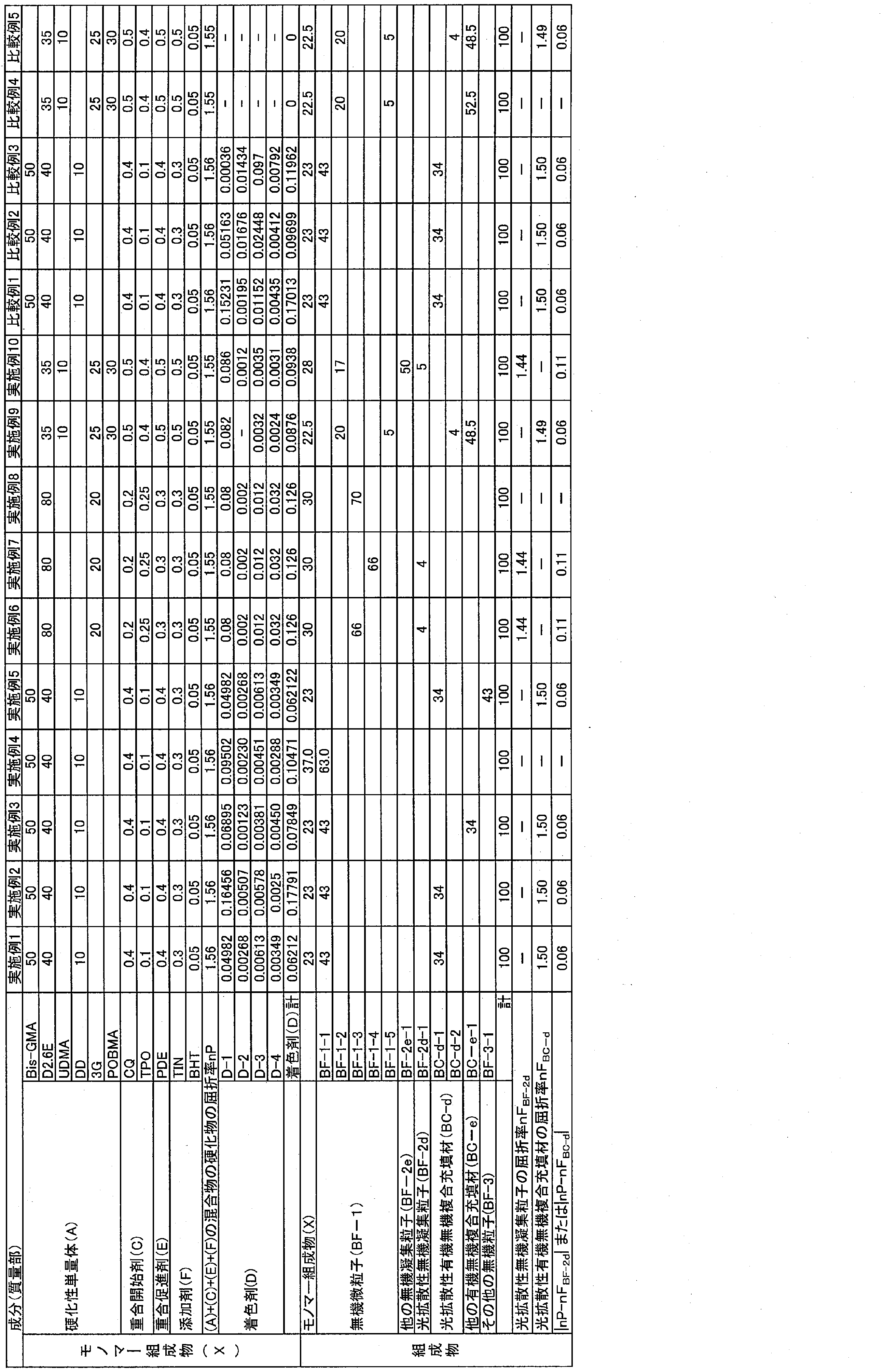
WO2022092193A1 - 良好な色調適合性を有する歯科用硬化性組成物 - Google Patents
良好な色調適合性を有する歯科用硬化性組成物 Download PDFInfo
- Publication number
- WO2022092193A1 WO2022092193A1 PCT/JP2021/039803 JP2021039803W WO2022092193A1 WO 2022092193 A1 WO2022092193 A1 WO 2022092193A1 JP 2021039803 W JP2021039803 W JP 2021039803W WO 2022092193 A1 WO2022092193 A1 WO 2022092193A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- curable composition
- inorganic
- dental curable
- filler
- light
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 243
- 239000000945 filler Substances 0.000 claims abstract description 126
- 239000000178 monomer Substances 0.000 claims abstract description 67
- 230000003595 spectral effect Effects 0.000 claims abstract description 36
- 239000003086 colorant Substances 0.000 claims abstract description 26
- 239000003505 polymerization initiator Substances 0.000 claims abstract description 22
- 239000002245 particle Substances 0.000 claims description 189
- 239000002131 composite material Substances 0.000 claims description 68
- 239000010419 fine particle Substances 0.000 claims description 34
- 239000011164 primary particle Substances 0.000 claims description 25
- 229920000642 polymer Polymers 0.000 claims description 15
- 230000000379 polymerizing effect Effects 0.000 claims description 8
- 230000004931 aggregating effect Effects 0.000 claims description 2
- -1 2,3-dibromopropyl Chemical group 0.000 description 108
- 239000000047 product Substances 0.000 description 75
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 57
- 238000000034 method Methods 0.000 description 52
- 235000019646 color tone Nutrition 0.000 description 49
- 239000011256 inorganic filler Substances 0.000 description 37
- 229910003475 inorganic filler Inorganic materials 0.000 description 37
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 36
- 238000011049 filling Methods 0.000 description 33
- 238000004519 manufacturing process Methods 0.000 description 31
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 29
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 28
- 239000011521 glass Substances 0.000 description 25
- 239000000463 material Substances 0.000 description 22
- 208000002925 dental caries Diseases 0.000 description 20
- 238000012360 testing method Methods 0.000 description 16
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 15
- 238000006116 polymerization reaction Methods 0.000 description 15
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 14
- 125000004432 carbon atom Chemical group C* 0.000 description 14
- 239000011350 dental composite resin Substances 0.000 description 14
- 238000010438 heat treatment Methods 0.000 description 14
- 238000005259 measurement Methods 0.000 description 14
- 239000001294 propane Substances 0.000 description 14
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 13
- 238000002156 mixing Methods 0.000 description 12
- AMFGWXWBFGVCKG-UHFFFAOYSA-N Panavia opaque Chemical compound C1=CC(OCC(O)COC(=O)C(=C)C)=CC=C1C(C)(C)C1=CC=C(OCC(O)COC(=O)C(C)=C)C=C1 AMFGWXWBFGVCKG-UHFFFAOYSA-N 0.000 description 11
- 125000003118 aryl group Chemical group 0.000 description 11
- 229910052757 nitrogen Inorganic materials 0.000 description 11
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 11
- 239000000049 pigment Substances 0.000 description 11
- 239000000843 powder Substances 0.000 description 11
- GOLORTLGFDVFDW-UHFFFAOYSA-N 3-(1h-benzimidazol-2-yl)-7-(diethylamino)chromen-2-one Chemical compound C1=CC=C2NC(C3=CC4=CC=C(C=C4OC3=O)N(CC)CC)=NC2=C1 GOLORTLGFDVFDW-UHFFFAOYSA-N 0.000 description 10
- XDLMVUHYZWKMMD-UHFFFAOYSA-N 3-trimethoxysilylpropyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCOC(=O)C(C)=C XDLMVUHYZWKMMD-UHFFFAOYSA-N 0.000 description 10
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 10
- 125000000217 alkyl group Chemical group 0.000 description 10
- 150000003839 salts Chemical class 0.000 description 10
- 238000004381 surface treatment Methods 0.000 description 10
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 9
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 9
- 229910052788 barium Inorganic materials 0.000 description 9
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 9
- 229910052796 boron Inorganic materials 0.000 description 9
- 230000001965 increasing effect Effects 0.000 description 9
- 238000005498 polishing Methods 0.000 description 9
- 239000000126 substance Substances 0.000 description 9
- 125000001931 aliphatic group Chemical group 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 8
- 238000007561 laser diffraction method Methods 0.000 description 8
- 238000000790 scattering method Methods 0.000 description 8
- 239000000377 silicon dioxide Substances 0.000 description 8
- 239000012756 surface treatment agent Substances 0.000 description 8
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 7
- 239000010954 inorganic particle Substances 0.000 description 7
- 239000012766 organic filler Substances 0.000 description 7
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 7
- 159000000000 sodium salts Chemical class 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- HWSSEYVMGDIFMH-UHFFFAOYSA-N 2-[2-[2-(2-methylprop-2-enoyloxy)ethoxy]ethoxy]ethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCOCCOCCOC(=O)C(C)=C HWSSEYVMGDIFMH-UHFFFAOYSA-N 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 6
- 239000006087 Silane Coupling Agent Substances 0.000 description 6
- 150000003926 acrylamides Chemical class 0.000 description 6
- 230000001588 bifunctional effect Effects 0.000 description 6
- 239000003999 initiator Substances 0.000 description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 6
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 6
- 230000008439 repair process Effects 0.000 description 6
- 229910000077 silane Inorganic materials 0.000 description 6
- QTUVQQKHBMGYEH-UHFFFAOYSA-N 2-(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC=NC=N1 QTUVQQKHBMGYEH-UHFFFAOYSA-N 0.000 description 5
- UEKHZPDUBLCUHN-UHFFFAOYSA-N 2-[[3,5,5-trimethyl-6-[2-(2-methylprop-2-enoyloxy)ethoxycarbonylamino]hexyl]carbamoyloxy]ethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCOC(=O)NCCC(C)CC(C)(C)CNC(=O)OCCOC(=O)C(C)=C UEKHZPDUBLCUHN-UHFFFAOYSA-N 0.000 description 5
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 5
- OKIZCWYLBDKLSU-UHFFFAOYSA-M N,N,N-Trimethylmethanaminium chloride Chemical class [Cl-].C[N+](C)(C)C OKIZCWYLBDKLSU-UHFFFAOYSA-M 0.000 description 5
- 239000002253 acid Substances 0.000 description 5
- 239000000654 additive Substances 0.000 description 5
- 230000000996 additive effect Effects 0.000 description 5
- 239000000853 adhesive Substances 0.000 description 5
- ODWXUNBKCRECNW-UHFFFAOYSA-M bromocopper(1+) Chemical compound Br[Cu+] ODWXUNBKCRECNW-UHFFFAOYSA-M 0.000 description 5
- 239000000919 ceramic Substances 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- 239000006185 dispersion Substances 0.000 description 5
- 238000009826 distribution Methods 0.000 description 5
- 238000009499 grossing Methods 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 5
- 239000003960 organic solvent Substances 0.000 description 5
- 238000007665 sagging Methods 0.000 description 5
- 239000002335 surface treatment layer Substances 0.000 description 5
- ATTSZAWLFKAZTF-UHFFFAOYSA-N (3-nitrophenyl)boron Chemical compound [B]C1=CC=CC([N+]([O-])=O)=C1 ATTSZAWLFKAZTF-UHFFFAOYSA-N 0.000 description 4
- PVLNHYPAZWPHDM-UHFFFAOYSA-N (4-chlorophenyl)boron Chemical compound [B]C1=CC=C(Cl)C=C1 PVLNHYPAZWPHDM-UHFFFAOYSA-N 0.000 description 4
- YGGMQWSJXSNGDT-UHFFFAOYSA-N (4-fluorophenyl)boron Chemical compound [B]C1=CC=C(F)C=C1 YGGMQWSJXSNGDT-UHFFFAOYSA-N 0.000 description 4
- RVSLSGJUKZJRIW-UHFFFAOYSA-N (4-nitrophenyl)boron Chemical compound [B]C1=CC=C([N+]([O-])=O)C=C1 RVSLSGJUKZJRIW-UHFFFAOYSA-N 0.000 description 4
- AZWDISHHPONIAX-UHFFFAOYSA-N 1-butylquinolin-1-ium Chemical class C1=CC=C2[N+](CCCC)=CC=CC2=C1 AZWDISHHPONIAX-UHFFFAOYSA-N 0.000 description 4
- OIDIRWZVUWCCCO-UHFFFAOYSA-N 1-ethylpyridin-1-ium Chemical class CC[N+]1=CC=CC=C1 OIDIRWZVUWCCCO-UHFFFAOYSA-N 0.000 description 4
- RTQPKEOYPPMVGQ-UHFFFAOYSA-N 1-methylquinolin-1-ium Chemical class C1=CC=C2[N+](C)=CC=CC2=C1 RTQPKEOYPPMVGQ-UHFFFAOYSA-N 0.000 description 4
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 description 4
- INQDDHNZXOAFFD-UHFFFAOYSA-N 2-[2-(2-prop-2-enoyloxyethoxy)ethoxy]ethyl prop-2-enoate Chemical compound C=CC(=O)OCCOCCOCCOC(=O)C=C INQDDHNZXOAFFD-UHFFFAOYSA-N 0.000 description 4
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical class OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 4
- 229910002012 Aerosil® Inorganic materials 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 4
- 239000004342 Benzoyl peroxide Substances 0.000 description 4
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 4
- ZWBGKXMWYNNSRH-UHFFFAOYSA-N C(CCC)C1=CC=C(C=C1)[B] Chemical compound C(CCC)C1=CC=C(C=C1)[B] ZWBGKXMWYNNSRH-UHFFFAOYSA-N 0.000 description 4
- VEQIGVGWYNVQHM-UHFFFAOYSA-N C(CCC)C=1C=C(C=CC=1)[B] Chemical compound C(CCC)C=1C=C(C=CC=1)[B] VEQIGVGWYNVQHM-UHFFFAOYSA-N 0.000 description 4
- HWPDIBGCECPDNL-UHFFFAOYSA-N C(CCC)OC1=CC=C(C=C1)[B] Chemical compound C(CCC)OC1=CC=C(C=C1)[B] HWPDIBGCECPDNL-UHFFFAOYSA-N 0.000 description 4
- UFUGBQFBWUBVSP-UHFFFAOYSA-N C(CCCCCCC)OC=1C=C(C=CC1)[B] Chemical compound C(CCCCCCC)OC=1C=C(C=CC1)[B] UFUGBQFBWUBVSP-UHFFFAOYSA-N 0.000 description 4
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 4
- PQBAWAQIRZIWIV-UHFFFAOYSA-N N-methylpyridinium Chemical class C[N+]1=CC=CC=C1 PQBAWAQIRZIWIV-UHFFFAOYSA-N 0.000 description 4
- LCKNZCHKSVSFHH-UHFFFAOYSA-N [3,5-bis(1,1,1,3,3,3-hexafluoro-2-methoxypropan-2-yl)phenyl]boron Chemical compound [B]C1=CC(C(OC)(C(F)(F)F)C(F)(F)F)=CC(C(OC)(C(F)(F)F)C(F)(F)F)=C1 LCKNZCHKSVSFHH-UHFFFAOYSA-N 0.000 description 4
- 230000001070 adhesive effect Effects 0.000 description 4
- 229910052782 aluminium Inorganic materials 0.000 description 4
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 4
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 4
- 235000019400 benzoyl peroxide Nutrition 0.000 description 4
- 239000005388 borosilicate glass Substances 0.000 description 4
- 239000008119 colloidal silica Substances 0.000 description 4
- 238000013329 compounding Methods 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- 239000000551 dentifrice Substances 0.000 description 4
- VFHVQBAGLAREND-UHFFFAOYSA-N diphenylphosphoryl-(2,4,6-trimethylphenyl)methanone Chemical compound CC1=CC(C)=CC(C)=C1C(=O)P(=O)(C=1C=CC=CC=1)C1=CC=CC=C1 VFHVQBAGLAREND-UHFFFAOYSA-N 0.000 description 4
- 239000002612 dispersion medium Substances 0.000 description 4
- GHLKSLMMWAKNBM-UHFFFAOYSA-N dodecane-1,12-diol Chemical compound OCCCCCCCCCCCCO GHLKSLMMWAKNBM-UHFFFAOYSA-N 0.000 description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 4
- 159000000003 magnesium salts Chemical class 0.000 description 4
- 150000001451 organic peroxides Chemical class 0.000 description 4
- 239000010453 quartz Substances 0.000 description 4
- 239000011347 resin Substances 0.000 description 4
- 229920005989 resin Polymers 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 238000003860 storage Methods 0.000 description 4
- DZLFLBLQUQXARW-UHFFFAOYSA-N tetrabutylammonium Chemical class CCCC[N+](CCCC)(CCCC)CCCC DZLFLBLQUQXARW-UHFFFAOYSA-N 0.000 description 4
- CBXCPBUEXACCNR-UHFFFAOYSA-N tetraethylammonium Chemical class CC[N+](CC)(CC)CC CBXCPBUEXACCNR-UHFFFAOYSA-N 0.000 description 4
- 235000010215 titanium dioxide Nutrition 0.000 description 4
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 4
- 150000003682 vanadium compounds Chemical class 0.000 description 4
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 3
- VNQXSTWCDUXYEZ-UHFFFAOYSA-N 1,7,7-trimethylbicyclo[2.2.1]heptane-2,3-dione Chemical compound C1CC2(C)C(=O)C(=O)C1C2(C)C VNQXSTWCDUXYEZ-UHFFFAOYSA-N 0.000 description 3
- KCWWCWMGJOWTMY-UHFFFAOYSA-N 1-benzyl-5-phenyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1C(C=2C=CC=CC=2)C(=O)NC(=O)N1CC1=CC=CC=C1 KCWWCWMGJOWTMY-UHFFFAOYSA-N 0.000 description 3
- REACWASHYHDPSQ-UHFFFAOYSA-N 1-butylpyridin-1-ium Chemical class CCCC[N+]1=CC=CC=C1 REACWASHYHDPSQ-UHFFFAOYSA-N 0.000 description 3
- CBMDQVNFHVUOIB-UHFFFAOYSA-N 1-ethylquinolin-1-ium Chemical class C1=CC=C2[N+](CC)=CC=CC2=C1 CBMDQVNFHVUOIB-UHFFFAOYSA-N 0.000 description 3
- TXBCBTDQIULDIA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)CO TXBCBTDQIULDIA-UHFFFAOYSA-N 0.000 description 3
- GAYWTJPBIQKDRC-UHFFFAOYSA-N 8-trimethoxysilyloctyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCCCCOC(=O)C(C)=C GAYWTJPBIQKDRC-UHFFFAOYSA-N 0.000 description 3
- NLZUEZXRPGMBCV-UHFFFAOYSA-N Butylhydroxytoluene Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NLZUEZXRPGMBCV-UHFFFAOYSA-N 0.000 description 3
- JTPHDBMUAITJHP-UHFFFAOYSA-N C(CCC)OC=1C=C(C=CC1)[B] Chemical compound C(CCC)OC=1C=C(C=CC1)[B] JTPHDBMUAITJHP-UHFFFAOYSA-N 0.000 description 3
- XNCFCJNBAMKOQT-UHFFFAOYSA-N C(CCCCCCC)OC1=CC=C(C=C1)[B] Chemical compound C(CCCCCCC)OC1=CC=C(C=C1)[B] XNCFCJNBAMKOQT-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical class OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 description 3
- 125000003342 alkenyl group Chemical group 0.000 description 3
- 125000000304 alkynyl group Chemical group 0.000 description 3
- UMGDCJDMYOKAJW-UHFFFAOYSA-N aminothiocarboxamide Natural products NC(N)=S UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 3
- 150000007656 barbituric acids Chemical class 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- MTZQAGJQAFMTAQ-UHFFFAOYSA-N benzoic acid ethyl ester Natural products CCOC(=O)C1=CC=CC=C1 MTZQAGJQAFMTAQ-UHFFFAOYSA-N 0.000 description 3
- 229930006711 bornane-2,3-dione Natural products 0.000 description 3
- 239000000805 composite resin Substances 0.000 description 3
- 150000004775 coumarins Chemical class 0.000 description 3
- 239000007822 coupling agent Substances 0.000 description 3
- 239000012933 diacyl peroxide Substances 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- UAOMVDZJSHZZME-UHFFFAOYSA-N diisopropylamine Chemical compound CC(C)NC(C)C UAOMVDZJSHZZME-UHFFFAOYSA-N 0.000 description 3
- 239000005350 fused silica glass Substances 0.000 description 3
- UCNNJGDEJXIUCC-UHFFFAOYSA-L hydroxy(oxo)iron;iron Chemical compound [Fe].O[Fe]=O.O[Fe]=O UCNNJGDEJXIUCC-UHFFFAOYSA-L 0.000 description 3
- 239000003112 inhibitor Substances 0.000 description 3
- 239000001023 inorganic pigment Substances 0.000 description 3
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 3
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 3
- 229910003002 lithium salt Inorganic materials 0.000 description 3
- 159000000002 lithium salts Chemical class 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 3
- OTLDLKLSNZMTTA-UHFFFAOYSA-N octahydro-1h-4,7-methanoindene-1,5-diyldimethanol Chemical compound C1C2C3C(CO)CCC3C1C(CO)C2 OTLDLKLSNZMTTA-UHFFFAOYSA-N 0.000 description 3
- 239000012860 organic pigment Substances 0.000 description 3
- 125000004430 oxygen atom Chemical group O* 0.000 description 3
- 150000002978 peroxides Chemical class 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 239000011163 secondary particle Substances 0.000 description 3
- 239000005361 soda-lime glass Substances 0.000 description 3
- GCLGEJMYGQKIIW-UHFFFAOYSA-H sodium hexametaphosphate Chemical compound [Na]OP1(=O)OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])O1 GCLGEJMYGQKIIW-UHFFFAOYSA-H 0.000 description 3
- 235000019982 sodium hexametaphosphate Nutrition 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 239000001577 tetrasodium phosphonato phosphate Substances 0.000 description 3
- 150000003606 tin compounds Chemical class 0.000 description 3
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 3
- 239000006097 ultraviolet radiation absorber Substances 0.000 description 3
- OTSKHUNLOQPIGN-UHFFFAOYSA-N 1,3,5-trimethyl-1,3-diazinane-2,4,6-trione Chemical compound CC1C(=O)N(C)C(=O)N(C)C1=O OTSKHUNLOQPIGN-UHFFFAOYSA-N 0.000 description 2
- DLKQHBOKULLWDQ-UHFFFAOYSA-N 1-bromonaphthalene Chemical compound C1=CC=C2C(Br)=CC=CC2=C1 DLKQHBOKULLWDQ-UHFFFAOYSA-N 0.000 description 2
- LRZPQLZONWIQOJ-UHFFFAOYSA-N 10-(2-methylprop-2-enoyloxy)decyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCOC(=O)C(C)=C LRZPQLZONWIQOJ-UHFFFAOYSA-N 0.000 description 2
- BXBOUPUNKULVKB-UHFFFAOYSA-N 10-trimethoxysilyldecyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCCCCCCOC(=O)C(C)=C BXBOUPUNKULVKB-UHFFFAOYSA-N 0.000 description 2
- RGOVHAJZSSODRY-UHFFFAOYSA-N 11-trimethoxysilylundecyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCCCCCCCOC(=O)C(C)=C RGOVHAJZSSODRY-UHFFFAOYSA-N 0.000 description 2
- ZUZAETTVAMCNTO-UHFFFAOYSA-N 2,3-dibutylbenzene-1,4-diol Chemical compound CCCCC1=C(O)C=CC(O)=C1CCCC ZUZAETTVAMCNTO-UHFFFAOYSA-N 0.000 description 2
- DXUMYHZTYVPBEZ-UHFFFAOYSA-N 2,4,6-tris(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 DXUMYHZTYVPBEZ-UHFFFAOYSA-N 0.000 description 2
- WJKHYAJKIXYSHS-UHFFFAOYSA-N 2-(4-chlorophenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(Cl)=CC=C1C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 WJKHYAJKIXYSHS-UHFFFAOYSA-N 0.000 description 2
- JUVSRZCUMWZBFK-UHFFFAOYSA-N 2-[n-(2-hydroxyethyl)-4-methylanilino]ethanol Chemical compound CC1=CC=C(N(CCO)CCO)C=C1 JUVSRZCUMWZBFK-UHFFFAOYSA-N 0.000 description 2
- JZXVADSBLRIAIB-UHFFFAOYSA-N 2-pyrrolidin-2-ylethanol Chemical compound OCCC1CCCN1 JZXVADSBLRIAIB-UHFFFAOYSA-N 0.000 description 2
- KBQVDAIIQCXKPI-UHFFFAOYSA-N 3-trimethoxysilylpropyl prop-2-enoate Chemical compound CO[Si](OC)(OC)CCCOC(=O)C=C KBQVDAIIQCXKPI-UHFFFAOYSA-N 0.000 description 2
- HTKIZIQFMHVTRJ-UHFFFAOYSA-N 5-butyl-1,3-diazinane-2,4,6-trione Chemical compound CCCCC1C(=O)NC(=O)NC1=O HTKIZIQFMHVTRJ-UHFFFAOYSA-N 0.000 description 2
- RCNRIKGRQSBQQB-UHFFFAOYSA-N 7-(dibutylamino)-3-[7-(dibutylamino)-2-oxochromene-3-carbonyl]chromen-2-one Chemical compound C1=C(N(CCCC)CCCC)C=C2OC(=O)C(C(=O)C3=CC4=CC=C(C=C4OC3=O)N(CCCC)CCCC)=CC2=C1 RCNRIKGRQSBQQB-UHFFFAOYSA-N 0.000 description 2
- SANIRTQDABNCHF-UHFFFAOYSA-N 7-(diethylamino)-3-[7-(diethylamino)-2-oxochromene-3-carbonyl]chromen-2-one Chemical compound C1=C(N(CC)CC)C=C2OC(=O)C(C(=O)C3=CC4=CC=C(C=C4OC3=O)N(CC)CC)=CC2=C1 SANIRTQDABNCHF-UHFFFAOYSA-N 0.000 description 2
- AXQFBPOHPYUZQC-UHFFFAOYSA-N 9-trimethoxysilylnonyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCCCCCOC(=O)C(C)=C AXQFBPOHPYUZQC-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 238000004438 BET method Methods 0.000 description 2
- OTFAQGCBYAXPCR-UHFFFAOYSA-N CC1=C(C(=O)COP(C2=CC=CC=C2)=O)C(=CC(=C1)C)C Chemical compound CC1=C(C(=O)COP(C2=CC=CC=C2)=O)C(=CC(=C1)C)C OTFAQGCBYAXPCR-UHFFFAOYSA-N 0.000 description 2
- 239000005749 Copper compound Substances 0.000 description 2
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 2
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 2
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 2
- 241000282320 Panthera leo Species 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Natural products NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 229910052769 Ytterbium Inorganic materials 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 2
- XQBCVRSTVUHIGH-UHFFFAOYSA-L [dodecanoyloxy(dioctyl)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCCCCCC)(CCCCCCCC)OC(=O)CCCCCCCCCCC XQBCVRSTVUHIGH-UHFFFAOYSA-L 0.000 description 2
- GUCYFKSBFREPBC-UHFFFAOYSA-N [phenyl-(2,4,6-trimethylbenzoyl)phosphoryl]-(2,4,6-trimethylphenyl)methanone Chemical compound CC1=CC(C)=CC(C)=C1C(=O)P(=O)(C=1C=CC=CC=1)C(=O)C1=C(C)C=C(C)C=C1C GUCYFKSBFREPBC-UHFFFAOYSA-N 0.000 description 2
- 238000005299 abrasion Methods 0.000 description 2
- 125000000218 acetic acid group Chemical group C(C)(=O)* 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 150000004982 aromatic amines Chemical class 0.000 description 2
- 238000005452 bending Methods 0.000 description 2
- 150000003935 benzaldehydes Chemical class 0.000 description 2
- JEHKKBHWRAXMCH-UHFFFAOYSA-N benzenesulfinic acid Chemical compound O[S@@](=O)C1=CC=CC=C1 JEHKKBHWRAXMCH-UHFFFAOYSA-N 0.000 description 2
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N benzo-alpha-pyrone Natural products C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 150000001880 copper compounds Chemical class 0.000 description 2
- 235000001671 coumarin Nutrition 0.000 description 2
- 239000006059 cover glass Substances 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- FOTKYAAJKYLFFN-UHFFFAOYSA-N decane-1,10-diol Chemical compound OCCCCCCCCCCO FOTKYAAJKYLFFN-UHFFFAOYSA-N 0.000 description 2
- 239000005548 dental material Substances 0.000 description 2
- 210000004268 dentin Anatomy 0.000 description 2
- ZQMIGQNCOMNODD-UHFFFAOYSA-N diacetyl peroxide Chemical compound CC(=O)OOC(C)=O ZQMIGQNCOMNODD-UHFFFAOYSA-N 0.000 description 2
- JQVDAXLFBXTEQA-UHFFFAOYSA-N dibutylamine Chemical compound CCCCNCCCC JQVDAXLFBXTEQA-UHFFFAOYSA-N 0.000 description 2
- GNTDGMZSJNCJKK-UHFFFAOYSA-N divanadium pentaoxide Chemical compound O=[V](=O)O[V](=O)=O GNTDGMZSJNCJKK-UHFFFAOYSA-N 0.000 description 2
- 229920001971 elastomer Polymers 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 238000010304 firing Methods 0.000 description 2
- RMBPEFMHABBEKP-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2C3=C[CH]C=CC3=CC2=C1 RMBPEFMHABBEKP-UHFFFAOYSA-N 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 150000002366 halogen compounds Chemical class 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 230000001678 irradiating effect Effects 0.000 description 2
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 2
- 238000004898 kneading Methods 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- CRVGTESFCCXCTH-UHFFFAOYSA-N methyl diethanolamine Chemical compound OCCN(C)CCO CRVGTESFCCXCTH-UHFFFAOYSA-N 0.000 description 2
- OSWPMRLSEDHDFF-UHFFFAOYSA-N methyl salicylate Chemical compound COC(=O)C1=CC=CC=C1O OSWPMRLSEDHDFF-UHFFFAOYSA-N 0.000 description 2
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N o-biphenylenemethane Natural products C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 description 2
- 125000003232 p-nitrobenzoyl group Chemical group [N+](=O)([O-])C1=CC=C(C(=O)*)C=C1 0.000 description 2
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 2
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 2
- 239000004926 polymethyl methacrylate Substances 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- YPFDHNVEDLHUCE-UHFFFAOYSA-N propane-1,3-diol Chemical compound OCCCO YPFDHNVEDLHUCE-UHFFFAOYSA-N 0.000 description 2
- 239000001054 red pigment Substances 0.000 description 2
- 229920003987 resole Polymers 0.000 description 2
- 239000005060 rubber Substances 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 2
- KFZUDNZQQCWGKF-UHFFFAOYSA-M sodium;4-methylbenzenesulfinate Chemical compound [Na+].CC1=CC=C(S([O-])=O)C=C1 KFZUDNZQQCWGKF-UHFFFAOYSA-M 0.000 description 2
- 238000001179 sorption measurement Methods 0.000 description 2
- 238000001694 spray drying Methods 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- UBXAKNTVXQMEAG-UHFFFAOYSA-L strontium sulfate Chemical compound [Sr+2].[O-]S([O-])(=O)=O UBXAKNTVXQMEAG-UHFFFAOYSA-L 0.000 description 2
- 229920003048 styrene butadiene rubber Polymers 0.000 description 2
- BUUPQKDIAURBJP-UHFFFAOYSA-N sulfinic acid Chemical compound OS=O BUUPQKDIAURBJP-UHFFFAOYSA-N 0.000 description 2
- 125000004434 sulfur atom Chemical group 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 150000003510 tertiary aliphatic amines Chemical class 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 238000002834 transmittance Methods 0.000 description 2
- 150000003918 triazines Chemical class 0.000 description 2
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 2
- XASAPYQVQBKMIN-UHFFFAOYSA-K ytterbium(iii) fluoride Chemical compound F[Yb](F)F XASAPYQVQBKMIN-UHFFFAOYSA-K 0.000 description 2
- 229910052726 zirconium Inorganic materials 0.000 description 2
- DTGKSKDOIYIVQL-WEDXCCLWSA-N (+)-borneol Chemical group C1C[C@@]2(C)[C@@H](O)C[C@@H]1C2(C)C DTGKSKDOIYIVQL-WEDXCCLWSA-N 0.000 description 1
- WRXCBRHBHGNNQA-UHFFFAOYSA-N (2,4-dichlorobenzoyl) 2,4-dichlorobenzenecarboperoxoate Chemical compound ClC1=CC(Cl)=CC=C1C(=O)OOC(=O)C1=CC=C(Cl)C=C1Cl WRXCBRHBHGNNQA-UHFFFAOYSA-N 0.000 description 1
- JLLAWIKMLAQZCZ-UHFFFAOYSA-N (2,6-dichlorophenyl)-diphenylphosphorylmethanone Chemical compound ClC1=CC=CC(Cl)=C1C(=O)P(=O)(C=1C=CC=CC=1)C1=CC=CC=C1 JLLAWIKMLAQZCZ-UHFFFAOYSA-N 0.000 description 1
- SUEDCWGEKSLKOM-UHFFFAOYSA-N (2,6-dimethoxyphenyl)-diphenylphosphorylmethanone Chemical compound COC1=CC=CC(OC)=C1C(=O)P(=O)(C=1C=CC=CC=1)C1=CC=CC=C1 SUEDCWGEKSLKOM-UHFFFAOYSA-N 0.000 description 1
- FVQMJJQUGGVLEP-UHFFFAOYSA-N (2-methylpropan-2-yl)oxy 2-ethylhexaneperoxoate Chemical compound CCCCC(CC)C(=O)OOOC(C)(C)C FVQMJJQUGGVLEP-UHFFFAOYSA-N 0.000 description 1
- QEQBMZQFDDDTPN-UHFFFAOYSA-N (2-methylpropan-2-yl)oxy benzenecarboperoxoate Chemical compound CC(C)(C)OOOC(=O)C1=CC=CC=C1 QEQBMZQFDDDTPN-UHFFFAOYSA-N 0.000 description 1
- NAWXUBYGYWOOIX-SFHVURJKSA-N (2s)-2-[[4-[2-(2,4-diaminoquinazolin-6-yl)ethyl]benzoyl]amino]-4-methylidenepentanedioic acid Chemical compound C1=CC2=NC(N)=NC(N)=C2C=C1CCC1=CC=C(C(=O)N[C@@H](CC(=C)C(O)=O)C(O)=O)C=C1 NAWXUBYGYWOOIX-SFHVURJKSA-N 0.000 description 1
- HRYZIYWIOWUKIP-UHFFFAOYSA-N (3-phenoxyphenyl)methyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC1=CC=CC(OC=2C=CC=CC=2)=C1 HRYZIYWIOWUKIP-UHFFFAOYSA-N 0.000 description 1
- NOBYOEQUFMGXBP-UHFFFAOYSA-N (4-tert-butylcyclohexyl) (4-tert-butylcyclohexyl)oxycarbonyloxy carbonate Chemical compound C1CC(C(C)(C)C)CCC1OC(=O)OOC(=O)OC1CCC(C(C)(C)C)CC1 NOBYOEQUFMGXBP-UHFFFAOYSA-N 0.000 description 1
- QBZIEGUIYWGBMY-FUZXWUMZSA-N (5Z)-5-hydroxyimino-6-oxonaphthalene-2-sulfonic acid iron Chemical compound [Fe].O\N=C1/C(=O)C=Cc2cc(ccc12)S(O)(=O)=O.O\N=C1/C(=O)C=Cc2cc(ccc12)S(O)(=O)=O.O\N=C1/C(=O)C=Cc2cc(ccc12)S(O)(=O)=O QBZIEGUIYWGBMY-FUZXWUMZSA-N 0.000 description 1
- BLKRGXCGFRXRNQ-SNAWJCMRSA-N (z)-3-carbonoperoxoyl-4,4-dimethylpent-2-enoic acid Chemical compound OC(=O)/C=C(C(C)(C)C)\C(=O)OO BLKRGXCGFRXRNQ-SNAWJCMRSA-N 0.000 description 1
- POILWHVDKZOXJZ-ARJAWSKDSA-M (z)-4-oxopent-2-en-2-olate Chemical compound C\C([O-])=C\C(C)=O POILWHVDKZOXJZ-ARJAWSKDSA-M 0.000 description 1
- WBYWAXJHAXSJNI-VOTSOKGWSA-M .beta-Phenylacrylic acid Natural products [O-]C(=O)\C=C\C1=CC=CC=C1 WBYWAXJHAXSJNI-VOTSOKGWSA-M 0.000 description 1
- TVHNLHDFHIHBRL-UHFFFAOYSA-N 1,1,3,3-tetracyclohexylthiourea Chemical compound C1CCCCC1N(C1CCCCC1)C(=S)N(C1CCCCC1)C1CCCCC1 TVHNLHDFHIHBRL-UHFFFAOYSA-N 0.000 description 1
- ZSPQVOFATJEJMT-UHFFFAOYSA-N 1,1,3,3-tetraethylthiourea Chemical compound CCN(CC)C(=S)N(CC)CC ZSPQVOFATJEJMT-UHFFFAOYSA-N 0.000 description 1
- MZRATIPJHWLMQH-UHFFFAOYSA-N 1,1,3-tricyclohexylthiourea Chemical compound C1CCCCC1N(C1CCCCC1)C(=S)NC1CCCCC1 MZRATIPJHWLMQH-UHFFFAOYSA-N 0.000 description 1
- HXMRAWVFMYZQMG-UHFFFAOYSA-N 1,1,3-triethylthiourea Chemical compound CCNC(=S)N(CC)CC HXMRAWVFMYZQMG-UHFFFAOYSA-N 0.000 description 1
- JAEZSIYNWDWMMN-UHFFFAOYSA-N 1,1,3-trimethylthiourea Chemical compound CNC(=S)N(C)C JAEZSIYNWDWMMN-UHFFFAOYSA-N 0.000 description 1
- VDUTWHBYCATQBI-UHFFFAOYSA-N 1,1,3-tripropylthiourea Chemical compound CCCNC(=S)N(CCC)CCC VDUTWHBYCATQBI-UHFFFAOYSA-N 0.000 description 1
- NALFRYPTRXKZPN-UHFFFAOYSA-N 1,1-bis(tert-butylperoxy)-3,3,5-trimethylcyclohexane Chemical compound CC1CC(C)(C)CC(OOC(C)(C)C)(OOC(C)(C)C)C1 NALFRYPTRXKZPN-UHFFFAOYSA-N 0.000 description 1
- HSLFISVKRDQEBY-UHFFFAOYSA-N 1,1-bis(tert-butylperoxy)cyclohexane Chemical compound CC(C)(C)OOC1(OOC(C)(C)C)CCCCC1 HSLFISVKRDQEBY-UHFFFAOYSA-N 0.000 description 1
- BEQKKZICTDFVMG-UHFFFAOYSA-N 1,2,3,4,6-pentaoxepane-5,7-dione Chemical compound O=C1OOOOC(=O)O1 BEQKKZICTDFVMG-UHFFFAOYSA-N 0.000 description 1
- KAJICSGLHKRDLN-UHFFFAOYSA-N 1,3-dicyclohexylthiourea Chemical compound C1CCCCC1NC(=S)NC1CCCCC1 KAJICSGLHKRDLN-UHFFFAOYSA-N 0.000 description 1
- VVSASNKOFCZVES-UHFFFAOYSA-N 1,3-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound CN1C(=O)CC(=O)N(C)C1=O VVSASNKOFCZVES-UHFFFAOYSA-N 0.000 description 1
- NFQAQKVFBFWVKQ-UHFFFAOYSA-N 1,3-dimethyl-5-(2-methylpropyl)-1,3-diazinane-2,4,6-trione Chemical compound CC(C)CC1C(=O)N(C)C(=O)N(C)C1=O NFQAQKVFBFWVKQ-UHFFFAOYSA-N 0.000 description 1
- UGFMJBRXTRUGJC-UHFFFAOYSA-N 1,3-dimethyl-5-phenyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1N(C)C(=O)N(C)C(=O)C1C1=CC=CC=C1 UGFMJBRXTRUGJC-UHFFFAOYSA-N 0.000 description 1
- FBQJKKPQBMSWEP-UHFFFAOYSA-N 1,3-diphenyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1CC(=O)N(C=2C=CC=CC=2)C(=O)N1C1=CC=CC=C1 FBQJKKPQBMSWEP-UHFFFAOYSA-N 0.000 description 1
- RXFDRTBMEIJQRH-UHFFFAOYSA-N 1,5-diethyl-1,3-diazinane-2,4,6-trione Chemical class CCC1C(=O)NC(=O)N(CC)C1=O RXFDRTBMEIJQRH-UHFFFAOYSA-N 0.000 description 1
- DABBHRQKJNIVFT-UHFFFAOYSA-N 1,5-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound CC1C(=O)NC(=O)N(C)C1=O DABBHRQKJNIVFT-UHFFFAOYSA-N 0.000 description 1
- UICXTANXZJJIBC-UHFFFAOYSA-N 1-(1-hydroperoxycyclohexyl)peroxycyclohexan-1-ol Chemical compound C1CCCCC1(O)OOC1(OO)CCCCC1 UICXTANXZJJIBC-UHFFFAOYSA-N 0.000 description 1
- 125000004972 1-butynyl group Chemical group [H]C([H])([H])C([H])([H])C#C* 0.000 description 1
- MSNOVYXXOZYRDG-UHFFFAOYSA-N 1-cyclohexyl-5-hexyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1C(CCCCCC)C(=O)NC(=O)N1C1CCCCC1 MSNOVYXXOZYRDG-UHFFFAOYSA-N 0.000 description 1
- NOMGRDIADBNCNU-UHFFFAOYSA-N 1-cyclohexyl-5-methyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1C(C)C(=O)NC(=O)N1C1CCCCC1 NOMGRDIADBNCNU-UHFFFAOYSA-N 0.000 description 1
- ADMXJQOJLRCZDT-UHFFFAOYSA-N 1-cyclohexyl-5-octyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1C(CCCCCCCC)C(=O)NC(=O)N1C1CCCCC1 ADMXJQOJLRCZDT-UHFFFAOYSA-N 0.000 description 1
- GSNNOBIKEKJNPW-UHFFFAOYSA-N 1-ethyl-5-(2-methylpropyl)-1,3-diazinane-2,4,6-trione Chemical compound CCN1C(=O)NC(=O)C(CC(C)C)C1=O GSNNOBIKEKJNPW-UHFFFAOYSA-N 0.000 description 1
- QPXXXBVJBVHTTJ-UHFFFAOYSA-N 1-ethyl-5-methyl-1,3-diazinane-2,4,6-trione Chemical compound CCN1C(=O)NC(=O)C(C)C1=O QPXXXBVJBVHTTJ-UHFFFAOYSA-N 0.000 description 1
- BMVXCPBXGZKUPN-UHFFFAOYSA-N 1-hexanamine Chemical compound CCCCCCN BMVXCPBXGZKUPN-UHFFFAOYSA-N 0.000 description 1
- XLPJNCYCZORXHG-UHFFFAOYSA-N 1-morpholin-4-ylprop-2-en-1-one Chemical compound C=CC(=O)N1CCOCC1 XLPJNCYCZORXHG-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- NKVCQMYWYHDOOF-UHFFFAOYSA-N 1-phenoxyethane-1,2-diol Chemical compound OCC(O)OC1=CC=CC=C1 NKVCQMYWYHDOOF-UHFFFAOYSA-N 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- 125000000530 1-propynyl group Chemical group [H]C([H])([H])C#C* 0.000 description 1
- GVSRNCLIDZDBPF-UHFFFAOYSA-N 11-(dichloromethylsilyl)undecyl 2-methylprop-2-enoate Chemical compound C(C(=C)C)(=O)OCCCCCCCCCCC[SiH2]C(Cl)Cl GVSRNCLIDZDBPF-UHFFFAOYSA-N 0.000 description 1
- GTTJFCFYQHWRHO-UHFFFAOYSA-N 11-trichlorosilylundecyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCC[Si](Cl)(Cl)Cl GTTJFCFYQHWRHO-UHFFFAOYSA-N 0.000 description 1
- CADOLDOQCJTEBR-UHFFFAOYSA-N 11-trimethoxysilylundecyl prop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCCCCCCCOC(=O)C=C CADOLDOQCJTEBR-UHFFFAOYSA-N 0.000 description 1
- HYQASEVIBPSPMK-UHFFFAOYSA-N 12-(2-methylprop-2-enoyloxy)dodecyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCCCCCCCCCOC(=O)C(C)=C HYQASEVIBPSPMK-UHFFFAOYSA-N 0.000 description 1
- OLZKHNBQPLVBGV-UHFFFAOYSA-N 13-trimethoxysilyltridecyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCCCCCCCCCOC(=O)C(C)=C OLZKHNBQPLVBGV-UHFFFAOYSA-N 0.000 description 1
- GLCVQJJSGABYQF-UHFFFAOYSA-M 16-pyridin-1-ium-1-ylhexadecyl prop-2-enoate chloride Chemical compound [Cl-].C(C=C)(=O)OCCCCCCCCCCCCCCCC[N+]1=CC=CC=C1 GLCVQJJSGABYQF-UHFFFAOYSA-M 0.000 description 1
- GKLHILWMQAPCGH-UHFFFAOYSA-M 16-pyridin-1-ium-1-ylhexadecyl prop-2-enoate;bromide Chemical compound [Br-].C=CC(=O)OCCCCCCCCCCCCCCCC[N+]1=CC=CC=C1 GKLHILWMQAPCGH-UHFFFAOYSA-M 0.000 description 1
- JPOUDZAPLMMUES-UHFFFAOYSA-N 2,2-bis(tert-butylperoxy)octane Chemical compound CCCCCCC(C)(OOC(C)(C)C)OOC(C)(C)C JPOUDZAPLMMUES-UHFFFAOYSA-N 0.000 description 1
- XCBBNTFYSLADTO-UHFFFAOYSA-N 2,3-Octanedione Chemical compound CCCCCC(=O)C(C)=O XCBBNTFYSLADTO-UHFFFAOYSA-N 0.000 description 1
- BVSXRCDUVXEDNL-UHFFFAOYSA-N 2,4,6-tri(propan-2-yl)benzenesulfinic acid Chemical compound CC(C)C1=CC(C(C)C)=C(S(O)=O)C(C(C)C)=C1 BVSXRCDUVXEDNL-UHFFFAOYSA-N 0.000 description 1
- TZSXNOJOZIODBO-UHFFFAOYSA-N 2,4,6-triethylbenzenesulfinic acid Chemical compound CCC1=CC(CC)=C(S(O)=O)C(CC)=C1 TZSXNOJOZIODBO-UHFFFAOYSA-N 0.000 description 1
- CGRMGOGJWHPCFJ-UHFFFAOYSA-N 2,4,6-trimethylbenzenesulfinic acid Chemical compound CC1=CC(C)=C(S(O)=O)C(C)=C1 CGRMGOGJWHPCFJ-UHFFFAOYSA-N 0.000 description 1
- URJAUSYMVIZTHC-UHFFFAOYSA-N 2,4,6-tris(tribromomethyl)-1,3,5-triazine Chemical compound BrC(Br)(Br)C1=NC(C(Br)(Br)Br)=NC(C(Br)(Br)Br)=N1 URJAUSYMVIZTHC-UHFFFAOYSA-N 0.000 description 1
- JJPKWNXRUDKBQT-UHFFFAOYSA-N 2,4-bis(trichloromethyl)-6-[2-(3,4,5-trimethoxyphenyl)ethenyl]-1,3,5-triazine Chemical compound COC1=C(OC)C(OC)=CC(C=CC=2N=C(N=C(N=2)C(Cl)(Cl)Cl)C(Cl)(Cl)Cl)=C1 JJPKWNXRUDKBQT-UHFFFAOYSA-N 0.000 description 1
- DMWVYCCGCQPJEA-UHFFFAOYSA-N 2,5-bis(tert-butylperoxy)-2,5-dimethylhexane Chemical compound CC(C)(C)OOC(C)(C)CCC(C)(C)OOC(C)(C)C DMWVYCCGCQPJEA-UHFFFAOYSA-N 0.000 description 1
- JGBAASVQPMTVHO-UHFFFAOYSA-N 2,5-dihydroperoxy-2,5-dimethylhexane Chemical compound OOC(C)(C)CCC(C)(C)OO JGBAASVQPMTVHO-UHFFFAOYSA-N 0.000 description 1
- DKCPKDPYUFEZCP-UHFFFAOYSA-N 2,6-di-tert-butylphenol Chemical compound CC(C)(C)C1=CC=CC(C(C)(C)C)=C1O DKCPKDPYUFEZCP-UHFFFAOYSA-N 0.000 description 1
- GUKYYIQMUCVKHI-UHFFFAOYSA-N 2-(2,4-dichlorophenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC1=CC(Cl)=CC=C1C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 GUKYYIQMUCVKHI-UHFFFAOYSA-N 0.000 description 1
- DQMOHZLFVGYNAN-UHFFFAOYSA-N 2-(2-phenylethenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=CC=2C=CC=CC=2)=N1 DQMOHZLFVGYNAN-UHFFFAOYSA-N 0.000 description 1
- XMNIXWIUMCBBBL-UHFFFAOYSA-N 2-(2-phenylpropan-2-ylperoxy)propan-2-ylbenzene Chemical compound C=1C=CC=CC=1C(C)(C)OOC(C)(C)C1=CC=CC=C1 XMNIXWIUMCBBBL-UHFFFAOYSA-N 0.000 description 1
- NVGHIMFCFZAKIF-UHFFFAOYSA-N 2-(3,4-dimethylanilino)ethanol Chemical compound CC1=CC=C(NCCO)C=C1C NVGHIMFCFZAKIF-UHFFFAOYSA-N 0.000 description 1
- JAHNSTQSQJOJLO-UHFFFAOYSA-N 2-(3-fluorophenyl)-1h-imidazole Chemical compound FC1=CC=CC(C=2NC=CN=2)=C1 JAHNSTQSQJOJLO-UHFFFAOYSA-N 0.000 description 1
- UKTRUOJPSJPVOZ-UHFFFAOYSA-N 2-(4-bromophenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=2C=CC(Br)=CC=2)=N1 UKTRUOJPSJPVOZ-UHFFFAOYSA-N 0.000 description 1
- MPNIGZBDAMWHSX-UHFFFAOYSA-N 2-(4-methylphenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(C)=CC=C1C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 MPNIGZBDAMWHSX-UHFFFAOYSA-N 0.000 description 1
- OZIUQIJHFBCZBX-UHFFFAOYSA-N 2-(4-methylsulfanylphenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(SC)=CC=C1C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 OZIUQIJHFBCZBX-UHFFFAOYSA-N 0.000 description 1
- IZBVPPRIBFECNZ-UHFFFAOYSA-N 2-(4-phenylphenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=2C=CC(=CC=2)C=2C=CC=CC=2)=N1 IZBVPPRIBFECNZ-UHFFFAOYSA-N 0.000 description 1
- IYAZLDLPUNDVAG-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4-(2,4,4-trimethylpentan-2-yl)phenol Chemical compound CC(C)(C)CC(C)(C)C1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 IYAZLDLPUNDVAG-UHFFFAOYSA-N 0.000 description 1
- NAHKJPXTQGGPSZ-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4-ethylphenol Chemical compound CCC1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 NAHKJPXTQGGPSZ-UHFFFAOYSA-N 0.000 description 1
- XYPMCBIMCCNXQE-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4-propylphenol Chemical compound CCCC1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 XYPMCBIMCCNXQE-UHFFFAOYSA-N 0.000 description 1
- FJGQBLRYBUAASW-UHFFFAOYSA-N 2-(benzotriazol-2-yl)phenol Chemical compound OC1=CC=CC=C1N1N=C2C=CC=CC2=N1 FJGQBLRYBUAASW-UHFFFAOYSA-N 0.000 description 1
- DGPBVJWCIDNDPN-UHFFFAOYSA-N 2-(dimethylamino)benzaldehyde Chemical compound CN(C)C1=CC=CC=C1C=O DGPBVJWCIDNDPN-UHFFFAOYSA-N 0.000 description 1
- WPTIDPUGZXGDEV-UHFFFAOYSA-N 2-[2-(2-methoxyphenyl)ethenyl]-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound COC1=CC=CC=C1C=CC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 WPTIDPUGZXGDEV-UHFFFAOYSA-N 0.000 description 1
- ZJRNXDIVAGHETA-UHFFFAOYSA-N 2-[2-(3,4-dimethoxyphenyl)ethenyl]-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=C(OC)C(OC)=CC=C1C=CC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 ZJRNXDIVAGHETA-UHFFFAOYSA-N 0.000 description 1
- WCXDCXLNIUKQHG-UHFFFAOYSA-N 2-[2-(4-butoxyphenyl)ethenyl]-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(OCCCC)=CC=C1C=CC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 WCXDCXLNIUKQHG-UHFFFAOYSA-N 0.000 description 1
- MCNPOZMLKGDJGP-UHFFFAOYSA-N 2-[2-(4-methoxyphenyl)ethenyl]-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(OC)=CC=C1C=CC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 MCNPOZMLKGDJGP-UHFFFAOYSA-N 0.000 description 1
- VWRRLTREYNMJPY-UHFFFAOYSA-N 2-[2-[[4,6-bis(trichloromethyl)-1,3,5-triazin-2-yl]oxy]ethyl-(2-hydroxyethyl)amino]ethanol Chemical compound OCCN(CCO)CCOC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 VWRRLTREYNMJPY-UHFFFAOYSA-N 0.000 description 1
- HLDMOJRATBJDBV-UHFFFAOYSA-N 2-[3,5-ditert-butyl-n-(2-hydroxyethyl)anilino]ethanol Chemical compound CC(C)(C)C1=CC(N(CCO)CCO)=CC(C(C)(C)C)=C1 HLDMOJRATBJDBV-UHFFFAOYSA-N 0.000 description 1
- IFMADWRJVDIYMF-UHFFFAOYSA-N 2-[4-ethyl-n-(2-hydroxyethyl)anilino]ethanol Chemical compound CCC1=CC=C(N(CCO)CCO)C=C1 IFMADWRJVDIYMF-UHFFFAOYSA-N 0.000 description 1
- IEXYKOLCMBHVMG-UHFFFAOYSA-N 2-[4-tert-butyl-n-(2-hydroxyethyl)anilino]ethanol Chemical compound CC(C)(C)C1=CC=C(N(CCO)CCO)C=C1 IEXYKOLCMBHVMG-UHFFFAOYSA-N 0.000 description 1
- DEWPRFSWNWKLHF-UHFFFAOYSA-N 2-[bis(2-hydroxyethyl)amino]ethanol;2-methylprop-2-enoic acid Chemical compound CC(=C)C(O)=O.CC(=C)C(O)=O.OCCN(CCO)CCO DEWPRFSWNWKLHF-UHFFFAOYSA-N 0.000 description 1
- YCHXXZUEOABJIB-UHFFFAOYSA-N 2-[bis(2-hydroxyethyl)amino]ethanol;2-methylprop-2-enoic acid Chemical compound CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.OCCN(CCO)CCO YCHXXZUEOABJIB-UHFFFAOYSA-N 0.000 description 1
- NKFNBVMJTSYZDV-UHFFFAOYSA-N 2-[dodecyl(2-hydroxyethyl)amino]ethanol Chemical compound CCCCCCCCCCCCN(CCO)CCO NKFNBVMJTSYZDV-UHFFFAOYSA-N 0.000 description 1
- BHICZSRCJVGOGG-UHFFFAOYSA-N 2-[ethyl-[2-(2-methylprop-2-enoyloxy)ethyl]amino]ethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCN(CC)CCOC(=O)C(C)=C BHICZSRCJVGOGG-UHFFFAOYSA-N 0.000 description 1
- IMKJXRBWMSKNQE-UHFFFAOYSA-N 2-[n-(2-hydroxyethyl)-3,5-di(propan-2-yl)anilino]ethanol Chemical compound CC(C)C1=CC(C(C)C)=CC(N(CCO)CCO)=C1 IMKJXRBWMSKNQE-UHFFFAOYSA-N 0.000 description 1
- JWQRYFYKHVZIDQ-UHFFFAOYSA-N 2-[n-(2-hydroxyethyl)-3,5-dimethylanilino]ethanol Chemical compound CC1=CC(C)=CC(N(CCO)CCO)=C1 JWQRYFYKHVZIDQ-UHFFFAOYSA-N 0.000 description 1
- OMBRORBBHNMRDM-UHFFFAOYSA-N 2-[n-(2-hydroxyethyl)-4-propan-2-ylanilino]ethanol Chemical compound CC(C)C1=CC=C(N(CCO)CCO)C=C1 OMBRORBBHNMRDM-UHFFFAOYSA-N 0.000 description 1
- UAMPJVJAALGMNG-UHFFFAOYSA-N 2-acetylbenzo[f]chromen-3-one Chemical compound C1=CC=C2C(C=C(C(O3)=O)C(=O)C)=C3C=CC2=C1 UAMPJVJAALGMNG-UHFFFAOYSA-N 0.000 description 1
- JPPGTVYDWHSNLJ-UHFFFAOYSA-N 2-benzoylbenzo[f]chromen-3-one Chemical compound C=1C(C2=CC=CC=C2C=C2)=C2OC(=O)C=1C(=O)C1=CC=CC=C1 JPPGTVYDWHSNLJ-UHFFFAOYSA-N 0.000 description 1
- NMBQBIACXMPEQB-UHFFFAOYSA-N 2-butoxyethyl benzoate Chemical compound CCCCOCCOC(=O)C1=CC=CC=C1 NMBQBIACXMPEQB-UHFFFAOYSA-N 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- CVICJYZKEWRDKT-UHFFFAOYSA-N 2-carbamoyloxyethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCOC(N)=O.CC(=C)C(=O)OCCOC(N)=O CVICJYZKEWRDKT-UHFFFAOYSA-N 0.000 description 1
- IJVRPNIWWODHHA-UHFFFAOYSA-N 2-cyanoprop-2-enoic acid Chemical compound OC(=O)C(=C)C#N IJVRPNIWWODHHA-UHFFFAOYSA-N 0.000 description 1
- ASEUXRQULQEGGL-UHFFFAOYSA-N 2-decyl-2-prop-2-enoyloxypropanedioic acid Chemical compound CCCCCCCCCCC(C(O)=O)(C(O)=O)OC(=O)C=C ASEUXRQULQEGGL-UHFFFAOYSA-N 0.000 description 1
- VGZZAZYCLRYTNQ-UHFFFAOYSA-N 2-ethoxyethoxycarbonyloxy 2-ethoxyethyl carbonate Chemical compound CCOCCOC(=O)OOC(=O)OCCOCC VGZZAZYCLRYTNQ-UHFFFAOYSA-N 0.000 description 1
- ISTSJNBVMFHDRK-UHFFFAOYSA-N 2-ethyl-2-(2,2,4-trimethylpentylperoxy)hexanoic acid Chemical compound CCCCC(CC)(C(=O)O)OOCC(C)(C)CC(C)C ISTSJNBVMFHDRK-UHFFFAOYSA-N 0.000 description 1
- ZACVGCNKGYYQHA-UHFFFAOYSA-N 2-ethylhexoxycarbonyloxy 2-ethylhexyl carbonate Chemical compound CCCCC(CC)COC(=O)OOC(=O)OCC(CC)CCCC ZACVGCNKGYYQHA-UHFFFAOYSA-N 0.000 description 1
- DILXLMRYFWFBGR-UHFFFAOYSA-N 2-formylbenzene-1,4-disulfonic acid Chemical compound OS(=O)(=O)C1=CC=C(S(O)(=O)=O)C(C=O)=C1 DILXLMRYFWFBGR-UHFFFAOYSA-N 0.000 description 1
- MIRQGKQPLPBZQM-UHFFFAOYSA-N 2-hydroperoxy-2,4,4-trimethylpentane Chemical compound CC(C)(C)CC(C)(C)OO MIRQGKQPLPBZQM-UHFFFAOYSA-N 0.000 description 1
- WFUGQJXVXHBTEM-UHFFFAOYSA-N 2-hydroperoxy-2-(2-hydroperoxybutan-2-ylperoxy)butane Chemical compound CCC(C)(OO)OOC(C)(CC)OO WFUGQJXVXHBTEM-UHFFFAOYSA-N 0.000 description 1
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 1
- FLFWJIBUZQARMD-UHFFFAOYSA-N 2-mercapto-1,3-benzoxazole Chemical compound C1=CC=C2OC(S)=NC2=C1 FLFWJIBUZQARMD-UHFFFAOYSA-N 0.000 description 1
- GOTIJEQQGSMAIN-UHFFFAOYSA-N 2-methyl-4,6-bis(tribromomethyl)-1,3,5-triazine Chemical compound CC1=NC(C(Br)(Br)Br)=NC(C(Br)(Br)Br)=N1 GOTIJEQQGSMAIN-UHFFFAOYSA-N 0.000 description 1
- LETDRANQSOEVCX-UHFFFAOYSA-N 2-methyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound CC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 LETDRANQSOEVCX-UHFFFAOYSA-N 0.000 description 1
- 125000003229 2-methylhexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- WZXYIWMZCIFNRB-UHFFFAOYSA-N 2-methylprop-2-enoate;tris(2-hydroxyethyl)azanium Chemical compound CC(=C)C(O)=O.OCCN(CCO)CCO WZXYIWMZCIFNRB-UHFFFAOYSA-N 0.000 description 1
- RPBWMJBZQXCSFW-UHFFFAOYSA-N 2-methylpropanoyl 2-methylpropaneperoxoate Chemical compound CC(C)C(=O)OOC(=O)C(C)C RPBWMJBZQXCSFW-UHFFFAOYSA-N 0.000 description 1
- ACMLKANOGIVEPB-UHFFFAOYSA-N 2-oxo-2H-chromene-3-carboxylic acid Chemical compound C1=CC=C2OC(=O)C(C(=O)O)=CC2=C1 ACMLKANOGIVEPB-UHFFFAOYSA-N 0.000 description 1
- ZXTHWIZHGLNEPG-UHFFFAOYSA-N 2-phenyl-4,5-dihydro-1,3-oxazole Chemical compound O1CCN=C1C1=CC=CC=C1 ZXTHWIZHGLNEPG-UHFFFAOYSA-N 0.000 description 1
- HAZQZUFYRLFOLC-UHFFFAOYSA-N 2-phenyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=2C=CC=CC=2)=N1 HAZQZUFYRLFOLC-UHFFFAOYSA-N 0.000 description 1
- NUIZZJWNNGJSGL-UHFFFAOYSA-N 2-phenylpropan-2-yl 2,2-dimethyloctaneperoxoate Chemical compound CCCCCCC(C)(C)C(=O)OOC(C)(C)c1ccccc1 NUIZZJWNNGJSGL-UHFFFAOYSA-N 0.000 description 1
- UDXXYUDJOHIIDZ-UHFFFAOYSA-N 2-phosphonooxyethyl prop-2-enoate Chemical compound OP(O)(=O)OCCOC(=O)C=C UDXXYUDJOHIIDZ-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- DOSGQNSHFPTAOA-UHFFFAOYSA-N 2-propyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound CCCC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 DOSGQNSHFPTAOA-UHFFFAOYSA-N 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- VEBFFMASUFIZKN-UHFFFAOYSA-N 2-tert-butylperoxy-3,3,5-trimethylhexanoic acid Chemical compound CC(C)CC(C)(C)C(C(O)=O)OOC(C)(C)C VEBFFMASUFIZKN-UHFFFAOYSA-N 0.000 description 1
- BIISIZOQPWZPPS-UHFFFAOYSA-N 2-tert-butylperoxypropan-2-ylbenzene Chemical compound CC(C)(C)OOC(C)(C)C1=CC=CC=C1 BIISIZOQPWZPPS-UHFFFAOYSA-N 0.000 description 1
- RDCTZTAAYLXPDJ-UHFFFAOYSA-N 2-trimethoxysilylethyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCOC(=O)C(C)=C RDCTZTAAYLXPDJ-UHFFFAOYSA-N 0.000 description 1
- KFGFVPMRLOQXNB-UHFFFAOYSA-N 3,5,5-trimethylhexanoyl 3,5,5-trimethylhexaneperoxoate Chemical compound CC(C)(C)CC(C)CC(=O)OOC(=O)CC(C)CC(C)(C)C KFGFVPMRLOQXNB-UHFFFAOYSA-N 0.000 description 1
- FIJRQSPKMCQFJU-UHFFFAOYSA-N 3,5-ditert-butyl-n,n-dimethylaniline Chemical compound CN(C)C1=CC(C(C)(C)C)=CC(C(C)(C)C)=C1 FIJRQSPKMCQFJU-UHFFFAOYSA-N 0.000 description 1
- FRIBMENBGGCKPD-UHFFFAOYSA-N 3-(2,3-dimethoxyphenyl)prop-2-enal Chemical compound COC1=CC=CC(C=CC=O)=C1OC FRIBMENBGGCKPD-UHFFFAOYSA-N 0.000 description 1
- LXCJXYYYOPFEDH-UHFFFAOYSA-N 3-(2-oxochromene-3-carbonyl)chromen-2-one Chemical compound C1=CC=C2OC(=O)C(C(C=3C(OC4=CC=CC=C4C=3)=O)=O)=CC2=C1 LXCJXYYYOPFEDH-UHFFFAOYSA-N 0.000 description 1
- YSAUSJGMEDJGDI-UHFFFAOYSA-N 3-(2-prop-2-enoyloxyethoxycarbonyl)phthalic acid Chemical compound OC(=O)C1=CC=CC(C(=O)OCCOC(=O)C=C)=C1C(O)=O YSAUSJGMEDJGDI-UHFFFAOYSA-N 0.000 description 1
- AZESABVBPAGIPJ-UHFFFAOYSA-N 3-(4-methoxybenzoyl)chromen-2-one Chemical compound C1=CC(OC)=CC=C1C(=O)C1=CC2=CC=CC=C2OC1=O AZESABVBPAGIPJ-UHFFFAOYSA-N 0.000 description 1
- IDQPLXGUEBBMQN-UHFFFAOYSA-N 3-(thiophene-3-carbonyl)chromen-2-one Chemical compound C=1C2=CC=CC=C2OC(=O)C=1C(=O)C=1C=CSC=1 IDQPLXGUEBBMQN-UHFFFAOYSA-N 0.000 description 1
- MYKMRINTJPZLQB-UHFFFAOYSA-N 3-acetyl-7-(dimethylamino)chromen-2-one Chemical compound C1=C(C(C)=O)C(=O)OC2=CC(N(C)C)=CC=C21 MYKMRINTJPZLQB-UHFFFAOYSA-N 0.000 description 1
- POLTYHPZKRUHOQ-UHFFFAOYSA-N 3-amino-4-(2-butoxyethyl)chromen-2-one Chemical compound C1=CC=CC2=C1OC(=O)C(N)=C2CCOCCCC POLTYHPZKRUHOQ-UHFFFAOYSA-N 0.000 description 1
- ITJVTQLTIZXPEQ-UHFFFAOYSA-N 3-benzoyl-5,7-dimethoxychromen-2-one Chemical compound O=C1OC2=CC(OC)=CC(OC)=C2C=C1C(=O)C1=CC=CC=C1 ITJVTQLTIZXPEQ-UHFFFAOYSA-N 0.000 description 1
- OXSWLSMWLZZECK-UHFFFAOYSA-N 3-benzoyl-6-bromochromen-2-one Chemical compound C=1C2=CC(Br)=CC=C2OC(=O)C=1C(=O)C1=CC=CC=C1 OXSWLSMWLZZECK-UHFFFAOYSA-N 0.000 description 1
- IIUIPOQYIBJROA-UHFFFAOYSA-N 3-benzoyl-6-methoxychromen-2-one Chemical compound C=1C2=CC(OC)=CC=C2OC(=O)C=1C(=O)C1=CC=CC=C1 IIUIPOQYIBJROA-UHFFFAOYSA-N 0.000 description 1
- FZFUZACPHSNNEC-UHFFFAOYSA-N 3-benzoyl-6-nitrochromen-2-one Chemical compound C=1C2=CC([N+](=O)[O-])=CC=C2OC(=O)C=1C(=O)C1=CC=CC=C1 FZFUZACPHSNNEC-UHFFFAOYSA-N 0.000 description 1
- CPVJWBWVJUAOMV-UHFFFAOYSA-N 3-benzoyl-7-(diethylamino)chromen-2-one Chemical compound O=C1OC2=CC(N(CC)CC)=CC=C2C=C1C(=O)C1=CC=CC=C1 CPVJWBWVJUAOMV-UHFFFAOYSA-N 0.000 description 1
- DWDONSBFIIUPQS-UHFFFAOYSA-N 3-benzoyl-7-(dimethylamino)chromen-2-one Chemical compound O=C1OC2=CC(N(C)C)=CC=C2C=C1C(=O)C1=CC=CC=C1 DWDONSBFIIUPQS-UHFFFAOYSA-N 0.000 description 1
- HYORIVUCOQKMOC-UHFFFAOYSA-N 3-benzoyl-7-methoxychromen-2-one Chemical compound O=C1OC2=CC(OC)=CC=C2C=C1C(=O)C1=CC=CC=C1 HYORIVUCOQKMOC-UHFFFAOYSA-N 0.000 description 1
- XMULXKFGESYAEG-UHFFFAOYSA-N 3-benzoyl-8-methoxychromen-2-one Chemical compound O=C1OC=2C(OC)=CC=CC=2C=C1C(=O)C1=CC=CC=C1 XMULXKFGESYAEG-UHFFFAOYSA-N 0.000 description 1
- LPBMPRKJYKSRLL-UHFFFAOYSA-N 3-benzoylchromen-2-one Chemical compound C=1C2=CC=CC=C2OC(=O)C=1C(=O)C1=CC=CC=C1 LPBMPRKJYKSRLL-UHFFFAOYSA-N 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- QOXOZONBQWIKDA-UHFFFAOYSA-N 3-hydroxypropyl Chemical group [CH2]CCO QOXOZONBQWIKDA-UHFFFAOYSA-N 0.000 description 1
- UUEWCQRISZBELL-UHFFFAOYSA-N 3-trimethoxysilylpropane-1-thiol Chemical compound CO[Si](OC)(OC)CCCS UUEWCQRISZBELL-UHFFFAOYSA-N 0.000 description 1
- JRHHJNMASOIRDS-UHFFFAOYSA-N 4-ethoxybenzaldehyde Chemical compound CCOC1=CC=C(C=O)C=C1 JRHHJNMASOIRDS-UHFFFAOYSA-N 0.000 description 1
- KLQNCSLBKKYPET-UHFFFAOYSA-N 4-ethyl-n,n-dimethylaniline Chemical compound CCC1=CC=C(N(C)C)C=C1 KLQNCSLBKKYPET-UHFFFAOYSA-N 0.000 description 1
- SXIFAEWFOJETOA-UHFFFAOYSA-N 4-hydroxy-butyl Chemical group [CH2]CCCO SXIFAEWFOJETOA-UHFFFAOYSA-N 0.000 description 1
- VGVHNLRUAMRIEW-UHFFFAOYSA-N 4-methylcyclohexan-1-one Chemical compound CC1CCC(=O)CC1 VGVHNLRUAMRIEW-UHFFFAOYSA-N 0.000 description 1
- KQLZAABIBODGSQ-UHFFFAOYSA-N 4-propoxybutyl prop-2-enoate Chemical compound CCCOCCCCOC(=O)C=C KQLZAABIBODGSQ-UHFFFAOYSA-N 0.000 description 1
- SJDILFZCXQHCRB-UHFFFAOYSA-N 4-tert-butyl-n,n-dimethylaniline Chemical compound CN(C)C1=CC=C(C(C)(C)C)C=C1 SJDILFZCXQHCRB-UHFFFAOYSA-N 0.000 description 1
- ODSZSDRDEZFANY-UHFFFAOYSA-N 5-butyl-1,3-diethyl-1,3-diazinane-2,4,6-trione Chemical compound CCCCC1C(=O)N(CC)C(=O)N(CC)C1=O ODSZSDRDEZFANY-UHFFFAOYSA-N 0.000 description 1
- PAAOBODHLHVLMN-UHFFFAOYSA-N 5-butyl-1,3-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound CCCCC1C(=O)N(C)C(=O)N(C)C1=O PAAOBODHLHVLMN-UHFFFAOYSA-N 0.000 description 1
- DVEQCIBLXRSYPH-UHFFFAOYSA-N 5-butyl-1-cyclohexylbarbituric acid Chemical compound O=C1C(CCCC)C(=O)NC(=O)N1C1CCCCC1 DVEQCIBLXRSYPH-UHFFFAOYSA-N 0.000 description 1
- IYHIFXGFKVJNBB-UHFFFAOYSA-N 5-chloro-2-[(2-hydroxynaphthalen-1-yl)diazenyl]-4-methylbenzenesulfonic acid Chemical compound C1=C(Cl)C(C)=CC(N=NC=2C3=CC=CC=C3C=CC=2O)=C1S(O)(=O)=O IYHIFXGFKVJNBB-UHFFFAOYSA-N 0.000 description 1
- PZBQVZFITSVHAW-UHFFFAOYSA-N 5-chloro-2h-benzotriazole Chemical compound C1=C(Cl)C=CC2=NNN=C21 PZBQVZFITSVHAW-UHFFFAOYSA-N 0.000 description 1
- RATSVJLVTJDKSI-UHFFFAOYSA-N 5-cyclohexyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1NC(=O)NC(=O)C1C1CCCCC1 RATSVJLVTJDKSI-UHFFFAOYSA-N 0.000 description 1
- ANBKBYQJFJUEBR-UHFFFAOYSA-N 5-cyclohexyl-1,3-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1N(C)C(=O)N(C)C(=O)C1C1CCCCC1 ANBKBYQJFJUEBR-UHFFFAOYSA-N 0.000 description 1
- WTUYGYIHVPSHQF-UHFFFAOYSA-N 5-cyclopentyl-1,3-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1N(C)C(=O)N(C)C(=O)C1C1CCCC1 WTUYGYIHVPSHQF-UHFFFAOYSA-N 0.000 description 1
- FMTLDVACNZDTQL-UHFFFAOYSA-N 5-ethyl-1,3-diazinane-2,4,6-trione Chemical compound CCC1C(=O)NC(=O)NC1=O FMTLDVACNZDTQL-UHFFFAOYSA-N 0.000 description 1
- QBCQXNDNMOHQIU-UHFFFAOYSA-N 5-ethyl-1,3-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound CCC1C(=O)N(C)C(=O)N(C)C1=O QBCQXNDNMOHQIU-UHFFFAOYSA-N 0.000 description 1
- GOMAEJQBTWAPAN-UHFFFAOYSA-N 5-methylbarbituric acid Chemical compound CC1C(=O)NC(=O)NC1=O GOMAEJQBTWAPAN-UHFFFAOYSA-N 0.000 description 1
- GFPIGNBQTXNNAG-UHFFFAOYSA-N 5-propan-2-yl-1,3-diazinane-2,4,6-trione Chemical compound CC(C)C1C(=O)NC(=O)NC1=O GFPIGNBQTXNNAG-UHFFFAOYSA-N 0.000 description 1
- ZWSXCQXSBNMGFC-UHFFFAOYSA-N 5-propyl-1,3-diazinane-2,4,6-trione Chemical compound CCCC1C(=O)NC(=O)NC1=O ZWSXCQXSBNMGFC-UHFFFAOYSA-N 0.000 description 1
- DKRGZWMTKHYZGY-UHFFFAOYSA-N 5-trimethoxysilylpentyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCOC(=O)C(C)=C DKRGZWMTKHYZGY-UHFFFAOYSA-N 0.000 description 1
- WIYVVIUBKNTNKG-UHFFFAOYSA-N 6,7-dimethoxy-3,4-dihydronaphthalene-2-carboxylic acid Chemical compound C1CC(C(O)=O)=CC2=C1C=C(OC)C(OC)=C2 WIYVVIUBKNTNKG-UHFFFAOYSA-N 0.000 description 1
- YBHSRYKMANGQQS-UHFFFAOYSA-N 6-methoxy-3-(6-methoxy-2-oxochromene-3-carbonyl)chromen-2-one Chemical compound COC1=CC=C2OC(=O)C(C(=O)C=3C(=O)OC4=CC=C(C=C4C=3)OC)=CC2=C1 YBHSRYKMANGQQS-UHFFFAOYSA-N 0.000 description 1
- GWKBSZQPLYRAAH-UHFFFAOYSA-N 6-trimethoxysilylhexyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCCCCOC(=O)C(C)=C GWKBSZQPLYRAAH-UHFFFAOYSA-N 0.000 description 1
- RVCQMFUJJMHLHQ-UHFFFAOYSA-N 7-(dimethylamino)-3-[7-(dimethylamino)-2-oxochromene-3-carbonyl]chromen-2-one Chemical compound C1=C(N(C)C)C=C2OC(=O)C(C(=O)C3=CC4=CC=C(C=C4OC3=O)N(C)C)=CC2=C1 RVCQMFUJJMHLHQ-UHFFFAOYSA-N 0.000 description 1
- VEEGNDSSWAOLFN-UHFFFAOYSA-N 7-methoxy-2-oxochromene-3-carboxylic acid Chemical compound C1=C(C(O)=O)C(=O)OC2=CC(OC)=CC=C21 VEEGNDSSWAOLFN-UHFFFAOYSA-N 0.000 description 1
- ODRDTKMYQDXVGG-UHFFFAOYSA-N 8-methoxycoumarin Natural products C1=CC(=O)OC2=C1C=CC=C2OC ODRDTKMYQDXVGG-UHFFFAOYSA-N 0.000 description 1
- YYVYAPXYZVYDHN-UHFFFAOYSA-N 9,10-phenanthroquinone Chemical compound C1=CC=C2C(=O)C(=O)C3=CC=CC=C3C2=C1 YYVYAPXYZVYDHN-UHFFFAOYSA-N 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 229910000497 Amalgam Inorganic materials 0.000 description 1
- 229910001020 Au alloy Inorganic materials 0.000 description 1
- VVAVKBBTPWYADW-UHFFFAOYSA-L Biebrich scarlet Chemical compound [Na+].[Na+].OC1=CC=C2C=CC=CC2=C1N=NC(C(=C1)S([O-])(=O)=O)=CC=C1N=NC1=CC=C(S([O-])(=O)=O)C=C1 VVAVKBBTPWYADW-UHFFFAOYSA-L 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- SDKBUGQFAYJVAA-UHFFFAOYSA-N C(C=C)(=O)OCC(OC(=O)N)CC(CC(CCC(COC(C=C)=O)OC(=O)N)C)(C)C Chemical compound C(C=C)(=O)OCC(OC(=O)N)CC(CC(CCC(COC(C=C)=O)OC(=O)N)C)(C)C SDKBUGQFAYJVAA-UHFFFAOYSA-N 0.000 description 1
- NPIAYTKVIYZUQB-UHFFFAOYSA-N C(CC)C1=CC=C(C=C1)[PH2]=O Chemical compound C(CC)C1=CC=C(C=C1)[PH2]=O NPIAYTKVIYZUQB-UHFFFAOYSA-N 0.000 description 1
- YEROZEJIWWDMAJ-UHFFFAOYSA-O C=CC1=CC=C(CCCCC(NC(N2)=[S+]N)=NC2=S)C=C1 Chemical compound C=CC1=CC=C(CCCCC(NC(N2)=[S+]N)=NC2=S)C=C1 YEROZEJIWWDMAJ-UHFFFAOYSA-O 0.000 description 1
- VDVWJKNSZQPORG-UHFFFAOYSA-N CC1=C(C(=CC=C1)C)OP(O)=O Chemical compound CC1=C(C(=CC=C1)C)OP(O)=O VDVWJKNSZQPORG-UHFFFAOYSA-N 0.000 description 1
- BUBJBVLNMSTXBG-UHFFFAOYSA-N CC1=C(C(=O)P(C2=CC=CC=C2)=O)C(=CC(=C1)C)C.[Na] Chemical class CC1=C(C(=O)P(C2=CC=CC=C2)=O)C(=CC(=C1)C)C.[Na] BUBJBVLNMSTXBG-UHFFFAOYSA-N 0.000 description 1
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 1
- 241000282465 Canis Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- WBYWAXJHAXSJNI-SREVYHEPSA-N Cinnamic acid Chemical compound OC(=O)\C=C/C1=CC=CC=C1 WBYWAXJHAXSJNI-SREVYHEPSA-N 0.000 description 1
- QSJXEFYPDANLFS-UHFFFAOYSA-N Diacetyl Chemical group CC(=O)C(C)=O QSJXEFYPDANLFS-UHFFFAOYSA-N 0.000 description 1
- PQUCIEFHOVEZAU-UHFFFAOYSA-N Diammonium sulfite Chemical compound [NH4+].[NH4+].[O-]S([O-])=O PQUCIEFHOVEZAU-UHFFFAOYSA-N 0.000 description 1
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- GTAXGNCCEYZRII-UHFFFAOYSA-N Eperisone hydrochloride Chemical compound Cl.C1=CC(CC)=CC=C1C(=O)C(C)CN1CCCCC1 GTAXGNCCEYZRII-UHFFFAOYSA-N 0.000 description 1
- 239000004386 Erythritol Substances 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical group C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 1
- 241000692870 Inachis io Species 0.000 description 1
- YIVJZNGAASQVEM-UHFFFAOYSA-N Lauroyl peroxide Chemical compound CCCCCCCCCCCC(=O)OOC(=O)CCCCCCCCCCC YIVJZNGAASQVEM-UHFFFAOYSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- QPJVMBTYPHYUOC-UHFFFAOYSA-N Methyl benzoate Natural products COC(=O)C1=CC=CC=C1 QPJVMBTYPHYUOC-UHFFFAOYSA-N 0.000 description 1
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 1
- 101000954570 Mus musculus V-type proton ATPase 16 kDa proteolipid subunit c Proteins 0.000 description 1
- VLCDUOXHFNUCKK-UHFFFAOYSA-N N,N'-Dimethylthiourea Chemical compound CNC(=S)NC VLCDUOXHFNUCKK-UHFFFAOYSA-N 0.000 description 1
- FLVIGYVXZHLUHP-UHFFFAOYSA-N N,N'-diethylthiourea Chemical compound CCNC(=S)NCC FLVIGYVXZHLUHP-UHFFFAOYSA-N 0.000 description 1
- KQJQICVXLJTWQD-UHFFFAOYSA-N N-Methylthiourea Chemical compound CNC(N)=S KQJQICVXLJTWQD-UHFFFAOYSA-N 0.000 description 1
- AKNUHUCEWALCOI-UHFFFAOYSA-N N-ethyldiethanolamine Chemical compound OCCN(CC)CCO AKNUHUCEWALCOI-UHFFFAOYSA-N 0.000 description 1
- GMEHFXXZSWDEDB-UHFFFAOYSA-N N-ethylthiourea Chemical compound CCNC(N)=S GMEHFXXZSWDEDB-UHFFFAOYSA-N 0.000 description 1
- OPKOKAMJFNKNAS-UHFFFAOYSA-N N-methylethanolamine Chemical compound CNCCO OPKOKAMJFNKNAS-UHFFFAOYSA-N 0.000 description 1
- 229920000459 Nitrile rubber Polymers 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-L Phosphate ion(2-) Chemical compound OP([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-L 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical class C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- MNOILHPDHOHILI-UHFFFAOYSA-N Tetramethylthiourea Chemical compound CN(C)C(=S)N(C)C MNOILHPDHOHILI-UHFFFAOYSA-N 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- WGLPBDUCMAPZCE-UHFFFAOYSA-N Trioxochromium Chemical compound O=[Cr](=O)=O WGLPBDUCMAPZCE-UHFFFAOYSA-N 0.000 description 1
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical class C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 description 1
- 239000004234 Yellow 2G Substances 0.000 description 1
- 229910021536 Zeolite Inorganic materials 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 239000005083 Zinc sulfide Substances 0.000 description 1
- PRRGXULZOZTZDK-UHFFFAOYSA-N [(2,6-dichlorobenzoyl)-(2,5-dimethylphenyl)phosphoryl]-(2,6-dichlorophenyl)methanone Chemical compound CC1=CC=C(C)C(P(=O)(C(=O)C=2C(=CC=CC=2Cl)Cl)C(=O)C=2C(=CC=CC=2Cl)Cl)=C1 PRRGXULZOZTZDK-UHFFFAOYSA-N 0.000 description 1
- YNJCLWHSZGZEAS-UHFFFAOYSA-N [(2,6-dichlorobenzoyl)-naphthalen-1-ylphosphoryl]-(2,6-dichlorophenyl)methanone Chemical compound ClC1=CC=CC(Cl)=C1C(=O)P(=O)(C=1C2=CC=CC=C2C=CC=1)C(=O)C1=C(Cl)C=CC=C1Cl YNJCLWHSZGZEAS-UHFFFAOYSA-N 0.000 description 1
- DNRISHWSPBDRTH-UHFFFAOYSA-N [(2,6-dichlorobenzoyl)-phenylphosphoryl]-(2,6-dichlorophenyl)methanone Chemical compound ClC1=CC=CC(Cl)=C1C(=O)P(=O)(C=1C=CC=CC=1)C(=O)C1=C(Cl)C=CC=C1Cl DNRISHWSPBDRTH-UHFFFAOYSA-N 0.000 description 1
- ZTCAGYRXUCWHPV-UHFFFAOYSA-N [(2,6-dimethoxybenzoyl)-(2,5-dimethylphenyl)phosphoryl]-(2,6-dimethoxyphenyl)methanone Chemical compound COC1=CC=CC(OC)=C1C(=O)P(=O)(C=1C(=CC=C(C)C=1)C)C(=O)C1=C(OC)C=CC=C1OC ZTCAGYRXUCWHPV-UHFFFAOYSA-N 0.000 description 1
- QISAYNXDUCNISJ-UHFFFAOYSA-N [(2,6-dimethoxybenzoyl)-phenylphosphoryl]-(2,6-dimethoxyphenyl)methanone Chemical compound COC1=CC=CC(OC)=C1C(=O)P(=O)(C=1C=CC=CC=1)C(=O)C1=C(OC)C=CC=C1OC QISAYNXDUCNISJ-UHFFFAOYSA-N 0.000 description 1
- WJHVGPHBIZTOHI-UHFFFAOYSA-N [2-(3-butoxyphenyl)phenyl]-diphenylborane Chemical compound CCCCOC1=CC=CC(C=2C(=CC=CC=2)B(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 WJHVGPHBIZTOHI-UHFFFAOYSA-N 0.000 description 1
- GNOXDPXZDZKCTN-UHFFFAOYSA-N [2-(3-octoxyphenyl)phenyl]-diphenylborane Chemical compound CCCCCCCCOC1=CC=CC(C=2C(=CC=CC=2)B(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 GNOXDPXZDZKCTN-UHFFFAOYSA-N 0.000 description 1
- YKNPUKLMZUVJTE-UHFFFAOYSA-N [2-(4-butoxyphenyl)phenyl]-diphenylborane Chemical compound C1=CC(OCCCC)=CC=C1C1=CC=CC=C1B(C=1C=CC=CC=1)C1=CC=CC=C1 YKNPUKLMZUVJTE-UHFFFAOYSA-N 0.000 description 1
- YYIVTXYZCMNNJI-UHFFFAOYSA-N [2-(4-fluorophenyl)phenyl]-diphenylborane Chemical compound C1=CC(F)=CC=C1C1=CC=CC=C1B(C=1C=CC=CC=1)C1=CC=CC=C1 YYIVTXYZCMNNJI-UHFFFAOYSA-N 0.000 description 1
- ZCSZKJOLHQJJED-UHFFFAOYSA-N [2-(4-nitrophenyl)phenyl]-diphenylborane Chemical compound C1=CC([N+](=O)[O-])=CC=C1C1=CC=CC=C1B(C=1C=CC=CC=1)C1=CC=CC=C1 ZCSZKJOLHQJJED-UHFFFAOYSA-N 0.000 description 1
- XCHUKVDNAYDTQG-UHFFFAOYSA-N [2-(4-octoxyphenyl)phenyl]-diphenylborane Chemical compound C1=CC(OCCCCCCCC)=CC=C1C1=CC=CC=C1B(C=1C=CC=CC=1)C1=CC=CC=C1 XCHUKVDNAYDTQG-UHFFFAOYSA-N 0.000 description 1
- PKRLTUNIIDPMER-UHFFFAOYSA-N [2-[3,5-bis(trifluoromethyl)phenyl]phenyl]-diphenylborane Chemical compound FC(F)(F)C1=CC(C(F)(F)F)=CC(C=2C(=CC=CC=2)B(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 PKRLTUNIIDPMER-UHFFFAOYSA-N 0.000 description 1
- YPBNDGVRPOECEP-UHFFFAOYSA-N [2-hydroxy-3-[2-[2-hydroxy-3-(2-methylprop-2-enoyloxy)propoxy]ethoxy]propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(O)COCCOCC(O)COC(=O)C(C)=C YPBNDGVRPOECEP-UHFFFAOYSA-N 0.000 description 1
- VZTQQYMRXDUHDO-UHFFFAOYSA-N [2-hydroxy-3-[4-[2-[4-(2-hydroxy-3-prop-2-enoyloxypropoxy)phenyl]propan-2-yl]phenoxy]propyl] prop-2-enoate Chemical compound C=1C=C(OCC(O)COC(=O)C=C)C=CC=1C(C)(C)C1=CC=C(OCC(O)COC(=O)C=C)C=C1 VZTQQYMRXDUHDO-UHFFFAOYSA-N 0.000 description 1
- TYJKIEGXKNWFJY-UHFFFAOYSA-N [3-(7-acetyloxy-2-oxochromene-3-carbonyl)-2-oxochromen-7-yl] acetate Chemical compound C1=C(OC(C)=O)C=C2OC(=O)C(C(=O)C3=CC4=CC=C(C=C4OC3=O)OC(=O)C)=CC2=C1 TYJKIEGXKNWFJY-UHFFFAOYSA-N 0.000 description 1
- BEUGBYXJXMVRFO-UHFFFAOYSA-N [4-(dimethylamino)phenyl]-phenylmethanone Chemical compound C1=CC(N(C)C)=CC=C1C(=O)C1=CC=CC=C1 BEUGBYXJXMVRFO-UHFFFAOYSA-N 0.000 description 1
- NUYFQCRLMKAHTJ-UHFFFAOYSA-M [Br-].C(CCCCCCCCC)[N+]1=CC=CC=C1.C(C=C)(=O)OC(C=C)=O Chemical compound [Br-].C(CCCCCCCCC)[N+]1=CC=CC=C1.C(C=C)(=O)OC(C=C)=O NUYFQCRLMKAHTJ-UHFFFAOYSA-M 0.000 description 1
- GRWDNMIZBWODSA-UHFFFAOYSA-M [Cl-].C(CCCCCCCCC)[N+]1=CC=CC=C1.C(C=C)(=O)OC(C=C)=O Chemical compound [Cl-].C(CCCCCCCCC)[N+]1=CC=CC=C1.C(C=C)(=O)OC(C=C)=O GRWDNMIZBWODSA-UHFFFAOYSA-M 0.000 description 1
- XHCLAFWTIXFWPH-UHFFFAOYSA-N [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] XHCLAFWTIXFWPH-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- AFPRJLBZLPBTPZ-UHFFFAOYSA-N acenaphthoquinone Chemical compound C1=CC(C(C2=O)=O)=C3C2=CC=CC3=C1 AFPRJLBZLPBTPZ-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 1
- XECAHXYUAAWDEL-UHFFFAOYSA-N acrylonitrile butadiene styrene Chemical compound C=CC=C.C=CC#N.C=CC1=CC=CC=C1 XECAHXYUAAWDEL-UHFFFAOYSA-N 0.000 description 1
- 229920001893 acrylonitrile styrene Polymers 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- GHPGOEFPKIHBNM-UHFFFAOYSA-N antimony(3+);oxygen(2-) Chemical compound [O-2].[O-2].[O-2].[Sb+3].[Sb+3] GHPGOEFPKIHBNM-UHFFFAOYSA-N 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- IRERQBUNZFJFGC-UHFFFAOYSA-L azure blue Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[S-]S[S-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-] IRERQBUNZFJFGC-UHFFFAOYSA-L 0.000 description 1
- HNYOPLTXPVRDBG-UHFFFAOYSA-N barbituric acid Chemical compound O=C1CC(=O)NC(=O)N1 HNYOPLTXPVRDBG-UHFFFAOYSA-N 0.000 description 1
- HEQCHSSPWMWXBH-UHFFFAOYSA-L barium(2+) 1-[(2-carboxyphenyl)diazenyl]naphthalen-2-olate Chemical compound [Ba++].Oc1ccc2ccccc2c1N=Nc1ccccc1C([O-])=O.Oc1ccc2ccccc2c1N=Nc1ccccc1C([O-])=O HEQCHSSPWMWXBH-UHFFFAOYSA-L 0.000 description 1
- 239000010953 base metal Substances 0.000 description 1
- 239000000981 basic dye Substances 0.000 description 1
- UIJGNTRUPZPVNG-UHFFFAOYSA-N benzenecarbothioic s-acid Chemical compound SC(=O)C1=CC=CC=C1 UIJGNTRUPZPVNG-UHFFFAOYSA-N 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- JBIROUFYLSSYDX-UHFFFAOYSA-M benzododecinium chloride Chemical compound [Cl-].CCCCCCCCCCCC[N+](C)(C)CC1=CC=CC=C1 JBIROUFYLSSYDX-UHFFFAOYSA-M 0.000 description 1
- XSIFPSYPOVKYCO-UHFFFAOYSA-N benzoic acid butyl ester Natural products CCCCOC(=O)C1=CC=CC=C1 XSIFPSYPOVKYCO-UHFFFAOYSA-N 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- 150000001565 benzotriazoles Chemical class 0.000 description 1
- KXHPPCXNWTUNSB-UHFFFAOYSA-M benzyl(trimethyl)azanium;chloride Chemical compound [Cl-].C[N+](C)(C)CC1=CC=CC=C1 KXHPPCXNWTUNSB-UHFFFAOYSA-M 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 238000012662 bulk polymerization Methods 0.000 description 1
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 description 1
- 239000001273 butane Substances 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- BXIQXYOPGBXIEM-UHFFFAOYSA-N butyl 4,4-bis(tert-butylperoxy)pentanoate Chemical compound CCCCOC(=O)CCC(C)(OOC(C)(C)C)OOC(C)(C)C BXIQXYOPGBXIEM-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- SXPLZNMUBFBFIA-UHFFFAOYSA-N butyl(trimethoxy)silane Chemical compound CCCC[Si](OC)(OC)OC SXPLZNMUBFBFIA-UHFFFAOYSA-N 0.000 description 1
- 229910052793 cadmium Inorganic materials 0.000 description 1
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 description 1
- CJOBVZJTOIVNNF-UHFFFAOYSA-N cadmium sulfide Chemical compound [Cd]=S CJOBVZJTOIVNNF-UHFFFAOYSA-N 0.000 description 1
- WUKWITHWXAAZEY-UHFFFAOYSA-L calcium difluoride Chemical compound [F-].[F-].[Ca+2] WUKWITHWXAAZEY-UHFFFAOYSA-L 0.000 description 1
- 229910001634 calcium fluoride Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- GBAOBIBJACZTNA-UHFFFAOYSA-L calcium sulfite Chemical compound [Ca+2].[O-]S([O-])=O GBAOBIBJACZTNA-UHFFFAOYSA-L 0.000 description 1
- 235000010261 calcium sulphite Nutrition 0.000 description 1
- ZDWKLZYHSINFKA-UHFFFAOYSA-L calcium;2,4,6-tri(propan-2-yl)benzenesulfinate Chemical compound [Ca+2].CC(C)C1=CC(C(C)C)=C(S([O-])=O)C(C(C)C)=C1.CC(C)C1=CC(C(C)C)=C(S([O-])=O)C(C(C)C)=C1 ZDWKLZYHSINFKA-UHFFFAOYSA-L 0.000 description 1
- JDFASTJCXKFWFY-UHFFFAOYSA-L calcium;2,4,6-triethylbenzenesulfinate Chemical compound [Ca+2].CCC1=CC(CC)=C(S([O-])=O)C(CC)=C1.CCC1=CC(CC)=C(S([O-])=O)C(CC)=C1 JDFASTJCXKFWFY-UHFFFAOYSA-L 0.000 description 1
- MGKWLBCLSKZROU-UHFFFAOYSA-L calcium;2,4,6-trimethylbenzenesulfinate Chemical compound [Ca+2].CC1=CC(C)=C(S([O-])=O)C(C)=C1.CC1=CC(C)=C(S([O-])=O)C(C)=C1 MGKWLBCLSKZROU-UHFFFAOYSA-L 0.000 description 1
- JPLNMWITWCOINR-UHFFFAOYSA-L calcium;4-methylbenzenesulfinate Chemical compound [Ca+2].CC1=CC=C(S([O-])=O)C=C1.CC1=CC=C(S([O-])=O)C=C1 JPLNMWITWCOINR-UHFFFAOYSA-L 0.000 description 1
- CQTBCRGSMDMCKL-UHFFFAOYSA-L calcium;benzenesulfinate Chemical compound [Ca+2].[O-]S(=O)C1=CC=CC=C1.[O-]S(=O)C1=CC=CC=C1 CQTBCRGSMDMCKL-UHFFFAOYSA-L 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 150000001733 carboxylic acid esters Chemical class 0.000 description 1
- 125000002843 carboxylic acid group Chemical group 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- SXPWTBGAZSPLHA-UHFFFAOYSA-M cetalkonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+](C)(C)CC1=CC=CC=C1 SXPWTBGAZSPLHA-UHFFFAOYSA-M 0.000 description 1
- 229960000228 cetalkonium chloride Drugs 0.000 description 1
- 239000012986 chain transfer agent Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- PZTQVMXMKVTIRC-UHFFFAOYSA-L chembl2028348 Chemical compound [Ca+2].[O-]S(=O)(=O)C1=CC(C)=CC=C1N=NC1=C(O)C(C([O-])=O)=CC2=CC=CC=C12 PZTQVMXMKVTIRC-UHFFFAOYSA-L 0.000 description 1
- ZLFVRXUOSPRRKQ-UHFFFAOYSA-N chembl2138372 Chemical compound [O-][N+](=O)C1=CC(C)=CC=C1N=NC1=C(O)C=CC2=CC=CC=C12 ZLFVRXUOSPRRKQ-UHFFFAOYSA-N 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 229930002875 chlorophyll Natural products 0.000 description 1
- 235000019804 chlorophyll Nutrition 0.000 description 1
- ATNHDLDRLWWWCB-AENOIHSZSA-M chlorophyll a Chemical compound C1([C@@H](C(=O)OC)C(=O)C2=C3C)=C2N2C3=CC(C(CC)=C3C)=[N+]4C3=CC3=C(C=C)C(C)=C5N3[Mg-2]42[N+]2=C1[C@@H](CCC(=O)OC\C=C(/C)CCC[C@H](C)CCC[C@H](C)CCCC(C)C)[C@H](C)C2=C5 ATNHDLDRLWWWCB-AENOIHSZSA-M 0.000 description 1
- ZCDOYSPFYFSLEW-UHFFFAOYSA-N chromate(2-) Chemical compound [O-][Cr]([O-])(=O)=O ZCDOYSPFYFSLEW-UHFFFAOYSA-N 0.000 description 1
- 229910000423 chromium oxide Inorganic materials 0.000 description 1
- 235000013985 cinnamic acid Nutrition 0.000 description 1
- 229930016911 cinnamic acid Natural products 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 239000002734 clay mineral Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 239000013065 commercial product Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 1
- OPQARKPSCNTWTJ-UHFFFAOYSA-L copper(ii) acetate Chemical compound [Cu+2].CC([O-])=O.CC([O-])=O OPQARKPSCNTWTJ-UHFFFAOYSA-L 0.000 description 1
- SVOAENZIOKPANY-CVBJKYQLSA-L copper;(z)-octadec-9-enoate Chemical compound [Cu+2].CCCCCCCC\C=C/CCCCCCCC([O-])=O.CCCCCCCC\C=C/CCCCCCCC([O-])=O SVOAENZIOKPANY-CVBJKYQLSA-L 0.000 description 1
- QNZRVYCYEMYQMD-UHFFFAOYSA-N copper;pentane-2,4-dione Chemical compound [Cu].CC(=O)CC(C)=O QNZRVYCYEMYQMD-UHFFFAOYSA-N 0.000 description 1
- 229960000956 coumarin Drugs 0.000 description 1
- VBVAVBCYMYWNOU-UHFFFAOYSA-N coumarin 6 Chemical compound C1=CC=C2SC(C3=CC4=CC=C(C=C4OC3=O)N(CC)CC)=NC2=C1 VBVAVBCYMYWNOU-UHFFFAOYSA-N 0.000 description 1
- LDHQCZJRKDOVOX-NSCUHMNNSA-N crotonic acid Chemical compound C\C=C\C(O)=O LDHQCZJRKDOVOX-NSCUHMNNSA-N 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- ZXJXZNDDNMQXFV-UHFFFAOYSA-M crystal violet Chemical compound [Cl-].C1=CC(N(C)C)=CC=C1[C+](C=1C=CC(=CC=1)N(C)C)C1=CC=C(N(C)C)C=C1 ZXJXZNDDNMQXFV-UHFFFAOYSA-M 0.000 description 1
- SPTHWAJJMLCAQF-UHFFFAOYSA-M ctk4f8481 Chemical compound [O-]O.CC(C)C1=CC=CC=C1C(C)C SPTHWAJJMLCAQF-UHFFFAOYSA-M 0.000 description 1
- 210000003464 cuspid Anatomy 0.000 description 1
- 125000002993 cycloalkylene group Chemical group 0.000 description 1
- 125000001047 cyclobutenyl group Chemical group C1(=CCC1)* 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000298 cyclopropenyl group Chemical group [H]C1=C([H])C1([H])* 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- VTXVGVNLYGSIAR-UHFFFAOYSA-N decane-1-thiol Chemical compound CCCCCCCCCCS VTXVGVNLYGSIAR-UHFFFAOYSA-N 0.000 description 1
- KQAHMVLQCSALSX-UHFFFAOYSA-N decyl(trimethoxy)silane Chemical compound CCCCCCCCCC[Si](OC)(OC)OC KQAHMVLQCSALSX-UHFFFAOYSA-N 0.000 description 1
- GWUJPMKBSYJFCK-UHFFFAOYSA-N decyl-dimethoxy-methylsilane Chemical compound CCCCCCCCCC[Si](C)(OC)OC GWUJPMKBSYJFCK-UHFFFAOYSA-N 0.000 description 1
- 239000003479 dental cement Substances 0.000 description 1
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 description 1
- GUJOJGAPFQRJSV-UHFFFAOYSA-N dialuminum;dioxosilane;oxygen(2-);hydrate Chemical compound O.[O-2].[O-2].[O-2].[Al+3].[Al+3].O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O GUJOJGAPFQRJSV-UHFFFAOYSA-N 0.000 description 1
- 125000004915 dibutylamino group Chemical group C(CCC)N(CCCC)* 0.000 description 1
- XRWMGCFJVKDVMD-UHFFFAOYSA-M didodecyl(dimethyl)azanium;bromide Chemical compound [Br-].CCCCCCCCCCCC[N+](C)(C)CCCCCCCCCCCC XRWMGCFJVKDVMD-UHFFFAOYSA-M 0.000 description 1
- WLCFKPHMRNPAFZ-UHFFFAOYSA-M didodecyl(dimethyl)azanium;chloride Chemical compound [Cl-].CCCCCCCCCCCC[N+](C)(C)CCCCCCCCCCCC WLCFKPHMRNPAFZ-UHFFFAOYSA-M 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 229940043279 diisopropylamine Drugs 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- HGQSXVKHVMGQRG-UHFFFAOYSA-N dioctyltin Chemical compound CCCCCCCC[Sn]CCCCCCCC HGQSXVKHVMGQRG-UHFFFAOYSA-N 0.000 description 1
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 1
- MIBMTBXKARQXFP-UHFFFAOYSA-N diphenylphosphoryl-(2,3,5,6-tetramethylphenyl)methanone Chemical compound CC1=CC(C)=C(C)C(C(=O)P(=O)(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1C MIBMTBXKARQXFP-UHFFFAOYSA-N 0.000 description 1
- SUXCALIDMIIJCK-UHFFFAOYSA-L disodium;4-amino-3-[[4-[4-[(1-amino-4-sulfonatonaphthalen-2-yl)diazenyl]-3-methylphenyl]-2-methylphenyl]diazenyl]naphthalene-1-sulfonate Chemical compound [Na+].[Na+].C1=CC=CC2=C(N)C(N=NC3=CC=C(C=C3C)C=3C=C(C(=CC=3)N=NC=3C(=C4C=CC=CC4=C(C=3)S([O-])(=O)=O)N)C)=CC(S([O-])(=O)=O)=C21 SUXCALIDMIIJCK-UHFFFAOYSA-L 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- CIKJANOSDPPCAU-UHFFFAOYSA-N ditert-butyl cyclohexane-1,4-dicarboperoxoate Chemical compound CC(C)(C)OOC(=O)C1CCC(C(=O)OOC(C)(C)C)CC1 CIKJANOSDPPCAU-UHFFFAOYSA-N 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- MCPKSFINULVDNX-UHFFFAOYSA-N drometrizole Chemical compound CC1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 MCPKSFINULVDNX-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000000635 electron micrograph Methods 0.000 description 1
- 238000007720 emulsion polymerization reaction Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 1
- 229940009714 erythritol Drugs 0.000 description 1
- 235000019414 erythritol Nutrition 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- YGHIIBLLAKLTBT-UHFFFAOYSA-N ethyl 6-methoxy-2-oxochromene-3-carboxylate Chemical compound COC1=CC=C2OC(=O)C(C(=O)OCC)=CC2=C1 YGHIIBLLAKLTBT-UHFFFAOYSA-N 0.000 description 1
- ATXWFSDEXYKQST-UHFFFAOYSA-N ethyl 8-methoxy-2-oxochromene-3-carboxylate Chemical compound C1=CC(OC)=C2OC(=O)C(C(=O)OCC)=CC2=C1 ATXWFSDEXYKQST-UHFFFAOYSA-N 0.000 description 1
- 239000005038 ethylene vinyl acetate Substances 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- FPVGTPBMTFTMRT-NSKUCRDLSA-L fast yellow Chemical compound [Na+].[Na+].C1=C(S([O-])(=O)=O)C(N)=CC=C1\N=N\C1=CC=C(S([O-])(=O)=O)C=C1 FPVGTPBMTFTMRT-NSKUCRDLSA-L 0.000 description 1
- 235000019233 fast yellow AB Nutrition 0.000 description 1
- 238000010528 free radical solution polymerization reaction Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000003178 glass ionomer cement Substances 0.000 description 1
- 239000002241 glass-ceramic Substances 0.000 description 1
- 239000003353 gold alloy Substances 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 229910001385 heavy metal Inorganic materials 0.000 description 1
- LIIALPBMIOVAHH-UHFFFAOYSA-N herniarin Chemical compound C1=CC(=O)OC2=CC(OC)=CC=C21 LIIALPBMIOVAHH-UHFFFAOYSA-N 0.000 description 1
- JHGVLAHJJNKSAW-UHFFFAOYSA-N herniarin Natural products C1CC(=O)OC2=CC(OC)=CC=C21 JHGVLAHJJNKSAW-UHFFFAOYSA-N 0.000 description 1
- VLKZOEOYAKHREP-UHFFFAOYSA-N hexane Substances CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 1
- XXMIOPMDWAUFGU-UHFFFAOYSA-N hexane-1,6-diol Chemical compound OCCCCCCO XXMIOPMDWAUFGU-UHFFFAOYSA-N 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 150000002432 hydroperoxides Chemical class 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 229910052588 hydroxylapatite Inorganic materials 0.000 description 1
- 238000010191 image analysis Methods 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- NVBMGOHJUIFNAH-UHFFFAOYSA-M lithium;2,4,6-tri(propan-2-yl)benzenesulfinate Chemical compound [Li+].CC(C)C1=CC(C(C)C)=C(S([O-])=O)C(C(C)C)=C1 NVBMGOHJUIFNAH-UHFFFAOYSA-M 0.000 description 1
- XFBCAFRNNFDRNG-UHFFFAOYSA-M lithium;2,4,6-triethylbenzenesulfinate Chemical compound [Li+].CCC1=CC(CC)=C(S([O-])=O)C(CC)=C1 XFBCAFRNNFDRNG-UHFFFAOYSA-M 0.000 description 1
- MIVOPSLBXCQIBW-UHFFFAOYSA-M lithium;2,4,6-trimethylbenzenesulfinate Chemical compound [Li+].CC1=CC(C)=C(S([O-])=O)C(C)=C1 MIVOPSLBXCQIBW-UHFFFAOYSA-M 0.000 description 1
- MSUZXYWQEDRGFN-UHFFFAOYSA-M lithium;4-methylbenzenesulfinate Chemical compound [Li+].CC1=CC=C(S([O-])=O)C=C1 MSUZXYWQEDRGFN-UHFFFAOYSA-M 0.000 description 1
- NVSKMOMNGUJKCL-UHFFFAOYSA-M lithium;benzenesulfinate Chemical compound [Li+].[O-]S(=O)C1=CC=CC=C1 NVSKMOMNGUJKCL-UHFFFAOYSA-M 0.000 description 1
- 235000010187 litholrubine BK Nutrition 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- ALTWGIIQPLQAAM-UHFFFAOYSA-N metavanadate Chemical compound [O-][V](=O)=O ALTWGIIQPLQAAM-UHFFFAOYSA-N 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- ARYZCSRUUPFYMY-UHFFFAOYSA-N methoxysilane Chemical compound CO[SiH3] ARYZCSRUUPFYMY-UHFFFAOYSA-N 0.000 description 1
- 229940095102 methyl benzoate Drugs 0.000 description 1
- WBYWAXJHAXSJNI-UHFFFAOYSA-N methyl p-hydroxycinnamate Natural products OC(=O)C=CC1=CC=CC=C1 WBYWAXJHAXSJNI-UHFFFAOYSA-N 0.000 description 1
- 229960001047 methyl salicylate Drugs 0.000 description 1
- LVHBHZANLOWSRM-UHFFFAOYSA-N methylenebutanedioic acid Natural products OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 description 1
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 239000011325 microbead Substances 0.000 description 1
- 229910052901 montmorillonite Inorganic materials 0.000 description 1
- LKNDYQNNDRTHFP-UHFFFAOYSA-N n,n,3,4-tetramethylaniline Chemical compound CN(C)C1=CC=C(C)C(C)=C1 LKNDYQNNDRTHFP-UHFFFAOYSA-N 0.000 description 1
- NBFRQCOZERNGEX-UHFFFAOYSA-N n,n,3,5-tetramethylaniline Chemical compound CN(C)C1=CC(C)=CC(C)=C1 NBFRQCOZERNGEX-UHFFFAOYSA-N 0.000 description 1
- CWOMTHDOJCARBY-UHFFFAOYSA-N n,n,3-trimethylaniline Chemical compound CN(C)C1=CC=CC(C)=C1 CWOMTHDOJCARBY-UHFFFAOYSA-N 0.000 description 1
- GYVGXEWAOAAJEU-UHFFFAOYSA-N n,n,4-trimethylaniline Chemical compound CN(C)C1=CC=C(C)C=C1 GYVGXEWAOAAJEU-UHFFFAOYSA-N 0.000 description 1
- HKJNHYJTVPWVGV-UHFFFAOYSA-N n,n-diethyl-4-methylaniline Chemical compound CCN(CC)C1=CC=C(C)C=C1 HKJNHYJTVPWVGV-UHFFFAOYSA-N 0.000 description 1
- MJRUTYCVCLZWSR-UHFFFAOYSA-N n,n-dimethyl-4-propan-2-ylaniline Chemical compound CC(C)C1=CC=C(N(C)C)C=C1 MJRUTYCVCLZWSR-UHFFFAOYSA-N 0.000 description 1
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- OFBQJSOFQDEBGM-UHFFFAOYSA-N n-pentane Natural products CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 1
- ZHHKVLCBIBQGKO-UHFFFAOYSA-H naphthol green B Chemical compound [Na+].[Na+].[Na+].[Fe+3].[O-]S(=O)(=O)C1=CC=C2C(=N[O-])C(=O)C=CC2=C1.[O-]S(=O)(=O)C1=CC=C2C(=N[O-])C(=O)C=CC2=C1.[O-]S(=O)(=O)C1=CC=C2C(=N[O-])C(=O)C=CC2=C1 ZHHKVLCBIBQGKO-UHFFFAOYSA-H 0.000 description 1
- CTIQLGJVGNGFEW-UHFFFAOYSA-L naphthol yellow S Chemical compound [Na+].[Na+].C1=C(S([O-])(=O)=O)C=C2C([O-])=C([N+]([O-])=O)C=C([N+]([O-])=O)C2=C1 CTIQLGJVGNGFEW-UHFFFAOYSA-L 0.000 description 1
- SLCVBVWXLSEKPL-UHFFFAOYSA-N neopentyl glycol Chemical compound OCC(C)(C)CO SLCVBVWXLSEKPL-UHFFFAOYSA-N 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 125000000018 nitroso group Chemical group N(=O)* 0.000 description 1
- 229910000510 noble metal Inorganic materials 0.000 description 1
- IOQPZZOEVPZRBK-UHFFFAOYSA-N octan-1-amine Chemical compound CCCCCCCCN IOQPZZOEVPZRBK-UHFFFAOYSA-N 0.000 description 1
- 239000011022 opal Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- MHHDXUNFNAZUGB-UHFFFAOYSA-N oxidovanadium(2+) Chemical compound [V+2]=O MHHDXUNFNAZUGB-UHFFFAOYSA-N 0.000 description 1
- BFYJDHRWCNNYJQ-UHFFFAOYSA-N oxo-(3-oxo-3-phenylpropoxy)-(2,4,6-trimethylphenyl)phosphanium Chemical compound CC1=CC(C)=CC(C)=C1[P+](=O)OCCC(=O)C1=CC=CC=C1 BFYJDHRWCNNYJQ-UHFFFAOYSA-N 0.000 description 1
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 1
- ZRSNZINYAWTAHE-UHFFFAOYSA-N p-methoxybenzaldehyde Chemical compound COC1=CC=C(C=O)C=C1 ZRSNZINYAWTAHE-UHFFFAOYSA-N 0.000 description 1
- NWVVVBRKAWDGAB-UHFFFAOYSA-N p-methoxyphenol Chemical compound COC1=CC=C(O)C=C1 NWVVVBRKAWDGAB-UHFFFAOYSA-N 0.000 description 1
- 239000003002 pH adjusting agent Substances 0.000 description 1
- FZUGPQWGEGAKET-UHFFFAOYSA-N parbenate Chemical compound CCOC(=O)C1=CC=C(N(C)C)C=C1 FZUGPQWGEGAKET-UHFFFAOYSA-N 0.000 description 1
- 235000012736 patent blue V Nutrition 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical compound [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 description 1
- TZMFJUDUGYTVRY-UHFFFAOYSA-N pentane-2,3-dione Chemical compound CCC(=O)C(C)=O TZMFJUDUGYTVRY-UHFFFAOYSA-N 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 125000005634 peroxydicarbonate group Chemical group 0.000 description 1
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 description 1
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920001084 poly(chloroprene) Polymers 0.000 description 1
- 229920001483 poly(ethyl methacrylate) polymer Polymers 0.000 description 1
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 230000037048 polymerization activity Effects 0.000 description 1
- 229920000193 polymethacrylate Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229910052573 porcelain Inorganic materials 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- DJEHXEMURTVAOE-UHFFFAOYSA-M potassium bisulfite Chemical compound [K+].OS([O-])=O DJEHXEMURTVAOE-UHFFFAOYSA-M 0.000 description 1
- 235000010259 potassium hydrogen sulphite Nutrition 0.000 description 1
- BHZRJJOHZFYXTO-UHFFFAOYSA-L potassium sulfite Chemical compound [K+].[K+].[O-]S([O-])=O BHZRJJOHZFYXTO-UHFFFAOYSA-L 0.000 description 1
- 235000019252 potassium sulphite Nutrition 0.000 description 1
- HPWMVNFJSFHJIW-UHFFFAOYSA-M potassium;2,4,6-tri(propan-2-yl)benzenesulfinate Chemical compound [K+].CC(C)C1=CC(C(C)C)=C(S([O-])=O)C(C(C)C)=C1 HPWMVNFJSFHJIW-UHFFFAOYSA-M 0.000 description 1
- NLZLDWFYQXFPSZ-UHFFFAOYSA-M potassium;2,4,6-triethylbenzenesulfinate Chemical compound [K+].CCC1=CC(CC)=C(S([O-])=O)C(CC)=C1 NLZLDWFYQXFPSZ-UHFFFAOYSA-M 0.000 description 1
- SCRWQCKSJIHKKV-UHFFFAOYSA-M potassium;2,4,6-trimethylbenzenesulfinate Chemical compound [K+].CC1=CC(C)=C(S([O-])=O)C(C)=C1 SCRWQCKSJIHKKV-UHFFFAOYSA-M 0.000 description 1
- YVHWGMMGEAQQRT-UHFFFAOYSA-M potassium;4-methylbenzenesulfinate Chemical compound [K+].CC1=CC=C(S([O-])=O)C=C1 YVHWGMMGEAQQRT-UHFFFAOYSA-M 0.000 description 1
- LZSBNSVOXWMXLL-UHFFFAOYSA-M potassium;benzenesulfinate Chemical compound [K+].[O-]S(=O)C1=CC=CC=C1 LZSBNSVOXWMXLL-UHFFFAOYSA-M 0.000 description 1
- 238000000634 powder X-ray diffraction Methods 0.000 description 1
- 150000003139 primary aliphatic amines Chemical class 0.000 description 1
- SCUZVMOVTVSBLE-UHFFFAOYSA-N prop-2-enenitrile;styrene Chemical compound C=CC#N.C=CC1=CC=CC=C1 SCUZVMOVTVSBLE-UHFFFAOYSA-N 0.000 description 1
- BWJUFXUULUEGMA-UHFFFAOYSA-N propan-2-yl propan-2-yloxycarbonyloxy carbonate Chemical compound CC(C)OC(=O)OOC(=O)OC(C)C BWJUFXUULUEGMA-UHFFFAOYSA-N 0.000 description 1
- SLUHLANJIVXTRQ-UHFFFAOYSA-N pyridin-2-ylthiourea Chemical compound NC(=S)NC1=CC=CC=N1 SLUHLANJIVXTRQ-UHFFFAOYSA-N 0.000 description 1
- 235000012752 quinoline yellow Nutrition 0.000 description 1
- 239000004172 quinoline yellow Substances 0.000 description 1
- IZMJMCDDWKSTTK-UHFFFAOYSA-N quinoline yellow Chemical compound C1=CC=CC2=NC(C3C(C4=CC=CC=C4C3=O)=O)=CC=C21 IZMJMCDDWKSTTK-UHFFFAOYSA-N 0.000 description 1
- 229940051201 quinoline yellow Drugs 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 229920005604 random copolymer Polymers 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 239000003829 resin cement Substances 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 239000003566 sealing material Substances 0.000 description 1
- 150000005619 secondary aliphatic amines Chemical class 0.000 description 1
- 230000001568 sexual effect Effects 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 1
- CMZUMMUJMWNLFH-UHFFFAOYSA-N sodium metavanadate Chemical compound [Na+].[O-][V](=O)=O CMZUMMUJMWNLFH-UHFFFAOYSA-N 0.000 description 1
- 235000010265 sodium sulphite Nutrition 0.000 description 1
- WWYACYKHLMCNBG-UHFFFAOYSA-M sodium;2,4,6-tri(propan-2-yl)benzenesulfinate Chemical compound [Na+].CC(C)C1=CC(C(C)C)=C(S([O-])=O)C(C(C)C)=C1 WWYACYKHLMCNBG-UHFFFAOYSA-M 0.000 description 1
- KNHRGMOGYVTHPU-UHFFFAOYSA-M sodium;2,4,6-triethylbenzenesulfinate Chemical compound [Na+].CCC1=CC(CC)=C(S([O-])=O)C(CC)=C1 KNHRGMOGYVTHPU-UHFFFAOYSA-M 0.000 description 1
- GRBCNEIBDFNEES-UHFFFAOYSA-M sodium;2,4,6-trimethylbenzenesulfinate Chemical compound [Na+].CC1=CC(C)=C(S([O-])=O)C(C)=C1 GRBCNEIBDFNEES-UHFFFAOYSA-M 0.000 description 1
- CHLCPTJLUJHDBO-UHFFFAOYSA-M sodium;benzenesulfinate Chemical compound [Na+].[O-]S(=O)C1=CC=CC=C1 CHLCPTJLUJHDBO-UHFFFAOYSA-M 0.000 description 1
- 239000002689 soil Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 239000012798 spherical particle Substances 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 1
- 150000003440 styrenes Chemical class 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-L sulfite Chemical class [O-]S([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-L 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 230000003746 surface roughness Effects 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 238000010557 suspension polymerization reaction Methods 0.000 description 1
- KUCOHFSKRZZVRO-UHFFFAOYSA-N terephthalaldehyde Chemical compound O=CC1=CC=C(C=O)C=C1 KUCOHFSKRZZVRO-UHFFFAOYSA-N 0.000 description 1
- OPQYOFWUFGEMRZ-UHFFFAOYSA-N tert-butyl 2,2-dimethylpropaneperoxoate Chemical compound CC(C)(C)OOC(=O)C(C)(C)C OPQYOFWUFGEMRZ-UHFFFAOYSA-N 0.000 description 1
- JIYXDFNAPHIAFH-UHFFFAOYSA-N tert-butyl 3-tert-butylperoxycarbonylbenzoate Chemical compound CC(C)(C)OOC(=O)C1=CC=CC(C(=O)OC(C)(C)C)=C1 JIYXDFNAPHIAFH-UHFFFAOYSA-N 0.000 description 1
- NMOALOSNPWTWRH-UHFFFAOYSA-N tert-butyl 7,7-dimethyloctaneperoxoate Chemical compound CC(C)(C)CCCCCC(=O)OOC(C)(C)C NMOALOSNPWTWRH-UHFFFAOYSA-N 0.000 description 1
- SWAXTRYEYUTSAP-UHFFFAOYSA-N tert-butyl ethaneperoxoate Chemical compound CC(=O)OOC(C)(C)C SWAXTRYEYUTSAP-UHFFFAOYSA-N 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- CIHOLLKRGTVIJN-UHFFFAOYSA-N tert‐butyl hydroperoxide Chemical compound CC(C)(C)OO CIHOLLKRGTVIJN-UHFFFAOYSA-N 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- ZEMGGZBWXRYJHK-UHFFFAOYSA-N thiouracil Chemical class O=C1C=CNC(=S)N1 ZEMGGZBWXRYJHK-UHFFFAOYSA-N 0.000 description 1
- 150000003585 thioureas Chemical class 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- LDHQCZJRKDOVOX-UHFFFAOYSA-N trans-crotonic acid Natural products CC=CC(O)=O LDHQCZJRKDOVOX-UHFFFAOYSA-N 0.000 description 1
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 1
- HQYALQRYBUJWDH-UHFFFAOYSA-N trimethoxy(propyl)silane Chemical compound CCC[Si](OC)(OC)OC HQYALQRYBUJWDH-UHFFFAOYSA-N 0.000 description 1
- UOKUUKOEIMCYAI-UHFFFAOYSA-N trimethoxysilylmethyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)COC(=O)C(C)=C UOKUUKOEIMCYAI-UHFFFAOYSA-N 0.000 description 1
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 1
- 150000003673 urethanes Chemical class 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 1
- 229910001935 vanadium oxide Inorganic materials 0.000 description 1
- 229920001567 vinyl ester resin Polymers 0.000 description 1
- 239000012463 white pigment Substances 0.000 description 1
- 229910052724 xenon Inorganic materials 0.000 description 1
- FHNFHKCVQCLJFQ-UHFFFAOYSA-N xenon atom Chemical compound [Xe] FHNFHKCVQCLJFQ-UHFFFAOYSA-N 0.000 description 1
- 239000001052 yellow pigment Substances 0.000 description 1
- NAWDYIZEMPQZHO-UHFFFAOYSA-N ytterbium Chemical compound [Yb] NAWDYIZEMPQZHO-UHFFFAOYSA-N 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
- NWONKYPBYAMBJT-UHFFFAOYSA-L zinc sulfate Chemical compound [Zn+2].[O-]S([O-])(=O)=O NWONKYPBYAMBJT-UHFFFAOYSA-L 0.000 description 1
- 229960001763 zinc sulfate Drugs 0.000 description 1
- 229910000368 zinc sulfate Inorganic materials 0.000 description 1
- 229910052984 zinc sulfide Inorganic materials 0.000 description 1
- NDKWCCLKSWNDBG-UHFFFAOYSA-N zinc;dioxido(dioxo)chromium Chemical compound [Zn+2].[O-][Cr]([O-])(=O)=O NDKWCCLKSWNDBG-UHFFFAOYSA-N 0.000 description 1
- DRDVZXDWVBGGMH-UHFFFAOYSA-N zinc;sulfide Chemical compound [S-2].[Zn+2] DRDVZXDWVBGGMH-UHFFFAOYSA-N 0.000 description 1
- 229910001928 zirconium oxide Inorganic materials 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/15—Compositions characterised by their physical properties
- A61K6/16—Refractive index
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/15—Compositions characterised by their physical properties
- A61K6/17—Particle size
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/30—Compositions for temporarily or permanently fixing teeth or palates, e.g. primers for dental adhesives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/70—Preparations for dentistry comprising inorganic additives
- A61K6/71—Fillers
- A61K6/76—Fillers comprising silicon-containing compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/70—Preparations for dentistry comprising inorganic additives
- A61K6/71—Fillers
- A61K6/77—Glass
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/70—Preparations for dentistry comprising inorganic additives
- A61K6/78—Pigments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/884—Preparations for artificial teeth, for filling teeth or for capping teeth comprising natural or synthetic resins
- A61K6/887—Compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
Definitions
- the present invention relates to a dental curable composition that can be suitably used as a dental material that can replace a part or all of natural teeth, particularly a dental composite resin, in the field of dentistry.
- dental composite resins especially dental composite resins, amalgam, gold alloys, etc. were used for caries filling treatment.
- dental composite resins have rapidly become widespread because they are inexpensive and can achieve a color tone close to that of natural teeth relatively easily.
- due to the improvement of mechanical strength of dental filling restoration materials and the improvement of adhesive strength with teeth it is used not only for anterior teeth but also for molars where high occlusal pressure is applied. Made with filling repair materials.
- Patent Document 1 a filler that is close to the refractive index of a cured product of a polymerizable monomer is used as a base filler, and a cured product of a composition containing the filler and the polymerizable monomer is used.
- a dental composite material containing a different kind of filler having a different refractive index from the cured product of the monomer and having an average particle size of 1 ⁇ m or more has been proposed. Since such dental composites have a specific transmitted light diffusivity and light diffusivity similar to that of natural teeth, they can provide highly aesthetic restorations.
- the present invention has been made to solve the problems of the prior art, and is capable of highly aesthetic restoration in harmony with the surroundings so as to be indistinguishable from natural teeth, and is used for dentistry having one kind of composition. It is an object of the present invention to provide a dental curable composition which is a curable composition and exhibits good color compatibility with a wide range of natural teeth.
- the present invention is a dental curable composition having one composition, particularly for cases where a cavity bottom is present, such as class I, class II, or class V, and has a good color tone for natural teeth having a wide range of color tones. It is an object of the present invention to provide a dental curable composition showing compatibility.
- the present inventors measured a cured product of a dental curable composition on a white background using a spectrocolorimeter, and found that in a certain range of wavelength regions. It has been found that the dental curable composition exhibits good color compatibility with a wide range of natural teeth due to the substantially constant spectral reflectance.
- a cured product having a thickness of 1.0 mm of the dental curable composition was measured at wavelengths of 650 nm, 700 nm, and 750 nm with respect to the spectral reflectance at a wavelength of 600 nm when measured on a white background using a spectrocolorimeter.
- LD (I 5 / cos 5 °) / I 0 (2)
- I represents the luminous intensity of the light transmitted through the cured product
- I 0 and I 5 are the directions inclined by 0 degrees and 5 degrees with respect to the direction perpendicular to the sample plate (light incident direction), respectively.
- the dental curable composition according to any one of [1] to [3], which is 3.0 to 2.0.
- the filler (B) contains inorganic agglomerated particles (BF-2) formed by aggregating inorganic primary particles (x), and the average particle size of the inorganic primary particles (x) is 0.001 to 1 ⁇ m.
- the dental curable composition according to any one of [1] to [5].
- the inorganic agglomerated particles (BF-2) contain light-diffusing inorganic agglomerated particles (BF-2d), and the refractive index of the light-diffusing inorganic agglomerated particles (BF-2d) has the following formula (3).
- the filler (B) contains an organic-inorganic composite filler (BC) containing the inorganic primary particles (x), and the average particle diameter of the inorganic primary particles (x) is 0.001 to 1 ⁇ m. , [1] to [7].
- the dental curable composition according to any one of [1] to [7].
- the organic-inorganic composite filler (BC) contains a light-diffusing organic-inorganic composite filler (BC-d), and the refractive index of the light-diffusing organic-inorganic composite filler (BC-d) is as follows.
- nP is the refractive index of the polymer obtained by polymerizing the polymerizable monomer (A)
- nF BC-d is the refractive index of the light diffusing organic-inorganic composite filler (BC-d). Represents the rate.
- a dental curable composition having one composition is good for natural teeth having a wide range of colors. It is possible to provide a dental curable composition showing a good color compatibility. Further, according to the present invention, one type of dental curable composition is good for natural teeth having a wide range of tones, especially for cases in which the bottom of the cavity is present, such as class I, class II, or class V. It is possible to provide a dental curable composition showing a good color compatibility. Furthermore, the dental curable composition of the present invention can be suitably used for dental composite resins. Further, according to the present invention, it is possible to provide a dental curable composition in which the cured product is excellent in easy-polishing property and slipping durability. Further, according to the present invention, it is possible to provide a dental curable composition having excellent operability.
- the dental curable composition of the present invention is a dental curable composition containing a polymerizable monomer (A), a filler (B), a polymerization initiator (C), and a colorant (D). , 650, 700, and 750 nm with respect to the spectral reflectance at a wavelength of 600 nm when a cured product having a thickness of 1.0 mm of the dental curable composition was measured on a white background using a spectrocolorimeter.
- the ratio R of the reflectance in each of the above is in the range of 97% to 103%.
- one type of dental curable composition is a dental curable composition that exhibits good color compatibility with natural teeth having a wide range of colors.
- the dental curable composition of the present invention has a spectral reflectance at a wavelength of 600 nm when a cured product having a thickness of 1.0 mm of the dental curable composition is measured on a white background using a spectrocolorimeter.
- the R 650/600 , R 700/600 , and R 750/600 are all preferably in the range of 97.5% to 102.5%, preferably in the range of 97.8% to 102.2%. It is more preferably within the range of 98.0% to 102.0%.
- the color tone of the bottom of the natural tooth is reflected in the filling portion of the tooth filled with the dental curable composition. It will be easier. Due to this effect, even if it is not necessary to prepare multiple types of dental curable compositions having different color tones, one type of dental curable composition is very aesthetically pleasing to natural teeth having a wide range of color tones.
- r650 ⁇ 100 is preferably in the range of 97.0% to 103.0%, more preferably in the range of 97.5% to 102.5%, and 97.8% to 97.8%. It is more preferably in the range of 102.2%, and particularly preferably in the range of 98.0% to 102.0%.
- the incident light passes through the plate of the cured product, is reflected on the white background, and the light (measurement light) transmitted through the plate of the cured product is measured again.
- the curve of the spectral reflectance becomes almost a plateau in the above range, it means that the measured light is constant in the range.
- this measurement light is constant means that the light absorbed by the cured product when the light reflected on the white background passes through the cured product is constant regardless of the wavelength, and the background color remains the same. It means that it can be reflected on the surface. Since the cured product of the dental curable composition of the present invention shows a constant reflected light with respect to the wavelength of yellow to red, which is the color tone of natural teeth of 600 to 750 nm, the background is very large for colors in this range. In other words, it is possible to pick up the background color well. This characteristic is clinically very effective, especially in cases where the bottom of the tooth cavity (bottom of the cavity) such as class I, class II, and class V in the Black classification is present.
- the classification of Black means the classification of the type of tooth cavity, and includes the class I cavity, the class II cavity, the class III cavity, the class IV cavity, and the class V cavity.
- Class I cavities are cavities formed for caries that originate in the premolar fissures and the like of molars.
- Class II cavities are cavities that originate on the adjacent surface of the molars.
- the class III tooth cavity is a tooth cavity that originates on the adjacent surface of the anterior teeth and canines and does not include the angle of the incision.
- a class IV tooth cavity is a tooth cavity that originates on the adjacent surface of the anterior tooth and canine tooth and includes the angle of the incision.
- the V-class cavity is a cavity for the gingival side 1/3 of the lip (cheek) side or tongue (palatal) side of the crown.
- the cured product reflects the color tone of the bottom of the cavity of the natural tooth well, so that a highly aesthetic restoration that harmonizes with the surroundings is possible.
- one composition of the dental curable composition exhibits good color compatibility with a wide range of natural teeth.
- the spectral reflectance can be measured by using a spectral colorimeter capable of measuring the reflected light.
- the spectral colorimeter can be measured by, for example, a commercially available product such as the spectral colorimeters "SE7700" and "SE6000” manufactured by Nippon Denshoku Industries Co., Ltd.
- the XYZ tristimulus values measured by the 0 ° -45 ° method of the C / 2 light source, JIS Z8722 are 90 ⁇ X ⁇ 96, 92 ⁇ Y ⁇ 98, and 100 ⁇ Z ⁇ 116.
- the spectral reflectance of the cured product of the dental curable composition of the present invention can be specifically measured by the method described in Examples described later.
- the adjustment of the spectral reflectance can be achieved by increasing or decreasing the types and blending ratios of the filler (B) and the colorant (D) described later.
- a curable composition without coloring is prepared using the polymerizable monomer (A), the filler (B) and the polymerization initiator (C), and the spectral reflectance is measured.
- the spectral reflectance curve in the wavelength range of 600 to 750 nm tends to increase slightly or is preferably plateau.
- This can be controlled by the type and compounding ratio of the polymerizable monomer (A) and the filler (B).
- a colorant (D) particularly a yellow or red pigment
- the contrast ratio of the cured product having a thickness of 1.0 mm in the dental curable composition in the present invention is preferably 0.35 or more, more preferably 0.40 or more, and 0.43. The above is more preferable, and 0.45 or more is particularly preferable.
- the contrast ratio is preferably 0.65 or less, more preferably 0.60 or less, further preferably 0.57 or less, and particularly preferably 0.55 or less.
- the contrast ratio is a value obtained based on the following formula (1).
- Y b is the Y value of the XYZ color system measured on a black background using a color difference meter
- Y w is the Y value of the XYZ color system measured on a white background using a color difference meter.
- Contrast ratio Y b / Y w (1)
- the contrast ratio is an index of transparency, and the closer the value is to 1, the more opaque the material is, and the closer the value is to 0, the more transparent the material is.
- the contrast ratio can be measured by using the above-mentioned spectrocolorimeter or the like.
- a standard white plate is used as the white background, and the measurement is performed by adhering it to the cured plate of the dental curable composition to be measured.
- a matte dark box is used as the black background.
- the contrast ratio of the cured product of the dental curable composition of the present invention can be specifically measured by the method described in Examples described later.
- the adjustment of the contrast ratio can be achieved by adding the colorant (D) or increasing or decreasing the compounding ratio thereof. It is preferable to make adjustments according to the blending amount of a white pigment such as zinc oxide and titanium oxide because it has little effect on other color factors such as hue and saturation.
- the dental curable composition in the present invention preferably has further light diffusivity.
- the light diffusivity can be measured by using a variable angle photometer or a goniometer. That is, the light is incident vertically on the cured product, the luminous intensity of the positively transmitted light transmitted vertically and the diffused transmitted light of the portion other than the positively transmitted light are measured, and the luminous intensity of the diffused transmitted light with respect to the positively transmitted light is measured. By obtaining it, the degree of light diffusivity can be measured.
- the boundary between the natural tooth and the filling is blurred when the natural tooth cavity is filled and repaired with the dental curable composition of the present invention. The effect is obtained.
- the natural tooth and the filling portion are color-friendly, and the filling portion becomes inconspicuous, so that a restoration method having a very high aesthetic can be provided. It is also very effective especially in cases of repairing a cavity that penetrates from the labial side to the lingual side, such as class III or IV in the Black classification. Normally, in such cases, when repairing is performed using a dental curable composition having a low contrast ratio (high transparency), the repaired part looks dark and the color is lost, so that it is oxidized.
- the aesthetics can be improved by using a dental curable composition having an increased contrast ratio (decreased transparency) by increasing the amount of pigment such as titanium, but for dentistry with light diffusivity. In the curable composition, even a composition having a relatively low contrast ratio (high transparency) can give a natural and highly aesthetic appearance.
- the light diffusivity LD defined by the following formula (2) of the cured product having a thickness of 0.25 mm is preferably 0.0001 or more, preferably 0.001 or more. Is more preferable, and 0.0015 or more is further preferable.
- the light diffusivity LD is preferably 0.99 or less, more preferably 0.97 or less, and even more preferably 0.95 or less.
- the light diffusivity LD is defined by the following equation (2).
- I the luminous intensity of the light transmitted through the cured product
- I 0 and I 5 are the directions inclined by 0 degrees and 5 degrees with respect to the direction perpendicular to the sample plate (light incident direction), respectively. Represents the luminous intensity of transmitted light.
- the value of the luminous intensity of 5 degrees is divided by the cosine of that angle in order to make it a shape corresponding to the scale that can be perceived by the human eye.
- it can be converted to illuminance by dividing the luminous intensity by the cosine of the measurement angle from the definition of illuminance. Therefore, the closer the LD value is to 1, the stronger the light diffusivity.
- the light diffusivity LD can be measured by the method described in Examples described later.
- the adjustment of the light diffusivity can be achieved by blending the light diffusing filler and adjusting the blending amount, which will be described later.
- the dental curable composition of the present invention in order to reproduce the color tone of a natural tooth, even if various color tone parameters (contrast ratio, saturation, etc.) are not strictly controlled to be close to those of the natural tooth, the natural tooth is used.
- the cured product having a thickness of 1.0 mm of the dental curable composition in the present invention has an L * a * b * color on a white background.
- the reddish chromaticity index a * / w in the system is preferably -3.0 or more, more preferably -2.5 or more, and further preferably -2.0 or more. , -1.8 or more is particularly preferable.
- a * / w is preferably 2.0 or less, more preferably 1.5 or less, further preferably 1.0 or less, and particularly preferably 0.5 or less. .. When a * / w is within the above range, the color tone of the bottom of the cavity can be effectively reflected in the filling portion.
- a * / w can be measured by using the above-mentioned spectrocolorimeter or the like.
- a standard white plate is used as the white background, and the measurement can be performed by adhering it to the cured plate of the dental curable composition to be measured.
- the cured product a * / w of the dental curable composition of the present invention can be specifically measured by the method described in Examples described later.
- the adjustment of a * / w can be achieved by adding a pigment or increasing or decreasing the compounding ratio thereof.
- the L * a * b * table of the cured product having a thickness of 1.0 mm of the dental curable composition of the present invention is shown on a white background.
- the L * / w in the color system is preferably 60.0 or more, more preferably 65.0 or more, further preferably 68.0 or more, and particularly preferably 70.0 or more. preferable. Further, L * / w is preferably 90.0 or less, more preferably 88.0 or less, further preferably 85.0 or less, and particularly preferably 82.0 or less. ..
- the L * a * b * table of the cured product having a thickness of 1.0 mm of the dental curable composition of the present invention is shown on a white background.
- the b * / w in the color system is preferably 0.0 or more, more preferably 1.0 or more, further preferably 2.0 or more, and particularly preferably 3.0 or more. preferable. Further, b * / w is preferably 50.0 or less, more preferably 45.0 or less, further preferably 40.0 or less, and particularly preferably 35.0 or less. ..
- Polymerizable monomer (A) As the polymerizable monomer (A) in the dental curable composition of the present invention, known polymerizable monomers used in the dental curable composition can be used, and particularly radically polymerizable. Monomers can be preferably used. Examples of the radically polymerizable monomer include unsaturated carboxylic acids such as ⁇ -cyanoacrylic acid, (meth) acrylic acid, ⁇ -halogenated acrylic acid, crotonic acid, cinnamic acid, sorbic acid, maleic acid, and itaconic acid.
- unsaturated carboxylic acids such as ⁇ -cyanoacrylic acid, (meth) acrylic acid, ⁇ -halogenated acrylic acid, crotonic acid, cinnamic acid, sorbic acid, maleic acid, and itaconic acid.
- the polymerizable monomer (A) one type may be used alone, or two or more types may be used in combination. Among these, unsaturated carboxylic acid esters and (meth) acrylamide derivatives are preferable, (meth) acrylic acid esters and (meth) acrylamide derivatives are more preferable, and (meth) acrylic acid esters are even more preferable.
- the notation "(meth) acrylic” is used in the meaning which includes both methacrylic and acrylic.
- (meth) acrylic monomer is used in the sense of including both the (meth) acrylic acid ester and the (meth) acrylamide derivative. Examples of (meth) acrylic acid esters and (meth) acrylamide derivatives are shown below.
- (I) Monofunctional (meth) acrylic acid ester and (meth) acrylamide derivative For example, methyl (meth) acrylate, isobutyl (meth) acrylate, benzyl (meth) acrylate, dodecyl (meth) acrylate, 2- (N).
- Bifunctional (meth) acrylic acid ester For example, aromatic bifunctional (meth) acrylic acid ester, aliphatic bifunctional (meth) acrylic acid ester, and the like can be mentioned.
- aromatic bifunctional (meth) acrylic acid ester examples include 2,2-bis ((meth) acryloyloxyphenyl) propane and 2,2-bis [4- (3-acryloyloxy-2-). Hydroxypropoxy) Phenyl] Propane, 2,2-Bis [4- (3-methacryloyloxy-2-hydroxypropoxy) Phenyl] Propane (commonly known as Bis-GMA), 2,2-Bis (4- (meth) acryloyloxy) Polyethoxyphenyl) propane, 2- (4- (meth) acryloyloxydiethoxyphenyl) -2- (4- (meth) acryloyloxyethoxyphenyl) propane, 2- (4- (meth) acryloyloxydiethoxyphenyl) -2- (4- (meth) acryloyloxytriethoxyphenyl) propane, 2,2-bis (4- (meth) acryloyloxypropoxyphenyl) propane, 2,2-bis (4- (meth) acryl
- Examples of the aliphatic bifunctional (meth) acrylic acid ester include glycerol di (meth) acrylate, ethylene glycol di (meth) acrylate, diethylene glycol di (meth) acrylate, triethylene glycol diacrylate, and triethylene glycol.
- Dimethacrylate (commonly known as 3G), propylene glycol di (meth) acrylate, butylene glycol di (meth) acrylate, neopentyl glycol di (meth) acrylate, 1,3-butanediol di (meth) acrylate, 1,5-pentane Didioldi (meth) acrylate, 1,6-hexanediol di (meth) acrylate, 1,10-decanediol di (meth) acrylate, 1,12-dodecanediol di (meth) acrylate, 1,2-bis (3) -(Meta) acryloyloxy-2-hydroxypropyloxy) ethane, tricyclodecanedimethanol di (meth) acrylate, 2,2,4-trimethylhexamethylenebis (2-carbamoyloxyethyl) diacrylate, 2,2 Examples thereof include 4-trimethylhexamethylene bis (2-carbamoyl
- (Iii) Trifunctional or higher (meth) acrylic acid ester
- trimethylol propanetri (meth) acrylate trimethylol ethanetri (meth) acrylate, trimethylol methanetri (meth) acrylate, pentaerythritol tri (meth) acrylate.
- an oligomer or a polymer having a radically polymerizable group such as a (meth) acrylic acid ester group can also be preferably used, and for example, the polymerization described in JP-A No. 50-42696.
- Sexual prepolymers, unsaturated urethane-based oligomers described in JP-A-2011-144121, polyfunctional acrylate compounds described in JP-A-2006-510583, prepolymers described in WO2020 / 122192, and the like can be used.
- the polymerizable monomer (A) is a dental curable composition having excellent adhesiveness to dentin, metal, ceramics, etc., it is possible to impart adhesiveness to these adherends. It may be preferable to include a capable functional monomer.
- the functional monomer for example, 2- (meth) acryloyloxyethyl dihydrogen phosphate, 10- (meth) acryloyloxydecyldi, from the viewpoint of exhibiting excellent adhesion to dentin and base metal.
- Polymerizable monomer having a phosphate group such as hydrogen phosphate, 2- (meth) acryloyloxyethylphenylhydrogen phosphate; 11- (meth) acryloyloxyundecane-1,1-dicarboxylic acid, 4- (meth)
- Examples thereof include polymerizable monomers having a carboxylic acid group such as acryloyloxyethoxycarbonylphthalic acid.
- silane coupling agents such as ⁇ - (meth) acryloyloxypropyltrimethoxysilane can be mentioned from the viewpoint of being effective for adhesion to ceramics, porcelain, and other dental curable compositions. ..
- the content of the polymerizable monomer (A) in the dental curable composition of the present invention is not particularly limited, but from the viewpoint of operability of the obtained dental curable composition and mechanical strength of the cured product. Based on the total mass of the dental curable composition, it is preferably 1% by mass or more, more preferably 2% by mass or more, further preferably 5% by mass or more, and 8% by mass or more. It may be 15% by mass or more, preferably 70% by mass or less, more preferably 50% by mass or less, and further preferably 40% by mass or less. It is preferably 30% by mass or less, and particularly preferably 30% by mass or less.
- the dental curable composition of the present invention contains a filler (B).
- the overall shape of the particles of the filler (B) is not particularly limited, and can be used as an amorphous or spherical powder.
- a dental curable composition having particularly excellent mechanical strength, smoothing durability, and abrasion resistance of the cured product can be obtained, and a spherical filler (spherical filler) can be obtained.
- a dental curable composition having a smooth paste property, good elongation, and excellent operability can be obtained. It is considered that the reason why the amorphous filler is superior in mechanical strength to the spherical filler is that the particles of the filler mesh with each other in the cured product.
- the smoothing durability and wear resistance are superior to those of a spherical filler because the particles of the filler are less likely to fall off from the surface of the cured product, and the dropout marks are enlarged and the cured product is used. It is considered that this is because the increase in the surface roughness of the surface can be suppressed. It is considered that the reason why the spherical filler is superior in operability to the amorphous filler is that the spherical particles have a smaller specific surface area and a smaller contact area with the polymerizable monomer (A). Be done.
- the overall shape of the filler (B) may be appropriately selected according to the purpose of the dental curable composition, but the amorphous filler (B) is selected from the viewpoint of excellent mechanical strength and slipping durability. It is preferable to use it.
- the filler (B) in the present invention is roughly classified into an inorganic filler (BF), an organic-inorganic composite filler (BC), and an organic filler (BP).
- glass powders of common composition such as fused silica, quartz, soda lime silica glass, E glass, C glass, borosilicate glass (Pylex® glass); barium glass (GM27884, 8235, Schott, Inc., E-2000, E-3000, ESSTECH), Strontium borosilicate glass (E-4000, ESSTECH), Lantern glass ceramics (GM31684, Shot), Fluoroaluminosilicate glass (GM35429, G018-091), Dental glass powder such as G018-117 (manufactured by Shot)], various ceramics, alumina, silica-titania, silica-zirconia, silica-ittelvia and other composite oxides, diatomaceous soil, kaolin, clay minerals (montmorillonite, etc.),
- two or more types may be used in combination.
- those containing silica as a main component are preferable.
- inorganic fillers containing metal elements such as barium, zirconium, aluminum, and ytterbium, which have high X-ray contrast properties (barium glass, alumina, silica-titania, silica-zirconia, silica-ytterbium, etc.) and ytterbium fluoride are used. It is preferable to contain it.
- the inorganic filler (BF) may be amorphous, crystalline, or a mixture of both, but preferably contains at least an amorphous portion. Transparency is improved when the proportion of amorphous portions in the inorganic filler (BF) is large.
- the inorganic filler (BF) in the present invention preferably contains inorganic fine particles (BF-1) having an average particle size of 0.05 to 1 ⁇ m, more preferably 0.08 to 0.9 ⁇ m. It is more preferably 0.1 to 0.8 ⁇ m.
- the average particle size of the inorganic fine particles (BF-1) is at least the above lower limit, stickiness and stringiness of the obtained dental curable composition can be more effectively suppressed, and operability is improved.
- the average particle size of the inorganic fine particles (BF-1) is not more than the above upper limit, the obtained cured product of the dental curable composition is improved in easy polishing property and smoothness durability.
- the average particle size of the inorganic fine particles (BF-1) can be determined by a laser diffraction / scattering method or electron microscopic observation of the particles.
- the laser diffraction / scattering method is performed on a volume basis by using, for example, a laser diffraction type particle size distribution measuring device (“SALD-2300” manufactured by Shimadzu Corporation, etc.) and ethanol or a 0.2% sodium hexametaphosphate aqueous solution as a dispersion medium. Can be measured.
- SALD-2300 laser diffraction type particle size distribution measuring device
- the laser diffraction / scattering method is convenient for measuring the particle size of particles having a diameter of 0.1 ⁇ m or more.
- a scanning electron microscope (manufactured by Hitachi High-Technologies Corporation, SU3500, SU3800, S-4000, etc.) can be used for electron microscope observation.
- an electron micrograph of particles is taken, and the particle size of the particles (200 or more) observed in the unit field of the photograph is measured by image analysis type particle size distribution measurement software (Mac-View). It can be obtained by measuring using (manufactured by Mountech Co., Ltd.).
- the particle size is obtained as an arithmetic mean value of the longest length and the shortest length of the particles, and the average particle size is calculated from the number of particles and the particle size thereof.
- the average particle size of the inorganic fine particles (BF-1) can be measured more specifically by the method described in Examples.
- the average particle size of the inorganic filler (BF) means the average particle size before the surface treatment.
- the refractive index of the inorganic fine particles (BF-1) is not particularly limited, but can be obtained by approximating the refractive index of components other than the inorganic fine particles (BF-1) in the cured product of the dental curable composition. It becomes easy to increase the transparency of an object. From this, the refractive index of the inorganic fine particles (BF-1) is preferably 1.40 or more, more preferably 1.45 or more, further preferably 1.50 or more, and further. It is preferably 1.63 or less, more preferably 1.60 or less, and even more preferably 1.58 or less. The refractive index of the inorganic fine particles (BF-1) can be controlled by adjusting the type and ratio of the metal elements contained therein.
- the inorganic filler (BF) in the dental curable composition of the present invention preferably contains inorganic agglomerated particles (BF-2) in which the inorganic primary particles (x) are agglomerated.
- the average particle size of the inorganic primary particles (x) is preferably 0.001 to 1 ⁇ m, more preferably 0.005 to 0.8 ⁇ m, and even more preferably 0.01 to 0.5 ⁇ m. ..
- the average particle size of the inorganic primary particles (x) is at least the above lower limit, it is possible to prevent the specific surface area of the inorganic aggregated particles (BF-2) from increasing more than necessary, and thus the dental curable composition is sticky. And stringing can be suppressed more effectively and operability is improved.
- the average particle size of the inorganic primary particles (x) is not more than the above upper limit, the obtained cured product of the dental curable composition is improved in easy polishing property and smoothness durability. In addition, it is possible to provide a filling restoration having high color tone compatibility in that it is easy to obtain a glossiness equivalent to that of a natural tooth.
- the average particle size of the inorganic primary particles (x) can be determined by the above-mentioned laser diffraction / scattering method or a method by electron microscope observation.
- the inorganic aggregated particles (BF-2) are in the form of aggregated particles formed by agglomerating the inorganic primary particles (x).
- a commercially available inorganic filler exists as an agglomerate, but the powder of the inorganic filler is added to 300 mL of a dispersion medium such as water, water containing a surfactant such as 5% by mass or less of sodium hexametaphosphate, and ethanol.
- a dispersion medium such as water, water containing a surfactant such as 5% by mass or less of sodium hexametaphosphate, and ethanol.
- the inorganic aggregated particles (BF-2) in the present invention are particles in which particles that are hardly dispersed even under such conditions are strongly aggregated.
- agglomerated particles in which particles are strongly agglomerated from commercially available agglomerated particles of an inorganic filler, in order to further increase the cohesive force, the particles are heated to near the temperature immediately before melting and contacted.
- a method of heating to the extent that the particles of the obtained inorganic filler are slightly fused to each other is preferably used.
- an agglomerated morph may be formed before heating. Examples of the method include a method in which an inorganic filler is placed in an appropriate container and pressurized, or a method in which the solvent is once dispersed in a solvent and then the solvent is removed by a method such as spray drying.
- a sol such as silica sol, alumina sol, titania sol, or zirconia sol produced by a wet method is used. Examples thereof include a method of drying this by a method such as freeze-drying or spray-drying, and heat-treating it if necessary.
- a sol such as silica sol, alumina sol, titania sol, or zirconia sol produced by a wet method.
- examples thereof include a method of drying this by a method such as freeze-drying or spray-drying, and heat-treating it if necessary.
- sol examples include silica spherical fine particles (trade name "Sea Hoster (registered trademark)” (KE series, surface treatment type, etc .; above, manufactured by Nissan Chemical Industries, Ltd.)) and silica organosol (trade name "OSCAL (registered trademark)”.
- the shape of the inorganic agglomerated particles (BF-2) is not particularly limited, and can be appropriately selected and used.
- the conditions for heat treatment in the method for producing inorganic aggregated particles (BF-2) cannot be unconditionally defined because the optimum treatment conditions (temperature, time) differ depending on the composition of the inorganic primary particles (x) and the like.
- the heat treatment temperature (firing temperature) is preferably in the range of 500 to 1,200 ° C. If the heat treatment temperature is too low, the mechanical strength of the cured product of the finally obtained dental curable composition tends to decrease, and if the heat treatment temperature is too high, the inorganic primary particles (x) are charged with each other. The fusion of the particles is excessively advanced, and the polishability and smoothing durability of the cured product of the finally obtained dental curable composition are liable to decrease.
- inorganic agglomerated particles (aggregated particles) as secondary particles (aggregated particles) are obtained by firing under several conditions within the heat treatment temperature range as described above.
- BF-2 inorganic agglomerated particles
- it can be carried out by performing powder X-ray diffraction analysis on them and adopting conditions such that the crystal structure cannot be confirmed.
- a dental curable composition is produced using them, and the bending strength of the cured product formed from them and the cured product are obtained. It may be determined by measuring the smoothness of the polished surface in the above.
- insufficient heat treatment tends to result in insufficient bending strength of the cured product.
- the heat treatment is excessive, not only the appearance of the cured product becomes unnaturally opaque, but also the smoothness of the polished surface of the cured product tends to decrease. This is because when the heat treatment is excessive, some of the constituents start to crystallize, and the refractive index of the inorganic agglomerated particles (BF-2) increases accordingly, and the dental curable composition using the refractive index increases.
- the cured product formed from the dental curable composition shows an unnatural whiteness different from that of the natural tooth, and the hardness of the inorganic agglomerated particles (BF-2) is increased. It is considered that the cause is that the object becomes difficult to scrape and the polishability is deteriorated.
- the specific surface area of the inorganic agglomerated particles is preferably 10 m 2 / g or more, preferably 15 m 2 / g or more, more preferably 18 m 2 / g or more, and more preferably 20 m. It is more preferably 2 / g or more, more preferably 300 m 2 / g or less, preferably 250 m 2 / g or less, more preferably 200 m 2 / g or less, and 190 m 2 / g or less. It may be more preferably 170 m 2 / g or less, and further may be 150 m 2 / g or less.
- the specific surface area of the inorganic agglomerated particles (BF-2) is at least the above lower limit, the easy-polishing property of the cured product of the obtained dental curable composition is improved, and the color tone compatibility is also improved. Further, when the specific surface area of the inorganic agglomerated particles (BF-2) is not more than the above upper limit, the blending amount of the inorganic agglomerated particles (BF-2) can be further increased, and the mechanical strength of the obtained cured product is improved. ..
- the specific surface area of the inorganic agglomerated particles (BF-2) means the specific surface area of the agglomerated particles (secondary particles).
- the specific surface area of the inorganic agglomerated particles (BF-2) can be determined by the BET method. Specifically, for example, it can be measured using a specific surface area measuring device (“BELSORP-mini” series manufactured by Microtrac Bell Co., Ltd., etc.). The specific surface area of the inorganic agglomerated particles (BF-2) can be measured more specifically by the method described in Examples.
- the average particle size of the inorganic aggregated particles (BF-2) in the present invention is preferably 0.5 ⁇ m or more, more preferably 1 ⁇ m or more, further preferably 1.5 ⁇ m or more, and 2 ⁇ m or more. It is particularly preferably 50 ⁇ m or less, more preferably 40 ⁇ m or less, further preferably 30 ⁇ m or less, and particularly preferably 20 ⁇ m or less.
- the average particle size of the inorganic agglomerated particles (BF-2) is at least the above lower limit, stickiness and stringiness of the obtained dental curable composition can be more effectively suppressed, and operability is improved.
- the average particle size of the inorganic agglomerated particles (BF-2) means the average particle size of the agglomerated particles (secondary particles).
- the average particle size of the inorganic agglomerated particles (BF-2) can be determined by the above-mentioned laser diffraction / scattering method or a method by electron microscope observation.
- the average particle size of the inorganic agglomerated particles (BF-2) can be measured in the same manner as the measuring method of the inorganic fine particles (BF-1), and specifically, can be measured by the method described in Examples described later.
- the inorganic agglomerated particles (BF-2) in the dental curable composition of the present invention preferably contain light diffusible inorganic agglomerated particles (BF-2d).
- the light diffusing inorganic agglomerated particles (BF-2d) are formed by agglomerating the inorganic primary particles (x) as described in the inorganic agglomerated particles (BF-2).
- the light-diffusing inorganic agglomerated particles (BF-2d) mean that the refractive index satisfies the following formula (3), preferably satisfying the following formula (4), and more preferably satisfying the following formula (5). It is preferable that the following formula (6) is satisfied.
- the refractive index of the light diffusible inorganic agglomerated particles can be measured by the method described in Examples described later. 0.03 ⁇ nP-nF BF-2d
- the refractive index of the light-diffusing inorganic agglomerated particles (BF-2d) is at least the lower limit of the above formula (3), sufficient light-diffusing property can be imparted to the obtained dental curable composition.
- the margin (boundary) between the natural tooth and the filling is blurred and the filling portion becomes inconspicuous, so that it is aesthetically pleasing. Can provide a very high repair method.
- the content is not more than the upper limit of the above formula, a certain degree of transparency can be imparted to the obtained dental curable composition.
- the color tone of the bottom of the natural tooth can be reflected in the filling portion. Due to this effect, even if it is not necessary to prepare multiple types of dental curable compositions having different color tones, one type of dental curable composition is very aesthetically pleasing to natural teeth having a wide range of color tones. It can provide a high repair method.
- the refractive index nP of the polymer obtained by polymerizing the polymerizable monomer (A) is filled in the polymerizable monomer (A) as shown in Examples described later.
- a polymer having a thickness of 1.0 mm obtained by casting and polymerizing a mixture of all components (polymerization initiator (C), etc.) other than the material (B) and the colorant (D) under predetermined conditions. It means a value measured using an Abbe's fluorometer.
- the polymerizable monomer (A) is one kind, it is the refractive index of the mixture containing the homopolymer of the polymerizable monomer, and the polymerizable monomer (A) is the polymerizable of a plurality of kinds.
- the monomer is contained, it is the refractive index of the mixture containing the random copolymer of the plurality of kinds of polymerizable monomers.
- the "refractive index” means a value at 25 ° C. unless otherwise specified.
- the raw materials mentioned as usable in the inorganic filler (BF) can be used without any limitation, but the above formula is satisfied.
- Various glass powders with common compositions such as fused silica, quartz, soda lime silica glass, E glass, C glass, borosilicate glass (Pylex® glass), ceramics, alumina to control the refractive index.
- Silica-titania, silica-zirconia, silica-ittelvia and other composite oxides can be particularly preferably used. Each of these may be used alone or in combination of two or more.
- the average particle size of the light diffusing inorganic agglomerated particles (BF-2d) those having an average particle size in the range mentioned as being suitably usable for the inorganic agglomerated particles (BF-2) can be preferably used.
- the average particle size of the light-diffusing inorganic agglomerated particles (BF-2d) is equal to or larger than the above lower limit, the stickiness and stringiness of the obtained dental curable composition can be more effectively suppressed, and the operability is improved.
- the average particle size of the light-diffusing inorganic agglomerated particles (BF-2d) is not more than the above upper limit, it is possible to more effectively suppress the roughness and sagging of the obtained dental curable composition. Operability is improved.
- the inorganic agglomerated particles (BF-2) include other inorganic agglomerated particles (BF-2e) other than the light diffusing inorganic agglomerated particles (BF-2d) (hereinafter, simply “other inorganic agglomerated particles (BF-2e)). It may also contain.).
- the average particle size of the other inorganic agglomerated particles (BF-2e) is in the range mentioned as being suitable for use in the inorganic agglomerated particles (BF-2) as well as the light diffusing inorganic agglomerated particles (BF-2d). Those having an average particle size can be preferably used.
- the other inorganic agglomerated particles (BF-2e) can be produced by the method described for the inorganic agglomerated particles (BF-2).
- the inorganic primary particles (x) of the other inorganic agglomerated particles (BF-2e) the raw materials mentioned as being usable in the inorganic filler (BF) can be used without any limitation.
- the inorganic filler (BF) is an inorganic particle (BF-3) other than the inorganic fine particles (BF-1) and the inorganic agglomerated particles (BF-2) (hereinafter, simply “another inorganic particle (BF-3)). It may also contain.).
- the average particle size of the other inorganic particles (BF-3) is preferably more than 1 ⁇ m and 30 ⁇ m or less, more preferably more than 1 ⁇ m and 20 ⁇ m or less, and further preferably more than 1 ⁇ m and 10 ⁇ m or less.
- the average particle size of the other inorganic particles (BF-3) can be determined by a laser diffraction / scattering method or electron microscopic observation of the particles.
- the laser diffraction / scattering method is performed on a volume basis by using, for example, a laser diffraction type particle size distribution measuring device (“SALD-2300” manufactured by Shimadzu Corporation, etc.) and ethanol or a 0.2% sodium hexametaphosphate aqueous solution as a dispersion medium. Can be measured.
- SALD-2300 laser diffraction type particle size distribution measuring device
- SALD-2300 manufactured by Shimadzu Corporation, etc.
- ethanol or a 0.2% sodium hexametaphosphate aqueous solution as a dispersion medium.
- the organic-inorganic composite filler (BC) in the present invention refers to a filler containing the inorganic filler (BF) and a polymer of a polymerizable monomer (A').
- the average particle size of the organic-inorganic composite filler (BC) is preferably 0.5 ⁇ m or more, more preferably 1 ⁇ m or more, further preferably 1.5 ⁇ m or more, and further preferably 2 ⁇ m or more. Is particularly preferable, and it is preferably 50 ⁇ m or less, more preferably 40 ⁇ m or less, further preferably 30 ⁇ m or less, and particularly preferably 20 ⁇ m or less.
- the average particle size of the organic-inorganic composite filler (BC) is at least the above lower limit, stickiness and stringiness of the obtained dental curable composition can be more effectively suppressed, and operability is improved.
- the average particle size of the organic-inorganic composite filler (BC) is not more than the above upper limit, it is possible to more effectively suppress the roughness and sagging of the obtained dental curable composition, and the operability is also improved. improves.
- the average particle size of the organic-inorganic composite filler (BC) can be determined by the above-mentioned laser diffraction / scattering method or a method by electron microscope observation.
- the method for producing the organic-inorganic composite filler (BC) in the present invention is not particularly limited, but for example, a polymerizable monomer (A') and a known polymerization initiator are added in advance to the inorganic filler (BF). , After making it into a paste, it may be polymerized by solution polymerization, suspension polymerization, emulsion polymerization or bulk polymerization and pulverized.
- the content of the inorganic filler (BF) in the organic-inorganic composite filler (BC) is 100 in total of the inorganic filler (BF) and the polymerizable monomer (A') in the organic-inorganic composite filler (BC). 10 parts by mass or more is preferable, 20 parts by mass or more is more preferable, 30 parts by mass or more is further preferable, and 40 parts by mass or more is particularly preferable. When it is at least the above lower limit, the mechanical strength of the obtained dental curable composition is improved.
- the upper limit is not particularly limited, but 99 parts by mass or less is preferable, and 95 parts by mass or less is more preferable, because the viscosity of the paste increases in production.
- the polymerizable monomer listed as the polymerizable monomer (A) that can be used in the dental curable composition of the present invention is preferably used.
- the absolute value of the difference between the refractive index when the polymerizable monomer (A') is cured and the refractive index of the inorganic filler (BF) in the organic-inorganic composite filler (BC) is 0.30. It is preferably 0 or less, and more preferably 0.20 or less.
- the inorganic primary particles (x) are preferably used from the viewpoint of easy polishing and the like.
- the organic-inorganic composite filler (BC) in the dental curable composition of the present invention preferably contains a light-diffusing organic-inorganic composite filler (BC-d).
- the light-diffusing organic-inorganic composite filler (BC-d) is a polymer of the inorganic filler (BF) and the polymerizable monomer (A'), similarly to the organic-inorganic composite filler (BC). including.
- the light diffusible organic-inorganic composite filler (BC-d) means that the refractive index satisfies the following formula (7), preferably the following formula (8), and preferably the following formula (9). Is more preferable, and it is further preferable to satisfy the following formula (10).
- the refractive index of the light-diffusing organic-inorganic composite filler (BC-d) can be measured by the method described in Examples described later. 0.03 ⁇ nP-nF BC-d
- the refractive index of the light-diffusing organic-inorganic composite filler (BC-d) is at least the lower limit of the above formula, sufficient light-diffusing property can be imparted to the obtained dental curable composition.
- the margin (boundary) between the natural tooth and the filling is blurred and the filling portion becomes inconspicuous, so that it is aesthetically pleasing.
- the content is not more than the upper limit of the above formula, a certain degree of transparency can be imparted to the obtained dental curable composition.
- the color tone of the bottom of the natural tooth can be reflected in the filling portion. Due to this effect, even if it is not necessary to prepare multiple types of dental curable compositions having different color tones, one type of dental curable composition is very aesthetically pleasing to natural teeth having a wide range of color tones. It can provide a high repair method.
- the inorganic primary particles (x) can be preferably used.
- the raw materials mentioned as being usable in the inorganic filler (BF) can be used without any limitation, but in order to control the refractive index so as to satisfy the above formula, fused silica, quartz, etc.
- Various glass powders with general composition such as soda lime silica glass, E glass, C glass, borosilicate glass (Pylex (registered trademark) glass), ceramics, alumina, silica-titania, silica-zirconia, silica-ittelvia, etc.
- the composite oxide of the above can be used particularly preferably. Each of these may be used alone or in combination of two or more.
- the average particle size of the light-diffusing organic-inorganic composite filler (BC-d) those having an average particle size in the range mentioned as being suitable for use in the inorganic aggregated particles (BF-2) can be preferably used. ..
- the average particle size of the light-diffusing organic-inorganic composite filler (BC-d) is at least the above lower limit, the stickiness and stringiness of the obtained dental curable composition can be more effectively suppressed.
- it is not more than the above lower limit the bluish scattered light becomes strong, so that the color tone compatibility with the natural tooth is lowered.
- the average particle size of the light-diffusing organic-inorganic composite filler (BC-d) is not more than the above upper limit, it is possible to more effectively suppress the roughness and sagging of the obtained dental curable composition. After all, operability is improved.
- the average particle size of the light-diffusing organic-inorganic composite filler (BC-d) can be measured in the same manner as the measuring method of the inorganic fine particles (BF-1) in the same manner as the average particle size of the inorganic aggregated particles (BF-2). Specifically, it can be measured by the method described in Examples described later.
- the inorganic filler (BF) and the organic-inorganic composite filler (BC) may not be surface-treated, but the surfaces of the inorganic filler (BF) and the organic-inorganic composite filler (BC) are made hydrophobic and polymerizable.
- Surface treatment is preferable because the affinity with the monomer (A) is improved and the blending amount of the inorganic filler (BF) and the organic-inorganic composite filler (BC) can be increased. ..
- the surface treatment can be performed using a surface treatment agent.
- the type of the surface treatment agent is not particularly limited, and known surface treatment agents can be used.
- a silane coupling agent for example, a silane coupling agent, an organic titanium-based coupling agent, an organic zirconium-based coupling agent, and an organic aluminum-based coupling agent can be used. Etc. can be used.
- the surface treatment agent one type may be used alone, or two or more types may be used in combination.
- a silane coupling agent is preferable from the viewpoint of affinity and availability of the polymerizable monomer (A) with the inorganic filler (BF) and the organic-inorganic composite filler (BC).
- the type of the silane coupling agent is not particularly limited, but it is preferably a compound represented by the following general formula (11).
- H 2 C CR 4 -CO-R 5- (CH 2 ) q -SiR 6 p R 7 (3-p) (11)
- R 4 is a hydrogen atom or a methyl group
- R 5 is an oxygen atom, a sulfur atom, or -NR 8-
- R 8 is a hydrogen atom or an aliphatic group having 1 to 8 carbon atoms
- R 8 is a hydrogen atom or an aliphatic group having 1 to 8 carbon atoms.
- R 6 is a hydrolyzable group
- R 7 is a hydrocarbon group having 1 to 6 carbon atoms
- p is an integer of 1 to 3.
- Q is an integer of 1 to 13
- R 6 and R 7 existing in a plurality thereof may be the same or different from each other.
- R4 is a hydrogen atom or a methyl group, and is preferably a methyl group.
- R 5 is an oxygen atom, a sulfur atom, or ⁇ NR8 ⁇ , and is preferably an oxygen atom.
- the R 8 is a hydrogen atom or an aliphatic group having 1 to 8 carbon atoms (may be linear, branched or cyclic), and is represented by the R 8 and has 1 to 8 carbon atoms.
- the group may be either a saturated aliphatic group (alkyl group, cycloalkylene group (cyclohexyl group, etc.), etc.) or an unsaturated aliphatic group (alkenyl group, alkynyl group, etc.), and is available and easy to manufacture. From the viewpoint of chemical stability and the like, a saturated aliphatic group is preferable, and an alkyl group is more preferable.
- alkyl group examples include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, an isobutyl group, a sec-butyl group, a tert-butyl group, an n-pentyl group, an isopentyl group, and n-.
- examples thereof include a hexyl group, an n-heptyl group, a 2-methylhexyl group, and an n-octyl group.
- R 8 is preferably a hydrogen atom or an alkyl group having 1 to 4 carbon atoms, more preferably a hydrogen atom or an alkyl group having 1 to 3 carbon atoms, and even more preferably a hydrogen atom.
- examples of the hydrolable group represented by R 6 include an alkoxy group such as a methoxy group, an ethoxy group and a butoxy group; a halogen atom such as a chlorine atom and a bromine atom; an isocyanate group and the like. Can be mentioned. When a plurality of R 6s exist, the R 6s may be the same or different from each other. Among these, R6 is preferably an alkoxy group, more preferably a methoxy group or an ethoxy group, and even more preferably a methoxy group.
- the hydrocarbon group having 1 to 6 carbon atoms represented by R 7 is, for example, an alkyl group having 1 to 6 carbon atoms (which may be cyclic) or an alkenyl group having 2 to 6 carbon atoms. (It may be cyclic), an alkynyl group having 2 to 6 carbon atoms, and the like. When a plurality of R 7s exist, the R 7s may be the same or different from each other.
- alkyl group having 1 to 6 carbon atoms examples include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, an isobutyl group, a sec-butyl group, a tert-butyl group and an n-pentyl group. , Isopentyl group, neopentyl group, tert-pentyl group, n-hexyl group, cyclopropyl group, cyclobutyl group, cyclopentyl group, cyclohexyl group and the like.
- alkenyl group having 2 to 6 carbon atoms examples include a vinyl group, an allyl group, a 1-methylvinyl group, a 1-propenyl group, a butenyl group, a pentenyl group, a hexenyl group, a cyclopropenyl group, a cyclobutenyl group and a cyclopentenyl group. , Cyclohexenyl group and the like.
- alkynyl group having 2 to 6 carbon atoms examples include ethynyl, 1-propynyl, 2-propynyl, 1-butynyl, 1-methyl-2-propynyl, 2-butynyl, 3-butynyl, 1-pentynyl and 1-.
- p is an integer of 1 to 3, preferably 2 or 3, and more preferably 3.
- q is an integer of 1 to 13, preferably an integer of 2 to 12, and more preferably an integer of 3 to 11.
- silane coupling agent represented by the general formula (11) examples include methacryloyloxymethyltrimethoxysilane, 2-methacryloyloxyethyltrimethoxysilane, 3-methacryloxypropyltrimethoxysilane, and 4-methacryloyloxy.
- silane coupling agent may be used alone, or two or more types may be used in combination.
- 3-methacryloyloxypropyltrimethoxysilane is preferable from the viewpoint of availability, and the polymerizable monomer (A), the inorganic filler (BF), and the organic-inorganic composite filler (BC) are used.
- 8-methacryloyloxyoctyltrimethoxysilane, 9-methacryloyloxynonyltrimethoxysilane, 10-methacryloyloxydecyltrimethoxysilane, and 11-methacryloyloxyundecyltrimethoxysilane are preferable.
- the surface treatment method is not particularly limited, and a known method can be applied.
- the amount of the surface treatment agent used is not particularly limited, but the amount of the surface treatment agent used can be, for example, 1 part by mass or more with respect to 100 parts by mass of the filler before the surface treatment, and the amount used is 5 parts by mass. It is preferably 4 parts or more, more preferably 6 parts by mass or more, further preferably 8 parts by mass or more, particularly preferably 10 parts by mass or more, and most preferably 20 parts by mass or more. It is preferable, and it is preferably 50 parts by mass or less, more preferably 45 parts by mass or less, and further preferably 40 parts by mass or less.
- the amount used is at least the above lower limit, the mechanical strength of the obtained cured product is more likely to be improved, and when the amount used is not more than the above upper limit, the cured product obtained by the surplus surface treatment agent is used. It is possible to suppress a decrease in mechanical strength.
- Examples of the organic filler (BP) in the present invention include polymethyl methacrylate, ethyl polymethacrylate, methyl methacrylate-ethyl methacrylate copolymer, crosslinked polymethylmethacrylate, crosslinked polyethyl methacrylate, polyamide and the like.
- Acrylic polymers Polyvinyl chloride, polystyrene, chloroprene rubber, nitrile rubber, ethylene-vinyl acetate copolymer, styrene-butadiene copolymer, acrylonitrile-styrene copolymer, acrylonitrile-styrene-butadiene copolymer, etc.
- the shape of the organic filler is not particularly limited, and the particle size of the filler can be appropriately selected and used. From the viewpoint of handleability and mechanical strength of the obtained dental curable composition, the average particle size of the organic filler (BP) is preferably 0.001 to 50 ⁇ m, preferably 0.001 to 10 ⁇ m. It is more preferable to have.
- the content of the filler (B) in the dental curable composition of the present invention (the content of the filler (B) after the surface treatment when the surface is treated) is not particularly limited, but the obtained dentistry can be obtained. From the viewpoint of operability of the curable composition and mechanical strength of the cured product, the content of the filler (B) in the dental curable composition of the present invention is filled with the polymerizable monomer (A). In the total 100 parts by mass of the material (B), it is preferably 10 parts by mass or more, more preferably 30 parts by mass or more, further preferably 50 parts by mass or more, and 65 parts by mass or more.
- Is particularly preferable and is preferably 97 parts by mass or less, more preferably 96 parts by mass or less, further preferably 95 parts by mass or less, and particularly preferably 90 parts by mass or less.
- the content of the filler (B) is at least the above lower limit, the mechanical strength of the cured product is improved, and the stickiness and stringiness of the dental curable composition are more effectively suppressed, and the operability thereof is improved. improves. Further, when the content of the filler (B) is not more than the above upper limit, it is possible to prevent the obtained dental curable composition from becoming excessively hard, and the operability is improved.
- the content of the inorganic fine particles (BF-1) (the content of the inorganic fine particles (BF-1) after the surface treatment when the surface is treated) is not particularly limited.
- the content of the inorganic fine particles (BF-1) in the dental curable composition of the present invention is a polymerizable single.
- the total of 100 parts by mass of the weight (A) and the filler (B) it is preferably 1 part by mass or more, more preferably 3 parts by mass or more, and further preferably 5 parts by mass or more.
- the content of the inorganic fine particles (BF-1) is equal to or higher than the above lower limit, the mechanical strength of the cured product is improved, and the easy-polishing property of the obtained dental curable composition is improved. The color tone compatibility is improved. Further, when the content of the inorganic fine particles (BF-1) is not more than the above upper limit, it is possible to prevent the obtained dental curable composition from becoming excessively hard, and the operability is improved.
- the dental curable composition of the present invention preferably contains inorganic agglomerated particles (BF-2) and / or an organic-inorganic composite filler (BC).
- Total content of inorganic agglomerated particles (BF-2) and organic-inorganic composite filler (BC) (in the case of surface-treated inorganic agglomerated particles (BF-2) and organic-inorganic composite filler (if surface-treated)
- the content of BC) is not particularly limited, but from the viewpoint of operability of the obtained dental curable composition and mechanical strength of the cured product, the inorganic aggregated particles (inorganic aggregated particles) in the dental curable composition of the present invention.
- the total content of the BF-2) and the organic-inorganic composite filler (BC) is preferably 1 part by mass or more in a total of 100 parts by mass of the polymerizable monomer (A) and the filler (B). It is more preferably 2 parts by mass or more, further preferably 3 parts by mass or more, particularly preferably 4 parts by mass or more, and preferably 95 parts by mass or less, and 90 parts by mass or less. It is more preferably 80 parts by mass or less, and particularly preferably 70 parts by mass or less.
- the total content of the inorganic agglomerated particles (BF-2) and the organic-inorganic composite filler (BC) is at least the above lower limit, stickiness and stringing are more effectively suppressed and the operability is improved.
- the obtained dental curable composition is more effective in roughness and dripping. It can be suppressed to the above, and the operability is also improved.
- the dental curable composition of the present invention may contain either or both of light-diffusing inorganic agglomerated particles (BF-2d) and light-diffusing organic-inorganic composite filler (BC-d). preferable.
- Total content of light-diffusing inorganic agglomerated particles (BF-2d) and light-diffusing organic-inorganic composite filler (BC-d) is not particularly limited, but the operability of the obtained dental curable composition, the mechanical strength of the cured product, and the filler are not particularly limited.
- the total content of the light-diffusing inorganic agglomerated particles (BF-2d) and the light-diffusing organic-inorganic composite filler (BC-d) in the dental curable composition of the present invention is determined from the viewpoint of aesthetics and the like.
- it is preferably 0.5 parts by mass or more, more preferably 1 part by mass or more, and 2 parts by mass or more. It is more preferably 3 parts by mass or more, more preferably 50 parts by mass or less, further preferably 45 parts by mass or less, still more preferably 40 parts by mass or less. It is particularly preferably 35 parts by mass or less.
- the total content of the light-diffusing inorganic agglomerated particles (BF-2d) and the light-diffusing organic-inorganic composite filler (BC-d) is equal to or higher than the above lower limit, stickiness and stringiness are more effectively suppressed.
- it is possible to impart sufficient light diffusivity to the obtained dental curable composition and it is possible to provide a restoration method having very high aesthetics as described above.
- the dental curable composition obtained by the total content of the light-diffusing inorganic agglomerated particles (BF-2d) and the light-diffusing organic-inorganic composite filler (BC-d) is not more than the above upper limit.
- one type of dental curable composition has a wide range of color tones. It is possible to provide a highly aesthetic restoration method for natural teeth.
- the content of the organic filler (BP) in the dental curable composition of the present invention is not particularly limited and may not be contained.
- the dental curable composition containing the organic filler (BP) has reduced polymerization shrinkage during curing.
- the content of the organic filler (BP) in the dental curable composition of the present invention is the polymerizable monomer (A) and the inorganic filler ( In the total of 100 parts by mass of BF), it is preferably 10 parts by mass or less, 5 parts by mass or less, 3 parts by mass or less, or not included.
- the dental curable composition of the present invention contains a polymerization initiator (C).
- a polymerization initiator used in the general industry can be selected and used, and among them, the polymerization initiator used for dental use is preferably used.
- photopolymerization initiators and chemical polymerization initiators are preferably used.
- the polymerization initiator (C) one type may be used alone, or two or more types may be used in combination as appropriate.
- photopolymerization initiator examples include (bis) acylphosphine oxides, ketals, ⁇ -diketones, coumarins and the like.
- the acylphosphine oxides include 2,4,6-trimethylbenzoyldiphenylphosphine oxide, 2,6-dimethoxybenzoyldiphenylphosphine oxide, and 2,6-dichlorobenzoyldiphenylphosphine oxide, 2 , 4,6-trimethylbenzoylmethoxyphenylphosphine oxide, 2,4,6-trimethylbenzoylethoxyphenylphosphine oxide, 2,3,5,6-tetramethylbenzoyldiphenylphosphine oxide, benzoyldi (2,6-dimethylphenyl) phosphonate And these salts (sodium salt, potassium salt, ammonium salt, etc.) and the like.
- bisacylphosphine oxides include bis (2,6-dichlorobenzoyl) phenylphosphine oxide, bis (2,6-dichlorobenzoyl) -2,5-dimethylphenylphosphine oxide, and bis (2,6-dichlorobenzoyl)-.
- ketals examples include benzyldimethyl ketal, benzyldiethyl ketal and the like.
- ⁇ -diketones examples include diacetyl, benzyl, camphorquinone, 2,3-pentadione, 2,3-octadione, 9,10-phenanthrenequinone, 4,4'-oxybenzyl, acenaphtenquinone and the like. .. Among these, camphorquinone is preferable from the viewpoint of having a maximum absorption wavelength in the visible light region.
- Examples of the coumarin compound include 3,3'-carbonylbis (7-diethylaminocoumarin), 3- (4-methoxybenzoyl) coumarin, 3-thienoylcoumarin, 3-benzoyl-5,7-dimethoxycoumarin, 3-.
- 3,3'-carbonylbis (7-diethylaminocoumarin) and 3,3'-carbonylbis (7-dibutylaminocoumarin) are particularly preferable.
- the photopolymerization initiators selected from the group consisting of (bis) acylphosphine oxides, ⁇ -diketones, and coumarin compounds
- the photocurability in the visible and near-ultraviolet regions can be achieved.
- a dental curable composition that is excellent and exhibits sufficient photocurability can be obtained by using any light source of a halogen lamp, a light emitting diode (LED), and a xenon lamp.
- LED light emitting diode
- Organic peroxides are preferably used as the chemical polymerization initiator.
- the organic peroxide used for the above-mentioned chemical polymerization initiator is not particularly limited, and known ones can be used.
- Typical organic peroxides include ketone peroxides, hydroperoxides, diacyl peroxides, dialkyl peroxides, peroxyketals, peroxyesters, peroxydicarbonates and the like.
- ketone peroxide examples include methyl ethyl ketone peroxide, methyl isobutyl ketone peroxide, methyl cyclohexanone peroxide, cyclohexanone peroxide and the like.
- hydroperoxide examples include 2,5-dimethylhexane-2,5-dihydroperoxide, diisopropylbenzenehydroperoxide, cumene hydroperoxide, t-butylhydroperoxide, 1,1,3,3-tetramethylbutylhydroperoxide and the like. Be done.
- diacyl peroxide examples include acetyl peroxide, isobutyryl peroxide, benzoyl peroxide, decanoy peroxide, 3,5,5-trimethylhexanoyl peroxide, 2,4-dichlorobenzoyl peroxide, and lauroyl peroxide.
- dialkyl peroxide examples include di-t-butyl peroxide, dicumyl peroxide, t-butyl cumyl peroxide, 2,5-dimethyl-2,5-di (t-butylperoxy) hexane, and 1,3-bis (t-). Butylperoxyisopropyl) benzene, 2,5-dimethyl-2,5-di (t-butylperoxy) -3-hexane and the like can be mentioned.
- peroxyketal examples include 1,1-bis (t-butylperoxy) -3,3,5-trimethylcyclohexane, 1,1-bis (t-butylperoxy) cyclohexane, and 2,2-bis (t-butylperoxy).
- Butane, 2,2-bis (t-butylperoxy) octane, 4,4-bis (t-butylperoxy) valeric acid-n-butyl ester and the like can be mentioned.
- peroxy ester examples include ⁇ -cumyl peroxyneodecanoate, t-butylperoxyneodecanoate, t-butylperoxypivalate, 2,2,4-trimethylpentylperoxy-2-ethylhexanoate, and t-.
- peroxydicarbonate examples include di-3-methoxyperoxydicarbonate, di (2-ethylhexyl) peroxydicarbonate, bis (4-t-butylcyclohexyl) peroxydicarbonate, diisopropylperoxydicarbonate, and di-n-propylperoxydi. Examples thereof include carbonate, di (2-ethoxyethyl) peroxydicarbonate, and diallylperoxydicarbonate.
- diacyl peroxide is preferably used from the viewpoint of overall balance of safety, storage stability and radical generation ability, and among them, benzoyl peroxide is more preferably used.
- the content of the polymerization initiator (C) in the dental curable composition of the present invention is not particularly limited, but the polymerizable monomer (A) can be obtained from the viewpoint of curability of the obtained dental curable composition. It is preferably 0.001 part by mass or more, more preferably 0.01 part by mass or more, further preferably 0.02 part by mass or more, and 0.1. It is particularly preferable that the content is by mass or more. Further, if the content of the polymerization initiator (C) is too large, precipitation from the dental curable composition may occur. Therefore, the content is 100% by mass in total of the polymerizable monomer (A). It is preferably 30 parts by mass or less, more preferably 20 parts by mass or less, further preferably 15 parts by mass or less, particularly preferably 10 parts by mass or less, and 5 parts by mass. It may be 2 parts or less and 2 parts by mass or less.
- the dental curable composition of the present invention contains a colorant (D).
- the type of the colorant (D) and the like are not particularly limited, and any of the inorganic pigment and / or the organic pigment can be used without limitation depending on the color tone of the target dental curable composition.
- the shape of the colored particles is not particularly limited and may be any particle shape such as spherical, needle-shaped, plate-shaped, crushed-shaped, scaly-shaped, etc., and can be used without limitation.
- inorganic pigments include chromate such as chlorophyll, zinc yellow, and barium yellow; ferrocyanides such as navy blue; sulfides such as silver vermilion, cadmium yellow, zinc sulfide, and cadmium red; barium sulfate, zinc sulfate. , Sulfates such as strontium sulfate; oxides such as zinc flower, titanium white, antimony white, red iron oxide, iron black, chromium oxide; hydroxides such as aluminum hydroxide; silicates such as calcium silicate and ultramarine; carbon Examples include carbon such as black and graphite.
- organic pigments include nitroso pigments such as naphthol green B and naphthol green Y; nitro pigments such as naphthol yellow S and resole fast yellow 2G, permanent red 4R, brilliant fast scarlet, hanza yellow, benzine yellow and the like.
- Insoluble azo pigments Insoluble azo pigments; sparingly soluble azo pigments such as resole red, lake red C, lake red D; soluble azo pigments such as Brilliant Carmine 6B, Permanent Red F5R, Pigment Scarlet 3B, Bordeaux 10B; , Sky blue and other phthalocyanine pigments; Rhodamine lakes, malakite green lakes, methyl violet lakes and other basic dye pigments; Peacock blue lakes, eosin lakes, quinoline yellow lakes, aluminum lakes and other acidic dye pigments. ..
- One of these colorants (D) may be used alone, or two or more thereof may be used in combination.
- titanium white, red iron oxide, iron black, yellow iron oxide and the like which are inorganic pigments having excellent heat resistance and light resistance as compared with organic pigments, are preferable.
- the content of the colorant (D) in the dental curable composition of the present invention is not limited as long as it is within the range in which the effect of the present invention is exhibited, but from the viewpoint of aesthetics, the polymerizable monomer (A) 100 It is preferably 0.0005 parts by mass or more, more preferably 0.002 parts by mass or more, further preferably 0.004 parts by mass or more, and 0.006 parts by mass or more with respect to parts by mass. Is particularly preferable. When it is at least the above lower limit, it is possible to effectively suppress the impression that the filled portion is dark and sunk. Further, it is preferably 0.5 parts by mass or less, more preferably 0.4 parts by mass or less, and further preferably 0.3 parts by mass or less.
- the content of the colorant (D) is preferably 0.00001 parts by mass or more, more preferably 0.0001 or more, and 0.0005 with respect to 100 parts by mass of the dental curable composition. It is more preferably parts by mass or more, and particularly preferably 0.001 part by mass or more.
- the content of the colorant (D) is preferably 0.3 parts by mass or less, more preferably 0.1 parts by mass or less, and further preferably 0.07 parts by mass or less.
- the dental curable composition of the present invention may further contain a polymerization accelerator (E).
- a polymerization accelerator examples include amines, sulfinic acid and salts thereof, borate compounds, barbituric acid derivatives, triazine compounds, copper compounds, tin compounds, vanadium compounds, halogen compounds, aldehydes, thiol compounds, sulfites, and bisulfites. , Thiourea compounds and the like.
- the polymerization accelerator (E) one type may be used alone, or two or more types may be used in combination.
- Amines are divided into aliphatic amines and aromatic amines.
- the aliphatic amine include primary aliphatic amines such as n-butylamine, n-hexylamine and n-octylamine; and secondary aliphatic amines such as diisopropylamine, dibutylamine and N-methylethanolamine; N-Methyldiethanolamine, N-ethyldiethanolamine, Nn-butyldiethanolamine, N-lauryldiethanolamine, 2- (dimethylamino) ethylmethacrylate, N-methyldiethanolaminedimethacrylate, N-ethyldiethanolaminedimethacrylate, triethanolamine monomethacrylate , Triethanolamine dimethacrylate, triethanolamine trimethacrylate, triethanolamine, trimethylamine, triethylamine, tributylamine and other tertiary aliphatic amines.
- aromatic amine examples include N, N-bis (2-hydroxyethyl) -3,5-dimethylaniline, N, N-bis (2-hydroxyethyl) -p-toluidine, and N, N-bis. (2-Hydroxyethyl) -3,4-dimethylaniline, N, N-bis (2-hydroxyethyl) -4-ethylaniline, N, N-bis (2-hydroxyethyl) -4-isopropylaniline, N, N-bis (2-hydroxyethyl) -4-t-butylaniline, N, N-bis (2-hydroxyethyl) -3,5-di-isopropylaniline, N, N-bis (2-hydroxyethyl)- 3,5-Di-t-butylaniline, N, N-dimethylaniline, N, N-dimethyl-p-toluidine, N, N-dimethyl-m-toluidine, N, N-die
- sulfinic acid and salts thereof include p-toluenesulfinic acid, sodium p-toluenesulfinate, potassium p-toluenesulfinate, lithium p-toluenesulfinate, calcium p-toluenesulfinate, benzenesulfinic acid, and benzenesulfin.
- an aryl borate compound is preferable.
- a borate compound having one aryl group in one molecule a borate compound having two aryl groups in one molecule, a borate compound having three aryl groups in one molecule, and one molecule.
- examples thereof include a borate compound having 4 aryl groups, and from the viewpoint of storage stability, a borate compound having 3 or 4 aryl groups in one molecule is preferable.
- borate compound having one aryl group in one molecule examples include trialkylphenyl boron, trialkyl (p-chlorophenyl) boron, trialkyl (p-fluorophenyl) boron, and trialkyl [3,5-bis.
- At least one selected from these) and salts thereof (sodium salt, lithium salt, potassium salt, magnesium salt, tetrabutylammonium salt, tetramethylammonium salt, tetraethylammonium salt, methylpyridinium salt, ethylpyridinium salt, butyl).
- salts thereof sodium salt, lithium salt, potassium salt, magnesium salt, tetrabutylammonium salt, tetramethylammonium salt, tetraethylammonium salt, methylpyridinium salt, ethylpyridinium salt, butyl.
- Pyridinium salt, methyl quinolinium salt, ethyl quinolinium salt, butyl quinolinium salt, etc. can be mentioned.
- borate compound having two aryl groups in one molecule examples include dialkyldiphenylboron, dialkyldi (p-chlorophenyl) boron, dialkyldi (p-fluorophenyl) boron, and dialkyldi [3,5-bis (trifluoromethyl).
- Phenyl] boron dialkyldi [3,5-bis (1,1,1,3,3,3-hexafluoro-2-methoxy-2-propyl) phenyl] boron, dialkyldi (p-nitrophenyl) boron, dialkyldi (M-Nitrophenyl) boron, dialkyldi (p-butylphenyl) boron, dialkyldi (m-butylphenyl) boron, dialkyldi (p-butyloxyphenyl) boron, dialkyldi (m-butyloxyphenyl) boron, dialkyldi (p-) Octyloxyphenyl) boron, dialkyldi (m-octyloxyphenyl) boron (alkyl groups in each of the above examples are n-butyl group, n-octyl group, n-dodecyl group, etc.),
- Salt potassium salt, magnesium salt, tetrabutylammonium salt, tetramethylammonium salt, tetraethylammonium salt, methylpyridinium salt, ethylpyridinium salt, butylpyridinium salt, methylquinolinium salt, ethylquinolinium salt, butylquinolinium Salt, etc.).
- borate compound having three aryl groups in one molecule examples include monoalkyltriphenylboron, monoalkyltri (p-chlorophenyl) boron, monoalkyltri (p-fluorophenyl) boron, and monoalkyltri [3].
- 5-bis (trifluoromethyl) phenyl] boron monoalkyltri [3,5-bis (1,1,1,3,3,3-hexafluoro-2-methoxy-2-propyl) phenyl] boron, Monoalkyltri (p-nitrophenyl) boron, monoalkyltri (m-nitrophenyl) boron, monoalkyltri (p-butylphenyl) boron, monoalkyltri (m-butylphenyl) boron, monoalkyltri (p- Butyloxyphenyl) boron, monoalkyltri (m-butyloxyphenyl) boron, monoalkyltri (p-octyloxyphenyl) boron and monoalkyltri (m-octyloxyphenyl) boron (alkyl group is n-butyl group, One selected from n-octyl group, n-dodecyl
- borate compound having four aryl groups in one molecule examples include tetraphenyl boron, tetrakis (p-chlorophenyl) boron, tetrakis (p-fluorophenyl) boron, and tetrakis [3,5-bis (trifluoromethyl).
- barbituric acid derivatives include barbituric acid, 1,3-dimethylbarbituric acid, 1,3-diphenylbarbituric acid, 1,5-dimethylbarbituric acid, 5-butylbarbituric acid, and 5-ethylbarbituric acid.
- barbituric acid derivatives include 5-butylbarbituric acid, 1,3,5-trimethylbarbituric acid, 1-cyclohexyl-5-ethylbarbituric acid, 1-benzyl-5-phenylbarbituric acid, and the like. And these sodium salts.
- triazine compound examples include 2,4,6-tris (trichloromethyl) -s-triazine, 2,4,6-tris (tribromomethyl) -s-triazine and 2-methyl-4,6-bis ( Trichloromethyl) -s-triazine, 2-methyl-4,6-bis (tribromomethyl) -s-triazine, 2-phenyl-4,6-bis (trichloromethyl) -s-triazine, 2- (p-) Methoxyphenyl) -4,6-bis (trichloromethyl) -s-triazine, 2- (p-methylthiophenyl) -4,6-bis (trichloromethyl) -s-triazine, 2- (p-chlorophenyl) -4 , 6-bis (trichloromethyl) -s-triazine, 2- (2,4-dichlorophenyl) -4,6-bis (trichloromethyl) -s-tria
- 2,4,6-tris (trichloromethyl) -s-triazine is preferable in terms of polymerization activity, and 2-phenyl-4, in terms of storage stability. 6-bis (trichloromethyl) -s-triazine, 2- (p-chlorophenyl) -4,6-bis (trichloromethyl) -s-triazine, and 2- (4-biphenylyl) -4,6-bis (trichloro) Methyl) -s-triazine.
- the above triazine compound may be used alone or in combination of two or more.
- the copper compound for example, acetylacetone copper, copper acetate (II), copper oleate, copper chloride (II), copper bromide (II) and the like are preferably used.
- tin compound examples include di-n-butyl tin dilaurate, di-n-octyl tin dimareate, di-n-octyl tin dilaurate, and di-n-butyl tin dilaurate.
- Particularly suitable tin compounds are di-n-octyl tin dilaurate and di-n-butyl tin dilaurate.
- the vanadium compound is preferably an IV-valent and / or V-valent vanadium compound.
- IV-valent and / or V-valent vanadium compounds include divanadium tetraoxide (IV), vanadium oxide acetylacetonate (IV), vanadil oxalate (IV), vanadil sulfate (IV), and oxobis (1-).
- Japanese Patent Laid-Open No. 2003 such as phenyl-1,3-butangionate) vanadium (IV), bis (maltrat) oxovanadium (IV), vanadium pentoxide (V), sodium metavanadate (V), ammon metavanadate (V). Examples thereof include the compounds described in Japanese Patent Publication No. 96122.
- halogen compound for example, dilauryldimethylammonium chloride, lauryldimethylbenzylammonium chloride, benzyltrimethylammonium chloride, tetramethylammonium chloride, benzyldimethylcetylammonium chloride, dilauryldimethylammonium bromide and the like are preferably used.
- aldehydes include terephthalaldehyde and benzaldehyde derivatives.
- the benzaldehyde derivative include dimethylaminobenzaldehyde, p-methoxybenzaldehyde, p-ethoxybenzaldehyde, pn-octyloxybenzaldehyde and the like. Among them, pn-octyloxybenzaldehyde is preferably used from the viewpoint of curability.
- thiol compound examples include 3-mercaptopropyltrimethoxysilane, 2-mercaptobenzoxazole, decanethiol, and thiobenzoic acid.
- sulfite salt examples include sodium sulfite, potassium sulfite, calcium sulfite, ammonium sulfite and the like.
- Examples of the hydrogen sulfite salt include sodium bisulfite and potassium hydrogen sulfite.
- thiourea compound examples include 1- (2-pyridyl) -2-thiourea, thiourea, methylthiourea, ethylthiourea, N, N'-dimethylthiourea, N, N'-diethylthiourea, N, N'-di.
- N N'-dicyclohexylthiourea, trimethylthiourea, triethylthiourea, tri-n-propylthiourea, tricyclohexylthiourea, tetramethylthiourea, tetraethylthiourea, tetra-n-propylthiourea, Examples thereof include tetracyclohexylthiourea.
- the content thereof is not particularly limited, but from the viewpoint of the curability of the obtained dental curable composition, a single polymerizable mass. It is preferably 0.001 part by mass or more, more preferably 0.01 part by mass or more, and further preferably 0.02 part by mass or more with respect to a total of 100 parts by mass of the body (A). , 0.03 parts by mass or more, 0.05 parts by mass or more, and further 0.1 parts by mass or more. Further, if the content of the polymerization accelerator (E) is too large, precipitation may occur in the dental curable composition. Therefore, the content of the polymerization accelerator (E) is the polymerizable monomer (A).
- the dental curable composition of the present invention is other than the above-mentioned polymerizable monomer (A), filler (B), polymerization initiator (C), colorant (D), and polymerization accelerator (E). Further, if necessary, an additive (F) such as a polymerization inhibitor, a chain transfer agent, an ultraviolet absorber, an antioxidant, an antibacterial agent, a dispersant, and a pH adjuster may be further contained. As the additive (F), one type may be used alone, or two or more types may be used in combination.
- polymerization inhibitor examples include 3,5-di-t-butyl-4-hydroxytoluene, hydroquinone, dibutylhydroquinone, dibutylhydroquinone monomethyl ether, hydroquinone monomethyl ether, 2,6-di-t-butylphenol and the like. , These may be used alone or in combination of two or more.
- UV absorber examples include 2- (2-hydroxyphenyl) benzotriazole, 2- (2-hydroxy-5-methylphenyl) benzotriazole, 2- (2-hydroxy-5-ethylphenyl) benzotriazole, 2 -(2-Hydroxy-5-propylphenyl) benzotriazole, 2- (2-hydroxy-5-tert-octylphenyl) benzotriazole, 2- (2-hydroxy-3-tert-butyl-5-methylphenyl)- Examples thereof include benzotriazole compounds such as 5-chloro-2H-benzotriazole (tinubin 326), benzomidazole compounds and the like, and these may be used alone or in combination of two or more.
- benzotriazole compounds such as 5-chloro-2H-benzotriazole (tinubin 326), benzomidazole compounds and the like, and these may be used alone or in combination of two or more.
- the dental curable composition of the present invention is a dental curable composition having a composition of one type for cases in which a cavity bottom is present, such as a class I cavity, a class II cavity, or a class V cavity.
- the cured product has excellent easy-to-polishability and slip durability.
- an embodiment in which the slipping durability is particularly emphasized may be selected.
- the color tone compatibility ( ⁇ E * represented by the following formula) for a class I cavity is a shade guide (trade name “Vita Classical”) from the viewpoint of particularly placing importance on smoothness durability.
- a dental curable composition that is 6.0 or less for both shades A1 and A4 of "Shade Guide", manufactured by VITA (VITA Zahnfabrik, Germany), and has a slipping durability of 40% or more. Can be mentioned.
- ⁇ E * ((L * 1 -L * 0 ) 2 + (a * 1 -a * 0 ) 2 + (b * 1 -b * 0 ) 2 ) 1/2 (In the formula, the explanation of the symbols is as described in the examples described later.)
- the method for measuring the color tone compatibility with the class I cavity and the method for measuring the slip resistance are as described in the examples described later.
- the dental curable composition of the present invention exhibits good color compatibility with a wide range of natural teeth with only one composition without changing the composition. Therefore, in a certain embodiment, the color tone compatibility ( ⁇ E *) is excellent for both A1 and A4 of the shade guide for the case where the class I cavity is present.
- the dental curable composition according to the embodiment (Y-1) can be appropriately modified based on the description of the present specification.
- the composition of the dental curable composition according to the embodiment (Y-1) is not particularly limited as long as the color tone compatibility with the class I cavity and the slip resistance are within a predetermined range.
- the dental curable composition in the embodiment (Y-1) preferably contains the polymerizable monomer (A).
- the dental curable composition in the embodiment (Y-1) preferably contains a filler (B).
- the dental curable composition in the embodiment (Y-1) contains a polymerization initiator (C).
- the dental curable composition in the embodiment (Y-1) preferably contains a colorant (D).
- the dental curable composition in the embodiment (Y-1) may not contain the colorant (D).
- the type of polymerization initiator (C) for example, (bis) acylphosphine oxides and ⁇ -diketones used in combination as a photopolymerization initiator
- the type of filler (B), size such as average particle size, shape (for example, amorphous filler, etc.), manufacturing method (for example, surface treatment with surface treatment agent; heat treatment, etc.), etc. are appropriately combined. Since the spectral reflectance ratios R 650/600 , R 700/600 , and R 750/600 are all within the above-mentioned predetermined ranges, the composition is not changed and only one composition is used.
- the dental curable composition of the present invention also includes those showing good color compatibility with natural teeth having a wide range of colors and having excellent smoothing durability.
- the sliding durability is preferably 55% or more, more preferably 60% or more, still more preferably 65% or more.
- the shade compatibility ( ⁇ E *) with respect to the class I cavity is shade guide (trade name “Vita classical shade guide”, manufactured by VITA (VITA Zhanfabrik, Germany)) shades A1 and A4. It is preferably 5.5 or less, more preferably 5.0 or less, and even more preferably 4.5 or less.
- the method for preparing the dental curable composition of the present invention is not particularly limited, and can be obtained by blending each component in a predetermined blending amount. At this time, the blending order is not particularly limited, and each component may be blended together or may be blended in two or more times. Further, if necessary, they may be mixed or kneaded, or may be subjected to a defoaming treatment such as a vacuum defoaming treatment.
- the obtained dental curable composition can be filled in a single container (syringe or the like) to obtain a one-material type (one-paste type) dental curable composition.
- the use of the dental curable composition of the present invention is not particularly limited and can be used as various dental materials.
- a dental composite resin for example, a composite resin for filling a carious cavity, a composite for abutment construction
- a composite resin for filling a carious cavity for example, a composite resin for filling a carious cavity, a composite for abutment construction
- Resin composite resin for crown, self-adhesive composite resin, etc.
- resin for denture base lining material for denture base, impression material, joint wearing material (for example, resin cement, resin-added glass ionomer cement, etc.), dentistry
- an adhesive material for dentures for example, an adhesive material for orthodontics, an adhesive material for cavity coating, etc.
- a tooth fissure groove sealing material for example, a resin block for CAD / CAM, a temporary crown, an artificial tooth material, and the like.
- the dental curable composition of the present invention is particularly preferable to be used as a dental composite resin and a CAD / CAM resin block because it has high aesthetics and excellent mechanical strength.
- the present invention includes embodiments in which the above configurations are variously combined within the scope of the technical idea of the present invention as long as the effects of the present invention are exhibited.
- refractive index of filler For the following fillers, various refractive indexes are obtained by mixing two or more organic solvents selected from the group consisting of organic solvents having a known refractive index (1-bromonaphthalene, methyl salicylate, dimethylformamide, and 1-pentanol). Disperse each filler in a fixed amount (0.1 g / mL) to prepare a dispersion, and determine the refractive index (nD25) of the organic solvent that maximizes the light transmittance of the dispersion at 589 nm. , This refractive index was taken as the refractive index of the filler.
- the type of organic solvent having a known refractive index can be selected based on the assumed refractive index according to the type of the filler to which the refractive index is to be measured.
- the refractive index of the organic solvent was measured with a Na-D light source using an Abbe refractometer (manufactured by Atago Co., Ltd., trade name: NAR-1T LIQUID), and the light transmittance of the dispersion was measured by an ultraviolet-visible spectrophotometer. It was measured in a quartz cell having an optical path length of 10 mm using (trade name: UV-2400, manufactured by Shimadzu Corporation).
- the refractive index of the polymer of the polymerizable monomer was measured in a constant temperature room at 25 ° C. using the above Abbe refractometer. Specifically, in each of the Examples and Comparative Examples of Table 1, the polymerizable monomer (A), the polymerization initiator (C), the polymerization accelerator (E), and the additive (F) are shown in Table 1. A mixture containing a uniform polymerizable monomer mixed in the above compounding ratio was placed in a mold having a hole of ⁇ 10 mm ⁇ 1.0 mm, and slide glass was pressure-welded on both sides.
- one side was irradiated with light for 45 seconds on each side with an LED photopolymerizer ( ⁇ -light V, manufactured by Morita Seisakusho Co., Ltd., wavelength: 400 to 408 nm, 465 to 475 nm) to cure, and then taken out from the mold.
- a cured product of a mixture containing a polymerizable monomer was prepared.
- a solvent (1-bromonaphthalene) that does not dissolve the sample and has a higher refractive index than the sample is dropped onto the sample for the purpose of bringing the cured product into close contact with the measurement surface. It was measured.
- the inorganic fillers obtained in each of the following production examples were vacuum devolatile at 100 ° C. for 2 hours using a specific surface area measuring device (“BELSORP-mini II” manufactured by Microtrac Bell Co., Ltd.), and then adsorbed gas:
- the analysis by the BET multipoint method used was adopted.
- the light diffusivity LD of the cured product was evaluated using a goniometer (Murakami Color Technology Laboratory Co., Ltd., GP-200). Specifically, the LED photopolymerizer ( ⁇ -light V, Morita Co., Ltd.) was placed in a state where the dental curable composition was sandwiched between cover glass on the top and bottom using a spacer made of stainless steel having a thickness of 0.25 mm and pressed against each other. A cured plate having a diameter of 30 mm and a thickness of 0.25 mm was produced by irradiating both sides with light for 45 seconds on each side at a wavelength of 400 to 408 nm and 465 to 475 nm manufactured by Mfg. Co., Ltd.
- the composition was filled into a plastic simulated tooth cavity (a hole having a diameter of 3 mm and a depth of 2 mm imitating a fifth-class tooth cavity) using a metal filling device at 25 ° C., and the ease of filling operation was evaluated.
- a metal dental filling device manufactured by Yoshida Co., Ltd., kneading fillers # 4 and # 5.
- the dental curable composition is liquid, the dental curable composition is filled in a syringe container (Clearfil Majesty ES Flow syringe container manufactured by Clarenoritake Dental Co., Ltd.), and the attached needle tip is used.
- the dental curable composition was extruded from the syringe container to which the dental curable composition was attached to fill the simulated cavity, and the stringing of the dental curable composition at that time was evaluated according to the following criteria.
- ⁇ No stickiness or stringiness
- ⁇ Slightly sticky or slightly stringy
- the color tone compatibility of the molar portion of the dental curable composition was evaluated by filling and repairing the tooth cavity formed in the artificial tooth and measuring the portion thereof with a dental color measuring device. Specifically, first, the artificial tooth No. 6 (Zen opal tooth, size: PL16, shade: A1, A3, and A4, manufactured by GC Corporation) is used as a dental colorimeter (Crystaleye Spectrophotometer, manufactured by Olympus Corporation). Was taken using. At this time, the artificial tooth was measured in the dark box (check box web cover) attached to the dental color measuring device.
- a class I cavity with a diameter of 4 mm and a depth of 2 mm was formed in the center of the artificial tooth, and a dental etching material (K Echanto GEL, manufactured by Kuraray Noritake Dental Co., Ltd.) was used, and the method recommended by the operator (in the package insert).
- the cavity surface was etched by the method described).
- the cavity surface is bonded by the method recommended by the operator, and a dental visible light irradiator (Pencure 2000, Pencure 2000, Using Morita Seisakusho Co., Ltd.), light irradiation was performed for 10 seconds in normal mode.
- the tooth cavity is filled with a dental curable composition and pressed with an artificial tooth mold prepared in advance with a silicone impression material (Memosil 2, manufactured by Heraeus Kurzer Japan Co., Ltd.) to give the surface shape of the artificial tooth. Shaped.
- the rotation speed is approximately 10,000 rpm.
- the filling part was polished for 30 seconds.
- the dental curable composition is irradiated with light from the top of the mold for 10 seconds in the normal mode, then the mold is removed and the mold is irradiated with the light for 10 seconds in the normal mode. Curing was performed to prepare a filled sample. The prepared sample was photographed by the same method as that photographed at the beginning.
- each ⁇ E * is preferably 6.0 or less, more preferably 5.5 or less, still more preferably 5.0 or less.
- the color tone compatibility of the anterior tooth portion of the dental curable composition was evaluated by filling and repairing the tooth cavity formed in the shade guide and measuring the color of the portion with a dental color measuring device. Specifically, first, a shade guide (Vita classical shade guide, shades: A1, A3, and A4, manufactured by VITA (VITA Zahnfabrik, Germany)) is used in the same manner using the above-mentioned dental color measuring device. I took a picture. Subsequently, a class IV cavity penetrating from the lingual side to the labial side of a ⁇ 4 mm quadrant was formed at the cut edge of the shade guide, and then a filling sample was prepared by the same method as described above.
- ⁇ E * is preferably 7.0 or less, more preferably 6.0 or less, further preferably 5.0 or less, and A1, A3, respectively. It is particularly preferable that both ⁇ E * are 6.0 or less with respect to the shade guides of A4 and A4.
- the glossiness is preferably 20% or more, more preferably 25% or more, still more preferably 30% or more.
- the smoothness durability was evaluated based on the change in glossiness before and after the wear resistance test. Specifically, it is as follows.
- the dental curable composition (paste) was filled in a SUS mold (length 30 mm ⁇ width 20 mm ⁇ thickness 2 mm), and the upper and lower sides (30 mm ⁇ 20 mm surface) of the paste were pressed with a slide glass.
- the paste was cured by irradiating the front and back of the paste with light for 180 seconds each through a slide glass with a visible light irradiator for dental technicians ( ⁇ -light V, manufactured by Morita Co., Ltd.).
- ⁇ -light V manufactured by Morita Co., Ltd.
- the test piece was used with a toothbrush wear tester (manufactured by Daiei Kagaku Seiki Seisakusho Co., Ltd.) using a suspension of dentifrice and a commercially available toothbrush (bitine, size: regular, hardness: normal, manufactured by Lion Corporation).
- a wear resistance test of 40,000 cycles was performed under a load of 250 g.
- the suspension of the dentifrice was prepared by using a commercially available dentifrice (Denter Clearmax, manufactured by Lion Corporation) at a mass ratio of dentifrice / distilled water of 10/90.
- the glossiness (G2) of the surface of the test piece after the wear resistance test was determined by the same method as before the wear resistance test.
- the sliding durability was preferably 40% or more, more preferably 45% or more, further preferably 50% or more, and 55% or more, 60% or more, 65% or more. Especially preferable.
- Filler (B) As the filler, the one obtained in the following production example was used. Moreover, the commercial product was used as it was.
- the obtained inorganic agglomerated particles (BF-2e-1) had an average particle diameter of 5.7 ⁇ m, a refractive index of 1.53, and a specific surface area of 95.8 m 2 / g.
- the silane-treated light-diffusible inorganic agglomerated particles (BF-2d-1) have an average particle size of 1.6 ⁇ m, a refractive index of 1.44, a specific surface area of 99 m 2 / g, and a pore volume of 0.19 mL / g. there were.
- the composition was polymerized by heating at 130 ° C. for 3 hours under reduced pressure, and the obtained cured product was further pulverized with a ball mill to obtain a surface-untreated organic-inorganic composite filler. Further, 100 parts by mass of the surface-untreated organic-inorganic composite filler was surface-treated with 1 part by mass of ⁇ -methacryloxypropyltrimethoxysilane to obtain a light-diffusing organic-inorganic composite filler (BC-d-1). ..
- the light diffusible organic-inorganic composite filler (BC-d-1) had an average particle size of 11 ⁇ m and a refractive index of 1.50.
- the composition was polymerized by heating at 130 ° C. for 3 hours under reduced pressure, and the obtained cured product was further pulverized with a ball mill to obtain a surface-untreated organic-inorganic composite filler. Further, 100 parts by mass of the surface-untreated organic-inorganic composite filler was surface-treated with 1 part by mass of ⁇ -methacryloxypropyltrimethoxysilane to obtain a light-diffusing organic-inorganic composite filler (BC-d-2). ..
- the light diffusible organic-inorganic composite filler (BC-d-2) had an average particle size of 15 ⁇ m and a refractive index of 1.49.
- TIN Chinubin 326
- BASF Japan Ltd. ultraviolet absorber
- BHT 3,5-di-t-butyl-4-hydroxytoluene (polymerization inhibitor)
- Example 1 to 10 and Comparative Examples 1 to 5 (dental composite resin)
- the dental curable composition was prepared by mixing and kneading the materials and ratios shown in Table 1 at room temperature (23 ° C.) and in a dark place, and vacuum-defoaming the uniformed material. Each test was performed on these dental curable compositions by the method described above. The results are shown in Table 2.
- the dental curable composition of the present invention is one type of dental curable composition for the bottom of a class I cavity and is good for natural teeth of a wide range of tones. Shows color compatibility. Further, among the dental curable compositions of the present invention, Example 1 and the like have light diffusivity and therefore have high color tone compatibility with class IV cavities, and also contain a specific filler (B). It can be seen that it has good sliding durability. It can be seen that Comparative Examples 1 to 5 are inferior in color tone compatibility because the ratio of the spectral reflectance is not within the preferable range.
- the dental curable composition of the present invention exhibits good color compatibility with a wide range of natural teeth with only one composition, has good slip durability, and is a dental composite resin or the like. It is preferably used as.
Landscapes
- Health & Medical Sciences (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Plastic & Reconstructive Surgery (AREA)
- Dental Preparations (AREA)
Abstract
Description
[1]重合性単量体(A)、充填材(B)、重合開始剤(C)、及び着色剤(D)を含む歯科用硬化性組成物であって、
前記歯科用硬化性組成物の厚さ1.0mmの硬化物を、分光色差計を用いて白背景で測定した際の、600nmの波長における分光反射率に対する、650nm、700nm、及び750nmの波長における分光反射率の比R650/600、R700/600、及びR750/600がいずれも、97%~103%の範囲内である、歯科用硬化性組成物。
[2]厚さ1.0mmの硬化物における、以下の式(1)で定義されるコントラスト比が、0.35~0.65を満たす、[1]に記載の歯科用硬化性組成物。
コントラスト比=Yb/Yw (1)
(ここで、Ybは黒背景において測定したXYZ表色系のY値を表し、Ywは白背景において測定したXYZ表色系のY値を表す。)
[3]厚さ0.25mmの硬化物における、以下の式(2)で定義される光拡散度LDが、0.0001~0.99を満たす、[1]又は[2]に記載の歯科用硬化性組成物。
LD=(I5/cos5°)/I0 (2)
(ここで、Iは硬化物を透過した光の光度を表し、I0、及びI5は試料板に垂直な方向(光の入射方向)に対し、それぞれ0度、及び5度傾いた方向の透過光の光度を表す。)
[4]厚さ1.0mmの硬化物における、硬化物の背後に標準白色板を置いて測定した際のL*a*b*表色系における色度指数であるa*/wが、-3.0~2.0である、[1]~[3]のいずれかに記載の歯科用硬化性組成物。
[5]前記充填材(B)が、平均粒子径が0.05~1μmである無機微粒子(BF-1)を含む、[1]~[4]のいずれかに記載の歯科用硬化性組成物。
[6]前記充填材(B)が、無機一次粒子(x)が凝集してなる無機凝集粒子(BF-2)を含み、前記無機一次粒子(x)の平均粒子径が0.001~1μmである、[1]~[5]のいずれかに記載の歯科用硬化性組成物。
[7]前記無機凝集粒子(BF-2)が、光拡散性無機凝集粒子(BF-2d)を含み、前記光拡散性無機凝集粒子(BF-2d)の屈折率が下記式(3)を満たす、[6]に記載の歯科用硬化性組成物。
0.03<|nP-nFBF-2d|<1.0 (3)
(ここで、nPは、前記重合性単量体(A)を重合して得られる重合体の屈折率、nFBF-2dは、前記光拡散性無機凝集粒子(BF-2d)の屈折率を表す。)
[8]前記充填材(B)が、無機一次粒子(x)を含有する有機無機複合充填材(BC)を含み、前記無機一次粒子(x)の平均粒子径が0.001~1μmである、[1]~[7]のいずれかに記載の歯科用硬化性組成物。
[9]前記有機無機複合充填材(BC)が、光拡散性有機無機複合充填材(BC-d)を含み、前記光拡散性有機無機複合充填材(BC-d)の屈折率が下記式(7)を満たす、[8]に記載の歯科用硬化性組成物。
0.03<|nP-nFBC-d|<1.0 (7)
(ここで、nPは、前記重合性単量体(A)を重合して得られる重合体の屈折率、nFBC-dは、前記光拡散性有機無機複合充填材(BC-d)の屈折率を表す。)
コントラスト比=Yb/Yw (1)
コントラスト比は透明性の指標であり、値が1に近いほど不透明な材料で、0近いほど透明な材料であることを表す。
LD=(I5/cos5°)/I0 (2)
(ここで、Iは硬化物を透過した光の光度を表し、I0、及びI5は試料板に垂直な方向(光の入射方向)に対し、それぞれ0度、及び5度傾いた方向の透過光の光度を表す。)
本発明の歯科用硬化性組成物における重合性単量体(A)としては、歯科用硬化性組成物に使用される重合性単量体として公知のものを用いることができ、特にラジカル重合性単量体を好ましく用いることができる。当該ラジカル重合性単量体としては、例えば、α-シアノアクリル酸、(メタ)アクリル酸、α-ハロゲン化アクリル酸、クロトン酸、桂皮酸、ソルビン酸、マレイン酸、イタコン酸等の不飽和カルボン酸のエステル;(メタ)アクリルアミド;(メタ)アクリルアミド誘導体;ビニルエステル類;ビニルエーテル類;モノ-N-ビニル誘導体;スチレン誘導体などが挙げられる。重合性単量体(A)は、1種を単独で使用してもよいし、2種以上を併用してもよい。これらの中でも、不飽和カルボン酸のエステル、(メタ)アクリルアミド誘導体が好ましく、(メタ)アクリル酸エステル、(メタ)アクリルアミド誘導体がより好ましく、(メタ)アクリル酸エステルがさらに好ましい。なお、本明細書において「(メタ)アクリル」との表記は、メタクリルとアクリルの両者を包含する意味で用いられる。また、「(メタ)アクリル系単量体」は、(メタ)アクリル酸エステルと(メタ)アクリルアミド誘導体の両者を包含する意味で用いられる。(メタ)アクリル酸エステル及び(メタ)アクリルアミド誘導体の例を以下に示す。
例えば、メチル(メタ)アクリレート、イソブチル(メタ)アクリレート、ベンジル(メタ)アクリレート、ドデシル(メタ)アクリレート、2-(N,N-ジメチルアミノ)エチル(メタ)アクリレート、2,3-ジブロモプロピル(メタ)アクリレート、3-(メタ)アクリロイルオキシプロピルトリメトキシシラン、11-(メタ)アクリロイルオキシウンデシルトリメトキシシラン、2-ヒドロキシエチル(メタ)アクリレート、3-ヒドロキシプロピル(メタ)アクリレート、4-ヒドロキシブチル(メタ)アクリレート、6-ヒドロキシヘキシル(メタ)アクリレート、10-ヒドロキシデシル(メタ)アクリレート、プロピレングリコールモノ(メタ)アクリレート、グリセリンモノ(メタ)アクリレート、エリスリトールモノ(メタ)アクリレート、フェノキシエチレングリコール(メタ)アクリレート、イソボルニル(メタ)アクリレート、3-フェノキシベンジル(メタ)アクリレート、N-メチロール(メタ)アクリルアミド、N-ヒドロキシエチル(メタ)アクリルアミド、N,N-ビス(ヒドロキシエチル)(メタ)アクリルアミド、N,N-ジメチル(メタ)アクリルアミド、N,N-ジエチル(メタ)アクリルアミド、N,N-ジ-n-プロピル(メタ)アクリルアミド、N-エチル-N-メチル(メタ)アクリルアミド、(メタ)アクリロイルモルホリン、(メタ)アクリロイルオキシドデシルピリジニウムブロミド、(メタ)アクリロイルオキシドデシルピリジニウムクロリド、(メタ)アクリロイルオキシヘキサデシルピリジニウムブロミド、(メタ)アクリロイルオキシヘキサデシルピリジニウムクロリド、エトキシ化-o-フェニルフェノール(メタ)アクリレート、エトキシ化-m-フェニルフェノール(メタ)アクリレート、エトキシ化-p-フェニルフェノール(メタ)アクリレート、プロポキシ化-o-フェニルフェノール(メタ)アクリレート、プロポキシ化-m-フェニルフェノール(メタ)アクリレート、プロポキシ化-p-フェニルフェノール(メタ)アクリレート、o-フェノキシベンジル(メタ)アクリレート、m-フェノキシベンジル(メタ)アクリレート、p-フェノキシベンジル(メタ)アクリレート、2-(o-フェノキシフェニル)エチル(メタ)アクリレート、2-(m-フェノキシフェニル)エチル(メタ)アクリレート、2-(p-フェノキシフェニル)エチル(メタ)アクリレートなどが挙げられる。これらの中でも、得られる歯科用硬化性組成物のペーストの操作性が良く、硬化後の機械的強度に優れる点で、エトキシ化-o-フェニルフェノール(メタ)アクリレート、及びm-フェノキシベンジル(メタ)アクリレートが最も好ましい。
例えば、芳香族系の二官能性の(メタ)アクリル酸エステル、脂肪族系の二官能性の(メタ)アクリル酸エステルなどが挙げられる。
例えば、トリメチロールプロパントリ(メタ)アクリレート、トリメチロールエタントリ(メタ)アクリレート、トリメチロールメタントリ(メタ)アクリレート、ペンタエリスリトールトリ(メタ)アクリレート、ペンタエリスリトールテトラ(メタ)アクリレート、ジペンタエリスリトールトリ(メタ)アクリレート、ジペンタエリスリトールテトラ(メタ)アクリレート、ジペンタエリスリトールペンタ(メタ)アクリレート、N,N’-(2,2,4-トリメチルヘキサメチレン)ビス〔2-(アミノカルボキシ)プロパン-1,3-ジオール〕テトラ(メタ)アクリレート、1,7-ジ(メタ)アクリロイルオキシ-2,2,6,6-テトラ(メタ)アクリロイルオキシメチル-4-オキサヘプタンなどが挙げられる。
本発明の歯科用硬化性組成物は充填材(B)を含む。
0.03<|nP-nFBF-2d|<1.0 (3)
0.04<|nP-nFBF-2d|<0.6 (4)
0.05<|nP-nFBF-2d|<0.4 (5)
0.06<|nP-nFBF-2d|<0.2 (6)
(ここで、nPは前記重合性単量体(A)を重合して得られる重合体の屈折率、nFBF-2dは前記光拡散性無機凝集粒子(BF-2d)の屈折率を表す。)
0.03<|nP-nFBC-d|<1.0 (7)
0.04<|nP-nFBC-d|<0.6 (8)
0.05<|nP-nFBC-d|<0.4 (9)
0.06<|nP-nFBC-d|<0.2 (10)
(ここで、nPは、前記重合性単量体(A)を重合して得られる重合体の屈折率、nFBC-dは、前記光拡散性有機無機複合充填材(BC-d)の屈折率を表す。)
H2C=CR4-CO-R5-(CH2)q-SiR6 pR7 (3-p) (11)
(式中、R4は水素原子又はメチル基であり、R5は酸素原子、硫黄原子、又は-NR8-であり、ここでR8は水素原子又は炭素数1~8の脂肪族基(直鎖状でも分岐鎖状でも環状でもよい)であり、R6は加水分解可能な基であり、R7は炭素数1~6の炭化水素基であり、pは1~3の整数であり、qは1~13の整数であり、複数存在するR6及びR7は、それぞれ、互いに同一であっても異なっていてもよい。)
本発明の歯科用硬化性組成物は重合開始剤(C)を含む。重合開始剤(C)としては、一般工業界で使用されている重合開始剤から選択して使用でき、中でも歯科用途に用いられている重合開始剤が好ましく用いられる。特に、光重合開始剤及び化学重合開始剤が好ましく用いられる。重合開始剤(C)は、1種を単独で用いてもよく、2種以上を適宜組み合わせて使用してもよい。
本発明の歯科用硬化性組成物は着色剤(D)を含む。前記着色剤(D)の種類等は特に限定されず、目的とする歯科用硬化性組成物の色調に応じて無機顔料及び/又は有機顔料のいずれでも制限なく用いることができる。着色粒子の形状は、特に制限はなく球状、針状、板状、破砕状、鱗片状等の任意の粒子形状でもよく、制限なく用いることができる。無機顔料として具体的に例示すると、黄鉛、亜鉛黄、バリウム黄等のクロム酸塩;紺青等のフェロシアン化物;銀朱、カドミウム黄、硫化亜鉛、カドミウムレッド等の硫化物;硫酸バリウム、硫酸亜鉛、硫酸ストロンチウム等の硫酸塩;亜鉛華、チタン白、アンチモン白、ベンガラ、鉄黒、酸化クロム等の酸化物;水酸化アルミニウム等の水酸化物;ケイ酸カルシウム、群青等のケイ酸塩;カーボンブラック、グラファイト等の炭素等が挙げられる。有機顔料として具体的に例示すると、ナフトールグリーンB、ナフトールグリーンY等のニトロソ系顔料;ナフトールイエローS、リソールファストイエロー2G等のニトロ系顔料、パーマネントレッド4R、ブリリアントファストスカーレット、ハンザイエロー、ベンジジンイエロー等の不溶性アゾ系顔料;リソールレッド、レーキレッドC、レーキレッドD等の難溶性アゾ系顔料;ブリリアントカーミン6B、パーマネントレッドF5R、ピグメントスカーレット3B、ボルドー10B等の可溶性アゾ系顔料;フタロシアニンブルー、フタロシアニングリーン、スカイブルー等のフタロシアニン系顔料;ローダミンレーキ、マラカイトグリーンレーキ、メチルバイオレットレーキ等の塩基性染料系顔料;ピーコックブルーレーキ、エオシンレーキ、キノリンイエローレーキ、アルミニウムレーキ等の酸性染料系顔料等が挙げられる。これらの着色剤(D)は1種を単独で使用してもよく、2種以上を併用できる。これらの着色剤(D)の中でも有機顔料に比べて耐熱性や耐光性等に優れる無機顔料であるチタン白、ベンガラ、鉄黒及び黄酸化鉄等が好ましい。
本発明の歯科用硬化性組成物は、重合促進剤(E)をさらに含んでいてもよい。重合促進剤としては、アミン類、スルフィン酸及びその塩、ボレート化合物、バルビツール酸誘導体、トリアジン化合物、銅化合物、錫化合物、バナジウム化合物、ハロゲン化合物、アルデヒド類、チオール化合物、亜硫酸塩、亜硫酸水素塩、チオ尿素化合物などが挙げられる。重合促進剤(E)は、1種を単独で使用してもよく、2種以上を併用してもよい。
本発明の歯科用硬化性組成物は、上記した重合性単量体(A)、充填材(B)、重合開始剤(C)、着色剤(D)、並びに重合促進剤(E)以外にも、必要に応じて、重合禁止剤、連鎖移動剤、紫外線吸収剤、酸化防止剤、抗菌剤、分散剤、pH調整剤等の添加剤(F)を、さらに含んでいてもよい。添加剤(F)は、1種を単独で使用してもよく、2種以上を併用してもよい。
ΔE*=((L*1-L*0)2+(a*1-a*0)2+(b*1-b*0)2)1/2
(式中、記号の説明は、後記する実施例に記載のとおりである。)
本発明の歯科用硬化性組成物の調製方法に特に制限はなく、各成分を所定の配合量で配合することにより得ることができる。この際の配合順序に特に制限はなく、各成分を一括して配合してもよいし、2回以上に分けて配合してもよい。また、必要に応じて混合ないし練合したり、あるいは、真空脱泡処理等の脱泡処理を施したりしてもよい。得られた歯科用硬化性組成物は、単一の容器(シリンジ等)に充填するなどして、1材型(1ペースト型)の歯科用硬化性組成物とすることができる。
本発明の歯科用硬化性組成物の用途に特に制限はなく、各種歯科材料として用いることができ、具体的には、歯科用コンポジットレジン(例えば、齲蝕窩洞充填用コンポジットレジン、支台築造用コンポジットレジン、歯冠用コンポジットレジン、自己接着性コンポジットレジン等)、義歯床用レジン、義歯床用裏装材、印象材、合着用材料(例えば、レジンセメント、レジン添加型グラスアイオノマーセメント等)、歯科用接着材(例えば、歯列矯正用接着材、窩洞塗布用接着材等)、歯牙裂溝封鎖材、CAD/CAM用レジンブロック、テンポラリークラウン、人工歯材料などとして好ましく用いることができる。これらの中でも、本発明の歯科用硬化性組成物は、審美性が高く、機械的強度も優れることから、歯科用コンポジットレジン、及びCAD/CAM用レジンブロックとして用いることが特に好ましい。
〔充填材の平均粒子径〕
下記の充填材の平均粒子径について、エタノールを分散媒とし、レーザー回折式粒度分布測定装置(株式会社島津製作所製「SALD-2300」)を用いて体積基準で測定(特に0.1μm以上の粒子の粒子径測定の場合)、あるいは、走査型電子顕微鏡(SU3500、株式会社日立ハイテクノロジーズ製)を使用して測定した(n=1)。
下記の充填材について、屈折率が既知である有機溶媒(1-ブロモナフタレン、サリチル酸メチル、ジメチルホルムアミド、及び1-ペンタノールからなる群より選ばれる2種以上の有機溶媒を混合して各種の屈折率に調整)中に各充填材を一定量(0.1g/mL)分散させて分散液を調製し、分散液の589nmの光透過率が最大となる有機溶媒の屈折率(nD25)を求め、この屈折率を該充填材の屈折率とした。屈折率の測定対象である充填材の種類に応じて、想定される屈折率を元に、屈折率が既知である有機溶媒の種類を選択できる。該有機溶媒の屈折率は、アッベ屈折計(株式会社アタゴ製、商品名:NAR-1T LIQUID)を用いてNa-D光源で測定し、該分散液の光透過率は、紫外可視分光光度計(商品名:UV-2400、株式会社島津製作所製)を用いて、光路長10mmの石英セル中で測定した。
重合性単量体の重合体の屈折率は、上記アッベ屈折計を用いて25℃の恒温室にて測定した。具体的には、表1の各実施例及び比較例において、重合性単量体(A)、重合開始剤(C)、重合促進剤(E)、及び添加剤(F)を表1に記載の配合比にて混合した均一な重合性単量体を含有する混合物を、φ10mm×1.0mmの孔を有する金型に入れ、両面にスライドガラスを圧接した。その後、LED光重合器(αライトV、株式会社モリタ製作所製、波長:400~408nm,465~475nm)で片面を45秒ずつ、両面に光を照射して硬化させた後、型から取り出して重合性単量体を含有する混合物の硬化物を作製した。上記アッベ屈折計に硬化物をセットする際に、硬化物と測定面を密着させる目的で、試料を溶解せず、かつ試料よりも屈折率の高い溶媒(1-ブロモナフタレン)を試料に滴下し測定した。
下記の各製造例で得られた無機充填材について、比表面積測定装置(マイクロトラック・ベル株式会社製「BELSORP-mini II」)を用い、100℃で2時間真空脱揮した後、吸着ガス:窒素、測定温度:77Kの条件でBET法に基づき無機充填材の比表面積を測定した(n=1)。なお当該測定においては、飽和蒸気圧P0(kPa)に対する吸着平衡圧P(kPa)の比(P/P0)が0.05~0.3の範囲にある吸着側等温線上の5点を用いたBET多点法による解析を採用した。
硬化物の分光反射率の比は、分光色差計(SE6000、日本電色工業株式会社製、光源:D65/2、測定窓:φ6mm)を用いて評価した。具体的には、歯科用硬化性組成物をステンレス製の金型(φ10mm×厚さ1mm)に充填し、上下をカバーガラスで挟み、圧接した状態で、LED光重合器(αライトV、株式会社モリタ製作所製、波長:400~408nm,465~475nm)で片面を45秒ずつ、両面に光を照射して硬化させた。硬化板を金型から取り出し、硬化板の背後に構成された標準白色板(X=93.94、Y=95.90、Z=112.92)を置いた状態で、380~780nmの波長に対する分光反射率を測定した(n=1)。既述の計算式に基づいて分光反射率の比(R650/600、R700/600、R750/600)を算出した。
硬化物のコントラスト比は、上記の分光色差計を用いて評価した。具体的には、上記と同様な方法でφ10mm×厚さ1mmの硬化板を作製し、三刺激値のY値を黒背景色及び白背景で測定した。白背景には標準白色板、黒背景には艶消し暗箱を用いた。既述の計算式に基づいてコントラスト比を計算した(n=1)。
硬化物の光拡散度LDは、ゴニオフォトメーター(株式会社村上色彩技術研究所、GP-200)を用いて評価した。具体的には、歯科用硬化性組成物を厚さ0.25mmのステンレス製のスペーサーを用いて、上下をカバーガラスで挟み、圧接した状態で、LED光重合器(αライトV、株式会社モリタ製作所製、波長:400~408nm,465~475nm)で片面を45秒ずつ、両面に光を照射して硬化させ、φ30mm×厚さ0.25mmの硬化板を作製した。ゴニオフォトメーターを用いて、入射光角度0°にて、この硬化板の-90°~+90°における透過光の光度分布を測定し、既述の計算式(2)に従って光拡散度LDを算出した(n=1)。
硬化物の明度及び色度は、分光反射率の比で使用した上記の分光色差計(SE6000、日本電色工業株式会社製、光源:D65/2、測定窓:φ6mm)を用いて評価した。具体的には、上記と同様な方法でφ10mm×厚さ1mmの硬化板を作製し、硬化板の背後に標準白色板を置いた状態で、L*a*b*表色系における明度(L*/w)、及び色度指数(a*/w、b*/w)を測定した(n=1)。
金属製の充填器具を用いてプラスチック製の模擬窩洞(5級窩洞を模したφ3mm×深さ2mmの穴)に25℃で組成物の充填操作を行い、充填操作のしやすさを評価した。具体的には、歯科用硬化性組成物の性状が粘土状である場合、金属製の歯科用充填器(株式会社ヨシダ製、練成充填器#4及び#5)を用いて、上記模擬窩洞に各実施例及び比較例の歯科用硬化性組成物を充填し、その際に感じるベタツキを以下の基準に従い評価した。また、歯科用硬化性組成物の性状が液状である場合、歯科用硬化性組成物をシリンジ容器(クラレノリタケデンタル株式会社製、クリアフィルマジェスティESフローのシリンジ容器)に充填し、付属のニードルチップを装着した前記シリンジ容器から歯科用硬化性組成物を押し出すことで上記模擬窩洞に充填し、その際の歯科用硬化性組成物の糸引きを以下の基準に従い評価した。
〇:ベタツキを感じない又は糸引きがない
△:ややベタツキを感じる又はやや糸引きがある
×:ひどいベタツキを感じる又は糸引きがある
歯科用硬化性組成物の臼歯部の色調適合性は、人工歯に形成した窩洞を充填修復し、その部分を歯科用測色装置で測色することで評価した。具体的には、まず、人工歯6番臼歯(ゼンオパール臼歯、サイズ:PL16、シェード:A1、A3、及びA4、株式会社ジーシー製)を歯科用測色装置(Crystaleye Spectrophotometer、オリンパス株式会社製)を用いて撮影した。この際、人工歯は歯科用測色装置に付属の暗箱(チェックボックス ウエカバー)内で測定した。続いて、その人工歯の中央にφ4mm×深さ2mmのI級窩洞を形成し、歯科用エッチング材(K エッチャント GEL、クラレノリタケデンタル株式会社製)を用い、事業者推奨の方法(添付文書に記載方法)にて窩洞面にエッチング処理を行った。続いて歯科用接着材(クリアフィルユニバーサルボンドQuickER、クラレノリタケデンタル株式会社製)を用いて、事業者推奨の方法にて窩洞面にボンディング処理を行い、歯科用可視光線照射器(ペンキュアー2000、株式会社モリタ製作所製)を用いて、ノーマルモードで10秒光照射した。その後、窩洞に歯科用硬化性組成物を充填し、事前にシリコーン印象材(メモジル2、ヘレウスクルツァージャパン株式会社製)で作製した人工歯の鋳型で押さえつけることにより、人工歯の表面形状を賦形した。歯科用ユニット(ポータケア21、株式会社モリタ製作所製)を使用し、注水下にて、歯科用ゴム製研磨材(コンポマスター、CA、13S、株式会社松風製)を用いて回転速度約10,000rpmで充填部を30秒間研磨した。最後に上記の歯科用可視光線照射器を用いて、鋳型の上からノーマルモードで10秒光照射し、続いて鋳型を取り外し、ノーマルモードで10秒光照射することで歯科用硬化性組成物の硬化を行い、充填試料を作製した。作製した試料を初めに撮影した方法と同様の方法にて撮影した。歯科用測色装置に付属のソフトにて、撮影した充填試料の画像中の充填部の明度(L*1)、及び色度(a*1、b*1)を測定した。さらに、初めに撮影した未処理人工歯の画像についても、人工歯の充填試料の画像で測定した箇所と同じ箇所の明度(L*0)、及び色度(a*0、b*0)を測定した。下記式に基づいて色差ΔE*を計算し、色調適合性の指標とした(n=1)。
ΔE*=((L*1-L*0)2+(a*1-a*0)2+(b*1-b*0)2)1/2
A1、A3、及びA4の人工歯に対して、いずれのΔE*も6.0以下であることが好ましく、5.5以下がより好ましく、5.0以下がさらに好ましい。
歯科用硬化性組成物の前歯部の色調適合性は、シェードガイドに形成した窩洞を充填修復し、その部分を歯科用測色装置で測色することで評価した。具体的には、まず、シェードガイド(ビタ クラシカル シェードガイド、シェード:A1、A3、及びA4、ビタ社(VITA Zahnfabrik、Germany)製)を、上記歯科用測色装置を用いて同様の方法にて撮影した。続いて、そのシェードガイドの切縁部に、φ4mmの四分円状の舌側から唇側まで貫通するIV級窩洞を形成し、後は上記と同様な方法で充填試料を作製した。また、上記と同様な方法にて色差ΔE*を計算し、色調適合性の指標とした(n=1)。A1、A3、及びA4のシェードガイドに対して、それぞれ、ΔE*が7.0以下であることが好ましく、6.0以下がより好ましく、5.0以下がさらに好ましく、また、A1、A3、及びA4のシェードガイドに対していずれのΔE*も6.0以下であることが特に好ましい。
ポリテトラフルオロエチレン製の型(φ10mm×厚さ2.0mm)に歯科用硬化性組成物を充填し、歯科用可視光線照射器(ペンキュアー2000、株式会社モリタ製作所製)で10秒光照射を行い、硬化物を型から取り出し試験片とした。この試験片の平滑面を、乾燥条件下、#600の研磨紙にて研磨した。続いて、歯科用ユニット(ポータケア21、株式会社モリタ製作所製)を使用し、注水下にて、歯科用ゴム製研磨材(コンポマスター、CA、13S、株式会社松風製)を用いて回転速度約10,000rpmで10秒間研磨した。その後、この研磨面の光沢を光沢計(VG2000、日本電色工業株式会社製、測定角度:60度、JIS Z 8741:1997に準拠)を用いて測定し、鏡を100%とした時の割合(光沢度)を求め、これを硬化物の易研磨性の指標とした(n=3)。測定値の平均値を表2に示す。光沢度は、20%以上であることが好ましく、25%以上であることがより好ましく、30%以上であることがさらに好ましい。
耐摩耗試験前後の光沢度の変化に基づき滑沢耐久性を評価した。具体的には以下のとおりである。歯科用硬化性組成物(ペースト)をSUS製の金型(縦30mm×横20mm×厚さ2mm)に充填し、ペーストの上下(30mm×20mmの面)をスライドガラスで圧接した。次いで、歯科技工用可視光照射器(αライトV、株式会社モリタ製)で、スライドガラス越しに180秒間ずつペーストの裏表に光照射してペーストを硬化させた。得られた硬化物を試験片として用いて、この表面を1500番の耐水研磨紙で研磨し、研磨面を作製した。次いで、その研磨面を技工用ポリッシングボックス(EWL80、KaVo社製)を用いて3000rpmで20秒間バフ研磨した。バフ研磨後の試験片を耐摩耗試験に供した。バフ研磨には、歯科用研磨材(商品名:ポーセニイ ハイドン(PORCENY HYDON)、株式会社東京歯材社製)を用いた。耐摩耗試験前の試験片の光沢度(G1)を、光沢計(VG2000、日本電色工業株式会社製、JIS Z 8741:1997に準拠)を用いて、鏡を100%としたときの割合として求めた。測定角度は60度とした。同試験片を歯ブラシ摩耗試験機(株式会社大栄科学精器製作所製)により、歯磨材の懸濁液、及び市販の歯ブラシ(ビトイーン、サイズ:レギュラー、かたさ:ふつう、ライオン株式会社製)を用いて、荷重250g下、40,000サイクルの耐摩耗試験を行った。前記歯磨材の懸濁液は、市販の歯磨材(デンタークリアマックス、ライオン株式会社製)を用いて、歯磨材/蒸留水を質量比で10/90として調製した。耐摩耗試験後の試験片の表面の光沢度(G2)を耐摩耗試験前と同様の方法で求めた。耐摩耗試験前後の試験片の表面の光沢度から、滑沢耐久性(%)を{(G2)×100}/(G1)として算出した。滑沢耐久性は、40%以上であることが好ましく、45%以上であることがより好ましく、50%以上であることがさらに好ましく、55%以上、60%以上、65%以上であることが特に好ましい。
(重合性単量体(A))
Bis-GMA:2,2-ビス〔4-(3-メタクリロイルオキシ-2-ヒドロキシプロポキシ)フェニル〕プロパン
D2.6E:2,2-ビス(4-メタクリロイルオキシポリエトキシフェニル)プロパン(エチレンオキシ基の平均付加モル数:2.6)
UDMA:2,2,4-トリメチルヘキサメチレンビス(2-カルバモイルオキシエチル)ジメタクリレート
DD:1,10-デカンジオールジメタクリレート
3G:トリエチレングリコールジメタクリレート
POBMA:m-フェノキシベンジルメタクリレート
充填材としては、下記の製造例で得られたものを用いた。また、市販品はそのまま用いた。
・無機微粒子(BF-1-1)の製造
市販のバリウムガラス(8235 UF0.7、ショット社製、平均粒子径0.7μm、屈折率:1.55)100質量部、3-メタクリロイルオキシプロピルトリメトキシシラン2質量部、及びトルエン170質量部を三口フラスコに入れ、2時間、室温下で撹拌した。トルエンを減圧下で留去した後、40℃で16時間真空乾燥を行い、さらに90℃で3時間加熱し、表面処理層が設けられた無機微粒子(BF-1-1)を得た。得られた無機微粒子(BF-1-1)の平均粒子径は0.7μmであった。
・無機微粒子(BF-1-2)の製造
市販のバリウムガラス(GM27884 NanoFine180、ショット社製、平均粒子径0.2μm、屈折率:1.53)100質量部、3-メタクリロイルオキシプロピルトリメトキシシラン11質量部、及びトルエン170質量部を三口フラスコに入れ、2時間、室温下で撹拌した。トルエンを減圧下で留去した後、40℃で16時間真空乾燥を行い、さらに90℃で3時間加熱し、表面処理層が設けられた無機微粒子(BF-1-2)を得た。得られた無機微粒子(BF-1-2)の平均粒子径は0.2μmであった。
・無機微粒子(BF-1-3)の製造
市販のバリウムガラス(GM27884 NanoFine180、ショット社製、平均粒子径0.2μm、屈折率:1.53)100質量部、8-メタクリロイルオキシオクチルトリメトキシシラン11質量部、及びトルエン170質量部を三口フラスコに入れ、2時間、室温下で撹拌した。トルエンを減圧下で留去した後、40℃で16時間真空乾燥を行い、さらに90℃で3時間加熱し、表面処理層が設けられた無機微粒子(BF-1-3)を得た。得られた無機微粒子(BF-1-3)の平均粒子径は0.2μmであった。
・無機微粒子(BF-1-4)の製造
市販のバリウムガラス(GM27884 NanoFine180、ショット社製、平均粒子径0.2μm、屈折率:1.53)100質量部、11-メタクリロイルオキシウンデシルトリメトキシシラン11質量部、及びトルエン170質量部を三口フラスコに入れ、2時間、室温下で撹拌した。トルエンを減圧下で留去した後、40℃で16時間真空乾燥を行い、さらに90℃で3時間加熱し、表面処理層が設けられた無機微粒子(BF-1-4)を得た。得られた無機微粒子(BF-1-4)の平均粒子径は0.2μmであった。
・無機微粒子(BF-1-5)の製造
市販品である表面処理シリカコート-フッ化イッテルビウム(SG-YBF100WSCMP10、一次粒子平均粒子径:110nm、屈折率:1.54、Sukgyung AT社製)をそのまま用いた。
・他の無機凝集粒子(BF-2e-1)の製造
市販のシリカ-イッテルビウム酸化物水分散液(SG-YBSO30SW、Sukgyung AT社製、一次粒子平均粒子径:30nm)中の水を、エバポレーターを用いて留去し、得られた固体成分を遊星型ボールミル(フリッチュ社(Fritsch、Germany)製、「クラシックラインP-6」、ジルコニアボール)により180分間粉砕した。得られた粉体を800℃に設定した電気炉にて1時間焼成し、これをさらに前記遊星型ボールミルにて180分間粉砕した。得られた粉末100質量部に対して10質量部の3-メタクリロイルオキシプロピルトリメトキシシランで表面処理することにより疎水化して、他の無機凝集粒子(BF-2e-1)を得た。得られた無機凝集粒子(BF-2e-1)の平均粒子径は5.7μmであり、屈折率1.53、比表面積は95.8m2/gであった。
・光拡散性無機凝集粒子(BF-2d-1)の製造
凝集シリカ「シリカマイクロビード P-500(一次粒子平均粒子径:12nm、凝集体平均粒子径2μm)」(日揮触媒化成株式会社製)100質量部、3-メタクリロキシプロピルトリメトキシシラン20質量部、及びトルエン170質量部を三口フラスコに入れ、2時間、室温下で撹拌した。トルエンを減圧下で留去した後、40℃で16時間真空乾燥を行い、さらに90℃で3時間加熱し、シラン処理した光拡散性無機凝集粒子(BF-2d-1)を得た。シラン処理した光拡散性無機凝集粒子(BF-2d-1)の平均粒子径は1.6μm、屈折率は1.44、比表面積は99m2/g、細孔容積は0.19mL/gであった。
・光拡散性有機無機複合充填材(BC-d-1)の製造
Bis-GMA70質量部、3G30質量部、及び過酸化ベンゾイル0.5質量部を混合し、均一に溶解し、重合性単量体含有組成物を得た。一方、平均粒子径0.04μmのコロイドシリカ粉末(日本アエロジル株式会社製、アエロジル(登録商標)OX50)に対し、常法によりγ-メタクリロキシプロピルトリメトキシシランで表面処理を行った。該表面処理コロイドシリカ100質量部と上記重合性単量体100質量部を混練し、ペースト状の組成物を得た。該組成物を減圧下、130℃で3時間加熱して重合させ、得られた硬化物をさらにボールミルで粉砕して、表面未処理有機無機複合フィラーを得た。さらに表面未処理有機無機複合フィラー100質量部に対し、1質量部のγ-メタクリロキシプロピルトリメトキシシランで表面処理を行い、光拡散性有機無機複合充填材(BC-d-1)を得た。光拡散性有機無機複合充填材(BC-d-1)の平均粒子径は11μm、屈折率は1.50であった。
・光拡散性有機無機複合充填材(BC-d-2)の製造
UDMA70質量部、DD30質量部、及び過酸化ベンゾイル0.5質量部を混合し、均一に溶解し、重合性単量体含有組成物を得た。一方、平均粒子径0.04μmのコロイドシリカ粉末(日本アエロジル株式会社製、アエロジル(登録商標)OX50)に対し、常法によりγ-メタクリロキシプロピルトリメトキシシランで表面処理を行った。該表面処理コロイドシリカ100質量部と上記重合性単量体100質量部を混練し、ペースト状の組成物を得た。該組成物を減圧下、130℃で3時間加熱して重合させ、得られた硬化物をさらにボールミルで粉砕して、表面未処理有機無機複合フィラーを得た。さらに表面未処理有機無機複合フィラー100質量部に対し、1質量部のγ-メタクリロキシプロピルトリメトキシシランで表面処理を行い、光拡散性有機無機複合充填材(BC-d-2)を得た。光拡散性有機無機複合充填材(BC-d-2)の平均粒子径は15μm、屈折率1.49であった。
・有機無機複合充填材(BC-e-1)の製造
予め重合開始剤としてアゾビスイソブチロニトリル(AIBN)を1質量%溶解した、Bis-GMAと3Gの重合性単量体混合物(質量比1:1)100質量部に対して、無機充填材として上記で製造した無機微粒子(BF-1-2)を100質量部添加、混合しペースト化した。これを100℃、減圧雰囲気下で5時間加熱重合した。得られた重合硬化物を、振動ボールミルを用いて、平均粒子径が約5μmとなるまで粉砕した。得られた粉末100gに対して、γ-メタクリロイルオキシプロピルトリメトキシシラン2質量%含有エタノール溶液200mL中、90℃で5時間還流することで表面処理を行ない、有機無機複合充填材(BC-e-1)を得た。得られた有機無機複合充填材(BC-e-1)の平均粒子径は5.2μm、屈折率は1.54であった。
・その他の無機粒子(BF-3-1)の製造
市販のバリウムガラス(8235 UF1.5、ショット社製、平均粒子径1.5μm、屈折率:1.55)100質量部、3-メタクリロイルオキシプロピルトリメトキシシラン2質量部、及びトルエン170質量部を三口フラスコに入れ、2時間、室温下で撹拌した。トルエンを減圧下で留去した後、40℃で16時間真空乾燥を行い、さらに90℃で3時間加熱し、表面処理層が設けられた無機粒子(BF-3-1)を得た。得られた無機粒子(BF-3-1)の平均粒子径は1.5μmであった。
CQ:カンファーキノン
TPO:2,4,6-トリメチルベンゾイルジフェニルホスフィンオキシド
D-1:酸化チタン
D-2:鉄黒
D-3:黄酸化鉄
D-4:ベンガラ
PDE:4-(N,N-ジメチルアミノ)安息香酸エチル
TIN:「チヌビン326」(BASFジャパン株式会社製)(紫外線吸収剤)
BHT:3,5-ジ-t-ブチル-4-ヒドロキシトルエン(重合禁止剤)
表1に記載された材料及び割合にて、これらを常温(23℃)及び暗所で混合練和し、均一にしたものを真空脱泡することにより、歯科用硬化性組成物を調製した。これらの歯科用硬化性組成物について、上記した方法で各試験を行った。結果を表2に示した。
Claims (9)
- 重合性単量体(A)、充填材(B)、重合開始剤(C)、及び着色剤(D)を含む歯科用硬化性組成物であって、
前記歯科用硬化性組成物の厚さ1.0mmの硬化物を、分光色差計を用いて白背景で測定した際の、600nmの波長における分光反射率に対する、650nm、700nm、及び750nmの波長における分光反射率の比R650/600、R700/600、及びR750/600がいずれも、97%~103%の範囲内である、歯科用硬化性組成物。 - 厚さ1.0mmの硬化物における、以下の式(1)で定義されるコントラスト比が、0.35~0.65を満たす、請求項1に記載の歯科用硬化性組成物。
コントラスト比=Yb/Yw (1)
(ここで、Ybは黒背景において測定したXYZ表色系のY値を表し、Ywは白背景において測定したXYZ表色系のY値を表す。) - 厚さ0.25mmの硬化物における、以下の式(2)で定義される光拡散度LDが、0.0001~0.99を満たす、請求項1又は2に記載の歯科用硬化性組成物。
LD=(I5/cos5°)/I0 (2)
(ここで、Iは硬化物を透過した光の光度を表し、I0、及びI5は試料板に垂直な方向(光の入射方向)に対し、それぞれ0度、及び5度傾いた方向の透過光の光度を表す。) - 厚さ1.0mmの硬化物における、硬化物の背後に標準白色板を置いて測定した際のL*a*b*表色系における色度指数であるa*/wが、-3.0~2.0である、請求項1~3のいずれかに記載の歯科用硬化性組成物。
- 前記充填材(B)が、平均粒子径が0.05~1μmである無機微粒子(BF-1)を含む、請求項1~4のいずれかに記載の歯科用硬化性組成物。
- 前記充填材(B)が、無機一次粒子(x)が凝集してなる無機凝集粒子(BF-2)を含み、前記無機一次粒子(x)の平均粒子径が0.001~1μmである、請求項1~5のいずれかに記載の歯科用硬化性組成物。
- 前記無機凝集粒子(BF-2)が、光拡散性無機凝集粒子(BF-2d)を含み、前記光拡散性無機凝集粒子(BF-2d)の屈折率が下記式(3)を満たす、請求項6に記載の歯科用硬化性組成物。
0.03<|nP-nFBF-2d|<1.0 (3)
(ここで、nPは、前記重合性単量体(A)を重合して得られる重合体の屈折率、nFBF-2dは、前記光拡散性無機凝集粒子(BF-2d)の屈折率を表す。) - 前記充填材(B)が、無機一次粒子(x)を含有する有機無機複合充填材(BC)を含み、前記無機一次粒子(x)の平均粒子径が0.001~1μmである、請求項1~7のいずれかに記載の歯科用硬化性組成物。
- 前記有機無機複合充填材(BC)が、光拡散性有機無機複合充填材(BC-d)を含み、前記光拡散性有機無機複合充填材(BC-d)の屈折率が下記式(7)を満たす、請求項8に記載の歯科用硬化性組成物。
0.03<|nP-nFBC-d|<1.0 (7)
(ここで、nPは、前記重合性単量体(A)を重合して得られる重合体の屈折率、nFBC-dは、前記光拡散性有機無機複合充填材(BC-d)の屈折率を表す。)
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US18/034,500 US20230381066A1 (en) | 2020-10-28 | 2021-10-28 | Dental curable composition having good color compatibility |
| CN202180074243.8A CN116367806A (zh) | 2020-10-28 | 2021-10-28 | 具有良好的色调适应性的齿科用固化性组合物 |
| AU2021372118A AU2021372118A1 (en) | 2020-10-28 | 2021-10-28 | Dental curable composition having good color compatibility |
| JP2022559226A JPWO2022092193A1 (ja) | 2020-10-28 | 2021-10-28 | |
| EP21886310.8A EP4238546A1 (en) | 2020-10-28 | 2021-10-28 | Dental curable composition having good color compatibility |
| CA3196983A CA3196983A1 (en) | 2020-10-28 | 2021-10-28 | Dental curable composition having good color compatibility |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020-180960 | 2020-10-28 | ||
| JP2020180960 | 2020-10-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022092193A1 true WO2022092193A1 (ja) | 2022-05-05 |
Family
ID=81382625
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2021/039803 WO2022092193A1 (ja) | 2020-10-28 | 2021-10-28 | 良好な色調適合性を有する歯科用硬化性組成物 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20230381066A1 (ja) |
| EP (1) | EP4238546A1 (ja) |
| JP (1) | JPWO2022092193A1 (ja) |
| CN (1) | CN116367806A (ja) |
| AU (1) | AU2021372118A1 (ja) |
| CA (1) | CA3196983A1 (ja) |
| WO (1) | WO2022092193A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023191112A1 (ja) * | 2022-04-01 | 2023-10-05 | クラレノリタケデンタル株式会社 | 良好な色調適合性を有する歯科用硬化性組成物 |
| JP2023176641A (ja) * | 2022-05-31 | 2023-12-13 | Yamakin株式会社 | 歯科用硬化性組成物 |
| WO2024150826A1 (ja) * | 2023-01-13 | 2024-07-18 | クラレノリタケデンタル株式会社 | 歯科用組成物 |
Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5042696A (ja) | 1973-04-24 | 1975-04-17 | ||
| JPH093109A (ja) | 1995-04-19 | 1997-01-07 | Tokuyama Corp | 可視光線重合開始剤および可視光線重合性組成物 |
| JPH09255516A (ja) | 1996-03-25 | 1997-09-30 | Kuraray Co Ltd | 歯科用複合材料 |
| JPH101473A (ja) | 1996-04-18 | 1998-01-06 | Tokuyama Corp | チオウラシル誘導体 |
| JPH10245525A (ja) | 1997-03-03 | 1998-09-14 | Tokuyama Corp | 接着性組成物 |
| JPH1192461A (ja) | 1997-09-19 | 1999-04-06 | Tokuyama Corp | ジスルフィド化合物および金属表面処理剤 |
| JP2003096122A (ja) | 2001-09-21 | 2003-04-03 | Tokuyama Corp | ラジカル重合触媒 |
| JP2006510583A (ja) | 2002-08-21 | 2006-03-30 | ビスコ インコーポレイテッド | 不揮発性歯科組成物 |
| JP2011144121A (ja) | 2010-01-13 | 2011-07-28 | Tokuyama Dental Corp | 歯科用充填修復キット |
| JP2015067594A (ja) * | 2013-09-30 | 2015-04-13 | 株式会社松風 | 異なる屈折率の粒子を含む硬化性歯科用組成物 |
| WO2015064090A1 (ja) * | 2013-10-31 | 2015-05-07 | クラレノリタケデンタル株式会社 | 歯科用色調調整材キット |
| WO2018043595A1 (ja) * | 2016-08-31 | 2018-03-08 | 株式会社トクヤマデンタル | 光硬化性組成物 |
| WO2019062144A1 (zh) * | 2017-09-30 | 2019-04-04 | 辽宁爱尔创生物材料有限公司 | 透光率和颜色渐变的牙科材料的制备方法及其制备的产品 |
| WO2019189698A1 (ja) * | 2018-03-30 | 2019-10-03 | 株式会社トクヤマデンタル | 歯科切削加工用レジン系ブロック |
| JP2020011917A (ja) * | 2018-07-17 | 2020-01-23 | 株式会社トクヤマデンタル | 歯科用硬化性組成物 |
| WO2020122192A1 (ja) | 2018-12-13 | 2020-06-18 | クラレノリタケデンタル株式会社 | 歯科用組成物 |
| WO2020138472A1 (ja) * | 2018-12-27 | 2020-07-02 | クラレノリタケデンタル株式会社 | 色調最適化された歯科用ミルブランク |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6250245B2 (ja) * | 2015-10-21 | 2017-12-20 | 株式会社トクヤマデンタル | 硬化性組成物及び歯科用充填修復材料 |
| KR102491300B1 (ko) * | 2017-03-06 | 2023-01-25 | 가부시키가이샤 도쿠야마 덴탈 | 광경화성 조성물 및 치과용 수복 충전 재료 |
-
2021
- 2021-10-28 WO PCT/JP2021/039803 patent/WO2022092193A1/ja active Application Filing
- 2021-10-28 EP EP21886310.8A patent/EP4238546A1/en active Pending
- 2021-10-28 AU AU2021372118A patent/AU2021372118A1/en active Pending
- 2021-10-28 JP JP2022559226A patent/JPWO2022092193A1/ja active Pending
- 2021-10-28 US US18/034,500 patent/US20230381066A1/en active Pending
- 2021-10-28 CA CA3196983A patent/CA3196983A1/en active Pending
- 2021-10-28 CN CN202180074243.8A patent/CN116367806A/zh active Pending
Patent Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5042696A (ja) | 1973-04-24 | 1975-04-17 | ||
| JPH093109A (ja) | 1995-04-19 | 1997-01-07 | Tokuyama Corp | 可視光線重合開始剤および可視光線重合性組成物 |
| JPH09255516A (ja) | 1996-03-25 | 1997-09-30 | Kuraray Co Ltd | 歯科用複合材料 |
| JPH101473A (ja) | 1996-04-18 | 1998-01-06 | Tokuyama Corp | チオウラシル誘導体 |
| JPH10245525A (ja) | 1997-03-03 | 1998-09-14 | Tokuyama Corp | 接着性組成物 |
| JPH1192461A (ja) | 1997-09-19 | 1999-04-06 | Tokuyama Corp | ジスルフィド化合物および金属表面処理剤 |
| JP2003096122A (ja) | 2001-09-21 | 2003-04-03 | Tokuyama Corp | ラジカル重合触媒 |
| JP2006510583A (ja) | 2002-08-21 | 2006-03-30 | ビスコ インコーポレイテッド | 不揮発性歯科組成物 |
| JP2011144121A (ja) | 2010-01-13 | 2011-07-28 | Tokuyama Dental Corp | 歯科用充填修復キット |
| JP2015067594A (ja) * | 2013-09-30 | 2015-04-13 | 株式会社松風 | 異なる屈折率の粒子を含む硬化性歯科用組成物 |
| WO2015064090A1 (ja) * | 2013-10-31 | 2015-05-07 | クラレノリタケデンタル株式会社 | 歯科用色調調整材キット |
| WO2018043595A1 (ja) * | 2016-08-31 | 2018-03-08 | 株式会社トクヤマデンタル | 光硬化性組成物 |
| WO2019062144A1 (zh) * | 2017-09-30 | 2019-04-04 | 辽宁爱尔创生物材料有限公司 | 透光率和颜色渐变的牙科材料的制备方法及其制备的产品 |
| WO2019189698A1 (ja) * | 2018-03-30 | 2019-10-03 | 株式会社トクヤマデンタル | 歯科切削加工用レジン系ブロック |
| JP2020011917A (ja) * | 2018-07-17 | 2020-01-23 | 株式会社トクヤマデンタル | 歯科用硬化性組成物 |
| WO2020122192A1 (ja) | 2018-12-13 | 2020-06-18 | クラレノリタケデンタル株式会社 | 歯科用組成物 |
| WO2020138472A1 (ja) * | 2018-12-27 | 2020-07-02 | クラレノリタケデンタル株式会社 | 色調最適化された歯科用ミルブランク |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023191112A1 (ja) * | 2022-04-01 | 2023-10-05 | クラレノリタケデンタル株式会社 | 良好な色調適合性を有する歯科用硬化性組成物 |
| JP2023176641A (ja) * | 2022-05-31 | 2023-12-13 | Yamakin株式会社 | 歯科用硬化性組成物 |
| JP7420405B2 (ja) | 2022-05-31 | 2024-01-23 | Yamakin株式会社 | 歯科用硬化性組成物 |
| WO2024150826A1 (ja) * | 2023-01-13 | 2024-07-18 | クラレノリタケデンタル株式会社 | 歯科用組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4238546A1 (en) | 2023-09-06 |
| JPWO2022092193A1 (ja) | 2022-05-05 |
| US20230381066A1 (en) | 2023-11-30 |
| AU2021372118A1 (en) | 2023-06-22 |
| CA3196983A1 (en) | 2022-05-05 |
| CN116367806A (zh) | 2023-06-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2472708C2 (ru) | Наполнители и композитные материалы с наночастицами диоксида циркония и кремнезема | |
| WO2022092193A1 (ja) | 良好な色調適合性を有する歯科用硬化性組成物 | |
| JP4782251B2 (ja) | 歯科用硬化性組成物及びそれを用いたコンポジットレジン | |
| US9993394B2 (en) | Dental restorative composition | |
| JP6930993B2 (ja) | 複数色積層歯科用ミルブランク | |
| JP6929014B2 (ja) | 歯科用ミルブランクの製造方法 | |
| JP2022106700A (ja) | 重合性樹脂と充填剤との間に屈折率差をもたらすナノ粒子を含む歯科用組成物 | |
| WO2020218446A1 (ja) | 歯科修復用硬化性組成物 | |
| KR102698521B1 (ko) | 치과 절삭 가공용 레진계 블록 | |
| JP7426343B2 (ja) | 歯科用組成物 | |
| JP5331697B2 (ja) | 歯科用重合性組成物およびそのキット | |
| WO2021125246A1 (ja) | 蛍光性を有する歯科用硬化性組成物及びその硬化物 | |
| JP6846167B2 (ja) | 歯科用ミルブランクアセンブリ | |
| WO2021132463A1 (ja) | 歯科用修復材組成物 | |
| JPS63303906A (ja) | 人工歯材料 | |
| JP7061826B2 (ja) | 歯科用ミルブランク | |
| JP7464615B2 (ja) | 歯科用硬化性組成物 | |
| WO2023191112A1 (ja) | 良好な色調適合性を有する歯科用硬化性組成物 | |
| JP2016153382A (ja) | 歯科用硬化性組成物 | |
| JP7308612B2 (ja) | プレポリマー及びそれを用いた歯科用組成物 | |
| US20230225829A1 (en) | Process for producing a surface-modified dental article |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21886310 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 3196983 Country of ref document: CA Ref document number: 2022559226 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18034500 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2021886310 Country of ref document: EP Effective date: 20230530 |
|
| ENP | Entry into the national phase |
Ref document number: 2021372118 Country of ref document: AU Date of ref document: 20211028 Kind code of ref document: A |