WO2014175355A1 - 非水電解質二次電池 - Google Patents
非水電解質二次電池 Download PDFInfo
- Publication number
- WO2014175355A1 WO2014175355A1 PCT/JP2014/061483 JP2014061483W WO2014175355A1 WO 2014175355 A1 WO2014175355 A1 WO 2014175355A1 JP 2014061483 W JP2014061483 W JP 2014061483W WO 2014175355 A1 WO2014175355 A1 WO 2014175355A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- active material
- electrode active
- positive electrode
- composite oxide
- lithium
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings, jackets or wrappings of a single cell or a single battery
- H01M50/116—Primary casings, jackets or wrappings of a single cell or a single battery characterised by the material
- H01M50/117—Inorganic material
- H01M50/119—Metals
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/409—Separators, membranes or diaphragms characterised by the material
- H01M50/449—Separators, membranes or diaphragms characterised by the material having a layered structure
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/028—Positive electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2220/00—Batteries for particular applications
- H01M2220/20—Batteries in motive systems, e.g. vehicle, ship, plane
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings, jackets or wrappings of a single cell or a single battery
- H01M50/116—Primary casings, jackets or wrappings of a single cell or a single battery characterised by the material
- H01M50/121—Organic material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings, jackets or wrappings of a single cell or a single battery
- H01M50/116—Primary casings, jackets or wrappings of a single cell or a single battery characterised by the material
- H01M50/124—Primary casings, jackets or wrappings of a single cell or a single battery characterised by the material having a layered structure
- H01M50/126—Primary casings, jackets or wrappings of a single cell or a single battery characterised by the material having a layered structure comprising three or more layers
- H01M50/129—Primary casings, jackets or wrappings of a single cell or a single battery characterised by the material having a layered structure comprising three or more layers with two or more layers of only organic material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/489—Separators, membranes, diaphragms or spacing elements inside the cells, characterised by their physical properties, e.g. swelling degree, hydrophilicity or shut down properties
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02T—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO TRANSPORTATION
- Y02T10/00—Road transport of goods or passengers
- Y02T10/60—Other road transportation technologies with climate change mitigation effect
- Y02T10/70—Energy storage systems for electromobility, e.g. batteries
Definitions
- the present invention relates to a non-aqueous electrolyte secondary battery.
- a nonaqueous electrolyte secondary battery generally includes a positive electrode obtained by applying a positive electrode active material or the like to a current collector, and a negative electrode obtained by applying a negative electrode active material or the like to a current collector. It has the structure connected through the electrolyte layer holding electrolyte gel. Then, when ions such as lithium ions are occluded / released in the electrode active material, a charge / discharge reaction of the battery occurs.
- non-aqueous electrolyte secondary batteries with a low environmental load are being used not only for portable devices, but also for power supply devices for electric vehicles such as hybrid vehicles (HEV), electric vehicles (EV), and fuel cell vehicles. .
- HEV hybrid vehicles
- EV electric vehicles
- fuel cell vehicles fuel cell vehicles.
- Non-aqueous electrolyte secondary batteries intended for application to electric vehicles are required to have high output and high capacity.
- a positive electrode active material used for a positive electrode of a non-aqueous electrolyte secondary battery for an electric vehicle a lithium-cobalt composite oxide, which is a layered composite oxide, can obtain a high voltage of 4V and has a high energy density.
- cobalt which is a raw material
- there is anxiety in terms of supply of raw materials considering the possibility that demand will increase significantly in the future.
- the price of cobalt raw materials may rise. Therefore, a composite oxide having a low cobalt content is desired.
- Spinel-based lithium manganese composite oxide (LiMn 2 O 4 ) has a spinel structure and functions as a 4V-class positive electrode material between the composition and ⁇ -MnO 2 . Since the spinel-type lithium manganese composite oxide has a three-dimensional host structure different from the layered structure such as that of LiCoO 2 or the like, most of the theoretical capacity can be used and is expected to have excellent cycle characteristics. .
- lithium ion secondary batteries using spinel-based lithium manganese composite oxide as the positive electrode material cannot avoid the capacity degradation that gradually decreases in capacity due to repeated charge and discharge. There was a big problem left.
- the present invention provides a non-aqueous electrolyte secondary battery including a spinel-based lithium manganese composite oxide as a positive electrode active material and encapsulating a power generation element inside an outer package made of a laminate film. It aims at providing the means which can be improved.
- a spinel lithium manganese composite oxide is used in combination with a lithium nickel composite oxide, and the mixing ratio of the lithium nickel composite oxide is within a specific range.
- a positive electrode in which a positive electrode active material layer containing a positive electrode active material is formed on the surface of a positive electrode current collector, and a negative electrode active material containing a negative electrode active material on the surface of the negative electrode current collector
- a nonaqueous electrolyte secondary battery in which a power generation element including a negative electrode formed with a layer and a separator is enclosed in an exterior body made of a laminate film.
- the positive electrode active material includes a spinel manganese positive electrode active material and a lithium nickel composite oxide, and the spinel manganese positive electrode active material and the lithium nickel composite oxide
- the mixing ratio of the lithium nickel composite oxide with respect to the total of 100% by weight is 30 to 70% by weight.
- FIG. 1 is a schematic cross-sectional view showing a basic configuration of a non-aqueous electrolyte lithium ion secondary battery that is not a flat (stacked) bipolar type, which is an embodiment of a non-aqueous electrolyte secondary battery. It is a perspective view showing the appearance of a flat lithium ion secondary battery which is a typical embodiment of a nonaqueous electrolyte secondary battery.
- a positive electrode in which a positive electrode active material layer containing a positive electrode active material is formed on the surface of a positive electrode current collector, and a negative electrode active material layer containing a negative electrode active material on the surface of the negative electrode current collector are provided.
- a power generation element including a formed negative electrode and a separator is enclosed in an exterior body made of a laminate film, and the positive electrode active material includes a spinel-based manganese positive electrode active material, a lithium nickel-based composite oxide, A non-aqueous electrolyte secondary battery in which the mixing ratio of the lithium nickel composite oxide is 30 to 70% by weight with respect to a total of 100% by weight of the spinel manganese positive electrode active material and the lithium nickel composite oxide Provided.
- a mixture of a spinel-based lithium manganese composite oxide and a lithium-nickel composite oxide is mixed in a positive electrode active material layer by mixing a predetermined amount of lithium-nickel composite oxide. Potential is generated. For this reason, microscopic lithium ion desorption / insertion occurs between these active materials during discharge, thereby improving the lithium ion conductivity in the electrolyte contained in the power generation element.
- output characteristics under low temperature conditions are improved. It becomes possible.
- FIG. 1 is a schematic cross-sectional view schematically showing an outline of a stacked battery as an embodiment of the battery of the present invention.
- the flat type (stacked type) lithium ion secondary battery shown in FIG. 1 will be described in detail as an example, but the technical scope of the present invention is only such a form. Not limited to.
- FIG. 1 is a schematic cross-sectional view schematically showing a basic configuration of a non-aqueous electrolyte lithium ion secondary battery (hereinafter also simply referred to as “stacked battery”) that is not a flat type (stacked type) bipolar type.
- the stacked battery 10 of the present embodiment has a structure in which a substantially rectangular power generation element 21 in which a charge / discharge reaction actually proceeds is sealed inside a battery exterior material 29 that is an exterior body.
- the power generation element 21 has a configuration in which a positive electrode, a separator 17, and a negative electrode are stacked.
- the separator 17 contains a nonaqueous electrolyte (for example, a liquid electrolyte).
- the positive electrode has a structure in which the positive electrode active material layers 15 are disposed on both surfaces of the positive electrode current collector 12.
- the negative electrode has a structure in which the negative electrode active material layer 13 is disposed on both surfaces of the negative electrode current collector 11.
- the negative electrode, the electrolyte layer, and the positive electrode are laminated in this order so that one positive electrode active material layer 15 and the negative electrode active material layer 13 adjacent thereto face each other with a separator 17 therebetween.
- the adjacent positive electrode, electrolyte layer, and negative electrode constitute one unit cell layer 19. Therefore, it can be said that the stacked battery 10 shown in FIG. 1 has a configuration in which a plurality of single battery layers 19 are stacked so that they are electrically connected in parallel.
- the negative electrode active material layer 13 is arrange
- the positive electrode current collector 12 and the negative electrode current collector 11 are each provided with a positive electrode current collector plate (tab) 27 and a negative electrode current collector plate (tab) 25 that are electrically connected to the respective electrodes (positive electrode and negative electrode). It has the structure led out of the battery exterior material 29 so that it may be pinched
- the positive electrode current collector 27 and the negative electrode current collector 25 are ultrasonically welded to the positive electrode current collector 12 and the negative electrode current collector 11 of each electrode, respectively, via a positive electrode lead and a negative electrode lead (not shown) as necessary. Or resistance welding or the like.
- FIG. 1 shows a flat battery (stacked battery) that is not a bipolar battery, but a positive electrode active material layer that is electrically coupled to one surface of the current collector and the opposite side of the current collector.
- a bipolar battery including a bipolar electrode having a negative electrode active material layer electrically coupled to the surface.
- one current collector also serves as a positive electrode current collector and a negative electrode current collector.
- the positive electrode has a positive electrode current collector and a positive electrode active material layer formed on the surface of the positive electrode current collector.
- a metal is preferably used.
- the metal include aluminum, nickel, iron, stainless steel, titanium, copper, and other alloys.
- a clad material of nickel and aluminum, a clad material of copper and aluminum, or a plating material of a combination of these metals can be preferably used.
- covered on the metal surface may be sufficient.
- aluminum, stainless steel, and copper are preferable from the viewpoints of electronic conductivity and battery operating potential.
- the size of the current collector is determined according to the intended use of the battery. For example, if it is used for a large battery that requires a high energy density, a current collector having a large area is used. There is no particular limitation on the thickness of the current collector.
- the thickness of the current collector is usually about 1 to 100 ⁇ m.
- the positive electrode active material layer includes a positive electrode active material.
- the positive electrode active material essentially includes a spinel-based lithium manganese composite oxide and a lithium-nickel-based composite oxide.
- the ratio of the total amount of spinel-based lithium manganese composite oxide and lithium-nickel-based composite oxide in the total amount of 100% by weight of the positive electrode active material contained in the positive electrode active material layer is preferably 50% by weight or more, and more It is preferably 70% by weight or more, more preferably 85% by weight or more, still more preferably 90% by weight or more, particularly preferably 95% by weight or more, and most preferably 100% by weight.
- the average particle size of the positive electrode active material is not particularly limited, but is preferably 6 to 11 ⁇ m, more preferably 7 to 10 ⁇ m in terms of secondary particle size from the viewpoint of increasing output.
