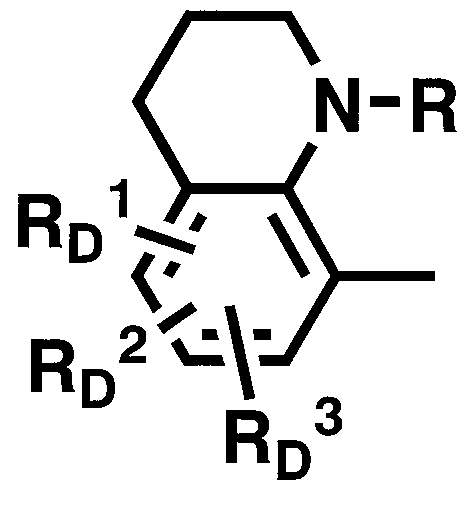
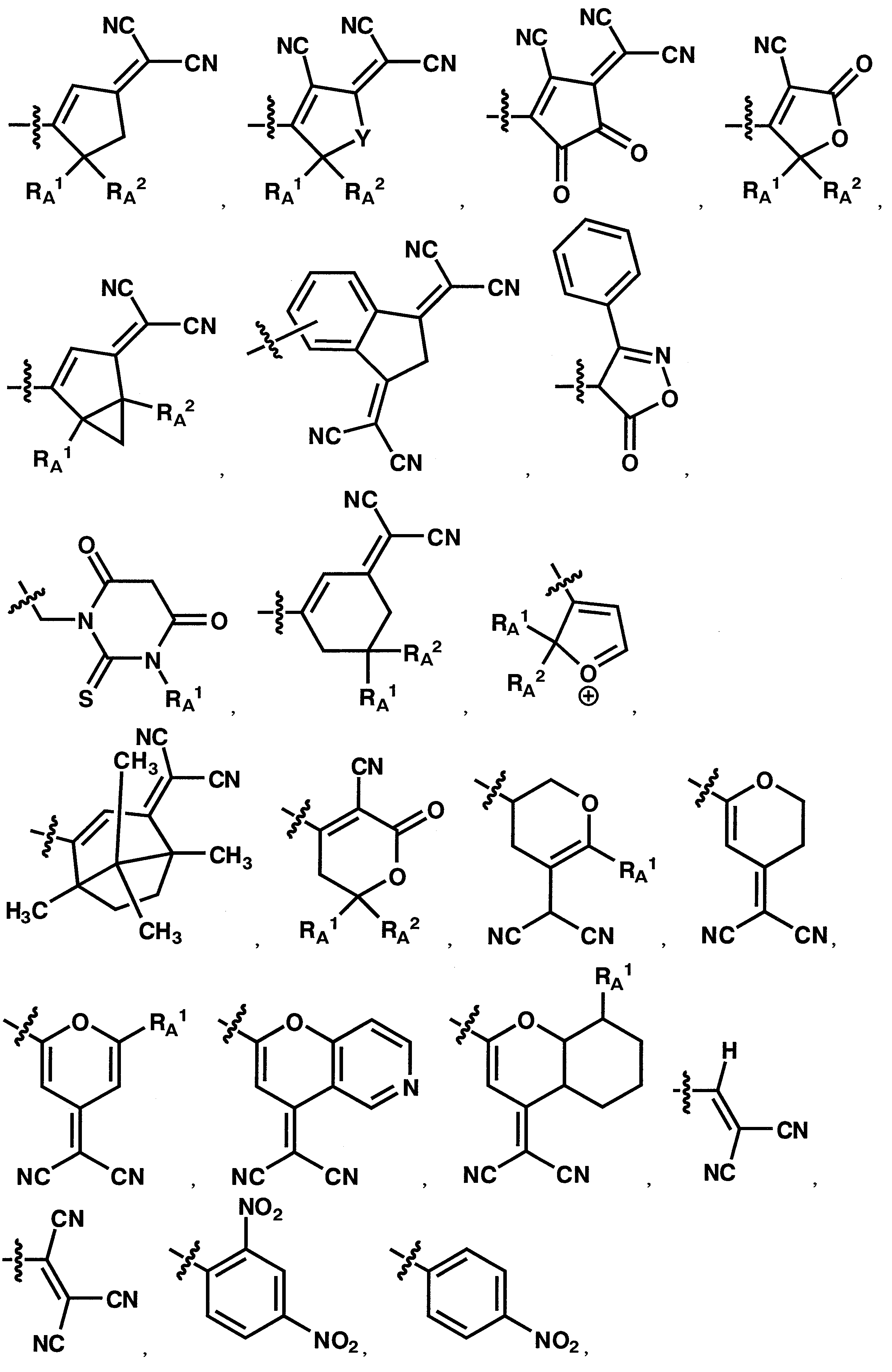
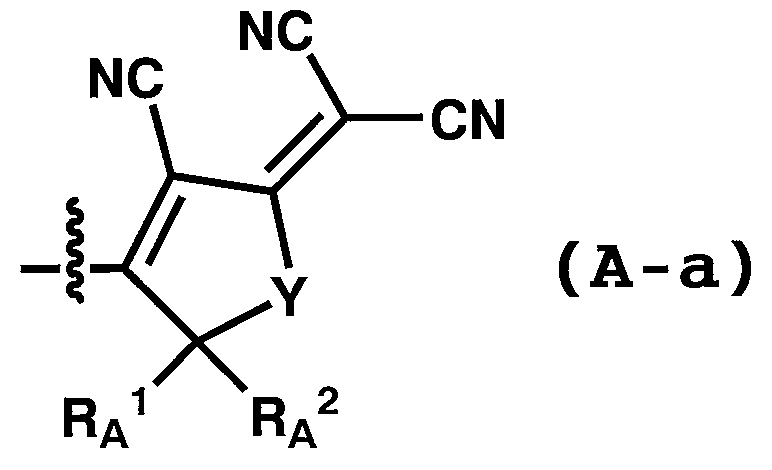
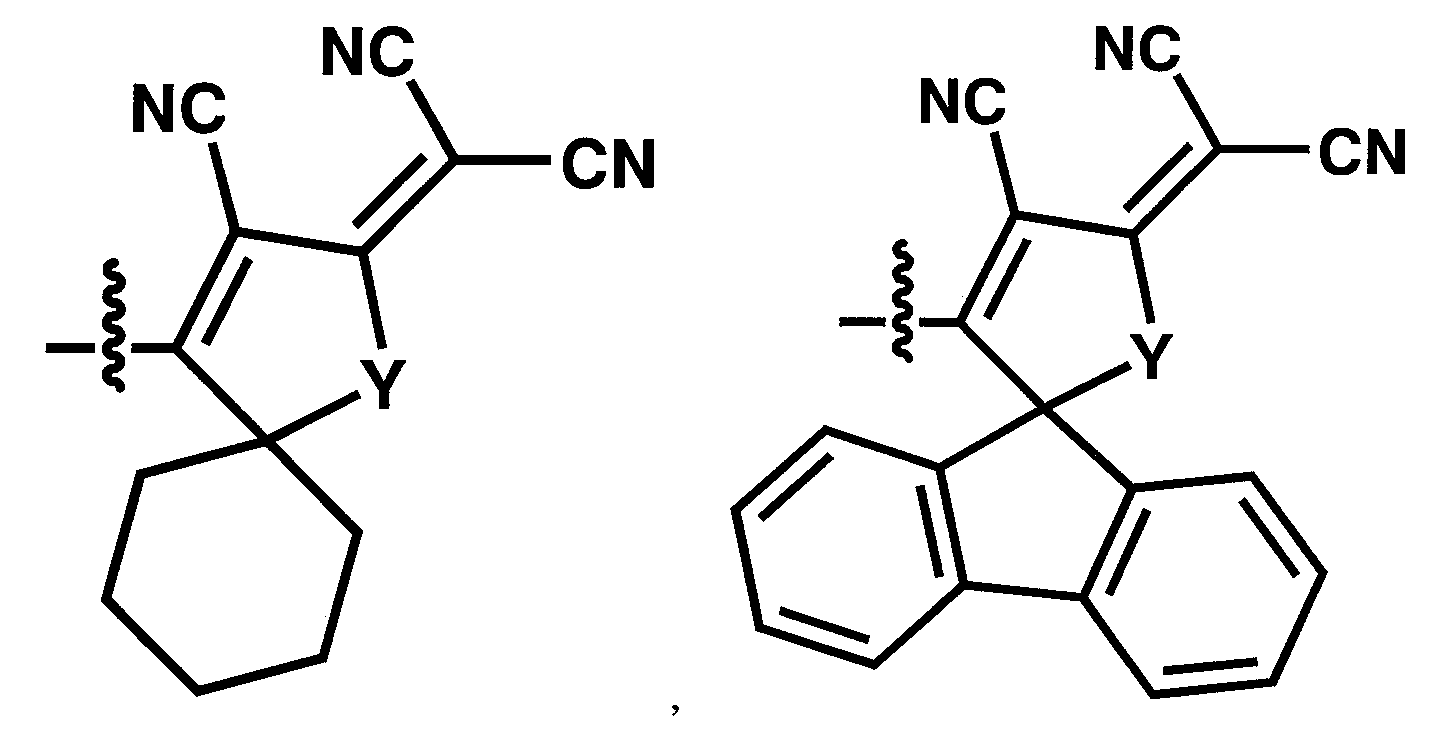
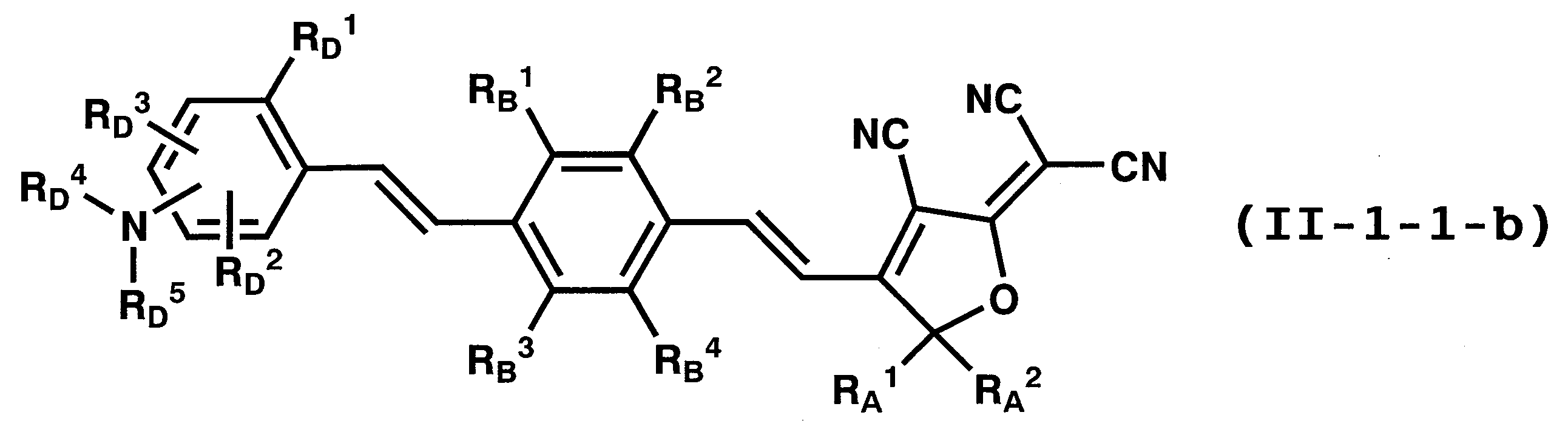
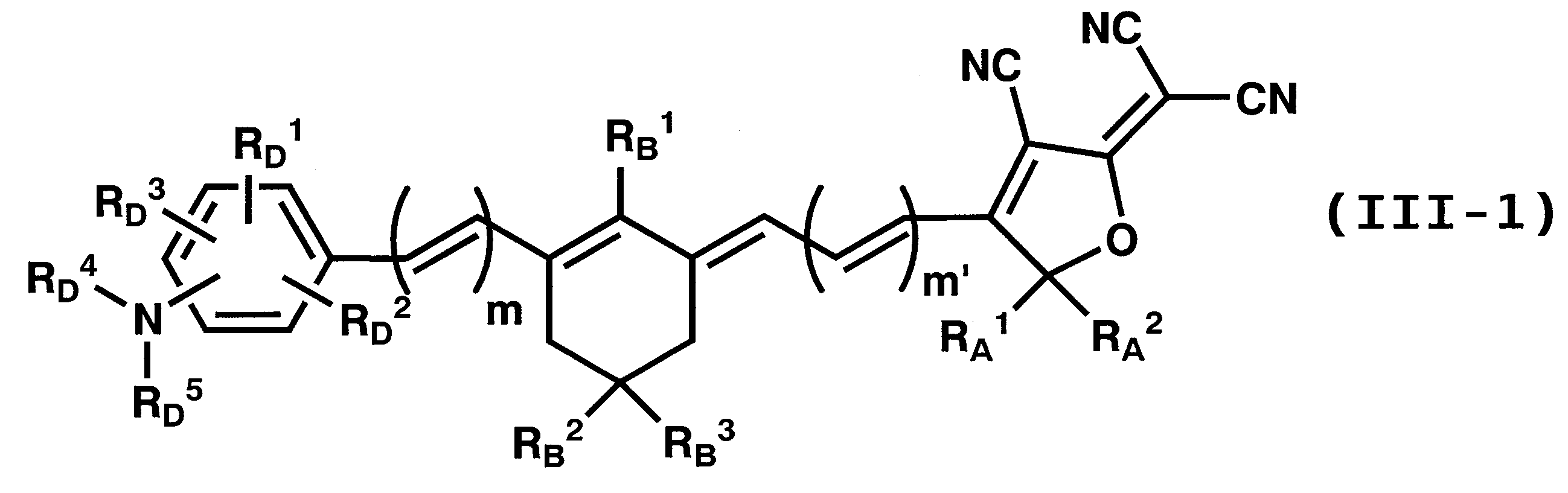
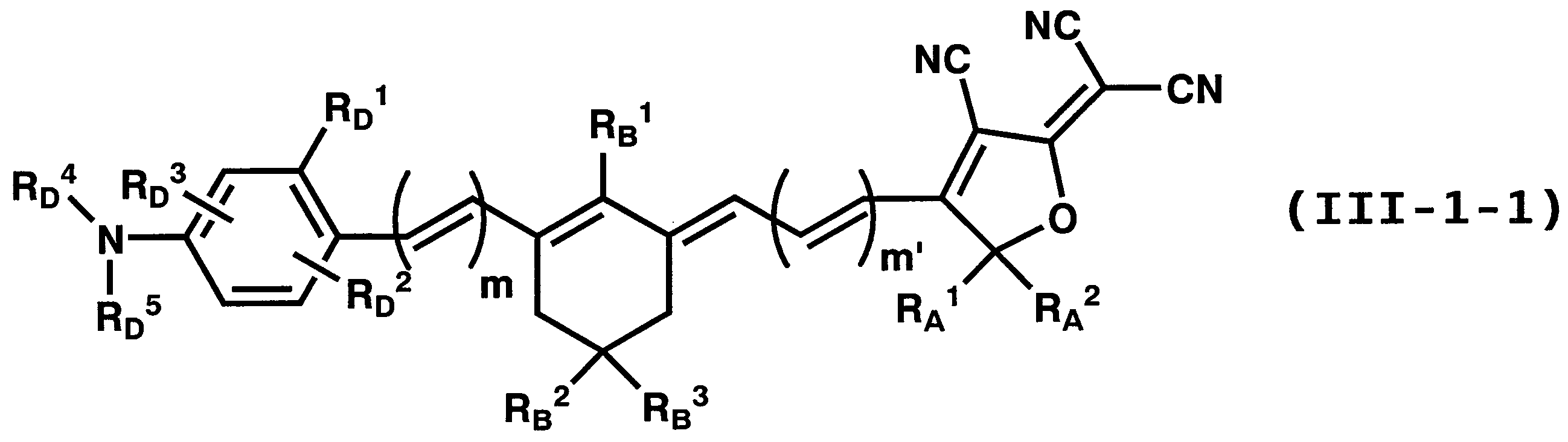
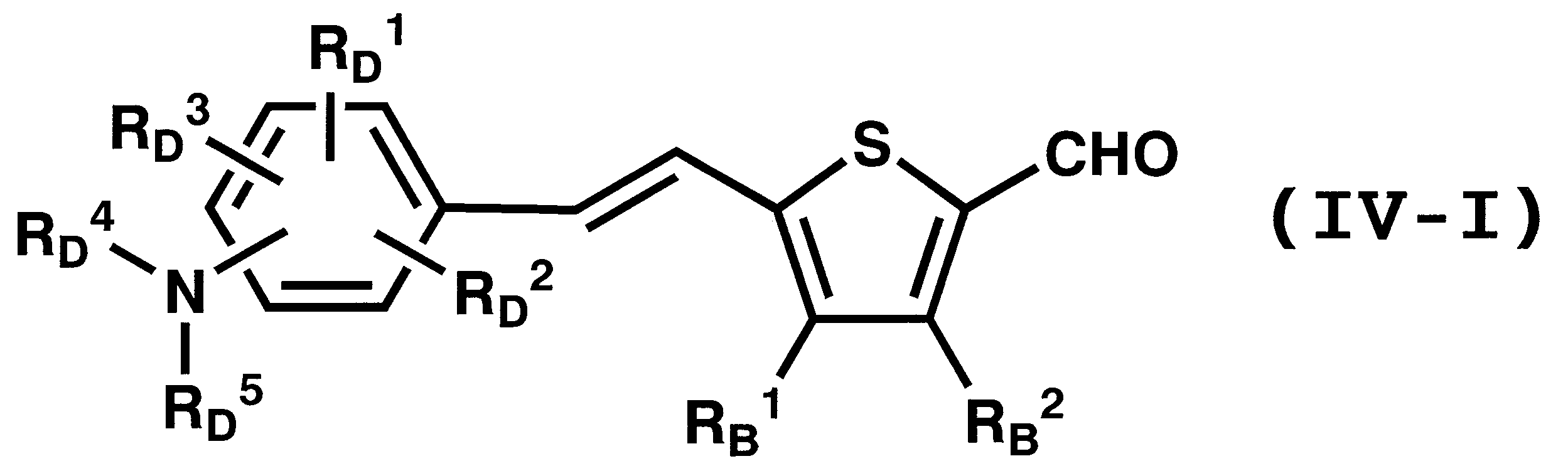
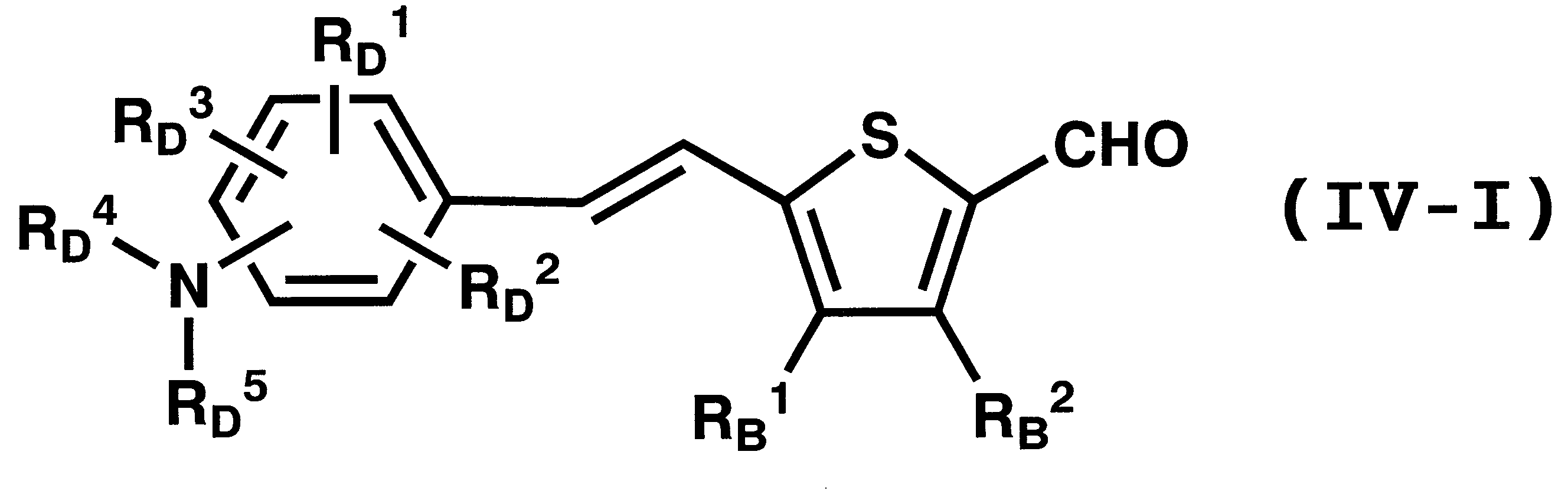
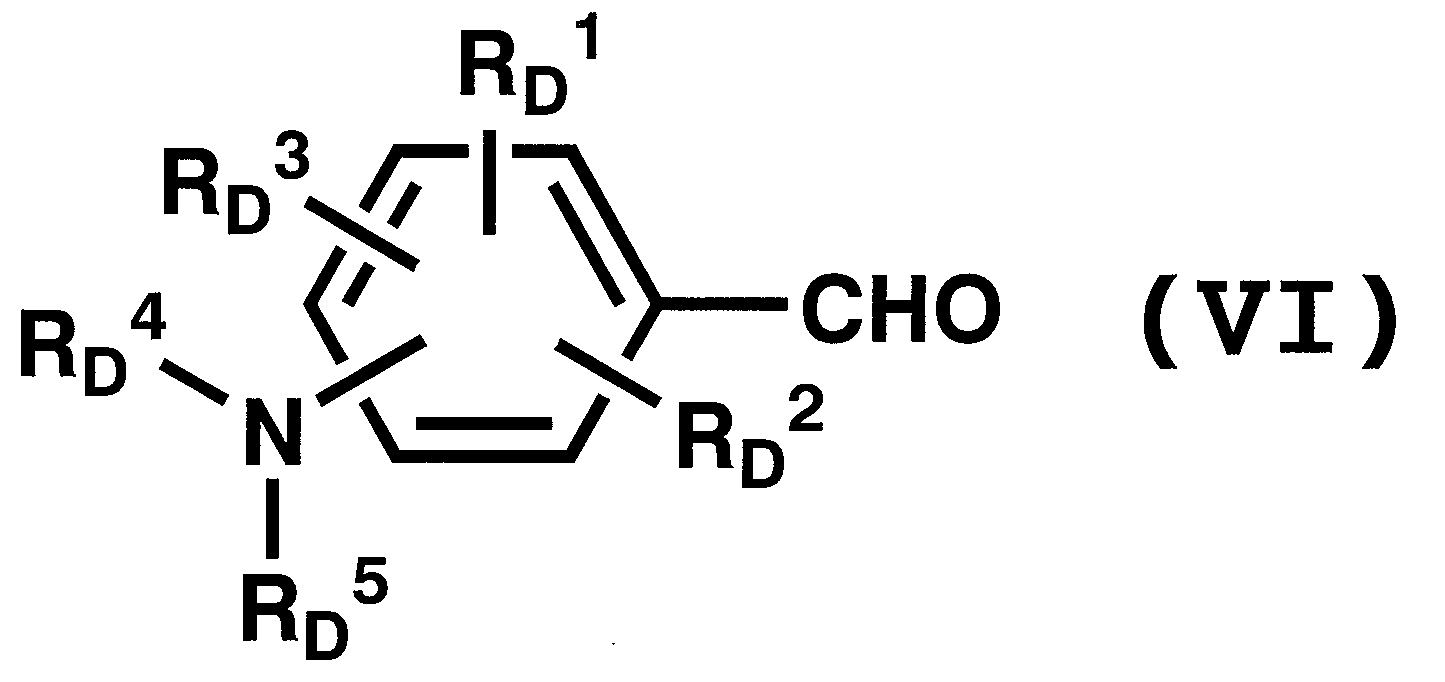
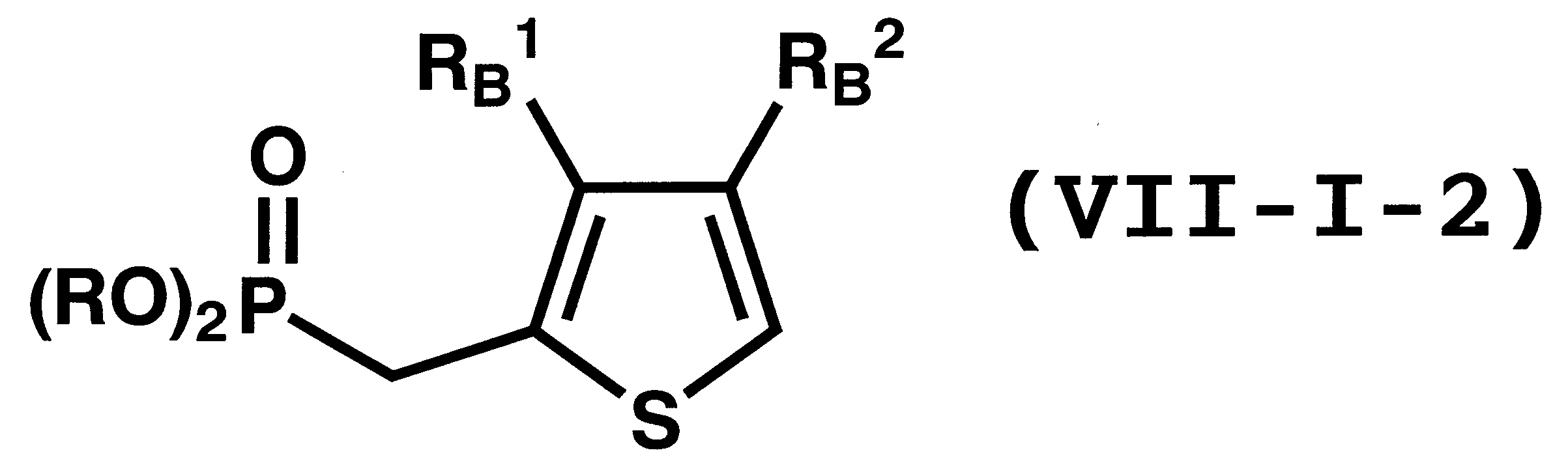
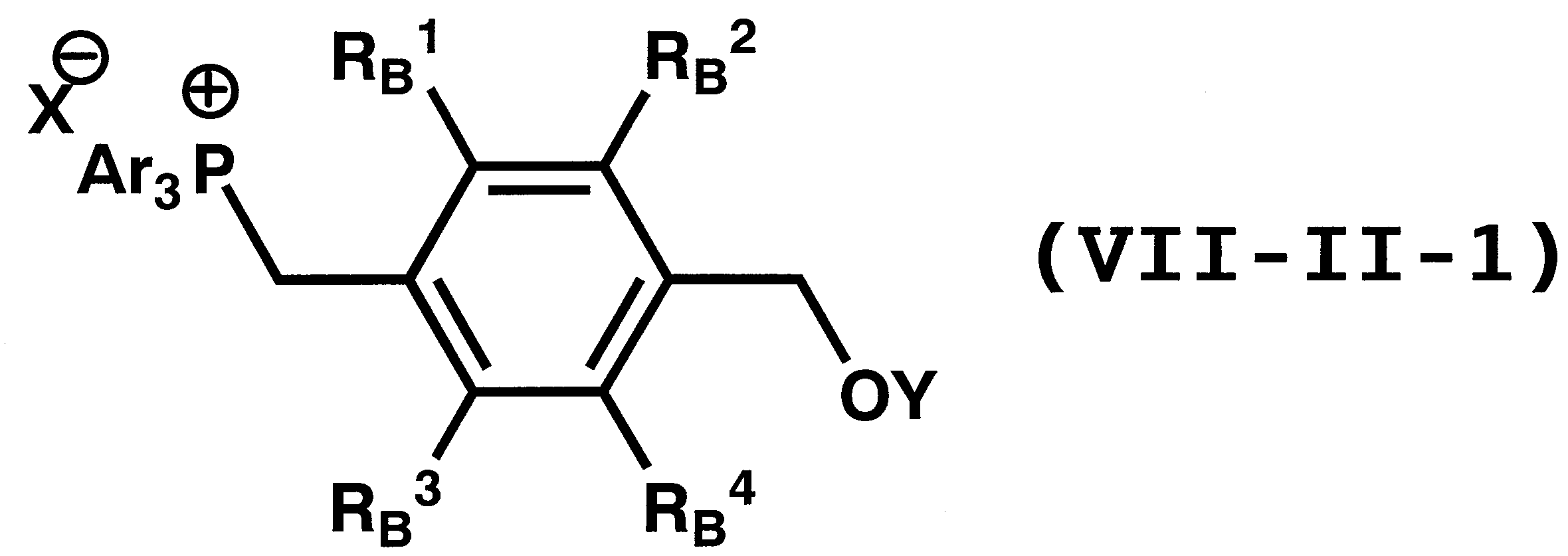
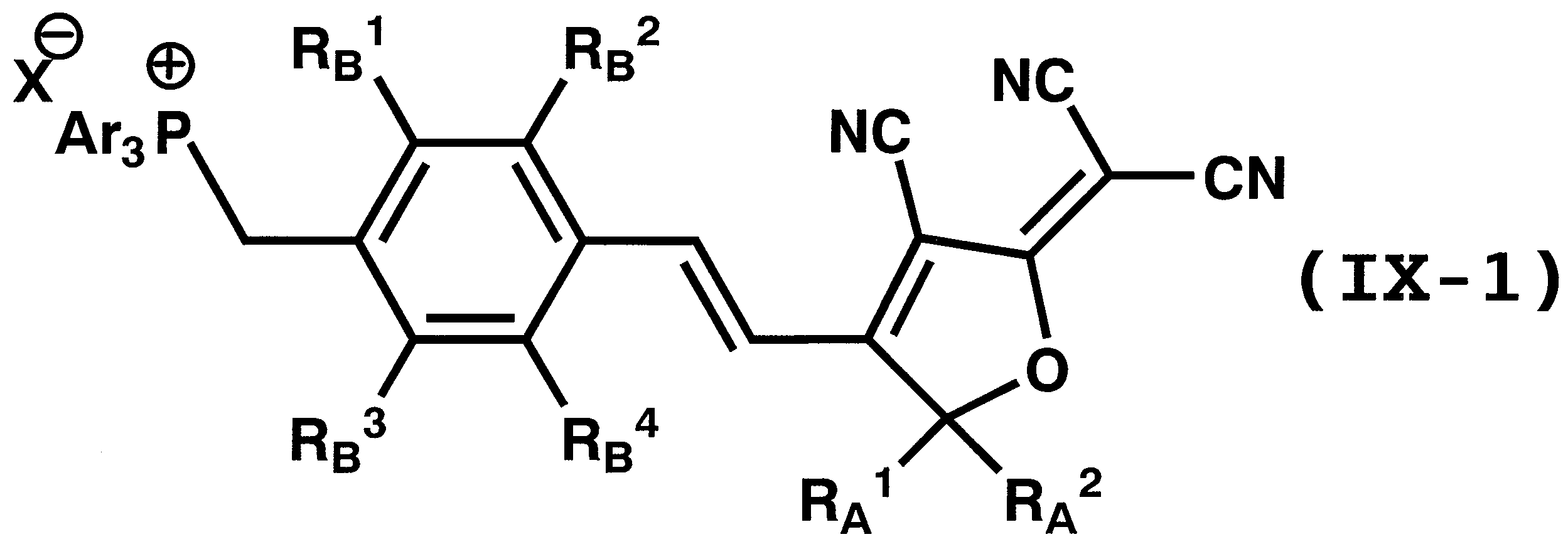
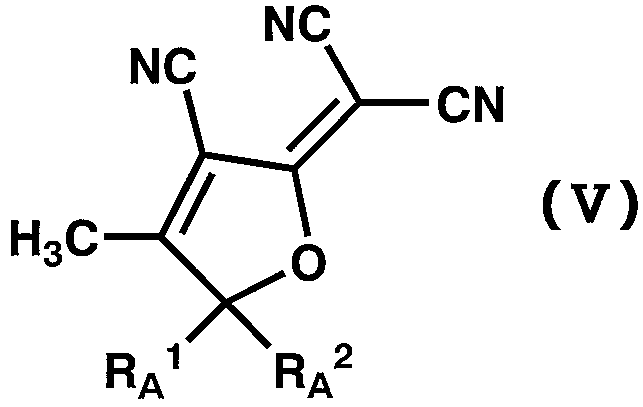
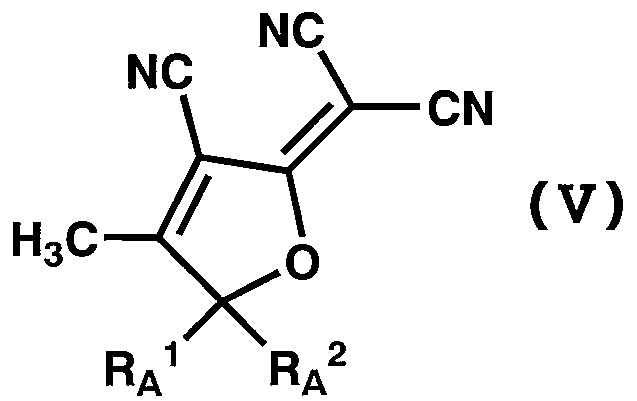
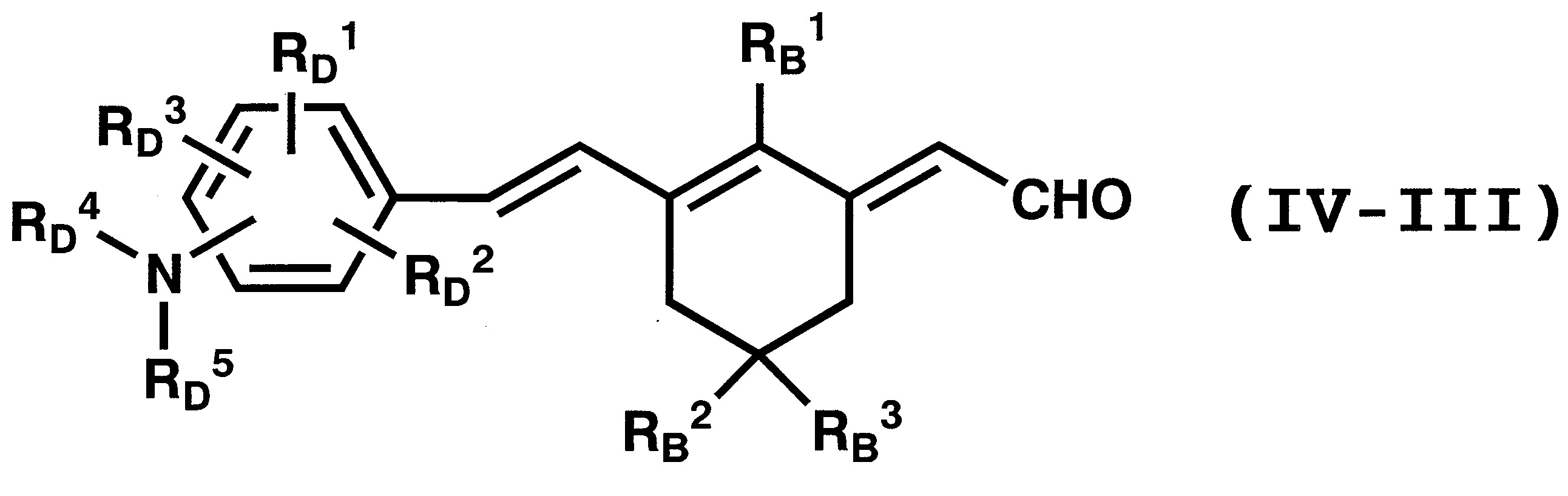
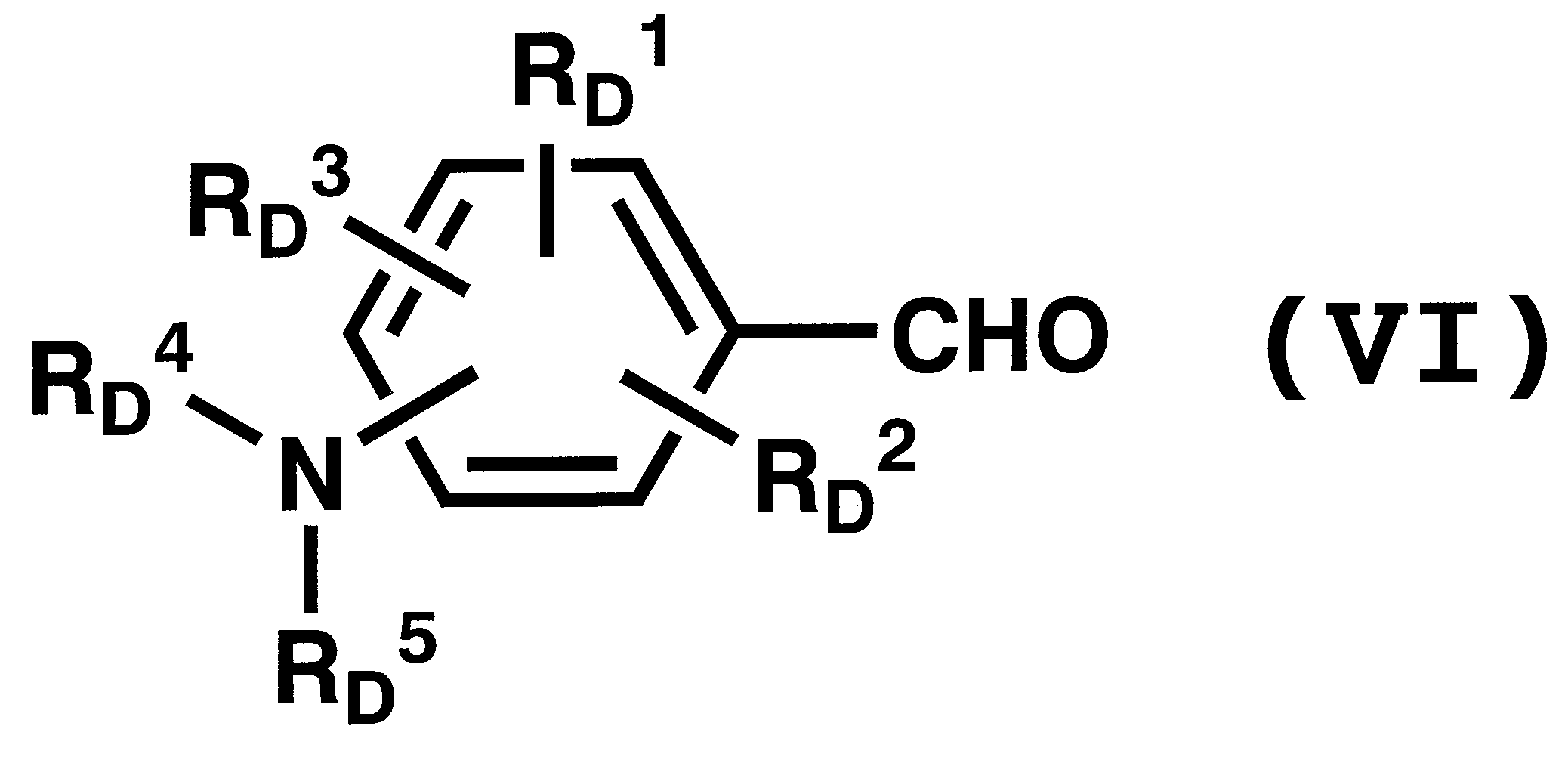
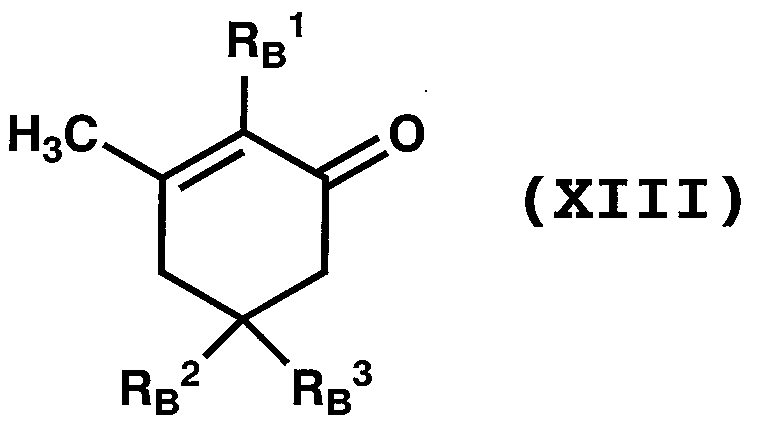
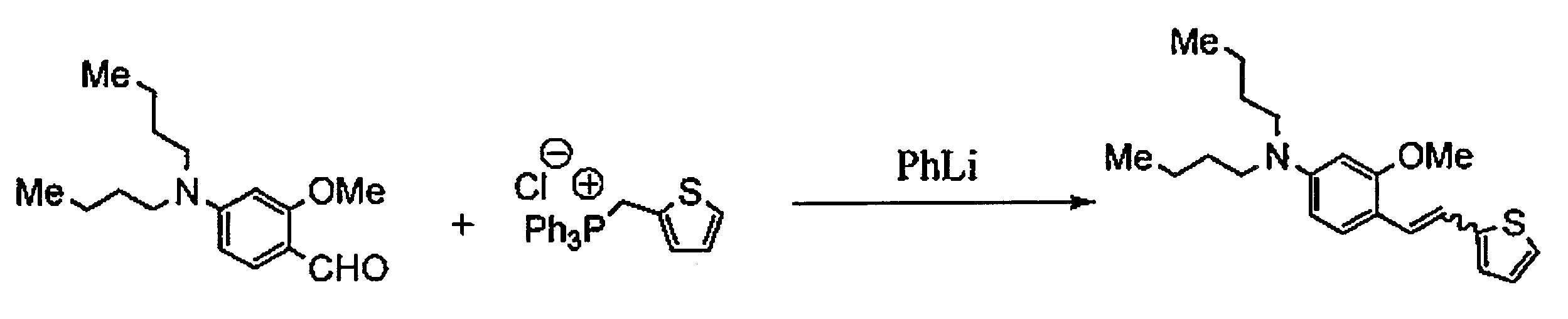
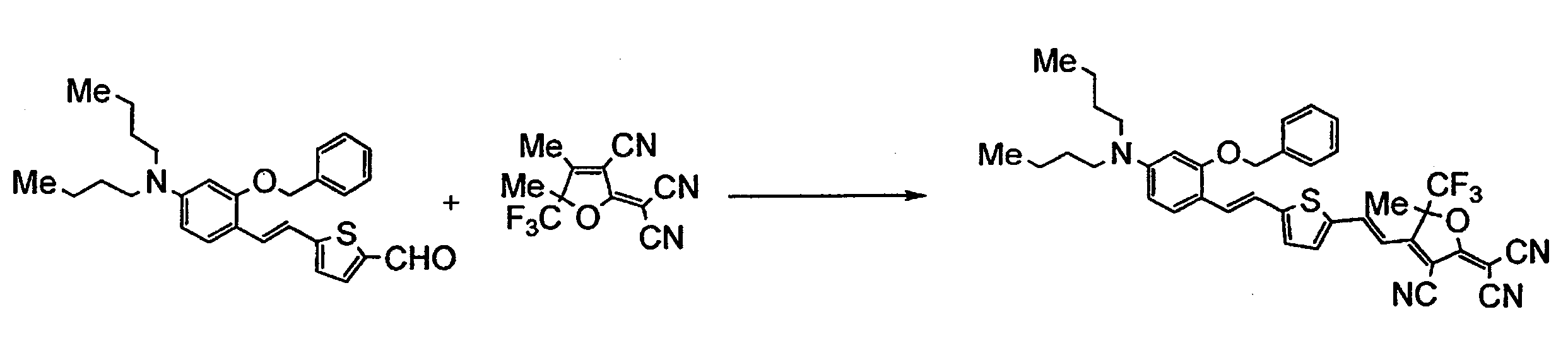
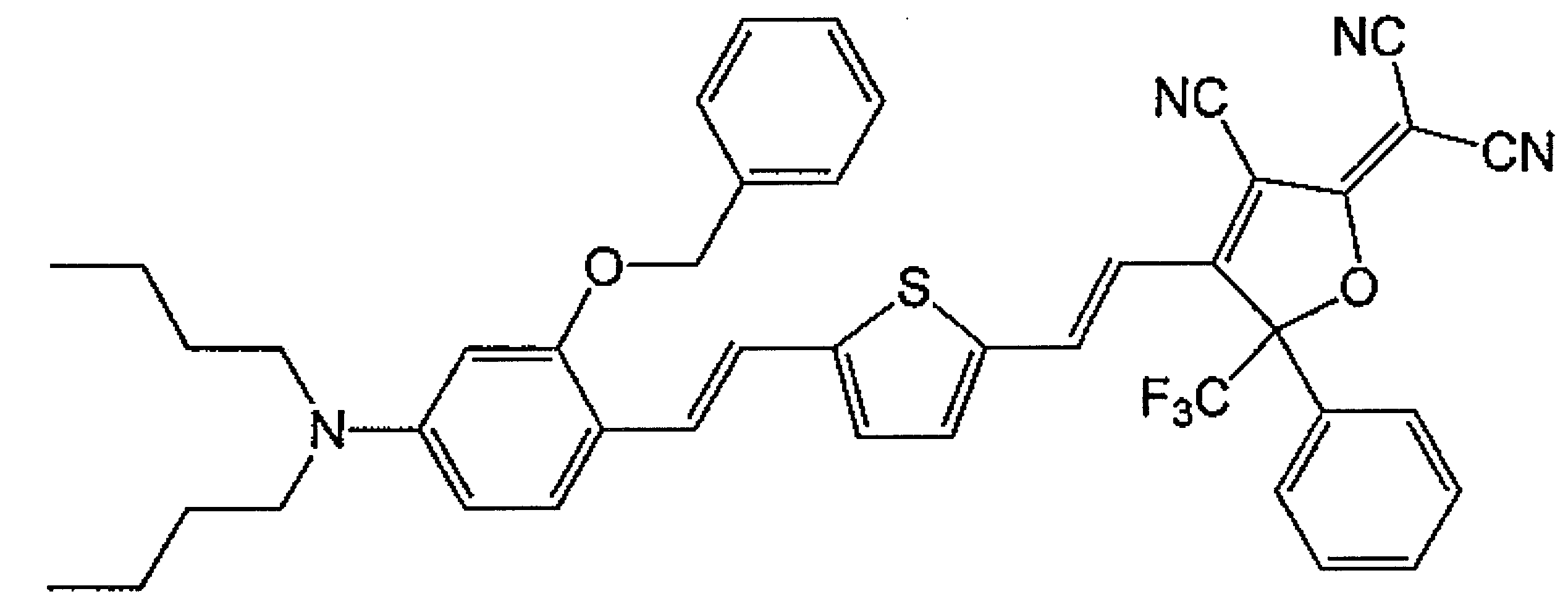
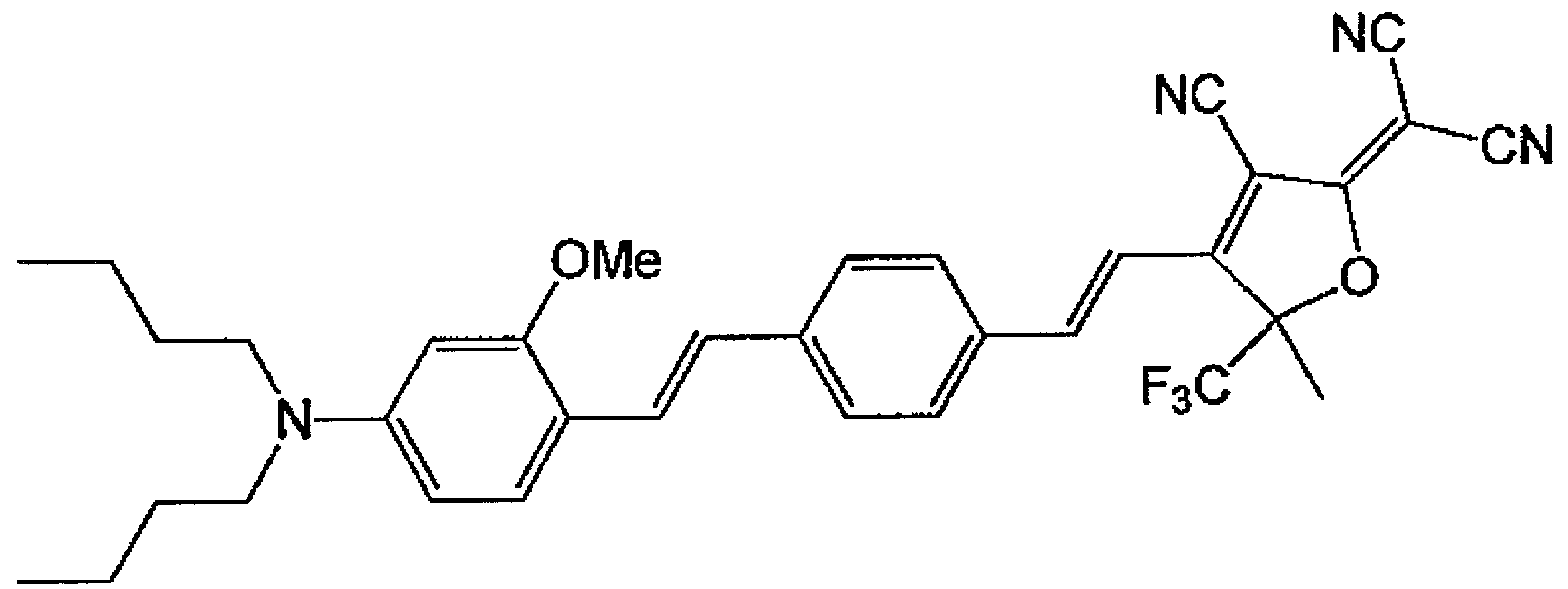
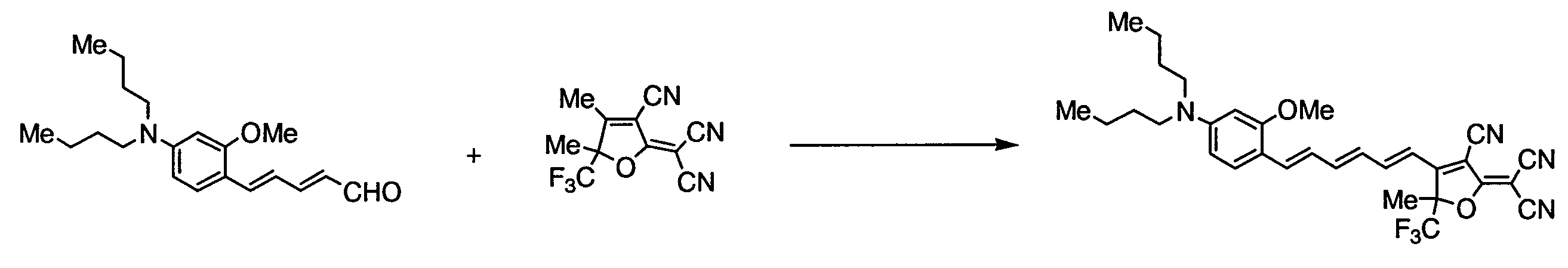
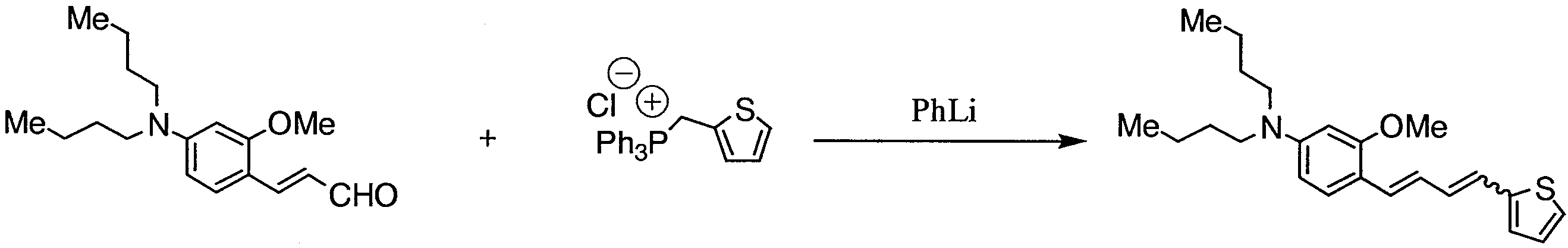
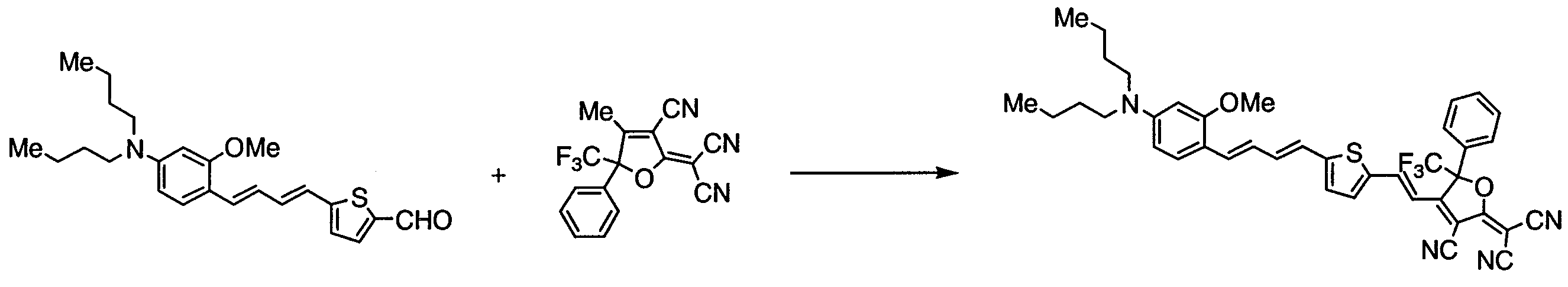
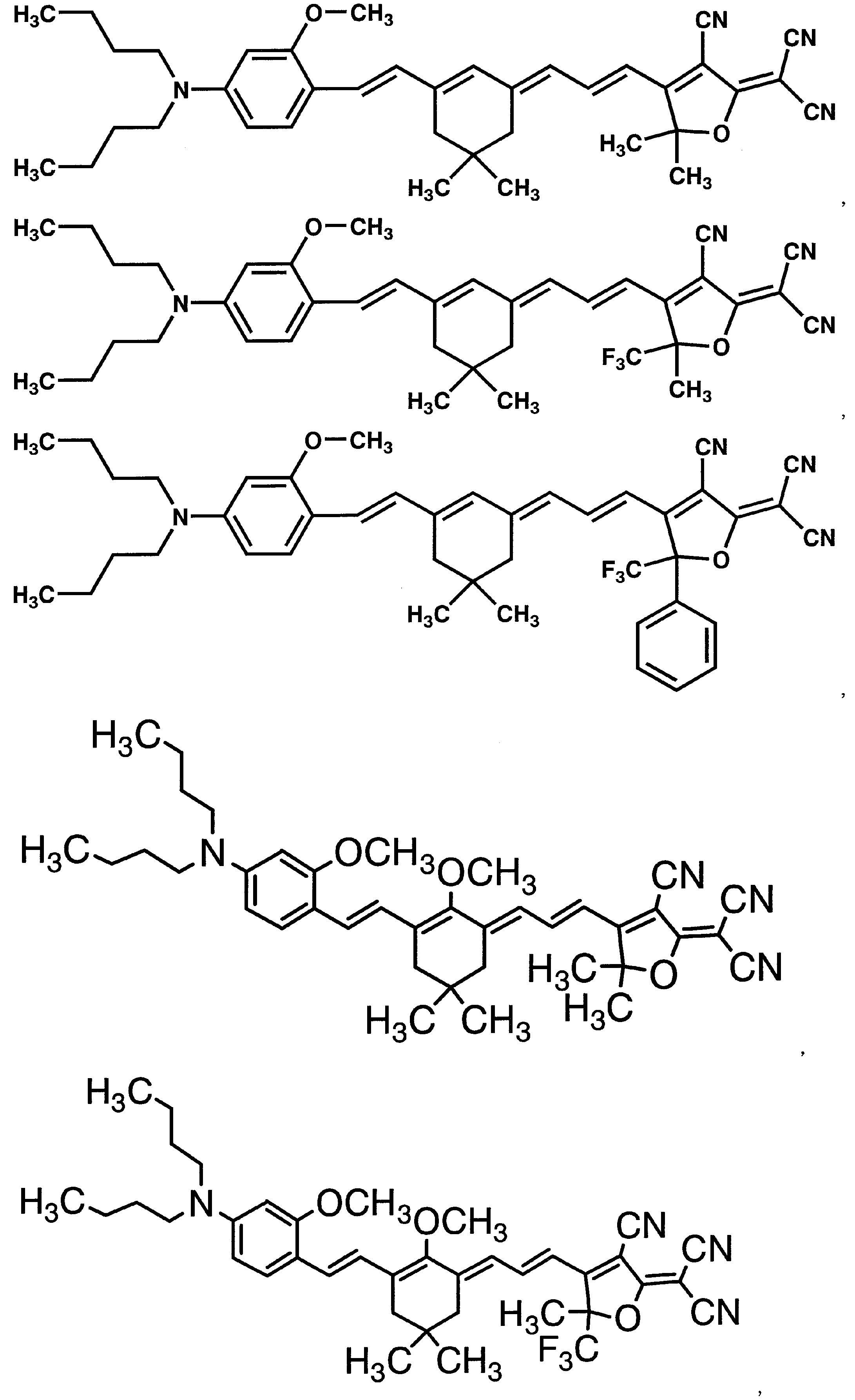
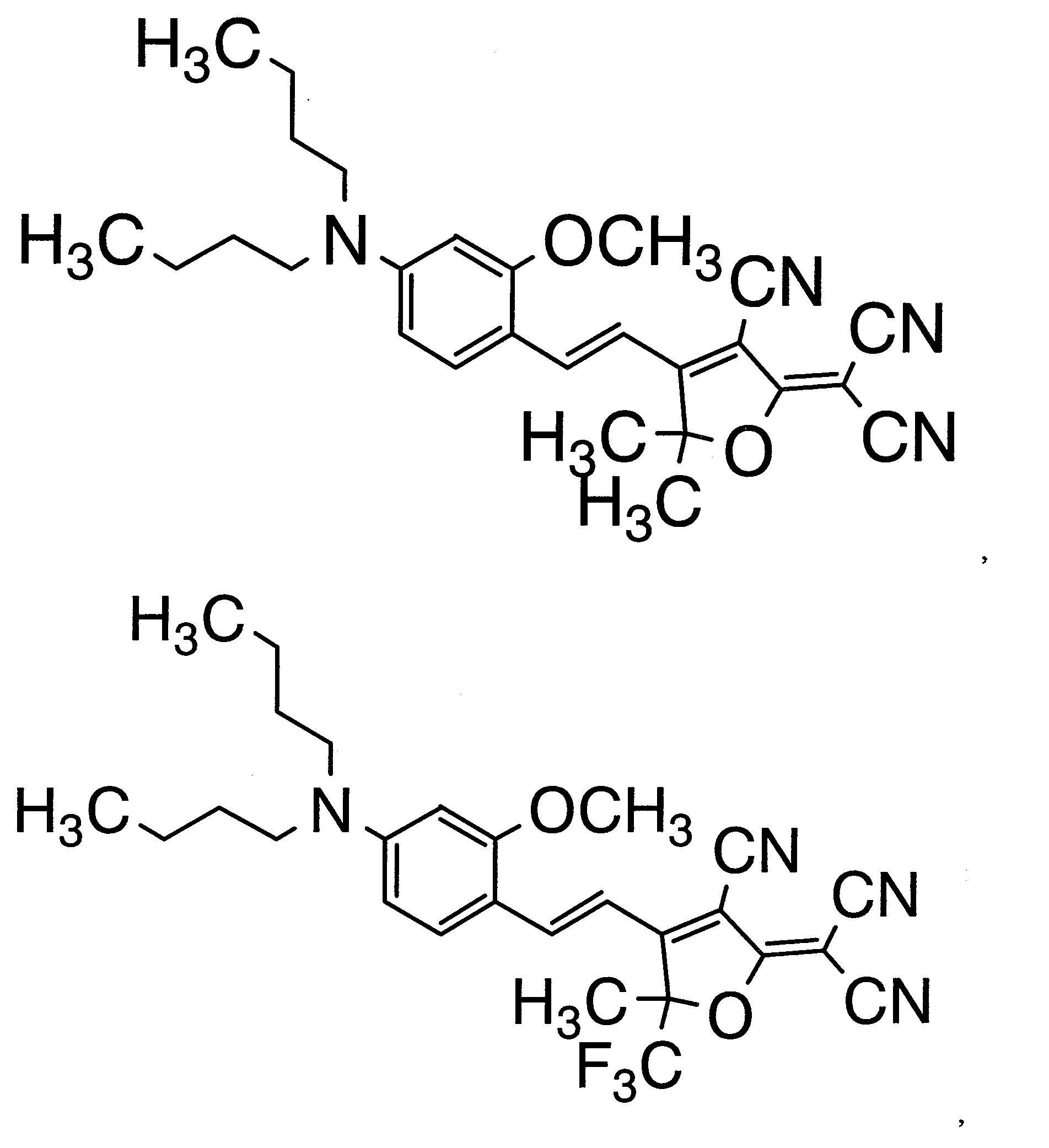
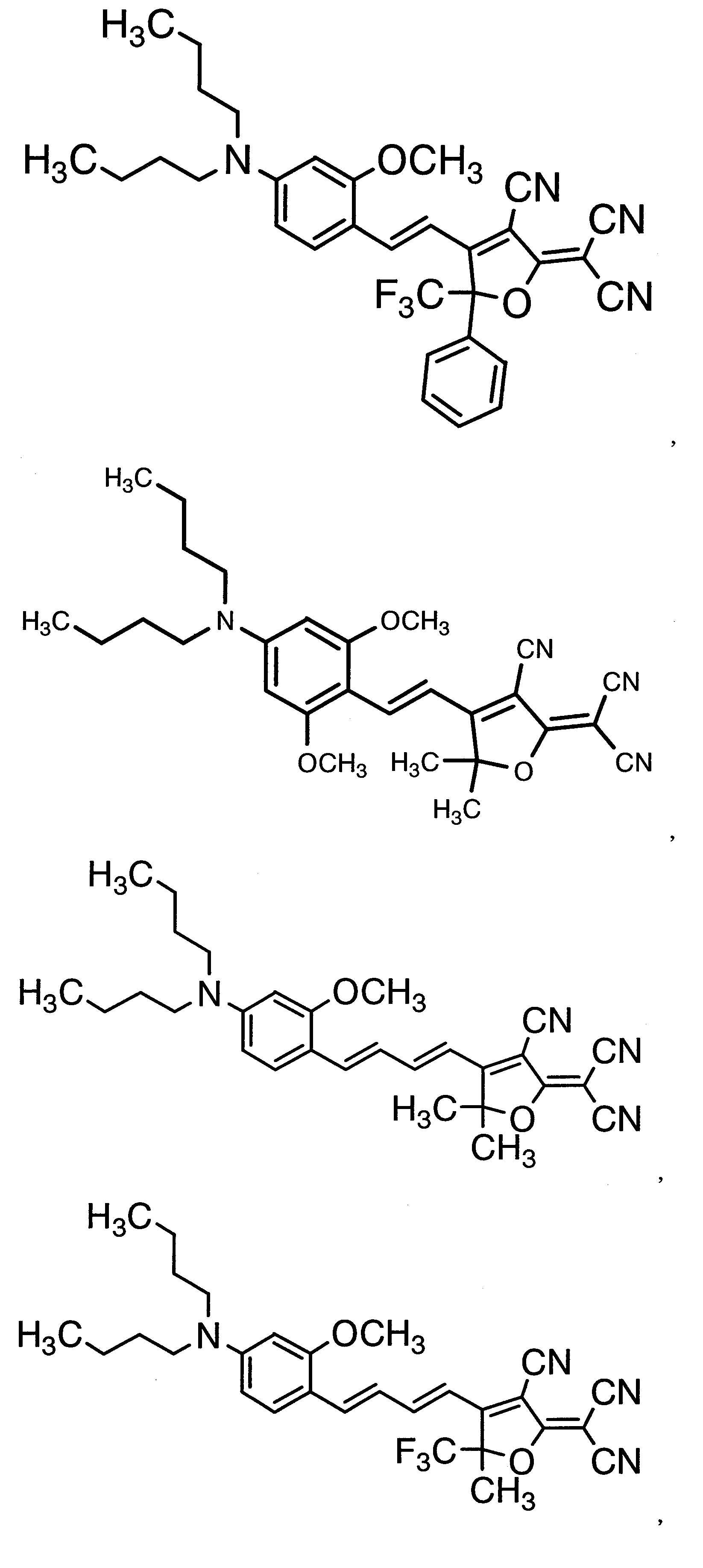
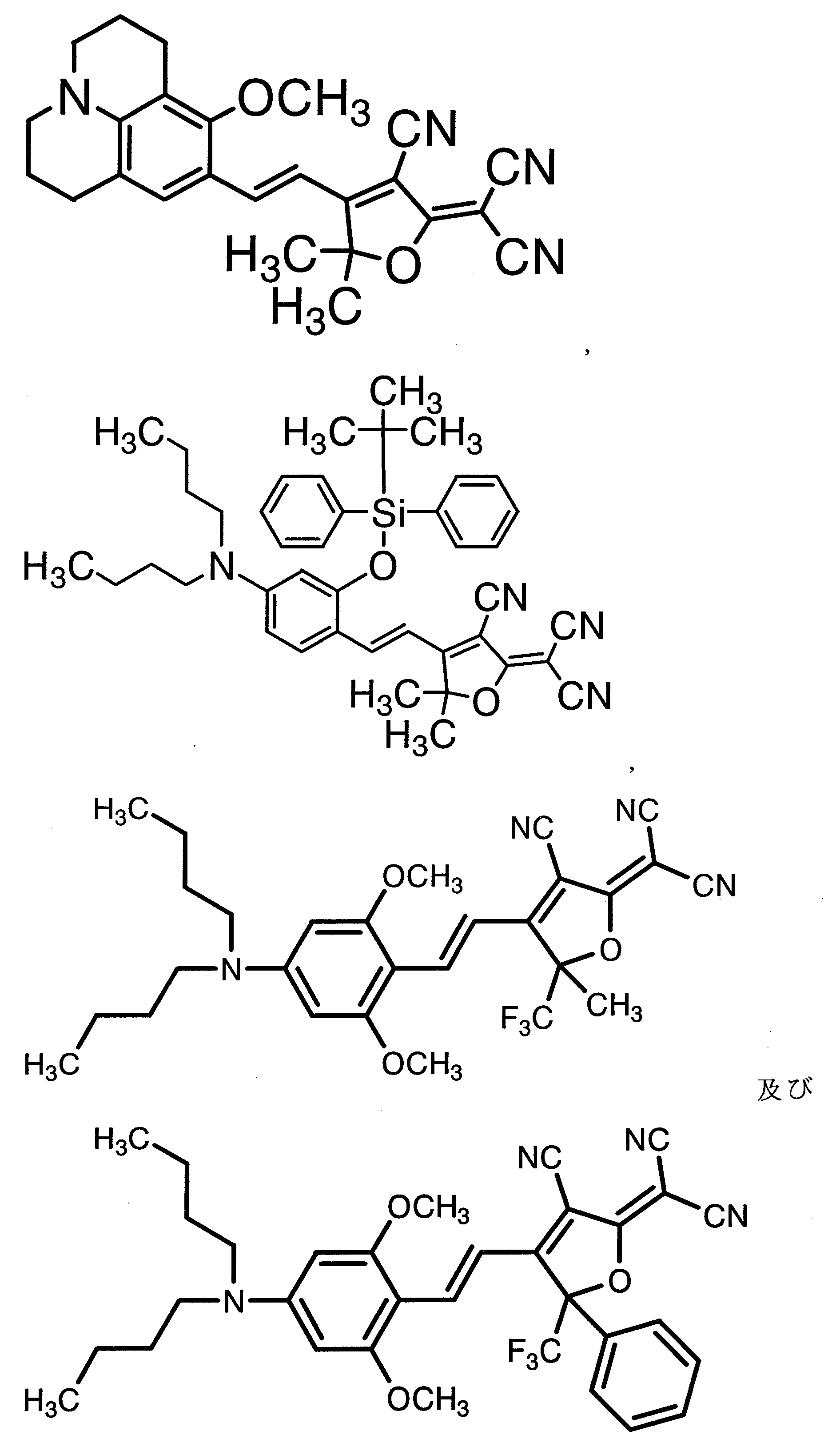
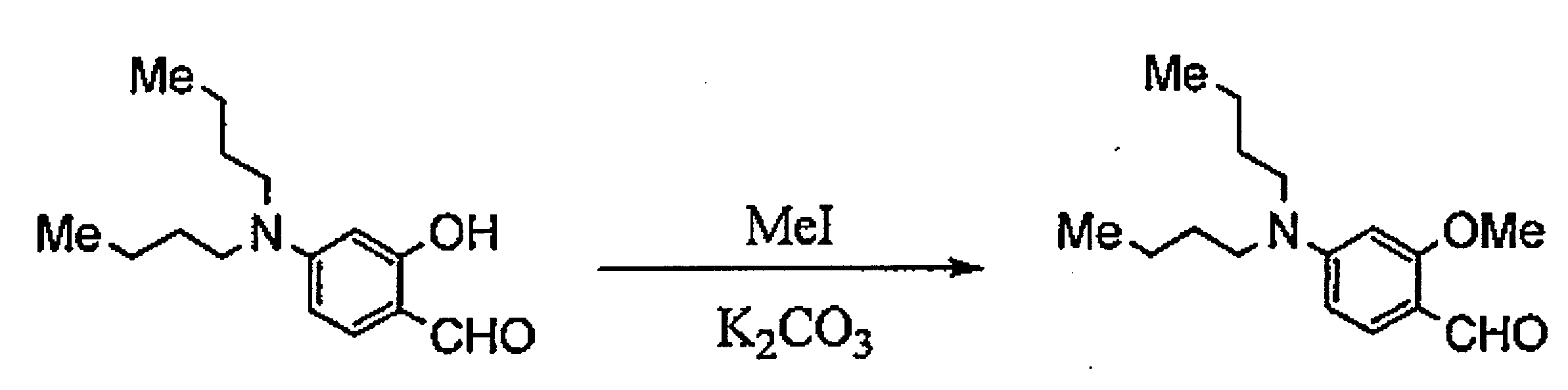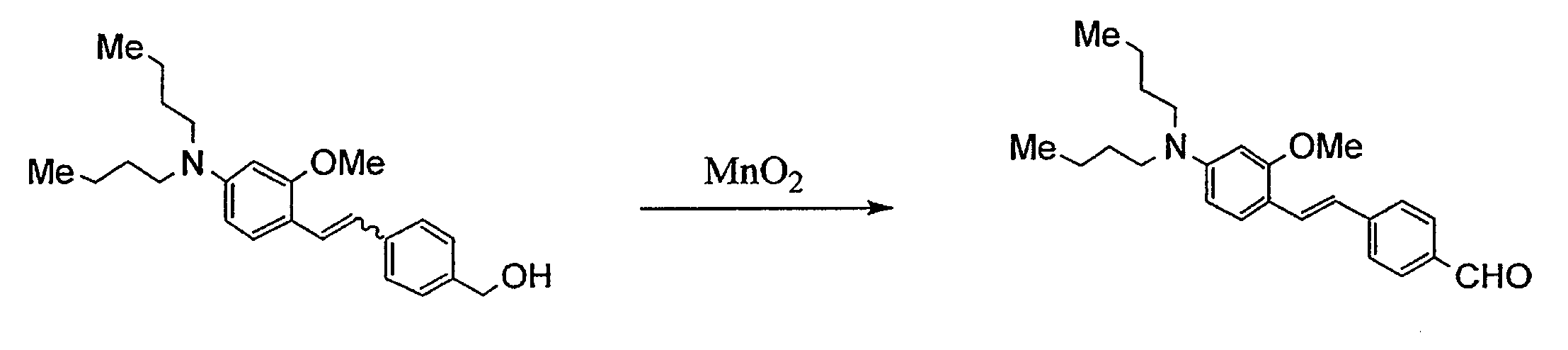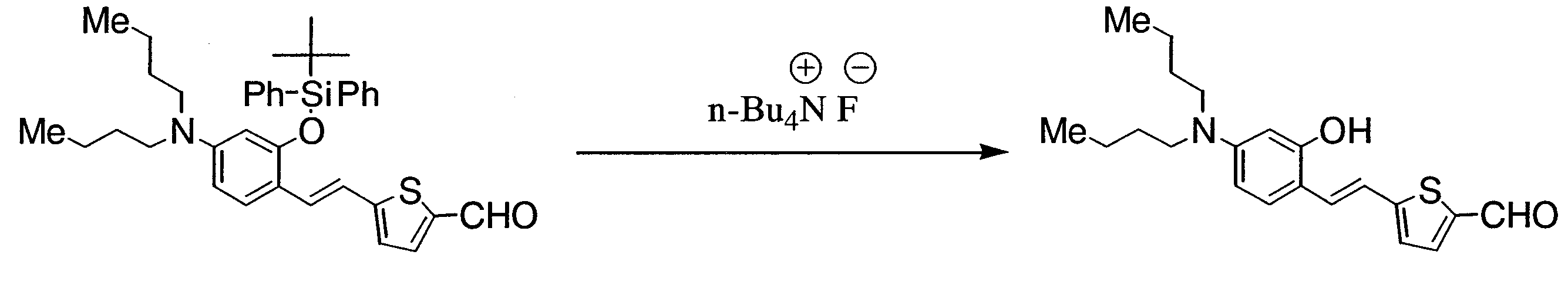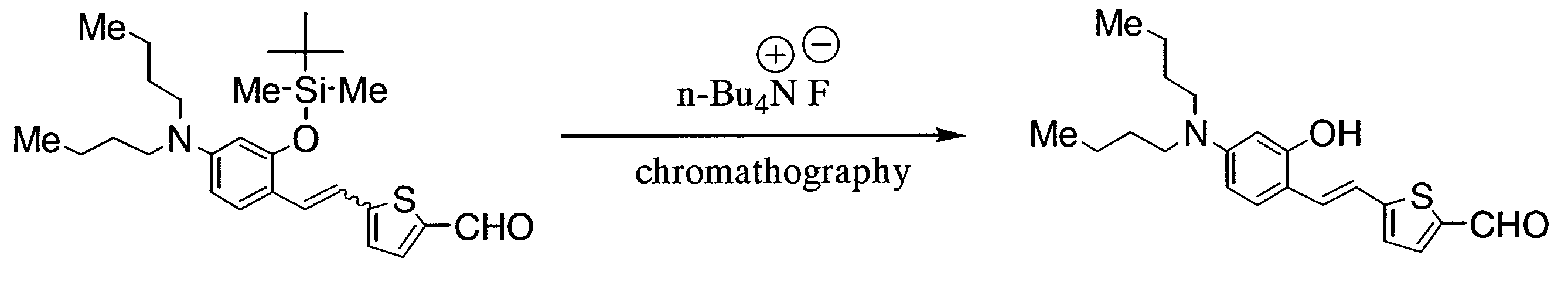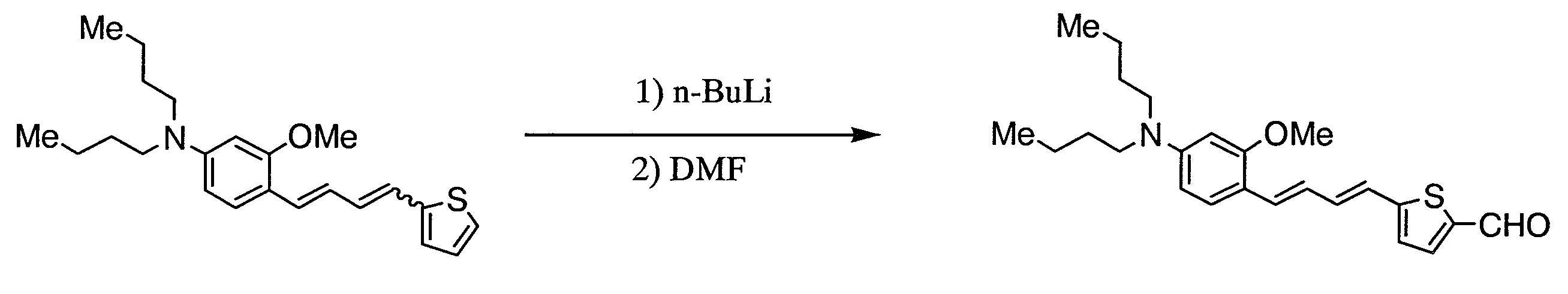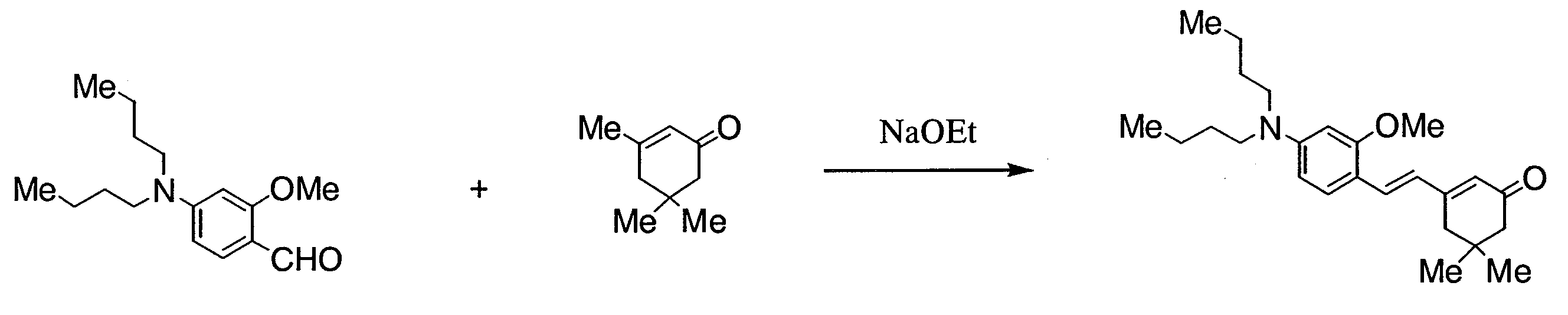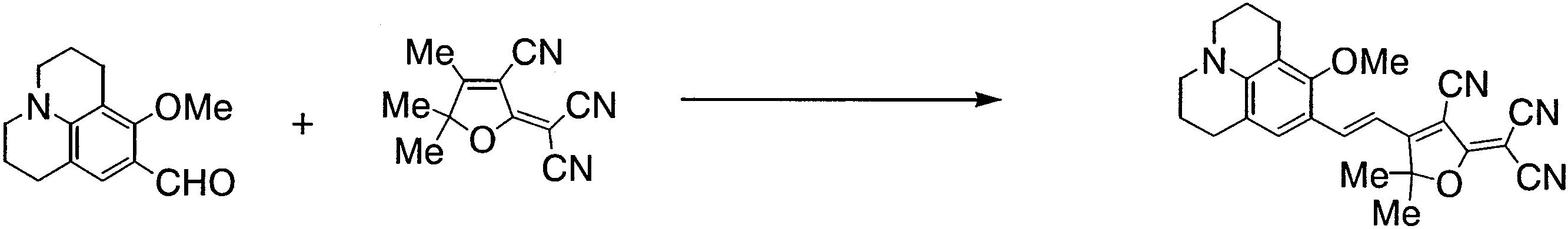WO2011024774A1 - 2次非線形光学化合物及びそれを含む非線形光学素子 - Google Patents
2次非線形光学化合物及びそれを含む非線形光学素子 Download PDFInfo
- Publication number
- WO2011024774A1 WO2011024774A1 PCT/JP2010/064216 JP2010064216W WO2011024774A1 WO 2011024774 A1 WO2011024774 A1 WO 2011024774A1 JP 2010064216 W JP2010064216 W JP 2010064216W WO 2011024774 A1 WO2011024774 A1 WO 2011024774A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituent
- aryl
- same
- atom
- Prior art date
Links
- 230000003287 optical effect Effects 0.000 title claims abstract description 153
- 150000001875 compounds Chemical class 0.000 title description 131
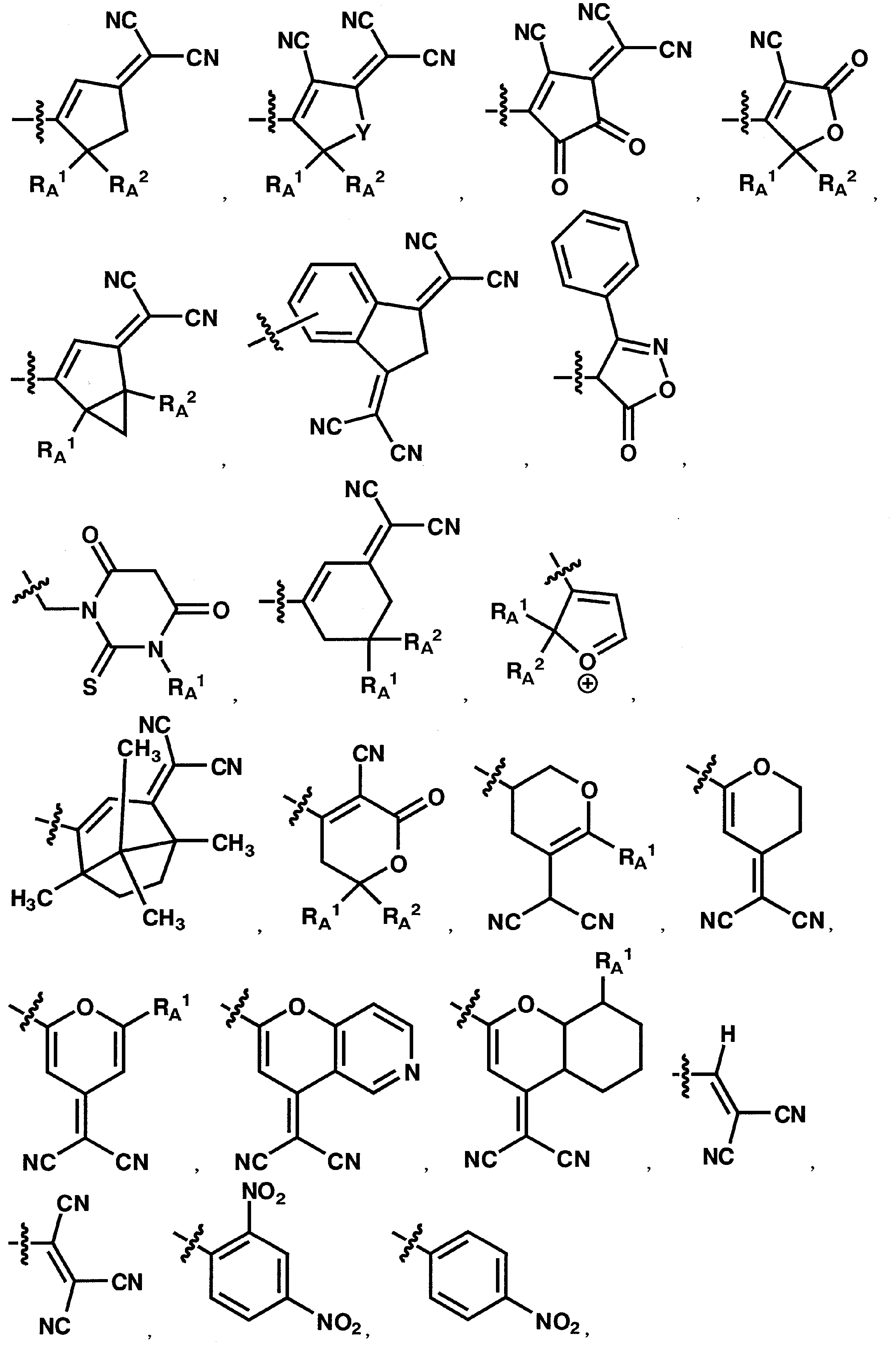
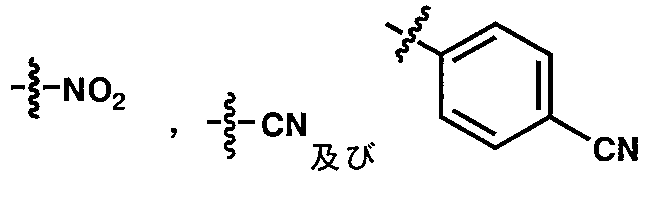
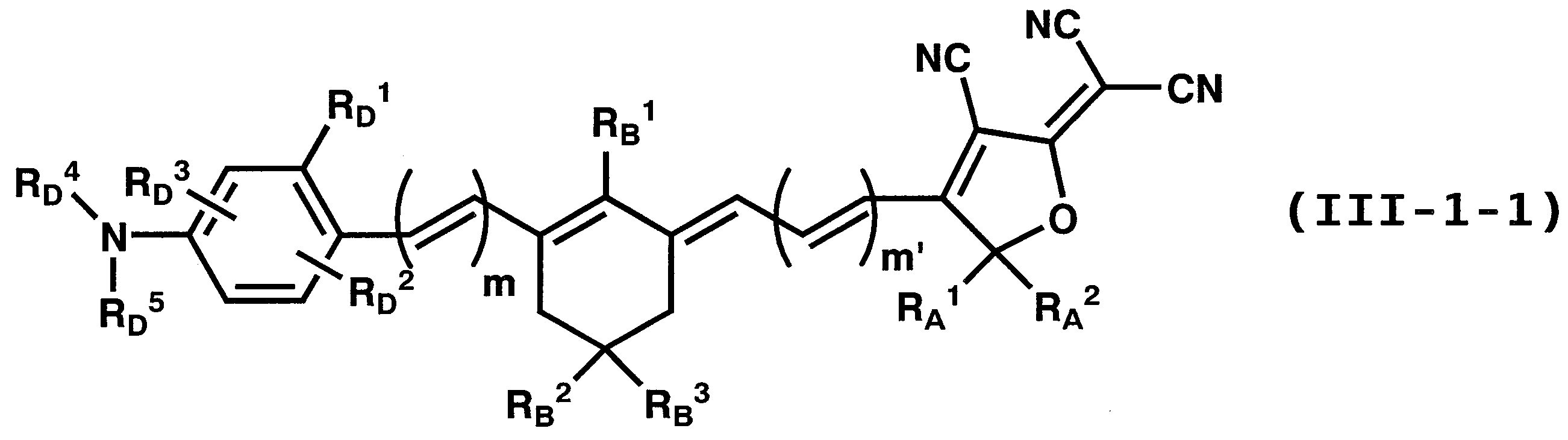
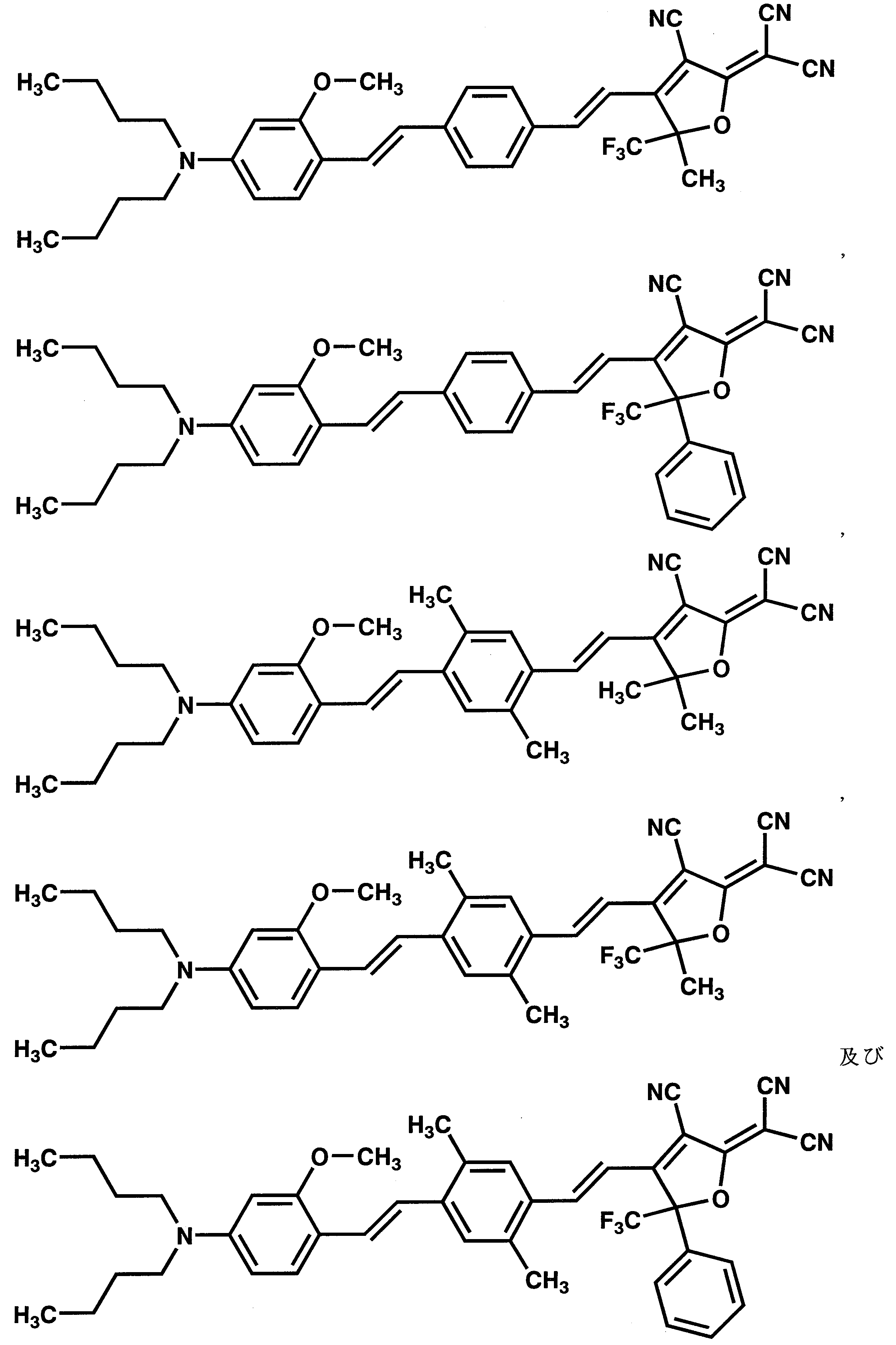
- 125000003118 aryl group Chemical group 0.000 claims abstract description 250
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 claims abstract description 49
- 125000001424 substituent group Chemical group 0.000 claims description 466
- 125000000217 alkyl group Chemical group 0.000 claims description 195
- 125000000623 heterocyclic group Chemical group 0.000 claims description 185
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 166
- 125000003545 alkoxy group Chemical group 0.000 claims description 150
- 125000002102 aryl alkyloxo group Chemical group 0.000 claims description 143
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 126
- 125000004104 aryloxy group Chemical group 0.000 claims description 118
- 125000005193 alkenylcarbonyloxy group Chemical group 0.000 claims description 110
- 125000005133 alkynyloxy group Chemical group 0.000 claims description 110
- 125000004469 siloxy group Chemical group [SiH3]O* 0.000 claims description 110
- 229910052757 nitrogen Inorganic materials 0.000 claims description 108
- 125000003302 alkenyloxy group Chemical group 0.000 claims description 107
- 125000001188 haloalkyl group Chemical group 0.000 claims description 107
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 106
- 125000004432 carbon atom Chemical group C* 0.000 claims description 94
- 229910052799 carbon Inorganic materials 0.000 claims description 92
- 150000001721 carbon Chemical group 0.000 claims description 89
- 125000005842 heteroatom Chemical group 0.000 claims description 89
- 239000000463 material Substances 0.000 claims description 55
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 49
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims description 40
- 125000005041 acyloxyalkyl group Chemical group 0.000 claims description 36
- 125000003342 alkenyl group Chemical group 0.000 claims description 36
- 125000004103 aminoalkyl group Chemical group 0.000 claims description 36
- CREMABGTGYGIQB-UHFFFAOYSA-N carbon carbon Chemical group C.C CREMABGTGYGIQB-UHFFFAOYSA-N 0.000 claims description 35
- 239000011203 carbon fibre reinforced carbon Substances 0.000 claims description 35
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 34
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 30
- 125000000392 cycloalkenyl group Chemical group 0.000 claims description 22
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 22
- 125000003277 amino group Chemical group 0.000 claims description 21
- 239000011347 resin Substances 0.000 claims description 13
- 229920005989 resin Polymers 0.000 claims description 13
- 125000000051 benzyloxy group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])O* 0.000 claims description 10
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 9
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 7
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 6
- 125000003320 C2-C6 alkenyloxy group Chemical group 0.000 claims description 6
- 125000000524 functional group Chemical group 0.000 claims description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 289
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 198
- -1 1,2-difluoroethyl group Chemical group 0.000 description 194
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 141
- 238000010898 silica gel chromatography Methods 0.000 description 126
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical group C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 111
- 238000005160 1H NMR spectroscopy Methods 0.000 description 110
- 238000003756 stirring Methods 0.000 description 109
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 100
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 99
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 92
- 239000013078 crystal Substances 0.000 description 90
- 239000000203 mixture Substances 0.000 description 87
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 75
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 75
- 229920002554 vinyl polymer Polymers 0.000 description 74
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 69
- 239000000243 solution Substances 0.000 description 63
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 60
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 59
- 239000000284 extract Substances 0.000 description 55
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 54
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 52
- CUONGYYJJVDODC-UHFFFAOYSA-N malononitrile Chemical compound N#CCC#N CUONGYYJJVDODC-UHFFFAOYSA-N 0.000 description 49
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 48
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 45
- 238000001816 cooling Methods 0.000 description 43
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 38
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 37
- 238000006243 chemical reaction Methods 0.000 description 36
- 239000002904 solvent Substances 0.000 description 35
- 238000000034 method Methods 0.000 description 31
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 description 29
- 239000003921 oil Substances 0.000 description 28
- 235000019198 oils Nutrition 0.000 description 28
- 229910052786 argon Inorganic materials 0.000 description 26
- 238000000746 purification Methods 0.000 description 26
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 24
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 22
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 21
- 239000007788 liquid Substances 0.000 description 20
- 239000011734 sodium Substances 0.000 description 20
- USFZMSVCRYTOJT-UHFFFAOYSA-N Ammonium acetate Chemical compound N.CC(O)=O USFZMSVCRYTOJT-UHFFFAOYSA-N 0.000 description 19
- 239000005695 Ammonium acetate Substances 0.000 description 19
- 235000019257 ammonium acetate Nutrition 0.000 description 19
- 229940043376 ammonium acetate Drugs 0.000 description 19
- 0 C*c1c(*)c(*)c(*)c(*)c1* Chemical compound C*c1c(*)c(*)c(*)c(*)c1* 0.000 description 18
- 239000011541 reaction mixture Substances 0.000 description 18
- 229930192474 thiophene Natural products 0.000 description 18
- 230000005684 electric field Effects 0.000 description 17
- 238000001914 filtration Methods 0.000 description 17
- 238000010438 heat treatment Methods 0.000 description 16
- 229910052739 hydrogen Inorganic materials 0.000 description 16
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 15
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 15
- 229910052740 iodine Inorganic materials 0.000 description 15
- 238000005481 NMR spectroscopy Methods 0.000 description 14
- 239000012044 organic layer Substances 0.000 description 14
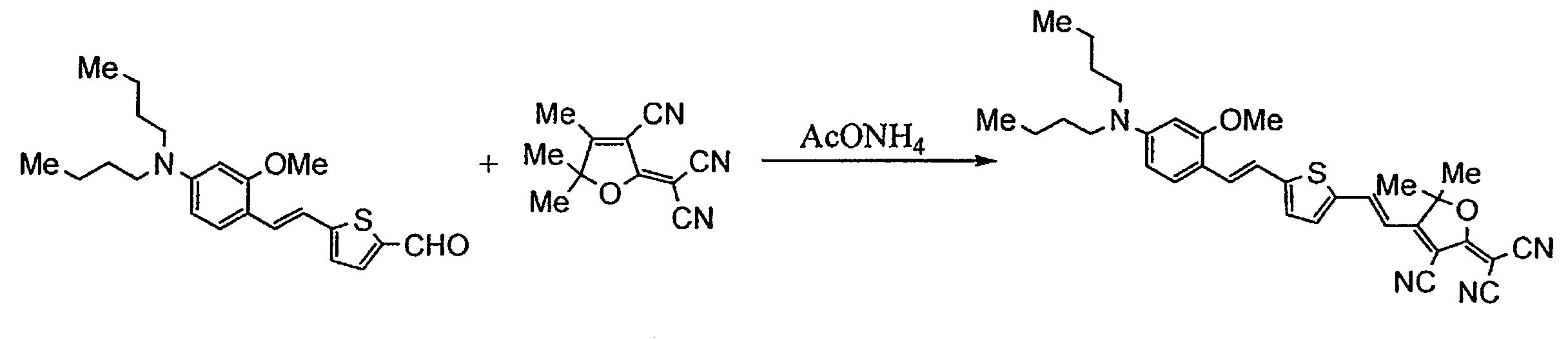
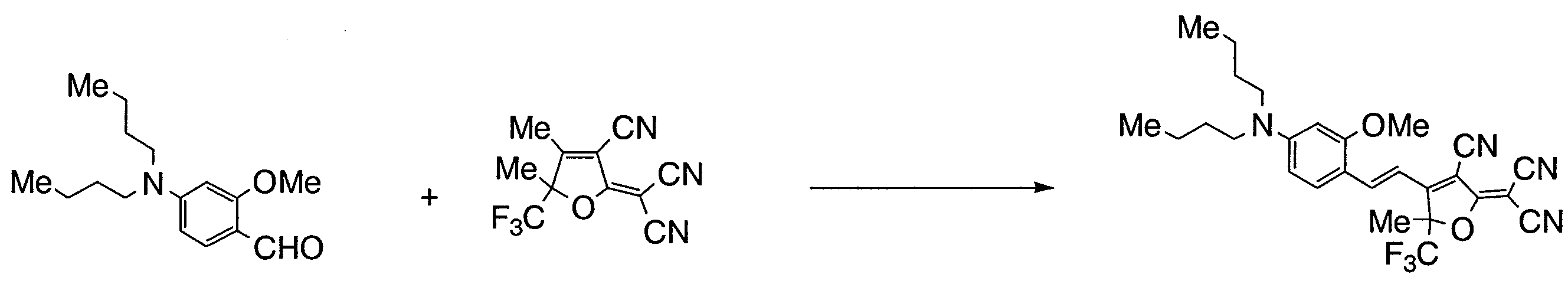
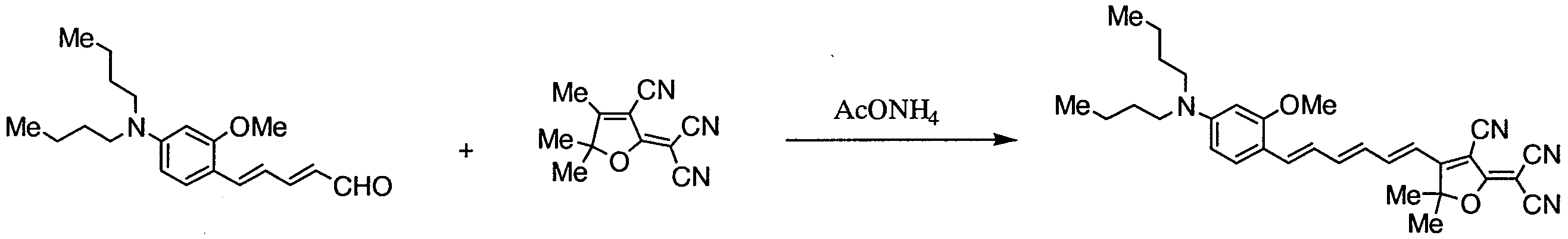
- FLMBQNOAWLPZPJ-UHFFFAOYSA-N 2-(3-cyano-4,5,5-trimethylfuran-2-ylidene)propanedinitrile Chemical compound CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C FLMBQNOAWLPZPJ-UHFFFAOYSA-N 0.000 description 13
- 125000002252 acyl group Chemical group 0.000 description 13
- 125000000304 alkynyl group Chemical group 0.000 description 13
- 238000005259 measurement Methods 0.000 description 13
- NHKJPPKXDNZFBJ-UHFFFAOYSA-N phenyllithium Chemical compound [Li]C1=CC=CC=C1 NHKJPPKXDNZFBJ-UHFFFAOYSA-N 0.000 description 13
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 12
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 12
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 12
- 239000011630 iodine Substances 0.000 description 12
- 238000002844 melting Methods 0.000 description 12
- 230000008018 melting Effects 0.000 description 12
- 239000010502 orange oil Substances 0.000 description 12
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 12
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 12
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 description 11
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 11
- 150000001412 amines Chemical class 0.000 description 11
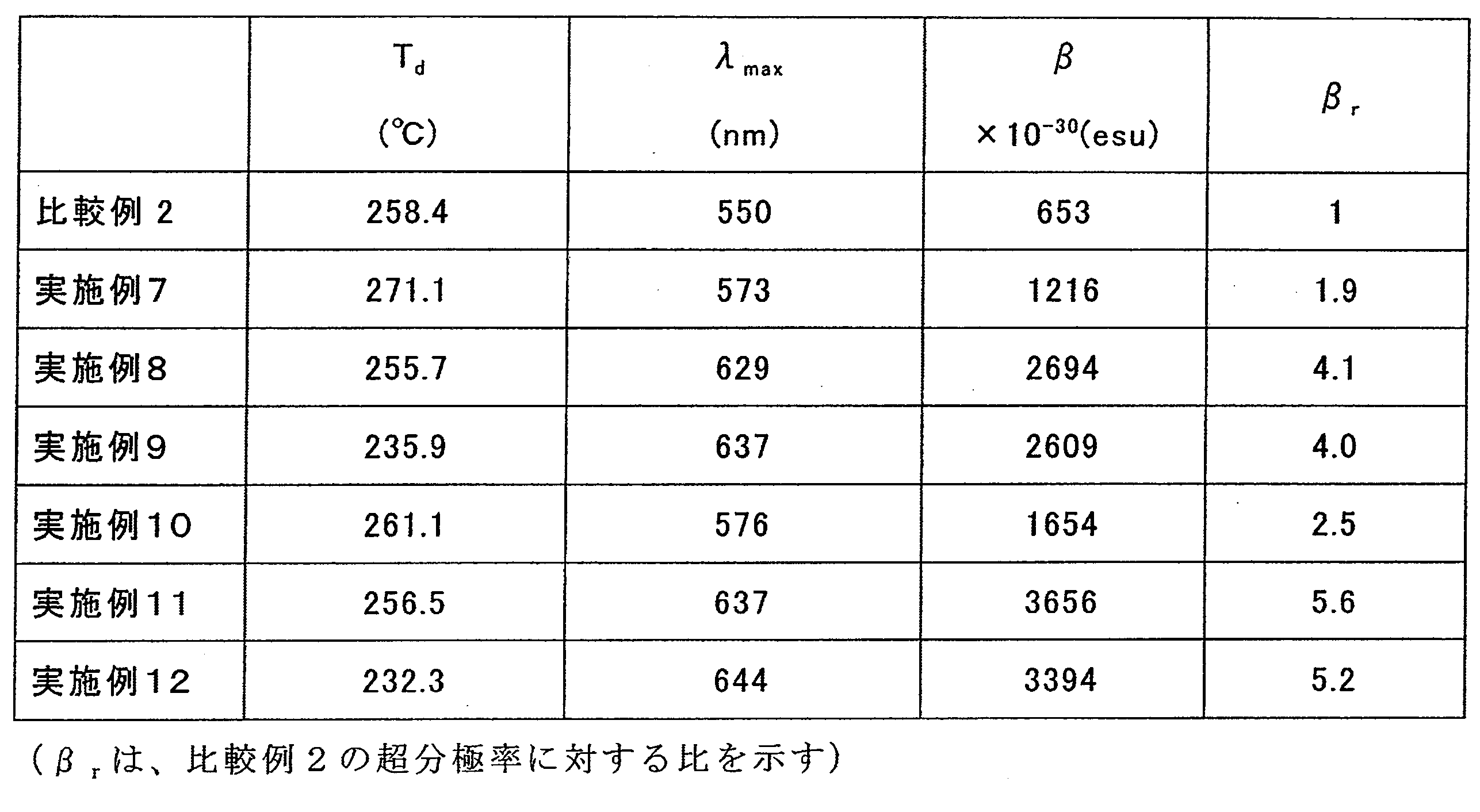
- 230000000052 comparative effect Effects 0.000 description 11
- 125000002950 monocyclic group Chemical group 0.000 description 11
- 125000006239 protecting group Chemical group 0.000 description 11
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 11
- 239000000126 substance Substances 0.000 description 11
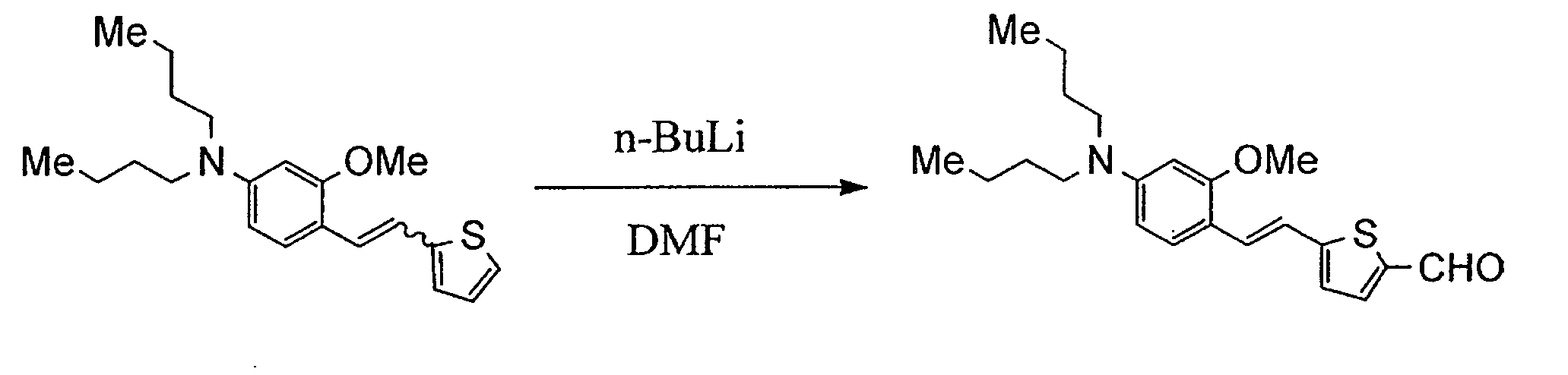
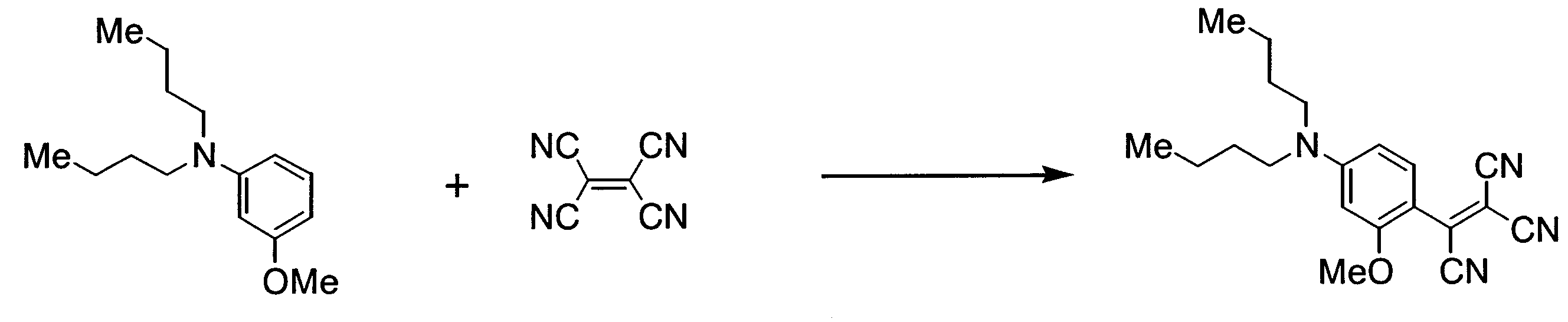
- UXJSJRDEGZSYRK-UHFFFAOYSA-N 4-(dibutylamino)-2-methoxybenzaldehyde Chemical compound CCCCN(CCCC)C1=CC=C(C=O)C(OC)=C1 UXJSJRDEGZSYRK-UHFFFAOYSA-N 0.000 description 10
- 238000004519 manufacturing process Methods 0.000 description 10
- 239000000047 product Substances 0.000 description 10
- HALXLBYMOUPGRG-UHFFFAOYSA-N 2-[3-cyano-4-methyl-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 HALXLBYMOUPGRG-UHFFFAOYSA-N 0.000 description 9
- 235000019502 Orange oil Nutrition 0.000 description 9
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 9
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 125000005843 halogen group Chemical group 0.000 description 9
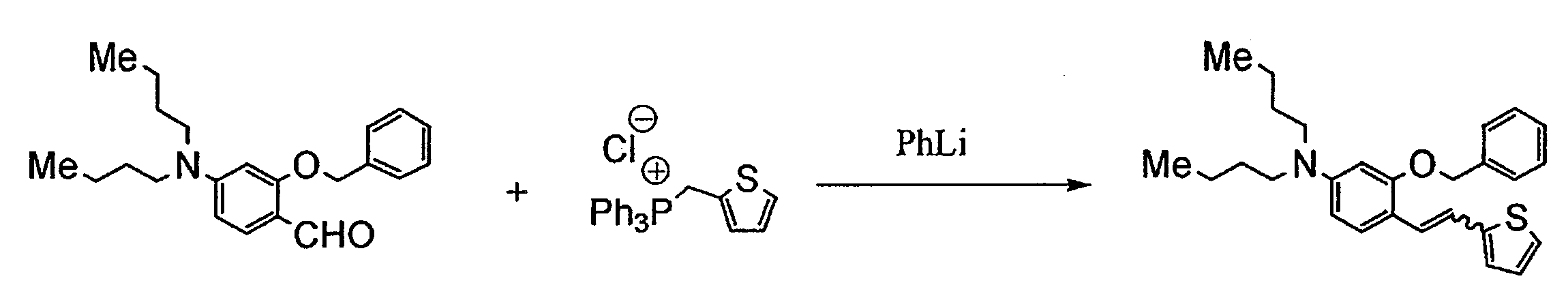
- NPMHPUVPHLQJSP-UHFFFAOYSA-M triphenyl(thiophen-2-ylmethyl)phosphanium;chloride Chemical compound [Cl-].C=1C=CC=CC=1[P+](C=1C=CC=CC=1)(C=1C=CC=CC=1)CC1=CC=CS1 NPMHPUVPHLQJSP-UHFFFAOYSA-M 0.000 description 9
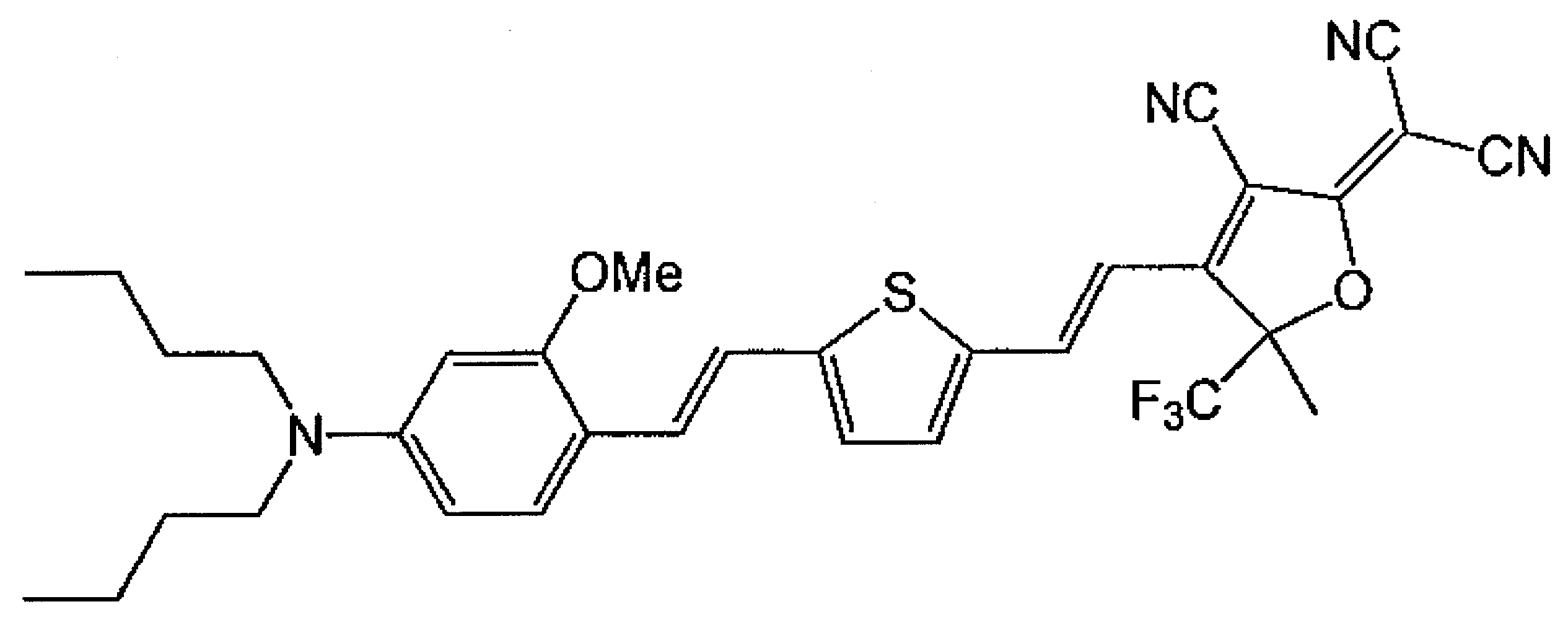
- USENAQUOBGJVHI-UHFFFAOYSA-N 2-[3-cyano-4,5-dimethyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C(F)(F)F USENAQUOBGJVHI-UHFFFAOYSA-N 0.000 description 8
- 239000012043 crude product Substances 0.000 description 8
- 239000005457 ice water Substances 0.000 description 8
- 239000010410 layer Substances 0.000 description 8
- 238000005979 thermal decomposition reaction Methods 0.000 description 8
- 238000005406 washing Methods 0.000 description 8
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 description 7
- 125000001931 aliphatic group Chemical group 0.000 description 7
- 125000005090 alkenylcarbonyl group Chemical group 0.000 description 7
- 230000000694 effects Effects 0.000 description 7
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 7
- 125000001072 heteroaryl group Chemical group 0.000 description 7
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 7
- 239000002798 polar solvent Substances 0.000 description 7
- 239000000843 powder Substances 0.000 description 7
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 6
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 6
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 6
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- 235000019270 ammonium chloride Nutrition 0.000 description 6
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 6
- SIPUZPBQZHNSDW-UHFFFAOYSA-N bis(2-methylpropyl)aluminum Chemical compound CC(C)C[Al]CC(C)C SIPUZPBQZHNSDW-UHFFFAOYSA-N 0.000 description 6
- 238000005253 cladding Methods 0.000 description 6
- 239000012230 colorless oil Substances 0.000 description 6
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 6
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 6
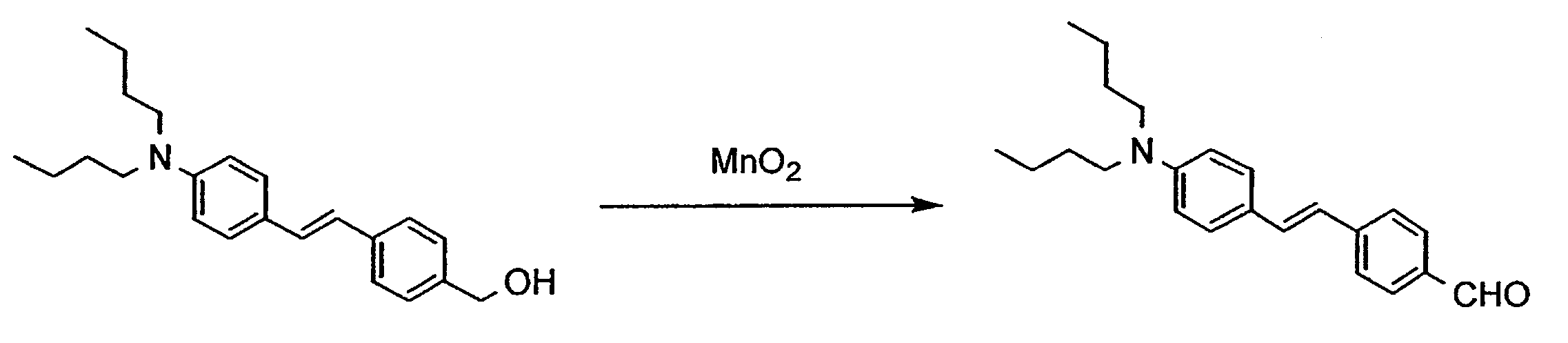
- NUJOXMJBOLGQSY-UHFFFAOYSA-N manganese dioxide Chemical compound O=[Mn]=O NUJOXMJBOLGQSY-UHFFFAOYSA-N 0.000 description 6
- 229910000027 potassium carbonate Inorganic materials 0.000 description 6
- 239000013074 reference sample Substances 0.000 description 6
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 6
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 5
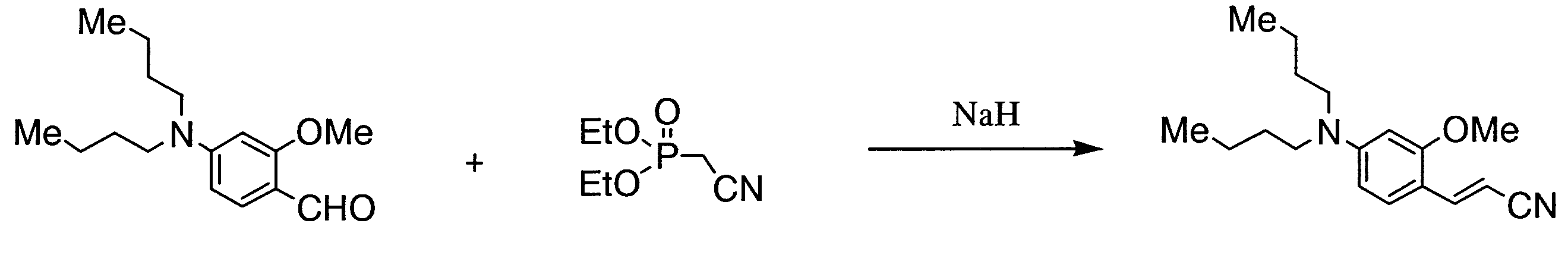
- KWMBADTWRIGGGG-UHFFFAOYSA-N 2-diethoxyphosphorylacetonitrile Chemical compound CCOP(=O)(CC#N)OCC KWMBADTWRIGGGG-UHFFFAOYSA-N 0.000 description 5
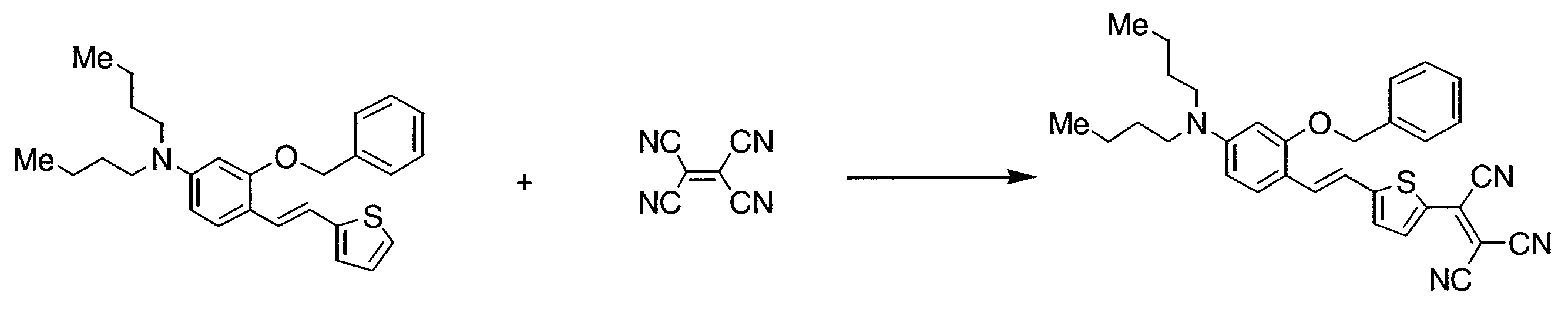
- BHBASVVNOGPRFQ-UHFFFAOYSA-N 5-[2-[2-[tert-butyl(diphenyl)silyl]oxy-4-(dibutylamino)phenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound C=1C=CC=CC=1[Si](C(C)(C)C)(C=1C=CC=CC=1)OC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 BHBASVVNOGPRFQ-UHFFFAOYSA-N 0.000 description 5
- 125000003341 7 membered heterocyclic group Chemical group 0.000 description 5
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- 239000012792 core layer Substances 0.000 description 5
- 238000001035 drying Methods 0.000 description 5
- 125000003253 isopropoxy group Chemical group [H]C([H])([H])C([H])(O*)C([H])([H])[H] 0.000 description 5
- 125000001624 naphthyl group Chemical group 0.000 description 5
- 239000000523 sample Substances 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 239000012312 sodium hydride Substances 0.000 description 5
- 229910000104 sodium hydride Inorganic materials 0.000 description 5
- 229940079827 sodium hydrogen sulfite Drugs 0.000 description 5
- PADLLLYWNNPBLV-UHFFFAOYSA-N 4-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-2,5-dimethylbenzaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC(C)=C(C=O)C=C1C PADLLLYWNNPBLV-UHFFFAOYSA-N 0.000 description 4
- ZWHGEVRFCWCEHP-UHFFFAOYSA-N 5-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 ZWHGEVRFCWCEHP-UHFFFAOYSA-N 0.000 description 4
- RZCPWYFMQWWAHL-UHFFFAOYSA-N 5-[2-[4-(dibutylamino)-2-phenylmethoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound C=1C=CC=CC=1COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 RZCPWYFMQWWAHL-UHFFFAOYSA-N 0.000 description 4
- XBAYGNRBGMPLJQ-UHFFFAOYSA-N 5-[2-[4-[butyl(4-hydroxybutyl)amino]-2-phenylmethoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound C=1C=CC=CC=1COC1=CC(N(CCCCO)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 XBAYGNRBGMPLJQ-UHFFFAOYSA-N 0.000 description 4
- BQPDCHGGVRLVKC-UHFFFAOYSA-N 5-[4-[4-(dibutylamino)-2-methoxyphenyl]buta-1,3-dienyl]thiophene-2-carbaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC=CC1=CC=C(C=O)S1 BQPDCHGGVRLVKC-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 4
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 4
- CSCPPACGZOOCGX-WFGJKAKNSA-N acetone d6 Chemical compound [2H]C([2H])([2H])C(=O)C([2H])([2H])[2H] CSCPPACGZOOCGX-WFGJKAKNSA-N 0.000 description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 4
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 4
- 235000011089 carbon dioxide Nutrition 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 4
- 238000010511 deprotection reaction Methods 0.000 description 4
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 4
- 239000010408 film Substances 0.000 description 4
- WVDDGKGOMKODPV-UHFFFAOYSA-N hydroxymethyl benzene Natural products OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 4
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 4
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 4
- 239000012046 mixed solvent Substances 0.000 description 4
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 4
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 4
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 4
- AOJFQRQNPXYVLM-UHFFFAOYSA-N pyridin-1-ium;chloride Chemical compound [Cl-].C1=CC=[NH+]C=C1 AOJFQRQNPXYVLM-UHFFFAOYSA-N 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 4
- 230000008054 signal transmission Effects 0.000 description 4
- 125000005017 substituted alkenyl group Chemical group 0.000 description 4
- 125000000547 substituted alkyl group Chemical group 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 4
- PLWLVSUJPBYXMS-UHFFFAOYSA-N 2-[(3-methoxyphenyl)methylamino]ethyl 2,2-dimethylpropanoate Chemical compound COC1=CC=CC(CNCCOC(=O)C(C)(C)C)=C1 PLWLVSUJPBYXMS-UHFFFAOYSA-N 0.000 description 3
- HUFLNPIRBGHPSX-UHFFFAOYSA-N 2-[(3-phenylmethoxyphenyl)methylamino]ethyl 2,2-dimethylpropanoate Chemical compound CC(C)(C)C(=O)OCCNCC1=CC=CC(OCC=2C=CC=CC=2)=C1 HUFLNPIRBGHPSX-UHFFFAOYSA-N 0.000 description 3
- CQHNGCXPNGOTFY-UHFFFAOYSA-N 2-[2-(2,4,6-trimethoxyphenyl)ethenyl]thiophene Chemical compound COC1=CC(OC)=CC(OC)=C1C=CC1=CC=CS1 CQHNGCXPNGOTFY-UHFFFAOYSA-N 0.000 description 3
- KBUHYIYWDCQODE-UHFFFAOYSA-N 2-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]acetaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC(=CC=O)CC(C)(C)C1 KBUHYIYWDCQODE-UHFFFAOYSA-N 0.000 description 3
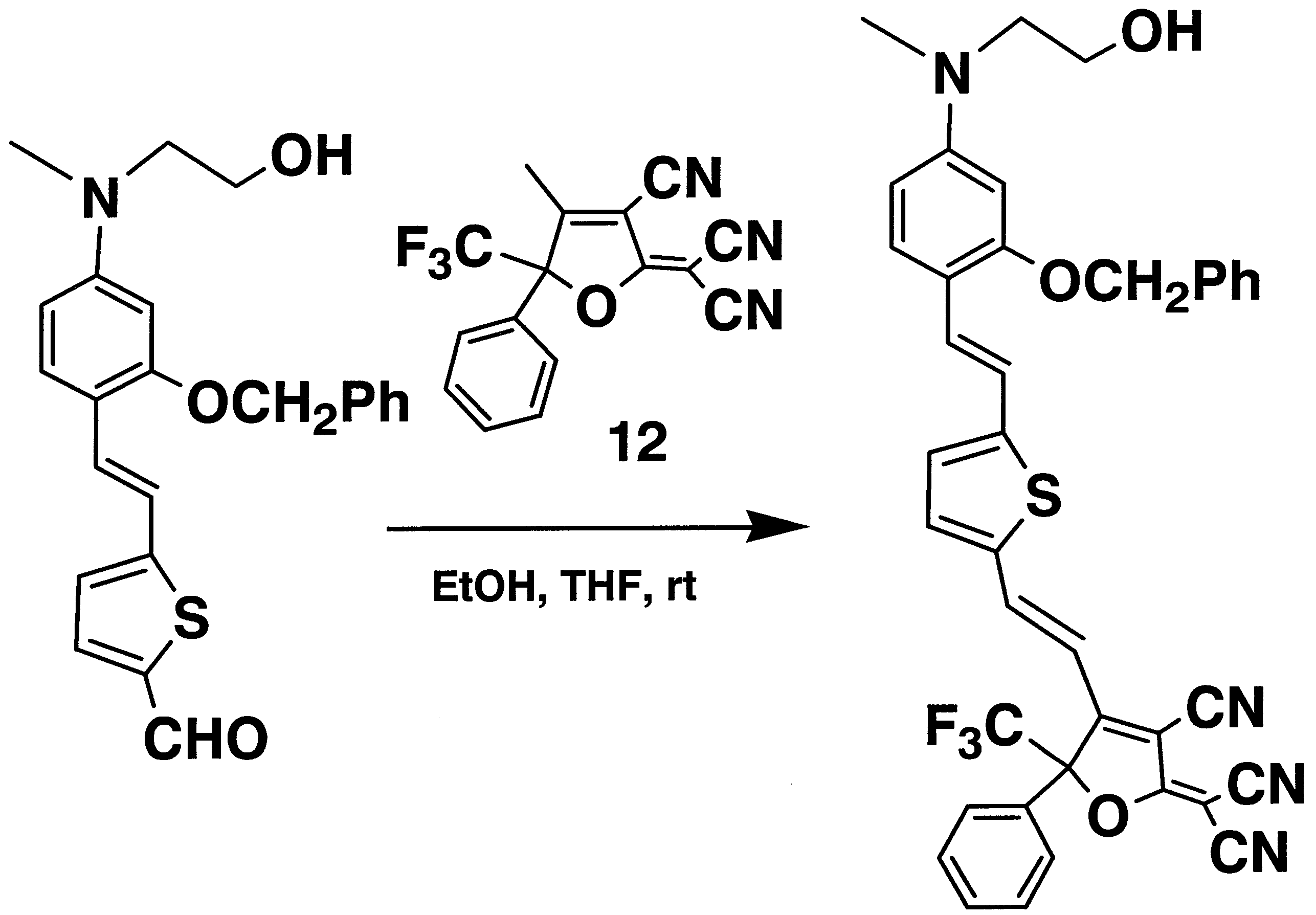
- UDPJEMQBVKVGKY-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[2-[4-[2-hydroxyethyl(methyl)amino]-2-phenylmethoxyphenyl]ethenyl]thiophen-2-yl]ethenyl]-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound C=1C=CC=CC=1COC1=CC(N(CCO)C)=CC=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 UDPJEMQBVKVGKY-UHFFFAOYSA-N 0.000 description 3
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 3
- 125000004777 2-fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 3
- SYXACGHAQSPPFI-UHFFFAOYSA-N 3-[4-(dibutylamino)-2-methoxyphenyl]prop-2-enal Chemical compound CCCCN(CCCC)C1=CC=C(C=CC=O)C(OC)=C1 SYXACGHAQSPPFI-UHFFFAOYSA-N 0.000 description 3
- XDTSUOLJKOWVTL-UHFFFAOYSA-N 4-(dibutylamino)-2,6-dimethoxybenzaldehyde Chemical compound CCCCN(CCCC)C1=CC(OC)=C(C=O)C(OC)=C1 XDTSUOLJKOWVTL-UHFFFAOYSA-N 0.000 description 3
- JPEVCYKADKXHKK-UHFFFAOYSA-N 4-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]benzaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)C=C1 JPEVCYKADKXHKK-UHFFFAOYSA-N 0.000 description 3
- OMUARTWUDVRUGO-UHFFFAOYSA-N 5-[2-(2,4,6-trimethoxyphenyl)ethenyl]thiophene-2-carbaldehyde Chemical compound COC1=CC(OC)=CC(OC)=C1C=CC1=CC=C(C=O)S1 OMUARTWUDVRUGO-UHFFFAOYSA-N 0.000 description 3
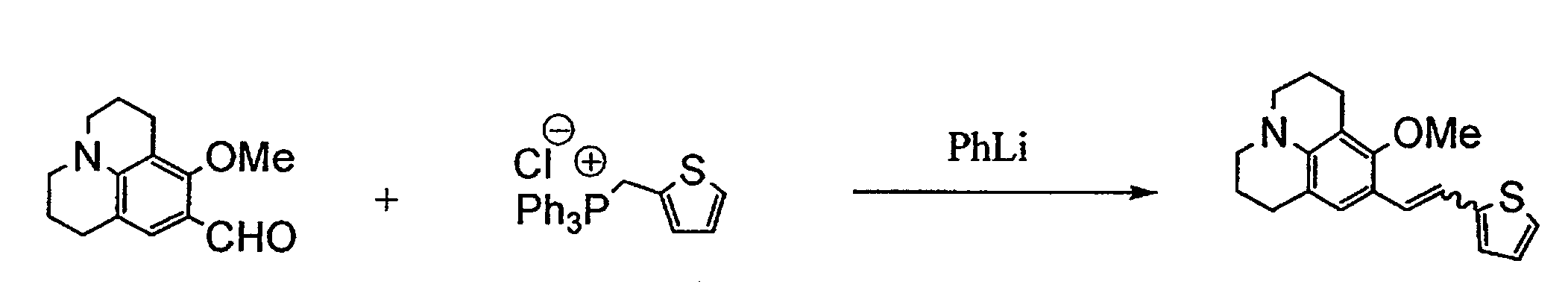
- BXTMBNXOCBKKEZ-UHFFFAOYSA-N 5-[2-(8-methoxy-2,3,6,7-tetrahydro-1h,5h-benzo[ij]quinolizine-9-yl)vinyl]thiophene-2-carboaldehyde Chemical compound C1=C(C=23)CCCN3CCCC=2C(OC)=C1C=CC1=CC=C(C=O)S1 BXTMBNXOCBKKEZ-UHFFFAOYSA-N 0.000 description 3
- ZUWAVFGEFDDRIO-UHFFFAOYSA-N 5-[2-[4-(dibutylamino)-2,6-dimethoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC(OC)=C1C=CC1=CC=C(C=O)S1 ZUWAVFGEFDDRIO-UHFFFAOYSA-N 0.000 description 3
- RHWUCWXRQVXORZ-UHFFFAOYSA-N 5-[2-[4-(dibutylamino)-2-hydroxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound OC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 RHWUCWXRQVXORZ-UHFFFAOYSA-N 0.000 description 3
- CWWSMJCKFQBUCO-UHFFFAOYSA-N 5-[2-[4-[2-hydroxyethyl(methyl)amino]-2-methoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound COC1=CC(N(C)CCO)=CC=C1C=CC1=CC=C(C=O)S1 CWWSMJCKFQBUCO-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 3
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 3
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 3
- 239000012300 argon atmosphere Substances 0.000 description 3
- 239000012298 atmosphere Substances 0.000 description 3
- 125000005997 bromomethyl group Chemical group 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 3
- 230000008878 coupling Effects 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- 238000005859 coupling reaction Methods 0.000 description 3
- 125000004093 cyano group Chemical group *C#N 0.000 description 3
- 230000018044 dehydration Effects 0.000 description 3
- 238000006297 dehydration reaction Methods 0.000 description 3
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 3
- 230000001747 exhibiting effect Effects 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 3
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 125000002510 isobutoxy group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])O* 0.000 description 3
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 3
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 3
- IUPSYLYTLDHOSP-UHFFFAOYSA-N n,n-dibutyl-3,5-dimethoxy-4-(2-thiophen-2-ylethenyl)aniline Chemical compound COC1=CC(N(CCCC)CCCC)=CC(OC)=C1C=CC1=CC=CS1 IUPSYLYTLDHOSP-UHFFFAOYSA-N 0.000 description 3
- BHRRIXUIPSCYJA-UHFFFAOYSA-N n,n-dibutyl-3-methoxyaniline Chemical compound CCCCN(CCCC)C1=CC=CC(OC)=C1 BHRRIXUIPSCYJA-UHFFFAOYSA-N 0.000 description 3
- UGQXBZFTOQBDKH-UHFFFAOYSA-N n,n-dibutyl-3-phenylmethoxy-4-(2-thiophen-2-ylethenyl)aniline Chemical compound C=1C=CC=CC=1COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=CS1 UGQXBZFTOQBDKH-UHFFFAOYSA-N 0.000 description 3
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 3
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 3
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 230000035484 reaction time Effects 0.000 description 3
- 125000005920 sec-butoxy group Chemical group 0.000 description 3
- 239000001632 sodium acetate Substances 0.000 description 3
- 235000017281 sodium acetate Nutrition 0.000 description 3
- 235000017557 sodium bicarbonate Nutrition 0.000 description 3
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 3
- 125000005415 substituted alkoxy group Chemical group 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 125000004213 tert-butoxy group Chemical group [H]C([H])([H])C(O*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 3
- GRRIMVWABNHKBX-UHFFFAOYSA-N (3-methoxyphenyl)methanamine Chemical compound COC1=CC=CC(CN)=C1 GRRIMVWABNHKBX-UHFFFAOYSA-N 0.000 description 2
- WAMBEHWAKBRTGT-UHFFFAOYSA-N (3-phenylmethoxyphenyl)methanamine Chemical compound NCC1=CC=CC(OCC=2C=CC=CC=2)=C1 WAMBEHWAKBRTGT-UHFFFAOYSA-N 0.000 description 2
- FEWLNYSYJNLUOO-UHFFFAOYSA-N 1-Piperidinecarboxaldehyde Chemical compound O=CN1CCCCC1 FEWLNYSYJNLUOO-UHFFFAOYSA-N 0.000 description 2
- 125000004343 1-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C([H])([H])[H] 0.000 description 2
- JVSFQJZRHXAUGT-UHFFFAOYSA-N 2,2-dimethylpropanoyl chloride Chemical compound CC(C)(C)C(Cl)=O JVSFQJZRHXAUGT-UHFFFAOYSA-N 0.000 description 2
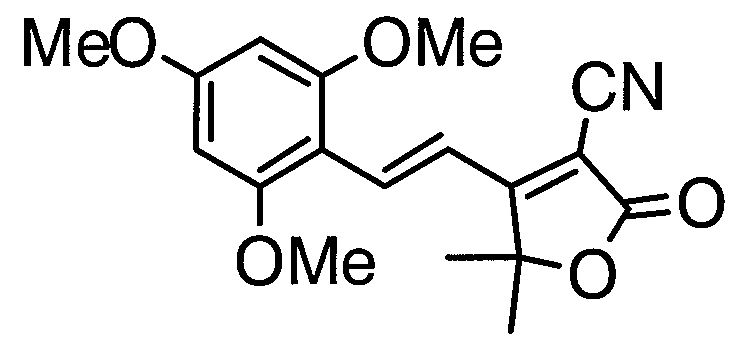
- CRBZVDLXAIFERF-UHFFFAOYSA-N 2,4,6-trimethoxybenzaldehyde Chemical compound COC1=CC(OC)=C(C=O)C(OC)=C1 CRBZVDLXAIFERF-UHFFFAOYSA-N 0.000 description 2
- 125000005808 2,4,6-trimethoxyphenyl group Chemical group [H][#6]-1=[#6](-[#8]C([H])([H])[H])-[#6](-*)=[#6](-[#8]C([H])([H])[H])-[#6]([H])=[#6]-1-[#8]C([H])([H])[H] 0.000 description 2
- BQOZTZUPCCMROE-UHFFFAOYSA-N 2-[(3-methoxyphenyl)methylamino]ethanol Chemical compound COC1=CC=CC(CNCCO)=C1 BQOZTZUPCCMROE-UHFFFAOYSA-N 0.000 description 2
- HRCFVAPRZYAOAA-UHFFFAOYSA-N 2-[(3-phenylmethoxyphenyl)methylamino]ethanol Chemical compound OCCNCC1=CC=CC(OCC=2C=CC=CC=2)=C1 HRCFVAPRZYAOAA-UHFFFAOYSA-N 0.000 description 2
- RWOSASOVVDBNFZ-UHFFFAOYSA-N 2-[(4-formyl-3-methoxyphenyl)methylamino]ethyl 2,2-dimethylpropanoate Chemical compound COC1=CC(CNCCOC(=O)C(C)(C)C)=CC=C1C=O RWOSASOVVDBNFZ-UHFFFAOYSA-N 0.000 description 2
- RPVSECPKTHFFNL-UHFFFAOYSA-N 2-[(4-formyl-3-phenylmethoxyphenyl)methylamino]ethyl 2,2-dimethylpropanoate Chemical compound CC(C)(C)C(=O)OCCNCC1=CC=C(C=O)C(OCC=2C=CC=CC=2)=C1 RPVSECPKTHFFNL-UHFFFAOYSA-N 0.000 description 2
- BFICZHJSKDVIDE-UHFFFAOYSA-N 2-[2-butyl-3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]acetonitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=C(CCCC)C(=CC#N)CC(C)(C)C1 BFICZHJSKDVIDE-UHFFFAOYSA-N 0.000 description 2
- NJPQSRLKVIXIKN-UHFFFAOYSA-N 2-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-2-methoxy-5,5-dimethylcyclohex-2-en-1-ylidene]acetaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=C(OC)C(=CC=O)CC(C)(C)C1 NJPQSRLKVIXIKN-UHFFFAOYSA-N 0.000 description 2
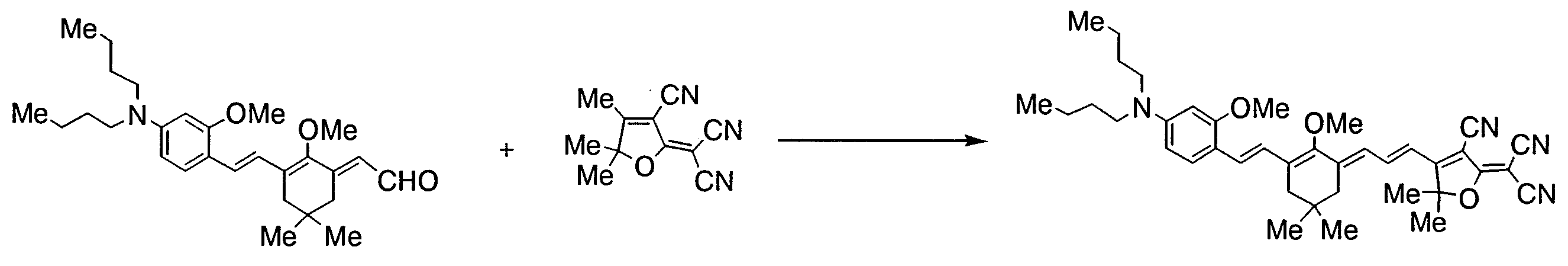
- AUFOLAMNTKIUGI-UHFFFAOYSA-N 2-[3-cyano-4-[3-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-2-methoxy-5,5-dimethylcyclohex-2-en-1-ylidene]prop-1-enyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(CC(C)(C)C1)=C(OC)C1=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C AUFOLAMNTKIUGI-UHFFFAOYSA-N 0.000 description 2
- BRHDJINJLAXJRW-UHFFFAOYSA-N 2-[3-cyano-4-[3-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]prop-1-enyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(CC(C)(C)C1)=CC1=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C BRHDJINJLAXJRW-UHFFFAOYSA-N 0.000 description 2
- HYUCTWATXYDDCQ-UHFFFAOYSA-N 2-[3-cyano-4-[4-[4-(dibutylamino)-2-methoxyphenyl]buta-1,3-dienyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C HYUCTWATXYDDCQ-UHFFFAOYSA-N 0.000 description 2
- NUQALBNUJLZCIO-UHFFFAOYSA-N 2-[3-cyano-4-[6-[4-(dibutylamino)-2-methoxyphenyl]hexa-1,3,5-trienyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C NUQALBNUJLZCIO-UHFFFAOYSA-N 0.000 description 2
- IQATUQWJEZGRDR-UHFFFAOYSA-N 2-[4-[2-[5-[2-[4-[butyl(4-hydroxybutyl)amino]-2-phenylmethoxyphenyl]ethenyl]thiophen-2-yl]ethenyl]-3-cyano-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound C=1C=CC=CC=1COC1=CC(N(CCCCO)CCCC)=CC=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C IQATUQWJEZGRDR-UHFFFAOYSA-N 0.000 description 2
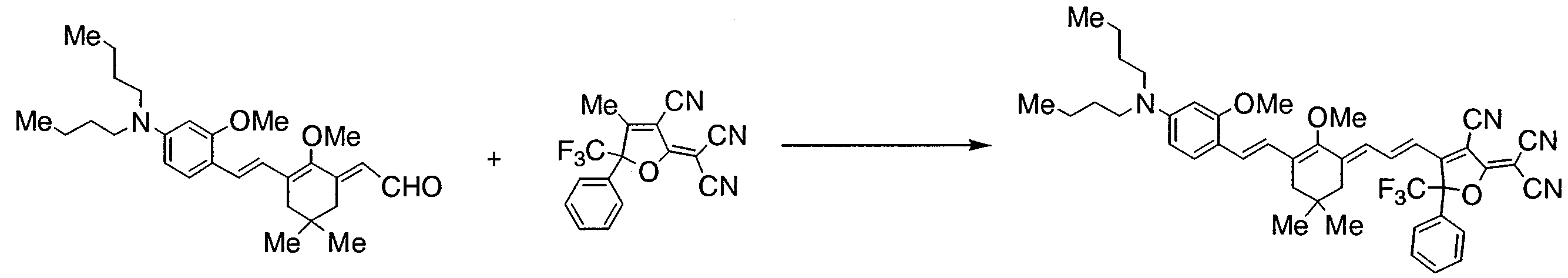
- DIUXTPCKKOFPGM-UHFFFAOYSA-N 2-[4-[3-[2-butyl-3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]prop-1-enyl]-3-cyano-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(CC(C)(C)C1)=C(CCCC)C1=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 DIUXTPCKKOFPGM-UHFFFAOYSA-N 0.000 description 2
- ZCTPKLBBEDZEQM-UHFFFAOYSA-N 2-[[3-methoxy-4-(2-thiophen-2-ylethenyl)phenyl]methylamino]ethanol Chemical compound COC1=CC(CNCCO)=CC=C1C=CC1=CC=CS1 ZCTPKLBBEDZEQM-UHFFFAOYSA-N 0.000 description 2
- QHQSVMOJQYNXSO-UHFFFAOYSA-N 2-butyl-3,5,5-trimethylcyclohex-2-en-1-one Chemical compound CCCCC1=C(C)CC(C)(C)CC1=O QHQSVMOJQYNXSO-UHFFFAOYSA-N 0.000 description 2
- UJOGYONNXWKUAI-UHFFFAOYSA-N 2-butyl-3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-one Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=C(CCCC)C(=O)CC(C)(C)C1 UJOGYONNXWKUAI-UHFFFAOYSA-N 0.000 description 2
- 125000006022 2-methyl-2-propenyl group Chemical group 0.000 description 2
- 125000004924 2-naphthylethyl group Chemical group C1=C(C=CC2=CC=CC=C12)CC* 0.000 description 2
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 2
- XUIDKEKENFBXKM-UHFFFAOYSA-N 3-[5-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]thiophen-2-yl]prop-2-enal Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=CC=O)S1 XUIDKEKENFBXKM-UHFFFAOYSA-N 0.000 description 2
- YPBLKTPTHKTSSQ-UHFFFAOYSA-N 3-[5-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]thiophen-2-yl]prop-2-enenitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=CC#N)S1 YPBLKTPTHKTSSQ-UHFFFAOYSA-N 0.000 description 2
- VNWPLOWYHIDMEB-UHFFFAOYSA-N 4-(dibutylamino)benzaldehyde Chemical compound CCCCN(CCCC)C1=CC=C(C=O)C=C1 VNWPLOWYHIDMEB-UHFFFAOYSA-N 0.000 description 2
- UODXTDOQNNLWSH-UHFFFAOYSA-N 4-[4-(3-phenylmethoxyphenyl)butylamino]butyl acetate Chemical compound CC(=O)OCCCCNCCCCC1=CC=CC(OCC=2C=CC=CC=2)=C1 UODXTDOQNNLWSH-UHFFFAOYSA-N 0.000 description 2
- JXZQWAAHLJXECQ-UHFFFAOYSA-N 4-[4-(4-formyl-3-phenylmethoxyphenyl)butylamino]butyl acetate Chemical compound CC(=O)OCCCCNCCCCC1=CC=C(C=O)C(OCC=2C=CC=CC=2)=C1 JXZQWAAHLJXECQ-UHFFFAOYSA-N 0.000 description 2
- IOZGOLSOBZJRMQ-UHFFFAOYSA-N 4-[butyl(4-hydroxybutyl)amino]-2-phenylmethoxybenzaldehyde Chemical compound OCCCCN(CCCC)C1=CC=C(C=O)C(OCC=2C=CC=CC=2)=C1 IOZGOLSOBZJRMQ-UHFFFAOYSA-N 0.000 description 2
- VFZRIQDDCOLUNB-UHFFFAOYSA-N 4-[butyl-[4-[tert-butyl(diphenyl)silyl]oxybutyl]amino]-2-phenylmethoxybenzaldehyde Chemical compound C=1C=C(C=O)C(OCC=2C=CC=CC=2)=CC=1N(CCCC)CCCCO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 VFZRIQDDCOLUNB-UHFFFAOYSA-N 0.000 description 2
- YVPNPAYQLSEULM-UHFFFAOYSA-N 5-[2-[4-(dibutylamino)-2-(oxiran-2-ylmethoxy)phenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound C1OC1COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 YVPNPAYQLSEULM-UHFFFAOYSA-N 0.000 description 2
- CQRJLNADPDVWNG-UHFFFAOYSA-N 5-[2-[4-(dibutylamino)phenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound C1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 CQRJLNADPDVWNG-UHFFFAOYSA-N 0.000 description 2
- GLBYQKLCUORPSD-UHFFFAOYSA-N 5-[2-[4-[2-[tert-butyl(dimethyl)silyl]oxyethyl-methylamino]-2-methoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound [Si](C)(C)(C(C)(C)C)OCCN(C1=CC(=C(C=C1)C=CC1=CC=C(S1)C=O)OC)C GLBYQKLCUORPSD-UHFFFAOYSA-N 0.000 description 2
- GVLGLFOEEQXKOE-UHFFFAOYSA-N 5-[2-[4-[2-[tert-butyl(dimethyl)silyl]oxyethyl-methylamino]-2-phenylmethoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound CN(CCO[Si](C)(C)C(C)(C)C)c1ccc(C=Cc2ccc(C=O)s2)c(OCc2ccccc2)c1 GVLGLFOEEQXKOE-UHFFFAOYSA-N 0.000 description 2
- JEBDYZIFAQXPLP-UHFFFAOYSA-N 5-[2-[4-[butyl-[4-[tert-butyl(diphenyl)silyl]oxybutyl]amino]-2-phenylmethoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound C=1C=C(C=CC=2SC(C=O)=CC=2)C(OCC=2C=CC=CC=2)=CC=1N(CCCC)CCCCO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 JEBDYZIFAQXPLP-UHFFFAOYSA-N 0.000 description 2
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 2
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 2
- 241001177361 Breda Species 0.000 description 2
- 125000003358 C2-C20 alkenyl group Chemical group 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 239000004642 Polyimide Substances 0.000 description 2
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 2
- XBDBWNDYGGPWQI-UHFFFAOYSA-N [4-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-2,5-dimethylphenyl]methanol Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC(C)=C(CO)C=C1C XBDBWNDYGGPWQI-UHFFFAOYSA-N 0.000 description 2
- CYRJPMUCGQIMBQ-UHFFFAOYSA-N [4-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]phenyl]methanol Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(CO)C=C1 CYRJPMUCGQIMBQ-UHFFFAOYSA-N 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 125000004429 atom Chemical group 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 2
- 235000019445 benzyl alcohol Nutrition 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 239000007810 chemical reaction solvent Substances 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 238000004891 communication Methods 0.000 description 2
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000003678 cyclohexadienyl group Chemical group C1(=CC=CCC1)* 0.000 description 2
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 2
- 125000006547 cyclononyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 2
- 125000000522 cyclooctenyl group Chemical group C1(=CCCCCCC1)* 0.000 description 2
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 2
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 description 2
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000000298 cyclopropenyl group Chemical group [H]C1=C([H])C1([H])* 0.000 description 2
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 2
- 230000000593 degrading effect Effects 0.000 description 2
- JQVDAXLFBXTEQA-UHFFFAOYSA-N dibutylamine Chemical compound CCCCNCCCC JQVDAXLFBXTEQA-UHFFFAOYSA-N 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- GKIPXFAANLTWBM-UHFFFAOYSA-N epibromohydrin Chemical compound BrCC1CO1 GKIPXFAANLTWBM-UHFFFAOYSA-N 0.000 description 2
- 239000003822 epoxy resin Substances 0.000 description 2
- VMZXXMQEPUPGHT-UHFFFAOYSA-N ethyl 2-[(3-methoxyphenyl)methylamino]acetate Chemical compound CCOC(=O)CNCC1=CC=CC(OC)=C1 VMZXXMQEPUPGHT-UHFFFAOYSA-N 0.000 description 2
- YPPQBSOJMIROEK-UHFFFAOYSA-N ethyl 2-[(3-phenylmethoxyphenyl)methylamino]acetate Chemical compound CCOC(=O)CNCC1=CC=CC(OCC=2C=CC=CC=2)=C1 YPPQBSOJMIROEK-UHFFFAOYSA-N 0.000 description 2
- PQJJJMRNHATNKG-UHFFFAOYSA-N ethyl bromoacetate Chemical compound CCOC(=O)CBr PQJJJMRNHATNKG-UHFFFAOYSA-N 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 230000009477 glass transition Effects 0.000 description 2
- 238000005658 halogenation reaction Methods 0.000 description 2
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 2
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 2
- 125000001041 indolyl group Chemical group 0.000 description 2
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 2
- HJOVHMDZYOCNQW-UHFFFAOYSA-N isophorone Chemical compound CC1=CC(=O)CC(C)(C)C1 HJOVHMDZYOCNQW-UHFFFAOYSA-N 0.000 description 2
- DLEDOFVPSDKWEF-UHFFFAOYSA-N lithium butane Chemical compound [Li+].CCC[CH2-] DLEDOFVPSDKWEF-UHFFFAOYSA-N 0.000 description 2
- ZCSHNCUQKCANBX-UHFFFAOYSA-N lithium diisopropylamide Chemical compound [Li+].CC(C)[N-]C(C)C ZCSHNCUQKCANBX-UHFFFAOYSA-N 0.000 description 2
- DBYQHFPBWKKZAT-UHFFFAOYSA-N lithium;benzene Chemical compound [Li+].C1=CC=[C-]C=C1 DBYQHFPBWKKZAT-UHFFFAOYSA-N 0.000 description 2
- 230000015654 memory Effects 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- MMTCINLVFUNXLN-UHFFFAOYSA-N n,n-dibutyl-2,6-dimethoxyaniline Chemical compound CCCCN(CCCC)C1=C(OC)C=CC=C1OC MMTCINLVFUNXLN-UHFFFAOYSA-N 0.000 description 2
- WRMZYFVPBPNKAQ-UHFFFAOYSA-N n,n-dibutyl-4-[2-[4-[[tert-butyl(diphenyl)silyl]oxymethyl]-2,5-dimethylphenyl]ethenyl]-3-methoxyaniline Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(C(=C1)C)=CC(C)=C1CO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 WRMZYFVPBPNKAQ-UHFFFAOYSA-N 0.000 description 2
- VVPZNKUHLIRTRQ-UHFFFAOYSA-N n,n-dibutyl-4-[2-[4-[[tert-butyl(diphenyl)silyl]oxymethyl]phenyl]ethenyl]-3-methoxyaniline Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(C=C1)=CC=C1CO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 VVPZNKUHLIRTRQ-UHFFFAOYSA-N 0.000 description 2
- GGSSXTWGEIRDKI-UHFFFAOYSA-N n-[2-[tert-butyl(dimethyl)silyl]oxyethyl]-3-methoxy-n-methyl-4-(2-thiophen-2-ylethenyl)aniline Chemical compound COC1=CC(N(C)CCO[Si](C)(C)C(C)(C)C)=CC=C1C=CC1=CC=CS1 GGSSXTWGEIRDKI-UHFFFAOYSA-N 0.000 description 2
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000005186 naphthyloxy group Chemical group C1(=CC=CC2=CC=CC=C12)O* 0.000 description 2
- 239000012299 nitrogen atmosphere Substances 0.000 description 2
- 239000011368 organic material Substances 0.000 description 2
- 125000000466 oxiranyl group Chemical group 0.000 description 2
- RLOWWWKZYUNIDI-UHFFFAOYSA-N phosphinic chloride Chemical compound ClP=O RLOWWWKZYUNIDI-UHFFFAOYSA-N 0.000 description 2
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 2
- 125000003367 polycyclic group Chemical group 0.000 description 2
- 229920000647 polyepoxide Polymers 0.000 description 2
- 229920001721 polyimide Polymers 0.000 description 2
- 239000004926 polymethyl methacrylate Substances 0.000 description 2
- IUBQJLUDMLPAGT-UHFFFAOYSA-N potassium bis(trimethylsilyl)amide Chemical compound C[Si](C)(C)N([K])[Si](C)(C)C IUBQJLUDMLPAGT-UHFFFAOYSA-N 0.000 description 2
- 239000003586 protic polar solvent Substances 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- 230000000630 rising effect Effects 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 125000004426 substituted alkynyl group Chemical group 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- MHYGQXWCZAYSLJ-UHFFFAOYSA-N tert-butyl-chloro-diphenylsilane Chemical compound C=1C=CC=CC=1[Si](Cl)(C(C)(C)C)C1=CC=CC=C1 MHYGQXWCZAYSLJ-UHFFFAOYSA-N 0.000 description 2
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 description 2
- JRMUNVKIHCOMHV-UHFFFAOYSA-M tetrabutylammonium bromide Chemical compound [Br-].CCCC[N+](CCCC)(CCCC)CCCC JRMUNVKIHCOMHV-UHFFFAOYSA-M 0.000 description 2
- NLDYACGHTUPAQU-UHFFFAOYSA-N tetracyanoethylene Chemical group N#CC(C#N)=C(C#N)C#N NLDYACGHTUPAQU-UHFFFAOYSA-N 0.000 description 2
- PCCVSPMFGIFTHU-UHFFFAOYSA-N tetracyanoquinodimethane Chemical compound N#CC(C#N)=C1C=CC(=C(C#N)C#N)C=C1 PCCVSPMFGIFTHU-UHFFFAOYSA-N 0.000 description 2
- PJQVALHFZIYJMT-UHFFFAOYSA-M triphenyl(thiophen-2-ylmethyl)phosphanium;bromide Chemical compound [Br-].C=1C=CC=CC=1[P+](C=1C=CC=CC=1)(C=1C=CC=CC=1)CC1=CC=CS1 PJQVALHFZIYJMT-UHFFFAOYSA-M 0.000 description 2
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 2
- DYLIWHYUXAJDOJ-OWOJBTEDSA-N (e)-4-(6-aminopurin-9-yl)but-2-en-1-ol Chemical compound NC1=NC=NC2=C1N=CN2C\C=C\CO DYLIWHYUXAJDOJ-OWOJBTEDSA-N 0.000 description 1
- KRWRFIMBWRVMKE-UHFFFAOYSA-N 1-bromo-3,5-dimethoxybenzene Chemical compound COC1=CC(Br)=CC(OC)=C1 KRWRFIMBWRVMKE-UHFFFAOYSA-N 0.000 description 1
- MPPPKRYCTPRNTB-UHFFFAOYSA-N 1-bromobutane Chemical compound CCCCBr MPPPKRYCTPRNTB-UHFFFAOYSA-N 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000006019 1-methyl-1-propenyl group Chemical group 0.000 description 1
- 125000006018 1-methyl-ethenyl group Chemical group 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- YQTCQNIPQMJNTI-UHFFFAOYSA-N 2,2-dimethylpropan-1-one Chemical group CC(C)(C)[C]=O YQTCQNIPQMJNTI-UHFFFAOYSA-N 0.000 description 1
- GVNVAWHJIKLAGL-UHFFFAOYSA-N 2-(cyclohexen-1-yl)cyclohexan-1-one Chemical compound O=C1CCCCC1C1=CCCCC1 GVNVAWHJIKLAGL-UHFFFAOYSA-N 0.000 description 1
- SLGWYHZXRTYOFA-UHFFFAOYSA-N 2-[2-butyl-3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]acetaldehyde Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=C(CCCC)C(=CC=O)CC(C)(C)C1 SLGWYHZXRTYOFA-UHFFFAOYSA-N 0.000 description 1
- HBZYYOYCJQHAEL-UHFFFAOYSA-N 2-[3-(dicyanomethylidene)inden-1-ylidene]propanedinitrile Chemical compound C1=CC=C2C(=C(C#N)C#N)CC(=C(C#N)C#N)C2=C1 HBZYYOYCJQHAEL-UHFFFAOYSA-N 0.000 description 1
- MCWCIIIPSZRQQJ-UHFFFAOYSA-N 2-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-2-methoxy-5,5-dimethylcyclohex-2-en-1-ylidene]acetonitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=C(OC)C(=CC#N)CC(C)(C)C1 MCWCIIIPSZRQQJ-UHFFFAOYSA-N 0.000 description 1
- IAGHSHPBZPKROQ-UHFFFAOYSA-N 2-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]acetonitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC(=CC#N)CC(C)(C)C1 IAGHSHPBZPKROQ-UHFFFAOYSA-N 0.000 description 1
- WMHCINIHMSAVTF-UHFFFAOYSA-N 2-[3-cyano-4-[2-[4-(dibutylamino)-2,6-dimethoxyphenyl]ethenyl]-5-methyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC(OC)=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C(F)(F)F WMHCINIHMSAVTF-UHFFFAOYSA-N 0.000 description 1
- NFEUGKZSVQFVHG-UHFFFAOYSA-N 2-[3-cyano-4-[2-[4-(dibutylamino)-2,6-dimethoxyphenyl]ethenyl]-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC(OC)=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 NFEUGKZSVQFVHG-UHFFFAOYSA-N 0.000 description 1
- DMUSDPFGIPNNHH-UHFFFAOYSA-N 2-[3-cyano-4-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5-methyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C(F)(F)F DMUSDPFGIPNNHH-UHFFFAOYSA-N 0.000 description 1
- XFHFJAZEJGLOSP-UHFFFAOYSA-N 2-[3-cyano-4-[2-[4-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]phenyl]ethenyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(C=C1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C XFHFJAZEJGLOSP-UHFFFAOYSA-N 0.000 description 1
- XTKSYYVFKJTOMW-UHFFFAOYSA-N 2-[3-cyano-4-[2-[4-[2-[4-(dibutylamino)phenyl]ethenyl]phenyl]ethenyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound C1=CC(N(CCCC)CCCC)=CC=C1C=CC(C=C1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C XTKSYYVFKJTOMW-UHFFFAOYSA-N 0.000 description 1
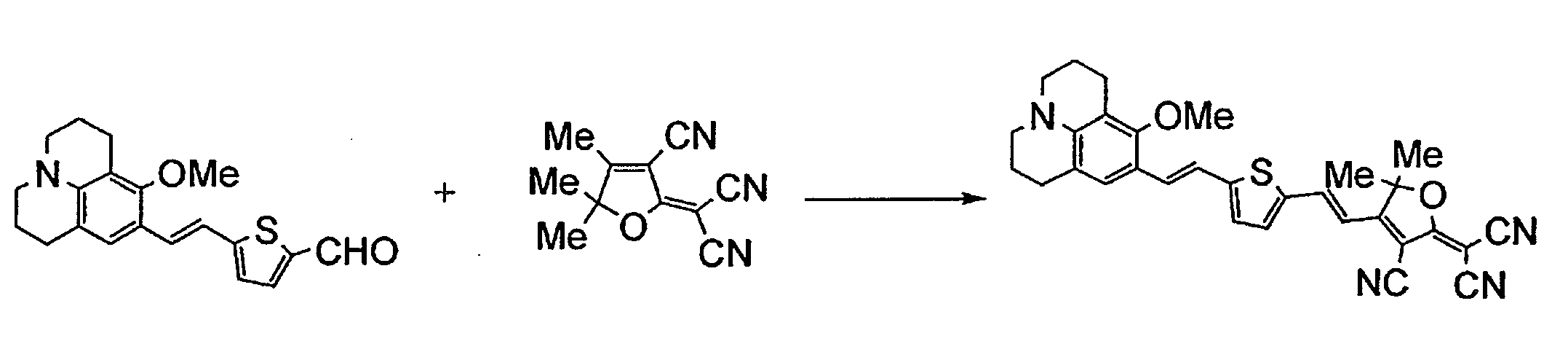
- YOROEHQKMUTOKO-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[2-(8-methoxy-2,3,6,7-tetrahydro-1h,5h-benzo[ij]quinolizine-9-yl)vinyl]thiophene-2-yl]vinyl]-5,5-dimethyl-2(5h)-furanylidene]propanedinitrile Chemical compound C1=C(C=23)CCCN3CCCC=2C(OC)=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C YOROEHQKMUTOKO-UHFFFAOYSA-N 0.000 description 1
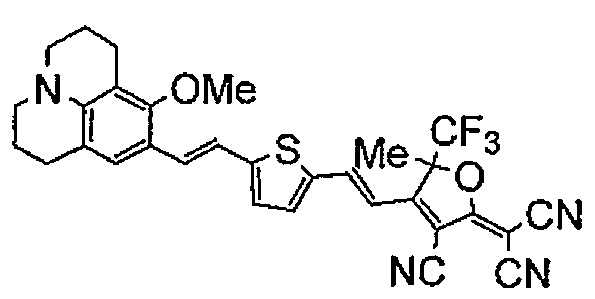
- YODUOSWKWGDHHU-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[2-(8-methoxy-2,3,6,7-tetrahydro-1h,5h-benzo[ij]quinolizine-9-yl)vinyl]thiophene-2-yl]vinyl]-5-phenyl-5-trifluoromethyl-2(5h)-furanylidene]propanedinitrile Chemical compound C1=C(C=23)CCCN3CCCC=2C(OC)=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 YODUOSWKWGDHHU-UHFFFAOYSA-N 0.000 description 1
- JYMNWHBFSBLAKH-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[2-[4-(dibutylamino)phenyl]ethenyl]thiophen-2-yl]ethenyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound C1=CC(N(CCCC)CCCC)=CC=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C JYMNWHBFSBLAKH-UHFFFAOYSA-N 0.000 description 1
- AOLRDRUJXYWUDM-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[2-[4-[2-hydroxyethyl(methyl)amino]-2-methoxyphenyl]ethenyl]thiophen-2-yl]ethenyl]-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(C)CCO)=CC=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 AOLRDRUJXYWUDM-UHFFFAOYSA-N 0.000 description 1
- ITENJFHRZJSHFM-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[4-[4-(dibutylamino)-2-methoxyphenyl]buta-1,3-dienyl]thiophen-2-yl]ethenyl]-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C ITENJFHRZJSHFM-UHFFFAOYSA-N 0.000 description 1
- CLRKJMBLGUNVJF-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[4-[4-(dibutylamino)-2-methoxyphenyl]buta-1,3-dienyl]thiophen-2-yl]ethenyl]-5-methyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C(F)(F)F CLRKJMBLGUNVJF-UHFFFAOYSA-N 0.000 description 1
- HZOLDTPPHMDKAF-UHFFFAOYSA-N 2-[3-cyano-4-[2-[5-[4-[4-(dibutylamino)-2-methoxyphenyl]buta-1,3-dienyl]thiophen-2-yl]ethenyl]-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 HZOLDTPPHMDKAF-UHFFFAOYSA-N 0.000 description 1
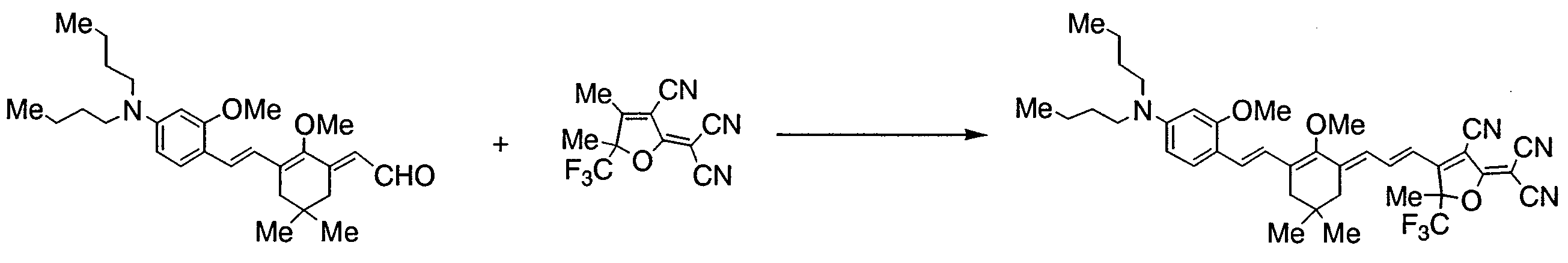
- RLZWHOYCEBXLIC-UHFFFAOYSA-N 2-[3-cyano-4-[3-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]prop-1-enyl]-5-methyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(CC(C)(C)C1)=CC1=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C(F)(F)F RLZWHOYCEBXLIC-UHFFFAOYSA-N 0.000 description 1
- DQPZONARUJMOMN-UHFFFAOYSA-N 2-[3-cyano-4-[3-[3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-ylidene]prop-1-enyl]-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(CC(C)(C)C1)=CC1=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 DQPZONARUJMOMN-UHFFFAOYSA-N 0.000 description 1
- HFOQTALETRIZHR-UHFFFAOYSA-N 2-[3-cyano-4-[4-[5-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]thiophen-2-yl]buta-1,3-dienyl]-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC(S1)=CC=C1C=CC=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 HFOQTALETRIZHR-UHFFFAOYSA-N 0.000 description 1
- OWYQGBOGFKWNRO-UHFFFAOYSA-N 2-[3-cyano-5-phenyl-5-(trifluoromethyl)-4-[2-[5-[2-(2,4,6-trimethoxyphenyl)ethenyl]thiophen-2-yl]ethenyl]furan-2-ylidene]propanedinitrile Chemical compound COC1=CC(OC)=CC(OC)=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 OWYQGBOGFKWNRO-UHFFFAOYSA-N 0.000 description 1
- JOVWGZLJDJSIJB-UHFFFAOYSA-N 2-[4-(dibutylamino)-2-methoxyphenyl]ethene-1,1,2-tricarbonitrile Chemical compound CCCCN(CCCC)C1=CC=C(C(C#N)=C(C#N)C#N)C(OC)=C1 JOVWGZLJDJSIJB-UHFFFAOYSA-N 0.000 description 1
- ZAYSNPGDEDWBKI-UHFFFAOYSA-N 2-[4-[2-[5-[2-[2-[tert-butyl(diphenyl)silyl]oxy-4-(dibutylamino)phenyl]ethenyl]thiophen-2-yl]ethenyl]-3-cyano-5,5-dimethylfuran-2-ylidene]propanedinitrile Chemical compound C=1C=CC=CC=1[Si](C(C)(C)C)(C=1C=CC=CC=1)OC1=CC(N(CCCC)CCCC)=CC=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C ZAYSNPGDEDWBKI-UHFFFAOYSA-N 0.000 description 1
- KVDFMOJGTKYIKL-UHFFFAOYSA-N 2-[4-[2-[5-[2-[2-[tert-butyl(diphenyl)silyl]oxy-4-(dibutylamino)phenyl]ethenyl]thiophen-2-yl]ethenyl]-3-cyano-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound CCCCN(CCCC)c1ccc(C=Cc2ccc(C=CC3=C(C#N)C(OC3(c3ccccc3)C(F)(F)F)=C(C#N)C#N)s2)c(O[Si](c2ccccc2)(c2ccccc2)C(C)(C)C)c1 KVDFMOJGTKYIKL-UHFFFAOYSA-N 0.000 description 1
- IIPFYGCOMRUNGN-UHFFFAOYSA-N 2-[4-[2-[5-[2-[4-[butyl(4-hydroxybutyl)amino]-2-phenylmethoxyphenyl]ethenyl]thiophen-2-yl]ethenyl]-3-cyano-5-phenyl-5-(trifluoromethyl)furan-2-ylidene]propanedinitrile Chemical compound C=1C=CC=CC=1COC1=CC(N(CCCCO)CCCC)=CC=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C(F)(F)F)C1=CC=CC=C1 IIPFYGCOMRUNGN-UHFFFAOYSA-N 0.000 description 1
- GMXNZHFGNXWCTF-UHFFFAOYSA-N 2-[4-[cyano-[4-(dibutylamino)-2-methoxyphenyl]methylidene]cyclohexa-2,5-dien-1-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C(C#N)=C1C=CC(=C(C#N)C#N)C=C1 GMXNZHFGNXWCTF-UHFFFAOYSA-N 0.000 description 1
- DALRTXMTJKCFGT-UHFFFAOYSA-N 2-[4-[cyano-[5-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]thiophen-2-yl]methylidene]cyclohexa-2,5-dien-1-ylidene]propanedinitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C(C#N)=C2C=CC(C=C2)=C(C#N)C#N)S1 DALRTXMTJKCFGT-UHFFFAOYSA-N 0.000 description 1
- AMPGMPACLOOCJE-UHFFFAOYSA-N 2-[5-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]thiophen-2-yl]ethene-1,1,2-tricarbonitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C(C#N)=C(C#N)C#N)S1 AMPGMPACLOOCJE-UHFFFAOYSA-N 0.000 description 1
- OQBHAZKJEDJMMA-UHFFFAOYSA-N 2-[5-[2-[4-(dibutylamino)-2-phenylmethoxyphenyl]ethenyl]thiophen-2-yl]ethene-1,1,2-tricarbonitrile Chemical compound C=1C=CC=CC=1COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C(C#N)=C(C#N)C#N)S1 OQBHAZKJEDJMMA-UHFFFAOYSA-N 0.000 description 1
- FFKZSMMQACBLBO-UHFFFAOYSA-N 2-[n-methyl-3-phenylmethoxy-4-(2-thiophen-2-ylethenyl)anilino]ethanol Chemical compound C=1C=CC=CC=1COC1=CC(N(CCO)C)=CC=C1C=CC1=CC=CS1 FFKZSMMQACBLBO-UHFFFAOYSA-N 0.000 description 1
- XUJYVPLKSDNTLG-UHFFFAOYSA-N 2-[tert-butyl(diphenyl)silyl]oxy-4-(dibutylamino)benzaldehyde Chemical compound CCCCN(CCCC)C1=CC=C(C=O)C(O[Si](C=2C=CC=CC=2)(C=2C=CC=CC=2)C(C)(C)C)=C1 XUJYVPLKSDNTLG-UHFFFAOYSA-N 0.000 description 1
- 125000005999 2-bromoethyl group Chemical group 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- BRXDVFXLLXAJKA-UHFFFAOYSA-N 2-methoxy-3,5,5-trimethylcyclohex-2-en-1-one Chemical compound COC1=C(C)CC(C)(C)CC1=O BRXDVFXLLXAJKA-UHFFFAOYSA-N 0.000 description 1
- 125000006020 2-methyl-1-propenyl group Chemical group 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- RSEBUVRVKCANEP-UHFFFAOYSA-N 2-pyrroline Chemical compound C1CC=CN1 RSEBUVRVKCANEP-UHFFFAOYSA-N 0.000 description 1
- KHSTZMGCKHBFJX-UHFFFAOYSA-N 3-(dibutylamino)phenol Chemical compound CCCCN(CCCC)C1=CC=CC(O)=C1 KHSTZMGCKHBFJX-UHFFFAOYSA-N 0.000 description 1
- YODYFLQTADVJPD-UHFFFAOYSA-N 3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-2-methoxy-5,5-dimethylcyclohex-2-en-1-one Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=C(OC)C(=O)CC(C)(C)C1 YODYFLQTADVJPD-UHFFFAOYSA-N 0.000 description 1
- GZGDCNQLRRBJFM-UHFFFAOYSA-N 3-[2-[4-(dibutylamino)-2-methoxyphenyl]ethenyl]-5,5-dimethylcyclohex-2-en-1-one Chemical compound COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC(=O)CC(C)(C)C1 GZGDCNQLRRBJFM-UHFFFAOYSA-N 0.000 description 1
- XHPZKAKWCCXHJM-UHFFFAOYSA-N 3-[4-(dibutylamino)-2-methoxyphenyl]prop-2-enenitrile Chemical compound CCCCN(CCCC)C1=CC=C(C=CC#N)C(OC)=C1 XHPZKAKWCCXHJM-UHFFFAOYSA-N 0.000 description 1
- 125000004975 3-butenyl group Chemical group C(CC=C)* 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- IGPFOKFDBICQMC-UHFFFAOYSA-N 3-phenylmethoxyaniline Chemical compound NC1=CC=CC(OCC=2C=CC=CC=2)=C1 IGPFOKFDBICQMC-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- OPUJAKVDYGQVHP-UHFFFAOYSA-N 4-(butylamino)butan-1-ol Chemical compound CCCCNCCCCO OPUJAKVDYGQVHP-UHFFFAOYSA-N 0.000 description 1
- NFDINLYPRUEBML-UHFFFAOYSA-N 4-(dibutylamino)-2-phenylmethoxybenzaldehyde Chemical compound CCCCN(CCCC)C1=CC=C(C=O)C(OCC=2C=CC=CC=2)=C1 NFDINLYPRUEBML-UHFFFAOYSA-N 0.000 description 1
- UYIWMMVKRHJJHW-UHFFFAOYSA-N 4-[2-[4-(dibutylamino)-2,6-dimethoxyphenyl]ethenyl]-5,5-dimethyl-2-oxofuran-3-carbonitrile Chemical compound COC1=CC(N(CCCC)CCCC)=CC(OC)=C1C=CC1=C(C#N)C(=O)OC1(C)C UYIWMMVKRHJJHW-UHFFFAOYSA-N 0.000 description 1
- SPHPWARFCMCCHV-UHFFFAOYSA-N 4-[2-[4-(dibutylamino)phenyl]ethenyl]benzaldehyde Chemical compound C1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)C=C1 SPHPWARFCMCCHV-UHFFFAOYSA-N 0.000 description 1
- YDPGZQDQPLNUMB-UHFFFAOYSA-N 4-[4-(3-phenylmethoxyphenyl)butylamino]butan-1-ol Chemical compound OCCCCNCCCCC1=CC=CC(OCC=2C=CC=CC=2)=C1 YDPGZQDQPLNUMB-UHFFFAOYSA-N 0.000 description 1
- AWQOWBKEEZTLNU-UHFFFAOYSA-N 5,5-dimethyl-2-oxo-4-[2-(2,4,6-trimethoxyphenyl)ethenyl]furan-3-carbonitrile Chemical compound COC1=CC(OC)=CC(OC)=C1C=CC1=C(C#N)C(=O)OC1(C)C AWQOWBKEEZTLNU-UHFFFAOYSA-N 0.000 description 1
- MXVOIGVDZAXUNR-UHFFFAOYSA-N 5-[2-[2-(3-bromo-2-hydroxypropoxy)-4-(dibutylamino)phenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound BrCC(O)COC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 MXVOIGVDZAXUNR-UHFFFAOYSA-N 0.000 description 1
- OXVRXCDSUQYEFA-UHFFFAOYSA-N 5-[2-[2-[tert-butyl(dimethyl)silyl]oxy-4-(dibutylamino)phenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound CC(C)(C)[Si](C)(C)OC1=CC(N(CCCC)CCCC)=CC=C1C=CC1=CC=C(C=O)S1 OXVRXCDSUQYEFA-UHFFFAOYSA-N 0.000 description 1
- DKEAMCZDOFKHER-UHFFFAOYSA-N 5-[2-[4-[2-hydroxyethyl(methyl)amino]-2-phenylmethoxyphenyl]ethenyl]thiophene-2-carbaldehyde Chemical compound C=1C=CC=CC=1COC1=CC(N(CCO)C)=CC=C1C=CC1=CC=C(C=O)S1 DKEAMCZDOFKHER-UHFFFAOYSA-N 0.000 description 1
- RLFBBWAULIGONR-UHFFFAOYSA-N 5-[4-(dibutylamino)-2-methoxyphenyl]penta-2,4-dienal Chemical compound CCCCN(CCCC)C1=CC=C(C=CC=CC=O)C(OC)=C1 RLFBBWAULIGONR-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- WZWBEYANEKEEBN-UHFFFAOYSA-N C(CCC)N(C1=CC(=C(C=C1)C=CC=1C=C(SC1)C=O)OC)CCCC Chemical compound C(CCC)N(C1=CC(=C(C=C1)C=CC=1C=C(SC1)C=O)OC)CCCC WZWBEYANEKEEBN-UHFFFAOYSA-N 0.000 description 1
- 125000003860 C1-C20 alkoxy group Chemical group 0.000 description 1
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 description 1
- ZADBFSKJOXMYSR-UHFFFAOYSA-N CC(C1C#N)C(C(F)(F)F)(c2ccccc2)OC1=C(C#N)C#N Chemical compound CC(C1C#N)C(C(F)(F)F)(c2ccccc2)OC1=C(C#N)C#N ZADBFSKJOXMYSR-UHFFFAOYSA-N 0.000 description 1
- NRJCAZLHXNBGQE-UHFFFAOYSA-N CC1=CCC1C1C#[N]C1 Chemical compound CC1=CCC1C1C#[N]C1 NRJCAZLHXNBGQE-UHFFFAOYSA-N 0.000 description 1
- BHUUDFAEFLJXOW-SIDAKMONSA-N CCCCN(CCCC)c1cc(OC)c(/C=C/c2c(C)cc(/C=C/C(C(C)(C(F)(F)F)OC3=C(C#N)C#N)=C3C#N)c(C)c2)cc1 Chemical compound CCCCN(CCCC)c1cc(OC)c(/C=C/c2c(C)cc(/C=C/C(C(C)(C(F)(F)F)OC3=C(C#N)C#N)=C3C#N)c(C)c2)cc1 BHUUDFAEFLJXOW-SIDAKMONSA-N 0.000 description 1
- LLZXDNMCPLRJLF-OJWGMWDRSA-N CCCCN(CCCC)c1cc(OC)c(/C=C/c2ccc(/C=C/C(C(C)(C)OC3=C(C#N)C#N)=C3C#N)[s]2)cc1 Chemical compound CCCCN(CCCC)c1cc(OC)c(/C=C/c2ccc(/C=C/C(C(C)(C)OC3=C(C#N)C#N)=C3C#N)[s]2)cc1 LLZXDNMCPLRJLF-OJWGMWDRSA-N 0.000 description 1
- MCWCIIIPSZRQQJ-YYTNQIMQSA-N CCCCN(CCCC)c1ccc(/C=C/C(CC(C)(C)C/C2=C\C#N)=C2OC)c(OC)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/C(CC(C)(C)C/C2=C\C#N)=C2OC)c(OC)c1 MCWCIIIPSZRQQJ-YYTNQIMQSA-N 0.000 description 1
- NJPQSRLKVIXIKN-YYTNQIMQSA-N CCCCN(CCCC)c1ccc(/C=C/C(CC(C)(C)C/C2=C\C=O)=C2OC)c(OC)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/C(CC(C)(C)C/C2=C\C=O)=C2OC)c(OC)c1 NJPQSRLKVIXIKN-YYTNQIMQSA-N 0.000 description 1
- PEKAUQJEZJNDTQ-DCIPZJNNSA-N CCCCN(CCCC)c1ccc(/C=C/C=C/C(C(C)=[O](C)C2=C(C#N)C#N)=C2C#N)c(OC)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/C=C/C(C(C)=[O](C)C2=C(C#N)C#N)=C2C#N)c(OC)c1 PEKAUQJEZJNDTQ-DCIPZJNNSA-N 0.000 description 1
- SYXACGHAQSPPFI-CMDGGOBGSA-N CCCCN(CCCC)c1ccc(/C=C/C=O)c(OC)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/C=O)c(OC)c1 SYXACGHAQSPPFI-CMDGGOBGSA-N 0.000 description 1
- PADLLLYWNNPBLV-ZHACJKMWSA-N CCCCN(CCCC)c1ccc(/C=C/c2c(C)cc(C=O)c(C)c2)c(OC)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/c2c(C)cc(C=O)c(C)c2)c(OC)c1 PADLLLYWNNPBLV-ZHACJKMWSA-N 0.000 description 1
- FKTQRKBYILAIMS-NETDEZLDSA-N CCCCN(CCCC)c1ccc(/C=C/c2cc(C)c(/C=C/C(C(C)(C)OC3=C(C#N)C#N)=C3C#N)cc2C)c(OC)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/c2cc(C)c(/C=C/C(C(C)(C)OC3=C(C#N)C#N)=C3C#N)cc2C)c(OC)c1 FKTQRKBYILAIMS-NETDEZLDSA-N 0.000 description 1
- RZCPWYFMQWWAHL-FYWRMAATSA-N CCCCN(CCCC)c1ccc(/C=C/c2ccc(C=O)[s]2)c(OCc2ccccc2)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/c2ccc(C=O)[s]2)c(OCc2ccccc2)c1 RZCPWYFMQWWAHL-FYWRMAATSA-N 0.000 description 1
- SPHPWARFCMCCHV-BQYQJAHWSA-N CCCCN(CCCC)c1ccc(/C=C/c2ccc(C=O)cc2)cc1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/c2ccc(C=O)cc2)cc1 SPHPWARFCMCCHV-BQYQJAHWSA-N 0.000 description 1
- LTXQSNXBYRNEPM-WJTDDFOZSA-N CCCCN(CCCC)c1ccc(/C=C/c2ccc[s]2)c(OCCS(C(C)(C)C)(c2ccccc2)c2ccccc2)c1 Chemical compound CCCCN(CCCC)c1ccc(/C=C/c2ccc[s]2)c(OCCS(C(C)(C)C)(c2ccccc2)c2ccccc2)c1 LTXQSNXBYRNEPM-WJTDDFOZSA-N 0.000 description 1
- TUSCZPXSZLDKRR-FNORWQNLSA-O CCCCNC(CC1[OH2+])=CC=C1/C=C/c1ccc(C=O)[s]1 Chemical compound CCCCNC(CC1[OH2+])=CC=C1/C=C/c1ccc(C=O)[s]1 TUSCZPXSZLDKRR-FNORWQNLSA-O 0.000 description 1
- RYLHRNNKENXNSW-UHFFFAOYSA-N COC1=CC(N(CCCC)CCCC)=CC(OC)=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C Chemical compound COC1=CC(N(CCCC)CCCC)=CC(OC)=C1C=CC(S1)=CC=C1C=CC1=C(C#N)C(=C(C#N)C#N)OC1(C)C RYLHRNNKENXNSW-UHFFFAOYSA-N 0.000 description 1
- YODUOSWKWGDHHU-YBAIQQBHSA-N COc1c(CCCN2CCC3)c2c3cc1/C=C/c1ccc(/C=C/C(C(C(F)(F)F)(c2ccccc2)OC2=C(C#N)C#N)=C2C#N)[s]1 Chemical compound COc1c(CCCN2CCC3)c2c3cc1/C=C/c1ccc(/C=C/C(C(C(F)(F)F)(c2ccccc2)OC2=C(C#N)C#N)=C2C#N)[s]1 YODUOSWKWGDHHU-YBAIQQBHSA-N 0.000 description 1
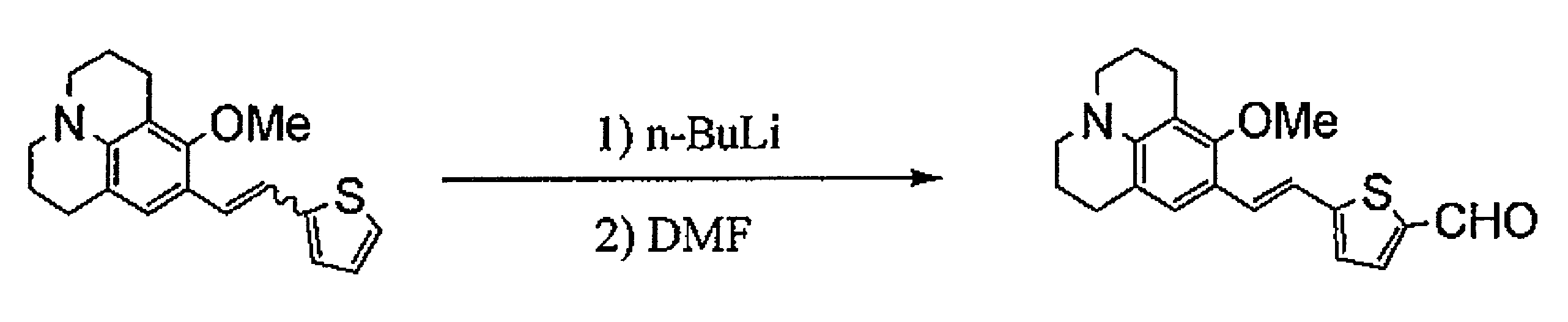
- BXTMBNXOCBKKEZ-VOTSOKGWSA-N COc1c(CCCN2CCC3)c2c3cc1/C=C/c1ccc(C=O)[s]1 Chemical compound COc1c(CCCN2CCC3)c2c3cc1/C=C/c1ccc(C=O)[s]1 BXTMBNXOCBKKEZ-VOTSOKGWSA-N 0.000 description 1
- IUBIQSNHMJUCRI-UHFFFAOYSA-N COc1c(CCCN2CCC3)c2c3cc1C=O Chemical compound COc1c(CCCN2CCC3)c2c3cc1C=O IUBIQSNHMJUCRI-UHFFFAOYSA-N 0.000 description 1
- OWYQGBOGFKWNRO-PHEQNACWSA-N COc1cc(OC)c(/C=C/c2ccc(/C=C/C(C(C(F)(F)F)(c3ccccc3)OC3=C(C#N)C#N)=C3C#N)[s]2)c(OC)c1 Chemical compound COc1cc(OC)c(/C=C/c2ccc(/C=C/C(C(C(F)(F)F)(c3ccccc3)OC3=C(C#N)C#N)=C3C#N)[s]2)c(OC)c1 OWYQGBOGFKWNRO-PHEQNACWSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 101150065749 Churc1 gene Proteins 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- FNJSWIPFHMKRAT-UHFFFAOYSA-N Monomethyl phthalate Chemical compound COC(=O)C1=CC=CC=C1C(O)=O FNJSWIPFHMKRAT-UHFFFAOYSA-N 0.000 description 1
- RSLSJQPCIOIUTN-UHFFFAOYSA-N N-[2-[tert-butyl(dimethyl)silyl]oxyethyl]-N-methyl-3-phenylmethoxy-4-(2-thiophen-2-ylethenyl)aniline Chemical compound C=1C=CC=CC=1COC1=CC(N(CCO[Si](C)(C)C(C)(C)C)C)=CC=C1C=CC1=CC=CS1 RSLSJQPCIOIUTN-UHFFFAOYSA-N 0.000 description 1
- OPFJDXRVMFKJJO-ZHHKINOHSA-N N-{[3-(2-benzamido-4-methyl-1,3-thiazol-5-yl)-pyrazol-5-yl]carbonyl}-G-dR-G-dD-dD-dD-NH2 Chemical compound S1C(C=2NN=C(C=2)C(=O)NCC(=O)N[C@H](CCCN=C(N)N)C(=O)NCC(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(N)=O)=C(C)N=C1NC(=O)C1=CC=CC=C1 OPFJDXRVMFKJJO-ZHHKINOHSA-N 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 102100038239 Protein Churchill Human genes 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- IBAOSSVIJDZMHY-UHFFFAOYSA-M [4-(hydroxymethyl)phenyl]methyl-triphenylphosphanium;bromide Chemical compound [Br-].C1=CC(CO)=CC=C1C[P+](C=1C=CC=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 IBAOSSVIJDZMHY-UHFFFAOYSA-M 0.000 description 1
- RDGVZHSAIDEHCQ-UHFFFAOYSA-M [4-[[tert-butyl(diphenyl)silyl]oxymethyl]phenyl]methyl-triphenylphosphanium;bromide Chemical compound [Br-].C=1C=CC=CC=1[Si](C=1C=CC=CC=1)(C(C)(C)C)OCC(C=C1)=CC=C1C[P+](C=1C=CC=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 RDGVZHSAIDEHCQ-UHFFFAOYSA-M 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- 125000000777 acyl halide group Chemical group 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910000102 alkali metal hydride Inorganic materials 0.000 description 1
- 150000008046 alkali metal hydrides Chemical class 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 125000004849 alkoxymethyl group Chemical group 0.000 description 1
- 125000005103 alkyl silyl group Chemical group 0.000 description 1
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 description 1
- 125000001204 arachidyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 description 1
- 125000005161 aryl oxy carbonyl group Chemical group 0.000 description 1
- 125000005874 benzothiadiazolyl group Chemical group 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- AGEZXYOZHKGVCM-UHFFFAOYSA-N benzyl bromide Chemical compound BrCC1=CC=CC=C1 AGEZXYOZHKGVCM-UHFFFAOYSA-N 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 125000003739 carbamimidoyl group Chemical group C(N)(=N)* 0.000 description 1
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 125000005708 carbonyloxy group Chemical group [*:2]OC([*:1])=O 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
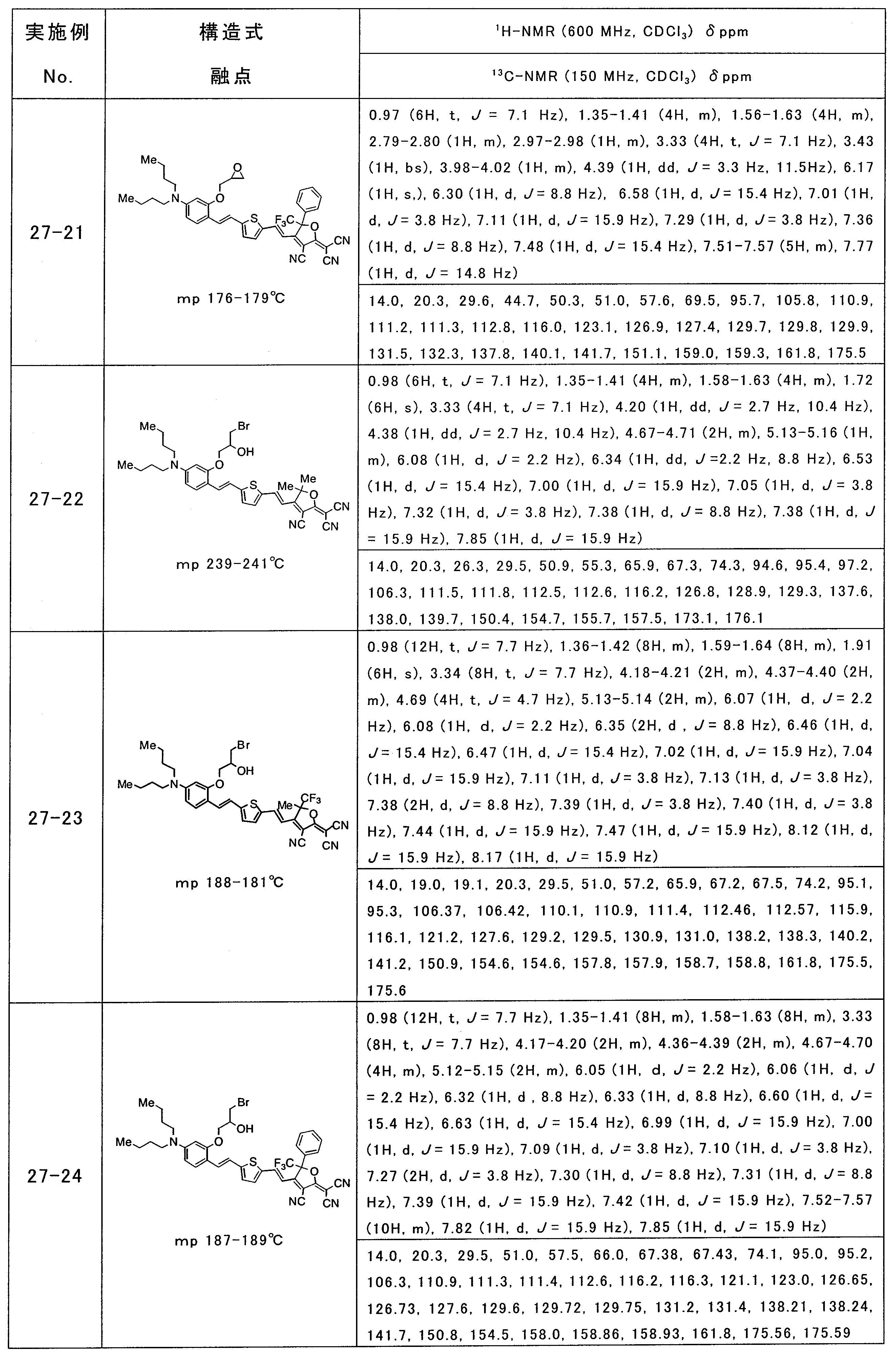
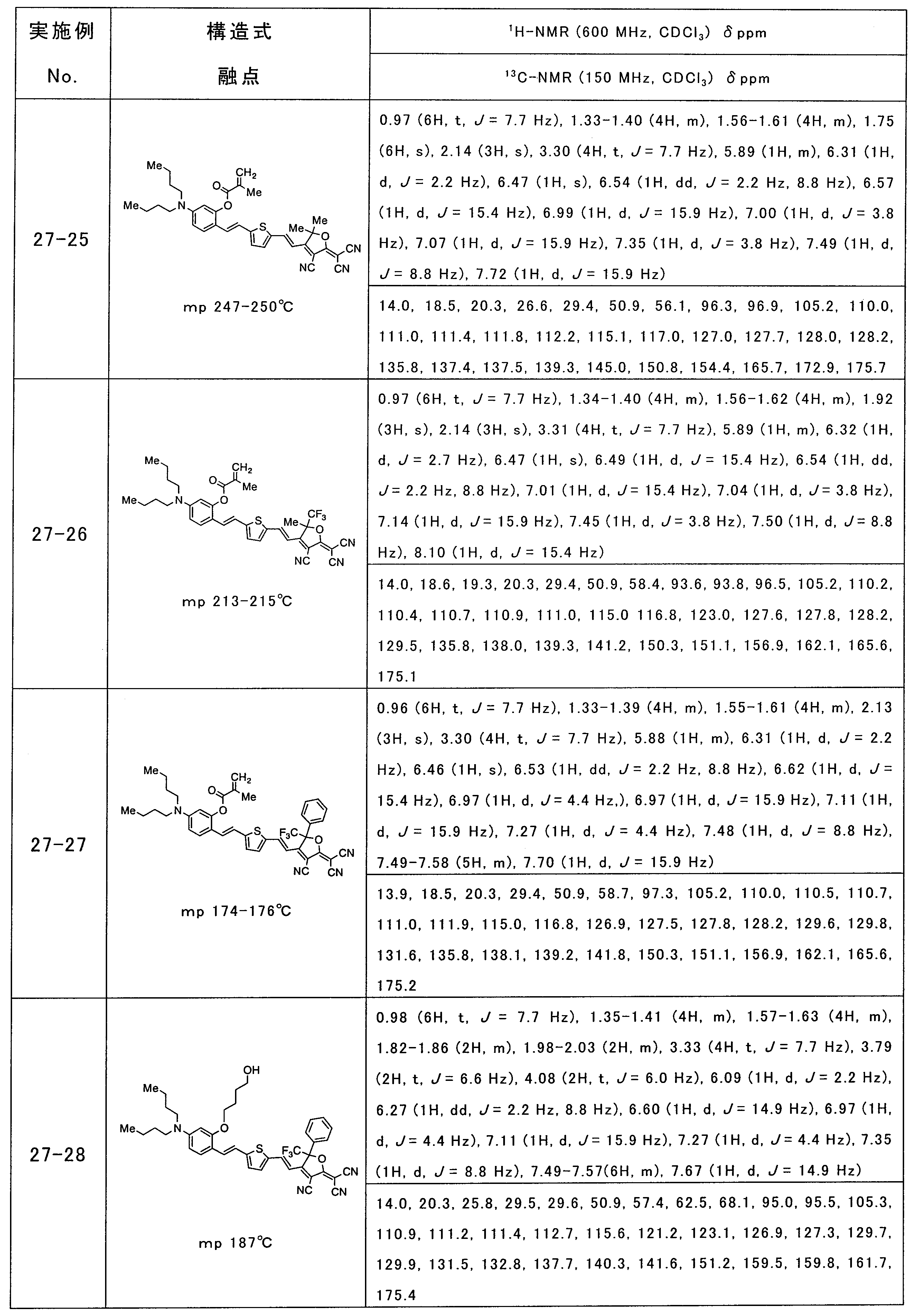
- 229940126086 compound 21 Drugs 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000000412 dendrimer Substances 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 238000004508 fractional distillation Methods 0.000 description 1
- 238000010575 fractional recrystallization Methods 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- ZLTPDFXIESTBQG-UHFFFAOYSA-N isothiazole Chemical group C=1C=NSC=1 ZLTPDFXIESTBQG-UHFFFAOYSA-N 0.000 description 1
- GQYHUHYESMUTHG-UHFFFAOYSA-N lithium niobate Chemical compound [Li+].[O-][Nb](=O)=O GQYHUHYESMUTHG-UHFFFAOYSA-N 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- NCBZRJODKRCREW-UHFFFAOYSA-N m-anisidine Chemical compound COC1=CC=CC(N)=C1 NCBZRJODKRCREW-UHFFFAOYSA-N 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 125000002960 margaryl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 1
- 235000019796 monopotassium phosphate Nutrition 0.000 description 1
- 239000012452 mother liquor Substances 0.000 description 1
- 230000003183 myoelectrical effect Effects 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- SLPWFXHWNVMESS-UHFFFAOYSA-N n-butyl-3,5-dimethoxyaniline Chemical compound CCCCNC1=CC(OC)=CC(OC)=C1 SLPWFXHWNVMESS-UHFFFAOYSA-N 0.000 description 1
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 239000012454 non-polar solvent Substances 0.000 description 1
- 125000001196 nonadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229920000620 organic polymer Polymers 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 125000004043 oxo group Chemical group O=* 0.000 description 1
- 125000006503 p-nitrobenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1[N+]([O-])=O)C([H])([H])* 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002958 pentadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- XYFCBTPGUUZFHI-UHFFFAOYSA-O phosphonium Chemical compound [PH4+] XYFCBTPGUUZFHI-UHFFFAOYSA-O 0.000 description 1
- 125000001476 phosphono group Chemical group [H]OP(*)(=O)O[H] 0.000 description 1
- PJNZPQUBCPKICU-UHFFFAOYSA-N phosphoric acid;potassium Chemical compound [K].OP(O)(O)=O PJNZPQUBCPKICU-UHFFFAOYSA-N 0.000 description 1
- 125000004592 phthalazinyl group Chemical group C1(=NN=CC2=CC=CC=C12)* 0.000 description 1
- 238000001020 plasma etching Methods 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 125000005575 polycyclic aromatic hydrocarbon group Chemical group 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920000193 polymethacrylate Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-OUBTZVSYSA-N potassium-40 Chemical compound [40K] ZLMJMSJWJFRBEC-OUBTZVSYSA-N 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 125000001042 pteridinyl group Chemical group N1=C(N=CC2=NC=CN=C12)* 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- ZVJHJDDKYZXRJI-UHFFFAOYSA-N pyrroline Natural products C1CC=NC1 ZVJHJDDKYZXRJI-UHFFFAOYSA-N 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000213 sulfino group Chemical group [H]OS(*)=O 0.000 description 1
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 description 1
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 description 1
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 125000001981 tert-butyldimethylsilyl group Chemical group [H]C([H])([H])[Si]([H])(C([H])([H])[H])[*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- DZLFLBLQUQXARW-UHFFFAOYSA-N tetrabutylammonium Chemical compound CCCC[N+](CCCC)(CCCC)CCCC DZLFLBLQUQXARW-UHFFFAOYSA-N 0.000 description 1
- DPKBAXPHAYBPRL-UHFFFAOYSA-M tetrabutylazanium;iodide Chemical compound [I-].CCCC[N+](CCCC)(CCCC)CCCC DPKBAXPHAYBPRL-UHFFFAOYSA-M 0.000 description 1
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 1
- 125000003698 tetramethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 125000000464 thioxo group Chemical group S=* 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000002221 trityl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C([*])(C1=C(C(=C(C(=C1[H])[H])[H])[H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000002948 undecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B23/00—Methine or polymethine dyes, e.g. cyanine dyes
- C09B23/0066—Methine or polymethine dyes, e.g. cyanine dyes the polymethine chain being part of a carbocyclic ring,(e.g. benzene, naphtalene, cyclohexene, cyclobutenene-quadratic acid)
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B1/00—Optical elements characterised by the material of which they are made; Optical coatings for optical elements
- G02B1/04—Optical elements characterised by the material of which they are made; Optical coatings for optical elements made of organic materials, e.g. plastics
- G02B1/045—Light guides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C223/00—Compounds containing amino and —CHO groups bound to the same carbon skeleton
- C07C223/06—Compounds containing amino and —CHO groups bound to the same carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/26—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D307/30—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/34—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D307/56—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D307/68—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/22—Radicals substituted by doubly bound hetero atoms, or by two hetero atoms other than halogen singly bound to the same carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/06—Peri-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
- C07F7/1804—Compounds having Si-O-C linkages
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B23/00—Methine or polymethine dyes, e.g. cyanine dyes
- C09B23/0075—Methine or polymethine dyes, e.g. cyanine dyes the polymethine chain being part of an heterocyclic ring
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B69/00—Dyes not provided for by a single group of this subclass
- C09B69/008—Dyes containing a substituent, which contains a silicium atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B69/00—Dyes not provided for by a single group of this subclass
- C09B69/10—Polymeric dyes; Reaction products of dyes with monomers or with macromolecular compounds
- C09B69/107—Polymeric dyes; Reaction products of dyes with monomers or with macromolecular compounds containing an azomethine dye
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B1/00—Optical elements characterised by the material of which they are made; Optical coatings for optical elements
- G02B1/04—Optical elements characterised by the material of which they are made; Optical coatings for optical elements made of organic materials, e.g. plastics
- G02B1/045—Light guides
- G02B1/046—Light guides characterised by the core material
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B6/00—Light guides; Structural details of arrangements comprising light guides and other optical elements, e.g. couplings
- G02B6/10—Light guides; Structural details of arrangements comprising light guides and other optical elements, e.g. couplings of the optical waveguide type
- G02B6/105—Light guides; Structural details of arrangements comprising light guides and other optical elements, e.g. couplings of the optical waveguide type having optical polarisation effects
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/0009—Materials therefor
- G02F1/0063—Optical properties, e.g. absorption, reflection or birefringence
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/35—Non-linear optics
- G02F1/355—Non-linear optics characterised by the materials used
- G02F1/361—Organic materials
- G02F1/3611—Organic materials containing Nitrogen
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/35—Non-linear optics
- G02F1/355—Non-linear optics characterised by the materials used
- G02F1/361—Organic materials
- G02F1/3611—Organic materials containing Nitrogen
- G02F1/3612—Heterocycles having N as heteroatom
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/35—Non-linear optics
- G02F1/355—Non-linear optics characterised by the materials used
- G02F1/361—Organic materials
- G02F1/3613—Organic materials containing Sulfur
- G02F1/3614—Heterocycles having S as heteroatom
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/35—Non-linear optics
- G02F1/355—Non-linear optics characterised by the materials used
- G02F1/361—Organic materials
- G02F1/3615—Organic materials containing polymers
Definitions
- the present invention relates to a nonlinear optical compound, a nonlinear optical material including the nonlinear optical compound, and a nonlinear optical element.
- Nonlinear optical materials have been put to practical use as light control elements in optical communication equipment and laser equipment because the intensity and phase of light can be changed by an external field such as an electric field or a magnetic field.
- devices such as optical modulators, optical switches, and optical memories utilize the electro-optic effect of nonlinear optical materials.
- Non-Patent Documents 1 to 3 below more advanced nonlinear optical performance, Organic nonlinear optical materials have attracted attention and various studies have been conducted for their practical use in order to meet demands such as reduction in manufacturing costs and compounding with electronic integrated circuits.
- Nonlinear optical compound a compound having nonlinear optical activity
- a host material such as a polymer material.
- Nonlinear optical compounds exhibiting an electro-optic effect include an electron donor group (donor structure part D) and an electron acceptor group (acceptor structure part A) positioned at both ends of the molecular structure, and a ⁇ -conjugated chain ( ⁇ conjugate) connecting them.
- Push-pull type ⁇ -conjugated compounds having a bridge structure B) are known.
- Patent Document 1 below describes a nonlinear optical compound having a thiophene ring in the ⁇ -conjugated bridge structure B represented by the following formula.
- Patent Document 2 describes an approach to improve the performance of the nonlinear optical compound by adopting a predetermined acceptor structure part A.
- Non-Patent Document 4 describes an approach for improving the performance of a nonlinear optical compound by adopting a predetermined donor structure portion D.
- an alignment treatment of the nonlinear optical compound may be performed in order to generate a second-order nonlinear optical activity of the nonlinear optical material.
- An electric field poling method is generally used as a method of aligning the nonlinear optical compound.
- the electric field poling method is a method in which an electric field is applied to a nonlinear optical material, and the nonlinear optical compound is oriented in the applied electric field direction by the Coulomb force between the dipole moment of the nonlinear optical compound and the applied electric field.
- the electric field is usually applied in a state where the host material is heated to a temperature near the glass transition temperature of the host material and the molecular motion of the nonlinear optical compound is promoted. Therefore, in order to obtain a non-linear optical element that exhibits excellent non-linear optical performance, in addition to the non-linear optical compound having excellent non-linear optical characteristics, the non-linear optical compound must have heat resistance that is not altered by heating in the alignment treatment. Required.
- Electro-optical elements using non-linear optical materials are converted into electric signals and optical signals. It is being considered for use in the conversion of.
- the electronic circuit operating at high speed becomes high temperature, the molecular motion of the nonlinear optical compound becomes active and the orientation may be relaxed.
- the glass transition temperature of the host material is required to be higher, and accordingly, the nonlinear optical compound is also required to have heat resistance at a higher temperature.
- the present invention has been made in view of such circumstances, and by using the donor structure portion D that improves the nonlinear optical characteristics without significantly impairing the heat resistance, both the nonlinear optical characteristics and the heat resistance are sufficiently obtained.
- An object is to provide a nonlinear optical compound that can be achieved at a high level.
- Another object of the present invention is to provide a nonlinear optical material and a nonlinear optical element using the nonlinear optical compound.
- An object of the present invention is to provide a chromophore having nonlinear optical activity far superior to conventional chromophores and a nonlinear optical element including the chromophore.
- the present inventors have found that in the chromophore including the donor structure part D, the ⁇ -conjugated bridge structure part B, and the acceptor structure part A, the donor structure part D contains a substituted oxyaryl. As a result, the present inventors have found that the chromophore has a nonlinear optical activity that is far superior to that of the conventional chromophore.
- the present invention ⁇ 1>
- the donor structure D includes an aryl group substituted with a substituted oxy group
- the acceptor structure A is ⁇ A chromophore characterized by being free of SO 2-
- the substituted oxy group is bonded to a carbon atom at the ortho position of the aryl group, or is bonded to a carbon atom at the ortho position and the para position.
- the donor structure D is represented by the formula D-1 (here, At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a heterocycle containing a nitrogen atom as a heteroatom, the heterocycle having a substituent
- (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- a heterocycle containing a nitrogen atom as a heteroatom, and the heterocycle may have a substituent)
- the chromophore according to any one of the above ⁇ 1> to ⁇ 5>, represented by:
- the donor structure D is represented by the formula D-1-1 (here, R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent; R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a heterocycle containing a nitrogen atom as a heteroatom, the heterocycle having a substituent Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached. And a heterocycle containing a nitrogen atom as a heteroatom, and the heterocycle may have a substituent)
- the chromophore according to any one of the above items ⁇ 1> to ⁇ 6>, represented by:
- the donor structure D is represented by the formula D-1-2 (here, At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a saturated heterocyclic ring that may have a substituent, or R D 4 and R D D 5 , together with the nitrogen atom to which the nitrogen atom is bonded, the carbon atom of the aryl to which the nitrogen atom is bonded and the carbon atom of the aryl adjacent to the carbon atom, contains a nitrogen atom as a hetero atom and has a substituent.
- Form a heterocycle that may be The chromophore according to any one of the above items ⁇ 1> to ⁇ 6>, represented by:
- Donor structure D is represented by formula D-2 (here, At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 when R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, (1) R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent, in which case R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent, or (2) R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent. Also good) The chromophore according to any one of ⁇ 1>, ⁇ 2>, ⁇ 4> and ⁇ 5>, represented by:
- the donor structure D is represented by the formula D-2-1 (here, R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent; R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent. May form a ring that may have The chromophore according to any one of ⁇ 1>, ⁇ 2>, ⁇ 4>, ⁇ 5> and ⁇ 9>, represented by
- R A 1 and R A 2 may have a substituent together with the carbon atoms to which they are bonded.
- Carbon-carbon conjugate bridge structure B is (here, R B 1 to R B 8 each independently represents a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an alkenyl group, a cycloalkyl group, a cycloalkenyl group, a haloalkyl group, an aralkyl group, an aryloxy group or an aralkyloxy group.
- R B 1 to R B 8 each independently represents a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an alkenyl group, a cycloalkyl group, a cycloalkenyl group, a haloalkyl group, an aralkyl group, an aryloxy group or an aralkyloxy group.
- n represents an integer of 1 to 5
- m and m ′ each independently represents an integer of 0 to 3
- the chromophore according to any one of the above
- Carbon-carbon conjugated bridge structure B is represented by formula BI (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent; R B 1 and R B 2 are each independently a hydrogen atom, alkyl group, alkoxy group, aryl group, alkenyl group, cycloalkyl group, cycloalkenyl group, haloalkyl group, aralkyl group, aryloxy group or aralkyloxy group. Each having the same or different substituents)
- the chromophore according to any one of ⁇ 1> to ⁇ 12>, represented by:
- Formula I-1 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents. it may have, or R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 and R B 2 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and may have the same or different substituents.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- Formula I-1-1 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent;
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent;
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a heterocyclic ring that may have a substituent or (a) R D 2 and -NR D 4 R D 5 and (b) R D 3 and -NR D 4 R D 5 are each independently a heterocycle containing a nitrogen atom as a hetero atom together with the carbon atom to which it is bonded. And the heterocycle may have a substituent; R B 1 and R B 2 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and may have the same or different substituents.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- Formula I-2 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R B 1 and R B 2 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and may have the same or different substituents.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- the chromophore according to any one of ⁇ 1>, ⁇ 2>, ⁇ 4>, ⁇ 5>, ⁇ 9> and ⁇ 11> to ⁇ 13>, represented by:
- the carbon-carbon conjugated bridge structure B is represented by the formula B-II (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent;
- R B 1 , R B 2 , R B 3 and R B 4 are each independently a hydrogen atom, alkyl group, alkoxy group, aryl group, alkenyl group, cycloalkyl group, cycloalkenyl group, haloalkyl group, aralkyl group, An aryloxy group or an aralkyloxy group, each having the same or different substituents)
- the chromophore according to any one of ⁇ 1> to ⁇ 12>, represented by:
- Formula II-1 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl group of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents. it may have, or R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 , R B 2 , R B 3 and R B 4 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and are the same or different.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- the chromophore according to any one of ⁇ 1> to ⁇ 6>, ⁇ 11>, ⁇ 12> and ⁇ 17>,
- Formula II-1-1 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent;
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent;
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent.
- R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 , R B 2 , R B 3 and R B 4 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and are the same or different.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- Carbon-carbon conjugated bridge structure B is represented by formula B-III (here, m and m ′ each independently represents an integer of 0 to 3; R B 1 , R B 2 and R B 3 are each independently a hydrogen atom, alkyl group, alkoxy group, aryl group, alkenyl group, cycloalkyl group, cycloalkenyl group, haloalkyl group, aralkyl group, aryloxy group or An aralkyloxy group, which may have the same or different substituents)
- the chromophore according to any one of ⁇ 1> to ⁇ 12>, represented by:
- Formula III-1 (here, m and m ′ each independently represents an integer of 0 to 3; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl group of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents. it may have, or R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 , R B 2 and R B 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, each having the same or different substituent.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- Formula III-1-1 (here, m and m ′ each independently represents an integer of 0 to 3; R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent; R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to adjacent carbon atoms of the aryl group of the donor structure D, R D 2 and R D 3 together with the two adjacent carbon atoms are substituted.
- a ring which may have a group may be formed;
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 , R B 2 and R B 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, each having the same or different substituent.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- the chromophore according to any one of the above ⁇ 1> to ⁇ 7>, ⁇ 11>, ⁇ 12>, ⁇ 20> and ⁇ 21>,
- Carbon-carbon conjugated bridge structure B is represented by formula B-IV (Where n is an integer from 1 to 5) The chromophore according to any one of the above items ⁇ 1> to ⁇ 12>,
- n represents an integer of 1 to 5
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to adjacent carbon atoms of the aryl group of the donor structure D, R D 2 and R D 3 together with the two adjacent carbon atoms are substituted.
- a ring which may have a group may be formed;
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- Formula IV-1-b (Where n represents an integer of 1 to 5; R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent; R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to adjacent carbon atoms of the aryl group of the donor structure D, R D 2 and R D 3 together with the two adjacent carbon atoms are substituted.
- a ring which may have a group may be formed;
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- Formula IV-2-a (here, n represents an integer of 1 to 5; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- the chromophore according to any one of the above ⁇ 1>, ⁇ 2>, ⁇ 4>, ⁇ 5>, ⁇ 9>, ⁇ 11>, ⁇ 12> and ⁇ 23>,
- n represents an integer of 1 to 5;
- R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy
- Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- the chromophore according to any one of the above ⁇ 1>, ⁇ 2>, ⁇ 4>, ⁇ 5>, ⁇ 9>, ⁇ 11>, ⁇ 12> and ⁇ 23>,
- R D 1 represents a C1-6 alkoxy group, a benzyloxy group, a silyloxy group, a C2-6 alkenyloxy group, a C2-6 alkenylcarbonyloxy group, a C3-6 alkynyloxy group or a hydroxy group, May have;
- R D 2 and R D 3 are each independently a hydrogen atom, C1-6 alkoxy group, benzyloxy group, silyloxy group, C2-6 alkenyloxy group, C2-6 alkenylcarbonyloxy group, C3-6 alkynyloxy group Or ⁇ 6> to ⁇ 10>, ⁇ 14> to ⁇ 16>, ⁇ 18>, ⁇ 19>, ⁇ 21>, ⁇ 21>, which represent a hydroxy group and may have the same or different substituents. 22> and the chromophore according to any one of ⁇ 24> to ⁇ 27>,
- R D 4 and R D 5 each independently represent an alkyl group, a hydroxyalkyl group, or a silyloxyalkyl group, and each may have the same or different substituent, ⁇ 8>, ⁇ 14>, ⁇ 15>, ⁇ 18>, ⁇ 19>, ⁇ 21>, ⁇ 22>, ⁇ 24> and the chromophore according to ⁇ 25>,
- R A 1 and R A 2 each independently represent a methyl group, a trifluoromethyl group, or a phenyl group, and each may have the same or different substituent, ⁇ 11>, ⁇ 14 > To ⁇ 16>, ⁇ 18>, ⁇ 19>, ⁇ 21>, ⁇ 22>, ⁇ 24>, ⁇ 25>, ⁇ 26> and ⁇ 27>
- ⁇ 31> ⁇ 1 and ⁇ 2 are each independently The chromophore according to any one of the above ⁇ 13> to ⁇ 19>, which may be represented by the same or different substituents,
- ⁇ 32> ⁇ 1 and ⁇ 2 are the formulas The chromophore according to any one of the above ⁇ 13> to ⁇ 19>, which may be represented by the same or different substituents,
- ⁇ 40> formula The chromophore according to any one of ⁇ 1>, ⁇ 2>, ⁇ 4>, ⁇ 5>, ⁇ 9> to ⁇ 12>, ⁇ 26>, ⁇ 28> and ⁇ 30>, represented by: And ⁇ 41> formula The chromophore according to any one of ⁇ 1>, ⁇ 2>, ⁇ 4>, ⁇ 5>, ⁇ 9> to ⁇ 12>, ⁇ 27>, ⁇ 28> and ⁇ 30>, .
- the present invention also provides: ⁇ 42> A nonlinear optical material containing the chromophore according to any one of ⁇ 1> to ⁇ 41> above and a host material in which the chromophore is dispersed, and ⁇ 43> the host material is the color developing material
- the resin includes a resin having a reactive functional group capable of forming a covalent bond with the group, and at least a part of the chromophore is bonded to the resin.
- the present invention also provides: ⁇ 44> a nonlinear optical element having a film formed of the chromophore according to any one of ⁇ 1> to ⁇ 41> above or the nonlinear optical material according to ⁇ 42> or ⁇ 43> above, ⁇ 45> A nonlinear optical element having an optical waveguide formed of the chromophore according to any one of ⁇ 1> to ⁇ 41> or the nonlinear optical material according to ⁇ 42> or ⁇ 43>, and ⁇ 46 > It relates to a non-linear optical element comprising the chromophore according to any one of ⁇ 1> to ⁇ 41> above.
- the nonlinear optical compound according to the present invention includes the aryl group substituted with the substituted oxy group in the donor structure part D, and thus has the effect of improving the nonlinear optical characteristics without significantly impairing the heat resistance.
- an optical modulator using the electro-optic effect it is possible to drive the optical modulator with lower electric power, thereby saving energy and reducing the size.
- a large nonlinear optical effect changes the intensity and phase of light even in a weak electric field, it can be used for an electric field sensor for measuring a leakage electric field of an electronic integrated circuit, a terahertz electromagnetic wave sensor, and the like.
- it can also be used for signal transmission by light between electronic circuits by combining with electronic circuits (for example, “Low (Sub ⁇ ETH> 1-Volt) Halfwave Voltage Polymeric Electro-optic Modulators Achieved by Controlling Chromophore Shape. ”Y. Shi, et. Al., Science, encevol.288, .119 (2000)).
- the “donor structure part D” is not particularly limited as long as it includes an aryl group substituted with a substituted oxy group.
- the “substituted oxy group” means a structure in which a hydroxy group (—OH) or a hydrogen atom of the hydroxy group is substituted with a substituent.
- aryl group in the above “aryl group substituted with a substituted oxy group”, a monocyclic aromatic hydrocarbon group (hereinafter, referred to as monocyclic aryl group) or a polycyclic aromatic hydrocarbon group (hereinafter, referred to as “cyclic group”).
- monocyclic aryl group for example, preferably a C5 to 10 ring group, more preferably a C5 to 7 ring group, still more preferably a C5 to 6 ring group, and most preferably a C6 ring group (that is, a phenyl group) Group) and the like.
- C5-10 ring means that 5-10 carbon atoms form the ring, and so on.
- polycyclic aryl group include an aryl group condensed with two rings and an aryl group condensed with three rings.
- the aryl group condensed with two rings include preferably a C8 to 12 ring group, more preferably a C9 to 10 ring group, and most preferably a C10 ring group (that is, a naphthyl group).
- Examples of the “substituent” in the “substituted oxy group” include (1) an alkyl group which may have a substituent, (2) a haloalkyl group which may have a substituent, and (3) a substituent.
- alkyl group in the “(1) optionally substituted alkyl group”, a linear or branched C1-20 alkyl group and the like can be mentioned.
- C1-20 alkyl group means an alkyl group having 1-20 carbon atoms constituting the alkyl group, and the same applies hereinafter.
- Examples of the alkyl group include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, hexyl, and isohexyl.
- the alkyl group is preferably a C1-6 alkyl group.
- haloalkyl group in the above “(2) optionally substituted haloalkyl group” is at least one same or different halogen atom (for example, fluorine atom, chlorine atom, bromine atom, iodine atom, etc.) Examples thereof include a substituted linear or branched C1-20 alkyl group.
- the haloalkyl group is preferably a halo C1-6 alkyl group, for example.
- fluoromethyl group More preferably, for example, fluoromethyl group, difluoromethyl group, trifluoromethyl group, 2-fluoroethyl group, 1,2-difluoroethyl group, chloromethyl group, 2-chloroethyl group, 1,2-dichloroethyl group, Examples include bromomethyl group, 2-bromoethyl group, 1-bromopropyl group, 2-bromopropyl group, 3-bromopropyl group, iodomethyl group and the like.
- Examples of the “aryl group” in the “(3) aryl group optionally having substituents” include the same aryl groups as the “aryl group” in the substituted oxyaryl.
- Examples of the aryl group include a phenyl group and a naphthyl group.
- heteroaryl group in the “(4) optionally substituted heteroaryl group” is selected from the group consisting of (a) an oxygen atom, a nitrogen atom and a sulfur atom, the same or different.
- a 5-membered or 6-membered monocyclic aromatic heterocyclic group containing 1 or 2 or more, preferably 1 to 3 heteroatoms (b) the monocyclic aromatic heterocyclic group and an aryl group (in the substituted oxyaryl)
- a cyclic group etc. are mentioned.
- the heteroaryl group include pyrrolyl group, furyl group, thienyl group, imidazolyl group, pyrazolyl group, thiazolyl group, isothiazole group, oxazolyl group, triazolyl group, tetrazolyl group, oxadiazolyl group, thiadiazol group, pyridyl group, pyrazinyl group , Pyrimidinyl group, pyridazinyl group, triazinyl group, indolyl group, benzofuranyl group, indolyl group, benzofuranyl group, benzothienyl group, benzoimidazolyl group, benzoxazolyl group, benzoisoxazolyl group, benzothiadiazolyl group, benzoisothiyl group Asiazolyl group, indazolyl group, indazolyl group, prynyl group, quinoly
- the “aralkyl group” in the “(5) aralkyl group optionally having substituent (s)” includes an alkyl group substituted with at least one aryl group.
- Examples of the aryl group include the “aryl group” in the above substituted oxyaryl.
- Examples of the “alkyl group” include “alkyl group” in “(1) an optionally substituted alkyl group”.
- Examples of the aralkyl group include benzyl group, 1-phenylethyl group, phenethyl group, 1-naphthylmethyl group, 2-naphthylmethyl group, 1-naphthylethyl group, 2-naphthylethyl group and the like.
- alkenyl group examples include linear or branched C2-20 alkenyl groups.
- the “alkenyl group” is preferably, for example, a C2-6 alkenyl group. More preferably, for example, ethenyl group, 1-propenyl group, 2-propenyl group, 1-methylethenyl group, 1-butenyl group, 2-butenyl group, 3-butenyl group, 1-methyl-1-propenyl group, 1- Examples thereof include a methyl-2-propenyl group, a 2-methyl-1-propenyl group, and a 2-methyl-2-propenyl group.
- alkynyl group in the “(8) alkynyl group optionally having substituent (s)”, a linear or branched C3-20 alkynyl group and the like can be mentioned.
- the “alkynyl group” is preferably, for example, a C3-6 alkynyl group. More preferably, for example, 2-propynyl group, 1-methyl-2-propynyl group, 1,1-dimethyl-2-propynyl group, 2-butynyl group, 3-butynyl group, 1-pentynyl group, 2-pentynyl group, 3 -Pentynyl group, 4-pentynyl group and the like.
- acyl group in the “(9) acyl group optionally having substituent (s)”, a linear or branched C 1-20 acyl group and the like can be mentioned.
- the “acyl group” is preferably a C1-6 acyl group, for example. More preferably, for example, formyl group, acetyl group, propionyl group, butyryl group, isobutyryl group, valeryl group, isovaleryl group, pivaloyl group and the like can be mentioned.
- alkenyl in the “(10) optionally substituted alkenylcarbonyl group”
- alkenyl group in the “(7) optionally substituted alkenyl group” and the like. Is mentioned.
- the substituent may be one substituent, or two or more substituents that are the same or different.
- alkyl group examples include, for example, “(1) substituent "Alkyl group” in “optionally substituted alkyl group”, “haloalkyl group” in “(2) optionally substituted haloalkyl group”, “(3) optionally substituted” "Aryl group” in “aryl group”, “alkenyl group” in “(7) optionally substituted alkenyl group”, “alkynyl in” (8) optionally substituted alkynyl group " Group “,” (9) acyl group optionally having substituent ", and the like.
- substituted oxy group in the present invention, preferably, for example, (a) an optionally substituted alkoxy group, (b) an optionally substituted aryloxy group, (c) An aralkyloxy group which may have a substituent, (d) a silyloxy group which may have a substituent, (e) an alkenyloxy group which may have a substituent, and (f) a substituent.
- substituents examples thereof include an alkenylcarbonyloxy group which may have, (g) an alkynyloxy group which may have a substituent, and (h) a hydroxy group.
- alkoxy group” in the “(a) optionally substituted alkoxy group” includes, for example, a linear or branched C1-20 alkoxy group.
- Preferable examples include C1-6 alkoxy groups. More preferred examples include methoxy group, ethoxy group, propoxy group, isopropoxy group, butoxy group, isobutoxy group, sec-butoxy group, tert-butoxy group and the like.
- aryloxy group in the “(b) optionally substituted aryloxy group” include C5-10 monocyclic aryloxy group, C8-12 bicyclic aryloxy group and the like. Can be mentioned. Preferable examples include a phenoxy group and a naphthyloxy group, and more preferable examples include a phenoxy group.
- Examples of the “aralkyloxy group” in the “(c) optionally substituted aralkyloxy group” include, for example, the “aralkyl group” in the “(5) optionally substituted aralkyl group”.
- Group having an oxy group bonded thereto Preferable examples include benzyloxy group, phenethyloxy group, 1-naphthylmethoxy group, 2-naphthylmethoxy group and the like. More preferably, a benzyloxy group etc. are mentioned, for example.
- Preferred examples of the “(d) optionally substituted silyloxy group” include tert-butyldiphenylsiloxy group and tert-butyldimethylsiloxy group.
- alkenyloxy group in the “(e) optionally substituted alkenyloxy group” include linear or branched C2-20 alkenyloxy groups. Preferable examples include C2-6 alkenyloxy groups. More preferably, for example, ethenyloxy group, 1-propenyloxy group, 2-propenyloxy group, 1-methylethenyloxy group, 1-butenyloxy group, 2-butenyloxy group, 3-butenyloxy group, 1-methyl-1- Examples include a propenyloxy group, a 1-methyl-2-propenyloxy group, a 2-methyl-1-propenyloxy group, and a 2-methyl-2-propenyloxy group.
- alkenylcarbonyloxy group” in the “(f) optionally substituted alkenylcarbonyloxy group” includes, for example, a linear or branched C2-20 alkenylcarbonyloxy group. It is done. Preferable examples include C2-6 alkenylcarbonyloxy groups.
- ethenylcarbonyloxy group 1-propenylcarbonyloxy group, 2-propenylcarbonyloxy group, 1-methylethenylcarbonyloxy group, 1-butenylcarbonyloxy group, 2-butenylcarbonyloxy group 3-butenylcarbonyloxy group, 1-methyl-1-propenylcarbonyloxy group, 1-methyl-2-propenylcarbonyloxy group, 2-methyl-1-propenylcarbonyloxy group, 2-methyl-2-propenylcarbonyl An oxy group etc. are mentioned.
- alkynyloxy group in the “(g) optionally substituted alkynyloxy group” includes, for example, a linear or branched C2-20 alkynyloxy group. Preferable examples include C3-6 alkynyloxy groups. More preferably, for example, 2-propynyloxy group, 1-methyl-2-propynyloxy group, 1,1-dimethyl-2-propynyloxy group, 2-butynyloxy group, 3-butynyloxy group, 1-pentynyloxy group 2-pentynyloxy group, 3-pentynyloxy group, 4-pentynyloxy group and the like.
- Nyl group ”,“ (8) alkynyl group optionally having substituent ”,“ (9) acyl group optionally having substituent ”and“ (10) optionally having substituent ” “Substituent” in “good alkenylcarbonyl group” and the like can be mentioned.
- More preferable “substituted oxy group” in the present invention includes, for example, a methoxy group, an oxiranylmethoxy group, an isopropoxy group, a hydroxybutoxy group, a 3-bromo-2-hydroxypropoxy group, a benzyloxy group, and a methoxybenzyloxy group.
- a methoxy group an oxiranylmethoxy group
- an isopropoxy group a hydroxybutoxy group
- a 3-bromo-2-hydroxypropoxy group a benzyloxy group
- a benzyloxy group and a methoxybenzyloxy group.
- Tert-butyldiphenylsiloxy group tert-butyldimethylsiloxy group, 2-methyl-2-propoxy group, 1-methylethenylcarbonyloxy group, hydroxy group and the like.
- the substituted oxy group may be bonded to any of the ortho, meta and para carbon atoms of the aryl group, and the same or different substituted oxy groups are aryl groups. It may be bonded to a plurality of carbon atoms in the ortho, meta or / and para positions.
- the substituted oxy group is bonded to a carbon atom at the ortho position of the aryl group, or an embodiment in which it is bonded to two or more carbon atoms in the ortho and para positions is more preferred.
- the substituted oxy group in the donor structure part D is combined with the carbon atom of the aryl group to which the substituted oxy group is bonded and the carbon atom adjacent to the carbon atom to form an oxygen atom as a hetero atom.
- a heterocyclic ring containing may be formed.
- the heterocycle may have a substituent. Examples of the heterocyclic ring include a 5- to 7-membered heterocyclic ring, and may be an aliphatic ring or an aromatic ring.
- the aryl group in the donor structure part D is preferably further substituted with an amino group which may have a substituent.
- Examples of the “substituent” in the “amino group optionally having substituent (s)” include ( ⁇ ) alkyl group, ( ⁇ ) haloalkyl group, ( ⁇ ) hydroxyalkyl group, ( ⁇ ) acyloxyalkyl group, ( ⁇ ) silyloxyalkyl group, ( ⁇ ) aminoalkyl group, ( ⁇ ) aryl group and the like.
- the embodiment in which the substituent is substituted with an amino group may be mono-substituted or di-substituted, but di-substituted is preferable.
- ( ⁇ ) alkyl group as the “substituent” in the “amino group optionally having substituent (s)”, for example, the above “(1) alkyl group optionally having substituent (s)” And the like.
- Preferable examples include C1-6 alkyl groups, and more preferable examples include methyl group, ethyl group, propyl group, isopropyl group, butyl group, isobutyl group, sec-butyl group, tert-butyl group and the like. Can be mentioned.
- the “( ⁇ ) hydroxyalkyl group” as the “substituent” in the “amino group optionally having substituent (s)” is, for example, a linear or branched chain substituted with at least one hydroxy group C1-C20 alkyl group and the like.
- the hydroxyalkyl group include hydroxy C1-6 alkyl groups such as hydroxymethyl group, hydroxyethyl group, hydroxypropyl group, and hydroxybutyl group.
- the “( ⁇ ) acyloxyalkyl group” as the “substituent” in the “amino group optionally having substituent (s)” is, for example, linear or substituted with at least one same or different acyloxy group Examples thereof include a branched C1-20 alkyl group.
- the “( ⁇ ) silyloxyalkyl group” as the “substituent” in the “amino group optionally having substituent (s)” is, for example, a linear or branched group substituted with at least one silyloxy group Examples thereof include a chain-like C1-20 alkyl group.
- the “( ⁇ ) aminoalkyl group” as the “substituent” in the “amino group optionally having substituent (s)” is, for example, a linear or branched chain substituted with at least one amino group C1-C20 alkyl group and the like.
- the “( ⁇ ) aryl group” as the “substituent” in the “amino group optionally having substituent (s)” includes, for example, the “aryl group” in the substituted oxyaryl, preferably phenyl Group, naphthyl group and the like.
- the substituent may be combined with the nitrogen atom to be bonded to form a heterocycle containing a nitrogen atom as a hetero atom.
- the heterocycle may have a substituent.
- the heterocyclic ring include a 5- to 7-membered heterocyclic ring, and may be an aliphatic ring or an aromatic ring.
- the amino group having the substituent may form a heterocycle containing a nitrogen atom as a hetero atom together with the carbon atom of the aryl group to be bonded.
- the heterocycle may have a substituent.
- the heterocyclic ring include a 5- to 7-membered heterocyclic ring, and may be an aliphatic ring or an aromatic ring.
- each substituent may form a heterocycle containing a nitrogen atom as a heteroatom.
- a preferable donor structure part D is described below.
- preferable donor structure D include, for example, formula D-1 (here, At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and R D 1 , R D 2 and R D 3 may have the same or different substituents,
- R D 2 and R D 3 when R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl group of the donor structure D, (1) R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent, in which case R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyl
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a saturated heterocyclic ring that may have a substituent, or (a) R D 2 And -NR D 4 R D 5 , and (b) R D 3 and -NR D 4 R D 5 each independently, together with the carbon atom to which it is bonded, contains a nitrogen atom as a heteroatom and is substituted A heterocyclic ring optionally having a group is formed)
- the structural part etc. which are represented by are mentioned.
- R D 1 , R D 2 and R D 3 is independently an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group
- Alkynyloxy group or hydroxy group means (1) any one of R D 1 , R D 2 and R D 3 is an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenyl It may indicate a carbonyl group, an alkynyloxy group or a hydroxy group, (2) R D 1, 2 turn, each independently alkoxy group of R D 2 and R D 3, aryloxy group, aralkyloxy group, silyloxy Group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group May be a group or a hydroxy group, (3) R D 1, all R D 2 and R D
- the above-mentioned “respectively independently represents a hydrogen atom or an alkyl group” means an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group R D 1 , R D 2, and / or R D 3 which do not represent each independently represent a hydrogen atom or an alkyl group.
- the ⁇ alkynyloxy group '' for example, the ⁇ alkoxy group '' in the ⁇ (a) optionally substituted alkoxy group '' as the ⁇ substituted oxy group '' and ⁇ (b) a substituent '' ⁇ Aryloxy group '' in ⁇ optionally aryloxy group '', ⁇ aralkyloxy group '' in ⁇ (c) aralkyloxy group optionally having substituents '', ⁇ (d) having substituents ''
- examples of the “alkyl group” represented by R D 1 , R D 2 and R D 3 include the “alkyl group” in the above “(1) alkyl group optionally having substituent (s)”. Can be mentioned.
- R D 1 , R D 2 and R D 3 may each have the same or different substituents” means that R D 1 , R D 2 and R D 3 are independent of each other. It means that it may be substituted with one substituent or two or more identical or different substituents. Examples of the substituent include the above-mentioned “(1) optionally substituted alkyl group”, “(2) optionally substituted haloalkyl group”, and “(3) substituent.
- R D 2 and R D 3 are each bonded to adjacent carbon atoms of the aryl of the donor structure D, R D 2 and R D 3 together with the two adjacent carbon atoms are substituted.
- a ring which may have a group may be formed.
- R D 1 has an alkoxy group which may have a substituent, an aryloxy group which may have a substituent, an aralkyloxy group which may have a substituent, and a substituent.
- R D 2 and R D 3 are formed together with two adjacent carbon atoms, and the ring which may have a substituent may be an aliphatic or aromatic carbocycle, and may be aliphatic or An aromatic heterocyclic ring may be used.
- R represents an alkyl group, an aryl group, an aralkyl group, a silyl group, an alkenyl group, an alkenylcarbonyl group, an alkynyl group or a hydrogen atom, and may have a substituent;
- R D 4 and R D 5 Has the same meaning as defined in Formula D-1 above) and the like, and is also included in the present invention.
- R D 2 and R D 3 are each bonded to adjacent carbon atoms of the aryl of the donor structure D
- R D 2 and R D 3 together with the two adjacent carbon atoms are heterogeneous.
- R represents an alkyl group, an aryl group, an aralkyl group, a silyl group, an alkenyl group, an alkenylcarbonyl group, an alkynyl group or a hydrogen atom, and may have a substituent;
- R D 4 and R D 5 Has the same meaning as defined in Formula D-1 above
- alkyl group As “alkyl group”, “haloalkyl group”, “hydroxyalkyl group”, “acyloxyalkyl group”, “silyloxyalkyl group”, “aminoalkyl group” or “aryl group” represented by R D 4 and R D 5
- alkyl group As “alkyl group”, “haloalkyl group”, “hydroxyalkyl group”, “acyloxyalkyl group”, “silyloxyalkyl group”, “aminoalkyl group” or “aryl group” represented by R D 4 and R D 5
- alkyl group As “alkyl group”, “haloalkyl group”, “hydroxyalkyl group”, “acyloxyalkyl group”, “silyloxyalkyl group”, “aminoalkyl group” or “aryl group” represented by R D 4 and R D 5
- alkyl group As “alkyl group”, “haloalkyl group”, “hydroxyalkyl group”, “acyloxyalkyl group”, “silyloxyal
- R D 4 and R D 5 may be combined with the nitrogen atom to be bonded to form a heterocyclic ring which may have a substituent.
- the heterocycle may have a substituent.
- the heterocyclic ring include a 5- to 7-membered heterocyclic ring, and may be an aliphatic ring or an aromatic ring.
- R D 2 and —NR D 4 R D 5 and (b) R D 3 and —NR D 4 R D 5, independently of each other, together with the carbon atoms to which they are bonded,
- a heterocycle containing a nitrogen atom as a heteroatom may be formed.
- the heterocycle may have a substituent.
- the heterocyclic ring include a 5- to 7-membered heterocyclic ring, and may be an aliphatic ring or an aromatic ring.
- R D 1 , R D 4 and R D 5 have the same meaning as defined in Formula D-1 above
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a saturated heterocyclic ring that may have a substituent, or (a) R D 2 And -NR D 4 R D 5 , and (b) R D 3 and -NR D 4 R D 5 each independently, together with the carbon atom to which it is bonded, contains a nitrogen atom as a heteroatom and is substituted A heterocyclic ring optionally having a group is formed) Etc.
- R D 1 , R D 2 and R D 3 are each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy
- R D 1 , R D 2 and R D 3 may have the same or different substituents
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent, in which case R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbon
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a saturated heterocyclic ring that may have a substituent, or R D 4 and R D D 5 , together with the nitrogen atom to which the nitrogen atom is bonded, the carbon atom of the aryl to which the nitrogen atom is bonded and the carbon atom of the aryl adjacent to the carbon atom, contains a nitrogen atom as a hetero atom and has a substituent.
- the donor structure D is defined as “R D 4 and R D 5 are combined with the nitrogen atom to be bonded, the aryl carbon atom to which the nitrogen atom is bonded, and the aryl carbon atom adjacent to the carbon atom.
- R D 4 and R D 5 are combined with the nitrogen atom to be bonded, the aryl carbon atom to which the nitrogen atom is bonded, and the aryl carbon atom adjacent to the carbon atom.
- R D 1 , R D 2 and R D 3 are each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy
- R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy
- Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent. Also good)
- the structure etc. which are represented by these are mentioned.
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent. May form a ring that may have The structure etc. which are represented by these are mentioned.
- R D 1 , R D 2 and R D 3 is a C1-6 alkoxy group (for example, methoxy group, ethoxy group, propoxy group, isopropoxy group).
- R D 4 and R D 5 are each independently an alkyl group (for example, a C1-6 alkyl group such as a methyl group, an ethyl group, a propyl group, or a butyl group).
- Etc. hydroxyalkyl groups
- hydroxy C1-6 alkyl groups such as hydroxymethyl group, hydroxyethyl group, hydroxypropyl group, hydroxybutyl group, etc.
- silyloxyalkyl groups each being the same or different substituent
- the aspect etc. which may have are further more preferable.
- the acceptor structure A in the present invention is not particularly limited as long as it is an electron accepting group and does not contain —SO 2 —.
- Examples of the “alkyl group” represented by R A 1 and R A 2 include the “alkyl group” in the above “(1) alkyl group optionally having substituent (s)”. Preferred examples of the alkyl group include a methyl group and an isopropyl group. Examples of the “alkenyl group” represented by R A 1 and R A 2 include the “alkenyl group” in the above “(7) alkenyl group optionally having substituent (s)”. Examples of the “cycloalkyl group” represented by R A 1 and R A 2 include C3-15 monocyclic or polycyclic saturated aliphatic cyclic groups.
- Examples of the cycloalkyl group include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclooctyl group, a cyclononyl group, a cyclodecyl group, a cycloundecyl group, a cyclododecyl group, and the like.
- a cyclohexyl group etc. are mentioned.
- Examples of the “cycloalkenyl group” represented by R A 1 and R A 2 include C3-15 monocyclic or polycyclic unsaturated aliphatic cyclic groups.
- Examples of the cycloalkenyl group include a cyclopropenyl group, a cyclopentenyl group, a cyclohexenyl group, a cycloheptynyl group, a cyclooctenyl group, a cyclopentadienyl group, a cyclohexadienyl group, a cycloheptadienyl group, and a cyclooctadienyl group.
- Examples of the “alkoxy group” represented by R A 1 and R A 2 include the “alkoxy group” in the above-mentioned “(a) alkoxy group optionally having substituent (s)”.
- alkoxy group examples include methoxy group, ethoxy group, propoxy group, isopropoxy group, butoxy group, isobutoxy group, sec-butoxy group, tert-butoxy group and the like.
- haloalkyl group represented by R A 1 and R A 2 include the “haloalkyl group” in the above “(2) haloalkyl group optionally having substituent (s)”.
- haloalkyl group examples include a fluoromethyl group, a difluoromethyl group, a trifluoromethyl group, a 2-fluoroethyl group, a 1,2-difluoroethyl group, a chloromethyl, a 2-chloroethyl group, and a 1,2-dichloroethyl group.
- the “aryl group” represented by R A 1 and R A 2 include a phenyl group and a naphthyl group, and a phenyl group is preferable.
- Another specific embodiment of the preferred acceptor structure A includes, for example, the formula The structure etc. which are represented by these are mentioned.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, an alkenyl group, a cycloalkyl group, a cycloalkenyl group, an alkoxy group, a haloalkyl group or an aryl group, each having the same or different substituent. May be)
- the structure etc. which are represented by these are mentioned.
- acceptor structure A for example, a compound represented by the formula Aa-1 (Here, R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, or an aryl group, and each may have the same or different substituent)
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, or an aryl group, and each may have the same or different substituent
- R A 1 and R A 2 in the above specific embodiment of the acceptor structure part A of the chromophore according to the present invention may have a substituent together with the carbon atom to which they are bonded. You may form the structure represented by these. That is, for example, the formula (Wherein, Y is, -CR A 1 R A 2 - , - O -, - S -, - SO -, - SiR A 1 R A 2 -, - NR- ( wherein, R represents a hydrogen atom or an alkyl
- the ⁇ -conjugated bridge structure B in the present invention has a conjugated system having conjugated multiple bonds, and ⁇ electrons are not transferred from the donor structure D to the acceptor structure A through an electron orbit in the bridge (bridge). There is no particular limitation as long as it is localized.
- the conjugated multiple bond of the ⁇ -conjugated bridge structure part B may be either a double bond or a triple bond, preferably a double bond.
- Examples of the ⁇ conjugate include a carbon-carbon conjugate and a conjugate containing nitrogen.
- a preferred conjugate in the chromophore of the present invention is a carbon-carbon conjugate.
- R B 1 to R B 8 each independently represents a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an alkenyl group, a cycloalkyl group, a cycloalkenyl group, a haloalkyl group, an aralkyl group, an aryloxy group or an aralkyloxy group.
- R B 1 to R B 8 each independently represents a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an alkenyl group, a cycloalkyl group, a cycloalkenyl group, a haloalkyl group, an aralkyl group, an aryloxy group or an aralkyloxy group.
- n represents an integer of 1 to 5
- m and m ′ each independently represents an integer of 0 to 3
- alkyl group represented by R B 1 to R B 8 include “alkyl group” in the above “(1) alkyl group optionally having substituent (s)”.
- the alkyl group is preferably, for example, methyl group, ethyl group, propyl group, isopropyl group, n-butyl group, isobutyl group, sec-butyl group, tert-butyl group, pentyl group, isopentyl group, hexyl group, An isohexyl group, a heptyl group, etc. are mentioned.
- Examples of the “alkoxy group” represented by R B 1 to R B 8 include the “alkoxy group” in the above-mentioned “(a) alkoxy group optionally having substituent (s)”.
- Examples of the alkoxy group include a methoxy group, an ethoxy group, a propoxy group, an isopropoxy group, a butoxy group, an isobutoxy group, a sec-butoxy group, and a tert-butoxy group, preferably a methoxy group.
- Examples of the “aryl group” represented by R B 1 to R B 8 include a phenyl group and a naphthyl group, and preferably include a phenyl group.
- Examples of the “alkenyl group” represented by R B 1 to R B 8 include a linear or branched C2-20 alkenyl group.
- Examples of the alkenyl group include an ethenyl group and a propenyl group. Group, butenyl group, pentenyl group, hexenyl group and the like.
- Examples of the “cycloalkyl group” represented by R B 1 to R B 8 include C3-15 monocyclic or polycyclic saturated aliphatic cyclic groups.
- Examples of the cycloalkyl group include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclooctyl group, a cyclononyl group, a cyclodecyl group, a cycloundecyl group, a cyclododecyl group, and the like.
- a cyclohexyl group etc. are mentioned.
- Examples of the “cycloalkenyl group” represented by R B 1 to R B 8 include C3-15 monocyclic or polycyclic unsaturated aliphatic cyclic groups.
- Examples of the cycloalkenyl group include a cyclopropenyl group, a cyclopentenyl group, a cyclohexenyl group, a cycloheptynyl group, a cyclooctenyl group, a cyclopentadienyl group, a cyclohexadienyl group, a cycloheptadienyl group, and a cyclooctadienyl group.
- Examples of the “haloalkyl group” represented by R B 1 to R B 8 include the “haloalkyl group” in the above “(2) haloalkyl group optionally having substituent (s)”.
- haloalkyl group examples include a fluoromethyl group, a difluoromethyl group, a trifluoromethyl group, a 2-fluoroethyl group, a 1,2-difluoroethyl group, a chloromethyl, a 2-chloroethyl group, and a 1,2-dichloroethyl group.
- Examples of the “aralkyl group” represented by R B 1 to R B 8 include the “aralkyl group” in the above “(5) aralkyl group optionally having substituent (s)”.
- Examples of the aralkyl group include benzyl group, 1-phenylethyl group, phenethyl group, 1-naphthylmethyl group, 2-naphthylmethyl group, 1-naphthylethyl group, 2-naphthylethyl group, and the like. , A benzyl group and the like.
- Examples of the “aryloxy group” represented by R B 1 to R B 8 include the “aryloxy group” in the above “(b) aryloxy group optionally having substituent (s)”.
- Examples of the aryloxy group include a phenoxy group and a naphthyloxy group, preferably a phenoxy group.
- Examples of the “aralkyloxy group” represented by R B 1 to R B 8 include the “aralkyloxy group” in the above “(c) aralkyloxy group optionally having substituent (s)”.
- Examples of the aralkyloxy group include a benzyloxy group, a phenethyloxy group, a 1-naphthylmethoxy group, and a 2-naphthylmethoxy group, and a benzyloxy group is preferable.
- R B 1 and R B 2 are each independently a hydrogen atom, alkyl group, alkoxy group, aryl group, alkenyl group, cycloalkyl group, cycloalkenyl group, haloalkyl group, aralkyl group, aryloxy group or aralkyloxy group.
- R B 9 and R B 10 each independently represent a hydrogen atom or a cyano group (—CN); m represents an integer of 0 to 3)
- m represents an integer of 0 to 3
- the formula BI (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent;
- R B 1 and R B 2 are each independently a hydrogen atom, alkyl group, alkoxy group, aryl group, alkenyl group, cycloalkyl group, cycloalkenyl group, haloalkyl group, aralkyl group, aryloxy group or aralkyloxy group.
- formula B-II (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent;
- R B 1 , R B 2 , R B 3 and R B 4 are each independently a hydrogen atom, alkyl group, alkoxy group, aryl group, alkenyl group, cycloalkyl group, cycloalkenyl group, haloalkyl group, aralkyl group, An aryloxy group or an aralkyloxy group, each of which may have the same or different substituents),
- formula B-III (here, m and m ′ each independently represents an integer of 0 to 3;
- R B 1 , R B 2 and R B 3 are each independently a hydrogen atom, alkyl group, alkoxy group, aryl group, alkenyl group, cycloalkyl group, cycloalkenyl group, haloalkyl
- the “same or different carbon-carbon conjugated ⁇ bond” is preferably represented by, for example, the formula And more preferably, a structure represented by the formula The structure represented by etc. is mentioned.
- the chromophore according to the present invention has, for example, the formula DBA (where D represents the donor structure D; B represents the ⁇ -conjugated bridge structure B; A represents the acceptor structure A), etc. It is represented by
- Preferred specific embodiments of the chromophore according to the present invention include, for example, formula I-1 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents. it may have, or R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 and R B 2 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and may have the same or different substituents.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- a compound of formula I-1-a (here, m and m ′ each independently represents an integer of 0 to 3; R D 1 to R D 5 , R B 1 , R B 2 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula I-1 above)
- a compound of formula I-1-b (Wherein R D 1 to R D 5 , R B 1 , R B 2 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula I-1)
- a compound of formula I-2 (here, At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alky
- R D 2 and R D 3 when R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, (1) R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent, in which case R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent, or (2) R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent. May be; ⁇ 1 , ⁇ 2 , R B 1 , R B 2 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula I-1) The compound etc. which are represented by these are mentioned.
- More preferred specific embodiments of the chromophore according to the present invention include, for example, formula I-1-1 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent;
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent;
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, (1) R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent, in which case R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent, or (2) R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a saturated heterocyclic ring that may have a substituent or (a) R D 2 And -NR D 4 R D 5 and (b) R D 3 and -NR D 4 R D 5 are each independently a heterocycle containing a nitrogen atom as a hetero atom, together with the carbon atom to which it is attached And the heterocycle may have a substituent; R B 1 and R B 2 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and may have the same or different substituents.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- a compound of formula I-1-1-a (here, m and m ′ each independently represents an integer of 0 to 3; R D 1 to R D 5 , R B 1 , R B 2 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula I-1-1)
- a compound of formula I-1-1-b (Wherein R D 1 to R D 5 , R B 1 , R B 2 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula I-1-1) The compound etc. which are represented by these are mentioned.
- chromophore includes, for example, formula II-1 (here, ⁇ 1 and ⁇ 2 each independently represent the same or different carbon-carbon conjugated ⁇ bond, and each may have the same or different substituent; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents. it may have, or R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 , R B 2 , R B 3 and R B 4 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and are the same or different.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- a compound of formula II-1-a here, m and m ′ each independently represents an integer of 0 to 3; R D 1 to R D 5 , R B 1 to R B 4 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula II-1)
- a compound of formula II-1-b (Here, R D 1 to R D 5 , R B 1 to R B 4 , R A 1 and R A 2 have the same meaning as defined in the compound represented by Formula II-1 above) And more preferably a compound represented by formula II-1-1.
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent.
- R D 4 and R D 5 together with the nitrogen atom to which they are bonded form a saturated heterocyclic ring that may have a substituent, or (a) R D 2 And -NR D 4 R D 5 , and (b) R D 3 and -NR D 4 R D 5 are each independently, together with the carbon atom to which they are attached, containing a nitrogen atom as a heteroatom, substituted Forming an optionally substituted heterocyclic ring; R B 1 , R B 2 , R B 3 and R B 4 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and are the same or different.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent
- a compound of formula II-1-1-a here, m and m ′ each independently represents an integer of 0 to 3; R D 1 to R D 5 , R B 1 to R B 4 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula II-1-1)
- a compound of formula II-1-1-b (Wherein R D 1 to R D 5 , R B 1 to R B 4 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula II-1-1) The compound etc. which are represented by these are mentioned.
- Preferred specific embodiments of the chromophore according to the present invention include, for example, formula III-1 (here, m and m ′ each independently represents an integer of 0 to 3; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- formula III-1 here, m and m ′ each independently represents an integer of 0 to 3; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy
- Each other group independently represents
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents. it may have, or R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 , R B 2 and R B 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, each having the same or different substituent.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- a compound of formula III-1-a (Wherein R D 1 to R D 5 , R B 1 to R B 3 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the above formula III-1) And more preferably a compound represented by formula III-1-1.
- R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group or a hydroxy group, and may have a substituent
- R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, or a hydroxy group, May have different substituents,
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D, R D 2 and R D 3 are combined with the two adjacent carbon atoms to form a substituent.
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R B 1 , R B 2 , R B 3 and R B 4 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryl group, an aralkyl group, an aryloxy group or an aralkyloxy group, and are the same or different.
- R A 1 and R A 2 each independently represents a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituents
- a compound represented by the formula III-1 -1-a (Here, R D 1 to R D 5 , R B 1 to R B 3 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the above formula III-1-1) The compound etc. which are represented by these are mentioned.
- chromophore includes, for example, formula IV-1-a (here, n represents an integer of 1 to 5; R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, and may have a substituent; R D 2 and R D 3 each independently represent a hydrogen atom, an alkyl group, an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group.
- formula IV-1-a here, n represents an integer of 1 to 5; R D 1 represents an alkoxy group, an aryloxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an
- R D 2 and R D 3 are each bonded to adjacent carbon atoms of the aryl group of the donor structure D, R D 2 and R D 3 together with the two adjacent carbon atoms are substituted.
- a ring which may have a group may be formed;
- R D 4 and R D 5 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group, a hydroxyalkyl group, an acyloxyalkyl group, a silyloxyalkyl group, an aminoalkyl group, or an aryl group, and the same or different substituents.
- R D 4 and R D 5 together with the nitrogen atom to which they are attached a heterocyclic ring containing a nitrogen atom to form a hetero atom, said heterocyclic is substituted Or (a) R D 2 and —NR D 4 R D 5 , and (b) R D 3 and —NR D 4 R D 5 are each independently combined with the carbon atom to which they are attached.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- a compound of formula IV-1-b (Here, n, R D 1 to R D 5 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the above formula IV-1-a)
- a compound of formula IV-2-a (here, n represents an integer of 1 to 5; At least one of R D 1 , R D 2 and R D 3 is each independently an alkoxy group, aryloxy group, aralkyloxy group, silyloxy group, alkenyloxy group, alkenylcarbonyloxy group, alkynyloxy group or hydroxy
- Each other group independently represents a hydrogen atom or an alkyl group, and each may have the same or different substituents.
- R D 2 and R D 3 are each bonded to the adjacent carbon atom of the aryl of the donor structure D
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a ring which may have a substituent
- R D 1 is an alkoxy group, aryl An oxy group, an aralkyloxy group, a silyloxy group, an alkenyloxy group, an alkenylcarbonyloxy group, an alkynyloxy group or a hydroxy group, which may have a substituent
- R D 2 and R D 3 may be combined with two adjacent carbon atoms to form a heterocycle containing an oxygen atom as a heteroatom, and the heterocycle has a substituent.
- R A 1 and R A 2 each independently represent a hydrogen atom, an alkyl group, a haloalkyl group or an aryl group, and each may have the same or different substituent)
- a compound of formula IV-2-b (Here, n, R D 1 to R D 3 , R A 1 and R A 2 have the same meaning as defined in the compound represented by the formula IV-2-a) The compound etc. which are represented by these are mentioned.
- the chromophore according to the present invention can be produced by a method known per se. For example, Ann., 580, 44 (1953), Angew.Chem., 92, 671 (1980), Chem. Ber., 95, 581 (1962), Macromolecules, 2001, 34, 253, Chem. Mater., 2007 , 19, 1154, Org. Synth., VI, 901 (1980), Chem. Mater., 2002, 14, 2393, J. Mater. Sci., 39, 2335 (2004), “Preparative Organic Chemistry”, John Wiley (1975), p.217, J. Org. Chem., 42, 353 (1977), J. Org. Chem., 33, 3382 (1968), Synthesis, 1981, 165, etc.
- the chromophore according to the present invention can be produced by various methods such as a method improved as appropriate, a method combining these methods, and a method described in the Examples below.
- the chromophore according to the present invention can be produced by the production method shown below.
- the method for producing the chromophore of the present invention is not limited to these reaction examples.
- the formula I-1-b (Wherein R D 1 to R D 5 , R B 1 and R B 2 and R A 1 and R A 2 have the same meaning as defined in the chromophore represented by the formula I-1-b)
- a compound represented by the formula V (Wherein R A 1 and R A 2 have the same meaning as defined in the chromophore represented by the formula I-1-b above) to react with the color represented by the formula I-1-b A group can be manufactured.
- the reaction of the compound represented by Formula IV-I and the compound represented by Formula V is usually performed in a polar solvent.
- the polar solvent may be a protic polar solvent or an aprotic polar solvent, or a polar solvent obtained by mixing them.
- the protic polar solvent include water, methanol, ethanol, 1-propanol, 2-propanol, 1-butanol, acetic acid, formic acid, and the like, or a mixture thereof.
- reaction temperature is usually about 0 ° C to about 150 ° C, preferably about 20 ° C to about 80 ° C.
- reaction time is affected by the reaction temperature, it is usually about 1 hour to about 3 days.
- the reaction atmosphere is usually performed in the air or the like.
- Formula IV-I which is a raw material for producing the compound represented by Formula I-1-b (Wherein R D 1 to R D 5 and R B 1 and R B 2 have the same meaning as defined in the chromophore represented by the above formula I-1-b), for example, It can be manufactured by the method described in the above.
- R D 1 to R D 5 have the same meaning as defined in the chromophore represented by the formula I-1-b above, and the formula VII-I-1 (Wherein R B 1 and R B 2 have the same meaning as defined in the chromophore represented by the formula I-1-b; Ar represents an aryl group; X represents a halogen atom), or Formula VII-I-2 (Wherein R B 1 and R B 2 have the same meaning as defined in the chromophore represented by the formula I-1-b; R represents an alkyl group)
- an organic solvent for example, tetrahydrofuran or the like
- R D 1 to R D 5 have the same meaning as defined in the chromophore represented by the formula I-1-b above, and the formula VII-I-3 (Wherein R B 1 , R B 2 and R B 3 have the same meaning as defined in the chromophore represented by the formula I-1-b; Ar represents an aryl group; X represents a halogen atom; Y represents a protecting group for a hydrogen atom or a hydroxy group) and a compound represented by formula VIII-I-2 (Wherein R D 1 to R D 5 and R B 1 and R B 2 have the same meaning as defined in the chromophore represented by the above formula I-1-b; Y represents a hydrogen atom or a hydroxy group Is obtained by subjecting it to an oxidation reaction, and when Y represents a protective group for a hydroxy group, by subjecting it to an oxidation reaction after the deprotection reaction of the protective group for the hydroxy group.
- a compound represented by the formula IV wherein R D 1 to
- Examples of the aryl group of the “aryl group optionally having substituent (s)” represented by Ar include a phenyl group and the like.
- Examples of the “halogen atom” represented by X include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom.
- the “protecting group for hydroxy group” represented by Y is not particularly limited as long as it has a function as a protecting group.
- an alkyl group for example, methyl group, ethyl group, isopropyl group, tert-butyl group) Group
- alkylsilyl group eg, trimethylsilyl group, tert-butyldimethylsilyl group, etc.
- alkoxymethyl group eg, methoxymethyl group, 2-methoxyethoxymethyl group, etc.
- tetrahydropyranyl group triethylsilylethoxymethyl Group
- aralkyl group for example, benzyl group, p-methoxybenzyl group, 2,3-dimethoxybenzyl group, o-nitrobenzyl group, p-nitrobenzyl group, trityl group, etc.
- acyl group for example, formyl group, acetyl group) Group, etc.
- a method known per se can be used as a deprotection reaction method. For example, it can be carried out according to the method described in the literature such as T.W. Greene, “Protective” Groups “Organic” Synthesis, “John Wiley” & “Sons,” 1981, or the like.
- a chromophore represented by formula IV-II Wherein R D 1 to R D 5 and R B 1 to R B 4 have the same meaning as defined in the chromophore represented by the formula II-1-b, and a compound represented by the formula V (Wherein R A 1 and R A 2 have the same meaning as defined in the chromophore represented by the above formula II-1-b), by reacting with a compound represented by the formula II-1-b
- the chromophore represented by can be produced.
- the reaction between the compound represented by Formula IV-II and the compound represented by Formula V is usually carried out under the conditions used for the reaction between the compound represented by Formula IV-I and the compound represented by Formula V. it can.
- the compound represented by the formula II-1-b can be obtained by purifying the crude product obtained by carrying out ordinary treatment according to a usual method or without purification.
- Formula IV-II which is a raw material for producing the compound represented by Formula II-1-b Where R D 1 to R D 5 and R B 1 to R B 4 have the same meaning as defined in the chromophore represented by the formula II-1-b above. It can be manufactured by the method described in the above.
- the formula VI (Wherein R D 1 to R D 5 have the same meaning as defined in the chromophore represented by the formula II-1-b), and a compound represented by the formula VII-II-1 (Wherein R B 1 to R B 4 have the same meaning as defined in the chromophore represented by the above formula II-1-b; Ar represents an aryl group; X represents a halogen atom; Y represents Represents a protecting group for a hydrogen atom or a hydroxy group), or formula VII-II-2 (Wherein R B 1 to R B 4 have the same meaning as defined in the chromophore represented by the formula II-1-b above; R represents an alkyl group)
- formula VIII-II (Wherein R D 1 to R D 5 and R B 1 to R B 4 have the same meaning as defined in the chromophore represented by the formula II-1-b above; Y is a hydrogen atom or a hydroxy group
- the formula IX-1 (Wherein R B 1 to R B 4 , R A 1 and R A 2 have the same meaning as defined in the chromophore represented by the above formula II-1-b; Ar represents an aryl group; X Represents a halogen atom), or a compound of formula IX-2 (Wherein R B 1 to R B 4 , R A 1 and R A 2 have the same meaning as defined in the chromophore represented by Formula II-1-b above; R represents an alkyl group)
- a compound of formula VI (Wherein R D 1 to R D 5 have the same meaning as defined in the chromophore represented by the above formula II-1-b), and by reacting them in the presence of a base, A chromophore represented by Formula II-1-b can be prepared.
- the reaction of the compound represented by Formula IX-1 or the compound represented by Formula IX-2 with the compound represented by Formula VI is usually carried out in a polar solvent or a nonpolar solvent by using an alkali metal alkoxide, an alkali metal hydride, n-
- the reaction is performed in the presence of a base such as butyl lithium or phenyl lithium.
- the reaction temperature is usually about ⁇ 30 ° C. to about 100 ° C., preferably about ⁇ 10 ° C. to about 50 ° C.
- the reaction time is usually from several minutes to about 5 hours, etc., but is appropriately determined while confirming the progress of the reaction by thin layer chromatography or the like.
- the reaction atmosphere is usually preferably carried out under an inert gas such as nitrogen or argon under moisture prevention.
- an inert gas such as nitrogen or argon under moisture prevention.
- Formula IX-1 which is a raw material for producing a compound represented by Formula II-1-b (Wherein R B 1 to R B 4 , R A 1 and R A 2 have the same meaning as defined in the chromophore represented by the above formula II-1-b; Ar represents an aryl group; X Can be produced by, for example, the following method.
- the chromophore represented by Formula IV-III wherein R D 1 to R D 5 and R B 1 to R B 3 have the same meaning as defined in the chromophore represented by the above formula III-1-a, and the formula V (Wherein R A 1 and R A 2 have the same meaning as defined in the chromophore represented by the above formula III-1-a), by reacting the compound represented by the formula III-1-a
- the chromophore represented by can be produced.
- the above reaction between the compound of formula IV-III and the compound of formula V is usually carried out in a polar solvent.
- the reaction temperature is usually about 0 ° C to about 150 ° C, preferably about 20 ° C to about 80 ° C.
- the reaction time is affected by the reaction temperature, it is usually about 1 hour to about 3 days.
- the reaction atmosphere is usually performed in the air.
- the compound represented by the formula III-1-a can be obtained by purifying the crude product obtained by carrying out the usual treatment according to the usual method or without purification.
- Formula IV-III which is a raw material for producing a compound represented by Formula III-1-a (Wherein R D 1 to R D 5 and R B 1 to R B 3 have the same meaning as defined in the chromophore represented by the above formula III-1-a), for example, It can be manufactured by the method described in the above.
- the formula VI Wherein R D 1 to R D 5 have the same meaning as defined in the chromophore represented by the above formula III-1-a
- the formula XIII (Wherein R B 1 to R B 3 have the same meaning as defined in the chromophore represented by the above formula III-1-a)
- the compound represented by the formula XIV obtained by reacting Wherein R D 1 to R D 5 and R B 1 to R B 3 have the same meaning as defined in the chromophore represented by the above formula III-1-a
- C A compound represented by the formula IV-III can be produced by reacting with alkylideneimine such as 6 H 11 N ⁇ CHCH 2 Li and then treating with water.
- a cis isomer or a trans isomer can be obtained stereoselectively by selecting a reaction, but usually obtained as a mixture thereof. It is often done.
- the object can be achieved by a method known per se such as chromatography, fractional recrystallization, distillation, or isomerization reaction.
- Nonlinear optical material and nonlinear optical element according to the present invention
- the nonlinear optical material and the nonlinear optical element according to the present invention can be obtained by a known method (for example, Oh et al., IEEE Journal on Selected Topics in Quantum Electronics, Vol. 7, No. 5, pp. 826-835, Sept./ Oct. 2001; Dalton et al., Journal of Materials Chemistry, 1999, 9, pp.1905-1920; Kai, IEICE Transactions, C Vol. J84-C, No. 9, pp. 744-755, September 2001; a method described in Ma et al., Advanced Materials, 2002, 14, No. 19, 2002, pp. 1339-1365, etc.).
- the nonlinear optical material according to the present invention contains the chromophore according to the present invention and a host material in which the chromophore is dispersed.
- the chromophore according to the present invention is preferably uniformly dispersed at a high concentration in the host material. From this viewpoint, the host material preferably exhibits high compatibility with the chromophore according to the present invention.
- the host material examples include resins such as polymethacrylate such as polymethyl methacrylate (PMMA), polyimide, polycarbonate, polystyrene, polysulfone, polyethersulfone, silicon resin, and epoxy resin. These resins are preferably used because they are excellent in compatibility with the chromophore according to the present invention and are excellent in transparency and moldability when used as a nonlinear optical element.
- resins such as polymethacrylate such as polymethyl methacrylate (PMMA), polyimide, polycarbonate, polystyrene, polysulfone, polyethersulfone, silicon resin, and epoxy resin.
- the chromophore and the host material are dissolved in an organic solvent at an appropriate mixing ratio, and then applied onto the substrate by spin coating.
- examples include a method of obtaining a thin film of a nonlinear optical material by performing a heat treatment.
- the host material preferably contains a resin having a reactive functional group capable of forming a covalent bond with the chromophore according to the present invention. Furthermore, it is preferable that at least a part of the chromophore according to the present invention is bonded to the resin having the reactive functional group.
- a nonlinear optical material can disperse the chromophore in the host material at high density, and can achieve high nonlinearity.
- the reactive functional group examples include a haloalkyl group, an acyl halide group, an alkoxycarbonyl group, an aryloxycarbonyl group, a hydroxy group, an amino group, an isocyanate group, an epoxy group, and a carboxy group.
- the reactive functional group can react with, for example, a hydroxy group, an amino group, an alkoxycarbonyl group or the like in the chromophore according to the present invention to form a covalent bond.
- the nonlinear optical element according to the present invention can be manufactured using the nonlinear optical material according to the present invention. Since the nonlinear optical material according to the present invention is excellent in nonlinearity and heat resistance, a nonlinear optical element having excellent optical performance and excellent durability that can withstand long-term use can be obtained.
- FIG. 1 shows a schematic cross-sectional view of an optical waveguide constituting an arm of a Mach-Zehnder modulator which is an example of a specific embodiment of a nonlinear optical element according to the present invention, but the present invention is not limited to this.
- a lower electrode 2 a first cladding layer 3, a core layer 4, a second cladding layer 6, and an upper electrode 8 are laminated in this order from the substrate 1 side.
- the core layer 4 is formed of the nonlinear optical material subjected to the electric field poling process
- the optical waveguide core 9 is formed by reactive ion etching or the like, and configures a Mach-Zehnder interferometer. Yes.
- the refractive index of the core layer 4 located between these electrodes changes, so that the phase difference between the Mach-Zehnder arms changes, and the propagation light propagates.
- the intensity can be modulated.
- the first cladding layer 3 and the second cladding layer 6 are not particularly limited as long as they have a refractive index lower than that of the core layer 4.
- ultraviolet curable resins such as acrylic resins, epoxy resins, and silicon resins are used.
- a thermosetting resin, an organic or inorganic composite sol-gel curable material such as polyimide or glass, silicon oxide or the like is preferably used.
- the lower electrode 2 is, for example, a conductive film such as a metal, a conductive oxide film, or a conductive organic polymer, and is used as an electrode at the time of polling or operation as an element.
- the upper electrode 8 is an electrode for applying an input electric signal.
- the nonlinear optical element according to the present invention is not limited to the Mach-Zehnder type, but other types (for example, a directional coupler type) ).
- the use of the nonlinear optical element according to the present invention is not limited to the optical modulator as long as it has a chromophore according to the present invention or a film or an optical waveguide formed by the nonlinear optical material according to the present invention.
- the nonlinear optical element according to the present invention includes, for example, an optical switch, an optical memory, a wavelength converter, a microwave, a millimeter wave, and a terahertz in addition to an optical modulator (for an ultrahigh-speed application, an optical interconnect application, an optical signal processing application, etc.). It can be used for electric field sensors such as waves, biopotential sensors such as myoelectric and electroencephalograms, spatial light modulators, optical scanners, etc., and also for signal transmission by light between electronic circuits by combining with electronic circuits Can be used.
- the extract was washed with 5% aqueous sodium hydrogen sulfite and then with saturated brine, and then dehydrated over anhydrous magnesium sulfate. Ether was distilled off and the residue was purified by silica gel column chromatography to obtain 1.54 g of an orange oil.
- the extract was washed with 5% aqueous sodium hydrogen sulfite and then with saturated brine, and then dehydrated with anhydrous magnesium sulfate. Ether was distilled off and the residue was purified by silica gel column chromatography to obtain 1.09 g of reddish orange crystals.
- Example 19 In the same manner as in Examples 1-1, 4-1, 13-1, and 16-1, 2-substituted oxybenzaldehyde compounds 19-1 to 19-4 shown in Table 1 were synthesized.
- Example 20 Dibutyl [3-substitutedoxy-4- [2- (thiophen-2-yl) vinyl] phenyl] amine as shown in Table 2 in the same manner as in Examples 1-2, 4-2, 13-2 and 16-2 Compounds 20-1 to 20-4 were synthesized.
- Example 21 5- [2- [2,4-disubstituted phenyl] vinyl] thiophene-2-carbaldehyde compound 21 shown in Table 3 in the same manner as in Examples 1-3, 4-3, 13-3 and 16-3 -1 to 21-4 were synthesized.
- Example 22 5- [2- (4-Dibutylamino-2-hydroxyphenyl) vinyl] thiophene-2-carbaldehyde 488 mg (0.82 mmol) of 5- [2- [4-dibutylamino-2- (tert-butyldiphenylsiloxy) phenyl] vinyl] thiophene-2-carbaldehyde is dissolved in 10 ml of tetrahydrofuran, and the mixture is stirred while stirring at room temperature. 2.5 ml of tetrabutylammonium bromide (1 mol tetrahydrofuran solution) was added dropwise. After stirring for 35 minutes, the mixture was poured into water and extracted with ethyl acetate.
- Example 24 5- [2- [4-Dibutylamino-2- (oxiranylmethoxy) phenyl] vinyl] thiophene-2-carbaldehyde 390 mg (1.09 mmol) of 5- [2- (4-dibutylamino-2-hydroxyphenyl) vinyl] thiophene-2-carbaldehyde and 0.22 g (1.61 mmol) of epibromohydrin were dissolved in 10 ml of acetonitrile. To this, 0.3 g (2.17 mmol) of anhydrous potassium carbonate and 40 mg of tetrabutylammonium iodide were added and stirred at 60 ° C. for 4 hours while heating.
- the reaction mixture was poured into water and extracted with ethyl acetate.
- the extract was washed with saturated brine, dehydrated with anhydrous sodium sulfate, and concentrated.
- the oily substance obtained by purification by silica gel column chromatography was dissolved in 50 ml of ether, 10 mg of iodine was added, and the mixture was stirred for 1 hour.
- the extract was washed with 5% aqueous sodium hydrogen sulfite and then with saturated brine, dried over anhydrous magnesium sulfate, and concentrated.
- the residual liquid was purified by silica gel column chromatography to obtain 264 mg of a dark reddish brown oily substance.
- Example 25 5- [2- [2- (3-Bromo-2-hydroxypropoxy) -4-dibutylaminophenyl] vinyl] thiophene-2-carbaldehyde 5- [2- (4-Dibutylamino-2-hydroxyphenyl) vinyl] thiophene-2-carbaldehyde 0.76 g (2.13 mmol) and epibromohydrin 0.8 g (5.84 mmol) were converted to 1-methyl-2-pyrrolidone 15 Dissolved in ml. To this was added anhydrous potassium carbonate 0.59 g (4.27 mmol), and the mixture was stirred at 60 ° C. for 4 hours. The reaction mixture was poured into water and extracted with ethyl acetate.
- the extract was washed with saturated brine, dehydrated with anhydrous sodium sulfate, and concentrated.
- the residual liquid was purified by silica gel column chromatography, and 0.31 g of 5- [2- [4-dibutylamino-2- (oxiranylmethoxy) phenyl] vinyl] thiophene-2-carbaldehyde was obtained from the previous fraction. It was. From the subsequent fraction, 0.45 g of 5- [2- [2- (3-bromo-2-hydroxypropoxy) -4-dibutylaminophenyl] vinyl] thiophene-2-carbaldehyde was obtained.
- Example 26 In the same manner as in Examples 24 and 25, 5- [2- [2,4-disubstituted phenyl] vinyl] thiophene-2-carbaldehyde compounds 26-1 to 26-3 shown in Table 4 were synthesized.
- Example 27 Second-order nonlinear optical compounds 27-1 to 27-29 shown in Table 5 were synthesized in the same manner as in Examples 1-4, 2, and 3.
- Example 28 2- [4- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -3-cyano-5,5-dimethyl-2 (5H) -furanylidene] propanedinitrile 4-Dibutylamino-2-methoxybenzaldehyde 170 mg (0.65 mmol) and 2- (3-cyano-4,5,5-trimethyl-2 (5H) -furanylidene) propanedinitrile 141 mg (0.71 mmol) in 5 ml of ethanol ) was dissolved. To this, 50 mg of ammonium acetate was added and stirred at 50 ° C. for 4 hours.
- Example 29 2- [4- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -3-cyano-5-methyl-5-trifluoromethyl-2 (5H) -furanylidene] propanedinitrile 149 mg (0.57 mmol) of 4-dibutylamino-2-methoxybenzaldehyde and 2- (3-cyano-4,5-dimethyl-5-trifluoromethyl-2 (5H) -furanylidene) propanedinitrile 158 in 5 ml of ethanol mg (0.62 mmol) was dissolved. After stirring at 50 ° C. for 2 hours, ethanol was distilled off and the residue was purified by silica gel column chromatography to obtain 262 mg of dark brown crystals.
- Example 30 2- [4- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -3-cyano-5-phenyl-5-trifluoromethyl-2 (5H) -furanylidene] propanedinitrile 4-Dibutylamino-2-methoxybenzaldehyde 150 mg (0.57 mmol) and 2- (3-cyano-4-methyl-5-phenyl-5-trifluoromethyl-2 (5H) -furanylidene) propanedi in 5 ml of ethanol Nitori 197 mg (0.63 mmol) was dissolved. After stirring at 50 ° C.
- Example 31 5,5-Dimethyl-2-oxo-4- [2- (2,4,6-trimethoxyphenyl) vinyl] -2,5-dihydrofuran-3-carbonitrile
- Example 32 2- ⁇ 3-Cyano-4- [2- (2,4,6-trimethoxyphenyl) vinyl] -5,5-dimethyl-2 (5H) -furanilidene ⁇ propanedinitrile
- Example 33 4- [2- (4-Dibutylamino-2,6-dimethoxyphenyl) vinyl] -5,5-dimethyl-2-oxo-2,5-dihydrofuran-3-carbonitrile The compound was synthesized in the same manner as in Example 30.
- Example 34 2- ⁇ 4- [2- (4-Dibutylamino-2,6-dimethoxyphenyl) vinyl] -3-cyano-5,5-dimethyl-2 (5H) -furanilidene ⁇ propanedinitrile
- the compound was synthesized in the same manner as in Example 30.
- Example 36 2- [4- [4- (4-Dibutylamino-2-methoxyphenyl) -1,3-butadienyl] -3-cyano-5-methyl-5-trifluoromethyl-2 (5H) -furanylidene] propanedi Nitrile 3- (4-Dibutylamino-2-methoxyphenyl) propenal 145 mg (0.50 mmol) and 2- (3-cyano-4,5-dimethyl-5-trifluoromethyl-2 (5H) -furanilidene in 5 ml of ethanol ) 140 mg (0.55 mmol) of propanedinitrile was dissolved. After stirring at 50 ° C.
- Example 39 2- [4- [6- (4-Dibutylamino-2-methoxyphenyl) -1,3,5-hexatrienyl] -3-cyano-5-methyl 5-trifluoromethyl-2 (5H) -furanylidene] Propanedinitrile 5- (4-Dibutylamino-2-methoxyphenyl) -2,4-pentadienal 130 mg (0.41 mmol) and 2- (3-cyano-4,5-dimethyl-5-trifluoromethyl in 5 ml of ethanol -2 (5H) -furanylidene) propanedinitrile (115 mg, 0.45 mmol) was added, and the mixture was stirred at 50 ° C. for 2 hours.
- Example 40 2- [4- [6- (4-Dibutylamino-2-methoxyphenyl) -1,3,5-hexatrienyl] -3-cyano-5-phenyl-5-trifluoromethyl-2 (5H) -furanylidene ] Propanedinitrile 5- (4-Dibutylamino-2-methoxyphenyl) -2,4-pentadienal 100 mg (0.32 mmol) and 2- (3-cyano-4-methyl-5-phenyl-5-trimethyl in 5 ml of ethanol Fluoromethyl-2 (5H) -furanylidene) propanedinitrile (110 mg, 0.35 mmol) was added, and the mixture was stirred at 50 ° C. with heating for 2.5 hours.
- Example 42 2- [4- [2- [5- [4- (4-Dibutylamino-2-methoxyphenyl) -1,3-butadienyl] thiophen-2-yl] vinyl] -3-cyano-5methyl-5- Trifluoromethyl-2 (5H) -furanylidene] propanedinitrile 5- [4- (4-Dibutylamino-2-methoxyphenyl) -1,3-butadienyl] thiophene-2-carbaldehyde 230 mg (0.58 mmol) and 2- (3-cyano-4,5-dimethyl-5 -Trifluoromethyl-2 (5H) -furanylidene) propanedinitrile 160 mg (0.63 mmol) was dissolved in ethanol 8 ml.
- Example 43 2- [4- [2- [5- [4- (4-Dibutylamino-2-methoxyphenyl) -1,3-butadienyl] thiophen-2-yl] vinyl] -3-cyano-5-phenyl-5 -Trifluoromethyl-2 (5H) -furanylidene] propanedinitrile 5- [4- (4-Dibutylamino-2-methoxyphenyl) -1,3-butadienyl] thiophene-2-carbaldehyde 220 mg (0.55 mmol) and 2- (3-cyano-4-methyl-5-phenyl 192 mg (0.61 mmol) of -5-trifluoromethyl-2 (5H) -furanylidene) propanedinitrile was dissolved in 8 ml of ethanol.
- Example 48 2- [4- [3- [3- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -5,5-dimethyl-2-cyclohexenylidene] -1-propenyl] -3-cyano- 5-Phenyl-5-trifluoromethyl-2 (5H) -furanylidene] propanedinitrile [3- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -5,5-dimethyl-2-cyclohexenylidene] acetaldehyde 207 mg (0.51 mmol) and 2- (3-cyano-4-methyl -5-Phenyl-5-trifluoromethyl-2 (5H) -furanylidene) propanedinitrile (175 mg, 0.56 mmol) was dissolved, and the mixture was stirred at 50 ° C for 3 hours while heating.
- Example 50 2- [4- [3- [3- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -2-methoxy-5,5-dimethyl-2-cyclohexenylidene] -1-propenyl]- 3-Cyano-5-methyl-5-trifluoromethyl-2 (5H) -furanilidene] propanedinitrile [3- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -2-methoxy-5,5-dimethyl-2-cyclohexenylidene] acetaldehyde 200 mg (0.46 mmol) and 2- (3-cyano -4,5-Dimethyl-5-trifluoromethyl-2 (5H) -furanylidene) propanedinitrile 127 mg (0.50 mmol) was dissolved in 5 ml of ethanol.
- Example 51 2- [4- [3- [3- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -2-methoxy-5,5-dimethyl-2-cyclohexenylidene] -1-propenyl]- 3-Cyano-5-phenyl-5-trifluoromethyl-2 (5H) -furanilidene] propanedinitrile 3- [2- (4-Dibutylamino-2-methoxyphenyl) vinyl] -2-methoxy-5,5-dimethyl-2-cyclohexenylidene] acetaldehyde 185 mg (0.42 mmol) and 2- (3-cyano- 146 mg (0.46 mmol) of 4-methyl-5-phenyl-5-trifluoromethyl-2 (5H) -furanylidene) propanedinitrile was dissolved and stirred at 60 ° C.
- Example 54 In the same manner as in Example 53, 2- [3-cyano-5-methyl-5-trifluoromethyl-4- (2- ⁇ 5- [2- (4-dibutylamino-2,6-dimethoxyphenyl)] Vinyl] -2-thienyl ⁇ vinyl) -2 (5H) -furanylidene] propanedinitrile was synthesized.
- Example 55 In the same manner as in Example 53, 2- [3-cyano-5-phenyl-5-trifluoromethyl-4- [2- [5- [2- (4-dibutylamino-2,6-dimethoxyphenyl)] Vinyl] -2-thienyl] vinyl] -2 (5H) -furanylidene] propanedinitrile was synthesized.
- Example 58 2- [3-Cyano-4- [2- (8-methoxy-2,3,6,7-tetrahydro-1H, 5H-benzo [ij] quinolizin-9-yl) vinyl] -5,5-dimethyl- 2 (5H) -Furanilidene] propanedinitrile
- Example 60 2- [5- [2- (2-Benzyloxy-4-dibutylaminophenyl) vinyl] thiophen-2-yl] -3-cyano-2-butenedinitrile Dissolve 0.13 g (1.01 mmol) of tetracyanoethylene in 3 ml of N, N-dimethylformamide and stir under ice-cooling [3-benzyloxy-4- [2- (thiophen-2-yl) vinyl] phenyl] Dibutylamine 0.4 g (0.95 mmol) was dissolved in 1 ml of N, N-dimethylformamide and added dropwise. After stirring for 40 minutes, the mixture was further stirred at 40 ° C. overnight.
- reaction mixture was added to 100 ml of water, extracted with chloroform, and washed with saturated brine. The mixture was dehydrated with anhydrous sodium sulfate and concentrated. The residue was purified by silica gel column chromatography, and further washed with ethanol. 157 mg of black crystals of mp125-130 ° C. were obtained.
- the extract was washed with saturated brine, dehydrated with anhydrous sodium sulfate, and concentrated. This was purified by silica gel column chromatography and further washed with hexane. 0.88 g of black crystals of mp 86 ° C. were obtained.
- Example 62 2-Cyano-3- [5- [2- (4-dibutylamino-2-methoxyphenyl) vinyl] thiophen-2-yl] -2-butenedinitrile 235 mg (1.83 mmol) of tetracyanoethylene was dissolved in 7 ml of N, N-dimethylformamide. While stirring with ice cooling, 600 mg (1.75 mmol) of dibutyl [3-methoxy-4- [2- (thiophen-2-yl) vinyl] phenyl] amine was dissolved in 2 ml of N, N-dimethylformamide and added dropwise. And stirred for 1.5 hours.
- the ice bath was removed and the mixture was stirred at room temperature for 1 hour and further heated to 50 ° C. overnight.
- the reaction solution was added to 80 ml of water and extracted with chloroform. The extract was washed with saturated brine, dehydrated with anhydrous sodium sulfate, and concentrated. The residue was purified by silica gel column chromatography and further washed with methanol. 324 mg of dark reddish brown crystals of mp189-192 ° C were obtained.
- Example 63 2- [4- [Cyano- (4-dibutylamino-2-methoxyphenyl) methylene] -2,5-cyclohexadienylidene] malononitrile 460 mg (2.25 mmol) of 7,7,8,8-tetracyanoquinodimethane was dissolved in 8 ml of N, N-dimethylformamide. While stirring with ice cooling, 500 mg (2.12 mmol) of dibutyl (3-methoxyphenyl) amine was dissolved in 2 ml of N, N-dimethylformamide and added dropwise, followed by stirring for 1.5 hours.
- Example 64 2- [4- [Cyano- [5- [2- (4-dibutylamino-2-methoxyphenyl) vinyl] thiophen-2-yl] methylene] -2,5-cyclohexadienylidene] malononitrile 375 mg (1.84 mmol) of 7,7,8,8-tetracyanoquinodimethane was dissolved in 8 ml of N, N-dimethylformamide. While stirring with ice cooling, 600 mg (1.75 mmol) of dibutyl [3-methoxy-4- [2- (thiophen-2-yl) vinyl] phenyl] amine was dissolved in 2 ml of N, N-dimethylformamide and added dropwise.
- Example 65 2- [2- (4-Dibutylamino-2-methoxybenzylidene) -3-dicyanomethyleneindan-1-ylidene] malononitrile 4- (Dibutylamino) -2-methoxybenzaldehyde 0.8 g (3.04 mmol) and 1,3-bis (dicyanomethylidene) indane 0.73 g (3.01 mmol) were dissolved in 10 ml of acetic anhydride and heated at 80 ° C. with heating. Stir for hours. The reaction solution was poured into 200 ml of water, and the precipitated crystals were collected by filtration, washed with water and dried.
- Example 69 2- [4- [3- [2-Butyl-3- [2- (4-dibutylamino-2-methoxyphenyl) vinyl] -5,5-dimethyl-2-cyclohexenylidene] -1-propenyl]- 3-cyano-5-phenyl-5-trifluoromethyl-2 (5H) -furanylidene] propanedinitrile (69-1) 2-butyl-3,5,5-trimethyl-2-cyclohexenone 10.0 g (72.36 mmol) of isophorone and 14.9 g (95.6 mmol) of 1-bromobutane were dissolved in 60 ml of 1-methyl-2-pyrrolidone.
- Example 70 2- ⁇ 3-Cyano-4- [2- (4-dibutylamino-2,6-dimethoxyphenyl) vinyl] -5-methyl-5-trifluoromethyl-2 (5H) -furanylidene ⁇ propanedinitrile
- the compound was synthesized in the same manner as in Example 30.
- Example 71 2- ⁇ 3-Cyano-4- [2- (4-dibutylamino-2,6-dimethoxyphenyl) vinyl] -5-phenyl-5-trifluoromethyl-2 (5H) -furanylidene ⁇ propanedinitrile
- the compound was synthesized in the same manner as in Example 30.
- the extract was washed with saturated brine, dried over anhydrous sodium sulfate, and concentrated.
- the residue was purified by silica gel column chromatography, and the resulting orange solution was dissolved in 200 ml of toluene, and 200 mg of iodine was added. After stirring at room temperature for 15 minutes, the mixture was added to water and separated. The organic layer was dehydrated with anhydrous sodium sulfate and concentrated. Purification by silica gel column chromatography gave 6.09 g of an orange oil.
- Test example 1 For the compounds obtained in Examples 1 to 15 and Comparative Examples 1 and 2, the thermal decomposition onset temperature (Td), maximum absorption spectrum ( ⁇ max), and hyperpolarizability ( ⁇ ) were measured as follows. The results are shown in Tables 6 and 7.
- the thermal decomposition starting temperature (Td) is a differential differential thermal balance device TG8120 manufactured by Rigaku Corporation.
- the measurement sample is 5 mg
- the reference sample is Al 2 O 3
- a nitrogen atmosphere is used as a nitrogen atmosphere
- the temperature rising rate is 10 ° C./min. The measurement was performed.
- a photomultiplier tube (R3896) manufactured by Hamamatsu Photonics Co., Ltd. was used.
- a nonlinear optical compound was adjusted to a concentration of 5 ⁇ mol / l in 1,4-dioxane to obtain a measurement sample.
- Laser light is irradiated by the method described in the reference paper, the generated second optical harmonic intensity is detected by a photomultiplier tube, and compared with the hyperpolarizability of the reference sample, the hyperpolarizability ( ⁇ r ).
- the maximum absorption wavelength ( ⁇ max) of the nonlinear optical compound is adjusted with a concentration of 5 ⁇ mol / l of the nonlinear optical compound in 1,4-dioxane, and an ultraviolet-visible spectrophotometer (UVmini-1240) manufactured by Shimadzu Corporation is used. It was measured.
- UVmini-1240 ultraviolet-visible spectrophotometer
- the thermal decomposition onset temperature (Td) of the nonlinear optical compounds according to the present invention is significantly higher than that of the nonlinear optical compounds of Comparative Examples 1 and 2. There was no difference. However, it has been shown that the hyperpolarizability ( ⁇ ) of the nonlinear optical compounds according to the present invention (Examples 1 to 15) is remarkably improved as compared with the nonlinear optical compounds of Comparative Examples 1 and 2.
- the nonlinear optical compound according to the present invention was significantly superior in nonlinear optical characteristics without significantly degrading heat resistance as compared with the nonlinear optical compounds of Comparative Examples 1 and 2.
- Test example 2 For the compounds obtained in Examples 1 to 6, 13 to 18, 27-1 to 27-28, 28 to 30, 35 to 57 and Comparative Example 1, the thermal decomposition onset temperature (Td) and the maximum absorption were as follows: The spectrum ( ⁇ max) and hyperpolarizability ( ⁇ ) were measured. The results are shown in Table 8.
- the thermal decomposition starting temperature (Td) is a differential differential thermal balance device TG8120 manufactured by Rigaku Corporation.
- the measurement sample is 5 mg
- the reference sample is Al 2 O 3
- a nitrogen atmosphere is used as a nitrogen atmosphere
- the temperature rising rate is 10 ° C./min. The measurement was performed.
- an Nd: YAG laser OPO system Quanta Ray Nd: YAG laser, OPO versaScan, wavelength 1.90 ⁇ m, pulse width 20 nsec, repetition rate 10 Hz, output 90 mW
- a photomultiplier tube R2658 manufactured by Hamamatsu Photonics Co., Ltd. was used.
- the nonlinear optical compound was adjusted to a concentration of 5 ⁇ mol / l in chloroform to prepare a measurement sample.
- Laser light is irradiated by the method described in the reference paper, the generated second optical harmonic intensity is detected by a photomultiplier tube, and compared with the hyperpolarizability of the reference sample, the hyperpolarizability ( ⁇ )
- the hyperpolarizability of chloroform used as a solvent ⁇ 0.49 ⁇ 10-30 esu
- a reference paper “Pyrroline Chromophores for Electro-Optics”, Sei-Hum Jang, Jingdong Luo, Neil M.
- the maximum absorption wavelength ( ⁇ max) of the nonlinear optical compound was measured by adjusting the nonlinear optical compound at a concentration of 10 ⁇ mol / l in chloroform and using an ultraviolet-visible spectrophotometer (U-4000) manufactured by Hitachi, Ltd.
- the thermal decomposition start temperature (Td) of the nonlinear optical compound according to the present invention (Examples 1 to 6, 13 to 18, 27-1 to 27-28, 28 to 30, and 35 to 57).
- Td thermal decomposition start temperature
- ⁇ hyperpolarizabilities
- the nonlinear optical compound according to the present invention was significantly superior in nonlinear optical characteristics without significantly degrading heat resistance as compared with the nonlinear optical compound of Comparative Example 1.
- the nonlinear optical compound according to the present invention includes the aryl group substituted with the substituted oxy group in the donor structure part D, and thus has the effect of improving the nonlinear optical characteristics without significantly impairing the heat resistance.
- an optical modulator using the electro-optic effect it is possible to drive the optical modulator with lower electric power, thereby saving energy and reducing the size.
- a large nonlinear optical effect changes the intensity and phase of light even in a weak electric field, it can be used for an electric field sensor for measuring a leakage electric field of an electronic integrated circuit, a terahertz electromagnetic wave sensor, and the like.
- it can be used for signal transmission by light between electronic circuits by combination with the electronic circuit.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Nonlinear Science (AREA)
- General Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Plural Heterocyclic Compounds (AREA)
- Optical Modulation, Optical Deflection, Nonlinear Optics, Optical Demodulation, Optical Logic Elements (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
また、本発明は、当該非線形光学化合物を用いた非線形光学材料及び非線形光学素子を提供することも目的とする。
<1>ドナー構造部Dとπ共役ブリッジ構造部Bとアクセプター構造部Aを含む発色団において、当該ドナー構造部Dが置換オキシ基で置換されたアリール基を含み、当該アクセプター構造部Aが-SO2-を含まないことを特徴とする発色団、
<2>当該置換オキシ基が当該アリール基のオルト位の炭素原子に結合している、又は、オルト位及びパラ位の炭素原子に結合している、上記<1>項に記載の発色団、
<3>当該アリール基がさらに置換基を有していてもよいアミノ基で置換されていてもよい、上記<1>又は<2>項に記載の発色団、
<4>当該π共役ブリッジ構造部Bのπ共役が炭素-炭素共役である、上記<1>~<3>項のいずれかに記載の発色団、
<5>式D-B-A(ここで、Dはドナー構造部Dを示す;Bはπ共役ブリッジ構造部Bを示す;Aはアクセプター構造部Aを示す)で表わされる、上記<1>~<4>項のいずれかに記載の発色団、
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい
又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい)
で表わされる、上記<1>~<5>項のいずれかに記載の発色団、
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい)
で表わされる、上記<1>~<6>項のいずれかに記載の発色団、
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は、結合する窒素原子と一緒になって、置換基を有していてもよい飽和複素環を形成する、又は
RD 4及びRD 5は、結合する窒素原子、当該窒素原子が結合するアリールの炭素原子及び当該炭素原子に隣接するアリールの炭素原子と一緒になって、ヘテロ原子として窒素原子を含み、置換基を有していてもよい複素環を形成する)
で表わされる、上記<1>~<6>項のいずれかに記載の発色団、
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい)
で表わされる、上記<1>、<2>、<4>及び<5>項のいずれかに記載の発色団、
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい)
で表わされる、上記<1>、<2>、<4>、<5>及び<9>項のいずれかに記載の発色団、
Yは、-CRA 1RA 2-、-O-、-S-、-SO-、-SiRA 1RA 2-、-NR-(ここで、Rは水素原子又はアルキル基を示す)又は-C(=CH2)-を示す;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、アルケニル基、シクロアルキル基、シクロアルケニル基、アルコキシ基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RA 1及びRA 2は結合する炭素原子と一緒になって、置換基を有していてもよい
からなる群より選択される式で表される、上記<1>~<10>項のいずれかに記載の発色団、
RB 1~RB 8はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
nは1~5の整数を示す;
m及びm’はそれぞれ独立して、0~3の整数を示す)
からなる群より選択される式で表され、置換基を有していてもよい、上記<1>~<11>項のいずれかに記載の発色団、
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる、上記<1>~<12>項のいずれかに記載の発色団、
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<6>及び<11>~<13>項のいずれかに記載の発色団、
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、置換基を有していてもよい複素環を形成する、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<7>及び<11>~<14>項のいずれかに記載の発色団、
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>、<2>、<4>、<5>、<9>及び<11>~<13>項のいずれかに記載の発色団、
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる、上記<1>~<12>項のいずれかに記載の発色団、
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<6>、<11>、<12>及び<17>項のいずれかに記載の発色団、
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<7>、<11>、<12>、<17>及び<18>項のいずれかに記載の発色団、
m及びm’はそれぞれ独立して、0~3の整数を示す;
RB 1、RB 2及びRB 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる、上記<1>~<12>項のいずれかに記載の発色団、
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2及びRB 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<6>、<11>、<12>及び<20>項のいずれかに記載の発色団、
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2及びRB 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<7>、<11>、<12>、<20>及び<21>項のいずれかに記載の発色団、
nは1~5の整数を示す;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<6>、<11>、<12>及び<23>項のいずれかに記載の発色団、
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>~<6>、<11>、<12>及び<23>項のいずれかに記載の発色団、
nは1~5の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>、<2>、<4>、<5>、<9>、<11>、<12>及び<23>項のいずれかに記載の発色団、
nは1~5の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、上記<1>、<2>、<4>、<5>、<9>、<11>、<12>及び<23>項のいずれかに記載の発色団、
RD 2及びRD 3はそれぞれ独立して、水素原子、C1~6アルコキシ基、ベンジルオキシ基、シリルオキシ基、C2~6アルケニルオキシ基、C2~6アルケニルカルボニルオキシ基、C3~6アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、上記<6>~<10>、<14>~<16>、<18>、<19>、<21>、<22>及び<24>~<27>項のいずれかに記載の発色団、
<41>式
に関する。
<42>上記<1>~<41>項のいずれかに記載の発色団と、当該発色団が分散されるホスト材料とを含有する非線形光学材料、及び
<43>前記ホスト材料は、前記発色団との間に共有結合を形成し得る反応性官能基を有する樹脂を含有し、前記発色団の少なくとも一部は、当該樹脂と結合している上記<42>項に記載の非線形光学材料
に関する。
<44>上記<1>~<41>項のいずれかに記載の発色団又は上記<42>若しくは<43>項に記載の非線形光学材料によって形成される膜を有する非線形光学素子、
<45>上記<1>~<41>項のいずれかに記載の発色団又は上記<42>若しくは<43>に記載の非線形光学材料によって形成される光導波路を有する非線形光学素子、及び
<46>上記<1>~<41>項のいずれかに記載の発色団を含む非線形光学素子
に関する。
「単環式アリール基」としては、例えば、好ましくはC5~10環基、より好ましくはC5~7環基、さらにより好ましくはC5~6環基、最も好ましくは、C6環基(すなわち、フェニル基)等が挙げられる。ここで、例えば、C5~10環とは、環を形成する炭素原子が5~10個であることを意味し、以下同様である。
「多環式アリール基」としては、例えば、二環が縮合したアリール基及び三環が縮合したアリール基等が挙げられる。二環が縮合したアリール基としては、例えば、好ましくはC8~12環基等、より好ましくはC9~10環基等、最も好ましくはC10環基(すなわち、ナフチル基)等が挙げられる。
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、RD 1、RD 2及びRD 3はそれぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、置換基を有していてもよい飽和複素環を形成する、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含み、置換基を有していてもよい複素環を形成する)
で表わされる構造部等が挙げられる。
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、置換基を有していてもよい飽和複素環を形成する、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含み、置換基を有していてもよい複素環を形成する)
等が挙げられる。
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、RD 1、RD 2及びRD 3はそれぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は、結合する窒素原子と一緒になって、置換基を有していてもよい飽和複素環を形成する、又は
RD 4及びRD 5は、結合する窒素原子、当該窒素原子が結合するアリールの炭素原子及び当該炭素原子に隣接するアリールの炭素原子と一緒になって、ヘテロ原子として窒素原子を含み、置換基を有していてもよい複素環を形成する)
等が挙げられる。
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい)
で表される構造等が挙げられる。
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい)
で表される構造等が挙げられる。
Yは、-CRA 1RA 2-、-O-、-S-、-SO-、-SiRA 1RA 2-、-NR-(ここで、Rは水素原子又はアルキル基を示す)又は-C(=CH2)-を示す;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、アルケニル基、シクロアルキル基、シクロアルケニル基、アルコキシ基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RA 1及びRA 2は結合する炭素原子と一緒になって、置換基を有していてもよい
等で表される構造が挙げられる。
RA 1及びRA 2が示す「アルケニル基」としては、例えば、上記「(7)置換基を有していてもよいアルケニル基」における「アルケニル基」等が挙げられる。
RA 1及びRA 2が示す「シクロアルキル基」としては、例えば、C3~15の単環式又は多環式の飽和脂肪族環基等が挙げられる。当該シクロアルキル基としては、例えば、シクロプロピル基、シクロブチル基、シクロペンチル基、シクロヘキシル基、シクロヘプチル基、シクロオクチル基、シクロノニル基、シクロデシル基、シクロウンデシル基、シクロドデシル基等が挙げられ、より好ましくは、シクロヘキシル基等が挙げられる。
RA 1及びRA 2が示す「シクロアルケニル基」としては、例えば、C3~15の単環式又は多環式の不飽和脂肪族環基等が挙げられる。当該シクロアルケニル基としては、例えば、シクロプロペニル基、シクロペンテニル基、シクロヘキセニル基、シクロヘプチニル基、シクロオクテニル基、シクロペンタジエニル基、シクロヘキサジエニル基、シクロヘプタジエニル基、シクロオクタジエニル基等が挙げられる。
RA 1及びRA 2が示す「アルコキシ基」としては、例えば、上記「(a)置換基を有していてもよいアルコキシ基」における「アルコキシ基」等が挙げられる。当該アルコキシ基としては、例えば、メトキシ基、エトキシ基、プロポキシ基、イソプロポキシ基、ブトキシ基、イソブトキシ基、sec-ブトキシ基、tert-ブトキシ基等が挙げられる。
RA 1及びRA 2が示す「ハロアルキル基」としては、例えば、上記「(2)置換基を有していてもよいハロアルキル基」における「ハロアルキル基」等が挙げられる。当該ハロアルキル基としては、例えば、フルオロメチル基、ジフルオロメチル基、トリフルオロメチル基、2-フルオロエチル基、1,2-ジフルオロエチル基、クロロメチル、2-クロロエチル基、1,2-ジクロロエチル基、ブロモメチル基、ヨードメチル基等が挙げられ、好ましくは、例えば、トリフルオロメチル基等が挙げられる。
RA 1及びRA 2が示す「アリール基」としては、例えば、フェニル基、ナフチル基等が挙げられ、好ましくはフェニル基等が挙げられる。
Yは、-CRA 1RA 2-、-O-、-S-、-SO-、-SiRA 1RA 2-、-NR-(ここで、Rは水素原子又はアルキル基を示す)又は-C(=CH2)-を示す;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、アルケニル基、シクロアルキル基、シクロアルケニル基、アルコキシ基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される構造、式A-b
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、アルケニル基、シクロアルキル基、シクロアルケニル基、アルコキシ基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される構造等が挙げられる。
で表される構造等が挙げられる。
RB 1~RB 8はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
nは1~5の整数を示す;
m及びm’はそれぞれ独立して、0~3の整数を示す)
等で表される構造が挙げられる。
RB 1~RB 8が示す「アルコキシ基」としては、例えば、上記「(a)置換基を有していてもよいアルコキシ基」における「アルコキシ基」等が挙げられる。当該アルコキシ基としては、例えば、メトキシ基、エトキシ基、プロポキシ基、イソプロポキシ基、ブトキシ基、イソブトキシ基、sec-ブトキシ基、tert-ブトキシ基等が挙げられ、好ましくはメトキシ基等が挙げられる。
RB 1~RB 8が示す「アリール基」としては、例えば、フェニル基、ナフチル基等が挙げられ、好ましくはフェニル基等が挙げられる。
RB 1~RB 8が示す「アルケニル基」としては、例えば、直鎖状または分岐鎖状のC2~20アルケニル基等が挙げられる、当該アルケニル基としては、例えば、エテニル基、プロぺニル基、ブテニル基、ペンテニル基、ヘキセニル基等が挙げられる。
RB 1~RB 8が示す「シクロアルキル基」としては、例えば、C3~15の単環式又は多環式の飽和脂肪族環基等が挙げられる。当該シクロアルキル基としては、例えば、シクロプロピル基、シクロブチル基、シクロペンチル基、シクロヘキシル基、シクロヘプチル基、シクロオクチル基、シクロノニル基、シクロデシル基、シクロウンデシル基、シクロドデシル基等が挙げられ、より好ましくは、シクロヘキシル基等が挙げられる。
RB 1~RB 8が示す「シクロアルケニル基」としては、例えば、C3~15の単環式又は多環式の不飽和脂肪族環基等が挙げられる。当該シクロアルケニル基としては、例えば、シクロプロペニル基、シクロペンテニル基、シクロヘキセニル基、シクロヘプチニル基、シクロオクテニル基、シクロペンタジエニル基、シクロヘキサジエニル基、シクロヘプタジエニル基、シクロオクタジエニル基等が挙げられる。
RB 1~RB 8が示す「ハロアルキル基」としては、例えば、上記「(2)置換基を有していてもよいハロアルキル基」における「ハロアルキル基」等が挙げられる。当該ハロアルキル基としては、例えば、フルオロメチル基、ジフルオロメチル基、トリフルオロメチル基、2-フルオロエチル基、1,2-ジフルオロエチル基、クロロメチル、2-クロロエチル基、1,2-ジクロロエチル基、ブロモメチル基、ヨードメチル基等が挙げられ、好ましくは、例えば、トリフルオロメチル基等が挙げられる。
RB 1~RB 8が示す「アラルキル基」としては、例えば、上記「(5)置換基を有していてもよいアラルキル基」における「アラルキル基」等が挙げられる。当該アラルキル基としては、例えば、ベンジル基、1-フェニルエチル基、フェネチル基、1-ナフチルメチル基、2-ナフチルメチル基、1-ナフチルエチル基、2-ナフチルエチル基等が挙げられ、好ましくは、ベンジル基等が挙げられる。
RB 1~RB 8が示す「アリールオキシ基」としては、例えば、上記「(b)置換基を有していてもよいアリールオキシ基」における「アリールオキシ基」等が挙げられる。当該アリールオキシ基としては、例えば、フェノキシ基、ナフチルオキシ基等が挙げられ、好ましくはフェノキシ基等が挙げられる。
RB 1~RB 8が示す「アラルキルオキシ基」としては、上記「(c)置換基を有していてもよいアラルキルオキシ基」における「アラルキルオキシ基」等が挙げられる。当該アラルキルオキシ基としては、例えば、ベンジルオキシ基、フェネチルオキシ基、1-ナフチルメトキシ基、2-ナフチルメトキシ基等が挙げられ、好ましくは、ベンジルオキシ基等が挙げられる。
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RB 9及びRB 10はそれぞれ独立して、水素原子又はシアノ基(-CN)を示す;
mは、0~3の整数を示す)
等で表される構造が挙げられる。
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい)、式B-II
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい)、式B-III
m及びm’はそれぞれ独立して、0~3の整数を示す;
RB 1、RB 2及びRB 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アルケニル基、シクロアルキル基、シクロアルケニル基、ハロアルキル基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい)、及び、式B-IV
等で表される構造が挙げられる。
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる化合物、式I-1-a
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1~RD 5、RB 1、RB 2、RA 1及びRA 2は、上記式I-1で表される化合物における定義と同一の意味を有する)
で表わされる化合物、式I-1-b
で表わされる化合物、式I-2
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
π1、π2、RB 1、RB 2、RA 1及びRA 2は、上記式I-1で表される化合物における定義と同一の意味を有する)
で表される化合物等が挙げられる。
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、置換基を有していてもよい飽和複素環を形成する、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる化合物、式I-1-1-a
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1~RD 5、RB 1、RB 2、RA 1及びRA 2は、上記式I-1-1で表される化合物における定義と同一の意味を有する)
で表わされる化合物、式I-1-1-b
で表わされる化合物等が挙げられる。
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる化合物、式II-1-a
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1~RD 5、RB 1~RB 4、RA 1及びRA 2は、上記式II-1で表される化合物における定義と同一の意味を有する)
で表わされる化合物、式II-1-b
で表わされる化合物等が挙げられ、より好ましくは、式II-1-1
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、置換基を有していてもよい飽和複素環を形成する、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含み、置換基を有していてもよい複素環を形成する;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる化合物、式II-1-1-a
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1~RD 5、RB 1~RB 4、RA 1及びRA 2は、上記式II-1-1で表される化合物における定義と同一の意味を有する)
で表わされる化合物、式II-1-1-b
で表わされる化合物等が挙げられる。
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2及びRB 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表わされる化合物、式III-1-a
で表わされる化合物等が挙げられ、より好ましくは、式III-1-1
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)で表わされる化合物、式III-1-1-a
で表わされる化合物等が挙げられる。
nは1~5の整数を示す;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される化合物、式IV-1-b
で表される化合物、式IV-2-a
nは1~5の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される化合物、式IV-2-b
で表される化合物等が挙げられる。
本発明の発色団のうち、式I-1-b
反応温度は、通常約0℃~約150℃等であり、好ましくは約20℃~約80℃等である。
反応時間は、反応温度に影響されるが通常約1時間~約3日等である。
反応雰囲気は、通常大気中等で行われる。
反応終了後、通常の処理を行い得られた粗生成物を、通常の方法に従って精製することにより、又は、精製することなく、式I-1-bで表わされる化合物を得ることができる。
上記Xが示す「ハロゲン原子」としては、例えばフッ素原子、塩素原子、臭素原子、ヨウ素原子等が挙げられる。
上記Yが示す「ヒドロキシ基の保護基」としては、保護基としての機能を有するものであれば、特に限定されないが、例えば、アルキル基(例えば、メチル基、エチル基、イソプロピル基、tert-ブチル基等)、アルキルシリル基(例えば、トリメチルシリル基、tert-ブチルジメチルシリル基等)、アルコキシメチル基(例えば、メトキシメチル基、2-メトキシエトキシメチル基等)、テトラヒドロピラニル基、トリエチルシリルエトキシメチル基、アラルキル基(例えば、ベンジル基、p-メトキシベンジル基、2,3-ジメトキシベンジル基、o-ニトロベンジル基、p-ニトロベンジル基、トリチル基等)、アシル基(例えば、ホルミル基、アセチル基等)等が挙げられる。
本発明の発色団のうち、式II-1-b
反応終了後、通常の処理を行い得られた粗生成物を、通常の方法に従って精製することにより、又は、精製することなく、式II-1-bで表わされる化合物を得ることができる。
(ここで、RD 1~RD 5及びRB 1~RB 4は、上記式II-1-bで表わされる発色団における定義と同一の意味を有する;Yは水素原子又はヒドロキシ基の保護基を示す)で表わされる化合物を得た後に酸化反応に付し、Yがヒドロキシ基の保護基を示すときは、ヒドロキシ基の保護基の脱保護反応の後に酸化反応に付することにより、式IV-IIで表わされる化合物を得ることができる。
上記に述べた製造法B以外にも、式II-1-b
反応温度は、通常約-30℃~約100℃等であり、好ましくは約-10℃~約50℃等である。
反応時間は、通常数分~約5時間等であるが、反応の進行を薄層クロマトグラフィー等で確認しつつ適宜決定される。
反応雰囲気は、通常防湿下窒素又はアルゴン等不活性気体下等で行うのが好ましい。
反応終了後、通常の処理を行い得られた粗生成物を、通常の方法に従って精製することにより、又は、精製することなく、式II-1-bで表わされる化合物を得ることができる。
本発明の発色団のうち、式III-1-a
式IV-III
反応温度は、通常約0℃~約150℃等であり、好ましくは約20℃~約80℃等である。
反応時間は、反応温度に影響されるが通常約1時間~約3日等である。
反応雰囲気は、通常大気中で行われる。
反応終了後、通常の処理を行い得られた粗生成物を、通常の方法に従って精製することにより、又は、精製することなく、式III-1-aで表わされる化合物を得ることができる。
本発明に係る非線形光学材料は、本発明に係る発色団と、当該発色団が分散されるホスト材料とを含有する。本発明に係る非線形光学材料が優れた非線形性を有するためには、本発明に係る発色団が当該ホスト材料中に高濃度で均一に分散されていることが好ましい。この観点からは、当該ホスト材料は、本発明に係る発色団と高い相溶性を示すことが好ましい。
s : シングレット、d : ダブレット、dd : ダブルダブレット、t : トリプレット、m : マルチプレット、b: ブロード、J : 結合定数
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.1 Hz), 1.35-1.41 (4H, m), 1.59-1.63 (4H, m), 3.34 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.02 (1H, s), 6.26 (1H, d, J = 8.8 Hz), 7.69 (1H, d, J= 8.8 Hz), 10.12 (1H, s)
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.40 (4H, m), 1.57-1.63 (4H, m), 3.31 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.12 (1H, d, J = 2.2 Hz), 6.25 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.02 (1H, d, J = 3.8 Hz), 7.06 (1H, d, J = 15.9 Hz), 7.35 (1H, d, J = 8.8 Hz), 7.41 (1H, d, J = 15.9 Hz), 7.61 (1H, d, J = 3.8 Hz), 9.79 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.5, 50.9, 55.2, 94.4, 104.6, 112.4, 116.0, 124.3, 128.7, 129.2, 137.7, 139.6, 150.1, 155.8, 159.1, 182.2
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.59-1.64 (4H, m), 1.74 (6H, s), 3.33 (4H, t, J = 7.7 Hz), 3.91 (3H, s), 6.11 (1H, d, J = 2.2 Hz), 6.28 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.53 (1H, d, J = 15.4 Hz), 6.99 (1H, d, J = 4.4 Hz), 7.08 (1H, d, J = 15.9 Hz), 7.34 (1H, d, J = 4.4 Hz), 7.37 (1H, d, J = 8.8 Hz), 7.44 (1H, d, J = 15.9 Hz), 7.75 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 26.6, 29.6, 50.9, 55.2, 55.3, 94.2, 96.7, 104.9, 111.1, 111.3, 111.7, 112.4, 115.7, 126.5, 129.0, 130.8, 137.0, 138.1, 139.4, 150.7, 156.6, 159.6, 172.8, 175.9
2-[4-[2-[5-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, DMSO-d6) δ: 0.93 (6H, t, J = 7.7 Hz), 1.31-1.37 (4H, m), 1.52-1.57 (4H, m), 2.09 (3H, s), 3.38 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.17 (1H, d, J = 2.2 Hz), 6.35 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.65 (1H, d, J = 14.8 Hz), 7.30 (1H, d, J = 3.8 Hz), 7.34 (1H, d, J = 15.9 Hz), 7.47 (1H, d, J = 15.9 Hz), 7.50 (1H, d, J = 8.8 Hz), 7.87 (1H, d, J = 3.8 Hz), 8.22 (1H, d, J = 14.8 Hz)
13C-NMR (150 MHz, DMSO-d6) δ: 13.7, 17.9, 19.5, 29.1, 49.9, 54.3, 55.2, 94.0, 94.7, 105.1, 110.2, 111.25, 111.34, 111.8, 112.0, 115.7, 120.8, 128.3, 129.7, 131.6, 137.5, 141.1, 141.6, 150.9, 158.7, 159.5, 161.9, 176.1
2-[4-[2-[5-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.59-1.64 (4H, m), 3.34 (4H, t, J = 7.7 Hz), 3.90 (3H, s), 6.09 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.56 (1H, d, J = 15.4 Hz), 6.99 (1H, d, J = 4.4 Hz), 7.09 (1H, d, J = 15.4 Hz), 7.29 (1H, d, J = 4.4 Hz), 7.37 (1H, d, J = 8.8 Hz), 7.49 (1H, d, J = 15.9 Hz), 7.51-7.56 (5H, m), 7.76 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.6, 51.0, 55.2, 57.4, 94.0, 105.2, 110.9, 111.1, 111.3, 111.4, 112.5, 115.7, 126.9, 127.3, 129.6, 129.7, 129.9, 131.4, 132.6, 137.7, 140.2, 141.6, 151.2, 159.7, 160.1, 161.7, 175.5
2-[4-[2-[5-[2-(2-ベンジルオキシ-4-ジブチルアミノフェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.94 (6H, t, J = 7.1 Hz), 1.29-1.34 (4H, m), 1.49-1.54 (4H, m), 3.27 (4H, t, J = 7.1 Hz), 5.18 (2H, s), 6.02 (1H, s), 6.26 (1H, d, J = 8.8 Hz), 7.31 (1H, m), 7.37-7.40 (2H, m), 7.40-7.44 (2H, m), 7.72 (1H, d, J= 8.8 Hz), 10.24 (1H, s)
1H-NMR (600 MHz, CDCl3) δ: 0.94 (6H, t, J = 7.7 Hz), 1.28-1.34 (4H, m), 1.48-1.53 (4H, m), 3.23 (4H, t, J = 7.7 Hz), 5.16 (2H, s), 6.12 (1H, d, J = 2.2 Hz), 6.26 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.98 (1H, d, J = 3.8 Hz), 7.11 (1H, d, J = 15.9 Hz), 7.32-7.48 (7H, m), 7.60 (1H, d, J = 3.8 Hz), 9.79 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.1, 20.4, 29.6, 51.0, 70.5, 96.5, 105.0, 112.8, 116.0, 116.3, 127.1, 128.0, 128.8, 129.0, 129.4, 137.3, 137.8, 139.8, 150.0, 155.8, 158.4, 182.3
1H-NMR (600 MHz, DMSO-d6) δ: 0.89 (6H, t, J = 7.7 Hz), 1.25-1.311 (4H, m), 1.40-1.45 (4H, m), 1.78 (6H, s), 3.27 (4H, t, J = 7.7 Hz), 5.27 (2H, s), 6.18 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.63 (1H, d, J = 15.4 Hz), 7.15 (1H, d, J = 3.8 Hz), 7.30 (1H, d, J = 15.9 Hz), 7.32-7.35 (1H, m), 7.41-7.48 (5H, m), 7.43 (1H, d, J = 15.9 H), 7.73 (1H, d, J = 3.8 Hz), 8.09 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, DMSO-d6) δ: 13.7, 19.5, 25.5, 29.0, 49.9, 52.3, 69.3, 95.5, 96.0, 98.3, 104.9, 111.25, 111.33, 111.9, 112.3, 113.0, 115.9, 126.9, 127.2, 127.6, 128.4, 129.1, 137.0, 137.3, 139.1, 140.2, 150.0, 154.7, 157.9, 174.3, 176.8
2-[4-[2-[5-[2-(2-ベンジルオキシ-4-ジブチルアミノフェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl6) δ: 0.94 (6H, t, J = 7.7 Hz), 1.28-1.35 (4H, m), 1.49-1.54 (4H, m), 1.90 (3H, s), 3.26 (4H, t, J = 7.7 Hz), 5.21 (2H, s), 6.09 (1H, d, J = 2.2 Hz), 6.28 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.43 (1H, d, J = 15.4 Hz), 7.00 (1H, d, J = 3.8 Hz), 7.17 (1H, d, J = 15.9 Hz), 7.34-7.46 (6H, m), 7.38 (1H, d, J = 8.8 Hz), 7.56 (1H, d, J = 15.9 Hz), 8.15 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.3, 20.3, 29.5, 51.0, 70.4, 96.1, 105.4, 109.9, 110.8, 111.4, 112.8, 116.0, 122.4, 126.9, 127.4, 128.1, 128.8, 129.9, 132.7, 136.9, 137.7, 140.2, 141.1, 151.0159.2, 159.4, 161.7, 174.9, 175.5
2-[4-[2-[5-[2-(2-ベンジルオキシ-4-ジブチルアミノフェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl6) δ: 0.94 (6H, t, J = 7.7 Hz), 1.28-1.34 (4H, m), 1.48-1.53 (4H, m), 3.25 (4H, t, J = 7.7 Hz), 5.20 (2H, s), 6.08 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.55 (1H, d, J = 15.4 Hz), 6.93 (1H, d, J = 3.8 Hz), 7.13 (1H, d, J = 15.9 Hz), 7.28 (1H, d, J = 3.8 Hz), 7.33-7.45 (6H, m), 7.50-7.56 (6H, m), 7.79 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.5, 51.0, 57.5, 70.4, 96.1, 105.4, 110.9, 111.1, 111.2, 111.3, 112.8, 116.0, 126.8, 126.9, 127.3, 128.1, 128.7, 129.7, 129.9, 131.4, 132.7, 136.9, 137.8, 140.1, 141.6, 151.0, 159.2, 159.6, 161.7, 175.5
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 3.31 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.15 (1H, d, J = 2.2 Hz), 6.28 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.95 (1H, d, J = 15.9 Hz), 7.45 (1H, d, J = 8.8 Hz), 7.56 (1H, d, J = 15.9 Hz), 7.60 (2H, d, J = 8.2 Hz), 7.80 (2H, d, J = 8.2 Hz), 9.94 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.4, 29.6, 50.9, 55.3, 94.6, 104.6, 113.2, 122.3, 126.1, 127.6, 128.0, 130.2, 134.1, 145.6, 149.9, 158.8, 191.7
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.59-1.64 (4H, m), 1.80 (6H, s), 3.33 (4H, t, J = 7.7 Hz), 3.90 (3H, s), 6.14 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.94 (1H, d, J = 16.5 Hz), 6.98 (1H, d, J = 15.9 Hz), 7.45 (1H, d, J = 8.8 Hz), 7.54 (2H, d, J = 8.8 Hz), 7.56 (2H, d, J = 8.8 Hz), 7.58 (1H, d, J = 15.9 Hz), 7.59 (1H, d, J = 16.5 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.4, 26.6, 29.6, 50.9, 55.3, 57.0, 94.4, 97.3, 98.4, 104.8, 110.6, 111.2, 111.9, 113.0, 122.0, 126.7, 128.0, 128.2, 129.8, 131.3, 144.7, 147.3, 150.1, 159.0, 173.8, 175.5
2-[4-[2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]フェニル]ビニル]-3-シアノ-5-メチル-5-トリフルオロメチル- 2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.42 (4H, m), 1.59-1.64 (4H, m), 1.97 (3H, s), 3.33 (4H, t, J = 7.7 Hz), 3.90 (3H, s), 6.14 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.91 (1H, d, J = 15.9 Hz), 6.96 (1H, d, J = 16.5 Hz), 7.46 (1H, d, J = 8.8 Hz), 7.55 (2H, d, J = 8.8 Hz), 7.58 (2H, d, J = 8.8 Hz), 7.63 (1H, d, J = 16.5 Hz), 8.00 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.2, 20.3, 29.6, 50.9, 55.3, 59.8, 94.3, 99.3, 104.8, 110.0, 110.4, 110.6, 111.9, 113.0, 121.9, 126.8, 128.4, 128.9, 130.5, 131.4, 145.8, 149.6, 150.3, 159.2, 163.3, 174.8, 186.4
2-[4-[2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]フェニル]ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 3.32 (4H, t, J = 7.7 Hz), 3.89 (3H, s), 6.12 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.92 (1H, d, J = 15.9 Hz), 7.00 (1H, d, J = 15.9 Hz), 7.43-7.59 (10H, m), 7.59 (1H, d, J = 15.9 Hz), 7.66 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.6, 50.9, 55.3, 60.0, 94.3, 99.9, 104.8, 110.1, 110.3, 110.6, 112.9. 113.1, 121.9, 126.7, 126.9, 128.4, 128.9, 129.3, 129.9, 130.5, 131.4, 131.7, 145.8, 150.3, 150.4, 159.2, 163.5, 175.0
2-[4-[2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-2,5-ジメチルフェニル]ビニル]-3-シアノ-5,5-ジメチル- 2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 2.41 (3H, s), 2.65 (3H, s), 3.32 (4H, t, J = 7.7 Hz), 3.89 (3H, s), 6.16 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.10 (1H, d, J = 15.9 Hz), 7.44 (1H, d, J = 15.9 Hz), 7.46 (1H, d, J = 8.8 Hz), 7.49 (1H, s), 7.56 (1H, s), 10.17 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.3, 19.4, 20.4, 29.5, 50.9, 55.3, 94.7, 104.6, 113.7, 120.1, 127.5, 128.0, 131.8, 132.8, 134.0, 134.8, 138.1, 143.4, 149.7, 158.7, 192.2
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.36-1.42 (4H, m), 1.59-1.64 (4H, m), 1.79 (6H, s), 2.42 (3H, s), 2.47 (3H, s), 3.33 (4H, t, J = 7.7 Hz), 3.90 (3H, s), 6.15 (1H, s), 6.30 (1H, d, J = 8.8 Hz), 6.89 (1H, d, J = 15.9 Hz), 7.10 (1H, d, J = 15.9 Hz), 7.47 (1H, d, J = 8.8 Hz), 7.49 (1H, d, J = 15.9 Hz), 7.50 (1H, s), 7.53 (1H, s), 8.03 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.4, 19.6, 20.4, 26.6, 29.6, 50.9, 55.3, 56.6, 94.5, 97.3, 104.7, 111.0, 111.3, 112.1, 112.9, 113.5, 119.6, 126.8, 128.2, 128.25, 128.28, 130.0, 133.8, 137.7, 143.4, 144.8, 150.1, 159.0, 174.2, 175.8
2-[4-[2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-2,5-ジメチルフェニル]ビニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.36-1.42 (4H, m), 1.60-1.65 (4H, m), 1.96 (3H, s), 2.42 (3H, s), 2.48 (3H, s), 3.33 (4H, t, J = 7.7 Hz), 3.90 (3H, s), 6.15 (1H, d, J = 2.2 Hz), 6.30 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.84 (1H, d, J = 15.9 Hz), 7.11 (1H, d, J = 15.9 Hz), 7.48 (1H, d, J = 8.8 Hz), 7.50 (1H, s), 7.55 (1H, s), 7.55 (1H, d, J = 15.9 Hz), 8.43 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.2, 19.4, 19.7, 20.3, 29.6, 50.9, 55.3, 59.3, 94.4, 98.1, 104.8, 110.2, 110.8, 110.9, 111.5, 119.3, 126.8, 128.4, 128.6, 129.2, 130.1, 134.0, 139.3, 144.7, 147.0, 150.3, 159.2, 163.7, 175.0
2-[4-[2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-2,5-ジメチルフェニル]ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 2.14 (3H, s), 2.38 (3H, s), 3.32 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.13 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, J = 8.8 Hz), 7.00 (1H, d, J = 15.4 Hz), 7.08 (1H, d, J = 15.9 Hz), 7.45-7.59 (9H, m), 7.86 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.1, 19.6, 20.3, 29.5, 50.9, 55.3, 59.6, 94.3, 99.5, 104.8, 110.3, 110.4, 110.7, 112.7, 113.5, 119.4, 126.7, 126.9, 128.4, 128.6, 129.1, 129.6, 129.9, 130.1, 131.7, 134.0, 139.1, 144.7, 148.1, 150.2, 159.1, 164.2, 175.1
2-[4-[2-[5-[2-[4-ジブチルアミノ-2-(tert-ブチルジフェニルシロキシ)フェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.74 (6H, t, J = 7.1 Hz), 0.97-1.03 (4H, m), 1.10 (9H, s), 1.13-1.18 (4H, m), 2.84 (4H, t, J = 7.1 Hz), 5.56 (1H, d, J = 2.2 Hz), 6.18 (1H, dd, J = 2.2 Hz, 9.3 Hz), 7.37-7.40 (4H, m), 7.41-7.45 (2H, m), 7.69-7.76 (5H, m), 10.48 (1H, s)
1H-NMR (600 MHz, CDCl3) δ: 0.74 (6H, t, J = 7.1 Hz), 0.97-1.03 (4H, m), 1.06-1.21 (4H, m), 1.17 (9H,s), 2.82 (4H, t, J = 7.1 Hz), 5.70 (1H, s,), 6.18 (1H, d, J = 8.8 Hz), 7.02 (1H, d, J = 3.8 Hz), 7.06 (1H, d, J = 15.9 Hz), 7.37-7.43 (7H, m), 7.63 (1H, d, J = 3.8 Hz), 7.75-7.78 (5H, m), 9.81 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 13.8, 19.6, 20.0, 26.6, 26.6, 29.2, 50.7, 102.8, 105.5, 113.8, 115.1, 124.4, 127.1, 127.8, 129.0, 129.9, 132.7, 135.4, 137.9, 139.6, 149.4, 155.1, 155.8, 182.2
1H-NMR (600 MHz, CDCl3) δ: 0.75 (6H, t, J = 7.1 Hz), 0.98-1.04 (4H, m), 1.14-1.21 (4H, m), 1.19 (9H, s), 1.74 (6H, s), 2.84 (4H, t, J = 7.7 Hz), 5.71 (1H, d, J = 2.2 Hz), 6.21 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.52 (1H, d, J = 15.4 Hz), 7.01 (1H, d, J = 3.8 Hz), 7.09 (1H, d, J = 15.9 Hz), 7.38-7.45 (8H, m), 7.75-7.79 (5H, m), 7.82 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.8, 19.7, 20.0, 26.6, 26.7, 29.3, 50.7, 55.5, 95.2, 96.7, 102.7, 105.9, 111.1, 111.2, 111.7, 112.5, 113.9, 114.8, 126.3, 127.7, 127.9, 130.1, 130.9, 132.5, 135.4, 136.9, 138.0, 139.3, 150.0, 155.7, 156.5, 172.7, 176.0
2-[4-[2-[5-[2-[4-ジブチルアミノ-2-(tert-ブチルジフェニルシロキシ)フェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.76 (6H, t, J = 7.1 Hz), 0.98-1.04 (4H, m), 1.08-1.21 (4H, m), 1.19 (9H), 1.89 (3H, s), 2.86 (4H, t, J = 7.1 Hz), 5.70 (1H, d, J = 2.2 Hz), 6.22 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.42 (1H, d, J = 15.4 Hz), 7.05 (1H, d, J = 4.4 Hz), 7.10 (1H, d, J = 15.4 Hz), 7.38-7.46 (7H, m), 7.48 (1H, d, J = 4.4 Hz), 7.74-7.76 (4H, m), 7.93 (1H, d, J = 15.4 Hz), 8.18 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.8, 19.3, 19.7, 20.0, 26.7, 29.3, 50.8, 57.3, 93.3, 102.6, 106.2, 109.8, 110.8, 111.4, 114.0, 114.7, 121.2, 127.2, 128.0, 130.1, 132.3, 132.4, 132.6, 135.4, 137.6, 140.3, 141.0, 150.6, 156.2, 159.5, 161.5, 175.5
2-[4-[2-[5-[2-[4-ジブチルアミノ-2-(tert-ブチルジフェニルシロキシ)フェニル)ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.75 (6H, t, J = 7.1 Hz), 0.98-1.04 (4H, m), 1.10-1.21 (4H, m), 1.17 (9H, s), 2.85 (4H, t, J = 7.7 Hz), 5.69 (1H, d, J = 2.2 Hz), 6.21 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.52 (1H, d, J = 15.9 Hz), 6.98 (1H, d, J = 4.4 Hz), 7.06 (1H, d, J = 15.9 Hz), 7.33 (1H, d, J = 4.4 Hz), 7.37-7.45 (7H, m), 7.50-7.55 (5H, m), 7.73-7.75 (4H, m), 7.88-7.90 (1H, m), 7.90 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.8, 19.7, 20.0, 26.6, 29.3, 50.8, 57.5, 95.9, 102.6, 106.2, 110.9, 111.1, 111.3, 114.0, 114.6, 121.2, 123.1, 126.7, 127.2, 127.86, 127.95, 129.7, 129.9, 130.4, 131.4, 132.3, 132.6, 135.4, 137.7, 140.3, 141.4, 150.6, 156.2, 159.6, 161.4, 175.6
2-[3-シアノ-4-[2-[5-[2-(8-メトキシ-2,3,6,7-テトラヒドロ-1H,5H-ベンゾ[ij]キノリジン-9-イル)ビニル]チオフェン-2-イル]ビニル]-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.91-1.96 (4H, m), 2.71 (2H, t, J = 6.6 Hz), 2.75 (2H, t, J = 6.6 Hz), 3.26-3.28 (4H, m), 3.81 (3H, s), 7.33 (1H, s), 10.00 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 20.8, 21.4, 27.3, 49.8, 50.1, 62.8, 112.3, 116.8, 117.2, 127.0, 149.0, 160.7, 187.6
1H-NMR (600 MHz, CDCl3) δ: 1.93-1.98 (4H, m), 2.73 (2H, t, J = 6.6 Hz), 2.77 (2H, t, J = 6.6 Hz),3.18-3.12 (4H, m), 3.72 (3H, s), 7.02 (1H, d, J = 15.9 Hz), 7.04 (1H, d, J = 3.8 Hz), 7.06 (1H, s), 7.34 (1H, d, J = 15.9 Hz), 7.62 (1H, d, J = 3.8 Hz), 9.80 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 21.3, 21.4, 21.9, 27.5, 49.6, 49.9, 61.1, 116.1, 124.6, 128.8, 137.6, 139.9, 155.3, 155.5, 182.3
1H-NMR (600 MHz, CDCl3) δ: 1.75 (6H, s), 1.93-1.99 (4H, m), 2.74 (2H, t, J = 6.6 Hz), 2.77 (2H, t, J = 6.6 Hz), 3.22-3.25 (4H, m), 3.76 (3H, s), 6.53 (1H, d, J = 15.4 Hz), 7.01 (1H, d, J = 3.8 Hz), 7.03 (1H, d, J = 15.9 Hz), 7.08 (1H, s), 7.35 (1H, d, J = 15.9 Hz), 7.36 (1H, d, J = 3.8 Hz), 7.77 (1H, d, J = 15.9Hz)
13C-NMR (150 MHz, CDCl3) δ: 21.2, 21.3, 21.8, 26.6, 27.6, 49.7, 50.0, 55.6, 61.3, 95.3, 96.8, 111.2, 111.6, 112.4, 113.9, 115.7, 115.8, 117.9, 124.8, 126.7, 130.4, 137.1, 138.0, 139.4, 145.3, 155.9, 156.0, 172.8, 175.9
2-[3-シアノ-4-[2-[5-[2-(8-メトキシ-2,3,6,7-テトラヒドロ-1H,5H-ベンゾ[ij]キノリジン-9-イル)ビニル]チオフェン- 2-イル]ビニル]- 5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.91 (3H, s), 1.94-1.99 (4H, m), 2.74 (2H, t, J = 6.0 H), 2.77 (2H, t, J = 6.6 Hz), 3.25-3.28 (4H, m), 3.76 (3H, s), 6.43 (1H, d, J = 15.4 Hz), 7.05 (1H, d, J = 4.4 Hz), 7.06 (1H, d, J = 15.9 Hz), 7.10 (1H, s), 7.42 (1H, d, J = 15.9 Hz), 7.45 (1H, d, J = 4.4 Hz), 8.15 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 19.4, 21.1, 21.4, 21.8, 27.7, 49.8, 50.1, 57.5, 61.5, 93.5, 93.7, 94.9, 110.1, 110.9, 111.4, 114.0, 115.7, 118.2, 122.5, 125.4, 127.6, 132.1, 137.9, 140.3, 141.1, 146.0, 156.5, 158.9, 161.8, 175.5
2-[3-シアノ-4-[2-[5-[2-(8-メトキシ-2,3,6,7-テトラヒドロ-1H,5H-ベンゾ[ij]キノリジン-9-イル)ビニル]チオフェン-2-イル]ビニル]-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.93-1.98 (4H, m), 2.72 (2H, t, J = 6.0 Hz), 2.77 (2H, t, J = 6.6 Hz), 3.24-3.27 (4H, m), 3.75 (3H, s), 6.57 (1H, d, J = 14.9 Hz), 6.99 (1H, d, J = 3.8 Hz), 7.03 (1H, d, J = 15.9 Hz), 7.07 (1H, s), 7.29 (1H, d, J = 3.8 Hz), 7.39 (1H, d, J = 15.9 Hz), 7.51-7.57 (5H, m,), 7.77 (1H, d, J = 14.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 21.1, 21.3, 21.7, 27.6, 49.7, 50.0, 57.7, 61.5, 95.8, 104.3, 110.8, 111.3, 113.9, 115.7, 118.1, 125.3, 126.8, 127.4, 129.76, 129.83, 131.5, 132.0, 137.8, 134.0, 141.6, 145.9, 156.4, 158.8, 161.8, 175.5
実施例1-1、4-1、13-1および16-1と同様にして表1に示した2-置換オキシベンズアルデヒド化合物19-1~19-4を合成した。
実施例1-2、4-2、13-2および16-2と同様にして表2に示したジブチル[3-置換オキシ-4-[2-(チオフェン-2-イル)ビニル]フェニル]アミン化合物20-1~20-4を合成した。
実施例1-3、4-3、13-3および16-3と同様にして表3に示した5-[2-[2,4-ジ置換フェニル]ビニル]チオフェン-2-カルボアルデヒド化合物21-1~21-4を合成した。
5-[2-(4-ジブチルアミノ-2-ヒドロキシフェニル)ビニル]チオフェン-2-カルボアルデヒド
1H-NMR (600 MHz, CDCl3) δ: 0.96 (6H, t, J = 7.7 Hz), 1.33-1.39 (4H, m), 1.53-1.58 (4H, m), 3.26 (4H, t, J = 7.7 Hz), 5.07 (1H, s), 6.01 (1H, s), 6.26 (1H, d, J = 8.8 Hz), 7.04 (1H, d, J = 3.8 Hz), 7.08 (1H, d, J = 15.9 Hz), 7.32 (1H, d, J = 8.8 Hz), 7.35 (1H, d, J = 16.5 Hz), 7.62 (1H, d, J = 3.8 Hz), 9.80 (1H, s)
5-[2-(4-ジブチルアミノ-2-ヒドロキシフェニル)ビニル]チオフェン-2-カルボアルデヒド
5-[2-[4-ジブチルアミノ-2-(オキシラニルメトキシ)フェニル]ビニル]チオフェン-2-カルボアルデヒド
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.39 (4H, m), 1.56-1.61 (4H, m), 2.81 (1H, dd, J = 2.5 Hz, 4.6 Hz), 2.96 (1H, t, J = 4.6 Hz), 3.30 (4H, t, J = 7.7 Hz), 3.40-3.43 (1H, m), 4.05 (1H, dd, J = 5.5 Hz, 11.0 Hz), 4.26 (2H, dd, J = 3.3 Hz, 11.5Hz), 6.16 (1H, d, J = 2.2 Hz), 6.28 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.04 (1H, d, J = 3.8 Hz), 7.07 (1H, d, J = 15.9 Hz), 7.37 (1H, d, J = 8.8 Hz), 7.43 (1H, d, J = 15.9 Hz), 7.62 (1H, d, J = 3.8 Hz), 9.80 (1H, s)
5-[2-[2-(3-ブロム-2-ヒドロキシプロポキシ)-4-ジブチルアミノフェニル]ビニル]チオフェン-2-カルボアルデヒド
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.40 (4H, m), 1.56-1.60 (4H, m), 3.30 (4H, t, J = 7.7 Hz), 4.19 (1H, dd, J = 3.3 Hz, 10.5 Hz), 4.30 (1H, dd, J = 3.8 Hz, 10.4 Hz,), 4.61-4.63 (1H, m), 4.69 (1H, t, J = 8.5 Hz), 5.09-5.12 (1H, m), 6.08 (1H, d, J = 2.2 Hz), 6.32 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.00 (1H, d, J = 15.9 Hz), 7.13 (1H, d, J = 3.8 Hz), 7.31 (1H, d, J = 15.9 Hz), 7.37 (1H, d, J = 8.8 Hz), 7.63 (1H, d, J = 3.8 Hz), 9.80 (1H, s)
実施例24および25と同様にして表4に示した5-[2-[2,4-ジ置換フェニル]ビニル]チオフェン-2-カルボアルデヒド化合物26-1~26-3を合成した。
実施例1-4、2、および3と同様にして表5に示した2次非線形光学化合物27-1~27-29を合成した。
2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.99 (6H, t, J = 7.7 Hz), 1.37-1.43 (4H, m), 1.62-1.67 (4H, m), 1.74 (6H, s), 3.41 (4H, t, J = 7.7 Hz), 3.91 (3H, s), 6.05 (1H, s), 6.35 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.82 (1H, d, J = 15.9 Hz), 7.53 (1H, d, J = 8.8 Hz), 7.95 (1H, d, J =15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 27.1, 29.6, 51.2, 53.1, 55.5, 93.3, 96.4, 106.5, 108.1, 111.8, 112.2, 112.4, 113.3, 131.4, 143.4, 154.2, 162.4, 175.0, 176.6
2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.00 (6H, t, J = 7.7 Hz), 1.38-1.44 (4H, m), 1.63-1.69 (4H, m), 1.89 (3H, s), 3.44 (4H, t, J = 7.7 Hz), 3.93 (3H, s), 6.03 (1H, d, J = 2.2 Hz), 6.39 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.76 (1H, b), 7.51 (1H, d, J = 8.8 Hz), 8.43 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 19.7, 20.2, 29.6, 51.5, 54.6, 55.6, 93.2, 93.4, 107.4, 111.6, 112.2, 112.3, 113.7, 121.4, 123.3, 145.7, 146.0, 155.3, 155.4, 163.5, 163.6, 176.3
2-[4-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.60-1.65 (4H, m), 3.40 (4H, t, J = 7.7 Hz), 3.83 (3H, s), 5.96 (1H, s), 6.32 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.87 (1H, b), 7.37 (1H, b), 7.46-7.52 (5H, m), 8.12 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 13.8, 20.2, 29.6, 51.5, 54.9, 55.5, 93.2, 95.7, 107.2, 111.6, 112.0, 112.1, 113.8, 121.3, 123.2, 126.8, 129.4, 130.8, 131.0, 146.8, 155.3, 163.5, 163.6, 176.5
5,5-ジメチル-2-オキソ-4-[2-(2,4,6-トリメトキシフェニル)ビニル]-2,5-ジヒドロフラン-3-カルボニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.67 (6H, s), 3.91 (3H, s), 3.92 (6H, s), 6.12 (2H, s), 7.36 (1H, d, J=18Hz), 8.12 (1H, d, J=18Hz)
13C-NMR (150 MHz, CDCl3) δ: 26.4, 55.5, 56.0, 86.7, 90.7, 94.8, 106.0, 112.8, 114.7, 137.9, 162.0, 164.7, 167.2, 179.4
2-{3-シアノ-4-[2-(2,4,6-トリメトキシフェニル)ビニル]-5,5-ジメチル-2(5H)-フラニリデン}プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.77 (6H, s), 3.93 (3H, s), 3.94 (6H, s), 6.12 (2H, s), 7.34 (1H, d, J=18Hz), 8.09 (1H, d, J=18Hz)
4-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]-5,5-ジメチル-2-オキソ-2,5-ジヒドロフラン-3-カルボニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.00 (6H, t,J=7Hz), 1.40(4H,m), 1.64(6H,s), 1.67(4H,m), 3.38 (4H, t,J=8Hz), 3.90 (6H, s), 5.78 (2H, s), 7.18 (1H, d, J=16Hz), 8.17 (1H, d, J=16Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.2, 26.7, 29.5, 50.9, 55.6, 86.4, 87.6, 90.8, 102.4, 111.0, 113.8, 138.7, 152.9, 162.6, 168.3, 179.6
2-{4-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン}プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.00 (6H, t,J=7Hz), 1.41(4H,m), 1.68(4H,m), 1.73(6H,s), 3.42 (4H, t,J=8Hz), 3.92 (6H, s), 5.78 (2H, s), 7.26 (1H, d, J=16Hz), 8.14 (1H, d, J=16Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.8, 20.2, 27.2, 29.6, 51.2, 55.7, 87.9, 96.1, 103.9, 110.2, 112.0, 112.9, 113.8, 140.8, 154.7, 163.6, 177.0, 177.1
2-[4-[4-(4-ジブチルアミノ-2-メトキシフェニル)-1,3-ブタジエニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.1 Hz), 1.34-1.40 (4H, m), 1.56-1.60 (4H, m), 3.31 (4H, t, J = 7.1 Hz), 3.85 (3H, s), 5.74 (1H, d, J = 16.5 Hz), 6.06 (1H, s), 6.21 (1H, d, J = 8.8 Hz), 7.18 (1H, d, J = 8.8 Hz), 7.46 (1H, d, J = 16.5 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.4, 50.9, 55.1, 89.2, 93.8, 104.3, 110.5, 120.9, 130.6, 146.4, 151.5, 160.2
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 3.33 (4H, t, J = 7.1 Hz), 3.87 (3H, s), 6.08 (1H, s), 6.26 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.61 (1H, dd, J = 8.2 Hz, 15.4 Hz), 7.39 (1H, d, J = 8.8 Hz), 7.70 (1H, d, J = 15.4 Hz), 9.55 (1H, d, J = 8.2 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.4, 50.9, 55.1, 93.7, 104.8, 110.9, 123.6, 130.8, 149.4, 152.3, 160.5
1H-NMR (600 MHz, CDCl3) δ: 0.99 (6H, t, J = 7.7 Hz), 1.37-1.42 (4H, m), 1.61-1.66 (4H, m), 1.69 (6H, s), 3.37 (4H, t, J = 7.7 Hz), 3.91 (3H, s), 6.06 (1H, b), 6.30 (1H, d, J = 14.8 Hz), 6.32 (1H, b), 6.94 (1H, dd, J = 14.9 Hz, 11.5Hz), 7.40 (1H, d, J = 8.8 Hz), 7.46 (1H, d, J = 14.8 Hz), 7.63 (1H, dd, J = 13.7 Hz, 13.2 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 26.7, 29.6, 51.2, 54.0, 55.3, 96.4, 105.9, 111.9, 112.2, 113.0, 113.3, 122.9, 131.4, 145.3, 151.7, 161.4, 173.4, 176.5
2-[4-[4-(4-ジブチルアミノ-2-メトキシフェニル)-1,3-ブタジエニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.00 (6H, t, J = 7.7 Hz), 1.37-1.44 (4H, m), 1.62-1.68 (4H, m), 1.83 (3H, s), 3.41 (4H, t, J = 7.7 Hz), 3.92 (3H, s), 6.04 (1H, s), 6.18 (1H, d, J = 14.8 H), 6.34 (1H, dd, J = 13.7 Hz, 13.2 Hz), 6.35 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.44 (1H, d, J = 8.8 Hz), 7.63 (1H, d, J = 14.8 Hz), 8.09 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 19.4, 20.3, 29.6, 51.3, 54.5, 55.3, 92.7, 93.4, 106.7, 111.7, 112.0, 112.3, 112.5, 114.3, 121.4, 123.3, 123.7, 132.5, 148.7, 154.0, 154.2, 161.2, 162.6, 176.2
2-[4-[4-(4-ジブチルアミノ-2-メトキシフェニル)-1,3-ブタジエニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.99 (6H, t, J = 7.7 Hz), 1.36-1.43 (4H, m), 1.60-1.66 (4H, m), 3.40 (4H, t, J = 7.7 Hz), 3.89 (3H, s), 6.01 (1H, s), 6.26 (1H, d, J = 13.7 Hz), 6.31 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.92 (1H, dd, J = 14.3 Hz, 13.2 Hz), 7.38 (1H, d, J = 8.8 Hz), 7.48-7.52 (6H, m), 7.90 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.6, 51.3, 55.0, 55.3, 93.4, 106.7, 111.7, 112.2, 112.4, 113.4, 114.4, 123.9, 126.8, 129.5, 130.6, 131.0, 148.7, 154.17, 154.24, 161.1, 162.6, 176.3
2-[4-[6-(4-ジブチルアミノ-2-メトキシフェニル)-1,3,5-へキサトリエニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.35-1.40 (4H, m), 1.55-1.60 (4H, m), 3.31 (4H, t, J = 7.7 Hz), 3.85 (3H, s), 4.97 and 5.20 (1H, d, J = 15.4 Hz), 6.04 (1H, s), 6.22-6.26 (1H, m), 6.71 and 6.73 (1H, dd, J = 0.7 Hz, 15.4 Hz), 7.07-7.18 (2H, m), 7.29 and 7.42 (1H, d, J = 8.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.5, 50.8, 55.2, 92.8, 94.1, 104.6, 104.7, 111.9, 119.8, 128.0, 129.2, 129.3, 137.4, 137.6, 150.7, 152.6, 159.6
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.1 Hz), 1.34-1.40 (4H, m), 1.58-1.63 (4H, m), 3.32 (4H, t, J = 7.1 Hz), 3.86 (3H, s), 6.09 (1H, s), 6.14 (1H, dd, J = 15.4 Hz, 8.2 Hz), 6.25 (1H, d, J = 8.8 Hz), 6.87 (1H, dd, J =15.4 Hz, 11.5 Hz), 7.26 (1H, d, J = 15.4 Hz), 7.28 (1H, d, J = 15.4 Hz), 7.37 (1H, d, J = 8.8 Hz), 9.53 (1H, d, J = 8.2 Hz)
13C-NMR (150 Hz, CDCl3) δ: 13.9, 20.3, 29.5, 50.9, 55.2, 94.1, 104.7, 121.4, 128.0, 129.4, 139.1, 150.9, 155.4, 159.7
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.60-1.63 (4H, m), 1.68 (6H, s), 3.35 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.07 (1H, s), 6.26 (1H, d, J = 8.8 Hz), 6.27 (1H, d, J = 15.4 Hz), 6.45 (1H, dd, J = 13.2 Hz, 12.1 Hz), 6.90 (1H, dd, J = 14.8 Hz, 11.5 Hz), 7.08 (1H, dd, J = 14.3 Hz, 11.5 Hz), 7.27 (1H, d, J = 15.4 Hz), 7.31 (1H, d, J = 8.8 Hz), 7.52 (1H, dd, J = 14.8 Hz, 11.5 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 26.6, 29.6, 51.0, 55.3, 93.9, 96.6, 105.3, 111.6, 112.0, 112.8, 113.3, 114.4, 123.3, 127.8, 130.1, 140.5, 149.4, 151.9, 160.4, 173.0, 176.2
2-[4-[6-(4-ジブチルアミノ-2-メトキシフェニル)-1,3,5-へキサトリエニル]-3-シアノ-5-メチル5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.99 (6H, t, J = 7.7 Hz), 1.36-1.42 (4H, m), 1.61-1.66 (4H, m), 1.82 (3H, s), 3.38 (4H, t, J = 7.7 Hz), 3.90 (3H, s), 6.05 (1H, s), 6.15 (1H, d, J = 14.3 Hz), 6.32 (1H, d, J = 8.8 Hz), 6.49 (1H, dd, J = 13.2 Hz, 12.6 Hz), 6.97 (1H, dd, J = 14.7 Hz, 11.5 Hz), 7.23 (1H, d, J = 13.2 Hz), 7.42 (1H, d, J = 8.8 Hz), 7.43 (1H, d, J = 13.7 Hz), 7.97 (1H, bt)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 19.3, 20.3, 29.6, 51.16, 51.21, 55.3, 93.1, 93.6, 106.1, 111.5, 112.1, 113.0, 114.1, 121.3. 123.2, 123.8, 128.6, 131.1, 144.2, 151.5, 155.4, 160.9, 161.4, 175.9
2-[4-[6-(4-ジブチルアミノ-2-メトキシフェニル)-1,3,5-へキサトリエニル]-3-シアノ- 5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.1 Hz), 1.35-1.41 (4H, m), 1.60-1.63 (4H, m), 3.37 (4H, t, J = 7.7 Hz), 3.87 (3H, s), 6.03 (1H, s), 6.25 (1H, d, J = 14.3 Hz), 6.30 (1H, d, 8.8 Hz), 6.42 (1H, dd, J = 13.2 Hz, 12.1 Hz), 6.91 (1H, dd, J = 14.8 Hz, 12.1 Hz), 7.10 (1H, dd, J = 12.6 Hz, 12.3 Hz), 7.36 (1H, d, J = 13.7 Hz), 7.39 (1H, d, J = 8.8 Hz), 7.48-7.53 (5H, m), 7.71 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.6, 51.2, 55.3, 93.6, 106.1, 111.5, 112.0, 112.1, 114.4, 121.3, 123.2, 123.7, 126.6, 126.8, 128.7, 129.5, 130.4, 131.1, 144.2, 151.9, 152.8, 154.2, 155.3, 160.8, 161.4, 176.0
2-[4-[2-[5-[4-(4-ジブチルアミノ-2-メトキシフェニル)-1,3-ブタジエニル]チオフェン-2-イル]ビニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.40 (4H, m), 1.57-1.62 (4H, m), 3.30 (4H, t, J = 7.7 Hz), 3.86 (3H, s), 6.11 (1H, d, J = 2.2 Hz), 6.25 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.62 (1H, d, J = 15.4 Hz), 6.78 (1H, dd, J = 15.4 Hz, 11.0 Hz), 6.99 (1H, d, J = 3.8 Hz), 7.01 (1H, dd, J = 15.4 Hz, 12.7 Hz), 7.02 (1H, d, J = 15.4 Hz), 7.34 (1H, d, J = 8.8 Hz), 7.60 (1H, d, J = 3.8 Hz), 9.80 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.6, 50.9, 55.3, 94.5, 104.6, 113.4, 120.7, 123.4, 124.9, 128.2, 132.2, 136.1, 137.6, 140.3, 149.7, 154.2, 158.8, 182.3
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.41 (4H, m), 1.58-1.63 (4H, m), 1.74 (6H, s), 3.32 (4H, t, J = 7.7 Hz), 3.87 (3H, s), 6.11 (1H, d, J = 2.2 Hz), 6.26 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.51 (1H, d, J = 15.9 Hz), 6.62 (1H, d, J = 15.4 Hz), 6.81 (1H, dd, J = 14.8 Hz, 11.0 Hz), 6.96 (1H, d, J = 3.8 Hz), 7.02 (1H, dd, J = 14.8 Hz, 11.0 H), 7.07 (1H, d, J = 14.8 Hz), 7.34 (1H, d, J = 3.8 Hz), 7.35 (1H, d, J = 8.8 Hz), 7.76 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 26.5, 29.6, 50.9, 55.2, 55.7, 94.3, 95.6, 96.9, 104.8, 111.2, 111.5, 112.4, 113.3, 120.4, 123.3, 126.9, 128.6, 133.9, 137.6, 137.7, 137.8, 139.2, 150.2, 154.5, 159.1, 172.7, 175.8
2-[4-[2-[5-[4-(4-ジブチルアミノ-2-メトキシフェニル)-1,3-ブタジエニル]チオフェン-2-イル]ビニル]-3-シアノ-5メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.58-1.64 (4H, m), 1.91 (3H, s), 3.33 (4H, t, J = 7.7 Hz), 3.87 (3H, s), 6.10 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.43 (1H, d, J = 14.8 Hz), 6.64 (1H, d, J = 14.8 Hz), 6.84 (1H, dd, J = 14.8 Hz, 11.0 Hz), 7.00 (1H, d, J = 3.8 Hz), 7.09 (1H, dd, J = 14.8 Hz, 11.0 Hz), 7.14 (1H, d, J = 15.4 Hz), 7.36 (1H, d, J = 8.8 Hz), 7.43 (1H, d, J = 3.8 Hz), 8.13 (1H, d, J = 14.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.2, 20.3, 29.6, 50.9, 55.2, 57.8, 94.2, 104.9, 110.4, 110.7, 111.2, 113.3, 120.4, 121.1, 123.0, 123.3, 127.7, 128.9, 135.4, 138.3, 139.5, 139.8, 140.9, 150.6, 157.1, 159.4, 161.7, 175.3
2-[4-[2-[5-[4-(4-ジブチルアミノ-2-メトキシフェニル)-1,3-ブタジエニル]チオフェン-2-イル]ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.41 (4H, m), 1.58-1.63 (4H, m), 3.32 (4H, t, J = 7.7 Hz), 3.87 (3H, s), 6.09 (1H, d, J = 2.2 Hz), 6.25 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.55 (1H, d, J = 14.8 Hz), 6.61 (1H, d, J = 14.8 Hz), 6.83 (1H, dd, J = 14.8 Hz, 11.0 Hz), 6.95 (1H, d, J = 4.4 Hz), 7.07 (1H, dd, J = 14.8 Hz, 11.0 Hz), 7.13 (1H, d, J = 15.4 Hz), 7.28 (1H, d, J = 4.4 Hz), 7.34 (1H, d, J = 8.8 Hz), 7.50-7.57 (5H, m), 7.79 (1H, d, J = 14.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.6, 50.9, 55.2, 58.0, 94.2, 104.9, 110.7, 111.1, 111.2, 111.6, 113.4, 120.4, 121.1, 123.3, 126.8, 127.6, 128.9, 129.8, 131.5, 135.4, 138.4, 139.5, 139.7, 141.4, 150.5, 157.2, 159.4, 161.7, 175.4
2-[4-[4-[5-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]チオフェン-2-イル]-1,3-ブタジエニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.40 (4H, m), 1.57-1.62 (4H, m), 3.30 (4H, t, J = 7.7 Hz), 3.87 (3H, s), 5.48 (1H, d, J = 16.5 Hz), 6.12 (1H, d, J = 2.2 Hz), 6.25 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.87 (1H, d, J = 3.8 Hz), 7.01 (1H, d, J = 15.9 Hz), 7.07 (1H, d, J = 3.8 Hz), 7.24 (1H, d, J = 15.9 Hz) 7.33 (1H, d, J = 8.8 Hz), 7.38 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.5, 50.9, 55.2, 91.8, 94.5, 104.6, 112.7, 116.2, 118.8, 124.8, 127.5, 128.3, 132.9, 134.9, 142.8, 149.8, 149.9, 158.8
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.34-1.40 (4H, m), 1.57-1.63 (4H, m), 3.31 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.13 (1H, d, J = 2.2 Hz), 6.25 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.41 (1H, dd, J = 15.4 Hz, 7.7 Hz), 6.93 (1H, d, J = 3.3 Hz), 7.04 (1H, d, J = 15.9 Hz), 7.21 (1H, d, J = 3.3 Hz), 7.28 (1H, d, J = 15.9 Hz), 7.35 (1H, d, J = 8.8 Hz), 7.50 (1H, d, J = 15.4 Hz), 9.59 (1H, d, J = 7.7 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.6, 50.9, 55.3, 94.5, 104.6, 112.7, 116.4, 125.2, 125.7, 127.6, 128.4, 134.0, 135.9, 144.8, 149.8, 151.3, 158.9, 192.8
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 1.70 (6H, s), 3.32 (4H, t, J = 7.7 Hz), 3.89 (3H, s), 6.12 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.37 (1H, d, J = 14.8 Hz), 6.69 (1H, dd, J = 14.8 Hz, 11.5 Hz), 6.94 (1H, d, J = 3.8 Hz), 7.06 (1H, d, J = 15.9 Hz), 7.16 (1H, d, J = 3.8 Hz), 7.24 (1H, d, J = 14.8 Hz), 7.32 (1H, d, J = 15.9 Hz), 7.36 (1H, d, J = 8.8 Hz), 7.55 (1H, dd, J = 14.8 Hz, 11.5 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 26.4, 29.6, 50.9, 55.3, 55.8, 94.4, 95.8, 96.9, 104.8, 111.2, 111.5, 112.4, 112.7, 116.0, 116.3, 125.5, 126.1, 128.6, 134.4, 138.1, 139.4, 148.1, 150.2, 152.5, 159.1, 172.9, 175.9
2-[4-[4-[5-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]チオフェン-2-イル]-1,3-ブタジエニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.1 Hz), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 3.32 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.10 (1H, s), 6.26 (1H, d, 8.8 Hz), 6.39 (1H, d, J = 14.8 Hz), 6.65 (1H, dd, J = 14.3 Hz, 11.5 Hz), 6.96 (1H, d, J = 3.8 Hz), 7.07 (1H, d, J = 15.9 Hz), 7.20 (1H, d, J = 3.8 Hz), 7.21 (1H, d, J = 14.8 Hz), 7.35 (1H, d, J = 8.8 Hz), 7.36 (1H, d, J = 15.9 Hz), 7.48-7.57 (5H, m), 7.69 (1H, bt)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.6, 50.9, 55.2, 58.0, 94.2, 96.0, 104.9, 110.7, 111.2, 112.7, 116.1, 121.1, 123.0, 126.0, 126.67, 126.76, 128.9, 129.69, 129.75, 129.9, 131.4, 136.3, 138.4, 141.6, 150.5, 150.8, 155.0, 159.4, 161.8, 175.5
2-[4-[3-[3-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-5,5-ジメチル-2-シクロヘキセニリデン]-1-プロペニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.1 Hz), 1.34-1.40 (4H, m), 1.57-1.62 (4H, m), 2.28 (2H, s), 2.49 (2H, s), 3.31 (4H, t, J = 7.1 Hz), 3.87 (3H, s), 5.99 (1H, s), 6.10 (1H, d, J = 2.2 Hz), 6.26 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.75 (1H, d, J = 15.9 Hz), 7.31 (1H, d, J = 15.9 Hz), 7.42 (1H, d, J = 8.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 28.6, 29.5, 33.3, 39.1, 50.8, 51.5, 55.2, 94.2, 104.7, 112.5, 124.2, 124.3, 128.3, 130.5, 150.4, 157.1, 159.2, 200.2
1H-NMR (600 MHz, CDCl3) δ: 0.96 and 0.97 (6H, t, J = 7.7 Hz), 1.00 and 1.03 (6H, s), 1.34-1.40 (4H, m), 1.56-1.62 (4H, m), 2.21 and 2.33 (2H, s), 2.33 and 2.45 (2H, s), 3.28-3.31 (4H, m), 3.856 and 3.864 (3H, s), 4.85 and 5.03 (1H, s), 6.11 (1H, d, J = 2.2 Hz), 6.19 and 6.64 (1H, s), 6.24 and 6.26 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.69 and 6.81 (1H, d, J = 16.5 Hz), 7.07 and 7.09 (1H, d, J = 16.5 Hz), 7.36 and 7.37 (1H, d, J = 8.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 14.2, 20.3, 21.1, 28.159, 28.197, 29.6, 31.1, 31.3, 38.9, 42.2, 44.9, 50.9, 55.2, 60.4, 89.5, 91.1, 94.4, 104.67, 104.74, 113.1, 118.2, 119.0, 122.1, 124.2, 125.2, 125.6, 127.1, 127.4, 127.7, 128.0, 146.5, 146.9, 149.8, 158.4, 158.69, 158.73
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.03 and 1.06 (6H, s), 1.34-1.40 (4H, m), 1.57-1.62 (4H, m), 2.27 and 2.39 (2H, s), 2.38 and 2.67 (2H, s), 3.30 (4H, m), 3.86 and 3.87 (3H, s), 5.67 and 5.89 (1H, d, J = 8.2 Hz), 6.11 (1H, d, J = 2.2 Hz), 6.24 and 7.15 (1H, s), 6.25-6.27 (1H, m), 6.76 and 6.80 (1H, d, J = 16.5 Hz), 7.10 and 7.12 (1H, d, J = 16.5 Hz), 7.38 and 7.40 (1H, d, J = 8.8 Hz), 10.03 and 10.21 (1H, d, J = 8.2 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 28.3, 28.4, 29.6, 31.1, 39.0, 39.2, 39.5, 46.5, 50.8, 55.2, 94.4, 104.8, 113.2, 120.0, 123.7, 125.69, 125.75, 125.9, 127.3, 127.4, 127.5, 127.91, 127.93, 147.7, 147.9, 150.0, 157.3, 157.6, 158.8, 158.9, 189.8, 190.6
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.03 and 1.05 (6H, s), 1.35-1.41 (4H, m), 1.58-1.63 (4H, m), 1.69 and 1.74 (6H, s), 2.34 and 2.44 (2 H, s), 2.42 (2H, s), 3.37 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.07 and 6.32 (1H, d, J = 12.6 Hz), 6.09 (1H, s), 6.15 and 6.21 (1H, d, J = 14.8 Hz), 6.27 (1H, d, J = 8.8 H), 6.36 and 6.78 (1 H, s), 6.84 and 6.86 (1H, d, J = 15.9 Hz,), 7.24 and 7.25 (1H, d, J = 15.9 Hz), 7.42 and 7.46 (1 H, d, J = 8.8 Hz), 8.00 and 8.30 (1 H, b)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 26.6, 28.3, 28.5, 29.6, 31.5, 31.8, 39.5, 39.7, 40.0, 50.9, 54.3, 55.2, 94.1, 96.5, 105.0, 112.0, 112.2, 112.9, 113.3, 114.5, 125.5, 127.3, 128.3, 129.0, 129.8, 143.4, 144.1, 150.6, 150.8, 156.2, 159.4, 172.8, 176.4
2-[4-[3-[3-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-5,5-ジメチル-2-シクロヘキセニリデン]-1-プロペニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.1 Hz), 1.05 (6 H, s), 1.35-1.41 (4H, m), 1.61-1.63 (4H, m), 1.82 (3H, s), 2.49 (4 H, s), 3.35 (4H, t, J = 7.1 Hz), 3.89 (3H, s), 6.08 (1H, s), 6.12 (1H, b), 6.29 (1H, d, J = 8.2 Hz), 6.38 (1H, d, J = 12.6 Hz), 6.43 (1H, s), 6.89 (1 H, d, J = 15.9 Hz), 7.42 (1H, d, J = 15.9 Hz), 7.46 (1 H, d, J = 8.8 Hz), 8.39 (1 H, b)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 14.2, 19.4, 20.3, 28.4, 29.6, 31.8, 39.8, 40.1, 51.0, 55.3, 60.4, 93.9, 105.5, 111.5, 112.2, 113.2, 125.3, 128.6, 129.1, 129.7, 132.8, 145.8, 151.5, 160.1, 171.2, 176.0
2-[4-[3-[3-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-5,5-ジメチル-2-シクロヘキセニリデン]-1-プロペニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (3H, s), 0.98 (6H, t, J = 7.7 Hz), 1.01 (3H, s), 1.35-1.41 (4H, m), 1.59-1.64 (4H, m), 2.26-2.34 (2H, b), 2.44 (2H, s), 3.35 (4H, t, J = 7.7 Hz), 3.88 (3H, s), 6.07 (1H, d, J = 2.2 Hz), 6.25 (2H, b), 6.29 (1H, d, J = 8.8 Hz), 6.37 (1H, b), 6.85 (1H, d, J = 15.4 Hz), 7.40 (1H, d, J = 15.4 Hz), 7.45 (1H, d, J = 8.8 Hz), 7.48-7.53 (5H, m), 8.08 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 28.1, 28.5, 29.6, 39.7, ,39.8, 51.0, 55.2, 55.3, 93.9, 105.5, 111.6, 112.2, 113.6, 114.47, 114.52, 123.2, 125.3, 126.7, 128.5, 129.1, 129.5, 130.7, 131.0, 132.9, 151.5, 160.2, 176.2
2-[4-[3-[3-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-2-メトキシ-5,5-ジメチル-2-シクロヘキセニリデン]-1-プロペニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.11 (6H, s), 1.34-1.40 (4H, m), 1.58-1.63 (4H, m), 2.36 (2H, s), 2.56 (2H, s), 3.31 (4H, t, J = 7.7 Hz), 3.73 (3H, s), 3.87 (3H, s), 6.11 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.23 (1H, d, J = 16.5 Hz), 7.28 (1H, d, J = 16.5 Hz), 7.53 (1H, d, J = 8.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 28.5, 29.5, 32.7, 38.8, 50.9, 52.1, 55.3, 60.5, 94.2, 104.8, 113.3, 118.1, 128.0, 129.6, 141.2, 147.2, 150.2, 159.0, 194.5
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.04 (6H, s), 1.34-1.40 (4H, m), 1.57-1.62 (4H, m), 2.39 (2H, s), 2.53 (2H, s), 3.30 (4H, m), 3.63 (3H, s), 3.86 (3H, s), 5.44 (1H, s), 6.11 (1H, d, J = 2.2 Hz), 6.26 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.05 (1H, d, J = 15.9 Hz), 7.15 (1H, d, J = 16.5 Hz), 7.46 (1H, d, J = 8.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 28.1, 29.6, 30.8, 39.0, 42.7, 50.9, 55.3, 60.8, 88.7, 94.4, 104.7, 113.6, 118.6, 119.1, 127.3, 127.7, 131.7, 148.0, 149.8, 153.2, 158.7
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz), 1.07 (6H, s), 1.34-1.40 (4H, m), 1.57-1.62 (4H, m), 2.43 (2H, s), 2.73 (2H, s), 3.31 (4H, t, J = 7.7 Hz), 3.64 (3H, s), 3.87 (3H, s), 6.12 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.30 (1H, d, J = 8.2 Hz), 7.09 (1H, d, J = 16.5 Hz), 7.24 (1H, d, J = 16.5 Hz), 7.48 (1H, d, J = 8.8 Hz), 10.07 (1H, d, J = 8.2 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 28.3, 29.6, 30.7, 38.8, 39.2, 50.9, 55.3, 60.9, 94.4, 104.8, 113.7, 119.2, 121.2, 127.4, 127.7, 133.8, 149.0, 149.9, 151.6, 158.8, 191.1
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.06 (6H, s), 1.35-1.41 (4H, m), 1.59-1.63 (4H, m), 1.69 (6H, s), 2.27 (2 H, s), 2.48 (2H, s), 3.33 (4H, t, J = 7.7 Hz), 3.69 (3H, s), 3.88 (3H, s), 6.10 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.33 (1H, d, J = 14.9 Hz), 6.76 (1H, d, J = 14.9 Hz), 7.21 (1 H, d, J = 14.9 Hz), 7.29 (1H, d, J = 16.5 Hz), 7.51 (1 H, d, J = 8.8 Hz), 8.00 (1H, t, J = 13.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 26.5, 28.3, 29.6, 31.0, 39.1, 40.1, 50.9, 54.7, 55.3, 61.3, 93.5, 94.2 96.6, 105.1, 111.9, 112.1, 112.8, 113.9, 115.8, 119.1, 122.4, 128.2, 129.6, 137.1, 144.2, 148.8, 150.5, 151.0, 159.3, 173.0, 176.3
2-[4-[3-[3-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-2-メトキシ-5,5-ジメチル-2-シクロヘキセニリデン]-1-プロペニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.98 (6H, t, J = 7.7 Hz), 1.06 (3H, s), 1.07 (3H, s), 1.36-1.42 (4H, m), 1.60-1.65 (4H, m), 1.84 (3H, s), 2.53 (4 H, s), 3.36 (4H, t, J = 7.7 Hz), 3.71 (3H, s), 3.90 (3H, s), 6.09 (1H, d, J = 2.2 Hz), 6.26 (1H, d, J = 13.7 Hz), 6.31 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.81 (1H, d, J = 12.6 Hz), 7.33 (1 H, d, J = 15.9 Hz), 7.38 (1H, d, J = 16.5 Hz), 7.55 (1 H, d, J = 8.8 Hz), 8.37 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.3, 20.3, 28.2, 29.6, 31.2, 39.4, 40.2, 51.0, 55.3, 55.7, 61.5, 92.9, 93.9, 105.5, 111.4, 112.0, 112.6, 114.1, 114.5, 119.0, 123.3, 128.9, 132.4, 141.7, 146.0, 151.3, 151.7, 151.9, 160.1, 161.1, 175.9
2-[4-[3-[3-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-2-メトキシ-5,5-ジメチル-2-シクロヘキセニリデン]-1-プロペニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.97 (3H, s), 0.98 (6H, t, J = 7.7 Hz), 1.02 (3H, s), 1.35-1.41 (4H, m), 1.59-1.64 (4H, m), 2.27 (1H, d, J = 15.9 Hz), 2.36 (1H, d, J = 15.9 Hz), 2.49 (2H, s), 3.35 (4H, t, J = 7.7 Hz), 3.66 (3H, s), 3.89 (3H, s), 6.08 (1H, d, J = 2.2 Hz), 6.30 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.37 (1H, d, J = 13.8 Hz), 6.72 (1H, d, J = 12.6 Hz), 7.30 (1H, d, J = 15.9 Hz), 7.36 (1H, d, J = 15.9 Hz), 7.50-7.54 (6H, m), 8.02 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 28.0, 28.4, 29.6, 31.2, 39.3, 51.0, 55.3, 55.9, 61.5, 91.9, 105.5, 111.4, 111.9, 114.2, 115.8, 119.0, 121.2, 123.2, 123.3, 126.8, 128.9, 129.5, 130.6, 131.1, 132.5, 141.7, 146.4, 151.4, 151.7, 151.8, 160.1, 161.2, 176.0
2-[3-シアノ-5-フェニル-5-トリフルオロメチル-4-[2-[5-[2-(2,4,6-トリメトキシフェニル)ビニル]-2-チエニル]ビニル]-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR(600MHz,CDCl3) δ:3.80(3H, s), 3.85(6H, s), 6.13(2H, s), 6.98(1H, d, J=6Hz), 7.29(1H, s), 7.10(1H, d, J=6Hz), 7.25(1H, d, J=18Hz), 7.58(1H, d, J=18Hz)
13C-NMR(150MHz,CDCl3) δ:55.2, 55.7, 90.7, 107.6, 119.6, 122.9, 123.0, 124.3, 127.3, 145.6, 159.4, 160.1
1H-NMR(600MHz,CDCl3) δ:3.74(3H, s), 3.79(6H, s), 6.03(2H, s), 6.98(1H, d, J=3.8Hz), 7.51(2H, s), 7.54(1H, d, J=3.8Hz), 9.70(1H, s)
1H-NMR(600MHz,CDCl3) δ:3.86(3H, s), 3.89(6H, s), 6.14(2H, s), 6.65(1H, d, J=12Hz), 7.04(1H, d, J=4Hz), 7.26(1H,s), 7.29(1H, d, J=4Hz), 7.49-7.56(5H, m), 7.61(1H, s), 7.75(1H, d, J=12Hz)
1H-NMR(600MHz,CDCl3) δ:0.946(6H, t, J=8Hz), 1.40(4H, m), 1.56(4H, m), 3.22(4H, t, J=7Hz), 3.75(6H, s), 5.83(3H, s)
13C-NMR(150MHz,CDCl3) δ:13.8, 20.2, 29.4, 50.7, 54.8, 87.0, 91.0, 149.8, 161.6
1H-NMR(600MHz,CDCl3) δ:0.99(6H, t, J=8Hz), 1.39(4H, m), 1.62(4H, m), 3.34(4H, t, J=7Hz), 3.85(6H, s,), 5.71(2H, s), 9.98(1H, s)
13C-NMR(150MHz,CDCl3) δ:13.9, 20.2, 29.4, 50.8, 55.5, 86.8, 104.7, 153.8, 164.2, 186.2
1H-NMR(600MHz,CDCl3) δ:0.96(6H, t, J=8Hz), 1.35(4H, m), 1.57(4H, m), 3.27(4H, t, J=7Hz), 3.86(6H, s,), 5.84(2H, s), 6.92(2H, d, J=2Hz), 7.03(1H, d, J=2Hz), 7.26(1H, d, J=16Hz), 7.48(1H, d, J=16Hz)
13C-NMR(150MHz,CDCl3) δ:13.9, 20.3, 29.5, 50.7, 54.4, 88.5, 103.1, 120.3, 121.9, 123.2, 127.2, 146.5, 148.5, 159.7
融点105~106℃
1H-NMR(600MHz,CDCl3) δ:0.96(6H, t, J=8Hz), 1.36(4H, m), 1.59(4H, m), 3.26(4H, t, J=7Hz), 3.84(6H, s,), 5.79(2H, s), 6.97(1H, d, J=4Hz), 7.46(1H, d, J=16Hz), 7.55(1H, d, J=4Hz), 7.60(1H, d, J=16Hz), 9.73(1H, s)
13C-NMR(150MHz,CDCl3) δ:13.9, 20.3, 29.6, 50.8, 55.4, 88.1, 101.6, 102.0, 118.4, 123.8, 125.6, 137.8, 139.0, 149.6, 157.7, 160.5, 182.1
アセトニトリル20 mlに5-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]チオフェン-2-カルバルデヒド401 mg(1.00 mmol)、2-(3-シアノ-4,5,5-トリメチル-2(5H)-フラニリデン)プロパンジニトリル199 mg(1.00 mmol)及び触媒量の酢酸アンモニウムを加えて一夜攪拌した。反応後、濃縮し、残渣をシリカゲルカラムクロマトグラフィーで精製すると目的化合物2-[3-シアノ-5,5-ジメチル-4-(2-{5-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]-2-チエニル}ビニル)-2(5H)-フラニリデン]プロパンジニトリルが暗赤色結晶として280 mg(収率48%)得られた。融点230℃
1H-NMR(600MHz,CDCl3) δ:0.99(6H, t, J=8Hz), 1.39(4H, m), 1.63(4H, m), 1.72(6H, s), 3.35(4H, t, J=7Hz), 3.91(6H, s), 5.80(2H, s), 6.48(1H, d, J=17Hz), 6.96(1H, d, J=4Hz), 7.31(1H, d, J=4Hz), 7.49(1H, d, J=16Hz), 7.62(2H, d, J=16Hz), 7.70(1H, d, J=17Hz)
13C-NMR(150MHz,CDCl3) δ:13.9, 19.2, 20.3, 29.7, 51.0, 54.8, 88.0, 92.7, 103.2, 109.1, 111.2, 111.9, 118.0, 121.3, 123.2, 127.1, 129.7, 137.4, 140.6, 151.1, 161.1, 161.8, 162.8, 175.5
実施例53と同様の方法で、2-[3-シアノ-5-メチル-5-トリフルオロメチル-4-(2-{5-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]-2-チエニル}ビニル)-2(5H)-フラニリデン]プロパンジニトリルを合成した。
黒色結晶、280mg(収率40%)融点203℃
13C-NMR(150MHz,THF-d8) δ:14.3, 18.6, 21.0, 30.6, 51.4, 55.8, 89.1, 104.1, 110.3, 111.4, 112.0, 112.1, 119.0, 127.8, 129.5, 138.3, 141.7, 141.9, 152.0, 162.0, 162.2, 176.3
実施例53と同様の方法で、2-[3-シアノ-5-フェニル-5-トリフルオロメチル-4-[2-[5-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]-2-チエニル]ビニル]-2(5H)-フラニリデン]プロパンジニトリルを合成した。
暗赤色結晶、550mg(収率77%)融点229~230℃
13C-NMR(150MHz,CDCl3) δ:14.1, 20.3, 29.8, 51.1, 55.6, 56.2, 88.0, 103.6, 110.5, 111.2, 111.5, 111.6, 118.2, 123.0, 127.2, 127.5, 129.8, 130.3, 130.5, 131.2, 137.0, 141.2, 151.2, 161.0, 161.3, 163.0, 175.4
2-(4-{2-[5-(2-{2-ベンジロキシ-4-[(2-ヒドロキシエチル)-メチルアミノ]-フェニル}-ビニル)-チオフェン-2-イル]-ビニル}-3シアノ-5-フェニル-5-トリフルオロメチル-5H-フラン-2-イリデン)-マロノニトリル
1H NMR (396 MHz, CDCl3):δ 7.14 (m, 1H, benzene ring), 6.36 (d, 1H, benzene ring), 6.29-6.28 (m, 2H, benzene ring), 4.23 (t, 2H, CH2), 3.78 (s,3H, OCH3), 3.60 (t, 2H, CH2), 2.97 (s, 3H, CH3), 1.20 (m, 9H, t-Bu)
1H NMR (396 MHz, CDCl3):δ 10.16 (s, 1H, CHO), 7.71 (m, 1H, benzene ring), 6.34 (m, 1H ,benzene ring), 6.13 (d, 1H, benzene ring), 4.26 (t, 2H, CH2), 3.91 (s, 3H, OCH3), 3.70 (t, 2H, CH2), 3.10 (s, 3H, CH3), 1.16 (m, 9H, t-Bu)
1H NMR (396 MHz, CDCl3):δ 7.38-6.85 (m, 5H, thiophene, CH=CH, benzene ring), 6.56 (dd, 1H, CH=CH), 6.34-6.24 (m, 2H, benzene ring), 4.22 (t, 2H, CH2), 3.82 (d, 3H, OCH3), 3.59 (t, 2H, CH2), 2.99 (s, 3H, CH3), 1.14 (m, 9H, t-Bu)
1H NMR (396 MHz, CDCl3):δ 7.34-6.83 (m, 5H, thiophene, CH=CH, benzene ring), 6.54 (dd, 1H, CH=CH), 6.33-6.23 (m, 2H, benzene ring), 3.83-3.75 (m, 5H, CH2,OCH3), 3.45 (m, 2H, CH2), 3.40 (d, 3H, CH3)
DMF(8 ml)中に2-{[3-メトキシ-4-(2-チオフェン-2-イルビニル)フェニル]メチルアミノ}エタノール(3.1 g, 10.8 mmol)とイミダゾール(1.47 g, 21.6 mmol)を溶解し、室温でDMF(5 ml)に溶解したtert-ブチルジメチルシリルクロリド(2.4 g,16.2 mmol)を滴下した。1時間撹拌後水300 mlを加え、油状物をジクロロメタンで抽出した。飽和食塩水で洗浄後、Na2SO4で乾燥、濃縮し、シリカゲルクロマトグラフィーで精製して黄色油状物([2-(tert-ブチルジメチルシラニロキシ)エチル][3-メトキシ-4-(2-チオフェン-2-イルビニル)フェニル]メチルアミン)を得た。収量4.3 g(98.6%)
1H NMR (396 MHz, CDCl3):δ 9.80 (s, 1H, CHO), 7.62 (d, 1H, thiophene), 7.40 (t, 2H, thiophene, CH=CH), 7.08 (q, 2H, benzene ring, CH=CH), 6.39 (m,1H, benzene ring), 6.27(m,1H, benzene ring), 3.90-3.84 (m, 5H,CH2,OCH3), 3.55 (t, 2H, CH2), 3.06 (s, 3H, CH3)
1H NMR (270 MHz, DMSO-d6):δ 7.75-7.61 (m, 7H, benzene ring, CH=CH), 7.51-7.26 (m, 4H, thiophene, benzene ring, CH=CH), 6.55-6.39 (m, 2H, CH=CH, benzene ring), 6.26 (s, 1H ,benzene ring), 3.88 (s, 3H, OCH3), 3.59-3.51 (m, 4H, CH2), 3.06 (s, 3H, CH3)
2-(4-{2-[5-(2-{2-ベンジロキシ-4-[(2-ヒドロキシエチル)-メチルアミノ]-フェニル}-ビニル)-チオフェン-2-イル]-ビニル}-3シアノ-5-フェニル-5-トリフルオロメチル-5H-フラン-2-イリデン)-マロノニトリル
1H NMR (396 MHz, CDCl3):δ 7.54 (d, 1H, benzene ring), 7.50 (d, 2H, benzene ring), 7.43 (dd, 2H, benzene ring), 7.24 (t, 1H, benzene ring), 6.49-6.47 (m, 3H, benzene ring), 5.13 (s, 2H, OCH2Ph), 4.31 (m, 2H, CH2), 3.64 (t, 2H, CH2), 3.00 (s, 3H, CH3), 1.30 (s, 9H, t-Bu)
1H NMR (396Hz, CDCl3):δ 10.28 (s, 1H, CHO), 7.73 (d, 1H, benzene ring), 7.44 (d, 1H, benzene ring), 7.40 (d, 2H, benzene ring), 7.40-7.27 (m, 2H, benzene ring), 6.33 (d, 1H, benzene ring), 6.19 (s, 1H, benzene ring), 5.17 (s, 2H, CH2Ph), 4.20 (t, 2H, CH2), 3.62 (t, 2H, CH2), 3.04 (s, 3H, CH3), 1.16 (m, 9H, t-Bu)
1H NMR (396 MHz, CDCl3):δ 7.48 (d, 1H, thiophene), 7.40-6.93 (m, 8H, thiophene ,benzene ring, CH=CH), 6.88-6.86 (q, 1H, benzene ring), 6.59 (dd, 1H, CH=CH), 6.35-6.26 (m, 2H, benzene ring), 5.11 (d, 2H, CH2Ph), 4.19 (m, 2H, CH2), 3.55 (m, 2H, CH2), 2.97 (s, 3H, CH3), 1.17 (s, 9H, t-Bu)
1H NMR (396 MHz, CDCl3):δ 7.48 (d,1H, thiophene), 7.42-6.94 (m, 8H, thiophene, benzene ring, CH=CH), 6.89 (q, 1H, benzene ring), 6.60 (dd, 1H, CH=CH), 6.41-6.33 (m, 2H, benzene ring), 5.12 (d, 2H, CH2Ph), 3.77 (m, 2H, CH2), 3.46 (t, 2H, CH2), 2.97 (s, 3H, CH3)
1H NMR (396 MHz, CDCl3):δ 7.46 (d, 1H, thiophene), 7.39-6.84 (m, 9H, thiophene, benzene ring, CH=CH), 6.58 (dd, 1H, CH=CH), 6.30-6.22 (m, 2H, benzene ring), 5.08 (d, 2H, CH2Ph), 3.71 (t, 2H, CH2), 3.42 (t, 2H, CH2), 2.95 (d, 3H, CH3), 0.86 (s, 9H, t-Bu), 0.00 (d, 6H, Si-CH3)
1H NMR (396 MHz, CDCl3):δ 9.77 (s, 1H, CHO), 7.58 (d, 1H, thiophene), 7.47-7.33 (m, 7H, benzene ring, CH=CH, thiophene), 7.40 (d, 1H, CH=CH), 6.96 (d, 1H, benzene ring), 6.31 (d, 1H, benzene ring), 6.21 (s, 1H, benzene ring), 5.13 (s,2H, CH2Ph), 3.72 (t, 2H, CH2), 3.46 (t, 2H, CH2), 3.00 (s, 3H, CH3), 0.87 (t, 9H, t-Bu), 0.00 (m, 6H, Si-CH3)
1H NMR (396 MHz, CDCl3):δ 9.77 (s, 1H, CHO), 7.60 (d, 1H, thiophene), 7.46 (d, 2H, benzene ring), 7.43-7.39 (t, 3H, benzene ring, CH=CH), 7.37-7.32 (m, 2H, benzene ring), 7.13 (d, 1H, CH=CH) ,6.98 (d, 1H, thiophene), 6.39-6.29 (m, 2H, benzene ring), 5.15 (s, 2H, CH2Ph), 3.77 (t, 2H, CH2), 3.49 (s, 2H, CH2), 3.00 (s, 3H, CH3)
1H NMR (270 MHz, CDCl3):δ 7.52-7.35 (m, 12H, benzene ring, thiophene, CH=CH), 7.47 (dd, 2H, CH=CH), 6.92 (d, 1H, thiophene), 6.58 (dd, 2H,CH=CH), 6.38(d, 1H, benzene ring), 6.28 (m, 1H, benzene ring), 5.19 (s, 2H, CH2Ph), 3.78 (t, 2H, CH2), 3.52 (t, 2H, CH2), 3.05 (s, 3H, CH3)
2-[3-シアノ-4-[2-(8-メトキシ-2,3,6,7-テトラヒドロ-1H,5H-ベンゾ[ij]キノリジン-9-イル)ビニル]-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.74 (6H, s), 1.94-2.01 (4H, m), 2.74-2.77 (4H, m), 3.36-3.39 (4H, m), 3.76 (3H, s), 6.70 (1H, d, J = 15.9 Hz), 7.24 (1H, s), 7.94 (1H, d, J =15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 20.5, 21.0, 21.2, 24.4, 27.0, 27.4, 50.1, 50.5, 53.0, 61.9, 91.9, 96.3, 107.5, 112.1, 112.5, 113.3, 113.7, 114.8, 119.3, 126.5, 143.2, 149.7, 159.1, 174.4, 176.6
2-[4-[2-[2-(tert-ブチルジフェニルシロキシ)-4-ジブチルアミノフェニル]ビニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.75 (6H, t, J = 7.1 Hz), 0.99-1.03 (4H, m), 1.14 (13H, m), 1.79 (6H, s), 2.87 (4H, br), 5.64 (1H, s), 6.29 (1H, d, J = 9.3 Hz), 6.88 (1H, d, J = 15.9 Hz), 7.40-7.42 (4H, m), 7.46-7.49 (2H, m), 7.66 (1H, d, J = 9.3 Hz), 7.71-7.73 (4H, m), 8.15 (1H, d, J =15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.7, 19.5, 20.2, 26.5, 27.2, 29.2, 51.0, 53.4, 93.4, 96.2, 102.2, 107.6, 111.6, 112.4, 113.2, 113.7, 128.2, 129.3, 130.5, 131.4, 135.2, 142.0, 153.5, 158.7, 174.5, 176.4
2-[5-[2-(2-ベンジルオキシ-4-ジブチルアミノフェニル)ビニル]チオフェン-2-イル]-3-シアノ-2-ブテンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.95 (6H, t, J = 7.7 Hz), 1.29-1.35 (4H, m), 1.50-1.55 (4H, m), 3.27 (4H, t, J = 7.7 Hz), 5.21 (2H, s), 6.08 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.03 (1H, d, J = 4.4 Hz), 7.18 (1H, d, J = 15.9 Hz), 7.34-7.37 (1H, m), 7.36 (1H, d, J = 8.8 Hz), 7.41-7.46 (4H, m), 7.60 (1H, d, J = 15.9 Hz), 7.85 (1H, d, J = 4.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.5, 51.0, 70.4, 75.4, 95.8, 105.6, 112.6, 113.3, 113.4, 113.8, 115.0, 126.1, 126.9, 128.2, 128.8, 129.4, 130.9, 131.3, 135.2, 136.7, 141.7, 151.6, 159.7, 162.7
2-シアノ-3-(4-ジブチルアミノ-2-メトキシフェニル)-2-ブテンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.94 (6H, t, J = 7.1 Hz), 1.31-1.37 (4H, m), 1.55-1.59 (4H, m), 3.24 (4H, t, J = 7.7 Hz), 3.78 (3H, s), 6.18-6.21 (2H, m), 6.27 (1H, dd, J = 2.2 Hz, 8.8Hz), 7.09-7.12 (1H, m)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.4, 50.8, 55.0, 98.3, 99.6, 105.0, 129.8, 149.6, 160.8
1H-NMR (600 MHz, CDCl3) δ: 0.99 (6H, t, J = 7.1 Hz), 1.37-1.43 (4H, m), 1.61-1.66 (4H, m,), 3.40 (4H, t, J = 7.7 Hz), 3.94 (3H, s), 6.04 (1H, d, J = 2.2 Hz), 6.34 (1H, dd, J = 2.2 Hz, 9.3 Hz), 7.61 (1H, d, J = 9.3 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.6, 51.4, 55.0, 81.7, 93.4, 106.2, 108.3, 113.8, 114.1, 115.0, 133.6, 135.4, 155.3, 161.4
2-シアノ-3-[5-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]チオフェン-2-イル]-2-ブテンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.99 (6H, t, J = 7.7 Hz), 1.36-1.42 (4H, m), 1.60-1.66 (4H, m), 3.36 (4H, t, J = 7.7 Hz), 3.92 (3H, s), 6.09 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.11 (1H, d, J = 4.4 Hz), 7.12 (1H, d, J = 15.9 Hz), 7.36 (1H, d, J = 8.8 Hz), 7.55 (1H, d, J = 15.4 Hz), 7.44 (1H, d, J = 4.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 29.6, 51.0, 55.2, 76.8, 93.8, 105.4, 112.3, 113.3, 113.5, 113.9, 114.6, 126.0, 129.2, 130.6, 131.4, 135.0, 141.6, 151.9, 160.6, 162.7
2-[4-[シアノ-(4-ジブチルアミノ-2-メトキシフェニル)メチレン]-2,5-シクロヘキサジエニリデン]マロンニトリル
1H-NMR (600 MHz, acetone-d6) δ: 0.95 (6H, t, J = 7.7 Hz), 1.34-1.41 (4H, m), 1.57-1.62 (4H, m), 3.36 (4H, t, J = 7.7 Hz), 3.86 (3H, s), 6.24 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.36 (1H, d, J = 2.2 Hz), 6.71 (1H, d, J = 8.8 Hz), 6.96 (2H, d, J = 8.2 Hz), 7.15 (2H, d, J = 8.2 Hz)
13C-NMR (150 MHz, acetone-d6) δ: 14.2, 20.8, 43.0, 51.3, 56.0, 96.7, 104.6, 109.2, 116.9, 119.4, 120.2, 126.1, 127.5, 129.4, 145.8, 151.9, 158.8
2-[4-[シアノ-[5-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]チオフェン-2-イル]メチレン]-2,5-シクロヘキサジエニリデン]マロンニトリル
1H-NMR (600 MHz, acetone-d6) δ: 0.96 (6H, t, J = 7.7 Hz), 1.37-1.42 (4H, m), 1.58-1.63 (4H, m), 3.37 (4H, t, J = 7.7 Hz), 3.85 (3H, s), 6.24 (1H, d, J = 2.2 Hz), 6.30 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.94 (1H, d, J = 3.8 Hz), 7.11 (2H, d, J = 8.8 Hz), 7.10 (1H, d, J = 16.5 Hz), 7.14 (1H, d, J = 16.5 Hz), 7.19 (1H, d, J = 3.8 Hz), 7.25 (2H, d, J = 8.8 Hz), 7.38 (1H, d, J = 8.8 Hz)
13C-NMR (150 MHz, acetone-d6) δ: 14.3, 20.9, 43.2, 51.3, 55.5, 95.5, 105.4, 111.3, 115.9, 116.7, 119.6, 119.7, 124.5, 125.5, 126.8, 127.0, 129.0, 129.5, 134.5, 147.3, 149.6, 150.7, 159.7
2-[2-(4-ジブチルアミノ-2-メトキシベンジリデン)-3-ジシアノメチレンインダン-1-イリデン]マロンニトリル
1H-NMR (600 MHz, CDCl3) δ: 1.01 (6H, t, J = 7.7 Hz), 1.39-1.45 (4H, m), 1.65-1.71 (4H, m), 3.45 (4H, t, J = 7.7 Hz), 3.97 (3H, s,), 6.02 (1H, d, J = 1.6 Hz), 6.40 (1H, d, J = 8.8 Hz), 7.40 (1H, d, J = 8.8 Hz), 7.63-7.65 (2H, m), 8.54-8.56 (2H, m), 9.13 1H, b)
13C-NMR (150 MHz, CDCl3) δ: 13.8, 20.3, 29.7, 51.6, 55.9, 67.4, 92.9, 107.9, 114.8, 115.5, 115.8, 120.5, 124.8, 133.2, 137.9, 142.2, 156.5, 161.1, 164.4
2-[4-[2-[5-[2-[2-ベンジルオキシ-4-[ブチル(4-ヒドロキシブチル)アミノ]フェニル]ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5,5-ジメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.93 (3H, t, J = 7.7 Hz), 1.29-1.35 (2H, m), 1.51-1.66 (6H, m), 3.22 (2H, t, J = 7.7 Hz), 3.26 (2H, t, J = 7.7 Hz), 3.66 (2H, t, J = 6.9 Hz), 5.05 (2H, s), 6.27-6.31 (3H, m), 7.11 (1H, t, J = 8.2 Hz), 7.30-7.33 (1H, m), 7.37-7.39 (2H, m), 7.43-7.45 (2H, m)
1H-NMR (600 MHz, CDCl3) δ: 0.93 (3H, t, J = 7.1 Hz), 1.29-1.35 (2H, m), 1.51-1.56 (2H, m), 1.62-1.63 (4H, m), 2.04 (3H, s), 3.22 (2H, t, J = 7.7 Hz), 3.25 (2H, t, J = 7.1 Hz), 4.07 (2H, t, J = 6.0 Hz), 5.04 (2H, s), 6.25-6.30 (3H, m), 7.10 (1H, dt, J = 1.1 Hz, 8.2 Hz), 7.30-7.32 (1H, m), 7.36-7.39 (2H, m), 7.43-7.44 (2H, m)
13C-NMR (150 MHz, CDCl3) δ: 14.01, 20.3, 21.0, 23.8, 26.2, 29.4, 50.6, 50.9, 64.3, 69.9, 99.4, 101.1, 105.3, 127.5, 127.8, 128.6, 129.9, 137.5, 149.4, and 160.2, 171.2
1H-NMR (600 MHz, CDCl3) δ: 0.94 (3H, t, J = 7.7 Hz), 1.29-1.35 (2H, m), 1.49-1.54 (2H, m), 1.58-1.64 (4H, m), 2.05 (3H, s), 3.26 (2H, t, J = 7.7 Hz), 3.31 (2H, t, J = 7.7 Hz), 4.07 (2H, t, J = 6.0 Hz), 5.18 (2H, s), 6.01 (1H, d, J = 2.2 Hz), 6.25 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.33 (1H, t, J = 7.1 Hz), 7.38-7.44 (4H, m), 7.73 (1H, d, J = 8.8 Hz), 10.25 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 13.7, 20.0, 20.7, 23.6, 25.9, 29.1, 50.4, 50.7, 63.7, 69.9, 94.3, 104.5, 114.4, 126.6, 127.8, 128.5, 130.1, 136.5, 153.6, 162.9, 170.8, 186.9
1H-NMR (600 MHz, CDCl3) δ: 0.94 (3H, t, J = 7.7 Hz), 1.29-1.35 (2H, m), 1.50-1.66 (6H, m), 3.27 (2H, t, J = 7.7 Hz), 3.32 (2H, t, J = 7.7 Hz), 3.66 (2H, t, J = 6.0 Hz), 5.18 (2H, s), 6.03 (1H, d, J = 2.2 Hz), 6.26 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.31-7.43 (6H, m), 7.72 (1H, d, J = 8.8 Hz), 10.24 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 23.8, 29.5, 30.0, 51.0, 51.1, 62.5, 70.2, 94.6, 104.8, 114.6, 127.0, 128.1, 128.8, 130.4, 136.9, 154.16, 163.3, 187.2
1H-NMR (600 MHz, CDCl3) δ: 0.93 (3H, t, J = 7.7 Hz), 1.04 (9H, s), 1.26-1.31 (2H, m), 1.47-1.55 (4H, m), 1.60-1.64 (2H, m), 3.21 (2H, t, J = 7.7 Hz), 3.24 (2H, t, J = 7.7 Hz), 3.67 (2H, t, J = 6.0 Hz), 5.13 (2H, s), 6.10 (1H, d, J = 2.2 Hz), 6.25 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.98 (1H, d, J = 3.8 Hz), 7.10 (1H, d, J = 15.9 Hz), 7.29-7.43 (12H, m), 7.46 (1H, d, J = 15.9 Hz), 7.60 (1H, d, J = 3.8 Hz), 7.65-7.66 (4H, m), 9.79 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 19.2, 20.3, 23.9, 26.9, 29.5, 30.0, 50.9, 51.0, 63.6, 70.4, 96.5, 105.0, 112.8, 116.3, 124.4, 127.0, 127.7, 127.9, 128.7, 128.9, 129.3, 129.6, 133.9, 135.6, 137.2, 137.7, 139.7, 149.8, 155.7, 158.3, 182.3
1H-NMR (600 MHz, CDCl3) δ: 0.93 (3H, t, J = 7.7 Hz), 1.28-1.34 (2H, m), 1.48-1.63 (6H, m), 3.24 (2H, t, J = 7.7 Hz), 3.28 (2H, t, J = 7.7 Hz), 3.65 (2H, t, J = 6.0 Hz), 5.17 (2H, s), 6.13 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.98 (1H, d, J = 3.8 Hz), 7.12 (1H, d, J = 15.9 Hz), 7.32-7.48 (7H, m), 7.60 (1H, d, J = 3.8 Hz), 9.79 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 23.8, 29.5, 30.1, 50.9, 51.0, 62.6, 70.4, 96.6, 105.1, 113.0, 116.4, 124.5, 127.0, 127.9, 128.7, 128.9, 129.2, 137.2, 137.7, 139.7, 149.8, 155.7, 158.3, 182.3
1H-NMR (600 MHz, CDCl3) δ: 0.94 (3H, t, J = 7.7 Hz), 1.28-1.34 (2H, m), 1.48-1.64 (6H, m), 1.73 (6H, s), 3.26 (2H, t, J = 7.7 Hz), 3.30 (2H, t, J = 7.7 Hz), 3.66 (2H, t, J = 6.0 Hz), 5.21 (2H, s), 6.12 (1H, d, J = 2.2 Hz), 6.29 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.50 (1H, d, J = 15.4 Hz), 6.94 (1H, d, J = 4.4 Hz), 7.14 (1H, d, J = 15.9 Hz), 7.31 (1H, d, J = 4.4 Hz), 7.34-7.49 (7H, m), 7.75 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 23.8, 26.6, 29.5, 30.0, 50.9, 51.0, 55.3, 62.6, 70.4, 95.2, 96.4, 96.8, 105.3, 111.1, 111.3, 111.7, 112.5, 112.9, 116.2, 126.6, 126.9, 128.0, 128.7, 129.4, 130.9, 137.0, 137.1, 138.0, 139.4, 150.4, 156.4, 158.7, 172.8, 175.9
2-[4-[2-[5-[2-[2-ベンジルオキシ-4-[ブチル(4-ヒドロキシブチル)アミノ]フェニル]ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-メチル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.94 (3H, t, J = 7.1 Hz), 1.28-1.34 (2H, m), 1.49-1.66 (6H, m), 1.88 (3H, s), 3.27 (2H, t, J = 7.7 Hz), 3.32 (2H, t, J = 7.1 Hz), 3.66 (2H, t, J = 6.0 Hz), 5.22 (2H, s), 6.12 (1H, s), 6.30 (1H, d, J = 8.8 Hz), 6.40 (1H, d, J = 15.4 Hz), 6.99 (1H, d, J = 3.8 Hz), 7.17 (1H, d, J = 15.4 Hz), 7.35-7.47 (7H, m), 7.54 (1H, d, J = 15.4 Hz), 8.12 (1H, d, J = 15.4 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 19.3, 20.3, 23.9, 29.5, 30.0, 50.99, 51.04, 57.2, 62.5, 70.4, 93.3, 93.5, 94.6, 96.2, 105.5, 109.9, 110.9, 111.4, 112.9, 116.1, 122.1, 127.0, 127.4, 128.1, 128.7, 130.0, 132.6, 137.0, 137.8, 140.3, 141.1, 150.9, 159.2, 159.4, 161.6, 175.4
2-[4-[2-[5-[2-[2-ベンジルオキシ-4-[ブチル(4-ヒドロキシブチル)アミノ]フェニル]ビニル]チオフェン-2-イル]ビニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
1H-NMR (600 MHz, CDCl3) δ: 0.93 (3H, t, J = 7.1 Hz), 1.29-1.34 (2H, m), 1.48-1.64 (6H, m), 3.26 (2H, t, J = 7.7 Hz), 3.31 (2H, t, J = 7.1 Hz), 3.66 (2H, t, J = 6.0 Hz), 5.21 (2H, s), 6.11 (1H, s), 6.28 (1H, d, J = 8.8 Hz), 6.55 (1H, d, J = 14.9 Hz), 6.94 (1H, d, J = 3.8 Hz), 7.14 (1H, d, J = 15.9 Hz), 7.20 (1H, d, J = 3.8 Hz), 7.33-7.56 (12H, m), 7.78 (1H, d, J = 14.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 13.9, 20.3, 23.8, 29.5, 30.0, 51.0, 57.5, 62.5, 70.4, 96.2, 105.5, 110.9, 111.1, 111.2, 111.3, 113.0, 116.2, 122.3, 126.8, 126.9, 127.4, 128.1, 128.7, 129.7, 129.8, 129.9, 131.5, 132.6, 137.0, 137.8, 140.1, 141.6150.8, 159.2, 159.5, 161.7, 175.5
2-[4-[3-[2-ブチル-3-[2-(4-ジブチルアミノ-2-メトキシフェニル)ビニル]-5,5-ジメチル-2-シクロヘキセニリデン]-1-プロペニル]-3-シアノ-5-フェニル-5-トリフルオロメチル-2(5H)-フラニリデン]プロパンジニトリル
(69-1) 2-ブチル-3,5,5-トリメチル-2-シクロヘキセノン
1H-NMR (600 MHz, CDCl3) δ: 0.90 (3H, t, J = 7.1 Hz), 1.00 (6H, s), 1.25-1.35 (4H, m), 1.91 (3H, s), 2.21 (2H, s), 2.23 (2H, s), 2.28 (2H, t, J = 7.7 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 21.3, 22.9, 24.8, 28.2, 31.4, 32.7, 47.0, 51.4, 134.8, 152.3, 199.0
1H-NMR (600 MHz, CDCl3) δ: 0.93 (3H, t, J = 6.6 Hz), 0.97 (6H, t, J = 7.7 Hz), 1.05 (6H, s), 1.33-1.40 (8H, m), 1.58-1.63 (4H, m), 2.29 (2H, s), 2.51 (2H, s), 2.51-2.53 (2H, m), 3.31 (4H, t, J = 7.7 Hz), 3.87 (3H, s), 6.13 (1H, d, J = 2.2 Hz), 6.28 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.20 (1H, d, J = 15.9 Hz), 7.24 (1H, d, J = 15.9 Hz), 7.41 (1H, d, J = 8.8 Hz,)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 14.1, 20.4, 23.0, 24.3, 28.5, 29.5, 32.2, 32.4, 39.9, 50.8, 51.6, 55.3, 94.4, 104.7, 113.4, 122.0, 128.3, 129.6, 133.7, 149.0, 150.0, 159.0, 199.3
1H-NMR (600 MHz, CDCl3) δ: 0.96 (3H, t, J = 6.6 Hz), 0.97 (6H, t, J = 7.7 Hz), 1.00 (6H, s), 1.33-1.43 (8H, m), 1.55-1.62 (4H, m), 2.38 (2H, s), 2.44 (2H, t, J = 7.7 Hz), 2.50 (2H, s), 3.30 (4H, t, J = 7.7 Hz), 3.86 (3H, s), 6.13 (1H, d, J = 2.2 Hz), 6.27 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.09 (1H, d, J = 15.9 Hz), 7.15 (1H, d, J = 15.9 Hz), 7.36 (1H, d, J = 8.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.4, 23.1, 26.9, 27.7, 28.1, 29.5, 30.4, 31.7, 40.4, 43.6, 50.9, 55.3, 89.5, 94.6, 104.7, 113.8, 119.7, 122.2, 127.7, 128.0, 130.6, 140.4, 149.7, 158.7, 159.3
1H-NMR (600 MHz, CDCl3) δ: 0.95 (3H, t, J = 7.1 Hz), 0.97 (6H, t, J = 7.7 Hz), 1.02 (6H, s), 1.34-1.44 (8H, m), 1.56-1.63 (4H, m), 2.42 (2H, s), 2.51 (2H, t, J = 7.4 Hz), 2.69 (2H, s), 3.31 (4H, t, J = 7.7 Hz), 3.87 (3H, s), 6.13 (1H, d, J = 2.2 Hz), 6.19 (1H, d, J = 8.2 Hz), 6.28 (1H, dd, J = 2.2 Hz, 8.8 Hz), 7.11 (1H, d, J = 15.9 Hz), 7.23 (1H, d, J = 15.9 Hz), 7.38 (1H, d, J = 8.8 Hz), 10.13 (1H, d, J = 8.2 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.4, 23.2, 27.2, 28.3, 29.6, 30.3, 32.0, 39.6, 40.4, 50.9, 55.3, 60.4, 94.5, 104.7, 113.9, 122.7, 123.2, 127.9, 128.1, 132.6, 141.9, 149.7, 158.1, 158.8, 191.8
1H-NMR (600 MHz, CDCl3) δ: 0.88 (3H, s), 0.97-1.00 (12H, m), 1.34-1.47 (8H, m), 1.59-1.64 (4H, m), 2.17 (1H, d, J = 15.4 Hz), 2.28 (1H, d, J = 15.9 Hz), 2.47 (2H, s), 2.25 (2H, b), 3.35 (4H, t, J = 7.7 Hz), 3.89 (3H, s), 6.09 (1H, d, J = 2.2 Hz), 6.30 (1H, dd, J = 2.2 Hz, 8.8 Hz), 6.37 (1H, d, J = 13.7 Hz), 6.58 (1H, d, J = 12.6 Hz), 7.32 (1H, d, J = 15.9 Hz), 7.40 (1H, d, J = 15.9 Hz), 7.41 (1H, d, J = 8.8 Hz), 7.51-7.54 (5H, m), 8.00 (1H, b)
13C-NMR (150 MHz, CDCl3) δ: 14.0 14.1, 20.3, 23.2, 27.0, 27.8, 28.5, 29.6, 30.7, 32.2, 40.6, 40.8, 55.3, 94.0, 95.2, 105.4, 111.6, 112.1, 114.1, 115.1, 121.3, 122.2, 123.2, 124.9, 126.9, 129.5, 130.8, 131.0, 133.5, 135.6, 147.5, 150.7, 151.3, 159.5, 160.2, 161.3, 176.0
2-{3-シアノ-4-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]-5-メチル-5-トリフルオロメチル-2 (5H)-フラニリデン}プロパンジニトリル
1H-NMR (600MHz, CDCl3) δ: 1.00 (6H, t, J=7.7Hz), 1.42 (4H, m), 1.68 (4H, m), 1.86 (3H, s), 3.44 (4H, m), 3.92 (6H, s), 5.76 (2H, s), 7.17 (1H, d, J=10.4Hz), 8.70 (1H, d, J=10.4Hz)
13C-NMR (150MHz, CDCl3) δ: 13.8, 20.2, 27.2, 29.6, 51.2, 55.7, 87.8, 96.1, 104.0, 110.0, 112.1, 113.2, 114.1, 131.6, 14.07, 154.5, 163.4, 176.6, 176.9
2-{3-シアノ-4-[2-(4-ジブチルアミノ-2,6-ジメトキシフェニル)ビニル]-5-フェニル-5-トリフルオロメチル-2 (5H)-フラニリデン}プロパンジニトリル
1H-NMR (600MHz, CDCl3) δ: 0.98 (6H, t, J=7.7Hz), 1.39 (4H, m), 1.64 (4H, m), 3.41 (4H, t, 7.7Hz), 3.83 (6H, s), 5.69 (2H, s), 7.31 (1H, b), 7.46-7.48 (3H, m), 7.52-7.53 (2H, m), 8.42 (1H, b)
13C-NMR (150MHz, CDCl3) δ: 13.8, 20.1, 29.7, 51.4, 55.7, 88.0, 105.3, 106.4, 110.8, 112.1, 113.0, 126.7, 129.1, 130.6, 131.5, 144.0, 155.9, 164.3, 177.0, 183.6
1H-NMR (600 MHz, CDCl3) δ: 0.96 (6H, t, J = 7.7 Hz) , 1.32-1.39 (4H, m), 1.55-1.60 (4H, m), 3.28 (4H, t, J = 7.7 Hz), 6.60 (2H, d, J = 8.8 Hz), 6.84 (1H, d, J = 15.9 Hz), 6.95 (1H, d, J = 3.9 Hz), 6.96 (1H, d, J = 3.9 Hz,), 6.99 (1H, d, J = 15.9 Hz), 7.09 (1H, t, J = 3.0 Hz), 7.31 (2H, d, J = 8.8 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.5, 50.8, 116.9, 128.8, 111.7, 122.7, 124.1,124.2, 127.4 127.6, 128.8, 144.2, 147.8
1H-NMR (600 MHz, CDCl3) δ: 0.96 (6H, t, J = 7.7 Hz) , 1.33-1.39 (4H, m), 1.55-1.61 (4H, m), 3.30 (4H, t, J = 7.7 Hz), 6.61 (2H, d, J = 8.8 Hz), 6.96 (1H, d, J = 15.9 Hz), 7.07 (1H, d, J = 15.9 Hz), 7.03 (1H, d, J = 4.2 Hz), 7.35 (2H, d, J = 8.8 Hz,),7.61 (1H, t, J = 4.2 Hz), 9.81 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.4, 50.7, 115.5, 133.7, 111.5, 122.8, 124.7, 128.5, 137.6, 140.0, 148.7, 154.5, 182.2
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.1 Hz), 1.34-1.40 (4H, m), 1.57-1.62 (4H, m), 1.75 (6H, s), 3.32 (4H, t, J = 7.7 Hz), 6.55 (1H, d, J = 15.9 Hz), 6.63 (2H, d, J = 8.8 Hz), 6.98 (1H, d, J = 15.9 Hz), 7.02 (1H, d, J = 3.8 Hz), 7.08 (1H, d, J = 15.9 Hz), 7.36 (1H, d, J = 3.8 Hz), 7.37 (2H, d, J = 8.8 Hz), 7.77 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 26.6, 29.5, 50.8, 55.8, 95.8, 96.9, 111.1, 111.5, 111.7, 112.3, 115.3, 122.7, 126.7, 129.0, 135.1, 137.2, 137.8, 139.4, 149.2, 154.8, 172.9, 175.8
1H-NMR (600 MHz, CDCl3) δ: 0.96 (6H, t, J = 7.7 Hz) , 1.33-1.39 (4H, m), 1.55-1.60 (4H, m), 3.28 (4H, t, J = 7.7 Hz), 4.66 (2H, d, J = 4.9 Hz), 6.62 (2H, d, J = 8.8 Hz), 6.86 (1H, d, J = 16.5 Hz), 7.02 (1H, d, J = 16.5 Hz), 7.31 (2H, d, J = 8.3 Hz), 7.37 (2H, d, J = 8.8 Hz), 7.45 (2H, d, J = 8.3 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.5, 50.8, 65.3, 111.6, 123.2, 124.1, 126.1, 127.4, 127.7, 129.0, 138.0, 139.2,148.0
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.7 Hz) , 1.34-1.40 (4H, m), 1.56-1.62 (4H, m), 3.30 (4H, t, J = 7.7 Hz), 6.63 (2H, d, J = 8.2 Hz), 6.89 (1H, d, J = 15.9 Hz), 7.19 (1H, d, J = 15.9 Hz), 7.40 (2H, d, J = 8.8 Hz), 7.59 (2H, d, J = 8.3 Hz), 7.82 (2H, d, J = 8.3 Hz), 9.95 (1H, s)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 29.5, 50.8, 111.6, 122.0, 123.5, 126.1, 128.4, 130.2, 132.7, 134.4, 144.8, 148.5, 191.6
1H-NMR (600 MHz, CDCl3) δ: 0.97 (6H, t, J = 7.1 Hz), 1.34-1.41 (4H, m), 1.57-1.62 (4H, m), 1.80 (6H, s), 3.31 (4H, t, J = 7.7 Hz), 6.63 (2H, d, J = 8.8 Hz), 6.88 (1H, d, J = 15.9 Hz), 7.00 (1H, d, J = 16.5 Hz), 7.21 (1H, d, J = 16.5 Hz), 7.41 (2H, d, J = 8.8 Hz), 7.54 (2H, d, J = 8.8 Hz), 7.59 (2H, d, J = 8.8 Hz), 7.61 (1H, d, J = 15.9 Hz)
13C-NMR (150 MHz, CDCl3) δ: 14.0, 20.3, 26.6, 29.5, 50.8, 57.2, 97.4, 98.7, 110.5, 111.1, 111.6, 111.9, 113.3, 121.7, 123.5, 126.7, 128.7, 129.8, 131.6, 132.9, 134.8, 147.1, 148.7, 173.7, 175.5
実施例1~15及び比較例1~2で得られた化合物について、以下のように熱分解開始温度(Td)、最大吸収スペクトル(λmax)、超分極率(β)を測定した。結果を表6及び表7に示す。
熱分解開始温度(Td)は、株式会社リガク製差動型示差熱天秤装置TG8120を使用し、測定試料5mg、基準試料はAl2O3、窒素雰囲気下、昇温速度10℃/分の条件で測定を行った。
超分極率(β)は、参考論文(“Intermolecular Coupling Enhancement of the Molecular Hyperpolarizability in Multichromophoric Dipolar Dendrons”, Shiyoshi Yokoyama, Tatsuo Nakahama, Akira Otomo, and Shinro Mashiko, Journal of the American Chemical Society, vol. 122, pages 3174-3181 (2000).)に記載の方法と同様に行った。レーザー光源は、アイシン精機株式会社製エルビウムドープファイバーレーザー(フェムトライトC-40-SP,BS-60、波長1.56μm、パルス幅100fsec、繰り返し50MHz、出力60mW)を用いた。光検出器は、浜松ホトニクス株式会社製光電子増倍管(R3896)を用いた。非線形光学化合物を1,4-ジオキサンに5μmol/lの濃度に調整し測定試料とした。参考論文に記載の方法でレーザー光を照射し、発生する第2光調波強度を光電子増倍管で検出し、参照試料の超分極率と比較することによって非線形光学化合物の超分極率(βr)を求めた。参照試料としては、溶媒として用いたクロロホルムの超分極率(-0.49×10-30esu)、または参考論文(“Pyrroline Chromophores for Electro-Optics”,Sei-Hum Jang,Jingdong Luo,Neil M.Tucker,Amalia Leclercq,Egbert Zojer,Marnie A.Haller,Tae-Dong Kim,Jae-Wook Kang,Kimberly Firestone,Denise Bale,David Lao,Jason B. Benedict,Dawn Cohen,Werner Kaminsky,Bart Kahr,Jean-Juc Bredas, Philip Reid,Larry R. Dalton,and Alex K.-Y.Jen,Chemistry of Materials,18(13),pages2982-2988 (2006))に記載の非線形光学化合物(No.5b)の超分極率を用いた。
非線形光学化合物の極大吸収波長(λmax)は、1,4-ジオキサン中に非線形光学化合物を5μmol/lの濃度で調整し、株式会社島津製作所製紫外可視分光光度計(UVmini‐1240)を用いて測定した。
実施例1~6、13~18、27-1~27-28、28~30、35~57及び比較例1で得られた化合物について、以下のように熱分解開始温度(Td)、最大吸収スペクトル(λmax)、超分極率(β)を測定した。結果を表8に示す。
熱分解開始温度(Td)は、株式会社リガク製差動型示差熱天秤装置TG8120を使用し、測定試料5mg、基準試料はAl2O3、窒素雰囲気下、昇温速度10℃/分の条件で測定を行った。
超分極率(β)の測定および非共鳴超分極率(β0)の算出は、参考論文(“Intermolecular Coupling Enhancement of the Molecular Hyperpolarizability in Multuchromophoric Dipolar Dendron”, Shiyoshi Yokoyama, Tatsuo Nakahama, Akira Otomo, and Shinro Mashiko, Journal of the American Chemical Society, vol. 122, pages 3174-318 (2000).)に記載の方法と同様に行った。レーザー光源は、スペクトラフィジックス社製Nd:YAGレーザーOPOシステム(Quanta Ray Nd:YAGレーザー、OPO versaScan、波長1.90μm、パルス幅20nsec、繰り返し10Hz、出力90mW)を用いた。光検出器は、浜松ホトニクス株式会社製光電子増倍管(R2658)を用いた。非線形光学化合物をクロロホルムに5μmol/lの濃度に調整し測定試料とした。参考論文に記載の方法でレーザー光を照射し、発生する第2光調波強度を光電子増倍管で検出し、参照試料の超分極率と比較することによって非線形光学化合物の超分極率(β)を求めた。参照試料としては、溶媒として用いたクロロホルムの超分極率(-0.49×10-30esu)、または参考論文(“Pyrroline Chromophores for Electro-Optics”,Sei-Hum Jang,Jingdong Luo,Neil M.Tucker,Amalia Leclercq,Egbert Zojer,Marnie A.Haller,Tae-Dong Kim,Jae-Wook Kang,Kimberly Firestone,Denise Bale,David Lao,Jason B. Benedict,Dawn Cohen,Werner Kaminsky,Bart Kahr,Jean-Juc Bredas, Philip Reid,Larry R. Dalton,and Alex K.-Y.Jen,Chemistry Materials,vol.28,pages2982-2988 (2006))に記載の非線形光学化合物(No.5b)の超分極率を用いた。
非線形光学化合物の極大吸収波長(λmax)は、クロロホルム中に非線形光学化合物を10μmol/lの濃度で調整し、株式会社日立製作所製紫外可視分光光度計(U-4000)を用いて測定した。
Claims (46)
- ドナー構造部Dとπ共役ブリッジ構造部Bとアクセプター構造部Aを含む発色団において、当該ドナー構造部Dが置換オキシ基で置換されたアリール基を含み、当該アクセプター構造部Aが-SO2-を含まないことを特徴とする発色団。
- 当該置換オキシ基が当該アリール基のオルト位の炭素原子に結合している、又は、オルト位及びパラ位の炭素原子に結合している、請求項1に記載の発色団。
- 当該アリール基がさらに置換基を有していてもよいアミノ基で置換されていてもよい、請求項1又は2に記載の発色団。
- 当該π共役ブリッジ構造部Bのπ共役が炭素-炭素共役である、請求項1~3のいずれかに記載の発色団。
- 式D-B-A(ここで、Dはドナー構造部Dを示す;Bはπ共役ブリッジ構造部Bを示す;Aはアクセプター構造部Aを示す)で表わされる、請求項1~4のいずれかに記載の発色団。
- ドナー構造部Dが、式D-1
(ここで、
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい)
で表わされる、請求項1~5のいずれかに記載の発色団。 - ドナー構造部Dが、式D-1-1
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい)
で表わされる、請求項1~6のいずれかに記載の発色団。 - ドナー構造部Dが、式D-1-2
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は、結合する窒素原子と一緒になって、置換基を有していてもよい飽和複素環を形成する、又は
RD 4及びRD 5は、結合する窒素原子、当該窒素原子が結合するアリールの炭素原子及び当該炭素原子に隣接するアリールの炭素原子と一緒になって、ヘテロ原子として窒素原子を含み、置換基を有していてもよい複素環を形成する)
で表わされる、請求項1~6のいずれかに記載の発色団。 - ドナー構造部Dが、式D-2
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい)
で表わされる、請求項1、2、4及び5のいずれかに記載の発色団。 - ドナー構造部Dが、式D-2-1
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい)
で表わされる、請求項1、2、4、5及び9のいずれかに記載の発色団。 - 式I-1
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、若しくは、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~6及び11~13のいずれかに記載の発色団。 - 式I-1-1
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、置換基を有していてもよい複素環を形成する、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~7及び11~14のいずれかに記載の発色団。 - 式I-2
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RB 1及びRB 2はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1、2、4、5、9及び11~13のいずれかに記載の発色団。 - 式II-1
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~6、11、12及び17のいずれかに記載の発色団。 - 式II-1-1
π1及びπ2はそれぞれ独立して、同一又は異なる炭素-炭素共役π結合を示し、それぞれ同一又は異なる置換基を有していてもよい;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2、RB 3及びRB 4はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~7、11、12、17及び18のいずれかに記載の発色団。 - 式III-1
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2及びRB 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~6、11、12及び20のいずれかに記載の発色団。 - 式III-1-1
m及びm’はそれぞれ独立して、0~3の整数を示す;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RB 1、RB 2及びRB 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリール基、アラルキル基、アリールオキシ基又はアラルキルオキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~7、11、12、20及び21のいずれかに記載の発色団。 - 式IV-1-a
nは1~5の整数を示す;
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~6、11、12及び23のいずれかに記載の発色団。 - 式IV-1-b
RD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、アルキル基、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリール基の隣接する炭素原子に結合するとき、RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよい;
RD 4及びRD 5はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基、ヒドロキシアルキル基、アシルオキシアルキル基、シリルオキシアルキル基、アミノアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい、又は
RD 4及びRD 5は結合する窒素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい、又は
(a)RD 2及び-NRD 4RD 5、並びに、(b)RD 3及び-NRD 4RD 5はそれぞれ独立して、結合する炭素原子と一緒になって、ヘテロ原子として窒素原子を含む複素環を形成し、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1~6、11、12及び23のいずれかに記載の発色団。 - 式IV-2-a
nは1~5の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1、2、4、5、9、11、12及び23のいずれかに記載の発色団。 - 式IV-2-b
nは1~5の整数を示す;
RD 1、RD 2及びRD 3のうち少なくとも1つはそれぞれ独立して、アルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、それ以外はそれぞれ独立して、水素原子又はアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、
ここで、RD 2及びRD 3がそれぞれドナー構造部Dのアリールの隣接する炭素原子に結合するとき、
(1)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、置換基を有していてもよい環を形成してもよく、このときRD 1はアルコキシ基、アリールオキシ基、アラルキルオキシ基、シリルオキシ基、アルケニルオキシ基、アルケニルカルボニルオキシ基、アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい、又は、
(2)RD 2及びRD 3は隣接する2つの炭素原子と一緒になって、ヘテロ原子として酸素原子を含む複素環を形成してもよく、当該複素環は置換基を有していてもよい;
RA 1及びRA 2はそれぞれ独立して、水素原子、アルキル基、ハロアルキル基又はアリール基を示し、それぞれ同一又は異なる置換基を有していてもよい)
で表される、請求項1、2、4、5、9、11、12及び23のいずれかに記載の発色団。 - RD 1はC1~6アルコキシ基、ベンジルオキシ基、シリルオキシ基、C2~6アルケニルオキシ基、C2~6アルケニルカルボニルオキシ基、C3~6アルキニルオキシ基又はヒドロキシ基を示し、置換基を有していてもよい;
RD 2及びRD 3はそれぞれ独立して、水素原子、C1~6アルコキシ基、ベンジルオキシ基、シリルオキシ基、C2~6アルケニルオキシ基、C2~6アルケニルカルボニルオキシ基、C3~6アルキニルオキシ基又はヒドロキシ基を示し、それぞれ同一又は異なる置換基を有していてもよい、請求項6~10、14~16、18、19、21、22及び24~27のいずれかに記載の発色団。 - RD 4及びRD 5はそれぞれ独立して、アルキル基、ヒドロキシアルキル基又はシリルオキシアルキル基を示し、それぞれ同一又は異なる置換基を有していてもよい、請求項6~8、14、15、18、19、21、22、24及び25のいずれかに記載の発色団。
- RA 1及びRA 2はそれぞれ独立して、メチル基、トリフルオロメチル基又はフェニル基を示し、それぞれ同一又は異なる置換基を有していてもよい、請求項11、14~16、18、19、21、22、及び24~27のいずれかに記載の発色団。
- 請求項1~41のいずれかに記載の発色団と、当該発色団が分散されるホスト材料とを含有する非線形光学材料。
- 前記ホスト材料は、前記発色団との間に共有結合を形成し得る反応性官能基を有する樹脂を含有し、前記発色団の少なくとも一部は、当該樹脂と結合している請求項42に記載の非線形光学材料。
- 請求項1~41のいずれかに記載の発色団又は請求項42若しくは43に記載の非線形光学材料によって形成される膜を有する非線形光学素子。
- 請求項1~41のいずれかに記載の発色団又は請求項42若しくは43に記載の非線形光学材料によって形成される光導波路を有する非線形光学素子。
- 請求項1~41のいずれかに記載の発色団を含む非線形光学素子。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201080035813.4A CN102482504B (zh) | 2009-08-24 | 2010-08-24 | 二阶非线性光学化合物及含有它的非线性光学元件 |
| US13/391,948 US8846955B2 (en) | 2009-08-24 | 2010-08-24 | Second-order nonlinear optical compound and nonlinear optical element comprising the same |
| JP2011528785A JP5945905B2 (ja) | 2009-08-24 | 2010-08-24 | 2次非線形光学化合物及びそれを含む非線形光学素子 |
| US14/317,192 US9488755B2 (en) | 2009-08-24 | 2014-06-27 | Second-order nonlinear optical compound and nonlinear optical element comprising the same |
| US15/208,976 US9977150B2 (en) | 2009-08-24 | 2016-07-13 | Second-order nonlinear optical compound and nonlinear optical element comprising the same |
| US15/944,019 US10754064B2 (en) | 2009-08-24 | 2018-04-03 | Second-order nonlinear optical compound and nonlinear optical element comprising the same |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009192738 | 2009-08-24 | ||
| JP2009-192738 | 2009-08-24 | ||
| JP2010-104951 | 2010-04-30 | ||
| JP2010104951 | 2010-04-30 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/391,948 A-371-Of-International US8846955B2 (en) | 2009-08-24 | 2010-08-24 | Second-order nonlinear optical compound and nonlinear optical element comprising the same |
| US14/317,192 Division US9488755B2 (en) | 2009-08-24 | 2014-06-27 | Second-order nonlinear optical compound and nonlinear optical element comprising the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011024774A1 true WO2011024774A1 (ja) | 2011-03-03 |
Family
ID=43627879
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2010/064216 WO2011024774A1 (ja) | 2009-08-24 | 2010-08-24 | 2次非線形光学化合物及びそれを含む非線形光学素子 |
Country Status (4)
| Country | Link |
|---|---|
| US (4) | US8846955B2 (ja) |
| JP (2) | JP5945905B2 (ja) |
| CN (2) | CN104610774B (ja) |
| WO (1) | WO2011024774A1 (ja) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105441062A (zh) * | 2014-09-29 | 2016-03-30 | 中国科学院大连化学物理研究所 | 一种人肠道羧酸酯酶活性检测的荧光探针底物及其应用 |
| JP2017521431A (ja) * | 2014-07-08 | 2017-08-03 | バレント・バイオサイエンシーズ・コーポレイションValent Biosciences Corporation | 3’置換−アブシジン酸誘導体 |
| WO2018003842A1 (ja) | 2016-06-29 | 2018-01-04 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー |
| WO2019039530A1 (ja) | 2017-08-24 | 2019-02-28 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー層を含む非線形光学用積層体及びその製造方法 |
| WO2019151318A1 (ja) | 2018-01-30 | 2019-08-08 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー |
| WO2019168186A1 (ja) | 2018-03-02 | 2019-09-06 | 帝人株式会社 | 電気光学効果を有するポリカーボネート及びその製造方法、並びにそのポリカーボネートを用いた光制御素子 |
| WO2022070667A1 (ja) | 2020-09-30 | 2022-04-07 | 国立研究開発法人情報通信研究機構 | 判定方法、光制御素子、及びその製造方法 |
| WO2023021921A1 (ja) | 2021-08-20 | 2023-02-23 | 国立研究開発法人情報通信研究機構 | ポリマー及び光制御素子 |
| WO2024024154A1 (ja) * | 2022-07-28 | 2024-02-01 | 株式会社村田製作所 | 電気光学ポリマー |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013172342A1 (ja) * | 2012-05-14 | 2013-11-21 | 国立大学法人九州大学 | 有機非線形光学化合物を含むポリマー組成物 |
| WO2015026370A1 (en) * | 2013-08-23 | 2015-02-26 | Empire Technology Development Llc | Cross-linked co-polymers for making optoelectronic devices |
| CN104804723A (zh) * | 2014-01-28 | 2015-07-29 | 中国科学院大连化学物理研究所 | 一类基于dcdhf发色团的溶致变色荧光探针、其制备及应用 |
| CN106279133A (zh) * | 2015-06-11 | 2017-01-04 | 中国科学院理化技术研究所 | 有机二阶非线性光学发色团及合成方法和应用 |
| CN105418560B (zh) * | 2015-11-09 | 2017-11-07 | 晋江尚京富本环保科技有限公司 | 长波长肼比色荧光探针及其制备方法 |
| EP3431515B1 (en) * | 2016-03-17 | 2021-04-21 | Kyushu University | Nonlinear optically active copolymer into which alicyclic group is introduced |
| CN107663384B (zh) | 2016-07-20 | 2020-05-12 | 上海高驰资产管理有限公司 | 一种荧光染料及其制备方法和用途 |
| CN108610325A (zh) * | 2018-05-08 | 2018-10-02 | 成都师范学院 | 一种基于二噻吩乙烯的非线性光学发色团及其制备和用途 |
| CN113582950B (zh) * | 2021-08-10 | 2022-07-29 | 中国科学院深圳先进技术研究院 | 一种比率型多硫化氢荧光探针及其制备方法和应用 |
| CN115960093A (zh) * | 2021-10-09 | 2023-04-14 | 香港城市大学深圳研究院 | 一种光学发色团化合物及含其的复合材料、薄膜和光电集成器件 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01179041A (ja) * | 1987-12-30 | 1989-07-17 | Konica Corp | イエロー染料を含有するハロゲン化銀写真感光材料 |
| JPH04116640A (ja) * | 1990-09-07 | 1992-04-17 | Konica Corp | 固体微粒子分散物を含有するハロゲン化銀写真感光材料 |
| JPH04226393A (ja) * | 1990-04-20 | 1992-08-17 | Agfa Gevaert Nv | 黒色着色熱染料昇華転写供与体材料 |
| JPH04265792A (ja) * | 1990-10-31 | 1992-09-21 | Eastman Kodak Co | 感熱カラープルーフ用イエロー色素混合物 |
| US6067186A (en) * | 1998-07-27 | 2000-05-23 | Pacific Wave Industries, Inc. | Class of high hyperpolarizability organic chromophores and process for synthesizing the same |
| JP2004501159A (ja) * | 2000-06-16 | 2004-01-15 | コーニング インコーポレイテッド | 高分子薄膜のための新規な発色団並びにそれを有してなる光導波路および素子 |
| JP2004243596A (ja) * | 2003-02-12 | 2004-09-02 | Mitsui Chemicals Inc | 光記録媒体および3,3−ジ置換−2−シアノアクリル酸誘導体 |
| WO2005073697A1 (en) * | 2004-01-20 | 2005-08-11 | The Curators Of The University Of Missouri | Local flow and shear stress sensor based on molecular rotors |
| WO2005072216A2 (en) * | 2004-01-20 | 2005-08-11 | The Curators Of The University Of Missouri | Supported molecular biofluid viscosity sensors for in vitro and in vivo use |
Family Cites Families (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3551502A (en) * | 1968-02-10 | 1970-12-29 | Meiji Seika Co | (2-propynyloxy)-beta-nitro-styrenes |
| ZA724018B (en) | 1971-06-29 | 1973-07-25 | Ciba Geigy Ag | Process for the production of new o-substituted phenylethylguanidines |
| JPS61169832A (ja) * | 1985-01-23 | 1986-07-31 | Fuji Photo Film Co Ltd | ハロゲン化銀写真感光材料 |
| JPS6330853A (ja) * | 1986-07-25 | 1988-02-09 | Canon Inc | 電子写真感光体 |
| JPH0211660A (ja) * | 1988-04-20 | 1990-01-16 | Eastman Kodak Co | ポリマービーズ |
| FR2636441B1 (fr) * | 1988-09-15 | 1993-11-05 | Rhone Poulenc Chimie | Composes organiques actif en optique non lineaire et dispositifs electrooptiques les contenant |
| FR2636745B1 (fr) * | 1988-09-16 | 1990-11-09 | Rhone Poulenc Chimie | Polymeres et materiaux les contenant, actifs en optique non lineaire, procedes de fabrication de ces polymeres et materiaux et dispositif optoelectrique les contenant |
| JP2876053B2 (ja) * | 1989-06-30 | 1999-03-31 | 日本電信電話株式会社 | 光非線形材料およびその製造方法 |
| JPH03109554A (ja) * | 1989-09-25 | 1991-05-09 | Fuji Electric Co Ltd | 電子写真用感光体 |
| DE4244195A1 (de) * | 1992-12-24 | 1994-06-30 | Basf Ag | Verfahren zur Herstellung von strukturierten Polymerschichten mit nichtlinear optischen Eigenschaften |
| TW243501B (en) * | 1994-07-22 | 1995-03-21 | Akzo Nobel Nv | Bleachable optical waveguide component |
| JPH08184866A (ja) * | 1994-12-28 | 1996-07-16 | Toyo Ink Mfg Co Ltd | 有機非線形光学材料用スチルベン誘導体及びその使用 |
| US5578416A (en) * | 1995-11-20 | 1996-11-26 | Eastman Kodak Company | Cinnamal-nitrile dyes for laser recording element |
| US6555027B2 (en) | 1998-07-27 | 2003-04-29 | Pacific Wave Industries, Inc. | Second-order nonlinear optical chromophores containing dioxine and/or bithiophene as conjugate bridge and devices incorporating the same |
| US6361717B1 (en) | 1998-07-27 | 2002-03-26 | Pacific Wave Industries, Inc. | Sterically stabilized second-order nonlinear optical chromophores and devices incorporating the same |
| US6348992B1 (en) | 1998-07-27 | 2002-02-19 | Pacific Wave Industries, Inc. | Sterically stabilized polyene-bridged second-order nonlinear optical chromophores and devices incorporating the same |
| US6616865B1 (en) | 1998-07-27 | 2003-09-09 | Pacific Wave Industries, Inc. | Sterically stabilized second-order nonlinear optical chromophores with improved stability and devices incorporating the same |
| US6652779B1 (en) | 1998-07-27 | 2003-11-25 | Pacific Wave Industries, Inc. | Polymers containing polyene-based second-order nonlinear optical chromophores and devices incorporating the same |
| US6584266B1 (en) | 2000-06-16 | 2003-06-24 | Corning Incorporated | Chromophores for polymeric thin films and optical waveguides and devices comprising the same |
| JP4778165B2 (ja) | 2001-07-19 | 2011-09-21 | 型研精工株式会社 | トランスファ送り装置 |
| US6931164B2 (en) * | 2002-11-20 | 2005-08-16 | Optimer Photonics, Inc. | Waveguide devices incorporating Kerr-based and other similar optically functional mediums |
| AU2003294380A1 (en) * | 2003-05-30 | 2005-01-04 | The Arizona Board Of Regents On Behalf Of The University Of Arizona | Third-order optical autocorrelator for time-domain opertion at the telecommunication wavelenghts |
| EP1742906A4 (en) * | 2004-05-03 | 2010-09-29 | Optimer Photonics Inc | NONLINEAR OPTICALLY ACTIVE MOLECULES, THEIR COMPOSITION AND USE |
| CN100400537C (zh) * | 2005-05-09 | 2008-07-09 | 北京大学 | 一种红色荧光染料化合物及其制备方法与应用 |
| JP2007057940A (ja) * | 2005-08-25 | 2007-03-08 | Fuji Xerox Co Ltd | 非線形光学材料製造用原料溶液、非線形光学材料、及び非線形光学素子 |
| EP2090576A1 (en) * | 2008-02-01 | 2009-08-19 | Merz Pharma GmbH & Co.KGaA | 6-halo-pyrazolo[1,5-a]pyridines, a process for their preparation and their use as metabotropic glutamate receptor (mGluR) modulators |
| US20100310456A1 (en) | 2009-06-04 | 2010-12-09 | General Electric Company | Imaging of myelin basic protein |
| US8658129B2 (en) | 2009-06-04 | 2014-02-25 | General Electric Company | Agents and methods for the imaging of myelin basic protein |
| US8617515B2 (en) | 2009-06-04 | 2013-12-31 | General Electric Company | Imaging of myelin basic protein |
| US7885011B1 (en) | 2009-08-19 | 2011-02-08 | General Electric Company | Objective for optical imaging systems |
| US8169696B2 (en) | 2009-06-04 | 2012-05-01 | General Electric Company | Systems for intraoperative nerve imaging |
-
2010
- 2010-08-24 JP JP2011528785A patent/JP5945905B2/ja active Active
- 2010-08-24 WO PCT/JP2010/064216 patent/WO2011024774A1/ja active Application Filing
- 2010-08-24 CN CN201410799080.1A patent/CN104610774B/zh active Active
- 2010-08-24 CN CN201080035813.4A patent/CN102482504B/zh active Active
- 2010-08-24 US US13/391,948 patent/US8846955B2/en active Active
-
2014
- 2014-06-27 US US14/317,192 patent/US9488755B2/en active Active
-
2015
- 2015-07-28 JP JP2015148928A patent/JP6304823B2/ja active Active
-
2016
- 2016-07-13 US US15/208,976 patent/US9977150B2/en active Active
-
2018
- 2018-04-03 US US15/944,019 patent/US10754064B2/en active Active
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01179041A (ja) * | 1987-12-30 | 1989-07-17 | Konica Corp | イエロー染料を含有するハロゲン化銀写真感光材料 |
| JPH04226393A (ja) * | 1990-04-20 | 1992-08-17 | Agfa Gevaert Nv | 黒色着色熱染料昇華転写供与体材料 |
| JPH04116640A (ja) * | 1990-09-07 | 1992-04-17 | Konica Corp | 固体微粒子分散物を含有するハロゲン化銀写真感光材料 |
| JPH04265792A (ja) * | 1990-10-31 | 1992-09-21 | Eastman Kodak Co | 感熱カラープルーフ用イエロー色素混合物 |
| US6067186A (en) * | 1998-07-27 | 2000-05-23 | Pacific Wave Industries, Inc. | Class of high hyperpolarizability organic chromophores and process for synthesizing the same |
| JP2004501159A (ja) * | 2000-06-16 | 2004-01-15 | コーニング インコーポレイテッド | 高分子薄膜のための新規な発色団並びにそれを有してなる光導波路および素子 |
| JP2004243596A (ja) * | 2003-02-12 | 2004-09-02 | Mitsui Chemicals Inc | 光記録媒体および3,3−ジ置換−2−シアノアクリル酸誘導体 |
| WO2005073697A1 (en) * | 2004-01-20 | 2005-08-11 | The Curators Of The University Of Missouri | Local flow and shear stress sensor based on molecular rotors |
| WO2005072216A2 (en) * | 2004-01-20 | 2005-08-11 | The Curators Of The University Of Missouri | Supported molecular biofluid viscosity sensors for in vitro and in vivo use |
Non-Patent Citations (3)
| Title |
|---|
| COLLECT. CZECH. CHEM. COMMUN., vol. 54, no. 3, 1989, pages 1666 - 1674 * |
| JOURNAL OF MOLECULAR STRUCTURE(THEOCHEM), vol. 543, 2001, pages 129 - 146 * |
| PIGMENT & RESIN TECHNOLOGY, vol. 33, no. 6, 2004, pages 370 - 374 * |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017521431A (ja) * | 2014-07-08 | 2017-08-03 | バレント・バイオサイエンシーズ・コーポレイションValent Biosciences Corporation | 3’置換−アブシジン酸誘導体 |
| CN105441062A (zh) * | 2014-09-29 | 2016-03-30 | 中国科学院大连化学物理研究所 | 一种人肠道羧酸酯酶活性检测的荧光探针底物及其应用 |
| CN105441062B (zh) * | 2014-09-29 | 2017-12-01 | 中国科学院大连化学物理研究所 | 一种人肠道羧酸酯酶活性检测的荧光探针底物及其应用 |
| US11236188B2 (en) | 2016-06-29 | 2022-02-01 | National Institute Of Information And Communications Technology | Electro-optic polymer |
| JPWO2018003842A1 (ja) * | 2016-06-29 | 2019-06-06 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー |
| WO2018003842A1 (ja) | 2016-06-29 | 2018-01-04 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー |
| JP7161195B2 (ja) | 2016-06-29 | 2022-10-26 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー |
| WO2019039530A1 (ja) | 2017-08-24 | 2019-02-28 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー層を含む非線形光学用積層体及びその製造方法 |
| US11194225B2 (en) | 2017-08-24 | 2021-12-07 | National Institute Of Information And Communications Technology | Laminate for non-linear optics containing electro-optic polymer layer and method for producing same |
| WO2019151318A1 (ja) | 2018-01-30 | 2019-08-08 | 国立研究開発法人情報通信研究機構 | 電気光学ポリマー |
| WO2019168186A1 (ja) | 2018-03-02 | 2019-09-06 | 帝人株式会社 | 電気光学効果を有するポリカーボネート及びその製造方法、並びにそのポリカーボネートを用いた光制御素子 |
| WO2022070667A1 (ja) | 2020-09-30 | 2022-04-07 | 国立研究開発法人情報通信研究機構 | 判定方法、光制御素子、及びその製造方法 |
| WO2023021921A1 (ja) | 2021-08-20 | 2023-02-23 | 国立研究開発法人情報通信研究機構 | ポリマー及び光制御素子 |
| WO2024024154A1 (ja) * | 2022-07-28 | 2024-02-01 | 株式会社村田製作所 | 電気光学ポリマー |
Also Published As
| Publication number | Publication date |
|---|---|
| US10754064B2 (en) | 2020-08-25 |
| US20180224577A1 (en) | 2018-08-09 |
| US20120172599A1 (en) | 2012-07-05 |
| JP2016006079A (ja) | 2016-01-14 |
| JP5945905B2 (ja) | 2016-07-05 |
| JPWO2011024774A1 (ja) | 2013-01-31 |
| CN102482504A (zh) | 2012-05-30 |
| JP6304823B2 (ja) | 2018-04-04 |
| US9977150B2 (en) | 2018-05-22 |
| CN104610774A (zh) | 2015-05-13 |
| US20150048284A1 (en) | 2015-02-19 |
| CN104610774B (zh) | 2017-07-18 |
| US20160318889A1 (en) | 2016-11-03 |
| CN102482504B (zh) | 2016-01-20 |
| US9488755B2 (en) | 2016-11-08 |
| US8846955B2 (en) | 2014-09-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6304823B2 (ja) | 2次非線形光学化合物及びそれを含む非線形光学素子 | |
| EP1313729B1 (en) | Design and synthesis of thiophene derived nlo materials for electro-optic applications | |
| US7307173B1 (en) | Pyrroline chromophores | |
| US7425643B1 (en) | Electron acceptors for nonlinear optical chromophores | |
| Huo et al. | Novel nonlinear optical push–pull fluorene dyes chromophore as promising materials for telecommunications | |
| Liu et al. | Synthesis of julolidine-containing nonlinear optical chromophores: Achieving excellent electro-optic activity by optimizing the bridges and acceptors | |
| US20040135130A1 (en) | Nonlinear optical compounds and methods for their preparation | |
| CN109438459A (zh) | 一种有机二阶非线性光学发色团及其合成方法和应用 | |
| Farsadpour et al. | New class of hyperpolarizable push–pull organic chromophores by applying a novel and convenient synthetic strategy | |
| CN105968130B (zh) | 一种中位含咔唑及桥联基团的双中心氟化硼络合二吡咯甲川衍生物及其制备方法 | |
| US8648332B2 (en) | Phenyltetraene-based nonlinear optical chromophores | |
| CN103373990B (zh) | 含有三蝶烯刚性立体结构的发色团分子及其合成方法和应用 | |
| CN104558005A (zh) | 微波合成具有D-π-A结构的二阶非线性光学发色团的方法 | |
| Li et al. | Synthesis and nonlinear optical properties of novel conjugated small molecules based on coumarin donor | |
| Casmier et al. | New paradigm in NLO chromophore design through a gradient bridge concept | |
| Chen et al. | Preparation and nonlinear optical properties of hybrid sol–gel films doped with triphenylamino-substituted chromophores | |
| EP1760080A1 (en) | Design and synthesis of thiophene derived NLO materials for electro-optic applications | |
| CN117126131A (zh) | 一种γ‐噻喃酮衍生物的高效合成方法 | |
| Shi et al. | Synthesis and characterization of thiophene-containing chromophores for nonlinear optical (NLO) materials |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080035813.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10811824 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011528785 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13391948 Country of ref document: US |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 10811824 Country of ref document: EP Kind code of ref document: A1 |