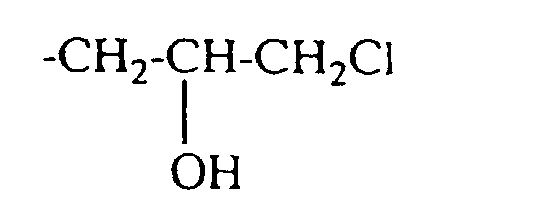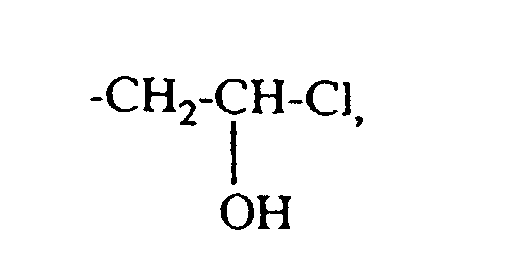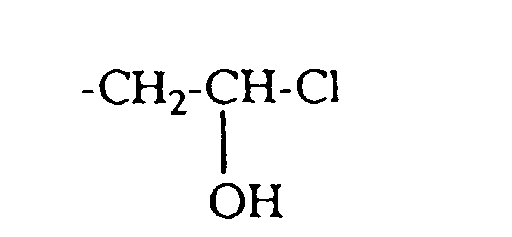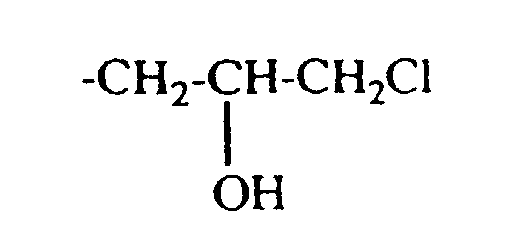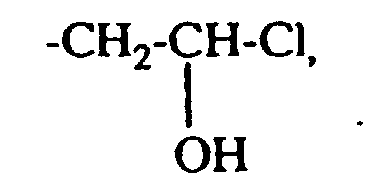EP0442844B2 - Verfahren zum Färben von Wolle mit Reaktivfarbstoffen - Google Patents
Verfahren zum Färben von Wolle mit Reaktivfarbstoffen Download PDFInfo
- Publication number
- EP0442844B2 EP0442844B2 EP91810078A EP91810078A EP0442844B2 EP 0442844 B2 EP0442844 B2 EP 0442844B2 EP 91810078 A EP91810078 A EP 91810078A EP 91810078 A EP91810078 A EP 91810078A EP 0442844 B2 EP0442844 B2 EP 0442844B2
- Authority
- EP
- European Patent Office
- Prior art keywords
- component
- formula
- hydrogen
- compound
- process according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P3/00—Special processes of dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form, classified according to the material treated
- D06P3/02—Material containing basic nitrogen
- D06P3/04—Material containing basic nitrogen containing amide groups
- D06P3/14—Wool
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/44—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders
- D06P1/60—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders using compositions containing polyethers
- D06P1/607—Nitrogen-containing polyethers or their quaternary derivatives
- D06P1/6076—Nitrogen-containing polyethers or their quaternary derivatives addition products of amines and alkylene oxides or oxiranes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P3/00—Special processes of dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form, classified according to the material treated
- D06P3/02—Material containing basic nitrogen
- D06P3/04—Material containing basic nitrogen containing amide groups
- D06P3/14—Wool
- D06P3/148—Wool using reactive dyes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S516/00—Colloid systems and wetting agents; subcombinations thereof; processes of
- Y10S516/01—Wetting, emulsifying, dispersing, or stabilizing agents
- Y10S516/07—Organic amine, amide, or n-base containing
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S8/00—Bleaching and dyeing; fluid treatment and chemical modification of textiles and fibers
- Y10S8/916—Natural fiber dyeing
- Y10S8/917—Wool or silk
Definitions
- the present invention relates to a new method for fiber and flat level dyeing of wool with reactive dyes, the material dyed according to the new process and a means of carrying out the Procedure.
- the present method is particularly suitable for producing light to medium shades.
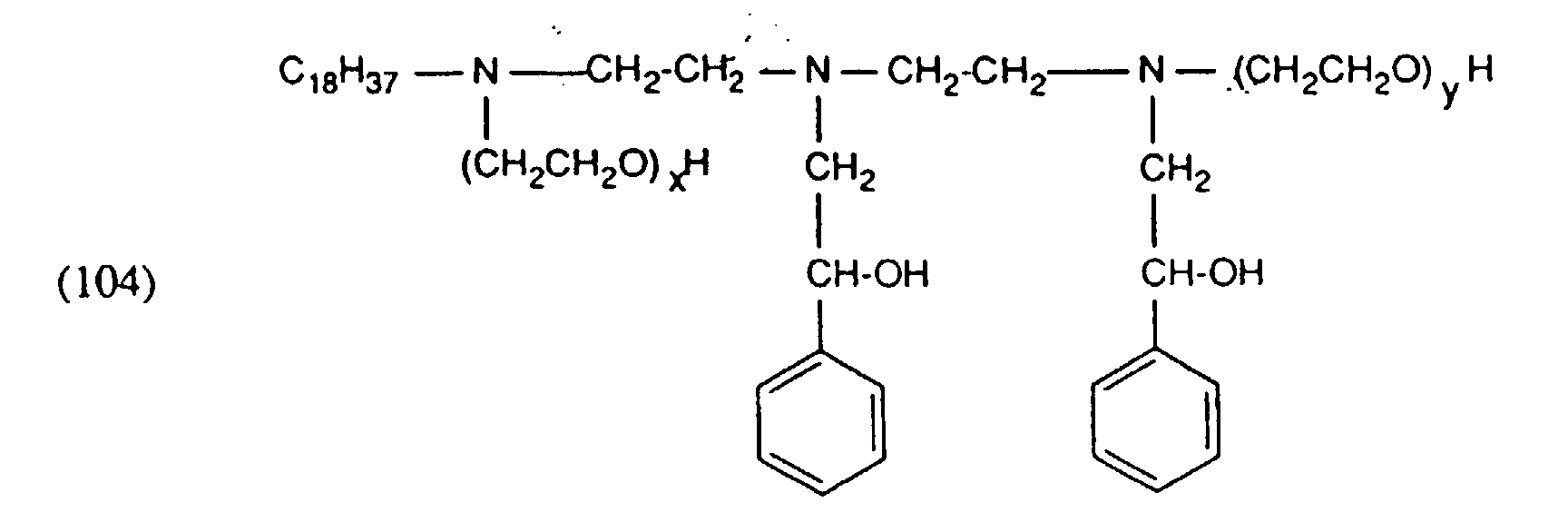
- auxiliary mixture as component (c) can be a nonionic compound of the formula wherein R "is an alkyl or alkenyl radical having 12 to 22 carbon atoms and x and y are integers, the sum of x and y being 80 to 140.
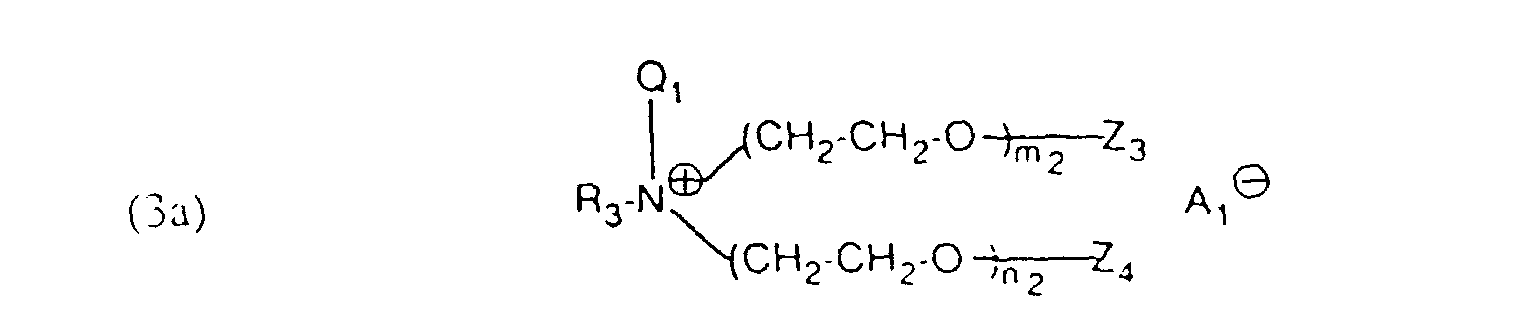
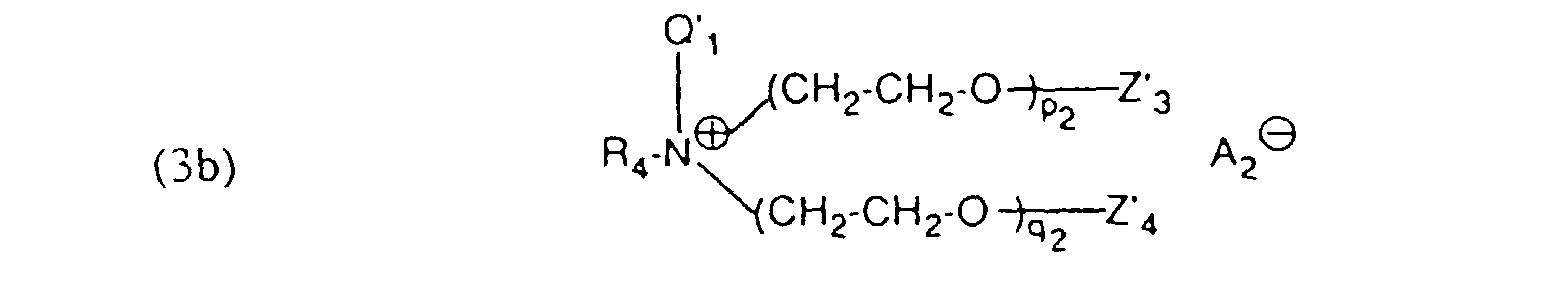
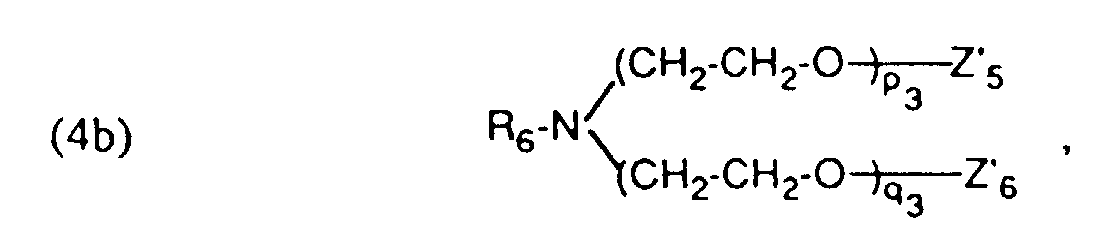
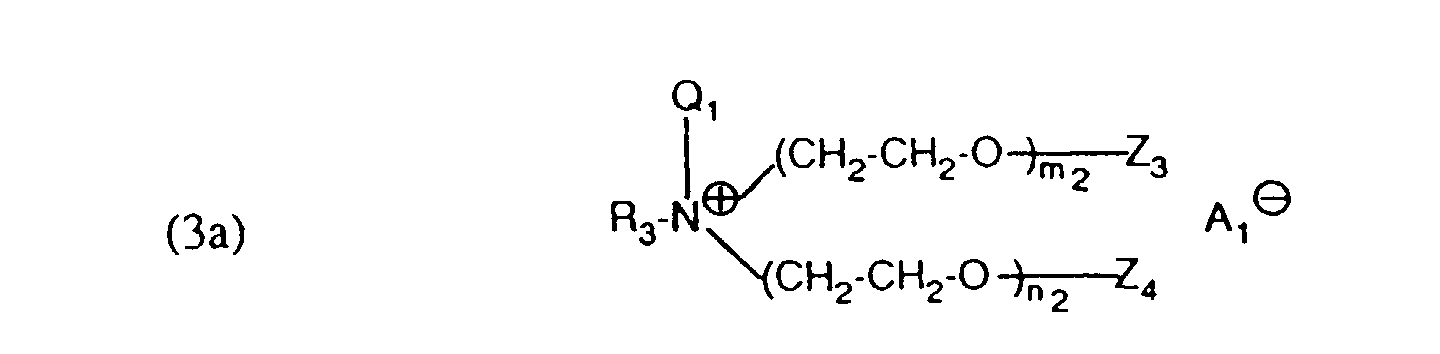
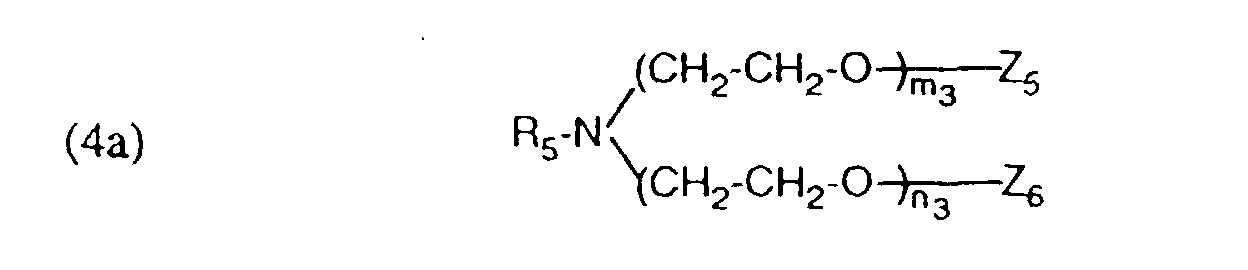
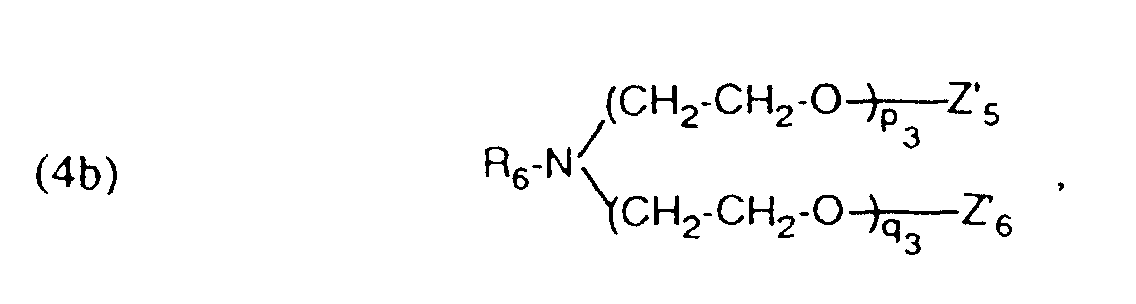
- auxiliary mixtures which, as component (a), are a compound of the formula (3a) and as component (b) a compound of the formula (4b) or auxiliary mixtures which, as component (a) contain a compound of formula (3b) and as component (b) a compound of formula (4a).
- M in the formulas (1), (3) and (4) denotes hydrogen, alkali metal, such as sodium, potassium and in particular ammonium.
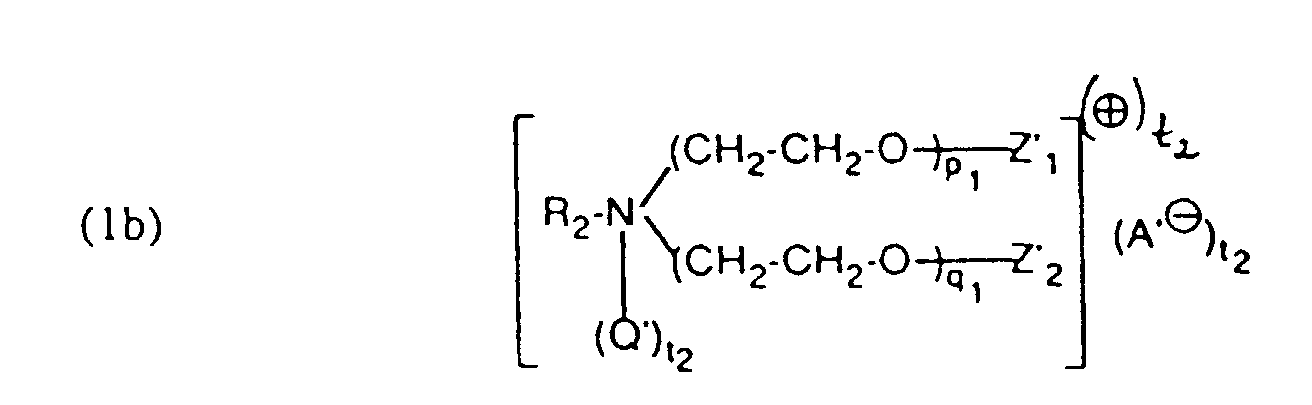
- the residues Q. Q ', Q 1 and Q' 1 and A ⁇ , A ' ⁇ , A 1 ⁇ and A 2 ⁇ in formulas (1) and (3) are derived from quaternizing agents , QC 1 to C 4 -Alkyl, -CH 2 -CO-NH 2 . or is.
- quaternizing agents are e.g. Acetyl bromide, ethyl bromide, ethylene chlorohydrin, Ethylene bromohydrin, epichlorohydrin, epibromohydrin, dimethyl sulfate, diethyl sulfate and in particular Chloroacetamide into consideration.
- the aliphatic radicals R 1 , R 2 , R 3 , R 4 , R 5 and R 6 in the formulas (1), (3) and (4) are alkyl or alkenyl radicals having 12 to 24, preferably 16 to 22, carbon atoms Consideration.
- the following may be mentioned as examples: the n-dodecyl, myristyl, n-hexadecyl, n-heptadecyl, n-octadecyl, arachidyl, behenyl, dodecenyl, hexadecenyl, oleyl and octadecenyl radical.
- the compounds of components (a), (b) and (c) are e.g. (in part) from US-A-4,444,564 known.
- the compounds of component (a) according to formula (1a) are prepared by 2 to 15 moles of ethylene oxide to aliphatic amines, which is an aliphatic radical having 12 to 24 carbon atoms have accumulated and optionally the adduct in the acidic monoester and optionally the acidic monoester converted into the alkali or ammonium salts or the adduct one of the quaternizing agents mentioned above.
- the compounds of component (b) accordingly Formula (1b) are prepared by adding 25 to 200 moles of ethylene oxide to aliphatic amines which have an aliphatic radical having 12 to 24 carbon atoms, attached and optionally that Addition product in the acidic ester and optionally the acidic ester in the alkali or ammonium salts transferred or the adduct with one of the ouating agents mentioned above implements.
- the compounds of formula (2) are prepared by adding 80 to 140 moles of ethylene oxide to a compound of formula where R "has the meaning given under formula (2).
- the amines which are required as starting materials for the preparation of the compounds of the formulas (1), (3) and (4) can have saturated or unsaturated, branched or unbranched hydrocarbon radicals having 12 to 24, preferably 16 to 22, carbon atoms.
- the amines can be chemically uniform or in the form of mixtures.
- the amine mixtures used are preferably those which arise when natural fats or oils, such as tallow fat, soybean or coconut oil, are converted into the corresponding amines.
- Dodecylamine, hexadecylamine, octadecylamine, arachidylamine, behenylamine and octadecenylamine may be mentioned as amines.
- Tallow fatty amine is a mixture of approx. 30% hexadecylamine, 25% octadecylamine and 45% octadecenylamine.
- Both the ethylene oxide addition and the esterification can be carried out according to methods known per se be performed.
- sulfuric acid or its functional derivatives such as e.g. Chlorosulfonic acid and especially sulfamic acid are used.
- the esterification is usually carried out by simply mixing the reactants with heating, expediently carried out at a temperature between 50 and 100 ° C.
- the free acids can then be converted into the alkali metal or ammonium salts by the usual way Bases such as Ammonia, sodium or potassium hydroxide can be added.
- an auxiliary mixture which 10 to 80 parts, preferably 20 to 70 parts of component (a), 5 to 70 parts, preferably 5 to 50 Parts of component (b) and 0 to 70, preferably 0 to 50 parts of the compound of component (c) and contains ad 100 parts of water.
- auxiliary mixture consists of components (a), (b) and optionally (c) is added to the dyebath, range between 0.5 and 4 percent by weight, based on the fiber material to be dyed. Preferably 1 to 2 percent by weight of the Auxiliary mixture, based on the fiber material.
- the weight ratio of components (a) and (b) is between 1: 5 and 10: 1, preferably between 1: 2 and 5: 1.
- Wool can be considered as the fiber material that can be dyed according to the invention.
- the fiber material can be in various forms. Examples include: Flake, sliver, yarn, fabric, knitwear or carpets.
- the wool can be normal or felt-free be equipped.
- This class of dyes is referred to in the Color Index 3rd edition 1971 as "reactive dyes”. It deals mainly dyes that contain at least one with polyhydroxy fibers (cellulose fibers) or polyamide fibers, especially wool, reactive group, a precursor for this or one with Contain polyhydroxyl fibers or polyamide fibers reactive substituents.
- Suitable bases for the reactive dyes are, in particular, those from the series of mono-, dis- or Polyazo dyes including the formazan dyes and the anthraquinone, xanthene, nitro, Triphenylmethane, naphthoquinoneimine, dioxazine and phthalocyanine dyes.
- being the azo and phthalo cyanine dyes can be both metal-free and metal-containing.
- reactive groups and precursors which form such reactive groups are Epoxy groups, the ethylene imide group, the vinyl grouping in vinyl sulfone or in acrylic acid residue and the ⁇ -sulfatoethylsulfone group, the ⁇ -chloroethylsuffone group or the ⁇ -dialkylaminoethylsulfone group called.
- the reactive substituents in reactive dyes are those which are easily removable and leave an electrophilic residue.
- substituents are 1 or 2 halogen atoms in an aliphatic one Acyl residue e.g. in the ⁇ -position or ⁇ - and ⁇ -position of a propionyl radical or in the ⁇ - and / or ⁇ -position of an acrylic acid residue. or 1 to 3 halogen atoms on the following ring system: pyridazine, Pyrimidine, pyridazone, triazine, quinoxaline or phthalazine.
- Dyes with two or more identical or different reactive groups can also be used be used.
- Preferred reactive dyes contain chloroacetyl, bromoacrylic or as reactive substituents Dibromopropionyl.
- the reactive dyes can be acidic, salt-forming substituents, e.g. Carboxylic acid groups, sulfuric acid and phosphonic acid ester groups, phosphonic acid groups or preferably sulfonic acid groups contain.
- Reactive dyes with at least one sulfonic acid group are preferred with an azo or anthraquinone base, which preferably has two to three sulfonic acid groups having.
- Mixtures of reactive dyes can also be used, with bichromatic or trichromatic dyeings can be generated.
- the coloring takes place after the pull-out procedure.
- the amount of dyes added to the dye liquor depends on the desired color strength. Generally, amounts are from 0.01 to 10 Percent by weight, preferably 0.01 to 2 percent by weight, based on the fiber material used, proven.
- the liquor ratio can be chosen within a wide range e.g. 1: 3 to 1: 100, preferably 1: 8 to 1:30.
- the dye baths can contain mineral acids, e.g. Sulfuric acid or phosphoric acid, organic acids, expediently aliphatic carboxylic acids such as formic acid, acetic acid, oxalic acid or citric acid and / or salts such as ammonium acetate, ammonium sulfate or sodium acetate.
- the acids serve especially the adjustment of the pH of the liquors used according to the invention, which is between 4 and 5 lies.
- the dye liquors can also contain other additives, e.g. Wool protection, dispersing and wetting agents and also contain defoamers.
- Special devices are not required in the method according to the invention. They can usual dyeing machines, such as, for example, open baths, worsted, stranded yarn or packers, Jigger, paddle, tree dyeing, circulation or jet dyeing or reel skids be used.
- dyeing machines such as, for example, open baths, worsted, stranded yarn or packers, Jigger, paddle, tree dyeing, circulation or jet dyeing or reel skids be used.
- the dyeing is advantageously carried out at a temperature in the range from 60 to 120 ° C., preferably 70 to 105 ° C.
- the dyeing time is within the usual range and is usually 20 to 120 minutes.
- the dyeing process can be followed by an alkaline after-treatment, e.g. With aqueous ammonia, alkali metal hydroxides, alkali metal carbonates. -hydrogen carbonates or hexamethylenetetraamine be connected.
- the pH of the dye baths containing alkali is expediently 7.5 to 9, preferably 8 to 8.5.
- the dyeing of the fiber material is expediently carried out in such a way that the material to be dyed with a aqueous liquor containing the acid, the auxiliary mixture of components (a) and (b) and, if appropriate contains component (c) and has a temperature of 30 to 60 ° C, briefly treated and the the same bath adds the reactive dye. Then you slowly increase the temperature to in one Color range from 80 to 100 ° C and for 20 to 90 minutes, preferably 30 to 60 minutes.
- the dyed material is then, if necessary, after the addition of alkalis, preferably sodium hydrogen carbonate or treated with sodium carbonate for 10 to 20 minutes at 70 to 90 ° C. In the end it will pulled out colored material and rinsed, acidified and dried as usual.
- Fiber and surface level dyeings are obtained in particular by the dyeing process according to the invention in light to medium shades with good light and wet fastness properties.
- the pH of the liquor is 4.5.
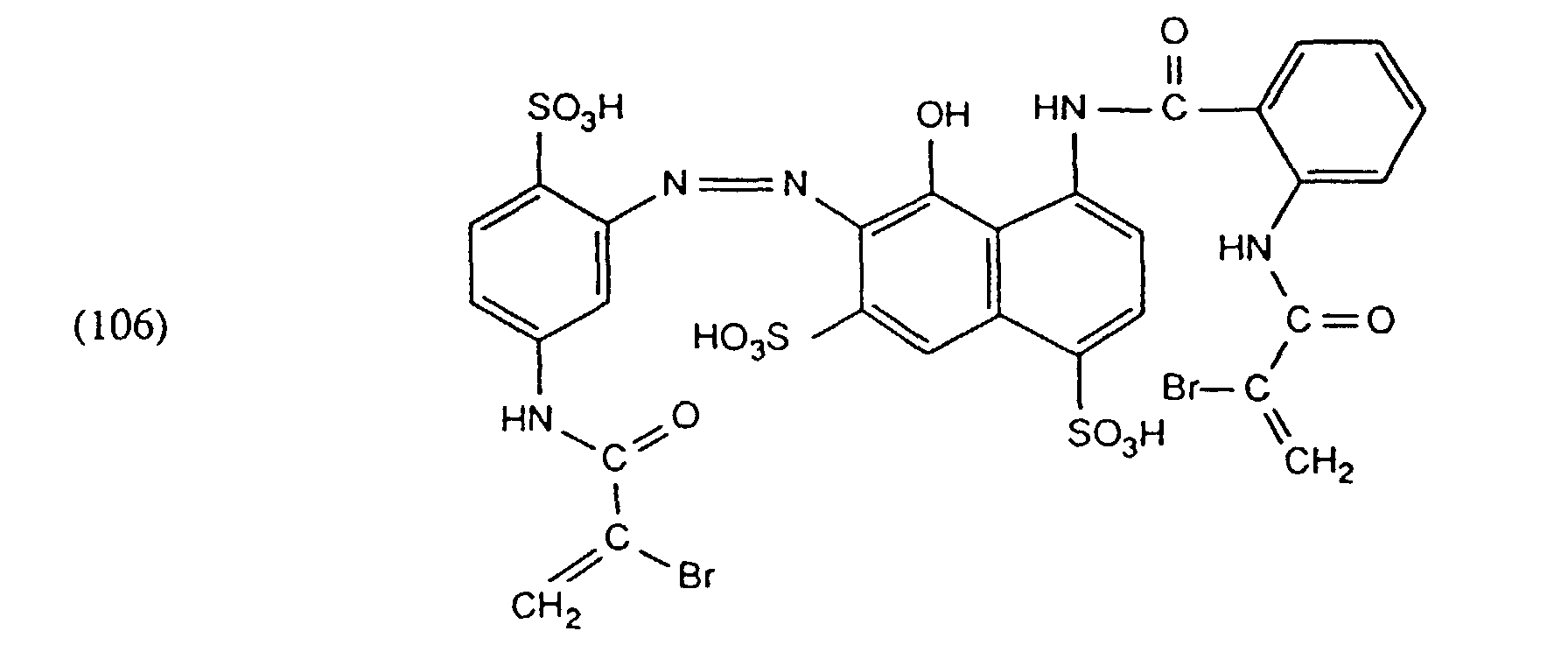
- the 12 mg of the dye of the formula 24 mg of the dye of the formula and 44 mg of the dye of the formula contains, the dyebath is kept at 40 ° for about 5 minutes and then heated to 60 ° at a rate of 1 ° / min and held at 60 ° for 20 minutes. The mixture is then heated to 98 ° at 1 ° / min and dyed for 30 minutes. After the liquor has cooled to 70 °, rinsing is carried out as usual. A fiber- and flat-level dyeing with good fastness properties is obtained.
- EXAMPLE 3 The procedure is as described in Example 1, with the difference that 0.2 g of the adduct of 34 mol of ethylene oxide and 1 mol of a C 20 -C 22 fatty amine is used instead of auxiliary B 2 . A fiber- and flat-level dyeing with good fastness properties is obtained.
- EXAMPLE 4 The procedure is as described in Example 1, with the difference that 0.2 g of the quaternized adduct of 34 mol of ethylene oxide and 1 mol of a C 20 -C 22 fatty amine is used instead of auxiliary B 2 . A fiber- and flat-level dyeing with good fastness properties is obtained.
- EXAMPLE 5 The procedure is as described in Example 1, with the difference that 0.2 g of the adduct of 30 mol of ethylene oxide and 1 mol of tallow fatty amine is used instead of B 2 . A fiber- and flat-level dyeing with good fastness properties is obtained.
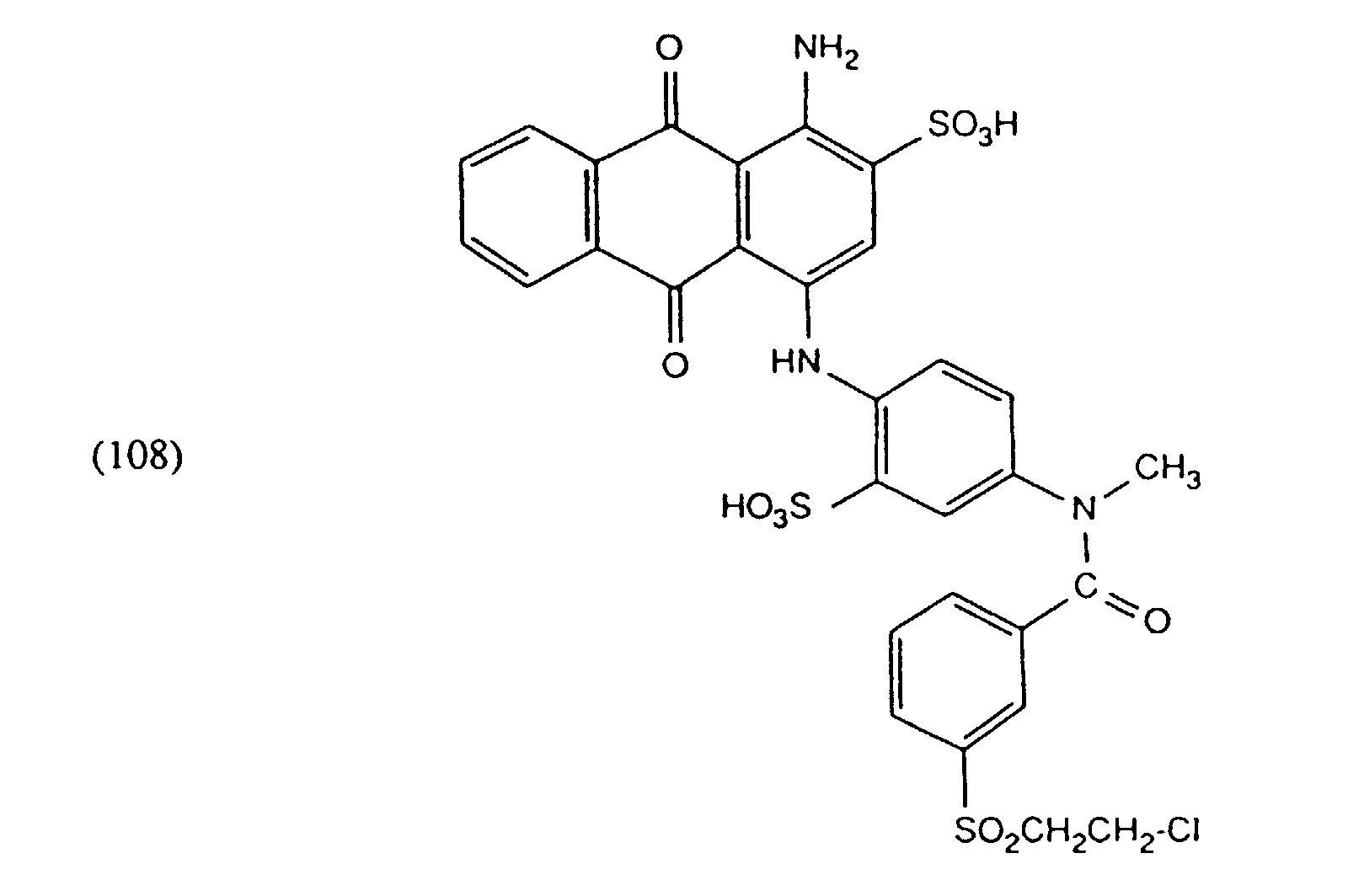
- EXAMPLE 9 The procedure is as described in Example 2, with the difference that, instead of the dye mixture, 80 mg of the dye of the formula is used.
- Example 10 One proceeds as described in Example 9 with the difference. that instead of 80 mg of the dye of the formula (105), 80 mg of the dye of the formula starts.
- EXAMPLE 11 The procedure is as described in Example 9, with the difference that instead of 80 mg of the dye of the formula (105), 200 mg of the dye of the formula used.
- EXAMPLE 12 The procedure is as described in Example 9, with the difference that instead of 80 mg of the dye of the formula (105), 320 mg of the dye of the formula used.
- EXAMPLE 13 The procedure is as described in Example 9, with the difference that instead of 80 mg of the dye of the formula (105), 100 mg of the dye of the formula used.
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Coloring (AREA)
Description
Claims (14)
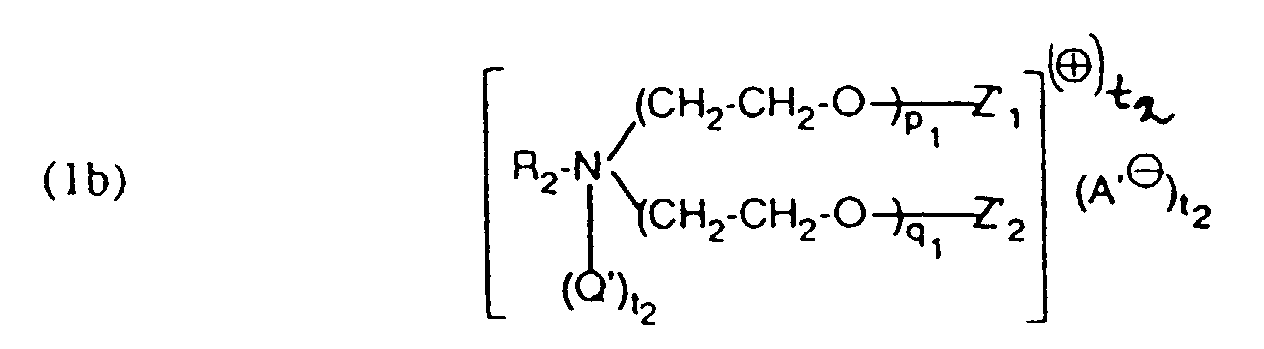
- Verfahren zum faser- und flächenegalen Färben von Wolle mit Reaktivfarbstoffen in Gegenwart eines Hilfsmittelgemisches dadurch gekennzeichnet, dass man zum Färben dieser Materialien eine wässrige Flotte verwendet, welche mindestens einen Reaktivfarbstoff und ein Hilfsmittelgemisch enthaltend als Komponente (a) mindestens eine Verbindung der Formel und als Komponente (b) mindestens eine Verbindung der Formel enthält, worinR1 und R2 unabhängig voneinander einen aliphatischen Rest mit 12 bis 24 C-Atomen, Q und Q' unabhängig voneinander C1 bis C4-Alkyl, -CH2-CO-NH2, oderA⊖ und A'⊖ ein Anion.Z1, Z2, Z'1 und Z'2 unabhängig voneinander Wasserstoff, SO3M oder PO3M, wobei M Wasserstoff, Alkalimetall oder Ammonium, t1 0 oder 1 und t2 0 oder 1 bedeuten, wobei wenn t1 0 ist, Z1 und Z2 Wasserstoff oder von Z1 und Z2 der eine Rest Wasserstoff und der andere SO3M oder PO3M bedeutet, und wenn t2 0 ist, Z'1 und Z'2 Wasserstoff oder von Z'1 und Z'2 der eine Rest Wasserstoff und der andere SO3M oder PO3M bedeutet, m1, n1, p1 und q1 ganze Zahlen bedeuten, wobei die Summe von (m1 + n1) 2 bis 15 und (p1 + q1) 25 bis 200 ist, und die Färbung unabhängig von der Farbtiefe bei einem pH-Wert von 4,0 bis 5,0 fertigstellt.
- Verfahren gemäss Anspruch 1, dadurch gekennzeichnet, dass Wolle in hellen bis mittleren Farbtönen gefärbt wird.
- Verfahren gemäss einem der Ansprüche 1 oder 2, dadurch gekennzeichnet, dass die Summen (m1 + n1) 5 bis 12 und (p1 + q1) 25 bis 100 bedeuten.
- Verfahren gemäss einem der Ansprüche 1 bis 3, dadurch gekennzeichnet, dass das Hilfsmittelgemisch zusätzlich als Komponente (c) eine nichtionogene Verbindung der Formel worin R" einen Alkyl- oder Alkenylrest mit 12 bis 22 Kohlenstoffatomen und x und y ganze Zahlen bedeuten, wobei die Summe von x und y 80 bis 140 ist, enthält.
- Verfahren gemäss einem der Ansprüche 1 bis 4, dadurch gekennzeichnet, dass ein Hilfsmittelgemisch enthaltend als Komponente (a) eine Verbindung der Formel als Komponente (b) eine Verbindung der Formel und als Komponente (c) eine Verbindung der Formel (2) verwendet wird, worin R3 und R4 unabhängig voneinander einen aliphatischen Rest mit 12 bis 24 C-Atomen,Q1 und Q'1 unabhängig voneinander C1 bis C4-Alkyl oder -CH2-CO-NH2,Z3, Z4, Z'3 und Z'4 unabhängig voneinander Wasserstoff oder SO3M,M Wasserstoff, Alkalimetall oder Ammonium,m2, n2, p2 und q2 ganze Zahlen bedeuten, wobei die Summe von (m2 + n2) 2 bis 15 und (p2 +q2) 25 bis 100 ist undA1 ⊖ und A2 ⊖ ein Anion bedeuten.
- Verfahren gemäss einem der Ansprüche 1 bis 4, dadurch gekennzeichnet, dass ein Hilfsmittelgemisch enthaltend als Komponente (a) eine Verbindung der Formel und als Komponente (b) eine Verbindung der Formel worinR5 und R6 unabhängig voneinander einen aliphatischen Rest mit 12 bis 24 C-Atomen.Z5 und Z6 Wasserstoff oder von Z5 und Z6 der eine Rest Wasserstoff und der andere SO3M bedeutet,Z'5 und Z'6 Wasserstoff oder von Z'5 und Z'6 der eine Rest Wasserstoff und der andere SO3M bedeutet,M Wasserstoff, Alkalimetall oder Ammonium undm3, n3, p3 und q3 ganze Zahlen bedeuten, wobei die Summe von (m3 + n3) 5 bis 12 und (p3 +q3) 25 bis 100 ist, verwendet wird.
- Verfahren gemäss einem der Ansprüche 1 bis 4, dadurch gekennzeichnet, dass ein Hilfsmittelgemisch enthaltend als Komponente (a) eine Verbindung der Formel (3a) gemäss Anspruch 5 und als Komponente (b) eine Verbindung der Formel (4b) gemäss Anspruch 6 verwendet wird.
- Verfahren gemäss einem der Ansprüche 1 bis 4, dadurch gekennzeichnet, dass ein Hilfsmittelgemisch enthaltend als Komponente (a) eine Verbindung der Formel (3b) gemäss Anspruch 5 und als Komponente (b) eine Verbindung der Formel (4a) gemäss Anspruch 6 verwendet wird.
- Verfahren gemäss einem der Ansprüche 1 bis 8, dadurch gekennzeichnet, das man ein Hilfsmittelgemisch enthaltendverwendet.10 bis 80 Teile der Komponente (a)5 bis 70 Teile der Komponente (b) und0 bis 70 Teile der Komponente (c)
und ad 100 Teile Wasser - Verfahren gemäss einem der Ansprüche 1 bis 9, dadurch gekennzeichnet, dass die Komponenten (a) und (b) in einem Gewichtsverhältnis zueinander von 1:5 bis 10:1 vorliegen.
- Verfahren gemäss einem der Ansprüche 1 bis 10, dadurch gekennzeichnet, dass man Reaktivfarbstoffe verwendet, die mindestens eine Sulfogruppe enthalten.
- Verfahren gemäss Anspruch 11, dadurch gekennzeichnet, dass Reaktivfarbstoffe verwendet werden, die als reaktionsfähige Substituenten Chloracetyl, Bromacetyl oder Dibrompropionyl enthalten.
- Verfahren gemäss einem der Ansprüche 1 bis 12, dadurch gekennzeichnet, dass man bei einer Temperatur zwischen 60 und 120°C färbt.
- Verfahren gemäss einem der Ansprüche 1 bis 13, dadurch gekennzeichnet, dass man bei einem Flottenverhältnis 1:3 bis 1:100, vorzugsweise 1:8 bis 1:30 färbt.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH47790 | 1990-02-14 | ||
| CH477/90 | 1990-02-14 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0442844A1 EP0442844A1 (de) | 1991-08-21 |
| EP0442844B1 EP0442844B1 (de) | 1995-02-01 |
| EP0442844B2 true EP0442844B2 (de) | 1998-01-21 |
Family
ID=4187901
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP91810078A Expired - Lifetime EP0442844B2 (de) | 1990-02-14 | 1991-02-05 | Verfahren zum Färben von Wolle mit Reaktivfarbstoffen |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US5145486A (de) |
| EP (1) | EP0442844B2 (de) |
| JP (1) | JPH04214476A (de) |
| KR (1) | KR910015749A (de) |
| AU (1) | AU638793B2 (de) |
| CA (1) | CA2036224A1 (de) |
| DE (1) | DE59104446D1 (de) |
| ES (1) | ES2067902T5 (de) |
| NZ (1) | NZ237087A (de) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0548013A1 (de) * | 1991-12-16 | 1993-06-23 | Ciba-Geigy Ag | Verfahren zum Färben von Wolle mit Hilfe einer Niedertemperaturplasma- oder Corona-Vorbehandlung |
| IT1261839B (it) * | 1992-08-03 | 1996-06-03 | Sandoz Ag | Procedimento per la produzione di effetti speciali di coloritura su prodotti tessili e miscele di coloranti impiegate. |
| ES2141211T3 (es) * | 1993-07-28 | 2000-03-16 | Ciba Sc Holding Ag | Procedimiento para el teñido de materiales de fibras que contienen lana. |
| DE19848894A1 (de) * | 1998-10-23 | 2000-04-27 | Bayer Ag | Phosphorsäureester |
| MXPA05013495A (es) * | 2003-06-26 | 2006-03-09 | Ciba Sc Holding Ag | Composiciones liquidas acuosas de ciclodextrina o derivados de ciclodextrina y proceso que usa dichas composiciones. |
| EP1777337A1 (de) * | 2005-10-07 | 2007-04-25 | Clariant International Ltd. | Verfahren zum Färben von Polyamidfasern |
| DE102008058921A1 (de) * | 2008-11-25 | 2010-05-27 | Henkel Ag & Co. Kgaa | Neue Färbemittel |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3919283A (en) * | 1965-03-18 | 1975-11-11 | Ciba Geigy Ag | Sulfate derivatives of polyglycol compounds |
| DE2847913B1 (de) * | 1978-11-04 | 1980-02-07 | Hoechst Ag | Verfahren zum Faerben von Wolle mit Reaktivfarbstoffen |
| DE3363011D1 (en) * | 1982-03-12 | 1986-05-22 | Ciba Geigy Ag | Process for dyeing fibrous material from natural polyamides |
| FR2574791A1 (fr) * | 1984-12-14 | 1986-06-20 | Sandoz Sa | Sulfates d'amines grasses, leur preparation et leur utilisation comme produits auxiliaires |
| DE3872824D1 (de) * | 1987-10-14 | 1992-08-20 | Ciba Geigy Ag | Verfahren zum faerben von natuerlichen polyamidfasern mit reaktivfarbstoffen. |
| EP0414631A1 (de) * | 1989-02-22 | 1991-02-27 | Ciba-Geigy Ag | Hilfsmittelgemisch und seine Verwendung beim Färben von Polyesterfasermaterialien |
| EP0417040A1 (de) * | 1989-09-06 | 1991-03-13 | Ciba-Geigy Ag | Verfahren zum Färben von Wolle |
-
1991
- 1991-02-05 EP EP91810078A patent/EP0442844B2/de not_active Expired - Lifetime
- 1991-02-05 DE DE59104446T patent/DE59104446D1/de not_active Expired - Fee Related
- 1991-02-05 ES ES91810078T patent/ES2067902T5/es not_active Expired - Lifetime
- 1991-02-11 US US07/654,139 patent/US5145486A/en not_active Expired - Fee Related
- 1991-02-12 NZ NZ237087A patent/NZ237087A/xx unknown
- 1991-02-12 CA CA002036224A patent/CA2036224A1/en not_active Abandoned
- 1991-02-12 KR KR1019910002313A patent/KR910015749A/ko not_active Withdrawn
- 1991-02-12 AU AU70994/91A patent/AU638793B2/en not_active Ceased
- 1991-02-14 JP JP3020240A patent/JPH04214476A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| CA2036224A1 (en) | 1991-08-15 |
| NZ237087A (en) | 1993-04-28 |
| EP0442844B1 (de) | 1995-02-01 |
| JPH04214476A (ja) | 1992-08-05 |
| ES2067902T3 (es) | 1995-04-01 |
| EP0442844A1 (de) | 1991-08-21 |
| AU638793B2 (en) | 1993-07-08 |
| ES2067902T5 (es) | 1998-04-16 |
| US5145486A (en) | 1992-09-08 |
| KR910015749A (ko) | 1991-09-30 |
| DE59104446D1 (de) | 1995-03-16 |
| AU7099491A (en) | 1991-08-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0089004B1 (de) | Verfahren zum Färben von Fasermaterial aus natürlichen Polyamiden | |
| EP0074923B1 (de) | Färbereihilfsmittel und seine Verwendung beim Färben oder Bedrucken von synthetischen Polyamidfasermaterialien | |
| EP0102926B1 (de) | Färbereihilfsmittel und seine Verwendung beim Färben oder Bedrucken von synthetischen Polyamidfasermaterialien | |
| EP0135198A2 (de) | Verfahren zum Färben von Fasermaterial aus synthetischen Polyamiden | |
| DE1940178A1 (de) | AEthansulfonsaeurederivate | |
| EP0197005B1 (de) | Maleinsäure- oder Phthalsäurehalbester von alkoxylierten Fettaminen | |
| EP0442844B2 (de) | Verfahren zum Färben von Wolle mit Reaktivfarbstoffen | |
| EP0309405B1 (de) | Verfahren zum Färben oder Bedrucken von Fasermaterial aus natürlichen oder synthetischen Polyamiden mit Reaktivfarbstoffen | |
| EP0312493B1 (de) | Verfahren zum Färben von natürlichen Polyamidfasern mit Reaktivfarbstoffen | |
| DE1924765B2 (de) | Polyoxyalkylierte Amine und deren Verwendung beim Färben mit Reaktivfarbstoffen | |
| EP0235088B1 (de) | Maleinsäure- oder Phthalsäurehalbester von alkoxylierten Fettaminen | |
| EP0745720B1 (de) | Die Verwendung von Hilfsmittelzubereitungen beim Färben von Wolle | |
| EP0163608B1 (de) | Verfahren zum Färben von Fasermaterial aus natürlichen oder synthetischen Polyamiden mit 1:1-Metallkomplexfarbstoffen | |
| EP0114574B1 (de) | Flüssige Handelsform von kationischen Farbstoffen | |
| EP0593392B1 (de) | Verfahren zum Färben von natürlichen und synthetischen Polyamidfasermaterialien mit Farbstoffmischungen | |
| EP0302013A1 (de) | Verfahren zum Färben von textilen Flächengebilden aus Polyamiden | |
| EP0474594B1 (de) | Verfahren zum Färben von Wolle und deren Mischungen mit anderen Fasern mit Reaktivfarbstoffen | |
| DE2841800C2 (de) | ||
| DE2926184A1 (de) | Neue disazofarbstoffe, deren herstellung und verwendung | |
| WO2000024749A1 (de) | Phosphorsäureester | |
| EP1162195A2 (de) | Mischungen von Schwefelsäureestern | |
| DE1963015B2 (de) | Verfahren zur kontinuierlichen Fixierung von Drucken und Klotzfarbungen in Schmelzen | |
| DE2405057C3 (de) | Verfahren zum Klotzfärben oder Bedrucken von Textilmischmaterial aus Cellulose- und Polyesterfasern | |
| DE2747699C3 (de) | Kurzflottenfärbeverfahren für Stückwaren aus Cellulosefasern in Strangform | |
| DE1619385C3 (de) | Verfahren und Mittel zum Färben von stickstoffhaltigen Textilfaser n |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19910207 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): BE CH DE ES FR GB IT LI |
|
| 17Q | First examination report despatched |
Effective date: 19930622 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE CH DE ES FR GB IT LI |
|
| ET | Fr: translation filed | ||
| REF | Corresponds to: |
Ref document number: 59104446 Country of ref document: DE Date of ref document: 19950316 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2067902 Country of ref document: ES Kind code of ref document: T3 |
|
| ITF | It: translation for a ep patent filed | ||
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19950314 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| 26 | Opposition filed |
Opponent name: BAYER AG, LEVERKUSEN KONZERNVERWALTUNG RP PATENTE Effective date: 19951027 |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| ITTA | It: last paid annual fee | ||
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: CIBA-GEIGY AG TRANSFER- CIBA SC HOLDING AG |
|
| BECN | Be: change of holder's name |
Effective date: 19961129 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 19970214 Year of fee payment: 7 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP Ref country code: CH Ref legal event code: PFA Free format text: CIBA SC HOLDING AG TRANSFER- CIBA SPECIALTY CHEMICALS HOLDING INC. |
|
| RAP2 | Party data changed (patent owner data changed or rights of a patent transferred) |
Owner name: CIBA SPECIALTY CHEMICALS HOLDING INC. |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PUAH | Patent maintained in amended form |
Free format text: ORIGINAL CODE: 0009272 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19971223 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19980108 Year of fee payment: 8 Ref country code: CH Payment date: 19980108 Year of fee payment: 8 |
|
| 27A | Patent maintained in amended form |
Effective date: 19980121 |
|
| AK | Designated contracting states |
Kind code of ref document: B2 Designated state(s): BE CH DE ES FR GB IT LI |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19980121 Year of fee payment: 8 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: AEN Free format text: AUFRECHTERHALTUNG DES PATENTES IN GEAENDERTER FORM |
|
| ITF | It: translation for a ep patent filed | ||
| GBTA | Gb: translation of amended ep patent filed (gb section 77(6)(b)/1977) | ||
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: CD |
|
| ET3 | Fr: translation filed ** decision concerning opposition | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19980403 Year of fee payment: 8 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: DC2A Kind code of ref document: T5 Effective date: 19980310 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: PC2A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990205 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990206 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990228 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990228 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990228 |
|
| BERE | Be: lapsed |
Owner name: CIBA SPECIALTY CHEMICALS HOLDING INC. Effective date: 19990228 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19990205 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19991029 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19991201 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20010503 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050205 |