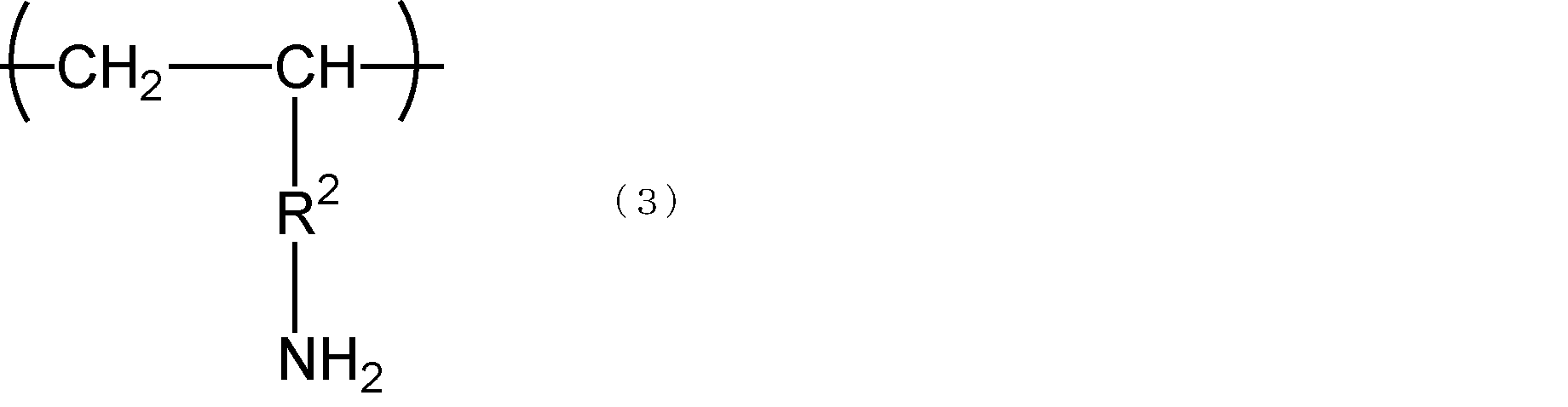WO2016043009A1 - 水溶性包装用フィルム - Google Patents
水溶性包装用フィルム Download PDFInfo
- Publication number
- WO2016043009A1 WO2016043009A1 PCT/JP2015/074217 JP2015074217W WO2016043009A1 WO 2016043009 A1 WO2016043009 A1 WO 2016043009A1 JP 2015074217 W JP2015074217 W JP 2015074217W WO 2016043009 A1 WO2016043009 A1 WO 2016043009A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- water
- packaging film
- soluble packaging
- film
- starch
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/052—Forming heat-sealable coatings
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D65/00—Wrappers or flexible covers; Packaging materials of special type or form
- B65D65/38—Packaging materials of special type or form
- B65D65/40—Applications of laminates for particular packaging purposes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D65/00—Wrappers or flexible covers; Packaging materials of special type or form
- B65D65/38—Packaging materials of special type or form
- B65D65/46—Applications of disintegrable, dissolvable or edible materials
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/042—Coating with two or more layers, where at least one layer of a composition contains a polymer binder
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D103/00—Coating compositions based on starch, amylose or amylopectin or on their derivatives or degradation products
- C09D103/02—Starch; Degradation products thereof, e.g. dextrin
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2329/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal, or ketal radical; Hydrolysed polymers of esters of unsaturated alcohols with saturated carboxylic acids; Derivatives of such polymer
- C08J2329/02—Homopolymers or copolymers of unsaturated alcohols
- C08J2329/04—Polyvinyl alcohol; Partially hydrolysed homopolymers or copolymers of esters of unsaturated alcohols with saturated carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2403/00—Characterised by the use of starch, amylose or amylopectin or of their derivatives or degradation products
- C08J2403/02—Starch; Degradation products thereof, e.g. dextrin
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02W—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO WASTEWATER TREATMENT OR WASTE MANAGEMENT
- Y02W90/00—Enabling technologies or technologies with a potential or indirect contribution to greenhouse gas [GHG] emissions mitigation
- Y02W90/10—Bio-packaging, e.g. packing containers made from renewable resources or bio-plastics
Definitions
- the present invention is capable of suppressing the occurrence of roll contamination, has good feedability and excellent process passability, can improve productivity, and further has water solubility, chemical resistance, visibility and
- the present invention relates to a water-soluble packaging film with improved packaging properties.
- Polyvinyl alcohol (hereinafter also referred to as PVA) is widely used as a packaging material because it is excellent in transparency, oil resistance, chemical resistance, and gas barrier properties such as oxygen. In recent years, it has been widely used as a packaging material for foods, pharmaceuticals, industrial chemicals, agricultural chemicals and the like in which deterioration due to oxidation has a great influence on properties.
- PVA has high water solubility, it is utilized also as a temporary protective film or sheet
- PVA has a problem that its water solubility decreases when it receives a thermal history during surface protection.
- the protective film may receive a heat history in a process such as hot pressing, and in such a case, there is a problem that PVA cannot be sufficiently removed due to a decrease in water solubility. there were.
- Patent Document 1 contains 20 to 40 parts by weight of a plasticizer and 10 to 35 parts by weight of starch with respect to 100 parts by weight of polyvinyl alcohol, thereby suppressing a decrease in water solubility and heat sealing. And techniques for improving processability and process passability are described.
- the method of Patent Document 1 has a problem that the film is still insufficient and the feeding property is inferior.
- action which improves pay-out property cannot be acquired only by mix
- Patent Document 1 describes a technique for embossing the surface of a water-soluble film as a method for improving the blocking property and process passability of the water-soluble film.
- embossing is performed, the transparency of the water-soluble film is impaired, and there is a problem that the visibility for discriminating the inclusion of chemicals such as agricultural chemicals and detergents deteriorates.
- Patent Document 2 describes a method of adding a polybasic acid or an alkali metal salt thereof and a hydrazine compound to a polyvinyl alcohol resin. Has been.
- Patent Document 3 discloses a water-soluble film in which a plasticizer and sulfite are added to a polyvinyl alcohol resin, which is less colored at the time of film formation. It is said that a water-soluble film with little coloring is obtained.
- the dissolution time cannot be shortened even if sulfite is added to the water-soluble film.
- sulfite reacts with a weak acid to generate sulfur dioxide gas, and there is also a problem that a bad odor is generated from the water-soluble film.
- the present invention is capable of suppressing the occurrence of roll contamination, has good feedability and excellent process passability, can improve productivity, and further has water solubility, chemical resistance, visibility and
- An object is to provide a water-soluble packaging film having improved packaging properties.
- the present invention contains polyvinyl alcohol, a plasticizer and starch, and the starch is applied to the front and back surfaces of the water-soluble packaging film, and the amount of the starch applied per 1 m 2 of the water-soluble packaging film. It is a water-soluble packaging film having a contact angle of 20 to 50 ° as measured with distilled water by a droplet method, and having a contact angle of 20 to 50 °.
- the present invention is described in detail below.
- the present inventor added a plasticizer as a component of the water-soluble packaging film, in addition to polyvinyl alcohol, and applied a predetermined amount of starch to the front and back surfaces of the water-soluble packaging film, so that the contact angle is suitable.
- a plasticizer as a component of the water-soluble packaging film, in addition to polyvinyl alcohol, and applied a predetermined amount of starch to the front and back surfaces of the water-soluble packaging film, so that the contact angle is suitable.
- the water-soluble packaging film of the present invention contains polyvinyl alcohol.
- the polyvinyl alcohol is a main constituent of the water-soluble packaging film of the present invention.
- the polyvinyl alcohol is obtained by polymerizing a vinyl ester to obtain a polymer according to a conventionally known method, and then saponifying the polymer, that is, hydrolyzing.
- saponification an alkali or an acid is generally used. It is preferable to use an alkali for the saponification.
- As said polyvinyl alcohol only 1 type may be used and 2 or more types may be used together.
- vinyl ester examples include vinyl acetate, vinyl formate, vinyl propionate, vinyl butyrate, vinyl pivalate, vinyl versatate, vinyl laurate, vinyl stearate and vinyl benzoate.
- the method for polymerizing the vinyl ester is not particularly limited.
- Examples of the polymerization method include a solution polymerization method, a bulk polymerization method, and a suspension polymerization method.
- polymerization catalyst used for polymerizing the vinyl ester examples include 2-ethylhexyl peroxydicarbonate (“TrigonoxEHP” manufactured by Tianjin McEIT), 2,2′-azobisisobutyronitrile (AIBN), t-butyl.
- Peroxyneodecanoate bis (4-tert-butylcyclohexyl) peroxydicarbonate, di-n-propylperoxydicarbonate, di-n-butylperoxydicarbonate, di-cetylperoxydicarbonate and di-s-butylperoxy Examples include dicarbonate.
- the said polymerization catalyst only 1 type may be used and 2 or more types may be used together.
- the polymer obtained by polymerizing the vinyl ester is preferably a polyvinyl ester.
- the polymer obtained by polymerizing the vinyl ester may be a copolymer of the vinyl ester and another monomer. That is, the polyvinyl alcohol may be formed using a copolymer of vinyl ester and another monomer.
- examples of other monomers, that is, comonomers to be copolymerized include olefins, (meth) acrylic acid and salts thereof, (meth) acrylic acid esters, (meth) acrylamide derivatives, N-vinylamides, vinyl ethers, and nitriles.
- the other monomer only one kind may be used, or two or more kinds may be used in combination.
- Examples of the olefins include ethylene, propylene, 1-butene and isobutene.
- the (meth) acrylic acid esters include methyl (meth) acrylate, ethyl (meth) acrylate, n-propyl (meth) acrylate, i-propyl (meth) acrylate, and n- (meth) acrylic acid. Examples include butyl and 2-ethylhexyl (meth) acrylate.
- Examples of the (meth) acrylamide derivative include acrylamide, n-methylacrylamide, N-ethylacrylamide, N, N-dimethylacrylamide, (meth) acrylamidepropanesulfonic acid and salts thereof, and the like.
- N-vinylamides examples include N-vinylpyrrolidone.
- vinyl ethers include methyl vinyl ether, ethyl vinyl ether, n-propyl vinyl ether, i-propyl vinyl ether, and n-butyl vinyl ether.
- nitriles include (meth) acrylonitrile.
- vinyl halides include vinyl chloride and vinylidene chloride.
- allyl compound include allyl acetate and allyl chloride.
- Examples of the vinylsilyl compound include vinyltrimethoxysilane.
- the modification amount is preferably 15 mol% or less, more preferably 5 mol% or less. That is, in the total 100 mol% of the content of the structural unit derived from the vinyl ester in the modified PVA and the content of the structural unit derived from the other monomer, the structural unit derived from the vinyl ester is preferably 85 mol%. As mentioned above, More preferably, it is 95 mol% or more, The structural unit derived from said other monomer becomes like this. Preferably it is 15 mol% or less, More preferably, it is 5 mol% or less.
- polyvinyl alcohol includes modified polyvinyl alcohol (modified PVA).
- the modified PVA is preferably modified with a hydrophilic group.
- a hydrophilic group for example, at least one selected from the group consisting of a sulfonic acid group, a pyrrolidone ring group, an amino group, and a carboxyl group is preferable. Of these, a sulfonic acid group and a pyrrolidone ring group are more preferable.
- the hydrophilic group includes salts such as sodium and potassium in addition to the functional group described above.
- the modified PVA modified with the hydrophilic group is obtained by copolymerizing the polyvinyl alcohol and another monomer having the hydrophilic group, or adding a hydrophilic group to the polyvinyl alcohol. The thing obtained by is also included.
- modified PVA examples include pyrrolidone ring-modified polyvinyl alcohol, sodium sulfonate-modified polyvinyl alcohol, amino group-modified polyvinyl alcohol, and carboxyl group-modified polyvinyl alcohol.
- the pyrrolidone ring-modified polyvinyl alcohol preferably has a structural unit represented by the following formula (1).
- the sodium sulfonate-modified polyvinyl alcohol preferably has a structural unit represented by the following formula (2).
- R 1 represents an alkylene group having 1 to 4 carbon atoms.
- the amino group-modified polyvinyl alcohol preferably has a structural unit represented by the following formula (3).
- R 2 represents a single bond or an alkylene group having 1 to 10 carbon atoms.
- the carboxyl group-modified polyvinyl alcohol has a structural unit represented by the following formula (4-1), (4-2) or (4-3). Is preferred.
- X 1 , X 2 , X 3 , X 4 and X 5 are each independently a hydrogen atom, a metal atom or a methyl group.
- the carboxyl group contained in the structural unit having a carboxyl group includes a salt of the carboxyl group and a methyl ester.
- a metal atom a sodium atom etc. are mentioned, for example.
- R 3 represents an alkylene group having 1 to 10 carbon atoms.
- modified PVA examples include “KL-318”, “KL-118”, “KM-618”, and “KM-118” (all manufactured by Kuraray Co., Ltd.).
- the content of the structural unit having a hydrophilic group in the modified PVA is preferably 0.1 mol%, more preferably 0.2 mol%, still more preferably 0.5 mol%, and particularly preferably 1 mol.
- the preferable upper limit is 15 mol%, the more preferable upper limit is 10 mol%, and the more preferable upper limit is 8 mol%.
- the film may be discolored or broken over a long period of time against chlorine-containing sanitary agents or oxidizing chemicals. A film with no resistance can be obtained.
- the saponification degree of the polyvinyl alcohol has a preferred lower limit of 80 mol%, a more preferred lower limit of 85 mol%, a still more preferred lower limit of 88 mol%, a particularly preferred lower limit of 90 mol%, and a more particularly preferred lower limit of 92 mol%.
- the preferred upper limit is 99.9 mol%, the more preferred upper limit is 99.0 mol%, the still more preferred upper limit is 98 mol%, the particularly preferred upper limit is 97 mol%, more particularly preferably 96 mol%.
- the saponification degree of the PVA is particularly preferably 85 mol% or more and 96 mol% or less.
- the saponification degree is measured according to JIS K6726.
- the saponification degree indicates the proportion of units that are actually saponified to vinyl alcohol units among the units converted to vinyl alcohol units by saponification.
- the method for adjusting the saponification degree is not particularly limited.
- the saponification degree can be appropriately adjusted according to saponification conditions, that is, hydrolysis conditions.
- the PVA preferably has a saponification degree distribution standard deviation ( ⁇ ) of 0.1 to 1.0 mol%.
- ⁇ saponification degree distribution standard deviation
- the more preferable lower limit of the saponification degree distribution standard deviation of the PVA is 0.2 mol%, and the more preferable upper limit is 0.9 mol%.
- the saponification degree distribution standard deviation is an index indicating the saponification degree variation in PVA and can be measured and calculated using, for example, FT-IR.
- the degree of polymerization of the PVA is not particularly limited.
- the preferable lower limit of the polymerization degree of the PVA is 400, the more preferable lower limit is 500, the still more preferable lower limit is 600, the particularly preferable lower limit is 900, the preferable upper limit is 2000, the more preferable upper limit is 1800, and the further preferable upper limit is 1500.
- the polymerization degree is not less than the above lower limit and not more than the above upper limit, the viscosity of the aqueous solution becomes appropriate when a water-soluble packaging film is formed.
- the degree of polymerization is not more than the above upper limit, the strength of the water-soluble packaging film is further increased and water resistance is obtained.
- the degree of polymerization is measured according to JIS K6726.
- the preferable lower limit of the viscosity measured at 20 ° C. is 3 mPa ⁇ s
- the preferable upper limit is 35 mPa ⁇ s
- the more preferable lower limit is 5 mPa ⁇ s
- the more preferable upper limit is 30 mPa ⁇ s.
- Water resistance can be improved by making the said viscosity into 3 mPa * s or more. By setting the viscosity to 35 mPa ⁇ s or less, the dissolution time can be shortened.
- the more preferable lower limit of the viscosity is 8 mPa ⁇ s
- the more preferable upper limit is 20 mPa ⁇ s.
- the viscosity can be measured according to JIS K6726.
- the preferable lower limit of the content of the polyvinyl alcohol is preferably 70% by weight, and the preferable upper limit is 97% by weight.
- the content of the polyvinyl alcohol is not less than the above lower limit, the water-soluble packaging film may have a better quality without bleeding out of the plasticizer from the water-soluble packaging film.
- the content of the polyvinyl alcohol is not more than the above upper limit, the strength of the water-soluble packaging film is further increased, and water resistance may be obtained.
- the water-soluble packaging film of the present invention contains a plasticizer.
- Water-soluble packaging films are required to have high tensile strength and durability because they can be transported, stored, and used in hot and humid regions and cold regions. Particularly, impact resistance at low temperatures is important.
- the water-soluble packaging film of the present invention contains a plasticizer, the glass transition point can be lowered, and durability at low temperatures can be improved.
- the solubility with respect to the water of the film for water-soluble packaging can also be improved by containing the said plasticizer.
- the plasticizer is not particularly limited as long as it is generally used as a plasticizer for PVA, and examples thereof include glycerin, diglycerin, diethylene glycol, trimethylolpropane, triethylene glycol, dipropylene glycol, and propylene glycol.
- Polyethylene alcohols such as polyhydric alcohols, polyethers such as polyethylene glycol and polypropylene glycol, phenol derivatives such as bisphenol A and bisphenol S, amide compounds such as N-methylpyrrolidone, and polyhydric alcohols such as glycerin, pentaerythritol and sorbitol with ethylene oxide. Examples include added compounds and water. These may be used alone or in combination of two or more.
- glycerin, trimethylolpropane, polyethylene glycol, polypropylene glycol, triethylene glycol, dipropylene glycol, and propylene glycol are preferable because water solubility can be improved, and the effect of improving water solubility is particularly large. Therefore, glycerin and trimethylolpropane are particularly preferable.
- the preferred lower limit of the plasticizer content relative to 100 parts by weight of polyvinyl alcohol is 3 parts by weight, and the preferred upper limit is 15 parts by weight.
- the content of the plasticizer is less than 3 parts by weight, the plasticizer blending effect may not be obtained.
- the blending ratio of the plasticizer exceeds 15 parts by weight, the bleedout of the plasticizer becomes large, and the anti-blocking property of the resulting water-soluble packaging film may be deteriorated.
- the minimum with said more preferable plasticizer content is 3.2 weight part, and a more preferable upper limit is 13 weight part.
- the water-soluble packaging film of the present invention contains starch.
- the starch is applied to the front and back surfaces of the water-soluble packaging film.
- the water-soluble packaging film of the present invention can be improved in feeding performance by applying the starch, and can be excellent in anti-blocking property and process passability.
- the surface and the back surface of the water-soluble packaging film refer to the main surface located on one main surface and the opposite side of the water-soluble packaging film.
- the term “application” includes spraying, dipping and the like in addition to application, and means that starch is attached to at least a part of the front surface and the back surface.
- the starch used in the present invention is not particularly limited, for example, starch derived from corn, wheat, potato, tapioca, taro, sweet potato, rice and the like, pregelatinized starch, dextrin, oxidized starch, aldehyde starch, Examples thereof include modified starches such as esterified starch, etherified starch, cationized starch and crosslinked starch, raw corn starch, oxidized corn starch, etherified corn starch, phosphate esterified potato starch and acetylated wheat starch. These may be used alone or in combination of two or more.
- the compatibility of polyvinyl alcohol and starch is somewhat low, and the film surface is preferably a pear ground, corn-derived starch, sweet potato Derived starch, raw corn starch, oxidized corn starch, etherified corn starch, phosphate esterified potato starch, acetylated wheat starch and the like are preferred. Furthermore, corn-derived starch, sweet potato-derived starch, and oxidized corn starch are particularly preferred because fine irregularities are favorably formed on the film surface.
- the lower limit per 1 m 2 of the water-soluble packaging film is 0.001 g, and the upper limit is 0.10 g.
- the coating amount of the starch is 0.001 g or more per 1 m 2 of the water-soluble packaging film, the feeding property can be improved, and the antiblocking property and process passability can be improved.
- the coating amount of the starch is 0.10 g or less per 1 m 2 of the water-soluble packaging film, it is possible to suppress the starch from adhering to the transport roll during the process of packaging the drug, thereby improving productivity. be able to.
- the preferable lower limit per 1 m 2 of water-soluble packaging film is 0.005 g, and the preferable upper limit is 0.07 g.
- the said application amount means the quantity of the starch apply
- the minimum with preferable content of the said starch with respect to 100 weight part of polyvinyl alcohol resin is 0.001 weight part, and a preferable upper limit is 1 weight part.
- the starch content relative to 100 parts by weight of the polyvinyl alcohol resin is 0.001 part by weight or more, the feeding property can be improved, and the anti-blocking property and the process passability can be improved.
- it is 1 part by weight or less it is possible to suppress starch from adhering to the transport roll during the process of packaging the medicine, and to improve productivity.
- the minimum with more preferable content of the said starch with respect to 100 weight part of polyvinyl alcohol resin is 0.05 weight part, and a more preferable upper limit is 0.7 weight part.
- a granular form, powder form, flake form, etc. are mentioned. Especially, it is preferable that it is a powder form.
- the preferable lower limit of the average particle diameter of the starch is 5 ⁇ m, and the preferable upper limit is 40 ⁇ m.
- the visibility of a film can be maintained as the said average particle diameter is 5 micrometers or more.
- the water-soluble packaging film which suppressed roll contamination as it is 40 micrometers or less can be obtained.
- the average particle diameter can be measured with a particle size distribution meter such as an optical microscope, an electron microscope, or laser diffraction.
- the water-soluble packaging film of the present invention preferably has an orientation of 0.5 to 9.9 nm.
- the orientation is not less than the above lower limit and not more than the above upper limit, the solubility and packaging properties of the water-soluble packaging film are improved, the dissolution time when the contents are opened, and the storage when the contents are packaged Both sexes can be improved in a balanced manner.
- the more preferable lower limit of the orientation is 1.0 nm, and the more preferable upper limit is 8.0 nm.
- the orientation is an index indicating the regularity of the PVA molecular arrangement in the water-soluble packaging film, and can be measured, for example, by using a phase difference measuring device (KOBRA-WR, manufactured by Oji Scientific Instruments). .
- the preferable upper limit of the thickness of the water-soluble packaging film of the present invention is 100 ⁇ m, the more preferable upper limit is 80 ⁇ m, and the still more preferable upper limit is 75 ⁇ m.
- the preferable lower limit of the thickness of the water-soluble packaging film of the present invention is 10 ⁇ m.
- medical agent becomes still higher that the thickness of the said water-soluble packaging film is more than the said minimum.
- the water-soluble packaging film of the present invention may further contain, as necessary, usual additives such as a colorant, a fragrance, an extender, an antifoaming agent, a release agent, an ultraviolet absorber, and a surfactant.
- usual additives such as a colorant, a fragrance, an extender, an antifoaming agent, a release agent, an ultraviolet absorber, and a surfactant.
- the lower limit of the contact angle measured using distilled water by the droplet method is 20 °, and the upper limit is 50 °.
- the contact angle is 20 ° or more, a water-soluble packaging film having excellent drawability can be obtained.
- the contact angle is 50 ° or less, a water-soluble packaging film in which roll contamination is suppressed can be obtained.
- the preferable lower limit of the contact angle is 23 °, the more preferable lower limit is 30 °, the preferable upper limit is 49 °, and the more preferable upper limit is 40 °.
- the contact angle can be measured according to JIS R 3257. For example, it can be measured by dropping distilled water onto a water-soluble packaging film using a contact angle meter Theta Light (manufactured by Biolin Scientific).
- Method for producing water-soluble packaging film Although it does not specifically limit as a manufacturing method of the water-soluble packaging film of this invention, After casting the PVA aqueous solution containing PVA, a plasticizer, and water to a supporting member and drying, the surface of the obtained PVA film and A method of applying starch on the back surface can be used. Specific examples include a solution casting method (casting method), a roll coating method, a spin coating method, a screen coating method, a fountain coating method, a dipping method, and a spray method.
- the PVA aqueous solution contains water together with the PVA and the plasticizer.
- the PVA is mainly dissolved in the water.
- the content of the water is 300 parts by weight or more, preferably 400 parts by weight or more, more preferably 500 parts by weight or more with respect to 100 parts by weight of components other than water containing the PVA.
- the water content is 900 parts by weight or less, preferably 800 parts by weight or less, more preferably 700 parts by weight or less.
- the viscosity of the PVA aqueous solution becomes moderately high, the casting of the PVA aqueous solution becomes easy, the drying time is further shortened, and the orientation of the water-soluble packaging film is more An even higher and better quality water-soluble packaging film is obtained.
- the support member is preferably capable of supporting the PVA film obtained by maintaining the PVA aqueous solution on the surface and casting the PVA aqueous solution.
- the material for the support member include polyolefin, polyester, acrylic resin, and the like. You may use the supporting member formed with materials other than these.
- the polyolefin include ethylene, polypropylene, ethylene-vinyl acetate copolymer, and ethylene-vinyl alcohol copolymer.
- Examples of the polyester include polyethylene terephthalate and polyethylene naphthalate.
- the material of the support member is preferably not PVA.
- the drying method after casting the PVA aqueous solution on the support member can be any suitable method and is not particularly limited.
- Examples of the drying method include a method of natural drying and a method of heat drying at a temperature lower than the glass transition temperature of PVA.
- coating starch to the said PVA film is not specifically limited, For example, methods, such as spraying (spray, mist), transfer (sponge roll, gravure roll), immersion (dipping), can be used.
- the stretching step may be performed during or after the drying. By performing such a process, the orientation of the water-soluble packaging film can be more suitably improved.
- the stretching step include stretching using a roll, stretching using a tenter, stretching using a winding device, stretching using drying shrinkage, and stretching combining these.
- the stretching ratio in the stretching step is preferably 1.01 to 3 times. Further, 1.1 to 2 times is more preferable.
- the use of the water-soluble packaging film include a medicine packaging film used for medicine packaging such as detergents, agricultural chemicals, and pharmaceuticals.
- the present invention it is possible to suppress the occurrence of roll contamination, and it is possible to improve productivity because it has good feedability and excellent process passability. Furthermore, water solubility, chemical resistance, visual recognition Water-soluble packaging film having improved properties and packaging properties can be provided.
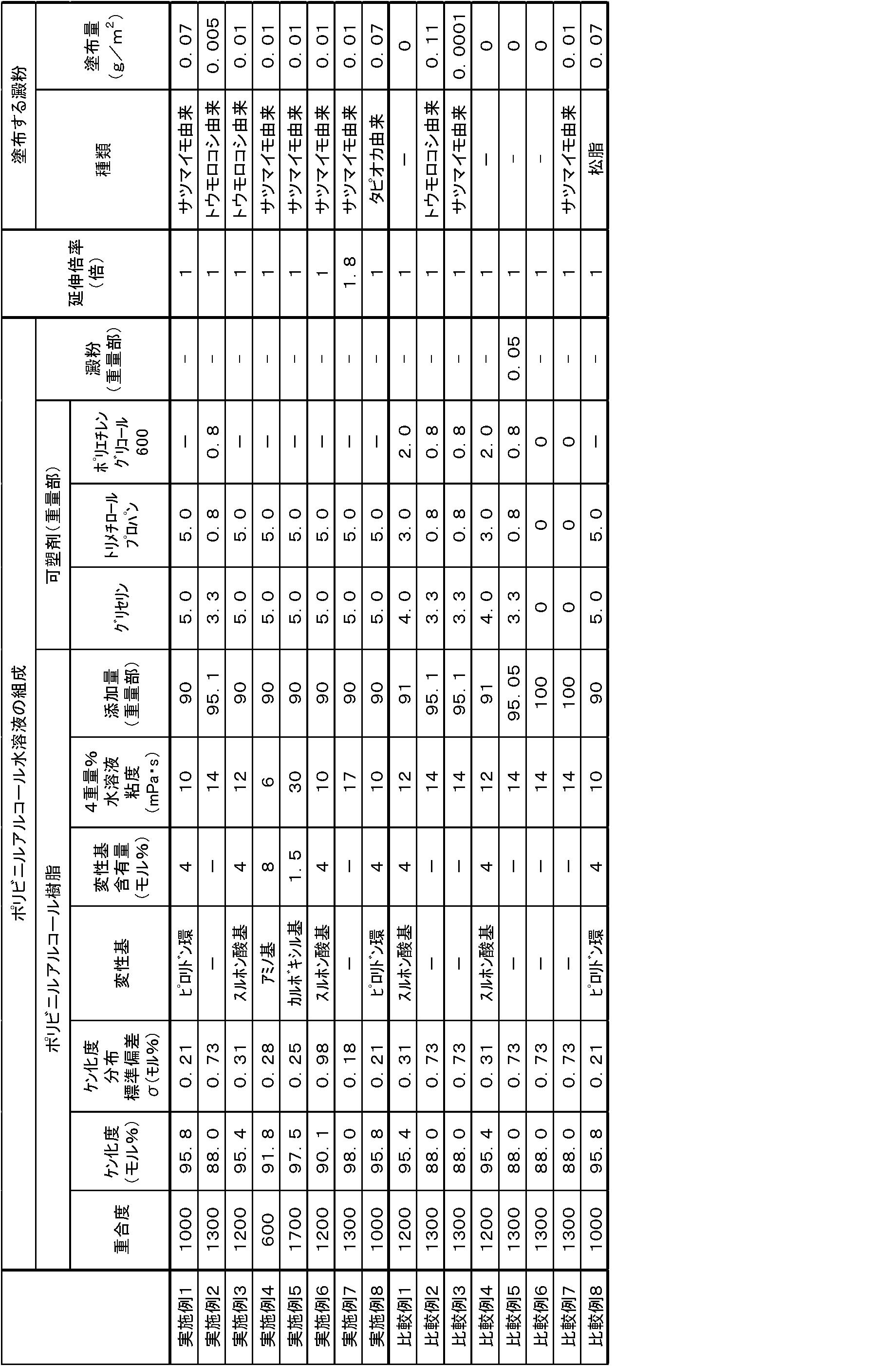
- Example 1 Pyrrolidone ring-modified polyvinyl alcohol having a structural unit represented by the above formula (1) as polyvinyl alcohol (polymerization degree 1000, saponification degree 95.8 mol%, saponification degree distribution standard deviation 0.21 mol%, pyrrolidone ring modification 4.
- the standard deviation of saponification degree distribution of the polyvinyl alcohol was measured by the following procedure. (Saponification degree distribution standard deviation measurement) Forty particles were randomly selected from the polyvinyl alcohol particles, and the amount of acetyl groups of the polyvinyl alcohol was measured using ATR-specific FT-IR (manufactured by Shimadzu Corporation, IRAffinity-1). The saponification degree of each particle was obtained from the obtained acetyl group amount, and the standard deviation ( ⁇ ) of the saponification degree distribution was calculated from the variation in the saponification degree.
- the obtained PVA aqueous solution was coated on a polyethylene terephthalate (PET) film (thickness 50 ⁇ m) as a support member by a lip coater method, dried at 70 ° C. for 10 minutes, and then dried at 110 ° C. for 10 minutes.
- a wound body of a laminated film in which a PVA film (thickness: 50 ⁇ m) was laminated on a support member was obtained by winding it around a 3-inch paper core.
- the PVA film was obtained by peeling the support from the wound body of the obtained laminated film, and then derived from sweet potato having an average particle diameter of 20 ⁇ m using a powder spray device (“K-III” manufactured by Nikka).
- the starch was applied to the front and back surfaces of the PVA film so that the amount of starch (manufactured by Wako Pure Chemical Industries, Ltd.) was 0.07 g per 1 m 2 of the water-soluble packaging film to obtain a water-soluble packaging film.
- Example 2 Unmodified polyvinyl alcohol as polyvinyl alcohol (polymerization degree 1300, saponification degree 88.0 mol%, saponification degree distribution standard deviation 0.73 mol%, 4 wt% aqueous solution viscosity 14 mPa ⁇ s) 95.1 parts by weight, plastic As an agent, 3.3 parts by weight of glycerin (manufactured by Wako Pure Chemical Industries), 0.8 part by weight of trimethylolpropane (manufactured by Wako Pure Chemical Industries), 0.8 weight of polyethylene glycol 600 (manufactured by Wako Pure Chemical Industries) A part was dissolved in 667 parts by weight of water to prepare a 15% by weight aqueous solution.
- glycerin manufactured by Wako Pure Chemical Industries
- trimethylolpropane manufactured by Wako Pure Chemical Industries
- polyethylene glycol 600 manufactured by Wako Pure Chemical Industries
- Example 2 In the same manner as in Example 1, a wound body of a laminated film in which a PVA film (thickness 50 ⁇ m) was laminated was obtained. After the support was peeled off from the wound body of the obtained laminated film to obtain a PVA film, corn-derived starch having an average particle size of 20 ⁇ m (manufactured by Wako Pure Chemical Industries, Ltd.) was obtained in the same manner as in Example 1. Starch was applied to the front and back surfaces of the PVA film so that the coating amount was 0.005 g per 1 m 2 of the water-soluble packaging film to obtain a water-soluble packaging film.
- Example 3 Polyvinyl alcohol having a structural unit represented by the above formula (2), in which R 1 is a 2-methylenepropylene group, sodium sulfonate-modified polyvinyl alcohol A (degree of polymerization 1200, degree of saponification) 95.4 mol%, saponification degree distribution standard deviation 0.31 mol%, sulfonic acid group modification amount 4 mol%, 4 wt% aqueous solution viscosity 12 mPa ⁇ s)
- a water-soluble packaging film was obtained in the same manner as in Example 1 except that the amount was 0.01 g per 1 m 2 of the water-soluble packaging film.
- Example 4 Polyvinyl alcohol having a structural unit represented by the above formula (3), wherein R 2 is a single bond in the above formula (3) (polymerization degree 600, saponification degree 91.8 mol%) , Saponification degree distribution standard deviation 0.28 mol%, amino group modification amount 8 mol%, 4 wt% aqueous solution viscosity 6 mPa ⁇ s) and average particle size 10 ⁇ m sweet potato-derived starch (manufactured by Wako Pure Chemical Industries, Ltd.)
- a water-soluble packaging film was obtained in the same manner as in Example 1 except that the coating amount was 0.01 g per 1 m 2 of the water-soluble packaging film.
- Example 5 Polyvinyl alcohol having a structural unit represented by the above formula (4-2) as the polyvinyl alcohol, wherein R 3 is a methylene group, and X 2 and X 3 are sodium atoms in the above formula (4-2) (Kuraray Co., Ltd., KL-118, polymerization degree 1700, saponification degree 97.5 mol%, saponification degree distribution standard deviation 0.25 mol%, carboxyl group modification amount 1.5 mol%, 4 wt% aqueous solution viscosity 30 mPa S), and the application amount of starch derived from sweet potato having an average particle size of 30 ⁇ m (manufactured by Wako Pure Chemical Industries, Ltd.) was changed to 0.01 g per 1 m 2 of water-soluble packaging film, in the same manner as in Example 1. A water-soluble packaging film was obtained.
- Example 7 Unmodified polyvinyl alcohol (polymerization degree 1300, saponification degree 98.0 mol%, saponification degree distribution standard deviation 0.18 mol%, 4 wt% aqueous solution viscosity 17 mPa ⁇ s) 90 parts by weight, plasticizer glycerin ( 5.0 parts by weight of Wako Pure Chemical Industries, Ltd.) and 5.0 parts by weight of trimethylolpropane (Wako Pure Chemical Industries, Ltd.) were dissolved in 667 parts by weight of water to prepare a 15% by weight aqueous solution.
- the obtained PVA aqueous solution was applied onto a polyethylene terephthalate (PET) film (thickness 50 ⁇ m) as a supporting member using an auto film applicator (“PI-1210” manufactured by Tester Sangyo Co., Ltd.) and dried at 80 ° C. for 20 minutes. And a PVA film (thickness 50 ⁇ m) was obtained on the support member. After removing the support from the obtained PVA film to obtain a water-soluble packaging film, the film was exposed to an environment of a temperature of 23 ° C. and a relative humidity of 50% RH for 24 hours.
- PET polyethylene terephthalate
- PI-1210 manufactured by Tester Sangyo Co., Ltd.
- the water-soluble packaging film was cut into a size of 600 mm ⁇ 500 mm and set in a tensile tester so that the width was 500 mm. Then, after extending
- Example 8 A water-soluble packaging film was prepared in the same manner as in Example 1 except that starch derived from sweet potato having an average particle size of 20 ⁇ m (manufactured by Wako Pure Chemical Industries, Ltd.) was changed to starch derived from tapioca having an average particle size of 20 ⁇ m (manufactured by GABAN). Obtained.
- a 15% by weight aqueous solution was prepared.
- a wound body of a laminated film in which a PVA film (thickness 50 ⁇ m) was laminated was obtained. From the wound body of the obtained laminated film, the support was peeled off to obtain a water-soluble packaging film.
- Example 2 A water-soluble packaging film was obtained in the same manner as in Example 2 except that the amount of starch derived from corn having an average particle size of 20 ⁇ m was 0.11 g per 1 m 2 of the water-soluble packaging film.
- Example 3 A water-soluble packaging film was obtained in the same manner as in Example 2 except that the amount of the starch derived from sweet potato having an average particle size of 30 ⁇ m was 0.0001 g per 1 m 2 of the water-soluble packaging film.
- Comparative Example 4 The water-soluble packaging film obtained in Comparative Example 1 was embossed in a lattice shape, the thickness of the concave portion was 17 ⁇ m, the height difference between the concave portion and the convex portion was 30 ⁇ m, the groove width was 0.1 mm, and the groove spacing was 0. A water-soluble packaging film of 1 mm was obtained.
- Example 2 In the same manner as in Example 1, a wound body of a laminated film in which a PVA film (thickness 50 ⁇ m) was laminated was obtained. From the wound body of the obtained laminated film, the support was peeled off to obtain a water-soluble packaging film. In addition, the obtained water-soluble packaging film had starch inside the film, and there was no precipitation of starch on the film surface.
- Example 6 As polyvinyl alcohol, 100 parts by weight of unmodified polyvinyl alcohol (polymerization degree 1300, saponification degree 88.0 mol%, saponification degree distribution standard deviation 0.73 mol%, 4 wt% aqueous solution viscosity 14 mPa ⁇ s) 667 weight parts water A 15% by weight aqueous solution was prepared by dissolving in part. In the same manner as in Example 1, a wound body of a laminated film in which a PVA film (thickness 50 ⁇ m) was laminated was obtained. From the wound body of the obtained laminated film, the support was peeled off to obtain a water-soluble packaging film.
- Comparative Example 7 Water-soluble in the same manner as in Comparative Example 6 except that starch was applied to the front and back surfaces of the PVA film so that the amount of starch derived from sweet potato having an average particle size of 20 ⁇ m was 0.01 g per 1 m 2 of water-soluble packaging film. A packaging film was obtained.
- Example 8 A water-soluble packaging film was obtained in the same manner as in Example 1 except that sweet potato-derived starch having an average particle size of 20 ⁇ m (manufactured by Wako Pure Chemical Industries, Ltd.) was changed to pine resin having an average particle size of 20 ⁇ m (manufactured by MIKASA).
- the obtained water-soluble packaging film was cut into a size of 10 mm ⁇ 30 mm. Next, after being exposed to a 50 ° C. environment for 24 hours, it was attached to a glass substrate. Thereafter, about 5.0 ⁇ L of distilled water was dropped onto the water-soluble packaging film in an atmosphere of 23 ° C. and 50% RH by a droplet method using a contact angle meter Theta light manufactured by Biolin Scientific. The angle formed by the water-soluble packaging film and the tangent of the droplet after 1 second from the dropping of the droplet was measured.
- the obtained water-soluble packaging film was exposed to an environment of a temperature of 23 ° C. and a relative humidity of 50% RH for 24 hours. Thereafter, the water-soluble packaging film was measured with a phase difference measuring device (KOBRA-WR, manufactured by Oji Scientific Instruments) at an incident angle of 45 °, and the measured value of the phase difference R was defined as an orientation value.
- a phase difference measuring device manufactured by Oji Scientific Instruments
- the present invention it is possible to suppress the occurrence of roll contamination, and it is possible to improve productivity because it has good feedability and excellent process passability. Furthermore, water solubility, chemical resistance, visual recognition Water-soluble packaging film having improved properties and packaging properties can be provided.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Mechanical Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Polymers & Plastics (AREA)
- Medicinal Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wrappers (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
- Coating Of Shaped Articles Made Of Macromolecular Substances (AREA)
- Laminated Bodies (AREA)
- Paints Or Removers (AREA)
Abstract
Description
しかしながら、PVAは、表面保護時に熱履歴を受けるとその水溶性が低下するという課題があった。例えば、金属加工時においては、熱プレス等の工程で保護膜が熱履歴を受けることがあり、このような場合に、水溶性の低下に起因して、PVAの除去を充分にできないという問題があった。
しかしながら、PVAは、吸湿性が高く、フィルム化した際に弾性率が不充分でありフィルムに腰がないという問題があった。このため、PVAからなるフィルムは、表面の滑りが悪くスリップ性に劣るものとなり、フィルムの繰り出し性を悪化させる原因となっていた。その結果、製袋機等を用いてヒートシールする際に、ピッチが合わなくなったり、しわが入ったりする等の不具合が生じ、工程通過性に劣るという問題があった。
しかしながら、特許文献1の方法では、フィルムの腰が依然として不充分であり、繰り出し性に劣るという問題があった。また、水溶性フィルムに澱粉を配合するだけでは、繰り出し性を改善する効果を得ることができず、工程通過性を改善する効果が得られない場合もあった。更に、特許文献1の方法では、澱粉を多量に配合する必要があり、澱粉配合量が多いことで、水溶性フィルム表面に塗布すると農薬や洗剤等の薬剤を包装する加工の際に搬送ロールに澱粉が大量に付着してしまい、搬送ロールを清掃する手間が増え、生産性が低下するという問題があった。
しかしながら、エンボス処理を行うと、水溶性フィルムの透明性が損なわれ、農薬や洗剤等の薬剤の内包を判別する視認性が悪化するという問題があった。
また、水溶性包装用フィルムは、袋状とした後、ヒートシール等によって封止されることが多いが、シール部分は耐久性が低下する傾向にあり、内容物を包装して長期間経過すると、環境等の影響により、シール部分に不良が生じる等の不具合が生じ、内容物を長期間安定して包装することができないという問題が生じていた。
しかしながら、水溶性フィルム中に亜硫酸塩を添加しても溶解時間が短縮できないという問題があった。また、亜硫酸塩は弱酸と反応して、二酸化硫黄ガスを発生することがあり、水溶性フィルムから異臭が発生するという問題もあった。
以下、本発明を詳述する。
以下、本発明に係るPVA水溶液に用いられる各成分の詳細を説明する。
本発明の水溶性包装用フィルムは、ポリビニルアルコールを含有する。
上記ポリビニルアルコールは、本発明の水溶性包装用フィルムの主たる構成成分となる。
上記親水性基としては、例えば、スルホン酸基、ピロリドン環基、アミノ基及びカルボキシル基からなる群より選択される少なくとも1種が好ましい。なかでも、スルホン酸基、ピロリドン環基がより好ましい。上記親水性基には、上述した官能基に加えて、ナトリウム、カリウム等の塩も含む。
なお、上記親水性基で変性された変性PVAとしては、上記ポリビニルアルコールと上記親水性基を有する他のモノマーとを共重合して得られるもののほか、上記ポリビニルアルコールに親水性基を付加することによって得られるものも含まれる。
上記式(4-2)中、R3は炭素数1~10のアルキレン基を表す。
上記ケン化度の調整方法は特に限定されない。ケン化度は、ケン化条件、すなわち加水分解条件により適宜調整可能である。
上記ケン化度分布標準偏差が上記下限以上及び上記上限以下であると、水溶性包装用フィルムの溶解性、薬品耐性がよくなり、薬剤を開放するときの溶解時間、及び薬品を包装した際の保存期間の双方をバランスよく高めることができる。
上記PVAのケン化度分布標準偏差のより好ましい下限は0.2モル%、より好ましい上限は0.9モル%である。
なお、上記ケン化度分布標準偏差は、PVA中のケン化度バラつきを示す指標であり、例えば、FT-IR等を用いて測定し、算出することができる。
なお、上記粘度はJIS K6726に準じて測定することができる。
上記ポリビニルアルコールの含有量が上記下限以上であると、水溶性包装用フィルムから可塑剤のブリードアウトがないより一層良好な品質の水溶性包装用フィルムとなることがある。上記ポリビニルアルコールの含有量が上記上限以下であると、水溶性包装用フィルムの強度がより一層高くなり、耐水性が得られることがある。
水溶性包装用フィルムは、高温多湿の地域や寒冷地でも運搬、貯蔵、使用がなされるため、高い引張強度や耐久性が要求される。特に低温での耐衝撃性が重視される。本発明の水溶性包装用フィルムは、可塑剤を含有することで、ガラス転移点を下げることが可能となり、低温での耐久性を向上させることができる。また、上記可塑剤を含有することで、水溶性包装用フィルムの水に対する溶解性を向上させることもできる。
上記可塑剤のなかでは、水溶性を向上させることができることから、グリセリン、トリメチロールプロパン、ポリエチレングリコール、ポリプロピレングリコール、トリエチレングリコール、ジプロピレングリコール、プロピレングリコールが好ましく、特に水溶性向上の効果が大きいことからグリセリン、トリメチロールプロパンが特に好ましい。
上記可塑剤含有量のより好ましい下限は3.2重量部、より好ましい上限は13重量部である。
本発明の水溶性包装用フィルムにおいて、上記澱粉は、水溶性包装用フィルムの表面及び裏面に塗布される。本発明の水溶性包装用フィルムは、上記澱粉が塗布されることにより、繰り出し性を向上させることができ、ブロッキング防止性や工程通過性に優れたものとすることができる。
なお、水溶性包装用フィルムの表面及び裏面とは、水溶性包装用フィルムの一方の主面及び反対側に位置する主面のことをいう。
また、本発明において、塗布するとは、塗布することの他、吹き付け、浸漬すること等も含み、表面及び裏面の少なくとも一部に澱粉が付着していることを意味する。
なお、上記塗布量とは、水溶性包装用フィルムの表面及び裏面に塗布された澱粉の量を意味する。
上記平均粒子径は、例えば、光学顕微鏡、電子顕微鏡、レーザー回析等の粒度分布計にて測定することができる。
上記配向性が上記下限以上及び上記上限以下であると、水溶性包装用フィルムの溶解性、パッケージング性がよくなり、内容物を開放するときの溶解時間、及び内容物を包装した際の保存性の双方をバランスよく高めることができる。
上記配向性のより好ましい下限は1.0nm、より好ましい上限は8.0nmである。
なお、上記配向性は水溶性包装用フィルム中のPVA分子配列の規則正しさを示す指標であり、例えば、位相差測定装置(KOBRA-WR、王子計測機器製)を用いることで測定することができる。
上記接触角は、JIS R 3257に準じて、測定することができる。例えば、接触角計Thetaライト(BiolinScientific社製)を用い、蒸留水を水溶性包装用フィルムに滴下することにより測定することができる。
本発明の水溶性包装用フィルムの製造方法としては、特に限定されないが、PVA、可塑剤及び水を含有するPVA水溶液を支持部材に流延し、乾燥した後、得られたPVAフィルムの表面及び裏面に澱粉を塗布する方法を用いることができる。具体的には、溶液流延法(キャスト法)、ロールコーティング法、スピンコーティング法、スクリーンコーティング法、ファウンテンコーティング法、ディッピング法及びスプレー法が挙げられる。
また、上記水の含有量は900重量部以下、好ましくは800重量部以下、より好ましくは700重量部以下である。上記水の含有量が上記下限以上であると、PVA水溶液の粘度が適度に低くなり、PVA水溶液の流延が容易になる。上記水の含有量が上記上限以下であると、PVA水溶液の粘度が適度に高くなり、PVA水溶液の流延が容易になり、乾燥時間がより一層短くなり、水溶性包装用フィルムの配向がより一層高められた、より一層良好な品質の水溶性包装用フィルムが得られる。
上記延伸工程としては、例えば、ロールを用いた延伸、テンターを用いた延伸、巻取装置を用いた延伸、乾燥収縮を利用した延伸、又は、これらを組み合わせた延伸等の方法が挙げられる。なお、上記延伸工程を行う場合、澱粉を塗布する工程は、延伸工程後に行うことが好ましい。
また、上記延伸工程における延伸倍率としては、1.01~3倍が好ましい。また、1.1~2倍がより好ましい。
上記水溶性包装用フィルムの用途としては、例えば、洗剤、農薬、医薬品等の薬剤包装用等に用いられる薬剤包装用フィルム等が挙げられる。
ポリビニルアルコールとして上記式(1)で表される構成単位を有するピロリドン環変性ポリビニルアルコール(重合度1000、ケン化度95.8モル%、ケン化度分布標準偏差0.21モル%、ピロリドン環変性量4モル%、4重量%水溶液粘度10mPa・s)90重量部、可塑剤として、グリセリン(和光純薬工業社製)5.0重量部、トリメチロールプロパン(和光純薬工業社製)5.0重量部、を水667重量部に溶解させて15重量%の水溶液を作製した。
(ケン化度分布標準偏差測定)
上記ポリビニルアルコールの粒子から、40個をランダムに選別し、ATR仕様のFT-IR(島津製作所製、IRAffinity-1)を用いて、ポリビニルアルコールのアセチル基量を測定した。得られたアセチル基量から、各粒子のケン化度を求め、ケン化度のバラつきから、ケン化度分布の標準偏差(σ)を算出した。
ポリビニルアルコールとして未変性のポリビニルアルコール(重合度1300、ケン化度88.0モル%、ケン化度分布標準偏差0.73モル%、4重量%水溶液粘度14mPa・s)95.1重量部、可塑剤として、グリセリン(和光純薬工業社製)3.3重量部、トリメチロールプロパン(和光純薬工業社製)0.8重量部、ポリエチレングリコール600(和光純薬工業社製)0.8重量部を水667重量部に溶解させて15重量%の水溶液を作製した。
実施例1と同様にしてPVAフィルム(厚さ50μm)が積層された積層フィルムの巻回体を得た。得られた積層フィルムの巻回体から、支持体を剥離してPVAフィルムを得た後、実施例1と同様にして、平均粒径20μmのトウモロコシ由来の澱粉(和光純薬工業社製)の塗布量が水溶性包装用フィルム1m2当たり0.005gになるようにPVAフィルムの表面及び裏面に澱粉を塗布し、水溶性包装用フィルムを得た。
ポリビニルアルコールとして上記式(2)で表される構成単位を有し、上記式(2)中、R1が2-メチレンプロピレン基であるスルホン酸ナトリウム変性ポリビニルアルコールA(重合度1200、ケン化度95.4モル%、ケン化度分布標準偏差0.31モル%、スルホン酸基変性量4モル%、4重量%水溶液粘度12mPa・s)を用い、平均粒径20μmのトウモロコシ由来の澱粉の塗布量を水溶性包装用フィルム1m2当たり0.01gとした以外は、実施例1と同様にして水溶性包装用フィルムを得た。
ポリビニルアルコールとして上記式(3)で表される構成単位を有し、上記式(3)中、R2が単結合であるアミノ基変性ポリビニルアルコール(重合度600、ケン化度91.8モル%、ケン化度分布標準偏差0.28モル%、アミノ基変性量8モル%、4重量%水溶液粘度6mPa・s)を用い、平均粒径10μmのサツマイモ由来の澱粉(和光純薬工業社製)の塗布量を水溶性包装用フィルム1m2当たり0.01gとした以外は、実施例1と同様にして水溶性包装用フィルムを得た。
ポリビニルアルコールとして上記式(4-2)で表される構成単位を有し、上記式(4-2)中、R3がメチレン基、X2及びX3がナトリウム原子であるカルボン酸変性ポリビニルアルコール(クラレ社製、KL-118、重合度1700、ケン化度97.5モル%、ケン化度分布標準偏差0.25モル%、カルボキシル基変性量1.5モル%、4重量%水溶液粘度30mPa・s)を用い、平均粒径30μmのサツマイモ由来の澱粉(和光純薬工業社製)の塗布量を水溶性包装用フィルム1m2当たり0.01gとした以外は、実施例1と同様にして水溶性包装用フィルムを得た。
ポリビニルアルコールとして上記式(2)で表される構成単位を有し、スルホン酸ナトリウム変性ポリビニルアルコールB(重合度1200、R1=CH2、ケン化度90.1モル%、ケン化度分布標準偏差0.98モル%、スルホン酸基変性量4モル%、4重量%水溶液粘度10mPa・s)を用い、平均粒径30μmのサツマイモ由来の澱粉(和光純薬工業社製)の塗布量を水溶性包装用フィルム1m2当たり0.01gとした以外は、実施例1と同様にして水溶性包装用フィルムを得た。
未変性のポリビニルアルコール(重合度1300、ケン化度98.0モル%、ケン化度分布標準偏差0.18モル%、4重量%水溶液粘度17mPa・s)90重量部、可塑剤として、グリセリン(和光純薬工業社製)5.0重量部、トリメチロールプロパン(和光純薬工業社製)5.0重量部、を水667重量部に溶解させて15重量%の水溶液を作製した。
得られたPVA水溶液を支持部材であるポリエチレンテレフタレート(PET)フィルム(厚さ50μm)上に、オートフィルムアプリケーター(テスター産業社製「PI-1210」)を用いて塗布し、80℃で20分間乾燥させ、支持部材上にPVAフィルム(厚さ50μm)を得た。
得られたPVAフィルムから支持体を剥離して水溶性包装用フィルムを得た後、温度23℃、相対湿度50%RHの環境下に24時間暴露した。
その後、水溶性包装用フィルムを600mm×500mmのサイズにカットして、幅が500mmになるように引張試験器にセットした。その後、幅が900mmになるまで延伸(延伸倍率:1.8倍)した後、平均粒径30μmのサツマイモ由来の澱粉(和光純薬工業社製)の塗布量が水溶性包装用フィルム1m2当たり0.01gになるようにPVAフィルムの表面及び裏面に澱粉を塗布し、水溶性包装用フィルムを得た。
平均粒径20μmのサツマイモ由来の澱粉(和光純薬工業社製)を平均粒径20μmのタピオカ由来の澱粉(GABAN社製)とした以外は、実施例1と同様にして水溶性包装用フィルムを得た。
ポリビニルアルコールとして上記式(2)で表される構成単位を有し、上記式(2)中、R1が2-メチレンプロピレン基であるスルホン酸ナトリウム変性ポリビニルアルコール(重合度1200、ケン化度95.4モル%、ケン化度分布標準偏差0.31モル%、スルホン酸基変性量4モル%、4重量%水溶液粘度12mPa・s)91重量部、可塑剤として、グリセリン(和光純薬工業社製)4.0重量部、トリメチロールプロパン(和光純薬工業社製)3.0重量部、ポリエチレングリコール600(和光純薬工業社製)2.0重量部を水667重量部に溶解させて15重量%の水溶液を作製した。
実施例1と同様にしてPVAフィルム(厚さ50μm)が積層された積層フィルムの巻回体を得た。得られた積層フィルムの巻回体から、支持体を剥離して水溶性包装用フィルムを得た。
平均粒径20μmのトウモロコシ由来の澱粉の塗布量を水溶性包装用フィルム1m2当たり0.11gとした以外は実施例2と同様にして水溶性包装用フィルムを得た。
平均粒径30μmのサツマイモ由来の澱粉の塗布量を水溶性包装用フィルム1m2当たり0.0001gとした以外は実施例2と同様にして水溶性包装用フィルムを得た。
比較例1で得られた水溶性包装用フィルムに格子状にエンボス加工を施し、凹部厚さが17μm、凹部と凸部との高さの差が30μm、溝幅0.1mm、溝間0.1mmである水溶性包装用フィルムを得た。
ポリビニルアルコールとして未変性のポリビニルアルコール(重合度1300、ケン化度88.0モル%、ケン化度分布標準偏差0.73モル%、4重量%水溶液粘度14mPa・s)95.05重量部、可塑剤として、グリセリン(和光純薬工業社製)3.3重量部、トリメチロールプロパン(和光純薬工業社製)0.8重量部、ポリエチレングリコール600(和光純薬工業社製)0.8重量部、平均粒径20μmのトウモロコシ由来の澱粉(和光純薬工業社製)0.05重量部を水667重量部に溶解させて15重量%の水溶液を作製した。
実施例1と同様にしてPVAフィルム(厚さ50μm)が積層された積層フィルムの巻回体を得た。得られた積層フィルムの巻回体から、支持体を剥離して水溶性包装用フィルムを得た。なお、得られた水溶性包装用フィルムは、澱粉をフィルムの内部に有するものであり、フィルム表面への澱粉の析出は無かった。
ポリビニルアルコールとして未変性のポリビニルアルコール(重合度1300、ケン化度88.0モル%、ケン化度分布標準偏差0.73モル%、4重量%水溶液粘度14mPa・s)100重量部を水667重量部に溶解させて15重量%の水溶液を作製した。
実施例1と同様にしてPVAフィルム(厚さ50μm)が積層された積層フィルムの巻回体を得た。得られた積層フィルムの巻回体から、支持体を剥離して水溶性包装用フィルムを得た。
平均粒径20μmのサツマイモ由来の澱粉の塗布量を水溶性包装用フィルム1m2当たり0.01gとなるようにPVAフィルムの表面及び裏面に澱粉を塗布した以外は比較例6と同様にして水溶性包装用フィルムを得た。
平均粒径20μmのサツマイモ由来の澱粉(和光純薬工業社製)を平均粒径20μmの松脂(MIKASA社製)とした以外は、実施例1と同様にして水溶性包装用フィルムを得た。
実施例及び比較例で得られたフィルムについて以下の評価を行った。結果を表2に示した。
得られた水溶性包装用フィルムを10mm×30mmのサイズにカットした。次に、50℃の環境下に24時間暴露した後、ガラス基板に貼付した。その後、BiolinScientific社製の接触角計Thetaライトを用いて、液滴法により、23℃、50%RHの雰囲気下で、約5.0μLの蒸留水を水溶性包装用フィルムの上に滴下して、液滴の滴下から1秒後の水溶性包装用フィルムと液滴の接線とからなる角度を測定した。
得られた水溶性包装用フィルムの50mm幅の巻回体をJIS Z0237に準拠し、巻き戻し速度20m/分の速度で50m巻き戻した時の金属ロールを指でなぞり、水溶性包装用フィルムに塗布した澱粉が付着しているかを目視確認し、以下の基準により評価した。
なお、比較例1で得られた水溶性包装用フィルムは、搬送中に金属ロールに水溶性包装用フィルムが貼り付いたため、ロール汚染性を測定することができなかった。
○○:澱粉の付着を観察できない
○:僅かに澱粉の付着を観察
×:指が真っ白になるほど澱粉が付着している、又は、測定不可
得られた水溶性包装用フィルムを80mm×200mmのサイズにカットした。JIS K7125に準拠して水溶性包装用フィルム同士の摩擦係数を測定し、以下の基準により評価した。
○○:0.8未満
○:0.8以上1.8未満
×:1.8以上
得られた水溶性包装用フィルムについて、ヘイズメーター(東京電色社製、TC-H3DPK)を用いて20℃でのヘイズを測定し、以下の基準により評価した。
○:ヘイズが3.5%未満
×:ヘイズが3.5%以上
得られた水溶性包装用フィルムから5cm×4cmの袋を作製し、該袋でトリクロロイソシアヌル酸ナトリウム20gを実包し、更にアルミ袋に入れ密封した後、温度40℃、湿度70%RHの恒温恒湿オーブンに1ヶ月間放置した。その後、フィルム部分を切り取り、ヘイズメーター(東京電色社製、TC-H3DPK)を用いて20℃でのヘイズを測定し、以下の基準により評価した。
○:ヘイズが3.5%未満
×:ヘイズが3.5%以上
得られた積層フィルムから支持体を剥離して水溶性包装用フィルムを得た後、温度23℃、相対湿度50%RHの環境下に24時間暴露した。
その後、水溶性包装用フィルムを35mm×40mmのサイズにカットして治具に固定し、500mlビーカーに水(500ml)を入れてスターラーにより撹拌(400mlの印に渦巻の下が到達)しながら、水温を23℃に保ちつつ、治具に固定したフィルムをかかる水中に浸漬した。治具からフィルムの残査が視認できなくなった時間を測定し、以下の基準により評価した。
○○:20秒未満
○:20秒以上30秒未満
×:30秒以上
得られた積層フィルムから支持体を剥離して水溶性包装用フィルムを得た後、アルミラミジップに入れて、温度23℃、相対湿度50%RHの環境下に1ヶ月間放置した。
その後、水溶性包装用フィルムを取り出して、1ヶ月間の放置前後のフィルムから可塑剤や添加剤等のブリード性を目視確認し、以下の基準により評価した。
○:フィルム外観が透明でブリードなし
×:フィルム外観が白濁してブリードあり
得られた水溶性包装用フィルムを温度23℃、相対湿度50%RHの環境下に24時間暴露した。その後、水溶性包装用フィルムを位相差測定装置(KOBRA-WR、王子計測機器社製)にて、入射角45°で測定し、測定した位相差Rの数値を配向性の値とした。
得られた水溶性包装用フィルムから5cm×4cmの袋を作製し、該袋で粉末洗剤20gを150℃のヒートシールで100個実包し、温度40℃、湿度70%RHの恒温恒湿オーブンに1ヶ月間放置した。その後、水溶性包装用フィルムのシール状況を目視観察した。
○:シール不良1個以下
×:2個以上シール不良あり
得られた水溶性包装用フィルムを5cm×4cmの袋を作製し、該袋でトリクロロイソシアヌル酸ナトリウム20gを実包し、更にアルミ袋に入れ密封した後、温度40℃、湿度70%RHの恒温恒湿オーブンに1ヶ月間放置した。その後、水溶性包装用フィルムの外観を目視観察した。
○:外観変化なし
×:外観が黄変又は茶変の着色あり
Claims (5)
- ポリビニルアルコール、可塑剤及び澱粉を含有し、
前記澱粉は、水溶性包装用フィルムの表面及び裏面に塗布されており、
前記澱粉の塗布量は、前記水溶性包装用フィルム1m2当たり0.001~0.10gであり、
液滴法により蒸留水を用いて測定した接触角が20~50°である
ことを特徴とする水溶性包装用フィルム。 - ポリビニルアルコールは、ケン化度が80~99.9モル%であることを特徴とする請求項1記載の水溶性包装用フィルム。
- ポリビニルアルコールは、ケン化度分布標準偏差(σ)が0.1~1.0モル%であることを特徴とする1又は2記載の水溶性包装用フィルム。
- ポリビニルアルコールは、4重量%水溶液として、20℃で測定した粘度が5~35mPa・sであることを特徴とする請求項1、2又は3記載の水溶性包装用フィルム。
- 配向性が0.5~9.9nmであることを特徴とする請求項1、2、3又は4記載の水溶性包装用フィルム。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15842220.4A EP3196015A4 (en) | 2014-09-17 | 2015-08-27 | Water-soluble packaging film |
| CN201580004936.4A CN105916685A (zh) | 2014-09-17 | 2015-08-27 | 水溶性包装用膜 |
| US15/509,587 US10351685B2 (en) | 2014-09-17 | 2015-08-27 | Water-soluble packaging film |
| JP2015544228A JP6546532B2 (ja) | 2014-09-17 | 2015-08-27 | 水溶性包装用フィルム |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-189014 | 2014-09-17 | ||
| JP2014189014 | 2014-09-17 | ||
| JP2015032186 | 2015-02-20 | ||
| JP2015-032186 | 2015-02-20 | ||
| JP2015-034360 | 2015-02-24 | ||
| JP2015034360 | 2015-02-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016043009A1 true WO2016043009A1 (ja) | 2016-03-24 |
Family
ID=55533050
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/074217 WO2016043009A1 (ja) | 2014-09-17 | 2015-08-27 | 水溶性包装用フィルム |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US10351685B2 (ja) |
| EP (1) | EP3196015A4 (ja) |
| JP (1) | JP6546532B2 (ja) |
| CN (1) | CN105916685A (ja) |
| TW (1) | TWI664125B (ja) |
| WO (1) | WO2016043009A1 (ja) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019006423A (ja) * | 2017-06-21 | 2019-01-17 | 日本合成化学工業株式会社 | 薬剤包装体及び薬剤包装体の製造方法 |
| JP2019521919A (ja) * | 2016-06-13 | 2019-08-08 | モノソル リミテッド ライアビリティ カンパニー | 異なるフィルムの組み合わせから作製された水溶性単位用量物品 |
| JP2019525874A (ja) * | 2016-06-13 | 2019-09-12 | モノソル リミテッド ライアビリティ カンパニー | 異なるフィルムの組み合わせから作製された水溶性単位用量物品 |
| WO2023054721A1 (ja) * | 2021-10-01 | 2023-04-06 | 株式会社クラレ | 水溶性フィルム、製造方法及び包装体 |
| WO2023054720A1 (ja) * | 2021-10-01 | 2023-04-06 | 株式会社クラレ | 水溶性フィルム、製造方法及び包装体 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019044751A1 (ja) * | 2017-08-30 | 2019-03-07 | 積水化学工業株式会社 | 水溶性包装用フィルム |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS51134766A (en) * | 1975-05-16 | 1976-11-22 | Kuraray Co | Surface treated polyvinyl alcohol film |
| JPS51138771A (en) * | 1975-05-28 | 1976-11-30 | Kuraray Co | Method of surface treatment of polyvinyl alcohol film |
| JPS51138772A (en) * | 1975-05-28 | 1976-11-30 | Kuraray Co | Method of modification of surface property of polyvinyl alcohol film |
| JPS51139868A (en) * | 1975-05-28 | 1976-12-02 | Kuraray Co | Method of improvement of surface property of polyvinyl alcohol film |
| JPH07501351A (ja) * | 1991-08-12 | 1995-02-09 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | 分解可能な、撥液コーティングされた物品 |
| WO2001023460A1 (en) * | 1999-09-30 | 2001-04-05 | Kao Corporation | Film and process for producing film |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4514858B2 (ja) | 1999-10-08 | 2010-07-28 | 株式会社クラレ | 樹脂組成物および水溶性フィルム |
| GB0023713D0 (en) | 2000-09-27 | 2000-11-08 | Unilever Plc | A water soluble package |
| DE60201142T2 (de) | 2001-04-20 | 2005-10-20 | Kuraray Co., Ltd., Kurashiki | Wasserlösliche Folie und Verpackung, welche dieselbe verwendet |
| EP1314653B1 (en) * | 2001-11-23 | 2004-12-08 | The Procter & Gamble Company | Water-soluble pouch |
| JP4433446B2 (ja) | 2001-12-10 | 2010-03-17 | 日本合成化学工業株式会社 | 熱履歴を受けても水溶性に優れた樹脂組成物およびその用途 |
| JP4630396B2 (ja) | 2003-12-16 | 2011-02-09 | 日本合成化学工業株式会社 | 水溶性フィルム |
| US7754318B2 (en) * | 2005-06-16 | 2010-07-13 | The Nippon Synthetic Chemical Industry Co., Ltd. | Water-soluble film |
| EP1900488B1 (en) * | 2005-07-04 | 2014-06-25 | Denki Kagaku Kogyo Kabushiki Kaisha | METHOD FOR PRODUCING A CERAMIC SUBSTRATE IN Si3N4 |
| KR101515653B1 (ko) * | 2007-05-14 | 2015-04-27 | 가부시키가이샤 구라레 | 수용성 폴리비닐알코올계 필름의 롤 형상물 및 그 보관 방법 |
| JP5754584B2 (ja) | 2010-05-27 | 2015-07-29 | 荒川化学工業株式会社 | 製紙用表面サイズ剤、表面サイズ塗工液および紙 |
| EP2679637B1 (en) | 2011-02-23 | 2017-05-10 | Dainichiseika Color & Chemicals Mfg. Co., Ltd. | Aqueous liquid composition, aqueous coating, functional coating film, and composite material |
| US9394092B2 (en) * | 2012-04-16 | 2016-07-19 | Monosol, Llc | Powdered pouch and method of making same |
| TW201420344A (zh) | 2012-09-21 | 2014-06-01 | Nippon Synthetic Chem Ind | 聚乙烯醇系膜及其製造方法、以及使用聚乙烯醇系膜之熱轉印用積層體及使用其之熱轉印方法 |
-
2015
- 2015-08-27 CN CN201580004936.4A patent/CN105916685A/zh active Pending
- 2015-08-27 JP JP2015544228A patent/JP6546532B2/ja active Active
- 2015-08-27 WO PCT/JP2015/074217 patent/WO2016043009A1/ja active Application Filing
- 2015-08-27 US US15/509,587 patent/US10351685B2/en active Active
- 2015-08-27 EP EP15842220.4A patent/EP3196015A4/en active Pending
- 2015-08-31 TW TW104128565A patent/TWI664125B/zh active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS51134766A (en) * | 1975-05-16 | 1976-11-22 | Kuraray Co | Surface treated polyvinyl alcohol film |
| JPS51138771A (en) * | 1975-05-28 | 1976-11-30 | Kuraray Co | Method of surface treatment of polyvinyl alcohol film |
| JPS51138772A (en) * | 1975-05-28 | 1976-11-30 | Kuraray Co | Method of modification of surface property of polyvinyl alcohol film |
| JPS51139868A (en) * | 1975-05-28 | 1976-12-02 | Kuraray Co | Method of improvement of surface property of polyvinyl alcohol film |
| JPH07501351A (ja) * | 1991-08-12 | 1995-02-09 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | 分解可能な、撥液コーティングされた物品 |
| WO2001023460A1 (en) * | 1999-09-30 | 2001-04-05 | Kao Corporation | Film and process for producing film |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3196015A4 * |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019521919A (ja) * | 2016-06-13 | 2019-08-08 | モノソル リミテッド ライアビリティ カンパニー | 異なるフィルムの組み合わせから作製された水溶性単位用量物品 |
| JP2019525874A (ja) * | 2016-06-13 | 2019-09-12 | モノソル リミテッド ライアビリティ カンパニー | 異なるフィルムの組み合わせから作製された水溶性単位用量物品 |
| JP7133475B2 (ja) | 2016-06-13 | 2022-09-08 | モノソル リミテッド ライアビリティ カンパニー | 異なるフィルムの組み合わせから作製された水溶性単位用量物品 |
| JP7178345B2 (ja) | 2016-06-13 | 2022-11-25 | モノソル リミテッド ライアビリティ カンパニー | 異なるフィルムの組み合わせから作製された水溶性単位用量物品 |
| JP2019006423A (ja) * | 2017-06-21 | 2019-01-17 | 日本合成化学工業株式会社 | 薬剤包装体及び薬剤包装体の製造方法 |
| JPWO2023054721A1 (ja) * | 2021-10-01 | 2023-04-06 | ||
| WO2023054721A1 (ja) * | 2021-10-01 | 2023-04-06 | 株式会社クラレ | 水溶性フィルム、製造方法及び包装体 |
| WO2023054720A1 (ja) * | 2021-10-01 | 2023-04-06 | 株式会社クラレ | 水溶性フィルム、製造方法及び包装体 |
| CN116368076A (zh) * | 2021-10-01 | 2023-06-30 | 株式会社可乐丽 | 水溶性膜、制造方法和包装体 |
| JP7314438B1 (ja) * | 2021-10-01 | 2023-07-25 | 株式会社クラレ | 水溶性フィルム、製造方法及び包装体 |
| JP7343732B2 (ja) | 2021-10-01 | 2023-09-12 | 株式会社クラレ | 水溶性フィルム、製造方法及び包装体 |
| US11939442B2 (en) | 2021-10-01 | 2024-03-26 | Kuraray Co., Ltd. | Water-soluble film, manufacturing method, and package |
| US11939441B2 (en) | 2021-10-01 | 2024-03-26 | Kuraray Co., Ltd. | Water-soluble film, manufacturing method, and package |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201615516A (zh) | 2016-05-01 |
| JP6546532B2 (ja) | 2019-07-17 |
| US10351685B2 (en) | 2019-07-16 |
| US20170253708A1 (en) | 2017-09-07 |
| CN105916685A (zh) | 2016-08-31 |
| TWI664125B (zh) | 2019-07-01 |
| EP3196015A1 (en) | 2017-07-26 |
| EP3196015A4 (en) | 2018-03-28 |
| JPWO2016043009A1 (ja) | 2017-06-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6546532B2 (ja) | 水溶性包装用フィルム | |
| US11840590B2 (en) | Polyvinyl alcohol film | |
| US9868574B2 (en) | Water-soluble packaging film | |
| JP6089149B1 (ja) | 薬剤包装用フィルム | |
| JP2017078166A (ja) | 水溶性フィルム及び薬剤包装体 | |
| CN111032312B (zh) | 水溶性包装用膜 | |
| JP2017052897A (ja) | 水溶性包装用フィルム | |
| JPWO2017043511A1 (ja) | 水溶性フィルム及び薬剤包装体 | |
| JP2017115128A (ja) | 水溶性フィルム及び薬剤包装体 | |
| JP2024032867A (ja) | 水溶性包装用フィルム | |
| JPWO2017043514A1 (ja) | フィルムロール及び薬剤包装体 | |
| JP6397323B2 (ja) | 多層薬剤包装用フィルム | |
| US10450118B2 (en) | Chemical product packaging film | |
| JP2018184500A (ja) | 水溶性包装用フィルム | |
| JP2017106008A (ja) | 水溶性フィルム及び薬剤包装体 | |
| JP2017114568A (ja) | 液体洗剤包装用水溶性フィルム及び液体洗剤包装体 | |
| JP7339721B2 (ja) | 水溶性包装用フィルム | |
| JP5653680B2 (ja) | 表面処理されたポリビニルアルコール系重合体フィルム | |
| JP2016156013A (ja) | 薬剤包装用フィルム | |
| JPWO2017043507A1 (ja) | 水溶性フィルムおよびそれを用いた薬剤包装体、並びに水溶性フィルムの製造方法 | |
| JP2018070169A (ja) | 水溶性包装用フィルム | |
| JP2000263712A (ja) | 多層構造体 | |
| JP2021143296A (ja) | ポリビニルアルコールフィルム、及びパッケージ材料 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2015544228 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15842220 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15509587 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015842220 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015842220 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |