WO2010146912A1 - 空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物、構造体及びその施工方法 - Google Patents
空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物、構造体及びその施工方法 Download PDFInfo
- Publication number
- WO2010146912A1 WO2010146912A1 PCT/JP2010/056010 JP2010056010W WO2010146912A1 WO 2010146912 A1 WO2010146912 A1 WO 2010146912A1 JP 2010056010 W JP2010056010 W JP 2010056010W WO 2010146912 A1 WO2010146912 A1 WO 2010146912A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- meth
- resin composition
- acrylate resin
- air
- polyester
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F290/00—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups
- C08F290/02—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups on to polymers modified by introduction of unsaturated end groups
- C08F290/04—Polymers provided for in subclasses C08C or C08F
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D167/00—Coating compositions based on polyesters obtained by reactions forming a carboxylic ester link in the main chain; Coating compositions based on derivatives of such polymers
- C09D167/06—Unsaturated polyesters having carbon-to-carbon unsaturation
- C09D167/07—Unsaturated polyesters having carbon-to-carbon unsaturation having terminal carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F290/00—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups
- C08F290/02—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups on to polymers modified by introduction of unsaturated end groups
- C08F290/06—Polymers provided for in subclass C08G
- C08F290/061—Polyesters; Polycarbonates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F290/00—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups
- C08F290/02—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups on to polymers modified by introduction of unsaturated end groups
- C08F290/06—Polymers provided for in subclass C08G
- C08F290/064—Polymers containing more than one epoxy group per molecule
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L33/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- C08L33/04—Homopolymers or copolymers of esters
- C08L33/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur, or oxygen atoms in addition to the carboxy oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur or oxygen atoms in addition to the carboxy oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D151/00—Coating compositions based on graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Coating compositions based on derivatives of such polymers
- C09D151/08—Coating compositions based on graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Coating compositions based on derivatives of such polymers grafted on to macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
Definitions
- the present invention relates to an air-drying polyester (meth) acrylate resin composition excellent in non-foaming property and room temperature curability used for a top coat in the field of civil engineering and construction, a structure on which it is constructed, and a construction method therefor.
- an acrylic monomer having a high boiling point is used instead of a styrene monomer, and a polymer such as an epoxy acrylate resin, a polyester acrylate resin, or a urethane acrylate resin is dissolved to achieve environmental friendliness and low VOC.
- a polymer such as an epoxy acrylate resin, a polyester acrylate resin, or a urethane acrylate resin is dissolved to achieve environmental friendliness and low VOC.
- These resins are cured at room temperature with a combination of a ketone peroxide-based curing agent such as methyl ethyl ketone peroxide (hereinafter MEKPO) and a metal soap-based cobalt accelerator such as cobalt naphthenate.
- MEKPO methyl ethyl ketone peroxide
- Patent Document 1 As a method for suppressing foam generation, use of an organic peroxide other than MEKPO has been proposed (Patent Document 1). However, since the curability is not sufficient, there is a problem that a special curing accelerator is necessary. It was. In addition, a method for suppressing foam by adding a monoalkyl ether (monomethyl malate) of an unsaturated dibasic acid to the resin composition has been proposed. (Patent Document 2) However, in this case, there is a problem that the cobalt-based accelerator is deactivated during storage, the gelation time is delayed, and it is not cured at room temperature.
- Patent Document 2 As a method for suppressing foam generation, use of an organic peroxide other than MEKPO has been proposed (Patent Document 1). However, since the curability is not sufficient, there is a problem that a special curing accelerator is necessary. It was. In addition, a method for suppressing foam by adding a monoalkyl ether (monomethyl malate) of an unsaturated di
- An object of the present invention is to provide a foaming agent in a curing reaction process in an air-drying polyester (meth) acrylate resin composition of a curing system of a ketone peroxide-based curing agent and a cobalt-based curing accelerator used for a top coat in the field of civil engineering and construction. Due to its non-foaming property, it can be applied or laminated without any bubbles remaining in the cured coating, causing problems with room temperature curability such as delayed gelation even after the composition has been stored for about 1 month. It is in providing the structure which applied the resin composition which was not excellent, and the construction method, and its construction method.
- the present invention relates to a polyester (meth) acrylate resin (A) and a (meth) acrylic monomer (B) having a structural unit derived from a cycloaliphatic unsaturated dibasic acid and a structural unit derived from an ether bond-containing glycol.
- a cobalt-based curing accelerator (C) wherein the ratio of the cycloaliphatic unsaturated dibasic acid in the total dibasic acid of the resin (A) is 5 mol% or more and less than 50 mol%, and 1 mol% to 10 mol% of at least one unsaturated dibasic acid of maleic acid and maleic anhydride is used in all dibasic acids.
- the present invention has, as the polyester structure, a structural unit derived from a cycloaliphatic unsaturated dibasic acid and a structural unit derived from an ether bond-containing glycol, and the cyclic aliphatic unsaturated dibasic acid in all dibasic acids.
- the ratio is 5 mol% or more and less than 50 mol%, and 1 mol% to 10 mol% of at least one unsaturated dibasic acid of maleic acid and maleic anhydride is used in all dibasic acids.
- the air-drying polyester (meth) acrylate resin (A) of the present invention has two or more (meth) acryloyl groups in one molecule at the end of the polyester resin, and is a cyclic aliphatic unsaturated dibasic acid. It is a polyester (meth) acrylate resin having a structure derived from a structural unit derived from and an ether bond-containing glycol, and its air drying property is the inhibition of curing by oxygen molecules due to the introduction of specific functional groups in the resin structure. It means that curing proceeds rapidly even in the air.
- a cycloaliphatic unsaturated dibasic acid plays the role.
- a terminal functional group of a polyester obtained by a polycondensation reaction between a dibasic acid containing a cyclic aliphatic unsaturated dibasic acid and a glycol containing an ether bond-containing glycol is reacted with this. It is obtained by reacting a functional group-containing (meth) acryloyl compound.
- the ratio of the cycloaliphatic unsaturated dibasic acid is 5 mol% or more and less than 50 mol%, preferably 10 mol% to 45 mol%, as the dibasic acid component having a polyester structure.
- an aromatic dibasic acid an alicyclic dibasic acid and an aliphatic dibasic acid, one or more saturated dibasic acids are used in an amount of 50 to 95 mol%, preferably 45 to 89 mol%.
- the other acid component it is preferable to use an aromatic dibasic acid.
- the resin is inferior in the balance between air drying properties and physical properties.
- 1 mol% to 10 mol% of at least one unsaturated dibasic acid of maleic acid and maleic anhydride in all dibasic acid components generation of bubbles during coating can be suppressed.
- it exceeds 10 mol% the cured product will be inferior in tensile strength.
- it is 1 to 8 mol%.
- the number average molecular weight in terms of polystyrene measured by gel permeation chromatography of the polyester (meth) acrylate resin (A) is preferably 500 to 3000, more preferably 500 to 2000.
- Examples of the functional group-containing (meth) acryloyl compound that reacts with the terminal functional group of the air-drying polyester resin include various unsaturated monobasic acids such as glycidyl (meth) acrylate, acrylic acid or methacrylic acid, and Examples thereof include glycidyl esters. Among these, glycidyl (meth) acrylate is preferable, and glycidyl methacrylate is particularly preferable.
- the cycloaliphatic unsaturated dibasic acid contains a derivative thereof, and this structure has two active hydrogens.
- tetrahydrophthalic anhydride endomethylenetetrahydrophthalic anhydride, methyltetrahydroanhydride.
- examples thereof include phthalic acid (cis-3-methyl-4-cyclohexene-cis-1,2-dicarboxylic anhydride), ⁇ -terhinene / maleic anhydride adduct, trans-piperylene / maleic anhydride adduct, and the like.
- Examples of the aliphatic dibasic acid include oxalic acid, succinic acid, malonic acid, glutaric acid, adipic acid, sebacic acid, 1,12-dodecanedioic acid, and the like.
- Examples thereof include hexahydrophthalic acid, hexahydrophthalic anhydride, hexahydroterephthalic acid, hexahydroisophthalic acid and the like.
- the unsaturated dibasic acid means that at least one of maleic acid and maleic anhydride is used in the total dibasic acid component in an amount of 1 mol% to 10 mol%.
- Fumaric acid, itaconic acid, itaconic anhydride and the like can be used in combination. Further, derivatives such as these dialkyl esters can be used.
- the unsaturated dibasic acid functions to increase the effect of suppressing foam generation, and is preferably used in an amount of 1 mol% to 8 mol%.
- the copolymerizability with acrylic monomer is different, and the copolymerizability between glycidyl methacrylate and acrylic monomer is It is superior to the copolymerizability of a saturated dibasic acid and an acrylic monomer. For this reason, when the usage-amount of unsaturated dibasic acid exceeds 10 mol%, since the tensile strength physical property of hardened
- aromatic dibasic acid examples include phthalic anhydride, halogenated phthalic anhydride, orthophthalic acid, isophthalic acid, terephthalic acid and the like, preferably phthalic anhydride.
- ether bond-containing glycol examples include ethylene glycol, diethylene glycol, triethylene glycol, polyethylene glycol, propylene glycol, dipropylene glycol, tripropylene glycol, polypropylene glycol, preferably diethylene glycol, triethylene glycol and polyethylene glycol. it can.
- glycols can be used in combination, for example, 2-methyl-1,3-propanediol, 1,3-butanediol, neopentyl glycol, hydrogenated bisphenol A, 1,4-butanediol, 1,6-hexane Diol, Adduct of bisphenol A and propylene oxide or ethylene oxide, 1,2,3,4-tetrahydroxybutane, glycerin, trimethylolpropane, 1,3-propanediol, 1,2-cyclohexane glycol, 1,3-cyclohexane And glycol, 1,4-cyclohexane glycol, 1,4-cyclohexanedimethanol, para-xylene glycol, bicyclohexyl-4,4′-diol, 2,6-decalin glycol, 2,7-decalin glycol and the like. .
- the polyester (meth) acrylate resin (A) may be used in combination with a resin such as epoxy (meth) acrylate, urethane (meth) acrylate, and other polyester (meth) acrylates, as long as the effects of the present invention are not impaired. good.
- Examples of the (meth) acrylic monomer (B) include methyl acrylate, ethyl acrylate, n-butyl acrylate, isobutyl acrylate, t-butyl acrylate, 2-ethylhexyl acrylate, and n-acrylate.
- phenoxyethyl (meth) acrylate, dicyclopentenyloxyethyl (meth) acrylate, and dicyclopentenyloxyethyl acrylate which have a molecular weight of 180 or more and are less likely to volatilize, have a hydrogen bond and are less likely to volatilize. Even if the 2-hydroxyethyl (meth) acrylate has a small amount of unreacted in the coating film, it is preferable in that it does not easily become TVOC. Further, styrene, vinyl acetate, vinyltoluene, ⁇ -methyltoluene and the like can be used in combination as long as the effects are not impaired.
- a monomer having at least two ethylenically unsaturated groups in one molecule can be used in combination with the (meth) acrylic monomer (B) as long as the effects of the invention are not impaired.
- this monomer in combination, it is possible to improve the wear resistance, scratch resistance, peristaltic resistance, chemical resistance and the like of the cured product surface.
- the monomer having at least two ethylenically unsaturated groups in one molecule include ethylene glycol di (meth) acrylate, 1,2-propylene glycol di (meth) acrylate, and 1,3-butylene glycol diester.
