WO2009154148A1 - 消炎鎮痛外用剤 - Google Patents
消炎鎮痛外用剤 Download PDFInfo
- Publication number
- WO2009154148A1 WO2009154148A1 PCT/JP2009/060768 JP2009060768W WO2009154148A1 WO 2009154148 A1 WO2009154148 A1 WO 2009154148A1 JP 2009060768 W JP2009060768 W JP 2009060768W WO 2009154148 A1 WO2009154148 A1 WO 2009154148A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- oxybuprocaine
- inflammatory
- preparation
- external preparation
- external
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
- A61K9/7023—Transdermal patches and similar drug-containing composite devices, e.g. cataplasms
- A61K9/703—Transdermal patches and similar drug-containing composite devices, e.g. cataplasms characterised by shape or structure; Details concerning release liner or backing; Refillable patches; User-activated patches
- A61K9/7038—Transdermal patches of the drug-in-adhesive type, i.e. comprising drug in the skin-adhesive layer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/06—Ointments; Bases therefor; Other semi-solid forms, e.g. creams, sticks, gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/192—Carboxylic acids, e.g. valproic acid having aromatic groups, e.g. sulindac, 2-aryl-propionic acids, ethacrynic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/235—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids having an aromatic ring attached to a carboxyl group
- A61K31/24—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids having an aromatic ring attached to a carboxyl group having an amino or nitro group
- A61K31/245—Amino benzoic acid types, e.g. procaine, novocaine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/38—Heterocyclic compounds having sulfur as a ring hetero atom
- A61K31/381—Heterocyclic compounds having sulfur as a ring hetero atom having five-membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
- A61K31/405—Indole-alkanecarboxylic acids; Derivatives thereof, e.g. tryptophan, indomethacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/54—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame
- A61K31/5415—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame ortho- or peri-condensed with carbocyclic ring systems, e.g. phenothiazine, chlorpromazine, piroxicam
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
- A61K9/7023—Transdermal patches and similar drug-containing composite devices, e.g. cataplasms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P23/00—Anaesthetics
- A61P23/02—Local anaesthetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
Definitions
- the present invention relates to an anti-inflammatory analgesic, and in particular, has anti-inflammatory / analgesic effects comprising a non-steroidal anti-inflammatory analgesic and a local anesthetic oxybuprocaine or pharmaceutically acceptable salts as medicinal ingredients.
- the present invention relates to a remarkably improved external preparation.
- non-steroidal anti-inflammatory analgesics have been developed and used clinically in place of steroidal anti-inflammatory analgesics with many side effects.
- these non-steroidal anti-inflammatory analgesics exhibit excellent anti-inflammatory and analgesic effects, the side effects of gastrointestinal disorders are strong depending on oral administration, and therefore, percutaneous absorption preparations as administration routes with reduced side effects have been studied.
- external preparations containing non-steroidal anti-inflammatory analgesics for example, ointments, plasters (sticks), poultices, tapes, suspensions, emulsions, lotions and other liquids, aerosols, etc. are being developed.
- ointments plasters (sticks), poultices, tapes, suspensions, emulsions, lotions and other liquids, aerosols, etc.
- the transdermal absorbability of non-steroidal anti-inflammatory analgesics is generally not so high, and when administered as an external preparation, the effect tends to decrease compared to oral administration. Therefore, various studies have been made to improve this transdermal absorbability. For example, the content of a non-steroidal anti-inflammatory analgesic, which is an active ingredient in the preparation, is increased, or a transdermal absorption enhancer is added. Have been devised.
- Patent Document 1 proposes an external patch containing a non-steroidal anti-inflammatory analgesic such as indomethacin, ketoprofen or the like and a local anesthetic such as lidocaine or benzocaine, particularly rheumatoid arthritis and deformity. It is said to be an external patch excellent in analgesic effect for pain accompanied by inflammation such as osteoarthritis and low back pain.
- a non-steroidal anti-inflammatory analgesic such as indomethacin, ketoprofen or the like
- a local anesthetic such as lidocaine or benzocaine, particularly rheumatoid arthritis and deformity. It is said to be an external patch excellent in analgesic effect for pain accompanied by inflammation such as osteoarthritis and low back pain.
- Patent Document 2 proposes an external preparation containing diclofenac sodium and lidocaine or benzocaine as a local anesthetic
- Patent Document 3 further discloses an external preparation containing lidocaine together with piroxicam.
- References 4 to 6 propose external patches containing non-steroidal anti-inflammatory analgesics such as indomethacin, diclofenac and the like, and a local anesthetic such as lidocaine or tetracaine.
- the external preparations proposed above are intended to reduce the skin irritation of non-steroidal anti-inflammatory analgesics, for example, or to improve the penetration and diffusion of drugs in tissues. It is said to be an external preparation that effectively exhibits the anti-inflammatory / analgesic effect of steroidal anti-inflammatory analgesics.
- An object of the present invention is to provide an external preparation having an excellent effect on pain accompanied by inflammation such as low back pain.
- the present inventors have conducted intensive studies, and in the external preparations formulated with non-steroidal anti-inflammatory analgesics and local anesthetics so far proposed, the results of examining the effects of local anesthetics in particular The present inventors have found that, among local anesthetics, when oxybuprocaine is used in combination with a non-steroidal anti-inflammatory analgesic, the analgesic and anti-inflammatory effects of the non-steroidal anti-inflammatory analgesic are effective.
- a topical analgesic / antipruritic preparation containing oxybuprocaine, a local anesthetic, together with a nonsteroidal anti-inflammatory analgesic was prepared, and this preparation was applied to the affected skin area with pain or with inflammation and pain.
- the skin irritation was alleviated and the analgesic action of oxybuprocaine, a local anesthetic, was found to be extremely high, and the present invention was completed. It was.
- the present invention is, as a basic aspect thereof, an external preparation characterized by containing a non-steroidal anti-inflammatory agent and oxybuprocaine or a pharmaceutically acceptable salt as medicinal ingredients.
- the present invention is an external preparation in which the content of the non-steroidal anti-inflammatory analgesic is 0.1 to 10% by weight based on the total weight of the drug-containing preparation, and oxybuprocaine or pharmaceutical Is a topical preparation having a salt content of 0.01 to 60% by weight based on the total weight of the drug-containing preparation.
- the present invention relates to an external preparation characterized by containing 0.1 to 10 parts by weight of oxybuprocaine or a pharmaceutically acceptable salt with respect to 1 part by weight of a nonsteroidal anti-inflammatory analgesic. It is.
- the non-steroidal anti-inflammatory analgesic agent to be incorporated is indomethacin, ketoprofen, piroxicam, felbinac, bufexamac, suprofen, flurbiprofen, diclofenac, ibuprofen and their pharmacologically acceptable. It is an external preparation that is selected from salts.
- the present invention is the above external preparation in which the preparation form as an external preparation is a preparation form of an ointment, liquid, suspension, emulsion, lotion, poultice, tape, aerosol, or external powder.
- a cataplasm or tape containing oxybuprocaine or a pharmaceutically acceptable salt together with felbinac which is a nonsteroidal anti-inflammatory analgesic. It is the external preparation in the form of.
- an external preparation that effectively exhibits the anti-inflammatory / analgesic effect of a non-steroidal anti-inflammatory analgesic, relieves skin irritation at the site of application, and has an excellent effect on pain accompanied by inflammation.
- the external preparation provided by the present invention is excellent in transdermal absorbability and excellent in penetrability and diffusibility of the active ingredient into the tissue after in vivo transfer. Therefore, according to the present invention, there are provided external preparations having various dosage forms that have a sufficient therapeutic effect on various pains and itching of the skin in combination with the action of oxybuprocaine and have very few side effects. And its medical value is tremendous.
- the anti-inflammatory analgesic external preparation of the present invention includes, for example, chronic pain such as rheumatoid arthritis, osteoarthritis, low back pain, inflammatory diseases such as peri-shoulderitis and tendonitis, pain due to surgery or trauma, etc.
- the present invention is an external preparation characterized by containing a non-steroidal anti-inflammatory agent and oxybuprocaine as medicinal ingredients.
- Non-steroidal anti-inflammatory analgesic contained as an active ingredient is selected from indomethacin, ketoprofen, piroxicam, felbinac, bufexamac, suprofen, flurbiprofen, diclofenac, ibuprofen and pharmacologically acceptable salts thereof Compounds, but are not limited to these.
- These nonsteroidal anti-inflammatory analgesics may be used alone or in combination of two or more. Especially, it turned out that a very effective external preparation is provided by selecting felbinac as a non-steroidal anti-inflammatory analgesic.
- the content of the non-steroidal anti-inflammatory analgesic varies depending on the non-steroidal anti-inflammatory analgesic used and the dosage form of the intended external preparation, but is preferably 0.1 to 10 with respect to the total weight of the drug-containing preparation. % By weight, more preferably 0.2 to 5% by weight. If the content is less than the above range, the effect is insufficient, which is not preferable. If the content exceeds the above range, no further effect can be expected, and side effects may occur.
- oxybuprocaine blended with these non-steroidal anti-inflammatory analgesics is a drug developed as a local anesthetic and has surface, infiltration and transmission anesthesia, and is mainly used for surface anesthesia in the ophthalmic field. is there.
- this oxybuprocaine by combining this oxybuprocaine with a non-steroidal anti-inflammatory agent, the analgesic effect of the non-steroidal anti-inflammatory agent at the application site is synergistically enhanced by the local anesthetic effect of oxybuprocaine. It was also found that the skin irritation was alleviated and the transdermal absorbability from the preparation for external use was enhanced.
- the content of oxybuprocaine or a pharmaceutically acceptable salt varies depending on the type of nonsteroidal anti-inflammatory analgesic compounded together and cannot be generally limited, but is preferably 0.01 with respect to the total weight of the drug-containing preparation. -60% by weight, more preferably 0.1-30% by weight. If the content of oxybuprocaine or a pharmaceutically acceptable salt is less than the above range, the combined effect is insufficient, which is not preferable, and if it exceeds the above range, the physical properties of the preparation are affected. In addition, side effects may occur, which is not preferable.
- 0.1 to 10 parts by weight of oxybuprocaine or a pharmaceutically acceptable salt is further contained with respect to 1 part by weight of the nonsteroidal anti-inflammatory analgesic. It turned out to be more effective.
- the external preparation provided by the present invention is not particularly limited as long as the active ingredient can be directly administered to the surface of the diseased part of the skin.
- ointments, liquids (suspensions, emulsions, lotions, etc.), patches It can be prepared and used for preparations such as an agent, a tape, an aerosol, and an external powder.
- preparations such as an agent, a tape, an aerosol, and an external powder.
- Such ingredients include white petrolatum, yellow petrolatum, lanolin, white beeswax, cetanol, stearyl alcohol, stearic acid, hardened oil, gelled carbonization in the case of ointments, creams, gels and lotions.
- Bases such as hydrogen, polyethylene glycol, liquid paraffin, squalane; oleic acid, isopropyl myristate, glycerin triisooctanoate, crotamiton, diethyl sebacate, diisopropyl adipate, hexyl laurate, fatty acid, fatty acid ester, aliphatic alcohol, vegetable oil Solvents and solubilizers such as: Tocopherol derivatives, antioxidants such as L-ascorbic acid, dibutylhydroxytoluene and butylhydroxyanisole; Preservatives such as parahydroxybenzoic acid esters; Glycerin and propylene Moisturizers such as recall and sodium hyaluronate; surfactants such as polyoxyethylene derivatives, glycerin fatty acid ester, sucrose fatty acid ester, sorbitan fatty acid ester, propylene glycol fatty acid ester, lecithin; carboxyviny
- tackifiers such as polyacrylic acid and polyacrylic acid copolymers
- cross-linking agents such as aluminum sulfate, potassium aluminum sulfate, aluminum chloride, magnesium metasilicate aluminate, and dihydroxyaluminum acetate
- Thickeners such as sodium polyacrylate, polyvinyl alcohol, polyvinylpyrrolidone, gelatin, sodium alginate, carboxymethylcellulose, sodium carboxymethylcellulose, hydroxypropylcellulose, hydroxypropylmethylcellulose; glycerin, polyethylene glycol (macrogol), propylene glycol, Polyhydric alcohols such as 1,3-butanediol; surfactants such as polyoxyethylene derivatives; fragrances such as l-menthol; Preservatives such as carboxy acid ester; purified water and the like.
- stabilizers, preservatives, absorption promoters, pH adjusters, and other suitable additives can be blended.
- adhesives such as styrene / isoprene / styrene block copolymers (SIS block copolymers) and acrylic resins; alicyclic saturated hydrocarbon resins, rosin resins, terpene resins, etc. Tackifier resins; softeners such as liquid rubber and liquid paraffin; antioxidants such as dibutylhydroxytoluene; polyhydric alcohols such as propylene glycol; absorption accelerators such as oleic acid; surfactants such as polyoxyethylene derivatives; Other suitable additives can be blended.
- SIS block copolymers styrene / isoprene / styrene block copolymers
- acrylic resins alicyclic saturated hydrocarbon resins, rosin resins, terpene resins, etc. Tackifier resins
- softeners such as liquid rubber and liquid paraffin
- antioxidants such as dibutylhydroxytoluene
- polyhydric alcohols such as
- a water-containing tape can be obtained by adding a water-containing polymer such as sodium polyacrylate or polyvinyl alcohol and a small amount of purified water. Even in this case, a stabilizer, a preservative, an absorption accelerator, a pH adjuster, and other suitable additives can be further blended as desired.
- Bases such as stearic acid, hydrogenated oil, gelled hydrocarbon, polyethylene glycol, liquid paraffin, squalane; oleic acid, isopropyl myristate, diisopropyl adipate, isopropyl sebacate, glycerin triisooctanoate, crotamiton, diethyl sebacate, laurin Solvents and solubilizing agents such as acid hexyl, fatty acid, fatty acid ester, aliphatic alcohol, vegetable oil; antioxidants such as tocopherol derivatives, L-ascorbic acid, dibutylhydroxytoluene, butylhydroxyanisole; Preservatives such as loxybenzoic acid esters; moisturizing agents such as glycerin, propylene glycol, sodium hyaluronate; surface activity such as polyoxyethylene derivatives, glycerin fatty acid esters, sucrose fatty acid esters, sorbitan fatty
- excipients such as potato starch, rice starch, corn starch, talc, zinc oxide or other suitable additives can be blended. Even in this case, various stabilizers, preservatives, absorption accelerators, and other suitable additives can be further blended as desired.
- the means for preparing the external preparation provided by the present invention is not particularly limited.
- a method for producing an ordinary external preparation such as kneading each component and the necessary base component well.
- Manufactured using In preparation of a poultice and a tape, the kneaded mixture is spread on a release paper, dried, further bonded to a flexible support, and cut into a desired size.
- the external preparation provided by the present invention for example, in the case of ointments, liquids (suspensions, emulsions, lotions, etc.), aerosols and powders for external use, may be applied directly to the affected area of the skin, or It is used by a normal method of use such as applying or impregnating a support such as cloth. In the case of a cataplasm or a tape, these preparations are used by directly attaching them to the affected skin area.
- Example 1 A drug-containing base having the formulation shown in Table 1 below was prepared. Specifically, felbinac is dissolved in crotamiton, oxybuprocaine is dissolved in propylene glycol, both are mixed, and then these dissolved materials are kneaded with the other ingredients shown in Table 1 until uniform. A containing base was obtained. The drug-containing base thus prepared was spread on a nonwoven fabric at 1000 g / m 2 , a polypropylene liner was affixed, and cut to 10 ⁇ 14 cm 2 to obtain an external patch.
- Table 1 A drug-containing base having the formulation shown in Table 1 below was prepared. Specifically, felbinac is dissolved in crotamiton, oxybuprocaine is dissolved in propylene glycol, both are mixed, and then these dissolved materials are kneaded with the other ingredients shown in Table 1 until uniform. A containing base was obtained. The drug-containing base thus prepared was spread on a nonwoven fabric at 1000 g / m 2
- Example 2 A drug-containing base having the formulation shown in Table 2 below was prepared. Specifically, indomethacin was dissolved in crotamiton, and oxybuprocaine was dissolved in propylene glycol. Next, these dissolved products were kneaded with the other components shown in Table 2 until uniform, to obtain a drug-containing base.
- the drug-containing base thus prepared was spread on a nonwoven fabric at 1000 g / m 2 , a polypropylene liner was affixed, and cut to 10 ⁇ 14 cm 2 to obtain an external patch.
- Example 3 Drug-containing bases having the formulations shown in Table 3 below were prepared. Specifically, diclofenac sodium was dissolved in N-methyl-2-pyrrolidone, and oxybuprocaine was dissolved in propylene glycol. Next, these dissolved products were kneaded with the other components shown in Table 3 until uniform, to obtain a drug-containing base. The drug-containing base thus prepared was spread on a nonwoven fabric at 1000 g / m 2 , a polypropylene liner was affixed, and cut to 10 ⁇ 14 cm 2 to obtain an external patch.
- Example 4 In accordance with the formulation shown in Table 4 below, styrene / isoprene / styrene block copolymer (SIS block copolymer), hydrogenated rosin glycerin ester, liquid paraffin, polybutene, antioxidant, etc. are added, mixed and melted with toluene, Felbinac and oxybuprocaine are added to this mixture and mixed, and the resulting mixture is spread on release paper, dried with toluene, bonded to a flexible support, cut into a desired size, and tape An agent was obtained.
- SIS block copolymer SIS block copolymer
- hydrogenated rosin glycerin ester liquid paraffin
- polybutene antioxidant
- Felbinac and oxybuprocaine are added to this mixture and mixed, and the resulting mixture is spread on release paper, dried with toluene, bonded to a flexible support, cut into a desired size, and tape An agent was obtained.
- Comparative Example 1 In Example 1, the same amount of purified water was blended in place of oxybuprocaine, and an external patch was obtained by the same method.
- Comparative Example 2 In Example 1, the same amount of purified water was blended in place of felbinac, and an external patch was obtained by the same method.
- Example 3 In Example 1, the same amount of purified water was blended in place of felbinac and oxybuprocaine, and an external patch was obtained in the same manner.
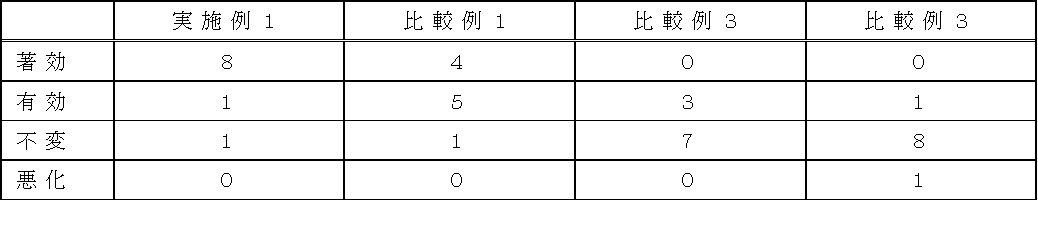
- Test example The external patches obtained in Example 1 and Comparative Examples 1 to 3 were applied to affected areas of 10 male volunteers each having low back pain, and a sensory test was conducted. The administration time was 12 hours per day for 7 days. After the test was completed, the volunteers were asked to evaluate the effect in four levels: “effective”, “effective”, “unchangeable”, and “deteriorated”. Further, the same test was repeated one week after the withdrawal, and the evaluation was performed until the evaluation of all patches was completed. The results are shown in Table 5 below.
- the patch of Example 1 which is an externally applied patch of the present invention containing both ferbinac and oxybuprocaine which are non-steroidal anti-inflammatory analgesics as active ingredients has an improvement rate ( Effective or better) was 90% (9/10), which was the same as the improvement rate of 90% (9/10) in the patch of Comparative Example 1 in which oxybuprocaine was not used in combination, but the improvement rate of the efficacy was 80 % (8/10) and 40% (4/10) are markedly different, and the effectiveness of the present invention is understood.
- the anti-inflammatory / analgesic effect of the non-steroidal anti-inflammatory analgesic agent is effectively exhibited, the skin irritation at the applied site is alleviated, rheumatoid arthritis and osteoarthritis, and External preparations of various dosage forms having excellent effects on pain accompanied by inflammation such as low back pain are provided.
- the external preparation provided by the present invention is excellent in percutaneous absorbability, and has excellent penetrability and diffusibility of an active ingredient into a tissue after in vivo transfer, and is an external preparation with very few side effects. Therefore, its medical value is tremendous.
Abstract
Description
これらの非ステロイド系消炎鎮痛剤は、優れた消炎・鎮痛効果を発揮するものの、経口投与によっては胃腸障害の副作用が強く、したがって、副作用を軽減した投与経路としての経皮吸収製剤が検討され、非ステロイド系消炎鎮痛剤を含有する外用剤として、例えば軟膏剤、硬膏剤(スチック剤)、パップ剤、テープ剤、懸濁剤、乳剤、ローション剤等の液剤、エアゾール剤等の開発が進められ、既に幾つかの製剤が臨床的に使用されるに至っている。
そこでこの経皮吸収性を向上されるため種々の検討がなされており、例えば、製剤中への有効成分である非ステロイド系消炎鎮痛剤の含有量を高めたり、経皮吸収促進剤を配合したりする工夫がなされている。
例えば、特許文献1には、非ステロイド系消炎鎮痛剤である例えば、インドメタシン、ケトプロフェン等と共に局所麻酔剤であるリドカイン又はベンゾカイン等を配合した外用貼付剤が提案されており、特に慢性関節リウマチや変形性関節症、腰痛症等の炎症を伴う痛みの鎮痛効果に優れた外用貼付剤であるとされている。
また本発明が提供する外用剤は、経皮吸収性に優れると共に、生体内移行後の組織中への有効成分の浸透性並びに拡散性に優れたものである。したがって、本発明により、オキシブプロカインの作用と相俟って、皮膚の様々な痛み及び痒みに対して十分な治療効果を有し、副作用の非常に少ない各種の剤型を有する外用剤が提供され、その医療上の価値は多大なものである。
有効成分として含有される非ステロイド系消炎鎮痛剤としては、インドメタシン、ケトプロフェン、ピロキシカム、フェルビナク、ブフェキサマク、スプロフェン、フルルビプロフェン、ジクロフェナック、イブプロフェン及びこれらの薬理学的に許容される塩から選択される化合物が挙げられるが、これらに限定させるものではない。
また、これらの非ステロイド系消炎鎮痛剤は、1種のみでもよく、また2種以上を併用してもよい。
なかでも、非ステロイド系消炎鎮痛剤としてフェルビナクを選択することにより、極めて効果的な外用剤が提供されることが判明した。
含有量が上記の範囲未満であると効果が不十分であるので好ましくなく、また上記の範囲を超えても、それ以上の効果は望めず、かえって副作用が発現する恐れがあり好ましくない。
本発明にあっては、このオキシブプロカインを非ステロイド系消炎鎮痛剤と共に配合することにより、オキシブプロカインの有する局所麻酔効果により適用部位における非ステロイド系消炎鎮痛剤の鎮痛効果が相乗的に高められ、また皮膚刺激性を緩和すると共に、外用剤製剤からの経皮吸収性が高められるものであることが判明した。
オキシブプロカイン又は薬学的に許容される塩類の含有量が上記の範囲未満であると併用効果が不十分であり、好ましいものではなく、また上記の範囲を超えると、製剤の物性に影響を与え、また、副作用が発現する恐れがあり好ましいものではない。
これらの製剤を調製するに際して、有効成分として含有する非ステロイド系消炎鎮痛剤並びにオキシブプロカイン以外に、通常の外用剤を調製するのに使用される各種配合成分を適宜選択して使用することが可能である。
更に、所望により安定剤、保存剤、吸収促進剤、pH調整剤、その他の適当な添加剤を配合することができる。
更に、所望により安定剤、保存剤、吸収促進剤、pH調整剤、その他の適当な添加剤を配合することができる。
また、ポリアクリル酸ナトリウムやポリビニルアルコールのような含水可能な高分子と少量の精製水を加えて含水テープ剤とすることもできる。
この場合にあっても、更に、所望により安定剤、保存剤、吸収促進剤、pH調整剤、その他の適当な添加剤を配合することができる。
この場合にあっても、更に、所望により各種安定剤、保存剤、吸収促進剤、その他の適当な添加剤を配合することができる。
またパップ剤並びにテープ剤の調製にあっては、混練した混合物を剥離紙上に展延、乾燥し、さらに柔軟な支持体と貼り合わせ、所望の大きさに裁断することにより調製することができる。
また、パップ剤或いはテープ剤の場合には、これらの製剤を皮膚患部に直接貼付する方法により使用される。
下記表1に示す処方の薬物含有基剤を調製した。具体的には、フェルビナクはクロタミトンに溶解させ、オキシブプロカインはプロピレングリコールに溶解させ、両者を混合し、次いで、これら溶解物を表1に示す他の成分と均一になるまで練合し、薬物含有基剤を得た。かくして調製された薬物含有基剤を、不織布上に1000g/m2で展延し、ポリプロピレン製のライナーを添着し、10×14cm2になるように裁断して外用貼付剤を得た。
下記表2に示す処方の薬物含有基剤を調製した。具体的には、インドメタシンはクロタミトンに溶解させ、オキシブプロカインはプロピレングリコールに溶解させた。
次いで、これら溶解物を表2に示す他の成分と均一になるまで練合し、薬物含有基剤を得た。かくして調製された薬物含有基剤を、不織布上に1000g/m2で展延し、ポリプロピレン製のライナーを添着し、10×14cm2になるように裁断して外用貼付剤を得た。
下記表3に示す処方の薬物含有基剤を調製した。具体的には、ジクロフェナックナトリウムはN-メチル-2-ピロリドンに溶解させ、オキシブプロカインはプロピレングリコールに溶解させた。
次いで、これら溶解物を表3に示す他の成分と均一になるまで練合し、薬物含有基剤を得た。かくして調製された薬物含有基剤を、不織布上に1000g/m2で展延し、ポリプロピレン製のライナーを添着し、10×14cm2になるように裁断して外用貼付剤を得た。
下記表4に示す処方に従って、スチレン・イソプレン・スチレンブロック共重合体(SISブロック共重合体)、水素添加ロジングリセリンエステル、流動パラフィン、ポリブテン、酸化防止剤等を加え、トルエンにて混合融解させ、この混合物にフェルビナク及びオキシブプロカインを投入混合し、よく混練し得られた混合物を剥離紙上に展延後、トルエンを乾燥し、柔軟な支持体と貼り合わせ、所望の大きさに裁断してテープ剤を得た。
実施例1において、オキシブプロカインの代わりに同量の精製水を配合し、同様の方法により外用貼付剤を得た。
実施例1において、フェルビナクの代わりに同量の精製水を配合し、同様の方法により外用貼付剤を得た。
実施例1において、フェルビナク及びオキシブプロカインの代わりに同量の精製水を配合し、同様の方法により外用貼付剤を得た。
実施例1及び比較例1~3で得られた外用貼付剤を、それぞれ腰痛を有する男性ボランティア10名に患部に貼付し、官能試験を実施した。
投与時間は1日12時間とし、7日間行った。
試験終了後、ボランティアにその効果を「著効」、「有効」、「不変」、「悪化」の4段階で評価してもらった。
更に休薬1週間後に同様の試験を繰り返し、すべての貼付剤の評価が終了するまで行った。
その結果を下記表5に示した。
本発明が提供する外用剤は、経皮吸収性に優れると共に、生体内移行後の組織中への有効成分の浸透性並びに拡散性に優れたものであり、副作用の非常に少ない外用剤であることから、その医療上の価値は多大なものである。
Claims (8)
- 薬効成分として、非ステロイド系消炎鎮痛剤及びオキシブプロカイン又は薬学的に許容される塩類を含有してなることを特徴とする外用剤。
- 非ステロイド系消炎鎮痛剤の含有量が、薬物含有製剤全重量に対して0.1~10重量%である請求項1に記載の外用剤。
- オキシブプロカイン又は薬学的に許容される塩類の含有量が、薬物含有製剤全重量に対して0.01~60重量%である請求項1に記載の外用剤。
- 非ステロイド系消炎鎮痛剤1重量部に対してオキシブプロカインを0.1~10重量部含有したことを特徴とする請求項1に記載の外用剤。
- 非ステロイド系消炎鎮痛剤が、インドメタシン、ケトプロフェン、ピロキシカム、フェルビナク、ブフェキサマク、スプロフェン、フルルビプロフェン、ジクロフェナック、イブプロフェン及びこれらの薬理学的に許容される塩から選択されるものである請求項1ないし4のいずれかに記載の外用剤。
- 非ステロイド系消炎鎮痛剤が、フェルビナクである請求項1ないし4のいずれかに記載の外用剤。
- 軟膏剤、液剤、懸濁剤、乳剤、ローション剤、パップ剤、テープ剤、エアゾール剤又は外用散剤の製剤形態である請求項1ないし6のいずれかに記載の外用剤。
- 薬効成分として、フェルビナク及びオキシブプロカイン又は薬学的に許容される塩類を含有してなることを特徴とする請求項7に記載の外用剤。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2009801234174A CN102065896A (zh) | 2008-06-16 | 2009-06-12 | 消炎镇痛外用剂 |
| CA2727155A CA2727155C (en) | 2008-06-16 | 2009-06-12 | Dermatological preparation comprising oxybuprocaine and a non-steroidal anti-inflammatory |
| AU2009261264A AU2009261264B2 (en) | 2008-06-16 | 2009-06-12 | Analgesic anti-inflammatory preparation for external application |
| US12/999,164 US20110160194A1 (en) | 2008-06-16 | 2009-06-12 | Anti-Inflammatory Analgesic External Preparation |
| EP09766596A EP2305307A4 (en) | 2008-06-16 | 2009-06-12 | ANALGESIC ANTI-INFLAMMATORY PREPARATION FOR EXTERNAL APPLICATION |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008156713A JP2009298741A (ja) | 2008-06-16 | 2008-06-16 | 消炎鎮痛外用剤 |
| JP2008-156713 | 2008-06-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009154148A1 true WO2009154148A1 (ja) | 2009-12-23 |
Family
ID=41434063
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/060768 WO2009154148A1 (ja) | 2008-06-16 | 2009-06-12 | 消炎鎮痛外用剤 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20110160194A1 (ja) |
| EP (1) | EP2305307A4 (ja) |
| JP (1) | JP2009298741A (ja) |
| KR (1) | KR20110017451A (ja) |
| CN (2) | CN102065896A (ja) |
| AU (1) | AU2009261264B2 (ja) |
| CA (1) | CA2727155C (ja) |
| TW (1) | TWI510235B (ja) |
| WO (1) | WO2009154148A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011074565A1 (ja) * | 2009-12-15 | 2011-06-23 | 帝國製薬株式会社 | ピロキシカム含有経皮吸収製剤 |
| WO2011083787A1 (ja) * | 2010-01-07 | 2011-07-14 | 帝國製薬株式会社 | 消炎鎮痛外用貼付剤 |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5677680B2 (ja) * | 2009-12-15 | 2015-02-25 | 帝國製薬株式会社 | 塩基性消炎鎮痛剤含有経皮吸収製剤 |
| US20140113970A1 (en) * | 2012-10-22 | 2014-04-24 | Mallinckrodt LLC Covidien | Dispensing system |
| WO2014068600A1 (en) * | 2012-11-02 | 2014-05-08 | Zydus Technologies Limited | Stable transdermal pharmaceutical drug delivery system comprising diclofenac |
| CN103006764A (zh) * | 2012-12-27 | 2013-04-03 | 上海海虹实业(集团)巢湖今辰药业有限公司 | 一种消炎利胆软胶囊的制作方法 |
| JP6893086B2 (ja) | 2013-03-13 | 2021-06-23 | エーブリー デニソン コーポレイション | 接着特性の向上 |
| KR101439032B1 (ko) * | 2013-06-13 | 2014-09-05 | 동아에스티 주식회사 | 피록시캄과 히알루론산을 포함하는 골관절염 치료를 위한 액상 조성물 |
| CN104367566A (zh) * | 2013-08-12 | 2015-02-25 | 和心医药科技(上海)有限公司 | 一种吲哚美辛巴布剂及其组合物 |
| CN104173322B (zh) * | 2014-09-16 | 2017-06-06 | 徐淑峰 | 一种含吡罗昔康的经皮吸收制剂及其制备方法 |
| CN105287361B (zh) * | 2015-11-13 | 2019-09-03 | 北京泰德制药股份有限公司 | 含有非甾体抗炎药微乳的皮肤外用制剂 |
| CN105663032A (zh) * | 2016-02-23 | 2016-06-15 | 青岛科技大学 | 一种维他昔布软膏剂的制备方法 |
| CN107028921A (zh) * | 2017-05-16 | 2017-08-11 | 蔡志浩 | 一种吡罗昔康巴布贴剂 |
| CN108451902A (zh) * | 2018-06-29 | 2018-08-28 | 佛山市南海东方澳龙制药有限公司 | 治疗动物乳房炎的酮洛芬药浴液及其制备方法 |
| WO2020021670A1 (ja) * | 2018-07-26 | 2020-01-30 | マルホ株式会社 | 液状外用剤 |
| JP2022096997A (ja) * | 2020-12-18 | 2022-06-30 | 小林製薬株式会社 | 外用医薬組成物 |
| WO2022193185A1 (en) * | 2021-03-17 | 2022-09-22 | Jiangyin Usun Pharmaceutical Co., Ltd. | New conjugates |
| TWI789000B (zh) * | 2021-09-09 | 2023-01-01 | 竟天生物科技股份有限公司 | 局部麻醉劑-黏土複合組合物 |
| CN117794524A (zh) * | 2021-09-09 | 2024-03-29 | 竟天生物科技股份有限公司 | 局部麻醉剂-粘土复合组合物 |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH024A (ja) | 1987-02-04 | 1990-01-05 | Asahi Optical Co Ltd | カメラの自動焦点検出装置 |
| JPH0323502A (ja) | 1989-06-21 | 1991-01-31 | Matsushita Electric Ind Co Ltd | 磁気ヘッド及びその製造方法 |
| JPH101441A (ja) * | 1996-06-14 | 1998-01-06 | Tendou Seiyaku Kk | 局所麻酔組成物 |
| WO2001047559A1 (fr) | 1999-12-27 | 2001-07-05 | Teikoku Seiyaku Co., Ltd | Pastilles a usage externe |
| JP2002128699A (ja) * | 2000-10-26 | 2002-05-09 | Sankyo Co Ltd | 消炎鎮痛外用剤組成物 |
| JP2003335663A (ja) | 2002-05-20 | 2003-11-25 | Medorekkusu:Kk | 消炎鎮痛外用剤 |
| JP2004123632A (ja) | 2002-10-03 | 2004-04-22 | Medorekkusu:Kk | 消炎鎮痛外用剤 |
| WO2005007140A1 (en) * | 2003-07-16 | 2005-01-27 | Jong-Hoon Kim | Pharmaceutical composition containing insect repellent |
| JP2005068035A (ja) | 2003-08-20 | 2005-03-17 | Medorekkusu:Kk | 慢性疼痛治療用外用剤 |
| JP2005145932A (ja) | 2003-11-19 | 2005-06-09 | Medorekkusu:Kk | 消炎鎮痛外用剤 |
| JP2005145931A (ja) * | 2003-11-19 | 2005-06-09 | Medorekkusu:Kk | 非ステロイド系消炎鎮痛剤を含有するテープ剤 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1232792B1 (en) * | 2001-02-20 | 2006-09-06 | F. Hoffmann-La Roche Ag | Linear cuvette array, a two-dimensional cuvette array built therewith and a system comprising such two-dimensional cuvette arrays |
| JP2010024223A (ja) * | 2008-06-16 | 2010-02-04 | Teikoku Seiyaku Co Ltd | オキシブプロカイン含有鎮痛・鎮痒用外用剤 |
| JP5677680B2 (ja) * | 2009-12-15 | 2015-02-25 | 帝國製薬株式会社 | 塩基性消炎鎮痛剤含有経皮吸収製剤 |
| CN102753178B (zh) * | 2009-12-15 | 2014-07-30 | 帝国制药株式会社 | 含吡罗昔康的经皮吸收制剂 |
-
2008
- 2008-06-16 JP JP2008156713A patent/JP2009298741A/ja active Pending
-
2009
- 2009-06-12 CN CN2009801234174A patent/CN102065896A/zh active Pending
- 2009-06-12 AU AU2009261264A patent/AU2009261264B2/en not_active Ceased
- 2009-06-12 WO PCT/JP2009/060768 patent/WO2009154148A1/ja active Application Filing
- 2009-06-12 CA CA2727155A patent/CA2727155C/en not_active Expired - Fee Related
- 2009-06-12 CN CN2013103347739A patent/CN103394091A/zh active Pending
- 2009-06-12 EP EP09766596A patent/EP2305307A4/en not_active Withdrawn
- 2009-06-12 US US12/999,164 patent/US20110160194A1/en not_active Abandoned
- 2009-06-12 TW TW098119643A patent/TWI510235B/zh not_active IP Right Cessation
- 2009-06-12 KR KR1020117000892A patent/KR20110017451A/ko not_active Application Discontinuation
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH024A (ja) | 1987-02-04 | 1990-01-05 | Asahi Optical Co Ltd | カメラの自動焦点検出装置 |
| JPH0323502A (ja) | 1989-06-21 | 1991-01-31 | Matsushita Electric Ind Co Ltd | 磁気ヘッド及びその製造方法 |
| JPH101441A (ja) * | 1996-06-14 | 1998-01-06 | Tendou Seiyaku Kk | 局所麻酔組成物 |
| WO2001047559A1 (fr) | 1999-12-27 | 2001-07-05 | Teikoku Seiyaku Co., Ltd | Pastilles a usage externe |
| JP2002128699A (ja) * | 2000-10-26 | 2002-05-09 | Sankyo Co Ltd | 消炎鎮痛外用剤組成物 |
| JP2003335663A (ja) | 2002-05-20 | 2003-11-25 | Medorekkusu:Kk | 消炎鎮痛外用剤 |
| JP2004123632A (ja) | 2002-10-03 | 2004-04-22 | Medorekkusu:Kk | 消炎鎮痛外用剤 |
| WO2005007140A1 (en) * | 2003-07-16 | 2005-01-27 | Jong-Hoon Kim | Pharmaceutical composition containing insect repellent |
| JP2005068035A (ja) | 2003-08-20 | 2005-03-17 | Medorekkusu:Kk | 慢性疼痛治療用外用剤 |
| JP2005145932A (ja) | 2003-11-19 | 2005-06-09 | Medorekkusu:Kk | 消炎鎮痛外用剤 |
| JP2005145931A (ja) * | 2003-11-19 | 2005-06-09 | Medorekkusu:Kk | 非ステロイド系消炎鎮痛剤を含有するテープ剤 |
Non-Patent Citations (2)
| Title |
|---|
| See also references of EP2305307A4 |
| TAKAHIRO AKAISHI ET AL.: "Heiken Hanno o Shihyo to shita Kato Kakumaku Chikaku Hyokakei no Kakuritsu", FOLIA OPHTHALMOGICA JAPONICA, vol. 52, no. 5, 2001, pages 377 - 382, XP008139188 * |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011074565A1 (ja) * | 2009-12-15 | 2011-06-23 | 帝國製薬株式会社 | ピロキシカム含有経皮吸収製剤 |
| CN102753178A (zh) * | 2009-12-15 | 2012-10-24 | 帝国制药株式会社 | 含吡罗昔康的经皮吸收制剂 |
| AU2010331355B2 (en) * | 2009-12-15 | 2014-04-24 | Teikoku Seiyaku Co., Ltd. | Piroxicam-containing endermic preparation |
| CN102753178B (zh) * | 2009-12-15 | 2014-07-30 | 帝国制药株式会社 | 含吡罗昔康的经皮吸收制剂 |
| US9119861B2 (en) | 2009-12-15 | 2015-09-01 | Teikoku Seiyaku Co., Ltd. | Piroxicam-containing transdermally absorbable preparation |
| WO2011083787A1 (ja) * | 2010-01-07 | 2011-07-14 | 帝國製薬株式会社 | 消炎鎮痛外用貼付剤 |
| CN102791267A (zh) * | 2010-01-07 | 2012-11-21 | 帝国制药株式会社 | 消炎镇痛外用贴剂 |
| US8657798B2 (en) | 2010-01-07 | 2014-02-25 | Teikoku Seiyaku Co., Ltd. | Anti-inflammatory analgesic adhesive patch for external use |
| AU2011204228B2 (en) * | 2010-01-07 | 2014-03-27 | Teikoku Seiyaku Co., Ltd. | Anti-inflammatory analgesic adhesive patch for external use |
| CN102791267B (zh) * | 2010-01-07 | 2015-06-10 | 帝国制药株式会社 | 消炎镇痛外用贴剂 |
| JP5748671B2 (ja) * | 2010-01-07 | 2015-07-15 | 帝國製薬株式会社 | 消炎鎮痛外用貼付剤 |
| KR101797787B1 (ko) | 2010-01-07 | 2017-11-15 | 데이고꾸세이약꾸가부시끼가이샤 | 소염진통제의 외용 패치제 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2305307A1 (en) | 2011-04-06 |
| AU2009261264A1 (en) | 2009-12-23 |
| CA2727155A1 (en) | 2009-12-23 |
| CN103394091A (zh) | 2013-11-20 |
| US20110160194A1 (en) | 2011-06-30 |
| CN102065896A (zh) | 2011-05-18 |
| TWI510235B (zh) | 2015-12-01 |
| AU2009261264B2 (en) | 2014-07-24 |
| CA2727155C (en) | 2016-04-26 |
| TW201004617A (en) | 2010-02-01 |
| EP2305307A4 (en) | 2012-09-05 |
| JP2009298741A (ja) | 2009-12-24 |
| KR20110017451A (ko) | 2011-02-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2009154148A1 (ja) | 消炎鎮痛外用剤 | |
| JPH058169B2 (ja) | ||
| JPWO2009075094A1 (ja) | イオン液体化したエトドラクのテープ剤 | |
| WO2020262057A1 (ja) | 水性貼付剤 | |
| KR20070059079A (ko) | 경피 페로스피론 투여용 의약적 조성물 | |
| JP4813725B2 (ja) | 鎮痒用外用剤 | |
| US8658625B2 (en) | External preparation for treating painful skin wound | |
| JP4791682B2 (ja) | 痔疾患用治療剤 | |
| CN105327351A (zh) | 一种消炎镇痛外用剂 | |
| JP2010024223A (ja) | オキシブプロカイン含有鎮痛・鎮痒用外用剤 | |
| US9833417B2 (en) | Felbinac-containing external patch | |
| US20070196460A1 (en) | External preparation for treating skin or mucosal injury caused by viral infection | |
| JP4313003B2 (ja) | 血液透析による皮膚疾患及び痒みの治療用外用剤 | |
| CN104208698A (zh) | 一种经皮吸收镇痛消炎药物组合物及其应用 | |
| JP2004137215A (ja) | 有痛性皮膚創傷の治療用外用剤 | |
| ZA200606352B (en) | External preparation for treating painful skin wound | |
| MXPA06009275A (en) | External preparation for treating painful skin wound | |
| JP2004137216A (ja) | ウイルス感染による皮膚・粘膜傷害症状に対する外用治療剤 | |
| ZA200606351B (en) | Remedy for external use for skin and mucosal injuries caused by viral infection | |
| MXPA06009302A (en) | Remedy for external use for skin and mucosal injuries caused by viral infection |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980123417.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09766596 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 4641/KOLNP/2010 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2727155 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009261264 Country of ref document: AU |
|
| REEP | Request for entry into the european phase |
Ref document number: 2009766596 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009766596 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2009261264 Country of ref document: AU Date of ref document: 20090612 Kind code of ref document: A Ref document number: 20117000892 Country of ref document: KR Kind code of ref document: A |