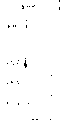CN1643587A - 在光数据载体信息层中作为光吸收化合物的方酸类染料 - Google Patents
在光数据载体信息层中作为光吸收化合物的方酸类染料 Download PDFInfo
- Publication number
- CN1643587A CN1643587A CNA038064146A CN03806414A CN1643587A CN 1643587 A CN1643587 A CN 1643587A CN A038064146 A CNA038064146 A CN A038064146A CN 03806414 A CN03806414 A CN 03806414A CN 1643587 A CN1643587 A CN 1643587A
- Authority
- CN
- China
- Prior art keywords
- light
- replacement
- general formula
- optical data
- phthalocyanine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 68
- 230000003287 optical effect Effects 0.000 title claims abstract description 55
- 239000000975 dye Substances 0.000 title description 19
- 239000000969 carrier Substances 0.000 title description 3
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 claims abstract description 45
- 239000000758 substrate Substances 0.000 claims abstract description 12
- 239000000463 material Substances 0.000 claims description 35
- -1 4Be hydrogen Chemical class 0.000 claims description 31
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 28
- 239000002253 acid Substances 0.000 claims description 27
- 239000002585 base Substances 0.000 claims description 27
- 229910052739 hydrogen Inorganic materials 0.000 claims description 25
- 239000001257 hydrogen Substances 0.000 claims description 25
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 21
- 125000000217 alkyl group Chemical group 0.000 claims description 18
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 16
- 230000001681 protective effect Effects 0.000 claims description 16
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 15
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 15
- 238000000034 method Methods 0.000 claims description 15
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 15
- 238000002360 preparation method Methods 0.000 claims description 15
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 14
- 125000001318 4-trifluoromethylbenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])*)C(F)(F)F 0.000 claims description 13
- 125000003118 aryl group Chemical group 0.000 claims description 13
- 150000003851 azoles Chemical class 0.000 claims description 12
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 10
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 10
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims description 9
- 239000007767 bonding agent Substances 0.000 claims description 9
- 229910052794 bromium Inorganic materials 0.000 claims description 9
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 9
- 238000000576 coating method Methods 0.000 claims description 9
- 238000003860 storage Methods 0.000 claims description 9
- 229910052801 chlorine Inorganic materials 0.000 claims description 8
- 239000000460 chlorine Substances 0.000 claims description 8
- 239000011248 coating agent Substances 0.000 claims description 8
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 7
- 239000002671 adjuvant Substances 0.000 claims description 7
- 125000004448 alkyl carbonyl group Chemical group 0.000 claims description 7
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 7
- 229910052740 iodine Inorganic materials 0.000 claims description 7
- 150000003233 pyrroles Chemical class 0.000 claims description 7
- 239000002904 solvent Substances 0.000 claims description 7
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 6
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 6
- 125000004210 cyclohexylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 claims description 6
- 125000006494 2-trifluoromethyl benzyl group Chemical group [H]C1=C([H])C([H])=C(C(=C1[H])C([H])([H])*)C(F)(F)F 0.000 claims description 5
- 125000006495 3-trifluoromethyl benzyl group Chemical group [H]C1=C([H])C(=C([H])C(=C1[H])C([H])([H])*)C(F)(F)F 0.000 claims description 5
- 125000004217 4-methoxybenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1OC([H])([H])[H])C([H])([H])* 0.000 claims description 5
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 claims description 5
- 125000004429 atom Chemical group 0.000 claims description 5
- 229910052732 germanium Inorganic materials 0.000 claims description 5
- 239000011630 iodine Substances 0.000 claims description 5
- 229910052710 silicon Inorganic materials 0.000 claims description 5
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 5
- 229910052718 tin Inorganic materials 0.000 claims description 5
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims description 4
- 239000003513 alkali Substances 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 3
- UTVVREMVDJTZAC-UHFFFAOYSA-N furan-2-amine Chemical class NC1=CC=CO1 UTVVREMVDJTZAC-UHFFFAOYSA-N 0.000 claims description 2
- 150000002366 halogen compounds Chemical class 0.000 claims description 2
- 125000000623 heterocyclic group Chemical group 0.000 claims description 2
- PWEBUXCTKOWPCW-UHFFFAOYSA-N squaric acid Chemical compound OC1=C(O)C(=O)C1=O PWEBUXCTKOWPCW-UHFFFAOYSA-N 0.000 claims description 2
- AOSZTAHDEDLTLQ-AZKQZHLXSA-N (1S,2S,4R,8S,9S,11S,12R,13S,19S)-6-[(3-chlorophenyl)methyl]-12,19-difluoro-11-hydroxy-8-(2-hydroxyacetyl)-9,13-dimethyl-6-azapentacyclo[10.8.0.02,9.04,8.013,18]icosa-14,17-dien-16-one Chemical compound C([C@@H]1C[C@H]2[C@H]3[C@]([C@]4(C=CC(=O)C=C4[C@@H](F)C3)C)(F)[C@@H](O)C[C@@]2([C@@]1(C1)C(=O)CO)C)N1CC1=CC=CC(Cl)=C1 AOSZTAHDEDLTLQ-AZKQZHLXSA-N 0.000 claims 1
- 229940126657 Compound 17 Drugs 0.000 claims 1
- 239000010410 layer Substances 0.000 abstract description 39
- 239000011230 binding agent Substances 0.000 abstract 1
- 239000011241 protective layer Substances 0.000 abstract 1
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 21
- 239000000047 product Substances 0.000 description 18
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 15
- 239000000853 adhesive Substances 0.000 description 14
- 230000001070 adhesive effect Effects 0.000 description 14
- 239000012071 phase Substances 0.000 description 14
- 239000000843 powder Substances 0.000 description 14
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 14
- 239000007787 solid Substances 0.000 description 13
- 239000000243 solution Substances 0.000 description 13
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 12
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 12
- 150000007513 acids Chemical class 0.000 description 11
- 238000007740 vapor deposition Methods 0.000 description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 10
- 238000006243 chemical reaction Methods 0.000 description 10
- 238000003756 stirring Methods 0.000 description 10
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- 238000002835 absorbance Methods 0.000 description 9
- 229910052751 metal Inorganic materials 0.000 description 8
- 239000002184 metal Substances 0.000 description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 7
- 238000010521 absorption reaction Methods 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical group O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 6
- 238000000354 decomposition reaction Methods 0.000 description 6
- 239000006185 dispersion Substances 0.000 description 6
- 230000031700 light absorption Effects 0.000 description 6
- 239000002245 particle Substances 0.000 description 6
- APSBXTVYXVQYAB-UHFFFAOYSA-M sodium docusate Chemical compound [Na+].CCCCC(CC)COC(=O)CC(S([O-])(=O)=O)C(=O)OCC(CC)CCCC APSBXTVYXVQYAB-UHFFFAOYSA-M 0.000 description 6
- 238000005406 washing Methods 0.000 description 6
- 238000002425 crystallisation Methods 0.000 description 5
- 229910052736 halogen Inorganic materials 0.000 description 5
- 150000002367 halogens Chemical class 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 238000000465 moulding Methods 0.000 description 5
- 238000002310 reflectometry Methods 0.000 description 5
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- 230000015556 catabolic process Effects 0.000 description 4
- 230000008859 change Effects 0.000 description 4
- 238000001816 cooling Methods 0.000 description 4
- 230000008025 crystallization Effects 0.000 description 4
- 238000006731 degradation reaction Methods 0.000 description 4
- 238000010790 dilution Methods 0.000 description 4
- 239000012895 dilution Substances 0.000 description 4
- 238000005469 granulation Methods 0.000 description 4
- 230000003179 granulation Effects 0.000 description 4
- 239000008188 pellet Substances 0.000 description 4
- 238000004528 spin coating Methods 0.000 description 4
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- AGEZXYOZHKGVCM-UHFFFAOYSA-N benzyl bromide Chemical compound BrCC1=CC=CC=C1 AGEZXYOZHKGVCM-UHFFFAOYSA-N 0.000 description 3
- 230000006837 decompression Effects 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 239000012530 fluid Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 238000000016 photochemical curing Methods 0.000 description 3
- XQZYPMVTSDWCCE-UHFFFAOYSA-N phthalonitrile Chemical group N#CC1=CC=CC=C1C#N XQZYPMVTSDWCCE-UHFFFAOYSA-N 0.000 description 3
- 229920006391 phthalonitrile polymer Polymers 0.000 description 3
- 229920000515 polycarbonate Polymers 0.000 description 3
- 239000004417 polycarbonate Substances 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 235000012239 silicon dioxide Nutrition 0.000 description 3
- 239000012279 sodium borohydride Substances 0.000 description 3
- 229910000033 sodium borohydride Inorganic materials 0.000 description 3
- 238000001694 spray drying Methods 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 238000000967 suction filtration Methods 0.000 description 3
- 239000000080 wetting agent Substances 0.000 description 3
- JTPNRXUCIXHOKM-UHFFFAOYSA-N 1-chloronaphthalene Chemical compound C1=CC=C2C(Cl)=CC=CC2=C1 JTPNRXUCIXHOKM-UHFFFAOYSA-N 0.000 description 2
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 2
- 241000931526 Acer campestre Species 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 2
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 2
- 239000004820 Pressure-sensitive adhesive Substances 0.000 description 2
- 229910052581 Si3N4 Inorganic materials 0.000 description 2
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- 230000004447 accommodation reflex Effects 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 239000004411 aluminium Substances 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 238000013500 data storage Methods 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 239000012065 filter cake Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 239000010931 gold Substances 0.000 description 2
- 229910052737 gold Inorganic materials 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000005286 illumination Methods 0.000 description 2
- 238000001746 injection moulding Methods 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 229910001507 metal halide Inorganic materials 0.000 description 2
- 150000005309 metal halides Chemical class 0.000 description 2
- 238000001208 nuclear magnetic resonance pulse sequence Methods 0.000 description 2
- 239000003973 paint Substances 0.000 description 2
- 230000010287 polarization Effects 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 238000012797 qualification Methods 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 2
- 239000004332 silver Substances 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 239000007921 spray Substances 0.000 description 2
- 238000003892 spreading Methods 0.000 description 2
- 230000007480 spreading Effects 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 239000012749 thinning agent Substances 0.000 description 2
- TXUICONDJPYNPY-UHFFFAOYSA-N (1,10,13-trimethyl-3-oxo-4,5,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-17-yl) heptanoate Chemical compound C1CC2CC(=O)C=C(C)C2(C)C2C1C1CCC(OC(=O)CCCCCC)C1(C)CC2 TXUICONDJPYNPY-UHFFFAOYSA-N 0.000 description 1
- TXVVVEUSVBLDED-UHFFFAOYSA-N 1-(bromomethyl)-2-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=CC=C1CBr TXVVVEUSVBLDED-UHFFFAOYSA-N 0.000 description 1
- ATLQGZVLWOURFU-UHFFFAOYSA-N 1-(bromomethyl)-3,5-bis(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC(CBr)=CC(C(F)(F)F)=C1 ATLQGZVLWOURFU-UHFFFAOYSA-N 0.000 description 1
- IKSNDOVDVVPSMA-UHFFFAOYSA-N 1-(bromomethyl)-4-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=C(CBr)C=C1 IKSNDOVDVVPSMA-UHFFFAOYSA-N 0.000 description 1
- MFFMQGGZCLEMCI-UHFFFAOYSA-N 2,4-dimethyl-1h-pyrrole Chemical compound CC1=CNC(C)=C1 MFFMQGGZCLEMCI-UHFFFAOYSA-N 0.000 description 1
- 125000001731 2-cyanoethyl group Chemical group [H]C([H])(*)C([H])([H])C#N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004801 4-cyanophenyl group Chemical group [H]C1=C([H])C(C#N)=C([H])C([H])=C1* 0.000 description 1
- 125000004860 4-ethylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004199 4-trifluoromethylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C(F)(F)F 0.000 description 1
- PAPNRQCYSFBWDI-UHFFFAOYSA-N DMP Natural products CC1=CC=C(C)N1 PAPNRQCYSFBWDI-UHFFFAOYSA-N 0.000 description 1
- COXRPBBNPBCNDM-UHFFFAOYSA-N Dioxamide Natural products C1=C2OCOC2=CC(C=CC(=O)N(CCC=2C=C3OCOC3=CC=2)C)=C1 COXRPBBNPBCNDM-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 240000007651 Rubus glaucus Species 0.000 description 1
- 235000011034 Rubus glaucus Nutrition 0.000 description 1
- 235000009122 Rubus idaeus Nutrition 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- 229910021626 Tin(II) chloride Inorganic materials 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- KURZCZMGELAPSV-UHFFFAOYSA-N [Br].[I] Chemical compound [Br].[I] KURZCZMGELAPSV-UHFFFAOYSA-N 0.000 description 1
- SYKNUAWMBRIEKB-UHFFFAOYSA-N [Cl].[Br] Chemical compound [Cl].[Br] SYKNUAWMBRIEKB-UHFFFAOYSA-N 0.000 description 1
- 239000011358 absorbing material Substances 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000012752 auxiliary agent Substances 0.000 description 1
- 238000000498 ball milling Methods 0.000 description 1
- 230000008033 biological extinction Effects 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 125000004744 butyloxycarbonyl group Chemical group 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- 125000002603 chloroethyl group Chemical group [H]C([*])([H])C([H])([H])Cl 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 239000011362 coarse particle Substances 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 239000007859 condensation product Substances 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 238000001723 curing Methods 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 125000006165 cyclic alkyl group Chemical group 0.000 description 1
- 125000004186 cyclopropylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C1([H])[H] 0.000 description 1
- DUVPPTXIBVUIKL-UHFFFAOYSA-N dibromogermanium Chemical compound Br[Ge]Br DUVPPTXIBVUIKL-UHFFFAOYSA-N 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 125000004185 ester group Chemical class 0.000 description 1
- 230000005281 excited state Effects 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 230000005283 ground state Effects 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000008676 import Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 125000004344 phenylpropyl group Chemical group 0.000 description 1
- 239000001007 phthalocyanine dye Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 239000011164 primary particle Substances 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000007127 saponification reaction Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 239000001119 stannous chloride Substances 0.000 description 1
- 235000011150 stannous chloride Nutrition 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005389 trialkylsiloxy group Chemical group 0.000 description 1
- 125000004044 trifluoroacetyl group Chemical group FC(C(=O)*)(F)F 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/242—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers
- G11B7/244—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only
- G11B7/246—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B47/00—Porphines; Azaporphines
- C09B47/04—Phthalocyanines abbreviation: Pc
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B47/00—Porphines; Azaporphines
- C09B47/04—Phthalocyanines abbreviation: Pc
- C09B47/08—Preparation from other phthalocyanine compounds, e.g. cobaltphthalocyanineamine complex
- C09B47/085—Preparation from other phthalocyanine compounds, e.g. cobaltphthalocyanineamine complex substituting the central metal atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B57/00—Other synthetic dyes of known constitution
- C09B57/007—Squaraine dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0097—Dye preparations of special physical nature; Tablets, films, extrusion, microcapsules, sheets, pads, bags with dyes
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/242—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers
- G11B7/244—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/242—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers
- G11B7/244—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only
- G11B7/246—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing dyes
- G11B7/248—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing dyes porphines; azaporphines, e.g. phthalocyanines
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/242—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers
- G11B7/244—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only
- G11B7/249—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing organometallic compounds
- G11B7/2492—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of recording layers comprising organic materials only containing organometallic compounds neutral compounds
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/252—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers
- G11B7/253—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers of substrates
- G11B7/2533—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers of substrates comprising resins
- G11B7/2534—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers of substrates comprising resins polycarbonates [PC]
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/252—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers
- G11B7/254—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers of protective topcoat layers
- G11B7/2542—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers of protective topcoat layers consisting essentially of organic resins
-
- G—PHYSICS
- G11—INFORMATION STORAGE
- G11B—INFORMATION STORAGE BASED ON RELATIVE MOVEMENT BETWEEN RECORD CARRIER AND TRANSDUCER
- G11B7/00—Recording or reproducing by optical means, e.g. recording using a thermal beam of optical radiation by modifying optical properties or the physical structure, reproducing using an optical beam at lower power by sensing optical properties; Record carriers therefor
- G11B7/24—Record carriers characterised by shape, structure or physical properties, or by the selection of the material
- G11B7/241—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material
- G11B7/252—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers
- G11B7/258—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers of reflective layers
- G11B7/259—Record carriers characterised by shape, structure or physical properties, or by the selection of the material characterised by the selection of the material of layers other than recording layers of reflective layers based on silver
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Optical Record Carriers And Manufacture Thereof (AREA)
- Thermal Transfer Or Thermal Recording In General (AREA)
- Pyrrole Compounds (AREA)
Abstract
本发明公开了光数据载体用的新的方酸类化合物。优选透明的基材,其表面施涂了可光写入的信息层、一个或两个任选的反射层以及另一基材或保护层,所述光数据载体可用红光,优选激光写入和读出。信息层包含光吸收化合物和任选的粘合剂,其中至少一种本发明方酸类化合物被用作光吸收化合物。
Description
技术领域
本发明涉及方酸类(squarylium)染料,其制备方法以及方酸类染料的主要成分及其制备,还涉及在其信息层中包含方酸类染料的光数据载体。
采用特殊光吸收物质或其混合物的“一次写入”光数据载体特别适合用于以红(635~660nm)激光二极管操作的DVD-R光盘,和上述染料在聚合物基材上的施涂,特别是采用旋涂涂布到聚碳酸酯上。
一次写入光盘(CD-R,780nm)近来有了大幅增长,代表着技术上成熟的系统。
下一代光数据存储介质——DVD——目前正在市场上推出。较短波长激光辐射(635~660nm)和较高数值孔径NA(NA=数值孔径)的应用使存储密度得以增加。在此种情况下的可写入格式是DVD-R。
可达到的存储密度取决于激光斑在信息平面内的聚焦。斑点的大小用激光波长λ/NA衡量。NA是所用物镜的数值孔径。为获得尽可能高的存储密度,应以用尽可能小的波长λ作为目标。目前,在采用半导体激光二极管的条件下可能达到390nm。
除了上述光学性质之外,含有光吸收有机物质的可写入信息层必须具有基本上无定形的形态才能将写入和读出期间的噪音信号维持在尽可能低的水平。正因为如此,特别优选的是,在随后的减压下涂布金属或电介质层期间采用从溶液旋涂、蒸汽沉积和/或升华的物质施涂中,防止光吸收物质的结晶。
含有光吸收物质的无定形层优选具有高抗热变形性,因为否则通过溅射或蒸汽沉积所施涂的有机或无机材料等以后各层将由于扩散而形成模糊的边界并从而对反射率产生负面影响。再者,抗热畸变能力不足的光吸收物质可能,在与聚合物载体的边界处,扩散到后者中并从而对反射率产生不利影响。
蒸汽压过高的光吸收物质会在上述以后各层的高真空溅射或蒸汽沉积施涂期间发生升华,从而减少所要求的层厚。这又进而对反射率产生负面影响。
因此,本发明的目的是提供满足在一次写入光数据载体中的信息层中使用时对于在600~680nm激光波长范围可写入的光数据存储格式的高要求(例如,光稳定性、有利的信噪比、对基材的无损施涂等)的适宜化合物。
现已惊奇地发现,选自特定对称方酸类化合物的光吸收化合物能特别好地满足上述综合要求。
因此,本发明提供了通式I的方酸类化合物,
其中
R是五元杂环,特别是取代或未取代的吡咯,但氨基取代的呋喃环除外。
优选对应于通式Ia的通式I的方酸类化合物,
其中
R1是氢、取代或未取代的烷基或取代或未取代的芳烷基,
R2是取代或未取代的烷基、取代或未取代的芳基、烷氧羰基或取代或未取代的烷羰基,且
R3和R4彼此独立地是氢、取代或未取代的烷基、取代或未取代的芳基、烷氧羰基或取代或未取代的烷羰基。
通式Ia是可能的内消旋结构式之一。
就本发明目的而言,“烷基”优选是C1-C6-烷基;“芳基”优选是C6~C10-芳基;“芳烷基”优选是C7~C16-芳烷基;“烷氧基”优选是C1~C6-烷氧基。
在烷基、芳基或芳烷基基团上可能的取代基是卤素,特别是F,羟基、硝基、氰基、羧基、烷氧基、三烷基甲硅烷基和三烷基甲硅烷氧基。这些烷基基团可以是直链、环状或支链的。它们可以是部分卤代的或者全卤代的。取代的烷基基团的例子是三氟甲基、氯乙基、氰基乙基、甲氧基乙基。环状烷基基团的例子是环己基甲基和环丙基甲基。支链烷基基团的例子是异丙基、叔丁基、2-丁基、新戊基。可能的芳基的例子是苯基、4-甲氧基苯基、4-氰基苯基、3,5-双(三氟甲基)苯基、4-三氟甲基苯基和4-乙基苯基。芳烷基基团的例子是苄基、苯乙基、苯丙基、4-甲氧基苄基、4-氰基苄基、3,5-双(三氟甲基)苄基、4-三氟甲基苄基和4-乙基苄基。羧基基团的例子是乙氧羰基、丁氧羰基、和三氟甲氧羰基。烷基羰基的例子是乙酰基、三氟乙酰基、丙酰基、丁酰基、戊酰基和己酰基。
优选的取代或未取代的烷基基团是甲基、乙基、正丙基、正戊基、异丁基、异丙基、全氟代甲基和乙基。
优选的取代或未取代的芳烷基基团,例如是,4-三氟甲基苄基、2-三氟甲基苄基、3,5-双三氟甲基苄基和4-氟-2-三氟甲基苄基。
优选的烷氧羧基基团是乙氧羰基。
特别优选这样的通式Ia的方酸类化合物,其中
R1是氢、甲基、乙基、丙基、丁基、环己基甲基、苄基、4-甲氧基苄基、4-三氟甲基苄基、3-三氟甲基苄基、2-三氟甲基苄基、3,5-双(三氟甲基)苄基或4-氟-2-三氟甲基苄基,
R2是甲基、乙基、丙基或苯基,且
R3和R4彼此独立地是氢、甲基、乙基、丙基、丁基、苯基、乙酰基或乙氧羰基。
更优选这样的通式Ia的方酸类化合物,其中
R1是4-三氟甲基苄基或3,5-双(三氟甲基)苄基,特别是4-三氟甲基苄基,
R2是甲基、乙基或苯基,特别是甲基,
R3是氢、乙基、乙酰基或乙氧羰基,特别是乙基,且
R4是甲基、乙基或苯基,特别是甲基或乙基。
本发明还提供了制备本发明方酸类化合物的方法,其特征在于,将3,4-二羟基-3-环丁烯-1,2-二酮(方形酸)与至少一种通式III的化合物起反应
R-R5 (III)
特别是与通式(IIIa)的吡咯化合物反应,优选在适当溶剂中反应,
其中
R5是氢、烷氧羰基,特别是叔丁氧羰基或乙氧羰基或羧基,且
R和R1~R4如上所定义。
本发明方法优选在醇中实施,特别是在乙醇中。优选的反应温度大于70℃,特别是75~85℃。本发明方法同样也优选地在含水乙酸中实施。乙酸/水的混合比例如介于3∶1~1∶3,优选2∶1~1∶2,尤其优选1∶1。优选的反应温度介于室温~介质的沸点。同样也优选采用催化量的无机酸,特别是盐酸。产物通常作为纯固体形式从反应溶液中沉淀,并在分离出来以后优选地以醚洗涤。
本发明还提供优选用于制备本发明方酸类化合物的通式(IIIa)的吡咯化合物。
因此本发明还提供了通式(IIIa)的吡咯化合物,
其中
R1是取代或未取代的C3~C12-烷基或取代或未取代的芳烷基,
R2是取代或未取代的烷基、取代或未取代的芳基、烷氧羰基、羰基或取代或未取代的烷基羰基,
R3和R4彼此独立地是氢、取代或未取代的烷基、取代或未取代的芳基、烷氧羰基、羰基或取代或未取代的烷氧羰基,且
R5是氢、烷氧羰基或羧基。
特别优选这样的通式IIIa的吡咯化合物,其中
R1是丙基、丁基、环己基甲基、苄基、4-甲氧基苄基、4-三氟甲基苄基、3-三氟甲基苄基、2-三氟甲基苄基、3,5-双(三氟甲基)苄基或4-氟-2-三氟甲基苄基,
R2是甲基、乙基、丙基或苯基,
R3和R4彼此独立地是氢、甲基、乙基、丙基、丁基、苯基、乙酰基或乙氧羰基,且
R5是氢、叔丁氧羰基或羧基。
本发明同样也提供了制备通式IIIa的新吡咯化合物的方法,其特征在于,将通式(II)的吡咯化合物
其中
R2~R4如上面针对通式IIIa的新吡咯所定义,且
R5是氢、烷氧羰基,特别是叔丁氧羰基、乙氧羰基或羧基,与通式IV的卤素化合物
R1-X (IV),
其中
R1如上面针对通式IIIa的新吡咯所定义,
X是Cl、Br或I,
以及至少2当量碱进行反应。
特别合适的碱是KOH。反应优选在适当溶剂,例如,在二甲亚砜(DMSO)、二甲基甲酰胺(DMF)或其混合物中实施。反应优选在20~100℃,尤其优选50~90℃,特别是65~80℃的温度实施。如果通式II中的R5是乙氧羰基,则该酯基团皂化生成吡咯羧酸的对应碱金属盐。该产物可通过酸化从水溶液中沉淀,然后过滤出来。
按此方式,该产物可以以满意的纯度获得,而不需要复杂的结晶步骤或类似的提纯步骤。
本发明还提供本发明方酸类染料作为光吸收化合物在一次写入光数据载体的信息层中的应用。
在此种应用中,光数据载体优选地用红激光,特别是波长介于600~680nm的激光写入和读出。
本发明还提供了方酸类化合物作为光吸收化合物在可用红激光,特别是波长介于600~680nm的激光写入和读出的一次写入光数据载体信息层中的应用。
本发明还提供了光数据载体,它包含优选透明的基材,所述基材的表面施涂了可光写入信息层、任选的一个或多个反射层以及另外的基材或保护层,其可用红光,优选波长介于600~680nm的光,优选激光写入和读出,其中信息层包含光吸收化合物以及任选的粘合剂,其特征在于,至少一种本发明方酸类染料被用作光吸收化合物。
该光吸收化合物优选应能遇热变化。此种热变化优选发生在<600℃的温度,尤其优选在<400℃的温度,非常尤其优选在<300℃的温度,特别是<200℃的温度。此种变化可以是,例如,光吸收化合物的发色中心的分解或化学变化。
同样也优选的是,光吸收化合物仅在高于100℃时发生热变化。
本发明光数据存储介质中的光吸收化合物的优选实施方案对应于本发明方酸类染料的优选实施方案。
在优选的实施方案中,所用光吸收化合物是这样的通式(Ia)的化合物,其中
R1是氢、甲基、乙基、丙基、丁基、环己基甲基、苄基、4-甲氧基苯基、4-三氟甲基苄基、3-三氟甲基苄基或3,5-双(三氟甲基)苄基,
R2、R3和R4彼此独立地是氢、甲基、乙基、丙基、丁基、苯基、乙酰基或乙氧羰基。
在特别优选的实施方案中,所用光吸收化合物是这样的通式(Ia)的化合物,其中
R1是4-三氟甲基苄基或3,5-双(三氟甲基)苄基或,特别是,4-三氟甲基苄基,
R2和R4彼此独立地是甲基、乙基或苯基,特别是甲基或乙基,且
R3是氢、乙基、乙酰基或乙氧羰基,特别是乙基。
在利用红激光写入和读出的本发明一次写入光数据载体的情况下,优选这样的光吸收化合物,其最大吸收的波长λmax介于500~650nm,其中波长λ1/2即,波长λmax2处的、吸光最大值的长波长侧翼中的吸光度是λmax处吸光度值一半时的波长位置,与波长λ1/10,即,λmax处的吸光最大值的长波长侧翼中的吸光度是λmax处吸光度值的十分之一时的波长位置,之间相距不超过60nm。此种光吸收化合物优选地直至750nm,尤其优选800nm,非常尤其优选直至850nm不具有较长-波长最大值λmax3。
优选具有510~620nm的吸光最大值(波长位置)λmax的光吸收化合物。
尤其优选具有530~610nm的吸光最大值λmax的光吸收化合物。
非常尤其优选具有550~600nm的吸光最大值λmax的光吸收化合物。
在此种光吸收化合物中,上面规定的λ1/2和λ1/10优选相距不超过50nm,尤其优选不超过40nm,非常尤其优选不超过30nm。
光吸收化合物优选具有>60000l/mol cm,优选>80000l/mol cm,尤其优选>100000l/mol cm,非常尤其优选>120000l/mol cm的摩尔消光系数。
特别合适的方酸类化合物是那些,其偶极矩变化Δμ=|μg-μag|,即,基态与第一受激态之间的偶极矩之差的绝对值非常小,优选<5D,尤其优选<2D。测定该偶极矩变化Δμ的方法描述在,例如,F.Wurthner等人的《Angew.Chem.》1997,109,2933以及其中所援引的文献中。低的溶剂-诱导波长移动(二噁烷/DMF)同样也是一个适当的选择标准。优选其溶剂-诱导波长移动Δλ=|λDMF-λ二噁烷|,即,在溶剂二甲基甲酰胺与在二噁烷中的吸收,波长之差的绝对值为<20nm,尤其优选<10nm,非常尤其优选<5nm的方酸类化合物。
吸收光谱优选在溶液中测定。
本发明使用的光吸收化合物优选地能够达到>10%的在未写入状态光数据载体中的反射率,和高到足以使信息层在以汇聚光,如果该光的波长介于600~680nm,逐点照射后发生热降解的吸收。数据载体上写入和未写入点之间的反差是通过因热降解后信息层光学性质的改变导致入射光振幅和相位的反射率变化实现的。
本发明方酸类染料优选通过旋涂而施加到光数据载体上。它们可以彼此混合或与具有类似光谱性质的其他染料混合。信息层不仅可包含本发明方酸类染料,而且可包含添加剂,例如,粘合剂、润湿剂、稳定剂、稀释剂和增感剂以及进一步的成分。
除了信息层之外,其他层如金属层、电介质层和保护层也可存在于本发明的光数据储存器中。金属和电介质用于,特别是,调节反射率和热吸收/保持。金属,视激光波长而定,可以是金、银、铝等。电介质层的例子是二氧化硅和氮化硅。保护层是,例如,(可)光固化表面涂层(压敏)粘合剂层和保护膜。
压敏粘合剂层主要由丙烯酸类粘合剂组成。例如Nitto Denko DA-8320或DA-8310,公开在专利JP-A 11-273147中,可用于此目的。
本发明光数据载体具有,例如,下列层结构(参见附图2):优选透明的基材(11)、信息层(12),任选的反射层(13)、任选的粘合剂层(14)、另外的优选透明的基材(15)。
在附图2中,基材(15)优选地换成由层(13)、(12)和(11)组成的顺序层。
或者,光数据载体的结构可
●包含多个优选地被适当层分开的信息层。特别优选的分隔层是光固化表面涂层、粘合剂层或反射层。
附图1、附图2和附图3中所示箭头指出入射光的路径。
本发明还提供了用红光,特别是红激光,尤其优选具有600~680nm波长的红激光,写入的本发明光数据载体。
本发明同样也提供了一次写入光数据载体,其信息层包含至少一种酞菁染料作为光吸收化合物,还提供生产它的方法。
因此,本发明的目的是提供合适的化合物,它满足一次写入光数据载体中的信息层,特别是在360~460nm激光波长范围高密度可写入光数据存储格式的高要求(例如,光稳定性、有利的信噪比、对基材的无损施涂等)。
现已惊奇地发现,作为光吸收化合物的特定酞菁能特别好地满足上述综合要求。酞菁表现出在波长范围360~460nm这一对激光,即,B或Soret带特别重要范围的强烈吸收。
因此,本发明提供了包含优选透明的基材的光数据载体,基材可任选具有预先涂布的一个或多个反射层并且其表面上涂布了可光写入信息层、任选的一个或多个反射层,以及任选的保护层或另外的基材或覆盖层,该光数据载体可用蓝光,优选激光,特别优选波长介于360~460nm,特别是380~420nm,非常特别优选390~410nm的光,或者用红外光,优选激光,特别优选波长介于760~830nm的光写入和读出,其中信息层包含光吸收化合物以及任选的粘合剂,其特征在于,采用至少一种通式(I)的酞菁作为光吸收化合物
其中
Me是双轴向取代的金属原子,选自Si、Ge和Sn,
Pc是未取代的酞菁,且
X1和X2彼此独立地是溴或碘,且X1也可以是氯。
优选采用通式(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)和(Ih)的酞菁
通式(Ic)、(If)和(Ih)应理解为指出,这两个卤素原子,或是两个都位于酞菁环平面上方,或是一个位于上方,一个位于酞菁环平面下方。
本发明使用的酞菁是公知的,例如可见诸于J.N.Esposito,L.E.Sutton,M.E.Kenney 《无机化学》6,1967,1116和W.J.Kroenke,M.E.Kenney,《无机化学》3,1964,696,或者可按照其中的描述制备。
它们原则上可按照公知的方法制备,例如:
√通过以邻苯二甲腈或氨基-亚氨基-异吲哚为原料在适当金属卤
化物存在下的环合成,
√任选地,令该产物与水在适当溶剂,例如,吡啶中起反应生成
通式(I)的酞菁,其中X1=X2=OH,
√任选地,用其他适当卤化物取代轴向取代基X1=X2=卤素,
√任选地,通过与HX1/HX2的反应用适当卤素取代轴向取代基
X1=X2=OH,
√任选地,利用溴、碘、溴氯或碘溴氧化通式(II)的非轴向取代的酞菁
MePc (II)。
通式(II)的酞菁,优选其中Me=Sn者,例如可通过通式(I)的酞菁的还原来制备,优选Me=Sn,其中X1和X2是卤素,例如,氯。合适的还原剂例如是四氢硼酸钠(NaBH4)。
光吸收化合物可遇热变化。热变化优选地发生在<600℃的温度。此种变化例如是光吸收化合物的发色中心的分解或化学变化。
所描述的光吸收物质保证该光数据载体在未写入状态具有足够高的反射率,和在汇聚蓝光,特别是激光,优选波长介于360~460nm的激光,逐点照射后高到足以使信息层热降解的吸收。数据载体上写入和未写入点之间的反差是由因热降解后信息层光学性质的变化所致入射光振幅和相位的反射率变化实现的。
这就是说,光数据载体可借助波长介于360~460nm的激光写入和读出。
光数据载体同样也可借助红外光,特别是波长介于760~830nm的激光写入和读出,这时沟纹间距和沟纹几何形状应与波长和数值孔径相匹配。
本发明还提供了通式(I)的酞菁作为光吸收化合物在光存储介质的信息层中的应用。
本发明同样也提供通式(I)的酞菁在生产光存储介质中的应用。酞菁优选地用作信息层中的光吸收化合物。
特别优选用于此种用途的酞菁具有大于90wt%,特别是大于95wt%,特别优选大于98wt%的含量,以通式(I)的酞菁为基准计。
本发明还提供通式(I)酞菁的粒状固体制剂,其特征在于,该颗粒具有0.5μm~10mm的平均粒度。
在粒状固体制剂的优选实施方案中,优选平均粒度介于0.5~20μm,特别是1~10μm,以下称细粉末的颗粒。此种细粉末可采用,例如研磨来生产。
同样也优选平均粒度介于50~300μm的粒状固体制剂,以下称细结晶形式。
另一类优选的粒状固体制剂是平均粒度介于50μm~10mm,优选100μm~800μm,并成形为一次颗粒的附聚物或凝聚物形式的粒状造型体。此种造型体例如可具有滴状、覆盆子、薄片或棒状的形状,以下称做粒料。
细结晶形式的粒度例如可通过合成中参数的选择来达到。例如,诸组分(邻苯二甲腈或氨基-亚氨基-异吲哚和适当金属卤化物在适当溶剂中)的混合物在例如30~60min时间内迅速加热至反应温度,例如,160~220℃,将择优地形成精细分散的形式。类似的结果在仅当反应温度介于,例如,160~190℃时才将金属卤化物加入到反应混合物(邻苯二甲腈或氨基-亚氨基-异吲哚在适当溶剂中)的情况下获得。诸组分的混合物的慢加热,例如,在65~250min时间内加热至反应温度,例如,160~220℃,将择优地形成粗颗粒形式。
本发明粒状固体制剂优选地包含
80~100wt%,优选95~100wt%通式(I)的酞菁,
0.1~1.0wt%,优选0.1~0.5wt%残余水份,
0~10wt%无机盐,
0~10wt%,优选0~5wt%其他添加剂,例如,分散剂、表面活性剂和/或润湿剂,
其中每种情况的百分数都以制剂为基准计并且给出的诸比例之和是100%。
本发明固体制剂优选具有低粉尘含量、能自由流动并且具有良好贮存稳定性。
该粒料可采用各种不同方法生产,例如,通过喷雾干燥、流化床喷雾造粒、流化床生长造粒或粉末流化床附聚。
优选采用喷雾干燥的造粒法,其中特别是旋转圆盘分析器和单-流体或双流体喷嘴,都可以作为喷雾装置。优选采用单流体喷嘴,特别是旋涡腔喷嘴,并优选令其操作在20~80巴压力下。
喷雾干燥期间进、出口温度取决于所要求的残余水份含量、安全措施和经济因素。进口温度优选介于120~200℃,尤其是140~180℃,出口温度优选介于40~80℃。
粒料一般是这样生产的,首先,在搅拌容器中将染料过滤滤饼,连同,恰当的话,助剂和添加剂一起,剧烈地混合。将该悬浮液的晶体优选地放在磨,例如,球磨中粉碎,从而获得进行分散的可微粒化悬浮液。
在优选的实施方案中,该染料悬浮液是一种水悬浮液。造粒是通过喷雾干燥实施的。
本发明还提供了固体成形体,例如,丸、挤出物等,其中包含通式(I)的酞菁,优选含量大于90wt%,特别是大于95wt%,优选大于98wt%,以成形体为基准计。加入到固体造型体中的其他添加剂可以是粘合剂。通式(I)酞菁和粘合剂之和优选大于95wt%,尤其优选大于99wt%。
此种造型体例如可通过通式(I)的酞菁,任选在粘合剂存在下,在5~50巴,优选10~20巴压力下压制来生产。
本发明同样也提供一种分散体,优选水分散体,含有通式(I)的金属络合物,其优选含量介于10~90wt%,以分散体为基准计。可能的分散剂例如是:基于丙烯酸酯、聚氨酯含长链聚氧乙烯化合物的聚合物分散剂。合适的产品的例子是:Solsperse 32000或Solsperse 38000,由Avecia出品。
本发明还提供了用通式(I)的酞菁涂布基材的方法。这优选地通过旋涂、溅射或真空蒸汽沉积来实施。通式(Ia)~(Ih)的酞菁可特别好地通过真空蒸汽沉积或溅射,特别是真空蒸汽沉积,来施涂。
采用溅射或真空蒸汽沉积施涂的此种涂层用的原料是通式(I)酞菁的所有上述形式,即,细粉末、细结晶形式或粒料、粒状固体制剂、固体造型体和分散体。后者尤其被用于将精细分散形式的酞菁施涂到某一表面,随后从该表面再通过溅射或真空蒸汽沉积施涂到基材上的场合。
这些操作优选使用大于50%,尤其优选大于85%,非常尤其优选大于90%,特别是大于95%或大于98%的酞菁纯度。
酞菁可彼此或者与其他具有类似光谱性质的染料进行混合。信息层不仅可包含酞菁,而且可包含添加剂,如粘合剂、润湿剂、稳定剂、稀释剂和增感剂以及其他成分。
本发明还提供一种用于将光吸收化合物的蒸汽沉积到生产光存储介质用的基材上的设备,其特征在于,染料可通过在低背景压力下加热而蒸发,并沉积到基材上。背景压力低于10-1Pa,优选低于10-3Pa,尤其优选低于10-4Pa。染料优选通过电阻加热或通过微波吸收加热。
特别是,本发明提供一种如上所述光数据载体,其中通式(I)的光吸收化合物连同,恰当的话,上述添加剂一起形成任选为无定形的信息层。就本发明目的而言,无定形是指,在光学显微镜下观察不到晶体并且在X-射线衍射图中观察不到布拉格反射,只能观察到无定形晕圈。
除了信息层之外,其他层如金属层、电介质层和保护层也可存在于光数据储存器中。金属和电介质层用于,特别是,调节反射率和热吸收/保持。视激光波长而定,金属可以是金、银、铝、合金等。电介质层的例子是二氧化硅和氮化硅。保护层例如是光固化表面涂层、粘合剂层和保护膜。
粘合剂层可以是压敏的。
压敏粘合剂层主要由丙烯酸类粘合剂组成。例如Nitto Denko DA-8320或DA-8310,公开在专利JP-A 11-273147中,可用于此目的。
光数据载体具有,例如,下列层结构(参见附图1):透明的基材(1)、任选的保护层(2),信息层(3)、任选的保护层(4)、任选的粘合剂层(5)、覆盖层(6)。
光数据载体的结构优选地:
√包含优选透明的基材(1),在基材表面上施涂了至少一个可用光,优选激光写入的可光写入信息层(3)、任选的保护层(4)、任选的粘合剂层(5)和透明覆盖层(6)。
√包含优选透明的基材(1),在基材表面上施涂了保护层(2)、至少一个可用光,优选激光写入的信息层(3)、任选的粘合剂层(5)和透明覆盖层(6)。
√包含优选透明的基材(1),在基材表面上施涂了保护层(2)和任选的至少一个可用光,优选激光写入的信息层(3)、任选的保护层(4)、任选的粘合剂层(5)和透明覆盖层(6)。
√包含优选透明的基材(1),在基材表面上施涂了至少一个可用光,优选激光写入的信息层(3)、任选的粘合剂层(5)和透明覆盖层(6)。
或者,光数据载体具有,例如,下列层结构(参见附图2):优选透明的基材(11)、信息层(12)、任选的反射层(13)、任选的粘合剂层(14)、另一优选透明的基材(15)。
或者,光数据载体具有,例如,下列层结构(参见附图3):优选透明的基材(21)、信息层(22)、任选的反射层(23)、保护层(24)。
替代地,该光数据载体的结构可
●包含多个信息层,中间优选地由适当层隔开。尤其优选的分隔层是光固化树脂、粘合剂层、电介质层或反射层。
本发明还提供了本发明光数据载体,其上已用蓝光,特别是激光,特别优选波长介于360~460nm的激光写入。
下面的实施例举例说明本发明。
实施例
实例施1
a)将4.75g 2-甲基-3,4-二乙基吡咯-5-甲酸叔丁酯和6.73g氢氧化钾粉末悬浮到40mL二甲亚砜中,并搅拌1h。随后滴加3.42g苄基溴,将溶液先在室温再搅拌1h,然后在70℃搅拌30min。悬浮液以50mL水稀释,以白色粉末形式沉淀出的下列通式
的产物被滤出并以水洗涤。干燥后获得5.5g(产率为84%)产物,熔点=75~77℃。
b)将5.43g来自a)的化合物和0.80g 3,4-二羟基-3-环丁烯-1,2-二酮在20mL乙醇中的溶液与0.5mL 37%浓度盐酸进行混合,混合物回流4h。除去溶剂后,残余物在乙醚中进行煮解,沉淀的固体过滤出来,并以乙醚洗涤。干燥后获得2.5g(产率为47%)下列通式绿色粉末
熔点=203℃(分解)
分子量532.73
λmax=590nm(二氯甲烷)
ε=167000l/mol cm
λ1/2~λ1/10(长波长侧翼)=21nm
Δλ=|λDMF-λ二噁烷|=2nm
实施例2
a)将4.19g 2-甲基-3,4-二乙基吡咯-5-甲酸乙酯和3.37g氢氧化钾粉末在30mL二甲亚砜中于80℃搅拌10min。冷却后,滴加6.14g 3,5-双三氟甲基苄基溴,将溶液先在室温搅拌1h,然后在70℃搅拌2h。溶液以200mL水稀释,以二氯甲烷萃取,然后产物以盐酸进行酸化而沉淀出来。过滤并以水洗涤,干燥后获得4.29g(产率为53%)下列通式的产物
,呈无色粉末状,熔点=126~128℃。
b)将4.07g来自a)的吡咯化合物和0.57g 3,4-二羟基-3-环丁烯-1,2-二酮用于类似于实例1b的方法中,从而获得了3.29g(产率为82%)下列通式绿色粉末
熔点=185℃(分解)
分子量=804.73
λmax=590nm(二氯甲烷)
ε=184652l/mol cm
λ1/2~λ1/10(长波长侧翼)=19nm
实施例3
a)采用类似于实例2a的方法,将1.90g 2,4-二甲基吡咯、2.24g氢氧化钾粉末和4.78g 4-三氟甲基苄基溴在20mL二甲亚砜中进行反应。反应完成后,溶液以300mL水稀释,产物以二氯甲烷萃取。将有机相用硫酸钠干燥并过滤。除去溶剂后,获得4.8g(产率为95%)产物,呈褐色树脂状,具有通式
b)将1.77g来自a)的产物和0.4g 3,4-二羟基-3-环丁烯-1,2-二酮按照类似于实例1b的方法进行反应。冷却后,产物在400mL二异丙基醚中进行沉淀,过滤后获得0.2g产物
滤液进行蒸发并在二氧化硅上以二氯甲烷为洗脱剂进行色谱处理。在除去溶剂后,第一紫色级分进一步产出0.72g产物。总收率:0.92g(产率为45%)。
熔点=220℃(分解)
分子量584.57
λmax=577nm(二氯甲烷)
ε=139000l/mol cm
λ1/2~λ1/10(长波长侧翼)=20nm
实施例4
a)将4.19g 2-甲基-3,4-二乙基吡咯-5-甲酸乙酯和3.37g氢氧化钾粉末在20mL二甲亚砜中于80℃搅拌10min。冷却后,滴加4.78g 2-三氟甲基苄基溴,将溶液先在室温再搅拌1h,然后在70℃搅拌3h。溶液以300mL水稀释,以二氯甲烷萃取,然后产物以盐酸进行酸化而沉淀出来。过滤并以水洗涤,干燥后获得3.80g(产率为56%)下列通式的产物
,呈无色粉末状,熔点=124~126℃。
b)将3.39g来自a)的化合物和0.57g 3,4-二羟基-3-环丁烯-1,2-二酮在20mL乙醇中与0.5mL 37%浓度盐酸混合,化合物回流4h。冷却后,沉淀的固体经过滤,以二异丙基醚洗涤。干燥后获得2.58g(产率为77%)绿色粉末,具有通式
熔点=206℃(分解)
分子量668.73
λmax=588nm(丙酮)
ε=202000l/mol cm
λ1/2~λ1/10(长波长侧翼)=22nm
Δλ=|λDMF-λ二噁烷|=1nm
另外的合适的方酸类染料示于表格中。它们是通过类似的组分和方酸类染料的制备方法制取的。
1)除非另外指出,在丙酮中
2)在长波长侧翼
3)Δλ=|λDMF-λ二噁烷|
实施例20
将染料酞菁二溴化锗(GeBr2Pc)在高真空(压力p近似于2×10-5mbar)下从电阻加热的钼船中以约5埃/s的速率蒸汽沉积到预制沟纹的聚碳酸酯基材上。层厚为约55nm。预制沟纹的聚碳酸酯基材是通过注塑制造的盘片。盘片的直径是120mm,其厚度是0.6mm。在注塑方法中产生的沟纹结构具有约1μm的道间距,沟纹的深度和沟纹半高宽度分别为约150nm和约260nm。带有作为信息载体的染料层的盘片通过蒸汽沉积被镀以100nm Ag。随后,通过旋涂施加一层紫外固化丙烯酸酯涂层并利用紫外光灯固化。盘片利用安装在光学试验台上的动态写入试验装置进行试验,包括用以发生线性偏振激光的GaN二极管激光器(λ=405nm)、偏振-敏感的射束分离器、λ/4板和移动悬吊式采集镜头,具有数值孔径NA=0.65(执行器镜头)。从盘片反射的光借助上述偏振-敏感射束分离器被从光束路径中取出并借助象散透镜聚焦到四象限检测器上。在线速度V=5.00m/s和写入功率Pw=13mW条件下,测得信噪比C/N=41dB。写入功率是以脉冲顺序的形式施加的,其间盘片被交替地用先上述写入功率Pw辐照1μs,随后用读出功率Pr=0.44mW辐照4μs。盘片以此种脉冲顺序辐照,直至它完成一周的旋转。随后,按此方式产生的标记以读出功率=0.44mW读出,并测定上述信噪比C/N。
实施例21
9.95g下列通式的酞菁二氯化锡
在125mL吡啶中的溶液在室温与4.9g四氢硼酸钠进行混合。混合物在回流(115℃)下搅拌75min,并慢慢冷却到90℃。在此温度,慢慢滴加100mL水。随后,混合物再回流30min,冷却至室温,然后抽滤。滤饼以200mL甲醇搅拌,抽滤并以甲醇洗涤至洗涤水变清。以50mL水洗涤后,将其在30℃、减压下干燥。于是,获得4.03g(产率为63%)下列通式的蓝色粉末
将1.6g该产物在32mL预先在分子筛上干燥过的氯萘中,在室温下与0.2mL溴在10mL氯萘中的溶液在搅拌下混合。随后,混合物在70~75℃搅拌1h,温度很快升高到90℃。冷却至室温后,混合物进行抽滤,以甲苯洗涤,直至洗涤水变清,随后以甲醇洗涤至洗涤液变为基本上无色,然后在30℃、减压下干燥。于是,获得1.46g(产率为74%)
下列通式的蓝色粉末
Claims (21)
2.权利要求1的化合物,其特征在于,R是取代或未取代的吡咯。
4.权利要求3的化合物,其特征在于,
R1是氢、甲基、乙基、丙基、丁基、环己基甲基、苄基、4-甲氧基苄基、4-三氟甲基苄基、3-三氟甲基苄基、2-三氟甲基苄基、3,5-双(三氟甲基)苄基或4-氟-2-三氟甲基苄基,
R2是甲基、乙基、丙基或苯基,且
R3和R4彼此独立地是氢、甲基、乙基、丙基、丁基、苯基、乙酰基或乙氧羰基。
5.权利要求3的化合物,其特征在于,
R1是4-三氟甲基苄基或3,5-双(三氟甲基)苄基,特别是4-三氟甲基苄基,
R2是甲基、乙基或苯基,特别是甲基,
R3是氢、乙基、乙酰基或乙氧羰基,特别是乙基,且
R4是甲基、乙基或苯基,特别是甲基或乙基。
7.权利要求1的方酸类化合物作为光吸收化合物在一次写入光数据载体的信息层中的应用。
8.权利要求7的应用,其特征在于,光数据载体可用红激光,特别是波长介于600~680nm的激光写入和读出。
10.权利要求9的吡咯化合物,其特征在于,
R1是丙基、丁基、环己基甲基、苄基、4-甲氧基苄基、4-三氟甲基苄基、3-三氟甲基苄基、2-三氟甲基苄基、3,5-双(三氟甲基)苄基或4-氟-2-三氟甲基苄基,
R2是甲基、乙基、丙基或苯基,
R3和R4彼此独立地是氢、甲基、乙基、丙基、丁基、苯基、乙酰基或乙氧羰基,且
R5是氢、叔丁氧羰基或羧基。
12.包含优选透明的基材的光数据载体,基材表面施涂了可光写入信息层、优选一个或多个反射层以及另外的基材或保护层,其可用红光,优选激光写入和读出,其中信息层包含光吸收化合物,其特征在于,至少一种权利要求1~5至少之一的方酸类化合物被用作光吸收化合物。
13.生产权利要求12的光数据载体的方法,其特征在于,优选透明的基材被涂以权利要求1的方酸类化合物、任选地连同适当粘合剂和添加剂以及任选的适当溶剂,并且任选提供有反射层、另外的中间层,以及任选的保护层或另外的基材。
14.权利要求12的光数据载体,其中所述光数据载体已用红光,特别是红激光写入。
15.包含优选透明的基材的光数据载体,基材可任选具有预先涂布的一个或多个反射层并且其表面上涂布了可光写入信息层、任选的一个或多个反射层,以及任选的保护层或另外的基材或覆盖层,该光数据载体可用蓝光,优选激光,特别优选波长介于360~460nm,特别是380~420nm,非常特别优选390~410nm的光,或者用红外光,优选激光,特别优选波长介于760~830nm的光写入和读出,其中信息层包含光吸收化合物以及任选的粘合剂,其特征在于,采用至少一种通式(I)的酞菁作为光吸收化合物
其中
Me是双轴向取代的金属原子,选自Si、Ge和Sn,
Pc是未取代的酞菁,且
X1和X2彼此独立地是溴或碘,且X1也可以是氯。
19.权利要求17或18的应用,其特征在于,所用酞菁的纯度大于90wt%,特别是大于95wt%,尤其优选大于98wt%,以通式(I)的酞菁为基准计。
21.权利要求15的光数据载体,其中所述光数据载体已用蓝光,特别是激光,尤其优选波长介于360~460nm的激光写入。
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10212199 | 2002-03-19 | ||
| DE10212199.0 | 2002-03-19 | ||
| DE10245581.3 | 2002-09-27 | ||
| DE10245581A DE10245581A1 (de) | 2002-03-19 | 2002-09-27 | Squaryliumfarbstoffe als lichtabsorbierende Verbindung in der Informationsschicht von optischen Datenträgern |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN1643587A true CN1643587A (zh) | 2005-07-20 |
Family
ID=28042851
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA038064146A Pending CN1643587A (zh) | 2002-03-19 | 2003-03-18 | 在光数据载体信息层中作为光吸收化合物的方酸类染料 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20050142489A1 (zh) |
| EP (1) | EP1488418A1 (zh) |
| JP (1) | JP2005520835A (zh) |
| CN (1) | CN1643587A (zh) |
| AU (1) | AU2003226651A1 (zh) |
| WO (1) | WO2003079339A1 (zh) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101544843B (zh) * | 2008-03-26 | 2013-05-01 | 富士施乐株式会社 | 着色剂 |
| TWI691565B (zh) * | 2017-11-29 | 2020-04-21 | 日商日東電工股份有限公司 | 附黏著劑層之偏光薄膜及液晶顯示裝置 |
| CN111417875A (zh) * | 2017-11-29 | 2020-07-14 | 日东电工株式会社 | 色彩校正构件及使用色彩校正构件的光学膜 |
| CN112470045A (zh) * | 2018-07-23 | 2021-03-09 | 住友化学株式会社 | 滤光片及显示装置 |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007526835A (ja) * | 2003-06-27 | 2007-09-20 | チバ スペシャルティ ケミカルズ ホールディング インコーポレーテッド | 高記憶密度を有する光学記録材料 |
| JP2008501548A (ja) * | 2004-06-03 | 2008-01-24 | クラリアント・インターナシヨナル・リミテツド | 光学データ記録用光学層におけるスクアリン酸色素の使用 |
| JP5286726B2 (ja) * | 2007-09-25 | 2013-09-11 | 富士ゼロックス株式会社 | 黒色色材及びトナー |
| US9353092B2 (en) | 2013-06-27 | 2016-05-31 | University Of Notre Dame Du Lac | Synthesis and use of croconaine compounds |
| JP6638948B2 (ja) * | 2014-10-20 | 2020-02-05 | 国立大学法人愛媛大学 | 新規オキソカーボン系化合物 |
| JP6506529B2 (ja) * | 2014-10-20 | 2019-04-24 | 株式会社日本触媒 | オキソカーボン系化合物を含む樹脂組成物及びこれからなる成形体 |
| KR20210034554A (ko) * | 2018-07-23 | 2021-03-30 | 스미또모 가가꾸 가부시키가이샤 | 화합물 및 그 제조 방법 |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1154745A (en) * | 1966-05-17 | 1969-06-11 | Whitefin Holding Sa | Haloketones |
| DE2055894A1 (en) * | 1970-11-13 | 1972-05-18 | Badische Anilin- & Soda-Fabrik Ag, 6700 Ludwigshafen | Dyes from quadrate acid and pyrazolones - for colouring synthetic fri esp metal-modified polypropylene |
| JPS54128567A (en) * | 1978-03-28 | 1979-10-05 | Teijin Ltd | 1-substituted-4-acyl-2-pyrrolecarboxylic acid derivative |
| US4256759A (en) * | 1979-06-11 | 1981-03-17 | Ciba-Geigy Corporation | Alphacarbamoyl-pyrrolpropionitriles |
| GR69239B (zh) * | 1979-06-11 | 1982-05-11 | Ciba Geigy | |
| JPS5856239A (ja) * | 1981-09-28 | 1983-04-02 | Tdk Corp | 光記録媒体 |
| DE3531504A1 (de) * | 1985-09-04 | 1987-03-12 | Basf Ag | 3-aminomethylpyrrol-1-yl-alkylamine und diese verbindungen enthaltende therapeutische mittel |
| US4743531A (en) * | 1986-11-21 | 1988-05-10 | Eastman Kodak Company | Dye sensitized photographic imaging system |
| AU625688B2 (en) * | 1989-01-31 | 1992-07-16 | Merrell Dow Pharmaceuticals Inc. | Method of using aryl- or heteroaryl-1-alkyl-pyrrole-2- carboxylic acid compounds in the treatment of interleukin-1 mediated conditions |
| US5039695A (en) * | 1989-01-31 | 1991-08-13 | Merrell Dow Pharmaceuticals Inc. | Method of using aryl-or heteroaryl-1-alkyl-pyrrole-2-carboxylic acid compounds in the treatment of interleukin-1 mediated conditions |
| DD294962A5 (de) * | 1990-06-05 | 1991-10-17 | Th "Carl Schorlemmer",De | Verfahren zur herstellung neuer thiophenhaltiger quadratsaeurefarbstoffe |
| DE4122563C2 (de) * | 1991-07-08 | 1994-12-08 | Syntec Ges Fuer Chemie Und Tec | Neue thiazolsubstituierte Quadratsäurefarbstoffe und Verfahren zu ihrer Herstellung |
| DE4215826A1 (de) * | 1992-05-09 | 1993-11-11 | Tech Hochschule C Schorlemmer | Selenazol- und selenophensubstituierte Quadratsäurefarbstoffe und Verfahren zu ihrer Herstellung |
| GB9513114D0 (en) * | 1995-06-28 | 1995-08-30 | Kodak Ltd | Novel image-dye-forming couplers and photographic elements containing them |
| WO1997008692A1 (en) * | 1995-08-30 | 1997-03-06 | Akzo Nobel N.V. | Optical recording medium containing a thiophene squarilium or croconium dye |
| JPH1158953A (ja) * | 1997-08-19 | 1999-03-02 | Matsushita Electric Ind Co Ltd | フタロシアニン昇華性材料及びそれを用いた光記録媒体、並びにその光記録再生方法 |
| JP4193285B2 (ja) * | 1999-06-04 | 2008-12-10 | コニカミノルタホールディングス株式会社 | カラートナー |
| ATE248833T1 (de) * | 1999-12-16 | 2003-09-15 | Kyowa Hakko Kogyo Kk | Squarylium-verbindung und diese enthaltendes optisches aufzeichungsmedium |
| US6558768B2 (en) * | 2000-03-07 | 2003-05-06 | Ricoh Company, Ltd. | Optical recording medium and optical recording and reading method using the same |
| EP1377977A1 (en) * | 2001-03-28 | 2004-01-07 | Bayer Chemicals AG | Optical recording medium |
| EP1377975A2 (en) * | 2001-03-28 | 2004-01-07 | Bayer Chemicals AG | Optical data medium containing, in the information layer, a dye as a lightabsorbing compound and having a protective covering layer of predetermined thickness |
| JP2004524201A (ja) * | 2001-03-28 | 2004-08-12 | バイエル アクチェンゲゼルシャフト | 情報層中に吸光性化合物としてジアザヘミシアニン色素を含有する光学データ記憶媒体 |
| US6896945B2 (en) * | 2001-08-22 | 2005-05-24 | Bayer Aktiengesellschaft | Optical data carrier comprising a phthalocyanine dye as light-absorbent compound in the information layer |
| EP1435094A2 (en) * | 2001-10-04 | 2004-07-07 | Bayer Chemicals AG | Optical data medium containing; in the information layer, a dye as a light-absorbing compound |
-
2003
- 2003-03-18 AU AU2003226651A patent/AU2003226651A1/en not_active Abandoned
- 2003-03-18 WO PCT/EP2003/002789 patent/WO2003079339A1/de not_active Application Discontinuation
- 2003-03-18 US US10/508,417 patent/US20050142489A1/en not_active Abandoned
- 2003-03-18 JP JP2003577251A patent/JP2005520835A/ja not_active Withdrawn
- 2003-03-18 EP EP03744371A patent/EP1488418A1/de not_active Withdrawn
- 2003-03-18 CN CNA038064146A patent/CN1643587A/zh active Pending
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101544843B (zh) * | 2008-03-26 | 2013-05-01 | 富士施乐株式会社 | 着色剂 |
| TWI691565B (zh) * | 2017-11-29 | 2020-04-21 | 日商日東電工股份有限公司 | 附黏著劑層之偏光薄膜及液晶顯示裝置 |
| CN111417875A (zh) * | 2017-11-29 | 2020-07-14 | 日东电工株式会社 | 色彩校正构件及使用色彩校正构件的光学膜 |
| CN111480099A (zh) * | 2017-11-29 | 2020-07-31 | 日东电工株式会社 | 带粘合剂层的偏振膜及液晶显示设备 |
| CN111699220A (zh) * | 2017-11-29 | 2020-09-22 | 日东电工株式会社 | 用于显示装置的方酸内鎓盐化合物 |
| CN111699220B (zh) * | 2017-11-29 | 2022-08-23 | 日东电工株式会社 | 用于显示装置的方酸内鎓盐化合物 |
| CN112470045A (zh) * | 2018-07-23 | 2021-03-09 | 住友化学株式会社 | 滤光片及显示装置 |
| CN112470045B (zh) * | 2018-07-23 | 2023-08-08 | 住友化学株式会社 | 滤光片及显示装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2005520835A (ja) | 2005-07-14 |
| US20050142489A1 (en) | 2005-06-30 |
| AU2003226651A1 (en) | 2003-09-29 |
| EP1488418A1 (de) | 2004-12-22 |
| WO2003079339A1 (de) | 2003-09-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1434771A (zh) | 光记录介质和氮杂卟啉化合物 | |
| CN1153761C (zh) | 铵盐或二亚铵盐化合物及其用途 | |
| CN1175058A (zh) | 光学记录介质 | |
| CN1643587A (zh) | 在光数据载体信息层中作为光吸收化合物的方酸类染料 | |
| CN1287369C (zh) | 在信息层中含有具有多个发色中心的光吸收化合物的光学数据载体 | |
| US20030064192A1 (en) | Optical data carrier comprising a phthalocyanine dye as light-absorbent compound in the information layer | |
| CN1656059A (zh) | 二亚胺盐混合物铵盐混合物及其用途 | |
| CN1193363C (zh) | 光记录媒体 | |
| CN1910179A (zh) | 在光学数据载体的信息层中用作吸光性化合物的金属配合物 | |
| CN1659640A (zh) | 在信息层中含有作为光吸收化合物的染料的光数据载体 | |
| CN1575236A (zh) | 酰亚胺化合物及使用该化合物的光记录媒体 | |
| CN1197067C (zh) | 光记录介质和类卟吩化合物 | |
| CN1406182A (zh) | 苯并双吡咯基化合物,光记录介质用化合物 | |
| CN1751101A (zh) | 在光数据载体信息层中的作为光吸收化合物的金属配合物 | |
| CN1304486C (zh) | 酞菁化合物,制备该酞菁化合物的方法以及含有该酞菁化合物的光记录介质 | |
| CN1195298C (zh) | 光信息记录介质 | |
| CN1261308A (zh) | 光记录媒体及其制作方法 | |
| CN1609973A (zh) | 光信息记录介质和信息记录方法以及色素化合物 | |
| CN100343340C (zh) | 偶氮金属螯合染料及光学记录媒体 | |
| CN1503827A (zh) | 作为光记录介质的金属茂基酞菁 | |
| CN1922028A (zh) | 光记录材料和光记录介质 | |
| CN1902057A (zh) | 光学记录介质和染料 | |
| CN1495747A (zh) | 光记录介质 | |
| CN101052533A (zh) | 光学信息记录介质和信息记录方法 | |
| CN1513172A (zh) | 在信息层中包含作为吸光化合物的二氮杂半花青染料的光学数据存储介质 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1080983 Country of ref document: HK |
|
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: WD Ref document number: 1080983 Country of ref document: HK |