CN113380552B - P掺杂FeS/Co3S4/Co9S8复合材料的制备方法及其应用 - Google Patents
P掺杂FeS/Co3S4/Co9S8复合材料的制备方法及其应用 Download PDFInfo
- Publication number
- CN113380552B CN113380552B CN202110547956.3A CN202110547956A CN113380552B CN 113380552 B CN113380552 B CN 113380552B CN 202110547956 A CN202110547956 A CN 202110547956A CN 113380552 B CN113380552 B CN 113380552B
- Authority
- CN
- China
- Prior art keywords
- fes
- doped
- composite material
- temperature
- sample
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 9
- 239000002131 composite material Substances 0.000 title claims description 37
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims abstract description 16
- DDFHBQSCUXNBSA-UHFFFAOYSA-N 5-(5-carboxythiophen-2-yl)thiophene-2-carboxylic acid Chemical compound S1C(C(=O)O)=CC=C1C1=CC=C(C(O)=O)S1 DDFHBQSCUXNBSA-UHFFFAOYSA-N 0.000 claims abstract description 12
- 229910052979 sodium sulfide Inorganic materials 0.000 claims abstract description 10
- GRVFOGOEDUUMBP-UHFFFAOYSA-N sodium sulfide (anhydrous) Chemical compound [Na+].[Na+].[S-2] GRVFOGOEDUUMBP-UHFFFAOYSA-N 0.000 claims abstract description 10
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 9
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 claims abstract description 8
- 239000004202 carbamide Substances 0.000 claims abstract description 8
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 8
- 150000001868 cobalt Chemical class 0.000 claims abstract description 7
- 239000003792 electrolyte Substances 0.000 claims abstract description 6
- KWSLGOVYXMQPPX-UHFFFAOYSA-N 5-[3-(trifluoromethyl)phenyl]-2h-tetrazole Chemical compound FC(F)(F)C1=CC=CC(C2=NNN=N2)=C1 KWSLGOVYXMQPPX-UHFFFAOYSA-N 0.000 claims abstract description 5
- 229910001379 sodium hypophosphite Inorganic materials 0.000 claims abstract description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 19
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 claims description 14
- 238000006243 chemical reaction Methods 0.000 claims description 10
- 239000008367 deionised water Substances 0.000 claims description 9
- 229910021641 deionized water Inorganic materials 0.000 claims description 9
- 238000001035 drying Methods 0.000 claims description 9
- 229910052786 argon Inorganic materials 0.000 claims description 7
- 229910001220 stainless steel Inorganic materials 0.000 claims description 7
- 239000010935 stainless steel Substances 0.000 claims description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 6
- 239000003990 capacitor Substances 0.000 claims description 4
- 229920001343 polytetrafluoroethylene Polymers 0.000 claims description 4
- 239000004810 polytetrafluoroethylene Substances 0.000 claims description 4
- -1 polytetrafluoroethylene Polymers 0.000 claims description 3
- 238000003756 stirring Methods 0.000 claims description 3
- 238000004506 ultrasonic cleaning Methods 0.000 claims description 2
- 238000007789 sealing Methods 0.000 claims 2
- 239000012300 argon atmosphere Substances 0.000 claims 1
- 238000001816 cooling Methods 0.000 claims 1
- 239000000203 mixture Substances 0.000 claims 1
- 238000004321 preservation Methods 0.000 claims 1
- 239000010453 quartz Substances 0.000 claims 1
- 150000003839 salts Chemical class 0.000 claims 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims 1
- 238000011144 upstream manufacturing Methods 0.000 claims 1
- 238000005406 washing Methods 0.000 claims 1
- 241001506047 Tremella Species 0.000 abstract description 18
- 230000014759 maintenance of location Effects 0.000 abstract description 13
- 239000002114 nanocomposite Substances 0.000 abstract description 13
- 150000002505 iron Chemical class 0.000 abstract description 5
- 239000000463 material Substances 0.000 abstract description 4
- 238000000034 method Methods 0.000 abstract description 4
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 abstract description 2
- 238000005229 chemical vapour deposition Methods 0.000 abstract description 2
- 239000002184 metal Substances 0.000 abstract description 2
- 229910052751 metal Inorganic materials 0.000 abstract description 2
- 229910052698 phosphorus Inorganic materials 0.000 abstract description 2
- 239000011574 phosphorus Substances 0.000 abstract description 2
- 230000001376 precipitating effect Effects 0.000 abstract description 2
- 239000000758 substrate Substances 0.000 abstract description 2
- 239000007772 electrode material Substances 0.000 description 10
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 8
- 238000001000 micrograph Methods 0.000 description 8
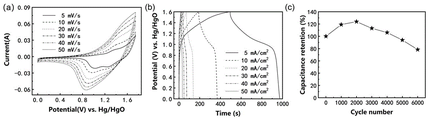
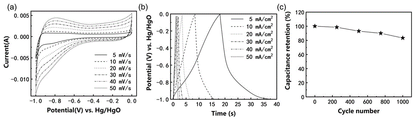
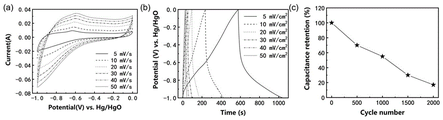
- 238000002484 cyclic voltammetry Methods 0.000 description 7
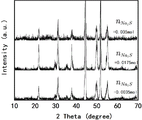
- 238000002441 X-ray diffraction Methods 0.000 description 6
- 239000007789 gas Substances 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 4
- 239000002086 nanomaterial Substances 0.000 description 4
- 239000010405 anode material Substances 0.000 description 3
- 239000003575 carbonaceous material Substances 0.000 description 3
- 239000006260 foam Substances 0.000 description 3
- 238000001027 hydrothermal synthesis Methods 0.000 description 3
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- VCJMYUPGQJHHFU-UHFFFAOYSA-N iron(3+);trinitrate Chemical compound [Fe+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O VCJMYUPGQJHHFU-UHFFFAOYSA-N 0.000 description 2
- 238000006479 redox reaction Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 241000233866 Fungi Species 0.000 description 1
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- UFMZWBIQTDUYBN-UHFFFAOYSA-N cobalt dinitrate Chemical compound [Co+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O UFMZWBIQTDUYBN-UHFFFAOYSA-N 0.000 description 1
- 229910001981 cobalt nitrate Inorganic materials 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000001351 cycling effect Effects 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 238000007599 discharging Methods 0.000 description 1
- 238000004146 energy storage Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 229910001416 lithium ion Inorganic materials 0.000 description 1
- 238000003760 magnetic stirring Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000007774 positive electrode material Substances 0.000 description 1
- 238000004073 vulcanization Methods 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G11/00—Hybrid capacitors, i.e. capacitors having different positive and negative electrodes; Electric double-layer [EDL] capacitors; Processes for the manufacture thereof or of parts thereof
- H01G11/22—Electrodes
- H01G11/30—Electrodes characterised by their material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G11/00—Hybrid capacitors, i.e. capacitors having different positive and negative electrodes; Electric double-layer [EDL] capacitors; Processes for the manufacture thereof or of parts thereof
- H01G11/22—Electrodes
- H01G11/24—Electrodes characterised by structural features of the materials making up or comprised in the electrodes, e.g. form, surface area or porosity; characterised by the structural features of powders or particles used therefor
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G11/00—Hybrid capacitors, i.e. capacitors having different positive and negative electrodes; Electric double-layer [EDL] capacitors; Processes for the manufacture thereof or of parts thereof
- H01G11/22—Electrodes
- H01G11/26—Electrodes characterised by their structure, e.g. multi-layered, porosity or surface features
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G11/00—Hybrid capacitors, i.e. capacitors having different positive and negative electrodes; Electric double-layer [EDL] capacitors; Processes for the manufacture thereof or of parts thereof
- H01G11/84—Processes for the manufacture of hybrid or EDL capacitors, or components thereof
- H01G11/86—Processes for the manufacture of hybrid or EDL capacitors, or components thereof specially adapted for electrodes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/13—Energy storage using capacitors
Landscapes
- Engineering & Computer Science (AREA)
- Power Engineering (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Chemical & Material Sciences (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Electric Double-Layer Capacitors Or The Like (AREA)
- Secondary Cells (AREA)
Abstract
Description
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110547956.3A CN113380552B (zh) | 2021-05-19 | 2021-05-19 | P掺杂FeS/Co3S4/Co9S8复合材料的制备方法及其应用 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110547956.3A CN113380552B (zh) | 2021-05-19 | 2021-05-19 | P掺杂FeS/Co3S4/Co9S8复合材料的制备方法及其应用 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113380552A CN113380552A (zh) | 2021-09-10 |
| CN113380552B true CN113380552B (zh) | 2022-10-21 |
Family
ID=77571360
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110547956.3A Active CN113380552B (zh) | 2021-05-19 | 2021-05-19 | P掺杂FeS/Co3S4/Co9S8复合材料的制备方法及其应用 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113380552B (zh) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115094476B (zh) * | 2022-07-11 | 2023-09-22 | 南京师范大学 | 一种Co9S8/Co3S4/Cu2S杂化纳米材料及其制备方法和应用 |
| CN117512656B (zh) * | 2023-09-27 | 2024-09-10 | 暨南大学 | 一种一体式金属-磷共掺杂Co9S8催化剂及其制备方法和应用 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106076377B (zh) * | 2016-06-06 | 2018-12-11 | 复旦大学 | 一种磷掺杂的硫化钴镍/碳纳米管-碳纳米纤维复合材料及其制备方法 |
| CN106340396B (zh) * | 2016-11-02 | 2018-03-09 | 信阳师范学院 | 一种泡沫镍为基底的CdCo2S4纳米结构超级电容器电极材料的制备方法 |
| CN109411239B (zh) * | 2018-08-22 | 2023-10-03 | 浙江师范大学 | 一种用于超级电容器的Cu掺杂Co9S8纳米管阵列简便制备方法 |
| CN110767466B (zh) * | 2019-10-08 | 2021-07-06 | 电子科技大学 | 一种超级电容器电极材料Ni掺杂CoP3/泡沫镍的制备方法 |
-
2021
- 2021-05-19 CN CN202110547956.3A patent/CN113380552B/zh active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN113380552A (zh) | 2021-09-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN106531456B (zh) | 一种基于CuCo2S4的超级电容器材料及其制备和应用 | |
| CN108447696B (zh) | 一种聚吡咯/导电碳布复合电极的制备方法及其应用 | |
| CN105957728B (zh) | 一种镍‑钴双氢氧化物/NiCo2S4复合纳米材料、其制备方法及作为超级电容器电极材料的应用 | |
| CN111029160B (zh) | 一种锌钴双金属硒化物纳米片电极及其制备方法 | |
| CN106601500A (zh) | 一种Ni‑Fe LDH电极材料的硫化改性方法 | |
| CN106340396B (zh) | 一种泡沫镍为基底的CdCo2S4纳米结构超级电容器电极材料的制备方法 | |
| CN109616331B (zh) | 一种核壳型的氢氧化镍纳米片/锰钴氧化物复合电极材料及其制备方法 | |
| CN113380552B (zh) | P掺杂FeS/Co3S4/Co9S8复合材料的制备方法及其应用 | |
| CN108010736A (zh) | 基于泡沫镍模板构筑NiCo2S4@Ni(OH)2@PPy材料用于超级电容器的方法 | |
| CN110098068A (zh) | 一种三元镍钴锰硫化物赝电容超级电容器材料的制备方法 | |
| CN111268745A (zh) | 一种NiMoO4@Co3O4核壳纳米复合材料、制备方法和应用 | |
| CN102874882B (zh) | 一种多孔Co3O4纳米片的制备方法 | |
| CN109962229A (zh) | 一种钼掺杂片状二硒化钴/石墨烯复合电极材料的制备方法 | |
| CN109817475B (zh) | 硫化铋镍正极材料的制备方法及其应用 | |
| CN102436936A (zh) | 双3d结构的二氧化锰薄膜电极及其制备方法 | |
| CN107146711A (zh) | 一种导电基底生长纳米层状金属化合物电极材料及其制备与应用 | |
| CN102897849A (zh) | 一种超级电容器电极材料的制备方法 | |
| CN105957723A (zh) | 一种化学气相沉积法制备硒化钴超级电容器材料的方法 | |
| CN111646510A (zh) | 高倍率钛铌氧化物微球及其制备方法和应用 | |
| CN115376835A (zh) | 一种网状铜铁属硫化物纳米薄片电极材料及其制备方法和应用 | |
| CN108448073B (zh) | 锂离子电池C@TiO2复合负极材料及其制备方法 | |
| CN107720822B (zh) | 一种海胆状锂离子电池正极材料的制备方法 | |
| CN108305790B (zh) | 一种低共熔型离子液体中制备Ni-Co-S电极材料的方法 | |
| CN110853933B (zh) | 一种基于碳布原位合成三氧化钨/五氧化二钒复合电极材料及其制备方法 | |
| CN108265283A (zh) | 低共熔型离子液体中镍基体原位硫化制备Ni3S2的方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20210910 Assignee: Hubei Yuchu Zhuxing Building Materials Co.,Ltd. Assignor: CHINA THREE GORGES University Contract record no.: X2023980054569 Denomination of invention: Preparation method and application of P-doped FeS/Co3S4/Co9S8composite materials Granted publication date: 20221021 License type: Common License Record date: 20240103 |
|
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20210910 Assignee: Hubei Fengwei Project Management Co.,Ltd. Assignor: CHINA THREE GORGES University Contract record no.: X2025980003464 Denomination of invention: Preparation method and application of P-doped FeS/Co3S4/Co9S8composite materials Granted publication date: 20221021 License type: Common License Record date: 20250219 |