WO2006022420A1 - 腹圧性尿失禁の予防・治療剤及びそのスクリーニング方法 - Google Patents
腹圧性尿失禁の予防・治療剤及びそのスクリーニング方法 Download PDFInfo
- Publication number
- WO2006022420A1 WO2006022420A1 PCT/JP2005/015830 JP2005015830W WO2006022420A1 WO 2006022420 A1 WO2006022420 A1 WO 2006022420A1 JP 2005015830 W JP2005015830 W JP 2005015830W WO 2006022420 A1 WO2006022420 A1 WO 2006022420A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pressure
- urinary incontinence
- substance
- receptor
- serotonin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
- A61K31/5517—1,4-Benzodiazepines, e.g. diazepam or clozapine condensed with five-membered rings having nitrogen as a ring hetero atom, e.g. imidazobenzodiazepines, triazolam
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
- A61K31/136—Amines having aromatic rings, e.g. ketamine, nortriptyline having the amino group directly attached to the aromatic ring, e.g. benzeneamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
- A61K31/137—Arylalkylamines, e.g. amphetamine, epinephrine, salbutamol, ephedrine or methadone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/38—Heterocyclic compounds having sulfur as a ring hetero atom
- A61K31/381—Heterocyclic compounds having sulfur as a ring hetero atom having five-membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/397—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having four-membered rings, e.g. azetidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/4025—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil not condensed and containing further heterocyclic rings, e.g. cromakalim
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
- A61K31/4045—Indole-alkylamines; Amides thereof, e.g. serotonin, melatonin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
- A61K31/416—1,2-Diazoles condensed with carbocyclic ring systems, e.g. indazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/437—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a five-membered ring having nitrogen as a ring hetero atom, e.g. indolizine, beta-carboline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4965—Non-condensed pyrazines
- A61K31/497—Non-condensed pyrazines containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4985—Pyrazines or piperazines ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5383—1,4-Oxazines, e.g. morpholine ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/54—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame
- A61K31/542—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/565—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol
- A61K31/568—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids not substituted in position 17 beta by a carbon atom, e.g. estrane, estradiol substituted in positions 10 and 13 by a chain having at least one carbon atom, e.g. androstanes, e.g. testosterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/02—Drugs for disorders of the urinary system of urine or of the urinary tract, e.g. urine acidifiers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/10—Drugs for disorders of the urinary system of the bladder
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/24—Drugs for disorders of the endocrine system of the sex hormones
- A61P5/26—Androgens
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5082—Supracellular entities, e.g. tissue, organisms
- G01N33/5088—Supracellular entities, e.g. tissue, organisms of vertebrates
Definitions
- the present invention relates to a stress urinary incontinence prevention / therapeutic agent and a stress pressure urinary incontinence prevention / therapeutic drug screening method comprising a substance that increases the leakage pressure when abdominal pressure increases.
- Stress urinary incontinence is a disease characterized by symptoms of urine leaking when the abdominal pressure rises transiently due to light exercise such as coughing, sneezing, sneezing or climbing stairs or heavy luggage. It is a disorder of the urinary restraint mechanism. This disease is more common in women and is said to occur because the pelvic floor muscles are weakened by childbirth and aging, and the anatomical location of organs in the pelvic floor, including the bladder and urethra, changes (for example, " The J our na 1 of Family Paratice ", 1982, pp. 14, 935-936).
- urinary leakage is avoided by the reflex increase in urethral pressure that antagonizes the increase in intravesical pressure that accompanies a rapid increase in abdominal pressure.
- a disorder related to the urinary restraint mechanism such as a neuronal circuit related to reflexes or a decrease in muscle group contraction force related to an increase in urethral pressure
- the bladder associated with a sudden increase in abdominal pressure Increased internal pressure increases urethral pressure reflexes and urine leakage Get up. Therefore, in order to screen for therapeutic agents for stress urinary incontinence, an evaluation system that reflects such pathological conditions is important.
- a model rat is created by vaginal dilation in the virgin state, ovariectomy, or a combination of these (for example, "Ur olo gy”, 1 998, Vol. 52, p. 143— 1 5 1, and “The Journal of Urology”, 2001, 1 166, p. 3 1 1-3 1 7).
- Leak point pressure indicating the urethral resistance in the urine collection phase is used as an evaluation item used to diagnose stress urinary incontinence in clinical practice.
- the most common method is to stimulate the abdominal wall to increase intravesical pressure or to stimulate the nasal mucous membrane with a sputum to sneeze and observe the presence of urine leakage (eg "Ur olo gy”, 1 998, 52, p. 143— 1 51, and “The Journal of Urology”, 20 01, 1 66, p. 3 1 1-3 1 7).
- a screening method for a preventive / therapeutic drug for stress urinary incontinence characterized in that the reflex contractile force of the pelvic floor muscles in small mammals is measured by urethral pressure (Japanese Patent Laid-Open No. 20 04). -1 599 1 9).
- WO 9 9/202 79 describes that serotonin uptake inhibitors are useful for the treatment of urinary incontinence.
- Japanese Patent Application Laid-Open No. 7-188003 describes that duloxetine, a serotonin uptake inhibitor, is useful for the treatment of urge incontinence and stress incontinence. Disclosure of the invention In screening new therapeutic agents for stress urinary incontinence, it is important to construct a simple, useful and efficient in vivo evaluation system. However, no medicinal evaluation system has been established for measuring the leakage pressure due to an increase in intravesical pressure based on a sudden increase in abdominal pressure in animals, except for the sneezing pressure. Also, the pressure at the time of sneezing leak is not a simple method suitable for screening because it is difficult to induce sneezing and control the increase in intravesical pressure.
- the present inventors have electrically stimulated the abdominal wall of an anesthetized female rat to increase the intravesical pressure via increased abdominal pressure and cause urine leakage. This makes it possible to measure the leakage pressure. Since the pressure at the time of leakage due to electrical stimulation of the abdominal wall is significantly reduced by bilaterally cutting the pelvic nerve including sensory nerves from the bladder, this is an evaluation item involving reflex urethral contraction response due to bladder compression. It was found that the introduction of the evaluation items provides a new method for evaluating the in vivo efficacy of therapeutic agents for stress urinary incontinence.
- the present inventors use this in vivo drug efficacy evaluation method, etc., so that a substance that activates serotonin 5-HT 2 C receptor or androgen increases urethral resistance to prevent and treat stress urinary incontinence. I found out that I can do it. As a result of further studies based on these findings, the present inventors have completed the present invention.
- the present invention is as follows.
- Serotonin 5--prophylaxis / treatment for stress urinary incontinence comprising a substance that activates the HT 2 C receptor
- Serotonin 5-HT 2 C substance activating receptor is a serotonin 5-HT 2 c receptor Agonisuto [1] prophylactic or therapeutic agent according,
- substance activating serotonin 5-HT 2C receptor is serotonin 5-HT 2.
- An abdominal pressure characterized by increasing the abdominal pressure by electrically stimulating the animal's abdominal muscles or diaphragm or the nerves that control them, and measuring the leakage pressure (1 eak point pr ⁇ ssure)
- a screening method for substances that increase the leakage pressure when rising
- a pressure-sensitive urine characterized by increasing the abdominal pressure by electrically stimulating the animal's abdominal muscles or diaphragm or the nerves that control them, and measuring the leakage pressure (1 e ak o int pressure) at this time Screening methods for incontinence preventive drugs,
- Figure 1 shows a typical example of the effect of duloxetine on urethral contraction induced by increased intravesical pressure in urethane anesthetized female rats.
- the substance used in the preventive / therapeutic agent for stress urinary incontinence of the present invention is a substance that increases urethral resistance when abdominal pressure increases.
- Substances that increase urethral resistance when increasing abdominal pressure are, for example, leaking pressure when increasing abdominal pressure (leak po int pre s sure; urethra)
- the substance used in the preventive / therapeutic agent for stress urinary incontinence of the present invention is preferably a substance that activates serotonin 5-HT 2C receptor, a substance that stimulates an androgen binding site, or the like. Used.
- serotonin 5-HT 2C receptors for example, (including partial Agonisuto) Seroto Nin 5- HT 2C receptor Agonisuto, serotonin 5 - HT 2C receptor partial en evening agonist, among others serotonin 5- HT 2C receptor agonists (particularly fluagonists) are preferably used.
- the serotonin 5-HT 2C receptor agonist has, for example, an inhibitory activity with a 50 % inhibitory concentration (IC 50 ) of about 100 OnM or less, preferably about 10 OnM or less as determined by a binding test.
- serotonin 2C receptor agonists include EP0572863, EP0863136, EP 12 13017, USP3253989, USP3676558, USP 3652 588, USP4082844, USP4971969 USP 549492 8, USP 5646173, USP6310208, W097 / 42183, W098 / 30546, WO 98/30548, W ⁇ 98/33504, WO 99/02 159, WO 99/43647 (USP 628 1243) S WO 0 0/12475 (USP 6380238), WO 00/12502 (USP 6 365598), WO 00/125 10 (USP 6433 175), WO 00/12475 WO
- Etc. are preferably used.
- the androgen binding site refers to a site (eg, receptor) to which androgen can bind, and is not limited to the androgen receptor.
- Substances that stimulate the androgen binding site include agonists (including partial agonists) and partial antagonists.
- substances that stimulate androgen binding sites include:
- Testosterone Tes t os t erone
- Dihydrotestosterone Dihydrot es t os t erone, DH T
- Testosterone propionate (Te s t o s t e r one p r op i o n at e)
- the substance of the present invention can increase the leakage pressure when the abdominal pressure rises, mammals such as mice, rats, hams, magpies, cats, innu, sushi, sheep, monkeys, It is useful as a preventive and therapeutic agent for stress urinary incontinence that is safe and less toxic to humans.
- Stress urinary incontinence is an involuntary leakage of urine that occurs during cough, sneezing, laughing or other physical activity that increases intra-abdominal pressure in the absence of bladder contraction and is due to overactive bladder It is different from urinary urgency and urge urinary incontinence, which are symptoms of urine.
- Stress urinary incontinence is classified into normal, mild, moderate, advanced, and extremely high according to the pad test (1 hour method) proposed by the International Association for Urinary Continence. That is, after ingesting 50 Oml of water, the abdominal pressure is increased by exercising and moving for 1 hour, and the amount of incontinence during that time is measured by the weight of the pad (or disposable diaper). g), mild (2.1 g to 5.0 g), moderate (5.1 g to l 0.0 g), altitude (10.1 g to 50.0 g), very high (50.
- the substance of the present invention is useful for any stress urinary incontinence.
- the pharmaceutical composition containing the substance of the present invention may be any of solid preparations such as powders, granules, tablets, capsules and suppositories, and liquids such as syrups, emulsions, injections and suspensions.
- the pharmaceutical composition of the present invention can be produced by a conventional method such as mixing, kneading, granulation, tableting, coating, sterilization, emulsification, etc., depending on the form of the preparation.
- a conventional method such as mixing, kneading, granulation, tableting, coating, sterilization, emulsification, etc., depending on the form of the preparation.
- the pharmaceutical composition of the present invention may be formed into a sustained release agent containing an active ingredient and a biodegradable polymer compound.
- the sustained-release agent can be prepared according to the method described in JP-A No. 9 2 6 3 5 4 5.
- the content of the substance of the present invention or a salt thereof varies depending on the form of the preparation, but is usually about 0.01 to 100% by weight, preferably about It is about 0.1 to 50% by weight, more preferably about 0.5 to 20% by weight.
- the substance of the present invention is used as the above-mentioned pharmaceutical composition, it is used as it is or as an appropriate pharmaceutically acceptable carrier, for example, excipient (for example, starch, lactose, sucrose, calcium carbonate, calcium phosphate, etc.), binding Agents (eg, starch, gum arabic, carboxymethylcellulose, hydroxypropylcellulose, crystalline cellulose, alginic acid, gelatin, polyvinylpyrrolidone, etc.), lubricants (eg, stearic acid, magnesium stearate, calcium stearate, talc, etc.), Disintegrating agents (eg, carboxymethylcellulose calcium, talc, etc.), diluents (eg, water for injection, physiological saline, etc.), additives as required (stabilizers, preservatives, coloring agents, fragrances, solubilizers, emulsifiers) Buffer, isotonic Can be administered orally or parenterally in the form of solids such as powder
- the dosage is the type of the substance of the present invention or a pharmaceutically acceptable salt thereof, the degree of symptoms, the age, sex, body weight, sensitivity difference, timing of administration, administration route, administration interval, pharmaceutical composition
- about 0.005 to 10 of the substance of the present invention per kg of body weight per day Omg preferably about 0.05 to 50 mg, more preferably about 0.2 to 3 Omg can be administered in 1 to 3 divided doses.
- the dosage is the type and content of the substance of the present invention, dosage form, duration of drug release, animal to be administered (eg, human, rat, mouse) Mammals such as cats, dogs, rabbits, cows, pigs, etc.), depending on the purpose of administration. For example, when applied parenterally, about 0.1 to about 10 Omg of book per week.
- the substance of the invention may be released from the dosage formulation.
- the substance of the present invention can be used in an appropriate amount or in combination with other pharmaceutically active ingredients as appropriate.
- the dose can be reduced compared with the case where the substance of the present invention or other pharmaceutically active ingredient is administered alone. More specifically, when the substance of the present invention and clenbuterol (Clenbuterol, a therapeutic agent for stress urinary incontinence), which is a two-agonist, are used in combination, the dosage is higher than that when clenbuterol is administered alone. For example, side effects such as tremor can be reduced.
- clenbuterol a therapeutic agent for stress urinary incontinence
- the drug used in combination with the substance of the present invention can be selected according to the patient's symptoms (mild, severe, etc.) (3)
- the treatment period can be set longer by selecting another pharmaceutically active ingredient having a different mechanism of action from the substance of the present invention.
- the therapeutic effect can be sustained by selecting another pharmaceutically active ingredient that has a different mechanism of action from the substance of the present invention.
- concomitant drugs examples include the following.
- Adrenaline ⁇ 1 receptor agonist eg, ephedrine hydrochloride, mitorine hydrochloride
- adrenaline 52 receptor agonist eg, Clen bu tero 1
- Tricyclic antidepressants eg, imibramine hydrochloride
- anticholinergic agents or smooth muscle stimulants eg, oxiptinin hydrochloride, probiverin hydrochloride, serimevelin hydrochloride
- female hormone drugs eg, conjugated estrogen (premarin) ), Estriol
- Insulin preparations eg, animal insulin preparations extracted from sushi and bronze spleen; human insulin preparations genetically engineered using Escherichia coli and yeast; insulin zinc; protamine insulin zinc; insulin fragments or Derivatives (eg, INS-1 etc.), etc., insulin sensitivity enhancers (eg, pioglitazone hydrochloride, troglitazone, oral diglizone or its maleate, JTT-501, MCC-555, YM-440, GI-262570 , KRP-297, FK-614, CS-011, etc.), cc-glucosidase inhibitors (eg, voglibose, carbose, miglitol, emiglitate, etc.), biguanides (eg, phenformin, metformin, buformin, etc.) ), Sulfonylurea agents (eg, tolptamide, glibenclamide, gliclazide, chlorp) Pami
- Aldose reductase inhibitors eg, torres, epalles, zena restat, zoborrestat, fidarestat (SNK-860), minarerestat (ARI-509), CT-1-12, etc.
- neurotrophic factors eg, , NGF, NT-3, etc.
- AGE inhibitors eg, ALT-945, pimagedin, viratoxatin, N-phenacylthiazolium promide (ALT-766), EX 0-226, etc.
- active oxygen Eliminating agents eg, thioctic acid
- cerebral vasodilators eg, thioprid
- Subtilin compounds that are cholesterol synthesis inhibitors eg, Pravas Yuchin, Symbus Yuchin, Mouth Bath Yuchin, Atrubas Yuchin, Full Bath Yuchin, Ceribasu Yuchin, or salts thereof (eg, sodium salt) Etc.
- squalene synthase inhibitors or fibrates having a triglyceride-reducing action eg, bezafibrate, clofibrate, simfibrate, clinofibrate, etc.
- Antihypertensive Angiotensin converting enzyme inhibitors eg, captopril, ⁇ nalapril, delapril, etc.
- angiotensin II antagonists eg, oral sultan, candesal evening, cilexetil, etc.
- calcium antagonists eg, manidipine, diphedipine, Amlodipine, efonidipine, dicardipine, etc.
- clonidine etc.
- Central anti-obesity drugs eg, dexfenfluramine, fenfluramine, phentermine, sibutramine, amphephramone, dexamphetamine, mazindol, phenylpropanolamine, clobenzolex, etc.
- ⁇ lipase inhibitor Eg, orlistate
- 3 agonists eg, CL-316243, SR-58611-A, UL-TG-307, AJ-9677, AZ 4014 0, etc.
- peptidic appetite suppressants eg, lebutin, CNTF ( Ciliary neurotrophic factor) etc.
- cholecystokininagonist eg, Lynch tribute, FPL-15 849 etc.
- Xanthine derivatives eg, sodium salicylate theopromin, salicylic acid lucumteopromine, etc.
- thiazide preparations eg, ethiazide, cyclopenthiazide, trichloromethiazide, hydrochlorothiazide, hydroflumethiazide, benzylhydrocrothiazide, Penflutizide, polythiazide, methical oral thiazide, etc.
- anti-aldosterone preparation eg, spironolactone, triamterene, etc.
- carbonic anhydrase inhibitor eg, acetozolamide, etc.
- chlorobenzenesulfonamide eg, chlor
- azosemide isosorbide, ethacrynic acid, pyrethanodide, bumetanide, furosemide, etc
- Alkylating agents eg, cyclophosphamide, ifosfamide, etc.
- antimetabolites eg, methotrexate, 5-fluorouracil, etc.
- anticancer antibiotics eg, mitomycin, adriamycin, etc.
- plant-derived anticancer agents eg, The pink Listin, vindesine, taxol, etc.
- cisbratine etepoxide, etc.
- 5-flurouracil derivatives such as flurolone or neofluron.
- Microorganisms or bacterial components eg, muramyl dipeptide derivatives, picibanil, etc.
- polysaccharides with immunopotentiating activity eg, lentinan, schizophyllan, krestin, etc.
- site force-in obtained by genetic engineering techniques (eg, interferon, Interleukin (IL), etc.)
- colony stimulating factors eg, granulocyte colony stimulating factor, erythropoietin, etc.
- Progesterone derivatives eg, megesterol acetate
- Metocro Bramidic drugs e.g., tetrahydrocannabinol drugs (the literature is the same as above)
- fat metabolism improving drugs eg, eicosapenic acid, etc.
- TNF-H, LIF, IL-6, Oncos Yuchin a factor that induces growth hormone, IGF- or cachexia Antibodies against M.
- Steroids eg, dexamethasone
- sodium hyaluronate e.g., sodium hyaluronate
- cycloxygenase inhibitors eg, indometacin, ketoprofen, loxoprofen, meloxicam, ampiroxicam, celecoxib, oral fuecoxib, etc.
- Glycation inhibitors eg, ALT-711, etc.
- nerve regeneration promoters eg, Y_128, VX853, pro sapt ide, etc.
- central nervous system agonists eg, decibram
- Amitriptyline imipramine, floxetine, paroxetine, doxepin and other antidepressants
- antiepileptic drugs eg, lamotrigine, carbamazepine
- antiarrhythmic drugs eg, mexiletine
- acetylcholine receptor ligands eg, ABT-15 9 4
- endothelin receptor antagonists eg, ABT— 6 2 7
- monoamine uptake inhibitors eg, tramadol
- indoleamine uptake inhibitors eg, floxetine, paroxetine
- narcotic analgesics eg, , morphine
- GABA receptor agonists e.g., Giyaba
- Anticholinergic agents include, for example, Attoguchi pin, scopolamine, homatropine, tropicamide, cyclopentolate, butyl scopolamine bromide, propane terin bromide, methyl penactidium bromide, mepenzolate bromide, flavoxate, pyrensebine, odor Ipratopium, trihexyphenidyl, oxiputinine, propiverine, darifenacin, tolterodine, temiverine, trospium chloride or its salts (eg, sulfate mouthpiece, scopolamine hydrobromide, homato mouthpin hydrobromide, cyclohydrochloride hydrochloride)
- bentolate, flavoxate hydrochloride, pyrenecebine hydrochloride, trihexyphenidyl hydrochloride, oxyputinine chloride, tolterodine tartrate among others, oxybutynin, propiverine, darifen
- NK-2 receptor antagonists examples include GR 159897, GR 149861, SR 48968 (sar edut ant), SR 144190, YM35375, YM38336, ZD 7944, L—743986, MD L 105212 A, ZD6021, MDL 105172 A , S CH 205528, SCH 62373, piperidine derivatives such as R—113281, perhydroisoindole derivatives such as RPR—10 6145, quinoline derivatives such as SB—414240, pyropyrimidine derivatives such as ZM—253270, MEN 11420 (nepadut ant) s SCH217048, L-65587 7, PD-147714 (CAM-2291), pseudopeptide derivatives such as MEN10376, S16474, etc., GR 100679, DNK33 3, GR94800, UK—224671, MEN 10376, MEN 106 27 Or a salt thereof. .
- the pharmaceutical composition in which the substance of the present invention and the concomitant drug are combined or used in combination includes those formulated as a single pharmaceutical composition containing the substance of the present invention and the concomitant drug, and Any of those in which the light substance and the concomitant drug are separately formulated are included. Hereinafter, these are collectively referred to as the combination agent of the present invention.
- the concomitant drug of the present invention comprises the substance of the present invention and the concomitant drug, separately or simultaneously, mixed as it is or with a pharmaceutically acceptable carrier, etc. It can be formulated by a method.
- the daily dose of the concomitant drug of the present invention depends on the degree of symptoms, age of the subject, sex, body weight, sensitivity difference, timing of administration, interval, properties of the pharmaceutical composition, formulation, type, type of active ingredient, etc. Therefore, there is no particular limitation.
- Each dose of the substance of the present invention and the concomitant drug is not particularly limited as long as it does not cause a problem of side effects, but is usually about 0.05 to 10 O mg per kg body weight of a mammal by oral administration. Preferably it is about 0.05 to 50 mg, more preferably about 0.2 to 30 mg, and this is usually administered in 1 to 3 divided doses.
- the substance of the present invention and the concomitant drug may be administered at the same time, or the concomitant drug may be administered first and then the substance of the present invention may be administered.
- the substance of the present invention may be administered first, and then the concomitant drug may be administered.
- the time difference varies depending on the active ingredient, dosage form, and method of administration. For example, when administering a concomitant drug first, 1 minute to 3% after administering the concomitant drug Within 10 minutes, preferably within 10 minutes to 1 day, more preferably within 15 minutes to 1 hour.
- the substance of the present invention is administered first, the concomitant drug within 1 minute to 1 day, preferably within 10 minutes to 6 hours, more preferably within 15 minutes to 1 hour after the administration of the substance of the present invention.
- the method of administering is mentioned.
- the respective contents of the substance of the present invention and the concomitant drug differ depending on the form of the preparation, but are usually about 0 with respect to the whole preparation. It is about 0.1 to 90% by weight, preferably about 0.1 to 50% by weight, and more preferably about 0.5 to 20% by weight.
- the content of the carrier in the concomitant drug is usually about 0 to 99.8% by weight, preferably about 10 to 99.8% by weight, more preferably about 10 to 90%, based on the entire preparation. It is about wt%.
- the pharmaceutical composition containing the concomitant drug can be produced and used in the same manner as the pharmaceutical composition containing the substance of the present invention.
- the effect of the test substance on the total urethral resistance of the animal eg, non-human mammal
- the animal eg, non-human mammal
- Leak pressure (l e a k o nt tp s s ure) refers to the intravesical pressure when urinary leakage occurs without contraction of the detrusor, and indicates the maximum urethral resistance that can resist the increase in intravesical pressure.
- urinary incontinence is observed when the abdominal pressure is transiently increased by electrically stimulating the abdominal muscles of the animal or the diaphragm or the nerves that control them to measure the maximum intravesical pressure. The lowest urinary bladder pressure among the trials performed was taken as the leak pressure. The specific method for measuring the leakage pressure is described in detail in the examples described later.
- the “animal” used in the present invention is preferably female, and the species includes non-human animals such as monkeys, dogs, cats, rabbits, guinea pigs, hamsters, rats, mice, gerbils, and particularly rats (W istar SD, etc.) are most preferred.
- non-human animals such as monkeys, dogs, cats, rabbits, guinea pigs, hamsters, rats, mice, gerbils, and particularly rats (W istar SD, etc.) are most preferred.
- the age, weight, presence / absence of delivery, etc. of the “animal” used in the present invention are not particularly limited as long as they can be applied to the intended screening, but these conditions may be appropriately changed.
- a normal animal an animal that does not show a disease state
- the state of low leakage pressure is that the nerve involved in the urethral contraction reflex is cut or damaged. Those that are injured or have reduced weight of the pelvic floor muscles and the external urethral sphincter are preferred.
- the nerves involved in the urethral contraction reflex eg, pelvic nerve, pudendal nerve, i 1 io coccygeous i / v ubococcygeous muscle Etc.
- pelvic nerve, pudendal nerve, i 1 io coccygeous i / v ubococcygeous muscle Etc. whether physically, chemically or biologically cut or damaged, giving birth, removing the ovaries, mechanically expanding the vagina, inducing diabetes, or administering drugs Or just combine them.
- drugs examples include the neuromuscular junction blocker Hiichi Banga Mouth Toxin, d-Vococlarin, Panchloronium, Decamesonium, Suxamesonium, and the like.
- the pressure at the time of leakage is measured when the abdominal pressure rises rapidly in animals.
- abdominal muscles or diaphragm or those are used as a method for causing a sudden increase in abdominal pressure.
- Electrical stimulation to the governing nerve eg, duration of about 0.01 to about 10 milliseconds, preferably about 0.1 to about 1 millisecond, voltage of about 1 to about 100 V, preferably about 3 to about 50 V Muscle contraction due to electrical stimulation (single or repeated rectangular waves).
- test substances include known or novel synthetic compounds, physiologically active substances derived from natural products, peptides, proteins, etc., for example, warm-blooded mammals (for example, mice, rats, bushes, bushes, sheep, monkeys, Human tissue), cell culture supernatant, etc. are used.
- warm-blooded mammals for example, mice, rats, bushes, bushes, sheep, monkeys, Human tissue
- cell culture supernatant etc.
- the leakage pressure when administered is about 5 cmH 20 , preferably about 10 cmH 20 or more, more preferably than when no test substance is administered. If elevated approximately 15 cm H 2 0 or more, the test substance can be judged to have the effect of improving stress incontinence.
- the screening method of the present invention measures the leakage pressure at the time of rapid increase in abdominal pressure in animals, and is used for the prevention and treatment of stress urinary incontinence (for example, adrenaline ⁇ : 1 receptor). It can be usefully and efficiently applied to screening of body antagonists, adrenaline ⁇ 2 receptor agonists, serotonin uptake inhibitors, noradrenaline uptake inhibitors, or serotonin and noradrenaline uptake inhibitors.
- a test substance for example, about 0.0001 to about 100 Omg kg (preferably about 0.001 to about 10 Omg / kg) of a test substance is administered to a non-human mammal in the screening method of the present invention, and the leakage pressure is used as an indicator.
- the leakage pressure is used as an indicator.
- preventive and therapeutic agents for stress urinary incontinence by examining the therapeutic effect of the test substance, it is possible to evaluate preventive and therapeutic agents for stress urinary incontinence.
- the concept of prevention of stress urinary incontinence includes suppression of decreased urethral resistance
- the concept of treatment of stress urinary incontinence includes improvement of stress urinary incontinence, suppression of progression, and prevention of seriousness. .
- the time when the test substance is administered to the animal in the evaluation method of the present invention includes the time before or after the leakage pressure reduction treatment, and also during the time of leakage pressure measurement. Accordingly, it is possible to evaluate drugs for the purpose of preventing or treating stress urinary incontinence.
- the animal used in the present invention may be a normal animal (animal that does not show a disease state), for example, stress urinary incontinence, overactive bladder, prostatic hypertrophy, bladder hypotonia, diabetes, diabetic Animals with pathological conditions such as neurosis, hypertension, obesity, hyperlipidemia, arteriosclerosis, gastric ulcer, asthma, chronic obstructive respiratory disease, uterine cancer, cerebrovascular disorder, brain injury, spinal cord injury (for example, The above-described leakage pressure measurement may be carried out using an obesity rat (such as Wistar Fatty Rat). A new stress incontinence model is necessary when the above-mentioned pressure measurement at the time of leakage is carried out on animals exhibiting such pathological conditions.
- the pathological conditions eg, gastrointestinal diseases such as stomach ulcers, etc.
- the screening of pharmaceutical substances that are effective only for the treatment and have no effect on stress urinary incontinence, or exclude test substances that cause stress urinary incontinence from the selected pharmaceutical substances. It can also be applied to screening for these purposes.
- the screening method of the present invention is not only effectively applied to screening of a pharmaceutical substance for prevention / treatment of stress urinary incontinence by applying a test substance and examining the effect of increasing pressure at the time of leakage. It can also be applied effectively for screening various medicinal substances by applying test substances and examining the effects on urethral resistance (including exacerbations, no effects, and improvements) when abdominal pressure rises. Is possible. In other words, test substances that exacerbate stress urinary incontinence should be excluded from the medicinal substances to be selected; test substances that do not affect stress urinary incontinence may be used to prevent or treat diseases other than stress urinary incontinence.
- test substance that shows an improvement effect on stress urinary incontinence
- certain diseases eg, urinary tract diseases such as overactive bladder
- the screening method of the present invention can be evaluated in a state almost in agreement with the pathological condition, and evaluation of stress urinary incontinence using a parametric index rather than the presence / absence determination ( It can be applied as a useful evaluation system even when screening various kinds of pharmaceutical substances for any of the above-mentioned purposes.
- the substance obtained by the screening method of the present invention can be formulated in the same manner as the substance of the present invention described above and used as a prophylactic / therapeutic agent for stress urinary incontinence.
- Example 1 The present invention will be described more specifically with reference to examples and formulation examples below, but the present invention is not limited thereto.
- Example 1
- the bladder catheter is connected to the pressure transducer, and the signal from the transducer is sent to the computer via the amplifier (blood pressure amplification unit AP—641 G; Nihon Kohden) and the data analysis device (BI OP AC; MP 100). Recorded on the hard disk. The data was analyzed on the computer evening using software (BIOPAC; AcqKnowledge). The exposed abdominal muscles were electrostimulated using an electrical stimulator (SEN-3301; Nihon Kohden) and Isore Ichiyo (S S-202 J; Nihon Kohden) to observe the presence of urinary incontinence from the urethral orifice.
- SEN-3301 Nihon Kohden
- Isore Ichiyo S S-202 J
- Nihon Kohden Isore Ichiyo
- the electrical stimulation was performed for 1 second at a voltage of 3 to 50 V, a duration of 0.05 to 0.5 millisecond, and a frequency of 50 Hz to gradually increase the intravesical pressure.
- the maximum intravesical pressure during electrical stimulation of the abdominal wall was measured, and the lowest maximum intravesical pressure among trials in which urinary incontinence was observed was defined as the leakage pressure.
- the abdominal muscles were electrically stimulated with a constant stimulation intensity, then the abdomen was opened, and the abdominal muscles were electrically stimulated again under the same stimulation conditions.
- the pelvic nerve, the hypogastric nerve, or the pudendal nerve and the nerve toward the i io coccygeou s muscle / p ubo coc cygeou s muscle were cut in both directions, and the effect of nerve cutting was also examined.
- Table 1 Effects of bilateral amputation of the nerve toward the pelvic nerve, hypogastric nerve, or pudendal nerve and iliococc yge ous muscle on the pressure during leakage due to electrical stimulation of the abdominal wall in urethane anesthetized female rats
- Pelvic nerve amputation group 3 1. 7 ⁇ 2. 7 * *
- Somatic nerve amputation group 3 9. 3 ⁇ 3. 8 * Data show mean SEM of 5-7 cases.
- the somatic nerve amputation group refers to a group in which the nerves toward the pudendal nerve, the i io co ccygeou s muscle and the pubococ c cygeou s muscle are cut together. * P ⁇ 0. 05, P ⁇ 0. 01 vs. sham surgery group (two-sided test, Dun n e t t test)
- Evans blue dye (Me rck) solution was injected into the bladder, and the bladder volume was adjusted to 0.2 to 0.3 ml. Connect the bladder catheter to the pressure transducer device, and send the signal from the transducer to the computer via the amplifier (blood pressure amplification unit AP—641 G; Nihon Kohden) and the device analyzer (BI OP AC MP 100). Recorded on a hard disk. The data was analyzed on a computer using software (BI OPAC; Ac qKnow ledge).
- the exposed abdominal muscles were electrically stimulated using an electrical stimulator (SEN-3301; Nihon Kohden) and Isoray Yuichi (SS-202 J; Nihon Kohden) to observe the presence of urinary incontinence from the urethral orifice.
- the electrical stimulation was performed at a voltage of 3 to 50 V, a duration of 0.05 to 0.5 milliseconds, and a degree of 50 Hz for 1 second to gradually increase the intravesical pressure.
- the maximum intravesical pressure during abdominal wall stimulation was measured, and the lowest maximum intravesical pressure among trials in which urinary incontinence was observed was defined as the leakage pressure.
- the leakage pressure was compared before and after administration of duloxetine and midodrine.
- Duloxetine was dissolved in N, N-dimethylformamide (DMA) / olyethylene glycol 400 (PEG400) (1: 1) and administered intravenously at a rate of 0.5m 1 / kg.
- DMA N, N-dimethylformamide
- PEG400 olyethylene glycol 400
- (Hydrochloride, Sigma) was dissolved in physiological saline and administered intravenously at a rate of 1 m 1 / kg.
- urinary incontinence was observed in 7 of 10 cases in the sham operation group, and the leakage pressure in 10 cases was 57 ⁇ 3 ⁇ 4.4 cmH 2 0 or more (previous item).
- duloxetine or middine which has an effect on stress urinary incontinence, was used in rats with the nerve cut to the i 1 io coccygeou s muscle on one side cut to the ubococcygeou s muscle.
- drin was administered intravenously and the leakage pressure was compared before and after that, any drug significantly increased the leakage pressure (Table 2).
- Table 2 Effect of intravenous administration of duloxetine or midodrine on leak pressure during abdominal wall electrical stimulation in urethane anesthetized female rats Number of cases Pressure at leak
- the exposed abdominal muscles were electrically stimulated using an electrical stimulator (SEN-3301; Nihon Kohden) and an isolator (SS-202 J; Nihon Kohden) to observe the presence of urinary incontinence from the urethral orifice.
- the electrical stimulation was performed at a voltage of 2.5 to 50 V, a duration of 0.5 milliseconds, and a frequency of 50 Hz for 1 second to gradually increase the intravesical pressure.
- the maximum intravesical pressure during abdominal wall stimulation was measured, and the lowest maximum intravesical pressure among trials in which urinary incontinence was observed was defined as the leakage pressure.
- the SB 221284 is a serotonin 5-HT 2 c receptor en evening is agonist RS- 102221 or serotonin 5-HT 2B / 2C receptor en evening agonist, WAY- 161503 or WAY- 163 f 909 It was administered intravenously 5 minutes before administration, and an antagonistic test for the effect of WAY-161503 or WAY-163909 was conducted.
- RS-1 02221 and SB 221284 were both dissolved in DMA / PEG400 (1: 1) and administered intravenously at a rate of 0.5 ml / kg.
- Serotonin 5 HT 2C receptor agonist D ⁇ I (5— HT 2A / 2B / 2C receptor agonist), mCPP (5— HT 2B / 2C receptor partial agonist), eltoprazine (5—HT 1A / 1B / 2C receptor partial agonist) significantly increased the leakage pressure (Table 3).
- the pressure on both sides of the bladder was gradually compressed with two cotton swabs, and the intravesical pressure when physiological saline leaked from the urethral orifice was measured and defined as the leakage pressure.
- the measurement at the time of leakage was repeated, and the average value of the last 5 stable measurements was taken as the value before drug administration. Thereafter, the drug was intravenously administered, and after 20 minutes, the leakage pressure was measured again to obtain the value after drug administration.
- DO I, m CPP, and eltobradine are dissolved in physiological saline, and other drugs are dissolved in DMA / PEG400 (1: 1), and intravenously at a rate of 1 m 1 / kg or 0.5 mg / kg. It was administered internally.
- Duloxetine a treatment for stress urinary incontinence, increased the leakage pressure as measured by directly compressing the bladder (Table 5).
- Intravenous administration of serotonin 5-HT 2C receptor agonists WAY-16 1503 and WAY-163909 increased the leakage pressure in a dose-dependent manner, both of which were less than 0.1 mg / kg. The upper dose had a significant effect (Table 5).
- WAY-163907 which is an optical isomer of WAY-163909 and has no 5-HT 2C receptor agonist action, did not show any action (Table 5).
- Serotonin 5-HT 2C receptor Agonisuto has an action DO I (5- HT 2A / 2B / 2C receptor Agonisuto), mCPP (5- HT 2B / 2C receptor partial Agonisu g), Erutopurajin (5- HT 1A / 1B / 2C receptor partial agonist) significantly increased the leakage pressure (Table 5).
- Intravenous administration of serotonin 5—HT 2C receptor antagonist RS—102221 suppressed the increase in leakage pressure due to WAY—161503 (0.3 mg / kg, iv) in a dose-dependent manner and was suppressed at 3 mgZkg. Was significant (Table 6).
- Table 5 Effects of intravenous administration of serotonin 5 _HT receptor agonist and duloxetine on leakage pressure due to direct bladder compression in urethane anesthetized female rats Pressure at leakage
- Solvent-7 60. 1 ⁇ 4. 7 60. 9 ⁇ 5.2 0. 7 ⁇ 0. 7 Duloxetine 0.01 6 58. 1 ⁇ 6. 0 60. 5 ⁇ 5. 1 2. 4 ⁇ 2. 6
- WAY-163909 0.3 mg / kg, V, SB221284 0.3 mg / kg, i. V. 6 60. 3 ⁇ 4.0 60.7 ⁇ 3.9 0.4 ⁇ 1.73; Leakage time was measured using a method of urinating the bladder. The data shows the ⁇ mean value SEM for each group.
- a microchip 'transducer catheter (SPR-524, Milarlar nstrument s Inc.) is inserted into the bladder from the urethral orifice, and the transducer is placed on the surface of the catheter using the scale written on the catheter surface.
- a part of the support was positioned 12.5 to 15.0 mm from the urethral opening, and the transducer surface was adjusted so that it touched the 3 o'clock direction of the inner surface of the urethra.
- Microchip 'Transdeuser measured local pressure change in the urethra (hereinafter referred to as urethral pressure for convenience)
- Amplifier blood pressure amplification unit AP— 641 G; Nihon Kohden
- AP blood pressure amplification unit
- biopack sampled at 500 Hz
- the urethral response induced by an increase in intravesical pressure was measured three times, and the average of the last two was taken as the value before drug administration.
- the evaluation items were resting urethral pressure and reflex urethral contraction response.After calculating the average urethral pressure per second, the values immediately before raising the bladder pressure were calculated from the static urethral pressure and the maximum value when the bladder pressure was raised. The value obtained by subtracting the urethral pressure at rest was taken as the urethral contraction response.
- the specific serotonin 5—HT 2C receptor agonist WAY—161503 (0.3 mg / kg) or duloxetine (10 mg / kg) was administered intravenously for 5 minutes. Later increased the intravesical pressure again The urethral response was measured. Both drugs were dissolved in DMA-PEG400 (1: 1) and administered at a rate of 0.5 ml / kg.
- the data shows the average soil SEM of 5 cases in each group.
- Changes in the intravesical pressure measured by the transducer were sent to a computer via an amplifier (amplification unit for blood pressure AP—641 G; Nihon Kohden) and a data analysis device (MP—100; biopack) and recorded on the hard disk.
- an amplifier amplification unit for blood pressure AP—641 G; Nihon Kohden
- MP data analysis device
- the intravesical pressure was increased for 90 seconds, and the presence or absence of saline leakage from the urethral orifice was observed.
- the intravesical pressure was increased every 2.5 cmH 2 0, and after the 90-second observation period, the intravesical pressure was temporarily returned to 0 cmH 2 O, and then the next step was started.
- the intravesical pressure when the leakage of physiological saline was observed from the urethral orifice was taken as the leakage pressure.
- the measurement of the leak time pressure was repeated, and the average value of the last three times was taken as the value before drug administration. Thereafter, the drug was intravenously administered, and the leak time pressure was measured again 10 minutes later.
- the drug was dissolved in DMA-PEG400 (1: 1) and administered at a rate of 0.5 mlZkg.
- duloxetine and WAY-161503 increased the leakage pressure in the intravesical pressure fixation method (Table 8).
- Androgen dihydrotestosterone (DHT) also increased the leakage pressure in the intravesical pressure fixation method (Table 8).
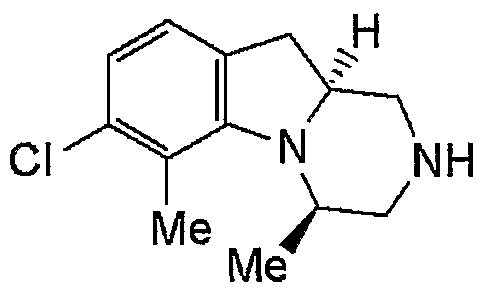
- WAY— 161503 1 A mixture of Omg, lactose 60 mg and corn starch 35 mg, 10% by weight hydroxypropyl methylcellulose aqueous solution 0. Use 03mL (3mg as hydroxypropylmethylcellulose), granulate, dry at 40 ° C and sieve. The resulting granules are mixed with 2 mg of magnesium stearate and compressed. The resulting uncoated tablet is coated with a sugar coating made from an aqueous suspension of sucrose, titanium dioxide, talc and gum arabic. The coated tablet is squeezed out with beeswax to obtain a coated tablet.
- WAY- 161503 1 Omg and magnesium stearate 3 mg are granulated with 0.07 mL soluble starch aqueous solution (7 mg as soluble starch), dried and mixed with 7 Omg lactose and 5 Omg corn starch. Compress the mixture to obtain tablets.
- the method for screening for the prevention and treatment of stress urinary incontinence according to the present invention is an excellent in vivo evaluation system because it measures the urethral resistance at the time of rapid and transient increase in abdominal pressure according to the pathological condition by the pressure at the time of leakage. Therefore, it can be usefully and efficiently applied to the screening of substances used for the prevention and treatment of stress urinary incontinence. On the other hand, it is also useful as an evaluation system to test that things used for the prevention and treatment of other diseases do not cause stress urinary incontinence.
- a substance that can be obtained by the screening method of the present invention for example, a substance that increases the leakage pressure when the abdominal pressure increases, can be used as a preventive / therapeutic agent for stress urinary incontinence.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Urology & Nephrology (AREA)
- Immunology (AREA)
- Hematology (AREA)
- Molecular Biology (AREA)
- Cell Biology (AREA)
- Diabetes (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Tropical Medicine & Parasitology (AREA)
- Biochemistry (AREA)
- Food Science & Technology (AREA)
- Pathology (AREA)
- Biotechnology (AREA)
- Toxicology (AREA)
- Microbiology (AREA)
- General Physics & Mathematics (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Obesity (AREA)
- Reproductive Health (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/660,751 US8685924B2 (en) | 2004-08-25 | 2005-08-24 | Preventives/remedies for stress urinary incontinence and method of screening the same |
| EP05781418A EP1792629A4 (en) | 2004-08-25 | 2005-08-24 | MEANS FOR THE PREVENTION / TREATMENT OF STRESS-RELATED HARNINE INCONTINENCE AND PRECAUTIONARY METHOD FOR THIS |
| JP2006532742A JP5173190B2 (ja) | 2004-08-25 | 2005-08-24 | 腹圧性尿失禁の予防・治療剤及びそのスクリーニング方法 |
| US14/156,677 US20140155369A1 (en) | 2004-08-25 | 2014-01-16 | Preventives/remedies for stress urinary incontinence and method of screening the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004-245931 | 2004-08-25 | ||
| JP2004245931 | 2004-08-25 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/660,751 A-371-Of-International US8685924B2 (en) | 2004-08-25 | 2005-08-24 | Preventives/remedies for stress urinary incontinence and method of screening the same |
| US14/156,677 Division US20140155369A1 (en) | 2004-08-25 | 2014-01-16 | Preventives/remedies for stress urinary incontinence and method of screening the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006022420A1 true WO2006022420A1 (ja) | 2006-03-02 |
Family
ID=35967615
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2005/015830 Ceased WO2006022420A1 (ja) | 2004-08-25 | 2005-08-24 | 腹圧性尿失禁の予防・治療剤及びそのスクリーニング方法 |
Country Status (4)
| Country | Link |
|---|---|
| US (2) | US8685924B2 (ja) |
| EP (3) | EP1792629A4 (ja) |
| JP (2) | JP5173190B2 (ja) |
| WO (1) | WO2006022420A1 (ja) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007132841A1 (ja) * | 2006-05-16 | 2007-11-22 | Takeda Pharmaceutical Company Limited | 縮合複素環化合物およびその用途 |
| EP2009374A2 (en) | 2007-06-28 | 2008-12-31 | Whirlpool Corporation | Distributed refrigeration system with custom storage modules |
| WO2009063992A1 (ja) * | 2007-11-15 | 2009-05-22 | Takeda Pharmaceutical Company Limited | 縮合ピリジン誘導体およびその用途 |
| JP2009273396A (ja) * | 2008-05-14 | 2009-11-26 | Mitsubishi Chemical Medience Corp | 腹圧性尿失禁症モデル動物の作製方法 |
| WO2011071136A1 (ja) | 2009-12-11 | 2011-06-16 | アステラス製薬株式会社 | 線維筋痛症治療剤 |
| EP2123644A4 (en) * | 2007-03-07 | 2011-06-29 | Takeda Pharmaceutical | BENZOXAZEPINE DERIVATIVES AND THEIR USE |
| US8480437B2 (en) | 2008-04-11 | 2013-07-09 | Bal Seal Engineering, Inc. | Connector cartridge stack for electrical transmission |
| JPWO2013133325A1 (ja) * | 2012-03-06 | 2015-07-30 | 武田薬品工業株式会社 | 三環性化合物 |
| JP2017531012A (ja) * | 2014-10-16 | 2017-10-19 | ジーティーエックス・インコーポレイテッド | Sarmを使用した泌尿器系障害の治療方法 |
| WO2019131902A1 (ja) | 2017-12-27 | 2019-07-04 | 武田薬品工業株式会社 | 腹圧性尿失禁および便失禁の治療薬 |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GT200500317A (es) | 2004-11-05 | 2006-10-27 | Proceso para preparar compuestos de quinolina y productos obtenidos de los mismos | |
| AR054849A1 (es) | 2005-07-26 | 2007-07-18 | Wyeth Corp | Diazepinoquinolinas, sintesis de las mismas, e intermediarios para obtenerlas |
| TW200806298A (en) * | 2006-03-24 | 2008-02-01 | Wyeth Corp | Methods for modulating bladder function |
| AU2013235518B2 (en) * | 2012-03-19 | 2017-03-02 | Wellesley Pharmaceuticals, Llc | Extended-release formulation for reducing the frequency of urination and method of use thereof |
| CN111956640B (zh) * | 2020-09-14 | 2022-07-15 | 长春金赛药业有限责任公司 | 含酯基芳香丙酰胺类化合物在制备治疗尿失禁药物中的应用 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998033504A1 (en) * | 1997-02-03 | 1998-08-06 | Akzo Nobel N.V. | Treatment of urinary incontinence |
| WO2000076984A2 (en) * | 1999-05-21 | 2000-12-21 | Biovitrum Ab | Novel compounds, their use and preparation |
| WO2003097636A1 (en) * | 2002-05-17 | 2003-11-27 | Biovitrum Ab | Novel compounds and their use |
Family Cites Families (131)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3253989A (en) | 1963-02-11 | 1966-05-31 | American Cyanamid Co | Process for producing anorexia |
| US3652588A (en) | 1969-10-23 | 1972-03-28 | Upjohn Co | 6-alkyl-1 2 3 4 5 6-hexahydroazepino(4 5-b)indoles |
| US3676558A (en) | 1970-08-28 | 1972-07-11 | Upjohn Co | Anorexigenic compositions comprising 6-alkyl-1,2,3,4,5,6-hexahydroazepino 4,5-indoles as active ingredient and process of treatment |
| US4082844A (en) | 1976-02-09 | 1978-04-04 | Merck & Co., Inc. | 6-chloro-2-(1-piperazinyl)pyrazine |
| JPH0755904B2 (ja) | 1986-05-14 | 1995-06-14 | 小玉株式会社 | 膀胱疾患治療剤 |
| EP0370560B1 (en) | 1988-11-24 | 1994-01-12 | Akzo Nobel N.V. | Pharmaceutical composition containing 1-[mono- or bis (trifluoromethyl)-2-pyridinyl] piperazines |
| EP0572863A1 (de) | 1992-06-05 | 1993-12-08 | F. Hoffmann-La Roche Ag | ZNS Pyrazinoindole |
| TW270114B (ja) | 1993-10-22 | 1996-02-11 | Hoffmann La Roche | |
| TW334423B (en) | 1993-10-22 | 1998-06-21 | Hoffmann La Roche | Tricyclic 1-aminoethylpyrrole-derivatives |
| TW344661B (en) * | 1993-11-24 | 1998-11-11 | Lilly Co Eli | Pharmaceutical composition for treatment of incontinence |
| WO1996010567A1 (en) | 1994-09-30 | 1996-04-11 | Maruho Co., Ltd. | Aminoketone derivative, physiologically acceptable salt thereof, and use of the same |
| JP2992677B2 (ja) | 1995-06-05 | 1999-12-20 | 武田薬品工業株式会社 | 骨形成促進医薬組成物 |
| US5955495A (en) | 1996-05-03 | 1999-09-21 | Hoffmann-La Roche Inc. | Method of treating diseases of the CNS |
| GB9616700D0 (en) * | 1996-08-09 | 1996-09-25 | Carey Beverly J | Hormone supplement |
| EP0958285B1 (en) | 1997-01-08 | 2004-01-07 | F.Hoffmann-La Roche Ag | TRICYCLIC BENZO[e]ISOINDOLES AND BENZO[h]ISOQUINOLINES |
| ZA9817B (en) | 1997-01-08 | 1998-07-08 | Hoffmann La Roche | Tricyclic benzo[e]isoindoles and benzo[h]isoquinolines |
| WO1998030548A1 (en) | 1997-01-13 | 1998-07-16 | Yamanouchi Pharmaceutical Co., Ltd. | 5-HT2c RECEPTOR AGONISTS AND AMINOALKYLINDAZOLE DERIVATIVES |
| EP0863136B1 (en) | 1997-02-25 | 2003-09-24 | Akzo Nobel N.V. | Derivatives of azetidine and pyrrolidine |
| EP0995741A4 (en) * | 1997-06-27 | 2002-11-20 | Nippon Kayaku Kk | MEDICINES FOR THE TREATMENT / PREVENTION OF POLLAKIURIA OR INCONTINENCE AND TROPON DERIVATIVES |
| US6399618B1 (en) | 1997-07-09 | 2002-06-04 | Cardiome Pharma Corp | Compositions and methods for modulating sexual activity |
| JPH11106334A (ja) | 1997-09-30 | 1999-04-20 | Saitama Daiichi Seiyaku Kk | 尿失禁治療剤 |
| JP2001520195A (ja) | 1997-10-17 | 2001-10-30 | イーライ・リリー・アンド・カンパニー | 医薬の増強 |
| JP4422331B2 (ja) | 1998-02-26 | 2010-02-24 | ナームローゼ・フエンノートチヤツプ・オルガノン | アゼチジン誘導体およびピロリジン誘導体 |
| PA8469101A1 (es) | 1998-04-09 | 2000-09-29 | Pfizer Prod Inc | Ligandos azabiciclicos de receptores 5ht1 |
| GB9819020D0 (en) | 1998-09-01 | 1998-10-28 | Cerebrus Ltd | Chemical compounds III |
| GB9819019D0 (en) | 1998-09-01 | 1998-10-28 | Cerebrus Ltd | Chemical compounds II |
| GB9819033D0 (en) | 1998-09-01 | 1998-10-28 | Cerebrus Ltd | Chemical compounds VI |
| GB9819035D0 (en) | 1998-09-01 | 1998-10-28 | Cerebrus Res Ltd | Chemical compounds VII |
| GB9819032D0 (en) | 1998-09-01 | 1998-10-28 | Cerebrus Ltd | Chemical compounds IV |
| DE69928542T2 (de) | 1998-09-18 | 2006-03-30 | Alcon Manufacturing Ltd., Fort Worth | 5HT2-Agonisten zur Behandlung des Glaukoms |
| GB9820767D0 (en) | 1998-09-23 | 1998-11-18 | Cerebrus Ltd | Chemical compounds VIII |
| WO2000028993A1 (en) | 1998-11-19 | 2000-05-25 | Nortran Pharmaceuticals, Inc. | Serotonin ligands as pro-erectile compounds |
| US6372745B1 (en) | 1999-12-06 | 2002-04-16 | American Home Products Corporation | 2,3,4,4A-tetrahydro-1H-pyrazino[1,2-A]quinoxalin-5(6H)one derivatives |
| IL143323A0 (en) | 1998-12-17 | 2002-04-21 | American Home Prod | 2,3,4,4a-tetrahydro-1h-pyrazino (1,2-a) quinoxalin-5 (6h) one derivatives being 5ht2c agonists |
| CA2361516A1 (en) | 1999-01-27 | 2000-08-03 | Jeffrey Thomas Mullaney | Aminoalkylbenzofurans as serotonin (5-ht(2c)) agonists |
| GB9902047D0 (en) | 1999-01-29 | 1999-03-17 | Cerebrus Ltd | Chemical compounds XI |
| AR023574A1 (es) | 1999-04-23 | 2002-09-04 | Pharmacia & Upjohn Co Llc | Compuestos de azepinindol tetraciclico,composiciones farmaceuticas y el uso de dichos compuestos para preparar un medicamento, e intermediarios |
| WO2000069437A1 (en) | 1999-05-18 | 2000-11-23 | Synaptic Pharmaceutical Corporation | Use of agonists or antagonists of the 5-ht7 receptor to treat disorders of the bladder |
| US6465467B1 (en) | 1999-05-21 | 2002-10-15 | Biovitrum Ab | Certain aryl-aliphatic and heteroaryl-aliphatic piperazinyl pyrazines and their use in the treatment of serotonin-related diseases |
| MXPA01012969A (es) | 1999-06-15 | 2003-10-14 | Bristol Myers Squibb Pharma Co | Gamma-carbolinas fusionadas de heterociclo sustituido. |
| AU768310B2 (en) | 1999-07-29 | 2003-12-04 | Eli Lilly And Company | Benzofurylpiperazines and benzofurylhomopiperazines: serotonin agonists |
| WO2001009126A1 (en) | 1999-07-29 | 2001-02-08 | Eli Lilly And Company | Serotonergic benzothiophenes |
| WO2001009123A1 (en) | 1999-07-29 | 2001-02-08 | Eli Lilly And Company | Benzofurylpiperazine serotonin agonists |
| DE60006825T2 (de) | 1999-07-29 | 2004-08-12 | Eli Lilly And Co., Indianapolis | Serotonerge benzofurane |
| GB9918965D0 (en) | 1999-08-11 | 1999-10-13 | Cerebrus Ltd | Chemical compounds xxi |
| GB9918962D0 (en) | 1999-08-11 | 1999-10-13 | Cerebrus Ltd | Chemical compounds xxii |
| US20030004207A1 (en) * | 1999-11-29 | 2003-01-02 | Douglas A. Craig | Use of compounds which activate a 5-ht receptor to treat urinary incontinence |
| AU2042301A (en) | 1999-12-03 | 2001-06-12 | Alcon Universal Limited | 1-aminoalkyl-1H-indoles for treating glaucoma |
| US20020183357A1 (en) | 2000-02-17 | 2002-12-05 | Brioni Jorge D. | Use of alpha- 1- alpha adrenoceptor agonists with alpha-1-Beta antagonism for the treatment of stress urinary incontinence |
| EP1132389A1 (en) | 2000-03-06 | 2001-09-12 | Vernalis Research Limited | New aza-indolyl derivatives for the treatment of obesity |
| MXPA02008893A (es) | 2000-03-14 | 2003-02-10 | Upjohn Co | Compuestos novedosos de 2,3,4,5-tetrahidro-1h (1,4)diazepino (1,7-a)indol. |
| PT1267847E (pt) | 2000-03-17 | 2004-05-31 | Yamanouchi Pharma Co Ltd | Agonistas 5ht2 para controlar a pio e para tratamento do glaucoma |
| AU2001238552A1 (en) | 2000-03-17 | 2001-10-03 | Alcon, Inc. | Compounds with 5-HT2 and 5-HT1A agonist activity for treating glaucoma |
| WO2001083487A1 (en) | 2000-04-28 | 2001-11-08 | Yamanouchi Pharmaceutical Co., Ltd. | Froindazole derivative |
| AU2001269781A1 (en) | 2000-07-06 | 2002-01-21 | Pharmacia And Upjohn Company | Substituted 2,3,7,8,9,10,11,12-octahydroazepino(4,5-b)pyrano(3,2-e) indoles |
| SE0002754D0 (sv) | 2000-07-21 | 2000-07-21 | Pharmacia & Upjohn Ab | New pharmaceutical combination formulation and method of treatment with the combination |
| JP4180365B2 (ja) | 2000-07-31 | 2008-11-12 | エフ.ホフマン−ラ ロシュ アーゲー | ピペラジン誘導体 |
| EP1319004A2 (en) | 2000-09-20 | 2003-06-18 | PHARMACIA & UPJOHN COMPANY | Substituted azepino[4,5-b]indole derivatives |
| JP2002114684A (ja) | 2000-10-03 | 2002-04-16 | Eisai Co Ltd | 尿路疾患治療剤 |
| ATE403647T1 (de) | 2000-10-16 | 2008-08-15 | Hoffmann La Roche | Indolinderivate und deren verwendung als 5-ht2 rezeptor liganden |
| AU2002239463A1 (en) | 2000-11-03 | 2002-06-03 | Wyeth | Cyclopenta(b)(1,4) diazepino(6,7,1-hi)indoles as 5ht2c antagonists |
| AU2002227170A1 (en) | 2000-11-03 | 2002-05-15 | Wyeth | Cycloalkyl(b)(1,4)diazepino(6,7,1-hi)indoles and derivatives |
| DE60142355D1 (de) | 2000-11-20 | 2010-07-22 | Biovitrum Ab Publ | Piperazinylpyrazinverbindungen als agonisten oder antagonisten am serotonin-5ht-2-rezeptor |
| ATE433962T1 (de) | 2000-11-20 | 2009-07-15 | Biovitrum Ab Publ | Piperazinylpyrazinverbindungen als antagonisten des serotonin-5-ht2-rezeptors |
| EP1213017A3 (en) | 2000-12-05 | 2003-11-12 | Akzo Nobel N.V. | Use of a 5-HT2C receptor agonist for the treatment of hot flushes |
| GB0030580D0 (en) | 2000-12-15 | 2001-01-31 | Medicare Man Consultancy Ltd | Composition and method |
| GB0030710D0 (en) | 2000-12-15 | 2001-01-31 | Hoffmann La Roche | Piperazine derivatives |
| WO2002059124A2 (en) | 2000-12-20 | 2002-08-01 | Bristol-Myers Squibb Company | Substituted pyrroloquinolines and pyridoquinolines as serotonin agonists and antagonists |
| PL366060A1 (en) | 2000-12-20 | 2005-01-24 | Bristol-Myers Squibb Pharma Company | Substituted pyrazinoquinoxaline derivatives as serotonin receptor agonists and antagonists |
| MXPA03005438A (es) | 2000-12-20 | 2004-05-05 | Bristol Myers Squibb Co | Pirioindoles substituidos como agonistas y antagonistas de serotonina. |
| EP1347979B1 (en) | 2000-12-27 | 2007-08-08 | F. Hoffmann-La Roche Ag | Indole derivatives and their use as 5-ht2b and 5-ht2c receptor ligands |
| JP4680483B2 (ja) | 2001-02-23 | 2011-05-11 | ユニバーシティ オブ ピッツバーグ オブ ザ コモンウェルス システム オブ ハイヤー エデュケイション | 組織および臓器の治療および修復に用いるための幹細胞マトリックスの迅速な調製方法 |
| GB0106177D0 (en) | 2001-03-13 | 2001-05-02 | Hoffmann La Roche | Piperazine derivatives |
| JPWO2002074746A1 (ja) | 2001-03-16 | 2004-07-08 | 山之内製薬株式会社 | ベンゾアゼピン誘導体 |
| AU2002314744A1 (en) | 2001-04-17 | 2002-10-28 | Sepracor, Inc. | Thiazole and other heterocyclic ligands and use thereof |
| BR0210238A (pt) | 2001-06-01 | 2004-07-20 | Alcon Inc | Novos análogos de aril-amino-propano e seu uso para o tratamento de glaucoma |
| WO2002098860A1 (en) | 2001-06-01 | 2002-12-12 | Alcon, Inc. | Novel fused indazoles and indoles and their use for the treatment of glaucoma |
| MXPA03010807A (es) | 2001-06-01 | 2004-11-22 | Alcon Inc | Piranoidazoles y su uso para el tratamiento del glaucoma. |
| US6825198B2 (en) | 2001-06-21 | 2004-11-30 | Pfizer Inc | 5-HT receptor ligands and uses thereof |
| CA2451606A1 (en) | 2001-07-05 | 2003-01-16 | Pharmacia & Upjohn Company | (hetero) aryl substituted benzofurans as 5-ht ligands |
| JP2005522408A (ja) | 2001-07-13 | 2005-07-28 | ファルマシア・アンド・アップジョン・カンパニー・エルエルシー | 5−HT受容体リガントとしてのヘキサヒドロアゼピノ(4,5−g)インドールおよびインドリン |
| US6667303B1 (en) | 2001-07-30 | 2003-12-23 | Wyeth | Aryl substituted 1,4-diazepanes and method of use thereof |
| GB0118892D0 (en) | 2001-08-02 | 2001-09-26 | Vernalis Res Ltd | Method of treatment |
| US6720316B2 (en) | 2001-08-06 | 2004-04-13 | Pharmacia & Upjohn Company | Therapeutic 5-HT ligand compounds |
| US20030060462A1 (en) | 2001-08-06 | 2003-03-27 | Hoffman Robert L. | Therapeutic compounds |
| EP1414819B1 (en) | 2001-08-08 | 2005-10-12 | Pharmacia & Upjohn Company LLC | THERAPEUTIC 1H-PYRIDO[4,3-b]INDOLES |
| AU2002336749A1 (en) | 2001-09-21 | 2003-04-01 | Pharmacia And Upjohn Company | Tricyclic indole derivatives as 5-ht ligands |
| CA2464412A1 (en) | 2001-10-18 | 2003-04-24 | Pharmacia & Upjohn Company | Tetracyclic azaindoles and -indolines having serotonin 5-ht2cactivity |
| US20030118633A1 (en) * | 2001-11-09 | 2003-06-26 | Ebrahim Versi | Combination therapy |
| EP1456178A1 (en) | 2001-12-21 | 2004-09-15 | Smithkline Beecham Plc | 7-sulfonyl-3-benzazepine derivatives as modulators of the dopamine receptor and their use for the treatment of cns disorders |
| AU2002367323A1 (en) | 2001-12-28 | 2003-07-24 | Bayer Pharmaceuticals Corporation | Benzothieno (3,2- |
| AU2002364035A1 (en) | 2001-12-28 | 2003-07-24 | Bayer Pharmaceuticals Corporation | 4-sulfide / sulfoxide / sulfonyl-1h-pyrazolyl derivative compounds, for use in diseases associated with the 5-ht2c receptor |
| MXPA04006184A (es) | 2001-12-28 | 2004-12-06 | Bayer Pharmaceuticals Corp | Compuestos derivados de 1h-pirazolilo, para el uso en enfermedades asociadas con el receptor 5-ht2c. |
| AU2002360819A1 (en) | 2001-12-28 | 2003-07-24 | Bayer Corporation | Cyclohexano- and cycloheptapyrazole derivative compounds, for use in diseases associated with the 5-ht2c receptor |
| GB0202015D0 (en) | 2002-01-29 | 2002-03-13 | Hoffmann La Roche | Piperazine Derivatives |
| DOP2003000587A (es) | 2002-02-27 | 2003-08-30 | Pfizer Prod Inc | AGONISTAS DEL RECEPTOR ß3-ADRENERGICO |
| JP2003252863A (ja) * | 2002-03-05 | 2003-09-10 | Kyowa Hakko Kogyo Co Ltd | イミダゾリジン誘導体 |
| US6953787B2 (en) | 2002-04-12 | 2005-10-11 | Arena Pharmaceuticals, Inc. | 5HT2C receptor modulators |
| DE10217006A1 (de) | 2002-04-16 | 2003-11-06 | Merck Patent Gmbh | Substituierte Indole |
| WO2003089409A1 (en) | 2002-04-19 | 2003-10-30 | Glaxo Group Limited | Compounds having affinity at 5ht2c receptor and use thereof in therapy |
| TW200307540A (en) | 2002-04-25 | 2003-12-16 | Wyeth Corp | [1, 4]Diazocino[7, 8, 1-hi] indole derivatives as antipsychotic and antiobesity agents |
| TW200307682A (en) | 2002-04-25 | 2003-12-16 | Wyeth Corp | 1,2,3,4,7,8-Hexahydro-6H-[1,4]diazepino[6,7,1-ij]quinoline derivatives as antipsychotic and antiobesity agents |
| TWI312781B (en) | 2002-04-25 | 2009-08-01 | [1,4]diazepino[6,7,1-ij]quinoline derivatives as antipsychotic and antiobesity agents | |
| ATE398119T1 (de) | 2002-06-19 | 2008-07-15 | Biovitrum Ab Publ | Piperazinylpyrazine als modulatoren des serotonin 5-ht2c rezeptors |
| WO2004000830A1 (en) | 2002-06-19 | 2003-12-31 | Biovitrum Ab | Novel compounds, their use and preparation |
| TW200404550A (en) | 2002-07-08 | 2004-04-01 | Sankyo Co | Cepharospolin formulation for oral use |
| JP2004159919A (ja) | 2002-11-13 | 2004-06-10 | Takeda Chem Ind Ltd | 腹圧性尿失禁予防治療薬のスクリーニング方法 |
| ATE509929T1 (de) | 2002-12-19 | 2011-06-15 | Bristol Myers Squibb Co | Substituierte tricyclische gamma-carbolineals serotonin-rezeptor-agonisten und antagonisten |
| GB0305024D0 (en) | 2003-03-05 | 2003-04-09 | Glaxo Group Ltd | Compounds |
| GB0305553D0 (en) | 2003-03-11 | 2003-04-16 | Glaxo Group Ltd | Compounds |
| US20040192754A1 (en) | 2003-03-24 | 2004-09-30 | Shapira Nathan Andrew | Methods for treating idiopathic hyperhidrosis and associated conditions |
| DE10315285A1 (de) | 2003-04-04 | 2004-10-14 | Merck Patent Gmbh | Chromenonindole |
| AU2004226838B8 (en) | 2003-04-04 | 2009-06-11 | H. Lundbeck A/S | 4-(2-phenylsulfanyl-phenyl)-piperidine derivatives as serotonin reuptake inhibitors |
| UA81300C2 (en) | 2003-04-04 | 2007-12-25 | Lundbeck & Co As H | Derivates of 4-(2-fenilsulfanilfenil)-1,2,3,6-tetrahydropiridin as retarding agents of serotonin recapture |
| GB0308025D0 (en) | 2003-04-07 | 2003-05-14 | Glaxo Group Ltd | Compounds |
| CL2004000826A1 (es) | 2003-04-25 | 2005-03-04 | Pfizer | Uso de un agonista para el receptor 5-ht2c para preparar un medicamento util en el tratamiento de la incontinencia urinaria provocada por estres, con la condicion de que el agonista no sea 1-[6-cloro-5-(trifluorometil)-2-piridinil]piperazina (org-129 |
| WO2004096201A1 (en) | 2003-04-29 | 2004-11-11 | Orexigen Therapeutics, Inc. | Compositions for affecting weight loss |
| US20050004164A1 (en) | 2003-04-30 | 2005-01-06 | Caggiano Thomas J. | 2-Cyanopropanoic acid amide and ester derivatives and methods of their use |
| MXPA05013366A (es) | 2003-06-17 | 2006-04-05 | Arena Pharm Inc | Derivados de benzazepina utiles para el tratamiento de enfermedades asociadas con el receptor 5ht2c. |
| JP2007523861A (ja) | 2003-06-20 | 2007-08-23 | アリーナ ファーマシューティカルズ, インコーポレイテッド | N−フェニル−ピペラジン誘導体および5ht2cレセプター関連疾患の予防方法または処置方法 |
| HUP0301906A3 (en) | 2003-06-23 | 2006-03-28 | Egis Gyogyszergyar Nyilvanosan | Use of bicyclo[2.2.1]heptane derivatives for producing of pharmaceutical compositions having neuroprotectiv activity |
| GB0314967D0 (en) | 2003-06-26 | 2003-07-30 | Hoffmann La Roche | Piperazine derivatives |
| EP1500391A1 (en) | 2003-07-24 | 2005-01-26 | Neuro3D | Therapeutic use of bicycloheptane derivatives |
| TW200510324A (en) | 2003-08-11 | 2005-03-16 | Lilly Co Eli | 6-(2,2,2-trifluoroethylamino)-7-chiloro-2, 3, 4, 5-tetrahydro-1h-benzo[d]azepine as a 5-ht2c receptor agonist |
| US7244843B2 (en) | 2003-10-07 | 2007-07-17 | Bristol-Myers Squibb Company | Modulators of serotonin receptors |
| WO2005042491A1 (en) | 2003-10-22 | 2005-05-12 | Arena Pharmaceuticals, Inc. | Benzazepine derivatives and methods of prophylaxis or treatment of 5ht2c receptor associated diseases |
| WO2005042490A1 (en) | 2003-10-22 | 2005-05-12 | Arena Pharmaceuticals, Inc. | Benzazepine derivatives and methods of prophylaxis or treatment of 5ht2c receptor associated diseases |
| US7435837B2 (en) | 2003-10-24 | 2008-10-14 | Wyeth | Dihydrobenzofuranyl alkanamine derivatives and methods for using same |
| US7728155B2 (en) | 2003-10-24 | 2010-06-01 | Wyeth Llc | Dihydrobenzofuranyl alkanamines and methods for using same as cns agents |
| HRP20030885A2 (en) | 2003-11-03 | 2005-08-31 | Pliva-Istra�iva�ki institut d.o.o. | USE OF 2-THIA-DIBENZO[e,h]AZULENES FOR THE MANUFACTURE OF PHARMACEUTICAL FORMULATIONS FOR THE TREATMENT AND PREVENTION OF CENTRAL NERVOUS SYYTEM DISEASES AND DISORDERS |
| WO2012000510A1 (en) | 2010-06-29 | 2012-01-05 | Vestas Wind Systems A/S | Method and system for monitoring structural health of a filter in a wind turbine, and a wind turbine |
-
2005
- 2005-08-24 US US11/660,751 patent/US8685924B2/en not_active Expired - Fee Related
- 2005-08-24 EP EP05781418A patent/EP1792629A4/en not_active Withdrawn
- 2005-08-24 EP EP11181877A patent/EP2400300A1/en not_active Withdrawn
- 2005-08-24 EP EP10174167A patent/EP2248524A3/en not_active Ceased
- 2005-08-24 WO PCT/JP2005/015830 patent/WO2006022420A1/ja not_active Ceased
- 2005-08-24 JP JP2006532742A patent/JP5173190B2/ja not_active Expired - Fee Related
-
2011
- 2011-03-07 JP JP2011049685A patent/JP5341933B2/ja not_active Expired - Fee Related
-
2014
- 2014-01-16 US US14/156,677 patent/US20140155369A1/en not_active Abandoned
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998033504A1 (en) * | 1997-02-03 | 1998-08-06 | Akzo Nobel N.V. | Treatment of urinary incontinence |
| WO2000076984A2 (en) * | 1999-05-21 | 2000-12-21 | Biovitrum Ab | Novel compounds, their use and preparation |
| WO2003097636A1 (en) * | 2002-05-17 | 2003-11-27 | Biovitrum Ab | Novel compounds and their use |
Non-Patent Citations (4)
| Title |
|---|
| KITAMURA M.: "Byoki to Kusuri, Hainyo Shogai to Kusuri", PHARMACIA, vol. 38, no. 7, 2002, pages 665 - 667, XP002999882 * |
| LEYSEN D.C.M. ET AL: "Selective 5-HT2C agonists as potential antidepressants", IDRUGS, vol. 2, no. 2, 1999, pages 109 - 120, XP000922503 * |
| See also references of EP1792629A4 * |
| STEERS W.D. ET AL: "Effects of m-chlorophenylpiperazine on penile and bladder function in rats", AMERICAN JOURNAL OF PHYSIOLOGY, vol. 257, no. 6, 1989, pages R1441 - R1449, XP000654934 * |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8158617B2 (en) | 2006-05-16 | 2012-04-17 | Takeda Pharmaceutical Company Limited | Fused heterocyclic compound and use thereof |
| US9079907B2 (en) | 2006-05-16 | 2015-07-14 | Takeda Pharmaceutical Company Limited | Fused heterocyclic compound and use thereof |
| EP2742936A1 (en) * | 2006-05-16 | 2014-06-18 | Takeda Pharmaceutical Company Limited | Fused heterocyclic compound and use thereof |
| JPWO2007132841A1 (ja) * | 2006-05-16 | 2009-09-24 | 武田薬品工業株式会社 | 縮合複素環化合物およびその用途 |
| WO2007132841A1 (ja) * | 2006-05-16 | 2007-11-22 | Takeda Pharmaceutical Company Limited | 縮合複素環化合物およびその用途 |
| JP2014088449A (ja) * | 2006-05-16 | 2014-05-15 | Takeda Chem Ind Ltd | 縮合複素環化合物およびその用途 |
| JP5497429B2 (ja) * | 2007-03-07 | 2014-05-21 | 武田薬品工業株式会社 | ベンゾオキサゼピン誘導体およびその用途 |
| EP2123644A4 (en) * | 2007-03-07 | 2011-06-29 | Takeda Pharmaceutical | BENZOXAZEPINE DERIVATIVES AND THEIR USE |
| US8247403B2 (en) | 2007-03-07 | 2012-08-21 | Takeda Pharmaceutical Company Limited | Benzoxazepine derivatives and use thereof |
| EP2009374A2 (en) | 2007-06-28 | 2008-12-31 | Whirlpool Corporation | Distributed refrigeration system with custom storage modules |
| EP2216023A4 (en) * | 2007-11-15 | 2013-03-13 | Takeda Pharmaceutical | CONDENSED PYRIDINE DERIVATIVE AND ITS USE |
| JP5520051B2 (ja) * | 2007-11-15 | 2014-06-11 | 武田薬品工業株式会社 | 縮合ピリジン誘導体およびその用途 |
| WO2009063992A1 (ja) * | 2007-11-15 | 2009-05-22 | Takeda Pharmaceutical Company Limited | 縮合ピリジン誘導体およびその用途 |
| US8480437B2 (en) | 2008-04-11 | 2013-07-09 | Bal Seal Engineering, Inc. | Connector cartridge stack for electrical transmission |
| JP2009273396A (ja) * | 2008-05-14 | 2009-11-26 | Mitsubishi Chemical Medience Corp | 腹圧性尿失禁症モデル動物の作製方法 |
| WO2011071136A1 (ja) | 2009-12-11 | 2011-06-16 | アステラス製薬株式会社 | 線維筋痛症治療剤 |
| JPWO2013133325A1 (ja) * | 2012-03-06 | 2015-07-30 | 武田薬品工業株式会社 | 三環性化合物 |
| JP2017531012A (ja) * | 2014-10-16 | 2017-10-19 | ジーティーエックス・インコーポレイテッド | Sarmを使用した泌尿器系障害の治療方法 |
| WO2019131902A1 (ja) | 2017-12-27 | 2019-07-04 | 武田薬品工業株式会社 | 腹圧性尿失禁および便失禁の治療薬 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20070274913A1 (en) | 2007-11-29 |
| EP2400300A1 (en) | 2011-12-28 |
| EP1792629A4 (en) | 2010-08-25 |
| US20140155369A1 (en) | 2014-06-05 |
| JP2011141287A (ja) | 2011-07-21 |
| JPWO2006022420A1 (ja) | 2008-05-08 |
| EP2248524A3 (en) | 2011-03-09 |
| EP1792629A1 (en) | 2007-06-06 |
| EP2248524A2 (en) | 2010-11-10 |
| JP5173190B2 (ja) | 2013-03-27 |
| US8685924B2 (en) | 2014-04-01 |
| JP5341933B2 (ja) | 2013-11-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5341933B2 (ja) | 腹圧性尿失禁の予防・治療剤及びそのスクリーニング方法 | |
| US20220152052A1 (en) | Methods and compositions for treating various disorders | |
| RU2336871C2 (ru) | Применение соединений, которые являются эффективными как селективные модуляторы опиатного рецептора | |
| JP2004531468A5 (ja) | ||
| AU2018390990B2 (en) | Treatment of pain and/or pain related symptoms associated with dysmenorrhea | |
| TW202114654A (zh) | 包含如sglt 1/2抑制劑的sglt抑制劑之治療 | |
| US20140045788A1 (en) | Method of stably treating incontinence using a bulking agent | |
| WO2024054412A1 (en) | Methods and compositions for treating amyotrophic lateral sclerosis | |
| HK40081935A (en) | Treatment of amyotrophic lateral sclerosis | |
| Brethauer et al. | William Carey, MD | |
| HK1079451B (en) | Use of compounds that are effective as selective opiate receptor modulators | |
| NZ614725B2 (en) | Methods and compositions for treating depression using cyclobenzaprine |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU LV MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2006532742 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11660751 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005781418 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005781418 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 11660751 Country of ref document: US |