CN100357312C - 赖氨酸七叶皂苷及其制备方法和其用途 - Google Patents
赖氨酸七叶皂苷及其制备方法和其用途 Download PDFInfo
- Publication number
- CN100357312C CN100357312C CNB2005101055854A CN200510105585A CN100357312C CN 100357312 C CN100357312 C CN 100357312C CN B2005101055854 A CNB2005101055854 A CN B2005101055854A CN 200510105585 A CN200510105585 A CN 200510105585A CN 100357312 C CN100357312 C CN 100357312C
- Authority
- CN
- China
- Prior art keywords
- methionin
- aescin
- aescine
- total
- lysine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
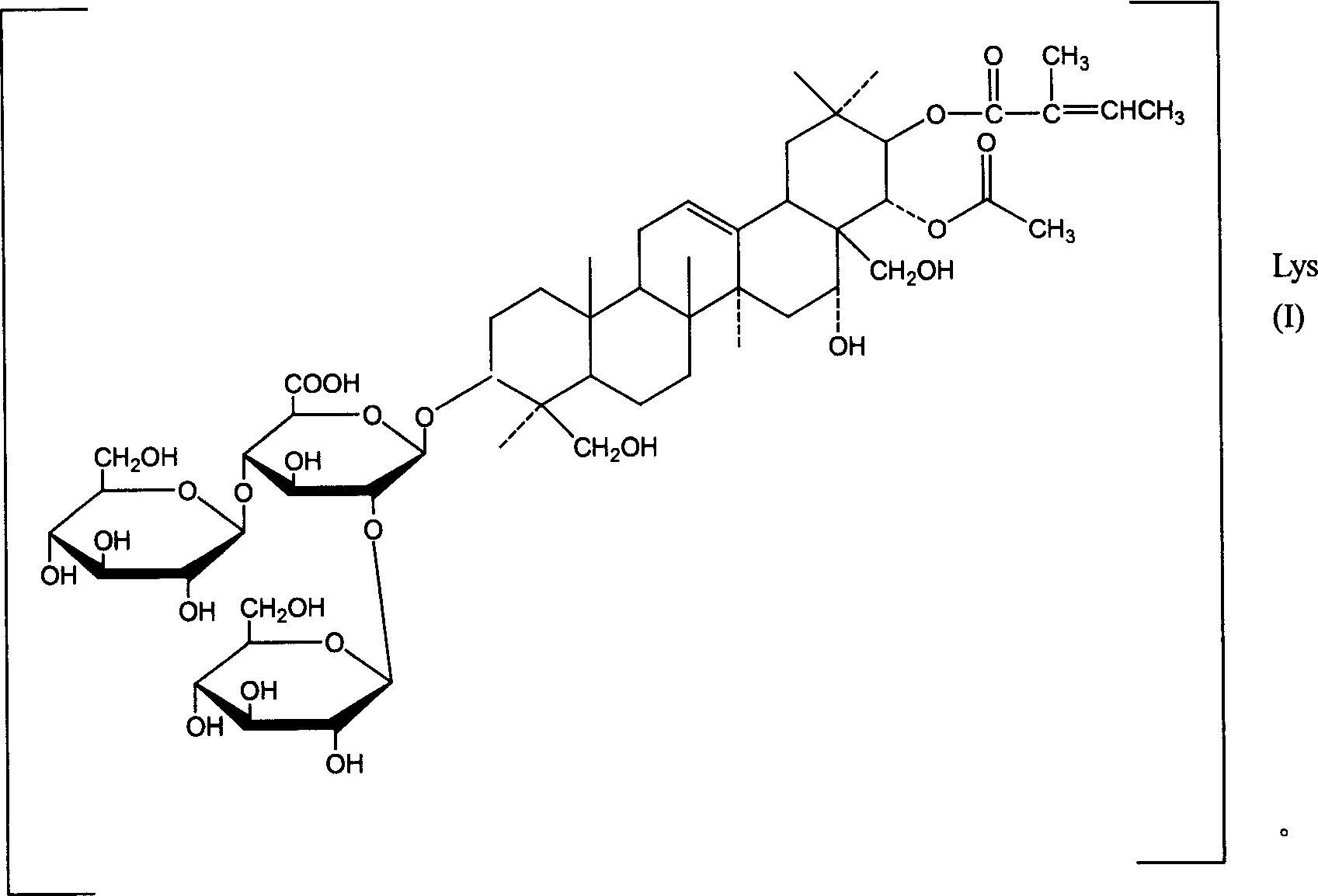
- AXNVHPCVMSNXNP-GKTCLTPXSA-N Aescin Natural products O=C(O[C@H]1[C@@H](OC(=O)C)[C@]2(CO)[C@@H](O)C[C@@]3(C)[C@@]4(C)[C@@H]([C@]5(C)[C@H]([C@](CO)(C)[C@@H](O[C@@H]6[C@@H](O[C@H]7[C@@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O7)[C@@H](O)[C@H](O[C@H]7[C@H](O)[C@@H](O)[C@H](O)[C@H](CO)O7)[C@@H](C(=O)O)O6)CC5)CC4)CC=C3[C@@H]2CC1(C)C)/C(=C/C)/C AXNVHPCVMSNXNP-GKTCLTPXSA-N 0.000 title claims abstract description 68
- 239000004472 Lysine Substances 0.000 title claims abstract description 28
- -1 Lysine aescin saponin Chemical class 0.000 title claims abstract description 28
- 238000002360 preparation method Methods 0.000 title claims description 36
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 title abstract description 21
- 229930182490 saponin Natural products 0.000 title description 23
- 239000001397 quillaja saponaria molina bark Substances 0.000 title description 19
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 claims abstract description 39
- 230000003110 anti-inflammatory effect Effects 0.000 claims abstract description 38
- 239000000203 mixture Substances 0.000 claims abstract description 33
- 150000001875 compounds Chemical class 0.000 claims abstract description 18
- 206010048962 Brain oedema Diseases 0.000 claims abstract description 12
- 208000006752 brain edema Diseases 0.000 claims abstract description 11
- XUYPXLNMDZIRQH-LURJTMIESA-N N-acetyl-L-methionine Chemical compound CSCC[C@@H](C(O)=O)NC(C)=O XUYPXLNMDZIRQH-LURJTMIESA-N 0.000 claims description 172
- 229930182817 methionine Natural products 0.000 claims description 171
- 239000000243 solution Substances 0.000 claims description 47
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 38
- 238000000034 method Methods 0.000 claims description 32
- BVHLGVCQOALMSV-JEDNCBNOSA-N L-lysine hydrochloride Chemical compound Cl.NCCCC[C@H](N)C(O)=O BVHLGVCQOALMSV-JEDNCBNOSA-N 0.000 claims description 31
- 239000003814 drug Substances 0.000 claims description 31
- 229960005337 lysine hydrochloride Drugs 0.000 claims description 31
- 239000008194 pharmaceutical composition Substances 0.000 claims description 27
- 229960003646 lysine Drugs 0.000 claims description 26
- 238000002347 injection Methods 0.000 claims description 21
- 239000007924 injection Substances 0.000 claims description 21
- 239000003937 drug carrier Substances 0.000 claims description 20
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 18
- 239000000843 powder Substances 0.000 claims description 17
- 231100001274 therapeutic index Toxicity 0.000 claims description 15
- AXNVHPCVMSNXNP-OXPBSUTMSA-N Aescin Chemical compound O([C@@H]1[C@H](O[C@H]([C@@H]([C@H]1O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@H]1CC[C@]2(C)[C@H]3CC=C4[C@@]([C@@]3(CC[C@H]2[C@]1(CO)C)C)(C)C[C@@H](O)[C@@]1(CO)[C@@H](OC(C)=O)[C@@H](C(C[C@H]14)(C)C)OC(=O)C(\C)=C/C)C(O)=O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O AXNVHPCVMSNXNP-OXPBSUTMSA-N 0.000 claims description 12
- 230000000638 stimulation Effects 0.000 claims description 12
- 239000007788 liquid Substances 0.000 claims description 10
- 235000019766 L-Lysine Nutrition 0.000 claims description 9
- 206010030113 Oedema Diseases 0.000 claims description 9
- 229960004756 ethanol Drugs 0.000 claims description 8
- 208000027418 Wounds and injury Diseases 0.000 claims description 7
- 239000003795 chemical substances by application Substances 0.000 claims description 7
- 238000002425 crystallisation Methods 0.000 claims description 7
- 230000008025 crystallization Effects 0.000 claims description 7
- 229960000935 dehydrated alcohol Drugs 0.000 claims description 7
- 238000005057 refrigeration Methods 0.000 claims description 7
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 claims description 6
- 229930195722 L-methionine Natural products 0.000 claims description 6
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims description 5
- 102000008100 Human Serum Albumin Human genes 0.000 claims description 5
- 108091006905 Human Serum Albumin Proteins 0.000 claims description 5
- 229930195725 Mannitol Natural products 0.000 claims description 5
- 238000009472 formulation Methods 0.000 claims description 5
- 239000000594 mannitol Substances 0.000 claims description 5
- 235000010355 mannitol Nutrition 0.000 claims description 5
- 239000012141 concentrate Substances 0.000 claims description 4
- 210000000936 intestine Anatomy 0.000 claims description 4
- 239000007921 spray Substances 0.000 claims description 4
- 230000000472 traumatic effect Effects 0.000 claims description 4
- 208000038016 acute inflammation Diseases 0.000 claims description 3
- 230000006022 acute inflammation Effects 0.000 claims description 3
- 239000002775 capsule Substances 0.000 claims description 3
- 239000008187 granular material Substances 0.000 claims description 3
- 210000004072 lung Anatomy 0.000 claims description 3
- 239000006187 pill Substances 0.000 claims description 3
- 230000002980 postoperative effect Effects 0.000 claims description 3
- 238000005507 spraying Methods 0.000 claims description 3
- 239000003826 tablet Substances 0.000 claims description 3
- 238000001291 vacuum drying Methods 0.000 claims description 3
- 239000012153 distilled water Substances 0.000 claims description 2
- 238000001035 drying Methods 0.000 claims description 2
- YFESOSRPNPYODN-RSMWSHJLSA-N (2s,3s,4s,5r,6r)-6-[[(4s,6ar,6bs,8r,8ar,9r,10r,14br)-9-acetyloxy-8-hydroxy-4,8a-bis(hydroxymethyl)-4,6a,6b,11,11,14b-hexamethyl-10-[(z)-2-methylbut-2-enoyl]oxy-1,2,3,4a,5,6,7,8,9,10,12,12a,14,14a-tetradecahydropicen-3-yl]oxy]-4-hydroxy-3,5-bis[[(2s,3r,4s, Chemical compound O([C@@H]1[C@H](O[C@H]([C@@H]([C@H]1O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)OC1CC[C@]2(C)C3CC=C4[C@@]([C@@]3(CCC2[C@]1(CO)C)C)(C)C[C@@H](O)[C@@]1(CO)[C@@H](OC(C)=O)[C@@H](C(CC14)(C)C)OC(=O)C(\C)=C/C)C(O)=O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O.O([C@@H]1[C@H](O[C@H]([C@@H]([C@H]1O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)OC1CC[C@]2(C)C3CC=C4[C@@]([C@@]3(CCC2[C@]1(CO)C)C)(C)C[C@@H](O)[C@@]1(CO)[C@@H](OC(C)=O)[C@@H](C(CC14)(C)C)OC(=O)C(/C)=C/C)C(O)=O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O YFESOSRPNPYODN-RSMWSHJLSA-N 0.000 claims 30
- 230000002265 prevention Effects 0.000 claims 2
- 239000002994 raw material Substances 0.000 claims 1
- 229940011399 escin Drugs 0.000 abstract description 8
- 229930186222 escin Natural products 0.000 abstract description 8
- 230000017531 blood circulation Effects 0.000 abstract description 3
- 229930192064 isoescin Natural products 0.000 abstract description 3
- 238000004519 manufacturing process Methods 0.000 abstract description 2
- 230000002322 anti-exudative effect Effects 0.000 abstract 1
- 229940093314 beta-escin Drugs 0.000 abstract 1
- AXNVHPCVMSNXNP-BEJCRFBNSA-N beta-escin Natural products CC=C(/C)C(=O)O[C@H]1[C@H](OC(=O)C)[C@]2(CO)[C@H](O)C[C@@]3(C)C(=CC[C@@H]4[C@@]5(C)CC[C@H](O[C@H]6O[C@@H]([C@H](O[C@H]7O[C@H](CO)[C@@H](O)[C@H](O)[C@H]7O)[C@H](O)[C@@H]6O[C@@H]8O[C@H](CO)[C@@H](O)[C@H](O)[C@H]8O)C(=O)O)[C@](C)(CO)[C@@H]5CC[C@@]34C)[C@@H]2CC1(C)C AXNVHPCVMSNXNP-BEJCRFBNSA-N 0.000 abstract 1
- 239000000969 carrier Substances 0.000 abstract 1
- 241001465754 Metazoa Species 0.000 description 38
- 238000012360 testing method Methods 0.000 description 36
- 206010020565 Hyperaemia Diseases 0.000 description 35
- 238000006243 chemical reaction Methods 0.000 description 31
- 230000000694 effects Effects 0.000 description 25
- 235000017709 saponins Nutrition 0.000 description 22
- 210000003462 vein Anatomy 0.000 description 21
- 239000004475 Arginine Substances 0.000 description 19
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 19
- 241000283973 Oryctolagus cuniculus Species 0.000 description 17
- 230000002401 inhibitory effect Effects 0.000 description 17
- 235000018977 lysine Nutrition 0.000 description 17
- 239000002085 irritant Substances 0.000 description 16
- 231100000021 irritant Toxicity 0.000 description 16
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 13
- 230000034994 death Effects 0.000 description 13
- 239000002504 physiological saline solution Substances 0.000 description 13
- 230000008961 swelling Effects 0.000 description 13
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 12
- 238000005259 measurement Methods 0.000 description 12
- 235000019633 pungent taste Nutrition 0.000 description 12
- 150000003839 salts Chemical class 0.000 description 12
- 238000010998 test method Methods 0.000 description 11
- 210000005069 ears Anatomy 0.000 description 10
- 230000007794 irritation Effects 0.000 description 10
- 230000001575 pathological effect Effects 0.000 description 10
- BMFMQGXDDJALKQ-BYPYZUCNSA-N Argininic acid Chemical class NC(N)=NCCC[C@H](O)C(O)=O BMFMQGXDDJALKQ-BYPYZUCNSA-N 0.000 description 9
- 210000004556 brain Anatomy 0.000 description 9
- 230000002490 cerebral effect Effects 0.000 description 9
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 description 9
- 210000003205 muscle Anatomy 0.000 description 9
- 231100000419 toxicity Toxicity 0.000 description 9
- 230000001988 toxicity Effects 0.000 description 9
- 230000008859 change Effects 0.000 description 8
- 150000007949 saponins Chemical class 0.000 description 8
- 241000700159 Rattus Species 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 7
- 238000010171 animal model Methods 0.000 description 7
- 229940079593 drug Drugs 0.000 description 7
- 239000011734 sodium Substances 0.000 description 7
- 229910052717 sulfur Inorganic materials 0.000 description 7
- 239000008215 water for injection Substances 0.000 description 7
- 201000006474 Brain Ischemia Diseases 0.000 description 6
- 206010008120 Cerebral ischaemia Diseases 0.000 description 6
- 241000699666 Mus <mouse, genus> Species 0.000 description 6
- 206010028851 Necrosis Diseases 0.000 description 6
- 206010008118 cerebral infarction Diseases 0.000 description 6
- 235000012000 cholesterol Nutrition 0.000 description 6
- 210000001508 eye Anatomy 0.000 description 6
- 230000000302 ischemic effect Effects 0.000 description 6
- 206010023683 lagophthalmos Diseases 0.000 description 6
- 230000017074 necrotic cell death Effects 0.000 description 6
- 230000008569 process Effects 0.000 description 6
- 239000007787 solid Substances 0.000 description 6
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 5
- 206010052428 Wound Diseases 0.000 description 5
- 229960000583 acetic acid Drugs 0.000 description 5
- 238000013019 agitation Methods 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- 238000001990 intravenous administration Methods 0.000 description 5
- 238000010253 intravenous injection Methods 0.000 description 5
- 238000004811 liquid chromatography Methods 0.000 description 5
- 229910052708 sodium Inorganic materials 0.000 description 5
- 159000000000 sodium salts Chemical class 0.000 description 5
- 238000005406 washing Methods 0.000 description 5
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 4
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 4
- 230000005856 abnormality Effects 0.000 description 4
- 231100000652 hormesis Toxicity 0.000 description 4
- 239000012071 phase Substances 0.000 description 4
- 210000003314 quadriceps muscle Anatomy 0.000 description 4
- 230000009467 reduction Effects 0.000 description 4
- 239000011347 resin Substances 0.000 description 4
- 229920005989 resin Polymers 0.000 description 4
- 210000000689 upper leg Anatomy 0.000 description 4
- 230000002792 vascular Effects 0.000 description 4
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 230000000996 additive effect Effects 0.000 description 3
- 230000037396 body weight Effects 0.000 description 3
- 238000004090 dissolution Methods 0.000 description 3
- 239000000890 drug combination Substances 0.000 description 3
- 239000003889 eye drop Substances 0.000 description 3
- 238000004108 freeze drying Methods 0.000 description 3
- 208000014674 injury Diseases 0.000 description 3
- 238000010255 intramuscular injection Methods 0.000 description 3
- 239000007927 intramuscular injection Substances 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- 231100000331 toxic Toxicity 0.000 description 3
- 230000002588 toxic effect Effects 0.000 description 3
- 230000008733 trauma Effects 0.000 description 3
- SFLSHLFXELFNJZ-QMMMGPOBSA-N (-)-norepinephrine Chemical compound NC[C@H](O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-QMMMGPOBSA-N 0.000 description 2
- 238000005160 1H NMR spectroscopy Methods 0.000 description 2
- 240000006409 Acacia auriculiformis Species 0.000 description 2
- 241000157282 Aesculus Species 0.000 description 2
- 206010002091 Anaesthesia Diseases 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- 210000000683 abdominal cavity Anatomy 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 230000037005 anaesthesia Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 210000001218 blood-brain barrier Anatomy 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- XHCADAYNFIFUHF-TVKJYDDYSA-N esculin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC(C(=C1)O)=CC2=C1OC(=O)C=C2 XHCADAYNFIFUHF-TVKJYDDYSA-N 0.000 description 2
- 230000003203 everyday effect Effects 0.000 description 2
- 239000000284 extract Substances 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 229940012356 eye drops Drugs 0.000 description 2
- 238000004992 fast atom bombardment mass spectroscopy Methods 0.000 description 2
- 235000010181 horse chestnut Nutrition 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 239000013002 intravenous (IV) drug Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 230000007170 pathology Effects 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 210000003752 saphenous vein Anatomy 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 235000014347 soups Nutrition 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 230000008719 thickening Effects 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- MPDDTAJMJCESGV-CTUHWIOQSA-M (3r,5r)-7-[2-(4-fluorophenyl)-5-[methyl-[(1r)-1-phenylethyl]carbamoyl]-4-propan-2-ylpyrazol-3-yl]-3,5-dihydroxyheptanoate Chemical compound C1([C@@H](C)N(C)C(=O)C2=NN(C(CC[C@@H](O)C[C@@H](O)CC([O-])=O)=C2C(C)C)C=2C=CC(F)=CC=2)=CC=CC=C1 MPDDTAJMJCESGV-CTUHWIOQSA-M 0.000 description 1
- 229930182836 (R)-noradrenaline Natural products 0.000 description 1
- DYLIWHYUXAJDOJ-OWOJBTEDSA-N (e)-4-(6-aminopurin-9-yl)but-2-en-1-ol Chemical compound NC1=NC=NC2=C1N=CN2C\C=C\CO DYLIWHYUXAJDOJ-OWOJBTEDSA-N 0.000 description 1
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 1
- QCVGEOXPDFCNHA-UHFFFAOYSA-N 5,5-dimethyl-2,4-dioxo-1,3-oxazolidine-3-carboxamide Chemical compound CC1(C)OC(=O)N(C(N)=O)C1=O QCVGEOXPDFCNHA-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 206010010904 Convulsion Diseases 0.000 description 1
- 102000002322 Egg Proteins Human genes 0.000 description 1
- 108010000912 Egg Proteins Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- 241001143502 Hippocastanaceae Species 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 206010022998 Irritability Diseases 0.000 description 1
- 238000012449 Kunming mouse Methods 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241000237502 Ostreidae Species 0.000 description 1
- 208000037273 Pathologic Processes Diseases 0.000 description 1
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- 208000005392 Spasm Diseases 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- 230000003187 abdominal effect Effects 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 231100000215 acute (single dose) toxicity testing Toxicity 0.000 description 1
- 238000011047 acute toxicity test Methods 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 230000000274 adsorptive effect Effects 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 125000000637 arginyl group Chemical group N[C@@H](CCCNC(N)=N)C(=O)* 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000002146 bilateral effect Effects 0.000 description 1
- 230000008499 blood brain barrier function Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 208000025698 brain inflammatory disease Diseases 0.000 description 1
- 210000005252 bulbus oculi Anatomy 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 210000001715 carotid artery Anatomy 0.000 description 1
- 239000003729 cation exchange resin Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 238000005352 clarification Methods 0.000 description 1
- 210000000078 claw Anatomy 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 230000036461 convulsion Effects 0.000 description 1
- 210000004087 cornea Anatomy 0.000 description 1
- 208000029078 coronary artery disease Diseases 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 230000002526 effect on cardiovascular system Effects 0.000 description 1
- 235000014103 egg white Nutrition 0.000 description 1
- 210000000969 egg white Anatomy 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 206010014599 encephalitis Diseases 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N ethyl acetate Substances CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 230000008014 freezing Effects 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 125000003147 glycosyl group Chemical group 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 229910017053 inorganic salt Inorganic materials 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 208000028867 ischemia Diseases 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 231100000053 low toxicity Toxicity 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 210000004877 mucosa Anatomy 0.000 description 1
- 231100000286 mucous membrane, eye irritation or corrosion testing Toxicity 0.000 description 1
- 230000003387 muscular Effects 0.000 description 1
- 208000031225 myocardial ischemia Diseases 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 235000020636 oyster Nutrition 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 230000009054 pathological process Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- DDBREPKUVSBGFI-UHFFFAOYSA-N phenobarbital Chemical compound C=1C=CC=CC=1C1(CC)C(=O)NC(=O)NC1=O DDBREPKUVSBGFI-UHFFFAOYSA-N 0.000 description 1
- 208000001297 phlebitis Diseases 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- 239000012716 precipitator Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000003672 processing method Methods 0.000 description 1
- 230000000770 proinflammatory effect Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 244000157013 shichito matgrass Species 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 229960004249 sodium acetate Drugs 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- PBDAWQLIMPWEEF-JEDNCBNOSA-M sodium;(2s)-2,6-diaminohexanoate Chemical compound [Na+].NCCCC[C@H](N)C([O-])=O PBDAWQLIMPWEEF-JEDNCBNOSA-M 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 238000011287 therapeutic dose Methods 0.000 description 1
- 231100000816 toxic dose Toxicity 0.000 description 1
- 231100000027 toxicology Toxicity 0.000 description 1
- 238000005292 vacuum distillation Methods 0.000 description 1
- 230000024883 vasodilation Effects 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
| 剂量(escin计)(mg·kg-1) | 动物数(n) | 肿胀值[X±S(mg)] | 抑制率(%) | ED50(p=0.95)(mg·kg-1) |
| 1.000.500.250.1250.4ml NS. | 1010101010 | 8.2±3.6210.31±4.5412.24±4.6714.94±4.2318.16±4.82 | 54.8***43.2**32.6*17.7△/ | 0.74(0.88~0.62) |
| 剂量(escin计)(mg·kg-1) | 动物数(n) | 肿胀值[X±S(mg)] | 抑制率(%) | ED50(p=0.95)(mg·kg-1) |
| 1.000.500.250.1250.4ml NS. | 1212121212 | 8.14±3.2511.82±2.9514.29±2.6916.20±1.9221.21±2.49 | 61.6***44.3***32.6***23.6***/ | 0.54(0.82~0.44) |
| 剂量(β-七叶皂苷计)(mg·kg-1) | 动物数(n) | 肿胀值(X±S,mg) | 抑制率(%) | ED50(p=0.95)(mg·kg-1) |
| 0.600.300.150.0750.4ml NS | 1010101010 | 9.25±3.0112.78±2.8315.75±2.5917.18±1.8820.91±2.31 | 55.8**38.8**24.7**17.8*/ | 0.497(0.69~0.36) |
| 剂量(β-七叶皂苷计)(mg·kg-1) | 动物数(n) | 肿胀值(X±S,mg) | 抑制率(%) | ED50(p=0.95)(mg·kg-1) |
| 0.600.300.150.0750.4ml NS | 1010101010 | 8.02±2.2511.71±2.3114.14±2.3916.12±2.0121.12±1.96 | 62.0***44.6***33.0**23.7**/ | 0.35(0.56~0.21) |
| 剂量(mg·kg-1) | 动物数(n) | 肿胀值(X±S,mg) | 抑制率(%) | ED50(mg·kg-1) |
| 248160.4ml NS | 1010101010 | 16.02±2.12*14.13±1.91**10.21±1.87***8.16±2.01***20.86±1.85 | 23.2032.2651.0560.88/ | 8.70 |
| 剂量(mg·kg-1) | 动物数(n) | 肿胀值(X±S,mg) | 抑制率(%) | ED50(mg·kg-1) |
| 1.53.06.01200.4ml NS | 1010101010 | 15.28±2.21*12.62±2.31**10.02±1.98***7.59±2.33***19.92±1.93 | 23.3236.6449.7061.89/ | 6.18 |
| 剂量(七叶皂苷计)(mg·kg-1) | 动物数(n) | 肿胀值(X±S,mg) | 抑制率(%) | ED50(p=0.95)(mg·kg-1) |
| 0.9000.4500.2250.1120.4ml NS | 1010101010 | ***6.20±1.89**9.01±1.42**9.52±2.12*12.01±3.7215.42±2.96 | 59.841.638.322.1/ | 0.5(0.89~0.41) |
| 组别 | 剂量(mg·kg-1) | 动物数(n) | 吸光度 | 抑制率(%) |
| 总七叶皂苷钠赖氨酸总七叶皂苷 | 1.51.2 | 1010 | 0.2057±0.055**0.1995±0.038** | 40.342.1 |
| 谷胱苷肽∶赖氨酸总七叶皂苷(3∶1)赖氨酸β-七叶皂苷谷胱苷肽∶赖氨酸β-七叶皂苷(3∶1)空白对照 | 1.01.00.8同体积生理盐水 | 10101010 | 0.1938±0.042**0.1765±0.052**0.1674±0.046**0.3446±0.086 | 43.848.751.4/ |
| 组别 | 剂量 | 动物数(n) | 脑含水量(%) | 脑指数 |
| 假手术对照脑缺血模型组总七叶皂苷钠组Esc-Lys∶Lys.HCl(3∶1) | N.SN.S1 mg·kg-11mg·kg-1 | 10101010 | 74.0±1.680.1±2.2△△77.5±2.2*75.10±1.9** | 0.801±0.0310.890±0.034△△0.855±0.036*0.820±0.040** |
| 剂量(mg·kg-1) | 对数剂量(X) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(Y) |
| 2.052.563.204.005.006.25 | 0.3120.4080.5050.6020.6990.796 | 151515151515 | 1236914 | 61320406093 | 3.453.874.164.755.256.48 |
| 剂量(mg·kg-1) | 对数剂量 | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(Y) |
| 10.007.004.903.432.40 | 1.0000.8450.6900.5350.380 | 1818181818 | 139631 | 725033175.5 | 5.585.004.564.053.45 |
| 剂量(mg·kg-1) | 对数剂量(x) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(Y) |
| 10.007.004.903.432.40 | 1.0000.8450.6900.5350.380 | 1616161616 | 129631 | 755638196 | 5.675.154.694.123.45 |
| 剂 量(mg·kg-1) | 对数剂量(x) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(Y) |
| 5.003.502.451.72 | 0.6990.5440.3890.234 | 16161616 | 12975 | 75564431 | 5.675.154.854.50 |
| 剂量(mg·kg-1) | 对数剂量(x) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(Y) |
| 45.0036.0028.8023.0418.43 | 1.6531.5561.4591.3621.266 | 1818181818 | 1713831 | 947244165 | 6.555.584.854.013.36 |
| 剂量(mg·kg-1) | 对数剂量(x) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(Y) |
| 45.0036.0028.8023.0418.43 | 1.6531.5561.4591.3621.266 | 1818181818 | 139531 | 725027175 | 5.585.004.394.053.36 |
| 组别 | 剂量(mg·kg-1) | 对数剂量 | 动物数(n) | 死亡数(r) | 死亡率(%) | LD50(mg·kg-1)P=0.95 |
| 谷胱苷肽∶赖氨酸总七叶皂苷(10∶1) | 42.0029.4020.5814.4110.08 | 1.6231.4681.3131.1591.003 | 1010101010 | 107763 | 10070706030 | 14.22(15.36~12.88mg·kg-1) |
| 谷胱苷肽∶赖氨酸总七叶皂苷(3∶1) | 10.008.006.405.124.10 | 1.0000.9030.8060.7090.613 | 1212121212 | 107531 | 835842258 | 7.00mg·kg-1(8.21~6.01mg·kg-1) |
| 剂量(mg·kg-1) | 对数剂量(X) | 动物数(n) | 动物死亡数(r) | 死亡率(%)(p) | 概率(Y) |
| 10.007.505.364.223.17 | 1.0000.8750.7500.6260.500 | 1212121212 | 97421 | 755833178 | 5.675.204.564.053.59 |
| 盐类名 | 成分名称 | 最小有效量(mg·kg-1) | ED50(mg·kg-1) | LD50(mg·kg-1) | 治疗指数Ti(LD50/ED50) |
| 钠盐赖氨酸盐 | 总七叶皂苷B-七叶皂苷异七叶皂苷总七叶皂苷B-七叶皂苷 | 0.125~0.25≤0.25-≤0.125≤0.075 | 0.740.58.70.540.35 | 4.22.8130.046.766.27 | 5.75.63.4513.517.9 |
| 异七叶皂苷 | -- | 6.18 | 35.75 | 5.78 |
| 反应级 | 刺激反应 |
| 012345 | 无明显变化轻度充血,其范围在0.5×1.0厘米以下中度充血,其范围在0.5×1.0厘米以上重度充血,伴有肌肉变性出现坏死,有褐色变性出现广泛坏死 |
| 品名(注射部位总固体) | 股四头肌刺激反应(测量单位cm) | 反应级数 | 股四头肌刺激反应(测量单位:cm) | 反应级数 | 反应级数和 |
| 总七叶皂苷钠(5mg) | ①重度充血4.7×4.8②重度充血4.0×4.7 | 33 | ③重度充血4.4×4.9④重度充血4.4×4.7 | 33 | 12 |
| 赖氨酸总七叶皂苷(5.5mg) | ①轻度充血0.56×0.9②轻度充血2.0×0.15 | 11 | ③中度充血1.8×0.44④轻度充血1.46×0.3 | 21 | 5 |
| 赖氨酸七叶皂苷∶盐酸赖氨酸(1∶3)(20mg) | ①轻度充血0.3×0.4②轻度充血1.0×0.5 | 11 | ③中度充血0.5×1.2④无充血 | 20 | 4 |
| B-七叶皂苷钠(5mg) | ①有褐色变性②出现坏死 | 44 | 出现坏死有褐色变性 | 44 | 16 |
| 赖氨酸七叶皂苷(5.5mg) | ①轻度充血0.5×1.0②轻度充血0.6×1.2 | 22 | ③轻度充血0.5×1.0④轻度充血0.6×0.9 | 12 | 6 |
| 赖氨酸总七叶皂苷∶谷胱甘肽(1∶3)(20mg) | ①中度充血4.4×0.52②中度充血2.6×0.95 | 22 | ③中度充血3.2×0.49④中度充血3.11×1.06 | 22 | 8 |
| 剂量10-5g/ml | 例数 | 犬大隐静脉条张力增加(%) | |
| 总七叶皂苷钠 | 赖氨酸总七叶皂苷 | ||
| 5.09.016.229.252.5 | 55555 | 8.315±9.30513.016±8.67820.913±6.95634.889±9.00342.158±7.681 | 7.9987±8.36412.8865±7.41219.8983±7.03135.023±10.62543.012±6.983 |
| 谷胱甘肽∶赖氨酸七叶皂苷 | 剂量(以Escin计) | 动物数(n) | 肿胀值(X±S,mg) | 抑制率(%) |
| 10∶16∶13∶10.4ml NS. | 0.45mg·kg-1 | 10101010 | **12.31±1.92**12.19±1.83**12.89±2.0520.56±1.88 | 40.140.737.3/ |
| 谷胱甘肽∶赖氨酸总七叶皂苷(总固体物) | 剂量(Escin计)mg·kg-1 | 股四头肌刺激反应(测量单位:cm) | 反应级数 | 股四头肌刺激反应(测量单位:cm) | 反应级数 | 总反应级数 |
| 10∶1(55mg) | 5 | ①重度充血4.5×4.2②重度充血3.4×1.49 | 33 | ①中度充血2.4×0.96②中度充血2.9×0.56 | 22 | 10 |
| 6∶1(35mg) | 5 | ①中度充血1.85×0.5②中度充血2.78×0.48 | 22 | ①中度充血1.1×0.52②中度充血3.2×1.12 | 22 | 8 |
| 3∶1(20mg) | 5 | ①中度充血4.4×0.52②中度充血2.6×0.95 | 22 | ①中度充血2.4×0.96②中度充血2.9×0.56 | 22 | 8 |
| 组别 | 剂量(以escin计,iv) | 动物数(n) | 脑含水量(%) | 抑制率(%) |
| 假手术对照组脑缺血模型1∶31∶61∶10 | NS.同体积NS.同体积0.5mg.kg-10.5mg.kg-10.5mg.kg-1 | 1010101010 | 74.00±1.6080.10±2.2077.81±1.9076.96±1.8077.00±2.00 | //37.551.450.8 |
| 剂量(mg.kg-1)(以总七叶皂苷计) | 动物数(n) | 肿胀值X±S(mg) | 抑制率(%) | ED50(p=0.95)mg.kg-1 |
| 0.8700.4350.2180.109空白对照组(NS.) | 1212121212 | 8.26±3.7611.30±2.9912.42±3.0416.22±3.0118.39±2.34 | 55***38.6***32.5***11.8*/ | 0.68(1.08~0.56) |
| 剂量(mg.kg-1,以皂苷计) | 动物数(n) | 肿胀值X±S(mg) | 抑制率(%) | ED50(p=0.95)mg.kg-1 |
| 0.8700.4350.2180.109空白对照组NS、0.5ml | 1212121212 | 8.06±3.6911.18±2.8912.29±3.0215.06±3.0419.19±2.16 | 57.99**41.70**35.90**21.50**/ | 0.59(0.99~0.46) |
| 剂量(mg.kg-1,以皂苷计) | 动物数(n) | 肿胀值(X±S,mg) | 抑制率(%) | ED50(p=0.95)(mg.kg-1) |
| 0.8700.4350.2180.109空白对照组NS、0.5ml | 1212121212 | 7.46±3.019.05±2.7212.98±2.5614.61 ±2.8319.76±2.23 | 62.2**54.2**34.3**26.0**/ | 6.73(10.2~5.5) |
| 剂量(mg.kg-1)(以总七叶皂苷计) | 对数剂量(x) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(y) |
| 15.0010.507.355.143.60 | 1.1761.0210.8660.7110.556 | 1010101010 | 87642 | 8070604020 | 5.845.525.254.754.16 |
| 剂量(mg.kg-1)(以β-七叶皂苷计) | 对数剂量(x) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(y) |
| 15.0010.507.355.143.60 | 1.1761.0210.8660.7110.556 | 1010101010 | 98742 | 9080704020 | 6.285.845.524.754.16 |
| 剂量mg.kg-1(以异七叶皂苷计) | 对数剂量(x) | 动物数(n) | 死亡数(r) | 死亡率(%) | 概率单位(y) |
| 45.0036.0028.8023.0418.43 | 1.6531.5561.4591.3621.266 | 1010101010 | 97531 | 9070503010 | 6.285.525.004.483.72 |
| 皂苷及其盐名称 | 剂量计算方法 | 抗炎活性(ED50mg.kg-1) | LD50mg.kg-1 | 相对治疗指数(Ti) | 对静脉血管刺激作用(注射1ml含5mg) | |
| 皂苷名称 | 盐的名称 | |||||
| 总七叶皂苷 | 赖氨酸盐 | 以总七叶皂苷计 | 0.54 | 6.76 | 13.50 | 几乎无刺激作用 |
| 精氨酸盐 | 0.68 | 6.68 | 9.82 | 有严重刺激反应 | ||
| B-七叶皂苷 | 赖氨酸盐 | 以β-七叶皂苷计 | 0.35 | 6.27 | 17.90 | 几乎无刺激作用 |
| 精氨酸盐 | 0.59 | 6.11 | 10.36 | |||
| 异七叶皂苷 | 赖氨酸盐 | 以异七叶皂苷计 | 6.18 | 35.75 | 5.78 | |
| 精氨酸盐 | 6.73 | 28.37 | 4.21 |
Claims (49)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB2005101055854A CN100357312C (zh) | 2005-07-15 | 2005-09-29 | 赖氨酸七叶皂苷及其制备方法和其用途 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN200510084022 | 2005-07-15 | ||
| CN200510084022.1 | 2005-07-15 | ||
| CNB2005101055854A CN100357312C (zh) | 2005-07-15 | 2005-09-29 | 赖氨酸七叶皂苷及其制备方法和其用途 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1896092A CN1896092A (zh) | 2007-01-17 |
| CN100357312C true CN100357312C (zh) | 2007-12-26 |
Family
ID=37608753
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB2005101055854A Active CN100357312C (zh) | 2005-07-15 | 2005-09-29 | 赖氨酸七叶皂苷及其制备方法和其用途 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN100357312C (zh) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102755297A (zh) * | 2011-04-29 | 2012-10-31 | 天津药物研究院 | 一种七叶皂苷b冻干粉针剂及其制备方法和应用 |
| CN102805750B (zh) * | 2011-05-31 | 2015-09-23 | 天津药物研究院 | 一种七叶皂苷衍生物及其盐的组合物、其制备方法和医药用途 |
| CN104147055A (zh) * | 2014-07-08 | 2014-11-19 | 武汉爱民制药有限公司 | 赖氨酸七叶皂苷及其盐在制备治疗i型超敏反应药物中的用途 |
| CN104288167B (zh) * | 2014-10-29 | 2017-08-25 | 马应龙药业集团股份有限公司 | 一种治疗痔疮的七叶皂苷钠制剂及其制备方法 |
| CN106580891B (zh) * | 2016-11-15 | 2019-08-27 | 武汉爱民制药股份有限公司 | 一种七叶皂苷b注射剂及其制备方法 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1017591A (ja) * | 1996-06-27 | 1998-01-20 | Jintan Dorufu:Kk | 抗炎症剤 |
| CN1241571A (zh) * | 1998-07-15 | 2000-01-19 | 烟台绿叶制药有限公司 | 一种低毒的抗炎抗渗出药物组合物 |
| CN1376675A (zh) * | 2002-04-10 | 2002-10-30 | 成都圣诺科技发展有限公司 | 具有消肿抗炎改善血液循环的精氨酸七叶皂苷及制备方法 |
| CN1156278C (zh) * | 1999-12-30 | 2004-07-07 | 山东绿叶制药股份有限公司 | 低毒的抗炎抗渗出药物组合物 |
| CN1575797A (zh) * | 2003-07-05 | 2005-02-09 | 山东绿叶制药股份有限公司 | 七叶皂苷及其盐的药物新用途 |
| CN1687103A (zh) * | 2005-03-24 | 2005-10-26 | 成都圣诺科技发展有限公司 | 水溶性七叶皂苷盐化合物及其制备方法 |
-
2005
- 2005-09-29 CN CNB2005101055854A patent/CN100357312C/zh active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1017591A (ja) * | 1996-06-27 | 1998-01-20 | Jintan Dorufu:Kk | 抗炎症剤 |
| CN1241571A (zh) * | 1998-07-15 | 2000-01-19 | 烟台绿叶制药有限公司 | 一种低毒的抗炎抗渗出药物组合物 |
| CN1156278C (zh) * | 1999-12-30 | 2004-07-07 | 山东绿叶制药股份有限公司 | 低毒的抗炎抗渗出药物组合物 |
| CN1376675A (zh) * | 2002-04-10 | 2002-10-30 | 成都圣诺科技发展有限公司 | 具有消肿抗炎改善血液循环的精氨酸七叶皂苷及制备方法 |
| CN1575797A (zh) * | 2003-07-05 | 2005-02-09 | 山东绿叶制药股份有限公司 | 七叶皂苷及其盐的药物新用途 |
| CN1687103A (zh) * | 2005-03-24 | 2005-10-26 | 成都圣诺科技发展有限公司 | 水溶性七叶皂苷盐化合物及其制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1896092A (zh) | 2007-01-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6027722B2 (ja) | 脳梗塞の予防及び治療用医薬品の製造におけるl−ブチルフタリドの使用 | |
| CN100418535C (zh) | 一种七叶皂苷及其制备方法 | |
| EP2573070A1 (en) | Crystal form of (s)-4-hydroxy-2-oxo-1-pyrrolidine acetamide, preparation method and use thereof | |
| CN100428940C (zh) | 七叶皂苷的药物组合物及其制备方法和其制药用途 | |
| CN100357312C (zh) | 赖氨酸七叶皂苷及其制备方法和其用途 | |
| WO2014005084A9 (en) | Oxabicycloheptanes and oxabicycloheptenes for the treatment of diabetes | |
| JP2015525232A (ja) | 水酸基ベニバナ黄色素aナトリウムの製造方法及び用途 | |
| WO2014005080A1 (en) | Oxabicycloheptanes and oxabicycloheptenes for the treatment of reperfusion injury | |
| EP2589382A1 (en) | Pharmaceutical composition comprising levocarnitine and dobesilate | |
| WO2000043392A2 (de) | Verwendung von imidazo[1,5-a]-pyrido[3,2-e]-pyrazinonen als arzneimittel | |
| KR20050003378A (ko) | 심장-뇌 혈관 질환들 및 안저 질환들 치료용고들빼기로부터 제조된 주사제 및 그것의 제조방법 | |
| TWI472335B (zh) | 用以治療腸激躁症之山薑屬植物萃取物 | |
| CN101289438A (zh) | 3-(3′-羟基)-丁基苯酞酯及其制法和用途 | |
| DE60125955T2 (de) | Bioaktive fraktion von eurycoma longifolia | |
| CN1698835A (zh) | 一种治疗心脑血管病的复方中药注射剂 | |
| DE2538573A1 (de) | Uridine und diese enthaltende pharmazeutische praeparate | |
| US6475520B1 (en) | Pharmaceutical composition with low toxicity for anti-inflammation and anti-exudation | |
| CN1314402C (zh) | 含有蒺藜果总皂苷的药物组合物及其制备方法 | |
| CN102727481B (zh) | 包含左卡尼汀和羟苯磺酸盐的药物组合物 | |
| CN112409443B (zh) | 一种脯氨酸茨醇衍生物及其制备方法和在制备治疗心脑血管类疾病的药物中的应用 | |
| CN100525759C (zh) | 一种用于治疗缺血性脑血管疾病的中成药 | |
| CN101255149B (zh) | 7、4'-二(琥珀酸单酯)氧乙氧基-葛根黄豆苷元及其医药新用途 | |
| KR20060066177A (ko) | 데커신 및/또는 데커시놀 안젤레이트, 또는 데커신및/또는 데커시놀 안젤레이트를 유효성분으로 하는당귀추출물을 포함하는 간기능 치료제 조성물 | |
| CN101007829A (zh) | 1,6-二磷酸-d-果糖酯精氨酸盐及其药物用途 | |
| KR20060066176A (ko) | 데커신 및/또는 데커시놀 안젤레이트, 또는 데커신및/또는 데커시놀 안젤레이트를 유효성분으로 하는당귀추출물을 포함하는 신기능 치료제 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| ASS | Succession or assignment of patent right |
Free format text: FORMER OWNER: LI XINJIONG Effective date: 20150424 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20150424 Address after: 430010 Hubei province Wuhan City Jiang'an District 27 Street No. 69 Zhao Patentee after: Wuhan Aimin Pharmaceutical Co., Ltd. Address before: 430010 Hubei province Wuhan City Jiang'an District 27 Street No. 69 Zhao Patentee before: Wuhan Aimin Pharmaceutical Co., Ltd. Patentee before: Li Xinjiong |
|
| DD01 | Delivery of document by public notice |
Addressee: Feng Yu Document name: Notification of Passing Examination on Formalities |
|
| C56 | Change in the name or address of the patentee | ||
| CP01 | Change in the name or title of a patent holder |
Address after: 430010 Hubei province Wuhan City Jiang'an District 27 Street No. 69 Zhao Patentee after: Wuhan Aimin Pharmaceutical Co., Ltd Address before: 430010 Hubei province Wuhan City Jiang'an District 27 Street No. 69 Zhao Patentee before: Wuhan Aimin Pharmaceutical Co., Ltd. |
|
| PE01 | Entry into force of the registration of the contract for pledge of patent right |
Denomination of invention: Lysine aescin saponin, its preparation and use Effective date of registration: 20190129 Granted publication date: 20071226 Pledgee: Ezhou SME credit Company limited by guarantee Pledgor: Wuhan Aimin Pharmaceutical Co., Ltd Registration number: 2019420000004 Denomination of invention: Lysine aescin saponin, its preparation and use Effective date of registration: 20190129 Granted publication date: 20071226 Pledgee: Ezhou SME credit Company limited by guarantee Pledgor: Wuhan Aimin Pharmaceutical Co., Ltd Registration number: 2019420000004 |
|
| PE01 | Entry into force of the registration of the contract for pledge of patent right | ||
| PC01 | Cancellation of the registration of the contract for pledge of patent right | ||
| PC01 | Cancellation of the registration of the contract for pledge of patent right |
Date of cancellation: 20200513 Granted publication date: 20071226 Pledgee: Ezhou SME credit Company limited by guarantee Pledgor: WUHAN AIMIN PHARMACEUTICAL Co.,Ltd. Registration number: 2019420000004 |