- the average particle diameter of the primary particles is 0.4 to 0.65 ⁇ m, more preferably 0.45 to 0.55 ⁇ m.
- the “particle diameter” in the present specification means the maximum distance L among the distances between any two points on the particle outline.
- the average particle diameter the average particle diameter of particles observed in several to several tens of fields using an observation means such as a scanning electron microscope (SEM) or a transmission electron microscope (TEM). A value calculated as a value is adopted.
- Spinel-based lithium manganese composite oxide is a composite oxide that typically has a composition of LiMn 2 O 4 and has a spinel structure and essentially contains lithium and manganese.
- conventionally known knowledge such as JP-A-2000-77071 can be referred to as appropriate.
- lithium nickel complex oxide is not specifically limited as long as it is a complex oxide containing lithium and nickel.
- a typical example of a composite oxide containing lithium and nickel is lithium nickel composite oxide (LiNiO 2 ).
- a composite oxide in which some of the nickel atoms of the lithium nickel composite oxide are substituted with other metal atoms is more preferable.
- a preferable example is a lithium-nickel-manganese-cobalt composite oxide (hereinafter simply referred to as “NMC composite”).
- oxide (Also referred to as “oxide”) has a layered crystal structure in which lithium atomic layers and transition metals (Mn, Ni and Co are arranged in an orderly manner) atomic layers are alternately stacked via oxygen atomic layers.
- Li atom is contained per atom, and the amount of Li that can be taken out is twice that of the spinel-based lithium-manganese composite oxide, that is, the supply capacity is doubled, and a high capacity can be obtained.
- it since it has higher thermal stability than LiNiO 2 , it is particularly advantageous among lithium nickel-based composite oxides used as a positive electrode active material.
- the NMC composite oxide includes a composite oxide in which a part of the transition metal element is substituted with another metal element.
- Other elements in that case include Ti, Zr, Nb, W, P, Al, Mg, V, Ca, Sr, Cr, Fe, B, Ga, In, Si, Mo, Y, Sn, V, Cu , Ag, Zn, etc., preferably Ti, Zr, Nb, W, P, Al, Mg, V, Ca, Sr, Cr, more preferably Ti, Zr, P, Al, Mg, From the viewpoint of improving cycle characteristics, Ti, Zr, Al, Mg, and Cr are more preferable.
- a represents the atomic ratio of Li
- b represents the atomic ratio of Ni
- c represents the atomic ratio of Co
- d represents the atomic ratio of Mn
- x represents the atomic ratio of M. Represents. From the viewpoint of cycle characteristics, it is preferable that 0.4 ⁇ b ⁇ 0.6 in the general formula (1).
- the composition of each element can be measured by, for example, inductively coupled plasma (ICP) emission spectrometry.
- ICP inductively coupled plasma
- Ni nickel
- Co cobalt
- Mn manganese
- Ti or the like partially replaces the transition metal in the crystal lattice. From the viewpoint of cycle characteristics, it is preferable that a part of the transition element is substituted with another metal element, and it is particularly preferable that 0 ⁇ x ⁇ 0.3 in the general formula (1). Since at least one selected from the group consisting of Ti, Zr, Nb, W, P, Al, Mg, V, Ca, Sr, and Cr is dissolved, the crystal structure is stabilized. It is considered that the battery capacity can be prevented from decreasing even if the above is repeated, and that excellent cycle characteristics can be realized.
- b, c and d are composite oxides in which 0.44 ⁇ b ⁇ 0.51, 0.27 ⁇ c ⁇ 0.31, and 0.19 ⁇ d ⁇ 0.26
- the positive electrode active material is preferable from the viewpoint of improving the balance between capacity and life characteristics.
- the lithium nickel composite oxide such as NMC composite oxide can be prepared by selecting various known methods such as coprecipitation method and spray drying method.
- the coprecipitation method is preferably used because the complex oxide according to this embodiment is easy to prepare.
- a nickel-cobalt-manganese composite oxide is manufactured by a coprecipitation method as in the method described in JP2011-105588A, and then nickel-cobalt. It can be obtained by mixing and firing a manganese composite oxide and a lithium compound. This will be specifically described below.
- the raw material compound of the composite oxide for example, Ni compound, Mn compound and Co compound is dissolved in an appropriate solvent such as water so as to have a desired composition of the active material.
- the Ni compound, Mn compound, and Co compound include sulfates, nitrates, carbonates, acetates, oxalates, oxides, hydroxides, and halides of the metal elements.
- Specific examples of the Ni compound, Mn compound, and Co compound include, but are not limited to, nickel sulfate, cobalt sulfate, manganese sulfate, nickel acetate, cobalt acetate, and manganese acetate.
- Ti, Zr, Nb as a metal element that substitutes a part of the layered lithium metal composite oxide constituting the active material so as to have a desired active material composition.
- W, P, Al, Mg, V, Ca, Sr, and a compound containing at least one metal element such as Cr may be further mixed.
- the coprecipitation reaction can be performed by neutralization and precipitation reaction using the above raw material compound and an alkaline solution.
- the metal composite hydroxide and metal composite carbonate containing the metal contained in the said raw material compound are obtained.
- the alkaline solution for example, an aqueous solution of sodium hydroxide, potassium hydroxide, sodium carbonate, ammonia or the like can be used, but sodium hydroxide, sodium carbonate or a mixed solution thereof is preferably used for the neutralization reaction. .
- an aqueous ammonia solution or an ammonium salt is preferably used for the complex reaction.
- the addition amount of the alkaline solution used for the neutralization reaction may be an equivalent ratio of 1.0 with respect to the neutralized content of all the metal salts contained, but it is preferable to add the alkali excess together for pH adjustment.
- the addition amount of the aqueous ammonia solution or ammonium salt used for the complex reaction is preferably such that the ammonia concentration in the reaction solution is in the range of 0.01 to 2.00 mol / l.
- the pH of the reaction solution is preferably controlled in the range of 10.0 to 13.0.
- the reaction temperature is preferably 30 ° C. or higher, more preferably 30 to 60 ° C.
- the composite hydroxide obtained by the coprecipitation reaction is then preferably suction filtered, washed with water and dried.
- the particle size of the composite hydroxide can be controlled by adjusting the conditions (stirring time, alkali concentration, etc.) for carrying out the coprecipitation reaction, which is the secondary of the positive electrode active material finally obtained. Affects the average particle size of the particles.
- the nickel-cobalt-manganese composite hydroxide is mixed with a lithium compound and fired to obtain a lithium-nickel-manganese-cobalt composite oxide.
- the Li compound include lithium hydroxide or a hydrate thereof, lithium peroxide, lithium nitrate, and lithium carbonate.
- the firing treatment may be performed in one stage, but is preferably performed in two stages (temporary firing and main firing).
- a composite oxide can be obtained efficiently by two-stage firing.
- the pre-baking conditions are not particularly limited, and differ depending on the lithium raw material, so that it is difficult to uniquely define them.
- the temperature rising rate is preferably 1 to 20 ° C./min from room temperature.
- the atmosphere is preferably in air or in an oxygen atmosphere.
- the pre-baking temperature is preferably 500 to 900 ° C., more preferably 600 to 800 ° C., further preferably 650. ⁇ 750 ° C.
- the pre-baking time is preferably 0.5 to 10 hours, more preferably 4 to 6 hours.
- the conditions for the main firing are not particularly limited, but the rate of temperature rise is preferably from room temperature to 1 to 20 ° C./min.
- the atmosphere is preferably in air or in an oxygen atmosphere.
- the firing temperature is preferably 800 to 1200 ° C., more preferably 850 to 1100 ° C., and further preferably 900 to 1050. ° C.
- the pre-baking time is preferably 1 to 20 hours, more preferably 8 to 12 hours.
- a method of previously mixing with nickel, cobalt, manganate Any means such as a method of adding nickel, cobalt and manganate simultaneously, a method of adding to the reaction solution during the reaction, a method of adding to the nickel-cobalt-manganese composite oxide together with the Li compound may be used.
- the lithium nickel composite oxide can be produced by appropriately adjusting the reaction conditions such as pH of the reaction solution, reaction temperature, reaction concentration, addition rate, and stirring time.
- non-aqueous electrolyte secondary battery using a spinel-based lithium manganese composite oxide as a positive electrode active material according to the technique described in Japanese Patent Laid-Open No. 10-112318, output characteristics under low temperature conditions are disclosed. Found that may not always be sufficient. Such a decrease in output characteristics under low temperature conditions is due to a non-aqueous electrolyte secondary battery comprising a spinel-based lithium manganese composite oxide as a positive electrode active material, and a power generation element enclosed in an exterior body made of a laminate film. It has also been found that this can occur significantly.
- the present inventors searched for the cause of the decrease in output characteristics under the low temperature conditions as described above. As a result, it was found that an increase in internal resistance due to a decrease in lithium ion conductivity in the power generation element under low temperature conditions causes a decrease in output characteristics. And since the capacity
- a spinel lithium manganese composite oxide is used in combination with a lithium nickel composite oxide, and the mixing ratio of the lithium nickel composite oxide is within a specific range.
- the mixing ratio of the lithium nickel composite oxide is 30 to 70 wt% with respect to 100 wt% in total of the spinel manganese positive electrode active material and the lithium nickel composite oxide. It is characterized in that
- the mechanism that prevents the deterioration of the output characteristics under the low temperature condition by having the above-described features is estimated as follows. That is, the spinel lithium manganese composite oxide and the lithium nickel composite oxide have different charge / discharge profiles. More specifically, when a charge / discharge curve is drawn using each composite oxide alone, with the capacity (SOC) on the horizontal axis and the potential of the composite oxide on the vertical axis, the spinel-based lithium manganese composite oxide is Almost all SOC regions show a higher potential than the lithium nickel composite oxide. If the mixing ratio of the lithium nickel composite oxide when the total amount of the two types is 100% by weight is 70% by weight or less, the mixture of the two types of composite oxides can be used as the positive electrode active material.
- the upper limit value of the mixing ratio of the lithium nickel composite oxide with respect to the total of 100 wt% of the two positive electrode active materials is defined as 70 wt%. It is.
- the mixing ratio of the lithium nickel based composite oxide in the total of 100% by weight of the spinel based manganese positive electrode active material and the lithium nickel based composite oxide is low, and conversely, the mixing ratio of the spinel based lithium manganese composite oxide is Think about the case of increasing. Since the spinel-type lithium manganese oxide composite oxide has a relatively small capacity, if the capacity of the positive electrode active material layer is kept constant in such a case, the basis weight of the positive electrode active material layer increases. When the weight per unit area of the positive electrode active material layer increases, the decrease in lithium ion conductivity under low temperature conditions is prominent, preventing the increase in internal resistance under low temperature conditions and the resulting decrease in output characteristics. Can not do it.
- the lithium nickel composite oxide with respect to a total of 100% by weight of the two positive electrode active materials as a range in which the effect according to the present embodiment can be exhibited within a range in which the basis weight is not significantly increased.
- the lower limit of the mixing ratio is defined as 30% by weight.
- the mixing ratio of the lithium nickel composite oxide to the total of 100% by weight of the spinel manganese positive electrode active material and the lithium nickel composite oxide is preferably 30 to 80% by weight, more preferably 50 to 70% by weight. %.
- the mixing ratio of the spinel-based lithium manganese composite oxide to the total of 100% by weight of the spinel-based manganese positive electrode active material and the lithium nickel-based composite oxide is not particularly limited, but is preferably 20 to 70% by weight, and more preferably Is 30 to 50% by weight.
- the basis weight of the positive electrode active material layer is not particularly limited, but is preferably 18.5 to 23.5 mg / cm 2 from the viewpoint of sufficiently exerting the effects of the present invention. More preferably, it is 22.0 to 23.5 mg / cm 2 .
- the positive electrode active material layer is used to increase the conductivity, as necessary, a conductive additive, a binder, an electrolyte (polymer matrix, ion conductive polymer, electrolyte, etc.), and ion conductivity. It further includes other additives such as lithium salts.
- a material that can function as an active material in the positive electrode active material layer and the negative electrode active material layer described later is preferably 85 to 99.5% by weight.
- binder Although it does not specifically limit as a binder used for a positive electrode active material layer, for example, the following materials are mentioned. Polyethylene, polypropylene, polyethylene terephthalate (PET), polyether nitrile, polyacrylonitrile, polyimide, polyamide, cellulose, carboxymethyl cellulose (CMC) and its salts, ethylene-vinyl acetate copolymer, polyvinyl chloride, styrene-butadiene rubber (SBR) ), Isoprene rubber, butadiene rubber, ethylene / propylene rubber, ethylene / propylene / diene copolymer, styrene / butadiene / styrene block copolymer and hydrogenated product thereof, styrene / isoprene / styrene block copolymer and hydrogenated product thereof.
- Thermoplastic polymers such as products, polyvinylidene fluoride (P
- the amount of the binder contained in the positive electrode active material layer is not particularly limited as long as it is an amount capable of binding the active material, but preferably 0.5 to 15% by weight with respect to the active material layer. More preferably, it is 1 to 10% by weight.
- the positive electrode active material layer further contains other additives such as a conductive additive, an electrolyte (polymer matrix, ion conductive polymer, electrolytic solution, etc.), and a lithium salt for improving ion conductivity, as necessary.
- the conductive assistant means an additive blended to improve the conductivity of the positive electrode active material layer or the negative electrode active material layer.
- the conductive auxiliary agent include carbon materials such as carbon black such as ketjen black and acetylene black, graphite, and carbon fiber.
- electrolyte salt examples include Li (C 2 F 5 SO 2 ) 2 N, LiPF 6 , LiBF 4 , LiClO 4 , LiAsF 6 , LiCF 3 SO 3 and the like.
- Examples of the ion conductive polymer include polyethylene oxide (PEO) and polypropylene oxide (PPO) polymers.
- the compounding ratio of the components contained in the positive electrode active material layer and the negative electrode active material layer described later is not particularly limited.
- the blending ratio can be adjusted by appropriately referring to known knowledge about lithium ion secondary batteries.
- the thickness of each active material layer is not particularly limited, and conventionally known knowledge about the battery can be appropriately referred to. As an example, the thickness of each active material layer is about 2 to 100 ⁇ m.
- the negative electrode active material layer contains an active material, and other additives such as a conductive additive, a binder, an electrolyte (polymer matrix, ion conductive polymer, electrolyte, etc.), and a lithium salt to enhance ionic conductivity as necessary.
- a conductive additive such as a conductive additive, a binder, an electrolyte (polymer matrix, ion conductive polymer, electrolyte, etc.), and a lithium salt to enhance ionic conductivity as necessary.
- An agent is further included.
- Other additives such as conductive assistants, binders, electrolytes (polymer matrix, ion conductive polymers, electrolytes, etc.) and lithium salts for improving ion conductivity are those described in the above positive electrode active material layer column. It is the same.
- the negative electrode active material examples include carbon materials such as graphite (graphite), soft carbon, and hard carbon, lithium-transition metal composite oxides (for example, Li 4 Ti 5 O 12 ), metal materials, lithium alloy negative electrode materials, and the like. Is mentioned. In some cases, two or more negative electrode active materials may be used in combination. Preferably, from the viewpoint of capacity and output characteristics, a carbon material or a lithium-transition metal composite oxide is used as the negative electrode active material. Of course, negative electrode active materials other than those described above may be used.
- the average particle diameter of the negative electrode active material is not particularly limited, but is preferably 1 to 100 ⁇ m, more preferably 1 to 20 ⁇ m from the viewpoint of increasing the output.
- the negative electrode active material layer preferably contains at least an aqueous binder.
- a water-based binder has a high binding power.
- it is easy to procure water as a raw material and since steam is generated at the time of drying, the capital investment in the production line can be greatly suppressed, and the environmental load can be reduced. There is.
- the water-based binder refers to a binder using water as a solvent or a dispersion medium, and specifically includes a thermoplastic resin, a polymer having rubber elasticity, a water-soluble polymer, or a mixture thereof.
- the binder using water as a dispersion medium refers to a polymer that includes all expressed as latex or emulsion and is emulsified or suspended in water.
- kind a polymer latex that is emulsion-polymerized in a system that self-emulsifies.
- water-based binders include styrene polymers (styrene-butadiene rubber, styrene-vinyl acetate copolymer, styrene-acrylic copolymer, etc.), acrylonitrile-butadiene rubber, methyl methacrylate-butadiene rubber, ) Acrylic polymers (polyethyl acrylate, polyethyl methacrylate, polypropyl acrylate, polymethyl methacrylate (methyl methacrylate rubber), polypropyl methacrylate, polyisopropyl acrylate, polyisopropyl methacrylate, polybutyl acrylate, polybutyl methacrylate, polyhexyl acrylate , Polyhexyl methacrylate, polyethylhexyl acrylate, polyethylhexyl methacrylate, polylauryl acrylate, polylauryl meta Acrylate, etc.), polytyren
- the aqueous binder may contain at least one rubber binder selected from the group consisting of styrene-butadiene rubber, acrylonitrile-butadiene rubber, methyl methacrylate-butadiene rubber, and methyl methacrylate rubber from the viewpoint of binding properties. preferable. Furthermore, it is preferable that the water-based binder contains styrene-butadiene rubber because of good binding properties.
- Water-soluble polymers suitable for use in combination with styrene-butadiene rubber include polyvinyl alcohol and modified products thereof, starch and modified products thereof, cellulose derivatives (such as carboxymethyl cellulose, methyl cellulose, hydroxyethyl cellulose, and salts thereof), polyvinyl Examples include pyrrolidone, polyacrylic acid (salt), or polyethylene glycol. Among them, it is preferable to combine styrene-butadiene rubber and carboxymethyl cellulose (salt) as a binder.
- the content of the aqueous binder is preferably 80 to 100% by weight, preferably 90 to 100% by weight, and preferably 100% by weight.
- the separator has a function of holding an electrolyte and ensuring lithium ion conductivity between the positive electrode and the negative electrode, and a function as a partition wall between the positive electrode and the negative electrode.
- separator examples include a separator made of a porous sheet made of a polymer or fiber that absorbs and holds the electrolyte and a nonwoven fabric separator.
- a microporous (microporous film) can be used as the separator of the porous sheet made of polymer or fiber.
- the porous sheet made of the polymer or fiber include polyolefins such as polyethylene (PE) and polypropylene (PP); a laminate in which a plurality of these are laminated (for example, three layers of PP / PE / PP) And a microporous (microporous membrane) separator made of a hydrocarbon resin such as polyimide, aramid, polyvinylidene fluoride-hexafluoropropylene (PVdF-HFP), glass fiber, and the like.
- PE polyethylene
- PP polypropylene
- a microporous (microporous membrane) separator made of a hydrocarbon resin such as polyimide, aramid, polyvinylidene fluoride-hexafluoropropylene (PVdF-HFP), glass fiber, and the like.
- the thickness of the microporous (microporous membrane) separator cannot be uniquely defined because it varies depending on the intended use. For example, in applications such as secondary batteries for driving motors such as electric vehicles (EV), hybrid electric vehicles (HEV), and fuel cell vehicles (FCV), it is 4 to 60 ⁇ m in a single layer or multiple layers. Is desirable.
- the fine pore diameter of the microporous (microporous membrane) separator is desirably 1 ⁇ m or less (usually a pore diameter of about several tens of nm).
- nonwoven fabric separator cotton, rayon, acetate, nylon, polyester; polyolefins such as PP and PE; conventionally known ones such as polyimide and aramid are used alone or in combination.
- the bulk density of the nonwoven fabric is not particularly limited as long as sufficient battery characteristics can be obtained by the impregnated polymer gel electrolyte.
- the thickness of the nonwoven fabric separator may be the same as that of the electrolyte layer, and is preferably 5 to 200 ⁇ m, particularly preferably 10 to 100 ⁇ m.
- the separator includes an electrolyte.
- the electrolyte is not particularly limited as long as it can exhibit such a function, but a liquid electrolyte or a gel polymer electrolyte is used.
- a gel polymer electrolyte By using the gel polymer electrolyte, the distance between the electrodes is stabilized, the occurrence of polarization is suppressed, and the durability (cycle characteristics) is improved.
- the liquid electrolyte functions as a lithium ion carrier.
- the liquid electrolyte constituting the electrolytic solution layer has a form in which a lithium salt as a supporting salt is dissolved in an organic solvent as a plasticizer.
- organic solvent include carbonates such as ethylene carbonate (EC), propylene carbonate (PC), dimethyl carbonate (DMC), diethyl carbonate (DEC), and ethyl methyl carbonate.
- EC ethylene carbonate
- PC propylene carbonate
- DMC dimethyl carbonate
- DEC diethyl carbonate
- ethyl methyl carbonate ethyl methyl carbonate.
- Li (CF 3 SO 2) 2 N Li (C 2 F 5 SO 2) 2 N, LiPF 6, LiBF 4, LiClO 4, LiAsF 6, LiTaF such 6, LiCF 3 SO 3
- a compound that can be added to the active material layer of the electrode can be similarly employed.
- the liquid electrolyte may further contain additives other than the components described above.
