WO2020255458A1 - 透明導電フィルム積層体及びその加工方法 - Google Patents
透明導電フィルム積層体及びその加工方法 Download PDFInfo
- Publication number
- WO2020255458A1 WO2020255458A1 PCT/JP2019/050513 JP2019050513W WO2020255458A1 WO 2020255458 A1 WO2020255458 A1 WO 2020255458A1 JP 2019050513 W JP2019050513 W JP 2019050513W WO 2020255458 A1 WO2020255458 A1 WO 2020255458A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- transparent conductive
- film
- conductive film
- transparent
- resin
- Prior art date
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/32—Layered products comprising a layer of synthetic resin comprising polyolefins
- B32B27/325—Layered products comprising a layer of synthetic resin comprising polyolefins comprising polycycloolefins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B1/00—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors
- H01B1/20—Conductive material dispersed in non-conductive organic material
- H01B1/22—Conductive material dispersed in non-conductive organic material the conductive material comprising metals or alloys
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J7/00—Adhesives in the form of films or foils
- C09J7/20—Adhesives in the form of films or foils characterised by their carriers
- C09J7/29—Laminated material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/02—Layer formed of wires, e.g. mesh
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/085—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising polyolefins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/06—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B27/08—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/12—Layered products comprising a layer of synthetic resin next to a fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/18—Layered products comprising a layer of synthetic resin characterised by the use of special additives
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/32—Layered products comprising a layer of synthetic resin comprising polyolefins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B33/00—Layered products characterised by particular properties or particular surface features, e.g. particular surface coatings; Layered products designed for particular purposes not covered by another single class
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B7/00—Layered products characterised by the relation between layers; Layered products characterised by the relative orientation of features between layers, or by the relative values of a measurable parameter between layers, i.e. products comprising layers having different physical, chemical or physicochemical properties; Layered products characterised by the interconnection of layers
- B32B7/02—Physical, chemical or physicochemical properties
- B32B7/023—Optical properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B7/00—Layered products characterised by the relation between layers; Layered products characterised by the relative orientation of features between layers, or by the relative values of a measurable parameter between layers, i.e. products comprising layers having different physical, chemical or physicochemical properties; Layered products characterised by the interconnection of layers
- B32B7/02—Physical, chemical or physicochemical properties
- B32B7/025—Electric or magnetic properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B7/00—Layered products characterised by the relation between layers; Layered products characterised by the relative orientation of features between layers, or by the relative values of a measurable parameter between layers, i.e. products comprising layers having different physical, chemical or physicochemical properties; Layered products characterised by the interconnection of layers
- B32B7/04—Interconnection of layers
- B32B7/06—Interconnection of layers permitting easy separation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B7/00—Layered products characterised by the relation between layers; Layered products characterised by the relative orientation of features between layers, or by the relative values of a measurable parameter between layers, i.e. products comprising layers having different physical, chemical or physicochemical properties; Layered products characterised by the interconnection of layers
- B32B7/04—Interconnection of layers
- B32B7/12—Interconnection of layers using interposed adhesives or interposed materials with bonding properties
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/34—Carboxylic acids; Esters thereof with monohydroxyl compounds
- C08G18/348—Hydroxycarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/42—Polycondensates having carboxylic or carbonic ester groups in the main chain
- C08G18/44—Polycarbonates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/72—Polyisocyanates or polyisothiocyanates
- C08G18/74—Polyisocyanates or polyisothiocyanates cyclic
- C08G18/75—Polyisocyanates or polyisothiocyanates cyclic cycloaliphatic
- C08G18/758—Polyisocyanates or polyisothiocyanates cyclic cycloaliphatic containing two or more cycloaliphatic rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/042—Coating with two or more layers, where at least one layer of a composition contains a polymer binder
- C08J7/0423—Coating with two or more layers, where at least one layer of a composition contains a polymer binder with at least one layer of inorganic material and at least one layer of a composition containing a polymer binder
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/044—Forming conductive coatings; Forming coatings having anti-static properties
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D163/00—Coating compositions based on epoxy resins; Coating compositions based on derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D175/00—Coating compositions based on polyureas or polyurethanes; Coating compositions based on derivatives of such polymers
- C09D175/04—Polyurethanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J7/00—Adhesives in the form of films or foils
- C09J7/40—Adhesives in the form of films or foils characterised by release liners
-
- G—PHYSICS
- G06—COMPUTING; CALCULATING OR COUNTING
- G06F—ELECTRIC DIGITAL DATA PROCESSING
- G06F3/00—Input arrangements for transferring data to be processed into a form capable of being handled by the computer; Output arrangements for transferring data from processing unit to output unit, e.g. interface arrangements
- G06F3/01—Input arrangements or combined input and output arrangements for interaction between user and computer
- G06F3/03—Arrangements for converting the position or the displacement of a member into a coded form
- G06F3/041—Digitisers, e.g. for touch screens or touch pads, characterised by the transducing means
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B13/00—Apparatus or processes specially adapted for manufacturing conductors or cables
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B13/00—Apparatus or processes specially adapted for manufacturing conductors or cables
- H01B13/0026—Apparatus for manufacturing conducting or semi-conducting layers, e.g. deposition of metal
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B5/00—Non-insulated conductors or conductive bodies characterised by their form
- H01B5/14—Non-insulated conductors or conductive bodies characterised by their form comprising conductive layers or films on insulating-supports
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/02—2 layers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/04—4 layers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/05—5 or more layers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/24—All layers being polymeric
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/02—Coating on the layer surface on fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/10—Coating on the layer surface on synthetic resin layer or on natural or synthetic rubber layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/20—Inorganic coating
- B32B2255/205—Metallic coating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/26—Polymeric coating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/28—Multiple coating on one surface
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/10—Inorganic fibres
- B32B2262/103—Metal fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2264/00—Composition or properties of particles which form a particulate layer or are present as additives
- B32B2264/10—Inorganic particles
- B32B2264/105—Metal
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2264/00—Composition or properties of particles which form a particulate layer or are present as additives
- B32B2264/10—Inorganic particles
- B32B2264/105—Metal
- B32B2264/1051—Silver or gold
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2264/00—Composition or properties of particles which form a particulate layer or are present as additives
- B32B2264/20—Particles characterised by shape
- B32B2264/204—Rod- or needle-shaped particles
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2264/00—Composition or properties of particles which form a particulate layer or are present as additives
- B32B2264/30—Particles characterised by physical dimension
- B32B2264/301—Average diameter smaller than 100 nm
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2264/00—Composition or properties of particles which form a particulate layer or are present as additives
- B32B2264/30—Particles characterised by physical dimension
- B32B2264/302—Average diameter in the range from 100 nm to 1000 nm
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2264/00—Composition or properties of particles which form a particulate layer or are present as additives
- B32B2264/30—Particles characterised by physical dimension
- B32B2264/304—Particle length
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/20—Properties of the layers or laminate having particular electrical or magnetic properties, e.g. piezoelectric
- B32B2307/202—Conductive
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/40—Properties of the layers or laminate having particular optical properties
- B32B2307/402—Coloured
- B32B2307/4023—Coloured on the layer surface, e.g. ink
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/40—Properties of the layers or laminate having particular optical properties
- B32B2307/412—Transparent
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/50—Properties of the layers or laminate having particular mechanical properties
- B32B2307/546—Flexural strength; Flexion stiffness
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/702—Amorphous
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/732—Dimensional properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/732—Dimensional properties
- B32B2307/737—Dimensions, e.g. volume or area
- B32B2307/7375—Linear, e.g. length, distance or width
- B32B2307/7376—Thickness
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/75—Printability
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2457/00—Electrical equipment
- B32B2457/20—Displays, e.g. liquid crystal displays, plasma displays
- B32B2457/208—Touch screens
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2345/00—Characterised by the use of homopolymers or copolymers of compounds having no unsaturated aliphatic radicals in side chain, and having one or more carbon-to-carbon double bonds in a carbocyclic or in a heterocyclic ring system; Derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2475/00—Characterised by the use of polyureas or polyurethanes; Derivatives of such polymers
- C08J2475/04—Polyurethanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2203/00—Applications of adhesives in processes or use of adhesives in the form of films or foils
- C09J2203/318—Applications of adhesives in processes or use of adhesives in the form of films or foils for the production of liquid crystal displays
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2301/00—Additional features of adhesives in the form of films or foils
- C09J2301/30—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier
- C09J2301/314—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier the adhesive layer and/or the carrier being conductive
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2301/00—Additional features of adhesives in the form of films or foils
- C09J2301/40—Additional features of adhesives in the form of films or foils characterized by the presence of essential components
- C09J2301/41—Additional features of adhesives in the form of films or foils characterized by the presence of essential components additives as essential feature of the carrier layer
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2423/00—Presence of polyolefin
- C09J2423/006—Presence of polyolefin in the substrate
Definitions
- the present invention relates to a transparent conductive film laminate and a processing method thereof, and particularly to a transparent conductive film laminate useful for suppressing curls generated during a heating step and a processing method thereof.
- PET film polyethylene terephthalate (PET) film has been widely used as a base film for a transparent conductive film constituting a capacitance type touch panel.
- PET film has high birefringence as it is formed by biaxial stretching, a phase difference occurs when the incident linearly polarized light passes through it. Therefore, it is difficult to use it under a polarizing plate. Therefore, a transparent conductive film using a cycloolefin resin is used as a base film for low retardation.
- a conductive layer or a protective layer is formed on a base film to produce a transparent conductive film
- a protective film is attached to the back surface to prevent damage to the base material during transportation, and a laminate is used.
- Patent Document 1 discloses a laminate in which both the substrate film of the transparent conductive film and the surface protective film (release film) are PET films. Curling is reduced by adjusting the heat shrinkage of the transparent conductive film with the amorphous transparent conductive thin film (ITO film) and the protective film (release film), but cycloolefin resin is used for the substrate film. No description or suggestion has been made regarding its use.
- Patent Document 2 describes a carrier film having an adhesive layer on at least one surface side of the protective film, and a transparent resin film and a transparent conductive film which are peelably laminated via the adhesive layer.
- the transparent resin film includes a film and the transparent resin film is made of an amorphous cycloolefin resin, and the protective film is formed of an amorphous resin having a glass transition temperature of 130 ° C. or higher, which is different from the transparent resin film.
- the transparent conductive film laminate is disclosed. Although the glass transition temperature and thickness of the transparent resin film and the protective film are adjusted to reduce curl during heating, the use of a thin transparent resin film and a PET protective film has been described and suggested. Absent.
- a cycloolefin resin film is used as the base film of the transparent conductive film and a PET film is used as the protective film, it is transparent during the heating process and after heating and cooling due to the difference in heat shrinkage and linear expansion coefficient between the two. Curling (warp) is likely to occur in the conductive film laminate.
- problems such as the warped transparent conductive film laminate coming into contact with the upper or lower part of the drying furnace occur, and it is difficult to perform stable and continuous production. It becomes.
- the step of manufacturing the touch panel using the transparent conductive film laminate the step of printing the wiring portion by screen printing or the like with silver paste or the like and heating the printed silver paste for drying and / or heat curing is performed. is there. If curl (warp) occurs in the transparent conductive film laminate during this heating, there arises a problem that the silver paste cannot be uniformly dried and / or thermoset, and a problem that the pattern accuracy becomes unstable. Yield deteriorates.
- Patent Document 1 In both Patent Document 1 and Patent Document 2, what is presented as a problem is curl remaining after heating and allowing to cool, and there is no mention of suppressing curl generated during heating. ..
- An object of the present invention is to suppress curling of the transparent conductive film laminate during and after the heating process when a thin-film cycloolefin resin is used as the base material of the transparent conductive film, and to secure the subsequent process yield. It is an object of the present invention to provide a possible transparent conductive film laminate and a method for processing the same.
- the present invention includes the following embodiments.
- a transparent conductive film laminate containing a carrier film and a transparent conductive film laminated on the carrier film The carrier film includes a protective film and an adhesive layer on one of the main surfaces thereof.
- the transparent conductive film is formed by laminating a transparent conductive layer containing a metal nanowire and a binder resin on one or both main surfaces of the transparent resin film and an overcoat layer in this order, and the transparent resin.
- the film is made of an amorphous cycloolefin resin, the thickness T 1 of the transparent resin film is 5 to 25 ⁇ m, and the transparent conductive film laminate is transparent so that the overcoat layer is the outermost layer.
- the conductive film is releasably laminated on the pressure-sensitive adhesive layer of the carrier film, the protective film is made of polyester having an aromatic ring in the molecular skeleton, and the thickness T 2 of the protective film is the thickness T of the transparent resin film.
- a transparent conductive film laminate having a size of 5 times or more and 150 ⁇ m or less of 1 .
- the present invention when a thin-film cycloolefin-based resin is used as the base material of the transparent conductive film, curling of the transparent conductive film laminate is suppressed during and after the heating step, and the subsequent process yield is ensured. It is possible to provide a possible transparent conductive film laminate and a processing method thereof.
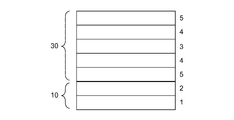
- the transparent conductive film laminate of the present invention is a transparent conductive film laminate including the carrier film 10 and the transparent conductive film 20 or 30 laminated on the carrier film 10.
- the carrier film 10 includes a protective film 1 and an adhesive layer 2 on one of the main surfaces thereof.
- the transparent conductive film 20 is composed of a transparent conductive layer 4 containing a metal nanowire and a binder resin on one main surface of the transparent resin film 3 and an overcoat layer 5 laminated in this order.
- Reference numeral 30 denotes a transparent conductive layer 4 containing a metal nanowire and a binder resin on both main surfaces of the transparent resin film 3, and an overcoat layer 5 are laminated in this order.
- the transparent resin film 3 is made of an amorphous cycloolefin resin, and the thickness T 1 of the transparent resin film is 5 to 25 ⁇ m.
- the transparent conductive film laminate the transparent conductive film is such that the overcoat layer (one overcoat layer in the case of a double-sided structure having two overcoat layers like the transparent conductive film 30) 5 is the outermost layer.
- the film 20 or 30 is releasably laminated on the pressure-sensitive adhesive layer 2 of the carrier film 10.
- the protective film 1 is made of polyester having an aromatic ring in its molecular skeleton, and the thickness T 2 of the protective film 1 is 5 times or more and 150 ⁇ m or less of the thickness T 1 of the transparent resin film 3.
- 1 and 2 show schematic cross-sectional views of the transparent conductive film laminate according to the embodiment.
- a transparent conductive layer 4 containing metal nanowires and a binder resin and an overcoat layer 5 are laminated on one main surface of the transparent resin film 3 in this order.
- the transparent conductive film laminate is a transparent conductive film laminate including the transparent conductive film 20 and the carrier film 10 laminated on the transparent conductive film 20, and the carrier film 10 is the protective film 1 and its carrier film 10.
- the pressure-sensitive adhesive layer 2 is provided on one main surface, and the transparent conductive film 20 includes a transparent conductive layer 4 containing a metal nanowire and a binder resin on one main surface of the transparent resin film 3, and an overcoat layer 5.
- the carrier film 10 is laminated in this order, and the carrier film 10 is releasably laminated on the main surface of the transparent resin film 3 on the opposite side of the transparent conductive layer 4 via the pressure-sensitive adhesive layer 2.
- the transparent conductive layer 4 containing the metal nanowires and the binder resin and the overcoat layer 5 are laminated on both main surfaces of the transparent resin film 3 in this order.
- the transparent conductive film laminate is a transparent conductive film laminate including the transparent conductive film 30 and the carrier film 10 laminated on the transparent conductive film 30, and the carrier film 10 is the protective film 1 and its carrier film 10.
- the pressure-sensitive adhesive layer 2 is provided on one main surface, and the transparent conductive film 30 includes a transparent conductive layer 4 containing a metal nanowire and a binder resin on both main surfaces of the transparent resin film 3, and an overcoat layer 5.
- the carrier film 10 is detachably laminated on the surface of one of the overcoat layers 5 of the transparent conductive film 30 via the pressure-sensitive adhesive layer 2.
- the transparent resin film 3 is formed of an amorphous cycloolefin resin, and has high transparency and low water absorption.
- an amorphous cycloolefin-based resin film as the transparent resin film 3, a transparent conductive film laminate having a transparent conductive film having good optical characteristics can be obtained.
- the amorphous cycloolefin resin is a (co) polymer containing 50 mol% or more of cycloolefin such as norbornene, and is a hydride ring-opening metathesis polymerized cycloolefin polymer of norbornene (ZEONOR®, registered trademark, Nippon Zeon Co., Ltd.). , ZEONEX (registered trademark, manufactured by Nippon Zeon), ARTON (registered trademark, manufactured by JSR), norbornene / ethylene-added copolymer cycloolefin polymer (APEL (registered trademark, manufactured by Mitsui Chemicals), TOPAS ( Registered trademark, manufactured by Polyplastics Co., Ltd.)) can be used.
- ZEONOR® registered trademark, Nippon Zeon Co., Ltd.
- ZEONEX registered trademark, manufactured by Nippon Zeon
- ARTON registered trademark, manufactured by JSR
- APEL registered trademark, manufactured by Mitsui Chemical
- the term "transparent" means that the total light transmittance is 70% or more.
- the surface of the transparent resin film 3 may be subjected to a treatment such as plasma treatment or corona treatment in advance to improve the adhesion of the transparent conductive layer 4 or the like formed on the transparent resin film 3. Further, before forming the transparent conductive layer 4, the surface of the transparent resin film 3 may be dust-removed and cleaned by solvent cleaning, ultrasonic cleaning, or the like, if necessary.
- a treatment such as plasma treatment or corona treatment in advance to improve the adhesion of the transparent conductive layer 4 or the like formed on the transparent resin film 3.
- the surface of the transparent resin film 3 may be dust-removed and cleaned by solvent cleaning, ultrasonic cleaning, or the like, if necessary.
- the thickness T 1 of the transparent resin film 3 is in the range of 5 to 25 ⁇ m, preferably in the range of 10 to 23 ⁇ m, and more preferably in the range of 13 to 23 ⁇ m. If the thickness of the transparent resin film 3 exceeds the upper limit of the above range, cracks and the like are likely to occur during bending. On the other hand, if the thickness is less than the lower limit of the above range, it is difficult to produce a film having smoothness and uniform thickness.
- the glass transition temperature (Tg) of the amorphous cycloolefin resin of the transparent resin film 3 is not particularly limited, but is preferably in the range of 130 to 170 ° C. As a result, the amount and direction of curl generated after the heating step such as drying can be controlled, so that the transparent conductive film laminate can be easily processed and conveyed.
- metal nanowires can be preferably used as the conductive material constituting the transparent conductive layer 4 formed on the transparent resin film 3.
- the metal nanowire is a metal having a diameter on the order of nanometers, and is a conductive material having a wire-like shape.
- metal nanotubes which are conductive materials having a porous or non-porous tubular shape, may be used together with (mixed) metal nanowires or instead of metal nanowires.
- both “wire-like” and “tube-like” are linear, but the former is intended to have a hollow center and the latter to be hollow in the center.
- the properties may be flexible or rigid.
- metal nanowires in a narrow sense The former is referred to as “metal nanowires in a narrow sense” and the latter is referred to as “metal nanotubes in a narrow sense”.
- metal nanowires are used in the meaning of including metal nanowires in a narrow sense and metal nanotubes in a narrow sense.
- Metal nanowires in a narrow sense and metal nanotubes in a narrow sense may be used alone or in combination.
- the "transparent conductive layer” is a thin film-like layer containing the above-mentioned metal nanowires and a binder resin described later, and is not necessarily limited to having a uniform thickness.
- the thickness is preferably in the range of 20 to 200 nm.
- metal nanowires As a method for producing metal nanowires, a known production method can be used. For example, silver nanowires can be synthesized by reducing silver nitrate in the presence of poly-N-vinylpyrrolidone using the Poly-ol method (see Chem. Matter., 2002, 14, 4736). Gold nanowires can also be similarly synthesized by reducing chloroauric acid hydrate in the presence of polyvinylpyrrolidone (see J. Am. Chem. Soc., 2007, 129, 1733). The techniques for large-scale synthesis and purification of silver nanowires and gold nanowires are described in detail in International Publication No. 2008/073143 and International Publication No. 2008/046058.
- Gold nanotubes having a porous structure can be synthesized by reducing a gold chloride solution using silver nanowires as a template.
- the silver nanowires used in the template dissolve in the solution by a redox reaction with chloroauric acid, resulting in gold nanotubes having a porous structure (JAm. Chem. Soc., 2004, 126, 3892). See -3901).
- the average diameter (average diameter) of the metal nanowires is preferably 1 to 500 nm, more preferably 5 to 200 nm, further preferably 5 to 100 nm, and particularly preferably 10 to 50 nm.
- the average length (average length) of the major axis of the metal nanowire is preferably 1 to 100 ⁇ m, more preferably 1 to 80 ⁇ m, further preferably 2 to 70 ⁇ m, and particularly preferably 5 to 50 ⁇ m.
- the average diameter and the average length of the major axis satisfy the above range, and the average aspect ratio is preferably larger than 5, more preferably 10 or more, and more than 100. It is more preferable, and it is particularly preferable that it is 200 or more.
- the aspect ratio is a value obtained by a / b when the average diameter of the metal nanowire is approximated to b and the average length of the major axis is approximated to a.
- a and b can be measured using a scanning electron microscope (SEM) and an optical microscope. Specifically, for b (average diameter), the diameter of 100 arbitrarily selected silver nanowires was measured using a field emission scanning electron microscope JSM-7000F (manufactured by JEOL Ltd.), and the arithmetic mean value was used. Can be sought.
- the shape measurement laser microscope VK-X200 manufactured by Keyence Co., Ltd. was used to calculate a (average length), and the lengths of 100 arbitrarily selected silver nanowires were measured, and the arithmetic mean value thereof was measured. Can be obtained as.
- such a metal nanowire As the material of such a metal nanowire, at least one selected from the group consisting of gold, silver, platinum, copper, nickel, iron, cobalt, zinc, ruthenium, rhodium, palladium, cadmium, osmium, and iridium, and metals thereof. Examples include alloys that combine the above.
- Optimal embodiments include silver nanowires.
- the transparent conductive layer 4 contains a binder resin in addition to the metal nanowires.
- the binder resin can be applied without limitation as long as it has transparency, but when a metal nanowire using the polyol method is used as the conductive material, from the viewpoint of compatibility with the manufacturing solvent (polyol), the binder resin can be applied without limitation. It is preferable to use a binder resin that is soluble in alcohol or water. Specifically, water-soluble cellulosic resins such as poly-N-vinylpyrrolidone, methylcellulose, hydroxyethylcellulose, and carboxymethylcellulose, butyral resin, and poly-N-vinylacetamide (PNVA (registered trademark)) can be used.
- PNVA poly-N-vinylacetamide
- Poly-N-vinylacetamide is a homopolymer of N-vinylacetamide (NVA), but a copolymer containing 70 mol% or more of N-vinylacetamide (NVA) can also be used.
- NVA N-vinylacetamide
- Examples of the monomer copolymerizable with NVA include N-vinylformamide, N-vinylpyrrolidone, acrylic acid, methacrylic acid, sodium acrylate, sodium methacrylate, acrylamide, acrylonitrile and the like.
- the monomer unit derived from N-vinylacetamide is preferably contained in the polymer in an amount of 70 mol% or more, more preferably 80 mol% or more. It is more preferable to contain 90 mol% or more.
- Polymers (homomopolymers and copolymers) containing N-vinylacetamide as a monomer unit preferably have a weight average molecular weight of 30,000 to 4 million, more preferably 100,000 to 3 million. It is more preferably 300,000 to 1,500,000.
- the weight average molecular weight based on the absolute molecular weight is measured by the following method.
- the binder resin was dissolved in the following eluent and allowed to stand for 20 hours.
- the concentration of the binder resin in this solution is 0.05% by mass. This was filtered through a 0.45 ⁇ m membrane filter, and the filtrate was measured by GPC-MALS to calculate the weight average molecular weight based on the absolute molecular weight.
- GPC Showa Denko Corporation Shodex (registered trademark) SYSTEM21
- Multi-angle fit method Berry method
- the transparent conductive layer 4 is printed on one or both main surfaces of the transparent resin film 3 using the conductive ink (metal nanowire ink) containing the metal nanowire, the binder resin and the solvent as a coating liquid, and the solvent is dried and removed. Formed by
- the solvent is not particularly limited as long as the metal nanowires exhibit good dispersibility and the binder resin dissolves in the solvent.
- the solvent for producing the metal nanowires From the viewpoint of compatibility with polyol, alcohol, water or a mixed solvent of alcohol and water is preferable.
- a saturated monohydric alcohol (methanol, ethanol, normal propanol and isopropanol) having 1 to 3 carbon atoms represented by C n H 2n + 1 OH (n is an integer of 1 to 3) [hereinafter, simply "carbon”. Notated as “saturated monohydric alcohol with 1 to 3 atomic numbers"]. It is preferable to contain 40% by mass or more of saturated monohydric alcohol having 1 to 3 carbon atoms in the total alcohol. Using a saturated monohydric alcohol having 1 to 3 carbon atoms facilitates drying, which is convenient in the process.
- an alcohol other than the saturated monohydric alcohol having 1 to 3 carbon atoms represented by C n H 2n + 1 OH (n is an integer of 1 to 3) can also be used in combination.
- examples of alcohols other than saturated monohydric alcohols having 1 to 3 carbon atoms that can be used in combination include ethylene glycol, propylene glycol, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, propylene glycol monomethyl ether, and propylene glycol monoethyl ether.
- the drying rate can be adjusted by using these in combination with the saturated monohydric alcohol having 1 to 3 carbon atoms represented by the above C n H 2n + 1 OH (n is an integer of 1 to 3).
- the total alcohol content in the mixed solvent is preferably 5 to 90% by mass. If the content of alcohol in the mixed solvent is less than 5% by mass or more than 90% by mass, a striped pattern (coating spot) may occur when coating, which may be inappropriate.
- the conductive ink can be produced by stirring and mixing the metal nanowires, the binder resin and the solvent with a rotating revolution stirrer or the like.
- the content of the binder resin contained in the conductive ink is preferably in the range of 0.01 to 1.0% by mass.
- the content of the metal nanowires contained in the conductive ink is preferably in the range of 0.01 to 1.0% by mass.
- the content of the solvent contained in the conductive ink is preferably in the range of 98.0 to 99.98% by mass.
- a conductive ink having a viscosity of 1 to 50 mPa ⁇ s can be obtained, which is printed on the main surface of the transparent resin film 3 and the solvent is dried and removed to obtain a transparent conductive ink having a thickness of 20 to 200 nm.
- Layer 4 is obtained.
- the more preferable viscosity of the conductive ink is 1 to 20 mPa ⁇ s, and the more preferable viscosity is 1 to 10 mPa ⁇ s.
- the viscosity is a value measured at 25 ° C. with a digital viscometer DV-E (spindle: SC4-18) manufactured by Brookfield.
- a printing method such as a bar coat printing method, a gravure printing method, an inkjet method, or a slit coating method can be applied to the manufacturing methods (conducting ink printing method) of the transparent conductive films 20 and 30.
- the bar coat printing method has good coatability of low-viscosity ink and is excellent in forming a thin film.
- the bar coat printing method can print low-viscosity ink containing inorganic or metal particles without clogging.
- the overcoat layer 5 that protects the transparent conductive layer 4 is preferably a cured film of a curable resin composition.
- the curable resin composition preferably contains (A) a polyurethane containing a carboxy group, (B) an epoxy compound, (C) a curing accelerator, and (D) a solvent.
- the curable resin composition is formed on the transparent conductive layer 4 by printing, coating, or the like, and cured to form the overcoat layer 5. Curing of the curable resin composition can be performed by heating and drying the thermosetting resin composition.
- the weight average molecular weight of the polyurethane (A) containing the carboxy group is preferably 1,000 to 100,000, more preferably 2,000 to 70,000, and 3,000 to 50, It is more preferably 000.
- the molecular weight is a polystyrene-equivalent value measured by gel permeation chromatography (hereinafter referred to as GPC). If the molecular weight is less than 1,000, the elongation, flexibility, and strength of the coating film after printing may be impaired, and if it exceeds 100,000, the solubility of polyurethane in the solvent becomes low and the polyurethane dissolves. However, the viscosity becomes too high, which may increase restrictions on use.
- the measurement conditions of GPC are as follows.
- Device name HPLC unit HSS-2000 manufactured by JASCO Corporation
- Detector RI-2031Plus manufactured by JASCO Corporation Temperature: 40.0 ° C
- Sample amount Sample loop 100 ⁇ L
- Sample concentration Prepared to about 0.1% by mass
- the acid value of the polyurethane (A) containing a carboxy group is preferably 10 to 140 mg-KOH / g, and more preferably 15 to 130 mg-KOH / g.
- the acid value is 10 mg-KOH / g or more, both curability and solvent resistance are good.
- it is 140 mg-KOH / g or less, the solubility in a solvent as polyurethane is good, and it is easy to adjust the viscosity to a desired value.
- problems such as warpage of the base film due to the cured product becoming too hard are less likely to occur.
- Acid value (mg-KOH / g) [B ⁇ f ⁇ 5.611] / S B: Amount of 0.1N potassium hydroxide-ethanol solution used (mL) f: Factor S of 0.1N potassium hydroxide-ethanol solution: Sample collection amount (g)
- the polyurethane (A) containing a carboxy group is more specifically a polyurethane synthesized using (a1) a polyisocyanate compound, (a2) a polyol compound, and (a3) a dihydroxy compound having a carboxy group as a monomer. is there. From the viewpoint of light resistance, it is desirable that (a1), (a2), and (a3) do not contain a functional group having conjugation such as an aromatic compound. Hereinafter, each monomer will be described in more detail.
- polyisocyanate compound As the polyisocyanate compound, diisocyanate having two isocyanato groups per molecule is usually used. Examples of the polyisocyanate compound include aliphatic polyisocyanates and alicyclic polyisocyanates, and one of these compounds can be used alone or in combination of two or more. (A) A small amount of polyisocyanate having 3 or more isocyanato groups can be used as long as the polyurethane containing a carboxy group does not gel.
- Examples of the aliphatic polyisocyanate include 1,3-trimethylene diisocyanate, 1,4-tetramethylene diisocyanate, 1,6-hexamethylene diisocyanate, 1,9-nonamethylene diisocyanate, and 1,10-decamethylene diisocyanate, and 2, , 2,4-trimethylhexamethylene diisocyanate, 2,4,4-trimethylhexamethylene diisocyanate, lysine diisocyanate, 2,2'-diethyleterdiisocyanate, dimerate diisocyanate and the like.
- Examples of the alicyclic polyisocyanate include 1,4-cyclohexanediisocyanate, 1,3-bis (isocyanatomethyl) cyclohexane, 1,4-bis (isocyanatomethyl) cyclohexane, and 3-isocyanatomethyl-3,3. , 5-trimethylcyclohexane (IPDI, isophorone diisocyanate), bis- (4-isocyanatocyclohexyl) methane (hydrogenated MDI), hydrogenated (1,3- or 1,4-) xylylene diisocyanate, norbornandiisocyanate, etc. Be done.
- the polyurethane resin according to the embodiment is used by using an alicyclic compound having 6 to 30 carbon atoms other than carbon atoms in the isocyanato group (-NCO group).
- the protective film formed from is highly reliable especially at high temperature and high humidity, and is suitable for members of electronic device parts.
- the aromatic polyisocyanate and the aromatic aliphatic polyisocyanate are 50 mol% or less, preferably 30 mol% or less, based on the total amount (100 mol%) of the (a1) polyisocyanate compound in the (a1) polyisocyanate compound. More preferably, it may be contained in an amount of 10 mol% or less.
- the number average molecular weight of (a2) polyol compound (a2) polyol compound is usually 250 to 50,000. Yes, preferably 400-10,000, more preferably 500-5,000. This molecular weight is a polystyrene-equivalent value measured by GPC under the above-mentioned conditions.
- the polyol compound (a2) is preferably a diol having a hydroxy group at both ends.
- polyvalent values derived from C18 (18 carbon atoms) unsaturated fatty acids made from polycarbonate polyols, polyether polyols, polyester polyols, polylactone polyols, hydroxylated polysilicones at both ends, and vegetable fats and oils and their polymers.
- It is a polyol compound having 18 to 72 carbon atoms obtained by hydrogenating a carboxylic acid to convert the carboxylic acid into a hydroxyl group.
- polycarbonate polyol is preferable in consideration of the balance between water resistance as a protective film, insulation reliability, and adhesion to a base material.
- the polycarbonate polyol can be obtained by reacting a diol having 3 to 18 carbon atoms with a carbonic acid ester or phosgene, and is represented by, for example, the following structural formula (1).
- R 3 is a residue obtained by removing the hydroxyl group from the corresponding diol (HO-R 3- OH) and is an alkylene group having 3 to 18 carbon atoms, and n 3 is a positive integer, preferably a positive integer. Is 2 to 50.
- the polycarbonate polyol represented by the formula (1) is 1,3-propanediol, 1,4-butanediol, 1,5-pentanediol, 1,6-hexanediol, or 3-methyl-1.
- 5-Pentanediol, 1,8-octanediol, 1,3-cyclohexanedimethanol, 1,4-cyclohexanedimethanol, 1,9-nonanediol, 2-methyl-1,8-octanediol, 1,10 -It can be produced by using decamethylene glycol, 1,2-tetradecanediol or the like as a raw material.
- the above-mentioned polycarbonate polyol may be a polycarbonate polyol (copolymerized polycarbonate polyol) having a plurality of types of alkylene groups in its skeleton.
- the use of a copolymerized polycarbonate polyol is often advantageous from the viewpoint of preventing crystallization of the polyurethane (A) containing a carboxy group. Further, considering the solubility in a solvent, it is preferable to use a polycarbonate polyol having a branched skeleton and a hydroxyl group at the end of the branched chain in combination.
- the above-mentioned polyether polyol is obtained by dehydration condensation of a diol having 2 to 12 carbon atoms or ring-opening polymerization of an oxylan compound, an oxetane compound, or a tetrahydrofuran compound having 2 to 12 carbon atoms, for example. It is represented by the following structural formula (2).
- R 4 is a residue obtained by removing the hydroxyl group from the corresponding diol (HO-R 4- OH) and is an alkylene group having 2 to 12 carbon atoms, and n 4 is a positive integer, preferably a positive integer. Is 4 to 50.
- the diol having 2 to 12 carbon atoms may be used alone to form a homopolymer, or may be used in combination of two or more to form a copolymer.
- polyether polyol represented by the above formula (2) examples include polyethylene glycol, polypropylene glycol, poly-1,2-butylene glycol, polytetramethylene glycol (poly1,4-butanediol), and the like.
- examples thereof include polyalkylene glycols such as poly-3-methyltetramethylene glycol and polyneopentyl glycol.
- a copolymer of these for example, a copolymer of 1,4-butanediol and neopentyl glycol can also be used.
- the polyester polyol is obtained by dehydration condensation of a dicarboxylic acid and a diol or a transesterification reaction of a lower alcohol esterified product of the dicarboxylic acid with a diol, and is represented by, for example, the following structural formula (3).
- R 5 is a residue obtained by removing the hydroxyl group from the corresponding diol (HO-R 5- OH), and is an alkylene group or an organic group having 2 to 10 carbon atoms, and R 6 corresponds to the corresponding diol.
- diol examples include ethylene glycol, 1,2-propanediol, 1,3-propanediol, 1,2-butanediol, 1,3-butanediol, and 1 , 4-butanediol, 1,5-pentanediol, 1,6-hexanediol, 3-methyl-1,5-pentanediol, 1,8-octanediol, 1,3-cyclohexanedimethanol, 1,4- Cyclohexanedimethanol, 1,9-nonanediol, 2-methyl-1,8-octanediol, 1,10-decamethylene glycol or 1,2-tetradecanediol, 2,4-diethyl-1,5-pentanediol, Examples thereof include butyl ethyl propanediol, 1,3-cyclohexanedimethanol, diethylene glycol, 1,2-propanedio
- dicarboxylic acid examples include succinic acid, glutaric acid, adipic acid, azelaic acid, sebacic acid, decandicarboxylic acid, brassic acid, and 1,4-cyclohexanedicarboxylic acid.
- succinic acid glutaric acid, adipic acid, azelaic acid, sebacic acid, decandicarboxylic acid, brassic acid, and 1,4-cyclohexanedicarboxylic acid.
- Hexahydrophthalic acid methyltetrahydrophthalic acid, endomethylenetetrahydrophthalic acid, methylendomethylenetetrahydrophthalic acid, chlorendic acid, fumaric acid, maleic acid, itaconic acid, citraconic acid.
- the polylactone polyol is obtained by a condensation reaction of a ring-opening polymer of a lactone with a diol or a condensation reaction of a diol and a hydroxyalkanoic acid, and is represented by, for example, the following structural formula (4).
- R 7 is a residue obtained by removing hydroxyl groups and carboxy groups from the corresponding hydroxyalkanoic acid (HO-R 7- COOH) and is an alkylene group having 4 to 8 carbon atoms
- R 8 is It is a residue obtained by removing the hydroxyl group from the corresponding diol (HO-R 8- OH) and is an alkylene group having 2 to 10 carbon atoms
- n 6 is a positive integer, preferably 2 to 50.
- hydroxyalkanoic acid examples include 3-hydroxybutanoic acid, 4-hydroxypentanoic acid, 5-hydroxyhexanoic acid and the like.
- lactone examples include ⁇ -caprolactone.
- the bi-terminal hydroxyl grouped polysilicone is represented by, for example, the following structural formula (5).
- R 9 is independently an aliphatic hydrocarbon divalent residue having 2 to 50 carbon atoms, and n 7 is a positive integer, preferably 2 to 50.
- R 9 may contain an ether group, and a plurality of R 10 are independently aliphatic hydrocarbon groups having 1 to 12 carbon atoms.
- Examples of commercially available products of the above-mentioned double-ended hydroxyl group-coupled polysilicone include "X-22-160AS, KF6001, KF6002, KF-6003" manufactured by Shin-Etsu Chemical Co., Ltd.
- polypoly compound having 18 to 72 carbon atoms obtained by hydrogenating a C18 unsaturated fatty acid made from a plant-based fat or oil and a polyvalent carboxylic acid derived from a polymer thereof to convert the carboxylic acid into a hydroxyl group Specifically, a diol compound having a skeleton obtained by hydrogenating dimer acid can be mentioned, and examples of a commercially available product thereof include "Sovermol (registered trademark) 908" manufactured by Cognis.
- a diol having a molecular weight of 300 or less which is usually used as a diol component when synthesizing polyester or polycarbonate as the polyol compound, can also be used.
- specific examples of such low molecular weight diols include ethylene glycol, 1,2-propanediol, 1,3-propanediol, 1,2-butanediol, 1,3-butanediol, and 1,4-butane.
- Diol 1,5-pentanediol, 1,6-hexanediol, 3-methyl-1,5-pentanediol, 1,8-octanediol, 1,3-cyclohexanedimethanol, 1,4-cyclohexanedimethanol, 1,9-Nonandiol, 2-methyl-1,8-octanediol, 1,10-decamethylene glycol, 1,2-tetradecanediol, 2,4-diethyl-1,5-pentanediol, butylethylpropanediol , 1,3-Cyclohexanedimethanol, diethylene glycol, triethylene glycol, dipropylene glycol and the like.
- (A3) Dihydroxy compound containing a carboxy group (a3)
- the dihydroxy compound containing a carboxy group has a molecular weight of 200 or less having two of a hydroxy group and a hydroxyalkyl group having 1 or 2 carbon atoms.
- the carboxylic acid or aminocarboxylic acid of the above is preferable because the cross-linking point can be controlled. Specific examples thereof include 2,2-dimethylolpropionic acid, 2,2-dimethyrolbutanoic acid, N, N-bishydroxyethylglycine, N, N-bishydroxyethylalanine, and the like. 2,2-Dimethylolpropionic acid and 2,2-dimethyrolbutanoic acid are particularly preferable because of their solubility.
- These (a3) carboxy group-containing dihydroxy compounds can be used alone or in combination of two or more.
- the polyurethane containing the (A) carboxy group described above can be synthesized only from the above three components ((a1), (a2) and (a3)). Further, it can also be synthesized by reacting (a4) a monohydroxy compound and / or (a5) a monoisocyanate compound. From the viewpoint of light resistance, it is preferable to use a compound that does not contain an aromatic ring or a carbon-carbon double bond in the molecule.
- Monohydroxy Compound (a4) examples include compounds having a carboxylic acid such as glycolic acid and hydroxypivalic acid.
- the monohydroxy compound can be used alone or in combination of two or more.
- (a4) monohydroxy compound examples include methanol, ethanol, n-propanol, isopropanol, n-butanol, isobutanol, sec-butanol, t-butanol, amyl alcohol, hexyl alcohol, and octyl alcohol.
- Monoisocyanate compound (a5) examples include hexyl isocyanate and dodecyl isocyanate.
- the polyurethane containing the (A) carboxy group can be obtained from the above-mentioned (a1) polyisocyanate compound, (a1), in the presence or absence of a known urethanization catalyst such as dibutyltin dilaurylate, using an appropriate organic solvent. It can be synthesized by reacting a2) a polyol compound and (a3) a dihydroxy compound having a carboxy group, but it is preferable to react without a catalyst without considering the final contamination of tin and the like.
- the organic solvent is not particularly limited as long as it has low reactivity with the isocyanate compound, but does not contain a basic functional group such as amine and has a boiling point of 50 ° C. or higher, preferably 80 ° C. or higher, more preferably 100 ° C. or higher. Certain solvents are preferred. Examples of such a solvent include toluene, xylene, ethylbenzene, nitrobenzene, cyclohexane, isophorone, diethylene glycol dimethyl ether, ethylene glycol diethyl ether, ethylene glycol monomethyl ether acetate, propylene glycol monomethyl ether acetate, and the like.
- Examples thereof include n-butyl, isoamyl acetate, ethyl lactate, acetone, methyl ethyl ketone, cyclohexanone, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, ⁇ -butyrolactone, and dimethylsulfoxide.
- polyurethane is used as a raw material for a curable resin composition for the overcoat layer 5 in electronic material applications, among these, particularly Propylene glycol monomethyl ether acetate, propylene glycol monoethyl ether acetate, dipropylene glycol monomethyl ether acetate, diethylene glycol monoethyl ether acetate, ⁇ -butyrolactone and the like are preferable.
- the order in which the raw materials are charged is not particularly limited, but usually, (a2) a polyol compound and (a3) a dihydroxy compound having a carboxy group are charged first, dissolved or dispersed in a solvent, and then 20 to 150 ° C.
- the polyisocyanate compound (a1) is added dropwise at 60 to 120 ° C., and then reacted at 30 to 160 ° C., more preferably 50 to 130 ° C.
- the molar ratio of the raw material is adjusted according to the molecular weight and acid value of the target polyurethane, but when the (a4) monohydroxy compound is introduced into the polyurethane, the end of the polyurethane molecule becomes an isocyanate group. It is necessary to use (a1) a polyisocyanate compound in excess of (a2) a polyol compound and (a3) a dihydroxy compound having a carboxy group (so that the isocyanato group is in excess of the total number of hydroxyl groups).
- the (a1) polyisocyanate compound is more than the (a2) polyol compound and the (a3) dihydroxy compound having a carboxy group so that the end of the polyurethane molecule becomes a hydroxy group. Should be used less (so that there are fewer isocyanato groups than the total number of hydroxyl groups).
- the molar ratio of these charges is as follows: (a1) isocyanato group of polyisocyanate compound: ((a2) hydroxyl group of polyol compound + hydroxyl group of dihydroxy compound having (a3) carboxy group) is 0.5 to 1. It is .5: 1, preferably 0.8 to 1.2: 1, more preferably 0.95 to 1.05: 1.
- the hydroxyl group of the (a2) polyol compound the hydroxyl group of the dihydroxy compound having the (a3) carboxy group is 1: 0.1 to 30, preferably 1: 0.3 to 10.
- the number of moles of the (a1) polyisocyanate compound is excessively larger than the number of moles of ((a2) polyol compound + (a3) dihydroxy compound having a carboxy group), and (a4) It is preferable to use the monohydroxy compound in an amount of 0.5 to 1.5 times, preferably 0.8 to 1.2 times, the molar amount of the excess molar number of the isocyanate group.
- the number of moles of ((a2) polyol compound + (a3) dihydroxy compound having a carboxy group) is excessively larger than the number of moles of (a1) polyisocyanate compound, and (a5) It is preferable to use the monoisocyanate compound in an amount of 0.5 to 1.5 times, preferably 0.8 to 1.2 times, the molar amount of the excess number of hydroxyl groups.
- Examples of the (B) epoxy compound include bisphenol A type epoxy compound, hydrogenated bisphenol A type epoxy resin, bisphenol F type epoxy resin, novolac type epoxy resin, phenol novolac type epoxy resin, cresol novolac type epoxy resin, and N-glycidyl type.
- Epoxy resin bisphenol A novolak type epoxy resin, chelate type epoxy resin, glioxal type epoxy resin, amino group containing epoxy resin, rubber modified epoxy resin, dicyclopentadiene phenolic type epoxy resin, silicone modified epoxy resin, ⁇ -caprolactone modified epoxy
- examples thereof include an epoxy compound having two or more epoxy groups in one molecule, such as a resin, an aliphatic epoxy resin containing a glycidyl group, and an alicyclic epoxy resin containing a glycidyl group.
- an epoxy compound having three or more epoxy groups in one molecule can be used more preferably.
- examples of such epoxy compounds include EHPE (registered trademark) 3150 (manufactured by Daicel Chemical Co., Ltd.), jER (registered trademark) 604 (manufactured by Mitsubishi Chemical Co., Ltd.), EPICLON (registered trademark) EXA-4700 (manufactured by DIC), and the like.
- EPICLON (registered trademark) HP-7200 (manufactured by DIC), pentaerythritol tetraglycidyl ether, pentaerythritol triglycidyl ether, TEPIC (registered trademark) -S (manufactured by Nissan Chemical Co., Ltd.) and the like can be mentioned.
- the epoxy compound (B) may have an aromatic ring in the molecule, and in that case, the mass of (B) is 20% by mass or less with respect to the total mass of (A) and (B). preferable.
- the compounding ratio of the polyurethane containing the (A) carboxy group to the (B) epoxy compound is 0.5 to 1.5, which is the equivalent ratio of the carboxy group in the polyurethane to the epoxy group of the (B) epoxy compound. It is preferably 0.7 to 1.3, more preferably 0.9 to 1.1.
- Examples of the above (C) curing accelerator include phosphine compounds such as triphenylphosphine and tributylphosphine (manufactured by Hokuko Kagaku Co., Ltd.), curesol (registered trademark) (imidazole-based epoxy resin curing agent: manufactured by Shikoku Kasei Co., Ltd.), and 2-phenyl. Examples thereof include -4-methyl-5-hydroxymethylimidazole, U-CAT (registered trademark) SA series (DBU salt: manufactured by San-Apro), Irgacure (registered trademark) 184 and the like.
- phosphine compounds such as triphenylphosphine and tributylphosphine (manufactured by Hokuko Kagaku Co., Ltd.), curesol (registered trademark) (imidazole-based epoxy resin curing agent: manufactured by Shikoku Kasei Co., Ltd.), and 2-phenyl. Examples thereof include -4-methyl-5-hydroxy
- the total of 100 parts by mass of (A) and (B) is 0. 1 to 10 parts by mass, more preferably 0.5 to 6 parts by mass, still more preferably 0.5 to 5 parts by mass, and particularly preferably 0.5 to 3 parts by mass.
- a curing aid may be used in combination.
- the curing aid include polyfunctional thiol compounds and oxetane compounds.
- the polyfunctional thiol compound include pentaerythritol tetrakis (3-mercaptopropionate), tris-[(3-mercaptopropionyloxy) -ethyl] -isocyanurate, trimethylolpropane tris (3-mercaptopropionate), and Karenz. (Registered trademark) MT series (manufactured by Showa Denko Co., Ltd.) and the like.
- oxetane compound examples include Aron Oxetane (registered trademark) series (manufactured by Toagosei Co., Ltd.), ETERNCOLL (registered trademark) OXBP and OXMA (manufactured by Ube Industries, Ltd.).
- Aron Oxetane registered trademark
- ETERNCOLL registered trademark
- OXBP registered trademark
- OXMA manufactured by Ube Industries, Ltd.
- the curable resin composition preferably contains the solvent (D) in an amount of 95.0% by mass or more and 99.9% by mass or less, more preferably 96% by mass or more and 99.7% by mass or less, and 97% by mass or more. It is more preferable to contain 99.5% by mass or less.
- the solvent (D) the solvent used for synthesizing the polyurethane (A) containing a carboxy group can be used as it is, or another solvent can be used to adjust the solubility and printability of the polyurethane. it can. When another solvent is used, the reaction solvent may be distilled off before and after the addition of the new solvent to replace the solvent.
- the boiling point of the solvent is preferably 80 ° C. to 300 ° C., more preferably 80 ° C. to 250 ° C.
- the boiling point is less than 80 ° C., it tends to dry during printing and unevenness is likely to occur.
- the boiling point is higher than 300 ° C., it is not suitable for industrial production because it requires a long heat treatment at a high temperature during drying and curing.
- Examples of such a solvent include propylene glycol monomethyl ether acetate (boiling point of 146 ° C.), ⁇ -butyrolactone (boiling point of 204 ° C.), diethylene glycol monoethyl ether acetate (boiling point of 218 ° C.), tripropylene glycol dimethyl ether (boiling point of 243 ° C.) and the like.
- Solvents used for polyurethane synthesis ether-based solvents such as propylene glycol dimethyl ether (boiling point 97 ° C.), diethylene glycol dimethyl ether (boiling point 162 ° C.), isopropyl alcohol (boiling point 82 ° C.), t-butyl alcohol (boiling point 82 ° C.), 1 -Hexanol (boiling point 157 ° C), propylene glycol monomethyl ether (boiling point 120 ° C.), diethylene glycol monomethyl ether (boiling point 194 ° C.), diethylene glycol monoethyl ether (boiling point 196 ° C.), diethylene glycol monobutyl ether (boiling point 230 ° C.), triethylene glycol (boiling temperature 230 ° C.)
- a solvent containing a hydroxyl group such as ethyl lactate (boiling point
- solvents may be used alone or in combination of two or more. When two or more types are mixed, aggregation and precipitation occur in consideration of the solubility of the polyurethane resin, epoxy resin, etc. used in addition to the solvent used for the synthesis of (A) carboxy group-containing polyurethane. It is preferable to use a solvent having a hydroxy group and having a boiling point of more than 100 ° C. or a solvent having a boiling point of 100 ° C. or less from the viewpoint of drying property of the ink.
- the curable resin composition contains the above-mentioned (A) carboxy group-containing polyurethane, (B) epoxy compound, (C) curing accelerator, (D) solvent, and (D) solvent content. It can be produced by blending so as to be 95.0% by mass or more and 99.9% by mass or less and stirring so as to be uniform.
- the solid content concentration in such a curable resin composition varies depending on the desired film thickness and printing method, but is preferably 0.1 to 5.0% by mass, preferably 0.5% by mass to 3.0%. More preferably, it is by mass%. If the solid content concentration is in the range of 0.1 to 5.0% by mass, there is a problem that the film thickness becomes too thick when applied on the transparent conductive film, so that electrical contact from silver paste or the like cannot be obtained.
- the overcoat layer 5 polyurethane containing (A) carboxy group, which is a solid content in the curable resin composition, (B) epoxy compound, and (C) curing residue in the curing accelerator.
- the proportion of the aromatic ring-containing compound defined in the following formula contained in the group is preferably suppressed to 15% by mass or less.
- (C) curing residue in the curing accelerator as used herein means that all or part of the (C) curing accelerator disappears (decomposes, volatilizes, etc.) depending on the curing conditions. It means the (C) curing accelerator remaining in the coat layer 5.
- aromatic ring-containing compound means a compound having at least one aromatic ring in the molecule.
- curability is formed on the transparent resin film 3 on which the transparent conductive layer 4 is formed by a printing method such as a bar coat printing method, a gravure printing method, an inkjet method, or a slit coat method. After applying the resin composition, drying and removing the solvent, the curable resin is cured by heat treatment and light irradiation as necessary to form the overcoat layer 5.
- a printing method such as a bar coat printing method, a gravure printing method, an inkjet method, or a slit coat method.
- the carrier film 10 is a protective film 1 having an adhesive layer 2 on one main surface.
- the carrier film 10 forms a transparent conductive film laminate together with the transparent conductive film 20 or 30 that is detachably laminated via the pressure-sensitive adhesive layer 2.
- the adhesive may be peeled off together with the protective film 1, or only the protective film 1 may be peeled off.
- a film made of polyester having an aromatic ring in its molecular skeleton (hereinafter, referred to as “aromatic polyester”) is used.
- the aromatic polyester used in the present invention has crystallinity, and specific examples thereof include polyethylene terephthalate (PET) -based resins.
- PET polyethylene terephthalate
- the polyethylene terephthalate (PET) -based resin used in the present invention may be a polyethylene terephthalate (PET) resin alone, or may contain components other than polyethylene terephthalate (PET) as long as it has crystallinity.
- the ethylene isophthalate unit may be a copolymer copolymerized with the ethylene terephthalate unit, and the glass transition temperature thereof is preferably 50 to 90 ° C, more preferably 60 to 80 ° C.
- polyethylene terephthalate (PET) may be uniformly mixed with polyethylene isophthalate (PEI) or the like. Since an aromatic polyester film such as polyethylene terephthalate (PET) is usually biaxially stretched, it has better dimensional stability than an amorphous resin film.
- the thickness T 2 of the protective film 1 is 5 times or more the thickness T 1 of the transparent resin film 3, that is, T 2 / T 1 ⁇ 5, preferably T 2 / T 1 ⁇ 7, and more preferably T.
- T 2 is 150 ⁇ m or less, preferably 140 ⁇ m or less, and more preferably 130 ⁇ m or less. Further, it is preferably 100 ⁇ m or more, more preferably 110 ⁇ m or more, and further preferably 120 ⁇ m or more. Within this range, the curl generated when the transparent conductive film laminate is heated or the curl of the transparent conductive film laminate cooled after heating can be suppressed to a low level, and the transparent conductive film laminate can be suppressed to a low level. There will be no manufacturing problems during the manufacturing process and in the post-process using the transparent conductive film laminate. In addition, there is no problem in winding the long transparent conductive film laminate onto the roll after manufacturing.
- “having crystallinity” means having a property of crystallizing when the resin film is heated to a temperature equal to or higher than the melting point, melted once, and then cooled to room temperature. If the resin has crystallinity, the peak (crystallization temperature) associated with crystallization can be confirmed by performing differential scanning calorimetry (DSC) in accordance with the transition temperature measurement method of JIS K7121 plastic. The glass transition temperature can also be confirmed by performing differential scanning calorimetry (DSC) in accordance with the JIS K7121 plastic transition temperature measurement method. Polyethylene terephthalate (PET) is a resin having typical crystallinity.
- the surface may be subjected to a treatment such as plasma treatment or corona treatment in advance to improve the adhesion to the pressure-sensitive adhesive layer 2 or the like on the protective film 1. Further, before forming the pressure-sensitive adhesive layer 2, the surface of the protective film 1 may be dust-removed and cleaned by solvent cleaning, ultrasonic cleaning, or the like, if necessary.
- a treatment such as plasma treatment or corona treatment in advance to improve the adhesion to the pressure-sensitive adhesive layer 2 or the like on the protective film 1.
- the surface of the protective film 1 may be dust-removed and cleaned by solvent cleaning, ultrasonic cleaning, or the like, if necessary.
- the pressure-sensitive adhesive layer 2 can be used without particular limitation as long as it has transparency. Specifically, for example, acrylic polymers, silicone polymers, polyesters, polyurethanes, polyamides, polyvinyl ethers, vinyl acetate / vinyl chloride copolymers, modified polyolefins, epoxys, fluorines, natural rubbers, synthetic rubbers and other rubbers, etc. A polymer based on the above polymer can be appropriately selected and used. In particular, an acrylic pressure-sensitive adhesive is preferably used from the viewpoint of excellent optical transparency, appropriate wettability, cohesiveness, adhesiveness and other adhesive properties, and excellent weather resistance and heat resistance.
- the method for forming the pressure-sensitive adhesive layer 2 is not particularly limited, and the pressure-sensitive adhesive composition is applied to a release liner, dried, and then transferred to a base film (transfer method).
- the pressure-sensitive adhesive composition is directly applied to the protective film 1. Examples include a method of coating and drying (direct copying method) and a method of coextrusion. If necessary, a tackifier, a plasticizer, a filler, an antioxidant, an ultraviolet absorber, a silane coupling agent, or the like can be appropriately used as the pressure-sensitive adhesive.
- the preferable thickness of the pressure-sensitive adhesive layer 2 is 5 to 50 ⁇ m.
- the transparent conductive film laminate is a transparent conductive film laminate including a transparent conductive film 20 or 30 and a carrier film 10 laminated on the transparent conductive film 20 or 30, and the carrier film 10 is a protective film 1 and
- the adhesive layer 2 is provided on one of the main surfaces of the transparent conductive film 20, and the transparent conductive film 20 is provided on one main surface of the transparent resin film 3, and the transparent conductive film 30 is provided on both main surfaces of the transparent resin film 3.
- the transparent conductive layer 4 containing the metal nanowire and the binder resin and the overcoat layer 5 are laminated in this order, and the carrier film 10 is the transparent resin film of the transparent conductive film 20 in the transparent conductive film 20.
- the transparent conductive film 30 is releasably laminated on one of the overcoat layers 5 via the pressure-sensitive adhesive layer 2.
- the curl value immediately after cutting the transparent conductive film laminate having the above-mentioned structure into a size of 15 cm ⁇ 21 cm and heating at 100 ° C. for 30 minutes is 0 to 25 mm, and the curl value after allowing to cool for 30 minutes is 0 to 0. It is preferably 10 mm. As a result, the amount and direction of curl generated after the heating step such as drying can be controlled, so that the transparent conductive film laminate can be easily transported.
- a transparent conductive film 20 in which a transparent conductive layer 4 and an overcoat layer 5 are laminated in this order on one main surface of a transparent resin film 3 is prepared.
- a transparent conductive film 30 in which the transparent conductive layer 4 and the overcoat layer 5 are laminated in this order on both main surfaces of the transparent resin film 3 is prepared.
- the step of laminating the protective film 1 on one main surface (overcoat layer 5) of the transparent conductive film 30 via the pressure-sensitive adhesive layer 2 is included.
- the step of heat-processing the transparent conductive film laminate and the transparent conductive film 20 or 30 of the transparent conductive film laminate and the carrier film 10 are peeled off. Including the process.
- a conductive paste such as silver paste was used to print on the overcoat layer 5 of the transparent conductive film 20 or 30 so as to be electrically connected to the transparent conductive layer 4 constituting the transparent conductive film. Examples thereof include a step of drying and / or heat-curing a conductive paste pattern (pattern of wiring, electrodes, etc.) to form a conductive pattern (wiring, electrodes, etc.).
- FIGS. 4 (a) to 4 (d) show the first embodiment.
- a process diagram of a modified example of the method for manufacturing the transparent conductive film laminate according to the form is shown.
- a transparent conductive layer 4 is formed on one main surface of the transparent resin film 3 (FIG. 3A). , (B)), and then the overcoat layer 5 is formed on the transparent conductive layer 4 (FIG. 3 (c)).
- the protective film 1 is placed on the main surface of the transparent conductive film 20 on the opposite (other side) side of the transparent conductive film 20 from the side on which the transparent conductive layer 4 is laminated via the adhesive layer 2.
- a step of laminating (carrier film 10) so as to be peelable is carried out, and a transparent conductive film laminate is manufactured.
- the transparent conductive film laminate has a protective film 1 (carrier) on one main surface of the transparent resin film 3 via an adhesive layer 2 as in the modified examples shown in FIGS. 4 (a) to 4 (d).
- a transparent resin laminate in which the film 10) is detachably laminated is formed (FIGS. 4 (a) and 4 (b)), and the side of the transparent resin film 3 opposite to the main surface on which the carrier film 10 is laminated (the other side).
- the transparent conductive layer 4 is formed on the main surface of the film (FIG. 4 (c)), and then the overcoat layer 5 is formed on the transparent conductive layer 4 (FIG. 4 (d)).
- FIGS. 6 (a) to 6 (i) show the second embodiment.
- a process diagram of a modified example of the method for manufacturing the transparent conductive film laminate according to the form is shown.
- the transparent conductive layer 4 and the overcoat layer 5 are sequentially formed on one main surface of the transparent resin film 3 ( 5 (a) to 5 (c)), and subsequently, the transparent conductive layer 4 and the overcoat layer 5 are sequentially formed on the other (other) main surface of the transparent resin film 3 (FIGS. 5 (d) to 5 (c)).
- the transparent conductive film laminate is a transparent film in which the protective film 1 (carrier film 10) is detachably laminated via the pressure-sensitive adhesive layer 2 as in the modified examples shown in FIGS. 6 (a) to 6 (i).
- the transparent conductive layer 4 and the overcoat layer 5 were sequentially formed on the main surface of the resin film 3 on the side opposite to (the other side) the side on which the carrier film 10 is laminated (FIGS. 6A to 6D). ), Then the protective film 1 (carrier film 10) is detachably laminated on the overcoat layer 5 via the adhesive layer 2 (FIG. 6 (e)), and then the carrier film 10 laminated on the transparent resin film 3. (FIGS.
- the transparent conductive layer 4 and the overcoat layer are on the main surface of the transparent resin film 3 on the opposite (other) side of the main surface on which the transparent conductive layer 4 is laminated. It can also be produced in the steps of sequentially forming 5 (FIGS. 6 (h) and 6 (i)).
- the conductive paste is screened on the overcoat layer 5 on the patterned transparent conductive layer 4 (transparent conductive pattern). It can be obtained by drying and / or heat-curing a conductive paste pattern (pattern of wiring, electrodes, etc.) printed by printing or the like.
- a part of the silver nanowire is overcoated.
- the thickness of the overcoat layer is, for example, 500 nm or less, preferably 200 nm, although it cannot be said unconditionally because it is affected by the shape (diameter / length) of the silver nanowires and the number of silver nanowires coated on the base material.
- the overcoat layer can be removed by using a known etching technique to expose the silver nanowires.
- the heating temperature in the step of drying and / or thermosetting the conductive paste pattern is preferably 100 ° C. or lower, more preferably 80 ° C. or lower.
- Example 1 Manufacture of a transparent conductive film 20 having a transparent conductive layer 4 on one main surface of the transparent resin film 3> ⁇ Manufacturing of silver nanowires> Poly-N-vinylpyrrolidone K-90 (manufactured by Nippon Shokubai Co., Ltd.) (0.98 g), AgNO 3 (1.04 g) and FeCl 3 (0.8 mg) were dissolved in ethylene glycol (250 ml) at 150 ° C. Heat reaction was carried out for 1 hour.
- the obtained crude silver nanowire dispersion was dispersed in 2000 ml of methanol, and a small desktop tester (manufactured by Nippon Gaishi Co., Ltd., using ceramic membrane filter Sepilt, membrane area 0.24 m 2 , pore diameter 2.0 ⁇ m, size ⁇ 30 mm ⁇ 250 mm, filtration It was poured into a differential pressure of 0.01 MPa), cross-flow filtration was performed at a circulation flow rate of 12 L / min and a dispersion temperature of 25 ° C. to remove impurities, and then concentrated until the total amount reached 100 g, and silver nanowires (average diameter:: average diameter: A methanol dispersion (26 nm, average length: 20 ⁇ m) was obtained.
- a field emission scanning electron microscope JSM-7000F (manufactured by JEOL Ltd.) was used to measure the diameters of 100 arbitrarily selected silver nanowires, and the arithmetic average thereof was measured. The value was calculated. Further, in order to calculate the average length of the obtained silver nanowires, a shape measurement laser microscope VK-X200 (manufactured by Keyence Co., Ltd.) was used to measure the lengths of 100 arbitrarily selected silver nanowires, and the arithmetic was performed. The average value was calculated. As the methanol, ethylene glycol, AgNO 3 , and FeCl 3 , reagents manufactured by Fujifilm Wako Pure Chemical Industries, Ltd. were used.
- the silver concentration of the obtained silver nanowire ink was measured by AA280Z Zeeman atomic absorption spectrophotometer manufactured by Varian. As a result, it was 0.17% by mass.
- ⁇ Formation of transparent conductive layer (silver nanowire layer) 4> As a transparent resin film 3 subjected to plasma processing (gas used: nitrogen, transport speed: 50 mm / sec, processing time: 6 sec, set voltage: 400 V) using a plasma processing device (AP-T03 manufactured by Sekisui Chemical Industry Co., Ltd.).
- TQC automatic film applicator standard (Cortec Co., Ltd.) on A4 size cycloolefin polymer film ZF14-013 (ZEONOR (registered trademark), manufactured by Nippon Zeon Co., Ltd., glass transition temperature 136 ° C [catalog value], thickness T 1 is 13 ⁇ m)
- Silver nanowire ink using wireless bar OSP-CN-22L manufactured by Cortec Co., Ltd., bar shape / P (groove pitch): 500 ⁇ m, H (groove depth): 42 m, material: SUS304) was applied to the entire surface (coating speed 500 mm / sec). Then, it was dried with hot air in an air atmosphere at 80 ° C.
- a transparent conductive layer (silver nanowire layer) 4 having a thickness of 90 nm as the transparent conductive layer 4.
- ⁇ Making overcoat resin> C-1015N manufactured by Kuraray Co., Ltd., polycarbonate diol, raw material diol molar ratio: 1,9-nonanediol: 2-methyl-1,8-octane
- C-1015N manufactured by Kuraray Co., Ltd., polycarbonate diol, raw material diol molar ratio: 1,9-nonanediol: 2-methyl-1,8-octane
- Diol 15:85, molecular weight 964) 42.32 g, carboxy group-containing dihydroxyl compound 2,2-dimethylolbutanoic acid (Huzhou Changsheng Chemical Co., Ltd.) 27.32 g, and diethylene glycol monoethyl ether acetate (manufactured by Huzhou Changsheng Chemicals) as a solvent. 158 g (manufactured by Daicel Co., Ltd.) was charged, and the 2,2-dimethylolbutanoic acid was dissolved at 90 ° C.
- the temperature of the reaction solution was lowered to 70 ° C., and by dropping funnel, Death Module (registered trademark) -W (bis- (4-isocyanatocyclohexyl) methane), manufactured by Sumika Covestro Urethane Co., Ltd., 59.69 g as polyisocyanate. Was added dropwise over 30 minutes.
- the temperature was raised to 120 ° C., the reaction was carried out at 120 ° C. for 6 hours, and after confirming by IR that the isocyanato group had almost disappeared, 0.5 g of isobutanol was added, and the reaction was further carried out at 120 ° C. for 6 hours. Was done.
- the weight average molecular weight of the obtained carboxy group-containing polyurethane (overcoat resin) determined by GPC was 32,300, and the acid value of the resin solution was 35.8 mgKOH / g.
- the proportion of the curing accelerator, which is an aromatic ring-containing compound, in the solid content of the overcoat ink is 5.7% by mass.
- Overcoat ink was applied on the transparent conductive layer (silver nanowire layer) 4 formed on the transparent resin film 3 by TQC automatic film applicator standard (manufactured by Cortec Co., Ltd.) as follows (coating speed 500 mm /). sec). It was applied using a wireless bar OSP-CN-05M so that the wet film thickness was 5 ⁇ m. Then, the overcoat layer 5 (90 nm) was formed by hot air drying and thermosetting in an air atmosphere at 80 ° C. for 1 minute in an incubator HISPEC HS350 (manufactured by Kusumoto Kasei Co., Ltd.).
- An acrylic pressure-sensitive adhesive was prepared by adding 6 parts by mass of an epoxy-based cross-linking agent to 100 parts by mass of this acrylic polymer. The acrylic pressure-sensitive adhesive was applied onto the release-treated surface of the release-treated PET film and heated at 120 ° C. for 60 seconds to form a pressure-sensitive adhesive layer 2 having a thickness of 20 ⁇ m. Next, a PET film having been released from the mold was attached to one side of a PET film (protective film 1) having a thickness T 2 of 125 ⁇ m via an adhesive layer 2.
- the release-treated PET film was peeled off to prepare a carrier film 10 having an adhesive layer 2 on one surface of the protective film 1.
- the PET film used as the protective film 1 was measured for differential scanning calorimetry (DSC) using a differential scanning calorimetry device DSC1 (manufactured by METTLER TOLEDO) in accordance with the transition temperature measurement method for JIS K7121 plastic. It was confirmed that the resin had a peak associated with crystallization and that the glass transition temperature (external glass transition start temperature) was 72 ° C.
- a carrier film 10 is placed on the surface of the transparent resin film 3 constituting the transparent conductive film 20 in which the overcoat layer 5 has already been formed, on the side where the transparent conductive layer (silver nanowire layer) 4 is not formed, via the pressure-sensitive adhesive layer 2. It was crimped and laminated to form a transparent conductive film laminate.
- Example 2 The preparation of the transparent conductive film 20, a cycloolefin polymer film RX4500 as the transparent resin film 3 (ARTON (registered trademark), manufactured by JSR Corporation, a glass transition temperature of 132 ° C. [catalog value], the thickness T 1 is 15 [mu] m) but using Was examined under the same conditions as in Example 1. The results are shown in Table 1.
- Example 3 To prepare the transparent conductive film 20, a cycloolefin polymer film ZF14-023 (ZEONOR (registered trademark), manufactured by Nippon Zeon Co., Ltd., glass transition temperature 136 ° C. [catalog value], thickness T 1 is 23 ⁇ m) was used as the transparent resin film 3. Examination was conducted under the same conditions as in Example 1 except that it was used. The results are shown in Table 1.
- Example 4 Preparation of a transparent conductive film laminate having a transparent conductive layer 4 on both main surfaces of the transparent resin film 3>
- a transparent conductive layer (silver nanowire layer) 4 and an overcoat layer 5 were prepared on one main surface (first surface) of the cycloolefin polymer film ZF14-013 under the same conditions as in Example 1.
- a transparent conductive layer (silver nanowire layer) 4 and an overcoat layer 5 are formed on the other main surface (second surface) on which the transparent conductive layer 4 is not formed under the same conditions as in Example 1, and are transparent.
- a transparent conductive film 30 having a transparent conductive layer (silver nanowire layer) 4 on both main surfaces of the resin film 3 was obtained.
- the carrier film 10 was pressure-bonded and laminated on the overcoat layer 5 on the first surface of the transparent conductive film 30 via the adhesive layer 2 to form a transparent conductive film laminate.
- Table 1 The results are shown in Table 1.
- Comparative Example 1 The same conditions as in Example 1 were examined except that a PET film having a thickness T 2 of 50 ⁇ m was used as the protective film 1 for producing the carrier film 10. The results are shown in Table 1.
- Comparative Example 2 The same conditions as in Example 2 were examined except that a PET film having a thickness T 2 of 50 ⁇ m was used as the protective film 1 for producing the carrier film 10. The results are shown in Table 1.
- Comparative Example 3 To prepare the transparent conductive film 20, a cycloolefin polymer film ZF14-050 (ZEONOR (registered trademark), manufactured by Nippon Zeon Co., Ltd., glass transition temperature 136 ° C. [catalog value], thickness T 1 is 50 ⁇ m) was used as the transparent resin film 3. Examination was conducted under the same conditions as in Example 1 except that it was used. The results are shown in Table 1.
- Comparative Example 4 To prepare the transparent conductive film 20, a cycloolefin polymer film ZF14-100 (ZEONOR (registered trademark), manufactured by Nippon Zeon Co., Ltd., glass transition temperature 136 ° C. [catalog value], thickness T 1 is 100 ⁇ m) was used as the transparent resin film 3. Examination was conducted under the same conditions as in Example 1 except that it was used. The results are shown in Table 1.
- Comparative Example 5 The study was conducted under the same conditions as in Example 4 except that a PET film having a thickness T 2 of 50 ⁇ m was used as the protective film 1 for producing the transparent conductive film 30. The results are shown in Table 1.
- ⁇ Evaluation> Measurement of thickness For those having a thickness of 1 ⁇ m or more, the thickness was measured with a microgauge type thickness gauge. The thickness of the transparent conductive layer 4 and the overcoat layer 5 containing metal nanowires of less than 1 ⁇ m was measured using a film thickness measuring system F20-UV (manufactured by Filmometrics Co., Ltd.) based on the optical interferometry.
- the uneven shape of the transparent conductive film laminate was observed from the glass window.
- the test piece is placed on a horizontal surface with the overcoat layer 5 (meaning the overcoat layer 5 on which the protective film 1 is not laminated in Example 4 and Comparative Example 5) is on top. Placed and quickly measured the curl value with a ruler. This was taken as the curl value during heating. Further, the mixture was allowed to cool at room temperature for 30 minutes, and the curl value was measured with a ruler. This was used as the curl value after cooling.
- FIG. 7 (a) and 7 (b) show explanatory diagrams of a method for measuring the curl value.
- FIG. 7A shows a case where the transparent conductive film laminate curls convexly
- FIG. 7B shows a case where the transparent conductive film laminate curls concavely.
- the height hmax of the highest point of the transparent conductive film laminate was measured from the horizontal plane Hs on which the transparent conductive film laminate was placed, and used as the curl value.
- both the curl value generated during heating and the curl value after allowing to cool are smaller than 5 mm (evaluation ⁇ ), and in Example 3, the curl value during heating is 20 mm. However, after allowing to cool, it became smaller than 5 mm (less than 1 mm) (evaluation ⁇ ). Therefore, in all of Examples 1 to 3, the deformation could be suppressed to the extent that it would not interfere with the roll transfer and the subsequent process. Even in Example 4 using the transparent conductive film 30 having the transparent conductive layer 4 on both sides of the transparent resin film 3, the curl value generated during heating and the curl value after allowing to cool are both as small as 10 mm (evaluation ⁇ ), and the roll is conveyed. Deformation could be suppressed to the extent that it would not interfere with the subsequent process.
- the criteria for the evaluations " ⁇ ", “ ⁇ ”, and “ ⁇ ” in Table 1 are that the curl value during heating is 5 mm or less and the curl value after cooling is 1 mm or less is " ⁇ ”, and the curl value during heating is 25 mm.
- the case where the curl value was 10 mm or less after allowing to cool and the curl value was more than 25 mm and / or the curl value after allowing to cool was more than 10 mm was evaluated as “x”.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Manufacturing & Machinery (AREA)
- Inorganic Chemistry (AREA)
- Physics & Mathematics (AREA)
- General Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Theoretical Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Dispersion Chemistry (AREA)
- Spectroscopy & Molecular Physics (AREA)
- General Physics & Mathematics (AREA)
- Human Computer Interaction (AREA)
- Laminated Bodies (AREA)
- Non-Insulated Conductors (AREA)
- Manufacturing Of Electric Cables (AREA)
- Paints Or Removers (AREA)
- Adhesive Tapes (AREA)
Abstract
Description
透明樹脂フィルム3は、非晶性シクロオレフィン系樹脂により形成されており、高透明性及び低吸水性の特性を有する。透明樹脂フィルム3として非晶性シクロオレフィン系樹脂フィルムを採用することにより、良好な光学特性を有する透明導電フィルムを備えた透明導電フィルム積層体が得られる。
透明樹脂フィルム3上に形成される透明導電層4を構成する導電材料としては、金属ナノワイヤを好適に使用することができる。金属ナノワイヤは、径がナノメーターオーダーのサイズである金属であり、ワイヤ状の形状を有する導電性材料である。なお、本実施形態では、金属ナノワイヤとともに(混合して)、または金属ナノワイヤに代えて、ポーラスあるいはノンポーラスのチューブ状の形状を有する導電性材料である金属ナノチューブを使用してもよい。本明細書において、「ワイヤ状」と「チューブ状」はいずれも線状であるが、前者は中央が中空ではないもの、後者は中央が中空であるものを意図する。性状は、柔軟であってもよく、剛直であってもよい。前者を「狭義の金属ナノワイヤ」、後者を「狭義の金属ナノチューブ」と呼び、以下、本願明細書において、「金属ナノワイヤ」は狭義の金属ナノワイヤと狭義の金属ナノチューブとを包括する意味で用いる。狭義の金属ナノワイヤ、狭義の金属ナノチューブは、単独で用いてもよく、混合して用いてもよい。
下記溶離液にバインダー樹脂を溶解させ、20時間静置した。この溶液におけるバインダー樹脂の濃度は0.05質量%である。
これを0.45μmメンブレンフィルターにて濾過し、濾液をGPC-MALSにて測定を実施し、絶対分子量基準の重量平均分子量を算出した。
GPC:昭和電工株式会社製Shodex(登録商標)SYSTEM21
カラム:東ソー株式会社製TSKgel(登録商標)G6000PW
カラム温度:40℃
溶離液:0.1mol/L NaH2PO4水溶液+0.1mol/L Na2HPO4水溶液
流速:0.64mL/min
試料注入量:100μL
MALS検出器:ワイアットテクノロジーコーポレーション、DAWN(登録商標) DSP
レーザー波長:633nm
多角度フィット法:Berry法
透明導電層4を保護するオーバーコート層5は、硬化性樹脂組成物の硬化膜であることが好ましい。硬化性樹脂組成物としては、(A)カルボキシ基を含有するポリウレタンと、(B)エポキシ化合物と、(C)硬化促進剤と、(D)溶媒と、を含むものが好ましい。硬化性樹脂組成物を上記透明導電層4上に印刷、塗布等により形成し、硬化させてオーバーコート層5を形成する。硬化性樹脂組成物の硬化は、熱硬化性樹脂組成物を加熱・乾燥させることにより行うことができる。
装置名:日本分光株式会社製HPLCユニット HSS-2000
カラム:ShodexカラムLF-804
移動相:テトラヒドロフラン
流速 :1.0mL/min
検出器:日本分光株式会社製 RI-2031Plus
温度 :40.0℃
試料量:サンプルル-プ 100μL
試料濃度:約0.1質量%に調製
100ml三角フラスコに試料約0.2gを精密天秤にて精秤し、これにエタノール/トルエン=1/2(質量比)の混合溶媒10mlを加えて溶解する。更に、この容器に指示薬としてフェノールフタレインエタノール溶液を1~3滴添加し、試料が均一になるまで十分に攪拌する。これを、0.1N水酸化カリウム-エタノール溶液で滴定し、指示薬の微紅色が30秒間続いたときを、中和の終点とする。その結果から下記の計算式を用いて得た値を、樹脂の酸価とする。
酸価(mg-KOH/g)=〔B×f×5.611〕/S
B:0.1N水酸化カリウム-エタノール溶液の使用量(mL)
f:0.1N水酸化カリウム-エタノール溶液のファクター
S:試料の採取量(g)
(a1)ポリイソシアネート化合物としては、通常、1分子当たりのイソシアナト基が2個であるジイソシアネートが用いられる。ポリイソシアネート化合物としては、たとえば、脂肪族ポリイソシアネート、脂環族ポリイソシアネート等が挙げられ、これらの1種を単独でまたは2種以上を組み合わせて用いることができる。(A)カルボキシ基を含有するポリウレタンがゲル化をしない範囲で、イソシアナト基を3個以上有するポリイソシアネートも少量使用することができる。
(a2)ポリオール化合物(ただし、(a2)ポリオール化合物には、後述する(a3)カルボキシ基を有するジヒドロキシ化合物は含まれない。)の数平均分子量は通常250~50,000であり、好ましくは400~10,000、より好ましくは500~5,000である。この分子量は前述した条件でGPCにより測定したポリスチレン換算の値である。
(a3)カルボキシ基を含有するジヒドロキシ化合物としては、ヒドロキシ基、炭素数が1または2のヒドロキシアルキル基から選択されるいずれかを2つ有する分子量が200以下のカルボン酸またはアミノカルボン酸であることが架橋点を制御できる点で好ましい。具体的には2,2-ジメチロ-ルプロピオン酸、2,2-ジメチロ-ルブタン酸、N,N-ビスヒドロキシエチルグリシン、N,N-ビスヒドロキシエチルアラニン等が挙げられ、この中でも、溶媒への溶解度から、2,2-ジメチロ-ルプロピオン酸、2,2-ジメチロ-ルブタン酸が特に好ましい。これらの(a3)カルボキシ基を含有するジヒドロキシ化合物は、1種単独でまたは2種以上を組み合わせて用いることができる。
(a4)モノヒドロキシ化合物として、グリコール酸、ヒドロキシピバリン酸等カルボン酸を有する化合物が挙げられる。
(a5)モノイソシアネート化合物としては、ヘキシルイソシアネート、ドデシルイソシアネート等が挙げられる。
[(芳香環含有化合物使用量)/(オーバーコート層5の質量((A)カルボキシ基を含有するポリウレタン質量+(B)エポキシ化合物質量+(C)硬化促進剤における硬化残基)]×100(%)
キャリアフィルム10は、一方の主面に粘着剤層2を有する保護フィルム1である。このキャリアフィルム10は、上記粘着剤層2を介して剥離可能に積層された透明導電フィルム20または30とともに透明導電フィルム積層体を形成する。キャリアフィルム10を透明導電フィルム積層体から剥離する際は、粘着剤は保護フィルム1とともに剥離されてもよいし、保護フィルム1のみが剥離されてもよい。
保護フィルム1には、分子骨格に芳香環を有するポリエステル(以下、「芳香族ポリエステル」と記載)からなるフィルムが用いられる。本発明で用いられる芳香族ポリエステルは、結晶性を有するものであり、具体的にはポリエチレンテレフタレート(PET)系樹脂が挙げられる。本発明で用いられるポリエチレンテレフタレート(PET)系樹脂は、ポリエチレンテレフタレート(PET)樹脂単独であってもよいし、結晶性を有する範囲で、ポリエチレンテレフタレート(PET)以外の他の成分を含んでもよい。例えば、エチレンイソフタレートユニットがエチレンテレフタレートユニットと共重合された共重合体であってもよく、そのガラス転移温度は50~90℃であることが好ましく、60~80℃であることがより好ましい。また、ポリエチレンテレフタレート(PET)にポリエチレンイソフタレート(PEI)等を均一混合したものであってもよい。ポリエチレンテレフタレート(PET)等の芳香族ポリエステルフィルムは通常ニ軸延伸されているため、非晶性の樹脂フィルムに比べて寸法安定性が良好である。保護フィルム1の厚みT2は、透明樹脂フィルム3の厚みT1の5倍以上、すなわち、T2/T1≧5であり、好ましくはT2/T1≧7であり、さらに好ましくはT2/T1≧8である。かつ、T2は150μm以下であり、140μm以下であることが好ましく、130μm以下であることがより好ましい。また、100μm以上であることが好ましく、110μm以上であることがより好ましく、120μm以上であることがさらに好ましい。この範囲であると、透明導電フィルム積層体が加熱された際に発生するカール、あるいは加熱後冷却された透明導電フィルム積層体のカールを、低レベルに抑制することができ、透明導電フィルム積層体の製造時および透明導電フィルム積層体を用いた後工程での製造上の不具合が発生することがない。また、長尺の透明導電フィルム積層体を製造後ロールへの巻取りをする際の不具合が発生することもない。
粘着剤層2としては、透明性を有するものであれば特に制限なく使用できる。具体的には、例えば、アクリル系ポリマー、シリコーン系ポリマー、ポリエステル、ポリウレタン、ポリアミド、ポリビニルエーテル、酢酸ビニル/塩化ビニルコポリマー、変性ポリオレフィン、エポキシ系、フッ素系、天然ゴム、合成ゴム等のゴム系などのポリマーをベースポリマーとするものを適宜に選択して用いることができる。特に、光学的透明性に優れ、適度な濡れ性、凝集性および接着性等の粘着特性を示し、耐候性や耐熱性等にも優れるという観点からは、アクリル系粘着剤が好ましく用いられる。
透明導電フィルム積層体は、透明導電フィルム20または30と、透明導電フィルム20または30に積層されたキャリアフィルム10と、を含む透明導電フィルム積層体であって、キャリアフィルム10は、保護フィルム1と、その一方の主面に粘着剤層2を備えており、透明導電フィルム20では、透明樹脂フィルム3の一方の主面に、透明導電フィルム30では、透明樹脂フィルム3の両方の主面に、金属ナノワイヤ及びバインダー樹脂を含む透明導電層4と、オーバーコート層5と、がこの順序に積層されて構成されており、キャリアフィルム10は、透明導電フィルム20では、透明導電フィルム20の透明樹脂フィルム3に、また、透明導電フィルム30では、透明導電フィルム30の一方のオーバーコート層5に、粘着剤層2を介して剥離可能に積層されている。
第一の実施形態にかかる透明導電フィルム積層体の製造方法は、透明樹脂フィルム3の一方の主面に透明導電層4とオーバーコート層5とがこの順序で積層された透明導電フィルム20を準備する工程と、透明導電フィルム20の透明導電層4が積層されている側とは反対(他方)側の主面に粘着剤層2を介して保護フィルム1(キャリアフィルム10)を積層する工程と、を含む。
<透明樹脂フィルム3の一方の主面に透明導電層4を有する透明導電フィルム20の作製>
<銀ナノワイヤの作製>
ポリ-N-ビニルピロリドンK-90(株式会社日本触媒製)(0.98g)、AgNO3(1.04g)及びFeCl3(0.8mg)を、エチレングリコール(250ml)に溶解し、150℃で1時間加熱反応した。得られた銀ナノワイヤ粗分散液をメタノール2000mlに分散させ、卓上小型試験機(日本ガイシ株式会社製、セラミック膜フィルター セフィルト使用、膜面積0.24m2、孔径2.0μm、寸法Φ30mm×250mm、ろ過差圧0.01MPa)に流し入れ、循環流速12L/min、分散液温度25℃にてクロスフロー濾過を実施し不純物を除去した後、全体量が100gになるまで濃縮し、銀ナノワイヤ(平均直径:26nm、平均長さ:20μm)のメタノール分散液を得た。得られた銀ナノワイヤの平均径の算出には、電界放出形走査電子顕微鏡JSM-7000F(日本電子株式会社製)を用い、任意に選択した100本の銀ナノワイヤの直径を測定し、その算術平均値を求めた。また、得られた銀ナノワイヤの平均長の算出には、形状測定レーザマイクロスコープVK-X200(キーエンス株式会社製)を用い、任意に選択した100本の銀ナノワイヤの長さを測定し、その算術平均値を求めた。また、上記メタノール、エチレングリコール、AgNO3、FeCl3は富士フイルム和光純薬株式会社製試薬を用いた。
上記ポリオール法で合成した銀ナノワイヤのメタノール分散液11g(銀ナノワイヤ濃度0.62質量%)、水3.5g、エタノール10.8g(富士フイルム和光純薬株式会社製)、プロピレングリコールモノメチルエーテル(PGME、富士フイルム和光純薬株式会社製)12.8g、プロピレングリコール1.2g(PG、旭硝子株式会社製)、PNVA(登録商標)水溶液(昭和電工株式会社製、固形分濃度10質量%、重量平均分子量90万)0.7gを混合し、ミックスローターVMR-5R(アズワン株式会社製)で1時間、室温、大気雰囲気下で撹拌(回転速度100rpm)して導電性インクとしての銀ナノワイヤインク40gを作製した。
プラズマ処理装置(積水化学工業株式会社製AP-T03)を用いてプラズマ処理(使用ガス:窒素、搬送速度:50mm/sec、処理時間:6sec、設定電圧:400V)した、透明樹脂フィルム3としてのA4サイズのシクロオレフィンポリマーフィルムZF14-013(ZEONOR(登録商標)、日本ゼオン株式会社製、ガラス転移温度136℃[カタログ値]、厚みT1が13μm)上に、TQC自動フィルムアプリケータースタンダード(コーテック株式会社製)とワイヤレスバーOSP-CN-22L(コーテック株式会社製、バー形状/P(溝のピッチ):500μm、H(溝の深さ):42m、材質:SUS304)とを用いて銀ナノワイヤインクを全面に塗布した(塗工速度500mm/sec)。その後、恒温器HISPEC HS350(楠本化成株式会社製)で80℃、1分間、大気雰囲気下で熱風乾燥し、透明導電層4としての厚み90nmの透明導電層(銀ナノワイヤ層)4を形成した。
攪拌装置、温度計、コンデンサーを備えた2L三口フラスコに、ポリオール化合物としてC-1015N(株式会社クラレ製、ポリカーボネートジオール、原料ジオールモル比:1,9-ノナンジオール:2-メチル-1,8-オクタンジオール=15:85、分子量964)42.32g、カルボキシ基を含有するジヒドロキシル化合物として2,2-ジメチロールブタン酸(湖州長盛化工製)27.32g、および溶媒としてジエチレングリコールモノエチルエーテルアセテート(株式会社ダイセル製)158gを仕込み、90℃で前記2,2-ジメチロールブタン酸を溶解させた。
上記得られた(A)カルボキシ基含有ポリウレタンの溶液(カルボキシ基含有ポリウレタン含有率:45質量%)10.0gをポリ容器に量り取り、(D)溶媒として1-ヘキサノール85.3gと酢酸エチル85.2gを加え、ミックスローターVMR-5R(アズワン株式会社製)で12時間、室温、大気雰囲気下で撹拌(回転速度100rpm)した。均一であることを目視で確認したのち、(B)エポキシ化合物としてペンタエリスリトールテトラグリシジルエーテル(昭和電工株式会社製)0.63g、(C)硬化促進剤として、U-CAT5003(サンアプロ株式会社製)0.31gを加え、再度ミックスローターを用いて1時間撹拌し、オーバーコートインクを得た。オーバーコートインクの固形分中の芳香環含有化合物である硬化促進剤の割合は5.7質量%である。
透明樹脂フィルム3上に形成した透明導電層(銀ナノワイヤ層)4の上に、TQC自動フィルムアプリケータースタンダード(コーテック株式会社製)により、以下のようにオーバーコートインクを塗布した(塗工速度500mm/sec)。ワイヤレスバーOSP-CN-05Mを用いてウェット膜厚が5μmになるように塗布した。その後、恒温器HISPEC HS350(楠本化成株式会社製)で80℃、1分間、大気雰囲気下で熱風乾燥及び熱硬化し、オーバーコート層5(90nm)を形成した。
通常の溶液重合により、ブチルアクリレート/アクリル酸=100/6(質量比)にて重量平均分子量60万のアクリル系ポリマーを得た。このアクリル系ポリマー100質量部に対し、エポキシ系架橋剤6質量部を加えてアクリル系粘着剤を準備した。離型処理されたPETフィルムの離型処理面上に上記アクリル系粘着剤を塗布し、120℃、60秒で加熱して、厚み20μmの粘着剤層2を形成した。次いで、厚みT2が125μmのPETフィルム(保護フィルム1)の片面に離型処理されたPETフィルムを粘着剤層2を介して貼りあわせた。その後、離型処理されたPETフィルムを剥がし、保護フィルム1の一方の面に粘着剤層2を有するキャリアフィルム10を作製した。なお、保護フィルム1として使用したPETフィルムをJIS K7121 プラスチックの転移温度測定方法に準拠して示差走査熱量測定装置DSC1(メトラー・トレド社製)を用いて示差走査熱量測定(DSC)した結果194℃に結晶化に伴うピークを有すること、ガラス転移温度(挿外ガラス転移開始温度)が72℃であることを確認した。
前記オーバーコート層5形成済みの透明導電フィルム20を構成する透明樹脂フィルム3の透明導電層(銀ナノワイヤ層)4が形成されていない側の面に、キャリアフィルム10を粘着剤層2を介して圧着、積層し、透明導電フィルム積層体を形成した。
透明導電フィルム20の作製に、透明樹脂フィルム3としてシクロオレフィンポリマーフィルムRX4500(ARTON(登録商標)、JSR株式会社製、ガラス転移温度132℃[カタログ値]、厚みT1が15μm)を用いた以外は実施例1と同条件で検討した。その結果を表1に示す。
透明導電フィルム20の作製に、透明樹脂フィルム3としてシクロオレフィンポリマーフィルムZF14-023(ZEONOR(登録商標)、日本ゼオン株式会社製、ガラス転移温度136℃[カタログ値]、厚みT1が23μm)を用いた以外は実施例1と同条件で検討した。その結果を表1に示す。
<透明樹脂フィルム3の両方の主面に透明導電層4を有する透明導電フィルム積層体の作製>
シクロオレフィンポリマーフィルムZF14-013の一方の主面(第一面)に、実施例1と同条件で透明導電層(銀ナノワイヤ層)4とオーバーコート層5を作製した。続いて、透明導電層4が形成されていないもう一方の主面(第二面)に、実施例1と同条件で透明導電層(銀ナノワイヤ層)4とオーバーコート層5を形成し、透明樹脂フィルム3の両方の主面に透明導電層(銀ナノワイヤ層)4を有する透明導電フィルム30を得た。この透明導電フィルム30の第一面のオーバーコート層5に、キャリアフィルム10を、粘着層2を介して圧着、積層し、透明導電フィルム積層体を形成した。その結果を表1に示す。
キャリアフィルム10の作製に、保護フィルム1として厚みT2が50μmのPETフィルムを用いた以外は実施例1と同条件で検討した。その結果を表1に示す。
キャリアフィルム10の作製に、保護フィルム1として厚みT2が50μmのPETフィルムを用いた以外は実施例2と同条件で検討した。その結果を表1に示す。
透明導電フィルム20の作製に、透明樹脂フィルム3としてシクロオレフィンポリマーフィルムZF14-050(ZEONOR(登録商標)、日本ゼオン株式会社製、ガラス転移温度136℃[カタログ値]、厚みT1が50μm)を用いた以外は実施例1と同条件で検討した。その結果を表1に示す。
透明導電フィルム20の作製に、透明樹脂フィルム3としてシクロオレフィンポリマーフィルムZF14-100(ZEONOR(登録商標)、日本ゼオン株式会社製、ガラス転移温度136℃[カタログ値]、厚みT1が100μm)を用いた以外は実施例1と同条件で検討した。その結果を表1に示す。
透明導電フィルム30の作製に、保護フィルム1として厚みT2が50μmのPETフィルムを用いた以外は実施例4と同条件で検討した。その結果を表1に示す。
(1)厚みの測定
厚みは1μm以上の厚みを有するものに関してはマイクロゲージ式厚み計にて測定を行った。また1μm未満の金属ナノワイヤを含む透明導電層4およびオーバーコート層5の厚みは光干渉法に基づく膜厚測定システムF20-UV(フィルメトリクス株式会社製)を用いて測定した。
実施例及び比較例で得られたA4サイズの透明導電フィルム積層体を長手方向の略中央で裁断して15cm(元のA4サイズの長手方向)×21cm(A4サイズの短手方向)のA5サイズの試験片を作製した。この試験片を、前面扉にガラス窓のついた乾燥機VO-420(Advantec製)に入れ、オーバーコート層5(実施例4と比較例5においては保護フィルム1を積層していないほうのオーバーコート層5を意味する)が上になる状態で100℃、30分加熱した。
Claims (7)
- キャリアフィルムと、前記キャリアフィルムに積層された透明導電フィルムと、を含む透明導電フィルム積層体であって、前記キャリアフィルムは、保護フィルムと、その一方の主面に粘着剤層を備えており、前記透明導電フィルムは、透明樹脂フィルムの一方又は両方の主面に金属ナノワイヤ及びバインダー樹脂を含む透明導電層と、オーバーコート層と、がこの順序に積層されて構成され、前記透明樹脂フィルムは、非晶性シクロオレフィン系樹脂からなり、前記透明樹脂フィルムの厚みT1は5~25μmであり、前記透明導電フィルム積層体は、前記オーバーコート層が最外層となるように、前記透明導電フィルムが前記キャリアフィルムの粘着剤層に剥離可能に積層されており、前記保護フィルムは、分子骨格に芳香環を有するポリエステルからなり、前記保護フィルムの厚みT2が前記透明樹脂フィルムの厚みT1の5倍以上かつ150μm以下であることを特徴とする透明導電フィルム積層体。
- 前記金属ナノワイヤ及びバインダー樹脂を含む透明導電層と、オーバーコート層と、がこの順序で、前記透明樹脂フィルムの一方の主面に積層されている、請求項1に記載の透明導電フィルム積層体。
- 前記金属ナノワイヤ及びバインダー樹脂を含む透明導電層と、オーバーコート層と、がこの順序で、前記透明樹脂フィルムの両方の主面にそれぞれ積層されている、請求項1に記載の透明導電フィルム積層体。
- 前記保護フィルムが、ポリエチレンテレフタレート系樹脂からなる請求項1~3のいずれかに記載の透明導電フィルム積層体。
- 前記透明導電層に含まれる金属ナノワイヤが、銀ナノワイヤである請求項1~4のいずれかに記載の透明導電フィルム積層体。
- 請求項1~5のいずれか一項に記載の透明導電フィルム積層体を加熱加工する工程と、前記透明導電フィルム積層体の透明導電フィルムとキャリアフィルムとを剥離する工程と、を含む透明導電フィルム積層体の加工方法。
- 前記加熱加工する工程は、オーバーコート層の上に導電ペーストにより形成した導電ペーストパターンを乾燥及び/または熱硬化し導電パターンを形成する工程である、請求項6に記載の透明導電フィルム積層体の加工方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP19934145.4A EP3988299A4 (en) | 2019-06-20 | 2019-12-24 | ELECTROCONDUCTIVE TRANSPARENT FILM LAMINATE AND METHOD FOR MAKING SUCH LAMINATE |
| US17/615,429 US11710581B2 (en) | 2019-06-20 | 2019-12-24 | Transparent conducting film laminate and processing method thereof |
| KR1020207034079A KR102265033B1 (ko) | 2019-06-20 | 2019-12-24 | 투명 도전 필름 적층체 및 그 가공 방법 |
| JP2020526457A JP6732161B1 (ja) | 2019-06-20 | 2019-12-24 | 透明導電フィルム積層体及びその加工方法 |
| CN201980030487.9A CN112292265B (zh) | 2019-06-20 | 2019-12-24 | 透明导电膜叠层体及其加工方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019114964 | 2019-06-20 | ||
| JP2019-114964 | 2019-06-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020255458A1 true WO2020255458A1 (ja) | 2020-12-24 |
Family
ID=73993747
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/050513 WO2020255458A1 (ja) | 2019-06-20 | 2019-12-24 | 透明導電フィルム積層体及びその加工方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US11710581B2 (ja) |
| EP (1) | EP3988299A4 (ja) |
| JP (2) | JP6732161B1 (ja) |
| KR (1) | KR102265033B1 (ja) |
| CN (2) | CN113954479A (ja) |
| TW (1) | TWI808289B (ja) |
| WO (1) | WO2020255458A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023195446A1 (ja) * | 2022-04-07 | 2023-10-12 | 日東電工株式会社 | 保護フィルム付透明導電性フィルム |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11535047B2 (en) | 2019-05-31 | 2022-12-27 | Showa Denko K.K. | Method for producing transparent conducting film |
| KR102316141B1 (ko) | 2019-12-27 | 2021-10-22 | 쇼와 덴코 가부시키가이샤 | 투명 도전 필름의 제조 방법 |
| CN112970075B (zh) | 2019-12-27 | 2021-11-19 | 昭和电工株式会社 | 透明导电膜的制造方法 |
| CN113556882B (zh) * | 2020-04-23 | 2022-08-16 | 鹏鼎控股(深圳)股份有限公司 | 透明电路板的制作方法以及透明电路板 |
| CN111752415B (zh) * | 2020-06-24 | 2024-01-19 | 京东方科技集团股份有限公司 | 一种触控模组、其制备方法及显示装置 |
| CN114531918A (zh) * | 2020-08-26 | 2022-05-24 | 昭和电工株式会社 | 透明导电基体 |
| CN113782256B (zh) * | 2021-11-09 | 2022-02-18 | 南京邮电大学 | 一种低表面粗糙度透明电极的制作方法 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008046058A2 (en) | 2006-10-12 | 2008-04-17 | Cambrios Technologies Corporation | Nanowire-based transparent conductors and applications thereof |
| WO2008073143A2 (en) | 2006-06-21 | 2008-06-19 | Cambrios Technologies Corporation | Methods of controlling nanostructure formations and shapes |
| JP2008251529A (ja) | 2007-03-02 | 2008-10-16 | Nitto Denko Corp | 粘着剤層付き透明導電性フィルムおよびその製造方法 |
| JP2016107503A (ja) | 2014-12-05 | 2016-06-20 | 日東電工株式会社 | 透明導電性フィルム積層体及びそれを用いて得られるタッチパネル、並びに透明導電性フィルムの製造方法 |
| WO2016104204A1 (ja) * | 2014-12-26 | 2016-06-30 | 日東電工株式会社 | 透明導電性フィルム、透明導電性フィルム積層体及びタッチパネル |
| JP2016173714A (ja) * | 2015-03-17 | 2016-09-29 | 東レフィルム加工株式会社 | 導電積層体およびそれを用いたタッチパネル |
| JP2019149012A (ja) * | 2018-02-27 | 2019-09-05 | 日東電工株式会社 | 透明導電性フィルム積層体および透明導電性フィルムの製造方法 |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4151821B2 (ja) | 2002-01-11 | 2008-09-17 | 日東電工株式会社 | 透明導電性フィルム用表面保護フィルム及び透明導電性フィルム |
| JP5844996B2 (ja) * | 2011-05-11 | 2016-01-20 | 日東電工株式会社 | 透明導電性積層体及びタッチパネル |
| US20150103269A1 (en) | 2012-04-26 | 2015-04-16 | Showa Denko K.K. | Transparent conductive substrate production method, transparent conductive substrate, and electrostatic capacitance touch panel |
| JP5944880B2 (ja) * | 2012-12-07 | 2016-07-05 | 日東電工株式会社 | 積層体 |
| JP2014191894A (ja) | 2013-03-26 | 2014-10-06 | Dic Corp | 透明導電フィルム及びタッチパネル |
| JP6433707B2 (ja) * | 2014-07-28 | 2018-12-05 | 日東電工株式会社 | 透明導電性積層体およびその製造方法、透明導電性フィルムの製造方法、ならびに透明導電性フィルム巻回体の製造方法 |
| JP6720481B2 (ja) | 2014-07-29 | 2020-07-08 | 大日本印刷株式会社 | 積層体、導電性積層体及びタッチパネル |
| JP6538432B2 (ja) | 2014-09-01 | 2019-07-03 | 日東電工株式会社 | 透明導電性フィルム用キャリアフィルム及び積層体 |
| JP5957133B2 (ja) | 2014-11-20 | 2016-07-27 | 日東電工株式会社 | 保護フィルム付き透明導電性フィルム |
| JP6512804B2 (ja) | 2014-12-05 | 2019-05-15 | 日東電工株式会社 | 透明導電性フィルム積層体及びその用途 |
| JP6835477B2 (ja) | 2015-04-30 | 2021-02-24 | 日東電工株式会社 | 透明導電性フィルム用キャリアフィルム及び積層体 |
| JP2017121696A (ja) | 2016-01-04 | 2017-07-13 | 積水化学工業株式会社 | 光透過性導電フィルム積層体の製造方法 |
| JP2017130419A (ja) | 2016-01-22 | 2017-07-27 | 日東電工株式会社 | 透明導電性フィルム |
| JP7007083B2 (ja) | 2016-09-30 | 2022-01-24 | 大日本印刷株式会社 | 導電性フィルム、タッチパネル、および画像表示装置 |
| TWI633563B (zh) * | 2016-12-15 | 2018-08-21 | 日商日東電工股份有限公司 | Transparent conductive film with carrier film and touch panel using the same |
| JP2018164985A (ja) | 2017-03-28 | 2018-10-25 | 積水化学工業株式会社 | 保護フィルム付き光透過性導電フィルム |
| JP6923415B2 (ja) * | 2017-10-23 | 2021-08-18 | 日東電工株式会社 | 透明導電性フィルムおよび透明導電性フィルム積層体 |
| JP7192798B2 (ja) | 2017-12-25 | 2022-12-20 | 大日本印刷株式会社 | 導電性フィルム、センサ、タッチパネル、画像表示装置、および保護フィルム付き導電性フィルム |
| JP6626996B2 (ja) * | 2019-01-17 | 2019-12-25 | 日東電工株式会社 | 透明導電性フィルム、透明導電性フィルム積層体及びタッチパネル |
| CN114555366B (zh) * | 2019-10-18 | 2023-03-24 | 昭和电工株式会社 | 透明导电膜层合体及其加工方法 |
-
2019
- 2019-12-24 EP EP19934145.4A patent/EP3988299A4/en active Pending
- 2019-12-24 CN CN202111353135.2A patent/CN113954479A/zh active Pending
- 2019-12-24 KR KR1020207034079A patent/KR102265033B1/ko active IP Right Grant
- 2019-12-24 US US17/615,429 patent/US11710581B2/en active Active
- 2019-12-24 JP JP2020526457A patent/JP6732161B1/ja active Active
- 2019-12-24 CN CN201980030487.9A patent/CN112292265B/zh active Active
- 2019-12-24 WO PCT/JP2019/050513 patent/WO2020255458A1/ja active Application Filing
- 2019-12-26 TW TW108147753A patent/TWI808289B/zh active
-
2020
- 2020-07-07 JP JP2020117151A patent/JP2021000826A/ja active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008073143A2 (en) | 2006-06-21 | 2008-06-19 | Cambrios Technologies Corporation | Methods of controlling nanostructure formations and shapes |
| WO2008046058A2 (en) | 2006-10-12 | 2008-04-17 | Cambrios Technologies Corporation | Nanowire-based transparent conductors and applications thereof |
| JP2008251529A (ja) | 2007-03-02 | 2008-10-16 | Nitto Denko Corp | 粘着剤層付き透明導電性フィルムおよびその製造方法 |
| JP2016107503A (ja) | 2014-12-05 | 2016-06-20 | 日東電工株式会社 | 透明導電性フィルム積層体及びそれを用いて得られるタッチパネル、並びに透明導電性フィルムの製造方法 |
| WO2016104204A1 (ja) * | 2014-12-26 | 2016-06-30 | 日東電工株式会社 | 透明導電性フィルム、透明導電性フィルム積層体及びタッチパネル |
| JP2016173714A (ja) * | 2015-03-17 | 2016-09-29 | 東レフィルム加工株式会社 | 導電積層体およびそれを用いたタッチパネル |
| JP2019149012A (ja) * | 2018-02-27 | 2019-09-05 | 日東電工株式会社 | 透明導電性フィルム積層体および透明導電性フィルムの製造方法 |
Non-Patent Citations (3)
| Title |
|---|
| CHEM. MATER., vol. 14, 2002, pages 4736 |
| J. AM. CHEM. SOC., vol. 126, 2004, pages 3892 - 3901 |
| J. AM. CHEM. SOC., vol. 129, 2007, pages 1733 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023195446A1 (ja) * | 2022-04-07 | 2023-10-12 | 日東電工株式会社 | 保護フィルム付透明導電性フィルム |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113954479A (zh) | 2022-01-21 |
| JP6732161B1 (ja) | 2020-07-29 |
| CN112292265A (zh) | 2021-01-29 |
| TWI808289B (zh) | 2023-07-11 |
| EP3988299A4 (en) | 2023-06-14 |
| US20220230774A1 (en) | 2022-07-21 |
| TW202108371A (zh) | 2021-03-01 |
| JP2021000826A (ja) | 2021-01-07 |
| KR20200145836A (ko) | 2020-12-30 |
| KR102265033B1 (ko) | 2021-06-15 |
| US11710581B2 (en) | 2023-07-25 |
| EP3988299A1 (en) | 2022-04-27 |
| JPWO2020255458A1 (ja) | 2021-09-13 |
| CN112292265B (zh) | 2021-11-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2020255458A1 (ja) | 透明導電フィルム積層体及びその加工方法 | |
| US11154902B2 (en) | Transparent conductive substrate and method for producing same | |
| JP7061734B2 (ja) | 透明導電フィルム積層体及びその加工方法 | |
| WO2020171022A1 (ja) | 透明導電基体及びこれを含むタッチパネル | |
| JP6855647B1 (ja) | 透明導電フィルムの製造方法 | |
| TWI775458B (zh) | 透明導電基體 | |
| JP6855648B1 (ja) | 透明導電フィルムの製造方法 | |
| WO2021131099A1 (ja) | 透明導電フィルムの製造方法 | |
| JP2024067269A (ja) | 透明導電フィルム積層体及びその製造方法並びに成形用透明導電フィルム積層体 | |
| WO2022138882A1 (ja) | 透明導電フィルム積層体 | |
| WO2021060149A1 (ja) | 透明ポリウレタン及びその製造方法並びに透明ポリウレタンを含む熱硬化性組成物及び透明導電フィルム |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2020526457 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20207034079 Country of ref document: KR Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19934145 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2019934145 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2019934145 Country of ref document: EP Effective date: 20220120 |