JP2010538457A - イオン性高分子電解質を含有する銅cmp組成物及び方法 - Google Patents
イオン性高分子電解質を含有する銅cmp組成物及び方法 Download PDFInfo
- Publication number
- JP2010538457A JP2010538457A JP2010522907A JP2010522907A JP2010538457A JP 2010538457 A JP2010538457 A JP 2010538457A JP 2010522907 A JP2010522907 A JP 2010522907A JP 2010522907 A JP2010522907 A JP 2010522907A JP 2010538457 A JP2010538457 A JP 2010538457A
- Authority
- JP
- Japan
- Prior art keywords
- composition
- polyelectrolyte
- copper
- complexing agent
- cmp
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K3/00—Materials not provided for elsewhere
- C09K3/14—Anti-slip materials; Abrasives
- C09K3/1454—Abrasive powders, suspensions and pastes for polishing
- C09K3/1463—Aqueous liquid suspensions
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09G—POLISHING COMPOSITIONS; SKI WAXES
- C09G1/00—Polishing compositions
- C09G1/02—Polishing compositions containing abrasives or grinding agents
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10P—GENERIC PROCESSES OR APPARATUS FOR THE MANUFACTURE OR TREATMENT OF DEVICES COVERED BY CLASS H10
- H10P52/00—Grinding, lapping or polishing of wafers, substrates or parts of devices
- H10P52/40—Chemomechanical polishing [CMP]
- H10P52/403—Chemomechanical polishing [CMP] of conductive or resistive materials
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Treatment Of Semiconductor (AREA)
- Finish Polishing, Edge Sharpening, And Grinding By Specific Grinding Devices (AREA)
Abstract
Description
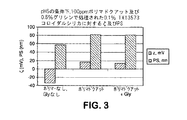
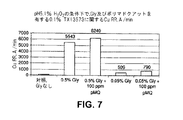
本発明のCMP組成物は、1質量パーセントの過酸化水素の存在下で4インチ径の銅ブランケットウエハを研磨するために利用された。2つの組成物は、0.1質量パーセントのコロイダルシリカ研磨剤(60nmの平均粒子サイズ)、15,000g/molの重量平均分子量を有する100ppmのポリ(マドクアット)を含み、さらに0.05か0.5質量パーセントのグリシンが併用された。他の2つの組成物は0.1質量パーセントの二酸化チタン及び100ppmのポリ(マドクアット)を含み、さらに0.05か1質量パーセントのグリシンが併用された。比較例を、研磨剤だけと、研磨剤と高分子電解質(グリシンを含まない)と、さらに研磨剤とグリシン(高分子電解質を含まない)とを含有する組成物とした。各々の組成物はpH5を有した。ウエハは、次の作業条件下でLogitech Model II CDP(イギリス、Glasgow、Logitech Ltd.製)上で研磨した。作業条件は、DlOO研磨パッド、80回毎分(rpm)のプラテンスピード、75rpmのキャリアスピード、3ポンド毎平方インチ(psi)のダウンフォース、及び200ミリリッター毎分(mL/min)のスラリー流速であった。
本発明のCMP組成物は4インチ径の銅ブランケットウエハを研磨するために利用された。その組成物は、0.1質量パーセントのコロイダルシリカ研磨剤(60nmの平均粒子サイズ)、200,000g/molの重量平均分子量を有して60:40のPAAのPAMに対するモル比を有する100〜1000ppmのPAA-PAMコポリマーを含み、さらに1質量パーセントのIDAが併用された。ウエハは、0.8〜1.6質量パーセントの範囲の様々な濃度の過酸化水素の存在下であって、5〜7の範囲のpHであって、さらに次の作業条件下でLogitech Model II CDP(イギリス、Glasgow、Logitech Ltd.製)上で研磨した。作業条件は、DlOO研磨パッド、80rpmのプラテンスピード、75rpmのキャリアスピード、3psiのダウンフォース、及び200mL/minのスラリー流速であった。
本発明のCMP組成物は、4インチ径の銅ブランケットウエハを研磨するために利用された。その組成物は、0.1質量パーセントのコロイダルシリカ研磨剤(60nmの平均粒子サイズ)、1000ppmのDISPERBYKTM191、及び0.1質量パーセントのシリコーングリコールコポリマーの非イオン性界面活性剤(SILWETTML7604、Connecticut、Danbury、OSi Specialties、報告によれば5〜8の範囲のHLBを有する。)を含み、さらに、1質量パーセントのIDAが併用された。ウエハは、0.8質量パーセントの過酸化水素又は0.1質量パーセントの過ヨウ素酸の存在下であって、pH7で、さらに次の作業条件下でLogitech Model II CDP(イギリス、Glasgow、Logitech Ltd.製)上で研磨した。作業条件は、DlOO研磨パッド、80rpmのプラテンスピード、75rpmのキャリアスピード、1psi又は3psiのダウンフォース、及び150mL/minのスラリー流速であった。各々の場合において、1psiダウンフォースの銅除去速度は1200Å/minであり、3psiダウンフォースの銅除去速度は3200Å/minであった。その組成物に対するスタチックエッチング速度は各々の酸化剤について18Å/minであった。
Claims (25)
- 銅含有基材を研磨するための化学機械研磨(CMP)組成物であって、該組成物が、
(a)1質量パーセント以下の粒子状研磨剤、
(b)高分子電解質、
(c)銅錯化剤、及び
(d)水性キャリア、
を含む、組成物。 - 前記高分子電解質が少なくとも10,000グラム-パー-モル(g/mol)の重量平均分子量を有する、請求項1に記載の組成物。
- 前記高分子電解質がアニオン性又は両性ポリマーを含む、請求項1に記載の組成物。
- 前記高分子電解質がアクリル酸のポリマー又はコポリマーを含む、請求項1に記載の組成物。
- 前記銅錯化剤がアミノポリカルボキシレートを含む、請求項1に記載の組成物。
- 前記高分子電解質がカチオン性ポリマーを含む、請求項1に記載の組成物。
- 前記銅錯化剤がアミノ酸を含む、請求項1に記載の組成物。
- 前記高分子電解質が50から1000ppmの範囲の濃度で前記組成物中に存在する、請求項1に記載の組成物。
- 前記銅錯化剤が0.5から1.5質量パーセントの範囲の濃度で前記組成物中に存在する、請求項1に記載の組成物。
- 粒子状研磨剤が100nm以下の平均粒子サイズを有する、請求項1に記載の組成物。
- 粒子状研磨剤が二酸化チタン及び二酸化ケイ素からなる群から選ばれる少なくとも一つの金属酸化物を含む、請求項1に記載の組成物。
- 銅含有基材を研磨するための化学機械研磨(CMP)組成物であって、該組成物が、
(a)100nm以下の平均粒子サイズを有する1質量パーセント以下の粒子状研磨剤、
(b)100から1000ppmのアニオン性又は両性高分子電解質、
(c)0.5から1.5質量パーセントのアミノポリカルボキシレート銅錯化剤、及び
(d)水性キャリア、
を含む、組成物。 - 前記高分子電解質が少なくとも50,000グラム-パー-モル(g/mol)の重量平均分子量を有する、請求項12に記載の組成物。
- 前記高分子電解質がアクリル酸のポリマー又はコポリマーを含む、請求項12に記載の組成物。
- 前記高分子電解質がアクリル酸とアクリルアミドのコポリマーを含む、請求項12に記載の組成物。
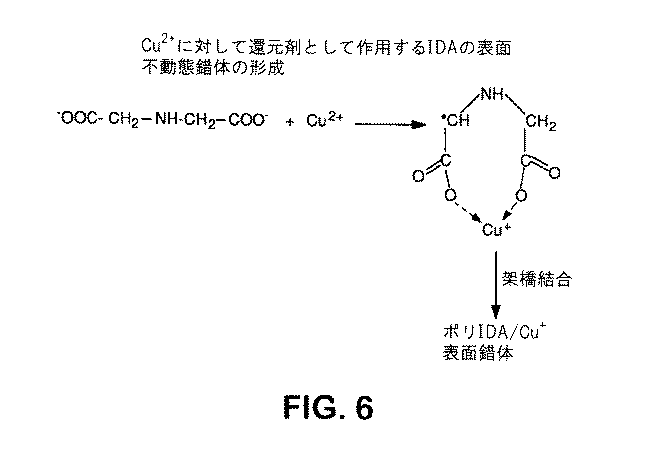
- 前記アミノポリカルボキシレートがイミノ二酢酸又はその塩を含む、請求項12に記載の組成物。
- 前記粒子状研磨剤が二酸化チタン及び二酸化ケイ素からなる群から選ばれる少なくとも一つの金属酸化物を含む、請求項12に記載の組成物。
- 銅含有基材を研磨するための化学機械研磨(CMP)組成物であって、該組成物が、
(a)100nm以下の平均粒子サイズを有する1質量パーセント以下の粒子状研磨剤、
(b)10から150ppmのカチオン性高分子電解質、
(c)0.5から1.5質量パーセントのアミノ酸銅錯化剤、及び
(d)水性キャリア、
を含む、組成物。 - 前記高分子電解質が少なくとも15,000グラム-パー-モル(g/mol)の重量平均分子量を有する、請求項18に記載の組成物。
- 前記カチオン性高分子電解質がポリ(2-[(メタクリロイルオキシ)エチル]トリメチル塩化アンモニウム)を含む、請求項18に記載の組成物。
- 前記アミノ酸がグリシンを含む、請求項18に記載の組成物。
- 前記粒子状研磨剤が二酸化チタン及び二酸化ケイ素からなる群から選ばれる少なくとも一つの金属酸化物を含む、請求項18に記載の組成物。
- 銅含有基材を研磨する方法であって、該方法が、請求項1に記載のCMP組成物で該基材の表面を摩耗させること含み、適宜、酸化剤の存在下で摩耗させることを含む、方法。
- 前記CMP組成物が、100から1000ppmの高分子電解質及び0.5から1.5質量パーセントの銅錯化剤を含み、並びに該高分子電解質がアニオン性又は両性ポリマーを含み、さらに該銅錯化剤がアミノポリカルボキシレート化合物を含む、請求項23に記載の方法。
- 前記CMP組成物が、10から150ppmの高分子電解質及び0.5から1.5質量パーセントの銅錯化剤を含み、並びに該高分子電解質がカチオン性ポリマーを含み、さらに該銅錯化剤がアミノ酸を含む、請求項23に記載の方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/895,896 | 2007-08-28 | ||
| US11/895,896 US20090056231A1 (en) | 2007-08-28 | 2007-08-28 | Copper CMP composition containing ionic polyelectrolyte and method |
| PCT/US2008/009852 WO2009032065A1 (en) | 2007-08-28 | 2008-08-19 | Copper cmp composition containing ionic polyelectrolyte and method |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2010538457A true JP2010538457A (ja) | 2010-12-09 |
| JP2010538457A5 JP2010538457A5 (ja) | 2015-09-24 |
| JP5960386B2 JP5960386B2 (ja) | 2016-08-02 |
Family
ID=40405295
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2010522907A Active JP5960386B2 (ja) | 2007-08-28 | 2008-08-19 | イオン性高分子電解質を含有する銅cmp組成物及び方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20090056231A1 (ja) |
| EP (1) | EP2190947A4 (ja) |
| JP (1) | JP5960386B2 (ja) |
| KR (1) | KR101305840B1 (ja) |
| CN (1) | CN101796160B (ja) |
| SG (1) | SG183780A1 (ja) |
| TW (1) | TWI434918B (ja) |
| WO (1) | WO2009032065A1 (ja) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012503880A (ja) * | 2008-09-26 | 2012-02-09 | ローディア・オペラシオン | 化学機械研磨用研磨剤組成物及びその使用法 |
| WO2014030570A1 (ja) * | 2012-08-23 | 2014-02-27 | 株式会社 フジミインコーポレーテッド | 研磨用組成物、研磨用組成物の製造方法、及び研磨用組成物原液の製造方法 |
| KR20140119096A (ko) | 2012-02-01 | 2014-10-08 | 히타치가세이가부시끼가이샤 | 금속용 연마액 및 연마 방법 |
| JP2016500747A (ja) * | 2012-11-02 | 2016-01-14 | ローレンス リバモア ナショナル セキュリティー, エルエルシー | 表面活性を失うことなく荷電コロイドの凝集を阻止する方法 |
| KR20190091467A (ko) | 2016-12-22 | 2019-08-06 | 카오카부시키가이샤 | 실리콘 웨이퍼용 린스제 조성물 |
| JP2019160875A (ja) * | 2018-03-08 | 2019-09-19 | 株式会社フジミインコーポレーテッド | 表面処理組成物、表面処理組成物の製造方法、表面処理方法および半導体基板の製造方法 |
| JP2021005704A (ja) * | 2019-06-26 | 2021-01-14 | 花王株式会社 | 酸化珪素膜用研磨液組成物 |
| JP2023536850A (ja) * | 2020-07-29 | 2023-08-30 | バーサム マテリアルズ ユーエス,リミティド ライアビリティ カンパニー | 銅及びスルーシリコンビア(tsv)化学機械平坦化(cmp)のためのパッドインボトル(pib)技術 |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101665661A (zh) * | 2008-09-05 | 2010-03-10 | 安集微电子科技(上海)有限公司 | 胺类化合物的应用以及一种化学机械抛光液 |
| US20120186572A1 (en) * | 2009-07-28 | 2012-07-26 | Helmuth Treichel | Silicon wafer sawing fluid and process for use thereof |
| JP5774283B2 (ja) * | 2010-04-08 | 2015-09-09 | 株式会社フジミインコーポレーテッド | 研磨用組成物及び研磨方法 |
| US8778212B2 (en) | 2012-05-22 | 2014-07-15 | Cabot Microelectronics Corporation | CMP composition containing zirconia particles and method of use |
| US8778211B2 (en) * | 2012-07-17 | 2014-07-15 | Cabot Microelectronics Corporation | GST CMP slurries |
| CN103865402A (zh) * | 2012-12-17 | 2014-06-18 | 安集微电子(上海)有限公司 | 一种化学机械抛光液 |
| US9303187B2 (en) | 2013-07-22 | 2016-04-05 | Cabot Microelectronics Corporation | Compositions and methods for CMP of silicon oxide, silicon nitride, and polysilicon materials |
| JP6400897B2 (ja) * | 2013-11-06 | 2018-10-03 | ニッタ・ハース株式会社 | 研磨組成物 |
| US10406652B2 (en) * | 2014-03-28 | 2019-09-10 | Fujimi Incorporated | Polishing composition and polishing method using the same |
| US9914852B2 (en) * | 2014-08-19 | 2018-03-13 | Fujifilm Planar Solutions, LLC | Reduction in large particle counts in polishing slurries |
| KR101874996B1 (ko) * | 2016-12-27 | 2018-07-05 | 한남대학교 산학협력단 | 연마효율이 우수한 화학-기계적 연마 슬러리 |
| US10170335B1 (en) * | 2017-09-21 | 2019-01-01 | Rohm And Haas Electronic Materials Cmp Holdings, Inc. | Chemical mechanical polishing method for cobalt |
| CN108930058B (zh) * | 2018-07-06 | 2020-07-21 | 鹤山市精工制版有限公司 | 一种电化学处理液及其应用 |
| US12234382B2 (en) * | 2020-07-28 | 2025-02-25 | Cmc Materials Llc | CMP composition including anionic and cationic inhibitors |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6348076B1 (en) * | 1999-10-08 | 2002-02-19 | International Business Machines Corporation | Slurry for mechanical polishing (CMP) of metals and use thereof |
| JP2002517593A (ja) * | 1998-06-10 | 2002-06-18 | ロデール ホールディングス インコーポレイテッド | 金属cmpにおける研磨用組成物および研磨方法 |
| JP2003303791A (ja) * | 2002-04-10 | 2003-10-24 | Nippon Shokubai Co Ltd | 化学機械研磨用水系分散体 |
| US20040229461A1 (en) * | 2003-05-12 | 2004-11-18 | Michael Darsillo | Chemical mechanical polishing compositions for copper and associated materials and method of using same |
| JP2005513765A (ja) * | 2001-12-05 | 2005-05-12 | キャボット マイクロエレクトロニクス コーポレイション | ポリマー性錯体形成剤を使用する銅cmpのための方法 |
| JP2005518091A (ja) * | 2002-02-11 | 2005-06-16 | キャボット マイクロエレクトロニクス コーポレイション | Cmpのための正電荷高分子電解質で処理したアニオン性研磨粒子 |
| JP2006511931A (ja) * | 2002-08-05 | 2006-04-06 | ピーピージー インダストリーズ オハイオ, インコーポレイテッド | 研磨スラリー系ならびに金属研磨および除去プロセス |
| JP2006093580A (ja) * | 2004-09-27 | 2006-04-06 | Fuji Photo Film Co Ltd | 化学的機械的研磨方法 |
| JP2007088424A (ja) * | 2005-08-24 | 2007-04-05 | Jsr Corp | 化学機械研磨用水系分散体、該水系分散体を調製するためのキット、化学機械研磨方法、および半導体装置の製造方法 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0852615B1 (en) * | 1996-07-25 | 2005-12-14 | DuPont Air Products NanoMaterials L.L.C. | Chemical mechanical polishing composition and process |
| US5958288A (en) * | 1996-11-26 | 1999-09-28 | Cabot Corporation | Composition and slurry useful for metal CMP |
| US6217416B1 (en) * | 1998-06-26 | 2001-04-17 | Cabot Microelectronics Corporation | Chemical mechanical polishing slurry useful for copper/tantalum substrates |
| US6503418B2 (en) * | 1999-11-04 | 2003-01-07 | Advanced Micro Devices, Inc. | Ta barrier slurry containing an organic additive |
| JP4213858B2 (ja) * | 2000-02-03 | 2009-01-21 | 花王株式会社 | 研磨液組成物 |
| TW586157B (en) * | 2000-04-13 | 2004-05-01 | Showa Denko Kk | Slurry composition for polishing semiconductor device, and method for manufacturing semiconductor device using the same |
| US6733553B2 (en) * | 2000-04-13 | 2004-05-11 | Showa Denko Kabushiki Kaisha | Abrasive composition for polishing semiconductor device and method for producing semiconductor device using the same |
| US6964923B1 (en) * | 2000-05-24 | 2005-11-15 | International Business Machines Corporation | Selective polishing with slurries containing polyelectrolytes |
| US6568997B2 (en) * | 2001-04-05 | 2003-05-27 | Rodel Holdings, Inc. | CMP polishing composition for semiconductor devices containing organic polymer particles |
| US6918820B2 (en) * | 2003-04-11 | 2005-07-19 | Eastman Kodak Company | Polishing compositions comprising polymeric cores having inorganic surface particles and method of use |
| US20060138087A1 (en) * | 2004-12-29 | 2006-06-29 | Simka Harsono S | Copper containing abrasive particles to modify reactivity and performance of copper CMP slurries |
| JP4776269B2 (ja) * | 2005-04-28 | 2011-09-21 | 株式会社東芝 | 金属膜cmp用スラリー、および半導体装置の製造方法 |
| KR100641348B1 (ko) * | 2005-06-03 | 2006-11-03 | 주식회사 케이씨텍 | Cmp용 슬러리와 이의 제조 방법 및 기판의 연마 방법 |
| WO2007077886A1 (ja) * | 2005-12-27 | 2007-07-12 | Hitachi Chemical Co., Ltd. | 金属用研磨液及び被研磨膜の研磨方法 |
-
2007
- 2007-08-28 US US11/895,896 patent/US20090056231A1/en not_active Abandoned
-
2008
- 2008-08-19 KR KR1020107006627A patent/KR101305840B1/ko active Active
- 2008-08-19 SG SG2012063707A patent/SG183780A1/en unknown
- 2008-08-19 CN CN200880104906.0A patent/CN101796160B/zh active Active
- 2008-08-19 JP JP2010522907A patent/JP5960386B2/ja active Active
- 2008-08-19 EP EP08795428.5A patent/EP2190947A4/en not_active Ceased
- 2008-08-19 WO PCT/US2008/009852 patent/WO2009032065A1/en not_active Ceased
- 2008-08-20 TW TW097131763A patent/TWI434918B/zh active
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002517593A (ja) * | 1998-06-10 | 2002-06-18 | ロデール ホールディングス インコーポレイテッド | 金属cmpにおける研磨用組成物および研磨方法 |
| US6348076B1 (en) * | 1999-10-08 | 2002-02-19 | International Business Machines Corporation | Slurry for mechanical polishing (CMP) of metals and use thereof |
| JP2005513765A (ja) * | 2001-12-05 | 2005-05-12 | キャボット マイクロエレクトロニクス コーポレイション | ポリマー性錯体形成剤を使用する銅cmpのための方法 |
| JP2005518091A (ja) * | 2002-02-11 | 2005-06-16 | キャボット マイクロエレクトロニクス コーポレイション | Cmpのための正電荷高分子電解質で処理したアニオン性研磨粒子 |
| JP2003303791A (ja) * | 2002-04-10 | 2003-10-24 | Nippon Shokubai Co Ltd | 化学機械研磨用水系分散体 |
| JP2006511931A (ja) * | 2002-08-05 | 2006-04-06 | ピーピージー インダストリーズ オハイオ, インコーポレイテッド | 研磨スラリー系ならびに金属研磨および除去プロセス |
| US20040229461A1 (en) * | 2003-05-12 | 2004-11-18 | Michael Darsillo | Chemical mechanical polishing compositions for copper and associated materials and method of using same |
| JP2006093580A (ja) * | 2004-09-27 | 2006-04-06 | Fuji Photo Film Co Ltd | 化学的機械的研磨方法 |
| JP2007088424A (ja) * | 2005-08-24 | 2007-04-05 | Jsr Corp | 化学機械研磨用水系分散体、該水系分散体を調製するためのキット、化学機械研磨方法、および半導体装置の製造方法 |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012503880A (ja) * | 2008-09-26 | 2012-02-09 | ローディア・オペラシオン | 化学機械研磨用研磨剤組成物及びその使用法 |
| KR20140119096A (ko) | 2012-02-01 | 2014-10-08 | 히타치가세이가부시끼가이샤 | 금속용 연마액 및 연마 방법 |
| US10037894B2 (en) | 2012-02-01 | 2018-07-31 | Hitachi Chemical Company, Ltd. | Polishing liquid for metal and polishing method |
| WO2014030570A1 (ja) * | 2012-08-23 | 2014-02-27 | 株式会社 フジミインコーポレーテッド | 研磨用組成物、研磨用組成物の製造方法、及び研磨用組成物原液の製造方法 |
| US9650544B2 (en) | 2012-08-23 | 2017-05-16 | Fujimi Incorporated | Polishing composition, method for manufacturing polishing composition, and method for manufacturing polishing composition liquid concentrate |
| JP2016500747A (ja) * | 2012-11-02 | 2016-01-14 | ローレンス リバモア ナショナル セキュリティー, エルエルシー | 表面活性を失うことなく荷電コロイドの凝集を阻止する方法 |
| US10865368B2 (en) | 2016-12-22 | 2020-12-15 | Kao Corporation | Rinse agent composition for silicon wafers |
| KR20190091467A (ko) | 2016-12-22 | 2019-08-06 | 카오카부시키가이샤 | 실리콘 웨이퍼용 린스제 조성물 |
| DE112017006489T5 (de) | 2016-12-22 | 2019-09-12 | Kao Corporation | Spülmittelzusammensetzung für Silicium-Wafer |
| JP2019160875A (ja) * | 2018-03-08 | 2019-09-19 | 株式会社フジミインコーポレーテッド | 表面処理組成物、表面処理組成物の製造方法、表面処理方法および半導体基板の製造方法 |
| JP7330668B2 (ja) | 2018-03-08 | 2023-08-22 | 株式会社フジミインコーポレーテッド | 表面処理組成物、表面処理組成物の製造方法、表面処理方法および半導体基板の製造方法 |
| JP2021005704A (ja) * | 2019-06-26 | 2021-01-14 | 花王株式会社 | 酸化珪素膜用研磨液組成物 |
| JP7041714B2 (ja) | 2019-06-26 | 2022-03-24 | 花王株式会社 | 酸化珪素膜用研磨液組成物 |
| TWI796575B (zh) * | 2019-06-26 | 2023-03-21 | 日商花王股份有限公司 | 氧化矽膜用研磨液組合物 |
| JP2023536850A (ja) * | 2020-07-29 | 2023-08-30 | バーサム マテリアルズ ユーエス,リミティド ライアビリティ カンパニー | 銅及びスルーシリコンビア(tsv)化学機械平坦化(cmp)のためのパッドインボトル(pib)技術 |
| JP7709517B2 (ja) | 2020-07-29 | 2025-07-16 | バーサム マテリアルズ ユーエス,リミティド ライアビリティ カンパニー | 銅及びスルーシリコンビア(tsv)化学機械平坦化(cmp)のためのパッドインボトル(pib)技術 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2190947A4 (en) | 2013-04-24 |
| EP2190947A1 (en) | 2010-06-02 |
| TWI434918B (zh) | 2014-04-21 |
| KR101305840B1 (ko) | 2013-09-23 |
| US20090056231A1 (en) | 2009-03-05 |
| JP5960386B2 (ja) | 2016-08-02 |
| WO2009032065A1 (en) | 2009-03-12 |
| KR20100065341A (ko) | 2010-06-16 |
| CN101796160B (zh) | 2013-07-31 |
| TW200927897A (en) | 2009-07-01 |
| SG183780A1 (en) | 2012-09-27 |
| CN101796160A (zh) | 2010-08-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5960386B2 (ja) | イオン性高分子電解質を含有する銅cmp組成物及び方法 | |
| KR101173753B1 (ko) | 구리-부동태화 cmp 조성물 및 방법 | |
| US8435421B2 (en) | Metal-passivating CMP compositions and methods | |
| JP6272842B2 (ja) | モリブデン研磨のための組成物および方法 | |
| KR102390227B1 (ko) | 텅스텐 버핑용 조성물 | |
| TWI865805B (zh) | 包含陰離子性及陽離子性抑制劑之cmp組合物 | |
| KR20210099166A (ko) | 텅스텐 cmp용 조성물 | |
| JP2010538457A5 (ja) | ||
| JP2020536386A (ja) | タングステンバフ用途のための表面処理研削粒子 | |
| TWI667337B (zh) | 用於研磨銅的cmp漿料組合物及使用其的研磨方法 | |
| JP5992925B2 (ja) | 金属を不動態化する化学機械研磨用組成物及び方法 | |
| WO2024064127A1 (en) | Tungsten cmp composition including a sulfur containing anionic surfactant |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110819 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110819 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130205 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130502 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130513 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130718 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20131022 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140224 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20140327 |
|
| A912 | Re-examination (zenchi) completed and case transferred to appeal board |
Free format text: JAPANESE INTERMEDIATE CODE: A912 Effective date: 20140523 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20150520 |
|
| A524 | Written submission of copy of amendment under article 19 pct |
Free format text: JAPANESE INTERMEDIATE CODE: A524 Effective date: 20150807 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160329 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20160623 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5960386 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |