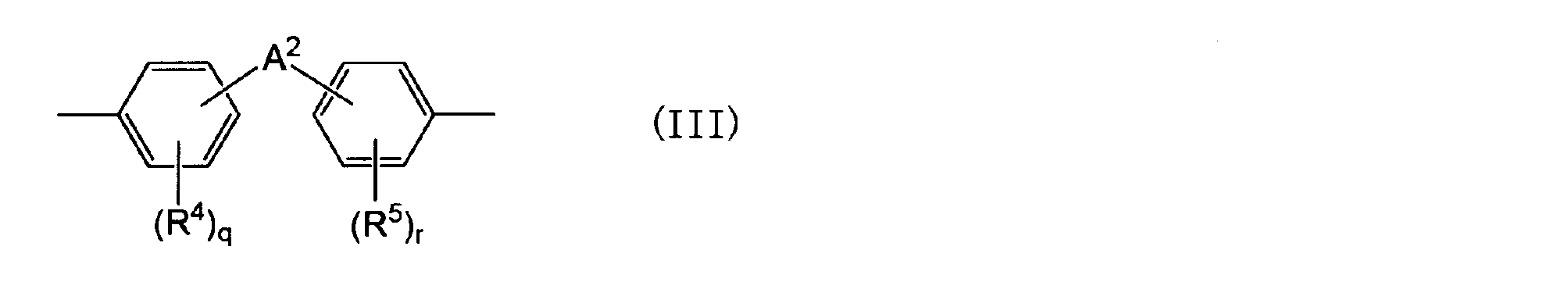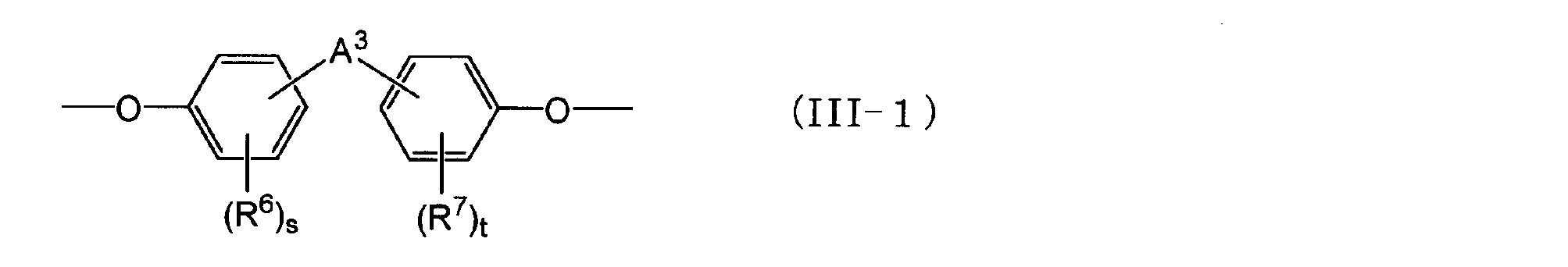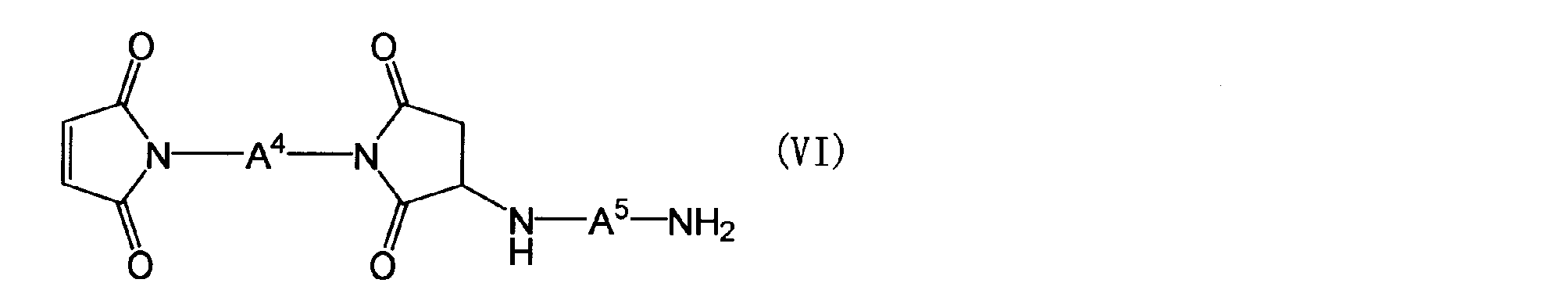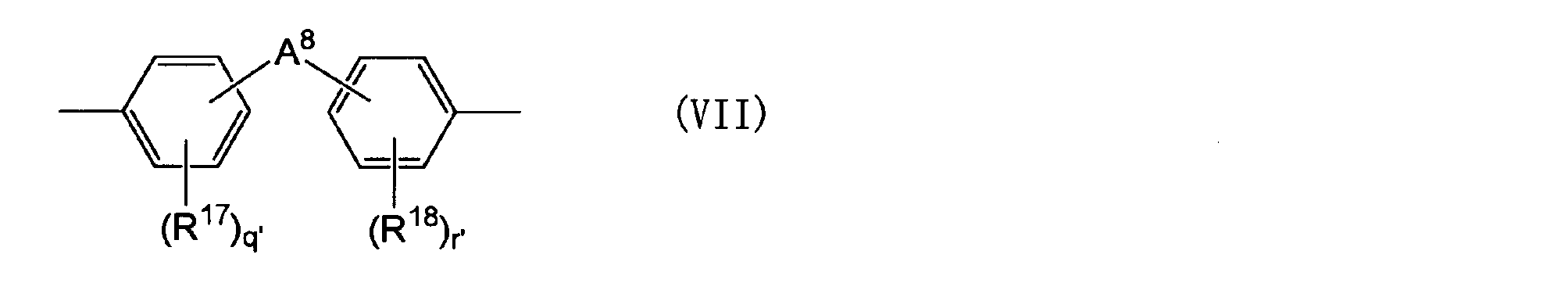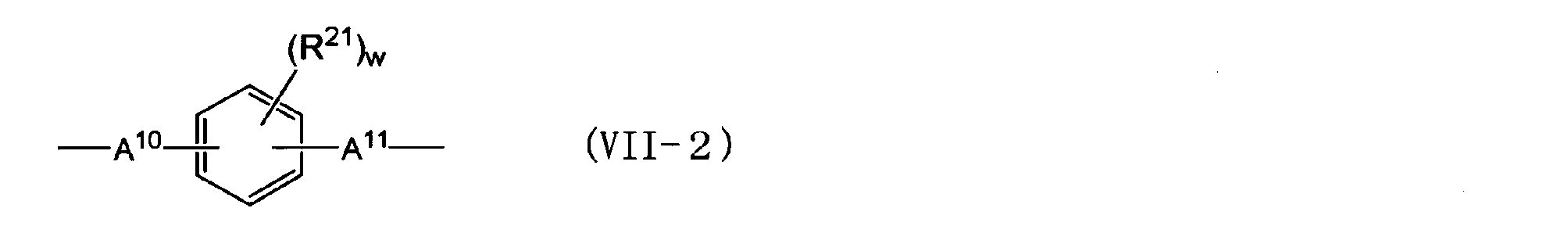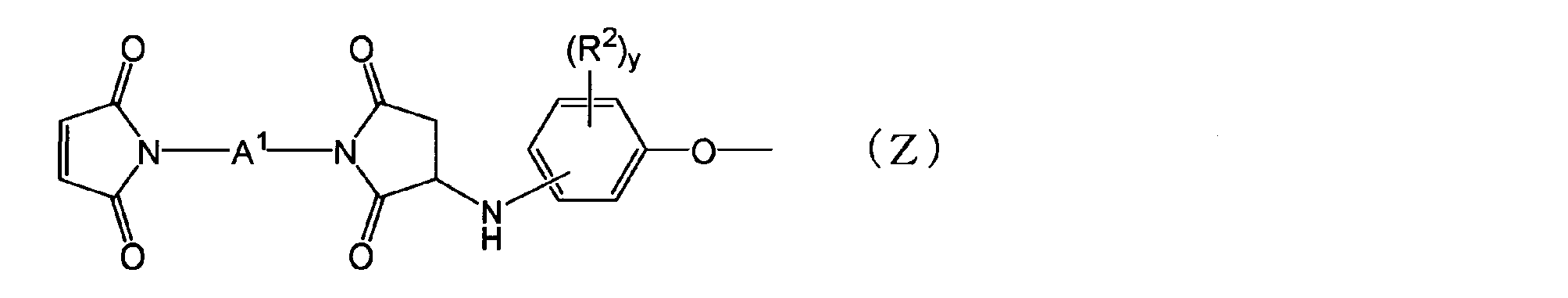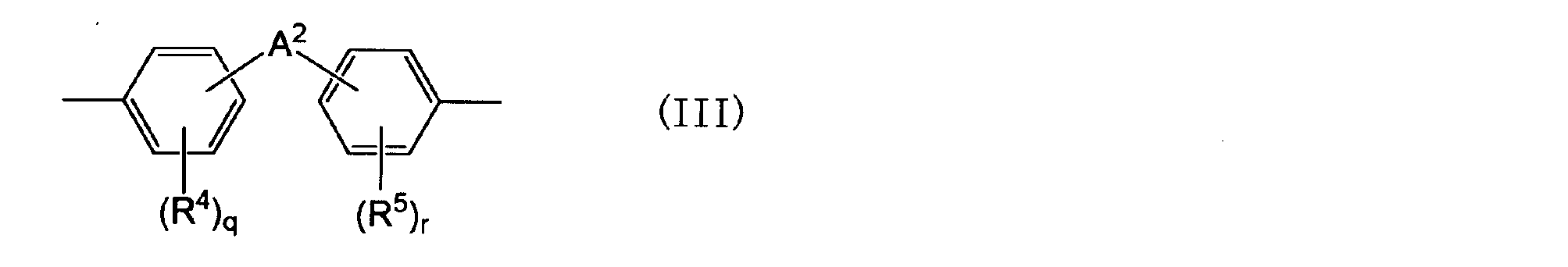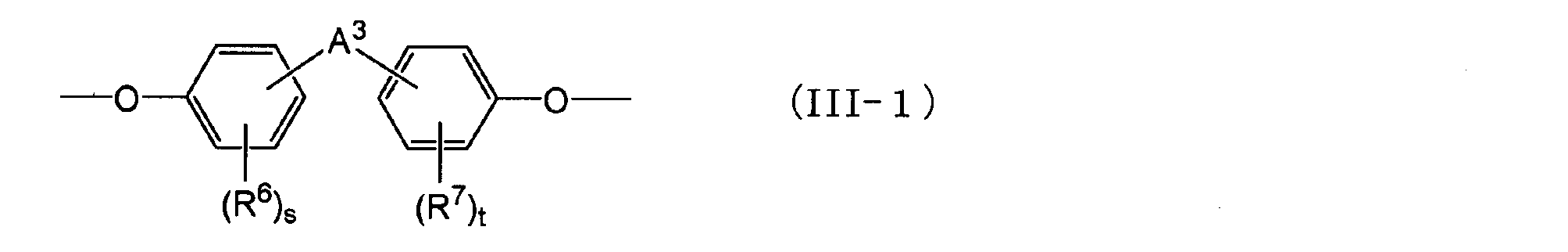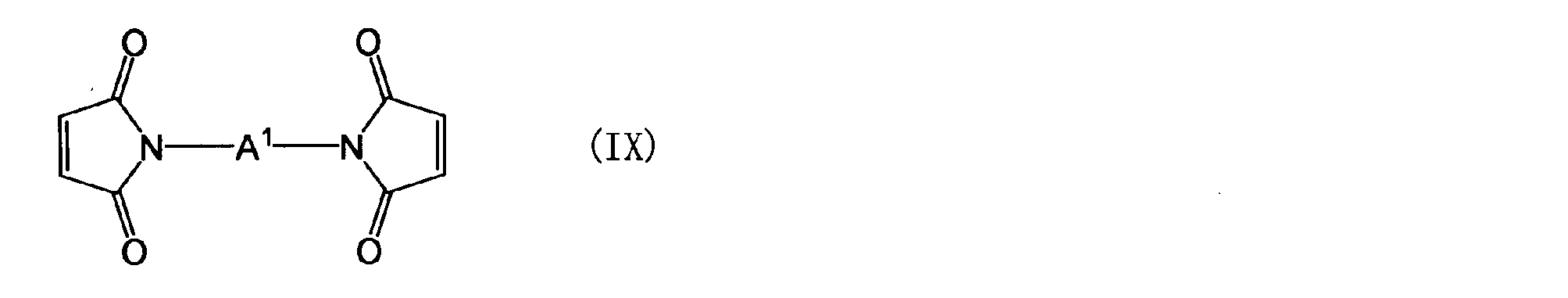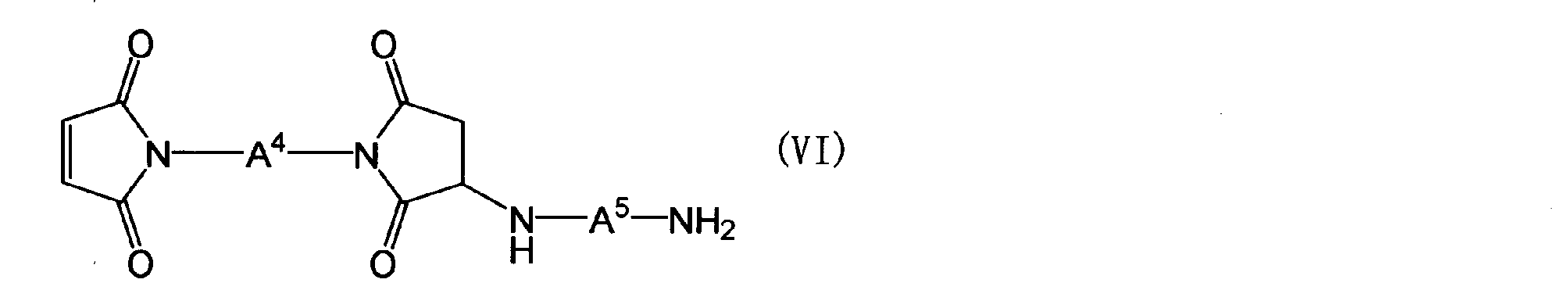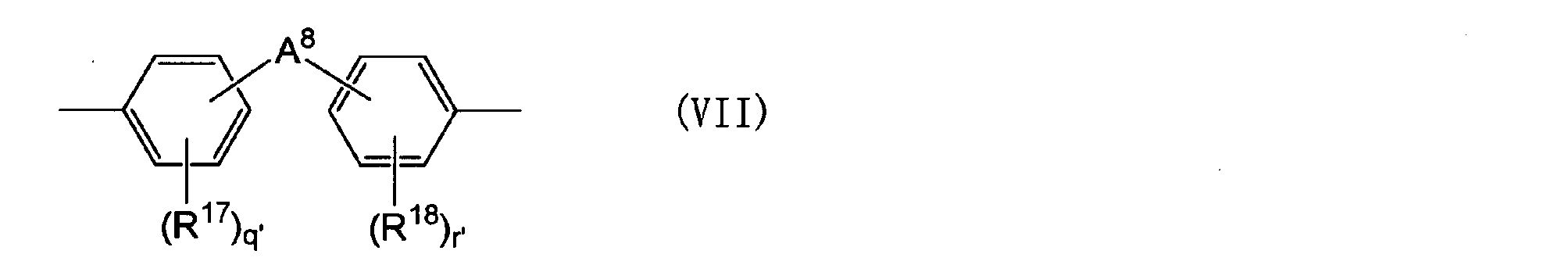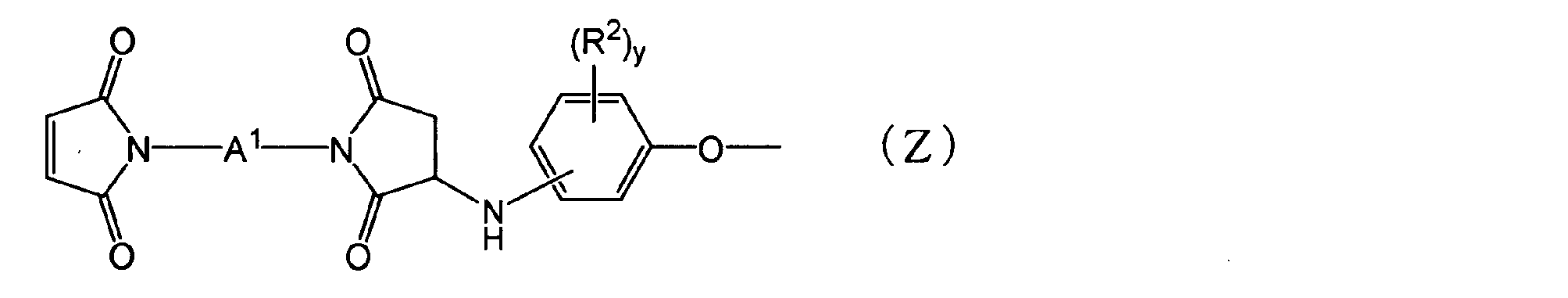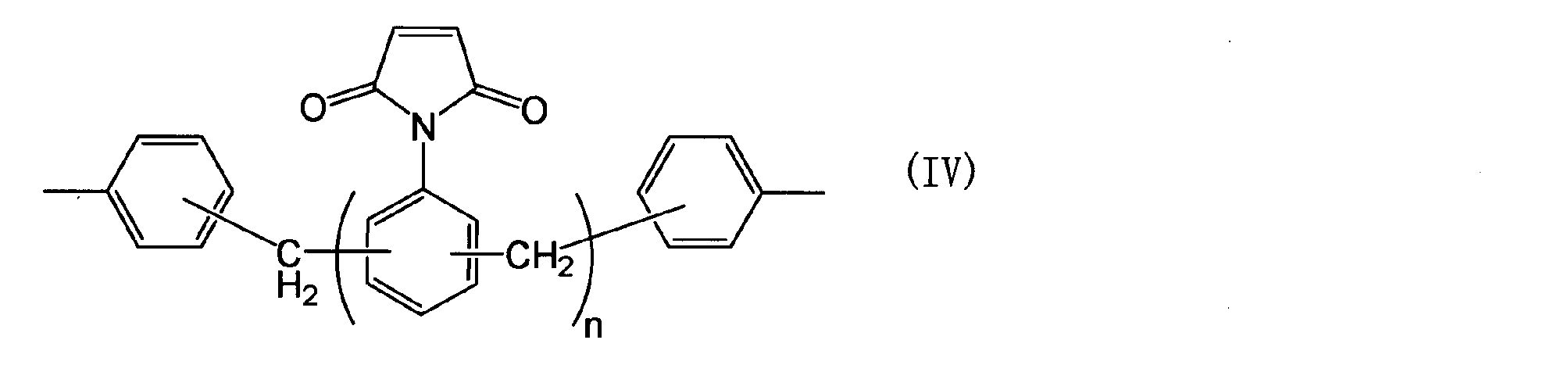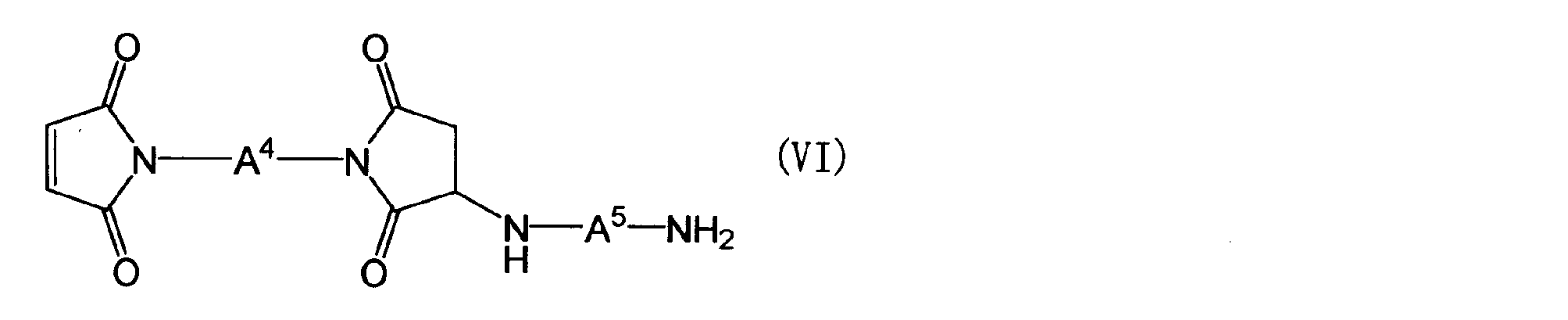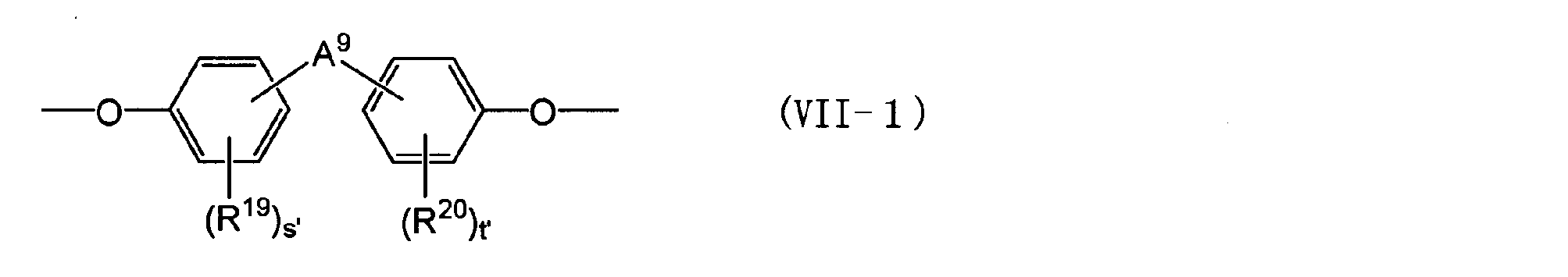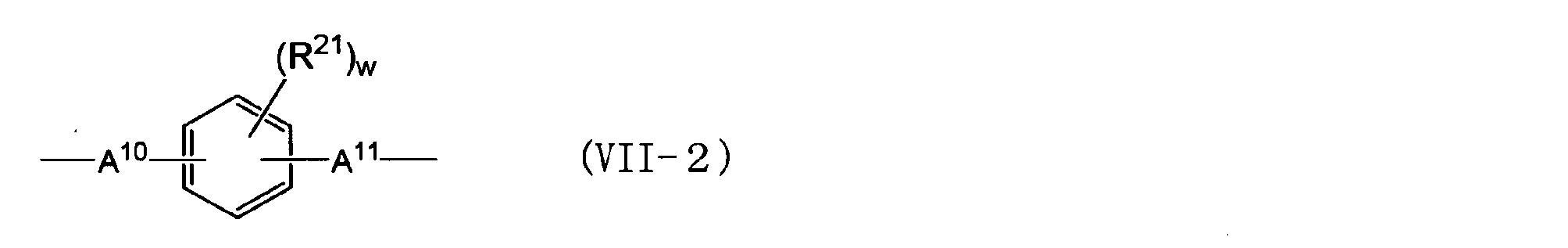WO2016175325A1 - 熱硬化性樹脂組成物、プリプレグ、積層板及び多層プリント配線板 - Google Patents
熱硬化性樹脂組成物、プリプレグ、積層板及び多層プリント配線板 Download PDFInfo
- Publication number
- WO2016175325A1 WO2016175325A1 PCT/JP2016/063497 JP2016063497W WO2016175325A1 WO 2016175325 A1 WO2016175325 A1 WO 2016175325A1 JP 2016063497 W JP2016063497 W JP 2016063497W WO 2016175325 A1 WO2016175325 A1 WO 2016175325A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- carbon atoms
- resin composition
- thermosetting resin
- integer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 N*NC(CC(N1*N(C(C=C2)=O)C2=O)=O)C1=O Chemical compound N*NC(CC(N1*N(C(C=C2)=O)C2=O)=O)C1=O 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N c1ccccc1 Chemical compound c1ccccc1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 2
- DBZVBKLKGDGTQE-UHFFFAOYSA-N CC(C)(c(cc1)ccc1Oc1ccc(C)cc1)c(cc1)ccc1Oc1ccc(C)cc1 Chemical compound CC(C)(c(cc1)ccc1Oc1ccc(C)cc1)c(cc1)ccc1Oc1ccc(C)cc1 DBZVBKLKGDGTQE-UHFFFAOYSA-N 0.000 description 1
- IVZSYJXREKSJTH-UHFFFAOYSA-N CC(Oc1ccccc1)P(OC)=O Chemical compound CC(Oc1ccccc1)P(OC)=O IVZSYJXREKSJTH-UHFFFAOYSA-N 0.000 description 1
- UOIWOHLIGKIYFE-UHFFFAOYSA-N CCCCCNC Chemical compound CCCCCNC UOIWOHLIGKIYFE-UHFFFAOYSA-N 0.000 description 1
- VCFUXSBUBBCXOE-UHFFFAOYSA-N CCc1c(C)c(C)cc(Cc2cc(CC)c(C)c(C)c2)c1 Chemical compound CCc1c(C)c(C)cc(Cc2cc(CC)c(C)c(C)c2)c1 VCFUXSBUBBCXOE-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1046—Polyimides containing oxygen in the form of ether bonds in the main chain
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/32—Polymers modified by chemical after-treatment
- C08G65/329—Polymers modified by chemical after-treatment with organic compounds
- C08G65/333—Polymers modified by chemical after-treatment with organic compounds containing nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/12—Unsaturated polyimide precursors
- C08G73/126—Unsaturated polyimide precursors the unsaturated precursors being wholly aromatic
- C08G73/127—Unsaturated polyimide precursors the unsaturated precursors being wholly aromatic containing oxygen in the form of ether bonds in the main chain
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/241—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres
- C08J5/244—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres using glass fibres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/249—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs characterised by the additives used in the prepolymer mixture
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/01—Use of inorganic substances as compounding ingredients characterized by their specific function
- C08K3/013—Fillers, pigments or reinforcing additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/0008—Organic ingredients according to more than one of the "one dot" groups of C08K5/01 - C08K5/59
- C08K5/0066—Flame-proofing or flame-retarding additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/34—Heterocyclic compounds having nitrogen in the ring
- C08K5/3412—Heterocyclic compounds having nitrogen in the ring having one nitrogen atom in the ring
- C08K5/3415—Five-membered rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/51—Phosphorus bound to oxygen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/51—Phosphorus bound to oxygen
- C08K5/52—Phosphorus bound to oxygen only
- C08K5/521—Esters of phosphoric acids, e.g. of H3PO4
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/51—Phosphorus bound to oxygen
- C08K5/52—Phosphorus bound to oxygen only
- C08K5/521—Esters of phosphoric acids, e.g. of H3PO4
- C08K5/523—Esters of phosphoric acids, e.g. of H3PO4 with hydroxyaryl compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/51—Phosphorus bound to oxygen
- C08K5/53—Phosphorus bound to oxygen bound to oxygen and to carbon only
- C08K5/5313—Phosphinic compounds, e.g. R2=P(:O)OR'
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/5399—Phosphorus bound to nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K9/00—Use of pretreated ingredients
- C08K9/04—Ingredients treated with organic substances
- C08K9/06—Ingredients treated with organic substances with silicon-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
- C08L71/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08L71/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08L71/12—Polyphenylene oxides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L71/00—Compositions of polyethers obtained by reactions forming an ether link in the main chain; Compositions of derivatives of such polymers
- C08L71/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08L71/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08L71/12—Polyphenylene oxides
- C08L71/126—Polyphenylene oxides modified by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08L79/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08L79/085—Unsaturated polyimide precursors
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K1/00—Printed circuits
- H05K1/02—Details
- H05K1/03—Use of materials for the substrate
- H05K1/0313—Organic insulating material
- H05K1/0353—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement
- H05K1/0366—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement reinforced, e.g. by fibres, fabrics
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K1/00—Printed circuits
- H05K1/02—Details
- H05K1/03—Use of materials for the substrate
- H05K1/0313—Organic insulating material
- H05K1/0353—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement
- H05K1/0373—Organic insulating material consisting of two or more materials, e.g. two or more polymers, polymer + filler, + reinforcement containing additives, e.g. fillers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2371/00—Characterised by the use of polyethers obtained by reactions forming an ether link in the main chain; Derivatives of such polymers
- C08J2371/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08J2371/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08J2371/12—Polyphenylene oxides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2379/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen, or carbon only, not provided for in groups C08J2361/00 - C08J2377/00
- C08J2379/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08J2379/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2463/00—Characterised by the use of epoxy resins; Derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2471/00—Characterised by the use of polyethers obtained by reactions forming an ether link in the main chain; Derivatives of such polymers
- C08J2471/08—Polyethers derived from hydroxy compounds or from their metallic derivatives
- C08J2471/10—Polyethers derived from hydroxy compounds or from their metallic derivatives from phenols
- C08J2471/12—Polyphenylene oxides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2479/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen, or carbon only, not provided for in groups C08J2461/00 - C08J2477/00
- C08J2479/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2479/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen, or carbon only, not provided for in groups C08J2461/00 - C08J2477/00
- C08J2479/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08J2479/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2203/00—Applications
- C08L2203/20—Applications use in electrical or conductive gadgets
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K1/00—Printed circuits
- H05K1/02—Details
- H05K1/03—Use of materials for the substrate
- H05K1/0313—Organic insulating material
Definitions
- the present invention relates to a thermosetting resin composition containing a polyphenylene ether derivative, a prepreg, a laminated board, and a multilayer printed wiring board.
- polyphenylene ether (PPE) resin has been used as a heat-resistant thermoplastic polymer excellent in high-frequency characteristics for printed wiring boards that require low transmission loss.
- PPE polyphenylene ether
- a method in which polyphenylene ether and a thermosetting resin are used in combination has been proposed.
- a resin composition containing polyphenylene ether and an epoxy resin for example, see Patent Document 1
- a resin composition containing polyphenylene ether and a cyanate resin having a low dielectric constant among thermosetting resins for example, Refer to Patent Document 2.
- Patent Documents 1 and 2 are generally insufficient in high-frequency characteristics in the GHz range, adhesion to conductors, low thermal expansion coefficient, flame retardancy, The heat resistance may decrease due to the low compatibility with the curable resin.
- the present inventors have made semi-IPN (semi-interpenetrating network) compatibility at the production stage (A stage stage) of a resin composition containing an organic solvent based on polyphenylene ether resin and polybutadiene resin.
- a resin composition for example, Patent Document 3
- PCB materials for printed wiring boards have high adhesion to conductors, a low coefficient of thermal expansion, a high thermal resistance, due to demands for higher density, higher reliability, and environmental friendliness. Further improvements in glass transition temperature, high flame retardancy, etc. are required.
- the adhesiveness to the conductor is 0.58 kN / m in terms of the peel strength of the copper foil when using a low profile copper foil (Rz: 1 to 2 ⁇ m) having a very low surface roughness on the resin adhesive surface side.
- Rz 1 to 2 ⁇ m
- 0.6 kN / m or more is desired.
- the glass transition temperature serving as a guide for these is 200 ° C. or higher, and that the thermal expansion coefficient (Z direction, Tg or lower) is 45 ppm / ° C. or lower, and further 43 ppm / ° C. or lower. ing.
- the thermal expansion coefficient Z direction, Tg or lower
- an inorganic filler is used to ensure resin flow for circuit filling. Is limited. Therefore, it is desirable to ensure the above required value even when the blending amount of the inorganic filler is relatively small.
- the dielectric constant of the substrate material when a general E glass base material is used is 3.8 or less, and further 3.7.
- the dielectric loss tangent is desired to be 0.007 or less, and further 0.006 or less.
- the substrate material generally shows a tendency that the dielectric loss tangent increases as the frequency increases.
- the conventional dielectric characteristic value at 1 to 5 GHz instead of the conventional dielectric characteristic value at 1 to 5 GHz, the necessity of satisfying the above required value at 10 GHz band or higher is increasing. Yes.
- the present invention has particularly good compatibility and dielectric characteristics (low dielectric constant and low dielectric loss tangent) in a high frequency band, high adhesion to a conductor, excellent heat resistance, high It is an object of the present invention to provide a thermosetting resin composition having a glass transition temperature, a low thermal expansion coefficient and high flame retardancy, a prepreg using the same, a laminate and a multilayer printed wiring board.
- thermosetting resin composition containing a polyphenylene ether derivative having a specific molecular structure, a specific thermosetting resin, and a phosphorus flame retardant
- the present invention is completed by finding that prepregs and laminates using a material exhibit excellent high-frequency characteristics, high heat resistance, high adhesion to conductors, high glass transition temperature, low thermal expansion coefficient and high flame retardancy. It came to. That is, the present invention relates to the following [1] to [14].
- thermosetting resin composition comprising: (In the formula, each R 1 is independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom. X is an integer of 0 to 4.) [2] The thermosetting resin composition according to the above [1], wherein the N-substituted maleimide structure-containing group is a group represented by the following general formula (Z).
- each R 2 independently represents an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- Y represents an integer of 0 to 4.
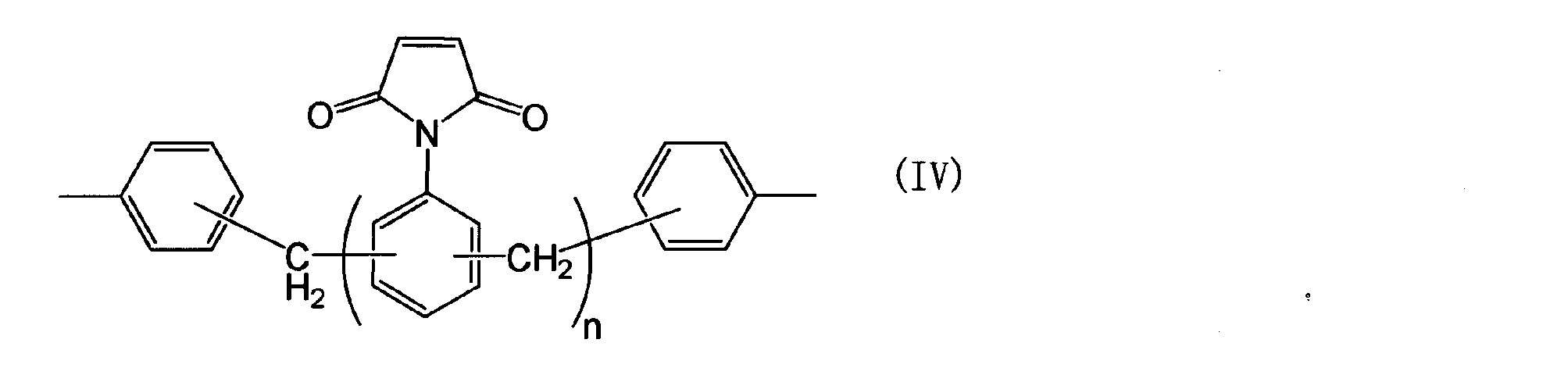
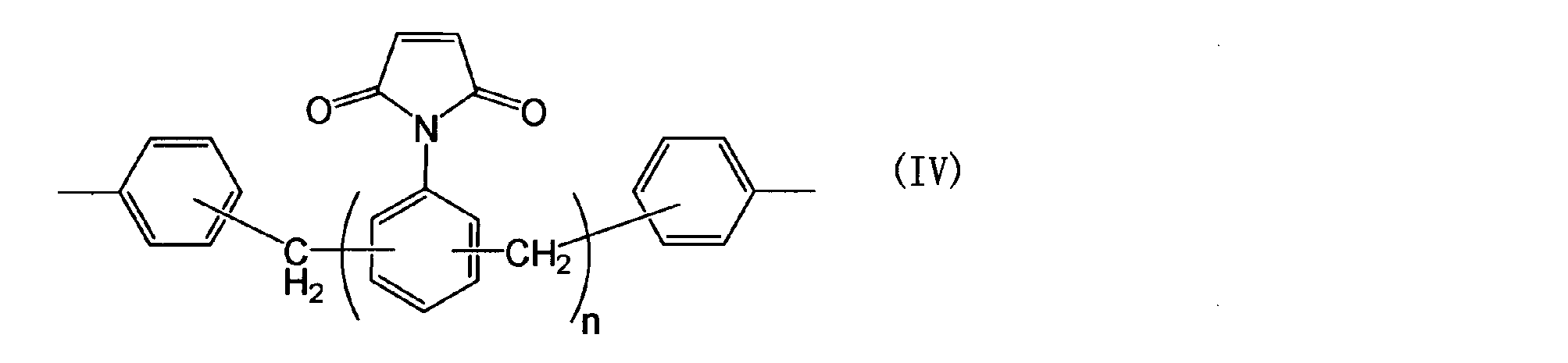
- a 1 represents the following general formula (II), ( III), a group represented by (IV) or (V).
- each R 3 independently represents an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- P is an integer of 0 to 4.
- R 4 and R 5 are each independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 2 is an alkylene group having 1 to 5 carbon atoms, an alkylidene having 2 to 5 carbon atoms A group, an ether group, a sulfide group, a sulfonyl group, a carbooxy group, a keto group, a single bond, or a group represented by the following general formula (III-1): q and r are each independently an integer of 0 to 4; is there.) (Wherein R 6 and R 7 are each independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 3 is an alkylene group having 1 to 5 carbon atoms, an isopropylidene group, an ether group, A sulfide group, a sulfonyl group, a carbooxy group, a keto group, or a single bond, and s and t are each independently an integer of 0 to 4.)
- n is an integer of 0 to 10.
- R 8 and R 9 are each independently a hydrogen atom or an aliphatic hydrocarbon group having 1 to 5 carbon atoms, u is an integer of 1 to 8)
- the thermosetting resin composition according to the above [1] or [2], wherein the structural unit represented by the general formula (I) is a structural unit represented by the following formula (I ′).
- thermosetting resin composition according to the above [2] or [3], wherein A 1 in the general formula (Z) is a group represented by any of the following formulas.
- a 1 in the general formula (Z) is a group represented by any of the following formulas.
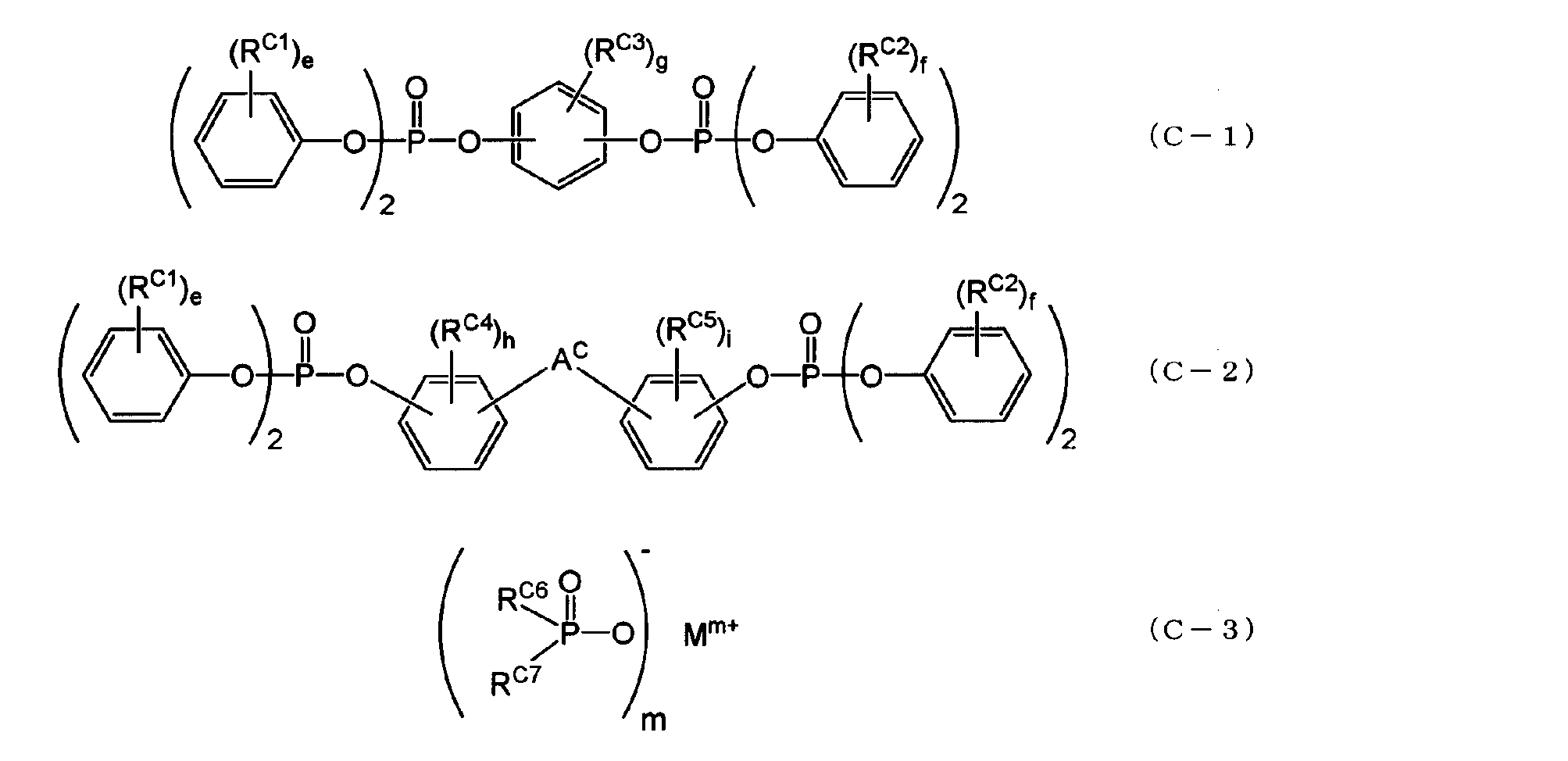
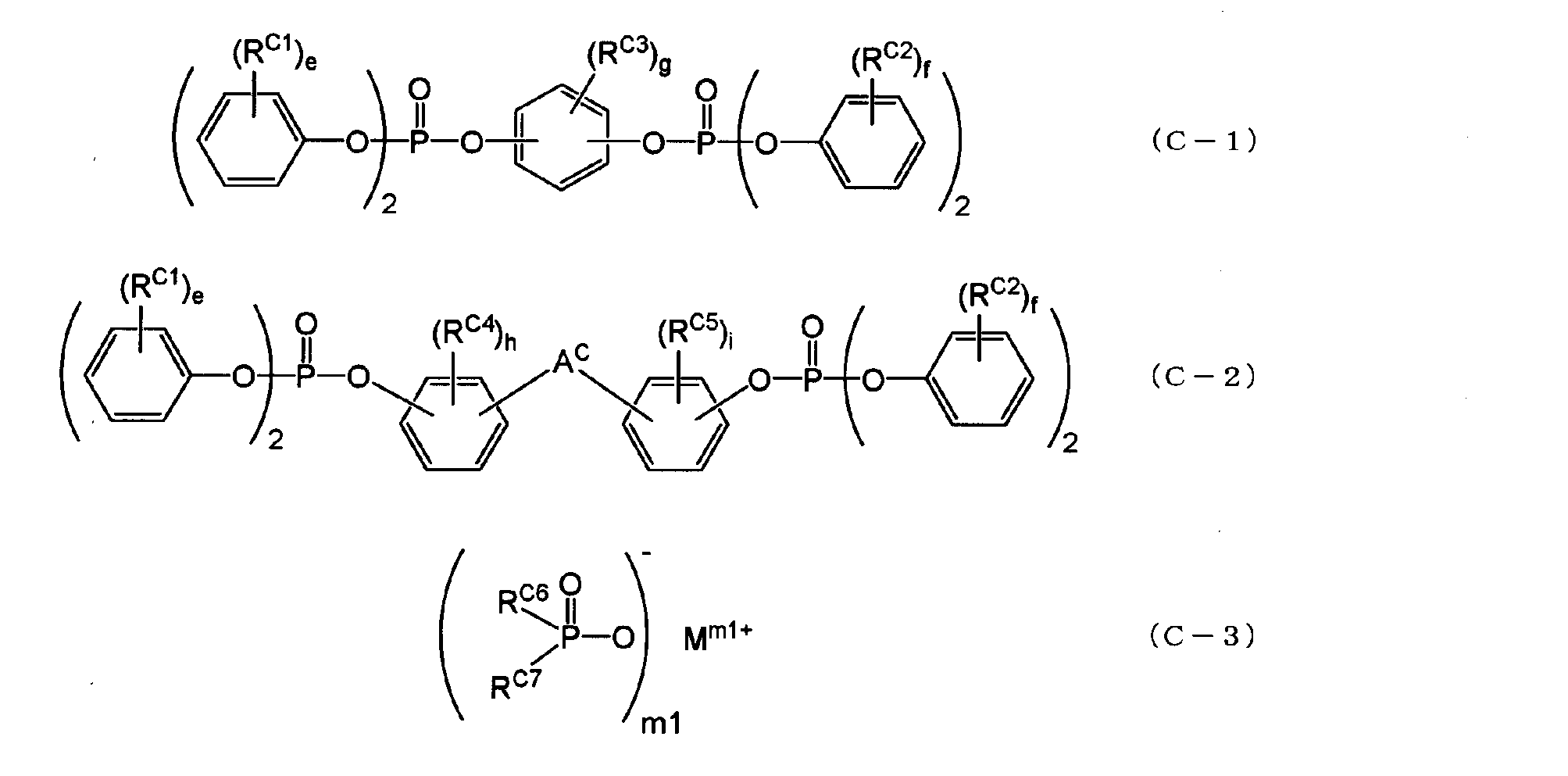
- the aromatic phosphate ester is represented by the following general formula (C-1) or (C-2), and the metal salt of the disubstituted phosphinic acid is represented by the following general formula (C-3).
- a c is an alkylene group having 1 to 5 carbon atoms, alkylidene of 2 to 5 carbon atoms A group, an ether group, a sulfide group, a sulfonyl group, a carbooxy group, a keto group, and a single bond, e and f are each independently an integer of 0 to 5, and g, h, and i are each independently an integer of 0 to 4. It is an integer.
