WO2012111614A1 - リチウムマンガン系固溶体正極材料 - Google Patents
リチウムマンガン系固溶体正極材料 Download PDFInfo
- Publication number
- WO2012111614A1 WO2012111614A1 PCT/JP2012/053275 JP2012053275W WO2012111614A1 WO 2012111614 A1 WO2012111614 A1 WO 2012111614A1 JP 2012053275 W JP2012053275 W JP 2012053275W WO 2012111614 A1 WO2012111614 A1 WO 2012111614A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- solid solution
- positive electrode
- electrode material
- solution positive
- lithium
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02T—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO TRANSPORTATION
- Y02T10/00—Road transport of goods or passengers
- Y02T10/60—Other road transportation technologies with climate change mitigation effect
- Y02T10/70—Energy storage systems for electromobility, e.g. batteries
Definitions
- the present invention relates to a solid solution positive electrode material that can be used as a positive electrode active material of a lithium battery such as a lithium primary battery, a lithium secondary battery, a lithium ion secondary battery, and a lithium polymer battery, particularly a lithium manganese-based solid solution containing lithium and manganese.
- a lithium battery such as a lithium primary battery, a lithium secondary battery, a lithium ion secondary battery, and a lithium polymer battery, particularly a lithium manganese-based solid solution containing lithium and manganese.
- the present invention relates to a positive electrode material.
- Lithium batteries especially lithium secondary batteries, have a low mass per unit of electricity and high energy density, so they are used for driving in portable electronic devices such as video cameras, laptop computers, and mobile phones, and electric vehicles. It is rapidly spreading as a power source.
- the high energy density of the lithium secondary battery is mainly due to the potential of the positive electrode material.
- the positive electrode active material lithium manganese oxide (LiMn 2 O 4 ) having a spinel structure and LiCoO 2 having a layered structure are used.
- LiNiO 2 , LiMnO 2, and other lithium composite oxides (LiMxOy) are known.
- the cathode active material of most of the lithium secondary batteries currently on the market is LiCoO 2 having a high voltage of 4 V, but Co is extremely expensive, so as an alternative material for LiCoO 2 , for example, LiFePO 4 or LiNi 1 / 3 Mn 1/3 Co 1/3 O 2 and the like are being studied.
- LiCoO 2 LiCoO 2 having a high voltage of 4 V
- LiFePO 4 or LiNi 1 / 3 Mn 1/3 Co 1/3 O 2 and the like are being studied.
- LiMO 2 and Li 2 MnO 3 LiNi 1 / 3 Mn 1/3 Co 1/3 O 2 and the like are being studied.
- Reference 1 is attracting attention as a so-called solid solution cathode material.
- a solid solution positive electrode material for example, in Patent Document 1, a general formula: xLiMO 2. (1-x) Li 2 NO 3 (where x is a number satisfying 0 ⁇ x ⁇ 1, and M is , One or more transition metals having an average oxidation state of 3 + , and N is one or more transition metals having an average oxidation state of 4 + .)
- a positive electrode material for a lithium ion battery characterized by being subjected to an oxidation treatment is disclosed.
- the main active material is represented by the general formula: xLi 2 MO 3. (1-x) Li [Ni 1-yz Co y A z ] O 2 (x is 0.4 ⁇ x ⁇ 1.0 is satisfied, M is one or more elements selected from the group consisting of Mn, Ti and Zr, and A is one selected from the group consisting of B, Al, Ga and In
- a positive electrode for a lithium ion battery which is expressed by the following elements: 0 ⁇ y ⁇ 0.3, 0 ⁇ z ⁇ 0.1.
- the lithium manganese solid solution positive electrode material is particularly promising in that it has a high discharge potential and can be expected to have a high energy density, and since it is based on Mn, the raw material cost can be reduced. Has been.
- this type of lithium manganese solid solution positive electrode material has a serious problem that gas is generated particularly during the initial cycle when charging and discharging. Therefore, the present invention is to provide a new lithium manganese solid solution positive electrode material capable of effectively suppressing gas generation during the initial cycle.
- the present inventor is a lithium manganese-based solid solution positive electrode material containing a C2 / m monoclinic structure in the hexagonal structure of the space group R-3m, the composition formula: xLi 4/3 Mn 2/3 O 2 + ( 1-x) Contains a solid solution represented by LiMn ⁇ Co ⁇ Ni ⁇ O 2 (where 0.2 ⁇ ⁇ ⁇ 0.6, 0 ⁇ ⁇ ⁇ 0.4, 0.2 ⁇ ⁇ ⁇ 0.6)
- a lithium manganese solid solution positive electrode material is proposed, wherein x in the composition formula is 0.36 or more and less than 0.50.
- Li 4/3 Mn 2/3 O 2 and LiMn ⁇ Co ⁇ Ni ⁇ O 2 (where 0.2 ⁇ ⁇ ⁇ 0.6, 0 ⁇ ⁇ ⁇ 0.4, 0.2 ⁇ ⁇ ⁇ 0.6 ) Is extremely narrow so that x indicating the molar ratio between the Li 4/3 Mn 2/3 O 2 and the LiMn ⁇ Co ⁇ Ni ⁇ O 2 is 0.36 or more and less than 0.50.
- gas generation during the initial cycle can be remarkably suppressed. Therefore, it can also be used for a large battery that could not be realized by this type of conventional solid solution positive electrode material.
- FIG. 2 is an XRD pattern of a solid solution powder (sample) obtained in Example 2.
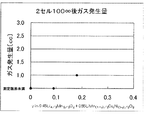
- FIG. 6 is a graph showing the composition dependency of the amount of gas generated based on the measured values of Example 1-5 and Comparative Example 1-2. It is the graph which showed the composition dependence of the amount of gas generation based on the measured value of the solid solution powder (sample) obtained in Example 2, 6 and 7.
- the solid solution positive electrode material according to the present embodiment includes a monoclinic structure of C2 / m in the hexagonal structure of the space group R-3m, and a composition formula: xLi 4 / 3 Mn 2/3 O 2 + (1-x) LiMn ⁇ Co ⁇ Ni ⁇ O 2 (where 0.2 ⁇ ⁇ ⁇ 0.6, 0 ⁇ ⁇ ⁇ 0.4, 0.2 ⁇ ⁇ ⁇ 0 .6) is a positive electrode material containing the solid solution.
- this solid solution positive electrode material does not belong to the hexagonal crystal structure of the space group R-3m, in addition to the peak attributed to the hexagonal crystal structure of the space group R-3m.
- Li 4/3 Mn 2/3 O 2 and LiMn ⁇ Co ⁇ Ni ⁇ O 2 (where 0.2 ⁇ ⁇ ⁇ 0.6, 0 ⁇ ⁇ ⁇ 0.4, 0.2 It is important that x indicating a molar ratio of ⁇ ⁇ ⁇ 0.6) is 0.36 or more and less than 0.50.
- x is 0.36 or more and less than 0.50, gas generation during the initial cycle can be remarkably suppressed. From the viewpoint of suppressing such gas generation, x is more preferably 0.38 or more and 0.48 or less, and more preferably 0.40 or more and 0.47 or less.
- LiMn ⁇ Co ⁇ Ni ⁇ O 2 (where 0.2 ⁇ ⁇ ⁇ 0.6, 0 ⁇ ⁇ ⁇ 0.4, 0.2 ⁇ ⁇ ⁇ 0.6), LiMn (1 A compound represented by - ⁇ ) / 2 Co ⁇ Ni (1- ⁇ ) / 2 O 2 is more preferable.
- ⁇ may be 0.0, and ⁇ may be in the range of 0.0 to 0.2.
- this solid solution positive electrode material may contain B (boron). At this time, B (boron) may exist in the solid solution or may exist outside the solid solution.
- the c-axis length of the crystal lattice of the hexagonal structure of the space group R-3m is preferably 14.255 ⁇ to 14.275 ⁇ , and in particular, 14.257 ⁇ . More preferably, it is 14.274 mm or less, more preferably 14.259 mm or more or 14.271 mm or less.
- the total area of peaks located in the range of ⁇ 18.0 ° to 19.5 ° in the XRD pattern obtained when the solid solution positive electrode material is subjected to XRD measurement.
- ⁇ Method for producing the solid solution positive electrode material First, raw materials such as a lithium salt compound, a manganese salt compound, a nickel salt compound, and a cobalt salt compound are mixed and pulverized with a wet pulverizer or the like. Thereafter, they are granulated and dried using a spray dryer or the like, and fired. If necessary, they are classified or pulverized using a collision pulverizer equipped with a classification mechanism. Furthermore, this solid solution positive electrode material can be obtained by heat-treating depending on the case or classification after that. However, the manufacturing method of this solid solution positive electrode material is not limited to this manufacturing method. For example, granulated powder to be subjected to firing may be produced by a method generally called a coprecipitation method.
- lithium salt compound examples include lithium hydroxide (LiOH), lithium carbonate (Li 2 CO 3 ), lithium nitrate (LiNO 3 ), lithium hydroxide hydrate (LiOH ⁇ H 2 O), and lithium oxide (Li 2 O). ) And other fatty acid lithium and lithium halide. Of these, lithium hydroxide salts, carbonates and nitrates are preferred.
- manganese salt compound there is no particular limitation on the manganese salt compound.
- manganese carbonate, manganese nitrate, manganese chloride, manganese dioxide and the like can be used, and among these, manganese carbonate and manganese dioxide are preferable.
- electrolytic manganese dioxide obtained by an electrolytic method is particularly preferable.
- the type of the nickel salt compound is not particularly limited, and for example, nickel carbonate, nickel nitrate, nickel chloride, nickel oxyhydroxide, nickel hydroxide, nickel oxide, etc. can be used, among which nickel carbonate, nickel hydroxide, nickel oxide are used. preferable.
- cobalt salt compound is not particularly limited, and for example, basic cobalt carbonate, cobalt nitrate, cobalt chloride, cobalt oxyhydroxide, cobalt hydroxide, cobalt oxide and the like can be used. Cobalt, cobalt oxide, and cobalt oxyhydroxide are preferred.
- the raw materials When mixing the raw materials, it is preferable to add a liquid medium such as water or a dispersant to the raw materials and wet-mix them to form a slurry, and the obtained slurry is preferably pulverized with a wet pulverizer. However, dry pulverization may be performed. Then, it is preferable to grind so that the average particle diameter (D50) is 0.2 ⁇ m to 1.0 ⁇ m.
- the granulation method may be wet or dry as long as the various raw materials pulverized in the previous step are dispersed in the granulated particles without being separated.
- an extrusion granulation method a rolling granulation method, a fluidized granulation method, a mixed granulation method, a spray drying granulation method, a pressure molding granulation method, or a flake granulation method using a roll or the like may be used.
- the drying method may be a known drying method such as a spray heat drying method, a hot air drying method, a vacuum drying method, a freeze drying method, etc. Among them, the spray heat drying method is preferable.
- the spray heat drying method is preferably performed using a spray dryer (spray dryer).
- the above firing is performed at a temperature of 850 to 1100 ° C. in a firing furnace in an air atmosphere, an atmosphere in which an oxygen partial pressure is adjusted, a carbon dioxide gas atmosphere, or other atmosphere (: firing in a firing furnace).
- the temperature when the thermocouple is brought into contact with the object is preferably calcined so as to hold for 0.5 to 30 hours.
- the kind of baking furnace is not specifically limited. For example, it can be fired using a rotary kiln, a stationary furnace, or other firing furnace.
- the classification after calcination has the technical significance of adjusting the particle size distribution of the agglomerated powder and removing foreign substances, and it is preferable to classify so that the average particle diameter (D50) is 2 ⁇ m to 50 ⁇ m.
- the pulverization after classification is performed by using a collision type pulverizer with a classification mechanism, for example, a counter jet mill with a classification rotor so that the ratio of the average particle diameter (D50) and the crystallite diameter falls within a predetermined range. Is preferred.
- the powder particles obtained by pulverization with a collision type pulverizer with a classification mechanism are usually non-spherical.
- the above heat treatment is preferably held in an atmosphere of 300 ° C. to 700 ° C., preferably 600 ° C. to 700 ° C. for 0.5 to 20 hours in an air atmosphere. At this time, if the temperature is lower than 300 ° C., the effect of the heat treatment is difficult to obtain and the fine powder may remain without being sintered. On the other hand, if the heat treatment is performed at a temperature higher than 700 ° C., sintering starts and the present invention is aimed. It becomes impossible to obtain the powder characteristics.
- the classification after the heat treatment has technical significance of adjusting the particle size distribution of the agglomerated powder and removing foreign matter, and it is preferable to classify the particles into an average particle diameter (D50) of 2 ⁇ m to 50 ⁇ m.
- the solid solution positive electrode material can be effectively used as a positive electrode active material of a lithium battery after being crushed and classified as necessary.
- a positive electrode mixture can be produced by mixing the solid solution positive electrode material, a conductive material made of carbon black or the like, and a binder made of Teflon (registered trademark) binder or the like.
- the positive electrode mixture thus obtained is used, the negative electrode is made of a material capable of inserting and extracting lithium such as lithium or carbon, and the nonaqueous electrolyte is made of lithium hexafluorophosphate (LiPF6) or the like.
- LiPF6 lithium hexafluorophosphate
- a lithium battery can be formed using a lithium salt dissolved in a mixed solvent such as ethylene carbonate and dimethyl carbonate.
- Lithium batteries configured in this way are, for example, notebook computers, mobile phones, cordless phones, video movies, LCD TVs, electric shavers, portable radios, headphone stereos, backup power supplies, memory cards, and other electronic devices, pacemakers, hearing aids It can be used as a drive power source for medical equipment such as electric vehicles.
- mobile phones that require excellent cycle characteristics
- portable computers such as PDAs (personal digital assistants) and notebook computers
- electric vehicles including hybrid vehicles
- power sources for driving power storage etc. It is valid.
- a laminate-type battery was produced using the solid solution powder (sample) produced in Examples / Comparative Examples as a positive electrode active material, and the following gas generation evaluation test and battery performance evaluation test were performed.
- the negative electrode current collector a copper foil having a thickness of 18 ⁇ m was used. 92% by weight of graphite as an active material and 8% by weight of PVDF as a binder were mixed, and NMP was added to prepare a paste. This paste was uniformly applied to the negative electrode current collector and dried at 100 ° C. Then, it compressed to thickness 80micrometer and produced the negative electrode sheet.
- the positive electrode sheet obtained above was cut into a size of 2.9 cm ⁇ 4.0 cm to be a positive electrode, while the negative electrode sheet obtained above was cut into a size of 3.1 cm ⁇ 4.2 cm to be a negative electrode,
- a separator (made of a porous polyethylene film) was placed between the positive electrode and the negative electrode to produce a laminate type battery.
- a separator impregnated with a volume% added electrolyte was used.
- the charge / discharge voltage range was 1.9 to 4.8 V at the first cycle, and 1.9 to 4.5 V at the second and subsequent cycles.
- the current value was determined and set so as to obtain a charge / discharge rate of 0.2 C from the content of the positive electrode active material in the positive electrode. Under these conditions, charge / discharge was repeated to calculate the capacity retention rate of each cycle relative to the initial discharge capacity.
- the measurement temperature was 25 ° C.
- XRD measurement> The XRD measurement was performed under the following conditions using the apparatus name “RINT-TTRIII” manufactured by Rigaku Corporation to obtain an XRD pattern, and the peak area was determined based on this.
- XRD measurement conditions X-ray source: CuK ⁇ , operation axis: 2 ⁇ / ⁇ , measurement method: continuous, counting unit: cps Start angle: 5 °, end angle: 80 °, Sampling width: 0.02 °, scan speed: 4 ° / min, Voltage: 50 kV, current: 300 mA Divergence slit: 2/3 °, divergence length: 10 mm Scattering slit: 2/3 °, light receiving slit: 0.15 mm
- the c-axis length was obtained by refining the lattice constant with the analysis software “JADE verson7.5.22 (Japanese version)”.
- a polycarboxylic acid ammonium salt (SN Dispersant 5468 manufactured by San Nopco Co., Ltd.) as a dispersant was added to the obtained slurry (raw material powder 20 kg) at 6% by weight of the slurry solid content, and 1300 rpm for 29 minutes using a wet pulverizer.
- the average particle size (D50) was adjusted to 0.7 ⁇ m by pulverization.
- the obtained pulverized slurry was granulated and dried using a spray dryer (spray dryer, OC-16 manufactured by Okawahara Chemical Co., Ltd.). At this time, a rotating disk was used for spraying, and granulation drying was performed by adjusting the rotation speed to 21000 rpm, the slurry supply amount to 24 kg / hr, and the outlet temperature of the drying tower to 100 ° C.
- the obtained granulated powder was baked at 950 ° C. for 20 hours in the air using a stationary electric furnace.
- the fired powder obtained by firing was classified with a sieve having a mesh opening of 75 ⁇ m to obtain a lithium manganese solid solution powder (sample).
- Example 2-7 and Comparative Example 1-2 A lithium manganese based solid solution powder (sample) was prepared in the same manner as in Example 1 except that the composition of the raw material was changed.
- the composition of the lithium manganese-based solid solution powder (sample) obtained in Example 1-7 and Comparative Example 1-2 was analyzed by ICP emission analysis, and the compositions shown in Table 1 were obtained.
Abstract
Description
近年、LiMO2 とLi2MnO3との固溶体を正極活物質として使用すると、4.5V以上で充電した場合に、LiCoO2 の2倍近くの高容量を示すことが報告されており(非特許文献1)、いわゆる固溶体正極材料として注目されている。
そこで本発明は、初期サイクル時のガス発生を有効に抑えることができる新たなリチウムマンガン系固溶体正極材料を提供せんとするものである。
本実施形態に係る固溶体正極材料(以下「本固溶体正極材料」と称する)は、空間群R-3mの六方晶構造にC2/mの単斜晶構造を含み、且つ、組成式:xLi4/3Mn2/3O2+(1-x)LiMnαCoβNiγO2(式中、0.2≦α≦0.6、0≦β≦0.4、0.2≦γ≦0.6)で示される固溶体を含有する正極材料である。
さらにまた、本固溶体正極材料は、B(ホウ素)を含有してもよい。この際、B(ホウ素)は固溶体中に存在してもよいし、固溶体の外に存在してもよい。
上記のθ=18.0°~19.5°の範囲に位置するピークは、空間群R-3mの003面に由来する回折ピークと空間群C2/mの001面に由来するピークに相当する。上記のθ=43.0°~46.0°の範囲に位置するピークは空間群R-3mの104面に由来する回折ピークと空間群C2/mの022面・220面・-202面・131面に由来するピークに相当する。上記のθ=19.5°~23.5°の範囲に位置するピークは空間群C2/mの020面及び110面に由来する回折ピークに相当する。よって、上記のピーク面積比率は、空間群R-3mとC2/mを合算した結晶構造全体に対する空間群C2/mの結晶構造の量と相関するため、当該ピーク面積比率を検討することで、結晶構造全体に対する空間群C2/mの結晶構造の含有量を検討することができる。
まず、例えばリチウム塩化合物、マンガン塩化合物、ニッケル塩化合物及びコバルト塩化合物などの原料を混合し、湿式粉砕機等で粉砕する。その後、それらを噴霧乾燥機等を用いて造粒乾燥させ、焼成する。必要に応じて、それらを分級したり、分級機構付衝突式粉砕機などを用いて粉砕したりする。さらには、場合によって熱処理したり、その後に分級したりすることで本固溶体正極材料を得ることができる。
ただし、本固溶体正極材料の製造方法がかかる製造方法に限定されるものではない。例えば一般的に共沈法と呼ばれる方法によって焼成に供する造粒粉を作製してもよい。
ニッケル塩化合物の種類も特に制限はなく、例えば炭酸ニッケル、硝酸ニッケル、塩化ニッケル、オキシ水酸化ニッケル、水酸化ニッケル、酸化ニッケルなどを用いることができ、中でも炭酸ニッケル、水酸化ニッケル、酸化ニッケルが好ましい。
コバルト塩化合物の種類も特に制限はなく、例えば塩基性炭酸コバルト、硝酸コバルト、塩化コバルト、オキシ水酸化コバルト、水酸化コバルト、酸化コバルトなどを用いることができ、中でも、塩基性炭酸コバルト、水酸化コバルト、酸化コバルト、オキシ水酸化コバルトが好ましい。
そして、平均粒径(D50)が0.2μm~1.0μmとなるように粉砕するのが好ましい。
焼成炉の種類は特に限定するものではない。例えばロータリーキルン、静置炉、その他の焼成炉を用いて焼成することができる。
分級機構付衝突式粉砕機で粉砕して得られる粉体粒子は、非真球形となるのが通常である。
本固溶体正極材料は、必要に応じて解砕・分級した後、リチウム電池の正極活物質として有効に利用することができる。
例えば、本固溶体正極材料と、カーボンブラック等からなる導電材と、テフロン(登録商標)バインダー等からなる結着剤とを混合して正極合剤を製造することができる。そして、そのようにして得られた正極合剤を用い、負極にはリチウムまたはカーボン等のリチウムを吸蔵、脱蔵できる材料を用い、非水系電解質には六フッ化リン酸リチウム(LiPF6)等のリチウム塩をエチレンカーボネート・ジメチルカーボネート等の混合溶媒に溶解したものを用いて、リチウム電池を構成することができる。
本明細書において「X~Y」(X,Yは任意の数字)と表現する場合、特にことわらない限り「X以上Y以下」の意と共に、「好ましくはXより大きい」或いは「好ましくはYより小さい」の意も包含する。
また、「X以上」(Xは任意の数字)或いは「Y以下」(Yは任意の数字)と表現した場合、「Xより大きいことが好ましい」或いは「Y未満であることが好ましい」旨の意図も包含する。
実施例・比較例で作製した固溶体粉末(サンプル)を正極活物質として用いてラミネート型電池を作製し、これを用いて以下に示すガス発生評価試験及び電池性能評価試験を行った。
実施例・比較例で作製した正極活物質としての固溶体粉末(サンプル)90重量%と、導電材としてのアセチレンブラック5重量%と、結着材としてのPVDF(ポリフッ化ビニリデン)5重量%とを混合し、NMP(N-メチルピロリドン)を加えてペースト状に調製した。このペーストを厚さ15μmのアルミニウム箔集電体に塗布し、100℃で乾燥させた。その後、厚さ80μmに圧縮して正極シートを作製した。
上記した方法で作製したラミネート型電池を、12時間放置した後、電流密度0.2CmA/cm2で、両電極間の電位差が4.8Vになるまで充電を行い、その後1.9Vになるまで0.2CmA/cm2で放電を行った。次に同電流値で、両電極間の電位差が4.5Vになるまでの充電と1.9Vになるまでの放電のサイクルを99回実施した。
ここまでに発生する膨れ量(mL)は、浸漬容積法(アルキメデスの原理に基づく溶媒置換法)により計測した。なお、表1の結果は、ラミネート型電池2個について、それぞれ測定した合計量である。
前記で作製したラミネート型電池を用いて次のように充放電を行い、初期容量に対する各サイクルの容量維持率を求めた。
XRD測定は、装置名「リガク社製RINT-TTRIII」を用い、次の条件で測定を行ってXRDパターンを得、これに基づいてピーク面積を求めた。
X線源:CuKα、操作軸:2θ/θ、測定方法:連続、計数単位:cps
開始角度:5°、終了角度:80°、
サンプリング幅:0.02°、スキャンスピード:4°/min、
電圧:50kV、電流:300mA
発散スリット:2/3°、発散縦:10mm
散乱スリット:2/3°、受光スリット:0.15mm
平均粒径(D50)8μmの炭酸リチウムと、平均粒径(D50)22μmの電解二酸化マンガンと、平均粒径(D50)14μmのオキシ水酸化コバルトと、平均粒径(D50)25μmの水酸化ニッケルとを、モル比でLi:Mn:Co:Ni=1.143:0.572:0:0.285となるように秤量し、水を加えて混合攪拌して固形分濃度50重量%のスラリーを調製した。
得られたスラリー(原料粉20kg)に、分散剤としてポリカルボン酸アンモニウム塩(サンノプコ(株)製 SNディスパーサント5468)を前記スラリー固形分の6重量%添加し、湿式粉砕機で1300rpm、29分間粉砕して平均粒径(D50)を0.7μmとした。
得られた粉砕スラリーを噴霧乾燥機(スプレードライヤー、大川原化工機(株)製OC‐16)を用いて造粒乾燥させた。この際、噴霧には回転ディスクを用い、回転数21000rpm、スラリー供給量24kg/hr、乾燥塔の出口温度100℃となるように調節して造粒乾燥を行なった。
原料の組成を変化させた以外は、実施例1と同様にリチウムマンガン系固溶体粉末(サンプル)を作製した。
実施例1-7及び比較例1-2で得られたリチウムマンガン系固溶体粉末(サンプル)をXRD測定で解析したところ、例えば図1に示すように、R-3mに由来せず、かつC2/mに属するピークが2θ=19.5~23.0°に認められた。よって、実施例1-7及び比較例1-2で得られたリチウムマンガン系固溶体は、空間群R-3mの六方晶構造にC2/mの単斜晶構造を含むものであって、Li4/3Mn2/3O2とLiMn1/2Ni1/2O2との固溶体であることが確認された。
また、実施例1―7及び比較例1-2で得られたリチウムマンガン系固溶体粉末(サンプル)をICP発光分析にて組成分析したところ、表1の組成であった。
これより、組成式:xLi4/3Mn2/3O2+(1-x)LiMn(1-β)/2CoβNi(1-β)/2O2(β=0~0.2)で示される固溶体を含有する正極材料においては、xが0.36以上0.50未満であれば、ガス発生を顕著に抑えることができることが分かった。
この結果とこれまでの試験経験を総合すると、xLi4/3Mn2/3O2+(1-x)LiMnαCoβNiγO2(式中、0.2≦α≦0.6、0≦β≦0.4、0.2≦γ≦0.6)で示される固溶体を含有する正極材料において、xが0.36以上0.50未満であれば、上記条件と同様にガス発生を顕著に抑えられるものと考えることができる。
Claims (8)
- 空間群R-3mの六方晶構造にC2/mの単斜晶構造を含むリチウムマンガン系固溶体正極材料であって、組成式:xLi4/3Mn2/3O2+(1-x)LiMnαCoβNiγO2(式中、0.2≦α≦0.6、0≦β≦0.4、0.2≦γ≦0.6)で示される固溶体を含有し、且つ、前記組成式におけるxが0.36以上0.50未満であることを特徴とするリチウムマンガン系固溶体正極材料。
- 空間群R-3mの六方晶構造にC2/mの単斜晶構造を含むリチウムマンガン系固溶体正極材料であって、組成式:xLi4/3Mn2/3O2+(1-x)LiMn(1-β)/2CoβNi(1-β)/2O2(式中、β=0~0.2)で示される固溶体を含有し、且つ、前記組成式におけるxが0.36以上0.50未満であることを特徴とするリチウムマンガン系固溶体正極材料。
- 上記組成式においてβ=0であることを特徴とする請求項2に記載のリチウムマンガン系固溶体正極材料。
- XRDパターンにおいて、空間群R-3mの六方晶構造に帰属するピークのほかに、空間群R-3mの六方晶構造には帰属しないが、C2/mの単斜晶構造には帰属するピークを2θ=19.5~23.0°に有することを特徴とする請求項1~3の何れかに記載のリチウムマンガン系固溶体正極材料。
- 空間群R-3mの六方晶構造の結晶格子のc軸長が14.255Å~14.275Åであることを特徴とする請求項1~4の何れかに記載のリチウムマンガン系固溶体正極材料。
- XRD測定によって求められるXRDパターンにおいて、θ=18.0°~19.5°の範囲に位置するピークの合計面積と、θ=43.0°~46.0°の範囲に位置するピークの合計面積との合算面積に対する、θ=19.5°~23.5°の範囲に位置するピークの合計面積の比率が0.145~0.185であることを特徴とする請求項1~5の何れかに記載のリチウムマンガン系固溶体正極材料。
- 上記組成式において、Li以外の金属元素の置換元素として、Nb、V、Mg、Al及びTiからなる群から選ばれる1種又は2種を含有し、且つ、当該置換元素の合計含有量が、Li以外の金属元素モル数の10モル%以下であることを特徴とする請求項1~6の何れかに記載のリチウムマンガン系固溶体正極材料。
- B(ホウ素)を含有することを特徴とする請求項1~7の何れかに記載のリチウムマンガン系固溶体正極材料。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012509405A JP5352736B2 (ja) | 2011-02-18 | 2012-02-13 | リチウムマンガン系固溶体正極材料 |
| KR1020137021332A KR101886174B1 (ko) | 2011-02-18 | 2012-02-13 | 리튬망간계 고용체 양극 재료 |
| US13/966,788 US9466829B2 (en) | 2011-02-18 | 2013-08-14 | Lithium—manganese-type solid solution positive electrode material |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011033071 | 2011-02-18 | ||
| JP2011-033071 | 2011-02-18 | ||
| JP2011143917 | 2011-06-29 | ||
| JP2011-143917 | 2011-06-29 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/966,788 Continuation US9466829B2 (en) | 2011-02-18 | 2013-08-14 | Lithium—manganese-type solid solution positive electrode material |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012111614A1 true WO2012111614A1 (ja) | 2012-08-23 |
Family
ID=46672533
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/053275 WO2012111614A1 (ja) | 2011-02-18 | 2012-02-13 | リチウムマンガン系固溶体正極材料 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US9466829B2 (ja) |
| JP (1) | JP5352736B2 (ja) |
| KR (1) | KR101886174B1 (ja) |
| WO (1) | WO2012111614A1 (ja) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014007358A1 (ja) * | 2012-07-06 | 2014-01-09 | 住友化学株式会社 | リチウム複合金属酸化物、正極活物質、正極および非水電解質二次電池 |
| JP2014049239A (ja) * | 2012-08-30 | 2014-03-17 | Gs Yuasa Corp | リチウム二次電池用正極活物質、リチウム二次電池用電極、リチウム二次電池 |
| JP2014063707A (ja) * | 2012-08-28 | 2014-04-10 | Gs Yuasa Corp | リチウム二次電池用正極活物質、その正極活物質の製造方法、リチウム二次電池用電極、及びリチウム二次電池 |
| WO2014181455A1 (ja) * | 2013-05-10 | 2014-11-13 | 株式会社 日立製作所 | 非水系二次電池用正極活物質、それを用いた非水系二次電池用正極、非水系二次電池、および、その製造方法 |
| WO2015019483A1 (ja) * | 2013-08-09 | 2015-02-12 | 株式会社日立製作所 | 非水系二次電池用正極活物質、それを用いた非水系二次電池用正極、非水系二次電池 |
| CN105247710A (zh) * | 2013-05-28 | 2016-01-13 | 旭硝子株式会社 | 正极活性物质 |
| US20160056462A1 (en) * | 2013-05-28 | 2016-02-25 | Asahi Glass Company, Limited | Cathode active material |
| JP2016136464A (ja) * | 2015-01-23 | 2016-07-28 | 旭硝子株式会社 | 正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| JP2016136463A (ja) * | 2015-01-23 | 2016-07-28 | 旭硝子株式会社 | 正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| JP2016199414A (ja) * | 2015-04-08 | 2016-12-01 | 旭硝子株式会社 | リチウム含有複合酸化物、その製造方法、正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| US20160365576A1 (en) * | 2015-06-10 | 2016-12-15 | Asahi Glass Company, Limited | Lithium-containing composite oxide, cathode active material, positive electrode for lithium ion secondary battery, and lithium ion secondary battery |
| WO2017082313A1 (ja) * | 2015-11-11 | 2017-05-18 | 旭硝子株式会社 | 正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| WO2020049794A1 (ja) * | 2018-09-05 | 2020-03-12 | パナソニックIpマネジメント株式会社 | 正極活物質およびそれを備えた電池 |
| WO2020049793A1 (ja) * | 2018-09-05 | 2020-03-12 | パナソニックIpマネジメント株式会社 | 正極活物質およびそれを備えた電池 |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6471025B2 (ja) * | 2014-06-27 | 2019-02-13 | 住友化学株式会社 | リチウム含有複合酸化物およびその製造方法 |
| JP6377983B2 (ja) * | 2014-07-23 | 2018-08-22 | 住友化学株式会社 | 正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| WO2017082314A1 (ja) * | 2015-11-11 | 2017-05-18 | 旭硝子株式会社 | 正極活物質の製造方法、正極活物質、正極およびリチウムイオン二次電池 |
| WO2018198410A1 (ja) * | 2017-04-24 | 2018-11-01 | パナソニックIpマネジメント株式会社 | 正極活物質、および、電池 |
| WO2022119156A1 (ko) * | 2020-12-04 | 2022-06-09 | 주식회사 에코프로비엠 | 양극 활물질 및 이를 포함하는 리튬 이차전지 |
| WO2024080470A1 (ko) * | 2022-10-14 | 2024-04-18 | 주식회사 에코프로비엠 | 양극 활물질 및 이를 포함하는 리튬 이차전지 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003044881A1 (fr) * | 2001-11-22 | 2003-05-30 | Yuasa Corporation | Materiau actif d'electrode positive pour cellule secondaire au lithium et cellule secondaire associee |
| JP2007503102A (ja) * | 2003-05-28 | 2007-02-15 | ナショナル・リサーチ・カウンシル・オブ・カナダ | リチウムセルおよびバッテリー用の酸化リチウム電極 |
| JP2007242581A (ja) * | 2006-02-08 | 2007-09-20 | Sanyo Electric Co Ltd | 非水電解質二次電池 |
| JP2008270201A (ja) * | 2007-03-27 | 2008-11-06 | Univ Kanagawa | リチウムイオン電池用正極材料 |
| JP2010277790A (ja) * | 2009-05-27 | 2010-12-09 | Toyota Motor Corp | リチウムイオン二次電池 |
| JP2010282874A (ja) * | 2009-06-05 | 2010-12-16 | Toyota Motor Corp | リチウム二次電池の製造方法 |
| JP2011034943A (ja) * | 2009-03-16 | 2011-02-17 | Sanyo Electric Co Ltd | 非水電解液二次電池 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090127520A1 (en) | 2003-05-28 | 2009-05-21 | Pamela Whitfield | Lithium metal oxide compositions |
| EP2062858A4 (en) * | 2006-09-12 | 2013-05-29 | Sumitomo Chemical Co | LITHIUM COMPOSITE METAL OXIDES AND NONAQUEOUS ELECTROLYTE SECONDARY CELL |
| JP5392813B2 (ja) | 2008-10-31 | 2014-01-22 | 日産自動車株式会社 | リチウムイオン電池とその使用方法 |
| JP5625273B2 (ja) * | 2009-07-24 | 2014-11-19 | 日産自動車株式会社 | リチウムイオン電池用正極材料の製造方法 |
-
2012
- 2012-02-13 KR KR1020137021332A patent/KR101886174B1/ko active IP Right Grant
- 2012-02-13 WO PCT/JP2012/053275 patent/WO2012111614A1/ja active Application Filing
- 2012-02-13 JP JP2012509405A patent/JP5352736B2/ja active Active
-
2013
- 2013-08-14 US US13/966,788 patent/US9466829B2/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003044881A1 (fr) * | 2001-11-22 | 2003-05-30 | Yuasa Corporation | Materiau actif d'electrode positive pour cellule secondaire au lithium et cellule secondaire associee |
| JP2007503102A (ja) * | 2003-05-28 | 2007-02-15 | ナショナル・リサーチ・カウンシル・オブ・カナダ | リチウムセルおよびバッテリー用の酸化リチウム電極 |
| JP2007242581A (ja) * | 2006-02-08 | 2007-09-20 | Sanyo Electric Co Ltd | 非水電解質二次電池 |
| JP2008270201A (ja) * | 2007-03-27 | 2008-11-06 | Univ Kanagawa | リチウムイオン電池用正極材料 |
| JP2011034943A (ja) * | 2009-03-16 | 2011-02-17 | Sanyo Electric Co Ltd | 非水電解液二次電池 |
| JP2010277790A (ja) * | 2009-05-27 | 2010-12-09 | Toyota Motor Corp | リチウムイオン二次電池 |
| JP2010282874A (ja) * | 2009-06-05 | 2010-12-16 | Toyota Motor Corp | リチウム二次電池の製造方法 |
Non-Patent Citations (4)
| Title |
|---|
| A. ITO ET AL.: "A new approach to improve the high-voltage cyclic performance of Li-rich layered cathode material by electrochemical pre-treatment", JOURNAL OF POWER SOURCES, vol. 183, 2008, pages 344 - 346 * |
| ATSUSHI ITO ET AL.: "Koyoryo Li-ion Denchi-yo Seikyoku Zairyo Li2MnO3-LiMO2 no Sosei to Denchi Tokusei", THE ELECTROCHEMICAL SOCIETY OF JAPAN DAI 77 KAI TAIKAI KOEN YOKOSHU, 29 March 2010 (2010-03-29), pages 1B22 * |
| J. KIM ET AL.: "Electrochemical and Structural Properties of xLi2M'O3 - (1-x) LiMno.5Nio.5O2 Electrodes for Lithium Batteries (M'=Ti,Mn, Zr;0<x<0.3)", CHEM. MATER., vol. 16, 2004, pages 1996 - 2006 * |
| L.ZHANG ET AL.: "Synthesis and electrochemistry of layered 0.6LiNio.5Mno.5O2 - xLi2MnO3 - yLiCoO2 (x+y=0.4) cathode materials", MATERIALS LETTERS, vol. 58, 2004, pages 3197 - 3200 * |
Cited By (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9583763B2 (en) | 2012-07-06 | 2017-02-28 | Sumitomo Chemical Company, Limited | Lithium composite metal oxide, positive electrode active substance, positive electrode, and non-aqueous electrolyte secondary battery |
| WO2014007358A1 (ja) * | 2012-07-06 | 2014-01-09 | 住友化学株式会社 | リチウム複合金属酸化物、正極活物質、正極および非水電解質二次電池 |
| JP2014063707A (ja) * | 2012-08-28 | 2014-04-10 | Gs Yuasa Corp | リチウム二次電池用正極活物質、その正極活物質の製造方法、リチウム二次電池用電極、及びリチウム二次電池 |
| JP2014049239A (ja) * | 2012-08-30 | 2014-03-17 | Gs Yuasa Corp | リチウム二次電池用正極活物質、リチウム二次電池用電極、リチウム二次電池 |
| WO2014181455A1 (ja) * | 2013-05-10 | 2014-11-13 | 株式会社 日立製作所 | 非水系二次電池用正極活物質、それを用いた非水系二次電池用正極、非水系二次電池、および、その製造方法 |
| CN105247710A (zh) * | 2013-05-28 | 2016-01-13 | 旭硝子株式会社 | 正极活性物质 |
| US20160043396A1 (en) * | 2013-05-28 | 2016-02-11 | Asahi Glass Company, Limited | Cathode active material |
| US20160056462A1 (en) * | 2013-05-28 | 2016-02-25 | Asahi Glass Company, Limited | Cathode active material |
| WO2015019483A1 (ja) * | 2013-08-09 | 2015-02-12 | 株式会社日立製作所 | 非水系二次電池用正極活物質、それを用いた非水系二次電池用正極、非水系二次電池 |
| JP2016136464A (ja) * | 2015-01-23 | 2016-07-28 | 旭硝子株式会社 | 正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| JP2016136463A (ja) * | 2015-01-23 | 2016-07-28 | 旭硝子株式会社 | 正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| JP2016199414A (ja) * | 2015-04-08 | 2016-12-01 | 旭硝子株式会社 | リチウム含有複合酸化物、その製造方法、正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| JP2017001910A (ja) * | 2015-06-10 | 2017-01-05 | 旭硝子株式会社 | リチウム含有複合酸化物、正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| US20160365576A1 (en) * | 2015-06-10 | 2016-12-15 | Asahi Glass Company, Limited | Lithium-containing composite oxide, cathode active material, positive electrode for lithium ion secondary battery, and lithium ion secondary battery |
| US10355276B2 (en) | 2015-06-10 | 2019-07-16 | Sumitomo Chemical Co., Ltd. | Lithium-containing composite oxide, cathode active material, positive electrode for lithium ion secondary battery, and lithium ion secondary battery |
| US10964944B2 (en) | 2015-06-10 | 2021-03-30 | Sumitomo Chemical Co., Ltd. | Lithium-containing composite oxide, cathode active material, positive electrode for lithium ion secondary battery, and lithium ion secondary battery |
| WO2017082313A1 (ja) * | 2015-11-11 | 2017-05-18 | 旭硝子株式会社 | 正極活物質、リチウムイオン二次電池用正極およびリチウムイオン二次電池 |
| US10811682B2 (en) | 2015-11-11 | 2020-10-20 | Sumitomo Chemical Company, Limited | Cathode active material, positive electrode for lithium ion secondary battery and lithium ion secondary battery |
| WO2020049794A1 (ja) * | 2018-09-05 | 2020-03-12 | パナソニックIpマネジメント株式会社 | 正極活物質およびそれを備えた電池 |
| WO2020049793A1 (ja) * | 2018-09-05 | 2020-03-12 | パナソニックIpマネジメント株式会社 | 正極活物質およびそれを備えた電池 |
| JPWO2020049793A1 (ja) * | 2018-09-05 | 2021-08-12 | パナソニックIpマネジメント株式会社 | 正極活物質およびそれを備えた電池 |
| JP7209163B2 (ja) | 2018-09-05 | 2023-01-20 | パナソニックIpマネジメント株式会社 | 正極活物質およびそれを備えた電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5352736B2 (ja) | 2013-11-27 |
| KR101886174B1 (ko) | 2018-08-08 |
| JPWO2012111614A1 (ja) | 2014-07-07 |
| US20130327979A1 (en) | 2013-12-12 |
| KR20140008344A (ko) | 2014-01-21 |
| US9466829B2 (en) | 2016-10-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5352736B2 (ja) | リチウムマンガン系固溶体正極材料 | |
| KR101746187B1 (ko) | 리튬 이차 전지용 양극 활물질, 및 이를 포함하는 리튬 이차 전지 | |
| US9640794B2 (en) | Lithium transition metal oxide having layered structure | |
| JP4839633B2 (ja) | 非水電解質二次電池および非水電解質二次電池用正極活物質の製造方法 | |
| JP5523637B2 (ja) | マンガン系スピネル型リチウム遷移金属酸化物 | |
| EP2207227A1 (en) | Positive electrode active material for non-aqueous electrolyte secondary battery, and non-aqueous electrolyte secondary battery comprising the same | |
| JP5572268B1 (ja) | スピネル型リチウムマンガンニッケル含有複合酸化物 | |
| JPWO2005020354A1 (ja) | リチウム二次電池用の正極活物質粉末 | |
| JP2006107845A (ja) | 非水系電解質二次電池用正極活物質およびこれを用いた非水系電解質二次電池およびその製造方法 | |
| KR20180059736A (ko) | 리튬 이차 전지용 양극 활물질, 이의 제조방법 및 이를 포함하는 리튬 이차 전지 | |
| US9786914B2 (en) | Spinel-type lithium cobalt manganese-containing complex oxide | |
| JP2006093067A (ja) | リチウム二次電池用正極活物質及びその製造方法 | |
| Ye et al. | Al, B, and F doped LiNi 1/3 Co 1/3 Mn 1/3 O 2 as cathode material of lithium-ion batteries | |
| KR20170076348A (ko) | 리튬 이차 전지용 양극 활물질, 이의 제조방법 및 이를 포함하는 리튬 이차 전지 | |
| WO2016103571A1 (ja) | リチウムコバルト系複合酸化物及びその製造方法、並びに電気化学デバイス及びリチウムイオン二次電池 | |
| JP5951518B2 (ja) | 層構造を有するリチウム金属複合酸化物の製造方法 | |
| WO2014050812A1 (ja) | スピネル型リチウムマンガン含有複合酸化物 | |
| JP2016051548A (ja) | 非水系電解質二次電池用正極材料とその製造方法、および該正極材料を用いた非水系電解質二次電池 | |
| JP6754891B2 (ja) | スピネル型リチウムニッケルマンガン含有複合酸化物 | |
| JP2004186149A (ja) | Liイオン二次電池用正極材料 | |
| JP7308586B2 (ja) | 非水系電解質二次電池用正極活物質 | |
| JP2004339027A (ja) | アルミニウム置換リチウムマンガン酸化物及びその製造方法 | |
| JP6200932B2 (ja) | 層構造を有するリチウム金属複合酸化物の製造方法 | |
| Tan et al. | Synthesis and characterization of spherical LiNi 0.7 Co 0.15 Mn 0.15 O 2 via co-precipitation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012509405 Country of ref document: JP |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12747500 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20137021332 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12747500 Country of ref document: EP Kind code of ref document: A1 |