WO2018084012A1 - 反応性ホットメルトシリコーン充填容器および反応性ホットメルトシリコーンの製造方法 - Google Patents
反応性ホットメルトシリコーン充填容器および反応性ホットメルトシリコーンの製造方法 Download PDFInfo
- Publication number
- WO2018084012A1 WO2018084012A1 PCT/JP2017/038184 JP2017038184W WO2018084012A1 WO 2018084012 A1 WO2018084012 A1 WO 2018084012A1 JP 2017038184 W JP2017038184 W JP 2017038184W WO 2018084012 A1 WO2018084012 A1 WO 2018084012A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- reactive hot
- container
- organopolysiloxane
- melt silicone
- group
- Prior art date
Links
- 229920001296 polysiloxane Polymers 0.000 title claims abstract description 155
- 239000012943 hotmelt Substances 0.000 title claims abstract description 75
- 238000004519 manufacturing process Methods 0.000 title claims description 12
- 238000000034 method Methods 0.000 title claims description 5
- 239000000203 mixture Substances 0.000 claims abstract description 55
- 125000003342 alkenyl group Chemical group 0.000 claims abstract description 25
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims abstract description 25
- 238000006459 hydrosilylation reaction Methods 0.000 claims abstract description 16
- 239000000155 melt Substances 0.000 claims abstract description 9
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 22
- 229910052710 silicon Inorganic materials 0.000 claims description 22
- 239000010703 silicon Substances 0.000 claims description 20
- 239000003054 catalyst Substances 0.000 claims description 13
- 238000010438 heat treatment Methods 0.000 claims description 11
- 150000001451 organic peroxides Chemical class 0.000 claims description 7
- 230000009969 flowable effect Effects 0.000 claims description 6
- 230000003197 catalytic effect Effects 0.000 claims description 5
- 238000006243 chemical reaction Methods 0.000 claims description 5
- 239000002683 reaction inhibitor Substances 0.000 claims description 5
- 239000012530 fluid Substances 0.000 claims 1
- 238000004132 cross linking Methods 0.000 abstract description 3
- 239000007809 chemical reaction catalyst Substances 0.000 abstract 1
- -1 siloxane unit Chemical group 0.000 description 60
- 229910004283 SiO 4 Inorganic materials 0.000 description 9
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 9
- 239000003795 chemical substances by application Substances 0.000 description 8
- 239000000047 product Substances 0.000 description 8
- 125000003118 aryl group Chemical group 0.000 description 7
- 125000004432 carbon atom Chemical group C* 0.000 description 7
- 150000003961 organosilicon compounds Chemical class 0.000 description 6
- 125000003545 alkoxy group Chemical group 0.000 description 5
- 125000005388 dimethylhydrogensiloxy group Chemical group 0.000 description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 5
- 230000005764 inhibitory process Effects 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 239000002184 metal Substances 0.000 description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 5
- VMAWODUEPLAHOE-UHFFFAOYSA-N 2,4,6,8-tetrakis(ethenyl)-2,4,6,8-tetramethyl-1,3,5,7,2,4,6,8-tetraoxatetrasilocane Chemical compound C=C[Si]1(C)O[Si](C)(C=C)O[Si](C)(C=C)O[Si](C)(C=C)O1 VMAWODUEPLAHOE-UHFFFAOYSA-N 0.000 description 4
- 239000004831 Hot glue Substances 0.000 description 4
- 229910004298 SiO 2 Inorganic materials 0.000 description 4
- 125000000217 alkyl group Chemical group 0.000 description 4
- 229910052697 platinum Inorganic materials 0.000 description 4
- DMWVYCCGCQPJEA-UHFFFAOYSA-N 2,5-bis(tert-butylperoxy)-2,5-dimethylhexane Chemical compound CC(C)(C)OOC(C)(C)CCC(C)(C)OOC(C)(C)C DMWVYCCGCQPJEA-UHFFFAOYSA-N 0.000 description 3
- 125000003710 aryl alkyl group Chemical group 0.000 description 3
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 3
- 239000004205 dimethyl polysiloxane Substances 0.000 description 3
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 3
- 150000002430 hydrocarbons Chemical group 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- 125000000962 organic group Chemical group 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 239000004033 plastic Substances 0.000 description 3
- 229920003023 plastic Polymers 0.000 description 3
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 3
- 229920005989 resin Polymers 0.000 description 3
- 239000011347 resin Substances 0.000 description 3
- 239000004065 semiconductor Substances 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 125000003944 tolyl group Chemical group 0.000 description 3
- UBRWPVTUQDJKCC-UHFFFAOYSA-N 1,3-bis(2-tert-butylperoxypropan-2-yl)benzene Chemical compound CC(C)(C)OOC(C)(C)C1=CC=CC(C(C)(C)OOC(C)(C)C)=C1 UBRWPVTUQDJKCC-UHFFFAOYSA-N 0.000 description 2
- XMNIXWIUMCBBBL-UHFFFAOYSA-N 2-(2-phenylpropan-2-ylperoxy)propan-2-ylbenzene Chemical compound C=1C=CC=CC=1C(C)(C)OOC(C)(C)C1=CC=CC=C1 XMNIXWIUMCBBBL-UHFFFAOYSA-N 0.000 description 2
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 description 2
- 239000012933 diacyl peroxide Substances 0.000 description 2
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 125000003700 epoxy group Chemical group 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 125000006038 hexenyl group Chemical group 0.000 description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- CRJSCSRODDRNDN-UHFFFAOYSA-N methyl-tris(2-methylbut-3-yn-2-yloxy)silane Chemical compound C#CC(C)(C)O[Si](C)(OC(C)(C)C#C)OC(C)(C)C#C CRJSCSRODDRNDN-UHFFFAOYSA-N 0.000 description 2
- 125000005375 organosiloxane group Chemical group 0.000 description 2
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 2
- 229920001843 polymethylhydrosiloxane Polymers 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 238000007789 sealing Methods 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 229920001187 thermosetting polymer Polymers 0.000 description 2
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 2
- 125000005023 xylyl group Chemical group 0.000 description 2
- HGXJDMCMYLEZMJ-UHFFFAOYSA-N (2-methylpropan-2-yl)oxy 2,2-dimethylpropaneperoxoate Chemical compound CC(C)(C)OOOC(=O)C(C)(C)C HGXJDMCMYLEZMJ-UHFFFAOYSA-N 0.000 description 1
- KDGNCLDCOVTOCS-UHFFFAOYSA-N (2-methylpropan-2-yl)oxy propan-2-yl carbonate Chemical compound CC(C)OC(=O)OOC(C)(C)C KDGNCLDCOVTOCS-UHFFFAOYSA-N 0.000 description 1
- NOBYOEQUFMGXBP-UHFFFAOYSA-N (4-tert-butylcyclohexyl) (4-tert-butylcyclohexyl)oxycarbonyloxy carbonate Chemical compound C1CC(C(C)(C)C)CCC1OC(=O)OOC(=O)OC1CCC(C(C)(C)C)CC1 NOBYOEQUFMGXBP-UHFFFAOYSA-N 0.000 description 1
- HMVBQEAJQVQOTI-SOFGYWHQSA-N (e)-3,5-dimethylhex-3-en-1-yne Chemical compound CC(C)\C=C(/C)C#C HMVBQEAJQVQOTI-SOFGYWHQSA-N 0.000 description 1
- GRGVQLWQXHFRHO-AATRIKPKSA-N (e)-3-methylpent-3-en-1-yne Chemical compound C\C=C(/C)C#C GRGVQLWQXHFRHO-AATRIKPKSA-N 0.000 description 1
- BEQKKZICTDFVMG-UHFFFAOYSA-N 1,2,3,4,6-pentaoxepane-5,7-dione Chemical compound O=C1OOOOC(=O)O1 BEQKKZICTDFVMG-UHFFFAOYSA-N 0.000 description 1
- QYLFHLNFIHBCPR-UHFFFAOYSA-N 1-ethynylcyclohexan-1-ol Chemical compound C#CC1(O)CCCCC1 QYLFHLNFIHBCPR-UHFFFAOYSA-N 0.000 description 1
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 description 1
- CRJIYMRJTJWVLU-UHFFFAOYSA-N 2,4,4-trimethylpentan-2-yl 3-(5,5-dimethylhexyl)dioxirane-3-carboxylate Chemical compound CC(C)(C)CCCCC1(C(=O)OC(C)(C)CC(C)(C)C)OO1 CRJIYMRJTJWVLU-UHFFFAOYSA-N 0.000 description 1
- DPGYCJUCJYUHTM-UHFFFAOYSA-N 2,4,4-trimethylpentan-2-yloxy 2-ethylhexaneperoxoate Chemical compound CCCCC(CC)C(=O)OOOC(C)(C)CC(C)(C)C DPGYCJUCJYUHTM-UHFFFAOYSA-N 0.000 description 1
- ODBCKCWTWALFKM-UHFFFAOYSA-N 2,5-bis(tert-butylperoxy)-2,5-dimethylhex-3-yne Chemical compound CC(C)(C)OOC(C)(C)C#CC(C)(C)OOC(C)(C)C ODBCKCWTWALFKM-UHFFFAOYSA-N 0.000 description 1
- ZACVGCNKGYYQHA-UHFFFAOYSA-N 2-ethylhexoxycarbonyloxy 2-ethylhexyl carbonate Chemical compound CCCCC(CC)COC(=O)OOC(=O)OCC(CC)CCCC ZACVGCNKGYYQHA-UHFFFAOYSA-N 0.000 description 1
- YAQDPWONDFRAHF-UHFFFAOYSA-N 2-methyl-2-(2-methylpentan-2-ylperoxy)pentane Chemical compound CCCC(C)(C)OOC(C)(C)CCC YAQDPWONDFRAHF-UHFFFAOYSA-N 0.000 description 1
- CEBKHWWANWSNTI-UHFFFAOYSA-N 2-methylbut-3-yn-2-ol Chemical compound CC(C)(O)C#C CEBKHWWANWSNTI-UHFFFAOYSA-N 0.000 description 1
- KHTWSJRZFJREDU-UHFFFAOYSA-N 2-methylbutan-2-yl 2-ethyl-2-hydroperoxyhexanoate Chemical compound CCCCC(CC)(OO)C(=O)OC(C)(C)CC KHTWSJRZFJREDU-UHFFFAOYSA-N 0.000 description 1
- RTEZVHMDMFEURJ-UHFFFAOYSA-N 2-methylpentan-2-yl 2,2-dimethylpropaneperoxoate Chemical compound CCCC(C)(C)OOC(=O)C(C)(C)C RTEZVHMDMFEURJ-UHFFFAOYSA-N 0.000 description 1
- NUIZZJWNNGJSGL-UHFFFAOYSA-N 2-phenylpropan-2-yl 2,2-dimethyloctaneperoxoate Chemical compound CCCCCCC(C)(C)C(=O)OOC(C)(C)c1ccccc1 NUIZZJWNNGJSGL-UHFFFAOYSA-N 0.000 description 1
- BIISIZOQPWZPPS-UHFFFAOYSA-N 2-tert-butylperoxypropan-2-ylbenzene Chemical compound CC(C)(C)OOC(C)(C)C1=CC=CC=C1 BIISIZOQPWZPPS-UHFFFAOYSA-N 0.000 description 1
- VKERWIBXKLNXCY-UHFFFAOYSA-N 3,5,5-trimethyl-2-(2-methylbutan-2-ylperoxy)hexanoic acid Chemical compound CCC(C)(C)OOC(C(O)=O)C(C)CC(C)(C)C VKERWIBXKLNXCY-UHFFFAOYSA-N 0.000 description 1
- NECRQCBKTGZNMH-UHFFFAOYSA-N 3,5-dimethylhex-1-yn-3-ol Chemical compound CC(C)CC(C)(O)C#C NECRQCBKTGZNMH-UHFFFAOYSA-N 0.000 description 1
- XYFRHHAYSXIKGH-UHFFFAOYSA-N 3-(5-methoxy-2-methoxycarbonyl-1h-indol-3-yl)prop-2-enoic acid Chemical compound C1=C(OC)C=C2C(C=CC(O)=O)=C(C(=O)OC)NC2=C1 XYFRHHAYSXIKGH-UHFFFAOYSA-N 0.000 description 1
- CARSMBZECAABMO-UHFFFAOYSA-N 3-chloro-2,6-dimethylbenzoic acid Chemical compound CC1=CC=C(Cl)C(C)=C1C(O)=O CARSMBZECAABMO-UHFFFAOYSA-N 0.000 description 1
- XDLMVUHYZWKMMD-UHFFFAOYSA-N 3-trimethoxysilylpropyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCOC(=O)C(C)=C XDLMVUHYZWKMMD-UHFFFAOYSA-N 0.000 description 1
- 239000004342 Benzoyl peroxide Substances 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- YIVJZNGAASQVEM-UHFFFAOYSA-N Lauroyl peroxide Chemical compound CCCCCCCCCCCC(=O)OOC(=O)CCCCCCCCCCC YIVJZNGAASQVEM-UHFFFAOYSA-N 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- AEHGADJCYDXLCC-UHFFFAOYSA-N [3-tert-butyl-2-(3-tert-butyl-4,4-dimethyl-2-phenylpentan-2-yl)peroxy-4,4-dimethylpentan-2-yl]benzene Chemical compound C=1C=CC=CC=1C(C)(C(C(C)(C)C)C(C)(C)C)OOC(C)(C(C(C)(C)C)C(C)(C)C)C1=CC=CC=C1 AEHGADJCYDXLCC-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- WNLRTRBMVRJNCN-UHFFFAOYSA-L adipate(2-) Chemical compound [O-]C(=O)CCCCC([O-])=O WNLRTRBMVRJNCN-UHFFFAOYSA-L 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- QRUDEWIWKLJBPS-UHFFFAOYSA-N benzotriazole Chemical compound C1=CC=C2N[N][N]C2=C1 QRUDEWIWKLJBPS-UHFFFAOYSA-N 0.000 description 1
- 239000012964 benzotriazole Substances 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000006482 condensation reaction Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- DDJSWKLBKSLAAZ-UHFFFAOYSA-N cyclotetrasiloxane Chemical compound O1[SiH2]O[SiH2]O[SiH2]O[SiH2]1 DDJSWKLBKSLAAZ-UHFFFAOYSA-N 0.000 description 1
- XJOBOFWTZOKMOH-UHFFFAOYSA-N decanoyl decaneperoxoate Chemical compound CCCCCCCCCC(=O)OOC(=O)CCCCCCCCC XJOBOFWTZOKMOH-UHFFFAOYSA-N 0.000 description 1
- 229940057404 di-(4-tert-butylcyclohexyl)peroxydicarbonate Drugs 0.000 description 1
- RQCQBDUYEHRNPP-UHFFFAOYSA-N dimethyl-bis(2-methylbut-3-yn-2-yloxy)silane Chemical compound C#CC(C)(C)O[Si](C)(C)OC(C)(C)C#C RQCQBDUYEHRNPP-UHFFFAOYSA-N 0.000 description 1
- YCWQBZCTYWZZAX-UHFFFAOYSA-N ditert-butyl 7,8-dioxabicyclo[4.2.0]octane-3,6-dicarboxylate Chemical compound C1C(C(=O)OC(C)(C)C)CCC2(C(=O)OC(C)(C)C)OOC21 YCWQBZCTYWZZAX-UHFFFAOYSA-N 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- BITPLIXHRASDQB-UHFFFAOYSA-N ethenyl-[ethenyl(dimethyl)silyl]oxy-dimethylsilane Chemical compound C=C[Si](C)(C)O[Si](C)(C)C=C BITPLIXHRASDQB-UHFFFAOYSA-N 0.000 description 1
- RCNRJBWHLARWRP-UHFFFAOYSA-N ethenyl-[ethenyl(dimethyl)silyl]oxy-dimethylsilane;platinum Chemical compound [Pt].C=C[Si](C)(C)O[Si](C)(C)C=C RCNRJBWHLARWRP-UHFFFAOYSA-N 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 239000003063 flame retardant Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 125000005417 glycidoxyalkyl group Chemical group 0.000 description 1
- QWVBGCWRHHXMRM-UHFFFAOYSA-N hexadecoxycarbonyloxy hexadecyl carbonate Chemical compound CCCCCCCCCCCCCCCCOC(=O)OOC(=O)OCCCCCCCCCCCCCCCC QWVBGCWRHHXMRM-UHFFFAOYSA-N 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- NYMPGSQKHIOWIO-UHFFFAOYSA-N hydroxy(diphenyl)silicon Chemical class C=1C=CC=CC=1[Si](O)C1=CC=CC=C1 NYMPGSQKHIOWIO-UHFFFAOYSA-N 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000011256 inorganic filler Substances 0.000 description 1
- 229910003475 inorganic filler Inorganic materials 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000002985 plastic film Substances 0.000 description 1
- 229920006255 plastic film Polymers 0.000 description 1
- 229920000193 polymethacrylate Polymers 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- RGBXDEHYFWDBKD-UHFFFAOYSA-N propan-2-yl propan-2-yloxy carbonate Chemical compound CC(C)OOC(=O)OC(C)C RGBXDEHYFWDBKD-UHFFFAOYSA-N 0.000 description 1
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 239000010948 rhodium Substances 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 150000004756 silanes Chemical class 0.000 description 1
- SCPYDCQAZCOKTP-UHFFFAOYSA-N silanol Chemical group [SiH3]O SCPYDCQAZCOKTP-UHFFFAOYSA-N 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- WYKYCHHWIJXDAO-UHFFFAOYSA-N tert-butyl 2-ethylhexaneperoxoate Chemical compound CCCCC(CC)C(=O)OOC(C)(C)C WYKYCHHWIJXDAO-UHFFFAOYSA-N 0.000 description 1
- PFBLRDXPNUJYJM-UHFFFAOYSA-N tert-butyl 2-methylpropaneperoxoate Chemical compound CC(C)C(=O)OOC(C)(C)C PFBLRDXPNUJYJM-UHFFFAOYSA-N 0.000 description 1
- NMOALOSNPWTWRH-UHFFFAOYSA-N tert-butyl 7,7-dimethyloctaneperoxoate Chemical compound CC(C)(C)CCCCCC(=O)OOC(C)(C)C NMOALOSNPWTWRH-UHFFFAOYSA-N 0.000 description 1
- GJBRNHKUVLOCEB-UHFFFAOYSA-N tert-butyl benzenecarboperoxoate Chemical compound CC(C)(C)OOC(=O)C1=CC=CC=C1 GJBRNHKUVLOCEB-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- CSKKAINPUYTTRW-UHFFFAOYSA-N tetradecoxycarbonyloxy tetradecyl carbonate Chemical compound CCCCCCCCCCCCCCOC(=O)OOC(=O)OCCCCCCCCCCCCCC CSKKAINPUYTTRW-UHFFFAOYSA-N 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- DQZNLOXENNXVAD-UHFFFAOYSA-N trimethoxy-[2-(7-oxabicyclo[4.1.0]heptan-4-yl)ethyl]silane Chemical compound C1C(CC[Si](OC)(OC)OC)CCC2OC21 DQZNLOXENNXVAD-UHFFFAOYSA-N 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J183/00—Adhesives based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon, with or without sulfur, nitrogen, oxygen, or carbon only; Adhesives based on derivatives of such polymers
- C09J183/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L83/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers
- C08L83/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/34—Filling pastes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D183/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon, with or without sulfur, nitrogen, oxygen, or carbon only; Coating compositions based on derivatives of such polymers
- C09D183/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J11/00—Features of adhesives not provided for in group C09J9/00, e.g. additives
- C09J11/02—Non-macromolecular additives
- C09J11/06—Non-macromolecular additives organic
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/29—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the material, e.g. carbon
- H01L23/293—Organic, e.g. plastic
- H01L23/296—Organo-silicon compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/12—Polysiloxanes containing silicon bound to hydrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/20—Polysiloxanes containing silicon bound to unsaturated aliphatic groups
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2301/00—Additional features of adhesives in the form of films or foils
- C09J2301/30—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier
- C09J2301/304—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier the adhesive being heat-activatable, i.e. not tacky at temperatures inferior to 30°C
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J2301/00—Additional features of adhesives in the form of films or foils
- C09J2301/30—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier
- C09J2301/312—Additional features of adhesives in the form of films or foils characterized by the chemical, physicochemical or physical properties of the adhesive or the carrier parameters being the characterizing feature
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L33/00—Semiconductor devices having potential barriers specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof
- H01L33/48—Semiconductor devices having potential barriers specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof characterised by the semiconductor body packages
- H01L33/52—Encapsulations
- H01L33/56—Materials, e.g. epoxy or silicone resin
Definitions
- the present invention relates to a reactive hot melt silicone filling container and a method for producing reactive hot melt silicone.
- Patent Document 1 discloses an organopolysiloxane having at least two alkenylsilyl groups in one molecule, an organopolysiloxane having at least two hydrosilyl groups in one molecule, a catalyst for hydrosilylation reaction, and reaction inhibition.
- a reactive hot melt silicone obtained by semi-curing a crosslinkable silicone composition containing an agent into a sheet has been proposed.
- this sheet-like reactive hot-melt silicone easily entrains air during sealing or coating because of the gap between the LED element and the substrate, and the appearance or reliability of the LED sealed or coated is deteriorated. There are issues such as.
- the object of the present invention is to provide a reactive hot melt silicone-filled container capable of taking out a reactive hot melt silicone excellent in gap fill property by heating, and volatilization and air inhibition of components during heating are suppressed.
- Another object of the present invention is to provide a method for producing a reactive hot melt silicone having excellent gap fill properties.
- Reactive hot melt silicone filled container of the present invention (A) an alkenyl group-bonded organopolysiloxane having at least a branched organopolysiloxane having an alkenyl group and a softening point of 50 ° C. or higher, (B) Organopolysiloxane having at least two silicon atom-bonded hydrogen atoms in one molecule ⁇ the amount of silicon atom-bonded hydrogen atoms in this component is 0.01 to 10 mol) and (C) a crosslinkable silicone composition containing a catalytic amount of a catalyst for hydrosilylation reaction is filled into a container, and the composition is crosslinked in a B-stage by heating the container. It is characterized by forming a reactive hot melt silicone which is non-flowable at 25 ° C. and has a melt viscosity at 120 ° C. of 5,000 Pa ⁇ s or less.
- the crosslinkable silicone composition further comprises 0.0001 to 5 parts by mass of (D) reaction inhibitor and / or 0.01 to 10 parts per 100 parts by mass of components (A) to (C). It is preferable to contain (E) an organic peroxide in parts by mass.
- the container is preferably a cartridge, a flexible container, a pail can, or a drum can.
- the method for producing a reactive hot melt silicone of the present invention is a method for producing a reactive hot melt silicone that is non-flowable at 25 ° C. and has a melt viscosity at 120 ° C. of 5,000 Pa ⁇ s or less, (A) an alkenyl group-bonded organopolysiloxane having at least a branched organopolysiloxane having an alkenyl group and a softening point of 50 ° C.
- the crosslinkable silicone composition further comprises 0.0001 to 5 parts by mass of (D) reaction inhibitor and / or 0.01 to 10 parts per 100 parts by mass of components (A) to (C). It is preferable to contain (E) an organic peroxide in parts by mass.
- the container is preferably a cartridge, a flexible container, a pail can, or a drum can.
- the reactive hot melt silicone-filled container of the present invention is characterized in that the reactive hot melt silicone excellent in gap fill property can be taken out by heating, and the method for producing the reactive hot melt silicone of the present invention includes: There is a feature that the reactive hot-melt silicone can be efficiently produced, which suppresses volatilization of components and air inhibition during heating, has fluidity by heating, and has excellent gap fill properties.
- Reactive hot melt silicone filled container of the present invention (A) an alkenyl group-bonded organopolysiloxane having at least a branched organopolysiloxane having an alkenyl group and a softening point of 50 ° C. or higher, (B) Organopolysiloxane having at least two silicon atom-bonded hydrogen atoms in one molecule ⁇ the amount of silicon atom-bonded hydrogen atoms in this component is 0.01 to 10 mol) and (C) a crosslinkable silicone composition containing a catalytic amount of a catalyst for hydrosilylation reaction is filled into a container, and the composition is crosslinked in a B-stage by heating the container. It is characterized by that.
- B-stage refers to the state of B-stage (cured intermediate of thermosetting resin) as defined in JIS K 6800, and incompletely cures the crosslinkable silicone composition. Although it is swollen by the solvent, it does not dissolve completely, and the “reactive hot melt” is a state in which the fluidity is lost at room temperature (25 ° C.), but at a high temperature (eg, 120 ° C. ) Is melted again when heated and then cured.
- the component (A) is an alkenyl group-bonded organopolysiloxane containing at least a branched organopolysiloxane having an alkenyl group and a softening point of 50 ° C. or higher.
- the branched organopolysiloxane having a softening point has a siloxane unit represented by the formula: R 1 SiO 3/2 and / or a siloxane unit represented by the formula: SiO 4/2 in the molecular chain.
- an organopolysiloxane which may have a siloxane unit represented by the formula: R 1 3 SiO 1/2 and / or a siloxane unit represented by the formula: R 1 2 SiO 2/2 .
- R 1 is the same or different monovalent hydrocarbon group, and examples thereof include 1 to 12 carbon atoms such as a methyl group, an ethyl group, a propyl group, a butyl group, a pentyl group, a hexyl group, a cyclopentyl group, and a cyclohexyl group.
- An alkyl group having 2 to 12 carbon atoms such as a vinyl group, an allyl group, a butenyl group, a pentenyl group or a hexenyl group; an aryl group having 6 to 12 carbon atoms such as a phenyl group, a tolyl group or a xylyl group; a benzyl group And an aralkyl group having 7 to 12 carbon atoms such as phenethyl group; a group in which part or all of the hydrogen atoms of these groups are substituted with a halogen atom such as a chlorine atom or a fluorine atom.
- at least one R 1 in one molecule is the alkenyl group.
- Such a branched organopolysiloxane has a softening point of 50 ° C. or higher.
- the softening point means, for example, a temperature measured by a softening point test method using a ring-and-ball method of a hot melt adhesive specified in JIS K 6863-1994 “Testing method for softening point of hot melt adhesive”.
- an organopolysiloxane having such a softening point an organopolysiloxane comprising a siloxane unit represented by the formula: R 1 SiO 3/2 , wherein 50 to 80 mol% of all R 1 in one molecule is an aryl group.
- Siloxane consisting of a siloxane unit represented by the formula: R 1 SiO 3/2 and a siloxane unit represented by the formula: R 1 3 SiO 1/2 , wherein 50 to 80 mol% of all R 1 in one molecule is aryl Organopolysiloxane as a group, comprising a siloxane unit represented by the formula: R 1 SiO 3/2 and a siloxane unit represented by the formula: R 1 2 SiO 2/2 , and 50 to 50 of all R 1 in one molecule
- organopolysiloxane 50-80 mol% of the total R 1 in one molecule is an aryl group, wherein: the siloxane units, organopol
- the component (A) may be only the branched organopolysiloxane, or a mixture of the branched organopolysiloxane and the linear organopolysiloxane.
- This linear organopolysiloxane is a component for adjusting the hardness of a cured product obtained by curing a reactive hot-melt silicone or imparting its flexibility.
- R 2 3 SiO (R 2 2 SiO) n SiR 2 3 It is represented by
- R 2 is the same or different monovalent hydrocarbon group, and the same group as R 1 is exemplified. However, at least two R 2 in one molecule is the alkenyl group.
- n is an integer in the range of 1 to 1,000, an integer in the range of 1 to 500, an integer in the range of 5 to 500, or an integer in the range of 10 to 500. preferable.
- n is not less than the lower limit of the above range, the resulting cured product has good mechanical strength.
- n is not more than the upper limit of the above range, the resulting crosslinkable silicone composition has good fillability. Because it becomes.
- the ratio of the branched organopolysiloxane to the linear organopolysiloxane is not limited, and the linear organopolysiloxane is in the range of 0 to 80% by mass in the component (A).
- Component (B) is an organopolysiloxane having at least two silicon-bonded hydrogen atoms in one molecule.
- the silicon-bonded organic group in the component (B) is an alkyl group having 1 to 12 carbon atoms such as a methyl group, an ethyl group, a propyl group, a butyl group, a pentyl group, a hexyl group, a cyclopentyl group, or a cyclohexyl group;
- Aryl groups having 6 to 12 carbon atoms such as benzyl, tolyl and xylyl groups; aralkyl groups having 7 to 12 carbon atoms such as benzyl and phenethyl groups; some or all of hydrogen atoms of these groups may be chlorine atoms or fluorine atoms And a group substituted with a halogen atom such as
- the molecular structure of the component (B) is not limited, and examples thereof include a straight chain
- molecular chain both ends dimethylhydrogensiloxy group-blocked dimethylpolysiloxane, molecular chain both ends dimethylhydrogensiloxy group-blocked diphenylsiloxane oligomer, molecular chain both ends dimethylhydrogensiloxy group-blocked methylphenyl Polysiloxane, dimethylhydrogensiloxy group-capped methylphenylsiloxane oligomer at both ends of molecular chain, dimethylhydrogensiloxy group-capped dimethylsiloxane / methylhydrogensiloxane copolymer, molecular chain both-ends trimethylsiloxy group-capped methylhydrogen polysiloxane with both molecular chain terminals blocked with trimethylsiloxy groups dimethylsiloxane-methylhydrogensiloxane copolymer, wherein: the siloxane units of the formula represented by SiO 4/2 :( H 3) 2 HSiO organopoly
- the content of the component (B) is an amount such that the silicon-bonded hydrogen atoms in this component are in the range of 0.01 to 10 mol with respect to a total of 1 mol of alkenyl groups in the component (A), Preferably, the amount is in the range of 0.01 to 5 mol, the amount is in the range of 0.05 to 5 mol, or the amount is in the range of 0.01 to 5 mol. This is because when the content of component (B) is not less than the lower limit of the above range, the hydrosilylation reaction of the composition proceeds sufficiently, and a B-staged reactive hot melt silicone can be obtained.
- curing reactive hot melt silicone is favorable as it is below the upper limit of the said range.
- the amount of the alkenyl group in the component (A) is 1 mol in the component (B).
- the amount of silicon atom-bonded hydrogen atoms may be 1 mol or less, or less than 1 mol. This is because even when all of the silicon-bonded hydrogen atoms in the component (B) have reacted in order to crosslink the composition, the resulting reactive hot melt silicone can be thermally cured with an organic peroxide. Because it can.
- the component (C) is a catalyst for promoting a hydrosilylation reaction between the alkenyl group in the component (A) and the silicon atom-bonded hydrogen atom in the component (B).
- a platinum-based catalyst for promoting a hydrosilylation reaction between the alkenyl group in the component (A) and the silicon atom-bonded hydrogen atom in the component (B).
- a platinum-based catalyst for example, a platinum-based catalyst, a rhodium-based catalyst, Palladium-based catalysts are mentioned.
- a platinum-based catalyst is preferable because the hydrosilylation reaction of the present composition can be significantly accelerated.
- the platinum-based catalyst include platinum fine powder, chloroplatinic acid, chloroplatinic acid alcohol solution, platinum-alkenylsiloxane complex, platinum-olefin complex, and platinum-carbonyl complex, and platinum-alkenylsiloxane complex is particularly preferable. .
- alkenylsiloxane examples include 1,3-divinyl-1,1,3,3-tetramethyldisiloxane, 1,3,5,7-tetramethyl-1,3,5,7-tetravinylcyclotetrasiloxane, Examples thereof include alkenyl siloxanes in which part of the methyl groups of these alkenyl siloxanes are substituted with ethyl groups, phenyl groups, and the like, and alkenyl siloxanes in which the vinyl groups of these alkenyl siloxanes are substituted with allyl groups, hexenyl groups, and the like.
- the content of the component (C) is a catalyst amount that promotes the hydrosilylation reaction of the composition, and preferably, the metal atoms in the component are added to the total amount of the components (A) and (B).
- the present composition may contain (D) a reaction inhibitor in order to control the hydrosilylation reaction of the present composition.
- component (D) 1-ethynylcyclohexane-1-ol, 2-methyl-3-butyn-2-ol, 3,5-dimethyl-1-hexyn-3-ol, 2-phenyl-3-butyne- Alkyne alcohols such as 2-ol; Enyne compounds such as 3-methyl-3-penten-1-yne and 3,5-dimethyl-3-hexen-1-yne; Tris (1,1-dimethylpropyneoxy) methylsilane Alkyneoxysilanes such as bis (1,1-dimethylpropyneoxy) dimethylsilane; 1,3,5,7-tetramethyl-1,3,5,7-tetravinylcyclotetrasiloxane, 1,3,5 Alkenyl group-containing cyclosiloxane not corresponding to component (A), such as 1,7-tetramethyl
- the content of the component (D) is not limited, but is in the range of 0 to 5 parts by mass, or in the range of 0.0001 to 5 parts by mass with respect to 100 parts by mass in total of the components (A) to (C). It is preferable to be within. This is because the component (D) can be blended arbitrarily, but when the content of the component (D) is within the above range, reactive hot melt silicone can be easily prepared.
- blend (E) organic peroxide in order to thermoset the reactive hot melt silicone obtained in this composition, you may mix
- the component (E) preferably has no activity when the present composition is crosslinked in a B-stage shape, and has an activity when the resulting reactive hot melt silicone is heated. Those having a time half-life temperature of 90 ° C. or higher are preferred.
- Examples of the component (E) include alkyl peroxides, diacyl peroxides, peroxide esters, and carbonates.
- alkyl peroxides examples include dicumyl peroxide, di-tert-butyl peroxide, di-tert-butylcumyl peroxide, and 2,5-dimethyl-2,5-di (tert-butylperoxy) hexane. 2,5-dimethyl-2,5-di (tert-butylperoxy) hexyne-3, tert-butylcumyl, 1,3-bis (tert-butylperoxyisopropyl) benzene, 3,6,9-triethyl- 3,6,9-trimethyl-1,4,7-triperoxonan.
- diacyl peroxides examples include benzoyl peroxide, lauroyl peroxide, and decanoyl peroxide.
- peroxide esters examples include 1,1,3,3-tetramethylbutylperoxyneodecanoate, ⁇ -cumylperoxyneodecanoate, tert-butylperoxyneodecanoate, and tert-butyl.
- Peroxyneoheptanoate tert-butyl peroxypivalate, tert-hexyl peroxypivalate, 1,1,3,3-tetramethylbutylperoxy-2-ethylhexanoate, tert-amyl peroxyl- 2-ethylhexanoate, tert-butylperoxy-2-ethylhexanoate, tert-butylperoxyisobutyrate, di-tert-butylperoxyhexahydroterephthalate, tert-amylperoxy-3,5, 5-trimethylhexanoate, tert-butylperoxy -3,5,5-trimethylhexanoate, tert-butyl peroxyacetate, tert-butyl peroxybenzoate, and di-butyl peroxytrimethyl adipate.
- peroxide carbonates examples include di-3-methoxybutyl peroxydicarbonate, di (2-ethylhexyl) peroxydicarbonate, diisopropyl peroxycarbonate, tert-butylperoxyisopropyl carbonate, and di (4-tert- Butylcyclohexyl) peroxydicarbonate, dicetylperoxydicarbonate, dimyristylperoxydicarbonate.
- alkyl peroxides are preferable, and those having a 10-hour half-life temperature of 90 ° C. or higher, or 95 ° C. or higher are particularly preferable.
- Examples of such component (E) include dicumyl peroxide, di-t-butyl peroxide, di-t-hexyl peroxide, t-butylcumyl peroxide, 2,5-dimethyl-2,5-di ( tert-butylperoxy) hexane, 1,3-bis (tert-butylperoxyisopropyl) benzene, di- (2-tert-butylperoxyisopropyl) benzene, 3,6,9-triethyl-3,6,9- And trimethyl-1,4,7-triperoxonan.
- the content of the component (E) is not limited, but is in the range of 0.01 to 10 parts by mass and in the range of 0.05 to 10 parts by mass with respect to the total of 100 parts by mass of the components (A) to (C). Of these, the range of 0.05 to 5 parts by mass or the range of 0.01 to 5 parts by mass is preferable. This is because when the content of the component (E) is not less than the lower limit of the above range, the resulting reactive hot melt silicone can be sufficiently heat-cured, and on the other hand, not more than the upper limit of the above range. This is because bubbles are hardly generated in the obtained cured product.
- an adhesion-imparting agent may be added to the present composition in order to impart adhesion to the resulting reactive hot melt silicone.
- an adhesion-imparting agent an organosilicon compound having at least one alkoxy group bonded to a silicon atom in one molecule is preferable.
- the alkoxy group include a methoxy group, an ethoxy group, a propoxy group, a butoxy group, and a methoxyethoxy group, and a methoxy group is particularly preferable.
- Examples of the group other than the alkoxy group bonded to the silicon atom of the organosilicon compound include halogen-substituted or unsubstituted monovalent hydrocarbon groups such as an alkyl group, an alkenyl group, an aryl group, an aralkyl group, and a halogenated alkyl group; Glycidoxyalkyl groups such as 3-glycidoxypropyl group and 4-glycidoxybutyl group; 2- (3,4-epoxycyclohexyl) ethyl group, 3- (3,4-epoxycyclohexyl) propyl group and the like
- Examples include epoxycyclohexylalkyl groups; epoxyalkyl groups such as 3,4-epoxybutyl groups and 7,8-epoxyoctyl groups; acrylic group-containing monovalent organic groups such as 3-methacryloxypropyl groups; and hydrogen atoms.
- This organosilicon compound preferably has an alkenyl group or a group capable of reacting with a silicon atom-bonded hydrogen atom in the composition, and specifically, preferably has a silicon atom-bonded hydrogen atom or an alkenyl group. Moreover, since it can provide favorable adhesiveness to various types of substrates, the organosilicon compound preferably has at least one epoxy group-containing monovalent organic group in one molecule. Examples of such organosilicon compounds include organosilane compounds, organosiloxane oligomers, and alkyl silicates.
- Examples of the molecular structure of the organosiloxane oligomer or alkyl silicate include linear, partially branched linear, branched, cyclic, and network, particularly linear, branched, and network. Preferably there is.
- organosilicon compounds include silane compounds such as 3-glycidoxypropyltrimethoxysilane, 2- (3,4-epoxycyclohexyl) ethyltrimethoxysilane, and 3-methacryloxypropyltrimethoxysilane;
- Examples thereof include a mixture of a siloxane compound having at least one atom-bonded hydroxy group and at least one silicon atom-bonded alkenyl group, methyl polysilicate, ethyl polysilicate, and epoxy group-containing ethyl polysilicate.
- the content of the adhesion-imparting agent is not limited, but is preferably in the range of 0.01 to 10 parts by mass with respect to 100 parts by mass in total of the components (A) to (C). This is because the adhesiveness of the resulting reactive hot melt silicone is good when the content of the adhesion-imparting agent is within the above range.
- the present composition includes trimethylsiloxy-capped dimethylpolysiloxane, trimethylsiloxy-capped dimethylsiloxane / methylphenylsiloxane copolymer, formula: (CH 3 ) 3
- An organopolysiloxane having no alkenyl group and no silicon-bonded hydrogen atom such as an organopolysiloxane composed of a siloxane unit represented by SiO 1/2 and a siloxane unit represented by the formula: SiO 4/2 Good.
- This organopolysiloxane preferably has a softening point of 50 ° C. or higher.
- the amount of the organopolysiloxane blended is not limited, but is preferably in the range of 0 to 250 parts by mass with respect to 100 parts by mass in total of the components (A) to (C). This is because it is easy to prepare a reactive hot melt silicone when the content of such an organopolysiloxane is within the above range.
- the present composition includes, as other optional components, inorganic fillers such as silica, titanium oxide, glass, alumina, and zinc oxide; fine organic resin powders such as polymethacrylate resins; heat-resistant agents, phosphors, dyes, and pigments
- inorganic fillers such as silica, titanium oxide, glass, alumina, and zinc oxide
- fine organic resin powders such as polymethacrylate resins
- heat-resistant agents such as phosphors, dyes, and pigments
- a flame retardant imparting agent may be blended.
- the container is filled with a crosslinkable silicone composition containing the components (A) to (C) and other components as necessary, and the container is heated.
- the composition is preferably degassed.
- the composition is crosslinked in a B-stage by heating the container filled with the composition.
- the reactive hot-melt silicone thus obtained is non-flowable at 25 ° C. and has a melt viscosity at 120 ° C. of 5,000 Pa ⁇ s or less, preferably in the range of 10 to 3,500 Pa ⁇ s.
- non-fluidity means that the material does not flow in an unloaded state.
- the ring-and-ball method of a hot melt adhesive specified in JIS K 6863-1994 “Testing method for softening point of hot melt adhesive” In order to show a state below the softening point measured by the softening point test method according to, that is, to be non-flowable at 25 ° C, the softening point needs to be higher than 25 ° C.
- the container that can be used as the reactive hot melt silicone filled container of the present invention is not particularly limited as long as it has heat resistance and does not inhibit the hydrosilylation reaction of the crosslinkable silicone composition.
- Such containers include plastic or metal cartridges; plastic film packs, metal foil laminated film packs, flexible containers such as bellows-like containers, tube-like containers; plastic or metal pail cans; and plastic or A metal drum can is illustrated, and a cartridge is particularly preferable.
- a cartridge for example, PSY-30FH2-P or PSY-30FH-P manufactured by Musashi Engineering Co., Ltd. can be used.
- FIG. 1 is a perspective view showing a partially broken surface of a cartridge which is an example of a reactive hot melt silicone-filled container of the present invention.
- a reactive hot-melt silicone 2 is filled in a cartridge 1
- a plunger 3 for melting and extruding the reactive hot-melt silicone is provided inside the cartridge.
- the reactive hot melt silicone is put into a melter to bring the reactive hot melt silicone into a molten state and the plunger is moved by mechanical force or gas pressure to extrude the reactive hot melt silicone.
- the shape, film thickness, coating area, and the like can be controlled by controlling the discharge amount of the reactive hot melt silicone.
- any shape such as a hemispherical shape, a semi-cylindrical shape, a dome shape, etc. can be obtained without entraining air on the optical semiconductor element mounted on the substrate having a flat surface. Can be sealed or covered.
- the method for producing the reactive hot melt silicone of the present invention is as described above, and a crosslinkable silicone composition containing the components (A) to (C) and other optional components as necessary is used as a container. It is characterized in that it is heated in a B-stage to cause crosslinking.
- the present composition is heated in a container to be crosslinked in a B-stage shape, volatilization such as low molecular weight components and reaction suppression contained in the present composition is suppressed, and hydrosilylation reaction It is difficult to cause problems such as air inhibition.
- the reactive hot melt silicone filling container of the present invention and the method for producing reactive hot melt silicone will be described in detail with reference to examples.
- Me, Ph, and Vi represent a methyl group, a phenyl group, and a vinyl group, respectively.
- the melt viscosity at 120 ° C. of the reactive hot melt silicone, the time for the initial torque value to develop, and the hardness of the cured product were measured as follows.
- melt viscosity of reactive hot melt silicone The melt viscosity at 120 ° C. of the reactive hot melt silicone was measured at a shear rate of 1 / s using an AR550 rheometer manufactured by TA Instruments using a cone plate having a diameter of 20 mm and a cone angle of 2 °. .
- Examples 1 to 5 The following components were uniformly mixed in the composition (parts by mass) shown in Table 1 to prepare a crosslinkable silicone composition.
- SiH / Vi represents the number of moles of silicon-bonded hydrogen atoms in component (B) relative to 1 mole of vinyl group in component (A).
- the crosslinkable silicone composition was filled in a cartridge of 30 cc capacity, heated under the conditions shown in Table 1, and then cooled to room temperature to prepare a reactive hot melt silicone filled container.
- (B) As the component (B), the following components were used.
- (B-1) Component: Formula: Me 2 HSiOPh 2 SiOSiMe 2 H Organopolysiloxane represented by
- Component (c) 1,3,5,7-tetramethyl-1,3,5,7-tetravinyl of platinum-1,3-divinyl-1,1,3,3-tetramethyldisiloxane complex Cyclotetrasiloxane solution (solution containing 0.1 wt% platinum metal)
- Component (f-1) Adhesion-imparting agent comprising a condensation reaction product of a silanol group-blocked methylvinylsiloxane oligomer having a viscosity of 30 mPa ⁇ s and 3-glycidoxypropyltrimethoxysilane
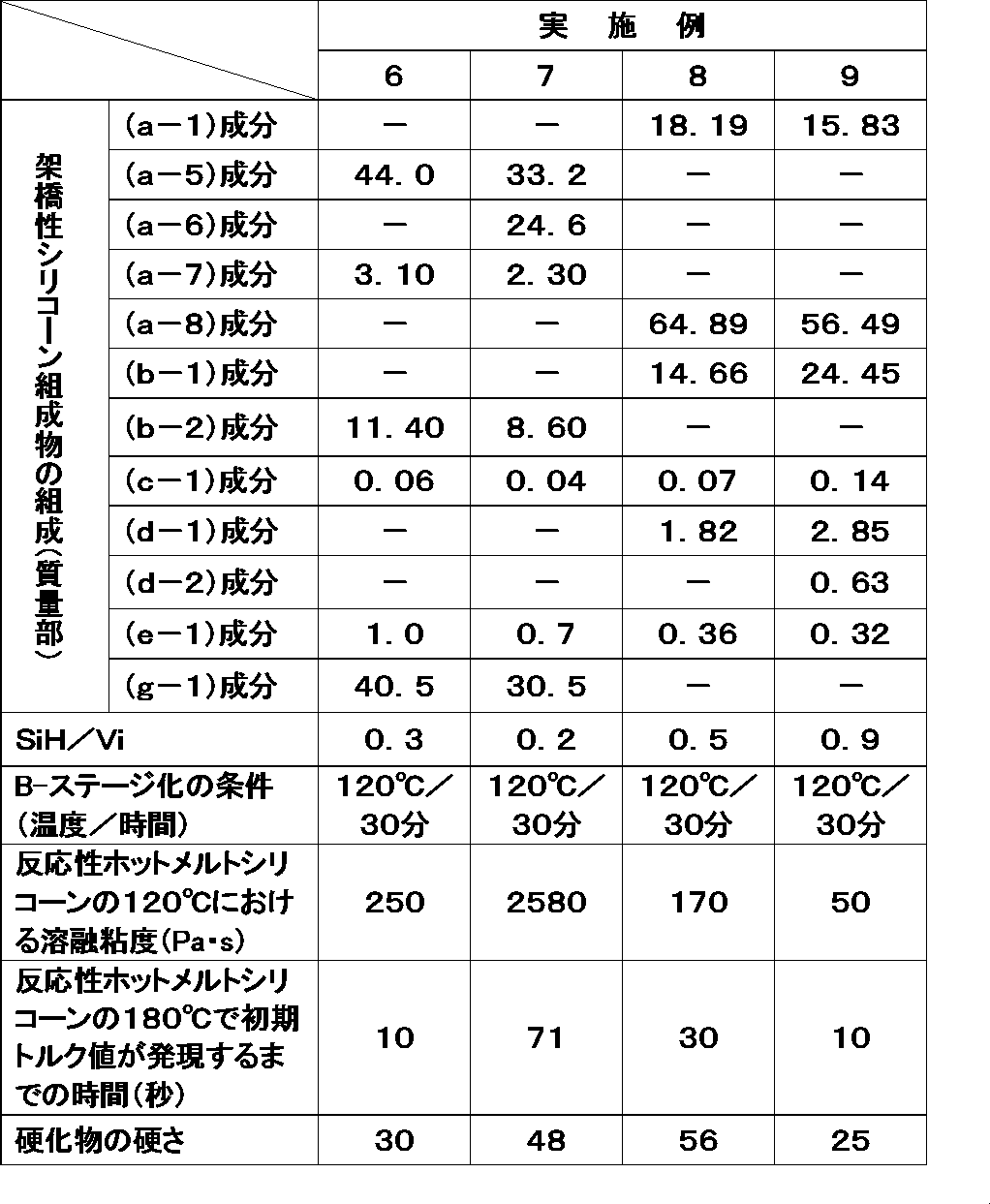
- Examples 6 to 9 The above and the following components were uniformly mixed in the composition (parts by mass) shown in Table 2 to prepare a crosslinkable silicone composition.
- SiH / Vi represents the number of moles of silicon-bonded hydrogen atoms in the component (B) with respect to 1 mole of the vinyl group in the component (A).
- this crosslinkable silicone composition was filled in a cartridge of 30 cc capacity, heated under the conditions shown in Table 2, and then cooled to room temperature to prepare a reactive hot melt silicone filled container.
- Component (a-5) average unit formula having a softening point of 300 ° C .: (Me 2 ViSiO 1/2 ) 0.10 (Me 3 SiO 1/2 ) 0.40 (SiO 4/2 ) 0.50
- (B-2) Component: Formula: Me 2 HSiO (Me 2 SiO) 25 SiMe 2 H Organopolysiloxane represented by
- component (D) 1,3,5,7-tetramethyl-1,3,5,7-tetravinylcyclotetrasiloxane
- component (d-2) component tris (1,1-dimethylpropyneoxy) methylsilane
- (E) As the component (E), the following components were used in addition to the above.
- the reactive hot melt silicone-filled container of the present invention can extract reactive hot melt silicone with good gap fill property by heating, in a chip array module in which an optical semiconductor element such as an LED is mounted on a substrate.
- the optical semiconductor element can be efficiently sealed and covered.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Materials Engineering (AREA)
- Polymers & Plastics (AREA)
- Medicinal Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Power Engineering (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Computer Hardware Design (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Adhesives Or Adhesive Processes (AREA)
Abstract
Description
(A)アルケニル基を有し、軟化点が50℃以上である分岐鎖状のオルガノポリシロキサンを少なくとも含む、アルケニル基結合オルガノポリシロキサン、
(B)一分子中に少なくとも2個のケイ素原子結合水素原子を有するオルガノポリシロキサン{(A)成分中のアルケニル基1モルに対して、本成分中のケイ素原子結合水素原子が0.01~10モルとなる量}、および
(C)触媒量のヒドロシリル化反応用触媒
を含む架橋性シリコーン組成物を容器に充填し、該容器を加熱することにより前記組成物をB-ステージ状に架橋させ、25℃で非流動性であり、120℃での溶融粘度が5,000Pa・s以下の反応性ホットメルトシリコーンを形成することを特徴とする。
(A)アルケニル基を有し、軟化点が50℃以上である分岐鎖状のオルガノポリシロキサンを少なくとも含む、アルケニル基結合オルガノポリシロキサン、
(B)一分子中に少なくとも2個のケイ素原子結合水素原子を有するオルガノポリシロキサン{(A)成分中のアルケニル基1モルに対して、本成分中のケイ素原子結合水素原子が0.01~10モルとなる量}、および
(C)触媒量のヒドロシリル化反応用触媒
を含む架橋性シリコーン組成物を容器中で加熱してB-ステージ状に架橋させることを特徴とする。
(A)アルケニル基を有し、軟化点が50℃以上である分岐鎖状のオルガノポリシロキサンを少なくとも含む、アルケニル基結合オルガノポリシロキサン、
(B)一分子中に少なくとも2個のケイ素原子結合水素原子を有するオルガノポリシロキサン{(A)成分中のアルケニル基1モルに対して、本成分中のケイ素原子結合水素原子が0.01~10モルとなる量}、および
(C)触媒量のヒドロシリル化反応用触媒
を含む架橋性シリコーン組成物を容器に充填し、該容器を加熱することにより前記組成物をB-ステージ状に架橋させることを特徴とする。なお、この「B-ステージ状」とは、JIS K 6800に定義されているB-ステージ(熱硬化性樹脂の硬化中間体)の状態をいい、架橋性シリコーン組成物を不完全に硬化させることにより、溶剤により膨潤するものの、完全に溶解することがなく、「反応性ホットメルト」とは、室温(25℃)では流動性を失ったような状態であるが、高温(例えば、120℃以上)に加熱すると再び溶融し、次いで硬化するものをいう。
R2 3SiO(R2 2SiO)nSiR2 3
で表される。
反応性ホットメルトシリコーンの120℃の溶融粘度を、ティー・エイ・インスツルメント社製のAR550レオメーターにより、直径20mm、コーン角2°のコーンプレートを用いて、せん断速度1/sで測定した。
所定温度に設定した測定装置(ALPHA TECHNOLOGIES Rheometer MDR 2000P)の固定された下ダイと昇降する上ダイから構成される円盤状のダイ中空部に6gの反応性ホットメルトシリコーンをセットし、上下のダイを密閉した後、振動数1.66Hz、振動角=1°の条件において、トルク値が1dNmに到達するまでの時間を測定し、これを初期トルク値が発現するまでの時間とした。
反応性ホットメルトシリコーンを150℃で2時間プレス成形することによりシート状の硬化物を作製した。このシート状硬化物の硬さをJIS K 6253に規定されるタイプDデュロメータにより測定した。
下記の成分を表1に示した組成(質量部)で均一に混合して架橋性シリコーン組成物を調製した。なお、表1中、SiH/Viは、(A)成分中のビニル基1モルに対する、(B)成分中のケイ素原子結合水素原子のモル数を示す。その後、この架橋性シリコーン組成物を容量30ccのカートリッジ中に充填し、表1に示した条件で加熱し、次いで、室温まで冷却することにより反応性ホットメルトシリコーン充填容器を作製した。
(a-1)成分:式:
ViMe2SiO(MePhSiO)80SiMe2Vi
で表されるメチルフェニルポリシロキサン
(a-2)成分:式:
ViMe2SiO(MePhSiO)18SiMe2Vi
で表されるメチルフェニルポリシロキサン
(a-3)成分:軟化点が100℃である、平均単位式:
(MeViSiO2/2)0.3(PhSiO3/2)0.7
で表されるオルガノポリシロキサン
(a-4)成分:軟化点が150℃である、平均単位式
(MeViSiO2/2)0.10(Me2SiO2/2)0.15(PhSiO3/2)0.75
で表されるオルガノポリシロキサン
(b-1)成分:式:
Me2HSiOPh2SiOSiMe2H
で表されるオルガノポリシロキサン
(c-1)成分:白金-1,3-ジビニル-1,1,3,3-テトラメチルジシロキサン錯体の1,3,5,7-テトラメチル-1,3,5,7-テトラビニルシクロテトラシロキサン溶液(白金金属を0.1重量%含有する溶液)
(f-1)成分:粘度が30mPa・sである分子鎖両末端シラノール基封鎖メチルビニルシロキサンオリゴマーと3-グリシドキシプロピルトリメトキシシランの縮合反応物からなる接着付与剤
上記および下記の成分を表2に示した組成(質量部)で均一に混合して架橋性シリコーン組成物を調製した。なお、表2中、SiH/Viは、(A)成分中のビニル基1モルに対する、(B)成分中のケイ素原子結合水素原子のモル数を示す。その後、この架橋性シリコーン組成物を容量30ccのカートリッジ中に充填し、表2に示した条件で加熱し、次いで室温まで冷却することにより反応性ホットメルトシリコーン充填容器を作製した。
(a-5)成分:軟化点が300℃である、平均単位式:
(Me2ViSiO1/2)0.10(Me3SiO1/2)0.40(SiO4/2)0.50
で表されるオルガノポリシロキサン
(a-6)成分:軟化点が300℃である、平均単位式
(Me2ViSiO1/2)0.04(Me3SiO1/2)0.40(SiO4/2)0.56
で表されるオルガノポリシロキサン
(a-7)成分:式:
ViMe2SiO(Me2SiO)770SiMe2Vi
で表されるジメチルポリシロキサン
(a-8)成分:軟化点が100℃である、平均単位式:
(Me2ViSiO1/2)0.25(PhSiO3/2)0.75
で表されるオルガノポリシロキサン
(b-2)成分:式:
Me2HSiO(Me2SiO)25SiMe2H
で表されるオルガノポリシロキサン
(d-1)成分:1,3,5,7-テトラメチル-1,3,5,7-テトラビニルシクロテトラシロキサン
(d-2)成分:トリス(1,1-ジメチルプロピンオキシ)メチルシラン
(e-1)成分:2,5-ジメチル-2,5-ジ(t-ブチルパオキシ)ヘキサン
(g-1)成分:軟化点100℃である、平均単位式:
(Me3SiO1/2)0.44(SiO4/2)0.56
で表されるオルガノポリシロキサン
2 反応性ホットメルトシリコーン
3 プランジャー
Claims (8)
- (A)アルケニル基を有し、軟化点が50℃以上である分岐鎖状のオルガノポリシロキサンを少なくとも含む、アルケニル基結合オルガノポリシロキサン、
(B)一分子中に少なくとも2個のケイ素原子結合水素原子を有するオルガノポリシロキサン{(A)成分中のアルケニル基1モルに対して、本成分中のケイ素原子結合水素原子が0.01~10モルとなる量}、および
(C)触媒量のヒドロシリル化反応用触媒
を含む架橋性シリコーン組成物を容器に充填し、該容器を加熱することにより前記組成物をB-ステージ状に架橋させ、25℃で非流動性であり、120℃での溶融粘度が5,000Pa・s以下の反応性ホットメルトシリコーンを形成することを特徴とする反応性ホットメルトシリコーン充填容器。 - 架橋性シリコーン組成物が、さらに、(A)成分~(C)成分の合計100質量部に対して0.0001~5質量部の(D)反応抑制剤を含む、請求項1に記載の反応性ホットメルトシリコーン充填容器。
- 架橋性シリコーン組成物が、さらに、(A)成分~(C)成分の合計100質量部に対して0.01~10質量部の(E)有機パーオキサイドを含む、請求項1または2に記載の反応性ホットメルトシリコーン充填容器。
- 容器が、カートリッジ、フレキシブル容器、ペール缶、またはドラム缶である、請求項1乃至3のいずれか1項に記載の反応性ホットメルトシリコーン充填容器。
- (A)アルケニル基を有し、軟化点が50℃以上である分岐鎖状のオルガノポリシロキサンを少なくとも含む、アルケニル基結合オルガノポリシロキサン、
(B)一分子中に少なくとも2個のケイ素原子結合水素原子を有するオルガノポリシロキサン{(A)成分中のアルケニル基1モルに対して、本成分中のケイ素原子結合水素原子が0.01~10モルとなる量}、および
(C)触媒量のヒドロシリル化反応用触媒
を含む架橋性シリコーン組成物を容器中で加熱してB-ステージ状に架橋させることを特徴とする、25℃で非流動性であり、120℃での溶融粘度が5,000Pa・s以下の反応性ホットメルトシリコーンの製造方法。 - 架橋性シリコーン組成物が、さらに、(A)成分~(C)成分の合計100質量部に対して0.0001~5質量部の(D)反応抑制剤を含む、請求項5に記載の反応性ホットメルトシリコーンの製造方法。
- 架橋性シリコーン組成物が、さらに、(A)成分~(C)成分の合計100質量部に対して0.01~10質量部の(E)有機パーオキサイドを含む、請求項5または6に記載の反応性ホットメルトシリコーンの製造方法。
- 容器が、カートリッジ、フレキシブル容器、ペール缶、またはドラム缶である、請求項5乃至7のいずれか1項に記載の反応性ホットメルトシリコーンの製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP17866632.7A EP3536747A4 (en) | 2016-11-02 | 2017-10-23 | FILLING CONTAINER FOR REACTIVE HOT MEL SILICONE AND METHOD FOR THE PRODUCTION OF REACTIVE HOT MEL SILICONE |
| CN201780065370.5A CN109963911A (zh) | 2016-11-02 | 2017-10-23 | 反应性热熔硅酮填充容器及用于制造反应性热熔硅酮的方法 |
| KR1020197013355A KR20190080884A (ko) | 2016-11-02 | 2017-10-23 | 반응성 핫멜트 실리콘 충전 용기 및 반응성 핫멜트 실리콘의 제조방법 |
| US16/346,015 US20190276684A1 (en) | 2016-11-02 | 2017-10-23 | Reactive hot-melt silicone filling container and method for manufacturing reactive hot-melt silicone |
| JP2018548632A JP7009381B2 (ja) | 2016-11-02 | 2017-10-23 | 反応性ホットメルトシリコーン充填容器および反応性ホットメルトシリコーンの製造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016214766 | 2016-11-02 | ||
| JP2016-214766 | 2016-11-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018084012A1 true WO2018084012A1 (ja) | 2018-05-11 |
Family
ID=62075797
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/038184 WO2018084012A1 (ja) | 2016-11-02 | 2017-10-23 | 反応性ホットメルトシリコーン充填容器および反応性ホットメルトシリコーンの製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20190276684A1 (ja) |
| EP (1) | EP3536747A4 (ja) |
| JP (1) | JP7009381B2 (ja) |
| KR (1) | KR20190080884A (ja) |
| CN (1) | CN109963911A (ja) |
| TW (1) | TWI765925B (ja) |
| WO (1) | WO2018084012A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020203304A1 (ja) * | 2019-03-29 | 2020-10-08 | ダウ・東レ株式会社 | 硬化性シリコーン組成物、その硬化物、およびその製造方法 |
| WO2020203307A1 (ja) * | 2019-03-29 | 2020-10-08 | ダウ・東レ株式会社 | 硬化性シリコーン組成物、その硬化物、およびその製造方法 |
| WO2020203305A1 (ja) * | 2019-03-29 | 2020-10-08 | ダウ・東レ株式会社 | 硬化性シリコーン組成物、その硬化物、およびその製造方法 |
| WO2022138335A1 (ja) * | 2020-12-25 | 2022-06-30 | ダウ・東レ株式会社 | 積層体の製造方法 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111278747B (zh) * | 2018-09-07 | 2021-08-03 | 瓦克化学股份公司 | 用于封装硅酮化合物的方法 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011219597A (ja) | 2010-04-08 | 2011-11-04 | Nitto Denko Corp | シリコーン樹脂シート |
| WO2015093329A1 (ja) * | 2013-12-19 | 2015-06-25 | 東レ・ダウコーニング株式会社 | シリコーン接着性フィルム、および半導体装置 |
| JP2016504461A (ja) * | 2012-12-21 | 2016-02-12 | ダウ コーニング コーポレーションDow Corning Corporation | 圧縮成形又はラミネート用ホットメルト型硬化性シリコーン組成物 |
| WO2016103654A1 (ja) * | 2014-12-26 | 2016-06-30 | 東レ・ダウコーニング株式会社 | 硬化性オルガノポリシロキサン組成物、それからなる半導体用封止剤および半導体装置 |
| JP2016124967A (ja) * | 2014-12-26 | 2016-07-11 | 東レ・ダウコーニング株式会社 | 硬化性シリコーン組成物、それからなる半導体用封止剤および半導体装置 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08295783A (ja) * | 1995-04-26 | 1996-11-12 | Toray Dow Corning Silicone Co Ltd | 硬化性樹脂組成物 |
| CN101935455B (zh) * | 2010-07-28 | 2012-10-24 | 杭州师范大学 | 一种led封装用有机硅材料及其制备方法 |
| KR20150097947A (ko) * | 2014-02-19 | 2015-08-27 | 다우 코닝 코포레이션 | 반응성 실리콘 조성물, 이로부터 제조되는 핫멜트 재료, 및 경화성 핫멜트 조성물 |
| JP6607644B2 (ja) * | 2014-09-01 | 2019-11-20 | ダウ・東レ株式会社 | 硬化性シリコーン組成物、硬化性ホットメルトシリコーン、および光デバイス |
| EP3365391A4 (en) * | 2015-10-19 | 2019-06-12 | Dow Corning Toray Co., Ltd. | MELT SILICONE COMPOSITION CURABLE BY ACTIVE ENERGY RADIATION, CURED PRODUCT THEREOF, AND FILM PRODUCTION PROCESS |
| KR102358091B1 (ko) * | 2016-08-08 | 2022-02-07 | 듀폰 도레이 스페셜티 머티리얼즈 가부시키가이샤 | 경화성 입상 실리콘 조성물, 이것으로 이루어지는 반도체용 부재 및 이의 성형 방법 |
-
2017
- 2017-10-23 JP JP2018548632A patent/JP7009381B2/ja active Active
- 2017-10-23 CN CN201780065370.5A patent/CN109963911A/zh active Pending
- 2017-10-23 EP EP17866632.7A patent/EP3536747A4/en not_active Withdrawn
- 2017-10-23 WO PCT/JP2017/038184 patent/WO2018084012A1/ja unknown
- 2017-10-23 US US16/346,015 patent/US20190276684A1/en not_active Abandoned
- 2017-10-23 KR KR1020197013355A patent/KR20190080884A/ko active IP Right Grant
- 2017-10-26 TW TW106136908A patent/TWI765925B/zh active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011219597A (ja) | 2010-04-08 | 2011-11-04 | Nitto Denko Corp | シリコーン樹脂シート |
| JP2016504461A (ja) * | 2012-12-21 | 2016-02-12 | ダウ コーニング コーポレーションDow Corning Corporation | 圧縮成形又はラミネート用ホットメルト型硬化性シリコーン組成物 |
| WO2015093329A1 (ja) * | 2013-12-19 | 2015-06-25 | 東レ・ダウコーニング株式会社 | シリコーン接着性フィルム、および半導体装置 |
| WO2016103654A1 (ja) * | 2014-12-26 | 2016-06-30 | 東レ・ダウコーニング株式会社 | 硬化性オルガノポリシロキサン組成物、それからなる半導体用封止剤および半導体装置 |
| JP2016124967A (ja) * | 2014-12-26 | 2016-07-11 | 東レ・ダウコーニング株式会社 | 硬化性シリコーン組成物、それからなる半導体用封止剤および半導体装置 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3536747A4 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020203304A1 (ja) * | 2019-03-29 | 2020-10-08 | ダウ・東レ株式会社 | 硬化性シリコーン組成物、その硬化物、およびその製造方法 |
| WO2020203307A1 (ja) * | 2019-03-29 | 2020-10-08 | ダウ・東レ株式会社 | 硬化性シリコーン組成物、その硬化物、およびその製造方法 |
| WO2020203305A1 (ja) * | 2019-03-29 | 2020-10-08 | ダウ・東レ株式会社 | 硬化性シリコーン組成物、その硬化物、およびその製造方法 |
| CN113631659A (zh) * | 2019-03-29 | 2021-11-09 | 陶氏东丽株式会社 | 固化性有机硅组合物、其固化物及其制造方法 |
| CN113631660A (zh) * | 2019-03-29 | 2021-11-09 | 陶氏东丽株式会社 | 固化性有机硅组合物、其固化物及其制造方法 |
| WO2022138335A1 (ja) * | 2020-12-25 | 2022-06-30 | ダウ・東レ株式会社 | 積層体の製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2018084012A1 (ja) | 2019-09-19 |
| EP3536747A1 (en) | 2019-09-11 |
| JP7009381B2 (ja) | 2022-01-25 |
| TW201829628A (zh) | 2018-08-16 |
| EP3536747A4 (en) | 2020-07-01 |
| CN109963911A (zh) | 2019-07-02 |
| TWI765925B (zh) | 2022-06-01 |
| US20190276684A1 (en) | 2019-09-12 |
| KR20190080884A (ko) | 2019-07-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2018084012A1 (ja) | 反応性ホットメルトシリコーン充填容器および反応性ホットメルトシリコーンの製造方法 | |
| CN109689791B (zh) | 固化性粒状硅组合物、由其构成的半导体用构件及其成型方法 | |
| JP5638714B2 (ja) | 発光素子封止用シリコーン組成物及び発光装置 | |
| JP4965111B2 (ja) | 硬化性シリコーン組成物 | |
| CN106604970B (zh) | 可固化硅酮组合物、可固化热熔硅酮以及光学装置 | |
| JP5992666B2 (ja) | 架橋性シリコーン組成物及びその架橋物 | |
| KR102282547B1 (ko) | 다이 본딩용 경화성 실리콘 조성물 | |
| JP2009242627A (ja) | 硬化性オルガノポリシロキサン組成物およびその硬化物 | |
| WO2018030286A1 (ja) | 硬化性粒状シリコーン組成物、それからなる光反射材、およびその製造方法 | |
| JP2006348119A (ja) | 硬化性オルガノポリシロキサン組成物 | |
| CN113631660B (zh) | 固化性有机硅组合物、其固化物及其制造方法 | |
| CN113330071A (zh) | 固化性有机硅组合物、其固化物及其制造方法 | |
| EP3587498B1 (en) | Curable organopolysiloxane composition and semiconductor device | |
| TWI786120B (zh) | 固化性有機矽組合物、由其構成之光反射材料、及其製造方法 | |
| TWI787444B (zh) | 可固化聚矽氧組成物、其固化產物、及光學半導體裝置 | |
| JP2020033407A (ja) | 付加硬化型シリコーン組成物及び半導体装置 | |
| WO2020154272A1 (en) | Curable silicone composition and cured product thereof | |
| JP2023132308A (ja) | 硬化性シリコーン組成物 | |
| JP2023132309A (ja) | 硬化性シリコーン組成物 | |
| JP5931999B2 (ja) | 半導体パッケージ用硬化性樹脂組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17866632 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2018548632 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20197013355 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2017866632 Country of ref document: EP Effective date: 20190603 |