNZHY-UHFFFAOYSA-N phthalimide Chemical compound C1=CC=C2C(=O)NC(=O)C2=C1 XKJCHHZQLQNZHY-UHFFFAOYSA-N 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229920000083 poly(allylamine) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 239000004584 polyacrylic acid Substances 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 238000004262 preparative liquid chromatography Methods 0.000 description 1
- VWQXLMJSFGLQIT-UHFFFAOYSA-N prop-2-enoyl bromide Chemical compound BrC(=O)C=C VWQXLMJSFGLQIT-UHFFFAOYSA-N 0.000 description 1
- GLFSWJDJMXUVEV-UHFFFAOYSA-N prop-2-enoyl iodide Chemical compound IC(=O)C=C GLFSWJDJMXUVEV-UHFFFAOYSA-N 0.000 description 1
- 150000003151 propanoic acid esters Chemical class 0.000 description 1
- 125000001325 propanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- QLNJFJADRCOGBJ-UHFFFAOYSA-N propionamide Chemical compound CCC(N)=O QLNJFJADRCOGBJ-UHFFFAOYSA-N 0.000 description 1
- 229940080818 propionamide Drugs 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 description 1
- 150000003220 pyrenes Chemical class 0.000 description 1
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical compound C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 1
- MCJGNVYPOGVAJF-UHFFFAOYSA-N quinolin-8-ol Chemical compound C1=CN=C2C(O)=CC=CC2=C1 MCJGNVYPOGVAJF-UHFFFAOYSA-N 0.000 description 1
- 229920003987 resole Polymers 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229910000679 solder Inorganic materials 0.000 description 1
- 230000007928 solubilization Effects 0.000 description 1
- 238000005063 solubilization Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 125000005017 substituted alkenyl group Chemical group 0.000 description 1
- 125000005415 substituted alkoxy group Chemical group 0.000 description 1
- 229960002317 succinimide Drugs 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 238000004381 surface treatment Methods 0.000 description 1
- IQAPWZNFJIMMMQ-UHFFFAOYSA-N tert-butyl 4-(2-hydroxypropan-2-yl)benzoate Chemical compound CC(C)(C)OC(=O)C1=CC=C(C(C)(C)O)C=C1 IQAPWZNFJIMMMQ-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000003536 tetrazoles Chemical group 0.000 description 1
- 238000002411 thermogravimetry Methods 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 125000005425 toluyl group Chemical group 0.000 description 1
- 125000005270 trialkylamine group Chemical group 0.000 description 1
- 150000003918 triazines Chemical class 0.000 description 1
- 150000003852 triazoles Chemical group 0.000 description 1
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 1
- WTVXIBRMWGUIMI-UHFFFAOYSA-N trifluoro($l^{1}-oxidanylsulfonyl)methane Chemical group [O]S(=O)(=O)C(F)(F)F WTVXIBRMWGUIMI-UHFFFAOYSA-N 0.000 description 1
- IZBIRHQNPWSIET-UHFFFAOYSA-M trifluoromethanesulfonate;tris(4-fluorophenyl)sulfanium Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(F)=CC=C1[S+](C=1C=CC(F)=CC=1)C1=CC=C(F)C=C1 IZBIRHQNPWSIET-UHFFFAOYSA-M 0.000 description 1
- OXWFVYDECNDRMT-UHFFFAOYSA-M trifluoromethanesulfonate;tris(4-methoxyphenyl)sulfanium Chemical compound [O-]S(=O)(=O)C(F)(F)F.C1=CC(OC)=CC=C1[S+](C=1C=CC(OC)=CC=1)C1=CC=C(OC)C=C1 OXWFVYDECNDRMT-UHFFFAOYSA-M 0.000 description 1
- ODHXBMXNKOYIBV-UHFFFAOYSA-N triphenylamine Chemical compound C1=CC=CC=C1N(C=1C=CC=CC=1)C1=CC=CC=C1 ODHXBMXNKOYIBV-UHFFFAOYSA-N 0.000 description 1
- XZZGCKRBJSPNEF-UHFFFAOYSA-M triphenylsulfanium;acetate Chemical compound CC([O-])=O.C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 XZZGCKRBJSPNEF-UHFFFAOYSA-M 0.000 description 1
- KOFQUBYAUWJFIT-UHFFFAOYSA-M triphenylsulfanium;hydroxide Chemical compound [OH-].C1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 KOFQUBYAUWJFIT-UHFFFAOYSA-M 0.000 description 1
- YFTHZRPMJXBUME-UHFFFAOYSA-N tripropylamine Chemical compound CCCN(CCC)CCC YFTHZRPMJXBUME-UHFFFAOYSA-N 0.000 description 1
- 229910021642 ultra pure water Inorganic materials 0.000 description 1
- 239000012498 ultrapure water Substances 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 238000001039 wet etching Methods 0.000 description 1
- 125000005023 xylyl group Chemical group 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/52—Esters of acyclic unsaturated carboxylic acids having the esterified carboxyl group bound to an acyclic carbon atom
- C07C69/533—Monocarboxylic acid esters having only one carbon-to-carbon double bond
- C07C69/54—Acrylic acid esters; Methacrylic acid esters
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/027—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
- C07C67/14—Preparation of carboxylic acid esters from carboxylic acid halides
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/038—Macromolecular compounds which are rendered insoluble or differentially wettable
- G03F7/0382—Macromolecular compounds which are rendered insoluble or differentially wettable the macromolecular compound being present in a chemically amplified negative photoresist composition
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/14—The ring being saturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/92—Systems containing at least three condensed rings with a condensed ring system consisting of at least two mutually uncondensed aromatic ring systems, linked by an annular structure formed by carbon chains on non-adjacent positions of the aromatic system, e.g. cyclophanes
Definitions
- the present invention relates to a cyclic compound represented by a specific chemical structural formula, a radiation-sensitive composition containing the same, and a resist pattern forming method using the composition.
- the conventional general resist material is a polymer resist material capable of forming an amorphous thin film.
- examples thereof include polymer resist materials such as polymethyl methacrylate, polyhydroxystyrene having an acid dissociable reactive group, and polyalkyl methacrylate.
- a resist thin film prepared by applying such a solution of a polymer resist material onto a substrate is irradiated with ultraviolet rays, far ultraviolet rays, electron beams, extreme ultraviolet rays (EUV), X-rays, etc., so that the thickness is 45 to 100 nm. About a line pattern is formed.
- the polymer resist material has a large molecular weight of about 10,000 to 100,000 and a wide molecular weight distribution
- roughness is generated on the surface of the fine pattern, and the pattern dimension can be controlled. It becomes difficult and the yield decreases. Therefore, there is a limit to miniaturization in conventional lithography using a polymer resist material.
- various low molecular weight resist materials have been proposed.
- the alkali-developable negative radiation-sensitive compositions described in Patent Documents 1 and 2 have insufficient heat resistance and may not form a resist pattern with a good shape.
- the alkali-developable negative radiation-sensitive composition described in Patent Documents 3 and 4 and Non-Patent Document 1 has a low molecular weight and a low molecular weight, so that the molecular size is small and the resolution is low. It is expected to provide a resist pattern with high roughness and low roughness.
- the low molecular weight cyclic polyphenol compound has a rigid cyclic structure in its skeleton, and thus provides high heat resistance despite its low molecular weight.
- currently known low molecular weight cyclic polyphenol compounds have low solubility in safety solvents used in semiconductor manufacturing processes.
- the radiation-sensitive composition containing a low molecular weight cyclic polyphenol compound and a safety solvent has low sensitivity and may not form a resist pattern with a good shape.
- improvement of a low molecular weight cyclic polyphenol compound is desired.
- an object of the present invention is to provide a cyclic compound capable of forming a radiation-sensitive composition having high solubility in a safe solvent, high sensitivity, low roughness, and a good resist pattern shape, a method for producing the same,
- the object is to provide a radiation-sensitive composition containing the same and a resist pattern forming method using the radiation-sensitive composition.
- the inventors of the present invention provide a cyclic compound having a specific structure with high solubility in a safe solvent, high sensitivity, low roughness, and good resist pattern shape.
- the present inventors have found that a radiation-sensitive composition can be formed and have reached the present invention.
- each R 0 independently represents a hydrogen atom, a hydroxyl group, a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted carbon atom having 1 to 20 carbon atoms.
- Linear aliphatic hydrocarbon group substituted or unsubstituted branched aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted cyclic aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted An aryl group having 6 to 20 carbon atoms, a substituted or unsubstituted aralkyl group having 7 to 30 carbon atoms, a substituted or unsubstituted alkoxy group having 1 to 20 carbon atoms, a substituted or unsubstituted amino group having 0 to 20 carbon atoms Group, substituted or unsubstituted alkenyl group having 2 to 20 carbon atoms, substituted or unsubstituted acyl group having 1 to 20 carbon atoms, substituted or unsubstituted alkoxycarbonyl group having 2 to 20 carbon atoms, substituted or unsubstituted A substituted or unsubstituted alkyl
- a valent group (one or more groups selected from the group consisting of a substituted or unsubstituted alkylene group, a substituted or unsubstituted arylene group and an ether group), At least one of R 0 is a monovalent group containing a (meth) acryloyl group. ) 2.
- each R 1 independently represents a hydrogen atom, a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted straight chain having 1 to 20 carbon atoms.
- R 2 each independently represents a hydrogen atom, a cyano group, a nitro group, a halogen atom, a substituted or unsubstituted linear aliphatic hydrocarbon group having 1 to 20 carbon atoms, a substituted or unsubstituted carbon number of 3 to 20 branched aliphatic hydrocarbon groups, substituted or unsubstituted cyclic aliphatic hydrocarbon groups having 3 to
- each R 4 independently represents a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted carbon number of 1 to 20 Linear aliphatic hydrocarbon group, substituted or unsubstituted branched aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted cyclic aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted A substituted aryl group having 6 to 20 carbon atoms, a substituted or unsubstituted alkoxy group having 1 to 20 carbon atoms, or a substituted or unsubstituted alkylsilyl group having 1 to 20 carbon atoms, and p is an integer of 0 to 5 Yes, At least one
- each R 1 independently represents a hydrogen atom, a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted straight chain having 1 to 20 carbon atoms.
- R 4 each independently represents a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted linear aliphatic hydrocarbon group having 1 to 20 carbon atoms, a substituted or unsubstituted group.
- each R 4 independently represents a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted linear aliphatic group having 1 to 20 carbon atoms.
- Hydrocarbon group substituted or unsubstituted branched aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted cyclic aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted 6 to 6 carbon atoms 20 aryl groups, substituted or unsubstituted alkoxy groups having 1 to 20 carbon atoms, or substituted or unsubstituted alkylsilyl groups having 1 to 20 carbon atoms, p is an integer from 0 to 5, R 5 is a (meth) acryloyl group. ) 5.
- a method for producing the cyclic compound according to any one of items 1 to 4 A step of subjecting one or more aldehyde compounds (A1) and one or more phenolic compounds (A2) to a condensation reaction to obtain a cyclic compound (A); A process for producing a cyclic compound, comprising a step of dehydrohalogenating the cyclic compound (A) and one or more (meth) acryloyl halides. 6).
- the aldehyde compound (A1) has 1 to 4 formyl groups and has 1 to 59 carbon atoms; 6.
- a radiation-sensitive composition comprising a solid component containing the cyclic compound according to any one of items 1 to 4 and a solvent. 8).
- Acid generator (C) that generates acid directly or indirectly by irradiation with any radiation selected from the group consisting of visible light, ultraviolet light, excimer laser, electron beam, extreme ultraviolet light (EUV), X-ray and ion beam 10.
- the solid component is a cyclic compound of 50 to 99.4% by weight, an acid generator (C) of 0.001 to 49% by weight, an acid crosslinking agent (G) of 0.5 to 49% by weight, and an acid diffusion controller (E 13)
- the dissolution rate of the amorphous film after irradiation with KrF excimer laser, extreme ultraviolet light, electron beam or X-ray, or the amorphous film after heating at 20 to 250 ° C. is 5 ⁇ / sec or less.
- the radiation sensitive composition of 14 or 15. 17 Applying the radiation-sensitive composition according to any one of items 7 to 16 on a substrate to form a resist film; Exposing the resist film; And a step of developing the exposed resist film.
- a cyclic compound having high solubility in a safe solvent, high sensitivity, low roughness, and capable of forming a radiation-sensitive composition that gives a good resist pattern shape, and a method for producing the same, including the same A radiation-sensitive composition and a method for forming a resist pattern using the radiation-sensitive composition can be provided.
- the cyclic compound of this embodiment is a cyclic compound represented by the following formula (1) having a molecular weight of 500 to 5,000.
- the cyclic compound of this embodiment is useful as a resist material.
- each R 0 independently represents a hydrogen atom, a hydroxyl group, a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted straight chain having 1 to 20 carbon atoms.
- Chain aliphatic hydrocarbon group substituted or unsubstituted branched aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted cyclic aliphatic hydrocarbon group having 3 to 20 carbon atoms, substituted or unsubstituted Aryl group having 6 to 20 carbon atoms, substituted or unsubstituted aralkyl group having 7 to 30 carbon atoms, substituted or unsubstituted alkoxy group having 1 to 20 carbon atoms, substituted or unsubstituted amino group having 0 to 20 carbon atoms Substituted or unsubstituted alkenyl group having 2 to 20 carbon atoms, substituted or unsubstituted acyl group having 1 to 20 carbon atoms, substituted or unsubstituted alkoxycarbonyl group having 2 to 20 carbon atoms, substituted or unsubstituted Having 1 to 20 carbon atoms, a substituted or unsubstituted
- a group (one or more groups selected from the group consisting of a substituted or unsubstituted alkylene group, a substituted or unsubstituted arylene group and an ether group), and at least one of R 0 is (meta ) A monovalent group containing an acryloyl group.
- R 0 is a monovalent group containing a (meth) acryloyl group
- anionic polymerization of cyclic compounds with an acid becomes possible.
- the radiation-sensitive composition containing the cyclic compound of this embodiment The sensitivity of (resist) can be further increased.
- EUV extreme ultraviolet
- substituted means that one or more hydrogen atoms in the functional group are a halogen atom, a hydroxyl group, a cyano group, a nitro group, a heterocyclic group, a carbon number of 1 to 20 Linear aliphatic hydrocarbon group, branched aliphatic hydrocarbon group having 3 to 20 carbon atoms, cyclic aliphatic hydrocarbon group having 3 to 20 carbon atoms, aryl group having 6 to 20 carbon atoms, and 7 to 7 carbon atoms 30 aralkyl groups, alkoxy groups having 1 to 20 carbon atoms, amino groups having 0 to 20 carbon atoms, alkenyl groups having 2 to 20 carbon atoms, acyl groups having 1 to 20 carbon atoms, alkoxycarbonyl groups having 2 to 20 carbon atoms Means substituted with an alkyloyloxy group having 1 to 20 carbon atoms, an aryloyloxy group having 7 to 30 carbon
- the unsubstituted heterocyclic group is not particularly limited.
- a triazole group, a tetrazole group, etc. are mentioned.
- substituted heterocyclic group is not particularly limited, for example, N-methylpyridyl group, N-fluoropyridyl group, N-hydroxypyridyl group, N-cyanopyridyl group, methylbipyridyl group, methylpyrrolidyl group, methylpyrazolyl group Methylimidazolyl group, methylisoxazolyl group, methylisothiazolyl group, methylpiperidyl group, methylpiperazyl group, methylmorpholyl group, methylthiomorpholyl group, methyltriazole group, and methyltetrazole group.
- the unsubstituted straight-chain aliphatic hydrocarbon group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a methyl group, an ethyl group, a propyl group, a butyl group, a pentyl group, a hexyl group, an octyl group, and a decyl group. , Dodecyl group, hexadecyl group, octadecyl group and the like.
- the substituted linear aliphatic hydrocarbon group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a fluoromethyl group, a 2-hydroxyethyl group, a 3-cyanopropyl group, and a 20-nitrooctadecyl group. It is done.
- the unsubstituted branched aliphatic hydrocarbon group having 3 to 20 carbon atoms is not particularly limited.
- isopropyl group, isobutyl group, tertiary butyl group, neopentyl group, 2-hexyl group, 2-octyl group examples include 2-decyl group, 2-dodecyl group, 2-hexadecyl group, 2-octadecyl group and the like.
- the substituted branched aliphatic hydrocarbon group having 3 to 20 carbon atoms is not particularly limited, and examples thereof include 1-fluoroisopropyl group and 1-hydroxy-2-octadecyl group.
- the unsubstituted cyclic aliphatic hydrocarbon group having 3 to 20 carbon atoms is not particularly limited, and examples thereof include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a cyclooctyl group, a cyclodecyl group, a cyclododecyl group, a cyclohexyl group, and the like.
- a sadecyl group, a cyclooctadecyl group, etc. are mentioned.
- the substituted cyclic aliphatic hydrocarbon group having 3 to 20 carbon atoms is not particularly limited, and examples thereof include a 2-fluorocyclopropyl group and a 4-cyanocyclohexyl group.
- the unsubstituted aryl group having 6 to 20 carbon atoms is not particularly limited, and examples thereof include a phenyl group and a naphthyl group.
- the substituted aryl group having 6 to 20 carbon atoms is not particularly limited, and examples thereof include a 4-methylphenyl group and a 6-fluoronaphthyl group.
- the unsubstituted aralkyl group having 7 to 30 carbon atoms is not particularly limited, and examples thereof include a methylphenyl group, an ethylphenyl group, a methylnaphthyl group, and a dimethylnaphthyl group.
- the substituted aralkyl group having 7 to 30 carbon atoms is not particularly limited, and examples thereof include a 4-fluoro-3-methylphenyl group.
- the unsubstituted alkoxy group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a methoxy group, an ethoxy group, a propoxy group, a butoxy group, a pentyloxy group, a hexyloxy group, an octyloxy group, a decyloxy group, and a dodecyloxy group.
- a methoxy group an ethoxy group, a propoxy group, a butoxy group, a pentyloxy group, a hexyloxy group, an octyloxy group, a decyloxy group, and a dodecyloxy group.
- the substituted alkoxy group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a chloromethoxy group and a bromoethoxy group.
- the unsubstituted amino group having 0 to 20 carbon atoms is not particularly limited, and examples thereof include an amino group, a methylamino group, a dimethylamino group, an ethylamino group, a diethylamino group, a dipropylamino group, and a dibutylamino group. It is done.
- the substituted amino group having 0 to 20 carbon atoms is not particularly limited, and examples thereof include a chloromethylamino group and a dibromomethylamino group.
- the unsubstituted alkenyl group having 2 to 20 carbon atoms is not particularly limited.
- the substituted alkenyl group having 2 to 20 carbon atoms is not particularly limited, and examples thereof include a chloropropynyl group.
- the unsubstituted acyl group having 1 to 20 carbon atoms is not particularly limited, in addition to the above-mentioned (meth) acryloyl group.
- the substituted acyl group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a chloroacetyl group.
- the unsubstituted alkoxycarbonyl group having 2 to 20 carbon atoms is not particularly limited, and examples thereof include a methoxycarbonyl group, an ethoxycarbonyl group, a propoxycarbonyl group, a butoxycarbonyl group, a pentyloxycarbonyl group, a hexyloxycarbonyl group, and an octyloxy group. Examples include carbonyl group, decyloxycarbonyl group, dodecyloxycarbonyl group, hexadecyloxycarbonyl group and the like.
- the substituted alkoxycarbonyl group having 2 to 20 carbon atoms is not particularly limited, and examples thereof include a chloromethoxycarbonyl group.
- the unsubstituted alkyloyloxy group having 1 to 20 carbon atoms is not particularly limited.
- methoxycarbonyloxy group, ethoxycarbonyloxy group, propoxycarbonyloxy group, butoxycarbonyloxy group, pentyloxycarbonyloxy group, hexyl examples include oxycarbonyloxy group, octyloxycarbonyloxy group, decyloxycarbonyloxy group, dodecyloxycarbonyloxy group, hexadecyloxycarbonyloxy group and the like.
- the substituted alkyloyloxy group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a chloromethoxycarbonyloxy group.
- the unsubstituted aryloyloxy group having 7 to 30 carbon atoms is not particularly limited, and examples thereof include a benzoyloxy group and a naphthylcarbonyloxy group.
- the substituted aryloyloxy group having 7 to 30 carbon atoms is not particularly limited, and examples thereof include a chlorobenzoyloxy group.
- the unsubstituted alkylsilyl group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a methylsilyl group, an ethylsilyl group, a propylsilyl group, a butylsilyl group, a pentylsilyl group, a hexylsilyl group, an octylsilyl group, a decylsilyl group, A dodecylsilyl group, a hexadecylsilyl group, an octadecylsilyl group, etc. are mentioned.
- the substituted alkylsilyl group having 1 to 20 carbon atoms is not particularly limited, and examples thereof include a chloromethylsilyl group.
- the monovalent group containing a (meth) acryloyl group is not particularly limited.
- the cyclic compound represented by the formula (1) is preferably a compound represented by the formula (2).
- each R 1 independently represents a hydrogen atom, a cyano group, a nitro group, a substituted or unsubstituted heterocyclic group, a halogen atom, a substituted or unsubstituted straight chain having 1 to 20 carbon atoms.
- the compound represented by the formula (2) is more preferably a compound represented by the formula (4).
- the compound represented by the formula (4) has a high solubility in a safe solvent, and can form a radiation-sensitive composition having high sensitivity, low roughness, and giving a resist pattern having a good shape.
- R 1 , R 4 and p are the same as described above.
- the compound represented by the formula (4) is more preferably a compound represented by the formula (5).
- the compound represented by the formula (5) can form a radiation-sensitive composition having high solubility in a safe solvent, high sensitivity, low roughness, and giving a resist pattern having a good shape.
- R 4 and p are the same as described above, and R 5 is a (meth) acryloyl group. More preferably, the compound represented by the formula (4) is a compound represented by the formula (6) or the formula (7).
- R 5 is the same as described above.
- R 5 is the same as described above.
- the compound represented by the formula (6) or the formula (7) has a higher solubility in a safe solvent, a higher sensitivity, a lower roughness, and a feeling that gives a resist pattern having a better shape.
- a radiation composition can be formed.
- the molecular weight of the cyclic compound represented by the above formula (1) is 500 to 5000, preferably 800 to 2000, more preferably 1000 to 2000. By setting it as the said range, when forming a resist, resolution can be improved, maintaining required film formability.
- the cyclic compound of the present embodiment can take a cis isomer and a trans isomer, but may have any structure or mixture.
- the cyclic compound of this embodiment is used as a resist component of a radiation-sensitive composition, it is preferable to use only one of a cis isomer and a trans isomer because the uniformity of the components in the resist film is high.
- a method for obtaining a cyclic compound consisting of only one of a cis isomer and a trans isomer can be performed by a known method such as separation by column chromatography or preparative liquid chromatography or optimization of a reaction solvent and a reaction temperature during production.
- the cyclic compound of this embodiment includes a step of obtaining a cyclic compound (A) by subjecting one or more aldehyde compounds (A1) and one or more phenolic compounds (A2) to a condensation reaction; And a step of dehydrohalogenating one or more (meth) acryloyl halides.
- the cyclic compound of this embodiment is a condensation of one or more compounds selected from various aromatic aldehyde compounds (A1A) and one or more compounds selected from various phenolic compounds (A2). It can be obtained by dehydrohalogenation reaction between the cyclic compound (A) obtained by the reaction and one or more compounds selected from various (meth) acryloyl halides (A3).
- the cyclic compound of this embodiment is a dehalogenation of one or more compounds selected from various phenolic compounds (A2) and one or more compounds selected from various (meth) acryloyl halides (A3). It can also be obtained by a condensation reaction between a phenolic compound (A2A) obtained by a hydrogen reaction and one or more compounds selected from various aldehyde compounds (A1).
- the aldehyde compound (A1) preferably has 1 to 59 carbon atoms, and preferably has 1 to 4 formyl groups.
- the aldehyde compound (A1) is selected from an aromatic aldehyde compound (A1A) and an aliphatic aldehyde compound (A1B).
- the aromatic aldehyde compound (A1A) is preferably a benzaldehyde compound having 7 to 24 carbon atoms.
- the aromatic aldehyde compound (A1A) is not particularly limited, and examples thereof include benzaldehyde, methylbenzaldehyde, dimethylbenzaldehyde, ethylbenzaldehyde, propylbenzaldehyde, butylbenzaldehyde, ethylmethylbenzaldehyde, isopropylmethylbenzaldehyde, diethylbenzaldehyde, anisaldehyde, naphthoaldehyde.
- the aromatic aldehyde compound (A1A) may have a linear or branched alkyl group having 1 to 4 carbon atoms, a cyano group, a hydroxyl group, a halogen or the like as long as the effects of the present invention are not impaired.
- the aromatic aldehyde compound (A1A) may be used alone or in combination of two or more.
- the aliphatic aldehyde compound (A1B) is preferably a compound having 1 to 24 carbon atoms.
- the aliphatic aldehyde compound (A1B) is not particularly limited.
- Carboxymethyl aldehydes are preferred, octanal, decanal, dodecanal, cyclohexane carboxaldehyde more preferable.
- the aliphatic aldehyde compound (A1B) may have a cyano group, a hydroxyl group, a halogen or the like as long as the effects of the present invention are not impaired.
- the aliphatic aldehyde compound (A1B) may be used alone or in combination of two or more.
- the phenolic compound (A2) preferably has 6 to 15 carbon atoms, and preferably has 1 to 3 phenolic hydroxyl groups. Although it does not specifically limit as a phenolic compound (A2), For example, a phenol, catechol, resorcinol, hydroquinone, pyrogallol etc. are mentioned, Resorcinol and pyrogallol are preferable and resorcinol is more preferable.
- the phenolic compound (A2) may have a linear or branched alkyl group having 1 to 4 carbon atoms, a cyano group, a hydroxyl group, a halogen or the like as long as the effects of the present invention are not impaired. You may use a phenolic compound (A2) individually or in combination of 2 or more types.
- (Meth) acryloyl halide (A3) is preferably, for example, (meth) acryloyl chloride, (meth) acryloyl bromide, or (meth) acryloyl iodide.
- the (meth) acryloyl halide (A3) may have a linear or branched alkyl group having 1 to 4 carbon atoms, a cyano group, a hydroxyl group, a halogen or the like as long as the effects of the present invention are not impaired.
- the (meth) acryloyl halide (A3) may be used alone or in combination of two or more.
- the cyclic compound of the present embodiment can be produced, for example, by the following method.
- an acid catalyst hydroochloric acid, sulfuric acid, paratoluenesulfonic acid, etc.
- organic solvent such as methanol or ethanol
- 0.1 mol of the phenolic compound (A2) is added to 1 mol of the aldehyde compound (A1).
- ⁇ 10 mol is reacted at 60 to 150 ° C. for about 0.5 to 20 hours.
- filtration, washing with an alcohol such as methanol, washing with water, separation by filtration, and drying are performed to obtain a cyclic compound (A) having a molecular weight of preferably 700 to 5000.
- a basic catalyst sodium hydroxide, barium hydroxide or 1,8-diazabicyclo [5.4.0] undecene-7 etc.
- the cyclic compound (A) Is obtained.
- the cyclic compound (A) can also be produced by converting the aldehyde compound (A1) into a dihalide with hydrogen halide or halogen gas, and reacting the isolated dihalide with the phenolic compound (A2).
- (meth) acryloyl halide (A3) is added to 1 mol of the cyclic compound (A).
- a base catalyst such as triethylamine, ammonia or sodium hydroxide
- organic solvent such as N-methyl-2-pyrrolidone
- (meth) acryloyl halide (A3) is added to 1 mol of the cyclic compound (A).
- 0.1-100 mol is reacted at 0-150 ° C. for about 0.5-20 hours.
- the phenolic hydroxyl group in the cyclic compound (A) can be converted into a (meth) acryloyl group.
- the cyclic compound shown by Formula (1) is obtained by drying after filtration, washing with alcohols such as methanol, washing with water, and separation by filtration.
- the solubility of the obtained cyclic compound in the semiconductor safety solvent is improved, it is more preferable to use at least one of the aldehyde compound (A1) or the phenolic compound (A2).
- a purification step may be performed as necessary in order to improve the purity in the cyclic compound and reduce the amount of remaining metal. Further, if the acid catalyst and the co-catalyst remain, generally the storage stability of the radiation-sensitive composition is lowered, and if the basic catalyst remains, the sensitivity of the radiation-sensitive composition is generally lowered. Purification may be performed.
- the purification method can be performed by a known method as long as the cyclic compound is not denatured, and is not particularly limited.
- a method of washing with water a method of washing with an acidic aqueous solution, a method of washing with a basic aqueous solution, ion exchange Examples include a method of treating with a resin and a method of treating with silica gel column chromatography.
- These purification methods are more preferably performed in combination of two or more.
- acidic aqueous solution, basic aqueous solution, ion exchange resin, and silica gel column chromatography select the optimal one according to the metal to be removed, the amount and type of acidic compound and basic compound, the type of cyclic compound to be purified, etc.
- Amberlyst 15J-HG Dry made by Organo can be mentioned. You may dry after refinement
- cyclic compound of this embodiment When the cyclic compound of this embodiment is used as a component of a radiation sensitive composition, an amorphous film can be formed by spin coating. Moreover, the cyclic compound of this embodiment can be applied to a general semiconductor manufacturing process.
- the cyclic compound of the present embodiment is useful as a negative resist material that becomes a compound that is hardly soluble in a developer by irradiation with a KrF excimer laser, extreme ultraviolet light, electron beam, or X-ray.
- the mechanism by which the cyclic compound of the present embodiment becomes a compound that is hardly soluble in the developer is not clear, but the cyclic reaction between the compounds by irradiating the cyclic compound with KrF excimer laser, extreme ultraviolet light, electron beam or X-ray. Is considered to be a compound that is hardly soluble in an alkaline developer.
- the resist pattern obtained using the cyclic compound of this embodiment has a very small line edge roughness (LER).
- the cyclic compound of the present embodiment can be used as a main component of a negative radiation-sensitive composition, and can be added to the radiation-sensitive composition as an additive for improving sensitivity and etching resistance, for example. it can.
- the use amount of the cyclic compound of the present embodiment as an additive is preferably 1 to 49.999% by weight based on the total solid component weight of the radiation-sensitive composition.
- the glass transition temperature of the cyclic compound of the present embodiment is preferably 100 ° C. or higher, more preferably 120 ° C. or higher, still more preferably 140 ° C. or higher, and particularly preferably 150 ° C. or higher.
- the semiconductor lithography process has heat resistance capable of maintaining a pattern shape, and performance such as high resolution is improved.
- the crystallization calorific value obtained by differential scanning calorimetric analysis of the glass transition temperature of the cyclic compound of the present embodiment is preferably less than 20 J / g.
- (crystallization temperature) ⁇ (glass transition temperature) is preferably 70 ° C. or higher, more preferably 80 ° C. or higher, further preferably 100 ° C. or higher, particularly preferably 130 ° C. or higher. is there.
- the crystallization heat generation amount is less than 20 J / g, or (crystallization temperature) ⁇ (glass transition temperature) is within the above range, an amorphous film can be easily formed by spin coating the radiation-sensitive composition.
- the film formability required for the resist can be maintained for a long time, and the resolution can be improved.
- the crystallization calorific value, the crystallization temperature, and the glass transition temperature can be determined as follows by differential scanning calorimetry using DSC / TA-50WS manufactured by Shimadzu Corporation.
- About 10 mg of a sample is put in an aluminum non-sealed container and heated to a melting point or higher at a temperature rising rate of 20 ° C./min in a nitrogen gas stream (50 mL / min).
- the temperature is raised to the melting point or higher at a heating rate of 20 ° C./min in a nitrogen gas stream (30 mL / min) again. Further, after rapid cooling, the temperature is increased again to 400 ° C.
- the temperature at the midpoint of the step difference of the baseline that has changed in a step shape (where the specific heat has changed to half) is the glass transition temperature (Tg), and the temperature of the exothermic peak that appears thereafter is the crystallization temperature.
- Tg glass transition temperature
- the calorific value is obtained from the area of the region surrounded by the exothermic peak and the baseline, and is defined as the crystallization calorific value.
- the cyclic compound of the present embodiment preferably has low sublimation property under normal pressure at 100 ° C. or lower, preferably 120 ° C. or lower, more preferably 130 ° C. or lower, further preferably 140 ° C. or lower, particularly preferably 150 ° C. or lower.
- the low sublimation property means that in thermogravimetric analysis, the weight loss when held at a predetermined temperature for 10 minutes is 10%, preferably 5%, more preferably 3%, still more preferably 1%, particularly preferably. It shows that it is 0.1% or less. Since the sublimation property is low, it is possible to prevent exposure apparatus from being contaminated by outgas during exposure. Moreover, a favorable pattern shape can be obtained with low LER.
- the cyclic compound of the present embodiment is preferably F ⁇ 3.0 (F represents the total number of atoms / (total number of carbon atoms ⁇ total number of oxygen atoms)), and more preferably F ⁇ 2.5. is there.
- the cyclic compound of this embodiment is excellent in dry etching resistance by satisfying the above conditions.
- the cyclic compound of the present embodiment includes propylene glycol monomethyl ether acetate (PGMEA), propylene glycol monomethyl ether (PGME), cyclohexanone (CHN), cyclopentanone (CPN), 2-heptanone, anisole, butyl acetate, ethyl propionate and In a solvent selected from ethyl lactate and exhibiting the highest solubility for cyclic compounds, it is preferably dissolved at 23 ° C., preferably 1% by weight or more, more preferably 5% by weight or more, and even more preferably 10% by weight or more.
- cyclic compound of this embodiment can be used in a semiconductor manufacturing process in actual production by satisfying the above conditions.
- a nitrogen atom may be introduced into the cyclic compound of the present embodiment as long as the effects of the present invention are not impaired.
- the ratio of the number of nitrogen atoms to the total number of constituent atoms of the cyclic compound is preferably 0.1 to 40%, more preferably 0.1 to 20%, and preferably 0.1 to 10%. Further preferred is 0.1 to 5%.
- transduced into the cyclic compound of this embodiment is a secondary or tertiary nitrogen atom, and it is more preferable that it is a tertiary nitrogen atom.
- the cyclic compound of the present embodiment is irradiated with visible light, ultraviolet light, excimer laser, electron beam, extreme ultraviolet light (EUV), X-ray and ion beam, or a chemical reaction induced thereby.
- a crosslinking reactive group that causes a crosslinking reaction may be introduced. Introduction of the crosslinking reactive group into the cyclic compound of the present embodiment is performed, for example, by reacting the cyclic compound and the crosslinking reactive group introduction reagent in the presence of a base catalyst.
- the crosslinking reactive group is not particularly limited, and examples thereof include a carbon-carbon multiple bond, an epoxy group, an azide group, a halogenated phenyl group, and a chloromethyl group.
- the crosslinking reactive group introduction reagent is not particularly limited, and examples thereof include carboxylic acid derivatives such as acids, acid chlorides, acid anhydrides, dicarbonates, and alkyl halides having such a crosslinking reactive group.
- a radiation-sensitive composition containing a cyclic compound having a crosslinking reactive group is also useful as a non-polymeric radiation-sensitive composition having high resolution, high heat resistance and solvent solubility.
- a non-acid-dissociable functional group may be introduced into at least one of the phenolic hydroxyl groups as long as the effects of the present invention are not impaired.
- the non-acid-dissociable functional group refers to a characteristic group that does not cleave in the presence of an acid and does not generate an alkali-soluble group.
- Non-acid-dissociable functional groups include, for example, C1-20 alkyl groups, C3-20 cycloalkyl groups, C6-20 aryl groups, C1-20 alkoxyl groups that are not decomposed by the action of an acid, Examples thereof include a functional group selected from the group consisting of a cyano group, a nitro group, a hydroxyl group, a heterocyclic group, a halogen, a carboxyl group, a C1-20 alkylsilane, and derivatives thereof.
- a naphthoquinone diazide ester group may be introduced into at least one of the phenolic hydroxyl groups as long as the effects of the present invention are not impaired.
- a cyclic compound having a naphthoquinonediazide ester group introduced into at least one phenolic hydroxyl group can be used as a main component of a negative radiation-sensitive composition as a main component, or as a main component of a positive radiation-sensitive composition. It can be used or added to the radiation sensitive composition as an acid generator or additive.
- an acid-generating functional group that generates an acid upon irradiation with radiation may be introduced into at least one of the phenolic hydroxyl groups in the cyclic compound of the present embodiment.
- a cyclic compound in which an acid-generating functional group is introduced into at least one phenolic hydroxyl group can be used as a main component of a negative radiation-sensitive composition by itself as a main component, and a radiation-sensitive composition as an additive. Can be added to.
- the radiation-sensitive composition according to this embodiment contains a solid component containing the aforementioned cyclic compound and a solvent.
- the solid component is preferably 1 to 80% by weight, and the ratio of the cyclic compound to the total weight of the solid component is 50 to 99.999% by weight. More preferably.
- the radiation sensitive composition of the present embodiment can form an amorphous film by spin coating.
- the dissolution rate of the amorphous film formed by spin-coating the radiation-sensitive composition of the present embodiment in a developer at 23 ° C. is preferably 10 ⁇ / sec or more, more preferably 10 to 10000 ⁇ / sec. More preferably, it is 100 to 1000 kg / sec.
- the dissolution rate is 10 ⁇ / sec or more
- the radiation-sensitive composition of the present embodiment can be dissolved in a developer and used as a resist.
- the resolution of the radiation-sensitive composition of the present embodiment may be improved when the dissolution rate is 10,000 kg / sec or less.
- the dissolution rate of the amorphous film formed by spin-coating the radiation-sensitive composition of the present embodiment in a developer at 23 ° C. at a portion exposed by radiation such as KrF excimer laser, extreme ultraviolet light, electron beam or X-ray is It is preferably 5 K / sec or less, more preferably 0.05 to 5 K / sec, and further preferably 0.0005 to 5 K / sec.
- the radiation-sensitive composition of the present embodiment is insoluble in a developer when the dissolution rate is 5 kg / sec or less, and can be used as a resist. Further, when the dissolution rate of the radiation-sensitive composition of the present embodiment is 0.0005 K / sec or more, the resolution may be improved. This is presumed to be because the micro surface portion of the cyclic compound is dissolved and LER is reduced. There is also an effect of reducing defects.
- the radiation-sensitive composition of the present embodiment is preferably 1 to 80% by weight of the solid component and 20 to 99% by weight of the solvent, more preferably 1 to 50% by weight of the solid component, and 50% of the solvent. It is ⁇ 99 wt%, more preferably 2 to 40 wt% solid component and 60 to 98 wt% solvent, particularly preferably 2 to 10 wt% solid component and 90 to 98 wt% solvent.
- the amount of the above-mentioned cyclic compound is the total weight of the solid component (the above-mentioned cyclic compound, the acid generator (C), the acid cross-linking agent (G), and the acid diffusion controller described later). 50 to 99.4% by weight, more preferably 55 to 90% by weight of (E) and the sum of solid components optionally used such as other components (F), and so on)
- the amount is preferably 60 to 80% by weight, particularly preferably 60 to 70% by weight.
- the radiation-sensitive composition of this embodiment is an acid directly or indirectly by irradiation with radiation selected from visible light, ultraviolet light, excimer laser, electron beam, extreme ultraviolet light (EUV), X-ray and ion beam. It is preferable to include one or more acid generators (C) that generate water.
- the content of the acid generator (C) is preferably 0.001 to 49% by weight, more preferably 1 to 40% by weight, still more preferably 3 to 30% by weight, and more preferably 10 to 25% by weight based on the total weight of the solid components. Particularly preferred.
- a pattern profile with high sensitivity and low edge roughness can be obtained when the content of the acid generator (C) is within the above range.
- the acid generation method is not limited as long as the acid is generated in the system. If excimer laser is used instead of ultraviolet rays such as g-line and i-line, finer processing is possible, and if high-energy rays are used electron beams, extreme ultraviolet rays, X-rays, ion beams, further fine processing is possible. Is possible.
- the acid generator (C) is preferably at least one selected from the group consisting of compounds represented by the following formulas (7-1) to (7-8).
- R 13 may be the same or different, and each independently represents a hydrogen atom, a linear, branched or cyclic alkyl group, a linear, branched or cyclic alkoxy group, A hydroxyl group or a halogen atom;
- X ⁇ is a sulfonate ion or a halide ion having an alkyl group, an aryl group, a halogen-substituted alkyl group or a halogen-substituted aryl group.
- the compound represented by the formula (7-1) includes triphenylsulfonium trifluoromethanesulfonate, triphenylsulfonium nonafluoro-n-butanesulfonate, diphenyltolylsulfonium nonafluoro-n-butanesulfonate, triphenylsulfonium perfluoro-n- Octane sulfonate, diphenyl-4-methylphenylsulfonium trifluoromethanesulfonate, di-2,4,6-trimethylphenylsulfonium trifluoromethanesulfonate, diphenyl-4-t-butoxyphenylsulfonium trifluoromethanesulfonate, diphenyl-4-t-butoxyphenyl Sulfonium nonafluoro-n-butanesulfonate, diphenyl-4-hydroxyphenylsulfonium trifluoromethane Sulfon
- R 14 s may be the same or different and each independently represents a hydrogen atom, a linear, branched or cyclic alkyl group, a linear, branched or cyclic alkoxy group, Represents a hydroxyl group or a halogen atom, X ⁇ is as defined above.
- the compound represented by the formula (7-2) includes bis (4-t-butylphenyl) iodonium trifluoromethanesulfonate, bis (4-t-butylphenyl) iodonium nonafluoro-n-butanesulfonate, bis (4-t -Butylphenyl) iodonium perfluoro-n-octanesulfonate, bis (4-t-butylphenyl) iodonium-p-toluenesulfonate, bis (4-t-butylphenyl) iodoniumbenzenesulfonate, bis (4-t-butylphenyl) ) Iodonium-2-trifluoromethylbenzenesulfonate, bis (4-tert-butylphenyl) iodonium-4-trifluoromethylbenzenesulfonate, bis (4-tert-butylphenyl) iodonium-2,4-
- Q represents an alkylene group, an arylene group or an alkoxylene group
- R 15 represents an alkyl group, an aryl group, a halogen-substituted alkyl group or a halogen-substituted aryl group.
- the compound represented by the formula (7-3) includes N- (trifluoromethylsulfonyloxy) succinimide, N- (trifluoromethylsulfonyloxy) phthalimide, N- (trifluoromethylsulfonyloxy) diphenylmaleimide, N- ( Trifluoromethylsulfonyloxy) bicyclo [2.2.1] hept-5-ene-2,3-dicarboximide, N- (trifluoromethylsulfonyloxy) naphthylimide, N- (10-camphorsulfonyloxy) Succinimide, N- (10-camphorsulfonyloxy) phthalimide, N- (10-camphorsulfonyloxy) diphenylmaleimide, N- (10-camphorsulfonyloxy) bicyclo [2.2.1] hept-5-ene-2 , 3-Dicarboximide, N (10-camphorsulfonyloxy) naph
- R 16 may be the same or different, and each independently represents an optionally substituted linear, branched or cyclic alkyl group, an optionally substituted aryl group, and optionally substituted. A heteroaryl group or an optionally substituted aralkyl group.
- the compound represented by the formula (7-4) is diphenyl disulfone, di (4-methylphenyl) disulfone, dinaphthyl disulfone, di (4-tert-butylphenyl) disulfone, di (4-hydroxyphenyl) disulfone. At least one selected from the group consisting of di (3-hydroxynaphthyl) disulfone, di (4-fluorophenyl) disulfone, di (2-fluorophenyl) disulfone and di (4-trifluoromethylphenyl) disulfone It is preferable.
- R 17 may be the same or different and each independently represents an optionally substituted linear, branched or cyclic alkyl group, an optionally substituted aryl group, and optionally substituted. A heteroaryl group or an optionally substituted aralkyl group.
- the compound represented by the formula (7-5) is ⁇ - (methylsulfonyloxyimino) -phenylacetonitrile, ⁇ - (methylsulfonyloxyimino) -4-methoxyphenylacetonitrile, ⁇ - (trifluoromethylsulfonyloxyimino).
- R 18 may be the same or different and each independently represents a halogenated alkyl group having one or more chlorine atoms and one or more bromine atoms.
- the halogenated alkyl group preferably has 1 to 5 carbon atoms.
- R 19 and R 20 are each independently an alkyl group having 1 to 3 carbon atoms such as a methyl group, an ethyl group, an n-propyl group, an isopropyl group, or cyclopentyl.
- Group, cycloalkyl group such as cyclohexyl group, alkoxyl group having 1 to 3 carbon atoms such as methoxy group, ethoxy group, propoxy group, or aryl group such as phenyl group, toluyl group, naphthyl group, preferably 6 carbon atoms ⁇ 10 aryl groups.
- L 19 and L 20 are each independently an organic group having a 1,2-naphthoquinonediazide group.
- Specific examples of the organic group having a 1,2-naphthoquinonediazide group include a 1,2-naphthoquinonediazide-4-sulfonyl group, a 1,2-naphthoquinonediazide-5-sulfonyl group, and a 1,2-naphthoquinonediazide- Preferred examples include 1,2-quinonediazidosulfonyl groups such as a 6-sulfonyl group.
- 1,2-naphthoquinonediazido-4-sulfonyl group and 1,2-naphthoquinonediazide-5-sulfonyl group are preferable.
- p is an integer of 1 to 3
- q is an integer of 0 to 4
- 1 ⁇ p + q ⁇ 5 is preferable.
- J 19 is a single bond, a polymethylene group having 1 to 4 carbon atoms, a cycloalkylene group, a phenylene group, a group represented by the following formula (7-7-1), a carbonyl group, an ester group, an amide group or an ether group.
- Y 19 represents a hydrogen atom, an alkyl group or an aryl group
- X 20 each independently represents a group represented by the following formula (7-8-1).
- Z 22 each independently represents an alkyl group, a cycloalkyl group or an aryl group, R 22 represents an alkyl group, a cycloalkyl group or an alkoxyl group, and r represents 0 to 3 Is an integer.
- Examples of other acid generators include, but are not limited to, bis (p-toluenesulfonyl) diazomethane, bis (2,4-dimethylphenylsulfonyl) diazomethane, bis (tert-butylsulfonyl) diazomethane, bis (n-butylsulfonyl) ) Diazomethane, bis (isobutylsulfonyl) diazomethane, bis (isopropylsulfonyl) diazomethane, bis (n-propylsulfonyl) diazomethane, bis (cyclohexylsulfonyl) diazomethane, bis (isopropylsulfonyl) diazomethane, 1,3-bis (cyclohexylsulfonylazomethyl) Sulfonyl) propane, 1,4-bis (phenylsulfonylazomethylsulfony
- acid generators having an aromatic ring are preferable, and acid generators represented by formula (7-1) or (7-2) are more preferable.
- An acid generator having a sulfonate ion having X ⁇ in formula (7-1) or (7-2) having an aryl group or a halogen-substituted aryl group is more preferred, and an acid generator having a sulfonate ion having an aryl group are particularly preferable, and diphenyltrimethylphenylsulfonium-p-toluenesulfonate, triphenylsulfonium-p-toluenesulfonate, triphenylsulfonium trifluoromethanesulfonate, and triphenylsulfonium nonafluoromethanesulfonate are particularly preferable.
- the acid generator (C) can be used alone or in combination of two or more.
- the radiation-sensitive composition of the present embodiment preferably contains one or more acid crosslinking agents (G).
- the acid cross-linking agent (G) is a compound that can cross-link the above-mentioned cyclic compound within a molecule or between molecules in the presence of an acid generated from the acid generator (C).
- Examples of such an acid crosslinking agent (G) include compounds having one or more groups (hereinafter referred to as “crosslinkable groups”) capable of crosslinking the cyclic compound of the formula (1).
- crosslinkable group examples are not particularly limited. For example, (i) hydroxy (C1-C6 alkyl group), C1-C6 alkoxy (C1-C6 alkyl group), acetoxy (C1-C6 alkyl group) ) Or a group derived therefrom; (ii) a carbonyl group such as formyl group or carboxy (C1-C6 alkyl group) or a group derived therefrom; (iii) dimethylaminomethyl group, diethylaminomethyl Group, nitrogen-containing group such as dimethylolaminomethyl group, diethylolaminomethyl group, morpholinomethyl group; (iv) glycidyl group-containing group such as glycidyl ether group, glycidyl ester group, glycidylamino group; (v) benzyl C1-C6 allyloxy (C1-C) such as oxymethyl group and benzoyloxymethyl group
- the acid crosslinking agent having a crosslinkable group (G) is not particularly limited.
- the acid crosslinking agent (G) compounds having phenolic hydroxyl groups, and compounds and resins imparted with crosslinking properties by introducing the crosslinking groups into acidic functional groups in the alkali-soluble resin can be used.
- the introduction ratio of the crosslinkable group is usually 5 to 100 mol%, preferably 10 to 60 mol%, more preferably based on the total acidic functional group in the compound having a phenolic hydroxyl group and the alkali-soluble resin. Is adjusted to 15-40 mol%. It is preferable for the introduction rate of the crosslinkable group to be in the above-mentioned range since a sufficient crosslinking reaction occurs and a decrease in the remaining film rate, a pattern swelling phenomenon, meandering, and the like are avoided.
- the acid crosslinking agent (G) is preferably an alkoxyalkylated urea compound or a resin thereof, or an alkoxyalkylated glycoluril compound or a resin thereof.
- Particularly preferred acid crosslinking agents (G) include compounds represented by the following formulas (8-1) to (8-3) and alkoxymethylated melamine compounds (acid crosslinking agent (G1)).
- R 7 each independently represents a hydrogen atom, an alkyl group or an acyl group
- R 8 to R 11 each independently represents a hydrogen atom, a hydroxyl group
- And represents an alkyl group or an alkoxyl group
- X 2 represents a single bond, a methylene group or an oxygen atom.
- the alkyl group represented by R 7 preferably has 1 to 6 carbon atoms, more preferably 1 to 3 carbon atoms, and examples thereof include a methyl group, an ethyl group, and a propyl group.
- the acyl group represented by R 7 preferably has 2 to 6 carbon atoms, more preferably 2 to 4 carbon atoms, and examples thereof include an acetyl group and a propionyl group.
- the alkyl group represented by R 8 to R 11 preferably has 1 to 6 carbon atoms, more preferably 1 to 3 carbon atoms, and examples thereof include a methyl group, an ethyl group, and a propyl group.
- the alkoxyl group represented by R 8 to R 11 preferably has 1 to 6 carbon atoms, more preferably 1 to 3 carbon atoms, and examples thereof include a methoxy group, an ethoxy group, and a propoxy group.
- X 2 is preferably a single bond or a methylene group.
- R 7 to R 11 and X 2 may be substituted with an alkyl group such as a methyl group or an ethyl group, an alkoxy group such as a methoxy group or an ethoxy group, a hydroxyl group, or a halogen atom.
- the plurality of R 7 and R 8 to R 11 may be the same or different.
- Specific examples of the compound represented by the formula (8-2) include, but are not limited to, N, N, N, N, N-tetra (methoxymethyl) glycoluril, N, N, N, N-tetra ( Ethoxymethyl) glycoluril, N, N, N, N-tetra (n-propoxymethyl) glycoluril, N, N, N-tetra (isopropoxymethyl) glycoluril, N, N, N, N-tetra (N-Butoxymethyl) glycoluril, N, N, N, N-tetra (t-butoxymethyl) glycoluril and the like can be mentioned. Of these, N, N, N, N-tetra (methoxymethyl) glycoluril is particularly preferable.
- alkoxymethylated melamine compounds include N, N, N, N, N, N-hexa (methoxymethyl) melamine, N, N, N, N, N-hexa (ethoxymethyl) melamine N, N, N, N, N-hexa (n-propoxymethyl) melamine, N, N, N, N, N-hexa (isopropoxymethyl) melamine, N, N, N, N, Examples thereof include N, N-hexa (n-butoxymethyl) melamine, N, N, N, N, N-hexa (t-butoxymethyl) melamine and the like. Among these, N, N, N, N, N, N-hexa (methoxymethyl) melamine is particularly preferable.
- the acid cross-linking agent (G1) is obtained by, for example, condensing a urea compound or glycoluril compound and formalin to introduce a methylol group, and then ether with lower alcohols such as methyl alcohol, ethyl alcohol, propyl alcohol, and butyl alcohol. Then, the reaction solution is cooled and the precipitated compound or its resin is recovered.
- the acid cross-linking agent (G1) can also be obtained as a commercial product such as CYMEL (trade name, manufactured by Mitsui Cyanamid) or Nicalac (manufactured by Sanwa Chemical Co., Ltd.).
- the molecule has 1 to 6 benzene rings, and has at least two hydroxyalkyl groups and / or alkoxyalkyl groups in the molecule. And / or a phenol derivative in which an alkoxyalkyl group is bonded to any one of the benzene rings (acid crosslinking agent (G2)).
- the molecular weight is 1500 or less
- the molecule has 1 to 6 benzene rings
- the hydroxyalkyl group and / or alkoxyalkyl group has 2 or more in total
- the hydroxyalkyl group and / or alkoxyalkyl group is the benzene ring.
- Mention may be made of phenol derivatives formed by bonding to any one or a plurality of benzene rings.
- hydroxyalkyl group bonded to the benzene ring those having 1 to 6 carbon atoms such as hydroxymethyl group, 2-hydroxyethyl group, and 2-hydroxy-1-propyl group are preferable.
- the alkoxyalkyl group bonded to the benzene ring is preferably one having 2 to 6 carbon atoms. Specifically, methoxymethyl group, ethoxymethyl group, n-propoxymethyl group, isopropoxymethyl group, n-butoxymethyl group, isobutoxymethyl group, sec-butoxymethyl group, t-butoxymethyl group, 2-methoxyethyl Group or 2-methoxy-1-propyl group is preferred.
- L 1 to L 8 may be the same or different and each independently represents a hydroxymethyl group, a methoxymethyl group or an ethoxymethyl group.
- a phenol derivative having a hydroxymethyl group is obtained by reacting a phenol compound not having a corresponding hydroxymethyl group (a compound in which L 1 to L 8 are hydrogen atoms in the above formula) with formaldehyde in the presence of a base catalyst. Can do.
- the reaction temperature is preferably 60 ° C. or lower. Specifically, it can be synthesized by the methods described in JP-A-6-282067, JP-A-7-64285 and the like.
- a phenol derivative having an alkoxymethyl group can be obtained by reacting a corresponding phenol derivative having a hydroxymethyl group with an alcohol in the presence of an acid catalyst.
- the reaction temperature is preferably 100 ° C. or lower. Specifically, it can be synthesized by the method described in EP632003A1 and the like.
- a phenol derivative having a hydroxymethyl group and / or an alkoxymethyl group synthesized in this manner is preferable in terms of stability during storage, but a phenol derivative having an alkoxymethyl group is particularly preferable from the viewpoint of stability during storage.
- the acid crosslinking agent (G2) may be used alone or in combination of two or more.
- Another particularly preferable acid crosslinking agent (G) is a compound having at least one ⁇ -hydroxyisopropyl group (acid crosslinking agent (G3)).
- the structure is not particularly limited as long as it has an ⁇ -hydroxyisopropyl group.
- the hydrogen atom of the hydroxyl group in the ⁇ -hydroxyisopropyl group is one or more acid dissociable groups (R—COO— group, R—SO 2 — group, etc., R is a straight chain having 1 to 12 carbon atoms)
- R—COO— group, R—SO 2 — group, etc. R is a straight chain having 1 to 12 carbon atoms
- a hydrocarbon group having 3 to 12 carbon atoms, an alkoxy group having 1 to 12 carbon atoms, a 1-branched alkyl group having 3 to 12 carbon atoms and an aromatic hydrocarbon group having 6 to 12 carbon atoms (Represents a substituent selected from the group).
- the compound having an ⁇ -hydroxyisopropyl group is not particularly limited, and examples thereof include substituted or unsubstituted aromatic compounds containing at least one ⁇ -hydroxyisopropyl group, diphenyl compounds, naphthalene compounds, and furan compounds. 1 type or 2 types or more are mentioned.
- benzene compound (1) (hereinafter referred to as “benzene compound (1)”), represented by the following general formula (9-2): Compound (hereinafter referred to as “diphenyl compound (2)”), a compound represented by the following general formula (9-3) (hereinafter referred to as “naphthalene compound (3)”), and the following general formula And a compound represented by (9-4) (hereinafter referred to as “furan compound (4)”).
- benzene compound (1) represented by the following general formula (9-2): Compound (hereinafter referred to as “diphenyl compound (2)”), a compound represented by the following general formula (9-3) (hereinafter referred to as “naphthalene compound (3)”), and the following general formula And a compound represented by (9-4) (hereinafter referred to as “furan compound (4)”).
- each A 2 independently represents an ⁇ -hydroxyisopropyl group or a hydrogen atom, and at least one A 2 is an ⁇ -hydroxyisopropyl group.
- R 51 represents a hydrogen atom, a hydroxyl group, a linear or branched alkylcarbonyl group having 2 to 6 carbon atoms, or a linear or branched structure having 2 to 6 carbon atoms. An alkoxycarbonyl group is shown.
- R 52 represents a single bond, a linear or branched alkylene group having 1 to 5 carbon atoms, —O—, —CO— or —COO—.
- R 53 and R 54 each independently represent a hydrogen atom or a linear or branched alkyl group having 1 to 6 carbon atoms.
- benzene compound (1) examples include, but are not limited to, ⁇ -hydroxyisopropylbenzene, 1,3-bis ( ⁇ -hydroxyisopropyl) benzene, 1,4-bis ( ⁇ -hydroxyisopropyl).
- ⁇ -hydroxyisopropylbenzenes such as benzene, 1,2,4-tris ( ⁇ -hydroxyisopropyl) benzene, 1,3,5-tris ( ⁇ -hydroxyisopropyl) benzene; 3- ⁇ -hydroxyisopropylphenol, 4- ⁇ -hydroxyisopropylphenols such as ⁇ -hydroxyisopropylphenol, 3,5-bis ( ⁇ -hydroxyisopropyl) phenol, 2,4,6-tris ( ⁇ -hydroxyisopropyl) phenol; 3- ⁇ -hydroxyisopropylphenyl Methyl ketone, 4- ⁇ -hy Roxyisopropylphenyl methyl ketone, 4- ⁇ -hydroxyisopropylphenyl ethyl ketone, 4- ⁇ -hydroxyisopropylphenyl n-propyl ketone, 4- ⁇ -hydroxyisopropylphenyl isopropyl ketone, 4- ⁇ -hydroxyisopropylphenyl n- But
- diphenyl compound (2) is not particularly limited, but examples thereof include 3- ⁇ -hydroxyisopropylbiphenyl, 4- ⁇ -hydroxyisopropylbiphenyl, 3,5-bis ( ⁇ -hydroxyisopropyl) biphenyl.
- the naphthalene-based compound (3) is not specifically limited.
- the furan compound (4) is not particularly limited, but examples thereof include 3- ( ⁇ -hydroxyisopropyl) furan, 2-methyl-3- ( ⁇ -hydroxyisopropyl) furan, 2-methyl- 4- ( ⁇ -hydroxyisopropyl) furan, 2-ethyl-4- ( ⁇ -hydroxyisopropyl) furan, 2-n-propyl-4- ( ⁇ -hydroxyisopropyl) furan, 2-isopropyl-4- ( ⁇ -hydroxy) Isopropyl) furan, 2-n-butyl-4- ( ⁇ -hydroxyisopropyl) furan, 2-t-butyl-4- ( ⁇ -hydroxyisopropyl) furan, 2-n-pentyl-4- ( ⁇ -hydroxyisopropyl) Furan, 2,5-dimethyl-3- ( ⁇ -hydroxyisopropyl) furan, 2,5-diethyl-3- ( ⁇ -hydroxy) Cyisopropyl) furan, 3,4-bis ( ⁇ -hydroxyisopropyl) furan, 3,
- the acid crosslinking agent (G3) is preferably a compound having two or more free ⁇ -hydroxyisopropyl groups, the benzene compound (1) having two or more ⁇ -hydroxyisopropyl groups, and two or more ⁇ -hydroxyisopropyl groups. More preferably, the diphenyl compound (2) having two or more ⁇ -hydroxyisopropyl groups, and the naphthalene compound (3) having two or more ⁇ -hydroxyisopropyl groups, ⁇ -hydroxyisopropylbiphenyls having two or more ⁇ -hydroxyisopropyl groups, ⁇ -hydroxy A naphthalene compound (3) having two or more isopropyl groups is particularly preferred.
- the acid cross-linking agent (G3) is usually obtained by a method in which a acetyl group-containing compound such as 1,3-diacetylbenzene is reacted with a Grignard reagent such as CH 3 MgBr to be methylated and then hydrolyzed. It can be obtained by a method in which an isopropyl group-containing compound such as diisopropylbenzene is oxidized with oxygen or the like to generate a peroxide and then reduced.
- the content of the acid crosslinking agent (G) is preferably 0.5 to 49% by weight, more preferably 0.5 to 40% by weight, based on the total weight of the solid component, 1 to 30% by weight is more preferred, and 2 to 20% by weight is particularly preferred.
- the content of the acid crosslinking agent (G) is 0.5% by weight or more, the radiation-sensitive composition of the present embodiment improves the effect of suppressing the solubility of the resist film in an alkaline developer, and the remaining film ratio
- the content of the acid cross-linking agent (G) is 50% by weight or less. It is preferable because the decrease can be suppressed.
- the blending ratio of at least one compound selected from the acid crosslinking agent (G1), the acid crosslinking agent (G2) and the acid crosslinking agent (G3) in the acid crosslinking agent (G) is not particularly limited. Various ranges can be used depending on the type of substrate used when forming the pattern.
- the content of the alkoxymethylated melamine compound and / or the compound represented by (9-1) to (9-3) is 50 to 99% by weight, preferably 60 to 99% by weight, More preferred is 70 to 98% by weight, and still more preferred is 80 to 97% by weight.
- the content of the alkoxymethylated melamine compound and / or the compounds represented by (9-1) to (9-3) is 50% by weight or more of the total acid crosslinking agent component. Therefore, the resolution can be improved, and it is preferable to set it to 99% by weight or less because it is easy to obtain a rectangular cross-sectional shape as the pattern cross-sectional shape.
- the radiation-sensitive composition of the present embodiment may further contain an acid diffusion controller (E).
- the acid diffusion control agent (E) is a compound having a function of controlling an undesired chemical reaction in an unexposed region by controlling diffusion of an acid generated from an acid generator by irradiation in a resist film. .
- the radiation sensitive composition of this embodiment improves storage stability by containing such an acid diffusion controller (E). In addition, the resolution is improved, and a change in the line width of the resist pattern due to fluctuations in the holding time before irradiation and the holding time after irradiation can be suppressed, and the process stability is extremely excellent.
- the acid diffusion controller (E) is not particularly limited, and examples thereof include radiolytic decomposable basic compounds such as nitrogen atom-containing basic compounds, basic sulfonium compounds, and basic iodonium compounds.
- the acid diffusion controller (E) can be used alone or in combination of two or more.
- the acid diffusion control agent (E) is not particularly limited, and examples thereof include nitrogen-containing organic compounds and basic compounds that are decomposed by exposure.
- examples of the nitrogen-containing organic compound include the following general formula (10):
- nitrogen-containing compound (I) a diamino compound having two nitrogen atoms in the same molecule
- nitrogen-containing compound (II) a diamino compound having two nitrogen atoms in the same molecule
- nitrogen-containing compound (II) a diamino compound having two nitrogen atoms in the same molecule
- nitrogen-containing compound (III) polyamino compounds and polymers having three or more compounds
- nitrogen-containing compound (III) examples thereof include polyamino compounds and polymers having three or more compounds
- amide group-containing compounds amide group-containing compounds
- urea compounds urea compounds
- nitrogen-containing heterocyclic compounds nitrogen-containing heterocyclic compounds
- an acid diffusion control agent (E) may be used individually by 1 type, and may use 2 or more types together.
- R 61 , R 62 and R 63 each independently represent a hydrogen atom, a linear, branched or cyclic alkyl group, an aryl group or an aralkyl group.
- the alkyl group, aryl group or aralkyl group may be unsubstituted or substituted with a hydroxyl group or the like.
- the linear, branched or cyclic alkyl group is not particularly limited, and examples thereof include those having 1 to 15 carbon atoms, preferably 1 to 10 carbon atoms.
- the aryl group is not particularly limited, and examples thereof include those having 6 to 12 carbon atoms.
- aralkyl group is not particularly limited, and examples thereof include those having 7 to 19 carbon atoms, preferably 7 to 13 carbon atoms.
- Specific examples include benzyl group, ⁇ -methylbenzyl group, phenethyl group, naphthyl group. A methyl group etc. are mentioned.
- nitrogen-containing compound (I) examples include, but are not particularly limited to, for example, n-hexylamine, n-heptylamine, n-octylamine, n-nonylamine, n-decylamine, n-dodecylamine, cyclohexylamine.
- Mono- (cyclo) alkylamines such as: di-n-butylamine, di-n-pentylamine, di-n-hexylamine, di-n-heptylamine, di-n-octylamine, di-n-nonylamine, Di (cyclo) alkylamines such as di-n-decylamine, methyl-n-dodecylamine, di-n-dodecylmethyl, cyclohexylmethylamine, dicyclohexylamine; triethylamine, tri-n-propylamine, tri-n-butylamine , Tri-n-pentylamine, tri-n-hexylamine, tri- Tri (cyclohexylamine, tri-n-octylamine, tri-n-nonylamine, tri-n-decylamine, dimethyl-n-dodecylamine, di-n-dodecylmethylamine
- Alkylamines Alkanolamines such as monoethanolamine, diethanolamine, triethanolamine; aniline, N-methylaniline, N, N-dimethylaniline, 2-methylaniline, 3-methylaniline, 4-methylaniline, 4 -Aromatic amines such as nitroaniline, diphenylamine, triphenylamine, 1-naphthylamine and the like.
- nitrogen-containing compound (II) examples include, but are not limited to, ethylenediamine, N, N, N ′, N′-tetramethylethylenediamine, N, N, N ′, N′-tetrakis (2- Hydroxypropyl) ethylenediamine, tetramethylenediamine, hexamethylenediamine, 4,4'-diaminodiphenylmethane, 4,4'-diaminodiphenyl ether, 4,4'-diaminobenzophenone, 4,4'-diaminodiphenylamine, 2,2-bis (4-aminophenyl) propane, 2- (3-aminophenyl) -2- (4-aminophenyl) propane, 2- (4-aminophenyl) -2- (3-hydroxyphenyl) propane, 2- (4 -Aminophenyl) -2- (4-hydroxyphenyl) propane, 1,4-bis [1 (4-aminophenyl)
- the nitrogen-containing compound (III) is not particularly limited, and examples thereof include polyethyleneimine, polyallylamine, N- (2-dimethylaminoethyl) acrylamide polymer, and the like.
- amide group-containing compound examples include, but are not particularly limited to, for example, formamide, N-methylformamide, N, N-dimethylformamide, acetamide, N-methylacetamide, N, N-dimethylacetamide, propionamide, benzamide Pyrrolidone, N-methylpyrrolidone and the like.
- urea compound examples include, but are not limited to, urea, methylurea, 1,1-dimethylurea, 1,3-dimethylurea, 1,1,3,3-tetramethylurea, 1,3- Examples thereof include diphenylurea and tri-n-butylthiourea.
- nitrogen-containing heterocyclic compound examples include, but are not limited to, imidazoles such as imidazole, benzimidazole, 4-methylimidazole, 4-methyl-2-phenylimidazole, 2-phenylbenzimidazole; 2-methylpyridine, 4-methylpyridine, 2-ethylpyridine, 4-ethylpyridine, 2-phenylpyridine, 4-phenylpyridine, 2-methyl-4-phenylpyridine, nicotine, nicotinic acid, nicotinamide, quinoline Pyridines such as 8-oxyquinoline and acridine; and pyrazine, pyrazole, pyridazine, quinosaline, purine, pyrrolidine, piperidine, morpholine, 4-methylmorpholine, piperazine, 1,4-dimethylpiperazine, 1,4-diazabicyclo [ 2 2.2] octane and the like can be mentioned.
- imidazoles such as imidazole,
- radiolytic basic compound is not particularly limited, but for example, the following general formula (11-1): A sulfonium compound represented by the general formula (11-2): The iodonium compound etc. which are represented by these can be mentioned.
- R 71 , R 72 , R 73 , R 74 and R 75 are each independently a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, or 1 carbon atom.
- Z ⁇ represents HO ⁇ , R—COO ⁇ (wherein R represents an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 11 carbon atoms, or an alkaryl group having 7 to 12 carbon atoms) or the following general formula (11-3):
- the radiation-decomposable basic compound include, but are not limited to, triphenylsulfonium hydroxide, triphenylsulfonium acetate, triphenylsulfonium salicylate, diphenyl-4-hydroxyphenylsulfonium hydroxide, diphenyl- 4-hydroxyphenylsulfonium acetate, diphenyl-4-hydroxyphenylsulfonium salicylate, bis (4-tert-butylphenyl) iodonium hydroxide, bis (4-tert-butylphenyl) iodonium acetate, bis (4-tert-butyl) Phenyl) iodonium hydroxide, bis (4-tert-butylphenyl) iodonium acetate, bis (4-tert-butylphenyl) iodonium salicylate, -t- butylphenyl-4-hydroxyphenyl iodonium hydroxide, 4-t
- the content of the acid diffusion control agent (E) is preferably 0.001 to 49% by weight, more preferably 0.01 to 10% by weight, based on the total weight of the solid component, and 0 0.01 to 5% by weight is more preferable, and 0.01 to 3% by weight is particularly preferable.
- the radiation-sensitive composition of the present embodiment can prevent degradation of resolution, pattern shape, dimensional fidelity, and the like. Furthermore, even if the holding time from electron beam irradiation to heating after radiation irradiation becomes longer, the shape of the pattern upper layer portion does not deteriorate.
- the radiation sensitive composition of this embodiment can prevent a fall of a sensitivity, the developability of an unexposed part, etc. as content of an acid diffusion control agent (E) is 10 weight% or less.
- an acid diffusion controller (E) by using such an acid diffusion controller (E), the storage stability of the radiation-sensitive composition is improved, the resolution is improved, the holding time before irradiation, and the pulling time after irradiation. Changes in the line width of the resist pattern due to fluctuations in placement time can be suppressed, and the process stability is extremely excellent.
- a dissolution accelerator such as organic carboxylic acids or phosphorus oxo acids or derivatives thereof can be added.
- Dissolution Accelerator when the solubility of the cyclic compound in the developer is too low, the dissolution accelerator increases its solubility and moderately increases the dissolution rate of the cyclic compound during development. It can be used in the range which does not impair the effect of this invention. Although it does not specifically limit as said dissolution promoter, For example, a low molecular weight phenolic compound can be mentioned, Specifically, bisphenols, a tris (hydroxyphenyl) methane, etc. can be mentioned, for example. These dissolution promoters can be used alone or in admixture of two or more.
- the content of the dissolution accelerator is appropriately adjusted according to the type of cyclic compound to be used, but is preferably 0 to 49% by weight based on the total weight of the solid component, and 0 to 5 % By weight is more preferred, 0 to 1% by weight is more preferred, and 0% by weight is particularly preferred.
- Solubility control agent for example, has a function of controlling the solubility to moderately reduce the dissolution rate during development when the cyclic compound described above is too soluble in the developer. It is.
- a dissolution control agent those that do not chemically change in steps such as baking of resist film, irradiation with radiation, and development are preferable.
- dissolution control agent for example, aromatic hydrocarbons, such as phenanthrene, anthracene, and acenaphthene; Ketones, such as acetophenone, benzophenone, and phenyl naphthyl ketone; Examples include sulfones.
- aromatic hydrocarbons such as phenanthrene, anthracene, and acenaphthene
- Ketones such as acetophenone, benzophenone, and phenyl naphthyl ketone
- examples include sulfones.
- the content of the dissolution control agent is appropriately adjusted according to the type of cyclic compound to be used, but is preferably 0 to 49% by weight of the total weight of the solid component, and 0 to 5 % By weight is more preferred, 0 to 1% by weight is more preferred, and 0% by weight is particularly preferred.
- Sensitizer absorbs the energy of the irradiated radiation and transmits the energy to the acid generator (C), thereby increasing the amount of acid generated. It is a component that improves the apparent sensitivity.
- a sensitizer is not particularly limited, and examples thereof include benzophenones, biacetyls, pyrenes, phenothiazines, and fluorenes. These sensitizers can be used alone or in combination of two or more.
- the content of the sensitizer is appropriately adjusted according to the type of the cyclic compound to be used, but is preferably 0 to 49% by weight based on the total weight of the solid component, and 0 to 5 % By weight is more preferred, 0 to 1% by weight is more preferred, and 0% by weight is particularly preferred.
- the surfactant is a component having an action of improving the coating property and striation of the radiation-sensitive composition of the present embodiment, the developability of the resist, and the like.
- a surfactant may be anionic, cationic, nonionic or amphoteric.
- a preferred surfactant is a nonionic surfactant.
- Nonionic surfactants have better affinity with the solvent used in the production of the radiation-sensitive composition and are more effective.
- Examples of the nonionic surfactant are not particularly limited, and examples thereof include polyoxyethylene higher alkyl ethers, polyoxyethylene higher alkyl phenyl ethers, and polyethylene glycol higher fatty acid diesters.
- Ftop made by Gemco
- Megafuck made by Dainippon Ink and Chemicals
- Florard made by Sumitomo 3M
- Asahi Guard Surflon
- Pepol manufactured by Toho Chemical Industry Co., Ltd.
- KP manufactured by Shin-Etsu Chemical Co., Ltd.
- Polyflow manufactured by Kyoeisha Yushi Chemical Co., Ltd.
- the content of the surfactant is appropriately adjusted according to the kind of the cyclic compound to be used, but is preferably 0 to 49% by weight based on the total weight of the solid component, and 0 to 5 % By weight is more preferred, 0 to 1% by weight is more preferred, and 0% by weight is particularly preferred.
- Organic carboxylic acid or phosphorous oxo acid or derivative thereof is further used as an optional component for the purpose of preventing sensitivity deterioration or improving the resist pattern shape, retention stability, etc.
- Organic carboxylic acids or phosphorus oxo acids or their derivatives can be included. In addition, it can use together with an acid diffusion control agent, and may be used independently. Although it does not specifically limit as organic carboxylic acid, For example, malonic acid, a citric acid, malic acid, a succinic acid, a benzoic acid, a salicylic acid etc. are suitable.
- Phosphorus oxoacids or derivatives thereof are not particularly limited.
- phosphoric acid, phosphoric acid di-n-butyl ester, phosphoric acid diphenyl ester and other phosphoric acid or derivatives thereof such as phosphonic acid, phosphonic acid Phosphonic acid such as dimethyl ester, phosphonic acid di-n-butyl ester, phenylphosphonic acid, phosphonic acid diphenyl ester, phosphonic acid dibenzyl ester or derivatives thereof, phosphinic acid, phosphinic acid such as phenylphosphinic acid, and the like And derivatives thereof such as phosphonic acid are particularly preferred.
- Organic carboxylic acids or phosphorus oxo acids or derivatives thereof may be used alone or in combination of two or more.
- the content of the organic carboxylic acid or phosphorus oxo acid or derivative thereof is appropriately adjusted according to the type of cyclic compound to be used, but 0 to 49 of the total weight of the solid component. % By weight is preferable, 0 to 5% by weight is more preferable, 0 to 1% by weight is further preferable, and 0% by weight is particularly preferable.
- the radiation-sensitive composition of this embodiment includes the present invention. As long as it does not impede the purpose, one or more additives other than the above-mentioned dissolution control agent, sensitizer, and surfactant can be contained. Such additives are not particularly limited, and examples thereof include dyes, pigments, and adhesion aids.
- the radiation-sensitive composition of this embodiment is preferable to contain, for example, a dye or a pigment because it can visualize the latent image in the exposed area and reduce the influence of halation during exposure.
- the radiation-sensitive composition of the present embodiment contains an adhesion assistant because adhesion with the substrate can be improved.
- examples of other additives include an antihalation agent, a storage stabilizer, an antifoaming agent, a shape improving agent, and the like, specifically 4-hydroxy-4′-methylchalcone.
- the total content of the optional component (F) is preferably 0 to 49% by weight, more preferably 0 to 5% by weight, and more preferably 0 to 1% by weight based on the total weight of the solid component. More preferred is 0% by weight.
- the content ratio of each component cyclic compound / acid generator (C) / acid crosslinking agent (G) / acid diffusion controller (E) / optional component (F)) %
- solid component preferably 50-99.4 / 0.001-49 / 0.5-49 / 0.001-49 / 0-49, more preferably 55-90 / 1-40 / 0 5 to 40 / 0.01 to 10/0 to 5, more preferably 60 to 80/3 to 30/1 to 30 / 0.01 to 5/0 to 1, particularly preferably 60 to 70/10 to 25 / 2 to 20 / 0.01 to 3/0.
- the content ratio of each component is selected from each range so that the sum is 100% by weight.
- the radiation sensitive composition of this embodiment is excellent in performance, such as sensitivity, resolution, developability, etc., when the content ratio of each of the above components is used.
- the radiation-sensitive composition of the present embodiment is usually obtained by dissolving each solid component in a solvent at the time of use to obtain a uniform solution, and then filtering with a filter having a pore size of about 0.2 ⁇ m as necessary. Prepared.
- the solvent used for the preparation of the radiation-sensitive composition of the present embodiment is not particularly limited, and examples thereof include ethylene glycol monomethyl ether acetate, ethylene glycol monoethyl ether acetate, ethylene glycol mono-n-propyl ether acetate, Ethylene glycol monoalkyl ether acetates such as ethylene glycol mono-n-butyl ether acetate; ethylene glycol monoalkyl ethers such as ethylene glycol monomethyl ether and ethylene glycol monoethyl ether; propylene glycol monomethyl ether acetate, propylene glycol monoethyl ether acetate, Propylene glycol mono-n-propyl ether acetate, propylene glycol mono-n-butyl Propylene glycol monoalkyl ether acetates such as ether acetate; Propylene glycol monoalkyl ethers such as propylene glycol monomethyl ether and propylene glycol monoethyl
- the radiation-sensitive composition of the present embodiment can contain a resin as long as the object of the present invention is not impaired.
- the resin is not particularly limited, and includes, for example, novolac resin, polyvinylphenols, polyacrylic acid, polyvinyl alcohol, styrene-maleic anhydride resin, and acrylic acid, vinyl alcohol, or vinylphenol as monomer units. Examples thereof include polymers and derivatives thereof.
- the content of the resin is appropriately adjusted according to the type of the above-described cyclic compound to be used, but is preferably 30 parts by weight or less per 100 parts by weight of the cyclic compound.
- the amount is preferably 10 parts by weight or less, more preferably 5 parts by weight or less, and particularly preferably 0 part by weight.
- the resist pattern forming method of the present embodiment includes a step of coating the radiation sensitive composition on a substrate to form a resist film, a step of exposing the resist film, and developing the exposed resist film. Forming a resist pattern.
- the resist pattern of this embodiment can also be formed as an upper layer resist in a multilayer process.
- a resist film is formed by applying the radiation-sensitive composition on a conventionally known substrate by a coating means such as spin coating, cast coating, roll coating or the like.
- the conventionally known substrate is not particularly limited, and examples thereof include a substrate for electronic parts and a substrate on which a predetermined wiring pattern is formed. More specifically, although not particularly limited, for example, a silicon substrate, a metal substrate such as copper, chromium, iron, and aluminum, a glass substrate, and the like can be given.
- the material for the wiring pattern is not particularly limited, and examples thereof include copper, aluminum, nickel, and gold. If necessary, an inorganic and / or organic film may be provided on the substrate.
- an inorganic antireflection film inorganic BARC
- organic film is not particularly limited, and examples thereof include an organic antireflection film (organic BARC). Surface treatment with hexamethylene disilazane or the like may be performed.
- the coated substrate is heated as necessary.
- the heating temperature varies depending on the composition of the radiation sensitive composition, but is preferably 20 to 250 ° C, more preferably 20 to 150 ° C.
- the adhesion of the radiation-sensitive composition (resist) to the substrate may be improved, which is preferable.
- the resist film is exposed to a desired pattern with any radiation selected from the group consisting of visible light, ultraviolet light, excimer laser, electron beam, extreme ultraviolet light (EUV), X-ray, and ion beam.
- the exposure conditions and the like are appropriately selected according to the composition of the radiation sensitive composition.
- the heating temperature varies depending on the composition of the radiation sensitive composition, but is preferably 20 to 250 ° C, more preferably 20 to 150 ° C.
- a predetermined resist pattern is formed by developing the exposed resist film with a developer (development process).
- a developer it is preferable to select a solvent having a solubility parameter (SP value) close to that of the above-mentioned cyclic compound to be used.
- SP value solubility parameter
- ketone solvents, ester solvents, alcohol solvents, amide solvents, ethers A polar solvent such as a solvent, a hydrocarbon solvent, or an alkaline aqueous solution can be used.
- the ketone solvent is not particularly limited.
- the ester solvent is not particularly limited.
- the alcohol solvent is not particularly limited.
- the ether solvent is not particularly limited, and examples thereof include dioxane, tetrahydrofuran and the like in addition to the glycol ether solvent.
- the amide solvent is not particularly limited.
- N-methyl-2-pyrrolidone, N, N-dimethylacetamide, N, N-dimethylformamide, hexamethylphosphoric triamide, 1,3-dimethyl-2- Imidazolidinone can be used.
- the hydrocarbon solvent is not particularly limited, and examples thereof include aromatic hydrocarbon solvents such as toluene and xylene, and aliphatic hydrocarbon solvents such as pentane, hexane, octane and decane.
- the water content of the developer as a whole is less than 70% by mass, preferably less than 50% by mass, and more preferably less than 30% by mass.
- it is more preferably less than 10% by mass, and it is particularly preferable that it contains substantially no water.
- the content of the organic solvent with respect to the developer is 30% by mass to 100% by mass, preferably 50% by mass to 100% by mass, and preferably 70% by mass to 100% by mass with respect to the total amount of the developer. More preferably, it is 90 mass% or less, More preferably, it is 90 mass% or more and 100 mass% or less, It is especially preferable that it is 95 mass% or more and 100 mass% or less.
- the alkaline aqueous solution is not particularly limited, and examples thereof include mono-, di- or trialkylamines, mono-, di- or trialkanolamines, heterocyclic amines, tetramethylammonium hydroxide (TMAH), choline. And alkaline compounds such as
- a developing solution containing at least one solvent selected from a ketone solvent, an ester solvent, an alcohol solvent, an amide solvent, and an ether solvent is used. It is preferable for improving the resist performance.
- the vapor pressure of the developer is preferably 5 kPa or less, more preferably 3 kPa or less, and particularly preferably 2 kPa or less at 20 ° C.
- Specific examples of the developer having a vapor pressure of 5 kPa or less at 20 ° C. are not particularly limited.
- 1-octanone, 2-octanone, 1-nonanone, 2-nonanone, 4-heptanone, 2- Ketone solvents such as hexanone, diisobutyl ketone, cyclohexanone, methylcyclohexanone, phenylacetone, methylisobutylketone, butyl acetate, amyl acetate, propylene glycol monomethyl ether acetate, ethylene glycol monoethyl ether acetate, diethylene glycol monobutyl ether acetate, diethylene glycol monoethyl ether Acetate, ethyl-3-ethoxypropionate, 3-methoxybutyl acetate, 3-methyl-3-methoxybutyl acetate, butyl formate, propionic acid Ester solvents such as ethyl lactate, but
- the developer having a vapor pressure of 2 kPa or less which is a particularly preferable range, are not particularly limited.
- 1-octanone, 2-octanone, 1-nonanone, 2-nonanone, 4-heptanone, 2 -Ketone solvents such as hexanone, diisobutyl ketone, cyclohexanone, methylcyclohexanone, phenylacetone, butyl acetate, amyl acetate, propylene glycol monomethyl ether acetate, ethylene glycol monoethyl ether acetate, diethylene glycol monobutyl ether acetate, diethylene glycol monoethyl ether acetate, ethyl -3-Ethoxypropionate, 3-methoxybutyl acetate, 3-methyl-3-methoxybutyl acetate, ethyl lactate, butyl lactate, propyl lactate, etc.
- Steal solvent n-butyl alcohol, sec-butyl alcohol, tert-butyl alcohol, isobutyl alcohol, n-hexyl alcohol, 4-methyl-2-pentanol, n-heptyl alcohol, n-octyl alcohol, n-decanol, etc.
- Alcohol solvents such as ethylene glycol, diethylene glycol, triethylene glycol, etc., ethylene glycol monomethyl ether, propylene glycol monomethyl ether, ethylene glycol monoethyl ether, propylene glycol monoethyl ether, diethylene glycol monomethyl ether, triethylene glycol mono Glycol ether solvents such as ethyl ether and methoxymethylbutanol, N-methyl-2-pyrrolidone, N, N Dimethylacetamide, N, N-dimethylformamide amide solvents, aromatic hydrocarbon solvents such as xylene, octane, aliphatic hydrocarbon solvents decane.
- An appropriate amount of a surfactant can be added to the developer as necessary.
- the surfactant is not particularly limited.
- ionic or nonionic fluorine-based and / or silicon-based surfactants can be used.
- fluorine and / or silicon surfactants include, for example, JP-A No. 62-36663, JP-A No. 61-226746, JP-A No. 61-226745, JP-A No. 62-170950, JP-A-63-34540, JP-A-7-230165, JP-A-8-62834, JP-A-9-54432, JP-A-9-5988, US Pat. No. 5,405,720, The surfactants described in US Pat. Nos.
- Nonionic surfactant it is a nonionic surfactant.
- a nonionic surfactant it does not specifically limit as a nonionic surfactant,
- the amount of the surfactant used is usually 0.001 to 5% by mass, preferably 0.005 to 2% by mass, and more preferably 0.01 to 0.5% by mass with respect to the total amount of the developer.
- the development method is not particularly limited.
- the substrate is immersed in a tank filled with a developer for a certain period of time (dip method), and the developer is raised on the surface of the substrate by surface tension and left stationary for a certain period of time.
- Development method (paddle method), spraying developer solution onto the substrate surface (spray method), developing solution is continuously applied to the substrate rotating at a constant speed while scanning the developer application nozzle at a constant speed.
- a method (dynamic dispensing method) or the like can be applied.
- the time for developing the pattern is not particularly limited, but is preferably 10 seconds to 90 seconds.
- a step of stopping the development may be performed while substituting with another solvent.
- a cleaning step (rinsing step) using a rinsing solution containing an organic solvent is included after the development step.
- the rinsing solution used in the rinsing step after the development step is not particularly limited as long as the resist pattern cured by crosslinking is not dissolved, and a solution or water containing a general organic solvent can be used.
- the rinsing liquid is not particularly limited, and includes, for example, at least one organic solvent selected from hydrocarbon solvents, ketone solvents, ester solvents, alcohol solvents, amide solvents, and ether solvents. It is preferable to use a rinse solution. More preferably, after the development step, a washing step is performed using a rinse solution containing at least one organic solvent selected from the group consisting of ketone solvents, ester solvents, alcohol solvents, and amide solvents. .
- a washing step using a rinse solution containing an alcohol solvent or an ester solvent is performed. Even more preferably, after the development step, a step of washing with a rinsing liquid containing a monohydric alcohol is performed. Particularly preferably, after the development step, a washing step is performed using a rinse solution containing a monohydric alcohol having 5 or more carbon atoms.
- the time for rinsing the pattern is not particularly limited, but is preferably 10 seconds to 90 seconds.
- the monohydric alcohol used in the rinsing step after the development step is not particularly limited, and examples thereof include linear, branched, and cyclic monohydric alcohols. Specific examples include 1-butanol, 2-butanol, 3-methyl-1-butanol, tert-butyl alcohol, 1-pentanol, 2-pentanol, 1-hexanol, 4-methyl-2-pentanol, 1-heptanol, 1-octanol, 2- Hexanol, cyclopentanol, 2-heptanol, 2-octanol, 3-hexanol, 3-heptanol, 3-octanol, 4-octanol and the like can be used, and particularly preferable monohydric alcohols having 5 or more carbon atoms include Without limitation, for example, 1-hexanol, 2-hexanol, 4-methyl-2-pen Nord, 1-pentanol, 3-methyl-1-butanol and the like can be
- a plurality of the above components may be mixed, or may be used by mixing with an organic solvent other than the above.
- the water content in the rinse liquid is preferably 10% by mass or less, more preferably 5% by mass or less, and particularly preferably 3% by mass or less. By setting the water content in the rinse liquid to 10% by mass or less, better development characteristics can be obtained.
- the vapor pressure of the rinsing liquid used after the development step is preferably 0.05 kPa or more and 5 kPa or less, more preferably 0.1 kPa or more and 5 kPa or less, and further preferably 0.12 kPa or more and 3 kPa or less at 20 ° C.
- An appropriate amount of a surfactant can be added to the rinse solution.
- the wafer that has been subjected to the developing step is cleaned using a rinsing solution containing the organic solvent.
- the method of the cleaning treatment is not particularly limited. For example, a method of continuously applying a rinse liquid onto a substrate rotating at a constant speed (rotary coating method), or immersing the substrate in a tank filled with the rinse liquid for a certain period of time. A method (dip method), a method of spraying a rinsing liquid onto the substrate surface (spray method), etc. can be applied.
- a cleaning process is performed by a spin coating method, and after cleaning, the substrate is rotated at a speed of 2000 rpm to 4000 rpm. It is preferable to rotate and remove the rinse liquid from the substrate.
- the pattern wiring board is obtained by etching.
- the etching method include known methods such as dry etching using plasma gas and wet etching using an alkali solution, a cupric chloride solution, a ferric chloride solution, or the like.
- plating after forming the resist pattern.
- plating method for example, there exist copper plating, solder plating, nickel plating, gold plating, etc.
- the residual resist pattern after etching can be stripped with an organic solvent.
- organic solvent For example, PGMEA (propylene glycol monomethyl ether acetate), PGME (propylene glycol monomethyl ether), EL (ethyl lactate) etc. are mentioned.
- peeling method For example, the immersion method, a spray system, etc. are mentioned.
- the wiring board on which the resist pattern is formed may be a multilayer wiring board or may have a small diameter through hole.
- the wiring board can also be formed by a method of depositing a metal in a vacuum after forming a resist pattern and then dissolving the resist pattern with a solution, that is, a lift-off method.
- this compound showed a molecular weight 960 of the target compound represented by the following formula (CR-1A).
- the chemical shift value ( ⁇ ppm, TMS standard) of 1 H-NMR in the heavy dimethyl sulfoxide solvent of the obtained compound is 1.1 to 1.2 (m, 24H), 2.6 to 2.7 ( m, 4H), 5.5 (s, 4H), 6.0 to 6.8 (m, 24H), and 8.4 to 8.5 (d, 8H).
- the compound obtained from these results was identified as the target compound (hereinafter also referred to as “CR-1A”) represented by the following formula (CR-1A) (yield 95%).
- Synthesis Example 2 Synthesis of CR-2A (Cyclic Compound (A)) Synthesis was performed in the same manner as in Synthesis Example 1 except that 4-isopropylbenzaldehyde was replaced with 4-cyclohexylbenzaldehyde (46.0 g, 0.2 mol), and 50 g To give a compound. As a result of analysis by LC-MS, this compound showed a molecular weight 1121 of the target compound represented by the following formula (CR-2A).
- the chemical shift value ( ⁇ ppm, TMS standard) of 1 H-NMR in the heavy dimethyl sulfoxide solvent of the obtained compound is 0.8 to 1.9 (m, 44H), 5.5 to 5.6 ( d, 4H), 6.0 to 6.8 (m, 24H), and 8.4 to 8.5 (m, 8H).
- the compound obtained from these results was identified as the target compound represented by the following formula (CR-2A) (hereinafter also referred to as “CR-2A”) (yield 91%).
- the chemical shift value ( ⁇ ppm, TMS standard) of 1 H-NMR in the heavy dimethyl sulfoxide solvent of the obtained compound is 1.1 to 1.2 (m, 24H), 2.6 to 2.7 (m, 4H), 5.5 (m, 4H), 5.7 (m, 4H), 6.0 to 6.8 (m, 32H), and 8.4 to 8.5 (m, 4H).
- the compound obtained from these results was converted to a compound represented by the following formula (CR-1) in which 50 mol% of the hydrogen atoms of the phenolic hydroxyl group of CR-1A were substituted with an acryloyl group (hereinafter referred to as “CR-1”). Also identified).
- Synthesis Example 2 Synthesis of CR-2 (Cyclic Compound) Synthesis was performed in the same manner as in Synthesis Example 1 except that CR-1A was replaced with CR-2A (11.2 g, 10 mmol). Obtained. Further, the chemical shift value ( ⁇ ppm, TMS standard) of 1 H-NMR in the heavy dimethyl sulfoxide solvent of the obtained compound is 0.8 to 1.9 (m, 44H), 5.5 to 5.6 ( d, 4H), 5.7 (s, 4H), 6.0 to 6.8 (m, 32H), and 8.4 to 8.5 (m, 4H).
- CR-2 a compound represented by the following formula (CR-2) in which 50 mol% of the hydrogen atoms of the phenolic hydroxyl group of CR-2A were substituted with an acryloyl group (hereinafter referred to as “CR-2”). Also identified).
- Synthesis Example 3 Synthesis of CR-3 (Cyclic Compound) Synthesis was performed in the same manner as in Synthesis Example 1 except that the amount of acryloyl chloride was changed from 3.6 g (40 mmol) to 10.8 g (120 mmol). 0 g of compound was obtained. As a result of analysis by LC-MS, this compound showed a molecular weight of 1393 of the target compound represented by the following formula (CR-3).
- the chemical shift value ( ⁇ ppm, TMS standard) of 1 H-NMR in the heavy dimethyl sulfoxide solvent of the obtained compound is 1.1 to 1.2 (m, 24H), 2.6 to 2.7 ( m, 4H), 5.5 (s, 4H), 5.7 (s, 8H), and 6.0 to 6.8 (m, 40H).
- the compound obtained from these results was converted to a compound represented by the following formula (CR-3) (hereinafter referred to as “CR-3”) in which 100 mol% of the hydrogen atom of the phenolic hydroxyl group of CR-1A was substituted with an acryloyl group. Also identified).
- Synthesis Example 4 Synthesis of CR-4 (Cyclic Compound) Synthesis was performed in the same manner as in Synthesis Example 2 except that the amount of acryloyl chloride was changed from 3.6 g (40 mmol) to 10.8 g (120 mmol). 4 g of compound was obtained. As a result of analysis by LC-MS, this compound showed a molecular weight 1553 of the target compound represented by the following formula (CR-4).
- the chemical shift value ( ⁇ ppm, TMS standard) of 1 H-NMR in the heavy dimethyl sulfoxide solvent of the obtained compound is 0.8 to 1.9 (m, 44H), 5.5 to 5.6 ( d, 4H), 5.7 (s, 8H), and 6.0 to 6.8 (m, 40H).
- the compound obtained from these results was converted to a compound represented by the following formula (CR-4) (hereinafter referred to as “CR-4”) in which 100 mol% of the hydrogen atom of the phenolic hydroxyl group of CR-2A was substituted with an acryloyl group. Also identified).
- Synthesis Example 5 Synthesis of CR-5 (Cyclic Compound) Synthesis was performed in the same manner as in Synthesis Example 3 except that 10.8 g (120 mmol) of acryloyl chloride was replaced with 12.48 g (120 mmol) of methacryloyl chloride. To give a compound. As a result of analysis by LC-MS, this compound showed a molecular weight 1521 of the target compound represented by the following formula (CR-5). Further, the chemical shift value ( ⁇ ppm, TMS standard) of 1 H-NMR in the heavy dimethyl sulfoxide solvent of the obtained compound is 1.1 to 1.2 (m, 24H), 1.9 (s, 12H).
- CR-5 a compound represented by the following formula (hereinafter referred to as “CR-5”) in which 100 mol% of the hydrogen atom of the phenolic hydroxyl group of CR-1A was substituted with a methacryloyl group. Also identified).
- Acid generator (C) P-1 Triphenylbenzenesulfonium trifluoromethanesulfonate (Midori Chemical Co., Ltd.) Acid crosslinking agent (G) C-1: Nikarac MW-100LM (Sanwa Chemical Co., Ltd.) Acid diffusion controller (E) Q-1: Trioctylamine (Tokyo Chemical Industry Co., Ltd.) Solvent S-1: Propylene glycol monomethyl ether (Tokyo Chemical Industry Co., Ltd.)
- the patterning test was done as follows using the prepared radiation sensitive composition.
- the prepared radiation sensitive composition was spin-coated on a clean silicon wafer and then pre-exposure baked (PB) in an oven at 110 ° C. to form a resist film having a thickness of 60 nm.
- the resist film was irradiated with an electron beam with a 1: 1 line and space setting at intervals of 50 nm, 40 nm, and 30 nm using an electron beam drawing apparatus (ELS-7500, manufactured by Elionix Co., Ltd.). After irradiation, the resist film formed from the radiation-sensitive compositions prepared in Examples 7 and 8 was heated at 110 ° C.
- the obtained resist pattern was observed with a scanning electron microscope (S-4800, manufactured by Hitachi High-Technology Corporation).
- the resolution of the pattern was the minimum line width of the formed pattern.
- the pattern shape was good if it was rectangular.
- the line edge roughness was good when the pattern irregularities were less than 5 nm. Further to the dose at that time a ( ⁇ C / cm 2) and sensitivity, was considered good than 150 ⁇ C / cm 2.
- the radiation sensitive compositions of Comparative Examples 3 and 4 were able to obtain a good resist pattern with a resolution of 40 nm, but could not obtain a good resist pattern with a resolution of 30 nm.
- the composition containing the compound of the present embodiment (for example, CR-3 and CR-4) is more effective than the composition containing the corresponding comparative compound (for example, CR-1A and CR-2A). It was found that a resist pattern with high sensitivity, low roughness, and good shape can be formed. As long as the requirements of the present embodiment described above are satisfied, compounds other than those described in the examples also show the same effect.
- the present invention relates to a cyclic compound represented by a specific chemical structural formula useful as an acid amplification type non-polymer resist material, a radiation sensitive composition containing the same, and a resist pattern forming method using the radiation sensitive composition. Preferably used.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- General Physics & Mathematics (AREA)
- Materials For Photolithography (AREA)
- Exposure And Positioning Against Photoresist Photosensitive Materials (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
1. 分子量500~5000の、式(1)で示される環状化合物。
前記R0の少なくともひとつが(メタ)アクリロイル基を含む一価の基である。)
2. 前記式(1)で示される環状化合物が、式(2)で示される前項1に記載の環状化合物。
R2は、それぞれ独立して、水素原子、シアノ基、ニトロ基、ハロゲン原子、置換若しくは無置換の炭素数1~20の直鎖状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の分岐状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の環状脂肪族炭化水素基、置換若しくは無置換の炭素数6~20のアリール基、置換若しくは無置換の炭素数7~30のアラルキル基、置換若しくは無置換の炭素数1~20のアルコキシ基、置換若しくは無置換の炭素数2~20のアルケニル基、置換若しくは無置換の炭素数1~20のアシル基、置換若しくは無置換の炭素数2~20のアルコキシカルボニル基、置換若しくは無置換の炭素数1~20のアルキロイルオキシ基、置換若しくは無置換の炭素数7~30のアリーロイルオキシ基又は置換若しくは無置換の炭素数1~20のアルキルシリル基であり、
R’は、それぞれ独立して、水素原子、置換若しくは無置換の炭素数1~20の直鎖状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の分岐状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の環状脂肪族炭化水素基又は下記式(3)
前記R1の少なくともひとつが、(メタ)アクリロイル基である。)
3. 前記式(2)で示される環状化合物が、式(4)で示される前項2に記載の環状化合物。
R4は、それぞれ独立して、シアノ基、ニトロ基、置換若しくは無置換の複素環基、ハロゲン原子、置換若しくは無置換の炭素数1~20の直鎖状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の分岐状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の環状脂肪族炭化水素基、置換若しくは無置換の炭素数6~20のアリール基、置換若しくは無置換の炭素数1~20のアルコキシ基又は置換若しくは無置換の炭素数1~20のアルキルシリル基であり、
pは0~5の整数であり、
前記R1の少なくともひとつが、(メタ)アクリロイル基である。)
4. 前記式(4)で示される環状化合物が、式(5)で示される前項3に記載の環状化合物。
pは0~5の整数であり、
R5は(メタ)アクリロイル基である。)
5. 前項1~4のいずれか1項に記載の環状化合物を製造する方法であって、
1種以上のアルデヒド性化合物(A1)と1種以上のフェノール性化合物(A2)とを縮合反応させて環状化合物(A)を得る工程と、
該環状化合物(A)と1種以上の(メタ)アクリロイルハライドとを脱ハロゲン化水素反応させる工程とを備える、環状化合物の製造方法。
6. 前記アルデヒド性化合物(A1)が、1~4個のホルミル基を有し、かつ炭素数1~59であり、
前記フェノール性化合物(A2)が、1~3個のフェノール性水酸基を有し、かつ炭素数6~15である、前項5に記載の環状化合物の製造方法。
7. 前項1~4のいずれか1項に記載の環状化合物を含む固形成分及び溶媒を含有する、感放射線性組成物。
8. 前記固形成分の含有量が1~80重量%である、前項7に記載の感放射線性組成物。
9. 前記固形成分の全重量に占める前記環状化合物の割合が50~99.999重量%である、前項7又は8に記載の感放射線性組成物。