- additives include, for example, vinylene carbonate, methyl vinylene carbonate, dimethyl vinylene carbonate, phenyl vinylene carbonate, diphenyl vinylene carbonate, ethyl vinylene carbonate, diethyl vinylene carbonate, vinyl ethylene carbonate, 1,2-divinyl ethylene carbonate.
- vinylene carbonate, methyl vinylene carbonate, and vinyl ethylene carbonate are preferable, and vinylene carbonate and vinyl ethylene carbonate are more preferable.
- These cyclic carbonates may be used alone or in combination of two or more.
- the gel polymer electrolyte has a configuration in which the above liquid electrolyte is injected into a matrix polymer (host polymer) made of an ion conductive polymer.
- a gel polymer electrolyte as the electrolyte is superior in that the fluidity of the electrolyte is lost and the ion conductivity between the layers is easily cut off.
- ion conductive polymer used as the matrix polymer (host polymer) examples include polyethylene oxide (PEO), polypropylene oxide (PPO), polyethylene glycol (PEG), polyacrylonitrile (PAN), polyvinylidene fluoride-hexafluoropropylene ( PVdF-HEP), poly (methyl methacrylate (PMMA), and copolymers thereof.
- PEO polyethylene oxide
- PPO polypropylene oxide
- PEG polyethylene glycol
- PAN polyacrylonitrile
- PVdF-HEP polyvinylidene fluoride-hexafluoropropylene
- PMMA methyl methacrylate
- the matrix polymer of gel electrolyte can express excellent mechanical strength by forming a crosslinked structure.
- thermal polymerization, ultraviolet polymerization, radiation polymerization, electron beam polymerization, etc. are performed on a polymerizable polymer (for example, PEO or PPO) for forming a polymer electrolyte using an appropriate polymerization initiator.
- a polymerization treatment may be performed.
- the separator is preferably a separator in which a heat-resistant insulating layer is laminated on a porous substrate (a separator with a heat-resistant insulating layer).
- the heat-resistant insulating layer is a ceramic layer containing inorganic particles and a binder.
- a highly heat-resistant separator having a melting point or a heat softening point of 150 ° C. or higher, preferably 200 ° C. or higher is used.
- the separator is less likely to curl in the battery manufacturing process due to the effect of suppressing thermal shrinkage and high mechanical strength.
- the inorganic particles in the heat resistant insulating layer contribute to the mechanical strength and heat shrinkage suppressing effect of the heat resistant insulating layer.
- the material used as the inorganic particles is not particularly limited. Examples thereof include silicon, aluminum, zirconium, titanium oxides (SiO 2 , Al 2 O 3 , ZrO 2 , TiO 2 ), hydroxides and nitrides, and composites thereof. These inorganic particles may be derived from mineral resources such as boehmite, zeolite, apatite, kaolin, mullite, spinel, olivine and mica, or may be artificially produced. Moreover, only 1 type may be used individually for these inorganic particles, and 2 or more types may be used together. Of these, silica (SiO 2 ) or alumina (Al 2 O 3 ) is preferably used, and alumina (Al 2 O 3 ) is more preferably used from the viewpoint of cost.
- the basis weight of the heat-resistant particles is not particularly limited, but is preferably 5 to 15 g / m 2 . If it is this range, sufficient ion conductivity will be acquired and it is preferable at the point which maintains heat resistant strength.
- the binder in the heat-resistant insulating layer has a role of adhering the inorganic particles and the inorganic particles to the resin porous substrate layer. With the binder, the heat-resistant insulating layer is stably formed, and peeling between the porous substrate layer and the heat-resistant insulating layer is prevented.
- the binder used for the heat-resistant insulating layer is not particularly limited.
- a compound such as butadiene rubber, polyvinylidene fluoride (PVDF), polytetrafluoroethylene (PTFE), polyvinyl fluoride (PVF), or methyl acrylate can be used as a binder.
- PVDF polyvinylidene fluoride
- PTFE polytetrafluoroethylene
- PVF polyvinyl fluoride
- methyl acrylate methyl acrylate
- PVDF polyvinylidene fluoride
- these compounds only 1 type may be used independently and 2 or more types may be used together.
- the binder content in the heat resistant insulating layer is preferably 2 to 20% by weight with respect to 100% by weight of the heat resistant insulating layer.
- the binder content is 2% by weight or more, the peel strength between the heat-resistant insulating layer and the porous substrate layer can be increased, and the vibration resistance of the separator can be improved.
- the binder content is 20% by weight or less, the gaps between the inorganic particles are appropriately maintained, so that sufficient lithium ion conductivity can be ensured.
- the thermal contraction rate of the separator with a heat-resistant insulating layer is preferably 10% or less for both MD and TD after holding for 1 hour at 150 ° C. and 2 gf / cm 2 .
- the material which comprises a current collector plate (25, 27) is not restrict
- a constituent material of the current collector plate for example, metal materials such as aluminum, copper, titanium, nickel, stainless steel (SUS), and alloys thereof are preferable. From the viewpoint of light weight, corrosion resistance, and high conductivity, aluminum and copper are more preferable, and aluminum is particularly preferable.
- the same material may be used for the positive electrode current collecting plate 27 and the negative electrode current collecting plate 25, and different materials may be used.
- the battery outer body 29 As the battery outer body 29, a bag-like case using a laminate film containing a metal such as aluminum that can cover the power generating element is also used. It is. When such a laminate film is used as an exterior body, the heat dissipation of the battery is high, and the power generation element is not easily warmed even when the power generation element generates Joule heat when used under a low temperature condition. Therefore, it can be said that the output characteristics are likely to deteriorate due to the decrease in lithium ion conductivity under low temperature conditions.
- a laminate film having a three-layer structure in which PP, aluminum, and nylon are laminated in this order can be used as the laminate film, but the laminate film is not limited thereto.
- a laminate film is desirable from the viewpoint that it is excellent in high output and cooling performance, and can be suitably used for a battery for large equipment for EV and HEV.
- the outer package is more preferably a laminate film containing aluminum.
- FIG. 2 is a perspective view showing the appearance of a flat lithium ion secondary battery which is a typical embodiment of the secondary battery.
- a flat laminated battery having a structure in which the power generation element is enclosed in a battery outer package made of a laminate film containing aluminum.
- the flat lithium ion secondary battery 50 has a rectangular flat shape, and a positive electrode tab 58 and a negative electrode tab 59 for taking out electric power are drawn out from both sides thereof.
- the power generation element 57 is encased by the battery outer packaging material 52 of the lithium ion secondary battery 50, and the periphery thereof is heat-sealed. The power generation element 57 is sealed with the positive electrode tab 58 and the negative electrode tab 59 pulled out to the outside.
- the power generation element 57 corresponds to the power generation element 21 of the lithium ion secondary battery 10 shown in FIG. 1 described above.
- the power generation element 57 is formed by laminating a plurality of single battery layers (single cells) 19 composed of a positive electrode (positive electrode active material layer) 15, an electrolyte layer 17, and a negative electrode (negative electrode active material layer) 13.
- the lithium ion secondary battery is not limited to a stacked flat shape.
- the wound lithium ion secondary battery may have a cylindrical shape, or may have a shape that is a flattened rectangular shape by deforming such a cylindrical shape.
- a laminate film may be used for the exterior material, and the conventional cylindrical can (metal can) may be used, for example, It does not restrict
- the power generation element is covered with an aluminum laminate film. With this configuration, weight reduction can be achieved.
- the tabs 58 and 59 shown in FIG. 2 are not particularly limited.
- the positive electrode tab 58 and the negative electrode tab 59 may be drawn out from the same side, or the positive electrode tab 58 and the negative electrode tab 59 may be divided into a plurality of parts and taken out from each side, as shown in FIG. It is not limited to.
- a terminal may be formed using a cylindrical can (metal can).
- the battery storage space is about 170L. Since auxiliary devices such as cells and charge / discharge control devices are stored in this space, the storage efficiency of a normal cell is about 50%. The efficiency of loading cells into this space is a factor that governs the cruising range of electric vehicles. If the size of the single cell is reduced, the loading efficiency is impaired, so that the cruising distance cannot be secured.
- the battery structure in which the power generation element is covered with the exterior body is preferably large.
- the length of the short side of the laminated cell battery is preferably 100 mm or more. Such a large battery can be used for vehicle applications.
- the length of the short side of the laminated cell battery refers to the side having the shortest length.
- the upper limit of the short side length is not particularly limited, but is usually 400 mm or less.
- volume energy density and rated discharge capacity In a general electric vehicle, a travel distance (cruising range) by a single charge is 100 km. Considering such a cruising distance, the volume energy density of the battery is preferably 157 Wh / L or more, and the rated capacity is preferably 20 Wh or more.
- the nonaqueous electrolyte secondary battery according to this embodiment is a flat laminated battery, and the ratio of the battery area to the rated capacity (projected area of the battery including the battery outer package) is 5 cm 2 / It is preferably Ah or more and the rated capacity is 3 Ah or more.
- the aspect ratio of the rectangular electrode is preferably 1 to 3, and more preferably 1 to 2.
- the electrode aspect ratio is defined as the aspect ratio of the rectangular positive electrode active material layer.
- the assembled battery is configured by connecting a plurality of batteries. Specifically, at least two or more are used, and are configured by serialization, parallelization, or both. Capacitance and voltage can be freely adjusted by paralleling in series.
- a small assembled battery that can be attached and detached by connecting a plurality of batteries in series or in parallel. Then, a plurality of small assembled batteries that can be attached and detached are connected in series or in parallel to provide a large capacity and large capacity suitable for vehicle drive power supplies and auxiliary power supplies that require high volume energy density and high volume output density.
- An assembled battery having an output can also be formed. How many batteries are connected to make an assembled battery, and how many small assembled batteries are stacked to make a large-capacity assembled battery depends on the battery capacity of the mounted vehicle (electric vehicle) It may be determined according to the output.
- the nonaqueous electrolyte secondary battery of the present invention maintains a discharge capacity even when used for a long period of time, and has good cycle characteristics. Furthermore, the volume energy density is high. Vehicle applications such as electric vehicles, hybrid electric vehicles, fuel cell vehicles, and hybrid fuel cell vehicles require higher capacity, larger size, and longer life than electric and portable electronic devices. . Therefore, the nonaqueous electrolyte secondary battery can be suitably used as a vehicle power source, for example, a vehicle driving power source or an auxiliary power source.
- a battery or an assembled battery formed by combining a plurality of these batteries can be mounted on the vehicle.
- a plug-in hybrid electric vehicle having a long EV mileage or an electric vehicle having a long charge mileage can be formed by mounting such a battery.
- a car a hybrid car, a fuel cell car, an electric car (four-wheeled vehicles (passenger cars, trucks, buses, commercial vehicles, light cars, etc.) This is because it can be used for motorcycles (including motorcycles) and tricycles) to provide a long-life and highly reliable automobile.
- the application is not limited to automobiles.
- it can be applied to various power sources for moving vehicles such as other vehicles, for example, trains, and power sources for mounting such as uninterruptible power supplies. It is also possible to use as.
- the metal composite hydroxide and lithium carbonate were weighed so that the ratio of the total number of moles of metals other than Li (Ni, Co, Mn) and the number of moles of Li was 1: 1, and then mixed well.
- the temperature was raised at a rate of temperature increase of 5 ° C./min, calcined at 900 ° C. for 2 hours in an air atmosphere, then heated at a rate of temperature increase of 3 ° C./min.
- an NMC composite oxide which is a positive electrode active material having a composition of LiNi 0.50 Mn 0.30 Co 0.20 O 2 was obtained.
- the average secondary particle diameter of the obtained NMC composite oxide was 10 ⁇ m.
- a positive electrode active material layer was produced (the porosity of the positive electrode active material layer was set to 25%).
- a positive electrode active material layer was formed on the back surface to produce a positive electrode in which a positive electrode active material layer was formed on both surfaces of a positive electrode current collector (aluminum foil).
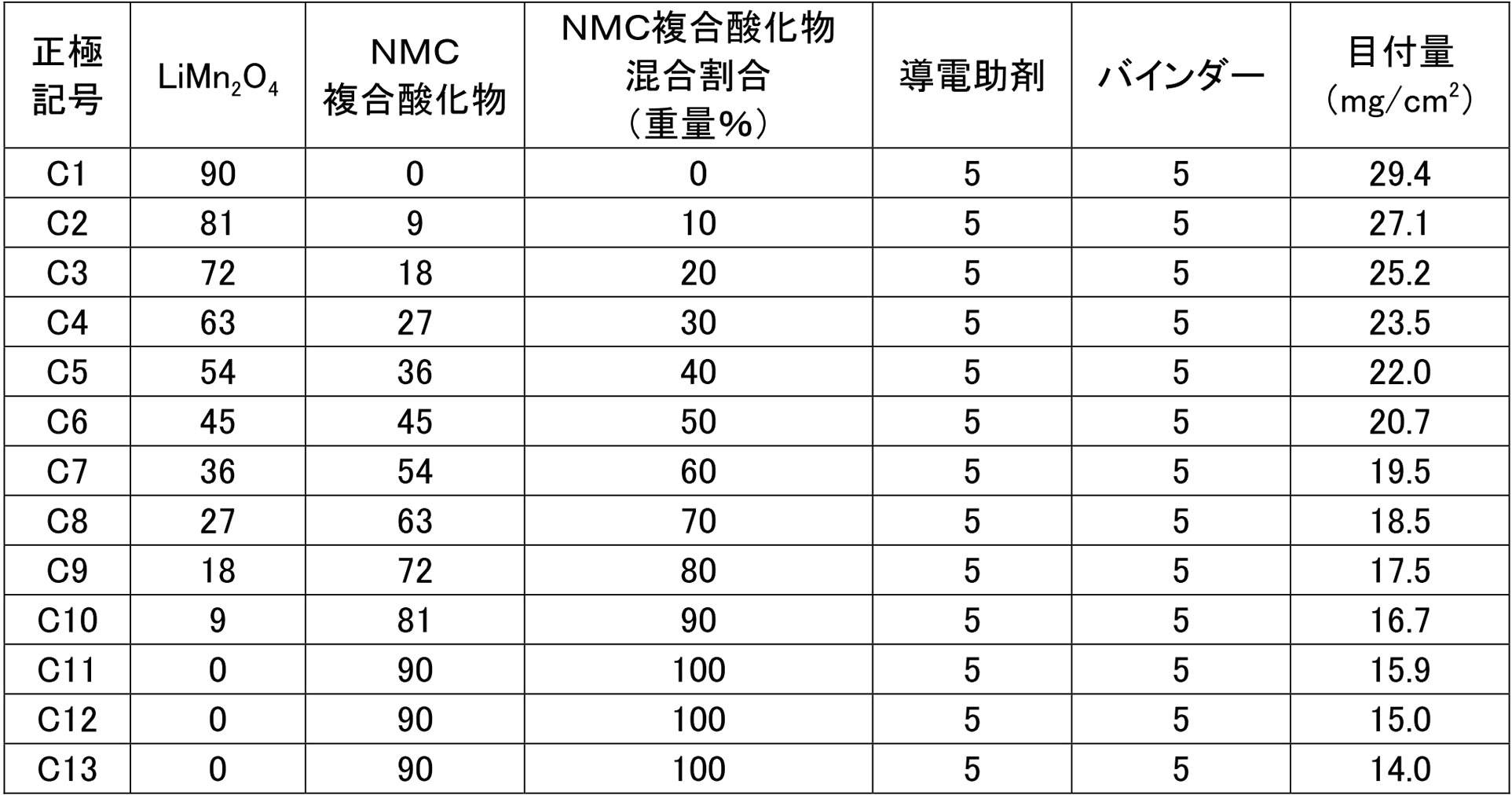
- the single-side coating amount of the positive electrode active material layer is set so that the charging capacity per unit area is 2.65 mAh / cm 2 for C1 to C11, and the charging amount per single area is for C12 and C13. There were set so that each becomes 2.55mAh / cm 2 and 2.33mAh / cm 2. In this way, 13 types of positive electrodes having different compositions of the positive electrode active material were produced as shown in Table 1 below (numbers are% by weight). These positive electrodes were cut into 200 mm squares.
- negative electrode 95% by weight of artificial graphite as a negative electrode active material 2% by weight of carbon black (Super-P, manufactured by 3M) as a conductive auxiliary agent, 1% by weight of ammonium salt of carboxymethyl cellulose as a binder and styrene-butadiene
- a negative electrode active material slurry was prepared by dispersing 2% by weight of polymer latex in purified water. This negative electrode active material slurry was applied to a copper foil (thickness 10 ⁇ m) serving as a negative electrode current collector, dried at 120 ° C. for 3 minutes, and then compression molded with a roll press to produce a negative electrode.
- a negative electrode active material layer was formed on the back surface to prepare a negative electrode in which a negative electrode active material layer was formed on both sides of a negative electrode current collector (copper foil).
- the coating amount of the negative electrode active material layer was adjusted such that C1 to C11 had an A / C ratio of 1.20 with the positive electrode facing when the test cell described later was prepared, C12 and C13 were adjusted so that the A / C ratios were 1.27 and 1.36, respectively. Further, this negative electrode was cut into a 202 mm square.
- the injection amount of the electrolytic solution was an amount that is 1.40 times the total pore volume (calculated by calculation) of the positive electrode active material layer, the negative electrode active material layer, and the separator.
- the opening of the aluminum laminate sheet bag was sealed so that the current extraction tabs connected to both electrodes were led out, and a test cell that was a laminated lithium ion secondary battery was completed.
- 16 types of test cells with different types of positive electrodes were produced as shown in Table 2 below.
- Table 2 below shows the rated capacity (Ah) of each test cell and the ratio of the battery area to the rated capacity (cm 2 / Ah).
- the rated capacity of the battery was determined as follows.
- the rated capacity of the test cell is left for about 10 hours after injecting the electrolyte, and the initial charge is performed. Thereafter, the temperature is measured by the following procedures 1 to 5 at a temperature of 25 ° C. and a voltage range of 3.0 V to 4.15 V.
- Procedure 1 After reaching 4.15V by constant current charging at 0.2C, pause for 5 minutes.
- Procedure 2 After Procedure 1, charge for 1.5 hours with constant voltage charging and rest for 5 minutes.
- Procedure 3 After reaching 3.0 V by constant current discharge of 0.2 C, discharge at constant voltage discharge for 2 hours, and then rest for 10 seconds.
- Procedure 4 After reaching 4.15 V by constant current charging at 0.2 C, charge for 2.5 hours by constant voltage charging, and then rest for 10 seconds.
- Procedure 5 After reaching 3.0V by constant current discharge of 0.2 C, discharge at constant voltage discharge for 2 hours, and then stop for 10 seconds.
- the discharge capacity (CCCV discharge capacity) in the discharge from the constant current discharge to the constant voltage discharge in the procedure 5 was defined as the rated capacity.
Abstract
Description
図1は、本発明の電池の一実施形態である積層型電池の概要を模式的に表した断面概略図である。なお、本明細書においては、図1に示す扁平型(積層型)の双極型でないリチウムイオン二次電池を例に挙げて詳細に説明するが、本発明の技術的範囲はかような形態のみに制限されない。
図1は、扁平型(積層型)の双極型ではない非水電解質リチウムイオン二次電池(以下、単に「積層型電池」ともいう)の基本構成を模式的に表した断面概略図である。図1に示すように、本実施形態の積層型電池10は、実際に充放電反応が進行する略矩形の発電要素21が、外装体である電池外装材29の内部に封止された構造を有する。ここで、発電要素21は、正極と、セパレータ17と、負極とを積層した構成を有している。なお、セパレータ17は、非水電解質(例えば、液体電解質)を内蔵している。正極は、正極集電体12の両面に正極活物質層15が配置された構造を有する。負極は、負極集電体11の両面に負極活物質層13が配置された構造を有する。具体的には、1つの正極活物質層15とこれに隣接する負極活物質層13とが、セパレータ17を介して対向するようにして、負極、電解質層および正極がこの順に積層されている。これにより、隣接する正極、電解質層および負極は、1つの単電池層19を構成する。したがって、図1に示す積層型電池10は、単電池層19が複数積層されることで、電気的に並列接続されてなる構成を有するとも言える。
正極は、正極集電体と、前記正極集電体の表面に形成された正極活物質層とを有するものである。
正極集電体を構成する材料に特に制限はないが、好適には金属が用いられる。具体的には、金属としては、アルミニウム、ニッケル、鉄、ステンレス、チタン、銅、その他合金等などが挙げられる。これらのほか、ニッケルとアルミニウムとのクラッド材、銅とアルミニウムとのクラッド材、またはこれらの金属の組み合わせのめっき材などが好ましく用いられうる。また、金属表面にアルミニウムが被覆されてなる箔であってもよい。なかでも、電子伝導性や電池作動電位の観点からは、アルミニウム、ステンレス、銅が好ましい。
正極活物質層は、正極活物質を含む。本形態において、正極活物質は、スピネル系リチウムマンガン複合酸化物およびリチウムニッケル系複合酸化物を必須に含む。なお、正極活物質層に含まれる正極活物質の全量100重量%に占めるスピネル系リチウムマンガン複合酸化物およびリチウムニッケル系複合酸化物の合計量の割合は、好ましくは50重量%以上であり、より好ましくは70重量%以上であり、さらに好ましくは85重量%以上であり、いっそう好ましくは90重量%以上であり、特に好ましくは95重量%以上であり、最も好ましくは100重量%である。
スピネル系リチウムマンガン複合酸化物は、典型的にはLiMn2O4の組成を有し、スピネル構造を有する、リチウムおよびマンガンを必須に含有する複合酸化物であり、その具体的な構成や製造方法については、特開2000-77071号公報等の従来公知の知見が適宜参照されうる。
リチウムニッケル系複合酸化物は、リチウムとニッケルとを含有する複合酸化物である限り、その組成は具体的に限定されない。リチウムとニッケルとを含有する複合酸化物の典型的な例としては、リチウムニッケル複合酸化物(LiNiO2)が挙げられる。ただし、リチウムニッケル複合酸化物のニッケル原子の一部が他の金属原子で置換された複合酸化物がより好ましく、好ましい例として、リチウム-ニッケル-マンガン-コバルト複合酸化物(以下、単に「NMC複合酸化物」とも称する)は、リチウム原子層と遷移金属(Mn、NiおよびCoが秩序正しく配置)原子層とが酸素原子層を介して交互に積み重なった層状結晶構造を持ち、遷移金属Mの1原子あたり1個のLi原子が含まれ、取り出せるLi量が、スピネル系リチウムマンガン複合酸化物の2倍、つまり供給能力が2倍になり、高い容量を持つことができる。加えて、LiNiO2より高い熱安定性を有しているため、正極活物質として用いられるリチウムニッケル系複合酸化物の中でも特に有利である。
正極活物質層は上述した正極活物質の他、必要に応じて、導電助剤、バインダー、電解質(ポリマーマトリックス、イオン伝導性ポリマー、電解液など)、イオン伝導性を高めるためのリチウム塩などのその他の添加剤をさらに含む。ただし、正極活物質層および後述の負極活物質層中、活物質として機能しうる材料の含有量は、85~99.5重量%であることが好ましい。
正極活物質層に用いられるバインダーとしては、特に限定されないが、例えば、以下の材料が挙げられる。ポリエチレン、ポリプロピレン、ポリエチレンテレフタレート(PET)、ポリエーテルニトリル、ポリアクリロニトリル、ポリイミド、ポリアミド、セルロース、カルボキシメチルセルロース(CMC)およびその塩、エチレン-酢酸ビニル共重合体、ポリ塩化ビニル、スチレン・ブタジエンゴム(SBR)、イソプレンゴム、ブタジエンゴム、エチレン・プロピレンゴム、エチレン・プロピレン・ジエン共重合体、スチレン・ブタジエン・スチレンブロック共重合体およびその水素添加物、スチレン・イソプレン・スチレンブロック共重合体およびその水素添加物などの熱可塑性高分子、ポリフッ化ビニリデン(PVdF)、ポリテトラフルオロエチレン(PTFE)、テトラフルオロエチレン・ヘキサフルオロプロピレン共重合体(FEP)、テトラフルオロエチレン・パーフルオロアルキルビニルエーテル共重合体(PFA)、エチレン・テトラフルオロエチレン共重合体(ETFE)、ポリクロロトリフルオロエチレン(PCTFE)、エチレン・クロロトリフルオロエチレン共重合体(ECTFE)、ポリフッ化ビニル(PVF)等のフッ素樹脂、ビニリデンフルオライド-ヘキサフルオロプロピレン系フッ素ゴム(VDF-HFP系フッ素ゴム)、ビニリデンフルオライド-ヘキサフルオロプロピレン-テトラフルオロエチレン系フッ素ゴム(VDF-HFP-TFE系フッ素ゴム)、ビニリデンフルオライド-ペンタフルオロプロピレン系フッ素ゴム(VDF-PFP系フッ素ゴム)、ビニリデンフルオライド-ペンタフルオロプロピレン-テトラフルオロエチレン系フッ素ゴム(VDF-PFP-TFE系フッ素ゴム)、ビニリデンフルオライド-パーフルオロメチルビニルエーテル-テトラフルオロエチレン系フッ素ゴム(VDF-PFMVE-TFE系フッ素ゴム)、ビニリデンフルオライド-クロロトリフルオロエチレン系フッ素ゴム(VDF-CTFE系フッ素ゴム)等のビニリデンフルオライド系フッ素ゴム、エポキシ樹脂等が挙げられる。これらのバインダーは、単独で用いてもよいし、2種以上を併用してもよい。
負極活物質層は活物質を含み、必要に応じて、導電助剤、バインダー、電解質(ポリマーマトリックス、イオン伝導性ポリマー、電解液など)、イオン伝導性を高めるためのリチウム塩などのその他の添加剤をさらに含む。導電助剤、バインダー、電解質(ポリマーマトリックス、イオン伝導性ポリマー、電解液など)、イオン伝導性を高めるためのリチウム塩などのその他の添加剤については、上記正極活物質層の欄で述べたものと同様である。
セパレータは、電解質を保持して正極と負極との間のリチウムイオン伝導性を確保する機能、および正極と負極との間の隔壁としての機能を有する。
十分な電池特性が得られるものであればよく、特に制限されるべきものではない。さらに、不織布セパレータの厚さは、電解質層と同じであればよく、好ましくは5~200μmであり、特に好ましくは10~100μmである。
集電板(25、27)を構成する材料は、特に制限されず、リチウムイオン二次電池用の集電板として従来用いられている公知の高導電性材料が用いられうる。集電板の構成材料としては、例えば、アルミニウム、銅、チタン、ニッケル、ステンレス鋼(SUS)、これらの合金等の金属材料が好ましい。軽量、耐食性、高導電性の観点から、より好ましくはアルミニウム、銅であり、特に好ましくはアルミニウムである。なお、正極集電板27と負極集電板25とでは、同一の材料が用いられてもよいし、異なる材料が用いられてもよい。
また、図示は省略するが、集電体11と集電板(25、27)との間を正極リードや負極リードを介して電気的に接続してもよい。正極および負極リードの構成材料としては、公知のリチウムイオン二次電池において用いられる材料が同様に採用されうる。なお、外装から取り出された部分は、周辺機器や配線などに接触して漏電したりして製品(例えば、自動車部品、特に電子機器等)に影響を与えないように、耐熱絶縁性の熱収縮チューブなどにより被覆することが好ましい。
本形態に係る非水電解質二次電池では、電池外装体29として、発電要素を覆うことができる、アルミニウムなどの金属を含むラミネートフィルムを用いた袋状のケースが用いられることも特徴の1つである。かようなラミネートフィルムを外装体として用いた場合には、電池の放熱性が高く、低温条件下で使用される場合に発電要素がジュール発熱しても発電要素が温まりにくい。したがって、低温条件下におけるリチウムイオン伝導度の低下に起因する出力特性の低下が生じやすいと言える。一方、従来の巻回型電池に用いられているような缶ケースでは、電池の放熱性が低いことからジュール発熱によって発電要素の温度は容易に上昇することができる。したがって、低温条件下における出力特性の低下の課題が生じにくいのである。
図2は、二次電池の代表的な実施形態である扁平なリチウムイオン二次電池の外観を表した斜視図である。このリチウムイオン二次電池のように、本発明における好ましい実施形態によれば、アルミニウムを含むラミネートフィルムからなる電池外装体に前記発電要素が封入されてなる構成を有する扁平積層型ラミネート電池が提供される。
一般的な電気自動車では、一回の充電による走行距離(航続距離)は100kmが市場要求である。かような航続距離を考慮すると、電池の体積エネルギー密度は157Wh/L以上であることが好ましく、かつ定格容量は20Wh以上であることが好ましい。
組電池は、電池を複数個接続して構成した物である。詳しくは少なくとも2つ以上用いて、直列化あるいは並列化あるいはその両方で構成されるものである。直列、並列化することで容量および電圧を自由に調節することが可能になる。
本発明の非水電解質二次電池は、長期使用しても放電容量が維持され、サイクル特性が良好である。さらに、体積エネルギー密度が高い。電気自動車やハイブリッド電気自動車や燃料電池車やハイブリッド燃料電池自動車などの車両用途においては、電気・携帯電子機器用途と比較して、高容量、大型化が求められるとともに、長寿命化が必要となる。したがって、上記非水電解質二次電池は、車両用の電源として、例えば、車両駆動用電源や補助電源に好適に利用することができる。
硫酸ニッケル、硫酸コバルト、および硫酸マンガンを溶解した水溶液(1mol/L)に、60℃にて水酸化ナトリウムおよびアンモニアを連続的に供給してpHを11.3に調整し、共沈法によりニッケルとマンガンとコバルトとが50:30:20のモル比で固溶してなる金属複合水酸化物を作製した。
他の正極活物質として、スピネル系リチウムマンガン複合酸化物(LiMn2O4)を準備した。なお、ここで準備したスピネル系リチウムマンガン複合酸化物の平均2次粒子径は10μmであった。
上記で調製・準備したNMC複合酸化物およびスピネル系リチウムマンガン複合酸化物を合計で90重量%、並びに、導電助剤としてカーボンブラック(Super-P、3M社製)5重量%、バインダーとしてポリフッ化ビニリデン(PVDF)(クレハ社製、♯7200)5重量%、およびスラリー粘度調整溶媒であるN-メチル-2-ピロリドン(NMP)を適量混合して正極活物質スラリーを調製し、得られた正極活物質スラリーを集電体であるアルミニウム箔(厚さ:20μm)の表面に塗布し、120℃で3分間乾燥後、ロールプレス機で圧縮成形して平面形状が矩形の正極活物質層を作製した(正極活物質層の空孔率が25%となるようにした)。裏面にも同様にして正極活物質層を形成して、正極集電体(アルミニウム箔)の両面に正極活物質層が形成されてなる正極を作製した。なお、正極活物質層の片面塗工量は、C1~C11については単位面積当たりの充電容量が2.65mAh/cm2となるように設定し、C12およびC13については単一面積あたりの充電量がそれぞれ2.55mAh/cm2および2.33mAh/cm2となるように設定した。このようにして、下記の表1に示すように正極活物質の組成の異なる正極を13種類作製した(数字は重量%である)。また、これらの正極は200mm四方にカットして用いた。
負極活物質として人造グラファイト95重量%、導電助剤としてカーボンブラック(Super-P、3M社製)2重量%、バインダーとしてカルボキシメチルセルロースのアンモニウム塩1重量%およびスチレン-ブタジエン共重合体ラテックス2重量%を精製水中に分散させて負極活物質スラリーを調製した。この負極活物質スラリーを負極集電体となる銅箔(厚さ10μm)に塗布し、120℃で3分間乾燥後、ロールプレス機で圧縮成形して負極を作製した。裏面にも同様にして負極活物質層を形成して、負極集電体(銅箔)の両面に負極活物質層が形成されてなる負極を作製した。なお、負極活物質層の塗工量については、C1~C11については後述する試験用セルの作製の際に対向する正極との間でA/C比が1.20となるように調整し、C12およびC13についてはA/C比がそれぞれ1.27および1.36となるように調整した。また、この負極は202mm四方にカットして用いた。
上記(3)で作製した正極と、上記(4)で作製した負極とを、下記の表2に示すように選択し、セパレータ(厚さ25μm、セルガード♯2500、ポリポア社製)を介して交互に積層(正極3層、負極4層)することによって発電要素を作製した。得られた発電要素を外装であるアルミラミネートシート製のバッグ中に載置し、電解液を注液した。電解液としては、1.0M LiPF6をエチレンカーボネート(EC)とジエチルカーボネート(DEC)との3:7(EC:DECの体積比)混合溶媒に溶解した溶液100重量%に対して、添加剤であるビニレンカーボネートを1質量%添加したものを用いた。ここで、電解液の注液量は、正極活物質層、負極活物質層およびセパレータの全空孔容積(計算により算出した)に対して1.40倍となる量とした。次いで、真空条件下において、両電極に接続された電流取り出しタブが導出するようにアルミラミネートシート製バッグの開口部を封止し、ラミネート型リチウムイオン二次電池である試験用セルを完成させた。このようにして、下記の表2に示すように正極の種類の異なる試験用セルを16種類作製した。なお、得られた各試験用セルの定格容量(Ah)および定格容量に対する電池面積の比(cm2/Ah)を下記の表2に示す。ここで、電池の定格容量は、以下により求めた。
定格容量は、試験用セルについて、電解液を注入した後で、10時間程度放置し、初期充電を行う。その後、温度25℃、3.0Vから4.15Vの電圧範囲で、次の手順1~5によって測定される。
≪低温条件下での出力特性の測定≫
上記(5)で作製した試験用セルを24時間放置し、開回路電圧(OCV:Open Circuit Voltage)が安定した後、0.2Cレートでカットオフ電圧4.15Vまで充電した。次いで、1時間の休止後、DOD10%、30%、50%、70%または90%のいずれかに対応するOCVまで0.2Cレートで放電した。その後、各DODの状態から1Cレートで5秒間放電し、その際の電流値および電圧をプロットしたグラフの傾きから電池の内部抵抗を算出した。この内部抵抗の測定を0℃および-20℃の2つの温度条件下で行い、-20℃における内部抵抗の0℃における内部抵抗の比の値を算出した。結果を下記の表2に示す。
11 負極集電体、
12 正極集電体、
13 負極活物質層、
15 正極活物質層、
17 セパレータ、
19 単電池層、
21、57 発電要素、
25 負極集電板、
27 正極集電板、
29、52 電池外装材、
58 正極タブ、
59 負極タブ。
Claims (8)
- 正極集電体の表面に正極活物質を含む正極活物質層が形成されてなる正極と、
負極集電体の表面に負極活物質を含む負極活物質層が形成されてなる負極と、
セパレータと、
を含む発電要素が、ラミネートフィルムからなる外装体の内部に封入されてなり、
前記正極活物質が、スピネル系マンガン正極活物質と、リチウムニッケル系複合酸化物とを含み、かつスピネル系マンガン正極活物質とリチウムニッケル系複合酸化物との合計100重量%に対する前記リチウムニッケル系複合酸化物の混合比率が30~70重量%である、非水電解質二次電池。 - 前記リチウムニッケル系複合酸化物は、
一般式:LiaNibMcCodO2(但し、式中、a、b、c、dは、0.9≦a≦1.2、0<b<1、0<c≦0.5、0<d≦0.5、b+c+d=1を満たす。Mは、Mn、Ti、Zr、Nb、W、P、Al、Mg、V、Ca、SrおよびCrからなる群から選ばれる少なくとも1種である)で表される組成を有する、請求項1に記載の非水電解質二次電池。 - 前記b、cおよびdが、0.44≦b≦0.51、0.27≦c≦0.31、0.19≦d≦0.26である、請求項2に記載の非水電解質二次電池。
- 前記正極活物質層の目付量(片面塗工量)が18.5~23.5mg/cm2である、請求項1~3のいずれか1項に記載の非水電解質二次電池。
- 前記セパレータが耐熱絶縁層付セパレータである、請求項1~4のいずれか1項に記載の非水電解質二次電池。
- 定格容量に対する電池面積(電池外装体まで含めた電池の投影面積)の比の値が5cm2/Ah以上であり、かつ、定格容量が3Ah以上である、請求項1~5のいずれか1項に記載の非水電解質二次電池。
- 矩形状の正極活物質層の縦横比として定義される電極のアスペクト比が1~3である、請求項1~6のいずれか1項に記載の非水電解質二次電池。
- 前記ラミネートフィルムが、アルミニウムを含むラミネートフィルムである、請求項1~7のいずれか1項に記載の非水電解質二次電池。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201480023723.1A CN105144458B (zh) | 2013-04-26 | 2014-04-23 | 非水电解质二次电池 |
| JP2015513817A JP6036999B2 (ja) | 2013-04-26 | 2014-04-23 | 非水電解質二次電池 |
| KR1020157030582A KR101798313B1 (ko) | 2013-04-26 | 2014-04-23 | 비수전해질 이차 전지 |
| EP14789007.3A EP2991152B1 (en) | 2013-04-26 | 2014-04-23 | Nonaqueous-electrolyte secondary battery |
| US14/786,934 US20160079589A1 (en) | 2013-04-26 | 2014-04-23 | Non-aqueous electrolyte secondary battery |
| US16/391,800 US20190252675A1 (en) | 2013-04-26 | 2019-04-23 | Non-Aqueous Electrolyte Secondary Battery |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-094522 | 2013-04-26 | ||
| JP2013094522 | 2013-04-26 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/786,934 A-371-Of-International US20160079589A1 (en) | 2013-04-26 | 2014-04-23 | Non-aqueous electrolyte secondary battery |
| US16/391,800 Continuation US20190252675A1 (en) | 2013-04-26 | 2019-04-23 | Non-Aqueous Electrolyte Secondary Battery |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014175355A1 true WO2014175355A1 (ja) | 2014-10-30 |
Family
ID=51791922
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/061483 WO2014175355A1 (ja) | 2013-04-26 | 2014-04-23 | 非水電解質二次電池 |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US20160079589A1 (ja) |
| EP (1) | EP2991152B1 (ja) |
| JP (1) | JP6036999B2 (ja) |
| KR (1) | KR101798313B1 (ja) |
| CN (1) | CN105144458B (ja) |
| WO (1) | WO2014175355A1 (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2975676A4 (en) * | 2013-03-15 | 2016-04-06 | Nissan Motor | NONAQUEOUS ELECTROLYTE SECONDARY BATTERY CATHODE AND NONAQUEOUS ELECTROLYTE SECONDARY BATTERY USING THE SAME |
| EP2975677A4 (en) * | 2013-03-15 | 2016-04-13 | Nissan Motor | CATHODE FOR SECONDARY BATTERY WITH A WATER-FREE ELECTROLYTE AND SECONDARY BATTERY WITH A WATER-FREE ELECTROLYTE THEREWITH |
| JP2016184521A (ja) * | 2015-03-26 | 2016-10-20 | オートモーティブエナジーサプライ株式会社 | 非水電解質二次電池 |
| CN107851838A (zh) * | 2015-08-04 | 2018-03-27 | 汽车能源供应公司 | 锂离子二次电池 |
| US20190355969A1 (en) * | 2016-06-08 | 2019-11-21 | Nissan Motor Co., Ltd. | Non-Aqueous Electrolyte Secondary Battery |
| JP2022141208A (ja) * | 2021-03-15 | 2022-09-29 | プライムプラネットエナジー&ソリューションズ株式会社 | 正極および当該正極を備える非水電解質二次電池 |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108352557A (zh) * | 2015-11-13 | 2018-07-31 | 松下知识产权经营株式会社 | 非水电解质电池和非水电解质电池用部件 |
| WO2017212597A1 (ja) * | 2016-06-08 | 2017-12-14 | 日産自動車株式会社 | 非水電解質二次電池 |
| CN109565071A (zh) * | 2016-08-05 | 2019-04-02 | Nec能源元器件株式会社 | 电池和电池模块 |
| US9893361B1 (en) | 2016-09-19 | 2018-02-13 | Marc Jaker | Electrochemical cells and methods for making same |
| US11043659B2 (en) * | 2016-09-29 | 2021-06-22 | Panasonic Corporation | Positive electrode for nonaqueous electrolyte secondary battery and nonaqueous electrolyte secondary battery |
| CN110249471A (zh) * | 2017-01-26 | 2019-09-17 | 日本电气株式会社 | 二次电池 |
| US20190386284A1 (en) * | 2017-02-22 | 2019-12-19 | Envision Aesc Energy Devices Ltd. | Lithium ion battery |
| US20180261827A1 (en) * | 2017-03-08 | 2018-09-13 | Ricoh Company, Ltd. | Electrode, electrode element, nonaqueous electrolytic power storage device |
| CN110574194B (zh) | 2017-11-06 | 2022-06-03 | 株式会社Lg化学 | 尖晶石结构的锂锰基正极活性材料和包含所述正极活性材料的正极和锂二次电池 |
| JP7209475B2 (ja) * | 2018-04-09 | 2023-01-20 | 日産自動車株式会社 | 電池の製造方法 |
| CN112909318B (zh) * | 2018-08-31 | 2022-06-03 | 宁德时代新能源科技股份有限公司 | 锂离子二次电池及其制备方法与包含锂离子二次电池的电子产品和电动产品 |
| CN111354949B (zh) * | 2018-12-24 | 2021-12-07 | 宁德时代新能源科技股份有限公司 | 一种锂离子电池 |
| WO2020119431A1 (zh) * | 2018-12-14 | 2020-06-18 | 宁德时代新能源科技股份有限公司 | 一种锂离子电池 |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10112318A (ja) | 1996-10-08 | 1998-04-28 | Fuji Elelctrochem Co Ltd | 非水電解液二次電池 |
| JP2000077071A (ja) | 1998-08-27 | 2000-03-14 | Nec Corp | 非水電解液二次電池 |
| JP2006216305A (ja) * | 2005-02-02 | 2006-08-17 | Nissan Motor Co Ltd | 二次電池 |
| JP2007335157A (ja) * | 2006-06-13 | 2007-12-27 | Toshiba Corp | 蓄電池システム、車載電源システム、車両、および蓄電池システムの充電方法 |
| JP2008098142A (ja) * | 2006-09-14 | 2008-04-24 | Nissan Motor Co Ltd | 非水電解質二次電池用正極およびこれを用いた非水電解質二次電池 |
| JP2008532221A (ja) * | 2005-02-23 | 2008-08-14 | エルジー・ケム・リミテッド | リチウムイオン移動度及び電池容量が改良された二次バッテリー |
| JP2009087889A (ja) * | 2007-10-03 | 2009-04-23 | Sony Corp | 耐熱絶縁層付きセパレータ及び非水電解質二次電池 |
| JP2011105588A (ja) | 2009-10-22 | 2011-06-02 | Toda Kogyo Corp | ニッケル・コバルト・マンガン系化合物粒子粉末及びその製造方法、リチウム複合酸化物粒子粉末及びその製造方法並びに非水電解質二次電池 |
| WO2012036127A1 (ja) * | 2010-09-14 | 2012-03-22 | 日立マクセルエナジー株式会社 | 非水二次電池 |
| JP2012182025A (ja) * | 2011-03-01 | 2012-09-20 | Toyota Motor Corp | 二次電池 |
| JP2013094522A (ja) | 2011-11-04 | 2013-05-20 | Seong-Yoon Kim | 歩行補助具の制動装置 |
| JP2014060143A (ja) * | 2012-08-22 | 2014-04-03 | Sony Corp | 正極活物質、正極および電池、並びに電池パック、電子機器、電動車両、蓄電装置および電力システム |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003297338A (ja) * | 2002-03-29 | 2003-10-17 | Japan Storage Battery Co Ltd | 非水電解質二次電池用正極及びそれを用いた非水電解質二次電池 |
| JP3729164B2 (ja) * | 2002-08-05 | 2005-12-21 | 日産自動車株式会社 | 自動車用電池 |
| CN101388448A (zh) * | 2006-09-14 | 2009-03-18 | 日产自动车株式会社 | 非水电解质二次电池的正极及采用它的非水电解质二次电池 |
| DE102010011413A1 (de) * | 2010-03-15 | 2011-09-15 | Li-Tec Battery Gmbh | Kathodische Elektrode und elektrochemische Zelle für dynamische Einsätze |
| CN101867060B (zh) * | 2010-05-12 | 2013-09-18 | 清华大学 | 锂离子储能电池 |
| US8571218B2 (en) * | 2010-06-01 | 2013-10-29 | GreatCall, Inc. | Short message service cipher |
| EP2669986B1 (en) * | 2011-01-25 | 2016-08-31 | Murata Manufacturing Co., Ltd. | Nonaqueous electrolyte secondary cell |
| CN103066269B (zh) * | 2012-12-25 | 2015-08-19 | 贵州安达科技能源股份有限公司 | 一种三元正极活性材料制备方法及电池 |
-
2014
- 2014-04-23 EP EP14789007.3A patent/EP2991152B1/en active Active
- 2014-04-23 US US14/786,934 patent/US20160079589A1/en not_active Abandoned
- 2014-04-23 KR KR1020157030582A patent/KR101798313B1/ko active IP Right Grant
- 2014-04-23 WO PCT/JP2014/061483 patent/WO2014175355A1/ja active Application Filing
- 2014-04-23 JP JP2015513817A patent/JP6036999B2/ja active Active
- 2014-04-23 CN CN201480023723.1A patent/CN105144458B/zh active Active
-
2019
- 2019-04-23 US US16/391,800 patent/US20190252675A1/en not_active Abandoned
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10112318A (ja) | 1996-10-08 | 1998-04-28 | Fuji Elelctrochem Co Ltd | 非水電解液二次電池 |
| JP2000077071A (ja) | 1998-08-27 | 2000-03-14 | Nec Corp | 非水電解液二次電池 |
| JP2006216305A (ja) * | 2005-02-02 | 2006-08-17 | Nissan Motor Co Ltd | 二次電池 |
| JP2008532221A (ja) * | 2005-02-23 | 2008-08-14 | エルジー・ケム・リミテッド | リチウムイオン移動度及び電池容量が改良された二次バッテリー |
| JP2007335157A (ja) * | 2006-06-13 | 2007-12-27 | Toshiba Corp | 蓄電池システム、車載電源システム、車両、および蓄電池システムの充電方法 |
| JP2008098142A (ja) * | 2006-09-14 | 2008-04-24 | Nissan Motor Co Ltd | 非水電解質二次電池用正極およびこれを用いた非水電解質二次電池 |
| JP2009087889A (ja) * | 2007-10-03 | 2009-04-23 | Sony Corp | 耐熱絶縁層付きセパレータ及び非水電解質二次電池 |
| JP2011105588A (ja) | 2009-10-22 | 2011-06-02 | Toda Kogyo Corp | ニッケル・コバルト・マンガン系化合物粒子粉末及びその製造方法、リチウム複合酸化物粒子粉末及びその製造方法並びに非水電解質二次電池 |
| WO2012036127A1 (ja) * | 2010-09-14 | 2012-03-22 | 日立マクセルエナジー株式会社 | 非水二次電池 |
| JP2012182025A (ja) * | 2011-03-01 | 2012-09-20 | Toyota Motor Corp | 二次電池 |
| JP2013094522A (ja) | 2011-11-04 | 2013-05-20 | Seong-Yoon Kim | 歩行補助具の制動装置 |
| JP2014060143A (ja) * | 2012-08-22 | 2014-04-03 | Sony Corp | 正極活物質、正極および電池、並びに電池パック、電子機器、電動車両、蓄電装置および電力システム |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2991152A4 |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2975676A4 (en) * | 2013-03-15 | 2016-04-06 | Nissan Motor | NONAQUEOUS ELECTROLYTE SECONDARY BATTERY CATHODE AND NONAQUEOUS ELECTROLYTE SECONDARY BATTERY USING THE SAME |
| EP2975677A4 (en) * | 2013-03-15 | 2016-04-13 | Nissan Motor | CATHODE FOR SECONDARY BATTERY WITH A WATER-FREE ELECTROLYTE AND SECONDARY BATTERY WITH A WATER-FREE ELECTROLYTE THEREWITH |
| US9608261B2 (en) | 2013-03-15 | 2017-03-28 | Nissan Motor Co., Ltd. | Positive electrode for non-aqueous electrolyte secondary battery and non-aqueous electrolyte secondary battery using the same |
| US9716266B2 (en) | 2013-03-15 | 2017-07-25 | Nissan Motor Co., Ltd. | Positive electrode for non-aqueous electrolyte secondary battery and non-aqueous electrolyte secondary battery using the same |
| JP2016184521A (ja) * | 2015-03-26 | 2016-10-20 | オートモーティブエナジーサプライ株式会社 | 非水電解質二次電池 |
| EP3073555A3 (en) * | 2015-03-26 | 2016-12-14 | Automotive Energy Supply Corporation | Nonaqueous electrolyte secondary battery |
| CN107851838A (zh) * | 2015-08-04 | 2018-03-27 | 汽车能源供应公司 | 锂离子二次电池 |
| US20180241075A1 (en) * | 2015-08-04 | 2018-08-23 | Automotive Energy Supply Corporation | Lithium-Ion Secondary Cell |
| CN107851838B (zh) * | 2015-08-04 | 2021-01-22 | 远景Aesc日本有限公司 | 锂离子二次电池 |
| US20190355969A1 (en) * | 2016-06-08 | 2019-11-21 | Nissan Motor Co., Ltd. | Non-Aqueous Electrolyte Secondary Battery |
| JP2022141208A (ja) * | 2021-03-15 | 2022-09-29 | プライムプラネットエナジー&ソリューションズ株式会社 | 正極および当該正極を備える非水電解質二次電池 |
| JP7320012B2 (ja) | 2021-03-15 | 2023-08-02 | プライムプラネットエナジー&ソリューションズ株式会社 | 正極および当該正極を備える非水電解質二次電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN105144458B (zh) | 2017-08-29 |
| EP2991152A4 (en) | 2016-05-11 |
| US20190252675A1 (en) | 2019-08-15 |
| US20160079589A1 (en) | 2016-03-17 |
| EP2991152A1 (en) | 2016-03-02 |
| KR20150135450A (ko) | 2015-12-02 |
| KR101798313B1 (ko) | 2017-11-15 |
| CN105144458A (zh) | 2015-12-09 |
| EP2991152B1 (en) | 2018-08-29 |
| JPWO2014175355A1 (ja) | 2017-02-23 |
| JP6036999B2 (ja) | 2016-11-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6036999B2 (ja) | 非水電解質二次電池 | |
| JP5967287B2 (ja) | 正極活物質、正極材料、正極および非水電解質二次電池 | |
| JP6070824B2 (ja) | 非水電解質二次電池用正極およびこれを用いた非水電解質二次電池 | |
| JP6229709B2 (ja) | 正極活物質、正極材料、正極および非水電解質二次電池 | |
| JP6008041B2 (ja) | 正極活物質、正極材料、正極および非水電解質二次電池 | |
| JP6075440B2 (ja) | 正極活物質、正極材料、正極および非水電解質二次電池 | |
| JP6156491B2 (ja) | 非水電解質二次電池 | |
| JP6176317B2 (ja) | 非水電解質二次電池用正極およびこれを用いた非水電解質二次電池 | |
| JP6112204B2 (ja) | 非水電解質二次電池用正極およびこれを用いた非水電解質二次電池 | |
| JP6070822B2 (ja) | 非水電解質二次電池 | |
| JP6070823B2 (ja) | 非水電解質二次電池 | |
| WO2015156400A1 (ja) | 電気デバイス用正極、およびこれを用いた電気デバイス | |
| JP6241543B2 (ja) | 電気デバイス | |
| JP6414229B2 (ja) | 電気デバイス | |
| WO2016103511A1 (ja) | 電気デバイス | |
| JP2014216264A (ja) | 非水電解質二次電池用正極活物質、非水電解質二次電池用正極および非水電解質二次電池 | |
| JP2017073281A (ja) | 非水電解質二次電池用正極材料、並びにこれを用いた非水電解質二次電池用正極および非水電解質二次電池 | |
| JP2016225185A (ja) | 非水電解質二次電池用正極およびこれを用いた非水電解質二次電池 | |
| JP2017004696A (ja) | 非水電解質二次電池用正極 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480023723.1 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14789007 Country of ref document: EP Kind code of ref document: A1 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| ENP | Entry into the national phase |
Ref document number: 2015513817 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20157030582 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14786934 Country of ref document: US Ref document number: 2014789007 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |