WO2003027040A1 - Materiau de revetement thermo-isolant destine a un refractaire contenant du carbone - Google Patents
Materiau de revetement thermo-isolant destine a un refractaire contenant du carbone Download PDFInfo
- Publication number
- WO2003027040A1 WO2003027040A1 PCT/JP2002/009765 JP0209765W WO03027040A1 WO 2003027040 A1 WO2003027040 A1 WO 2003027040A1 JP 0209765 W JP0209765 W JP 0209765W WO 03027040 A1 WO03027040 A1 WO 03027040A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- weight
- refractory
- coating material
- carbon
- hollow
- Prior art date
Links
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 title claims abstract description 48
- 239000011248 coating agent Substances 0.000 title claims abstract description 47
- 238000000576 coating method Methods 0.000 title claims abstract description 47
- 239000000463 material Substances 0.000 title claims abstract description 47
- 229910052799 carbon Inorganic materials 0.000 title claims abstract description 45
- 239000002245 particle Substances 0.000 claims abstract description 64
- 239000011819 refractory material Substances 0.000 claims abstract description 21
- 239000004927 clay Substances 0.000 claims abstract description 17
- 239000011230 binding agent Substances 0.000 claims abstract description 16
- 239000007788 liquid Substances 0.000 claims abstract description 16
- 239000011521 glass Substances 0.000 claims description 18
- 239000000843 powder Substances 0.000 claims description 18
- 238000010438 heat treatment Methods 0.000 claims description 16
- 239000010455 vermiculite Substances 0.000 claims description 14
- 229910052902 vermiculite Inorganic materials 0.000 claims description 14
- 235000019354 vermiculite Nutrition 0.000 claims description 14
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 12
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims description 12
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 claims description 10
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 claims description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 7
- 150000001875 compounds Chemical class 0.000 claims description 6
- 238000002425 crystallisation Methods 0.000 claims description 6
- 230000008025 crystallization Effects 0.000 claims description 6
- 238000009413 insulation Methods 0.000 claims description 6
- 239000000155 melt Substances 0.000 claims description 6
- 238000010301 surface-oxidation reaction Methods 0.000 claims description 5
- KZHJGOXRZJKJNY-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Si]=O.O=[Al]O[Al]=O.O=[Al]O[Al]=O.O=[Al]O[Al]=O KZHJGOXRZJKJNY-UHFFFAOYSA-N 0.000 claims description 4
- 239000005350 fused silica glass Substances 0.000 claims description 4
- 239000000395 magnesium oxide Substances 0.000 claims description 4
- 229910052863 mullite Inorganic materials 0.000 claims description 4
- 229910052611 pyroxene Inorganic materials 0.000 claims description 4
- -1 chamotte Inorganic materials 0.000 claims description 2
- 239000013078 crystal Substances 0.000 claims 1
- 239000002023 wood Substances 0.000 claims 1
- 230000003647 oxidation Effects 0.000 abstract description 2
- 238000007254 oxidation reaction Methods 0.000 abstract description 2
- 239000010410 layer Substances 0.000 description 22
- 239000002994 raw material Substances 0.000 description 8
- 238000007654 immersion Methods 0.000 description 6
- 230000003078 antioxidant effect Effects 0.000 description 5
- 238000010924 continuous production Methods 0.000 description 5
- 239000010439 graphite Substances 0.000 description 5
- 229910002804 graphite Inorganic materials 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 239000003963 antioxidant agent Substances 0.000 description 3
- 239000011247 coating layer Substances 0.000 description 3
- 238000010276 construction Methods 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 238000002844 melting Methods 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 238000005245 sintering Methods 0.000 description 3
- 229910052596 spinel Inorganic materials 0.000 description 3
- 239000011029 spinel Substances 0.000 description 3
- 238000005507 spraying Methods 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 2
- 238000009529 body temperature measurement Methods 0.000 description 2
- 239000004568 cement Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000013329 compounding Methods 0.000 description 2
- 238000005336 cracking Methods 0.000 description 2
- 230000005183 environmental health Effects 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 230000009970 fire resistant effect Effects 0.000 description 2
- 239000003365 glass fiber Substances 0.000 description 2
- 239000002241 glass-ceramic Substances 0.000 description 2
- 230000017525 heat dissipation Effects 0.000 description 2
- 239000011810 insulating material Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000005332 obsidian Substances 0.000 description 2
- 229910001562 pearlite Inorganic materials 0.000 description 2
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- 235000019738 Limestone Nutrition 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 239000004111 Potassium silicate Substances 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- ILRRQNADMUWWFW-UHFFFAOYSA-K aluminium phosphate Chemical compound O1[Al]2OP1(=O)O2 ILRRQNADMUWWFW-UHFFFAOYSA-K 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000001680 brushing effect Effects 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000008119 colloidal silica Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000002657 fibrous material Substances 0.000 description 1
- 238000005188 flotation Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 238000005187 foaming Methods 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 239000012774 insulation material Substances 0.000 description 1
- 239000006028 limestone Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 235000014380 magnesium carbonate Nutrition 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000010451 perlite Substances 0.000 description 1
- 235000019362 perlite Nutrition 0.000 description 1
- NNHHDJVEYQHLHG-UHFFFAOYSA-N potassium silicate Chemical compound [K+].[K+].[O-][Si]([O-])=O NNHHDJVEYQHLHG-UHFFFAOYSA-N 0.000 description 1
- 229910052913 potassium silicate Inorganic materials 0.000 description 1
- 235000019353 potassium silicate Nutrition 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 229910021487 silica fume Inorganic materials 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 239000012720 thermal barrier coating Substances 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B32/00—Artificial stone not provided for in other groups of this subclass
- C04B32/005—Artificial stone obtained by melting at least part of the composition, e.g. metal
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/01—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics
- C04B35/013—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics containing carbon
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/01—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics
- C04B35/16—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics based on silicates other than clay
- C04B35/18—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics based on silicates other than clay rich in aluminium oxide
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/62605—Treating the starting powders individually or as mixtures
- C04B35/62645—Thermal treatment of powders or mixtures thereof other than sintering
- C04B35/62665—Flame, plasma or melting treatment
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/63—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B using additives specially adapted for forming the products, e.g.. binder binders
- C04B35/6303—Inorganic additives
- C04B35/6316—Binders based on silicon compounds
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/66—Monolithic refractories or refractory mortars, including those whether or not containing clay
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/009—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone characterised by the material treated
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/45—Coating or impregnating, e.g. injection in masonry, partial coating of green or fired ceramics, organic coating compositions for adhering together two concrete elements
- C04B41/50—Coating or impregnating, e.g. injection in masonry, partial coating of green or fired ceramics, organic coating compositions for adhering together two concrete elements with inorganic materials
- C04B41/5025—Coating or impregnating, e.g. injection in masonry, partial coating of green or fired ceramics, organic coating compositions for adhering together two concrete elements with inorganic materials with ceramic materials
- C04B41/5037—Clay, Kaolin
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/80—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone of only ceramics
- C04B41/81—Coating or impregnation
- C04B41/85—Coating or impregnation with inorganic materials
- C04B41/87—Ceramics
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/18—Fireproof paints including high temperature resistant paints
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/60—Additives non-macromolecular
- C09D7/61—Additives non-macromolecular inorganic
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/66—Additives characterised by particle size
- C09D7/69—Particle size larger than 1000 nm
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/70—Additives characterised by shape, e.g. fibres, flakes or microspheres
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2111/00—Mortars, concrete or artificial stone or mixtures to prepare them, characterised by specific function, property or use
- C04B2111/00474—Uses not provided for elsewhere in C04B2111/00
- C04B2111/0087—Uses not provided for elsewhere in C04B2111/00 for metallurgical applications
- C04B2111/00879—Non-ferrous metallurgy
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3205—Alkaline earth oxides or oxide forming salts thereof, e.g. beryllium oxide
- C04B2235/3206—Magnesium oxides or oxide-forming salts thereof
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3217—Aluminum oxide or oxide forming salts thereof, e.g. bauxite, alpha-alumina
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3217—Aluminum oxide or oxide forming salts thereof, e.g. bauxite, alpha-alumina
- C04B2235/3222—Aluminates other than alumino-silicates, e.g. spinel (MgAl2O4)
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3231—Refractory metal oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3244—Zirconium oxides, zirconates, hafnium oxides, hafnates, or oxide-forming salts thereof
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/34—Non-metal oxides, non-metal mixed oxides, or salts thereof that form the non-metal oxides upon heating, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3418—Silicon oxide, silicic acids or oxide forming salts thereof, e.g. silica sol, fused silica, silica fume, cristobalite, quartz or flint
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/34—Non-metal oxides, non-metal mixed oxides, or salts thereof that form the non-metal oxides upon heating, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3427—Silicates other than clay, e.g. water glass
- C04B2235/3463—Alumino-silicates other than clay, e.g. mullite
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/34—Non-metal oxides, non-metal mixed oxides, or salts thereof that form the non-metal oxides upon heating, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3427—Silicates other than clay, e.g. water glass
- C04B2235/3463—Alumino-silicates other than clay, e.g. mullite
- C04B2235/3481—Alkaline earth metal alumino-silicates other than clay, e.g. cordierite, beryl, micas such as margarite, plagioclase feldspars such as anorthite, zeolites such as chabazite
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/34—Non-metal oxides, non-metal mixed oxides, or salts thereof that form the non-metal oxides upon heating, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/349—Clays, e.g. bentonites, smectites such as montmorillonite, vermiculites or kaolines, e.g. illite, talc or sepiolite
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/36—Glass starting materials for making ceramics, e.g. silica glass
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/42—Non metallic elements added as constituents or additives, e.g. sulfur, phosphor, selenium or tellurium
- C04B2235/422—Carbon
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/50—Constituents or additives of the starting mixture chosen for their shape or used because of their shape or their physical appearance
- C04B2235/54—Particle size related information
- C04B2235/5418—Particle size related information expressed by the size of the particles or aggregates thereof
- C04B2235/5427—Particle size related information expressed by the size of the particles or aggregates thereof millimeter or submillimeter sized, i.e. larger than 0,1 mm
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/72—Products characterised by the absence or the low content of specific components, e.g. alkali metal free alumina ceramics
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/346—Clay
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/25—Web or sheet containing structurally defined element or component and including a second component containing structurally defined particles
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/25—Web or sheet containing structurally defined element or component and including a second component containing structurally defined particles
- Y10T428/252—Glass or ceramic [i.e., fired or glazed clay, cement, etc.] [porcelain, quartz, etc.]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/26—Web or sheet containing structurally defined element or component, the element or component having a specified physical dimension
- Y10T428/263—Coating layer not in excess of 5 mils thick or equivalent
- Y10T428/264—Up to 3 mils
- Y10T428/265—1 mil or less
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/30—Self-sustaining carbon mass or layer with impregnant or other layer
Definitions
- the present invention relates to a heat insulating coating material which prevents surface oxidation of a carbon-containing refractory, suppresses heat dissipation from the surface as a heat insulating layer, and does not cause environmental health problems.
- Carbon-containing refractories such as alumina-graphite and zirconia-graphite, are used as refractories for construction used in continuous production of steel.
- the carbon-containing refractory is coated with an antioxidant mainly composed of glass powder on the surface to prevent carbon oxidation during preheating and during use.
- heat insulating material consisting of glass fiber or ceramic fiber is usually wound around the outer surface of carbon-containing refractories. Is widely practiced.
- heat insulation materials made of fibrous materials such as glass fibers or ceramic fibers have become an environmental health problem.
- Japanese Patent Application Laid-Open No. 7-147174 discloses that vermiculite heat-treated at 300 to 1200 ° C. is 3 to 30% by weight, Obsidian that forms a hollow structure with heat of 800 ° C or more ⁇ Pearlite ⁇ Pinestone ⁇
- One or more types of expansive shale are 1 to 30% by weight in an unheated state, and 400 to 150% Glass powder that softens and melts in the range of ° C, or pyroxene 'silica stone' chamotte 'mullitealumina' fused silica-zirconiamagnesia or refractory powder of at least 40 to 96% by weight
- a heat-insulating antioxidant for graphite-containing refractories having a heat-insulating property in which a liquid binder is blended in an amount of 20 to 250% by weight with the mixture is disclosed.
- Japanese Unexamined Patent Publication (Kokai) No. Hei 9-157,045 discloses that refractory raw materials having a particle size of 0.5 to 6.0 mm, having a hollow particle size of 15 to 70% by weight, and having a particle size of 1 mm or less. 0-3 5% by weight, d, Xy-alumina intermediate 10 to 40% by weight, microsilica 0.5 to 5.0% by weight, air entrainer 0.01 to 0.4% by weight, and thickening Disclosed is a refractory insulated castable refractory comprising 0.1 to 0.5% by weight of an agent.
- Japanese Patent Application Laid-Open No. 11-49757 discloses that alumina hollow particles having a particle size of 0.5 to 6.0 mm are 15 to 70% by weight, and magnesia having a particle size of 1 mm or less. 2 to 30% by weight of at least one of the first fine refractory raw materials mainly containing subinel, and a main component mainly containing at least one of alumina, mullite, silica, and zirconia having a particle size of 1 mm or less. 8 to 60% by weight of the second fine refractory raw material, and 5 to 45% by weight of at least one of alumina cement having hydraulic component and alumina component of 65% by weight or more, and 5 to 45% by weight of air entraining agent. Disclosed is an amorphous refractory characterized by the fact that it comprises a mixture of 0.4% by weight and a thickener (0.1-0.5% by weight).
- the heat-insulating antioxidant for graphite-containing refractories disclosed in Japanese Patent Application Laid-Open No. 7-24771-4 is an unheated obsidian, perlite, pinestone, expansive shale (hereinafter referred to as foaming). Content is defined as 1 to 30% by weight. However, even if 30% by weight of expandable pearlite is blended, a sufficient foamed heat insulating layer can be obtained during heating. I can't.
- the foamable foam is foamed when heated, but when foamed, the adhesion to the graphite-containing refractory as a base material is deteriorated, and a gap is formed between the base material and the foamed layer, and the foamed layer may also come off. is there.
- the fire-resistant heat-insulated cast refractory disclosed in Japanese Patent Application Laid-Open No. 9-157045 uses refractory hollow particles having a large particle size. When it is used for heat treatment, it becomes difficult to form a heat insulating layer. In addition, since it does not contain a glass phase forming component, there is a problem that the bonding strength of the heat insulating layer in the operating temperature range of the carbon-containing refractory is insufficient.
- the heat-insulating coating material for refractory containing carbon is used because refractory hollow particles having a large particle size are used. When it is used for heat treatment, it becomes difficult to form a heat insulating layer.
- it contains hydraulic alumina and alumina cement, when it is applied as a heat-insulating coating material for refractories containing carbon, the heat-insulating layer cracks during use, and the There is a problem that it is easy to release.
- an object of the present invention is to provide a heat-insulating coating material for carbon-containing refractories which can prevent surface oxidation of a carbon-containing refractory at the time of high-temperature heating and can form a heat-insulating layer.
- the heat insulating coating material for a carbon-containing refractory of the present invention employs a hollow refractory powder.
- the heat-insulating coating material for a carbon-containing refractory of the present invention contains hollow refractory particles, the content of the hollow particles is 597% by weight, the remaining part or all is clay, and A liquid binder is added to a refractory material having a clay content of 30% by weight or less, and 210.70% by weight of a liquid binder is added.
- the heat insulating coating material for refractory containing carbon of the present invention contains hollow refractory particles, the content of the hollow particles is 509% by weight, and the content of the clay is 225% by weight. % Or the remaining part is vermiculite in which water of crystallization is dehydrated and / or glass powder that softens and melts in a temperature range of 500 1100 ° C., and / or vermiculite in which the water of crystallization is dehydrated. Liquid binder is added to the refractory material whose total amount of glass powder softening and melting in the temperature range of 500 ° C. is not more than 30% by weight, and a liquid binder is added at a rate of 201% to 0% by weight. And
- the heat-insulating coating material for the refractory containing carbon of the present invention is one or two or more selected from fused quartz, magnesite, magnesia, zirconia, spinel, alumina, chamotte, limestone, and clay shale. It is characterized by containing at least 10% by weight of at least one compound.
- insulation co one coating material forces one carbon-containing refractory of the present invention, the hollow refractory particles child, S i 0 2 and A 1 2 0 3 containing, the content S i 0 2: 40 to 80% by weight,
- a 1 2 0 3 wherein the 2 0 6 0% by weight.
- the heat-insulating coating material for refractory containing carbon according to the present invention is characterized in that the particle size range of the hollow refractory particles is in the range of 380 microns.
- FIG. 1 is a schematic diagram of an apparatus for explaining measurement of a temperature drop in an embodiment.
- 1 denotes a heating box
- 2 denotes an immersion nozzle for continuous production
- 3 denotes a coating material coating film
- 4 denotes a gas burner
- 5 denotes a temperature measurement position.
- the present inventors have applied hollow refractory particles to solve the above-described problems and achieve a stable antioxidant effect and the formation of a heat insulating layer, thereby minimizing the volume change of the heat insulating layer during heating.
- the present invention was completed by studying in detail the amount of refractory raw materials to be added to maintain the bonding of the structure of the heat insulating material and to improve the adhesion between the heat insulating layer and the refractory base material. is there. '
- the heat-insulating coating material for refractory containing carbon according to the present invention is characterized by containing hollow refractory particles, and the content of the hollow refractory particles is 50 to 97 in the refractory material. % By weight. More preferably, it is 55 to 94% by weight.
- the content of the hollow refractory particles is less than 50% by weight, a sufficient heat insulating effect cannot be obtained, which is not preferable.
- the content of the hollow refractory particles exceeds 97% by weight, the structure of the heat insulating layer becomes insufficiently bonded, causing problems such as peeling and cracking, which is also unsuitable.
- the hollow refractory particles used in the present invention represent hollow particles having a hollow center, and the thickness of the shell and the shape of the hollow particles are not particularly specified. These hollow particles include not only spherical closed hollow particles but also hollow particles partially having a hole. Generally, those having an open area of 1 to 4 or less on the particle surface can be used as the hollow particles of the present invention.
- the hollow volume of these hollow particles is usually 50 to 90%, preferably 60 to 80%.
- These hollow particles are obtained by crushing, sintering, flotation, drying, and classification of mineral raw materials mainly composed of silica and alumina, and are commercially available under trade names such as ES pheres. be able to.
- S i 0 2 content of the hollow refractory particles child is less than 4 0% by weight
- a 1 2 0 3 content exceeds 6 0 wt% If, sinterability of the hollow refractory particles And the bonding of the structure of the heat insulating layer at the time of heating at a high temperature becomes insufficient, and the coating material is easily peeled off from the base material, which is not preferable.
- S i 0 2 content of the hollow refractory particles is more than 8 0% by weight
- the content of A 1 2 0 3 is less than 2 0% by weight
- the use of carbon-containing refractories causes problems such as increased erosion of the thermal insulation coating.
- the content of S i 0 2 and A 1 2 0 3 is more preferably S i 0 2 5 0-7 0 wt%, A 1 2 0 3 is 3 0-5 0% by weight.
- the content in this case S i 0 2, A 1 2 0 3 other components is preferably 1 0% by weight or less. If the total of these components exceeds 10% by weight, the heat resistance of the hollow refractory particles decreases, and the coating material softens and melts when used at a high temperature.
- the particle size range of the hollow refractory particles of the present invention is in the range of 30 to 800 microns. If it is less than 30 microns, the kneadability with the liquid binder will be poor, and the workability on refractory containing carbon fiber will be poor, which is inappropriate. Further, when the hollow refractory particles are smaller than 30 microns, there is a problem that the heat insulating property is deteriorated. On the other hand, if the particle size of the hollow refractory particles exceeds 800 ⁇ m, the heat insulating layer is not suitable because the tissue bonding force of the heat insulating layer at the time of heating is reduced, causing cracks and easy detachment from the base material. .
- the size range of the hollow refractory particles is more preferably between 50 and 700 microns.
- the particle size referred to here indicates the average particle size of the hollow refractory particles.
- the compounding amount of the clay in the heat insulating coating material of the carbon-containing refractory of the present invention is not more than 30% by weight, and preferably in the range of 3 to 20% by weight. If the amount of the clay exceeds 30% by weight, the sintering shrinkage of the heat insulating layer due to the clay component increases during preheating and use, and cracks tend to occur in the heat insulating coating layer. Appropriate.
- the heat-insulating coating material for a carbon-containing refractory of the present invention comprises As components other than the fire particles and clay, vermiculite dehydrated from water of crystallization and / or glass powder that softens and melts in a temperature range of 500 to 110 ° C. can be used in combination. By using the vermiculite together, the heat insulating effect can be exhibited from a lower temperature range. Also, by using a glass powder that softens and melts in a temperature range of 500 to 110 ° C., the structure of the heat insulating layer can be bonded from a lower temperature.
- the mixing ratio of vermiculite is desirably 20% by weight or less.
- the amount of glass powder added is preferably 10% by weight or less. This is because if it exceeds 10% by weight, the melting of the glass at a high temperature becomes excessive, and it becomes difficult to hold the coating layer. Further, it is desirable that the total added amount of vermiculite and glass powder is 30% by weight or less.
- the softening temperature of the glass powder is optimally within the range of 500 to 110 ° C considering the general preheating conditions and the temperature conditions during use of the carbon-containing refractory. .
- the heat-insulating coating material for carbon-containing refractories of the present invention comprises, as components other than the hollow refractory particles, clay, vermiculite, and glass powder, fused quartz, mullite, magnesium, zirconia, spinel, alumina, It is also possible to use one or two or more selected from the group of the compounds of chamotte, pyroxene, and clay shale. These compounds are used to adjust the heat resistance of the heat-insulating coating layer at high temperatures and to control cracking and peeling of the heat-insulating coating material during use, depending on the heating and use conditions of the refractory containing carbon. Is to be blended.
- the addition amount of these compounds is desirably 10% by weight or less. If the content exceeds 10% by weight, sintering shrinkage due to these components will increase during heating and during use, causing cracks in the heat insulating layer and problems such as easy separation of the base material and the heat insulating layer. It is inappropriate.
- the above-mentioned hollow heat-resistant particles and refractory raw materials such as clay, vermiculite and glass powder are mixed using a liquid binder.
- a liquid binder it is preferable to apply a coating material to the surface of the carbon-containing refractory and then dry it at a temperature of 300 ° C. or less to form a coating film.
- a coating material for example, potassium silicate, aluminum phosphate, colloidal Silica or the like can be used. It is also possible to use several kinds of liquid binders together
- the amount of liquid binder to be added is 20 to 170 weight per unit of refractory material. %, Preferably 30 to 150% by weight. If the added amount of the liquid binder is less than 20% by weight, the effect of the addition is not exhibited, and it is difficult to apply the liquid binder to the surface of the refractory containing carbon. On the other hand, if the amount is more than 170% by weight, it is not preferable because the coating property on the carbon-containing refractory decreases due to the decrease in viscosity.
- the method of applying to the surface of the refractory containing carbon is not limited, and the method may be selected according to the shape and size of the refractory containing carbon to be applied, the construction environment, and the like. it can.
- the coating material of the carbon-containing refractory of the present invention will be further described with reference to examples.
- the present invention is not limited by the examples.
- the coating materials of the present invention and comparative products were obtained at the mixing ratios shown in Table 1 below.
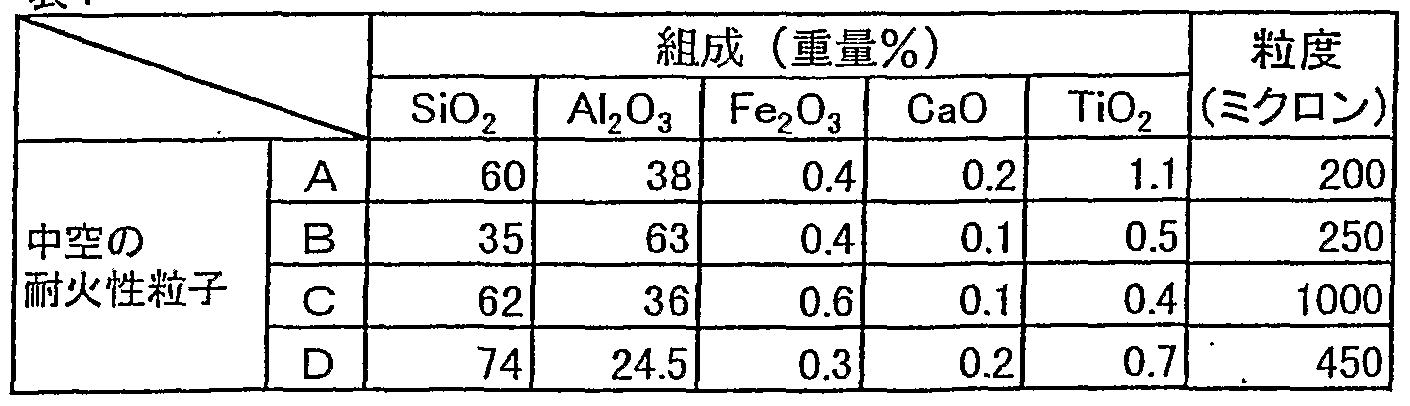
- the hollow refractory particles used here had the following appearance: white, bulk specific gravity: 0.25-0.42 g / cm 3 , and the composition and particle size were as follows:
- the obtained coating material A 1 2 0 3 5 0 wt%, S i 0 2 2 5 wt%, the immersion nozzle for continuous ⁇ made of carbon-containing refractory having a composition of carbon 2 5 wt% After spray coating the outer surface to a thickness of about 3 mm and drying at 130 ° C for 4 hours, set the continuous production immersion nozzle (2) in the heating box (1) shown in Fig. 1 Heat to 125 ° C with a submerged (4), hold at that temperature for 6 hours, Remove from the box (1), leave the continuous production immersion nozzle (2) in the air, and check the temperature drop (after 10 minutes) at the temperature measurement position (5) under the coating material coating (3). It was measured. After cooling, the state of the coating film of the coating material was visually observed.
- the glass powder used in the product 1 of the present invention had a melting softening temperature of 1000 ° C.
- Comparative product 1 was a blank that was not sprayed with a coating material.
- Comparative product 2 was a 3 mm-thick ceramic paper stuck to a continuous production immersion nozzle of the same material as above. Things.
- Example 3 Compounds of the present invention and comparative products were obtained at the compounding ratios shown in Table 3 below.
- the hollow refractory particles D of Example 1 were used as the hollow refractory particles used in Example 2.
- the same raw materials as those used in Example 1 were used for glass powder, clay and vermiculite.
- Fused quartz, mullite, magnesia, zirconia, spinel, alumina, chamotte, pyroxene, and sand shale were all made from raw materials of 0.3 mm or less.
- the resulting coating material A 1 2 0 3 60 wt%, Si0 2 10 wt%, the continuous ⁇ kiln immersion nozzle outer surface of Le composed of carbon-containing refractory having a composition of forces one carbon 30 wt% to about
- a heating test and evaluation were performed in the same manner as in Example 1. Table 3 shows the obtained results.
- the heat insulation layer which consists of a coating material coating film can be formed, and it has the effect that the continuous Useful as thermal insulation coating material in construction 0
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Ceramic Engineering (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Structural Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Inorganic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Physics & Mathematics (AREA)
- Plasma & Fusion (AREA)
- Thermal Sciences (AREA)
- Nanotechnology (AREA)
- Ceramic Products (AREA)
- Carbon Steel Or Casting Steel Manufacturing (AREA)
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/489,620 US20040241446A1 (en) | 2001-09-26 | 2002-09-24 | Thermally insulating coating material, for refractory containing carbon |
| EP02768012A EP1443031A1 (en) | 2001-09-26 | 2002-09-24 | Thermally insulating coating material for refractory containing carbon |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2001-294583 | 2001-09-26 | ||
| JP2001294583A JP2003095757A (ja) | 2001-09-26 | 2001-09-26 | カーボン含有耐火物の断熱コーティング材 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2003027040A1 true WO2003027040A1 (fr) | 2003-04-03 |
Family
ID=19116158
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2002/009765 WO2003027040A1 (fr) | 2001-09-26 | 2002-09-24 | Materiau de revetement thermo-isolant destine a un refractaire contenant du carbone |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20040241446A1 (ja) |
| EP (1) | EP1443031A1 (ja) |
| JP (1) | JP2003095757A (ja) |
| WO (1) | WO2003027040A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107840671A (zh) * | 2017-11-08 | 2018-03-27 | 武汉钢铁有限公司 | 一种焦炉用轻质保温涂层及其施工工艺 |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9968994B2 (en) * | 2005-03-24 | 2018-05-15 | Sumitomo Electric Industries, Ltd. | Casting nozzle |
| JP5465396B2 (ja) * | 2008-04-22 | 2014-04-09 | Agcセラミックス株式会社 | 低熱伝導性の断熱キャスタブル用粉体組成物 |
| CN102557683B (zh) * | 2011-12-23 | 2013-11-20 | 中钢集团洛阳耐火材料研究院有限公司 | 一种铝铬质耐火材料 |
| CN102557694B (zh) * | 2012-03-13 | 2013-06-12 | 湖南省涟源市合成耐火材料厂 | 钢包精炼炉回转台、炉盖用的隔钢分离剂及防粘钢方法 |
| DE102013009148B4 (de) * | 2013-05-31 | 2016-02-11 | Fl Services Gmbh | Mischung zur Herstellung einer Schicht in einem Schichtaufbau, Verfahren zum Herstellen eines Schichtaufbaus, Schichtaufbau und Verwendung des Schichtaufbaus |
| WO2017073115A1 (ja) * | 2015-10-27 | 2017-05-04 | 株式会社Inui | コーティング液、コーティング液用組成物及びコーティング層を有する耐火物 |
| CN105439581A (zh) * | 2015-11-27 | 2016-03-30 | 蚌埠海明压铸机有限公司 | 一种隔热耐火材料 |
| CN105439585A (zh) * | 2015-11-30 | 2016-03-30 | 蚌埠市华科机电有限责任公司 | 一种抗侵蚀耐火材料 |
| JP6372539B2 (ja) * | 2016-10-12 | 2018-08-15 | 品川リフラクトリーズ株式会社 | 連続鋳造ノズル用断熱コーティング材 |
| JP7251823B2 (ja) * | 2021-03-01 | 2023-04-04 | 明智セラミックス株式会社 | 耐火物用断熱材 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5232910A (en) * | 1975-09-09 | 1977-03-12 | Nippon Steel Corp | Lining refractories for ingot steel vessels |
| JP3080941B1 (ja) * | 1999-03-30 | 2000-08-28 | 日本プライブリコ株式会社 | 断熱吹付プラスチック耐火物 |
| JP2001335375A (ja) * | 2000-05-23 | 2001-12-04 | Shinagawa Refract Co Ltd | カーボン含有耐火物用断熱コーティング材 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4037015A (en) * | 1974-03-29 | 1977-07-19 | Hitachi, Ltd. | Heat insulating coating material |
| US4664712A (en) * | 1986-04-01 | 1987-05-12 | Ignacio Cisneros | Thermal insulating material and method of production thereof |
| WO1988002741A1 (fr) * | 1986-10-14 | 1988-04-21 | Nicolas Davidovits | Materiau composite ceramique-ceramique et procede d'obtention |
| US5482904A (en) * | 1993-03-10 | 1996-01-09 | Krosaki Corporation | Heat-insulating refractory material |
| FR2730227B1 (fr) * | 1995-02-03 | 1997-03-14 | Pechiney Recherche | Composition pour un revetement de produits carbones et ce revetement |
-
2001
- 2001-09-26 JP JP2001294583A patent/JP2003095757A/ja active Pending
-
2002
- 2002-09-24 US US10/489,620 patent/US20040241446A1/en not_active Abandoned
- 2002-09-24 EP EP02768012A patent/EP1443031A1/en not_active Withdrawn
- 2002-09-24 WO PCT/JP2002/009765 patent/WO2003027040A1/ja not_active Application Discontinuation
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5232910A (en) * | 1975-09-09 | 1977-03-12 | Nippon Steel Corp | Lining refractories for ingot steel vessels |
| JP3080941B1 (ja) * | 1999-03-30 | 2000-08-28 | 日本プライブリコ株式会社 | 断熱吹付プラスチック耐火物 |
| JP2001335375A (ja) * | 2000-05-23 | 2001-12-04 | Shinagawa Refract Co Ltd | カーボン含有耐火物用断熱コーティング材 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107840671A (zh) * | 2017-11-08 | 2018-03-27 | 武汉钢铁有限公司 | 一种焦炉用轻质保温涂层及其施工工艺 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20040241446A1 (en) | 2004-12-02 |
| JP2003095757A (ja) | 2003-04-03 |
| EP1443031A1 (en) | 2004-08-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8235092B2 (en) | Insulated investment casting mold and method of making | |
| TW562789B (en) | Insulating refractory material | |
| JPH0521865B2 (ja) | ||
| JPH0413308B2 (ja) | ||
| JPH06317383A (ja) | 断熱用耐火材料 | |
| JPS5924111B2 (ja) | ムライト質セラミツクフアイバ−の生成方法 | |
| WO2003027040A1 (fr) | Materiau de revetement thermo-isolant destine a un refractaire contenant du carbone | |
| TWI267548B (en) | Insulating refractory material | |
| US5155070A (en) | Refractory coating composition | |
| US5602063A (en) | Lightweight sprayable tundish lining composition | |
| JP2007326733A (ja) | 断熱傾斜材の製造方法及び断熱傾斜材 | |
| CA1098292A (en) | Method of making an insulating refractory | |
| BRPI0714034A2 (pt) | mistura refratária para a produção de um artigo refratário, artigo refratário, e, método para fabricar o artigo | |
| JPH04124073A (ja) | ジルコニア質複合耐火断熱材 | |
| JP2987094B2 (ja) | 高耐熱性無機質繊維成形体 | |
| JPS61295276A (ja) | 耐磨耗耐火組成物 | |
| JP5465396B2 (ja) | 低熱伝導性の断熱キャスタブル用粉体組成物 | |
| EP3464488B1 (en) | Refractory coating material containing low biopersistent fibers and method for making the same | |
| JPH01160854A (ja) | 高強度、耐摩耗性耐火キャスタブル | |
| JP2657042B2 (ja) | 黒鉛含有耐火物の断熱性酸化防止剤 | |
| JP6372539B2 (ja) | 連続鋳造ノズル用断熱コーティング材 | |
| JP3868186B2 (ja) | カーボン含有耐火物用断熱コーティング材 | |
| KR100670905B1 (ko) | 카본함유 내화물 표면에 코팅되는 코팅재 | |
| JP2004299960A (ja) | 繊維質成形体、耐熱構造体及びこれらの製造方法 | |
| JP2001206783A (ja) | 無機繊維質成形体、断熱材及びこれらの製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AU |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FR GB GR IE IT LU MC NL PT SE SK TR |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 10489620 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002332240 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002768012 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2002768012 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2002768012 Country of ref document: EP |