- R C6 and R C7 are each independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or an aromatic hydrocarbon group having 6 to 14 carbon atoms.
- M is a lithium atom, a sodium atom, a potassium atom, a calcium atom, a magnesium atom, an aluminum atom, a titanium atom, or a zinc atom.
- m is an integer of 1 to 4.
- the maleimide compound in the component (B) is a polymaleimide compound (a) having at least two N-substituted maleimide groups in one molecule, or a polyaminobismaleimide represented by the following general formula (VI)
- a 4 is the same as the definition of A 1 in the general formula (Z)
- a 5 is a group represented by the following general formula (VII).
- R 17 and R 18 each independently represents an aliphatic hydrocarbon group having 1 to 5 carbon atoms, an alkoxy group having 1 to 5 carbon atoms, a hydroxyl group or a halogen atom.
- a 8 represents 1 to 5 carbon atoms.
- Q ′ and r ′ are each independently an integer of 0 to 4.)
- R 19 and R 20 each independently represents an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 9 represents an alkylene group having 1 to 5 carbon atoms, an isopropylidene group, m-phenyl group)
- R 21 is an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 10 and A 11 are each independently an alkylene group having 1 to 5 carbon atoms, an isopropylidene group, an ether group, A sulfide group, a sulfonyl group, a carbooxy group, a keto group, or a single bond, w is an integer of 0 to 4.)
- w is an integer of 0 to 4.
- thermosetting resin composition according to any one of [1] to [9], further including a curing accelerator (E).
- a prepreg comprising the thermosetting resin composition according to any one of [1] to [11] above and a sheet-like fiber reinforced base material.
- a laminate comprising the prepreg according to the above [12] and a metal foil.
- a multilayer printed wiring board comprising the prepreg according to [12] or the laminated board according to [13].
- thermosetting resin composition of the present invention has particularly good compatibility, excellent high-frequency characteristics (low dielectric constant, low dielectric loss tangent), high adhesion to conductors, excellent heat resistance, high glass transition temperature, It has a low coefficient of thermal expansion and high flame resistance. Therefore, the prepreg and laminated board obtained using this thermosetting resin composition can be used suitably for electronic component uses, such as a multilayer printed wiring board.
- thermosetting resin composition One embodiment of the present invention is a polyphenylene ether derivative (A) having an N-substituted maleimide structure-containing group and a structural unit represented by the following general formula (I) in one molecule [hereinafter simply referred to as a polyphenylene ether derivative (A) or (A) may be abbreviated as component]
- At least one thermosetting resin (B) selected from an epoxy resin, a cyanate resin, and a maleimide compound hereinafter, may be simply referred to as a thermosetting resin (B) or (B) component]
- phosphorus-based Flame retardant (C) phosphorus-based Flame retardant
- component (C) Is a thermosetting resin composition.
- each R 1 is independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- X is an integer of 0 to 4.
- the polyphenylene ether derivative (A) has an N-substituted maleimide structure-containing group and a structural unit represented by the general formula (I) in one molecule.
- the polyphenylene ether derivative (A) since it has at least one N-substituted maleimide structure-containing group in one molecule, it has excellent high frequency characteristics (low dielectric constant, low dielectric loss tangent), high adhesion to a conductor, It becomes a thermosetting resin composition having excellent heat resistance, high glass transition temperature, low thermal expansion coefficient and high flame retardancy.
- the thermal expansion coefficient referred to in the present invention is a value called a linear expansion coefficient.
- each R 1 is independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- the aliphatic hydrocarbon group include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, an isobutyl group, a t-butyl group, and an n-pentyl group.
- the aliphatic hydrocarbon group may be an aliphatic hydrocarbon group having 1 to 3 carbon atoms or a methyl group.
- R 1 may be an aliphatic hydrocarbon group having 1 to 5 carbon atoms.
- x is an integer of 0 to 4, may be an integer of 0 to 2, or may be 2.
- R 1 may be substituted at the ortho position on the benzene ring (provided that the substitution position of the oxygen atom is a reference).
- the plurality of R 1 may be the same or different.
- the structural unit represented by the general formula (I) may be a structural unit represented by the following general formula (I ′).
- the polyphenylene ether derivative (A) has an N-substituted maleimide structure-containing group, including high-frequency characteristics (low dielectric constant, low dielectric loss tangent), adhesion to conductors, heat resistance, glass transition temperature, thermal expansion coefficient, and flame retardancy From this point of view, it may be a group containing a bismaleimide structure in which nitrogen atoms of two maleimide groups are bonded via an organic group, or may be a group represented by the following general formula (Z) Good.
- each R 2 independently represents an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- Y represents an integer of 0 to 4.
- a 1 represents the following general formula (II), ( III), a group represented by (IV) or (V).)
- y is an integer of 0 to 4, may be an integer of 0 to 2, or may be 0.
- y is an integer of 2 or more, the plurality of R 2 may be the same or different.
- a 1 represents, formula (II), the group represented by (III), (IV) or (V) are as follows.
- each R 3 independently represents an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- P is an integer of 0 to 4.
- the aliphatic hydrocarbon group having 1 to 5 carbon atoms and the halogen atom represented by R 3 are described in the same manner as in R 1 .
- p is an integer of 0 to 4, and may be an integer of 0 to 2, 0, 1 or 0 from the viewpoint of availability. When p is an integer of 2 or more, the plurality of R 3 may be the same or different.
- R 4 and R 5 are each independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 2 is an alkylene group having 1 to 5 carbon atoms, an alkylidene having 2 to 5 carbon atoms A group, an ether group, a sulfide group, a sulfonyl group, a carbooxy group, a keto group, a single bond, or a group represented by the following general formula (III-1): q and r are each independently an integer of 0 to 4; is there.)
- Examples of the aliphatic hydrocarbon group having 1 to 5 carbon atoms and the halogen atom represented by R 4 and R 5 include the same as those in the case of R 1 .
- the aliphatic hydrocarbon group may be an aliphatic hydrocarbon group having 1 to 3 carbon atoms, and may be a methyl group, an ethyl group, or an ethyl group.
- Examples of the alkylene group having 1 to 5 carbon atoms represented by A 2 include a methylene group, a 1,2-dimethylene group, a 1,3-trimethylene group, a 1,4-tetramethylene group, a 1,5-pentamethylene group, and the like. Is mentioned.
- the alkylene group is an alkylene group having 1 to 3 carbon atoms from the viewpoint of high-frequency characteristics (low dielectric constant, low dielectric loss tangent), adhesion to a conductor, heat resistance, glass transition temperature, thermal expansion coefficient, and flame retardancy. It may be a methylene group.
- Examples of the alkylidene group having 2 to 5 carbon atoms represented by A 2 include an ethylidene group, a propylidene group, an isopropylidene group, a butylidene group, an isobutylidene group, a pentylidene group, and an isopentylidene group.
- an isopropylidene group may be used from the viewpoints of high-frequency characteristics (low dielectric constant, low dielectric loss tangent), adhesion to a conductor, heat resistance, glass transition temperature, thermal expansion coefficient, and flame retardancy.
- a 2 may be an alkylene group having 1 to 5 carbon atoms or an alkylidene group having 2 to 5 carbon atoms, among the above options.
- q and r are each independently an integer of 0 to 4, and from the viewpoint of easy availability, either may be an integer of 0 to 2, or may be 0 or 2. When q or r is an integer greater than or equal to 2, several R ⁇ 4 > or R ⁇ 5 > may be same or different, respectively.
- the groups represented by general formula (III-1) represented by A 2 are as follows.
- R 6 and R 7 are each independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 3 is an alkylene group having 1 to 5 carbon atoms, an isopropylidene group, an ether group, A sulfide group, a sulfonyl group, a carbooxy group, a keto group, or a single bond, and s and t are each independently an integer of 0 to 4.
- Examples of the aliphatic hydrocarbon group having 1 to 5 carbon atoms and the halogen atom represented by R 6 and R 7 are the same as those for R 4 and R 5 .
- Examples of the alkylene group having 1 to 5 carbon atoms represented by A 3 include the same groups as the alkylene group having 1 to 5 carbon atoms represented by A 2 .
- a 3 among the above options, an alkylidene group having 2 to 5 carbon atoms may be selected.
- s and t are integers of 0 to 4, and from the viewpoint of easy availability, each of them may be an integer of 0 to 2, 0 or 1, or 0.
- s or t is an integer of 2 or more, a plurality of R 6 s or R 7 s may be the same or different.
- n is an integer of 0 to 10.
- n may be 0 to 5 or 0 to 3 from the viewpoint of availability.
- R 8 and R 9 are each independently a hydrogen atom or an aliphatic hydrocarbon group having 1 to 5 carbon atoms, u is an integer of 1 to 8)
- the aliphatic hydrocarbon group having 1 to 5 carbon atoms and the halogen atom represented by R 8 and R 9 are described in the same manner as in the case of R 1 .
- u is an integer of 1 to 8, may be an integer of 1 to 3, or may be 1.
- a 1 in the group represented by the general formula (Z) includes high-frequency characteristics (low dielectric constant, low dielectric loss tangent), adhesion to a conductor, heat resistance, glass transition temperature, thermal expansion coefficient, and flame retardancy. From the viewpoint, a group represented by any of the following formulas may be used.
- the polyphenylene ether derivative (A) may be a polyphenylene ether derivative represented by the following general formula (A ′).
- a 1 , R 1 , R 2 , x and y are as defined above.
- M is an integer of 1 or more.
- m may be an integer of 1 to 300, an integer of 10 to 300, an integer of 30 to 200, or an integer of 50 to 150.
- the polyphenylene ether derivative (A) may be a polyphenylene ether derivative represented by any of the following formulas. (In the formula, m is the same as m in the general formula (A ′).)
- the raw material may be a polyphenylene ether derivative of the above formula (A′-1), and from the viewpoint of excellent dielectric properties and low water absorption, the above formula (A′-2) It may be a polyphenylene ether derivative, or may be a polyphenylene ether derivative of the above formula (A′-3) from the viewpoint of excellent adhesion to a conductor and mechanical properties (elongation, breaking strength, etc.). Therefore, one polyphenylene ether derivative represented by any one of the above formulas (A′-1) to (A′-3) is used alone or in combination of two or more according to the intended characteristics. Can do.
- the number average molecular weight of the polyphenylene ether derivative (A) of the present invention may be 5000 to 12000, 7000 to 12000, or 7000 to 10,000.
- the number average molecular weight is 5000 or more, a better glass transition temperature tends to be obtained in the thermosetting resin composition of the present invention, a prepreg and a laminate using the same.
- a number average molecular weight is 12000 or less, when the thermosetting resin composition of this invention is used for a laminated board, it exists in the tendency for a better moldability to be obtained.
- the number average molecular weight is a value converted from a calibration curve using standard polystyrene by gel permeation chromatography (GPC), and more specifically, measurement of the number average molecular weight described in Examples. The value obtained by the method.
- the polyphenylene ether derivative (A) can be obtained, for example, by the following production method.
- an aminophenol compound represented by the following general formula (VIII) [hereinafter referred to as aminophenol compound (VIII)] and, for example, a polyphenylene ether having a number average molecular weight of 15000 to 25000 are re-distributed in an organic solvent.
- the polyphenylene ether compound (A ′′) having a primary amino group in one molecule hereinafter, also simply referred to as polyphenylene ether compound (A ′′)
- the polyphenylene ether compound (A ′′) and the bismaleimide compound represented by the general formula (IX) [hereinafter referred to as bismaleimide compound (IX)]. ] Can be produced by a Michael addition reaction to produce a polyphenylene ether derivative (A).
- aminophenol compound (VIII) examples include o-aminophenol, m-aminophenol, and p-aminophenol.
- m-aminophenol and p-aminophenol are preferable from the viewpoint of the reaction yield when producing the polyphenylene ether compound (A ′′) and the heat resistance when the resin composition, prepreg and laminate are used. It may be p-aminophenol.
- the molecular weight of the polyphenylene ether compound (A ′′) can be controlled by the amount of aminophenol compound (VIII) used.
- the That is, the use amount of the aminophenol compound (VIII) may be appropriately adjusted so that the number average molecular weight of the finally produced polyphenylene ether derivative (A) falls within a suitable range.
- the compounding amount of the aminophenol compound (VIII) is not particularly limited.
- the polyphenylene ether to be reacted with the aminophenol compound (VIII) is 15000 to 25000
- the polyphenylene ether When used in the range of 0.5 to 6 parts by mass with respect to 100 parts by mass, the polyphenylene ether derivative (A) having a number average molecular weight of 5000 to 12000 can be obtained.
- the organic solvent used in the production process of the polyphenylene ether compound (A ′′) is not particularly limited.
- alcohols such as methanol, ethanol, butanol, butyl cellosolve, ethylene glycol monomethyl ether, propylene glycol monomethyl ether; acetone, methyl ethyl ketone Ketones such as methyl isobutyl ketone and cyclohexanone; aromatic hydrocarbons such as toluene, xylene and mesitylene; esters such as methoxyethyl acetate, ethoxyethyl acetate, butoxyethyl acetate and ethyl acetate; N, N-dimethylformamide, N, N And nitrogen-containing compounds such as dimethylacetamide and N-methyl-2-pyrrolidone. These may be used alone or in combination of two or more. Among these, toluene, xylene, and mesitylene may be used from the viewpoint
- a reaction catalyst can be used as necessary.
- this reaction catalyst a known reaction catalyst in a redistribution reaction can be applied.
- an organic peroxide such as t-butylperoxyisopropyl monocarbonate and a metal carboxylate such as manganese naphthenate May be used in combination.
- the amount of reaction catalyst used is not particularly limited.
- the organic peroxide is reduced to 0 with respect to 100 parts by mass of the polyphenylene ether to be reacted with the aminophenol compound (VIII).
- 0.5 to 5 parts by mass and 0.05 to 0.5 parts by mass of the carboxylic acid metal salt may be used.
- a predetermined amount of the aminophenol compound (VIII), the polyphenylene ether having a number average molecular weight of 15000 to 25000, an organic solvent, and, if necessary, a reaction catalyst is charged into a reactor, and the reaction is carried out while heating, keeping warm, and stirring. '') Is obtained.
- the reaction temperature and reaction time in this step the reaction conditions for known redistribution reactions can be applied.
- the reaction temperature is 70 to 110.
- the reaction may be carried out at a temperature of 1 ° C. for 1 to 8 hours.
- the solution of the polyphenylene ether compound (A ′′) produced as described above may be continuously supplied to the production process of the polyphenylene ether derivative (A) in the next step as it is. At this time, the solution of the polyphenylene ether compound (A ′′) may be cooled, or may be adjusted to the reaction temperature in the next step. Further, as described later, this solution may be concentrated as necessary to remove a part of the organic solvent, or may be diluted by adding an organic solvent.
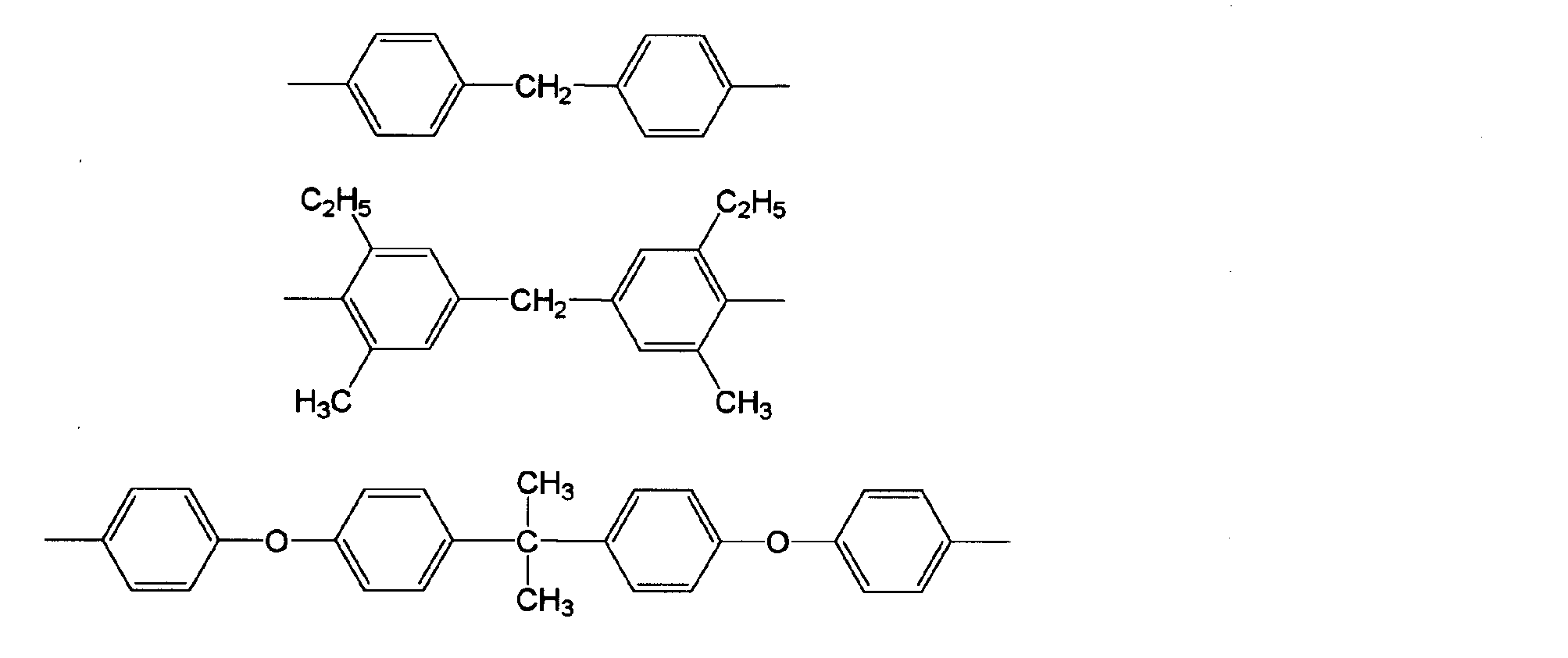
- Examples of the bismaleimide compound (IX) used in producing the polyphenylene ether derivative (A) include bis (4-maleimidophenyl) methane, polyphenylmethanemaleimide, bis (4-maleimidophenyl) ether, bis ( 4-maleimidophenyl) sulfone, 3,3′-dimethyl-5,5′-diethyl-4,4′-diphenylmethane bismaleimide, 4-methyl-1,3-phenylenebismaleimide, m-phenylenebismaleimide, 2, 2-bis (4- (4-maleimidophenoxy) phenyl) propane, bis (4-maleimidophenyl) sulfone, bis (4-maleimidophenyl) sulfide, bis (4-maleimidophenyl) ketone, 2,2-bis (4 -(4-maleimidophenoxy) phenyl) propane, bis ( - (4-maleimide phenoxy) phenyl)
- bis (4-maleimidophenyl) methane, 3,3′-dimethyl-5,5′-diethyl-4,4′-diphenylmethane bismaleimide, 2,2-bis (4- (4-maleimidophenoxy) Phenyl) propane may be selected.
- Bis (4-maleimidophenyl) methane may be used from the viewpoint that a polyphenylene ether derivative containing the formula (A′-1) is obtained and is inexpensive.
- the amount of bismaleimide compound (IX) used is determined by the amount of aminophenol compound (VIII) used. That is, the equivalent ratio (Tb1 / Ta1) of the —NH 2 group equivalent (Ta1) of the aminophenol compound (VIII) to the maleimide group equivalent (Tb1) of the bismaleimide compound (IX) can be in the range of 2-6. It may be blended in the range of 2-4.
- the bismaleimide compound within the above equivalent ratio range, in the thermosetting resin composition, prepreg and laminate of the present invention, more excellent heat resistance, high glass transition temperature and high flame retardance are obtained. It tends to be.
- a reaction catalyst can be used as necessary.
- the reaction catalyst that can be used is not particularly limited.
- an acidic catalyst such as p-toluenesulfonic acid
- an amine such as triethylamine, pyridine, and tributylamine
- an imidazole compound such as methylimidazole and phenylimidazole
- a triphenylphosphine examples thereof include phosphorus-based catalysts. These may be used alone or in combination of two or more.
- the amount of the reaction catalyst is not particularly limited, but may be, for example, 0.01 to 5 parts by mass with respect to 100 parts by mass of the polyphenylene ether compound (A ′′).
- the above-mentioned bismaleimide compound (IX) and, if necessary, a reaction catalyst, etc. are charged into a polyphenylene ether compound (A ′′) solution in a predetermined amount, and subjected to a Michael addition reaction while heating, keeping warm, and stirring, to thereby produce a polyphenylene ether derivative (A). Is obtained.
- the reaction conditions in this step may be, for example, a reaction temperature of 50 to 160 ° C. and a reaction time of 1 to 10 hours from the viewpoint of workability and gelation suppression.
- the organic solvent can be added or concentrated to adjust the reaction concentration (solid content concentration) and the solution viscosity.
- the organic solvent used additionally the organic solvent exemplified in the production process of the polyphenylene ether compound (A ′′) can be applied, and these may be used alone or in combination of two or more. Good. Of these, methyl ethyl ketone, cyclohexanone, propylene glycol monomethyl ether, N, N-dimethylformamide, and N, N-dimethylacetamide may be selected from the viewpoint of solubility.
- the reaction concentration (solid content concentration) in the production process of the polyphenylene ether derivative (A) and the polyphenylene ether compound (A ′′) is not particularly limited.
- 10 to 60% by mass It may be 20 to 50% by mass.
- the reaction concentration is 10% by mass or more, the reaction rate does not become too slow and tends to be more advantageous in terms of production cost.
- the reaction temperature is 60% by mass or less, better solubility tends to be obtained.
- the solution viscosity is low, the stirring efficiency is good, and gelation tends to be less.
- thermosetting resin composition of this invention the thermosetting resin composition of this invention
- the organic solvent in the solution may be partially or entirely removed and concentrated as appropriate. Additional dilution may be used. There is no restriction
- the production of the polyphenylene ether compound (A ′′) and the polyphenylene ether derivative (A) obtained by the above production process can be confirmed by GPC measurement and IR measurement after taking out a small amount of sample after the completion of each process.
- the polyphenylene ether compound (A ′′) has a molecular weight lower than that of a polyphenylene ether having a number average molecular weight of 15000 to 25000 from GPC measurement, and the peak of the aminophenol compound (VIII) as a raw material disappears. From the IR measurement, it can be confirmed that the desired polyphenylene ether compound (A ′′) is produced by the appearance of a primary amino group of 3300 to 3500 cm ⁇ 1 .
- the polyphenylene ether derivative (A) was purified by reprecipitation, and from the IR measurement, the disappearance of the peak of the primary amino group of 3300 to 3500 cm ⁇ 1 and the appearance of the peak of the carbonyl group of maleimide of 1700 to 1730 cm ⁇ 1 were observed. By confirming, it can confirm that the desired polyphenylene ether derivative (A) is manufactured.
- thermosetting resin composition of the present invention is more adhesive to the conductor, heat resistance, thermal expansion coefficient than the resin composition containing the polyphenylene ether compound (A ′′) and the component (B) described later. It tends to be more excellent in flame retardancy and workability (drilling, cutting).
- thermosetting resin (B) The component (B) contained in the thermosetting resin composition of the present invention is at least one thermosetting resin selected from an epoxy resin, a cyanate resin, and a maleimide compound.
- the maleimide compound does not include the polyphenylene ether derivative (A).
- the epoxy resin may be an epoxy resin having two or more epoxy groups in one molecule.
- the epoxy resin is classified into a glycidyl ether type epoxy resin, a glycidyl amine type epoxy resin, a glycidyl ester type epoxy resin, and the like.
- a glycidyl ether type epoxy resin may be selected.
- Epoxy resins are classified into various epoxy resins depending on the difference in the main skeleton. In each of the above types of epoxy resins, bisphenols such as bisphenol A type epoxy resin, bisphenol F type epoxy resin, and bisphenol S type epoxy resin are further used.
- Type epoxy resin alicyclic epoxy resin; aliphatic chain epoxy resin; phenol novolac type epoxy resin, cresol novolac type epoxy resin, bisphenol A novolac type epoxy resin, bisphenol F novolac type epoxy resin, etc .; phenol Aralkyl epoxy resin; Stilbene epoxy resin; Dicyclopentadiene epoxy resin; Naphthalene skeleton such as naphthol novolac epoxy resin and naphthol aralkyl epoxy resin It is classified into dicyclopentadiene type epoxy resin; type epoxy resins; biphenyl type epoxy resins; biphenyl aralkyl type epoxy resins; xylylene-type epoxy resins; dihydroanthracene type epoxy resin.
- An epoxy resin may be used individually by 1 type, and may use 2 or more types together.
- naphthalene skeleton-containing epoxy resins and biphenyl aralkyl epoxy resins may be used from the viewpoints of high-frequency characteristics, heat resistance, glass transition temperature, thermal expansion coefficient, flame retardancy, and the like.
- curing agent and hardening adjuvant can be used together as needed.
- polyamine compounds such as diethylenetriamine, triethylenetetramine, diaminodiphenylmethane, m-phenylenediamine, dicyandiamide; bisphenol A, phenol novolac resin, cresol novolac resin, bisphenol A novolak resin, phenol And polyphenol compounds such as aralkyl resins; acid anhydrides such as phthalic anhydride and pyromellitic anhydride; carboxylic acid compounds; and active ester compounds. These may be used alone or in combination of two or more.
- the amount used is not particularly limited and can be appropriately adjusted according to the purpose. Among these, from the viewpoints of heat resistance, glass transition temperature, thermal expansion coefficient, storage stability, and insulation reliability, polyphenol compounds and active ester compounds may be used.
- the cyanate resin is not particularly limited.
- Examples include benzene, a cyanate ester compound of a phenol-added dicyclopentadiene polymer, a phenol novolac-type cyanate ester compound, and a cresol novolac-type cyanate ester compound.
- Cyanate resin may be used individually by 1 type, and may use 2 or more types together.
- 2,2-bis (4-cyanatophenyl) propane may be used from the viewpoint of manufacturing cost and from the viewpoint of the overall balance of high-frequency characteristics and other characteristics.
- curing agent and hardening adjuvant of cyanate resin can be used together as needed.
- these are not specifically limited,
- curing agent and a hardening adjuvant is not restrict
- a monophenol compound may be used from the viewpoints of high-frequency characteristics, heat resistance, moisture absorption resistance, and storage stability.
- a monophenol compound is used in combination with the cyanate resin
- the monophenol compound to be used in combination may be blended in all specified amounts at the time of prepolymerization, or may be blended separately before and after prepolymerization, but from the viewpoint of storage stability, a method of blending separately Can be adopted.
- the maleimide compound is not particularly limited.
- a polymaleimide compound (a) having at least two N-substituted maleimide groups in one molecule [hereinafter sometimes referred to as component (a). ]
- a polyamino bismaleimide compound (c) represented by the following general formula (VI) [hereinafter sometimes referred to as component (c). ] can be contained.
- the maleimide compound may be a polyaminobismaleimide compound (c).
- the polyamino bismaleimide compound (c) is, for example, the component (a) and an aromatic diamine compound (b) having two primary amino groups in one molecule [hereinafter referred to as component (b). Can be obtained by Michael addition reaction in an organic solvent.
- a 4 is the same as the definition of A 1 in the general formula (Z), and A 5 is a group represented by the following general formula (VII).
- R 17 and R 18 are each independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms, an alkoxy group having 1 to 5 carbon atoms, a hydroxyl group or a halogen atom.
- An alkylene group having 1 to 5 carbon atoms) An alkylidene group having 2 to 5 carbon atoms, an ether group, a sulfide group, a sulfonyl group, a carbooxy group, a keto group, a fluorenylene group, a single bond, or the following general formula (VII-1) or (VII-2)
- Q ′ and r ′ are each independently an integer of 0 to 4.
- R 19 and R 20 each independently represents an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 9 represents an alkylene group having 1 to 5 carbon atoms, an isopropylidene group, m-phenyl group)
- R 21 is an aliphatic hydrocarbon group having 1 to 5 carbon atoms or a halogen atom.
- a 10 and A 11 are each independently an alkylene group having 1 to 5 carbon atoms, an isopropylidene group, an ether group, A sulfide group, a sulfonyl group, a carbooxy group, a keto group, or a single bond, w is an integer of 0 to 4.
- R 1 in the general formula (I) may be the same.
- the aliphatic hydrocarbon group may be an aliphatic hydrocarbon group having 1 to 3 carbon atoms, and may be a methyl group or an ethyl group.
- a 4 in the general formula (VI) is the same as the definition of A 1 in the general formula (Z).
- the alkylene group having 1 to 5 carbon atoms represented by A 10 and A 11 in the general formula (VII-2) is described in the same manner as in the case of A 2 in the general formula (III).
- q ′ and r ′ are integers of 0 to 4, and from the viewpoint of availability, both of them may be integers of 0 to 2, and may be 0 or 2.
- s ′ and t ′ are integers of 0 to 4, and from the viewpoint of availability, both may be integers of 0 to 2, may be 0 or 1, or may be 0.
- w is an integer of 0 to 4, and may be an integer of 0 to 2 or 0 from the viewpoint of availability.
- the component (a) is not particularly limited, and for example, the same component as the bismaleimide compound (IX) may be applied.
- the component (a) include bis (4-maleimidophenyl) methane, polyphenylmethanemaleimide, bis (4-maleimidophenyl) ether, bis (4-maleimidophenyl) sulfone, 3,3′-dimethyl-5, 5′-diethyl-4,4′-diphenylmethane bismaleimide, 4-methyl-1,3-phenylene bismaleimide, m-phenylene bismaleimide, 2,2-bis (4- (4-maleimidophenoxy) phenyl) propane, Bis (4-maleimidophenyl) sulfone, bis (4-maleimidophenyl) sulfide, bis (4-maleimidophenyl) ketone, 2,2-bis (4- (4-maleimidophenoxy) phenyl) propane, bis (4- ( 4-maleimid
- a component may be used individually by 1 type according to the objective, a use, etc., and may use 2 or more types together.
- the component (a) may be a bismaleimide compound, and may be bis (4-maleimidophenyl) methane from the viewpoint of being inexpensive, and has excellent dielectric properties and low water absorption. From a certain point of view, 3,3′-dimethyl-5,5′-diethyl-4,4′-diphenylmethane bismaleimide may be used, and it has high adhesion to the conductor and mechanical properties (elongation, breaking strength, etc.) From the standpoint of superiority, 2,2-bis (4- (4-maleimidophenoxy) phenyl) propane may be used.
- the polyamino bismaleimide compound (c) is obtained by Michael addition of the component (a) and the aromatic diamine compound (b) having two primary amino groups in one molecule in an organic solvent. It can be obtained by reacting.
- the component (b) is not particularly limited.
- 4,4′-diaminodiphenylmethane, 4,4′-diamino-3,3 is highly soluble in organic solvents, has a high reaction rate during synthesis, and can have high heat resistance.
- 4,4′-diaminodiphenylmethane, 4,4′-diamino-3,3′-dimethyl-diphenylmethane, 4,4 ′ from the viewpoint of low cost -Diamino-3,3'-diethyl-diphenylmethane may be used.
- 2,2-bis (4- (4-aminophenoxy) phenyl) propane It may be 4 ′-[1,3-phenylenebis (1-methylethylidene)] bisaniline and 4,4 ′-[1,4-phenylenebis (1-methylethylidene)] bisaniline.
- 4,4 ′-[1,3-phenylenebis (1- Methylethylidene)] bisaniline, 4,4 ′-[1,4-phenylenebis (1-methylethylidene)] bisaniline may be selected. These may be used alone or in combination of two or more according to the purpose and application.
- the organic solvent used in producing the polyaminobismaleimide compound (c) is not particularly limited, and for example, the organic solvent exemplified in the production process of the polyphenylene ether compound (A ′′) can be applied. These may be used alone or in combination of two or more. Among these, methyl ethyl ketone, cyclohexanone, propylene glycol monomethyl ether, N, N-dimethylformamide, and N, N-dimethylacetamide may be used from the viewpoint of solubility.
- the amount of the component (a) and the component (b) used in the production of the component (c) is as follows: —NH 2 group equivalent (Ta2) of the component (b) and maleimide group equivalent (Tb2) of the component (a) Equivalent ratio (Tb2 / Ta2) may be in the range of 1 to 10, or in the range of 2 to 10.
- reaction catalyst In the Michael addition reaction for producing the polyaminobismaleimide compound (c), it is not necessary to use a reaction catalyst, but it can also be used as necessary. Although it does not restrict
- the blending amount of the reaction catalyst is not particularly limited as described above.
- a maleimide compound curing agent such as styrene monomer, divinylbenzene and divinylbiphenyl; (meth) acrylate compounds; allyl compounds such as triallyl cyanurate and triallyl isocyanurate; diaminodiphenylmethane and the like
- the polyamine compounds may be used alone or in combination of two or more. These usage amounts are not particularly limited, and can be appropriately adjusted according to the purpose.
- vinyl compounds and polyamine compounds may be used from the viewpoints of high-frequency characteristics and heat resistance.
- a polyamino bismaleimide compound (c) is obtained by charging a predetermined amount of the above component (a), component (b), an organic solvent and, if necessary, a reaction catalyst into a reactor, followed by Michael addition reaction while heating, keeping warm, and stirring. .
- the reaction conditions such as the reaction temperature and reaction time in this step, for example, the reaction conditions during the Michael addition reaction during the production of the polyphenylene ether derivative (A) described above can be applied.
- the reaction concentration (solid content concentration) is not particularly limited, but may be 10 to 90% by mass or 20 to 80% by mass. When the reaction concentration is 10 mass or more, the reaction rate does not become too slow and tends to be more advantageous in terms of production cost.
- the content of the component (A) is not particularly limited, but it is preferably 3% by mass or more with respect to 100 parts by mass of the total of the components (A) to (C) from the viewpoint of high frequency characteristics, and 5% by mass. More preferably.
- the content of the component (B) is not particularly limited, but is 10 to 90% by mass with respect to 100 parts by mass of the sum of the components (A) to (C) from the viewpoint of high frequency characteristics and moldability. It is preferably 20 to 80% by mass or more.
- the content ratio [(A) :( B)] of the component (A) and the component (B) is not particularly limited, but may be 5:95 to 80:20 by mass ratio.
- the content ratio of the component (A) with respect to the total amount of the component (A) and the component (B) is 5% by mass or more, more excellent high frequency characteristics and low hygroscopicity tend to be obtained. Moreover, if it is 80 mass% or less, it exists in the tendency for the more excellent heat resistance, the more excellent moldability, and the more excellent workability to be obtained.
- thermosetting resin composition of the present invention further contains a phosphorus-based flame retardant (C).
- a phosphorus-based flame retardant C
- Low dielectric loss tangent high adhesion to the conductor, excellent heat resistance, low thermal expansion coefficient and high glass transition temperature.
- a phosphorus flame retardant (C) may be used individually by 1 type, and may use 2 or more types together.
- the phosphorus-based flame retardant (C) is not particularly limited as long as it contains a phosphorus atom among those generally used as a flame retardant, and may be an inorganic phosphorus-based flame retardant, It may be an organic phosphorus flame retardant. From the viewpoint of environmental problems, it is preferable not to contain a halogen atom. From the viewpoints of high-frequency characteristics (low dielectric constant, low dielectric loss tangent), adhesion to conductors, heat resistance, glass transition temperature, thermal expansion coefficient, and flame retardancy, organic phosphorus flame retardants may be used.
- inorganic phosphorus flame retardants include red phosphorus; ammonium phosphates such as monoammonium phosphate, diammonium phosphate, triammonium phosphate and ammonium polyphosphate; inorganic nitrogen-containing phosphorus compounds such as phosphate amides Phosphoric acid; phosphine oxide and the like.
- organic phosphorus flame retardants include aromatic phosphoric acid esters, monosubstituted phosphonic acid diesters, disubstituted phosphinic acid esters, disubstituted phosphinic acid metal salts, organic nitrogenous phosphorus compounds, and cyclic organic phosphorus compounds. Is mentioned.
- an aromatic phosphate compound and a metal salt of a disubstituted phosphinic acid may be selected.
- the “metal salt” may be any one of a lithium salt, a sodium salt, a potassium salt, a calcium salt, a magnesium salt, an aluminum salt, a titanium salt, and a zinc salt, or an aluminum salt.
- aromatic phosphates may be used.
- aromatic phosphate ester examples include triphenyl phosphate, tricresyl phosphate, trixylenyl phosphate, cresyl diphenyl phosphate, cresyl di-2,6-xylenyl phosphate, resorcinol bis (diphenyl phosphate), 1,3 -Phenylenebis (di-2,6-xylenyl phosphate), bisphenol A-bis (diphenyl phosphate), 1,3-phenylenebis (diphenyl phosphate) and the like.
- Examples of monosubstituted phosphonic acid diesters include divinyl phenylphosphonate, diallyl phenylphosphonate, and bis (1-butenyl) phenylphosphonate.
- Examples of the disubstituted phosphinic acid ester include phenyl diphenylphosphinate and methyl diphenylphosphinate.
- Examples of the metal salt of disubstituted phosphinic acid include a metal salt of dialkylphosphinic acid, a metal salt of diallylphosphinic acid, a metal salt of divinylphosphinic acid, and a metal salt of diarylphosphinic acid.
- these metal salts may be any of lithium salt, sodium salt, potassium salt, calcium salt, magnesium salt, aluminum salt, titanium salt, and zinc salt, and an aluminum salt may be selected.
- organic nitrogen-containing phosphorus compounds include phosphazene compounds such as bis (2-allylphenoxy) phosphazene and dicresyl phosphazene; melamine compounds such as melamine phosphate, melamine pyrophosphate, melamine polyphosphate, and melam polyphosphate. Can be mentioned.
- cyclic organophosphorus compounds include 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide, 10- (2,5-dihydroxyphenyl) -9,10-dihydro-9-oxa-10- And phosphaphenanthrene-10-oxide.
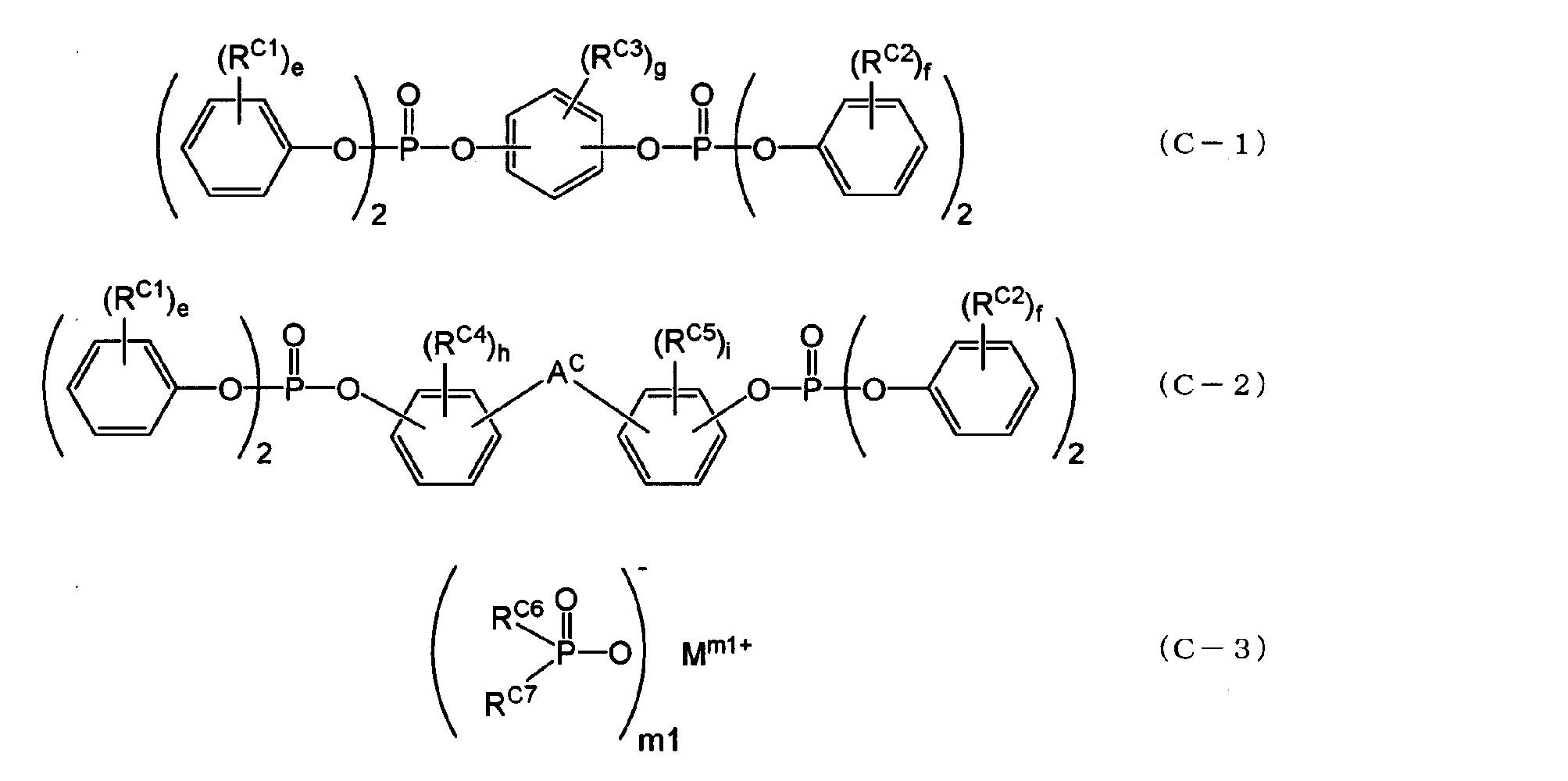
- the aromatic phosphate ester may be an aromatic phosphate ester represented by the following general formula (C-1) or (C-2), and the metal salt of the disubstituted phosphinic acid is: It may be a metal salt of a disubstituted phosphinic acid represented by the general formula (C-3).
- a c is an alkylene group having 1 to 5 carbon atoms, alkylidene of 2 to 5 carbon atoms A group, an ether group, a sulfide group, a sulfonyl group, a carbooxy group, a keto group, and a single bond, e and f are each independently an integer of 0 to 5, and g, h, and i are each independently an integer of 0 to 4. It is an integer.
- R C6 and R C7 are each independently an aliphatic hydrocarbon group having 1 to 5 carbon atoms or an aromatic hydrocarbon group having 6 to 14 carbon atoms.
- M is a lithium atom, a sodium atom, a potassium atom, a calcium atom, a magnesium atom, an aluminum atom, a titanium atom, or a zinc atom.
- m1 is an integer of 1 to 4.
- Examples of the aliphatic hydrocarbon group having 1 to 5 carbon atoms and the halogen atom represented by R C1 to R C5 include the same as those in the case of R 1 in the general formula (I).
- Alkylidene group alkylene group and having 2 to 5 carbon atoms having 1 to 5 carbon atoms represented by A c is described as in the case of the A 2.
- Ac may be an alkylene group having 1 to 5 carbon atoms, an alkylidene group having 2 to 5 carbon atoms, a single bond, an isopropylidene group, or a single bond among the above options.
- e and f may be an integer of 0 to 2, or may be 2.
- g, h, and i may be integers of 0 to 2, may be 0 or 1, and may be 0.
- Examples of the aliphatic hydrocarbon group having 1 to 5 carbon atoms represented by R C6 and R C7 are the same as those in the case of R 1 in the general formula (I).
- the aliphatic hydrocarbon group may be an aliphatic hydrocarbon group having 1 to 3 carbon atoms or an ethyl group.
- the aromatic hydrocarbon group having 6 to 14 carbon atoms represented by R C6 and R C7 include a phenyl group, a naphthyl group, a biphenylyl group, and an anthryl group.
- the aromatic hydrocarbon group may be an aromatic hydrocarbon group having 6 to 10 carbon atoms.
- m1 represents the valence of the metal ion, that is, changes within the range of 1 to 4 corresponding to the type of M.
- M may be an aluminum atom. Note that m1 is 3 when M is an aluminum atom.
- the content ratio of the phosphorus-based flame retardant (C) in the thermosetting resin composition of the present invention is not particularly limited.
- the thermosetting resin composition in terms of solid content excluding the component (D) described later
- the content of phosphorus atoms in the sum of the other components may be 0.2 to 5% by mass, or 0.3 to 3% by mass.
- the phosphorus atom content is 0.2% by mass or more, better flame retardancy tends to be obtained.
- content of a phosphorus atom is 5 mass% or less, it exists in the tendency for better moldability, more high adhesiveness with a conductor, more excellent heat resistance, and a high glass transition temperature.
- thermosetting resin composition of the present invention may optionally be referred to as an inorganic filler (D) [hereinafter referred to as component (D) as required. ] And a hardening accelerator (E) [Hereafter, it may be called (E) component. ] May be included.
- D inorganic filler
- E hardening accelerator
- various characteristics when it is used as a laminate can be further improved. For example, a low thermal expansion coefficient, high elastic modulus, heat resistance, and flame retardance can be improved by arbitrarily including an appropriate inorganic filler (D) in the thermosetting resin composition of the present invention.
- thermosetting resin composition is improved, and the high frequency characteristics, heat resistance, adhesion to the conductor, elastic modulus, and glass transition temperature are improved. be able to.
- flame retardants and flame retardant aids other than the phosphorus flame retardant (C) may be used in combination with the component (C).
- the component (D) is not particularly limited.
- examples include magnesium, aluminum hydroxide, aluminum silicate, calcium carbonate, calcium silicate, magnesium silicate, silicon nitride, boron nitride, clay (fired clay, etc.), talc, aluminum borate, aluminum borate, silicon carbide, and the like. . These may be used alone or in combination of two or more.
- silica, alumina, mica, talc, silica, alumina, or silica may be used from the viewpoints of thermal expansion coefficient, elastic modulus, heat resistance, and flame retardancy.
- the silica include a precipitated silica produced by a wet method and having a high water content, and a dry method silica produced by a dry method and containing almost no bound water, and the dry method silica further includes a difference in production method. Crushed silica, fumed silica, fused silica (fused spherical silica) and the like.
- the shape and particle size of the inorganic filler (D) are not particularly limited.
- the particle size may be 0.01 to 20 ⁇ m or 0.1 to 10 ⁇ m.
- the particle diameter means an average particle diameter, and is a particle diameter at a point corresponding to a volume of 50% when a cumulative frequency distribution curve based on the particle diameter is obtained with the total volume of the particles being 100%. It can be measured by a particle size distribution measuring apparatus using a laser diffraction scattering method.
- the content ratio of the component (D) in the thermosetting resin composition is not particularly limited, but from the viewpoint of thermal expansion coefficient, elastic modulus, heat resistance and flame retardancy,
- the content of component (D) in the thermosetting resin composition may be 3 to 65% by volume, 5 to 60% by volume, or 15 to 55% by volume.
- a coupling agent is used in combination as necessary for the purpose of improving the dispersibility of the component (D) and the adhesion between the component (D) and the organic component in the resin composition. May be.
- the coupling agent is not particularly limited, and for example, a silane coupling agent or a titanate coupling agent can be appropriately selected and used.
- a coupling agent may be used individually by 1 type, and may use 2 or more types together.
- the amount of the coupling agent used is not particularly limited, and may be 0.1 to 5 parts by mass, for example, 0.5 to 3 parts by mass with respect to 100 parts by mass of component (D). There may be. If it is this range, there will be little fall of various characteristics and it exists in the tendency which can exhibit the characteristic by use of said (D) component effectively.
- a method using an inorganic filler surface-treated by a wet method can be adopted. By adopting this method, the characteristics of the component (D) can be expressed more effectively.
- the component (D) When the component (D) is contained, it is used as a slurry in which the component (D) is dispersed in an organic solvent in advance, for the purpose of improving the dispersibility of the component (D) in the thermosetting resin composition. be able to.
- the organic solvent illustrated by the manufacturing process of the polyphenylene ether compound (A '') mentioned above is applicable. These may be used alone or in combination of two or more. Among these, methyl ethyl ketone, methyl isobutyl ketone, and cyclohexanone can be selected from the viewpoint of dispersibility.
- the solid content (nonvolatile content) concentration of the slurry is not particularly limited, but may be, for example, 50 to 80% by mass from the viewpoint of sedimentation and dispersibility of the inorganic filler (D), and 60 to 80%. It may be mass%.
- thermosetting resin composition of the present invention a suitable (E) component can be used according to the type of the (B) component to be used.
- suitable (E) component when an epoxy resin is used as the component (B) include imidazole compounds and derivatives thereof; tertiary amine compounds; quaternary ammonium compounds; and phosphorus compounds such as triphenylphosphine. . These may be used alone or in combination of two or more. Among these, from the viewpoint of heat resistance, glass transition temperature, and storage stability, an imidazole compound and its derivative or phosphorus compound may be used.
- imidazole compound examples include methyl imidazole, phenyl imidazole, and isocyanate mask imidazole (for example, an addition reaction product of hexamethylene diisocyanate resin and 2-ethyl-4-methyl imidazole). May be.
- Examples of the component (E) when a cyanate resin is used as the component (B) include imidazole compounds and derivatives thereof; carboxylates such as manganese, cobalt, and zinc; acetylacetone complexes of transition metals such as manganese, cobalt, and zinc; And organometallic compounds. These may be used alone or in combination of two or more. Among these, an organic metal compound may be used from the viewpoints of heat resistance, glass transition temperature, and storage stability.
- examples of the component (E) include acidic catalysts such as p-toluenesulfonic acid; amine compounds such as triethylamine, pyridine, and tributylamine; methylimidazole, phenylimidazole, isocyanate mask imidazole (Eg, addition reaction product of hexamethylene diisocyanate resin and 2-ethyl-4-methylimidazole); tertiary amine compound; quaternary ammonium compound; phosphorus compound such as triphenylphosphine; dicumyl Peroxide, 2,5-dimethyl-2,5-bis (t-butylperoxy) hexyne-3,2,5-dimethyl-2,5-bis (t-butylperoxy) hexane, t-butylperoxy Isopropyl monocarbonate, ⁇ , ⁇ ' -Organic peroxides such as
- an imidazole compound from the viewpoint of heat resistance, glass transition temperature and storage stability, it may be an imidazole compound, an organic peroxide, or a carboxylate salt, and from the viewpoint of heat resistance, glass transition temperature, elastic modulus, and thermal expansion coefficient. Therefore, an imidazole compound and an organic peroxide may be used in combination.
- organic peroxides ⁇ , ⁇ '-bis (t-butylperoxy) diisopropylbenzene may be selected.
- the content ratio of the (E) component is not particularly limited, but for example, the (A) component and the (B) component of the present invention.
- the total amount may be 0.01 to 10 parts by mass, or 0.01 to 5 parts by mass.
- the thermosetting resin composition of the present invention may contain a flame retardant other than the phosphorus-based flame retardant (C) and a flame retardant aid as long as the effects of the present invention are not impaired.
- a flame retardant other than the phosphorus-based flame retardant (C) from the viewpoint of environmental problems, for example, a metal hydrate such as a hydrate of aluminum hydroxide or a hydrate of magnesium hydroxide may be used. These may be used alone or in combination of two or more.
- the metal hydroxide may correspond to an inorganic filler, but is classified as a flame retardant in the case of a material that can impart flame retardancy.
- thermosetting resin composition of the present invention contains a flame retardant other than the phosphorus flame retardant (C)
- the content of the flame retardant is 50 masses with respect to 100 parts by mass of the phosphorus flame retardant (C). Part or less, 30 parts by weight or less, or 15 parts by weight or less.
- the thermosetting resin composition of the present invention may contain a flame retardant aid, for example, an inorganic flame retardant aid such as antimony trioxide and zinc molybdate.
- a flame retardant aid for example, an inorganic flame retardant aid such as antimony trioxide and zinc molybdate.
- the content ratio is not particularly limited, but for example, with respect to 100 parts by mass of the sum of the component (A) and the component (B)
- the amount may be 0.1 to 20 parts by mass, or 0.1 to 10 parts by mass.
- thermosetting resin composition of the present invention includes, as necessary, resin materials such as known thermoplastic resins and elastomers, as well as coupling agents, antioxidants, thermal stabilizers, antistatic agents, and ultraviolet rays. Absorbers, pigments, colorants, lubricants and the like can be appropriately selected and contained. These may be used alone or in combination of two or more. Moreover, these usage-amounts are not specifically limited.
- the resin composition of the present invention may contain an organic solvent from the viewpoint of facilitating handling by dilution and easy manufacture of a prepreg described later.
- organic solvent include, but are not limited to, alcohol solvents such as ethanol, propanol, butanol, methyl cellosolve, butyl cellosolve, and propylene glycol monomethyl ether; ketone solvents such as acetone, methyl ethyl ketone, methyl isobutyl ketone, and cyclohexanone; tetrahydrofuran Ether solvents such as toluene; xylene, mesitylene and other aromatic solvents; dimethylformamide, dimethylacetamide, N-methylpyrrolidone and other nitrogen atom containing solvents; dimethyl sulfoxide and other sulfur atom containing solvents; ⁇ -butyrolactone and other esters System solvents and the like.
- alcohol solvents, ketone solvents, nitrogen atom-containing solvents may be used, ketone solvents may be used, and acetone, methyl ethyl ketone, methyl isobutyl ketone, and cyclohexanone may be used. It can be methyl ethyl ketone.
- One organic solvent may be used alone, or two or more organic solvents may be used in combination.
- the content of the organic solvent in the thermosetting resin composition of the present invention is not particularly limited, but the solid content may be 30 to 90% by mass, 40 to 80% by mass, It may be -70% by mass or 40-60% by mass.
- thermosetting resin composition having a solid content concentration within the above range, the handleability becomes easy, the impregnation property to the base material and the appearance of the prepreg produced are good, Adjustment of the solid content concentration of the resin is facilitated, and production of a prepreg having a desired thickness tends to be facilitated.
- thermosetting resin composition of the present invention can be obtained by mixing the components (A) to (C), other components used in combination as necessary, and an organic solvent as necessary, by a known method. Can do. At this time, it may be dissolved or dispersed while stirring. Conditions such as mixing order, temperature and time during mixing and stirring are not particularly limited and can be arbitrarily set.
- the glass transition temperature when a laminate is prepared from the thermosetting resin composition of the present invention is not particularly limited, but has good heat resistance and through-hole connection reliability, and excellent workability when manufacturing electronic components, etc. From this point of view, it may be 175 ° C. or higher, 180 ° C. or higher, or 190 ° C. or higher.
- the upper limit of the glass transition temperature is not particularly limited, but may be, for example, 1000 ° C. or less, 500 ° C. or less, 300 ° C. or less, or 230 ° C. or less.
- the coefficient of thermal expansion (Z direction, Tg or less) when a laminate is produced from the thermosetting resin composition of the present invention is not particularly limited, but is 45 ppm / ° C.
- the glass transition temperature and the thermal expansion coefficient are values measured in accordance with the IPC standard as described in the examples.
- the dielectric constant and dielectric loss tangent are not particularly limited.
- the dielectric constant at 10 Ghz is preferably small, may be 3.8 or less, may be 3.75 or less, or may be 3.65 or less.
- the lower limit of the dielectric constant is not particularly limited, but may be, for example, 0.5 or more, 1 or more, 3 or more, or 3.5 or more.
- the dielectric loss tangent is preferably small, and may be 0.007 or less, or 0.006 or less.
- the lower limit of the dielectric constant is not particularly limited and may be as small as possible.
- the dielectric constant and dielectric loss tangent are values measured according to JPCA-TM001 (triplate resonator method) as in the examples.
- the present invention also provides a prepreg comprising the resin composition of the present invention and a sheet-like fiber reinforced substrate.
- the prepreg is formed using the resin composition of the present invention and a sheet-like fiber reinforced substrate, and more specifically, impregnating or impregnating the sheet-like fiber reinforced substrate of the present invention with the thermosetting resin composition of the present invention. It can be obtained by coating and drying. More specifically, for example, the prepreg of the present invention can be produced by drying by heating in a drying furnace at a temperature of usually 80 to 200 ° C. for 1 to 30 minutes and semi-curing (B-stage). .
- the amount of the thermosetting resin composition used can be determined so that the solid content concentration derived from the thermosetting resin composition in the prepreg after drying is 30 to 90% by mass. When the solid content concentration is within the above range, when a laminate is obtained, better formability tends to be obtained.
- the prepreg sheet-like fiber reinforced substrate known materials used for various types of laminates for electrical insulating materials are used.
- the material for the sheet-like reinforcing substrate include inorganic fibers such as E glass, D glass, S glass, and Q glass; organic fibers such as polyimide, polyester, and tetrafluoroethylene; and mixtures thereof.
- These sheet-like reinforcement base materials have shapes, such as a woven fabric, a nonwoven fabric, a robink, a chopped strand mat, a surfacing mat, for example.
- the thickness of the sheet-like fiber reinforced base material is not particularly limited, and for example, a thickness of 0.02 to 0.5 mm can be used.
- the surface treated with a coupling agent or the like, or the one mechanically subjected to the fiber opening treatment is impregnated with the resin composition, heat resistance when used as a laminate, moisture resistance, and workability. Can be preferably used.
- the hot melt method is a method in which an organic solvent is not contained in a thermosetting resin composition, and (1) a coated paper having good peelability from the composition is once coated and laminated on a sheet-like reinforcing substrate. Or (2) A method of directly coating the sheet-like reinforcing substrate with a die coater.
- the solvent method an organic solvent is contained in the thermosetting resin composition, and a sheet-like reinforcing base material is immersed in the obtained thermosetting resin composition, so that the thermosetting resin composition is made into a sheet-like reinforcing group. In this method, the material is impregnated and then dried.
- the laminate of the present invention comprises the prepreg of the present invention and a metal foil.
- the laminated plate of the present invention is formed using the prepreg of the present invention and a metal foil. More specifically, the metal plate is arranged on one or both sides of one prepreg of the present invention, or the prepreg of the present invention. It can be obtained by placing a metal foil on one side or both sides of a prepreg obtained by stacking two or more sheets, and then heat-pressing.
- the metal of the metal foil is not particularly limited as long as it is used for electrical insulating materials, but from the viewpoint of conductivity, copper, gold, silver, nickel, platinum, molybdenum, ruthenium, aluminum, tungsten, iron, titanium , Chromium, or an alloy containing at least one of these metal elements, copper, aluminum, or copper.
- the conditions for the heat and pressure molding are not particularly limited. For example, the temperature is 100 ° C. to 300 ° C., the pressure is 0.2 to 10.0 MPa, and the time is 0.1 to 5 hours. Can do.
- the heat and pressure molding can employ a method in which a vacuum state is maintained for 0.5 to 5 hours using a vacuum press or the like.
- the multilayer printed wiring board of this invention contains the prepreg or laminated board of this invention.
- the multilayer printed wiring board of the present invention is formed using the prepreg or laminate of the present invention, and using the prepreg or laminate of the present invention, a known method for drilling, metal plating, metal It can be manufactured by performing circuit formation processing such as etching of the foil and multilayer adhesion processing.
- thermosetting resin composition, resin film, prepreg, metal-clad laminate, and multilayer printed wiring board of the present invention can be suitably used for electronic devices that handle high-frequency signals of 1 GHz or higher, particularly high-frequency signals of 10 GHz or higher. It can use suitably for the electronic device to handle.
- perbutyl (registered trademark) I [t-butylperoxyisopropyl monocarbonate, manufactured by NOF Corporation, trade name] (2 parts by mass) and manganese naphthenate [manufactured by Wako Pure Chemical Industries, Ltd.] ( 0.15 parts by mass) was added and reacted at a solution temperature of 90 ° C. for 4 hours, and then cooled to 70 ° C. to obtain a polyphenylene ether compound (A ′) having a primary amino group at the molecular end. A small amount of this reaction solution was taken out and measured by gel permeation chromatography (GPC).
- GPC gel permeation chromatography
- the peak derived from p-aminophenol disappeared and the number average molecular weight of the polyphenylene ether compound (A ′) was about 9200. Met. Further, when a small amount of the reaction solution taken out was dropped into a methanol / benzene mixed solvent (mixing mass ratio: 1: 1) and reprecipitated, and the solid content (reaction product) purified by FT-IR measurement, 3400 cm ⁇ 1 was measured. The appearance of a peak derived from a nearby primary amino group was confirmed.
- the number average molecular weight was converted from a calibration curve using standard polystyrene by gel permeation chromatography (GPC).
- the calibration curve is standard polystyrene: TSK standard POLYSTYRENE (Type; A-2500, A-5000, F-1, F-2, F-4, F-10, F-20, F-40) [manufactured by Tosoh Corporation, The product name] was used to approximate the cubic equation.
- the measurement conditions for GPC are shown below.
- BMI-4000 2,2-bis (4- (4-maleimidophenoxy) phenyl) propane, manufactured by Daiwa Kasei Kogyo Co., Ltd., trade name] (4.5 parts by mass), propylene glycol monomethyl was added to the above reaction solution.
- Ether (10 parts by mass) was added, the temperature of the solution was raised while stirring, and the reaction was allowed to proceed for 4 hours while maintaining at 120 ° C., followed by cooling and filtration through a 200 mesh filter, polyphenylene ether derivative (A-1) Manufactured.
- thermosetting resin (B) A 1 L glass flask container with a thermometer, a reflux condenser, and a stirrer that can be heated and cooled, and BMI-4000 [2,2-bis (4- (4-maleimidophenoxy) phenyl) propane, Yamato Kasei Kogyo Co., Ltd., trade name] (100 parts by mass), Bisaniline-M [4,4 '-[1,3-phenylenebis (1-methylethylidene)] bisaniline, Mitsui Chemicals, trade name] (14 (Mass parts) and propylene glycol monomethyl ether (50 parts by mass), with the liquid temperature kept at 120 ° C., the mixture was reacted for 3 hours with stirring, then cooled and filtered through a 200 mesh filter, and a thermosetting resin. A polyaminobismaleimide compound (B-1) as (B) was produced.
- thermosetting resin composition having a solid content (nonvolatile content) concentration of 40 to 60% by mass was prepared by stirring and mixing while heating at room temperature according to the blending amount (unit: parts by mass).
- the density of the resin composition (excluding the inorganic filler) is usually 1.20 to 1.25 g / cm 3 , and the density of the inorganic filler used is 2. From 2 to 3.01 g / cm 3 , when 80 parts by mass of the inorganic filler is added to 100 parts by mass of the resin composition (excluding the inorganic filler), the amount is about 30 to 34% by volume.
- thermosetting resin composition (1. Compatibility evaluation of thermosetting resin composition)
- thermosetting resin composition obtained in each example was applied to a glass cloth [E glass, manufactured by Nitto Boseki Co., Ltd.] having a thickness of 0.1 mm, and then heat-dried at 160 ° C. for 7 minutes to obtain a resin content.
- Resin content A prepreg of about 54% by mass was produced.
- These 6 prepregs are stacked, and on the top and bottom, low profile copper foil [trade name “FV-WS”, M surface Rz: 1.5 ⁇ m, manufactured by Furukawa Electric Co., Ltd.] 18 ⁇ m thick is in contact with the M surface.
- the copper clad laminate obtained above was evaluated for formability, dielectric properties, copper foil peeling strength, glass transition temperature, thermal expansion coefficient, solder heat resistance and flame retardancy.
- the characteristic evaluation method of the copper clad laminate is as follows.
- (2-2-1) Formability The formability was evaluated by observing the appearance of the laminated plate obtained by etching the copper foils on both sides. The moldability was evaluated visually and evaluated according to the following criteria. A: There is no abnormality in appearance. C: There are some unevenness, streaks, blurring, voids, etc., and the surface smoothness is poor. (2-2-2) Dielectric properties The dielectric properties (relative permittivity and dielectric loss tangent) were measured in accordance with JPCA-TM001 (Triplate Resonator Method) at 1 GHz band and 10 GHz band.
- surface means the number of sheets in which abnormality, such as generation
- Tg Glass Transition Temperature
- Tg Thermal Expansion Coefficient Glass transition temperature
- Tg thermal expansion coefficient
- each material in Table 1 is as follows.
- Flame retardant OP-935 Dialkylphosphinic acid aluminum salt [metal salt of disubstituted phosphinic acid, phosphorus content: 23.5% by mass, manufactured by Clariant Corporation]
- PX-200 1,3-phenylenebis (di-2,6-xylenyl phosphate), [aromatic phosphate ester, phosphorus content: 9% by mass, manufactured by Daihachi Chemical Industry Co., Ltd.]
- PX-202 4,4′-biphenylene-tetrakis (2,6-dimethylphenyl phosphate) [aromatic phosphate ester, phosphorus content: 8% by mass, manufactured by Daihachi Chemical Industry Co., Ltd.]
- SPB-100 Polybisphenoxyphosphazene [Organic nitrogen-containing phosphorus compound, phosphorus content: 13% by mass, manufactured by Otsuka Chemical Co., Ltd.] HCA-
- Inorganic filler SC-2050 KNK: spherical fused silica, average particle size: 0.5 ⁇ m, surface treatment: vinylsilane coupling agent (1% by mass / solid content), dispersion medium: methyl isobutyl ketone, solid content concentration 70% by mass, density 2.2 g / cm 3 , manufactured by Admatechs Co., Ltd., trade name (3) curing accelerator, perbutyl (registered trademark) P: ⁇ , ⁇ ′-bis (t-butylperoxy) diisopropylbenzene, Made by NOF Corporation, trade name G-8809L: Isocyanate mask imidazole (addition reaction product of hexamethylene diisocyanate resin and 2-ethyl-4-methylimidazole), trade name made by Daiichi Kogyo Seiyaku Co., Ltd.
- the compatibility of the thermosetting resin composition and the appearance of the prepreg are excellent, and the copper-clad laminate produced using these is molded. It is good and balanced in all of the properties, high-frequency characteristics (dielectric characteristics), adhesion to the conductor, solder heat resistance, glass transition temperature, thermal expansion coefficient and flame retardancy. On the other hand, as shown in Table 1, in some comparative examples, the compatibility of the resin composition and the appearance of the prepreg are not good (Comparative Examples 1 to 3).
- the copper clad laminated board of each comparative example is not more favorable than an Example in a moldability, a dielectric characteristic, adhesiveness with a conductor, solder heat resistance, a glass transition temperature, a thermal expansion coefficient, and a flame retardance.
- thermosetting resin composition having high adhesiveness, excellent heat resistance, high glass transition temperature, low thermal expansion coefficient, and high flame retardancy of V-0 is obtained.
- thermosetting resin composition can keep raw material costs and substrate material manufacturing costs low, and is excellent in work environment. Therefore, a prepreg and a laminate provided by using this thermosetting resin composition Can be suitably used for electronic component applications such as multilayer printed wiring boards.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- General Chemical & Material Sciences (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Inorganic Chemistry (AREA)
- Reinforced Plastic Materials (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Laminated Bodies (AREA)
- Polyethers (AREA)
Abstract
Description
また、近年の環境問題から、鉛フリーはんだによる電子部品の実装やハロゲンフリーによる難燃化が要求されるようになってきたため、プリント配線板用材料にはこれまでよりも高い耐熱性や難燃性が必要とされている。
すなわち、本発明は、下記[1]~[14]に関するものである。
エポキシ樹脂、シアネート樹脂及びマレイミド化合物からなる群より選ばれる少なくとも1種類の熱硬化性樹脂(B)、及び
リン系難燃剤(C)
を含む熱硬化性樹脂組成物。
(式中、R1は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。xは0~4の整数である。)
[2]前記N-置換マレイミド構造含有基が下記一般式(Z)で表される基である、上記[1]に記載の熱硬化性樹脂組成物。
(式中、R2は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。yは0~4の整数である。A1は、下記一般式(II)、(III)、(IV)又は(V)で表される基である。)
(式中、R3は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。pは0~4の整数である。)
(式中、R4及びR5は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A2は炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、単結合、又は下記一般式(III-1)で表される基である。q及びrは各々独立に0~4の整数である。)
(式中、R6及びR7は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A3は炭素数1~5のアルキレン基、イソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。s及びtは各々独立に0~4の整数である。)
(式中、nは0~10の整数である。)
(式中、R8及びR9は各々独立に、水素原子又は炭素数1~5の脂肪族炭化水素基である。uは1~8の整数である。)
[3]前記一般式(I)で表される構造単位が、下記式(I’)で表される構造単位である、上記[1]又は[2]に記載の熱硬化性樹脂組成物。
[4]前記一般式(Z)中のA1が下記式のいずれかで表される基である、上記[2]又は[3]に記載の熱硬化性樹脂組成物。
[5]前記リン系難燃剤(C)が、芳香族リン酸エステル、及び2置換ホスフィン酸の金属塩から選択される少なくとも1種類である、上記[1]~[4]のいずれか1つに記載の熱硬化性樹脂組成物。
[6]前記芳香族リン酸エステルが下記一般式(C-1)又は(C-2)で表され、前記2置換ホスフィン酸の金属塩が下記一般式(C-3)で表される、上記[5]に記載の熱硬化性樹脂組成物。
(式中、RC1~RC5は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。Acは炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、単結合である。e及びfは各々独立に0~5の整数であり、g、h及びiは各々独立に0~4の整数である。
RC6及びRC7は各々独立に、炭素数1~5の脂肪族炭化水素基又は炭素数6~14の芳香族炭化水素基である。Mは、リチウム原子、ナトリウム原子、カリウム原子、カルシウム原子、マグネシウム原子、アルミニウム原子、チタン原子、亜鉛原子である。mは、1~4の整数である。)
[7]前記(B)成分中のマレイミド化合物が、1分子中に少なくとも2個のN-置換マレイミド基を有するポリマレイミド化合物(a)、又は下記一般式(VI)で表されるポリアミノビスマレイミド化合物(c)である、上記[1]~[6]のいずれか1つに記載の熱硬化性樹脂組成物。
(式中、A4は前記一般式(Z)中のA1の定義と同じであり、A5は下記一般式(VII)で表される基である。)
(式中、R17及びR18は各々独立に、炭素数1~5の脂肪族炭化水素基、炭素数1~5のアルコキシ基、水酸基又はハロゲン原子である。A8は炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、フルオレニレン基、単結合、又は下記一般式(VII-1)もしくは(VII-2)で表される基である。q’及びr’は各々独立に0~4の整数である。)
(式中、R19及びR20は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A9は炭素数1~5のアルキレン基、イソプロピリデン基、m-フェニレンジイソプロピリデン基、p-フェニレンジイソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。s’及びt’は各々独立に0~4の整数である。)
(式中、R21は炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A10及びA11は各々独立に、炭素数1~5のアルキレン基、イソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。wは0~4の整数である。)
[8](A)成分と(B)成分の含有割合[(A):(B)]が、質量比で、5:95~80:20である、上記[1]~[7]のいずれか1つに記載の熱硬化性樹脂組成物。
[9]更に無機充填剤(D)を含有する、上記[1]~[8]のいずれか1つに記載の熱硬化性樹脂組成物。
[10]更に硬化促進剤(E)を含有する、上記[1]~[9]のいずれか1つに記載の熱硬化性樹脂組成物。
[11]更に有機溶媒を含有する、上記[1]~[10]のいずれか1つに記載の熱硬化性樹脂組成物。
[12]上記[1]~[11]のいずれか1つに記載の熱硬化性樹脂組成物とシート状繊維補強基材と含有してなるプリプレグ。
[13]上記[12]に記載のプリプレグと金属箔と含有してなる積層板。
[14]上記[12]に記載のプリプレグ又は上記[13]に記載の積層板を含有してなる多層プリント配線板。
本発明の一態様は、1分子中にN-置換マレイミド構造含有基及び下記一般式(I)で表される構造単位を有するポリフェニレンエーテル誘導体(A)[以下、単にポリフェニレンエーテル誘導体(A)又は(A)成分と略称することがある]、
エポキシ樹脂、シアネート樹脂及びマレイミド化合物から選択される少なくとも1種類の熱硬化性樹脂(B)[以下、単に熱硬化性樹脂(B)又は(B)成分と略称することがある]、及び
リン系難燃剤(C)[以下、(C)成分と略称することがある]
を含む熱硬化性樹脂組成物である。
(式中、R1は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。xは0~4の整数である。)
以下、各成分について順に説明する。
ポリフェニレンエーテル誘導体(A)は、1分子中にN-置換マレイミド構造含有基及び前記一般式(I)で表される構造単位を有する。特に、ポリフェニレンエーテル誘導体(A)が1分子中に少なくとも1個のN-置換マレイミド構造含有基を有することにより、優れた高周波特性(低誘電率、低誘電正接)、導体との高接着性、優れた耐熱性、高ガラス転移温度、低熱膨張係数及び高難燃性を有する熱硬化性樹脂組成物となる。ここで、本発明でいう熱膨張係数は、線膨張係数とも呼ばれる値である。
前記一般式(I)中のR1は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。該脂肪族炭化水素基としては、例えば、メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基、イソブチル基、t-ブチル基、n-ペンチル基等が挙げられる。該脂肪族炭化水素基としては、炭素数1~3の脂肪族炭化水素基であってもよく、メチル基であってもよい。また、ハロゲン原子としては、例えば、フッ素原子、塩素原子、臭素原子、ヨウ素原子等が挙げられる。ハロゲン原子としては、ハロゲンフリーとする観点から、フッ素原子であってもよい。
以上の中でも、R1としては炭素数1~5の脂肪族炭化水素基であってもよい。
xは0~4の整数であり、0~2の整数であってもよく、2であってもよい。なお、xが1又は2である場合、R1はベンゼン環上のオルト位(但し、酸素原子の置換位置を基準とする。)に置換していてもよい。また、xが2以上である場合、複数のR1同士は同一であっても異なっていてもよい。
前記一般式(I)で表される構造単位としては、具体的には、下記一般式(I')で表される構造単位であってもよい。
(式中、R2は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。yは0~4の整数である。A1は、下記一般式(II)、(III)、(IV)又は(V)で表される基である。)
yは0~4の整数であり、0~2の整数であってもよく、0であってもよい。yが2以上の整数である場合、複数のR2同士は同一であっても異なっていてもよい。
A1が表す、一般式(II)、(III)、(IV)又は(V)で表される基は、以下のとおりである。
(式中、R3は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。pは0~4の整数である。)
R3が表す炭素数1~5の脂肪族炭化水素基、ハロゲン原子としては、R1の場合と同様に説明される。
pは0~4の整数であり、入手容易性の観点から、0~2の整数であってもよく、0又は1であってもよく、0であってもよく。pが2以上の整数である場合、複数のR3同士は同一であっても異なっていてもよい。
(式中、R4及びR5は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A2は炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、単結合、又は下記一般式(III-1)で表される基である。q及びrは各々独立に0~4の整数である。)
R4及びR5が表す炭素数1~5の脂肪族炭化水素基、ハロゲン原子としては、R1の場合と同じものが挙げられる。該脂肪族炭化水素基としては、炭素数1~3の脂肪族炭化水素基であってもよく、メチル基、エチル基であってもよく、エチル基であってもよい。
A2が表す炭素数1~5のアルキレン基としては、例えば、メチレン基、1,2-ジメチレン基、1,3-トリメチレン基、1,4-テトラメチレン基、1,5-ペンタメチレン基等が挙げられる。該アルキレン基としては、高周波特性(低誘電率、低誘電正接)、導体との接着性、耐熱性、ガラス転移温度、熱膨張係数及び難燃性の観点から、炭素数1~3のアルキレン基であってもよく、メチレン基であってもよい。
A2が表す炭素数2~5のアルキリデン基としては、例えば、エチリデン基、プロピリデン基、イソプロピリデン基、ブチリデン基、イソブチリデン基、ペンチリデン基、イソペンチリデン基等が挙げられる。これらの中でも、高周波特性(低誘電率、低誘電正接)、導体との接着性、耐熱性、ガラス転移温度、熱膨張係数及び難燃性の観点から、イソプロピリデン基であってもよい。
A2としては、上記選択肢の中でも、炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基であってもよい。
q及びrは各々独立に0~4の整数であり、入手容易性の観点から、いずれも、0~2の整数であってもよく、0又は2であってもよい。q又はrが2以上の整数である場合、複数のR4同士又はR5同士は、それぞれ同一であっても異なっていてもよい。
なお、A2が表す一般式(III-1)で表される基は以下のとおりである。
(式中、R6及びR7は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A3は炭素数1~5のアルキレン基、イソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。s及びtは各々独立に0~4の整数である。)
R6及びR7が表す炭素数1~5の脂肪族炭化水素基、ハロゲン原子としては、R4及びR5の場合と同様に説明される。
A3が表す炭素数1~5のアルキレン基としては、A2が表す炭素数1~5のアルキレン基と同じものが挙げられる。
A3としては、上記選択肢の中でも、炭素数2~5のアルキリデン基を選択してもよい。
s及びtは0~4の整数であり、入手容易性の観点から、いずれも、0~2の整数であってもよく、0又は1であってもよく、0であってもよい。s又はtが2以上の整数である場合、複数のR6同士又はR7同士は、それぞれ同一であっても異なっていてもよい。
(式中、R8及びR9は各々独立に、水素原子又は炭素数1~5の脂肪族炭化水素基である。uは1~8の整数である。)
R8及びR9が表す炭素数1~5の脂肪族炭化水素基、ハロゲン原子としては、R1の場合と同様に説明される。
uは1~8の整数であり、1~3の整数であってもよく、1であってもよい。
(式中、A1、R1、R2、x及びyは前記定義のとおりである。mは1以上の整数である。)
mは、1~300の整数であってもよく、10~300の整数であってもよく、30~200の整数であってもよく、50~150の整数であってもよい。
なお、本明細書において、数平均分子量は、ゲルパーミエーションクロマトグラフィー(GPC)により、標準ポリスチレンを用いた検量線から換算した値であり、より詳細には実施例に記載の数平均分子量の測定方法により求めた値である。
ポリフェニレンエーテル誘導体(A)は、例えば、以下の製造方法によって得ることができる。
まず、下記一般式(VIII)で表されるアミノフェノール化合物[以下、アミノフェノール化合物(VIII)と称する]と、例えば、数平均分子量15000~25000のポリフェニレンエーテルを有機溶媒中で、公知の再分配反応をさせることにより、ポリフェニレンエーテルの低分子量化を伴いながら、1分子中に第一級アミノ基を有するポリフェニレンエーテル化合物(A'')(以下、単に、ポリフェニレンエーテル化合物(A'')ともいう)を製造し、次いで、前記ポリフェニレンエーテル化合物(A'')と一般式(IX)で表されるビスマレイミド化合物[以下、ビスマレイミド化合物(IX)と称する。]をマイケル付加反応させることによって、ポリフェニレンエーテル誘導体(A)を製造することができる。
アミノフェノール化合物(VIII)の配合量としては、特に制限されるものではないが、例えば、アミノフェノール化合物(VIII)と反応させる前記ポリフェニレンエーテルの数平均分子量が15000~25000であれば、該ポリフェニレンエーテル100質量部に対して0.5~6質量部の範囲で使用することにより、数平均分子量が5000~12000であるポリフェニレンエーテル誘導体(A)が得られる。
作業性及びゲル化抑制の観点から、及び所望の数平均分子量の(A)成分を得るためのポリフェニレンエーテル化合物(A’’)の分子量を制御できる等の観点から、例えば、反応温度70~110℃、反応時間1~8時間で反応を行ってもよい。
これらの中でも、ビス(4-マレイミドフェニル)メタン、3,3’-ジメチル-5,5’-ジエチル-4,4’-ジフェニルメタンビスマレイミド、2,2-ビス(4-(4-マレイミドフェノキシ)フェニル)プロパンを選択してもよい。
前記式(A'-1)を含むポリフェニレンエーテル誘導体が得られ、安価であるという観点から、ビス(4-マレイミドフェニル)メタンを用いてもよい。
前記式(A'-2)を含むポリフェニレンエーテル誘導体が得られ、誘電特性に優れ、低吸水性であるという観点から、3,3’-ジメチル-5,5’-ジエチル-4,4’-ジフェニルメタンビスマレイミドを用いてもよい。
前記式(A'-3)を含むポリフェニレンエーテル誘導体が得られ、導体との高接着性及び機械特性(伸びや破断強度等)に優れるという観点から、2,2-ビス(4-(4-マレイミドフェノキシ)フェニル)プロパンを用いてもよい。
なお、ポリフェニレンエーテル誘導体(A)を製造後は、反応器から取り出す際の作業性や、ポリフェニレンエーテル誘導体(A)に種々の熱硬化性樹脂等を加えて、本発明の熱硬化性樹脂組成物とする際の使用状況(例えば、プリプレグの製造に適した溶液粘度や溶液濃度)に合わせて、適宜、溶液中の有機溶媒の一部又は全部を除去して濃縮してもよく、有機溶媒を追加して希釈してもよい。追加する際の有機溶媒は特に制限はなく、上述した1種類以上の有機溶媒が適用できる。
まず、ポリフェニレンエーテル化合物(A’’)は、GPC測定から数平均分子量15000~25000のポリフェニレンエーテルよりも分子量が低下し、かつ原材料のアミノフェノール化合物(VIII)のピークが消失していること、またIR測定から3300~3500cm-1の第一級アミノ基の出現により所望のポリフェニレンエーテル化合物(A’’)が製造されていることを確認できる。次いでポリフェニレンエーテル誘導体(A)は、再沈殿により精製後、IR測定から3300~3500cm-1の第一級アミノ基のピークの消失と、1700~1730cm-1のマレイミドのカルボニル基のピークの出現を確認することにより、所望のポリフェニレンエーテル誘導体(A)が製造されていることを確認できる。
本発明の熱硬化性樹脂組成物に含まれる(B)成分は、エポキシ樹脂、シアネート樹脂及びマレイミド化合物から選択される少なくとも1種類の熱硬化性樹脂である。なお、該マレイミド化合物は、前記ポリフェニレンエーテル誘導体(A)を包含しない。
エポキシ樹脂は、主骨格の違いによっても種々のエポキシ樹脂に分類分けされ、上記それぞれのタイプのエポキシ樹脂において、更に、ビスフェノールA型エポキシ樹脂、ビスフェノールF型エポキシ樹脂、ビスフェノールS型エポキシ樹脂等のビスフェノール型エポキシ樹脂;脂環式エポキシ樹脂;脂肪族鎖状エポキシ樹脂;フェノールノボラック型エポキシ樹脂、クレゾールノボラック型エポキシ樹脂、ビスフェノールAノボラック型エポキシ樹脂、ビスフェノールFノボラック型エポキシ樹脂等のノボラック型エポキシ樹脂;フェノールアラルキル型エポキシ樹脂;スチルベン型エポキシ樹脂;ジシクロペンタジエン型エポキシ樹脂;ナフトールノボラック型エポキシ樹脂やナフトールアラルキル型エポキシ樹脂等のナフタレン骨格含有型エポキシ樹脂;ビフェニル型エポキシ樹脂;ビフェニルアラルキル型エポキシ樹脂;キシリレン型エポキシ樹脂;ジヒドロアントラセン型エポキシ樹脂;ジシクロペンタジエン型エポキシ樹脂などに分類される。
エポキシ樹脂は、1種類を単独で用いてもよく、2種類以上を併用してもよい。これらの中でも、高周波特性、耐熱性、ガラス移転温度、熱膨張係数及び難燃性等の観点から、ナフタレン骨格含有型エポキシ樹脂、ビフェニルアラルキル型エポキシ樹脂を用いてもよい。
シアネート樹脂にモノフェノール化合物を併用する場合、有機溶媒への溶解性の観点から、予備反応させてフェノール変性シアネートプレポリマーとして用いる方法を採用できる。併用するモノフェノール化合物はプレポリマー化時に規定量全てを配合してもよく、又はプレポリマー化前後で規定量を分けて配合してもよいが、保存安定性の観点から、分けて配合する方法を採用できる。
ポリアミノビスマレイミド化合物(c)は、例えば、(a)成分と1分子中に2個の第一級アミノ基を有する芳香族ジアミン化合物(b)[以下、(b)成分と称することがある。]とを有機溶媒中でマイケル付加反応させることにより得られる。
(式中、A4は前記一般式(Z)中のA1の定義と同じであり、A5は下記一般式(VII)で表される基である。)
(式中、R17及びR18は各々独立に、炭素数1~5の脂肪族炭化水素基、炭素数1~5のアルコキシ基、水酸基又はハロゲン原子である。炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、フルオレニレン基、単結合、又は下記一般式(VII-1)もしくは(VII-2)で表される基である。q’及びr’は各々独立に0~4の整数である。)
(式中、R19及びR20は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A9は炭素数1~5のアルキレン基、イソプロピリデン基、m-フェニレンジイソプロピリデン基、p-フェニレンジイソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。s’及びt’は各々独立に0~4の整数である。)
(式中、R21は炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A10及びA11は各々独立に、炭素数1~5のアルキレン基、イソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。wは0~4の整数である。)
上記一般式(VI)中のA4は前記一般式(Z)中のA1の定義と同じである。
上記一般式(VII)、(VII-1)又は(VII-2)中のA8、A9及びA10が表す炭素数1~5のアルキレン基及び炭素数2~5のアルキリデン基としては、前記一般式(III)中のA2の場合と同様に説明される。また、上記一般式(VII-2)中のA10及びA11が表す炭素数1~5のアルキレン基は、前記一般式(III)中のA2の場合と同様に説明される。
q’及びr’は0~4の整数であり、入手容易性の観点から、いずれも0~2の整数であってもよく、0又は2であってもよい。s’及びt’は0~4の整数であり、入手容易性の観点から、いずれも0~2の整数であってもよく、0又は1であってもよく、0であってもよい。wは0~4の整数であり、入手容易性の観点から、0~2の整数であってもよく、0であってもよい。
また、これらの中でも、有機溶媒への溶解性が高く、合成時の反応率が高く、かつ耐熱性を高くできる観点から、4,4’-ジアミノジフェニルメタン、4,4’-ジアミノ-3,3’-ジメチル-ジフェニルメタン、4,4’-ジアミノ-3,3’-ジエチル-ジフェニルメタン、2,2-ビス(4-(4-アミノフェノキシ)フェニル)プロパン、4,4’-[1,3-フェニレンビス(1-メチルエチリデン)]ビスアニリン、4,4’-[1,4-フェニレンビス(1-メチルエチリデン)]ビスアニリンから選択してもよい。溶解性、反応率及び耐熱性が優れることに加えて、安価であるという観点から、4,4’-ジアミノジフェニルメタン、4,4’-ジアミノ-3,3’-ジメチル-ジフェニルメタン、4,4’-ジアミノ-3,3’-ジエチル-ジフェニルメタンであってもよい。また、溶解性、反応率及び耐熱性に優れることに加えて、導体との高接着性を発現できるという観点から、2,2-ビス(4-(4-アミノフェノキシ)フェニル)プロパン、4,4’-[1,3-フェニレンビス(1-メチルエチリデン)]ビスアニリン、4,4’-[1,4-フェニレンビス(1-メチルエチリデン)]ビスアニリンであってもよい。更に、溶解性、反応率、耐熱性、及び導体との接着性に優れることに加えて、高周波特性及び耐吸湿性の観点からは、4,4’-[1,3-フェニレンビス(1-メチルエチリデン)]ビスアニリン、4,4’-[1,4-フェニレンビス(1-メチルエチリデン)]ビスアニリンを選択してもよい。これらは目的や用途等に合せて、1種類を単独で用いてもよく、2種類以上を併用してもよい。
反応濃度(固形分濃度)は特に制限はないが、10~90質量%であってもよく、20~80質量%であってもよい。反応濃度が10質量以上の場合、反応速度が遅くなりすぎず、製造コストの面でより有利となる傾向にある。90質量%以下の場合、より良好な溶解性が得られる傾向にある。また、溶液粘度が低いので攪拌効率がよく、ゲル化することも少ない。なお、ポリアミノビスマレイミド化合物(c)の製造後は、ポリフェニレンエーテル誘導体(A)の製造時と同様に、目的に合わせて有機溶媒の一部又は全部を除去(濃縮)したり、有機溶媒を追加して希釈したりすることができる。
前記(A)成分の含有量は特に制限はないが、高周波特性の観点から、(A)~(C)成分の総和100質量部に対して3質量%以上であることが好ましく、5質量%以上であることがより好ましい。
前記(B)成分の含有量は特に制限はないが、高周波特性及び成形性の観点から、(A)~(C)成分の総和100質量部に対して、10~90質量%であることが好ましく、20~80質量%以上であることがより好ましい。
前記(A)成分と前記(B)成分の含有割合[(A):(B)]は特に制限はないが、質量比で、5:95~80:20であってもよく、5:95~75:25であってもよく、5:95~70:30であってもよく、10:90~70:30であってもよい。(A)成分と(B)成分合計量に対する(A)成分の含有割合が5質量%以上であれば、より優れた高周波特性及び低吸湿性が得られる傾向にある。また、80質量%以下であれば、より優れた耐熱性、より優れた成形性及びより優れた加工性が得られる傾向にある。
本発明の熱硬化性樹脂組成物は、更にリン系難燃剤(C)を含む。リン系難燃剤(C)を用いることにより、ハロゲンフリーを実現しながら難燃性を付与することが可能となり、それのみならず、リン系難燃剤(C)によって優れた高周波特性(低誘電率、低誘電正接)、導体との高接着性、優れた耐熱性、低熱膨張係数及び高ガラス転移温度を達成することもできる。
リン系難燃剤(C)は、1種類を単独で用いてもよく、2種類以上を併用してもよい。
無機系のリン系難燃剤としては、例えば、赤リン;リン酸一アンモニウム、リン酸二アンモニウム、リン酸三アンモニウム、ポリリン酸アンモニウム等のリン酸アンモニウム;リン酸アミド等の無機系含窒素リン化合物;リン酸;ホスフィンオキシドなどが挙げられる。
有機系のリン系難燃剤としては、例えば、芳香族リン酸エステル、1置換ホスホン酸ジエステル、2置換ホスフィン酸エステル、2置換ホスフィン酸の金属塩、有機系含窒素リン化合物、環状有機リン化合物等が挙げられる。これらの中でも芳香族リン酸エステル化合物、2置換ホスフィン酸の金属塩を選択してもよい。ここで、「金属塩」としては、リチウム塩、ナトリウム塩、カリウム塩、カルシウム塩、マグネシウム塩、アルミニウム塩、チタン塩、亜鉛塩のいずれかであってもよく、アルミニウム塩であってもよい。また、有機系のリン系難燃剤の中でも、芳香族リン酸エステルであってもよい。
1置換ホスホン酸ジエステルとしては、例えば、フェニルホスホン酸ジビニル、フェニルホスホン酸ジアリル、フェニルホスホン酸ビス(1-ブテニル)等が挙げられる。
2置換ホスフィン酸エステルとしては、例えば、ジフェニルホスフィン酸フェニル、ジフェニルホスフィン酸メチル等が挙げられる。
2置換ホスフィン酸の金属塩としては、ジアルキルホスフィン酸の金属塩、ジアリルホスフィン酸の金属塩、ジビニルホスフィン酸の金属塩、ジアリールホスフィン酸の金属塩等が挙げられる。これら金属塩は、前述の通り、リチウム塩、ナトリウム塩、カリウム塩、カルシウム塩、マグネシウム塩、アルミニウム塩、チタン塩、亜鉛塩のいずれかであってもよく、アルミニウム塩を選択してもよい。
有機系含窒素リン化合物としては、例えば、ビス(2-アリルフェノキシ)ホスファゼン、ジクレジルホスファゼン等のホスファゼン化合物;リン酸メラミン、ピロリン酸メラミン、ポリリン酸メラミン、ポリリン酸メラム等のメラミン系化合物などが挙げられる。
環状有機リン化合物としては、9,10-ジヒドロ-9-オキサ-10-ホスファフェナントレン-10-オキシド、10-(2,5-ジヒドロキシフェニル)-9,10-ジヒドロ-9-オキサ-10-ホスファフェナントレン-10-オキシド等が挙げられる。
RC6及びRC7は各々独立に、炭素数1~5の脂肪族炭化水素基又は炭素数6~14の芳香族炭化水素基である。Mは、リチウム原子、ナトリウム原子、カリウム原子、カルシウム原子、マグネシウム原子、アルミニウム原子、チタン原子、亜鉛原子である。m1は、1~4の整数である。)
Acが表す炭素数1~5のアルキレン基及び炭素数2~5のアルキリデン基は、前記A2の場合と同様に説明される。Acとしては、前記選択肢の中でも、炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、単結合であってもよく、イソプロピリデン基、単結合であってもよい。
e及びfは、0~2の整数であってもよく、2であってもよい。g、h及びiは、0~2の整数であってもよく、0又は1であってもよく、0であってもよい。
RC6及びRC7が表す炭素数1~5の脂肪族炭化水素基としては、前記一般式(I)中のR1の場合と同じものが挙げられる。該脂肪族炭化水素基としては、炭素数1~3の脂肪族炭化水素基であってもよく、エチル基であってもよい。
RC6及びRC7が表す炭素数6~14の芳香族炭化水素基としては、例えば、フェニル基、ナフチル基、ビフェニリル基、アントリル基等が挙げられる。該芳香族炭化水素基としては、炭素数6~10の芳香族炭化水素基であってもよい。
m1は金属イオンの価数を表しており、つまり、Mの種類に対応して1~4の範囲内で変化する。
Mとしては、アルミニウム原子であってもよい。なお、Mがアルミニウム原子である場合、m1は3である。
本発明の熱硬化樹脂組成物におけるリン系難燃剤(C)の含有割合は特に制限されるものではないが、例えば、固形分換算の熱硬化性樹脂組成物(後述する(D)成分を除くその他成分の総和)中のリン原子の含有量が0.2~5質量%であってもよく、0.3~3質量%であってもよい。リン原子の含有量が0.2質量%以上の場合、より良好な難燃性が得られる傾向にある。また、リン原子の含有量が5質量%以下である場合、より良好な成形性、より導体との高接着性、より優れた耐熱性及び高ガラス転移温度が得られる傾向にある。
本発明の熱硬化性樹脂組成物は、任意に必要に応じて、無機充填剤(D)[以下、(D)成分と称することがある。]及び硬化促進剤(E)[以下、(E)成分と称することがある。]から選択される少なくとも1種類を含有していてもよい。これらを含有させることにより、積層板とした際の諸特性を更に向上させることができる。
例えば、本発明の熱硬化性樹脂組成物に任意に適切な無機充填剤(D)を含有させることで、低熱膨張係数、高弾性率性、耐熱性及び難燃性を向上させることができる。また、適切な硬化促進剤(E)を含有させることで、熱硬化性樹脂組成物の硬化性を向上させ、高周波特性、耐熱性、導体との接着性、弾性率及びガラス転移温度を向上させることができる。
更に、前記リン系難燃剤(C)以外の難燃剤及び難燃助剤を、(C)成分と併用してもよい。
(D)成分としては、特に制限されるものではないが、例えば、シリカ、アルミナ、酸化チタン、マイカ、ベリリア、チタン酸バリウム、チタン酸カリウム、チタン酸ストロンチウム、チタン酸カルシウム、炭酸アルミニウム、水酸化マグネシウム、水酸化アルミニウム、ケイ酸アルミニウム、炭酸カルシウム、ケイ酸カルシウム、ケイ酸マグネシウム、窒化ケイ素、窒化ホウ素、クレー(焼成クレー等)、タルク、ホウ酸アルミニウム、ホウ酸アルミニウム、炭化ケイ素等が挙げられる。これらは1種類を単独で用いてもよく、2種類以上を併用してもよい。これらの中でも、熱膨張係数、弾性率、耐熱性及び難燃性の観点から、シリカ、アルミナ、マイカ、タルクであってもよく、シリカ、アルミナであってもよく、シリカであってもよい。シリカとしては、例えば、湿式法で製造され含水率の高い沈降シリカと、乾式法で製造され結合水等をほとんど含まない乾式法シリカ等が挙げられ、乾式法シリカとしては更に、製造法の違いにより破砕シリカ、フュームドシリカ、溶融シリカ(溶融球状シリカ)等が挙げられる。
また、無機充填剤(D)の形状及び粒径についても特に制限はないが、例えば、粒径は0.01~20μmであってもよく、0.1~10μmであってもよい。ここで、粒径とは、平均粒子径を指し、粒子の全体積を100%として粒子径による累積度数分布曲線を求めた時、体積50%に相当する点の粒子径のことである。レーザ回折散乱法を用いた粒度分布測定装置等で測定することができる。
また、(D)成分を用いる場合、(D)成分の分散性や(D)成分と樹脂組成物中の有機成分との密着性を向上させる目的で、必要に応じ、カップリング剤を併用してもよい。該カップリング剤としては特に限定されるものではなく、例えば、シランカップリング剤又はチタネートカップリング剤を適宜選択して用いることができる。カップリング剤は1種類を単独で用いてもよく、2種類以上を併用してもよい。また、カップリング剤の使用量も特に限定されるものではなく、例えば、(D)成分100質量部に対して0.1~5質量部であってもよく、0.5~3質量部であってもよい。この範囲であれば、諸特性の低下が少なく、上記の(D)成分の使用による特長を効果的に発揮できる傾向にある。
なお、カップリング剤を用いる場合、樹脂組成物中に(D)成分を配合した後、カップリング剤を添加する、いわゆるインテグラルブレンド処理方式ではなく、予め無機充填剤にカップリング剤を乾式又は湿式で表面処理した無機充填剤を使用する方式を採用できる。この方法を採用することで、より効果的に上記(D)成分の特長を発現できる。
本発明の熱硬化性樹脂組成物に(E)成分を含有させる場合、使用する(B)成分の種類に合わせて好適な(E)成分を用いることができる。
(B)成分としてエポキシ樹脂を用いる場合の(E)成分としては、例えば、イミダゾール化合物及びその誘導体;第3級アミン化合物;第4級アンモニウム化合物;トリフェニルホスフィン等のリン系化合物などが挙げられる。これらは1種類を単独で用いてもよく、2種類以上を併用してもよい。これらの中でも、耐熱性、ガラス転移温度及び保存安定性の観点から、イミダゾール化合物及びその誘導体又はリン系化合物を用いてもよい。該イミダゾール化合物としては、例えば、メチルイミダゾール、フェニルイミダゾール、イソシアネートマスクイミダゾール(例えば、ヘキサメチレンジイソシアネート樹脂と2-エチル-4-メチルイミダゾールの付加反応物等)等が挙げられ、イソシアネートマスクイミダゾールを選択してもよい。
本発明の熱硬化性樹脂組成物には、本発明の効果を損なわない範囲で、リン系難燃剤(C)以外の難燃剤及び難燃助剤を含有させてもよい。
リン系難燃剤(C)以外の難燃剤としては、環境問題の観点からは、例えば、水酸化アルミニウムの水和物、水酸化マグネシウムの水和物等の金属水和物であってもよく、これらは1種類を単独で用いてもよく、2種類以上を併用してもよい。該金属水酸化物は無機充填剤にも該当し得るが、難燃性を付与し得る材料の場合には難燃剤に分類する。
本発明の熱硬化性樹脂組成物にリン系難燃剤(C)以外の難燃剤を含有させる場合、該難燃剤の含有割合は、リン系難燃剤(C)100質量部に対して、50質量部以下であってもよく、30質量部以下であってもよく、15質量部以下であってもよい。
本発明の熱硬化性樹脂組成物には、難燃助剤、例えば、三酸化アンチモン、モリブデン酸亜鉛等の無機系難燃助剤を含有させることができる。
本発明の熱硬化性樹脂組成物に難燃助剤を含有させる場合、その含有割合は特に制限されるものではないが、例えば、(A)成分と(B)成分の総和100質量部に対して、0.1~20質量部であってもよく、0.1~10質量部であってもよい。このような範囲で難燃助剤を用いると、より良好な耐薬品性が得られる傾向にある。
本発明の樹脂組成物は、希釈することによって取り扱いを容易にするという観点及び後述するプリプレグを製造し易くする観点から、有機溶剤を含有させてもよい。
該有機溶剤としては、特に制限されないが、例えば、エタノール、プロパノール、ブタノール、メチルセロソルブ、ブチルセロソルブ、プロピレングリコールモノメチルエーテル等のアルコール系溶剤;アセトン、メチルエチルケトン、メチルイソブチルケトン、シクロヘキサノン等のケトン系溶剤;テトラヒドロフラン等のエーテル系溶剤;トルエン、キシレン、メシチレン等の芳香族系溶剤;ジメチルホルムアミド、ジメチルアセトアミド、N-メチルピロリドン等の窒素原子含有溶剤;ジメチルスルホキシド等の硫黄原子含有溶剤;γ-ブチロラクトン等のエステル系溶剤などが挙げられる。
これらの中でも、溶解性の観点から、アルコール系溶剤、ケトン系溶剤、窒素原子含有溶剤であってもよく、ケトン系溶剤であってもよく、アセトン、メチルエチルケトン、メチルイソブチルケトン、シクロヘキサノンであってもよく、メチルエチルケトンであってもよく。
有機溶剤は、1種類を単独で用いてもよく、2種類以上を併用してもよい。
本発明の熱硬化性樹脂組成物における有機溶剤の含有量は、特に制限はないが、固形分濃度が30~90質量%であってもよく、40~80質量%であってもよく、40~70質量%であってもよく、40~60質量%であってもよい。固形分濃度が上記の範囲内である熱硬化性樹脂組成物を用いることで、取り扱い性が容易となり、さらに基材への含浸性や製造されるプリプレグの外観が良好で、後述するプリプレグ中の樹脂の固形分濃度の調整が容易となり、所望の厚みとなるようなプリプレグの製造が容易となる傾向にある。
また、本発明の熱硬化性樹脂組成物から積層板を作成したときの熱膨張係数(Z方向、Tg以下)は特に限定されないが、積層板のそりを抑制する観点から、45ppm/℃以下であることが好ましく、43ppm/℃以下であることがより好ましい。熱膨張係数の下限値には制限はないが、通常、30ppm/℃以上、さらには35ppm/℃以上である。
なお、ガラス転移温度及び熱膨張係数は、実施例に記載のとおり、IPC規格に準じて測定した値である。
また、誘電正接は小さいことが好ましく、0.007以下であってもよく、0.006以下であってもよい。誘電率の下限については特に限定はなく、小さいほどよいが、例えば0.0001以上であってもよく、0.002以上であってもよく、0.004以上であってもよく、0.005以上であってもよい。
なお、誘電率及び誘電正接は実施例のとおり、JPCA-TM001(トリプレート共振器法)に準拠して測定した値である。
本発明は、本発明の樹脂組成物とシート状繊維補強基材とを含有してなるプリプレグをも提供する。該プリプレグは、本発明の樹脂組成物とシート状繊維補強基材とを用いて形成され、より具体的には、本発明の熱硬化性樹脂組成物を、シート状繊維補強基材に含浸又は塗工し、乾燥させることによって得ることができる。より具体的には、例えば、乾燥炉中で通常、80~200℃の温度で、1~30分間加熱乾燥し、半硬化(Bステージ化)させることにより本発明のプリプレグを製造することができる。熱硬化性樹脂組成物の使用量は、乾燥後のプリプレグ中の熱硬化性樹脂組成物由来の固形分濃度が30~90質量%となるように決定することができる。固形分濃度を上記の範囲とすることで積層板とした際、より良好な成形性が得られる傾向にある。
ホットメルト法は、熱硬化性樹脂組成物に有機溶剤を含有させず、(1)該組成物との剥離性の良い塗工紙に一旦コーティングし、それをシート状補強基材にラミネートする方法、又は(2)ダイコーターによりシート状補強基材に直接塗工する方法である。
一方、ソルベント法は、熱硬化性樹脂組成物に有機溶剤を含有させ、得られた熱硬化性樹脂組成物にシート状補強基材を浸漬して、熱硬化性樹脂組成物をシート状補強基材に含浸させ、その後、乾燥させる方法である。
本発明の積層板は、本発明のプリプレグと金属箔とを含有してなるものである。本発明の積層板は、本発明のプリプレグと金属箔とを用いて形成され、より具体的には、本発明のプリプレグ1枚の片面もしくは両面に金属板を配置するか、又は本発明のプリプレグ2枚以上を重ねて得られるプリプレグの片面もしくは両面に金属箔を配置し、次いで加熱加圧成形することによって得ることができる。
金属箔の金属としては、電気絶縁材料用途で用いられるものであれば特に制限されないが、導電性の観点から、銅、金、銀、ニッケル、白金、モリブデン、ルテニウム、アルミニウム、タングステン、鉄、チタン、クロム、又はこれらの金属元素のうちの少なくとも1種類を含む合金であってもよく、銅、アルミニウムであってもよく、銅であってもよい。
加熱加圧成形の条件は特に制限されるものではなくが、例えば、温度が100℃~300℃、圧力が0.2~10.0MPa、時間が0.1~5時間の範囲で実施することができる。また加熱加圧成形は真空プレス等を用いて真空状態を0.5~5時間保持する方法を採用できる。
また、本発明の多層プリント配線板は、本発明のプリプレグ又は積層板を含有してなるものである。本発明の多層プリント配線板は、本発明のプリプレグ又は積層板を用いて形成されるものであり、本発明のプリプレグ又は積層板を用い、公知の方法によって、穴明け加工、金属めっき加工、金属箔のエッチング等による回路形成加工及び多層化接着加工を行うことによって製造することができる。
温度計、還流冷却管、撹拌装置を備えた加熱及び冷却可能な容積2Lのガラス製フラスコ容器に、トルエン(190質量部)、PPO640 [ポリフェニレンエーテル、数平均分子量:約16000、SABICイノベーティブプラスチックス社製、商品名] (100質量部)、p-アミノフェノール[イハラケミカル工業株式会社製] (1.35質量部)を投入し、フラスコ内の温度を90℃に設定、保温して撹拌しながら溶解した。溶解を目視で確認後、パーブチル(登録商標)I[t-ブチルペルオキシイソプロピルモノカーボネート、日油株式会社製、商品名](2質量部)とナフテン酸マンガン[和光純薬工業株式会社製](0.15質量部)とを添加し、溶液温度90℃で4時間反応させた後、70℃に冷却して分子末端に第一級アミノ基を有するポリフェニレンエーテル化合物(A’)を得た。
この反応溶液を少量取り出し、ゲルパーミエーションクロマトグラフィー(GPC)により、測定を行ったところ、p-アミノフェノールに由来するピークが消失し、かつポリフェニレンエーテル化合物(A’)の数平均分子量は約9200であった。また少量取り出した反応溶液をメタノール/ベンゼン混合溶媒(混合質量比:1:1)に滴下、再沈殿させて精製した固形分(反応生成物)のFT-IR測定を行ったところ、3400cm-1付近の第一級アミノ基由来のピークの出現が確認された。
装置:
ポンプ:L-6200型[株式会社日立ハイテクノロジーズ製]
検出器:L-3300型RI[株式会社日立ハイテクノロジーズ製]
カラムオーブン:L-655A-52[株式会社日立ハイテクノロジーズ製]
カラム:ガードカラム;TSK Guardcolumn HHR-L+カラム;TSKgel G4000HHR+TSKgel G2000HHR[すべて東ソー株式会社製、商品名]
カラムサイズ:6.0×40mm(ガードカラム)、7.8×300mm(カラム)
溶離液:テトラヒドロフラン
試料濃度:30mg/5mL
注入量:20μL
流量:1.00mL/分
測定温度:40℃
この反応溶液を少量取り出し、上記同様に再沈殿、精製した固形物のFT-IR測定を行い、3400cm-1付近の第一級アミノ基由来ピークの消失と、1700~1730cm-1のマレイミドのカルボニル基のピークの出現が確認された。またこの固形物のGPC(上記と同条件)を測定したところ、数平均分子量は約9400であった。
温度計、還流冷却管、撹拌装置を備えた加熱及び冷却可能な容積1Lのガラス製フラスコ容器に、BMI-4000[2,2-ビス(4-(4-マレイミドフェノキシ)フェニル)プロパン、大和化成工業株式会社製、商品名] (100質量部)、ビスアニリン-M[4,4’-〔1,3-フェニレンビス(1-メチルエチリデン)〕ビスアニリン、三井化学株式会社製、商品名](14質量部)及びプロピレングリコールモノメチルエーテル(50質量部)を投入し、液温を120℃に保ったまま、撹拌しながら3時間反応させた後、冷却及び200メッシュフィルターを通して濾過し、熱硬化性樹脂(B)としてのポリアミノビスマレイミド化合物(B-1)を製造した。
製造例Aで得たポリフェニレンエーテル誘導体(A-1)、製造例Bで得たポリアミノビスマレイミド化合物(B-1)、難燃剤、無機充填剤、硬化促進剤及び有機溶媒を表1に記載の配合量(単位:質量部)に従って室温で加熱しながら攪拌、混合して、固形分(不揮発分)濃度40~60質量%の熱硬化性樹脂組成物を調製した。
ここで、無機充填剤の配合量としては、通常、樹脂組成物(無機充填剤を除く)の密度が1.20~1.25g/cm3であり、用いた無機充填剤の密度が2.2~3.01g/cm3であることから、無機充填剤を樹脂組成物(無機充填剤を除く)100質量部に対して80質量部配合した場合、30~34体積%程度となる。
上記実施例及び比較例で得られた熱硬化性樹脂組成物を用いて、下記方法に従って各評価及び測定を行った。結果を表1に示す。
各例で得た熱硬化性樹脂組成物及びこれを160℃で10分間乾燥して有機溶媒を揮発させた後の外観を目視で観察して、それぞれの相容性(巨視的(マクロ)な相分離及びムラの有無)を以下の基準に従い評価した。
A:巨視的(マクロ)な相分離及びムラがない。
C:巨視的(マクロ)な相分離又はムラがある。
各例で得た熱硬化性樹脂組成物を、厚さ0.1mmのガラス布[Eガラス、日東紡績株式会社製]に塗工した後、160℃で7分間加熱乾燥して、樹脂含有量(樹脂分)約54質量%のプリプレグを作製した。これらのプリプレグ6枚を重ね、その上下に、厚さ18μmのロープロファイル銅箔[商品名「FV-WS」、M面Rz:1.5μm、古河電気工業株式会社製]をM面が接するように配置し、温度230℃、圧力3.9MPa、時間180分間の条件で加熱加圧成形して、両面銅張積層板(厚さ:0.8mm)を作製した。
(2-1.プリプレグの外観評価)
上記で得られたプリプレグの外観を観察した。外観は目視により評価し、以下の基準に従い評価した。
A:外観上の異常がない。
C:プリプレグ表面に多少なりともムラ、スジ、発泡、相分離等があり、表面平滑性に欠ける。
(2-2.銅張積層板の特性評価)
上記で得られた銅張積層板について、成形性、誘電特性、銅箔引きはがし強さ、ガラス転移温度、熱膨張係数、はんだ耐熱性及び難燃性を評価した。銅張積層板の特性評価方法は、以下の通りである。
両面の銅箔をエッチングした積層板の外観を観察して成形性を評価した。成形性は目視により評価し、以下の基準に従い評価した。
A:外観上の異常がない。
C:多少なりともムラ、スジ、カスレ、ボイド等があり、表面平滑性に欠ける。
(2-2-2)誘電特性
誘電特性(比誘電率、誘電正接)はJPCA-TM001(トリプレート共振器法)に準拠して、1GHz帯及び10GHz帯で誘電率及び誘電正接を測定した。
銅箔引きはがし強さは、JIS C6481(1996年)に準拠して測定し、導体との接着性を測定した。
(2-2-4)はんだ耐熱性
はんだ耐熱性は、両面の銅箔をエッチングした50mm角の試験片を用いて、常態及びプレッシャークッカーテスト(PCT)用装置(条件:121℃、2.2気圧)中に所定時間(1時間、3時間及び5時間)処理した後のものを、288℃の溶融はんだ中に20秒間浸漬した後の試験片の外観を目視観察した。なお、表中の数字は、はんだ浸漬後の試験片3枚のうち、積層板内に膨れやミーズリングの発生等の異常が認められなかったものの枚数を意味する。
(2-2-5)ガラス転移温度(Tg)及び熱膨張係数
ガラス転移温度(Tg)と熱膨張係数(板厚方向、温度範囲:30~150℃)は、両面の銅箔をエッチングした5mm角の試験片を用いて、熱機械測定装置(TMA)[ティー・エイ・インスツルメント・ジャパン株式会社製、Q400(型番)]により、IPC(The Institute for Interconnecting and Packaging Electronic Circuits)規格に準拠して測定した。
(2-2-6)難燃性
銅張積層板を銅エッチング液である過硫酸アンモニウム[三菱ガス化学株式会社製]10質量%溶液に浸漬することにより銅箔を取り除いた評価基板から、長さ127mm及び幅12.7mmの試験片を切り出し、該試験片を用いて、UL94の試験法(V法)に準じて難燃性を試験及び評価した。
つまり、垂直に保持した試験片の下端に20mm炎による10秒間の接炎を2回行なった。評価は、UL94のV法の基準に従って行なった。
(1)難燃剤
・OP-935:ジアルキルホスフィン酸アルミニウム塩[2置換ホスフィン酸の金属塩、リン含有量:23.5質量%、クラリアント社製]
・PX-200:1,3-フェニレンビス(ジ-2,6-キシレニルホスフェート)、[芳香族リン酸エステル、リン含有量:9質量%、大八化学工業株式会社製]
・PX-202:4,4'-ビフェニレン-テトラキス(2,6-ジメチルフェニルホスフェート)[芳香族リン酸エステル、リン含有量:8質量%、大八化学工業株式会社製]
・SPB-100:ポリビスフェノキシホスファゼン[有機系含窒素リン化合物、リン含有量:13質量%、大塚化学株式会社製]
・HCA-HQ:10-(2,5-ジヒドロキシフェニル)-9,10-ジヒドロ-9-オキサ-10-ホスファフェナントレン-10-オキシド[環状有機リン化合物、リン含有量:9.6質量%、三光株式会社製]
・SAYTEX8010:1,2-ビス(2,3,4,5,6-ペンタブロモフェニル)エタン[臭素系難燃剤、臭素含有量:82質量%、アルベマール日本株式会社製]
・AlOOH:ベーマイト型水酸化アルミニウム[金属水和物、密度3.0g/cm3、河合石灰工業株式会社製]
なお、上記のOP-935、PX-200、PX-202、SPB-100、HCA-HQ、SAYTEX8010は商品名である。
・SC-2050 KNK:球状溶融シリカ、平均粒子径:0.5μm、表面処理:ビニルシランカップリング剤(1質量%/固形分)、分散媒:メチルイソブチルケトン、固形分濃度70質量%、密度2.2g/cm3、株式会社アドマテックス製、商品名
(3)硬化促進剤
・パーブチル(登録商標)P:α,α’-ビス(t-ブチルパーオキシ)ジイソプロピルベンゼン、日油株式会社製、商品名
・G-8009L:イソシアネートマスクイミダゾール(ヘキサメチレンジイソシアネート樹脂と2-エチル-4-メチルイミダゾールの付加反応物)、第一工業製薬株式会社製、商品名
一方、表1が示すように、比較例においては、樹脂組成物の相容性やプリプレグの外観が良好ではないものがある(比較例1~3)。また各比較例の銅張積層板は、成形性、誘電特性、導体との接着性、はんだ耐熱性、ガラス転移温度、熱膨張係数及び難燃性のいずれかが実施例よりも良好ではない。
また、前記熱硬化性樹脂組成物は、原材料コストや基板材料の製造コストを低く抑えられ、更に作業環境性にも優れるため、この熱硬化性樹脂組成物を用いて提供されるプリプレグ及び積層板は、多層プリント配線板等の電子部品用途に好適に使用することができる。
Claims (14)
- 前記N-置換マレイミド構造含有基が下記一般式(Z)で表される基である、請求項1に記載の熱硬化性樹脂組成物。
(式中、R2は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。yは0~4の整数である。A1は、下記一般式(II)、(III)、(IV)又は(V)で表される基である。)
(式中、R3は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。pは0~4の整数である。)
(式中、R4及びR5は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A2は炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、単結合、又は下記一般式(III-1)で表される基である。q及びrは各々独立に0~4の整数である。)
(式中、R6及びR7は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A3は炭素数1~5のアルキレン基、イソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。s及びtは各々独立に0~4の整数である。)
(式中、nは0~10の整数である。)
(式中、R8及びR9は各々独立に、水素原子又は炭素数1~5の脂肪族炭化水素基である。uは1~8の整数である。) - 前記リン系難燃剤(C)が、芳香族リン酸エステル、及び2置換ホスフィン酸の金属塩から選択される少なくとも1種類である、請求項1~4のいずれか1項に記載の熱硬化性樹脂組成物。
- 前記芳香族リン酸エステルが下記一般式(C-1)又は(C-2)で表され、前記2置換ホスフィン酸の金属塩が下記一般式(C-3)で表される、請求項5に記載の熱硬化性樹脂組成物。
(式中、RC1~RC5は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。Acは炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、単結合である。e及びfは各々独立に0~5の整数であり、g、h及びiは各々独立に0~4の整数である。
RC6及びRC7は各々独立に、炭素数1~5の脂肪族炭化水素基又は炭素数6~14の芳香族炭化水素基である。Mは、リチウム原子、ナトリウム原子、カリウム原子、カルシウム原子、マグネシウム原子、アルミニウム原子、チタン原子、亜鉛原子である。m1は、1~4の整数である。) - 前記(B)成分中のマレイミド化合物が、1分子中に少なくとも2個のN-置換マレイミド基を有するポリマレイミド化合物(a)、又は下記一般式(VI)で表されるポリアミノビスマレイミド化合物(c)である、請求項1~6のいずれか1項に記載の熱硬化性樹脂組成物。
(式中、A4は前記一般式(Z)中のA1の定義と同じであり、A5は下記一般式(VII)で表される基である。)
(式中、R17及びR18は各々独立に、炭素数1~5の脂肪族炭化水素基、炭素数1~5のアルコキシ基、水酸基又はハロゲン原子である。A8は炭素数1~5のアルキレン基、炭素数2~5のアルキリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基、フルオレニレン基、単結合、又は下記一般式(VII-1)もしくは(VII-2)で表される基である。q’及びr’は各々独立に0~4の整数である。)
(式中、R19及びR20は各々独立に、炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A9は炭素数1~5のアルキレン基、イソプロピリデン基、m-フェニレンジイソプロピリデン基、p-フェニレンジイソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。s’及びt’は各々独立に0~4の整数である。)
(式中、R21は炭素数1~5の脂肪族炭化水素基又はハロゲン原子である。A10及びA11は各々独立に、炭素数1~5のアルキレン基、イソプロピリデン基、エーテル基、スルフィド基、スルホニル基、カルボオキシ基、ケト基又は単結合である。wは0~4の整数である。) - (A)成分と(B)成分の含有割合[(A):(B)]が、質量比で、5:95~80:20である、請求項1~7のいずれか1項に記載の熱硬化性樹脂組成物。
- 更に無機充填剤(D)を含有する、請求項1~8のいずれか1項に記載の熱硬化性樹脂組成物。
- 更に硬化促進剤(E)を含有する、請求項1~9のいずれか1項に記載の熱硬化性樹脂組成物。
- 更に有機溶媒を含有する、請求項1~10のいずれか1項に記載の熱硬化性樹脂組成物。
- 請求項1~11のいずれか1項に記載の熱硬化性樹脂組成物とシート状繊維補強基材とを含有してなるプリプレグ。
- 請求項12に記載のプリプレグと金属箔とを含有してなる積層板。
- 請求項12に記載のプリプレグ又は請求項13に記載の積層板を含有してなる多層プリント配線板。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/570,174 US10519279B2 (en) | 2015-04-30 | 2016-04-28 | Thermosetting resin composition, prepreg, laminate and multilayer printed wiring board |
| JP2017515634A JP6705446B2 (ja) | 2015-04-30 | 2016-04-28 | 熱硬化性樹脂組成物、プリプレグ、積層板及び多層プリント配線板 |
| SG11201708808SA SG11201708808SA (en) | 2015-04-30 | 2016-04-28 | Thermosetting resin composition, prepreg, laminate and multilayer printed wiring board |
| KR1020177030709A KR102466877B1 (ko) | 2015-04-30 | 2016-04-28 | 열경화성 수지 조성물, 프리프레그, 적층판 및 다층 프린트 배선판 |
| EP16786601.1A EP3290479A4 (en) | 2015-04-30 | 2016-04-28 | Thermosetting resin composition, prepreg, laminate and multilayer printed wiring board |
| CN201680024864.4A CN107531992B (zh) | 2015-04-30 | 2016-04-28 | 热固性树脂组合物、预浸料、层叠板和多层印刷线路板 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-093508 | 2015-04-30 | ||
| JP2015093508 | 2015-04-30 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/570,174 A-371-Of-International US10519279B2 (en) | 2015-04-30 | 2016-04-28 | Thermosetting resin composition, prepreg, laminate and multilayer printed wiring board |
| US16/676,501 Division US11359055B2 (en) | 2015-04-30 | 2019-11-07 | Thermosetting resin composition, prepreg, laminate and multilayer printed wiring board |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016175325A1 true WO2016175325A1 (ja) | 2016-11-03 |
Family
ID=57199777
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/063497 Ceased WO2016175325A1 (ja) | 2015-04-30 | 2016-04-28 | 熱硬化性樹脂組成物、プリプレグ、積層板及び多層プリント配線板 |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US10519279B2 (ja) |
| EP (1) | EP3290479A4 (ja) |
| JP (1) | JP6705446B2 (ja) |
| KR (1) | KR102466877B1 (ja) |
| CN (1) | CN107531992B (ja) |
| SG (1) | SG11201708808SA (ja) |
| TW (1) | TWI721978B (ja) |
| WO (1) | WO2016175325A1 (ja) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020031495A1 (ja) | 2018-08-06 | 2020-02-13 | 大八化学工業株式会社 | 芳香族リン酸エステルを含む熱硬化性樹脂用難燃剤、それを含む熱硬化性樹脂組成物、その硬化物およびその用途 |
| KR20200060292A (ko) * | 2018-11-16 | 2020-05-29 | 리쇼 고교 가부시키가이샤 | 수지 조성물, 프리프레그, 및 적층판 |
| WO2020157828A1 (ja) * | 2019-01-29 | 2020-08-06 | 日立化成株式会社 | 樹脂組成物、半導体装置の製造方法及び半導体装置 |
| WO2020204175A1 (ja) * | 2019-04-03 | 2020-10-08 | 日立化成株式会社 | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び半導体パッケージ |
| JP2020169273A (ja) * | 2019-04-03 | 2020-10-15 | 日立化成株式会社 | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び半導体パッケージ |
| JPWO2019172342A1 (ja) * | 2018-03-06 | 2021-03-18 | 昭和電工マテリアルズ株式会社 | プリプレグ、積層板、多層プリント配線板、半導体パッケージ及び樹脂組成物、並びに、プリプレグ、積層板及び多層プリント配線板の製造方法 |
| JP2021075729A (ja) * | 2021-01-26 | 2021-05-20 | 昭和電工マテリアルズ株式会社 | 樹脂組成物、プリプレグ、積層板及び多層プリント配線板 |
| JPWO2022124129A1 (ja) * | 2020-12-09 | 2022-06-16 | ||
| JPWO2022124130A1 (ja) * | 2020-12-09 | 2022-06-16 | ||
| JPWO2022149440A1 (ja) * | 2021-01-06 | 2022-07-14 | ||
| WO2022163505A1 (ja) | 2021-01-27 | 2022-08-04 | デンカ株式会社 | 球状アルミナ粒子及びその製造方法、並びに樹脂組成物 |
| EP3467028B1 (en) * | 2016-05-31 | 2024-04-24 | Mitsubishi Gas Chemical Company, Inc. | Resin composition, laminate, semiconductor wafer with resin composition layer, substrate for mounting semiconductor with resin composition layer, and semiconductor device |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6822610B2 (ja) * | 2019-02-14 | 2021-01-27 | Dic株式会社 | 硬化性樹脂組成物、硬化物、酸変性マレイミド樹脂および硬化剤 |
| TWI833906B (zh) * | 2019-04-26 | 2024-03-01 | 日商Dic股份有限公司 | 硬化性樹脂組成物 |
| KR102699413B1 (ko) * | 2019-08-01 | 2024-08-28 | 가부시키가이샤 가네카 | 열경화성 수지 조성물, 열경화성 수지막, 열경화막, 적층체, 그리고 프린트 배선판 및 그의 제조 방법 |
| CN115698178A (zh) * | 2020-05-29 | 2023-02-03 | 蓝立方知识产权有限责任公司 | 无卤素的极低损耗树脂组合物 |
| CN115667393A (zh) * | 2020-09-11 | 2023-01-31 | 电化株式会社 | 组合物及其固化体 |
| CN116410596B (zh) * | 2021-12-31 | 2025-02-14 | 广东生益科技股份有限公司 | 一种树脂组合物及其应用 |
| US11596066B1 (en) * | 2022-03-22 | 2023-02-28 | Thintronics. Inc. | Materials for printed circuit boards |
| CN120230416A (zh) * | 2023-12-29 | 2025-07-01 | 广东生益科技股份有限公司 | 一种树脂组合物及包含其的预浸料、覆金属箔层压板 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001302900A (ja) * | 2000-04-26 | 2001-10-31 | Asahi Kasei Corp | ポリフェニレンエーテル樹脂組成物 |
| JP2003292767A (ja) * | 2002-04-08 | 2003-10-15 | Asahi Kasei Corp | 高分子ワニス |
| JP2005225913A (ja) * | 2004-02-10 | 2005-08-25 | Kyocera Chemical Corp | 難燃性樹脂組成物とそれを用いた樹脂部品および複合部品 |
| JP2010018791A (ja) * | 2008-06-09 | 2010-01-28 | Mitsubishi Gas Chemical Co Inc | ビスマレアミック酸、ビスマレイミドおよびその硬化物 |
| JP2010138364A (ja) * | 2008-02-12 | 2010-06-24 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグおよび金属箔張り積層板 |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5869046A (ja) | 1981-10-21 | 1983-04-25 | 旭化成株式会社 | 積層板及びその成形法 |
| JPS6118937A (ja) | 1984-07-06 | 1986-01-27 | Mitsubishi Electric Corp | 工業用テレビカメラの照明装置 |
| EP0610890A3 (en) * | 1993-02-09 | 1995-09-13 | Mitsubishi Gas Chemical Co | Thermoplastic resin compositions containing polyphenylene ethers. |
| TWI441866B (zh) * | 2006-02-17 | 2014-06-21 | Hitachi Chemical Co Ltd | A thermosetting resin composition of a semi-IPN type composite and a varnish, a prepreg and a metal laminate |
| JP5261943B2 (ja) * | 2006-02-17 | 2013-08-14 | 日立化成株式会社 | セミipn型複合体の熱硬化性樹脂組成物並びにこれを用いたワニス、プリプレグ及び金属張積層板 |
| JP2011225639A (ja) | 2010-04-15 | 2011-11-10 | Hitachi Chem Co Ltd | 熱硬化性樹脂組成物並びにこれを用いた樹脂ワニス、プリプレグ及び金属張積層板 |
| US9422412B2 (en) * | 2011-09-22 | 2016-08-23 | Elite Material Co., Ltd. | Halogen-free resin composition and copper clad laminate and printed circuit board using same |
| CN103013110B (zh) * | 2011-09-27 | 2014-12-17 | 台光电子材料股份有限公司 | 无卤素树脂组成物及应用其的铜箔基板及印刷电路板 |
| CN103012688A (zh) * | 2012-12-27 | 2013-04-03 | 哈尔滨工业大学 | 一种聚苯醚接枝n-取代马来酰亚胺及其制备方法 |
| CN106255713B (zh) * | 2014-04-04 | 2017-12-19 | 日立化成株式会社 | 聚苯醚衍生物、热固化性树脂组合物、树脂清漆、预浸渍体、层叠板及多层印制线路板 |
-
2016
- 2016-04-28 WO PCT/JP2016/063497 patent/WO2016175325A1/ja not_active Ceased
- 2016-04-28 JP JP2017515634A patent/JP6705446B2/ja active Active
- 2016-04-28 EP EP16786601.1A patent/EP3290479A4/en active Pending
- 2016-04-28 SG SG11201708808SA patent/SG11201708808SA/en unknown
- 2016-04-28 US US15/570,174 patent/US10519279B2/en active Active
- 2016-04-28 KR KR1020177030709A patent/KR102466877B1/ko active Active
- 2016-04-28 CN CN201680024864.4A patent/CN107531992B/zh active Active
- 2016-04-29 TW TW105113506A patent/TWI721978B/zh active
-
2019
- 2019-11-07 US US16/676,501 patent/US11359055B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001302900A (ja) * | 2000-04-26 | 2001-10-31 | Asahi Kasei Corp | ポリフェニレンエーテル樹脂組成物 |
| JP2003292767A (ja) * | 2002-04-08 | 2003-10-15 | Asahi Kasei Corp | 高分子ワニス |
| JP2005225913A (ja) * | 2004-02-10 | 2005-08-25 | Kyocera Chemical Corp | 難燃性樹脂組成物とそれを用いた樹脂部品および複合部品 |
| JP2010138364A (ja) * | 2008-02-12 | 2010-06-24 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグおよび金属箔張り積層板 |
| JP2010018791A (ja) * | 2008-06-09 | 2010-01-28 | Mitsubishi Gas Chemical Co Inc | ビスマレアミック酸、ビスマレイミドおよびその硬化物 |
Cited By (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3467028B1 (en) * | 2016-05-31 | 2024-04-24 | Mitsubishi Gas Chemical Company, Inc. | Resin composition, laminate, semiconductor wafer with resin composition layer, substrate for mounting semiconductor with resin composition layer, and semiconductor device |
| JP7656422B2 (ja) | 2018-03-06 | 2025-04-03 | 株式会社レゾナック | プリプレグ、積層板、多層プリント配線板、半導体パッケージ及び樹脂組成物、並びに、プリプレグ、積層板及び多層プリント配線板の製造方法 |
| JPWO2019172342A1 (ja) * | 2018-03-06 | 2021-03-18 | 昭和電工マテリアルズ株式会社 | プリプレグ、積層板、多層プリント配線板、半導体パッケージ及び樹脂組成物、並びに、プリプレグ、積層板及び多層プリント配線板の製造方法 |
| JP2023134512A (ja) * | 2018-03-06 | 2023-09-27 | 株式会社レゾナック | プリプレグ、積層板、多層プリント配線板、半導体パッケージ及び樹脂組成物、並びに、プリプレグ、積層板及び多層プリント配線板の製造方法 |
| WO2020031495A1 (ja) | 2018-08-06 | 2020-02-13 | 大八化学工業株式会社 | 芳香族リン酸エステルを含む熱硬化性樹脂用難燃剤、それを含む熱硬化性樹脂組成物、その硬化物およびその用途 |
| US11965126B2 (en) | 2018-08-06 | 2024-04-23 | Daihachi Chemical Industry Co., Ltd. | Flame retardant for thermosetting resin including aromatic phosphoric acid ester, thermosetting resin composition including same, and cured material and application of same |
| KR20210040388A (ko) | 2018-08-06 | 2021-04-13 | 다이하치 카가쿠 고교 가부시키가이샤 | 방향족 인산 에스테르를 포함하는 열경화성 수지용 난연제, 그것을 포함하는 열경화성 수지 조성물, 그 경화물 및 그 용도 |
| KR20200060292A (ko) * | 2018-11-16 | 2020-05-29 | 리쇼 고교 가부시키가이샤 | 수지 조성물, 프리프레그, 및 적층판 |
| JP2020083931A (ja) * | 2018-11-16 | 2020-06-04 | 利昌工業株式会社 | 樹脂組成物、プリプレグ、および、積層板 |
| KR102141432B1 (ko) * | 2018-11-16 | 2020-08-05 | 리쇼 고교 가부시키가이샤 | 수지 조성물, 프리프레그, 및 적층판 |
| WO2020157828A1 (ja) * | 2019-01-29 | 2020-08-06 | 日立化成株式会社 | 樹脂組成物、半導体装置の製造方法及び半導体装置 |
| JP7272068B2 (ja) | 2019-04-03 | 2023-05-12 | 株式会社レゾナック | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び半導体パッケージ |
| US11905412B2 (en) | 2019-04-03 | 2024-02-20 | Resonac Corporation | Resin composition, prepreg, laminate, multilayer printed wiring board, and semiconductor package |
| WO2020204175A1 (ja) * | 2019-04-03 | 2020-10-08 | 日立化成株式会社 | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び半導体パッケージ |
| JP7480777B2 (ja) | 2019-04-03 | 2024-05-10 | 株式会社レゾナック | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び半導体パッケージ |
| JPWO2020204175A1 (ja) * | 2019-04-03 | 2020-10-08 | ||
| JP2020169273A (ja) * | 2019-04-03 | 2020-10-15 | 日立化成株式会社 | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び半導体パッケージ |
| JPWO2022124129A1 (ja) * | 2020-12-09 | 2022-06-16 | ||
| JPWO2022124130A1 (ja) * | 2020-12-09 | 2022-06-16 | ||
| WO2022124129A1 (ja) * | 2020-12-09 | 2022-06-16 | 三菱瓦斯化学株式会社 | 樹脂組成物、プリプレグ、樹脂シート、金属箔張積層板、及びプリント配線板 |
| WO2022124130A1 (ja) * | 2020-12-09 | 2022-06-16 | 三菱瓦斯化学株式会社 | 銅箔張積層板及びプリント配線板 |
| WO2022149440A1 (ja) * | 2021-01-06 | 2022-07-14 | 昭和電工マテリアルズ株式会社 | 熱硬化性樹脂組成物、プリプレグ、積層板、金属張り積層板、プリント配線板及び高速通信対応モジュール |
| JPWO2022149440A1 (ja) * | 2021-01-06 | 2022-07-14 | ||
| US12168709B2 (en) | 2021-01-06 | 2024-12-17 | Resonac Corporation | Thermosetting resin composition, prepreg, laminate, metal-clad laminate, printed wiring board, and high-speed communication compatible module |
| JP2021075729A (ja) * | 2021-01-26 | 2021-05-20 | 昭和電工マテリアルズ株式会社 | 樹脂組成物、プリプレグ、積層板及び多層プリント配線板 |
| WO2022163505A1 (ja) | 2021-01-27 | 2022-08-04 | デンカ株式会社 | 球状アルミナ粒子及びその製造方法、並びに樹脂組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107531992B (zh) | 2021-06-11 |
| US20180127547A1 (en) | 2018-05-10 |
| JPWO2016175325A1 (ja) | 2018-02-22 |
| EP3290479A1 (en) | 2018-03-07 |
| KR20180002625A (ko) | 2018-01-08 |
| US20200071464A1 (en) | 2020-03-05 |
| US10519279B2 (en) | 2019-12-31 |
| TW201702310A (zh) | 2017-01-16 |
| KR102466877B1 (ko) | 2022-11-11 |
| EP3290479A4 (en) | 2018-09-19 |
| CN107531992A (zh) | 2018-01-02 |
| US11359055B2 (en) | 2022-06-14 |
| SG11201708808SA (en) | 2017-12-28 |
| JP6705446B2 (ja) | 2020-06-03 |
| TWI721978B (zh) | 2021-03-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6705446B2 (ja) | 熱硬化性樹脂組成物、プリプレグ、積層板及び多層プリント配線板 | |
| JP6705447B2 (ja) | 樹脂組成物、プリプレグ、積層板及び多層プリント配線板 | |
| JP6079930B2 (ja) | N−置換マレイミド基を有するポリフェニレンエーテル誘導体、並びにそれを用いた熱硬化性樹脂組成物、樹脂ワニス、プリプレグ、金属張積層板、及び多層プリント配線板 | |
| JP6863126B2 (ja) | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び半導体パッケージ | |
| TWI775544B (zh) | 印刷配線板用的樹脂組成物、帶樹脂層支撐體、預浸體、積層板、多層印刷配線板及其應用、毫米波雷達用印刷配線板 | |
| JP6896993B2 (ja) | 樹脂組成物、プリプレグ、積層板及び多層プリント配線板 | |
| TWI769169B (zh) | 樹脂組成物、樹脂膜、積層板、多層印刷配線板及多層印刷配線板的製造方法 | |
| JP7106819B2 (ja) | 樹脂ワニス、樹脂組成物、プリプレグ、積層板、多層プリント配線板及び樹脂ワニスの保存方法 | |
| JP7656422B2 (ja) | プリプレグ、積層板、多層プリント配線板、半導体パッケージ及び樹脂組成物、並びに、プリプレグ、積層板及び多層プリント配線板の製造方法 | |
| WO2020095422A1 (ja) | 樹脂組成物、樹脂組成物の硬化物、プリプレグ、積層板、樹脂フィルム、多層プリント配線板、ミリ波レーダー用多層プリント配線板及びポリフェニレンエーテル誘導体 | |
| JP2017066280A (ja) | 熱硬化性樹脂組成物とその製造方法、並びに前記熱硬化性樹脂組成物を有するプリプレグ、金属張積層板、及び多層プリント配線板 | |
| JP2017071689A (ja) | 樹脂組成物、プリプレグ、積層板、多層プリント配線板及び多層プリント配線板の製造方法 | |
| JP2021080459A (ja) | 樹脂組成物、プリプレグ、積層板及び多層プリント配線板 | |
| JP6896994B2 (ja) | 樹脂組成物、プリプレグ、積層板及び多層プリント配線板 | |
| JP2021075729A (ja) | 樹脂組成物、プリプレグ、積層板及び多層プリント配線板 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16786601 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017515634 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2016786601 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20177030709 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15570174 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11201708808S Country of ref document: SG |