- examples thereof include polyoxyalkylene-glycol di (meth) acrylates such as acrylate polyethylene glycol di (meth) acrylate, and these are used alone or in combination of two or more.
- divinylbenzene, diallyl phthalate, diallyl isophthalate, diallyl tetrabromophthalate, triallyl phthalate and the like can be used as long as the effects of the invention are not impaired.
- the resin composition of the present invention contains cobalt soap as the cobalt-based curing accelerator (C), and examples of the cobalt soap include cobalt naphthenate and cobalt octylate.
- the addition amount of the curing accelerator (C) is preferably 0.01 to 5 parts by mass in the resin composition composed of (A) and (B). Outside this range, long-term storage stability cannot be obtained. This curing accelerator is added in advance to the resin composition.
- curing accelerators other than cobalt-based ones may be added as long as the effects of the present invention are not impaired.
- metal soaps such as zinc octylate, vanadium octylate, copper naphthenate, barium naphthenate
- metal chelates such as vanadium acetyl acetate, cobalt acetyl acetate, iron acetylacetonate, aniline, N, N-dimethylaniline, N, N-diethylaniline, p-toluidine, N, N-dimethyl-p-toluidine, N, N-bis (2-hydroxyethyl) -p-toluidine, 4- (N, N-dimethylamino) benzaldehyde, 4- [ N, N-bis (2-hydroxyethyl) amino] benzaldehyde, 4- (N-methyl-N-hydroxyethylamino) benzaldehyde, N, N-bis
- the resin composition of the present invention is used by adding a ketone peroxide-based curing agent (D) for curing at room temperature.
- the amount used is preferably 0.1 to 6 parts by mass with respect to 100 parts by mass of the total amount of the resin composition.
- the curing agent (D) is preferably methyl ethyl ketone peroxide.
- the resin composition of the present invention uses a radical curing agent other than the curing agent (D), a photo radical initiator, and a polymerization inhibitor in a range that does not impair the effects of the present invention in order to adjust the curing rate.
- radical curing agents other than the above (D) include organic peroxides, specifically, diacyl peroxides, peroxyesters, hydroperoxides, dialkyl peroxides, peroxyketals.
- publicly known ones such as alkyl peresters and percarbonates can be used. Specific examples include benzoyl peroxide.
- Examples of the photo radical initiator that is, a photosensitizer, include benzoin ethers such as benzoin alkyl ether, benzophenones such as benzophenone, benzyl and methyl orthobenzoylbenzoate, benzyl dimethyl ketal, 2,2-diethoxyacetophenone, Acetophenones such as 2-hydroxy-2-methylpropiophenone, 4-isopropyl-2-hydroxy-2-methylpropiophenone, 1,1-dichloroacetophenone, 2-chlorothioxanthone, 2-methylthioxanthone, 2-isopropyl Examples include thioxanthone series such as thioxanthone.
- polymerization inhibitor examples include trihydrobenzene, toluhydroquinone, 14-naphthoquinone, parabenzoquinone, hydroquinone, benzoquinone, hydroquinone monomethyl ether, p-tert-butylcatechol, 2,6-di-tert-butyl-4-methyl. Phenol and the like can be mentioned. Preferably, 10 to 1000 ppm can be added to the resin composition.
- the fat composition of the present invention may contain petroleum wax, synthetic wax, that is, polyethylene wax, oxidized paraffin, alcohol type wax, and the like as components for assisting in drying the coating film.
- the resin composition of the present invention includes various additives such as pigments, heat shielding pigments, fillers, ultraviolet absorbers, thickeners, low shrinkage agents, anti-aging agents, plasticizers, and aggregates. Flame retardants, stabilizers, reinforcing materials and the like can be used.
- the pigment may be any pigment as long as it is used in preparing a colored paint, but typical examples include carbon black, phthalocyanine blue, phthalocyanine green, and quinacridone red.
- Organic pigments include carbon black, phthalocyanine blue, phthalocyanine green, and quinacridone red.
- Organic pigments such as titanium oxide, iron oxide, titanium yellow, and copper chrome black; and inorganic flake pigments such as aluminum flakes and pearl mica.
- the above-mentioned heat-shielding pigment has a solar reflectance of 15% or more in the wavelength range of 350 to 2100 nm as defined in JIS A 5759 for the purpose of heat-shielding properties, and L in the CIE 1976 L * a * b * color space.
- a pigment with a value of 30 or less, more preferably a L * value of 24 or less may be added.
- coloring pigments examples include yellow pigments such as monoazo yellow (trade name Hoster Palm Yellow H3G: manufactured by Hoechst), iron oxide (trade name Toda Color 120ED: manufactured by Toda Kogyo Co., Ltd.), Red pigments such as quinacridone red (trade name Hostaperm Red E2B70: manufactured by Hoechst), blue pigments such as phthalocyanine blue (trade name cyanine blue SPG-8: manufactured by DIC Corporation), phthalocyanine green (trade name cyanine) Green 5310: manufactured by Dainichi Seika Kogyo Co., Ltd.) and the like.
- the resin composition of the present invention can be preferably used as a thermal barrier coating by adding the thermal barrier pigment.
- Examples of the filler include hydraulic silicate material, calcium carbonate powder, clay, alumina powder, aragonite powder, talc, barium sulfate, silica powder, glass powder, glass beads, mica, aluminum hydroxide, cellulose-based, Examples include dredged sand, river sand, and cold water stones.
- the resin composition of the present invention is excellent in non-foaming property at room temperature and curing property at room temperature, it can be used in construction methods for outdoor structures, and any structure related to civil engineering and construction can be used.
- Examples of the civil engineering-related structure include a floor, a wall, a ceiling, a road, a sidewalk, a pool floor, and a rooftop floor. It can also be laminated on the FRP waterproof layer.
- the construction method of the present invention is performed by adding a radical curing agent to the resin composition of the present invention and performing coating operations such as spray coating, brush coating, and roll coating.
- Example 1 was 2 hours in Example 1, 3 hours in Example 2, 2 hours in Example 3, 2 hours in Example 4, and 3 hours in Example 5. Thereafter, it is cooled to 140 ° C., and then a predetermined amount of glycidyl methacrylate (GMA) is added and reacted for 3 hours. After cooling to 60 ° C., phenoxyethyl methacrylate (B) is used as the (meth) acrylic monomer (B). PhOEMA) was added to obtain a resin composition.
- the input amount of PhOEMA was 40% by mass in Example 1 and Example 4, 50% by mass in Example 2, Example 3 and Example 5, Example 1 was composition a1, and Example 4 was composition a5.
- Example 2 as composition a2, Example 3 as composition a3, and Example 5 as composition a6.
- Synthesis Example 1 (Preparation of epoxy methacrylate resin (composition b)) 1850 g of Epicron 850 (made by DIC Corporation) having an epoxy equivalent of 185 obtained by the reaction of bisphenol A and epichlorohydrin in a four-necked flask equipped with a thermometer, a stirrer, an inert gas inlet and a reflux condenser, methacrylic acid 860 g, 1.36 g of hydroquinone and 10.8 g of triethylamine were charged, the temperature was raised to 120 ° C., and the mixture was reacted at the same temperature for 10 hours to obtain an epoxy acrylate having an acid value of 3.5. Phenoxyethyl methacrylate (PhOEMA) was added thereto to obtain an epoxy methacrylate resin. The input amount of PhOEMA was 60% by mass, and this resin was used as composition b.
- Comparative Example 1 (Preparation of resin composition a0) Without using maleic anhydride, phthalic anhydride (PA), diethylene glycol (DEG) and cis-3-methyl-4-cyclohexene-cis-1,2-dicarboxylic anhydride (PMAA) were thermometer, stirrer, Into a four-necked flask equipped with an inert gas inlet and a reflux condenser, 0.05% by mass of dibutyltin oxide was added as an esterification catalyst and reacted at 205 ° C. The reaction time at 205 ° C.
- PA phthalic anhydride
- DEG diethylene glycol
- PMAA cis-3-methyl-4-cyclohexene-cis-1,2-dicarboxylic anhydride
- PhOEMA acrylic Phenoxyethyl methacrylate
- PhOEMA glycidyl methacrylate
- Comparative Example 3 One part of monomethyl malate was added as an additive to the composition b, and the presence or absence of foam and curability were confirmed.
- Non-foaming How to check the presence or absence of bubbles during curing" 100 g of the resin composition was taken in a 200 cc polycup, the resin was cured under the following formulation and temperature conditions, and the presence or absence of bubbles until gelation was visually observed. When bubbles remained in the resin or the liquid surface in the polycup at the time when the resin was gelled, “bubbles” was indicated, and when bubbles did not remain, “no bubbles” was indicated.
- the gel time measurement method is based on JIS K6901 5.10.1 [normal temperature gelation time (Method A)]. The gel time was measured in the same manner as in the above composition and temperature conditions. After blending the curing accelerator and auxiliary accelerator, the gel time measurement result on the day and the gel time measurement result one month later were confirmed, and the degree of deactivation of the accelerator cobalt I saw.
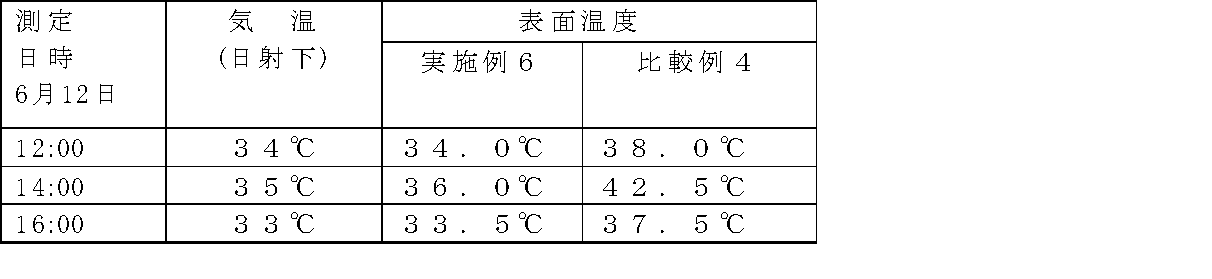
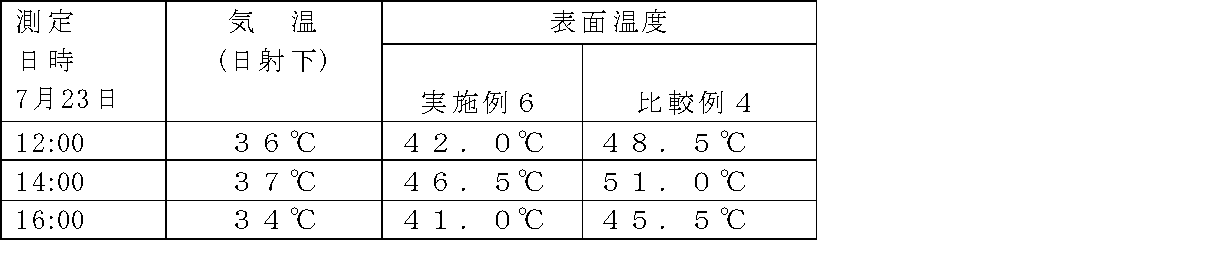
- Example 6 After applying 0.2 kg / m 2 of urethane primer (Priadec T-150-35DNX) manufactured by DIC Corporation on the ground concrete and drying, it is applied to methacrylic resin (Diobar NS-313TM) manufactured by DIC Corporation and 55% methyl ethyl ketone par. After blending 1.0% of oxide and applying with a brush, the glass mat for waterproofing # 450 was impregnated and defoamed to form an FRP waterproof layer. After the impregnation and defoaming, the resin was cured and dried within about 60 minutes. On top of that, the gray topcoat composition of Example 6 in Table 3 was applied with a brush at a thickness of 0.3 kg / m 2 with the following composition. An outdoor exposure experiment was conducted to measure the surface temperature. The results are shown in Tables 4 and 5.
- Comparative Example 4 In the same manner as in Example 6, after applying a urethane primer and a methacrylic resin on the foundation concrete, the gray top coat composition of Comparative Example 4 in Table 3 was added on the concrete with 0.3 kg / m 2 in the following composition. The thickness was applied with a brush. An outdoor exposure experiment was conducted to measure the surface temperature. The results are shown in Tables 4 and 5.
- Example 6 It can be seen from Example 6 and Comparative Example 4 that a surface temperature difference of about 4 to 7 ° C. is caused by the application of the thermal barrier coating.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Macromonomer-Based Addition Polymer (AREA)
- Polymerization Catalysts (AREA)
- Paints Or Removers (AREA)
Abstract
Description
不飽和二塩基酸の不飽和基とグリシジルメタクリレートのメタアクリロイル基とを比較すると、アクリル系単量体との共重合性が異なり、グリシジルメタクリレートとアクリル系単量体との共重合性が、不飽和二塩基酸とアクリル系単量体との共重合性よりも優れる。このため不飽和二塩基酸の使用量が10モル%を超える場合は、硬化物の引張強度物性が低下するので好ましくない。
また、ジビニルベンゼン、ジアリルフタレート、ジアリルイソフタレート、ジアリルテトラブロムフタレート、トリアリルフタレート等も、発明の効果を損なわない範囲で使用することができる。
硬化促進剤(C)の添加量は、前記(A)と前記(B)からなる樹脂組成物中に、好ましくは0.01~5質量部である。この範囲を外れると長期保存安定性が得られないものとなる。この硬化促進剤は、予め樹脂組成物に添加しておくものである。
表1の配合量で無水フタル酸(PA)、ジエチレングリコール(DEG)、無水マレイン酸(MA)およびシス-3-メチル-4-シクロヘキセン-シス-1,2-ジカルボキシリックアンハイドライド(PMAA)を温度計、攪拌機、不活性ガス導入口および還流冷却器を備えた四口フラスコに仕込み、エステル化触媒としてジブチルチンオキサイドを0.05質量%添加し、205℃で反応させた。 205℃での反応時間は、実施例1で2時間、実施例2で3時間、実施例3で2時間、実施例4で2時間、実施例5で3時間行った。その後、140℃まで冷却し、次いでグリシジルメタクリレート(GMA)を所定量投入し、3時間反応させた後、60℃まで冷却してから(メタ)アクリル系単量体(B)としてフェノキシエチルメタクリレート(PhOEMA)を投入し、樹脂組成物を得た。PhOEMAの投入量は、実施例1及び実施例4で40質量%、実施例2、実施例3及び実施例5で50質量%とし、実施例1を組成物a1、実施例4を組成物a5、及び実施例2を組成物a2、実施例3を組成物a3、実施例5を組成物a6とした。
温度計、攪拌機、不活性ガス導入口および還流冷却器を備えた四口フラスコにビスフェノールAとエピクロルヒドリンとの反応により得られたエポキシ当量が185なるエピクロン850(DIC(株)製)1850g、メタクリル酸860g、ハイドロキノン1.36gおよびトリエチルアミン10.8gを仕込み、120℃まで昇温させ、同温度で10時間反応させ、酸価3.5のエポキシアクリレートとした。これにフェノキシエチルメタクリレート(PhOEMA)を投入し、エポキシメタクリレート樹脂を得た。PhOEMAの投入量は60質量%とし、この樹脂を組成物bとした。
無水マレイン酸を使用しないで、無水フタル酸(PA)、ジエチレングリコール(DEG)およびシス-3-メチル-4-シクロヘキセン-シス-1,2-ジカルボキシリックアンハイドライド(PMAA)を温度計、攪拌機、不活性ガス導入口および還流冷却器を備えた四口フラスコに仕込み、エステル化触媒としてジブチルチンオキサイドを0.05質量%添加し、205℃で反応させた。205℃での反応時間は2時間とし、その後、140℃まで冷却し、次いでグリシジルメタクリレート(GMA)を所定量投入し、3時間反応させた後、60℃まで冷却してから(メタ)アクリル系単量体(B)としてフェノキシエチルメタクリレート(PhOEMA)を投入し、樹脂組成物a0を得た。PhOEMAの投入量は40質量%とした。
表1の配合量で無水マレイン酸(MA)、無水フタル酸(PA)、ジエチレングリコール(DEG)およびシス-3-メチル-4-シクロヘキセン-シス-1,2-ジカルボキシリックアンハイドライド(PMAA)を温度計、攪拌機、不活性ガス導入口および還流冷却器を備えた四口フラスコに仕込み、エステル化触媒としてジブチルチンオキサイドを0.05質量%添加し、205℃で反応させた。205℃での反応時間は2時間とし、その後、140℃まで冷却し、次いでグリシジルメタクリレート(GMA)を所定量投入し、3時間反応させた後、60℃まで冷却してから反応性モノマーとしてフェノキシエチルメタクリレート(PhOEMA)を投入し、樹脂組成物a4を得た。PhOEMAの投入量は40質量%とした。
上記の組成物bに添加剤としてモノメチルマレート1部を添加し、泡の有無、硬化性を確認した。
200ccのポリカップに樹脂組成物100gを採り、以下の配合および温度条件で樹脂を硬化させ、ゲル化に至るまでの間の泡の有無を目視観察した。
樹脂がゲル化した時点でポリカップ中の樹脂中あるいは液面に泡が残存する場合は、「泡有」とし、泡が残存していない場合は、「泡無」とした。
樹脂組成物:各サンプル 100g
硬化促進剤:8%ナフテン酸コバルト 1g
助促進剤 :アセチルブチロラクトン 0.05g
硬化剤 :55%メチルエチルケトンパーオキサイド 1g
ゲルタイム測定方法は、JIS K6901 5.10.1[常温ゲル化時間(A法)]による。
上記配合、温度条件と同様にしてゲルタイムを測定、硬化促進剤、助促進剤を配合後、当日ゲルタイム測定した結果と、一ヵ月後にゲルタイム測定した結果について確認し、促進剤コバルトの失活の程度をみた。
厚み3mmの樹脂注型板を作成し、常温養生1日後から80℃×6時間、後硬化した。
(配 合)
樹 脂 :各サンプル 100g
硬化促進剤:8%ナフテン酸コバルト 1g
助促進剤 :アセチルブチロラクトン 0.05g
硬化剤 :55%メチルエチルケトンパーオキサイド 1g
引張物性の測定は、JISK7113法、1号ダンベルで実施した。
DEG:ジエチレングリコール
PA:無水フタル酸
MA:無水マレイン酸
PMAA:シス-3-メチル-4-シクロヘキセン-シス-1,2-ジカルボキシリックアンハイドライド(メチルテトラヒドロ無水フタル酸)
GMA:グリシジルメタクリレート
PhOEMA:フェノキシエチルメタクリレート
比較例3のエポキシアクリレート樹脂では、保存後の常温硬化性が悪いものとなる。
下地コンクリート上に、DIC株式会社製ウレタンプライマー(プライアデックT-150-35DNX)を0.2kg/m2塗布乾燥後、DIC株式会社製メタクリル樹脂(ディオバー NS-313TM)に硬化剤55%メチルエチルケトンパーオキサイド1.0%を配合し、刷毛にて塗布後、防水用ガラスマット#450に含浸、脱泡させ、FRP防水層とした。含浸、脱泡終了後、約60分以内に硬化、乾燥した。その上に、表3の実施例6のグレー色のトップコート組成物を以下の配合で、0.3kg/m2の厚みで刷毛により塗布した。屋外での暴露実験を実施し、表面温度を測定した。結果を表4,5に示した。
トップコート組成物:実施例3 100g
硬化促進剤:8%ナフテン酸コバルト 1g
助促進剤 :アセチルブチロラクトン 0.05g
硬化剤 :55%メチルエチルケトンパーオキサイド 1g
実施例6と同様に、下地コンクリート上にウレタンプライマー、メタクリル樹脂を塗布した後、その上に表3の比較例4のグレー色のトップコート組成物を以下の配合で、0.3kg/m2の厚みで刷毛により塗布した。屋外での暴露実験を実施し、表面温度を測定した。結果を表4,5に示した。
トップコート組成物:比較例4 100g
硬化促進剤:8%ナフテン酸コバルト 1g
助促進剤 :アセチルブチロラクトン 0.05g
硬化剤 :55%メチルエチルケトンパーオキサイド 1g
測定方法:非接触型赤外線式表面温度計(Raytek社製)
場 所:DIC株式会社堺工場屋外暴露場
日 時:2009年6月12日測定、2009年7月23日測定
Claims (7)
- 環状脂肪族不飽和二塩基酸由来の構造単位及びエーテル結合含有グリコール由来の構造単位を有するポリエステル(メタ)アクリレート樹脂(A)と(メタ)アクリル系単量体(B)、コバルト系硬化促進剤(C)とを含む空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物であって、
前記ポリエステル(メタ)アクリレート樹脂(A)が、全二塩基酸中に環状脂肪族不飽和二塩基酸(A1)を5モル%~50モル%未満使用するものであり、かつ全二塩基酸中にマレイン酸、無水マレイン酸のうちの少なくとも1種の不飽和二塩基酸(A2)を1モル%~10モル%使用していることを特徴とする空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物。 - 前記ポリエステル(メタ)アクリレート樹脂(A)が、環状脂肪族不飽和二塩基酸(A1)10~45モル%、不飽和二塩基酸(A2)1~10モル%及び飽和二塩基酸(A3)45~89モル%からなる二塩基酸を使用するものである請求項1記載の空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物。
- さらに、硬化剤(D)として、ケトンパーオキサイド系硬化剤を使用するものである請求項1記載の空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物。
- 前記(メタ)アクリル系単量体(B)が、フェノキシメチルメタクリレートである請求項1記載の空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物。
- 前記ポリエステル(メタ)アクリレート樹脂(A)が60~30質量%、前記(メタ)アクリル系単量体(B)が40~70質量%含まれるものである請求項1記載の空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物。
- 請求項1~3いずれか1項に記載の空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物を施工したことを特徴とする構造体。
- 請求項1~3いずれか1項に記載の空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物に硬化剤を添加して、土木建築構造体に施工することを特徴とする施工方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/259,508 US20120108700A1 (en) | 2009-06-18 | 2010-04-01 | Air-drying polyester (meth)acrylate resin composition, structure, and method for applying the resin composition |
| CN201080024154.4A CN102449008B (zh) | 2009-06-18 | 2010-04-01 | 空气干燥性聚酯(甲基)丙烯酸酯树脂组合物、结构体及其施工方法 |
| JP2010538668A JP4716150B2 (ja) | 2009-06-18 | 2010-04-01 | 空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物、構造体及びその施工方法 |
| EP10789289.5A EP2395033A4 (en) | 2009-06-18 | 2010-04-01 | AIR-DRYING POLYESTER (METH) ACRYLATE RESIN COMPOSITION AND STRUCTURE AND METHOD FOR THE PRODUCTION THEREOF |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009-145316 | 2009-06-18 | ||
| JP2009145316 | 2009-06-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010146912A1 true WO2010146912A1 (ja) | 2010-12-23 |
Family
ID=43356241
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2010/056010 Ceased WO2010146912A1 (ja) | 2009-06-18 | 2010-04-01 | 空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物、構造体及びその施工方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20120108700A1 (ja) |
| EP (1) | EP2395033A4 (ja) |
| JP (1) | JP4716150B2 (ja) |
| KR (1) | KR20120037360A (ja) |
| CN (1) | CN102449008B (ja) |
| WO (1) | WO2010146912A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012180444A (ja) * | 2011-03-01 | 2012-09-20 | Dic Corp | ラジカル硬化性樹脂組成物及びその硬化方法 |
| WO2016171034A1 (ja) * | 2015-04-22 | 2016-10-27 | Dic株式会社 | ラジカル重合性樹脂組成物及び土木建築用プライマー |
| WO2024090516A1 (ja) * | 2022-10-28 | 2024-05-02 | 株式会社レゾナック | ラジカル重合性樹脂用硬化剤組成物、及びラジカル重合性樹脂組成物 |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6281217B2 (ja) * | 2013-09-09 | 2018-02-21 | Dic株式会社 | ラジカル重合性樹脂組成物及び土木建築材料 |
| US12187900B2 (en) | 2016-06-27 | 2025-01-07 | Viavi Solutions Inc. | High chromaticity pigment flakes and foils |
| US10928579B2 (en) | 2016-06-27 | 2021-02-23 | Viavi Solutions Inc. | Optical devices |
| KR102052720B1 (ko) | 2016-06-27 | 2019-12-05 | 비아비 솔루션즈 아이엔씨. | 고채도 플레이크 |
| US12552939B2 (en) | 2018-06-29 | 2026-02-17 | Viavi Solutions Inc. | Optical devices with functional molecules |
| CA3236836A1 (en) * | 2021-11-24 | 2023-06-01 | Kareem Kaleem | (co)polymer-acrylic block copolymers and coating compositions containing the same |
| JP2024005580A (ja) * | 2022-06-30 | 2024-01-17 | Dic株式会社 | ラジカル重合性組成物 |
| CN116854866B (zh) * | 2023-06-05 | 2024-02-06 | 浙江创赢新材料有限公司 | 一种道路涂料用mma双组分不饱和聚酯树脂及其制备方法 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5441987A (en) | 1977-09-09 | 1979-04-03 | Nippon Oil & Fats Co Ltd | Curing of vinyl ester resin |
| JPS6094415A (ja) | 1983-10-28 | 1985-05-27 | Dainippon Ink & Chem Inc | 貯蔵安定性の改良された不飽和エポキシ樹脂組成物 |
| JP2004099770A (ja) * | 2002-09-10 | 2004-04-02 | Japan Composite Co Ltd | エポキシ(メタ)アクリレートおよび/またはウレタン(メタ)アクリレート樹脂組成物、およびその硬化方法 |
| JP2005248131A (ja) * | 2004-03-08 | 2005-09-15 | Japan Composite Co Ltd | ラジカル硬化性樹脂組成物 |
| JP2007326934A (ja) * | 2006-06-07 | 2007-12-20 | Dainippon Ink & Chem Inc | 硬化性樹脂組成物及び防水材組成物 |
| JP2008106169A (ja) * | 2006-10-26 | 2008-05-08 | Dainippon Ink & Chem Inc | 硬化性樹脂組成物 |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3776980A (en) * | 1970-04-30 | 1973-12-04 | Velsicol Chemical Corp | Polyesters of methylcyclohexene-dicarboxylic anhydride |
| US3663658A (en) * | 1970-04-30 | 1972-05-16 | Velsicol Chemical Corp | Polyester compositions and air-drying compositions thereof |
| US3674727A (en) * | 1970-10-20 | 1972-07-04 | Frank Fekete | Unsaturated polyesters |
| JPS5645951B2 (ja) * | 1973-05-21 | 1981-10-29 | ||
| NL7309060A (ja) * | 1973-06-29 | 1974-12-31 | ||
| US3989767A (en) * | 1974-03-05 | 1976-11-02 | Dainippon Ink & Chemicals, Incorporated | Powder coating resin compositions of glycidyl copolymers having an internal flowing agent |
| DE2644691B2 (de) * | 1976-10-02 | 1981-02-12 | Hoechst Ag, 6000 Frankfurt | Copolymerisatlösung aus Acrylharz, Verfahren zu ihrer Herstellung und Verwendung derselben in Reaktionslacken |
| US4130520A (en) * | 1977-03-25 | 1978-12-19 | Westinghouse Electric Corp. | Aqueous polyester coating composition, method of making, and coated conductor |
| US4348506A (en) * | 1979-08-06 | 1982-09-07 | The Dow Chemical Company | Process for curing vinyl ester resins and composition useful therein |
| NL7909040A (nl) * | 1979-12-14 | 1981-07-16 | Unilever Nv | Polyesters. |
| US4391947A (en) * | 1980-11-06 | 1983-07-05 | Westinghouse Electric Corp. | Low viscosity polyester coating compositions |
| US5747597A (en) * | 1995-09-25 | 1998-05-05 | Dainippon Ink And Chemicals, Inc. | Curable resin composition and coating material composition |
| EP1149874A1 (en) * | 2000-04-17 | 2001-10-31 | Dainippon Ink And Chemicals, Inc. | Polymerizable unsaturated polyester resin composition |
| WO2003057792A2 (en) * | 2001-12-29 | 2003-07-17 | 3M Innovative Properties Company | Composition containing a polymerizable reducing agent, kit, and method |
| WO2008003493A1 (en) * | 2006-07-06 | 2008-01-10 | Dsm Ip Assets B.V. | Unsaturated polyester resin or vinyl ester resin compositions |
| JPWO2009011211A1 (ja) * | 2007-07-13 | 2010-09-16 | 昭和電工株式会社 | 硬化性組成物およびその硬化物 |
-
2010
- 2010-04-01 EP EP10789289.5A patent/EP2395033A4/en not_active Withdrawn
- 2010-04-01 WO PCT/JP2010/056010 patent/WO2010146912A1/ja not_active Ceased
- 2010-04-01 JP JP2010538668A patent/JP4716150B2/ja active Active
- 2010-04-01 CN CN201080024154.4A patent/CN102449008B/zh active Active
- 2010-04-01 KR KR1020117010751A patent/KR20120037360A/ko not_active Ceased
- 2010-04-01 US US13/259,508 patent/US20120108700A1/en not_active Abandoned
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5441987A (en) | 1977-09-09 | 1979-04-03 | Nippon Oil & Fats Co Ltd | Curing of vinyl ester resin |
| JPS6094415A (ja) | 1983-10-28 | 1985-05-27 | Dainippon Ink & Chem Inc | 貯蔵安定性の改良された不飽和エポキシ樹脂組成物 |
| JP2004099770A (ja) * | 2002-09-10 | 2004-04-02 | Japan Composite Co Ltd | エポキシ(メタ)アクリレートおよび/またはウレタン(メタ)アクリレート樹脂組成物、およびその硬化方法 |
| JP2005248131A (ja) * | 2004-03-08 | 2005-09-15 | Japan Composite Co Ltd | ラジカル硬化性樹脂組成物 |
| JP2007326934A (ja) * | 2006-06-07 | 2007-12-20 | Dainippon Ink & Chem Inc | 硬化性樹脂組成物及び防水材組成物 |
| JP2008106169A (ja) * | 2006-10-26 | 2008-05-08 | Dainippon Ink & Chem Inc | 硬化性樹脂組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2395033A4 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012180444A (ja) * | 2011-03-01 | 2012-09-20 | Dic Corp | ラジカル硬化性樹脂組成物及びその硬化方法 |
| WO2016171034A1 (ja) * | 2015-04-22 | 2016-10-27 | Dic株式会社 | ラジカル重合性樹脂組成物及び土木建築用プライマー |
| JP6066027B1 (ja) * | 2015-04-22 | 2017-01-25 | Dic株式会社 | ラジカル重合性樹脂組成物及び土木建築用プライマー |
| WO2024090516A1 (ja) * | 2022-10-28 | 2024-05-02 | 株式会社レゾナック | ラジカル重合性樹脂用硬化剤組成物、及びラジカル重合性樹脂組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20120037360A (ko) | 2012-04-19 |
| JP4716150B2 (ja) | 2011-07-06 |
| EP2395033A1 (en) | 2011-12-14 |
| CN102449008A (zh) | 2012-05-09 |
| EP2395033A4 (en) | 2013-12-04 |
| US20120108700A1 (en) | 2012-05-03 |
| JPWO2010146912A1 (ja) | 2012-12-06 |
| CN102449008B (zh) | 2015-02-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4716150B2 (ja) | 空気乾燥性ポリエステル(メタ)アクリレート樹脂組成物、構造体及びその施工方法 | |
| JP5346522B2 (ja) | 防食塗料組成物およびそれを用いた防食塗膜構造体 | |
| JP3994297B2 (ja) | 樹脂組成物及び土木建築材料 | |
| JP2003503515A (ja) | 構造適用のための、不飽和ポリエステル樹脂、ビニルエステル及びビニルエステルウレタン中のビニルエーテル | |
| JP5527036B2 (ja) | モルタル組成物、コンクリート構造体及び補修方法 | |
| JP4623242B1 (ja) | ポリエステル(メタ)アクリレート樹脂組成物、塗布構造体及びその施工方法 | |
| JP2009263445A (ja) | 空気乾燥性不飽和樹脂組成物の保存方法、それを用いた土木建築構造体、及び土木建築物の施工方法 | |
| TW570935B (en) | Curable unsaturated resin composition | |
| JP4973914B2 (ja) | 硬化性樹脂組成物及び防水材組成物 | |
| JP2006206863A (ja) | 不飽和ポリエステル樹脂組成物及びこれを用いた塗装方法 | |
| JP4151310B2 (ja) | 硬化性不飽和樹脂組成物 | |
| JP4441868B2 (ja) | 土木建築材用硬化性樹脂組成物、塗布用土木建築材および土木建築物 | |
| JP2008106169A (ja) | 硬化性樹脂組成物 | |
| JP4102379B2 (ja) | 不飽和ポリエステル樹脂組成物およびその硬化体 | |
| JP2003268054A (ja) | ラジカル硬化性樹脂組成物及びそれを用いた土木建築材料 | |
| JP2006096911A (ja) | 靭性を有する不飽和ポリエステル樹脂、その製造方法及びその利用 | |
| JP4911485B2 (ja) | 不飽和ポリエステル樹脂組成物 | |
| JP2006282767A (ja) | 不飽和ポリエステル樹脂組成物およびその硬化体 | |
| JP5288180B2 (ja) | ポリエステル(メタ)アクリレート樹脂組成物、塗布構造体及びその施工方法 | |
| JP4272500B2 (ja) | 樹脂組成物 | |
| JP5166967B2 (ja) | 耐アルコール性能が求められる用途のための不飽和ポリエステル樹脂組成物、それを用いた耐アルコール性能が求められる用途のための樹脂複合組成物及びアルコール系燃料用frp二重殻タンク | |
| JP4710365B2 (ja) | 硬化性樹脂組成物、土木建築材及び土木建築物 | |
| JP2003082038A (ja) | 不飽和ポリエステル樹脂組成物 | |
| JP2001151832A (ja) | 硬化性樹脂組成物およびその製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080024154.4 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 2010538668 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10789289 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20117010751 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010789289 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13259508 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |