WO2020031952A1 - 血糖値上昇抑制用組成物及び血糖値上昇抑制方法 - Google Patents
血糖値上昇抑制用組成物及び血糖値上昇抑制方法 Download PDFInfo
- Publication number
- WO2020031952A1 WO2020031952A1 PCT/JP2019/030711 JP2019030711W WO2020031952A1 WO 2020031952 A1 WO2020031952 A1 WO 2020031952A1 JP 2019030711 W JP2019030711 W JP 2019030711W WO 2020031952 A1 WO2020031952 A1 WO 2020031952A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- increase
- suppressing
- composition
- blood glucose
- beverage
- Prior art date
Links
- 239000008280 blood Substances 0.000 title claims abstract description 204
- 210000004369 blood Anatomy 0.000 title claims abstract description 204
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 title claims abstract description 153
- 239000008103 glucose Substances 0.000 title claims abstract description 153
- 239000000203 mixture Substances 0.000 title claims abstract description 106
- 238000000034 method Methods 0.000 title claims abstract description 38
- YKGCBLWILMDSAV-GOSISDBHSA-N Isoxanthohumol Natural products O(C)c1c2C(=O)C[C@H](c3ccc(O)cc3)Oc2c(C/C=C(\C)/C)c(O)c1 YKGCBLWILMDSAV-GOSISDBHSA-N 0.000 claims abstract description 143
- YKGCBLWILMDSAV-SFHVURJKSA-N isoxanthohumol Chemical compound C1([C@H]2OC=3C(CC=C(C)C)=C(O)C=C(C=3C(=O)C2)OC)=CC=C(O)C=C1 YKGCBLWILMDSAV-SFHVURJKSA-N 0.000 claims abstract description 106
- 235000012054 meals Nutrition 0.000 claims abstract description 18
- 230000000291 postprandial effect Effects 0.000 claims abstract description 15
- 239000004480 active ingredient Substances 0.000 claims abstract description 12
- 235000013361 beverage Nutrition 0.000 claims description 53
- 235000013305 food Nutrition 0.000 claims description 33
- 201000010099 disease Diseases 0.000 claims description 18
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 18
- 235000013405 beer Nutrition 0.000 claims description 16
- 201000001421 hyperglycemia Diseases 0.000 claims description 16
- 235000014171 carbonated beverage Nutrition 0.000 claims description 10
- 235000013616 tea Nutrition 0.000 claims description 10
- 235000013399 edible fruits Nutrition 0.000 claims description 9
- 235000013334 alcoholic beverage Nutrition 0.000 claims description 7
- 235000013353 coffee beverage Nutrition 0.000 claims description 7
- 230000001476 alcoholic effect Effects 0.000 claims description 6
- 235000013322 soy milk Nutrition 0.000 claims description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 6
- 235000020510 functional beverage Nutrition 0.000 claims description 5
- 235000013311 vegetables Nutrition 0.000 claims description 5
- 235000013365 dairy product Nutrition 0.000 claims description 4
- 239000000126 substance Substances 0.000 abstract description 23
- FUSADYLVRMROPL-UHFFFAOYSA-N demethylxanthohumol Natural products CC(C)=CCC1=C(O)C=C(O)C(C(=O)C=CC=2C=CC(O)=CC=2)=C1O FUSADYLVRMROPL-UHFFFAOYSA-N 0.000 description 37
- ORXQGKIUCDPEAJ-YRNVUSSQSA-N xanthohumol Chemical compound COC1=CC(O)=C(CC=C(C)C)C(O)=C1C(=O)\C=C\C1=CC=C(O)C=C1 ORXQGKIUCDPEAJ-YRNVUSSQSA-N 0.000 description 37
- UVBDKJHYMQEAQV-UHFFFAOYSA-N xanthohumol Natural products OC1=C(CC=C(C)C)C(OC)=CC(OC)=C1C(=O)C=CC1=CC=C(O)C=C1 UVBDKJHYMQEAQV-UHFFFAOYSA-N 0.000 description 37
- 235000008209 xanthohumol Nutrition 0.000 description 37
- 230000000694 effects Effects 0.000 description 28
- 239000003814 drug Substances 0.000 description 27
- 230000037406 food intake Effects 0.000 description 25
- 238000012360 testing method Methods 0.000 description 22
- 229940079593 drug Drugs 0.000 description 19
- 241000218228 Humulus Species 0.000 description 15
- 206010012601 diabetes mellitus Diseases 0.000 description 15
- 239000000284 extract Substances 0.000 description 14
- 241001465754 Metazoa Species 0.000 description 10
- 230000037396 body weight Effects 0.000 description 9
- 235000009200 high fat diet Nutrition 0.000 description 8
- 230000001629 suppression Effects 0.000 description 8
- 230000036541 health Effects 0.000 description 7
- 238000010438 heat treatment Methods 0.000 description 7
- 238000002360 preparation method Methods 0.000 description 7
- 244000269722 Thea sinensis Species 0.000 description 6
- 230000009471 action Effects 0.000 description 6
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 235000021590 normal diet Nutrition 0.000 description 6
- 230000001225 therapeutic effect Effects 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical class CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- 241000699670 Mus sp. Species 0.000 description 5
- 230000008859 change Effects 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 241000282412 Homo Species 0.000 description 4
- 244000025221 Humulus lupulus Species 0.000 description 4
- 235000008694 Humulus lupulus Nutrition 0.000 description 4
- 241000699666 Mus <mouse, genus> Species 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 235000013376 functional food Nutrition 0.000 description 4
- 239000008187 granular material Substances 0.000 description 4
- 235000013402 health food Nutrition 0.000 description 4
- 238000004128 high performance liquid chromatography Methods 0.000 description 4
- 230000002401 inhibitory effect Effects 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 238000002156 mixing Methods 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 102000004877 Insulin Human genes 0.000 description 3
- 108090001061 Insulin Proteins 0.000 description 3
- 235000006468 Thea sinensis Nutrition 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 239000001768 carboxy methyl cellulose Substances 0.000 description 3
- 230000001684 chronic effect Effects 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 230000003345 hyperglycaemic effect Effects 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 229940125396 insulin Drugs 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 235000013336 milk Nutrition 0.000 description 3
- 239000008267 milk Substances 0.000 description 3
- 210000004080 milk Anatomy 0.000 description 3
- 235000019520 non-alcoholic beverage Nutrition 0.000 description 3
- -1 prenyl chalcone Chemical compound 0.000 description 3
- 235000014214 soft drink Nutrition 0.000 description 3
- 102000011690 Adiponectin Human genes 0.000 description 2
- 108010076365 Adiponectin Proteins 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- 241000725101 Clea Species 0.000 description 2
- 206010052804 Drug tolerance Diseases 0.000 description 2
- 240000005979 Hordeum vulgare Species 0.000 description 2
- 235000007340 Hordeum vulgare Nutrition 0.000 description 2
- 230000005856 abnormality Effects 0.000 description 2
- 239000003674 animal food additive Substances 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 235000020279 black tea Nutrition 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 150000001720 carbohydrates Chemical class 0.000 description 2
- 235000014633 carbohydrates Nutrition 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 235000005911 diet Nutrition 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 239000000469 ethanolic extract Substances 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 235000019634 flavors Nutrition 0.000 description 2
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 230000026781 habituation Effects 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 229930014626 natural product Natural products 0.000 description 2
- 238000007410 oral glucose tolerance test Methods 0.000 description 2
- 150000008442 polyphenolic compounds Chemical class 0.000 description 2
- 235000013824 polyphenols Nutrition 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 238000007619 statistical method Methods 0.000 description 2
- 230000001954 sterilising effect Effects 0.000 description 2
- 238000004659 sterilization and disinfection Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 238000011282 treatment Methods 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 241000271566 Aves Species 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000218235 Cannabaceae Species 0.000 description 1
- DQFBYFPFKXHELB-UHFFFAOYSA-N Chalcone Natural products C=1C=CC=CC=1C(=O)C=CC1=CC=CC=C1 DQFBYFPFKXHELB-UHFFFAOYSA-N 0.000 description 1
- 241000272201 Columbiformes Species 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 206010022489 Insulin Resistance Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 208000017442 Retinal disease Diseases 0.000 description 1
- 206010038923 Retinopathy Diseases 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- 238000000692 Student's t-test Methods 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 210000001789 adipocyte Anatomy 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 235000021329 brown rice Nutrition 0.000 description 1
- 235000019577 caloric intake Nutrition 0.000 description 1
- 235000021074 carbohydrate intake Nutrition 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 235000005513 chalcones Nutrition 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 102000038379 digestive enzymes Human genes 0.000 description 1
- 108091007734 digestive enzymes Proteins 0.000 description 1
- 150000002016 disaccharides Chemical class 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 239000003651 drinking water Substances 0.000 description 1
- 235000020188 drinking water Nutrition 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 235000015897 energy drink Nutrition 0.000 description 1
- 239000003889 eye drop Substances 0.000 description 1
- 229940012356 eye drops Drugs 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- ZONYXWQDUYMKFB-UHFFFAOYSA-N flavanone group Chemical group O1C(CC(=O)C2=CC=CC=C12)C1=CC=CC=C1 ZONYXWQDUYMKFB-UHFFFAOYSA-N 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 235000012631 food intake Nutrition 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 235000014080 ginger ale Nutrition 0.000 description 1
- 238000007446 glucose tolerance test Methods 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 230000002641 glycemic effect Effects 0.000 description 1
- 235000009569 green tea Nutrition 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- 239000008274 jelly Substances 0.000 description 1
- 208000017169 kidney disease Diseases 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 235000020094 liqueur Nutrition 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- 235000021056 liquid food Nutrition 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 208000030159 metabolic disease Diseases 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 235000020124 milk-based beverage Nutrition 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 239000007923 nasal drop Substances 0.000 description 1
- 229940100662 nasal drops Drugs 0.000 description 1
- 201000001119 neuropathy Diseases 0.000 description 1
- 230000007823 neuropathy Effects 0.000 description 1
- 235000020333 oolong tea Nutrition 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 208000033808 peripheral neuropathy Diseases 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 125000001844 prenyl group Chemical group [H]C([*])([H])C([H])=C(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000002953 preparative HPLC Methods 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 235000020083 shōchū Nutrition 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 235000015096 spirit Nutrition 0.000 description 1
- 235000011496 sports drink Nutrition 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- DQFBYFPFKXHELB-VAWYXSNFSA-N trans-chalcone Chemical group C=1C=CC=CC=1C(=O)\C=C\C1=CC=CC=C1 DQFBYFPFKXHELB-VAWYXSNFSA-N 0.000 description 1
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 1
- 235000015041 whisky Nutrition 0.000 description 1
- 235000013618 yogurt Nutrition 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23C—DAIRY PRODUCTS, e.g. MILK, BUTTER OR CHEESE; MILK OR CHEESE SUBSTITUTES; MAKING THEREOF
- A23C9/00—Milk preparations; Milk powder or milk powder preparations
- A23C9/152—Milk preparations; Milk powder or milk powder preparations containing additives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23F—COFFEE; TEA; THEIR SUBSTITUTES; MANUFACTURE, PREPARATION, OR INFUSION THEREOF
- A23F3/00—Tea; Tea substitutes; Preparations thereof
- A23F3/16—Tea extraction; Tea extracts; Treating tea extract; Making instant tea
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23F—COFFEE; TEA; THEIR SUBSTITUTES; MANUFACTURE, PREPARATION, OR INFUSION THEREOF
- A23F5/00—Coffee; Coffee substitutes; Preparations thereof
- A23F5/24—Extraction of coffee; Coffee extracts; Making instant coffee
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L11/00—Pulses, i.e. fruits of leguminous plants, for production of food; Products from legumes; Preparation or treatment thereof
- A23L11/60—Drinks from legumes, e.g. lupine drinks
- A23L11/65—Soy drinks
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/02—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation containing fruit or vegetable juices
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/38—Other non-alcoholic beverages
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/40—Complete food formulations for specific consumer groups or specific purposes, e.g. infant formula
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
- A61K31/352—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom condensed with carbocyclic rings, e.g. methantheline
- A61K31/353—3,4-Dihydrobenzopyrans, e.g. chroman, catechin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0056—Mouth soluble or dispersible forms; Suckable, eatable, chewable coherent forms; Forms rapidly disintegrating in the mouth; Lozenges; Lollipops; Bite capsules; Baked products; Baits or other oral forms for animals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0087—Galenical forms not covered by A61K9/02 - A61K9/7023
- A61K9/0095—Drinks; Beverages; Syrups; Compositions for reconstitution thereof, e.g. powders or tablets to be dispersed in a glass of water; Veterinary drenches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12G—WINE; PREPARATION THEREOF; ALCOHOLIC BEVERAGES; PREPARATION OF ALCOHOLIC BEVERAGES NOT PROVIDED FOR IN SUBCLASSES C12C OR C12H
- C12G3/00—Preparation of other alcoholic beverages
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2200/00—Function of food ingredients
- A23V2200/30—Foods, ingredients or supplements having a functional effect on health
- A23V2200/328—Foods, ingredients or supplements having a functional effect on health having effect on glycaemic control and diabetes
Definitions
- the present invention relates to a composition for suppressing an increase in blood glucose level.
- the present invention also relates to a method for suppressing an increase in blood glucose level and to the use of isoxanthohumol for suppressing an increase in blood glucose level.
- Diabetes refers to a group of metabolic diseases characterized by chronic hyperglycemia due to lack of insulin action.
- the number of patients and reserves have increased, especially in developed and emerging countries from the perspective of increasing medical costs.
- Past large-scale clinical trials and the like have shown that lifestyle-related interventions such as restriction of food intake (total energy intake) and moderate exercise are effective as approaches to prevent and improve the onset of diabetes.
- restriction of food intake total energy intake
- moderate exercise are effective as approaches to prevent and improve the onset of diabetes.
- forcing a strong lifestyle intervention for a long period of time entails a physical burden on the subject, and there is a need for a method of preventing or improving diabetes that has less physical burden.
- Non-patent Document 1 It is considered clinically meaningful to correct postprandial hyperglycemia, that is, to suppress an increase in blood glucose after ingesting foods containing carbohydrates.
- the use of functional foods is considered to be beneficial from the viewpoint of controlling medical costs.
- Labeled food is commercially available. However, most of them suppress the physical absorption of sugar in the small intestine and suppress the function of digestive enzymes that break down polysaccharides and disaccharides into monosaccharides, and the timing of ingestion is limited to before and during meals There is a problem that it is done.
- Xanthohumol is known as a component contained in hops (scientific name: Humulus lupulus), which is a plant of the hemp family and used as a raw material for beer.
- Xanthohumol is a kind of polyphenol whose molecular formula is represented by C 21 H 22 O 5 and is classified as “prenyl chalcone” among polyphenols.
- Non-Patent Document 2 shows that when xanthohumol is continuously administered at 60 mg / kg / day to a high-fat diet-ingested model mouse, the fasting blood glucose level decreases. In addition, it has been reported that, when xanthohumol is continuously administered at 30 or 60 mg / kg / day, the blood insulin level during fasting decreases.
- Non-Patent Literature 3 shows that, in a high-fat diet-ingested model mouse, the blood sugar level decreases when xanthohumol is mixed at a concentration of 10 mg / L in drinking water for a long time.
- Patent Literature 1 shows the result that when a diabetic model mouse was bred with a feed in which a xanthohumol-rich material was mixed at a ratio of 1%, an increase in blood glucose in a glucose tolerance test was suppressed. .
- These results shown in Non-Patent Literatures 2 and 3 and Patent Literature 1 are the results of continuous ingestion of xanthohumol, and no effect is reported by a single ingestion of xanthohumol.
- xanthohumol lowers blood glucose levels, but xanthohumol is irreversibly transformed into isoxanthohumol with heating.
- xanthohumol is irreversibly transformed into isoxanthohumol with heating.
- most of the beer is converted to isoxanthohumol during the production process.
- foods with functional indication, and the like for which it is required to guarantee the amount of functional components during long-term storage, it is desired to provide particularly stable substances and compositions.
- Non-Patent Document 4 describes that anticancer activity is very effective with xanthohumol, but very weak with isoxanthohumol. Therefore, it is not easy to estimate the biological activity of isoxanthohumol based on information on the biological activity of xanthohumol.
- Patent Document 2 discloses that xanthohumol and hops dehydrated ethanolic extract promote secretion of adiponectin, a physiologically active substance from 3T3-L1 adipocytes, which contributes to improvement of insulin resistance (a state in which insulin action is not sufficiently exerted).
- adiponectin a physiologically active substance from 3T3-L1 adipocytes
- a heat-stable substance that can be used for specified health foods or functionally labeled foods or the like that requires the assurance of the amount of functionally-related components during long-term storage, and that contributes to suppression of blood sugar level rise is required.
- the timing of ingestion is not limited to before or during a meal, and a substance that can suppress an increase in blood glucose level after a meal is desirable.
- An object of the present invention is to provide a composition for suppressing an increase in blood glucose level, comprising a heat stable substance as an active ingredient. Further, another object of the present invention is to provide a composition for suppressing an increase in blood glucose level, which can suppress the increase in blood glucose level after meals without limiting the timing of ingestion to before or during a meal. Another object of the present invention is to provide a method for suppressing an increase in blood glucose level and the like.
- isoxanthohumol has a blood sugar level increase inhibitory action.
- Isoxanthohumol is a compound having higher thermal stability than xanthohumol. Isoxanthohumol exhibited a stronger blood sugar level inhibitory effect than xanthohumol. Also, isoxanthohumol does not show a blood sugar rise inhibitory effect in a single ingestion, blood sugar rise suppression effect is recognized only by continuous ingestion, instead of physically suppressing sugar absorption derived from carbohydrates, It can be said that the blood glucose level is suppressed by an action mechanism other than the above.
- the timing of ingesting isoxanthohumol to obtain the effect of suppressing an increase in postprandial blood glucose level is not limited to before and during a meal.
- Isoxanthohumol exhibits an effect of suppressing an increase in blood sugar level and is excellent in beverage suitability. For this reason, by blending isoxanthohumol, it becomes possible to develop functional beverages and the like that contribute to maintenance and promotion of health.
- the present invention relates to, but is not limited to, the following composition for suppressing an increase in blood sugar level, a method for suppressing an increase in blood sugar level, and the like.
- a composition for suppressing an increase in blood glucose level comprising isoxanthohumol as an active ingredient.
- the composition for suppressing an increase in blood sugar level according to [1] which is used for suppressing an increase in blood sugar level after meals.
- beverage is a tea-based beverage, a coffee beverage, an alcoholic beverage, a non-alcoholic beer-taste beverage, a carbonated beverage, a functional beverage, a fruit / vegetable beverage, a dairy beverage, a soy milk beverage, or a flavored water.
- the composition for blood-sugar level rise suppression containing a heat-stable substance as an active ingredient can be provided. Further, according to the present invention, the composition for suppressing an increase in blood glucose level, which can suppress the rise in blood glucose level after meals, without limiting the timing of ingestion to before or during a meal. ADVANTAGE OF THE INVENTION According to this invention, the blood glucose level rise suppression method can be provided.
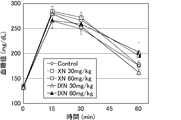
- FIG. 1 is a graph showing the change in blood glucose level after glucose administration in each group to which isoxanthohumol or xanthohumol was administered once.
- FIG. 2 is a graph showing a change in blood glucose level after glucose administration in each group to which isoxanthohumol or xanthohumol was continuously administered (#: p ⁇ 0.05 vs. high fat diet group).
- FIG. 3 is a graph showing the area under the time curve of blood glucose concentration (AUC) of each group to which isoxanthohumol or xanthohumol was continuously administered (++: p ⁇ 0.01% vs. normal diet group) ).
- AUC blood glucose concentration
- composition for suppressing an increase in blood glucose level of the present invention contains isoxanthohumol as an active ingredient.
- isoxanthohumol has an action of suppressing an increase in blood sugar level.
- Ingestion of isoxanthohumol can suppress a postprandial rise in blood glucose level.
- the effect of suppressing the rise in blood glucose level by isoxanthohumol can be obtained by continuously taking isoxanthohumol.
- the intake timing of isoxanthohumol is not limited to before or during a meal, and at any timing. Can be taken.
- Isoxanthohumol can be prepared, for example, from hop (Humulus upulus) extract via a process such as heating. By heating the hop extract, isoxanthohumol can be generated in the extract.
- the hop extract is usually prepared by extracting hop cones with a solvent and, if necessary, through a process relating to purification, and can be obtained by a known hop extract preparation method. Examples of the extraction method include an extraction method using an ethanol solvent used as a method for preparing a hop extract used for beer brewing. Hop extracts are commercially available, and commercially available hop extracts can also be used.
- the hop extract is preferably heated at 80 to 140 ° C.
- isoxanthohumol (more preferably 85 to 100 ° C.) for 15 minutes to 5 hours (more preferably 20 minutes to 3 hours) to produce isoxanthohumol.
- Purification of the hop extract for preparing isoxanthohumol is performed by a known method. Examples of the purification method include methods such as use of an HPLC or an adsorption column, and precipitation using a change in solubility.
- Isoxanthohumol can also be produced by heating xanthohumol to isomerize it. The heating temperature at this time is preferably 80 to 140 ° C. (more preferably 85 to 100 ° C.) for 15 minutes to 5 hours (more preferably 20 minutes to 3 hours).
- the isoxanthohumol obtained by the isomerization treatment can be concentrated or purified by a known method (for example, filtration, concentration under reduced pressure, lyophilization and the like) as necessary.
- Isoxanthohumol is a compound contained in natural products and foods and drinks, and has a dietary experience. For this reason, from the viewpoint of safety, it is considered that isoxanthohumol has few problems even when taken daily, for example. Therefore, according to the present invention, it is possible to provide a composition for suppressing an increase in blood sugar level, which contains a highly safe component as an active ingredient, and a method for suppressing an increase in blood sugar level. In addition, it has been reported that isoxanthohumol is stable even at a high temperature condition of, for example, 100 ° C.
- sterilization conditions are defined as standard standards for soft drinks, etc., for example, those with a pH of 4.0 or more (excluding those with a pH of 4.6 or more and a water activity exceeding 0.94) Requires heating at 85 ° C. for 30 minutes.
- isoxanthohumol is considered to have a high beverage suitability. Therefore, according to the present invention, it is possible to provide various functional foods, functional drinks, and the like that exhibit a blood sugar level increase suppressing action, are stable to heat, and contribute to maintenance and promotion of health.
- the timing of ingestion is not limited to before or during a meal, and provides various functional foods, functional drinks, and the like that exhibit a blood sugar level increase suppressing effect, for example, a postprandial blood glucose level increase suppressing effect. It becomes possible.
- Isoxanthohumol can be used for suppressing an increase in blood sugar level, for example, for suppressing an increase in postprandial blood sugar level.
- the composition for suppressing an increase in blood sugar level of the present invention by using isoxanthohumol as an active ingredient, exhibits an effect of suppressing an increase in blood sugar level, and is suitably used, for example, to suppress an increase in postprandial blood sugar level. .
- the use of the composition for suppressing an increase in blood glucose level of the present invention enables prevention or improvement of postprandial hyperglycemia.
- the composition for suppressing an increase in blood glucose level of the present invention can be used for preventing or improving postprandial hyperglycemia, and for preventing or improving a condition or disease caused by postprandial hyperglycemia.
- Postprandial hyperglycemia means a condition in which the blood sugar level increases after carbohydrate intake.
- Conditions or diseases caused by postprandial hyperglycemia include, for example, diabetes, arteriosclerosis, neuropathy, nephropathy, and retinopathy.
- the composition for suppressing an increase in blood glucose level of the present invention is useful for preventing or improving such a condition or disease.
- prevention of a condition or disease includes preventing the onset of the condition or disease, delaying the onset of the condition or disease, reducing the incidence of the condition or disease, reducing the risk of developing the condition or disease. It refers to reducing. Improvement of a condition or disease refers to relieving the subject from the condition or disease, reducing the symptoms of the condition or disease, delaying or preventing the progress of the condition or disease, and the like.
- the composition for suppressing an increase in blood glucose level of the present invention may contain only isoxanthohumol as an active ingredient.
- the composition for suppressing an increase in blood glucose level of the present invention can be applied to either therapeutic use (medical use) or non-therapeutic use (non-medical use).
- the composition for suppressing an increase in blood glucose level of the present invention can be in the form of food and drink, medicine, quasi-drug, feed and the like.
- the composition for suppressing an increase in blood sugar level of the present invention may itself be a food or drink for suppressing an increase in blood sugar level, a medicine, a quasi-drug, a feed, etc. Alternatively, it may be a preparation or the like.
- the composition for suppressing an increase in blood glucose level of the present invention can be provided in the form of an agent as an example, but is not limited thereto.
- the agent can be provided as it is as a composition or as a composition containing the agent.
- the composition for suppressing an increase in blood sugar level of the present invention can also be referred to as an agent for suppressing an increase in blood sugar level.
- the composition for suppressing an increase in blood glucose level of the present invention is preferably an oral composition.
- the oral composition include foods and drinks, oral medicines, quasi-drugs, and feeds, and are preferably foods and drinks or oral medicines, and more preferably foods and drinks.
- the composition for suppressing an increase in blood glucose level of the present invention contains isoxanthohumol as an active ingredient.
- the composition for suppressing an increase in blood glucose level of the present invention can contain any additive and any component in addition to isoxanthohumol as long as the effects of the present invention are not impaired.
- These additives and components can be selected according to the form of the composition and the like, and those generally usable for foods and drinks, medicines, quasi-drugs, feed, feed additives and the like can be used.
- the production method is not particularly limited, and it can be produced by a general method.
- the composition for suppressing an increase in blood glucose level containing isoxanthohumol can also be produced, for example, by blending a hop extract in the production thereof and heating the hop extract.
- composition for suppressing an increase in blood glucose level of the present invention when used as a food or drink, isoxanthohumol is added to a component (for example, a food material or a food additive used as needed) that can be used in the food or drink.
- a component for example, a food material or a food additive used as needed
- foods and drinks can be obtained.
- Foods and drinks are not particularly limited, and include, for example, general foods and drinks, health foods, health drinks, functionally labeled foods, foods for specified health use, foods and drinks for the sick.
- isoxanthohumol is heat-stable, so that the composition for suppressing an increase in blood glucose level of the present invention can be provided in various beverage forms.
- a preferred example of the composition for suppressing an increase in blood glucose level of the present invention is a beverage.
- the beverage may be a non-alcoholic beverage or an alcoholic beverage.
- the non-alcoholic beverage include a tea-based beverage, a coffee beverage, a non-alcoholic beer taste beverage, a carbonated beverage, a functional beverage, a fruit / vegetable beverage, a dairy beverage, a soy milk beverage, and a flavored water.
- the composition for suppressing an increase in blood glucose level of the present invention is a beverage, a tea beverage, a coffee beverage, an alcoholic beverage, a non-alcoholic beer taste beverage, a carbonated beverage, a functional beverage, a fruit / vegetable beverage, a dairy beverage, and soy milk It is preferably a beverage or a flavored water.
- the composition for suppressing an increase in blood glucose level of the present invention is a tea-based beverage, it is preferably a black tea beverage or a sugar-free tea beverage.
- the sugar-free tea beverage include a green tea beverage, an oolong tea beverage, a barley tea beverage, a brown rice tea beverage, a pigeon barley tea beverage, and a sugar-free black tea beverage.
- the composition for suppressing an increase in blood glucose level of the present invention is a coffee beverage, it is preferably a packaged coffee or a liquid coffee.
- alcoholic beverages examples include beer, beer-based beverages, beer and alcoholic beverages other than beer-based beverages.
- the composition for suppressing an increase in blood glucose level of the present invention is a beer-based beverage, it is preferably a low-malt beer or a third beer.
- the composition for suppressing an increase in blood glucose level of the present invention is an alcoholic beverage other than beer and beer-based beverages, it is preferably shochu, chuhai, liqueur, cocktail, spirits, or whiskey.
- non-alcoholic beer-taste beverage in the present specification means a carbonated beverage having a beer-like flavor, which is usually a non-fermented non-alcoholic beverage, which does not substantially contain alcohol.
- the non-alcoholic beer taste beverage does not exclude a beverage containing an extremely small amount of alcohol that cannot be detected.
- composition for suppressing an increase in blood sugar level of the present invention is a carbonated beverage
- it is preferably a cola flavor beverage, a transparent carbonated beverage, ginger ale, a juice-based carbonated beverage, a milk-containing carbonated beverage, or a sugar-free carbonated beverage.
- the composition for suppressing an increase in blood glucose level of the present invention is a functional drink, it is preferably a sports drink, an energy drink, a health support drink, or a pouch jelly drink.
- composition for suppressing an increase in blood glucose level of the present invention is a fruit / vegetable beverage, it is preferably a 100% fruit beverage, a fruit-containing beverage, a low juice-containing soft drink, a fruit-containing fruit beverage, or a pulp beverage.
- composition for suppressing an increase in blood glucose level of the present invention is a milk-based beverage, it is preferably milk, a drink yogurt, a lactic acid bacterium beverage or a milk-containing soft drink.
- composition for suppressing an increase in blood glucose level of the present invention is a soymilk drink, it is preferably a soymilk or soybean drink.
- the form of the beverage is not particularly limited, and may be a packaged beverage.
- Containers for packaged beverages are not particularly limited, and containers of any form and material may be used.
- metal containers such as aluminum cans and steel cans; resin containers such as PET bottles; paper such as paper packs Containers; glass containers such as glass bottles; and commonly used containers such as wooden containers such as barrels can be used.
- composition for suppressing an increase in blood glucose level of the present invention is used as a drug or a quasi-drug
- a pharmacologically acceptable carrier, additives and the like added as necessary are mixed with isoxanthohumol.
- Such carriers, additives, and the like can be used in medicines or quasi-drugs, as long as they are pharmacologically acceptable, for example, excipients, binders, disintegrants, lubricants, One or more of an antioxidant, a coloring agent, and the like are included.
- Examples of the administration (ingestion) form of the medicament or quasi-drug include oral, enteral, transmucosal, and injection, and the oral administration form is preferable from the viewpoint of more sufficiently obtaining the effects of the present invention.
- the composition for suppressing an increase in blood glucose level is used as a medicine, it is preferably used as an oral medicine.
- Examples of the dosage form of the preparation for oral administration include liquids, tablets, powders, fine granules, granules, dragees, capsules, suspensions, emulsions, chewables and the like.
- dosage forms for parenteral administration include injections, inhalants, infusions, suppositories, transdermal absorbers, nasal drops, eye drops, creams, gels, lotions and the like.
- the medicament may be a non-human veterinary medicament.
- Feed also includes feed additives.
- feed include feed for livestock used for cattle, pigs, chickens, sheep, horses, and the like; feed for small animals used for rabbits, rats, mice, and the like; and pet food used for dogs, cats, small birds, and the like.
- the content of isoxanthohumol in the composition for suppressing an increase in blood glucose level of the present invention is not particularly limited, and can be set according to the form and the like.
- the content of isoxanthohumol is, for example, preferably 0.0001% by weight or more, more preferably 0.001% by weight or more, and further preferably 90% by weight or less in the composition for suppressing an increase in blood glucose level.
- the content of isoxanthohumol in the composition for suppressing an increase in blood sugar level is preferably 0.0001 to 90% by weight, more preferably 0.001 to 90% by weight.
- the content of isoxanthohumol is preferably in the above range.
- Isoxanthohumol contained in the composition for suppressing an increase in blood glucose level can be quantified by, for example, high performance liquid chromatography (HPLC) or LC-MS / MS.
- composition for suppressing an increase in blood glucose level of the present invention can be taken or administered by an appropriate method depending on the form. From the viewpoint of sufficiently obtaining the effects of the present invention, the composition for suppressing an increase in blood glucose level of the present invention is preferably orally ingested (orally administered).
- the amount of intake (also referred to as dose) of the composition for suppressing an increase in blood glucose level of the present invention is not particularly limited, and may be any amount (effective amount) capable of achieving an effect of suppressing an increase in blood glucose level. What is necessary is just to set suitably according to an administration method, a subject's weight, etc.
- the amount of the composition for suppressing an increase in blood glucose level is 60 kg / day, preferably 1 to 200 mg / day as isoxanthohumol. And more preferably 5 to 60 mg. It is preferable to take the above amount once or more times a day, for example, once to several times a day (eg two to three times).
- the timing of ingesting the composition for suppressing an increase in blood glucose level of the present invention is not particularly limited, and the composition may be ingested at least once a day at any time.
- the composition for suppressing an increase in blood glucose level of the present invention may be an oral composition for allowing an adult to ingest the above amount of isoxanthohumol per day per 60 kg body weight.
- the composition for suppressing an increase in blood glucose level of the present invention is to be taken continuously.
- the composition for suppressing an increase in blood sugar level is preferably taken continuously for 2 weeks or more, more preferably for 3 weeks or more.
- the subject to which the composition for suppressing an increase in blood glucose level of the present invention is to be taken (also referred to as the subject to be administered) is not particularly limited. Preferably it is a human or non-human mammal, more preferably a human.
- the subject to which the composition for suppressing an increase in blood sugar level of the present invention is to be taken is preferably a subject that requires or wants to suppress the increase in blood sugar level, and more preferably a subject that requires or wants to suppress the increase in blood sugar level after eating.
- the subject to which the composition for suppressing an increase in blood sugar level of the present invention is taken is not limited to diabetic patients or diabetic reserves.
- the composition for suppressing an increase in the blood glucose level of the present invention can be used, for example, for hyperglycemic humans whose blood glucose level is 126 mg / dL or more on an empty stomach or whose oral glucose tolerance test (OGTT) 2 hour value is 200 mg / dL or more.
- the composition for suppressing an increase in blood glucose level of the present invention can be used for therapeutic purposes.
- the composition for suppressing an increase in blood glucose level of the present invention can be used for humans (boundary type) having an OGTT 2 hour value of 140 to 199 mg / dL in order to prevent the deterioration of diabetes.
- the composition for suppressing an increase in blood glucose level of the present invention can also be used for healthy persons for preventing or improving a hyperglycemic state.
- the composition for suppressing an increase in blood glucose level of the present invention contains a highly safe component contained in natural products and foods and drinks as an active ingredient.
- the composition for suppressing an increase in blood sugar level of the present invention exerts an effect of suppressing an increase in blood sugar level when continuously taken.
- the composition for suppressing an increase in blood glucose level of the present invention is not limited to be taken before or during a meal, and can be taken at any timing.
- the composition for suppressing an increase in blood glucose level of the present invention for example, the above-mentioned diabetic patients, diabetic reserve army, borderline diabetic patients, humans who are concerned about postprandial hyperglycemic state, healthy subjects, and the like are taken daily.
- it is useful as a health food, a functionally labeled food, or the like to be used for preventing or improving diabetes by suppressing an increase in blood sugar level.
- the composition for suppressing an increase in blood glucose level of the present invention may have an indication of a function based on the action of suppressing an increase in blood glucose level.
- the composition for suppressing an increase in the blood glucose level of the present invention includes, for example, “suppress the increase in the blood glucose level after eating”, “reduce the increase in the blood glucose level after eating”, “reduce the blood glucose level”, and " One or two or more indications of "for those who are concerned,””for those who are concerned about their blood sugar level after meals” and "improve their constitution in which the blood sugar level tends to increase” may be given.
- the composition for suppressing an increase in blood glucose level of the present invention is preferably a food or drink to which the above-mentioned function is indicated.
- the display may be a display indicating that the display is used to obtain the above function.
- the present invention also encompasses a method for suppressing an increase in blood glucose level in which isoxanthohumol is ingested or administered to a subject.
- the method for suppressing an increase in blood glucose level is preferably a method for suppressing an increase in blood glucose level after a meal.
- the present invention also includes a method for preventing or ameliorating a condition or disease caused by postprandial hyperglycemia, in which isoxanthohumol is ingested or administered.
- Isoxanthohumol can be used to prevent or ameliorate a condition or disease caused by postprandial hyperglycemia by suppressing an increase in postprandial blood glucose level.
- the method may be a therapeutic or a non-therapeutic method.
- “Non-therapeutic" is a concept that does not include medical practice, i.e., human surgery, treatment or diagnosis.
- isoxanthohumol is continuously taken or administered to the subject.
- the subject in the above method is preferably a human or non-human mammal, more preferably a human.
- the present invention also includes the following uses.
- the use is preferably for suppressing an increase in postprandial blood glucose level.
- the invention also includes the use of isoxanthohumol for preventing or ameliorating a condition or disease caused by postprandial hyperglycemia.
- the above uses are preferably for use in human or non-human mammals, more preferably humans.
- the use may be a therapeutic use or a non-therapeutic use.
- isoxanthohumol when isoxanthohumol is continuously taken or administered to a subject, it is preferably taken or administered continuously for 2 weeks or more, and is taken or administered continuously for 3 weeks or more. Is more preferable.
- the timing of ingesting or administering isoxanthohumol is not particularly limited.
- the subject may be administered or ingested isoxanthohumol once or more times a day, for example, once to several times a day (eg, two to three times).
- isoxanthohumol is orally administered or taken.
- the subject may be administered or ingested in an amount (also referred to as an effective amount) of isoxanthohumol in which an effect of suppressing an increase in blood glucose level is obtained.
- the preferred ingestion amount and administration subject of isoxanthohumol are the same as those of the above-described composition for suppressing an increase in blood glucose level of the present invention.
- Isoxanthohumol may be administered or taken as it is, or may be administered or taken as a composition containing isoxanthohumol.
- the composition for suppressing an increase in blood glucose level of the present invention described above may be administered or taken.
- isoxanthohumol can be used for production of foods and drinks, medicines, quasi-drugs, feeds and the like used for suppressing an increase in blood sugar level.
- the present invention also includes use of isoxanthohumol for producing a composition for suppressing an increase in blood glucose level.
- ⁇ Preparation Example 1> Preparation of Xanthohumol and Isoxanthohumol Isoxanthohumol and xanthohumol were isolated and purified from a hop extract (manufactured by Asama Kasei Co., Ltd.) by the following method. That is, isoxanthohumol and xanthohumol are purified from a hop extract by normal phase column chromatography, reverse phase column chromatography, and preparative HPLC purification, respectively, and the purity is 95% or more by HPLC analysis. It was confirmed.
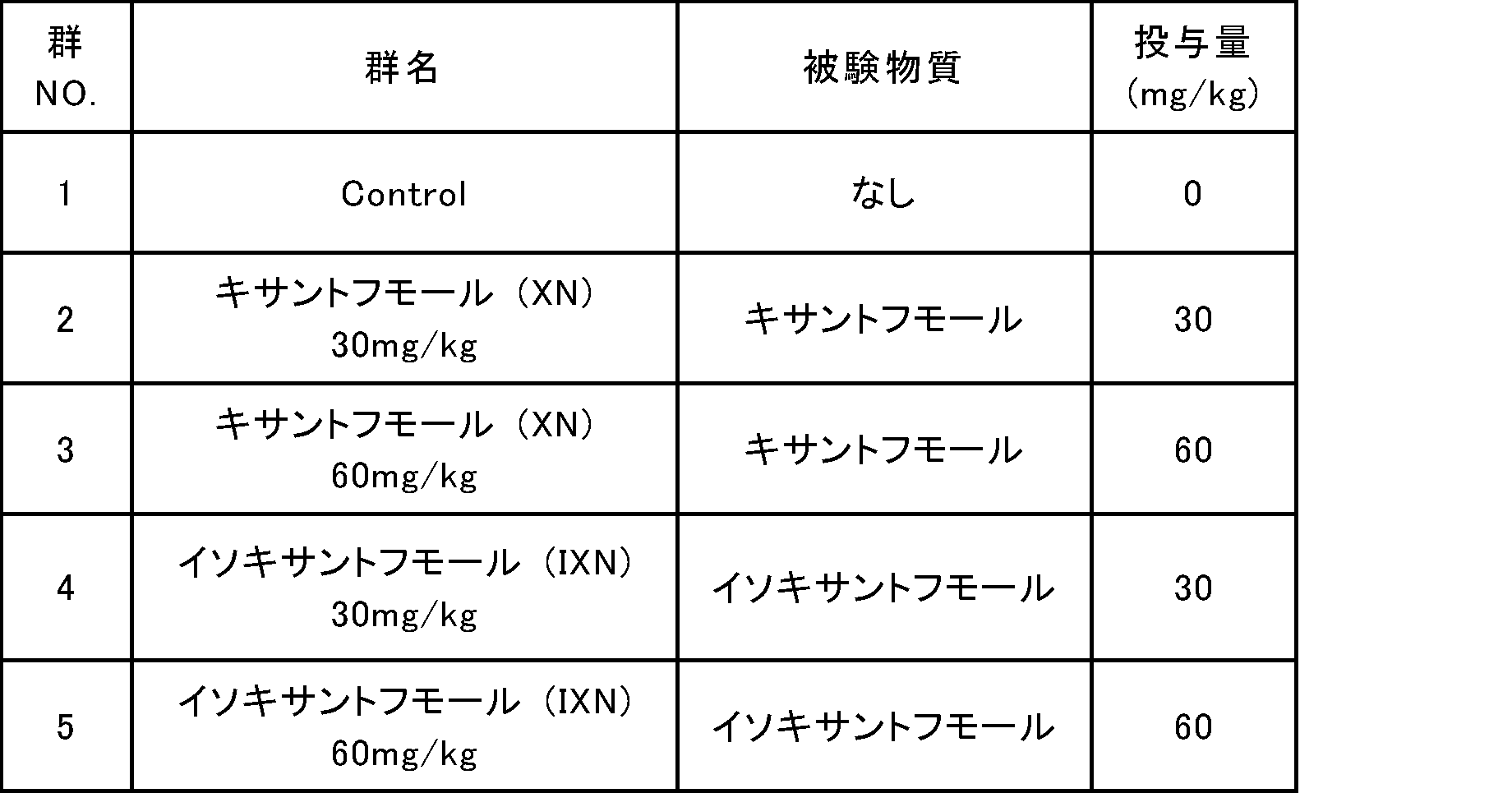
- Table 1 shows the group composition (group name, test substance, dose of test substance) in this test.
- Xanthohumol and isoxanthohumol used were the samples (purity 95% or more) obtained in Preparation Example 1.
- Dose in the table is the dose (mg) of the test substance per kg of body weight.
- an administration liquid obtained by mixing the test substance and a solvent was used.
- a 0.5% by weight aqueous solution of sodium carboxymethylcellulose (CMC) was used as the solvent.
- the administration liquid volume was 10 mL / kg.
- For the ingestion of glucose a 50% by weight aqueous glucose solution was used.
- mice C57BL / 6J, male, 7 weeks old, CLEA Japan
- test substance was orally administered to each group as shown in Table 1, 30 minutes later, glucose was orally ingested so as to be 1 g per kg of body weight, and blood was collected from the tail vein with time. Immediately after blood collection, the blood glucose level was measured using a Glutest Neo sensor (manufactured by Sanwa Chemical Laboratory Co., Ltd.).
- FIG. 1 is a graph showing the change in blood glucose level of each group after ingesting glucose.
- XN is xanthohumol
- IXN is isoxanthohumol.
- ⁇ indicates the control group
- ⁇ indicates the xanthohumol 30 mg / kg group
- ⁇ indicates the xanthohumol 60 mg / kg group
- ⁇ indicates the isoxanthohumol 30 mg / kg group
- ⁇ indicates the isoxanthohumol 60 mg / kg group. It is. Compared with the Control group, no significant suppression of blood sugar level increase was observed at any time by a single ingestion of xanthohumol or a single ingestion of isoxanthohumol.
- Example 1 Evaluation of the Effect of Continuous Intake of Isoxanthohumol on the Increase in Blood Sugar Level
- the effect of continuous intake of isoxanthohumol on the increase in blood glucose level after glucose ingestion was examined by the following procedure using a high fat diet-ingested model mouse.
- Table 2 shows the group composition (group name, basal feed, test substance, dose of test substance) in this test.
- "normal feed” used "control feed” (D12450J), and 60 kcal% high fat meal used “ultra high fat feed” (D12492) (all manufactured by Research Diets).
- Xanthohumol and isoxanthohumol used were the samples (purity 95% or more) obtained in Preparation Example 1.
- "Dose” in the table is the dose (mg) of the test substance per day per kg of body weight.
- an administration liquid in which the test substance and a solvent (0.5% by weight CMC aqueous solution) were mixed was used. The dose was 10 mL / kg, and the dose and fluid volume were calculated based on the latest body weight on the test substance administration day.
- For the ingestion of glucose a 50% by weight aqueous glucose solution was used.
- mice C57BL / 6J, male, 7 weeks old, CLEA Japan, Inc.
- quarantine and acclimatizing for 1 week, animals were selected based on changes in body weight from animals with no abnormalities observed in general condition observation did.
- the test substance was orally administered once a day for 3 weeks.
- glucose tolerance test After a 6-hour fast on the last day of the test substance administration, glucose was orally ingested so as to be 1 g per kg of body weight, and blood was collected from the tail vein with time. Immediately after blood collection, the blood glucose level was measured using a Glutest Neo sensor (manufactured by Sanwa Chemical Laboratory Co., Ltd.).
- FIG. 2 is a graph showing the change in blood glucose level after glucose ingestion in each group (#: p ⁇ 0.05 vs. high fat diet group).
- n 8
- FIG. 2 shows a graph showing the area under the blood glucose concentration time curve (AUC) of each group (++: p ⁇ 0.01 vs. normal diet group).
- XN in FIGS. 2 and 3 is xanthohumol, and IXN is isoxanthohumol.
Abstract
Description
〔1〕イソキサントフモールを有効成分とする血糖値上昇抑制用組成物。
〔2〕食後血糖値の上昇を抑制するために使用される上記〔1〕に記載の血糖値上昇抑制用組成物。
〔3〕継続して摂取されるものである上記〔1〕又は〔2〕に記載の血糖値上昇抑制用組成物。
〔4〕食後高血糖に起因する状態又は疾患を予防又は改善するために使用される上記〔1〕~〔3〕のいずれかに記載の血糖値上昇抑制用組成物。
〔5〕飲食品である上記〔1〕~〔4〕のいずれかに記載の血糖値上昇抑制用組成物。
〔6〕飲料である上記〔1〕~〔5〕のいずれかに記載の血糖値上昇抑制用組成物。
〔7〕上記飲料が、茶系飲料、コーヒー飲料、アルコール飲料、ノンアルコールビールテイスト飲料、炭酸飲料、機能性飲料、果実・野菜系飲料、乳性飲料、豆乳飲料又はフレーバーウォーターである上記〔6〕に記載の血糖値上昇抑制用組成物。
〔8〕「食後の血糖値の上昇を抑える」、「食後の血糖値の上昇をおだやかにする」、「血糖値を下げる」、「血糖値が気になる方へ」、「食後の血糖値が気になる方へ」及び「血糖値が上がりやすい体質を改善する」の1又は2以上の表示を付した上記〔1〕~〔7〕のいずれかに記載の血糖値上昇抑制用組成物。
〔9〕イソキサントフモールを対象に摂取させる又は投与する、血糖値上昇抑制方法。
〔10〕イソキサントフモールを継続して対象に摂取させる又は投与する、上記〔9〕に記載の血糖値上昇抑制方法。
〔11〕血糖値上昇を抑制するための、イソキサントフモールの使用。
〔12〕イソキサントフモールを継続して対象に摂取させる又は投与する、上記〔11〕に記載の使用。
また、イソキサントフモールは、例えば100℃の高温条件でも安定であることが報告されている。食品衛生法においては清涼飲料水等の規格基準として殺菌条件が定められているが、例えばpH4.0以上のもの(pH4.6以上で、かつ、水分活性が0.94を超えるものを除く)においては85℃において30分間の加熱が必要である。このような殺菌工程における成分変換を考慮した場合、イソキサントフモールは飲料適正が高いと考えられる。
従って本発明によれば、血糖値上昇抑制作用を示し、熱に安定であり、健康の維持、増進に資する種々の機能性食品、機能性飲料等を提供することが可能となる。また本発明によれば、摂取のタイミングが、食前又は食中に限定されず、血糖値上昇抑制効果、例えば食後血糖値上昇抑制効果を発揮する種々の機能性食品、機能性飲料等を提供することが可能となる。
本発明の血糖値上昇抑制用組成物は、イソキサントフモールのみを有効成分とするものであってよい。
本発明の血糖値上昇抑制用組成物は、飲食品、医薬、医薬部外品、飼料等の形態とすることができる。本発明の血糖値上昇抑制用組成物は、それ自体が、血糖値上昇抑制のための飲食品、医薬、医薬部外品、飼料等であってもよく、これらに配合して使用される素材又は製剤等であってもよい。
本発明の血糖値上昇抑制用組成物は、一例として、剤の形態で提供することができるが、本形態に限定されるものではない。当該剤をそのまま組成物として、又は、当該剤を含む組成物として提供することもできる。本発明の血糖値上昇抑制用組成物は、血糖値上昇抑制剤ということもできる。
本発明の効果を充分に得る観点から、本発明の血糖値上昇抑制用組成物は、好ましくは経口用組成物である。経口用組成物として、飲食品、経口用の医薬、医薬部外品、飼料が挙げられ、好ましくは飲食品又は経口用医薬であり、より好ましくは飲食品である。
本発明の血糖値上昇抑制用組成物を、飲食品、医薬、医薬部外品、飼料等とする場合、その製造方法は特に限定されず、一般的な方法により製造することができる。またイソキサントフモールを含む血糖値上昇抑制用組成物は、例えば、その製造において、ホップ抽出物を配合し、該ホップ抽出物を加熱する工程を行うことによって製造することもできる。
飲料は、ノンアルコール飲料、アルコール飲料のいずれであってもよい。ノンアルコール飲料として、例えば、茶系飲料、コーヒー飲料、ノンアルコールビールテイスト飲料、炭酸飲料、機能性飲料、果実・野菜系飲料、乳性飲料、豆乳飲料、フレーバーウォーター等が挙げられる。
本発明の血糖値上昇抑制用組成物が茶系飲料である場合、紅茶飲料又は無糖茶飲料であることが好ましい。無糖茶飲料として、緑茶飲料、ウーロン茶飲料、麦茶飲料、玄米茶飲料、ハト麦茶飲料、無糖の紅茶飲料等が挙げられる。
本発明の血糖値上昇抑制用組成物がコーヒー飲料である場合、容器詰コーヒー又はリキッドコーヒーであることが好ましい。
本発明の血糖値上昇抑制用組成物がビール系飲料である場合、発泡酒又は第三のビールであることが好ましい。
本発明の血糖値上昇抑制用組成物がビール及びビール系飲料以外のアルコール飲料である場合、焼酎、チューハイ、リキュール、カクテル、スピリッツ、ウイスキーであることが好ましい。
本発明の血糖値上昇抑制用組成物が機能性飲料である場合、スポーツドリンク、エナジードリンク、健康サポート飲料又はパウチゼリー飲料であることが好ましい。
本発明の血糖値上昇抑制用組成物が乳性飲料である場合、牛乳、ドリンクヨーグルト、乳酸菌飲料又は乳類入清涼飲料であることが好ましい。
本発明の血糖値上昇抑制用組成物が豆乳飲料である場合、豆乳又は大豆飲料であることが好ましい。
血糖値上昇抑制用組成物に含まれるイソキサントフモールは、例えば、高速液体クロマトグラフィ(HPLC)やLC-MS/MSにより定量することが可能である。
本発明の血糖値上昇抑制用組成物を摂取させる対象として、血糖値上昇抑制を必要とする又は希望する対象が好ましく、食後血糖値上昇抑制を必要とする又は希望する対象がより好ましい。本発明の血糖値上昇抑制用組成物を摂取させる対象は、糖尿病患者又は糖尿病予備軍に限定されない。本発明の血糖値上昇抑制用組成物は、例えば、血糖値が空腹時126mg/dL以上、又は、経口的ブドウ糖負荷試験(OGTT)2時間値が200mg/dL以上の高血糖症のヒトに対して、治療目的で本発明の血糖値上昇抑制用組成物を使用することができる。また例えば、OGTT2時間値で140~199mg/dLであるヒト(境界型)に対して、糖尿病の悪化予防のために、本発明の血糖値上昇抑制用組成物を使用することもできる。本発明の血糖値上昇抑制用組成物は、高血糖状態の予防又は改善のために、健常者に対して使用することもできる。
本発明の血糖値上昇抑制用組成物は、天然物や飲食品に含まれる安全性が高い成分を有効成分として含む。また、本発明の血糖値上昇抑制用組成物は、継続的に摂取されることによって血糖値上昇抑制効果を発揮する。また、本発明の血糖値上昇抑制用組成物は、摂取するタイミングが食前や食中に限定されず、任意のタイミングで摂取することができる。このため本発明の血糖値上昇抑制用組成物は、例えば、上記の糖尿病患者、糖尿病予備軍、境界型糖尿病患者、食後の高血糖状態が気になるヒト、健常者等が日常的に摂取することにより、血糖値上昇を抑制し、糖尿病の予防又は改善に使用するための健康食品、機能性表示食品等として有用である。
本発明の血糖値上昇抑制用組成物には、例えば、「食後の血糖値の上昇を抑える」、「食後の血糖値の上昇をおだやかにする」、「血糖値を下げる」、「血糖値が気になる方へ」、「食後の血糖値が気になる方へ」及び「血糖値が上がりやすい体質を改善する」の1又は2以上の表示が付されていてもよい。
本発明の一実施態様において、本発明の血糖値上昇抑制用組成物は、上記の機能の表示が付された飲食品であることが好ましい。また上記表示は、上記の機能を得るために用いる旨の表示であってもよい。
血糖値上昇抑制方法は、好ましくは食後血糖値上昇抑制方法である。
本発明は、イソキサントフモールを摂取させる又は投与する、食後高血糖に起因する状態又は疾患を予防又は改善する方法も包含する。イソキサントフモールは、食後血糖値上昇を抑制することにより、食後高血糖に起因する状態又は疾患を予防又は改善するために使用され得る。
上記方法は、治療的な方法であってもよく、非治療的な方法であってもよい。「非治療的」とは、医療行為、すなわち人間の手術、治療又は診断を含まない概念である。
血糖値上昇を抑制するための、イソキサントフモールの使用。
上記使用は、好ましくは、食後血糖値の上昇を抑制するための使用である。
本発明は、食後高血糖に起因する状態又は疾患を予防又は改善するための、イソキサントフモールの使用も包含する。
上記の使用は、好ましくはヒト又は非ヒト哺乳動物、より好ましくはヒトにおける使用である。使用は、治療的な使用であってもよく、非治療的な使用であってもよい。
上記使用においては、血糖値上昇抑制効果を得る観点から、イソキサントフモールを継続して対象に摂取させる又は投与することが好ましい。
また、イソキサントフモールは、血糖値上昇抑制のために使用される飲食品、医薬、医薬部外品、飼料等の製造のために使用することができる。本発明は一態様において、血糖値上昇抑制用組成物を製造するための、イソキサントフモールの使用、も包含する。
キサントフモール及びイソキサントフモールの調製
ホップ抽出物(アサマ化成株式会社製)から、以下の方法でイソキサントフモール及びキサントフモールを単離精製した。すなわち、順相カラムクロマトグラフィー、逆相カラムクロマトグラフィー、分取HPLC精製によりホップ抽出物を原料に、イソキサントフモール及びキサントフモールをそれぞれ精製し、HPLC分析により純度が95%以上であることを確認した。なおHPLC分析においては、カラムとしてDevelosil C30-UG-5(野村化学株式会社)を使用し、検出器の紫外線吸収測定波長は280nm(イソキサントフモール)及び350nm(キサントフモール)とした。
得られたイソキサントフモール及びキサントフモールを、それぞれ標品(いずれも純度95%以上)として以下の実験で使用した。
イソキサントフモールの単回摂取による血糖値上昇抑制作用の評価
イソキサントフモール単回摂取によるグルコース摂取後の血糖値上昇に対する影響を、以下の手順で検討した。
本試験における群構成(群名、被験物質、被験物質の投与量)を表1に示す。キサントフモール及びイソキサントフモールは、調製例1で得られた標品(純度95%以上)を使用した。表中の「投与量」は、体重1kgあたりの被験物質の投与量(mg)である。被験物質の投与には、被験物質と溶媒とを混合した投与液を使用した。溶媒には、0.5重量%カルボキシメチルセルロースナトリウム(CMC)水溶液を使用した。なお投与液量は10mL/kgとした。グルコースの摂取には、50重量%グルコース水溶液を使用した。
マウス(C57BL/6J、雄性、7週齢、日本クレア株式会社)を入荷して1週間検疫及び馴化した後、一般状態観察において異常の認められなかった動物より、6時間の絶食時血糖値を基準に動物を選択した。選択した動物は、馴化期間終了時の体重による層別連続無作為化法により、表1に示す群構成中の各群(1群につきn=9)に割り付けた。
群分け後、各群に表1で表記した通りに被験物質を経口投与し、その30分後にグルコースを体重1kg当たり1gとなるよう経口摂取させ、経時的に尾静脈から採血した。採血後速やかにグルテストNeoセンサー(株式会社三和化学研究所製)を用いて血糖値を測定した。
統計処理はMicrosoft Office Excel 2003を用いて実施した。有意差検定は、Control群と各群との間でDunnett多重比較検定を行い、有意水準は5%とした。
図1に、各群の、グルコース摂取後の血糖値の変動を示すグラフを示す。図1のグラフは、グルコース摂取後(グルコース摂取時:0分)の各時刻における各群の血糖値の平均値±標準誤差(n=9)で示した。図1中のXNはキサントフモール、IXNはイソキサントフモールである。図1中、○はControl群、□はキサントフモール30mg/kg群、■はキサントフモール60mg/kg群、△はイソキサントフモール30mg/kg群、▲はイソキサントフモール60mg/kg群である。
Control群と比較して、いずれの時刻においてもキサントフモール単回摂取又はイソキサントフモール単回摂取による有意な血糖値上昇抑制作用は確認されなかった。
イソキサントフモールの継続摂取による血糖値上昇抑制作用の評価
イソキサントフモール継続摂取によるグルコース摂取後の血糖値上昇に対する影響を、高脂肪食摂取モデルマウスを用いて以下の手順で検討した。
本試験における群構成(群名、基礎飼料、被験物質、被験物質の投与量)を表2に示す。なお表中、普通食は「コントロール飼料」(D12450J)を、60kcal%高脂肪食は「超高脂肪飼料」(D12492)(以上、Research Diets社製)を使用した。キサントフモール及びイソキサントフモールは、調製例1で得られた標品(純度95%以上)を使用した。表中の「投与量」は、体重1kgあたり1日あたりの被験物質の投与量(mg)である。被験物質の投与には、参考例1と同様に、被験物質と溶媒(0.5重量%CMC水溶液)とを混合した投与液を使用した。なお投与液量は10mL/kgとし、被験物質投与日における最新体重を基準として投与量及び液量を算出した。グルコースの摂取には、50重量%グルコース水溶液を使用した。
マウス(C57BL/6J、雄性、7週齢、日本クレア株式会社)を入荷して1週間検疫及び馴化した後、一般状態観察において異常の認められなかった動物より、体重推移を基準に動物を選択した。選択した動物は、馴化期間終了時の体重による層別連続無作為化法により、表2に示す群構成中の各群(1群につきn=8)に割り付け、所定の基礎飼料及び水は自由摂取できる状況で飼育を行った。被験物質は3週間、1日1回経口投与を行った。
被験物質投与最終日に6時間絶食した後、グルコースを体重1kg当たり1gとなるよう経口摂取させ、経時的に尾静脈から採血した。採血後速やかにグルテストNeoセンサー(株式会社三和化学研究所製)を用いて血糖値を測定した。
統計処理はMicrosoft Office Excel 2003を用いて実施した。有意差検定は、普通食群と高脂肪食群との間をStudentのt検定で行い(有意水準1%(p<0.01))、高脂肪食群と、普通食を除く各群との間でDunnett多重比較検定を行った(有意水準5%(p<0.05))。
図2に、各群の、グルコース摂取後の血糖値の変動を示すグラフを示す(#:p<0.05 vs.高脂肪食群)。図2のグラフは、グルコース摂取後(グルコース摂取時:0分)の各時刻における各群の血糖値の平均値±標準誤差(n=8)で示した。図2中、○は普通食群、●は高脂肪食群、□はキサントフモール30mg/kg群、■はキサントフモール60mg/kg群、△はイソキサントフモール30mg/kg群、▲はイソキサントフモール60mg/kg群である。
図2に示さないが、高脂肪食群と普通食群との血糖値の比較では、いずれの時刻においても有意差が認められた(p<0.01)。また図3に、各群の、血中グルコース濃度の時間曲線下面積(AUC)を示すグラフを示す(++:p<0.01 vs.普通食群)。図3に示す結果は、グルコース摂取(0分)から120分後までの血中グルコース濃度の時間曲線下面積(AUC)の平均値±標準誤差(n=8)である。図2及び図3中のXNはキサントフモール、IXNはイソキサントフモールである。
一方、イソキサントフモール30mg/kg群においては、高脂肪食群と比較して血糖値上昇抑制傾向(p<0.10)が認められ、イソキサントフモール60mg/kg群においては有意な血糖値上昇抑制作用が認められた。以上より、イソキサントフモールは血糖値上昇抑制作用を有し、その作用はキサントフモールと比較して強力であることが示された。
Claims (12)
- イソキサントフモールを有効成分とする血糖値上昇抑制用組成物。
- 食後血糖値の上昇を抑制するために使用される請求項1に記載の血糖値上昇抑制用組成物。
- 継続して摂取されるものである請求項1又は2に記載の血糖値上昇抑制用組成物。
- 食後高血糖に起因する状態又は疾患を予防又は改善するために使用される請求項1~3のいずれか一項に記載の血糖値上昇抑制用組成物。
- 飲食品である請求項1~4のいずれか一項に記載の血糖値上昇抑制用組成物。
- 飲料である請求項1~5のいずれか一項に記載の血糖値上昇抑制用組成物。
- 前記飲料が、茶系飲料、コーヒー飲料、アルコール飲料、ノンアルコールビールテイスト飲料、炭酸飲料、機能性飲料、果実・野菜系飲料、乳性飲料、豆乳飲料又はフレーバーウォーターである請求項6に記載の血糖値上昇抑制用組成物。
- 「食後の血糖値の上昇を抑える」、「食後の血糖値の上昇をおだやかにする」、「血糖値を下げる」、「血糖値が気になる方へ」、「食後の血糖値が気になる方へ」及び「血糖値が上がりやすい体質を改善する」の1又は2以上の表示を付した請求項1~7のいずれか一項に記載の血糖値上昇抑制用組成物。
- イソキサントフモールを対象に摂取させる又は投与する、血糖値上昇抑制方法。
- イソキサントフモールを継続して対象に摂取させる又は投与する、請求項9に記載の血糖値上昇抑制方法。
- 血糖値上昇を抑制するための、イソキサントフモールの使用。
- イソキサントフモールを継続して対象に摂取させる又は投与する、請求項11に記載の使用。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2019317043A AU2019317043A1 (en) | 2018-08-10 | 2019-08-05 | Composition and method for suppressing increase in blood glucose level |
| US17/265,612 US20210161857A1 (en) | 2018-08-10 | 2019-08-05 | Composition and method for suppressing increase in blood glucose level |
| EP19846174.1A EP3834629A4 (en) | 2018-08-10 | 2019-08-05 | COMPOSITION AND METHOD OF SUPPRESSING RAISED BLOOD SUGAR LEVELS |
| KR1020217007024A KR20210042949A (ko) | 2018-08-10 | 2019-08-05 | 혈당치 상승 억제용 조성물 및 혈당치 상승 억제 방법 |
| SG11202100264YA SG11202100264YA (en) | 2018-08-10 | 2019-08-05 | Composition and method for suppressing increase in blood glucose level |
| JP2020535753A JPWO2020031952A1 (ja) | 2018-08-10 | 2019-08-05 | 血糖値上昇抑制用組成物及び血糖値上昇抑制方法 |
| CN201980052790.9A CN112566516A (zh) | 2018-08-10 | 2019-08-05 | 血糖值上升抑制用组合物及血糖值上升抑制方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018151540 | 2018-08-10 | ||
| JP2018-151540 | 2018-08-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020031952A1 true WO2020031952A1 (ja) | 2020-02-13 |
Family
ID=69413755
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/030711 WO2020031952A1 (ja) | 2018-08-10 | 2019-08-05 | 血糖値上昇抑制用組成物及び血糖値上昇抑制方法 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20210161857A1 (ja) |
| EP (1) | EP3834629A4 (ja) |
| JP (1) | JPWO2020031952A1 (ja) |
| KR (1) | KR20210042949A (ja) |
| CN (1) | CN112566516A (ja) |
| AU (1) | AU2019317043A1 (ja) |
| SG (1) | SG11202100264YA (ja) |
| TW (1) | TW202017486A (ja) |
| WO (1) | WO2020031952A1 (ja) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001252064A (ja) * | 2000-03-13 | 2001-09-18 | Matsutani Chem Ind Ltd | 食物繊維を含有するビール又は発泡酒 |
| JP2006306800A (ja) | 2005-04-28 | 2006-11-09 | Kirin Brewery Co Ltd | ファルネソイドx受容体活性化剤 |
| US20070218155A1 (en) * | 2004-08-20 | 2007-09-20 | Kuhrts Eric H | Methods and compositions for treating dyslipidaemia |
| JP2009518429A (ja) | 2005-12-09 | 2009-05-07 | メタプロテオミクス,エルエルシー | 代謝症候群および糖尿病の治療のための抗炎症植物産物 |
| JP2010513360A (ja) * | 2006-12-22 | 2010-04-30 | ジョー. バース ウント ソーン ゲーエムベーハー ウントコー. カーゲー | 肝疾患を予防および/または制御するための、キサントフモールまたはイソキサントフモールの活性物質としての使用 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002345433A (ja) * | 2001-05-28 | 2002-12-03 | Takara Bio Inc | 食品、飲料又は調味料の製造方法 |
| JP2006028143A (ja) * | 2004-07-21 | 2006-02-02 | Shiseido Co Ltd | Mitf遺伝子発現促進物質 |
| AU2012204133B2 (en) * | 2005-08-09 | 2013-09-19 | Metaproteomics, Llc | Protein kinase modulation by hops and acacia products |
| KR20140035331A (ko) * | 2011-01-07 | 2014-03-21 | 엘셀릭스 테라퓨틱스 인코포레이티드 | 화학감각 수용체 리간드-기반 요법 |
-
2019
- 2019-08-05 WO PCT/JP2019/030711 patent/WO2020031952A1/ja unknown
- 2019-08-05 AU AU2019317043A patent/AU2019317043A1/en not_active Abandoned
- 2019-08-05 JP JP2020535753A patent/JPWO2020031952A1/ja active Pending
- 2019-08-05 KR KR1020217007024A patent/KR20210042949A/ko unknown
- 2019-08-05 CN CN201980052790.9A patent/CN112566516A/zh active Pending
- 2019-08-05 US US17/265,612 patent/US20210161857A1/en not_active Abandoned
- 2019-08-05 EP EP19846174.1A patent/EP3834629A4/en not_active Withdrawn
- 2019-08-05 SG SG11202100264YA patent/SG11202100264YA/en unknown
- 2019-08-08 TW TW108128218A patent/TW202017486A/zh unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001252064A (ja) * | 2000-03-13 | 2001-09-18 | Matsutani Chem Ind Ltd | 食物繊維を含有するビール又は発泡酒 |
| US20070218155A1 (en) * | 2004-08-20 | 2007-09-20 | Kuhrts Eric H | Methods and compositions for treating dyslipidaemia |
| JP2006306800A (ja) | 2005-04-28 | 2006-11-09 | Kirin Brewery Co Ltd | ファルネソイドx受容体活性化剤 |
| JP2009518429A (ja) | 2005-12-09 | 2009-05-07 | メタプロテオミクス,エルエルシー | 代謝症候群および糖尿病の治療のための抗炎症植物産物 |
| JP2010513360A (ja) * | 2006-12-22 | 2010-04-30 | ジョー. バース ウント ソーン ゲーエムベーハー ウントコー. カーゲー | 肝疾患を予防および/または制御するための、キサントフモールまたはイソキサントフモールの活性物質としての使用 |
Non-Patent Citations (9)
| Title |
|---|
| ANDO, Y ET AL.: "Tumor Necrosis Factor (TNF) - a -induced Repression of GKAP42 Protein Levels through cGMP-dependent Kinase (cGK)-Ialpha Causes Insulin Resistance in 3T3-L1 Adipocytes", THE JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 290, no. 9, 27 February 2015 (2015-02-27), pages 5881 - 5892, XP055683583, ISSN: 1083-351X * |
| ARCH BIOCHEM BIOPHYS, vol. 599, 2016, pages 22 - 30 |
| FREE RADICAL BIO MED, vol. 89, 2015, pages 486 - 497 |
| J NUTR BIOCHEM, vol. 45, 2017, pages 39 - 47 |
| LANCET, vol. 354, 1999, pages 617 - 621 |
| NEGRAO, R ET AL.: "Isoxanthohumol modulates angiogenesis and inflammation via vascular endothelial growth factor receptor, tumor necrosis factor alpha and nuclear factor kappa B pathways", BIOFACTORS, vol. 39, no. 6, November 2013 (2013-11-01), pages 608 - 622, XP055683580, ISSN: 1872-8081 * |
| RAMIREZ, AG ET AL.: "Highly isoxanthohumol enriched hop extract obtained by pressurized hot water extraction (PHWE", CHEMICAL AND FUNCTIONAL CHARACTERIZATION INNOVATIVE FOOD SCIENCE AND EMERGING TECHNOLOGIES, vol. 16, October 2012 (2012-10-01), pages 54 - 60, XP055683579, ISSN: 1466-8564 * |
| See also references of EP3834629A4 |
| YANG, JY ET AL.: "Effect of xanthohumol and isoxanthohumol on 3T3-L1 cell apoptosis and adipogenesis", APOPTOSIS, vol. 12, 2007, pages 1953 - 1963, XP019523444, ISSN: 1573-675X, DOI: 10.1007/s10495-007-0130-4 * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3834629A4 (en) | 2022-04-27 |
| JPWO2020031952A1 (ja) | 2021-08-26 |
| KR20210042949A (ko) | 2021-04-20 |
| AU2019317043A1 (en) | 2021-02-04 |
| EP3834629A1 (en) | 2021-06-16 |
| SG11202100264YA (en) | 2021-02-25 |
| US20210161857A1 (en) | 2021-06-03 |
| TW202017486A (zh) | 2020-05-16 |
| CN112566516A (zh) | 2021-03-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP3756438B2 (ja) | 容器詰飲料 | |
| US20030161903A1 (en) | Carbohydrate Aborption Inhibitor and Method for Manufacturing the Same | |
| EP2409696A1 (en) | Agent for promoting energy consumption | |
| Butacnum et al. | Black tea consumption improves postprandial glycemic control in normal and pre-diabetic subjects: A randomized, double-blind, placebo-controlled crossover study | |
| WO2018117041A1 (ja) | イソキサントフモールを含む脂質代謝促進用組成物 | |
| EP3302453B1 (en) | Compositions comprising cinnamaldehyde and zinc and methods of using such compositions | |
| WO2020031961A1 (ja) | 尿酸排出促進用組成物、urat1阻害用組成物及び血中尿酸値低下用組成物 | |
| WO2020031957A1 (ja) | 腸内環境改善用組成物及び腸内細菌叢改善方法 | |
| CN106028837B (zh) | 使用肉桂醛和锌进行体重管理的方法和组合物 | |
| JP2006117687A (ja) | 体脂肪の燃焼のための健康食品 | |
| WO2020031952A1 (ja) | 血糖値上昇抑制用組成物及び血糖値上昇抑制方法 | |
| JP3756510B2 (ja) | 体脂肪燃焼のための容器詰飲料。 | |
| JP4611775B2 (ja) | 血圧降下剤 | |
| JP7352570B2 (ja) | 血流改善用組成物及び血管内皮機能改善用組成物 | |
| WO2018101231A1 (ja) | 交感神経活性化用組成物 | |
| WO2021131568A1 (ja) | 身体活動量低下の抑制又は改善用組成物 | |
| JP5048258B2 (ja) | リバウンド抑制剤 | |
| WO2021131570A1 (ja) | 認知機能の低下抑制又は改善用組成物 | |
| Vishwakarma et al. | Litchi: Queen of Fruit | |
| TWI424819B (zh) | 裝於容器內之含異白胺酸的食品 | |
| KR20190136333A (ko) | 들깨박 추출물을 포함하는 장기능 및 변비 개선용 조성물 | |
| JP2006077026A (ja) | 体脂肪燃焼のための容器詰飲料。 | |
| KR20170087064A (ko) | 아위버섯 물 추출물을 유효성분으로 함유하는 대사성질환의 예방 및 치료용 약학적 조성물 또는 건강기능성식품 | |
| KR20180079023A (ko) | 치아씨드 및 선인장열매 혼합물을 유효성분으로 포함하는 대사증후군 예방, 치료 또는 개선용 조성물 | |
| JP2017218410A (ja) | 糖尿病の予防または治療用組成物およびインスリン抵抗性の改善用組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19846174 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020535753 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019317043 Country of ref document: AU Date of ref document: 20190805 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20217007024 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019846174 Country of ref document: EP Effective date: 20210310 |