WO2009122686A1 - リン酸鉄リチウム粒子粉末の製造方法、オリビン型構造のリン酸鉄リチウム粒子粉末、該リン酸鉄リチウム粒子粉末を用いた正極材シート及び非水溶媒系二次電池 - Google Patents
リン酸鉄リチウム粒子粉末の製造方法、オリビン型構造のリン酸鉄リチウム粒子粉末、該リン酸鉄リチウム粒子粉末を用いた正極材シート及び非水溶媒系二次電池 Download PDFInfo
- Publication number
- WO2009122686A1 WO2009122686A1 PCT/JP2009/001374 JP2009001374W WO2009122686A1 WO 2009122686 A1 WO2009122686 A1 WO 2009122686A1 JP 2009001374 W JP2009001374 W JP 2009001374W WO 2009122686 A1 WO2009122686 A1 WO 2009122686A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- iron phosphate
- olivine
- lithium iron
- raw material
- lithium
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/5825—Oxygenated metallic salts or polyanionic structures, e.g. borates, phosphates, silicates, olivines
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B25/00—Phosphorus; Compounds thereof
- C01B25/16—Oxyacids of phosphorus; Salts thereof
- C01B25/26—Phosphates
- C01B25/45—Phosphates containing plural metal, or metal and ammonium
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- olivine type LiFePO 4 has attracted attention as a battery having a high charge / discharge capacity.
- this material has an essentially large electrical resistivity of 10 9 ⁇ ⁇ cm and poor fillability as an electrode, improvement in characteristics is required.
- LiFePO 4 having an olivine structure is composed of a strong phosphoric acid tetrahedral skeleton, an oxygen octahedron centered on iron ions contributing to redox, and lithium ions that are current carriers. Because of this crystal structure, the crystal structure is stable even by repeating the charge / discharge reaction, and the characteristics are less likely to deteriorate compared to other lithium ion cathode materials even if charge / discharge is repeated. On the other hand, there are drawbacks in that the movement path of lithium ions is one-dimensional and has high electrical resistance due to a small number of free electrons. In order to solve these problems, productivity has not been considered, and primary particles of olivine-type LiFePO 4 have been reduced to 200 to 300 nm or less, and research has been conducted on materials substituted with different elements (non-patent literature). 1-5).
- LiFePO 4 tends to have better charge / discharge characteristics under a high current load as the primary particle size of the powder is smaller.
- an olivine-type LiFePO 4 composite oxide positive electrode with excellent characteristics It is necessary to control the aggregate state of each so that they form a network with moderately agglomerated secondary particles and more conductive assistants such as graphitized carbon.
- the positive electrode combined with a large amount of carbon or the like is bulky and has a drawback that the substantial lithium ion density that can be filled per unit volume is lowered.
- LiFePO 4 having an olivine structure with low filling and high impurity content and a small electric resistance is produced by an industrial method with a small environmental load. Is required.
- Patent Document 1 a technology for reducing the electrical resistance by adding a different metal element of LiFePO 4 having an olivine type structure
- Patent Document 2 a technology for improving the tap density when producing olivine type LiFePO 4 and forming a composite with carbon
- Patent Document 3 a technique for obtaining an excellent positive electrode active material by adding a dissimilar metal element using an iron raw material having a valence of 3
- Patent Document 4 a technique using an iron compound of a valence of 3 as a raw material
- Patent Document 4 a technique using an iron compound of a valence of 3 as a raw material
- Non-Patent Documents 1 to 5 do not industrially obtain olivine-type LiFePO 4 having a high filling property and a small amount of amorphous portions and consisting of small primary particles.
- Patent Document 1 is a technique of adding other kinds of metals in order to propose structural stabilization and electrical resistance of the olivine-type LiFePO 4 composite oxide. There is no mention of state controls.
- Patent Document 2 is a technique for forming an aggregate with carbon in the production of a composite oxide of olivine type LiFePO 4 , but it is difficult to say that the battery performance is high.
- Patent Document 3 is difficult to synthesize fine primary particles because the solid phase reactivity of iron oxide used as a raw material is not sufficient.
- Patent Document 4 is a technique that can perform a synthesis reaction while maintaining the particle shape using a general-purpose and inexpensive trivalent iron compound as a raw material. Ion diffusion efficiency is low.
- the present invention establishes an efficient industrial method with a small environmental load of olivine-type LiFePO 4 having a high filling property and a small impurity crystal phase, and as a secondary battery containing a positive electrode material with a high filling property.
- the technical problem is that a high capacity can be obtained even in the current load characteristics, and that it can be used repeatedly sufficiently.
- the present invention uses iron oxide or hydrous oxide as an iron raw material, and contains at least one of the elements Na, Mg, Al, Si, Cr, Mn, and Ni in an amount of 0.1 to 2 mol% with respect to Fe.
- an iron oxide or a hydrous oxide containing 5 to 10 mol% of element C with respect to Fe, Fe 2+ of 40 mol% or less with respect to the amount of Fe, and an average primary particle diameter of 5 to 300 nm is added to the lithium raw material and phosphorus
- the first step of mixing with the raw material the second step of adjusting the resulting mixture so that the aggregated particle diameter is 0.3 to 5.0 ⁇ m, and then the oxygen concentration of the mixture after the second step is 0.1% or less
- This is a method for producing lithium iron phosphate particles having an olivine structure which comprises a third step of firing at a temperature of 250 to 750 ° C. in an inert gas atmosphere or a reducing gas atmosphere (Invention 1).
- the iron raw material used contains at least one of the elements Na, Mg, Al, Si, Cr, Mn, and Ni in an amount of 0.1 to 2 mol% with respect to Fe, and the seven Fe 3 O containing 1.5 to 4 mol% of the total amount of elements, 5 to 10 mol% of element C to Fe, and an average primary particle diameter of 5 to 300 nm. 4.
- a method for producing an olivine-type lithium iron phosphate particle powder according to the present invention 1 comprising at least one of ⁇ -FeOOH, ⁇ -FeOOH, and ⁇ -FeOOH (invention 2).
- the iron raw material used contains at least one of the elements Na, Mg, Al, Si, Cr, Mn, and Ni in an amount of 0.1 to 2 mol% with respect to Fe, and the seven
- the total amount of elements is 1.5 to 4 mol% with respect to Fe
- the element C is 5 to 10 mol% with respect to Fe
- the average primary particle diameter is 5 to 300 nm.
- the additive element C in the iron raw material used is an organic substance capable of reducing Fe 3+ to Fe 2+ in an inert gas atmosphere having an oxygen concentration of 0.1% or less.
- a method for producing a lithium iron phosphate particle powder having an olivine structure described in any one of the present invention (Invention 4).
- the present invention provides at least one selected from conductive carbon, an organic substance capable of reducing Fe 3+ to Fe 2+ , and an organic binder in the middle of the second step or immediately before the third step.
- Step A is mixed as an electron conduction assistant for the lithium iron phosphate particle powder to be produced, a reducing agent for Fe 3+ contained in the iron raw material to Fe 2+ or a precursor aggregate particle size control agent of 0.3 to 30 ⁇ m.
- a method for producing an olivine-type lithium iron phosphate particle powder according to any one of the present inventions 1 to 4 (Invention 5).
- the obtained lithium, iron and phosphorus main content is re-ground and re-precise mixed, and then the re-precise mixture is added to conductive carbon, Fe 3+ to Fe 2+ .
- At least one selected from organic substances having a reducing ability and organic binders is remixed and remixed at a temperature of 250 to 750 ° C. in an inert gas or reducing gas atmosphere having an oxygen concentration of 0.1% or less. 6.
- a method for producing a lithium iron phosphate particle powder having an olivine structure according to any one of the first to fifth aspects of the present invention, wherein firing is performed (Invention 6).
- the solid content concentration of the raw materials is 30 wt% or more.
- the olivine-type structure in which the slurry is adjusted so that 1 to 25 wt% is added to LiFePO 4 that produces ascorbic acid or sucrose and mixed at 50 ° C. or lower to adjust the pH of the raw slurry to 4 to 8 This is a method for producing a lithium iron phosphate particle powder (Invention 7).
- the present invention also relates to a lithium iron phosphate particle powder having an olivine type structure, wherein the lithium and phosphorus contents are 0.95 to 1.05 in terms of a molar ratio to iron, and Fe 3+ Impurity crystals other than the olivine structure are less than 5 mol%, have a BET specific surface area of 6 to 30 m 2 / g, have a residual carbon content of 0.5 to 8 wt%, have a residual sulfur content of 0.08 wt% or less.
- the amount of the phase Li 3 PO 4 is 5 wt% or less, the crystallite size is 25 to 300 nm, the aggregate particle diameter is 0.3 to 20 ⁇ m, and the compression molding density is 2.0 to 2.8 g / cc.
- This is an olivine type lithium iron phosphate particle powder characterized by having an electrical resistivity of 1 to 1.0 ⁇ 10 5 ⁇ ⁇ cm (Invention 8).
- the olivine type lithium iron phosphate particle powder according to the present invention 8 is combined with 0.1 to 10 wt% carbon of a conductive auxiliary agent and 1 to 10 wt% of a binder. It is a secondary battery positive electrode material sheet having a density of 8 g / cc or more (Invention 9).
- the present invention is a secondary battery manufactured using the secondary battery positive electrode material sheet according to the present invention 9 (Invention 10).
- the method for producing olivine-type lithium iron phosphate particles according to the present invention is low in cost and can be produced with a small environmental load, and the powder obtained by the method has a defect structure due to uniform solid solution of additive elements or surface modification. Therefore, the movement of electrons and Li ions is facilitated, and since the aggregated particles are controlled, the filling property is high.
- a secondary battery using it as a positive electrode active material has a high capacity in terms of current load characteristics, and can be used repeatedly for charging and discharging sufficiently.
- the olivine-type LiFePO 4 composite oxide powder according to the present invention has a compactness of 2.0 g / cc or more at 0.5 t / cm 2 or more, so that the filling property is improved. , Energy density per volume can be improved.
- the LiFePO 4 powder having an olivine structure according to the present invention has a lithium and phosphorus content of 0.95 to 1.05 mol ratio to iron, Fe 3+ is less than 5 mol% with respect to the Fe amount, BET Specific surface area of 6-30 m 2 / g, residual carbon content of 0.5-8 wt%, residual sulfur content of 0.08 wt% or less, amount of impurity crystal phase Li 3 PO 4 other than olivine structure is 5 wt% or less, crystallite size 25 ⁇ 300nm, Agglomerated particle size 0.3 ⁇ 20 ⁇ m, compression molding density 2.0 ⁇ 2.8g / cc, powder electrical resistivity 1 ⁇ 10 5 ⁇ ⁇ cm Current load characteristics and charge / discharge repetition characteristics during battery charge / discharge can be improved.
- the LiFePO 4 powder having an olivine structure according to the present invention is suitable as a positive electrode active material for a non-aqueous solvent secondary battery.
- FIG. 2 is a secondary electron image of a Fe 3 O 4 iron raw material shown in Table 1 by a scanning electron microscope. It is the reflected electron image by the scanning electron microscope of the lithium, phosphorus, and iron element containing precursor after the 2nd process obtained in Example 1.
- FIG. 2 is a secondary electron image of the olivine-type lithium iron phosphate particle powder obtained in Example 1 by a scanning electron microscope.
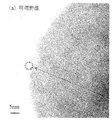
- 7 is a high-resolution TEM bright-field image of olivine-type lithium iron phosphate particle powder obtained in Example 5.
- FIG. 4 is a limited-field electron diffraction pattern inside the particles of lithium iron phosphate having an olivine structure obtained in Example 5.
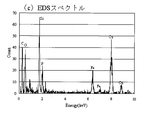
- FIG. 6 is a local elemental analysis EDS spectrum on the surface of lithium iron phosphate particles having an olivine structure obtained in Example 5.
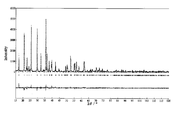
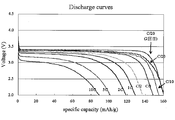
- FIG. 7 is a Rietveld analysis result of an X-ray diffraction pattern of olivine-type lithium iron phosphate particle powder obtained in Example 7. It is the discharge characteristic of the sheet number 2 of Table 5.
- the olivine-type lithium iron phosphate particle powder according to the present invention has solid solution or adsorption of additive elements such as elements Na, Mg, Al, Si, Cr, Mn, and Ni (hereinafter referred to as “foreign metal elements”).
- additive elements such as elements Na, Mg, Al, Si, Cr, Mn, and Ni (hereinafter referred to as “foreign metal elements”).
- the iron raw material, the lithium raw material and the phosphorus raw material can be uniformly and precisely mixed and obtained by appropriate heat treatment.
- the iron raw material in which different metal elements (Na, Mg, Al, Si, Cr, Mn, Ni) are dissolved is 0.1 to 1.8 mol / l of ferrous sulfate or ferric sulfate.
- a mixed solution of iron and a sulfate, nitrate, chloride, or organic substance containing a different metal element is prepared so as to have a predetermined molar ratio. Necessary while slowly supplying 0.1 to 18.5 mol / l aqueous alkali solution to the mixed solution to the stirred reaction vessel and maintaining the reaction vessel at a pH of 8 or higher and a temperature of room temperature to 105 ° C. Can be obtained by performing an air oxidation reaction.
- the generated iron oxide or hydrated oxide may be adsorbed with an additive element-containing sulfate, nitrate, chloride, or organic substance in a predetermined molar ratio.
- organic substances include carboxylates, alcohols, saccharides, and the like that are easily incorporated into or adsorbed into the iron oxide or hydrated oxide to be generated.
- NaOH, Na 2 CO 3 , NH 4 OH, ethanolamine or the like is used as an alkali source, and filtration washing or decantation washing is performed to remove impurity sulfate ions and control the composition ratio of the additive to Fe.
- the apparatus include a press filter and a filter thickener.
- the reaction concentration, temperature, pH, time, degree of air oxidation, etc. may be controlled.
- An iron raw material containing at least one of Fe 3 O 4 , ⁇ -FeOOH, ⁇ -FeOOH, and ⁇ -FeOOH having an average primary particle diameter of 5 to 300 nm is obtained.
- (NH 4 ) H 2 PO 4 , (NH 4 ) 2 HPO 4 is a coprecipitation method using NH 4 OH with H 3 PO 4
- LiH 2 PO 4 is LiOH or LiOH ⁇ nH 2 O aqueous solution of H 3 PO 4 solution
- Li 3 PO 4 is obtained by a coprecipitation method by mixing Li 3 or LiOH ⁇ nH 2 O aqueous solution of H 3 PO 4 solution.
- the average particle size of these lithium raw material and phosphorus raw material is preferably 10 ⁇ m or less, and a predetermined amount is mixed with the iron raw material described above so as to obtain a lithium iron phosphate particle powder having a predetermined olivine structure (first step). .
- Equipment includes Henschel mixer, Raiki machine, and high speed mixer.
- the mixture obtained in the first step is adjusted so that the aggregated particle diameter is 0.3 to 5.0 ⁇ m (second step).
- the Fe element is present at a ratio of 19/20 or more in a 2 ⁇ m ⁇ 2 ⁇ m visual field excluding voids.
- the adjustment method in the second step is a precision mixing mainly with an iron raw material accompanied by pulverization of a lithium raw material and a phosphorus-containing raw material, and a ball mill, a vibration mill, or a medium stirring mill is used.
- a ball mill, a vibration mill, or a medium stirring mill is used.
- LiFePO 4 obtained after the third step undergoes grain growth, and satisfactory battery characteristics cannot be obtained.
- LiFePO 4 obtained after the third step does not undergo grain growth unless the Fe element is present in a 2 ⁇ m ⁇ 2 ⁇ m visual field excluding voids at a ratio of 19/20 or more.
- satisfactory battery characteristics cannot be obtained.
- the proportion of 19/20 or more empirically, preferably LiFePO 4 is produced.
- the agglomerated particle diameter is controlled by the organic substance added in the first step and appropriate dry compaction.
- the aggregated particle diameter of the olivine-type LiFePO 4 powder according to the present invention is the aggregated particle diameter of the obtained olivine-type lithium iron phosphate particle powder before and after firing, that is, after the second and third steps. There is almost no change and it is necessary to adjust in the second step.
- the precursor obtained in the second step is baked at a temperature of 250 to 750 ° C. in an inert gas or reducing gas atmosphere having an oxygen concentration of 0.1% or less (third step).
- Examples of the apparatus include a gas flow type box muffle furnace, a gas flow type rotary furnace, and a fluidized heat treatment furnace.
- As the inert gas N 2 , Ar, H 2 O, CO 2 or a mixed gas thereof is used.
- As the reducing gas, H 2 , CO, or a mixed gas of these gases and the inert gas is used.
- LiFePO 4 Since Fe 3+ contained in the Fe raw material is changed to Fe 2+ by the additive element C or reducing gas and LiFePO 4 is generated, it is necessary to perform firing in an atmosphere having an oxygen concentration of 0.1% or less. Empirically, LiFePO 4 is sufficiently produced at 350 ° C. or higher, but heat treatment is performed at 400 to 700 ° C. for several hours in order to simplify the solid-phase reaction and to form a graphite phase with high electron conductivity of the additive element C. It is preferable to carry out.
- raw materials containing Fe 3+ tend to grow more easily in the firing process than raw materials containing Fe 2+ .
- the preferred average primary particle size is 30 to 250 nm.
- Li, Fe, P raw material composition ratio and composition ratio of the additive element with respect to Fe are almost the same as those obtained in the first step, with the exception of additive element C, which hardly changes before and after the heat treatment. Is the ratio.
- the additive element C may be reduced to less than 50% by reduction heat treatment of Fe 3+ to Fe 2+ , and it is necessary to measure the amount of C remaining in each firing condition in advance and adjust it in the first step (this Inventions 1 to 3).
- the first step of mixing the raw materials in the present invention it is preferable to mix the raw materials in an aqueous solvent, and it is more preferable to adjust the slurry concentration so that the solid content concentration of the raw material is 30 wt% or more.
- the first step it is preferable to add 1 to 25 wt% of ascorbic acid or sucrose with respect to LiFePO 4 to be produced.
- ascorbic acid or sucrose By adding ascorbic acid or sucrose to the slurry, the reaction of Li, Fe, and P is promoted, compositional segregation is reduced during drying, and a heterogeneous phase is less likely to be generated after firing.
- the amount of ascorbic acid or sucrose added is less than 1 wt%, the effect of adding does not appear. If it exceeds 25 wt%, the heterogeneous precipitation cannot be effectively reduced. More preferably, it is 2 to 10 wt%.
- the reaction temperature in the first step is preferably 50 ° C. or lower. When the temperature during mixing exceeds 50 ° C., it is difficult to obtain an olivine single phase. More preferably, it is from room temperature to 45 ° C, and even more preferably from 25 to 43 ° C.
- the pH of the slurry in the first step is adjusted to 4-8.
- the pH is less than 4, P ions are present in the solution, segregate during drying, and a heterogeneous phase is likely to be generated after firing.
- the pH exceeds 8, it is difficult in principle, more preferably 4.5 to 6.5.
- an organic substance having a high reducing ability is included to positively promote organic reduction of Fe 3+ to Fe 2+ .
- An iron raw material can be used, and the amount thereof is adjusted so that the amount of residual carbon is 0.5 to 8.0 wt% or less with respect to the generated lithium iron phosphate particle powder.
- the organic substance having a high reducing ability carboxylate, alcohols, and saccharides that are easily taken into or adsorbed by iron oxide or hydrous oxide are preferable. However, it is necessary to be careful in handling the organic substance having a high reducing ability so as to reduce it by firing without reducing the iron raw material (Invention 4).
- the compression molded body of LiFePO 4 powder having an olivine structure obtained at a low temperature firing of 400 to 500 ° C. satisfies the electrical resistivity of 1 to 10 5 ⁇ ⁇ cm and exhibits high performance secondary battery characteristics.
- the organic substance having a high reducing ability when an inert gas having an oxygen concentration of 0.1% or less is used in the heat treatment in the third step, in order to positively promote organic reduction of Fe 3+ to Fe 2+ , It is possible to add the organic substance having a high reducing ability before the three steps.
- the amount thereof can be adjusted so that the residual carbon amount is 0.5 to 8 wt% or less with respect to the lithium iron phosphate particle powder to be produced.
- resin powder such as polyethylene is used as the organic substance.
- the agglomerated particle diameter of the olivine-type LiFePO 4 composite oxide powder obtained in the present invention hardly changes before and after the third step, that is, firing, as described above. Therefore, the aggregate particle diameter of the precursor is adjusted to 0.3 to 30 ⁇ m by adding an organic binder during the second step or before the third step, and after firing, the aggregate is 0.3 to 30 ⁇ m. It is possible to obtain LiFePO 4 having a particle size.
- the precursor aggregate particle size control agent having a particle size of 0.3 to 30 ⁇ m is, for example, organic binders such as polyvinyl alcohol and sucrose.
- the conductive carbon, the reducing agent during the firing, and the precursor aggregate particle size controlling agent can be added,
- the amount of residual carbon is adjusted to 0.5 to 8 wt% with respect to the lithium iron phosphate particle powder (Description of Step A of Invention 5).
- the local gas concentration distribution may affect the quality due to the generation of water vapor and the generation of oxidizing gas accompanying the precursor Fe 3+ reduction. Therefore, it is also possible to carry out heat treatment (main firing) again by performing so-called calcination, mixing the carbon-containing additive, pulverizing and mixing again.
- the calcination temperature is preferably as low as about 250 to 500 ° C.
- the main calcination temperature is preferably as high as 400 to 750 ° C.
- the order of operations between calcination and main calcination is not particularly limited.
- the carbon-containing additive added before the second heat treatment is conductive carbon, an organic reducing agent, and a binder for controlling the aggregated particle size of the precursor, and at least one of these is mixed. It is also possible (description of step A of the present invention 6).
- FIG. 1 shows a flowchart of a method for producing olivine type lithium iron phosphate particles in the present invention.
- the composition of the LiFePO 4 powder having an olivine structure according to the present invention is Li x FeP y O 4 (0.95 ⁇ x, y ⁇ 1.05).
- x and z are out of the above ranges, it is easy to form a heterogeneous phase, and in some cases, grain growth is promoted, and LiFePO 4 having high battery characteristics cannot be obtained.
- the contents of the different metal elements are preferably 0.1 to 2 mol% with respect to Fe.
- the mol% of Fe 3+ / Fe in the olivine-type LiFePO 4 composite oxide powder according to the present invention is less than 5%. It is known that LiFePO 4 produced after firing is oxidized by exposure to air to form an Fe 3+ amorphous phase. The formed Fe 3+ compound does not contribute to charging / discharging of the secondary battery, and forms a dendrite at the negative electrode and promotes an internal short circuit of the electrode, and it is necessary to reduce it as much as possible.
- the BET specific surface area of the LiFePO 4 powder having an olivine structure according to the present invention is 6 to 30 m 2 / g.
- the BET specific surface area value is less than 6 m 2 / g, the movement of Li ions in LiFePO 4 is slow, so that it is difficult to take out current.
- it exceeds 30 m ⁇ 2 > / g, since the fall with the packing density of a positive electrode and the reactivity with electrolyte solution increase, it is unpreferable.
- it is 8 to 28 m 2 / g, more preferably 9 to 25 m 2 / g.
- the residual carbon content of the olivine-type LiFePO 4 composite oxide powder according to the present invention is 0.5 to 8.0 wt%.
- the carbon content is less than 0.5%, the particle growth during the heat treatment cannot be suppressed, and the electric resistance of the obtained powder is increased, which deteriorates the charge / discharge characteristics of the secondary battery.
- it exceeds 8.0% the positive electrode packing density decreases, and the energy density per volume of the secondary battery decreases. More preferably, it is 0.6 to 6.0%.
- the LiFePO 4 composite oxide powder having an olivine structure according to the present invention has an impurity sulfur content of 0.08 wt% or less, and good storage characteristics can be obtained in a non-aqueous electrolyte secondary battery.
- impurities such as lithium sulfate are formed, and these impurities undergo a decomposition reaction during charge and discharge, and the reaction with the electrolyte during high-temperature storage is promoted, and after storage Resistance rises intensely. More preferably, it is 0.05 wt% or less.
- the crystal phase of Li 3 PO 4 may be detected at 5 wt% or less other than the olivine structure.
- Li 3 PO 4 is detected, sometimes LiFePO 4 particles obtained by solid phase reaction may be fine, resulting in a high discharge capacity.
- Li 3 PO 4 itself does not contribute to charging / discharging, 5 wt% or less is desirable.
- the crystallite size of the LiFePO 4 powder having an olivine structure according to the present invention is 25 to 300 nm. It is extremely difficult to mass-produce powders of 25 nm or less while satisfying other powder characteristics, and it takes time for Li to move at a crystallite size of 300 nm. The current load characteristic of the secondary battery is deteriorated.
- the thickness is preferably 30 nm to 200 nm, more preferably 40 nm to 150 nm.
- the aggregated particle diameter of the LiFePO 4 powder having an olivine structure according to the present invention is 0.3 to 30 ⁇ m.
- the thickness is less than 0.3 ⁇ m, the positive electrode packing density is decreased and the reactivity with the electrolytic solution is increased.
- it is extremely difficult to mass-produce powders exceeding 30 ⁇ m while satisfying other powder characteristics by the production method.
- it is 0.5 to 15 ⁇ m.
- the density of the compression molded body of the LiFePO 4 powder having an olivine structure according to the present invention is preferably 2.0 g / cc or more.
- the true density of the general-purpose layered compound LiCoO 2 is 5.1 g / cc, whereas the true density of LiFePO 4 is as low as 3.6 g / cc. Therefore, a preferable compression-molded body density is 2.0 g / cc or more which is 50% or more of the true density, and the closer to the true density, the better the filling property.
- the powder electrical resistivity of the LiFePO 4 powder having an olivine structure according to the present invention is 1 to 10 5 ⁇ ⁇ cm, more preferably 10 to 5 ⁇ 10 4 ⁇ ⁇ cm.
- a conductive agent and a binder are added and mixed according to a conventional method.
- the conductive agent acetylene black, carbon black, graphite and the like are preferable, and as the binder, polytetrafluoroethylene, polyvinylidene fluoride and the like are preferable.
- the solvent for example, N-methyl-pyrrolidone is used, and the positive electrode active material sieved to 45 to 105 ⁇ m or less and the slurry containing the additive are kneaded until they become honey. The obtained slurry is applied onto the current collector with a doctor blade having a groove of 25 ⁇ m to 500 ⁇ m.
- the coating speed is about 60 cm / sec, and an Al foil of about 20 ⁇ m is usually used as a current collector.
- drying is performed at 80 to 180 ° C. in a non-oxidizing atmosphere of Fe 2+ .
- the sheet is subjected to a calender roll treatment so as to have a pressure of 1 to 3 t / cm 2 .
- an oxidation reaction of Fe 2+ to Fe 3+ occurs even at room temperature.
- the density of the positive electrode made of the positive electrode active material, carbon, and binder on the current collector of the obtained positive electrode sheet is 1.8 g / cc or more.
- the density of the compression molded body of the positive electrode active material is as high as 2.0 g / cc or more, and the electrical resistivity of the compression molded body of the positive electrode active material is 1 to 10 5 ⁇ ⁇ cm.
- the amount of carbon added during sheet preparation can be suppressed because it is low, and the amount of binder added can be suppressed because the BET specific surface area of the positive electrode active material is as low as 6 to 30 m 2 / g, resulting in a positive electrode sheet with high density. Is obtained.
- the negative electrode active material lithium metal, lithium / aluminum alloy, lithium / tin alloy, graphite or the like can be used, and the negative electrode sheet is produced by the same doctor blade method as that of the positive electrode.
- an organic solvent containing at least one of carbonates such as propylene carbonate and dimethyl carbonate and ethers such as dimethoxyethane can be used as the solvent for the electrolytic solution.
- At least one lithium salt such as lithium perchlorate and lithium tetrafluoroborate can be dissolved in the above solvent and used.
- the secondary battery manufactured using the positive electrode sheet according to the present invention has a discharge capacity of 150 mAh / g or more at C / 10 at room temperature and a capacity deterioration rate of less than 10% in the 50 charge / discharge repetition characteristics.
- the discharge capacity at 1 C is 120 mAh / g or more, the capacity deterioration rate in the 50 charge / discharge repetition characteristics is less than 5%, and the discharge capacity at 5 C at room temperature is 80 mAh / g or more.
- the capacity deterioration rate is defined as (C 50 ⁇ C 1 ) / C 1 ⁇ 100, which is the discharge capacity C 50 obtained in the 50th charge / discharge cycle from the discharge capacity C 1 obtained in the first charge / discharge cycle. It is represented by Discharge capacity C n from C n + 1 (n is an integer) value changes continuously, it is confirmed with a validity evaluation.
- C / 20 is a current value fixed so that a current of LiFePO 4 theoretical capacity of 170 mAh / g flows in 20 hours, and 5C is a current of LiFePO 4 theoretical capacity of 170 mAh / g in 1/5 hours.
- the current value is fixed as follows.
- a higher C coefficient means higher current load characteristics.
- the current value at the time of charging is not particularly limited, in the present invention, it was confirmed that a theoretical capacity was almost obtained using a constant current value of C / 20.
- the voltage range during charging and discharging is not particularly limited, but in the present invention, the voltage range was 2.0 to 4.5V.
- the LiFePO 4 powder having an olivine structure according to the present invention uses an inexpensive and stable iron raw material containing Fe 3+ , it can be manufactured at low cost and with a small environmental load.
- the secondary battery characteristics are satisfied in order to satisfy the powder characteristics described in the present invention 7, and in particular, since the modifying element and the solid solution element are controlled, a high capacity is obtained even in the current load characteristics, and sufficient charge / discharge is achieved. The inventor presumes that it can be used repeatedly.
- a typical embodiment of the present invention is as follows.
- the Fe concentration of the iron raw material is quantified by titration (JIS K5109), and an X-ray diffractometer RINT-2500 [manufactured by Rigaku Corporation] is used to identify the crystal phase. It was carried out at 40 kV and 300 mA, and it was confirmed that there was no crystallized additive element.
- Element C added to the iron raw material was quantified by burning the iron raw material in an oxygen stream in a combustion furnace using EMIA-820 [manufactured by Horiba Seisakusho].
- Elements Na, Mg, Al, Si, Ca, Ti, Cr, Mn, Co, Ni and Zn added in addition to Li, Fe, P main elements and C are the emission plasma analyzer ICAP-6500 [Thermo Fisher Scientific Measured using
- a Hitachi S-4800 scanning electron microscope (SEM) was used to calculate the average primary particle size of the iron raw material, and the short axis and long axis of about 200 particles that could be judged from the obtained images were measured and averaged. The primary particle size was calculated. Since the ratio of the major axis to the minor axis differs greatly only for ⁇ -FeOOH, the aspect ratio was calculated.
- Table 1 shows the characteristics of the iron raw materials used in the present invention.
- the aspect ratio (major axis diameter / minor axis diameter) of iron raw material number 4 of ⁇ -FeOOH was 5, and the aspect ratio (major axis diameter / minor axis diameter) of iron raw material number 5 was 2.5.
- the Li and P concentrations of the lithium and phosphorus-containing main raw materials were measured by neutralization titration using a pH meter and hydrochloric acid or NaOH reagent.
- the impurity element concentration contained in the main raw material containing lithium and phosphorus was such a concentration that the above-mentioned emission plasma analyzer was used and the addition amount could be corrected without affecting the present invention.
- the scanning electron microscope SEM was used to determine that Fe element was present at a ratio of 19/20 or more in a 2 ⁇ m ⁇ 2 ⁇ m visual field excluding voids.
- HELOS manufactured by Nippon Laser Co., Ltd.
- a dry laser diffraction / scattering particle size distribution meter was used for the measurement of the aggregate particle size of the precursor or olivine type lithium iron phosphate particles.
- used was quantified with a median diameter D 50.
- the lithium iron phosphate particles having an olivine structure manufactured according to the present invention were dissolved in an acid solution at 200 ° C. using an autoclave for sample dissolution.
- the above-mentioned emission plasma analyzer was used for the contents of lithium and phosphorus with respect to iron.
- Rietveld analysis of the X-ray diffraction pattern by the above-described apparatus and local elemental analysis by JEOL JEM-2010F high resolution TEM and its associated EDS were used.
- the X-ray diffraction pattern was measured in steps of 0.02 ° at 2.5 ° / min and 2 ⁇ in the range of 15 to 120 ° so that the maximum peak intensity count number was 5000 to 8000.
- Rietan 2000 was used as the Rietveld analysis program.
- the TCH pseudo-void function is used as the profile function, the method such as Finger is used for asymmetry of the function, and the reliability factor S value is 1.5. Analyzed to cut.
- This program was applied to the identification of impurity crystal phases other than the olivine structure, the quantification of impurity crystal phases Li 3 PO 4 other than the olivine structure, and the crystallite size quantification of LiFePO 4 particles of 80 nm or more.
- the crystallite size of LiFePO 4 particles of less than 80 nm calculation was made from the half-value width of the (101) plane X-ray diffraction pattern. The spectrum by EDS ended when the maximum peak intensity exceeded 60.
- the amount of Fe 3+ was calculated from the amount of Fe described above and Fe 2+ titration (JIS K1462) for quantification of Fe 3+ .
- the specific surface area is a specific surface area determined by a BET one-point continuous method using MONOSORB [manufactured by Yuasa Ionics Co., Ltd.] after drying and deaeration of the sample under nitrogen gas at 120 ° C. for 45 minutes.
- the amount of residual sulfur was quantified using the carbon and sulfur measuring device EMIA-820 [manufactured by Horiba Ltd.] and applied to the amount of residual carbon.
- the density of the compression-molded body was compacted to 1.5 t / cm 2 with a 13 mm ⁇ jig and calculated from the weight and volume. Simultaneously, the powder electrical resistivity was measured by the two-terminal method.
- the carbon of the conductive auxiliary agent used is acetylene black, ketjen black and graphite KS-6.
- the binder used was polyvinylidene fluoride having a polymerization degree of 540,000 (manufactured by Aldrich) and was dissolved in N-methylpyrrolidone (manufactured by Kanto Chemical Co., Inc.).
- a CR22032 type coin cell (manufactured by Hosen Co., Ltd.) was prepared using the electrolytic solution (manufactured by Kishida Chemical Co., Ltd.) mixed in 3: 7).
- ball mixed powder and a prescribed amount of acetylene black obtained in the first step made ZrO 2 - mill vessel to the slurry - to have a concentration 30 wt% ethanol - Adjust Le, ZrO 2 ball of 5 mm.phi - Le
- the mixture was pulverized for 24 hours, mixed finely, and the slurry at room temperature was dried (solvent removed) to obtain a precursor.
- the secondary electron image of the iron raw material used here is shown in FIG. 2, and the reflected electron image of the obtained precursor is shown in FIG.
- the iron raw material used had an average primary particle size of 200 nm. 24 squares of 2 ⁇ m ⁇ 2 ⁇ m were added to the reflected electron image of FIG. 2, and it was confirmed that the Fe element was present in the field of view excluding voids in the squares.
- the obtained precursor had an agglomerated particle diameter D 50 of 1.4 ⁇ m (step A, second step).
- the obtained precursor was put into an alumina crucible and subjected to the heat treatment described in Table 2. That is, the temperature rising rate was 200 ° C./hr, the reached temperature was 500 ° C., the reached temperature holding time was 2 hours, the gas used was 95% N 2 -5% H 2 , and the gas flow rate was 1 L / min (third step).
- Table 3 shows the powder characteristics of the obtained powder.
- the obtained powder is fine, has an olivine structure, is not different from the composition ratio of Li, Fe, P prepared in the first step, and the composition ratio of all additive elements and Fe except additive element C Matched within a measurement error range of 3%.
- FIG. 4 shows an SEM photograph (secondary electron image) of the obtained lithium iron phosphate particles having an olivine structure.
- Examples 2, 3, and 8 The experiment was conducted with the contents shown in Table 2. Parts not described were performed in the same manner as in Example 1. However, a predetermined amount of the carbon-containing additive was blended in a dry ball mill after the second step. Table 3 shows the powder characteristics of the obtained lithium iron phosphate particles having an olivine structure. In the fine powder having an olivine structure, the composition ratios of all additive elements and Fe except for the composition ratio of Li, Fe, and P and additive element C were within the measurement error range of 3%, as in Example 1. Matched.
- Examples 4, 5, and 7 A predetermined amount of the main raw material was mixed in a wet (water solvent) ball mill so that 150 g of lithium iron phosphate particle powder was formed, and then dried at 70 ° C. for 12 hours. Lithium and phosphorus-containing main raw materials Li 3 PO 4 and H 3 PO 4 were used (first step).
- step A The dried product and a predetermined amount of carbon-containing additive were pulverized with a 5 mm ⁇ ZrO 2 dry ball mill for 24 hours (step A, second step), and calcined in a nitrogen atmosphere at 400 ° C. for 2 hours (third) Process). After pulverizing and mixing with a dry ball mill, heat treatment was performed again at 650 ° C. for 2 hours in a nitrogen atmosphere (operation A).
- Table 3 shows the powder characteristics of the obtained olivine-type lithium iron phosphate particles.
- the composition ratios of all additive elements and Fe except for the composition ratio of Li, Fe, and P and additive element C were within the measurement error range of 3%, as in Example 1. Matched.
- FIG. 5 to 7 show the high-resolution TEM bright-field image (FIG. 5), the limited-field electron diffraction pattern (FIG. 6), and the local elemental analysis EDS spectrum (FIG. 7) obtained in Example 5.
- FIG. 8 shows the Rietveld analysis result of the X-ray diffraction pattern of Example 7.
- the dotted line shows the measured diffraction pattern
- the curve shows the calculated diffraction pattern
- the bottom horizontal straight waveform shows the measured and calculated diffraction patterns. This means that the closer to the straight line, the more the measured value and the calculated value match.
- the same analysis was performed on all the samples of Examples 1 to 8. No impurity crystal phase other than Li 3 PO 4 was observed, and crystalline compound segregation due to the additive element was not confirmed.
- Example 6 As shown in Table 2, experiments similar to those in Examples 4, 5, and 7 were performed. However, the predetermined amount of the carbon-containing additive was performed after the second step, and the heat treatment was performed once without performing calcination, pulverization, and mixing (operation A) after firing. It was kept at 400 ° C. for 2 hours in hydrogen, then switched to N 2 and kept at 650 ° C. for 2 hours. In the fine powder having an olivine structure, the composition ratio of all additive elements and Fe except for the composition ratio of Li, Fe, P and additive element C is within the measurement error range of 3%, as in the other examples. Matched.
- the electrode slurry was applied onto the Al foil current collector with a doctor blade having a gap of 100 ⁇ m. Sheet - After preparative drying, was punched into 3t / cm 2 pressurized to 2 cm 2.
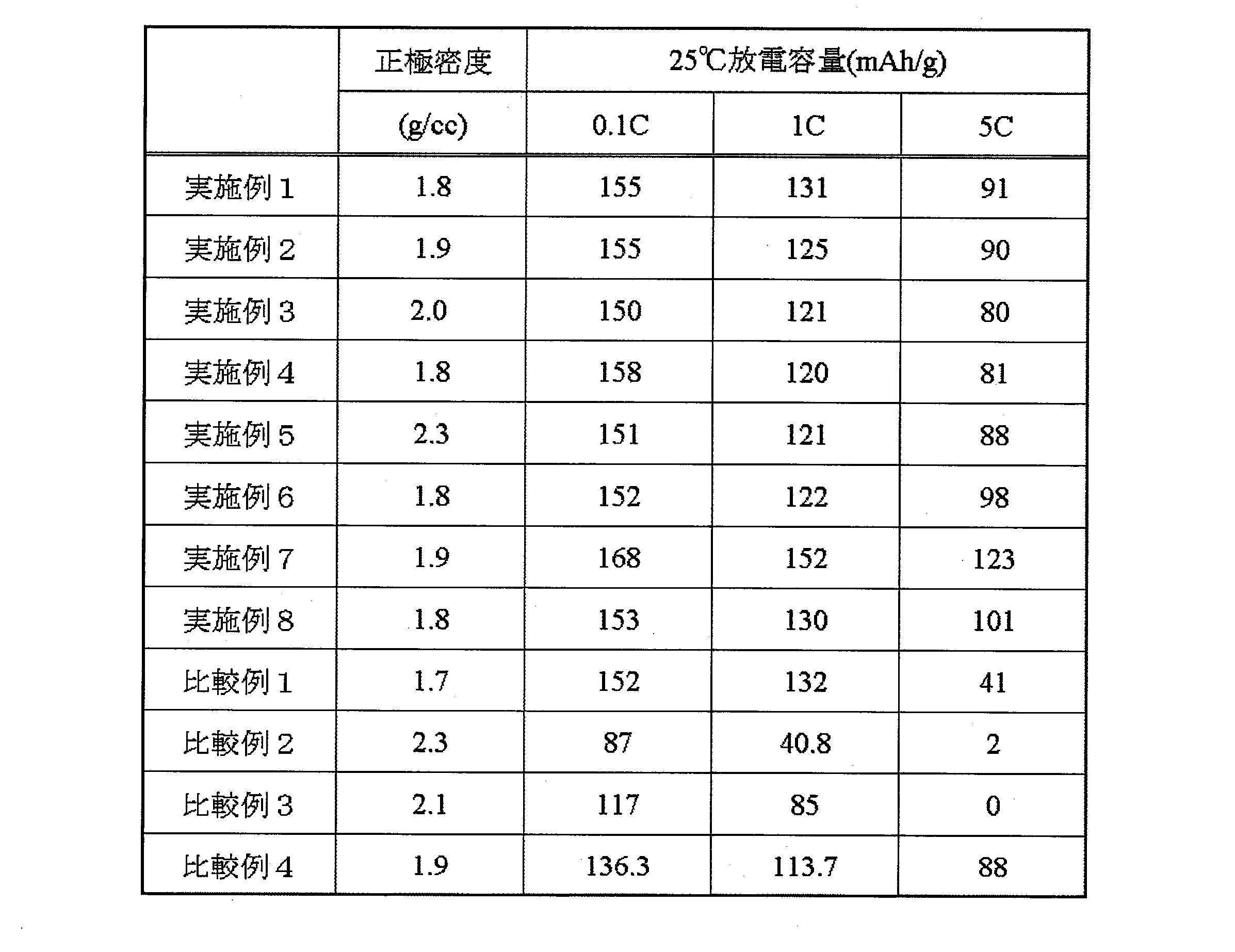
- Table 4 shows the density of the positive electrode on the current collector. Table 4 also shows the characteristics of the secondary battery using this sheet as the positive electrode.
- the lithium iron phosphate particles having an olivine structure according to the present invention satisfy high cathode density and secondary battery characteristics.
- Comparative Example 1 in which the positive electrode active material powder had a low compression molding density had a low positive electrode density. The reason why the discharge capacity at 5C was low is thought to be because the influence of the additive did not appear. In Comparative Examples 2 to 4 having a large crystallite size, almost all the discharge capacities were low. Comparative Example 4 has a high capacity deterioration rate, and it is considered that the surface modification and uniform solid solution of the additive element were insufficient.
- the secondary battery according to the present invention has an excellent capacity maintenance rate.
- the film thickness and density of the positive electrode when the electrode composition ratio of the active material obtained in Example 5 was changed to form a sheet and the characteristics of the secondary battery are shown.
- the thickness of the positive electrode was a value obtained by subtracting the thickness of the Al foil current collector contained in the positive electrode sheet, and was adjusted by the amount of solvent added at the time of coating and the depth of the groove of the doctor blade.
- the carbon added was acetylene black and KS-6 graphite in a weight ratio of 1: 1.
- the amount of PVDF and carbon increased, the positive electrode density decreased.
- high secondary battery characteristics were obtained with all these electrode compositions.
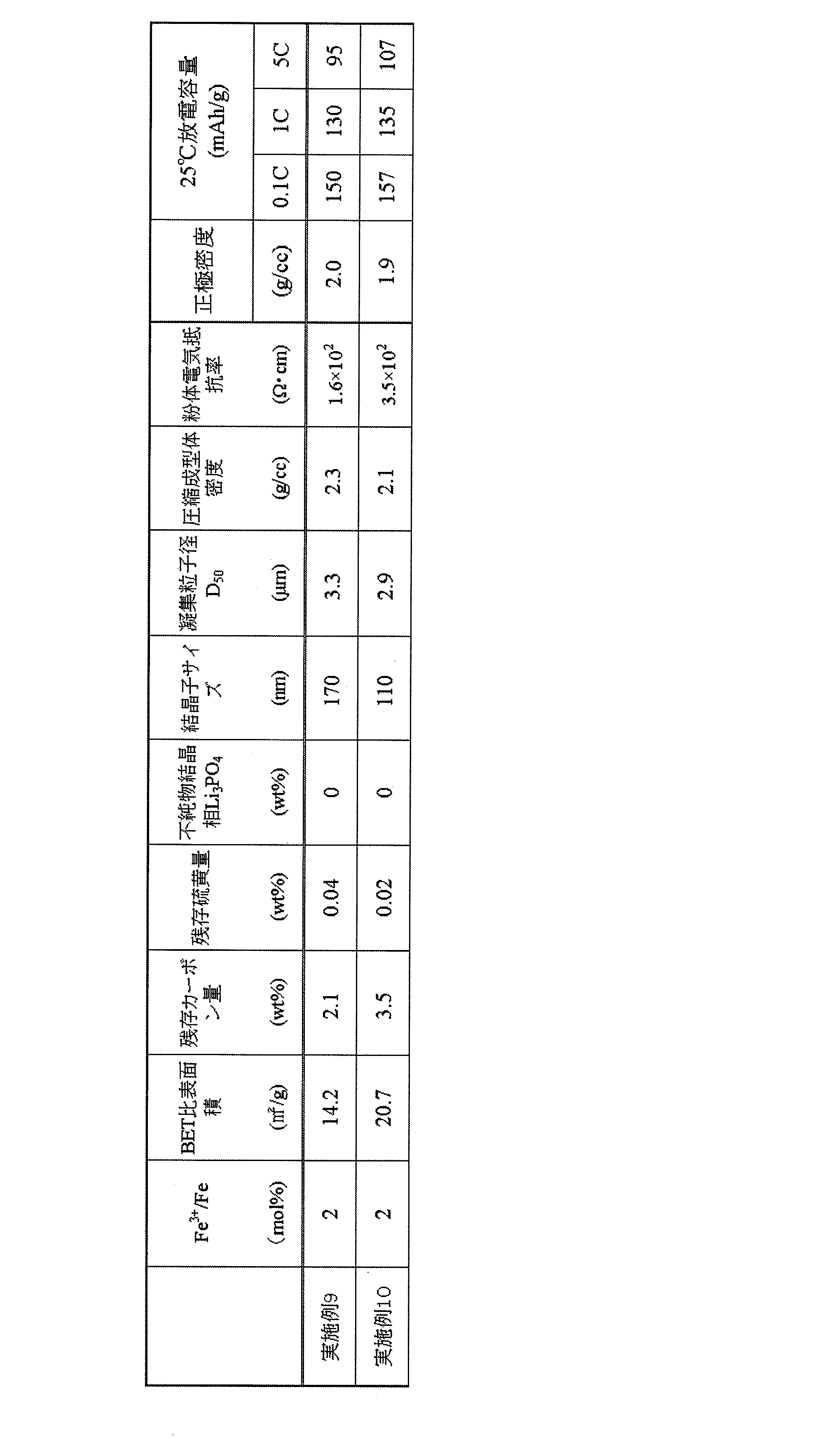
- Example 10 In the same Li, P, and Fe raw material slurry as in Example 6 (the solid content concentration was adjusted to 50 wt%), 5 wt% and 5 wt% sucrose were similarly added to LiFePO 4 that produces ascorbic acid, and room temperature ( After reaction for 1 day at 25 ° C.), a paste containing Li, P and Fe was obtained. After drying at 70 ° C., the second and third steps were performed in the same manner as in Example 6. In the precision mixture after the second step, the presence of Fe element in the 2 ⁇ m ⁇ 2 ⁇ m field of view was 19/20 or more by SEM observation, and the precursor aggregated particle diameter D 50 was 3.1 ⁇ m.
- Table 6 shows the powder characteristics and battery characteristics of the olivine-type lithium iron phosphate obtained in Examples 9 and 10. Battery characteristics were evaluated by producing coin cells by the same method as described in Table 4.
- the method for producing olivine-type lithium iron phosphate particle powder according to the present invention is a low-cost production method with little environmental load.
- the olivine-type lithium iron phosphate particle powder according to the present invention can produce a highly filled positive electrode sheet, and a secondary battery using the same has a high capacity in terms of current load characteristics. It has been confirmed that charging and discharging can be sufficiently repeated.
- the present invention uses an olivine-type lithium iron phosphate particle powder produced by a low-cost and low environmental load manufacturing method as a positive electrode active material for a secondary battery, resulting in a high energy density per volume and a high current load.
- a high capacity can be obtained, and a non-aqueous solvent secondary battery that can be sufficiently charged and discharged repeatedly can be obtained.
Abstract
Description
有機物としては、生成する鉄酸化物又は含水酸化物に取り込まれやすい若しくは吸着しやすいカルボン酸塩、アルコール類、糖類等がある。
本発明に係るオリビン型構造のLiFePO4粉末は安価で安定なFe3+を含む鉄原料を用いるため、低コストで、環境負荷が小さく製造できる。前記二次電池特性を満たすのは、本発明7記載の粉体特性を満たすため、特に、修飾元素と固溶元素制御したため、電流負荷特性においても高容量が得られ、且つ十分に充放電を繰返し使えると本発明者は推定している。
F. Izumi and T. Ikeda, Mater. Sci. Forum, 2000, Vol. 198, p.321-324.
表1記載の番号1の鉄原料を用いて、10gのリン酸鉄リチウム粒子粉末ができるようLiH2PO4を表2記載の仕込み比Li/Fe=1.01、P/Fe=1.01で、ライカイ機を用いて混合した(第一工程)。
表2記載の内容で実験を行った。記載の無い部分は実施例1と同様に行った。但し、所定量の炭素含有添加物は、第二工程後、乾式ボ-ルミルにて配合した。得られたオリビン型構造のリン酸鉄リチウム粒子粉末の粉体特性を表3に示す。微細で、オリビン型構造を有する該粉末において、実施例1と同様、Li、Fe、Pの組成比と添加元素Cを除く、すべての添加元素とFeの組成比は測定誤差範囲3%内で一致した。
150gのリン酸鉄リチウム粒子粉末ができるよう主原料を所定量湿式(水溶媒)ボ-ルミルにて混合後、70℃、12時間で乾燥した。リチウム及びリン含有主原料Li3PO4とH3PO4を用いた(第一工程)。
表2に記すよう、実施例4、5、7と同様の実験を行った。但し、所定量の炭素含有添加物は、第二工程後行い、また、焼成後の仮焼、粉砕、混合(A操作)を施すことなく、熱処理は1度で行った。水素中400℃、2時間保持し、その後、N2に切り替えて、650℃、2時間保持した。微細で、オリビン型構造を有する該粉末において、他の実施例と同様、Li、Fe、Pの組成比と添加元素Cを除く、すべての添加元素とFeの組成比は測定誤差範囲3%内で一致した。
表2に記す内容で、実施例1同様の第一工程、第二工程を経て、仮焼、粉砕、混合、カ-ボン源添加、再焼成を行った。微細な粒子径のリン酸リチウムが生成したものの、低い成型体密度であった。
表2に記す内容で、実施例4同様の第一工程を経て、該第二工程を経由せず、所定量の炭素含有添加物をライカイ機で混合後(工程A)、第三工程の熱処理を行った。得られた粉末はオリビン構造を有するものの、Fe3+不純物が多く、微細でなく、電気抵抗が高かった。
表2に記す内容で、実施例1同様の第一工程を経て、所定量の炭素含有添加物をライカイ機で混合後(工程A)、第二、第三工程を経て、粉砕、混合後、再焼成を行った(A操作)。第一工程におけるLi,Fe,P組成比を本発明の範囲外としたため、得られた粉末は比表面積が小さく、残存カ-ボンが少なく、不純物結晶相Li3PO4が多く、結晶子サイズも大きかった。
表2に記す内容で、実施例1同様の第一工程を経て、所定量の炭素含有添加物をライカイ機で混合後、第二工程を経由せず、第三工程を経てリン酸鉄リチウム粒子粉末を作製した。その粉体特性は残存硫黄が高く、粗大な粒子であった。
実施例6と同一のLi、P、Fe原料スラリ-(固形分濃度を35wt%に調整)に、アスコルビン酸を生成するLiFePO4に対し5wt%添加し、40℃、3時間反応後、pH=5のスラリ-を得た。70℃で乾燥後、第二、第三工程は実施例6と同様に行った。第二工程後の精密混合物はSEM観察で2μm×2μm視野内にFe元素の存在は19/20以上であり、前駆体凝集粒子径D50は3.5μmであった。
実施例6と同一のLi、P、Fe原料スラリ-(固形分濃度を50wt%に調整)に、アスコルビン酸を生成するLiFePO4に対し5wt%、ショ糖を同様に5wt%添加し、室温(25℃)で、1日間反応後、Li、P、Fe含有ペ-ストを得た。70℃で乾燥後、第二、第三工程は実施例6と同様に行った。第二工程後の精密混合物はSEM観察で2μm×2μm視野内にFe元素の存在は19/20以上であり、前駆体凝集粒子径D50は3.1μmであった。
Claims (10)
- 鉄原料として鉄酸化物又は含水酸化物を用い、元素Na、Mg、Al、Si、Cr、Mn、Niのうち少なくとも1種をFeに対し各々0.1~2mol%含み、且つ、元素CがFeに対して5~10mol%含み、Fe量に対しFe2+が40mol%以下で、平均一次粒子径が5~300nmの鉄酸化物又は含水酸化物を、リチウム原料及びリン原料とともに混合する第一工程、得られた混合物の凝集粒子径が0.3~5.0μmとなるように調整する第二工程、次いで、第二工程を経た混合物を酸素濃度0.1%以下の不活性ガス、又は還元性ガス雰囲気下で、温度250~750℃で焼成を行う第三工程からなるオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法。
- 用いる鉄原料が、元素Na、Mg、Al、Si、Cr、Mn、Niのうち少なくとも1種をFeに対し各々0.1~2mol%の量で含み、且つ当該7種の元素合計量がFeに対して1.5~4mol%となるように含み、且つ、元素CがFeに対して5~10mol%含み、且つ、平均一次粒子径が5~300nmのFe3O4、α-FeOOH、γ-FeOOH、δ-FeOOHのうち少なくとも1種を含む請求項1記載のオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法。
- 用いる鉄原料が、元素Na、Mg、Al、Si、Cr、Mn、Niのうち少なくとも1種をFeに対し各々0.1~2mol%の量で含み、且つ当該7種の元素合計量がFeに対して1.5~4mol%となるように含み、且つ、元素CがFeに対して5~10mol%含み、且つ、平均一次粒子径が5~300nmで長軸と短軸のアスペクト比が2以上の針状鉄原料である請求項2記載のオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法。
- 用いる鉄原料における添加元素Cが、酸素濃度0.1%以下の不活性ガス雰囲気下でFe3+をFe2+へ還元することが可能な有機物である請求項1~3の何れかに記載されたオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法。
- 第二工程の途中、或いは、第三工程の直前に、導電性カーボン、Fe3+のFe2+への還元能力のある有機物および有機系結着剤から選択される少なくとも1種を、生成するリン酸鉄リチウム粒子粉末の電子伝導補助剤、鉄原料に含まれるFe3+のFe2+への還元剤又は0.3~30μmの前駆体凝集粒子径制御剤として混合する工程Aを含む請求項1~4の何れかに記載のオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法。
- 第三工程の後、得られたリチウム、鉄およびリン主含有物を再粉砕し、再精密混合した後、当該再精密混合物に、導電性カーボン、Fe3+のFe2+への還元能力のある有機物および有機系結着剤から選択される少なくとも1種を再混合し、酸素濃度0.1%以下の不活性ガス又は還元性ガス雰囲気下で、温度250~750℃で再焼成を行う請求項1~5の何れかに記載のオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法。
- 請求項1~6の何れかに記載のオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法の原料を混合する第一工程において、原料の固形分濃度が30wt%以上となるようにスラリーを調整し、アスコルビン酸又はショ糖を生成するLiFePO4に対し1~25wt%添加して、50℃以下で混合を行い、原料スラリーのpHを4~8に調整するオリビン型構造のリン酸鉄リチウム粒子粉末の製造方法。
- オリビン型構造のリン酸鉄リチウム粒子粉末であって、リチウムとリンの含有量が鉄に対するmol比で各々0.95~1.05であり、Fe量に対しFe3+が5mol%未満であり、BET比表面積が6~30m2/gであり、残存カーボン量が0.5~8wt%であり、残存硫黄量が0.08wt%以下であり、オリビン構造以外の不純物結晶相Li3PO4の量が5wt%以下であり、結晶子サイズが25~300nmであり、凝集粒子径が0.3~20μmであり、圧縮成型体密度が2.0~2.8g/ccであり、粉体電気抵抗率が1~1.0×105Ω・cmであることを特徴とするオリビン型構造のリン酸鉄リチウム粒子粉末。
- 請求項8記載のオリビン型構造のリン酸鉄リチウム粒子粉末、0.1~10wt%の導電補助剤のカーボン及び1~10wt%の結着剤を複合化させた密度1.8g/cc以上の二次電池正極材シート。
- 請求項9記載の二次電池正極材シートを用いて作製した二次電池。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/935,456 US20110091772A1 (en) | 2008-03-31 | 2009-03-26 | Process for producing lithium iron phosphate particles, lithium iron phosphate particles having olivine type structure, and positive electrode sheet and non-aqueous solvent-based secondary battery using the lithium iron phosphate particles |
| EP09727078.9A EP2277828B1 (en) | 2008-03-31 | 2009-03-26 | Lithium iron phosphate powder manufacturing method, olivine structured lithium iron phosphate powder, cathode sheet using said lithium iron phosphate powder, and non-aqueous solvent secondary battery |
| CN2009801111827A CN101980956A (zh) | 2008-03-31 | 2009-03-26 | 磷酸铁锂颗粒粉末的制造方法、橄榄石型结构的磷酸铁锂颗粒粉末、使用该磷酸铁锂颗粒粉末的正极材料片和非水溶剂类二次电池 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008094056 | 2008-03-31 | ||
| JP2008-094056 | 2008-03-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009122686A1 true WO2009122686A1 (ja) | 2009-10-08 |
Family
ID=41135094
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/001374 WO2009122686A1 (ja) | 2008-03-31 | 2009-03-26 | リン酸鉄リチウム粒子粉末の製造方法、オリビン型構造のリン酸鉄リチウム粒子粉末、該リン酸鉄リチウム粒子粉末を用いた正極材シート及び非水溶媒系二次電池 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20110091772A1 (ja) |
| EP (1) | EP2277828B1 (ja) |
| JP (1) | JP5464322B2 (ja) |
| KR (1) | KR101587671B1 (ja) |
| CN (2) | CN101980956A (ja) |
| WO (1) | WO2009122686A1 (ja) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100239909A1 (en) * | 2008-10-22 | 2010-09-23 | Lg Chem, Ltd. | Cathode mix containing having improved efficiency and energy density of electrode |
| WO2011062232A1 (ja) * | 2009-11-18 | 2011-05-26 | 三井化学株式会社 | 電気化学セル用水性ペースト、該水性ペーストを塗布してなる電気化学セル用極板、および該極板を含む電池 |
| WO2011115211A1 (ja) * | 2010-03-19 | 2011-09-22 | 戸田工業株式会社 | リン酸マンガン鉄リチウム粒子粉末の製造方法、リン酸マンガン鉄リチウム粒子粉末、及び該粒子粉末を用いた非水電解質二次電池 |
| WO2011114918A1 (en) * | 2010-03-19 | 2011-09-22 | Semiconductor Energy Laboratory Co., Ltd. | Power storage device and manufacturing method thereof |
| JP2011210376A (ja) * | 2010-03-28 | 2011-10-20 | Niigata Univ | Liイオン電池用正極活物質およびその製造方法 |
| US20110269022A1 (en) * | 2010-04-28 | 2011-11-03 | Semiconductor Energy Laboratory Co., Ltd. | Positive electrode active material of power storage device, power storage device, electrically propelled vehicle, and method for manufacturing power storage device |
| US20110287315A1 (en) * | 2008-10-22 | 2011-11-24 | Lg Chem, Ltd. | Cathode active material providing improved efficiency and energy density of electrode |
| KR101186686B1 (ko) | 2009-12-29 | 2012-09-27 | 주식회사 에너세라믹 | 리튬 이차 전지용 양극 활물질의 제조 방법 |
| JP2012229147A (ja) * | 2011-04-27 | 2012-11-22 | Nichia Corp | オリビン型リチウム遷移金属酸化物及びその製造方法 |
| CN102844916A (zh) * | 2010-04-21 | 2012-12-26 | 株式会社Lg化学 | 橄榄石晶体结构的锂铁磷酸盐和使用所述锂铁磷酸盐的锂二次电池 |
| CN102859762A (zh) * | 2010-04-21 | 2013-01-02 | 株式会社Lg化学 | 含有基于硫键的硫化合物的锂铁磷酸盐和使用所述锂铁磷酸盐的锂二次电池 |
| CN102893432A (zh) * | 2010-04-21 | 2013-01-23 | 株式会社Lg化学 | 碳包覆的具有橄榄石晶体结构的锂铁磷酸盐和使用所述锂铁磷酸盐的锂二次电池 |
| JP2013032257A (ja) * | 2011-06-28 | 2013-02-14 | Nichia Corp | オリビン型リチウム遷移金属酸化物及びその製造方法 |
| EP2576439A1 (de) | 2010-05-27 | 2013-04-10 | Süd-Chemie AG | Kohlenstoff -lithiumübergangsmetallphosphat -verbundmaterial mit einem niedrigen kohlenstoffgehalt |
| CN103066293A (zh) * | 2011-10-20 | 2013-04-24 | 何金祥 | 锂离子电池正极材料的制备方法 |
| JP2013120621A (ja) * | 2011-12-06 | 2013-06-17 | Toyota Motor Corp | 非水系二次電池 |
| US9812707B2 (en) * | 2010-04-21 | 2017-11-07 | Lg Chem, Ltd. | Carbon-coated lithium iron phosphate of olivine crystal structure and lithium secondary battery using the same |
| CN107565132A (zh) * | 2017-08-24 | 2018-01-09 | 高延敏 | 磷酸铁的制备方法及其制备的磷酸铁、磷酸铁锂的制备方法及其制备的磷酸铁锂以及锂电池 |
Families Citing this family (41)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW201010944A (en) | 2008-04-17 | 2010-03-16 | Basf Se | Process for the preparation of crystalline lithium-, iron-and phosphate-comprising materials |
| TW200951066A (en) * | 2008-04-17 | 2009-12-16 | Basf Se | Process for the preparation of crystalline lithium-, iron-and phosphate-comprising materials |
| BRPI0919654B1 (pt) * | 2008-10-22 | 2019-07-30 | Lg Chem, Ltd. | Fosfato de ferro-lítio do tipo olivina, mistura de catodo e bateria secundária de lítio |
| ES2384675T3 (es) * | 2009-11-10 | 2012-07-10 | Rockwood Italia Spa | Procedimiento hidrotérmico para la producción de polvo de LiFePO4 |
| JP5502518B2 (ja) * | 2010-02-15 | 2014-05-28 | 旭化成イーマテリアルズ株式会社 | リチウムイオン二次電池 |
| JP5784292B2 (ja) * | 2010-09-13 | 2015-09-24 | 三井金属鉱業株式会社 | マグネタイト粒子 |
| US8980126B2 (en) | 2010-10-08 | 2015-03-17 | Semiconductor Energy Laboratory Co., Ltd. | Electrode material and method for manufacturing power storage device |
| JP5710788B2 (ja) * | 2011-03-16 | 2015-04-30 | 韓華石油化学株式会社Hanwhachemical Corporation | ロータリーキルンを用いた電極物質のか焼方法 |
| US8945498B2 (en) | 2011-03-18 | 2015-02-03 | Semiconductor Energy Laboratory Co., Ltd. | Method for manufacturing lithium-containing composite oxide |
| JP2012204322A (ja) * | 2011-03-28 | 2012-10-22 | Gs Yuasa Corp | 非水電解質二次電池用活物質の製造方法 |
| JP5772197B2 (ja) | 2011-05-09 | 2015-09-02 | ソニー株式会社 | リチウムイオン二次電池用活物質、リチウムイオン二次電池用電極、リチウムイオン二次電池、電子機器、電動工具、電動車両および電力貯蔵システム |
| US11476462B2 (en) | 2011-06-22 | 2022-10-18 | National Tsing Hua University | LiFePO4 precursor for manufacturing electrode material of Li-ion battery and method for manufacturing the same |
| US9112222B2 (en) | 2012-07-13 | 2015-08-18 | Sony Corporation | Lithium ion secondary battery active material, lithium ion secondary battery electrode, lithium ion secondary battery, electronic device, electronic power tool, electric vehicle, and power storage system |
| CN102867953B (zh) * | 2012-07-24 | 2015-01-21 | 龙能科技(苏州)有限公司 | 用氢氧化物或羟基氧化物生产锂离子电池正极材料的方法 |
| JP6207923B2 (ja) | 2012-08-27 | 2017-10-04 | 株式会社半導体エネルギー研究所 | 二次電池用正極の製造方法 |
| KR101733743B1 (ko) | 2012-10-05 | 2017-05-08 | 삼성에스디아이 주식회사 | 리튬 이차 전지용 양극 활물질의 제조 방법, 상기 제조 방법에 따라 제조된 리튬 이차 전지용 양극 활물질, 및 이를 포함하는 리튬 이차 전지 |
| JP6032410B2 (ja) * | 2012-10-30 | 2016-11-30 | 燐化学工業株式会社 | リン酸第二鉄含水和物粒子粉末の製造方法 |
| WO2014069207A1 (ja) * | 2012-10-31 | 2014-05-08 | シャープ株式会社 | 正極活物質および正極並びに非水電解質二次電池 |
| JP5928302B2 (ja) * | 2012-11-02 | 2016-06-01 | 日立金属株式会社 | リチウム二次電池用正極活物質の製造方法 |
| KR101561375B1 (ko) | 2013-01-10 | 2015-10-19 | 주식회사 엘지화학 | 리튬 인산철 나노분말 제조방법 |
| KR101586556B1 (ko) | 2013-01-10 | 2016-01-20 | 주식회사 엘지화학 | 탄소 코팅 리튬 인산철 나노분말 제조방법 |
| KR101542317B1 (ko) | 2013-01-10 | 2015-08-05 | 주식회사 엘지화학 | 리튬 인산철 나노분말 제조방법 |
| JP6028630B2 (ja) | 2013-03-12 | 2016-11-16 | ソニー株式会社 | 二次電池用活物質、二次電池用電極、二次電池、電池パック、電動車両、電力貯蔵システム、電動工具および電子機器 |
| CN105144440A (zh) | 2013-04-24 | 2015-12-09 | 住友大阪水泥股份有限公司 | 电极材料和电极以及锂离子电池 |
| JP2014216240A (ja) * | 2013-04-26 | 2014-11-17 | 住友大阪セメント株式会社 | 電極活物質と電極材料及び電極、リチウムイオン電池並びに電極材料の製造方法 |
| US9431651B2 (en) | 2013-08-30 | 2016-08-30 | Hong Kong Applied Science and Technology Research Institute Company Limited | Composite material for a lithium ion battery anode and a method of producing the same |
| US10446833B2 (en) * | 2013-09-20 | 2019-10-15 | Basf Se | Electrode material including lithium transition metal oxide, lithium iron phosphate, further iron-phosphorous compound. and carbon, and lithium battery including the same |
| JP5769140B2 (ja) * | 2014-01-30 | 2015-08-26 | 住友金属鉱山株式会社 | リチウム二次電池用正極活物質の製造方法 |
| JP5835446B1 (ja) * | 2014-10-28 | 2015-12-24 | 住友大阪セメント株式会社 | 正極材料、正極材料の製造方法、正極およびリチウムイオン電池 |
| DE102015214577A1 (de) * | 2015-07-31 | 2017-02-02 | Robert Bosch Gmbh | Verfahren zur Herstellung einer Elektrode eines Lithiumionenakkumulators |
| EP3184170B1 (en) * | 2015-12-25 | 2020-04-01 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Iron oxide-hydroxide particles, method for producing them and oxidation catalyst using the iron oxide-hydroxide particles |
| CN105680020A (zh) * | 2016-03-17 | 2016-06-15 | 贵州安达科技能源股份有限公司 | 一种制备磷酸铁锂和正极材料的方法 |
| JP6497462B1 (ja) | 2018-03-30 | 2019-04-10 | 住友大阪セメント株式会社 | リチウムイオン電池用電極材料及びリチウムイオン電池 |
| CN111081319B (zh) * | 2019-11-01 | 2023-07-25 | 力神(青岛)新能源有限公司 | 一种正极材料碳含量的建模方法 |
| CN111072003A (zh) * | 2019-12-20 | 2020-04-28 | 合肥师范学院 | 一种刺球状磷酸盐电极材料及其制备方法和锂离子电池 |
| CN113023703A (zh) * | 2021-03-24 | 2021-06-25 | 郑州中科新兴产业技术研究院 | 一种回收废旧磷酸铁锂粉的方法 |
| CN113120876B (zh) * | 2021-03-30 | 2022-06-10 | 中国石油大学(北京) | 一种磷酸铁锂废极片再生回收制备磷酸铁锂材料的方法 |
| CN114261952B (zh) * | 2021-12-21 | 2024-03-29 | 蜂巢能源科技股份有限公司 | 一种磷酸铁锂正极材料、其制备方法及用途 |
| CN114497479B (zh) * | 2021-12-30 | 2023-10-31 | 乳源东阳光新能源材料有限公司 | 一种高压实高性能磷酸铁锂正极材料及其制备方法 |
| WO2023192443A1 (en) | 2022-04-01 | 2023-10-05 | Aspen Aerogels, Inc. | Carbon powder containing lithium iron phosphate cathode materials |
| CN114988386B (zh) * | 2022-06-16 | 2024-02-02 | 蜂巢能源科技股份有限公司 | 一种磷酸锰铁锂正极材料及其制备方法和应用 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003520405A (ja) | 2000-01-18 | 2003-07-02 | ヴァレンス テクノロジー インコーポレーテッド | リチウム金属含有物質の製造方法、生成物、組成物およびバッテリー |
| JP2004079276A (ja) * | 2002-08-13 | 2004-03-11 | Sony Corp | 正極活物質及びその製造方法 |
| JP2005514304A (ja) | 2001-12-21 | 2005-05-19 | マサチューセッツ・インスティチュート・オブ・テクノロジー | 伝導性リチウム貯蔵電極 |
| JP2006032241A (ja) | 2004-07-21 | 2006-02-02 | Mitsui Mining Co Ltd | リチウムイオン二次電池用正極材料、その製造方法、及びリチウムイオン二次電池 |
| JP2006347805A (ja) | 2005-06-15 | 2006-12-28 | Seimi Chem Co Ltd | リチウム鉄複合酸化物の製造方法 |
| JP2007022894A (ja) * | 2005-07-21 | 2007-02-01 | Seimi Chem Co Ltd | リチウム鉄複合酸化物の製造方法 |
| JP2007230784A (ja) * | 2004-03-30 | 2007-09-13 | Agc Seimi Chemical Co Ltd | リチウム鉄複合酸化物の製造方法 |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6350539B1 (en) * | 1999-10-25 | 2002-02-26 | General Motors Corporation | Composite gas distribution structure for fuel cell |
| CA2320661A1 (fr) * | 2000-09-26 | 2002-03-26 | Hydro-Quebec | Nouveau procede de synthese de materiaux limpo4 a structure olivine |
| JP4963777B2 (ja) * | 2001-08-24 | 2012-06-27 | ソニー株式会社 | 電池 |
| US6960402B2 (en) * | 2002-06-28 | 2005-11-01 | Advanced Energy Technology Inc. | Perforated cylindrical fuel cells |
| US7390472B1 (en) * | 2002-10-29 | 2008-06-24 | Nei Corp. | Method of making nanostructured lithium iron phosphate—based powders with an olivine type structure |
| CN1305147C (zh) * | 2004-12-30 | 2007-03-14 | 清华大学 | 锂离子电池正极材料高密度球形磷酸铁锂的制备方法 |
| KR101264459B1 (ko) * | 2005-06-29 | 2013-05-14 | 썽뜨르 나쇼날르 드 라 르쉐르쉐 씨엉띠삐끄 | 결정성 나노메트릭 LiFePO₄ |
| US8323832B2 (en) * | 2005-08-08 | 2012-12-04 | A123 Systems, Inc. | Nanoscale ion storage materials |
| CN100385713C (zh) * | 2005-11-30 | 2008-04-30 | 重庆大学 | 一种制备磷酸亚铁锂的方法 |
| TWI346406B (en) * | 2006-02-16 | 2011-08-01 | Lg Chemical Ltd | Lithium secondary battery with enhanced heat-resistance |
| DE112007001410T5 (de) * | 2006-06-16 | 2009-04-23 | Sharp Kabushiki Kaisha | Positivelektrode, Herstellverfahren für diese sowie Lithiumsekundärbatterie unter Verwendung derselben |
| TWI319920B (en) * | 2006-07-06 | 2010-01-21 | The preparation and application of the lifepo4/li3v2(po4)3 composite cathode materials for lithium ion batteries | |
| JP5479096B2 (ja) * | 2006-08-21 | 2014-04-23 | エルジー・ケム・リミテッド | リチウム金属リン酸化物の製造方法 |
| KR100821832B1 (ko) * | 2007-04-20 | 2008-04-14 | 정성윤 | 리튬전이금속 인산화물의 나노입자 분말의 제조방법 |
| US20090155689A1 (en) * | 2007-12-14 | 2009-06-18 | Karim Zaghib | Lithium iron phosphate cathode materials with enhanced energy density and power performance |
| TWI466370B (zh) * | 2008-01-17 | 2014-12-21 | A123 Systems Inc | 鋰離子電池的混合式金屬橄欖石電極材料 |
| US8088305B2 (en) * | 2008-02-22 | 2012-01-03 | Byd Company Limited | Lithium iron phosphate cathode material |
| KR100939647B1 (ko) * | 2009-01-22 | 2010-02-03 | 한화석유화학 주식회사 | 전극 활물질인 음이온 부족형 비화학양론 리튬 전이금속 다중산 화합물, 그 제조 방법 및 그를 이용한 전기화학 소자 |
-
2009
- 2009-03-26 WO PCT/JP2009/001374 patent/WO2009122686A1/ja active Application Filing
- 2009-03-26 EP EP09727078.9A patent/EP2277828B1/en active Active
- 2009-03-26 CN CN2009801111827A patent/CN101980956A/zh active Pending
- 2009-03-26 KR KR1020107021513A patent/KR101587671B1/ko active IP Right Grant
- 2009-03-26 CN CN201510896278.6A patent/CN105355920A/zh active Pending
- 2009-03-26 US US12/935,456 patent/US20110091772A1/en not_active Abandoned
- 2009-03-31 JP JP2009087967A patent/JP5464322B2/ja active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003520405A (ja) | 2000-01-18 | 2003-07-02 | ヴァレンス テクノロジー インコーポレーテッド | リチウム金属含有物質の製造方法、生成物、組成物およびバッテリー |
| JP2005514304A (ja) | 2001-12-21 | 2005-05-19 | マサチューセッツ・インスティチュート・オブ・テクノロジー | 伝導性リチウム貯蔵電極 |
| JP2004079276A (ja) * | 2002-08-13 | 2004-03-11 | Sony Corp | 正極活物質及びその製造方法 |
| JP2007230784A (ja) * | 2004-03-30 | 2007-09-13 | Agc Seimi Chemical Co Ltd | リチウム鉄複合酸化物の製造方法 |
| JP2006032241A (ja) | 2004-07-21 | 2006-02-02 | Mitsui Mining Co Ltd | リチウムイオン二次電池用正極材料、その製造方法、及びリチウムイオン二次電池 |
| JP2006347805A (ja) | 2005-06-15 | 2006-12-28 | Seimi Chem Co Ltd | リチウム鉄複合酸化物の製造方法 |
| JP2007022894A (ja) * | 2005-07-21 | 2007-02-01 | Seimi Chem Co Ltd | リチウム鉄複合酸化物の製造方法 |
Non-Patent Citations (5)
| Title |
|---|
| A. YAMADA ET AL., J. ELECTROCHEM. SOC., vol. 148, 2001, pages A224 - 229 |
| D. MORGAN ET AL., ELECTROCHEM. AND SOLID-STATE LETT., vol. 7, 2004, pages A30 - 32 |
| H. HUANG ET AL., ELECTROCHEM. AND SOLID-STATE LETT., vol. 4, 2001, pages A170 - 172 |
| M. SAIFUL ISLAM ET AL., CHEM. MATER., vol. 17, 2005, pages 5085 - 5092 |
| ZHAOHUI CHEN ET AL., J. ELECTROCHEM. SOC., vol. 149, 2002, pages A1184 - 1189 |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8962185B2 (en) * | 2008-10-22 | 2015-02-24 | Lg Chem, Ltd. | Cathode mix having improved efficiency and energy density of electrode |
| US20100239909A1 (en) * | 2008-10-22 | 2010-09-23 | Lg Chem, Ltd. | Cathode mix containing having improved efficiency and energy density of electrode |
| US20110287315A1 (en) * | 2008-10-22 | 2011-11-24 | Lg Chem, Ltd. | Cathode active material providing improved efficiency and energy density of electrode |
| US8974957B2 (en) * | 2008-10-22 | 2015-03-10 | Lg Chem, Ltd. | Cathode active material providing improved efficiency and energy density of electrode |
| WO2011062232A1 (ja) * | 2009-11-18 | 2011-05-26 | 三井化学株式会社 | 電気化学セル用水性ペースト、該水性ペーストを塗布してなる電気化学セル用極板、および該極板を含む電池 |
| KR101186686B1 (ko) | 2009-12-29 | 2012-09-27 | 주식회사 에너세라믹 | 리튬 이차 전지용 양극 활물질의 제조 방법 |
| WO2011115211A1 (ja) * | 2010-03-19 | 2011-09-22 | 戸田工業株式会社 | リン酸マンガン鉄リチウム粒子粉末の製造方法、リン酸マンガン鉄リチウム粒子粉末、及び該粒子粉末を用いた非水電解質二次電池 |
| WO2011114918A1 (en) * | 2010-03-19 | 2011-09-22 | Semiconductor Energy Laboratory Co., Ltd. | Power storage device and manufacturing method thereof |
| CN102781827A (zh) * | 2010-03-19 | 2012-11-14 | 户田工业株式会社 | 磷酸锰铁锂颗粒粉末的制造方法、磷酸锰铁锂颗粒粉末和使用该颗粒粉末的非水电解质二次电池 |
| JP2011210376A (ja) * | 2010-03-28 | 2011-10-20 | Niigata Univ | Liイオン電池用正極活物質およびその製造方法 |
| US9214700B2 (en) * | 2010-04-21 | 2015-12-15 | Lg Chem, Ltd. | Lithium iron phosphate containing sulfur compound based upon sulfide bond and lithium secondary battery using the same |
| CN102844916A (zh) * | 2010-04-21 | 2012-12-26 | 株式会社Lg化学 | 橄榄石晶体结构的锂铁磷酸盐和使用所述锂铁磷酸盐的锂二次电池 |
| CN102893432A (zh) * | 2010-04-21 | 2013-01-23 | 株式会社Lg化学 | 碳包覆的具有橄榄石晶体结构的锂铁磷酸盐和使用所述锂铁磷酸盐的锂二次电池 |
| US20130029226A1 (en) * | 2010-04-21 | 2013-01-31 | Lg Chem, Ltd. | Lithium iron phosphate of olivine crystal structure and lithium secondary battery using the same |
| US20130034776A1 (en) * | 2010-04-21 | 2013-02-07 | Lg Chem, Ltd. | Lithium iron phosphate containing sulfur compound based upon sulfide bond and lithium secondary battery using the same |
| US9812707B2 (en) * | 2010-04-21 | 2017-11-07 | Lg Chem, Ltd. | Carbon-coated lithium iron phosphate of olivine crystal structure and lithium secondary battery using the same |
| US9331329B2 (en) * | 2010-04-21 | 2016-05-03 | Lg Chem, Ltd. | Carbon-coated lithium iron phosphate of olivine crystal structure and lithium secondary battery using the same |
| CN102859762A (zh) * | 2010-04-21 | 2013-01-02 | 株式会社Lg化学 | 含有基于硫键的硫化合物的锂铁磷酸盐和使用所述锂铁磷酸盐的锂二次电池 |
| CN102859762B (zh) * | 2010-04-21 | 2016-02-17 | 株式会社Lg化学 | 含有基于硫键的硫化合物的锂铁磷酸盐和使用所述锂铁磷酸盐的锂二次电池 |
| US20130216902A1 (en) * | 2010-04-21 | 2013-08-22 | Lg Chem, Ltd. | Carbon-coated lithium iron phosphate of olivine crystal structure and lithium secondary battery using the same |
| KR101400099B1 (ko) | 2010-04-21 | 2014-05-29 | 주식회사 엘지화학 | 올리빈 결정구조의 리튬 철인산화물 및 이를 이용한 리튬 이차전지 |
| US8906552B2 (en) | 2010-04-21 | 2014-12-09 | Lg Chem, Ltd. | Lithium iron phosphate of olivine crystal structure and lithium secondary battery using the same |
| US9318741B2 (en) * | 2010-04-28 | 2016-04-19 | Semiconductor Energy Laboratory Co., Ltd. | Positive electrode active material of power storage device, power storage device, electrically propelled vehicle, and method for manufacturing power storage device |
| US20110269022A1 (en) * | 2010-04-28 | 2011-11-03 | Semiconductor Energy Laboratory Co., Ltd. | Positive electrode active material of power storage device, power storage device, electrically propelled vehicle, and method for manufacturing power storage device |
| US10916774B2 (en) | 2010-04-28 | 2021-02-09 | Semiconductor Energy Laboratory Co., Ltd. | Positive electrode active material of power storage device, power storage device, electrically propelled vehicle, and method for manufacturing power storage |
| US9899678B2 (en) | 2010-04-28 | 2018-02-20 | Semiconductor Energy Laboratory Co., Ltd. | Positive electrode active material of power storage device, power storage device, electrically propelled vehicle, and method for manufacturing power storage device |
| US10224548B2 (en) | 2010-04-28 | 2019-03-05 | Semiconductor Energy Laboratory Co., Ltd. | Positive electrode active material of power storage device, power storage device, electrically propelled vehicle, and method for manufacturing power storage |
| EP2576439A1 (de) | 2010-05-27 | 2013-04-10 | Süd-Chemie AG | Kohlenstoff -lithiumübergangsmetallphosphat -verbundmaterial mit einem niedrigen kohlenstoffgehalt |
| EP2576439B1 (de) * | 2010-05-27 | 2016-08-24 | Johnson Matthey PLC | Kohlenstoff-lithiumübergangsmetallphosphat-verbundmaterial mit einem niedrigen kohlenstoffgehalt |
| JP2012229147A (ja) * | 2011-04-27 | 2012-11-22 | Nichia Corp | オリビン型リチウム遷移金属酸化物及びその製造方法 |
| JP2013032257A (ja) * | 2011-06-28 | 2013-02-14 | Nichia Corp | オリビン型リチウム遷移金属酸化物及びその製造方法 |
| CN103066293A (zh) * | 2011-10-20 | 2013-04-24 | 何金祥 | 锂离子电池正极材料的制备方法 |
| CN103066293B (zh) * | 2011-10-20 | 2016-11-09 | 何金祥 | 锂离子电池正极材料的制备方法 |
| JP2013120621A (ja) * | 2011-12-06 | 2013-06-17 | Toyota Motor Corp | 非水系二次電池 |
| CN107565132A (zh) * | 2017-08-24 | 2018-01-09 | 高延敏 | 磷酸铁的制备方法及其制备的磷酸铁、磷酸铁锂的制备方法及其制备的磷酸铁锂以及锂电池 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2277828B1 (en) | 2018-08-08 |
| JP2009263222A (ja) | 2009-11-12 |
| KR20110007112A (ko) | 2011-01-21 |
| US20110091772A1 (en) | 2011-04-21 |
| EP2277828A1 (en) | 2011-01-26 |
| EP2277828A4 (en) | 2014-12-31 |
| CN105355920A (zh) | 2016-02-24 |
| KR101587671B1 (ko) | 2016-01-21 |
| JP5464322B2 (ja) | 2014-04-09 |
| CN101980956A (zh) | 2011-02-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5464322B2 (ja) | リン酸鉄リチウム粒子粉末の製造方法、オリビン型構造のリン酸鉄リチウム粒子粉末、該リン酸鉄リチウム粒子粉末を用いた正極材シート及び非水溶媒系二次電池 | |
| US9847526B2 (en) | Lithium titanate particles and process for producing the lithium titanate particles, Mg-containing lithium titanate particles and process for producing the Mg-containing lithium titanate particles, negative electrode active substance particles for non-aqueous electrolyte secondary batteries, and non-aqueous electrolyte secondary battery | |
| JP5817963B2 (ja) | リン酸マンガン鉄リチウム粒子粉末の製造方法、リン酸マンガン鉄リチウム粒子粉末、及び該粒子粉末を用いた非水電解質二次電池 | |
| JP6107832B2 (ja) | Li−Ni複合酸化物粒子粉末及びその製造方法、並びに非水電解質二次電池 | |
| JP5376894B2 (ja) | オリビン構造を有する多元系リン酸型リチウム化合物粒子、その製造方法及びこれを正極材料に用いたリチウム二次電池 | |
| JP4211865B2 (ja) | 非水電解質二次電池用Li−Ni複合酸化物粒子粉末及びその製造方法、並びに非水電解質二次電池 | |
| JP5293936B2 (ja) | 非水電解質二次電池用オリビン型複合酸化物及びその製造方法、並びに二次電池 | |
| JP4829557B2 (ja) | リチウム鉄複合酸化物の製造方法 | |
| JP6112118B2 (ja) | Li−Ni複合酸化物粒子粉末並びに非水電解質二次電池 | |
| JP5517032B2 (ja) | 非水電解質二次電池用オリビン型複合酸化物粒子粉末及びその製造方法、並びに二次電池 | |
| JP6260535B2 (ja) | 炭素複合化リン酸マンガン鉄リチウム粒子粉末の製造方法、及び該粒子粉末を用いた非水電解質二次電池の製造方法 | |
| JP5364523B2 (ja) | オリビン型ケイ酸mリチウムの合成方法およびリチウムイオン二次電池 | |
| JP2011132095A (ja) | オリビン型化合物粒子粉末の製造方法、並びに非水電解質二次電池 | |
| JP2023027147A (ja) | 金属複合水酸化物とその製造方法、非水電解質二次電池用正極活物質とその製造方法、及び、それを用いた非水電解質二次電池 | |
| JP2019019047A (ja) | 金属複合水酸化物とその製造方法、非水電解質二次電池用正極活物質とその製造方法、及び、それを用いた非水電解質二次電池 | |
| JP6362033B2 (ja) | 非水系電解液二次電池用正極活物質と非水系電解液二次電池 | |
| JPWO2019065195A1 (ja) | 非水電解質二次電池 | |
| Yamashita et al. | Hydrothermal synthesis and electrochemical properties of Li2FexMnxCo1− 2xSiO4/C cathode materials for lithium-ion batteries | |
| JP2018147695A (ja) | 非水系電解質二次電池用正極活物質 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980111182.7 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09727078 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20107021513 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009727078 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12935456 Country of ref document: US |