RU2406722C2 - Соли бис-(тиогидразидамида) для лечения рака - Google Patents
Соли бис-(тиогидразидамида) для лечения рака Download PDFInfo
- Publication number
- RU2406722C2 RU2406722C2 RU2007101898/04A RU2007101898A RU2406722C2 RU 2406722 C2 RU2406722 C2 RU 2406722C2 RU 2007101898/04 A RU2007101898/04 A RU 2007101898/04A RU 2007101898 A RU2007101898 A RU 2007101898A RU 2406722 C2 RU2406722 C2 RU 2406722C2
- Authority
- RU
- Russia
- Prior art keywords
- thiohydrazidamide
- bis
- organic solvent
- base
- solvent
- Prior art date
Links
- 206010028980 Neoplasm Diseases 0.000 title claims abstract 5
- 201000011510 cancer Diseases 0.000 title claims abstract 5
- 150000001408 amides Chemical class 0.000 title abstract 2
- 150000003839 salts Chemical class 0.000 title abstract 2
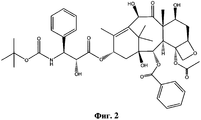
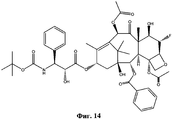
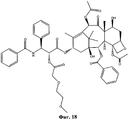
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 claims abstract description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims abstract 11
- 150000001875 compounds Chemical class 0.000 claims abstract 11
- 229930012538 Paclitaxel Natural products 0.000 claims abstract 4
- 229960001592 paclitaxel Drugs 0.000 claims abstract 4
- 230000001093 anti-cancer Effects 0.000 claims abstract 3
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract 3
- 230000002708 enhancing effect Effects 0.000 claims abstract 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims 14
- 239000003960 organic solvent Substances 0.000 claims 11
- 239000002585 base Substances 0.000 claims 10
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical group [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims 9
- 239000000243 solution Substances 0.000 claims 8
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 claims 7
- 230000007935 neutral effect Effects 0.000 claims 7
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical group OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims 6
- 239000012296 anti-solvent Substances 0.000 claims 6
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical group CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 claims 4
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 claims 4
- 229910052783 alkali metal Inorganic materials 0.000 claims 4
- 239000000203 mixture Substances 0.000 claims 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 claims 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 claims 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 claims 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims 3
- -1 alkyl glycol ether Chemical compound 0.000 claims 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 claims 3
- 239000011734 sodium Substances 0.000 claims 3
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 claims 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 claims 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims 2
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 claims 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 claims 2
- 150000001335 aliphatic alkanes Chemical class 0.000 claims 2
- 150000001924 cycloalkanes Chemical class 0.000 claims 2
- 201000001441 melanoma Diseases 0.000 claims 2
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 claims 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 claims 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 claims 1
- 206010006187 Breast cancer Diseases 0.000 claims 1
- 208000026310 Breast neoplasm Diseases 0.000 claims 1
- KCMZYCFSSYXEQR-UHFFFAOYSA-N CCCC[K] Chemical compound CCCC[K] KCMZYCFSSYXEQR-UHFFFAOYSA-N 0.000 claims 1
- IRDQNLLVRXMERV-UHFFFAOYSA-N CCCC[Na] Chemical compound CCCC[Na] IRDQNLLVRXMERV-UHFFFAOYSA-N 0.000 claims 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 claims 1
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 claims 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 claims 1
- PLOSSHSFATUNTF-UHFFFAOYSA-N [K]C1=CC=CC=C1 Chemical compound [K]C1=CC=CC=C1 PLOSSHSFATUNTF-UHFFFAOYSA-N 0.000 claims 1
- 125000001931 aliphatic group Chemical group 0.000 claims 1
- 229910000102 alkali metal hydride Inorganic materials 0.000 claims 1
- 150000008046 alkali metal hydrides Chemical class 0.000 claims 1
- 150000008044 alkali metal hydroxides Chemical class 0.000 claims 1
- 150000001412 amines Chemical class 0.000 claims 1
- 239000000908 ammonium hydroxide Substances 0.000 claims 1
- 239000007864 aqueous solution Substances 0.000 claims 1
- 150000001768 cations Chemical class 0.000 claims 1
- RXKJFZQQPQGTFL-UHFFFAOYSA-N dihydroxyacetone Chemical compound OCC(=O)CO RXKJFZQQPQGTFL-UHFFFAOYSA-N 0.000 claims 1
- 239000003085 diluting agent Substances 0.000 claims 1
- 239000003937 drug carrier Substances 0.000 claims 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims 1
- 208000032839 leukemia Diseases 0.000 claims 1
- 229910000103 lithium hydride Inorganic materials 0.000 claims 1
- SIAPCJWMELPYOE-UHFFFAOYSA-N lithium hydride Chemical group [LiH] SIAPCJWMELPYOE-UHFFFAOYSA-N 0.000 claims 1
- 208000025113 myeloid leukemia Diseases 0.000 claims 1
- 239000003208 petroleum Substances 0.000 claims 1
- NHKJPPKXDNZFBJ-UHFFFAOYSA-N phenyllithium Chemical compound [Li]C1=CC=CC=C1 NHKJPPKXDNZFBJ-UHFFFAOYSA-N 0.000 claims 1
- RPDAUEIUDPHABB-UHFFFAOYSA-N potassium ethoxide Chemical compound [K+].CC[O-] RPDAUEIUDPHABB-UHFFFAOYSA-N 0.000 claims 1
- 229910000105 potassium hydride Inorganic materials 0.000 claims 1
- NTTOTNSKUYCDAV-UHFFFAOYSA-N potassium hydride Chemical compound [KH] NTTOTNSKUYCDAV-UHFFFAOYSA-N 0.000 claims 1
- BDAWXSQJJCIFIK-UHFFFAOYSA-N potassium methoxide Chemical compound [K+].[O-]C BDAWXSQJJCIFIK-UHFFFAOYSA-N 0.000 claims 1
- 208000016691 refractory malignant neoplasm Diseases 0.000 claims 1
- 229910000104 sodium hydride Inorganic materials 0.000 claims 1
- 239000012312 sodium hydride Substances 0.000 claims 1
- KSMWLICLECSXMI-UHFFFAOYSA-N sodium;benzene Chemical compound [Na+].C1=CC=[C-]C=C1 KSMWLICLECSXMI-UHFFFAOYSA-N 0.000 claims 1
- 239000002904 solvent Substances 0.000 claims 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims 1
- 239000008096 xylene Substances 0.000 claims 1
- 239000003814 drug Substances 0.000 abstract 1
- 230000000694 effects Effects 0.000 abstract 1
- 239000000126 substance Substances 0.000 abstract 1
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 abstract 1
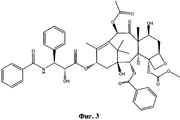
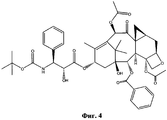
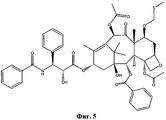
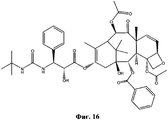
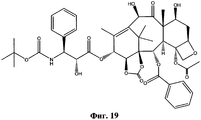
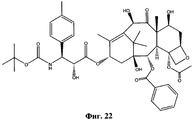
- NHITUGACHSROGE-MDMDSUOFSA-N CC(C)(C)OC(N[C@H]([C@H](C(O[C@@H](C1)C(C)=C([C@H](C([C@](C)([C@H](C[C@H]2OC3)O)[C@H]([C@@H]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)O)C(C)(C)[C@@]14O)=O)OC(COC(C[C@@H](C(N)=O)N)=O)=O)C1CC1)=O Chemical compound CC(C)(C)OC(N[C@H]([C@H](C(O[C@@H](C1)C(C)=C([C@H](C([C@](C)([C@H](C[C@H]2OC3)O)[C@H]([C@@H]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)O)C(C)(C)[C@@]14O)=O)OC(COC(C[C@@H](C(N)=O)N)=O)=O)C1CC1)=O NHITUGACHSROGE-MDMDSUOFSA-N 0.000 description 1
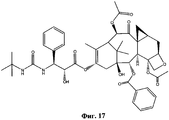
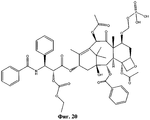
- UBCKDQWEECHQJC-MPSZKIJFSA-N CC(C)(C)OC(N[C@H]([C@H](C(O[C@@H](C1)C(C)=C([C@H](C([C@](C)([C@H]([C@@H]([C@H]2OC3)F)O)[C@H]([C@@H]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)OC(C)=O)C(C)(C)[C@@]14O)=O)O)c1ccccc1)=O Chemical compound CC(C)(C)OC(N[C@H]([C@H](C(O[C@@H](C1)C(C)=C([C@H](C([C@](C)([C@H]([C@@H]([C@H]2OC3)F)O)[C@H]([C@@H]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)OC(C)=O)C(C)(C)[C@@]14O)=O)O)c1ccccc1)=O UBCKDQWEECHQJC-MPSZKIJFSA-N 0.000 description 1
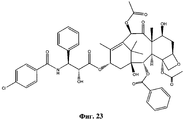
- 0 CC(C)(C)SC(N[C@]([C@](C(O[C@@](C1)C(C)=C([C@](C([C@](C)([C@](C[C@]2OC3)O)[C@]([C@@]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)OC(C)=O)C(C)(C)[C@@]14O)=O)O)*1ccc[o]1)=O Chemical compound CC(C)(C)SC(N[C@]([C@](C(O[C@@](C1)C(C)=C([C@](C([C@](C)([C@](C[C@]2OC3)O)[C@]([C@@]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)OC(C)=O)C(C)(C)[C@@]14O)=O)O)*1ccc[o]1)=O 0.000 description 1
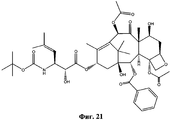
