JP2010046059A - バナナ処理方法 - Google Patents
バナナ処理方法 Download PDFInfo
- Publication number
- JP2010046059A JP2010046059A JP2009185595A JP2009185595A JP2010046059A JP 2010046059 A JP2010046059 A JP 2010046059A JP 2009185595 A JP2009185595 A JP 2009185595A JP 2009185595 A JP2009185595 A JP 2009185595A JP 2010046059 A JP2010046059 A JP 2010046059A
- Authority
- JP
- Japan
- Prior art keywords
- banana
- liquid composition
- groups
- cyclopropene
- bananas
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 235000018290 Musa x paradisiaca Nutrition 0.000 title claims abstract description 48
- 238000000034 method Methods 0.000 title claims abstract description 24
- 240000005561 Musa balbisiana Species 0.000 title 1
- 239000000203 mixture Substances 0.000 claims abstract description 62
- 241000234295 Musa Species 0.000 claims abstract description 47
- 239000007788 liquid Substances 0.000 claims abstract description 39
- OOXWYYGXTJLWHA-UHFFFAOYSA-N cyclopropene Chemical compound C1C=C1 OOXWYYGXTJLWHA-UHFFFAOYSA-N 0.000 claims abstract description 27
- 239000008393 encapsulating agent Substances 0.000 claims abstract description 24
- 239000002738 chelating agent Substances 0.000 claims description 17
- SHDPRTQPPWIEJG-UHFFFAOYSA-N 1-methylcyclopropene Chemical group CC1=CC1 SHDPRTQPPWIEJG-UHFFFAOYSA-N 0.000 claims description 16
- 229910052751 metal Inorganic materials 0.000 claims description 16
- 239000002184 metal Substances 0.000 claims description 16
- 239000002736 nonionic surfactant Substances 0.000 claims description 9
- 229920001450 Alpha-Cyclodextrin Polymers 0.000 claims description 6
- HFHDHCJBZVLPGP-RWMJIURBSA-N alpha-cyclodextrin Chemical group OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO HFHDHCJBZVLPGP-RWMJIURBSA-N 0.000 claims description 6
- 229940043377 alpha-cyclodextrin Drugs 0.000 claims description 6
- 239000005969 1-Methyl-cyclopropene Substances 0.000 claims description 2
- 238000007654 immersion Methods 0.000 claims description 2
- 240000008790 Musa x paradisiaca Species 0.000 description 42
- 235000021015 bananas Nutrition 0.000 description 37
- 125000001424 substituent group Chemical group 0.000 description 19
- -1 alkenoxy Chemical group 0.000 description 14
- 238000011282 treatment Methods 0.000 description 14
- 125000004122 cyclic group Chemical group 0.000 description 13
- 229910052739 hydrogen Inorganic materials 0.000 description 13
- 239000001257 hydrogen Substances 0.000 description 12
- 125000003636 chemical group Chemical group 0.000 description 11
- 125000001931 aliphatic group Chemical group 0.000 description 10
- 238000003306 harvesting Methods 0.000 description 10
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 9
- 239000005977 Ethylene Substances 0.000 description 9
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 9
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 9
- 229920000858 Cyclodextrin Polymers 0.000 description 8
- 125000004429 atom Chemical group 0.000 description 8
- 125000000623 heterocyclic group Chemical group 0.000 description 8
- 125000003118 aryl group Chemical group 0.000 description 7
- 239000008139 complexing agent Substances 0.000 description 7
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 7
- 235000003805 Musa ABB Group Nutrition 0.000 description 6
- 235000015266 Plantago major Nutrition 0.000 description 6
- 241000013557 Plantaginaceae Species 0.000 description 5
- 125000000217 alkyl group Chemical group 0.000 description 5
- 125000005842 heteroatom Chemical group 0.000 description 5
- 239000000243 solution Substances 0.000 description 5
- 230000035882 stress Effects 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 229940097362 cyclodextrins Drugs 0.000 description 4
- 238000007598 dipping method Methods 0.000 description 4
- 235000013399 edible fruits Nutrition 0.000 description 4
- 125000005843 halogen group Chemical group 0.000 description 4
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 description 4
- 230000005070 ripening Effects 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical class NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 3
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 125000003342 alkenyl group Chemical group 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 3
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical class OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 3
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 3
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 3
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 3
- 150000001943 cyclopropenes Chemical class 0.000 description 3
- 125000000298 cyclopropenyl group Chemical group [H]C1=C([H])C1([H])* 0.000 description 3
- 125000004663 dialkyl amino group Chemical group 0.000 description 3
- 239000006185 dispersion Substances 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 150000002431 hydrogen Chemical class 0.000 description 3
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 150000002923 oximes Chemical class 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 3
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 3
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 2
- QOPUBSBYMCLLKW-UHFFFAOYSA-N 2-[2-[bis(carboxymethyl)amino]ethyl-(carboxymethyl)amino]-4-hydroxybutanoic acid Chemical compound OCCC(C(O)=O)N(CC(O)=O)CCN(CC(O)=O)CC(O)=O QOPUBSBYMCLLKW-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- XTEGARKTQYYJKE-UHFFFAOYSA-M Chlorate Chemical compound [O-]Cl(=O)=O XTEGARKTQYYJKE-UHFFFAOYSA-M 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 238000006424 Flood reaction Methods 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- 229920002125 Sokalan® Polymers 0.000 description 2
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 description 2
- 125000005083 alkoxyalkoxy group Chemical group 0.000 description 2
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 2
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 2
- 125000005600 alkyl phosphonate group Chemical group 0.000 description 2
- 125000004414 alkyl thio group Chemical group 0.000 description 2
- 125000000304 alkynyl group Chemical group 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 125000002619 bicyclic group Chemical group 0.000 description 2
- 238000009835 boiling Methods 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 230000001143 conditioned effect Effects 0.000 description 2
- 150000003983 crown ethers Chemical class 0.000 description 2
- 125000004093 cyano group Chemical group *C#N 0.000 description 2
- 125000006310 cycloalkyl amino group Chemical group 0.000 description 2
- 125000005144 cycloalkylsulfonyl group Chemical group 0.000 description 2
- 125000005240 diheteroarylamino group Chemical group 0.000 description 2
- 125000004438 haloalkoxy group Chemical group 0.000 description 2
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 2
- VINBVOMNIBDIPH-UHFFFAOYSA-N isocyanoimino(oxo)methane Chemical compound O=C=N[N+]#[C-] VINBVOMNIBDIPH-UHFFFAOYSA-N 0.000 description 2
- 125000001810 isothiocyanato group Chemical group *N=C=S 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 2
- LHKVDVFVJMYULK-UHFFFAOYSA-N nitrosylazide Chemical compound [N-]=[N+]=NN=O LHKVDVFVJMYULK-UHFFFAOYSA-N 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 150000003839 salts Chemical group 0.000 description 2
- 239000002002 slurry Substances 0.000 description 2
- 238000002791 soaking Methods 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 125000004001 thioalkyl group Chemical group 0.000 description 2
- 125000004665 trialkylsilyl group Chemical group 0.000 description 2
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 description 1
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 description 1
- JPRPJUMQRZTTED-UHFFFAOYSA-N 1,3-dioxolanyl Chemical group [CH]1OCCO1 JPRPJUMQRZTTED-UHFFFAOYSA-N 0.000 description 1
- TYFSYONDMQEGJK-UHFFFAOYSA-N 2-(2,2-dihydroxyethylamino)acetic acid Chemical compound OC(O)CNCC(O)=O TYFSYONDMQEGJK-UHFFFAOYSA-N 0.000 description 1
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical class NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 description 1
- 125000001963 4 membered heterocyclic group Chemical group 0.000 description 1
- YCPXWRQRBFJBPZ-UHFFFAOYSA-N 5-sulfosalicylic acid Chemical compound OC(=O)C1=CC(S(O)(=O)=O)=CC=C1O YCPXWRQRBFJBPZ-UHFFFAOYSA-N 0.000 description 1
- FRBYJCORNHQJTG-UHFFFAOYSA-N C(CN(C(C1=CC=CC=C1)C(=O)O)O)N(C(C1=CC=CC=C1)C(=O)O)O Chemical compound C(CN(C(C1=CC=CC=C1)C(=O)O)O)N(C(C1=CC=CC=C1)C(=O)O)O FRBYJCORNHQJTG-UHFFFAOYSA-N 0.000 description 1
- 241001550206 Colla Species 0.000 description 1
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 241000252165 Elops saurus Species 0.000 description 1
- 239000001116 FEMA 4028 Substances 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 244000011211 Intsia bijuga Species 0.000 description 1
- 235000004462 Musa AAB Group Nutrition 0.000 description 1
- FSVCELGFZIQNCK-UHFFFAOYSA-N N,N-bis(2-hydroxyethyl)glycine Chemical compound OCCN(CCO)CC(O)=O FSVCELGFZIQNCK-UHFFFAOYSA-N 0.000 description 1
- JCXJVPUVTGWSNB-UHFFFAOYSA-N Nitrogen dioxide Chemical compound O=[N]=O JCXJVPUVTGWSNB-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 229920002845 Poly(methacrylic acid) Polymers 0.000 description 1
- 229920002873 Polyethylenimine Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 206010063493 Premature ageing Diseases 0.000 description 1
- 208000032038 Premature aging Diseases 0.000 description 1
- 239000002262 Schiff base Substances 0.000 description 1
- 150000004753 Schiff bases Chemical class 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 229920004482 WACKER® Polymers 0.000 description 1
- 229910021536 Zeolite Inorganic materials 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 125000005194 alkoxycarbonyloxy group Chemical group 0.000 description 1
- 125000000676 alkoxyimino group Chemical group 0.000 description 1
- 125000003282 alkyl amino group Chemical group 0.000 description 1
- 125000004471 alkyl aminosulfonyl group Chemical group 0.000 description 1
- 125000005196 alkyl carbonyloxy group Chemical group 0.000 description 1
- 125000005282 allenyl group Chemical group 0.000 description 1
- 150000001414 amino alcohols Chemical class 0.000 description 1
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 1
- 125000001769 aryl amino group Chemical group 0.000 description 1
- 125000005141 aryl amino sulfonyl group Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- WHGYBXFWUBPSRW-FOUAGVGXSA-N beta-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO WHGYBXFWUBPSRW-FOUAGVGXSA-N 0.000 description 1
- 235000011175 beta-cyclodextrine Nutrition 0.000 description 1
- 229960004853 betadex Drugs 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- SXDBWCPKPHAZSM-UHFFFAOYSA-M bromate Inorganic materials [O-]Br(=O)=O SXDBWCPKPHAZSM-UHFFFAOYSA-M 0.000 description 1
- SXDBWCPKPHAZSM-UHFFFAOYSA-N bromic acid Chemical compound OBr(=O)=O SXDBWCPKPHAZSM-UHFFFAOYSA-N 0.000 description 1
- 230000001680 brushing effect Effects 0.000 description 1
- 125000002837 carbocyclic group Chemical group 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 125000001651 cyanato group Chemical group [*]OC#N 0.000 description 1
- 125000001559 cyclopropyl group Chemical class [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 125000004472 dialkylaminosulfonyl group Chemical group 0.000 description 1
- LXCYSACZTOKNNS-UHFFFAOYSA-N diethoxy(oxo)phosphanium Chemical compound CCO[P+](=O)OCC LXCYSACZTOKNNS-UHFFFAOYSA-N 0.000 description 1
- JGUQDUKBUKFFRO-CIIODKQPSA-N dimethylglyoxime Chemical compound O/N=C(/C)\C(\C)=N\O JGUQDUKBUKFFRO-CIIODKQPSA-N 0.000 description 1
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 150000002118 epoxides Chemical class 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- GDSRMADSINPKSL-HSEONFRVSA-N gamma-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO GDSRMADSINPKSL-HSEONFRVSA-N 0.000 description 1
- 229940080345 gamma-cyclodextrin Drugs 0.000 description 1
- 239000000174 gluconic acid Substances 0.000 description 1
- 235000012208 gluconic acid Nutrition 0.000 description 1
- 125000004995 haloalkylthio group Chemical group 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 125000006517 heterocyclyl carbonyl group Chemical group 0.000 description 1
- 125000005844 heterocyclyloxy group Chemical group 0.000 description 1
- 235000015243 ice cream Nutrition 0.000 description 1
- ICIWUVCWSCSTAQ-UHFFFAOYSA-M iodate Chemical compound [O-]I(=O)=O ICIWUVCWSCSTAQ-UHFFFAOYSA-M 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 239000004816 latex Substances 0.000 description 1
- 229920000126 latex Polymers 0.000 description 1
- 150000002678 macrocyclic compounds Chemical class 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 239000004530 micro-emulsion Substances 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- MGFYIUFZLHCRTH-UHFFFAOYSA-N nitrilotriacetic acid Chemical compound OC(=O)CN(CC(O)=O)CC(O)=O MGFYIUFZLHCRTH-UHFFFAOYSA-N 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- JZRYQZJSTWVBBD-UHFFFAOYSA-N pentaporphyrin i Chemical class N1C(C=C2NC(=CC3=NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 JZRYQZJSTWVBBD-UHFFFAOYSA-N 0.000 description 1
- VLTRZXGMWDSKGL-UHFFFAOYSA-M perchlorate Inorganic materials [O-]Cl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-M 0.000 description 1
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical compound OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- ORIHZIZPTZTNCU-YVMONPNESA-N salicylaldoxime Chemical compound O\N=C/C1=CC=CC=C1O ORIHZIZPTZTNCU-YVMONPNESA-N 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- FQENQNTWSFEDLI-UHFFFAOYSA-J sodium diphosphate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]P([O-])(=O)OP([O-])([O-])=O FQENQNTWSFEDLI-UHFFFAOYSA-J 0.000 description 1
- 235000019832 sodium triphosphate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 125000003107 substituted aryl group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 125000004963 sulfonylalkyl group Chemical group 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 150000003464 sulfur compounds Chemical class 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 235000019818 tetrasodium diphosphate Nutrition 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23B—PRESERVATION OF FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES; CHEMICAL RIPENING OF FRUIT OR VEGETABLES
- A23B2/00—Preservation of foods or foodstuffs, in general
- A23B2/70—Preservation of foods or foodstuffs, in general by treatment with chemicals
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23B—PRESERVATION OF FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES; CHEMICAL RIPENING OF FRUIT OR VEGETABLES
- A23B7/00—Preservation of fruit or vegetables; Chemical ripening of fruit or vegetables
- A23B7/14—Preserving or ripening with chemicals not covered by group A23B7/08 or A23B7/10
- A23B7/153—Preserving or ripening with chemicals not covered by group A23B7/08 or A23B7/10 in the form of liquids or solids
- A23B7/154—Organic compounds; Microorganisms; Enzymes
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N27/00—Biocides, pest repellants or attractants, or plant growth regulators containing hydrocarbons
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N3/00—Preservation of plants or parts thereof, e.g. inhibiting evaporation, improvement of the appearance of leaves or protection against physical influences such as UV radiation using chemical compositions; Grafting wax
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- General Health & Medical Sciences (AREA)
- Health & Medical Sciences (AREA)
- Plant Pathology (AREA)
- Environmental Sciences (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- Chemical & Material Sciences (AREA)
- Polymers & Plastics (AREA)
- Food Science & Technology (AREA)
- Pest Control & Pesticides (AREA)
- Toxicology (AREA)
- General Chemical & Material Sciences (AREA)
- Microbiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Storage Of Fruits Or Vegetables (AREA)
- Preparation Of Fruits And Vegetables (AREA)
- Food Preservation Except Freezing, Refrigeration, And Drying (AREA)
- Detergent Compositions (AREA)
Abstract
【解決手段】シクロプロペン分子カプセル化剤複合体を含む液体組成物とバナナとを接触させ、当該接触の期間が1秒〜4分であることを含む、バナナを処理する方法が提供される。
【選択図】なし
Description
バナナは様々な問題を生じやすい。そのような問題の1つは、場合によって輸送中に起こる早期の熟成である。バナナは輸送時間よりも長い緑色期間(green life)(すなわち、バナナが緑色のままである期間)を有することが望まれる。ある場合には、ある出来事がバナナの緑色期間をより短くすることができる。例えば、輸送中にバナナの容器の内部がエチレンガスに曝露される場合には、バナナの多くがその目的地に到着する前に熟し、これらのバナナの多くは廃棄される必要があるであろう。この早期の熟成はバナナ産業に多大な損失を生じさせる。
本明細書において使用される場合、バナナが「処理される」と称される場合には、バナナが本発明の液体組成物と接触させられることを意味する。
式中、R1、R2、R3およびR4のそれぞれは独立して、Hおよび式:
また、好適なR1、R2、R3およびR4基には、例えば、置換および非置換の複素環式基(すなわち、芳香族または非芳香族で、環内に少なくとも1つのヘテロ原子を有する環式基)がある。
好適な金属錯化剤の混合物も好適である。
本発明のある実施形態においては、バナナは収穫後(すなわち、房が偽茎から分離された後)36時間以下で処理される。ある実施形態においては、収穫から処理までの時間は24時間以下;または10時間以下;または3時間以下;または1時間以下;または20分以下である。
本明細書および特許請求の範囲の目的のために、ここで開示されるそれぞれの操作は、他に示されない限りは、25℃で行われるものと理解される。
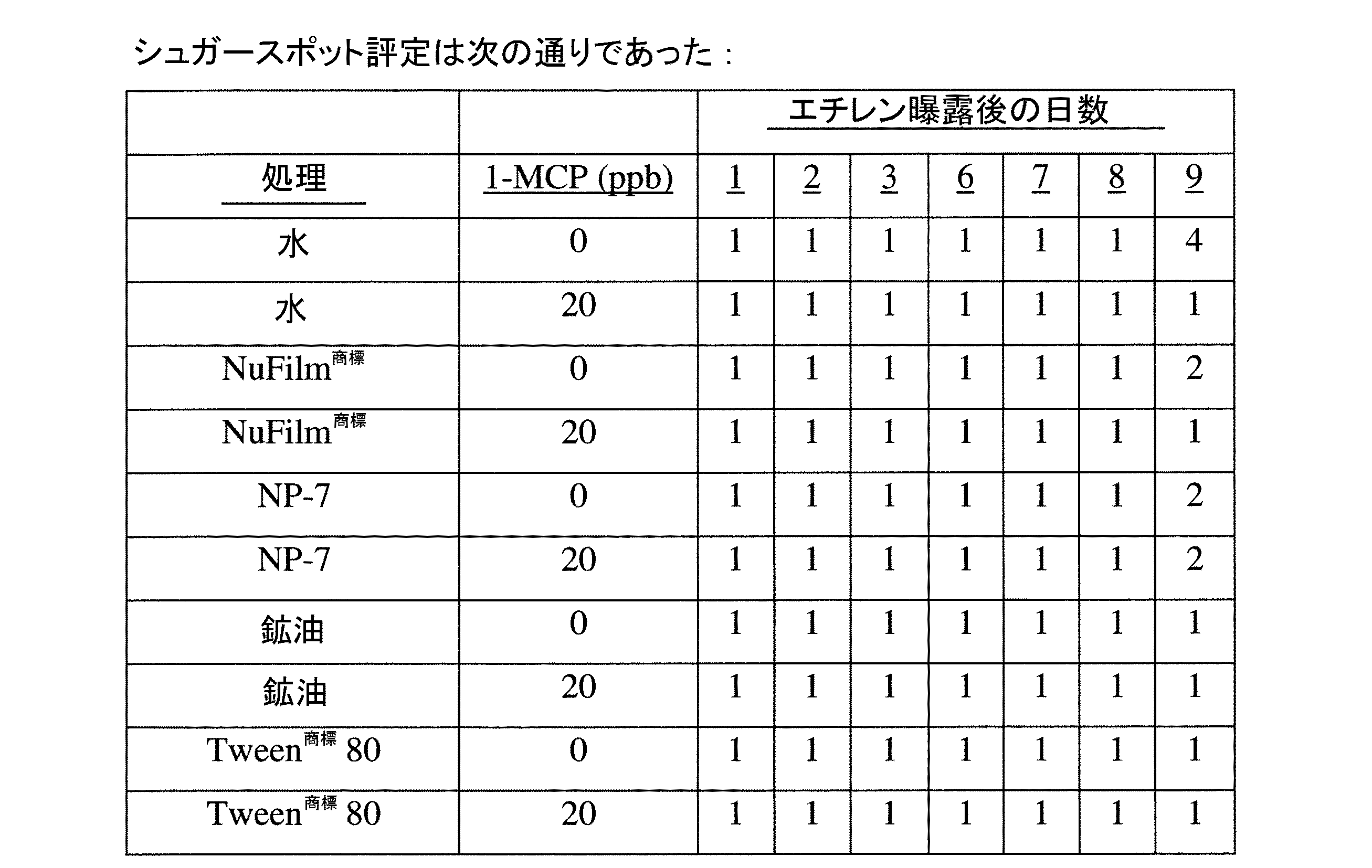
コスタリカで成長し処理された12週齢のバナナ果実。そのバナナは浸漬された(噴霧ではない)。すべての1−MCP処理の溶液は0.6ml/lのNuFilm 17(登録商標)96%(エラストマー形成性添加剤、Miller Chemical and Fertilizer Co.)を含んでいた。対照のサンプルは浸漬されなかった。アルファ−シクロデキストリン中の1−MCP封入複合体の粉体が水に添加されて、1リットルあたり20マイクログラムの1−MCPの濃度を与える水溶液中に他のサンプルは浸漬された。バナナはゼロ時間(入れおよび出し)、5分または20分間浸漬された。
4つの処理数および処理あたり2つの箱を用いて、完全無作為計画が使用された。それぞれの処理からの集団がエチレン処理後、2日ごとに評価された。
5分間または20分間浸漬されたバナナは正常に熟さなかった。それらは12日後でも緑色のままであった。
対照のバナナと素早い入れおよび出しで浸漬されたされたバナナとの間では熟成において有意な違いは観察されなかった。
バナナ(Cavendish Musa spp.)は以下に示される水溶液中に浸漬された。1−MCPを含む溶液は、1リットルあたり20マイクログラムの1−MCP濃度を与えるように、アルファ−シクロデキストリン中の1−MCPの封入複合体の粉体を水に添加することにより調製された。浸漬の時間は15秒であった。
バナナはコスタリカで成長し試験された12週齢のものであった。浸漬後、バナナは棚の上で乾燥させられ、箱に入れられた。箱は14℃で9日間貯蔵され、次いで20〜21℃で6時間調整され、次いで21℃のチャンバー内で24時間、1リットルあたり100マイクロリットルのエチレンの連続流れに曝露させられ;次いで、周囲条件(20℃および80%相対湿度(RH))に保たれた。
Claims (9)
- シクロプロペン分子封入剤複合体を含む液体組成物とバナナとを接触させ、当該接触の期間が1秒〜4分であることを含む、バナナを処理する方法。
- 液体組成物が水性である請求項1に記載の方法。
- 液体組成物が、液体組成物の全重量を基準にして0〜0.1重量%の非イオン性界面活性剤を含む、請求項1に記載の方法。
- 液体組成物が金属キレート剤を0.1〜100ミリモル/リットルの濃度で含む、請求項1に記載の方法。
- 接触がバナナを液体組成物中に浸漬することにより行われる請求項1に記載の方法。
- 浸漬が5〜60秒の期間を有する請求項5に記載の方法。
- 液体組成物中のシクロプロペンの量が5〜100マイクログラム/リットルである、請求項1に記載の方法。
- シクロプロペンが1−メチルシクロプロペンである請求項1に記載の方法。
- 分子封入剤がアルファ−シクロデキストリンである請求項1に記載の方法。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US18999508P | 2008-08-25 | 2008-08-25 | |
| US61/189,995 | 2008-08-25 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010046059A true JP2010046059A (ja) | 2010-03-04 |
| JP5161846B2 JP5161846B2 (ja) | 2013-03-13 |
Family
ID=40934081
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009185595A Expired - Fee Related JP5161846B2 (ja) | 2008-08-25 | 2009-08-10 | バナナ処理方法 |
Country Status (24)
| Country | Link |
|---|---|
| US (2) | US8377489B2 (ja) |
| EP (1) | EP2158812B1 (ja) |
| JP (1) | JP5161846B2 (ja) |
| KR (1) | KR101189814B1 (ja) |
| CN (1) | CN101658204A (ja) |
| AR (1) | AR073204A1 (ja) |
| AT (1) | ATE525904T1 (ja) |
| AU (1) | AU2009208109B2 (ja) |
| BR (1) | BRPI0902722A2 (ja) |
| CA (1) | CA2676584C (ja) |
| CL (1) | CL2009001754A1 (ja) |
| CO (1) | CO6200114A1 (ja) |
| CR (1) | CR10973A (ja) |
| EC (1) | ECSP099594A (ja) |
| ES (1) | ES2371603T3 (ja) |
| GT (1) | GT200900233A (ja) |
| IL (1) | IL200390A (ja) |
| MX (1) | MX2009008970A (ja) |
| MY (1) | MY149151A (ja) |
| NZ (1) | NZ579201A (ja) |
| PT (1) | PT2158812E (ja) |
| TW (1) | TWI422335B (ja) |
| UA (1) | UA104712C2 (ja) |
| ZA (1) | ZA200905792B (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011246463A (ja) * | 2010-05-25 | 2011-12-08 | Rohm & Haas Co | 植物部分上のワックス状コーティング |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2371603T3 (es) | 2008-08-25 | 2012-01-05 | Rohm And Haas Company | Tratamiento de platanos. |
| WO2011109144A1 (en) * | 2010-03-01 | 2011-09-09 | Rohm And Haas Company | Oil formulations comprising cylcopropene compounds |
| JP5562992B2 (ja) * | 2011-04-05 | 2014-07-30 | ローム アンド ハース カンパニー | 制御放出組成物 |
| US8822382B2 (en) | 2013-05-05 | 2014-09-02 | Nazir Mir | Hydrocolloid systems for reducing loss of volatile active compounds from their liquid formulations for pre- and post harvest use on agricultural crops |
| US8802140B2 (en) * | 2013-05-05 | 2014-08-12 | Nazir Mir | Situ mixing and application of hydrocolloid systems for pre- and post harvest use on agricultural crops |
| CN104855110B (zh) * | 2015-05-27 | 2017-03-01 | 广西壮族自治区农业科学院农产品加工研究所 | 一种延缓香蕉成熟上市的方法 |
| JP6935428B2 (ja) | 2016-02-19 | 2021-09-15 | ヘイゼル テクノロジーズ, インコーポレイテッド | 活性成分の制御放出のための組成物およびその作製方法 |
| EP3429351A4 (en) * | 2016-03-18 | 2019-08-14 | AgroFresh Inc. | LONG-TERM PROCEDURE FOR IMPROVING DISEASE TOLERANCE IN PLANTS |
| CN107467173B (zh) * | 2017-07-01 | 2020-08-07 | 华南农业大学 | 一种粉蕉的保鲜方法 |
| WO2023288294A1 (en) | 2021-07-16 | 2023-01-19 | Novozymes A/S | Compositions and methods for improving the rainfastness of proteins on plant surfaces |
| CA3257053A1 (en) | 2022-05-14 | 2023-11-23 | Novozymes A/S | COMPOSITIONS AND METHODS FOR THE PREVENTION, TREATMENT, SUPPRESSION AND/OR ELIMINATION OF PHYTOPATHOGENIC INFESTATIONS AND INFECTIONS |
| WO2025122503A1 (en) * | 2023-12-04 | 2025-06-12 | Tano Pharmaceuticals, Inc. | Banana stem sap compositions and methods of use thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10501231A (ja) * | 1994-06-03 | 1998-02-03 | ノース・キャロライナ・ステイト・ユニヴァーシティ | 植物中のエチレン反応の阻害方法 |
| JP2005145984A (ja) * | 1998-08-20 | 2005-06-09 | Agrofresh Inc | 植物エチレン応答阻害化合物および複合体 |
| JP2005330287A (ja) * | 2004-05-19 | 2005-12-02 | Rohm & Haas Co | シクロプロペン類および補助剤を有する組成物 |
| JP2005330288A (ja) * | 2004-05-19 | 2005-12-02 | Rohm & Haas Co | シクロプロペン類および金属錯化剤を有する組成物 |
| JP2007131621A (ja) * | 2005-11-08 | 2007-05-31 | Rohm & Haas Co | シクロプロペンおよび非炭化水素油を有する組成物 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4857333A (en) * | 1988-05-12 | 1989-08-15 | Harold Robert G | Food product for administering medication to animals |
| CA2394513C (en) | 1999-12-17 | 2010-03-09 | Agrofresh Inc. | Compounds and complexes for inhibition of ethylene response in plants |
| US20040072694A1 (en) * | 2002-02-25 | 2004-04-15 | Jacobson Richard Martin | Method to inhibit ethylene responses in plants |
| ES2193878B2 (es) | 2002-04-16 | 2004-08-01 | Angel Ruiz Gabaldon | Procedimiento de desecacion de las hojas de los plataneros, conservando los contenidos de tanino fisiologico y su uso como complemento en la alimentacion animal. |
| EP1609359A3 (en) | 2004-06-24 | 2011-10-05 | Rohm and Haas Company | A method for treating plants or plant parts |
| AU2007201831B8 (en) * | 2005-01-14 | 2013-02-21 | Agrofresh Inc. | Contacting crop plants with compositions |
| JP5128392B2 (ja) * | 2007-08-03 | 2013-01-23 | ローム アンド ハース カンパニー | 油配合物 |
| ES2371603T3 (es) | 2008-08-25 | 2012-01-05 | Rohm And Haas Company | Tratamiento de platanos. |
-
2009
- 2009-07-21 ES ES09165962T patent/ES2371603T3/es active Active
- 2009-07-21 AT AT09165962T patent/ATE525904T1/de not_active IP Right Cessation
- 2009-07-21 EP EP09165962A patent/EP2158812B1/en not_active Not-in-force
- 2009-07-21 PT PT09165962T patent/PT2158812E/pt unknown
- 2009-08-10 JP JP2009185595A patent/JP5161846B2/ja not_active Expired - Fee Related
- 2009-08-11 AU AU2009208109A patent/AU2009208109B2/en not_active Ceased
- 2009-08-12 CO CO09084380A patent/CO6200114A1/es active IP Right Grant
- 2009-08-12 TW TW098127071A patent/TWI422335B/zh not_active IP Right Cessation
- 2009-08-12 CR CR10973A patent/CR10973A/es not_active Application Discontinuation
- 2009-08-13 IL IL200390A patent/IL200390A/en not_active IP Right Cessation
- 2009-08-17 GT GT200900233A patent/GT200900233A/es unknown
- 2009-08-19 BR BRPI0902722-0A patent/BRPI0902722A2/pt not_active IP Right Cessation
- 2009-08-20 US US12/583,406 patent/US8377489B2/en not_active Expired - Fee Related
- 2009-08-20 CN CN200910166000A patent/CN101658204A/zh active Pending
- 2009-08-20 ZA ZA200905792A patent/ZA200905792B/xx unknown
