JP5658561B2 - ナノ表面 - Google Patents
ナノ表面 Download PDFInfo
- Publication number
- JP5658561B2 JP5658561B2 JP2010515496A JP2010515496A JP5658561B2 JP 5658561 B2 JP5658561 B2 JP 5658561B2 JP 2010515496 A JP2010515496 A JP 2010515496A JP 2010515496 A JP2010515496 A JP 2010515496A JP 5658561 B2 JP5658561 B2 JP 5658561B2
- Authority
- JP
- Japan
- Prior art keywords
- implant
- range
- titanium
- biocompatible
- nanostructure
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000007943 implant Substances 0.000 claims abstract description 183
- 239000002086 nanomaterial Substances 0.000 claims abstract description 110
- 210000000988 bone and bone Anatomy 0.000 claims abstract description 68
- 239000000758 substrate Substances 0.000 claims abstract description 13
- 238000000034 method Methods 0.000 claims description 87
- 239000010936 titanium Substances 0.000 claims description 82
- 229910052719 titanium Inorganic materials 0.000 claims description 79
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 claims description 78
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 74
- 239000000203 mixture Substances 0.000 claims description 51
- 229910044991 metal oxide Inorganic materials 0.000 claims description 34
- 150000004706 metal oxides Chemical class 0.000 claims description 34
- 239000000126 substance Substances 0.000 claims description 33
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 claims description 29
- 235000006408 oxalic acid Nutrition 0.000 claims description 26
- 238000011282 treatment Methods 0.000 claims description 26
- 239000000463 material Substances 0.000 claims description 18
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 17
- 230000008468 bone growth Effects 0.000 claims description 15
- 230000008569 process Effects 0.000 claims description 15
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 14
- 229910021645 metal ion Inorganic materials 0.000 claims description 14
- 238000005422 blasting Methods 0.000 claims description 13
- 230000002708 enhancing effect Effects 0.000 claims description 13
- 239000012298 atmosphere Substances 0.000 claims description 11
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 11
- 239000001301 oxygen Substances 0.000 claims description 11
- 229910052760 oxygen Inorganic materials 0.000 claims description 11
- 229910001416 lithium ion Inorganic materials 0.000 claims description 10
- 238000012545 processing Methods 0.000 claims description 10
- 229910001427 strontium ion Inorganic materials 0.000 claims description 10
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 claims description 9
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 8
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 8
- 239000002253 acid Substances 0.000 claims description 8
- 239000000460 chlorine Substances 0.000 claims description 8
- 229910052801 chlorine Inorganic materials 0.000 claims description 8
- 229910052731 fluorine Inorganic materials 0.000 claims description 8
- 239000011737 fluorine Substances 0.000 claims description 8
- 238000004381 surface treatment Methods 0.000 claims description 8
- 150000003839 salts Chemical class 0.000 claims description 7
- 230000000399 orthopedic effect Effects 0.000 claims description 6
- BHPQYMZQTOCNFJ-UHFFFAOYSA-N Calcium cation Chemical compound [Ca+2] BHPQYMZQTOCNFJ-UHFFFAOYSA-N 0.000 claims description 5
- JLVVSXFLKOJNIY-UHFFFAOYSA-N Magnesium ion Chemical compound [Mg+2] JLVVSXFLKOJNIY-UHFFFAOYSA-N 0.000 claims description 5
- 229910001069 Ti alloy Inorganic materials 0.000 claims description 5
- 229910001424 calcium ion Inorganic materials 0.000 claims description 5
- 238000004140 cleaning Methods 0.000 claims description 5
- 229910001425 magnesium ion Inorganic materials 0.000 claims description 5
- SOQBVABWOPYFQZ-UHFFFAOYSA-N oxygen(2-);titanium(4+) Chemical class [O-2].[O-2].[Ti+4] SOQBVABWOPYFQZ-UHFFFAOYSA-N 0.000 claims description 4
- -1 titanium ions Chemical class 0.000 claims description 4
- 230000004913 activation Effects 0.000 claims description 3
- 150000002500 ions Chemical class 0.000 claims description 3
- LCKIEQZJEYYRIY-UHFFFAOYSA-N Titanium ion Chemical compound [Ti+4] LCKIEQZJEYYRIY-UHFFFAOYSA-N 0.000 claims description 2
- 238000005238 degreasing Methods 0.000 claims description 2
- PWYYWQHXAPXYMF-UHFFFAOYSA-N strontium(2+) Chemical compound [Sr+2] PWYYWQHXAPXYMF-UHFFFAOYSA-N 0.000 claims 2
- 230000001568 sexual effect Effects 0.000 claims 1
- 238000010883 osseointegration Methods 0.000 abstract description 15
- 230000003993 interaction Effects 0.000 abstract description 5
- 239000000523 sample Substances 0.000 description 63
- 210000004027 cell Anatomy 0.000 description 35
- 238000012360 testing method Methods 0.000 description 28
- 229910052751 metal Inorganic materials 0.000 description 20
- 239000002184 metal Substances 0.000 description 20
- 239000012890 simulated body fluid Substances 0.000 description 20
- 238000009826 distribution Methods 0.000 description 18
- 230000015572 biosynthetic process Effects 0.000 description 15
- 238000002513 implantation Methods 0.000 description 15
- 238000001000 micrograph Methods 0.000 description 15
- 239000000243 solution Substances 0.000 description 15
- 241000283973 Oryctolagus cuniculus Species 0.000 description 14
- 230000000694 effects Effects 0.000 description 13
- 210000000963 osteoblast Anatomy 0.000 description 13
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 13
- 229910052586 apatite Inorganic materials 0.000 description 12
- 238000004519 manufacturing process Methods 0.000 description 12
- VSIIXMUUUJUKCM-UHFFFAOYSA-D pentacalcium;fluoride;triphosphate Chemical compound [F-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O VSIIXMUUUJUKCM-UHFFFAOYSA-D 0.000 description 12
- 230000003746 surface roughness Effects 0.000 description 11
- 210000001519 tissue Anatomy 0.000 description 11
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 10
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 10
- 238000007654 immersion Methods 0.000 description 10
- 239000010410 layer Substances 0.000 description 10
- 229910045601 alloy Inorganic materials 0.000 description 9
- 239000000956 alloy Substances 0.000 description 9
- 239000011575 calcium Substances 0.000 description 9
- XEYBRNLFEZDVAW-ARSRFYASSA-N dinoprostone Chemical compound CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O XEYBRNLFEZDVAW-ARSRFYASSA-N 0.000 description 9
- 230000001965 increasing effect Effects 0.000 description 9
- 230000011164 ossification Effects 0.000 description 9
- 238000001878 scanning electron micrograph Methods 0.000 description 9
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 8
- 238000000576 coating method Methods 0.000 description 8
- 229960002986 dinoprostone Drugs 0.000 description 8
- 229910052588 hydroxylapatite Inorganic materials 0.000 description 8
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical compound [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 description 8
- XEYBRNLFEZDVAW-UHFFFAOYSA-N prostaglandin E2 Natural products CCCCCC(O)C=CC1C(O)CC(=O)C1CC=CCCCC(O)=O XEYBRNLFEZDVAW-UHFFFAOYSA-N 0.000 description 8
- 239000007864 aqueous solution Substances 0.000 description 7
- 239000012620 biological material Substances 0.000 description 7
- 230000035876 healing Effects 0.000 description 7
- 229910052744 lithium Inorganic materials 0.000 description 7
- QPJSUIGXIBEQAC-UHFFFAOYSA-N n-(2,4-dichloro-5-propan-2-yloxyphenyl)acetamide Chemical compound CC(C)OC1=CC(NC(C)=O)=C(Cl)C=C1Cl QPJSUIGXIBEQAC-UHFFFAOYSA-N 0.000 description 7
- 229910052712 strontium Inorganic materials 0.000 description 7
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 7
- 238000012876 topography Methods 0.000 description 7
- 239000001506 calcium phosphate Substances 0.000 description 6
- 238000011555 rabbit model Methods 0.000 description 6
- 239000013074 reference sample Substances 0.000 description 6
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 5
- 229910000389 calcium phosphate Inorganic materials 0.000 description 5
- 235000011010 calcium phosphates Nutrition 0.000 description 5
- 230000004663 cell proliferation Effects 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 238000004090 dissolution Methods 0.000 description 5
- 238000002389 environmental scanning electron microscopy Methods 0.000 description 5
- 238000011156 evaluation Methods 0.000 description 5
- 229910052758 niobium Inorganic materials 0.000 description 5
- 239000010955 niobium Substances 0.000 description 5
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 description 5
- 238000010915 one-step procedure Methods 0.000 description 5
- 230000002829 reductive effect Effects 0.000 description 5
- 229910052715 tantalum Inorganic materials 0.000 description 5
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 5
- 229910052726 zirconium Inorganic materials 0.000 description 5
- 241001132374 Asta Species 0.000 description 4
- 238000004113 cell culture Methods 0.000 description 4
- 230000000875 corresponding effect Effects 0.000 description 4
- 239000004053 dental implant Substances 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 229910052735 hafnium Inorganic materials 0.000 description 4
- VBJZVLUMGGDVMO-UHFFFAOYSA-N hafnium atom Chemical compound [Hf] VBJZVLUMGGDVMO-UHFFFAOYSA-N 0.000 description 4
- 238000000338 in vitro Methods 0.000 description 4
- 238000011068 loading method Methods 0.000 description 4
- 102000004169 proteins and genes Human genes 0.000 description 4
- 108090000623 proteins and genes Proteins 0.000 description 4
- 230000004044 response Effects 0.000 description 4
- 238000001356 surgical procedure Methods 0.000 description 4
- 210000000689 upper leg Anatomy 0.000 description 4
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 3
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 3
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 241000607479 Yersinia pestis Species 0.000 description 3
- 230000001464 adherent effect Effects 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 210000001185 bone marrow Anatomy 0.000 description 3
- 210000002805 bone matrix Anatomy 0.000 description 3
- 230000002308 calcification Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 239000008367 deionised water Substances 0.000 description 3
- 229910021641 deionized water Inorganic materials 0.000 description 3
- 238000000151 deposition Methods 0.000 description 3
- 238000013461 design Methods 0.000 description 3
- 238000002845 discoloration Methods 0.000 description 3
- 230000004821 effect on bone Effects 0.000 description 3
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 3
- 238000005530 etching Methods 0.000 description 3
- 210000002744 extracellular matrix Anatomy 0.000 description 3
- 239000007769 metal material Substances 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 238000013425 morphometry Methods 0.000 description 3
- 239000002159 nanocrystal Substances 0.000 description 3
- 210000002997 osteoclast Anatomy 0.000 description 3
- 230000003239 periodontal effect Effects 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 230000035755 proliferation Effects 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 210000004872 soft tissue Anatomy 0.000 description 3
- 230000001954 sterilising effect Effects 0.000 description 3
- 238000004659 sterilization and disinfection Methods 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- 239000007995 HEPES buffer Substances 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 229910002091 carbon monoxide Inorganic materials 0.000 description 2
- 230000024245 cell differentiation Effects 0.000 description 2
- 230000003915 cell function Effects 0.000 description 2
- 239000011247 coating layer Substances 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 238000005553 drilling Methods 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 239000010931 gold Substances 0.000 description 2
- 229910052737 gold Inorganic materials 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000010874 in vitro model Methods 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 239000011261 inert gas Substances 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 238000003780 insertion Methods 0.000 description 2
- 230000037431 insertion Effects 0.000 description 2
- 230000001788 irregular Effects 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 210000005009 osteogenic cell Anatomy 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 238000007788 roughening Methods 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 238000004544 sputter deposition Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- AZUYLZMQTIKGSC-UHFFFAOYSA-N 1-[6-[4-(5-chloro-6-methyl-1H-indazol-4-yl)-5-methyl-3-(1-methylindazol-5-yl)pyrazol-1-yl]-2-azaspiro[3.3]heptan-2-yl]prop-2-en-1-one Chemical compound ClC=1C(=C2C=NNC2=CC=1C)C=1C(=NN(C=1C)C1CC2(CN(C2)C(C=C)=O)C1)C=1C=C2C=NN(C2=CC=1)C AZUYLZMQTIKGSC-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- XZKIHKMTEMTJQX-UHFFFAOYSA-N 4-Nitrophenyl Phosphate Chemical compound OP(O)(=O)OC1=CC=C([N+]([O-])=O)C=C1 XZKIHKMTEMTJQX-UHFFFAOYSA-N 0.000 description 1
- 238000008940 Alkaline Phosphatase assay kit Methods 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 102000007350 Bone Morphogenetic Proteins Human genes 0.000 description 1
- 108010007726 Bone Morphogenetic Proteins Proteins 0.000 description 1
- 208000006386 Bone Resorption Diseases 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 238000008157 ELISA kit Methods 0.000 description 1
- 208000002354 Edentulous Jaw Diseases 0.000 description 1
- 229910001200 Ferrotitanium Inorganic materials 0.000 description 1
- SXRSQZLOMIGNAQ-UHFFFAOYSA-N Glutaraldehyde Chemical compound O=CCCCC=O SXRSQZLOMIGNAQ-UHFFFAOYSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 238000000692 Student's t-test Methods 0.000 description 1
- 229910010413 TiO 2 Inorganic materials 0.000 description 1
- 229910000756 V alloy Inorganic materials 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 238000010306 acid treatment Methods 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 210000001789 adipocyte Anatomy 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 210000003484 anatomy Anatomy 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 210000002798 bone marrow cell Anatomy 0.000 description 1
- 229940112869 bone morphogenetic protein Drugs 0.000 description 1
- 230000010072 bone remodeling Effects 0.000 description 1
- 230000024279 bone resorption Effects 0.000 description 1
- 210000000845 cartilage Anatomy 0.000 description 1
- 230000021164 cell adhesion Effects 0.000 description 1
- 230000006037 cell lysis Effects 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 238000005524 ceramic coating Methods 0.000 description 1
- 229910010293 ceramic material Inorganic materials 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- HBXWYZMULLEJSG-UHFFFAOYSA-N chromium vanadium Chemical compound [V][Cr][V][Cr] HBXWYZMULLEJSG-UHFFFAOYSA-N 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 239000008139 complexing agent Substances 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 230000001054 cortical effect Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 239000012894 fetal calf serum Substances 0.000 description 1
- 239000000834 fixative Substances 0.000 description 1
- 210000001650 focal adhesion Anatomy 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 238000003018 immunoassay Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 238000007373 indentation Methods 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 102000006495 integrins Human genes 0.000 description 1
- 108010044426 integrins Proteins 0.000 description 1
- 238000004573 interface analysis Methods 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 230000004068 intracellular signaling Effects 0.000 description 1
- 229910052743 krypton Inorganic materials 0.000 description 1
- DNNSSWSSYDEUBZ-UHFFFAOYSA-N krypton atom Chemical compound [Kr] DNNSSWSSYDEUBZ-UHFFFAOYSA-N 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000002932 luster Substances 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 238000010297 mechanical methods and process Methods 0.000 description 1
- 230000005226 mechanical processes and functions Effects 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 150000002736 metal compounds Chemical class 0.000 description 1
- 238000007491 morphometric analysis Methods 0.000 description 1
- 230000003562 morphometric effect Effects 0.000 description 1
- 239000002105 nanoparticle Substances 0.000 description 1
- 229910052754 neon Inorganic materials 0.000 description 1
- GKAOGPIIYCISHV-UHFFFAOYSA-N neon atom Chemical compound [Ne] GKAOGPIIYCISHV-UHFFFAOYSA-N 0.000 description 1
- 238000011587 new zealand white rabbit Methods 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 229910052755 nonmetal Inorganic materials 0.000 description 1
- 230000006911 nucleation Effects 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 229910000489 osmium tetroxide Inorganic materials 0.000 description 1
- 239000012285 osmium tetroxide Substances 0.000 description 1
- 230000004072 osteoblast differentiation Effects 0.000 description 1
- 238000000059 patterning Methods 0.000 description 1
- 230000035479 physiological effects, processes and functions Effects 0.000 description 1
- 238000007750 plasma spraying Methods 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 229910052704 radon Inorganic materials 0.000 description 1
- SYUHGPGVQRZVTB-UHFFFAOYSA-N radon atom Chemical compound [Rn] SYUHGPGVQRZVTB-UHFFFAOYSA-N 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 230000024155 regulation of cell adhesion Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000010405 reoxidation reaction Methods 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- 239000002276 single tooth dental implant Substances 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 210000001082 somatic cell Anatomy 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 238000012414 sterilization procedure Methods 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 238000007669 thermal treatment Methods 0.000 description 1
- 230000008467 tissue growth Effects 0.000 description 1
- 229910000391 tricalcium phosphate Inorganic materials 0.000 description 1
- 235000019731 tricalcium phosphate Nutrition 0.000 description 1
- 229940078499 tricalcium phosphate Drugs 0.000 description 1
- 238000010246 ultrastructural analysis Methods 0.000 description 1
- 238000009827 uniform distribution Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 229910052724 xenon Inorganic materials 0.000 description 1
- FHNFHKCVQCLJFQ-UHFFFAOYSA-N xenon atom Chemical compound [Xe] FHNFHKCVQCLJFQ-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30767—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C8/00—Means to be fixed to the jaw-bone for consolidating natural teeth or for fixing dental prostheses thereon; Dental implants; Implanting tools
- A61C8/0012—Means to be fixed to the jaw-bone for consolidating natural teeth or for fixing dental prostheses thereon; Dental implants; Implanting tools characterised by the material or composition, e.g. ceramics, surface layer, metal alloy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/28—Materials for coating prostheses
- A61L27/30—Inorganic materials
- A61L27/306—Other specific inorganic materials not covered by A61L27/303 - A61L27/32
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y30/00—Nanotechnology for materials or surface science, e.g. nanocomposites
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y5/00—Nanobiotechnology or nanomedicine, e.g. protein engineering or drug delivery
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/46—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 containing oxalates
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C26/00—Coating not provided for in groups C23C2/00 - C23C24/00
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C8/00—Solid state diffusion of only non-metal elements into metallic material surfaces; Chemical surface treatment of metallic material by reaction of the surface with a reactive gas, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C8/00—Solid state diffusion of only non-metal elements into metallic material surfaces; Chemical surface treatment of metallic material by reaction of the surface with a reactive gas, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C8/06—Solid state diffusion of only non-metal elements into metallic material surfaces; Chemical surface treatment of metallic material by reaction of the surface with a reactive gas, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using gases
- C23C8/08—Solid state diffusion of only non-metal elements into metallic material surfaces; Chemical surface treatment of metallic material by reaction of the surface with a reactive gas, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using gases only one element being applied
- C23C8/10—Oxidising
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23G—CLEANING OR DE-GREASING OF METALLIC MATERIAL BY CHEMICAL METHODS OTHER THAN ELECTROLYSIS
- C23G1/00—Cleaning or pickling metallic material with solutions or molten salts
- C23G1/02—Cleaning or pickling metallic material with solutions or molten salts with acid solutions
- C23G1/10—Other heavy metals
- C23G1/106—Other heavy metals refractory metals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C8/00—Means to be fixed to the jaw-bone for consolidating natural teeth or for fixing dental prostheses thereon; Dental implants; Implanting tools
- A61C8/0018—Means to be fixed to the jaw-bone for consolidating natural teeth or for fixing dental prostheses thereon; Dental implants; Implanting tools characterised by the shape
- A61C8/0037—Details of the shape
- A61C2008/0046—Textured surface, e.g. roughness, microstructure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/3094—Designing or manufacturing processes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30667—Features concerning an interaction with the environment or a particular use of the prosthesis
- A61F2002/30719—Means for cleaning prostheses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30767—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth
- A61F2002/30906—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth shot- sand- or grit-blasted
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30767—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth
- A61F2002/30925—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth etched
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2/30767—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth
- A61F2002/30929—Special external or bone-contacting surface, e.g. coating for improving bone ingrowth having at least two superposed coatings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0058—Additional features; Implant or prostheses properties not otherwise provided for
- A61F2250/0092—Means for cleaning prostheses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2310/00—Prostheses classified in A61F2/28 or A61F2/30 - A61F2/44 being constructed from or coated with a particular material
- A61F2310/00005—The prosthesis being constructed from a particular material
- A61F2310/00011—Metals or alloys
- A61F2310/00023—Titanium or titanium-based alloys, e.g. Ti-Ni alloys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2310/00—Prostheses classified in A61F2/28 or A61F2/30 - A61F2/44 being constructed from or coated with a particular material
- A61F2310/00389—The prosthesis being coated or covered with a particular material
- A61F2310/00592—Coating or prosthesis-covering structure made of ceramics or of ceramic-like compounds
- A61F2310/00598—Coating or prosthesis-covering structure made of compounds based on metal oxides or hydroxides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2310/00—Prostheses classified in A61F2/28 or A61F2/30 - A61F2/44 being constructed from or coated with a particular material
- A61F2310/00389—The prosthesis being coated or covered with a particular material
- A61F2310/00592—Coating or prosthesis-covering structure made of ceramics or of ceramic-like compounds
- A61F2310/00598—Coating or prosthesis-covering structure made of compounds based on metal oxides or hydroxides
- A61F2310/00616—Coating made of titanium oxide or hydroxides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2400/00—Materials characterised by their function or physical properties
- A61L2400/12—Nanosized materials, e.g. nanofibres, nanoparticles, nanowires, nanotubes; Nanostructured surfaces
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Nanotechnology (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Transplantation (AREA)
- Epidemiology (AREA)
- Medicinal Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Crystallography & Structural Chemistry (AREA)
- Molecular Biology (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Medical Informatics (AREA)
- Biotechnology (AREA)
- Pharmacology & Pharmacy (AREA)
- Biophysics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Dermatology (AREA)
- Ceramic Engineering (AREA)
- Inorganic Chemistry (AREA)
- Dentistry (AREA)
- Physics & Mathematics (AREA)
- Composite Materials (AREA)
- General Engineering & Computer Science (AREA)
- General Physics & Mathematics (AREA)
- Cardiology (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
Description
a)少なくとも部分的に金属酸化物で被覆された生体適合性部材を備えること;及び
b)該部材の部分が該金属酸化物で被覆された、その部材の少なくとも一部を、シュウ酸を含む水性組成物で処理すること;
を含み、そのことによって改質された金属酸化物が得られる、方法に関する。
c)i)イオン化されたフッ素及びイオン化された塩素を含むグループから選ばれる少なくとも1つの物質;及び
ii)少なくとも1つの酸;
を含む第二の水性組成物で、前記改質された酸化物の少なくとも部分を処理する工程を更に含む。
a)プラトー及び/又はリッジによって分離された微細ピットを含む微細構造;及び
b)該微細構造の上に重なっている一次ナノ構造、ここで該一次ナノ構造は波状の形に配置された窪みを含む;
を含む表面を有する基体を含む生体適合性部材に関する。
T ら、「Ultrastructural analysis of the interface zone of titanium and gold implants」、Advances in Biomaterials 4, 167-177, 1982; Albrektsson, Tら,「Interface analysis of titanium and zirconium bone implants」、Biomaterials 6, 97-101, 1985; Albrektsson T, Hansson, H-A,「An ultrastructural characterization of the interface between bone and sputtered titanium or stainless steel surfaces」、Biomaterials 7, 201-205, 1986; Hansson, H-A ら、「Structural aspects of the interface between tissue and titanium implants」、Journal of Prosthetic Dentistry 50, 108-113, 1983; Johansson, C ら、「Ultrastructural differences of the interface zone between bone and Ti6A14V or commercially pure titanium」、Journal of Biomedical Engineering 11, 3-8, 1989; Johansson, C. ら、「Qualitative, interfacial study between bone and tantalum, niobium or commercially pure titanium」、Biomaterials 11, 277-280, 1990; Sennerby, L ら、「Structure of the bone-titanium interface in retrieved clinical oral implants」、Clinical Oral Implants Research 2, 103-111, 1991; Sennerby, L ら、「Ultrastructure of the bone-titanium interface in rabits」、Journal of Materials Science: Material in Medicine 3, 262-271, 1992; Sennerby,
L ら、「Early tissue response to titanium implants inserted in rabit cortical bone, Part II: Ultrastructural observations」、Journal of Materials Science: Material in Medicine 4, 494-502, 1993)。微細構造及び一次ナノ構造を含む階層的表面トポグラフィーによって、部材と引続いて形成される骨組織との間の改善された機械的相互作用が与えられ、その結果、強度が低下したより薄い組織層が形成されると考えられる。二次ナノ構造によって、埋め込み後の生体適合性部材と周囲の骨組織間の機械的相互作用が更に改善される。従って、本発明の生体適合性部材によって、改善されたせん断強さ及び引張り強さを有する骨組織−インプラント界面が供される。
i)上記記載の生体適合性部材を備えること;及び
ii)該生体適合性部材をヒト又は動物の体にインプラントすること;
を含むインプラント方法に関する。
実施例 1−表面改質
(i)サンプル調製
コインの形状を有するチタンサンプル(各々機械加工されたものとブラスチングされたもの)、固定具(ブラスチングされたもの)及び橋脚歯(機械加工されたもの)を従来の化学的処理によって洗浄した。これらのサンプルを1Mのシュウ酸水溶液中に浸漬し、そして80℃で30分間、激しい撹拌下に置いた。30分後シュウ酸水溶液から、サンプルを取出して水中でゆすぎ、続いて超音波浴中の水中で2分間、ゆすいだ。ゆすぎの約10分後、サンプルを0.1Mのフッ化水素酸(HF)水溶液中に室温で浸漬し、活発な溶解が始まるまで撹拌し、引続いて追加の活性化処理を40秒間行った。次に、HF溶液からサンプルを取出して水中でゆすぎ、引き続いて超音波浴中の水中で5分間、ゆすいだ。これらのサンプルを、滅菌前に室温で空気中において約60分間、乾燥した。
ESEM XL30(FEI)を用いて、工程bに続いてゆすいだ後のサンプル及び工程cに続いて乾燥した後のサンプルについて走査型電子顕微鏡(SEM)観察を実施した。500×及び15000×の間の倍率を用いて、立体画像を撮影し、そしてMeX 5.0 programme(Alicona)によって評価した。フィルターは使用しなかった。微細構造のピット及び一次ナノ構造の窪みの深さ及び直径、並びに及び微細構造の隣接ピットの間の距離を求めた。結果を一次微細構造に対して図13a−c(機械加工されたサンプル)、及び図14a−c(ブラスチングされたサンプル)、そして一次ナノ構造に対して図15a−b(機械加工されたサンプル)、及び図16a−b(ブラスチングされたサンプル)に提示する。滅菌後に撮られたSEM画像を図8、9及び11に提示する。
(コインの形状をした)ブラスチング処理されたチタンサンプルを、0.1Mのフッ化水素酸及び1Mのシュウ酸を含む水溶液中に室温で浸漬し、そして各々、5、15、30及び42分間、撹拌した。溶液から、サンプルを取出して水中でゆすぎ、続いて超音波浴中の水中で2分間、ゆすいだ。サンプルを乾燥後、走査型電子顕微鏡(ESEM XL30、FEI)によって表面トポグラフィーを調べた。
チタンサンプルを0.1MのHF水溶液中に室温で浸漬し、そして活発な溶解が始まるまで撹拌し、続いて40秒の追加の処理時間をかけた。次いでHF溶液から、サンプルを取出して水中でゆすぎ、続いて超音波浴中の水中で5分間、ゆすいだ。ゆすぎの約10分後、サンプルを1Mのシュウ酸水溶液中に浸漬し、80℃で30分間、激しく撹拌した。30分後、シュウ酸溶液からサンプルを取出して水中でゆすぎ、引き続いて超音波浴中の水中で2分間、ゆすいだ。これらのサンプルを、室温で1時間、乾燥させた。
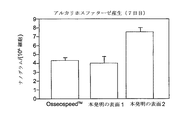
細胞増殖、及びアルカリホスファターゼ(ALP)及びプロスタグラジンE2(PGE2)各々の産生を、本発明に従ってチタン表面上でインビトロで成長させたヒト骨芽細胞に対して調べ、市販のインプラント表面(OsseoSpeed(商標);Asta Tech AB、スウェーデン)上で成長させた細胞と比較した。
MG−63は、骨芽細胞のインビトロの研究のために従来から使用されているヒト細胞株である。この研究においては、MG-63cells(MG-63、ATCC No CRL-1427、米国)を、300mLのFalcon細胞培養フラスコ(BD、WWR、スウェーデン)内の、5%のウシ胎児血清(FCS;Gibco、英国)及び1%のペニシリン−ストレプトマイシン(PEST;Gibco、英国)を含むダルベッコ最少必須培地(Dulbecco's Minimun Essential Medium(D-MEM))(Gibco、英国)中で、冷凍細胞の膨大部からの2回目の継代から、成長させた。 接着した細胞が密集度まで成長したら、0.05%トリプシン−EDTA(Gibco、英国)を用いて、それらを3継代の継代をさせた。光学顕微鏡を用いてカウントした細胞生存率は高かった(>98%)。
三つのコイン形状をした、β−滅菌されたチタン試験体;その中の1つは本発明の工程bにかけられたもの;その中の1つは本発明の工程b及び工程cにかけられたもの;そしてその中の1つは市販の表面(OsseoSpeed(商標);Asta Tech AB、スウェーデン)を有したもの;を各々別のFalcon24ウェルプレート(BD、WWR、スウェーデン)中に置いた。各々のウエルに、5%のFCS(Gibco、英国)及び1%のPEST(Gibco、英国)を含有し、20,000細胞/mLの細胞濃度を有する1mlのD−MEM(Gibco、英国)を添加した。これらのプレートを37℃、5%のCO2及び湿度100%で36時間、インキュベートした。従来のSEMサンプル調製手順に従って、サンプルを4℃でグルタルアルデヒドを用いて、続いて四酸化オスミウムによって固定し、脱水及び金スパッタリングした。細胞形態をSEM(ESEM XL30、FEI)によって調べた。細胞のSEM画像を図20a(従来の表面上で成長させた細胞)、図20b(本発明の工程bに従って処理された部材上で成長させた細胞)、図20c(本発明の工程b及び工程cに従って処理された部材上で成長させた細胞)に示す。
三セット(n=6)のコイン形状をした、β−滅菌されたチタン試験体;一組は本発明の工程bにかけられたもの(「本発明の表面1」);一組は本発明の工程b及び工程cにかけられたもの(「本発明の表面2」);そして一組は市販の表面(OsseoSpeed(商標);Asta Tech AB、スウェーデン)を有するもの;を各々別のFalcon24ウェルプレート(BD、WWR、スウェーデン)中に置いた。各々のウエルに5%のFCS(Gibco、英国)及び1%のPEST(Gibco、英国)を含有し、そして20,000細胞/mLの細胞濃度を有する1mlのD−MEM(Gibco、英国)を添加した。これらのプレートを37℃、5%のCO2及び湿度100%で14日間インキュベートした。
本発明に記載のインプラントの完全性をウサギモデルにおいて試験した。その目的は、市販の参照インプラントに対する応答と比較して、本発明に記載の二つのインプラント表面改質に対するインビボの骨組織応答を定性的に及び定量的に調べることであった。
シュウ酸中に、そして引き続きHF中に浸漬することによって、実施例1において述べられた通りに(つまり工程b及びcを含んで)、調製したチタントルク固定具(四角形の頭をした除去トルク設計、3.5×8.2ミリメーター)を使用した(試験インプラント2と称する)。また、シュウ酸中に浸漬することによって、実施例1において述べられた通りに(つまり工程cが省略されて)、調製したトルク固定具(3.5×8.2ミリメーター)(試験インプラント1と称する)も使用した。更に、市販のOsseoSpeed(商標)口腔インプラントを代表するトルク固定具(3.5×8.2ミリメーター)を参照固定具として使用した。
上記実施例1において述べられた通りに調製した口腔インプラント(3.5×8ミリメーター)のヒト設計の固定具を使用した(試験インプラント2)。また、HF処理(つまり工程c)を省略した以外は、実施例1において述べられた通りに調製した固定具(3.5×8ミリメーター)も使用した(試験インプラント1)。更に、市販のOsseoSpeed(商標)口腔インプラントを代表する固定具(3.5×8ミリメーター)を参照固定具として使用した。
12把の成体の雄のニュージーランド白ウサギを手術用として予定した。一把のウサギは初期の麻酔で死んだ(#8)。手術は平穏に進んだ。連続食塩冷却を用いて、低速穴開け(穴開けのために1500rpg、そしてインプラント挿入のために20rpm)を行った。
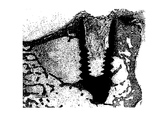
6週間後に、試験を終了し、そしてウサギを犠牲にした。インプラント及び周囲の組織を調べた。脛骨インプラントは容易に位置が分かり、それらの全てで、骨膜骨組織成長の兆候が見られた。除去トルク試験(RTQ)を用いて、インプラント−骨界面の生体力学的試験を実施した。RTQ装置(Detektor AB、Goteborg、スウェーデン)は、骨層におけるインプラントの安定性(Ncmを単位とするピークゆるみトルク)を試験するために使用される歪ゲージトランスジューサーを含む電子機器であり、従って、骨組織及びインプラントの間の界面せん断力を大まかに反映する三次元試験と見なすことができる(Johansson C. B.,Albrektsson T.,Clin Oral implants Res 1991;2:24-9)。直線的に増大するトルクがインプラントの同じ軸上に、一体化が破壊されるまでかけられ、そしてピーク値が記録された。大腿骨中に挿入されたインプラントは、よりしばしば骨組織によるインプラント頭部の「完全な被覆」を示した。大腿骨インプラントを固定液中に浸漬し、そして組織学及び組織学形態計測研究のために更に処理した。
6週間後に、試験を終了し、そしてウサギを犠牲にした。ウサギ#1及び#5からの骨組織及びインプラントを含む大腿骨インプラント部位の選択サンプルを、骨−インプラント接触(BIC)及び内側のネジ山(thread)内の骨領域(内部領域、ia)及び大腿骨から回収されたインプラント周辺の種々の領域における対応する鏡像(mi)に関して組織学形態計測的に評価した。
(a)ミクロ−ネジ山;
(b)マクロ−ネジ山;
(c)骨髄空洞における先端側(ネジ山がない)に沿った領域;及び
(d)インプラントの先端底部中(この領域は、骨インプラント接触についてのみ報告される)
図23aはウサギ#1、試験インプラント2を示す;
図23bはウサギ#1、参照インプラントを示す;
図24aはウサギ#5、試験インプラント1を示す;及び
図24bはウサギ#5、参照インプラントを示す;
骨形成を調べるための1つの従来からのインビトロのモデルは、疑似体液(SBFs)中への生体材料の浸漬である。SBFsはヒトの血漿のそれにほぼ等しいイオン濃度を有する溶液である(Kokubo T.,Kushitani H.,Sakka S.,Kitsugi T.,Yamamuro T.,J Biomed Mater Res 1990;24:721-734;Oyane A.,Kim H. K.,Furuya T.,Kokubo T.,Miyazaki T., Nakamura T.,J Biomed Mater Res 2003;65A、188-195)。生体材料の核形成能力に依存して、骨状のリン酸カルシウムがその表面上に沈殿することになる。インビボの骨生理活性を有するSBF中でのアパタイト形成の定量的相関が報告されている(Kokubo T.,Takadama H.,Biomaterials 2006;27:2907-2915)。今日では、SBFのインビトロモデルはしばしば使用され、そして国際標準、ISO 23317:2007Eによって記載されている。
ヒトの血漿の電解質濃度に類似した電解質濃度を有する修正SBF(Oyane A.ら、J Biomed Mater Res 2003;65A、188-195)が選択された(Vander A.J., Sherman J.H., Luciano D.S.,「Human physiology The mechanisms of body function)」、5th ed. McGraw-Hill Publishing Company、ニューヨーク、1990:349-400)。SBFは10.806gのNaCl、1.480gのNaHCO3、4.092gのNa2CO3、0.450gのKCl、0.460gのK2HPO4・3H2O、0.622gのMgCl2・6H2O、23.856gの2−(4−(2−ヒドロキシエチル)−1−ピペラジニル)エタンスルホン酸(HEPES)、0.776gのCaCl2及び0.144gのNa2SO4を、2000ミリリッターの脱イオン水中に溶解させることによって調製した。HEPESは溶液に添加する前に、200ミリリッターの脱イオン水中に溶解させた。最終的なpHは1.0MのNaOHを用いて37℃で7.40に調節された。全ての試薬は、NaCl及びNa2SO4 (これらはFluka(スウェーデン)から得られた)を除いて、Merck(スウェーデン)から得られた。
可能なアパタイトの形成の解析を、環境走査型電子顕微鏡(ESEM、XL 30、FEI)を用いて実施した。SBF浸漬前の表面構造のSEM画像を、図25a(参照)、26a(本発明の表面1)、及び27a(本発明の表面2)に提示する。SBF浸漬後の表面構造を調べたとき、サンプルの全ての組上でリン酸カルシウムの薄い層が形成されたと結論付けられた;参照(図25b)、本発明の表面1(図26b)、及び本発明の表面2(図27b)。
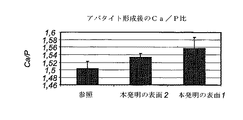
アパタイト形成前後のサンプルの化学的解析のためにエネルギー分散分光法(EDS,Apollo 40,EDAX)を使用した。チタンシグナルを解析することによって、リン酸カルシウムによるサンプルの被覆度が間接的に評価できた。本発明表面2は、SBF浸漬後のチタンシグナルにおいて最大の低下を示し(図28)、調査した資料の組の中で最も広範囲なアパタイト形成を示した。
Claims (51)
- 生体適合性部材の改質方法であって、以下の工程:
a)少なくとも部分的に金属酸化物で被覆された生体適合性部材を備えること;及び
b)該部材の部分が該金属酸化物で被覆された、その部材の少なくとも一部を、シュウ酸を含む水性組成物で処理すること;そして、さらに、
c)i)イオン化されたフッ素及びイオン化された塩素を含むグループから選ばれる少なくとも1つの材料;及び
ii)少なくとも1つの酸;
を含む第二の水性組成物で、改質された金属酸化物の少なくとも部分を処理すること、ここで、改質された金属酸化物上に不動態化酸化物が形成される前に、工程cが実行されること;
を含み、そのことによって、改質された金属酸化物の部分が溶解し、そして引き続いて沈殿して金属酸化物を含む二次ナノ構造を形成する、方法。 - 工程bの組成物が、0.001から5Mの範囲内のシュウ酸濃度を有し;工程bの処理が、10から60分の範囲内の処理時間で実行される、請求項1記載の方法。
- 処理時間が20から30分の範囲内である、請求項2記載の方法。
- 工程bの組成物が、20℃から100℃の範囲内の温度を有する、請求項1〜3の何れか1項に記載の方法。
- 工程bの完了後、0℃又はそれより高い温度で、そして酸素含有大気における通常の大気圧で、部材が保持される時間としてカウントされた、180時間又はそれより短い時間以内で、工程cが実行される、請求項1〜4の何れか1項に記載の方法。
- 工程bの完了後、72時間又はそれより短い時間以内で、工程cが実行される、請求項1〜5の何れか1項に記載の方法。
- 工程bの完了後、24時間又はそれより短い時間以内で、工程cが実行される、請求項1〜5の何れか1項に記載の方法。
- 工程bの完了後、1時間又はそれより短い時間以内で、工程cが実行される、請求項1〜5の何れか1項に記載の方法。
- 工程bの完了後、10分又はそれより短い時間以内で、工程cが実行される、請求項1〜5の何れか1項に記載の方法。
- 第二の水性組成物がフッ化水素酸を含む、請求項1〜9の何れか1項に記載の方法。
- 第二の水性組成物が、0.5から5の範囲内のpH;0.05から0.3Mの範囲内の、イオン化されたフッ素及びイオン化された塩素を含むグループから選ばれる少なくとも1つの材料の濃度を有し;そして10秒から3分の範囲内の活性化処理時間で工程cの処理が実行される、請求項1〜10の何れか1項に記載の方法。
- 第二の水性組成物が15℃から25℃の範囲内の温度を有する、請求項1〜11の何れか1項に記載の方法。
- 工程bの水性組成物が骨成長増強物質を含む、請求項1〜12の何れか1項に記載の方法。
- 第二の水性組成物が骨成長増強物質を含む、請求項1〜13の何れか1項に記載の方法。
- 骨成長増強物質が金属イオン又はそれらの塩を含む、請求項13又は14に記載の方法。
- 金属イオンが、チタンイオン、マグネシウムイオン、カルシウムイオン、リチウムイオン、ストロンチウムイオン、又はそれらの如何なる組合せから成るグループから選ばれるイオンを含む、請求項15に記載の方法。
- 金属イオンがリチウムイオンを含む、請求項15に記載の方法。
- 金属イオンがストロンチウムイオンを含む、請求項15に記載の方法。
- 部材が少なくとも部分的にチタン又はチタン合金から成る、請求項1〜18の何れか1項に記載の方法。
- 金属酸化物が酸化チタンを含む、請求項1〜19の何れか1項に記載の方法。
- 金属酸化物が本質的に酸化チタン又は酸化チタンの組合せから成る、請求項1〜20の何れか1項に記載の方法。
- 金属酸化物が不動態化酸化チタンを含む、請求項1〜21の何れか1項に記載の方法。
- 工程bの前の部材が機械的表面処理にかけられる、請求項1〜22の何れか1項に記載の方法。
- 機械的表面処理がブラスチングを含む、請求項23に記載の方法。
- 工程bの前の部材が化学的表面処理にかけられる、請求項1〜24の何れか1項に記載の方法。
- 化学的表面処理が脱脂又は洗浄処理を含む、請求項25記載の方法。
- 生体適合性部材が、インプラント、固定具、橋脚歯、ワンピースインプラント又はそれらの組合せから成るグループから選ばれる歯科用部材である、請求項1〜26の何れか1項に記載の方法。
- 生体適合性部材が整形外科用部材である、請求項1〜26の何れか1項に記載の方法。
- 請求項1〜28の何れか1項に記載の方法によって得られ得る部材。
- 少なくとも部分的に金属酸化物で被覆された生体適合性部材であって:
a)プラトー及び/又はリッジによって分離されたピットを含む微細構造;
b)該微細構造の上に重なっている一次ナノ構造、ここで該一次ナノ構造は波状の形に配置された窪みを含む;及び
c)前記一次ナノ構造の上に重なっている二次ナノ構造
を含む金属酸化物の表面を有する基体を含む生体適合性部材。 - 微細構造が、0.5から15μmの範囲内のピット直径;0.1から2.5μmの範囲内の深さ;及び0から10μmの範囲内の互いに隣接するピット間の距離を有する、請求項30に記載の生体適合性部材。
- 一次ナノ構造の窪みが10nmから1μmの範囲内の直径;及び10nmから300nmの範囲内の深さを有する、請求項30又は31に記載の生体適合性部材。
- 一次ナノ構造の個々の窪みの直径が、該個々の窪みの深さを超える、請求項30〜32の何れか1項に記載の生体適合性部材。
- 一次ナノ構造の窪みの直径が、該窪みが重なる微細構造のピットの直径より小さく、そして該一次ナノ構造の窪みの深さが、該窪みが重なる上記微細構造のピットの深さより小さい、請求項30〜33の何れか1項に記載の生体適合性部材。
- 一次ナノ構造の窪みの境界の少なくとも部分が、該一次ナノ構造の他の窪みの境界の少なくとも部分を構成する、請求項30〜34の何れか1項に記載の生体適合性部材。
- 部材が機械的表面処理にかけられている、請求項30〜35の何れか1項に記載の生体適合性部材。
- 機械的表面処理がブラスチングを含む、請求項36記載の生体適合性部材。
- 基体が少なくとも部分的にチタン又はチタン合金から成る、請求項30〜37の何れか1項に記載の生体適合性部材。
- 基体がチタンから成る、請求項30〜37の何れか1項に記載の生体適合性部材。
- 二次ナノ構造が一様に分布したパターンで一次ナノ構造の上に重なっていて、丸みのあるピークの形状を有する分離した突起を含む、請求項30〜39の何れか1項に記載の生体適合性部材。
- 二次ナノ構造が、20から550nmの範囲内のピーク直径;5から200nmの範囲内の平均ピーク高さ;そして10から450nmの範囲内のピーク間距離を有する、請求項40に記載の生体適合性部材。
- 二次ナノ構造が、15から150ピーク/平方μmの範囲内のピーク密度を含む、請求項40又は41に記載の生体適合性部材。
- ナノ要素が金属酸化物を含む、請求項30〜42の何れか1項に記載の生体適合性部材。
- ナノ要素が酸化チタンを含む、請求項43に記載の生体適合性部材。
- 表面が骨成長増強物質を含む、請求項30〜44の何れか1項に記載の生体適合性部材。
- 二次ナノ構造の少なくとも一部が骨成長増強物質を含む、請求項30〜45の何れか1項に記載の生体適合性部材。
- 骨成長増強物質が、チタンイオン、マグネシウムイオン、カルシウムイオン、リチウムイオン、ストロンチウムイオン、又はそれらの任意の組合せから成るグループから選ばれる金属イオン又はその塩を含む、請求項45又は46に記載の生体適合性部材。
- 骨成長増強物質がリチウムイオンを含む、請求項45又は46に記載の生体適合性部材。
- 骨成長増強物質がストロンチウムイオンを含む、請求項45、46及び48の何れか1項に記載の生体適合性部材。
- 部材が、インプラント、固定具、橋脚歯、ワンピースインプラント又はそれらの組合せから成るグループから選ばれる歯科用部材である、請求項30〜49の何れか1項に記載の生体適合性部材。
- 部材が整形外科用部材である、請求項30〜49の何れか1項に記載の生体適合性部材。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP07112076.0 | 2007-07-09 | ||
| EP07112076A EP2022447A1 (en) | 2007-07-09 | 2007-07-09 | Nanosurface |
| PCT/EP2008/058860 WO2009007373A1 (en) | 2007-07-09 | 2008-07-08 | Nanosurface |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2011509098A JP2011509098A (ja) | 2011-03-24 |
| JP2011509098A5 JP2011509098A5 (ja) | 2011-08-11 |
| JP5658561B2 true JP5658561B2 (ja) | 2015-01-28 |
Family
ID=38740247
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2010515496A Active JP5658561B2 (ja) | 2007-07-09 | 2008-07-08 | ナノ表面 |
Country Status (12)
| Country | Link |
|---|---|
| US (2) | US8632836B2 (ja) |
| EP (4) | EP2022447A1 (ja) |
| JP (1) | JP5658561B2 (ja) |
| KR (1) | KR20100055403A (ja) |
| CN (2) | CN102961193B (ja) |
| AT (1) | ATE494861T1 (ja) |
| AU (1) | AU2008274303A1 (ja) |
| BR (1) | BRPI0814563B8 (ja) |
| CA (1) | CA2693478A1 (ja) |
| DE (1) | DE602008004546D1 (ja) |
| ES (3) | ES2358776T3 (ja) |
| WO (1) | WO2009007373A1 (ja) |
Families Citing this family (55)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8585765B2 (en) | 2005-05-06 | 2013-11-19 | Titan Spine, Llc | Endplate-preserving spinal implant having a raised expulsion-resistant edge |
| US8562685B2 (en) | 2005-05-06 | 2013-10-22 | Titan Spine, Llc | Spinal implant and integration plate for optimizing vertebral endplate contact load-bearing edges |
| US8758443B2 (en) | 2005-05-06 | 2014-06-24 | Titan Spine, Llc | Implants with integration surfaces having regular repeating surface patterns |
| US8551176B2 (en) | 2005-05-06 | 2013-10-08 | Titan Spine, Llc | Spinal implant having a passage for enhancing contact between bone graft material and cortical endplate bone |
| US8403991B2 (en) | 2005-05-06 | 2013-03-26 | Titan Spine Llc | Implant with critical ratio of load bearing surface area to central opening area |
| US8617248B2 (en) | 2005-05-06 | 2013-12-31 | Titan Spine, Llc | Spinal implant having variable ratios of the integration surface area to the axial passage area |
| US9125756B2 (en) | 2005-05-06 | 2015-09-08 | Titan Spine, Llc | Processes for producing regular repeating patterns on surfaces of interbody devices |
| US8758442B2 (en) | 2005-05-06 | 2014-06-24 | Titan Spine, Llc | Composite implants having integration surfaces composed of a regular repeating pattern |
| US8480749B2 (en) | 2005-05-06 | 2013-07-09 | Titan Spine, Llc | Friction fit and vertebral endplate-preserving spinal implant |
| US8562684B2 (en) | 2005-05-06 | 2013-10-22 | Titan Spine, Llc | Endplate-preserving spinal implant with an integration plate having a roughened surface topography |
| US8435302B2 (en) | 2005-05-06 | 2013-05-07 | Titan Spine, Llc | Instruments and interbody spinal implants enhancing disc space distraction |
| US8262737B2 (en) | 2005-05-06 | 2012-09-11 | Titan Spine, Llc | Composite interbody spinal implant having openings of predetermined size and shape |
| US8992622B2 (en) | 2005-05-06 | 2015-03-31 | Titan Spine, Llc | Interbody spinal implant having a roughened surface topography |
| US20120312779A1 (en) | 2005-05-06 | 2012-12-13 | Titian Spine, LLC | Methods for manufacturing implants having integration surfaces |
| US8585767B2 (en) | 2005-05-06 | 2013-11-19 | Titan Spine, Llc | Endplate-preserving spinal implant with an integration plate having durable connectors |
| US9168147B2 (en) | 2005-05-06 | 2015-10-27 | Titan Spine, Llc | Self-deploying locking screw retention device |
| US8585766B2 (en) | 2005-05-06 | 2013-11-19 | Titan Spine, Llc | Endplate-preserving spinal implant with an integration plate having durable connectors |
| US8591590B2 (en) | 2005-05-06 | 2013-11-26 | Titan Spine, Llc | Spinal implant having a transverse aperture |
| US8545568B2 (en) | 2005-05-06 | 2013-10-01 | Titan Spine, Llc | Method of using instruments and interbody spinal implants to enhance distraction |
| US8814939B2 (en) | 2005-05-06 | 2014-08-26 | Titan Spine, Llc | Implants having three distinct surfaces |
| US11096796B2 (en) | 2005-05-06 | 2021-08-24 | Titan Spine, Llc | Interbody spinal implant having a roughened surface topography on one or more internal surfaces |
| EP2022447A1 (en) * | 2007-07-09 | 2009-02-11 | Astra Tech AB | Nanosurface |
| US8696759B2 (en) * | 2009-04-15 | 2014-04-15 | DePuy Synthes Products, LLC | Methods and devices for implants with calcium phosphate |
| US10493793B2 (en) * | 2010-04-09 | 2019-12-03 | Brown University | Nanostructured surfaces |
| EP2394602B1 (en) * | 2010-06-10 | 2016-05-18 | Dentsply IH AB | A dental fixture |
| JP2012143416A (ja) * | 2011-01-13 | 2012-08-02 | Gc Corp | 歯科用インプラント及び歯科用インプラントの表面処理方法 |
| KR20130032507A (ko) * | 2011-09-23 | 2013-04-02 | 포항공과대학교 산학협력단 | 유기 금속을 포함하는 자기조립성 고분자를 포함하는 전극 및 그 제조 방법 |
| US8992619B2 (en) | 2011-11-01 | 2015-03-31 | Titan Spine, Llc | Microstructured implant surfaces |
| TWI469762B (zh) * | 2011-11-25 | 2015-01-21 | Ind Tech Res Inst | 具有微奈米結構之電燒刀及其製造方法 |
| WO2013109078A1 (ko) * | 2012-01-20 | 2013-07-25 | 오스템임플란트 주식회사 | 거시-미세-나노규모의 삼중 구조를 지니는 임플란트의 골 유착 능력을 향상시키기 위한 다공성 표면과 그 제조 방법 |
| EP2827806B1 (en) | 2012-03-20 | 2020-06-24 | Titan Spine, Inc. | Process of fabricating bioactive spinal implant endplates |
| EP2716261A1 (en) | 2012-10-02 | 2014-04-09 | Titan Spine, LLC | Implants with self-deploying anchors |
| US9498349B2 (en) | 2012-10-09 | 2016-11-22 | Titan Spine, Llc | Expandable spinal implant with expansion wedge and anchor |
| WO2014195027A2 (en) * | 2013-06-07 | 2014-12-11 | Straumann Holding Ag | Abutment |
| US9615935B2 (en) | 2014-01-30 | 2017-04-11 | Titan Spine, Llc | Thermally activated shape memory spring assemblies for implant expansion |
| AU2015220178A1 (en) * | 2014-02-21 | 2016-09-01 | Maruemu Works Co., Ltd. | Dental member |
| US10687956B2 (en) | 2014-06-17 | 2020-06-23 | Titan Spine, Inc. | Corpectomy implants with roughened bioactive lateral surfaces |
| US20150366668A1 (en) * | 2014-06-23 | 2015-12-24 | Community Blood Center | Cellular-scale surface modification for increased osteogenic protein expression |
| CN105441960A (zh) * | 2014-08-22 | 2016-03-30 | 广州中国科学院先进技术研究所 | 钛牙种植体表面超亲水性微/纳分级结构的构建方法 |
| EP3034033A1 (en) | 2014-12-16 | 2016-06-22 | Nobel Biocare Services AG | Dental implant |
| TWI726940B (zh) | 2015-11-20 | 2021-05-11 | 美商泰坦脊柱股份有限公司 | 積層製造整形外科植入物之方法 |
| JP6914266B2 (ja) | 2015-11-20 | 2021-08-04 | タイタン スパイン インコーポレイテッドTitan Spine,Inc. | 整形外科インプラントを付加製作するための方法 |
| CN105537589B (zh) * | 2016-01-28 | 2018-12-25 | 佛山市安齿生物科技有限公司 | 一种slm成型钛种植体表面处理方法 |
| WO2018026448A1 (en) * | 2016-08-03 | 2018-02-08 | Titan Spine, Llc | Titanium implant surfaces free from alpha case and with enhanced osteoinduction |
| US11051915B2 (en) * | 2016-11-10 | 2021-07-06 | Nantoh. Co., Ltd. | Biological tissue rootage face, implant, method for forming biological tissue rootage face, and method for producing implant |
| CN108096640A (zh) * | 2016-11-24 | 2018-06-01 | 重庆润泽医药有限公司 | 一种多孔材料 |
| JP6893838B2 (ja) * | 2017-07-12 | 2021-06-23 | 日本ピストンリング株式会社 | インプラントの表面構造およびインプラントの表面構造の製造方法 |
| US11419705B2 (en) | 2017-09-27 | 2022-08-23 | Straumann Holding Ag | Process for the preparation of nanostructures on a dental implant |
| GB201802109D0 (en) * | 2018-02-09 | 2018-03-28 | Depuy Ireland Ultd Co | Medical implants comprising anti-infective surfaces |
| US20200306015A1 (en) * | 2018-06-07 | 2020-10-01 | Taiwan Shan Yin International Co., Ltd. | Implant device |
| US11485052B2 (en) | 2018-07-30 | 2022-11-01 | Canon Kabushiki Kaisha | Resin product, method of making resin product, interchangeable lens, and optical device |
| JP7423202B2 (ja) * | 2018-07-30 | 2024-01-29 | キヤノン株式会社 | 樹脂製品、樹脂製品の製造方法、交換レンズ、および光学機器 |
| WO2021153658A1 (ja) * | 2020-01-31 | 2021-08-05 | 京セラ株式会社 | 生体インプラントおよび生体インプラントの製造方法 |
| KR102399423B1 (ko) * | 2021-03-16 | 2022-05-19 | 주식회사 도이프 | 교정용 미니스크류의 제조 및 그 표면처리 방법 |
| EP4169485B1 (fr) * | 2021-10-21 | 2024-07-03 | Anthogyr | Implant à ancrage osseux et procédé de fabrication d'un tel implant |
Family Cites Families (75)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4015A (en) * | 1845-04-26 | Hand-loom for weaving figured fabrics | ||
| CA962806A (en) * | 1970-06-04 | 1975-02-18 | Ontario Research Foundation | Surgical prosthetic device |
| US3707006A (en) * | 1970-08-26 | 1972-12-26 | Gulf Oil Corp | Orthopedic device for repair or replacement of bone |
| US3906550A (en) * | 1973-12-27 | 1975-09-23 | William Rostoker | Prosthetic device having a porous fiber metal structure |
| US4101984A (en) * | 1975-05-09 | 1978-07-25 | Macgregor David C | Cardiovascular prosthetic devices and implants with porous systems |
| US4374669A (en) * | 1975-05-09 | 1983-02-22 | Mac Gregor David C | Cardiovascular prosthetic devices and implants with porous systems |
| SE416175B (sv) * | 1979-03-07 | 1980-12-08 | Per Ingvar Branemark | For implantering i kroppsvevnad serskilt benvevnad, avsett material |
| GB2189815B (en) * | 1986-03-24 | 1990-10-10 | Permelec Electrode Ltd | Titanium composite materials coated with calcium phosphate compound and process for production thereof |
| DE58905636D1 (de) * | 1989-03-23 | 1993-10-21 | Straumann Inst Ag | Metallisches Implantat. |
| EP0401793B1 (en) | 1989-06-06 | 1995-10-04 | TDK Corporation | Use of ceramic materials for living hard tissue replacements |
| JPH0428374A (ja) | 1990-05-23 | 1992-01-30 | Tdk Corp | 複合インプラント |
| JP2930619B2 (ja) | 1989-10-30 | 1999-08-03 | 春幸 川原 | チタンもしくはチタン基合金製生体修復部材及びその表面処理法 |
| DE4028374A1 (de) | 1990-09-07 | 1992-03-12 | Abb Patent Gmbh | Verfahren zum verbinden von bauteilen |
| TW197475B (ja) | 1990-12-26 | 1993-01-01 | Eltech Systems Corp | |
| US5282861A (en) * | 1992-03-11 | 1994-02-01 | Ultramet | Open cell tantalum structures for cancellous bone implants and cell and tissue receptors |
| US5356516A (en) * | 1993-02-26 | 1994-10-18 | Sandia Corporation | Process for etching mixed metal oxides |
| US5824651A (en) * | 1993-05-10 | 1998-10-20 | Universite De Montreal | Process for modification of implant surface with bioactive conjugates for improved integration |
| US5368881A (en) * | 1993-06-10 | 1994-11-29 | Depuy, Inc. | Prosthesis with highly convoluted surface |
| CA2175660C (en) | 1993-11-02 | 2005-07-05 | Charles S. Naiman | Control of cell growth |
| US7048870B1 (en) * | 1993-12-20 | 2006-05-23 | Astrazeneca Ab | Metallic implant and process for treating a metallic implant |
| US6652765B1 (en) * | 1994-11-30 | 2003-11-25 | Implant Innovations, Inc. | Implant surface preparation |
| US5863201A (en) * | 1994-11-30 | 1999-01-26 | Implant Innovations, Inc. | Infection-blocking dental implant |
| US6491723B1 (en) * | 1996-02-27 | 2002-12-10 | Implant Innovations, Inc. | Implant surface preparation method |
| DE69536061D1 (de) * | 1994-11-30 | 2010-05-12 | Biomet 3I Llc | Vorbereitung einer Implantatoberfläche |
| DE19506188C2 (de) | 1995-02-22 | 2003-03-06 | Miladin Lazarov | Implantat und dessen Verwendung |
| EP0798992B1 (en) * | 1995-12-08 | 2008-11-26 | Zimmer Dental Inc. | Dental implant having multiple textured surfaces |
| EP0806212B1 (en) | 1996-05-10 | 2003-04-02 | IsoTis N.V. | Device for incorporation and release of biologically active agents |
| DE69716505T2 (de) * | 1996-05-10 | 2003-06-26 | Isotis N.V., Bilthoven | Implantatwerkstoff und Verfahren zu seiner Herstellung |
| EP0806211B1 (en) | 1996-05-10 | 2002-10-23 | IsoTis N.V. | Implant material and process for producing it |
| KR100492920B1 (ko) | 1996-11-19 | 2005-06-02 | 캄주크, 알렉산더 콘스탄티노비치 | 차량의 트랜스퍼 기어박스에 의해 구동되는 호이스트 |
| DE69734921T2 (de) | 1996-12-09 | 2006-09-28 | Nippon Sheet Glass Co., Ltd. | Antibeschlag-gegenstand und dessen herstellungsverfahren |
| US20050031663A1 (en) * | 1997-05-16 | 2005-02-10 | Cecilia Larsson | Implant element |
| JP3404286B2 (ja) * | 1998-04-16 | 2003-05-06 | 日本パーカライジング株式会社 | 金属の表面処理方法、および該表面処理方法により得られた表面を有する金属部材 |
| US6261322B1 (en) * | 1998-05-14 | 2001-07-17 | Hayes Medical, Inc. | Implant with composite coating |
| SE514202C2 (sv) * | 1999-05-31 | 2001-01-22 | Nobel Biocare Ab | På implantat till ben- eller vävnadsstruktur anordnat skikt samt sådant implantat och förfarande för applicering av skiktet |
| TW491714B (en) * | 1999-12-08 | 2002-06-21 | Wen-Jing Shiue | Orthopedic implant having a porous surface and method of making same |
| US7048541B2 (en) * | 2000-04-04 | 2006-05-23 | Nobel Biocare Ab | Implant having attachment and hole-insert parts, and method for producing such an implant |
| IT1317969B1 (it) * | 2000-06-09 | 2003-07-21 | Nora Elettrodi De | Elettrodo caratterizzato da elevata adesione di uno strato cataliticosuperficiale. |
| CH694935A5 (de) * | 2000-07-26 | 2005-09-30 | Straumann Holding Ag | Oberflaechenmodifizierte Implantate. |
| CA2402578C (en) | 2000-09-19 | 2011-06-07 | Eduardo Anitua Aldecoa | Method for surface treatment of implants or prosthesis made of titanium or other materials |
| CN1237945C (zh) * | 2000-10-09 | 2006-01-25 | 刘宝林 | 改良的牙种植体表面的喷砂处理方法 |
| US6921544B2 (en) * | 2001-03-06 | 2005-07-26 | Rutgers, The State University | Magnesium-substituted hydroxyapatites |
| CA2442582C (en) * | 2001-04-02 | 2011-01-04 | Stratec Medical Ag | Bioactive surface layer, particularly for medical implants and prostheses |
| US6527938B2 (en) * | 2001-06-21 | 2003-03-04 | Syntheon, Llc | Method for microporous surface modification of implantable metallic medical articles |
| CN100515504C (zh) * | 2001-10-12 | 2009-07-22 | 美国英佛曼公司 | 涂层,涂布体及其制造方法 |
| EP1448908B1 (de) | 2001-11-23 | 2006-02-01 | Universität Duisburg-Essen | Implantat |
| SE523288C2 (sv) * | 2002-07-19 | 2004-04-06 | Astra Tech Ab | Ett implantat och ett förfarande för behandling av en implantatyta |
| US7537664B2 (en) * | 2002-11-08 | 2009-05-26 | Howmedica Osteonics Corp. | Laser-produced porous surface |
| EP1440669B1 (de) | 2003-01-23 | 2006-11-22 | Dinkelacker, Wolfgang, Dr. med. dent. | Knochenimplantat und Verfahren zu seiner Herstellung |
| US20040167632A1 (en) * | 2003-02-24 | 2004-08-26 | Depuy Products, Inc. | Metallic implants having roughened surfaces and methods for producing the same |
| JP5079208B2 (ja) * | 2003-02-24 | 2012-11-21 | デピュイ・プロダクツ・インコーポレイテッド | 粗面を有する金属製インプラント及びその製造方法 |
| US20060216358A1 (en) * | 2003-05-07 | 2006-09-28 | Christian Hansen | Controlled release composition containing a strontium salt |
| US8251700B2 (en) * | 2003-05-16 | 2012-08-28 | Biomet 3I, Llc | Surface treatment process for implants made of titanium alloy |
| US20050119758A1 (en) * | 2003-07-30 | 2005-06-02 | Bio-Lok International Inc. | Surgical implant for promotion of osseo-integration |
| BRPI0411404A (pt) * | 2004-03-04 | 2006-07-25 | Young-Taek Sul | implante de titanato de magnésio osseoindutivo e processo de fabricação do mesmo |
| KR100714244B1 (ko) * | 2004-04-26 | 2007-05-02 | 한국기계연구원 | 생체용 골유도성 금속 임플란트 및 그 제조방법 |
| US8329202B2 (en) * | 2004-11-12 | 2012-12-11 | Depuy Products, Inc. | System and method for attaching soft tissue to an implant |
| WO2006091582A2 (en) | 2005-02-24 | 2006-08-31 | Implant Innovations, Inc. | Surface treatment methods for implants made of titanium or titanium alloy |
| US7601149B2 (en) | 2005-03-07 | 2009-10-13 | Boston Scientific Scimed, Inc. | Apparatus for switching nominal and attenuated power between ablation probes |
| JP2008538515A (ja) | 2005-03-21 | 2008-10-30 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | マイクロ構造表面上における制御可能なナノ構造化 |
| US7368065B2 (en) * | 2005-06-23 | 2008-05-06 | Depuy Products, Inc. | Implants with textured surface and methods for producing the same |
| JP2007001826A (ja) * | 2005-06-24 | 2007-01-11 | Hitachi Ltd | 金属酸化物材料とその製造方法及びその利用 |
| EP1764116A1 (en) * | 2005-09-16 | 2007-03-21 | Debiotech S.A. | Porous coating process using colloidal particles |
| US20090207790A1 (en) | 2005-10-27 | 2009-08-20 | Qualcomm Incorporated | Method and apparatus for settingtuneawaystatus in an open state in wireless communication system |
| EP3517137A1 (en) | 2005-11-14 | 2019-07-31 | Biomet 3I, LLC | Deposition of discrete nanoparticles on an implant surface |
| US7749555B2 (en) * | 2006-01-25 | 2010-07-06 | Medtronic, Inc | Modification of chemical forces of bone constructs |
| US9327056B2 (en) * | 2006-02-14 | 2016-05-03 | Washington State University | Bone replacement materials |
| US20070264303A1 (en) * | 2006-05-12 | 2007-11-15 | Liliana Atanasoska | Coating for medical devices comprising an inorganic or ceramic oxide and a therapeutic agent |
| EP2104471B1 (de) * | 2006-12-22 | 2017-01-25 | Thommen Medical Ag | Dentalimplantat und verfahren zu dessen herstellung |
| EP2022447A1 (en) * | 2007-07-09 | 2009-02-11 | Astra Tech AB | Nanosurface |
| US20090035723A1 (en) * | 2007-08-03 | 2009-02-05 | Claus Daniel | Material with a repetitive pattern of micro-features for application in a living organism and method of fabrication |
| ES2315194B1 (es) * | 2007-09-10 | 2010-02-26 | Francisco J. GARCIA SABAN | Procedimiento para obtener una nueva superficie de un implante metalico a base de titanio destinado a ser insertado en tejido oseo. |
| WO2009061887A2 (en) * | 2007-11-06 | 2009-05-14 | University Of Connecticut | Ceramic/structural protein composites and method of preparation thereof |
| EP3159018B1 (en) * | 2008-02-29 | 2022-04-20 | Smith & Nephew, Inc | Gradient coating for biomedical applications |
| CA2722661A1 (en) * | 2008-04-28 | 2009-11-05 | Kuraray Medical Inc. | Dental composition and composite resin |
-
2007
- 2007-07-09 EP EP07112076A patent/EP2022447A1/en not_active Withdrawn
-
2008
- 2008-07-08 CN CN201210472825.4A patent/CN102961193B/zh active Active
- 2008-07-08 EP EP20120179573 patent/EP2537485B1/en active Active
- 2008-07-08 CA CA2693478A patent/CA2693478A1/en not_active Abandoned
- 2008-07-08 EP EP08774897A patent/EP2178466B1/en active Active
- 2008-07-08 DE DE602008004546T patent/DE602008004546D1/de active Active
- 2008-07-08 CN CN200880024004.6A patent/CN101686862B/zh active Active
- 2008-07-08 AT AT08774897T patent/ATE494861T1/de active
- 2008-07-08 ES ES08774897T patent/ES2358776T3/es active Active
- 2008-07-08 WO PCT/EP2008/058860 patent/WO2009007373A1/en active Application Filing
- 2008-07-08 US US12/298,884 patent/US8632836B2/en active Active
- 2008-07-08 ES ES10194575T patent/ES2397633T3/es active Active
- 2008-07-08 ES ES12179573.6T patent/ES2542243T3/es active Active
- 2008-07-08 JP JP2010515496A patent/JP5658561B2/ja active Active
- 2008-07-08 BR BRPI0814563A patent/BRPI0814563B8/pt active IP Right Grant
- 2008-07-08 EP EP10194575A patent/EP2319461B1/en active Active
- 2008-07-08 AU AU2008274303A patent/AU2008274303A1/en not_active Abandoned
- 2008-07-08 KR KR1020107002403A patent/KR20100055403A/ko not_active Application Discontinuation
-
2012
- 2012-09-14 US US13/620,079 patent/US9642708B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| AU2008274303A1 (en) | 2009-01-15 |
| US20130013081A1 (en) | 2013-01-10 |
| US8632836B2 (en) | 2014-01-21 |
| CN101686862A (zh) | 2010-03-31 |
| CN102961193A (zh) | 2013-03-13 |
| BRPI0814563A2 (pt) | 2015-01-06 |
| EP2319461B1 (en) | 2012-09-19 |
| CN101686862B (zh) | 2014-08-06 |
| EP2537485B1 (en) | 2015-05-13 |
| BRPI0814563B8 (pt) | 2021-06-22 |
| ES2358776T3 (es) | 2011-05-13 |
| DE602008004546D1 (en) | 2011-02-24 |
| EP2022447A1 (en) | 2009-02-11 |
| BRPI0814563B1 (pt) | 2018-12-26 |
| EP2537485A1 (en) | 2012-12-26 |
| ES2397633T3 (es) | 2013-03-08 |
| EP2178466B1 (en) | 2011-01-12 |
| EP2178466A1 (en) | 2010-04-28 |
| ES2542243T3 (es) | 2015-08-03 |
| CA2693478A1 (en) | 2009-01-15 |
| ATE494861T1 (de) | 2011-01-15 |
| CN102961193B (zh) | 2016-05-18 |
| JP2011509098A (ja) | 2011-03-24 |
| EP2319461A1 (en) | 2011-05-11 |
| US20100173264A1 (en) | 2010-07-08 |
| US9642708B2 (en) | 2017-05-09 |
| WO2009007373A1 (en) | 2009-01-15 |
| KR20100055403A (ko) | 2010-05-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5658561B2 (ja) | ナノ表面 | |
| US9744263B2 (en) | Bone tissue implant comprising strontium ions | |
| AU2006315629B2 (en) | Deposition of discrete nanoparticles on an implant surface | |
| Xue et al. | In vivo evaluation of plasma-sprayed titanium coating after alkali modification | |
| JP5487101B2 (ja) | リチウムイオンを含む骨組織インプラント | |
| Nazir et al. | Biomimetic coating of modified titanium surfaces with hydroxyapatite using simulated body fluid | |
| Fouziya et al. | Surface modifications of titanium implants–The new, the old, and the never heard of options |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110624 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110624 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130129 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130426 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130508 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130729 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20131126 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140221 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140715 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20141006 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20141104 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20141128 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5658561 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |