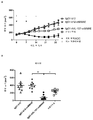
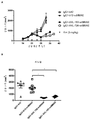
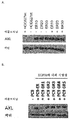
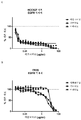
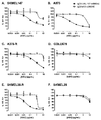
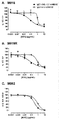
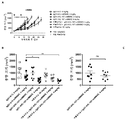
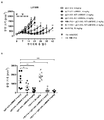
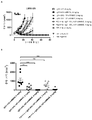
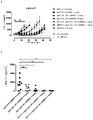
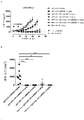
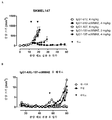
KR20180033523A - 암 치료를 위한 axl-특이적 항체-약물 접합체 - Google Patents
암 치료를 위한 axl-특이적 항체-약물 접합체 Download PDFInfo
- Publication number
- KR20180033523A KR20180033523A KR1020187004011A KR20187004011A KR20180033523A KR 20180033523 A KR20180033523 A KR 20180033523A KR 1020187004011 A KR1020187004011 A KR 1020187004011A KR 20187004011 A KR20187004011 A KR 20187004011A KR 20180033523 A KR20180033523 A KR 20180033523A
- Authority
- KR
- South Korea
- Prior art keywords
- region
- seq
- cdr2
- cdr1
- adc
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001102—Receptors, cell surface antigens or cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/68031—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug being an auristatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6849—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a receptor, a cell surface antigen or a cell surface determinant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6855—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from breast cancer cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6857—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from lung cancer cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6869—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from a cell of the reproductive system: ovaria, uterus, testes, prostate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2863—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against receptors for growth factors, growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/34—Identification of a linear epitope shorter than 20 amino acid residues or of a conformational epitope defined by amino acid residues
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
- C07K2317/732—Antibody-dependent cellular cytotoxicity [ADCC]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/77—Internalization into the cell
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Immunology (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Cell Biology (AREA)
- Organic Chemistry (AREA)
- Oncology (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pulmonology (AREA)
- Reproductive Health (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EPPCT/EP2015/065900 | 2015-07-10 | ||
| PCT/EP2015/065900 WO2016005593A1 (en) | 2014-07-11 | 2015-07-10 | Antibodies binding axl |
| US201662278283P | 2016-01-13 | 2016-01-13 | |
| US62/278,283 | 2016-01-13 | ||
| PCT/EP2016/066353 WO2017009258A1 (en) | 2015-07-10 | 2016-07-08 | Axl-specific antibody-drug conjugates for cancer treatment |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20180033523A true KR20180033523A (ko) | 2018-04-03 |
Family
ID=57756861
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187004011A Ceased KR20180033523A (ko) | 2015-07-10 | 2016-07-08 | 암 치료를 위한 axl-특이적 항체-약물 접합체 |
Country Status (22)
| Country | Link |
|---|---|
| US (4) | US20180326084A1 (enExample) |
| EP (3) | EP3319993B1 (enExample) |
| JP (5) | JP6892431B2 (enExample) |
| KR (1) | KR20180033523A (enExample) |
| CN (3) | CN108368171A (enExample) |
| AU (3) | AU2016292762B2 (enExample) |
| CA (2) | CA2991805A1 (enExample) |
| CY (1) | CY1123983T1 (enExample) |
| DK (1) | DK3319993T3 (enExample) |
| EA (2) | EA201890272A1 (enExample) |
| ES (2) | ES2784685T3 (enExample) |
| HR (1) | HRP20200551T1 (enExample) |
| HU (1) | HUE049072T2 (enExample) |
| IL (2) | IL256790B (enExample) |
| LT (1) | LT3319993T (enExample) |
| ME (1) | ME03772B (enExample) |
| PL (1) | PL3319993T3 (enExample) |
| PT (1) | PT3319993T (enExample) |
| RS (1) | RS60141B1 (enExample) |
| SI (1) | SI3319993T1 (enExample) |
| SM (1) | SMT202000359T1 (enExample) |
| WO (2) | WO2017009258A1 (enExample) |
Families Citing this family (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL296633A (en) | 2014-07-11 | 2022-11-01 | Genmab As | Antibodies that bind axl |
| CA2991805A1 (en) * | 2015-07-10 | 2017-01-19 | Genmab A/S | Axl-specific antibody-drug conjugates for cancer treatment |
| JP2019505520A (ja) | 2016-01-13 | 2019-02-28 | ゲンマブ エー/エス | 抗体およびその薬物コンジュゲートの製剤 |
| CN109311858B (zh) | 2016-05-26 | 2021-12-03 | 里科瑞尔姆Ip控股有限责任公司 | Egfr抑制剂化合物 |
| WO2018007592A1 (en) | 2016-07-08 | 2018-01-11 | Genmab A/S | New dosage regimens for antibody drug conjugates based on anti-axl antibodies |
| WO2018051306A1 (en) | 2016-09-19 | 2018-03-22 | Novartis Ag | Therapeutic combinations comprising a raf inhibitor and a erk inhibitor |
| MX393780B (es) | 2017-01-17 | 2025-03-24 | Heparegenix Gmbh | Inhibidores de proteina cinasa para promover la regeneracion hepatica o reducir o prevenir la muerte de hepatocitos |
| KR20240032157A (ko) | 2017-05-02 | 2024-03-08 | 노파르티스 아게 | 병용 요법 |
| KR102811888B1 (ko) * | 2017-09-08 | 2025-05-27 | 에프. 호프만-라 로슈 아게 | 암의 진단 및 치료 방법 |
| SG11202002331QA (en) * | 2017-09-13 | 2020-04-29 | Nat Res Council Canada | Axl-specific antibodies and uses thereof |
| CN111936141A (zh) * | 2018-03-30 | 2020-11-13 | 诺华股份有限公司 | 包含达拉菲尼、曲美替尼和erk抑制剂的三重药物组合 |
| MA52657A (fr) * | 2018-04-10 | 2021-02-17 | Genmab As | Anticorps spécifiques d'axl pour le traitement du cancer |
| CN110483639A (zh) | 2018-05-15 | 2019-11-22 | 复旦大学 | 靶向axl的抗体及抗体-药物偶联物及其制备方法和用途 |
| CN110540592B (zh) | 2018-05-29 | 2022-08-09 | 杭州尚健生物技术有限公司 | 结合axl蛋白的抗体及其应用 |
| EP3856783A1 (en) * | 2018-09-26 | 2021-08-04 | Genmab A/S | Axl-specific antibodies for treatment of non-small cell lung cancer |
| WO2020188086A1 (en) * | 2019-03-20 | 2020-09-24 | Imcheck Therapeutics Sas | Antibodies having specificity for btn2 and uses thereof |
| WO2020205576A1 (en) * | 2019-03-29 | 2020-10-08 | Celldex Therapeutics, Inc. | Anti-axl antibodies and methods of use thereof |
| MX2021013817A (es) | 2019-05-13 | 2021-12-14 | Novartis Ag | Nuevas formas cristalinas de n-(3-(2-(2-hidroxietoxi)-6-morfolinop iridin-4-il)-4-metilfenil)-2-(trifluorometil)isonicotinamida como inhibidores de raf para el tratamiento del cancer. |
| WO2021154156A1 (en) * | 2020-01-31 | 2021-08-05 | Agency For Science, Technology And Research | Anti-axl antibody and uses thereof |
| CN115210263A (zh) * | 2020-02-28 | 2022-10-18 | 西福根有限公司 | 抗axl抗体和组合物 |
| CN115551509A (zh) * | 2020-05-12 | 2022-12-30 | 诺华股份有限公司 | 包含craf抑制剂的治疗组合 |
| WO2022026807A2 (en) * | 2020-07-30 | 2022-02-03 | Albert Einstein College Of Medicine | Antibodies targeting sars-cov-2 and uses thereof |
| KR20230107606A (ko) * | 2020-11-06 | 2023-07-17 | 데이 원 바이오파마슈티칼즈, 인크. | 저등급 신경교종 치료를 위한 raf 억제제 |
| KR102549520B1 (ko) | 2021-01-29 | 2023-07-03 | 일리미스테라퓨틱스 주식회사 | 비염증성 식세포작용 유도 활성을 갖는 융합분자 |
| CN117177754A (zh) * | 2021-02-19 | 2023-12-05 | 首日生物制药公司 | Raf抑制剂和mek抑制剂的组合 |
| AU2024224437A1 (en) * | 2023-02-22 | 2025-08-14 | Resolute Science, Inc. | Compositions and methods for targeting tumor-associated macrophages |
| WO2024178140A1 (en) * | 2023-02-22 | 2024-08-29 | Resolute Science, Inc. | Compositions and methods for targeting tumor-associated macrophages |
Family Cites Families (102)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US761218A (en) | 1899-10-05 | 1904-05-31 | U S Standard Voting Machine Co | Register or counter. |
| US4179337A (en) | 1973-07-20 | 1979-12-18 | Davis Frank F | Non-immunogenic polypeptides |
| JPS5896026A (ja) | 1981-10-30 | 1983-06-07 | Nippon Chemiphar Co Ltd | 新規ウロキナ−ゼ誘導体およびその製造法ならびにそれを含有する血栓溶解剤 |
| US4609546A (en) | 1982-06-24 | 1986-09-02 | Japan Chemical Research Co., Ltd. | Long-acting composition |
| US4681581A (en) | 1983-12-05 | 1987-07-21 | Coates Fredrica V | Adjustable size diaper and folding method therefor |
| US4766106A (en) | 1985-06-26 | 1988-08-23 | Cetus Corporation | Solubilization of proteins for pharmaceutical compositions using polymer conjugation |
| US4735210A (en) | 1985-07-05 | 1988-04-05 | Immunomedics, Inc. | Lymphographic and organ imaging method and kit |
| US5776093A (en) | 1985-07-05 | 1998-07-07 | Immunomedics, Inc. | Method for imaging and treating organs and tissues |
| US5101827A (en) | 1985-07-05 | 1992-04-07 | Immunomedics, Inc. | Lymphographic and organ imaging method and kit |
| US5057313A (en) | 1986-02-25 | 1991-10-15 | The Center For Molecular Medicine And Immunology | Diagnostic and therapeutic antibody conjugates |
| US5648471A (en) | 1987-12-03 | 1997-07-15 | Centocor, Inc. | One vial method for labeling antibodies with Technetium-99m |
| GB8823869D0 (en) | 1988-10-12 | 1988-11-16 | Medical Res Council | Production of antibodies |
| US5703055A (en) | 1989-03-21 | 1997-12-30 | Wisconsin Alumni Research Foundation | Generation of antibodies through lipid mediated DNA delivery |
| ATE172879T1 (de) | 1989-08-09 | 1998-11-15 | Rhomed Inc | Direkte radioetikettierung von antikörpern und sonstigen proteinen mittels technetium oder rhenium |
| ES2108048T3 (es) | 1990-08-29 | 1997-12-16 | Genpharm Int | Produccion y utilizacion de animales inferiores transgenicos capaces de producir anticuerpos heterologos. |
| US5789650A (en) | 1990-08-29 | 1998-08-04 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5545806A (en) | 1990-08-29 | 1996-08-13 | Genpharm International, Inc. | Ransgenic non-human animals for producing heterologous antibodies |