- UBJAHGAUPNGZFF-XOVTVWCYSA-N CC(C)([C@@](C1)([C@H]([C@H]([C@]2([C@@H](C[C@@H]3OCSC)OC2)OC(C)=O)[C@]3(C)C([C@@H]2OC(C)=O)=O)OC(c3ccccc3)=O)O)C2=C(C)[C@H]1OC([C@@H]([C@H](c1ccccc1)NC(c1ccccc1)=O)O)=O Chemical compound CC(C)([C@@](C1)([C@H]([C@H]([C@]2([C@@H](C[C@@H]3OCSC)OC2)OC(C)=O)[C@]3(C)C([C@@H]2OC(C)=O)=O)OC(c3ccccc3)=O)O)C2=C(C)[C@H]1OC([C@@H]([C@H](c1ccccc1)NC(c1ccccc1)=O)O)=O UBJAHGAUPNGZFF-XOVTVWCYSA-N 0.000 description 1
- RRZWOVDBXXQXBW-WCFPGISDSA-N CC(C)CSC(N[C@H]([C@H](C(O[C@@H](C1)C(C)=C([C@H](C([C@](C)([C@H](C[C@H]2OC3)O)[C@H]([C@@H]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)OC(C)=O)C(C)(C)[C@@]14O)=O)O)/C(/C)=C/C=C\O)=O Chemical compound CC(C)CSC(N[C@H]([C@H](C(O[C@@H](C1)C(C)=C([C@H](C([C@](C)([C@H](C[C@H]2OC3)O)[C@H]([C@@H]4OC(c5ccccc5)=O)[C@@]23OC(C)=O)=O)OC(C)=O)C(C)(C)[C@@]14O)=O)O)/C(/C)=C/C=C\O)=O RRZWOVDBXXQXBW-WCFPGISDSA-N 0.000 description 1
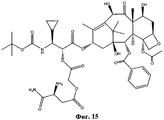
- MGJBERWKPZUCJM-UHFFFAOYSA-L CN(C(C1CC1)=S)/N=C(/C/C(/[O-])=N/N(C)C(C1CC1)=S)\[O-] Chemical compound CN(C(C1CC1)=S)/N=C(/C/C(/[O-])=N/N(C)C(C1CC1)=S)\[O-] MGJBERWKPZUCJM-UHFFFAOYSA-L 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D305/00—Heterocyclic compounds containing four-membered rings having one oxygen atom as the only ring hetero atoms
- C07D305/14—Heterocyclic compounds containing four-membered rings having one oxygen atom as the only ring hetero atoms condensed with carbocyclic rings or ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C327/00—Thiocarboxylic acids
- C07C327/38—Amides of thiocarboxylic acids
- C07C327/56—Amides of thiocarboxylic acids having nitrogen atoms of thiocarboxamide groups further bound to another hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/02—Systems containing only non-condensed rings with a three-membered ring
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pyridine Compounds (AREA)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US58259604P | 2004-06-23 | 2004-06-23 | |
| US60/582,596 | 2004-06-23 | ||
| US68136805P | 2005-05-16 | 2005-05-16 | |
| US60/681,368 | 2005-05-16 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2010145056/04A Division RU2010145056A (ru) | 2004-06-23 | 2010-11-08 | Соли бис (тиогидразидамида) для лечения рака |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2007101898A RU2007101898A (ru) | 2008-07-27 |
| RU2406722C2 true RU2406722C2 (ru) | 2010-12-20 |
Family
ID=34993190
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2007101898/04A RU2406722C2 (ru) | 2004-06-23 | 2005-06-20 | Соли бис-(тиогидразидамида) для лечения рака |
| RU2010145056/04A RU2010145056A (ru) | 2004-06-23 | 2010-11-08 | Соли бис (тиогидразидамида) для лечения рака |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2010145056/04A RU2010145056A (ru) | 2004-06-23 | 2010-11-08 | Соли бис (тиогидразидамида) для лечения рака |
Country Status (21)
| Country | Link |
|---|---|
| US (5) | US7385084B2 (enExample) |
| EP (2) | EP1781604B1 (enExample) |
| JP (1) | JP5362986B2 (enExample) |
| KR (1) | KR101313027B1 (enExample) |
| CN (1) | CN1993318B (enExample) |
| AU (1) | AU2005265202B2 (enExample) |
| BR (1) | BRPI0512526A (enExample) |
| CA (1) | CA2570698C (enExample) |
| DK (1) | DK1781604T3 (enExample) |
| ES (1) | ES2430373T3 (enExample) |
| HR (1) | HRP20131023T1 (enExample) |
| IL (1) | IL179632A (enExample) |
| ME (1) | ME01644B (enExample) |
| NO (1) | NO20070378L (enExample) |
| NZ (1) | NZ552159A (enExample) |
| PL (1) | PL1781604T3 (enExample) |
| PT (1) | PT1781604E (enExample) |
| RS (1) | RS52999B (enExample) |
| RU (2) | RU2406722C2 (enExample) |
| TW (1) | TWI389684B (enExample) |
| WO (1) | WO2006009940A1 (enExample) |
Families Citing this family (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI332943B (en) * | 2001-07-10 | 2010-11-11 | Synta Pharmaceuticals Corp | Taxol enhancer compounds |
| TWI252847B (en) | 2001-07-10 | 2006-04-11 | Synta Pharmaceuticals Corp | Synthesis of taxol enhancers |
| TWI297335B (en) * | 2001-07-10 | 2008-06-01 | Synta Pharmaceuticals Corp | Taxol enhancer compounds |
| TWI330079B (en) | 2003-01-15 | 2010-09-11 | Synta Pharmaceuticals Corp | Treatment for cancers |
| PL1781604T3 (pl) * | 2004-06-23 | 2014-01-31 | Synta Pharmaceuticals Corp | Sole bis(tiohydrazydoamidu) do leczenia nowotworów |
| AU2005306471B2 (en) | 2004-11-19 | 2009-12-17 | Synta Pharmaceuticals Corp. | Bis(thio-hydrazide amides) for increasing Hsp70 expression |
| US20090042991A1 (en) * | 2005-04-15 | 2009-02-12 | James Barsoum | Methods of increasing natural killer cell activity for therapy |
| MX2007012688A (es) | 2005-04-15 | 2008-03-14 | Synta Pharmaceuticals Corp | Terapia de combinacion para el cancer con compuestos de bis(tiohidrazida) amida. |
| US7709683B2 (en) | 2005-05-16 | 2010-05-04 | Synta Pharmaceuticals Corp. | Synthesis of bis(thio-hydrazide amide) salts |
| US7678832B2 (en) * | 2005-08-16 | 2010-03-16 | Synta Pharmaceuticals Corp. | Bis(thio-hydrazide amide) formulation |
| US20100310587A1 (en) * | 2005-08-29 | 2010-12-09 | Academia Sinica | Immunomodulatory compositions |
| GB0607950D0 (en) * | 2006-04-21 | 2006-05-31 | Novartis Ag | Organic compounds |
| US8785451B2 (en) | 2006-08-21 | 2014-07-22 | Synta Pharmaceuticals Corp. | Compounds for the treatment of proliferative disorders |
| AU2007288340A1 (en) * | 2006-08-21 | 2008-02-28 | Synta Pharmaceuticals Corp. | Bis (thiohydrazide amides) for treating melanoma |
| RU2475478C2 (ru) * | 2006-08-21 | 2013-02-20 | Синта Фармасьютиклз Корп. | Соединения для лечения пролиферативных расстройств |
| WO2008024299A2 (en) * | 2006-08-21 | 2008-02-28 | Synta Pharmaceuticals Corp. | Combination with bis(thiohydrazide amides) for treating cancer |
| EP2063878A2 (en) * | 2006-08-21 | 2009-06-03 | Synta Pharmaceuticals Corporation | Bis(thiohydrazide amides) for use in preventing or delaying the recurrence of melanoma |
| JP2010502616A (ja) | 2006-08-31 | 2010-01-28 | シンタ ファーマシューティカルズ コーポレーション | 癌を治療するためのビス(チオヒドラジドアミド)の組み合わせ |
| US9498528B2 (en) * | 2006-09-13 | 2016-11-22 | Genzyme Corporation | Treatment of multiple sclerosis (MS) |
| WO2008033494A2 (en) | 2006-09-15 | 2008-03-20 | Synta Pharmaceuticals Corp. | Purification of bis(thiohydrazide amides) |
| WO2009064374A2 (en) * | 2007-11-09 | 2009-05-22 | Synta Pharmaceuticals Corp. | Oral formulations of bis(thiohydrazide amides) |
| US8581004B2 (en) | 2008-02-21 | 2013-11-12 | Synta Pharmaceuticals Corp. | Compounds for treating proliferative disorders |
| US8633323B2 (en) * | 2008-03-31 | 2014-01-21 | Synta Pharmaceuticals Corp. | Process for preparing bis(thiohydrazide amides) |
| NZ592400A (en) * | 2008-10-22 | 2013-09-27 | Synta Pharmaceuticals Corp | Transition metal complexes of bis[thiohydrazide amide] compounds |
| MX2011004114A (es) | 2008-10-22 | 2011-08-15 | Synta Pharmaceuticals Corp | Complejos de metales de transicion de un compuesto bis (tiohidrazida amida). |
| US8525776B2 (en) * | 2008-10-27 | 2013-09-03 | Lenovo (Singapore) Pte. Ltd | Techniques for controlling operation of a device with a virtual touchscreen |
| EP2370402A2 (en) | 2008-12-01 | 2011-10-05 | Synta Pharmaceuticals Corp. | Compounds for treating proliferative disorders |
| RU2400471C1 (ru) * | 2009-04-30 | 2010-09-27 | Учреждение Российской академии наук Институт органической химии им. Н.Д. Зелинского РАН (ИОХ РАН) | N-замещенные производные тиогидразидов оксаминовых кислот, способ их получения и их использование |
| WO2011069159A2 (en) * | 2009-12-04 | 2011-06-09 | Synta Pharmaceuticals Corp. | Bis[thiohydrazide amide] compounds for treating leukemia |
| US8815945B2 (en) | 2010-04-20 | 2014-08-26 | Masazumi Nagai | Use of bis [thiohydrazide amide] compounds such as elesclomol for treating cancers |
| JP2013545759A (ja) | 2010-11-18 | 2013-12-26 | シンタ ファーマスーティカルズ コーポレーション | 低酸素状態に基づくエレスクロモールによる治療に適した被験体の事前選択 |
| JP2014534238A (ja) * | 2011-11-10 | 2014-12-18 | シンタ ファーマシューティカルズ コーポレーション | 癌を治療するためのビス(チオヒドラジドアミド)化合物の投与 |
| US20130149392A1 (en) | 2011-12-12 | 2013-06-13 | Synta Pharmaceuticals Corp. | Method of treating non-small cell lung cancer with bis-(thiohydrazide)amide compounds |
| WO2013103795A1 (en) | 2012-01-05 | 2013-07-11 | The Board Of Trustees Of The Leland Stanford Junior University | Bis (thiohydrazide amide) compounds for treating cancers |
| US12610818B2 (en) | 2023-02-07 | 2026-04-21 | Semiconductor Components Industries, Llc | Single switch direct cooling assemblies and related methods |
Family Cites Families (78)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2097737A5 (en) | 1970-07-14 | 1972-03-03 | Berlin Chemie Veb | Virustatic 4-substd 1-acylthiosemicarbazides -from carboxylic acid - hydrazide and isothiocyanates or from carboxylic acid chloride and 4- |
| DE2037257A1 (en) | 1970-07-28 | 1972-02-03 | Farbwerke Hoechst AG, vorm. Meister Lucius & Brüning, 6000 Frankfurt | Poly-(5-amino-1,3,4-thiadiazol-2-yl) derivs prepn - intermediates for drug and polymer prodn |
| GB1272920A (en) | 1971-03-15 | 1972-05-03 | Berlin Chemie Veb | New thiosemicarbazides |
| US4012360A (en) * | 1973-12-03 | 1977-03-15 | Ciba-Geigy Corporation | Bis-salicyloyl-dicarboxylic acid dihydrazides as stabilizers for polyolefines |
| JPS5091056A (enExample) | 1973-12-17 | 1975-07-21 | ||
| US4822777A (en) | 1987-02-27 | 1989-04-18 | Liposome Technology, Inc. | Amphotericin B/cholesterol sulfate composition |
| JP2767241B2 (ja) | 1987-04-15 | 1998-06-18 | ロ−ム アンド ハ−ス コンパニ− | 殺虫性のn−(場合により置換された)−n′−置換−n,n′−ジ置換ヒドラジン |
| US6013836A (en) * | 1992-02-28 | 2000-01-11 | Rohm And Haas Company | Insecticidal N'-substituted-N,N'-disubstitutedhydrazines |
| FR2697752B1 (fr) | 1992-11-10 | 1995-04-14 | Rhone Poulenc Rorer Sa | Compositions antitumorales contenant des dérivés du taxane. |
| CH683965A5 (it) * | 1993-02-19 | 1994-06-30 | Limad Marketing Exp & Imp | Composti della classe dei ftalidrazidici come sostanza attiva in agenti antinfiammatori ed antitossici. |
| US6749868B1 (en) | 1993-02-22 | 2004-06-15 | American Bioscience, Inc. | Protein stabilized pharmacologically active agents, methods for the preparation thereof and methods for the use thereof |
| US6096331A (en) | 1993-02-22 | 2000-08-01 | Vivorx Pharmaceuticals, Inc. | Methods and compositions useful for administration of chemotherapeutic agents |
| US5439686A (en) | 1993-02-22 | 1995-08-08 | Vivorx Pharmaceuticals, Inc. | Methods for in vivo delivery of substantially water insoluble pharmacologically active agents and compositions useful therefor |
| NZ262679A (en) | 1993-02-22 | 1997-08-22 | Vivorx Pharmaceuticals Inc | Compositions for in vivo delivery of pharmaceutical agents where the agents are contained in a polymeric shell |
| US6753006B1 (en) | 1993-02-22 | 2004-06-22 | American Bioscience, Inc. | Paclitaxel-containing formulations |
| US6537579B1 (en) | 1993-02-22 | 2003-03-25 | American Bioscience, Inc. | Compositions and methods for administration of pharmacologically active compounds |
| US5916596A (en) | 1993-02-22 | 1999-06-29 | Vivorx Pharmaceuticals, Inc. | Protein stabilized pharmacologically active agents, methods for the preparation thereof and methods for the use thereof |
| US5665382A (en) | 1993-02-22 | 1997-09-09 | Vivorx Pharmaceuticals, Inc. | Methods for the preparation of pharmaceutically active agents for in vivo delivery |
| US5840746A (en) | 1993-06-24 | 1998-11-24 | Merck Frosst Canada, Inc. | Use of inhibitors of cyclooxygenase in the treatment of neurodegenerative diseases |
| US5523325A (en) | 1993-09-09 | 1996-06-04 | Jacobson; Richard M. | Amidrazones and their use as pesticides |
| EP0763014A1 (de) | 1994-06-03 | 1997-03-19 | Basf Aktiengesellschaft | Carbamoylcarbonsäurehydrazide und ihre verwendung zur bekämpfung von schadpilzen |
| ATE184613T1 (de) * | 1995-09-22 | 1999-10-15 | Novo Nordisk As | Varianten des grünen fluoreszenzproteins, gfp |
| US5739686A (en) | 1996-04-30 | 1998-04-14 | Naughton; Michael J. | Electrically insulating cantilever magnetometer with mutually isolated and integrated thermometry, background elimination and null detection |
| US6235787B1 (en) * | 1997-06-30 | 2001-05-22 | Hoffmann-La Roche Inc. | Hydrazine derivatives |
| GB9727524D0 (en) | 1997-12-31 | 1998-02-25 | Pharmacia & Upjohn Spa | Synergistic antitumor composition containing a biologically active ureido compound |
