EP0904533B1 - Sensor zur bestimmung der konzentration oxidierbarer bestandteile in einem gasgemisch - Google Patents
Sensor zur bestimmung der konzentration oxidierbarer bestandteile in einem gasgemisch Download PDFInfo
- Publication number
- EP0904533B1 EP0904533B1 EP97914159A EP97914159A EP0904533B1 EP 0904533 B1 EP0904533 B1 EP 0904533B1 EP 97914159 A EP97914159 A EP 97914159A EP 97914159 A EP97914159 A EP 97914159A EP 0904533 B1 EP0904533 B1 EP 0904533B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- sensor according
- semiconductor
- acceptor
- solid electrolyte
- donor
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 239000000203 mixture Substances 0.000 title claims description 11
- 239000007789 gas Substances 0.000 claims description 39
- 239000007784 solid electrolyte Substances 0.000 claims description 20
- 239000004065 semiconductor Substances 0.000 claims description 13
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 12
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N titanium dioxide Inorganic materials O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 8
- 229910052759 nickel Inorganic materials 0.000 claims description 6
- 229910052758 niobium Inorganic materials 0.000 claims description 6
- 239000010955 niobium Substances 0.000 claims description 6
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 claims description 5
- 239000000758 substrate Substances 0.000 claims description 5
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims description 3
- 239000000470 constituent Substances 0.000 claims description 3
- 229910052802 copper Inorganic materials 0.000 claims description 3
- 239000010949 copper Substances 0.000 claims description 3
- 239000000463 material Substances 0.000 claims description 3
- 238000005259 measurement Methods 0.000 claims description 3
- 239000011148 porous material Substances 0.000 claims description 3
- 239000000654 additive Substances 0.000 claims description 2
- 229910052751 metal Inorganic materials 0.000 claims description 2
- 239000002184 metal Substances 0.000 claims description 2
- 229930195735 unsaturated hydrocarbon Natural products 0.000 claims description 2
- 239000010941 cobalt Substances 0.000 claims 2
- 229910017052 cobalt Inorganic materials 0.000 claims 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 claims 2
- 230000003647 oxidation Effects 0.000 claims 2
- 238000007254 oxidation reaction Methods 0.000 claims 2
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 claims 1
- 229910052804 chromium Inorganic materials 0.000 claims 1
- 239000011651 chromium Substances 0.000 claims 1
- 239000006104 solid solution Substances 0.000 claims 1
- 229910052715 tantalum Inorganic materials 0.000 claims 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical group [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 claims 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims 1
- 230000007704 transition Effects 0.000 claims 1
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 11
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 9
- 229910052760 oxygen Inorganic materials 0.000 description 9
- 239000001301 oxygen Substances 0.000 description 9
- 229910052697 platinum Inorganic materials 0.000 description 6
- 238000006243 chemical reaction Methods 0.000 description 5
- 230000035945 sensitivity Effects 0.000 description 5
- QQONPFPTGQHPMA-UHFFFAOYSA-N Propene Chemical compound CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 4
- 230000003197 catalytic effect Effects 0.000 description 4
- 238000002485 combustion reaction Methods 0.000 description 3
- 229930195733 hydrocarbon Natural products 0.000 description 3
- 150000002430 hydrocarbons Chemical class 0.000 description 3
- 230000002452 interceptive effect Effects 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical compound O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 2
- 238000011088 calibration curve Methods 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 229910052737 gold Inorganic materials 0.000 description 2
- 239000010931 gold Substances 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- -1 platinum metals Chemical class 0.000 description 2
- 230000036647 reaction Effects 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 230000005678 Seebeck effect Effects 0.000 description 1
- 230000000274 adsorptive effect Effects 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 239000007772 electrode material Substances 0.000 description 1
- 238000011067 equilibration Methods 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- JUWSSMXCCAMYGX-UHFFFAOYSA-N gold platinum Chemical compound [Pt].[Au] JUWSSMXCCAMYGX-UHFFFAOYSA-N 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 230000007257 malfunction Effects 0.000 description 1
- 238000000034 method Methods 0.000 description 1
- 239000011241 protective layer Substances 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/26—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating electrochemical variables; by using electrolysis or electrophoresis
- G01N27/403—Cells and electrode assemblies
- G01N27/406—Cells and probes with solid electrolytes
- G01N27/407—Cells and probes with solid electrolytes for investigating or analysing gases
- G01N27/4073—Composition or fabrication of the solid electrolyte
- G01N27/4074—Composition or fabrication of the solid electrolyte for detection of gases other than oxygen
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/26—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating electrochemical variables; by using electrolysis or electrophoresis
- G01N27/403—Cells and electrode assemblies
- G01N27/406—Cells and probes with solid electrolytes
- G01N27/407—Cells and probes with solid electrolytes for investigating or analysing gases
Definitions
- the invention is based on a sensor for determining the concentration of oxidizable constituents in a gas mixture, in particular for determining one or more of the gases NO x , CO, H2 and preferably unsaturated hydrocarbons according to the preamble of the main claim.
- Exhaust gases from gasoline and diesel engines, internal combustion engines and incineration plants can cause increased concentrations of oxidizable components, in particular NO x , CO, H2 and hydrocarbons, for example as a result of a component malfunction, such as an injection valve, or as a result of incomplete combustion. To optimize the combustion reactions, it is therefore necessary to know the concentration of these exhaust gas components.
- JP-OS 60-61654 describes a method for determining oxidizable gases, according to which stoichiometric reaction with oxygen takes place on a first measuring electrode made of platinum metals and quasi-equilibrium states on one or more further metallic measuring electrodes with reduced catalytic activity for the oxygen equilibrium reaction can be set.
- the Nernst voltages E1 and E2 are measured between the measuring electrodes and a reference electrode which is exposed to a reference gas with constant oxygen partial pressure, and the concentration of the gas components is calculated from their difference on the basis of calibration curves.
- Document DE-A-42 25 775 discloses a sensor for determination oxidizable components (NO) in a gas mixture accordingly the preamble of claim 1.
- the sensor according to the invention with the features of In contrast, the main claim enables an increased Miniaturization, a constructive simplification and one cheaper manufacturing.
- the molecules of the measuring gas can therefore by the Pores of the solid electrolyte to the reference electrode diffuse where the thermodynamic equilibrium is established.
- the reference electrode is thus one Exposed to partial pressure of oxygen, which equals thermodynamic equilibrium. So it can there is no need to add a reference gas, which considerably simplifies the construction of the probe.
- thermodynamic equilibrium will already in the solid electrolyte by choosing a catalytically effective one Solid electrolyte material.
- a catalytically effective one Solid electrolyte material As a special advantage is to be seen that thereby the Reference signal interfering gases can be oxidized what the signal evaluation simplified or in the first place allows.
- Advantageously, in addition to Solid electrolytes also make the measuring electrodes porous, which means the diffusion of the molecules of the measuring gas Reference electrode is further improved.
- Electrode materials correspond to the electrode adhesion and thus improves the life of the sensor. Especially It is also advantageous to remove the measuring electrodes Build up semiconductors, reducing the selectivity significant of the gas components to be detected can be increased.
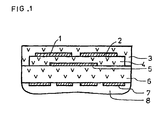
- Figure 1 shows a section through the sensor according to the invention.
- a sensor according to the invention is in section shown.
- An insulating planar ceramic substrate 6 carries on one large area in superimposed Layers a reference electrode 5 preferably made of platinum, a porous solid electrolyte 4, measuring electrodes 1 and 2 and a gas-permeable protective layer 3.
- a reference electrode 5 preferably made of platinum
- a porous solid electrolyte 4 measuring electrodes 1 and 2
- a gas-permeable protective layer 3 On the opposite large area of the substrate is one Heater device 7 applied with cover 8.
- the senor is used in exhaust gases by means of the heating device 7 to a temperature between 300 and 1000 ° C, more advantageous Way heated to 600 ° C.
- the sample gas diffuses through the porous solid electrolyte to the reference electrode 5, which is the setting of the Oxygen equilibrium potential catalyzed.
- the Measuring electrodes 1, 2 are constructed so that they have a reduced catalytic activity for the oxygen equilibrium reaction exhibit.
- the sensor generates over the oxygen ion-conducting solid electrolyte Cell voltage by first using the Reference electrode set half-cell reaction and a second by the oxidizable to be determined Gas components affected at least half-cell reaction a measuring electrode. About the calibration curves are from the concentrations of the gas components determined.
- the sensor according to the invention is thus in the simplest case with a reference electrode that the Equilibration of the gas mixture catalyzed and a measuring electrode, the equilibrium of the Gas mixture is not able to catalyze or only catalyzes little, operational.
- the measuring elements then react with different, from the gas type dependent voltage, based on the reference electrode.
- At least two measuring electrodes furthermore the possibility of the cross sensitivity of a the first measuring electrode completely or at least partially by the signal of another measuring electrode compensate by the sensitivity of this further Measuring electrode on the interfering gas components accordingly is set.
- the solid electrolyte is so trained, e.g. by adding 0.01 to 10% by volume Platinum powder that the solid electrolyte is the gases to be measured catalytically implemented, so that only the thermodynamic Equivalent gases at the reference electrode arrive or that the solid electrolyte only the Reference signal interfering gases.
- metallic electrode fabrics as they e.g. are described in JP-OS 60-61654, or else Semiconductors that have a high specific sensitivity for have certain oxidizable gases. Are particularly suitable semiconducting oxides or mixed oxides, the acceptor and / or Can be donor-doped.
- the high sensitivity of e.g. Acceptor and donor doped n-type Titanium dioxide especially for unsaturated ones Hydrocarbons is due to the adsorptive interaction the orbitals of the unsaturated pi bonds Hydrocarbons with the acceptor sites on the Conditional on semiconductor surface.
- the following example describes a production method for a sensor according to the invention: 7% niobium and 3% of one of the transition metals nickel, copper or iron doped rutile is printed as a 30 ⁇ m thick screen printing layer on a substrate which carries a reference electrode made of platinum, for example, and a solid electrolyte layer above it. A heater device is applied to the opposite side of the substrate. The sensor is sintered at 1200 ° C for 90 minutes with a heating / cooling ramp of 300 ° C / hour. After sintering, the solid electrolyte has pores in the size range from 10 nm to 100 ⁇ m.
- the voltage across the cell constructed in this way is measured at a resistance of 1 MOhm between the reference and the rutile electrode.
- the sensor is heated to 600 ° C with its heater.
- Simulated exhaust gas with 10% oxygen, 5% water and 5% carbon dioxide and 30 ppm sulfur dioxide is used as the measurement gas.
- Oxidizable gases are mixed in the amounts given in the table.
- the last line of the table below shows the voltage values for a mixed potential electrode made of 20% gold and 80% platinum, which represents a measuring electrode according to the prior art.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Physics & Mathematics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Molecular Biology (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Measuring Oxygen Concentration In Cells (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19623212A DE19623212A1 (de) | 1996-06-11 | 1996-06-11 | Sensor zur Bestimmung der Konzentration oxidierbarer Bestandteile in einem Gasgemisch |
| DE19623212 | 1996-06-11 | ||
| PCT/DE1997/000380 WO1997047962A1 (de) | 1996-06-11 | 1997-03-03 | Sensor zur bestimmung der konzentration oxidierbarer bestandteile in einem gasgemisch |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0904533A1 EP0904533A1 (de) | 1999-03-31 |
| EP0904533B1 true EP0904533B1 (de) | 2003-06-04 |
Family
ID=7796596
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP97914159A Expired - Lifetime EP0904533B1 (de) | 1996-06-11 | 1997-03-03 | Sensor zur bestimmung der konzentration oxidierbarer bestandteile in einem gasgemisch |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US6174421B1 (enExample) |
| EP (1) | EP0904533B1 (enExample) |
| JP (1) | JP3933697B2 (enExample) |
| KR (1) | KR20000016502A (enExample) |
| CN (1) | CN1221490A (enExample) |
| DE (2) | DE19623212A1 (enExample) |
| WO (1) | WO1997047962A1 (enExample) |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19901956C2 (de) * | 1999-01-20 | 2003-06-18 | Bosch Gmbh Robert | Sensor zur Analyse von Gasen |
| DE19932048A1 (de) * | 1999-07-09 | 2001-01-11 | Bosch Gmbh Robert | Meßfühler zur Bestimmung einer Konzentration von Gaskomponenten in Gasgemischen |
| DE10111586A1 (de) * | 2001-03-10 | 2002-09-12 | Volkswagen Ag | Verfahren zum Betrieb von Brennkraftmaschinen |
| DE10160704B4 (de) | 2001-12-11 | 2013-07-18 | Volkswagen Ag | Verfahren zum Betrieb von Abgasreinigungsvorrichtungen |
| DE10319664A1 (de) * | 2003-05-02 | 2004-11-18 | Robert Bosch Gmbh | Sensor zur Detektion von Teilchen |
| US7228725B2 (en) * | 2004-01-27 | 2007-06-12 | H2Scan Llc | Thin film gas sensor configuration |
| DE102005015569A1 (de) * | 2005-04-05 | 2006-10-12 | Robert Bosch Gmbh | Keramisches Widerstands- oder Sensorelement |
| JP4430591B2 (ja) * | 2005-07-25 | 2010-03-10 | 日本特殊陶業株式会社 | ガスセンサ素子及びガスセンサ |
| JP4061556B2 (ja) * | 2005-08-12 | 2008-03-19 | 株式会社新潟Tlo | 水素量センサーおよび水素貯蔵装置 |
| DE102006061954A1 (de) * | 2006-12-29 | 2008-07-03 | Robert Bosch Gmbh | Sensorelement mit zusätzlicher Fettgasregelung |
| KR100864381B1 (ko) * | 2007-04-05 | 2008-10-21 | 한국과학기술원 | 질소산화물 센서 및 이를 이용한 전체 질소산화물 농도산출 방법 |
| KR100938673B1 (ko) * | 2007-12-31 | 2010-01-25 | 주식회사 시오스 | 이산화탄소센서 및 상기 이산화탄소센서가 구비된이산화탄소 측정장치 |
| WO2010010978A1 (en) * | 2008-07-23 | 2010-01-28 | Cios Inc. | Nox sensor |
| US8852950B2 (en) * | 2008-09-03 | 2014-10-07 | Testo Ag | Method and device for measuring NOx concentration using measurements of NOx and a second gas component |
| US20110168557A1 (en) * | 2008-09-30 | 2011-07-14 | Iljin Copper Foil Co., Ltd. | Nitrogen-oxide gas sensor |
| EP2330410A4 (en) * | 2008-09-30 | 2013-01-16 | Iljin Copper Foil Co Ltd | NITROGEN OXIDE SENSOR |
| KR101133267B1 (ko) * | 2008-09-30 | 2012-04-05 | 주식회사 시오스 | 질소산화물 가스센서 |
| WO2010038987A2 (ko) * | 2008-09-30 | 2010-04-08 | 일진소재산업(주) | 장기 신호 안정성을 갖는 질소산화물 가스센서 |
| EP2799858A1 (de) * | 2013-05-02 | 2014-11-05 | E+E Elektronik Ges.m.b.H. | Amperometrischer Gassensor |
| CA2951690A1 (en) * | 2014-06-09 | 2015-12-17 | Biometry Inc. | Low cost test strip and method to measure analyte |
| CN105334256A (zh) * | 2015-10-29 | 2016-02-17 | 宁波大学 | 一种基于Ni掺杂PdO敏感电极电位型CO传感器及其制备方法 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DD138245B1 (de) * | 1978-08-30 | 1980-12-10 | Moebius Hans Heinrich | Einrichtung zur gasanalyse mit galvanischen festelektrolytzellen |
| JP2541530B2 (ja) * | 1985-10-29 | 1996-10-09 | コモンウェルス、サイエンティフィク、エンド、インダストリアル、リサ−チ、オ−ガナイゼ−ション | 固体電解質装置及びその製造方法 |
| GB8602544D0 (en) * | 1986-02-03 | 1986-03-12 | Atomic Energy Authority Uk | Sensor |
| DE4225775C2 (de) * | 1992-08-04 | 2002-09-19 | Heraeus Electro Nite Int | Anordnung zum kontinuierlichen Überwachen der Konzentration von NO in Gasgemischen |
| GB2285689A (en) * | 1993-06-18 | 1995-07-19 | Capteur Sensors & Analysers | Semiconducting oxide gas sensors and materials therefor |
| JP3287096B2 (ja) * | 1993-12-27 | 2002-05-27 | 株式会社豊田中央研究所 | イオン導電体を用いたガスセンサおよびその製造方法 |
| DE4408361C2 (de) * | 1994-03-14 | 1996-02-01 | Bosch Gmbh Robert | Elektrochemischer Sensor zur Bestimmung der Sauerstoffkonzentration in Gasgemischen |
| JP3377016B2 (ja) * | 1996-01-26 | 2003-02-17 | 矢崎総業株式会社 | 排気ガス中の酸素濃度測定用限界電流式酸素センサ |
| DE19623434A1 (de) * | 1996-06-12 | 1997-12-18 | Bosch Gmbh Robert | Sensor zur Bestimmung der Konzentration oxidierbarer Bestandteile in einem Gasgemisch |
-
1996
- 1996-06-11 DE DE19623212A patent/DE19623212A1/de not_active Withdrawn
-
1997
- 1997-03-03 WO PCT/DE1997/000380 patent/WO1997047962A1/de not_active Ceased
- 1997-03-03 KR KR1019980710088A patent/KR20000016502A/ko not_active Ceased
- 1997-03-03 EP EP97914159A patent/EP0904533B1/de not_active Expired - Lifetime
- 1997-03-03 JP JP50103298A patent/JP3933697B2/ja not_active Expired - Fee Related
- 1997-03-03 DE DE59710230T patent/DE59710230D1/de not_active Expired - Lifetime
- 1997-03-03 CN CN97195441A patent/CN1221490A/zh active Pending
- 1997-03-03 US US09/202,313 patent/US6174421B1/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| KR20000016502A (ko) | 2000-03-25 |
| JP2000512013A (ja) | 2000-09-12 |
| US6174421B1 (en) | 2001-01-16 |
| EP0904533A1 (de) | 1999-03-31 |
| WO1997047962A1 (de) | 1997-12-18 |
| CN1221490A (zh) | 1999-06-30 |
| DE19623212A1 (de) | 1997-12-18 |
| JP3933697B2 (ja) | 2007-06-20 |
| DE59710230D1 (de) | 2003-07-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0904533B1 (de) | Sensor zur bestimmung der konzentration oxidierbarer bestandteile in einem gasgemisch | |
| EP1166096B1 (de) | Elektrochemischer gassensor | |
| DE69713698T2 (de) | Gasfühler | |
| DE19623434A1 (de) | Sensor zur Bestimmung der Konzentration oxidierbarer Bestandteile in einem Gasgemisch | |
| DE69521451T2 (de) | Verfahren und Vorrichtung zum Messen von einem Gaskomponenten | |
| DE4333232B4 (de) | Meßfühler zur Bestimmung des Sauerstoffgehaltes von Gasgemischen | |
| DE3019072A1 (de) | Vorrichtung zur bestimmung der sauerstoffkonzentration in verbrennungsgasen | |
| DE4408504A1 (de) | Sensor zur Bestimmung der Konzentration von Gaskomponenten in Gasgemischen | |
| DE19861198B4 (de) | Sensor für die Untersuchung von Abgasen und Untersuchungsverfahren | |
| DE19549147C2 (de) | Gassensor | |
| DE19963008B4 (de) | Sensorelement eines Gassensors zur Bestimmung von Gaskomponenten | |
| DE19960338A1 (de) | Gassensor zur Bestimmung der Konzentration von Gaskomponenten in Gasgemischen und dessen Verwendung | |
| DE4021929A1 (de) | Sensor | |
| DE69228507T2 (de) | Sauerstoffsensor | |
| DE19937016A1 (de) | Sensorelement und Verfahren zur Bestimmung der Sauerstoffkonzentration in Gasgemischen | |
| DE10023062B4 (de) | Messeinrichtung zur Konzentrationsbestimmung von Gaskomponenten im Abgas einer Brennkraftmaschine und Verfahren zur Steuerung eines Betriebs der Messeinrichtung | |
| EP0892922B1 (de) | Sensor zur bestimmung der konzentration oxidierbarer bestandteile in einem gasgemisch | |
| DE19806308A1 (de) | Gassensor zur Sauerstoffmessung mit Verwendung und Meßverfahren | |
| DE4243734C2 (de) | Sensor zur Bestimmung von Gaskomponenten und/oder von Gaskonzentrationen von Gasgemischen | |
| DE102009011298A1 (de) | Gassensor | |
| DE10332519A1 (de) | Elektrochemische Pumpzelle für Gassensoren | |
| DE10207229A1 (de) | Katalytisch aktive Schicht | |
| WO2004072634A1 (de) | Sensorelement | |
| DE10319878A1 (de) | Potentiometrischer Kohlenwasserstoffsensor mit sauerstoffpartialdruckunabhängiger Kennlinie | |
| DE102008014655A1 (de) | Sensorelement eines Gassensors und Verfahren zur Bestimmung von Gaskomponenten in Gasgemischen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19990111 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): DE FR GB IT |
|
| RTI1 | Title (correction) |
Free format text: SENSOR FOR DETERMINING THE CONCENTRATION OF OXIDISABLE ELEMENTS IN A GAS MIXTURE |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| RTI1 | Title (correction) |
Free format text: SENSOR FOR DETERMINING THE CONCENTRATION OF OXIDISABLE ELEMENTS IN A GAS MIXTURE |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): DE FR GB IT |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REF | Corresponds to: |
Ref document number: 59710230 Country of ref document: DE Date of ref document: 20030710 Kind code of ref document: P |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 20031007 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20040305 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20060322 Year of fee payment: 10 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20070303 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20071130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070303 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20080326 Year of fee payment: 12 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070402 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20060323 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20100512 Year of fee payment: 14 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090303 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20111001 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 59710230 Country of ref document: DE Effective date: 20111001 |