JP2013509914A - 複数層フィラメント状デバイスまたは血管障害処置 - Google Patents
複数層フィラメント状デバイスまたは血管障害処置 Download PDFInfo
- Publication number
- JP2013509914A JP2013509914A JP2012537226A JP2012537226A JP2013509914A JP 2013509914 A JP2013509914 A JP 2013509914A JP 2012537226 A JP2012537226 A JP 2012537226A JP 2012537226 A JP2012537226 A JP 2012537226A JP 2013509914 A JP2013509914 A JP 2013509914A
- Authority
- JP
- Japan
- Prior art keywords
- shell
- filaments
- filament
- state
- permeable shell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000011282 treatment Methods 0.000 title claims abstract description 162
- 230000002792 vascular Effects 0.000 title description 32
- 210000005166 vasculature Anatomy 0.000 claims abstract description 144
- 238000000034 method Methods 0.000 claims abstract description 95
- 230000017531 blood circulation Effects 0.000 claims abstract description 55
- 208000019553 vascular disease Diseases 0.000 claims description 64
- 206010059245 Angiopathy Diseases 0.000 claims description 54
- 208000007536 Thrombosis Diseases 0.000 claims description 47
- 239000004744 fabric Substances 0.000 claims description 29
- 210000004369 blood Anatomy 0.000 claims description 15
- 239000008280 blood Substances 0.000 claims description 15
- 230000005540 biological transmission Effects 0.000 claims description 14
- 239000000758 substrate Substances 0.000 claims 1
- 230000002490 cerebral effect Effects 0.000 abstract description 6
- 206010002329 Aneurysm Diseases 0.000 description 159
- UQMRAFJOBWOFNS-UHFFFAOYSA-N butyl 2-(2,4-dichlorophenoxy)acetate Chemical compound CCCCOC(=O)COC1=CC=C(Cl)C=C1Cl UQMRAFJOBWOFNS-UHFFFAOYSA-N 0.000 description 92
- 239000000463 material Substances 0.000 description 65
- 239000011148 porous material Substances 0.000 description 46
- 231100000216 vascular lesion Toxicity 0.000 description 40
- 210000003739 neck Anatomy 0.000 description 32
- 210000004204 blood vessel Anatomy 0.000 description 23
- 239000010410 layer Substances 0.000 description 22
- 239000007943 implant Substances 0.000 description 21
- 229920000642 polymer Polymers 0.000 description 19
- 201000008450 Intracranial aneurysm Diseases 0.000 description 18
- 238000000576 coating method Methods 0.000 description 18
- 230000036961 partial effect Effects 0.000 description 18
- 230000015572 biosynthetic process Effects 0.000 description 17
- 239000000835 fiber Substances 0.000 description 16
- 239000011248 coating agent Substances 0.000 description 15
- 238000004519 manufacturing process Methods 0.000 description 15
- 229910052751 metal Inorganic materials 0.000 description 15
- 239000002184 metal Substances 0.000 description 15
- 230000008569 process Effects 0.000 description 15
- 210000001519 tissue Anatomy 0.000 description 15
- 238000010438 heat treatment Methods 0.000 description 12
- 238000007789 sealing Methods 0.000 description 12
- 230000007246 mechanism Effects 0.000 description 11
- -1 polyparaphenylene terephthalamide Polymers 0.000 description 11
- 230000000903 blocking effect Effects 0.000 description 10
- 229910045601 alloy Inorganic materials 0.000 description 9
- 239000000956 alloy Substances 0.000 description 9
- 210000001367 artery Anatomy 0.000 description 9
- 239000004020 conductor Substances 0.000 description 9
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 9
- 208000035475 disorder Diseases 0.000 description 9
- 239000003550 marker Substances 0.000 description 9
- 210000004556 brain Anatomy 0.000 description 8
- 230000035602 clotting Effects 0.000 description 8
- 229940079593 drug Drugs 0.000 description 8
- 239000003814 drug Substances 0.000 description 8
- 230000035876 healing Effects 0.000 description 8
- 238000002513 implantation Methods 0.000 description 8
- 230000003902 lesion Effects 0.000 description 8
- 239000004753 textile Substances 0.000 description 8
- 230000001154 acute effect Effects 0.000 description 7
- 239000012530 fluid Substances 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 229910001000 nickel titanium Inorganic materials 0.000 description 7
- 238000001356 surgical procedure Methods 0.000 description 7
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical compound O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 6
- 238000009954 braiding Methods 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 239000002131 composite material Substances 0.000 description 6
- 229920001577 copolymer Polymers 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 230000003073 embolic effect Effects 0.000 description 6
- 230000023597 hemostasis Effects 0.000 description 6
- 230000001788 irregular Effects 0.000 description 6
- 238000005259 measurement Methods 0.000 description 6
- 150000002739 metals Chemical class 0.000 description 6
- 238000002203 pretreatment Methods 0.000 description 6
- 230000002441 reversible effect Effects 0.000 description 6
- 238000004381 surface treatment Methods 0.000 description 6
- 229920000954 Polyglycolide Polymers 0.000 description 5
- 239000012620 biological material Substances 0.000 description 5
- 230000004087 circulation Effects 0.000 description 5
- 230000010102 embolization Effects 0.000 description 5
- 238000003780 insertion Methods 0.000 description 5
- 230000037431 insertion Effects 0.000 description 5
- 229920000728 polyester Polymers 0.000 description 5
- 239000004633 polyglycolic acid Substances 0.000 description 5
- 230000007556 vascular defect Effects 0.000 description 5
- 238000003466 welding Methods 0.000 description 5
- 206010053567 Coagulopathies Diseases 0.000 description 4
- 230000006978 adaptation Effects 0.000 description 4
- 238000002583 angiography Methods 0.000 description 4
- 230000009286 beneficial effect Effects 0.000 description 4
- 239000012867 bioactive agent Substances 0.000 description 4
- 230000006835 compression Effects 0.000 description 4
- 238000007906 compression Methods 0.000 description 4
- 230000007547 defect Effects 0.000 description 4
- 210000001105 femoral artery Anatomy 0.000 description 4
- 238000011049 filling Methods 0.000 description 4
- 238000009998 heat setting Methods 0.000 description 4
- 230000001965 increasing effect Effects 0.000 description 4
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 238000005476 soldering Methods 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- 102000008186 Collagen Human genes 0.000 description 3
- 108010035532 Collagen Proteins 0.000 description 3
- 229920000106 Liquid crystal polymer Polymers 0.000 description 3
- 239000004977 Liquid-crystal polymers (LCPs) Substances 0.000 description 3
- 239000000853 adhesive Substances 0.000 description 3
- 230000001070 adhesive effect Effects 0.000 description 3
- 238000013459 approach Methods 0.000 description 3
- 230000000975 bioactive effect Effects 0.000 description 3
- 229920001436 collagen Polymers 0.000 description 3
- 238000010276 construction Methods 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 3
- 229910052737 gold Inorganic materials 0.000 description 3
- 239000010931 gold Substances 0.000 description 3
- 238000002955 isolation Methods 0.000 description 3
- 238000002844 melting Methods 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 230000035699 permeability Effects 0.000 description 3
- 238000001259 photo etching Methods 0.000 description 3
- 229920001610 polycaprolactone Polymers 0.000 description 3
- 239000004632 polycaprolactone Substances 0.000 description 3
- 239000004626 polylactic acid Substances 0.000 description 3
- 239000004814 polyurethane Substances 0.000 description 3
- 108090000765 processed proteins & peptides Proteins 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 230000001732 thrombotic effect Effects 0.000 description 3
- 238000009941 weaving Methods 0.000 description 3
- 208000005189 Embolism Diseases 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 239000004593 Epoxy Substances 0.000 description 2
- OHCQJHSOBUTRHG-KGGHGJDLSA-N FORSKOLIN Chemical compound O=C([C@@]12O)C[C@](C)(C=C)O[C@]1(C)[C@@H](OC(=O)C)[C@@H](O)[C@@H]1[C@]2(C)[C@@H](O)CCC1(C)C OHCQJHSOBUTRHG-KGGHGJDLSA-N 0.000 description 2
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 2
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 2
- 108010007267 Hirudins Proteins 0.000 description 2
- 102000007625 Hirudins Human genes 0.000 description 2
- 229920000271 Kevlar® Polymers 0.000 description 2
- 229920001410 Microfiber Polymers 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 239000004696 Poly ether ether ketone Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- HZEWFHLRYVTOIW-UHFFFAOYSA-N [Ti].[Ni] Chemical compound [Ti].[Ni] HZEWFHLRYVTOIW-UHFFFAOYSA-N 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 238000004026 adhesive bonding Methods 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 239000003146 anticoagulant agent Substances 0.000 description 2
- 210000000709 aorta Anatomy 0.000 description 2
- 239000004760 aramid Substances 0.000 description 2
- 229920006231 aramid fiber Polymers 0.000 description 2
- KXNPVXPOPUZYGB-XYVMCAHJSA-N argatroban Chemical compound OC(=O)[C@H]1C[C@H](C)CCN1C(=O)[C@H](CCCN=C(N)N)NS(=O)(=O)C1=CC=CC2=C1NC[C@H](C)C2 KXNPVXPOPUZYGB-XYVMCAHJSA-N 0.000 description 2
- 229960003856 argatroban Drugs 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000002872 contrast media Substances 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 230000010339 dilation Effects 0.000 description 2
- 229940088598 enzyme Drugs 0.000 description 2
- 229960001123 epoprostenol Drugs 0.000 description 2
- KAQKFAOMNZTLHT-VVUHWYTRSA-N epoprostenol Chemical compound O1C(=CCCCC(O)=O)C[C@@H]2[C@@H](/C=C/[C@@H](O)CCCCC)[C@H](O)C[C@@H]21 KAQKFAOMNZTLHT-VVUHWYTRSA-N 0.000 description 2
- 239000010408 film Substances 0.000 description 2
- 238000002594 fluoroscopy Methods 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- 229960002897 heparin Drugs 0.000 description 2
- 229920000669 heparin Polymers 0.000 description 2
- WQPDUTSPKFMPDP-OUMQNGNKSA-N hirudin Chemical compound C([C@@H](C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C=CC(OS(O)(=O)=O)=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCCCN)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H]1NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@@H]2CSSC[C@@H](C(=O)N[C@@H](CCC(O)=O)C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](C(=O)N[C@H](C(NCC(=O)N[C@@H](CCC(N)=O)C(=O)NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N2)=O)CSSC1)C(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=2C=CC(O)=CC=2)NC(=O)[C@@H](NC(=O)[C@@H](N)C(C)C)C(C)C)[C@@H](C)O)CSSC1)C(C)C)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 WQPDUTSPKFMPDP-OUMQNGNKSA-N 0.000 description 2
- 229940006607 hirudin Drugs 0.000 description 2
- 238000003384 imaging method Methods 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 239000004761 kevlar Substances 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 229910052697 platinum Inorganic materials 0.000 description 2
- 229920001432 poly(L-lactide) Polymers 0.000 description 2
- 229920002530 polyetherether ketone Polymers 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 description 2
- 229920002635 polyurethane Polymers 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 210000002321 radial artery Anatomy 0.000 description 2
- 230000000452 restraining effect Effects 0.000 description 2
- 229910000679 solder Inorganic materials 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 229910052715 tantalum Inorganic materials 0.000 description 2
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 2
- 239000010409 thin film Substances 0.000 description 2
- SJZRECIVHVDYJC-UHFFFAOYSA-N 4-hydroxybutyric acid Chemical compound OCCCC(O)=O SJZRECIVHVDYJC-UHFFFAOYSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 108010074415 Angiogenic Proteins Proteins 0.000 description 1
- 102000008076 Angiogenic Proteins Human genes 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- 239000005552 B01AC04 - Clopidogrel Substances 0.000 description 1
- 239000005528 B01AC05 - Ticlopidine Substances 0.000 description 1
- 235000014036 Castanea Nutrition 0.000 description 1
- 241001070941 Castanea Species 0.000 description 1
- 206010008111 Cerebral haemorrhage Diseases 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- SUZLHDUTVMZSEV-UHFFFAOYSA-N Deoxycoleonol Natural products C12C(=O)CC(C)(C=C)OC2(C)C(OC(=O)C)C(O)C2C1(C)C(O)CCC2(C)C SUZLHDUTVMZSEV-UHFFFAOYSA-N 0.000 description 1
- 208000036829 Device dislocation Diseases 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- 238000012276 Endovascular treatment Methods 0.000 description 1
- 108010056764 Eptifibatide Proteins 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 1
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 1
- 102000009123 Fibrin Human genes 0.000 description 1
- 108010073385 Fibrin Proteins 0.000 description 1
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 1
- 108010049003 Fibrinogen Proteins 0.000 description 1
- 102000008946 Fibrinogen Human genes 0.000 description 1
- 102000016359 Fibronectins Human genes 0.000 description 1
- 108010067306 Fibronectins Proteins 0.000 description 1
- 206010016654 Fibrosis Diseases 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 description 1
- 229920002633 Kraton (polymer) Polymers 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- OVRNDRQMDRJTHS-FMDGEEDCSA-N N-acetyl-beta-D-glucosamine Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-FMDGEEDCSA-N 0.000 description 1
- 229910000990 Ni alloy Inorganic materials 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 229920002732 Polyanhydride Polymers 0.000 description 1
- 229920002614 Polyether block amide Polymers 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 229920002873 Polyethylenimine Polymers 0.000 description 1
- 108010039918 Polylysine Proteins 0.000 description 1
- 229920000388 Polyphosphate Polymers 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- 208000002847 Surgical Wound Diseases 0.000 description 1
- 108090000190 Thrombin Proteins 0.000 description 1
- 102000003938 Thromboxane Receptors Human genes 0.000 description 1
- 108090000300 Thromboxane Receptors Proteins 0.000 description 1
- 229910001069 Ti alloy Inorganic materials 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 206010053648 Vascular occlusion Diseases 0.000 description 1
- 229920000508 Vectran Polymers 0.000 description 1
- 239000004979 Vectran Substances 0.000 description 1
- 229910001080 W alloy Inorganic materials 0.000 description 1
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 1
- 229960000446 abciximab Drugs 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 230000002745 absorbent Effects 0.000 description 1
- 239000002250 absorbent Substances 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 239000002390 adhesive tape Substances 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 229940061720 alpha hydroxy acid Drugs 0.000 description 1
- 150000001280 alpha hydroxy acids Chemical class 0.000 description 1
- WYTGDNHDOZPMIW-RCBQFDQVSA-N alstonine Natural products C1=CC2=C3C=CC=CC3=NC2=C2N1C[C@H]1[C@H](C)OC=C(C(=O)OC)[C@H]1C2 WYTGDNHDOZPMIW-RCBQFDQVSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 125000000539 amino acid group Chemical class 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 230000002965 anti-thrombogenic effect Effects 0.000 description 1
- 229940127219 anticoagulant drug Drugs 0.000 description 1
- 229940030225 antihemorrhagics Drugs 0.000 description 1
- 229940127218 antiplatelet drug Drugs 0.000 description 1
- 239000004019 antithrombin Substances 0.000 description 1
- 229960004676 antithrombotic agent Drugs 0.000 description 1
- 210000002376 aorta thoracic Anatomy 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 210000001841 basilar artery Anatomy 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000000227 bioadhesive Substances 0.000 description 1
- 239000003124 biologic agent Substances 0.000 description 1
- OIRCOABEOLEUMC-GEJPAHFPSA-N bivalirudin Chemical compound C([C@@H](C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CC(C)C)C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)CNC(=O)CNC(=O)CNC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 OIRCOABEOLEUMC-GEJPAHFPSA-N 0.000 description 1
- 108010055460 bivalirudin Proteins 0.000 description 1
- 229960001500 bivalirudin Drugs 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 230000023555 blood coagulation Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 210000002302 brachial artery Anatomy 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 210000001715 carotid artery Anatomy 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000013043 chemical agent Substances 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000000788 chromium alloy Substances 0.000 description 1
- 229960004588 cilostazol Drugs 0.000 description 1
- RRGUKTPIGVIEKM-UHFFFAOYSA-N cilostazol Chemical compound C=1C=C2NC(=O)CCC2=CC=1OCCCCC1=NN=NN1C1CCCCC1 RRGUKTPIGVIEKM-UHFFFAOYSA-N 0.000 description 1
- GKTWGGQPFAXNFI-HNNXBMFYSA-N clopidogrel Chemical compound C1([C@H](N2CC=3C=CSC=3CC2)C(=O)OC)=CC=CC=C1Cl GKTWGGQPFAXNFI-HNNXBMFYSA-N 0.000 description 1
- 229960003009 clopidogrel Drugs 0.000 description 1
- 230000015271 coagulation Effects 0.000 description 1
- 238000005345 coagulation Methods 0.000 description 1
- OHCQJHSOBUTRHG-UHFFFAOYSA-N colforsin Natural products OC12C(=O)CC(C)(C=C)OC1(C)C(OC(=O)C)C(O)C1C2(C)C(O)CCC1(C)C OHCQJHSOBUTRHG-UHFFFAOYSA-N 0.000 description 1
- 229960005188 collagen Drugs 0.000 description 1
- 238000007428 craniotomy Methods 0.000 description 1
- 238000002788 crimping Methods 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000006837 decompression Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 229960002768 dipyridamole Drugs 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 238000005868 electrolysis reaction Methods 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 229960004468 eptifibatide Drugs 0.000 description 1
- GLGOPUHVAZCPRB-LROMGURASA-N eptifibatide Chemical compound N1C(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CCCCNC(=N)N)NC(=O)CCSSC[C@@H](C(N)=O)NC(=O)[C@@H]2CCCN2C(=O)[C@@H]1CC1=CN=C2[C]1C=CC=C2 GLGOPUHVAZCPRB-LROMGURASA-N 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- VJDOPFARMOLELX-ZDUSSCGKSA-N ethyl 3-[[(3s)-1-(4-carbamimidoylphenyl)-2-oxopyrrolidin-3-yl]carbamoylamino]propanoate Chemical compound O=C1[C@@H](NC(=O)NCCC(=O)OCC)CCN1C1=CC=C(C(N)=N)C=C1 VJDOPFARMOLELX-ZDUSSCGKSA-N 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 229950003499 fibrin Drugs 0.000 description 1
- 229940012952 fibrinogen Drugs 0.000 description 1
- 230000004761 fibrosis Effects 0.000 description 1
- 230000003176 fibrotic effect Effects 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 230000036449 good health Effects 0.000 description 1
- 239000002874 hemostatic agent Substances 0.000 description 1
- 230000002439 hemostatic effect Effects 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 description 1
- 230000003100 immobilizing effect Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- FZWBNHMXJMCXLU-BLAUPYHCSA-N isomaltotriose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O)O1 FZWBNHMXJMCXLU-BLAUPYHCSA-N 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 229950003178 lamifiban Drugs 0.000 description 1
- FPKOGTAFKSLZLD-FQEVSTJZSA-N lamifiban Chemical compound C1=CC(C(=N)N)=CC=C1C(=O)N[C@H](C(=O)N1CCC(CC1)OCC(O)=O)CC1=CC=C(O)C=C1 FPKOGTAFKSLZLD-FQEVSTJZSA-N 0.000 description 1
- 238000003698 laser cutting Methods 0.000 description 1
- 239000004816 latex Substances 0.000 description 1
- 229920000126 latex Polymers 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000003055 low molecular weight heparin Substances 0.000 description 1
- 229940127215 low-molecular weight heparin Drugs 0.000 description 1
- 238000003754 machining Methods 0.000 description 1
- 230000013011 mating Effects 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 239000003658 microfiber Substances 0.000 description 1
- 238000002324 minimally invasive surgery Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 229950006780 n-acetylglucosamine Drugs 0.000 description 1
- 238000011587 new zealand white rabbit Methods 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 239000013307 optical fiber Substances 0.000 description 1
- 229950002383 orbofiban Drugs 0.000 description 1
- 229940019331 other antithrombotic agent in atc Drugs 0.000 description 1
- 238000000059 patterning Methods 0.000 description 1
- 230000010412 perfusion Effects 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 229940096701 plain lipid modifying drug hmg coa reductase inhibitors Drugs 0.000 description 1
- 239000000106 platelet aggregation inhibitor Substances 0.000 description 1
- ZONODCCBXBRQEZ-UHFFFAOYSA-N platinum tungsten Chemical compound [W].[Pt] ZONODCCBXBRQEZ-UHFFFAOYSA-N 0.000 description 1
- 229920001308 poly(aminoacid) Polymers 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 229920002463 poly(p-dioxanone) polymer Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 239000000622 polydioxanone Substances 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 229920000656 polylysine Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920006124 polyolefin elastomer Polymers 0.000 description 1
- 239000001205 polyphosphate Substances 0.000 description 1
- 235000011176 polyphosphates Nutrition 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 150000004804 polysaccharides Chemical class 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 238000004382 potting Methods 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 150000003815 prostacyclins Chemical class 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 108091006082 receptor inhibitors Proteins 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 239000012783 reinforcing fiber Substances 0.000 description 1
- 238000007634 remodeling Methods 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 239000012858 resilient material Substances 0.000 description 1
- 150000003839 salts Chemical group 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 239000012781 shape memory material Substances 0.000 description 1
- 238000007493 shaping process Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 238000004513 sizing Methods 0.000 description 1
- 210000003625 skull Anatomy 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 239000002195 soluble material Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 208000010110 spontaneous platelet aggregation Diseases 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 210000004876 tela submucosa Anatomy 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- 230000003685 thermal hair damage Effects 0.000 description 1
- 238000007669 thermal treatment Methods 0.000 description 1
- 229960004072 thrombin Drugs 0.000 description 1
- 230000002885 thrombogenetic effect Effects 0.000 description 1
- 108010065972 tick anticoagulant peptide Proteins 0.000 description 1
- PHWBOXQYWZNQIN-UHFFFAOYSA-N ticlopidine Chemical compound ClC1=CC=CC=C1CN1CC(C=CS2)=C2CC1 PHWBOXQYWZNQIN-UHFFFAOYSA-N 0.000 description 1
- 229960005001 ticlopidine Drugs 0.000 description 1
- 229960003425 tirofiban Drugs 0.000 description 1
- COKMIXFXJJXBQG-NRFANRHFSA-N tirofiban Chemical compound C1=CC(C[C@H](NS(=O)(=O)CCCC)C(O)=O)=CC=C1OCCCCC1CCNCC1 COKMIXFXJJXBQG-NRFANRHFSA-N 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000005945 translocation Effects 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- SBHRWOBHKASWGU-UHFFFAOYSA-M tridodecyl(methyl)azanium;chloride Chemical compound [Cl-].CCCCCCCCCCCC[N+](C)(CCCCCCCCCCCC)CCCCCCCCCCCC SBHRWOBHKASWGU-UHFFFAOYSA-M 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- 229920000785 ultra high molecular weight polyethylene Polymers 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 208000021331 vascular occlusion disease Diseases 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 108010047303 von Willebrand Factor Proteins 0.000 description 1
- 102100036537 von Willebrand factor Human genes 0.000 description 1
- 229960001134 von willebrand factor Drugs 0.000 description 1
- 229960005080 warfarin Drugs 0.000 description 1
- PJVWKTKQMONHTI-UHFFFAOYSA-N warfarin Chemical compound OC=1C2=CC=CC=C2OC(=O)C=1C(CC(=O)C)C1=CC=CC=C1 PJVWKTKQMONHTI-UHFFFAOYSA-N 0.000 description 1
- ZXIBCJHYVWYIKI-PZJWPPBQSA-N ximelagatran Chemical compound C1([C@@H](NCC(=O)OCC)C(=O)N2[C@@H](CC2)C(=O)NCC=2C=CC(=CC=2)C(\N)=N\O)CCCCC1 ZXIBCJHYVWYIKI-PZJWPPBQSA-N 0.000 description 1
- 229960001522 ximelagatran Drugs 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
- A61B17/12113—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
- A61B17/12113—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm
- A61B17/12118—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm for positioning in conjunction with a stent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/12168—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a mesh structure
- A61B17/12172—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a mesh structure having a pre-set deployed three-dimensional shape
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/12168—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a mesh structure
- A61B17/12177—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a mesh structure comprising additional materials, e.g. thrombogenic, having filaments, having fibers or being coated
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B2017/00526—Methods of manufacturing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B2017/1205—Introduction devices
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/22—Implements for squeezing-off ulcers or the like on inner organs of the body; Implements for scraping-out cavities of body organs, e.g. bones; for invasive removal or destruction of calculus using mechanical vibrations; for removing obstructions in blood vessels, not otherwise provided for
- A61B2017/22051—Implements for squeezing-off ulcers or the like on inner organs of the body; Implements for scraping-out cavities of body organs, e.g. bones; for invasive removal or destruction of calculus using mechanical vibrations; for removing obstructions in blood vessels, not otherwise provided for with an inflatable part, e.g. balloon, for positioning, blocking, or immobilisation
- A61B2017/22065—Functions of balloons
- A61B2017/22067—Blocking; Occlusion
Landscapes
- Health & Medical Sciences (AREA)
- Surgery (AREA)
- Life Sciences & Earth Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Molecular Biology (AREA)
- Vascular Medicine (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Reproductive Health (AREA)
- Medical Informatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Neurosurgery (AREA)
- Surgical Instruments (AREA)
- Prostheses (AREA)
- External Artificial Organs (AREA)
Applications Claiming Priority (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US25854109P | 2009-11-05 | 2009-11-05 | |
| US61/258,541 | 2009-11-05 | ||
| US29476010P | 2010-01-13 | 2010-01-13 | |
| US61/294,760 | 2010-01-13 | ||
| US33413010P | 2010-05-12 | 2010-05-12 | |
| US61/334,130 | 2010-05-12 | ||
| PCT/US2010/055494 WO2011057002A2 (en) | 2009-11-05 | 2010-11-04 | Multiple layer filamentary devices or treatment of vascular defects |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2013509914A true JP2013509914A (ja) | 2013-03-21 |
| JP2013509914A5 JP2013509914A5 (enExample) | 2013-11-21 |
Family
ID=43970761
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012537226A Pending JP2013509914A (ja) | 2009-11-05 | 2010-11-04 | 複数層フィラメント状デバイスまたは血管障害処置 |
Country Status (8)
| Country | Link |
|---|---|
| US (3) | US20110152993A1 (enExample) |
| EP (1) | EP2496299B1 (enExample) |
| JP (1) | JP2013509914A (enExample) |
| CN (1) | CN102639181A (enExample) |
| AU (1) | AU2010315106A1 (enExample) |
| BR (1) | BR112012010758A2 (enExample) |
| CA (1) | CA2778639A1 (enExample) |
| WO (1) | WO2011057002A2 (enExample) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014512872A (ja) * | 2011-02-11 | 2014-05-29 | コヴィディエン リミテッド パートナーシップ | 二段階配備動脈瘤塞栓用デバイス |
| JP2016096917A (ja) * | 2014-11-19 | 2016-05-30 | グンゼ株式会社 | 医療用材料 |
| JP2017511202A (ja) * | 2014-04-14 | 2017-04-20 | シークエント メディカル インコーポレイテッド | 処置的血管手技のためのデバイス |
| JP2018538064A (ja) * | 2015-12-07 | 2018-12-27 | シーラス エンドバスキュラー リミテッド | 閉塞デバイス |
| JP2019506230A (ja) * | 2016-02-10 | 2019-03-07 | マイクロベンション インコーポレイテッドMicrovention, Inc. | 血管閉塞用デバイス |
| JP2019111343A (ja) * | 2017-12-22 | 2019-07-11 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 動脈瘤装置及び送達システム |
| JP2019209145A (ja) * | 2018-05-31 | 2019-12-12 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 動脈瘤装置及び送達システム |
| JP2022525317A (ja) * | 2019-03-15 | 2022-05-12 | シークエント メディカル インコーポレイテッド | 血管障害の治療のための可撓性継合部を有するフィラメント状デバイス |
| JP2022525745A (ja) * | 2019-03-15 | 2022-05-19 | シークエント メディカル インコーポレイテッド | 血管障害の治療のためのフィラメント状デバイス |
| JP2022532912A (ja) * | 2019-05-25 | 2022-07-20 | ギャラクシー セラピューティクス, インコーポレイテッド | 動脈瘤を治療するためのシステムおよび方法 |
| WO2022162971A1 (ja) * | 2021-01-27 | 2022-08-04 | 株式会社SG-1Medical | 血管プラグ及び治療デバイス |
| US11406404B2 (en) | 2020-02-20 | 2022-08-09 | Cerus Endovascular Limited | Clot removal distal protection methods |
| US11648013B2 (en) | 2016-03-11 | 2023-05-16 | Cerus Endovascular Limited | Occlusion device |
| US12029431B2 (en) | 2014-04-30 | 2024-07-09 | Stryker Ireland Technology, Ltd. | Occlusion device |
Families Citing this family (199)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005094283A2 (en) | 2004-03-25 | 2005-10-13 | Hauser David L | Vascular filter device |
| EP1982655B2 (de) * | 2007-04-16 | 2022-12-07 | Occlutech Holding AG | Occluder zum Verschliessen eines Herzohres und Herstellungsverfahren dafür |
| EP2157937B1 (en) | 2007-06-04 | 2017-03-22 | Sequent Medical, Inc. | Devices for treatment of vascular defects |
| US9034007B2 (en) | 2007-09-21 | 2015-05-19 | Insera Therapeutics, Inc. | Distal embolic protection devices with a variable thickness microguidewire and methods for their use |
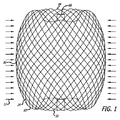
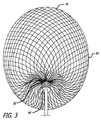
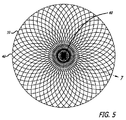
| ES2430117T3 (es) | 2008-04-21 | 2013-11-19 | Covidien Lp | Dispositivos embólicos de bola de trenza y sistemas de colocación |
| US12004750B2 (en) | 2008-05-01 | 2024-06-11 | Aneuclose Llc | Methods for creating an expandable two-part intrasacular aneurysm occlusion device from a tubular mesh |
| US11464518B2 (en) | 2008-05-01 | 2022-10-11 | Aneuclose Llc | Proximal concave neck bridge with central lumen and distal net for occluding cerebral aneurysms |
| US10716573B2 (en) | 2008-05-01 | 2020-07-21 | Aneuclose | Janjua aneurysm net with a resilient neck-bridging portion for occluding a cerebral aneurysm |
| US11484322B2 (en) | 2018-01-03 | 2022-11-01 | Aneuclose Llc | Aneurysm neck bridge with a closeable opening or lumen through which embolic material is inserted into the aneurysm sac |
| US11357511B2 (en) | 2008-05-01 | 2022-06-14 | Aneuclose Llc | Intrasacular aneurysm occlusion device with globular first configuration and bowl-shaped second configuration |
| US11471164B2 (en) | 2008-05-01 | 2022-10-18 | Aneuclose Llc | Methods of occluding a cerebral aneurysm by inserting embolic members or material into an intrasacular implant |
| US11583289B2 (en) | 2008-05-01 | 2023-02-21 | Aneuclose Llc | Aneurysm-occluding mesh ribbon with a series of loops or segments having distal-to-proximal variation in size, shape, and/or orientation |
| US11471163B2 (en) | 2008-05-01 | 2022-10-18 | Aneuclose Llc | Intrasaccular aneurysm occlusion device with net or mesh expanded by string-of-pearls embolies |
| US10028747B2 (en) | 2008-05-01 | 2018-07-24 | Aneuclose Llc | Coils with a series of proximally-and-distally-connected loops for occluding a cerebral aneurysm |
| BRPI0911923B8 (pt) * | 2008-05-02 | 2021-06-22 | Sequent Medical Inc | dispositivo para tratamento de um aneurisma cerebral |
| WO2009140437A1 (en) | 2008-05-13 | 2009-11-19 | Nfocus Neuromedical, Inc. | Braid implant delivery systems |
| CA2731735A1 (en) | 2008-07-22 | 2010-01-28 | Microtherapeutics, Inc. | Vascular remodeling device |
| US9402707B2 (en) | 2008-07-22 | 2016-08-02 | Neuravi Limited | Clot capture systems and associated methods |
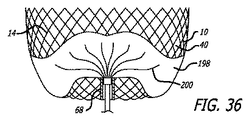
| US20110152993A1 (en) | 2009-11-05 | 2011-06-23 | Sequent Medical Inc. | Multiple layer filamentary devices or treatment of vascular defects |
| CN102791205B (zh) | 2009-11-09 | 2016-02-03 | 恩福克斯神经医学股份有限公司 | 栓塞装置 |
| US9358140B1 (en) | 2009-11-18 | 2016-06-07 | Aneuclose Llc | Stent with outer member to embolize an aneurysm |
| US9468442B2 (en) | 2010-01-28 | 2016-10-18 | Covidien Lp | Vascular remodeling device |
| EP2528541B1 (en) | 2010-01-28 | 2016-05-18 | Covidien LP | Vascular remodeling device |
| EP2539012B1 (en) | 2010-02-23 | 2018-01-24 | Covidien LP | Devices for vascular recanalization |
| US20180049731A1 (en) * | 2010-04-29 | 2018-02-22 | Muffin Incorporated | Closing device for tissue openings |
| US10568628B2 (en) | 2017-05-23 | 2020-02-25 | Muffin Incorporated | Closing device for tissue openings |
| AU2011298843A1 (en) * | 2010-09-06 | 2013-04-04 | Nonwotecc Medical Gmbh | Device for closing openings or cavities in blood vessels |
| EP2613735B1 (en) | 2010-09-10 | 2018-05-09 | Covidien LP | Devices for the treatment of vascular defects |
| US8998947B2 (en) | 2010-09-10 | 2015-04-07 | Medina Medical, Inc. | Devices and methods for the treatment of vascular defects |
| CN101933850B (zh) * | 2010-09-16 | 2012-07-18 | 先健科技(深圳)有限公司 | 封堵器及其制造方法 |
| ES2683943T3 (es) | 2010-10-22 | 2018-09-28 | Neuravi Limited | Sistema de captura y extirpación de coágulos |
| US12076037B2 (en) | 2011-03-09 | 2024-09-03 | Neuravi Limited | Systems and methods to restore perfusion to a vessel |
| ES3029850T3 (en) | 2011-03-09 | 2025-06-25 | Neuravi Ltd | A clot retrieval device for removing occlusive clot from a blood vessel |
| US11259824B2 (en) | 2011-03-09 | 2022-03-01 | Neuravi Limited | Clot retrieval device for removing occlusive clot from a blood vessel |
| US9089332B2 (en) | 2011-03-25 | 2015-07-28 | Covidien Lp | Vascular remodeling device |
| WO2013005195A1 (en) * | 2011-07-07 | 2013-01-10 | Jayandiran Pillai | Aneurysm occluder |
| EP2744423A4 (en) * | 2011-08-19 | 2015-06-24 | Inceptus Medical LLC | DEABLABLE OCCLUSION DEVICE AND METHOD THEREFOR |
| US9060886B2 (en) | 2011-09-29 | 2015-06-23 | Covidien Lp | Vascular remodeling device |
| EP2606829A1 (en) * | 2011-12-23 | 2013-06-26 | Occlutech Holding AG | A medical implant, a kit and a method of manufacturing a 3D fabric of strands for forming a medical implant |
| SG11201401703UA (en) * | 2011-10-27 | 2014-09-26 | Occlutech Holding Ag | A medical implant, a kit and a method of manufacturing a 3d fabric of strands for forming a medical implant |
| US8758389B2 (en) * | 2011-11-18 | 2014-06-24 | Aga Medical Corporation | Devices and methods for occluding abnormal openings in a patient's vasculature |
| CN104039246A (zh) * | 2012-01-06 | 2014-09-10 | 因赛普特斯医学有限责任公司 | 可膨胀的闭塞装置及使用方法 |
| US9113890B2 (en) | 2012-02-06 | 2015-08-25 | Aga Medical Corporation | Devices and methods for occluding vascular abnormalities |
| EP2827783B1 (en) * | 2012-03-22 | 2021-04-28 | C.R. Bard Inc. | Implantable prosthesis for soft tissue repair |
| DE102012102844B4 (de) * | 2012-04-02 | 2020-03-19 | Acandis Gmbh | Okklusionsvorrichtung zur Implantation innerhalb eines Aneurysmas und Anordnung mit einer derartigen Okklusionsvorrichtung |
| US20150133989A1 (en) * | 2012-04-20 | 2015-05-14 | Inceptus Medical, Llc | Expandable occlusion devices and methods of use |
| JP6133983B2 (ja) * | 2012-07-13 | 2017-05-24 | ボストン サイエンティフィック サイムド,インコーポレイテッドBoston Scientific Scimed,Inc. | 心耳用閉塞装置およびその製造方法 |
| DK3821830T3 (da) | 2012-09-24 | 2024-11-11 | Inari Medical Inc | Anordning og fremgangsmåde til behandling af vaskulær okklusion |
| US9314248B2 (en) | 2012-11-06 | 2016-04-19 | Covidien Lp | Multi-pivot thrombectomy device |
| CN108354645A (zh) | 2012-11-13 | 2018-08-03 | 柯惠有限合伙公司 | 封堵装置 |
| US8597323B1 (en) * | 2012-11-16 | 2013-12-03 | Sequent Medical, Inc. | Delivery and detachment systems and methods for vascular implants |
| US8784434B2 (en) | 2012-11-20 | 2014-07-22 | Inceptus Medical, Inc. | Methods and apparatus for treating embolism |
| CN103006286B (zh) * | 2012-12-18 | 2015-07-08 | 邹旭华 | 一种可显影封闭式编织体植入物及输送装置 |
| US10342546B2 (en) | 2013-01-14 | 2019-07-09 | Microvention, Inc. | Occlusive device |
| US9295571B2 (en) | 2013-01-17 | 2016-03-29 | Covidien Lp | Methods and apparatus for luminal stenting |
| WO2014134568A2 (en) | 2013-03-01 | 2014-09-04 | The Regents Of The University Of California | Apparatus and methods for bidirectional hyperelastic stent covers |
| US9463105B2 (en) | 2013-03-14 | 2016-10-11 | Covidien Lp | Methods and apparatus for luminal stenting |
| US9433429B2 (en) | 2013-03-14 | 2016-09-06 | Neuravi Limited | Clot retrieval devices |
| PL2967611T3 (pl) | 2013-03-14 | 2019-08-30 | Neuravi Limited | Urządzenie do usuwania ostrych blokad z naczyń krwionośnych |
| CN105208950A (zh) | 2013-03-14 | 2015-12-30 | 尼尔拉维有限公司 | 一种用于从血管去除堵塞凝块的凝块收取装置 |
| CN105142545B (zh) | 2013-03-15 | 2018-04-06 | 柯惠有限合伙公司 | 闭塞装置 |
| US8715315B1 (en) | 2013-03-15 | 2014-05-06 | Insera Therapeutics, Inc. | Vascular treatment systems |
| EP2967578A2 (en) * | 2013-03-15 | 2016-01-20 | Covidien LP | Occlusive implant structures |
| JP6437517B2 (ja) | 2013-03-15 | 2018-12-12 | インセラ セラピューティクス,インク. | 血管治療装置及び方法 |
| US8679150B1 (en) | 2013-03-15 | 2014-03-25 | Insera Therapeutics, Inc. | Shape-set textile structure based mechanical thrombectomy methods |
| US8715314B1 (en) | 2013-03-15 | 2014-05-06 | Insera Therapeutics, Inc. | Vascular treatment measurement methods |
| US9907684B2 (en) | 2013-05-08 | 2018-03-06 | Aneuclose Llc | Method of radially-asymmetric stent expansion |
| US9259237B2 (en) | 2013-07-12 | 2016-02-16 | Inceptus Medical, Llc | Methods and apparatus for treating pulmonary embolism |
| US9561103B2 (en) | 2013-07-17 | 2017-02-07 | Cephea Valve Technologies, Inc. | System and method for cardiac valve repair and replacement |
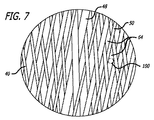
| US9078658B2 (en) | 2013-08-16 | 2015-07-14 | Sequent Medical, Inc. | Filamentary devices for treatment of vascular defects |
| US9955976B2 (en) | 2013-08-16 | 2018-05-01 | Sequent Medical, Inc. | Filamentary devices for treatment of vascular defects |
| WO2015061365A1 (en) | 2013-10-21 | 2015-04-30 | Inceptus Medical, Llc | Methods and apparatus for treating embolism |
| WO2015120155A1 (en) * | 2014-02-06 | 2015-08-13 | Boston Scientific Scimed, Inc. | Occlusion device detachable by inflation of a balloon |
| US11154302B2 (en) | 2014-03-31 | 2021-10-26 | DePuy Synthes Products, Inc. | Aneurysm occlusion device |
| US11076860B2 (en) | 2014-03-31 | 2021-08-03 | DePuy Synthes Products, Inc. | Aneurysm occlusion device |
| WO2015179377A1 (en) | 2014-05-20 | 2015-11-26 | Muffin Incorporated | Aneurysm stop pressure system |
| EP3094363A4 (en) | 2014-06-09 | 2017-10-04 | Inceptus Medical, LLC | Retraction and aspiration device for treating embolism and associated systems and methods |
| US10792043B2 (en) * | 2014-08-07 | 2020-10-06 | Perflow Medical Ltd. | Aneurysm treatment device and method |
| EP3171793B1 (en) * | 2014-08-14 | 2024-07-24 | Microvention, Inc. | Filamentary devices for treatment of vascular defects |
| US10617435B2 (en) | 2014-11-26 | 2020-04-14 | Neuravi Limited | Clot retrieval device for removing clot from a blood vessel |
| US11253278B2 (en) | 2014-11-26 | 2022-02-22 | Neuravi Limited | Clot retrieval system for removing occlusive clot from a blood vessel |
| WO2016083472A1 (en) | 2014-11-26 | 2016-06-02 | Neuravi Limited | A clot retrieval device for removing occlusive clot from a blood vessel |
| WO2016093877A1 (en) | 2014-12-09 | 2016-06-16 | Cephea Valve Technologies, Inc. | Replacement cardiac valves and methods of use and manufacture |
| CA2974532C (en) | 2015-01-23 | 2023-08-29 | Contego Medical Llc | Interventional device having an integrated embolic filter and associated methods |
| KR102586485B1 (ko) | 2015-02-25 | 2023-10-16 | 갤럭시 테라퓨틱스, 아이엔씨 | 동맥류 치료를 위한 시스템 및 방법 |
| US9375333B1 (en) | 2015-03-06 | 2016-06-28 | Covidien Lp | Implantable device detachment systems and associated devices and methods |
| EP3294220B1 (en) | 2015-05-14 | 2023-12-06 | Cephea Valve Technologies, Inc. | Cardiac valve delivery devices and systems |
| EP3294221B1 (en) | 2015-05-14 | 2024-03-06 | Cephea Valve Technologies, Inc. | Replacement mitral valves |
| WO2017035275A1 (en) * | 2015-08-24 | 2017-03-02 | Neurvana Medical Llc | Intrasacular occlusion devices methods processes and systems |
| US10478194B2 (en) | 2015-09-23 | 2019-11-19 | Covidien Lp | Occlusive devices |
| US9700332B2 (en) | 2015-10-23 | 2017-07-11 | Inari Medical, Inc. | Intravascular treatment of vascular occlusion and associated devices, systems, and methods |
| CN113796927B (zh) | 2015-10-23 | 2025-03-04 | 伊纳里医疗公司 | 脉管闭塞的脉管内治疗以及相关的装置、系统和方法 |
| US10342571B2 (en) | 2015-10-23 | 2019-07-09 | Inari Medical, Inc. | Intravascular treatment of vascular occlusion and associated devices, systems, and methods |
| ES2897755T3 (es) * | 2015-10-28 | 2022-03-02 | Jayandiran Pillai | Oclusor de aneurisma |
| WO2017072695A1 (en) | 2015-10-28 | 2017-05-04 | Jayandiran Pillai | Aneurysm occluder |
| CN109069790A (zh) | 2015-12-18 | 2018-12-21 | 伊纳里医疗公司 | 导管轴及相关装置、系统和方法 |
| CN108697423A (zh) | 2016-02-16 | 2018-10-23 | 伊瑟拉医疗公司 | 抽吸装置和锚定的分流装置 |
| US12232737B2 (en) | 2016-03-17 | 2025-02-25 | Eclipse Medical Limited | Occluding anatomical structures |
| JP6937327B2 (ja) * | 2016-03-17 | 2021-09-22 | スワミナサン ジャヤラマン | 解剖学的構造の閉塞 |
| WO2017214577A1 (en) * | 2016-06-10 | 2017-12-14 | Microvention, Inc. | Vessel occluder |
| EP3471665B1 (en) | 2016-06-17 | 2023-10-11 | Cephea Valve Technologies, Inc. | Cardiac valve delivery devices |
| US10478195B2 (en) | 2016-08-04 | 2019-11-19 | Covidien Lp | Devices, systems, and methods for the treatment of vascular defects |
| RU2019110141A (ru) | 2016-09-06 | 2020-10-08 | Ньюрави Лимитед | Устройство извлечения тромба для удаления окклюзионного тромба из кровеносного сосуда |
| US11877752B2 (en) | 2016-09-07 | 2024-01-23 | Daniel Ezra Walzman | Filterless aspiration, irrigating, macerating, rotating microcatheter and method of use |
| US11439492B2 (en) | 2016-09-07 | 2022-09-13 | Daniel Ezra Walzman | Lasso filter tipped microcatheter for simultaneous rotating separator, irrigator for thrombectomy and method for use |
| US12369932B2 (en) | 2016-09-07 | 2025-07-29 | Daniel Ezra Walzman | Methods and devices to ameliorate vascular obstruction |
| US12138149B2 (en) | 2016-09-07 | 2024-11-12 | Daniel Ezra Walzman | Endovascular devices and methods with filtering elements |
| US10314684B2 (en) | 2016-09-07 | 2019-06-11 | Daniel Ezra Walzman | Simultaneous rotating separator, irrigator microcatheter for thrombectomy |
| US11259820B2 (en) | 2016-09-07 | 2022-03-01 | Daniel Ezra Walzman | Methods and devices to ameliorate vascular obstruction |
| EP3913124A1 (en) | 2016-10-14 | 2021-11-24 | Inceptus Medical, LLC | Braiding machine and methods of use |
| US20180206851A1 (en) * | 2016-10-19 | 2018-07-26 | Daniel E. Walzman | Hydrogel intrasaccular occlusion device |
| US10576099B2 (en) | 2016-10-21 | 2020-03-03 | Covidien Lp | Injectable scaffold for treatment of intracranial aneurysms and related technology |
| WO2018080590A1 (en) | 2016-10-24 | 2018-05-03 | Inari Medical | Devices and methods for treating vascular occlusion |
| US12426885B2 (en) | 2016-12-05 | 2025-09-30 | Daniel Ezra Walzman | Mesh cap for ameliorating outpouchings |
| US20180206848A1 (en) | 2016-12-05 | 2018-07-26 | Daniel E. Walzman | Alternative Use for Hydrogel Intrasaccular Occlusion Device with Radial Bars for Structural Support |
| US10448970B2 (en) | 2016-12-05 | 2019-10-22 | Daniel E. Walzman | Alternative use for hydrogel intrasaccular occlusion device with telescoping central support element |
| US10548607B2 (en) | 2016-12-05 | 2020-02-04 | Daniel Ezra Walzman | Mesh caps |
| US10543015B2 (en) * | 2016-12-05 | 2020-01-28 | Daniel Ezra Walzman | Mesh disc for saccular aneurysms and cover for saccular out-pouching |
| CR20190381A (es) | 2017-01-23 | 2019-09-27 | Cephea Valve Tech Inc | Valvulas mitrales de reemplazo |
| AU2018203053B2 (en) | 2017-01-23 | 2020-03-05 | Cephea Valve Technologies, Inc. | Replacement mitral valves |
| AU2018224126A1 (en) | 2017-02-23 | 2019-09-12 | DePuy Synthes Products, Inc. | Aneurysm device and delivery system |
| CN110573092B (zh) | 2017-02-24 | 2023-04-18 | 因赛普特斯医学有限责任公司 | 血管阻塞装置和方法 |
| US10881413B2 (en) | 2017-03-24 | 2021-01-05 | Sequent Medical, Inc. | Systems and methods for embolization of body structures |
| EP4119066A1 (en) * | 2017-08-17 | 2023-01-18 | Arissa Medical, Inc. | Flow attenuation device |
| IL272716B2 (en) | 2017-08-21 | 2023-09-01 | Cerus Endovascular Ltd | Install a block |
| US10675036B2 (en) | 2017-08-22 | 2020-06-09 | Covidien Lp | Devices, systems, and methods for the treatment of vascular defects |
| PT3678731T (pt) | 2017-09-06 | 2024-12-10 | Inari Medical Inc | Válvulas de hemostase e métodos de utilização |
| EP3695037B1 (en) | 2017-10-14 | 2024-03-27 | Inceptus Medical, LLC | Braiding machine and methods of use |
| US11399845B2 (en) * | 2017-12-12 | 2022-08-02 | Penumbra, Inc. | Vascular cages and methods of making and using the same |
| US10716574B2 (en) | 2017-12-22 | 2020-07-21 | DePuy Synthes Products, Inc. | Aneurysm device and delivery method |
| US11185335B2 (en) | 2018-01-19 | 2021-11-30 | Galaxy Therapeutics Inc. | System for and method of treating aneurysms |
| US10905430B2 (en) | 2018-01-24 | 2021-02-02 | DePuy Synthes Products, Inc. | Aneurysm device and delivery system |
| US11154314B2 (en) | 2018-01-26 | 2021-10-26 | Inari Medical, Inc. | Single insertion delivery system for treating embolism and associated systems and methods |
| US11596412B2 (en) * | 2018-05-25 | 2023-03-07 | DePuy Synthes Products, Inc. | Aneurysm device and delivery system |
| US11058430B2 (en) * | 2018-05-25 | 2021-07-13 | DePuy Synthes Products, Inc. | Aneurysm device and delivery system |
| US11051825B2 (en) | 2018-08-08 | 2021-07-06 | DePuy Synthes Products, Inc. | Delivery system for embolic braid |
| CN112867455B (zh) | 2018-08-13 | 2025-07-29 | 伊纳里医疗有限公司 | 治疗栓塞的系统和相关的装置和方法 |
| US10842498B2 (en) | 2018-09-13 | 2020-11-24 | Neuravi Limited | Systems and methods of restoring perfusion to a vessel |
| US11123077B2 (en) * | 2018-09-25 | 2021-09-21 | DePuy Synthes Products, Inc. | Intrasaccular device positioning and deployment system |
| US11406416B2 (en) | 2018-10-02 | 2022-08-09 | Neuravi Limited | Joint assembly for vasculature obstruction capture device |
| US11076861B2 (en) * | 2018-10-12 | 2021-08-03 | DePuy Synthes Products, Inc. | Folded aneurysm treatment device and delivery method |
| JP7475020B2 (ja) | 2018-10-25 | 2024-04-26 | 株式会社東海メディカルプロダクツ | 医療用デバイス |
| US11406392B2 (en) | 2018-12-12 | 2022-08-09 | DePuy Synthes Products, Inc. | Aneurysm occluding device for use with coagulating agents |
| US11730485B2 (en) | 2018-12-17 | 2023-08-22 | Covidien Lp | Devices, systems, and methods for the treatment of vascular defects |
| US11272939B2 (en) | 2018-12-18 | 2022-03-15 | DePuy Synthes Products, Inc. | Intrasaccular flow diverter for treating cerebral aneurysms |
| WO2020139544A2 (en) * | 2018-12-27 | 2020-07-02 | Stryker Corporation | Shape adaptable multi-layered intra-saccular flow reduction device and methods of manufacturing same |
| US11134953B2 (en) | 2019-02-06 | 2021-10-05 | DePuy Synthes Products, Inc. | Adhesive cover occluding device for aneurysm treatment |
| EP3908209A4 (en) | 2019-03-15 | 2022-10-19 | Sequent Medical, Inc. | FIBROUS DEVICES FOR TREATMENT OF VASCULAR DEFECTS |
| US11337706B2 (en) | 2019-03-27 | 2022-05-24 | DePuy Synthes Products, Inc. | Aneurysm treatment device |
| US11602350B2 (en) | 2019-12-05 | 2023-03-14 | DePuy Synthes Products, Inc. | Intrasaccular inverting braid with highly flexible fill material |
| US11497504B2 (en) | 2019-05-21 | 2022-11-15 | DePuy Synthes Products, Inc. | Aneurysm treatment with pushable implanted braid |
| US11607226B2 (en) | 2019-05-21 | 2023-03-21 | DePuy Synthes Products, Inc. | Layered braided aneurysm treatment device with corrugations |
| US10653425B1 (en) | 2019-05-21 | 2020-05-19 | DePuy Synthes Products, Inc. | Layered braided aneurysm treatment device |
| US11413046B2 (en) | 2019-05-21 | 2022-08-16 | DePuy Synthes Products, Inc. | Layered braided aneurysm treatment device |
| US11672542B2 (en) | 2019-05-21 | 2023-06-13 | DePuy Synthes Products, Inc. | Aneurysm treatment with pushable ball segment |
| US11278292B2 (en) | 2019-05-21 | 2022-03-22 | DePuy Synthes Products, Inc. | Inverting braided aneurysm treatment system and method |
| US12446886B2 (en) | 2019-05-21 | 2025-10-21 | DePuy Synthes Products, Inc. | Semispherical braided aneurysm treatment system and method |
| US12102327B2 (en) | 2019-05-25 | 2024-10-01 | Galaxy Therapeutics, Inc. | Systems and methods for treating aneurysms |
| WO2021011033A1 (en) * | 2019-07-12 | 2021-01-21 | Sequent Medical, Inc. | Filamentary devices for treatment of vascular defects |
| EP3771437A1 (fr) * | 2019-07-29 | 2021-02-03 | Université de Montpellier | Dispositif intra-anévrismal |
| US11864779B2 (en) | 2019-10-16 | 2024-01-09 | Inari Medical, Inc. | Systems, devices, and methods for treating vascular occlusions |
| US11712231B2 (en) | 2019-10-29 | 2023-08-01 | Neuravi Limited | Proximal locking assembly design for dual stent mechanical thrombectomy device |
| EP4054440B1 (en) | 2019-11-04 | 2024-06-05 | Covidien LP | Manufacturing method of devices for treatment of intracranial aneurysms |
| US11517340B2 (en) | 2019-12-03 | 2022-12-06 | Neuravi Limited | Stentriever devices for removing an occlusive clot from a vessel and methods thereof |
| US11457926B2 (en) | 2019-12-18 | 2022-10-04 | DePuy Synthes Products, Inc. | Implant having an intrasaccular section and intravascular section |
| US12070220B2 (en) | 2020-03-11 | 2024-08-27 | Microvention, Inc. | Devices having multiple permeable shells for treatment of vascular defects |
| WO2021183793A2 (en) | 2020-03-11 | 2021-09-16 | Microvention, Inc. | Devices for treatment of vascular defects |
| US20210282789A1 (en) | 2020-03-11 | 2021-09-16 | Microvention, Inc. | Multiple layer devices for treatment of vascular defects |
| US11717308B2 (en) | 2020-04-17 | 2023-08-08 | Neuravi Limited | Clot retrieval device for removing heterogeneous clots from a blood vessel |
| US11730501B2 (en) | 2020-04-17 | 2023-08-22 | Neuravi Limited | Floating clot retrieval device for removing clots from a blood vessel |
| US11871946B2 (en) | 2020-04-17 | 2024-01-16 | Neuravi Limited | Clot retrieval device for removing clot from a blood vessel |
| US20210338247A1 (en) * | 2020-05-04 | 2021-11-04 | DePuy Synthes Products, Inc. | Double layer braid |
| US11931041B2 (en) | 2020-05-12 | 2024-03-19 | Covidien Lp | Devices, systems, and methods for the treatment of vascular defects |
| US11737771B2 (en) | 2020-06-18 | 2023-08-29 | Neuravi Limited | Dual channel thrombectomy device |
| US11937836B2 (en) | 2020-06-22 | 2024-03-26 | Neuravi Limited | Clot retrieval system with expandable clot engaging framework |
| US11395669B2 (en) | 2020-06-23 | 2022-07-26 | Neuravi Limited | Clot retrieval device with flexible collapsible frame |
| US11439418B2 (en) | 2020-06-23 | 2022-09-13 | Neuravi Limited | Clot retrieval device for removing clot from a blood vessel |
| AU2021296039A1 (en) * | 2020-06-25 | 2023-02-09 | Bae Systems Plc | Structural simulation of additively manufactured components |
| US12161344B2 (en) * | 2020-07-31 | 2024-12-10 | Covidien Lp | Expandable devices for treating body lumens |
| CN114098880B (zh) * | 2020-08-31 | 2025-08-01 | 微创神通医疗科技(上海)有限公司 | 血管瘤封堵装置、血管瘤封堵治疗装置及血管瘤封堵系统 |
| US12127743B2 (en) | 2020-09-23 | 2024-10-29 | DePuy Synthes Products, Inc. | Inverting braided aneurysm implant with dome feature |
| US11864781B2 (en) | 2020-09-23 | 2024-01-09 | Neuravi Limited | Rotating frame thrombectomy device |
| US20220192678A1 (en) * | 2020-12-18 | 2022-06-23 | Microvention, Inc. | Filamentary devices for treatment of vascular defects |
| US11937837B2 (en) | 2020-12-29 | 2024-03-26 | Neuravi Limited | Fibrin rich / soft clot mechanical thrombectomy device |
| CN112690855B (zh) * | 2020-12-31 | 2023-02-03 | 先健科技(深圳)有限公司 | 封堵器及封堵装置 |
| JP2024502376A (ja) * | 2021-01-10 | 2024-01-18 | ルシード ヴァスキュラ リミテッド | 血管内デバイス |
| US12029442B2 (en) | 2021-01-14 | 2024-07-09 | Neuravi Limited | Systems and methods for a dual elongated member clot retrieval apparatus |
| WO2022164957A1 (en) | 2021-01-27 | 2022-08-04 | Galaxy Therapeutics, Inc. | Systems and methods for treating aneurysms |
| US12064130B2 (en) | 2021-03-18 | 2024-08-20 | Neuravi Limited | Vascular obstruction retrieval device having sliding cages pinch mechanism |
| CN113180781B (zh) * | 2021-06-01 | 2025-03-21 | 上海融脉医疗科技有限公司 | 一种卷绕式网盘支架及其输送装置 |
| US11974764B2 (en) | 2021-06-04 | 2024-05-07 | Neuravi Limited | Self-orienting rotating stentriever pinching cells |
| CN113491556B (zh) * | 2021-06-25 | 2022-07-26 | 四川艾迈思生物医疗科技股份有限公司 | 一种栓塞微导管组件 |
| US12446891B2 (en) | 2021-08-30 | 2025-10-21 | Microvention, Inc. | Devices for treatment of vascular defects |
| AU2023208030A1 (en) | 2022-01-11 | 2024-07-18 | Inari Medical, Inc. | Devices for removing clot material from intravascularly implanted devices, and associated systems and methods |
| CN114246631A (zh) * | 2022-02-28 | 2022-03-29 | 上海介入医疗器械有限公司 | 封堵器及封堵组件 |
| US12408923B2 (en) | 2022-03-02 | 2025-09-09 | Covidien Lp | Occlusive devices with thrombogenic inserts |
| KR102772752B1 (ko) * | 2022-04-12 | 2025-02-26 | 한스바이오메드 주식회사 | 다층 구조 및 복합 소재를 포함하는 혈류 제한 장치 |
| US20230404590A1 (en) * | 2022-06-15 | 2023-12-21 | DePuy Synthes Products, Inc. | Inverting braided aneurysm treatment system having a semi-frustoconically-shaped portion |
| WO2025235015A1 (en) | 2024-05-10 | 2025-11-13 | Inari Medical, Inc. | Mechanical thrombectomy assemblies with relief features, and associated devices, systems, and methods |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008126060A (ja) * | 2006-11-20 | 2008-06-05 | Stout Medical Group Lp | 機械的組織デバイスおよび方法 |
| JP2009000497A (ja) * | 2007-06-21 | 2009-01-08 | Aga Medical Corp | 血管障害部位を閉塞するための多層編組構造 |
Family Cites Families (228)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3874388A (en) | 1973-02-12 | 1975-04-01 | Ochsner Med Found Alton | Shunt defect closure system |
| JPS52141092A (en) | 1976-05-20 | 1977-11-25 | Nippon Zeon Co | Blood bessel catheter |
| US4282875A (en) | 1979-01-24 | 1981-08-11 | Serbinenko Fedor A | Occlusive device |
| US4346712A (en) | 1979-04-06 | 1982-08-31 | Kuraray Company, Ltd. | Releasable balloon catheter |
| US4675361A (en) | 1980-02-29 | 1987-06-23 | Thoratec Laboratories Corp. | Polymer systems suitable for blood-contacting surfaces of a biomedical device, and methods for forming |
| DK151404C (da) | 1984-05-23 | 1988-07-18 | Cook Europ Aps William | Sammenklappeligt filter til implantation i en patients blodkar |
| SE453258B (sv) | 1986-04-21 | 1988-01-25 | Medinvent Sa | Elastisk, sjelvexpanderande protes samt forfarande for dess framstellning |
| US4729278A (en) * | 1987-05-04 | 1988-03-08 | Rockwell International Corporation | Vertical rotary braider |
| US5165421A (en) | 1987-09-30 | 1992-11-24 | Lake Region Manufacturing Co., Inc. | Hollow lumen cable apparatus |
| US5154705A (en) | 1987-09-30 | 1992-10-13 | Lake Region Manufacturing Co., Inc. | Hollow lumen cable apparatus |
| FR2624747A1 (fr) * | 1987-12-18 | 1989-06-23 | Delsanti Gerard | Dispositifs endo-arteriels amovibles destines a reparer des decollements de parois des arteres |
| US5263963A (en) | 1989-09-08 | 1993-11-23 | Advanced Cardiovascular Systems, Inc. | Expandable cage catheter for repairing a damaged blood vessel |
| US5122136A (en) | 1990-03-13 | 1992-06-16 | The Regents Of The University Of California | Endovascular electrolytically detachable guidewire tip for the electroformation of thrombus in arteries, veins, aneurysms, vascular malformations and arteriovenous fistulas |
| US5569245A (en) | 1990-03-13 | 1996-10-29 | The Regents Of The University Of California | Detachable endovascular occlusion device activated by alternating electric current |
| US5221261A (en) * | 1990-04-12 | 1993-06-22 | Schneider (Usa) Inc. | Radially expandable fixation member |
| US5071407A (en) | 1990-04-12 | 1991-12-10 | Schneider (U.S.A.) Inc. | Radially expandable fixation member |
| US5108407A (en) * | 1990-06-08 | 1992-04-28 | Rush-Presbyterian St. Luke's Medical Center | Method and apparatus for placement of an embolic coil |
| JPH0447415A (ja) | 1990-06-14 | 1992-02-17 | Amada Co Ltd | ワーク搬送ロボットにおけるリニアモータの制御方法及びその装置 |
| US5158545A (en) | 1991-05-02 | 1992-10-27 | Brigham And Women's Hospital | Diameter expansion cannula |
| JP2961287B2 (ja) * | 1991-10-18 | 1999-10-12 | グンゼ株式会社 | 生体管路拡張具、その製造方法およびステント |
| WO1993022986A1 (en) * | 1992-05-08 | 1993-11-25 | Schneider (Usa) Inc. | Esophageal stent and delivery tool |
| US5382259A (en) * | 1992-10-26 | 1995-01-17 | Target Therapeutics, Inc. | Vasoocclusion coil with attached tubular woven or braided fibrous covering |
| US5334210A (en) | 1993-04-09 | 1994-08-02 | Cook Incorporated | Vascular occlusion assembly |
| US5536247A (en) | 1993-06-10 | 1996-07-16 | Scimed Life Systems, Inc. | Method of treating cardiac conduction defects |
| RU2089131C1 (ru) | 1993-12-28 | 1997-09-10 | Сергей Апполонович Пульнев | Стент |
| JP2880070B2 (ja) | 1994-03-31 | 1999-04-05 | 株式会社カネカメディックス | 生体内留置部材を有する医療用ワイヤー |
| EP1221307B1 (en) * | 1994-07-08 | 2010-02-17 | ev3 Inc. | System for performing an intravascular procedure |
| US5846261A (en) | 1994-07-08 | 1998-12-08 | Aga Medical Corp. | Percutaneous catheter directed occlusion devices |
| US5725552A (en) * | 1994-07-08 | 1998-03-10 | Aga Medical Corporation | Percutaneous catheter directed intravascular occlusion devices |
| US6123715A (en) | 1994-07-08 | 2000-09-26 | Amplatz; Curtis | Method of forming medical devices; intravascular occlusion devices |
| US5556687A (en) | 1994-10-14 | 1996-09-17 | Acromed Corporation | Composite structure suitable for use as a bone plate and method for making said structure |
| US5601595A (en) * | 1994-10-25 | 1997-02-11 | Scimed Life Systems, Inc. | Remobable thrombus filter |
| US5578074A (en) | 1994-12-22 | 1996-11-26 | Target Therapeutics, Inc. | Implant delivery method and assembly |
| NO962336L (no) * | 1995-06-06 | 1996-12-09 | Target Therapeutics Inc | Vaso-okklusiv spiral |
| WO1997001368A1 (en) | 1995-06-26 | 1997-01-16 | Trimedyne, Inc. | Therapeutic appliance releasing device |
| US5749883A (en) * | 1995-08-30 | 1998-05-12 | Halpern; David Marcos | Medical instrument |
| US6168622B1 (en) * | 1996-01-24 | 2001-01-02 | Microvena Corporation | Method and apparatus for occluding aneurysms |
| JP2001502605A (ja) | 1996-01-30 | 2001-02-27 | メドトロニック,インコーポレーテッド | ステントを作るための物品および方法 |
| US5733294A (en) * | 1996-02-28 | 1998-03-31 | B. Braun Medical, Inc. | Self expanding cardiovascular occlusion device, method of using and method of making the same |
| US6592617B2 (en) * | 1996-04-30 | 2003-07-15 | Boston Scientific Scimed, Inc. | Three-dimensional braided covered stent |
| US5718159A (en) * | 1996-04-30 | 1998-02-17 | Schneider (Usa) Inc. | Process for manufacturing three-dimensional braided covered stent |
| US5927345A (en) | 1996-04-30 | 1999-07-27 | Target Therapeutics, Inc. | Super-elastic alloy braid structure |
| US6949116B2 (en) | 1996-05-08 | 2005-09-27 | Carag Ag | Device for plugging an opening such as in a wall of a hollow or tubular organ including biodegradable elements |
| US6190402B1 (en) * | 1996-06-21 | 2001-02-20 | Musc Foundation For Research Development | Insitu formable and self-forming intravascular flow modifier (IFM) and IFM assembly for deployment of same |
| US5964797A (en) | 1996-08-30 | 1999-10-12 | Target Therapeutics, Inc. | Electrolytically deployable braided vaso-occlusion device |
| US5980554A (en) | 1997-05-05 | 1999-11-09 | Micro Therapeutics, Inc. | Wire frame partial flow obstruction for aneurysm treatment |
| US5951599A (en) * | 1997-07-09 | 1999-09-14 | Scimed Life Systems, Inc. | Occlusion system for endovascular treatment of an aneurysm |
| US5928260A (en) | 1997-07-10 | 1999-07-27 | Scimed Life Systems, Inc. | Removable occlusion system for aneurysm neck |
| US7569066B2 (en) | 1997-07-10 | 2009-08-04 | Boston Scientific Scimed, Inc. | Methods and devices for the treatment of aneurysms |
| US5944733A (en) | 1997-07-14 | 1999-08-31 | Target Therapeutics, Inc. | Controlled detachable vasoocclusive member using mechanical junction and friction-enhancing member |
| GB9715241D0 (en) | 1997-07-18 | 1997-09-24 | Jeffree Martin A | Device for treating aneurysms |
| WO1999005977A1 (en) | 1997-08-04 | 1999-02-11 | Boston Scientific Corporation | Occlusion system for aneurysm repair |
| US6063070A (en) * | 1997-08-05 | 2000-05-16 | Target Therapeutics, Inc. | Detachable aneurysm neck bridge (II) |
| EP1003422B1 (en) | 1997-08-05 | 2006-06-14 | Boston Scientific Limited | Detachable aneurysm neck bridge |
| US5916235A (en) | 1997-08-13 | 1999-06-29 | The Regents Of The University Of California | Apparatus and method for the use of detachable coils in vascular aneurysms and body cavities |
| US6086577A (en) | 1997-08-13 | 2000-07-11 | Scimed Life Systems, Inc. | Detachable aneurysm neck bridge (III) |
| US5984929A (en) | 1997-08-29 | 1999-11-16 | Target Therapeutics, Inc. | Fast detaching electronically isolated implant |
| US6156061A (en) | 1997-08-29 | 2000-12-05 | Target Therapeutics, Inc. | Fast-detaching electrically insulated implant |
| US6066149A (en) | 1997-09-30 | 2000-05-23 | Target Therapeutics, Inc. | Mechanical clot treatment device with distal filter |
| US6511468B1 (en) * | 1997-10-17 | 2003-01-28 | Micro Therapeutics, Inc. | Device and method for controlling injection of liquid embolic composition |
| US6159206A (en) | 1997-10-30 | 2000-12-12 | Kaneka Medix Corporation | Medical implement for depositing implanted device and method of depositing implanted device |
| US6461370B1 (en) | 1998-11-03 | 2002-10-08 | C. R. Bard, Inc. | Temporary vascular filter guide wire |
| US20040260333A1 (en) * | 1997-11-12 | 2004-12-23 | Dubrul William R. | Medical device and method |
| US5873907A (en) * | 1998-01-27 | 1999-02-23 | Endotex Interventional Systems, Inc. | Electrolytic stent delivery system and methods of use |
| US5944738A (en) | 1998-02-06 | 1999-08-31 | Aga Medical Corporation | Percutaneous catheter directed constricting occlusion device |
| JP2003522550A (ja) * | 1998-02-10 | 2003-07-29 | アーテミス・メディカル・インコーポレイテッド | 咬合、固定、テンショニング、及び流向装置とその使用方法 |
| US5925060A (en) | 1998-03-13 | 1999-07-20 | B. Braun Celsa | Covered self-expanding vascular occlusion device |
| US6168615B1 (en) * | 1998-05-04 | 2001-01-02 | Micrus Corporation | Method and apparatus for occlusion and reinforcement of aneurysms |
| US6463317B1 (en) | 1998-05-19 | 2002-10-08 | Regents Of The University Of Minnesota | Device and method for the endovascular treatment of aneurysms |
| JP4741728B2 (ja) | 1998-06-04 | 2011-08-10 | ニューヨーク・ユニバーシティ | 血管内薄膜デバイスおよびストロークの予防治療法 |
| US6139564A (en) | 1998-06-16 | 2000-10-31 | Target Therapeutics Inc. | Minimally occlusive flow disruptor stent for bridging aneurysm necks |
| US5935148A (en) | 1998-06-24 | 1999-08-10 | Target Therapeutics, Inc. | Detachable, varying flexibility, aneurysm neck bridge |
| US6096175A (en) * | 1998-07-17 | 2000-08-01 | Micro Therapeutics, Inc. | Thin film stent |
| US7004962B2 (en) * | 1998-07-27 | 2006-02-28 | Schneider (Usa), Inc. | Neuroaneurysm occlusion and delivery device and method of using same |
| US6093199A (en) | 1998-08-05 | 2000-07-25 | Endovascular Technologies, Inc. | Intra-luminal device for treatment of body cavities and lumens and method of use |
| US6315709B1 (en) | 1998-08-07 | 2001-11-13 | Stereotaxis, Inc. | Magnetic vascular defect treatment system |
| US6478773B1 (en) | 1998-12-21 | 2002-11-12 | Micrus Corporation | Apparatus for deployment of micro-coil using a catheter |
| US6500149B2 (en) | 1998-08-31 | 2002-12-31 | Deepak Gandhi | Apparatus for deployment of micro-coil using a catheter |
| EP1109499B1 (en) | 1998-09-04 | 2007-08-15 | Boston Scientific Limited | Detachable aneurysm neck closure patch |
| US7410482B2 (en) | 1998-09-04 | 2008-08-12 | Boston Scientific-Scimed, Inc. | Detachable aneurysm neck bridge |
| US6277126B1 (en) * | 1998-10-05 | 2001-08-21 | Cordis Neurovascular Inc. | Heated vascular occlusion coil development system |
| US7044134B2 (en) | 1999-11-08 | 2006-05-16 | Ev3 Sunnyvale, Inc | Method of implanting a device in the left atrial appendage |
| US6152144A (en) | 1998-11-06 | 2000-11-28 | Appriva Medical, Inc. | Method and device for left atrial appendage occlusion |
| US6368338B1 (en) * | 1999-03-05 | 2002-04-09 | Board Of Regents, The University Of Texas | Occlusion method and apparatus |
| US6613074B1 (en) | 1999-03-10 | 2003-09-02 | Cordis Corporation | Endovascular aneurysm embolization device |
| US6428558B1 (en) | 1999-03-10 | 2002-08-06 | Cordis Corporation | Aneurysm embolization device |
| US6203779B1 (en) * | 1999-03-19 | 2001-03-20 | Charlie Ricci | Methods for treating endoleaks during endovascular repair of abdominal aortic aneurysms |
| US6375668B1 (en) * | 1999-06-02 | 2002-04-23 | Hanson S. Gifford | Devices and methods for treating vascular malformations |
| US20020169473A1 (en) * | 1999-06-02 | 2002-11-14 | Concentric Medical, Inc. | Devices and methods for treating vascular malformations |
| US7052711B2 (en) * | 1999-09-02 | 2006-05-30 | Rice University | Nitric oxide-producing hydrogel materials |
| US6325815B1 (en) | 1999-09-21 | 2001-12-04 | Microvena Corporation | Temporary vascular filter |
| US6375670B1 (en) * | 1999-10-07 | 2002-04-23 | Prodesco, Inc. | Intraluminal filter |
| US6689150B1 (en) | 1999-10-27 | 2004-02-10 | Atritech, Inc. | Filter apparatus for ostium of left atrial appendage |
| US6551303B1 (en) * | 1999-10-27 | 2003-04-22 | Atritech, Inc. | Barrier device for ostium of left atrial appendage |
| US6994092B2 (en) * | 1999-11-08 | 2006-02-07 | Ev3 Sunnyvale, Inc. | Device for containing embolic material in the LAA having a plurality of tissue retention structures |
| US6790218B2 (en) | 1999-12-23 | 2004-09-14 | Swaminathan Jayaraman | Occlusive coil manufacture and delivery |
| US6350270B1 (en) * | 2000-01-24 | 2002-02-26 | Scimed Life Systems, Inc. | Aneurysm liner |
| US7740637B2 (en) | 2000-02-09 | 2010-06-22 | Micrus Endovascular Corporation | Apparatus and method for deployment of a therapeutic device using a catheter |
| US20020043456A1 (en) * | 2000-02-29 | 2002-04-18 | Ho Ken K. | Bimorphic, compositionally-graded, sputter-deposited, thin film shape memory device |
| US6346117B1 (en) * | 2000-03-02 | 2002-02-12 | Prodesco, Inc. | Bag for use in the intravascular treatment of saccular aneurysms |
| US6391037B1 (en) * | 2000-03-02 | 2002-05-21 | Prodesco, Inc. | Bag for use in the intravascular treatment of saccular aneurysms |
| US6632241B1 (en) | 2000-03-22 | 2003-10-14 | Endovascular Technologies, Inc. | Self-expanding, pseudo-braided intravascular device |
| US6719778B1 (en) * | 2000-03-24 | 2004-04-13 | Endovascular Technologies, Inc. | Methods for treatment of aneurysms |
| US6844153B2 (en) * | 2000-03-27 | 2005-01-18 | Thomas Jefferson University | Compositions and methods for identifying and targeting cancer cells of alimentary canal origin |
| US20010031981A1 (en) | 2000-03-31 | 2001-10-18 | Evans Michael A. | Method and device for locating guidewire and treating chronic total occlusions |
| AU2001275376A1 (en) | 2000-06-08 | 2001-12-17 | John J Frantzen | Radially expandable stent featuring covering tip primarily for bifurcated arteryaneurysms |
| WO2002000139A1 (en) | 2000-06-23 | 2002-01-03 | Frantzen John J | Radially expandable aneurysm treatment stent |
| ES2341641T3 (es) | 2000-07-21 | 2010-06-24 | The Spineology Group, Llc | Un dispositivo de bolsa de malla porosa expansible y su uso para cirugia osea. |
| US6855154B2 (en) * | 2000-08-11 | 2005-02-15 | University Of Louisville Research Foundation, Inc. | Endovascular aneurysm treatment device and method |
| DE60115712T2 (de) * | 2000-08-23 | 2006-09-07 | Thoratec Corp., Pleasanton | Beschichtete gefässtransplantate und verwendungsverfahren |
| AU9109201A (en) | 2000-09-18 | 2002-03-26 | Organogenesis Inc | Methods for treating a patient using a bioengineered flat sheet graft prostheses |
| US6730119B1 (en) * | 2000-10-06 | 2004-05-04 | Board Of Regents Of The University Of Texas System | Percutaneous implantation of partially covered stents in aneurysmally dilated arterial segments with subsequent embolization and obliteration of the aneurysm cavity |
| US20060135947A1 (en) | 2000-10-27 | 2006-06-22 | Pulmonx | Occlusal stent and methods for its use |
| US6589265B1 (en) | 2000-10-31 | 2003-07-08 | Endovascular Technologies, Inc. | Intrasaccular embolic device |
| US6743251B1 (en) | 2000-11-15 | 2004-06-01 | Scimed Life Systems, Inc. | Implantable devices with polymeric detachment junction |
| US6878384B2 (en) * | 2001-03-13 | 2005-04-12 | Microvention, Inc. | Hydrogels that undergo volumetric expansion in response to changes in their environment and their methods of manufacture and use |
| US7044958B2 (en) * | 2001-04-03 | 2006-05-16 | Medtronic Vascular, Inc. | Temporary device for capturing embolic material |
| GB2374885B (en) * | 2001-04-27 | 2003-05-14 | Smith International | Method for hardfacing roller cone drill bit legs using a D-gun hardfacing application technique |
| US6855153B2 (en) * | 2001-05-01 | 2005-02-15 | Vahid Saadat | Embolic balloon |
| US6607539B1 (en) | 2001-05-18 | 2003-08-19 | Endovascular Technologies, Inc. | Electric endovascular implant depolyment system |
| EP2319430B1 (en) * | 2001-05-29 | 2013-11-13 | Microvention, Inc. | Vascular embolization device and method of manufacture |
| US20020187288A1 (en) | 2001-06-11 | 2002-12-12 | Advanced Cardiovascular Systems, Inc. | Medical device formed of silicone-polyurethane |
| US6454780B1 (en) | 2001-06-21 | 2002-09-24 | Scimed Life Systems, Inc. | Aneurysm neck obstruction device |
| US20030100945A1 (en) | 2001-11-23 | 2003-05-29 | Mindguard Ltd. | Implantable intraluminal device and method of using same in treating aneurysms |
| US7011671B2 (en) * | 2001-07-18 | 2006-03-14 | Atritech, Inc. | Cardiac implant device tether system and method |
| US8252040B2 (en) | 2001-07-20 | 2012-08-28 | Microvention, Inc. | Aneurysm treatment device and method of use |
| US20030028209A1 (en) * | 2001-07-31 | 2003-02-06 | Clifford Teoh | Expandable body cavity liner device |
| US6811560B2 (en) * | 2001-09-20 | 2004-11-02 | Cordis Neurovascular, Inc. | Stent aneurysm embolization method and device |
| US20050021075A1 (en) * | 2002-12-30 | 2005-01-27 | Bonnette Michael J. | Guidewire having deployable sheathless protective filter |
| JP2003190175A (ja) * | 2001-11-15 | 2003-07-08 | Cordis Neurovascular Inc | 動脈瘤を密閉するための動脈瘤頚部プラグ |
| JP4429589B2 (ja) | 2001-11-15 | 2010-03-10 | コーディス・ニューロバスキュラー・インコーポレイテッド | 閉塞部材を用いる動脈瘤塞栓装置 |
| US20060292206A1 (en) | 2001-11-26 | 2006-12-28 | Kim Steven W | Devices and methods for treatment of vascular aneurysms |
| US20040111147A1 (en) | 2002-12-03 | 2004-06-10 | Rabkin Dmitry J. | Temporary, repositionable or retrievable intraluminal devices |
| US6752826B2 (en) | 2001-12-14 | 2004-06-22 | Thoratec Corporation | Layered stent-graft and methods of making the same |
| US6953473B2 (en) | 2001-12-20 | 2005-10-11 | Boston Scientific Scimed, Inc. | Detachable device with electrically responsive element |
| FR2834199B1 (fr) | 2001-12-27 | 2004-10-15 | Doron Carmi | Endoprothese adaptee au milieu endoluminal |
| JP4328209B2 (ja) | 2002-01-25 | 2009-09-09 | アトリテック, インコーポレイテッド | 心耳血液ろ過システム |
| US6638257B2 (en) | 2002-03-01 | 2003-10-28 | Aga Medical Corporation | Intravascular flow restrictor |
| US7695488B2 (en) * | 2002-03-27 | 2010-04-13 | Boston Scientific Scimed, Inc. | Expandable body cavity liner device |
| US20050119684A1 (en) | 2002-07-12 | 2005-06-02 | Guterman Lee R. | Aneurysm buttress arrangement |
| US6746890B2 (en) | 2002-07-17 | 2004-06-08 | Tini Alloy Company | Three dimensional thin film devices and methods of fabrication |
| US6997904B2 (en) | 2002-12-24 | 2006-02-14 | Robert David Sculati | Viscous fluid injection system |
| US7229454B2 (en) | 2003-01-07 | 2007-06-12 | Boston Scientific Scimed, Inc. | Occlusive cinching devices and methods of use |
| ATE504241T1 (de) | 2003-01-10 | 2011-04-15 | Kaneka Corp | Embolus-bildendes in-vivo-verweilimplantat |
| US8377035B2 (en) * | 2003-01-17 | 2013-02-19 | Boston Scientific Scimed, Inc. | Unbalanced reinforcement members for medical device |
| US20040158311A1 (en) | 2003-02-06 | 2004-08-12 | Berhow Steven W. | Intravascular stent |
| US20040193208A1 (en) | 2003-03-27 | 2004-09-30 | Scimed Life Systems, Inc. | Radiopaque embolic protection filter membrane |
| US20040204737A1 (en) | 2003-04-11 | 2004-10-14 | Scimed Life Systems, Inc. | Embolic filter loop fabricated from composite material |
| US7597704B2 (en) | 2003-04-28 | 2009-10-06 | Atritech, Inc. | Left atrial appendage occlusion device with active expansion |
| MXPA05011896A (es) * | 2003-05-05 | 2006-05-25 | Univ Ben Gurion | Preparaciones polimericas reticuladas inyectables y sus usos. |
| CN1852688A (zh) * | 2003-05-19 | 2006-10-25 | 斯托特药物集团公司 | 组织扩张装置及用于治疗介入的相关方法 |
| US7101390B2 (en) * | 2003-05-27 | 2006-09-05 | Scimed Life Systems, Inc. | Staged deployment endograft |
| US20050053782A1 (en) * | 2003-09-04 | 2005-03-10 | Ayusman Sen | Process for forming polymeric micro and nanofibers |
| US6940209B2 (en) | 2003-09-08 | 2005-09-06 | New Scale Technologies | Ultrasonic lead screw motor |
| WO2005025630A1 (en) * | 2003-09-10 | 2005-03-24 | Cato T Laurencin | Polymeric nanofibers for tissue engineering and drug delivery |
| US7232461B2 (en) * | 2003-10-29 | 2007-06-19 | Cordis Neurovascular, Inc. | Neck covering device for an aneurysm |
| EP1689457A2 (en) * | 2003-11-10 | 2006-08-16 | Angiotech International Ag | Intravascular devices and fibrosis-inducing agents |
| US20050113868A1 (en) * | 2003-11-20 | 2005-05-26 | Devellian Carol A. | Device, with electrospun fabric, for a percutaneous transluminal procedure, and methods thereof |
| US20050133046A1 (en) | 2003-12-17 | 2005-06-23 | Becker Timothy A. | Compositions and methods for improved occlusion of vascular defects |
| US7329279B2 (en) * | 2003-12-23 | 2008-02-12 | Sadra Medical, Inc. | Methods and apparatus for endovascularly replacing a patient's heart valve |
| US7275471B2 (en) | 2003-12-29 | 2007-10-02 | Surpass Medical Ltd. | Mixed wire braided device with structural integrity |
| US20110208233A1 (en) | 2004-01-22 | 2011-08-25 | Mcguckin Jr James F | Device for preventing clot migration from left atrial appendage |
| CN1937976B (zh) * | 2004-02-02 | 2011-12-07 | 孕体有限公司 | 一种用于避孕的增强组织向内生长的装置 |
| US8398670B2 (en) | 2004-03-19 | 2013-03-19 | Aga Medical Corporation | Multi-layer braided structures for occluding vascular defects and for occluding fluid flow through portions of the vasculature of the body |
| US9039724B2 (en) | 2004-03-19 | 2015-05-26 | Aga Medical Corporation | Device for occluding vascular defects |
| US8313505B2 (en) * | 2004-03-19 | 2012-11-20 | Aga Medical Corporation | Device for occluding vascular defects |
| US20050228434A1 (en) | 2004-03-19 | 2005-10-13 | Aga Medical Corporation | Multi-layer braided structures for occluding vascular defects |
| US8715340B2 (en) | 2004-03-31 | 2014-05-06 | Merlin Md Pte Ltd. | Endovascular device with membrane |
| CA2549323C (en) * | 2004-04-08 | 2009-07-21 | Aga Medical Corporation | Flanged occlusion devices and methods |
| US8628564B2 (en) | 2004-05-25 | 2014-01-14 | Covidien Lp | Methods and apparatus for luminal stenting |
| US20050267516A1 (en) | 2004-06-01 | 2005-12-01 | Farzad Soleimani | Embolic protection device for the prevention of stroke |
| BE1016067A3 (fr) | 2004-06-03 | 2006-02-07 | Frid Noureddine | Endoprothese luminale pour occlusion d'anevrisme et procede de fabrication d'une telle endoprothese. |
| CN101415459B (zh) * | 2004-08-25 | 2014-11-05 | 麦克罗文逊股份有限公司 | 可植入物装置的热脱离系统 |
| EP1827250B1 (en) | 2004-08-31 | 2018-05-16 | Cook Medical Technologies LLC | Device for treating an aneurysm |
| US8444668B2 (en) | 2004-09-17 | 2013-05-21 | DePuy Synthes Products, LLC | Expandable vascular occlusion device |
| EP1804719A2 (en) * | 2004-09-22 | 2007-07-11 | Lee R. Guterman | Cranial aneurysm treatment arrangement |
| US7201918B2 (en) * | 2004-11-16 | 2007-04-10 | Microvention, Inc. | Compositions, systems and methods for treatment of defects in blood vessels |
| US20060155323A1 (en) * | 2005-01-07 | 2006-07-13 | Porter Stephen C | Intra-aneurysm devices |
| WO2006096449A2 (en) | 2005-03-03 | 2006-09-14 | Hines Richard A | Endovascular aneurysm treatment device and delivery system |
| DE102005013547B4 (de) | 2005-03-23 | 2009-02-05 | Admedes Schuessler Gmbh | Aneurysmen-Stent und Verfahren zu seiner Herstellung |
| US9327010B2 (en) | 2005-04-25 | 2016-05-03 | Massachusetts Institute Of Technology | Compositions and methods for promoting hemostasis and other physiological activities |
| DE102006020687A1 (de) * | 2005-07-19 | 2007-02-08 | Aesculap Ag & Co. Kg | Stentgraft-Prothese |
| WO2007013977A2 (en) * | 2005-07-21 | 2007-02-01 | The Research Foundation Of State University Of New York | Stent vascular intervention device and methods for treating aneurysms |
| US20070061006A1 (en) * | 2005-09-14 | 2007-03-15 | Nathan Desatnik | Methods of making shape memory films by chemical vapor deposition and shape memory devices made thereby |
| US8545530B2 (en) * | 2005-10-19 | 2013-10-01 | Pulsar Vascular, Inc. | Implantable aneurysm closure systems and methods |
| US20070100414A1 (en) * | 2005-11-02 | 2007-05-03 | Cardiomind, Inc. | Indirect-release electrolytic implant delivery systems |
| DE102005052628B4 (de) | 2005-11-04 | 2014-06-05 | Jenavalve Technology Inc. | Selbstexpandierendes, flexibles Drahtgeflecht mit integrierter Klappenprothese für den transvaskulären Herzklappenersatz und ein System mit einer solchen Vorrichtung und einem Einführkatheter |
| US8100938B2 (en) * | 2005-11-14 | 2012-01-24 | Occlutech Holding Ag | Occlusion device for occluding an atrial auricula and method for producing same |
| US8455088B2 (en) | 2005-12-23 | 2013-06-04 | Boston Scientific Scimed, Inc. | Spun nanofiber, medical devices, and methods |
| US7744652B2 (en) | 2006-01-23 | 2010-06-29 | Hesham Morsi | Aneurysm sealing device |
| GB0603685D0 (en) | 2006-02-23 | 2006-04-05 | Angiomed Ag | Vascular prosthesis for aneurysms, set of vascular prostheses, method for manufacturing a vascular prosthesis and method for inserting a vascular prosthesis |
| DE102006013770A1 (de) | 2006-03-24 | 2007-09-27 | Occlutech Gmbh | Occlusionsinstrument und Verfahren zu dessen Herstellung |
| WO2007134266A2 (en) | 2006-05-12 | 2007-11-22 | Electroformed Stents, Inc. | Exclusion device and system for delivery |
| US20080033341A1 (en) * | 2006-08-04 | 2008-02-07 | Bay Holdings Ltd. | Methods and devices for reducing or blocking blood flow to a selected blood vessel or part thereof |
| US20080221600A1 (en) | 2006-08-17 | 2008-09-11 | Dieck Martin S | Isolation devices for the treatment of aneurysms |
| GB0616738D0 (en) * | 2006-08-23 | 2006-10-04 | Evexar Medical Ltd | Improvements in and relating to medical devices |
| EP1982655B2 (de) | 2007-04-16 | 2022-12-07 | Occlutech Holding AG | Occluder zum Verschliessen eines Herzohres und Herstellungsverfahren dafür |
| US8574258B2 (en) * | 2007-02-16 | 2013-11-05 | Reverse Medical Corporation | Occlusion device and method of use |
| US7573382B2 (en) | 2007-04-02 | 2009-08-11 | General Electric Company | System and method to manage movement of assets |
| US8425542B2 (en) * | 2007-04-27 | 2013-04-23 | Wisconsin Alumni Research Foundation | Aneurysm occlusion device containing bioactive and biocompatible copolymer shell and biocompatible metallic frame member |
| EP2157937B1 (en) | 2007-06-04 | 2017-03-22 | Sequent Medical, Inc. | Devices for treatment of vascular defects |
| US8361138B2 (en) * | 2007-07-25 | 2013-01-29 | Aga Medical Corporation | Braided occlusion device having repeating expanded volume segments separated by articulation segments |
| US20110144669A1 (en) | 2007-09-11 | 2011-06-16 | Nfocus Neuromedical Inc. | Aneurysm cover device for embolic delivery and retention |
| US20090082803A1 (en) * | 2007-09-26 | 2009-03-26 | Aga Medical Corporation | Braided vascular devices having no end clamps |
| US9414842B2 (en) | 2007-10-12 | 2016-08-16 | St. Jude Medical, Cardiology Division, Inc. | Multi-component vascular device |
| CA2710781C (en) | 2007-12-21 | 2016-09-27 | Microvention, Inc. | A system and method of detecting implant detachment |
| JP5160878B2 (ja) | 2007-12-21 | 2013-03-13 | 浜松ホトニクス株式会社 | 試料同定装置および試料同定方法 |
| US20090227976A1 (en) | 2008-03-05 | 2009-09-10 | Calabria Marie F | Multiple biocompatible polymeric strand aneurysm embolization system and method |
| US20090264978A1 (en) | 2008-03-27 | 2009-10-22 | Dieck Martin S | Friction-Release Distal Latch Implant Delivery System and Components |
| US7806919B2 (en) * | 2008-04-01 | 2010-10-05 | Medtronic Vascular, Inc. | Double-walled stent system |
| ES2430117T3 (es) * | 2008-04-21 | 2013-11-19 | Covidien Lp | Dispositivos embólicos de bola de trenza y sistemas de colocación |
| WO2009132141A1 (en) | 2008-04-22 | 2009-10-29 | Coherex Medical, Inc. | Device, system and method for aneurysm embolization |
| WO2009134337A1 (en) | 2008-05-01 | 2009-11-05 | Aneuclose Llc | Aneurysm occlusion device |
| BRPI0911923B8 (pt) | 2008-05-02 | 2021-06-22 | Sequent Medical Inc | dispositivo para tratamento de um aneurisma cerebral |
| US8041095B2 (en) | 2008-06-11 | 2011-10-18 | Siemens Aktiengesellschaft | Method and apparatus for pretreatment planning of endovascular coil placement |
| US9351715B2 (en) * | 2008-07-24 | 2016-05-31 | St. Jude Medical, Cardiology Division, Inc. | Multi-layered medical device for treating a target site and associated method |
| US20100069948A1 (en) * | 2008-09-12 | 2010-03-18 | Micrus Endovascular Corporation | Self-expandable aneurysm filling device, system and method of placement |
| US9427304B2 (en) * | 2008-10-27 | 2016-08-30 | St. Jude Medical, Cardiology Division, Inc. | Multi-layer device with gap for treating a target site and associated method |
| US20120071911A1 (en) * | 2009-05-20 | 2012-03-22 | University Of Miami | Spherical helix embolic coils for the treatment of cerebral aneurysms |
| US20110054515A1 (en) * | 2009-08-25 | 2011-03-03 | John Bridgeman | Device and method for occluding the left atrial appendage |
| US20110152993A1 (en) | 2009-11-05 | 2011-06-23 | Sequent Medical Inc. | Multiple layer filamentary devices or treatment of vascular defects |
| CN102791205B (zh) * | 2009-11-09 | 2016-02-03 | 恩福克斯神经医学股份有限公司 | 栓塞装置 |
| US8771289B2 (en) | 2009-12-21 | 2014-07-08 | Acist Medical Systems, Inc. | Thrombus removal device and system |
| EP2613735B1 (en) | 2010-09-10 | 2018-05-09 | Covidien LP | Devices for the treatment of vascular defects |
| WO2012078678A1 (en) | 2010-12-06 | 2012-06-14 | Tyco Healthcare Group Lp | Vascular remodeling device |
| US20120283768A1 (en) | 2011-05-05 | 2012-11-08 | Sequent Medical Inc. | Method and apparatus for the treatment of large and giant vascular defects |
| US8261648B1 (en) * | 2011-10-17 | 2012-09-11 | Sequent Medical Inc. | Braiding mechanism and methods of use |
| US9211132B2 (en) | 2012-06-27 | 2015-12-15 | MicoVention, Inc. | Obstruction removal system |
-
2010
- 2010-11-04 US US12/939,901 patent/US20110152993A1/en not_active Abandoned
- 2010-11-04 WO PCT/US2010/055494 patent/WO2011057002A2/en not_active Ceased
- 2010-11-04 CN CN2010800543559A patent/CN102639181A/zh active Pending
- 2010-11-04 EP EP10829110.5A patent/EP2496299B1/en active Active
- 2010-11-04 BR BR112012010758A patent/BR112012010758A2/pt not_active IP Right Cessation
- 2010-11-04 JP JP2012537226A patent/JP2013509914A/ja active Pending
- 2010-11-04 AU AU2010315106A patent/AU2010315106A1/en not_active Abandoned
- 2010-11-04 CA CA2778639A patent/CA2778639A1/en not_active Abandoned
-
2012
- 2012-04-04 US US13/439,754 patent/US9918720B2/en active Active
-
2016
- 2016-04-08 US US15/094,084 patent/US10238393B2/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008126060A (ja) * | 2006-11-20 | 2008-06-05 | Stout Medical Group Lp | 機械的組織デバイスおよび方法 |
| JP2009000497A (ja) * | 2007-06-21 | 2009-01-08 | Aga Medical Corp | 血管障害部位を閉塞するための多層編組構造 |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014512872A (ja) * | 2011-02-11 | 2014-05-29 | コヴィディエン リミテッド パートナーシップ | 二段階配備動脈瘤塞栓用デバイス |
| JP2017511202A (ja) * | 2014-04-14 | 2017-04-20 | シークエント メディカル インコーポレイテッド | 処置的血管手技のためのデバイス |
| US12029431B2 (en) | 2014-04-30 | 2024-07-09 | Stryker Ireland Technology, Ltd. | Occlusion device |
| US12414775B1 (en) | 2014-04-30 | 2025-09-16 | Stryker Ireland Technology LTD | Occlusion device |
| JP2016096917A (ja) * | 2014-11-19 | 2016-05-30 | グンゼ株式会社 | 医療用材料 |
| JP2018538064A (ja) * | 2015-12-07 | 2018-12-27 | シーラス エンドバスキュラー リミテッド | 閉塞デバイス |
| JP2021120028A (ja) * | 2015-12-07 | 2021-08-19 | シーラス エンドバスキュラー リミテッド | 閉塞デバイス |
| JP7227307B2 (ja) | 2015-12-07 | 2023-02-21 | シーラス エンドバスキュラー リミテッド | 閉塞デバイス |
| JP2019506230A (ja) * | 2016-02-10 | 2019-03-07 | マイクロベンション インコーポレイテッドMicrovention, Inc. | 血管閉塞用デバイス |
| US12285175B2 (en) | 2016-03-11 | 2025-04-29 | Stryker Ireland Technology Ltd. | Occlusion device |
| US11648013B2 (en) | 2016-03-11 | 2023-05-16 | Cerus Endovascular Limited | Occlusion device |
| JP2019111343A (ja) * | 2017-12-22 | 2019-07-11 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 動脈瘤装置及び送達システム |
| JP7282514B2 (ja) | 2017-12-22 | 2023-05-29 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 動脈瘤装置及び送達システム |
| JP2019209145A (ja) * | 2018-05-31 | 2019-12-12 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 動脈瘤装置及び送達システム |
| JP7483744B2 (ja) | 2019-03-15 | 2024-05-15 | マイクロベンション インコーポレイテッド | 血管障害の治療のためのフィラメント状デバイス |
| JP7483743B2 (ja) | 2019-03-15 | 2024-05-15 | マイクロベンション インコーポレイテッド | 血管障害の治療のための可撓性継合部を有するフィラメント状デバイス |
| JP2024102213A (ja) * | 2019-03-15 | 2024-07-30 | マイクロベンション インコーポレイテッド | 血管障害の治療のための可撓性継合部を有するフィラメント状デバイス |
| JP2022525745A (ja) * | 2019-03-15 | 2022-05-19 | シークエント メディカル インコーポレイテッド | 血管障害の治療のためのフィラメント状デバイス |
| JP2022525317A (ja) * | 2019-03-15 | 2022-05-12 | シークエント メディカル インコーポレイテッド | 血管障害の治療のための可撓性継合部を有するフィラメント状デバイス |
| JP2022532912A (ja) * | 2019-05-25 | 2022-07-20 | ギャラクシー セラピューティクス, インコーポレイテッド | 動脈瘤を治療するためのシステムおよび方法 |
| JP7540734B2 (ja) | 2019-05-25 | 2024-08-27 | ギャラクシー セラピューティクス, インコーポレイテッド | 動脈瘤を治療するためのシステムおよび方法 |
| US11406404B2 (en) | 2020-02-20 | 2022-08-09 | Cerus Endovascular Limited | Clot removal distal protection methods |
| US12303153B2 (en) | 2020-02-20 | 2025-05-20 | Stryker Ireland Technology Ltd. | Clot removal distal protection methods |
| WO2022162971A1 (ja) * | 2021-01-27 | 2022-08-04 | 株式会社SG-1Medical | 血管プラグ及び治療デバイス |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2010315106A1 (en) | 2012-05-17 |
| US10238393B2 (en) | 2019-03-26 |
| CN102639181A (zh) | 2012-08-15 |
| US20120197283A1 (en) | 2012-08-02 |
| EP2496299A4 (en) | 2014-08-27 |
| EP2496299B1 (en) | 2019-03-06 |
| US20160249937A1 (en) | 2016-09-01 |
| WO2011057002A3 (en) | 2011-09-29 |
| BR112012010758A2 (pt) | 2019-09-24 |
| CA2778639A1 (en) | 2011-05-12 |
| EP2496299A2 (en) | 2012-09-12 |
| WO2011057002A2 (en) | 2011-05-12 |
| US9918720B2 (en) | 2018-03-20 |
| US20110152993A1 (en) | 2011-06-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7566843B2 (ja) | 血管系の治療のためのデバイスの製造方法 | |
| US10238393B2 (en) | Multiple layer filamentary devices for treatment of vascular defects | |
| US10136896B2 (en) | Filamentary devices for treatment of vascular defects | |
| US20120283768A1 (en) | Method and apparatus for the treatment of large and giant vascular defects |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20131004 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20131004 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140522 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20140523 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20141104 |