| TW479053B (en) * | 1998-10-19 | 2002-03-11 | Agro Kanesho Co Ltd | Hydrazineoxoacetamide derivatives and pesticides |
| ES2161594B1 (es) * | 1998-12-17 | 2003-04-01 | Servier Lab | Nuevos derivados de la hidrazida, su procedimiento de preparacion y las composiciones farmaceuticas que los contienen. |
| RU2191015C2 (ru) * | 1999-11-05 | 2002-10-20 | Общество с ограниченной ответственностью "Центр бифункциональных иммуномодуляторов" | Динатриевая соль n-(6-метил-2,4-диоксо-1,2,3,4-тетрагидро-5h-пиримидинсульфон)-n'- изоникотиноилгидразида, проявляющая антимикробную и иммунотропную активность, и лекарственное средство на ее основе |
| US6322303B1 (en) * | 2000-05-12 | 2001-11-27 | David M. John | Dunnage bag and method of making same |
| EP1164126A1 (de) * | 2000-06-16 | 2001-12-19 | Basf Aktiengesellschaft | Salicylsäurehydrazid-Derivate, Verfahren und Zwischenprodukte zu ihrer Herstellung, sie enthaltende Mittel und ihre Verwendung zur Bekämpfung von Schadpilzen |
| US6365745B1 (en) * | 2000-07-14 | 2002-04-02 | Sumika Fine Chemicals Co., Ltd. | Method for producing hydrazine derivative |
| US6656971B2 (en) * | 2001-01-25 | 2003-12-02 | Guilford Pharmaceuticals Inc. | Trisubstituted carbocyclic cyclophilin binding compounds and their use |
| TWI231211B (en) | 2001-05-01 | 2005-04-21 | Abbott Lab | Compositions and methods for enhancing the bioavailability of pharmaceutical agents |
| WO2002094259A1 (en) | 2001-05-03 | 2002-11-28 | MAX-PLANCK-Gesellschaft zur Förderung der Wissenschaften e.V. | Compounds that inhibit hsp90 and stimulate hsp70 and hsp40, useful in the prevention or treatment of diseases associated with protein aggregation and amyloid formation |
| US6602907B1 (en) * | 2001-06-08 | 2003-08-05 | University Of Central Florida | Treatment of breast cancer |
| TWI332943B (en) | 2001-07-10 | 2010-11-11 | Synta Pharmaceuticals Corp | Taxol enhancer compounds |
| TWI252847B (en) * | 2001-07-10 | 2006-04-11 | Synta Pharmaceuticals Corp | Synthesis of taxol enhancers |
| TWI297335B (en) | 2001-07-10 | 2008-06-01 | Synta Pharmaceuticals Corp | Taxol enhancer compounds |
| US6924312B2 (en) * | 2001-07-10 | 2005-08-02 | Synta Pharmaceuticals Corp. | Taxol enhancer compounds |
| NZ533397A (en) | 2001-11-28 | 2005-12-23 | Sod Conseils Rech Applic | 5-sulphanyl-4H-1,2,4-triazole derivatives and their use as a medicament |
| TW200408407A (en) | 2001-11-30 | 2004-06-01 | Dana Farber Cancer Inst Inc | Methods and compositions for modulating the immune system and uses thereof |
| TWI330079B (en) | 2003-01-15 | 2010-09-11 | Synta Pharmaceuticals Corp | Treatment for cancers |
| AU2006228035B2 (en) | 2003-01-15 | 2010-02-18 | Synta Pharmaceuticals Corp. | Bis (thio-hydrazide amide) compounds for treating multi-drug resistant cancer |
| SI1611112T1 (sl) | 2003-02-11 | 2012-12-31 | Vernalis (R&D) Limited | Izoksazolne spojine kot inhibitorji vroäśinskih ĺ ok proteinov |
| KR100575251B1 (ko) | 2003-03-03 | 2006-05-02 | 재단법인서울대학교산학협력재단 | p38/JTV-1을 유효성분으로 하는 암 치료용 약학적조성물 및 암 치료용 약학적 조성물의 스크리닝 방법 |
| KR100486693B1 (ko) * | 2003-03-17 | 2005-05-03 | 삼성전자주식회사 | 금속 매립 방법 |
| EP1493445A1 (en) | 2003-07-04 | 2005-01-05 | Max-Planck-Gesellschaft zur Förderung der Wissenschaften e.V. | Inhibition of stress-induced ligand-dependent EGFR activation |
| TWI339206B (en) | 2003-09-04 | 2011-03-21 | Vertex Pharma | Compositions useful as inhibitors of protein kinases |
| KR100544347B1 (ko) | 2003-12-11 | 2006-01-23 | 한국생명공학연구원 | 디아릴이소옥사졸계 화합물을 유효성분으로 함유하는 암 예방 및 치료용 약학적 조성물 |
| CA2560954A1 (en) | 2004-03-26 | 2005-10-20 | Amphora Discovery Corporation | Certain triazole-based compounds, compositions, and uses thereof |
| US7011923B2 (en) * | 2004-04-07 | 2006-03-14 | International Business Machines Corporation | Negative photoresist and method of using thereof |
| PL1781604T3 (pl) | 2004-06-23 | 2014-01-31 | Synta Pharmaceuticals Corp | Sole bis(tiohydrazydoamidu) do leczenia nowotworów |
| WO2006033913A2 (en) | 2004-09-16 | 2006-03-30 | Synta Pharmaceuticals Corp. | Bis (thio-hydrazide amides) for treament of hyperplasia |
| WO2006062732A2 (en) | 2004-11-19 | 2006-06-15 | Synta Pharmaceuticals Corp. | Compounds acting at the centrosome |
| AU2005306471B2 (en) * | 2004-11-19 | 2009-12-17 | Synta Pharmaceuticals Corp. | Bis(thio-hydrazide amides) for increasing Hsp70 expression |
| MX2007009888A (es) | 2005-02-17 | 2007-10-16 | Synta Pharmaceuticals Corp | Compuestos para el tratamiento de trastornos proliferativos. |
| WO2006113493A2 (en) | 2005-04-15 | 2006-10-26 | Synta Pharmaceuticals Corp. | Methods of determining cancer prognosis via natural killer cell activity |
| US20090042991A1 (en) | 2005-04-15 | 2009-02-12 | James Barsoum | Methods of increasing natural killer cell activity for therapy |
| MX2007012688A (es) | 2005-04-15 | 2008-03-14 | Synta Pharmaceuticals Corp | Terapia de combinacion para el cancer con compuestos de bis(tiohidrazida) amida. |
| US7709683B2 (en) | 2005-05-16 | 2010-05-04 | Synta Pharmaceuticals Corp. | Synthesis of bis(thio-hydrazide amide) salts |
| US7678832B2 (en) | 2005-08-16 | 2010-03-16 | Synta Pharmaceuticals Corp. | Bis(thio-hydrazide amide) formulation |
| WO2007139955A2 (en) | 2006-05-25 | 2007-12-06 | Synta Pharmaceuticals Corp. | Triazole compounds that modulate hsp90 activity |
| RU2475478C2 (ru) | 2006-08-21 | 2013-02-20 | Синта Фармасьютиклз Корп. | Соединения для лечения пролиферативных расстройств |
| AU2007288340A1 (en) | 2006-08-21 | 2008-02-28 | Synta Pharmaceuticals Corp. | Bis (thiohydrazide amides) for treating melanoma |
| EP2063878A2 (en) | 2006-08-21 | 2009-06-03 | Synta Pharmaceuticals Corporation | Bis(thiohydrazide amides) for use in preventing or delaying the recurrence of melanoma |
| WO2008024299A2 (en) | 2006-08-21 | 2008-02-28 | Synta Pharmaceuticals Corp. | Combination with bis(thiohydrazide amides) for treating cancer |
| US8497272B2 (en) | 2006-08-21 | 2013-07-30 | Synta Pharmaceuticals Corp. | Compounds for treating proliferative disorders |
| WO2008024298A1 (en) | 2006-08-21 | 2008-02-28 | Synta Pharmaceuticals Corp. | Bis(thiohydrazide amides) for inhibiting angiogenesis |
| JP2010502616A (ja) | 2006-08-31 | 2010-01-28 | シンタ ファーマシューティカルズ コーポレーション | 癌を治療するためのビス(チオヒドラジドアミド)の組み合わせ |
| US20080118562A1 (en) | 2006-09-11 | 2008-05-22 | Keizo Koya | Bis(thiohydrazide amides) formulation |
| JP2010503674A (ja) | 2006-09-14 | 2010-02-04 | シンタ ファーマシューティカルズ コーポレーション | 血管形成治療用化合物 |
| WO2008033494A2 (en) | 2006-09-15 | 2008-03-20 | Synta Pharmaceuticals Corp. | Purification of bis(thiohydrazide amides) |
| US20090093538A1 (en) | 2007-01-03 | 2009-04-09 | Synta Pharmaceuticals Corp | Method for treating cancer |
| US8093425B2 (en) | 2007-04-30 | 2012-01-10 | Synta Pharmaceuticals Corp. | Compounds for treating proliferative disorders |
| US8729111B2 (en) | 2007-08-07 | 2014-05-20 | Synta Pharmaceuticals Corp. | Compounds for treating proliferative disorders |
| WO2009064374A2 (en) | 2007-11-09 | 2009-05-22 | Synta Pharmaceuticals Corp. | Oral formulations of bis(thiohydrazide amides) |
| US8637704B2 (en) | 2007-11-28 | 2014-01-28 | Synta Pharmaceuticals Corp. | Polymorphs of N-malonyl-bis(N′-methyl-N′-thiobenzoylhydrazide) |
| JP5091056B2 (ja) | 2008-08-28 | 2012-12-05 | 三甲株式会社 | 密閉容器の蓋部シール構造 |
-
2005
- 2005-06-20 PL PL05762347T patent/PL1781604T3/pl unknown
- 2005-06-20 DK DK05762347.2T patent/DK1781604T3/da active
- 2005-06-20 PT PT57623472T patent/PT1781604E/pt unknown
- 2005-06-20 BR BRPI0512526-0A patent/BRPI0512526A/pt not_active IP Right Cessation
- 2005-06-20 HR HRP20131023TT patent/HRP20131023T1/hr unknown
- 2005-06-20 JP JP2007518152A patent/JP5362986B2/ja not_active Expired - Fee Related
- 2005-06-20 EP EP05762347.2A patent/EP1781604B1/en not_active Expired - Lifetime
- 2005-06-20 ME MEP-2013-124A patent/ME01644B/me unknown
- 2005-06-20 CN CN2005800264456A patent/CN1993318B/zh not_active Expired - Fee Related
- 2005-06-20 RU RU2007101898/04A patent/RU2406722C2/ru not_active IP Right Cessation
- 2005-06-20 US US11/157,213 patent/US7385084B2/en not_active Expired - Fee Related
- 2005-06-20 WO PCT/US2005/021642 patent/WO2006009940A1/en not_active Ceased
- 2005-06-20 NZ NZ552159A patent/NZ552159A/en not_active IP Right Cessation
- 2005-06-20 CA CA2570698A patent/CA2570698C/en not_active Expired - Fee Related
- 2005-06-20 KR KR1020077001513A patent/KR101313027B1/ko not_active Expired - Fee Related
- 2005-06-20 EP EP10009536A patent/EP2305642A3/en not_active Withdrawn
- 2005-06-20 ES ES05762347T patent/ES2430373T3/es not_active Expired - Lifetime
- 2005-06-20 AU AU2005265202A patent/AU2005265202B2/en not_active Ceased
- 2005-06-20 RS RS20130458A patent/RS52999B/sr unknown
- 2005-06-21 TW TW094120599A patent/TWI389684B/zh not_active IP Right Cessation
-
2006
- 2006-11-27 IL IL179632A patent/IL179632A/en not_active IP Right Cessation
-
2007
- 2007-01-19 NO NO20070378A patent/NO20070378L/no not_active Application Discontinuation
-
2008
- 2008-04-18 US US12/148,312 patent/US7579503B2/en not_active Expired - Fee Related
-
2009
- 2009-07-15 US US12/503,661 patent/US7795313B2/en not_active Expired - Fee Related
-
2010
- 2010-08-30 US US12/871,587 patent/US8048925B2/en not_active Expired - Fee Related
- 2010-11-08 RU RU2010145056/04A patent/RU2010145056A/ru not_active Application Discontinuation
-
2011
- 2011-10-14 US US13/273,807 patent/US8461208B2/en not_active Expired - Fee Related
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2406722C2 (ru) | Соли бис-(тиогидразидамида) для лечения рака | |
| US7709683B2 (en) | Synthesis of bis(thio-hydrazide amide) salts | |
| RU2002111333A (ru) | Способ получения производных 4''-замещенных 9-деоксо-9а-аза-9а-гомоэритромицина а | |
| PT100943B (pt) | Processo aperfeicoado para a sintese de (5r)-6-ciano-5-hidroxi-3-oxo-hexanoato de 1,1-dimetiletilo | |
| RU2010152758A (ru) | Кристаллическая форма и тозилат соединения тетрациклина, кристаллическая форма указанного тозилата и ее полиморфная модификация, способ получения тозилата соединения тетрациклина, полиморфная модификация, полученная указанным способом, и фармацевтические композиции на основе вышеуказанных кристаллической формы и полиморфной модификации | |
| JP2008504264A5 (enExample) | ||
| RU2018132264A (ru) | Промежуточные продукты в синтезе эрибулина и соответствующие способы синтеза | |
| RU2008141850A (ru) | Жидкий фармацевтический препарат с немедленным высвобождением | |
| RU2011140724A (ru) | Способ модификации гиалуроновой кислоты с помощью комплекса (о-ацил-о'-алкилкарбонат - замещенный пиридин) | |
| FI129024B (en) | Process for the preparation of bicyclic guanidine and its derivatives | |
| RU2011139325A (ru) | Аморфная соль макроциклического ингибитора hcv | |
| JP2008540658A5 (enExample) | ||
| Vashishtha et al. | Organobase catalysis using 1-(2-pyrimidyl) piperazine in micellar medium: an approach for better performance and reusability of organobase | |
| Das et al. | Stereoselective direct reductive amination of ketones with electron-deficient amines using Re 2 O 7/NaPF 6 catalyst | |
| AR051373A1 (es) | Procedimiento para la preparacion de analogos aciclicos de nucleotidos de fosfonatos | |
| AR047544A1 (es) | Proceso para la preparacion de compuestos de triazol sustituidos | |
| GB1340720A (en) | Benzomorphan derivatives processes for producing them and composi tions containing them | |
| CA2671816A1 (en) | Salts of imidazole-5-carboxylic acid derivatives, a method for preparing same and pharmaceutical compositions comprising same | |
| WO2013140417A4 (en) | BORONIC ACID BEARING LIPHAGANE COMPOUNDS AS INHIBITORS OF PI3K-α AND/OR β | |
| MX379419B (es) | Proceso para preparar compuestos de aminotioléster y sus sales. | |
| US10138188B2 (en) | Catalyst for producing methanol precursor, methanol precursor produced using the catalyst and methanol produced using the methanol precursor | |
| RU2012117977A (ru) | Способ синтеза 2-тиогистидина и его аналогов | |
| RU2013110311A (ru) | Синтез соединений тиазолидиндиона | |
| CN111471035B (zh) | 一类ido抑制剂的制备方法 | |
| US11020730B2 (en) | Halogen-containing metathesis catalysts and methods thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| MM4A | The patent is invalid due to non-payment of fees |
Effective date: 20160621 |