| US5661016A (en) | 1990-08-29 | 1997-08-26 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US6300129B1 (en) | 1990-08-29 | 2001-10-09 | Genpharm International | Transgenic non-human animals for producing heterologous antibodies |
| US5877397A (en) | 1990-08-29 | 1999-03-02 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5625126A (en) | 1990-08-29 | 1997-04-29 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5633425A (en) | 1990-08-29 | 1997-05-27 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5770429A (en) | 1990-08-29 | 1998-06-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5874299A (en) | 1990-08-29 | 1999-02-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5814318A (en) | 1990-08-29 | 1998-09-29 | Genpharm International Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5194594A (en) | 1990-09-07 | 1993-03-16 | Techniclone, Inc. | Modified antibodies |
| AU2235992A (en) | 1991-06-14 | 1993-01-12 | Genpharm International, Inc. | Transgenic immunodeficient non-human animals |
| JPH06508880A (ja) | 1991-07-08 | 1994-10-06 | ユニバーシティ オブ マサチューセッツ アット アムハースト | サーモトロピック液晶セグメント化ブロックコポリマー |
| US5635483A (en) | 1992-12-03 | 1997-06-03 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Tumor inhibiting tetrapeptide bearing modified phenethyl amides |
| US5780588A (en) | 1993-01-26 | 1998-07-14 | Arizona Board Of Regents | Elucidation and synthesis of selected pentapeptides |
| JPH08509612A (ja) | 1993-04-26 | 1996-10-15 | ジェンファーム インターナショナル インコーポレイテッド | 異種抗体を産生することができるトランスジェニック非ヒト動物 |
| US6214345B1 (en) | 1993-05-14 | 2001-04-10 | Bristol-Myers Squibb Co. | Lysosomal enzyme-cleavable antitumor drug conjugates |
| US6077835A (en) | 1994-03-23 | 2000-06-20 | Case Western Reserve University | Delivery of compacted nucleic acid to cells |
| US5663149A (en) | 1994-12-13 | 1997-09-02 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory pentapeptide heterocyclic and halophenyl amides |
| KR970029803A (ko) | 1995-11-03 | 1997-06-26 | 김광호 | 반도체 메모리장치의 프리차지 회로 |
| DK0979281T3 (da) | 1997-05-02 | 2005-11-21 | Genentech Inc | Fremgangsmåde til fremstilling af multispecifikke antistoffer med heteromultimere og fælles bestanddele |
| DE60013773T2 (de) | 1999-02-03 | 2005-11-10 | Biosante Pharmaceuticals, Inc. | Methoden zur Herstellung von therapeutischen Kalziumphosphat Partikeln |
| US6281005B1 (en) | 1999-05-14 | 2001-08-28 | Copernicus Therapeutics, Inc. | Automated nucleic acid compaction device |
| IL147765A0 (en) | 1999-07-29 | 2002-08-14 | Medarex Inc | HUMAN MONOCLONAL ANTIBODIES TO HER2/neu |
| DE10043437A1 (de) | 2000-09-04 | 2002-03-28 | Horst Lindhofer | Verwendung von trifunktionellen bispezifischen und trispezifischen Antikörpern zur Behandlung von malignem Aszites |
| EP1243276A1 (en) | 2001-03-23 | 2002-09-25 | Franciscus Marinus Hendrikus De Groot | Elongated and multiple spacers containing activatible prodrugs |
| US7129261B2 (en) | 2001-05-31 | 2006-10-31 | Medarex, Inc. | Cytotoxic agents |
| PT2314629E (pt) | 2002-07-18 | 2014-01-22 | Merus B V | Produção recombinante de misturas de anticorpos |
| DK1545613T3 (da) | 2002-07-31 | 2011-11-14 | Seattle Genetics Inc | Auristatinkonjugater og deres anvendelse til behandling af cancer, en autoimmun sygdom eller en infektiøs sygdom |
| WO2004043493A1 (en) | 2002-11-14 | 2004-05-27 | Syntarga B.V. | Prodrugs built as multiple self-elimination-release spacers |
| CN107213469A (zh) | 2003-11-06 | 2017-09-29 | 西雅图基因公司 | 能够与配体偶联的单甲基缬氨酸化合物 |
| US7741568B2 (en) | 2005-01-13 | 2010-06-22 | The Wiremold Company | Downward facing receptacle assembly for cable raceway |
| TWI671403B (zh) | 2005-03-31 | 2019-09-11 | 中外製藥股份有限公司 | 控制組裝之多肽的製造方法 |
| CA2617907A1 (en) | 2005-08-05 | 2007-02-15 | Syntarga B.V. | Triazole-containing releasable linkers and conjugates comprising the same |
| US7612181B2 (en) | 2005-08-19 | 2009-11-03 | Abbott Laboratories | Dual variable domain immunoglobulin and uses thereof |
| AU2006317242A1 (en) | 2005-11-28 | 2007-05-31 | Genmab A/S | Recombinant monovalent antibodies and methods for production thereof |
| CN101415679A (zh) | 2006-02-02 | 2009-04-22 | 辛塔佳有限公司 | 水溶性cc-1065类似物及其缀合物 |
| GEP20135917B (en) | 2006-03-17 | 2013-09-10 | Biogen Idec Inc | Stabilized polypeptide compositions |
| ES2395969T3 (es) | 2006-03-24 | 2013-02-18 | Merck Patent Gmbh | Dominios de proteínas heterodiméricas genéticamente modificados |
| AT503902B1 (de) | 2006-07-05 | 2008-06-15 | F Star Biotech Forsch & Entw | Verfahren zur manipulation von immunglobulinen |
| EP2070956A1 (en) | 2007-12-14 | 2009-06-17 | Total Petrochemicals Research Feluy | Process for the production of a bimodal polypropylene having low ash content |
| ES2702087T3 (es) | 2007-06-21 | 2019-02-27 | Macrogenics Inc | Diacuerpos covalentes y sus usos |
| WO2009017394A1 (en) | 2007-08-01 | 2009-02-05 | Syntarga B.V. | Substituted cc-1065 analogs and their conjugates |
| EP2050764A1 (en) | 2007-10-15 | 2009-04-22 | sanofi-aventis | Novel polyvalent bispecific antibody format and uses thereof |
| CN101939336B (zh) * | 2007-11-12 | 2014-05-14 | U3制药有限公司 | Axl抗体 |
| TW200936160A (en) | 2007-11-15 | 2009-09-01 | Chugai Pharmaceutical Co Ltd | Monoclonal antibodies that bind to anexelekto and uses thereof |
| US8227577B2 (en) | 2007-12-21 | 2012-07-24 | Hoffman-La Roche Inc. | Bivalent, bispecific antibodies |
| US20090162359A1 (en) | 2007-12-21 | 2009-06-25 | Christian Klein | Bivalent, bispecific antibodies |
| US9266967B2 (en) | 2007-12-21 | 2016-02-23 | Hoffmann-La Roche, Inc. | Bivalent, bispecific antibodies |
| US8242247B2 (en) | 2007-12-21 | 2012-08-14 | Hoffmann-La Roche Inc. | Bivalent, bispecific antibodies |
| SI2235064T1 (sl) | 2008-01-07 | 2016-04-29 | Amgen Inc. | Metoda za izdelavo heterodimernih molekul - protitelesa fc z uporabo elektrostatičnih usmerjevalnih učinkov |
| WO2010015792A1 (en) | 2008-08-06 | 2010-02-11 | Argenta Discovery Limited | Nitrogen containing heterocyclic compounds useful as bifunctional modulators of m3 receptors and beta-2 receptors |
| JP5397668B2 (ja) | 2008-09-02 | 2014-01-22 | ソニー株式会社 | 記憶素子および記憶装置 |
| CA2742568C (en) | 2008-11-03 | 2017-09-26 | Syntarga B.V. | Novel cc-1065 analogs and their conjugates |
| SG172254A1 (en) | 2008-12-19 | 2011-07-28 | Macrogenics Inc | Covalent diabodies and uses thereof |
| CN102459346B (zh) | 2009-04-27 | 2016-10-26 | 昂考梅德药品有限公司 | 制造异源多聚体分子的方法 |
| MX2011011825A (es) | 2009-05-11 | 2011-12-06 | U3 Pharma Gmbh | Anticuerpos humanizados para axl. |
| CN102459344A (zh) | 2009-05-15 | 2012-05-16 | 中外制药株式会社 | 抗axl抗体 |
| KR101747103B1 (ko) | 2009-06-26 | 2017-06-14 | 리제너론 파마슈티칼스 인코포레이티드 | 천연 면역글로불린 포맷을 가지는 용이하게 분리된 이중특이성 항체 |
| LT2769737T (lt) * | 2009-07-20 | 2017-06-26 | Bristol-Myers Squibb Company | Anti-ctla4 antikūno derinys su etopozidu, skirtas sinerginiam proliferacinių ligų gydymui |
| US9493578B2 (en) | 2009-09-02 | 2016-11-15 | Xencor, Inc. | Compositions and methods for simultaneous bivalent and monovalent co-engagement of antigens |
| EP2548027A2 (en) * | 2010-03-18 | 2013-01-23 | Institut National de la Santé et de la Recherche Médicale | Method for predicting the responsiveness to chemotherapy |
| TWI426920B (zh) | 2010-03-26 | 2014-02-21 | Hoffmann La Roche | 雙專一性、雙價抗-vegf/抗-ang-2抗體 |
| PL2560993T3 (pl) | 2010-04-20 | 2024-11-04 | Genmab A/S | Heterodimeryczne białka zawierające region FC przeciwciała i sposoby ich wytwarzania |
| CA2797981C (en) | 2010-05-14 | 2019-04-23 | Rinat Neuroscience Corporation | Heterodimeric proteins and methods for producing and purifying them |
| WO2011159980A1 (en) | 2010-06-18 | 2011-12-22 | Genentech, Inc. | Anti-axl antibodies and methods of use |
| RU2608640C2 (ru) | 2010-08-16 | 2017-01-23 | Новиммун С.А. | Способы получения мультиспецифичных и мультивалентных антител |
| RU2013110876A (ru) | 2010-08-24 | 2014-09-27 | Рош Гликарт Аг | Активируемые биспецифические антитела |
| RU2013110875A (ru) | 2010-08-24 | 2014-09-27 | Ф.Хоффманн-Ля Рош Аг | БИСПЕЦИФИЧЕСКИЕ АНТИТЕЛА, СОДЕРЖАЩИЕ СТАБИЛИЗИРОВАННЫЙ ДИСУЛЬФИДОМ ФРАГМЕНТ Fv |
| AU2011325833C1 (en) | 2010-11-05 | 2017-07-13 | Zymeworks Bc Inc. | Stable heterodimeric antibody design with mutations in the Fc domain |
| CN102250246A (zh) | 2011-06-10 | 2011-11-23 | 常州亚当生物技术有限公司 | 抗VEGF/PDGFRβ双特异性抗体及其应用 |
| JP6033293B2 (ja) * | 2011-06-22 | 2016-11-30 | インサーム(インスティテュ ナシオナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシェ メディカル)Inserm(Institut National Dela Sante Et De La Recherche Medicale) | 抗Axl抗体及びその使用 |
| WO2012175692A1 (en) | 2011-06-22 | 2012-12-27 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Anti-axl antibodies and uses thereof |
| EP2589609A1 (en) | 2011-11-03 | 2013-05-08 | Pierre Fabre Medicament | Antigen binding protein and its use as addressing product for the treatment of cancer |
| US9879061B2 (en) | 2011-12-15 | 2018-01-30 | The Board Of Trustees Of The Leland Stanford Junior University | Inhibition of AXL/GAS6 signaling in the treatment of liver fibrosis |
| HRP20201004T1 (hr) | 2011-12-20 | 2020-10-16 | Medimmune, Llc | Modificirani polipeptidi za skelete bispecifičnog protutijela |
| DK2838917T3 (da) | 2012-04-20 | 2019-08-26 | Merus Nv | Fremgangsmåder og midler til frembringelse af heterodimere ig-lignende molekyler |
| HK1208216A1 (en) | 2012-05-15 | 2016-02-26 | Concortis Biosystems, Corp | Drug-conjugates, conjugation methods, and uses thereof |
| US9624308B2 (en) * | 2012-11-05 | 2017-04-18 | Pierre Fabre Medicament | Antigen binding proteins |
| US20150315552A1 (en) | 2012-12-14 | 2015-11-05 | The Board Of Trustees Of The Leland Stanford Junior University | Inhibition of AXL Signaling in Primary Tumor Therapy |
| US20160097370A1 (en) * | 2013-03-26 | 2016-04-07 | James L. Rodgers | High-torque wind turbine |
| WO2014174111A1 (en) | 2013-04-26 | 2014-10-30 | Pierre Fabre Medicament | Axl antibody-drug conjugate and its use for the treatment of cancer |
| US10028958B2 (en) * | 2014-04-19 | 2018-07-24 | Massachusetts Institute Of Technology | Methods of treating cancer with a combination of selected MEK1/2 and AXL inhibitors |
| GB201410825D0 (en) * | 2014-06-18 | 2014-07-30 | Bergenbio As | Anti-axl antibodies |
| IL296633A (en) * | 2014-07-11 | 2022-11-01 | Genmab As | Antibodies that bind axl |
| CA2991805A1 (en) * | 2015-07-10 | 2017-01-19 | Genmab A/S | Axl-specific antibody-drug conjugates for cancer treatment |
| WO2018007592A1 (en) * | 2016-07-08 | 2018-01-11 | Genmab A/S | New dosage regimens for antibody drug conjugates based on anti-axl antibodies |
-
2016
- 2016-07-08 CA CA2991805A patent/CA2991805A1/en active Pending
- 2016-07-08 US US15/742,818 patent/US20180326084A1/en not_active Abandoned
- 2016-07-08 EA EA201890272A patent/EA201890272A1/ru unknown
- 2016-07-08 ME MEP-2020-74A patent/ME03772B/me unknown
- 2016-07-08 DK DK16739079.8T patent/DK3319993T3/da active
- 2016-07-08 SM SM20200359T patent/SMT202000359T1/it unknown
- 2016-07-08 HU HUE16739079A patent/HUE049072T2/hu unknown
- 2016-07-08 ES ES16739079T patent/ES2784685T3/es active Active
- 2016-07-08 HR HRP20200551TT patent/HRP20200551T1/hr unknown
- 2016-07-08 EP EP16739079.8A patent/EP3319993B1/en active Active
- 2016-07-08 CN CN201680052378.3A patent/CN108368171A/zh active Pending
- 2016-07-08 EP EP20151844.6A patent/EP3730520A1/en not_active Withdrawn
- 2016-07-08 AU AU2016292762A patent/AU2016292762B2/en active Active
- 2016-07-08 KR KR1020187004011A patent/KR20180033523A/ko not_active Ceased
- 2016-07-08 WO PCT/EP2016/066353 patent/WO2017009258A1/en not_active Ceased
- 2016-07-08 JP JP2018500605A patent/JP6892431B2/ja not_active Expired - Fee Related
- 2016-07-08 LT LTEP16739079.8T patent/LT3319993T/lt unknown
- 2016-07-08 PL PL16739079T patent/PL3319993T3/pl unknown
- 2016-07-08 SI SI201630699T patent/SI3319993T1/sl unknown
- 2016-07-08 PT PT167390798T patent/PT3319993T/pt unknown
- 2016-07-08 RS RS20200419A patent/RS60141B1/sr unknown
-
2017
- 2017-01-13 CN CN202310359872.6A patent/CN116617410A/zh active Pending
- 2017-01-13 JP JP2018536482A patent/JP2019509257A/ja active Pending
- 2017-01-13 WO PCT/EP2017/050718 patent/WO2017121877A1/en not_active Ceased
- 2017-01-13 ES ES17701812T patent/ES3029907T3/es active Active
- 2017-01-13 CN CN201780017252.7A patent/CN108884165A/zh active Pending
- 2017-01-13 EP EP17701812.4A patent/EP3402822B1/en active Active
- 2017-01-13 EA EA201891607A patent/EA201891607A1/ru unknown
- 2017-01-13 US US16/069,395 patent/US20190022243A1/en not_active Abandoned
- 2017-01-13 CA CA3010887A patent/CA3010887A1/en active Pending
- 2017-01-13 AU AU2017206967A patent/AU2017206967B2/en active Active
-
2018
- 2018-01-08 IL IL256790A patent/IL256790B/en active IP Right Grant
- 2018-06-17 IL IL260065A patent/IL260065A/en unknown
-
2020
- 2020-04-13 CY CY20201100339T patent/CY1123983T1/el unknown
- 2020-06-03 US US16/891,796 patent/US20200397913A1/en active Pending
-
2021
- 2021-05-27 JP JP2021088877A patent/JP7428680B2/ja active Active
- 2021-12-21 JP JP2021207104A patent/JP2022037170A/ja not_active Withdrawn
-
2022
- 2022-09-30 US US17/957,302 patent/US20230321261A1/en active Pending
- 2022-10-28 AU AU2022259847A patent/AU2022259847C1/en active Active
-
2024
- 2024-01-25 JP JP2024009200A patent/JP2024038480A/ja active Pending
Also Published As
Similar Documents
| Publication | Publication Date | Title |
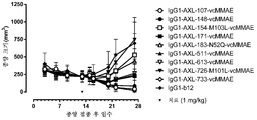
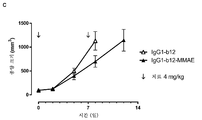
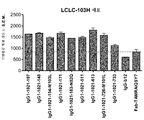
|---|---|---|
| AU2022259847C1 (en) | AXL-specific antibody-drug conjugates for cancer treatment | |
| KR102586656B1 (ko) | Axl에 결합하는 항체 | |
| HK1255412B (en) | Axl-specific antibody-drug conjugates for cancer treatment | |
| HK40000535B (en) | Axl‑specific antibody‑drug conjugates for cancer treatment | |
| HK40000535A (en) | Axl‑specific antibody‑drug conjugates for cancer treatment | |
| BR122024013731A2 (pt) | Anticorpo, imunoconjugado, composição, célula hospedeira, uso do anticorpo, métodos para produzir um anticorpo e para detectar a presença de um anticorpo axl ou uma célula que expressa axl, e, kit | |
| BR122024013731B1 (pt) | Anticorpo, imunoconjugado, composição, célula hospedeira, uso do anticorpo, métodos para produzir um anticorpo e para detectar a presença de um anticorpo axl ou uma célula que expressa axl, e, kit | |
| BR122024013726B1 (pt) | Anticorpo, imunoconjugado, composição, célula hospedeira, uso do anticorpo, métodos para produzir um anticorpo e para detectar a presença de um anticorpo axl ou uma célula que expressa axl, e, kit | |
| BR112017000316B1 (pt) | Anticorpo, imunoconjugado, composição, célula hospedeira, hibridoma, uso do anticorpo, métodos in vitro para produzir um anticorpo e para detectar a presença de um anticorpo axl ou uma célula que expressa axl, e, kit | |
| BR122024013726A2 (pt) | Anticorpo, imunoconjugado, composição, célula hospedeira, uso do anticorpo, métodos para produzir um anticorpo e para detectar a presença de um anticorpo axl ou uma célula que expressa axl, e, kit | |
| BR122024013722A2 (pt) | Anticorpo, imunoconjugado, composição, célula hospedeira, uso do anticorpo, métodos para produzir um anticorpo e para detectar a presença de um anticorpo axl ou uma célula que expressa axl, e, kit | |
| BR122024013722B1 (pt) | Anticorpo, imunoconjugado, composição, célula hospedeira, uso do anticorpo, métodos para produzir um anticorpo e para detectar a presença de um anticorpo axl ou uma célula que expressa axl, e, kit |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20180209 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20210708 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20240128 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20240329 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20240128 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |