WO2014129495A1 - 眼疾患処置薬 - Google Patents
眼疾患処置薬 Download PDFInfo
- Publication number
- WO2014129495A1 WO2014129495A1 PCT/JP2014/053898 JP2014053898W WO2014129495A1 WO 2014129495 A1 WO2014129495 A1 WO 2014129495A1 JP 2014053898 W JP2014053898 W JP 2014053898W WO 2014129495 A1 WO2014129495 A1 WO 2014129495A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- age
- alkyl
- macular degeneration
- related macular
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/655—Azo (—N=N—), diazo (=N2), azoxy (>N—O—N< or N(=O)—N<), azido (—N3) or diazoamino (—N=N—N<) compounds
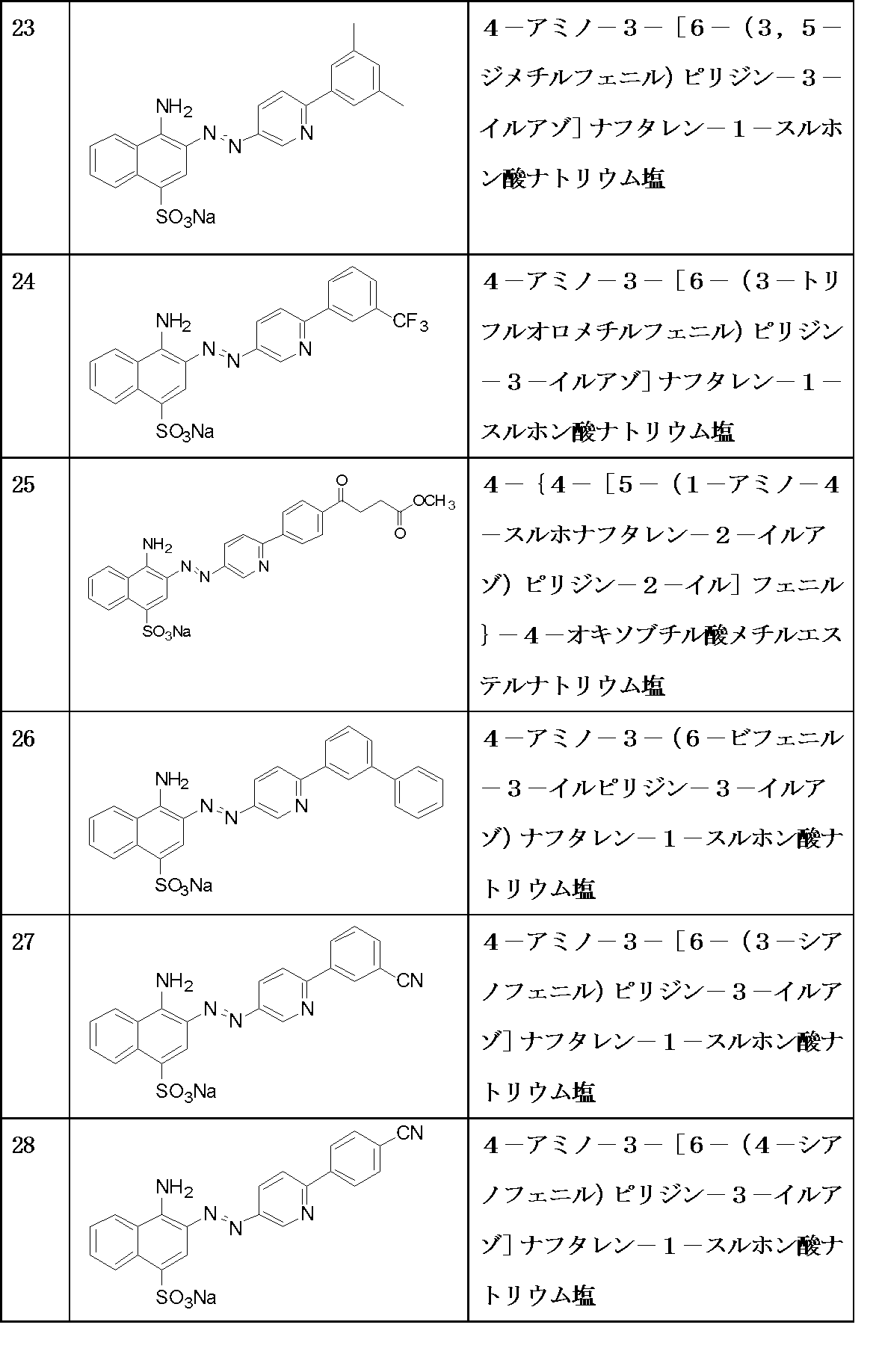
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
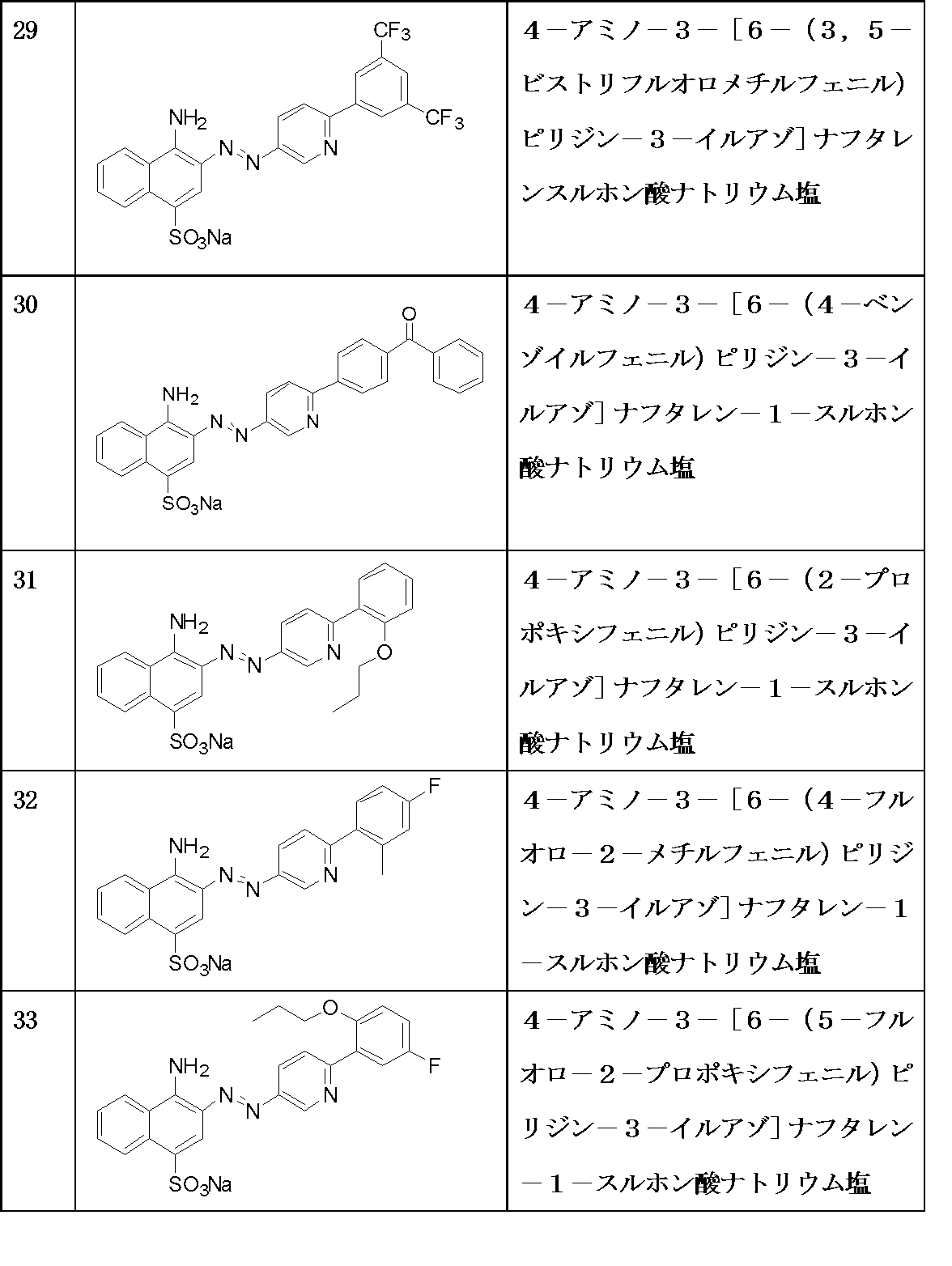
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4418—Non condensed pyridines; Hydrogenated derivatives thereof having a carbocyclic group directly attached to the heterocyclic ring, e.g. cyproheptadine
-
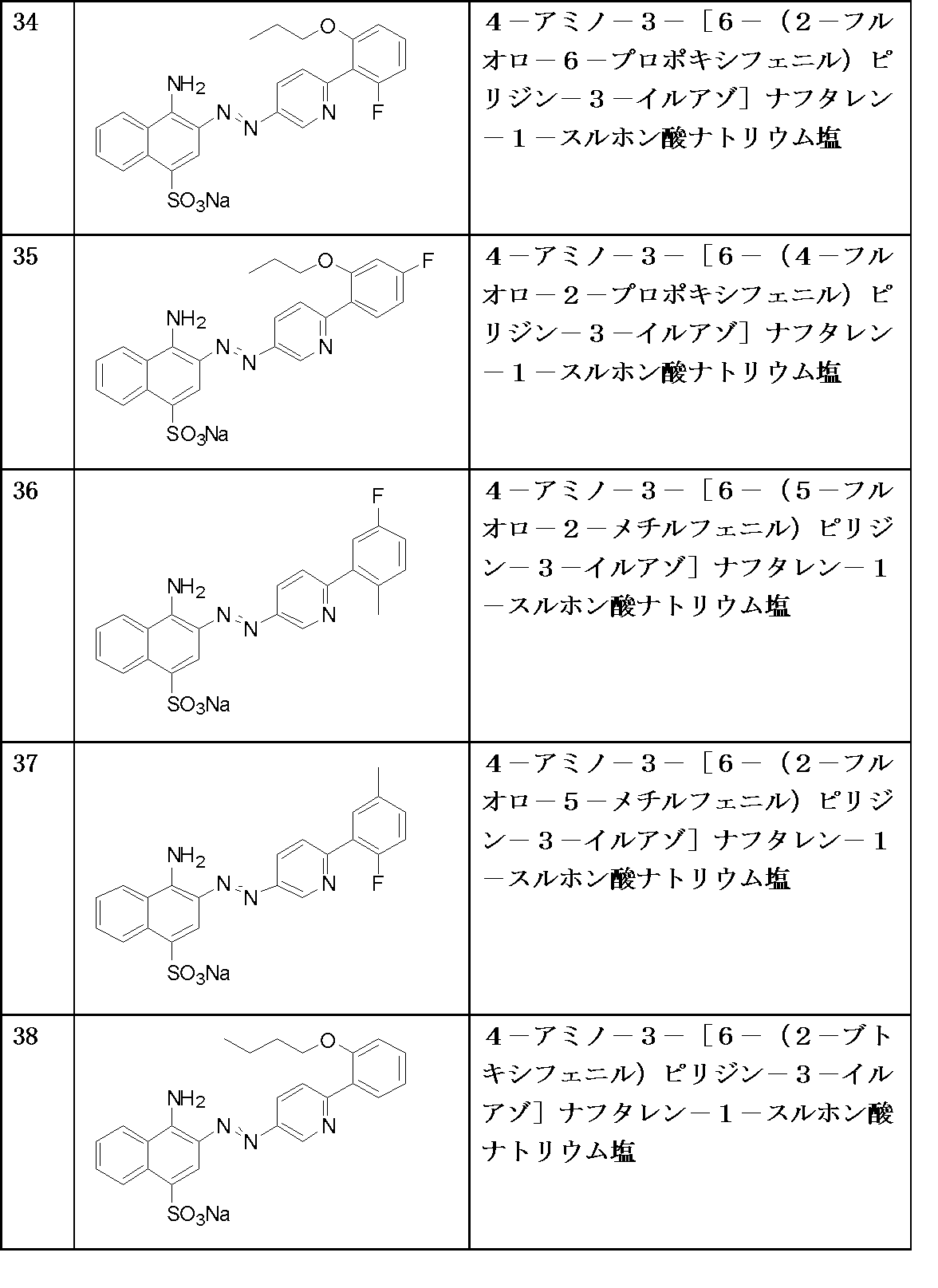
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
- A61P39/02—Antidotes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
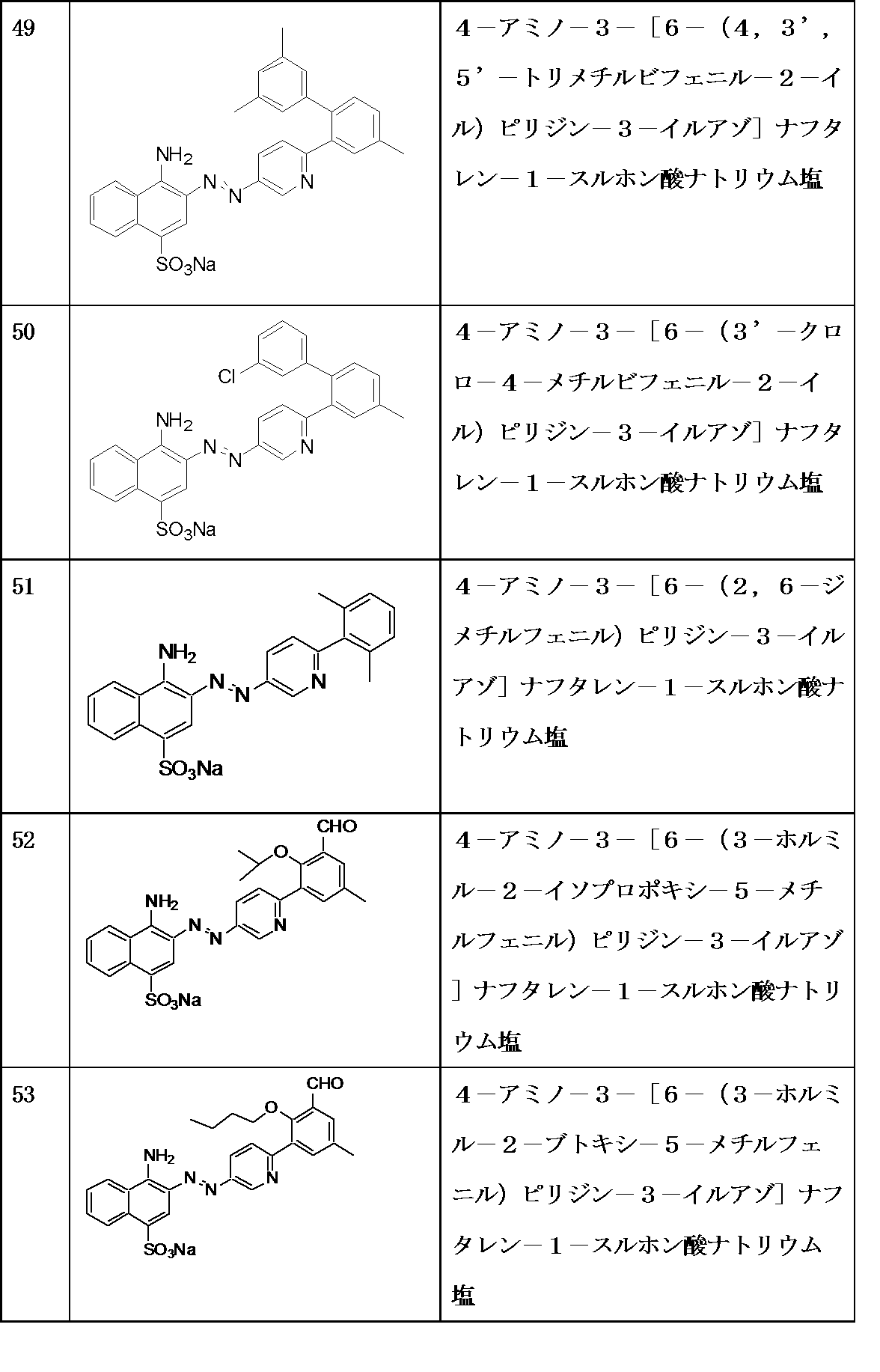
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/72—Nitrogen atoms
- C07D213/76—Nitrogen atoms to which a second hetero atom is attached
- C07D213/77—Hydrazine radicals
Definitions
- the present invention relates to a drug effective for the treatment of an eye disease, a pharmaceutical composition containing the drug, a method for producing a medicament for the treatment of an eye disease including the use of the drug, and the drug in the production of a medicament for the treatment of an eye disease And a method of treating an ophthalmic disease comprising administering the drug or pharmaceutical composition.
- the eye diseases treated according to the invention include in particular age-related macular degeneration, in particular wet age-related macular degeneration or atrophic age-related macular degeneration.
- Age-related macular degeneration is an eye disease that is difficult to see in the center and has symptoms such as distortion, and occupies the top cause of blindness in elderly people in developed countries. In the United States, about 15% of elderly people over 80 years old suffer from age-related macular degeneration in 2000. In Japan as well, the number of patients with age-related macular degeneration has increased in recent years with the aging of society and the westernization of food culture, which is the fourth cause of blindness.
- drusen which is a waste product mainly composed of lipids, accumulates under the retinal pigment. Thereafter, in wet age-related macular degeneration, subchoroidal neovascularization is formed, resulting in decreased visual acuity due to subretinal hemorrhage, retinal edema, and the like.
- the only effective treatment method is vascular regression treatment for the formed new blood vessels (intravitreal administration of anti-VEGF drug, photodynamic therapy), but once the new blood vessels are formed, such treatment is not possible. Even if it goes, the visual prognosis is poor. In order to maintain good visual acuity, it is important to perform progression-suppressing treatment during the drusen formation period, but at present there is no such treatment method.
- atrophic age-related macular degeneration after the drusen accumulation, the retina choroids gradually atrophy without neovascularization, resulting in decreased visual acuity. There is currently no effective treatment for atrophic age-related macular degeneration. There may be a transition from wet age-related macular degeneration to atrophic age-related macular degeneration.
- An object of the present invention is to provide a medicament capable of removing drusen, which is a precursor lesion of age-related macular degeneration, or suppressing the formation of drusen.
- the object of the present invention is also to provide a medicament capable of treating and / or preventing age-related macular degeneration, in particular wet age-related macular degeneration or atrophic age-related macular degeneration.
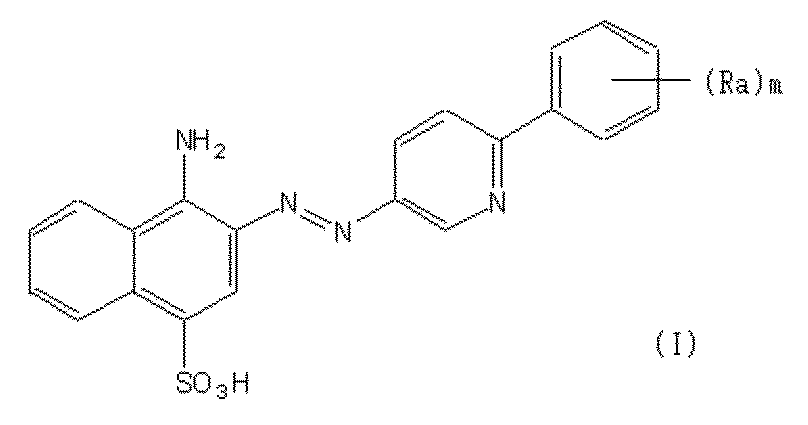
- the present invention provides a compound of formula (I) for removal and / or inhibition of drusen and / or treatment and / or prevention of age-related macular degeneration:
- Each Ra is independently halo, hydroxy, alkyl, halo substituted alkyl, aryl, halo or alkyl substituted aryl, alkoxy, hydroxy or carboxy substituted alkoxy, aryloxy, halo or alkyl substituted aryloxy, CHO, C (O) — Selected from the group consisting of alkyl, C (O) -aryl, C (O) -alkyl-carboxyl, C (O) -alkylene-carboxyester and cyano; m is an integer selected from 0 to 4] Or an oxide, ester, prodrug, pharmaceutically acceptable salt or solvate thereof (hereinafter referred to as the compound of the present invention).
- the present invention is directed to drusen removal and / or inhibition of formation comprising a compound of formula (I) or an oxide, ester, prodrug, pharmaceutically acceptable salt or solvate thereof, and / or Alternatively, a pharmaceutical composition for the treatment and / or prevention of age-related macular degeneration is provided.
- the present invention provides a compound of formula (I) as defined above for the manufacture of a pharmaceutical composition for the removal and / or inhibition of drusen and / or the treatment and / or prevention of age-related macular degeneration. Or the use of oxides, esters, prodrugs, pharmaceutically acceptable salts or solvates thereof.
- the present invention provides a method for the removal and / or inhibition of drusen and / or the treatment and / or prevention of age-related macular degeneration comprising a therapeutically effective amount of the above in a subject in need thereof
- a method comprising administering a compound of formula (I) or an oxide, ester, prodrug, pharmaceutically acceptable salt or solvate thereof.
- the age-related macular degeneration is wet age-related macular degeneration or atrophic age-related macular degeneration, in particular atrophic age-related macular degeneration.
- the compound of formula (I) or an oxide, ester, prodrug, pharmaceutically acceptable salt or solvate thereof is administered orally.
- the compound of the present invention can remove drusen, which is a precursor lesion of age-related macular degeneration, or suppress the formation of drusen. Therefore, according to the present invention, it is possible to provide a drug with a new mechanism of action that has never existed for the treatment and / or prevention of age-related macular degeneration.
- the compounds of the present invention can be used for the treatment of atrophic age-related macular degeneration for which no effective therapeutic method has been established.
- Experiment 1 An outline of Experiment 1 is shown. It is a fundus photograph of 9 month old and 15 month old mice of Experiment 1. Upper: control mice, lower: mice treated with compound 32. In the fundus photograph of the upper part of FIG. 2 (control mouse), drusen is indicated by an arrow, and an area where drusen increase is recognized is surrounded by a curve. In the fundus photograph of the lower part of FIG. 2 (compound 32-administered mouse), the area where drusen disappears is observed is surrounded by a curve. 3 is a graph showing the course of drusen numbers in the compound 32 administration group and the control group of Experiment 1.
- ns indicates no significant difference.
- the transition of drusen number in mouse individuals 1 to 4 to which compound 32 of Experiment 1 was administered is shown.
- An outline of Experiment 2 is shown.
- mice of Experiment 2 3.5, 6 and 9 months old.
- Upper and middle control mice
- mice treated with compound 32 In the fundus photographs at 3.5 and 9 months of age in the middle of FIG. 9 (control mice), drusen is indicated by an arrow, and a region where drusen increase is observed is surrounded by a curve.
- the area where drusen disappears is observed is surrounded by a curve. It is a graph which shows progress of the drusen number of the compound 32 administration group of experiment 2, and a control group.
- Alkyl means a monovalent saturated aliphatic hydrocarbyl group having 1 to 10 carbon atoms, preferably 1 to 6 carbon atoms.
- C xy alkyl means an alkyl group having x to y carbons.
- Alkyl is, for example, straight and branched chain hydrocarbyl groups such as methyl (CH 3 —), ethyl (CH 3 CH 2 —), n-propyl (CH 3 CH 2 CH 2 —), isopropyl ((CH 3 ) 2 CH-), n-butyl (CH 3 CH 2 CH 2 CH 2- ), isobutyl ((CH 3 ) 2 CHCH 2- ), sec-butyl ((CH 3 ) (CH 3 CH 2 ) CH-), t -Butyl ((CH 3 ) 3 C-), n-pentyl (CH 3 CH 2 CH 2 CH 2- ) and neopentyl ((CH 3 ) 3 CCH 2- ) are meant, but not limited to.
- substituted means that one or more hydrogen atoms of the group are replaced by the same or different designated substituents.
- Alkylene means a divalent saturated aliphatic hydrocarbyl group having 1 to 10 carbon atoms, preferably 1 to 6 carbon atoms.
- C xy alkylene means an alkylene having x to y carbons.
- Alkylidene and alkylene groups include branched and straight chain hydrocarbyl groups.
- Alkoxy means a group of —O-alkyl where alkyl is defined herein. Alkoxy includes, for example, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, t-butoxy, sec-butoxy and n-pentoxy.
- Aryl or “Ar” means a monovalent aromatic carbocyclic group of 6 to 14 carbon atoms having one ring (eg, phenyl) or multiple condensed rings (eg, naphthyl or anthryl). Aryl groups typically include phenyl and naphthyl. “Aryloxy” refers to the group —O-aryl where aryl is defined herein and includes, for example, phenoxy and naphthoxy.
- Cyano or “carbonitrile” refers to the group —CN.
- Carboxyl or “carboxy” means —COOH or a salt thereof.
- Carboxyl ester or “carboxy ester” refers to the group —C (O) O-alkyl, where alkyl is defined herein.
- Halo or “halogen” means fluoro, chloro, bromo and iodo.
- Hydroxy or “hydroxyl” refers to the group —OH.
- substituents not explicitly defined in this specification is performed by naming the terminal portion of the functional group and then naming the adjacent functional group toward the point of attachment.
- substituent “arylalkyloxycarbonyl” refers to (aryl)-(alkyl) -O—C (O) —.
- Compound and “compound (s)” as used herein refer to compounds included in Formula (I), as described herein, and specific compounds of Formula (I), and their It means an oxide, ester, prodrug, pharmaceutically acceptable salt or solvate. The term further includes stereoisomers and tautomers of compounds or groups of compounds.
- Solvate or “solvate” of a compound means a compound as defined above combined with a stoichiometric or non-stoichiometric amount of solvent.
- Solvates include solvates of oxides, esters, prodrugs or pharmaceutically acceptable salts of the compounds of formula (I).
- the solvent is volatile, non-toxic and / or acceptable for administration to humans in trace amounts. Suitable solvates include water.
- Stereoisomer or “stereoisomers” refer to compounds that differ in the chirality of one or more stereocenters. Stereoisomers include enantiomers and diastereomers.
- tautomeric forms of heteroaryl groups are meant, for example pyrazole, imidazole, benzimidazole, triazole and tetrazole.
- “Pharmaceutically acceptable salt” means pharmaceutically acceptable salts derived from a variety of organic and inorganic counterions well known in the art, such as sodium, potassium, calcium, magnesium, ammonium and tetraalkyl. Salts with ammonium, as well as salts with organic or inorganic acids such as hydrochloric acid, hydrobromic acid, tartaric acid, mesylic acid, acetic acid, maleic acid and oxalic acid.
- a pharmaceutically acceptable salt of a compound means a pharmaceutically acceptable salt, including a salt of an oxide, ester or prodrug of a compound of formula (I).
- the term “pharmaceutically acceptable salt” includes non-toxic acid or alkaline earth metal salts of compounds of formula (I). These salts can be prepared in situ during the final isolation and purification of the compound of formula (I) or by reacting the base or acid functionality separately with a suitable organic or inorganic acid or base, respectively.
- Typical salts are: acetate, adipate, alginate, citrate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, camphorate, camphorsulfonate, Digluconate, cyclopentanepropionate, dodecyl sulfate, ethane sulfonate, glucoheptanoate, glycerophosphate, hemisulfate, heptanoate, hexanoate, fumarate, hydrochloride, hydrogen bromide Acid salt, hydroiodide salt, 2-hydroxyethanesulfonate, lactate, maleate, methanesulfonate, nicotinate, 2-naphthalenesulfonate, oxalate, pamoate, pectinic acid Salt, persulfate, 3-phenylpropionate, picrate, pivalate, propionate, succinate, sulf
- Basic nitrogen-containing groups also include alkyl halides such as methyl, ethyl, propyl and butyl chlorides, bromides and iodides; dialkyl sulfates such as dimethyl, diethyl, dibutyl and diamyl sulfate, decyl, lauryl and myristyl. And quaternized with reactants such as long chain halides such as stearyl chloride, bromide and iodide, aralkyl halides such as benzyl and phenethyl chloride and others. Thereby a product is obtained which dissolves or disperses in water or oil.
- alkyl halides such as methyl, ethyl, propyl and butyl chlorides, bromides and iodides
- dialkyl sulfates such as dimethyl, diethyl, dibutyl and diamyl sulfate, decyl, lauryl and
- Base addition salts may be used in situ during the final isolation and purification of the compound of formula (I), or with a carboxylic acid group and a suitable base such as a pharmaceutically acceptable metal cation hydroxide, carbonate or bicarbonate or ammonia, Alternatively, it can be produced by separately reacting with an organic primary, secondary or tertiary amine.
- Pharmaceutically acceptable salts include alkali and alkaline earth metal cations, such as sodium, lithium, potassium, calcium, magnesium, aluminum salts, and the like, as well as non-toxic ammonium, quaternary ammonium and amine cations, such as limited Not including, but limited to, ammonium, tetramethylammonium, tetraethylammonium, methylamine, dimethylamine, trimethylamine, triethylamine, ethylamine, and the like.
- Other representative organic amines useful for the formation of base addition salts include diethylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine and the like.
- ester means an ester that is hydrolyzed in vivo, including those that are readily degraded in the human body to release the parent compound or a salt thereof.
- Suitable ester groups are, for example, those derived from pharmaceutically acceptable aliphatic carboxylic acids, in particular alkanoic acids, alkenoic acids, cycloalkanoic acids and alkanedioic acids, where each alkyl or alkenyl group is preferably 6 Having no more than carbon atoms).
- Specific examples of esters include formate, acetate, propionate, butyrate, acrylic acid and ethyl succinate.
- prodrug is within the scope of reasonable medical judgment and includes human or animal tissue without undue toxicity, irritation, allergic response, etc. Means a prodrug of a compound suitable for use in contact, balanced at a reasonable benefit / risk ratio and effective for the intended use, and possibly a zwitterionic form of the compound of the invention .
- prodrug means a compound that is rapidly transformed in vivo to yield the parent compound of the above formula, for example by hydrolysis in blood.
- General explanation is T. Higuchi and V. Stella, Pro drugs as Novel Delivery Systems, Vol. 14 of the ACS Symposium Series and Edward B. Roche, ed., Bioreversible Carriers in Drug Design, AmericanociPharmaceutical sga 1987 (both incorporated herein by reference).
- the compounds of formula (I) or any pharmaceutically acceptable salt, ester, oxide and prodrug thereof may be processed in vivo by metabolism in the human or animal body or cells to yield metabolites.
- the term “metabolite” as used herein means a derivative of any formula that is produced in a subject after administration of the parent compound. Derivatives may be produced from the parent compound by biochemical transformations such as oxidation, reduction, hydrolysis or conjugation in a variety of subjects, including, for example, oxide and demethylated derivatives. Metabolites of the compounds of the present invention can be identified using routine techniques known in the art. For example, Bertolini, G. et al., J. Med. Chem.
- Subject means mammals, including human and non-human mammals.
- prevent or “prevention” as used with respect to a subject disease means prevention of the onset of a disease in a subject who is susceptible or who has not yet shown symptoms of the disease.
- treating or “treatment” as used with respect to a subject disease means 1) prevention of disease or cessation of its progression; or 2) occurrence of disease remission or regression.
- the compounds of the invention have the compounds of formula (I) or an oxide, ester thereof, wherein Ra is independently selected from the group consisting of halo, hydroxy, alkyl, halo-substituted alkyl, and alkoxy: Prodrug, pharmaceutically acceptable salt or solvate.
- the compounds of the invention have the compounds of formula (I) or an oxide, ester, prodrug, pharmaceutically acceptable salt thereof, wherein Ra is independently selected from the group consisting of halo and alkyl, Salt or solvate.
- the compounds of the invention have the compounds of formula (I) or an oxide, ester, prodrug, pharmaceutically acceptable salt thereof, wherein two Ra are present, one is halo and the other is alkyl. Salt or solvate.
- the compound of the invention is a compound selected from compounds 1 to 53 listed in Table 1 below, or an oxide, ester, prodrug, pharmaceutically acceptable salt or solvate thereof.
- the compounds of the invention have the formula Or an oxide, ester, prodrug, pharmaceutically acceptable salt or solvate thereof, particularly a sodium salt thereof.
- the compounds of formula (I) and any pharmaceutically acceptable salts, esters, oxides and prodrugs thereof may contain asymmetrically substituted carbon atoms.
- Such asymmetrically substituted carbon atoms can result in compounds that exist in other stereoisomeric forms that can be defined as absolute stereochemistry, such as enantiomers, diastereomers, and (R)-or (S) -forms.
- absolute stereochemistry such as enantiomers, diastereomers, and (R)-or (S) -forms.
- S racemic mixtures (or racemates), mixtures of diastereomers as well as one diastereoisomer.
- Stereomers are intended.
- S and “R” configurations, as used herein, are as defined by IUPAC 1974 RECOMMENDATIONS FOR SECTION E, FUNDAMENTAL StereoCHEMISTRY, Pure Appl. Chem. 45: 13-30 (1976).
- Age-related macular degeneration is divided into wet and atrophic types.
- the exudative type is a disease characterized by choroidal neovascularization (CNV) generated by changes in the retinal pigment epithelial cells, Bruch's membrane, and choroid in the macula, and its proliferative changes.
- CNV choroidal neovascularization
- CNV grows under the retinal pigment subepithelium and then under the retina. It exhibits bleeding, exudation of retinal pigment epithelium, and retinal detachment. After they are absorbed, atrophy and scarring are formed, resulting in a high degree of permanent vision loss, and the progression is rapid.
- atrophic type a map-like atrophy lesion of retinal pigment epithelium-choriocapillary plate is formed in the macular region, but progress is slow.
- Retinal pigment epithelial cells play an important role in maintaining the neuroretinal environment, including the phagocytic function of the outer segment of photoreceptor cells, but as age-related changes, the accumulation of lipofuscin as digestive residues and retinal pigments Epithelial cells become lipidated and an abnormal structure called drusen is formed.
- the Bruch's subepithelial membrane thickens with age, and the physiological environment between the photoreceptor cells, the retinal pigment epithelium, and the Bruch's membrane changes.
- VEGF vascular endothelial growth factor
- the compound of formula (I) of the present invention eliminates drusen or suppresses the formation of drusen, thereby causing atrophic and exudative age-related macular degeneration with a mechanism of action different from that of vascular regression treatment. Suppress progress.
- the compound of formula (I) of the present invention can treat atrophic and exudative age-related macular degeneration with a mechanism of action different from that of vascular regression treatment. It is not intended to be excluded.
- Vascular regression treatment that can be used in combination with the compounds of formula (I) of the present invention includes, but is not limited to, intravitreal administration of ranibizumab, pegaptanib and aflibercept, and photodynamic therapy.
- a pharmaceutically acceptable carrier suitable for administration to a human or animal subject comprising at least one compound of formula (I), alone or with an additional agent.
- a pharmaceutical composition for removing or inhibiting drusen and / or for treating and / or preventing age-related macular degeneration is provided.
- a compound of formula (I) for the removal or inhibition of drusen and / or the treatment and / or prevention of age-related macular degeneration there is provided.
- a compound of formula (I) for the removal or inhibition of drusen and / or the treatment and / or prevention of age-related macular degeneration in a further embodiment of the invention, the use of a compound of formula (I) in the manufacture of a pharmaceutical composition for the removal or inhibition of drusen and / or the treatment and / or prevention of age-related macular degeneration.
- the compounds of this invention are administered in therapeutically effective amounts and in any acceptable dosage form for agents that provide similar utilities.
- the amount of actual compound, i.e., active ingredient will depend on many factors, such as the severity of the disease being treated, the age and relative health of the subject, the ability of the compound to be used, the route and form of administration and other factors.
- the compounds of the present invention can be administered one or more times per day, preferably 3 or 4 times per day. All these factors are within the skill of the attending clinician.
- the amount of active ingredient that can be combined with a carrier material to produce a single dosage form will vary depending upon the host treated and the particular mode of administration.
- the specific dose level for any specific subject is the activity, age, weight, general health, sex, diet, time of administration, route of administration, excretion rate of the specific compound used, It is understood that it is based on a variety of factors including the combination of drugs and the severity of the specific disease being treated.
- the therapeutically effective amount in a given situation can be readily determined by routine experimentation and is within the skill and judgment of the ordinary clinician.
- a therapeutically effective amount is a total daily dose administered to the host in a single or divided dose, for example, from about 0.001 to about 1000 mg / kg body weight / day, or from about 1.0 to about 30 mg / kg body weight. / Day amount.
- a unit dosage composition may contain a submultiple amount to constitute a daily dose.
- Suitable pharmaceutically acceptable carriers or diluents include, for example, processing agents and drug delivery modifiers and accelerators such as calcium phosphate, magnesium stearate, talc, monosaccharides, disaccharides, starch, gelatin, cellulose, methylcellulose, carboxy Includes sodium methylcellulose, dextrose, hydroxypropyl- ⁇ -cyclodextrin, polyvinylpyrrolidinone, low melting point wax, ion exchange resin, and the like, as well as combinations of one or more thereof.
- processing agents and drug delivery modifiers and accelerators such as calcium phosphate, magnesium stearate, talc, monosaccharides, disaccharides, starch, gelatin, cellulose, methylcellulose, carboxy Includes sodium methylcellulose, dextrose, hydroxypropyl- ⁇ -cyclodextrin, polyvinylpyrrolidinone, low melting point wax, ion exchange resin, and the like, as well as combinations of one or more thereof.
- Liquid and semi-solid excipients may be selected from glycerol, propylene glycol, water, ethanol and various oils such as those of petroleum, animal, vegetable or synthetic origin, such as peanut oil, soybean oil, mineral oil, sesame oil, etc. it can.
- liquid carriers particularly for injectable solutions, include water, saline, aqueous dextrose and glycol.
- suitable pharmaceutically acceptable excipients are described in “Remington's Pharmaceuticals Sciences,” “Mack, Pub., Co.,” New, Jersey, (1991).
- the choice of formulation is based on a variety of factors such as drug dosage form and bioavailability of the drug substance.
- the drug can be administered as a pharmaceutical composition by any one or a combination of two or more of the following routes: oral, systemic (eg, transdermally, intranasally or via suppositories), topical (eye drops, intravitreal, Subconjunctival, intratenon or transdermal) or parenteral administration, preferably oral, ophthalmic, intravitreal, subconjunctival or intratenon.
- An exemplary method of administration is oral using a convenient daily dosing regimen that can be adjusted according to the degree of disease.
- composition can take the form of a tablet, pill, capsule, semi-solid, powder, sustained release formulation, solution, suspension, elixir, aerosol or any other suitable composition.
- Another method of administration is inhalation, such as delivering the therapeutic agent directly to the respiratory tract (see US Pat. No. 5,607,915).
- Topical administration also includes the use of transdermal administration such as transdermal patches or iontophoresis devices.
- parenteral as used herein includes subcutaneous injections, intravenous, intraperitoneal, intramuscular, intrasternal injection or infusion techniques.
- sterile injectable aqueous or oleaginous suspensions can be formulated according to the known art using suitable dispersing or wetting agents or suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally acceptable diluent or solvent, for example as a solution in 1,3-propanediol.
- a non-toxic parenterally acceptable diluent or solvent for example as a solution in 1,3-propanediol.
- acceptable vehicles and solvents that can be employed are water, Ringer's solution and isotonic sodium chloride solution.
- sterile fixed oils are conveniently used as a solvent or dispersion medium.
- any grade of fixed oil can be used including synthetic mono- or diglycerides.
- fatty acids such as oleic acid can be used in injectable preparations.
- Suppositories for rectal administration of the drug are suitable non-irritating excipients such as cocoa butter and polyethylene that are solid at room temperature but liquid at the rectal temperature and thus melt at the rectum to release the drug. It can be produced by mixing glycol.
- Solid dosage forms for oral administration can include capsules, tablets, pills, powders and granules.
- the active compound may be admixed with at least one inert diluent such as sucrose, lactose or starch.
- Such dosage forms may also contain additives other than inert diluents, such as lubricants such as magnesium stearate, as is commonly practiced.
- the dosage form may contain a buffering agent. Tablets and pills may additionally be manufactured with enteric coatings.
- Liquid dosage forms for oral administration may include pharmaceutically acceptable emulsions, solutions, suspensions, syrups and elixirs containing inert diluents commonly used in the art, such as water. .
- Such compositions may also contain adjuvants such as wetting agents, emulsifying and suspending agents, cyclodextrins and sweetening, flavoring and perfuming agents.
- liposomes are generally derived from phospholipids or other lipid substances. Liposomes are formed by mono- or multilamellar hydrated liquid crystals that are dispersed in an aqueous medium. Any non-toxic, physiologically acceptable and metabolizable lipid capable of forming liposomes can be used.
- the present composition in liposome form may contain stabilizers, preservatives, excipients and the like. Examples of lipids are natural and synthetic phospholipids and phosphatidylcholines (lecithins). Methods for forming liposomes are known in the art. See, for example, Prescott, Ed., Methods Cell Biology, Volume XIV, Academic Press, New York, N.W., p. 33 et seq. (1976).
- Compressed gas can be used to disperse the compound of the present invention in aerosol form.
- Inert gases suitable for this purpose are nitrogen, carbon dioxide and the like.
- Other suitable pharmaceutical excipients and their formulations are described in Remington's Pharmaceutical Sciences, edited by E. W, Martin (Mack Publishing Company, 18th Ed., 1990).
- the compounds of the invention can be formulated as solutions, suspensions, aerosol propellants or dry powders and loaded into a suitable dispenser for administration.
- suitable dispenser for administration There are various types of pharmaceutical inhalation devices, nebulizers, inhalers, fixed dose inhalers (MDI) and dry powder inhalers (DPI).
- MDI fixed dose inhalers
- DPI dry powder inhalers
- the nebulizer device creates a high velocity air stream that sprays the therapeutic agent (which is formulated in liquid form) as a mist that is carried into the subject's respiratory tract.
- MDI is typically a formulation packaged in compressed gas. When activated, the device releases a predetermined amount of therapeutic agent by compressed gas and is therefore a reliable method of administering a fixed amount of compound.
- DPI releases a therapeutic agent in the form of a free flowing powder that can be dispersed by the device in the respiratory airflow of the subject.
- the therapeutic agent is formulated with an excipient such as lactose.
- a certain amount of therapeutic agent is stored in capsule form and is released with each movement.
- Eye drops include isotonic agents such as sodium chloride and concentrated glycerin; buffering agents such as sodium phosphate and sodium acetate; surface activity such as polyoxyethylene sorbitan monooleate, polyoxyl 40 stearate, polyoxyethylene hydrogenated castor oil Agents; Stabilizers such as sodium citrate and sodium edetate; can be selected and used as needed from preservatives such as benzalkonium chloride and paraben, and pH is acceptable for ophthalmic preparations Although it may be within the range, it is usually within the range of 4-8.
- the eye ointment can be prepared using a commonly used base such as white petrolatum or liquid paraffin.
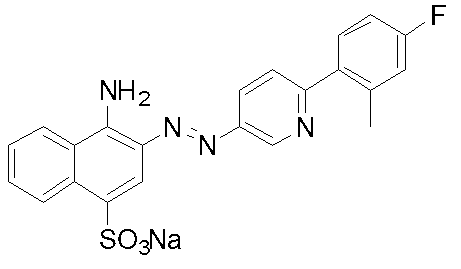
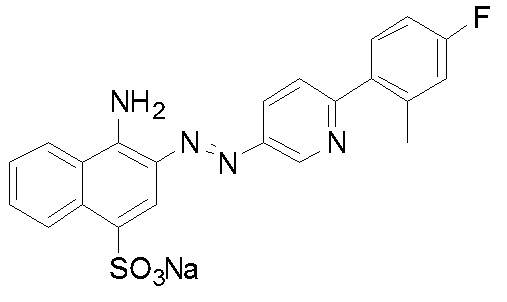
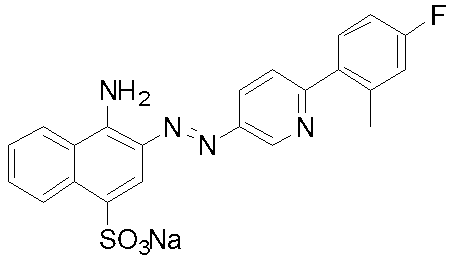
- Example 1 Compound 32: Synthesis of 4-amino-3- [6- (4-fluoro-2-methylphenyl) pyridin-3-ylazo] naphthalene-1-sulfonic acid sodium salt (I) 2- (4-Fluoro-2-methylphenyl) -5-nitropyridine 2-chloro-5-nitropyridine (5.0 g, 31.5 mmol), tetrakis (triphenylphosphine) palladium (0.35 g, 0.3 mmol) was added to 1,2-dimethoxyethane (50 ml), and the mixture was degassed and purged with nitrogen three times under reduced pressure.
- Example 2 Removal of drusen in aged mice
- CCR2 monocyte chemotactic activator (MCP-1) receptor
- MCP-1 monocyte chemotactic activator
- n 30 eyes
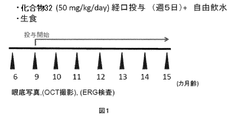
- compound 32 dissolved in physiological saline was orally administered at a dose of 50 mg / kg body weight / day for 5 days a week from 9 months of age.
- OCT optical coherence tomography
- ERG electroretinogram
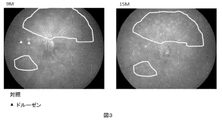
- FIG. 2 to 4 are fundus photographs of 9 months and 15 months of age.
- the white granular structure is drusen.
- an increase in drusen is observed in the area surrounded by the curve in FIG.
- mice administered with compound 32 drusen disappeared in the area surrounded by the curve in FIG.
- FIG. 5 is a graph obtained by counting drusen confirmed by fundus photographs. The graph shows that drusen increased over time in the control, but not in the mice that received compound 32.
- FIG. 6 shows the change in the number of drusen in mice administered with Compound 32. It can be seen that the drusen number increased until 9 months of administration, but gradually decreased after administration.
- FIG. 7 shows b-wave magnitudes in the electroretinogram of control mice and mice administered compound 32. This result means that the mouse treated with compound 32 has better retinal function than the control mouse.
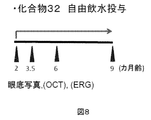
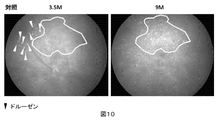
- Example 3 Experiment 2 Inhibition of drusen formation in young mice The effect of the compound of Compound No. 32 (compound 32) on the inhibition of drusen formation in young mice was examined. CCR2-deficient mice were used. From 2 months of age, drinking water containing Compound 32 (0.385 mg / ml) was freely given. The control group was allowed to freely drink drinking water not containing Compound 32. Fundus photographs, OCT and ERG were performed at 2, 3.5, 6 and 9 months of age. An outline of Experiment 2 is shown in FIG.
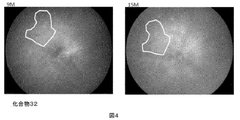
- FIG. 9 to 11 are fundus photographs of 3.5, 6 and 9 months of age.
- the white granular structure is drusen.
- In control mice there is a marked increase in drusen in the area enclosed by the curve in FIG.
- no increase in drusen was observed in the mice to which compound 32 was administered, and drusen formation suppression was particularly remarkably observed in the region surrounded by the curve in FIG.
- FIG. 12 is a graph obtained by counting drusen confirmed by fundus photographs. This graph shows that drusen increased over time in the controls, but not in mice receiving compound 32.
- FIG. 13 shows b-wave magnitudes in the electroretinogram of control mice and mice administered compound 32. This result means that the mouse treated with compound 32 has better retinal function than the control mouse.
- FIG. 14 shows the magnitude of the a wave in the electroretinogram of control mice and mice administered compound 32. This result means that photoreceptor cells function better in control mice than in compound 32 administered mice.
- the compound of the present invention particularly the compound of Synthesis Example 32, has a drusen removal and formation inhibitory effect and is therefore useful for the treatment and prevention of age-related macular degeneration.
- the compounds of the present invention are effective orally and have the advantage of being easily administrable.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Toxicology (AREA)
- Ophthalmology & Optometry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Pyridine Compounds (AREA)
Abstract
Description
Raはそれぞれ独立して、ハロ、ヒドロキシ、アルキル、ハロ置換アルキル、アリール、ハロまたはアルキル置換アリール、アルコキシ、ヒドロキシまたはカルボキシ置換アルコキシ、アリールオキシ、ハロまたはアルキル置換アリールオキシ、CHO、C(O)-アルキル、C(O)-アリール、C(O)-アルキル-カルボキシル、C(O)-アルキレン-カルボキシエステルおよびシアノから成る群から選択され、
mは0~4から選択される整数である〕
の化合物またはそのオキシド、エステル、プロドラッグ、薬学的に許容される塩もしくは溶媒和物(以下、本発明の化合物と称する)を提供する。
さらなる態様において、式(I)の化合物またはそのオキシド、エステル、プロドラッグ、薬学的に許容される塩もしくは溶媒和物は、経口投与される。
特に具体的な定めのない限り、本明細書で使用される用語は、有機化学、医学、薬学、分子生物学、微生物学等の分野における当業者に一般に理解されるとおりの意味を有する。以下にいくつかの本明細書で使用される用語についての定義を記載するが、これらの定義は、本明細書において、一般的な理解に優先する。
「アリールオキシ」は、-O-アリール(ここで、アリールは本明細書に定義されている)の基を意味し、例えばフェノキシおよびナフトキシを含む。
「カルボキシル」または「カルボキシ」は-COOHまたはその塩を意味する。
「カルボキシルエステル」または「カルボキシエステル」は、-C(O)O-アルキル(ここで、アルキルは本明細書に定義されている)の基を意味する。
「ハロ」または「ハロゲン」はフルオロ、クロロ、ブロモおよびヨードを意味する。
「ヒドロキシ」または「ヒドロキシル」は-OHの基を意味する。
加齢黄斑変性は滲出型と萎縮型に分けられる。滲出型は黄斑部の網膜色素上皮細胞-ブルッフ膜-脈絡膜の変化により発生する脈絡膜新生血管(choroidal neovascularization: CNV)とその増殖変化を本態とする疾患である。CNVは網膜色素上皮下、ついで網膜下に発育する。出血、滲出による網膜色素上皮剥離、網膜剥離を呈し、それらが吸収した後には萎縮・瘢痕が形成され、高度の永続する視力低下を生じ、その進行は速い。萎縮型では黄斑部に網膜色素上皮-脈絡毛細血管板の地図状萎縮病巣が形成されるが、進行は緩慢である。
本発明のある実施態様では、少なくとも1種の式(I)の化合物を、単独でまたはさらなる薬剤とともに含み、ヒトまたは動物対象への投与に適した薬学的に許容される担体を含む、ドルーゼンの除去または形成抑制用、および/または、加齢黄斑変性の処置および/または予防用の医薬組成物が提供される。
化合物32:4-アミノ-3-[6-(4-フルオロ-2-メチルフェニル)ピリジン-3-イルアゾ]ナフタレン-1-スルホン酸ナトリウム塩の合成
2-クロロ-5-ニトロピリジン(5.0g、31.5mmol)、テトラキス(トリフェニルホスフィン)パラジウム(0.35g、0.3mmol)を1,2-ジメトキシエタン(50ml)に加え、減圧下で脱気、窒素置換を3回行った。窒素雰囲気下、室温で20分間撹拌後、4-フルオロ-2-メチルフェニルボロン酸(31.5mmol)、2M炭酸ナトリウム水溶液(31.5ml)を注加し、80℃に昇温した。80℃で3時間反応後、室温まで冷却し酢酸エチルと水を加えて抽出した。有機層を無水硫酸ナトリウムで乾燥し、減圧下濃縮した。カラムクロマトグラフィーで精製し、表題化合物を得た。
エタノール(20ml)と水(5ml)を混合し、鉄粉を加え70-80℃に加熱した。塩化アンモニウム(0.1g、2.1mmol)を加え、次いで(i)で得られた2-(4-フルオロ-2-メチルフェニル)-5-ニトロピリジン(10.0mmol)を加え、70-80℃で1時間反応を行った。反応終了後、鉄粉をセライトで熱時ろ過し、ろ液を減圧下濃縮した。残渣をイソプロピルアルコールに溶解し、水を加え晶析・ろ過し、表題化合物を得た。
(ii)で得られた6-(4-フルオロ-2-メチルフェニル)ピリジン-3-イルアミン(58.9mmol)を99%酢酸(50ml)に溶解し、35%塩酸(25g)を加え塩酸塩とした。氷冷下、36%亜硝酸ナトリウム水溶液(12g、62.5mmol)を0-5℃で滴下し、約15分間反応を行った。アミド硫酸を加え、更に5分間反応を行い、ジアゾ液を得た。4-アミノ-1-ナフタレンスルホン酸(13.0g、58.4mmol)を水(130ml)に懸濁し、10%水酸化ナトリウム水溶液でpH8-9に調整した。5-10℃まで冷却し、得られたジアゾ液を5-10℃で滴下した。その際、pH7-9を維持するよう10%水酸化ナトリウム水溶液で調整した。滴下終了後、5-10℃で1時間反応を行い、その後室温まで昇温した。飽和食塩水で塩析し、析出した結晶を吸引ろ過した。カラムクロマトグラフィーで精製し、表題化合物を得た。
1H-NMR δ[ppm] = 9.22(1H, d, J = 2.4Hz), 8.76(1H, d, J = 8.1), 8.49-8.44(2H, m), 8.34(1H, s), 7.82(2H, bs), 7.67-7.47(4H, m), 7.21-7.11(2H, m), 2.41(3H, s)
13C-NMR(DMSO-d6) δ[ppm] = 163.5, 160.3, 158.2, 147.1, 146.7, 145.4, 138.8, 138.7, 136.1, 136.1, 132.4, 132.1, 131.8, 131.7, 129.2, 128.6, 128.3, 127.2, 125.1 , 124.6, 124.2, 124.0, 117.3, 117.1, 116.6, 112.9, 112.6, 20.4. 20.4
実験1:老齢マウスにおけるドルーゼンの除去
比較的老齢のマウスにおいて、化合物番号32の化合物(化合物32)が、既に形成されたドルーゼンを除去する効果について調べた。CCR2(単球走化活性化因子(MCP-1)受容体)欠損マウスを用いた。化合物32投与群(n=30眼)では、生理食塩水に溶解した化合物32を、生後9カ月齢から週に5日、50mg/体重kg/日で経口投与した。対照群(n=48眼)には生理食塩水を経口投与した。マウスを自由に飲水させた。6、9、10、11、12、13、14および15カ月齢で、眼底写真、光干渉断層法(optical coherence tomography、OCT)(Multiline OCT, Heidelberg Engineering)および網膜電図検査(electroretinogram;ERG)を実施した。実験1の概要を図1に示す。
実験2:若年マウスにおけるドルーゼンの形成抑制
若年のマウスにおいて、化合物番号32の化合物(化合物32)が、ドルーゼンの形成を抑制する効果について調べた。CCR2欠損マウスを用いた。生後2カ月齢から、化合物32(0.385mg/ml)を含む飲用水を自由に摂取させた。コントロール群には化合物32を含まない飲用水を自由に摂取させた。2、3.5、6および9カ月齢で、眼底写真、OCTおよびERGを実施した。実験2の概要を図8に示す。
Claims (11)
- ドルーゼンの除去および/または形成抑制用、および/または、加齢黄斑変性の処置および/または予防用の、式(I):
Raはハロ、ヒドロキシ、アルキル、ハロ置換アルキル、アリール、ハロまたはアルキル置換アリール、アルコキシ、ヒドロキシまたはカルボキシ置換アルコキシ、アリールオキシ、ハロまたはアルキル置換アリールオキシ、CHO、C(O)-アルキル、C(O)-アリール、C(O)-アルキル-カルボキシル、C(O)-アルキレン-カルボキシエステルおよびシアノから成る群から選択され、
mは0~4から選択される整数である〕
の化合物またはそのオキシド、エステル、薬学的に許容される塩もしくは溶媒和物を含む医薬組成物。 - Raが、それぞれ独立して、ハロ、ヒドロキシ、アルキル、ハロ置換アルキルおよびアルコキシから成る群から選択される、請求項1に記載の医薬組成物。
- Raが、それぞれ独立して、ハロおよびアルキルから成る群から選択される、請求項1または請求項2に記載の医薬組成物。
- Raが2個存在し、一方がハロであり、他方がアルキルである、請求項1ないし請求項3のいずれかに記載の医薬組成物。
- ドルーゼンの除去および/または形成抑制用の、請求項1ないし請求項5のいずれかに記載の医薬組成物。
- 加齢黄斑変性の処置および/または予防用の、請求項1ないし請求項5のいずれかに記載の医薬組成物。
- 加齢黄斑変性が、滲出型加齢黄斑変性または萎縮型加齢黄斑変性である、請求項7に記載の医薬組成物。
- 経口投与用の請求項1ないし請求項8のいずれかに記載の医薬組成物。
- ドルーゼンの除去および/または形成抑制用、および/または、加齢黄斑変性の処置および/または予防用の医薬組成物を製造するための、請求項1に記載の式(I)の化合物またはそのオキシド、エステル、薬学的に許容される塩もしくは溶媒和物の使用。
- ドルーゼンの除去および/または形成抑制、および/または、加齢黄斑変性の処置および/または予防の方法であって、それを必要とする対象に治療上有効量の請求項1に記載の式(I)の化合物またはそのオキシド、エステル、プロドラッグ、薬学的に許容される塩もしくは溶媒和物を投与することを含む方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DK14754749.1T DK2959903T3 (en) | 2013-02-20 | 2014-02-19 | Medicine for the treatment of eye disease |
| PL14754749T PL2959903T3 (pl) | 2013-02-20 | 2014-02-19 | Lek do leczenia chorób oczu |
| ES14754749.1T ES2691805T3 (es) | 2013-02-20 | 2014-02-19 | Medicamento para el tratamiento de una enfermedad ocular |
| EP14754749.1A EP2959903B1 (en) | 2013-02-20 | 2014-02-19 | Medicine for treating eye disease |
| JP2015501476A JP6261011B2 (ja) | 2013-02-20 | 2014-02-19 | 眼疾患処置薬 |
| US14/768,949 US9782420B2 (en) | 2013-02-20 | 2014-02-19 | Method for treating eye diseases |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-031190 | 2013-02-20 | ||
| JP2013031190 | 2013-02-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014129495A1 true WO2014129495A1 (ja) | 2014-08-28 |
Family
ID=51391281
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/053898 WO2014129495A1 (ja) | 2013-02-20 | 2014-02-19 | 眼疾患処置薬 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US9782420B2 (ja) |
| EP (1) | EP2959903B1 (ja) |
| JP (1) | JP6261011B2 (ja) |
| DK (1) | DK2959903T3 (ja) |
| ES (1) | ES2691805T3 (ja) |
| PL (1) | PL2959903T3 (ja) |
| PT (1) | PT2959903T (ja) |
| WO (1) | WO2014129495A1 (ja) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015033981A1 (ja) * | 2013-09-04 | 2015-03-12 | 国立大学法人京都大学 | レプチン抵抗性を改善する医薬組成物 |
| WO2017222042A1 (ja) * | 2016-06-24 | 2017-12-28 | 国立大学法人京都大学 | 眼用医薬組成物 |
| JP6308285B1 (ja) * | 2016-12-08 | 2018-04-11 | コニカミノルタ株式会社 | 透明基板積層体の製造方法および空中映像表示デバイスの製造方法 |
| WO2019131720A1 (ja) | 2017-12-27 | 2019-07-04 | 国立大学法人京都大学 | 角膜保護用の組成物 |
| WO2019203176A1 (ja) | 2018-04-16 | 2019-10-24 | 国立大学法人京都大学 | 心筋細胞の保護用の医薬組成物 |
| WO2020027137A1 (ja) | 2018-07-31 | 2020-02-06 | 国立大学法人京都大学 | 脳梗塞の処置用の医薬組成物 |
| WO2021079983A1 (ja) | 2019-10-24 | 2021-04-29 | 国立大学法人京都大学 | 軟骨保護用の医薬組成物 |
| WO2022230949A1 (ja) | 2021-04-28 | 2022-11-03 | 国立大学法人京都大学 | 皮膚組織の異常を改善するための組成物 |
| WO2024090512A1 (ja) * | 2022-10-27 | 2024-05-02 | 株式会社京都創薬研究所 | ナフタレン誘導体の結晶 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5607915A (en) | 1992-09-29 | 1997-03-04 | Inhale Therapeutic Systems | Pulmonary delivery of active fragments of parathyroid hormone |
| US20030065020A1 (en) * | 2001-07-13 | 2003-04-03 | Catharine Gale | Treatment of macular degeneration |
| WO2012014994A1 (ja) | 2010-07-30 | 2012-02-02 | ダイトーケミックス株式会社 | ナフタレン誘導体 |
| WO2012043891A1 (ja) | 2010-09-30 | 2012-04-05 | ダイトーケミックス株式会社 | 眼疾患処置薬 |
| JP2013031190A (ja) | 2007-03-26 | 2013-02-07 | Qualcomm Inc | 改善された周波数オフセット推定機 |
-
2014
- 2014-02-19 ES ES14754749.1T patent/ES2691805T3/es active Active
- 2014-02-19 PT PT14754749T patent/PT2959903T/pt unknown
- 2014-02-19 PL PL14754749T patent/PL2959903T3/pl unknown
- 2014-02-19 DK DK14754749.1T patent/DK2959903T3/en active
- 2014-02-19 JP JP2015501476A patent/JP6261011B2/ja active Active
- 2014-02-19 US US14/768,949 patent/US9782420B2/en active Active
- 2014-02-19 WO PCT/JP2014/053898 patent/WO2014129495A1/ja active Application Filing
- 2014-02-19 EP EP14754749.1A patent/EP2959903B1/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5607915A (en) | 1992-09-29 | 1997-03-04 | Inhale Therapeutic Systems | Pulmonary delivery of active fragments of parathyroid hormone |
| US20030065020A1 (en) * | 2001-07-13 | 2003-04-03 | Catharine Gale | Treatment of macular degeneration |
| JP2013031190A (ja) | 2007-03-26 | 2013-02-07 | Qualcomm Inc | 改善された周波数オフセット推定機 |
| WO2012014994A1 (ja) | 2010-07-30 | 2012-02-02 | ダイトーケミックス株式会社 | ナフタレン誘導体 |
| WO2012043891A1 (ja) | 2010-09-30 | 2012-04-05 | ダイトーケミックス株式会社 | 眼疾患処置薬 |
Non-Patent Citations (16)
| Title |
|---|
| "1974 RECOMMENDATIONS FOR SECTION E, FUNDAMENTAL STEREOCHEMISTRY", PURE APPL. CHEM., vol. 45, 1976, pages 13 - 30 |
| "Bioreversible Carriers in Drug Design", 1987, AMERICAN PHARMACEUTICAL ASSOCIATION AND PERGAMON PRESS |
| "Methods in Cell Biology", vol. XIV, 1976, ACADEMIC PRESS, pages: 33 |
| "Remington's Pharmaceutical Sciences", 1990, MACK PUBLISHING COMPANY |
| "Remington's Pharmaceutical Sciences", 1991, MACK PUB. CO. |
| ADV EXP MED BIOL., vol. 703, pages 127 - 35 |
| BAGSHAWE K., DRUG DEV. RES., vol. 34, 1995, pages 220 - 230 |
| BERTOLINI, G. ET AL., J. MED. CHEM., vol. 40, 1997, pages 2011 - 2016 |
| BODOR, N., ADVANCES IN DRUG RES., vol. 13, 1984, pages 224 - 331 |
| BUNDGAARD, H.: "Design of Prodrugs", 1985, ELSEVIER PRESS |
| CHI, Z.-L. ET AL.: "Suppression of drusen formation by compstatin, a peptide inhibitor of complement C3 activation, on cynomolgus monkey with early-onset macular degeneration", ADV EXP MED BIOL, vol. 703, 2010, pages 127 - 135, XP055273570 * |
| IOVS, vol. 52, 2011, pages 6004 |
| LARSEN, I. K. ET AL.: "Drug Design and Development", 1991, HARWOOD ACADEMIC PUBLISHERS, article "Design and Application of Prodrugs" |
| SHAN, D. ET AL., J. PHARM. SCI., vol. 86, no. 7, pages 765 - 767 |
| T. HIGUCHI; V. STELLA: "Pro-drugs as Novel Delivery Systems", vol. 14, A.C.S. SYMPOSIUM SERIES |
| YUKI MURAOKA ET AL.: "VCP Sogaizai ni yoru CCR2 Kesson Mouse no Drusen Yokusei Koka no Kento", JOURNAL OF JAPANESE OPHTHALMOLOGICAL SOCIETY, vol. 117, 4 March 2013 (2013-03-04), pages 295 * |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015033981A1 (ja) * | 2013-09-04 | 2015-03-12 | 国立大学法人京都大学 | レプチン抵抗性を改善する医薬組成物 |
| US9931351B2 (en) | 2013-09-04 | 2018-04-03 | Kyoto University | Method for improving leptin resistance |
| WO2017222042A1 (ja) * | 2016-06-24 | 2017-12-28 | 国立大学法人京都大学 | 眼用医薬組成物 |
| JP6308285B1 (ja) * | 2016-12-08 | 2018-04-11 | コニカミノルタ株式会社 | 透明基板積層体の製造方法および空中映像表示デバイスの製造方法 |
| JP2018097013A (ja) * | 2016-12-08 | 2018-06-21 | コニカミノルタ株式会社 | 透明基板積層体の製造方法および空中映像表示デバイスの製造方法 |
| KR20200123095A (ko) | 2017-12-27 | 2020-10-28 | 고쿠리츠 다이가쿠 호진 교토 다이가쿠 | 각막 보호용의 조성물 |
| WO2019131720A1 (ja) | 2017-12-27 | 2019-07-04 | 国立大学法人京都大学 | 角膜保護用の組成物 |
| WO2019203176A1 (ja) | 2018-04-16 | 2019-10-24 | 国立大学法人京都大学 | 心筋細胞の保護用の医薬組成物 |
| KR20200143441A (ko) | 2018-04-16 | 2020-12-23 | 고쿠리츠 다이가쿠 호진 교토 다이가쿠 | 심근 세포의 보호용의 의약 조성물 |
| JPWO2019203176A1 (ja) * | 2018-04-16 | 2021-05-13 | 国立大学法人京都大学 | 心筋細胞の保護用の医薬組成物 |
| JP7369454B2 (ja) | 2018-04-16 | 2023-10-26 | 国立大学法人京都大学 | 心筋細胞の保護用の医薬組成物 |
| WO2020027137A1 (ja) | 2018-07-31 | 2020-02-06 | 国立大学法人京都大学 | 脳梗塞の処置用の医薬組成物 |
| KR20210038587A (ko) | 2018-07-31 | 2021-04-07 | 고쿠리츠 다이가쿠 호진 교토 다이가쿠 | 뇌경색의 처치용의 의약 조성물 |
| WO2021079983A1 (ja) | 2019-10-24 | 2021-04-29 | 国立大学法人京都大学 | 軟骨保護用の医薬組成物 |
| KR20220087499A (ko) | 2019-10-24 | 2022-06-24 | 고쿠리츠 다이가쿠 호진 교토 다이가쿠 | 연골 보호용의 의약 조성물 |
| WO2022230949A1 (ja) | 2021-04-28 | 2022-11-03 | 国立大学法人京都大学 | 皮膚組織の異常を改善するための組成物 |
| WO2024090512A1 (ja) * | 2022-10-27 | 2024-05-02 | 株式会社京都創薬研究所 | ナフタレン誘導体の結晶 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2014129495A1 (ja) | 2017-02-02 |
| PT2959903T (pt) | 2019-01-17 |
| US9782420B2 (en) | 2017-10-10 |
| US20160000810A1 (en) | 2016-01-07 |
| JP6261011B2 (ja) | 2018-01-17 |
| PL2959903T3 (pl) | 2019-03-29 |
| EP2959903A4 (en) | 2016-08-03 |
| EP2959903B1 (en) | 2018-10-03 |
| DK2959903T3 (en) | 2019-01-21 |
| ES2691805T3 (es) | 2018-11-28 |
| EP2959903A1 (en) | 2015-12-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6261011B2 (ja) | 眼疾患処置薬 | |
| JP6628252B2 (ja) | 虚血性眼疾患の処置用の医薬組成物 | |
| WO2019042445A1 (zh) | 一类具有抑制并降解布鲁顿酪氨酸蛋白激酶Btk活性的化合物 | |
| JP5822840B2 (ja) | 眼疾患処置薬 | |
| JP5142714B2 (ja) | Hivプロテアーゼ阻害薬のプロドラッグ | |
| KR102332837B1 (ko) | 폐 병태의 치료를 위한 고침투 전구약물 조성물 및 그의 약제학적 조성물 | |
| AU2022256175B2 (en) | Antimicrobial compounds, compositions, and uses thereof | |
| JP2011510965A (ja) | 高眼圧症の治療のためのジフルオロビフェニルアミド誘導体 | |
| CN111662294A (zh) | 一类具有降解Btk活性的化合物 | |
| EP2139463B1 (en) | Anti-infective agents | |
| JP2010024243A (ja) | 緑内障および近視を処置するためのベンゾ[g]キノリン誘導体 | |
| CN113490669B (zh) | 一类具有降解Btk活性的化合物 | |
| JP2010095550A (ja) | キノリン誘導体 | |
| DE60027461T2 (de) | Megluminsalz einer spezifischen Chinolinsäure-Verbindung mit NMDA Rezeptor-Aktivität | |
| JP2001513532A (ja) | 痛みを処置するための方法 | |
| JP2022515549A (ja) | パントテンアミドアナログ | |
| CA2431761A1 (en) | Novel 2,3-benzodiazepine derivatives and pharmaceutical compositions containing the same as the active ingredient | |
| KR20220080176A (ko) | 퀴놀론 카르복실산 유도체 | |
| WO2019113312A1 (en) | Anticancer compound process | |
| JPH09227529A (ja) | ニトロイミダゾール誘導体 | |
| JP2005298344A (ja) | フェノール誘導体及びその製法 | |
| JPH1025280A (ja) | ニトロイミダゾール誘導体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14754749 Country of ref document: EP Kind code of ref document: A1 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| ENP | Entry into the national phase |
Ref document number: 2015501476 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14768949 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014754749 Country of ref document: EP |