EP0831165A2 - Mittel zur Vorbehandlung von Fasern - Google Patents
Mittel zur Vorbehandlung von Fasern Download PDFInfo
- Publication number
- EP0831165A2 EP0831165A2 EP97115512A EP97115512A EP0831165A2 EP 0831165 A2 EP0831165 A2 EP 0831165A2 EP 97115512 A EP97115512 A EP 97115512A EP 97115512 A EP97115512 A EP 97115512A EP 0831165 A2 EP0831165 A2 EP 0831165A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- acid
- bleaching
- mixtures
- fibers
- agents
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/322—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing nitrogen
- D06M13/325—Amines
- D06M13/342—Amino-carboxylic acids; Betaines; Aminosulfonic acids; Sulfo-betaines
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
- D06L4/10—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs using agents which develop oxygen
- D06L4/15—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs using agents which develop oxygen using organic agents
Definitions
- the invention relates to agents for pretreating fibers containing iminodisuccinic acids, their salts and their mixtures with one another, which may have an additional content of citric acid, gluconic acid and / or magnesium in ionic form, for pretreating bleaching with H 2 O 2 .
- agents for pretreating fibers containing iminodisuccinic acids, their salts and their mixtures with one another which may have an additional content of citric acid, gluconic acid and / or magnesium in ionic form, for pretreating bleaching with H 2 O 2 .
- Such agents are used in aqueous solution.
- Textile-forming natural vegetable fibers such as cotton, sisal, jute and others and animal fibers such as silk and wool contain waxes, fats and in raw form other plant components that cause a yellowish-brown coloring of the fiber. This means that not all of the desired colors are possible; the colorations are also uneven.
- a pretreatment of these fibers is therefore carried out in bleaching and washing are combined. Such pretreatment can are applied to the fibers of the named origin, to those made from them Yarns and on fabrics, knitted fabrics or nonwovens of such fibers. This pretreatment can also be applied to mixtures of such fibers with synthetic Fibers and products made from them.
- So-called bleaching liquors which contain water, hydrogen peroxide, wetting / washing and emulsifying agents, alkali for adjusting the pH and H 2 O 2 regulators (bleaching regulators, stabilizers) are used to carry out the aforementioned combined pretreatment.
- water glass and inorganic phosphates have long been used as regulators.
- water glass (sodium silicate) as a bleach stabilizer, insoluble calcium silicate deposits appear on the machine and material, especially in connection with calcium salts. Because of the over-fertilization of the waste water, the inorganic phosphates were later replaced by (poly) -phosphonates.
- EDTA ethylenediamine-tetraacetic acid
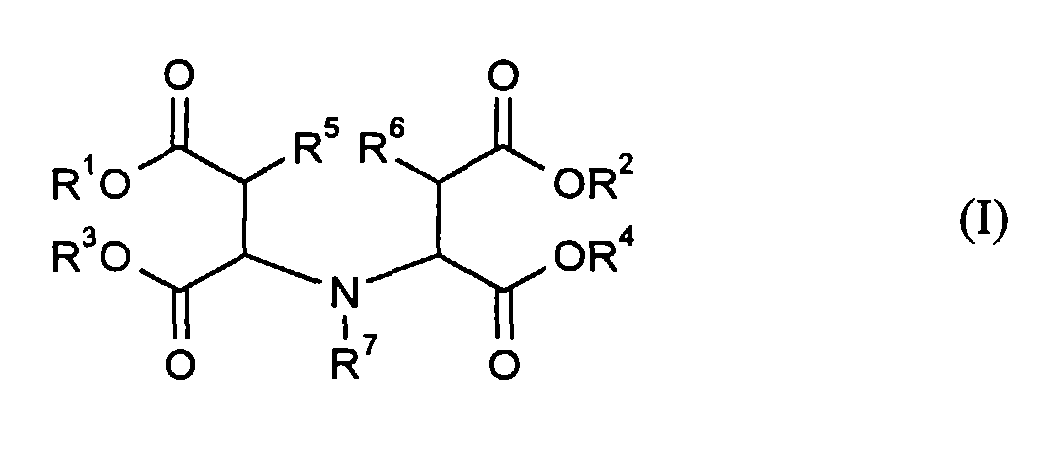
- R 6 preferably stands for H, particularly preferably R 5 and R 6 stand for H.
- R 7 also stands for H.
- R 5 , R 6 and R 7 stand for H, so that (I ) represents the unsubstituted iminodisuccinic acid and its salts (both, iminodisuccinic acid and its salts, collectively referred to as IDS) of the type described above.
- R 1 , R 2 , R 3 and R 4 are more preferably replaced by R 11 , R 12 , R 13 and R 14 with the meaning of H, Na, K, NH 4 or H 3 NCH 2 CH 2 OH.
- the agents used according to the invention as bleach stabilizers, containing substances of the formula (I), are in pure form or are associated with small amounts of secondary components from the preparation of (I), such as maleic acid, fumaric acid, aspartic acid, malic acid, asparagine, tartaric acid, hydroxyaspartic acid, Condensates of aspartic acid etc. or their salts with the cations R 1 to R 4 indicated above.
- the secondary components are present in the mixture in an amount of at most 35% by weight, preferably 30% by weight and particularly preferably 25% by weight; the rest is 100% by weight (I).
- (I) is prepared by known processes in an aqueous medium, for example from maleic anhydride, maleic acid or epoxysuccinic acid and ammonia or aspartic acid. Methods of this kind are described in GB 1 306 331, SU 0 639 863, JP 6/329 607, JP 6/330 020 and DE 3 739 610.
- an alkali metal hydroxide or NH 3 can be added until a pH of 7.5 to 14 is reached.
- a broad pH range up to a strongly alkaline setting is in principle possible because alkali must be added to the bleaching liquor to which the agents according to the invention are added.
- a lower pH value has proven to be more favorable.
- sodium hydroxide is preferred as the alkali hydroxide, although in principle KOH or LiOH are also possible; it can be used both in solid form and in the easy-to-handle aqueous solution with, for example, 10 to 60% by weight.
- the invention further relates to the use of the above agents containing Iminodisuccinic acids and their salts of the formula (I), as bleach regulators and thus also a process for the pretreatment of natural textiles vegetable or animal fibers or their mixtures with synthetic fibers or of yarns, woven fabrics, knitted fabrics or nonwovens made from such fibers or their Bleaching in bleaching liquors containing water, alkali, hydrogen peroxide, Contain wetting agents, detergents and emulsifiers as well as bleach regulators is characterized in that agents of the type described above as bleach regulators be used.
- the pretreatment process can take a number of different forms are carried out, all of which are known to the person skilled in the art as prior art are: pretreatment in a long liquor (discontinuous or continuous); Block-cold retention process (KKV); Pad steam process; Pad roll process and other.
- Textile-forming natural vegetable fibers for example those made from cellulose, such as cotton, jute, linen or regenerated cellulose, as well as animal fibers, such as silk and wool, as well as their mixtures with synthetics can according to the invention be bleached.
- Vegetable fibers are particularly preferred preferably called cotton and mixtures thereof.
- synthetic mixed ingredients be synthetic polyamide, such as nylon or Perlon, polyester and Called polyacrylonitrile.
- bleaching liquors to be used according to the invention in addition to water, Alkali hydroxide, wetting, washing and emulsifying agents and agents according to the invention as bleaching regulators, hydrogen peroxide in an amount of 0.1 to 30 g / l used.
- the alkali hydroxide is added in an amount and added, that the stated pH of 7 to 14 is maintained in the bleaching liquor.
- Wetting agents, detergents and emulsifiers are those that are active in this field Are known to those skilled in the art.
- anionic, cationic or non-ionic surfactants are individual substances or mixtures from the known anionic, cationic or non-ionic surfactants; it is preferably anionic or nonionic surfactants, such as Fatty acids and their salts, fatty acid alkyl esters, fatty alcohols, glycerides, alkyl aromatic sulfonic acids, their polyglycol ethers and others known manner, these surfactants are chosen and compiled so that the unwanted accompanying substances of natural vegetable and animal fibers, such as fats, waxes and other plant components (e.g. remains of seed capsules etc.) and animal non-fibrous components are removed.
- water can demineralized water or a customarily provided service water, the has a different degree of hardness depending on the occurrence.
- the Agents serve as stabilizers for the hydrogen peroxide. The levy regulates the oxygen to bleach the fibers.
- the agent containing substances Formula (I) is used for complexing and "masking" alkaline earth metals, especially the disruptive Ca ions and heavy metals.
- Mg ions and Ca ions as an example of alkaline earth ions and Fe as an example of Heavy metal ions that are to be complexed are natural components of the usually available process water. Alkaline earth ions and heavy metal ions can also be used as impurities in the natural vegetable to be bleached Fibers are introduced. When using demineralized water is missing both the Ca to be complexed and the one desired as a co-stabilizer Mg, while those introduced by impurities in the natural vegetable fibers Components must still be considered. Mg ions can therefore in amounts of 0.1 to 1 g / l the bleaching regulators in addition to citric acid and / or Gluconic acid can be added. Taking these dependencies into account of the available water and the quality of the fiber to be bleached are known to the person skilled in the art.
- the agents according to the invention can also be used.
- Typical bleaching liquors were prepared with water at 0-20 °, ie as follows: long fleet KKV bleach PAD-Steam Magnesium sulfate g / l 0.2 0.2 0.3 Bleach regulator IDS g / l 1.0 4.0 4.0 Sodium hydroxide solution (32% by weight) ml / l 6.0 30.0 20.0 Hydrogen peroxide (35% by weight) ml / l 10.0 60.0 40.0
- the composition had a pH of 6.7.
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Detergent Compositions (AREA)
- Paper (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
Abstract
Description
- R1, R2, R3 und R4
- unabhängig voneinander H, Li, Na, K, NH4, H3NCH2CH2OH, H2N(CH2CH2OH)2 oder HN(CH2CH2OH)3 bedeuten,
- R5 und R6
- unabhängig voneinander für H oder OH stehen und
- R7
- für H, CH2CH2OH, CH2CH2CH2OH, CH2CH(OH)CH3, CH2COOR8 oder CH2CH2COOR8 steht, worin R8 unabhängig von R1 den Bedeutungsumfang von R1 hat.
- Sengen, Abbrennen der abstehenden Faserenden, um eine glatte Oberfläche zu erzielen. Dies ist üblicherweise der 1. Arbeitsgang.
- Abkochen, Beuchen, d.h. heiße Alkalibehandlung mit dem Ziel einer VorExtraktion der Faserbegleitsubstanzen bzw. Aufquellung der Fasern und der Samenschalen der Baumwolle. Dies wird meist vor dem Bleichen durchgeführt.
- Laugieren, Merzerisieren, Behandlung mit hoch konzentriertem Alkali mehr oder weniger unter Spannung des Materiales zur Erzielung einer starken Faserquellung und damit Glanz sowie Eliminierung der unreifen bzw. toten Baumwollanteile, welche sich nicht oder schlecht anfärben lassen. Dies kann vor oder nach dem Bleichen durchgeführt werden.
- Säure-Extraktion, wird vor dem Bleichen durchgeführt, wenn extrem hohe Mengen an Schwermetallen vorliegen (aus dem Brauchwasser und/oder dem Fasersubstrat). Die Komplexbildung des Bleichregulators wäre sonst überfordert.
- Ferner kommen zur Erzielung sehr hoher Weißgrade auch weitere Bleichverfahren
vor oder nach der Wasserstoffperoxid-Bleiche zur Anwendung,
beispielsweise
- (a) die Natriumhypochlorit-Bleiche,
- (b) die Natriumchlorit-Bleiche,
- (c) die reduktive Bleiche und/oder
- (d) enzymatische Bleiche, jeweils
mit und ohne Zusatz von Weißtönern.
| lange Flotte | KKV-Bleiche | PAD-Steam | ||
| Magnesiumsulfat | g/l | 0,2 | 0,2 | 0,3 |
| Bleichregulator IDS | g/l | 1,0 | 4,0 | 4,0 |
| Natronlauge (32 gew.-%ig) | ml/l | 6,0 | 30,0 | 20,0 |
| Wasserstoffperoxid (35 gew.-%ig) | ml/l | 10,0 | 60,0 | 40,0 |
| Verfahren | |||
| A) | B) | C) | |
| Weißgrad Remission 460 nm (%) nach Bleiche | 81 | 70 | 70 |
| Verfahren | |||
| A) | B) | C) | |
| Weißgrad Remission 460 nm (%) nach Bleiche | 61 | 44 | 56 |
| Verfahren | |||
| A | B | C | |
| Weißgrad Remission 460 nm (%) nach Bleiche | 80 | 66 | 68 |
| Verfahren | |||
| A | B | C | |
| Weißgrad Remission 460 nm (%) nach Bleiche | 78 | 69 | 66 |
| Versuch | |||
| a) | b) | ||
| Natronlauge (32 gew.-%ig) | g/l | 6,0 | 6,0 |
| Regulator IDS | g/l | 0 | 4,0 |
| Wasserstoffperoxid (35 gew.-%ig) | ml/l | 10,0 | 10,0 |
| Fe+++Sulfatlösung (1:1000) | g/l | 20,0 | 20,0 |
| H2O2-Gehalt zu Beginn | ml/l | 10,4 | 10,2 |
| nach 15 min | ml/l | 0 | 5,7 |
| nach 30 min | ml/l | 0 | 2,4 |
Claims (9)
- Mittel zur Vorbehandlung von textilbildenden natürlichen pflanzlichen oder tierischen Fasern oder ihren Gemischen mit synthetischen Fasern oder von Garnen, Geweben, Gewirken oder Vliesen aus solchen Fasern oder ihren Gemischen durch Bleiche, enthaltend Iminodibernsteinsäuren, ihre Salze sowie ihre Mischungen untereinander der Formel in der
- R1, R2, R3 und R4
- unabhängig voneinander H, Li, Na, K, NH4, H3NCH2CH2OH, H2N(CH2CH2OH)2 oder HN(CH2CH2OH)3 bedeuten,
- R5 und R6
- unabhängig voneinander für H oder OH stehen und
- R7
- für H, CH2CH2OH, CH2CH2CH2OH, CH2CH(OH)CH3, CH2COOR8 oder CH2CH2COOR8 steht, worin R8 unabhängig von R1 den Bedeutungsumfang von R1 hat.
- Mittel nach Anspruch 1, dadurch gekennzeichnet, daß R6 für H steht, bevorzugt R5 und R6 für H stehen.
- Mittel nach Anspruch 1, dadurch gekennzeichnet, daß R7 für H steht, bevorzugt R5, R6 und R7 für H stehen.
- Mittel nach Anspruch 1, dadurch gekennzeichnet, daß an die Stelle von R1, R2, R3 und R4 die Reste R11, R12, R13 und R14 mit der Bedeutung von H, Na, K, NH4 oder H3NCH2CH2OH treten.
- Mittel nach Anspruch 1, dadurch gekennzeichnet, daß die Iminodibernsteinsäuren, ihre Salze oder Mischungen davon vergesellschaftet sind mit Maleinsäure, Fumarsäure, Asparaginsäure, Äpfelsäure, Asparagin, Weinsäure, Hydroxyasparaginsäure, Kondensate der Asparaginsäure oder deren Salzen, wobei die Vergesellschaftung maximal 35 Gew.-%, bevorzugt 30 Gew.-%, besonders bevorzugt 25 Gew.-% beträgt und wobei der Rest zu 100 Gew.-% (I) ist.
- Verfahren zur Vorbehandlung von textilbildenden natürlichen pflanzlichen oder tierischen Fasern oder ihren Gemischen mit synthetischen Fasern oder von Garnen, Geweben, Gewirken oder Vliesen aus solchen Fasern oder ihren Gemischen durch Bleiche in Bleichflotten, die Wasser, Alkali, Wasserstoffperoxid, Netz-, Wasch- und Emulgiermittel sowie Bleichregulatoren enthalten, dadurch gekennzeichnet, daß als Bleichregulatoren Mittel gemäß Anspruch 1 eingesetzt werden.
- Verfahren nach Anspruch 6, dadurch gekennzeichnet, daß als Fasern solche aus Cellulose, Regeneratcellulose, Wolle, Seide, Mischungen mit Synthesefasern oder ihre Verarbeitungsformen, bevorzugt solche aus Baumwolle und deren Mischungen vorbehandelt werden.
- Verfahren nach Anspruch 6, dadurch gekennzeichnet, daß die Mittel gemäß Anspruch 1 in einer Menge von 0,1 bis 30 g/l, bevorzugt 0,1 bis 20 g/l, besonders bevorzugt 0,2 bis 10 g/l in der Bleichflotte vorliegen.
- Verfahren nach Anspruch 6, dadurch gekennzeichnet, daß in der Bleichflotte zusätzlich zu den Mitteln gemäß Anspruch 1 eine oder mehrere Verbindungen aus der Gruppe von Zitronensäure, Gluconsäure, Polyasparaginsäure oder ihre Alkalisalze und Magnesiumionen vorliegen.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19638569A DE19638569A1 (de) | 1996-09-20 | 1996-09-20 | Bleichregulatoren und Bleichverfahren damit |
| DE19638569 | 1996-09-20 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0831165A2 true EP0831165A2 (de) | 1998-03-25 |
| EP0831165A3 EP0831165A3 (de) | 1998-07-15 |
Family
ID=7806336
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP97115512A Withdrawn EP0831165A3 (de) | 1996-09-20 | 1997-09-08 | Mittel zur Vorbehandlung von Fasern |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US6056787A (de) |
| EP (1) | EP0831165A3 (de) |
| JP (1) | JPH10110195A (de) |
| DE (1) | DE19638569A1 (de) |
| TR (1) | TR199700976A3 (de) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998045251A1 (de) * | 1997-04-04 | 1998-10-15 | Bayer Aktiengesellschaft | Herstellung und verwendung von iminodibernsteinsäuresalzen |
| EP0940495A1 (de) * | 1998-03-05 | 1999-09-08 | Bayer Aktiengesellschaft | Gleichzeitiges Waschen und Bleichen nativer Fasern und textiler Erzeugnisse daraus |
| WO2000068492A1 (de) * | 1999-05-08 | 2000-11-16 | Cognis Deutschland Gmbh | Verfahren zur rohwollwäsche |
| DE10301535A1 (de) * | 2003-01-17 | 2004-07-29 | Cht R. Beitlich Gmbh | Verfahren zur Veredlung von textilem Fasermaterial |
| EP1967577A1 (de) * | 2007-02-13 | 2008-09-10 | CHT R. BEITLICH GmbH | Katalysierte Peroxidbleiche ("Katalysator-Bleiche") |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1913194B1 (de) * | 2005-08-10 | 2015-02-18 | Perachem Limited | Verbesserungen bei oder in zusammenhang mit wollbehandlung |
| WO2020093280A1 (en) * | 2018-11-07 | 2020-05-14 | Kemira Oyj | A composition for use as peroxide stabilizer |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3755028A (en) * | 1967-12-18 | 1973-08-28 | Curlator Corp | Method for manufacturing non-woven textile articles |
| GB1306331A (de) * | 1970-12-07 | 1973-02-07 | ||
| SU639863A1 (ru) * | 1977-06-16 | 1978-12-30 | Калининский Ордена Трудового Красного Знамени Политехнический Институт Калининский Сельско-Хозяйственный Институт | Способ получени комплексона-иминоди нтарной кислоты |
| FR2545854B1 (fr) * | 1983-05-10 | 1985-07-26 | Ugine Kuhlmann | Procede de blanchiment des textiles en presence de particules de fer ou de metaux ferreux |
| DE3739610A1 (de) * | 1987-11-23 | 1989-06-01 | Basf Ag | Nitrilo-di-aepfel-mono-essigsaeuren, verfahren zu ihrer herstellung und ihre verwendung |
| US5362412A (en) * | 1991-04-17 | 1994-11-08 | Hampshire Chemical Corp. | Biodegradable bleach stabilizers for detergents |
| JPH06330020A (ja) * | 1993-05-20 | 1994-11-29 | Nippon Shokubai Co Ltd | キレート性組成物及びその製法並びに洗剤組成物 |
| JP3326867B2 (ja) * | 1993-05-24 | 2002-09-24 | 株式会社日本触媒 | イミノジコハク酸金属塩の製造方法 |
| US5670082A (en) * | 1993-06-11 | 1997-09-23 | Ciba-Geigy Corporation | Bleaching auxiliary |
| DE4340043A1 (de) * | 1993-11-24 | 1995-06-01 | Henkel Kgaa | Verwendung Stickstoff-haltiger Komplexbildner bei der Holzstoffbleiche |
| JPH09310097A (ja) * | 1996-05-22 | 1997-12-02 | Lion Corp | 高嵩密度漂白洗剤組成物 |
-
1996
- 1996-09-20 DE DE19638569A patent/DE19638569A1/de not_active Ceased
-
1997
- 1997-09-08 EP EP97115512A patent/EP0831165A3/de not_active Withdrawn
- 1997-09-12 JP JP9265132A patent/JPH10110195A/ja active Pending
- 1997-09-18 TR TR97/00976A patent/TR199700976A3/tr unknown
- 1997-09-19 US US08/933,769 patent/US6056787A/en not_active Expired - Fee Related
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998045251A1 (de) * | 1997-04-04 | 1998-10-15 | Bayer Aktiengesellschaft | Herstellung und verwendung von iminodibernsteinsäuresalzen |
| US6107518A (en) * | 1997-04-04 | 2000-08-22 | Bayer Aktiengesellschaft | Preparation and use of iminodisuccinic acid salts |
| US6207010B1 (en) | 1997-04-04 | 2001-03-27 | Bayer Aktiengesellschaft | Preparation and use of iminodisuccinic acid salts |
| EP1247800A1 (de) * | 1997-04-04 | 2002-10-09 | Bayer Ag | Verwendung von Iminodibernsteinsäuresalzen bei der Papierherstellung |
| EP0940495A1 (de) * | 1998-03-05 | 1999-09-08 | Bayer Aktiengesellschaft | Gleichzeitiges Waschen und Bleichen nativer Fasern und textiler Erzeugnisse daraus |
| US6096097A (en) * | 1998-03-05 | 2000-08-01 | Bayer Aktiengesellschaft | Simultaneous washing and bleaching of native fibres and textile products therefrom |
| WO2000068492A1 (de) * | 1999-05-08 | 2000-11-16 | Cognis Deutschland Gmbh | Verfahren zur rohwollwäsche |
| AU766557B2 (en) * | 1999-05-08 | 2003-10-16 | Cognis Ip Management Gmbh | Method for washing raw wool |
| DE10301535A1 (de) * | 2003-01-17 | 2004-07-29 | Cht R. Beitlich Gmbh | Verfahren zur Veredlung von textilem Fasermaterial |
| EP1967577A1 (de) * | 2007-02-13 | 2008-09-10 | CHT R. BEITLICH GmbH | Katalysierte Peroxidbleiche ("Katalysator-Bleiche") |
Also Published As
| Publication number | Publication date |
|---|---|
| TR199700976A2 (xx) | 1998-04-21 |
| TR199700976A3 (tr) | 1998-04-21 |
| DE19638569A1 (de) | 1998-04-02 |
| US6056787A (en) | 2000-05-02 |
| EP0831165A3 (de) | 1998-07-15 |
| JPH10110195A (ja) | 1998-04-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE2226784C3 (de) | Mittel zum Stabilisieren von Peroxyverbindungen in wäßrig-alkalischer Lösung und seine Verwendung | |
| DE976170C (de) | Verfahren zur Behandlung von Polyamidfasern mit peroxydhaltigen Bleich- oder Waschmitteln | |
| EP0233350B1 (de) | Silikat- und magnesiumfreie Peroxidstabilisatoren | |
| EP0557730B1 (de) | Bleichregulator-Zusammensetzungen und Bleichverfahren damit | |
| DE2735816A1 (de) | Verfahren zum bleichen und entschlichten von textilgut | |
| EP0831165A2 (de) | Mittel zur Vorbehandlung von Fasern | |
| DE60014975T2 (de) | Neue verfahren zur vorbehandlung von zellulosefaser und deren mischungen | |
| DE4208106B4 (de) | Vorbehandlung von Textilfasermaterial | |
| DE3820160C2 (de) | Konzentriertes Präparat und dessen Verwendung als Stabilisator für alkalische peroxydhaltige Flotten | |
| EP0315848B1 (de) | Verfahren zur Vorbehandlung von textilen Materialien | |
| EP1583861B1 (de) | Verfahren zur veredlung von textilem fasermaterial | |
| DE2012171C3 (de) | Vorbehandlung von Fasergut aus nativer Cellulose | |
| EP0775226B2 (de) | Verwendung eines stabilisierungsmittel-konzentrats für wässrige bleich- und behandlungslösungen auf basis von wasserstoffperoxid und/oder anderen anorganischen und/oder organischen peroxoverbindungen | |
| DE2554360C2 (de) | Verfahren zum Vorbehandeln und Veredeln von Fasergutaus nativer Cellulose | |
| EP0801165A2 (de) | Vorbehandlung von Textilien | |
| EP0997523B1 (de) | Wässrige Lösungen von Phosphonsäuren | |
| AT131584B (de) | Verfahren zum Waschen von Textilmaterialien. | |
| DE19533713C1 (de) | Wollschutzmittel für Naßbehandlungen bei Temperaturen oberhalb 98 DEG C, deren Verwendung bei Textilveredlungsprozessen sowie Verfahren zum Schutz von Wolle | |
| DE1617141C3 (de) | Verfahren zur Verminderung der Wiederverschmutzung von Wäsche wahrend des Waschens | |
| AT206859B (de) | Verfahren und Mittel zur Behandlung von synthetische Polyamide enthaltenden Fasern oder Faserprodukten | |
| DE2413867A1 (de) | Wirkstoffmischung fuer ein oberflaechenaktives, faserschonendes extraktionsmittel fuer cellulosefasern | |
| EP0671499A2 (de) | Bleichen von Synthesefasern | |
| DE1619081C3 (de) | Avivagemittel für Textilien | |
| DE3534524A1 (de) | Verfahren zum bleichen und oxidieren von textilien | |
| DE4424820A1 (de) | Bleichen von Synthesefasern |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): BE CH DE DK ES FR GB IT LI PT |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| 17P | Request for examination filed |
Effective date: 19990115 |
|
| AKX | Designation fees paid |
Free format text: BE CH DE DK ES FR GB IT LI PT |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): BE CH DE DK ES FR GB IT LI PT |
|
| 17Q | First examination report despatched |
Effective date: 20020927 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: BAYER CROPSCIENCE AG |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: BAYER AG |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: BAYER CHEMICALS AG |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: LANXESS DEUTSCHLAND GMBH |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20050803 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: LANXESS B.V. |