- 2009-08-21 MX MX2009008970A patent/MX2009008970A/es active IP Right Grant
- 2009-08-21 CL CL2009001754A patent/CL2009001754A1/es unknown
- 2009-08-21 EC EC2009009594A patent/ECSP099594A/es unknown
- 2009-08-21 NZ NZ579201A patent/NZ579201A/en not_active IP Right Cessation
- 2009-08-24 AR ARP090103251A patent/AR073204A1/es unknown
- 2009-08-24 MY MYPI20093497A patent/MY149151A/en unknown
- 2009-08-24 KR KR1020090078060A patent/KR101189814B1/ko not_active Expired - Fee Related
- 2009-08-25 UA UAA200908868A patent/UA104712C2/uk unknown
- 2009-08-25 CA CA2676584A patent/CA2676584C/en not_active Expired - Fee Related
-
2013
- 2013-01-15 US US13/741,537 patent/US20130129880A1/en not_active Abandoned
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10501231A (ja) * | 1994-06-03 | 1998-02-03 | ノース・キャロライナ・ステイト・ユニヴァーシティ | 植物中のエチレン反応の阻害方法 |
| JP2003201201A (ja) * | 1994-06-03 | 2003-07-18 | Univ North Carolina | 植物中のエチレン反応の阻害方法 |
| JP2005145984A (ja) * | 1998-08-20 | 2005-06-09 | Agrofresh Inc | 植物エチレン応答阻害化合物および複合体 |
| JP2005330287A (ja) * | 2004-05-19 | 2005-12-02 | Rohm & Haas Co | シクロプロペン類および補助剤を有する組成物 |
| JP2005330288A (ja) * | 2004-05-19 | 2005-12-02 | Rohm & Haas Co | シクロプロペン類および金属錯化剤を有する組成物 |
| JP2007131621A (ja) * | 2005-11-08 | 2007-05-31 | Rohm & Haas Co | シクロプロペンおよび非炭化水素油を有する組成物 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011246463A (ja) * | 2010-05-25 | 2011-12-08 | Rohm & Haas Co | 植物部分上のワックス状コーティング |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2009208109B2 (en) | 2014-10-30 |
| CA2676584A1 (en) | 2010-02-25 |
| CR10973A (es) | 2009-10-23 |
| ES2371603T3 (es) | 2012-01-05 |
| ZA200905792B (en) | 2010-06-30 |
| KR101189814B1 (ko) | 2012-10-11 |
| US20130129880A1 (en) | 2013-05-23 |
| EP2158812A1 (en) | 2010-03-03 |
| NZ579201A (en) | 2011-10-28 |
| MX2009008970A (es) | 2010-03-25 |
| BRPI0902722A2 (pt) | 2010-05-25 |
| US8377489B2 (en) | 2013-02-19 |
| MY149151A (en) | 2013-07-15 |
| US20100047408A1 (en) | 2010-02-25 |
| ATE525904T1 (de) | 2011-10-15 |
| AR073204A1 (es) | 2010-10-20 |
| CL2009001754A1 (es) | 2010-11-12 |
| AU2009208109A1 (en) | 2010-03-11 |
| EP2158812B1 (en) | 2011-09-28 |
| KR20100024362A (ko) | 2010-03-05 |
| GT200900233A (es) | 2011-08-18 |
| CN101658204A (zh) | 2010-03-03 |
| CO6200114A1 (es) | 2010-09-20 |
| UA104712C2 (uk) | 2014-03-11 |
| CA2676584C (en) | 2015-03-10 |
| IL200390A (en) | 2013-10-31 |
| IL200390A0 (en) | 2010-04-29 |
| TW201023751A (en) | 2010-07-01 |
| TWI422335B (zh) | 2014-01-11 |
| PT2158812E (pt) | 2011-11-30 |
| JP5161846B2 (ja) | 2013-03-13 |
| ECSP099594A (es) | 2010-03-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5161846B2 (ja) | バナナ処理方法 | |
| EP1856975B1 (en) | Treating horticultural crops | |
| KR101362841B1 (ko) | 식물 부위의 왁스 코팅 | |
| AU2007231682B2 (en) | Cyclopropene compositions | |
| JP2004521648A (ja) | 果物、野菜および鳥の卵、特に有機栽培生産物に有用な被覆用合成物。 | |
| JP2019511923A (ja) | 衛生化された産物を調製および保存するための方法 | |
| KR101250556B1 (ko) | 원예 농작물의 처리 | |
| TW201438577A (zh) | 用於保護和增進香蕉產量的方法和系統 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20101213 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20101215 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110314 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20110401 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20121214 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20151221 Year of fee payment: 3 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |