EP0804503B1 - Eine polyolefinlegierung mit verbesserter oberflächenhärte und kratzfestigkeit, und verfahren zu ihrer herstellung - Google Patents
Eine polyolefinlegierung mit verbesserter oberflächenhärte und kratzfestigkeit, und verfahren zu ihrer herstellung Download PDFInfo
- Publication number
- EP0804503B1 EP0804503B1 EP96901147A EP96901147A EP0804503B1 EP 0804503 B1 EP0804503 B1 EP 0804503B1 EP 96901147 A EP96901147 A EP 96901147A EP 96901147 A EP96901147 A EP 96901147A EP 0804503 B1 EP0804503 B1 EP 0804503B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- weight
- polyolefin alloy
- ethylene
- alloy according
- propylene
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/10—Homopolymers or copolymers of propene
- C08L23/14—Copolymers of propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/10—Homopolymers or copolymers of propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/16—Ethylene-propylene or ethylene-propylene-diene copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L51/00—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L51/06—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers grafted on to homopolymers or copolymers of aliphatic hydrocarbons containing only one carbon-to-carbon double bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/03—Polymer mixtures characterised by other features containing three or more polymers in a blend
Definitions
- the present invention relates to a polyolefin alloy with improved surface hardness and scratch resistance, and processes for producing same. More specifically, the invention relates to a polyolefin alloy on the basis of a polypropylene material, a functionalized polypropylene material, a mineral, an amorphous silica gel and optionally an ethylene/propylene/-polyene terpolymer (EPDM), as well as processes for producing said polyolefin alloy.
- EPDM ethylene/propylene/-polyene terpolymer
- JP 1318051 discloses polyolefins which for this purpose have been modified with a filler on silica basis. Said filler may have been surface treated with silane type compounds.
- the disclosed polyolefins are not alloys and no fatty acid amide-treated silica gel is used in the material.
- JP 1104637 (Showa Denko KK) discloses materials on a polypropylene basis to which silica/alumina spheroids and a polypropylene material functionalized with carboxylic acid or carboxylic anhydride have been added. These materials also contains no fatty acid amide-treated silica gel.
- the invention provides a new polyolefin alloy having improved surface hardness and scratch resistance, and having a melt index MI in the range of 0.1 - 50 g/10 min at 230°C/2.16 kg, particularly in the range of 3 to 40 g/10 min at 230°C/-2.16 kg.
- the polyolefin alloy is characterized in that it is constituted by a blend of:
- EP-A-O 567 058 discloses compositions having improved elasticity, peeling strength and recoating properties, comprising a polypropylene, an ethylene/a-olefin copolymer elastomer, and a modified polyolefin having a functional group in either or both ends thereof, as well as certain substituted amines/amides.
- the compositions can also contain various additives, among which talc is mentioned.
- compositions of EP-A-O 567 058 do not, however, contain the combination of a functionalized polypropylene material of the same kind as component (B) of the polyolefin alloy of the present invention, with a mineral having been surface-treated with an aminosilane, and a fatty acid-treated silica gel, and they do not suggest any improvement of the scratch resistance.
- US 4 675 122 (Lüers et al.) teaches a combined antiblocking agent and lubricant concentrate based on polyolefin, which contains diatomaceous earth, precipitated silica and/or silica gel as the antiblocking agent, and one more more amides of unsaturated C 18 -C 22 fatty acids. No mention is made of any improvement of the scratch-resistance. Also, it could not be considered to be near at hand to combine the teaching of US 4 675 122 with the teaching of the above-discussed EP-A-O 567 058, which also does not mention scratch-resistance.
- EP-A-0 297 693 (McKinney et al.) teaches a composition comprising an intimate mixture of at least one ethylene interpolymer with acrylic acid, methacrylic acid or an ionomer of either of said acids and a slip-enhancing amount of a secondary fatty acid amide.
- the composition may also contain small amounts of finely divided inorganic materials such as silica and talc.
- the aim of the invention of EP-A-O 297 693 is to improve the slip and/or block characteristics of the ethylene interpolymers. No mention is made of any improvement of the scratch-resistance.
- EP-A-O 297 693 is disclosing compositions in which silica may be used, and will come into contact with fatty acid amide, it is not question there of any fatty acid-treated silica gel comprising 25-75% by weight of silica and 75-25% by weight of fatty acid amide as in the polyolefin alloy of the present invention.
- any fatty acid-treated silica gel comprising 25-75% by weight of silica and 75-25% by weight of fatty acid amide as in the polyolefin alloy of the present invention.
- a finely inorganic material is used in the composition of EP-A-0 297 693, no surface treatment thereof with aminosilane is mentioned, neither is there any mention of any functionalized polypropylene material.
- EP-A-O 257 803 (Yamada et al.), comprising an ethylene/methyl methacrylate copolymer, a slip agent which may be a fatty acid amide, and a small amount (0.01-0.1 %wt) of an inorganic filler, which may be a silica or a talc.
- the composition is used to produce peelable protective films with low peeling strength for wrapping around e.g. rubbery articles having a tacky surface.
- EP-A-O 257 803 nor EP-A-O 297 693 suggests the polyolefin alloy of the present invention having improved surface hardness and scratch-resistance.
- US 5 286 791 discloses a filled composition having improved flexural modulus and impact strength, comprising a propylene polymer material, a propylene polymer material grafted with one or more vinyl monomers, and a rubber component.
- the composition may contain i.a. talc and silica, and column 3, lines 25-37, of the patent indicates that the filler may be coated with an organic compound.
- the patent does not suggest any fatty acid amide-treated silica gel in combination with an aminosilane-treated mineral and there is no suggestion in said patent that any such combination would improve the surface hardness and scratch-resistance of a polyolefin alloy as defined according to the present invention.
- the new polyolefin alloy of the present invention can be produced by blending the components (A), (B), (C) and (D), and optionally utilized component (E); melting and preferably kneading the blend; and then cooling and granulating the obtained blend.
- the new polyolefin alloy can be produced by blending the components (A) and (B) and optionally utilized component (E); melting the blend; blending the components (C) and (D) into the melt, preferably with kneading of the blend; and then cooling and granulating the obtained blend.
- the production of the polyolefin alloy can be effected in a mixer of any suitable type, for instance a continuous or batchwise mixer, although it is preferred to use an extruder.
- Particularly suitable polypropylene materials (A) for use in the new polyolefin alloy are copolymers of propylene and ethylene.
- a particularly suitable polypropylene material is a copolymer of propylene and ethylene having a content of 5 to 25% by weight of polymerized ethylene units.
- Suitable functionalized polypropylene materials (B) for use in the new polyolefin alloy are polypropylene homopolymers and copolymers of propylene and ethylene and/or butadiene (B), grafted with a compound selected from maleic anhydride, acrylic acid, acrylates and methacrylates, vinyl silanes and other vinyl compounds. Particularly good results are obtained by grafting maleic anhydride on the polypropylene material.
- the functionalized polypropylene material can have a melt index MI in the range of 5 to 250 g/10 min at 230°C/2.16 kg, more preferably in the range of 10 to 150 g/10 min at 230°C/2.16 kg.
- Suitable minerals (C) for use in the polyolefin alloy of the invention are finely divided minerals which have been silanized with one or more of the aminosilanes traditionally used for surface treatment of talc.
- Particularly suitable minerals for use in the alloy after such silanizing treatment are wollastonite, kaolin, mica, calcium carbonate and talc, particularly talc.
- the treated mineral has a mean particle size D 50 in the range of 0.5 to 10 ⁇ m, more preferably 1-6 ⁇ m.
- the silica gel material (D) which is used in the polyolefin alloy of the invention is preferably a fatty amide-treated, amorphous, synthetic silica gel containing 40 to 65% by weight of silica and 60 to 35% by weight of fatty acid amide.
- Ethylene/propylene/polyene terpolymers that are particularly useful as an optional component in the polymer alloy of the invention are such terpolymers which have an ethylene content of 30 to 90% by weight and a flow, determined according to the Mooney method (ML 101°C), of 0 to 60, more preferably 0 to 50.
- An alloy on polypropylene basis was prepared from the following components:
- the alloy was prepared in a "Werner & Pfleiderer" double screw extruder, type ZSK, having 57 mm co-rotating screws. Before and after the preparation, the hopper was flushed with nitrogen gas in order that the preparation should take place in an approximately inert atmosphere. The screw speed was 200 rpm and the rate of extrusion was 80 kg/h. All the raw material was introduced gravimetrically at feed point 1. The temperature profile of the extruder was maintained in the range of 210 to 230°C. The components added, the amounts thereof (in % by weight of the total blend) and the results are given in the table below.
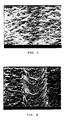
- Fig. 1 shows the appearance of a test sample of the alloy after the scratch resistance having been measured with an Ericsson pen according to ISO 1518.
- Example 2 shows the appearance of a test sample of the alloy after the scratch resistance having been measured with an Ericsson pen according to ISO 1518.
- Fig. 3 shows the appearance of a test sample of the alloy after the scratch resistance having been measured with an Ericsson pen according to ISO 1518.
- Fig. 4 shows the appearance of a test sample of the alloy after the scratch resistance having been measured with an Ericsson pen according to ISO 1518.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Laminated Bodies (AREA)
Claims (10)
- Polyolefinlegierung mit verbesserter Oberflächenhärte und Kratzfestigkeit und mit einem Schmelzindex MI im Bereich von 0,1 bis 50 g/10 min bei 230°C/2,16 kg, insbesondere im Bereich von 3 bis 40 g/10 min bei 230°C/2,16 kg, dadurch gekennzeichnet, daß sie sich zusammensetzt aus einer Mischung aus:(A) 40 bis 80 Gew. % eines Polypropylen-Materials, bestehend aus einem Polypropylen-Homopolymer oder einem Copolymer aus Propylen mit Ethylen und/oder Butadien, wobei das Polypropylen-Material einen Gehalt an polymerisierten Ethylen- und/oder Butadien-Einheiten von 5 bis 35 Mol% aufweisen kann und ein Molekulargewicht Mw von 100.000 bis 300.000 sowie einen Schmelzindex MI von 0,1 bis 20 g/10 min bei 230°C/2,16 kg aufweist,(B) 1 bis 10 Gew.% eines funktionalisierten Polypropylen-Materials, das aus einem bepfropften Polypropylen-Homopolymer oder Copolymer aus Propylen mit Ethylen und/oder Butadien besteht und einen Pfropfungsgrad von 0,2 bis 10 Gew. % aufweist,(C) 1 bis 50 Gew. % eines Minerals, das eine mittlere Teilchengröße von ungefähr 2,5 µm und eine maximale Teilchengröße von 20 µm aufweist und mit 0,2 bis 5 Gew. % Aminosilan oberflächenbehandelt wird,(D) 1 bis 15 Gew. % amorphen fettsäurebehandelten Silicagels, welches 25 bis 75 Gew. % Silicamasse und 75 bis 25 Gew. % Fettsäureamid aufweist,(E) 0 bis 40 Gew. % eines Ethylen/Propylen/Polyen-Terpolymers (EPDM) mit einem Schmelzindex MI im Bereich von 1 bis 10 g/10 min bei 230°C/2,16 kg und einer Shore-D-Härte von 30 bis 70.
- Polyolefinlegierung nach Anspruch 1, dadurch gekennzeichnet, daß das funktionalisierte Polypropylenhomopolymer oder Copolymer aus Propylen mit Ethylen und/oder Butadien (B) mit einer Verbindung aus der Gruppe umfassend Maleinsäureanhydrid, Acrylsäure, Acrylate und Methacrylate, Vinylsilane und anderen Vinylverbindungen, vorzugsweise mit Maleinsäureanhydrid, gepfropft wird.
- Polyolefinlegierung nach Anspruch 1 oder 2, dadurch gekennzeichnet, daß das Polypropylen-Material (A) ein Copolymer aus Propylen und Ethylen ist und einen Gehalt von 5 bis 25 Gew. % polymerisierte Ethyleneinheiten aufweist.
- Polyolefinlegierung nach irgend einem der Ansprüche 1 bis 3, dadurch gekennzeichnet, daß sie sich zusammensetzt aus einer Mischung aus:60 bis 80 Gew.% von (A);2,5 bis 7,5 Gew. % von (B);7,5 bis 35 Gew.% von (C);2,5 bis 7,5 Gew.% von (D);0 bis 25 Gew.% von (E).
- Polyolefinlegierung nach irgend einem der Ansprüche 1 bis 4, dadurch gekennzeichnet, daß das funktionalisierte Polypropylen-Material (B) einen Pfropfungsgrad von 0,5 bis 2 Gew. % aufweist.
- Polyolefinlegierung nach irgend einem der Ansprüche 1 bis 5, dadurch gekennzeichnet, daß das oberflächenbehandelte Mineral (C) mit ungefähr 1 Gew.% Aminosilan oberflächenbehandelt wurde.
- Polyolefinlegierung nach irgend einem der Ansprüche 1 bis 6, dadurch gekennzeichnet, daß das Mineral (C) aus der Gruppe umfassend Wollastonit, Kaolin, Glimmer, Calciumcarbonat und Talk ausgewählt wird.
- Polyolefinlegierung nach Anspruch 7, dadurch gekennzeichnet, daß das Mineral (C) Talk ist.
- Verfahren zur Herstellung einer Polyolefinlegierung nach irgend einem der Ansprüche 1 bis 8, gekennzeichnet durch die Schritte des Mischens der Komponenten (A), (B), (C) und (D) und der wahlweise verwendeten Komponente (E); des Schmelzens und vorzugsweise des Knetens der Mischung; und daraufhin des Kühlens und Granulierens der gewonnenen Mischung.
- Verfahren zur Herstellung einer Polyolefinlegierung nach einem beliebigen der Ansprüche 1 bis 8, gekennzeichnet durch die Schritte des Mischens der Komponenten (A) und (B) und der wahlweise verwendeten Komponente (E); des Schmelzens der Mischung; des Einmischens der Komponenten (C) und (D) in die Schmelze, vorzugsweise mit Kneten der Mischung; und daraufhin des Kühlens und Granulierens der gewonnenen Mischung.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| NO950160 | 1995-01-16 | ||
| NO950160A NO309384B1 (no) | 1995-01-16 | 1995-01-16 | Polyolefinlegering med forbedret overflatehårdhet og ripefasthet |
| PCT/NO1996/000003 WO1996022328A1 (en) | 1995-01-16 | 1996-01-09 | A polyolefin alloy with improved surface hardness and scratch resistance, and processes for producing same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0804503A1 EP0804503A1 (de) | 1997-11-05 |
| EP0804503B1 true EP0804503B1 (de) | 1999-03-17 |
Family
ID=19897839
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP96901147A Expired - Lifetime EP0804503B1 (de) | 1995-01-16 | 1996-01-09 | Eine polyolefinlegierung mit verbesserter oberflächenhärte und kratzfestigkeit, und verfahren zu ihrer herstellung |
Country Status (13)
| Country | Link |
|---|---|
| US (1) | US5973070A (de) |
| EP (1) | EP0804503B1 (de) |
| JP (1) | JP3162080B2 (de) |
| KR (1) | KR100239816B1 (de) |
| CN (1) | CN1168148A (de) |
| AR (1) | AR000661A1 (de) |
| AT (1) | ATE177772T1 (de) |
| AU (1) | AU4497696A (de) |
| BR (1) | BR9606915A (de) |
| DE (1) | DE69601780T2 (de) |
| ES (1) | ES2128831T3 (de) |
| NO (1) | NO309384B1 (de) |
| WO (1) | WO1996022328A1 (de) |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR970070085A (ko) * | 1996-04-29 | 1997-11-07 | 황선두 | 웰드강도가 향상된 폴리프로필렌 수지 조성물 |
| JP2002514244A (ja) * | 1996-12-13 | 2002-05-14 | デュポン ダウ エラストマーズ エルエルシー | 耐摩耗性シラン−架橋性ポリマーおよびポリマーブレンド組成物 |
| DE19817257A1 (de) | 1998-04-19 | 1999-12-09 | Grace Gmbh | Granulatzusammensetzung aus Antiblockingmitteln und Additiven für Polymerherstellung |
| US6177515B1 (en) * | 1998-12-17 | 2001-01-23 | Montell Technology Company Bv | Polypropylene graft copolymers with improved scratch and mar resistance |
| KR100578167B1 (ko) * | 1999-12-31 | 2006-05-12 | 현대자동차주식회사 | 폴리올레핀계 복합수지 조성물 |
| US6605656B2 (en) | 2000-11-29 | 2003-08-12 | Visteon Global Technologies, Inc. | Surface properties in thermoplastic olefin alloys |
| JP4795528B2 (ja) * | 2000-11-30 | 2011-10-19 | 住友電装株式会社 | オレフィン系樹脂組成物および被覆電線 |
| JP4808840B2 (ja) | 2000-12-01 | 2011-11-02 | 住友電装株式会社 | オレフィン系樹脂組成物および被覆電線 |
| EP1223588A1 (de) * | 2001-01-15 | 2002-07-17 | Sumitomo Wiring Systems, Ltd. | Zusammensetzung auf der Basis von Olefinharzen |
| US6414068B1 (en) | 2001-01-16 | 2002-07-02 | Sumitomo Wiring Systems, Ltd. | Olefin-based resin composition |
| WO2003008497A1 (en) * | 2001-07-12 | 2003-01-30 | Idemitsu Petrochemical Co., Ltd. | Polyolefin resin composition |
| EP1295910A1 (de) * | 2001-09-25 | 2003-03-26 | Borealis GmbH | Isolierschaumzusammensetzung |
| KR100925349B1 (ko) * | 2002-12-16 | 2009-11-09 | 다케모토 유시 가부시키 가이샤 | 합성 고분자 필름용 표면처리제, 합성 고분자 필름의표면처리방법 및 표면처리 합성 고분자 필름 |
| DE10305557B4 (de) * | 2003-02-10 | 2007-04-26 | Benefit Gmbh | Zusammensetzung zur Verstärkung von Thermoplasten mittels eines Additivs und dessen Herstellungsverfahren |
| JP4530657B2 (ja) * | 2003-12-22 | 2010-08-25 | 出光興産株式会社 | 軟質ポリオレフィン系樹脂の造粒方法及び造粒物 |
| US7390574B2 (en) * | 2004-07-07 | 2008-06-24 | Ciba Specialty Chemicals Corporation | Scratch resistant polyolefins |
| EP1888686B1 (de) * | 2005-06-07 | 2013-01-23 | Basf Se | Kratzfeste polyolefine |
| JP2009509016A (ja) * | 2005-09-22 | 2009-03-05 | チバ ホールディング インコーポレーテッド | 耐引掻性ポリマー及びコーティング組成物 |
| US7462670B2 (en) | 2005-09-22 | 2008-12-09 | Ciba Specialty Chemicals Corporation | Scratch resistant polymer compositions |
| US7790795B2 (en) * | 2006-05-25 | 2010-09-07 | Exxonmobil Chemical Patents Inc. | Scratch and mar resistant polymer compositions, methods for making and articles made from the same |
| EP2305751A1 (de) | 2009-10-01 | 2011-04-06 | Borealis AG | Mehrschichtiger Artikel |
| BRPI0904397A2 (pt) | 2009-10-07 | 2011-06-14 | Braskem Sa | processo via extrusço para preparar uma composiÇço polimÉrica hÍbrida, composiÇço polimÉrica hÍbrida e artigo |
| CN102127263B (zh) * | 2011-01-27 | 2013-01-23 | 中国汽车工程研究院股份有限公司 | 一种汽车用釜内合金聚丙烯材料的改性方法 |
| CN109401057B (zh) * | 2018-11-01 | 2021-12-14 | 常州工程职业技术学院 | 一种表面耐划伤聚丙烯及其制备方法 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5723642A (en) * | 1980-07-17 | 1982-02-06 | Mitsubishi Petrochem Co Ltd | Olefinic polymer composition containing inorganic filler |
| DE3337356A1 (de) * | 1983-10-14 | 1985-04-25 | Grace Gmbh, 2000 Norderstedt | Kombiniertes antiblocking- und gleitmittelkonzentrat |
| US4751262A (en) * | 1985-09-03 | 1988-06-14 | The Dow Chemical Company | Ethylene-acrylic acid type interpolymer compositions and films having increased slip and reduced block |
| JPS6356546A (ja) * | 1986-08-26 | 1988-03-11 | Sumitomo Chem Co Ltd | 剥離性保護フイルム |
| JPS6395252A (ja) * | 1986-10-13 | 1988-04-26 | Mitsui Toatsu Chem Inc | ポリプロピレン樹脂組成物 |
| EP0508415B1 (de) * | 1991-04-09 | 1996-08-28 | Nippon Petrochemicals Company, Limited | Matter Film und dessen Herstellungsverfahren |
| GB9116356D0 (en) * | 1991-07-29 | 1991-09-11 | Ici Plc | Scratch resistance polymer compositions and articles |
| CN1077466A (zh) * | 1992-04-09 | 1993-10-20 | 澳大利亚卜内门军品独占有限公司 | 聚合物组合物 |
| EP0567058A3 (de) * | 1992-04-21 | 1995-02-22 | Idemitsu Petrochemical Co | Polypropylenharz Zusammensetzung. |
| US5286791A (en) * | 1992-05-29 | 1994-02-15 | Himont Incorporated | Impact modified graft copolymer compositions containing broad molecular weight distribution polypropylene |
| GB9302069D0 (en) * | 1993-02-03 | 1993-03-24 | Ici Plc | Scratch resistant polymer compositions |
-
1995
- 1995-01-16 NO NO950160A patent/NO309384B1/no not_active IP Right Cessation
-
1996
- 1996-01-09 JP JP52218096A patent/JP3162080B2/ja not_active Expired - Fee Related
- 1996-01-09 BR BR9606915A patent/BR9606915A/pt not_active IP Right Cessation
- 1996-01-09 KR KR1019970704825A patent/KR100239816B1/ko not_active Expired - Fee Related
- 1996-01-09 AT AT96901147T patent/ATE177772T1/de not_active IP Right Cessation
- 1996-01-09 AU AU44976/96A patent/AU4497696A/en not_active Abandoned
- 1996-01-09 DE DE69601780T patent/DE69601780T2/de not_active Expired - Fee Related
- 1996-01-09 WO PCT/NO1996/000003 patent/WO1996022328A1/en not_active Ceased
- 1996-01-09 EP EP96901147A patent/EP0804503B1/de not_active Expired - Lifetime
- 1996-01-09 ES ES96901147T patent/ES2128831T3/es not_active Expired - Lifetime
- 1996-01-09 CN CN96191469A patent/CN1168148A/zh active Pending
- 1996-01-09 US US08/860,977 patent/US5973070A/en not_active Expired - Fee Related
- 1996-01-10 AR AR33498996A patent/AR000661A1/es unknown
Also Published As
| Publication number | Publication date |
|---|---|
| AU4497696A (en) | 1996-08-07 |
| JP3162080B2 (ja) | 2001-04-25 |
| JPH11511768A (ja) | 1999-10-12 |
| ATE177772T1 (de) | 1999-04-15 |
| EP0804503A1 (de) | 1997-11-05 |
| AR000661A1 (es) | 1997-07-10 |
| KR100239816B1 (ko) | 2000-01-15 |
| DE69601780T2 (de) | 1999-10-21 |
| ES2128831T3 (es) | 1999-05-16 |
| US5973070A (en) | 1999-10-26 |
| WO1996022328A1 (en) | 1996-07-25 |
| NO950160L (no) | 1996-07-17 |
| NO309384B1 (no) | 2001-01-22 |
| BR9606915A (pt) | 1997-11-11 |
| CN1168148A (zh) | 1997-12-17 |
| KR19980701435A (ko) | 1998-05-15 |
| DE69601780D1 (de) | 1999-04-22 |
| NO950160D0 (no) | 1995-01-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0804503B1 (de) | Eine polyolefinlegierung mit verbesserter oberflächenhärte und kratzfestigkeit, und verfahren zu ihrer herstellung | |
| KR860000770B1 (ko) | 폴리프로필렌수지 조성물 | |
| CA1156787A (en) | Polybutylene modified masterbatches for impact resistant polypropylene | |
| EP0578043B1 (de) | Glasfaserverstärkte Propylenpfropfpolymerzusammensetzung | |
| US5286791A (en) | Impact modified graft copolymer compositions containing broad molecular weight distribution polypropylene | |
| CA1165928A (en) | Molding resins based on blends of acid copolymer/linear polyolefin/reinforcing fiber | |
| CA2024058C (en) | Moldable polyblends of polyolefins and styrenic resins | |
| EP0204400B1 (de) | Polypropylenzusammensetzung | |
| EP1034200B1 (de) | Nicht agglomerierende zähigkeitsvermittler für polyamide | |
| EP0748354A1 (de) | Weiche, polare, thermoplastische polyolefinzusammensetzungen | |
| WO2007139622A2 (en) | Scratch and mar resistant polymer compositions, methods for making and articles made from the same | |
| JP2915142B2 (ja) | 低粘度を有する熱可塑性オレフィン | |
| EP1362079B1 (de) | Polypropylenharzzusammensetzung mit verbesserter oberflächenhärte und kratzfestigkeit | |
| EP0856555A1 (de) | Propylen Harzmischung | |
| EP0365234B1 (de) | Verfahren zur Herstellung von ethylenischen Pfropfcopolymeren | |
| JPH05194804A (ja) | 強化ポリプロピレン成形用組成物 | |
| WO1996037553A1 (en) | Vinyl chloride resin compositions | |
| JP3338247B2 (ja) | 熱可塑性重合体組成物 | |
| JP3323413B2 (ja) | 熱可塑性樹脂組成物 | |
| JP2943252B2 (ja) | 樹脂組成物 | |
| JP3240581B2 (ja) | 射出成形用ポリプロピレン | |
| JPH0517642A (ja) | ポリプロピレン系樹脂組成物 | |
| JPH08319390A (ja) | 繊維強化塩化ビニル系樹脂組成物 | |
| EP0615982A2 (de) | Modifizierte Alpha-Olefin-Polymere mit tertiären Kohlenstoffen enthaltenden Seitenketten und Herstellung von gestreckten Filmen | |
| KR970003307B1 (ko) | 필-러 함유 폴리올레핀 수지 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19970808 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL PT SE |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| 17Q | First examination report despatched |
Effective date: 19971223 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| ITF | It: translation for a ep patent filed | ||
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL PT SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19990317 Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990317 Ref country code: CH Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19990317 |
|
| REF | Corresponds to: |
Ref document number: 177772 Country of ref document: AT Date of ref document: 19990415 Kind code of ref document: T |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 69601780 Country of ref document: DE Date of ref document: 19990422 |
|
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2128831 Country of ref document: ES Kind code of ref document: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19990617 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 19990525 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000109 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000109 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 20000131 |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PT Payment date: 20081223 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20090122 Year of fee payment: 14 Ref country code: AT Payment date: 20090115 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20090114 Year of fee payment: 14 Ref country code: DE Payment date: 20090122 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20090122 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20090219 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20090114 Year of fee payment: 14 Ref country code: IT Payment date: 20090128 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20090115 Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: MM4A Free format text: LAPSE DUE TO NON-PAYMENT OF FEES Effective date: 20100709 |
|
| BERE | Be: lapsed |
Owner name: *BOREALIS A/S Effective date: 20100131 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20100801 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20100109 |
|
| EUG | Se: european patent has lapsed | ||
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20100930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100801 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100803 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100109 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100109 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100709 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100131 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100109 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20110331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110321 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100110 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100110 |