EP3346052B1 - Verfahren zur herstellung von verarbeiteter faser, verarbeitete faser, verfahren zur unterdrückung von schäden an tierfasern und verfahren zur verarbeitung von tierfaser - Google Patents
Verfahren zur herstellung von verarbeiteter faser, verarbeitete faser, verfahren zur unterdrückung von schäden an tierfasern und verfahren zur verarbeitung von tierfaser Download PDFInfo
- Publication number
- EP3346052B1 EP3346052B1 EP16841846.5A EP16841846A EP3346052B1 EP 3346052 B1 EP3346052 B1 EP 3346052B1 EP 16841846 A EP16841846 A EP 16841846A EP 3346052 B1 EP3346052 B1 EP 3346052B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- keratin
- fibers
- bleaching
- hairs
- hydrolyzed keratin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/01—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with natural macromolecular compounds or derivatives thereof
- D06M15/15—Proteins or derivatives thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
- D06L4/10—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs using agents which develop oxygen
- D06L4/12—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs using agents which develop oxygen combined with specific additives
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/44—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders
- D06P1/46—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders using compositions containing natural macromolecular substances or derivatives thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2101/00—Chemical constitution of the fibres, threads, yarns, fabrics or fibrous goods made from such materials, to be treated
- D06M2101/02—Natural fibres, other than mineral fibres
- D06M2101/10—Animal fibres
Definitions
- the present invention relates to a method for manufacturing processed fibers bleached and/or dyed, the processed fiber, a method for suppressing damage to animal fibers, and a method for processing animal fibers.
- the texture and hue of a fiber product made of animal fibers are important factors in determining the product value.
- the texture of animal fibers is expressed as "tender”, “smooth”, “frictional feel”, or the like, and it is thought that animal fibers have the most excellent texture when these fibers are collected and no chemical treatment other than scouring is performed.
- dyeing is performed in accordance with purposes, and when a vivid hue such as white or extremely light color to light color is required, it is necessary to bleach fibers prior to dyeing.
- oxidative bleaching by hydrogen peroxide or reductive bleaching by hydrosulfite or sodium bisulfite has been performed.
- these types of bleaching require long-term heating and an alkali agent, and thus fibers are damaged, causing deterioration of unique textures such as tenderness and frictional feel of animal fibers.
- Patent Literature 1 a bleaching sub-agent containing a polycarboxylic acid-based polymer and an amine-based compound, and a treating method by means of an alkaline protease, hydroxyalkyl phosphine, and derivatives thereof (Patent Literature 2) have been proposed.
- Patent Literature 2 a bleaching sub-agent containing a polycarboxylic acid-based polymer and an amine-based compound
- Patent Literature 2 a treating method by means of an alkaline protease, hydroxyalkyl phosphine, and derivatives thereof
- Patent Literature 3 a method applying an ultraviolet absorbing agent
- Patent Literature 4 a method applying a lower phosphorous acid and a lower phosphate to animal fibers
- Patent Literature 5 a method applying a fluorescent whitening agent to animal fibers
- their effects are not necessarily sufficient, and the textures are harmed, and their safety with regard to the human body may be also a concern.
- Patent Literatures 6 to 8 Conventionly, methods for providing fibers with the properties of an animal-derived protein by combining the animal-derived protein and animal fibers have been proposed (Patent Literatures 6 to 8). For example, it has been reported that use of sericin together with a dichlorotriazine-based compound provides fibers with durability and shape stability (Patent Literature 6); that in shape fixing, cloth made of animal protein fibers is soaked for absorption into a solution of a collagen protein derivative and a fibroin protein or a keratin solution, and thereby, the texture and touch feel are maintained (Patent Literature 7); that the physical properties of a protein fiber product is improved and the color of the protein fiber product is deepened by soaking the protein fiber product in an animal hair protein aqueous solution and a bridging agent (Patent Literature 8); and the like.
- a keratin hydrolytic product has the effect of suppressing deterioration of the texture and the light resistance of animal fibers caused by bleaching and/or dyeing.
- JP 2008 280326A JPH11 139940A and JPH07 300771A disclose the treatment of animal fibers with hydrolysed keratin.
- the present invention relates to providing a method for manufacturing processed fibers, the processed fibers, a method for suppressing damage to animal fibers, and a method for processing animal fibers, where a unique texture and light resistance of animal fibers are kept and a desired hue is realized even if the animal fibers are bleached and/or dyed.
- the inventors of the present invention found that when soaking animal fibers in a hydrolyzed keratin solution both before and after bleaching and /or dyeing the animal fibers, processed fibers with a desired hue are manufactured, while damage to the processed fibers is suppressed and while the original texture and light resistance of the animal fibers are kept.
- the method for manufacturing processed fibers, the method for suppressing damage to animal fibers, and the method for processing animal fibers, in accordance with the present invention fiber damage occurring when bleaching and/or dyeing animal fibers is suppressed or repaired, and processed fibers where strength and light resistance are kept without impairing a unique texture of the animal fibers are manufactured. Also, because a hydrolytic product obtained by hydrolyzing naturally derived keratin protein is used, the method according to the present invention is implemented substantially safely to the human body, the environment, and the like.
- a method for manufacturing processed fibers of the present invention is one of the methods for manufacturing processed fibers with bleaching and dyeing animal fibers and has the step for soaking the fibers in a hydrolyzed keratin solution.
- animal fibers are those animal fibers used in spinning, and the form thereof may be short fibers, long fibers, spun yarn, knitted fabric, woven fabric, felt, or the like, and may be apparel products such as clothes, bedding, cushions, and stuffed toys.
- the animal fibers include anyone of sheep wool, cashmere, mohair, camel hair, llama, alpaca, vicuna, angora, mink, and silk. These animal fibers may be used alone or in combination.
- the processed fibers of the present invention are obtained with bleaching and/or dyeing the above-described animal fibers, and thus, the processed fibers encompass bleached fibers, dyed fibers, and bleached and dyed fibers.
- bleaching is performed in order to increase the whiteness of animal fibers and is performed with an aqueous solution of hydrogen peroxide and a surface active agent, an aqueous solution of hydrogen peroxide and sodium silicate, or the like.
- dyeing is performed without particular limitation with a liquid flow method, a jigger method, a beam method, a cold pad-batch method, a pad steam method, a pad roll method, a continuous method, or the like, and a suitable dye and a suitable dyeing method may be selected in accordance with the type of animal fibers.
- the hydrolyzed keratins may be a hydrolytic product obtained by decomposing a keratin-containing raw material or a keratin extracted from the raw material under a reduction condition or the like by acid, alkali, peroxide, an enzyme, or the like, and the methods are all known (for example, see JP 2005-247692A , JP 2006-124341A , JP 2008-247925A , and JP H6-116300A ). Among them, hydrolytic products with an alkali or a peroxide are preferable.
- the number average molecular weight of this hydrolyzed keratin is 3,000 or less, and more preferably 2,000 or less, and is 150 or more, more preferably 200 or more, and more preferably 300 or more. Also, the number average molecular weight thereof is preferably 10,000 to 130, more preferably 8,000 to 130, more preferably 5,000 to 130, more preferably 3,000 to 150, more preferably 2,000 to 200, and more preferably 2,000 to 300.
- the keratin-containing raw material may be directly decomposed.
- an alkali metal hydroxide such as sodium hydroxide or potassium hydroxide, an alkaline earth metal hydroxides such as calcium hydroxide, or an alkaline compound such as ammonia is used, and in general, it is preferable to process the keratin-containing raw material at a concentration of 0.1 to 0.8 mol/L, preferably at a concentration of 0.2 to 0.5 mol/L, in general, at 20 to 120°C, and preferably for 0.1 to 72 hours.
- hydrolysis by a peroxide is performed by a peroxide such as hydrogen peroxide, performic acid, and peracetic acid, and in general, it is preferable to process the keratin-containing raw material at a concentration of 1 to 10%, preferably at a concentration of 3 to 8%, in general, at room temperature to 100°C, and preferably for 30 minutes to 48 hours.
- a peroxide such as hydrogen peroxide, performic acid, and peracetic acid
- this hydrolytic product may be cationized, silylated, acylated, or alkyl cationized so as to form derivatives of the hydrolyzed keratin, and these derivatives may also be used as long as the effects of the present invention are obtained.
- Examples of the keratin-containing raw materials include hairs of animals such as birds, sheep, horses, pigs, alpacas, mohair, angora, and cashmere, and among these, feathers obtained from birds or sheep wool is preferable, and it is particularly preferable to use feathers.
- any feathers such as down, feathers, and small feathers obtained from land birds such as chickens, quails, and turkeys, and water birds such as geese, call ducks, domestic ducks, European ducks, Peking ducks, and eider ducks, in particular, feathers of water birds are preferable.
- examples of preferred hydrolyzed keratins include hydrolyzed keratins derived from sheep wool or feathers, hydrolyzed keratins derived from feathers are more preferable, and alkaline hydrolyzed keratins or oxidatively hydrolyzed keratins derived from feathers are even more preferable.
- hydrolyzed keratin solutions examples include aqueous and ethanol solutions, and aqueous solutions are preferable.
- lipid components such as a chelating agent, a metal salt, ceramide, and fatty acid esters, an organic acid such as citric acid or ascorbic acid, a nonionic surface active agent, a cationic surface active agent, an amphoteric surface active agent, higher alcohols, lower alcohols, animal and vegetable oils, silicone oils, natural polysaccharides, an animal or plant extract, a hydrolytic product derived from animals or plants and derivatives thereof, a pH regulator, an antiseptic agent, and the like, in a range of not impairing the effects of the keratin.
- lipid components such as a chelating agent, a metal salt, ceramide, and fatty acid esters, an organic acid such as citric acid or ascorbic acid, a nonionic surface active agent, a cationic surface active agent, an amphoteric surface active agent, higher alcohols, lower alcohols, animal and vegetable oils, silicone oils, natural polysaccharides, an animal or plant extract, a hydrolytic product derived
- the concentration of the hydrolyzed keratin in the hydrolyzed keratin solution is preferably 0.01 mass% or more, more preferably 0.03 mass% or more, more preferably 0.05 mass% or more, and more preferably 0.1 mass% or more, and preferably 1 mass% or less, more preferably 0.7 mass% or less, and more preferably 0.5 mass% or less. Also, the concentration thereof is preferably 0.01 to 1 mass%, more preferably 0.03 to 0.7 mass%, more preferably 0.05 to 0.7 mass% or more, and more preferably 0.1 to 0.5 mass%.
- the animal fibers are preferably soaked in the hydrolyzed keratin solution, in general, at 30 to 80°C for 20 to 60 minutes, and are more preferably soaked at 40 to 60°C for 30 to 60 minutes.
- the animal fibers are soaked in the hydrolyzed keratin solution in a pretreatment process before a bleaching process and/or a dyeing process, and in a post-treatment process after a bleaching process and/or a dyeing process, and it is the invention to implement both the pretreatment process and the post-treatment process.
- a pretreatment process before a bleaching process and/or a dyeing process and in a post-treatment process after a bleaching process and/or a dyeing process, and it is the invention to implement both the pretreatment process and the post-treatment process.
- the processed fibers of the present invention obtained thus, fiber damage due to bleaching and/or dyeing is suppressed or repaired. Namely, in the processed fibers of the present invention, the unique texture (for example, the touch feel) of animal fibers is kept in spite of the bleaching and/or dyeing, and the strength and the light resistance are kept without being reduced.
- the method including the step for soaking these fibers in the hydrolyzed keratin solution serves as a method for suppressing damage to the fibers in bleaching and/or dyeing. Also, the method, including the step for soaking the animal fibers in the hydrolyzed keratin solution, before and after the fibers are bleached and/or dyed, serves as a processing method for suppressing the damage to the fibers.
- the present method for suppressing damage to animal fibers and the present method for processing animal fibers are useful for protecting the animal fibers from bleaching and/or dyeing and for keeping the texture of the fibers, or for keeping the light resistance.
- Texture refers to the material feel felt when a human touches the material, such as the touch feel, skin feel, or comfortability of feel
- unique texture of the animal fibers refers to the "tenderness”, “smoothness”, “frictional feel”, “suppleness”, “softness”, “sturdiness”, “bulkiness”, or the like.
- light resistance refers to resistance against deterioration caused by light (yellowing or decoloring of fibers).
- the dyeing (light resistant) fastness of fibers was measured in conformity with JIS L 0842, the third exposure method. The higher the grade obtained through this test is, the higher the fastness is.
- Fiber damage testing method Alkaline solubility method
- the alkaline solubility of fibers was measured in conformity with JIS L 1081, alkaline solubility method. The smaller the value obtained in this test is, the less damaged the fibers are.
- Fiber damage testing method Coloring method (methylene blue method)
- the fiber damage was determined in conformity with JIS L 1081, coloring (methylene blue method).
- the damage level is determined by means of a color concentration because undamaged fibers turn light blue and damaged fibers turn deep blue.
- ATR-FTIR spectra of fibers were recorded with a PerkinElmer Spectrum One FTIR spectrophotometer, manufactured by PerkinElmer, Inc., provided in Universal ATR Sampling Accessory. The measurements were performed under the condition that the accumulation number was 16 and the resolution was 4 cm -1 , and 4000 to 400 cm -1 .
- a peak for sulfonic acid was obtained at 1040 cm -1 .
- This sulfonic acid was derived from cystine in the fibers, and the cystine underwent cleavage due to excessive oxidation of cystine and changed into sulfonic acid.
- the lower the peak at 1040 cm -1 is, the lower the content of sulfonic acid is; indicating that excessive oxidation of fibers in the bleaching process has been suppressed.
- Panelist evaluation was performed on specimens. 25 panelists who had many opportunities to evaluate cashmere fibers in business touched specimens freely and evaluated the touch feel with the following criteria, and average values were obtained.
- SDS-PAGE electrophoresis was observed using a ready-made gel (e-PAGEL manufactured by ATTO CORPORATION). Dyeing was performed with Coomassie brilliant blue, and decoloring was performed with 10% acetic acid.
- Production Example 1 Production of sheep wool-derived solubilized (non hydrolytic) keratin
- Production Example 2 Production of feather-derived solubilized (non-hydrolytic) keratin
- the sheep wool was changed to feathers, and feather-derived solubilized keratin, not hydrolyzed, was produced similarly to Production Example 1.
- Production Example 3 Production of sheep wool-derived oxidatively hydrolyzed keratin
- Production Example 4 Production of feather-derived oxidatively hydrolyzed keratin
- the sheep wool was changed to feathers, and feather-derived oxidatively hydrolyzed keratin was produced similarly to Production Example 3.
- Production Example 5 Production of sheep wool-derived alkaline hydrolyzed keratin
- FIG. 1 shows the results of molecular weight analysis with SDS-PAGE for keratins produced in Production Examples 1 to 6.
- the feather and sheep wool-derived solubilized keratins bands with a molecular weight of 10,000 or more were confirmed, whereas with the feather and sheep wool-derived hydrolyzed keratins, only bands with a molecular weight of 10,000 or less were confirmed.
- the feather and sheep wool-derived solubilized keratins are found to contain keratin molecules with molecular weight of 10,000 or more, and the feather and sheep wool-derived hydrolyzed keratins contain only keratin molecules with a molecular weight of 10,000 or less.
- Pretreatment was performed by soaking cashmere hairs in an aqueous solution containing 0.1% feather-derived oxidatively hydrolyzed keratin produced in Production Example 4 at 40°C for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the hairs were soaked in a 0.1% hydrolyzed keratin solution, at a liquid ratio of 1: 20 at 40°C for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus feather-derived oxidatively hydrolyzed keratin treated hairs were produced.
- Feather-derived alkaline hydrolyzed keratin treated hairs were produced similarly to Embodiment 1, where the feather-derived oxidatively hydrolyzed keratin was changed to the feather-derived alkaline hydrolyzed keratin produced in Production Example 6.
- Sheep wool-derived oxidatively hydrolyzed keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived oxidatively hydrolyzed keratin produced in Production Example 3.
- Sheep wool-derived alkaline hydrolyzed keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived alkaline hydrolyzed keratin produced in Production Example 5.
- Sheep wool-derived solubilized keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the sheep wool-derived solubilized keratin produced in Production Example 1.
- Feather-derived solubilized keratin treated hairs were produced similarly to Embodiment 1, while the feather-derived oxidatively hydrolyzed keratin was changed to the feather-derived solubilized keratin produced in Production Example 2.
- FIG. 2 shows photographs of cotton-like fibers of Embodiments 1 to 4, Comparative Examples 1, 2, and unprocessed cashmere fibers, before and after the methylene blue dyeing.
- the fiber damage is evaluated on color concentrations because undamaged fibers turn light blue and damaged fibers turn deep blue.
- Comparative Examples 1, 2 were more deeply colored than unprocessed fibers.
- Embodiments 1 to 4 were more light in the color than Comparative Examples 1, 2, and in particular, Embodiments 1, 2 were equivalent to the unprocessed fibers in the color.
- Embodiments 1 to 4 suppressed, also according to the methylene blue dyeing method, fiber damage caused by bleaching, as is similar to the alkaline solubility method.
- the hydrolyzed keratins with an average molecular weight of 10,000 or less are found to effectively suppress fiber damage caused by bleaching.
- Pretreatment was performed by soaking cashmere hairs whose place of origin was different from those used in Embodiment 1 in a 0.5% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 30 minutes. Thereafter, the hairs were soaked in a diluted solution obtained of 35% hydrogen peroxide water with the concentration 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried to produce hydrolyzed keratin treated hairs.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 5, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 60 °C.
- Feather-derived alkaline hydrolyzed keratin treated hairs were produced similarly to Embodiment 5, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 80 °C.
- Cashmere hairs similar to those used in Embodiment 5 were soaked in a diluted solution of 35% hydrogen peroxide water in the concentration of 20 cc/L of, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried; thus keratin untreated hairs were produced.
- Embodiments 5 to 7 shows ATR-FTIR spectra of unprocessed hairs, Embodiments 5 to 7, and Comparative Example 3.
- the unprocessed hairs showed no peaks for sulfonic acid at 1040 cm -1
- Embodiments 5 to 7 and Comparative Example 3 showed peaks for sulfonic acid at 1040 cm -1 , and thus, it was suggested that excessive oxidation caused by bleaching occurred.
- Embodiments 5 to 7 had weaker peak intensities at 1040 cm -1 , and thus, it was suggested that excessive oxidation caused by bleaching was suppressed.
- Bleached cashmere hairs were soaked in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 60 minutes. After the hairs were sufficiently washed with water, they were dried, and hydrolyzed keratin treated hairs were produced.
- Embodiment 8 The fiber damages and light resistant fastnesses of Embodiment 8 and the bleached cashmere hairs are listed in Table 4.
- the fiber damage was smaller than the bleached cashmere hairs, suggesting the recovery from the fiber damage.
- the light resistant fastness increased, suggesting the increased light resistance.
- Table 4 Keratin Treatment Fiber Damage Light Resistant Fastness Processing Agent Temperatur e When Emb. 8 Feather-Derived Oxidatively Hydrolyzed Keratin 40°C After Bleaching 62.6 4 or mor e Un-processed - - - 71.2 3 to 4 *unprocessed: bleached cashmere hairs without keratin treatment
- Bleached and dyed cashmere hairs were soaked in a 0.5% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 60°C, for 20 minutes.
- the hairs were sufficiently washed with water and then dried, and thus, hydrolyzed keratin treated hairs were produced.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 7, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 80 °C.
- Embodiments 9 and 10 The light resistant fastness of Embodiments 9 and 10 and the bleached and dyed cashmere hairs are listed in Table 5. Embodiments 9 and 10 showed increased light resistant fastness.
- Table 5 Keratin Treatment Light Resistant Fastness Processed Temperature When Emb. 9 Feather-Derived Oxidatively Hydrolyzed Keratin 60°C After Bleaching and Dyeing 3 to 4 Emb. 10 80°C After Bleaching and Dyeing 3 to 4 Unprocessed - - - 3 *unprocessed: bleached and dyed cashmere hairs that did not undergo keratin treatment
- Pretreatment was performed by soaking cashmere hairs used in Embodiment 5, in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water having a concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the post-treatment was performed by soaking the hairs in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus hydrolyzed keratin pre/post-treated hairs. were produced.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 9, while the hairs were soaked in a feather-derived oxidatively hydrolyzed keratin solution, at 30 °C.
- FIG. 4 shows ATR-FTIR spectra of unprocessed hairs, Embodiments 11 and 12, and Comparative Example 3.
- the unprocessed hairs showed no peaks for sulfonic acid at 1040 cm -1
- Embodiments 11 and 12 and Comparative Example 3 showed peaks for sulfonic acid at 1040 cm -1 , and thus, excessive oxidation seems caused by bleaching.
- Embodiments 11 and 12 had weaker peak intensities at 1040 cm -1 , and thus, excessive oxidation caused by bleaching seems suppressed.
- Pretreatment was performed by soaking cashmere hairs used in Embodiment 5, in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 80°C, for 30 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, dyeing was performed by a 0.03% acidic dye.
- Hydrolyzed keratin treated hairs were produced similarly to Embodiment 13, while the hairs were soaked in a 0.5% feather-derived oxidatively hydrolyzed keratin solution.
- Cashmere hairs similar to Embodiment 5 were soaked in a diluted solution of 35% hydrogen peroxide water with the concentration of 20 cc/L, at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, dyeing was performed by a 0.03% acidic dye. The hairs were sufficiently washed with water and then dried, and thus, keratin untreated hairs were produced.
- Embodiments 13 and 14 and Comparative Example 4 are listed in Table 7. Compared with Comparative Example 4, in Embodiments 13 and 14, the fiber damages were small, revealing the suppression of the fiber damage. Also, the light resistant fastness increased, revealing the increase in the light resistance.
- Table 7 Keratin Treatment Fiber Damage Light Resistance Fastness Processing Agent Temperature When Emb. 13 Feather Oxidatively Hydrolyzed Keratin 80°C Before and After Bleaching and Dyeing 22.6 3 to 4 Emb. 14 80°C Before and After Bleaching and Dyeing 26.6 4 or more Com. Ex. 4 - - Before and After Bleaching and Dyeing 29.3 3
- Pretreatment was performed by soaking spun sheep wool yarn in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C. for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution), at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, post-treatment was performed by soaking the hairs in a 0.1% feather-derived oxidatively hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C, for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus, hydrolyzed keratin treated spun yarn was produced.
- the spun sheep wool yarn was soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution), at a liquid ratio of 1: 20, and was bleached at 60°C for 1 hour.
- the yarn was sufficiently washed with water and then dried, and thus bleached sheep wool spun yarn was produced.
- Hydrolyzed keratin treated cashmere spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to cashmere yarn.
- Hydrolyzed keratin treated silk spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to silk yarn.
- Hydrolyzed keratin treated alpaca spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to alpaca yarn.
- Hydrolyzed keratin treated mixed spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to mixed yarn of 80% mohair and 20% sheep wool.
- Hydrolyzed keratin treated angora spun yarn was produced similarly to Embodiment 15, while the sheep wool yarn was changed to angora yarn.
- Embodiments 16 to 20 Animal hairs similar to Embodiments 16 to 20 were soaked in a diluted solution of 35% hydrogen peroxide water(20 cc/L solution), at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. The hairs were sufficiently washed with water and then dried, and thus keratin untreated spun yarn was produced.
- Pretreatment was performed by soaking cashmere hairs in a 0.1% aqueous solution containing commercially available hydrolyzed keratin (average molecular weight of 1,000), at a temperature of 40°C, for 60 minutes. Thereafter, the hairs were soaked in a diluted solution of 35% hydrogen peroxide water (20 cc/L solution) at a liquid ratio of 1: 20, and were bleached at 60°C for 1 hour. After bleaching, the hairs were soaked in a 0.1% hydrolyzed keratin solution, at a liquid ratio of 1: 20, at 40°C for 60 minutes. The hairs were sufficiently washed with water and then dried, and thus hydrolyzed keratin treated hairs were produced.
- Hydrolyzed collagen treated hairs were produced similarly to Embodiment 21, while the hydrolyzed keratin was changed to a commercially available hydrolyzed collagen (average molecular weight of 1,000).
- Hydrolyzed silk treated hairs were produced similarly to Embodiment 21, while the hydrolyzed keratin was changed to commercially available hydrolyzed silk (average molecular weight of 1,000).
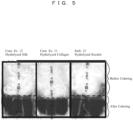
- FIG. 5 shows photographs of cotton-like fibers, before and after methylene blue dyeing on Embodiment 21, Comparative Examples 11 and 12.
- the fiber damage is evaluated with a color concentration because undamaged fibers turn light blue and damaged fibers turn deep blue.
- Comparative Examples 11 and 12 were more deeply colored than unprocessed fibers.
- Embodiment 21 was more lightly colored than Comparative Examples 11 and 12. This suggests that with Embodiment 21, in the evaluation with the methylene blue dyeing method as well, fiber damage caused by bleaching was suppressed similarly to the alkaline solubility method.
- the hydrolyzed keratin, among hydrolyzed proteins effectively suppressed fiber damage.
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
- Coloring (AREA)
Claims (7)
- Verfahren zur Herstellung von verarbeiteten Fasern durch Bleichen und/oder Färben von Tierfasern,
Durchführen eines Einweichschritts zum Einweichen der Tierfasern in einer Lösung aus hydrolysiertem Keratin, das ein zahlenmittleres Molekulargewicht von 150 bis 3000, gemessen durch Gelfiltrations-Molekulargewichtsanalyse, aufweist, wobei der Einweichschritt sowohl vor als auch nach dem Bleichen und/oder Färben durchgeführt wird, und wobei die Tierfasern eine oder mehrere ausgewählt aus der Gruppe von Schafwolle, Kaschmir, Mohair, Kamelhaar, Lama, Alpaka, Vikunja, Angora, Nerz und Seide sind. - Verfahren nach Anspruch 1, wobei die Lösung des hydrolysierten Keratins in dem Einweichschritt eine wässrige Lösung aus hydrolysiertem Keratin ist, die eine Konzentration von 0,01 bis 1 Masse-% aufweist.
- Verfahren nach Anspruch 1 oder Anspruch 2, wobei das hydrolysierte Keratin aus Federn abgeleitet ist.
- Verfahren nach einem der Ansprüche 1 bis 3, wobei das hydrolysierte Keratin ein zahlenmittleres Molekulargewicht von 300 bis 2000, gemessen durch Gelfiltrations-Molekulargewichtsanalyse, aufweist.
- Verfahren nach einem der Ansprüche 1 bis 4, wobei die Tierfasern gesponnenes Garn sind.
- Verarbeitete Faser, die nach dem Verfahren in einem der Ansprüche 1 bis 5 hergestellt wird.
- Verfahren nach einem der Ansprüche 1 bis 5, wobei Schäden an Tierfasern durch Bleichen und/oder Färben durch den Einweichschritt unterdrückt werden.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015171068 | 2015-08-31 | ||
| PCT/JP2016/075315 WO2017038814A1 (ja) | 2015-08-31 | 2016-08-30 | 加工繊維の製造方法及び当該加工繊維、動物繊維の損傷抑制方法、並びに動物繊維の加工方法 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP3346052A1 EP3346052A1 (de) | 2018-07-11 |
| EP3346052A4 EP3346052A4 (de) | 2019-05-01 |
| EP3346052B1 true EP3346052B1 (de) | 2025-06-18 |
Family
ID=58187543
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP16841846.5A Active EP3346052B1 (de) | 2015-08-31 | 2016-08-30 | Verfahren zur herstellung von verarbeiteter faser, verarbeitete faser, verfahren zur unterdrückung von schäden an tierfasern und verfahren zur verarbeitung von tierfaser |
Country Status (8)
| Country | Link |
|---|---|
| EP (1) | EP3346052B1 (de) |
| JP (1) | JP6587690B2 (de) |
| KR (1) | KR102129971B1 (de) |
| CN (1) | CN108026692A (de) |
| AU (1) | AU2016317514B2 (de) |
| MY (1) | MY193256A (de) |
| NZ (1) | NZ740282A (de) |
| WO (1) | WO2017038814A1 (de) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102279714B1 (ko) | 2017-05-15 | 2021-07-19 | 가부시키가이샤 시마세이키 세이사쿠쇼 | 표면가공섬유, 그 제조방법, 실 및 섬유제품 |
| US12018405B2 (en) * | 2018-03-22 | 2024-06-25 | Shima Seiki Mfg., Ltd. | Protein fiber crimping method, protein fiber production method, protein fibers, spun yarn, and textile product |
| US20220071355A1 (en) * | 2018-12-21 | 2022-03-10 | Ecco Sko A/S | Method of dyeing collagen staple fiber |
| CN110565357A (zh) * | 2019-06-11 | 2019-12-13 | 邵阳阳光发品有限公司 | 一种络合催化人发纤维漂白的方法 |
| CN110453522B (zh) * | 2019-06-20 | 2022-02-18 | 天津科技大学 | 一种100%天然纤维制造无纺墙纸原纸的方法 |
| JP7473955B2 (ja) * | 2020-04-03 | 2024-04-24 | 西川株式会社 | 獣毛製品製造方法 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01280074A (ja) | 1988-05-02 | 1989-11-10 | Daito Boshoku Kk | 布地およびこの布地の製造方法 |
| JPH02242970A (ja) | 1988-08-04 | 1990-09-27 | Honda Motor Co Ltd | 羊毛繊維の耐光耐熱性向上法 |
| JPH05156573A (ja) | 1991-03-11 | 1993-06-22 | Kanebo Ltd | 超耐光性獣毛系繊維品およびその製法 |
| JP3265329B2 (ja) * | 1993-05-31 | 2002-03-11 | 中日本繊維工業協同組合 | 蛋白繊維製品の加工方法 |
| JP3256748B2 (ja) * | 1993-06-30 | 2002-02-12 | ライオン株式会社 | 衣料用液体柔軟剤組成物 |
| JPH07300771A (ja) * | 1994-04-21 | 1995-11-14 | Kiyoujiyuu:Kk | 形状記憶ウール及びその製造方法 |
| DE69816135T2 (de) | 1997-03-25 | 2004-04-22 | Ciba Speciality Chemicals Holding Inc. | 4,4'-Diaminostilben-2,2'-disulfonsäurederivate |
| JPH11172580A (ja) | 1997-08-21 | 1999-06-29 | Nippon Chem Ind Co Ltd | 獣毛繊維処理剤、処理浴および処理方法 |
| JPH11139940A (ja) * | 1997-10-31 | 1999-05-25 | Seiwa Kasei Kk | 染毛用前処理剤 |
| JP4157338B2 (ja) | 2002-07-13 | 2008-10-01 | 敏夫 萩原 | 繊維材料の改質加工法 |
| JP2005146442A (ja) | 2003-11-12 | 2005-06-09 | Nicca Chemical Co Ltd | 羊毛系繊維用漂白助剤及び羊毛系繊維の漂白方法 |
| JP4931010B2 (ja) * | 2007-04-13 | 2012-05-16 | 東洋羽毛工業株式会社 | 毛髪処理方法 |
| CN103194919A (zh) * | 2013-04-24 | 2013-07-10 | 太仓市其盛化纤厂 | 一种有利于提高羊毛面料色彩饱和度的印花方法 |
| CN103735428B (zh) * | 2013-12-31 | 2015-04-08 | 广州市白云区大荣精细化工有限公司 | 一种角蛋白护发素及其制备方法 |
-
2016
- 2016-08-30 EP EP16841846.5A patent/EP3346052B1/de active Active
- 2016-08-30 AU AU2016317514A patent/AU2016317514B2/en active Active
- 2016-08-30 KR KR1020187009161A patent/KR102129971B1/ko active Active
- 2016-08-30 MY MYPI2018700748A patent/MY193256A/en unknown
- 2016-08-30 CN CN201680050316.9A patent/CN108026692A/zh active Pending
- 2016-08-30 JP JP2017538044A patent/JP6587690B2/ja active Active
- 2016-08-30 WO PCT/JP2016/075315 patent/WO2017038814A1/ja not_active Ceased
- 2016-08-30 NZ NZ740282A patent/NZ740282A/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| KR102129971B1 (ko) | 2020-07-03 |
| MY193256A (en) | 2022-09-28 |
| JPWO2017038814A1 (ja) | 2018-08-16 |
| KR20180042429A (ko) | 2018-04-25 |
| JP6587690B2 (ja) | 2019-10-09 |
| AU2016317514A1 (en) | 2018-03-22 |
| WO2017038814A1 (ja) | 2017-03-09 |
| EP3346052A1 (de) | 2018-07-11 |
| CN108026692A (zh) | 2018-05-11 |
| EP3346052A4 (de) | 2019-05-01 |
| AU2016317514B2 (en) | 2019-07-25 |
| NZ740282A (en) | 2019-07-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3346052B1 (de) | Verfahren zur herstellung von verarbeiteter faser, verarbeitete faser, verfahren zur unterdrückung von schäden an tierfasern und verfahren zur verarbeitung von tierfaser | |
| CN102560696B (zh) | 一种天然彩色桑蚕丝绵的生产方法 | |
| CN109537279A (zh) | 一种玻尿酸丝绸面料及其制作方法 | |
| CN108385382A (zh) | 一种抗菌防臭抗皱整理剂及其应用 | |
| JP2010132595A (ja) | 毛髪の保護、損傷防止、及び修復効果を有する毛髪用処理剤 | |
| CN103866575A (zh) | 一种改性蚕丝纤维的制备方法及其产品 | |
| US20150107616A1 (en) | Methods for processing keratinous fibers, and uses thereof | |
| Pekhtasheva et al. | Biodegradation and biodeterioration of some natural polymers | |
| JPH0610272A (ja) | 織物仕上加工剤 | |
| CN105421019B (zh) | 一种人发纤维漂白工艺 | |
| KR101207490B1 (ko) | 케라티네이즈를 포함하는 모발 보호 및 수복용 모발 화장품 조성물 | |
| JPH03213574A (ja) | 獣毛繊維の改質方法 | |
| CN106087432A (zh) | 一种棉纤维的柔化处理工艺 | |
| CN103494354A (zh) | 珍珠纤维和铜氨纤维混纺的面料 | |
| JP3895217B2 (ja) | 繊維製品およびその製造方法 | |
| KR102185434B1 (ko) | 효소처리 과정을 포함하는 친환경 삼베 섬유 제조 방법 | |
| JP3459367B2 (ja) | パーマネントウェーブ用剤、羊毛改質剤、羊毛仕上剤、パーマネントウェーブ用エンドペーパー及びパーマネントウェーブの処理方法 | |
| CN105671953A (zh) | 一种纯棉袜子的抗菌整理工艺 | |
| Pekhtasheva et al. | Biodegradation mechanism of some polymers | |
| Pekhtasheva et al. | Biodegradation and biodeterioration of some natural polymers | |
| KR102609988B1 (ko) | 콜라겐을 이용한 기능성 원단의 제조방법 및 이에 의해제조된 기능성 원단 | |
| CN110055771A (zh) | 一种纺织材料的漂白和改性方法和用途 | |
| JP7709187B2 (ja) | 羊毛繊維の黄変防止処理、鮮明染色処理、及び耐洗濯性の付与方法 | |
| CN106491490A (zh) | 一种抗菌消炎的丝素蛋白复合水凝胶及其制备方法 | |
| CN107675509A (zh) | 一种抗静电抗菌毛领 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20180323 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| DAV | Request for validation of the european patent (deleted) | ||
| DAX | Request for extension of the european patent (deleted) | ||
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20190403 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: D06M 101/12 20060101ALI20190328BHEP Ipc: D06L 4/12 20170101ALI20190328BHEP Ipc: D06M 15/15 20060101AFI20190328BHEP Ipc: D06P 1/46 20060101ALI20190328BHEP Ipc: D06L 4/00 20170101ALI20190328BHEP |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| 17Q | First examination report despatched |
Effective date: 20220929 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20250324 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602016092607 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG9D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250919 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250918 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20250922 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20250923 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250918 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20251020 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 1804260 Country of ref document: AT Kind code of ref document: T Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20251018 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602016092607 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: H13 Free format text: ST27 STATUS EVENT CODE: U-0-0-H10-H13 (AS PROVIDED BY THE NATIONAL OFFICE) Effective date: 20260324 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250618 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20250830 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |