EP1113070A2 - MGDA-haltige Maschinengeschirrspülmittel niederer Alkalität - Google Patents
MGDA-haltige Maschinengeschirrspülmittel niederer Alkalität Download PDFInfo
- Publication number
- EP1113070A2 EP1113070A2 EP01105118A EP01105118A EP1113070A2 EP 1113070 A2 EP1113070 A2 EP 1113070A2 EP 01105118 A EP01105118 A EP 01105118A EP 01105118 A EP01105118 A EP 01105118A EP 1113070 A2 EP1113070 A2 EP 1113070A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- mgda
- acid
- detergent
- salts
- residues
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/26—Organic compounds containing nitrogen
- C11D3/33—Amino carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3947—Liquid compositions
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D7/00—Compositions of detergents based essentially on non-surface-active compounds
- C11D7/22—Organic compounds
- C11D7/32—Organic compounds containing nitrogen
- C11D7/3245—Aminoacids
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D7/00—Compositions of detergents based essentially on non-surface-active compounds
- C11D7/22—Organic compounds
- C11D7/32—Organic compounds containing nitrogen
- C11D7/3281—Heterocyclic compounds
Definitions
- the invention relates to machine dishwashing detergents Alkalinity, methylglycinediacetic acid (MGDA) or a salt of which contain and which preferably have a reduced content of Have bleach.
- MGDA methylglycinediacetic acid
- US-A 3 673 098 discloses caustic compositions without Bleaching agents based on KOH and nitrilotriacetic acid (NTA) based.
- Fr-A 1 590 416 also discloses caustic compositions without bleach based on alkali hydroxides, Pyrophosphates and NTA based.
- EP-A-530 635 discloses acidic to mildly alkaline Compositions (pH 5-9) that do not contain complexing agents contain and on builders, enzymes and optionally Oxygen bleaching is based.
- DE-A-3 833 047 discloses acidic compositions (pH 2-6) with hydrolases and builders or complexing agents.
- the Examples disclose high NTA or EDTA share (up to 30%). While these compositions can e.g. Eliminate tea residues well, there is milk residue their effect, measured on a scale from 0 to 10, however always under 5.
- biodegradable complexing agents such as ⁇ -alanine diacetic acid ( ⁇ -ADA) and iso-serine diacetic acid (ISDA), in detergents is in the DE-A-3 829 847 and DE-A-4 036 995. If those Publications Application examples for dishwasher detergents contained, it is machine dishwashing detergent, which in about 0.5% aqueous solution has a pH of more than 10.5.
- DE-A-4 319 935 describes the use of MGDA in Dishwasher detergents disclosed without an example specify.
- MGDA methylglycinediacetic acid
- MGDA is supposed to be a better one, especially for blood-soiled laundry Have effect, as phosphonates and with tea soiled Laundry have about the same effect as phosphonates.
- the same publication discloses that MGDA in a dishwashing detergent, similar to NTA, a very good one Deposit inhibition on glass, metal and porcelain shows, whereby a dishwashing detergent is disclosed which in solution provides pH of more than 10.5. It will too suggested MGDA in very strongly alkaline conditions to use for dairy cleaning.
- a dishwashing detergent excellent cleaning performance for dishes have both milk and milk Tea residue is contaminated.
- This task is accomplished by a Dishwasher detergent dissolved, the methylglycinediacetic acid or their salts as complexing agents and is characterized in that it is approximately 0.5% aqueous solution does not exceed a pH of 10.4.
- Milk residues consist of a mixture of denatured proteins (casein), fats, sugars and mineral deposits and consist of around 70% calcium phosphate (Ca 3 (PO 4 ) 2 ).
- Complexing agents therefore have a significant influence on the removal of milk residues because they dissolve calcium phosphates by complexing the calcium ions.
- Tea residues are also based on Ca deposits, that arise when brewing tea with hard water.
- tea residues are usually different Requirements for a dishwasher detergent as Milk residues, which can be seen from the fact that the detergent disclosed in DE-A 3 833 047 in the acidic range (pH 2-6) Tea residues removed satisfactorily, at Milk residues cannot convince.
- the ability (based on the weight of the complexing agent) to complex calcium ions at a certain pH value depends on the stability constant of the calcium complex (pK c ), the acidic character of the complexing agent (pK 3 ) and its molecular weight.
- the theoretical is at pH 10.4 Degree of complexation for calcium phosphates when using NTA at 360 mg / g and when using MGDA at 340 mg / g, and at pH 8 the theoretical values are 510 mg / g for NTA and 490 mg / g for MGDA.
- MGDA was only used in dishwashing detergents used in about 0.5% aqueous solution have a pH of more than 10.4.
- Dishwashing detergents that contain MGDA and one of them 0.5% aqueous solution with a pH of up to 10.4 deliver better both tea residues and milk residues can remove and overall a better washing result have as dishwashing detergent, which instead of MGDA another Contain complexing agents such as the common NTA.
- the sufficient Amount of complexing agent MGDA has the amount of Oxygen bleach can be reduced without changing thereby the flushing performance and in particular the ability Remove milk residues and tea residues, worsened. This surprising finding even opens up the possibility of using a sufficiently large amount of complexing agents completely on the use of To avoid oxygen bleach.
- the dishwashing detergent according to the invention can be liquid or be powdery and also in the form of a single layer or multi-layer tablet. Appropriate Dosage forms and methods of obtaining them are the Known specialist.
- the dishwasher detergents according to the invention preferably contain one or more enzymes such as Amylase, protease, lipase or cellulase. Through the combined application of amylase and protease were the best cleaning results.
- the ones here are particularly suitable for the pH ranges described use effectively; for example, the temperature stable amylase Termamyl (Novo) at pH 7-8 das Optimal of your spectrum of activity.
- the enzymes come in Granulate form or in solution with commercially available Concentrations or activities are used, the Enzyme content is 0.5-6, preferably 1-4% by weight can.
- Suitable builders are homo- and copolymers Polycarboxylic acids and their partially or completely neutralized salts, monomeric polycarboxylic acids and Hydroxycarboxylic acids and their salts, carbonates, bicarbonates, Borates, phosphates, silicates, aluminosilicates and mixtures such substances.
- Preferred salts of the above compounds are the ammonium and / or alkali salts, i.e. the lithium, Sodium, potassium and rubidium salts and particularly preferred the sodium salt used.
- Suitable polycarboxylic acids are.acyclic, alicyclic, heterocyclic and aromatic carboxylic acids, these contain at least two carboxy groups, each through preferably no more than two carbon atoms from each other are separated.
- Polycarboxylates containing two carboxy groups include for example water-soluble salts of succinic acid, Malonic acid, (ethylenedioxy) diacetic acid, maleic acid, Diglycolic acid, tartaric acid, tartronic acid and fumaric acid.
- Polycarboxylates containing three carboxy groups include for example, water-soluble citrates.
- Corresponding is suitable as a hydroxycarboxylic acid, for example Citric acid.
- one of aspartic acid can also be used HOOC-CH (NH 2 ) -CH 2 -COOH derived polymer with monomer units of the formula to be named.
- the homopolymer of is also suitable as the polycarboxylic acid Acrylic acid.
- borate builders can also such builders are used, the borates among the Detergent storage conditions or rinsing conditions release.
- Suitable phosphates are polyphosphates such as tripolyphosphate, Pyrophosphate, orthophosphate and the polymeric metaphosphate.
- polyphosphates such as tripolyphosphate, Pyrophosphate, orthophosphate and the polymeric metaphosphate.
- Suitable silicates are sodium silicates such as sodium disilicate, Sodium metasilicate and crystalline layered silicates. Sodium aluminosilicates are also suitable (Zeolite).
- a builder system from the salt of one is particularly preferred Hydroxycarboxylic acid or from the mixture of one Hydroxycarboxylic acid and the salt of a hydroxycarboxylic acid.
- Both the hydroxycarboxylic acid and the salt of Hydroxycarboxylic acid can be completely or partially Tripolyphosphate can be replaced, although this is not preferred becomes.
- the builder system preferably consists of a Hydroxypolycarboxylic acid with 2-4 carboxyl groups (or acidic inorganic salts), which are used to adjust the pH value with their Salt can be mixed.
- Citric acid is preferred or a mixture of sodium citrate with citric acid used.
- the required pH value is suitable depending on the others Components of the mixture e.g. Mix with one predominant proportion of citric acid.
- the water content of liquid dishwashing detergent can be 40-80 % By weight.
- the liquid dishwashing detergents also contain 0.2-5.0% by weight, preferably 0.5-2% by weight, of a thickener.
- a thickener preferably 0.5-2% by weight
- Carboxyvinyl polymers such as those marketed by BF Goodrich under the production name "Carbopol”, proved to be very useful.
- Inorganic compounds which have thickening properties in this product composition can also be used. Examples of this are natural or synthetic thickeners based on differently structured silicate structures. Their use is described in various patents, for example in US-A-4 933 101 or EP-A2-0 407 187.
- nonionic surfactants are suitable as surfactants. These include, for example, water-soluble ethoxylated C 6-16 fatty alcohols and C 6-16 mixed ethoxylated / propoxylated fatty alcohols and mixtures thereof, and also polyglucosides.
- Another class of non-ionic surfactants includes polyhydroxy fatty acid amides.
- the dishwasher detergent according to the invention can further contain one or more foam control agents. All those used in this area are suitable for this Foam control agents such as silicones and Paraffin oil.
- the foam control agents are in the invention Machine dishwashing detergent preferably with less than 5% by weight of the total weight of the detergent.
- the machine dishwashing detergents according to the invention can be used as Bleaching agents prefer chlorine-free, active oxygen releasing bleaches such as inorganic perhydrates or contain organic peracids and their salts.
- inorganic perhydrates are perborates, Percarbonates and persulfates such as peroxymonopersulfate.
- the inorganic perhydrates are usually alkali metal salts, such as lithium, sodium, potassium or rubidium salts, especially the sodium salts.
- the inorganic perhydrates can in the detergent as crystalline solids without protection. For certain perhydrates however, it is advantageous to use them as granular To use compositions with a coating are provided, the better the granular products Gives storage stability.
- the preferred perborate is sodium perborate, which can be present as a monohydrate with the formula NaBO 2 H 2 O 2 or as a tetrahydrate with the formula NaBO 2 H 2 O 2 .3H 2 O.
- the preferred percarbonate is sodium percarbonate with the formula 2Na 2 CO 3 3H 2 O 2 .
- the percarbonate is preferably used in a coated form to increase its stability.
- Organic peracids all conventionally include bleaching agents used organic peracids, including for example perbenzoic acid and peroxycarboxylic acids such as Mono- or diperoxyphthalic acid, 2-octydiperoxysuccinic acid, Diperoxydodecanedicarboxylic acid, Diperoxyazelaic acid and Imidoperoxycarboxylic acid and optionally its salts.
- perbenzoic acid and peroxycarboxylic acids such as Mono- or diperoxyphthalic acid, 2-octydiperoxysuccinic acid, Diperoxydodecanedicarboxylic acid, Diperoxyazelaic acid and Imidoperoxycarboxylic acid and optionally its salts.
- the dishwasher detergent according to the invention can continue to contain one or more bleach activators. These are preferred in detergents for dishwashing operations at temperatures below 60 ° C used to achieve a sufficient bleaching effect.
- N- and O-acyl compounds are particularly suitable such as acylated amines, acylated glycolurils or acylated sugar compounds.
- TAED N, N ', N'-tetraacetylethylenediamine
- PAG pentaacetylglucose
- TAGU tetraacetylglycoluril
- Dishwasher detergent is an oxygen bleach an amount of less than 1% by weight, based on the active Oxygen on. The amount of bleach activator used will be adjusted accordingly.
- the machine dishwashing detergent s a large percentage of MGDA, like 25% or more, preferably 30% or more, in each case based on the free acid, and an oxygen bleach is not used.
- the dishwasher detergent according to the invention can also contain a silver / copper anti-corrosion agent.
- This term includes funds that start up Non-ferrous metals, especially silver and copper, prevent or mitigate.
- Preferred silver / copper anti-corrosion agents are benzotriazole or bis-benzotriazoles as well as their substituted derivatives.
- Suitable agents are organic and / or inorganic redox-active substances and paraffin oil.
- Benzotriazole derivatives are those compounds in which the available substitution sites of the aromatic ring are partially or completely substituted.
- Linear or branched chain C 1-20 alkyl groups as well as hydroxy, thio, phenyl or halogen such as fluorine, chlorine, bromine and iodine are suitable as substituents.
- Preferred substituted benzotriazole is tolyltriazole.
- Suitable bis-benzotriazoles are those in which the benzotriazole groups are each connected in the 6-position by a group X, in which X is a bond, a straight-chain alkylene group which is optionally substituted by one or more C 1-4 alkyl groups and preferably has 1-6 carbon atoms , a cycloalkyl radical having at least 5 carbon atoms, a carbonyl group, a sulfuryl group, an oxygen or a sulfur atom.
- the aromatic rings of the bis-benzotriazoles can be substituted as defined above for the benzotriazole.
- Suitable organic redox-active substances are, for example, ascorbic acid, indole, methionine, an N-mono- (C 1 -C 4 alkyl) glycine, an N, N-di (C 1 -C 4 alkyl) glycine, 2- phenylglycine or a coupler and / or developer compound chosen from the group of diaminopyridines, aminohydroxypyridines, dihydroxypyridines, heterocyclic hydrazones, aminohydroxypyrimidines, dihydroxypyrimidines, tetraaminopyrimidines, triaminohydroxypyrimidines, Diaminodihydroxypyrimidine, Dihydroxynaphtaline, naphthols, pyrazolones, hydroxyquinolines, aminoquinolines, primary aromatic amines, which in ortho, meta or para position have a further free or with C 1 -C 4 alkyl or C 2 -C 4 hydroxyalkyl groups substituted hydroxy or amino
- Suitable inorganic redox-active substances are for example, metal salts and / or metal complexes selected from the group of manganese, titanium, zirconium, hafnium, Vanadium, cobalt and cerium salts and / or complexes, wherein the metals in one of the oxidation states II, III, IV, V or VI are available.
- Organic and inorganic redox-active substances which as Silver / copper anti-corrosion agents are suitable also mentioned in WO 94/26860 and WO 94/26859, on the Content hereby referred.
- Suitable paraffin oil are predominantly branched aliphatic hydrocarbons with a number of carbon atoms in the range from 20 to 50.
- the paraffin oil is preferably selected from predominantly branched chain C 25-45 species with a ratio of cyclic to noncyclic hydrocarbons of 1:10 to 2: 1 , preferably from 1: 5 to 1: 1.
- a silver / copper anticorrosive in the machine dishwashing detergent according to the invention is present, it is preferably 0.01 to 5% by weight, particularly preferably with 0.1 to 2 wt .-% of the total weight.
- the proportion of MGDA on the dishwasher detergent is high enough, such as 25% or more, preferably 30% or more, each based on is the free acid, so that there is no bleaching agent can also be used on a silver / copper anti-corrosion agent to be dispensed with.
- the machine dishwashing detergents according to the invention are not caustic and therefore do not require a specially secured one Packaging. A good washing result can be achieved with one relatively low use of 15-20 g powder or 20-50 ml of liquid detergent can be achieved. All ingredients are according to current knowledge, neither toxic nor otherwise harmful to health.
- the cleaning agents described here therefore also represent an environmentally and consumer-friendly Alternative to conventional cleaners with Complexing agents in the strongly basic area work.
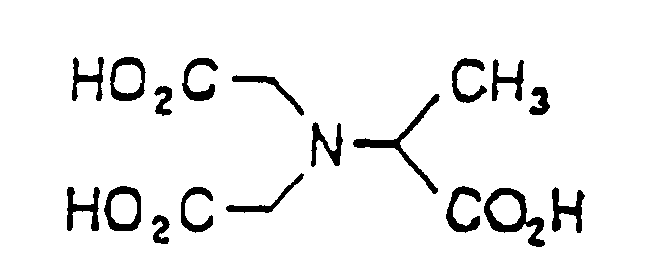
- the methylglycinediacetic acid used according to the invention has the following structural formula:
- Methylglycinediacetic acid is particularly preferred as free acid or used as sodium or potassium salt and is preferably in an amount of 3% to 90%, based on the free acid, contained in the detergent.
- the amount of MGDA is preferably at least 5%, based on the free acid.
- an approximately 0.5% aqueous solution of the detergent has a pH of no more than 10.4.
- the pH is preferably about 0.5% aqueous solution of the detergent according to the invention more than 6 to 10.4, particularly preferably from 7 to 10.0.
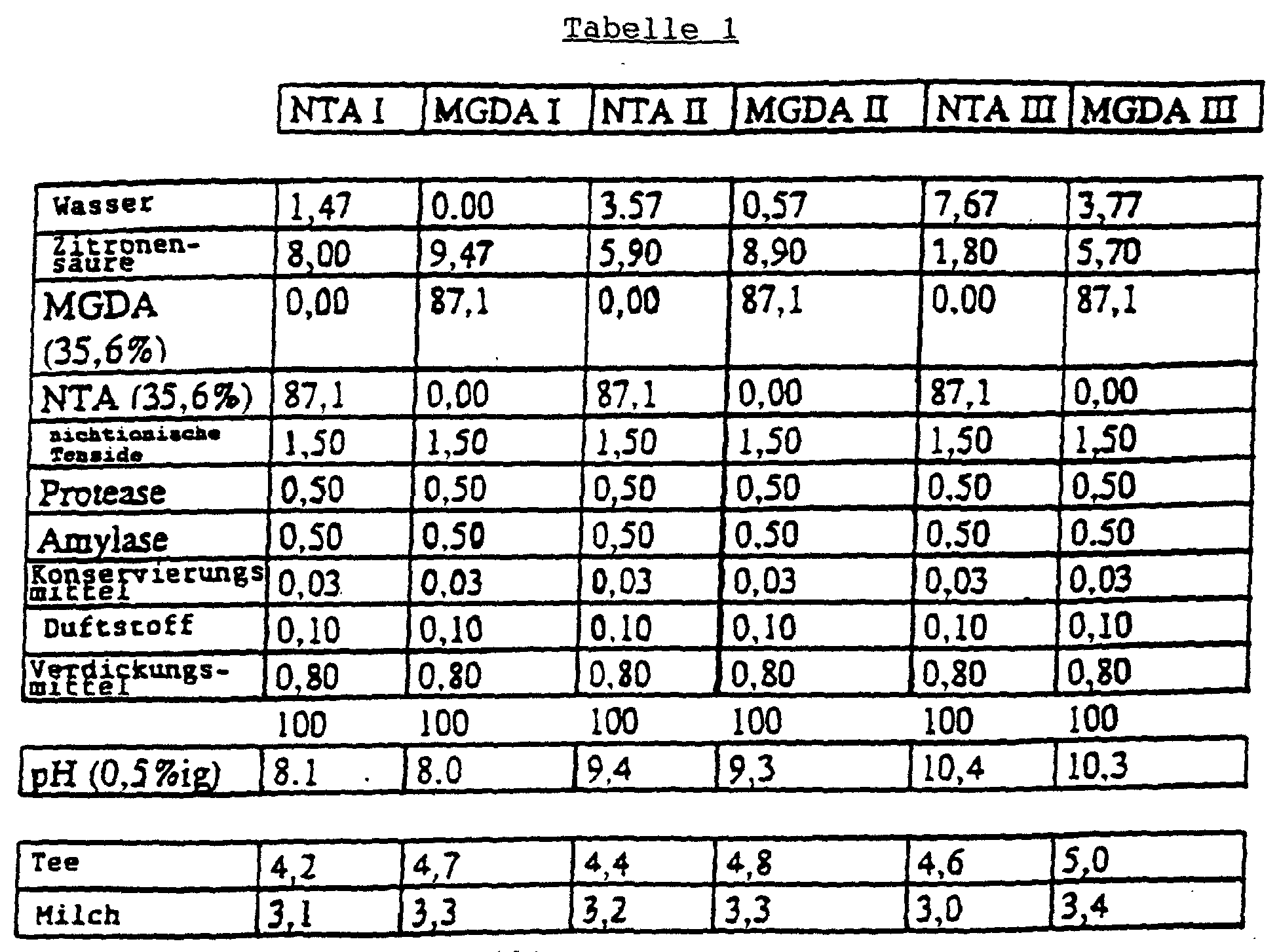
- MGDA is not commercially available in its pure form and therefore a 77% solution in water was used. A larger amount of the commercially available product had to be used, as shown in the table. Two values are given in the table for MGDA. The first value indicates the amount in g of the commercial product, and the second value corresponds to the corresponding amount of pure substance (100%). S ⁇ FW Journal 122, 1996, 392-397 Ex. 1 Ex. 2 Quantities in g. (%) Ex.
- the following example shows that when replacing part of the builder of a dishwasher detergent with methylglycinediacetic acid at low pH values excellent washing performance can be achieved and especially contrary to theoretical expectations Ability to remove milk residues and tea residues higher than other common complexing agents such as NTA.
- the example illustrates the invention.
- Liquid dishwashing detergents with a Composition as shown in Table 1 is made (both according to the invention using MGDA and using the NTA used in the prior art), and their washing performance was measured using dishes that both Contained milk and tea residues, determined. In this Test was essentially the DIN standard No. 44990, part 2 followed. It became a Bosch SMS 5062 dishwasher used. The water had a hardness of 19 ° dH (3.39 mmol Calcium carbonate), the temperature of the water was 65 ° C and 20 ml of detergent were used in each case. The results The flushing performance was assessed on a scale of 1-5 (5 for 100% clean dishes are rated) and are also listed in Table 1.
- the non-ionic surfactant is a common, low-foaming ethoxylated and propoxylated fatty alcohol. Common proteases and amylases are used.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
Description
| SÖFW-Journal 122, 1996, 392-397 | |||
| Beisp. 1 | Beisp. 2 Mengen in g. (%) | Beisp. 3 | |
| Citrat | 2 (50%) | 1,6 (40%) | 1 (25%) |
| Carbonat | 1 (25%) | 1 (25%) | 1 (25%) |
| Hydrogen- | |||
| carbonat | 0,36 (9%) | 0,36 (9%) | 0,36 (9%) |
| Perborat | 0,28 (7%) | 0,28 (7%) | 0,28 (7%) |
| TAED | 0,08 (2%) | 0,08 (2%) | 0,08 (2%) |
| nichtionisches | 0,08 (2%) | 0,08 (2%) | 0,08 (2%) |
| oberflächenaktives | |||
| Mittel | |||
| MGDA | 0,26/0,2 (5%) | 0,78/0,6 (15%) | 1,56/1,2 (30%) |
| (77% Lös.)/ | |||
| (100%) | |||
| Gesamt | 4,06 4 | 4,18 4 | 4,36 4 |
| pH | 10,64 | 10,79 | 10,82 |
Claims (4)
- Maschinengeschirrspülmittel, enthaltend Methylglycindiessigsäure oder deren Salze als Komplexierungsmittel, dadurch gekennzeichnet, daß das Mittel in etwa 0,5%-iger wäßriger Lösung einen pH-Wert von 10,4 nicht übersteigt.
- Maschinengeschirrspülmittel nach Anspruch 1, dadurch gekennzeichnet, daß es ein Sauerstoffbleichmittel in einer Menge von weniger als 1% bezogen auf das Gewicht aktiven Sauerstoffs enthält.
- Maschinengeschirrspülmittel nach Anspruch 1 oder 2, dadurch gekennzeichnet, daß das Mittel in etwa 0,5%-iger wäßriger Lösung einen pH-Wert von 6 bis 10,4 aufweist.
- Verwendung von Methylglycindiessigsäure oder deren Salze als vollständiger oder teilweiser Ersatz für Builder oder Komplexierungsmittel in Maschinengeschirrspülmitteln mit einem pH-Wert von nicht mehr als 10,4 in etwa 0,5%-iger wäßriger Lösung zur Verbesserung der Fähigkeit sowohl Milchrückstände als auch Teerückstände zu beseitigen.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19624839 | 1996-06-21 | ||
| DE19624839 | 1996-06-21 | ||
| EP97928266A EP0906407B1 (de) | 1996-06-21 | 1997-06-20 | Mgda-haltige maschinengeschirrspülmittel niederer alkalität |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP97928266A Division EP0906407B1 (de) | 1996-06-21 | 1997-06-20 | Mgda-haltige maschinengeschirrspülmittel niederer alkalität |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1113070A2 true EP1113070A2 (de) | 2001-07-04 |
| EP1113070A3 EP1113070A3 (de) | 2002-01-02 |
| EP1113070B1 EP1113070B1 (de) | 2008-03-19 |
Family
ID=7797614
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP97928266A Revoked EP0906407B1 (de) | 1996-06-21 | 1997-06-20 | Mgda-haltige maschinengeschirrspülmittel niederer alkalität |
| EP01105118A Revoked EP1113070B1 (de) | 1996-06-21 | 1997-06-20 | MGDA-haltige Maschinengeschirrspülmittel niederer Alkalität |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP97928266A Revoked EP0906407B1 (de) | 1996-06-21 | 1997-06-20 | Mgda-haltige maschinengeschirrspülmittel niederer alkalität |
Country Status (6)
| Country | Link |
|---|---|
| EP (2) | EP0906407B1 (de) |
| AT (2) | ATE205250T1 (de) |
| CA (1) | CA2258218A1 (de) |
| DE (2) | DE59712930D1 (de) |
| ES (2) | ES2160355T3 (de) |
| WO (1) | WO1997049792A1 (de) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102007006627A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007006629A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007006628A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007006630A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007044417A1 (de) | 2007-09-17 | 2009-03-19 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102008060470A1 (de) | 2008-12-05 | 2010-06-10 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102008063801A1 (de) | 2008-12-19 | 2010-06-24 | Henkel Ag & Co. Kgaa | Maschinelles Geschirrspülmittel |
| WO2010149695A1 (de) * | 2009-06-24 | 2010-12-29 | Henkel Ag & Co. Kgaa | Maschinelles reingungsmittel |
| WO2010149697A1 (de) * | 2009-06-24 | 2010-12-29 | Henkel Ag & Co. Kgaa | Maschinelles reinigungsmittel |
| WO2011032868A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschinelles geschirrspülmittel |
| DE102009029635A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschinelles Geschirrspülmittel |
| DE102009029636A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschine Geschirrspülmittel |
| WO2011051419A1 (de) | 2009-10-30 | 2011-05-05 | Henkel Ag & Co. Kgaa | Maschinelles geschirrspülmittel |
| DE102013019269A1 (de) | 2013-11-15 | 2015-06-03 | Weylchem Switzerland Ag | Geschirrspülmittel sowie dessen Verwendung |
| US9441189B2 (en) | 2005-11-07 | 2016-09-13 | Reckitt Benckiser Finish B.V. | Composition |
| WO2018007298A1 (de) | 2016-07-05 | 2018-01-11 | Henkel Ag & Co. Kgaa | Geschirrspülmittel enthaltend zuckersäure und aminocarbonsäure |
| EP2520641B1 (de) | 2005-06-30 | 2019-03-13 | The Procter & Gamble Company | Phosphatarme Maschinengeschirrspülmittel |
| DE102024205394A1 (de) * | 2024-06-12 | 2025-12-18 | BSH Hausgeräte GmbH | Geschirrspülzusammensetzung zum Reinigen von Geschirr, Verfahren zum Spülen von Geschirr und Geschirrspülmaschine |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2311537A (en) * | 1996-03-29 | 1997-10-01 | Procter & Gamble | Rinse composition for dishwashers |
| PL1721962T3 (pl) * | 2005-05-11 | 2009-01-30 | Unilever Nv | Kompozycja do mycia naczyń oraz sposób zmywania naczyń |
| DE102005041347A1 (de) † | 2005-08-31 | 2007-03-01 | Basf Ag | Reinigungsformulierungen für die maschinelle Geschirrreinigung enthaltend hydrophil modifizierte Polycarboxylate |
| GB0611206D0 (en) | 2006-06-07 | 2006-07-19 | Reckitt Benckiser Nv | Detergent composition |
| DE102007029643A1 (de) * | 2006-09-08 | 2009-01-15 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| US8709990B2 (en) | 2011-10-19 | 2014-04-29 | Basf Se | Formulations, their use as or for producing dishwashing detergents and their production |
| WO2014017564A1 (ja) * | 2012-07-27 | 2014-01-30 | 花王株式会社 | 食器用洗浄剤組成物 |
| CN103146506A (zh) * | 2013-03-05 | 2013-06-12 | 广州立白企业集团有限公司 | 一种具有清除茶渍功效的餐具洗涤剂组合物 |
| EA201690076A1 (ru) * | 2013-06-25 | 2016-06-30 | Юнилевер Н.В. | Гигроскопичный моющий состав, содержащий воду, аминокарбоксилатный хелатирующий агент и влагочувствительные ингредиенты |
| EP2821471A1 (de) * | 2013-07-02 | 2015-01-07 | Basf Se | Wässrige Lösungen mit Komplexbildner in hoher Konzentration |
| DE102013214780A1 (de) * | 2013-07-29 | 2015-01-29 | Henkel Ag & Co. Kgaa | Pflegemittel für automatische Geschirrspülmaschinen |
| EP3228686B1 (de) | 2016-04-08 | 2021-10-27 | The Procter & Gamble Company | Automatisches geschirrspülen |
| EP3623456A1 (de) | 2018-09-12 | 2020-03-18 | CLARO Products GmbH | Silberschutzmittel |
| DE102019219812A1 (de) * | 2019-12-17 | 2021-06-17 | Henkel Ag & Co. Kgaa | Mittel für das maschinelle Geschirrspülen mit verbesserter Reinigungsleistung für bleichbare Anschmutzungen |
| JP7667265B2 (ja) | 2020-11-17 | 2025-04-22 | ザ プロクター アンド ギャンブル カンパニー | アルカリ性すすぎによる自動食器洗浄方法 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE4036695C2 (de) * | 1990-11-17 | 1994-08-25 | Henkel Kgaa | Verwendung von beta-Alaninderivaten als Gerüstsubstanzen in phosphatfreien Mitteln für das maschinelle Geschirrspülen |
| JPH05105900A (ja) * | 1991-08-27 | 1993-04-27 | Kao Corp | 洗浄機用洗浄剤 |
| DE4319798A1 (de) * | 1993-06-15 | 1994-12-22 | Henkel Kgaa | Geschirrspülmittel mit verringerter Belagsbildungsneigung |
| DE4319935A1 (de) * | 1993-06-16 | 1994-12-22 | Basf Ag | Verwendung von Glycin-N,N-diessigsäure-Derivaten als Komplexbildner für Erdalkali- und Schwermetallionen |
| US5481018A (en) * | 1995-03-31 | 1996-01-02 | The Dow Chemical Company | Amino nitrile intermediate for the preparation of alanine diacetic acid |
| GB2311536A (en) * | 1996-03-29 | 1997-10-01 | Procter & Gamble | Dishwashing and laundry detergents |
-
1997
- 1997-06-20 ES ES97928266T patent/ES2160355T3/es not_active Expired - Lifetime
- 1997-06-20 DE DE59712930T patent/DE59712930D1/de not_active Expired - Lifetime
- 1997-06-20 WO PCT/EP1997/003256 patent/WO1997049792A1/de not_active Ceased
- 1997-06-20 EP EP97928266A patent/EP0906407B1/de not_active Revoked
- 1997-06-20 EP EP01105118A patent/EP1113070B1/de not_active Revoked
- 1997-06-20 CA CA002258218A patent/CA2258218A1/en not_active Abandoned
- 1997-06-20 AT AT97928266T patent/ATE205250T1/de not_active IP Right Cessation
- 1997-06-20 AT AT01105118T patent/ATE389709T1/de not_active IP Right Cessation
- 1997-06-20 DE DE59704543T patent/DE59704543D1/de not_active Expired - Fee Related
- 1997-06-20 ES ES01105118T patent/ES2300287T3/es not_active Expired - Lifetime
Cited By (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2520641B1 (de) | 2005-06-30 | 2019-03-13 | The Procter & Gamble Company | Phosphatarme Maschinengeschirrspülmittel |
| US10240109B2 (en) | 2005-11-07 | 2019-03-26 | Reckitt Benckiser Finish B.V. | Composition |
| US9441189B2 (en) | 2005-11-07 | 2016-09-13 | Reckitt Benckiser Finish B.V. | Composition |
| US9920283B2 (en) | 2005-11-07 | 2018-03-20 | Reckitt Benckiser Finish B.V. | Composition |
| WO2008095563A1 (de) * | 2007-02-06 | 2008-08-14 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007006627A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| EP3567094A1 (de) | 2007-02-06 | 2019-11-13 | Henkel AG & Co. KGaA | Reinigungsmittel |
| US7879154B2 (en) | 2007-02-06 | 2011-02-01 | Henkel Ag & Co. Kgaa | Phosphate-free dishwashing detergents comprising builder, bleaching agent, nonionic surfactant, copolymer and a phosphonate |
| US8303721B2 (en) | 2007-02-06 | 2012-11-06 | Henkel Ag & Co. Kgaa | Detergent comprising a builder, a bleaching agent, and a copolymer |
| DE102007006630A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007006628A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| US9752100B2 (en) | 2007-02-06 | 2017-09-05 | Henkel Ag & Co. Kgaa | Detergents |
| DE202007019720U1 (de) | 2007-02-06 | 2017-06-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007006629A1 (de) | 2007-02-06 | 2008-08-07 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102007044417A1 (de) | 2007-09-17 | 2009-03-19 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102008060470A1 (de) | 2008-12-05 | 2010-06-10 | Henkel Ag & Co. Kgaa | Reinigungsmittel |
| DE102008063801A1 (de) | 2008-12-19 | 2010-06-24 | Henkel Ag & Co. Kgaa | Maschinelles Geschirrspülmittel |
| WO2010149695A1 (de) * | 2009-06-24 | 2010-12-29 | Henkel Ag & Co. Kgaa | Maschinelles reingungsmittel |
| WO2010149697A1 (de) * | 2009-06-24 | 2010-12-29 | Henkel Ag & Co. Kgaa | Maschinelles reinigungsmittel |
| DE102009029636A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschine Geschirrspülmittel |
| US8551930B2 (en) | 2009-09-21 | 2013-10-08 | Henkel Ag & Co. Kgaa | Dishwasher detergent |
| WO2011032870A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschinelles geschirrspülmittel |
| WO2011032869A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschinelles geschirrspülmittel |
| DE102009029637A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschinelles Geschirrspülmittel |
| DE102009029635A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschinelles Geschirrspülmittel |
| WO2011032868A1 (de) | 2009-09-21 | 2011-03-24 | Henkel Ag & Co. Kgaa | Maschinelles geschirrspülmittel |
| DE102009046216A1 (de) | 2009-10-30 | 2011-05-12 | Henkel Ag & Co. Kgaa | Maschinelles Geschirrspülmittel |
| WO2011051419A1 (de) | 2009-10-30 | 2011-05-05 | Henkel Ag & Co. Kgaa | Maschinelles geschirrspülmittel |
| DE102013019269A1 (de) | 2013-11-15 | 2015-06-03 | Weylchem Switzerland Ag | Geschirrspülmittel sowie dessen Verwendung |
| WO2018007298A1 (de) | 2016-07-05 | 2018-01-11 | Henkel Ag & Co. Kgaa | Geschirrspülmittel enthaltend zuckersäure und aminocarbonsäure |
| DE102016212248A1 (de) | 2016-07-05 | 2018-01-11 | Henkel Ag & Co. Kgaa | Geschirrspülmittel enthaltend Zuckersäure und Aminocarbonsäure |
| DE102024205394A1 (de) * | 2024-06-12 | 2025-12-18 | BSH Hausgeräte GmbH | Geschirrspülzusammensetzung zum Reinigen von Geschirr, Verfahren zum Spülen von Geschirr und Geschirrspülmaschine |
Also Published As
| Publication number | Publication date |
|---|---|
| ATE389709T1 (de) | 2008-04-15 |
| WO1997049792A1 (de) | 1997-12-31 |
| EP1113070B1 (de) | 2008-03-19 |
| CA2258218A1 (en) | 1997-12-31 |
| ES2160355T3 (es) | 2001-11-01 |
| EP0906407A1 (de) | 1999-04-07 |
| DE59704543D1 (de) | 2001-10-11 |
| EP1113070A3 (de) | 2002-01-02 |
| ES2300287T3 (es) | 2008-06-16 |
| DE59712930D1 (de) | 2008-04-30 |
| EP0906407B1 (de) | 2001-09-05 |
| ATE205250T1 (de) | 2001-09-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0906407B1 (de) | Mgda-haltige maschinengeschirrspülmittel niederer alkalität | |
| EP0842257B1 (de) | Geschirrwaschmaschinenspülmittel in form einer tablette | |
| DE69808885T2 (de) | Verfahren zum spülen von geschirr | |
| EP1076683B1 (de) | Festes maschinelles geschirrspülmittel mit phosphat und kristallinen schichtförmigen silikaten | |
| DE69726165T2 (de) | Schwachschäumende maschinengeschirrspülmittel | |
| EP0697036B1 (de) | Silberkorrosionsschutzmittel ii | |
| DE69229366T2 (de) | Phosphatfreies Maschinengeschirrspülmittel | |
| DE69931812T2 (de) | Waschmitteltablette | |
| EP3068859B1 (de) | Geschirrspülmittel sowie dessen verwendung | |
| EP2113024B1 (de) | Geschirrreinigungsmittel | |
| EP2329001B1 (de) | Bleichkatalysatormischungen bestehend aus mangansalzen und oxalsäure oder deren salze | |
| EP0512371B1 (de) | Granuläres phosphatfreies Mittel für das maschinelle Geschirrspülen | |
| EP1103599A1 (de) | Teilchenförmiger Klarspüler und maschinelle Geschirrspülmittel | |
| DE69022515T3 (de) | Bei niedrigen Temperaturen wirksame Bleichmittelzusammensetzungen für Textilien. | |
| WO1996012783A1 (de) | Kristalline schichtsilikate in maschinellen geschirrspülmitteln | |
| DE19704229A1 (de) | Cobuilderhaltiges Additiv für Wasch- oder Reinigungsmittel | |
| DE20014919U1 (de) | Teilchenförmige maschinelle Geschirrspülmittel mit Klarspüleffekt | |
| EP0846155B1 (de) | Verwendung von lipasen in niederalkalischen mitteln zum maschinellen geschirrspülen | |
| EP1050575B1 (de) | Alkalische Wasch- und Reinigungsmittelzusammensetzung enthaltend Alkylbenzolsulfonate und Alkanolamine | |
| WO1996022353A1 (de) | Reinigungsmittel enthaltend silberkorrosionsschutzmittel | |
| EP1857124A1 (de) | Zusammensetzung zur Reinigung dentaler Instrumente und Verfahren | |
| DE20221701U1 (de) | Reinigungs- und/oder Entkalkungstabletten für harte Oberflächen mit diskreten Farbzentren | |
| WO2001072949A1 (de) | Teilchenförmige maschinelle geschirrspülmittel mit klarspüleffekt | |
| GB2321466A (en) | Process for making tabletted detergent compositions | |
| DE19918457A1 (de) | Leistungsgesteigerte teilchenförmige Reinigungsmittel für das maschinelle Geschirrspülen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 906407 Country of ref document: EP |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: WAESCHENBACH, GUIDO, DR. Inventor name: ROBINSON, PAUL WILLIAM Inventor name: GERET, LAURENCE, DR. |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| RIC1 | Information provided on ipc code assigned before grant |
Free format text: 7C 11D 7/32 A, 7C 11D 7/54 B, 7C 11D 3/33 B, 7C 11D 3/39 B |
|
| 17P | Request for examination filed |
Effective date: 20020614 |
|
| AKX | Designation fees paid |
Free format text: AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| 17Q | First examination report despatched |
Effective date: 20041014 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: RECKITT BENCKISER N.V. |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 0906407 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 20080319 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REF | Corresponds to: |
Ref document number: 59712930 Country of ref document: DE Date of ref document: 20080430 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2300287 Country of ref document: ES Kind code of ref document: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080319 |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080826 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080619 |
|
| ET | Fr: translation filed | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080319 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| BERE | Be: lapsed |
Owner name: RECKITT BENCKISER N.V. Effective date: 20080630 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| 26 | Opposition filed |
Opponent name: THE PROCTER & GAMBLE COMPANY Effective date: 20081204 |
|
| 26 | Opposition filed |
Opponent name: HENKEL AG & CO. KGAA Effective date: 20081215 Opponent name: THE PROCTER & GAMBLE COMPANY Effective date: 20081204 Opponent name: DALLI-WERKE GMBH & CO. KG Effective date: 20081211 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080319 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080319 Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080630 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| 26 | Opposition filed |
Opponent name: BASF SE Effective date: 20081218 Opponent name: THE PROCTER & GAMBLE COMPANY Effective date: 20081204 Opponent name: HENKEL AG & CO. KGAA Effective date: 20081215 Opponent name: UNILEVER N.V. Effective date: 20081219 Opponent name: DALLI-WERKE GMBH & CO. KG Effective date: 20081211 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080630 |
|
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080630 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080630 |
|
| PLAB | Opposition data, opponent's data or that of the opponent's representative modified |
Free format text: ORIGINAL CODE: 0009299OPPO |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080620 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080620 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080620 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R064 Ref document number: 59712930 Country of ref document: DE Ref country code: DE Ref legal event code: R103 Ref document number: 59712930 Country of ref document: DE |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20120626 Year of fee payment: 16 |
|
| RDAF | Communication despatched that patent is revoked |
Free format text: ORIGINAL CODE: EPIDOSNREV1 |
|
| PLAB | Opposition data, opponent's data or that of the opponent's representative modified |
Free format text: ORIGINAL CODE: 0009299OPPO |
|
| R26 | Opposition filed (corrected) |
Opponent name: DALLI-WERKE GMBH & CO. KG Effective date: 20081211 |
|
| RDAG | Patent revoked |
Free format text: ORIGINAL CODE: 0009271 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT REVOKED |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20130627 Year of fee payment: 17 Ref country code: GB Payment date: 20130627 Year of fee payment: 17 |
|
| 27W | Patent revoked |
Effective date: 20121206 |
|
| GBPR | Gb: patent revoked under art. 102 of the ep convention designating the uk as contracting state |
Effective date: 20121206 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20130702 Year of fee payment: 17 Ref country code: IT Payment date: 20130626 Year of fee payment: 17 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R107 Ref document number: 59712930 Country of ref document: DE Effective date: 20131010 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MA03 Ref document number: 389709 Country of ref document: AT Kind code of ref document: T Effective date: 20121206 |