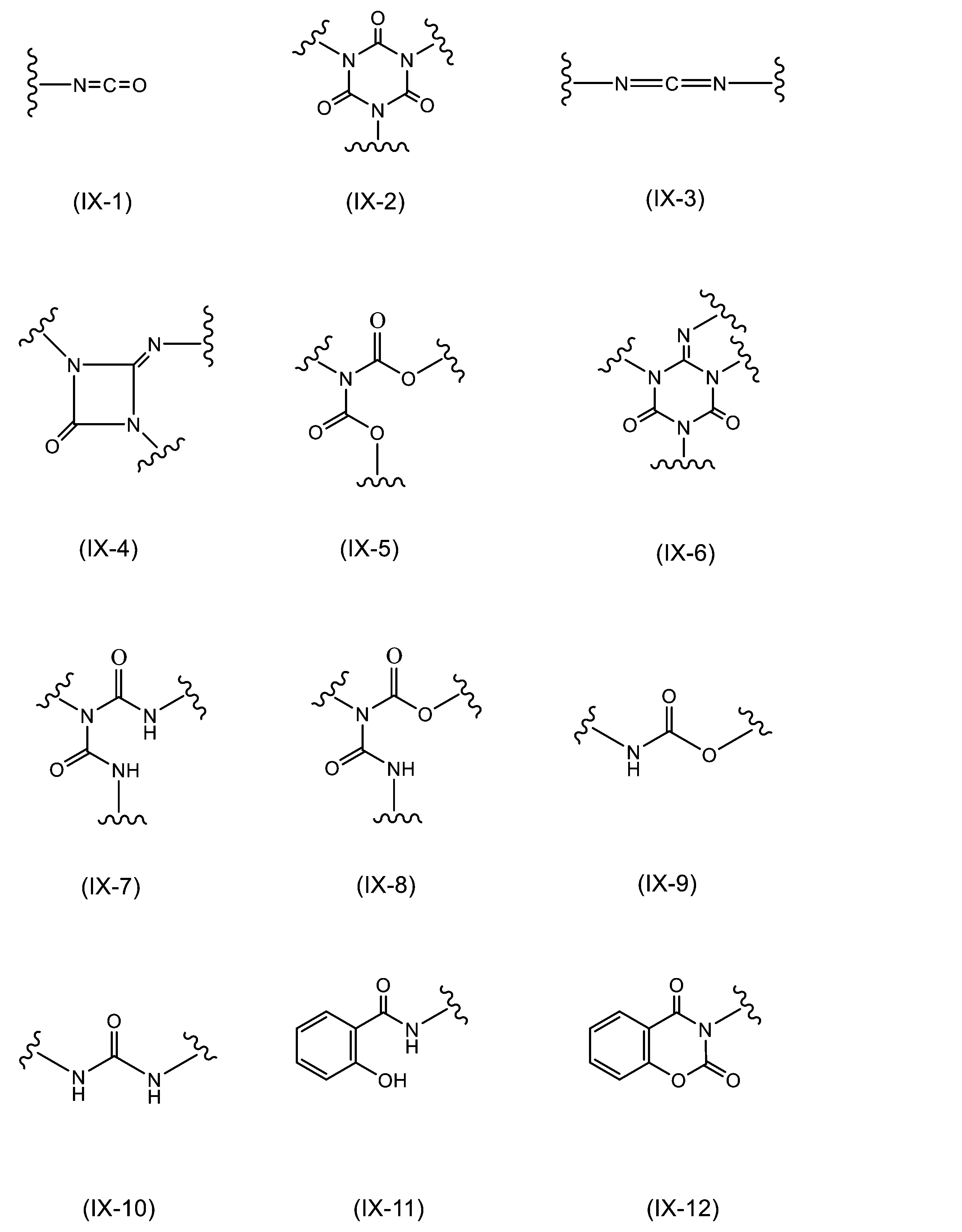
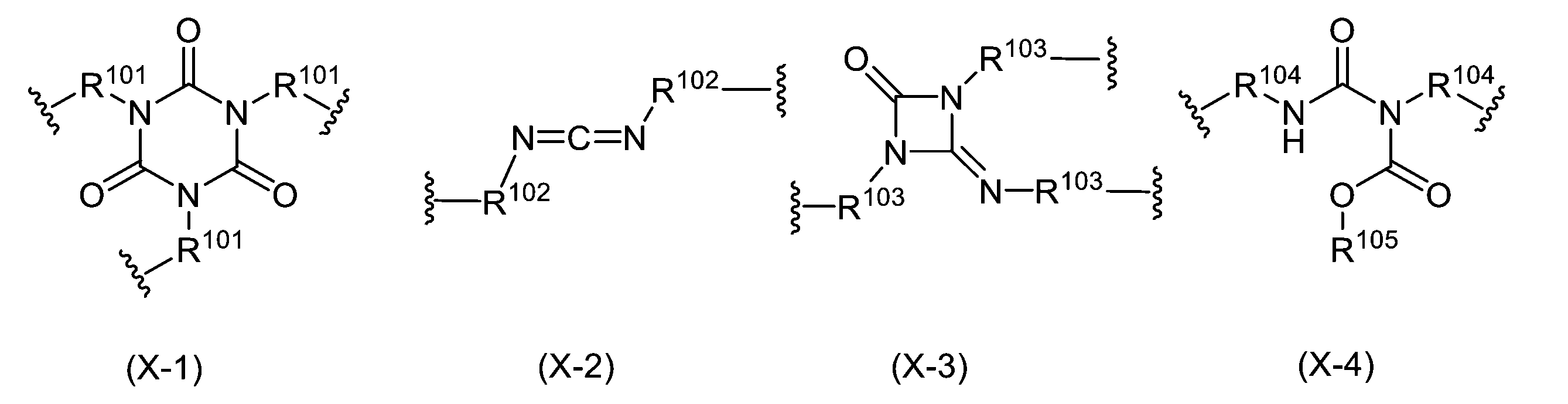
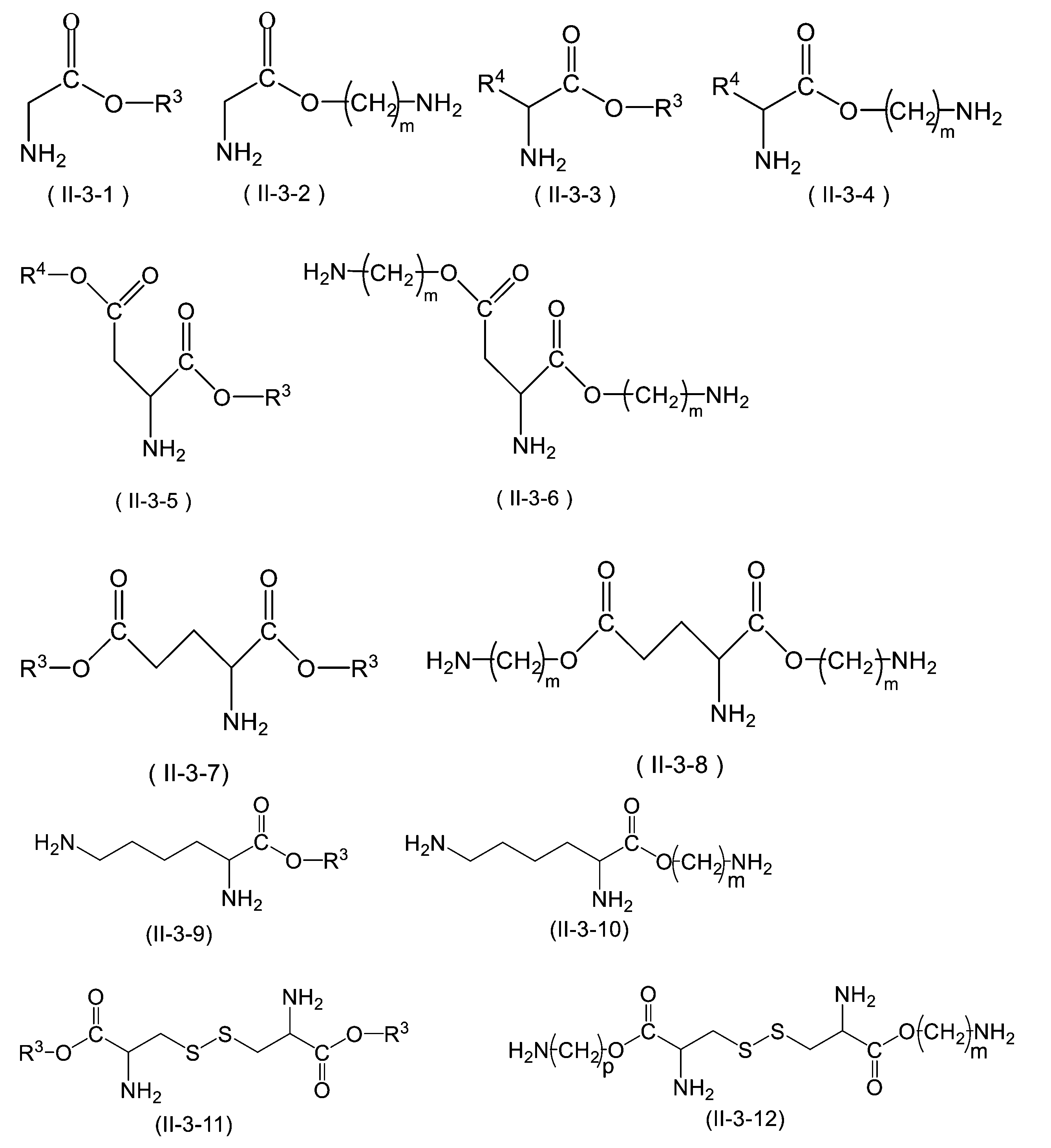
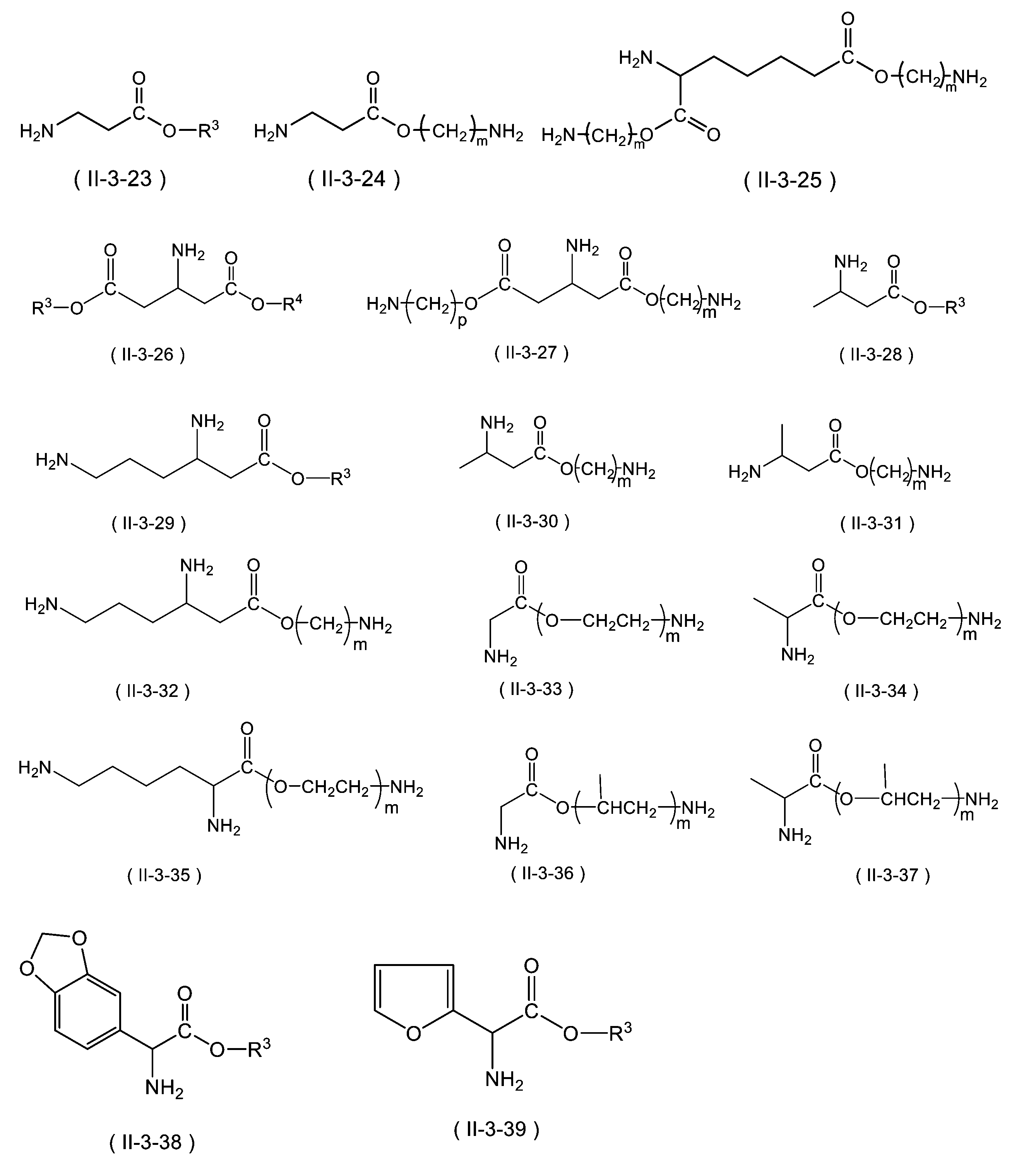
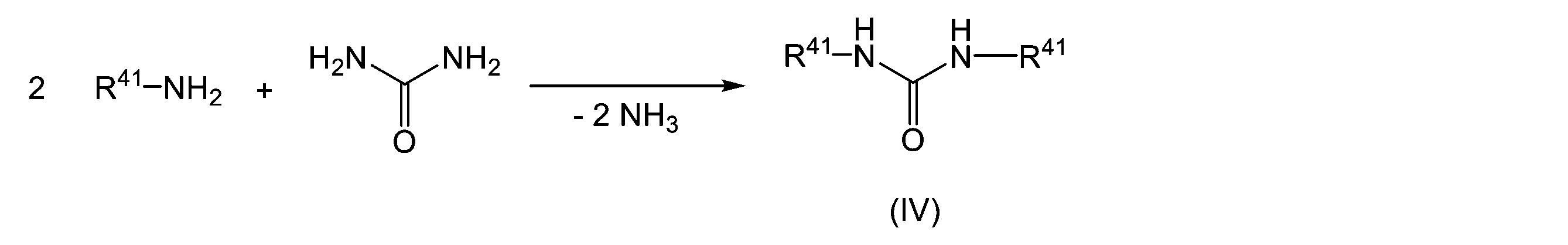
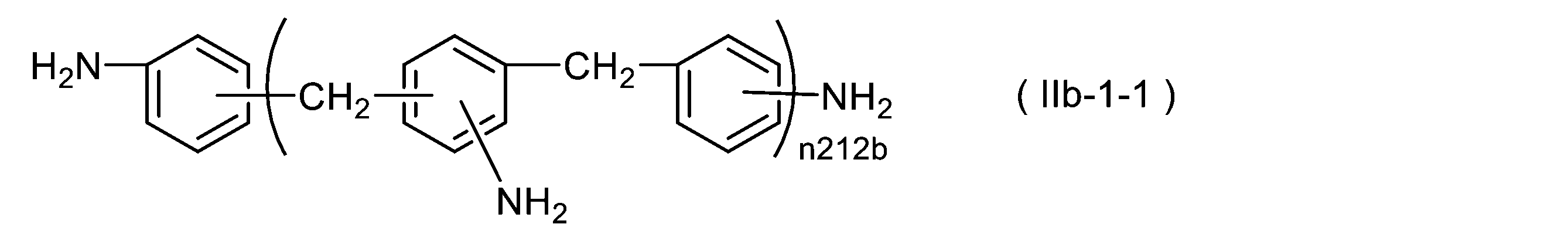
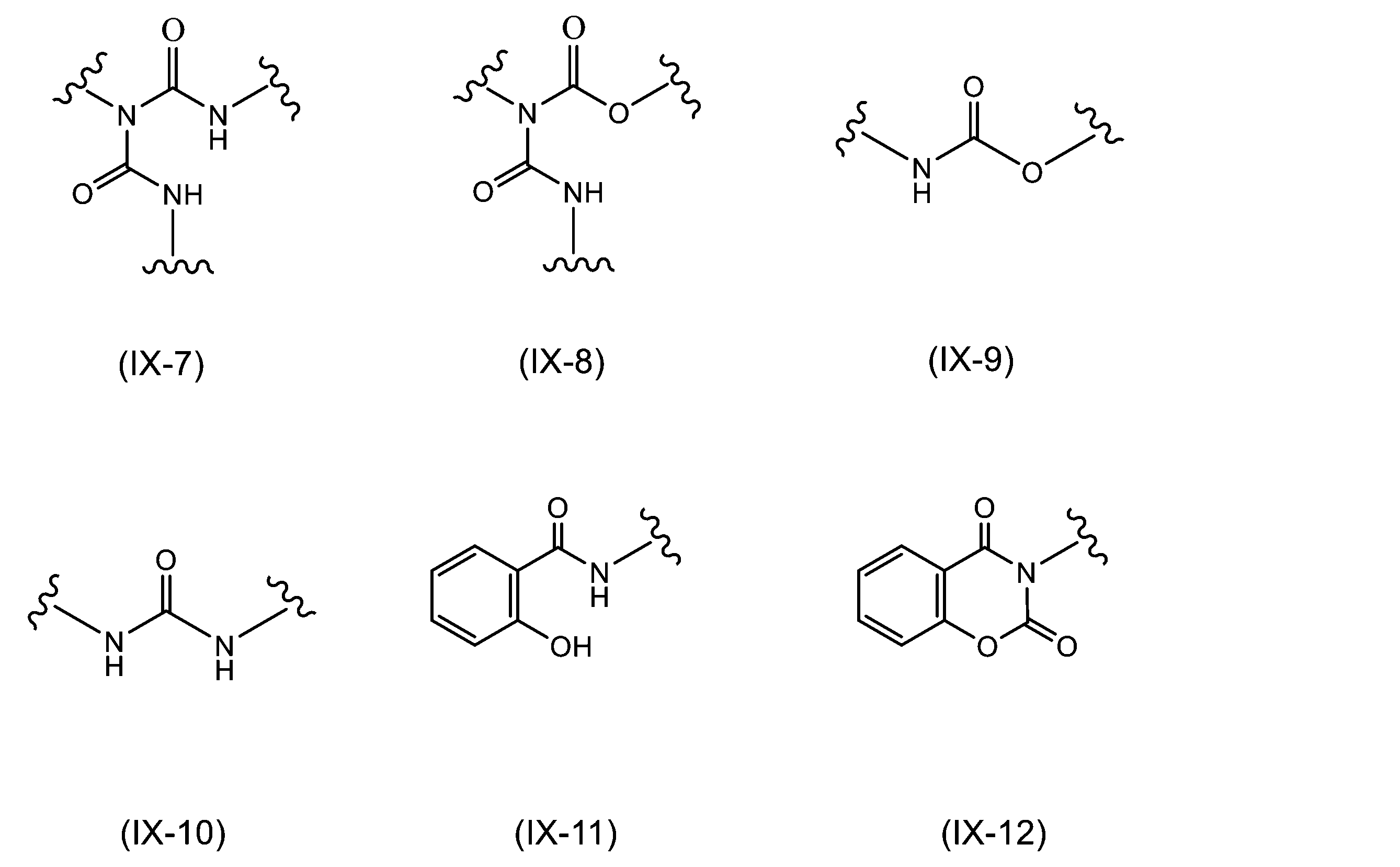
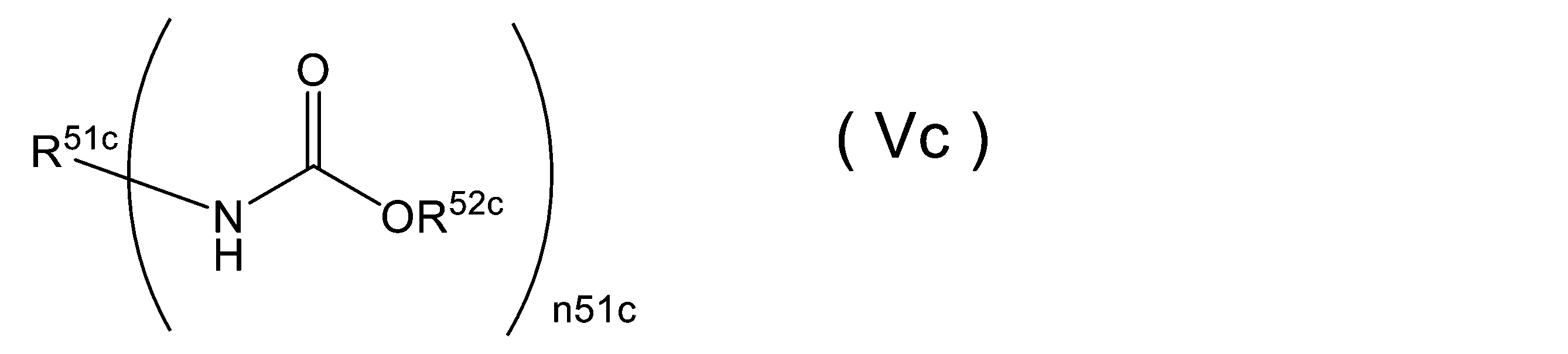
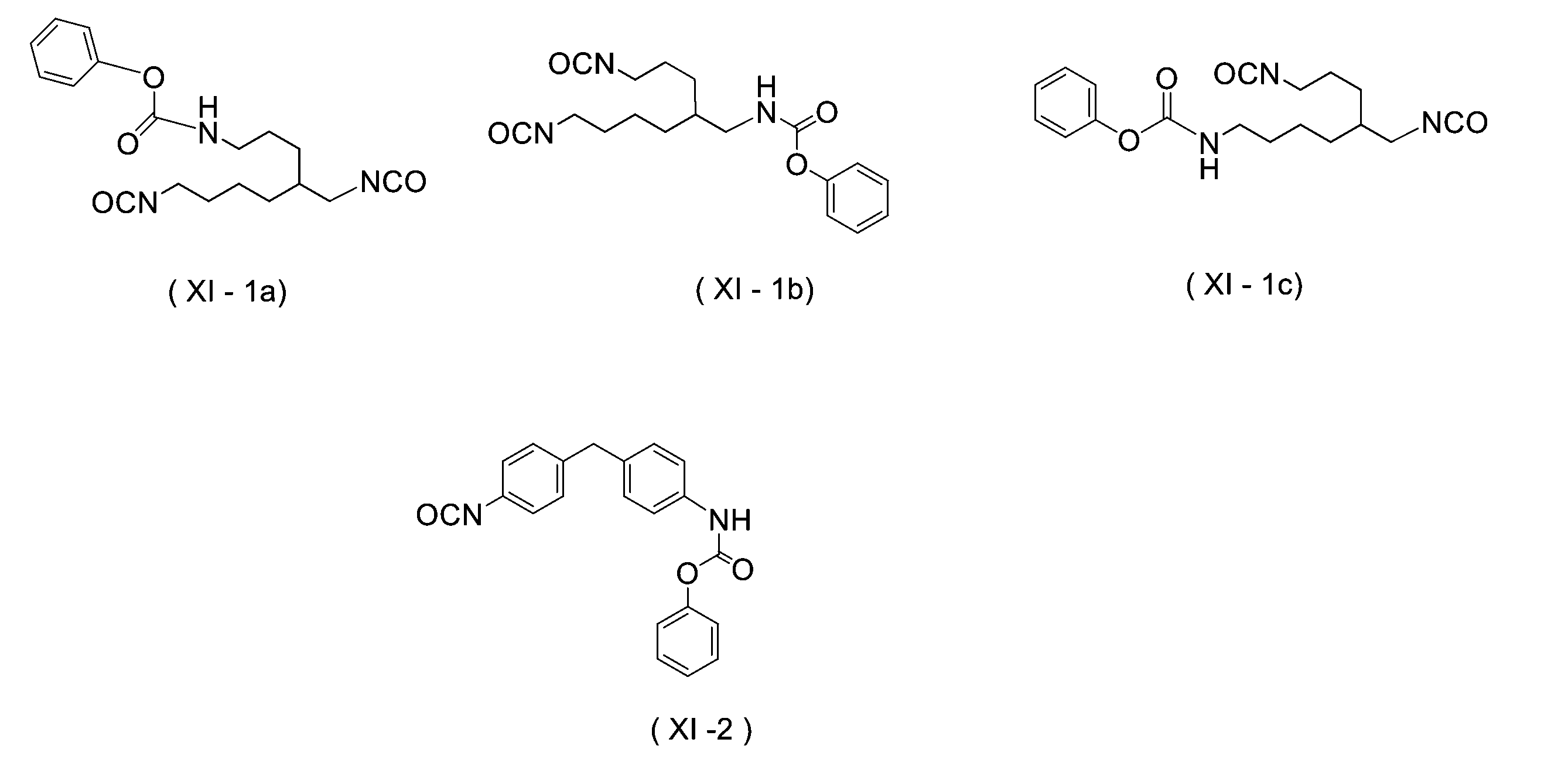
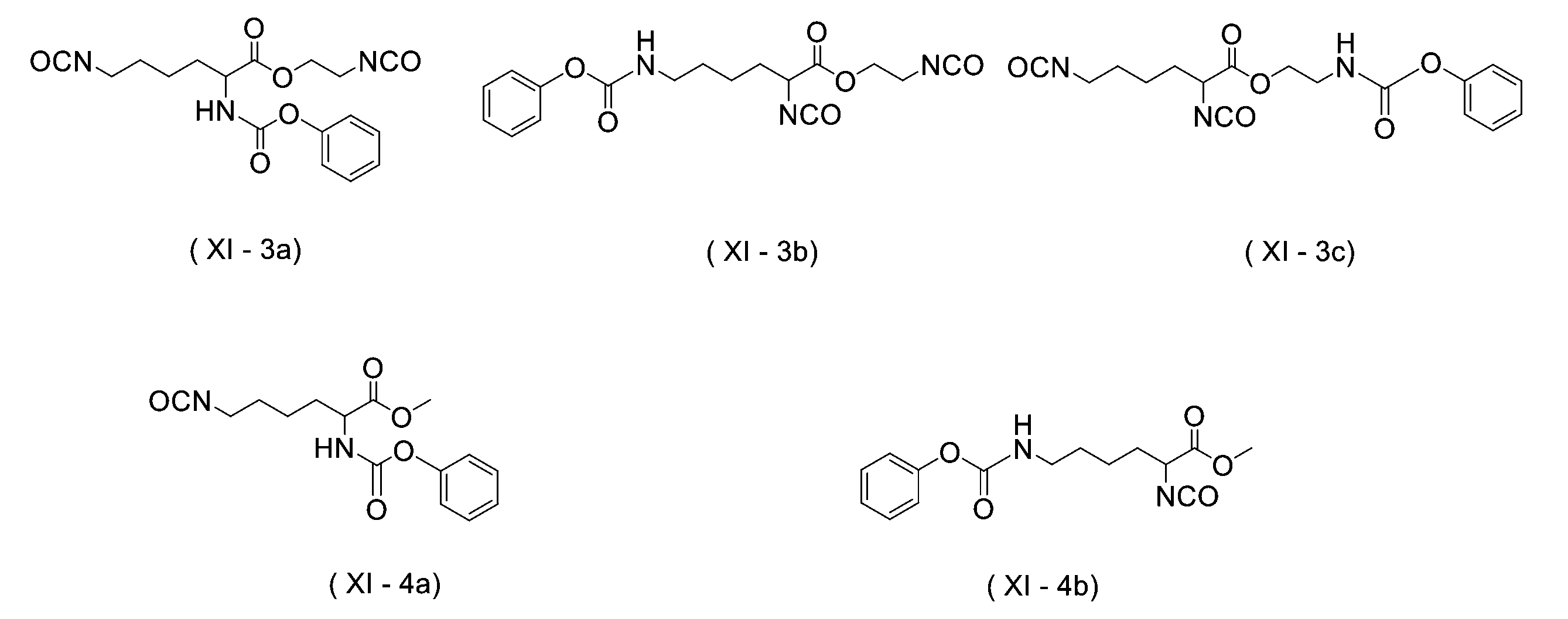
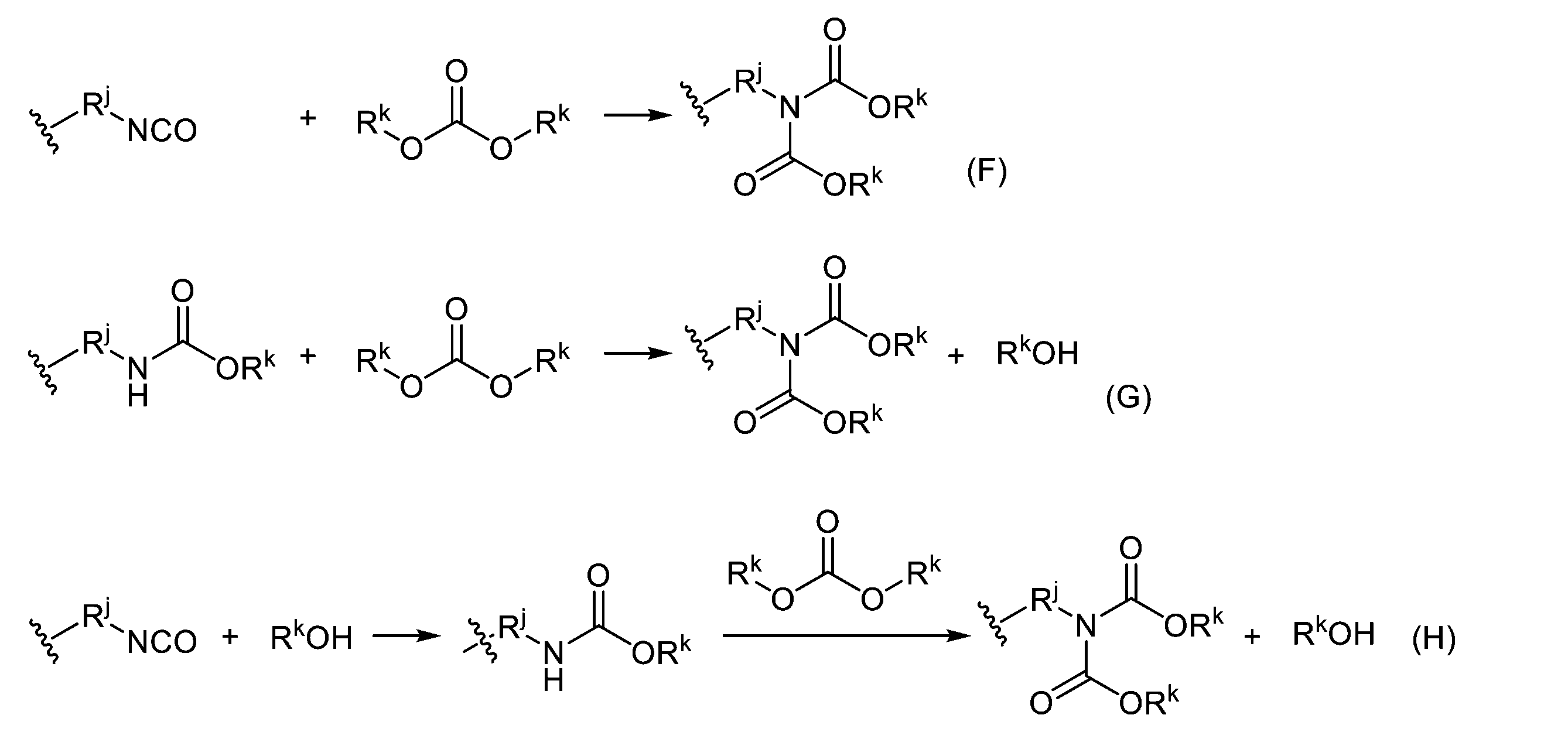
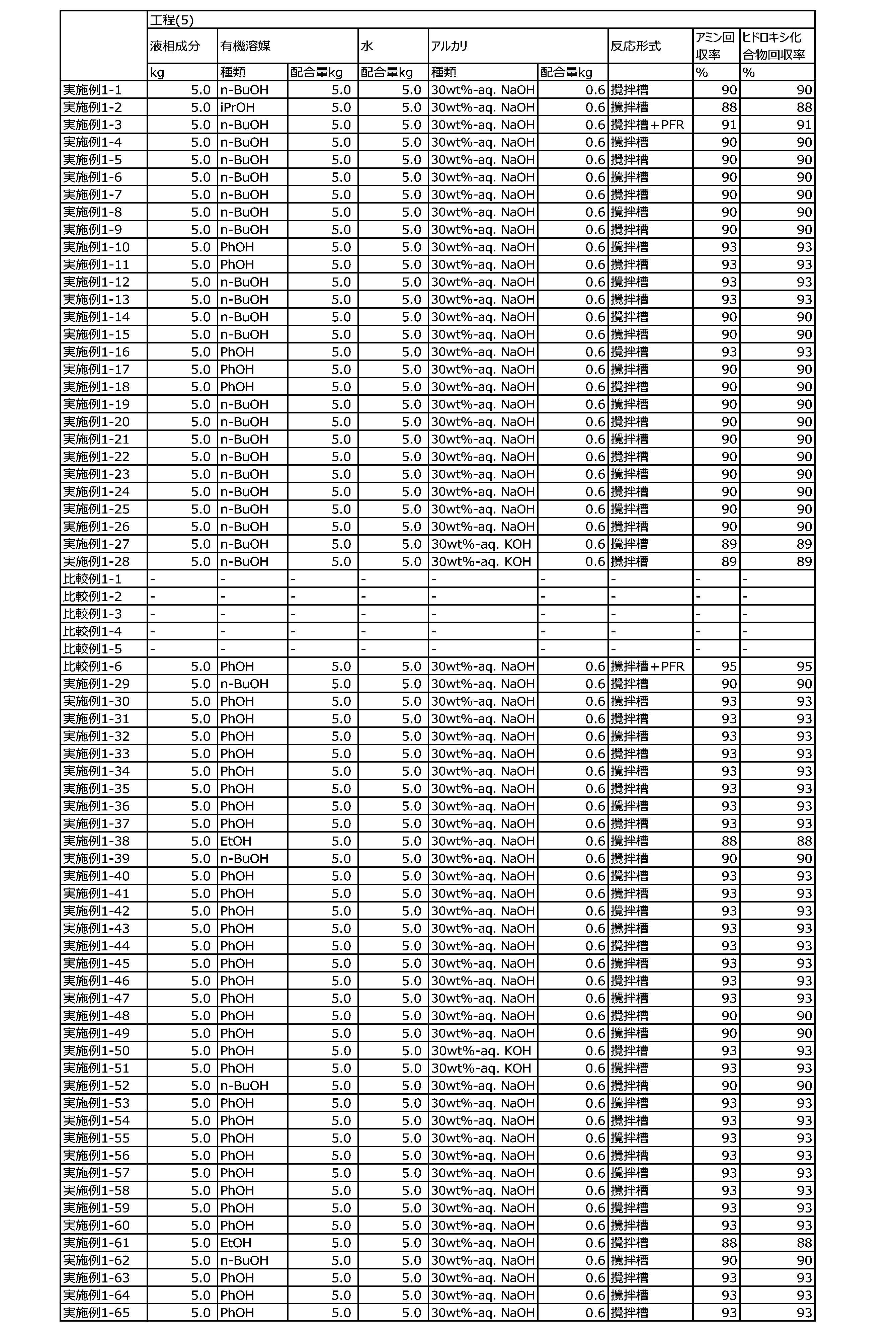
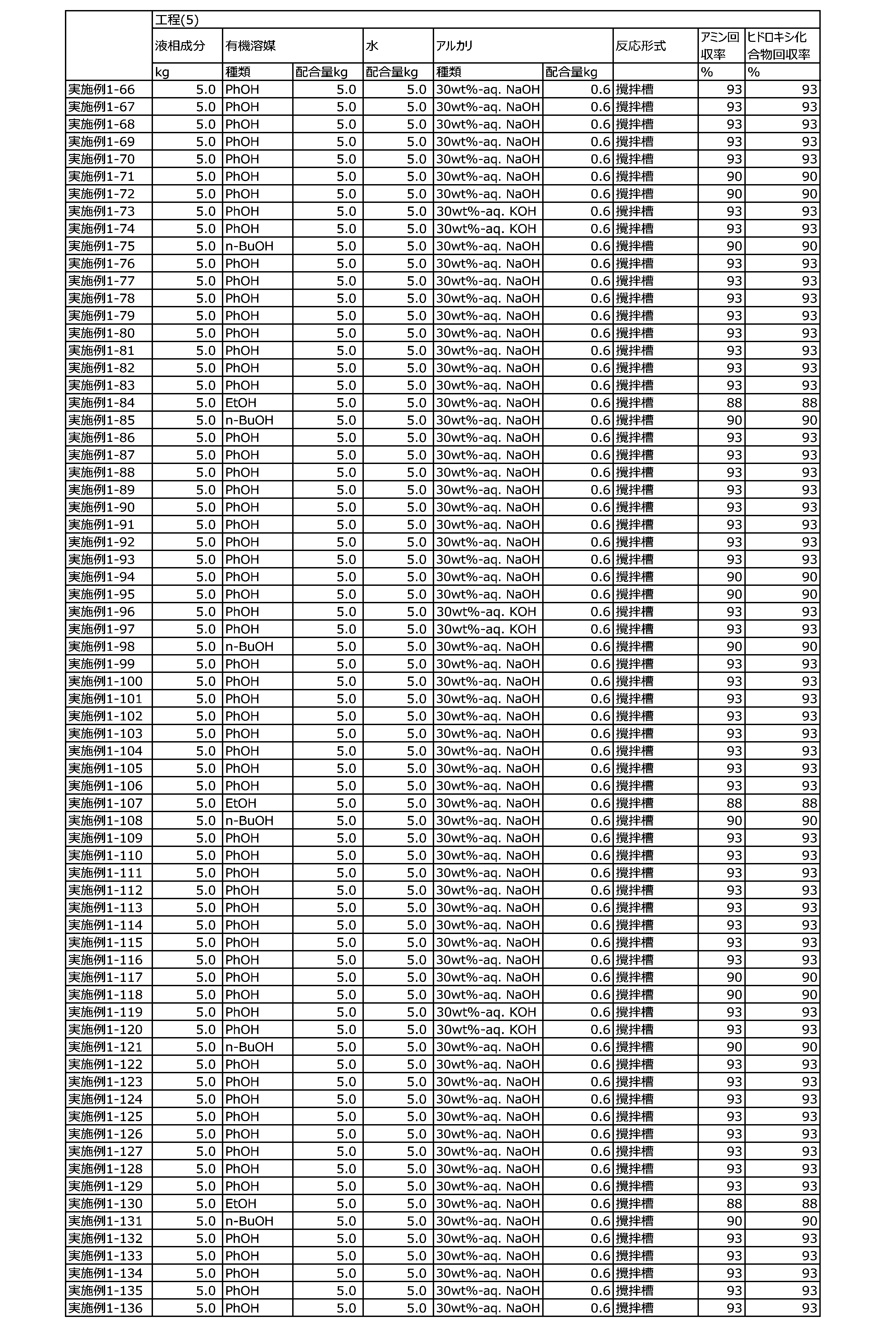
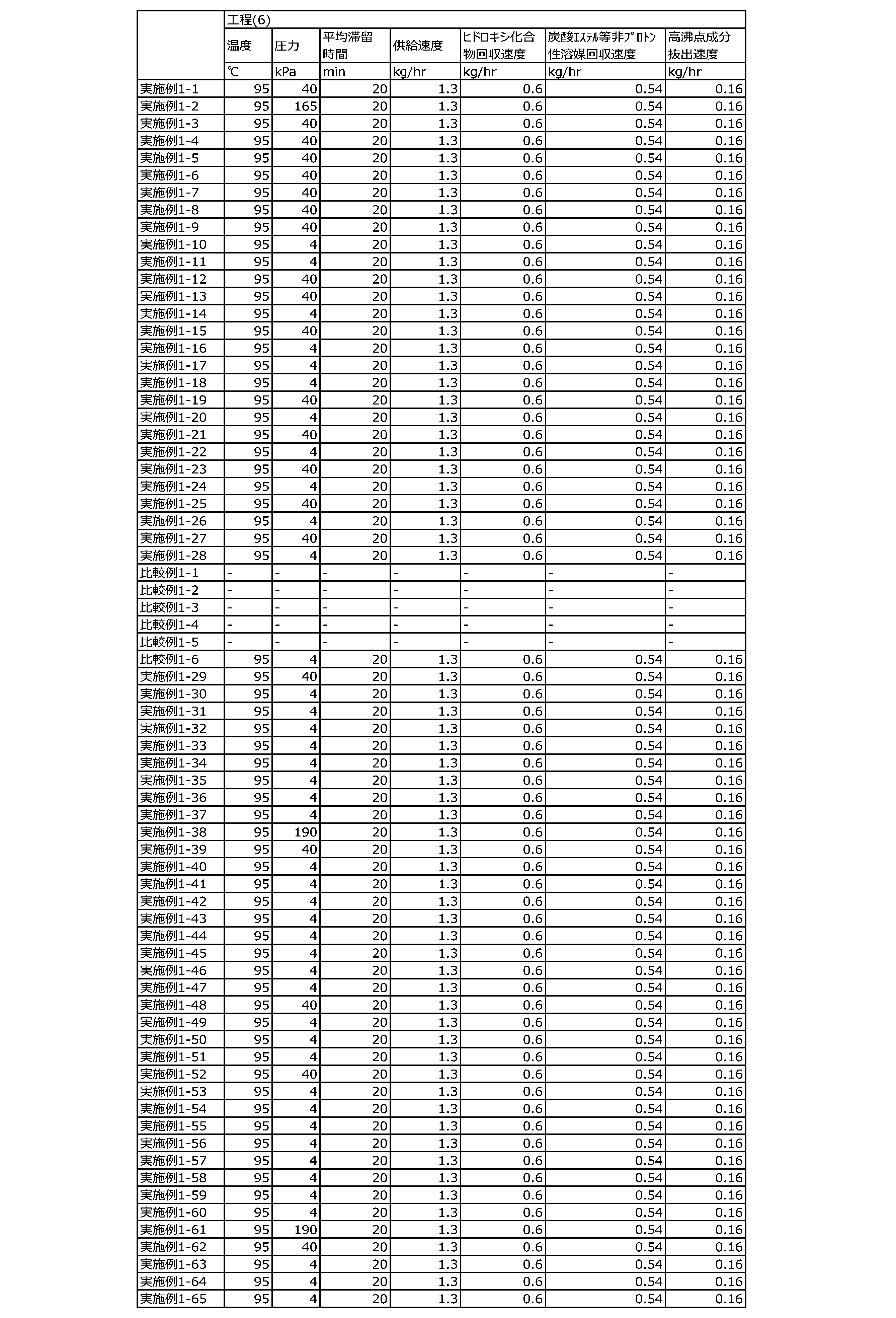
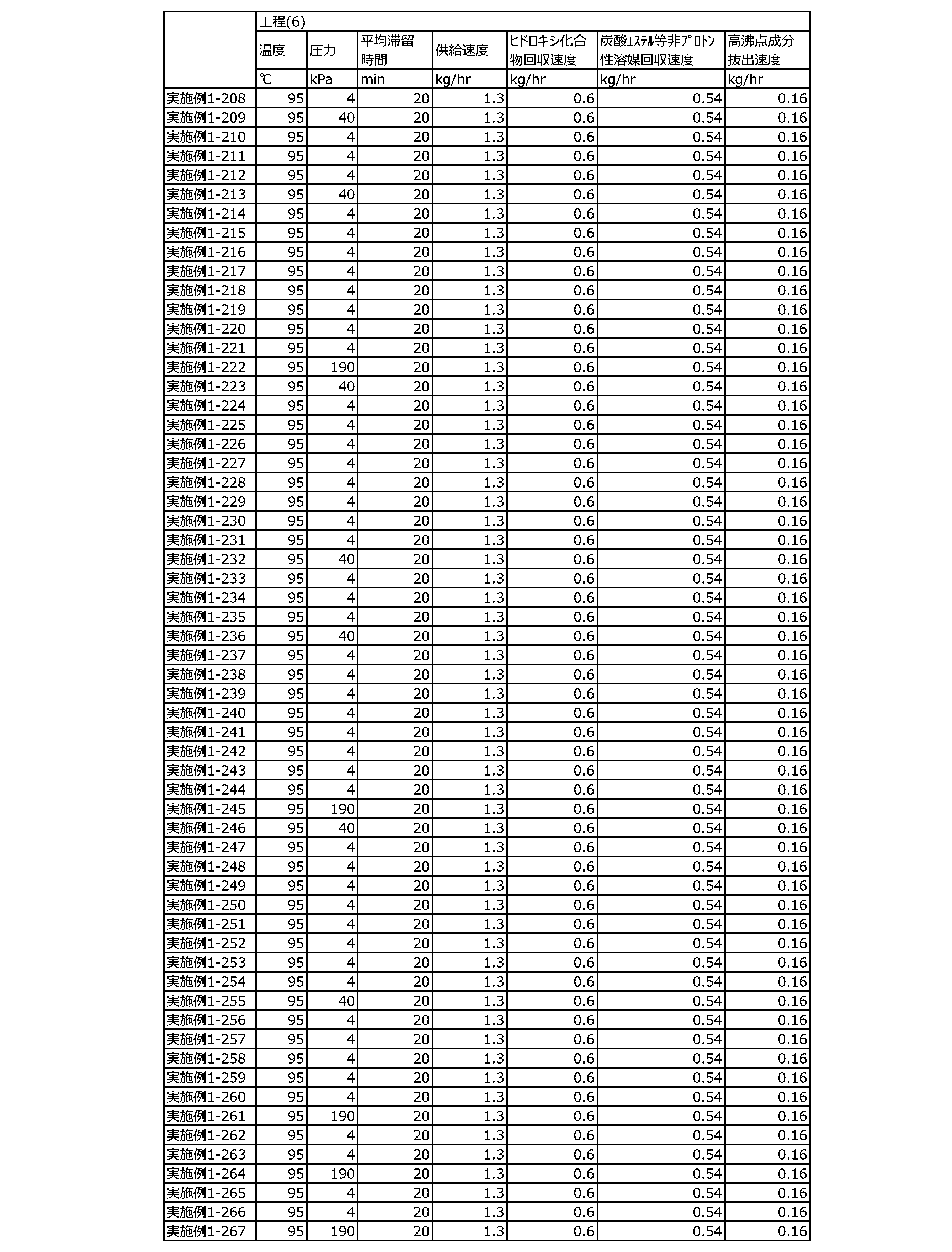
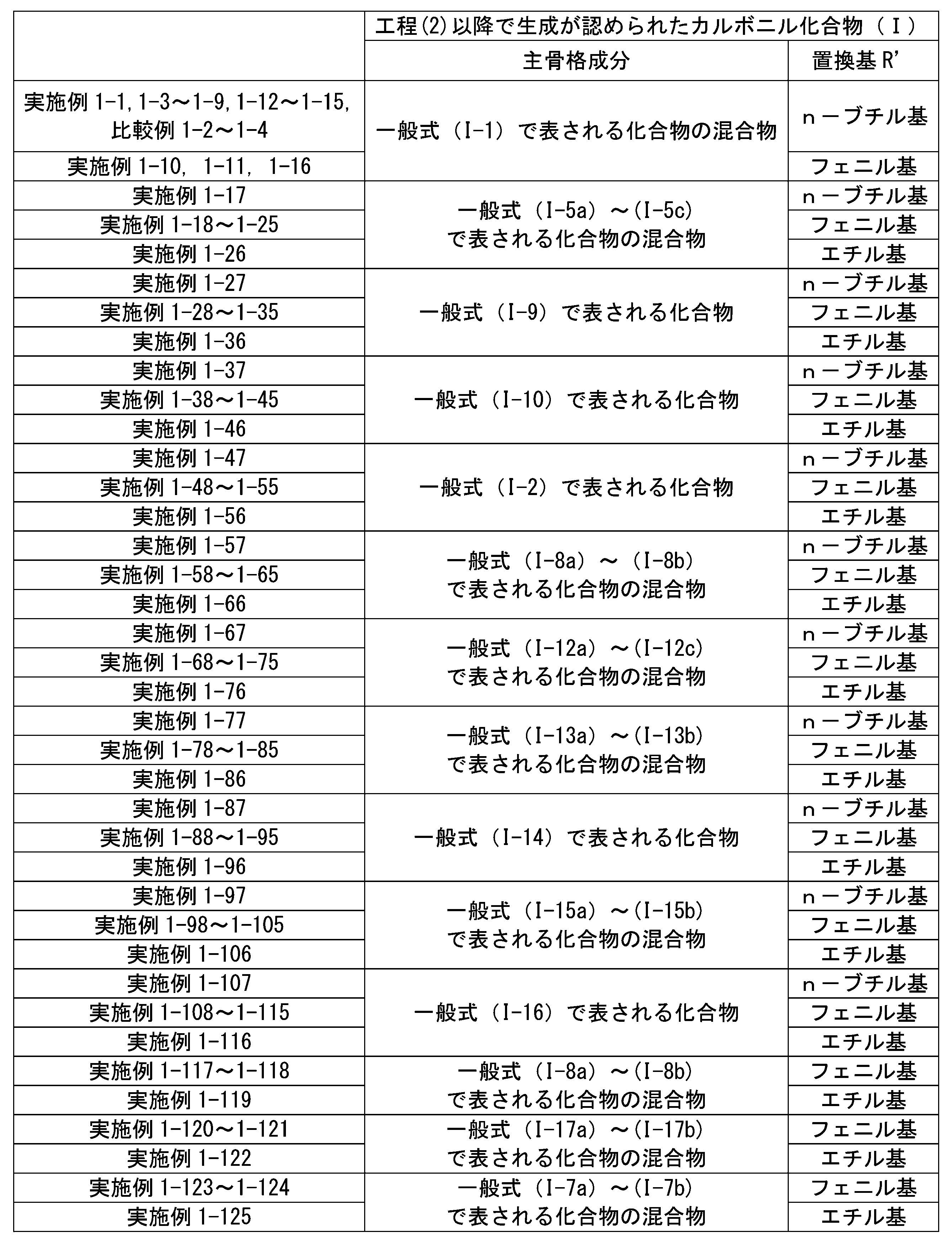
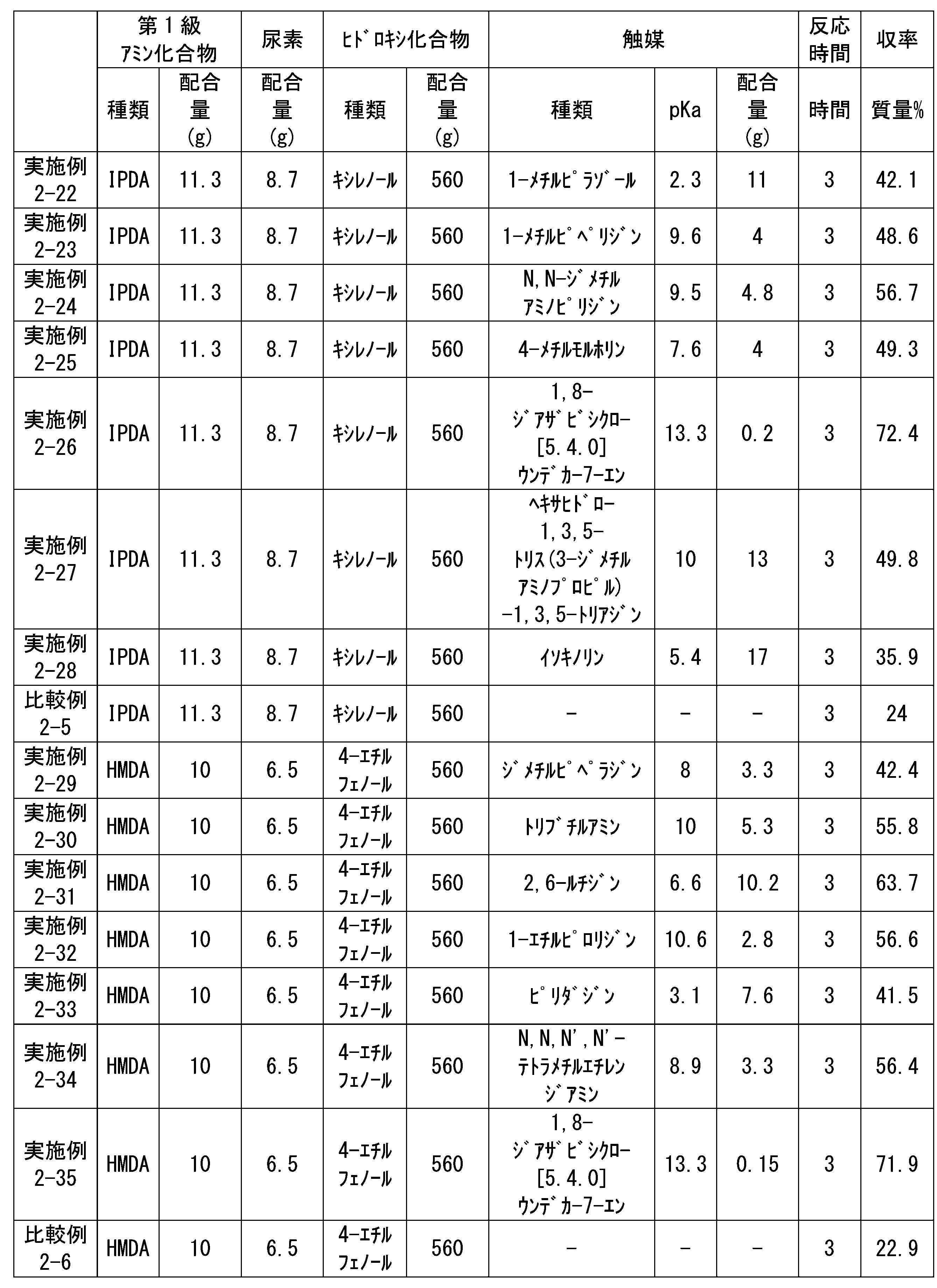
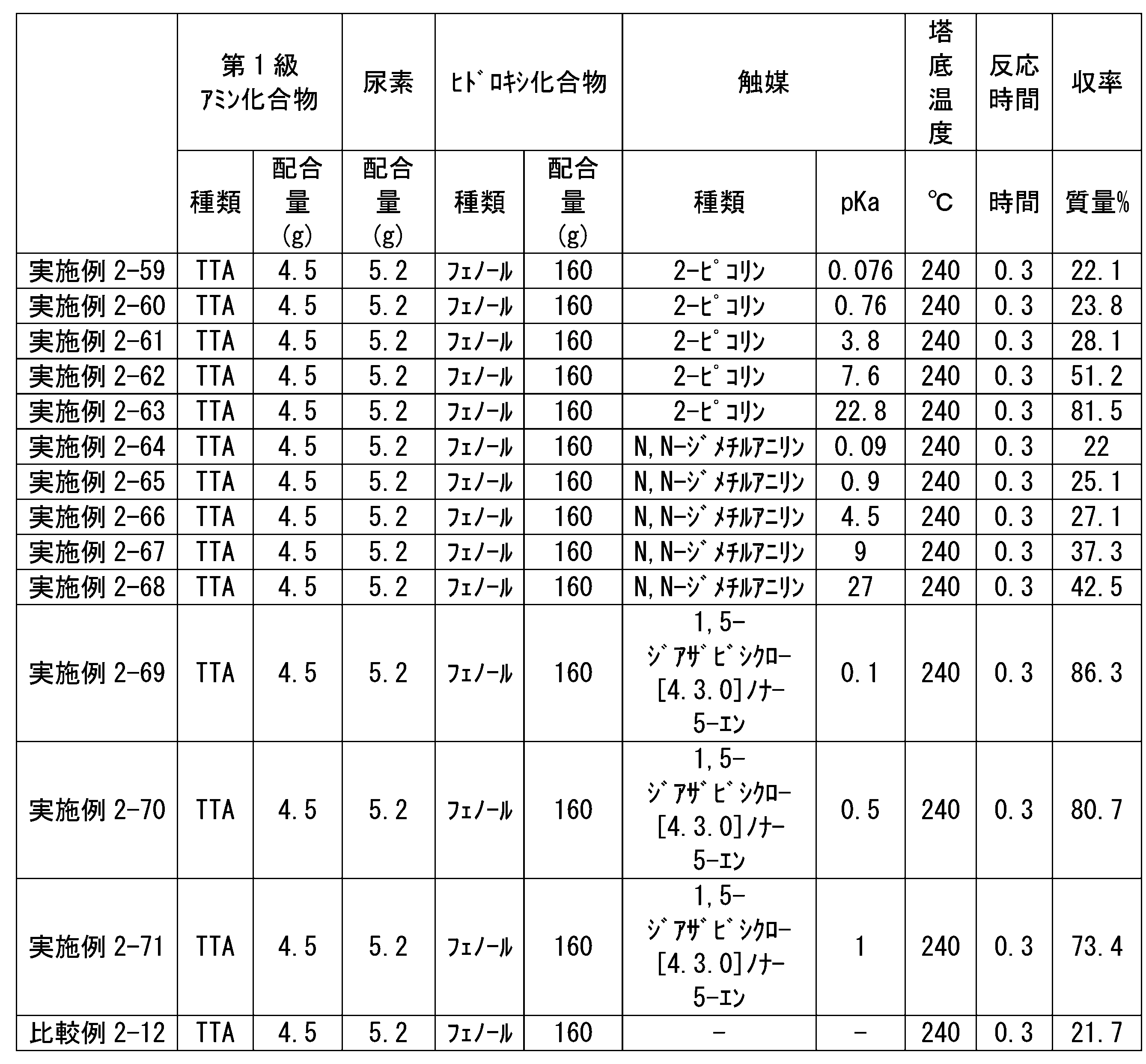
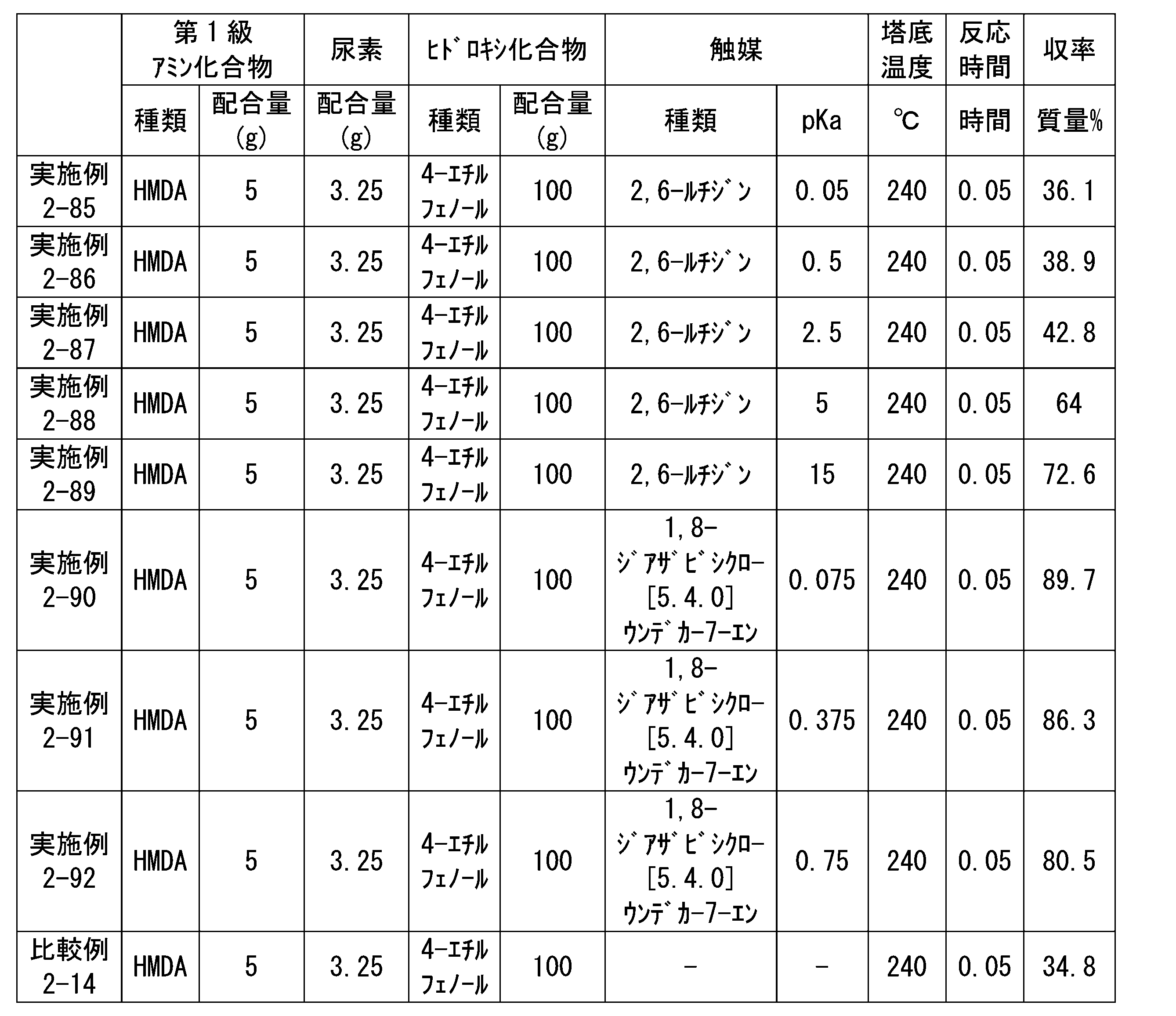
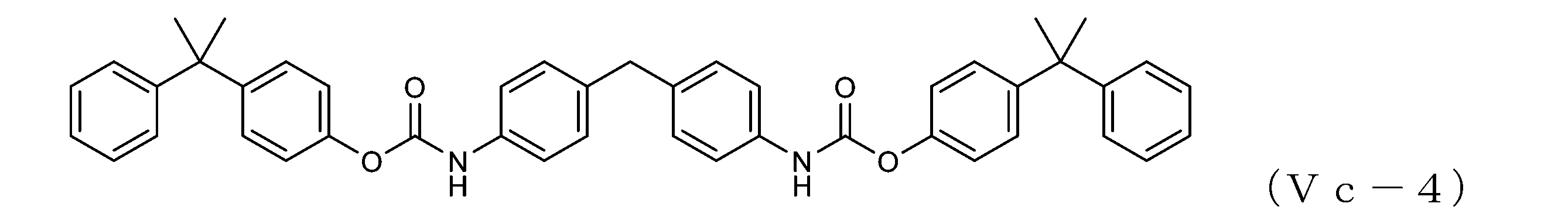
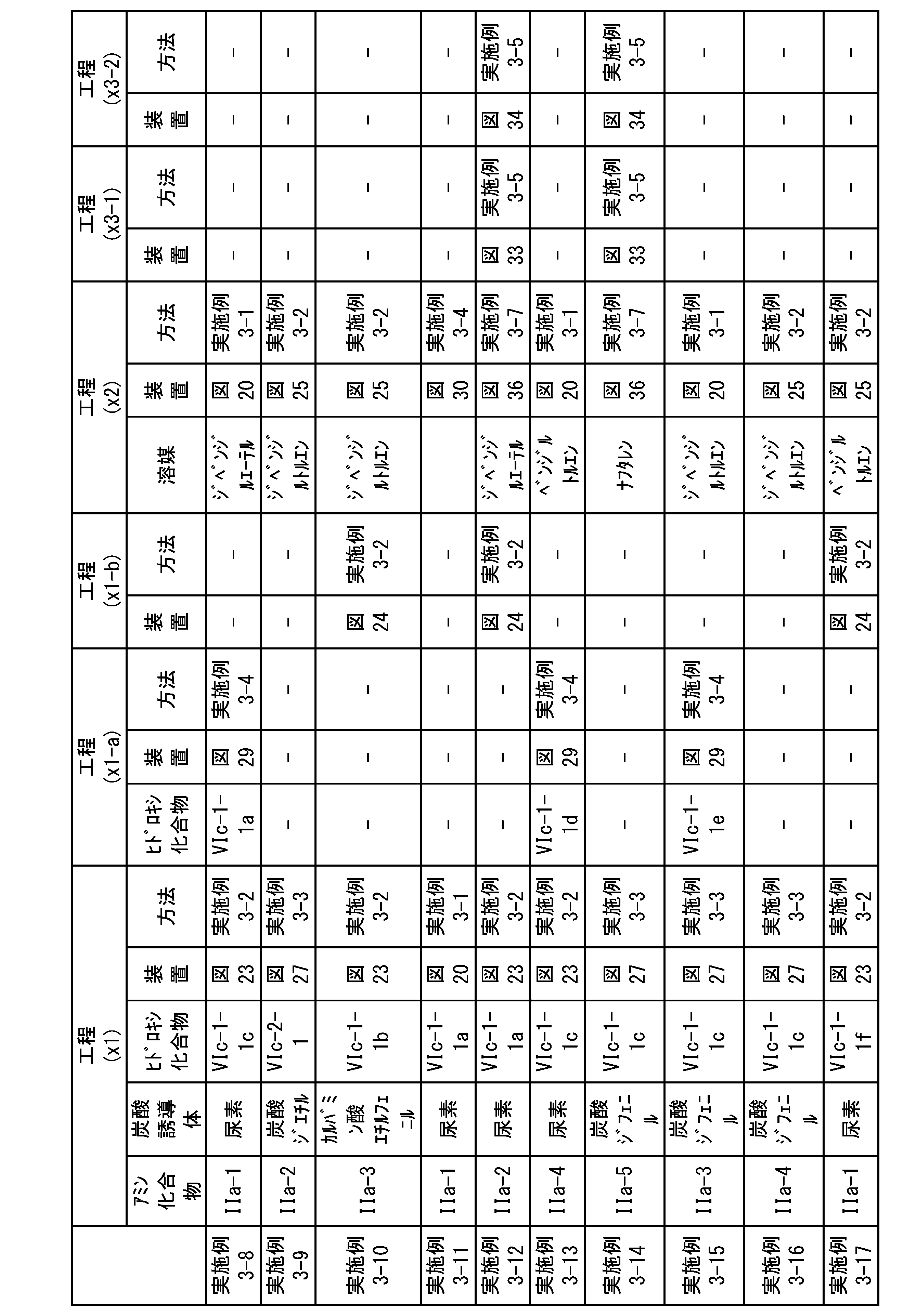
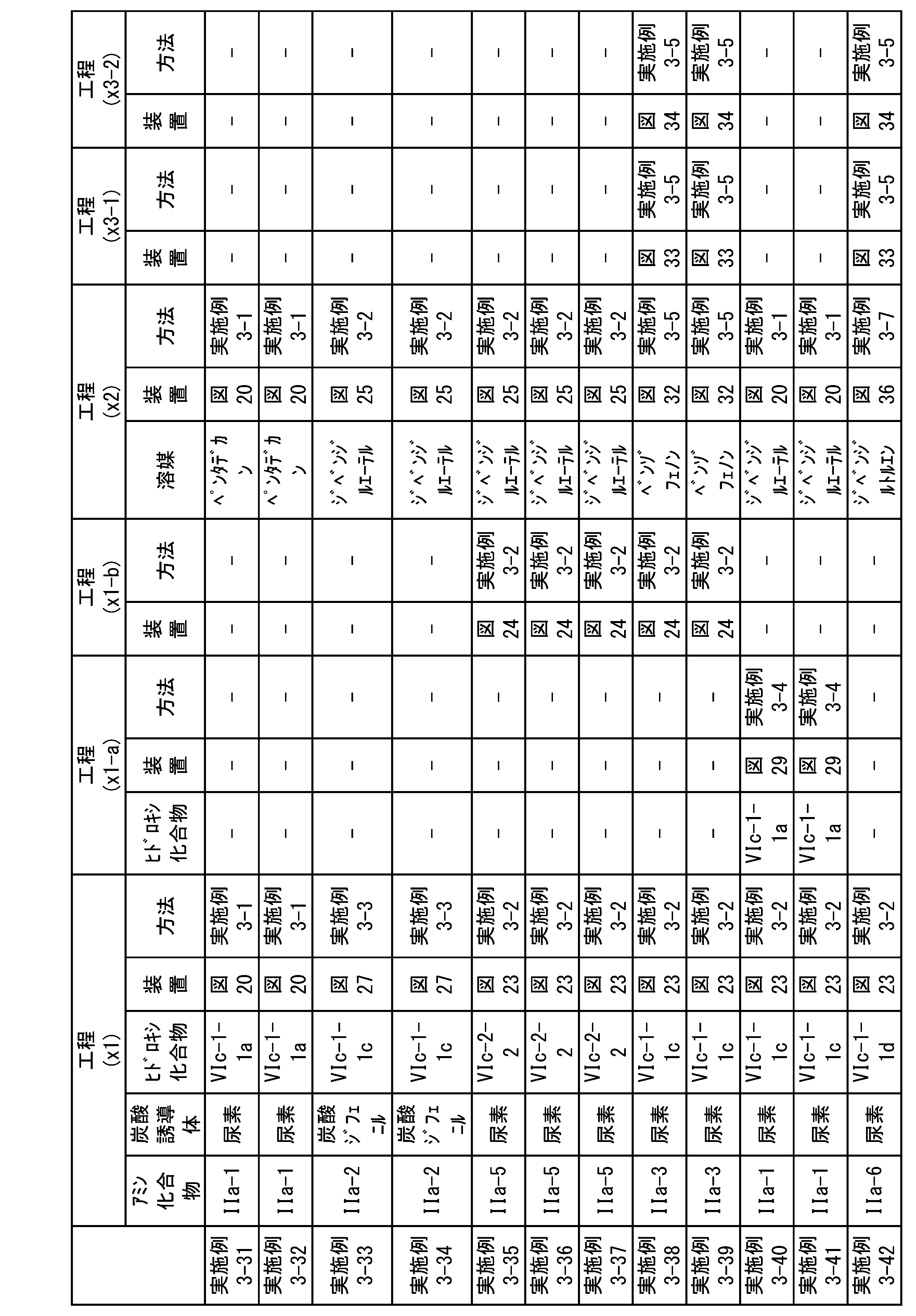
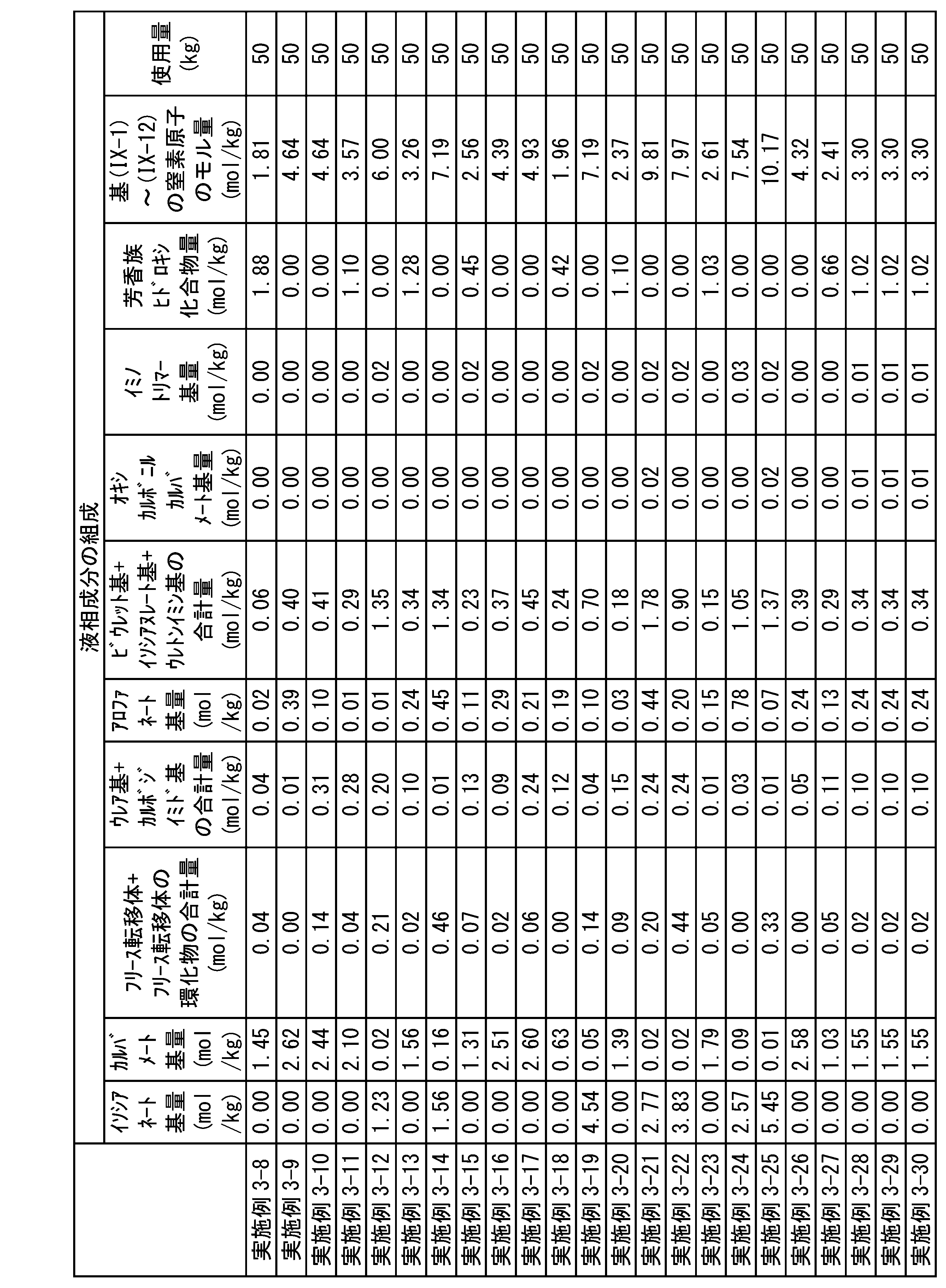
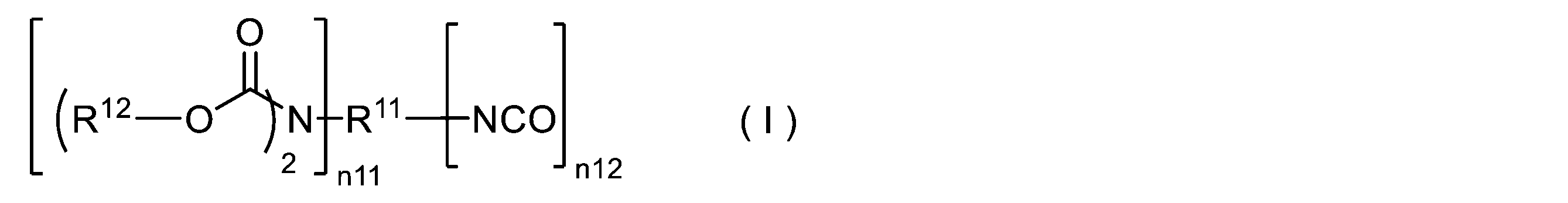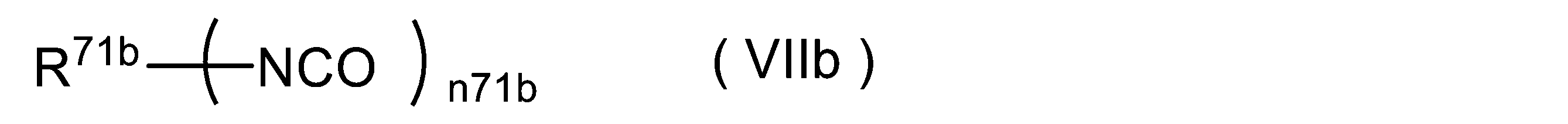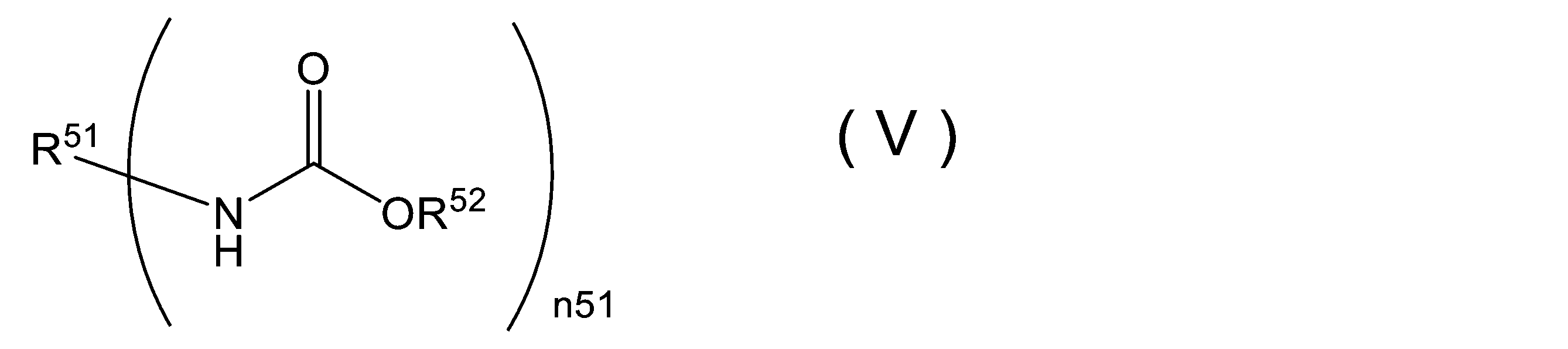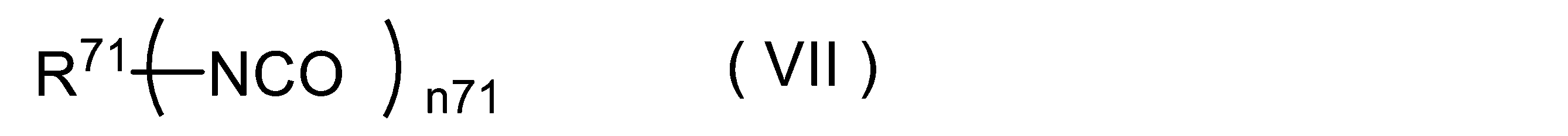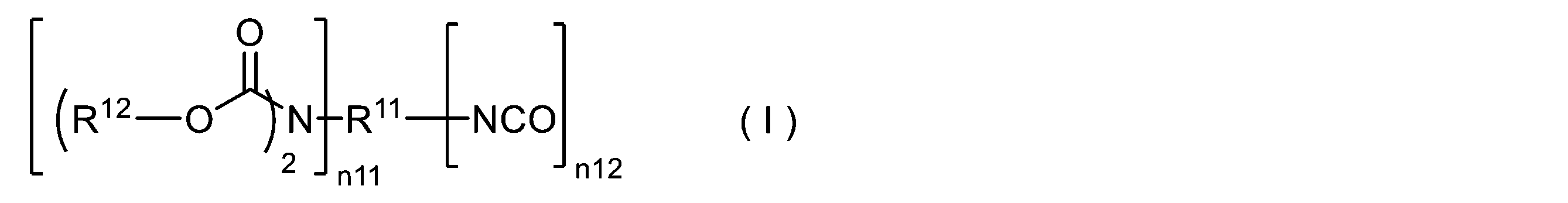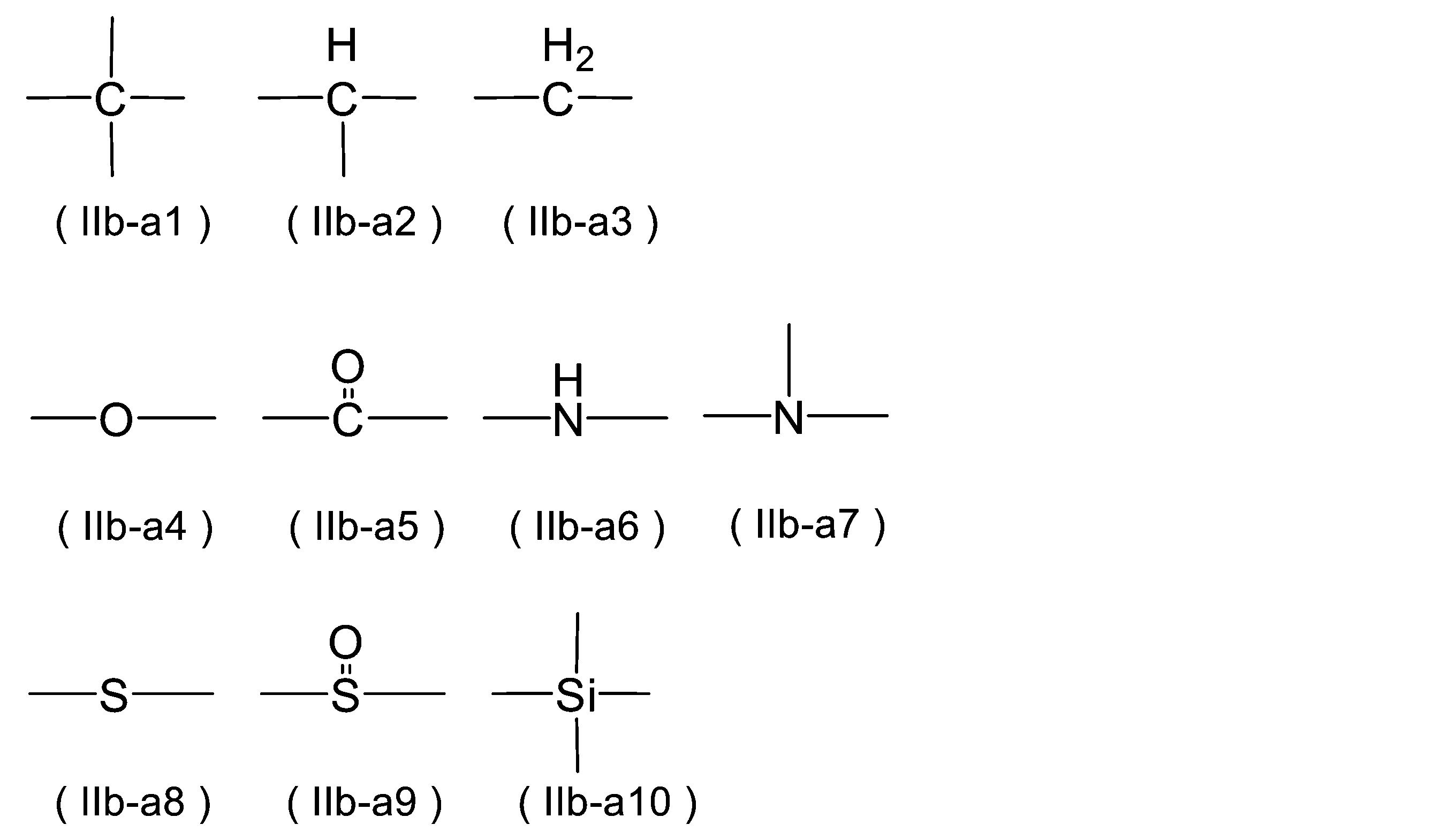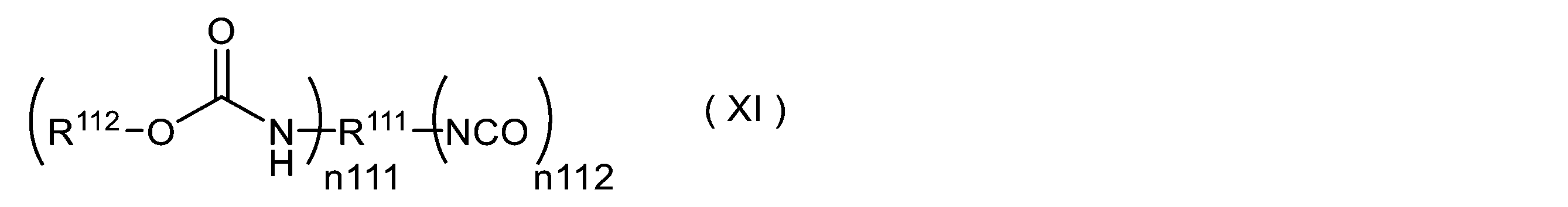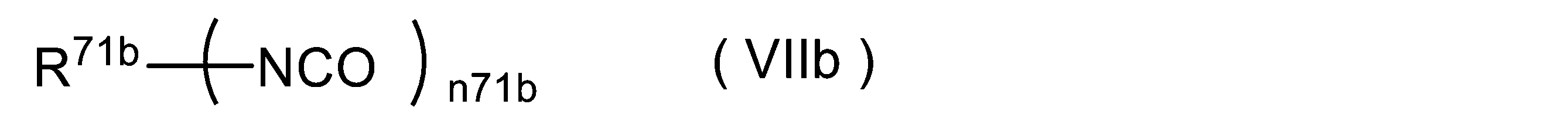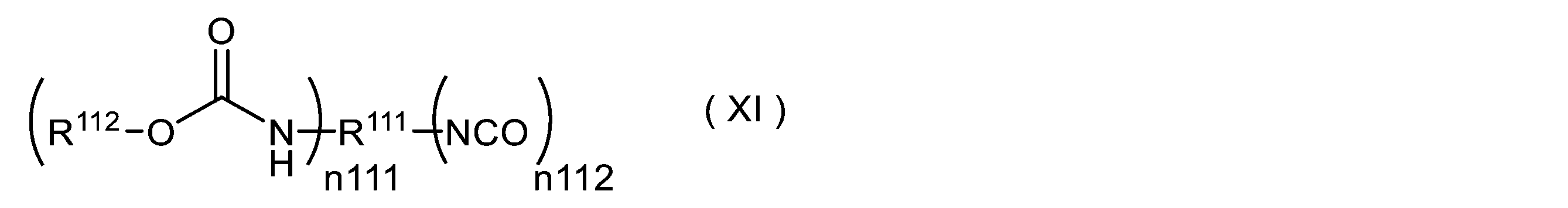WO2023080257A1 - イソシアネート化合物の製造方法、カルバメート化合物の製造方法、アミン化合物の回収方法、イソシアネート組成物 - Google Patents
イソシアネート化合物の製造方法、カルバメート化合物の製造方法、アミン化合物の回収方法、イソシアネート組成物 Download PDFInfo
- Publication number
- WO2023080257A1 WO2023080257A1 PCT/JP2022/041611 JP2022041611W WO2023080257A1 WO 2023080257 A1 WO2023080257 A1 WO 2023080257A1 JP 2022041611 W JP2022041611 W JP 2022041611W WO 2023080257 A1 WO2023080257 A1 WO 2023080257A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- isomer
- compound
- isocyanate
- general formula
- Prior art date
Links
- -1 isocyanate compounds Chemical class 0.000 title claims abstract description 1622
- 239000012948 isocyanate Substances 0.000 title claims abstract description 309
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 154
- 238000000034 method Methods 0.000 title claims description 198
- 150000002513 isocyanates Chemical class 0.000 title claims description 84
- 239000000203 mixture Substances 0.000 title claims description 69
- 150000004657 carbamic acid derivatives Chemical class 0.000 title description 59
- 238000006243 chemical reaction Methods 0.000 claims abstract description 332
- 150000001875 compounds Chemical class 0.000 claims abstract description 267
- 150000002440 hydroxy compounds Chemical class 0.000 claims abstract description 234
- 150000004649 carbonic acid derivatives Chemical class 0.000 claims abstract description 135
- 238000009835 boiling Methods 0.000 claims abstract description 121
- 239000000010 aprotic solvent Substances 0.000 claims abstract description 64
- 239000006227 byproduct Substances 0.000 claims abstract description 52
- 238000005979 thermal decomposition reaction Methods 0.000 claims abstract description 37
- 125000004432 carbon atom Chemical group C* 0.000 claims description 163
- 125000003118 aryl group Chemical group 0.000 claims description 130
- 125000001931 aliphatic group Chemical group 0.000 claims description 102
- 239000001257 hydrogen Substances 0.000 claims description 99
- 229910052739 hydrogen Inorganic materials 0.000 claims description 99
- 238000004821 distillation Methods 0.000 claims description 85
- 239000003054 catalyst Substances 0.000 claims description 77
- 125000000962 organic group Chemical group 0.000 claims description 75
- 239000007791 liquid phase Substances 0.000 claims description 66
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 claims description 59
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 57
- 238000000926 separation method Methods 0.000 claims description 55
- 150000001728 carbonyl compounds Chemical class 0.000 claims description 52
- 239000007789 gas Substances 0.000 claims description 51
- 239000012071 phase Substances 0.000 claims description 49
- 239000000243 solution Substances 0.000 claims description 49
- 238000011084 recovery Methods 0.000 claims description 45
- 239000004202 carbamide Substances 0.000 claims description 37
- 229910052799 carbon Inorganic materials 0.000 claims description 36
- 239000002994 raw material Substances 0.000 claims description 35
- 229910052757 nitrogen Inorganic materials 0.000 claims description 32
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 30
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 30
- 239000000047 product Substances 0.000 claims description 27
- 239000007788 liquid Substances 0.000 claims description 25
- 239000010409 thin film Substances 0.000 claims description 25
- 239000012295 chemical reaction liquid Substances 0.000 claims description 20
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 20
- 125000000320 amidine group Chemical group 0.000 claims description 18
- 239000011552 falling film Substances 0.000 claims description 18
- 150000003672 ureas Chemical class 0.000 claims description 18
- 125000000524 functional group Chemical group 0.000 claims description 17
- ZRALSGWEFCBTJO-UHFFFAOYSA-N guanidine group Chemical group NC(=N)N ZRALSGWEFCBTJO-UHFFFAOYSA-N 0.000 claims description 17
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 15
- 239000003513 alkali Substances 0.000 claims description 15
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 15
- 125000004185 ester group Chemical group 0.000 claims description 14
- 229920000768 polyamine Polymers 0.000 claims description 14
- 125000001302 tertiary amino group Chemical group 0.000 claims description 14
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical group NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 claims description 13
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 claims description 12
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 claims description 12
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 claims description 11
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical group OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 claims description 11
- 150000004706 metal oxides Chemical class 0.000 claims description 11
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 claims description 11
- 150000004651 carbonic acid esters Chemical class 0.000 claims description 10
- 238000000746 purification Methods 0.000 claims description 10
- 238000000066 reactive distillation Methods 0.000 claims description 10
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 claims description 9
- 229910044991 metal oxide Inorganic materials 0.000 claims description 9
- 238000002156 mixing Methods 0.000 claims description 9
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 claims description 8
- 229910052783 alkali metal Inorganic materials 0.000 claims description 8
- 150000001340 alkali metals Chemical class 0.000 claims description 8
- AVWRKZWQTYIKIY-UHFFFAOYSA-N urea-1-carboxylic acid Chemical group NC(=O)NC(O)=O AVWRKZWQTYIKIY-UHFFFAOYSA-N 0.000 claims description 8
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 7
- VPKDCDLSJZCGKE-UHFFFAOYSA-N carbodiimide group Chemical group N=C=N VPKDCDLSJZCGKE-UHFFFAOYSA-N 0.000 claims description 7
- RXYPXQSKLGGKOL-UHFFFAOYSA-N 1,4-dimethylpiperazine Chemical compound CN1CCN(C)CC1 RXYPXQSKLGGKOL-UHFFFAOYSA-N 0.000 claims description 6
- OEBXWWBYZJNKRK-UHFFFAOYSA-N 1-methyl-2,3,4,6,7,8-hexahydropyrimido[1,2-a]pyrimidine Chemical compound C1CCN=C2N(C)CCCN21 OEBXWWBYZJNKRK-UHFFFAOYSA-N 0.000 claims description 6
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 claims description 6
- OISVCGZHLKNMSJ-UHFFFAOYSA-N 2,6-dimethylpyridine Chemical compound CC1=CC=CC(C)=N1 OISVCGZHLKNMSJ-UHFFFAOYSA-N 0.000 claims description 6
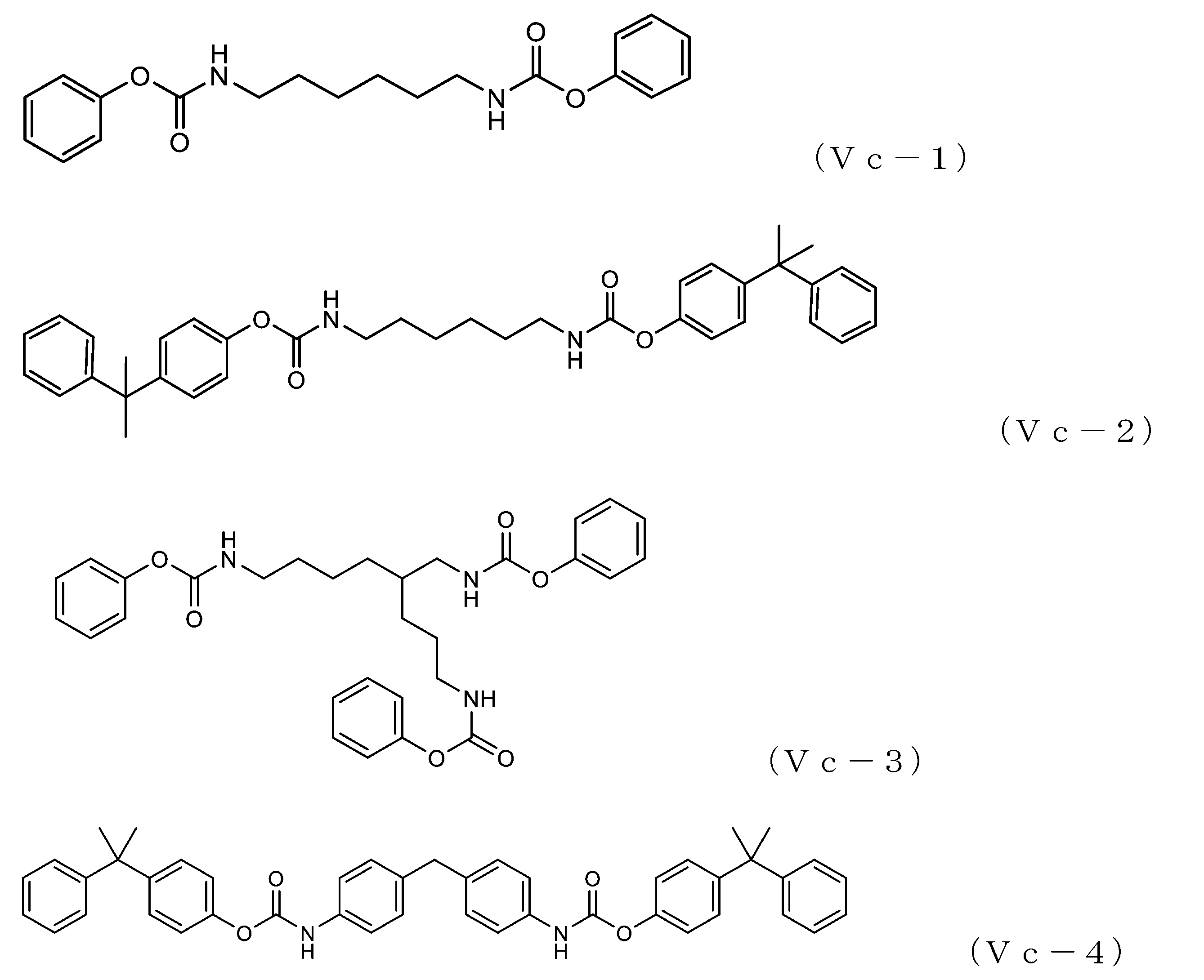
- 101100132433 Arabidopsis thaliana VIII-1 gene Proteins 0.000 claims description 6
- 101100459319 Arabidopsis thaliana VIII-2 gene Proteins 0.000 claims description 6
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 claims description 6
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 claims description 6
- 229910052784 alkaline earth metal Inorganic materials 0.000 claims description 6
- VKYKSIONXSXAKP-UHFFFAOYSA-N hexamethylenetetramine Chemical compound C1N(C2)CN3CN1CN2C3 VKYKSIONXSXAKP-UHFFFAOYSA-N 0.000 claims description 6
- 150000004679 hydroxides Chemical class 0.000 claims description 6
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 claims description 6
- SMUQFGGVLNAIOZ-UHFFFAOYSA-N quinaldine Chemical compound C1=CC=CC2=NC(C)=CC=C21 SMUQFGGVLNAIOZ-UHFFFAOYSA-N 0.000 claims description 6
- 150000008044 alkali metal hydroxides Chemical class 0.000 claims description 5
- 150000001342 alkaline earth metals Chemical class 0.000 claims description 5
- 238000004064 recycling Methods 0.000 claims description 5
- BSKHPKMHTQYZBB-UHFFFAOYSA-N 2-methylpyridine Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 claims description 4
- ISNICOKBNZOJQG-UHFFFAOYSA-N 1,1,2,3,3-pentamethylguanidine Chemical compound CN=C(N(C)C)N(C)C ISNICOKBNZOJQG-UHFFFAOYSA-N 0.000 claims description 3
- ZFDWWDZLRKHULH-UHFFFAOYSA-N 1,2-dimethyl-5,6-dihydro-4h-pyrimidine Chemical compound CN1CCCN=C1C ZFDWWDZLRKHULH-UHFFFAOYSA-N 0.000 claims description 3
- MCTWTZJPVLRJOU-UHFFFAOYSA-N 1-methyl-1H-imidazole Chemical compound CN1C=CN=C1 MCTWTZJPVLRJOU-UHFFFAOYSA-N 0.000 claims description 3
- CJVYYDCBKKKIPD-UHFFFAOYSA-N 1-n,1-n,2-n,2-n-tetramethylbenzene-1,2-diamine Chemical compound CN(C)C1=CC=CC=C1N(C)C CJVYYDCBKKKIPD-UHFFFAOYSA-N 0.000 claims description 3
- DTNQFFYVOVIRFQ-UHFFFAOYSA-N 2-[[2-(dimethylamino)phenyl]methyl]-n,n-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1CC1=CC=CC=C1N(C)C DTNQFFYVOVIRFQ-UHFFFAOYSA-N 0.000 claims description 3
- FZQMJOOSLXFQSU-UHFFFAOYSA-N 3-[3,5-bis[3-(dimethylamino)propyl]-1,3,5-triazinan-1-yl]-n,n-dimethylpropan-1-amine Chemical compound CN(C)CCCN1CN(CCCN(C)C)CN(CCCN(C)C)C1 FZQMJOOSLXFQSU-UHFFFAOYSA-N 0.000 claims description 3
- ZMSQJSMSLXVTKN-UHFFFAOYSA-N 4-[2-(2-morpholin-4-ylethoxy)ethyl]morpholine Chemical compound C1COCCN1CCOCCN1CCOCC1 ZMSQJSMSLXVTKN-UHFFFAOYSA-N 0.000 claims description 3
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 claims description 3
- UQFQONCQIQEYPJ-UHFFFAOYSA-N N-methylpyrazole Chemical compound CN1C=CC=N1 UQFQONCQIQEYPJ-UHFFFAOYSA-N 0.000 claims description 3
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 claims description 3
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 claims description 3
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 claims description 3
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 claims description 3
- 229910001860 alkaline earth metal hydroxide Inorganic materials 0.000 claims description 3
- 229910000287 alkaline earth metal oxide Inorganic materials 0.000 claims description 3
- HOPRXXXSABQWAV-UHFFFAOYSA-N anhydrous collidine Natural products CC1=CC=NC(C)=C1C HOPRXXXSABQWAV-UHFFFAOYSA-N 0.000 claims description 3
- UTBIMNXEDGNJFE-UHFFFAOYSA-N collidine Natural products CC1=CC=C(C)C(C)=N1 UTBIMNXEDGNJFE-UHFFFAOYSA-N 0.000 claims description 3
- HPYNZHMRTTWQTB-UHFFFAOYSA-N dimethylpyridine Natural products CC1=CC=CN=C1C HPYNZHMRTTWQTB-UHFFFAOYSA-N 0.000 claims description 3
- 239000004312 hexamethylene tetramine Substances 0.000 claims description 3
- 235000010299 hexamethylene tetramine Nutrition 0.000 claims description 3
- 230000003301 hydrolyzing effect Effects 0.000 claims description 3
- 125000002883 imidazolyl group Chemical group 0.000 claims description 3
- 125000005956 isoquinolyl group Chemical group 0.000 claims description 3
- 125000002971 oxazolyl group Chemical group 0.000 claims description 3
- UKODFQOELJFMII-UHFFFAOYSA-N pentamethyldiethylenetriamine Chemical compound CN(C)CCN(C)CCN(C)C UKODFQOELJFMII-UHFFFAOYSA-N 0.000 claims description 3
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 3
- 125000005412 pyrazyl group Chemical group 0.000 claims description 3
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical compound C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 claims description 3
- 125000005495 pyridazyl group Chemical group 0.000 claims description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 3
- 125000004076 pyridyl group Chemical group 0.000 claims description 3
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 3
- 125000005493 quinolyl group Chemical group 0.000 claims description 3
- SBYHFKPVCBCYGV-UHFFFAOYSA-N quinuclidine Chemical compound C1CC2CCN1CC2 SBYHFKPVCBCYGV-UHFFFAOYSA-N 0.000 claims description 3
- GFYHSKONPJXCDE-UHFFFAOYSA-N sym-collidine Natural products CC1=CN=C(C)C(C)=C1 GFYHSKONPJXCDE-UHFFFAOYSA-N 0.000 claims description 3
- 125000000335 thiazolyl group Chemical group 0.000 claims description 3
- 229910021482 group 13 metal Inorganic materials 0.000 claims description 2
- LHTGBZMVHWJBQB-UHFFFAOYSA-N n,2-diethylaniline Chemical compound CCNC1=CC=CC=C1CC LHTGBZMVHWJBQB-UHFFFAOYSA-N 0.000 claims description 2
- GUAWMXYQZKVRCW-UHFFFAOYSA-N n,2-dimethylaniline Chemical compound CNC1=CC=CC=C1C GUAWMXYQZKVRCW-UHFFFAOYSA-N 0.000 claims description 2
- 238000006467 substitution reaction Methods 0.000 claims description 2
- 229910001854 alkali hydroxide Inorganic materials 0.000 claims 1
- 229940125844 compound 46 Drugs 0.000 claims 1
- KPADFPAILITQBG-UHFFFAOYSA-N non-4-ene Chemical compound CCCCC=CCCC KPADFPAILITQBG-UHFFFAOYSA-N 0.000 claims 1
- 150000002430 hydrocarbons Chemical class 0.000 description 49
- 125000000217 alkyl group Chemical group 0.000 description 44
- 235000001014 amino acid Nutrition 0.000 description 43
- 229940024606 amino acid Drugs 0.000 description 43
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 41
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 38
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 37
- 125000000753 cycloalkyl group Chemical group 0.000 description 36
- 235000013877 carbamide Nutrition 0.000 description 35
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 35
- 125000003277 amino group Chemical group 0.000 description 33
- 125000004429 atom Chemical group 0.000 description 33
- 239000000463 material Substances 0.000 description 32
- 230000002829 reductive effect Effects 0.000 description 30
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 28
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 28
- 150000001413 amino acids Chemical class 0.000 description 27
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 27
- 239000002904 solvent Substances 0.000 description 27
- 230000008569 process Effects 0.000 description 26
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 24
- 238000000354 decomposition reaction Methods 0.000 description 24
- 125000001424 substituent group Chemical group 0.000 description 24
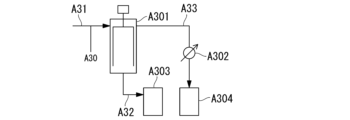
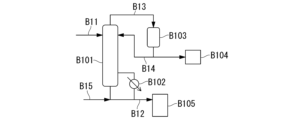
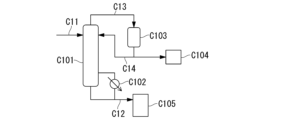
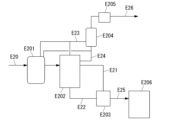
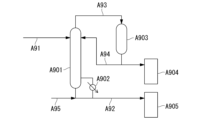
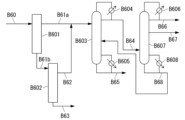
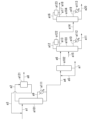
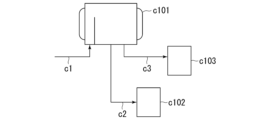
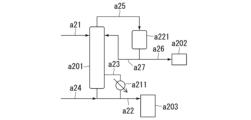
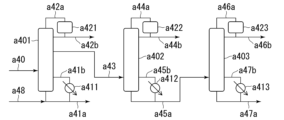
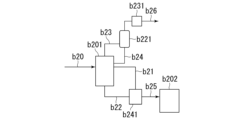
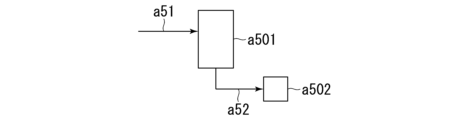
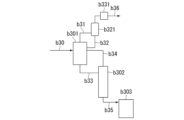
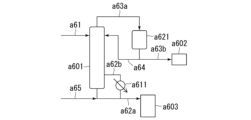
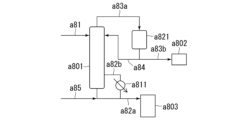
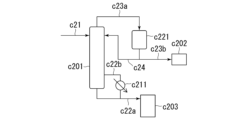
- 238000010586 diagram Methods 0.000 description 23
- 238000012856 packing Methods 0.000 description 22
- 238000003756 stirring Methods 0.000 description 22
- 229910052751 metal Inorganic materials 0.000 description 20
- 239000002184 metal Substances 0.000 description 20
- 150000003862 amino acid derivatives Chemical class 0.000 description 19
- 125000002015 acyclic group Chemical group 0.000 description 18
- 239000007810 chemical reaction solvent Substances 0.000 description 18
- 239000011521 glass Substances 0.000 description 18
- 229910052717 sulfur Inorganic materials 0.000 description 18
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 17
- 125000003545 alkoxy group Chemical group 0.000 description 17
- 229910052710 silicon Inorganic materials 0.000 description 17
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 16
- YGYAWVDWMABLBF-UHFFFAOYSA-N Phosgene Chemical compound ClC(Cl)=O YGYAWVDWMABLBF-UHFFFAOYSA-N 0.000 description 16
- 238000006460 hydrolysis reaction Methods 0.000 description 16
- 125000003710 aryl alkyl group Chemical group 0.000 description 15
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 15
- 229910052760 oxygen Inorganic materials 0.000 description 15
- 239000001301 oxygen Substances 0.000 description 15
- 239000010703 silicon Substances 0.000 description 15
- 239000007787 solid Substances 0.000 description 15
- 150000004945 aromatic hydrocarbons Chemical group 0.000 description 14
- 238000001816 cooling Methods 0.000 description 14
- 239000011261 inert gas Substances 0.000 description 14
- 238000007086 side reaction Methods 0.000 description 14
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 13
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 13
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 13
- 239000011593 sulfur Substances 0.000 description 13
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 12
- 229910021529 ammonia Inorganic materials 0.000 description 12
- 230000015572 biosynthetic process Effects 0.000 description 12
- 150000002148 esters Chemical class 0.000 description 12
- 125000005843 halogen group Chemical group 0.000 description 12
- 125000001624 naphthyl group Chemical group 0.000 description 12
- 239000000126 substance Substances 0.000 description 11
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 10
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 10
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 10
- 239000002253 acid Substances 0.000 description 10
- 238000011437 continuous method Methods 0.000 description 10
- 238000010438 heat treatment Methods 0.000 description 10
- 230000035484 reaction time Effects 0.000 description 10
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 9
- 150000001298 alcohols Chemical class 0.000 description 9
- 150000001721 carbon Chemical group 0.000 description 9
- 239000012528 membrane Substances 0.000 description 9
- 238000005191 phase separation Methods 0.000 description 9
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 8
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 8
- 239000004472 Lysine Substances 0.000 description 8
- 125000002723 alicyclic group Chemical group 0.000 description 8
- 125000002102 aryl alkyloxo group Chemical group 0.000 description 8
- 125000004104 aryloxy group Chemical group 0.000 description 8
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 8
- MWKFXSUHUHTGQN-UHFFFAOYSA-N decan-1-ol Chemical compound CCCCCCCCCCO MWKFXSUHUHTGQN-UHFFFAOYSA-N 0.000 description 8
- 230000007423 decrease Effects 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- UYMKPFRHYYNDTL-UHFFFAOYSA-N ethenamine Chemical compound NC=C UYMKPFRHYYNDTL-UHFFFAOYSA-N 0.000 description 8
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 8
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 8
- 239000000945 filler Substances 0.000 description 8
- 125000000623 heterocyclic group Chemical group 0.000 description 8
- ZSIAUFGUXNUGDI-UHFFFAOYSA-N hexan-1-ol Chemical compound CCCCCCO ZSIAUFGUXNUGDI-UHFFFAOYSA-N 0.000 description 8
- 235000018977 lysine Nutrition 0.000 description 8
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 8
- 230000004048 modification Effects 0.000 description 8
- 238000012986 modification Methods 0.000 description 8
- 150000003141 primary amines Chemical class 0.000 description 8
- 229920006395 saturated elastomer Polymers 0.000 description 8
- 150000003512 tertiary amines Chemical class 0.000 description 8
- VZSRBBMJRBPUNF-UHFFFAOYSA-N 2-(2,3-dihydro-1H-inden-2-ylamino)-N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]pyrimidine-5-carboxamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C(=O)NCCC(N1CC2=C(CC1)NN=N2)=O VZSRBBMJRBPUNF-UHFFFAOYSA-N 0.000 description 7
- 229910000975 Carbon steel Inorganic materials 0.000 description 7
- 239000004809 Teflon Substances 0.000 description 7
- 229920006362 Teflon® Polymers 0.000 description 7
- 230000002411 adverse Effects 0.000 description 7
- 125000002947 alkylene group Chemical group 0.000 description 7
- 239000002585 base Substances 0.000 description 7
- 239000010962 carbon steel Substances 0.000 description 7
- 239000000919 ceramic Substances 0.000 description 7
- 239000011248 coating agent Substances 0.000 description 7
- 238000000576 coating method Methods 0.000 description 7
- 238000010924 continuous production Methods 0.000 description 7
- 229910000856 hastalloy Inorganic materials 0.000 description 7
- 229930195733 hydrocarbon Natural products 0.000 description 7
- 238000006011 modification reaction Methods 0.000 description 7
- 125000002950 monocyclic group Chemical group 0.000 description 7
- 125000003367 polycyclic group Chemical group 0.000 description 7
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 7
- 230000009257 reactivity Effects 0.000 description 7
- 125000003003 spiro group Chemical group 0.000 description 7
- 239000010935 stainless steel Substances 0.000 description 7
- 229910001220 stainless steel Inorganic materials 0.000 description 7
- 239000000758 substrate Substances 0.000 description 7
- 125000003944 tolyl group Chemical group 0.000 description 7
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 6
- BBMCTIGTTCKYKF-UHFFFAOYSA-N 1-heptanol Chemical compound CCCCCCCO BBMCTIGTTCKYKF-UHFFFAOYSA-N 0.000 description 6
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 6
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 6
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 6
- 238000005618 Fries rearrangement reaction Methods 0.000 description 6
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 6
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 6
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 150000001412 amines Chemical class 0.000 description 6
- CKLJMWTZIZZHCS-REOHCLBHSA-N aspartic acid group Chemical group N[C@@H](CC(=O)O)C(=O)O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 6
- OHJMTUPIZMNBFR-UHFFFAOYSA-N biuret Chemical group NC(=O)NC(N)=O OHJMTUPIZMNBFR-UHFFFAOYSA-N 0.000 description 6
- 239000012267 brine Substances 0.000 description 6
- 239000001569 carbon dioxide Substances 0.000 description 6
- 229910002092 carbon dioxide Inorganic materials 0.000 description 6
- 239000000498 cooling water Substances 0.000 description 6
- 229920001577 copolymer Polymers 0.000 description 6
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 6
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 6
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 6
- ZWRUINPWMLAQRD-UHFFFAOYSA-N nonan-1-ol Chemical compound CCCCCCCCCO ZWRUINPWMLAQRD-UHFFFAOYSA-N 0.000 description 6
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 6
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 6
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 6
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 6
- 229910052814 silicon oxide Inorganic materials 0.000 description 6
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 6
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 5
- 239000004471 Glycine Substances 0.000 description 5
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 5
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 5
- 125000005577 anthracene group Chemical group 0.000 description 5
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 5
- 235000003704 aspartic acid Nutrition 0.000 description 5
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 5
- 125000006267 biphenyl group Chemical group 0.000 description 5
- 125000001951 carbamoylamino group Chemical group C(N)(=O)N* 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 125000004122 cyclic group Chemical group 0.000 description 5
- 125000002993 cycloalkylene group Chemical group 0.000 description 5
- 235000013922 glutamic acid Nutrition 0.000 description 5
- 239000004220 glutamic acid Substances 0.000 description 5
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 5
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 description 5
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 5
- 150000002483 hydrogen compounds Chemical class 0.000 description 5
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 5
- 150000007522 mineralic acids Chemical class 0.000 description 5
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 5
- 238000010992 reflux Methods 0.000 description 5
- 125000004434 sulfur atom Chemical group 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- JOYRKODLDBILNP-UHFFFAOYSA-N urethane group Chemical group NC(=O)OCC JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 5
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 4
- OOCCDEMITAIZTP-QPJJXVBHSA-N (E)-cinnamyl alcohol Chemical compound OC\C=C\C1=CC=CC=C1 OOCCDEMITAIZTP-QPJJXVBHSA-N 0.000 description 4
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 4
- IXQGCWUGDFDQMF-UHFFFAOYSA-N 2-Ethylphenol Chemical compound CCC1=CC=CC=C1O IXQGCWUGDFDQMF-UHFFFAOYSA-N 0.000 description 4
- GJYCVCVHRSWLNY-UHFFFAOYSA-N 2-butylphenol Chemical compound CCCCC1=CC=CC=C1O GJYCVCVHRSWLNY-UHFFFAOYSA-N 0.000 description 4
- HMJBXEZHJUYJQY-UHFFFAOYSA-N 4-(aminomethyl)octane-1,8-diamine Chemical compound NCCCCC(CN)CCCN HMJBXEZHJUYJQY-UHFFFAOYSA-N 0.000 description 4
- 239000004475 Arginine Substances 0.000 description 4
- 239000004215 Carbon black (E152) Substances 0.000 description 4
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 4
- 239000005977 Ethylene Substances 0.000 description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 4
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 4
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 4
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical group [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 4
- 150000007513 acids Chemical class 0.000 description 4
- 239000003463 adsorbent Substances 0.000 description 4
- 230000001476 alcoholic effect Effects 0.000 description 4
- 150000007824 aliphatic compounds Chemical class 0.000 description 4
- XXROGKLTLUQVRX-UHFFFAOYSA-N allyl alcohol Chemical compound OCC=C XXROGKLTLUQVRX-UHFFFAOYSA-N 0.000 description 4
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 4
- 235000009697 arginine Nutrition 0.000 description 4
- 229910052786 argon Inorganic materials 0.000 description 4
- 150000001491 aromatic compounds Chemical class 0.000 description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 4
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical class C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 4
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 4
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 4
- 238000009833 condensation Methods 0.000 description 4
- 230000005494 condensation Effects 0.000 description 4
- 239000010949 copper Substances 0.000 description 4
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 4
- XCIXKGXIYUWCLL-UHFFFAOYSA-N cyclopentanol Chemical compound OC1CCCC1 XCIXKGXIYUWCLL-UHFFFAOYSA-N 0.000 description 4
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 4
- 238000004925 denaturation Methods 0.000 description 4
- 230000036425 denaturation Effects 0.000 description 4
- 238000013461 design Methods 0.000 description 4
- 125000005442 diisocyanate group Chemical group 0.000 description 4
- LQZZUXJYWNFBMV-UHFFFAOYSA-N dodecan-1-ol Chemical compound CCCCCCCCCCCCO LQZZUXJYWNFBMV-UHFFFAOYSA-N 0.000 description 4
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- LHGVFZTZFXWLCP-UHFFFAOYSA-N guaiacol Chemical compound COC1=CC=CC=C1O LHGVFZTZFXWLCP-UHFFFAOYSA-N 0.000 description 4
- 229910052734 helium Inorganic materials 0.000 description 4
- 239000001307 helium Substances 0.000 description 4
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 4
- 125000001072 heteroaryl group Chemical group 0.000 description 4
- BTFJIXJJCSYFAL-UHFFFAOYSA-N icosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCO BTFJIXJJCSYFAL-UHFFFAOYSA-N 0.000 description 4
- 239000000543 intermediate Substances 0.000 description 4
- 238000002955 isolation Methods 0.000 description 4
- 150000002736 metal compounds Chemical class 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 125000005561 phenanthryl group Chemical group 0.000 description 4
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 4
- COLNVLDHVKWLRT-QMMMGPOBSA-N phenylalanine group Chemical group N[C@@H](CC1=CC=CC=C1)C(=O)O COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 4
- 125000000286 phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 4
- 229920000642 polymer Polymers 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 125000001725 pyrenyl group Chemical group 0.000 description 4
- 150000003335 secondary amines Chemical class 0.000 description 4
- 238000001179 sorption measurement Methods 0.000 description 4
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- KJIOQYGWTQBHNH-UHFFFAOYSA-N undecanol Chemical compound CCCCCCCCCCCO KJIOQYGWTQBHNH-UHFFFAOYSA-N 0.000 description 4
- HXKKHQJGJAFBHI-UHFFFAOYSA-N 1-aminopropan-2-ol Chemical compound CC(O)CN HXKKHQJGJAFBHI-UHFFFAOYSA-N 0.000 description 3
- OVENINIFSWEPGA-UHFFFAOYSA-N 2-aminopropyl prop-2-enoate Chemical compound CC(N)COC(=O)C=C OVENINIFSWEPGA-UHFFFAOYSA-N 0.000 description 3
- MHQULXYNBKWNDF-UHFFFAOYSA-N 3,4-dimethylbenzene-1,2-diamine Chemical group CC1=CC=C(N)C(N)=C1C MHQULXYNBKWNDF-UHFFFAOYSA-N 0.000 description 3
- RNLHGQLZWXBQNY-UHFFFAOYSA-N 3-(aminomethyl)-3,5,5-trimethylcyclohexan-1-amine Chemical compound CC1(C)CC(N)CC(C)(CN)C1 RNLHGQLZWXBQNY-UHFFFAOYSA-N 0.000 description 3
- YBRVSVVVWCFQMG-UHFFFAOYSA-N 4,4'-diaminodiphenylmethane Chemical compound C1=CC(N)=CC=C1CC1=CC=C(N)C=C1 YBRVSVVVWCFQMG-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical group [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 3
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 3
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 3
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 3
- 239000002841 Lewis acid Substances 0.000 description 3
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 3
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 3
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 238000009825 accumulation Methods 0.000 description 3
- CUJRVFIICFDLGR-UHFFFAOYSA-N acetylacetonate Chemical compound CC(=O)[CH-]C(C)=O CUJRVFIICFDLGR-UHFFFAOYSA-N 0.000 description 3
- 235000004279 alanine Nutrition 0.000 description 3
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 3
- 150000003973 alkyl amines Chemical class 0.000 description 3
- 125000005011 alkyl ether group Chemical group 0.000 description 3
- 125000005233 alkylalcohol group Chemical group 0.000 description 3
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 3
- JFDZBHWFFUWGJE-UHFFFAOYSA-N benzonitrile Chemical compound N#CC1=CC=CC=C1 JFDZBHWFFUWGJE-UHFFFAOYSA-N 0.000 description 3
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 3
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 3
- 239000007806 chemical reaction intermediate Substances 0.000 description 3
- 239000000460 chlorine Substances 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 3
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 3
- GGSUCNLOZRCGPQ-UHFFFAOYSA-N diethylaniline Chemical compound CCN(CC)C1=CC=CC=C1 GGSUCNLOZRCGPQ-UHFFFAOYSA-N 0.000 description 3
- 150000002170 ethers Chemical class 0.000 description 3
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 125000005842 heteroatom Chemical group 0.000 description 3
- 229960000310 isoleucine Drugs 0.000 description 3
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 3
- 150000002576 ketones Chemical class 0.000 description 3
- 150000003951 lactams Chemical class 0.000 description 3
- 239000011133 lead Substances 0.000 description 3
- 150000007517 lewis acids Chemical class 0.000 description 3
- 238000004811 liquid chromatography Methods 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- 238000002844 melting Methods 0.000 description 3
- 150000001247 metal acetylides Chemical class 0.000 description 3
- 229930182817 methionine Natural products 0.000 description 3
- YZOISHTVEWVNHA-UHFFFAOYSA-N n,n'-dicyclohexylmethanediamine Chemical compound C1CCCCC1NCNC1CCCCC1 YZOISHTVEWVNHA-UHFFFAOYSA-N 0.000 description 3
- 150000004767 nitrides Chemical class 0.000 description 3
- 229960003104 ornithine Drugs 0.000 description 3
- 150000002989 phenols Chemical class 0.000 description 3
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 3
- DYFXGORUJGZJCA-UHFFFAOYSA-N phenylmethanediamine Chemical compound NC(N)C1=CC=CC=C1 DYFXGORUJGZJCA-UHFFFAOYSA-N 0.000 description 3
- 239000005056 polyisocyanate Substances 0.000 description 3
- 229920001228 polyisocyanate Polymers 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 238000001556 precipitation Methods 0.000 description 3
- 230000001737 promoting effect Effects 0.000 description 3
- 239000001294 propane Substances 0.000 description 3
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 3
- 238000000197 pyrolysis Methods 0.000 description 3
- 239000011541 reaction mixture Substances 0.000 description 3
- 238000007363 ring formation reaction Methods 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- SQGYOTSLMSWVJD-UHFFFAOYSA-N silver(1+) nitrate Chemical compound [Ag+].[O-]N(=O)=O SQGYOTSLMSWVJD-UHFFFAOYSA-N 0.000 description 3
- CXWXQJXEFPUFDZ-UHFFFAOYSA-N tetralin Chemical compound C1=CC=C2CCCCC2=C1 CXWXQJXEFPUFDZ-UHFFFAOYSA-N 0.000 description 3
- 150000003568 thioethers Chemical class 0.000 description 3
- 229910052719 titanium Inorganic materials 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- OTJFQRMIRKXXRS-UHFFFAOYSA-N (hydroxymethylamino)methanol Chemical compound OCNCO OTJFQRMIRKXXRS-UHFFFAOYSA-N 0.000 description 2
- GEYOCULIXLDCMW-UHFFFAOYSA-N 1,2-phenylenediamine Chemical compound NC1=CC=CC=C1N GEYOCULIXLDCMW-UHFFFAOYSA-N 0.000 description 2
- PCHXZXKMYCGVFA-UHFFFAOYSA-N 1,3-diazetidine-2,4-dione Chemical compound O=C1NC(=O)N1 PCHXZXKMYCGVFA-UHFFFAOYSA-N 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- SGUVLZREKBPKCE-UHFFFAOYSA-N 1,5-diazabicyclo[4.3.0]-non-5-ene Chemical compound C1CCN=C2CCCN21 SGUVLZREKBPKCE-UHFFFAOYSA-N 0.000 description 2
- YIWGJFPJRAEKMK-UHFFFAOYSA-N 1-(2H-benzotriazol-5-yl)-3-methyl-8-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carbonyl]-1,3,8-triazaspiro[4.5]decane-2,4-dione Chemical compound CN1C(=O)N(c2ccc3n[nH]nc3c2)C2(CCN(CC2)C(=O)c2cnc(NCc3cccc(OC(F)(F)F)c3)nc2)C1=O YIWGJFPJRAEKMK-UHFFFAOYSA-N 0.000 description 2
- KODLUXHSIZOKTG-UHFFFAOYSA-N 1-aminobutan-2-ol Chemical compound CCC(O)CN KODLUXHSIZOKTG-UHFFFAOYSA-N 0.000 description 2
- QYYKZVMRNIHICX-UHFFFAOYSA-N 1-phenoxyethanamine Chemical compound CC(N)OC1=CC=CC=C1 QYYKZVMRNIHICX-UHFFFAOYSA-N 0.000 description 2
- WWKRJZZIOIXWMN-UHFFFAOYSA-N 10-aminodecyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCN WWKRJZZIOIXWMN-UHFFFAOYSA-N 0.000 description 2
- GHGMVUIQGHORMP-UHFFFAOYSA-N 10-aminodecyl prop-2-enoate Chemical compound C(C=C)(=O)OCCCCCCCCCCN GHGMVUIQGHORMP-UHFFFAOYSA-N 0.000 description 2
- PKNHMUUNDHSGKI-UHFFFAOYSA-N 12-aminododecyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCCCN PKNHMUUNDHSGKI-UHFFFAOYSA-N 0.000 description 2
- UKGQUCAKWDITNO-UHFFFAOYSA-N 12-aminododecyl prop-2-enoate Chemical compound NCCCCCCCCCCCCOC(=O)C=C UKGQUCAKWDITNO-UHFFFAOYSA-N 0.000 description 2
- QFASVUCTQSVGKG-UHFFFAOYSA-N 2,3-bis(2-cyclohexyloxypropan-2-yl)phenol Chemical compound C=1C=CC(O)=C(C(C)(C)OC2CCCCC2)C=1C(C)(C)OC1CCCCC1 QFASVUCTQSVGKG-UHFFFAOYSA-N 0.000 description 2
- ZKSAJVBCOJFDMT-UHFFFAOYSA-N 2,3-bis(3-cyclohexyloxypentan-3-yl)phenol Chemical compound C=1C=CC(O)=C(C(CC)(CC)OC2CCCCC2)C=1C(CC)(CC)OC1CCCCC1 ZKSAJVBCOJFDMT-UHFFFAOYSA-N 0.000 description 2
- JIVGSHFYXPRRSZ-UHFFFAOYSA-N 2,3-dimethoxybenzaldehyde Chemical compound COC1=CC=CC(C=O)=C1OC JIVGSHFYXPRRSZ-UHFFFAOYSA-N 0.000 description 2
- ZVDSMYGTJDFNHN-UHFFFAOYSA-N 2,4,6-trimethylbenzene-1,3-diamine Chemical group CC1=CC(C)=C(N)C(C)=C1N ZVDSMYGTJDFNHN-UHFFFAOYSA-N 0.000 description 2
- NXXYKOUNUYWIHA-UHFFFAOYSA-N 2,6-Dimethylphenol Chemical compound CC1=CC=CC(C)=C1O NXXYKOUNUYWIHA-UHFFFAOYSA-N 0.000 description 2
- GOJFAKBEASOYNM-UHFFFAOYSA-N 2-(2-aminophenoxy)aniline Chemical compound NC1=CC=CC=C1OC1=CC=CC=C1N GOJFAKBEASOYNM-UHFFFAOYSA-N 0.000 description 2
- WCASXYBKJHWFMY-NSCUHMNNSA-N 2-Buten-1-ol Chemical compound C\C=C\CO WCASXYBKJHWFMY-NSCUHMNNSA-N 0.000 description 2
- FYDMBFIJIQPURU-UHFFFAOYSA-N 2-[2-(2-aminophenyl)propan-2-yl]aniline Chemical compound C=1C=CC=C(N)C=1C(C)(C)C1=CC=CC=C1N FYDMBFIJIQPURU-UHFFFAOYSA-N 0.000 description 2
- XVCLWTSDDBFRET-UHFFFAOYSA-N 2-aminoethyl 2,5-diaminopentanoate Chemical compound NCCOC(C(CCCN)N)=O XVCLWTSDDBFRET-UHFFFAOYSA-N 0.000 description 2
- RHMJDCIYQZFNMK-UHFFFAOYSA-N 2-aminoethyl 2,6-diaminohexanoate Chemical compound NCCCCC(N)C(=O)OCCN RHMJDCIYQZFNMK-UHFFFAOYSA-N 0.000 description 2
- UGIJCMNGQCUTPI-UHFFFAOYSA-N 2-aminoethyl prop-2-enoate Chemical compound NCCOC(=O)C=C UGIJCMNGQCUTPI-UHFFFAOYSA-N 0.000 description 2
- QMQCYMRBZCWBNI-UHFFFAOYSA-N 2-aminopropyl 2-methylprop-2-enoate Chemical compound CC(N)COC(=O)C(C)=C QMQCYMRBZCWBNI-UHFFFAOYSA-N 0.000 description 2
- MOEFFSWKSMRFRQ-UHFFFAOYSA-N 2-ethoxyphenol Chemical compound CCOC1=CC=CC=C1O MOEFFSWKSMRFRQ-UHFFFAOYSA-N 0.000 description 2
- AVVVXUXMKWPKAJ-UHFFFAOYSA-N 2-ethyl-4-methylphenol Chemical compound CCC1=CC(C)=CC=C1O AVVVXUXMKWPKAJ-UHFFFAOYSA-N 0.000 description 2
- FIWYWGLEPWBBQU-UHFFFAOYSA-N 2-heptylphenol Chemical compound CCCCCCCC1=CC=CC=C1O FIWYWGLEPWBBQU-UHFFFAOYSA-N 0.000 description 2
- ABMULKFGWTYIIK-UHFFFAOYSA-N 2-hexylphenol Chemical compound CCCCCCC1=CC=CC=C1O ABMULKFGWTYIIK-UHFFFAOYSA-N 0.000 description 2
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 2
- MEEKGULDSDXFCN-UHFFFAOYSA-N 2-pentylphenol Chemical compound CCCCCC1=CC=CC=C1O MEEKGULDSDXFCN-UHFFFAOYSA-N 0.000 description 2
- WRMNZCZEMHIOCP-UHFFFAOYSA-N 2-phenylethanol Chemical compound OCCC1=CC=CC=C1 WRMNZCZEMHIOCP-UHFFFAOYSA-N 0.000 description 2
- LCHYEKKJCUJAKN-UHFFFAOYSA-N 2-propylphenol Chemical compound CCCC1=CC=CC=C1O LCHYEKKJCUJAKN-UHFFFAOYSA-N 0.000 description 2
- RXFCIXRFAJRBSG-UHFFFAOYSA-N 3,2,3-tetramine Chemical compound NCCCNCCNCCCN RXFCIXRFAJRBSG-UHFFFAOYSA-N 0.000 description 2
- OQEBBZSWEGYTPG-UHFFFAOYSA-N 3-aminobutanoic acid Chemical compound CC(N)CC(O)=O OQEBBZSWEGYTPG-UHFFFAOYSA-N 0.000 description 2
- SNCMCDMEYCLVBO-UHFFFAOYSA-N 3-aminopropyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCN SNCMCDMEYCLVBO-UHFFFAOYSA-N 0.000 description 2
- OTKLRHWBZHQJOP-UHFFFAOYSA-N 3-aminopropyl prop-2-enoate Chemical compound NCCCOC(=O)C=C OTKLRHWBZHQJOP-UHFFFAOYSA-N 0.000 description 2
- GBMGXXPFNLJLMP-UHFFFAOYSA-N 3-ethoxybenzene-1,2-diamine Chemical compound CCOC1=CC=CC(N)=C1N GBMGXXPFNLJLMP-UHFFFAOYSA-N 0.000 description 2
- KDHWOCLBMVSZPG-UHFFFAOYSA-N 3-imidazol-1-ylpropan-1-amine Chemical compound NCCCN1C=CN=C1 KDHWOCLBMVSZPG-UHFFFAOYSA-N 0.000 description 2
- BFLWXPJTAKXXKT-UHFFFAOYSA-N 3-methoxybenzene-1,2-diamine Chemical compound COC1=CC=CC(N)=C1N BFLWXPJTAKXXKT-UHFFFAOYSA-N 0.000 description 2
- AOIUPKVEPBMRDZ-UHFFFAOYSA-N 4-aminobutyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCN AOIUPKVEPBMRDZ-UHFFFAOYSA-N 0.000 description 2
- IEOMKERTJQIKKF-UHFFFAOYSA-N 4-aminobutyl prop-2-enoate Chemical compound NCCCCOC(=O)C=C IEOMKERTJQIKKF-UHFFFAOYSA-N 0.000 description 2
- PAEPSWBIJOCLLK-UHFFFAOYSA-N 5-aminopentyl prop-2-enoate Chemical compound NCCCCCOC(=O)C=C PAEPSWBIJOCLLK-UHFFFAOYSA-N 0.000 description 2
- PGXIJLSNAVMTTA-UHFFFAOYSA-N 6-aminohexyl prop-2-enoate Chemical compound NCCCCCCOC(=O)C=C PGXIJLSNAVMTTA-UHFFFAOYSA-N 0.000 description 2
- CIRRFAQIWQFQSS-UHFFFAOYSA-N 6-ethyl-o-cresol Chemical compound CCC1=CC=CC(C)=C1O CIRRFAQIWQFQSS-UHFFFAOYSA-N 0.000 description 2
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- IRIAEXORFWYRCZ-UHFFFAOYSA-N Butylbenzyl phthalate Chemical compound CCCCOC(=O)C1=CC=CC=C1C(=O)OCC1=CC=CC=C1 IRIAEXORFWYRCZ-UHFFFAOYSA-N 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- RGSFGYAAUTVSQA-UHFFFAOYSA-N Cyclopentane Chemical compound C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 2
- KCXZNSGUUQJJTR-UHFFFAOYSA-N Di-n-hexyl phthalate Chemical compound CCCCCCOC(=O)C1=CC=CC=C1C(=O)OCCCCCC KCXZNSGUUQJJTR-UHFFFAOYSA-N 0.000 description 2
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 description 2
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N Ethylbenzene Chemical compound CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 description 2
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 2
- PYVHTIWHNXTVPF-UHFFFAOYSA-N F.F.F.F.C=C Chemical compound F.F.F.F.C=C PYVHTIWHNXTVPF-UHFFFAOYSA-N 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 2
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 2
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 2
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 2
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 2
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 2
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 2
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 2
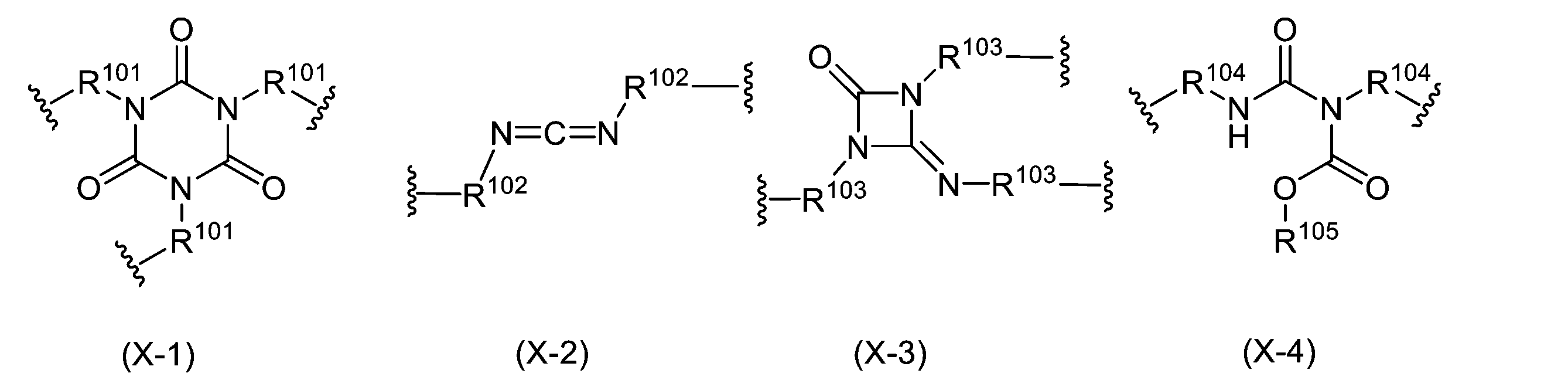
- 150000007945 N-acyl ureas Chemical class 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- 101100156282 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) vib-1 gene Proteins 0.000 description 2
- 239000002033 PVDF binder Substances 0.000 description 2
- 229920001774 Perfluoroether Polymers 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 229920005830 Polyurethane Foam Polymers 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- WUGQZFFCHPXWKQ-UHFFFAOYSA-N Propanolamine Chemical compound NCCCO WUGQZFFCHPXWKQ-UHFFFAOYSA-N 0.000 description 2
- NBBJYMSMWIIQGU-UHFFFAOYSA-N Propionic aldehyde Chemical compound CCC=O NBBJYMSMWIIQGU-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 2
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical group [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- JDPAVWAQGBGGHD-UHFFFAOYSA-N aceanthrylene Chemical group C1=CC=C2C(C=CC3=CC=C4)=C3C4=CC2=C1 JDPAVWAQGBGGHD-UHFFFAOYSA-N 0.000 description 2
- 125000004054 acenaphthylenyl group Chemical group C1(=CC2=CC=CC3=CC=CC1=C23)* 0.000 description 2
- SQFPKRNUGBRTAR-UHFFFAOYSA-N acephenanthrylene Chemical group C1=CC(C=C2)=C3C2=CC2=CC=CC=C2C3=C1 SQFPKRNUGBRTAR-UHFFFAOYSA-N 0.000 description 2
- YRKCREAYFQTBPV-UHFFFAOYSA-N acetylacetone Chemical compound CC(=O)CC(C)=O YRKCREAYFQTBPV-UHFFFAOYSA-N 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 2
- 125000004171 alkoxy aryl group Chemical group 0.000 description 2
- OOCCDEMITAIZTP-UHFFFAOYSA-N allylic benzylic alcohol Natural products OCC=CC1=CC=CC=C1 OOCCDEMITAIZTP-UHFFFAOYSA-N 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 150000001414 amino alcohols Chemical class 0.000 description 2
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 2
- 125000001204 arachidyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 150000004984 aromatic diamines Chemical class 0.000 description 2
- 235000009582 asparagine Nutrition 0.000 description 2
- 229960001230 asparagine Drugs 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 125000003828 azulenyl group Chemical group 0.000 description 2
- 229910052788 barium Inorganic materials 0.000 description 2
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 2
- QVQLCTNNEUAWMS-UHFFFAOYSA-N barium oxide Chemical compound [Ba]=O QVQLCTNNEUAWMS-UHFFFAOYSA-N 0.000 description 2
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 2
- RUOKPLVTMFHRJE-UHFFFAOYSA-N benzene-1,2,3-triamine Chemical compound NC1=CC=CC(N)=C1N RUOKPLVTMFHRJE-UHFFFAOYSA-N 0.000 description 2
- HFACYLZERDEVSX-UHFFFAOYSA-N benzidine Chemical group C1=CC(N)=CC=C1C1=CC=C(N)C=C1 HFACYLZERDEVSX-UHFFFAOYSA-N 0.000 description 2
- UCMIRNVEIXFBKS-UHFFFAOYSA-N beta-alanine Chemical compound NCCC(O)=O UCMIRNVEIXFBKS-UHFFFAOYSA-N 0.000 description 2
- 239000004305 biphenyl Substances 0.000 description 2
- 235000010290 biphenyl Nutrition 0.000 description 2
- LLEMOWNGBBNAJR-UHFFFAOYSA-N biphenyl-2-ol Chemical compound OC1=CC=CC=C1C1=CC=CC=C1 LLEMOWNGBBNAJR-UHFFFAOYSA-N 0.000 description 2
- 125000002529 biphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C12)* 0.000 description 2
- NWSMTZKOVRLRFA-UHFFFAOYSA-N bis(2-aminoethyl) 2-aminobutanedioate Chemical compound NCCOC(=O)CC(N)C(=O)OCCN NWSMTZKOVRLRFA-UHFFFAOYSA-N 0.000 description 2
- MYMAOTWUDGDSNS-UHFFFAOYSA-N bis(2-aminoethyl) 2-aminopentanedioate Chemical compound NCCOC(C(CCC(=O)OCCN)N)=O MYMAOTWUDGDSNS-UHFFFAOYSA-N 0.000 description 2
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 2
- QARVLSVVCXYDNA-UHFFFAOYSA-N bromobenzene Chemical compound BrC1=CC=CC=C1 QARVLSVVCXYDNA-UHFFFAOYSA-N 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- YCIMNLLNPGFGHC-UHFFFAOYSA-N catechol Chemical compound OC1=CC=CC=C1O YCIMNLLNPGFGHC-UHFFFAOYSA-N 0.000 description 2
- 238000006757 chemical reactions by type Methods 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 2
- 125000005578 chrysene group Chemical group 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- QTMDXZNDVAMKGV-UHFFFAOYSA-L copper(ii) bromide Chemical compound [Cu+2].[Br-].[Br-] QTMDXZNDVAMKGV-UHFFFAOYSA-L 0.000 description 2
- XRLHAJCIEMOBLT-UHFFFAOYSA-N cyclobutane-1,1-diamine Chemical compound NC1(N)CCC1 XRLHAJCIEMOBLT-UHFFFAOYSA-N 0.000 description 2
- YMHQVDAATAEZLO-UHFFFAOYSA-N cyclohexane-1,1-diamine Chemical compound NC1(N)CCCCC1 YMHQVDAATAEZLO-UHFFFAOYSA-N 0.000 description 2
- DIOQZVSQGTUSAI-UHFFFAOYSA-N decane Chemical compound CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 2
- OWEZJUPKTBEISC-UHFFFAOYSA-N decane-1,1-diamine Chemical compound CCCCCCCCCC(N)N OWEZJUPKTBEISC-UHFFFAOYSA-N 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 150000004985 diamines Chemical class 0.000 description 2
- DOIRQSBPFJWKBE-UHFFFAOYSA-N dibutyl phthalate Chemical compound CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 description 2
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 2
- JXCHMDATRWUOAP-UHFFFAOYSA-N diisocyanatomethylbenzene Chemical compound O=C=NC(N=C=O)C1=CC=CC=C1 JXCHMDATRWUOAP-UHFFFAOYSA-N 0.000 description 2
- IUNMPGNGSSIWFP-UHFFFAOYSA-N dimethylaminopropylamine Chemical compound CN(C)CCCN IUNMPGNGSSIWFP-UHFFFAOYSA-N 0.000 description 2
- ROORDVPLFPIABK-UHFFFAOYSA-N diphenyl carbonate Chemical compound C=1C=CC=CC=1OC(=O)OC1=CC=CC=C1 ROORDVPLFPIABK-UHFFFAOYSA-N 0.000 description 2
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 2
- 230000009977 dual effect Effects 0.000 description 2
- JBKVHLHDHHXQEQ-UHFFFAOYSA-N epsilon-caprolactam Chemical compound O=C1CCCCCN1 JBKVHLHDHHXQEQ-UHFFFAOYSA-N 0.000 description 2
- 125000006232 ethoxy propyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000005448 ethoxyethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])C([H])([H])* 0.000 description 2
- 125000005745 ethoxymethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])* 0.000 description 2
- MTZQAGJQAFMTAQ-UHFFFAOYSA-N ethyl benzoate Chemical compound CCOC(=O)C1=CC=CC=C1 MTZQAGJQAFMTAQ-UHFFFAOYSA-N 0.000 description 2
- IIEWJVIFRVWJOD-UHFFFAOYSA-N ethylcyclohexane Chemical compound CCC1CCCCC1 IIEWJVIFRVWJOD-UHFFFAOYSA-N 0.000 description 2
- 239000010408 film Substances 0.000 description 2
- 238000010304 firing Methods 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- HYBBIBNJHNGZAN-UHFFFAOYSA-N furfural Chemical compound O=CC1=CC=CO1 HYBBIBNJHNGZAN-UHFFFAOYSA-N 0.000 description 2
- WCASXYBKJHWFMY-UHFFFAOYSA-N gamma-methylallyl alcohol Natural products CC=CCO WCASXYBKJHWFMY-UHFFFAOYSA-N 0.000 description 2
- 239000007792 gaseous phase Substances 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- 235000004554 glutamine Nutrition 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 150000008282 halocarbons Chemical class 0.000 description 2
- 229910052736 halogen Inorganic materials 0.000 description 2
- 150000002367 halogens Chemical class 0.000 description 2
- 125000002192 heptalenyl group Chemical group 0.000 description 2
- JARKCYVAAOWBJS-UHFFFAOYSA-N hexanal Chemical compound CCCCCC=O JARKCYVAAOWBJS-UHFFFAOYSA-N 0.000 description 2
- 125000003707 hexyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 2
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 2
- 150000002466 imines Chemical class 0.000 description 2
- 125000003427 indacenyl group Chemical group 0.000 description 2
- 150000002484 inorganic compounds Chemical class 0.000 description 2
- 229910010272 inorganic material Inorganic materials 0.000 description 2
- 229910003480 inorganic solid Inorganic materials 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- 125000002960 margaryl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- AUHZEENZYGFFBQ-UHFFFAOYSA-N mesitylene Substances CC1=CC(C)=CC(C)=C1 AUHZEENZYGFFBQ-UHFFFAOYSA-N 0.000 description 2
- 125000001827 mesitylenyl group Chemical group [H]C1=C(C(*)=C(C([H])=C1C([H])([H])[H])C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- KPNBUPJZFJCCIQ-LURJTMIESA-N methyl L-lysinate Chemical compound COC(=O)[C@@H](N)CCCCN KPNBUPJZFJCCIQ-LURJTMIESA-N 0.000 description 2
- GTCAXTIRRLKXRU-UHFFFAOYSA-N methyl carbamate Chemical compound COC(N)=O GTCAXTIRRLKXRU-UHFFFAOYSA-N 0.000 description 2
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- QOHMWDJIBGVPIF-UHFFFAOYSA-N n',n'-diethylpropane-1,3-diamine Chemical compound CCN(CC)CCCN QOHMWDJIBGVPIF-UHFFFAOYSA-N 0.000 description 2
- DTSDBGVDESRKKD-UHFFFAOYSA-N n'-(2-aminoethyl)propane-1,3-diamine Chemical compound NCCCNCCN DTSDBGVDESRKKD-UHFFFAOYSA-N 0.000 description 2
- ITZPOSYADVYECJ-UHFFFAOYSA-N n'-cyclohexylpropane-1,3-diamine Chemical compound NCCCNC1CCCCC1 ITZPOSYADVYECJ-UHFFFAOYSA-N 0.000 description 2
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 2
- NTNWKDHZTDQSST-UHFFFAOYSA-N naphthalene-1,2-diamine Chemical compound C1=CC=CC2=C(N)C(N)=CC=C21 NTNWKDHZTDQSST-UHFFFAOYSA-N 0.000 description 2
- SLCVBVWXLSEKPL-UHFFFAOYSA-N neopentyl glycol Chemical compound OCC(C)(C)CO SLCVBVWXLSEKPL-UHFFFAOYSA-N 0.000 description 2
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 2
- LQNUZADURLCDLV-UHFFFAOYSA-N nitrobenzene Chemical compound [O-][N+](=O)C1=CC=CC=C1 LQNUZADURLCDLV-UHFFFAOYSA-N 0.000 description 2
- 125000001196 nonadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- BKIMMITUMNQMOS-UHFFFAOYSA-N nonane Chemical compound CCCCCCCCC BKIMMITUMNQMOS-UHFFFAOYSA-N 0.000 description 2
- 229910052755 nonmetal Inorganic materials 0.000 description 2
- NUJGJRNETVAIRJ-UHFFFAOYSA-N octanal Chemical compound CCCCCCCC=O NUJGJRNETVAIRJ-UHFFFAOYSA-N 0.000 description 2
- 150000002894 organic compounds Chemical class 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 150000002902 organometallic compounds Chemical class 0.000 description 2
- 239000003973 paint Substances 0.000 description 2
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000002958 pentadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 2
- GUVXZFRDPCKWEM-UHFFFAOYSA-N pentalene group Chemical group C1=CC=C2C=CC=C12 GUVXZFRDPCKWEM-UHFFFAOYSA-N 0.000 description 2
- GPCKFIWBUTWTDH-UHFFFAOYSA-N pentane-3,3-diamine Chemical compound CCC(N)(N)CC GPCKFIWBUTWTDH-UHFFFAOYSA-N 0.000 description 2
- 230000000737 periodic effect Effects 0.000 description 2
- YNPNZTXNASCQKK-UHFFFAOYSA-N phenanthrene Chemical group C1=CC=C2C3=CC=CC=C3C=CC2=C1 YNPNZTXNASCQKK-UHFFFAOYSA-N 0.000 description 2
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 2
- DTUQWGWMVIHBKE-UHFFFAOYSA-N phenylacetaldehyde Chemical compound O=CCC1=CC=CC=C1 DTUQWGWMVIHBKE-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 125000000109 phenylethoxy group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])O* 0.000 description 2
- MEGDIQXLXPPWGL-UHFFFAOYSA-N phenylmethanetriamine Chemical compound NC(N)(N)C1=CC=CC=C1 MEGDIQXLXPPWGL-UHFFFAOYSA-N 0.000 description 2
- 125000004344 phenylpropyl group Chemical group 0.000 description 2
- RGCLLPNLLBQHPF-HJWRWDBZSA-N phosphamidon Chemical compound CCN(CC)C(=O)C(\Cl)=C(/C)OP(=O)(OC)OC RGCLLPNLLBQHPF-HJWRWDBZSA-N 0.000 description 2
- 229920002493 poly(chlorotrifluoroethylene) Polymers 0.000 description 2
- 239000005023 polychlorotrifluoroethylene (PCTFE) polymer Substances 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 description 2
- 239000011496 polyurethane foam Substances 0.000 description 2
- 229920002620 polyvinyl fluoride Polymers 0.000 description 2
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- TVDSBUOJIPERQY-UHFFFAOYSA-N prop-2-yn-1-ol Chemical compound OCC#C TVDSBUOJIPERQY-UHFFFAOYSA-N 0.000 description 2
- ZNZJJSYHZBXQSM-UHFFFAOYSA-N propane-2,2-diamine Chemical compound CC(C)(N)N ZNZJJSYHZBXQSM-UHFFFAOYSA-N 0.000 description 2
- 125000005581 pyrene group Chemical group 0.000 description 2
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 2
- 230000008929 regeneration Effects 0.000 description 2
- 238000011069 regeneration method Methods 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- GHMLBKRAJCXXBS-UHFFFAOYSA-N resorcinol Chemical compound OC1=CC=CC(O)=C1 GHMLBKRAJCXXBS-UHFFFAOYSA-N 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 2
- 239000004065 semiconductor Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000011343 solid material Substances 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 238000003883 substance clean up Methods 0.000 description 2
- 125000001935 tetracenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC=CC=C4C=C3C=C12)* 0.000 description 2
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 2
- TXEYQDLBPFQVAA-UHFFFAOYSA-N tetrafluoromethane Chemical compound FC(F)(F)F TXEYQDLBPFQVAA-UHFFFAOYSA-N 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- 239000011135 tin Substances 0.000 description 2
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 2
- 231100000331 toxic Toxicity 0.000 description 2
- 230000002588 toxic effect Effects 0.000 description 2
- 150000003623 transition metal compounds Chemical class 0.000 description 2
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000005580 triphenylene group Chemical group 0.000 description 2
- CKANOPZMLBOZNJ-UHFFFAOYSA-N tris(2-aminoethyl) hexane-1,3,6-tricarboxylate Chemical compound NCCOC(=O)CCC(CCCC(=O)OCCN)C(=O)OCCN CKANOPZMLBOZNJ-UHFFFAOYSA-N 0.000 description 2
- WNVQBUHCOYRLPA-UHFFFAOYSA-N triuret Chemical compound NC(=O)NC(=O)NC(N)=O WNVQBUHCOYRLPA-UHFFFAOYSA-N 0.000 description 2
- 229940057402 undecyl alcohol Drugs 0.000 description 2
- 125000002948 undecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 239000004474 valine Substances 0.000 description 2
- 239000010457 zeolite Substances 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- DYLIWHYUXAJDOJ-OWOJBTEDSA-N (e)-4-(6-aminopurin-9-yl)but-2-en-1-ol Chemical compound NC1=NC=NC2=C1N=CN2C\C=C\CO DYLIWHYUXAJDOJ-OWOJBTEDSA-N 0.000 description 1
- VCGQROFHUSXQBU-UHFFFAOYSA-N 1,2,3-tris(3-isocyanatopropyl)benzene Chemical compound N(=C=O)CCCC=1C(=C(C=CC=1)CCCN=C=O)CCCN=C=O VCGQROFHUSXQBU-UHFFFAOYSA-N 0.000 description 1
- QMEVBXBOTMIPJZ-UHFFFAOYSA-N 1,2,3-tris(isocyanatomethyl)-4-methylbenzene Chemical compound N(=C=O)CC1=C(C(=C(C=C1)C)CN=C=O)CN=C=O QMEVBXBOTMIPJZ-UHFFFAOYSA-N 0.000 description 1
- OKIRBHVFJGXOIS-UHFFFAOYSA-N 1,2-di(propan-2-yl)benzene Chemical compound CC(C)C1=CC=CC=C1C(C)C OKIRBHVFJGXOIS-UHFFFAOYSA-N 0.000 description 1
- PKQYSCBUFZOAPE-UHFFFAOYSA-N 1,2-dibenzyl-3-methylbenzene Chemical compound C=1C=CC=CC=1CC=1C(C)=CC=CC=1CC1=CC=CC=C1 PKQYSCBUFZOAPE-UHFFFAOYSA-N 0.000 description 1
- WQONPSCCEXUXTQ-UHFFFAOYSA-N 1,2-dibromobenzene Chemical compound BrC1=CC=CC=C1Br WQONPSCCEXUXTQ-UHFFFAOYSA-N 0.000 description 1
- NMUWSGQKPAEPBA-UHFFFAOYSA-N 1,2-dibutylbenzene Chemical compound CCCCC1=CC=CC=C1CCCC NMUWSGQKPAEPBA-UHFFFAOYSA-N 0.000 description 1
- RAZWADXTNBRANC-UHFFFAOYSA-N 1,2-dimethylcyclohexan-1-ol Chemical compound CC1CCCCC1(C)O RAZWADXTNBRANC-UHFFFAOYSA-N 0.000 description 1
- OCJBOOLMMGQPQU-UHFFFAOYSA-N 1,4-dichlorobenzene Chemical compound ClC1=CC=C(Cl)C=C1 OCJBOOLMMGQPQU-UHFFFAOYSA-N 0.000 description 1
- GGMFZKQJNPNNRS-UHFFFAOYSA-N 1-(diisocyanatomethyl)naphthalene Chemical compound C1=CC=C2C(C(N=C=O)N=C=O)=CC=CC2=C1 GGMFZKQJNPNNRS-UHFFFAOYSA-N 0.000 description 1
- SBSMXBFGGRDASE-UHFFFAOYSA-N 1-[2-[(dimethylamino)methyl]phenyl]-n,n-dimethylmethanamine Chemical compound CN(C)CC1=CC=CC=C1CN(C)C SBSMXBFGGRDASE-UHFFFAOYSA-N 0.000 description 1
- DLKQHBOKULLWDQ-UHFFFAOYSA-N 1-bromonaphthalene Chemical compound C1=CC=C2C(Br)=CC=CC2=C1 DLKQHBOKULLWDQ-UHFFFAOYSA-N 0.000 description 1
- RCHLXMOXBJRGNX-UHFFFAOYSA-N 1-butylcyclohexan-1-ol Chemical compound CCCCC1(O)CCCCC1 RCHLXMOXBJRGNX-UHFFFAOYSA-N 0.000 description 1
- JTPNRXUCIXHOKM-UHFFFAOYSA-N 1-chloronaphthalene Chemical compound C1=CC=C2C(Cl)=CC=CC2=C1 JTPNRXUCIXHOKM-UHFFFAOYSA-N 0.000 description 1
- BUCJHJXFXUZJHL-UHFFFAOYSA-N 1-ethylcyclohexan-1-ol Chemical compound CCC1(O)CCCCC1 BUCJHJXFXUZJHL-UHFFFAOYSA-N 0.000 description 1
- LPCWIFPJLFCXRS-UHFFFAOYSA-N 1-ethylcyclopentan-1-ol Chemical compound CCC1(O)CCCC1 LPCWIFPJLFCXRS-UHFFFAOYSA-N 0.000 description 1
- FHCVFYFOWIAQDU-UHFFFAOYSA-N 1-hexylcyclohexan-1-ol Chemical compound CCCCCCC1(O)CCCCC1 FHCVFYFOWIAQDU-UHFFFAOYSA-N 0.000 description 1
- KYZJYDSSXRSBDB-UHFFFAOYSA-N 1-isocyanato-2-(2-isocyanatophenoxy)benzene Chemical compound O=C=NC1=CC=CC=C1OC1=CC=CC=C1N=C=O KYZJYDSSXRSBDB-UHFFFAOYSA-N 0.000 description 1
- JIABEENURMZTTI-UHFFFAOYSA-N 1-isocyanato-2-[(2-isocyanatophenyl)methyl]benzene Chemical compound O=C=NC1=CC=CC=C1CC1=CC=CC=C1N=C=O JIABEENURMZTTI-UHFFFAOYSA-N 0.000 description 1
- PYWYAAZCDBLBAN-UHFFFAOYSA-N 1-isocyanato-2-[2-(2-isocyanatophenyl)propan-2-yl]benzene Chemical compound C=1C=CC=C(N=C=O)C=1C(C)(C)C1=CC=CC=C1N=C=O PYWYAAZCDBLBAN-UHFFFAOYSA-N 0.000 description 1
- RRPOMRAIAKJYKL-UHFFFAOYSA-N 1-isocyanatoethoxybenzene Chemical compound O=C=NC(C)OC1=CC=CC=C1 RRPOMRAIAKJYKL-UHFFFAOYSA-N 0.000 description 1
- CAKWRXVKWGUISE-UHFFFAOYSA-N 1-methylcyclopentan-1-ol Chemical compound CC1(O)CCCC1 CAKWRXVKWGUISE-UHFFFAOYSA-N 0.000 description 1
- GNHXRDSGBDYEAF-UHFFFAOYSA-N 1-methylnaphthalene-2,3-diamine Chemical compound C1=CC=C2C(C)=C(N)C(N)=CC2=C1 GNHXRDSGBDYEAF-UHFFFAOYSA-N 0.000 description 1
- KJCVRFUGPWSIIH-UHFFFAOYSA-N 1-naphthol Chemical compound C1=CC=C2C(O)=CC=CC2=C1 KJCVRFUGPWSIIH-UHFFFAOYSA-N 0.000 description 1
- RUFPHBVGCFYCNW-UHFFFAOYSA-N 1-naphthylamine Chemical compound C1=CC=C2C(N)=CC=CC2=C1 RUFPHBVGCFYCNW-UHFFFAOYSA-N 0.000 description 1
- RJKGJBPXVHTNJL-UHFFFAOYSA-N 1-nitronaphthalene Chemical compound C1=CC=C2C([N+](=O)[O-])=CC=CC2=C1 RJKGJBPXVHTNJL-UHFFFAOYSA-N 0.000 description 1
- QKMOFWOJEZZHFD-UHFFFAOYSA-N 1-pentylcyclohexan-1-ol Chemical compound CCCCCC1(O)CCCCC1 QKMOFWOJEZZHFD-UHFFFAOYSA-N 0.000 description 1
- HQRWWHIETAKIMO-UHFFFAOYSA-N 1-phenylbutan-1-ol Chemical compound CCCC(O)C1=CC=CC=C1 HQRWWHIETAKIMO-UHFFFAOYSA-N 0.000 description 1
- UAJVCELPUNHGKE-UHFFFAOYSA-N 1-phenylheptan-1-ol Chemical compound CCCCCCC(O)C1=CC=CC=C1 UAJVCELPUNHGKE-UHFFFAOYSA-N 0.000 description 1
- KBXJSWRVYBTZPC-UHFFFAOYSA-N 1-phenylnonan-1-ol Chemical compound CCCCCCCCC(O)C1=CC=CC=C1 KBXJSWRVYBTZPC-UHFFFAOYSA-N 0.000 description 1
- MNBCPXKENXMEEJ-UHFFFAOYSA-N 1-phenyloctan-1-ol Chemical compound CCCCCCCC(O)C1=CC=CC=C1 MNBCPXKENXMEEJ-UHFFFAOYSA-N 0.000 description 1
- PYLPYOPJKOJRNP-UHFFFAOYSA-N 1-propylcyclohexan-1-ol Chemical compound CCCC1(O)CCCCC1 PYLPYOPJKOJRNP-UHFFFAOYSA-N 0.000 description 1
- RSVLLJMCLGUKLM-UHFFFAOYSA-N 10-isocyanatodecyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCN=C=O RSVLLJMCLGUKLM-UHFFFAOYSA-N 0.000 description 1
- QXOJEFDVHQCJNH-UHFFFAOYSA-N 10-isocyanatodecyl prop-2-enoate Chemical compound C=CC(=O)OCCCCCCCCCCN=C=O QXOJEFDVHQCJNH-UHFFFAOYSA-N 0.000 description 1
- YIZNDHIAODOIBD-UHFFFAOYSA-N 11-isocyanatoundecyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCCN=C=O YIZNDHIAODOIBD-UHFFFAOYSA-N 0.000 description 1
- PBLZLIFKVPJDCO-UHFFFAOYSA-N 12-aminododecanoic acid Chemical compound NCCCCCCCCCCCC(O)=O PBLZLIFKVPJDCO-UHFFFAOYSA-N 0.000 description 1
- WEOHWYYCMGEAQY-UHFFFAOYSA-N 12-isocyanatododecyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCCCN=C=O WEOHWYYCMGEAQY-UHFFFAOYSA-N 0.000 description 1
- FLBILZJKOCDIBX-UHFFFAOYSA-N 12-isocyanatododecyl prop-2-enoate Chemical compound C=CC(=O)OCCCCCCCCCCCCN=C=O FLBILZJKOCDIBX-UHFFFAOYSA-N 0.000 description 1
- KGRVJHAUYBGFFP-UHFFFAOYSA-N 2,2'-Methylenebis(4-methyl-6-tert-butylphenol) Chemical compound CC(C)(C)C1=CC(C)=CC(CC=2C(=C(C=C(C)C=2)C(C)(C)C)O)=C1O KGRVJHAUYBGFFP-UHFFFAOYSA-N 0.000 description 1
- HSQFVBWFPBKHEB-UHFFFAOYSA-N 2,3,4-trichlorophenol Chemical compound OC1=CC=C(Cl)C(Cl)=C1Cl HSQFVBWFPBKHEB-UHFFFAOYSA-N 0.000 description 1
- AFXQOXNRTJFOJV-UHFFFAOYSA-N 2,3,4-triethylphenol Chemical compound CCC1=CC=C(O)C(CC)=C1CC AFXQOXNRTJFOJV-UHFFFAOYSA-N 0.000 description 1
- UAJDPHHQCAYKRZ-UHFFFAOYSA-N 2,3,4-trimethylnaphthalen-1-amine Chemical compound C1=CC=CC2=C(C)C(C)=C(C)C(N)=C21 UAJDPHHQCAYKRZ-UHFFFAOYSA-N 0.000 description 1
- XRUGBBIQLIVCSI-UHFFFAOYSA-N 2,3,4-trimethylphenol Chemical compound CC1=CC=C(O)C(C)=C1C XRUGBBIQLIVCSI-UHFFFAOYSA-N 0.000 description 1
- YQHONIHINWAAFC-UHFFFAOYSA-N 2,3,4-tris(1-cyclohexyloxyethyl)phenol Chemical compound C=1C=C(O)C(C(C)OC2CCCCC2)=C(C(C)OC2CCCCC2)C=1C(C)OC1CCCCC1 YQHONIHINWAAFC-UHFFFAOYSA-N 0.000 description 1
- RXVHWFLTCLUXEV-UHFFFAOYSA-N 2,3,4-tris(1-cyclopentyloxyethyl)phenol Chemical compound C=1C=C(O)C(C(C)OC2CCCC2)=C(C(C)OC2CCCC2)C=1C(C)OC1CCCC1 RXVHWFLTCLUXEV-UHFFFAOYSA-N 0.000 description 1
- AVCGSBFXSSMYIV-UHFFFAOYSA-N 2,3,4-tris(1-cyclopentyloxypropyl)phenol Chemical compound C=1C=C(O)C(C(CC)OC2CCCC2)=C(C(CC)OC2CCCC2)C=1C(CC)OC1CCCC1 AVCGSBFXSSMYIV-UHFFFAOYSA-N 0.000 description 1
- JIUPIGIMTHGWSN-UHFFFAOYSA-N 2,3,4-tris(cyclohexyloxymethyl)phenol Chemical compound C1CCCCC1OCC1=C(COC2CCCCC2)C(O)=CC=C1COC1CCCCC1 JIUPIGIMTHGWSN-UHFFFAOYSA-N 0.000 description 1
- AUNJZVHNNMAWBI-UHFFFAOYSA-N 2,3,4-tris(ethoxymethyl)phenol Chemical compound CCOCC1=CC=C(O)C(COCC)=C1COCC AUNJZVHNNMAWBI-UHFFFAOYSA-N 0.000 description 1
- CRYHSFLDCJOGMP-UHFFFAOYSA-N 2,3,4-tris(methoxymethyl)phenol Chemical compound COCC1=CC=C(O)C(COC)=C1COC CRYHSFLDCJOGMP-UHFFFAOYSA-N 0.000 description 1
- NLPUNRLMQUKGHD-UHFFFAOYSA-N 2,3-bis(1-cyclopentyloxyethyl)phenol Chemical compound C=1C=CC(O)=C(C(C)OC2CCCC2)C=1C(C)OC1CCCC1 NLPUNRLMQUKGHD-UHFFFAOYSA-N 0.000 description 1
- DQQIVDYKBXCIHV-UHFFFAOYSA-N 2,3-bis(1-phenoxybutyl)phenol Chemical compound C=1C=CC(O)=C(C(CCC)OC=2C=CC=CC=2)C=1C(CCC)OC1=CC=CC=C1 DQQIVDYKBXCIHV-UHFFFAOYSA-N 0.000 description 1
- MIGXIFIKYPYBNQ-UHFFFAOYSA-N 2,3-bis(1-phenoxyethyl)phenol Chemical compound C=1C=CC(O)=C(C(C)OC=2C=CC=CC=2)C=1C(C)OC1=CC=CC=C1 MIGXIFIKYPYBNQ-UHFFFAOYSA-N 0.000 description 1
- ROZVFKZVOBITTM-UHFFFAOYSA-N 2,3-bis(1-phenoxypropyl)phenol Chemical compound C=1C=CC(O)=C(C(CC)OC=2C=CC=CC=2)C=1C(CC)OC1=CC=CC=C1 ROZVFKZVOBITTM-UHFFFAOYSA-N 0.000 description 1
- MGVDORSODDQAEP-UHFFFAOYSA-N 2,3-bis(2-phenoxypropan-2-yl)phenol Chemical compound C=1C=CC(O)=C(C(C)(C)OC=2C=CC=CC=2)C=1C(C)(C)OC1=CC=CC=C1 MGVDORSODDQAEP-UHFFFAOYSA-N 0.000 description 1
- DHVMUNUTYVCVPV-UHFFFAOYSA-N 2,3-bis(2-phenylethoxymethyl)phenol Chemical compound C=1C=CC=CC=1CCOCC=1C(O)=CC=CC=1COCCC1=CC=CC=C1 DHVMUNUTYVCVPV-UHFFFAOYSA-N 0.000 description 1
- PSSTXNKDBDKMJV-UHFFFAOYSA-N 2,3-bis(3-phenoxypentan-3-yl)phenol Chemical compound C=1C=CC(O)=C(C(CC)(CC)OC=2C=CC=CC=2)C=1C(CC)(CC)OC1=CC=CC=C1 PSSTXNKDBDKMJV-UHFFFAOYSA-N 0.000 description 1
- WPGIEDHJUPPZEG-UHFFFAOYSA-N 2,3-bis(3-phenylpropoxymethyl)phenol Chemical compound C=1C=CC=CC=1CCCOCC=1C(O)=CC=CC=1COCCCC1=CC=CC=C1 WPGIEDHJUPPZEG-UHFFFAOYSA-N 0.000 description 1
- DGMXMEXXKMFTCR-UHFFFAOYSA-N 2,3-bis(4-phenoxyheptan-4-yl)phenol Chemical compound C=1C=CC(O)=C(C(CCC)(CCC)OC=2C=CC=CC=2)C=1C(CCC)(CCC)OC1=CC=CC=C1 DGMXMEXXKMFTCR-UHFFFAOYSA-N 0.000 description 1
- XGEYPRBTHKTTCN-UHFFFAOYSA-N 2,3-bis(5-cyclohexyloxynonan-5-yl)phenol Chemical compound C=1C=CC(O)=C(C(CCCC)(CCCC)OC2CCCCC2)C=1C(CCCC)(CCCC)OC1CCCCC1 XGEYPRBTHKTTCN-UHFFFAOYSA-N 0.000 description 1
- NBKWBVCMAZAHJJ-UHFFFAOYSA-N 2,3-bis(5-phenoxynonan-5-yl)phenol Chemical compound C=1C=CC(O)=C(C(CCCC)(CCCC)OC=2C=CC=CC=2)C=1C(CCCC)(CCCC)OC1=CC=CC=C1 NBKWBVCMAZAHJJ-UHFFFAOYSA-N 0.000 description 1
- JRRQFNCTMDEFGG-UHFFFAOYSA-N 2,3-bis(butoxymethyl)phenol Chemical compound CCCCOCC1=CC=CC(O)=C1COCCCC JRRQFNCTMDEFGG-UHFFFAOYSA-N 0.000 description 1
- GQWHCRLPSIPCIS-UHFFFAOYSA-N 2,3-bis(cyclohexyloxymethyl)phenol Chemical compound C1CCCCC1OCC=1C(O)=CC=CC=1COC1CCCCC1 GQWHCRLPSIPCIS-UHFFFAOYSA-N 0.000 description 1
- OHPRKLGJCWRBDC-UHFFFAOYSA-N 2,3-bis(cyclopentyloxymethyl)phenol Chemical compound C1CCCC1OCC=1C(O)=CC=CC=1COC1CCCC1 OHPRKLGJCWRBDC-UHFFFAOYSA-N 0.000 description 1
- QBCOXNITRDHCNX-UHFFFAOYSA-N 2,3-bis(ethoxymethyl)phenol Chemical compound CCOCC1=CC=CC(O)=C1COCC QBCOXNITRDHCNX-UHFFFAOYSA-N 0.000 description 1
- XWRVPAWRRBUHHO-UHFFFAOYSA-N 2,3-bis(methoxymethyl)phenol Chemical compound COCC1=CC=CC(O)=C1COC XWRVPAWRRBUHHO-UHFFFAOYSA-N 0.000 description 1
- BHTIYWJUXGMOKO-UHFFFAOYSA-N 2,3-bis(phenylmethoxymethyl)phenol Chemical compound C=1C=CC=CC=1COCC=1C(O)=CC=CC=1COCC1=CC=CC=C1 BHTIYWJUXGMOKO-UHFFFAOYSA-N 0.000 description 1
- UKMPOLWIVGIZKU-UHFFFAOYSA-N 2,3-bis(propoxymethyl)phenol Chemical compound CCCOCC1=CC=CC(O)=C1COCCC UKMPOLWIVGIZKU-UHFFFAOYSA-N 0.000 description 1
- KFCXCDAGFZQFSG-UHFFFAOYSA-N 2,3-bis[(2,3,4-triethylphenoxy)methyl]phenol Chemical compound CCC1=C(CC)C(CC)=CC=C1OCC1=CC=CC(O)=C1COC1=CC=C(CC)C(CC)=C1CC KFCXCDAGFZQFSG-UHFFFAOYSA-N 0.000 description 1
- DCIZZZCFXDWSRW-UHFFFAOYSA-N 2,3-bis[(2,3,4-trimethylphenoxy)methyl]phenol Chemical compound CC1=C(C)C(C)=CC=C1OCC1=CC=CC(O)=C1COC1=CC=C(C)C(C)=C1C DCIZZZCFXDWSRW-UHFFFAOYSA-N 0.000 description 1
- QSKGKJKLMYSCJC-UHFFFAOYSA-N 2,3-bis[phenoxy(diphenyl)methyl]phenol Chemical compound OC1=CC=CC(C(OC=2C=CC=CC=2)(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1C(C=1C=CC=CC=1)(C=1C=CC=CC=1)OC1=CC=CC=C1 QSKGKJKLMYSCJC-UHFFFAOYSA-N 0.000 description 1
- FNAKEOXYWBWIRT-UHFFFAOYSA-N 2,3-dibromophenol Chemical compound OC1=CC=CC(Br)=C1Br FNAKEOXYWBWIRT-UHFFFAOYSA-N 0.000 description 1
- UMPSXRYVXUPCOS-UHFFFAOYSA-N 2,3-dichlorophenol Chemical compound OC1=CC=CC(Cl)=C1Cl UMPSXRYVXUPCOS-UHFFFAOYSA-N 0.000 description 1
- RLEWTHFVGOXXTN-UHFFFAOYSA-N 2,3-diethylphenol Chemical compound CCC1=CC=CC(O)=C1CC RLEWTHFVGOXXTN-UHFFFAOYSA-N 0.000 description 1
- QWBBPBRQALCEIZ-UHFFFAOYSA-N 2,3-dimethylphenol Chemical compound CC1=CC=CC(O)=C1C QWBBPBRQALCEIZ-UHFFFAOYSA-N 0.000 description 1
- BSWWXRFVMJHFBN-UHFFFAOYSA-N 2,4,6-tribromophenol Chemical compound OC1=C(Br)C=C(Br)C=C1Br BSWWXRFVMJHFBN-UHFFFAOYSA-N 0.000 description 1
- KOHSRHXKURUXCP-UHFFFAOYSA-N 2,4,6-triethylphenol Chemical compound CCC1=CC(CC)=C(O)C(CC)=C1 KOHSRHXKURUXCP-UHFFFAOYSA-N 0.000 description 1
- BPRYUXCVCCNUFE-UHFFFAOYSA-N 2,4,6-trimethylphenol Chemical compound CC1=CC(C)=C(O)C(C)=C1 BPRYUXCVCCNUFE-UHFFFAOYSA-N 0.000 description 1
- PHSXXPSPEBSWOK-UHFFFAOYSA-N 2,4,6-tripropylphenol Chemical compound CCCC1=CC(CCC)=C(O)C(CCC)=C1 PHSXXPSPEBSWOK-UHFFFAOYSA-N 0.000 description 1
- FMUYQRFTLHAARI-UHFFFAOYSA-N 2,4-bis(2-phenylpropan-2-yl)phenol Chemical compound C=1C=C(O)C(C(C)(C)C=2C=CC=CC=2)=CC=1C(C)(C)C1=CC=CC=C1 FMUYQRFTLHAARI-UHFFFAOYSA-N 0.000 description 1
- WHBDGWIYZWPCAY-UHFFFAOYSA-N 2,4-dibutyl-6-methylphenol Chemical compound CCCCC1=CC(C)=C(O)C(CCCC)=C1 WHBDGWIYZWPCAY-UHFFFAOYSA-N 0.000 description 1
- KPZBEZVZFBDKCG-UHFFFAOYSA-N 2,4-dibutylphenol Chemical compound CCCCC1=CC=C(O)C(CCCC)=C1 KPZBEZVZFBDKCG-UHFFFAOYSA-N 0.000 description 1
- KVMXOQBZXXCWQA-UHFFFAOYSA-N 2,4-diethyl-6-hexylphenol Chemical compound CCCCCCC1=CC(CC)=CC(CC)=C1O KVMXOQBZXXCWQA-UHFFFAOYSA-N 0.000 description 1
- VUDWBUZLGAXSDX-UHFFFAOYSA-N 2,4-diethyl-6-methylphenol Chemical compound CCC1=CC(C)=C(O)C(CC)=C1 VUDWBUZLGAXSDX-UHFFFAOYSA-N 0.000 description 1
- KHQXLKHARNCXJO-UHFFFAOYSA-N 2,4-diethyl-6-pentylphenol Chemical compound CCCCCC1=CC(CC)=CC(CC)=C1O KHQXLKHARNCXJO-UHFFFAOYSA-N 0.000 description 1
- OPDOEZVOQNNAFP-UHFFFAOYSA-N 2,4-diethyl-6-propylphenol Chemical compound CCCC1=CC(CC)=CC(CC)=C1O OPDOEZVOQNNAFP-UHFFFAOYSA-N 0.000 description 1
- LMLAXOBGXCTWBJ-UHFFFAOYSA-N 2,4-diethylphenol Chemical compound CCC1=CC=C(O)C(CC)=C1 LMLAXOBGXCTWBJ-UHFFFAOYSA-N 0.000 description 1
- OZYHDRHMWRNOID-UHFFFAOYSA-N 2,4-diheptylphenol Chemical compound CCCCCCCC1=CC=C(O)C(CCCCCCC)=C1 OZYHDRHMWRNOID-UHFFFAOYSA-N 0.000 description 1
- LUDSPYUFEAFRRF-UHFFFAOYSA-N 2,4-dihexyl-6-methylphenol Chemical compound CCCCCCC1=CC(C)=C(O)C(CCCCCC)=C1 LUDSPYUFEAFRRF-UHFFFAOYSA-N 0.000 description 1
- KFSJCAKQTAGWBH-UHFFFAOYSA-N 2,4-dihexylphenol Chemical compound CCCCCCC1=CC=C(O)C(CCCCCC)=C1 KFSJCAKQTAGWBH-UHFFFAOYSA-N 0.000 description 1
- LZCXVGMGRORTJD-UHFFFAOYSA-N 2,4-dimethyl-6-pentylphenol Chemical compound CCCCCC1=CC(C)=CC(C)=C1O LZCXVGMGRORTJD-UHFFFAOYSA-N 0.000 description 1
- QSEOBUMWQKSYAO-UHFFFAOYSA-N 2,4-dimethyl-6-propylphenol Chemical compound CCCC1=CC(C)=CC(C)=C1O QSEOBUMWQKSYAO-UHFFFAOYSA-N 0.000 description 1
- GKPNMUZVXNHWPX-UHFFFAOYSA-N 2,4-dipentylphenol Chemical compound CCCCCC1=CC=C(O)C(CCCCC)=C1 GKPNMUZVXNHWPX-UHFFFAOYSA-N 0.000 description 1
- BRBBSBYGIPRNSU-UHFFFAOYSA-N 2,4-dipropylphenol Chemical compound CCCC1=CC=C(O)C(CCC)=C1 BRBBSBYGIPRNSU-UHFFFAOYSA-N 0.000 description 1
- DKRVPEBSFVVKAH-UHFFFAOYSA-N 2,6-diethyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CCC1=C(O)C(CC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 DKRVPEBSFVVKAH-UHFFFAOYSA-N 0.000 description 1
- YAHDPZOCCUOXLW-UHFFFAOYSA-N 2,6-diethyl-4-hexylphenol Chemical compound CCCCCCC1=CC(CC)=C(O)C(CC)=C1 YAHDPZOCCUOXLW-UHFFFAOYSA-N 0.000 description 1
- HFVTUNMAGWBDGI-UHFFFAOYSA-N 2,6-diethyl-4-methylphenol Chemical compound CCC1=CC(C)=CC(CC)=C1O HFVTUNMAGWBDGI-UHFFFAOYSA-N 0.000 description 1
- LPVCACGMEHZJOY-UHFFFAOYSA-N 2,6-diethyl-4-pentylphenol Chemical compound CCCCCC1=CC(CC)=C(O)C(CC)=C1 LPVCACGMEHZJOY-UHFFFAOYSA-N 0.000 description 1
- FSOZDASILVCNHT-UHFFFAOYSA-N 2,6-diethyl-4-phenylphenol Chemical compound CCC1=C(O)C(CC)=CC(C=2C=CC=CC=2)=C1 FSOZDASILVCNHT-UHFFFAOYSA-N 0.000 description 1
- WZKGOTIKKZIUCO-UHFFFAOYSA-N 2,6-diethyl-4-propylphenol Chemical compound CCCC1=CC(CC)=C(O)C(CC)=C1 WZKGOTIKKZIUCO-UHFFFAOYSA-N 0.000 description 1
- METWAQRCMRWDAW-UHFFFAOYSA-N 2,6-diethylphenol Chemical compound CCC1=CC=CC(CC)=C1O METWAQRCMRWDAW-UHFFFAOYSA-N 0.000 description 1
- QHGSEUMPNSFCQX-UHFFFAOYSA-N 2,6-dimethyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CC1=C(O)C(C)=CC(C(C)(C)C=2C=CC=CC=2)=C1 QHGSEUMPNSFCQX-UHFFFAOYSA-N 0.000 description 1
- GARYJBCFXHQULL-UHFFFAOYSA-N 2,6-dimethyl-4-pentylphenol Chemical compound CCCCCC1=CC(C)=C(O)C(C)=C1 GARYJBCFXHQULL-UHFFFAOYSA-N 0.000 description 1
- SPDODUOTLRAEOR-UHFFFAOYSA-N 2,6-dimethyl-4-phenylphenol Chemical compound CC1=C(O)C(C)=CC(C=2C=CC=CC=2)=C1 SPDODUOTLRAEOR-UHFFFAOYSA-N 0.000 description 1
- HJZJMARGPNJHHG-UHFFFAOYSA-N 2,6-dimethyl-4-propylphenol Chemical compound CCCC1=CC(C)=C(O)C(C)=C1 HJZJMARGPNJHHG-UHFFFAOYSA-N 0.000 description 1
- NAILKKRDWBJCNH-UHFFFAOYSA-N 2,6-dipropylphenol Chemical compound CCCC1=CC=CC(CCC)=C1O NAILKKRDWBJCNH-UHFFFAOYSA-N 0.000 description 1
- BEHKQKDAIWPVKL-UHFFFAOYSA-N 2-(1-cyclohexyloxyethyl)phenol Chemical compound C=1C=CC=C(O)C=1C(C)OC1CCCCC1 BEHKQKDAIWPVKL-UHFFFAOYSA-N 0.000 description 1
- CSDQKOPMLYJJRK-UHFFFAOYSA-N 2-(1-cyclopentyloxyethyl)phenol Chemical compound C=1C=CC=C(O)C=1C(C)OC1CCCC1 CSDQKOPMLYJJRK-UHFFFAOYSA-N 0.000 description 1
- SHEVEUQGJRUUEX-UHFFFAOYSA-N 2-(1-phenoxyethyl)phenol Chemical compound C=1C=CC=C(O)C=1C(C)OC1=CC=CC=C1 SHEVEUQGJRUUEX-UHFFFAOYSA-N 0.000 description 1
- GIAFURWZWWWBQT-UHFFFAOYSA-N 2-(2-aminoethoxy)ethanol Chemical compound NCCOCCO GIAFURWZWWWBQT-UHFFFAOYSA-N 0.000 description 1
- CERXASTYBVJNEE-UHFFFAOYSA-N 2-(2-cyclohexyloxypropan-2-yl)phenol Chemical compound C=1C=CC=C(O)C=1C(C)(C)OC1CCCCC1 CERXASTYBVJNEE-UHFFFAOYSA-N 0.000 description 1
- PHEFPQZYYWWRDU-UHFFFAOYSA-N 2-(2-phenylethoxymethyl)phenol Chemical compound OC1=CC=CC=C1COCCC1=CC=CC=C1 PHEFPQZYYWWRDU-UHFFFAOYSA-N 0.000 description 1
- CJWNFAKWHDOUKL-UHFFFAOYSA-N 2-(2-phenylpropan-2-yl)phenol Chemical compound C=1C=CC=C(O)C=1C(C)(C)C1=CC=CC=C1 CJWNFAKWHDOUKL-UHFFFAOYSA-N 0.000 description 1
- PDKUJYBOPSDPMX-UHFFFAOYSA-N 2-(3-phenoxypentan-3-yl)phenol Chemical compound C=1C=CC=C(O)C=1C(CC)(CC)OC1=CC=CC=C1 PDKUJYBOPSDPMX-UHFFFAOYSA-N 0.000 description 1
- DRXPURBMNBRARO-UHFFFAOYSA-N 2-(3-phenylpropoxymethyl)phenol Chemical compound OC1=CC=CC=C1COCCCC1=CC=CC=C1 DRXPURBMNBRARO-UHFFFAOYSA-N 0.000 description 1
- GNNUWFUVNWRCEO-UHFFFAOYSA-N 2-(Ethoxymethyl)phenol Chemical compound CCOCC1=CC=CC=C1O GNNUWFUVNWRCEO-UHFFFAOYSA-N 0.000 description 1
- NPSWRQFCRZXYLO-UHFFFAOYSA-N 2-(cyclohexyloxymethyl)phenol Chemical compound OC1=CC=CC=C1COC1CCCCC1 NPSWRQFCRZXYLO-UHFFFAOYSA-N 0.000 description 1
- VXDHFIYVVXOUMZ-UHFFFAOYSA-N 2-(cyclopentyloxymethyl)phenol Chemical compound OC1=CC=CC=C1COC1CCCC1 VXDHFIYVVXOUMZ-UHFFFAOYSA-N 0.000 description 1
- TXHLGOIVNLSKPX-UHFFFAOYSA-N 2-(diisocyanatomethyl)pyridine Chemical compound O=C=NC(N=C=O)c1ccccn1 TXHLGOIVNLSKPX-UHFFFAOYSA-N 0.000 description 1
- SSICPQZWCDZSQA-UHFFFAOYSA-N 2-(methoxymethyl)phenol Chemical compound COCC1=CC=CC=C1O SSICPQZWCDZSQA-UHFFFAOYSA-N 0.000 description 1
- WNKFGPVMJCGWIA-UHFFFAOYSA-N 2-(phenoxymethyl)phenol Chemical compound OC1=CC=CC=C1COC1=CC=CC=C1 WNKFGPVMJCGWIA-UHFFFAOYSA-N 0.000 description 1
- KXRBJDHLRRVYAA-UHFFFAOYSA-N 2-(phenylmethoxymethyl)phenol Chemical compound OC1=CC=CC=C1COCC1=CC=CC=C1 KXRBJDHLRRVYAA-UHFFFAOYSA-N 0.000 description 1
- FEXBEKLLSUWSIM-UHFFFAOYSA-N 2-Butyl-4-methylphenol Chemical compound CCCCC1=CC(C)=CC=C1O FEXBEKLLSUWSIM-UHFFFAOYSA-N 0.000 description 1
- JTOXIUXNGPRKHI-UHFFFAOYSA-N 2-[(2,3,4-trimethylphenoxy)methyl]phenol Chemical compound CC1=C(C)C(C)=CC=C1OCC1=CC=CC=C1O JTOXIUXNGPRKHI-UHFFFAOYSA-N 0.000 description 1
- MNQJGSCHVDVZGH-UHFFFAOYSA-N 2-[phenoxy(diphenyl)methyl]phenol Chemical compound OC1=CC=CC=C1C(C=1C=CC=CC=1)(C=1C=CC=CC=1)OC1=CC=CC=C1 MNQJGSCHVDVZGH-UHFFFAOYSA-N 0.000 description 1
- WDNZXHUUNHEDJY-UHFFFAOYSA-N 2-[phenoxy(phenyl)methyl]phenol Chemical compound OC1=CC=CC=C1C(C=1C=CC=CC=1)OC1=CC=CC=C1 WDNZXHUUNHEDJY-UHFFFAOYSA-N 0.000 description 1
- JCBPETKZIGVZRE-UHFFFAOYSA-N 2-aminobutan-1-ol Chemical compound CCC(N)CO JCBPETKZIGVZRE-UHFFFAOYSA-N 0.000 description 1
- PQMCFTMVQORYJC-UHFFFAOYSA-N 2-aminocyclohexan-1-ol Chemical compound NC1CCCCC1O PQMCFTMVQORYJC-UHFFFAOYSA-N 0.000 description 1
- QLIBJPGWWSHWBF-UHFFFAOYSA-N 2-aminoethyl methacrylate Chemical compound CC(=C)C(=O)OCCN QLIBJPGWWSHWBF-UHFFFAOYSA-N 0.000 description 1
- BKMMTJMQCTUHRP-UHFFFAOYSA-N 2-aminopropan-1-ol Chemical compound CC(N)CO BKMMTJMQCTUHRP-UHFFFAOYSA-N 0.000 description 1
- VADKRMSMGWJZCF-UHFFFAOYSA-N 2-bromophenol Chemical compound OC1=CC=CC=C1Br VADKRMSMGWJZCF-UHFFFAOYSA-N 0.000 description 1
- PGGSLMBIBDGHCF-UHFFFAOYSA-N 2-butyl-4,6-diethylphenol Chemical compound CCCCC1=CC(CC)=CC(CC)=C1O PGGSLMBIBDGHCF-UHFFFAOYSA-N 0.000 description 1
- UIXRDRQSWYSVNK-UHFFFAOYSA-N 2-butyl-4,6-dimethylphenol Chemical compound CCCCC1=CC(C)=CC(C)=C1O UIXRDRQSWYSVNK-UHFFFAOYSA-N 0.000 description 1
- XETZPXGVUZURJO-UHFFFAOYSA-N 2-butyl-4,6-dipropylphenol Chemical compound CCCCC1=CC(CCC)=CC(CCC)=C1O XETZPXGVUZURJO-UHFFFAOYSA-N 0.000 description 1
- AGMVRFBVPSPDHB-UHFFFAOYSA-N 2-butyl-4-(2-phenylpropan-2-yl)-6-propylphenol Chemical compound CCCC1=C(O)C(CCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 AGMVRFBVPSPDHB-UHFFFAOYSA-N 0.000 description 1
- JBWWHHCUPVRNBH-UHFFFAOYSA-N 2-butyl-4-(2-phenylpropan-2-yl)phenol Chemical compound C1=C(O)C(CCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 JBWWHHCUPVRNBH-UHFFFAOYSA-N 0.000 description 1
- LAEAEMFNIHYYRJ-UHFFFAOYSA-N 2-butyl-4-ethyl-6-methylphenol Chemical compound CCCCC1=CC(CC)=CC(C)=C1O LAEAEMFNIHYYRJ-UHFFFAOYSA-N 0.000 description 1
- UGEUHNQEIFIZKX-UHFFFAOYSA-N 2-butyl-4-ethyl-6-propylphenol Chemical compound CCCCC1=CC(CC)=CC(CCC)=C1O UGEUHNQEIFIZKX-UHFFFAOYSA-N 0.000 description 1
- DAKZLOJXUDPQIL-UHFFFAOYSA-N 2-butyl-4-ethylphenol Chemical compound CCCCC1=CC(CC)=CC=C1O DAKZLOJXUDPQIL-UHFFFAOYSA-N 0.000 description 1
- MKFZUGOVEXSIRO-UHFFFAOYSA-N 2-butyl-4-heptyl-6-propylphenol Chemical compound CCCCCCCC1=CC(CCC)=C(O)C(CCCC)=C1 MKFZUGOVEXSIRO-UHFFFAOYSA-N 0.000 description 1
- CLZICFIXYOKGBO-UHFFFAOYSA-N 2-butyl-4-heptylphenol Chemical compound CCCCCCCC1=CC=C(O)C(CCCC)=C1 CLZICFIXYOKGBO-UHFFFAOYSA-N 0.000 description 1
- ISJQEEWPGOUIJQ-UHFFFAOYSA-N 2-butyl-4-hexyl-6-methylphenol Chemical compound CCCCCCC1=CC(C)=C(O)C(CCCC)=C1 ISJQEEWPGOUIJQ-UHFFFAOYSA-N 0.000 description 1
- ROFOSPRAPLFMME-UHFFFAOYSA-N 2-butyl-4-hexyl-6-propylphenol Chemical compound CCCCCCC1=CC(CCC)=C(O)C(CCCC)=C1 ROFOSPRAPLFMME-UHFFFAOYSA-N 0.000 description 1
- GZMOYWNMJYGXCM-UHFFFAOYSA-N 2-butyl-4-hexylphenol Chemical compound CCCCCCC1=CC=C(O)C(CCCC)=C1 GZMOYWNMJYGXCM-UHFFFAOYSA-N 0.000 description 1
- ANQBJSHUFLIDCH-UHFFFAOYSA-N 2-butyl-4-methyl-6-propylphenol Chemical compound CCCCC1=CC(C)=CC(CCC)=C1O ANQBJSHUFLIDCH-UHFFFAOYSA-N 0.000 description 1
- DQOUITOGWPGUBK-UHFFFAOYSA-N 2-butyl-4-octyl-6-propylphenol Chemical compound CCCCCCCCC1=CC(CCC)=C(O)C(CCCC)=C1 DQOUITOGWPGUBK-UHFFFAOYSA-N 0.000 description 1
- MYRPZOHNKFKKPR-UHFFFAOYSA-N 2-butyl-4-octylphenol Chemical compound CCCCCCCCC1=CC=C(O)C(CCCC)=C1 MYRPZOHNKFKKPR-UHFFFAOYSA-N 0.000 description 1
- LCWGFWANTWNHHA-UHFFFAOYSA-N 2-butyl-4-pentyl-6-propylphenol Chemical compound CCCCCC1=CC(CCC)=C(O)C(CCCC)=C1 LCWGFWANTWNHHA-UHFFFAOYSA-N 0.000 description 1
- QOUDALFLJMVOON-UHFFFAOYSA-N 2-butyl-4-pentylphenol Chemical compound CCCCCC1=CC=C(O)C(CCCC)=C1 QOUDALFLJMVOON-UHFFFAOYSA-N 0.000 description 1
- YIWYYKYOGOYTHH-UHFFFAOYSA-N 2-butyl-4-phenyl-6-propylphenol Chemical compound CCCC1=C(O)C(CCCC)=CC(C=2C=CC=CC=2)=C1 YIWYYKYOGOYTHH-UHFFFAOYSA-N 0.000 description 1
- HCZJXBXOJGEADP-UHFFFAOYSA-N 2-butyl-4-phenylphenol Chemical compound C1=C(O)C(CCCC)=CC(C=2C=CC=CC=2)=C1 HCZJXBXOJGEADP-UHFFFAOYSA-N 0.000 description 1
- DGZXXQOXJFRRDK-UHFFFAOYSA-N 2-butyl-4-propylphenol Chemical compound CCCCC1=CC(CCC)=CC=C1O DGZXXQOXJFRRDK-UHFFFAOYSA-N 0.000 description 1
- CGLFBHIJNGCEPI-UHFFFAOYSA-N 2-butyl-6-ethyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CCC1=C(O)C(CCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 CGLFBHIJNGCEPI-UHFFFAOYSA-N 0.000 description 1
- YUKAYLMZDNKXSK-UHFFFAOYSA-N 2-butyl-6-ethyl-4-heptylphenol Chemical compound CCCCCCCC1=CC(CC)=C(O)C(CCCC)=C1 YUKAYLMZDNKXSK-UHFFFAOYSA-N 0.000 description 1
- JVNPNBPLGNIKBD-UHFFFAOYSA-N 2-butyl-6-ethyl-4-hexylphenol Chemical compound CCCCCCC1=CC(CC)=C(O)C(CCCC)=C1 JVNPNBPLGNIKBD-UHFFFAOYSA-N 0.000 description 1
- UWKSIBQHTYCYAO-UHFFFAOYSA-N 2-butyl-6-ethyl-4-methylphenol Chemical compound CCCCC1=CC(C)=CC(CC)=C1O UWKSIBQHTYCYAO-UHFFFAOYSA-N 0.000 description 1
- YGFDLAXYYNSFTC-UHFFFAOYSA-N 2-butyl-6-ethyl-4-octylphenol Chemical compound CCCCCCCCC1=CC(CC)=C(O)C(CCCC)=C1 YGFDLAXYYNSFTC-UHFFFAOYSA-N 0.000 description 1
- ZEYZGXBEQKTVGJ-UHFFFAOYSA-N 2-butyl-6-ethyl-4-pentylphenol Chemical compound CCCCCC1=CC(CC)=C(O)C(CCCC)=C1 ZEYZGXBEQKTVGJ-UHFFFAOYSA-N 0.000 description 1
- WNXSKDHIBNIGKD-UHFFFAOYSA-N 2-butyl-6-ethyl-4-phenylphenol Chemical compound CCC1=C(O)C(CCCC)=CC(C=2C=CC=CC=2)=C1 WNXSKDHIBNIGKD-UHFFFAOYSA-N 0.000 description 1
- PYRWHDRDGFYEBJ-UHFFFAOYSA-N 2-butyl-6-ethyl-4-propylphenol Chemical compound CCCCC1=CC(CCC)=CC(CC)=C1O PYRWHDRDGFYEBJ-UHFFFAOYSA-N 0.000 description 1
- DKUKYQHDSXMXBF-UHFFFAOYSA-N 2-butyl-6-ethylphenol Chemical compound CCCCC1=CC=CC(CC)=C1O DKUKYQHDSXMXBF-UHFFFAOYSA-N 0.000 description 1
- ZOTYLDGDUURFLU-UHFFFAOYSA-N 2-butyl-6-methyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CC1=C(O)C(CCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 ZOTYLDGDUURFLU-UHFFFAOYSA-N 0.000 description 1
- XAKORSRYAJJDKM-UHFFFAOYSA-N 2-butyl-6-methyl-4-pentylphenol Chemical compound CCCCCC1=CC(C)=C(O)C(CCCC)=C1 XAKORSRYAJJDKM-UHFFFAOYSA-N 0.000 description 1
- VIGDBQRXWPOJBT-UHFFFAOYSA-N 2-butyl-6-methyl-4-phenylphenol Chemical compound CC1=C(O)C(CCCC)=CC(C=2C=CC=CC=2)=C1 VIGDBQRXWPOJBT-UHFFFAOYSA-N 0.000 description 1
- DSYYHZAARQXGOG-UHFFFAOYSA-N 2-butyl-6-methyl-4-propylphenol Chemical compound CCCCC1=CC(CCC)=CC(C)=C1O DSYYHZAARQXGOG-UHFFFAOYSA-N 0.000 description 1
- KUNNUNBSGQSGDY-UHFFFAOYSA-N 2-butyl-6-methylphenol Chemical compound CCCCC1=CC=CC(C)=C1O KUNNUNBSGQSGDY-UHFFFAOYSA-N 0.000 description 1
- DOISHLXLWZJLHG-UHFFFAOYSA-N 2-butyl-6-propylphenol Chemical compound CCCCC1=CC=CC(CCC)=C1O DOISHLXLWZJLHG-UHFFFAOYSA-N 0.000 description 1
- FPYUJUBAXZAQNL-UHFFFAOYSA-N 2-chlorobenzaldehyde Chemical compound ClC1=CC=CC=C1C=O FPYUJUBAXZAQNL-UHFFFAOYSA-N 0.000 description 1
- ISPYQTSUDJAMAB-UHFFFAOYSA-N 2-chlorophenol Chemical compound OC1=CC=CC=C1Cl ISPYQTSUDJAMAB-UHFFFAOYSA-N 0.000 description 1
- MXHAHSBTOVFDBK-UHFFFAOYSA-N 2-ethyl-4,6-dimethylphenol Chemical compound CCC1=CC(C)=CC(C)=C1O MXHAHSBTOVFDBK-UHFFFAOYSA-N 0.000 description 1
- ZILCDGZZSYOUMD-UHFFFAOYSA-N 2-ethyl-4-(2-phenylpropan-2-yl)-6-propylphenol Chemical compound CCC1=C(O)C(CCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 ZILCDGZZSYOUMD-UHFFFAOYSA-N 0.000 description 1
- RVKBHWQWKCPQML-UHFFFAOYSA-N 2-ethyl-4-(2-phenylpropan-2-yl)phenol Chemical compound C1=C(O)C(CC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 RVKBHWQWKCPQML-UHFFFAOYSA-N 0.000 description 1
- LHBWYCLWAUSYQB-UHFFFAOYSA-N 2-ethyl-4-heptyl-6-hexylphenol Chemical compound CCCCCCCC1=CC(CC)=C(O)C(CCCCCC)=C1 LHBWYCLWAUSYQB-UHFFFAOYSA-N 0.000 description 1
- UPFANZBLORSZTL-UHFFFAOYSA-N 2-ethyl-4-heptyl-6-pentylphenol Chemical compound CCCCCCCC1=CC(CC)=C(O)C(CCCCC)=C1 UPFANZBLORSZTL-UHFFFAOYSA-N 0.000 description 1
- CNWMXHLTDLVXOE-UHFFFAOYSA-N 2-ethyl-4-heptyl-6-propylphenol Chemical compound CCCCCCCC1=CC(CC)=C(O)C(CCC)=C1 CNWMXHLTDLVXOE-UHFFFAOYSA-N 0.000 description 1
- GNNUIASUJJVTDP-UHFFFAOYSA-N 2-ethyl-4-heptylphenol Chemical compound CCCCCCCC1=CC=C(O)C(CC)=C1 GNNUIASUJJVTDP-UHFFFAOYSA-N 0.000 description 1
- BLQMSJHBWHXGBT-UHFFFAOYSA-N 2-ethyl-4-hexyl-6-methylphenol Chemical compound CCCCCCC1=CC(C)=C(O)C(CC)=C1 BLQMSJHBWHXGBT-UHFFFAOYSA-N 0.000 description 1
- VBTUSRIKVQSEFC-UHFFFAOYSA-N 2-ethyl-4-hexyl-6-pentylphenol Chemical compound CCCCCCC1=CC(CC)=C(O)C(CCCCC)=C1 VBTUSRIKVQSEFC-UHFFFAOYSA-N 0.000 description 1
- JOCVUUIRSRPQJY-UHFFFAOYSA-N 2-ethyl-4-hexyl-6-propylphenol Chemical compound CCCCCCC1=CC(CC)=C(O)C(CCC)=C1 JOCVUUIRSRPQJY-UHFFFAOYSA-N 0.000 description 1
- LBNWCPFRHFUJNX-UHFFFAOYSA-N 2-ethyl-4-hexylphenol Chemical compound CCCCCCC1=CC=C(O)C(CC)=C1 LBNWCPFRHFUJNX-UHFFFAOYSA-N 0.000 description 1
- VWKBWEIXIRXQMO-UHFFFAOYSA-N 2-ethyl-4-methyl-6-pentylphenol Chemical compound CCCCCC1=CC(C)=CC(CC)=C1O VWKBWEIXIRXQMO-UHFFFAOYSA-N 0.000 description 1
- FHNYYGOJPPQFTP-UHFFFAOYSA-N 2-ethyl-4-methyl-6-propylphenol Chemical compound CCCC1=CC(C)=CC(CC)=C1O FHNYYGOJPPQFTP-UHFFFAOYSA-N 0.000 description 1
- WCIWNCBWTNFXON-UHFFFAOYSA-N 2-ethyl-4-octyl-6-pentylphenol Chemical compound CCCCCCCCC1=CC(CC)=C(O)C(CCCCC)=C1 WCIWNCBWTNFXON-UHFFFAOYSA-N 0.000 description 1
- XTPVPCGMGGEVMF-UHFFFAOYSA-N 2-ethyl-4-octyl-6-propylphenol Chemical compound CCCCCCCCC1=CC(CC)=C(O)C(CCC)=C1 XTPVPCGMGGEVMF-UHFFFAOYSA-N 0.000 description 1
- CABUGBFFTDFNIW-UHFFFAOYSA-N 2-ethyl-4-octylphenol Chemical compound CCCCCCCCC1=CC=C(O)C(CC)=C1 CABUGBFFTDFNIW-UHFFFAOYSA-N 0.000 description 1
- WRPFAYQYDVAVJG-UHFFFAOYSA-N 2-ethyl-4-pentyl-6-propylphenol Chemical compound CCCCCC1=CC(CC)=C(O)C(CCC)=C1 WRPFAYQYDVAVJG-UHFFFAOYSA-N 0.000 description 1
- JWZHJMPTAWZUNL-UHFFFAOYSA-N 2-ethyl-4-pentylphenol Chemical compound CCCCCC1=CC=C(O)C(CC)=C1 JWZHJMPTAWZUNL-UHFFFAOYSA-N 0.000 description 1
- POALPYSOUNDKEQ-UHFFFAOYSA-N 2-ethyl-4-phenyl-6-propylphenol Chemical compound CCC1=C(O)C(CCC)=CC(C=2C=CC=CC=2)=C1 POALPYSOUNDKEQ-UHFFFAOYSA-N 0.000 description 1
- XJYHDSXKWGRZKW-UHFFFAOYSA-N 2-ethyl-4-phenylphenol Chemical compound C1=C(O)C(CC)=CC(C=2C=CC=CC=2)=C1 XJYHDSXKWGRZKW-UHFFFAOYSA-N 0.000 description 1
- PBYQIPORGDZOGS-UHFFFAOYSA-N 2-ethyl-4-propylphenol Chemical compound CCCC1=CC=C(O)C(CC)=C1 PBYQIPORGDZOGS-UHFFFAOYSA-N 0.000 description 1
- RHPSHNZKGIFRPD-UHFFFAOYSA-N 2-ethyl-6-hexyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CCC1=C(O)C(CCCCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 RHPSHNZKGIFRPD-UHFFFAOYSA-N 0.000 description 1
- DZZXYDVGRGWNEE-UHFFFAOYSA-N 2-ethyl-6-hexyl-4-methylphenol Chemical compound CCCCCCC1=CC(C)=CC(CC)=C1O DZZXYDVGRGWNEE-UHFFFAOYSA-N 0.000 description 1
- DDTJCCBGTZTUBW-UHFFFAOYSA-N 2-ethyl-6-hexyl-4-octylphenol Chemical compound CCCCCCCCC1=CC(CC)=C(O)C(CCCCCC)=C1 DDTJCCBGTZTUBW-UHFFFAOYSA-N 0.000 description 1
- GXAADVPJLNEPIC-UHFFFAOYSA-N 2-ethyl-6-hexyl-4-pentylphenol Chemical compound CCCCCCC1=CC(CCCCC)=CC(CC)=C1O GXAADVPJLNEPIC-UHFFFAOYSA-N 0.000 description 1
- WWWUDHKAOHXYFZ-UHFFFAOYSA-N 2-ethyl-6-hexyl-4-phenylphenol Chemical compound CCC1=C(O)C(CCCCCC)=CC(C=2C=CC=CC=2)=C1 WWWUDHKAOHXYFZ-UHFFFAOYSA-N 0.000 description 1
- LVEFYOYYMIUFKV-UHFFFAOYSA-N 2-ethyl-6-hexyl-4-propylphenol Chemical compound CCCCCCC1=CC(CCC)=CC(CC)=C1O LVEFYOYYMIUFKV-UHFFFAOYSA-N 0.000 description 1
- IVRIRLUKIGLWNJ-UHFFFAOYSA-N 2-ethyl-6-methyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CC1=C(O)C(CC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 IVRIRLUKIGLWNJ-UHFFFAOYSA-N 0.000 description 1
- RYSLTGFNBYVPIO-UHFFFAOYSA-N 2-ethyl-6-methyl-4-pentylphenol Chemical compound CCCCCC1=CC(C)=C(O)C(CC)=C1 RYSLTGFNBYVPIO-UHFFFAOYSA-N 0.000 description 1
- VKYWLQLKFJGIPO-UHFFFAOYSA-N 2-ethyl-6-methyl-4-phenylphenol Chemical compound CC1=C(O)C(CC)=CC(C=2C=CC=CC=2)=C1 VKYWLQLKFJGIPO-UHFFFAOYSA-N 0.000 description 1
- VEGXDBZAPWMZIN-UHFFFAOYSA-N 2-ethyl-6-methyl-4-propylphenol Chemical compound CCCC1=CC(C)=C(O)C(CC)=C1 VEGXDBZAPWMZIN-UHFFFAOYSA-N 0.000 description 1
- YHENCQWUXKDQCM-UHFFFAOYSA-N 2-ethyl-6-pentyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CCC1=C(O)C(CCCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 YHENCQWUXKDQCM-UHFFFAOYSA-N 0.000 description 1
- CIZMZAUIJSVNPX-UHFFFAOYSA-N 2-ethyl-6-pentyl-4-phenylphenol Chemical compound CCC1=C(O)C(CCCCC)=CC(C=2C=CC=CC=2)=C1 CIZMZAUIJSVNPX-UHFFFAOYSA-N 0.000 description 1
- MPWRPYMJOGDTQL-UHFFFAOYSA-N 2-ethyl-6-pentyl-4-propylphenol Chemical compound CCCCCC1=CC(CCC)=CC(CC)=C1O MPWRPYMJOGDTQL-UHFFFAOYSA-N 0.000 description 1
- ICTYOCFDKKBVPG-UHFFFAOYSA-N 2-ethyl-6-pentylphenol Chemical compound CCCCCC1=CC=CC(CC)=C1O ICTYOCFDKKBVPG-UHFFFAOYSA-N 0.000 description 1
- AKZFZHNJLYDHKN-UHFFFAOYSA-N 2-ethyl-6-propylphenol Chemical compound CCCC1=CC=CC(CC)=C1O AKZFZHNJLYDHKN-UHFFFAOYSA-N 0.000 description 1
- ZWDVQMVZZYIAHO-UHFFFAOYSA-N 2-fluorobenzaldehyde Chemical compound FC1=CC=CC=C1C=O ZWDVQMVZZYIAHO-UHFFFAOYSA-N 0.000 description 1
- FPHHNBOGUFZGJG-UHFFFAOYSA-N 2-heptyl-4-(2-phenylpropan-2-yl)phenol Chemical compound C1=C(O)C(CCCCCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 FPHHNBOGUFZGJG-UHFFFAOYSA-N 0.000 description 1
- FZHFMOCXQPRZPI-UHFFFAOYSA-N 2-heptyl-4-hexylphenol Chemical compound CCCCCCCC1=CC(CCCCCC)=CC=C1O FZHFMOCXQPRZPI-UHFFFAOYSA-N 0.000 description 1
- OMYSNYQDIZTQJX-UHFFFAOYSA-N 2-heptyl-4-methylphenol Chemical compound CCCCCCCC1=CC(C)=CC=C1O OMYSNYQDIZTQJX-UHFFFAOYSA-N 0.000 description 1
- YSHHJJAMISGFCW-UHFFFAOYSA-N 2-heptyl-4-octylphenol Chemical compound CCCCCCCCC1=CC=C(O)C(CCCCCCC)=C1 YSHHJJAMISGFCW-UHFFFAOYSA-N 0.000 description 1
- VYTYAKKIAPXBRE-UHFFFAOYSA-N 2-heptyl-4-pentylphenol Chemical compound CCCCCCCC1=CC(CCCCC)=CC=C1O VYTYAKKIAPXBRE-UHFFFAOYSA-N 0.000 description 1
- BQFZIGDUYGUTCS-UHFFFAOYSA-N 2-heptyl-4-phenylphenol Chemical compound C1=C(O)C(CCCCCCC)=CC(C=2C=CC=CC=2)=C1 BQFZIGDUYGUTCS-UHFFFAOYSA-N 0.000 description 1
- PYFYOKIQAYUDMR-UHFFFAOYSA-N 2-heptyl-4-propylphenol Chemical compound CCCCCCCC1=CC(CCC)=CC=C1O PYFYOKIQAYUDMR-UHFFFAOYSA-N 0.000 description 1
- DWCYIKLVZZTRSZ-UHFFFAOYSA-N 2-hexyl-4,6-dimethylphenol Chemical compound CCCCCCC1=CC(C)=CC(C)=C1O DWCYIKLVZZTRSZ-UHFFFAOYSA-N 0.000 description 1
- YMKDGIODAYSRMM-UHFFFAOYSA-N 2-hexyl-4-(2-phenylpropan-2-yl)phenol Chemical compound C1=C(O)C(CCCCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 YMKDGIODAYSRMM-UHFFFAOYSA-N 0.000 description 1
- VRTAOZNHKCUURQ-UHFFFAOYSA-N 2-hexyl-4-methylphenol Chemical compound CCCCCCC1=CC(C)=CC=C1O VRTAOZNHKCUURQ-UHFFFAOYSA-N 0.000 description 1
- HCRSQBYUBIYKAJ-UHFFFAOYSA-N 2-hexyl-4-octylphenol Chemical compound CCCCCCCCC1=CC=C(O)C(CCCCCC)=C1 HCRSQBYUBIYKAJ-UHFFFAOYSA-N 0.000 description 1
- VSAITRBYBIETEP-UHFFFAOYSA-N 2-hexyl-4-pentylphenol Chemical compound CCCCCCC1=CC(CCCCC)=CC=C1O VSAITRBYBIETEP-UHFFFAOYSA-N 0.000 description 1
- OPBJSVOSZVQOOI-UHFFFAOYSA-N 2-hexyl-4-phenylphenol Chemical compound C1=C(O)C(CCCCCC)=CC(C=2C=CC=CC=2)=C1 OPBJSVOSZVQOOI-UHFFFAOYSA-N 0.000 description 1
- MJHMABCWBANWCN-UHFFFAOYSA-N 2-hexyl-4-propylphenol Chemical compound CCCCCCC1=CC(CCC)=CC=C1O MJHMABCWBANWCN-UHFFFAOYSA-N 0.000 description 1
- MCWAODAAQSDPNQ-UHFFFAOYSA-N 2-hexyl-6-methyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CC1=C(O)C(CCCCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 MCWAODAAQSDPNQ-UHFFFAOYSA-N 0.000 description 1
- RLSULJPVRFEPAH-UHFFFAOYSA-N 2-hexyl-6-methyl-4-pentylphenol Chemical compound CCCCCCC1=CC(CCCCC)=CC(C)=C1O RLSULJPVRFEPAH-UHFFFAOYSA-N 0.000 description 1
- WYMVOLIWROELQK-UHFFFAOYSA-N 2-hexyl-6-methyl-4-phenylphenol Chemical compound CC1=C(O)C(CCCCCC)=CC(C=2C=CC=CC=2)=C1 WYMVOLIWROELQK-UHFFFAOYSA-N 0.000 description 1
- JDBRODRKKNHDMJ-UHFFFAOYSA-N 2-hexyl-6-methyl-4-propylphenol Chemical compound CCCCCCC1=CC(CCC)=CC(C)=C1O JDBRODRKKNHDMJ-UHFFFAOYSA-N 0.000 description 1
- WCACBHGMPNHDPN-UHFFFAOYSA-N 2-isocyanatoethyl 2,5-diisocyanatopentanoate Chemical compound N(=C=O)CCOC(C(CCCN=C=O)N=C=O)=O WCACBHGMPNHDPN-UHFFFAOYSA-N 0.000 description 1
- GNDOBZLRZOCGAS-UHFFFAOYSA-N 2-isocyanatoethyl 2,6-diisocyanatohexanoate Chemical compound O=C=NCCCCC(N=C=O)C(=O)OCCN=C=O GNDOBZLRZOCGAS-UHFFFAOYSA-N 0.000 description 1
- DPNXHTDWGGVXID-UHFFFAOYSA-N 2-isocyanatoethyl prop-2-enoate Chemical compound C=CC(=O)OCCN=C=O DPNXHTDWGGVXID-UHFFFAOYSA-N 0.000 description 1
- HSSLFJICBPDMPH-UHFFFAOYSA-N 2-isocyanatopropyl 2-methylprop-2-enoate Chemical compound O=C=NC(C)COC(=O)C(C)=C HSSLFJICBPDMPH-UHFFFAOYSA-N 0.000 description 1
- RGDAQLOWLCIMDP-UHFFFAOYSA-N 2-isocyanatopropyl prop-2-enoate Chemical compound O=C=NC(C)COC(=O)C=C RGDAQLOWLCIMDP-UHFFFAOYSA-N 0.000 description 1
- ZTFAQVQNIDKNIQ-UHFFFAOYSA-N 2-methyl-4,6-dipentylphenol Chemical compound CCCCCC1=CC(C)=C(O)C(CCCCC)=C1 ZTFAQVQNIDKNIQ-UHFFFAOYSA-N 0.000 description 1
- KPBDGTGMUIZQAW-UHFFFAOYSA-N 2-methyl-4,6-dipropylphenol Chemical compound CCCC1=CC(C)=C(O)C(CCC)=C1 KPBDGTGMUIZQAW-UHFFFAOYSA-N 0.000 description 1
- QBYOXEQNZZLFLP-UHFFFAOYSA-N 2-methyl-4-(2-phenylpropan-2-yl)-6-propylphenol Chemical compound CC1=C(O)C(CCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 QBYOXEQNZZLFLP-UHFFFAOYSA-N 0.000 description 1
- SIQFMJHVPDNION-UHFFFAOYSA-N 2-methyl-4-pentyl-6-propylphenol Chemical compound CCCCCC1=CC(C)=C(O)C(CCC)=C1 SIQFMJHVPDNION-UHFFFAOYSA-N 0.000 description 1
- MUFIAECBWIRTHK-UHFFFAOYSA-N 2-methyl-4-phenyl-6-propylphenol Chemical compound CC1=C(O)C(CCC)=CC(C=2C=CC=CC=2)=C1 MUFIAECBWIRTHK-UHFFFAOYSA-N 0.000 description 1
- OZOJGNDZRKPTTK-UHFFFAOYSA-N 2-methyl-6-pentyl-4-(2-phenylpropan-2-yl)phenol Chemical compound CC1=C(O)C(CCCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 OZOJGNDZRKPTTK-UHFFFAOYSA-N 0.000 description 1
- SGDBHMWNLODQIL-UHFFFAOYSA-N 2-methyl-6-pentyl-4-phenylphenol Chemical compound CC1=C(O)C(CCCCC)=CC(C=2C=CC=CC=2)=C1 SGDBHMWNLODQIL-UHFFFAOYSA-N 0.000 description 1
- HEDPVRKSORXGRS-UHFFFAOYSA-N 2-methyl-6-pentyl-4-propylphenol Chemical compound CCCCCC1=CC(CCC)=CC(C)=C1O HEDPVRKSORXGRS-UHFFFAOYSA-N 0.000 description 1
- UZDBICBKUHGBSU-UHFFFAOYSA-N 2-methyl-6-pentylphenol Chemical compound CCCCCC1=CC=CC(C)=C1O UZDBICBKUHGBSU-UHFFFAOYSA-N 0.000 description 1
- NXSQQKKFGJHACS-UHFFFAOYSA-N 2-methyl-6-propylphenol Chemical compound CCCC1=CC=CC(C)=C1O NXSQQKKFGJHACS-UHFFFAOYSA-N 0.000 description 1
- CXCMHIHWZCWXQU-UHFFFAOYSA-N 2-methylpyridine-3,4-diamine Chemical compound CC1=NC=CC(N)=C1N CXCMHIHWZCWXQU-UHFFFAOYSA-N 0.000 description 1
- ZVKRNMZUPKAEDJ-UHFFFAOYSA-N 2-pentyl-4-(2-phenylpropan-2-yl)phenol Chemical compound C1=C(O)C(CCCCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 ZVKRNMZUPKAEDJ-UHFFFAOYSA-N 0.000 description 1
- MJRIGYZMXNOATK-UHFFFAOYSA-N 2-pentyl-4-phenylphenol Chemical compound C1=C(O)C(CCCCC)=CC(C=2C=CC=CC=2)=C1 MJRIGYZMXNOATK-UHFFFAOYSA-N 0.000 description 1
- DLZPATGLJANHOQ-UHFFFAOYSA-N 2-pentyl-4-propylphenol Chemical compound CCCCCC1=CC(CCC)=CC=C1O DLZPATGLJANHOQ-UHFFFAOYSA-N 0.000 description 1
- 229940061334 2-phenylphenol Drugs 0.000 description 1
- VYXRCBSLMKPUBV-UHFFFAOYSA-N 3-[2,3-bis(3-aminopropyl)-4-methylphenyl]propan-1-amine Chemical compound NCCCC1=C(C(=C(C=C1)C)CCCN)CCCN VYXRCBSLMKPUBV-UHFFFAOYSA-N 0.000 description 1
- HVQPNKXSWMVRDZ-UHFFFAOYSA-N 3-isocyanatopropyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCN=C=O HVQPNKXSWMVRDZ-UHFFFAOYSA-N 0.000 description 1
- ORTCGSWQDZPULK-UHFFFAOYSA-N 3-isocyanatopropyl prop-2-enoate Chemical compound C=CC(=O)OCCCN=C=O ORTCGSWQDZPULK-UHFFFAOYSA-N 0.000 description 1
- OXYRENDGHPGWKV-UHFFFAOYSA-N 3-methyl-5-phenylpentan-1-ol Chemical compound OCCC(C)CCC1=CC=CC=C1 OXYRENDGHPGWKV-UHFFFAOYSA-N 0.000 description 1
- AXNUJYHFQHQZBE-UHFFFAOYSA-N 3-methylbenzene-1,2-diamine Chemical compound CC1=CC=CC(N)=C1N AXNUJYHFQHQZBE-UHFFFAOYSA-N 0.000 description 1
- MWHLORMGUXPWLW-UHFFFAOYSA-N 4,4-diethylcyclohexan-1-ol Chemical compound CCC1(CC)CCC(O)CC1 MWHLORMGUXPWLW-UHFFFAOYSA-N 0.000 description 1
- QHQIFBYQGBWWKP-UHFFFAOYSA-N 4-(2-phenylpropan-2-yl)-2,6-dipropylphenol Chemical compound CCCC1=C(O)C(CCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 QHQIFBYQGBWWKP-UHFFFAOYSA-N 0.000 description 1
- GGGNEDHTYNAPLK-UHFFFAOYSA-N 4-(2-phenylpropan-2-yl)-2-propylphenol Chemical compound C1=C(O)C(CCC)=CC(C(C)(C)C=2C=CC=CC=2)=C1 GGGNEDHTYNAPLK-UHFFFAOYSA-N 0.000 description 1
- IJALWSVNUBBQRA-UHFFFAOYSA-N 4-Isopropyl-3-methylphenol Chemical compound CC(C)C1=CC=C(O)C=C1C IJALWSVNUBBQRA-UHFFFAOYSA-N 0.000 description 1
- ALYNCZNDIQEVRV-UHFFFAOYSA-N 4-aminobenzoic acid Chemical compound NC1=CC=C(C(O)=O)C=C1 ALYNCZNDIQEVRV-UHFFFAOYSA-N 0.000 description 1
- DPURNAQARVHQHE-UHFFFAOYSA-N 4-aminobutane-1,2,3-tricarboxylic acid Chemical compound NCC(C(O)=O)C(C(O)=O)CC(O)=O DPURNAQARVHQHE-UHFFFAOYSA-N 0.000 description 1
- DRNGLYHKYPNTEA-UHFFFAOYSA-N 4-azaniumylcyclohexane-1-carboxylate Chemical compound NC1CCC(C(O)=O)CC1 DRNGLYHKYPNTEA-UHFFFAOYSA-N 0.000 description 1
- VTVNEYMEIZYSKO-UHFFFAOYSA-N 4-butyl-2,6-diethylphenol Chemical compound CCCCC1=CC(CC)=C(O)C(CC)=C1 VTVNEYMEIZYSKO-UHFFFAOYSA-N 0.000 description 1
- RQQYPGZACLPRDX-UHFFFAOYSA-N 4-butyl-2,6-dimethylphenol Chemical compound CCCCC1=CC(C)=C(O)C(C)=C1 RQQYPGZACLPRDX-UHFFFAOYSA-N 0.000 description 1
- YKCDTYZKCQUPLB-UHFFFAOYSA-N 4-butyl-2,6-dipropylphenol Chemical compound CCCCC1=CC(CCC)=C(O)C(CCC)=C1 YKCDTYZKCQUPLB-UHFFFAOYSA-N 0.000 description 1
- JGPLWHNBQXFBQD-UHFFFAOYSA-N 4-butyl-2-ethyl-6-hexylphenol Chemical compound CCCCCCC1=CC(CCCC)=CC(CC)=C1O JGPLWHNBQXFBQD-UHFFFAOYSA-N 0.000 description 1
- RRGSURQFDSOLHZ-UHFFFAOYSA-N 4-butyl-2-ethyl-6-methylphenol Chemical compound CCCCC1=CC(C)=C(O)C(CC)=C1 RRGSURQFDSOLHZ-UHFFFAOYSA-N 0.000 description 1
- MYGQLGVSNBVJOM-UHFFFAOYSA-N 4-butyl-2-ethyl-6-pentylphenol Chemical compound CCCCCC1=CC(CCCC)=CC(CC)=C1O MYGQLGVSNBVJOM-UHFFFAOYSA-N 0.000 description 1
- DSPGRAACHYHUHA-UHFFFAOYSA-N 4-butyl-2-ethyl-6-propylphenol Chemical compound CCCCC1=CC(CC)=C(O)C(CCC)=C1 DSPGRAACHYHUHA-UHFFFAOYSA-N 0.000 description 1
- PJNOBPONKXGBDD-UHFFFAOYSA-N 4-butyl-2-ethylphenol Chemical compound CCCCC1=CC=C(O)C(CC)=C1 PJNOBPONKXGBDD-UHFFFAOYSA-N 0.000 description 1
- RGTRNGCUFHTWGG-UHFFFAOYSA-N 4-butyl-2-heptylphenol Chemical compound CCCCCCCC1=CC(CCCC)=CC=C1O RGTRNGCUFHTWGG-UHFFFAOYSA-N 0.000 description 1
- KQBZTJBQTRSWFZ-UHFFFAOYSA-N 4-butyl-2-hexyl-6-methylphenol Chemical compound CCCCCCC1=CC(CCCC)=CC(C)=C1O KQBZTJBQTRSWFZ-UHFFFAOYSA-N 0.000 description 1
- SHDCGBKRKJFGLB-UHFFFAOYSA-N 4-butyl-2-hexylphenol Chemical compound CCCCCCC1=CC(CCCC)=CC=C1O SHDCGBKRKJFGLB-UHFFFAOYSA-N 0.000 description 1
- VMOADSRAMIWOKE-UHFFFAOYSA-N 4-butyl-2-methyl-6-pentylphenol Chemical compound CCCCCC1=CC(CCCC)=CC(C)=C1O VMOADSRAMIWOKE-UHFFFAOYSA-N 0.000 description 1
- UIYXIVUJTWJUCD-UHFFFAOYSA-N 4-butyl-2-methyl-6-propylphenol Chemical compound CCCCC1=CC(C)=C(O)C(CCC)=C1 UIYXIVUJTWJUCD-UHFFFAOYSA-N 0.000 description 1
- IHUVDMKLWOUFLD-UHFFFAOYSA-N 4-butyl-2-pentylphenol Chemical compound CCCCCC1=CC(CCCC)=CC=C1O IHUVDMKLWOUFLD-UHFFFAOYSA-N 0.000 description 1
- FEYARRBYFHBZAP-UHFFFAOYSA-N 4-butyl-2-propylphenol Chemical compound CCCCC1=CC=C(O)C(CCC)=C1 FEYARRBYFHBZAP-UHFFFAOYSA-N 0.000 description 1
- PRRINTZNQPGZHB-UHFFFAOYSA-N 4-ethyl-2,6-dimethylphenol Chemical compound CCC1=CC(C)=C(O)C(C)=C1 PRRINTZNQPGZHB-UHFFFAOYSA-N 0.000 description 1
- ZCPIQEPQEVUZPZ-UHFFFAOYSA-N 4-ethyl-2,6-dipropylphenol Chemical compound CCCC1=CC(CC)=CC(CCC)=C1O ZCPIQEPQEVUZPZ-UHFFFAOYSA-N 0.000 description 1
- SWQNPONMAHJPPQ-UHFFFAOYSA-N 4-ethyl-2-heptylphenol Chemical compound CCCCCCCC1=CC(CC)=CC=C1O SWQNPONMAHJPPQ-UHFFFAOYSA-N 0.000 description 1
- FLUUKWIDBQGOQW-UHFFFAOYSA-N 4-ethyl-2-hexyl-6-methylphenol Chemical compound CCCCCCC1=CC(CC)=CC(C)=C1O FLUUKWIDBQGOQW-UHFFFAOYSA-N 0.000 description 1
- IAXARCCFCKCDNR-UHFFFAOYSA-N 4-ethyl-2-hexylphenol Chemical compound CCCCCCC1=CC(CC)=CC=C1O IAXARCCFCKCDNR-UHFFFAOYSA-N 0.000 description 1
- PTSPDOXBQOSHME-UHFFFAOYSA-N 4-ethyl-2-methyl-6-pentylphenol Chemical compound CCCCCC1=CC(CC)=CC(C)=C1O PTSPDOXBQOSHME-UHFFFAOYSA-N 0.000 description 1
- DFUXUUGXOMYKBN-UHFFFAOYSA-N 4-ethyl-2-methyl-6-propylphenol Chemical compound CCCC1=CC(CC)=CC(C)=C1O DFUXUUGXOMYKBN-UHFFFAOYSA-N 0.000 description 1
- ZBBCONZKZHEBPF-UHFFFAOYSA-N 4-ethyl-2-pentylphenol Chemical compound CCCCCC1=CC(CC)=CC=C1O ZBBCONZKZHEBPF-UHFFFAOYSA-N 0.000 description 1
- DTRXPNPTVRYGOE-UHFFFAOYSA-N 4-ethyl-2-propylphenol Chemical compound CCCC1=CC(CC)=CC=C1O DTRXPNPTVRYGOE-UHFFFAOYSA-N 0.000 description 1
- HVCNXQOWACZAFN-UHFFFAOYSA-N 4-ethylmorpholine Chemical compound CCN1CCOCC1 HVCNXQOWACZAFN-UHFFFAOYSA-N 0.000 description 1
- ZHTBTTWESPCXSC-UHFFFAOYSA-N 4-heptyl-2-hexylphenol Chemical compound CCCCCCCC1=CC=C(O)C(CCCCCC)=C1 ZHTBTTWESPCXSC-UHFFFAOYSA-N 0.000 description 1
- WGVSJLYSFDYQGL-UHFFFAOYSA-N 4-heptyl-2-pentylphenol Chemical compound CCCCCCCC1=CC=C(O)C(CCCCC)=C1 WGVSJLYSFDYQGL-UHFFFAOYSA-N 0.000 description 1
- GFDRZFNFQFSKNU-UHFFFAOYSA-N 4-heptyl-2-propylphenol Chemical compound CCCCCCCC1=CC=C(O)C(CCC)=C1 GFDRZFNFQFSKNU-UHFFFAOYSA-N 0.000 description 1
- SZQCPWQPLTWRHZ-UHFFFAOYSA-N 4-hexyl-2,6-dimethylphenol Chemical compound CCCCCCC1=CC(C)=C(O)C(C)=C1 SZQCPWQPLTWRHZ-UHFFFAOYSA-N 0.000 description 1
- RGZPSTNWQPYQSC-UHFFFAOYSA-N 4-hexyl-2,6-dipropylphenol Chemical compound CCCCCCC1=CC(CCC)=C(O)C(CCC)=C1 RGZPSTNWQPYQSC-UHFFFAOYSA-N 0.000 description 1
- FGIUXKCBUMGBDY-UHFFFAOYSA-N 4-hexyl-2-methyl-6-pentylphenol Chemical compound CCCCCCC1=CC(C)=C(O)C(CCCCC)=C1 FGIUXKCBUMGBDY-UHFFFAOYSA-N 0.000 description 1
- GQGAXBYQSCRANZ-UHFFFAOYSA-N 4-hexyl-2-methyl-6-propylphenol Chemical compound CCCCCCC1=CC(C)=C(O)C(CCC)=C1 GQGAXBYQSCRANZ-UHFFFAOYSA-N 0.000 description 1
- HMLDSEVOLDJUDC-UHFFFAOYSA-N 4-hexyl-2-pentylphenol Chemical compound CCCCCCC1=CC=C(O)C(CCCCC)=C1 HMLDSEVOLDJUDC-UHFFFAOYSA-N 0.000 description 1
- DTSRTHPBXBZMMP-UHFFFAOYSA-N 4-hexyl-2-propylphenol Chemical compound CCCCCCC1=CC=C(O)C(CCC)=C1 DTSRTHPBXBZMMP-UHFFFAOYSA-N 0.000 description 1
- FPMYOMFDJKFHBV-UHFFFAOYSA-N 4-isocyanatobutyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCN=C=O FPMYOMFDJKFHBV-UHFFFAOYSA-N 0.000 description 1
- OQEAEWQOPZQPSS-UHFFFAOYSA-N 4-isocyanatobutyl prop-2-enoate Chemical compound C=CC(=O)OCCCCN=C=O OQEAEWQOPZQPSS-UHFFFAOYSA-N 0.000 description 1
- ORGIXTZQPMWKGZ-UHFFFAOYSA-N 4-methyl-2,6-dipropylphenol Chemical compound CCCC1=CC(C)=CC(CCC)=C1O ORGIXTZQPMWKGZ-UHFFFAOYSA-N 0.000 description 1
- HOWYOMNTSWEROD-UHFFFAOYSA-N 4-methyl-2-pentylphenol Chemical compound CCCCCC1=CC(C)=CC=C1O HOWYOMNTSWEROD-UHFFFAOYSA-N 0.000 description 1
- NNPMYBBCIDUAJD-UHFFFAOYSA-N 4-methyl-2-propylphenol Chemical compound CCCC1=CC(C)=CC=C1O NNPMYBBCIDUAJD-UHFFFAOYSA-N 0.000 description 1
- MQWCXKGKQLNYQG-UHFFFAOYSA-N 4-methylcyclohexan-1-ol Chemical compound CC1CCC(O)CC1 MQWCXKGKQLNYQG-UHFFFAOYSA-N 0.000 description 1
- JKEMMMJZVBIURV-UHFFFAOYSA-N 4-octyl-2-pentylphenol Chemical compound CCCCCCCCC1=CC=C(O)C(CCCCC)=C1 JKEMMMJZVBIURV-UHFFFAOYSA-N 0.000 description 1
- PRIGLXJTGQLCMQ-UHFFFAOYSA-N 4-octyl-2-propylphenol Chemical compound CCCCCCCCC1=CC=C(O)C(CCC)=C1 PRIGLXJTGQLCMQ-UHFFFAOYSA-N 0.000 description 1
- BYGCHDJESMWCKK-UHFFFAOYSA-N 4-pentyl-2,6-dipropylphenol Chemical compound CCCCCC1=CC(CCC)=C(O)C(CCC)=C1 BYGCHDJESMWCKK-UHFFFAOYSA-N 0.000 description 1
- QPWWOWRMSUABAI-UHFFFAOYSA-N 4-pentyl-2-propylphenol Chemical compound CCCCCC1=CC=C(O)C(CCC)=C1 QPWWOWRMSUABAI-UHFFFAOYSA-N 0.000 description 1
- KHJZYKRUOFKZRV-UHFFFAOYSA-N 4-phenyl-2,6-dipropylphenol Chemical compound CCCC1=C(O)C(CCC)=CC(C=2C=CC=CC=2)=C1 KHJZYKRUOFKZRV-UHFFFAOYSA-N 0.000 description 1
- UIFYLOYBNHJKDV-UHFFFAOYSA-N 4-phenyl-2-propylphenol Chemical compound C1=C(O)C(CCC)=CC(C=2C=CC=CC=2)=C1 UIFYLOYBNHJKDV-UHFFFAOYSA-N 0.000 description 1
- DPZMVZIQRMVBBW-UHFFFAOYSA-N 5-Phenyl-1-pentanol Chemical compound OCCCCCC1=CC=CC=C1 DPZMVZIQRMVBBW-UHFFFAOYSA-N 0.000 description 1
- VNLFMDYLWALULR-UHFFFAOYSA-N 5-isocyanatopentyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCN=C=O VNLFMDYLWALULR-UHFFFAOYSA-N 0.000 description 1
- NXGVRYZWBLEASN-UHFFFAOYSA-N 5-isocyanatopentyl prop-2-enoate Chemical compound C=CC(=O)OCCCCCN=C=O NXGVRYZWBLEASN-UHFFFAOYSA-N 0.000 description 1
- SLXKOJJOQWFEFD-UHFFFAOYSA-N 6-aminohexanoic acid Chemical compound NCCCCCC(O)=O SLXKOJJOQWFEFD-UHFFFAOYSA-N 0.000 description 1
- ISDYQNBADWDQAB-UHFFFAOYSA-N 6-isocyanatohexyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCN=C=O ISDYQNBADWDQAB-UHFFFAOYSA-N 0.000 description 1
- XJRMEBKVZKZQGM-UHFFFAOYSA-N 6-isocyanatohexyl prop-2-enoate Chemical compound C=CC(=O)OCCCCCCN=C=O XJRMEBKVZKZQGM-UHFFFAOYSA-N 0.000 description 1
- ICIZZFOJFCBJLO-UHFFFAOYSA-N 8-isocyanatooctyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCN=C=O ICIZZFOJFCBJLO-UHFFFAOYSA-N 0.000 description 1
- KECRGISUZXMERG-UHFFFAOYSA-N 8-isocyanatooctyl prop-2-enoate Chemical compound C=CC(=O)OCCCCCCCCN=C=O KECRGISUZXMERG-UHFFFAOYSA-N 0.000 description 1
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 1
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 1
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- DLXDBOZVVLOXPE-UHFFFAOYSA-N C=CC(=O)OCCCCCCCCCCCN=C=O Chemical compound C=CC(=O)OCCCCCCCCCCCN=C=O DLXDBOZVVLOXPE-UHFFFAOYSA-N 0.000 description 1
- YSFJXSMRBVJVHO-UHFFFAOYSA-N CC1=CC=C(CN)C(CN)=C1CN Chemical compound CC1=CC=C(CN)C(CN)=C1CN YSFJXSMRBVJVHO-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical group [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 229910021589 Copper(I) bromide Inorganic materials 0.000 description 1
- 229910021591 Copper(I) chloride Inorganic materials 0.000 description 1
- 229910021595 Copper(I) iodide Inorganic materials 0.000 description 1
- 229910021590 Copper(II) bromide Inorganic materials 0.000 description 1
- 229910021592 Copper(II) chloride Inorganic materials 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- MQIUGAXCHLFZKX-UHFFFAOYSA-N Di-n-octyl phthalate Natural products CCCCCCCCOC(=O)C1=CC=CC=C1C(=O)OCCCCCCCC MQIUGAXCHLFZKX-UHFFFAOYSA-N 0.000 description 1
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 1
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 1
- JJHHIJFTHRNPIK-UHFFFAOYSA-N Diphenyl sulfoxide Chemical compound C=1C=CC=CC=1S(=O)C1=CC=CC=C1 JJHHIJFTHRNPIK-UHFFFAOYSA-N 0.000 description 1
- RYECOJGRJDOGPP-UHFFFAOYSA-N Ethylurea Chemical compound CCNC(N)=O RYECOJGRJDOGPP-UHFFFAOYSA-N 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- OWIKHYCFFJSOEH-UHFFFAOYSA-N Isocyanic acid Chemical compound N=C=O OWIKHYCFFJSOEH-UHFFFAOYSA-N 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- DWPCPZJAHOETAG-IMJSIDKUSA-N L-lanthionine Chemical compound OC(=O)[C@@H](N)CSC[C@H](N)C(O)=O DWPCPZJAHOETAG-IMJSIDKUSA-N 0.000 description 1
- JHWNWJKBPDFINM-UHFFFAOYSA-N Laurolactam Chemical compound O=C1CCCCCCCCCCCN1 JHWNWJKBPDFINM-UHFFFAOYSA-N 0.000 description 1
- NIPNSKYNPDTRPC-UHFFFAOYSA-N N-[2-oxo-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 NIPNSKYNPDTRPC-UHFFFAOYSA-N 0.000 description 1
- CHJJGSNFBQVOTG-UHFFFAOYSA-N N-methyl-guanidine Natural products CNC(N)=N CHJJGSNFBQVOTG-UHFFFAOYSA-N 0.000 description 1
- XGEGHDBEHXKFPX-UHFFFAOYSA-N N-methylthiourea Natural products CNC(N)=O XGEGHDBEHXKFPX-UHFFFAOYSA-N 0.000 description 1
- WFAYHLRNODZZHW-UHFFFAOYSA-N NCCCC=1C(=C(C=CC=1)CCCN)CCCN Chemical compound NCCCC=1C(=C(C=CC=1)CCCN)CCCN WFAYHLRNODZZHW-UHFFFAOYSA-N 0.000 description 1
- 238000004497 NIR spectroscopy Methods 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- IGFHQQFPSIBGKE-UHFFFAOYSA-N Nonylphenol Natural products CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- DYUQAZSOFZSPHD-UHFFFAOYSA-N Phenylpropanol Chemical compound CCC(O)C1=CC=CC=C1 DYUQAZSOFZSPHD-UHFFFAOYSA-N 0.000 description 1
- 229920002396 Polyurea Polymers 0.000 description 1
- 238000001069 Raman spectroscopy Methods 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- 238000007059 Strecker synthesis reaction Methods 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 1
- GKXVJHDEWHKBFH-UHFFFAOYSA-N [2-(aminomethyl)phenyl]methanamine Chemical compound NCC1=CC=CC=C1CN GKXVJHDEWHKBFH-UHFFFAOYSA-N 0.000 description 1
- JAWMENYCRQKKJY-UHFFFAOYSA-N [3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-ylmethyl)-1-oxa-2,8-diazaspiro[4.5]dec-2-en-8-yl]-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]methanone Chemical compound N1N=NC=2CN(CCC=21)CC1=NOC2(C1)CCN(CC2)C(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F JAWMENYCRQKKJY-UHFFFAOYSA-N 0.000 description 1
- KXBFLNPZHXDQLV-UHFFFAOYSA-N [cyclohexyl(diisocyanato)methyl]cyclohexane Chemical compound C1CCCCC1C(N=C=O)(N=C=O)C1CCCCC1 KXBFLNPZHXDQLV-UHFFFAOYSA-N 0.000 description 1
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 description 1
- LNWBFIVSTXCJJG-UHFFFAOYSA-N [diisocyanato(phenyl)methyl]benzene Chemical compound C=1C=CC=CC=1C(N=C=O)(N=C=O)C1=CC=CC=C1 LNWBFIVSTXCJJG-UHFFFAOYSA-N 0.000 description 1
- IKHGUXGNUITLKF-XPULMUKRSA-N acetaldehyde Chemical compound [14CH]([14CH3])=O IKHGUXGNUITLKF-XPULMUKRSA-N 0.000 description 1
- 125000003668 acetyloxy group Chemical group [H]C([H])([H])C(=O)O[*] 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 150000005215 alkyl ethers Chemical class 0.000 description 1
- FTWHFXMUJQRNBK-UHFFFAOYSA-N alpha-Methylen-gamma-aminobuttersaeure Natural products NCCC(=C)C(O)=O FTWHFXMUJQRNBK-UHFFFAOYSA-N 0.000 description 1
- 150000001371 alpha-amino acids Chemical class 0.000 description 1
- 235000008206 alpha-amino acids Nutrition 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical group [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 150000001409 amidines Chemical class 0.000 description 1
- 229960004050 aminobenzoic acid Drugs 0.000 description 1
- CBTVGIZVANVGBH-UHFFFAOYSA-N aminomethyl propanol Chemical compound CC(C)(N)CO CBTVGIZVANVGBH-UHFFFAOYSA-N 0.000 description 1
- XLJMAIOERFSOGZ-UHFFFAOYSA-N anhydrous cyanic acid Natural products OC#N XLJMAIOERFSOGZ-UHFFFAOYSA-N 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 1
- 150000008378 aryl ethers Chemical class 0.000 description 1
- 238000010923 batch production Methods 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960004217 benzyl alcohol Drugs 0.000 description 1
- WGQKYBSKWIADBV-UHFFFAOYSA-N benzylamine Chemical compound NCC1=CC=CC=C1 WGQKYBSKWIADBV-UHFFFAOYSA-N 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- LTAICIIOZBYBTN-UHFFFAOYSA-N bis(2-ethoxyphenyl) carbonate Chemical compound CCOC1=CC=CC=C1OC(=O)OC1=CC=CC=C1OCC LTAICIIOZBYBTN-UHFFFAOYSA-N 0.000 description 1
- BJQHLKABXJIVAM-UHFFFAOYSA-N bis(2-ethylhexyl) phthalate Chemical compound CCCCC(CC)COC(=O)C1=CC=CC=C1C(=O)OCC(CC)CCCC BJQHLKABXJIVAM-UHFFFAOYSA-N 0.000 description 1
- XXNJYJLOAWOENE-UHFFFAOYSA-N bis(2-isocyanatoethyl) 2-isocyanatobutanedioate Chemical compound N(=C=O)CCOC(C(CC(=O)OCCN=C=O)N=C=O)=O XXNJYJLOAWOENE-UHFFFAOYSA-N 0.000 description 1
- ILQQWSOGZZJPDL-UHFFFAOYSA-N bis(2-isocyanatoethyl) 2-isocyanatopentanedioate Chemical compound O=C=NCCOC(=O)CCC(N=C=O)C(=O)OCCN=C=O ILQQWSOGZZJPDL-UHFFFAOYSA-N 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- 239000002981 blocking agent Substances 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- 150000001639 boron compounds Chemical class 0.000 description 1
- GHWVXCQZPNWFRO-UHFFFAOYSA-N butane-2,3-diamine Chemical compound CC(N)C(C)N GHWVXCQZPNWFRO-UHFFFAOYSA-N 0.000 description 1
- SKKTUOZKZKCGTB-UHFFFAOYSA-N butyl carbamate Chemical compound CCCCOC(N)=O SKKTUOZKZKCGTB-UHFFFAOYSA-N 0.000 description 1
- ZTQSAGDEMFDKMZ-UHFFFAOYSA-N butyric aldehyde Natural products CCCC=O ZTQSAGDEMFDKMZ-UHFFFAOYSA-N 0.000 description 1
- BRPQOXSCLDDYGP-UHFFFAOYSA-N calcium oxide Chemical compound [O-2].[Ca+2] BRPQOXSCLDDYGP-UHFFFAOYSA-N 0.000 description 1
- 239000000292 calcium oxide Substances 0.000 description 1
- ODINCKMPIJJUCX-UHFFFAOYSA-N calcium oxide Inorganic materials [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 1
- 150000001718 carbodiimides Chemical class 0.000 description 1
- 150000001722 carbon compounds Chemical class 0.000 description 1
- 125000005587 carbonate group Chemical group 0.000 description 1
- HHTWOMMSBMNRKP-UHFFFAOYSA-N carvacrol Natural products CC(=C)C1=CC=C(C)C(O)=C1 HHTWOMMSBMNRKP-UHFFFAOYSA-N 0.000 description 1
- RECUKUPTGUEGMW-UHFFFAOYSA-N carvacrol Chemical compound CC(C)C1=CC=C(C)C(O)=C1 RECUKUPTGUEGMW-UHFFFAOYSA-N 0.000 description 1
- 235000007746 carvacrol Nutrition 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 238000006482 condensation reaction Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- OXBLHERUFWYNTN-UHFFFAOYSA-M copper(I) chloride Chemical compound [Cu]Cl OXBLHERUFWYNTN-UHFFFAOYSA-M 0.000 description 1
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 1
- 229910052593 corundum Inorganic materials 0.000 description 1
- 229930003836 cresol Natural products 0.000 description 1
- 229910052906 cristobalite Inorganic materials 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 125000000000 cycloalkoxy group Chemical group 0.000 description 1
- 125000004976 cyclobutylene group Chemical group 0.000 description 1
- QCRFMSUKWRQZEM-UHFFFAOYSA-N cycloheptanol Chemical compound OC1CCCCCC1 QCRFMSUKWRQZEM-UHFFFAOYSA-N 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- KQWGXHWJMSMDJJ-UHFFFAOYSA-N cyclohexyl isocyanate Chemical compound O=C=NC1CCCCC1 KQWGXHWJMSMDJJ-UHFFFAOYSA-N 0.000 description 1
- 125000004956 cyclohexylene group Chemical group 0.000 description 1
- 125000002933 cyclohexyloxy group Chemical group C1(CCCCC1)O* 0.000 description 1
- 125000006547 cyclononyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- WJTCGQSWYFHTAC-UHFFFAOYSA-N cyclooctane Chemical compound C1CCCCCCC1 WJTCGQSWYFHTAC-UHFFFAOYSA-N 0.000 description 1
- 239000004914 cyclooctane Substances 0.000 description 1
- FHADSMKORVFYOS-UHFFFAOYSA-N cyclooctanol Chemical compound OC1CCCCCCC1 FHADSMKORVFYOS-UHFFFAOYSA-N 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001887 cyclopentyloxy group Chemical group C1(CCCC1)O* 0.000 description 1
- 125000006612 decyloxy group Chemical group 0.000 description 1
- 238000007872 degassing Methods 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 229960002380 dibutyl phthalate Drugs 0.000 description 1
- RJGHQTVXGKYATR-UHFFFAOYSA-L dibutyl(dichloro)stannane Chemical compound CCCC[Sn](Cl)(Cl)CCCC RJGHQTVXGKYATR-UHFFFAOYSA-L 0.000 description 1
- 239000012975 dibutyltin dilaurate Substances 0.000 description 1
- 229940117389 dichlorobenzene Drugs 0.000 description 1
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 1
- SWSQBOPZIKWTGO-UHFFFAOYSA-N dimethylaminoamidine Natural products CN(C)C(N)=N SWSQBOPZIKWTGO-UHFFFAOYSA-N 0.000 description 1
- LTYMSROWYAPPGB-UHFFFAOYSA-N diphenyl sulfide Chemical compound C=1C=CC=CC=1SC1=CC=CC=C1 LTYMSROWYAPPGB-UHFFFAOYSA-N 0.000 description 1
- CZZYITDELCSZES-UHFFFAOYSA-N diphenylmethane Chemical compound C=1C=CC=CC=1CC1=CC=CC=C1 CZZYITDELCSZES-UHFFFAOYSA-N 0.000 description 1
- ZZTCPWRAHWXWCH-UHFFFAOYSA-N diphenylmethanediamine Chemical compound C=1C=CC=CC=1C(N)(N)C1=CC=CC=C1 ZZTCPWRAHWXWCH-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- CZEPJJXZASVXQF-ZETCQYMHSA-N ethyl (2s)-2,6-diaminohexanoate Chemical compound CCOC(=O)[C@@H](N)CCCCN CZEPJJXZASVXQF-ZETCQYMHSA-N 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 238000000855 fermentation Methods 0.000 description 1
- 230000004151 fermentation Effects 0.000 description 1
- KTWOOEGAPBSYNW-UHFFFAOYSA-N ferrocene Chemical compound [Fe+2].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 KTWOOEGAPBSYNW-UHFFFAOYSA-N 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 125000000291 glutamic acid group Chemical group N[C@@H](CCC(O)=O)C(=O)* 0.000 description 1
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 229960001867 guaiacol Drugs 0.000 description 1
- ORUJFMPWKPVXLZ-UHFFFAOYSA-N guaiacol carbonate Chemical compound COC1=CC=CC=C1OC(=O)OC1=CC=CC=C1OC ORUJFMPWKPVXLZ-UHFFFAOYSA-N 0.000 description 1
- DMEGYFMYUHOHGS-UHFFFAOYSA-N heptamethylene Natural products C1CCCCCC1 DMEGYFMYUHOHGS-UHFFFAOYSA-N 0.000 description 1
- 125000005446 heptyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 150000002391 heterocyclic compounds Chemical class 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000009776 industrial production Methods 0.000 description 1
- 238000006713 insertion reaction Methods 0.000 description 1
- 239000013067 intermediate product Substances 0.000 description 1
- 230000009878 intermolecular interaction Effects 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- WYXXLXHHWYNKJF-UHFFFAOYSA-N isocarvacrol Natural products CC(C)C1=CC=C(O)C(C)=C1 WYXXLXHHWYNKJF-UHFFFAOYSA-N 0.000 description 1
- 125000000741 isoleucyl group Chemical group [H]N([H])C(C(C([H])([H])[H])C([H])([H])C([H])([H])[H])C(=O)O* 0.000 description 1
- NFIDBGJMFKNGGQ-UHFFFAOYSA-N isopropylmethylphenol Natural products CC(C)CC1=CC=CC=C1O NFIDBGJMFKNGGQ-UHFFFAOYSA-N 0.000 description 1
- DWKPPFQULDPWHX-VKHMYHEASA-N l-alanyl ester Chemical compound COC(=O)[C@H](C)N DWKPPFQULDPWHX-VKHMYHEASA-N 0.000 description 1
- 150000002605 large molecules Chemical class 0.000 description 1
- 125000001909 leucine group Chemical group [H]N(*)C(C(*)=O)C([H])([H])C(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- DWPCPZJAHOETAG-UHFFFAOYSA-N meso-lanthionine Natural products OC(=O)C(N)CSCC(N)C(O)=O DWPCPZJAHOETAG-UHFFFAOYSA-N 0.000 description 1
- 239000003863 metallic catalyst Substances 0.000 description 1
- RBQRWNWVPQDTJJ-UHFFFAOYSA-N methacryloyloxyethyl isocyanate Chemical compound CC(=C)C(=O)OCCN=C=O RBQRWNWVPQDTJJ-UHFFFAOYSA-N 0.000 description 1
- 125000001434 methanylylidene group Chemical group [H]C#[*] 0.000 description 1
- 125000001360 methionine group Chemical group N[C@@H](CCSC)C(=O)* 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- CEMZBWPSKYISTN-YFKPBYRVSA-N methyl (2s)-2-amino-3-methylbutanoate Chemical compound COC(=O)[C@@H](N)C(C)C CEMZBWPSKYISTN-YFKPBYRVSA-N 0.000 description 1
- UIHPNZDZCOEZEN-YFKPBYRVSA-N methyl (2s)-2-amino-4-methylsulfanylbutanoate Chemical compound COC(=O)[C@@H](N)CCSC UIHPNZDZCOEZEN-YFKPBYRVSA-N 0.000 description 1
- YXMMTUJDQTVJEN-WDSKDSINSA-N methyl (2s,3s)-2-amino-3-methylpentanoate Chemical compound CC[C@H](C)[C@H](N)C(=O)OC YXMMTUJDQTVJEN-WDSKDSINSA-N 0.000 description 1
- XWBUDPXCXXQEOU-UHFFFAOYSA-N methyl 2,4-diamino-4-oxobutanoate Chemical compound COC(=O)C(N)CC(N)=O XWBUDPXCXXQEOU-UHFFFAOYSA-N 0.000 description 1
- QVDXUKJJGUSGLS-LURJTMIESA-N methyl L-leucinate Chemical compound COC(=O)[C@@H](N)CC(C)C QVDXUKJJGUSGLS-LURJTMIESA-N 0.000 description 1
- KQSSATDQUYCRGS-UHFFFAOYSA-N methyl glycinate Chemical compound COC(=O)CN KQSSATDQUYCRGS-UHFFFAOYSA-N 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- GRVDJDISBSALJP-UHFFFAOYSA-N methyloxidanyl Chemical compound [O]C GRVDJDISBSALJP-UHFFFAOYSA-N 0.000 description 1
- XGEGHDBEHXKFPX-NJFSPNSNSA-N methylurea Chemical compound [14CH3]NC(N)=O XGEGHDBEHXKFPX-NJFSPNSNSA-N 0.000 description 1
- AJUXDFHPVZQOGF-UHFFFAOYSA-N n,n-dimethyl-1-naphthylamine Chemical compound C1=CC=C2C(N(C)C)=CC=CC2=C1 AJUXDFHPVZQOGF-UHFFFAOYSA-N 0.000 description 1
- PSHKMPUSSFXUIA-UHFFFAOYSA-N n,n-dimethylpyridin-2-amine Chemical compound CN(C)C1=CC=CC=N1 PSHKMPUSSFXUIA-UHFFFAOYSA-N 0.000 description 1
- MMFBQHXDINNBMW-UHFFFAOYSA-N n,n-dipropylaniline Chemical compound CCCN(CCC)C1=CC=CC=C1 MMFBQHXDINNBMW-UHFFFAOYSA-N 0.000 description 1
- YNTOKMNHRPSGFU-UHFFFAOYSA-N n-Propyl carbamate Chemical compound CCCOC(N)=O YNTOKMNHRPSGFU-UHFFFAOYSA-N 0.000 description 1
- PPHQUIPUBYPZLD-UHFFFAOYSA-N n-ethyl-n-methylaniline Chemical compound CCN(C)C1=CC=CC=C1 PPHQUIPUBYPZLD-UHFFFAOYSA-N 0.000 description 1
- XWCCTMBMQUCLSI-UHFFFAOYSA-N n-ethyl-n-propylpropan-1-amine Chemical compound CCCN(CC)CCC XWCCTMBMQUCLSI-UHFFFAOYSA-N 0.000 description 1
- 150000004002 naphthaldehydes Chemical class 0.000 description 1
- NVSYANRBXPURRQ-UHFFFAOYSA-N naphthalen-1-ylmethanamine Chemical compound C1=CC=C2C(CN)=CC=CC2=C1 NVSYANRBXPURRQ-UHFFFAOYSA-N 0.000 description 1
- MCXRDRHASMNTGZ-UHFFFAOYSA-N naphthalen-1-ylmethanediamine Chemical compound C1=CC=C2C(C(N)N)=CC=CC2=C1 MCXRDRHASMNTGZ-UHFFFAOYSA-N 0.000 description 1
- 125000005186 naphthyloxy group Chemical group C1(=CC=CC2=CC=CC=C12)O* 0.000 description 1
- 229910052754 neon Inorganic materials 0.000 description 1
- GKAOGPIIYCISHV-UHFFFAOYSA-N neon atom Chemical compound [Ne] GKAOGPIIYCISHV-UHFFFAOYSA-N 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 125000006611 nonyloxy group Chemical group 0.000 description 1
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-M octanoate Chemical compound CCCCCCCC([O-])=O WWZKQHOCKIZLMA-UHFFFAOYSA-M 0.000 description 1
- 125000005447 octyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 125000002524 organometallic group Chemical group 0.000 description 1
- 235000010292 orthophenyl phenol Nutrition 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- HFPZCAJZSCWRBC-UHFFFAOYSA-N p-cymene Chemical compound CC(C)C1=CC=C(C)C=C1 HFPZCAJZSCWRBC-UHFFFAOYSA-N 0.000 description 1
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 1
- 125000004817 pentamethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- 125000004115 pentoxy group Chemical group [*]OC([H])([H])C([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- BSCCSDNZEIHXOK-UHFFFAOYSA-N phenyl carbamate Chemical compound NC(=O)OC1=CC=CC=C1 BSCCSDNZEIHXOK-UHFFFAOYSA-N 0.000 description 1
- DGTNSSLYPYDJGL-UHFFFAOYSA-N phenyl isocyanate Chemical compound O=C=NC1=CC=CC=C1 DGTNSSLYPYDJGL-UHFFFAOYSA-N 0.000 description 1
- KRIOVPPHQSLHCZ-UHFFFAOYSA-N phenyl propionaldehyde Natural products CCC(=O)C1=CC=CC=C1 KRIOVPPHQSLHCZ-UHFFFAOYSA-N 0.000 description 1
- 229940100595 phenylacetaldehyde Drugs 0.000 description 1
- 125000004346 phenylpentyl group Chemical group C1(=CC=CC=C1)CCCCC* 0.000 description 1
- 229950009195 phenylpropanol Drugs 0.000 description 1
- QCDYQQDYXPDABM-UHFFFAOYSA-N phloroglucinol Chemical compound OC1=CC(O)=CC(O)=C1 QCDYQQDYXPDABM-UHFFFAOYSA-N 0.000 description 1
- 229960001553 phloroglucinol Drugs 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- ZWLUXSQADUDCSB-UHFFFAOYSA-N phthalaldehyde Chemical compound O=CC1=CC=CC=C1C=O ZWLUXSQADUDCSB-UHFFFAOYSA-N 0.000 description 1
- SATCULPHIDQDRE-UHFFFAOYSA-N piperonal Chemical compound O=CC1=CC=C2OCOC2=C1 SATCULPHIDQDRE-UHFFFAOYSA-N 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229910052573 porcelain Inorganic materials 0.000 description 1
- 238000004094 preconcentration Methods 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- YSTAFYOHERLRBE-UHFFFAOYSA-N pyridin-2-ylmethanediamine Chemical compound NC(N)C1=CC=CC=N1 YSTAFYOHERLRBE-UHFFFAOYSA-N 0.000 description 1
- 229940079877 pyrogallol Drugs 0.000 description 1
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical compound O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 1
- 229910052761 rare earth metal Inorganic materials 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001172 regenerating effect Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- ADZWSOLPGZMUMY-UHFFFAOYSA-M silver bromide Chemical compound [Ag]Br ADZWSOLPGZMUMY-UHFFFAOYSA-M 0.000 description 1
- TYTYIUANSACAEM-UHFFFAOYSA-M silver;2,4,6-trinitrophenolate Chemical compound [Ag+].[O-]C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O TYTYIUANSACAEM-UHFFFAOYSA-M 0.000 description 1
- 238000007711 solidification Methods 0.000 description 1
- 230000008023 solidification Effects 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- DVKJHBMWWAPEIU-UHFFFAOYSA-N toluene 2,4-diisocyanate Chemical compound CC1=CC=C(N=C=O)C=C1N=C=O DVKJHBMWWAPEIU-UHFFFAOYSA-N 0.000 description 1
- 238000005809 transesterification reaction Methods 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- FOETTWZZVDEKIW-UHFFFAOYSA-N triisocyanatomethylbenzene Chemical compound O=C=NC(N=C=O)(N=C=O)C1=CC=CC=C1 FOETTWZZVDEKIW-UHFFFAOYSA-N 0.000 description 1
- 239000013638 trimer Substances 0.000 description 1
- 125000003258 trimethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- KXUFBFHIWJDZPO-UHFFFAOYSA-N tris(2-isocyanatoethyl) hexane-1,3,6-tricarboxylate Chemical compound O=C=NCCOC(=O)CCCC(C(=O)OCCN=C=O)CCC(=O)OCCN=C=O KXUFBFHIWJDZPO-UHFFFAOYSA-N 0.000 description 1
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 1
- 125000002987 valine group Chemical group [H]N([H])C([H])(C(*)=O)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- 150000003739 xylenols Chemical class 0.000 description 1
- 229910001845 yogo sapphire Inorganic materials 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 150000003752 zinc compounds Chemical class 0.000 description 1
- JLVUSDMLNQQPCD-UHFFFAOYSA-L zinc;phenylmethanesulfonate Chemical compound [Zn+2].[O-]S(=O)(=O)CC1=CC=CC=C1.[O-]S(=O)(=O)CC1=CC=CC=C1 JLVUSDMLNQQPCD-UHFFFAOYSA-L 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C269/00—Preparation of derivatives of carbamic acid, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C269/04—Preparation of derivatives of carbamic acid, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups from amines with formation of carbamate groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C269/00—Preparation of derivatives of carbamic acid, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C269/06—Preparation of derivatives of carbamic acid, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups by reactions not involving the formation of carbamate groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C269/00—Preparation of derivatives of carbamic acid, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C269/08—Separation; Purification; Stabilisation; Use of additives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C273/00—Preparation of urea or its derivatives, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups
- C07C273/18—Preparation of urea or its derivatives, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups of substituted ureas
- C07C273/189—Purification, separation, stabilisation, use of additives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B61/00—Other general methods
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/14—The ring being saturated
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/10—Process efficiency
Definitions
- the present invention relates to a method for producing an isocyanate compound, a method for producing a carbamate compound, a method for recovering an amine compound, and an isocyanate composition.
- This application is filed in Japan on November 8, 2021 in Japanese Patent Application Nos. 2021-182191, 2021-182194, and 2021-182195, and on December 8, 2021 in Japan. A priority is claimed based on Japanese Patent Application No. 2021-199645 filed, the content of which is incorporated herein.
- Isocyanates are widely used as raw materials for manufacturing polyurethane foams, polyureas, paints, adhesives, etc.
- the main industrial production method of isocyanate is the reaction of an amine compound with phosgene (phosgene method), and almost all of the production in the world is produced by the phosgene method.
- phosgene method has many problems.
- a method for producing isocyanate compounds that does not use phosgene is desired.
- the heat of a carbamate compound obtained by reacting an amine, at least one of urea and N-unsubstituted carbamic acid ester, and an alcohol (carbamate reaction).
- a decomposition method (urea method) has been proposed (see, for example, Patent Document 1, etc.). It has long been known that isocyanates and hydroxy compounds are obtained by thermal decomposition of carbamate compounds (see, for example, Non-Patent Document 1).
- the basic reaction is exemplified by the following formula (A).
- R is an a-valent organic group
- R' is a monovalent organic group
- a is an integer of 1 or more.
- R a to R h are each independently a monovalent organic group.
- Patent Literatures 2 and 3 disclose a method of continuously producing polyisocyanate by thermal decomposition of a carbamate compound in the presence of a high-boiling-point solvent without a catalyst.
- Patent Document 4 also discloses, at relatively low temperatures, preferably under reduced pressure, in the presence or absence of catalysts and/or stabilizers, without the use of solvents, in good yields, It is disclosed that monocarbamates can be decomposed.
- the decomposition products (monoisocyanates and alcohols) are removed from the boiling reaction mixture by distillation and collected separately by fractional condensation.
- a method for partially removing the reaction mixture to remove by-products formed in the pyrolysis is disclosed in general form.
- Patent Document 1 describes basic catalysts such as lithium, sodium, potassium, calcium, and barium methylate, ethylate, butyrate (each isomer), elemental rare earth elements, antimony, and bismuth, and their Elemental oxides, sulfides and salts, elemental boron and boron compounds, metals of the copper group, zinc group, aluminum group, carbon group and titanium group of the periodic table and their oxides and sulfides, elements of the periodic table Carbides and nitrides of carbon, titanium, vanadium, and chromium group elements other than carbon are disclosed.
- basic catalysts such as lithium, sodium, potassium, calcium, and barium methylate, ethylate, butyrate (each isomer), elemental rare earth elements, antimony, and bismuth, and their Elemental oxides, sulfides and salts, elemental boron and boron compounds, metals of the copper group, zinc group, aluminum group, carbon group and titanium group of the periodic table and their oxide
- Patent Document 5 discloses a method for producing carbamate using zinc toluenesulfonate as a catalyst.
- Patent Document 6 discloses two types of metal compounds
- Patent Document 7 discloses a metal compound containing a non-coordinating anion
- Patent Document 8 discloses the Compounds are disclosed that contain bound one or more cations of metals from Groups IIB, VB, VIIB and VIII.
- Patent Documents 9 to 11 disclose carbamate production methods in which various metal compounds may be used as catalysts.
- isocyanate is produced by the reaction of amine and phosgene, which causes unfavorable polymerization reactions such as multimerization of isocyanate. It is known that boiling point compounds are produced. However, it is known that even in the production of isocyanate by the urea method, a high boiling point compound having a boiling point higher than that of isocyanate is produced after the isocyanate is recovered. A composition containing this high boiling point compound may become a highly viscous liquid or solid at around room temperature, which may cause clogging or the like in the continuous production of the isocyanate compound. Further, the high boiling point compound is a by-product derived from the isocyanate compound and the organic amine compound that is the raw material of the isocyanate compound, and it would be industrially advantageous if it could be recovered as an active ingredient.
- Patent Document 12 discloses a method of separating isocyanate from a diisocyanate-containing organic residue under specific temperature and pressure conditions and carrying out the residue by forced transportation.
- Patent Documents 13 to 17 disclose post-treatment methods for residues generated in isocyanate production.
- Patent Document 18 discloses a method for post-treating the residue generated in the production of isocyanate, in which all of the by-produced gas components are absorbed as carbonate with an alkali metal.
- Patent Document 19 discloses a method of post-treating a distillation residue produced in the synthesis of tolylene diisocyanate by reacting it with water, wherein the distillation residue is treated continuously or semi-continuously in a back-mixing reactor in the presence of a hydrolyzate. is disclosed for reacting with water.
- Patent Document 20 discloses a decomposition recovery method of recovering an isocyanate compound as a raw material by contacting the isocyanate compound with high-temperature, high-pressure water containing one or more compounds selected from the group consisting of ammonia and aliphatic amines. disclosed.
- Patent Document 21 discloses a method of decomposing residues produced in isocyanate production in the presence of high-temperature, high-pressure water and hydroxides of alkali metals or alkaline earth metals.
- Patent Document 1 discloses a method of using a metal element or compound as a catalyst.
- carbamate is used as a raw material for isocyanate, it is desirable to separate it from isocyanate because metals and their compounds modify isocyanate.
- metals and their compounds are very difficult to volatilize, which can impose restrictions on process design.
- Patent Document 19 has the problem that the reaction efficiency is low and it takes a long time to complete the reaction.
- the reaction efficiency depends on the interfacial contact efficiency of the aqueous phase/organic phase, and liquid-liquid phase separation occurs in portions where there is no stirring power, and the reaction efficiency is low. .
- Patent Document 21 has the problem that the alkali metal or alkaline earth metal carbonate added to the reaction solution precipitates as a solid content, causing it to adhere to the inside of the apparatus during separation operations such as distillation. ing.
- the present invention has been made in view of the above circumstances, and provides a novel process that has a good yield of isocyanate compounds, enables stable continuous operation, and suppresses the amount of by-products mixed in.
- a method for producing an isocyanate compound is provided.
- the present invention has been made in view of the above circumstances, and provides a method for producing a carbamate compound that does not use a metal catalyst, is inexpensive, and has a good yield, and the carbamate obtained by the production method.
- a method for producing an isocyanate compound using the compound is provided.
- the present invention has been made in view of the above circumstances, and provides a method for recovering an amine compound capable of efficiently regenerating a useful component including an amine compound from a liquid phase component remaining after production of an isocyanate compound. do.
- the present invention has been made in view of the above circumstances, and when producing an isocyanate compound without using phosgene, it prevents by-products from sticking to the equipment and improves the yield of the isocyanate compound.
- an isocyanate composition that can be obtained and a method for producing an isocyanate compound using the isocyanate composition.
- the method for producing an isocyanate compound according to ⁇ 1> further comprising the step (5) of obtaining the primary amine compound and the hydroxy compound by hydrolyzing under ⁇ 1>.
- ⁇ 3> In the step (1), In the presence of a hydroxy compound, a primary amine compound is reacted with urea or a urea derivative as the carbonic acid derivative, and a compound having a boiling point lower than that of the N-substituted carbamate compound, which is a by-product, is extracted while N-substitution is performed.
- a primary amine compound is reacted with a carbonic acid ester as the carbonic acid derivative to obtain an N-substituted carbamate compound while extracting a hydroxy compound as a by-product having a boiling point lower than that of the N-substituted carbamate compound.
- a method for producing an isocyanate compound according to ⁇ 1> or ⁇ 2>. ⁇ 4> The isocyanate compound according to any one of ⁇ 1> to ⁇ 3>, wherein in the step (2), a liquid phase containing the isocyanate compound is obtained while extracting the by-produced hydroxy compound into the gas phase. Production method.
- the step (1) and the step (2) are performed in one or more selected from the group consisting of a tank reactor, a distillation column, a tubular evaporator, a thin film evaporator, and a falling film evaporator.
- ⁇ 6> The method for producing an isocyanate compound according to any one of ⁇ 1> to ⁇ 5>, wherein at least one of the steps (1) and (2) is performed by a reactive distillation method.
- ⁇ 7> The method for producing an isocyanate compound according to any one of ⁇ 1> to ⁇ 6>, wherein the hydroxy compound extracted in the step (2) is recycled to the step (1) for reuse. . ⁇ 8> Production of the isocyanate compound according to any one of ⁇ 1> to ⁇ 7>, wherein the aprotic solvent separated in the step (3) is recycled to the step (2) for reuse. Method. ⁇ 9> The method for producing an isocyanate compound according to any one of ⁇ 1> to ⁇ 8>, wherein the aprotic solvent is a carbonate ester.
- step (4) in the presence of a carbonyl compound represented by the following general formula (I) in an amount of 1 ppm by mass or more and 50% by mass or less with respect to the mass of the fraction containing the isocyanate compound,
- a carbonyl compound represented by the following general formula (I) in an amount of 1 ppm by mass or more and 50% by mass or less with respect to the mass of the fraction containing the isocyanate compound,
- R 11 is a (n11+n12)-valent organic group
- R 12 is a monovalent organic group.
- n11 is an integer of 1 or more and 8 or less
- n12 is 0 or more and 7 or less. It is an integer, and the sum of n11 and n12 is 2 or more and 8 or less.
- R 21 is an n21-valent organic group. n21 is an integer of 2 or more.
- a carbamate compound including a reaction step of reacting a primary amine compound, a carbonic acid derivative, and a hydroxy compound in the presence of an amine compound having no active hydrogen as a catalyst to obtain a carbamate compound.
- Production method. ⁇ 14>
- the amine compound having no active hydrogen has one or more functional groups selected from the group consisting of a tertiary amino group, a nitrogen-containing aromatic group, an amidine group, and a guanidine group, ⁇ 13> A method for producing the carbamate compound according to .
- ⁇ 15> The method for producing a carbamate compound according to ⁇ 14>, wherein the amine compound having no active hydrogen has one or more tertiary amino groups and has 3 or more and 85 or less carbon atoms. . ⁇ 16> The carbamate compound according to ⁇ 15>, wherein the amine compound having no active hydrogen has 1 to 6 tertiary amino groups and has 3 to 30 carbon atoms. Production method.
- the amine compound having no active hydrogen is N,N'-dimethylaniline, N,N'-diethylaniline, N-methyl-N'-ethylaniline, N,N'-dimethylaminopyridine, N , N,N',N'-tetramethylphenylenediamine, methylenebis(N,N'-dimethylaniline), triethylamine, ethyldiisopropylamine, N-methylmorpholine, N-methylpiperidine, quinuclidine, N,N'-dimethylpiperazine , triethylenediamine, N,N,N'N'-tetramethylethylenediamine, N,N,N'N'-tetramethylhexanediamine, N,N,N'N'-tetramethylxylylenediamine, pentamethyldiethylenetriamine, of the carbamate compound of ⁇ 16>, which is bis(2-morpholinoethyl
- the nitrogen-containing aromatic group is a substituted or unsubstituted pyridyl group, imidazolyl group, pyrazolyl group, quinolyl group, isoquinolyl group, oxazolyl group, thiazolyl group, pyridazyl group, pyrimidyl group, or pyrazyl group.
- the amine compound having no active hydrogen is pyridine, picoline, lutidine, collidine, 1-methylimidazole, 1-methylpyrazole, quinoline, isoquinoline, methylquinoline, oxazole, thiazole, pyridazine, pyrimidine, or pyrazine.
- R 811 , R 812 , R 813 and R 814 are each independently a monovalent organic group.
- R 811 and R 812 , R 812 and R 813 , R 813 and Each of R 814 , R 814 and R 811 may be independently bonded to each other to form a ring structure, and the total carbon number of R 811 , R 812 , R 813 and R 814 is 5 or more and 85 or less. be.
- the amidine group-containing compound has one or more ring structures, and the total number of carbon atoms of R 811 , R 812 , R 813 and R 814 is 5 or more and 30 or less.
- the amidine group-containing compound is 1,2-dimethyl-1,4,5,6-tetrahydropyrimidine, 1,8-diazabicyclo-[5.4.0]undec-7-ene, or 1,5 -Diazabicyclo-[4.3.0]non-5-ene, the method for producing the carbamate compound according to ⁇ 22>.
- ⁇ 24> The method for producing a carbamate compound according to ⁇ 14>, wherein the amine compound having no active hydrogen is a guanidine group-containing compound represented by the following general formula (VIII-2).
- R 821 , R 822 , R 823 , R 824 and R 825 are each independently a monovalent organic group.
- R 821 and R 822 , R 822 and R 823 , R 823 and R 824 , R 824 and R 825 , R 825 and R 821 may each independently combine with each other to form a ring structure, R 821 , R 822 , R 823 , R 824 , and
- the total carbon number of R825 is 5 or more and 85 or less.
- ⁇ 25> The method for producing a carbamate compound according to ⁇ 24>, wherein the total number of carbon atoms of R 821 , R 822 , R 823 , R 824 and R 825 is 5 or more and 30 or less.
- ⁇ 26> The carbamate according to ⁇ 25>, wherein the guanidine group-containing compound is pentamethylguanidine or 7-methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene A method for producing a compound.
- ⁇ 27> The method for producing a carbamate compound according to any one of ⁇ 13> to ⁇ 26>, wherein the primary amine compound is an aliphatic or aromatic primary polyamine compound.
- ⁇ 28> The method for producing a carbamate compound according to any one of ⁇ 13> to ⁇ 27>, wherein the hydroxy compound is an aromatic hydroxy compound.
- An amine compound represented by the following general formula (IIa) is obtained from a liquid phase component containing a compound having a boiling point higher than that of the isocyanate compound, which is by-produced in a method for producing an isocyanate compound represented by the following general formula (VIIa):
- a method of recovering a A step of reacting the liquid phase component, the aromatic hydroxy compound, the active hydrogen-containing compound, and the catalyst in a reactor to obtain a reaction liquid containing the amine compound represented by the general formula (IIa) ( A recovery method comprising a).
- R 71a is an n71a-valent organic group. n71a is an integer of 2 or more and 8 or less.
- step (a) includes the following steps (a1) and (a2).
- step (a2) ⁇ 32>
- step (a2-1) of mixing the mixture obtained in step (a1), an amine compound as an active hydrogen-containing compound, and a catalyst;
- step (a2-1) of reacting the mixture obtained in the step (a2-1) with water as an active hydrogen-containing compound in a reactor to obtain a reaction liquid containing the amine compound represented by the general formula (IIa).
- the liquid phase component is subjected to a thermal decomposition reaction of a reaction liquid containing a carbamate compound produced from a carbonic acid derivative, a hydroxy compound, and an amine compound represented by the general formula (IIa) to produce
- the composition containing the isocyanate compound represented by the general formula (VIIa) is supplied to a distillation apparatus, and when separating the isocyanate compound represented by the general formula (VIIa) as a gas phase component, the distillation
- the recovery method according to any one of ⁇ 30> to ⁇ 36>, wherein the liquid phase component is extracted from the device.
- the liquid phase component is a group represented by the following formula (IX-1), a group represented by the following formula (IX-2), a group represented by the following formula (IX-3), or a group represented by the following formula (IX-3).
- the liquid phase component is a group represented by the formula (IX-1), a group represented by the formula (IX-2), a group represented by the formula (IX-3), or a group represented by the formula (IX-3).
- the catalyst comprises alkali metal hydroxides and oxides, alkaline earth metal hydroxides and oxides, tertiary amine compounds, Group 12 metal oxides, and Group 13 metal oxides. and the recovery method according to any one of ⁇ 30> to ⁇ 39>, wherein the compound is one or more compounds selected from the group consisting of Group 14 metal oxides.
- ⁇ 42> The recovery method according to ⁇ 41>, wherein the catalyst is an alkali metal hydroxide or oxide.
- an isocyanate compound a carbonyl compound represented by the following general formula (I);
- An isocyanate composition comprising The isocyanate compound and the carbonyl compound are different compounds, ⁇ 3 ⁇ (molar amount of isocyanurate group) + 2 ⁇ (molar amount of carbodiimide group) + 3 ⁇ (molar amount of uretonimine group) + 2 ⁇ (molar amount of allophanate group) ⁇ ⁇ (molar amount of carbonyl compound) is An isocyanate composition that is 0.001 or more and 8.0 or less.
- R 11 is a (n11+n12)-valent organic group
- R 12 is a monovalent organic group.
- n11 is an integer of 1 or more and 8 or less
- n12 is 0 or more and 7 or less.
- the sum of n11 and n12 is an integer of 2 or more and 8 or less.
- R 11 is a divalent to tetravalent aliphatic hydrocarbon group having 1 to 20 carbon atoms, which may have 1 to 4 ester groups or nitrogen atoms, or 6 carbon atoms is a divalent or more and trivalent or less aromatic hydrocarbon group of 20 or less
- the isocyanate composition according to ⁇ 48> or ⁇ 49>, wherein the isocyanate compound is a compound represented by the following general formula (VIIb).
- the isocyanurate group, the carbodiimide group, the uretonimine group, and the allophanate group are represented by the following general formulas (X-1), (X-2), (X-3), and (X-4), respectively.
- the content of the isocyanate compound is 1% by mass or more and 99% by mass or less
- n111 is an integer of 1 or more and 8 or less
- n112 is an integer of 0 or more and 7 or less
- the sum of n111 and n112 is an integer of 2 or more and 8 or less
- satisfies the relational expression: n111+n112 n11+n12.
- ⁇ 54> The carbamate group-containing compound represented by the general formula (XI) and the general formula, each in an amount of 2.0 ppm by mass or more and 1.0 ⁇ 10 4 ppm by mass or less with respect to the total mass of the isocyanate composition
- ⁇ 55> A method for producing an isocyanate compound, comprising distilling and purifying the isocyanate composition according to any one of ⁇ 48> to ⁇ 54> and continuously recovering the isocyanate compound as a gas phase component.
- the yield is good, stable continuous operation is possible, and the amount of mixed by-products can be suppressed.
- the isocyanate composition of the above aspect when the isocyanate compound is produced without using phosgene, it is possible to prevent by-products from sticking to the equipment and to provide an isocyanate composition capable of improving the yield of the isocyanate compound. can do.
- the method for producing an isocyanate compound according to the above aspect is a method using the isocyanate composition, and can prevent by-products from sticking to equipment and improve the yield of the isocyanate compound.
- FIG. 1 is a schematic configuration diagram showing an apparatus used in step (1) in Example 1-1.
- FIG. 1 is a schematic configuration diagram showing an apparatus used in step (1a) in Example 1-1.
- FIG. 1 is a schematic configuration diagram showing an apparatus used in step (2) in Example 1-1.
- FIG. FIG. 2 is a schematic configuration diagram showing an apparatus used in step (3) in Example 1-1;
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (4) in Example 1-1;
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (6) in Example 1-1;
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (5) in Example 1-1;
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (5) in Example 1-3;
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (2) in Example 1-7;
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (2) in Example 1-8.
- 1 is a schematic configuration diagram showing an apparatus used in step (1) in Example 1-9.
- FIG. FIG. 2 is a schematic configuration diagram showing an apparatus used in step (2) in Example 1-9.
- 1 is a schematic configuration diagram showing an apparatus used in step (1) in Example 1-16.
- FIG. FIG. 2 is a schematic configuration diagram showing an apparatus used in step (1b) in Example 1-16;
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (2) in Example 1-16.
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (1a) in Comparative Example 1-6.
- FIG. 4 is a schematic configuration diagram showing an apparatus used in step (1b) in Comparative Example 1-6.
- FIG. 2 is a schematic configuration diagram showing an apparatus used in step (2) in Comparative Example 1-6;
- 1 is a schematic configuration diagram of an apparatus for producing a carbamate compound in Examples.
- FIG. It is an isocyanate compound manufacturing apparatus used in Examples. 1 shows an amine compound recovery apparatus used in Examples. It is a separation apparatus for amine compounds used in Examples. 1 shows an apparatus for producing a carbamate compound used in Examples. It is an apparatus used in the pre-concentration step of a reaction solution containing a carbamate compound in Examples. It is an isocyanate compound manufacturing apparatus used in Examples. 1 shows an amine compound recovery apparatus used in Examples.
- 1 shows an apparatus for producing a carbamate compound used in Examples.
- 1 shows an amine compound recovery apparatus used in Examples. It is an apparatus used in the transesterification step of a carbamate compound in Examples. It is an isocyanate compound manufacturing apparatus used in Examples.
- 1 shows an amine compound recovery apparatus used in Examples. It is an apparatus used in the thermal decomposition step of a carbamate compound in Examples. It is an apparatus used in the isocyanate compound purification step (light boiling point separation step) in Examples. It is an apparatus used in the isocyanate compound purification step (high boiling point separation step) in the examples.
- 1 shows an amine compound recovery apparatus used in Examples. It is an apparatus used in the thermal decomposition step of the carbamate compound used in the examples.
- FIG. 1 is a schematic configuration diagram showing an apparatus for producing a carbamate compound used in Examples.
- FIG. 1 is a schematic configuration diagram showing a pyrolysis reactor used in Examples.
- FIG. 1 is a schematic configuration diagram showing a low boiling point separation apparatus used in Examples.
- FIG. 1 is a schematic configuration diagram showing a high-boiling separator used in Examples.
- this embodiment The best mode for carrying out the present invention (hereinafter referred to as "this embodiment") will be described in detail below. It should be noted that the present invention is not limited to the following embodiments, and various modifications can be made within the scope of the gist of the present invention.
- organometallic compounds also include organometallic compounds and metal complexes.
- terms such as "organic group” and “substituent” mean groups composed of atoms that do not contain metal atoms and semimetals. Further, in the present embodiment, preferably H (hydrogen atom), C (carbon atom), N (nitrogen atom), O (oxygen atom), S (sulfur atom), Cl (chlorine atom), Br (bromine atom) , I (iodine atom).
- aliphatic and “aromatic” are used frequently. According to the IUPAC rules mentioned above, it is stated that organic compounds are classified into aliphatic compounds and aromatic compounds. Aliphatic is the definition of the group according to the 1995 IUPAC Recommendations for aliphatic compounds. The recommendation defines aliphatic compounds as "Acyclic or cyclic, saturated or unsaturated carbon compounds, excluding aromatic compounds".
- the "aliphatic compound” used in the description of the present embodiment contains both saturated and unsaturated, chain and cyclic, and the above H (hydrogen atom); C (carbon atom); N ( nitrogen atom); O (oxygen atom); S (sulfur atom); Si (silicon atom); Cl (chlorine atom), Br (bromine atom) or I (iodine atom).
- H hydrogen atom
- C carbon atom
- N nitrogen atom
- O oxygen atom
- S sulfur atom
- Si silicon atom
- Cl chlorine atom
- Br bromine atom
- I iodine atom
- an aromatic group such as an aralkyl group
- the term "aliphatic group substituted with an aromatic group” or "group consisting of an aliphatic group to which an aromatic group is bonded” is used. may be indicated. This is based on the reactivity in the present embodiment, and is because the reactive properties of groups such as aralkyl groups are very similar to the reactivity of aliphatic rather than aromatic groups.
- non-aromatic reactive groups including aralkyl groups, alkyl groups, etc. are defined as "aliphatic groups optionally substituted with aromatic groups” and "aliphatic groups optionally bonded with aromatic groups”. etc.
- active hydrogen refers to a hydrogen atom bonded to an oxygen atom, a sulfur atom, a nitrogen atom, a silicon atom, or the like, and a hydrogen atom of a terminal methine group.
- the hydrogen of the hydroxy group is also an active hydrogen, but the hydroxy group is also contained in the composition containing the product obtained by the production method of the present embodiment and the reaction solution containing the reaction raw materials, and is not a group that exerts an adverse effect. Therefore, unless otherwise specified, groups containing active hydrogen exclude hydroxy groups.
- the compound having a hydroxy group includes alcohols and aromatic hydroxy compounds.
- alcohol used herein refers to compounds in which a hydroxy group, -OH, is attached to a saturated carbon atom: R 3 COH)” and does not include aromatic hydroxy compounds in which a hydroxy group is attached to an aromatic ring.
- aromatic hydroxy compound refers to phenols described in the IUPAC definition (Rule C-202) "one or more hydroxy groups attached to a benzene ring or other arene ring. "Compounds having one or more hydroxy groups attached to a benzene or other arene ring.”
- the method for producing an isocyanate compound of the present embodiment includes steps (1) to (4) shown below.
- a primary amine compound and a carbonic acid derivative are reacted, and a compound having a boiling point lower than that of the N-substituted carbamate compound (hereinafter also referred to as a "low-boiling compound”), which is a by-product, is extracted while extracting the N-substituted carbamate.
- step (1) of obtaining a compound Using the reaction solution containing the N-substituted carbamate compound obtained in the step (1), the N-substituted carbamate compound is thermally decomposed in the presence of an aprotic solvent, and the hydroxy compound produced as a by-product is removed.
- Step (2) of obtaining an isocyanate compound while extracting Step (3) of separating the isocyanate compound and the aprotic solvent from the reaction solution containing the isocyanate compound obtained in the step (2); Purifying the isocyanate compound by removing a component having a boiling point higher than that of the isocyanate compound (hereinafter sometimes simply referred to as a "high boiling point component") from the fraction containing the isocyanate compound obtained in the step (3).
- the production reaction of the N-substituted carbamate compound is performed while extracting the low-boiling compound produced as a by-product in the step (1), and the hydroxy compound produced as a by-product in the step (2) is extracted.
- the reaction equilibrium constant K is about 10 ⁇ 3 or more and 10 ⁇ 1 or less, and the reactions of steps (1) and (2) that are biased toward the original system , the reaction equilibrium can be shifted toward the production system, and the yield of the isocyanate compound can be improved.
- the high boiling point component in the fraction containing the isocyanate compound functions as a solvent, so that the amount of the high boiling point component mixed is suppressed, and the isocyanate compound is isolated and purified with high purity. be able to.
- Step (1) N-substituted carbamate compound production step>
- a primary amine compound and a carbonic acid derivative are reacted, and an N-substituted carbamate compound is obtained while a low-boiling compound by-produced is extracted.
- Step (1) can also be called an N-substituted carbamate compound production step.
- step (1) a primary amine compound and a carbonic acid derivative are reacted to convert the terminal amino group of the primary amine compound to obtain an N-substituted carbamate compound.
- urea when urea is used as the carbonic acid derivative, a reaction intermediate is produced in which the terminal amino group of the primary amine compound becomes a ureido group in the presence of a hydroxy compound, and then the terminal ureido group and the hydroxy compound are further formed. reacts to give a compound in which the terminal ureido group is substituted with a terminal structure in which the residue obtained by removing the hydroxyl group from the hydroxy compound via the carbamate group is bonded, ie, an N-substituted carbamate compound. At this time, the by-produced low-boiling compound becomes ammonia.
- the low-boiling compound produced as a by-product corresponds to an alkyl group substituted for the amino group of ammonia and urea.
- urea is used as the carbonic acid derivative in step (1).
- the process is not particularly limited as long as the reaction to produce the N-substituted carbamate compound via the ureide intermediate and the extraction of by-product ammonia can be performed simultaneously. It is preferably carried out using an evaporator or a falling film evaporator, more preferably a distillation column, a thin film evaporator or a falling film evaporator, and still more preferably a distillation column or a falling film evaporator, Particular preference is given to using a distillation column. When a distillation column is used, it is preferable to use a technique called reactive distillation, because the process involves chemical changes and reactions.
- the reactive distillation method is a method in which the above-described reaction is performed in a reactor by a mixture of a hydroxy compound, a primary amine compound, and one or more compounds selected from the group consisting of urea and urea derivatives, and by-products Ammonia is extracted from the reactor by distillation. Since ammonia has a lower boiling point than the N-substituted carbamate compound, which is the main product, by extracting ammonia, which is a by-product, as a gas phase component, a fraction containing the N-substituted carbamate compound can be obtained as a liquid phase component. can be efficiently obtained.
- step (1) is preferably performed by a continuous method.
- the continuous method the above-described mixture is continuously supplied to a reactor to carry out a production reaction of an N-substituted carbamate compound via a ureide intermediate, and by-produced ammonia is continuously discharged from the reactor. It is a method of extracting.
- the rate at which the mixture is supplied to the reactor and the rate at which ammonia is withdrawn can be appropriately adjusted according to the rate of production of the N-substituted carbamate compound and ammonia as products.
- the amounts of the primary amine compound and one or more compounds selected from the group consisting of urea and urea derivatives are such that the molar ratio of the amino group of the primary amine compound to the urea group of the compound is 1:1.
- the amount can be 3 to 3:1, preferably 1:2 to 2:1, more preferably 1:1.5 to 1.5 to 1, and 1:1. Amount is more preferred. When the amount of the compound used for the two weeks is within the above range, the ureidation reaction can be carried out more efficiently.
- the amount of the hydroxy compound used is preferably such that the molar ratio of the hydroxyl group of the hydroxy compound to the amino group of the primary amine compound is 1/1 or more and 300/1 or less, and the amount that becomes 1/1 or more and 250/1 or less. More preferably, the amount of 1/1 or more and 200/1 or less is even more preferable.
- the molar ratio of the hydroxyl group of the hydroxy compound to the amino group of the primary amine compound is at least the above lower limit, the carbamate group can be produced more efficiently.
- the amount is equal to or less than the above upper limit, the amount of the hydroxy compound to be used can be further reduced while maintaining the efficiency of carbamate group formation.
- carrier agent refers to an agent that is substantially inert and in a gaseous state under pyrolysis reaction conditions.
- Such carriers include inert gases, hydrocarbon gases, and the like.
- inert gases include nitrogen, argon, helium, carbon dioxide, methane, ethane, and propane.
- an inert gas such as nitrogen is preferable as the carrier.
- a low boiling point organic solvent may also be used to achieve the same effect.
- Low boiling point organic solvents include, for example, halogenated hydrocarbons, lower hydrocarbons, ethers and the like.
- Halogenated hydrocarbons include, for example, dichloromethane, chloroform, carbon tetrachloride and the like.
- Examples of lower hydrocarbons include pentane, hexane, heptane, and benzene.
- Ethers include, for example, tetrahydrofuran and dioxane.
- carrier agents may be used alone or in combination of two or more. Moreover, it is preferable to heat these carriers in advance before use.
- the reaction temperature varies depending on the type of primary amine compound used and the reaction pressure, but is preferably 40° C. or higher and 380° C. or lower, more preferably 50° C. or higher and 320° C. or lower, and 60° C. or higher and 300° C. or lower. more preferably 80° C. or higher and 300° C. or lower, and most preferably 100° C. or higher and 280° C. or lower.
- the reaction pressure varies depending on the type of compound used and the reaction temperature, and may be any of reduced pressure, normal pressure, and increased pressure.
- the pressure is preferably 20 Pa or more and 2 ⁇ 10 6 Pa or less, more preferably 30 Pa or more and 1.5 ⁇ 10 6 Pa or less, and 50 Pa or more and 1 ⁇ 0 6 Pa or less. It is more preferable to carry out within the range of 50 Pa or more and 0.5 ⁇ 0 6 Pa or less, particularly preferably.
- the reaction time (residence time in the case of a continuous process) is not particularly limited, and is preferably 0.001 hours or more and 100 hours or less, more preferably 0.005 hours or more and 50 hours or less. More preferably, it is 0.01 hours or more and 10 hours or less.
- a carbonate ester is a compound in which one or two hydrogen atoms of carbonic acid CO(OH) 2 are substituted with an alkyl group or an aryl group, and is represented by the general formula (III) described later. is a compound.
- a compound substituted with an alkyl group or an aryl group derived from a carbonate via a carbamate group, that is, an N-substituted carbamate compound is obtained.
- the low-boiling compound produced as a by-product is a hydroxy compound composed of an alkyl group or aryl group derived from a carbonate ester and a hydroxy group.
- the reaction conditions between the carbonate ester and the primary amine compound vary depending on the compound to be reacted, but the molar amount of the carbonate ester relative to the molar amount of the terminal amino group of the primary amine compound is at least 1 times the stoichiometric ratio. is preferable, and an amount of 1.01 times or more and 1000 times or less is more preferable.
- the molar amount of the carbonate ester is preferably an excess amount relative to the molar amount of the terminal amino group of the primary amine, and the size of the carbamate-forming reactor is taken into consideration. For example, an amount of 1.01 times or more and 50 times or less is more preferable, and an amount of 1.2 times or more and 10 times or less is particularly preferable.
- the reaction between the carbonate ester and the primary amine compound is preferably carried out in a liquid phase using an appropriate solvent.
- the solvent can be appropriately selected depending on the compound to be used. Atomic compounds, halogenated aliphatic hydrocarbons, halogenated aromatic hydrocarbons, and water can be used, and these compounds can also be used in appropriate combinations.
- the primary amine compound is preferably present in a liquid state in the carbamate reaction reactor.
- the primary amine compound is preferably supplied in a state of being dissolved in a solvent, and the solvent described above is preferably used as the solvent for dissolving the primary amine compound.
- the primary amine compound feed is carried out as a mixture with the hydroxy compound, water, or carbonate.
- the carbamate reaction in step (1) is carried out by supplying a carbonate ester and a primary amine compound to the carbamate reaction reactor.
- the reaction temperature is preferably 0°C or higher and 150°C or lower.
- a high temperature is preferable for increasing the reaction rate, but from the viewpoint of suppressing undesirable side reactions, a temperature of 10° C. or more and 100° C. or less is more preferable.
- the carbamate reactor may be equipped with a known cooling device or heating device.
- the reaction pressure varies depending on the type of compound used and the reaction temperature, but may be reduced pressure, normal pressure, or increased pressure, and is usually carried out in the range of 20 Pa or more and 1 ⁇ 10 6 Pa or less.
- the reaction time (residence time in the case of a continuous method) is not particularly limited, and is usually preferably 0.001 hour or more and 50 hours or less, more preferably 0.01 hour or more and 20 hours or less, and 0.1 hour or more and 10 hours or less. is more preferred.
- the reaction solution can be sampled and, for example, by liquid chromatography to confirm that the desired amount of carbamic acid ester is produced, the reaction can be terminated.
- a catalyst may or may not be used in addition to these compounds in the reaction between the carbonate ester and the primary amine compound.
- no catalyst is used, thermal denaturation of the carbamic acid ester due to the influence of the metal component derived from the catalyst can be prevented.
- the reaction can be completed in a short time and the reaction temperature can be lowered.
- organometallic compounds or inorganic metal compounds such as tin, lead, copper and titanium; alcoholates of alkali metals or alkaline earth metals such as lithium, sodium, potassium, calcium or barium; Basic catalysts such as methylate, ethylate or butyrate (each isomer) and the like can be used.
- a reactor that meets the conditions is appropriately selected in the reaction for producing the N-substituted carbamate compound.
- the reactor may be a plug flow reactor or a tank reactor.
- conventionally known reactors such as a stirring tank, a pressurized stirring tank, a reduced pressure stirring tank, a column reactor, a distillation column, a packed column, and a thin film distiller can be used in combination as appropriate.
- a known distillation apparatus is preferably used to efficiently recover the gas phase component.
- step (1) is performed using one or more reactors selected from the group consisting of tank reactors, distillation columns, tubular evaporators, thin film evaporators, and falling film evaporators. is particularly preferred.
- a packing material generally used for distillation towers and absorption towers can be appropriately used.
- preferred solid fillers include Raschig rings, cascade mini rings, lessing rings, spiral rings, ball rings, interlox saddles, Stedman packings, McMahon packings, Dixon packings, helix packings, coil packings, heat pipe packings, and the like. is mentioned.
- the distillation column is a plate column, various plate columns having sieve trays, cascade trays, turbogrid trays, ripple trays, dual trays and the like can be used as appropriate.
- the material of the solid filler is preferably inert to the decomposition products, and is not particularly limited to porcelain, metal, or the like. Among them, a material having high thermal conductivity is preferable as the material of the solid filler.
- Materials include SUS304, SUS316, SUS316L, and generally silicon oxide (compositional formula: SiO2), aluminum oxide (compositional formula: Al2O3), and carbon fluoride (compounds with -CHF- or CF2- repeating units). preferable. Glass, ceramic, and fluororesin containing these as constituent components are also preferable. In the case of glass or ceramic, the contents of silicon oxide and aluminum oxide are not particularly limited, and even if the content of silicon oxide accounts for 60% by mass or more, the content of aluminum oxide accounts for 60% by mass or more. and various materials can be selected.
- Ceramics or “ceramics” here refers to sintered bodies made by firing inorganic substances.
- inorganic solid materials such as semiconductors such as silicon, and inorganic compounds such as carbides, nitrides, borides, and titanium oxide molded bodies, powders, and films.
- Fluorine resins include polytetrafluoroethylene, polychlorotrifluoroethylene, polyvinylidene fluoride, polyvinyl fluoride, perfluoroalkoxy fluororesin, ethylene tetrafluoride/propylene hexafluoride copolymer, and ethylene/tetrafluoroethylene copolymer. polymers, ethylene/chlorotrifluoroethylene copolymers, and the like.
- the preferred filler shown here may contain other metal atoms within the scope of the present embodiment.
- the type of condenser provided in the reactor is not particularly limited, and known condensers can be used.
- conventionally known condensers such as multi-tube cylindrical condensers, double-tube condensers, single-tube condensers, and air-cooled condensers can be appropriately combined and used.
- the condenser may be provided inside the reactor or may be provided outside the reactor and connected to the reactor by piping. Various forms are adopted in consideration of the handling method of .
- the materials of the reactor and condenser are not particularly limited, and known materials can be used as long as they do not adversely affect the N-substituted carbamate compound and the product isocyanate.
- it can be made of glass, stainless steel, carbon steel, hastelloy, or a substrate having a glass lining or a Teflon (registered trademark) coating.
- SUS304, SUS316, SUS316L, etc. can be preferably used because they are also inexpensive.
- known process devices such as instrumentation equipment such as flowmeters and thermometers, reboilers, pumps, and condensers may be added. , cooling water, brine and the like can be used. You may add a process as needed.
- (ureidation reaction) may be carried out in the same reactor or separately by reacting a reaction intermediate having a ureido group with a hydroxy compound to produce an N-substituted carbamate compound.
- a reactor of For the ureidation reaction conventionally known methods such as a stirred tank, a pressurized stirred tank, a reduced pressure stirred tank, a column reactor, a continuous stirred reactor, a distillation column, a packed column, a plug flow reactor, a thin film distiller, etc.
- a suitable combination of reactors can be used, preferably a stirring tank, a pressurized stirring tank, a reduced pressure stirring tank, a tower reactor, a continuous stirring reactor, or a plug flow reactor, and from the viewpoint of production efficiency, a continuous stirring type More preferably, it is carried out in a continuous process using a reactor or a plug flow reactor.
- step (1) other steps can be added as necessary.
- step (1) for example, using the N-substituted carbamate obtained in the above reaction and a hydroxy compound different from the hydroxy compound used in the above reaction, the N-substituted carbamate is transesterified to obtain a different N- A step of producing a substituted carbamate; a step of separating a part or all of the hydroxy compound from the reaction solution obtained by the above reaction; a step of recovering ammonia produced by the above reaction; process and the like.
- step (1) it is also one of preferred embodiments to control the reaction conditions by appropriately refluxing as necessary.
- Step (2) isocyanate compound production step (thermal decomposition step)>
- step (2) using the reaction solution containing the N-substituted carbamate compound obtained in step (1), the N-substituted carbamate compound is thermally decomposed in the presence of an aprotic solvent, and the by-produced hydroxy An isocyanate compound is obtained while extracting the compound.
- the step (2) can also be called an isocyanate compound production step or a thermal decomposition step.
- an isocyanate compound is obtained by a thermal decomposition reaction of the N-substituted carbamate compound.
- the hydroxy compound produced as a by-product is the hydroxy compound used in the step (1) or the hydroxy compound derived from the carbonate ester used in the step (1) (in the compound represented by the general formula (III-1) described later, It is a compound having the same structure as the hydroxy compound having a structure in which a hydroxy group is bonded to R 311 or R 312 .
- the step (2) there is no particular limitation as long as the thermal decomposition reaction of the N-substituted carbamate compound and the extraction of the by-product hydroxy compound can be performed simultaneously, but the step (2) includes a stirring tank and a pressurized stirring tank. , a pressure-reduced stirring tank, a column reactor, a continuous stirring reactor, a distillation column, a tubular evaporator, a thin film evaporator, or a falling film evaporator, preferably a distillation column, a thin film evaporator or a falling film evaporator is more preferred, a distillation column or a falling film evaporator is more preferred, and a distillation column is particularly preferred.
- step (2) using the reactive distillation method is a method in which the thermal decomposition reaction of the N-substituted carbamate compound is carried out in a reactor, and the by-produced hydroxy compound is extracted from the reactor by distillation.
- step (1) described above it is preferable to use a hydroxy compound having a normal boiling point lower than that of the isocyanate compound, which is the main product. A fraction containing an isocyanate compound as a phase component can be obtained more efficiently.
- the term "normal boiling point” as used herein refers to the boiling point under 1 atm.
- a hydroxy compound having a higher normal boiling point than the isocyanate compound which is the main product.
- a hydroxy compound can be extracted as a liquid phase component, and a fraction containing an isocyanate compound can be obtained as a gas phase component.
- the difference in normal boiling point between the isocyanate compound and the hydroxy compound is preferably 300°C or less, more preferably 250°C or less, even more preferably 200°C or less.
- the lower limit of the difference in normal boiling point between the isocyanate compound and the hydroxy compound is usually 10°C or higher, preferably 25°C or higher, more preferably 50°C or higher, and even more preferably 100°C or higher.
- step (2) is preferably performed by a continuous method.
- a reaction solution containing an N-substituted carbamate compound is continuously supplied to a reactor to carry out a thermal decomposition reaction of the N-substituted carbamate compound, and a by-produced hydroxy compound is continuously discharged from the reactor. It is a method of extracting effectively.
- the speed of supplying the reaction solution to the reactor and the speed of withdrawing the hydroxy compound described above can be appropriately adjusted according to the production speed of the isocyanate compound and the hydroxy compound, which are the products.
- the normal boiling point of the aprotic solvent is preferably lower than the normal boiling point of the isocyanate compound and higher than that of the hydroxy compound.
- the difference between the normal boiling points of the isocyanate compound and the aprotic solvent is preferably 100°C or less, more preferably 90°C or less, and even more preferably 80°C or less.
- the lower limit of the difference between the normal boiling points of the isocyanate compound and the aprotic solvent is usually 10°C or higher, preferably 15°C or higher, more preferably 20°C or higher, further preferably 25°C or higher, and 30°C or higher. is particularly preferred.
- the difference between the normal boiling points of the isocyanate compound and the aprotic solvent is equal to or less than the above upper limit, when the hydroxy compound after thermal decomposition is extracted into the gas phase, entrainment of the aprotic solvent can be further suppressed, and the liquid phase By preventing the isocyanate from increasing in concentration, it is possible to further reduce the reverse reaction between the generated isocyanate and the hydroxy compound, the modification reaction with the raw material carbamate, and the increase in the number of isocyanates. On the other hand, when it is at least the above lower limit, the aprotic solvent and the isocyanate compound can be separated more efficiently in the step (3) described later, and the yield of the isocyanate compound can be further improved. .
- the normal boiling point of the isocyanate compound is usually 150° C. or higher, preferably 250° C. or higher, more preferably 255° C. or higher, further preferably 300° C. or higher, and 350° C. or higher. is particularly preferred.
- the upper limit of the normal boiling point of the isocyanate compound is not particularly limited, it is preferably 450°C or lower, more preferably 430°C or lower, and even more preferably 420°C or lower. When the normal boiling point of the isocyanate compound is at least the above lower limit, there is a tendency that the isocyanate compound is more easily separated from the by-product hydroxy compound in step (2).
- the amount of the aprotic solvent used is not particularly limited, and is usually about 0.1% by mass or more and 100000% by mass or less with respect to the total mass of the reaction liquid containing the N-substituted carbamate compound as a starting material. range, preferably 1% by mass or more and 10000% by mass or less, more preferably 5% by mass or more and 5000% by mass or less, further preferably 10% by mass or more and 4000% by mass or less, and 20 It is more preferably 3000% by mass or less, particularly preferably 20% by mass or more and 2500% by mass or less, and most preferably 30% by mass or more and 2000% by mass or less.
- the amount of the aprotic solvent used is at least the above lower limit, the concentration of the isocyanate compound produced is further reduced, the reaction between the isocyanate compounds during thermal decomposition produces nurate, and the isocyanate compound and the raw material N-substituted carbamate. Allophanate formation due to reaction of the compound can be further suppressed, the thermal decomposition yield of the isocyanate compound can be further improved, and precipitation of the polymer can be further suppressed.
- the amount of the aprotic solvent used is equal to or less than the above upper limit, it becomes possible to further improve the production efficiency of the isocyanate compound in the same apparatus.
- a single aprotic solvent may be used for the reaction, or a plurality of types of aprotic solvents may be used in combination, which can be arbitrarily selected according to the reaction conditions.
- the thermal decomposition temperature of the N-substituted carbamate compound varies depending on the type of the N-substituted carbamate compound used, but is preferably 140°C or higher and 380°C or lower, more preferably 160°C or higher and 320°C or lower, and 180°C. It is more preferably 300°C or higher, particularly preferably 200°C or higher and 300°C or lower, and most preferably 220°C or higher and 280°C or lower.
- the thermal decomposition reactor may be equipped with a known cooling device and heating device.
- the thermal decomposition reaction of the N-substituted carbamate compound proceeds further, and the higher the temperature, the faster the thermal decomposition rate, and the thermal decomposition of the N-substituted carbamate compound occurs in a short time. complete.
- the modification reaction from isocyanate to nurate or the like can be further suppressed.
- the production amount of the isocyanate polymer produced by the nurate conversion can be further reduced, adhesion to the reaction apparatus and clogging of piping can be further suppressed, and operability can be further improved.
- reaction pressure varies depending on the type of compound used and the reaction temperature, and may be any of reduced pressure, normal pressure, and increased pressure.
- the pressure can be set to the saturated vapor pressure of the aprotic solvent used, and is preferably in the range of 20 Pa or more and 10 ⁇ 10 6 Pa or less.
- the reaction time (residence time in the case of a continuous process) is not particularly limited, and is preferably 0.001 hours or more and 100 hours or less, more preferably 0.005 hours or more and 50 hours or less. More preferably, it is 0.01 hours or more and 10 hours.
- a catalyst is not always necessary in the thermal decomposition reaction, but a catalyst may be used to lower the reaction temperature or complete the reaction early.
- the catalyst is preferably used in an amount of 0.01% to 30% by weight, more preferably 0.5% to 20% by weight, based on the weight of the N-substituted carbamate.
- catalysts examples include Lewis acids and transition metal compounds that generate Lewis acids, organotin compounds, various compounds of copper group metals, zinc, and iron group metals.
- Specific examples of catalysts include AlX 3 , TiX 3 , TiX 4 , VOX 3 , VX 5 , ZnX 2 , FeX 3 , SnX 4 (where X is an oxygen atom, a nitrogen atom, a halogen atom, an acetoxy group, an alkoxy group , or an aryloxy group with a valence that satisfies the metal oxidation number) and transition metal compounds that generate Lewis acids; (CH 3 ) 3 SnOCOCH 3 , (C 2 H 5 ) SnOCOC 6H5 , Bu3SnOCOCH3 , Ph3SnOCOCH3 , Bu2Sn ( OCOCH3 ) 2 , Bu2Sn ( OCOC11H23 ) 2 , Ph3SnOCH3 , ( C2H
- iron group metal compounds (in the above, Bu represents a butyl group, Ph represents a phenyl group, and acac represents an acetylacetone chelate ligand), 1,4-diazabicyclo[2,2,2]octane, triethylenediamine, amines such as triethylamine; Among them, organometallic catalysts such as dibutyltin dilaurate, lead octylate, and stanaoctoate can be mentioned. These compounds may be used alone or as a mixture of two or more. It is also preferable to carry the above metal component on a carrier that is substantially inert and does not inhibit the reaction. As the carrier, various materials such as activated carbon, silicon, aluminum, zirconium, titanium, oxides and composite oxides thereof are used.
- the shape of the catalyst various shapes such as powder, columnar, ring-shaped, and spherical are preferably used, and columnar, ring-shaped, and spherical are particularly preferable.
- step (1) when a catalyst is used in step (1) described above, the catalyst residue and the like may be supplied to step (2). In the thermal decomposition reaction of step (2), such catalyst residues and the like may be present.
- a reactor that meets the conditions is appropriately selected in the reaction for producing the N-substituted carbamate compound.
- Examples of the reactor include those exemplified in step (1) above.
- a process of recovering the cracked gas and separating it by distillation using the boiling point difference may be added.
- International Publication No. WO 2013/111353 discloses recovering isocyanate and hydroxy compounds produced by a thermal decomposition reaction as gas phase components and separating them by distillation in a multi-stage distillation column.
- the multi-stage distillation column is a plate column
- various plate columns having sieve trays, cascade trays, turbogrid trays, ripple trays, dual trays, etc. can be used as appropriate.
- step (2) it is also a preferred embodiment to control the reaction conditions by appropriately refluxing as necessary.
- the multi-stage distillation column is a packed column
- packing materials generally used for distillation columns and absorption columns can be appropriately used.
- preferred solid fillers include Raschig rings, cascade mini rings, lessing rings, spiral rings, ball rings, interlox saddles, Stedman packings, McMahon packings, Dixon packings, helix packings, coil packings, heat pipe packings, Melapak, Sulzer Packing, Goodroll Packing, Flexipak and the like.
- Internal materials such as fillers are preferably inert to the decomposition products, and are made of SUS304, SUS316, SUS316L, generally silicon oxide (composition formula: SiO 2 ), aluminum oxide (composition formula: Al 2 O 3 ), carbon fluoride (a compound having —CHF— or CF 2 — repeating units) is preferred. Glass, ceramic, and fluororesin containing these as constituent components are also preferable. In the case of glass or ceramic, the contents of silicon oxide and aluminum oxide are not particularly limited, and even if the content of silicon oxide accounts for 60% by mass or more, the content of aluminum oxide accounts for 60% by mass or more. and various materials can be selected.
- Ceramic here refers to a sintered body made by firing inorganic substances.
- inorganic solid materials such as semiconductors such as silicon, and inorganic compounds such as carbides, nitrides, borides, titanium oxide, molded bodies, powders, and films.
- Fluorine resins include polytetrafluoroethylene, polychlorotrifluoroethylene, polyvinylidene fluoride, polyvinyl fluoride, perfluoroalkoxy fluororesin, ethylene tetrafluoride/propylene hexafluoride copolymer, and ethylene/tetrafluoroethylene copolymer. polymers, ethylene/chlorotrifluoroethylene copolymers, and the like.
- the preferred fillers shown here may contain other metal atoms within the scope of the present embodiment.
- Step (3) Isocyanate compound concentration step>
- the isocyanate compound and the aprotic solvent are separated from the reaction solution containing the isocyanate compound obtained in step (2).
- the amount of the reaction solution can be reduced, and the size of equipment for each step can be reduced, thereby improving productivity and reducing equipment costs.
- the step (3) can also be called an isocyanate compound concentration step.
- the method for separating the aprotic solvent is not particularly limited, but from the viewpoint of productivity and ease of process design, known distillation separation is preferred.
- step (3) preferably separates the aprotic solvent from the reaction liquid containing the isocyanate compound under the lowest possible temperature conditions and in the shortest possible time. Therefore, step (3) is also preferably carried out in a continuous manner, like the thermal decomposition reaction in step (2).
- a reaction liquid containing an isocyanate compound is continuously supplied to a reactor, and an aprotic solvent is continuously withdrawn from the reactor. Further, in step (2) described above, it is preferable to use an aprotic solvent having a boiling point lower than that of the isocyanate compound. minutes can be separated more efficiently.
- an aprotic solvent having a boiling point higher than that of the isocyanate compound whereby the aprotic solvent is extracted as the liquid phase component, and the fraction containing the isocyanate compound is more efficiently extracted as the gas phase component.
- the pressure in step (3) varies depending on the type of compound used and the reaction temperature, but may be any of reduced pressure, normal pressure, and increased pressure as long as the isocyanate compound and the aprotic solvent can be separated. .
- the pressure can be a pressure that becomes the saturated vapor pressure of the aprotic solvent used, and is preferably in the range of 20 Pa or more and 1 ⁇ 10 6 Pa or less.
- the operation time (residence time in the case of a continuous method) in step (3) is not particularly limited as long as the isocyanate compound and the aprotic solvent can be separated, but it is preferably 5 seconds or more and 100 hours or less. preferably 10 seconds or more and 50 hours or less, more preferably 20 seconds or more and 10 hours or less, even more preferably 20 seconds or more and 1 hour or less, and 20 seconds or more and 50 minutes or less. 20 seconds or more and 45 minutes or less is particularly preferable, 20 seconds or more and 40 minutes or less is particularly preferable, 30 seconds or more and 30 minutes or less is even more preferable, and 30 seconds or more and 25 minutes or less is particularly preferable. Minutes or less is most preferred.
- the operation time When the operation time is equal to or less than the above upper limit, the operation time can be shortened, and the side reaction of the isocyanate compound can be further suppressed. Moreover, the aprotic solvent can be more sufficiently extracted into the gas phase because the operation time is equal to or longer than the above lower limit.
- the temperature in step (3) is not particularly limited as long as the isocyanate compound and the aprotic solvent are stable and the isocyanate compound and the aprotic solvent can be separated. Since uretdione is generated by the reaction below, it is preferable to separate from the aprotic solvent under as low temperature conditions as possible and in as short a time as possible. From the viewpoint of suppressing modification of the isocyanate compound, the temperature is preferably 300° C. or lower, more preferably 20° C. or higher and 290° C. or lower, and more preferably 30° C. or higher and 280° C. or lower. More preferred.
- distillation apparatus Although there is no particular limitation on the form of separation, it is preferable to use a known distillation apparatus in order to efficiently recover gas phase components.
- distillation apparatus include distillation columns, multi-stage distillation columns, multi-tubular reactors, continuous multi-stage distillation columns, packed columns, thin-film evaporators, reactors with internal supports, forced circulation reactors, Various known methods are used, such as a method using a reactor containing either a falling film evaporator or a falling drop evaporator, or a method combining these.
- a method using a distillation column, a multi-stage distillation column, a continuous multi-stage distillation column, a packed column, a tubular reactor, a thin film evaporator, a falling film evaporator, or a falling drop evaporator from the viewpoint of quickly removing the aprotic solvent from the reaction system. is preferred.
- the aprotic solvent can be rapidly transferred to the gas phase, a separation type having a structure with a large gas-liquid contact area is preferred.
- the solid packing material provided in the packed column may be a packing commonly used for distillation columns and absorption columns, similar to those mentioned in the step (2) described above. materials can be used as appropriate. Specific examples of preferred solid fillers include Raschig rings, Cascade mini rings, Mellapak, Sulzer packing, and Goodroll packing.
- the material of process equipment and lines may be any known material as long as it does not adversely affect isocyanate, etc., but SUS304, SUS316, SUS316L, etc. are preferably used because they are inexpensive.
- step (3) the above-described distillation separation may be performed once, or may be performed two or more times.
- the aprotic solvent can be removed to obtain a fraction containing a more concentrated isocyanate compound.
- the number of distillations can be 1 to 5 times, preferably 1 to 4 times, more preferably 2 to 3 times, and even more preferably 2 times, from the balance between cost and production efficiency.
- distillation it is also a preferred embodiment to control the conditions by appropriately refluxing as necessary.
- Step (4) isocyanate compound isolation step (purification step)>
- steps (4) components having a boiling point higher than that of the isocyanate compound (high boiling point components) are removed from the fraction containing the isocyanate compound obtained in step (3) to purify the isocyanate compound.
- the step (4) can also be called an isocyanate compound isolation step or purification step.
- the method for purifying the isocyanate compound is not particularly limited, a known distillation separation is preferable from the viewpoint of productivity and ease of process design.
- step (4) preferably removes high-boiling components from the fraction containing the isocyanate compound and purifies the isocyanate compound in as short a time as possible under low temperature conditions. Therefore, step (4) is also preferably carried out in a continuous manner, like the thermal decomposition reaction in step (2).
- a fraction containing an isocyanate compound is continuously supplied to a reactor, and the isocyanate compound is continuously withdrawn from the reactor.
- the fraction containing the isocyanate compound contains a high boiling point component having a boiling point higher than that of the isocyanate compound. It preferably contains a carbonyl compound represented by formula (I) (hereinafter sometimes referred to as "carbonyl compound (I)") as a high boiling point component.
- the carbonyl compound (I) is a solvent having a higher boiling point than the isocyanate compound (hereinafter referred to as The isocyanate compound can be extracted from the reactor as a gas phase component and isolated and purified more efficiently.
- the high boiling solvent remains at the bottom of the reactor as a liquid phase component.
- the content of the carbonyl compound (I) is preferably 1 mass ppm or more and 50 mass% or less, more preferably 10 mass ppm or more and 30 mass% or less, relative to the mass of the fraction containing the isocyanate compound, and 0.01 mass%. 15% by mass or less is more preferable, and 1% by mass or more and 10% by mass or less is particularly preferable.
- the content of the carbonyl compound (I) is at least the above lower limit value, the yield of the isocyanate compound can be further improved. By making the equipment for each process smaller, productivity can be improved and equipment costs can be further reduced.
- R 11 is a (n11+n12)-valent organic group
- R 12 is a monovalent organic group.
- n11 is an integer of 1 or more and 8 or less
- n12 is 0 or more and 7 or less. It is an integer, and the sum of n11 and n12 is 2 or more and 8 or less.
- the pressure in step (4) varies depending on the type of compound used and the reaction temperature, but may be any of reduced pressure, normal pressure, and increased pressure as long as the isocyanate compound and the high boiling point component can be separated.
- the pressure can be a pressure at which the isocyanate compound has a saturated vapor pressure, and is preferably in the range of 20 Pa or more and 1 ⁇ 10 6 Pa or less.
- the operation time (residence time in the case of a continuous method) in step (4) is not particularly limited as long as the isocyanate compound and the high boiling point component can be separated, but it is preferably 5 seconds or more and 100 hours or less. , more preferably 10 seconds or more and 50 hours or less, more preferably 20 seconds or more and 10 hours or less, still more preferably 20 seconds or more and 1 hour or less, and 20 seconds or more and 30 minutes or less.
- the operation time is even more preferably, 20 seconds or more and 25 minutes or less is particularly preferable, 20 seconds or more and 20 minutes or less is particularly preferable, 30 seconds or more and 15 minutes or less is even more preferable, 30 seconds or more and 10 minutes or less Most preferably:
- the operation time is equal to or less than the above upper limit, the operation time can be shortened, and the side reaction of the isocyanate compound can be further suppressed.
- an isocyanate compound can be more fully extracted to a gas phase because operation time is more than the said lower limit.
- the temperature in step (4) is not particularly limited as long as the isocyanate compound and the high boiling point component are stable and the isocyanate compound and the high boiling point component can be separated. Since uretdione is generated by the reaction, it is preferable to separate from the aprotic solvent under as low temperature conditions and in as short a time as possible. From the viewpoint of suppressing modification of the isocyanate compound, the temperature is preferably 250° C. or lower, more preferably 20° C. or higher and 240° C. or lower, and preferably 30° C. or higher and 230° C. or lower. More preferred.
- the type of isolation device is not particularly limited, but it is preferable to use a known distillation device in order to efficiently recover the gas phase components.
- distillation apparatus examples include distillation columns, multi-stage distillation columns, multi-tubular reactors, continuous multi-stage distillation columns, packed columns, thin-film evaporators, reactors with internal supports, forced circulation reactors, Various known methods are used, such as a method using a reactor containing either a falling film evaporator or a falling drop evaporator, or a method combining these.
- a method using a distillation column, a multi-stage distillation column, a tubular reactor, a continuous multi-stage distillation column, a packed column, a thin film evaporator, or a falling film evaporator is preferable.
- a method using a tower, a tubular thin film evaporator, a thin film evaporator or a tubular falling film evaporator or the like is more preferred.
- a reactor having a structure with a large gas-liquid contact area is preferred because the isocyanate compound can be rapidly transferred to the gas phase.
- the isolation device and line may be made of any known material as long as it does not adversely affect isocyanate, etc., but SUS304, SUS316, SUS316L, etc. are preferably used because they are inexpensive.
- the isocyanate compound and the aprotic solvent are separated from the component containing the isocyanate compound obtained in the step (3′), and the isocyanate compound is purified like the step (4′), the step (3 ) and the order of the components to be separated in step (4) can be changed.
- the step (3') is the same operation as the step (4), and the step (4') is the same operation as the step (3).
- the manufacturing method of the present embodiment preferably further includes the following step (5) in addition to the steps (1) to (4) described above.
- the fraction containing the aprotic solvent separated in step (3) may contain high boiling point components when the aprotic solvent has a higher boiling point than the isocyanate compound.
- the high-boiling components include the carbonyl compound (I) presumed to be produced by the reaction of an isocyanate compound and an aprotic solvent, biuret bond, allophanate bond, isocyanurate bond, urea bond, carbodiimide bond, uretonimine bond, and iminotrimer bonds.
- the primary amine compound and hydroxy compound used in step (1) are obtained by hydrolyzing the high-boiling components containing these under alkaline conditions. Therefore, step (5) can also be called a primary amine compound and hydroxy compound regeneration step.
- the hydroxy compound may be premixed with the high boiling point component before the hydrolysis reaction. Since hydroxy compounds tend to have high solubility in high-boiling components, by pre-dissolving the high-boiling components in the hydroxy compound, the reaction system can be homogenized and the reaction can proceed rapidly.
- the denaturation of various compounds contained in the high boiling point component can be further suppressed, and the liquid phase state can be maintained.
- the primary amine compound and the hydroxy compound obtained after the hydrolysis reaction are separated, in general, when the primary amine compound and water are mixed, they may be in a miscible state. Phase separation may become difficult. However, by adding the hydroxy compound to the high boiling point component in advance, the phase separation between the primary amine compound and water is facilitated.
- the used alkali may precipitate in the distillation column and the distillation may not be carried out significantly.
- the hydroxy compound by adding the hydroxy compound to the high-boiling point component in advance, it is possible to maintain the dissolved state of the alkali and continuously distill and separate it.
- the liquid phase component can be used as it is in step (5).
- the hydroxy compound may be added by providing a pipe for supplying the hydroxy compound to the pipe for recovering the high-boiling point component continuously extracted as the liquid phase component from the reactor in step (4).
- the content of the hydroxy compound is 20% by mass with respect to the total mass of the fraction containing the aprotic solvent separated in step (3) and the component having a higher boiling point than the isocyanate compound removed in step (4). 300 mass % or less is preferable, and 50 mass % or more and 150 mass % or less is more preferable.
- the reaction system in the step (5) can be homogenized and the reaction can proceed rapidly.
- the high boiling point component is preferably kept at 50° C. or higher and 300° C. or lower, more preferably 100° C. or higher and 260° C. or lower, and supplied to the reactor in step (5).
- the amount (molar amount) of water used is stoichiometric with respect to the total molar amount of isocyanate groups, biuret bonds, allophanate bonds, isocyanurate bonds, carbamate bonds, and urea bonds contained in the high boiling point component. It is preferably from 1.0 times to 200 times, more preferably from 5 times to 150 times, and even more preferably from 5 times to 100 times.
- the energy load in the subsequent dehydration step can be further reduced, and it is possible to reduce energy consumption in production and downsize the apparatus.
- the amount of water used is at least the above lower limit, the decomposition reaction of the high-boiling-point component proceeds more favorably.
- the amount of water used varies depending on the composition of the material to be decomposed because the hydrolyzability varies depending on the composition of the bonds contained in the high-boiling point components, but if it is within the above range, efficient decomposition is possible. is.
- the amount of water can be further reduced.
- crosslinkable bonds typified by urea bonds that can be produced as intermediates in the hydrolysis reaction increase, and high molecular weight substances, ie, gels, may be generated in the system.
- the gel becomes a scale and adheres to the inside of the apparatus, which not only significantly lowers the reaction efficiency, but also causes a decrease in economic efficiency due to the need to clean the apparatus.
- the amount of water to be used within the above range the decomposition proceeds more favorably and the generation of scale in the apparatus can be further suppressed, so that the primary amine compound can be efficiently recovered.
- the alkali is not particularly limited, but for example, hydroxides and oxides of alkali metals and alkaline earth metals such as sodium hydroxide, potassium hydroxide, magnesium hydroxide, magnesium oxide, calcium oxide and barium oxide; Heterocyclic compounds such as methylpyridine (including isomers), dipropylethylamine, N-methylmorpholine, N-ethylmorpholine, triethylamine and triethylenediamine, and tertiary amines. These alkalis can also be used individually or in mixture of 2 or more types. Among them, as the alkali, hydroxides and oxides of alkali metals and alkaline earth metals are preferable, and hydroxides of alkali metals such as sodium hydroxide and potassium hydroxide are more preferable.
- the alkali may be uniformly compatible with the reaction liquid, or may be non-uniform. It is preferable to use a uniform alkali, which can promote the decomposition reaction in the reactor quickly, contribute to the suppression of the production of by-products, and further improve the yield.
- the amount (molar amount) of alkali used is determined by isocyanate group, biuret bond, allophanate bond, isocyanurate bond, carbamate bond, urea bond, carbodiimide bond, uretonimine bond, imino trimer bond, and Fries rearrangement end contained in the high boiling point component.
- Fries rearrangement end cyclized product, and oxycarbonyl carbamate bond the stoichiometric ratio can be 0.001 to 10 times, preferably 0.01 to 1 time. , more preferably 0.03 times or more and 0.5 times or less.
- the yield of the primary amine compound decreases because the decomposition of the high-boiling point component is slow or not decomposed.
- the bonds contained in the high-boiling-point component have different decomposability depending on their bonding modes, and by appropriately selecting the type of alkali, the reaction mode, etc., it is possible to accelerate the decomposition of each bonding mode.
- Fries rearrangement ends, Fries rearrangement end cyclizations, isocyanurate linkages, and iminotrimer linkages are
- the decomposition reaction with water is difficult to proceed, and it is difficult to generate primary amine compounds.
- isocyanurate bonds and iminotrimer bonds are difficult to hydrolyze, and are known to undergo slow decomposition by known methods. These bonds are crosslinkable bonds, and if they remain in the reaction solution as high-molecular-weight substances, they may clog the apparatus due to precipitation or the like when the primary amine compound or hydroxy compound is separated from the reaction solution, or , the amount of primary amine compounds that can be recovered is reduced.
- the reaction temperature for hydrolysis in step (5) can be appropriately set according to the composition of the high-boiling component to be decomposed. It is preferably 160° C. or higher and 300° C. or lower, and even more preferably 200° C. or higher and 280° C. or lower. In the temperature range above the upper limit of the reaction temperature, the decomposition efficiency is almost saturated, and rather, the environment of high-temperature, high-pressure water becomes a reaction field that increases the ion product and improves the acidity. As a result, unfavorable reactions may occur, for example, the formation of imine compounds, which are internally cyclized by-products of deammonification of primary amine compounds.
- the decomposition efficiency decreases, and crosslinkable bonds typified by urea bonds that can be produced as intermediates in the hydrolysis reaction increase, and high molecular weight, In other words, gel may occur.
- the gel becomes a scale and adheres to the inside of the apparatus, which not only significantly lowers the reaction efficiency, but also causes a decrease in economic efficiency due to the need to clean the apparatus.
- the reaction temperature is within the above range, the decomposition of the crosslinkable molecule represented by the urea bond proceeds more favorably, and the generation of scale in the apparatus can be further suppressed. Primary amine compounds can be recovered more efficiently.
- the reaction pressure can be 0.01 kPa or more and 10 MPa (absolute pressure) or less, and can be carried out under reduced pressure, normal pressure, or increased pressure.
- the reaction time (residence time in the case of a continuous reaction) can be 0.01 hours or more and 100 hours or less. , the reaction can be terminated when the desired production amount is reached.
- a low boiling point component having a boiling point of 30° C. or lower may be generated. may be delayed. Therefore, it is preferable to carry out the hydrolysis reaction while withdrawing at least part of the low-boiling-point component out of the reaction system as a gas phase component. In this case, it is preferable to prevent water from being drawn out of the reaction system together with the low boiling point components, such as by installing a condenser in the middle of the line for drawing out the low boiling point components from the reactor in which the hydrolysis reaction is performed.
- an isocyanate group may be generated by the reaction with, and it is preferable to withdraw the generated carbon dioxide out of the reaction system.
- the generated carbon dioxide forms a carbonate with the primary amine compound produced by the hydrolysis reaction, which may slow down the hydrolysis reaction.
- the facilitation of carbonate formation is dependent on the nucleophilicity of the primary amine compound, and the order of carbonate formation is generally aliphatic amines > substituted cycloaliphatic polyamine > aromatic Since it is an amine, it is preferable to adjust the amount of carbon dioxide produced by the structure of the primary amine compound to be extracted out of the reaction system as necessary.
- the reactor for the hydrolysis reaction in step (5) is not particularly limited, and known reactors can be used.
- a vertical thin film evaporator equipped with a forced conveying device, a tubular reactor, etc. can be used in combination according to the reaction method and conditions.
- the reaction may be of a batch type or a continuous flow type, and a reaction apparatus may be selected according to each reaction type.
- the material of the reactor is also not particularly limited, and known materials can be used.
- SUS304, SUS316, SUS316L, etc. are inexpensive and can be preferably used.
- known process devices such as instrumentation equipment such as a flow meter and thermometer, a mechanism for maintaining pressure, a reboiler, a pump, a condenser and the like may be added.
- a known method such as steam or a heater may be used for heating, and a known method such as natural cooling, cooling water, or brine may be used for cooling.
- the primary amine compound can be recovered from the reaction liquid obtained by the hydrolysis reaction through known separation methods such as distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- the recovered primary amine compound is preferably reused as a raw material in step (1) described above.
- the method for recovering the primary amine compound and the hydroxy compound preferably includes steps (5a) to (5d) shown below, and these steps can be performed continuously, or steps (5a) to ( 5d) can also be performed simultaneously. Further, for example, the operations of steps (5c) to (5d) can be performed before the operations of steps (5a) to (5b), or the steps can be partially exchanged.
- the primary amine compound is separated from the reaction solution after the hydrolysis reaction.
- a known method can be used for separating the primary amine compound, and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- the primary amine compound can also be continuously separated from the liquid phase component (reaction solution) continuously discharged in step (5).
- Step (5b): Purification of primary amine compound In step (5b), the primary amine compound separated in step (5a) is purified.
- a known method can be used to purify the primary amine compound, and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- the primary amine compound continuously separated in step (5a) can also be purified continuously.
- Step (5c): Separation of hydroxy compound the hydroxy compound used as the raw material in step (1) is separated from the reaction solution after the hydrolysis reaction.
- a known method can be used to separate the hydroxy compound, and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- the hydroxy compound can also be continuously separated from the gas phase component continuously discharged in step (5).
- Step (5d): Purification of hydroxy compound In step (5d), the hydroxy compound separated in step (5c) above is purified. A known method can be used to purify the hydroxy compound, and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation. In step (5d), the hydroxy compound continuously separated in step (5c) above can also be purified continuously.
- the primary amine compound and hydroxy compound recovered by the recovery method can be reused in step (1) above. That is, the manufacturing method of the present embodiment preferably further includes step (7) shown below. A step (7) of reusing the primary amine compound and hydroxy compound recovered in the step (5) in the step (1).
- Step (7) Step of Reusing Primary Amine Compound and Hydroxy Compound
- the primary amine compound and hydroxy compound recovered in step (5) are reused in step (1).
- the production of the N-substituted carbamate compound in the above step (1) the quality of the obtained N-substituted carbamate compound and the isocyanate compound obtained in the subsequent step may be affected, so the above step ( 5) (Specifically, in the above steps (5b) and (5d)), the content of metal components is 1000 mass ppm or less and the content of halogen atoms is 1000 mass ppm or less with respect to the total mass of the primary amine compound. It is preferable to collect the primary amine compound and the hydroxy compound by distillation so that the is 1000 ppm by mass or less.
- the hydroxy compound can be reused for pre-mixing with the high boiling point component in step (5) above.
- the manufacturing method of the present embodiment can further include other steps in addition to the steps (1) to (5) described above.
- Step (1a): N-substituted carbamate compound concentration step In step (1a), the N-substituted carbamate compound obtained in step (1) is concentrated. That is, step (1a) can also be called an N-substituted carbamate compound concentration step.
- a method for concentrating the N-substituted carbamate compound is not particularly limited, but a known distillation separation is preferable from the viewpoint of productivity and ease of process design.
- step (1a) removes unreacted raw material components such as hydroxy compounds from the reaction liquid containing the N-substituted carbamate compound in as short a time as possible under low temperature conditions to obtain N It is preferred to concentrate the -substituted carbamate compound. Therefore, step (1a) is also preferably carried out in a continuous manner, like the reaction in step (1).
- a concentration method by a continuous method a reaction liquid containing an N-substituted carbamate compound is continuously supplied to a reactor, and the concentrated N-substituted carbamate compound is continuously extracted from the reactor.
- the speed of supplying the reaction solution to the reactor and the speed of withdrawing the N-substituted carbamate compound can be appropriately adjusted according to the degree of concentration of the N-substituted carbamate compound.
- step (1a) the concentration described above may be performed after premixing the aprotic solvent used in the subsequent step (1).
- the aprotic solvent used at this time it is preferable to use one having a boiling point higher than that of the hydroxy compound. This makes it possible to further prevent the liquid from drying up due to concentration.
- Step (6) solvent recovery step (solvent reuse step)
- step (6) the hydroxy compound extracted in the step (2) is recycled to the step (1) for reuse, and the aprotic solvent separated in the step (3) is recycled to the It is recycled and reused in step (2). That is, step (6) can be referred to as a solvent recovery step or a solvent recycling step.
- the hydroxy compound extracted in step (2) and the aprotic solvent separated in step (3) may be separated and purified separately to recover the hydroxy compound and the aprotic solvent, respectively.
- the components withdrawn from these steps may be mixed together and then separated and purified to recover the hydroxy compound and the aprotic solvent.
- a known method can be used for separating and purifying the hydroxy compound and the aprotic solvent, and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- distillation separation for example, a system including any one of a multistage distillation column, a plate column, a packed column, a thin film evaporator, a falling film evaporator, a drop evaporator, and a falling film type evaporator is used.
- step (6) the hydroxy compound and the aprotic solvent are continuously separated from the gas phase component continuously discharged in step (2) and the gas phase component continuously discharged in step (3), respectively. You can also Moreover, it is also one of the preferable aspects to control conditions by carrying out reflux suitably as needed.
- the recovered hydroxy compound can be reused for pre-mixing with the high boiling point component in step (5) above, or can be reused as a raw material for step (1).
- Primary amine compound examples include compounds represented by the following general formula (II) (hereinafter sometimes referred to as "primary amine compound (II)").
- R 21 is an n21-valent organic group. n21 is an integer of 2 or more.
- R21 is an n21-valent organic group, preferably an organic group having 3 or more and 85 or less carbon atoms, more preferably an organic group having 3 or more and 30 or less carbon atoms.
- the organic group for R 21 is an aliphatic hydrocarbon group, an aromatic hydrocarbon group, or a group formed by combining an aliphatic hydrocarbon group and an aromatic hydrocarbon group.
- R 21 include, for example, a cyclic hydrocarbon group, an acyclic hydrocarbon group, a group in which an acyclic hydrocarbon group and one or more cyclic groups are bonded, and these groups are specified and a group covalently bonded to a non-metallic atom of.
- the cyclic group include cyclic hydrocarbon groups, heterocyclic groups, heterocyclic spiro groups, and hetero bridged ring groups.
- Examples of the cyclic hydrocarbon group include monocyclic hydrocarbon groups, condensed polycyclic hydrocarbon groups, bridged cyclic hydrocarbon groups, spiro hydrocarbon groups, ring-assembled hydrocarbon groups, and cyclic hydrocarbon groups with side chains.
- a hydrocarbon group etc. are mentioned.
- Examples of the nonmetallic atoms include carbon, oxygen, nitrogen, sulfur, silicon and the like.
- covalently bonded to a specific non-metallic atom means, for example, that the groups exemplified above are covalently bonded to groups represented by the following formulas (II)-1a to (II)-1p. state.
- the organic group for R 21 is a cyclic hydrocarbon group, an acyclic hydrocarbon group, a group in which an acyclic hydrocarbon group and one or more cyclic groups are bonded, or these groups is a group covalently bonded to a group represented by formula (II)-1a, formula (II)-1b, formula (II)-1c, formula (II)-1e, or formula (II)-1j Preferably.
- R 21 is an aliphatic hydrocarbon group
- specific examples of the primary amine compound (II) include aliphatic diamines, aliphatic triamines, and substituted cycloaliphatic polyamines. be done.
- aliphatic diamines examples include ethylenediamine, diaminopropane (each isomer), diaminobutane (each isomer), diaminopentane (each isomer), diaminohexane (each isomer), and diaminodecane (each isomer). etc.
- aliphatic triamines examples include triaminohexane (each isomer), triaminononane (each isomer), triaminodecane (each isomer), triaminoundecane (each isomer), 4-aminomethyl- 1,8-octanediamine and the like.
- Substituted cycloaliphatic polyamines include, for example, diaminocyclobutane (each isomer), diaminocyclohexane (each isomer), 3-aminomethyl-3,5,5-trimethylcyclohexylamine (cis and trans at least one of the isomers), methylenebis(cyclohexylamine) (each isomer), and the like.
- R 21 is an aromatic group
- specific examples of the primary amine compound (II) include aromatic diamines and aromatic triamines.
- aromatic diamines examples include diaminobenzene (each isomer), diaminotoluene (each isomer), methylenedianiline (each isomer), diaminomesitylene (each isomer), diaminobiphenyl (each isomer), Diaminodibenzyl (each isomer), bis(aminophenyl)propane (each isomer), bis(aminophenyl)ether (each isomer), bis(aminophenoxyethane) (each isomer), diaminoxylene (each isomer) isomer), diaminoanisole (each isomer), diaminophenetole (each isomer), diaminonaphthalene (each isomer), diaminomethylbenzene (each isomer), diaminomethylpyridine (each isomer), diaminomethylnaphthalene ( each isomer), diaminodip
- aromatic triamines examples include triaminobenzene (each isomer), triaminomethylbenzene (each isomer), tris(aminopropan-yl)benzene (each isomer), tris(aminopropan-yl)- Examples include methylbenzene (each isomer), tris(aminomethyl)-methylbenzene (each isomer), ((aminophenylene)bis(methylene))bis(aminebenzene) (each isomer), and the like.
- the primary amine compound (II) specifically includes, for example, a reaction between the carboxy group of an amino acid and a hydroxy compound.
- An amine compound having an ester group obtained by Specific examples of such amine compounds having an ester group include 2-aminoethyl acrylate, 2-methyl-2-aminoethyl acrylate, 2-aminopropyl acrylate, 2- Methyl-Acrylic Acid-2-Aminopropyl Ester, Acrylic Acid-3-Aminopropyl Ester, 2-Methyl-Acrylic Acid-3-Aminopropyl Ester, Acrylic Acid-4-Aminobutyl Ester, 2-Methyl-Acrylic Acid-4 -aminobutyl ester, 5-aminopentyl acrylate, 2-methyl-5-aminopentyl acrylate, 6-aminohexyl acrylate, 2-methyl
- the primary amine compound (II) specifically includes, for example, an aliphatic hydrocarbon group or an aromatic group is preferably an amine compound having 1 to 4 secondary or tertiary amines in addition to the terminal of .
- amine compound having 1 to 4 secondary or tertiary amines other than the end of the aliphatic hydrocarbon group or aromatic group herein include, for example, 2-(dimethylamino)ethyleneamine, 2-( Diethylamino)ethyleneamine, 2-(diisopropylamino)ethyleneamine, 2-(cyclohexylamino)ethyleneamine, 3-(cyclohexylamino)propylamine, 3-(diethylamino)propylamine, 3-(dimethylamino)propylamine, diethylenetriamine , diisopropyltriamine, bis-(3-aminopropyl)methyleneamine, 3-(2-aminoethylamino)propylamine, N,N'-bis(3-aminopropyl)ethylenediamine, 1-(3-aminopropyl)imidazole , trisaminoethylamine, trisaminopropylamine, and the like.
- n21 represents the number of amino groups, and is an integer of 2 or more, preferably an integer of 2 or more and 10 or less, more preferably an integer of 3 or more, and still more preferably 3.
- the primary amine compound (II) includes diaminohexane (each isomer), 4-aminomethyl-1,8-octanediamine, triaminohexane (each isomer), triaminoundecane (each isomer isomer), 3-aminomethyl-3,5,5-trimethylcyclohexylamine, methylenebis(cyclohexylamine) (each isomer), diaminoxylene (each isomer), methylenedianiline (each isomer), triaminobenzene ( each isomer), triaminomethylbenzene (each isomer), or tris(aminopropan-yl)-methylbenzene (each isomer).
- Amino acid derivatives are also preferably used as the primary amine compound (II).
- amino acid derivative as used herein means a compound synthesized using an amino acid as a raw material. As will be described later, the amino acid may be a natural amino acid or a synthetic amino acid. Further, the derivative may be a derivative alone or in the form of an inorganic acid salt.
- Preferred amino acid derivatives include, for example, compounds represented by the following general formula (II-1) or (II-2) (hereinafter sometimes abbreviated as "amino acid derivative (II-1)", etc.). be done.
- R 211 is an n211-valent organic group and is the same as R 21 above.
- R 211 preferably has a structure obtained by removing the —NHCOOH group from an amino acid, and is an aliphatic hydrocarbon having 1 or more carbon atoms, which may contain a primary amino group, a sulfur atom, an oxygen atom, or a halogen atom. or an aromatic group having 6 or more carbon atoms, a divalent or trivalent aliphatic hydrocarbon group having 1 or more and 15 or less carbon atoms or a bivalent or trivalent hydrocarbon group having 6 or more and 15 or less carbon atoms More preferably, it is an aromatic group.
- X 211 is an oxygen atom or a secondary amino group (-NH-). Among them, X 211 is preferably an oxygen atom.
- R 212 is a monovalent aliphatic hydrocarbon group having 1 to 15 carbon atoms, an aromatic group having 6 to 15 carbon atoms, or a hydrogen atom. Among them, R 212 is preferably an alkyl group having 1 to 6 carbon atoms, more preferably an alkyl group having 1 to 3 carbon atoms.
- n211 is an integer of 2 or more, and n211 is preferably 2 or 3.
- amino acid derivative may be D-type, L-type, a mixture thereof, or a racemate.
- amino acids that are industrially available at low cost are amino acids produced by fermentation, and most of them are L-type, and they can be preferably used. Configurations are not indicated herein, but are indicated as either D- or L-forms.
- R 221 is an n221-valent organic group or a hydrogen atom, and when it is an n221-valent organic group, it is the same as R 21 above.
- R 221 is preferably a monovalent to tetravalent aliphatic hydrocarbon group having 1 to 15 carbon atoms, or a monovalent to tetravalent aromatic group having 6 to 15 carbon atoms.
- X 221 is an oxygen atom or a secondary amino group (--NH--), preferably an oxygen atom.
- n221 is an integer of 1 or more and 4 or less.
- R 222 is a monovalent aliphatic hydrocarbon group, monovalent aromatic group or hydrogen atom.
- R 222 is a monovalent aliphatic hydrocarbon group or carbon having 1 or more carbon atoms, which may contain one or more selected from the group consisting of a primary amino group, a sulfur atom, an oxygen atom, and a halogen atom. It is preferably a monovalent aromatic group of number 6 or more, or a hydrogen atom.
- a monovalent aliphatic hydrocarbon group having 1 to 15 carbon atoms or 6 carbon atoms which may contain one or more selected from the group consisting of a primary amino group, a sulfide bond, an ether bond, and a disulfide bond;
- One or more selected from the group consisting of a monovalent aromatic group of 15 or less, a primary amino group in which an aliphatic hydrocarbon group and an aromatic group are bonded, a sulfide bond, an ether bond, and a disulfide bond It is more preferably a group having 7 or more and 15 or less carbon atoms, a group represented by the following general formula (II-2a) or (II-2b), or a hydrogen atom, which may contain.
- R 223 is a group represented by the following general formula (II-2c), (II-2d), or (II-2e), or a hydrocarbon having 1 to 10 carbon atoms is the base.
- n222 is an integer of 0 or more and 5 or less.
- a wavy line represents a bond.
- R 224 is an aliphatic hydrocarbon group having 1 to 15 carbon atoms or an aromatic group having 6 to 15 carbon atoms.
- n223 is an integer of 0 or more and 5 or less.
- a wavy line represents a bond.
- R 225 is a divalent aliphatic hydrocarbon group having 1 to 10 carbon atoms, preferably a linear or branched alkylene group having 1 to 6 carbon atoms.
- n223 is an integer of 1 or more and 9 or less.
- n224 is an integer of 0 or more and 9 or less.
- wavy lines represent bonds.
- amino acid esters are preferred as amino acid derivatives. Accordingly, preferred amino acid derivatives (II-1) and (II-2) are exemplified below.
- Preferable amino acid derivatives (II-1) specifically include compounds represented by the following general formulas (II-1-1) to (II-1-3).
- R 213 is the same as R 212 above. Among them, R 213 is preferably an alkyl group having 1 to 6 carbon atoms, more preferably an alkyl group having 1 to 3 carbon atoms.
- Preferred amino acid derivatives (II-2) include, for example, compounds represented by general formulas (II-2-1) to (II-2-4).
- R 226 is a monovalent aliphatic hydrocarbon group having 1 to 15 carbon atoms, a monovalent aromatic group having 6 to 15 carbon atoms groups, or hydrogen atoms.
- R 227 is the same as R 222 .
- amino acid derivative a compound represented by the following general formula (II-3) (hereinafter sometimes referred to as "amino acid derivative (II-3)”) is also preferably used.
- n231 is 1 or 2 in general formula (II-3).
- R 231 is an aliphatic hydrocarbon group having 1 to 15 carbon atoms, which may contain an ether bond or a sulfide bond, or an aromatic group having 6 to 15 carbon atoms, or an aliphatic hydrocarbon.
- a group having 7 or more and 15 or less carbon atoms, which may contain an ether bond or a sulfide bond, in which a group and an aromatic group are bonded, or general formula (II-2c), (II-2d), or (II-2e) is a group represented by
- R 232 is one having 1 to 15 carbon atoms, which may contain one or more selected from the group consisting of a primary amino group, a sulfide bond, an ether bond, and a disulfide bond.
- Preferred amino acid derivatives (II-3) include compounds represented by the following general formulas (II-3-1) to (II-3-39).
- R 3 and R 4 are each independently an aliphatic hydrocarbon group having 1 to 10 carbon atoms or a hydrogen atom. Among them, R 3 is preferably an alkyl group having 1 to 4 carbon atoms. R 4 is preferably an alkyl group having 1 to 6 carbon atoms.
- R is a halogen atom, an alkyl group having 1 to 6 carbon atoms, or an alkoxy group having 1 to 6 carbon atoms.
- R is preferably a halogen atom, an alkyl group having 1 to 4 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms.
- n is an integer of 0 or more and 2 or less. Among them, n is preferably 1 or 2.
- m and p are each independently an integer of 1 or more and 10 or less, preferably an integer of 1 or more and 6 or less, and 1 or more and 3 The following integers are more preferable.
- amino acid derivative (II-3) includes the general formulas (II-3-3), (II-3-4), (II-3-13), (II-3-14), ( Compounds represented by II-3-38) or (II-3-39) are preferred.
- amino acid derivatives (II-3) include amino acid esters derived from lysine bone nuclei such as lysine methyl ester, lysine ethyl ester and lysine ⁇ -aminoethyl ester; ) Amino acid esters derived from glutamic acid skeleton such as ester; Amino acid esters derived from methionine skeleton such as methionine methyl ester; Amino acid esters derived from glycine skeleton such as glycine methyl ester; Amino acids derived from phenylalanine skeleton such as phenylalanine methyl ester Ester; amino acid ester derived from aspartic acid skeleton such as aspartic acid methyl ester; amino acid ester derived from alanine skeleton such as alanine methyl ester; amino acid ester derived from leucine skeleton such as leucine methyl ester; isoleucine
- the above-mentioned amino acid ester is produced by, for example, reacting an amino acid with a compound having an alcoholic hydroxy group in the presence of an inorganic acid, or reacting an amino acid inorganic acid salt and an amino alcohol inorganic acid salt in the presence of an inorganic acid. It can be manufactured by
- the inorganic acid may be any inorganic acid such as hydrochloric acid, nitric acid, phosphoric acid, sulfuric acid, boric acid and hydrofluoric acid, preferably sulfuric acid, phosphoric acid and hydrochloric acid, more preferably hydrochloric acid. be.
- Amino acid esters may be used in the form of inorganic acid salts.
- the amino acid ester inorganic acid salt is formed from the above inorganic acid, and is preferably an amino acid ester sulfate, an amino acid ester phosphate, or an amino acid ester hydrochloride, more preferably an amino acid ester hydrochloride.
- the amino acid is preferably an aliphatic or aromatic amino acid having 2 or more and 18 or less carbon atoms, or a 3- or 12-membered lactam having at least one amino group and at least one carboxy group. .
- the amino acid may be a natural amino acid or a synthetic amino acid.
- Natural amino acids include alanine, arginine, asparagine, glutamine, glutamic acid, glycine, histidine, isoleucine, leucine, lysine, aspartic acid, methionine, phenylalanine, tryptophan, valine, ornithine, and the like.
- Synthetic amino acids can be produced by known methods, for example, by Strecker synthesis using aldehyde compounds.
- aldehydes include compounds represented by the following general formula (A) (hereinafter sometimes abbreviated as "compound (A)").
- R a is a monovalent aliphatic hydrocarbon group having 1 or more carbon atoms or an aromatic group having 6 or more carbon atoms, which may contain an oxygen atom or a halogen atom.
- R a is preferably a monovalent aliphatic hydrocarbon group having 1 to 12 carbon atoms or an aromatic group having 6 or more carbon atoms.
- a is an integer of 1 or more and 3 or less.
- Preferred compounds (A) include, for example, acetaldehyde, propionaldehyde, hexylaldehyde, octylaldehyde, caprinsanaldehyde, phenylacetaldehyde, benzaldehyde, dimethoxybenzaldehyde, chlorobenzaldehyde, fluorobenzaldehyde, heliotropine, cyclamenbenzaldehyde, furfural, naphthaldehyde, phthalaldehyde and the like.
- isomers are also included.
- Amino acid inorganic acid salts are inorganic acid salts of the above amino acids.
- Particularly preferably used amino acids are aliphatic monoaminomonocarboxylic acids, diaminomonocarboxylic acids, monoaminodicarboxylic acids, diaminodicarboxylic acids and the like. Lactams formed by cyclization of these amino acids are also preferably used.
- the above compounds include glycine, 3-aminopropionic acid, ⁇ -aminocaproic acid, ⁇ -aminolauric acid, alanine, isoleucine, 3-aminobutyric acid, 4-aminocyclohexanecarboxylic acid, phenylalanine, methionine, and aminobenzoic acid. , aspartic acid, glutamic acid, lysine, lanthionine, 1-amino-2,3,4-butanetricarboxylic acid, lactams of the above amino acids, pyrrolidone, caprolactam, laurolactam and the like.
- the compound containing an alcoholic hydroxy group is preferably an aminoalcohol inorganic acid salt.
- the aminoalcohol inorganic acid salt can be produced by reacting the amino acid inorganic acid salt with alcohol.
- An aminoalcohol inorganic acid salt is an inorganic acid salt of an aminoalcohol having 2 or more and 12 or less carbon atoms having one primary or secondary hydroxyl group and one primary amino group.
- the above aminoalcohols include those containing heteroatoms such as oxygen and sulfur in the alkylene chain, and groups inert to esterification reactions, such as nitro, halogen, alkyl, and substituents such as phenyl groups. may be
- amino alcohols include ethanolamine, 1-amino-2-propanol, 2-amino-1-propanol, 2-aminoisobutanol, 2-amino-1-butanol, 2-(2-aminoethoxy)- ethanol, 2-aminocyclohexanol, and the like.
- a known alcohol can be used, but a monoalcohol having 1 to 10 carbon atoms is preferable. Specific examples include methanol, ethanol, propanol, butanol, pentanol, hexanol, octanol, decanol, cyclopentanol, cyclohexanol and the like. In addition, when these compounds contain isomers, the isomers can also be used.
- Hydroxy compounds include alcohols and aromatic hydroxy compounds.
- alcohols examples include methyl alcohol, propyl alcohol, butyl alcohol, amyl alcohol, hexyl alcohol, heptyl alcohol, octyl alcohol, nonyl alcohol, decyl alcohol, undecyl alcohol, lauryl alcohol, dothesyl alcohol, stearyl alcohol, and eicosyl alcohol.
- alcohol allyl alcohol, crotyl alcohol, propargyl alcohol, cyclopentanol, cyclohexanol, benzyl alcohol, cinnamyl alcohol, ethylene glycol, diethylene glycol, polyethylene glycol, propylene glycol, polypropylene glycol, 1,3-butanediol, 1, 4-butanediol, hydrogenated bisphenol A, neopentyl glycol, glycerin, trimethylolpropane, pentaerythritol, ethanolamine, propanolamine (1-amino-2-propanol), dimethanolamine, diethanolamine, dipropanolamine, 1- Amino-2-butanol and the like can be mentioned.
- aromatic hydroxy compounds include compounds represented by general formula (VI) (hereinafter sometimes referred to as "aromatic hydroxy compounds (VI)").
- ring A 61 is an aromatic hydrocarbon ring having 6 to 20 carbon atoms.
- R 61 is a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, and 1 to 20 carbon atoms.
- R 61 may combine with Ring A 61 to form a ring structure, and n61 is an integer of 1 or more and 10 or less.
- Examples of the alkyl group having 1 to 20 carbon atoms in R 61 include methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), pentyl group (each isomer), hexyl group ( isomer), heptyl group (each isomer), octyl group (each isomer), nonyl group (each isomer), decyl group (each isomer), dodecyl group (each isomer), octadecyl group (each isomer) body) and the like.
- the alkoxy group having 1 to 20 carbon atoms in R 61 includes, for example, methoxy group, ethoxy group, propoxy group (each isomer), butyloxy group (each isomer), pentyloxy group (each isomer), hexyloxy group (each isomer), heptyloxy group (each isomer), octyloxy group (each isomer), nonyloxy group (each isomer), decyloxy group (each isomer), dodecyloxy group (each isomer), octadecyloxy group (each isomer) and the like.
- Examples of the aryl group having 6 to 20 carbon atoms in R 61 include a phenyl group and a naphthyl group.
- Examples of the aryl group having an alkyl group as a substituent for R 61 include a methylphenyl group (each isomer), an ethylphenyl group (each isomer), a propylphenyl group (each isomer), a butylphenyl group (each isomer isomer), pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer), nonylphenyl group (each isomer), decylphenyl group (each isomer), biphenyl group (each isomer), dimethylphenyl group (each isomer), diethylphenyl group (each isomer), dipropylphenyl group (each isomer), dibutylphenyl group (each isomer ),
- Examples of the aryloxy group having 6 to 20 carbon atoms in R 61 include a phenoxy group, a methylphenoxy group (each isomer), an ethylphenoxy group (each isomer), a propylphenoxy group (each isomer), and a butylphenoxy group.
- each isomer pentylphenoxy group (each isomer), hexylphenoxy group (each isomer), heptylphenoxy group (each isomer), octylphenoxy group (each isomer), nonylphenoxy group (each isomer ), decylphenoxy group (each isomer), phenylphenoxy group (each isomer), dimethylphenoxy group (each isomer), diethylphenoxy group (each isomer), dipropylphenoxy group (each isomer), dibutylphenoxy group (each isomer), dipentylphenoxy group (each isomer), dihexylphenoxy group (each isomer), diheptylphenoxy group (each isomer), diphenylphenoxy group (each isomer), trimethylphenoxy group (each isomer isomer), triethylphenoxy group (each isomer), tripropy
- Examples of the aralkyl group having 7 to 20 carbon atoms in R 61 include a phenylmethyl group, a phenylethyl group (each isomer), a phenylpropyl group (each isomer), a phenylbutyl group (each isomer), and phenylpentyl. group (each isomer), phenylhexyl group (each isomer), phenylheptyl group (each isomer), phenyloctyl group (each isomer), phenylnonyl group (each isomer), and the like.
- Examples of the aralkyloxy group having 7 or more and 20 or less carbon atoms in R 61 include a phenylmethoxy group, a phenylethoxy group (each isomer), a phenylpropyloxy group (each isomer), and a phenylbutyloxy group (each isomer). , phenylpentyloxy group (each isomer), phenylhexyloxy group (each isomer), phenylheptyloxy group (each isomer), phenyloctyloxy group (each isomer), phenylnonyloxy group (each isomer) etc.
- Ring A 61 is an aromatic hydrocarbon ring having 6 or more and 20 or less carbon atoms. Ring A 61 may be monocyclic, polycyclic, or condensed. Specific examples of ring A 61 include benzene ring, naphthalene ring, anthracene ring, phenanthrene ring, naphthacene ring, chrysene ring, pyrene ring, triphenylene ring, pentalene ring, azulene ring, heptalene ring, indacene ring, biphenylene ring, acenaphthylene ring, aceanthrylene ring, acephenanthrylene ring, and the like.
- the ring A 11 is preferably a benzene ring, a naphthalene ring, or an anthracene ring, more preferably a benzene ring.
- R 61 may also have substituents other than R 61 above.
- Substituents other than R 61 include the same as those exemplified for R 61 .
- R 61 and substituents other than R 61 consist of different functional groups.
- n61 represents the number of substituents R61 and is an integer of 1 or more and 10 or less.
- examples of compounds in which ring A 61 is a benzene ring include compounds represented by the following general formula (VI-1).
- R 611 to R 615 are each independently the same as R 61 above.
- At least one of R 611 to R 615 is preferably a hydrogen atom, and more preferably all of R 611 to R 615 are hydrogen atoms.
- Preferred hydroxy compounds include, for example, phenol, 2-ethylphenol, 2-propylphenol (each isomer), 2-butylphenol (each isomer), 2-pentylphenol (each isomer), 2-hexylphenol (each isomer), 2-heptylphenol (each isomer), 2-phenylphenol, 2,6-dimethylphenol, 2,4-diethylphenol, 2,6-diethylphenol, 2,4-dipropylphenol (each isomer isomer), 2,6-dipropylphenol (each isomer), 2,4-dibutylphenol (each isomer), 2,4-dipentylphenol (each isomer), 2,4-dihexylphenol (each isomer ), 2,4-diheptylphenol (each isomer), 2-methyl-6-ethylphenol, 2-methyl-6-propylphenol (each isomer), 2-methyl-6-butylphenol (each iso
- the hydroxy compound is preferably a hydroxy compound having a normal boiling point lower than that of the isocyanate compound produced from the N-substituted carbamate compound described above.
- Carbonic acid derivatives examples include urea, urea derivatives, carbonate esters, and the like. The details of the carbonate ester will be described later.
- the urea derivative includes, for example, a compound represented by the general formula (IV) obtained by the following reaction formula (hereinafter sometimes referred to as "urea derivative (IV)").
- R 41 is a hydrogen atom or a monovalent organic group.
- R41 Examples of the monovalent organic group for R 41 are the same as those for R 52 described later. Among them, an aliphatic hydrocarbon group having 1 to 20 carbon atoms is preferable, and an alkyl group having 1 to 10 carbon atoms is more preferable.
- one type of primary amine having one primary amino group in one molecule i.e., monofunctional primary amine
- the primary amine used is a primary amine having two or more primary amino groups in one molecule (that is, a primary amine having two or more functional amine), or when a compound other than urea is used as the carbonic acid derivative, for example, when N-alkylurea or N,N'-dialkylurea in which each amino group of urea is substituted with an alkyl group is used as the urea derivative, the same applies.
- the by-produced low-boiling compounds are ammonia and an alkylamine corresponding to the alkyl group.
- N'-dialkylurea is used in step (1), the by-produced low-boiling compound is an alkylamine corresponding to the alkyl group.
- N-substituted carbamate compounds examples include compounds represented by general formula (V) (hereinafter sometimes referred to as “carbamate compound (V)").
- R 51 is a divalent or higher organic group
- R 52 is a monovalent organic group
- n51 is an integer of 2 or more.
- R 51 is an organic group having a valence of 2 or more, preferably an organic group having a valence of 2 or more and 10 or less.
- R 51 is a divalent to tetravalent aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have 1 to 4 ester groups or nitrogen atoms, or 1 to 4 or an aromatic group having 6 or more and 20 or less carbon atoms and having a valence of 2 or more and 3 or less and optionally having a nitrogen atom.
- the aliphatic hydrocarbon group for R 51 includes an alkylene group or an alkanetriyl group, a cycloalkyl group, a cycloalkylene group or a cycloalkanetriyl group, or the alkyl group, the alkylene group or the alkanetriyl group, A group composed of the cycloalkyl group, the cycloalkylene group or the cycloalkanetriyl group is preferable, and a linear or branched alkylene group or alkanetriyl group, cycloalkylene group or cycloalkanetriyl group , or a group composed of the alkylene group or the alkanetriyl group and the cycloalkyl group, the cycloalkylene group or the cycloalkanetriyl group is more preferable.
- linear or branched alkylene groups examples include methylene group, ethylene group, propylene group, trimethylene group, pentylene group, n-hexylene group and decamethylene group.
- the cycloalkylene group includes, for example, a cyclobutylene group, a cyclohexylene group, and the like.
- linear or branched alkanetriyl groups examples include hexanetriyl, nonanetriyl, and decantriyl groups.
- the cycloalkanetriyl group includes, for example, a cyclopropanetriyl group, a cyclobutanetriyl group, a cyclopentanetriyl group, a cyclohexanetriyl group, and the like.
- the aromatic hydrocarbon group for R 51 is preferably a substituted or unsubstituted group having an aromatic ring having 6 or more and 13 or less carbon atoms.
- substituents include alkyl groups, aryl groups, and aralkyl groups.
- the aromatic ring may be an aromatic hydrocarbon ring or a heteroaromatic ring, and specific examples thereof include benzene ring, naphthalene ring, pyridine ring and the like.
- R 51 is an amine compound having an ester group obtained by reacting the carboxy group of an amino acid with a hydroxy compound. is preferably a group other than the terminal primary amino group.
- amine compound having an ester group examples include, for example, acrylic acid-2-aminoethyl ester, 2-methyl-acrylic acid-2-aminoethyl ester, acrylic acid-2-aminopropyl ester, 2- Methyl-Acrylic Acid-2-Aminopropyl Ester, Acrylic Acid-3-Aminopropyl Ester, 2-Methyl-Acrylic Acid-3-Aminopropyl Ester, Acrylic Acid-4-Aminobutyl Ester, 2-Methyl-Acrylic Acid-4 -aminobutyl ester, 5-aminopentyl acrylate, 2-methyl-5-aminopentyl acrylate, 6-aminohexyl acrylate, 2-methyl-6-aminohexyl acrylate, acrylic Acid-8-aminoctyl ester, 2-methyl-acrylic acid-8-aminoctyl ester, acrylic acid-10-aminodecyl ester, 2-methyl-acrylic
- amino acids used for producing amine compounds having an ester group include lysine, alanine, arginine, asparagine, glutamine, glycine, aspartic acid, glutamic acid, ornithine, histidine, isoleucine, leucine, methionine, phenylalanine, tryptophan, and valine. mentioned.
- the amino acid is preferably lysine, arginine, glycine, aspartic acid, glutamic acid or ornithine, more preferably lysine, arginine, glycine, aspartic acid or glutamic acid.
- the hydroxy compounds used in the production of amine compounds having an ester group include alcohols and aromatic hydroxy compounds.
- alcohols examples include methyl alcohol, propyl alcohol, butyl alcohol, amyl alcohol, hexyl alcohol, heptyl alcohol, octyl alcohol, nonyl alcohol, decyl alcohol, undecyl alcohol, lauryl alcohol, dothesyl alcohol, stearyl alcohol, and eicosyl alcohol.
- alcohol allyl alcohol, crotyl alcohol, propargyl alcohol, cyclopentanol, cyclohexanol, benzyl alcohol, cinnamyl alcohol, ethylene glycol, diethylene glycol, polyethylene glycol, propylene glycol, polypropylene glycol, 1,3-butanediol, 1, 4-butanediol, hydrogenated bisphenol A, neopentyl glycol, glycerin, trimethylolpropane, pentaerythritol, ethanolamine, propanolamine (1-amino-2-propanol), dimethanolamine, diethanolamine, dipropanolamine, 1- Amino-2-butanol and the like can be mentioned.
- aromatic hydroxy compounds examples include phenol (carbolic acid), 2-methoxyphenol, cresol, xylenol, carvacrol, motyl, monophenols such as naphthol, catechol, resorcinol, hydroquinone, bisphenol A, bisphenol F, pyrogallol, phloroglucin. and polyhydric phenols such as
- R 51 When the aliphatic hydrocarbon group or aromatic group in R 51 has 1 or more and 4 or less nitrogen atoms, R 51 includes a secondary or tertiary amine in addition to the terminal of the aliphatic hydrocarbon group or aromatic group. It is preferably an organic group obtained by removing the terminal amino group from an amine compound having 1 or more and 4 or less.
- amine compound having 1 to 4 secondary or tertiary amines other than the end of the aliphatic hydrocarbon group or aromatic group herein include, for example, 2-(dimethylamino)ethyleneamine, 2-( Diethylamino)ethyleneamine, 2-(diisopropylamino)ethyleneamine, 2-(cyclohexylamino)ethyleneamine, 3-(cyclohexylamino)propylamine, 3-(diethylamino)propylamine, 3-(dimethylamino)propylamine, diethylenetriamine , diisopropyltriamine, bis-(3-aminopropyl)methyleneamine, 3-(2-aminoethylamino)propylamine, N,N'-bis(3-aminopropyl)ethylenediamine, 1-(3-aminopropyl)imidazole , trisaminoethylamine, trisaminopropylamine, and the like.
- the amine compound is preferably an amine compound having one or more and two or less tertiary amines in addition to the terminal and an aliphatic hydrocarbon group or an aromatic group, and a tertiary amine other than one terminal. and an aliphatic hydrocarbon group or an aromatic group.
- R 52 is a monovalent organic group. Among them, R 52 is preferably an aliphatic hydrocarbon group having 1 or more and 20 or less carbon atoms or an aromatic hydrocarbon group having 6 or more and 20 or less carbon atoms, which may contain an oxygen atom.
- Aliphatic hydrocarbon groups for R 52 include, for example, methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), pentyl group (each isomer), hexyl group (each isomer) , heptyl group (each isomer), octyl group (each isomer), nonyl group (each isomer), decyl group (each isomer), undecyl group (each isomer), dodecyl group (each isomer), tridecyl group (each isomer), tetradecyl group (each isomer), pentadecyl group (each isomer), hexadecyl group (each isomer), heptadecyl group (each isomer), octadecyl group (each isomer), nonadecyl (each isomer),
- the aliphatic hydrocarbon group which may contain an oxygen atom in R 52 includes, for example, a methoxymethyl group, a methoxyethyl group (each isomer), a methoxypropyl group (each isomer), a methoxybutyl group (each isomer), Methoxypentyl group (each isomer), methoxyhexyl group (each isomer), methoxyheptyl group (each isomer), methoxyoctyl group (each isomer), methoxynonyl group (each isomer), methoxydecyl group (each isomer), methoxyundecyl group (each isomer), methododecyl group (each isomer), methoxytridecyl group (each isomer), methoxytetradecyl group (each isomer), methoxypentadecyl group (each isomer isomer
- the aromatic hydrocarbon group for R 52 includes, for example, aryl groups such as phenyl group, naphthyl group, anthryl group, pyrenyl group and phenanthryl group; methylphenyl group (each isomer), ethylphenyl group (each isomer), Propylphenyl group (each isomer), butylphenyl group (each isomer), pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer), nonylphenyl group (each isomer), decylphenyl group (each isomer), undecylphenyl group (each isomer), dodecylphenyl group (each isomer), tridecylphenyl group (each isomer) , tetrade
- Examples of the aromatic hydrocarbon group which may contain an oxygen atom for R 52 include alkoxyaryl groups such as a methoxyphenyl group (each isomer) and an ethoxyphenyl group (each isomer).
- R52 includes phenyl group, naphthyl group, anthryl group, pyrenyl group, phenanthryl group, methylphenyl group (each isomer), ethylphenyl group (each isomer), propylphenyl group (each isomer), butyl Phenyl group (each isomer), pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer), nonylphenyl group (each isomer) isomer), decylphenyl group (each isomer), undecylphenyl group (each isomer), dodecylphenyl group (each isomer), tridecylphenyl group (each isomer), tetradecylphenyl group (each isomer
- n51 represents the number of carbamate groups and is an integer of 2 or more, preferably an integer of 2 or more and 10 or less, more preferably an integer of 3 or more, and 3 is more preferred.
- Preferred N-substituted carbamate compounds (V) include, for example, compounds represented by the following formulas (V-1) to (V-5).
- the aprotic solvent is preferably a carbonic acid derivative, an ether, a hydrocarbon, a ketone, or a mixture thereof, more preferably a carbonic acid derivative, a hydrocarbon, a ketone, or a mixture thereof. is more preferred.
- carbonic acid derivative refers to a compound having a carbonyl group.
- carbonic acid derivative a compound represented by the following general formula (III) (hereinafter sometimes referred to as “carbonic acid derivative (III)”) is preferable.
- R 31 and R 32 are each independently a substituted or unsubstituted alkoxy group having 1 to 20 carbon atoms or an aryloxy group having 6 to 20 carbon atoms.
- R 31 and R 32 may be the same or different, but are preferably the same.
- Examples of the alkoxy group having 1 to 20 carbon atoms in R 31 and R 32 include a methoxy group, an ethoxy group, a propyloxy group (each isomer), a butoxy group (each isomer), a hexyloxy group (each isomer ) and the like.
- Examples of the aryloxy group having 6 to 20 carbon atoms in R 31 and R 32 include a phenoxy group and a naphthyloxy group.
- substituents possessed by alkoxy groups, aryloxy groups, and aralkyloxy groups include alkyl groups and alkoxy groups.
- R 31 and R 32 are each independently preferably a substituted or unsubstituted aryloxy group having 6 or more and 20 or less carbon atoms.
- Preferred carbonic acid derivatives (III) include, for example, carbonic acid esters.
- a carbonate ester refers to a compound in which one or two of the diatomic hydrogen atoms of carbonic acid CO(OH) 2 are substituted with an alkyl group or an aryl group.
- Carbonic acid ester is a compound represented by the following general formula (III-1) (hereinafter sometimes referred to as “carbonic acid ester (III-1)").
- R 311 and R 312 are each independently a substituted or unsubstituted alkyl group having 1 to 20 carbon atoms or an aryl group having 6 to 20 carbon atoms.
- R 311 and R 312 may be the same or different, but are preferably the same.
- alkyl groups having 1 to 20 carbon atoms in R 311 and R 312 include methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), pentyl group (each isomer), Hexyl group (each isomer), heptyl group (each isomer), octyl group (each isomer), nonyl group (each isomer), decyl group (each isomer), undecyl group (each isomer), dodecyl group (each isomer), tridecyl group (each isomer), tetradecyl group (each isomer), pentadecyl group (each isomer), hexadecyl group (each isomer), heptadecyl group (each isomer), octadecyl group (each isomer), nonadecyl (each
- Examples of the aryl group having 6 to 20 carbon atoms in R 311 and R 312 include phenyl group, naphthyl group, anthryl group, pyrenyl group and phenanthryl group.
- An alkoxy group etc. are mentioned as a substituent which an alkyl group has.
- An alkyl group, an alkoxy group, etc. are mentioned as a substituent which an aryl group has.
- Alkyl groups having an alkoxy group as a substituent include, for example, a methoxyethyl group (each isomer), a methoxypropyl group (each isomer), a methoxybutyl group (each isomer), and a methoxypentyl group.
- each isomer methoxyhexyl group (each isomer), methoxyheptyl group (each isomer), methoxyoctyl group (each isomer), methoxynonyl group (each isomer), methoxydecyl group (each isomer) , methoxyundecyl group (each isomer), methododecyl group (each isomer), methoxytridecyl group (each isomer), methoxytetradecyl group (each isomer), methoxypentadecyl group (each isomer), Methoxyhexadecyl group (each isomer), methoxyheptadecyl group (each isomer), methoxyoctadecyl group (each isomer), methoxynonadecyl (each isomer), ethoxymethyl group, ethoxyethyl group (
- aryl group having an alkyl group as a substituent examples include a methylphenyl group (each isomer), an ethylphenyl group (each isomer), a propylphenyl group (each isomer), a butylphenyl group (each isomer), Pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer), nonylphenyl group (each isomer), decylphenyl group (each isomer), undecylphenyl group (each isomer), dodecylphenyl group (each isomer), tridecylphenyl group (each isomer), tetradecylphenyl group (each isomer), dimethylphenyl group (each isomer ),
- an alkoxyaryl group having an alkoxy group as a substituent includes, for example, a methoxyphenyl group (each isomer), an ethoxyphenyl group (each isomer), and the like.
- R 311 and R 312 are preferably substituted or unsubstituted aryl groups.
- a phenyl group, a methoxyphenyl group (each isomer), or an ethoxyphenyl group (each isomer) is more preferable.
- Preferred carbonate esters (III-1) include, for example, diphenyl carbonate, bis(2-methoxyphenyl) carbonate, bis(2-ethoxyphenyl) carbonate and the like.
- isocyanate compound examples include compounds represented by general formula (VII) (hereinafter sometimes referred to as “isocyanate compound (VII)").
- R 71 and n71 are the same as R 51 and n51 above, respectively.
- R 71 is an aliphatic hydrocarbon group
- specific examples of the isocyanate (VII) include aliphatic diisocyanates, aliphatic triisocyanates, and substituted cycloaliphatic polyisocyanates.
- aliphatic diisocyanates examples include diisocyanatoethane, diisocyanatopropane (each isomer), diisocyanatobutane (each isomer), diisocyanatopentane (each isomer), diisocyanatohexane (each isomer), diisocyanatodecane (each isomer), isophorone diisocyanate (each isomer), dicyclohexylmethane diisocyanate (each isomer), and the like.
- aliphatic triisocyanates examples include triisocyanatohexane (each isomer), 4-isocyanatomethyl-1,8-octamethylene diisocyanate, triisocyanatononane (each isomer), triisocyanatodecane (each isomer) and the like.
- Substituted cycloaliphatic polyisocyanates include, for example, diisocyanatocyclobutane (each isomer), diisocyanatocyclohexane (each isomer), 3-isocyanatomethyl-3,5,5-trimethylcyclohexylisocyanate (at least one of cis and trans isomers), methylenebis(cyclohexyl isocyanate) (each isomer), and the like.
- R 71 is an aromatic group
- specific examples of the isocyanate (VII) include aromatic diisocyanates and aromatic triisocyanates.
- aromatic diisocyanates include diisocyanatobenzene (each isomer), diisocyanatotoluene (each isomer), bis(isocyanatophenyl)methane (each isomer), diisocyanatomesitylene (each isomer ), diisocyanatobiphenyl (each isomer), diisocyanatodibenzyl (each isomer), bis(isocyanatophenyl)propane (each isomer), bis(isocyanatophenyl)ether (each isomer), bis (isocyanatophenoxyethane) (each isomer), diisocyanatoxylene (each isomer), diisocyanatoanisole (each isomer), diisocyanatophenetol (each isomer), diisocyanatonaphthalene (each isomer) isomer), diisocyanatomethylbenzene (each is
- aromatic triisocyanates examples include triisocyanatobenzene (each isomer), triisocyanato-methylbenzene (each isomer), tris(isocyanatopropan-yl)benzene (each isomer), tris(isocyanate Natopropan-yl)-methylbenzene (each isomer), Tris(isocyanatomethyl)-methylbenzene (each isomer), ((isocyanato-phenylene)bis(methylene))bis(isocyanatobenzene) (each isomer) etc.
- the electron-withdrawing effect of the aromatic group increases the reactivity of the directly attached isocyanate group. Furthermore, as the number of isocyanate groups directly bonded to the same aromatic compound increases, the reactivity increases due to mutual electron attraction. From the viewpoint of reactivity, the number of isocyanate groups for R 71 is preferably 2 or more, more preferably 3 or more. On the other hand, when the reactivity of the isocyanate increases, the reaction between moisture, the isocyanate itself, and other impurities occurs, and the stability of the compound at room temperature and during heating decreases. From the viewpoint of the stability of the isocyanate compound, the number of isocyanate groups directly bonded to R 71 is preferably 4 or less, more preferably 3 or less.
- isocyanate (VII) examples include acrylic acid-2-isocyanato-ethyl ester, 2-methyl-acrylic acid-2-isocyanato-ethyl ester, acrylic acid-2-isocyanato-propyl ester, 2-methyl-acrylic acid-2-isocyanato-propyl ester, acrylic acid-3-isocyanato-propyl ester, 2- Methyl-acrylic acid-3-isocyanato-propyl ester, acrylic acid-4-isocyanato-butyl ester, 2-methyl-acrylic acid-4-isocyanato-butyl ester, acrylic acid-5-isocyanato-pentyl ester, 2-methyl- Acrylic Acid-5-Isocyanato-Pentyl Ester, Acrylic Acid-6-Isocyanato-Hexyl Ester, 2-Methyl-Ac
- the isocyanate compound obtained by the production method of the present embodiment can be suitably used as a production raw material for polyurethane foams, paints, adhesives, and the like. According to the production method of the present embodiment, an isocyanate compound can be produced in good yield without using highly toxic phosgene.
- Carbonyl compound (I) is a by-product produced when the N-substituted carbamate compound in step (2) is thermally decomposed to produce an isocyanate compound.
- a compound having an isocyanate terminal or an N-substituted carbamate terminal and a carbonate ester undergo a reaction from intermolecular interaction under reaction conditions such as a temperature drop in thermal decomposition conditions, and each terminal group is formed on the carbonate skeleton. It is believed that the carbonyl compound (I) is produced as the insertion reaction proceeds.
- the carbonyl compound (I) acts as a good solvent for the modified isocyanate compound, and the carbonyl compound (I) acts as a terminal blocking agent to increase the molecular weight of the modified isocyanate compound.
- the carbonyl compound (I) By suppressing , it is possible to suppress the formation of solids and the increase in liquid viscosity in the isocyanate compound production system due to the thermal decomposition reaction of the carbamate compound.
- the carbonyl compound (I) since the carbonyl compound (I) has a higher boiling point than the isocyanate compound and has fewer cross-linking points than the modified isocyanate compound, it can also serve as a solvent during purification of the isocyanate compound.
- Examples of the carbonyl compound (I) include compounds represented by the following general formula (I).
- R 11 is a (n11+n12)-valent organic group
- R 12 is a monovalent organic group.
- n11 is an integer of 1 or more and 8 or less
- n12 is 0 or more and 7 or less. It is an integer, and the sum of n11 and n12 is 2 or more and 8 or less.
- R11 is a (n11+n12)-valent organic group, that is, an organic group having a valence of 2 or more and 8 or less.
- R 11 is a divalent to tetravalent aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have an ester group of 1 to 4 carbon atoms or a nitrogen atom, or 6 or more carbon atoms. It is preferably an aromatic hydrocarbon group having a valence of 20 or less and having a valence of 20 or more and 3 or less.
- R 11 are the same as those exemplified as the organic group for R 51 above.
- R 11 is preferably a group represented by any one of the following formulas (Ia-1) to (Ia-28), such as formulas (Ia-1), (Ia-2), (Ia-3 ), (Ia-14), (Ia-18), or (Ia-19).
- formulas (Ia-1) to (Ia-28) such as formulas (Ia-1), (Ia-2), (Ia-3 ), (Ia-14), (Ia-18), or (Ia-19).
- a wavy line indicates a bond.
- R12 is a monovalent organic group, preferably an aliphatic hydrocarbon group having 1 to 20 carbon atoms or an aromatic hydrocarbon group having 6 to 20 carbon atoms, which may contain an oxygen atom.
- R 12 include those exemplified for R 52 above.
- n11 represents the number of carbamate groups and is an integer of 1 or more and 8 or less.
- n12 represents the number of isocyanate groups and is an integer of 0 or more and 7 or less.
- the sum of n11 and n12 is an integer of 2 or more and 8 or less, preferably an integer of 2 or more and 6 or less, more preferably an integer of 2 or more and 5 or less, and still more preferably an integer of 3 or more and 4 or less.
- the sum of n11 and n12 is equal to or higher than the above lower limit, the normal boiling point is increased as the molecular weight of the carbonyl compound is increased, and separation from the isocyanate becomes easier.
- the modification reaction with the highly reactive isocyanate group of the isocyanate compound is further suppressed, and sticking and clogging to the device can be further reduced.
- Preferred carbonyl compounds (I) include, for example, compounds represented by the following formulas (I-1) to (I-17b).
- the substituent R' in the following formulas (I-1) to (I-17b) is a phenyl group, (o-, m-, or p-methyl)phenyl group, ethyl group, propyl group (each isomeric isomers), or butyl groups (each isomer).
- a primary amine compound, a carbonic acid derivative, and a hydroxy compound are reacted in the presence of an amine compound having no active hydrogen as a catalyst to obtain a carbamate compound. Including the reaction step.
- the reaction to produce a carbamate compound is an equilibrium reaction, but equilibrium is reached by using an amine compound having no active hydrogen as a catalyst.
- the time required can be shortened.
- favorable effects such as improvement in the selectivity of carbamate compounds due to downsizing of the device and reduction of side reactions are produced.
- amine compounds that do not have active hydrogen are non-metallic catalysts, they can be easily separated from the reaction mixture by selecting one that has an appropriate vapor pressure, and the carbamate compound is thermally decomposed into an isocyanate compound. Modification at the time of obtaining can be suppressed.
- any amine compound having no active hydrogen which is a catalyst
- the pressure is higher than the vapor pressure of the compound, it becomes easy to separate and recover the catalyst from the reaction liquid containing the hydroxy compound and the N-substituted carbamate.
- the vapor pressure of the catalyst is lower than the vapor pressure of the hydroxy compound under the reaction conditions of the reaction step, the concentration of the catalyst in the liquid phase is kept high, which is effective in promoting the reaction.
- a carbamate reaction step> In the reaction step, a carbamate compound is obtained by reacting a primary amine compound, a carbonic acid derivative, and a hydroxy compound in the presence of an amine compound having no active hydrogen as a catalyst.
- the reaction step preferably includes the following steps (X1) and (X2).
- a primary amine compound, a carbonic acid derivative, and a hydroxy compound are subjected to a carbamate reaction in the presence of an amine compound having no active hydrogen as a catalyst to form an N-substituted carbamate, resulting in a carbonyl derived from the carbonic acid derivative.
- Step (X1) carbamate synthesis step
- a primary amine compound, a carbonic acid derivative, and a hydroxy compound are used as starting materials, and in the presence of an amine compound having no active hydrogen as a catalyst, a carbamate reaction is performed to produce a carbamate compound.
- compound and a compound having a carbonyl group derived from a carbonic acid derivative may contain one or more compounds selected from the group consisting of an active hydrogen compound derived from a carbonic acid derivative and an amine compound having no active hydrogen.
- the step (X1) is roughly divided into A method for producing an N-substituted carbamate by "simultaneously" reacting a primary amine compound, a carbonic acid derivative, and a hydroxy compound in the presence of at least one amine compound having no active hydrogen as a catalyst (Method (1A)); A primary amine compound and a carbonic acid derivative are reacted in the presence or absence of an amine compound having no active hydrogen as a catalyst to produce a carbonyl compound having a group derived from the primary amine compound.
- a method (method (1B)) can be performed in two ways.
- carbonyl compound having a group derived from a primary amine compound is a compound produced by a reaction between a primary amine compound and a carbonic acid derivative and represented by the following general formula (XII).
- R 121 is a group derived from a primary amine compound, i.e., the same as R 21b in general formula (IIb) described later.
- R 122 is a group derived from a carbonic acid derivative, i.e., R 321 , R 322 , R 323 or R 324 in general formula (III-2), R 331 in general formula (III-3) described later, or R 331 in general formula (III-1b) described later (The same as R 311b or R 312b .
- n121 is the same as n21b in general formula (IIb) described later.
- method (1A) and method (1B) may be combined.
- Method (1A) "Simultaneously" in method (1A) means that the step of producing the N-substituted carbamate is divided into two steps in method (1B), whereas the steps are not separated in method (1A). However, it does not necessarily mean that the primary amine compound, the carbonic acid derivative and the hydroxy compound react all at the same time.
- the reaction conditions for producing an N-substituted carbamate by reacting a primary amine compound, a carbonic acid derivative and a hydroxy compound in the presence of a catalyst vary depending on the compound to be reacted, but the amount of the hydroxy compound used is usually The stoichiometric ratio of the hydroxyl group of the hydroxy compound to the molar amount of the amino group of the primary amine compound is in the range of 1 to 500 times the molar amount of the amino group of the primary amine compound. If the amount of the hydroxy compound used is small, complexly substituted carbonyl compounds and the like are likely to be produced, so it is preferable to use a large excess of the hydroxy compound, but considering the size of the reactor, it is preferably 1-fold molar. The range is from 1.5 to 100 mol, more preferably from 2 to 60 mol.
- the amount (molar amount) of the carbonic acid derivative to be used is usually in the range of 1 to 100 times the molar amount of the amino group of the primary amine compound in terms of stoichiometric ratio. Even if the amount of the carbonic acid derivative used is small, complicatedly substituted carbonyl compounds and the like are likely to be generated, so it is preferable to use an excessive amount of the carbonic acid derivative. In some cases, a carbonyl compound is likely to be generated, and an unreacted carbonic acid derivative may remain. Therefore, it is preferably 1.0 to 10-fold mol, more preferably 1.0 to 5-fold mol, and still more preferably 1.05 to 2-fold mol.
- the amount (molar amount) of the amine compound having no active hydrogen used as a catalyst may be adjusted as appropriate within the range in which the reaction proceeds well. On the other hand, it can be 0.0001 to 100 times mol, preferably 0.0001 to 30 times mol.
- the amount (molar amount) to be used is preferably 0.1 times the molar amount of the amino group of the primary amine compound, more preferably 0. 0.05 mol or less, more preferably 0.01 mol or less.
- a base whose conjugate acid has a pKa of 7 or more and 12 or less must be used in large amounts in order to obtain the same promotion effect as when using the above strong base. Since the effect of accelerating the modification reaction is smaller than that of a strong base, it may be used in a large amount, but the range of the preferable amount used is 0.5 to 30 times the molar amount of the amino group of the primary amine compound. mol or less, more preferably 0.5-fold mol or more and 10-fold mol or less, further preferably 1-fold mol or more and 3-fold mol or less.
- the preferred amount of use is 0.5 to 30 times the molar amount of the amino group of the primary amine compound, and more It is preferably 1-fold mol or more and 10-fold mol or less.
- the reaction temperature depends on the reactivity of the primary amine compound, carbonic acid derivative and hydroxy compound used, it is preferably in the range of 100° C. or higher and 350° C. or lower, more preferably in the range of 120° C. or higher and 320° C. or lower. More preferably, it is in the range of 140°C or higher and 300°C or lower.
- the reaction temperature is equal to or higher than the above lower limit, the reaction can be accelerated, and the increase in the complexly substituted carbonyl compound can be more effectively suppressed.
- reaction temperature is equal to or lower than the above upper limit
- decomposition of the carbonic acid derivative and accompanying side reactions can be more effectively suppressed, and the dehydrogenative modification or product of the hydroxy compound can be obtained. It is possible to more effectively suppress the decomposition reaction, modification reaction, and the like of the N-substituted carbamate.
- the reaction pressure varies depending on the composition of the reaction system, the reaction temperature, the reactor, etc., and can be carried out under reduced pressure, normal pressure, or increased pressure, but is usually carried out in the range of 0.01 kPa to 10 MPa (absolute pressure). is preferred.
- step (X1) the reaction to produce the N-substituted carbamate is often carried out mainly in the liquid phase. Therefore, the hydroxy compound and catalyst are preferably present as liquid phase components under the reaction conditions.
- the hydroxy compound and the compound having a carbonyl group derived from a carbonic acid derivative are introduced into the condenser as gas phase components and condensed in the condenser. It is preferably present as a phase component as well. Therefore, the reaction conditions are set so that the catalyst exists mainly as a liquid phase component, and the hydroxy compound exists partly as a liquid phase component and partly as a gaseous phase component.
- reaction conditions are set so that at least one hydroxy compound is present as a liquid phase component. Since such reaction conditions (reaction temperature, pressure) are closely related to the properties of the hydroxy compound and catalyst used, particularly the correlation between temperature and vapor pressure, the properties of the hydroxy compound and catalyst ( Correlation between temperature and vapor pressure) is measured or investigated and used as an index for determining reaction conditions. Incidentally, it is common knowledge for those skilled in the art that the correlation between the temperature and the vapor pressure of a substance varies greatly depending on the purity of the substance, coexisting compounds and their amounts. It is clear that not only the properties of the compounds and the catalyst (correlation between temperature and vapor pressure) but also the coexisting compounds and their amounts should be taken into account.
- a reaction in which an N-substituted carbamate is produced from a primary amine compound, a carbonic acid derivative, and a hydroxy compound is an equilibrium reaction, and the carbonic acid derivative is one or more selected from the group consisting of urea compounds and N-unsubstituted carbamates.
- the reaction is largely biased toward the original system. Therefore, in order to increase the yield of the N-substituted carbamate, it is necessary to carry out the reaction while removing by-product active hydrogen compounds out of the system as much as possible.
- the active hydrogen compound is removed so that the active hydrogen compound concentration in the reaction solution is 1000 mass ppm or less, more preferably 300 mass ppm or less, still more preferably 100 mass ppm or less, and particularly preferably 10 mass ppm or less. do.
- the method include a reactive distillation method, a method using an inert gas, a method using membrane separation, and a method using adsorption separation.
- the reactive distillation method is a method in which an active hydrogen compound that is successively produced under reaction is separated in a gaseous state by distillation. In order to increase the distillation efficiency of the active hydrogen compound, it can be carried out under boiling of the solvent or hydroxy compound.
- the method using an inert gas is a method in which an active hydrogen compound that is successively produced under the reaction is separated from the reaction system by entraining it in a gaseous state with an inert gas.
- an inert gas for example, nitrogen, helium, argon, carbon dioxide gas, methane, ethane, propane, etc. are used singly or in combination, and a method of introducing the inert gas into the reaction system is preferred.
- Adsorbents used in the adsorption separation method include, for example, adsorbents that can be used under the temperature conditions at which the reaction is carried out, such as silica, alumina, various zeolites, and diatomaceous earths.
- the method for removing these active hydrogen compounds out of the system may be carried out singly or in combination of a plurality of methods.
- the active hydrogen compound produced as a by-product here means, for example, a raw material primary amine compound produced from urea or substituted urea and an amine compound, an amine compound other than an amine compound having no active hydrogen as a catalyst, and a carbonate ester.
- Examples include hydroxy compounds other than solvents generated from amine compounds.
- the reaction time (residence time in the case of a continuous reaction) varies depending on the composition of the reaction system, the reaction temperature, the method for removing the active hydrogen compound, the reaction apparatus, the reaction pressure, etc., but is usually 0.01 hour or more and 100 hours or less. .
- the reaction time can also be determined according to the amount of the N-substituted carbamate that is the target compound. For example, the reaction solution is sampled, the content of the N-substituted carbamate in the reaction solution is quantified, and the yield is 10% by mass or more relative to the mass of the raw material primary amine compound used. After confirming that, the reaction may be stopped, or after confirming that the yield is 90% by mass or more, the reaction may be stopped.
- a suitable solvent such as Alkanes such as pentane (each isomer), hexane (each isomer), heptane (each isomer), octane (each isomer), nonane (each isomer), decane (each isomer); Aromatic hydrocarbons and alkyl-substituted aromatic hydrocarbons such as benzene, toluene, xylene (each isomer), ethylbenzene, diisopropylbenzene (each isomer), dibutylbenzene (each isomer), mesitylene, paracymene, tetralin, and naphthalene ; Nitrile compounds such as acetonitrile and benzonitrile; Aromatic compounds substituted by halogen or nitro group such as chlorobenzen
- the carbamate reaction contains a compound having a carbonyl group derived from a hydroxy compound and a carbonic acid derivative, and is selected from the group consisting of an active hydrogen compound derived from a carbonic acid derivative and an amine compound having no active hydrogen as a catalyst. It is carried out in a system having a gas phase which may contain the above compounds and a liquid phase in which the carbamate reaction is carried out. Although the carbamate reaction may occur in the gas phase depending on the reaction conditions, most of the carbamate reaction takes place in the liquid phase. At that time, the volume content of the liquid phase in the reactor in which the carbamate reaction is carried out is preferably 50% by volume or less with respect to the volume of the gas phase.
- the carbamate reaction When the carbamate reaction is carried out continuously over a long period of time, fluctuations in operating conditions (temperature, pressure, etc.) may produce polymeric by-products, but the liquid phase volume content in the reactor is large. and deposition and accumulation of such polymeric by-products on the reactor can be avoided.
- one or more compounds selected from the group consisting of urea compounds and N-unsubstituted carbamates are used as the carbonic acid derivative, if the liquid phase volume content is too large, the removal efficiency of the by-produced active hydrogen compound is reduced.
- the content of the liquid phase volume relative to the volume of the gas phase is preferably 50% by volume or less, more preferably 30% by volume or less, and even more preferably 20% by volume. % or less.
- liquid phase volume content here means the reaction tank section in the case of a tank reactor, the stage below the feed stage in the case of a tower reactor (not including the bottom part and reboiler part), the thin film distiller In the case of , represents the liquid phase volume ratio to the gas phase volume in the capacity of the thin film distiller.
- the reactor used in carrying out the carbamate reaction is not particularly limited as long as it is a reactor equipped with a condenser, and known reactors can be used.
- One or more reactors selected from the group consisting of are preferably used.
- the material of the reactor is also not particularly limited, and known materials can be used.
- it can be made of glass, stainless steel, carbon steel, hastelloy, or a substrate having a glass lining or a Teflon (registered trademark) coating.
- SUS304, SUS316, SUS316L and the like are inexpensive and can be preferably used.
- instrumentation equipment such as flowmeters and thermometers, reboilers, pumps, condensers and other known process devices may be added.
- a known method such as steam or a heater may be used for heating, and a known method such as natural cooling, cooling water, or brine may be used for cooling.
- a step as described later may be added.
- the steps that can be added include, for example, a step of removing the generated active hydrogen compound, a step of purifying the primary amine compound, a step of dissolving the carbonic acid derivative in the hydroxy compound, a step of dissolving the hydroxy compound, and separating the hydroxy compound.
- a step of purifying, a step of purifying N-substituted carbamate from the resulting reaction solution, a step of incinerating or discarding by-products, etc. may be added within the scope of those skilled in the art. .
- Step (b1) The amount (molar amount) of the hydroxy compound used and the amount (molar amount) of the amine compound having no active hydrogen used as a catalyst can be the same as the amount used in the above method (1A).
- the amount (molar amount) of the carbonic acid derivative to be used is usually in the range of 1 to 100 times the stoichiometric molar amount of the amino group of the primary amine compound. Even if the amount of the carbonic acid derivative used is small, complexly substituted carbonyl compounds and the like are likely to be produced, so it is preferable to use an excessive amount of the carbonic acid derivative. On the other hand, if too much carbonic acid derivative is used, complex substituted carbonyl compounds may rather tend to form, or unreacted carbonic acid derivative may remain. Therefore, it is preferably in the range of 1.0 to 10-fold mol, more preferably 1.1 to 5-fold mol.
- the reaction temperature of the step (b1) can be usually within the range of 30°C or higher and 250°C or lower. Higher temperatures are preferred to increase the reaction rate. On the other hand, at high temperatures, unfavorable reactions (for example, decomposition reactions of carbonic acid derivatives, complicated side reactions caused by such reactions, etc.) may occur to produce complexly substituted carbonyl compounds. Therefore, the range is preferably 50° C. or higher and 200° C. or lower, more preferably 70° C. or higher and 180° C. or lower. In order to keep the reaction temperature constant, a known cooling device or heating device may be installed in the reactor in which step (b1) is carried out.
- the reaction pressure varies depending on the type of compound used, the composition of the reaction system, the reaction temperature, the reactor, etc., but it is usually preferably carried out in the range of 0.01 kPa or more and 10 MPa or less (absolute pressure). Considering the easiness of the pressure, the range of 0.1 kPa or more and 5 MPa or less (absolute pressure) is preferable.
- the reaction time (residence time in the case of a continuous process) is not particularly limited and is usually 0.001 to 100 hours, preferably 0.01 to 80 hours, more preferably 0.1 to 50 hours. be.
- the reaction solution can be sampled and, for example, liquid chromatography can be used to confirm that a desired amount of a carbonyl compound having a group derived from a primary amine compound is produced, and then the reaction can be terminated.
- the step (b1) is a step of producing the carbonyl compound.
- a compound having a ureylene group or the like is produced, and not only the production amount of the N-substituted carbamate is reduced, but also adhesion to the reactor and solidification often occur.
- step (b1) it is preferable to reduce the amount of amino groups derived from the primary amine compound as much as possible.
- the ratio of the number of moles of amino groups derived from an organic amine to the number of moles of carbonyl groups constituting a carbonyl compound having a group derived from a primary amine compound is preferably 0.25 or less, more It is preferable to continue the reaction until it becomes 0.1 or less, more preferably 0.05 or less.
- the hydroxy compound used in step (b1) may be completely the same as the hydroxy compound used in step (b2), may be partially the same, or may be different. Above all, it is preferable that the hydroxy compound used in step (b1) is partially or wholly the same as the hydroxy compound used in step (b2) for ease of operation. As explained below, the reaction of step (b1) is carried out in the presence of an aromatic hydroxy compound, or the reaction of step (b1) is carried out in the presence of an alcohol or an aromatic hydroxy compound followed by the aromatic hydroxy Adding a compound is even more preferred.
- the reaction solvent shown here can be used in any amount, but when alcohol is used as the reaction solvent, the amount (molar amount) used is based on the molar amount of the amino group of the primary amine compound. On the other hand, it can be used in the range of more than 1-fold mol and less than 100-fold mol in stoichiometric ratio. In order to improve the fluidity of the reaction solution and allow the reaction to proceed efficiently, it is preferable to use excess alcohol relative to the amino groups of the primary amine compound. On the other hand, if too much alcohol is used, there are also adverse effects such as an increase in the size of the reactor.
- the amount (molar amount) used is preferably in the range of more than 5-fold mol and less than 50-fold mol, more preferably 8-fold mol in stoichiometric ratio with respect to the molar amount of the amino group of the primary amine compound. It can be used in the range of more than 20-fold molar.
- the amount (molar amount) used is stoichiometric with respect to the molar amount of the amino group of the primary amine compound. It can be used in the range of more than 1-fold mol and less than 100-fold mol. In order to improve the fluidity of the reaction solution and allow the reaction to proceed efficiently, it is preferable to use an excessive amount of the aromatic hydroxy compound relative to the amino groups of the primary amine compound. On the other hand, if too much aromatic hydroxy compound is used, there are also adverse effects such as an increase in the size of the reactor.
- the amount (molar amount) to be used is preferably more than 2-fold mol and less than 50-fold mol, more preferably more than 3-fold mol in stoichiometric ratio with respect to the molar amount of the amino group of the primary amine compound. It can be used in the range of less than 20 times molar.
- aromatic hydroxy compounds are preferably used as the hydroxy compound, considering the solubility of the carbonyl compound having a group derived from the primary amine compound to be produced.
- JP-A-6-41045 (Reference 1) describes that polyhexamethylene-urea produced by the reaction of urea and hexamethylenediamine is difficult to dissolve in n-butanol.
- aromatic hydroxy compounds are often excellent in the solubility of various reaction products.
- the aromatic hydroxy compound When an aromatic hydroxy compound is used as the reaction solvent, the aromatic hydroxy compound may be used alone or in combination with another solvent. range. When the aromatic hydroxy compound is added after step (b1) is performed in the presence of alcohol, the aromatic hydroxy compound is used within the above range. At that time, the amount of alcohol used in the reaction of step (b1) is also the amount of alcohol in the above-mentioned stoichiometric ratio with respect to the molar amount of the amino group of the primary amine compound.
- reactors used when carrying out the reaction in step (b1), and known reactors can be used.
- conventionally known reactors such as a stirring tank, a pressurized stirring tank, a reduced pressure stirring tank, a column reactor, a distillation column, a packed column, a thin film distiller, a tubular reactor, etc. They can be used in combination as appropriate.
- the reaction may be of a batch type or a continuous flow type, and a reaction apparatus may be selected according to each reaction type.
- a continuous flow system is preferable, and it is often efficient to conduct the reaction by allowing the raw material solution to flow through a tube-like channel (pipe) having a small inner diameter.
- the thickness and length of the flow path are important, but the amount of production of the carbonyl compound having a group derived from the primary amine compound, the area of the heat conducting surface with respect to the volume of the hollow interior, the required residence time (reaction time) can be determined as appropriate.
- a single channel can share the front part for the supply process and the rear part for the reaction process. In this case, the part where the solution flowing through the channel can reach the target temperature can be considered for the reaction process, and the other part can be considered for the supply process.
- the material of the reactor there are no particular restrictions on the material of the reactor, and known materials can be used.
- it can be made of glass, stainless steel, carbon steel, hastelloy, or a substrate having a glass lining or a Teflon (registered trademark) coating.
- SUS304, SUS316, SUS316L, etc. are inexpensive and can be preferably used.
- known process devices such as instrumentation equipment such as a flow meter and thermometer, a mechanism for maintaining pressure, a reboiler, a pump, a condenser and the like may be added.
- a known method such as steam or a heater may be used for heating, and a known method such as natural cooling, cooling water, or brine may be used for cooling.
- a step as described later may be added.
- the steps that can be added include, for example, a step of removing an active hydrogen compound produced as a by-product, a step of purifying a primary amine compound, a step of dissolving a carbonic acid derivative in a hydroxy compound, a step of dissolving a hydroxy compound, and a step of dissolving a hydroxy compound.
- step (b1) From the viewpoint of selectively producing a carbonyl compound having a group derived from a primary amine compound in the step (b1), it is preferable that a certain amount of an active hydrogen compound derived from a carbonic acid derivative is dissolved in the reaction solution. On the other hand, in step (b2) described later, the active hydrogen compound is preferably removed. Therefore, when the reaction liquid of step (b1) is used as the raw material of step (b2), it is also a preferable method to separate the active hydrogen compound from the reaction liquid in advance.
- the separation method is not particularly limited, but for example, the gas phase portion and the liquid phase portion are rapidly separated by sending the reaction liquid to a decompressed container, and the carbonyl compound having a group derived from a primary amine compound is included. The liquid phase can be recovered and used.
- step (b1) When a reaction solvent is used in step (b1), the reaction solvent may be removed from the reaction solution in step (b1) before performing step (b2), or step (b2) may be performed without removing it. good too.
- step (b2) it is preferable to use the hydroxy compound used as the reaction solvent in step (b1) as it is as part of the hydroxy compound in step (b2).
- Step (b2) is a step of producing an N-substituted carbamate by reacting the carbonyl compound having a group derived from the primary amine compound obtained in step (b1) with a hydroxy compound.
- step (b1) When a hydroxy compound is used as a reaction solvent in step (b1) and the hydroxy compound is the same as the hydroxy compound in step (b2), the reaction solution obtained in step (b1) is used as it is in step (b2). It can be performed.
- step (b2) may be performed by adding a new hydroxy compound to the reaction solution obtained in step (b1). .
- one or more types of hydroxy compounds are newly added to the reaction solution obtained in step (b1), and then part or all of the reaction solvent in step (b1) is separated, followed by step (b2). ) may be performed.
- step (b2) may be performed after newly adding a hydroxy compound.
- the hydroxy compound added here is one or more hydroxy compounds selected from the group consisting of an alcohol represented by general formula (VIa) described later and an aromatic hydroxy compound represented by general formula (VIb) described later. including.
- the method for separating the reaction solvent used in step (b1) is not particularly limited, and known methods such as distillation separation, membrane separation, and extraction separation can be used, but distillation separation is preferred.
- the hydroxy compound used in step (b2) is preferably an aromatic hydroxy compound represented by general formula (VIb) described later.
- Step (b2) may be performed after adding a catalyst to the reaction solution of step (b1) before performing step (b2) without using a catalyst in step (b1).
- the reaction conditions for producing an N-substituted carbamate by reacting a carbonyl compound having a group derived from a primary amine compound with a hydroxy compound vary depending on the compound to be reacted, but the amount of the hydroxy compound used is The (molar amount) is in the range of 1 to 500 times the stoichiometric ratio of the number of moles of the hydroxy compound to the number of moles of the carbonyl group of the carbonyl compound used.
- the amount of the hydroxy compound is small, a complexly substituted carbonyl compound or a high-molecular-weight compound having a carbonyl bond in the molecule is likely to be produced, so it is preferable to use a large excess of the hydroxy compound.
- the amount (molar amount) of the hydroxy compound used is such that the number of moles of the hydroxy compound is stoichiometric with respect to the number of moles of the carbonyl group of the carbonyl compound used.
- the range is preferably from 1 to 200 times, more preferably from 1.5 to 100 times, and even more preferably from 2 to 60 times.
- the reaction temperature depends on the compound used, it is preferably in the range of 40°C to 380°C, more preferably in the range of 50°C to 320°C, still more preferably in the range of 60°C to 300°C, particularly preferably It is in the range of 80°C or higher and 300°C or lower, most preferably in the range of 100°C or higher and 280°C or lower.
- the reaction temperature is equal to or higher than the above lower limit, the reaction can be accelerated, and the increase in the complexly substituted carbonyl compound can be more effectively suppressed.
- reaction temperature is equal to or lower than the above upper limit
- decomposition of the carbonic acid derivative and accompanying side reactions can be more effectively suppressed, and the dehydrogenative modification or product of the hydroxy compound can be obtained. It is possible to more effectively suppress the decomposition reaction, modification reaction, and the like of the N-substituted carbamate.
- the reaction pressure varies depending on the composition of the reaction system, the reaction temperature, the method for removing the active hydrogen compound derived from the carbonic acid derivative, the reaction apparatus, etc., but it is usually carried out in the range of 0.01 Pa or more and 10 MPa or less (absolute pressure). is preferable, and considering the ease of industrial implementation, the range of 0.1 Pa or more and 5 MPa or less (absolute pressure) is preferable, and 0.1 Pa or more and 1.5 MPa or less (absolute pressure) is more preferable.
- the reaction to produce the N-substituted carbamate is often carried out mainly in the liquid phase. Therefore, the hydroxy compound and catalyst are preferably present as liquid phase components under the reaction conditions.
- the hydroxy compound and the compound having a carbonyl group derived from a carbonic acid derivative are introduced into the condenser as gas phase components and condensed in the condenser.
- the compound is also present as a gas phase component under the reaction conditions. Therefore, the reaction conditions are set so that the catalyst exists mainly as a liquid phase component, and the hydroxy compound exists partly as a liquid phase component and partly as a gaseous phase component.
- reaction conditions are set so that at least one hydroxy compound is present as a liquid phase component. Since such reaction conditions (reaction temperature, pressure) are closely related to the properties of the hydroxy compound and catalyst used, particularly the correlation between temperature and vapor pressure, the properties of the hydroxy compound and catalyst ( Correlation between temperature and vapor pressure) is measured or investigated and used as an index for determining reaction conditions. Incidentally, it is common knowledge for those skilled in the art that the correlation between the temperature and the vapor pressure of a substance varies greatly depending on the purity of the substance, coexisting compounds and their amounts. It is clear that not only the properties of the compounds and the catalyst (correlation between temperature and vapor pressure) but also the coexisting compounds and their amounts should be taken into account.
- the reaction to produce an N-substituted carbamate is an equilibrium reaction, and when one or more selected from the group consisting of urea compounds and N-unsubstituted carbamates are used as the carbonic acid derivative, the reaction is large. biased toward the original system. Therefore, in order to increase the yield of the N-substituted carbamate, it is necessary to carry out the reaction while removing by-product active hydrogen compounds out of the system as much as possible.
- the active hydrogen compound is removed so that the active hydrogen compound concentration in the reaction solution is 1000 mass ppm or less, more preferably 300 mass ppm or less, still more preferably 100 mass ppm or less, and particularly preferably 10 mass ppm or less. do.
- the method examples include a reactive distillation method, a method using an inert gas, a method using membrane separation, and a method using adsorption separation.
- the reactive distillation method is a method in which an active hydrogen compound that is successively produced under reaction is separated in a gaseous state by distillation. In order to increase the distillation efficiency of the active hydrogen compound, it can be carried out under boiling of the solvent or hydroxy compound.
- the method using an inert gas is a method in which an active hydrogen compound that is successively produced under the reaction is separated from the reaction system by entraining it in a gaseous state with an inert gas.
- Adsorbents used in the adsorption separation method include, for example, adsorbents that can be used under the temperature conditions at which the reaction is carried out, such as silica, alumina, various zeolites, and diatomaceous earths.
- the method for removing these active hydrogen compounds out of the system may be carried out singly or in combination of a plurality of methods.
- the reaction time (residence time in the case of a continuous reaction) varies depending on the composition of the reaction system, the reaction temperature, the method for removing the active hydrogen compound, the reaction apparatus, the reaction pressure, etc., but is usually 0.01 hour or more and 100 hours or less. .
- the reaction time can also be determined according to the amount of the N-substituted carbamate that is the target compound. For example, the reaction solution is sampled, the content of the N-substituted carbamate in the reaction solution is quantified, and the yield is 10% by mass or more relative to the mass of the carbonyl compound having a group derived from a primary amine compound.
- the yield of the N-substituted carbamate is preferably 50% by mass or more, more preferably 80% by mass or more, and still more preferably 90% by mass or more, based on the mass of the carbonyl compound having a group derived from a primary amine compound.
- reaction solvent it is not always necessary to use a reaction solvent, but a suitable solvent may be used for the purpose of facilitating the reaction operation.
- the reaction solvent include those exemplified in method (1A).
- the hydroxy compound used in excess in the reaction is also preferably used as the reaction solvent.
- the reaction of step (b2) has a gas phase containing a hydroxy compound, a compound having a carbonyl group derived from a carbonic acid derivative, and an active hydrogen compound by-produced in the reaction, and a liquid phase in which the reaction of step (b2) is performed. system. Most of the reactions in step (b2) are carried out in the liquid phase, but depending on the reaction conditions, the reaction may also occur in the gas phase. In this case, the volume content of the liquid phase in the reactor in which the reaction of step (b2) is carried out is preferably 50% by volume or less with respect to the volume of the gas phase. When the reaction is carried out continuously over a long period of time, polymer-like by-products may be produced due to fluctuations in operating conditions (temperature, pressure, etc.).
- the liquid phase volume content here means the reaction tank section in the case of a tank reactor, the stage below the feed stage in the case of a tower reactor (not including the bottom part and reboiler part), the thin film distiller In the case of , represents the liquid phase volume ratio to the gas phase volume in the capacity of the thin film distiller.
- the reactor used for carrying out the reaction in step (b2) is not particularly limited as long as it is a reactor equipped with a condenser, and known reactors can be used.
- One or more reactors selected from the group consisting of tower types are preferably used.
- the reaction of step (b2) includes a gas phase containing a hydroxy compound, a compound having a carbonyl group derived from a carbonic acid derivative, and an active hydrogen compound by-produced in the reaction, and the reaction of step (b2).
- the volume content of the liquid phase in the reactor is preferably 50% by volume or less with respect to the volume of the gas phase.
- the reactor for carrying out the reaction in step (b2) is also selected to meet the conditions.
- reactors such as a stirring tank, a pressurized stirring tank, a reduced pressure stirring tank, a column reactor, a distillation column, a packed column, and a thin film distiller can be used in appropriate combination as the reactor.
- the type of condenser provided in the reactor is not particularly limited, and known condensers can be used.
- conventionally known condensers such as multi-tube cylindrical condensers, double-tube condensers, single-tube condensers, and air-cooled condensers can be appropriately combined and used.
- the condenser may be provided inside the reactor, may be provided outside the reactor, or may be connected to the reactor by piping, and the type of reactor or condenser , various forms are adopted in consideration of the handling method of the condensate, and the like.
- the materials of the reactor and condenser there are no particular restrictions on the materials of the reactor and condenser, and known materials can be used.
- glass, stainless steel, carbon steel, Hastelloy, substrates with glass lining or Teflon (registered trademark) coating can be used.
- SUS304, SUS316, SUS316L, etc. are inexpensive and can be preferably used.
- instrumentation equipment such as flowmeters and thermometers, reboilers, pumps, condensers and other known process devices may be added.
- a known method such as steam or a heater may be used for heating, and a known method such as natural cooling, cooling water, or brine may be used for cooling.
- a step as described later may be added.
- the steps that can be added include, for example, a step of removing the generated active hydrogen compound, a step of purifying the primary amine compound, a step of dissolving the carbonic acid derivative in the hydroxy compound, a step of separating or purifying the hydroxy compound, and a step of separating or purifying the hydroxy compound.
- Examples include a step of purifying a carbonyl compound having a group derived from a primary amine compound from the reaction solution, a step of incinerating or discarding by-products, etc., and adding steps and devices within the range that a person skilled in the art can envision. I don't mind.
- N-substituted carbamate obtained by the above reaction is an N-substituted carbamate represented by the general formula (Va) described below when alcohol is used as the hydroxy compound. Further, when an aromatic hydroxy compound is used as the hydroxy compound, it is an N-substituted carbamate represented by general formula (Vb) described later.
- step (c1) can be further performed in the step (X1). That is, the step (X1) may further include the following step (c1).
- a step of reacting a compound having one or more functional groups selected from the group consisting of a ureylene group and a biuret group with a carbonic acid derivative in the presence of at least one amine compound having no active hydrogen as a catalyst step (c1)).
- the step (c1) is preferably carried out in the presence of a hydroxy compound.
- the hydroxy compound may be an alcohol represented by general formula (VIa) described later, or an aromatic hydroxy compound represented by general formula (VIb) described later.
- the reaction between the ureylene group-containing compound represented by formula (XIII-1) and the carbonic acid derivative is preferably carried out at 100°C or higher and 350°C or lower. At low temperatures, the efficiency of the reaction is poor, while at too high a temperature, denaturation reactions occur. Therefore, the reaction is preferably carried out at 120°C or higher and 330°C or lower, more preferably 140°C or higher and 300°C or lower.
- the amount (molar amount) of the carbonic acid derivative to be used depends on the type of carbonic acid derivative and the reaction conditions, but usually the ratio of the number of moles of the carbonic acid derivative to the number of moles of the ureylene group in the compound having the ureylene group is 10 or less. In order to increase the reaction rate and improve the efficiency of the reaction, it is preferable to use a large amount of the carbonic acid derivative. be. Therefore, the ratio of the number of moles of the carbonic acid derivative to the number of moles of the ureylene group in the compound having the ureylene group is preferably 5 or less, more preferably 3 or less.
- the amount (molar amount) of the amine compound having no active hydrogen, which is used as a catalyst is not particularly limited. It can be 1-fold mol or less, preferably 20-fold mol or less.
- the amount used is preferably 1-fold mol or less, more preferably 0.2-fold mol, relative to the molar amount of the ureylene group. 0.1 mol or less, more preferably 0.1 mol or less.
- the reaction between the compound having a ureylene group and the carbonic acid derivative is preferably carried out in the presence of a solvent.
- the solvent is not particularly limited as long as it dissolves the compound having a ureylene group and the carbonic acid derivative and is stable at the reaction temperature.
- a hydroxy compound such as an alcohol represented by the formula (VIa) or an aromatic hydroxy compound represented by the general formula (VIb) described later can be used. In particular, aromatic hydroxy compounds are preferably used.
- the step (c1) is included in the reaction solution obtained in the step (X1) after the N-substituted carbamate is produced from the primary amine compound, the carbonic acid derivative and the hydroxy compound in the step (X1) (step ( It may be carried out using a compound having a ureylene group) produced as a by-product in X1), and the reaction of a primary amine compound, a carbonic acid derivative and a hydroxy compound may be carried out simultaneously with the production of an N-substituted carbamate to produce a by-product.
- N-substituted carbamates may be prepared by reacting a compound having a ureylene group with a carbonic acid derivative. In such a case, the solvent used in step (X1) or the hydroxy compound used in excess can also be used as a solvent.
- the reaction may be carried out under any of pressurized, normal pressure, and reduced pressure conditions. Moreover, the reaction is preferably carried out in an inert gas atmosphere such as nitrogen, argon, helium, neon, or the like.
- reactors such as a stirring tank, a pressurized stirring tank, a reduced pressure stirring tank, a column reactor, a distillation column, a packed column, and a thin film distiller can be used in appropriate combination.
- a known cooling device or heating device may be installed in order to keep the reaction temperature constant.
- the material is not particularly limited, and known materials can be used.
- glass, stainless steel, carbon steel, Hastelloy, substrates with glass lining or Teflon (registered trademark) coating can be used.
- Step (X2) Gas Phase Component Condensation Step> Step (X2) includes one or more selected from the group consisting of the carbonic acid derivative and the compound having a carbonyl group derived from the carbonic acid derivative recovered in step (X1), and a hydroxy compound derived from the carbonic acid derivative.
- a gas phase component optionally containing one compound selected from the group consisting of an active hydrogen compound and an amine compound having no active hydrogen as a catalyst is added to a reactor for carrying out the carbamate synthesis step (step (X1)). It is a condensation step of condensing with a condenser provided.
- the type of condenser is not particularly limited, and known condensers can be used.
- conventionally known condensers such as multi-tube cylindrical condensers, double-tube condensers, single-tube condensers, and air-cooled condensers can be appropriately combined and used.
- the condenser may be provided inside the reactor, may be provided outside the reactor, or may be connected to the reactor by piping. Various forms are adopted in consideration of the handling method of the condensate.
- the reactor and condenser there are no particular restrictions on the materials of the reactor and condenser, and known materials can be used.
- it can be made of glass, stainless steel, carbon steel, hastelloy, or a substrate having a glass lining or a Teflon (registered trademark) coating.
- SUS304, SUS316, SUS316L, etc. are inexpensive and can be preferably used.
- instrumentation equipment such as flowmeters and thermometers, reboilers, pumps, condensers and other known process devices may be added.
- a known method such as steam or a heater may be used for heating, and a known method such as natural cooling, cooling water, or brine may be used for cooling.
- step (X2) if necessary, steps and devices within the range that a person skilled in the art can envision may be added.
- a primary amine compound, a carbonic acid derivative, and a hydroxy compound are mixed in the presence of an amine compound having no active hydrogen as a catalyst using a reactor equipped with a condenser.
- This is a method for producing an N-substituted carbamate by a carbamate reaction.
- an active hydrogen compound derived from the carbonic acid derivative and active hydrogen as a catalyst containing one or more selected from the group consisting of a carbonic acid derivative and a compound having a carbonyl group derived from the carbonic acid derivative, and a hydroxy compound.
- a gas phase component is generated which may contain one compound selected from the group consisting of amine compounds that do not have.
- this gas phase component is introduced into a condenser provided in the reactor for performing the carbamate-forming step (step (1)), and part or all of the hydroxy compound, the carbonic acid derivative and the carbonic acid derivative derived from A part or all of one or more compounds selected from the group consisting of compounds having a carbonyl group and a part or all of an amine compound having no active hydrogen, which is a catalyst, are condensed.
- the molar amount of the hydroxy compound to be condensed is preferably at least 1 mol, more preferably 2 mol in stoichiometric ratio with respect to the molar amount of the compound having a carbonyl group derived from the condensed carbonic acid derivative. It is at least 1-fold mol, more preferably at least 3-fold mol.
- the "compound having a carbonyl group derived from a carbonic acid derivative" mentioned as a component to be condensed in the condenser is the carbonic acid derivative used in the reaction of the primary amine compound, the carbonic acid derivative and the hydroxy compound.
- is a compound having a carbonyl group derived from The compound having a carbonyl group derived from a carbonic acid derivative is the carbonic acid derivative itself used as a raw material (unreacted product, and one selected from the group consisting of the excess amount when used excessively with respect to the primary amine compound. above), a compound obtained by reacting a carbonic acid derivative with a hydroxy compound, and a compound obtained by reacting a carbonic acid derivative.
- specific compounds include carbonic acid derivatives used as raw materials and by-products such as isocyanic acid, biuret, and isocyanurate.
- Examples include urea compounds, N-unsubstituted carbamates whose carbamate group is derived from a hydroxy compound, carbonate esters whose ester group is derived from a hydroxy compound, and the like.
- a compound having a carbonyl group derived from urea can be quantified by a method of detecting the carbonyl group contained in the compound by a method such as infrared spectroscopy, near-infrared spectroscopy, Raman spectroscopy, or ultraviolet spectroscopy. can.
- urea in particular requires the most attention because its production amount (detected amount) is large and its melting point is 135°C.
- the condenser is preferably maintained at a temperature at least 90° C. below the normal boiling point of the hydroxy compound and at a temperature at which the hydroxy compound does not solidify.
- the mixture of the hydroxy compound and the compound having a carbonyl group derived from the carbonic acid derivative, condensed by the condenser, is circulated inside the reactor to mix the primary amine compound, the carbonic acid derivative, and the hydroxy compound. It may be reused in the reaction, or one or more selected from the group consisting of compounds having a carbonyl group derived from a hydroxy compound and a carbonic acid derivative may be recovered from the mixture to form a primary amine compound and a carbonic acid derivative. It may be reused for the reaction with the hydroxy compound, or may be reused for the step of producing the N-unsubstituted carbamate (referring to step (c1) described above).
- the amount of the active hydrogen compound derived from the carbonic acid derivative contained in the mixture of the hydroxy compound and the compound having a carbonyl group derived from the carbonic acid derivative is preferably based on the mass of the mixture. is 5000 mass ppm or less, more preferably 3000 ppm or less, and still more preferably 2000 ppm or less.
- the content of the active hydrogen compound exceeds the above upper limit, it can be reused for the reaction of the primary amine compound, the carbonic acid derivative and the hydroxy compound.
- the reaction between and a hydroxy compound is an equilibrium reaction, and in order to proceed the reaction efficiently, it is necessary to remove the active hydrogen compound, which is a by-product, out of the system.
- the amount of the active hydrogen compound in the mixture is preferably as small as possible, it is possible to reduce the amount of the active hydrogen compound as much as possible by setting the amount to the above upper limit or less, thereby saving a great deal of labor.
- various compounds may be recovered as compounds having a carbonyl group derived from a carbonic acid derivative. Even if it contains, there is no problem in reusing the condensed component.
- amine compound having no active hydrogen one having at least one functional group selected from the group consisting of a tertiary amino group, a nitrogen-containing aromatic group, an amidine group and a guanidine group is preferably used.
- the amine compound having no active hydrogen used as a catalyst preferably has a conjugate acid with a pKa of 6 or more and 17 or less, and a pKa of 10 or more and 15 or less, from the viewpoint of promoting the carbamate reaction. More preferably, it has one or more functional groups selected from the group consisting of a tertiary amino group, an amidine group, and a guanidine group, and is selected from the group consisting of an amidine group and a guanidine group having a pKa of 12 or more and 15 or less. Further preferred are those having one or more functional groups.
- the amine compound having no active hydrogen used as a catalyst preferably has a pKa of 1 to 15, and a tertiary amino compound having a pKa of 3 to 12. and one or more functional groups selected from the group consisting of a nitrogen-containing aromatic group, and more preferably a nitrogen-containing aromatic group having a conjugate acid with a pKa of 4 or more and 7 or less.
- tertiary amino group used herein is a group represented by formula (XIV).
- a wavy line means a bond and represents a bonding site with a monovalent organic group.
- the total number of carbon atoms constituting the compound having a tertiary amino group is preferably 3 or more and 85 or less, more preferably 3 or more and 30 or less.
- the number of tertiary amino groups contained in the compound having a tertiary amino group is preferably 1 or more and 6 or less, more preferably 1 or more and 3 or less.
- Preferred amine compounds having a tertiary amino group include aromatic organic mono-tertiary amines such as N,N-dimethylaniline, N,N-diethylaniline, N-methyl-N-ethylaniline, N,N-dimethylaminopyridine; aromatic organic polytertiary amines such as N,N,N',N'-tetramethylphenylenediamine (each isomer), methylenebis(N,N-dimethylaniline) (each isomer); aliphatic mono-tertiary amines such as triethylamine, ethyldiisopropylamine, N-methylmorpholine, N-methylpiperidine, quinuclidine; N,N'-dimethylpiperazine, triethylenediamine, N,N,N'N'-tetramethylethylenediamine, N,N,N'N'-tetramethylhexanediamine, N,N,N
- nitrogen-containing aromatic group refers to a heteroaromatic group in which at least one nitrogen atom is included in atoms constituting a heteroaromatic ring.
- the compound having a heteroaromatic group is composed of nonmetallic atoms (carbon, oxygen, nitrogen, sulfur, silicon), and the total number of atoms is preferably 5 or more and 85 or less, more preferably 6 or more and 30 or less.
- the non-metallic atom is preferably selected from carbon, oxygen and nitrogen, more preferably carbon atom.
- the total number of carbon atoms constituting the compound having a nitrogen-containing aromatic group is preferably 3 or more and 85 or less, more preferably 3 or more and 30 or less.
- the number of nitrogen-containing aromatic groups contained in the compound having a nitrogen-containing aromatic group is preferably 1 or more and 5 or less, more preferably 1 or more and 3 or less.
- Preferred nitrogen-containing aromatic groups include pyridyl group, imidazolyl group, pyrazolyl group, quinolyl group, isoquinolyl group, oxazolyl group, thiazolyl group, pyridazyl group, pyrimidyl group and pyrazyl group, all of which may have a substituent. etc.
- Preferred amine compounds having nitrogen-containing aromatic groups include pyridine, picoline (each isomer), lutidine (each isomer), collidine (each isomer), 1-methylimidazole, 1-methylpyrazole, quinoline, isoquinoline, Methylquinoline (each isomer), oxazole, thiazole, pyridazine, pyrimidine, pyrazine and the like.
- a compound represented by general formula (VIII-1) is preferably used as the compound having an amidine group.
- R 811 , R 812 , R 813 and R 814 are each independently a monovalent organic group.
- R 811 and R 812 , R 812 and R 813 , R 813 and Each of R 814 , R 814 and R 811 may be independently bonded to each other to form a ring structure, and the total carbon number of R 811 , R 812 , R 813 and R 814 is 5 or more and 85 or less. be.
- Examples of the monovalent organic group for R 811 , R 812 , R 813 and R 814 include an aliphatic group, an aromatic group, a group formed by combining an aliphatic group and an aromatic group, and the above-mentioned groups. and a group bonded via a covalent bond with a non-metallic atom (carbon, oxygen, nitrogen, sulfur, silicon).
- R 811 and R 812 , R 812 and R 813 , R 813 and R 814 , R 814 and R 811 may each independently combine with each other to form a ring structure together with the nitrogen atom.
- the total number of carbon atoms constituting the compound having an amidine group is 5 or more and 85 or less, preferably 5 or more and 30 or less.
- the compound having an amidine group preferably has one or more ring structures.
- the number of amidine groups contained in the compound having an amidine group is preferably 1 or more and 3 or less, more preferably 1 or more and 2 or less.
- Preferred amidine group-containing compounds include 1,2-dimethyl-1,4,5,6-tetrahydropyrimidine, 1,8-diazabicyclo-[5.4.0]undec-7-ene (DBU), 1, 5-diazabicyclo-[4.3.0]non-5-ene (DBN) and the like.
- a compound represented by the following general formula (VIII-2) is preferably used as the compound having a guanidine group.
- R 821 , R 822 , R 823 , R 824 and R 825 are each independently a monovalent organic group.
- R 821 and R 822 , R 822 and R 823 , R 823 and R 824 , R 824 and R 825 , R 825 and R 821 may each independently combine with each other to form a ring structure, R 821 , R 822 , R 823 , R 824 , and
- the total carbon number of R825 is 5 or more and 85 or less.
- the monovalent organic groups for R 821 , R 822 , R 823 , R 824 and R 825 include, for example, an aliphatic group, an aromatic group, a group formed by combining an aliphatic group and an aromatic group, and the above-mentioned Examples include groups in which the group is bonded via a covalent bond to a specific nonmetallic atom (carbon, oxygen, nitrogen, sulfur, silicon).
- R 821 and R 822 , R 822 and R 823 , R 823 and R 824 , R 824 and R 825 , R 825 and R 821 are each independently bonded to each other to form a ring structure together with the nitrogen atom; good too.
- the total number of carbon atoms constituting the compound having a guanidine group is 5 or more and 85 or less, preferably 5 or more and 30 or less.
- the number of guanidine groups contained in the compound having a guanidine group is preferably 1 or more and 3 or less, more preferably 1 or more and 2 or less.
- Preferred compounds having a guanidine group include pentamethylguanidine, 7-methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene, and the like.
- primary amine compounds refer to "primary amines" (mono-primary amines and poly-primary amines) as defined in IUPAC Nomenclature Regulation C-8.
- Such a primary amine compound is a compound represented by the following general formula (IIb) (hereinafter sometimes referred to as “primary amine compound (IIb)").
- R 21b is an n21b-valent organic group. n21b is an integer of 1 or more and 10 or less.
- the organic group for R 21b represents an aliphatic group, an aromatic group, or a group formed by combining an aliphatic group and an aromatic group, and is an acyclic hydrocarbon group or a cyclic hydrocarbon group (e.g. , monocyclic hydrocarbon group, condensed polycyclic hydrocarbon group, bridged cyclic hydrocarbon group, spiro hydrocarbon group, ring-assembled hydrocarbon group, side-chained cyclic hydrocarbon group, heterocyclic group, heterocycle formula spiro group, hetero bridged ring group, heterocyclic group), groups in which an acyclic hydrocarbon group and a cyclic hydrocarbon group are bonded, and these groups are specific nonmetallic atoms (carbon, oxygen, nitrogen, Sulfur, silicon) represents a group bonded via a covalent bond.
- acyclic hydrocarbon group or a cyclic hydrocarbon group e.g. , monocyclic hydrocarbon group, condensed polycyclic hydrocarbon group, bridged cyclic hydro
- covalent bond with the specific nonmetallic atom is, for example, one or more selected from the group consisting of the above groups and groups represented by the following formulas (IIb-a1) to (IIb-a10) group is bonded by a covalent bond.
- R 21b is an aliphatic group, an aromatic group, or a group formed by bonding an aliphatic group and an aromatic group, and an acyclic hydrocarbon group, or , cyclic hydrocarbon groups (monocyclic hydrocarbon groups, condensed polycyclic hydrocarbon groups, bridged cyclic hydrocarbon groups, spiro hydrocarbon groups, ring-assembled hydrocarbon groups, cyclic hydrocarbon groups with side chains) , and a group in which an acyclic hydrocarbon group and a cyclic hydrocarbon group are bonded (mutually substituted groups), preferably a group having from 1 to 85 carbon atoms, and considering fluidity, etc. , more preferably a group having 1 to 70 carbon atoms, more preferably a group having 1 to 13 carbon atoms.
- Preferred aliphatic groups for R 21b include a chain hydrocarbon group, a cyclic hydrocarbon group, and a group in which a chain hydrocarbon group and a cyclic hydrocarbon group are bonded, having 5 to 70 carbon atoms (e.g., cyclic hydrocarbon group substituted with a chain hydrocarbon group, chain hydrocarbon group substituted with a cyclic hydrocarbon group, etc.).
- preferred primary amine compounds include: 1) R 21b is an aromatic group having 6 to 85 carbon atoms which may be substituted with one or more groups selected from the group consisting of aliphatic groups and aromatic groups, and n21b is 1; an aromatic primary monoamine compound; 2) R 21b is a group having 6 to 85 carbon atoms and containing at least one aromatic ring optionally substituted with at least one group selected from the group consisting of an aliphatic group and an aromatic group; aromatic primary polyamine compounds in which n21b is 2 or more and the aromatic groups in R21b are substituted with NH2 groups; 3) an aliphatic primary (mono- or poly)amine compound in which R 21b is an aliphatic group having 1 to 85 carbon atoms and optionally substituted with an aromatic group; is.
- an atom (preferably a carbon atom) to which the NH2 group is bonded is represented as an aromatic amine compound that is contained in an aromatic ring, and is bonded to an atom (mainly carbon) that is not an aromatic ring.
- an aliphatic amine compound When there is, it is described as an aliphatic amine compound.
- the primary amine compound an aromatic primary polyamine compound or an aliphatic primary polyamine compound is preferable.
- Aromaatic primary monoamine compound Preferred aromatic primary monoamine compounds are compounds represented by the following general formula (IIb-1).
- ring A 211b is an aromatic hydrocarbon ring having 6 to 20 carbon atoms; R 211b is a hydrogen atom or an alkyl optionally substituted with an aromatic group; A cycloalkyl group, an aryl group, or a group linked via a covalent bond between these groups and certain non-metallic atoms (carbon, oxygen, nitrogen, sulfur, silicon).R 211b is ring A 211b to form a ring structure, and n211b is an integer of 1 or more and 10 or less.
- Ring A 211b is an aromatic hydrocarbon ring having 6 to 20 carbon atoms. Ring A 211b may be monocyclic, polycyclic, or condensed.
- ring A 211b examples include benzene ring, naphthalene ring, anthracene ring, phenanthrene ring, naphthacene ring, chrysene ring, pyrene ring, triphenylene ring, pentalene ring, azulene ring, heptalene ring, indacene ring, biphenylene ring, acenaphthylene ring, aceanthrylene ring, acephenanthrylene ring, and the like.
- the ring A 11 is preferably a benzene ring, a naphthalene ring, or an anthracene ring, more preferably a benzene ring.
- R211b When n211b is 2 or more, multiple R211b may be the same or different.
- the total carbon number of multiple R 211b is preferably 1 or more and 64 or less, and more preferably 0 or more and 7 or less in consideration of fluidity and the like.
- the total number of carbon atoms constituting the aromatic primary monoamine compound represented by the general formula (IIb-1) is 6 or more and 13 or less.
- n211b represents the number of R 211b and is an integer of 1 or more and 10 or less, preferably an integer of 0 or more and 4 or less, more preferably an integer of 0 or more and 3 or less, and still more preferably an integer of 0 or more and 2 or less.
- At least one selected from the group consisting of the ortho-position and para-position of the NH 2 group of the aromatic primary monoamine compound represented by the general formula (IIb-1) is unsubstituted, ie, is a hydrogen atom.
- R 211b is a hydrogen atom, a methyl group, or an ethyl group.
- preferred aromatic primary monoamine compounds include aniline, aminotoluene (each isomer), dimethylaniline (each isomer), diethylaniline (each isomer), dipropylaniline (each isomer), Aminonaphthalene (each isomer), aminomethylnaphthalene (each isomer), dimethylnaphthylamine (each isomer), trimethylnaphthylamine (each isomer), and the like.
- R 21b in the primary amine compound represented by the above general formula (IIb) is substituted with one or more groups selected from the group consisting of aliphatic groups and aromatic groups. an aromatic group having 6 or more and 85 or less carbon atoms containing one or more aromatic rings which may be optionally substituted, wherein n21b is 2 or more, and the aromatic group in R 21b is substituted with an NH 2 group. It is a primary polyamine compound.
- R 21b is a group having 6 or more and 13 or less carbon atoms which may be substituted with an alkyl group, an aryl group, or an aralkyl group, n21b is 2 or more and 8 or less, Also, it is an aromatic primary polyamine compound in which the aromatic group in R21b is substituted with NH2 group.
- aromatic primary polyamine compounds include diaminobenzene (each isomer), diaminotoluene (each isomer), methylenedianiline (each isomer), diaminomesitylene (each isomer), diaminobiphenyl ( isomer), diaminodibenzyl (each isomer), bis(aminophenyl)propane (each isomer), bis(aminophenyl)ether (each isomer), bis(aminophenoxyethane) (each isomer), Diaminoxylene (each isomer), diaminoanisole (each isomer), diaminophenetole (each isomer), diaminonaphthalene (each isomer), diamino-methylbenzene (each isomer), diamino-methylpyridine (each isomer) isomer), diamino-methylnaphthalene (each isomer), polym
- n212b is an integer of 0 or more and 6 or less.
- R 21b in the primary amine compound represented by the general formula (IIb) may be substituted with an aromatic group having 1 to 85 carbon atoms.
- Preferred aliphatic primary amine compounds include an acyclic aliphatic hydrocarbon group, a cyclic aliphatic (i.e., alicyclic) hydrocarbon group, and an acyclic aliphatic hydrocarbon group in which R 21b has 1 to 70 carbon atoms.
- More preferred aliphatic primary amine compounds include an acyclic aliphatic hydrocarbon group, a cyclic aliphatic (alicyclic) hydrocarbon group, and an acyclic aliphatic hydrocarbon group in which R 21b has 1 to 70 carbon atoms.
- a more preferable aliphatic primary amine compound is one in which R 21b is composed of carbon atoms and hydrogen atoms and has 5 or more and 13 or less carbon atoms.
- R 21b is an acyclic aliphatic hydrocarbon group having 5 or more and 13 or less carbon atoms, composed of carbon atoms and hydrogen atoms, and a specific nonmetallic atom (carbon , oxygen, nitrogen) and is an aliphatic primary amine compound in which n21b is 2 or 3.
- preferred aliphatic primary amine compounds include: Ethylenediamine, diaminopropane (each isomer), diaminopentane (each isomer), diaminohexane (each isomer), diaminodecane (each isomer), xylylenediamine (each isomer), diaminocyclobutane (each isomer) , diaminocyclohexane (each isomer), bis(diaminomethyl)cyclohexane, 3-aminomethyl-3,5,5-trimethylcyclohexylamine (cis and/or trans isomer), methylenebis(cyclohexylamine) (each isomer), etc.
- aliphatic primary diamines of aliphatic primary diamines of; Aliphatic primary triamines such as triaminohexane (each isomer), triaminononane (each isomer), triaminodecane (each isomer); Amino acid derivatives such as lysine methyl ester and lysine aminoethyl ester; etc.
- a carbonic acid derivative in the present specification is a component used as a raw material for producing a carbamate compound together with a primary amine compound and a hydroxy compound.
- Specific examples of carbonic acid derivatives include urea compounds, N-unsubstituted carbamates, carbonate esters, and the like.
- a urea compound as used herein is a compound having at least one urea bond in the molecule.
- a preferred urea compound is a compound having one urea bond, and specific examples thereof include compounds represented by the following general formula (III-2).
- R 321 , R 322 , R 323 and R 324 are each independently an organic group having 1 to 20 carbon atoms or a hydrogen atom.
- the sum of R 321 and R 322 The number of carbon atoms is an integer of 0 or more and 20 or less, and the total number of carbon atoms of R 323 and R 324 is an integer of 0 or more and 20 or less.
- Examples of organic groups having 1 to 20 carbon atoms in R 321 , R 322 , R 323 and R 324 include: Methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), pentyl group (each isomer), hexyl group (each isomer), heptyl group (each isomer), octyl group (each isomer) and other alkyl groups; Phenyl group, methylphenyl group (each isomer), biphenyl group (each isomer), dimethylphenyl group (each isomer), diethylphenyl group (each isomer), terphenyl group (each isomer), trimethylphenyl group (each isomer), triethylphenyl group (each isomer), tripropylphenyl group
- Aryl groups having 6 to 20 carbon atoms constituting the group an aralkyl group having 7 or more and 20 or less carbon atoms constituting the group, such as a phenylmethyl group and a phenylethyl group (each isomer); etc.
- urea compounds include urea, methylurea, and ethylurea.
- urea in which all of R 321 , R 322 , R 323 and R 324 in general formula (III-2) are hydrogen atoms is preferred.
- Urea may include biuret, triuret, and cyanuric acid.
- biuret, triuret and cyanuric acid generally have low solubility in solvents. Since it is preferable to use a uniform solution when transferring the raw materials and the reaction solution, it is preferable that the content of these components is as low as possible.
- urea has a melting point of 135° C. and is solid when handled at room temperature, but its shape is not particularly limited, and for example, powder or granules can be used.
- N-unsubstituted carbamate As the N-unsubstituted carbamate, a compound represented by the following general formula (III-3) is preferably used.
- R 331 is an organic group having 1 to 20 carbon atoms.
- R331 Examples of the organic group having 1 to 20 carbon atoms for R 331 include the same groups as those exemplified for R 321 , R 322 , R 323 and R 324 above.
- N-unsubstituted carbamate examples include methyl carbamate, ethyl carbamate, propyl carbamate, butyl carbamate, and phenyl carbamate.
- carbonic acid ester refers to a compound in which one or two of the two hydrogen atoms of carbonic acid CO(OH) 2 are substituted with an aliphatic or aromatic group.
- carbonate ester a compound represented by the following general formula (III-1b) is preferably used.
- R 311b and R 312b each independently represent an organic group having 1 to 20 carbon atoms.
- R 311b and R 312b examples of the organic group having 1 to 20 carbon atoms are the same as those exemplified for R 321 , R 322 , R 323 and R 324 above.
- carbonate esters include dimethyl carbonate, diethyl carbonate, and diphenyl carbonate.
- a hydroxy compound refers to a compound having a hydroxy group (--OH group).
- the hydroxy compound one or more compounds selected from the group consisting of alcohols and aromatic hydroxy compounds, which are compounds in which a hydroxy group is bonded to a carbon atom, are preferably used.
- alcohol refers to compounds in which a hydroxy group, -OH, is attached to a saturated carbon atom: R 3 COH)” and is a hydroxy compound represented by the following general formula (VIa).
- R 61a is an n61a-valent aliphatic group. However, R 61a is a group having no active hydrogen other than a hydroxy group. n61a is an integer of 1 or more and 5 or less. show.)
- the aliphatic group for R 61a may be bonded with an aliphatic group having 1 to 50 carbon atoms or an aromatic group having 7 to 50 carbon atoms as a substituent.
- the aliphatic group for R 61a is an aliphatic hydrocarbon group in which atoms other than hydrogen atoms constituting the aliphatic group are specific nonmetallic atoms (carbon, oxygen, nitrogen, sulfur, silicon, halogen atoms).
- Preferred aliphatic groups include a chain aliphatic hydrocarbon group, a cyclic aliphatic (alicyclic) hydrocarbon group, and a combination of a chain aliphatic hydrocarbon group and a cyclic aliphatic (alicyclic) hydrocarbon group. groups (for example, a cyclic hydrocarbon group substituted with a chain hydrocarbon group, a chain hydrocarbon group substituted with a cyclic hydrocarbon group, etc.), and the like.
- the aliphatic group to which the aromatic group is bonded includes a chain or branched alkyl group, an aliphatic group in which the cycloalkyl group is substituted with an aromatic group, and an alkyl group having 1 to 44 carbon atoms. is a group in which an aromatic group having 6 or more and 49 or less carbon atoms is bonded to As described above, the aromatic group is preferably an aromatic group in which atoms other than hydrogen atoms constituting the aromatic group are specific nonmetallic atoms (carbon, oxygen, nitrogen, sulfur, silicon, halogen atoms).
- a monocyclic aromatic group a condensed polycyclic aromatic group, a bridged cyclic aromatic group, a ring-assembled aromatic group, a heterocyclic aromatic group, etc., more preferably substituted or an unsubstituted phenyl group, a substituted or unsubstituted naphthyl group, or a substituted or unsubstituted anthryl group.
- R 61a Methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), pentyl group (each isomer), hexyl group (each isomer), heptyl group (each isomer), octyl group ( isomer), nonyl group (each isomer), decyl group (each isomer), dodecyl group (each isomer), octadecyl group (each isomer), cyclopentyl group, cyclohexyl group, methylcyclopentyl group (each isomer) ), methylcyclohexyl group (each isomer), dimethylcyclohexyl group (each isomer), diethylcyclohexyl group (each isomer), substituted or unsubstituted alkyl and cycloalkyl groups; Ar
- alcohols considering industrial use, they generally have low viscosity, so one or two alcoholic hydroxy groups (constituting hydroxy compounds, directly attached to carbon atoms other than aromatic rings hydroxy groups) are preferred, monoalcohols with one alcoholic hydroxy group are more preferred.
- alcohol Methanol, ethanol, propanol (each isomer), butanol (each isomer), pentanol (each isomer), hexanol (each isomer), heptanol (each isomer), octanol (each isomer), nonanol ( isomers), decanol (each isomer), dodecanol (each isomer), octadecanol (each isomer), cyclopentanol, cyclohexanol, cycloheptanol, cyclooctanol, methylcyclopentanol (each isomer) isomer), ethylcyclopentanol (each isomer), methylcyclohexanol (each isomer), ethylcyclohexanol (each isomer), propylcyclohexanol (each isomer),
- alkyl alcohols having 1 to 20 carbon atoms are preferably used from the viewpoint of availability, solubility of raw materials and products, and the like.
- aromatic hydroxy compound refers to the phenols described in the IUPAC definition (Rule C-202), "one or more hydroxy groups on a benzene ring or other arene ring. It is a compound having one or more hydroxy groups attached to a benzene or other arena ring, and is a compound represented by the following general formula (VIb).
- ring A 61b is an aromatic hydrocarbon ring having 6 to 20 carbon atoms; R 61b is a hydrogen atom or a monovalent organic group; n61b is 1 to 6; is an integer.
- the monovalent organic group for R 61b includes a halogen atom, an aliphatic group, an aromatic group, and a group in which these groups are bonded. More specifically, acyclic hydrocarbon groups, cyclic hydrocarbon groups (e.g., monocyclic hydrocarbon groups, condensed polycyclic hydrocarbon groups, bridged cyclic hydrocarbon groups, spiro hydrocarbon groups, ring assembly hydrocarbon group, cyclic hydrocarbon group with a side chain, heterocyclic group, heterocyclic spiro group, hetero-bridging ring group, heterocyclic group), acyclic hydrocarbon group and cyclic hydrocarbon group bonded groups and groups to which these groups are attached via covalent bonds to certain non-metallic atoms (carbon, oxygen, nitrogen, sulfur, silicon).
- acyclic hydrocarbon groups e.g., monocyclic hydrocarbon groups, condensed polycyclic hydrocarbon groups, bridged cyclic hydrocarbon groups, spiro hydrocarbon groups, ring assembly hydrocarbon group, cyclic hydro
- n61b represents the number of aromatic hydroxy groups bonded to ring A 61b , and is preferably 1 or 2, more preferably 1, because the viscosity is generally low. That is, aromatic di- or mono-hydroxy compounds are preferred, and aromatic mono-hydroxy compounds are more preferred.
- Ring A 61b is an aromatic hydrocarbon ring having 6 or more and 20 or less carbon atoms. Ring A 61b may be monocyclic, polycyclic, or condensed.
- ring A 61b examples include benzene ring, naphthalene ring, anthracene ring and the like.
- a benzene ring which is easily available, is preferable from the viewpoint of industrial use.
- aromatic hydroxy compounds include aromatic monohydroxy compounds represented by the following general formula (VIb-1).
- R 611b , R 612b , R 613b , R 614b and R 615b are each independently the same as R 61b above.
- R 611b , R 612b , R 613b , R 614b and R 615b preferred R 611b , R 612b , R 613b , R 614b and R 615b include alkyl groups, cycloalkyl groups, aryl groups, aralkyl groups and ether groups (substituted or unsubstituted, alkyl ethers, aryl ethers, or aralkyl ethers); groups to which two or more of these groups are attached; these groups through covalent bonds with certain nonmetallic atoms (carbon, oxygen, nitrogen, sulfur, silicon) a group bound by a group; a halogen atom; or a hydrogen atom.
- nonmetallic atoms carbon, oxygen, nitrogen, sulfur, silicon
- the total carbon number of R 611b , R 612b , R 613b , R 614b and R 615b is an integer of 0 or more and 44 or less.
- R 611b , R 612b , R 613b , R 614b and R 615b are each independently one or more groups selected from the group consisting of groups (i) to (v) below.
- ⁇ -position atom as used herein means that among the atoms constituting R 611b , R 612b , R 613b , R 614b and R 615b, R 611b , R 612b , R 613b , R 614b and R 615b are Refers to the atoms adjacent to the carbon atoms on the attached aromatic hydrocarbon ring.
- aralkyloxy group in this specification represents a group in which an oxygen atom is bonded to the previously defined aralkyl group.
- R 611b , R 612b , R 613b , R 614b and R 615b are specifically: Methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), cyclopentyl group, cyclohexyl group, methylcyclopentyl group (each isomer), ethylcyclopentyl group (each isomer), methylcyclohexyl group ( isomers), dimethylcyclohexyl group (each isomer), diethylcyclohexyl group (each isomer), substituted or unsubstituted alkyl groups and cycloalkyl groups; methoxy group, ethoxy group, propoxy group (each isomer), butyloxy group (each isomer), cyclopentyloxy group, cyclohexyloxy group, methylcyclopentyloxy group (each isomer),
- Preferred aromatic hydroxy compounds include, for example, the following compounds. Phenol, chlorophenol (each isomer), bromophenol (each isomer), dichlorophenol (each isomer), dibromophenol (each isomer), trichlorophenol (each isomer), tribromophenol (each isomer) , phenol, methylphenol (each isomer), ethylphenol (each isomer), propylphenol (each isomer), butylphenol (each isomer), pentylphenol (each isomer), hexylphenol (each isomer), heptyl Phenol (each isomer), Octylphenol (each isomer), Nonylphenol (each isomer), Cumylphenol (each isomer), Dimethylphenol (each isomer), Diethylphenol (each isomer), Isopropylmethylphenol ( isomers), trimethylphenol (each
- aromatic hydroxy compounds in which the total number of carbon atoms of R 611b , R 612b , R 613b , R 614b and R 615b is 0 or more and 13 or less are preferable because they are easy to transport.
- the total carbon number of R 611b , R 612b , R 613b , R 614b and R 615b is 0 or more and 9 or less, and each of R 611b , R 612b , R 613b , R 614b and R 615b is independently a hydrogen atom, linear or branched alkyl group, cycloalkyl group, substituted or unsubstituted aryl group, linear or branched alkoxy group, substituted or unsubstituted aryloxy group, or substituted or unsubstituted An aromatic hydroxy compound that is an aralkyl group is more preferred.
- Carbamate compounds obtained by the production method of the present embodiment include N-substituted carbamates.
- N-Substituted Carbamate Compound (Va) In the reaction of a primary amine compound, urea and a hydroxy compound, the N-substituted carbamate obtained when alcohol is used as the hydroxy compound is a compound represented by the following general formula (Va) (hereinafter referred to as "N-substituted (sometimes referred to as “carbamate compound (Va)").
- R 51a is an n51a-valent organic group and is a group derived from a primary amine compound. That is, R 51a is the same as R 21b above.
- R 52a is , is an alcohol-derived group.That is, R 52a is the same as R 61a above.n51a is the same as n21b above.
- Preferred N-substituted carbamate compounds (Va) are 1) N-aromatic monocarbamate compounds in which R 51a is a group containing at least one aromatic ring and having 6 to 85 carbon atoms and n51a is 1; 2) N-aromatic polycarbamate compounds in which R 51a is a group containing at least one aromatic ring and having 6 to 85 carbon atoms and n51a is 2 or more; 3) N-aliphatic polycarbamate compounds in which R 51a is an aliphatic group having 1 or more and 85 or less carbon atoms, which may be substituted with an aromatic group, and n51a is 2 or more; is.
- N-substituted carbamate compound (Va) Specific examples of the N-substituted carbamate compound (Va) are shown below.
- N-aromatic monocarbamate compound A preferable N-aromatic monocarbamate compound is an N-aromatic monocarbamate compound represented by the following general formula (Va-1).
- R 511a is the same as R 211b above.
- n511a is the same as n211b above.
- R 512a is the same as R 61a above.
- Ring A 511a is the same as R 61a above. Same as Ring A 211b .
- R 51a in the general formula (Va) is a group having 6 or more and 85 or less carbon atoms containing one or more aromatic rings, and n51a is 2 or more.
- - is an aromatic polycarbamate compound.
- R 51a in the general formula (Va) may be substituted with an aliphatic group or an aromatic group, and contains at least one aromatic ring and has 6 to 13 carbon atoms. and n51a is 2 or more, N-aromatic polycarbamate compounds are preferred.
- N-aromatic polycarbamate compounds include polymethylene polyphenyl polycarbamate represented by the following general formula (Va-2).
- R 521a is the same as R 61a above.
- n521a is the same as n212b above and is an integer of 0 or more and 6 or less.
- N-aliphatic polycarbamate compound In the N-aliphatic polycarbamate compound, R 51a in the general formula (Va) is an aliphatic group having 1 or more and 85 or less carbon atoms, which may be substituted with an aromatic group, and n51a is 2 or more. It is an N-aliphatic polycarbamate compound.
- the aliphatic group in R 51a is a chain aliphatic hydrocarbon group, a cyclic aliphatic hydrocarbon group, and a chain aliphatic hydrocarbon group and a cyclic aliphatic hydrocarbon group are bonded. groups, or groups in which these groups are bonded to specific nonmetallic atoms (carbon, oxygen, nitrogen, sulfur, silicon) via covalent bonds, and N-aliphatic polycarbamates having n51a of 2 or more Compounds are preferred.
- the aliphatic group in R 51a is an acyclic aliphatic hydrocarbon group having 1 to 70 carbon atoms, a cyclic aliphatic hydrocarbon group, an acyclic aliphatic hydrocarbon group and a cyclic aliphatic hydrocarbon group or a group to which these groups are bonded via a covalent bond with a specific nonmetallic atom (carbon, oxygen, nitrogen, sulfur, silicon), and n51a is 2 or more and 5 or less N-aliphatic polycarbamate compounds are more preferred.
- N-aliphatic polycarbamate compounds it is not possible to list all of them because they are determined by the types of primary amine compounds and alcohols used. , N,N'-pentanediyl-di(carbamic acid ethyl ester) (each isomer), N,N'-pentanediyl-di(carbamic acid propyl ester) (each isomer), N,N'-pentanediyl-di( carbamic acid butyl ester) (each isomer), N,N'-pentanediyl-di(carbamic acid pentyl ester) (each isomer), N,N'-pentanediyl-di(carbamic acid hexyl ester) (each isomer) , N,N'-pentanediyl-di(carbamic acid heptyl ester) (each isomer), N,N
- N-substituted carbamate compound (Vb) The N-substituted carbamate obtained when an aromatic hydroxy compound is used as the hydroxy compound in the reaction of the primary amine compound, urea and the hydroxy compound is a compound represented by the following general formula (Vb) (hereinafter referred to as " N-substituted carbamate compound (Vb)”).
- R 51b is an n51b-valent organic group and is a group derived from a primary amine compound. That is, R 51b is the same as R 21b above.
- R 52b is , is a group derived from an aromatic hydroxy compound.That is, R 52b is the same as R 61b above.n51b is the same as n21b above.
- N-substituted carbamate compounds include 1) N-aromatic monocarbamate compounds in which R 51b is an aromatic group having 6 to 85 carbon atoms which may be substituted with an aliphatic group or an aromatic group, and n51b is 1; 2) R 51b is a group containing one or more aromatic rings optionally substituted with an aliphatic group or an aromatic group and having 6 or more and 85 or less carbon atoms, and n51b is 2 or more; N-aromatic polycarbamate compound; 3) N-aliphatic polycarbamate compounds in which R 51b is an aliphatic group having 1 or more and 85 or less carbon atoms, which may be substituted with an aromatic group, and n51b is 2 or more; is.
- N-substituted carbamate compound (Vb) Specific examples of the N-substituted carbamate compound (Vb) are shown below.
- N-aromatic monocarbamate compound A preferred N-aromatic monocarbamate compound is an N-aromatic monocarbamate compound represented by the following general formula (Vb).
- R 511b is the same as R 211b above.
- n511b is the same as n211b above.
- R 512b is the same as R 61b above.
- Ring A 511b is the same as R 61b above. Same as Ring A 61b .
- N-aromatic polycarbamate compound In the N-aromatic polycarbamate compound, R 51b in the above general formula (Vb) may be substituted with an aliphatic group or an aromatic group, contains one or more aromatic rings, and has 6 to 85 carbon atoms. and n51b is 2 or more, it is an N-aromatic polycarbamate compound.
- the R 51b group in the general formula (Vb) is a group having 6 or more and 13 or less carbon atoms, which may be substituted with an alkyl group, an aryl group, or an aralkyl group, and , n51b of 2 or more and 8 or less.
- N-aromatic polycarbamate compounds include polymethylene polyphenyl polycarbamate represented by the following general formula (Vb-2).
- R 521b is the same as R 61b above.
- n521b is the same as 212b above and is an integer of 0 or more and 6 or less.
- N-aliphatic polycarbamate compound In the N-aliphatic polycarbamate compound, R 51b in the general formula (Vb) may be substituted with an aliphatic group or an aromatic group, contains one or more aromatic rings, and has 6 to 85 carbon atoms. and n51b is 2 or more, N-aromatic polycarbamate compounds.
- the aliphatic group in R 51b is an acyclic aliphatic hydrocarbon group having 1 or more and 70 or less carbon atoms, a cyclic aliphatic hydrocarbon group, or an acyclic aliphatic hydrocarbon A group in which a group and a cycloaliphatic hydrocarbon group are bonded, or a group in which these groups are bonded via a covalent bond to a specific nonmetallic atom (carbon, oxygen, nitrogen, sulfur, silicon),
- N-aliphatic polycarbamate compounds having n51b of 2 or more are preferred.
- R 51b is an acyclic aliphatic hydrocarbon group having 1 to 70 carbon atoms, a cyclic aliphatic hydrocarbon group, an acyclic aliphatic hydrocarbon group and a cyclic aliphatic hydrocarbon group bonded N-aliphatic groups, or groups in which these groups are bonded via covalent bonds to specific nonmetallic atoms (carbon, oxygen, nitrogen, sulfur, silicon) and n51b is 2 or more and 5 or less Polycarbamate compounds are more preferred.
- R 51b is an acyclic aliphatic hydrocarbon group composed of carbon atoms and hydrogen atoms and having 5 or more and 13 or less carbon atoms, a cyclic An aliphatic hydrocarbon group, a group in which an acyclic aliphatic hydrocarbon group and a cyclic aliphatic hydrocarbon group are bonded, or these groups are specific nonmetallic atoms (carbon, oxygen, nitrogen, sulfur, silicon) N-aliphatic polycarbamate compounds in which n51b is 2 or more and 4 or less are more preferable.
- N-aliphatic polycarbamate compounds it is not possible to list all of them because they are determined by the types of primary amine compounds and aromatic hydroxy compounds used.
- phenyl ester N,N'-pentanediyl-di(carbamate methylphenyl ester), N,N'-pentanediyl-di(carbamate ethylphenyl ester), N,N'-pentanediyl-di(carbamate propylphenyl ester) ), N,N′-pentanediyl-di(carbamate butylphenyl ester), N,N′-pentanediyl-di(carbamate pentylphenyl ester), N,N′-pentanediyl-di(carbamate hexylphenyl ester), N,N'-pentanediyl-di(carbamic acid dimethylphenyl ester
- the method for producing an isocyanate compound of the present embodiment includes a thermal decomposition step of thermally decomposing the carbamate compound obtained by the above-described production method.
- Thermal decomposition process In the thermal decomposition step, the carbamate compound obtained by the method for producing a carbamate compound described above is thermally decomposed.
- the reaction temperature for thermally decomposing the carbamate compound is usually in the range of 100° C. or higher and 300° C. or lower, and a high temperature is preferable in order to increase the reaction rate.
- One or more compounds selected from the group may cause side reactions. Therefore, it is preferably in the range of 150° C. or higher and 280° C. or lower.
- the reactor may be equipped with a known cooling device or heating device.
- the reaction pressure varies depending on the type of compound used and the reaction temperature, but may be reduced pressure, normal pressure, or increased pressure, and is usually carried out in the range of 20 Pa or more and 1 ⁇ 10 6 Pa or less.
- reaction time is not particularly limited, and is usually 0.001 to 100 hours, preferably 0.005 to 50 hours, more preferably 0.01 to 10 hours. is.
- reaction solvent other than the hydroxy compound
- a suitable solvent can be used for purposes such as facilitating the reaction operation.
- the reaction solvent include those exemplified in method (1A).
- Carbamate compounds may cause side reactions when kept at high temperatures for a long time. Therefore, it is preferable that the time during which the carbamate compound and the isocyanate compound are held at high temperature be as short as possible.
- the pyrolysis reaction is therefore preferably carried out in a continuous process.
- the continuous method is a method in which a mixture containing a carbamate compound is continuously supplied to a reactor, subjected to a thermal decomposition reaction, and the resulting isocyanate compound and hydroxy compound are continuously withdrawn from the thermal decomposition reactor. be.
- the low boiling point components produced by the thermal decomposition reaction of the carbamate compound are preferably recovered from the upper part of the thermal decomposition reactor as gas phase components, and the rest are recovered from the bottom of the thermal decomposition reactor as liquid phase components.
- all the compounds present in the pyrolysis reactor can be recovered as gaseous phase components, by allowing liquid phase components to exist in the pyrolysis reactor, 1 selected from the group consisting of carbamate compounds and isocyanate compounds It has the effect of dissolving polymeric compounds produced by side reactions caused by more than one kind of compound and preventing adhesion and accumulation of polymeric compounds on the thermal decomposition reactor.
- An isocyanate compound and a hydroxy compound are produced by the thermal decomposition reaction of the carbamate compound, and at least one of these compounds is recovered as a gas phase component. Which compound is recovered as a gas phase component depends on the thermal decomposition reaction conditions and the like.
- a known distillation apparatus is preferably used in order to efficiently recover gas phase components.
- a variety of known methods are used, such as a method using a reactor containing any of these vessels, and a method combining these. From the viewpoint of quickly removing low boiling point components from the reaction system, a method using a tubular reactor, more preferably a reactor such as a tubular thin film evaporator or a tubular falling film evaporator, is used.
- a structure with a large gas-liquid contact area that can be quickly transferred to the gas phase is preferred.
- the material of the pyrolysis reactor and lines may be of any known type as long as it does not adversely affect the carbamate compound and products such as hydroxy compounds and isocyanate compounds.
- SUS304, SUS316, SUS316L, etc. are inexpensive and can be preferably used.
- the recovered isocyanate compound may contain hydroxy compounds, etc., depending on the reaction conditions, the conditions for recovering the isocyanate compound, the reactor, etc. In such a case, further operations such as distillation can be performed to obtain the isocyanate compound of desired purity.
- the isocyanate compound After obtaining the isocyanate compound by thermal decomposition of the carbamate compound, the isocyanate compound may be purified. It is preferable to separate or remove the catalyst used in synthesizing the carbamate compound between the pyrolysis reaction and the purification. In a state where the isocyanate compound and the above catalyst coexist, undesirable phenomena such as modification or discoloration of the isocyanate compound may occur. Isocyanate-modified products cause clogging of piping, making continuous operation extremely difficult.
- the catalyst composed of an amine compound having no active hydrogen which is used in the method for producing a carbamate compound of the present embodiment, is easier to remove than an inorganic base, and is effective in suppressing the modification of the isocyanate compound in the thermal decomposition step of the carbamate compound. target.
- the low-boiling point components produced by the thermal decomposition reaction of carbamate compounds are mainly hydroxyl compounds
- a catalyst with a higher vapor pressure than the hydroxyl compounds is used in the production of carbamate compounds, there is no need to install a process for removing the catalyst.
- the catalyst can be separated from the isocyanate compound together with the hydroxy compound, which is economical.
- the low boiling point components generated by the thermal decomposition reaction of the carbamate compound are mainly isocyanate compounds
- the generated isocyanate compound can be quickly released from the catalyst. Since it is separated, it is effective in suppressing the modification of isocyanate.
- low-boiling-point component produced by the thermal decomposition reaction of the carbamate compound corresponds to one or more compounds selected from the group consisting of hydroxy compounds and isocyanate compounds produced by the thermal decomposition reaction of the carbamate compound. refers in particular to compounds that may exist as a gas under the conditions under which the pyrolysis reaction is carried out.
- the method for producing an isocyanate compound according to the present embodiment may optionally include other steps in addition to the thermal decomposition step.
- Other steps include, for example, a step of pre-concentrating by separating part or all of the hydroxy compound from the reaction solution containing the carbamate compound obtained by the above production method before the thermal decomposition step (“carbamate concentration step ”); a step of purifying the isocyanate compound; and the like.
- isocyanate compound (VIIc) As the isocyanate compound obtained by the method for producing an isocyanate compound of the present embodiment, a compound represented by the following general formula (VIIc) (hereinafter sometimes referred to as "isocyanate compound (VIIc)”) is preferably used.
- R 71c is an n71c-valent organic group. n71c is an integer of 1 or more and 12 or less.
- R 71c is an n71c valent organic group. That is, it is an organic group having a valence of 1 or more and 12 or less. Among them, R 71c is preferably an organic group consisting of a carbon atom, an oxygen atom and a hydrogen atom, and more preferably an organic group having no active hydrogen.
- the aliphatic hydrocarbon group for R 71c includes an alkylene group or an alkanetriyl group, a cycloalkyl group, a cycloalkylene group or a cycloalkanetriyl group, or the alkyl group, the alkylene group or the alkanetriyl group, A group composed of the cycloalkyl group, the cycloalkylene group or the cycloalkanetriyl group is preferable, and a linear or branched alkylene group or alkanetriyl group, cycloalkylene group or cycloalkanetriyl group , or a group composed of the alkylene group or the alkanetriyl group and the cycloalkyl group, the cycloalkylene group or the cycloalkanetriyl group is more preferable.
- linear or branched alkylene groups examples include methylene group, ethylene group, propylene group, trimethylene group, pentylene group, n-hexylene group and decamethylene group.
- the cycloalkylene group includes, for example, a cyclobutylene group, a cyclohexylene group, and the like.
- linear or branched alkanetriyl groups examples include hexanetriyl, nonanetriyl, and decantriyl groups.
- the cycloalkanetriyl group includes, for example, a cyclopropanetriyl group, a cyclobutanetriyl group, a cyclopentanetriyl group, a cyclohexanetriyl group, and the like.
- the aromatic hydrocarbon group for R 71c is preferably a substituted or unsubstituted group having an aromatic ring having 6 to 13 carbon atoms.
- substituents include alkyl groups, aryl groups, and aralkyl groups.
- the aromatic ring may be an aromatic hydrocarbon ring or a heteroaromatic ring, and specific examples thereof include benzene ring, naphthalene ring, pyridine ring and the like.
- Preferred isocyanate compounds (VIIc) include monofunctional isocyanate compounds, bifunctional isocyanate compounds, and polyfunctional isocyanate compounds.
- Examples of monofunctional isocyanate compounds include methyl isocyanate, ethyl isocyanate, propyl isocyanate (each isomer), butyl isocyanate (each isomer), pentyl isocyanate (each isomer), hexyl isocyanate (each isomer), octyl isocyanate ( isomer), nonyl isocyanate (each isomer), decyl isocyanate (each isomer), undecyl isocyanate (each isomer), dodecyl isocyanate (each isomer), tridecyl isocyanate (each isomer), tetradecyl isocyanate (each isomer), pentadecyl isocyanate (each isomer), hexadecyl isocyanate (each isomer), heptadecyl isocyanate (each isomer), oc
- bifunctional isocyanate compounds include dimethylene diisocyanate, trimethylene diisocyanate (each isomer), tetramethylene diisocyanate (each isomer), pentamethylene diisocyanate (each isomer), hexamethylene diisocyanate (each isomer), hepta Methylene diisocyanate (each isomer), octamethylene diisocyanate (each isomer), diisocyanatocyclohexane (each isomer), bisisocyanatomethylcyclohexane (each isomer), isophorone diisocyanate (each isomer), dicyclohexylmethane diisocyanate (each isomer), diisocyanatodimethylpropane (each isomer), diisocyanatodimethylpentane (each isomer), diisocyanatodimethylhexane (each isomer),
- polyfunctional isocyanates examples include propane triisocyanate, butane triisocyanate (each isomer), pentane triisocyanate (each isomer), hexane triisocyanate (each isomer), heptane triisocyanate (each isomer), octane triisocyanate (each isomer), nonane triisocyanate (each isomer), decane triisocyanate (each isomer), undecane triisocyanate (each isomer), dodecane triisocyanate (each isomer), tridecane triisocyanate (each isomer isomer), tetradecane triisocyanate (each isomer), pentadecane triisocyanate (each isomer), hexadecane triisocyanate (each isomer), heptadecane triisocyanate (each isomer), o
- the isocyanate compound represented by the following general formula (VIIa) (hereinafter sometimes referred to as “isocyanate compound (VIIa)”) is produced as a by-product in the production method.
- An amine compound represented by the following general formula (IIa) (hereinafter sometimes referred to as “amine compound (IIa)") is selected from liquid phase components containing high boiling point compounds (hereinafter sometimes referred to as "high boiling point compounds”) ) is a method of recovering.
- R 71a is an n71a-valent organic group. n71a is an integer of 2 or more and 8 or less.
- the recovery method of this embodiment includes the following step (a). A step of reacting the liquid phase component, the aromatic hydroxy compound, the active hydrogen-containing compound, and the catalyst in a reactor to obtain a reaction liquid containing the amine compound represented by the general formula (IIa) ( a).
- a liquid phase component containing a high boiling point compound, an aromatic hydroxy compound, an active hydrogen-containing compound, and a catalyst are mixed and reacted to regenerate the amine compound (IIa).
- the liquid phase component, aromatic hydroxy compound, active hydrogen-containing compound, and catalyst may be mixed in advance and supplied to the reactor where the decomposition reaction is performed, or may be supplied separately. In addition, it is also possible to pre-heat before supplying to the reactor in which the decomposition reaction is performed, as long as it does not interfere with the essence of the present embodiment.
- the high boiling point compound in the liquid phase component has Sum of biuret group, allophanate group, isocyanurate group, urea group, carbodiimide group, uretonimine group and imino trimer group (X), isocyanate group, carbamate group, rearranged Fries, cyclized product of rearranged Fries, and oxycarbonyl carbamate
- the relational expression [X/Y ⁇ (n71a) ⁇ 1) ⁇ ] is preferably 2 or less.
- the content of isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group, uretonimine group, imino trimer group, Fries rearrangement product, cyclized product of Fries rearrangement product, and oxycarbonylcarbamate group is For example, by infrared spectroscopy (IR measurement), nuclear magnetic resonance spectroscopy (NMR measurement), or simply by NMR measurement, the —CH 2 — group adjacent to the N atom of the functional group is quantified, and from this value By estimating the amount of the functional group, the value of [X/Y ⁇ (n71a) ⁇ 1) ⁇ ] can be calculated.
- the recovery method of the present embodiment can efficiently regenerate the useful components including the amine compound from the liquid phase component remaining after the production of the isocyanate compound.
- Step (a) is a step of reacting a liquid phase component, an aromatic hydroxy compound, an active hydrogen-containing compound, and a catalyst in a reactor to obtain a reaction liquid containing the amine compound (IIa).
- Step (a) preferably includes the following steps (a1) and (a2). Step (a1) of mixing the liquid phase component and the aromatic hydroxy compound; A step ( a2).
- Step (a1) is a step of mixing a liquid phase component containing a high-boiling compound by-produced in the method for producing the isocyanate compound (VIIa) and an aromatic hydroxy compound.
- the aromatic hydroxy compound used in step (a1) is preferably the aromatic hydroxy compound used in the method for producing the isocyanate compound (VIIa).
- Aromatic hydroxy compounds may be used alone or in combination of two or more.
- the aromatic hydroxy compound includes the above-described isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group, uretonimine group, imino trimer group, Fries rearrangement, cyclized Fries rearrangement, and In many cases, it has high solubility in high-boiling compounds containing oxycarbonyl carbamate groups and the like, and has the effect of making the reaction system uniform and allowing the reaction to proceed rapidly.
- the liquid phase component containing the high boiling point compound continuously extracted from the thermal decomposition reactor contains the aromatic hydroxy compound
- the liquid phase component may be used in the reaction as it is, and a pipe for supplying an aromatic hydroxy compound is provided to a pipe for recovering a liquid phase component containing a high boiling point compound continuously extracted from the separation device in the step (x2) described later.
- Group hydroxy compounds can also be added.
- the amount of the aromatic hydroxy compound used is preferably 10% by mass or more and 500% by mass or less, more preferably 20% by mass or more and 300% by mass or less, and 50% by mass or more and 300% by mass or less, relative to the total mass of the liquid phase components. is more preferred.
- the liquid phase component is transferred from the thermal decomposition reactor and the separation device of the step (x2) described later to the reactor in which the reaction of the step (a1) in the recovery method of the present embodiment is performed, the liquid phase component is It is preferable to transfer while maintaining the state. Therefore, the liquid phase component is preferably kept at 50° C. or higher and 300° C. or lower, more preferably 100° C. or higher and 260° C.
- step (a1) is performed.
- the isocyanate compound (VIIa), biuret group, urea group, carbodiimide group, and uretonimine group contained in the liquid phase component are suppressed from degenerating, and a liquid phase state is obtained. There is an effect of holding.
- the high boiling point compound and the active hydrogen-containing compound are separated by liquid-liquid phase separation in the steps (b) and (e) described later.
- the amine compound (IIa) and the isocyanate compound (VIIa) are mixed In this state, both may be in a mixed state, and phase separation cannot be performed. and can be phase-separated.
- steps (c) and (f) described later when purifying the amine compound (IIa) separated in step (b) and the hydroxy compound separated in step (e) by distillation separation,
- the catalyst used in the step (a2) or the method (B) for producing the isocyanate compound (VIIa) described later may precipitate in the distillation column and the distillation may not be significantly performed, but the aromatic hydroxy compound is added to the liquid phase component, the catalyst used in the following step (a2) or the method (B) for producing the isocyanate compound (VIIa) is maintained in a dissolved state and continuously separated by distillation. and the like.
- Step (a2) is a step of reacting the composition mixed in step (a1), the active hydrogen-containing compound, and the catalyst in a reactor.
- the active hydrogen-containing compound is at least one selected from the group consisting of water, urea, alcohols (not including aromatic hydroxy compounds), alkylthiols and amine compounds (preferably primary amine compounds). is preferred, and at least one selected from the group consisting of water, primary amine compounds, and hydroxy compounds is more preferred. These compounds may be used alone or in combination of two or more. Among them, more preferred are water, a combination of water and a primary amine compound, a combination of water and alcohol, or a combination of urea and alcohol. Also particularly preferred is water or a combination of water and a primary amine compound. Additionally, water or a combination of water and an aliphatic primary amine compound is most preferred.
- the target amine compound (IIa) can also be used as the active hydrogen-containing compound.
- the alcohol used in the reaction is preferably the alcohol that is used in the production of the carbamate compound in the production of the isocyanate compound (VIIa).
- the amount (molar amount) of water used is the isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, 5 times the total molar amount of nitrogen atoms contained in each functional group of carbodiimide group, uretonimine group, imino trimer group, rearranged Fries, cyclized product of rearranged Fries, and oxycarbonyl carbamate group in stoichiometric ratio 200 times or less is preferable, 5 times or more and 150 times or less is more preferable, and 5 times or more and 100 times or less is even more preferable.
- the amount (molar amount) of water used is equal to or less than the above lower limit, an internal ring is formed due to an unfavorable reaction caused by an acidic atmosphere associated with an increase in ion product caused by a large amount of high-temperature and high-pressure water, such as a deammonification reaction of hexamethylenediamine.
- the formation of hexamethyleneimine, which is a by-product of the reaction, can be further suppressed.
- the energy load in the subsequent dehydration step can be further reduced, and it is possible to reduce energy consumption in production and downsize the device.
- the amount (molar amount) of water used is at least the above lower limit, the decomposition reaction of the high-boiling point compound proceeds more satisfactorily.
- the amount of water can be further reduced if the reaction can be carried out efficiently by applying a catalyst, etc. Therefore, when a catalyst is used, the amount (molar amount) of water used is the isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group contained in the liquid phase component. , uretonimine group, imino trimer group, Fries rearrangement form, cyclized product of Fries rearrangement form, and oxycarbonylcarbamate group, at a stoichiometric ratio of at least 5 times the total molar amount of nitrogen atoms contained in each functional group. It is preferably 50 times or less.
- the amount (molar amount) of these active hydrogen-containing compounds used is the isocyanate group, biuret group, Allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group, uretonimine group, imino trimer group, Fries rearrangement, cyclized Fries rearrangement, and oxycarbonyl carbamate group
- the stoichiometric ratio is usually 0.1 to 100 times the molar amount.
- catalysts include, but are not limited to, hydroxides and oxides of alkali metals and alkaline earth metals such as sodium hydroxide, potassium hydroxide, magnesium hydroxide, magnesium oxide, calcium oxide, and barium oxide; pyridine; Heterocyclic compounds and tertiary amines such as methylpyridine (including isomers), dipropylethylamine, N-methylmorpholine, N-ethylmorpholine, triethylamine, triethylenediamine; aluminum oxide, zinc oxide, titanium oxide, zirconium oxide, Group 3-14 metal oxides such as cerium oxide, iron oxide, tin oxide, vanadium oxide, molybdenum oxide, manganese oxide, cobalt oxide and nickel oxide can be mentioned.
- alkali metals and alkaline earth metals such as sodium hydroxide, potassium hydroxide, magnesium hydroxide, magnesium oxide, calcium oxide, and barium oxide
- pyridine Heterocyclic compounds and tertiary amines such as
- alkali metal hydroxides and oxides alkaline earth metal hydroxides and oxides, tertiary amine compounds, group 12 metal oxides, group 13 metal oxides, and group 14
- One or more compounds selected from the group consisting of the group consisting of metal oxides are preferred.
- one or more compounds selected from the group consisting of hydroxides and oxides of alkali metals and hydroxides and oxides of alkaline earth metals are more preferable, and hydroxides and oxides of alkali metals are more preferable.
- alkali metal hydroxides such as sodium hydroxide and potassium hydroxide.
- the catalyst may be homogeneously compatible with the reaction liquid, or may be heterogeneous. Among them, it is preferable to use a homogeneous catalyst because it has the effects of accelerating the decomposition reaction in the reactor, contributing to the suppression of the production of by-products, and improving the yield.
- the amount (molar amount) of the catalyst used is the isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group, uretonimine group, imino trimer group, and Fries rearrangement that are contained in the liquid phase component.
- the cyclized product of the Fries rearrangement, and the total molar amount of nitrogen atoms contained in each functional group of the oxycarbonyl carbamate group the stoichiometric ratio is usually 0.001 to 10 times, and 0 0.005 times or more and 5.00 times or less is preferable, and 0.02 times or more and 3.00 times or less is more preferable.
- the high boiling point compound decomposes slowly or does not decompose, resulting in a lower yield of the amine compound (IIa).
- the functional groups contained in the liquid phase components have different decomposability depending on their bonding modes. It is possible to promote decomposition.
- the Fries rearrangement, the cyclized product of the Fries rearrangement, the isocyanurate group, and the iminotrimer group described above are compared to the biuret group, allophanate group, carbamate group, oxycarbonylcarbamate group, urea group, carbodiimide group, and uretonimine group. Therefore, the decomposition reaction with water is generally difficult to proceed, and the amine compound (IIa) is difficult to form.
- an isocyanurate group and an iminotrimer group are known to be difficult to hydrolyze, and are slow to decompose by known methods.
- the liquid phase component, the aromatic hydroxy compound, the active hydrogen-containing compound, and the catalyst, which are by-produced in the method for producing the isocyanate compound (VIIa), are used, the decomposition of the functional groups is accelerated, and the high molecular weight is added to the reaction solution. do not remain in the Therefore, when the amine compound (IIa) and the hydroxy compound are separated from the reaction solution, clogging of the apparatus due to precipitation or the like can be avoided, and the amine compound (IIa) can be efficiently recovered.
- the reaction temperature of the regeneration reaction can be set according to the compound used, but is preferably 100°C or higher and 300°C or lower, and considering the energy load in this step, is more preferably 160°C or higher and 270°C or lower, and 190°C. Above 240° C. or below is more preferable. Even if the temperature is raised above the above range, the decomposition efficiency is almost saturated. Rather, the environment of high-temperature, high-pressure water increases the ionic product and becomes a reaction field that improves the acidity, so an unfavorable reaction may occur.
- This unfavorable reaction includes, for example, the formation of hexamethyleneimine, an internal cyclized by-product of the deammonification reaction of hexamethylenediamine.
- crosslinkable bonds typified by urea groups that can be produced as intermediates in the hydrolysis reaction increase, and high molecular weight substances, ie, gels, may be generated in the system.
- the gel becomes a scale and adheres to the inside of the apparatus, which not only significantly lowers the reaction efficiency, but also causes a decrease in economic efficiency due to the need to clean the apparatus.
- the decomposition of the crosslinkable molecule represented by the urea group proceeds more favorably, and the generation of scale in the apparatus can be further suppressed. can be recovered more efficiently.
- the reaction pressure is usually in the range of 0.01 kPa to 10 MPa (absolute pressure), and can be carried out under reduced pressure, normal pressure, or increased pressure.
- the reaction time (residence time in the case of a continuous reaction) is usually 0.01 hours or more and 100 hours or less, preferably 0.1 hours or more and 10 hours or less, more preferably 0.5 hours or more and 3 hours or less. It is more preferably 3 hours or more and 3 hours or less.
- the reaction solution can be appropriately sampled to measure the production amount of the target compound, amine compound (IIa), and the reaction can be terminated when the desired production amount is reached.
- low boiling point components with a boiling point of 30 ° C or less may be generated, but the low boiling point components may increase the pressure of the reaction system, and the presence of low boiling point components may slow down the reaction. Therefore, it is preferable to carry out the reaction while withdrawing at least part of the low-boiling-point component as a gas phase component from the reaction system. In that case, it is preferable to prevent the active hydrogen-containing compound from being extracted out of the reaction system together with the low boiling point component, such as by installing a condenser in the middle of the line for extracting the low boiling point component from the reactor in which the reaction is performed.
- isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group, uretonimine group, imino trimer group, Fries rearrangement, Fries rearrangement and the oxycarbonylcarbamate group react with water to produce carbon dioxide in some cases, and it is preferable to withdraw the produced carbon dioxide out of the reaction system.
- the produced carbon dioxide forms a carbonate with the amine compound (IIa) produced by the reaction, which may slow down the reaction.
- the susceptibility to carbonate formation depends on the nucleophilicity of the amine compound (IIa), and the permutation of carbonate formation is generally aliphatic-aliphatic amines > substituted cycloaliphatic polyamines > aromatic Since it is a group amine, it is preferable to adjust the amount of the generated carbon dioxide to be extracted out of the reaction system as necessary depending on the structure of the amine compound (IIa).
- reactors there are no particular restrictions on the reactor for the reaction, and known reactors can be used.
- stirred tank pressurized stirred tank, vacuum stirred tank, column reactor, distillation column, packed column, thin film still, paddle dryer with forced conveying device, extruder with degassing function.
- a vertical thin film evaporator equipped with a forced conveying device, a tubular reactor, etc. according to the reaction method and conditions, conventionally known reactors can be used in combination as appropriate.
- reaction may be batch type or continuous flow type, and the reactor may be selected according to each reaction type.
- the material of the reactor there are no particular restrictions on the material of the reactor, and known materials can be used.
- glass, stainless steel, carbon steel, Hastelloy, substrates with glass lining, Teflon (registered trademark) coating, and the like can be used.
- SUS304, SUS316, SUS316L, etc. are also inexpensive and can be preferably used.
- known process devices such as instrumentation equipment such as a flow meter and thermometer, a mechanism for maintaining pressure, a reboiler, a pump, a condenser and the like may be added.
- a known method such as steam or a heater may be used for heating, and a known method such as natural cooling, cooling water, or brine may be used for cooling.
- Step (a2) preferably includes the following steps (a2-1) and (a2-2).
- Step (a2-1) is a step of mixing the mixture obtained in step (a1) with an amine compound as an active hydrogen-containing compound and a catalyst.
- the temperature in the reactor in step (a2-1) is preferably 100°C or higher and 350°C or lower, more preferably 150°C or higher and 300°C or lower, even more preferably 180°C or higher and 280°C or lower.
- the mixing time (residence time in the case of continuous mixing) in step (a2-1) is usually 0.01 hours or more and 10 hours or less. 0.1 hours or more and 5 hours or less are preferable, 0.2 hours or more and 2.5 hours or less are more preferable, and 0.3 hours or more and 2 hours or less are even more preferable. It is also possible to appropriately sample the reaction solution, measure the production amount of the urea group, which is the target compound, and terminate the mixing when the desired production amount is reached.
- the amine compound used in step (a2-1) is a primary amine compound or a secondary amine compound, preferably a primary amine compound, and more preferably an amine compound (IIa). That is, the target amine compound (IIa) can be used as the active hydrogen-containing compound.
- the amount (molar amount) of the amine used in step (a2-1) is the isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group, uretonimine contained in the liquid phase component. 0.5 times or more in stoichiometric ratio with respect to the total molar amount of nitrogen atoms contained in each functional group of group, imino trimer group, Fries rearrangement form, cyclized product of Fries rearrangement form, and oxycarbonyl carbamate group
- the range is preferably 200 times or less, more preferably 1 time or more and 50 times or less, and even more preferably 2 times or more and 20 times or less.
- the amine compound reacts with the biuret group, allophanate group, carbamate group, and uretonimine group contained in the liquid phase component to form the corresponding urea group, and the carbodiimide group is reacted with the corresponding urea group. and amidine groups, and these reactions proceed quantitatively upon mixing. Since the urea group and amidine group are crosslinkable bonds, they precipitate as a gel when they form a high molecular weight product. On the other hand, if the amount of the amine compound is within the above range, the decomposition of the crosslinkable molecule proceeds smoothly, and the generation of scale in the apparatus can be further suppressed. ) can be recovered more efficiently.
- step (a2-1) examples of the catalyst used in step (a2-1) are the same as those exemplified in step (a2) above. Among them, alkali metal hydroxides, oxides and phenoxides, or alkaline earth metal hydroxides, oxides and phenoxides are preferred. From the viewpoint of solubility in the reaction system, sodium phenoxide or potassium phenoxide is more preferred.
- step (a1) When the mixture obtained in step (a1) is mixed with an amine as an active hydrogen-containing compound, a urea group corresponding to the isocyanurate group contained in the liquid phase component is formed. This reaction proceeds quantitatively. For the imino trimer group, the corresponding urea group and amidine group are formed.
- the isocyanurate group and the imino trimer group are significantly decomposed by using a catalyst that homogenizes in the reaction system. be able to. As described above, the isocyanurate group and the imino trimer group, which are crosslinkable bonds, form high molecular weight substances and cause scale in the pipe. suppressing effect.
- the molar amount of the catalyst used in step (a2-1) is the isocyanate group, biuret group, allophanate group, isocyanurate group, carbamate group, urea group, carbodiimide group, uretonimine group, imino
- the stoichiometric ratio of the total molar amount of nitrogen atoms contained in each functional group of the trimer group, the Fries rearrangement form, the cyclized product of the Fries rearrangement form, and the oxycarbonyl carbamate group is usually 0.001 times or more 10 times or less, preferably 0.005 times or more and 5.00 times or less, and more preferably 0.02 times or more and 3.00 times or less.
- the liquid phase component, the amine compound and the catalyst may be mixed in advance and supplied to the reactor in which step (a2-2) is performed, or may be supplied separately and mixed in the reactor. Moreover, it is also possible to preheat before supplying to the reactor in which the step (a2-2) is carried out, as long as it does not interfere with the essence of the present embodiment.
- Step (a2-2) In step (a2-2), the mixture obtained in step (a2-1) and water as an active hydrogen-containing compound are reacted in a reactor to obtain a reaction liquid containing amine compound (II).
- the amount (molar amount) of water used in step (a2-2) is the same as the amount (molar amount) of water used in step (a2) above.
- the temperature in the reactor in step (a2-2) is preferably 100°C or higher and 350°C or lower, more preferably 150°C or higher and 300°C or lower, even more preferably 180°C or higher and 240°C or lower.
- the pressure in the reactor is preferably 0.01 kPa or more and 15 MPa or less (absolute pressure), and the reaction can be carried out under reduced pressure, normal pressure, or increased pressure.
- the reaction time (residence time in the case of continuous reaction) is 0.01 to 100 hours, preferably 0.1 to 10 hours, more preferably 0.2 to 3 hours, and 0.01 to 100 hours. 3 hours or more and 2 hours or less are more preferable.
- the reaction solution may be appropriately sampled, the amount of amine compound (IIa), which is the target compound, produced may be measured, and the reaction time may be the time when the desired amount of production is reached.
- the amine compound (IIa) can be recovered from the reaction liquid obtained by the reaction through distillation separation, liquid-liquid phase separation, solid-liquid separation, membrane separation, and the like.
- the recovered amine compound (IIa) is preferably reused as the amine compound (IIa) in step (a2-1).
- an aromatic hydroxy compound is produced by the reaction of the high boiling point compound and the active hydrogen-containing compound, the aromatic hydroxy compound is recovered and used as the aromatic hydroxy compound in step (a2-1) or step (a1). It can also be reused.
- the recovery method of the present embodiment preferably further includes the following steps (b), (c), (e), and (f). These steps (b), (c), (e), and (f) can be performed continuously, or step (b), step (c), step (e), and step (f) can also be performed simultaneously. Alternatively, the operations of steps (e) and (f) can be performed before steps (b) and (c). In this case, in step (e), the reaction solution containing the amine compound (IIa) obtained in step (a2) is used as a raw material, while in step (b), after the hydroxy compound is separated in step (e), The residual liquid of is used as a raw material. Alternatively, some steps can be exchanged and implemented.
- Step (b) is a step of separating the amine compound (IIa) from the liquid obtained in step (a2), step (a2-2), or step (e) or step (f) described later.
- a known method can be used to separate the amine compound (IIa) from the reaction solution obtained in step (a2) or step (a2-2). For example, distillation separation, liquid-liquid phase separation, Examples include solid-liquid separation and membrane separation.
- the step (b) can also continuously separate the amine compound (IIa) from the liquid phase component (reaction liquid) continuously discharged in the step (a2) or step (a2-2).
- Step (c) is a step of purifying the amine compound (IIa) separated in step (b).
- a known method can be used for purifying the amine compound (IIa), and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- the step (c) can also continuously purify the amine compound (IIa) continuously separated in the step (b).
- Step (e) Separats the hydroxy compound used in the production of the isocyanate compound (VIIa) from the liquid obtained in the step (a2), step (a2-2), step (b) or step (c). It is a process to do.
- a compound containing a carbamate group such as a carbamate compound described later
- step (e) when the hydroxy compound used as a raw material in the production of the isocyanate compound is different from the aromatic hydroxy compound used in the above step (a), depending on the separation method, mainly the It is a hydroxy compound used as a raw material in the production of isocyanate compounds.
- step (e) the hydroxy compound is separated from the liquid obtained in step (a2), step (a2-2), step (b) or step (c), and the hydroxy compound is recovered.
- a known method can be used for the separation in step (e), and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- Step (f) is a step of purifying the hydroxy compound separated in step (e).
- step (f) it is preferable to perform step (f) after step (e) to purify the hydroxy compound.
- a known method can be used to purify the hydroxy compound, and examples thereof include distillation separation, liquid-liquid phase separation, solid-liquid separation, and membrane separation.
- Step (f) can also continuously purify the hydroxy compound continuously separated in step (e).
- the recovery method of the present embodiment preferably further includes the following steps (d) and (g).
- Step (d) is a step of reusing the amine compound (IIa) purified in step (c) as a raw material for the isocyanate compound (VIIa).
- the method for producing the isocyanate compound (VIIa) and the quality of the isocyanate compound (VIIa) produced by the method are often affected, so in the step (c), the total mass of the amine compound (IIa)
- Step (g) is a step of reusing the hydroxy compound purified in step (f) as a raw material in the method for producing isocyanate compound (VIIa).
- the method for producing the isocyanate compound (VIIa) and the quality of the isocyanate compound (VIIa) produced by the method are often affected.
- the hydroxy compound is recovered by distillation so that the metal component content is 1000 mass ppm or less and the halogen atom content is 1000 mass ppm or less.
- the recovery method of the present embodiment preferably further includes the following step (h).
- the residual liquid after separating the amine compound (IIa) in the step (b), the residual liquid after purifying the amine compound (IIa) in the step (c), and the hydroxy compound separated in the step (e) a step (h) of reusing at least one residual liquid selected from the group consisting of the subsequent residual liquid and the residual liquid after purifying the hydroxy compound in the step (f) in the step (a).
- Step (h) is the residual liquid after separating the amine compound (IIa) in the step (b), and the residual liquid after purifying the amine compound represented by the general formula (IIa) in the step (c). , a residual liquid after separating the hydroxy compound in the step (e), and a residual liquid after purifying the hydroxy compound in the step (f); This is a step of recycling in step (a).
- the amine compound (IIa) can be more efficiently removed in the recovery method of the present embodiment. can be recovered.
- the high boiling point compound contained in the liquid phase component is a by-product produced during the production of the isocyanate compound (VIIa) using the amine compound (IIa) as a raw material, and is an amine compound (IIa) and a reaction intermediate, which will be described later. It is presumed that one or more compounds selected from the group consisting of carbamate compounds and isocyanate compounds (VIIa) react to form.
- the high boiling point compound is represented by a group represented by the following formula (IX-1) (isocyanate group), a group represented by the following formula (IX-2) (isocyanurate group), and represented by the following formula (IX-3).
- a group represented by the following formula (imino trimer group) A group represented by the following formula (imino trimer group), a group represented by the following formula (IX-7) (biuret group), a group represented by the following formula (IX-8) (allophanate group), the following formula (IX- 9) a group represented by the following formula (carbamate group), a group represented by the following formula (IX-10) (urea group), a group represented by the following formula (IX-11) (a functional group generated
- the high boiling point compound by-produced in the process of producing the isocyanate compound (VIIa) via the carbamate compound described below is a group represented by the following formula (IX-11) (Fries rearrangement), the following formula (IX- 12) (cyclized form of rearranged Fries), a group represented by the following formula (IX-5) (oxycarbonyl carbamate group), and the like.
- the high boiling point compound by-produced in the process of producing the isocyanate compound (VIIa) by thermal decomposition using a carbamate compound and a carbonate ester, which will be described later is a group represented by the following formula (IX-5) (oxycarbonyl carbamate group).
- the high boiling point compound by-produced in the process of producing the isocyanate compound (VIIa) via a carbamate compound synthesized from urea and N-unsubstituted carbamic acid which will be described later, is represented by the following formula (IX-2).
- (isocyanurate group) a group represented by the following formula (IX-3) (carbodiimide group), and the like.
- a group (isocyanurate group) represented by the following formula (IX-2) which is by-produced in the process of producing an isocyanate compound (VIIa) via a carbamate compound synthesized from urea or N-unsubstituted carbamic acid.
- each of the three nitrogen atoms in the following formula (IX-2) is bonded to R 71a or a hydrogen atom in the general formula (VIIa) described later, that is, 3 in the following formula (IX-2)
- a bond indicated by two wavy lines may be R 71a or a hydrogen atom in general formula (VIIa) described later.
- the high boiling point compound is a group (isocyanate group) represented by the following formula (IX-1), a group (isocyanurate group) represented by the following formula (IX-2), and a group represented by the following formula (IX-3).
- the group represented by the above formula (IX-1) isocyanate group
- the group represented by the above formula (IX-2) isocyanurate group
- the above The group represented by the formula (IX-3) carbbodiimide group
- the group represented by the formula (IX-4) (uretonimine group)
- the group represented by the formula (IX-5) oxycarbonyl carbamate group
- a group represented by the formula (IX-7) (biuret group)
- a group represented by the formula (IX-8) allophanate group
- the group represented by the formula (IX-9) carbarbamate group
- the group represented by the formula (IX-10) (urea group), the group represented by the formula (IX-11) (Fries rearrangement) and the group represented by the formula (IX-12) (cyclized Fries rearrangement), the total content of nitrogen atoms contained
- crosslinkable bonds typified by urea groups, which can be formed as intermediates in the hydrolysis reaction, increase, and high molecular weight substances, that is, gels, may occur in the system.
- the gel becomes scales and adheres to the inside of the apparatus, which not only significantly lowers the reaction efficiency, but also lowers economic efficiency because the apparatus needs to be cleaned.
- the content of the functional group described above is within the above range, the decomposition proceeds more favorably, and the generation of scale in the apparatus can be further suppressed, so that the amine compound (IIa) can be recovered more efficiently. can.
- the content of these functional groups can be determined, for example, by infrared spectroscopy (IR measurement), nuclear magnetic resonance spectroscopy (NMR measurement), or simply by NMR measurement of —CH 2 adjacent to the N atom of the functional group.
- IR measurement infrared spectroscopy
- NMR measurement nuclear magnetic resonance spectroscopy
- - groups are quantified, the total mass of the functional groups is estimated from this value, and the total mass is divided by the mass of the sample (liquid phase component) subjected to measurement, and the value can be calculated as a percentage.
- the high boiling point compound is a compound with a higher boiling point than the isocyanate compound (VIIa). Specifically, in the method for producing the isocyanate compound (VIIa) via a carbamate compound, a compound that did not become a gas phase component under the same conditions as the compound extracted as a gas phase component from the thermal decomposition reactor, or It is a compound that did not become a gas phase component in the distillation purification step of the isocyanate compound (VIIa).
- the boiling point of the high boiling point compound is not particularly limited, but is, for example, 300° C. or higher at the operating pressure of the thermal decomposition reactor or distillation apparatus.
- the isocyanate compound (VIIa) is a compound represented by the following general formula (VIIa).
- R 71a is an n71a-valent organic group. n71a is an integer of 2 or more and 8 or less.
- R 71a is an n71a valent organic group. That is, it is an organic group having a valence of 2 or more and 8 or less.
- R 71a is a divalent to tetravalent aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have an ester group of 1 or more and 4 or less or a nitrogen atom, or an aliphatic hydrocarbon group of 6 or more carbon atoms. It is preferably an aromatic hydrocarbon group having a valence of 20 or less and having a valence of 20 or more and 3 or less.
- the aliphatic hydrocarbon group for R 71a includes an alkylene group or an alkanetriyl group, a cycloalkyl group, a cycloalkylene group or a cycloalkanetriyl group, or the alkyl group, the alkylene group or the alkanetriyl group, A group composed of the cycloalkyl group, the cycloalkylene group or the cycloalkanetriyl group is preferable, and a linear or branched alkylene group or alkanetriyl group, cycloalkylene group or cycloalkanetriyl group , or a group composed of the alkylene group or the alkanetriyl group and the cycloalkyl group, the cycloalkylene group or the cycloalkanetriyl group is more preferable.
- linear or branched alkylene groups examples include methylene group, ethylene group, propylene group, trimethylene group, pentylene group, n-hexylene group and decamethylene group.
- the cycloalkylene group includes, for example, a cyclobutylene group, a cyclohexylene group, and the like.
- linear or branched alkanetriyl groups examples include hexanetriyl, nonanetriyl, and decantriyl groups.
- the cycloalkanetriyl group includes, for example, a cyclopropanetriyl group, a cyclobutanetriyl group, a cyclopentanetriyl group, a cyclohexanetriyl group, and the like.
- the aromatic hydrocarbon group for R 71a is preferably a substituted or unsubstituted group having an aromatic ring having 6 or more and 13 or less carbon atoms.
- substituents include alkyl groups, aryl groups, and aralkyl groups.
- the aromatic ring may be an aromatic hydrocarbon ring or a heteroaromatic ring, and specific examples thereof include benzene ring, naphthalene ring, pyridine ring and the like.
- R 71a is preferably a group represented by any one of the following formulas (Ia-1) to (Ia-24), such as formulas (Ia-1), (Ia-2), (Ia-3 ), (Ia-14), (Ia-18), or (Ia-19).
- formulas (Ia-1) to (Ia-24) such as formulas (Ia-1), (Ia-2), (Ia-3 ), (Ia-14), (Ia-18), or (Ia-19).
- a wavy line indicates a bond.
- n71a represents the number of isocyanate groups and is an integer of 2 or more and 8 or less, preferably an integer of 2 or more and 4 or less, and more preferably an integer of 2 or more and 3 or less.
- R 71a is an aliphatic hydrocarbon group
- specific examples of the isocyanate compound (VIIa) include aliphatic diisocyanates, aliphatic triisocyanates, and substituted cycloaliphatic polyisocyanates. .
- aliphatic diisocyanates examples include diisocyanatoethane, diisocyanatopropane (each isomer), diisocyanatobutane (each isomer), diisocyanatopentane (each isomer), diisocyanatohexane (each isomer), diisocyanatodecane (each isomer), and the like.
- aliphatic triisocyanates examples include triisocyanatohexane (each isomer), 4-isocyanatomethyl-1,8-octamethylene diisocyanate, triisocyanatononane (each isomer), triisocyanatodecane (each isomer) and the like.
- Substituted cycloaliphatic polyisocyanates include, for example, diisocyanatocyclobutane (each isomer), diisocyanatocyclohexane (each isomer), 3-isocyanatomethyl-3,5,5-trimethylcyclohexylisocyanate (also called isophorone diisocyanate) (each isomer), 1,3-bis(isocyanatomethyl)cyclohexane (at least one of cis and trans isomers), methylenebis(cyclohexyl isocyanate) (also called dicyclohexylmethane diisocyanate) ) (each isomer) and the like.
- diisocyanatocyclobutane each isomer
- diisocyanatocyclohexane each isomer
- 3-isocyanatomethyl-3,5,5-trimethylcyclohexylisocyanate also called isophorone diisocyanate
- R 71a is an aromatic group
- specific examples of the isocyanate compound (VIIa) include aromatic diisocyanates and aromatic triisocyanates.
- aromatic diisocyanates include diisocyanatobenzene (each isomer), diisocyanatotoluene (each isomer), bis(isocyanatophenyl)methane (each isomer), diisocyanatomesitylene (each isomer ), diisocyanatobiphenyl (each isomer), diisocyanatodibenzyl (each isomer), bis(isocyanatophenyl)propane (each isomer), bis(isocyanatophenyl) ether (each isomer), bis (isocyanatophenoxyethane) (each isomer), diisocyanatoxylene (each isomer), diisocyanatoanisole (each isomer), diisocyanatophenetol (each isomer), diisocyanatonaphthalene (each isomer) isomer), diisocyanatomethylbenzene (each
- aromatic triisocyanates examples include triisocyanatobenzene (each isomer), triisocyanato-methylbenzene (each isomer), tris(isocyanatopropan-yl)benzene (each isomer), tris(isocyanate Natopropan-yl)-methylbenzene (each isomer), Tris(isocyanatomethyl)-methylbenzene (each isomer), ((isocyanato-phenylene)bis(methylene))bis(isocyanatobenzene) (each isomer) , triphenylmethane triisocyanate, and the like.
- isocyanate compound (VIIa) examples include acrylic acid-2-isocyanato-ethyl ester, 2-methyl - acrylic acid-2-isocyanato-ethyl ester, acrylic acid-2-isocyanato-propyl ester, 2-methyl-acrylic acid-2-isocyanato-propyl ester, acrylic acid-3-isocyanato-propyl ester, 2-methyl-acrylic acid.
- Acid-3-isocyanato-propyl ester acrylic acid-4-isocyanato-butyl ester, 2-methyl-acrylic acid-4-isocyanato-butyl ester, acrylic acid-5-isocyanato-pentyl ester, 2-methyl-acrylic acid- 5-isocyanato-pentyl ester, acrylic acid-6-isocyanato-hexyl ester, 2-methyl-acrylic acid-6-isocyanato-hexyl ester, acrylic acid-8-isocyanato-octyl ester, 2-methyl-acrylic acid-8- Isocyanato-octyl Ester, Acrylic Acid-10-isocyanato-decyl Ester, 2-Methyl-acrylic Acid-10-isocyanato-decyl Ester, Acrylic Acid-11-isocyanato-undecyl Ester, 2-Methyl-acrylic Acid-11-isocyanato -undecyl ester, acrylic acid
- Preferred isocyanate compounds (VIIa) include, for example, pentamethylene diisocyanate, hexamethylene diisocyanate, 4-isocyanatomethyl-1,8-octamethylene diisocyanate, lysine ethyl ester diisocyanate, methylene bis(cyclohexyl isocyanate) (HMDI), 3-isocyanate Natomethyl-3,5,5-trimethylcyclohexyl isocyanate (isophorone diisocyanate), diisocyanatotoluene (each isomer), diisocyanatodiphenylmethane (MDI), triisocyanatobenzene (each isomer), triphenylmethane triisocyanate etc.
- HMDI methylene bis(cyclohexyl isocyanate)
- HMDI 3-isocyanate Natomethyl-3,5,5-trimethylcyclohexyl isocyanate
- MDI
- the isocyanate compound (VIIa) can be produced using the amine compound (IIa) as a starting material, and known methods can be used as the production method. For example, method (X) and method (Y) shown below may be mentioned.
- Method (X) can include other steps, if necessary, in addition to steps (x1) and (x2).
- steps (x1) and (x2) are used as another step, for example, the carbamate compound obtained in the step (x1) and a hydroxy compound different from the hydroxy compound used in the step (x1) are used to carry out a carbamate transesterification reaction to produce a different carbamate.
- Step (x1-a) Separating part or all of the hydroxy compound from the reaction solution of Step (x1) and pre-concentrating (Step (x1-b)); Purifying the isocyanate compound (VIIa) Step (step (x3)); a step of recovering ammonia, a hydroxy compound, etc., which are by-produced from the carbonic acid derivative in the step (x1), and the like.
- step (x3) is A gas phase component containing a compound having a boiling point lower than that of the isocyanate compound (VIIa) (hereinafter sometimes referred to as a "low boiling point compound”) is separated by distillation to separate a liquid phase component containing the isocyanate compound (VIIa) ( hereinafter sometimes referred to as “light boiling separation”) step (step (x3-1)); and The gas phase component containing the isocyanate compound (VIIa) is separated by distillation to separate the high boiling point compound from the isocyanate compound (VIIa) (hereinafter sometimes referred to as "high boiling point separation”) step (step (x3-2) ); Alternatively, the order of performing the step (x3-1) and the step (x3-2) can be changed.
- Step (x1) In the step (x1), the amine compound (IIa), a carbonic acid derivative and a hydroxy compound are reacted to produce a carbamate compound, and one or more compounds selected from the group consisting of urea and isocyanic acid, a hydroxy compound, and ammonia is a step of recovering a gaseous phase component containing separately from the produced carbamate compound.
- the step (x1) can be roughly divided into the following two methods: method (x1-1) and method (x1-2). In the manufacturing method of this embodiment, the following method (x1-1) and method (x1-2) may be combined.
- Method (x1-1) A method of producing a carbamate compound by "simultaneously" reacting amine compound (IIa), urea and a hydroxy compound.
- Method (x1-2) step (x1-2-1) of reacting amine compound (IIa) with urea to produce a compound having a ureido group; and ureide obtained in step (x1-2-1) a step of reacting a compound having a group with a hydroxy compound to produce a carbamate compound (x1-2-2); method including.
- the amount (molar amount) of the hydroxy compound used is usually 1 to 500 times the stoichiometric ratio with respect to the molar amount of the amino group of the amine compound (IIa) used. or less, preferably 5 times or more and 200 times or less, more preferably 10 times or more and 150 times or less, and even more preferably 10 times or more and 100 times or less.
- the amount (molar amount) of urea to be used is usually 1-fold to 100-fold, and 1-fold to 10-fold in stoichiometric ratio with respect to the molar amount of amino groups in the amine compound (IIa) used.
- the following is preferable, 1 to 3 times is more preferable, and 1 to 1.5 times is even more preferable.
- the reaction temperature is usually 100°C or higher and 350°C or lower, preferably 150°C or higher and 300°C or lower, more preferably 180°C or higher and 280°C or lower, even more preferably 200°C or higher and 260°C or lower.
- the reaction pressure is usually 0.01 kPa to 10 MPa (absolute pressure), preferably 1 kPa to 3 MPa, more preferably 10 kPa to 1 MPa, and even more preferably 101 kPa to 0.5 MPa.
- Methods for removing ammonia from the system include a reactive distillation method, a method using an inert gas, a method using membrane separation, a method using adsorption separation, and the like. Solvents and catalysts can be used if necessary in the reaction.
- the reaction time (residence time in the case of a continuous reaction) is usually 0.01 to 100 hours, preferably 0.1 to 10 hours, more preferably 0.2 to 2 hours, More preferably 0.3 hours or more and 1 hour or less. It can also be determined by the production amount of the carbamate compound, which is the target compound in method (x1-1).
- step (x1-2-1) the amine compound (IIa) and urea are reacted to produce a compound having a ureido group.
- step (x1-2-1) the number of ureas is in the range of 1 to 100 times the number of amino groups in the compound represented by general formula (III). Among them, 1-fold to 10-fold is preferable, 1-fold to 3-fold is more preferable, and 1-fold to 1.5-fold is even more preferable.
- the reaction temperature is usually 30°C or higher and 250°C or lower, preferably 100°C or higher and 200°C or lower, more preferably 120°C or higher and 200°C or lower, and even more preferably 130°C or higher and 180°C or lower.
- the reaction pressure is usually 0.01 kPa or more and 10 MPa (absolute pressure), preferably 1 kPa or more and 3 MPa or less, more preferably 10 kPa or more and 1 MPa or less, and even more preferably 50 kPa or more and 0.5 MPa or less.
- the reaction time (residence time in the case of a continuous method) is 0.01 to 100 hours, preferably 0.1 to 10 hours, more preferably 0.2 to 2 hours, and 0.01 to 100 hours. More preferably 3 hours or more and 1 hour or less. After confirming that a desired amount of the compound having a ureido group is produced, the reaction can be terminated.
- a catalyst or solvent can be used if necessary in step (x1-2-1).
- the aromatic hydroxy compound used in step (a) is used as the solvent.
- step (x1-2-2) the compound having a ureido group obtained in step (x1-2-1) is reacted with a hydroxy compound to produce a carbamate compound.
- the reaction solution obtained in the step (x1-2-1) is used as it is in the step (x1-2- 2) can be performed.
- the stoichiometric ratio of the amount (molar amount) of the hydroxy compound to be used is generally 1-fold to 500-fold, and 5-fold to 200-fold relative to the molar amount of the ureido group in the compound having a ureido group to be used. It is preferably 10 times or less, more preferably 10 times or more and 150 times or less, and even more preferably 10 times or more and 100 times or less.
- the reaction temperature is usually 100°C or higher and 350°C or lower, preferably 150°C or higher and 300°C or lower, more preferably 180°C or higher and 280°C or lower, even more preferably 200°C or higher and 260°C or lower.
- the reaction pressure is usually 0.01 kPa to 10 MPa (absolute pressure), preferably 1 kPa to 3 MPa, more preferably 10 kPa to 1 MPa, and even more preferably 101 kPa to 0.5 MPa.
- Methods for removing ammonia from the system include a reactive distillation method, a method using an inert gas, a method using membrane separation, a method using adsorption separation, and the like. Solvents and catalysts can be used if necessary in the reaction.
- the reaction time (residence time in the case of a continuous reaction) is usually 0.01 to 100 hours, preferably 0.1 to 10 hours, more preferably 0.2 to 2 hours, More preferably 0.3 hours or more and 1 hour or less. It can also be determined by the production amount of the carbamate compound, which is the target compound in step (x1-2-2).
- step (x1-2-2) a solvent or catalyst can be used if necessary.
- the catalyst is not particularly limited, but for example, alkali metals such as sodium hydroxide, potassium hydroxide, magnesium hydroxide, magnesium oxide, calcium oxide, barium oxide, hydroxides and oxides of alkaline earth metals; Heterocyclic compounds and tertiary amines such as methylpyridine (including isomers), dipropylethylamine, N-methylmorpholine, N-ethylmorpholine, triethylamine, triethylenediamine; aluminum oxide, zinc oxide, titanium oxide, zirconium oxide, oxide Group 3 to 14 metal oxides such as cerium, iron oxide, tin oxide, vanadium oxide, molybdenum oxide, manganese oxide, cobalt oxide, nickel oxide, and the like.
- alkali metals such as sodium hydroxide, potassium hydroxide, magnesium hydroxide, magnesium oxide, calcium oxide, barium oxide, hydroxides and oxides of alkaline earth metals
- Heterocyclic compounds and tertiary amines such as
- the catalyst may be homogeneously compatible with the reaction liquid, or may be heterogeneous.
- the amount of the catalyst used is such that the molar amount of the catalyst is usually 0.001 to 10 times the stoichiometric ratio with respect to the molar amount of the ureido group in the compound having the ureido group used. . These catalysts can be used alone or in combination of two or more.
- Step (x2) The carbamate compound produced in the step (x1) is a compound represented by the following general formula (Vc) (hereinafter sometimes referred to as “carbamate compound (Vc)").
- the step (x2) is a step of subjecting the carbamate compound (Vc) produced in the step (x1) to a thermal decomposition reaction to produce a component containing the isocyanate compound (VIIa).
- the step (x2) can be roughly divided into the following two methods: method (x2-1) and method (x2-2).
- a method for producing an isocyanate compound (VIIa) by separating a gas phase component containing the compound (VIIa) from a high boiling point compound.
- the method (x2-1) and method (x2-2) can be carried out using an appropriate solvent.
- the reaction temperature for thermally decomposing the carbamate compound is usually in the range of 100°C or higher and 300°C or lower.
- the reaction pressure is usually in the range of 20 Pa or more and 1 ⁇ 10 6 Pa or less.
- a catalyst is not always necessary, but a catalyst may be used to lower the reaction temperature or complete the reaction early.
- a catalyst may be used to lower the reaction temperature or complete the reaction early.
- organic tin compounds, compounds of copper group metals, zinc, iron group metals, and the like can be used in an amount of 0.01 to 30% by mass relative to the mass of the carbamate compound.
- the reaction time (residence time in the case of a continuous method) is preferably as short as possible within a range that does not hinder the progress of the desired reaction.
- the hydroxy compound or the like contained in the reaction solution obtained in step (x1) can be used as a solvent.
- the thermal decomposition reaction of the carbamate compound (Vc) is performed by placing a mixture containing the carbamate compound (Vc) in a reactor (sometimes referred to as a thermal decomposition reactor). and subjected to a thermal decomposition reaction, part of the resulting isocyanate compound (VIIa) and hydroxy compound are continuously extracted from the thermal decomposition reactor as gas phase components, and the remaining liquid phase components are , a method of continuously withdrawing from the pyrolysis reactor is adopted.
- the type of thermal decomposition reactor used in method (x2-1) is not particularly limited, but a known distillation apparatus is preferably used in order to efficiently recover gas phase components.
- Examples of the type of the pyrolysis reactor using a distillation apparatus include a distillation column, a multi-stage distillation column, a multi-tubular reactor, a continuous multi-stage distillation column, a packed column, a thin film evaporator, and a reaction equipped with a support inside.
- Various known methods are used, such as a method using a reactor including any of a reactor, a forced circulation reactor, a falling film evaporator, a falling drop evaporator, and a method combining these.
- the thermal decomposition reactor As a thermal decomposition reactor using the distillation apparatus used in this embodiment, the low boiling point components produced (isocyanate compound (VIIa) and hydroxy compound in method (x2-1)) are rapidly transferred to the gas phase.
- a structure with a large gas-liquid contact area is preferred, a method using a tubular reactor is more preferred, and a method using a tubular thin film evaporator or a tubular falling film evaporator is even more preferred.
- the thermal decomposition reactor comprises the above tubular reactor, the gas phase component and the liquid phase component. It is further preferable to comprise a separation tank for separating the .
- the thermal decomposition reaction of the carbamate compound (Vc) is performed by continuously feeding a mixture containing the carbamate compound (Vc) to a reactor (sometimes referred to as a thermal decomposition reactor). and subjected to a thermal decomposition reaction, the hydroxy compound as a gaseous phase component is continuously withdrawn from the thermal decomposition reactor, and the remaining isocyanate compound (VIIa) and the high-boiling point compound are contained as a liquid phase component of heat.
- a method of continuous withdrawal from the cracking reactor is employed.
- the type of thermal decomposition reactor used in method (x2-2) is not particularly limited, but a known distillation apparatus is preferably used in order to efficiently recover gas phase components.
- Examples of the type of the pyrolysis reactor using a distillation apparatus include a distillation column, a multi-stage distillation column, a multi-tubular reactor, a continuous multi-stage distillation column, a packed column, a thin film evaporator, and a reaction equipped with a support inside.
- Various known methods are used, such as a method using a reactor including any of a reactor, a forced circulation reactor, a falling film evaporator, a falling drop evaporator, and a method combining these.
- the low boiling point component (hydroxy compound in method (x2-2)) to be produced is rapidly transferred to the gas phase, and the carbamate compound A structure with a large gas-liquid contact area that allows efficient separation from (Vc) is preferred, and a method using a tubular thin film evaporator or a tubular falling film evaporator is more preferred, and a distillation column or multi-stage distillation column is used. A method is more preferred.
- the mixture is continuously supplied to a separation device.
- the isocyanate compound (VIIa) is continuously withdrawn from the separator as a gaseous phase component, and the residual liquid containing the high boiling point compound is continuously withdrawn from the separator as a liquid phase component.
- the type of the separation device is not particularly limited, but for example, a distillation column, a multi-stage distillation column, a multi-tubular reactor, a continuous multi-stage distillation column, a packed column, a method using a separation device including a thin film evaporator, and Various known methods such as a method combining these methods are used.
- the liquid phase component containing the high-boiling point compound, which is continuously withdrawn from the thermal decomposition reactor, or the isocyanate compound (VIIa) is continuously withdrawn in the distillation step.
- a liquid phase component containing a high boiling point compound is used as a liquid phase component containing a high boiling point compound which is a raw material for the recovery method of the present embodiment.
- the "high boiling point" of the high boiling point compound as used herein does not refer to the absolute boiling point, but rather the compound extracted as a gas phase component from the thermal decomposition reactor, or the isocyanate compound (VIIa). It represents a compound that did not become a gas phase component under the same conditions as a compound extracted as a gas phase component in the distillation process.
- the high-boiling compounds often contain biuret groups, allophanate groups, isocyanurate groups, urea groups, carbodiimide groups, uretonimine groups, iminotrimer groups, and the like.
- the high-boiling-point compound includes a rearranged Fries product obtained by rearranging a carbamate group, a cyclized product of the rearranged Fries product, an oxycarbonyl Carbamate groups may be included.
- the high boiling point compound may contain an isocyanurate group, a carbodiimide group, or the like.
- each of the three nitrogen atoms of the isocyanurate group is R 71a in the general formula (VIIa) described later.
- the liquid phase component continuously withdrawn from the thermal decomposition reactor may contain the carbamate compound (Vc) and the hydroxy compound used in the step (x1).
- the liquid phase component continuously withdrawn from the separator in method (x2-2) may contain the isocyanate compound (VIIa), carbodiimide group, uretonimine group, isocyanurate group and iminotrimer group.
- a high-boiling compound by-produced in the process of producing the isocyanate compound (VIIa) by thermal decomposition using a carbonate ester as a solvent may contain an oxycarbonylcarbamate group.
- the liquid phase component preferably has a viscosity of 100 mPa at 150° C. from the viewpoint of transfer to the reactor in which the liquid phase component, the aromatic hydroxy compound, the active hydrogen-containing compound, and the catalyst react. • s or less is used.
- the viscosity of the liquid phase component can be measured by a known measuring instrument such as a capillary viscometer, a falling ball viscometer, or a rotary viscometer. Specifically, for example, the liquid phase component can be heated to a predetermined temperature in a nitrogen atmosphere and measured using a Brookfield viscometer.
- the viscosity is measured at a low temperature, and the logarithm of the viscosity and the reciprocal of the measurement temperature (absolute temperature) are plotted to calculate the viscosity at 150°C. good.
- the method (Y) is a method comprising the step (y1) of reacting the amine compound (IIa) with phosgene to produce the isocyanate compound (VIIa) while dehydrochlorinating.
- the reaction of the amine compound (IIa) with phosgene is carried out in solution or in the gas phase to produce carbamic acid chloride, thermally decompose the carbamic acid chloride, and separate the by-produced hydrogen chloride.
- An isocyanate compound (VIIa) is produced.
- the production of carbamic acid chloride and the thermal decomposition of carbamic acid chloride may be carried out in separate steps or may be carried out simultaneously in the same step.
- Phosgene is used as a mixture with a solvent.
- the solvent to be used is not particularly limited as long as it is inert to phosgene, N-substituted carbamic acid chloride, and isocyanate which is a decomposition product of the N-substituted carbamic acid chloride.
- the liquefied phosgene can be used in the reaction either as it is or regasified.
- the oxygen concentration in the raw materials other than phosgene used in the present embodiment is preferably 0.1% by mass or less.
- the reaction temperature is preferably 10°C or higher and 300°C or lower, more preferably 30°C or higher and 250°C or lower, and even more preferably 50°C or higher and 200°C or lower.
- the reaction pressure may be under reduced pressure, under atmospheric pressure, or under increased pressure.
- reaction solvent the same solvent as the solvent for dissolving phosgene described above can be used. Among them, it is preferable to use the same solvent as the solvent for dissolving phosgene described above. Chlorobenzene, orthodichlorobenzene, or isoamyl acetate is preferably used as the reaction solvent in consideration of the solubility and ease of handling of the starting compounds and products.
- the reactor is not particularly limited, and known reactors can be used.
- conventionally known reactors such as a stirring tank, a pressurized stirring tank, a reduced pressure stirring tank, a column reactor, a distillation column, a packed column, and a thin film distiller can be used in combination as appropriate.
- liquid phase component after recovering the isocyanate compound (VIIa) is used as the liquid phase component containing the high-boiling point compound that is the raw material for the recovery method of the present embodiment.
- the liquid phase component obtained by method (Y) may contain the solvent used in the reaction.
- the amine compound used in the method for producing the isocyanate compound (VIIa) those exemplified as the "amine compound (IIa)" described later as the amine compound recovered by the recovery method of the present embodiment are preferably used.
- the hydroxy compound used in the method for producing the isocyanate compound (VIIa) those exemplified in the "hydroxy compound” described later as the hydroxy compound recovered by the recovery method of the present embodiment are preferably used.
- Carbamate compound (Vc) is a compound represented by the following general formula (Vc).
- R 51c is a divalent to tetravalent aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have an ester group of 1 or more and 4 or less or a nitrogen atom, or an aliphatic hydrocarbon group of 6 or more carbon atoms. It is preferably an aromatic hydrocarbon group having a valence of 20 or less and having a valence of 2 or more and 3 or less.
- R 51c is preferably a group represented by any one of the above formulas (Ia-1) to (Ia-24), such as formulas (Ia-1), (Ia-2), ( A group represented by Ia-3), (Ia-14), (Ia-18), or (Ia-19) is more preferable.
- R52c is a monovalent organic group, which is a residue obtained by removing the hydroxyl group from a hydroxy compound.
- R 52c is preferably an aromatic hydrocarbon group having 6 or more and 20 or less carbon atoms, which may contain an oxygen atom.
- R 52c examples include phenyl group, naphthyl group, anthryl group, pyrenyl group, phenanthryl group, methylphenyl group (each isomer), ethylphenyl group (each isomer), propylphenyl group (each isomer ), butylphenyl group (each isomer), pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer), nonylphenyl group (each isomer), decylphenyl group (each isomer), undecylphenyl group (each isomer), dodecylphenyl group (each isomer), tridecylphenyl group (each isomer), tetradecylphenyl group (each isomer),
- Preferred carbamate compounds (Vc) include, for example, compounds represented by the following formulas (Vc-1) to (Vc-4).
- carbonic acid derivative examples include urea, N-unsubstituted carbamic acid esters, carbonate esters and the like.
- N-unsubstituted carbamates examples include N-unsubstituted ethyl carbamate, N-unsubstituted butyl carbamate, N-unsubstituted hexyl carbamate, N-unsubstituted octyl carbamate, and N-unsubstituted phenyl carbamate. is preferably used.
- N-unsubstituted means H 2 N-COOR (R is a hydrocarbon group), and a structure R' in which one hydrogen atom bonded to a nitrogen atom is substituted with a hydrocarbon group.
- -NH-COOR R and R' each independently represent a hydrocarbon group
- carbonate ester dimethyl carbonate, diethyl carbonate, dibutyl carbonate, dihexyl carbonate, dioctyl carbonate, diphenyl carbonate, di(methylphenyl) carbonate and the like are preferably used.
- urea is preferred.
- hydroxy compound (VIc) As the hydroxy compound recovered in the recovery method of the present embodiment, a compound represented by the following general formula (VIc) (hereinafter sometimes referred to as "hydroxy compound (VIc)”) is preferably used. As described above, the hydroxy compound can also be said to be a raw material for producing the isocyanate compound (VIIa). In the recovery method of the present embodiment, among the hydroxy compounds described later, hydroxy compounds having an aromatic group are preferably used as the aromatic hydroxy compound used in step (a1).
- R 61c is a monovalent monovalent organic group.
- R 61c is a monovalent organic group, preferably an aliphatic hydrocarbon group having 1 to 20 carbon atoms or an aromatic hydrocarbon group having 6 to 20 carbon atoms, which may contain an oxygen atom.
- Aliphatic hydrocarbon groups for R 61c include, for example, methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), pentyl group (each isomer), hexyl group (each isomer) , heptyl group (each isomer), octyl group (each isomer), nonyl group (each isomer), decyl group (each isomer), undecyl group (each isomer), dodecyl group (each isomer), tridecyl group (each isomer), tetradecyl group (each isomer), pentadecyl group (each isomer), hexadecyl group (each isomer), heptadecyl group (each isomer), octadecyl group (each isomer), nonadecyl (each iso
- the aliphatic hydrocarbon group which may contain an oxygen atom in R 61c includes, for example, a methoxymethyl group, a methoxyethyl group (each isomer), a methoxypropyl group (each isomer), a methoxybutyl group (each isomer), Methoxypentyl group (each isomer), methoxyhexyl group (each isomer), methoxyheptyl group (each isomer), methoxyoctyl group (each isomer), methoxynonyl group (each isomer), methoxydecyl group (each isomer), methoxyundecyl group (each isomer), methododecyl group (each isomer), methoxytridecyl group (each isomer), methoxytetradecyl group (each isomer), methoxypentadecyl group (each isomer is
- aromatic hydrocarbon group for R 61c examples include aryl groups such as phenyl group, naphthyl group, anthryl group, pyrenyl group and phenanthryl group; methylphenyl group (each isomer), ethylphenyl group (each isomer), Propylphenyl group (each isomer), butylphenyl group (each isomer), pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer), nonylphenyl group (each isomer), decylphenyl group (each isomer), undecylphenyl group (each isomer), dodecylphenyl group (each isomer), tridecylphenyl group (each isomer) , tetradec
- Examples of the aromatic hydrocarbon group which may contain an oxygen atom for R 61c include alkoxyaryl groups such as a methoxyphenyl group (each isomer) and an ethoxyphenyl group (each isomer).
- R 61c is preferably an aromatic group such as a phenyl group, naphthyl group, anthryl group, pyrenyl group, phenanthryl group, methylphenyl group (each isomer), ethylphenyl group (each isomer), propyl Phenyl group (each isomer), butylphenyl group (each isomer), pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer) isomer), nonylphenyl group (each isomer), decylphenyl group (each isomer), undecylphenyl group (each isomer), dodecylphenyl group (each isomer), tridecylphenyl group (each isomer), Tetradec
- Preferred hydroxy compounds (VIc) include, for example, ethanol, 1-butanol, aromatic hydroxy compounds represented by the following general formula (VIc-1) (hereinafter referred to as "aromatic hydroxy compounds (VIc-1)" There is) and the like.
- ring A 611c is an aromatic hydrocarbon ring having 6 to 20 carbon atoms;
- R 611c is a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an alkoxy group having 20 or less, an aryl group having 6 to 20 carbon atoms, an aryloxy group having 6 to 20 carbon atoms, an aralkyl group having 7 to 20 carbon atoms, an aralkyloxy group having 7 to 20 carbon atoms, or hydroxy
- R 611c may combine with Ring A 611c to form a ring structure, and n 611c is an integer of 1 or more and 10 or less.
- R611c Examples of the alkyl group having 1 to 20 carbon atoms in R 611c include methyl group, ethyl group, propyl group (each isomer), butyl group (each isomer), pentyl group (each isomer), hexyl group ( isomer), heptyl group (each isomer), octyl group (each isomer), nonyl group (each isomer), decyl group (each isomer), dodecyl group (each isomer), octadecyl group (each isomer) body) and the like.
- the alkoxy group having 1 to 20 carbon atoms in R 611c includes, for example, methoxy group, ethoxy group, propoxy group (each isomer), butyloxy group (each isomer), pentyloxy group (each isomer), hexyloxy group (each isomer), heptyloxy group (each isomer), octyloxy group (each isomer), nonyloxy group (each isomer), decyloxy group (each isomer), dodecyloxy group (each isomer), octadecyloxy group (each isomer) and the like.
- Examples of the aryl group having 6 to 20 carbon atoms in R 611c include a phenyl group and a naphthyl group.
- Examples of the aryl group having an alkyl group as a substituent for R 611c include a methylphenyl group (each isomer), an ethylphenyl group (each isomer), a propylphenyl group (each isomer), a butylphenyl group (each isomer isomer), pentylphenyl group (each isomer), hexylphenyl group (each isomer), heptylphenyl group (each isomer), octylphenyl group (each isomer), nonylphenyl group (each isomer), decylphenyl group (each isomer), biphenyl group (each isomer), dimethylphenyl group (each isomer), diethylphenyl group (each isomer), dipropylphenyl group (each isomer), dibutylphenyl group (each isomer
- Examples of the aryloxy group having 6 to 20 carbon atoms in R 611c include a phenoxy group, a methylphenoxy group (each isomer), an ethylphenoxy group (each isomer), a propylphenoxy group (each isomer), and a butylphenoxy group.
- each isomer pentylphenoxy group (each isomer), hexylphenoxy group (each isomer), heptylphenoxy group (each isomer), octylphenoxy group (each isomer), nonylphenoxy group (each isomer ), decylphenoxy group (each isomer), phenylphenoxy group (each isomer), dimethylphenoxy group (each isomer), diethylphenoxy group (each isomer), dipropylphenoxy group (each isomer), dibutylphenoxy group (each isomer), dipentylphenoxy group (each isomer), dihexylphenoxy group (each isomer), diheptylphenoxy group (each isomer), diphenylphenoxy group (each isomer), trimethylphenoxy group (each isomer isomer), triethylphenoxy group (each isomer), tripropy
- Examples of the aralkyl group having 7 to 20 carbon atoms in R 611c include a phenylmethyl group, a phenylethyl group (each isomer), a phenylpropyl group (each isomer), a phenylbutyl group (each isomer), and phenylpentyl. group (each isomer), phenylhexyl group (each isomer), phenylheptyl group (each isomer), phenyloctyl group (each isomer), phenylnonyl group (each isomer), and the like.
- Examples of the aralkyloxy group having 7 to 20 carbon atoms in R 611c include a phenylmethoxy group, a phenylethoxy group (each isomer), a phenylpropyloxy group (each isomer), and a phenylbutyloxy group (each isomer). , phenylpentyloxy group (each isomer), phenylhexyloxy group (each isomer), phenylheptyloxy group (each isomer), phenyloctyloxy group (each isomer), phenylnonyloxy group (each isomer) etc.
- Ring A 611c is an aromatic hydrocarbon ring having 6 to 20 carbon atoms. Ring A 611c may be monocyclic, polycyclic, or condensed.
- Ring A 611c examples include benzene ring, naphthalene ring, anthracene ring, phenanthrene ring, naphthacene ring, chrysene ring, pyrene ring, triphenylene ring, pentalene ring, azulene ring, heptalene ring, indacene ring, biphenylene ring, acenaphthylene ring, aceanthrylene ring, acephenanthrylene ring, and the like.
- the ring A 611c is preferably a benzene ring, a naphthalene ring, or an anthracene ring, more preferably a benzene ring.
- R 611c may also have substituents other than R 611c .
- Substituents other than R 611c include the same as those exemplified for R 611c .
- R 611c and substituents other than R 611c consist of different functional groups.
- n611c represents the number of substituents R611c and is an integer of 1 or more and 10 or less.
- compounds in which ring A 611c is a benzene ring include, for example, compounds represented by the following general formula (VIc-1-1) (hereinafter referred to as "aromatic hydroxy compound (VIc-1 -1)”) and the like.
- R 612c , R 613c , R 614c , R 615c , and R 616c are each independently the same as R 611c above.
- R 612c , R 613c , R 614c , R 615c and R 616c is preferably a hydrogen atom, and all of R 612c , R 613c , R 614c , R 615c and R 616c are hydrogen atoms Atoms are more preferred.
- Preferred aromatic hydroxy compounds (VIc-1-1) include, for example, phenol, 2-ethylphenol, 2-propylphenol (each isomer), 2-butylphenol (each isomer), 2-pentylphenol (each isomer isomer), 2-hexylphenol (each isomer), 2-heptylphenol (each isomer), 2-phenylphenol, 2,6-dimethylphenol, 2,4-diethylphenol, 2,6-diethylphenol, 2 ,4-dipropylphenol (each isomer), 2,6-dipropylphenol (each isomer), 2,4-dibutylphenol (each isomer), 2,4-dipentylphenol (each isomer), 2 ,4-dihexylphenol (each isomer), 2,4-diheptylphenol (each isomer), 2-methyl-6-ethylphenol, 2-methyl-6-propylphenol (each isomer), 2-methyl
- the amine compound (IIa) recovered in the recovery method of the present embodiment is a compound represented by the following general formula (IIa). As described above, the amine compound (IIa) can also be said to be a raw material for producing the isocyanate compound (VIIa).
- R 21a is a divalent to tetravalent aliphatic hydrocarbon group having 1 to 20 carbon atoms which may have an ester group of 1 or more and 4 or less or a nitrogen atom, or 6 or more carbon atoms. It is preferably an aromatic hydrocarbon group having a valence of 20 or less and having a valence of 2 or more and 3 or less.
- R 21a is preferably a group represented by any one of the above formulas (Ia-1) to (Ia-24), and the groups represented by the formulas (Ia-1), (Ia-2), ( A group represented by Ia-3), (Ia-14), (Ia-18), or (Ia-19) is more preferable.
- R 21a is an aliphatic hydrocarbon group
- specific examples of the amine compound (IIa) include aliphatic diamines, aliphatic triamines, and substituted cycloaliphatic polyamines.
- aliphatic diamines examples include ethylenediamine, diaminopropane (each isomer), diaminobutane (each isomer), diaminopentane (each isomer), diaminohexane (each isomer), and diaminodecane (each isomer). etc.
- aliphatic triamines examples include triaminohexane (each isomer), triaminononane (each isomer), triaminodecane (each isomer), 4-aminomethyl-1,8-octanediamine, and the like. be done.
- Substituted cycloaliphatic polyamines include, for example, diaminocyclobutane (each isomer), diaminocyclohexane (each isomer), 3-aminomethyl-3,5,5-trimethylcyclohexylamine (cis and/or trans isomer), methylenebis(cyclohexylamine), triaminocyclohexane (each isomer), and the like.
- R 21a is an aromatic group
- specific examples of the amine compound (IIa) include aromatic diamines and aromatic triamines.
- aromatic diamines examples include diaminobenzene (each isomer), diaminotoluene (each isomer), methylenedianiline (each isomer), diaminomesitylene (each isomer), diaminobiphenyl (each isomer), Diaminodibenzyl (each isomer), bis(aminophenyl)propane (each isomer), bis(aminophenyl)ether (each isomer), bis(aminophenoxyethane) (each isomer), diaminoxylene (each isomer) isomer), diaminoanisole (each isomer), diaminophenetole (each isomer), diaminonaphthalene (each isomer), diamino-methylbenzene (each isomer), diamino-methylpyridine (each isomer), diamino- Examples include methylnaphthalene (each iso
- aromatic triamines examples include triaminobenzene (each isomer), methanetriyltrianiline (each isomer), and the like.
- amine compound (IIa) examples include 2-aminoethyl acrylate, 2-methyl acrylate, -acrylic acid-2-aminoethyl ester, acrylic acid-2-aminopropyl ester, 2-methyl-acrylic acid-2-aminopropyl ester, acrylic acid-3-aminopropyl ester, 2-methyl-acrylic acid-3- Aminopropyl ester, 4-aminobutyl acrylate, 2-methyl-4-aminobutyl acrylate, 5-aminopentyl acrylate, 5-aminopentyl acrylate 2-methyl-acrylate, acrylic acid -6-aminohexyl ester, 2-methyl-acrylic acid-6-aminohexyl ester, acrylic acid-8-aminoctyl ester, 2-methyl-acrylic acid-8-aminoctyl ester, acrylic acid-10-aminodecy
- Preferred amine compounds (IIa) include, for example, diaminopentane (each isomer), diaminohexane (each isomer), 4-aminomethyl-1,8-octanediamine, lysine ethyl ester diamine, 3-aminomethyl-3 , 5,5-trimethylcyclohexylamine (cis and/or trans), methylenebis(cyclohexylamine), triaminocyclohexane (each isomer), diaminotoluene (each isomer), methylenedianiline (each isomer), tri Aminobenzene (each isomer), methanetriyltrianiline (each isomer), and the like.
- the isocyanate composition of the present embodiment includes an isocyanate compound, a carbonyl compound represented by the following general formula (I) (hereinafter sometimes referred to as "carbonyl compound (I)");
- the isocyanate compound and the carbonyl compound are different compounds.
- R 11 is a (n11+n12)-valent organic group
- R 12 is a monovalent organic group.
- n11 is an integer of 1 or more and 8 or less
- n12 is 0 or more and 7 or less.
- the sum of n11 and n12 is an integer of 2 or more and 8 or less.
- the carbonyl compound (I) and the modified isocyanate compound are by-products produced when the isocyanate compound is produced by the thermal decomposition reaction of the carbamate compound described later.
- the carbonyl compound (I) acts as a good solvent for the modified isocyanate compound, and the carbonyl compound (I) acts as a terminal blocker, and the modified isocyanate compound has a high molecular weight.
- the carbonyl compound (I) since the carbonyl compound (I) has a higher boiling point than the isocyanate compound and has fewer cross-linking points than the modified isocyanate compound, it can also serve as a solvent during purification of the isocyanate compound. Alternatively, it binds to a functional group such as a carbodiimide group to prevent modified products of isocyanate from binding to each other and increasing the molecular weight. These actions improve the operability during the purification of the isocyanate compound and enable recovery of the isocyanate compound at a high yield.
- a functional group such as a carbodiimide group
- ⁇ 3 ⁇ (molar amount of isocyanurate groups) + 2 ⁇ (molar amount of carbodiimide groups) + 3 ⁇ (molar amount of uretonimine groups) + 2 ⁇ (molar amount of allophanate groups) ⁇ / ( carbonyl compound molar amount) is 0.001 to 8.0, preferably 0.01 to 7.8, more preferably 0.1 to 7.5, and 1.0 to 7.0 The following are more preferable, and 2.0 or more and 6.5 or less are particularly preferable.
- the above values are measured by combining 1 H-NMR, liquid chromatography, and gas chromatography to measure the respective molar amounts of isocyanurate groups, carbodiimide groups, uretonimine groups, allophanate groups, and carbonyl compounds, and then using the above formula. It can be calculated by substituting values.
- the conditions described in Examples can be adopted.
- the content of the isocyanate compound is preferably 1% by mass or more and 99% by mass or less, more preferably 5% by mass or more and 99% by mass or less, relative to the total mass of the isocyanate composition. 99% by mass or less is even more preferable, 30% by mass or more and 99% by mass or less is even more preferable, 50% by mass or more and 99% by mass or less is even more preferable, and 70% by mass or more and 99% by mass or less is particularly preferable.
- the total content of the carbonyl compound (I) and the modified product of the isocyanate compound is preferably 1% by mass or more and 99% by mass or less, relative to the total mass of the isocyanate composition, and 1% by mass. More preferably 95% by mass or less, even more preferably 1% by mass or more and 90% by mass or less, still more preferably 1% by mass or more and 70% by mass or less, even more preferably 1% by mass or more and 50% by mass or less, 1% by mass More than 30% by mass or less is particularly preferable.
- the content of the carbonyl compound (I) is preferably 1% by mass or more and 99% by mass or less, more preferably 3% by mass or more and 95% by mass or less, relative to the total mass of the isocyanate composition.
- 5% by mass or more and 90% by mass or less is even more preferable, 7% by mass or more and 80% by mass or less is still more preferable, 10% by mass or more and 70% by mass or less is even more preferable, and 10% by mass or more and 60% by mass or less is particularly preferable.
- the content of each of the above compounds in the isocyanate composition of the present embodiment can be calculated, for example, by combining 1 H-NMR liquid chromatography and gas chromatography analysis. As the detailed conditions for the above analysis, the conditions described in Examples can be adopted.
- the isocyanate composition of the present embodiment prevents by-products from sticking to the apparatus when producing an isocyanate compound without using phosgene, and improves the yield of the isocyanate compound. can be done.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
本願は、2021年11月8日に、日本に出願された特願2021-182191号、特願2021-182194号、及び特願2021-182195号、並びに、2021年12月8日に、日本に出願された特願2021-199645号に基づき優先権を主張し、その内容をここに援用する。
〈1〉 第一級アミン化合物と、炭酸誘導体と、を反応させて、副生する、N-置換カルバメート化合物よりも沸点の低い化合物を抜き出しながら、N-置換カルバメート化合物を得る工程(1)と、
前記工程(1)で得られた前記N-置換カルバメート化合物を含む反応液を用いて、非プロトン性溶媒存在下で、前記N-置換カルバメート化合物の熱分解を行い、副生するヒドロキシ化合物を抜き出しながら、イソシアネート化合物を得る工程(2)と、
前記工程(2)で得られた前記イソシアネート化合物を含む反応液から前記イソシアネート化合物と、前記非プロトン性溶媒と、を分離する工程(3)と、
前記工程(3)で得られた前記イソシアネート化合物を含む画分から前記イソシアネート化合物よりも沸点が高い成分を除去して、イソシアネート化合物を精製する工程(4)と、
を含む、イソシアネート化合物の製造方法。
〈2〉 前記工程(3)で分離された前記非プロトン性溶媒を含む画分、又は、前記工程(4)で除去された前記イソシアネート化合物よりも沸点が高い成分を、アルカリと水との共存下で、加水分解し、前記第一級アミン化合物及び前記ヒドロキシ化合物を得る工程(5)を更に含む、〈1〉記載のイソシアネート化合物の製造方法。
〈3〉 前記工程(1)において、
ヒドロキシ化合物存在下で、第一級アミン化合物と、前記炭酸誘導体として尿素又は尿素誘導体と、を反応させて、副生する、N-置換カルバメート化合物よりも沸点の低い化合物を抜き出しながら、N-置換カルバメート化合物を得る、又は、
第一級アミン化合物と、前記炭酸誘導体として炭酸エステルと、を反応させて、副生する、N-置換カルバメート化合物よりも沸点の低い化合物としてヒドロキシ化合物を抜き出しながら、N-置換カルバメート化合物を得る、〈1〉又は〈2〉に記載のイソシアネート化合物の製造方法。
〈4〉 前記工程(2)において、副生するヒドロキシ化合物を気相に抜き出しながら、前記イソシアネート化合物を含む液相を得る、〈1〉~〈3〉のいずれか一つに記載のイソシアネート化合物の製造方法。
〈5〉 前記工程(1)及び前記工程(2)を、槽型反応器、蒸留塔、管型蒸発器、薄膜蒸発器、及びフォーリングフィルム型蒸発器からなる群より選ばれる1種以上の反応器を用いて行う、〈1〉~〈4〉のいずれか一つに記載のイソシアネート化合物の製造方法。
〈6〉 前記工程(1)及び前記工程(2)のうち、少なくともいずれか一方を反応蒸留法により行う、〈1〉~〈5〉のいずれか一つに記載のイソシアネート化合物の製造方法。
〈7〉 前記工程(2)で抜き出された前記ヒドロキシ化合物を前記工程(1)に循環して再利用する、〈1〉~〈6〉のいずれか一つに記載のイソシアネート化合物の製造方法。
〈8〉 前記工程(3)で分離された前記非プロトン性溶媒を前記工程(2)に循環して再利用する、〈1〉~〈7〉のいずれか一つに記載のイソシアネート化合物の製造方法。
〈9〉 前記非プロトン性溶媒が炭酸エステルである、〈1〉~〈8〉のいずれか一つに記載のイソシアネート化合物の製造方法。
〈10〉 前記工程(4)において、前記イソシアネート化合物を含む画分の質量に対して、1質量ppm以上50質量%以下の、下記一般式(I)で表されるカルボニル化合物存在下で、前記イソシアネート化合物の精製を行う、〈1〉~〈9〉のいずれか一つに記載のイソシアネート化合物の製造方法。
〈14〉 前記活性水素を有さないアミン化合物が、第三級アミノ基、含窒素芳香族基、アミジン基、及びグアニジン基からなる群より選ばれる1種以上の官能基を有する、〈13〉に記載のカルバメート化合物の製造方法。
〈15〉 前記活性水素を有さないアミン化合物が、第三級アミノ基を1以上有し、且つ、炭素数が3以上85以下の化合物である、〈14〉に記載のカルバメート化合物の製造方法。
〈16〉 前記活性水素を有さないアミン化合物が、第三級アミノ基を1以上6以下有し、且つ、炭素数が3以上30以下の化合物である、〈15〉に記載のカルバメート化合物の製造方法。
〈17〉 前記活性水素を有さないアミン化合物が、N,N’-ジメチルアニリン、N,N’-ジエチルアニリン、N-メチル-N’-エチルアニリン、N,N’-ジメチルアミノピリジン、N,N,N’,N’-テトラメチルフェニレンジアミン、メチレンビス(N,N’-ジメチルアニリン)、トリエチルアミン、エチルジイソプロピルアミン、N-メチルモルホリン、N-メチルピペリジン、キヌクリジン、N,N’-ジメチルピペラジン、トリエチレンジアミン、N,N,N’ N’-テトラメチルエチレンジアミン、N,N,N’ N’-テトラメチルヘキサンジアミン、N,N,N’ N’-テトラメチルキシリレンジアミン、ペンタメチルジエチレントリアミン、ビス(2-モルホリノエチル)エーテル、ヘキサヒドロ-1,3,5-トリス(3-ジメチルアミノプロピル)-1,3,5-トリアジン、又はヘキサメチレンテトラミンである、〈16〉に記載のカルバメート化合物の製造方法。
〈18〉 前記活性水素を有さないアミン化合物が、含窒素芳香族基を1以上有し、且つ、炭素数が3以上85以下の化合物である、〈14〉に記載のカルバメート化合物の製造方法。
〈19〉 前記含窒素芳香族基が、置換又は無置換の、ピリジル基、イミダゾリル基、ピラゾリル基、キノリル基、イソキノリル基、オキサゾリル基、チアゾリル基、ピリダジル基、又はピリミジル基、ピラジル基である、〈18〉に記載のカルバメート化合物の製造方法。
〈20〉 前記活性水素を有さないアミン化合物が、ピリジン、ピコリン、ルチジン、コリジン、1-メチルイミダゾール、1-メチルピラゾール、キノリン、イソキノリン、メチルキノリン、オキサゾール、チアゾール、ピリダジン、ピリミジン、又はピラジンである、〈19〉に記載のカルバメート化合物の製造方法。
〈21〉 前記活性水素を有さないアミン化合物が、下記一般式(VIII-1)で表されるアミジン基含有化合物である、〈14〉に記載のカルバメート化合物の製造方法。
〈23〉 前記アミジン基含有化合物が、1,2-ジメチル-1,4,5、6-テトラヒドロピリミジン、1,8-ジアザビシクロ-[5.4.0]ウンデカ-7-エン、又は1,5-ジアザビシクロ-[4.3.0]ノナ-5-エンである、〈22〉に記載のカルバメート化合物の製造方法。
〈24〉 前記活性水素を有さないアミン化合物が、下記一般式(VIII-2)で表されるグアニジン基含有化合物である、〈14〉に記載のカルバメート化合物の製造方法。
〈26〉 前記グアニジン基含有化合物が、ペンタメチルグアニジン、又は7-メチル-1,5,7-トリアザビシクロ[4.4.0]デカ-5-エンである、〈25〉に記載のカルバメート化合物の製造方法。
〈27〉 前記第1級アミン化合物が、脂肪族又は芳香族第1級ポリアミン化合物である、〈13〉~〈26〉のいずれか一つに記載のカルバメート化合物の製造方法。
〈28〉 前記ヒドロキシ化合物が、芳香族ヒドロキシ化合物である、〈13〉~〈27〉のいずれか一つに記載のカルバメート化合物の製造方法。
〈29〉 〈13〉~〈28〉のいずれか一つに記載のカルバメート化合物の製造方法により得られたカルバメート化合物を熱分解する熱分解工程を含む、イソシアネート化合物の製造方法。
前記液相成分と、芳香族ヒドロキシ化合物と、活性水素含有化合物と、触媒と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a)を含む、回収方法。
前記液相成分と芳香族ヒドロキシ化合物を混合させる工程(a1);
前記工程(a1)で得られた混合物と、活性水素含有化合物と、触媒と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a2)
〈32〉 前記工程(a2)が、以下の工程(a2-1)及び工程(a2-2)を含む、〈31〉に記載の回収方法。
前記工程(a1)で得られた混合物と、活性水素含有化合物としてアミン化合物と、触媒と、を混合させる工程(a2-1);
前記工程(a2-1)で得られた混合物と、活性水素含有化合物として水と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a2-2)
〈33〉 前記活性水素含有化合物が水である、〈30〉~〈32〉のいずれか一つに記載の回収方法。
〈34〉 前記活性水素含有化合物が水及び第一級アミン化合物である、〈30〉~〈32〉のいずれか一つに記載の回収方法。
〈35〉 前記第一級アミン化合物が前記一般式(IIa)で表されるアミン化合物である、〈34〉に記載の回収方法。
〈36〉 前記一般式(VIIa)で表されるイソシアネート化合物の製造方法において、炭酸誘導体と、ヒドロキシ化合物と、前記一般式(IIa)で表されるアミン化合物と、を原料として用いる、〈30〉~〈35〉のいずれか一つに記載の回収方法。
〈37〉 前記液相成分が、炭酸誘導体と、ヒドロキシ化合物と、前記一般式(IIa)で表されるアミン化合物と、から生成されたカルバメート化合物を含む反応液を熱分解反応に供して、生成された前記一般式(VIIa)で表されるイソシアネート化合物を含む組成物を、蒸留装置に供給し、前記一般式(VIIa)で表されるイソシアネート化合物を気相成分として分離する際に、前記蒸留装置から抜き出される液相成分である、〈30〉~〈36〉のいずれか一つに記載の回収方法。
〈38〉 前記液相成分が、下記式(IX-1)で表される基、下記式(IX-2)で表される基、下記式(IX-3)で表される基、下記式(IX-4)で表される基、下記式(IX-5)で表される基、下記式(IX-6)で表される基、下記式(IX-7)で表される基、下記式(IX-8)で表される基、下記式(IX-9)で表される基、下記式(IX-10)で表される基、下記式(IX-11)で表される基、及び下記式(IX-12)で表される基からなる群より選ばれる1種以上の官能基を有する化合物を含む、〈30〉~〈37〉のいずれか一つに記載の回収方法。
〈40〉 前記触媒が、アルカリ金属の水酸化物及び酸化物、アルカリ土類金属の水酸化物及び酸化物、第三級アミン化合物、第12族の金属酸化物、第13族の金属酸化物、並びに、第14族の金属酸化物からなる群より選ばれる1種以上の化合物である、〈30〉~〈39〉のいずれか一つに記載の回収方法。
〈41〉 前記触媒が、アルカリ金属の水酸化物及び酸化物、並びに、アルカリ土類金属の水酸化物及び酸化物からなる群より選ばれる1種以上の化合物である、〈40〉に記載の回収方法。
〈42〉 前記触媒が、アルカリ金属の水酸化物及び酸化物である、〈41〉に記載の回収方法。
〈43〉 以下の工程(b)及び工程(c)を更に含む、〈30〉~〈42〉のいずれか一つに記載の回収方法。
前記一般式(IIa)で表されるアミン化合物を含む反応液から前記一般式(IIa)で表されるアミン化合物を分離する工程(b);
前記一般式(IIa)で表されるアミン化合物を精製する工程(c)
〈44〉 以下の工程(d)を更に含む、〈43〉に記載の回収方法。
前記工程(c)で精製された前記一般式(IIa)で表されるアミン化合物を、前記一般式(VIIa)で表されるイソシアネート化合物の製造方法において、原料として再利用する工程(d)
〈45〉 以下の工程(e)及び(f)を更に含む、〈30〉~〈44〉のいずれか一つに記載の回収方法。
前記一般式(IIa)で表されるアミン化合物を含む反応液からヒドロキシ化合物を分離する工程(e);
前記ヒドロキシ化合物を精製する工程(f)
〈46〉 以下の工程(g)を更に含む、〈45〉に記載の回収方法。
前記工程(f)で精製された前記ヒドロキシ化合物を、前記一般式(VIIa)で表されるイソシアネート化合物の製造方法において、原料として再利用する工程(g)
〈47〉 以下の工程(h)を更に含む、〈45〉に記載の回収方法。
前記工程(b)において前記一般式(IIa)で表されるアミン化合物を分離した後の残留液、前記工程(c)において前記一般式(IIa)で表されるアミン化合物を精製した後の残留液、前記工程(e)において前記ヒドロキシ化合物を分離した後の残留液、及び前記工程(f)において前記ヒドロキシ化合物を精製した後の残留液からなる群より選ばれる1種以上の残留液を、前記工程(a)において再利用する工程(h)
下記一般式(I)で表されるカルボニル化合物と、
前記イソシアネート化合物のイソシアネート基の一部が、イソシアヌレート基、カルボジイミド基、ウレトンイミン基、及びアロファネート基からなる群より選ばれる1種以上の官能基に変換された、イソシアネート化合物の変性物と、
を含む、イソシアネート組成物であって、
前記イソシアネート化合物と前記カルボニル化合物は異なる化合物であり、
{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値が0.001以上8.0以下である、イソシアネート組成物。
前記R12が、酸素原子を含んでもよい、炭素数6以上20以下の1価の芳香族炭化水素基である、〈48〉に記載のイソシアネート組成物。
〈50〉 前記イソシアネート化合物が下記一般式(VIIb)で表される化合物である、〈48〉又は〈49〉に記載のイソシアネート組成物。
前記イソシアネート化合物の含有量が1質量%以上99質量%以下であり、且つ、
前記カルボニル化合物及び前記イソシアネート化合物の変性物の合計含有量が1質量%以上99質量%以下である、〈48〉~〈51〉のいずれか一つに記載のイソシアネート組成物。
〈53〉 前記イソシアネート組成物の総質量に対して、それぞれ2.0質量ppm以上99質量%以下の、下記一般式(XI)で表されるカルバメート基含有化合物及び下記一般式(III-1a)で表される炭酸エステルからなる群より選ばれる1種以上の化合物を更に含有する、〈48〉~〈52〉のいずれか一つに記載のイソシアネート組成物。
〈55〉 〈48〉~〈54〉のいずれか一つに記載のイソシアネート組成物を蒸留精製し、連続的に気相成分として、前記イソシアネート化合物を回収することを含む、イソシアネート化合物の製造方法。
本実施形態のイソシアネート化合物の製造方法(以下、単に「本実施形態の製造方法」と称する場合がある)は、以下に示す工程(1)~工程(4)を含む。
第一級アミン化合物と、炭酸誘導体と、を反応させて、副生する、N-置換カルバメート化合物よりも沸点の低い化合物(以下、「低沸化合物」ともいう)を抜き出しながら、N-置換カルバメート化合物を得る工程(1);
前記工程(1)で得られた前記N-置換カルバメート化合物を含む反応液を用いて、非プロトン性溶媒存在下で、前記N-置換カルバメート化合物の熱分解を行い、副生する前記ヒドロキシ化合物を抜き出しながら、イソシアネート化合物を得る工程(2);
前記工程(2)で得られた前記イソシアネート化合物を含む反応液から前記イソシアネート化合物と、前記非プロトン性溶媒と、を分離する工程(3);
前記工程(3)で得られた前記イソシアネート化合物を含む画分から前記イソシアネート化合物よりも沸点が高い成分(以下、単に「高沸点成分」と称する場合がある)を除去して、イソシアネート化合物を精製する工程(4)。
工程(1)では、第一級アミン化合物と、炭酸誘導体と、を反応させて、副生する低沸化合物を抜き出しながら、N-置換カルバメート化合物を得る。
上記ウレイド中間体を介したN-置換カルバメート化合物の生成反応と、副生するアンモニアの抜き出しを同時に行える方法であれば特に限定されないが、工程(1)は、蒸留塔、管型蒸発器、薄膜蒸発器、又はフォーリングフィルム型蒸発器を用いて行うことが好ましく、蒸留塔、薄膜蒸発器、又はフォーリングフィルム型蒸発器がより好ましく、蒸留塔、又はフォーリングフィルム型蒸発器がさらに好ましく、蒸留塔を用いることが特に好ましい。蒸留塔を用いる際は、当該工程で化学変化や反応を伴うことから、反応蒸留法と呼ばれる手法により行うことが好ましい。反応蒸留法とは、ヒドロキシ化合物と、第一級アミン化合物と、尿素及び尿素誘導体からなる群より選ばれる1種以上の化合物との混合物による上述した反応を反応器内で行いながら、副生するアンモニアを、蒸留によって上記反応器から抜き出す方法である。アンモニアは、主生成物であるN-置換カルバメート化合物よりも沸点が低いことから、気相成分として副生成物であるアンモニアを抜き出すことで、液相成分として、N-置換カルバメート化合物を含む留分を効率良く得ることができる。
触媒を使用する場合には、反応を短時間で完結でき、反応温度を低くすることができる。
触媒を使用する場合には、例えば、スズ、鉛、銅、チタン等の有機金属化合物や無機金属化合物;アルカリ金属又はアルカリ土類金属のアルコラートであって、リチウム、ナトリウム、カリウム、カルシウム若しくはバリウムのメチラート、エチラート又はブチラート(各異性体)等の塩基性触媒等を使用することができる。
ウレイド化反応については、攪拌槽、加圧式攪拌槽、減圧式攪拌槽、塔型反応器、連続攪拌型反応器、蒸留塔、充填塔、プラグフロー反応器、薄膜蒸留器等の、従来公知の反応器を適宜組み合わせて使用でき、攪拌槽、加圧式攪拌槽、減圧式攪拌槽、塔型反応器、連続攪拌型反応器、又はプラグフロー反応器が好ましく、製造効率の観点から、連続攪拌型反応器、又はプラグフロー反応器を用いて連続法で行うことがより好ましい。
工程(2)では、工程(1)で得られたN-置換カルバメート化合物を含む反応液を用いて、非プロトン性溶媒存在下で、N-置換カルバメート化合物の熱分解を行い、副生するヒドロキシ化合物を抜き出しながら、イソシアネート化合物を得る。
イソシアネート化合物と、ヒドロキシ化合物の標準沸点の差が上記範囲であることで、熱分解後のヒドロキシ化合物を気相により効率的に抜き出すことができ、生成するイソシアネート化合物とヒドロキシ化合物との逆反応をより低減することができ、イソシアネート化合物の収率をより向上させることができる。
イソシアネート化合物と、非プロトン性溶媒の標準沸点の差が上記上限値以下であることで、熱分解後のヒドロキシ化合物を気相に抜き出す際、非プロトン性溶媒の同伴をより抑制でき、液相中のイソシアネートの高濃度化を防ぐことで、生成するイソシアネートとヒドロキシ化合物との逆反応、原料であるカルバメートとの変性反応及びイソシアネート同士の多量化をより低減することができる。一方、上記下限値以上であることで、後述する工程(3)において、非プロトン性溶媒とイソシアネート化合物とをより効率的に分離することができ、イソシアネート化合物の収率をより向上させることができる。
一方で、イソシアネート化合物の標準沸点の上限は特に限定されないが、450℃以下であることが好ましく、430℃以下であることがより好ましく、420℃以下であることがさらに好ましい。
イソシアネート化合物の標準沸点が上記下限値以上であることで、工程(2)において副生成物であるヒドロキシ化合物とより分離しやすい傾向がみられる。一方で、イソシアネート化合物の標準沸点が上記上限値以下であることで、後述する工程(3)及び工程(4)において非プロトン性溶媒や高沸点成分とより分離しやすい傾向がみられる。
非プロトン性溶媒の使用量が上記下限値以上であることで、生成されるイソシアネート化合物の濃度がより下がり、熱分解時のイソシアネート化合物同士の反応によるヌレートの生成、イソシアネート化合物と原料N-置換カルバメート化合物の反応によるアロファネート生成をより抑制することができ、イソシアネート化合物の熱分解収率をより向上し、且つ、ポリマーの析出をより抑制することができる。一方、非プロトン性溶媒の使用量が上記上限値以下であることで、同じ装置でのイソシアネート化合物の生産効率をより向上させることが可能となる。また、単一の非プロトン性溶媒を用いて反応させても、複数種の非プロトン性溶媒を併用してもよく、反応条件に合わせて任意に選択することができる。
熱分解温度が上記下限値以上であることで、N-置換カルバメート化合物の熱分解反応がより進行し、温度が高いほど熱分解速度が向上し、短時間でN-置換カルバメート化合物の熱分解が完了する。一方、上記上限値以下であることでイソシアネートからヌレート等への変性反応をより抑制できる。さらに、ヌレート化によって生成するイソシアネートのポリマーの生成量をより低減できるため、反応装置のへの付着や配管の閉塞がより抑制でき、運転性もより改善できる。
反応時間は、(連続法の場合は滞留時間)に、特に制限はなく、0.001時間以上100時間以下であることが好ましく、0.005時間以上50時間以下であることがより好ましく、0.01時間以上10時間であることがさらに好ましい。
工程(3)では、工程(2)で得られたイソシアネート化合物を含む反応液からイソシアネート化合物と、非プロトン性溶媒と、を分離する。過剰に含まれる非プロトン性溶媒を分離することで反応液量を減らし、各工程の設備を小さくすることで生産性の改善と設備費を低減することができる。
温度としては、イソシアネート化合物の変性を抑制する観点から、300℃以下であることが好ましく、20℃以上290℃以下の範囲であることがより好ましく、30℃以上280℃以下の範囲であることがさらに好ましい。
工程(4)では、工程(3)で得られたイソシアネート化合物を含む画分からイソシアネート化合物よりも沸点が高い成分(高沸点成分)を除去して、イソシアネート化合物を精製する。
温度としては、イソシアネート化合物の変性を抑制する観点から、250℃以下であることが好ましく、20℃以上240℃以下の範囲であることがより好ましく、30℃以上230℃以下の範囲であることがさらに好ましい。
本実施形態の製造方法は、上述した工程(1)~工程(4)に加えて、以下に示す工程を(5)を更に含むことが好ましい。
工程(3)で分離された非プロトン性溶媒を含む画分、又は、工程(4)で除去されたイソシアネート化合物よりも沸点が高い成分(高沸点成分)を、アルカリと水との共存下で、加水分解し、前記第一級アミン化合物及び前記ヒドロキシ化合物を得る工程(5)。
水の使用量が上記上限値以下であることで、多量の高温高圧水が引き起こすイオン積増大に伴う酸性雰囲気に起因する好ましくない反応、例えば、第一級アミン化合物の脱アンモニア反応によって内部環化した副生物であるイミン化合物の生成等をより抑制することができる。また、続く脱水工程におけるエネルギー負荷もより低減することができ、製造における消費エネルギーの抑制及び装置の小型化が可能である。一方、水の使用量が上記下限値以上であることで、高沸点成分の分解反応がより良好に進行する。
中でも、アルカリとしては、アルカリ金属及びアルカリ土類金属の水酸化物及び酸化物が好ましく、水酸化ナトリウム、水酸化カリウム等のアルカリ金属の水酸化物がより好ましい。
しかしながら、反応温度が上記範囲であることで、ウレア結合に代表される架橋性分子の分解がより良好に進行し、装置内のスケール発生をより抑制できるため、連続運転性を保持しつつ、第一級アミン化合物をより効率的に回収できる。
工程(5a)では、加水分解反応後の反応液から第一級アミン化合物を分離する。第一級アミン化合物の分離方法は、公知の方法を用いることができ、例えば、蒸留分離、液-液相分離、固液分離、膜分離等が挙げられる。工程(5a)では、上記工程(5)において連続的に排出された液相成分(反応液)から第一級アミン化合物を連続的に分離することもできる。
工程(5b)では、上記工程(5a)で分離された第一級アミン化合物を精製する。第一級アミン化合物を精製する方法は、公知の方法を用いることができ、例えば、蒸留分離、液-液相分離、固液分離、膜分離等が挙げられる。工程(5b)では、工程(5a)において連続的に分離された第一級アミン化合物を連続的に精製することもできる。
工程(5c)では、加水分解反応後の反応液から、上記工程(1)において原料となるヒドロキシ化合物を分離する。ヒドロキシ化合物の分離方法は、公知の方法を用いることができ、例えば、蒸留分離、液-液相分離、固液分離、膜分離等が挙げられる。工程(5c)では、上記工程(5)において連続的に排出された気相成分からヒドロキシ化合物を連続的に分離することもできる。
工程(5d)では、上記工程(5c)で分離されたヒドロキシ化合物を精製する。ヒドロキシ化合物を精製する方法は、公知の方法を用いることができ、例えば、蒸留分離、液-液相分離、固液分離、膜分離等が挙げられる。工程(5d)では、上記工程(5c)において連続的に分離されたヒドロキシ化合物を連続的に精製することもできる。
すなわち、本実施形態の製造方法は、以下に示す工程(7)を更に含むことが好ましい。
前記工程(5)で回収された第一級アミン化合物及びヒドロキシ化合物を前記工程(1)に再使用する工程(7)。
工程(7)では、工程(5)で回収された第一級アミン化合物及びヒドロキシ化合物を、上記工程(1)に再利用する。
その際に、上記工程(1)におけるN-置換カルバメート化合物の製造や、得られたN-置換カルバメート化合物及び続く工程で得られるイソシアネート化合物の品質に影響を与える虞があることから、上記工程(5)(具体的には、上記工程(5b)及び上記工程(5d))において、第一級アミン化合物の総質量に対して、金属成分の含有量が1000質量ppm以下及びハロゲン原子の含有量が1000質量ppm以下となるように第一級アミン化合物及びヒドロキシ化合物をそれぞれ蒸留回収することが好ましい。
本実施形態の製造方法は、上述した工程(1)~工程(5)に加えて、その他の工程を更に含むことができる。
工程(1a)では、工程(1)で得られたN-置換カルバメート化合物を濃縮する。すなわち、工程(1a)は、N-置換カルバメート化合物濃縮工程ということもできる。
N-置換カルバメート化合物の濃縮方法としては、特に限定されないが、生産性やプロセス設計の容易さの観点から、公知の蒸留分離が好ましい。
工程(6)では、工程(2)で抜き出された前記ヒドロキシ化合物を前記工程(1)に循環して再利用し、且つ、前記工程(3)で分離された前記非プロトン性溶媒を前記工程(2)に循環して再利用する。すなわち、工程(6)は、溶媒回収工程又は溶媒再利用工程ということができる。
本実施形態のイソシアネート化合物の製造方法において用いる原料及び生成物(中間生成物も含む)について、以下に詳細を説明する。
第一級アミン化合物としては、例えば、以下の一般式(II)で表される化合物(以下、「第一級アミン化合物(II)」と称する場合がある)等が挙げられる。
R21は、n21価の有機基であり、炭素数3以上85以下の有機基が好ましく、炭素数3以上30以下の有機基がより好ましい。
n21は、アミノ基の数を表し、2以上の整数であり、2以上10以下の整数であることが好ましく、3以上の整数であることがより好ましく、3であることがさらに好ましい。
ここでいう、アミノ酸誘導体は、アミノ酸を原料として合成される化合物という意味である。後述するように、アミノ酸としては天然アミノ酸であってもよく、合成アミノ酸であってもよい。また、当該誘導体は誘導体単体であっても、無機酸塩の形であってもよい。
また、第1級アミノ基、スルフィド結合、エーテル結合、及びジスルフィド結合からなる群から選ばれる1種以上を含んでもよい、炭素数1以上15以下の1価の脂肪族炭化水素基若しくは炭素数6以上15以下の1価の芳香族基、脂肪族炭化水素基と芳香族基とが結合した、第1級アミノ基、スルフィド結合、エーテル結合、及びジスルフィド結合からなる群から選ばれる1種以上を含んでもよい、炭素数7以上15以下の基、下記一般式(II-2a)又は(II-2b)で表される基、又は水素原子であることがより好ましい。
一般式(II-2a)中、n222は、0以上5以下の整数である。
一般式(II-2a)中、波線は結合手を表す。
一般式(II-2b)中、n223は、0以上5以下の整数である。
一般式(II-2b)中、波線は結合手を表す。
一般式(II-2d)中、n223は、1以上9以下の整数である。
一般式(II-2e)中、n224は、0以上9以下の整数である。
一般式(II-2c)~(II-2e)中、波線は結合手を表す。
一般式(II-2-1)~(II-2-4)中、R227は、R222と同じである。
特に好ましく用いられるアミノ酸は、脂肪族のモノアミノモノカルボン酸、ジアミノモノカルボン酸、モノアミノジカルボン酸、ジアミノジカルボン酸等である。これらのアミノ酸が環化して形成するラクタムもまた好ましく用いられる。
具体的には、メタノール、エタノール、プロパノール、ブタノール、ペンタノール、ヘキサノール、オクタノール、デカノール、シクロペンタノール、シクロヘキサノール等が挙げられる。なお、これらの化合物が異性体を含む場合は、該異性体も使用できる。
ヒドロキシ化合物としては、アルコール類及び芳香族ヒドロキシ化合物類が挙げられる。
R61における炭素数1以上20以下のアルキル基としては、例えば、メチル基、エチル基、プロピル基(各異性体)、ブチル基(各異性体)、ペンチル基(各異性体)、ヘキシル基(各異性体)、ヘプチル基(各異性体)、オクチル基(各異性体)、ノニル基(各異性体)、デシル基(各異性体)、ドデシル基(各異性体)、オクタデシル基(各異性体)等が挙げられる。
環A61は、炭素数6以上20以下の芳香族炭化水素環である。環A61は、単環であってもよく、多環であってもよく、縮合環であってもよい。
環A61として具体的は、例えば、ベンゼン環、ナフタレン環、アントラセン環、フェナントレン環、ナフタセン環、クリセン環、ピレン環、トリフェニレン環、ペンタレン環、アズレン環、ヘプタレン環、インダセン環、ビフェニレン環、アセナフチレン環、アセアントリレン環、アセフェナントリレン環等が挙げられる。中でも、環A11としては、ベンゼン環、ナフタレン環、又はアントラセン環が好ましく、ベンゼン環がより好ましい。
n61は、置換基R61の数を示し、1以上10以下の整数である。
炭酸誘導体としては、例えば、尿素、尿素誘導体、炭酸エステル等が挙げられる。炭酸エステルの詳細は後述する。
R41における1価の有機基としては、後述するR52と同じものが例示される。中でも、炭素数1以上20以下の脂肪族炭化水素基が好ましく、炭素数1以上10以下のアルキル基がより好ましい。
N-置換カルバメート化合物としては、例えば、一般式(V)で表される化合物(以下、「カルバメート化合物(V)」と称する場合がある)等が挙げられる。
一般式(V)中、R51は、2価以上の有機基であり、2価以上10価以下の有機基であることが好ましい。中でも、R51としては、1以上4以下のエステル基又は窒素原子を有してもよい、炭素数1以上20以下の2価以上4価以下の脂肪族炭化水素基、又は、1以上4以下のエステル基又は窒素原子を有してもよい、炭素数6以上20以下の2価以上3価以下の芳香族基であることが好ましい。
一般式(V)中、R52は1価の有機基である。中でも、R52としては、酸素原子を含んでもよい、炭素数1以上20以下の脂肪族炭化水素基又は炭素数6以上20以下の芳香族炭化水素基が好ましい。
一般式(V)中、n51はカルバメート基の数を表し、2以上の整数であり、2以上10以下の整数であることが好ましく、3以上の整数であることがより好ましく、3であることがさらに好ましい。
非プロトン性溶媒としては、イソシアネートと反応しないという観点から、炭酸誘導体、エーテル、炭化水素、ケトン、又はこれらの混合物が好ましく、炭酸誘導体、炭化水素、ケトン、又はこれらの混合物がより好ましく、炭酸誘導体が更に好ましい。
本明細書において、炭酸誘導体とは、カルボニル基を有する化合物を指す。
R31及びR32は互いに同一であってもよく、異なっていてもよいが、同一であることが好ましい。
R311及びR312は互いに同一であってもよく、異なっていてもよいが、同一であることが好ましい。
アリール基が有する置換基としては、アルキル基、アルコキシ基等が挙げられる。
イソシアネート化合物としては、例えば、一般式(VII)で表される化合物(以下、「イソシアネート化合物(VII)」と称する場合がある)等が挙げられる。
カルボニル化合物(I)は、工程(2)のN-置換カルバメート化合物を熱分解してイソシアネート化合物を生成する際に副生する副生成物である。当該化合物(I)はイソシアネート末端又はN-置換カルバメート末端を有する化合物と炭酸エステルが熱分解条件における降温等の反応条件下、分子間相互作用から反応が生起され、炭酸エステル骨格に各末端基が挿入する反応が進行することにより、当該カルボニル化合物(I)が生成すると考えられる。本実施形態の製造方法において、カルボニル化合物(I)が、イソシアネート化合物の変性物の良溶媒として働き、且つ、カルボニル化合物(I)が末端封止剤として働き、イソシアネート化合物の変性物の高分子量化を抑制することで、カルバメート化合物の熱分解反応によるイソシアネート化合物の生成系における固形物生成や液粘度上昇を抑えることができる。また、カルボニル化合物(I)は、イソシアネート化合物よりも沸点が高く、且つ、イソシアネート化合物の変性物よりも架橋点が少ないことから、イソシアネート化合物の精製時においても溶媒として働くことができる。或いは、カルボジイミド基等の官能基に結合し、イソシアネートの変性物同士が結合して高分子量化することを防ぐ。これらの作用により、イソシアネート化合物の精製時における運転性を良化させ、イソシアネート化合物を高収率で回収することを可能とする。
R11は(n11+n12)価の有機基、すなわち、2価以上8価以下の有機基である。中でも、R11としては、1以上4以下のエステル基又は窒素原子を有してもよい、炭素数1以上20以下の2価以上4価以下の脂肪族炭化水素基、又は、炭素数6以上20以下の2価以上3価以下の芳香族炭化水素基であることが好ましい。
R12は1価の有機基であり、酸素原子を含んでもよい、炭素数1以上20以下の脂肪族炭化水素基又は炭素数6以上20以下の芳香族炭化水素基が好ましい。
n11はカルバメート基の数を表し、1以上8以下の整数である。
n12はイソシアネート基の数を表し、0以上7以下の整数である。
n11とn12の和は2以上8以下の整数であり、2以上6以下の整数が好ましく、2以上5以下の整数がより好ましく、3以上4以下の整数がさらに好ましい。n11とn12の和が上記下限値以上であることで、カルボニル化合物の分子量が増加することに伴い、標準沸点も上昇し、イソシアネートとの分離がより容易となる。一方、上記上限値以下であることで、イソシアネート化合物が有する反応性の高いイソシアネート基との変性反応がより抑制されて、装置への固着やつまりをより低減することができる。
本実施形態のカルバメート化合物の製造方法は、触媒として、活性水素を有さないアミン化合物存在下で、第1級アミン化合物と、炭酸誘導体と、ヒドロキシ化合物と、を反応させて、カルバメート化合物を得る反応工程を含む。
反応工程では、触媒として、活性水素を有さないアミン化合物存在下で、第1級アミン化合物と、炭酸誘導体と、ヒドロキシ化合物と、を反応させて、カルバメート化合物を得る。
第1級アミン化合物と、炭酸誘導体と、ヒドロキシ化合物とから、触媒として、活性水素を有さないアミン化合物の存在下において、カルバメート化反応によりN-置換カルバメートを生成し、炭酸誘導体に由来するカルボニル基を有する化合物と、ヒドロキシ化合物とを含み、炭酸誘導体に由来する活性水素化合物及び活性水素を有さないアミン化合物からなる群より選ばれる1種以上の化合物を含んでいてもよい気相成分を回収する、カルバメート合成工程(工程(X1));
前記気相成分を凝縮器により凝縮する、気相成分の凝縮工程(工程(X2))
工程(X1)は、第1級アミン化合物と炭酸誘導体とヒドロキシ化合物とを原料として、触媒として、活性水素を有さないアミン化合物の存在下において、カルバメート化反応により、カルバメート化合物を生成し、ヒドロキシ化合物及び炭酸誘導体に由来するカルボニル基を有する化合物を含み、炭酸誘導体に由来する活性水素化合物及び活性水素を有さないアミン化合物からなる群より選ばれる1種以上の化合物を含んでいてもよい気相成分を回収する、カルバメート合成工程である。
第1級アミン化合物と炭酸誘導体とヒドロキシ化合物とを、触媒として少なくとも1種の、活性水素を有さないアミン化合物の存在下において、“同時に”反応させてN-置換カルバメートを製造する方法(方法(1A));
第1級アミン化合物と炭酸誘導体とを、触媒として、活性水素を有さないアミン化合物の存在下又は非存在下において反応させて、第1級アミン化合物に由来する基を有するカルボニル化合物を製造する工程(b1)、及び、前記カルボニル化合物とヒドロキシ化合物とを前記活性水素を有さないアミン化合物存在下において反応させて、N-置換カルバメートを製造する工程(b2)と、を含む、方法(方法(1B));
の2通りの方法をおこなうことができる。
方法(1A)における“同時”とは、方法(1B)ではN-置換カルバメートを製造する工程が2つの工程に分割されているのに対して、方法(1A)では工程が分割されていないという意であって、必ずしも、第1級アミン化合物と炭酸誘導体とヒドロキシ化合物とが全く同時に反応するという意味ではない。
ペンタン(各異性体)、ヘキサン(各異性体)、ヘプタン(各異性体)、オクタン(各異性体)、ノナン(各異性体)、デカン(各異性体)等のアルカン類;
ベンゼン、トルエン、キシレン(各異性体)、エチルベンゼン、ジイソプロピルベンゼン(各異性体)、ジブチルベンゼン(各異性体)、メシチレン、パラシメン、テトラリン、ナフタレン等の芳香族炭化水素及びアルキル置換芳香族炭化水素類;
アセトニトリル、ベンゾニトリル等のニトリル化合物;
クロロベンゼン、ジクロロベンゼン(各異性体)、ブロモベンゼン、ジブロモベンゼン(各異性体)、クロロナフタレン、ブロモナフタレン、ニトロベンゼン、ニトロナフタレン等のハロゲン又はニトロ基によって置換された芳香族化合物類;
ジフェニル、置換ジフェニル、ジフェニルメタン、ターフェニル、アントラセン、ジベンジルトルエン(各異性体)等の多環炭化水素化合物類;
シクロヘキサン、シクロペンタン、シクロオクタン、エチルシクロヘキサン等の脂環式炭化水素類;
メチルエチルケトン、アセトフェノン等のケトン類;
ジブチルフタレート、ジヘキシルフタレート、ジオクチルフタレート、ベンジルブチルフタレート等のエステル類;
テトラヒドロフラン、1,4-ジオキサン、1,2-ジメトキシエタン、ジフェニルエーテル、ジフェニルスルフィド等のエーテル類及びチオエーテル類;
アセトン、メチルエチルケトン等のケトン化合物;
酢酸エチル、安息香酸エチル等のエステル化合物;
ジメチルスルホキシド、ジフェニルスルホキシド等のスルホキシド類;
等を反応溶媒として好適に使用する。いうまでもなく、当該反応において過剰量使用するヒドロキシ化合物も、反応溶媒として好適に使用される。
以下、方法(1B)について述べる。
ヒドロキシ化合物の使用量(モル量)及び触媒として使用する活性水素を有さないアミン化合物の使用量(モル量)は、上記方法(1A)での使用量と同じ量とすることができる。
工程(b2)は、工程(b1)で得られた第1級アミン化合物に由来する基を有するカルボニル化合物と、ヒドロキシ化合物とを反応させて、N-置換カルバメートを製造する工程である。
ウレイレン基及びビウレット基からなる群より選ばれる1種以上の官能基を有する化合物と炭酸誘導体とを、触媒として少なくとも1種の、活性水素を有さないアミン化合物の存在下において反応させる工程(工程(c1))。
工程(X2)は、工程(X1)で回収された、炭酸誘導体及び炭酸誘導体に由来するカルボニル基を有する化合物からなる群より選ばれる1種以上と、ヒドロキシ化合物とを含み、炭酸誘導体に由来する活性水素化合物及び触媒である活性水素を有さないアミン化合物からなる群より選ばれる1種の化合物を含んでいてもよい気相成分を、カルバメート合成工程(工程(X1))を行う反応器に具備されている凝縮器による凝縮する凝縮工程である。
活性水素を有さないアミン化合物としては、第3級アミノ基、含窒素芳香族基、アミジン基、及びグアニジン基からなる群より選ばれる1種以上の官能基を有するものが好ましく用いられる。
本明細書中における「第3級アミノ基」とは、式(XIV)で表される基である。なお、式(XIV)中、波線は結合手を意味し、1価の有機基との結合部位を表す。
N,N-ジメチルアニリン、N,N-ジエチルアニリン、N-メチル-N-エチルアニリン、N,N-ジメチルアミノピリジン等の芳香族有機モノ第3級アミン;
N,N,N’,N’-テトラメチルフェニレンジアミン(各異性体)、メチレンビス(N,N-ジメチルアニリン)(各異性体)等の芳香族有機ポリ第3級アミン;
トリエチルアミン、エチルジイソプロピルアミン、N-メチルモルホリン、N-メチルピペリジン、キヌクリジン等の脂肪族モノ第3級アミン;
N,N’-ジメチルピペラジン、トリエチレンジアミン、N,N,N’ N’-テトラメチルエチレンジアミン、N,N,N’ N’-テトラメチルヘキサンジアミン、N,N,N’ N’-テトラメチルキシリレンジアミン、ペンタメチルジエチレントリアミン、ビス(2-モルホリノエチル)エーテル、ヘキサヒドロ-1,3,5-トリス(3-ジメチルアミノプロピル)-1,3,5-トリアジン、ヘキサメチレンテトラミン等の脂肪族ポリ第3級アミン;
等が挙げられる。
本明細書における「含窒素芳香族基」とは、ヘテロ芳香族基であって、少なくとも一つの窒素原子がヘテロ芳香環を構成する原子に含まれる基を指す。ヘテロ芳香族基を有する化合物は、非金属原子(炭素、酸素、窒素、硫黄、ケイ素)から構成され、その合計原子数は5以上85以下が好ましく、6以上30以下がより好ましい。非金属原子としては、炭素、酸素及び窒素から選ばれることが好ましく、さらに炭素原子であることがより好ましい。含窒素芳香族基を有する化合物を構成する合計炭素数は3以上85以下が好ましく、3以上30以下がより好ましい。
また、含窒素芳香族基を有する化合物に含まれる含窒素芳香族基の数は1以上5以下が好ましく、1以上3以下がより好ましい。
アミジン基を有する化合物としては、一般式(VIII-1)で表される化合物が好ましく使用される。
グアニジン基を有する化合物としては、下記一般式(VIII-2)で表される化合物が好ましく使用される。
本明細書における「第一級アミン化合物」とは、IUPACで定められたNomenclature規則C-8に定められる「第1アミン」(モノ第1アミン及びポリ第1アミン)を指す。このような第一級アミン化合物は、下記一般式(IIb)で表される化合物(以下、「第一級アミン化合物(IIb)」と称する場合がある)である。
1)R21bが、脂肪族基及び芳香族基からなる群より選ばれる1種以上の基で置換されてもよい、炭素数6以上85以下の芳香族基であり、且つ、n21bが1である、芳香族第1級モノアミン化合物;
2)R21bが、脂肪族基及び芳香族基からなる群より選ばれる1種以上の基で置換されてもよい芳香族環を1種以上含有する炭素数6以上85以下の基であり、n21bが2以上であり、且つ、R21b中の芳香族基がNH2基で置換された芳香族第1級ポリアミン化合物;
3)R21bが、炭素数1以上85以下の、芳香族基で置換されてもよい脂肪族基である、脂肪族第1級(モノ又はポリ)アミン化合物;
である。
好ましい芳香族第1級モノアミン化合物は、下記一般式(IIb-1)で表される化合物である。
環A211bは、炭素数6以上20以下の芳香族炭化水素環である。環A211bは、単環であってもよく、多環であってもよく、縮合環であってもよい。
n211bが2以上である場合に、複数存在するR211bは互いに同一であってもよく、異なっていてもよい。
n211bは、R211bの数を表し、1以上10以下の整数であり、0以上4以下の整数が好ましく、0以上3以下の整数がより好ましく、0以上2以下の整数がさらに好ましい。
芳香族第1級ポリアミン化合物は、上記一般式(IIb)で表される第1級アミン化合物におけるR21bが、脂肪族基及び芳香族基からなる群より選ばれる1種以上の基で置換されてもよい芳香族環を1種以上含有する炭素数6以上85以下の基であり、n21bが2以上であり、且つ、R21b中の芳香族基がNH2基で置換された芳香族第1級ポリアミン化合物である。
脂肪族第1級(モノ又はポリ)アミン化合物は、上記一般式(IIb)で表される第1級アミン化合物におけるR21bが、炭素数1以上85以下の、芳香族基で置換されてもよい脂肪族基である、脂肪族第1級(モノ又はポリ)アミン化合物である。
エチレンジアミン、ジアミノプロパン(各異性体)、ジアミノペンタン(各異性体)、ジアミノヘキサン(各異性体)、ジアミノデカン(各異性体)、キシリレンジアミン(各異性体)、ジアミノシクロブタン(各異性体)、ジアミノシクロヘキサン(各異性体)、ビス(ジアミノメチル)シクロヘキサン、3-アミノメチル-3,5,5-トリメチルシクロヘキシルアミン(シス及び/又はトランス体)、メチレンビス(シクロヘキシルアミン)(各異性体)等の脂肪族第1級ジアミン類;
トリアミノヘキサン(各異性体)、トリアミノノナン(各異性体)、トリアミノデカン(各異性体)等の脂肪族第1級トリアミン類;
リジンメチルエステル、リジンアミノエチルエステル等のアミノ酸誘導体;
等が挙げられる。
本明細書中における炭酸誘導体とは、第1級アミン化合物、ヒドロキシ化合物と共に、カルバメート化合物を製造するための原料として使用される成分である。炭酸誘導体として具体的には、尿素化合物、N-無置換カルバメート、炭酸エステル等が挙げられる。
本明細書中における尿素化合物とは、分子中に尿素結合を少なくとも1つ有する化合物である。好ましい尿素化合物としては、尿素結合を1つ有する化合物であり、具体的には、下記一般式(III-2)で表される化合物が挙げられる。
R321、R322、R323及びR324における炭素数1以上20以下の有機基としては、例えば、
メチル基、エチル基、プロピル基(各異性体)、ブチル基(各異性体)、ペンチル基(各異性体)、ヘキシル基(各異性体)、ヘプチル基(各異性体)、オクチル基(各異性体)等のアルキル基;
フェニル基、メチルフェニル基(各異性体)、ビフェニル基(各異性体)、ジメチルフェニル基(各異性体)、ジエチルフェニル基(各異性体)、ターフェニル基(各異性体)、トリメチルフェニル基(各異性体)、トリエチルフェニル基(各異性体)、トリプロピルフェニル基(各異性体)、トリブチルフェニル基(各異性体)等の、基を構成する炭素数が6以上20以下のアリール基;
フェニルメチル基、フェニルエチル基(各異性体)等の、該基を構成する炭素数が7以上20以下のアラルキル基;
等が挙げられる。
尿素には、ビウレット、トリウレット、及びシアヌル酸が含まれていてもよい。しかしながら、一般的にビウレット、トリウレット及びシアヌル酸は溶媒に対する溶解性が低い。原料や反応液の移送に際しては均一溶液であることが好ましいことから、これらの含有量は可能な限り少ない方が好ましい。また、尿素の融点は135℃であり常温での取り扱いでは固体であるが、その形状は、特に限定されず、例えば、粉体状でも顆粒状でも使用できる。
N-無置換カルバメートは、下記一般式(III-3)で表される化合物が好ましく使用される。
R331における炭素数1以上20以下の有機基としては、上記R321、R322、R323及びR324において例示されたものと同じものが挙げられる。
本明細書中における「炭酸エステル」とは、炭酸CO(OH)2の2つの水素原子のうち、その1原子又は2原子を、脂肪族基又は芳香族基で置換した化合物を指す。
R311b及びR312bにおける炭素数1以上20以下の有機基としては、上記R321、R322、R323及びR324において例示されたものと同じものが挙げられる。
ヒドロキシ化合物とは、ヒドロキシ基(-ОH基)を有する化合物を指す。ヒドロキシ化合物としては、ヒドロキシ基が炭素原子に結合した化合物である、アルコール及び芳香族ヒドロキシ化合物からなる群より選ばれる1種以上の化合物が好ましく使用される。
本明細書中における「アルコール」とは、IUPACの定義(Rule C-201)に記載された、「ヒドロキシ基が飽和炭素原子に結合した化合物(Compounds in which a hydroxy group, -OH, is attached to a saturated carbon atom:R3COH)」であり、下記一般式(VIa)で表されるヒドロキシ化合物である。
R61aにおける脂肪族基は、置換基として、炭素数1以上50以下の脂肪族基、又は炭素数7以上50以下の芳香族基が結合していてもよい。
メチル基、エチル基、プロピル基(各異性体)、ブチル基(各異性体)、ペンチル基(各異性体)、へキシル基(各異性体)、ヘプチル基(各異性体)、オクチル基(各異性体)、ノニル基(各異性体)、デシル基(各異性体)、ドデシル基(各異性体)、オクタデシル基(各異性体)、シクロペンチル基、シクロヘキシル基、メチルシクロペンチル基(各異性体)、メチルシクロヘキシル基(各異性体)、ジメチルシクロヘキシル基(各異性体)、ジエチルシクロヘキシル基(各異性体)等の、置換又は無置換のアルキル基及びシクロアルキル基;
フェニルメチル基、フェニルエチル基(各異性体)等のアラルキル基;
等が挙げられる。
メタノール、エタノール、プロパノール(各異性体)、ブタノール(各異性体)、ペンタノール(各異性体)、へキサノール(各異性体)、ヘプタノール(各異性体)、オクタノール(各異性体)、ノナノール(各異性体)、デカノール(各異性体)、ドデカノール(各異性体)、オクタデカノール(各異性体)、シクロペンタノール、シクロへキサノール、シクロヘプタノール、シクロオクタノール、メチルシクロペンタノール(各異性体)、エチルシクロペンタノール(各異性体)、メチルシクロへキサノール(各異性体)、エチルシクロへキサノール(各異性体)、プロピルシクロへキサノール(各異性体)、ブチルシクロへキサノール(各異性体)、ペンチルシクロへキサノール(各異性体)、へキシルシクロへキサノール(各異性体)、ジメチルシクロへキサノール(各異性体)、ジエチルシクロへキサノール(各異性体)、ジブチルシクロへキサノール(各異性体)等の、置換又は無置換のアルキルアルコール及びシクロアルキルアルコール;
フェニルメタノール、フェニルエタノール(各異性体)、フェニルプロパノール(各異性体)、フェニルブタノール(各異性体)、フェニルペンタノール(各異性体)、フェニルへキサノール(各異性体)、フェニルヘプタノール(各異性体)、フェニルオクタノール(各異性体)、フェニルノナノール(各異性体)等のアリール基で置換されたアルキルアルコール;
等が挙げられる。
本明細書中における「芳香族ヒドロキシ化合物」とは、IUPACの定義(Rule C-202)に記載されたフェノール類(phenols)「1つ又はそれ以上のヒドロキシ基がベンゼン環又は他のアレーン環に結合した化合物(Compounds having one or more hydroxy groups attached to a benzene or other arene ring.)」であり、下記一般式(VIb)で表される化合物である。
R61bにおける1価の有機基としては、ハロゲン原子、脂肪族基、芳香族基、及びこれらの基が結合した基である。より具体的には、非環式炭化水素基、環式炭化水素基(例えば、単環式炭化水素基、縮合多環式炭化水素基、架橋環式炭化水素基、スピロ炭化水素基、環集合炭化水素基、側鎖のある環式炭化水素基、ヘテロ環基、ヘテロ環式スピロ基、ヘテロ架橋環基、複素環基)、非環式炭化水素基と環式炭化水素基とが結合した基、及びこれら基が、特定の非金属原子(炭素、酸素、窒素、硫黄、ケイ素)との共有結合を介して結合している基である。
n61bは、環A61bに結合している芳香族性ヒドロキシ基の数を表し、一般に低粘度であるため、1又は2が好ましく、1がより好ましい。すなわち、芳香族ジ又はモノヒドロキシ化合物が好ましく、芳香族モノヒドロキシ化合物がより好ましい。
環A61bは、炭素数6以上20以下の芳香族炭化水素環である。環A61bは、単環であってもよく、多環であってもよく、縮合環であってもよい。
中でも、好ましいR611b、R612b、R613b、R614b及びR615bとしては、アルキル基、シクロアルキル基、アリール基、アラルキル基、エーテル基を含んでもよい、これらの基(置換又は無置換の、アルキルエーテル、アリールエーテル、又はアラルキルエーテル);これらの基のうち2種以上が結合した基;これらの基が特定の非金属原子(炭素、酸素、窒素、硫黄、ケイ素)との共有結合を介して結合された基;ハロゲン原子;又は水素原子である。
(i)水素原子;
(ii)ハロゲン原子;
(iii)α位の原子が炭素原子である、炭素数1以上44以下の基であって、α位の炭素原子に結合している3つの基が、それぞれ独立に、炭素数1以上43以下のアルキル基、炭素数4以上43以下のシクロアルキル基、炭素数1以上43以下のアルコキシ基、炭素数2以上43以下であって末端にOH基を有しないポリオキシアルキレンアルキルエーテル基、炭素数6以上43以下のアリール基、炭素数7以上43以下のアラルキル基、炭素数7以上43以下のアラルキルオキシ基、及びこれらの基のうち2種以上が結合した基;
(iv)炭素数1以上44以下のアリール基であって、該アリール基が置換基によって置換されており、該置換基は、以下に示す置換基で1以上5以下の整数の範囲で置換されてよいアリール基であり、該置換基は、水素原子、炭素数1以上38以下のアルキル基、炭素数4以上38以下のシクロアルキル基、炭素数1以上38以下のアルコキシ基、炭素数2以上38以下であって末端にOH基を有しないポリオキシアルキレンアルキルエーテル基、炭素数6以上38以下のアリール基、炭素数7以上38以下のアラルキル基、炭素数7以上38以下のアラルキルオキシ基、及びこれらの基のうち2種以上が結合した基;
(v)α位の原子が酸素原子である、炭素数1以上44以下の基であって、α位の酸素原子に結合している基が、炭素数1以上44以下のアルキル基、炭素数4以上44以下のシクロアルキル基、炭素数1以上44以下のアルコキシ基、炭素数2以上44以下であって末端にOH基を有しないポリオキシアルキレンアルキルエーテル基、炭素数6以上44以下のアリール基、炭素数7以上44以下のアラルキル基、炭素数7以上44以下のアラルキルオキシ基、及びこれらの基のうち2種以上が結合した基。
メチル基、エチル基、プロピル基(各異性体)、ブチル基(各異性体)、シクロペンチル基、シクロヘキシル基、メチルシクロペンチル基(各異性体)、エチルシクロペンチル基(各異性体)、メチルシクロヘキシル基(各異性体)、ジメチルシクロヘキシル基(各異性体)、ジエチルシクロヘキシル基(各異性体)等の、置換又は無置換のアルキル基及びシクロアルキル基;
メトキシ基、エトキシ基、プロポキシ基(各異性体)、ブチルオキシ基(各異性体)、シクロペンチルオキシ基、シクロヘキシルオキシ基、メチルシクロペンチルオキシ基(各異性体)、エチルシクロペンチルオキシ基(各異性体)、メチルシクロヘキシルオキシ基(各異性体)、ジメチルシクロヘキシルオキシ基(各異性体)、ジエチルシクロヘキシルオキシ基(各異性体)等の、置換又は無置換のアルコキシ基及びシクロアルコキシ基;
フェニル基、メチルフェニル基(各異性体)、エチルフェニル基(各異性体)、ビフェニル基(各異性体)、ジメチルフェニル基(各異性体)、ジエチルフェニル基(各異性体)、ターフェニル基(各異性体)、トリメチルフェニル基(各異性体)、トリエチルフェニル基(各異性体)等の、置換又は無置換のアリール基;
フェノキシ基、メチルフェノキシ基(各異性体)、エチルフェノキシ基(各異性体)、フェニルフェノキシ基(各異性体)、ジメチルフェノキシ基(各異性体)、ジエチルフェノキシ基(各異性体)、ジフェニルフェノキシ基(各異性体)、トリメチルフェノキシ基(各異性体)、トリエチルフェノキシ基(各異性体)等の、置換又は無置換のアリールオキシ基;
フェニルメチル基、フェニルエチル基(各異性体)、フェニルプロピル基(各異性体)等のアラルキル基、フェニルメトキシ基、フェニルエトキシ基(各異性体)等のアラルキルオキシ基;
等が挙げられる。
フェノール、クロロフェノール(各異性体)、ブロモフェノール(各異性体)、ジクロロフェノール(各異性体)、ジブロモフェノール(各異性体)、トリクロロフェノール(各異性体)、トリブロモフェノール(各異性体)、フェノール、メチルフェノール(各異性体)、エチルフェノール(各異性体)、プロピルフェノール(各異性体)、ブチルフェノール(各異性体)ペンチルフェノール(各異性体)、ヘキシルフェノール(各異性体)、ヘプチルフェノール(各異性体)、オクチルフェノール(各異性体)、ノニルフェノール(各異性体)、クミルフェノール(各異性体)、ジメチルフェノール(各異性体)、ジエチルフェノール(各異性体)、イソプロピルメチルフェノール(各異性体)、トリメチルフェノール(各異性体)、トリエチルフェノール(各異性体)、ジブチルメチルフェノール(各異性体)、(メトキシメチル)フェノール(各異性体)、(エトキシメチル)フェノール(各異性体)、((シクロペンチルオキシメチル)フェノール(各異性体)、(シクロヘキシルオキシメチル)フェノール(各異性体)、(メチルシクロペンチルオキシメチル)フェノール(各異性体)、(メチルシクロヘキシルオキシメチル)フェノール(各異性体)、(ジメチルシクロヘキシルオキシメチル)フェノール(各異性体)、(フェノキシメチル)フェノール、(メチルフェノキシメチル)フェノール(各異性体)、(フェニルフェノキシメチル)フェノール(各異性体)、(ジメチルフェノキシメチル)フェノール(各異性体)、(ジエチルフェノキシメチル)フェノール(各異性体)、(ジフェニルフェノキシメチル)フェノール(各異性体)、(トリメチルフェノキシメチル)フェノール(各異性体)、(フェニルメトキシメチル)フェノール、(フェニルエトキシメチル)フェノール(各異性体)、(フェニルプロピルオキシメチル)フェノール(各異性体)、ジ(メトキシメチル)フェノール、ジ(エトキシメチル)フェノール、ジ(プロポキシメチル)フェノール(各異性体)、ジ(ブチルオキシメチル)フェノール(各異性体)、ジ(シクロペンチルオキシメチル)フェノール、ジ(シクロヘキシルオキシメチル)フェノール、ジ(メチルシクロペンチルオキシメチル)フェノール(各異性体)ジ(メチルシクロヘキシルオキシメチル)フェノール(各異性体)ビス(ジメチルシクロヘキシルオキシメチル)フェノール(各異性体)、ビス(ジエチルシクロヘキシルオキシメチル)フェノール(各異性体)、ジ(メチルフェノキシメチル)フェノール(各異性体)、ジ(エチルフェノキシメチル)フェノール(各異性体)、ジ(プロピルフェノキシメチル)フェノール(各異性体)、ビス(ジメチルフェノキシメチル)フェノール(各異性体)、ビス(ジエチルフェノキシメチル)フェノール(各異性体)、ビス(ジプロピルフェノキシメチル)フェノール(各異性体)、ビス(ジブチルフェノキシメチル)フェノール(各異性体)、ビス(ジフェニルフェノキシメチル)フェノール(各異性体)、ジ(トリメチルフェノキシメチル)フェノール(各異性体)、ジ(トリエチルフェノキシメチル)フェノール(各異性体)ジ(フェニルメトキシメチル)フェノール、ジ(フェニルエトキシメチル)フェノール(各異性体)、ジ(フェニルプロピルオキシメチル)フェノール(各異性体)、トリ(メトキシメチル)フェノール、トリ(エトキシメチル)フェノールトリ(シクロヘキシルオキシメチル)フェノール、トリ(メチルシクロペンチルオキシメチル)フェノール(各異性体)、トリ(エチルシクロペンチルオキシメチル)フェノール(各異性体)、トリ(メチルシクロヘキシルオキシメチル)フェノール(各異性体)、ビス(ジメチルシクロヘキシルオキシメチル)フェノール(各異性体)、ビス(ジエチルシクロヘキシルオキシメチル)フェノール(各異性体)、ビス(ジブチルシクロヘキシルオキシメチル)フェノール(各異性体)、トリ(フェノキシメチル)フェノール、トリ(メチルフェノキシメチル)フェノール(各異性体)、トリ(エチルフェノキシメチル)フェノール(各異性体)、トリ(フェニルフェノキシメチル)フェノール(各異性体)、ビス(ジメチルフェノキシメチル)フェノール(各異性体)、ビス(ジエチルフェノキシメチル)フェノール(各異性体)、ビス(ジフェニルフェノキシメチル)フェノール(各異性体)、トリ(トリメチルフェノキシメチル)フェノール(各異性体)、トリ(トリエチルフェノキシメチル)フェノール(各異性体)、トリ(フェニルメトキシメチル)フェノール、(フェニルメチル)フェノール(各異性体)、((メチルフェニル)メチル)フェノール(各異性体)、((エチルフェニル)メチル)フェノール(各異性体)、((ビフェニル)メチル)フェノール(各異性体)、((ジメチルフェニル)メチル)フェノール(各異性体)、((ターフェニル)メチル)フェノール(各異性体)、((トリメチルフェニル)メチル)フェノール(各異性体)、((トリエチルフェニル)メチル)フェノール(各異性体)、ジ(フェニルメチル)フェノール(各異性体)、ジ((メチルフェニル)メチル)フェノール(各異性体)、ジ((エチルフェニル)メチル)フェノール(各異性体)、ジ((ビフェニル)メチル)フェノール(各異性体)、ジ((ジメチルフェニル)メチル)フェノール(各異性体)、ジ((ジエチルフェニル)メチル)フェノール(各異性体)、ジ((ターフェニル)メチル)フェノール(各異性体)、ジ((トリメチルフェニル)メチル)フェノール(各異性体)、ジ((トリエチルフェニル)メチル)フェノール(各異性体)、トリ(フェニルメチル)フェノール(各異性体)、トリ((メチルフェニル)メチル)フェノール(各異性体)、トリ((エチルフェニル)メチル)フェノール(各異性体)、トリ((ビフェニル)メチル)フェノール(各異性体)、トリ((ジメチルフェニル)メチル)フェノール(各異性体)、トリ((ジエチルフェニル)メチル)フェノール(各異性体)、トリ((ターフェニル)メチル)フェノール(各異性体)、トリ((トリメチルフェニル)メチル)フェノール(各異性体)、トリ((トリエチルフェニル)メチル)フェノール(各異性体)、フェニルエチルフェノール(各異性体)、フェニル-n-プロピルフェノール(各異性体)、フェニル-n-ブチルフェノール(各異性体)、フェニル-n-ペンチルフェノール(各異性体)、フェニル-n-ヘキシルフェノール(各異性体)、フェニル-n-ヘプチルフェノール(各異性体)、フェニル-n-オクチルフェノール(各異性体)、フェニル-n-ノニルフェノール(各異性体)、(メチルアミノ)フェノール、(エチルアミノ)フェノール、(プロピルアミノ)フェノール(各異性体)、(ブチルアミノ)フェノール(各異性体)、(ペンチルアミノ)フェノール(各異性体)、(ヘキシルアミノ)フェノール(各異性体)、ジ(メチルアミノ)フェノール、ジ(エチルアミノ)フェノール、ジ(プロピルアミノ)フェノール(各異性体)、ジ(ブチルアミノ)フェノール(各異性体)、トリ(メチルアミノ)フェノール、トリ(エチルアミノ)フェノール、メトキシフェノール(各異性体)、エトキシフェノール(各異性体)、プロピルオキシフェノール(各異性体)、ブチルオキシフェノール(各異性体)、ペンチルオキシフェノール(各異性体)、ヘキシルオキシフェノール(各異性体)、ヘプチルオキシフェノール(各異性体)、オクチルオキシフェノール(各異性体)、ノニルオキシフェノール(各異性体)、デシルオキシフェノール(各異性体)、ドデシルオキシフェノール(各異性体)、オクタデシルオキシフェノール(各異性体)、シクロペンチルオキシフェノール(各異性体)、シクロヘキシルオキシフェノール(各異性体)、(メチルシクロペンチルオキシ)フェノール(各異性体)、(エチルシクロペンチルオキシ)フェノール(各異性体)、(メチルシクロヘキシルオキシ)フェノール(各異性体)、(エチルシクロヘキシルオキシ)フェノール(各異性体)、(プロピルシクロヘキシルオキシ)フェノール(各異性体)、(ジメチルシクロヘキシルオキシ)フェノール(各異性体)、(ジエチルシクロヘキシルオキシ)フェノール(各異性体)、フェノキシフェノール、(メチルフェニルオキシ)フェノール(各異性体)、(ジメチルフェニルオキシ)フェノール(各異性体)、(トリメチルフェニルオキシ)フェノール(各異性体)、(フェニルメチルオキシ)フェノール、(フェニルエチルオキシ)フェノール(各異性体)、ジメトキシフェノール(各異性体)、ジエトキシフェノール(各異性体)、ジシクロヘキシルオキシフェノール(各異性体)、ジ(メチルシクロペンチルオキシ)フェノール(各異性体)、ビス(ジメチルシクロヘキシルオキシ)フェノール(各異性体)、ジ(メチルフェニルオキシ)フェノール(各異性体)、ビス(ジメチルフェニルオキシ)フェノール(各異性体)、ジ(トリメチルフェニルオキシ)フェノール(各異性体)、ジ(フェニルメチルオキシ)フェノール、トリメトキシフェノール(各異性体)、トリエトキシフェノール(各異性体)、トリプロピルオキシフェノール(各異性体)、トリブチルオキシフェノール(各異性体)、トリシクロペンチルオキシフェノール(各異性体)、トリシクロヘキシルオキシフェノール(各異性体)、トリフェニルオキシフェノール、トリ(メチルフェニルオキシ)フェノール(各異性体)、トリ(エチルフェニルオキシ)フェノール(各異性体)、トリ(プロピルフェニルオキシ)フェノール(各異性体)、トリ(ジメチルフェニルオキシ)フェノール(各異性体)、トリ(ジエチルフェニルオキシ)フェノール(各異性体)、トリ(トリメチルフェニルオキシ)フェノール(各異性体)、トリ(トリエチルフェニルオキシ)フェノール(各異性体)、トリ(フェニルメチルオキシ)フェノール、トリ(フェニルエチルオキシ)フェノール(各異性体)、フェニルフェノール(各異性体)、ヒドロキシフェニルフェノール(各異性体)、ヒドロキシフェノキシフェノール(各異性体)、ヒドロキシフェニル-プロピルフェノール(各異性体)、ナフトール(各異性体)。
また、R611b、R612b、R613b、R614b及びR615bの合計炭素数が0以上9以下であり、R611b、R612b、R613b、R614b及びR615bがそれぞれ独立に、水素原子、直鎖状若しくは分岐鎖状のアルキル基、シクロアルキル基、置換若しくは無置換のアリール基、直鎖状若しくは分岐鎖状のアルコキシ基、置換若しくは無置換のアリールオキシ基、又は、置換若しくは無置換のアラルキル基である芳香族ヒドロキシ化合物がより好ましい。
本実施形態の製造方法で得られるカルバメート化合物としては、N-置換カルバメートが挙げられる。
第1級アミン化合物と尿素とヒドロキシ化合物との反応において、ヒドロキシ化合物としてアルコールを使用した場合に得られるN-置換カルバメートは、下記一般式(Va)で表される化合物(以下、「N-置換カルバメート化合物(Va)」と称する場合がある)である。
1)R51aが1種以上の芳香族環を含有する炭素数6以上85以下の基であり、且つ、n51aが1であるN-芳香族モノカルバメート化合物;
2)R51aが、1種以上の芳香族環を含有する炭素数6以上85以下の基であり、且つ、n51aが2以上であるN-芳香族ポリカルバメート化合物;
3)R51aが、芳香族基で置換されてもよい、炭素数1以上85以下の脂肪族基であり、且つ、n51aが2以上のN-脂肪族ポリカルバメート化合物;
である。
好ましいN-芳香族モノカルバメート化合物としては、下記一般式(Va-1)で表されるN-芳香族モノカルバメート化合物である。
N-芳香族ポリカルバメート化合物は、上記一般式(Va)におけるR51aが、1種以上の芳香族環を含有する炭素数6以上85以下の基であり、且つ、n51aが2以上であるN-芳香族ポリカルバメート化合物である。中でも、流動性等を考慮して、上記一般式(Va)におけるR51aが、脂肪族基又は芳香族基で置換されてもよい、芳香族環を1種以上含有する炭素数6以上13以下の基であり、且つ、n51aが2以上であるN-芳香族ポリカルバメート化合物が好ましい。
N-脂肪族ポリカルバメート化合物は、上記一般式(Va)におけるR51aが、芳香族基で置換されてもよい、炭素数1以上85以下の脂肪族基であり、且つ、n51aが2以上のN-脂肪族ポリカルバメート化合物である。
第1級アミン化合物と尿素とヒドロキシ化合物との反応において、ヒドロキシ化合物として芳香族ヒドロキシ化合物を使用した場合に得られるN-置換カルバメートは、下記一般式(Vb)で表される化合物(以下、「N-置換カルバメート化合物(Vb)」と称する場合がある)である。
1)R51bが、脂肪族基又は芳香族基で置換されてもよい、炭素数6以上85以下の芳香族基であり、且つ、n51bが1であるN-芳香族モノカルバメート化合物;
2)R51bが、脂肪族基又は芳香族基で置換されてもよい、芳香族環を1種以上含有する、炭素数6以上85以下の基であり、且つ、n51bが2以上である、N-芳香族ポリカルバメート化合物;
3)R51bが、芳香族基で置換されてもよい、炭素数1以上85以下の脂肪族基であり、且つ、n51bが2以上のN-脂肪族ポリカルバメート化合物;
である。
好ましいN-芳香族モノカルバメート化合物は、下記一般式(Vb)で表されるN-芳香族モノカルバメート化合物である。
N-芳香族ポリカルバメート化合物は、上記一般式(Vb)におけるR51bが、脂肪族基又は芳香族基で置換されてもよい、芳香族環を1種以上含有する、炭素数6以上85以下の基であり、且つ、n51bが2以上である、N-芳香族ポリカルバメート化合物である。
N-脂肪族ポリカルバメート化合物は、一般式(Vb)におけるR51bが、脂肪族基又は芳香族基で置換されてもよい、芳香族環を1種以上含有する、炭素数6以上85以下の基であり、且つ、n51bが2以上である、N-芳香族ポリカルバメート化合物である。
N-ジメチルフェニルカルバミン酸(ヘキシルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ヘプチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(オクチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ノニルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(デシルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ドデシルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(オクタデシルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジメチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジエチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジプロピルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジブチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジペンチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジヘキシルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジヘプチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジオクチルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジノニルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジデシルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジドデシルフェニル)エステル(各異性体)、N-ジメチルフェニルカルバミン酸(ジオクタデシルフェニル)エステル(各異性体)等が挙げられる。
本実施形態のイソシアネート化合物の製造方法は、上述した製造方法により得られたカルバメート化合物を熱分解する熱分解工程を含む。
熱分解工程では、上述したカルバメート化合物の製造方法により得られたカルバメート化合物を熱分解する。
本実施形態のイソシアネート化合物の製造方法で得られるイソシアネート化合物としては、下記一般式(VIIc)で表される化合物(以下、「イソシアネート化合物(VIIc)」と称する場合がある)が好ましく用いられる。
R71cは、n71c価の有機基である。すなわち、1価以上12価以下の有機基である。中でも、R71cとしては、炭素原子、酸素原子及び水素原子からなる有機基であることが好ましく、活性水素を有しない有機基であることがより好ましい。
本実施形態の回収方法は、下記一般式(VIIa)で表されるイソシアネート化合物(以下、「イソシアネート化合物(VIIa)」と称する場合がある)の製造方法で副生する前記イソシアネート化合物よりも沸点の高い化合物(以下、「高沸点化合物」と称する場合がある)を含む液相成分から、下記一般式(IIa)で表されるアミン化合物(以下、「アミン化合物(IIa)」と称する場合がある)を回収する方法である。
前記液相成分と、芳香族ヒドロキシ化合物と、活性水素含有化合物と、触媒と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a)。
工程(a)は、液相成分と、芳香族ヒドロキシ化合物と、活性水素含有化合物と、触媒と、を反応器内で反応させて、アミン化合物(IIa)を含む反応液を得る工程である。
前記液相成分と芳香族ヒドロキシ化合物を混合させる工程(a1);
前記工程(a1)で得られた混合物と、活性水素含有化合物と、触媒と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a2)。
工程(a1)は、イソシアネート化合物(VIIa)の製造方法で副生する高沸点化合物を含有する液相成分と芳香族ヒドロキシ化合物を混合させる工程である。
工程(a2)は、前記工程(a1)で混合された組成物と活性水素含有化合物と触媒とを反応器内で反応させる工程である。
活性水素含有化合物としては、水、尿素、アルコール(なお、芳香族ヒドロキシ化合物は含まない)、アルキルチオール及びアミン化合物(好ましくは、第一級アミン化合物)からなる群より選ばれる少なくとも1種であることが好ましく、水、第一級アミン化合物、及びヒドロキシ化合物からなる群より選ばれる少なくとも1種であることがより好ましい。これらの化合物は単独で使用してもよく、2種以上組み合わせて使用してもよい。中でも、水、水及び第一級アミン化合物の組み合わせ、水及びアルコールの組み合わせ、又は、尿素及びアルコールの組み合わせがさらに好ましい。また、水、又は、水及び第一級アミン化合物の組み合わせが特に好ましい。さらに、水、又は、水及び脂肪族の第一級アミン化合物の組み合わせが最も好ましい。
触媒としては、特に制限されないが、例えば、水酸化ナトリウム、水酸化カリウム、水酸化マグネシウム、酸化マグネシウム、酸化カルシウム、酸化バリウム等のアルカリ金属及びアルカリ土類金属の水酸化物と酸化物;ピリジン、メチルピリジン(異性体含む)、ジプロピルエチルアミン、N-メチルモルホリン、N-エチルモルホリン、トリエチルアミン、トリエチレンジアミン等の複素環化合物や第三級アミン類;酸化アルミニウム、酸化亜鉛、酸化チタン、酸化ジルコニウム、酸化セリウム、酸化鉄、酸化スズ、酸化バナジウム、酸化モリブデン、酸化マンガン、酸化コバルト、酸化ニッケル等の第3~14族の金属酸化物が挙げられる。中でも、アルカリ金属の水酸化物及び酸化物、アルカリ土類金属の水酸化物及び酸化物、第三級アミン化合物、第12族の金属酸化物、第13族の金属酸化物、並びに、第14族の金属酸化物からなる群より選ばれる1種以上の化合物が好ましい。また、アルカリ金属の水酸化物及び酸化物、並びに、アルカリ土類金属の水酸化物及び酸化物からなる群より選ばれる1種以上の化合物がより好ましく、アルカリ金属の水酸化物及び酸化物がさらに好ましく、水酸化ナトリウム、水酸化カリウム等のアルカリ金属の水酸化物が特に好ましい。これらの触媒は単独又は二種以上を混合して使用することもできる。
前記工程(a1)で得られた混合物と、活性水素含有化合物としてアミン化合物と、触媒と、を混合させる工程(a2-1);
前記工程(a2-1)で得られた混合物と、活性水素含有化合物として水と、を反応器内で反応させて、アミン化合物(II)を含む反応液を得る工程(a2-2)。
工程(a2-1)は、前記工程(a1)で得られた混合物と活性水素含有化合物としてアミン化合物と触媒とを混合させる工程である。
工程(a2-2)は、工程(a2-1)で得られた混合物と、活性水素含有化合物として水と、を反応器内で反応させて、アミン化合物(II)を含む反応液を得る。
前記アミン化合物(IIa)を含む反応液から前記アミン化合物(IIa)を分離する工程(b);
前記一般式(IIa)で表されるアミン化合物を精製する工程(c);
前記アミン化合物(IIa)を含む反応液から前記ヒドロキシ化合物を分離する工程(e);及び
前記ヒドロキシ化合物を精製する工程(f)。
工程(b)は、前記工程(a2)、工程(a2-2)、又は後述する工程(e)若しくは工程(f)で得られた液から、アミン化合物(IIa)を分離する工程である。前記工程(a2)又は工程(a2-2)で得られた反応液から、アミン化合物(IIa)を分離する方法は公知の方法を用いることができ、例えば、蒸留分離、液-液相分離、固液分離、膜分離等が挙げられる。
工程(c)は、前記工程(b)で分離されたアミン化合物(IIa)を精製する工程である。アミン化合物(IIa)を精製する方法は、公知の方法を用いることができ、例えば、蒸留分離、液-液相分離、固液分離、膜分離等が挙げられる。
工程(e)は、前記工程(a2)、工程(a2-2)、又は工程(b)若しくは工程(c)で得られた液から、イソシアネート化合物(VIIa)の製造で用いたヒドロキシ化合物を分離する工程である。
工程(f)は、前記工程(e)で分離されたヒドロキシ化合物を精製する工程である。
前記工程(c)で精製されたアミン化合物(II)を、イソシアネート化合物(VIIa)の製造方法において、原料として再利用する工程(d);
前記工程(f)で精製されたヒドロキシ化合物を、イソシアネート化合物(VIIa)の製造方法において、原料として再利用する工程(g)。
工程(d)は、前記工程(c)で精製されたアミン化合物(IIa)を、イソシアネート化合物(VIIa)において、原料として再利用する工程である。
工程(g)は、前記工程(f)で精製されたヒドロキシ化合物を、イソシアネート化合物(VIIa)の製造方法において、原料として再利用する工程である。
前記工程(b)においてアミン化合物(IIa)を分離した後の残留液、前記工程(c)においてアミン化合物(IIa)を精製した後の残留液、前記工程(e)において前記ヒドロキシ化合物を分離した後の残留液、及び前記工程(f)において前記ヒドロキシ化合物を精製した後の残留液からなる群より選ばれる1種以上の残留液を、前記工程(a)において再利用する工程(h)。
工程(h)は、前記工程(b)においてアミン化合物(IIa)を分離した後の残留液、前記工程(c)において前記一般式(IIa)で表されるアミン化合物を精製した後の残留液、前記工程(e)において前記ヒドロキシ化合物を分離した後の残留液、及び前記工程(f)において前記ヒドロキシ化合物を精製した後の残留液からなる群より選ばれる1種以上の残留液を、前記工程(a)において再利用する工程である。
液相成分に含まれる高沸点化合物は、アミン化合物(IIa)を原料としてイソシアネート化合物(VIIa)の製造時において副生するものであって、アミン化合物(IIa)、反応中間生成物である後述するカルバメート化合物、及びイソシアネート化合物(VIIa)からなる群より選ばれる1種以上の化合物が反応して生成すると推定される。
イソシアネート化合物(VIIa)は、下記一般式(VIIa)で表される化合物である。
R71aはn71a価の有機基である。すなわち、2価以上8価以下の有機基である。中でも、R71aとしては、1以上4以下のエステル基又は窒素原子を有してもよい、炭素数1以上20以下の2価以上4価以下の脂肪族炭化水素基、又は、炭素数6以上20以下の2価以上3価以下の芳香族炭化水素基であることが好ましい。
n71aは、イソシアネート基の数を表し、2以上8以下の整数であり、2以上4以下の整数が好ましく、2以上3以下の整数がより好ましい。
イソシアネート化合物(VIIa)は、アミン化合物(IIa)を原料として製造することができ、その製造方法は、公知の方法を用いることができる。例えば、下記に示す方法(X)、方法(Y)が挙げられる。
方法(X):アミン化合物(IIa)と炭酸誘導体とヒドロキシ化合物を用いてカルバメート化合物を製造し(工程(x1))、該カルバメート化合物を熱分解反応に付してイソシアネート化合物(VIIa)を製造する(工程(x2))方法。
方法(Y):アミン化合物(IIa)とホスゲンを反応させ、脱塩化水素しながらイソシアネート化合物(VIIa)を製造する方法。
方法(X)は、工程(x1)、工程(x2)以外に、必要に応じてこれ以外のその他の工程を付加することができる。その他の工程としては、例えば、工程(x1)で得られるカルバメート化合物と、工程(x1)で用いるヒドロキシ化合物と別種のヒドロキシ化合物を用いて、カルバメートのエステル交換反応を行い、別種のカルバメートを製造する工程(工程(x1-a));工程(x1)の反応液からヒドロキシ化合物の一部又は全部を分離して予備濃縮する工程(工程(x1-b));イソシアネート化合物(VIIa)を精製する工程(工程(x3));工程(x1)で炭酸誘導体から副生するアンモニアやヒドロキシ化合物等を回収する工程等が挙げられる。
イソシアネート化合物(VIIa)よりも沸点の低い化合物(以下、「低沸点化合物」と称する場合がある)を含む気相成分を蒸留分離して、イソシアネート化合物(VIIa)を含む液相成分を分離する(以下、「軽沸分離」と称する場合がある)工程(工程(x3-1));及び、
イソシアネート化合物(VIIa)を含む気相成分を蒸留分離して、イソシアネート化合物(VIIa)から高沸点化合物を分離する(以下、「高沸分離」と称する場合がある)工程(工程(x3-2));
に分けて行うこともでき、或いは、工程(x3-1)及び工程(x3-2)の実施する順番を入れ替えて行うこともできる。
工程(x1)は、アミン化合物(IIa)と炭酸誘導体とヒドロキシ化合物とを反応させてカルバメート化合物を生成し、尿素及びイソシアン酸からなる群より選ばれる1種以上の化合物、ヒドロキシ化合物、並びに、アンモニアを含む気相成分を、生成されたカルバメート化合物とは別個に回収する工程である。
方法(x1-1):アミン化合物(IIa)と尿素とヒドロキシ化合物とを“同時に”反応させてカルバメート化合物を製造する方法。
方法(x1-2):アミン化合物(IIa)と尿素とを反応させてウレイド基を有する化合物を製造する工程(x1-2-1);及び
工程(x1-2-1)で得られたウレイド基を有する化合物とヒドロキシ化合物とを反応させてカルバメート化合物を製造する工程(x1-2-2);
を含む方法。
方法(x1-1)において、ヒドロキシ化合物の使用量(モル量)は、使用されるアミン化合物(IIa)のアミノ基のモル量に対して、化学量論比で、通常、1倍以上500倍以下であり、5倍以上200倍以下が好ましく、10倍以上150倍以下がより好ましく、10倍以上100倍以下がさらに好ましい。
次に、方法(x1-2)における前記工程(x1-2-1)~工程(x1-2-2)について説明する。
前記工程(x1)によって製造されるカルバメート化合物は、下記一般式(Vc)で表される化合物(以下、「カルバメート化合物(Vc)」と称する場合がある)である。
方法(x2-1):カルバメート化合物(Vc)を熱分解反応に付して、イソシアネート化合物(VIIa)を含む気相成分と、高沸点化合物と、を分離することで、イソシアネート化合物(VIIa)を含む成分を製造する方法。
方法(x2-2):カルバメート化合物(Vc)を熱分解反応に付して、イソシアネート化合物(VIIa)を含む成分と、高沸点化合物と、を共に液相成分として回収し、回収した液からイソシアネート化合物(VIIa)を含む気相成分と、高沸点化合物と、を分離することで、イソシアネート化合物(VIIa)を製造する方法。
方法(Y)は、アミン化合物(IIa)とホスゲンを反応させ、脱塩化水素しながら前記イソシアネート化合物(VIIa)を製造する工程(y1)を含む方法である。アミン化合物(IIa)とホスゲンとの反応は、溶液中で又は気相で行われ、カルバミン酸クロリドを生成し、該カルバミン酸クロリドを熱分解して、副生される塩化水素を分離することによりイソシアネート化合物(VIIa)を製造する。カルバミン酸クロリドの生成と、カルバミン酸クロリドの熱分解は、別の工程で行ってもよく、同じ工程で同時に行ってもよい。
カルバメート化合物(Vc)は、下記一般式(Vc)で表される化合物である。
R51cは、n51c価の有機基であり、関係式:R51c=R71aを満たす。すなわち、R51cは、上記R71aと同じである。
R52cは1価の有機基であり、ヒドロキシ化合物から水酸基を除いた残基である。
n51cは、カルバメート基の数を表し、関係式:n51c=n71aを満たす。すなわち、n51cは、上記n71aと同じである。n51cは、2以上8以下の整数であり、2以上4以下の整数が好ましく、2以上3以下の整数がより好ましい。
炭酸誘導体としては、尿素、N-無置換カルバミン酸エステル、炭酸エステル等が挙げられる。
本実施形態の回収方法において回収されるヒドロキシ化合物は、下記一般式(VIc)で表される化合物(以下、「ヒドロキシ化合物(VIc)」と称する場合がある)が好ましく使用される。ヒドロキシ化合物は、上述したように、イソシアネート化合物(VIIa)の製造原料ともいえる。また、本実施形態の回収方法において、工程(a1)において用いられる芳香族ヒドロキシ化合物としては、後述するヒドロキシ化合物のうち、芳香族基を有するヒドロキシ化合物が好ましく使用される。
R61cは、1価の有機基であり、酸素原子を含んでもよい、炭素数1以上20以下の脂肪族炭化水素基又は炭素数6以上20以下の芳香族炭化水素基が好ましい。
R611cにおける炭素数1以上20以下のアルキル基としては、例えば、メチル基、エチル基、プロピル基(各異性体)、ブチル基(各異性体)、ペンチル基(各異性体)、ヘキシル基(各異性体)、ヘプチル基(各異性体)、オクチル基(各異性体)、ノニル基(各異性体)、デシル基(各異性体)、ドデシル基(各異性体)、オクタデシル基(各異性体)等が挙げられる。
環A611cは、炭素数6以上20以下の芳香族炭化水素環である。環A611cは、単環であってもよく、多環であってもよく、縮合環であってもよい。
n611cは、置換基R611cの数を示し、1以上10以下の整数である。
本実施形態の回収方法において回収されるアミン化合物(IIa)は、下記一般式(IIa)で表される化合物である。アミン化合物(IIa)は、上述したように、イソシアネート化合物(VIIa)の製造原料ともいえる。
R21aは、n21a価の有機基であり、関係式:R21a=R71aを満たす。すなわち、R21aは、上記R71aと同じである。
n21aは、アミノ基の数を表し、2以上8以下の整数であり、関係式:n21a=n71aを満たす。すなわち、n21aは、上記n71aと同じである。n21aは、2以上8以下の整数であり、2以上6以下の整数が好ましく、2以上5以下の整数がより好ましく、3以上4以下の整数がさらに好ましい。
本実施形態のイソシアネート組成物は、イソシアネート化合物と、
下記一般式(I)で表されるカルボニル化合物(以下、「カルボニル化合物(I)」と称する場合がある)と、
を含む。
カルボニル化合物(I)は、下記一般式(I)で表される化合物である。
R11は(n11+n12)価の有機基、すなわち、2価以上8価以下の有機基である。中でも、R11としては、1以上4以下のエステル基又は窒素原子を有してもよい、炭素数1以上20以下の2価以上4価以下の脂肪族炭化水素基、又は、炭素数6以上20以下の2価以上3価以下の芳香族炭化水素基であることが好ましい。
R12は1価の有機基であり、酸素原子を含んでもよい、炭素数1以上20以下の脂肪族炭化水素基又は炭素数6以上20以下の芳香族炭化水素基が好ましい。
n11は1以上8以下の整数である。
イソシアネート化合物としては、下記一般式(VIIb)で表される化合物(以下、「イソシアネート化合物(VIIb)」と称する場合がある)が好ましく使用される。
R71bは、n71b価の有機基であり、関係式:R71b=R11を満たす。すなわち、R71bは、上記R11と同じである。
n71bは、イソシアネート基の数を表し、関係式:n71b=n11+n12を満たす。n71bは、2以上8以下の整数であり、2以上6以下の整数が好ましく、2以上5以下の整数がより好ましく、3以上4以下の整数がさらに好ましい。一般的に、n71b価以上のイソシアネートと活性水素基を有するジヒドロキシ化合物やジアミン化合物と反応させることでポリマーを得ることができる。このn71bの値が大きくなるほどイソシアネート分子あたりの架橋点(イソシアネート基)が多くなるため、ポリマー化した時の架橋密度が高くなり、硬化時間の短縮やポリマーの硬度向上させることができる。なお、ここでいう「架橋密度が高くなる」とは、架橋点と架橋点の間の平均分子鎖長が小さくなることを意味する。イソシアネート分子中のイソシアネート基が3以上(n71bが3以上)であると直鎖のポリマーとポリマーをさらに結合することができポリマーの分子量が高くなりやすいため、とりわけ硬化時間の大幅な短縮や硬度などのポリマー物性の劇的な向上が期待できる。一方、イソシアネート製造においては、反応性の高いイソシアネート化合物を加熱すると変性反応が想起され、装置への固着やつまりの原因となるため、イソシアネート化合物中のイソシアネート基の価数n71bが6以下であることが好ましく、5以下であることがより好ましく、4以下であることがさら好ましい。
イソシアネート化合物の変性物は、上述したイソシアネート化合物から誘導された化合物であって、前記イソシアネート化合物のイソシアネート基の一部が、イソシアヌレート基、カルボジイミド基、ウレトンイミン基、及びアロファネート基からなる群より選ばれる1種以上の官能基に変換された化合物である。
R101、R102、R103、及びR104はそれぞれ独立に、2価以上8価以下の有機基であり、関係式:R101=R71b、R102=R71b、R103=R71b、及びR104=R71bを満たす。すなわち、R101、R102、R103、及びR104は、上記R71bと同じである。
R105は1価の有機基であり、関係式:R105=R12を満たす。すなわち、R105は、上記R12と同じである。
カルバメート基含有化合物は、下記一般式(XI)で表される化合物である。
R111は(n111+n112)価の有機基であり、関係式:R111=R11を満たす。すなわち、R111は、上記R11と同じである。
R112は1価の有機基であり、関係式:R112=R12を満たす。すなわち、R112は、上記R12と同じである。
n111は1以上8以下の整数である。
n112は0以上7以下の整数である。
n111とn112の和は2以上8以下の整数であり、関係式:n111+n112=n11+n12を満たす。
炭酸エステルは、下記一般式(III―1a)で表される化合物である。
R311a及びR312aは互いに同一であり、関係式:R311a=R312a=R12を満たす。
本実施形態のイソシアネート組成物は、溶媒として上記炭酸エステル(III-1a)存在下で、下記一般式(Vd)で表されるカルバメート化合物(以下、「カルバメート化合物(Vd)」と称する場合がある)の熱分解反応を行う(熱分解工程)、或いは、溶媒として上記炭酸エステル(III-1a)存在下で、カルバメート化合物(Vd)の熱分解反応(熱分解工程)を行った後に、イソシアネート化合物よりも沸点の低い成分(以下、「軽沸成分」と称する場合がある)の留去(軽沸分離工程)を行うことで得られる。
熱分解工程では、溶媒として炭酸エステル(III-1a)存在下、カルバメート化合物(Vd)の熱分解反応を行うことにより、イソシアネート化合物、カルボニル化合物(I)、及びイソシアネート化合物の変性物を含むイソシアネート組成物を得る。
熱分解工程の後に、軽沸成分を留去してもよく、軽沸成分を気相成分として分離することができれば、留去方法は特に限定されない。ここでいう「軽沸成分(軽沸点成分)」とは、イソシアネート化合物よりも沸点の低い成分を指し、イソシアネート組成物の製造原料となる化合物の種類によって異なるが、主として熱分解工程で用いた炭酸エステル及びヒドロキシ化合物からなる群より選ばれる1種以上の化合物である。
カルバメート化合物(Vd)は、下記一般式(Vd)で表される化合物である。
R51dは、n51d価の有機基であり、関係式:R51d=R71bを満たす。すなわち、R51dは、上記R71bと同じである。
R52dは1価の有機基であり、関係式:R52d=R12を満たす。すなわち、R52dは、上記R12と同じである。
n51dは、カルバメート基の数を表し、関係式:n51d=n71bを満たす。n51dは、2以上8以下の整数であり、2以上6以下の整数が好ましく、2以上5以下の整数がより好ましく、3以上4以下の整数がさらに好ましい。
カルバメート化合物(Vd)の製造方法としては、例えば、炭酸誘導体及びアミン化合物から製造する方法、又は、炭酸誘導体とヒドロキシ化合物とアミン化合物とから製造する方法が好ましい。
炭酸誘導体としては、例えば、尿素、炭酸エステル等が挙げられる。
アミン化合物としては、例えば、下記一般式(IIc)で表される化合物(以下、「アミン化合物(IIc)」と称する場合がある)が好ましく使用される。
R21cは、n21c価の有機基であり、関係式:R21c=R51dを満たす。すなわち、R21cは、上記R51dと同じである。
n21cは、アミノ基の数を表し、関係式:n21c=n51dを満たす。n21cは、2以上8以下の整数であり、2以上6以下の整数が好ましく、2以上5以下の整数がより好ましく、3以上4以下の整数がさらに好ましい。
ヒドロキシ化合物としては、下記一般式(VId)で表される化合物(以下、「ヒドロキシ化合物(VId)」と称する場合がある)が好ましく使用される。
R61dは、1価の有機基であり、関係式:R61d=R12を満たす。すなわち、R61dは、上記R12と同じである。
R611dにおける炭素数1以上20以下のアルキル基としては、例えば、メチル基、エチル基、プロピル基(各異性体)、ブチル基(各異性体)、ペンチル基(各異性体)、ヘキシル基(各異性体)、ヘプチル基(各異性体)、オクチル基(各異性体)、ノニル基(各異性体)、デシル基(各異性体)、ドデシル基(各異性体)、オクタデシル基(各異性体)等が挙げられる。
環A611dは、炭素数6以上20以下の芳香族炭化水素環である。環A611dは、単環であってもよく、多環であってもよく、縮合環であってもよい。
n611dは、置換基R611dの数を示し、1以上10以下の整数である。
本実施形態のイソシアネート化合物の製造方法は、上述したイソシアネート組成物を蒸留精製し、連続的に気相成分として、イソシアネート化合物を回収すること(以下、「高沸分離工程」と称する場合がある)を含む。
高沸分離工程では、上述したイソシアネート組成物を蒸留精製し、連続的にイソシアネート化合物を気相成分として回収し、イソシアネート化合物よりも沸点の高い成分(高沸成分)と分離する(以降、「高沸分離」と称する)。
熱分解工程及び精製工程が行われる反応器及びラインの材質は、カルバメート化合物や、生成物であるヒドロキシ化合物及びイソシアネート化合物、溶媒である炭酸エステルに悪影響を及ぼさなければ、公知のどのようなものであってもよいが、SUS304やSUS316、SUS316L等が安価であり、好ましく使用できる。
<分析方法>
[液体クロマトグラフィー分析方法]
以下の条件で分析を行った。
装置:島津製作所社製、LC-10AT
カラム:Inertsil ODS
粒子径5μm、内径2.1mm、長さ250mm
カラム温度:40℃
展開溶媒:水/アセトニトリル=90/10
展開溶媒流量:1mL/min
検出器:フォトダイオードアレイ検出器
サンプル溶液を1.0g秤量し、酢酸10gを加えて均一に混合した溶液を液体クロマトグラフィー分析サンプルとした。
各標準物質について分析を実施し、作成した検量線を基に、分析サンプル溶液の定量分析を実施した。測定値から、各工程での生成物の収率を算出した。
以下の条件で分析を行った。
装置:島津製作所社製、GC-2010
カラム:DB-1
直径0.25mm、長さ30m、膜厚1.0μm
カラム温度:60℃~300℃
注入口温度:300℃
キャリアガス:ヘリウム
キャリアガス流量:40mL/min
検出器:FID(水素炎イオン化型検出器)
サンプル溶液を1.0g秤量し、アセトニトリル10gと内部標準物質としてアニソール0.1gを加えて均一に混合した溶液をガスクロマトグラフィー分析サンプルとした。
1.イソシアネート化合物の製造
(1)工程(1):カルバメート化合物の製造工程
図1に示す装置を使用して、工程(1)を実施した。
n-ブタノール28.4kg、尿素2.5kg、1,6-ヘキサメチレンジアミンを1.1kgの混合物を槽A101で予混合し、槽A102で170℃まで加温した後、塔底温度240℃、塔頂圧力0.4MPaとした蒸留塔A201に2kg/時間の速度でラインA21から供給し、カルバメート化反応液を、ラインA22を経て貯槽A205に回収した。蒸留塔A201で生じた気相成分はラインA23を通して凝縮器A203に導入し、n-ブタノールとアンモニアの混合物として凝縮した後に、槽A204に抜出し、当該混合液の一部はラインA24より1kg/時間の速度で還流した。
図2に示す装置を用いて、貯槽A205に回収した、工程(1)で得られたカルバメート化反応液に、炭酸ジブチルを1kg/時間の速度にてラインA30から供給し、ラインA31上で予混合し、温度150℃、内圧0.2MPaとした薄膜蒸発器A301に1.5kg/時間で供給し、カルバメート成分を濃縮した反応液を、ラインA32を経て貯槽A303に0.2kg/時間の速度で回収した。回収された反応液を液体クロマトグラフィーで分析したところ、下記式(V-1)で表されるN-置換カルバメート化合物が、1,6-ヘキサメチレンジアミンに対して収率95質量%で生成していた。
図3に示す装置を用いて、熱分解反応を行った。ラインB15から炭酸ジブチルを供給し260℃、800kPaの条件で全還流状態とした蒸留塔B101に、ラインB11から貯槽A303に回収された反応液を供給した。熱分解反応で生じたn-ブタノールを含む気相成分をラインB13に通して凝縮器B103に通じ、凝縮器A403で100℃に冷却して得た凝縮液の一部を、ラインB14を通じて蒸留塔B101に還流し、残りの凝縮液を槽B104に回収した。槽B104に回収した凝縮液を工程(1)の原料として再利用した。一方、イソシアネート化合物を含む反応液を、ラインB12を経て貯槽B105に回収した。回収された反応液を液体クロマトグラフィーで分析したところ、1,6-ヘキサメチレンジイソシアネートが、式(V-1)で表されるN-置換カルバメート化合物に対して収率89質量%で生成していた。また、この留分には、式(I-1-1)で表されるカルボニル化合物(高沸点生成物)が、生成した1,6-ヘキサメチレンジイソシアネートに対して5質量%相当で副生した。
図4に示す装置を用いて、イソシアネート化合物の濃縮を行った。貯槽B105に回収した反応液を塔頂圧20kPa、塔頂温度130℃とした蒸留塔C101に供給した。蒸留塔C101にて炭酸ジブチルを含む気相成分を蒸留分離し、得られた1,6-ヘキサメチレンジイソシアネートを含む液相分はラインC12を通じて貯槽C105へ回収した。蒸留塔C101で生じた気相成分はラインC13を通して凝縮器C103に導入し、炭酸ジブチルを含む溶液として凝縮した後に、槽C104に抜出し、当該溶液の一部はラインC14より還流した。槽C104に回収した凝縮液を工程(2)の原料として再利用した。
図5に示す装置を用いて、イソシアネート化合物の単離(精製)を行った。貯槽C105に回収した1.6-ヘキサメチレンジイソシアネートを含む留分をラインD11から圧力1kPa、150℃とした蒸発機D102で蒸発し、気相成分を蒸発機内部(凝縮部)D104で凝縮しラインD14から抜き出した。ラインD14を通して排出された反応液をガスクロマトグラフィーで分析したところ、回収した1,6-ヘキサメチレンジイソシアネートは純度が99質量%であり、式(I-1-1)で表される成分含有量は0.01質量%であった。高沸点成分は、蒸発機底部D103に回収後、ラインD13から抜き出した。ラインD13を通して排出された当該式(I-1-1)で表されるカルボニル化合物(高沸点生成物)を含む液相成分を分析したところ、液相成分の総質量に対して、6.8質量%の1,6-ヘキサメチレンジイソシアネート、25質量%の式(I-1-1)で表されるカルボニル化合物、68質量%のその他高沸点成分という組成であった。
図6に示す装置を用いて、工程(5)を実施した。
回収した式(I-1-1)で表されるカルボニル化合物(高沸点生成物)を含む液相成分5kgとn-ブタノール5kgとを180℃に保持した状態で、ラインF10から反応器F101に供給した。当該液相成分には、式(I-1-1)で表されるカルボニル化合物以外に、1,6-ヘキサメチレンジアミン骨格、n-ブチル骨格が含まれていた。次いで、水10kgと30質量%水酸化ナトリウム水溶液0.6kgを追添し、250℃で3時間加熱処理を行った。反応液をガスクロマトグラフィーにて分析したところ、1,6-ヘキサメチレンジアミンが含まれており、回収した反応液を、減圧蒸留器F102で減圧蒸留し、1,6-ヘキサメチレンジアミン及びn-ブタノールをそれぞれ収率90質量%(対供給液相成分中に含まれる1,6-ヘキサメチレンジアミン骨格、n-ブチル骨格成分)で回収した。回収した1,6-ヘキサメチレンジアミン及びn-ブタノールはいずれも工程(1)(N-置換カルバメート化合物製造工程)に再利用した。
図7に示す装置を用いて、工程(6)を実施した。
工程(1a)(N-置換カルバメート化合物濃縮工程)、工程(2)(イソシアネート化合物製造工程、熱分解工程)、及び工程(3)(イソシアネート化合物濃縮工程)の各工程から気相として留去された成分を、ラインE12から塔頂温度95℃、塔頂圧40kPaとした蒸留塔E102に1.3kg/時間の速度で供給し、蒸留分離を行った。塔頂からn-ブタノールを0.6kg/時間の速度で、中段成分(サイドカット留分)として炭酸ジブチルを0.54kg/時間の速度で、塔底から高沸点成分を0.16kg/時間の速度で抜出した。n-ブタノールは工程(1)(N-置換カルバメート化合物製造工程)に、炭酸ジブチルは工程(1a)(N-置換カルバメート化合物濃縮工程)及び工程(2)(イソシアネート化合物製造工程、熱分解工程)に用いられる溶媒として再利用し、塔底成分は高沸点成分として抜出した。
実施例1-1の工程(1)のn-ブタノールの代わりにイソプロパノール、工程(2)の炭酸ジブチルの代わりに炭酸ジイソプロピルを用いたこと以外は、実施例1-1に記載の条件で反応および精製を行った。なお、工程(1)~(3)の運転条件は、イソプロパノールならびに炭酸ジイソプロピルの蒸気圧に相当する下記表に記載の条件で運転を行った。
実施例1-1の工程(5)について、実施例1-1に記載の方法に代わり、以下の通り運転を行った。
回収した上記式(I-2)で表されるカルボニル化合物(高沸点生成物)を含む液相成分5kgとn-ブタノール5kgとを180℃に保持した状態で、水10kgと30質量%水酸化ナトリウム水溶液0.6kgを追添し、図8に示すラインE20から平均滞留時間30分となる様な容積を有する攪拌翼つき反応器E201に供給し、250℃で加温した。連続的に反応槽外に抜出したのち、反応器E202に供給し、反応器E201と同温度にて3時間加熱処理を行った。反応液をガスクロマトグラフィーにて分析したところ、1,6-ヘキサメチレンジアミンが含まれており、回収した反応液を、減圧蒸留器E203で減圧蒸留し、1,6-ヘキサメチレンジアミン及びn-ブタノールをそれぞれ収率98質量%で回収した。回収した1,6-ヘキサメチレンジアミン及びn-ブタノールはいずれも工程(1)(N-置換カルバメート化合物製造工程)に再利用した。
実施例1-1の工程(2)で使用した炭酸ジブチルの代わりに、炭酸ジブチル:m-キシレン=9:1(質量比)となる混合溶媒に変えたこと以外は、実施例1-1に記載の方法で運転を行った。なお、工程(3)の運転条件は、炭酸ジブチル及びm-キシレンの蒸気圧に相当する下記表に記載の条件で運転を行った。
実施例1-1の工程(1)で使用した尿素の代わりに、N-ブチル尿素を用いたこと以外は、実施例1-1に記載の方法並びに下記表に記載の条件で運転を行った。なお、蒸留塔A201で生じた気相成分はN-ブチル尿素由来のアンモニアとn-ブチルアミンであった。
実施例1-1の工程(1)で使用した尿素の代わりに、N,N-ジブチル尿素を用いたこと以外は、実施例1-1に記載の方法並びに下記表に記載の条件で運転を行った。なお、蒸留塔A201で生じた気相成分はN,N-ジブチル尿素由来のn-ブチルアミンであった。
実施例1-1の工程(2)について、実施例1-1に記載の方法に代わり、以下の通り運転を行った。
実施例1-1の工程(1a)で得られたN-置換カルバメート濃縮液に予め炭酸ジブチルを混合し、ラインB20から260℃、0.8MPaに保持した管型熱分解反応器B201に0.2kg/時間で供給し、熱分解反応を行ったところ、1,6-ヘキサメチレンジイソシアネートが、式(V-1)で表されるN-置換カルバメート化合物に対して収率86質量%で生成していた。熱分解で得られたイソシアネートを含む成分を回収し、蒸留塔B202に供給した。
実施例1-1の工程(2)について、実施例1-1に記載の方法に代わり、以下の通り運転を行った。
実施例1-1の工程(1)及び工程(2)について、実施例1-1に記載の方法に代わり、以下の通り運転を行った。
図11に示す装置を使用し、カルバメート化工程をおこなった。尿素2.3kg、n-ブタノール28.4kgをそれぞれラインA40、A41より撹拌槽A401に供給した。130℃に加熱して均一な溶液としたのち、ヘキサメチレンジアミン1.5kgをラインA42より撹拌槽A401に約0.3kg/時間で供給した。ヘキサメチレンジアミンを全量供給後2時間撹拌したのち反応液を分析したところ、1,6-ヘキサメチレンジウレアがヘキサメチレンジアミンに対して収率約95質量%で生成していた。ラインA43とラインA45を経て多段蒸留塔A403に供給した。途中、気液分離器A402にてアンモニアを分離しラインA44より抜き出した。
図12に示す装置を使用して、熱分解工程をおこなった。熱分解反応器B401、B402は共にフォーリングフィルム型蒸発器であり、内部圧力を0.8MPaとし、ジャケット温度を260℃とした。分離塔B403の塔頂圧力を0.5kPaとして、デカンの全還流状態とし、ラインB40よりN-置換カルバメートと炭酸ジブチルを予混合した原料を約2.1kg/時間で供給し、ラインB41よりデカンを約1kg/時間で供給した。熱分解反応器B401の液相成分はラインB42を経て連続的に熱分解反応器B402に供給した。熱分解反応器B402底部の液相成分はラインB43より回収した。
実施例1-10及び1-11では、それぞれ実施例1-7及び1-8の工程(1)で使用のn-ブタノールの代わりにフェノール、工程(2)で使用の炭酸ジブチルの代わりに炭酸ジフェニル:m-キシレン=95:5(質量比)としたこと、工程(2)でガス成分としてイソシアネート、フェノール、m-キシレン、炭酸ジフェニルを含む留分を回収し、工程(3)で塔頂成分としてフェノール、m-キシレン、塔底成分としてイソシアネート、炭酸ジフェニル、その他高沸点成分を含む成分を抜出したこと以外は、実施例1-7及び1-8に記載の条件で運転を行った。なお、工程(3)については、非ヒドロキシ化合物を含む低沸点成分が留去可能な蒸気圧に相当する下記表に記載の条件で運転を行った。また、工程(2)で副生するカルボニル化合物(高沸点生成物)は式(I-1-1)で表される構造のn-ブチル基に代わり、フェニル基の構造を有する化合物が副生していた。
実施例1-1の工程(1)の代わりに、下記工程により、N-置換カルバメートの製造を行った。
攪拌槽に炭酸ジフェニル8.9kgをアセトニトリル17.8kgに溶解し、1,6-ヘキサメチレンジアミン1.05kgのアセトニトリル4.2kgを混合し、50℃で3時間攪拌を行った。アセトニトリルを公知の手法で予め留去した混合物を塔底温度110℃、塔頂圧力5kPaとした蒸留塔A201に2kg/時間の速度でラインA21から供給し、カルバメート化反応液を、ラインA22を経て貯槽A205に回収した。蒸留塔A201で生じた気相成分はラインA23を通して凝縮器A203に導入し、フェノールと微量のアセトニトリルの混合物として凝縮した後に、槽A204に抜出し、当該混合液の一部はラインA24より3kg/時間の速度で還流した。
実施例1-12の工程(1)で用いた蒸留塔の代わりに、攪拌混合槽を用いたこと以外は実施例1-12に記載の条件で運転を行った。
実施例1-1の工程(1)において、運転圧力を2MPaGにしたこと以外は実施例1-1に記載の条件で運転を行ったところ、工程(1)の塔頂に低沸成分が殆ど得られず、N-置換カルバメートの収率は3質量%であった。
実施例1-1の工程(2)において、運転圧力を12MPaGにしたこと以外は実施例1-1に記載の条件で運転を行ったところ、工程(2)の塔頂に低沸成分が得られず、1,6-ヘキサメチレンイソシアネートの収率は13質量%であった。
実施例1-1の工程(2)において、滞留時間を90分に変更したこと以外は、実施例1-1に記載の条件で運転を行ったところ、1,6-ヘキサメチレンジイソシアネートの収率は90質量%となった。
実施例1-1の工程(2)において、蒸留塔の代わりに薄膜蒸発器を用いたこと以外は、実施例1-1に記載の条件で運転を行ったところ、1,6-ヘキサメチレンジイソシアネートの収率は89質量%となった。
実施例1-1の工程(2)までの工程について、国際公開第2019/131855号(参考文献2)の実施例1-4を参考に、下記運転を行った。
図13に示す装置を使用し、ラインA50を通じて、フェノール50kgを撹拌槽A501に供給し、次いで、炭酸ジフェニル38kgを供給し均一な溶液とした。撹拌槽A501を50℃に保持した状態で1,6-ヘキサメチレンジアミン8.2kgをゆっくりと添加した。添加終了後3時間撹拌を継続したのち、ラインA51を通じて貯槽A502に反応液を移送した。該反応液を液体クロマトグラフィーで分析したところ、下記式(V-2)で表されるN-置換カルバメートが1,6-ヘキサメチレンジアミンに対して収率95質量%で生成していた。
図14に示す装置を使用し、多段蒸留塔A601にラインA65を通じてp-クレゾールを供給し、リボイラーA602で加熱して多段蒸留塔A601を全還流状態とした。ここにラインA61を通じて上記工程(1)で得た反応液を供給し、上記式(V-2)で表される化合物のエステル交換反応をおこなった。エステル交換反応で生じたフェノールを含む気相成分をラインA63に通して凝縮器A703に供給した。凝縮器A603にて100℃に冷却して得た凝縮液の一部をラインA64を通じて多段蒸留塔A601に供給し、残りの凝縮液を貯槽A604に回収した。得られた反応液はラインA62を通じて貯槽A605に回収した。
反応液を液体クロマトグラフィーで分析したところ、下記式(V-3)で表される化合物が、式(V-2)に対して収率95質量%で生成していた。
図15に示す装置を使用し、熱分解を行った。ラインB50を経て熱分解装置B501に原料となるN-置換カルバメート液と炭酸ジフェニルを質量比で4:1となる様な組成で供給した。熱分解装置B501は、カルバメートの熱分解反応によってイソシアネートを生成するための装置であり、フォーリングフィルム型管型反応器と、イソシアネートを含む液相成分と気相成分を分離する分離槽からなるものであった。内部圧力を250kPaとし、外部加熱により250℃に加熱した。
炭酸ジフェニルと1,6-ヘキサメチレンジイソシアネートの分離を行った。蒸留分離に必要な熱量はリボイラーB505より供給し、分離塔B502の塔底より塔底成分をラインB53から回収した。
分離塔B502の塔頂から回収した1,6-ヘキサメチレンジイソシアネートを含む成分は、凝縮器B504及びラインB55を経てセラミック製ラシヒリングを充填した精製塔B503に供給し、1,6-ヘキサメチレンジイソシアネートの蒸留精製を行った。蒸留精製に必要な熱量はリボイラーB507より供給した。
精製塔B503の塔頂より、凝縮器B506及びラインB56を経て1,6-ヘキサメチレンジイソシアネートが2.4kg/時間で回収された。精製塔B503の塔底成分はラインB54より抜き出した。
実施例1-1の工程(2)で1,6-ヘキサメチレンジイソシアネートを含む留分を得た後、工程(3)を実施しなかったこと以外は実施例1-1に記載の条件で運転を行ったところ、工程(4)で回収される1,6-ヘキサメチレンジイソシアネート留分に低沸分である炭酸ジブチルが同伴し、製品イソシアネート純度が低下した。また、工程(4)に供給される液レートに対する高沸留分が少なく、液枯れが発生し、運転が困難となった。
実施例1-1の工程(3)で1,6-ヘキサメチレンジイソシアネートを含む留分を得た後、工程(4)を実施しなかったこと以外は実施例1-1に記載の条件で運転を行ったところ、得られた当該イソシアネート留分には、一般式(I)で表されるカルボニル化合物が9wt%、高沸点副生物が8wt%相当含まれていた。
実施例1-1の工程(2)において、熱分解溶媒として炭酸ジブチルからフェノールに変え、運転圧力を0.4MPaとしたこと以外は実施例1-1に記載の条件で運転を行ったところ、工程(2)の塔頂に低沸成分が殆ど得られず、1,6-ヘキサメチレンジイソシアネートの収率は5wt%であった。また、該イソシアネートを含む留分中には式(I-1-1)で表されるカルボニル化合物(高沸点生成物)が副生せず、工程(3)及び工程(4)で1,6-ヘキサメチレンジイソシアネートの単離を試みたが、当該イソシアネート化合物の変性及び高分子量化が進行し、運転困難となり運転停止した。
国際公開第2014-157636号(参考文献3)の実施例1-13を参考に、下記運転を行った。
尿素1.6kg、フェノール23kgをそれぞれ図11のラインA40、A41より撹拌槽A401に供給した。130℃に加熱して均一な溶液としたのち、ヘキサメチレンジアミン1.4kgをラインA42より撹拌槽A401に約0.3kg/時間で供給した。ヘキサメチレンジアミンを全量供給後2時間撹拌したのち反応液を分析したところ、1,6-ヘキサメチレンジウレアがヘキサメチレンジアミンに対して収率約95質量%で生成していた。1,6-ヘキサメチレンジウレアを含む当該反応液をラインA43とラインA45を経て多段蒸留塔A503に供給した。途中、気液分離器A402にてアンモニアを分離しラインA44より抜き出した。
次に図16に示す装置を使用して、予備濃縮をおこなった。予備濃縮器A701はフォーリングフィルム型蒸発器であり、ジャケット温度を250℃、内部の圧力を3kPaとした。カルバメート化工程で貯槽A407に回収した反応液を、ラインA71を通して約10kg/時間で予備濃縮器A701に供給した。生成した気相成分はラインA73より抜き出した。回収物はフェノールであった。一方、予備濃縮器A701の液相成分は、ラインA72を経て貯槽A702に約2.5kg/時間で回収した。1,6-ヘキサンジイル-ジ(カルバミン酸フェニル)がヘキサメチレンジアミンに対して収率96質量%で得られた。
図17に示す装置を使用し、エステル交換をおこなった。連続多段蒸留塔A801はラシヒリングを充填した充填塔であり、予め、塔底部に4-(α,α-ジメチルベンジル)フェノールを供給して、塔内を4-(α,α-ジメチルベンジル)フェノールの全還流状態とした。必要な熱量はリボイラーA802より供給した。塔底温度は250℃であった。予備濃縮工程で貯槽A702に回収した液をラインA81より約1kg/時間で供給し、ラインA85より4-(α,α-ジメチルベンジル)フェノールを約4.2kg/時間で供給した。ラインA84はラインA81からの反応液供給が開始すると同時に閉止した。塔頂よりフェノールを主成分とする気相成分を抜き出し、ラインA83を経て、凝縮器A803で凝縮して貯槽A804に回収した。一方、塔底部より反応液を抜き出し、ラインA82を経て貯槽A805に約2.1kg/時間で回収した。貯槽A805に回収した液を分析したところ、1,6-ヘキサンジイル-(カルバミン酸(4-(α,α-ジメチルベンジル)フェニル))が1,6-ヘキサンジイル-(カルバミン酸フェニル)に対して収率93質量%で生成していた。
図18に示す装置を使用して、熱分解をおこなった。熱分解反応器B601、B602は共にフォーリングフィルム型蒸発器であり、内部圧力を1kPaとし、ジャケット温度を250℃とした。分離塔B603の塔頂圧力を0.5kPaとして、ベンジルトルエンの全還流状態とし、ラインB60より前工程で得られた非プロトン性溶媒を含まないN-置換カルバメートの4-(α,α-ジメチルベンジル)フェノール溶液を約2.1kg/時間で供給した。熱分解反応器B601の液相成分はラインB61bを経て連続的に熱分解反応器B602に供給した。熱分解反応器B602の底部から回収される液相成分をラインB63より回収した。熱分解反応器で生成した気相成分は、ラインB61a及びB62より熱分解反応器より抜き出して、分離塔B603に供給した。ラインB61a及びB62に含まれるガス成分をサンプリングして分析したところ、対応する1,6-ヘキサメチレンジイソシアネートが原料N-置換カルバメートに対して収率88質量%で得られた。B63の払い出し液は高沸成分を含んでおり、式(I-1)で示されるカルボニル化合物が副生していなかったことから、高濃縮条件で運転するとB63流路に部分的にスケーリングが発生した。また、低沸抜出し成分量が多く、低圧での運転で圧力損失を低減するため、ラインB61a及びB62の配管径を大きく取る必要があった。
分離塔B603にはセラミック製ラシヒリングを充填し、蒸留分離に必要な熱量はリボイラーB605より供給した。
分離塔B603の塔頂よりヘキサメチレンジイソシアネートを含む成分を回収し、凝縮器B604で凝縮した後、ラインB64を経て分離塔B607に供給した。蒸留分離に必要な熱量をリボイラーB608より供給して、分離塔B607にてヘキサメチレンジイソシアネートの蒸留分離を行い、分離塔B607の中段に具備するラインB67より1,6-ヘキサメチレンジイソシアネートを回収した。
実施例1-10~1-12、及び1-14で使用した原料ヘキサメチレンジアミンの量を変えたこと以外は実施例1-10~1-12、及び1-14に記載の方法で製造した。
実施例1-12、及び1-14で使用した各工程の蒸留塔をSUS304製シーブトレイ塔に変えたこと以外は実施例1-12、及び1-14に記載の方法で製造した。
実施例1-12、及び1-14で使用した各工程の充填物をラシヒリングからSUS304製メラパック(スルザー社製)に変えたこと以外は実施例1-12、及び1-14に記載の方法で製造した。
実施例1-12、及び1-14の工程(4)の薄膜蒸発器に代わり、セラミックス製ラシヒリングを充填した充填塔で製造したこと以外は、実施例1-12、及び1-14に記載の方法で製造した。
実施例1-12、及び1-14の工程(5)で使用した30質量%水酸化ナトリウム水溶液の代わりに、30質量%水酸化カリウム水溶液を使用したこと以外は、実施例1-12、及び1-14に記載の方法で製造した。
(イソシアネート化合物の製造)
1.工程(1):N-置換カルバメート化合物製造工程
図1に示す装置を用いて、4-アミノメチル-1,8-オクタンジアミン3.1kg、尿素3.7kg、及びn-ブタノール88.2kgの混合物を槽A101で予混合し、槽A102で170℃まで加温した後、塔底温度240℃、塔頂圧力0.4MPaとした蒸留塔A201に1.0kg/時間の速度でラインA21から供給し、カルバメート化反応液を、ラインA22を経て貯槽A205に回収した。蒸留塔A201で生じた気相成分はラインA23を通して凝縮器A203に導入し、n-ブタノールとアンモニアの混合物として凝縮した後に、槽A204に抜出し、当該混合液の一部はラインA24より1.3kg/時間の速度で還流した。
図2に示す装置を用いて、貯槽A205に回収した、工程(1)で得られたカルバメート化反応液に、炭酸ジブチルを0.50kg/時間の速度にてラインA31上で予混合し、温度150℃、内圧30kPaとした薄膜蒸発器A301に2.1kg/時間で供給し、カルバメート成分を濃縮した反応液を、ラインA32を経て貯槽A303に0.2kg/時間の速度で回収した。回収された反応液を液体クロマトグラフィーで分析したところ、下記式(V-1)で表されるN-置換カルバメート化合物が、4-アミノメチル-1,8-オクタンジアミンに対して収率95質量%で生成していた。
図3に示す装置を用いて、熱分解反応を行った。ラインB15から炭酸ジフェニルを供給し250℃、25kPaの条件で全還流状態とした蒸留塔B101に、ラインB11から貯槽A303に回収された反応液を供給した。熱分解反応で生じたフェノールを含む気相成分をラインB13に通して凝縮器B103に通じ、凝縮器B103で100℃に冷却して得た凝縮液の一部を、ラインB14を通じて蒸留塔B101に還流し、残りの凝縮液を槽B104に回収した。槽B104に回収した凝縮液を工程(1)の原料として再利用した。一方、イソシアネート化合物を含む反応液を、ラインB12を経て貯槽B105に回収した。回収された反応液を液体クロマトグラフィーで分析したところ、式(VII-1)で表されるイソシアネート化合物(4-イソシアナトメチル-1,8-オクタメチレンジイソシアネート)が、式(V-4)で表されるN-置換カルバメート化合物に対して収率92質量%で生成していた。また、この留分には、式(I-5a-1)~(I-5c-1)で表されるカルボニル化合物の混合物(高沸点生成物)が、生成した式(VII-1)で表されるイソシアネート化合物に対して5質量%相当で副生した。
図4に示す装置を用いて、イソシアネート化合物の濃縮を行った。貯槽B105に回収した反応液を塔頂圧2kPa、塔頂温度170℃とした蒸留塔C101に供給した。蒸留塔C101にて炭酸ジフェニルを含む気相成分を蒸留分離し、得られた4-イソシアナトメチル-1,8-オクタメチレンジイソシアネートを含む液相分はラインC12を通じて貯槽C105へ回収した。蒸留塔C101で生じた気相成分はラインC13を通して凝縮器C103に導入し、炭酸ジフェニルを含む溶液として凝縮した後に、槽C104に抜出し、当該溶液の一部はラインC14より還流した。槽C104に回収した凝縮液を工程(2)の原料として再利用した。
図5に示す装置を用いて、イソシアネート化合物の単離(精製)を行った。貯槽C105に回収した4-イソシアナトメチル-1,8-オクタメチレンジイソシアネートを含む留分をラインD11から圧力100Pa、200℃とした蒸発機D102で蒸発し、気相成分を蒸発機内部(凝縮部)D104で凝縮しラインD14から抜き出した。ラインD14を通して排出された反応液をガスクロマトグラフィーで分析したところ、回収した4-イソシアナトメチル-1,8-オクタメチレンジイソシアネートは純度が99.1質量%であった。高沸点成分は、蒸発機底部D103に回収後、ラインD13から抜き出した。ラインD13を通して排出された当該式(I-5a-1)~(I-5c-1)で表されるカルボニル化合物(高沸点生成物)を含む液相成分を分析したところ、液相成分の総質量に対して、6.8質量%の4-イソシアナトメチル-1,8-オクタメチレンジイソシアネートを、25.0質量%の式(I-1)で表されるカルボニル化合物、68.2質量%のその他高沸点成分という組成であった。
図6に示す装置を用いて、工程(5)を行った。
回収した式(I-5a-1)~(I-5c-1)で表されるカルボニル化合物(高沸点生成物)を含む液相成分5kgとフェノール5kgとを180℃に保持した状態で、ラインF10から反応器F101に供給した。当該液相成分には、式(I-5a-1)~(I-5c-1)で表されるカルボニル化合物以外に4-アミノメチル-1,8-オクタンジアミン骨格、n-ブチル骨格が含まれていた。次いで、水10kgと30質量%水酸化ナトリウム水溶液0.6kgを追添し、240℃で3時間加熱処理を行った。反応液をガスクロマトグラフィーにて分析したところ、4-アミノメチル-1,8-オクタンジアミンが含まれており、回収した反応液を、減圧蒸留器F102で減圧蒸留し、4-アミノメチル-1,8-オクタンジアミン及びn-ブタノールをそれぞれ収率90質量%で回収した。回収した4-アミノメチル-1,8-オクタンジアミン及びn-ブタノールはいずれも工程(1)(N-置換カルバメート化合物製造工程)に再利用した。
図7に示す装置を用いて、工程(6)を実施した。
工程(1a)(N-置換カルバメート化合物濃縮工程)、工程(2)(イソシアネート化合物製造工程、熱分解工程)、及び工程(3)(イソシアネート化合物濃縮工程)の各工程から気相として留去された成分を、ラインE12から塔頂温度95℃、塔頂圧40kPaとした蒸留塔E101に1.3kg/時間の速度で供給し、蒸留分離を行った。塔頂からn-ブタノールを0.6kg/時間の速度で、中段成分(サイドカット留分)として炭酸ジブチルを0.54kg/時間の速度で、塔底から高沸点成分を0.16kg/時間の速度で抜出した。n-ブタノールは工程(1)(N-置換カルバメート化合物製造工程)に、炭酸ジブチルは工程(1a)(N-置換カルバメート化合物濃縮工程)及び工程(2)(イソシアネート化合物製造工程、熱分解工程)に用いられる溶媒として再利用し、塔底成分は高沸点成分として抜出した。
下記表に記載の通り、工程(1)~工程(5)の条件を種々変更して、各種イソシアネートの製造を行った。
HDA:ジアミノヘキサン
TAN:4-アミノメチル-1,8-オクタンジアミン
TAH:トリアミノヘキサン
TAU:トリアミノウンデカン
IPDA:3-アミノメチル-3,5,5-トリメチルシクロヘキシルアミン
HMDA:4,4’-メチレンビス(シクロヘキシルアミン)
XDA:ジアミノキシレン
MDA:4,4’-メチレンジアニリン
TAB:トリアミノベンゼン
TAMBトリアミノメチルベンゼン
TAPMB:トリス(アミノプロパン-イル)-メチルベンゼン
LTA:2,6-ジアミノヘキサン酸2-アミノエチル
ODA:オルニチンエチルエステルジアミン
LDA:リジンエチルエステルジアミン
DEC:炭酸ジエチル
DPC:炭酸ジフェニル
n-BuOH:n-ブタノール
iPrOH:イソプロパノール
PhOH:フェノール
MeCN:アセトニトリル
p-cresol:p-クレゾール
PCP:4-(α,α-ジメチルベンジル)フェノール
NH3:アンモニア
PhOH:フェノール
EtOH:エタノール
DBC:炭酸ジブチル
DiPrC:炭酸ジイソプロピル
m-Xy:m-キシレン
DEC:炭酸ジエチル
DPC:炭酸ジフェニル
PCP:4-(α,α-ジメチルベンジル)フェノール
FF:フォーリングフィルム型蒸発器
FF×2:2器のフォーリングフィルム型蒸発器を直列に接続
n-BuOH:n-ブタノール
PhOH:フェノール
EtOH:エタノール
DBC:炭酸ジブチル
DiPrC:炭酸ジイソプロピル
m-Xy:m-キシレン
DEC:炭酸ジエチル
DPC:炭酸ジフェニル
heavy:イソシアネート化合物よりも沸点の高い副生物
DPC:炭酸ジフェニル
n-BuOH:n-ブタノール
iPrOH:イソプロパノール
PhOH:フェノール
<分析方法>
(1)1H-NMR分析方法
装置として、日本電子(株)社製JNM-A400 FT-NMRシステムを用いて、1H-NMR分析を実施した。
サンプル溶液を0.3g秤量し、重クロロホルムを0.7gと内部標準物質としてジメチルジフェニルシラン0.05gを加えて均一に混合した溶液をNMR分析サンプルとした。
各標準物質について分析を実施し、作成した検量線を基に、分析サンプル溶液の定量分析を実施した。
以下の条件で分析を行った。
装置:島津製作所社製、GC-2010
カラム:DB-1
直径0.25mm、長さ30m、膜厚1.0μm
カラム温度:60℃~300℃
注入口温度:300℃
キャリアガス:ヘリウム
キャリアガス流量:40mL/min
検出器:FID(水素炎イオン化型検出器)
サンプル溶液を1.0g秤量し、アセトニトリルを10gと内部標準物質としてアニソールを0.1g加えて均一に混合した溶液をガスクロマトグラフィー分析サンプルとした。
以下の条件で分析を行った。
装置:島津製作所社製、LC-10AT
カラム:Inertsil ODS
粒子径5μm、内径2.1mm、長さ250mm
カラム温度:40℃
展開溶媒:水/アセトニトリル=90/10
展開溶媒流量:1mL/min
検出器:フォトダイオードアレイ検出器
サンプル溶液を1.0g秤量し、酢酸10gを加えて均一に混合した溶液を液体クロマトグラフィー分析サンプルとした。
各標準物質について分析を実施し、作成した検量線を基に、分析サンプル溶液の定量分析を実施した。
コンデンサーを取り付けた2L四ツ口フラスコに尿素13.2g、クレゾール500g、7-メチル-1,5,7-トリアザビシクロ[4.4.0]デカ-5-エン0.3gを投入し、攪拌しながら135℃に昇温した。これに、180℃に保温した配管から窒素ガスを吹き込み、大気圧(101.325kPa、以降同様)下にてマスフローメータにより流量を300cc/分に制御した。吹き込んだ窒素ガスはコンデンサーの上部より抜き出し、硫酸水溶液に通した後に系外へ放出した。次いで、滴下漏斗を用いてヘキサメチレンジアミン11.6gをクレゾール60gに溶解した溶液を投入し、大気圧下、180℃にて1時間攪拌した。反応液の一部を抜き出して液体クロマトグラフィーで分析したところ、N,N’-ヘキサンジイル-ジ(カルバミン酸クレゾイルエステル)がヘキサメチレンジアミンに対して収率72.6質量%で生成していた。
実施例2-1で使用した第1級アミン化合物、尿素、ヒドロキシ化合物、触媒を以下の表に記載の種類及び使用量に変更したこと以外は、同じ方法で対応するカルバメートを得た。得られたカルバメートの第1級アミン化合物に対する収率は以下の表に記載のとおりであった。なお、各表中のpKa値は、CAS SciFindernに収録された予測値である。
HDA:ヘキサメチレンジアミン
PDA:ペンタメチレンジアミン
TTA:4-(アミノメチル)オクタン-1,8-ジアミン
IPDA:イソホロンジアミン
HMDA:4、4’-メチレンビス(シクロヘキサンアミン)
TDA:ジアミノトルエン
図19に示す装置を使用し、カルバメート化工程を実施した。ヘキサメチレンジアミン6.3kg、尿素7.2kg、クレゾール180kg、1,5-ジアザビシクロ-[4.3.0]ノナ-5-エン0.13kgの混合物を約3.0kg/時間でライン11よりヘリパックNo.4を充填した蒸留塔101(理論段数39段)に供給した。ライン14より、凝縮器103で得られたクレゾールと尿素との混合液を約3.5kg/時間で供給した。余剰の凝縮成分は貯槽104に回収した。蒸留塔101は、カルバメート合成工程を行うための装置であり、リボイラー102で加熱することにより塔底温度を240℃とし、塔頂圧力0.15MPaとした。濃度調整のために、塔底のライン15よりクレゾールを供給した。塔底より反応液を抜き出し、ライン12を経て貯槽105に回収した。貯槽105より回収した液を液体クロマトグラフィーで分析したところ、N,N’-ヘキサンジイル-ジ(カルバミン酸クレゾイルエステル)がヘキサメチレンジアミンに対して収率90.4質量%で生成していた。
実施例2-41で使用した第1級アミン化合物、尿素、ヒドロキシ化合物、触媒を以下の表に記載の種類及び使用量に変更し、塔底温度を以下の表に記載の値に変更したこと以外は、同じ方法で対応するカルバメートを得た。得られたカルバメートの第1級アミン化合物に対する収率は以下の表に記載のとおりであった。
1.カルバメート合成工程
コンデンサーを取り付けた2L四ツ口フラスコに尿素13.2g、フェノール500g、2,4-ルチジン30.0gを投入し、攪拌しながら135℃に昇温した。これに、180℃に保温した配管から窒素ガスを吹き込み、大気圧下にてマスフローメータにより流量を300cc/分に制御した。吹き込んだ窒素ガスはコンデンサーの上部より抜き出し、硫酸水溶液に通した後に系外へ放出した。次いで、滴下漏斗を用いてイソホロンジアミン17.0gをフェノール60gに溶解した溶液を投入し、大気圧下、180℃にて12時間攪拌した。反応液の一部を抜き出して液体クロマトグラフィーで分析したところ、((3,3,5-トリメチル-5-((フェノキシカルボニル)アミノ))シクロヘキシル)メチルカルバミン酸フェニルがイソホロンジアミンに対して収率85.6質量%で生成していた。
上記で得られた溶液を60℃にて減圧して大部分のフェノールと2,4-ルチジンを除去し、得られた固体100gを80℃にてベンジルトルエン50gに溶解させた。
コンデンサーを取り付けた1L四ツ口フラスコにベンジルトルエン350gを投入し、攪拌しながら250℃に加熱し、内部の圧力を22kPaとした。これに、滴下漏斗を用いてカルバメート濃縮工程にて得られたカルバメートのベンジルトルエン溶液の全量を投入し、熱分解を実施した。熱分解により生成する軽沸化合物をコンデンサーの上部より回収しながら、3時間反応させた。その後、内部を大気圧として室温まで冷却し、四ツ口フラスコに残った反応液の一部を抜き出してガスクロマトグラフィーで分析したところ、イソホロンジイソシアネートが((3,3,5-トリメチル-5-((フェノキシカルボニル)アミノ))シクロヘキシル)メチルカルバミン酸フェニルに対して収率90.3質量%で生成していた。カルバメート合成工程で仕込んだイソホロンジアミンに対しての収率は、77.3質量%であった。
1.カルバメート合成工程
2,4-ルチジンを使用しなかったこと以外は、実施例2-123と同じ方法で対応するジカルバメートを得た。得られたジカルバメートのイソホロンジアミンに対する収率は56.3質量%であった。
上記で得られた溶液を60℃にて減圧してフェノールの大部分を除去し、得られた固体100gを80℃にてベンジルトルエン50gに溶解させた。
実施例2-123と同じ方法で熱分解を実施し、イソホロンジイソシアネートを得た。得られたジイソシアネートのジカルバメートに対する収率は72.3質量%であった。カルバメート合成工程で仕込んだイソホロンジアミンに対しての収率は、40.6質量%であった。
1.カルバメート合成工程
2,4-ルチジンの代わりに水酸化ナトリウム12.0gを使用したこと以外は、実施例2-123と同じ方法で対応するジカルバメートを得た。得られたジカルバメートのイソホロンジアミンに対する収率は79.2質量%であった。
上記で得られた溶液を60℃にて減圧してフェノールの大部分を除去し、得られた固体100gを80℃にてベンジルトルエン50gに溶解させた。
実施例2-123と同じ方法で熱分解を実施し、イソホロンジイソシアネートを得た。得られたジイソシアネートのジカルバメートに対する収率は49.0質量%であった。カルバメート合成工程で仕込んだイソホロンジアミンに対しての収率は、38.8質量%であった。また、反応液をNMRにより分析したところ、イソシアヌレート環が原料アミノ基に対して収率10.3質量%で生成しており、フラスコ表面にゲル状の付着物が析出していた。
1.カルバメート合成工程
尿素21.0gを使用し、2,4-ルチジンの代わりに4-ピコリン43.5gを使用し、イソホロンジアミンの代わりに4-(アミノメチル)オクタン-1,8-ジアミン18.0gを使用し、反応時間を18時間としたこと以外は、実施例2-123と同じ方法で対応するトリカルバメートを得た。得られたトリカルバメートの4-(アミノメチル)オクタン-1,8-ジアミンに対する収率は88.5質量%であった。
上記で得られた溶液を60℃にて減圧して大部分のフェノールと4-ピコリンを除去し、得られた固体100gを80℃にてベンジルトルエン50gに溶解させた。
実施例2-123と同じ方法で熱分解を実施し、4-(イソシアナトメチル)オクタン-1,8-ジイソシアネートを得た。得られたトリイソシアネートのトリカルバメートに対する収率は85.7質量%であった。カルバメート合成工程で仕込んだ4-(アミノメチル)オクタン-1,8-ジアミンに対しての収率は、75.9質量%であった。
1.カルバメート合成工程
4-ピコリンを使用しなかったこと以外は、実施例2-124と同じ方法で対応するトリカルバメートを得た。得られたトリカルバメートの4-(アミノメチル)オクタン-1,8-ジアミンに対する収率は47.5質量%であった。
上記で得られた溶液を60℃にて減圧してフェノールの大部分を除去し、得られた固体100gを80℃にてベンジルトルエン50gに溶解させた。
実施例2-123と同じ方法で熱分解を実施し、4-(イソシアナトメチル)オクタン-1,8-ジイソシアネートを得た。得られたトリイソシアネートのトリカルバメートに対する収率は51.2質量%であった。カルバメート合成工程で仕込んだ4-(アミノメチル)オクタン-1,8-ジアミンに対しての収率は、24.3質量%であった。
1.カルバメート合成工程
尿素14.0gを使用し、フェノールの代わりにクレゾールを使用し、2,4-ルチジンの代わりにジイソプロピルエチルアミン8.0gを使用し、イソホロンジアミンの代わりに4,4’-メチレンビス(シクロヘキシルアミン)22.0gを使用したこと以外は、実施例2-123と同じ方法で対応するジカルバメートを得た。得られたジカルバメートの4,4’-メチレンビス(シクロヘキシルアミン)に対する収率は90.3質量%であった。
上記で得られた溶液を60℃にて減圧して大部分のクレゾールとジイソプロピルエチルアミンを除去し、得られた固体100gを80℃にてベンジルトルエン50gに溶解させた。
実施例2-123と同じ方法で熱分解を実施し、ビス(4-イソシアナトシクロヘキシル)メタンを得た。得られたジイソシアネートのジカルバメートに対する収率は92.4質量%であった。カルバメート合成工程で仕込んだ4,4’-メチレンビス(シクロヘキシルアミン)に対しての収率は、83.3質量%であった。
1.カルバメート合成工程
ジイソプロピルエチルアミンを使用しなかったこと以外は、実施例2-125と同じ方法で対応するトリカルバメートを得た。得られたジカルバメートの4,4’-メチレンビス(シクロヘキシルアミン)に対する収率は51.8質量%であった。
上記で得られた溶液を60℃にて減圧してクレゾールの大部分を除去し、得られた固体100gを80℃にてベンジルトルエン50gに溶解させた。
実施例2-123と同じ方法で熱分解を実施し、ビス(4-イソシアナトシクロヘキシル)メタンを得た。得られたジイソシアネートのジカルバメートに対する収率は68.9質量%であった。カルバメート合成工程で仕込んだ4,4‘-メチレンビス(シクロヘキシルアミン)に対しての収率は、35.7質量%であった。
1.カルバメート合成工程
尿素6.7gを使用し、フェノールの代わりにp-クミルフェノールを使用し、2,4-ルチジンの代わりに1,5-ジアザビシクロ-[4.3.0]ノナ-5-エン0.13gを使用し、イソホロンジアミンの代わりにヘキサメチレンジアミン5.8gを使用したこと以外は、実施例2-123と同じ方法で対応するジカルバメートを得た。得られたジカルバメートのヘキサメチレンジアミンに対する収率は92.2質量%であった。
上記反応混合物を、同じ装置で攪拌しながら250℃に加熱し、内部の圧力を85kPaとした。熱分解により生成する軽沸化合物をコンデンサーの上部より回収しながら、3時間反応させた。その後、内部を大気圧として室温まで冷却し、コンデンサー上部から回収した溶液の一部をガスクロマトグラフィーで分析したところ、ヘキサメチレンイソシアネートがヘキサメチレンジアミンに対して収率88.4質量%で生成していた。カルバメート合成工程で仕込んだヘキサメチレンジアミンに対しての収率は、81.4質量%であった。
1.カルバメート合成工程
1,5-ジアザビシクロ-[4.3.0]ノナ-5-エン0.13gを使用なかった以外は、実施例2-126と同じ方法で対応するジカルバメートを得た。得られたジカルバメートのヘキサメチレンジアミンに対する収率は49.0質量%であった。
実施例2-123と同じ方法で熱分解を実施し、ヘキサメチレンイソシアネートを得た。得られたヘキサメチレンイソシアネートのヘキサメチレンジアミンに対する収率は65.6質量%であった。カルバメート合成工程で仕込んだヘキサメチレンジアミンに対しての収率は、32.1質量%であった。
<分析方法>
(1)1H-NMR分析方法
装置として、日本電子(株)社製JNM-A400 FT-NMRシステムを用いて、1H-NMR分析を実施した。
サンプル溶液を0.3g秤量し、重クロロホルムを0.7gと内部標準物質としてジメチルジフェニルシラン0.05gを加えて均一に混合した溶液をNMR分析サンプルとした。
各標準物質について分析を実施し、作成した検量線を基に、分析サンプル溶液の定量分析を実施した。
(2-1)ガスクロマトグラフィー分析サンプルの調製
サンプル溶液を1.0g秤量し、アセトニトリルを10gと内部標準物質としてアニソールを0.1g加えて均一に混合した溶液をガスクロマトグラフィー分析サンプルとした。
以下の条件で分析を行った。
装置:島津製作所社製、GC-2010
カラム:DB-1
直径0.25mm、長さ30m、膜厚1.0μm
カラム温度:60℃~300℃
注入口温度:300℃
キャリアガス:ヘリウム
キャリアガス流量:40mL/min
検出器:FID(水素炎イオン化型検出器)
(3-1)液体クロマトグラフィー分析サンプルの調製
サンプル溶液を1.0g秤量し、酢酸10gを加えて均一に混合した溶液を液体クロマトグラフィー分析サンプルとした。
以下の条件で分析を行った。
装置:島津製作所社製、LC-10AT
カラム:Inertsil ODS
粒子径5μm、内径2.1mm、長さ250mm
カラム温度:40℃
展開溶媒:水/アセトニトリル=90/10
展開溶媒流量:1mL/min
検出器:フォトダイオードアレイ検出器
各標準物質について分析を実施し、作成した検量線を基に、分析サンプル溶液の定量分析を実施した。
1.イソシアネート化合物(VIIa)分離後の液相成分の回収
図20に示す装置を使用して、以下に示す工程(x1)~工程(x4)を実施した。
2,4-トルエンジアミン9.8kg、尿素10.3kg、及び4-(1,1,3,3-テトラメチルブチル)フェノール261.9kgの混合物を90kg/時間でラインa1より連続多段蒸留塔a101に供給した。
連続多段蒸留塔a101の底部より反応液を、ラインa4を経て熱分解装置a102に供給した。熱分解装置a102は管型反応器であり、カルバメート化合物の熱分解反応によってイソシアネート化合物を生成するための装置である。内部圧力を1kPaとし、外部加熱により250℃に加熱した。
熱分解装置a102にて生成した成分をラインa6から分離塔a103に供給し、ラインa15によりペンタデカンを供給し、4-(1,1,3,3-テトラメチルブチル)フェノールと2,4-トルエンジイソシアネートの分離を行った。蒸留分離に必要な熱量はリボイラーa112より供給した。
熱分解装置a102の底部より回収される液相成分の一部はラインa7を経て回収し、液温度を180℃として、続く工程(a1)に高沸点化合物を含む液相成分として供給した。残りの液相成分はラインa5を経て、再び熱分解装置a102に供給した。
図21に示す装置を使用して、以下に示す工程(a1)及び工程(a2)を実施した。
ラインa7を経て回収した高沸点化合物を含む液相成分50kgと4-(1,1,3,3-テトラメチルブチル)フェノール50kgを180℃に保持した状態でラインb1を通じて耐圧反応器b101に供給した。
次いで、水50kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:30モル倍)と水酸化ナトリウム1.5kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:0.41モル倍)を耐圧反応器b101に供給し、混合液とした。該耐圧反応器b101を280℃、圧力6.6MPaで5時間加熱した。
図22に示す装置を使用して、工程(b)を実施した。
=(工程(b)で回収した2,4-トルエンジアミンの質量)×100/(工程(x4)で回収した液相成分中の高沸化合物が有する2,4-トルエンジアミンに由来する構造単位の質量)
前記工程(b)で回収した粗2,4-トルエンジアミン含有液を蒸留精製した。回収した2,4-トルエンジアミンの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、2,4-トルエンジアミンの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
貯槽c102へ回収した反応液を減圧蒸留し、粗2,4-トルエンジアミン含有液と粗4-(1,1,3,3-テトラメチルブチル)フェノール含有液をそれぞれ回収した。収率は84質量%であった。なお、収率は、以下の式により算出した。式中、「工程(x4)で回収した液相成分中の高沸化合物が有するヒドロキシ化合物に由来する構造単位の質量」は、カルバメート基、アロファネート基、フリース転位末端、フリース環化体、及びオキシカルボニルカルバメート基×2の合計質量である。
=(工程(d)で回収したヒドロキシ化合物の質量)×100/(工程(x4)で回収した液相成分中の高沸化合物が有するヒドロキシ化合物に由来する構造単位の質量)
前記工程(e)で回収した粗4-(1,1,3,3-テトラメチルブチル)フェノール含有液を蒸留精製した。回収した4-(1,1,3,3-テトラメチルブチル)フェノールの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、4-(1,1,3,3-テトラメチルブチル)フェノールの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
図20に示す装置において、工程(c)で精製した2,4-トルエンジアミンと工程(f)で精製した4-(1,1,3,3-テトラメチルブチル)フェノールを使用し、不足分の2,4-トルエンジアミンと4-(1,1,3,3-テトラメチルブチル)フェノールは新たに追加して、ラインa1より連続多段蒸留塔a101に供給し、上記工程(x1)~(x3)を行ったところ、ラインa18より2,4-トルエンジイソシアネートが2.4kg/時間で回収された。
図21に示す装置において、前記工程(b)で貯槽c103に回収した液相成分と、前記工程(c)及び前記工程(f)より回収された蒸留残渣を使用し、不足分の水と水酸化ナトリウムと4-(1,1,3,3-テトラメチルブチル)フェノールは新たに追加して、ラインb1を通じてこれらの回収した液相成分を耐圧反応器b101に供給し、前記工程(a1)と前記工程(a2)を行ったところ、ラインb5を通じて貯槽b102に回収した反応液には、2,4-トルエンジアミンが46モル含まれていた。
1.イソシアネート化合物(VIIa)分離後の液相成分の回収
(1)工程(x1):カルバメート化合物の製造工程
図23に示す装置を使用して、以下に示す工程(x1)を実施した。
次に、図24に示す装置を使用して、予備濃縮工程を行った。カルバメート化工程で貯槽a203に回収した反応液を、ラインa31を通して21kg/時間で薄膜蒸発器a301に供給した。薄膜蒸発器a301は加熱蒸発面の温度を130℃、内部の圧力を70kPaとした。
図25に示す装置を使用して、工程(x2)~(x4)を行った。
熱分解装置a401の中段に設けたラインa43よりイソホロンジイソシアネートを含む留分を回収し分離塔a402に供給した。分離塔a402にて1-ブタノールを含む気相成分を蒸留分離し、該気相成分は凝縮器a422を経てラインa44bより回収した。蒸留分離に必要な熱量はリボイラーa412より供給した。
ラインa41aより回収した液相成分を本実施形態の高沸点化合物を含む液相成分として使用した。
図26に示す装置を使用して、以下に示す工程(a1)及び工程(a2)を実施した。
ラインa41aを経て回収した高沸点化合物を含む液相成分50kgとフェノール50kgを180℃に保持した状態でラインb20を通じて耐圧反応器b201に供給した。
耐圧反応器b201に水100kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:35モル倍)と1-アミノ-3-アミノメチル-3,5,5-トリメチルシクロヘキサン50kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:1.9モル倍)と水酸化カルシウム3kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:0.26モル倍)を添加した後、ポンプb241により、ラインb21とラインb22を経由して液相成分を循環させながら250℃、4.1MPaで5時間反応を行った。
後述する工程(e)で、ヒドロキシ化合物を回収した際の残留液を減圧蒸留し、粗1-アミノ-3-アミノメチル-3,5,5-トリメチルシクロヘキサン含有液を回収した。収率は90質量%であった。
工程(b)で回収した粗1-アミノ-3-アミノメチル-3,5,5-トリメチルシクロヘキサン含有液を蒸留精製した。回収した1-アミノ-3-アミノメチル-3,5,5-トリメチルシクロヘキサンは純度が99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、1-アミノ-3-アミノメチル-3,5,5-トリメチルシクロヘキサンの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
前記工程(b)を実施する前に、貯槽b202に回収した反応液を減圧蒸留し、粗1-ブタノール含有液を回収した。収率は80質量%であった。
前記工程(e)で回収した粗1-ブタノール含有液を蒸留精製した。回収した1-ブタノールの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、1-ブタノールの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
1.イソシアネート化合物(VIIa)分離後の液相成分の回収
(1)工程(x1):カルバメート化合物の製造工程
図27に示す装置を使用して、工程(x1)を実施した。
ジベンジルエーテルの代わりにジベンジルトルエンを用い、且つ、実施例3-2の「工程(x2):カルバメート化合物の熱分解工程」と同様の方法を用いて、カルボニル化合物の熱分解反応を行い、高沸点化合物を含む液相成分を得た。
図28に示す装置を使用して、以下に示す工程(a1)及び工程(a2)を実施した。
工程(x2)でラインa41aを経て回収した高沸点化合物を含む液相成分50kgとフェノール25kgを180℃に保持した状態でラインb30を通じて撹拌槽b301に供給した。
撹拌槽B301に水100kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:24モル倍)と水酸化カリウム3kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:0.23モル倍)を添加して250℃、4.9MPaで均一な溶液とした。該溶液を、ラインb33を通じて、250℃に加熱され、4.1MPaに加圧された充填塔b302に連続的に供給した。
後述する工程(e)で、ヒドロキシ化合物を回収した際の残留液を減圧蒸留し、粗4、4’-ジシクロヘキシルメタンジアミン含有液を回収した。収率は88質量%であった。
工程(b)で回収した粗4、4’-ジシクロヘキシルメタンジアミン含有液を蒸留精製した。
貯槽b303に回収した反応液を減圧蒸留し、フェノールを回収した。収率は93質量%であった。
前記工程(e)で回収した粗フェノール含有液を蒸留精製した。回収したフェノールの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、フェノールの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
1.イソシアネート化合物(VIIa)分離後の液相成分の回収
(1)工程(x1):カルバメート化合物の製造工程
図27に示す装置を使用して、工程(x1)を実施した。
図29に示す装置を使用し、工程(x1-a)を実施した。
図30に示す装置を使用して、工程(x2)を実施した。
図31に示す装置を使用して、以下に示す工程(a1)及び工程(a2)を実施した。
前記工程(x2)で回収した高沸点化合物を含む液相成分50kgと4-(α,α-ジメチルベンジル)フェノール50kgを180℃に保持した状態でラインb40を通じて280℃に加熱し、6.6MPaに加圧された押出機b401に供給した。
前記工程(a1)と同時にラインb40を通じて、押出機b401に、水100kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:34モル倍)と炭酸セシウム2kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:0.04モル倍)の割合で連続的に添加した。
回収した反応液を減圧蒸留し、粗1,6-ヘキサメチレンジアミン含有液を回収した。収率は76質量%であった。
工程(b)で回収した粗1,6-ヘキサメチレンジアミン含有液を蒸留精製した。回収した1,6-ヘキサメチレンジアミンの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、1,6-ヘキサメチレンジアミンの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
工程(b)で、1,6-ヘキサメチレンジアミンを回収した後の残留液を減圧蒸留し、粗4-(α,α-ジメチルベンジル)フェノール含有液を回収した。収率は74質量%であった。
工程(e)で回収した粗4-(α,α-ジメチルベンジル)フェノール含有液を蒸留精製した。4-(α,α-ジメチルベンジル)フェノールの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、4-(α,α-ジメチルベンジル)フェノールの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
1.イソシアネート化合物(VIIa)分離後の液相成分の回収
(1)工程(x1):カルバメート化合物の製造工程、及び、工程(x1-b):予備濃縮工程
図23及び図24に示す装置を使用して、工程(x1)及び工程(x1-b)を実施した。
図32に示す装置を使用して、工程(x2)を行った。
(3-1)工程(x3-1):(多段蒸留塔による)軽沸分離工程
図33に示す装置を使用して、工程(x3-1)を実施した。
図34に示す装置を使用して、工程(x3-2)を実施した。
図26に示す装置を使用して、以下に示す工程(a1)及び工程(a2)を実施した。
ラインc33を経て回収した高沸点化合物を含む液相成分50kgとフェノール50kgとを180℃に保持した状態でラインb20を通じて耐圧反応器b201に供給した。
次いで、水100kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:11モル倍)とトリエチルアミン6kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:0.11モル倍)を使用し、240℃、3.4MPaで3時間反応を行った以外は、実施例3-2の「工程(a2):反応工程」と同様の方法を用いて、反応を行い、反応液を得た。
後述する工程(e)で、ヒドロキシ化合物を回収した際の残留液を減圧蒸留し、粗4-アミノメチル-1,8-オクタンジアミン含有液を回収した。収率は89質量%であった。
工程(b)で回収した粗4-アミノメチル-1,8-オクタンジアミン含有液を蒸留精製した。回収した4-アミノメチル-1,8-オクタンジアミンの純度は99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、4-アミノメチル-1,8-オクタンジアミンの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
工程(a2)で得た反応液を減圧蒸留し、粗フェノール含有液を回収した。工程(a1)及び工程(a2)で添加した量を除いて収率は84質量%であった。
工程(e)で回収した粗フェノール含有液を蒸留精製した。フェノールの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、フェノールの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
1.イソシアネート化合物(VIIa)分離後の液相成分の回収
(1)工程(y1):イソシアネート化合物(VIIa)の製造工程
図27及び図32に示す装置を使用し、反応溶媒としてジクロロベンゼンを用いて、1,5-ペンタメチレンジアミンとホスゲンとの反応により1,5-ペンタメチレンジイソシアネートを製造する際に生成する蒸留残渣(溶融物)を得た。これを、高沸点化合物を含有する組成物として使用した。
図35に示す装置を使用して、以下に示す工程(a1)及び工程(a2)を実施した。
工程(y1)で得られた蒸留残渣50kgとフェノール100kgを200℃に保持した状態で混合し、ラインb50を通じ薄膜蒸発器b502に供給した。
工程(a2)は前記工程(a1)と同時に実施した。ラインb50を通じて、水50kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:10モル倍)と酸化亜鉛9kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:0.20モル倍)を薄膜蒸発器b502に供給した。
回収した反応液を減圧蒸留し、粗1,5-ペンタメチレンジアミン含有液を回収した。収率は68質量%であった。
工程(6-3)で回収した粗1,5-ペンタメチレンジアミン含有液を蒸留精製した。回収した1,5-ペンタメチレンジアミンの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、1,5-ペンタメチレンジアミンの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
1.イソシアネート化合物(VIIa)分離後の液相成分の回収
(1)工程(x1):カルバメート化合物の製造工程
図23に示す装置を使用し、4,4’-メチレンジアニリン12.7kg、尿素15.1kg、及び4-(α,α-ジメチルベンジル)フェノール253.3kgを使用した以外は、実施例3-2の「工程(x1):カルバメート化合物の製造工程」と同様の方法を用いて、カルバメート化合物の製造を行い、下記式(Vc-4)で表されるカルバメート化合物を含む反応液を得た。
図36に示す装置を使用して、工程(x2)を行った。
(3-1)工程(x3-1):(多段蒸留塔による)軽沸分離工程
図33に示す装置を使用し、実施例3-5の「工程(x3-1):(多段蒸留塔による)軽沸分離工程」と同様の方法を用いて、多段蒸留塔による軽沸分離を行った。
図34に示す装置を使用し、実施例3-5の「工程(x3-2):高沸分離工程(薄膜蒸発器による精製工程)」と同様の方法を用いて、高沸分離を行った。
図26に示す装置を使用して、以下に示す工程(a1)及び工程(a2)(工程(a2-1)及び工程(a2-2))を実施した。
ラインc33を経て回収した高沸点化合物を含む液相成分50kgと4-(α,α-ジメチルベンジル)フェノール50kgを、ラインb20を通じて撹拌槽b201へ導入し180℃で0.5時間反応を行い、反応液を得た。
(2-1)工程(a2-1):組成物-アミン化合物-触媒混合工程
ラインb20を通じて4,4’-メチレンジアニリン200kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:3.1モル倍)と水酸化ナトリウム9kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:0.68モル倍)をB201へ導入した。供給された混合液は、240℃で1時間反応を行い、反応液を得た。
工程(a2-1)で混合された組成物300kgと水200kg(基(IX-1)~(IX-12)に含まれる窒素原子の合計モル量に対する比率:34モル倍)を使用し、190℃、1.3MPaで1.5時間反応を行った以外は、実施例3-2の「工程(a2):反応工程」と同様の方法を用いて、工程(a2-1)で得られた組成物と水の反応を行い、反応液を得た。
後述する工程(e)で、ヒドロキシ化合物を回収した際の残留液を減圧蒸留し、粗4,4’-メチレンジアニリン含有液を回収した。収率は77質量%であった。
工程(b)で回収した粗4,4’-メチレンジアニリン含有液を蒸留精製した。回収した4,4’-メチレンジアニリンの純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、4,4’-メチレンジアニリンの総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
工程(a2-2)で得た反応液を減圧蒸留し、粗4-(α,α-ジメチルベンジル)フェノール含有液を回収した。工程(a1)で添加した量を除いて収率は82質量%であった。
工程(e)で回収した粗4-(α,α-ジメチルベンジル)フェノール含有液を蒸留精製した。4-(α,α-ジメチルベンジル)の純度は、99質量%以上(ガスクロマトグラフィーの分析誤差を含む)であった。また、4-(α,α-ジメチルベンジル)の総質量に対する金属原子含有量は1000質量ppm未満、ハロゲン原子含有量は1000質量ppm未満であった。
以下の各表に示す条件にて、イソシアネート化合物(VIIa)の製造で副生する高沸点化合物を含む液相成分を得た後、該液相成分を用いて、アミン化合物(IIa)、及びヒドロキシ化合物を回収した。液相成分の組成及び回収後の結果についても、以下の各表に示す。
IIa-1:2,4-トルエンジアミン
IIa-2:4,4’-ジシクロヘキシルメタンジアミン
IIa-3:4-アミノメチル-1,8-オクタンジアミン
IIa-4:1,6-ヘキサメチレンジアミン
IIa-5:1-アミノ-3-アミノメチル-3,5,5-トリメチルシクロヘキサン
IIa-6:4,4’,4’’-メタントリアニリン
IIa-7:L-リジンアミノエチルエステル
IIa-8:ベンゼン-1,3,5-トリアミン
IIa-9:シクロヘキシル-1,3-5-トリアミン
IIa-10:4,4’-メチレンジアニリン
VIc-1-1a:4-(1,1,3,3-テトラメチルブチル)フェノール
VIc-1-1b:エチルフェノール
VIc-1-1c:フェノール
VIc-1-1d:4-(α,α-ジメチルベンジル)フェノール
VIc-1-1e:トリベンジルフェノール
VIc-1-1f:クレゾール
VIc-1-1g:p-ドデシルフェノール
VIc-1-1h:3-(3,4,6-トリベンジルフェニル)フェノール
VIc-2-1:エタノール
VIc-2-2:1-ブタノール
実施例3-1の工程(a2)に相当する工程で触媒を使用しなかった以外は、実施例3-1と同様の方法で行い、粗2,4-トルエンジアミン含有液を回収した。その収率は40質量%であった。また、粗4-(1,1,3,3-テトラメチルブチル)フェノール含有液を回収した。その収率は39質量%であった。前記成分を蒸留分離した後の蒸留塔内には固着物が発生していた。
実施例3-24の工程(a2)に相当する工程で触媒を使用しなかった以外は、実施例3-24と同様の方法で行った。反応初期に反応器b201内に閉塞が生じ、反応物を払い出すことができなかった。反応を継続し、さらに5時間の反応を実施したところ、閉塞は解消したがスラリー状の液層成分として反応液を回収した。粗1,6-ヘキサメチレンジアミン含有液を回収した。その収率は14質量%であった。また、粗1-ブタノール含有液を回収した。その収率は31質量%であった。
実施例3-13の工程(a2)に相当する工程で触媒を使用しなかった以外は、実施例3-13と同様の方法で行った。反応初期に攪拌槽b301及び充填塔b302内に閉塞が生じ、反応物を払い出すことができなかった。反応を継続し、さらに5時間の反応を実施したところ、閉塞が解消し液層成分として反応液を回収した。粗1,6-ヘキサメチレンジアミン含有液を回収した。その収率は27質量%であった。また、粗4-(α,α-ジメチルベンジル)フェノール含有液を回収した。その収率は47質量%であった。
実施例3-8の工程(a1)に相当する工程で芳香族ヒドロキシ化合物を使用しなかった以外は、実施例8と同様の方法を行ったが、反応器b201内に閉塞が生じ、反応物を払い出すことができなかった。
実施例3-5の工程(a2)に相当する工程で水を使用しなかった以外は、実施例3-5と同様の方法を行った。粗4-アミノメチル-1,8-オクタンジアミン含有液及び粗フェノール含有液を蒸留塔にて分離することを試みたが、蒸留塔内には固着物が発生し、払い出すことができなかったため運転を中断した。
実施例3-2の工程(a1)に相当する工程において、芳香族ヒドロキシ化合物の代わりに、1-ブタノールを使用した以外は、実施例3-2と同様の方法を行った。
実施例3-7の工程(a1)に相当する工程を実施せず、芳香族ヒドロキシ化合物を用いなかった以外は、実施例3-7と同様の方法を行った。粗4,4’-メチレンジアニリン含有液及び粗4-(α,α-ジメチルベンジル)フェノール含有液を蒸留塔にて分離することを試みたが、蒸留塔内には固着物が発生し、払い出すことができなかったため運転を中断した。一部回収できた4,4’-メチレンジアニリンの収率は38質量%であった。また、回収できた4-(α,α-ジメチルベンジル)フェノールの収率は67質量%であった。
実施例3-10の工程(a2-1)に相当する工程において、触媒を用いなかった以外は実施例10と同様の方法をおこない、粗4-アミノメチル-1,8-オクタンジアミン含有液を回収した。その収率は42質量%であった。また、粗エチルフェノール含有液を回収した。その収率は54質量%であった。
実施例3-11の工程(a1)に相当する工程において、芳香族ヒドロキシ化合物の代わりに、2-ブタノールを使用した以外は、実施例3-11と同様の方法を行った。粗2,4-トルエンジアミン含有液及び粗4-(1,1,3,3-テトラメチルブチル)フェノール含有液を蒸留塔にて分離することを試みたが、蒸留塔内には固着物が発生し、払い出すことができなかったため運転を中断した。一部回収できた2,4-トルエンジアミンの収率は62質量%であった。また、回収できた4-(1,1,3,3-テトラメチルブチル)フェノールの収率は37質量%であった。
実施例3-10の工程(a2-1)に相当する工程において、活性水素化合物を用いなかった以外は、実施例3-10と同様の方法を行い、粗4-アミノメチル-1,8-オクタンジアミン含有液を回収した。その収率は67質量%であった。また、粗エチルフェノール含有液を回収した。その収率は68質量%であった。
<分析方法>
(1)1H-NMR分析方法
装置として、日本電子(株)社製JNM-A400 FT-NMRシステムを用いて、1H-NMR分析を実施した。
サンプル溶液を0.3g秤量し、重クロロホルムを0.7gと内部標準物質としてジメチルジフェニルシラン0.05gを加えて均一に混合した溶液をNMR分析サンプルとした。
各標準物質について分析を実施し、作成した検量線を基に、分析サンプル溶液の定量分析を実施した。
以下の条件で分析を行った。
装置:島津製作所社製、GC-2010
カラム:DB-1
直径0.25mm、長さ30m、膜厚1.0μm
カラム温度:60℃~300℃
注入口温度:300℃
キャリアガス:ヘリウム
キャリアガス流量:40mL/min
検出器:FID(水素炎イオン化型検出器)
サンプル溶液を1.0g秤量し、アセトニトリルを10gと内部標準物質としてアニソールを0.1g加えて均一に混合した溶液をガスクロマトグラフィー分析サンプルとした。
以下の条件で分析を行った。
装置:島津製作所社製、LC-10AT
カラム:Inertsil ODS
粒子径5μm、内径2.1mm、長さ250mm
カラム温度:40℃
展開溶媒:水/アセトニトリル=90/10
展開溶媒流量:1mL/min
検出器:フォトダイオードアレイ検出器
サンプル溶液を1.0g秤量し、酢酸10gを加えて均一に混合した溶液を液体クロマトグラフィー分析サンプルとした。
各標準物質について分析を実施し、作成した検量線を基に、分析サンプル溶液の定量分析を実施した。
工程(1-1):カルバメート化合物の製造
図37に示す装置を使用して反応を行った。なお、実施例4-1以降のカルバメート化合物の製造においても図37に示す装置を使用した。
図38に示す装置を使用して反応を行った。なお、実施例4-1以降のカルバメート化合物の熱分解においても図38に示す装置を使用した。
図39に示す装置を使用して反応を行った。なお、実施例4-1以降の軽沸分離においても図39に示す装置を使用した。
図40に示す装置を使用して反応を行った。なお、実施例4-1以降の高沸分離においても図40に示す装置を使用した。
工程(2-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを19.4kg使用し、反応液(1-1)を約13分かけて反応器に供給して反応を開始し、ジャケット温度248℃、内温240℃、還流比0.4、圧力を14から26kPaの範囲で反応を行い、反応液(1-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は20.96kgであった。この反応液(以下、「反応液(2-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率78質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は4.3であった。また、反応液(2-2)の総質量に対して、DPCの濃度は68質量%であり、カルボニル化合物(I)の濃度は18質量%であった。
反応液(2-2)を4.19kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.26kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(2-3)」又は、「イソシアネート組成物(2-3)」と称する)は6.29kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(2-2)に対してTTIが収率77質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は6.3であった。また、反応液(2-3)の総質量に対して、DPCの濃度は550質量ppmであり、カルボニル化合物(I)の濃度は34質量%であった。
工程(2-3)で貯槽303に回収した液を使用したこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は3.36kgであり、TTIの回収率は116質量%であった。
工程(3-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを19.4kg使用し、反応液(1-1)を約9分かけて反応器に供給して反応を開始し、ジャケット温度258℃、内温250℃、還流比2.0、圧力を21から32kPaの範囲で反応を行い、反応液(1-1)を全て移送後2.5時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は22.12kgであった。この反応液(以下、「反応液(3-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率73質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は3.9であった。また、反応液(3-2)の総質量に対して、DPCの濃度は67質量%であり、カルボニル化合物(I)の濃度は13質量%であった。
反応液(3-2)を4.42kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.42kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(3-3)」、又は、「イソシアネート組成物(3-3)」と称する)は7.08kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(3-2)に対してTTIが収率80質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は5.5であった。また、反応液(3-3)の総質量に対して、DPCの濃度は320質量ppmであり、カルボニル化合物(I)の濃度は40質量%であった。
工程(3-3)で貯槽303に回収した液を使用したこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は3.35kgであり、TTIの回収率は119質量%であった。
工程(4-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを19.4kg使用し、反応液(1-1)を約14分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比1.5、圧力を8から14kPaの範囲で反応を行い、反応液(1-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の重量は22.12kgであった。この反応液(以下、「反応液(4-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率79質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は1.6であった。また、反応液(4-2)の総質量に対して、DPCの濃度は61質量%であり、カルボニル化合物(I)の濃度は19質量%であった。
反応液(4-2)を4.42kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.68kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(4-3)」、又は、「イソシアネート組成物(4-3)」と称する)は8.41kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(4-2)に対してTTIが収率85質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は2.6であった。また、反応液(4-3)の総質量に対して、DPCの濃度は730質量ppmであり、カルボニル化合物(I)の濃度は50質量%であった。
工程(4-3)で貯槽303に回収した液を使用したこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は4.05kgであり、TTIの回収率は125質量%であった。
工程(5-1):カルバメート化合物の製造
炭酸ジフェニルの代わりに尿素0.23kg(69.2mol)を使用し、貯槽102に供給したフェノールが25.87kg(275.2mol)、反応器104の反応温度を240℃にして30分攪拌したこと以外は実施例4-1と同様の操作でカルバメート合成を行った。貯槽107に抜き出したフェノール、及びアンモニア量は24.14kgであった。
反応液(5-1)を10.8kg、炭酸ジフェニルを28.0kg使用し、反応液(1-1)を約13分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比1.7、圧力を8から14kPaの範囲で反応を行い、反応液(5-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は21.34kgであった。この反応液(以下、「反応液(5-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率78質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は1.5であった。また、反応液(5-2)の総質量に対して、DPCの濃度は58質量%であり、カルボニル化合物(I)の濃度は21質量%であった。
反応液(5-2)を4.27kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.75kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(5-3)」、又は、「イソシアネート組成物(5-3)」と称する)は8.75kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(5-2)に対してTTIが収率84質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は2.5であった。また、反応液(5-3)の総質量に対して、DPCの濃度は510質量ppmであり、カルボニル化合物(I)の濃度は52質量%であった。
工程(5-3)で貯槽303に回収した液を使用したこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は4.05kgであり、TTIの回収率は128質量%であった。
工程(1’-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを5.45kg使用し、反応液(1-1)を約14分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比1.5、圧力を5から15kPaの範囲で反応を行い、反応液(1-1)を全て移送後1.5時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は5.71kgであった。この反応液(以下、「反応液(1’-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率16質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は64.5であった。また、反応液(1’-2)の総質量に対して、DPCの濃度は5質量%であり、カルボニル化合物(I)の濃度は12質量%であった。
反応液(1’-2)を1.14kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.09kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行ったところ運転途中で液が高粘度化し、運転継続が困難であった。
工程(2’-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを10.89kg使用し、反応液(1-1)を約13分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比0、圧力を11から15kPaの範囲で反応を行い、反応液(1-1)を全て移送後4時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は9.39kgであった。この反応液(以下、「反応液(2’-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率20質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は17.7であった。また、反応液(2’-2)の総質量に対して、DPCの濃度は27質量%であり、カルボニル化合物(I)の濃度は23質量%であった。
反応液(2’-2)を1.88kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.41kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行ったところ運転途中で液が高粘度化し、運転継続が困難であった。
工程(3’-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを10.89kg使用し、反応液(1-1)を約11分かけて反応器に供給して反応を開始し、ジャケット温度248℃、内温240℃、還流比2.5、圧力を30から32kPaの範囲で反応を行い、反応液(1-1)を全て移送後4時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は20.0kgであった。この反応液(以下、「反応液(3’-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率27質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は12.0であった。また、反応液(3’-2)の総質量に対して、DPCの濃度は63質量%であり、カルボニル化合物(I)の濃度は14質量%であった。
反応液(3’-2)を3.99kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.64kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行ったところ運転途中で液が高粘度化し、運転継続が困難であった。
工程(4’-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを19.4kg使用し、反応液(1-1)を約15分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比0、圧力を14から18kPaの範囲で反応を行い、反応液(1-1)を全て移送後4時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は18.2kgであった。この反応液(以下、「反応液(4’-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率59質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は12.8であった。また、反応液(4’-2)の総質量に対して、DPCの濃度は66質量%であり、カルボニル化合物(I)の濃度は8質量%であった。
反応液(4’-2)を3.65kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.46kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行ったところ運転途中で液が高粘度化し、運転継続が困難であった。
工程(5’-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを10.89kg使用し、反応液(1-1)を約10分かけて反応器に供給して反応を開始し、ジャケット温度258℃、内温250℃、還流比0.5、圧力を18から32kPaの範囲で反応を行い、反応液(1-1)を全て移送後2.5時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は16.1kgであった。この反応液(以下、「反応液(5’-2)」と称する)をNMR、LC、ガスクロマトグラフィーで分析した結果、TTIが収率71質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は6.4であった。また、反応液(5’-2)の総質量に対して、DPCの濃度は59質量%であり、カルボニル化合物(I)の濃度は13質量%であった。
反応液(5’-2)を3.21kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.22kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液は6.10kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(5’-2)に対してTTIが収率74質量%で回収されており、この時の反応液(以下、「反応液(5’-3)」と称する場合がある)の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は8.2であった。また、反応液(5’-3)の総質量に対して、DPCの濃度は850質量ppmであり、カルボニル化合物(I)の濃度は33質量%であった。
工程(5’-3)で貯槽303に回収した液を使用したこと以外は実施例4-1と同様の操作で高沸分離を行ったが、残渣が高粘度化し運転継続が困難であった。
工程(6’-2):カルバメート化合物の熱分解工程
実施例4-1の反応液(1-1)を19.4kg、炭酸ジフェニルを19.4kg使用し、反応液(1-1)を約12分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比0.2、圧力を11から16kPaの範囲で反応を行い、反応液(1-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は17.5kgであった。この反応液(以下、「反応液(6’-2)」と称する)をNMR、LC、及びガスクロマトグラフィーで分析した結果、TTIが収率79質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は5.5であった。また、反応液(6’-2)の総質量に対して、DPCの濃度は64質量%であり、カルボニル化合物(I)の濃度は9質量%であった。
反応液(6’-2)を3.49kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.15kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液は5.76kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(6’-2)に対してTTIが収率74質量%で回収されており、この時の反応液(以下、「反応液(6’-3)」と称する場合がある)の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は8.3であった。また、反応液(6’-3)の総質量に対して、DPCの濃度は920質量ppmであり、カルボニル化合物(I)の濃度は29質量%であった。
工程(6’-3)で貯槽303に回収した液を使用したこと以外は実施例4-1と同様の操作で高沸分離を行ったが、残渣が高粘度化し運転継続が困難であった。
工程(6-1):カルバメート化合物の製造
ライン14を閉止した状態で、4,4’-ジアミノジフェニルメタン3.33kg(16.8mol)を貯槽101aよりライン11aを経て、バッフル付きSUS製反応容器104aに供給し、フェノール2.53kg(27.0mol)を貯槽102aよりライン12aを経て、上記反応容器104aに供給し、攪拌することで均一化した。次に、ライン16を閉止した状態で、フェノール2.53kg(27.0mol)を貯槽102aよりライン15aを経て、バッフル付きSUS製反応容器105aに供給し、炭酸ジフェニル11.96kg(55.6mol)を貯槽103aよりライン13aを経て、上記反応容器105aに供給した。上記反応容器105aの液温を65℃に調整し、攪拌することで均一化した後、反応容器104aよりライン14aを経て、4,4’-ジアミノジフェニルメタンとフェノールの混合液を内温が70℃を超えないように供給した。2時間攪拌を継続した後、反応液を120℃まで昇温し、内部の圧力を約1kPaにすることで、液中のフェノール7.58kgをライン17及びコンデンサー(凝縮器)X11を通じて貯槽107に抜き出した。
反応液(6-1)を12.77kg、炭酸ジフェニルを12.77kg使用し、反応液(6-1)を約10分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比0.8、圧力を11から16kPaの範囲で反応を行い、反応液(6-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は11.75kgであった。この反応液(以下、「反応液(6-2)」と称する)を、NMR、及びガスクロマトグラフィーで分析した結果、ジフェニルメタンジイソシアネート(MDI)が収率70質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は1.3であった。また、反応液(6-2)の総質量に対して、DPCの濃度は15質量%であり、カルボニル化合物(I)の濃度は38質量%であった。
反応液(6-2)を2.35kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.97kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(6-3)」、又は、「イソシアネート組成物(6-3)」と称する)は9.87kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(6-2)に対してMDIが収率79質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は2.3であった。また、反応液(6-3)の総質量に対して、DPCの濃度は420質量ppmであり、カルボニル化合物(I)の濃度は45質量%であった。
工程(6-3)で貯槽303に回収した液を使用し、運転温度170℃、内部の圧力を0.3kPaとしたこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は4.54kgであり、MDIの回収率は130質量%であった。
工程(7-1):カルバメート化合物の製造
ライン14aを閉止した状態で、リジンβ-アミノエチルエステル3塩酸塩3.33kg(16.8mol)を貯槽101aよりライン11aを経て、バッフル付きSUS製反応容器104aに供給し、フェノール2.52kg(26.7mol)を貯槽102aよりライン12aを経て、上記反応容器104aに供給し、攪拌することで均一化した。次に、ライン16を閉止した状態で、フェノール2.52kg(26.7mol)を貯槽102aよりライン15aを経てバッフル付きSUS製反応容器105aに供給し、炭酸ジフェニル11.91kg(55.6mol)を貯槽103aよりライン13aを経て上記反応容器105aに供給した。上記反応容器105aの液温を65℃に調整し、攪拌することで均一化した後、反応容器104aよりライン14aを経てリジンβ-アミノエチルエステル3塩酸塩とフェノールの混合液を内温が70℃を超えないように供給した。2時間攪拌を継続した後、反応液を120℃まで昇温し、内部の圧力を約1kPaにすることで、液中のフェノール8.22kgをライン17及びコンデンサー(凝縮器)X11を通じて貯槽107に抜き出した。
反応液(7-1)を11.45kg、炭酸ジフェニルを10.00kg使用し、反応液(7-1)を約15分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比3.2、圧力を11から16kPaの範囲で反応を行い、反応液(7-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は9.01kgであった。この反応液(以下、「反応液(7-2)」と称する)を、NMR、及びガスクロマトグラフィーで分析した結果、リジントリイソシアネート(LTI)が収率77質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は1.4であった。また、反応液(7-2)の総質量に対して、DPCの濃度は38質量%であり、カルボニル化合物(I)の濃度は32質量%であった。
反応液(7-2)を1.80kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.10kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(7-3)」、又は、「イソシアネート組成物(7-3)」と称する)は5.50kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(7-2)に対してLTIが収率83質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は2.4であった。また、反応液(7-3)の総質量に対して、DPCの濃度は120質量ppmであり、カルボニル化合物(I)の濃度は53質量%であった。
工程(7-3)で貯槽303に回収した液を使用し、運転温度190℃、内部の圧力を0.1kPaとしたこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は2.38kgであり、LTIの回収率は125質量%であった。
工程(8-1):カルバメート化合物の製造
ライン14aを閉止した状態で、リジンメチルエステル二塩酸塩3.33kg(14.3mol)を貯槽101aよりライン11aを経て、バッフル付きSUS製反応容器104aに供給し、フェノール1.91kg(20.3mol)を貯槽102aよりライン12aを経て上記反応容器104aに供給し、攪拌することで均一化した。次に、ライン16を閉止した状態で、フェノール1.91kg(20.3mol)を貯槽102aよりライン15aを経て、バッフル付きSUS製反応容器105aに供給し、炭酸ジフェニル10.17kg(47.5mol)を貯槽103aよりライン13aを経て上記反応容器105aに供給した。上記反応容器105aの液温を65℃に調整し、攪拌することで均一化した後、反応容器104aよりライン14aを経て、リジンメチルエステル二塩酸塩とフェノールの混合液を内温が70℃を超えないように供給した。2時間攪拌を継続した後、反応液を120℃まで昇温し、内部の圧力を約1kPaにすることで、液中のフェノール5.93kgをライン17及びコンデンサー(凝縮器)X11を通じて貯槽107に抜き出した。
反応液(8-1)を11.38kg、炭酸ジフェニルを11.38kg使用し、反応液(8-1)を約12分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比0.9、圧力を20から29kPaの範囲で反応を行い、反応液(8-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は19.35kgであった。この反応液(以下、「反応液(8-2)」と称する)を、NMR、ガスクロマトグラフィーで分析した結果、リジンジイソシアネート(LDI)が収率81質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は1.4であった。また、反応液(8-2)の総質量に対して、DPCの濃度は61質量%であり、カルボニル化合物(I)の濃度は13質量%であった。
反応液(8-2)を3.87kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.47kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(8-3)」、又は、「イソシアネート組成物(8-3)」と称する)は7.35kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(8-2)に対してLDIが収率86質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は2.5であった。また、反応液(8-3)の総質量に対して、DPCの濃度は630質量ppmであり、カルボニル化合物(I)の濃度は35質量%であった。
工程(8-3)で貯槽303に回収した液を使用し、運転温度180℃、内部の圧力を0.1kPaとしたこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は4.57kgであり、LDIの回収率は122質量%であった。
工程(9-1):カルバメート化合物の製造
ライン14aを閉止した状態で、4,4’-メチレンビス(シクロヘキシルアミン)3.33kg(15.8mol)を貯槽101aよりライン11aを経て、バッフル付きSUS製反応容器104aに供給し、フェノール2.29kg(24.4mol)を貯槽102aよりライン12aを経て、上記反応容器104aに供給し、攪拌することで均一化した。次に、ライン16を閉止した状態で、フェノール2.29kg(24.4mol)を貯槽102aよりライン15aを経て、バッフル付きSUS製反応容器105aに供給し、炭酸ジフェニル11.27kg(52.7mol)を貯槽103aよりライン13aを経て、上記反応容器105aに供給した。上記反応容器105aの液温を65℃に調整し、攪拌することで均一化した後、反応容器104aよりライン14aを経て、4,4’-メチレンビス(シクロヘキシルアミン)とフェノールの混合液を内温が70℃を超えないように供給した。2時間攪拌を継続した後、反応液を120℃まで昇温し、内部の圧力を約1kPaにすることで、液中のフェノール6.94kgをライン17及びコンデンサー(凝縮器)X11を通じて貯槽107に抜き出した。
反応液(9-1)を12.24kg、炭酸ジフェニルを12.24kg使用し、反応液(9-1)を約10分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比4.5、圧力を11から16kPaの範囲で反応を行い、反応液(9-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は9.79kgであった。この反応液(以下、「反応液(9-2)」と称する)を、NMR、LC、及びガスクロマトグラフィーで分析した結果、メチレンビス(シクロヘキシルイソシアネート)(HMDI)が収率76質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は2.0であった。また、反応液(9-2)の総質量に対して、DPCの濃度は15質量%であり、カルボニル化合物(I)の濃度は27質量%であった。
反応液(9-2)を1.96kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.65kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(9-3)」、又は、「イソシアネート組成物(9-3)」と称する)は8.22kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(9-2)に対してHMDIが収率82質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は3.4であった。また、反応液(9-3)の総質量に対して、DPCの濃度は200質量ppmであり、カルボニル化合物(I)の濃度は32質量%であった。
工程(9-3)で貯槽303に回収した液を使用し、運転温度190℃、内部の圧力を0.3kPaとしたこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は4.85kgであり、HMDIの回収率は125質量%であった。
工程(10-1):カルバメート化合物の製造
ライン14aを閉止した状態で、1,3-ジ(アミノメチル)シクロヘキサン3.33kg(23.1mol)を貯槽101aよりライン11aを経て、バッフル付きSUS製反応容器104aに供給し、フェノール4.11kg(43.7mol)を貯槽102aよりライン12aを経て、上記反応容器104aに供給し、攪拌することで均一化した。次にライン16を閉止した状態で、フェノール4.11kg(43.7mol)を貯槽102aよりライン15aを経て、バッフル付きSUS製反応容器105aに供給し、炭酸ジフェニル16.44kg(76.8mol)を貯槽103aよりライン13aを経て、上記反応容器105aに供給した。上記反応容器105aの液温を65℃に調整し、攪拌することで均一化した後、反応容器104aよりライン14aを経て、1,3-ジ(アミノメチル)シクロヘキサンとフェノールの混合液を内温が70℃を超えないように供給した。2時間攪拌を継続した後、反応液を120℃まで昇温し、内部の圧力を約1kPaにすることで、液中のフェノール11.74kgをライン17及びコンデンサー(凝縮器)X11を通じて貯槽107に抜き出した。
反応液(10-1)を16.24kg、炭酸ジフェニルを16.24kg使用し、反応液(10-1)を約15分かけて反応器に供給して反応を開始し、ジャケット温度238℃、内温230℃、還流比4.0、圧力を11から16kPaの範囲で反応を行い、反応液(10-1)を全て移送後3時間フェノールの抜き出しを継続したこと以外は、実施例4-1と同様の操作で熱分解反応を行った。貯槽205に移送した反応液の質量は13.97kgであった。この反応液(以下、「反応液(10-2)」と称する)を、NMR、LC、及びガスクロマトグラフィーで分析した結果、1,3-ビス(イソシアナトメチル)シクロヘキサン(HXDI)が収率78質量%で生成していた。この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は1.7であった。また、反応液(10-2)の総質量に対して、DPCの濃度は31質量%であり、カルボニル化合物(I)の濃度は25質量%であった。
反応液(10-2)を2.79kg/時間で連続的にフィードし、ライン34の定常状態における抜き出し速度が1.90kg/時間であること以外は、実施例4-1と同様の操作で軽沸分離を行った。貯槽303に回収した液(以下、「反応液(10-3)」、又は、「イソシアネート組成物(10-3)」と称する)は9.50kgであり、NMR、LC、及びガスクロマトグラフィーで分析した結果、供給した反応液(10-2)に対してHXDIが収率77質量%で回収されており、この時の反応液の{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値は3.6であった。また、反応液(10-3)の総質量に対して、DPCの濃度は450質量ppmであり、カルボニル化合物(I)の濃度は37質量%であった。
工程(10-3)で貯槽303に回収した液を使用し、運転温度190℃、内部の圧力を0.3kPaとしたこと以外は実施例4-1と同様の操作で高沸分離を行った。貯槽402に回収した液は4.93kgであり、HXDIの回収率は122質量%であった。
A101,A102,A204,A304,B104,C104,C104,E101,F104:槽
A201,B101,B202,B204,C101,E102:蒸留塔
A202,A302,B102,B203,C102:圧力計
A203,A404,A603,A703,A803,B103,B204,B304,B306,B404,B406,B504,B506,B604,B606,C103,E204,F103:凝縮器
A205,A303,A405,A406,A407,A502,A604,A605,A702,A703,A804,A805,B105,B309,C105,E206,F105:貯槽
A301:薄膜蒸発器
E201,E202,F101:反応器
E203,F102:減圧蒸留器
E205:保存弁
A401,A501:攪拌槽
A402:気液分離器
A403,A601:多段蒸留塔
A701:予備濃縮器
A801:連続多段蒸留塔
B201:管型熱分解反応器
B301,B302,B401,B402,B601,B602:フォーリングフィルム型蒸発器
B303,B307,B403,B407,B502,B603,B607:分離塔
A408,A602,A802,B305,B308,B405,B408,B505,B507,B605,B608:リボイラー
B501:熱分解装置
B503:精製塔
D101:蒸発機側面部
D102:蒸発機
D103:蒸発機底部
D104:蒸発機内部(凝縮部)
101:蒸留塔
102:リボイラー
103:凝縮器
104:貯槽
105:貯槽
a101:連続多段蒸留塔
a102,a401,a701,a801:熱分解装置
a103,a402,a702:分離塔
a104,a403,a703:精製塔
a111,a112,a113,a211,a411,a412,a413,a611,a711,a712,a811,c211:リボイラー
a121,a122,a123,a221,a321,a421,a422,a423,a621,a721,a722,a821,a921,b121,b221,b321,b421,b521,c221,c321:凝縮器
a202,a203,a302,a303,a502,a602,a603,a802,a803,a902,a903,b102,b202,b303,b402,b504,c102,c103,c202,c203:貯槽
a301,a901,b502,c302:薄膜蒸発器
a501,b301:攪拌槽
b101,b201:耐圧反応器
b131,b231,b331,b431,b432,b531:保存弁
b241:ポンプ
b302:充填塔
b401:押出機
b451:ベント口
c101:ジャケットが付帯した貯槽
c201:分離塔
b501,c301:加熱蒸発面
b503,c303:回収部
X11,X21,X31,X41:コンデンサー
X32:リボイラー
101a,102a,103a,106,107,202,204,205,302,303,402,403:貯槽
104a,105a,201:バッフル付きSUS製反応容器
203,301:多段蒸留塔
401:薄膜蒸留装置
Claims (55)
- 第一級アミン化合物と、炭酸誘導体と、を反応させて、副生する、N-置換カルバメート化合物よりも沸点の低い化合物を抜き出しながら、N-置換カルバメート化合物を得る工程(1)と、
前記工程(1)で得られた前記N-置換カルバメート化合物を含む反応液を用いて、非プロトン性溶媒存在下で、前記N-置換カルバメート化合物の熱分解を行い、副生するヒドロキシ化合物を抜き出しながら、イソシアネート化合物を得る工程(2)と、
前記工程(2)で得られた前記イソシアネート化合物を含む反応液から前記イソシアネート化合物と、前記非プロトン性溶媒と、を分離する工程(3)と、
前記工程(3)で得られた前記イソシアネート化合物を含む画分から前記イソシアネート化合物よりも沸点が高い成分を除去して、イソシアネート化合物を精製する工程(4)と、
を含む、イソシアネート化合物の製造方法。 - 前記工程(3)で分離された前記非プロトン性溶媒を含む画分、又は、前記工程(4)で除去された前記イソシアネート化合物よりも沸点が高い成分を、アルカリと水との共存下で、加水分解し、前記第一級アミン化合物及び前記ヒドロキシ化合物を得る工程(5)を更に含む、請求項1に記載のイソシアネート化合物の製造方法。
- 前記工程(1)において、
ヒドロキシ化合物存在下で、第一級アミン化合物と、前記炭酸誘導体として尿素又は尿素誘導体と、を反応させて、副生する、N-置換カルバメート化合物よりも沸点の低い化合物を抜き出しながら、N-置換カルバメート化合物を得る、又は、
第一級アミン化合物と、前記炭酸誘導体として炭酸エステルと、を反応させて、副生する、N-置換カルバメート化合物よりも沸点の低い化合物としてヒドロキシ化合物を抜き出しながら、N-置換カルバメート化合物を得る、請求項1又は2に記載のイソシアネート化合物の製造方法。 - 前記工程(2)において、副生するヒドロキシ化合物を気相に抜き出しながら、前記イソシアネート化合物を含む液相を得る、請求項1又は2に記載のイソシアネート化合物の製造方法。
- 前記工程(1)及び前記工程(2)を、槽型反応器、蒸留塔、管型蒸発器、薄膜蒸発器、及びフォーリングフィルム型蒸発器からなる群より選ばれる1種以上の反応器を用いて行う、請求項1又は2に記載のイソシアネート化合物の製造方法。
- 前記工程(1)及び前記工程(2)のうち、少なくともいずれか一方を反応蒸留法により行う、請求項1又は2に記載のイソシアネート化合物の製造方法。
- 前記工程(2)で抜き出された前記ヒドロキシ化合物を前記工程(1)に循環して再利用する、請求項1又は2に記載のイソシアネート化合物の製造方法。
- 前記工程(3)で分離された前記非プロトン性溶媒を前記工程(2)に循環して再利用する、請求項1又は2に記載のイソシアネート化合物の製造方法。
- 前記非プロトン性溶媒が炭酸エステルである、請求項1又は2に記載のイソシアネート化合物の製造方法。
- 前記n21が3以上である、請求項11に記載のイソシアネート化合物の製造方法。
- 触媒として、活性水素を有さないアミン化合物存在下で、第1級アミン化合物と、炭酸誘導体と、ヒドロキシ化合物と、を反応させて、カルバメート化合物を得る反応工程を含む、カルバメート化合物の製造方法。
- 前記活性水素を有さないアミン化合物が、第三級アミノ基、含窒素芳香族基、アミジン基、及びグアニジン基からなる群より選ばれる1種以上の官能基を有する、請求項13に記載のカルバメート化合物の製造方法。
- 前記活性水素を有さないアミン化合物が、第三級アミノ基を1以上有し、且つ、炭素数が3以上85以下の化合物である、請求項14に記載のカルバメート化合物の製造方法。
- 前記活性水素を有さないアミン化合物が、第三級アミノ基を1以上6以下有し、且つ、炭素数が3以上30以下の化合物である、請求項15に記載のカルバメート化合物の製造方法。
- 前記活性水素を有さないアミン化合物が、N,N’-ジメチルアニリン、N,N’-ジエチルアニリン、N-メチル-N’-エチルアニリン、N,N’-ジメチルアミノピリジン、N,N,N’,N’-テトラメチルフェニレンジアミン、メチレンビス(N,N’-ジメチルアニリン)、トリエチルアミン、エチルジイソプロピルアミン、N-メチルモルホリン、N-メチルピペリジン、キヌクリジン、N,N’-ジメチルピペラジン、トリエチレンジアミン、N,N,N’ N’-テトラメチルエチレンジアミン、N,N,N’ N’-テトラメチルヘキサンジアミン、N,N,N’ N’-テトラメチルキシリレンジアミン、ペンタメチルジエチレントリアミン、ビス(2-モルホリノエチル)エーテル、ヘキサヒドロ-1,3,5-トリス(3-ジメチルアミノプロピル)-1,3,5-トリアジン、又はヘキサメチレンテトラミンである、請求項16に記載のカルバメート化合物の製造方法。
- 前記活性水素を有さないアミン化合物が、含窒素芳香族基を1以上有し、且つ、炭素数が3以上85以下の化合物である、請求項14に記載のカルバメート化合物の製造方法。
- 前記含窒素芳香族基が、置換又は無置換の、ピリジル基、イミダゾリル基、ピラゾリル基、キノリル基、イソキノリル基、オキサゾリル基、チアゾリル基、ピリダジル基、又はピリミジル基、ピラジル基である、請求項18に記載のカルバメート化合物の製造方法。
- 前記活性水素を有さないアミン化合物が、ピリジン、ピコリン、ルチジン、コリジン、1-メチルイミダゾール、1-メチルピラゾール、キノリン、イソキノリン、メチルキノリン、オキサゾール、チアゾール、ピリダジン、ピリミジン、又はピラジンである、請求項19に記載のカルバメート化合物の製造方法。
- 前記アミジン基含有化合物が、環構造を1以上有し、且つ、前記R811、前記R812、前記R813、及び前記R814の合計炭素数が5以上30以下である、請求項21に記載のカルバメート化合物の製造方法。
- 前記アミジン基含有化合物が、1,2-ジメチル-1,4,5、6-テトラヒドロピリミジン、1,8-ジアザビシクロ-[5.4.0]ウンデカ-7-エン、又は1,5-ジアザビシクロ-[4.3.0]ノナ-5-エンである、請求項22に記載のカルバメート化合物の製造方法。
- 前記R821、前記R822、前記R823、前記R824、及び前記R825の合計炭素数が5以上30以下である、請求項24に記載のカルバメート化合物の製造方法。
- 前記グアニジン基含有化合物が、ペンタメチルグアニジン、又は7-メチル-1,5,7-トリアザビシクロ[4.4.0]デカ-5-エンである請求項25に記載のカルバメート化合物の製造方法。
- 前記第1級アミン化合物が、脂肪族又は芳香族第1級ポリアミン化合物である、請求項13に記載のカルバメート化合物の製造方法。
- 前記ヒドロキシ化合物が、芳香族ヒドロキシ化合物である、請求項13に記載のカルバメート化合物の製造方法。
- 請求項13~28のいずれか一項に記載のカルバメート化合物の製造方法により得られたカルバメート化合物を熱分解する熱分解工程を含む、イソシアネート化合物の製造方法。
- 下記一般式(VIIa)で表されるイソシアネート化合物の製造方法で副生する前記イソシアネート化合物よりも沸点の高い化合物を含む液相成分から、下記一般式(IIa)で表されるアミン化合物を回収する方法であって、
前記液相成分と、芳香族ヒドロキシ化合物と、活性水素含有化合物と、触媒と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a)を含む、回収方法。
- 前記工程(a)が、以下の工程(a1)及び工程(a2)を含む、請求項30に記載の回収方法。
前記液相成分と芳香族ヒドロキシ化合物を混合させる工程(a1);
前記工程(a1)で得られた混合物と、活性水素含有化合物と、触媒と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a2) - 前記工程(a2)が、以下の工程(a2-1)及び工程(a2-2)を含む、請求項31に記載の回収方法。
前記工程(a1)で得られた混合物と、活性水素含有化合物としてアミン化合物と、触媒と、を混合させる工程(a2-1);
前記工程(a2-1)で得られた混合物と、活性水素含有化合物として水と、を反応器内で反応させて、前記一般式(IIa)で表されるアミン化合物を含む反応液を得る工程(a2-2) - 前記活性水素含有化合物が水である、請求項30~32のいずれか一項に記載の回収方法。
- 前記活性水素含有化合物が水及び第一級アミン化合物である、請求項30~32のいずれか一項に記載の回収方法。
- 前記第一級アミン化合物が前記一般式(IIa)で表されるアミン化合物である、請求項34に記載の回収方法。
- 前記一般式(VIIa)で表されるイソシアネート化合物の製造方法において、炭酸誘導体と、ヒドロキシ化合物と、前記一般式(IIa)で表されるアミン化合物と、を原料として用いる、請求項30~32のいずれか一項に記載の回収方法。
- 前記液相成分が、炭酸誘導体と、ヒドロキシ化合物と、前記一般式(IIa)で表されるアミン化合物と、から生成されたカルバメート化合物を含む反応液を熱分解反応に供して、生成された前記一般式(VIIa)で表されるイソシアネート化合物を含む組成物を、蒸留装置に供給し、前記一般式(VIIa)で表されるイソシアネート化合物を気相成分として分離する際に、前記蒸留装置から抜き出される液相成分である、請求項30~32のいずれか一項に記載の回収方法。
- 前記液相成分が、前記式(IX-1)で表される基、前記式(IX-2)で表される基、前記式(IX-3)で表される基、前記式(IX-4)で表される基、前記式(IX-5)で表される基、前記式(IX-6)で表される基、前記式(IX-7)で表される基、前記式(IX-8)で表される基、前記式(IX-9)で表される基、前記式(IX-10)で表される基、前記式(IX-11)で表される基、及び前記式(IX-12)で表される基からなる群より選ばれる2種以上の官能基を有する化合物を含む、請求項38に記載の回収方法。
- 前記触媒が、アルカリ金属の水酸化物及び酸化物、アルカリ土類金属の水酸化物及び酸化物、第三級アミン化合物、第12族の金属酸化物、第13族の金属酸化物、並びに、第14族の金属酸化物からなる群より選ばれる1種以上の化合物である、請求項30~32のいずれか一項に記載の回収方法。
- 前記触媒が、アルカリ金属の水酸化物及び酸化物、並びに、アルカリ土類金属の水酸化物及び酸化物からなる群より選ばれる1種以上の化合物である、請求項40に記載の回収方法。
- 前記触媒が、アルカリ金属の水酸化物及び酸化物である、請求項41に記載の回収方法。
- 以下の工程(b)及び工程(c)を更に含む、請求項30~32のいずれか一項に記載の回収方法。
前記一般式(IIa)で表されるアミン化合物を含む反応液から前記一般式(IIa)で表されるアミン化合物を分離する工程(b);
前記一般式(IIa)で表されるアミン化合物を精製する工程(c) - 以下の工程(d)を更に含む、請求項43に記載の回収方法。
前記工程(c)で精製された前記一般式(IIa)で表されるアミン化合物を、前記一般式(VIIa)で表されるイソシアネート化合物の製造方法において、原料として再利用する工程(d) - 以下の工程(e)及び(f)を更に含む、請求項30~32のいずれか一項に記載の回収方法。
前記一般式(IIa)で表されるアミン化合物を含む反応液からヒドロキシ化合物を分離する工程(e);
前記ヒドロキシ化合物を精製する工程(f) - 以下の工程(g)を更に含む、請求項45に記載の回収方法。
前記工程(f)で精製された前記ヒドロキシ化合物を、前記一般式(VIIa)で表されるイソシアネート化合物の製造方法において、原料として再利用する工程(g) - 以下の工程(h)を更に含む、請求項45に記載の回収方法。
前記工程(b)において前記一般式(IIa)で表されるアミン化合物を分離した後の残留液、前記工程(c)において前記一般式(IIa)で表されるアミン化合物を精製した後の残留液、前記工程(e)において前記ヒドロキシ化合物を分離した後の残留液、及び前記工程(f)において前記ヒドロキシ化合物を精製した後の残留液からなる群より選ばれる1種以上の残留液を、前記工程(a)において再利用する工程(h) - イソシアネート化合物と、
下記一般式(I)で表されるカルボニル化合物と、
前記イソシアネート化合物のイソシアネート基の一部が、イソシアヌレート基、カルボジイミド基、ウレトンイミン基、及びアロファネート基からなる群より選ばれる1種以上の官能基に変換された、イソシアネート化合物の変性物と、
を含む、イソシアネート組成物であって、
前記イソシアネート化合物と前記カルボニル化合物は異なる化合物であり、
{3×(イソシアヌレート基のモル量)+2×(カルボジイミド基のモル量)+3×(ウレトンイミン基のモル量)+2×(アロファネート基のモル量)}÷(カルボニル化合物のモル量)の値が0.001以上8.0以下である、イソシアネート組成物。
- 前記R11が、1以上4以下のエステル基又は窒素原子を有してもよい、炭素数1以上20以下の2価以上4価以下の脂肪族炭化水素基、又は、炭素数6以上20以下の2価以上3価以下の芳香族炭化水素基であり、且つ、
前記R12が、酸素原子を含んでもよい、炭素数6以上20以下の1価の芳香族炭化水素基である、請求項48に記載のイソシアネート組成物。 - 前記イソシアネート組成物の総質量に対して、
前記イソシアネート化合物の含有量が1質量%以上99質量%以下であり、且つ、
前記カルボニル化合物及び前記イソシアネート化合物の変性物の合計含有量が1質量%以上99質量%以下である、請求項48又は49に記載のイソシアネート組成物。 - 前記イソシアネート組成物の総質量に対して、それぞれ2.0質量ppm以上99質量%以下の、下記一般式(XI)で表されるカルバメート基含有化合物及び下記一般式(III-1a)で表される炭酸エステルからなる群より選ばれる1種以上の化合物を更に含有する、請求項48又は49に記載のイソシアネート組成物。
- 前記イソシアネート組成物の総質量に対して、それぞれ2.0質量ppm以上1.0×104質量ppm以下の、前記一般式(XI)で表されるカルバメート基含有化合物及び前記一般式(III-1a)で表される炭酸エステルからなる群より選ばれる1種以上の化合物を含有する、請求項53に記載のイソシアネート組成物。
- 請求項48又は49に記載のイソシアネート組成物を蒸留精製し、連続的に気相成分として、前記イソシアネート化合物を回収することを含む、イソシアネート化合物の製造方法。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202280073806.6A CN118215651A (zh) | 2021-11-08 | 2022-11-08 | 异氰酸酯化合物的制造方法、氨基甲酸酯化合物的制造方法、胺化合物的回收方法、异氰酸酯组合物 |
| JP2023558105A JPWO2023080257A1 (ja) | 2021-11-08 | 2022-11-08 | |
| EP22890077.5A EP4431490A1 (en) | 2021-11-08 | 2022-11-08 | Method for producing isocyanate compounds, method for producing carbamate compounds, method for recovering amine compounds, and isocyanate composition |
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021182195 | 2021-11-08 | ||
| JP2021182191 | 2021-11-08 | ||
| JP2021182194 | 2021-11-08 | ||
| JP2021-182195 | 2021-11-08 | ||
| JP2021-182194 | 2021-11-08 | ||
| JP2021-182191 | 2021-11-08 | ||
| JP2021199645 | 2021-12-08 | ||
| JP2021-199645 | 2021-12-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023080257A1 true WO2023080257A1 (ja) | 2023-05-11 |
Family
ID=86241638
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/041611 WO2023080257A1 (ja) | 2021-11-08 | 2022-11-08 | イソシアネート化合物の製造方法、カルバメート化合物の製造方法、アミン化合物の回収方法、イソシアネート組成物 |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP4431490A1 (ja) |
| JP (1) | JPWO2023080257A1 (ja) |
| WO (1) | WO2023080257A1 (ja) |
Citations (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5271443A (en) | 1975-12-03 | 1977-06-14 | Anic Spa | Production of aromatic urethane |
| JPS588380B2 (ja) | 1974-05-09 | 1983-02-15 | 武田薬品工業株式会社 | 有機アミン類の製造法 |
| US4386033A (en) | 1980-12-19 | 1983-05-31 | Bayer Aktiengesellschaft | Process for the continuous thermal cleavage of carbamic acid esters and preparation of isocyanates from the products thereof |
| JPS5848538B2 (ja) | 1978-03-31 | 1983-10-28 | 三井東圧化学株式会社 | アミンの回収方法 |
| JPS59205352A (ja) | 1983-04-23 | 1984-11-20 | バスフ アクチエンゲゼルシヤフト | ヘキサメチレン−ジイソシアネ−ト−1,6及び/又はアルキレン基中に6個の炭素原子を有する異性体の脂肪族ジイソシアネ−トの多工程式製法 |
| JPS6438055A (en) | 1979-04-30 | 1989-02-08 | Basf Ag | Manufacture of fatty, alicyclic and heterocyclicdi- and/or polyurethane |
| JPH03199645A (ja) | 1989-12-27 | 1991-08-30 | Yanmar Diesel Engine Co Ltd | 移動農機の電子制御式燃料噴射装置 |
| JPH05255227A (ja) | 1991-11-14 | 1993-10-05 | Bayer Ag | ポリイソシアネートの製造方法 |
| JPH0625136A (ja) | 1992-04-21 | 1994-02-01 | Basf Ag | 有機ポリイソシアネートを連続的に製造するための多工程法 |
| JPH0641045B2 (ja) | 1984-11-26 | 1994-06-01 | キヤノン株式会社 | 切削加工装置 |
| JPH08217744A (ja) | 1995-02-13 | 1996-08-27 | Asahi Chem Ind Co Ltd | ウレタンの連続的製造法 |
| JP2002173471A (ja) | 2000-12-07 | 2002-06-21 | Kobe Steel Ltd | イソシアネート系分解対象化合物の分解回収方法 |
| JP2002518369A (ja) | 1998-06-18 | 2002-06-25 | ビーエーエスエフ アクチェンゲゼルシャフト | トリレンジイソシアネートの合成で生成する蒸留残渣の後処理 |
| WO2007036479A1 (de) | 2005-09-29 | 2007-04-05 | Basf Se | Verfahren zur aufreinigung von isocyanathaltigen rückständen |
| WO2009127591A2 (de) | 2008-04-14 | 2009-10-22 | Basf Se | Verfahren zur aufarbeitung von rückständen aus der produktion von isocyanaten |
| WO2009130842A1 (ja) | 2008-04-26 | 2009-10-29 | 日本ポリウレタン工業株式会社 | ウレア化合物の分解処理方法 |
| WO2010110142A1 (ja) | 2009-03-26 | 2010-09-30 | 三井化学株式会社 | ウレタン化合物およびその製造方法、および、イソシアネートおよびその製造方法 |
| WO2010116871A1 (ja) | 2009-04-09 | 2010-10-14 | 三井化学株式会社 | ウレタン化合物およびその製造方法、および、イソシアネートおよびその製造方法 |
| WO2011021257A1 (ja) * | 2009-08-21 | 2011-02-24 | 旭化成ケミカルズ株式会社 | N-置換カルバミン酸エステルの製造方法、該n-置換カルバミン酸エステルを用いるイソシアネートの製造方法、ならびにn-置換カルバミン酸エステルおよび芳香族ヒドロキシ化合物を含むn-置換カルバミン酸エステルの移送用および貯蔵用組成物 |
| WO2011089932A1 (ja) | 2010-01-22 | 2011-07-28 | 三井化学株式会社 | トルエンスルホン酸亜鉛の製造方法、トルエンスルホン酸亜鉛およびカルバメートの製造方法 |
| WO2013111353A1 (ja) | 2012-01-25 | 2013-08-01 | 旭化成ケミカルズ株式会社 | 分離方法 |
| JP5563886B2 (ja) | 2010-05-11 | 2014-07-30 | 三井化学株式会社 | イソシアネート残渣の処理方法 |
| WO2014157636A1 (ja) | 2013-03-29 | 2014-10-02 | 旭化成ケミカルズ株式会社 | イソシアネートの製造方法 |
| WO2015030106A1 (ja) * | 2013-09-02 | 2015-03-05 | 国立大学法人京都大学 | Ggt阻害作用を有する化合物及びggtファミリー酵素阻害剤 |
| WO2019131855A1 (ja) | 2017-12-27 | 2019-07-04 | 旭化成株式会社 | 有機アミンの回収方法 |
| JP2021182194A (ja) | 2020-05-18 | 2021-11-25 | 株式会社ユニオンソフトウェアマネイジメント | 画像認識による寸法測定システム及び寸法測定方法 |
| JP2021182195A (ja) | 2020-05-18 | 2021-11-25 | 株式会社東海理化電機製作所 | 管理システム及び管理方法 |
| JP2021182191A (ja) | 2020-05-18 | 2021-11-25 | トヨタ自動車株式会社 | エージェント制御装置、エージェント制御方法及びエージェント制御プログラム |
-
2022
- 2022-11-08 WO PCT/JP2022/041611 patent/WO2023080257A1/ja active Application Filing
- 2022-11-08 JP JP2023558105A patent/JPWO2023080257A1/ja active Pending
- 2022-11-08 EP EP22890077.5A patent/EP4431490A1/en active Pending
Patent Citations (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS588380B2 (ja) | 1974-05-09 | 1983-02-15 | 武田薬品工業株式会社 | 有機アミン類の製造法 |
| JPS5271443A (en) | 1975-12-03 | 1977-06-14 | Anic Spa | Production of aromatic urethane |
| JPS5848538B2 (ja) | 1978-03-31 | 1983-10-28 | 三井東圧化学株式会社 | アミンの回収方法 |
| JPS6438055A (en) | 1979-04-30 | 1989-02-08 | Basf Ag | Manufacture of fatty, alicyclic and heterocyclicdi- and/or polyurethane |
| US4386033A (en) | 1980-12-19 | 1983-05-31 | Bayer Aktiengesellschaft | Process for the continuous thermal cleavage of carbamic acid esters and preparation of isocyanates from the products thereof |
| JPS59205352A (ja) | 1983-04-23 | 1984-11-20 | バスフ アクチエンゲゼルシヤフト | ヘキサメチレン−ジイソシアネ−ト−1,6及び/又はアルキレン基中に6個の炭素原子を有する異性体の脂肪族ジイソシアネ−トの多工程式製法 |
| JPH0641045B2 (ja) | 1984-11-26 | 1994-06-01 | キヤノン株式会社 | 切削加工装置 |
| JPH03199645A (ja) | 1989-12-27 | 1991-08-30 | Yanmar Diesel Engine Co Ltd | 移動農機の電子制御式燃料噴射装置 |
| JPH05255227A (ja) | 1991-11-14 | 1993-10-05 | Bayer Ag | ポリイソシアネートの製造方法 |
| JPH0625136A (ja) | 1992-04-21 | 1994-02-01 | Basf Ag | 有機ポリイソシアネートを連続的に製造するための多工程法 |
| JPH08217744A (ja) | 1995-02-13 | 1996-08-27 | Asahi Chem Ind Co Ltd | ウレタンの連続的製造法 |
| JP2002518369A (ja) | 1998-06-18 | 2002-06-25 | ビーエーエスエフ アクチェンゲゼルシャフト | トリレンジイソシアネートの合成で生成する蒸留残渣の後処理 |
| JP2002173471A (ja) | 2000-12-07 | 2002-06-21 | Kobe Steel Ltd | イソシアネート系分解対象化合物の分解回収方法 |
| WO2007036479A1 (de) | 2005-09-29 | 2007-04-05 | Basf Se | Verfahren zur aufreinigung von isocyanathaltigen rückständen |
| WO2009127591A2 (de) | 2008-04-14 | 2009-10-22 | Basf Se | Verfahren zur aufarbeitung von rückständen aus der produktion von isocyanaten |
| JP5240678B2 (ja) | 2008-04-26 | 2013-07-17 | 日本ポリウレタン工業株式会社 | ウレア化合物の分解処理方法 |
| WO2009130842A1 (ja) | 2008-04-26 | 2009-10-29 | 日本ポリウレタン工業株式会社 | ウレア化合物の分解処理方法 |
| WO2010110142A1 (ja) | 2009-03-26 | 2010-09-30 | 三井化学株式会社 | ウレタン化合物およびその製造方法、および、イソシアネートおよびその製造方法 |
| WO2010116871A1 (ja) | 2009-04-09 | 2010-10-14 | 三井化学株式会社 | ウレタン化合物およびその製造方法、および、イソシアネートおよびその製造方法 |
| WO2011021257A1 (ja) * | 2009-08-21 | 2011-02-24 | 旭化成ケミカルズ株式会社 | N-置換カルバミン酸エステルの製造方法、該n-置換カルバミン酸エステルを用いるイソシアネートの製造方法、ならびにn-置換カルバミン酸エステルおよび芳香族ヒドロキシ化合物を含むn-置換カルバミン酸エステルの移送用および貯蔵用組成物 |
| WO2011089932A1 (ja) | 2010-01-22 | 2011-07-28 | 三井化学株式会社 | トルエンスルホン酸亜鉛の製造方法、トルエンスルホン酸亜鉛およびカルバメートの製造方法 |
| JP5563886B2 (ja) | 2010-05-11 | 2014-07-30 | 三井化学株式会社 | イソシアネート残渣の処理方法 |
| WO2013111353A1 (ja) | 2012-01-25 | 2013-08-01 | 旭化成ケミカルズ株式会社 | 分離方法 |
| WO2014157636A1 (ja) | 2013-03-29 | 2014-10-02 | 旭化成ケミカルズ株式会社 | イソシアネートの製造方法 |
| WO2015030106A1 (ja) * | 2013-09-02 | 2015-03-05 | 国立大学法人京都大学 | Ggt阻害作用を有する化合物及びggtファミリー酵素阻害剤 |
| WO2019131855A1 (ja) | 2017-12-27 | 2019-07-04 | 旭化成株式会社 | 有機アミンの回収方法 |
| JP2021182194A (ja) | 2020-05-18 | 2021-11-25 | 株式会社ユニオンソフトウェアマネイジメント | 画像認識による寸法測定システム及び寸法測定方法 |
| JP2021182195A (ja) | 2020-05-18 | 2021-11-25 | 株式会社東海理化電機製作所 | 管理システム及び管理方法 |
| JP2021182191A (ja) | 2020-05-18 | 2021-11-25 | トヨタ自動車株式会社 | エージェント制御装置、エージェント制御方法及びエージェント制御プログラム |
Non-Patent Citations (6)
| Title |
|---|
| "Nomenclature of Organic Chemistry and Biochemistry", 1992, NANKODO CO., LTD |
| DRITTER JAHRGANG: "186. A. W. Hofmann: Ueber die aromatischen Cyanate", BERICHTE DER DEUTSCHEN CHEMISCHEN GESELLSCHAFT, vol. 3, 8 January 1970 (1970-01-08), pages 653 - 658 |
| DYER E ET AL.: "Thermal Degradation of Alkyl N-Phenylcarbomates", JOURNAL OF AMERICAN CHEMICAL SOCIETY, vol. 81, 1959, pages 2138 - 2143 |
| ENGLUND ETHAN A., GOPI HOSAHUDYA N., APPELLA DANIEL H.: "An Efficient Synthesis of a Probe for Protein Function: 2,3-Diaminopropionic Acid with Orthogonal Protecting Groups", ORGANIC LETTERS, AMERICAN CHEMICAL SOCIETY, US, vol. 6, no. 2, 1 January 2004 (2004-01-01), US , pages 213 - 215, XP093062691, ISSN: 1523-7060, DOI: 10.1021/ol0361599 * |
| SCHWETLICK K ET AL.: "Kinetics and Catalysis of Consecutive Isocyanate Reactions. Formation of Carbamates, Allophanates and Isocyanurates", JOURNAL OF THE CHEMICAL SOCIETY, PERKIN TRANSACTIONS II, vol. 2, 1995, pages 395 - 402, XP009077345, DOI: 10.1039/p29950000395 |
| ULRICH H ET AL.: "2+2] Cycloaddition Reactions of Unsymmetrically substituted Carbodiimides", JOURNAL OF HETEROCYCLIC CHEMISTRY, vol. 24, 1987, pages 1121 - 1123, XP055795653, DOI: 10.1002/jhet.5570240438 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2023080257A1 (ja) | 2023-05-11 |
| EP4431490A1 (en) | 2024-09-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6316491B2 (ja) | イソシアネートの製造方法 | |
| ES2710177T3 (es) | Proceso para la producción de isocianato | |
| JP6546233B2 (ja) | ポリイソシアネート組成物及びイソシアネート重合体組成物 | |
| JP4958971B2 (ja) | カルバミン酸エステルおよび芳香族ヒドロキシ化合物を含む組成物を用いるイソシアネートの製造方法、ならびにカルバミン酸エステル移送用および貯蔵用組成物 | |
| JP6412238B2 (ja) | イソチオシアネートを製造する方法 | |
| US9000219B2 (en) | Method for treatment of isocyanate residue, and method for treatment of carbonate | |
| WO2023080257A1 (ja) | イソシアネート化合物の製造方法、カルバメート化合物の製造方法、アミン化合物の回収方法、イソシアネート組成物 | |
| JP7214806B2 (ja) | 有機アミンの回収方法 | |
| CN118215651A (zh) | 异氰酸酯化合物的制造方法、氨基甲酸酯化合物的制造方法、胺化合物的回收方法、异氰酸酯组合物 | |
| WO2023080258A1 (ja) | カルボニル化合物、カルボニル化合物の製造方法、イソシアネート化合物の製造方法、及びイソシアネート組成物 | |
| JP2022170318A (ja) | イソシアネートの製造方法 | |
| JP2023092840A (ja) | イソシアネートの製造方法 | |
| JP2023091331A (ja) | イソシアネートの製造方法 | |
| CN118302407A (zh) | 羰基化合物、羰基化合物的制造方法、异氰酸酯化合物的制造方法以及异氰酸酯组合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22890077 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023558105 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18703834 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280073806.6 Country of ref document: CN |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112024008453 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022890077 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2022890077 Country of ref document: EP Effective date: 20240610 |
|
| ENP | Entry into the national phase |
Ref document number: 112024008453 Country of ref document: BR Kind code of ref document: A2 Effective date: 20240429 |