10. 可視光線、紫外線、エキシマレーザー、電子線、極端紫外線(EUV)、X線及びイオンビームからなる群から選ばれるいずれかの放射線の照射により直接的又は間接的に酸を発生する酸発生剤(C)をさらに含む、前項7~9のいずれか1項に記載の感放射線性組成物。
11. 酸架橋剤(G)をさらに含む、前項7~10のいずれか1項に記載の感放射線性組成物。
12. 酸拡散制御剤(E)をさらに含む、前項7~11のいずれか1項に記載の感放射線性組成物。
13. 前記固形成分が、環状化合物50~99.4重量%、酸発生剤(C)0.001~49重量%、酸架橋剤(G)0.5~49重量%、及び酸拡散制御剤(E)0.001~49重量%を含有する、前項7~12のいずれか1項に記載の感放射線性組成物。
14. スピンコートによりアモルファス膜を形成する、前項7~13のいずれか1項に記載の感放射線性組成物。
15. 前記アモルファス膜の23℃における現像液に対する溶解速度が10Å/sec以上である、前項14に記載の感放射線性組成物。
16. KrFエキシマレーザー、極端紫外線、電子線若しくはX線を照射した後の前記アモルファス膜、又は20~250℃で加熱した後の前記アモルファス膜の、現像液に対する溶解速度が5Å/sec以下である、前項14又は15に記載の感放射線性組成物。
17. 前項7~16のいずれか1項に記載の感放射線性組成物を、基板上に塗布してレジスト膜を形成する工程と、
該レジスト膜を露光する工程と、
該露光したレジスト膜を現像する工程とを備える、レジストパターン形成方法。
本実施形態の環状化合物は、分子量500~5000の、下記式(1)で示される環状化合物である。本実施形態の環状化合物は、レジスト材料として有用である。
本実施形態による感放射線性組成物は、前記した環状化合物を含む固形成分と、溶媒とを含有する。また、本実施形態の感放射線性組成物は、その固形成分が1~80重量%であることが好ましく、さらに、該環状化合物の固形成分全重量に占める割合が50~99.999重量%であることがより好ましい。
上記酸発生剤(C)は、単独で又は2種以上を使用することができる。
溶解促進剤は、例えば、上述の環状化合物の現像液に対する溶解性が低すぎる場合に、その溶解性を高めて、現像時の環状化合物の溶解速度を適度に増大させる作用を有する成分であり、本発明の効果を損なわない範囲で使用することができる。前記溶解促進剤としては、特に限定されないが、例えば、低分子量のフェノール性化合物を挙げることができ、具体的には、例えば、ビスフェノール類、トリス(ヒドロキシフェニル)メタン等を挙げることができる。これらの溶解促進剤は、単独で又は2種以上を混合して使用することができる。本実施形態の感放射線性組成物において、溶解促進剤の含有量は、使用する環状化合物の種類に応じて適宜調節されるが、固形成分全重量の0~49重量%が好ましく、0~5重量%がより好ましく、0~1重量%がさらに好ましく、0重量%が特に好ましい。
溶解制御剤は、例えば、上述の環状化合物が現像液に対する溶解性が高すぎる場合に、その溶解性を制御して現像時の溶解速度を適度に減少させる作用を有する成分である。このような溶解制御剤としては、レジスト被膜の焼成、放射線照射、現像等の工程において化学変化しないものが好ましい。
増感剤は、照射された放射線のエネルギーを吸収して、そのエネルギーを酸発生剤(C)に伝達し、それにより酸の生成量を増加する作用を有し、レジストの見掛けの感度を向上させる成分である。このような増感剤としては、特に限定されないが、例えば、ベンゾフェノン類、ビアセチル類、ピレン類、フェノチアジン類、フルオレン類等を挙げることができる。これらの増感剤は、単独で又は2種以上を使用することができる。本実施形態の感放射線性組成物において、増感剤の含有量は、使用する環状化合物の種類に応じて適宜調節されるが、固形成分全重量の0~49重量%が好ましく、0~5重量%がより好ましく、0~1重量%がさらに好ましく、0重量%が特に好ましい。
界面活性剤は、本実施形態の感放射線性組成物の塗布性やストリエーション、レジストの現像性等を改良する作用を有する成分である。このような界面活性剤は、アニオン系、カチオン系、ノニオン系あるいは両性のいずれでもよい。好ましい界面活性剤はノニオン系界面活性剤である。ノニオン系界面活性剤は、感放射線性組成物の製造に用いる溶媒との親和性がよく、より効果がある。ノニオン系界面活性剤の例としては、特に限定されないが、例えば、ポリオキシエチレン高級アルキルエーテル類、ポリオキシエチレン高級アルキルフェニルエーテル類、ポリエチレングリコールの高級脂肪酸ジエステル類等が挙げられる。市販品としては、特に限定されないが、例えば、以下商品名で、エフトップ(ジェムコ社製)、メガファック(大日本インキ化学工業社製)、フロラード(住友スリーエム社製)、アサヒガード、サーフロン(以上、旭硝子社製)、ペポール(東邦化学工業社製)、KP(信越化学工業社製)、ポリフロー(共栄社油脂化学工業社製)等を挙げることができる。本実施形態の感放射線性組成物において、界面活性剤の含有量は、使用する環状化合物の種類に応じて適宜調節されるが、固形成分全重量の0~49重量%が好ましく、0~5重量%がより好ましく、0~1重量%がさらに好ましく、0重量%が特に好ましい。
本実施形態の感放射線性組成物は、感度劣化防止又はレジストパターン形状、引き置き安定性等の向上の目的で、さらに任意の成分として、有機カルボン酸又はリンのオキソ酸若しくはその誘導体を含有させることができる。なお、酸拡散制御剤と併用することもできるし、単独で用いてもよい。有機カルボン酸としては、特に限定されないが、例えば、マロン酸、クエン酸、リンゴ酸、コハク酸、安息香酸、サリチル酸などが好適である。リンのオキソ酸若しくはその誘導体としては、特に限定されないが、例えば、リン酸、リン酸ジ-n-ブチルエステル、リン酸ジフェニルエステルなどのリン酸又はそれらのエステルなどの誘導体、ホスホン酸、ホスホン酸ジメチルエステル、ホスホン酸ジ-n-ブチルエステル、フェニルホスホン酸、ホスホン酸ジフェニルエステル、ホスホン酸ジベンジルエステルなどのホスホン酸又はそれらのエステルなどの誘導体、ホスフィン酸、フェニルホスフィン酸などのホスフィン酸及びそれらのエステルなどの誘導体が挙げられ、これらの中で特にホスホン酸が好ましい。
更に、本実施形態の感放射線性組成物には、本発明の目的を阻害しない範囲で、必要に応じて、上記溶解制御剤、増感剤、及び界面活性剤以外の添加剤を1種又は2種以上含有することができる。そのような添加剤としては、特に限定されないが、例えば、染料、顔料、及び接着助剤等が挙げられる。本実施形態の感放射線性組成物は、例えば、染料又は顔料を含有させると、露光部の潜像を可視化させて、露光時のハレーションの影響を緩和できるので好ましい。また、本実施形態の感放射線性組成物は、接着助剤を含有させると、基板との接着性を改善することができるので好ましい。更に、他の添加剤としては、ハレーション防止剤、保存安定剤、消泡剤、形状改良剤等、具体的には4-ヒドロキシ-4’-メチルカルコン等を挙げることができる。
本実施形態のレジストパターンの形成方法は、上記の感放射線性組成物を、基板上に塗布してレジスト膜を形成する工程、前記レジスト膜を露光する工程及び前記露光したレジスト膜を現像してレジストパターンを形成する工程を含む。本実施形態のレジストパターンは多層プロセスにおける上層レジストとして形成することもできる。
十分乾燥し、窒素置換した、滴下漏斗、ジム・ロート氏冷却管、温度計、攪拌翼を設置した四つ口フラスコ(1000mL)において、窒素気流下で、関東化学社製レゾルシノール(22g、0.2mol)と、4-イソプロピルベンズアルデヒド(29.6g,0.2mol)と、脱水エタノール(200mL)とを投入し、エタノール溶液を調製した。このエタノール溶液を攪拌しながらマントルヒーターで85℃まで加熱した。次いでこのエタノール溶液中に、濃塩酸(35%)75mLを、滴下漏斗により30分かけて滴下した後、引き続き85℃で3時間攪拌して反応を行った。反応終了後、得られた反応液を放冷し、室温に到達させた後、氷浴で冷却した。この反応液を1時間静置後、淡黄色粗結晶が生成した。生成した淡黄色粗結晶を濾別して粗結晶を得た。得られた粗結晶をメタノール500mLで2回洗浄し、濾別、真空乾燥させることにより45.6gの化合物を得た。この化合物は、LC-MS分析した結果、下記式(CR-1A)で表される目的化合物の分子量960を示した。また、得られた化合物の重ジメチルスルホキシド溶媒中での1H-NMRのケミカルシフト値(δppm,TMS基準)は1.1~1.2(m,24H)、2.6~2.7(m,4H)、5.5(s,4H)、6.0~6.8(m,24H)、8.4~8.5(d,8H)であった。これらの結果から得られた化合物を下記式(CR-1A)で表される目的化合物(以下「CR-1A」とも記す。)と同定した(収率95%)。
4-イソプロピルベンズアルデヒドを4-シクロヘキシルベンズアルデヒド(46.0g,0.2mol)に代えた以外は合成例1と同様に合成し、50gの化合物を得た。この化合物は、LC-MSで分析した結果、下記式(CR-2A)で表される目的化合物の分子量1121を示した。また、得られた化合物の重ジメチルスルホキシド溶媒中での1H-NMRのケミカルシフト値(δppm,TMS基準)は0.8~1.9(m,44H)、5.5~5.6(d,4H)、6.0~6.8(m,24H)、8.4~8.5(m,8H)であった。これらの結果から得られた化合物を下記式(CR-2A)で表される目的化合物(以下「CR-2A」とも記す。)と同定した(収率91%)。
十分乾燥し、窒素置換した、滴下漏斗、ジム・ロート氏冷却管、温度計、攪拌翼を設置した四つ口フラスコ(1000mL)において、窒素気流下、氷浴で冷却した、合成例1で合成したCR-1A9.6g(10mmol)、トリエチルアミン4g(40mmol)及び300mLのTHFからなる溶液に、アクリロイルクロライド3.6g(40mmol)を滴下し、1時間室温で撹拌して反応を行った。反応終了後、得られた反応液を冷水1000mlに加え、析出した固体を濾別した。得られた固体を、カラムクロマトで精製し、10.6gの化合物を得た。
CR-1AをCR-2A(11.2g,10mmol)に代えた以外は合成実施例1と同様に合成し、11.2gの化合物を得た。また、得られた化合物の重ジメチルスルホキシド溶媒中での1H-NMRのケミカルシフト値(δppm,TMS基準)は0.8~1.9(m,44H)、5.5~5.6(d,4H)、5.7(s,4H)、6.0~6.8(m,32H)、8.4~8.5(m,4H)であった。これらの結果から得られた化合物を、CR-2Aのフェノール性水酸基の水素原子の50mol%がアクリロイル基で置換された、下記式(CR-2)で表される化合物(以下「CR-2」とも記す。)と同定した。
アクリロイルクロライドの量を3.6g(40mmol)から10.8g(120mmol)に代えた以外は合成実施例1と同様に合成し、11.0gの化合物を得た。この化合物は、LC-MSで分析した結果、下記式(CR-3)で表される目的化合物の分子量1393を示した。また、得られた化合物の重ジメチルスルホキシド溶媒中での1H-NMRのケミカルシフト値(δppm,TMS基準)は1.1~1.2(m,24H)、2.6~2.7(m,4H)、5.5(s,4H)、5.7(s,8H)、6.0~6.8(m,40H)であった。これらの結果から得られた化合物を、CR-1Aのフェノール性水酸基の水素原子の100mol%がアクリロイル基で置換された、下記式(CR-3)で表される化合物(以下「CR-3」とも記す。)と同定した。
アクリロイルクロライドの量を3.6g(40mmol)から10.8g(120mmol)に代えた以外は合成実施例2と同様に合成し、11.4gの化合物を得た。この化合物は、LC-MSで分析した結果、下記式(CR-4)で表される目的化合物の分子量1553を示した。また、得られた化合物の重ジメチルスルホキシド溶媒中での1H-NMRのケミカルシフト値(δppm,TMS基準)は0.8~1.9(m,44H)、5.5~5.6(d,4H)、5.7(s,8H)、6.0~6.8(m,40H)であった。これらの結果から得られた化合物を、CR-2Aのフェノール性水酸基の水素原子の100mol%がアクリロイル基で置換された、下記式(CR-4)で表される化合物(以下「CR-4」とも記す。)と同定した。
アクリロイルクロライド10.8g(120mmol)をメタクリロイルクロライド12.48g(120mmol)に代えた以外は合成実施例3と同様に合成し、11.0gの化合物を得た。この化合物は、LC-MSで分析した結果、下記式(CR-5)で表される目的化合物の分子量1521を示した。また、得られた化合物の重ジメチルスルホキシド溶媒中での1H-NMRのケミカルシフト値(δppm,TMS基準)は1.1~1.2(m,24H)、1.9(s,12H)、2.6~2.7(m,4H)、5.5(s,4H)、5.7(s,8H)、6.0~6.8(m,32H)であった。これらの結果から得られた化合物を、CR-1Aのフェノール性水酸基の水素原子の100mol%がメタクリロイル基で置換された、下記式(CR-5)で表される化合物(以下「CR-5」とも記す。)と同定した。
アクリロイルクロライド10.8g(120mmol)をメタクリロイルクロライド12.48g(120mmol)に代えた以外は合成実施例4と同様に合成し、11.0gの化合物を得た。この化合物は、LC-MSで分析した結果、下記式(CR-6)で表される目的化合物の分子量1681を示した。また、得られた化合物の重ジメチルスルホキシド溶媒中での1H-NMRのケミカルシフト値(δppm,TMS基準)は0.8~1.9(m,44H)、2.0(s,24H)、5.5~5.6(d,4H)、5.7(s,8H)、6.0~6.8(m,32H)であった。これらの結果から得られた化合物を、CR-2Aのフェノール性水酸基の水素原子の100mol%がメタクリロイル基で置換された、下記式(CR-6)で表される化合物(以下「CR-6」とも記す。)と同定した。
・化合物の安全溶媒溶解度試験
上記合成実施例1~6及び合成例1~2で得られた化合物のプロピレングリコールモノメチルエーテル(PGME)、及びシクロヘキサノン(CHN)への溶解量を以下のとおり評価した。該評価結果を第1表に示す。
〔評価基準〕
A:5.0質量% ≦ 溶解量
B:3.0質量%≦ 溶解量 <5.0質量%
C:溶解量 <3.0質量%
※溶解量(質量%)=23℃の溶媒100g中に溶解した化合物の質量(g)/(溶媒の質量100g+23℃の溶媒100g中に溶解した化合物の質量(g))×100
(1)感放射線性組成物を調製
第2表に記載の成分を調合し、均一溶液とした後、該溶液を孔径0.1μmのテフロン製メンブランフィルターで濾過して、感放射線性組成物を調製した。
P-1:トリフェニルベンゼンスルホニウムトリフルオロメタンスルホネート(みどり化学(株))
酸架橋剤(G)
C-1:ニカラックMW-100LM(三和ケミカル(株))
酸拡散制御剤(E)
Q-1:トリオクチルアミン(東京化成工業(株))
溶媒
S-1:プロピレングリコールモノメチルエーテル(東京化成工業(株))
上記調製した感放射線性組成物を用いて以下のとおりパターニング試験を行った。
上記調製した感放射線性組成物を清浄なシリコンウェハー上に回転塗布した後、110℃のオーブン中で露光前ベーク(PB)して、厚さ60nmのレジスト膜を形成した。該レジスト膜を電子線描画装置(ELS-7500、(株)エリオニクス社製)を用いて、50nm、40nm及び30nm間隔の1:1のラインアンドスペース設定の電子線を照射した。照射後に、それぞれ110℃で、90秒間加熱し、実施例7及び8で調製した感放射線性組成物から形成されたレジスト膜は、プロピレングリコールモノメチルエーテルアセテート現像液に30秒間、比較例3及び4で調製した感放射線性組成物から形成されたレジスト膜は2.38質量%TMAH水溶液に浸漬して現像を行った。その後、該レジスト膜を超純水で30秒間洗浄し、乾燥して、ネガ型のレジストパターンを形成した。
Claims (17)
- 分子量500~5000の、式(1)で示される環状化合物。
前記R0の少なくともひとつが(メタ)アクリロイル基を含む一価の基である。) - 前記式(1)で示される環状化合物が、式(2)で示される請求項1に記載の環状化合物。
R2は、それぞれ独立して、水素原子、シアノ基、ニトロ基、ハロゲン原子、置換若しくは無置換の炭素数1~20の直鎖状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の分岐状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の環状脂肪族炭化水素基、置換若しくは無置換の炭素数6~20のアリール基、置換若しくは無置換の炭素数7~30のアラルキル基、置換若しくは無置換の炭素数1~20のアルコキシ基、置換若しくは無置換の炭素数2~20のアルケニル基、置換若しくは無置換の炭素数1~20のアシル基、置換若しくは無置換の炭素数2~20のアルコキシカルボニル基、置換若しくは無置換の炭素数1~20のアルキロイルオキシ基、置換若しくは無置換の炭素数7~30のアリーロイルオキシ基又は置換若しくは無置換の炭素数1~20のアルキルシリル基であり、
R’は、それぞれ独立して、水素原子、置換若しくは無置換の炭素数1~20の直鎖状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の分岐状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の環状脂肪族炭化水素基又は下記式(3)
前記R1の少なくともひとつが、(メタ)アクリロイル基である。) - 前記式(2)で示される環状化合物が、式(4)で示される請求項2に記載の環状化合物。
R4は、それぞれ独立して、シアノ基、ニトロ基、置換若しくは無置換の複素環基、ハロゲン原子、置換若しくは無置換の炭素数1~20の直鎖状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の分岐状脂肪族炭化水素基、置換若しくは無置換の炭素数3~20の環状脂肪族炭化水素基、置換若しくは無置換の炭素数6~20のアリール基、置換若しくは無置換の炭素数1~20のアルコキシ基又は置換若しくは無置換の炭素数1~20のアルキルシリル基であり、
pは0~5の整数であり、
前記R1の少なくともひとつが、(メタ)アクリロイル基である。) - 請求項1~4のいずれか1項に記載の環状化合物を製造する方法であって、
1種以上のアルデヒド性化合物(A1)と1種以上のフェノール性化合物(A2)とを縮合反応させて環状化合物(A)を得る工程と、
該環状化合物(A)と1種以上の(メタ)アクリロイルハライドとを脱ハロゲン化水素反応させる工程とを備える、環状化合物の製造方法。 - 前記アルデヒド性化合物(A1)が、1~4個のホルミル基を有し、かつ炭素数1~59であり、
前記フェノール性化合物(A2)が、1~3個のフェノール性水酸基を有し、かつ炭素数6~15である、請求項5に記載の環状化合物の製造方法。 - 請求項1~4のいずれか1項に記載の環状化合物を含む固形成分及び溶媒を含有する、感放射線性組成物。
- 前記固形成分の含有量が1~80重量%である、請求項7に記載の感放射線性組成物。
- 前記固形成分の全重量に占める前記環状化合物の割合が50~99.999重量%である、請求項7又は8に記載の感放射線性組成物。
- 可視光線、紫外線、エキシマレーザー、電子線、極端紫外線(EUV)、X線及びイオンビームからなる群から選ばれるいずれかの放射線の照射により直接的又は間接的に酸を発生する酸発生剤(C)をさらに含む、請求項7~9のいずれか1項に記載の感放射線性組成物。
- 酸架橋剤(G)をさらに含む、請求項7~10のいずれか1項に記載の感放射線性組成物。
- 酸拡散制御剤(E)をさらに含む、請求項7~11のいずれか1項に記載の感放射線性組成物。
- 前記固形成分が、環状化合物50~99.4重量%、酸発生剤(C)0.001~49重量%、酸架橋剤(G)0.5~49重量%、及び酸拡散制御剤(E)0.001~49重量%を含有する、請求項7~12のいずれか1項に記載の感放射線性組成物。
- スピンコートによりアモルファス膜を形成する、請求項7~13のいずれか1項に記載の感放射線性組成物。
- 前記アモルファス膜の23℃における現像液に対する溶解速度が10Å/sec以上である、請求項14に記載の感放射線性組成物。
- KrFエキシマレーザー、極端紫外線、電子線若しくはX線を照射した後の前記アモルファス膜、又は20~250℃で加熱した後の前記アモルファス膜の、現像液に対する溶解速度が5Å/sec以下である、請求項14又は15に記載の感放射線性組成物。
- 請求項7~16のいずれか1項に記載の感放射線性組成物を、基板上に塗布してレジスト膜を形成する工程と、
該レジスト膜を露光する工程と、
該露光したレジスト膜を現像する工程とを備える、レジストパターン形成方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201280056217.3A CN103958455A (zh) | 2011-11-18 | 2012-11-14 | 环状化合物、其制造方法、辐射敏感组合物及抗蚀图案形成方法 |
| KR1020147013241A KR102004692B1 (ko) | 2011-11-18 | 2012-11-14 | 환상 화합물, 그 제조방법, 감방사선성 조성물 및 레지스트 패턴 형성방법 |
| JP2013544300A JP6313045B2 (ja) | 2011-11-18 | 2012-11-14 | 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 |
| US14/359,081 US9182666B2 (en) | 2011-11-18 | 2012-11-14 | Cyclic compound, method for producing the same, radiation-sensitive composition, and resist pattern formation method |
| EP12849981.1A EP2781504B1 (en) | 2011-11-18 | 2012-11-14 | Cyclic compound, method for producing same, radiation-sensitive composition, and method for forming resist pattern |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011253312 | 2011-11-18 | ||
| JP2011-253312 | 2011-11-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013073582A1 true WO2013073582A1 (ja) | 2013-05-23 |
Family
ID=48429637
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/079528 WO2013073582A1 (ja) | 2011-11-18 | 2012-11-14 | 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9182666B2 (ja) |
| EP (1) | EP2781504B1 (ja) |
| JP (1) | JP6313045B2 (ja) |
| KR (1) | KR102004692B1 (ja) |
| CN (1) | CN103958455A (ja) |
| TW (1) | TW201339133A (ja) |
| WO (1) | WO2013073582A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017145320A (ja) * | 2016-02-17 | 2017-08-24 | 公立大学法人 富山県立大学 | テンプレート形成用重合性化合物及びその硬化性組成物並びにその硬化物 |
| JP2018536885A (ja) * | 2015-08-28 | 2018-12-13 | ヨンチャン ケミカル カンパニー リミテッドYoung Chang Chemical Co.,Ltd | エッチング耐性に優れたi線用ネガ型フォトレジスト組成物 |
| US10550068B2 (en) | 2015-07-23 | 2020-02-04 | Mitsubishi Gas Chemical Company, Inc. | Compound and method for producing same |
| US10723690B2 (en) | 2015-07-23 | 2020-07-28 | Mitsubishi Gas Chemical Company, Inc. | (Meth)acryloyl compound and method for producing same |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107430337A (zh) * | 2015-03-30 | 2017-12-01 | 三菱瓦斯化学株式会社 | 辐射敏感组合物及抗蚀图案形成方法 |
| US11487204B2 (en) * | 2017-07-27 | 2022-11-01 | Dic Corporation | Resist material |
| US11858899B2 (en) * | 2021-06-10 | 2024-01-02 | University Of Cincinnati | Complexation of pirfenidone with polyphenolic calixarene or resorcin[4]arenes |
Citations (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61226745A (ja) | 1985-03-30 | 1986-10-08 | Japan Synthetic Rubber Co Ltd | 半導体集積回路製造用のスピンコート用レジスト組成物 |
| JPS61226746A (ja) | 1985-03-30 | 1986-10-08 | Japan Synthetic Rubber Co Ltd | 半導体集積回路製造用のスピンコート用レジスト組成物 |
| JPS6236663A (ja) | 1985-08-12 | 1987-02-17 | Mitsubishi Chem Ind Ltd | ナフトキノンジアジド系化合物及び該化合物を含有するポジ型フオトレジスト組成物 |
| JPS62170950A (ja) | 1986-01-23 | 1987-07-28 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS6334540A (ja) | 1986-07-30 | 1988-02-15 | Mitsubishi Chem Ind Ltd | ポジ型フオトレジスト組成物 |
| US5294511A (en) | 1990-01-16 | 1994-03-15 | Fuji Photo Film Co., Ltd. | Photosensitive composition |
| US5296330A (en) | 1991-08-30 | 1994-03-22 | Ciba-Geigy Corp. | Positive photoresists containing quinone diazide photosensitizer, alkali-soluble resin and tetra(hydroxyphenyl) alkane additive |
| JPH06282067A (ja) | 1992-09-14 | 1994-10-07 | Fuji Photo Film Co Ltd | ポジ型感光性平版印刷版 |
| US5360692A (en) | 1992-06-04 | 1994-11-01 | Fuji Photo Film Co., Ltd. | Positive type 1,2-naphthoquinonediazide photoresist composition containing benzotriazole light absorbing agent |
| EP0632003A1 (en) | 1993-06-30 | 1995-01-04 | Fuji Photo Film Co., Ltd. | Novel phenol compounds containing methoxymethyl group or hydroxymethyl group |
| JPH0764285A (ja) | 1993-08-31 | 1995-03-10 | Fuji Photo Film Co Ltd | ポジ型感光性平版印刷版 |
| US5405720A (en) | 1985-08-07 | 1995-04-11 | Japan Synthetic Rubber Co., Ltd. | Radiation-sensitive composition containing 1,2 quinonediazide compound, alkali-soluble resin and monooxymonocarboxylic acid ester solvent |
| JPH07230165A (ja) | 1993-06-30 | 1995-08-29 | Tokyo Ohka Kogyo Co Ltd | ポジ型ホトレジスト組成物 |
| JPH0862834A (ja) | 1994-08-22 | 1996-03-08 | Mitsubishi Chem Corp | フォトレジスト組成物 |
| US5529881A (en) | 1994-03-17 | 1996-06-25 | Fuji Photo Film Co., Ltd. | Postive photoresist composition |
| US5576143A (en) | 1991-12-03 | 1996-11-19 | Fuji Photo Film Co., Ltd. | Light-sensitive composition |
| JPH095988A (ja) | 1995-06-21 | 1997-01-10 | Mitsubishi Chem Corp | 感放射線性塗布組成物 |
| JPH0954432A (ja) | 1995-08-18 | 1997-02-25 | Dainippon Ink & Chem Inc | フォトレジスト組成物 |
| US5824451A (en) | 1994-07-04 | 1998-10-20 | Fuji Photo Film Co., Ltd. | Positive photosensitive composition |
| JP2000047018A (ja) * | 1998-07-28 | 2000-02-18 | Jsr Corp | カラーフィルタ用感放射線性組成物 |
| JP2004018421A (ja) * | 2002-06-13 | 2004-01-22 | Tokuyama Corp | 新規カリックスレゾルシナレーン誘導体 |
| JP2005326838A (ja) | 2004-04-15 | 2005-11-24 | Mitsubishi Gas Chem Co Inc | レジスト組成物 |
| JP2008145539A (ja) | 2006-12-06 | 2008-06-26 | Mitsubishi Gas Chem Co Inc | 感放射線性レジスト組成物 |
| JP2009173623A (ja) | 2007-04-23 | 2009-08-06 | Mitsubishi Gas Chem Co Inc | 感放射線性組成物 |
| JP2010113117A (ja) * | 2008-11-06 | 2010-05-20 | Konica Minolta Business Technologies Inc | 電子写真感光体と画像形成装置 |
| WO2011024916A1 (ja) | 2009-08-31 | 2011-03-03 | 三菱瓦斯化学株式会社 | 環状化合物、その製造方法、感放射線性組成物およびレジストパターン形成方法 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4695615A (en) | 1984-11-21 | 1987-09-22 | Loctite (Ireland) Limited | Instant adhesive composition utilizing mixed functionality calixarenes as accelerators |
| US4642362A (en) | 1984-01-30 | 1987-02-10 | Loctite (Ireland) Limited | Polymer bound calixarenes |
| IE893986A1 (en) * | 1989-12-13 | 1991-06-19 | Loctite Ireland Ltd | Polymerisable calixarene and oxacalixarene derivatives,¹polymers thereof, and use of such derivatives and polymers¹for sequestration of metals |
| IE904444A1 (en) * | 1990-12-10 | 1992-06-17 | Stephen J Harris | Ion-selective electrodes |
| WO1995019974A2 (en) * | 1994-01-24 | 1995-07-27 | Harris Stephen J | Calixarene-based compounds having antibacterial, antifungal, anticancer-hiv activity |
| JPH10310545A (ja) | 1997-05-09 | 1998-11-24 | Jsr Corp | フェノール系デンドリマー化合物およびそれを含む感放射線性組成物 |
| JP2002072477A (ja) | 2000-06-12 | 2002-03-12 | Jsr Corp | 感放射線性樹脂組成物 |
| WO2006129574A1 (ja) * | 2005-06-01 | 2006-12-07 | Idemitsu Kosan Co., Ltd. | カリックスレゾルシナレン化合物、並びに、それからなるフォトレジスト基材及びその組成物 |
| US8110334B2 (en) | 2006-11-02 | 2012-02-07 | Mitsubishi Gas Chemical Company, Inc. | Radiation-sensitive composition |
| JP5750777B2 (ja) | 2009-04-24 | 2015-07-22 | 日立化成株式会社 | カリックスアレーン誘導体、カリックスアレーン誘導体の製造方法、エポキシ樹脂用硬化剤、エポキシ樹脂組成物及び電子部品装置 |
| KR101288573B1 (ko) | 2009-08-03 | 2013-07-22 | 제일모직주식회사 | 칼릭스 알렌이 함유된 고 내에칭성 반사방지 하드마스크 조성물, 이를 이용한 패턴화된 재료 형상의 제조방법 |
| JP5733211B2 (ja) | 2009-08-31 | 2015-06-10 | 三菱瓦斯化学株式会社 | 環状化合物、その製造方法、感放射線性組成物およびレジストパターン形成方法 |
| WO2012133040A1 (ja) | 2011-03-28 | 2012-10-04 | 株式会社トクヤマ | カリックスアレーン誘導体 |
| JP5618893B2 (ja) | 2011-04-20 | 2014-11-05 | 株式会社トクヤマ | カリックス[4]アレーン組成物 |
-
2012
- 2012-11-14 KR KR1020147013241A patent/KR102004692B1/ko active IP Right Grant
- 2012-11-14 US US14/359,081 patent/US9182666B2/en not_active Expired - Fee Related
- 2012-11-14 WO PCT/JP2012/079528 patent/WO2013073582A1/ja active Application Filing
- 2012-11-14 JP JP2013544300A patent/JP6313045B2/ja active Active
- 2012-11-14 CN CN201280056217.3A patent/CN103958455A/zh active Pending
- 2012-11-14 EP EP12849981.1A patent/EP2781504B1/en not_active Not-in-force
- 2012-11-16 TW TW101142887A patent/TW201339133A/zh unknown
Patent Citations (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61226746A (ja) | 1985-03-30 | 1986-10-08 | Japan Synthetic Rubber Co Ltd | 半導体集積回路製造用のスピンコート用レジスト組成物 |
| JPS61226745A (ja) | 1985-03-30 | 1986-10-08 | Japan Synthetic Rubber Co Ltd | 半導体集積回路製造用のスピンコート用レジスト組成物 |
| US5405720A (en) | 1985-08-07 | 1995-04-11 | Japan Synthetic Rubber Co., Ltd. | Radiation-sensitive composition containing 1,2 quinonediazide compound, alkali-soluble resin and monooxymonocarboxylic acid ester solvent |
| JPS6236663A (ja) | 1985-08-12 | 1987-02-17 | Mitsubishi Chem Ind Ltd | ナフトキノンジアジド系化合物及び該化合物を含有するポジ型フオトレジスト組成物 |
| JPS62170950A (ja) | 1986-01-23 | 1987-07-28 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS6334540A (ja) | 1986-07-30 | 1988-02-15 | Mitsubishi Chem Ind Ltd | ポジ型フオトレジスト組成物 |
| US5294511A (en) | 1990-01-16 | 1994-03-15 | Fuji Photo Film Co., Ltd. | Photosensitive composition |
| US5436098A (en) | 1991-08-30 | 1995-07-25 | Ciba-Geigy Corporation | Positive photoresists with enhanced resolution and reduced crystallization containing novel tetra(hydroxyphenyl)alkanes |
| US5296330A (en) | 1991-08-30 | 1994-03-22 | Ciba-Geigy Corp. | Positive photoresists containing quinone diazide photosensitizer, alkali-soluble resin and tetra(hydroxyphenyl) alkane additive |
| US5576143A (en) | 1991-12-03 | 1996-11-19 | Fuji Photo Film Co., Ltd. | Light-sensitive composition |
| US5360692A (en) | 1992-06-04 | 1994-11-01 | Fuji Photo Film Co., Ltd. | Positive type 1,2-naphthoquinonediazide photoresist composition containing benzotriazole light absorbing agent |
| JPH06282067A (ja) | 1992-09-14 | 1994-10-07 | Fuji Photo Film Co Ltd | ポジ型感光性平版印刷版 |
| EP0632003A1 (en) | 1993-06-30 | 1995-01-04 | Fuji Photo Film Co., Ltd. | Novel phenol compounds containing methoxymethyl group or hydroxymethyl group |
| JPH07230165A (ja) | 1993-06-30 | 1995-08-29 | Tokyo Ohka Kogyo Co Ltd | ポジ型ホトレジスト組成物 |
| JPH0764285A (ja) | 1993-08-31 | 1995-03-10 | Fuji Photo Film Co Ltd | ポジ型感光性平版印刷版 |
| US5529881A (en) | 1994-03-17 | 1996-06-25 | Fuji Photo Film Co., Ltd. | Postive photoresist composition |
| US5824451A (en) | 1994-07-04 | 1998-10-20 | Fuji Photo Film Co., Ltd. | Positive photosensitive composition |
| JPH0862834A (ja) | 1994-08-22 | 1996-03-08 | Mitsubishi Chem Corp | フォトレジスト組成物 |
| JPH095988A (ja) | 1995-06-21 | 1997-01-10 | Mitsubishi Chem Corp | 感放射線性塗布組成物 |
| JPH0954432A (ja) | 1995-08-18 | 1997-02-25 | Dainippon Ink & Chem Inc | フォトレジスト組成物 |
| JP2000047018A (ja) * | 1998-07-28 | 2000-02-18 | Jsr Corp | カラーフィルタ用感放射線性組成物 |
| JP2004018421A (ja) * | 2002-06-13 | 2004-01-22 | Tokuyama Corp | 新規カリックスレゾルシナレーン誘導体 |
| JP2005326838A (ja) | 2004-04-15 | 2005-11-24 | Mitsubishi Gas Chem Co Inc | レジスト組成物 |
| JP2008145539A (ja) | 2006-12-06 | 2008-06-26 | Mitsubishi Gas Chem Co Inc | 感放射線性レジスト組成物 |
| JP2009173623A (ja) | 2007-04-23 | 2009-08-06 | Mitsubishi Gas Chem Co Inc | 感放射線性組成物 |
| JP2010113117A (ja) * | 2008-11-06 | 2010-05-20 | Konica Minolta Business Technologies Inc | 電子写真感光体と画像形成装置 |
| WO2011024916A1 (ja) | 2009-08-31 | 2011-03-03 | 三菱瓦斯化学株式会社 | 環状化合物、その製造方法、感放射線性組成物およびレジストパターン形成方法 |
Non-Patent Citations (6)
| Title |
|---|
| FU,Q. ET AL.: "Effect of C-tetramethyl calix[4]resorcinarene acrylate on curing behavior and film properties of thiol-acrylate coating system", CHEMICAL RESEARCH IN CHINESE UNIVERSITIES, vol. 25, no. 5, 2009, pages 760 - 766, XP055150078 * |
| HUANG, Z. ET AL.: "Selective Esterification of Calix[4]arenes", SYNTHETIC COMMUNICATIONS, vol. 24, no. 1, 1994, pages 11 - 22, XP008072330 * |
| HUANG,Z. ET AL.: "Study of calixarenes. V. Friedel-Crafts reaction of calixarenes", CHEMISCHE BERICHTE, vol. 127, no. 3, 1994, pages 519 - 23, XP055150079 * |
| See also references of EP2781504A4 |
| SEE,K.A. ET AL.: "Calixarenes. 26. Selective esterification and selective ester cleavage of calix[4]arenes", JOURNAL OF ORGANIC CHEMISTRY, vol. 56, no. 26, 1991, pages 7256 - 68, XP055150082 * |
| T. NAKAYAMA; M. NOMURA; K. HAGA; M. UEDA, BULL. CHEM. SOC. JPN., vol. 71, 1998, pages 2979 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10550068B2 (en) | 2015-07-23 | 2020-02-04 | Mitsubishi Gas Chemical Company, Inc. | Compound and method for producing same |
| US10723690B2 (en) | 2015-07-23 | 2020-07-28 | Mitsubishi Gas Chemical Company, Inc. | (Meth)acryloyl compound and method for producing same |
| JP2018536885A (ja) * | 2015-08-28 | 2018-12-13 | ヨンチャン ケミカル カンパニー リミテッドYoung Chang Chemical Co.,Ltd | エッチング耐性に優れたi線用ネガ型フォトレジスト組成物 |
| JP2017145320A (ja) * | 2016-02-17 | 2017-08-24 | 公立大学法人 富山県立大学 | テンプレート形成用重合性化合物及びその硬化性組成物並びにその硬化物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2781504B1 (en) | 2017-01-04 |
| JPWO2013073582A1 (ja) | 2015-04-02 |
| EP2781504A4 (en) | 2015-06-17 |
| EP2781504A1 (en) | 2014-09-24 |
| TW201339133A (zh) | 2013-10-01 |
| JP6313045B2 (ja) | 2018-04-18 |
| US20140329178A1 (en) | 2014-11-06 |
| US9182666B2 (en) | 2015-11-10 |
| KR102004692B1 (ko) | 2019-07-29 |
| CN103958455A (zh) | 2014-07-30 |
| KR20140104949A (ko) | 2014-08-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6066333B2 (ja) | 環状化合物、その製造方法、組成物及びレジストパターン形成方法 | |
| JP5982823B2 (ja) | 環状化合物、その製造方法、感放射線性組成物およびレジストパターン形成方法 | |
| WO2013024778A1 (ja) | レジスト組成物、レジストパターン形成方法、それに用いるポリフェノール化合物及びそれから誘導され得るアルコール化合物 | |
| JP5857745B2 (ja) | 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 | |
| JP6313045B2 (ja) | 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 | |
| JP5796490B2 (ja) | 環状化合物、その製造方法、感放射線性組成物およびレジストパターン形成方法 | |
| JP5786713B2 (ja) | 環状化合物、その製造方法、感放射線性組成物およびレジストパターン形成方法 | |
| JP5733211B2 (ja) | 環状化合物、その製造方法、感放射線性組成物およびレジストパターン形成方法 | |
| WO2014196425A1 (ja) | レジスト組成物 | |
| JP6268677B2 (ja) | レジスト組成物 | |
| WO2012098828A1 (ja) | 低分子化合物、感放射線性組成物、およびレジストパターン形成方法 | |
| JP6313046B2 (ja) | 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 | |
| JP2013140342A (ja) | 感放射線性組成物 | |
| WO2011037072A1 (ja) | 環状化合物、感放射線性組成物およびレジストパターン形成法 | |
| JP5821343B2 (ja) | レジストパターン形成方法 | |
| JP5817524B2 (ja) | 感放射線性組成物及びレジストパターン形成方法 | |
| JP5413070B2 (ja) | 感放射線性組成物、その製造方法およびレジストパターン形成方法 | |
| JPWO2016158881A1 (ja) | 感放射線性組成物及びレジストパターン形成方法 | |
| JP2013107841A (ja) | 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 | |
| JP2013018711A (ja) | 環状化合物、その製造方法、感放射線性組成物及びレジストパターン形成方法 | |
| WO2011037071A1 (ja) | 環状化合物、感放射線性組成物およびレジストパターン形成法 | |
| JP2016109775A (ja) | レジスト組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12849981 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013544300 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20147013241 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012849981 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14359081 Country of ref document: US Ref document number: 2012849981 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |