WO2022172863A1 - 溶着フィルム及び接合体 - Google Patents
溶着フィルム及び接合体 Download PDFInfo
- Publication number
- WO2022172863A1 WO2022172863A1 PCT/JP2022/004352 JP2022004352W WO2022172863A1 WO 2022172863 A1 WO2022172863 A1 WO 2022172863A1 JP 2022004352 W JP2022004352 W JP 2022004352W WO 2022172863 A1 WO2022172863 A1 WO 2022172863A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- welding
- molecular weight
- film
- bisphenol
- resin
- Prior art date
Links
- 238000003466 welding Methods 0.000 title claims abstract description 67
- 239000013034 phenoxy resin Substances 0.000 claims abstract description 40
- 229920006287 phenoxy resin Polymers 0.000 claims abstract description 40
- 238000000034 method Methods 0.000 claims description 33
- 239000000463 material Substances 0.000 claims description 23
- 238000004519 manufacturing process Methods 0.000 claims description 21
- -1 polypropylene Polymers 0.000 claims description 17
- 239000000758 substrate Substances 0.000 claims description 17
- 229910052782 aluminium Inorganic materials 0.000 claims description 14
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 14
- 238000012360 testing method Methods 0.000 claims description 14
- 229920000515 polycarbonate Polymers 0.000 claims description 11
- 239000004417 polycarbonate Substances 0.000 claims description 11
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 10
- 238000010438 heat treatment Methods 0.000 claims description 7
- 229920002430 Fibre-reinforced plastic Polymers 0.000 claims description 6
- 239000011151 fibre-reinforced plastic Substances 0.000 claims description 6
- 239000011521 glass Substances 0.000 claims description 6
- 230000006698 induction Effects 0.000 claims description 6
- 239000000919 ceramic Substances 0.000 claims description 5
- 229910052742 iron Inorganic materials 0.000 claims description 5
- 229920001707 polybutylene terephthalate Polymers 0.000 claims description 5
- 239000004952 Polyamide Substances 0.000 claims description 4
- 239000004697 Polyetherimide Substances 0.000 claims description 4
- 239000004743 Polypropylene Substances 0.000 claims description 4
- 229920003229 poly(methyl methacrylate) Polymers 0.000 claims description 4
- 229920002647 polyamide Polymers 0.000 claims description 4
- 229920001601 polyetherimide Polymers 0.000 claims description 4
- 239000004926 polymethyl methacrylate Substances 0.000 claims description 4
- 229920001155 polypropylene Polymers 0.000 claims description 4
- 238000007731 hot pressing Methods 0.000 claims description 3
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 36
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 30
- 239000003822 epoxy resin Substances 0.000 description 25
- 229920000647 polyepoxide Polymers 0.000 description 25
- 239000007787 solid Substances 0.000 description 25
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 24
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 24
- 230000001588 bifunctional effect Effects 0.000 description 22
- 238000011282 treatment Methods 0.000 description 22
- 239000011342 resin composition Substances 0.000 description 19
- 150000001875 compounds Chemical class 0.000 description 18
- 229920005989 resin Polymers 0.000 description 16
- 239000011347 resin Substances 0.000 description 16
- 239000002904 solvent Substances 0.000 description 16
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 14
- 239000002585 base Substances 0.000 description 13
- 239000000126 substance Substances 0.000 description 13
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 12
- 238000006243 chemical reaction Methods 0.000 description 11
- 239000012299 nitrogen atmosphere Substances 0.000 description 11
- 238000004090 dissolution Methods 0.000 description 10
- 239000007789 gas Substances 0.000 description 10
- 239000000203 mixture Substances 0.000 description 10
- 238000005422 blasting Methods 0.000 description 9
- 238000010992 reflux Methods 0.000 description 9
- 238000005304 joining Methods 0.000 description 8
- 238000003756 stirring Methods 0.000 description 8
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 238000005227 gel permeation chromatography Methods 0.000 description 6
- 238000005498 polishing Methods 0.000 description 6
- 239000004593 Epoxy Substances 0.000 description 5
- 239000003054 catalyst Substances 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 239000002184 metal Substances 0.000 description 5
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 4
- 238000004140 cleaning Methods 0.000 description 4
- 238000005238 degreasing Methods 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 238000009832 plasma treatment Methods 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 239000000853 adhesive Substances 0.000 description 3
- 230000001070 adhesive effect Effects 0.000 description 3
- 239000010960 cold rolled steel Substances 0.000 description 3
- 238000003851 corona treatment Methods 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- 238000005530 etching Methods 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 239000012943 hotmelt Substances 0.000 description 3
- 230000014759 maintenance of location Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 3
- 229930185605 Bisphenol Natural products 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- 229910000831 Steel Inorganic materials 0.000 description 2
- KKEYFWRCBNTPAC-UHFFFAOYSA-N Terephthalic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-N 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000001746 injection moulding Methods 0.000 description 2
- QQVIHTHCMHWDBS-UHFFFAOYSA-N isophthalic acid Chemical compound OC(=O)C1=CC=CC(C(O)=O)=C1 QQVIHTHCMHWDBS-UHFFFAOYSA-N 0.000 description 2
- 238000013532 laser treatment Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 2
- 229910052753 mercury Inorganic materials 0.000 description 2
- 230000010355 oscillation Effects 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 229920005749 polyurethane resin Polymers 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 238000007788 roughening Methods 0.000 description 2
- 239000010959 steel Substances 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- AHDSRXYHVZECER-UHFFFAOYSA-N 2,4,6-tris[(dimethylamino)methyl]phenol Chemical compound CN(C)CC1=CC(CN(C)C)=C(O)C(CN(C)C)=C1 AHDSRXYHVZECER-UHFFFAOYSA-N 0.000 description 1
- HSDVRWZKEDRBAG-UHFFFAOYSA-N 2-[1-(oxiran-2-ylmethoxy)hexoxymethyl]oxirane Chemical compound C1OC1COC(CCCCC)OCC1CO1 HSDVRWZKEDRBAG-UHFFFAOYSA-N 0.000 description 1
- KXGFMDJXCMQABM-UHFFFAOYSA-N 2-methoxy-6-methylphenol Chemical compound [CH]OC1=CC=CC([CH])=C1O KXGFMDJXCMQABM-UHFFFAOYSA-N 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 238000004026 adhesive bonding Methods 0.000 description 1
- 239000004844 aliphatic epoxy resin Substances 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 238000004873 anchoring Methods 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 239000001273 butane Substances 0.000 description 1
- 238000003486 chemical etching Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000000567 combustion gas Substances 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- KPVWDKBJLIDKEP-UHFFFAOYSA-L dihydroxy(dioxo)chromium;sulfuric acid Chemical compound OS(O)(=O)=O.O[Cr](O)(=O)=O KPVWDKBJLIDKEP-UHFFFAOYSA-L 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000000866 electrolytic etching Methods 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 1
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical class [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 150000002505 iron Chemical class 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 229910001416 lithium ion Inorganic materials 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 description 1
- OFBQJSOFQDEBGM-UHFFFAOYSA-N n-pentane Natural products CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 1
- 229920003986 novolac Polymers 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 229920001568 phenolic resin Polymers 0.000 description 1
- 239000005011 phenolic resin Substances 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- YXJYBPXSEKMEEJ-UHFFFAOYSA-N phosphoric acid;sulfuric acid Chemical compound OP(O)(O)=O.OS(O)(=O)=O YXJYBPXSEKMEEJ-UHFFFAOYSA-N 0.000 description 1
- 238000001020 plasma etching Methods 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 239000012779 reinforcing material Substances 0.000 description 1
- 238000005488 sandblasting Methods 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- 229920005992 thermoplastic resin Polymers 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000013585 weight reducing agent Substances 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C65/00—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor
- B29C65/02—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C65/00—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor
- B29C65/02—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure
- B29C65/08—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure using ultrasonic vibrations
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C65/00—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor
- B29C65/02—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure
- B29C65/18—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure using heated tools
- B29C65/20—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure using heated tools with direct contact, e.g. using "mirror"
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C65/00—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor
- B29C65/02—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure
- B29C65/18—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure using heated tools
- B29C65/24—Joining or sealing of preformed parts, e.g. welding of plastics materials; Apparatus therefor by heating, with or without pressure using heated tools characterised by the means for heating the tool
- B29C65/30—Electrical means
- B29C65/32—Induction
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/18—Layered products comprising a layer of metal comprising iron or steel
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/20—Layered products comprising a layer of metal comprising aluminium or copper
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/28—Layered products comprising a layer of synthetic resin comprising synthetic resins not wholly covered by any one of the sub-groups B32B27/30 - B32B27/42
- B32B27/281—Layered products comprising a layer of synthetic resin comprising synthetic resins not wholly covered by any one of the sub-groups B32B27/30 - B32B27/42 comprising polyimides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/32—Layered products comprising a layer of synthetic resin comprising polyolefins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/34—Layered products comprising a layer of synthetic resin comprising polyamides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
- B32B27/365—Layered products comprising a layer of synthetic resin comprising polyesters comprising polycarbonates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B37/00—Methods or apparatus for laminating, e.g. by curing or by ultrasonic bonding
- B32B37/12—Methods or apparatus for laminating, e.g. by curing or by ultrasonic bonding characterised by using adhesives
- B32B37/1207—Heat-activated adhesive
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B7/00—Layered products characterised by the relation between layers; Layered products characterised by the relative orientation of features between layers, or by the relative values of a measurable parameter between layers, i.e. products comprising layers having different physical, chemical or physicochemical properties; Layered products characterised by the interconnection of layers
- B32B7/04—Interconnection of layers
- B32B7/12—Interconnection of layers using interposed adhesives or interposed materials with bonding properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B9/00—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00
- B32B9/005—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising one layer of ceramic material, e.g. porcelain, ceramic tile
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J171/00—Adhesives based on polyethers obtained by reactions forming an ether link in the main chain; Adhesives based on derivatives of such polymers
- C09J171/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C09J171/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J5/00—Adhesive processes in general; Adhesive processes not provided for elsewhere, e.g. relating to primers
- C09J5/10—Joining materials by welding overlapping edges with an insertion of plastic material
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J7/00—Adhesives in the form of films or foils
- C09J7/10—Adhesives in the form of films or foils without carriers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J7/00—Adhesives in the form of films or foils
- C09J7/30—Adhesives in the form of films or foils characterised by the adhesive composition
- C09J7/35—Heat-activated
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B37/00—Methods or apparatus for laminating, e.g. by curing or by ultrasonic bonding
- B32B37/12—Methods or apparatus for laminating, e.g. by curing or by ultrasonic bonding characterised by using adhesives
- B32B37/1207—Heat-activated adhesive
- B32B2037/1215—Hot-melt adhesive
- B32B2037/1223—Hot-melt adhesive film-shaped
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/50—Properties of the layers or laminate having particular mechanical properties
- B32B2307/542—Shear strength
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/732—Dimensional properties
- B32B2307/737—Dimensions, e.g. volume or area
- B32B2307/7375—Linear, e.g. length, distance or width
- B32B2307/7376—Thickness
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2371/00—Characterised by the use of polyethers obtained by reactions forming an ether link in the main chain; Derivatives of such polymers
- C08J2371/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08J2371/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2203/00—Applications of adhesives in processes or use of adhesives in the form of films or foils
- C09J2203/318—Applications of adhesives in processes or use of adhesives in the form of films or foils for the production of liquid crystal displays
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2203/00—Applications of adhesives in processes or use of adhesives in the form of films or foils
- C09J2203/326—Applications of adhesives in processes or use of adhesives in the form of films or foils for bonding electronic components such as wafers, chips or semiconductors
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2203/00—Applications of adhesives in processes or use of adhesives in the form of films or foils
- C09J2203/354—Applications of adhesives in processes or use of adhesives in the form of films or foils for automotive applications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2301/00—Additional features of adhesives in the form of films or foils
- C09J2301/30—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier
- C09J2301/304—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier the adhesive being heat-activatable, i.e. not tacky at temperatures inferior to 30°C
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2301/00—Additional features of adhesives in the form of films or foils
- C09J2301/30—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier
- C09J2301/312—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier parameters being the characterizing feature
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2463/00—Presence of epoxy resin
Definitions
- the present invention relates to a welding film suitable for use in firmly welding the same or different resin materials, the same or different metals, and metals and resins.
- Hot melt epoxy resin, reactive polyurethane resin, etc. have been used as welding materials.
- Hot melts have been developed using the number average molecular weight and the weight average molecular weight as indexes, and various formulations have been studied (for example, Patent Document 1).
- the present invention has been made in view of such circumstances, and aims to provide a welded film that is excellent in welding strength and shape retention, a joined body using the welded film, and a method for producing the same.
- a welding film made of a phenoxy resin having a z-average molecular weight and a ratio of the z-average molecular weight to the number-average molecular weight [Mz/Mn] of a specific value or more can solve the above problems.
- the welding film has a tensile shear strength of 10 MPa or more when welded between the first substrate and the second substrate in a test according to JIS K 6850:1999. Or the welding film according to [2].
- a joined body obtained by welding the welding film according to any one of [1] to [3] above between a first substrate and a second substrate.
- the first base material and the second base material are aluminum, iron, fiber-reinforced plastic, glass, ceramic, polypropylene, polycarbonate, polymethyl methacrylate, polyetherimide, polyamide, and polybutylene terephthalate.
- the welding film is welded between the first substrate and the second substrate by at least one method selected from the group consisting of heating, hot pressing, ultrasonic welding, and high-frequency induction welding.
- the manufacturing method of the joined body is welded between the first substrate and the second substrate by at least one method selected from the group consisting of heating, hot pressing, ultrasonic welding, and high-frequency induction welding.
- the present invention it is possible to provide a welding film excellent in welding strength and shape retention, a joined body using the welding film, and a method for producing the same.
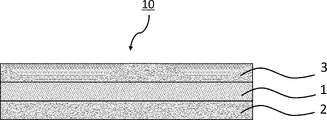
- FIG. 2 is an explanatory diagram showing the configuration of a joined body in one embodiment of the present invention
- joining means joining things together, and adhesion and welding are subordinate concepts thereof.
- Adhesion means bonding two adherends (things to be adhered) via an organic material (thermosetting resin, thermoplastic resin, etc.) such as tape and adhesive.
- the welding film of the present embodiment is made of a phenoxy resin having a z-average molecular weight of 70,000 or more and a ratio [Mz/Mn] of the z-average molecular weight to the number average molecular weight of the phenoxy resin of 5.5. It is characterized by being 0 or more.
- the phenoxy resin has a z-average molecular weight (Mz) of 70,000 or more. If the Mz is less than 70,000, the film formability and welding strength may be lowered.
- the Mz is preferably from 70,000 to 800,000, more preferably from 70,000 to 600,000, still more preferably from 70,000 to 70,000, from the viewpoint of improving film moldability and increasing welding strength. 400,000, more preferably 80,000 to 250,000.
- the Mz, and the Mn and Mw described later can be measured by a method using a device that separates the difference in molecular size using a column having pores. It can be measured by the method described.
- the Mz of the phenoxy resin is 70,000 or more, it is presumed that the phenoxy resin is appropriately mixed with a three-dimensionally branched resin and a linear resin. As a result, the phenoxy resin has good film formability, and the welding film made of the phenoxy resin has excellent welding strength. Moreover, the storage stability and repairability of the welding film are excellent.
- the phenoxy resin may contain a linear, relatively low molecular weight resin. By containing the low-molecular-weight resin, the wettability of the welding film to the adherend can be enhanced.
- the phenoxy resin preferably has a number average molecular weight (Mn) of 6,000 to 30,000, more preferably 6,000 to 25,000, still more preferably 6,000 to 20,000, Even more preferably 7,000 to 20,000.
- Mn number average molecular weight
- a resin having three-dimensional branches is present to a certain extent, and relatively low-molecular-weight resins are likely to be included, and welding strength and wettability to adherends are improved. will be excellent.
- the ratio [Mz/Mn] between the Mz and the Mn is 5.0 or more. If the ratio [Mz/Mn] is less than 5.0, film formability and welding strength may be lowered.
- the ratio [Mz/Mn] is preferably from 5.0 to 200.0, more preferably from 5.0 to 100.0, and even more preferably from the viewpoint of improving film formability and increasing welding strength. is 5.0 to 50.0, more preferably 5.0 to 35.0, still more preferably 5.0 to 30.0, still more preferably 5.0 to 24.0 is.
- the phenoxy resin preferably has a weight average molecular weight (Mw) of 10,000 to 500,000, more preferably 20,000 to 300,000, still more preferably 25,000 to 150,000, Even more preferably 25,000 to 120,000, and even more preferably 30,000 to 120,000.
- Mw weight average molecular weight
- a resin having three-dimensional branches is present to a certain extent, and relatively low-molecular-weight resins are likely to be included, resulting in excellent film moldability and welding strength. Become.
- the phenoxy resin used in the present invention is not particularly limited as long as the Mz and the ratio [Mz/Mn] are not less than the values described above. and a resin obtained by reacting a compound in the presence of a catalyst.
- bifunctional epoxy compound examples include aromatic epoxy resins such as bisphenol A type epoxy resin, bisphenol F type epoxy resin, bisphenol S type epoxy resin, biphenol type epoxy resin, and naphthalene type bifunctional epoxy resin; Aliphatic epoxy resins such as hexanediol diglycidyl ether can be mentioned. These may be used individually by 1 type, or may use 2 or more types together.
- a bisphenol A type epoxy resin is particularly preferred, and its molecular weight is preferably 250 to 6,000, more preferably 300 to 6,000, still more preferably 700 to 5,000. When the molecular weight is within this range, good film formability is obtained.
- Examples of the compound having a bifunctional hydroxyl group include phenols such as bisphenol A, bisphenol F, bisphenol S, and biphenol; and aliphatic glycols such as ethylene glycol, propylene glycol, and diethylene glycol.
- bisphenol A and bisphenol S are preferable, and bisphenol S is particularly preferable, from the viewpoint of cost, adhesiveness, and water resistance. These may be used individually by 1 type, or may use 2 or more types together.
- the amount of the compound having a bifunctional hydroxyl group is preferably 0.4 to 1.2 equivalents, more preferably 0.5 to 1.0 equivalents, per 1.0 equivalents of the bifunctional epoxy compound. Equivalent. When the compounding amount of the compound having a bifunctional hydroxyl group is within the above range, good film formability is obtained. Also, the Mz and the ratio [Mz/Mn] of the phenoxy resin can be easily adjusted to the above values or more.
- tertiary amines such as triethylamine and 2,4,6-tris(dimethylaminomethyl)phenol
- phosphorous compounds such as triphenylphosphine, and the like are preferably used.
- triphenylphosphine is preferable from the viewpoint of stability.
- the amount of the catalyst used is not particularly limited, but is preferably 0.01 to 10.00 parts by mass with respect to a total of 100 parts by mass of the bifunctional epoxy compound and the compound having a difunctional hydroxyl group, and more It is preferably 0.10 to 1.00 parts by mass, more preferably 0.15 to 0.50 parts by mass.
- the reaction between the bifunctional epoxy compound and the compound having a bifunctional hydroxyl group is preferably carried out at 50-200°C.
- bifunctional carboxy compounds and bifunctional thiol compounds can be used in addition to compounds having bifunctional hydroxyl groups.
- the bifunctional carboxy compound may be any compound having two carboxy groups in the molecule, for example, aromatic dicarboxylic acids such as terephthalic acid and isophthalic acid; acids, maleic acid, fumaric acid and other aliphatic dicarboxylic acids.
- aromatic dicarboxylic acids such as terephthalic acid and isophthalic acid
- acids maleic acid, fumaric acid and other aliphatic dicarboxylic acids.
- the bifunctional thiol compound any compound having two mercapto groups in the molecule may be used. 3-mercaptobutyryloxy)butane and the like.
- the method for producing the welded film of the present embodiment is not particularly limited, but for example, a resin composition obtained by heating and mixing the bifunctional epoxy resin and the compound having a bifunctional hydroxyl group, or A resin composition having a solid content of 95% by mass or more obtained by dissolving the bifunctional epoxy resin and the compound having a bifunctional hydroxyl group in a solvent, and optionally removing the solvent from the resulting mixture.
- a method of obtaining a welded film made of a phenoxy resin by heating and compressing, and the like can be mentioned. According to the manufacturing method, a welded film made of a phenoxy resin can be easily manufactured.
- the Mz and the ratio [Mz/Mn] of the phenoxy resin can be easily adjusted to the above values or more.
- solid content refers to the component in a composition except volatile components, such as a solvent.
- the catalyst may be added to the solvent at the same time as the bifunctional epoxy compound and the compound having a bifunctional hydroxyl group, and the bifunctional epoxy resin and the compound having a bifunctional hydroxyl group are You may add after melt
- Preferred solvents are methyl ethyl ketone, cyclohexanone, toluene, xylene, acetone, and the like.
- Examples of the method of heat-compressing the resin composition include a method of compressing with a heated press (heat press) or the like.
- the temperature of the hot press is preferably 120 to 250°C, more preferably 130 to 200°C, still more preferably 140 to 180°C.
- the compression time when compressing the resin composition by hot pressing is preferably 0.5 to 4 hours, more preferably 0.5 to 3 hours, and still more preferably 0.5 to 2 hours. is.
- the thickness of the welding film of the present embodiment is preferably 1-1000 ⁇ m, more preferably 10-800 ⁇ m, and still more preferably 20-500 ⁇ m.
- the welded film of the present embodiment preferably has a tensile shear strength of 10 MPa or more, and more preferably, when welded between the first substrate and the second substrate in a test according to JIS K 6850:1999. is 13 MPa or more. Specifically, the tensile shear strength can be measured by the method described in Examples.
- FIG. 1 is an explanatory diagram showing the configuration of a joined body in one embodiment (this embodiment) of the present invention.
- the joined body 10 of this embodiment is formed by welding a welding film 1 between a first base material 2 and a second base material 3 .
- the welding method include at least one method selected from the group consisting of heating, hot press, ultrasonic welding, and high-frequency induction welding. Among them, hot press, ultrasonic welding, and high-frequency induction welding are preferred.
- the first base material and the second base material are both aluminum, iron, fiber reinforced plastic (FRP), glass, ceramic, polypropylene, polycarbonate, polymethyl methacrylate, polyetherimide, polyamide, and polybutylene terephthalate. It is preferably composed of at least one selected, and more preferably composed of at least one selected from aluminum, iron, polycarbonate, and polybutylene terephthalate.
- the first base material and the second base material may be made of the same material or may be made of different materials.
- Both the first base material and the second base material are preferably subjected to surface pretreatment for the purpose of removing surface contaminants and/or for anchoring effect.
- pretreatment include degreasing treatment, UV ozone treatment, blasting treatment, polishing treatment, plasma treatment, corona discharge treatment, laser treatment, etching treatment, and flame treatment.
- the pretreatment is preferably a pretreatment for washing the surface of the substrate or a pretreatment for roughening the surface.
- the substrate is made of aluminum, glass, ceramic, or iron, at least one selected from the group consisting of degreasing treatment, UV ozone treatment, blasting treatment, polishing treatment, plasma treatment, and etching treatment is preferable.
- the substrate is made of FRP, polypropylene, polycarbonate, polymethyl methacrylate, polyetherimide, polyamide, or polybutylene terephthalate, from the group consisting of degreasing treatment, UV ozone treatment, blasting treatment, polishing treatment, plasma treatment and corona discharge treatment At least one selected is preferred. Only one type of pretreatment may be used, or two or more types may be applied. As specific methods for these pretreatments, known methods can be used. Usually, hydroxyl groups derived from resins or reinforcing materials exist on the surface of FRP, and it is thought that hydroxyl groups originally exist on the surface of glass and ceramics. can increase the hydroxyl group of
- the degreasing treatment is a method of removing dirt such as oil on the base material surface by dissolving it with an organic solvent such as acetone or toluene.
- the UV ozone treatment is a method of cleaning or modifying a surface by using the energy of short-wave ultraviolet rays emitted from a low-pressure mercury lamp and the power of ozone (O 3 ) generated thereby.
- O 3 ozone
- glass it is one of the surface cleaning methods for removing organic impurities on the surface.
- Cleaning and surface modification equipment using low-pressure mercury lamps are generally called “UV ozone cleaners”, “UV cleaning equipment”, “ultraviolet surface modification equipment” and the like.
- blasting examples include wet blasting, shot blasting, sandblasting, and the like. Among them, wet blasting is preferable because a more dense surface can be obtained than dry blasting.
- polishing treatment examples include buffing using an abrasive cloth, roll polishing using abrasive paper (sandpaper), electrolytic polishing, and the like.
- the corona discharge treatment includes a method applied to surface modification of a polymer film, in which electrons emitted from an electrode cut the polymer main chain or side chain of the polymer surface layer and generate radicals as starting points. It is a method of generating hydroxyl groups or polar groups on the surface.
- the laser treatment is a technique for improving surface characteristics by rapidly heating and cooling only the surface of the base material by laser irradiation, and is an effective method for roughening the surface.
- Known laser processing techniques can be used.
- etching treatment examples include chemical etching treatment such as alkali method, phosphoric acid-sulfuric acid method, fluoride method, chromic acid-sulfuric acid method, iron salt method; electrochemical etching treatment such as electrolytic etching method. be done.
- the flame treatment is a method of burning a mixed gas of combustion gas and air to convert oxygen in the air into plasma, and applying oxygen plasma to the object to be treated to make the surface hydrophilic.
- Known frame processing techniques can be used.
- Epomic R140P Mitsubishi Chemicals, Inc., bisphenol A type epoxy resin, molecular weight: about 378, 1.0 equivalent (189 g), bisphenol 0.95 equivalents (119 g) of S (molecular weight 250) and 0.8 g of triphenylphosphine were charged, and the temperature was raised to 100° C. while stirring in a nitrogen atmosphere. Dissolution was visually confirmed, and a resin composition (EP-5) having a solid content of 100% by mass was obtained.
- Epomic R140P (Mitsui Chemicals, Inc., bisphenol A type epoxy resin, molecular weight: about 378) 1.0 equivalent (189 g), bisphenol 0.95 equivalent (119 g) of S (molecular weight 250), 0.8 g of triphenylphosphine, and 1232 g of cyclohexanone were charged, and the temperature was raised to 170° C. under a nitrogen atmosphere for reaction. After reacting for 4 hours, it was confirmed by GPC that the bisphenol S peak had disappeared, and the reaction was terminated to obtain a phenoxy resin having a solid content of 25% by mass. By removing the solvent from this, a phenoxy resin (EP-8) having a solid content of 100% by mass was obtained.
- Thickness of Welded Film The thickness of the obtained welded film was measured using MDC-25MX manufactured by Mitutoyo Corporation after being left in an atmosphere of 23° C. and 50% humidity for 24 hours.
- Bonding test specimen-2 was prepared in the same manner as bonding test specimen-1 described above, except that aluminum A6061-T6 having a size of 25 mm ⁇ 100 mm and a thickness of 1.6 mm was used instead of the cold-rolled steel plate.
- the surface of aluminum A6061-T6 having a size of 25 mm ⁇ 100 mm and a thickness of 1.6 mm was blasted for 10 minutes, and then the blasted surface was degreased with methyl ethyl ketone.
- a welding film is placed on the degreased surface of the aluminum plate, and the polycarbonate plate is overlapped and fixed so that the overlapping length of the joint is 12.5 mm and the width is 25 mm.
- Industrial Co., Ltd., oscillator UH-2.5K, press JIIP30S) was used to heat the metal by high-frequency induction, and the aluminum plate and the polycarbonate plate were joined by heating and pressure to prepare a joining test specimen-3.
- the applied pressure was 110 N (pressure 2.2 MPa), the oscillation frequency was 900 kHz, and the oscillation time was 5 seconds.
- Example 1 to 8 in which a welded film made of a phenoxy resin having a Mz of 70,000 or more and a ratio [Mz/Mn] of 5.0 or more was used, it was difficult to handle because it had strength enough to be released from the mold. It can be seen that the weld strength is excellent, and the tensile shear strength is as high as 10 MPa or more.
- Joined bodies using the welding film of the present embodiment include, for example, door side panels, bonnet roofs, tailgates, steering hangers, A pillars, B pillars, C pillars, D pillars, crash boxes, and power control unit (PCU) housings. , electric compressor parts (inner wall, intake port, exhaust control valve (ECV) insertion part, mount boss, etc.), lithium ion battery (LIB) spacer, battery case, automotive parts such as LED headlamps, smartphones, It is used as a notebook computer, a tablet computer, a smart watch, a large liquid crystal television (LCD-TV), an outdoor LED lighting structure, etc., but is not particularly limited to these examples.
- PCU power control unit
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Mechanical Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Health & Medical Sciences (AREA)
- Polymers & Plastics (AREA)
- Medicinal Chemistry (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Ceramic Engineering (AREA)
- Adhesives Or Adhesive Processes (AREA)
- Lining Or Joining Of Plastics Or The Like (AREA)
- Epoxy Resins (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
- Laminated Bodies (AREA)
Abstract
Description
[1]フェノキシ樹脂からなる溶着フィルムであって、前記フェノキシ樹脂のz平均分子量が70,000以上であり、前記フェノキシ樹脂のz平均分子量と数平均分子量との比〔Mz/Mn〕が5.0以上である、溶着フィルム。
[2]前記溶着フィルムは厚さが1~1000μmである、上記[1]に記載の溶着フィルム。
[3]前記溶着フィルムを、第一基材と第二基材との間に溶着させた際の引張剪断強度が、JIS K 6850:1999に準じた試験において10MPa以上である、上記[1]または[2]に記載の溶着フィルム。
[4]上記[1]~[3]のいずれかに記載の溶着フィルムを、第一基材と第二基材との間に溶着させてなる、接合体。
[5]前記第一基材、及び前記第二基材が、いずれもアルミニウム、鉄、繊維強化プラスチック、ガラス、セラミック、ポリプロピレン、ポリカーボネート、ポリメチルメタクリレート、ポリエーテルイミド、ポリアミド、及びポリブチレンテレフタレートより選ばれる少なくとも1種からなる、上記[4]に記載の接合体。
[6]溶着フィルムを、加熱、熱プレス、超音波溶着、及び高周波誘導溶着からなる群より選ばれる少なくとも1種の方法で、第一基材及び第二基材との間に溶着させる、上記[4]または[5]に記載の接合体の製造方法。
本明細書において、接合とは、物と物とを繋合わせることを意味し、接着及び溶着はその下位概念である。接着とは、テープ及び接着剤の様な有機材(熱硬化性樹脂及び熱可塑性樹脂等)を介して、2つの被着材(接着しようとするもの)を接合状態とすることを意味する。
本実施形態の溶着フィルムは、フェノキシ樹脂からなり、該フェノキシ樹脂のz平均分子量が70,000以上であり、前記フェノキシ樹脂のz平均分子量と数平均分子量との比〔Mz/Mn〕が5.0以上であることを特徴とする。
なお、本明細書において、前記Mz、後述するMn及びMwは、細孔を有するカラムを用いて分子サイズの違いを分離する装置を用いる手法により測定することができ、具体的には実施例に記載の方法により測定することができる。
前記フェノキシ樹脂は、直鎖状の比較的低分子量の樹脂を含んでいてもよい。該低分子量の樹脂を含むことにより、溶着フィルムの被着材への濡れ性を高めることができる。
特にビスフェノールA型エポキシ樹脂が好ましく、その分子量は好ましくは250~6,000であり、より好ましくは300~6,000であり、更に好ましくは700~5,000である。前記分子量がこの範囲内であると、フィルム成形性が良好となる。
前記触媒の使用量は、特に制限はないが、2官能エポキシ化合物と2官能の水酸基を有する化合物との合計100質量部に対して、好ましくは0.01~10.00質量部であり、より好ましくは0.10~1.00質量部であり、更に好ましくは0.15~0.50質量部である。
本実施形態の溶着フィルムの製造方法は、特に限定されるものではないが、例えば、前記2官能エポキシ樹脂、及び前記2官能の水酸基を有する化合物を加熱混合して得られた樹脂組成物、もしくは前記2官能エポキシ樹脂、及び前記2官能の水酸基を有する化合物を溶媒に溶解し、得られた混合物から必要に応じて前記溶媒を除去して得られた固形分95質量%以上の樹脂組成物を加熱圧縮してフェノキシ樹脂からなる溶着フィルムを得る方法等が挙げられる。
前記製造方法によれば、フェノキシ樹脂からなる溶着フィルムを簡便に製造することができる。また、フェノキシ樹脂のMz及び比〔Mz/Mn〕をそれぞれ前述の値以上にしやすくなる。
なお、本明細書において「固形分」とは、溶媒等の揮発成分を除いた組成物中の成分を指す。
溶媒としては、メチルエチルケトン、シクロヘキサノン、トルエン、キシレン、アセトン等が好ましい。
前記熱プレスの温度は、好ましくは120~250℃であり、より好ましくは130~200℃であり、更に好ましくは140~180℃である。
また、前記樹脂組成物を熱プレスで圧縮する際の圧縮時間は、好ましくは0.5~4時間であり、より好ましくは0.5~3時間であり、更に好ましくは0.5~2時間である。
前記引張せん断強度は、具体的には実施例に記載の方法により測定することができる。
図1は、本発明の一実施形態(本実施形態)における接合体の構成を示す説明図である。図1に示すように、本実施形態の接合体10は、溶着フィルム1を第一基材2と第二基材3との間に溶着させてなる。前記溶着方法としては、加熱、熱プレス、超音波溶着、及び高周波誘導溶着からなる群より選ばれる少なくとも1種の方法が挙げられる。中でも、熱プレス、超音波溶着、高周波誘導溶着が好ましい。
前記第一基材、及び前記第二基材は、同じ材料からなるものでもよく、異なる材料からなるものでもよい。
前処理としては、例えば、脱脂処理、UVオゾン処理、ブラスト処理、研磨処理、プラズマ処理、コロナ放電処理、レーザー処理、エッチング処理、フレーム処理等が挙げられる。
前処理としては、基材の表面を洗浄する前処理または表面に凹凸を付ける前処理が好ましい。具体的には、基材がアルミニウム、ガラス、セラミック、又は鉄からなる場合、脱脂処理、UVオゾン処理、ブラスト処理、研磨処理、プラズマ処理、エッチング処理からなる群より選ばれる少なくとも1種が好ましく、基材がFRP、ポリプロピレン、ポリカーボネート、ポリメチルメタクリレート、ポリエーテルイミド、ポリアミド、又はポリブチレンテレフタレートからなる場合、脱脂処理、UVオゾン処理、ブラスト処理、研磨処理、プラズマ処理及びコロナ放電処理からなる群より選ばれる少なくとも1種が好ましい。
前処理は、1種のみであってもよく、2種以上を施してもよい。これらの前処理の具体的な方法としては、公知の方法を用いることができる。
通常、FRPの表面には樹脂又は補強材に由来する水酸基が存在し、ガラス及びセラミック表面には元々水酸基が存在すると考えられるが、前記の前処理によって新たに水酸基が生成され、基材の表面の水酸基を増やすことができる。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)1.0等量(12.5g)、トリフェニルホスフィン0.8g、及びメチルエチルケトン400gを仕込み、窒素雰囲気下で撹拌しながら100℃まで昇温した。目視で溶解したことを確認し、40℃まで冷却して固形分35質量%の樹脂組成物を得た。これから溶剤を除去して固形分100質量%の樹脂組成物(EP-1)を得た。
撹拌拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)0.8等量(10.0g)、トリフェニルホスフィン0.8g、及びメチルエチルケトン397gを仕込み、窒素雰囲気下で撹拌しながら100℃まで昇温した。目視で溶解したことを確認し、40℃まで冷却して固形分35質量%の樹脂組成物を得た。これから溶剤を除去して固形分100質量%の樹脂組成物(EP-2)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)0.5等量(6.3g)、トリフェニルホスフィン0.8g、及びメチルエチルケトン390gを仕込み、窒素雰囲気下で撹拌しながら100℃まで昇温した。目視で溶解したことを確認し、40℃まで冷却して固形分35質量%の樹脂組成物を得た。これから溶剤を除去して固形分100質量%の樹脂組成物(EP-3)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1001(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約900)1.0等量(270g)、ビスフェノールS(分子量250)0.95等量(71.3g)、トリフェニルホスフィン0.8g、及びメチルエチルケトン515gを仕込み、窒素雰囲気下で撹拌しながら100℃まで昇温した。目視で溶解したことを確認し、40℃まで冷却して固形分35質量%の樹脂組成物を得た。これから溶剤を除去して固形分100質量%の樹脂組成物(EP-4)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、エポミックR140P(三井化学株式会社製、ビスフェノールA型エポキシ樹脂、分子量約378)1.0等量(189g)、ビスフェノールS(分子量250)0.95等量(119g)、トリフェニルホスフィン0.8g、を仕込み、窒素雰囲気下で撹拌しながら100℃まで昇温した。目視で溶解したことを確認し、固形分100質量%の樹脂組成物(EP-5)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)0.25等量(3.1g)、トリフェニルホスフィン0.8g、及びメチルエチルケトン390gを仕込み、窒素雰囲気下で撹拌しながら100℃まで昇温した。目視で溶解したことを確認し、40℃まで冷却して固形分35質量%の樹脂組成物を得た。これから溶剤を除去して固形分100質量%の樹脂組成物(EP-6)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)0.95等量(11.8g)、トリフェニルホスフィン0.8g、及びシクロヘキサノン198gを仕込み、窒素雰囲気下で170℃まで昇温して反応させた。6.5時間反応後、ゲルパーミエーションクロマトグラフィー(GPC)でビスフェノールSのピークが消失したことを確認して反応を終了し、固形分52質量%のフェノキシ樹脂を得た。これから溶剤を除去して固形分100質量%のフェノキシ樹脂(EP-7)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、エポミックR140P(三井化学株式会社製、ビスフェノールA型エポキシ樹脂、分子量約378)1.0等量(189g)、ビスフェノールS(分子量250)0.95等量(119g)、トリフェニルホスフィン0.8g、及びシクロヘキサノン1232gを仕込み、窒素雰囲気下で170℃まで昇温して反応させた。4時間反応後、GPCでビスフェノールSのピークが消失したことを確認して反応を終了し、固形分25質量%のフェノキシ樹脂を得た。これから溶剤を除去して固形分100質量%のフェノキシ樹脂(EP-8)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)0.95等量(11.8g)、トリフェニルホスフィン0.8g、及びシクロヘキサノン400gを仕込み、窒素雰囲気下で170℃まで昇温して反応させた。6.5時間反応後、GPCでビスフェノールSのピークが消失したことを確認して反応を終了し、固形分35質量%のフェノキシ樹脂を得た。これから溶剤を除去して固形分100質量%のフェノキシ樹脂(EP-9)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)0.95等量(11.8g)、トリフェニルホスフィン7.2g、及びシクロヘキサノン200gを仕込み、窒素雰囲気下で170℃まで昇温して反応させた。4時間反応後、GPCでビスフェノールSのピークが消失したことを確認して反応を終了し、固形分52質量%のフェノキシ樹脂を得た。これから溶剤を除去して固形分100質量%のフェノキシ樹脂(EP-10)を得た。
140℃に昇温した2L混和機3011(高林理化株式会社製)に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(939g)を仕込み、低速で撹拌しながら溶解した。溶解後、ビスフェノールS(分子量250)0.95等量(56.3g)を4回に分けて投入し、140℃を維持した状態でビスフェノールSが溶解するまで回転数30rpmで撹拌を行った。ビスフェノールSの溶解を目視で確認後、トリフェニルホスフィン3.8gを投入し、温度140℃、回転数30rpmの条件で45分間撹拌し、固形分100質量%の樹脂組成物(EP-11)を得た。
140℃に昇温した2L混和機3011(高林理化株式会社製)に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(939g)を仕込み、低速で撹拌しながら溶解した。溶解後、ビスフェノールS(分子量250)1.0等量(57.5g)を4回に分けて投入し、140℃を維持した状態でビスフェノールSが溶解するまで回転数30rpmで撹拌を行った。ビスフェノールSの溶解を目視で確認後、トリフェニルホスフィン3.8gを投入し、温度160℃、回転数30rpmの条件で80分間撹拌し、固形分100質量%の樹脂組成物(EP-12)を得た。
撹拌機、環流冷却器、ガス導入管、及び温度計を付した反応装置に、jER(登録商標)1007(三菱ケミカル株式会社製、ビスフェノールA型エポキシ樹脂、分子量約4060)1.0等量(203g)、ビスフェノールS(分子量250)0.5等量(6.3g)、BRG-555 0.5等量(5.2g)(アイカ工業株式会社製、ノボラック系フェノール樹脂)、トリフェニルホスフィン0.8g、及びシクロヘキサノン400gを仕込み、窒素雰囲気下で170℃まで昇温して反応させた。6.5時間反応後、GPCでビスフェノールSのピークが消失したことを確認して反応を終了し、固形分35質量%のフェノキシ樹脂を得た。これから溶剤を除去して固形分100質量%のフェノキシ樹脂(EP-13)を得た。
〔溶着フィルムの作製〕
プレス機の上板及び下板に非粘着フッ素樹脂フィルム(ニトフロン(登録商標)No.900UL、日東電工株式会社製)を設置し、下板の非粘着フッ素樹脂フィルム上に製造例1~13で得られた樹脂組成物又はフェノキシ樹脂を配置した後、前記プレス機を160℃に加熱し、前記樹脂組成物又はフェノキシ樹脂を2時間加熱圧縮して厚みが50μmとなる溶着フィルムを作製した。得られた溶着フィルムを用い、以下の評価を実施した。結果を表1に示す。
(1)溶着フィルムの保形性
評価は溶着フィルムが成形できたものを「A」、やや脆さが見られたが離型でき、溶着フィルムが得られたものを「B」、溶着フィルムとして離形できなかったものを「C」とした。
各実施例及び比較例で作製した溶着フィルムをテトラヒドロフランに溶解し、Prominence 501(昭和サイエンス株式会社製、Detector:Shodex(登録商標) RI-501(昭和電工株式会社製))を用い、以下の条件で測定した。
カラム:昭和電工製 LF-804×2本
カラム温度:40℃
試料:重合体の0.4質量%テトラヒドロフラン溶液
流量:1ml/分
溶離液:テトラヒドロフラン
得られた溶着フィルムの厚みは23℃、湿度50%の雰囲気中に24時間放置後、株式会社ミツトヨ製のMDC-25MXを用いて測定した。
〔接合試験体-1(鋼板/溶着フィルム/鋼板)の作製〕
25mm×100mm、厚さ3mmの冷延鋼板の表面を10分間ブラスト処理後、ブラスト面をメチルエチルケトンで脱脂した。脱脂した面に溶着フィルムを置き、同様の処理を行った冷延鋼板を、接合部の重なり長さ12.5mm、幅25mmとなるように重ね、クリップで固定した。固定した状態で160℃雰囲気に2時間放置して溶着させ、接合試験体-1を作製した。ここで接合部とは、試験体用基材を重ね合わせた箇所を意味する。
冷延鋼板の代わりに25mm×100mm、厚さ1.6mmのアルミニウム A6061-T6を用いたこと以外は前述の接合試験体-1の作製と同じ方法で接合試験体-2を作製した。
ポリカーボネート 121R(SABIC社製)を、射出成形機(住友重機械工業株式会社製 SE100V)を用いて、シリンダー温度290℃、金型温度85℃、射出速度30mm/秒、保圧70MPaで3秒と50MPaで2秒、冷却時間13秒の条件で射出成型して、25mm×100mm、厚さ2mmのポリカーボネート板を得た。
試験体用基材として、得られたポリカーボネート板、及びアルミニウム板を用意した。アルミニウム板は、25mm×100mm、厚さ1.6mmのアルミニウム A6061-T6の表面を10分間ブラスト処理後、ブラスト面をメチルエチルケトンで脱脂した。
アルミニウム板の脱脂した面に溶着フィルムを置き、ポリカーボネート板を、接合部の重なり長さが12.5mm、幅25mmとなるように重ねて固定し、固定部を高周波誘導溶着機(精電舎電子工業株式会社製、発振器UH-2.5K、プレスJIIP30S)を用いて高周波誘導により金属を発熱させ、加熱・加圧によりアルミニウム板及びポリカーボネート板を接合し、接合試験体-3を作製した。加圧力は110N(圧力2.2MPa)、発振周波数は900kHz、発振時間は5秒とした。
1 溶着フィルム
2 第一基材
3 第二基材
Claims (6)
- フェノキシ樹脂からなる溶着フィルムであって、前記フェノキシ樹脂のz平均分子量が70,000以上であり、前記フェノキシ樹脂のz平均分子量と数平均分子量との比〔Mz/Mn〕が5.0以上である、溶着フィルム。
- 前記溶着フィルムは厚さが1~1000μmである、請求項1に記載の溶着フィルム。
- 前記溶着フィルムを、第一基材と第二基材との間に溶着させた際の引張剪断強度が、JIS K 6850:1999に準じた試験において10MPa以上である、請求項1または2に記載の溶着フィルム。
- 請求項1~3のいずれか1項に記載の溶着フィルムを、第一基材と第二基材との間に溶着させてなる、接合体。
- 前記第一基材、及び前記第二基材が、いずれもアルミニウム、鉄、繊維強化プラスチック、ガラス、セラミック、ポリプロピレン、ポリカーボネート、ポリメチルメタクリレート、ポリエーテルイミド、ポリアミド、及びポリブチレンテレフタレートより選ばれる少なくとも1種からなる、請求項4に記載の接合体。
- 溶着フィルムを、加熱、熱プレス、超音波溶着、及び高周波誘導溶着からなる群より選ばれる少なくとも1種の方法で、第一基材及び第二基材との間に溶着させる、請求項4または5に記載の接合体の製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020237027233A KR20230131902A (ko) | 2021-02-10 | 2022-02-04 | 용착 필름 및 접합체 |
| EP22752690.2A EP4292811A1 (en) | 2021-02-10 | 2022-02-04 | Welding film and joined body |
| JP2022580602A JP7414160B2 (ja) | 2021-02-10 | 2022-02-04 | 溶着フィルム及び接合体 |
| CN202280013961.9A CN116829664A (zh) | 2021-02-10 | 2022-02-04 | 熔接膜和接合体 |
| US18/276,510 US20240117228A1 (en) | 2021-02-10 | 2022-02-04 | Welding film and joined body |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-020100 | 2021-02-10 | ||
| JP2021020100 | 2021-02-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022172863A1 true WO2022172863A1 (ja) | 2022-08-18 |
Family
ID=82837894
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/004352 WO2022172863A1 (ja) | 2021-02-10 | 2022-02-04 | 溶着フィルム及び接合体 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20240117228A1 (ja) |
| EP (1) | EP4292811A1 (ja) |
| JP (1) | JP7414160B2 (ja) |
| KR (1) | KR20230131902A (ja) |
| CN (1) | CN116829664A (ja) |
| WO (1) | WO2022172863A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2023127667A1 (ja) * | 2021-12-27 | 2023-07-06 | ||
| JPWO2023127668A1 (ja) * | 2021-12-27 | 2023-07-06 | ||
| JP7485005B1 (ja) | 2022-12-19 | 2024-05-16 | 株式会社レゾナック | 接合体の製造方法、接合体、及び電気電子部品 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007277333A (ja) * | 2006-04-04 | 2007-10-25 | Dainippon Ink & Chem Inc | 樹脂組成物、フェノキシ樹脂、塗料組成物、接着剤組成物、接着フィルム、プリプレグ、多層プリント配線基板及び樹脂付銅箔 |
| JP2010126694A (ja) * | 2008-11-28 | 2010-06-10 | Nagase Chemtex Corp | 縮環構造含有フェノキシ樹脂 |
| JP2017119779A (ja) * | 2015-12-28 | 2017-07-06 | 新日鉄住金化学株式会社 | ビスフェノールf骨格含有フェノキシ樹脂、その製造方法、及びそれを用いた樹脂組成物 |
| JP2017171802A (ja) * | 2016-03-24 | 2017-09-28 | 新日鉄住金化学株式会社 | ビスフェノールf骨格含有フェノキシ樹脂、その製造方法、及びそれを用いた樹脂組成物 |
| JP2019147863A (ja) * | 2018-02-26 | 2019-09-05 | 日鉄ケミカル&マテリアル株式会社 | リン含有フェノキシ樹脂、その樹脂組成物、及び硬化物 |
| JP2020100728A (ja) * | 2018-12-21 | 2020-07-02 | 日鉄ケミカル&マテリアル株式会社 | フェノキシ樹脂、その樹脂組成物、その硬化物、およびその製造方法。 |
| JP2020125471A (ja) * | 2019-02-06 | 2020-08-20 | 日鉄ケミカル&マテリアル株式会社 | フェノキシ樹脂及びその製造方法、その樹脂組成物並びに硬化物 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7132048B2 (ja) | 2018-09-13 | 2022-09-06 | 藤森工業株式会社 | ホットメルト接着樹脂組成物及びホットメルト接着樹脂積層体 |
| JPWO2022014503A1 (ja) * | 2020-07-13 | 2022-01-20 | ||
| WO2022209116A1 (ja) * | 2021-03-31 | 2022-10-06 | 日鉄ケミカル&マテリアル株式会社 | 接着用樹脂組成物、接着フィルム及び接着構造体 |
-
2022
- 2022-02-04 EP EP22752690.2A patent/EP4292811A1/en active Pending
- 2022-02-04 US US18/276,510 patent/US20240117228A1/en active Pending
- 2022-02-04 WO PCT/JP2022/004352 patent/WO2022172863A1/ja active Application Filing
- 2022-02-04 JP JP2022580602A patent/JP7414160B2/ja active Active
- 2022-02-04 CN CN202280013961.9A patent/CN116829664A/zh active Pending
- 2022-02-04 KR KR1020237027233A patent/KR20230131902A/ko unknown
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007277333A (ja) * | 2006-04-04 | 2007-10-25 | Dainippon Ink & Chem Inc | 樹脂組成物、フェノキシ樹脂、塗料組成物、接着剤組成物、接着フィルム、プリプレグ、多層プリント配線基板及び樹脂付銅箔 |
| JP2010126694A (ja) * | 2008-11-28 | 2010-06-10 | Nagase Chemtex Corp | 縮環構造含有フェノキシ樹脂 |
| JP2017119779A (ja) * | 2015-12-28 | 2017-07-06 | 新日鉄住金化学株式会社 | ビスフェノールf骨格含有フェノキシ樹脂、その製造方法、及びそれを用いた樹脂組成物 |
| JP2017171802A (ja) * | 2016-03-24 | 2017-09-28 | 新日鉄住金化学株式会社 | ビスフェノールf骨格含有フェノキシ樹脂、その製造方法、及びそれを用いた樹脂組成物 |
| JP2019147863A (ja) * | 2018-02-26 | 2019-09-05 | 日鉄ケミカル&マテリアル株式会社 | リン含有フェノキシ樹脂、その樹脂組成物、及び硬化物 |
| JP2020100728A (ja) * | 2018-12-21 | 2020-07-02 | 日鉄ケミカル&マテリアル株式会社 | フェノキシ樹脂、その樹脂組成物、その硬化物、およびその製造方法。 |
| JP2020125471A (ja) * | 2019-02-06 | 2020-08-20 | 日鉄ケミカル&マテリアル株式会社 | フェノキシ樹脂及びその製造方法、その樹脂組成物並びに硬化物 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2023127667A1 (ja) * | 2021-12-27 | 2023-07-06 | ||
| JPWO2023127668A1 (ja) * | 2021-12-27 | 2023-07-06 | ||
| JP7491479B2 (ja) | 2021-12-27 | 2024-05-28 | 株式会社レゾナック | 接合体の製造方法 |
| JP7485005B1 (ja) | 2022-12-19 | 2024-05-16 | 株式会社レゾナック | 接合体の製造方法、接合体、及び電気電子部品 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20240117228A1 (en) | 2024-04-11 |
| JPWO2022172863A1 (ja) | 2022-08-18 |
| EP4292811A1 (en) | 2023-12-20 |
| KR20230131902A (ko) | 2023-09-14 |
| TW202235564A (zh) | 2022-09-16 |
| JP7414160B2 (ja) | 2024-01-16 |
| CN116829664A (zh) | 2023-09-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2022172863A1 (ja) | 溶着フィルム及び接合体 | |
| TWI838691B (zh) | 熔接薄膜及接合體 | |
| JP6923762B1 (ja) | 複合積層体及び、接合体 | |
| WO2022130833A1 (ja) | 金属部材-樹脂部材接合体の製造方法及びフィルム | |
| US20230330946A1 (en) | Method for joining metal and resin, and joined body thereof | |
| JP6923764B1 (ja) | 複合積層体及び、接合体 | |
| JP6923721B1 (ja) | プライマー付き基材及びその製造方法、並びに接合体 | |
| JP7491480B2 (ja) | 接合体の製造方法及び基材の製造方法 | |
| WO2023127667A1 (ja) | 接合体の製造方法 | |
| WO2023127666A1 (ja) | 接合体の製造方法 | |
| JP7491479B2 (ja) | 接合体の製造方法 | |
| TWI835487B (zh) | 接合體之製造方法 | |
| JP7485227B1 (ja) | 接合体の製造方法、接合体、及び電気電子部品 | |
| JP6923763B1 (ja) | 複合積層体及び接合体 | |
| JP6923706B1 (ja) | プライマー付材料及び接合体 | |
| JP6923707B1 (ja) | プライマー付材料及び接合体 | |
| JP6918894B2 (ja) | 複合積層体及び、金属―ポリアミド系樹脂接合体 | |
| WO2022270613A1 (ja) | 接合体の製造方法 | |
| JP2023003079A (ja) | 熱溶着性フィルム及びそれを用いた接合体 | |
| JP2022083664A (ja) | 接合体の製造方法 | |
| JP2022102952A (ja) | 複合積層体及びその製造方法、並びに複合積層体を使用した接合体及びその製造方法 | |
| JP2023096713A (ja) | 車載カメラの製造方法 | |
| JP2023096624A (ja) | 車載用ecuの製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22752690 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2022580602 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280013961.9 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18276510 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20237027233 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020237027233 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022752690 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022752690 Country of ref document: EP Effective date: 20230911 |