WO2017175552A1 - 光学ガラス、プリフォーム材及び光学素子 - Google Patents
光学ガラス、プリフォーム材及び光学素子 Download PDFInfo
- Publication number
- WO2017175552A1 WO2017175552A1 PCT/JP2017/010321 JP2017010321W WO2017175552A1 WO 2017175552 A1 WO2017175552 A1 WO 2017175552A1 JP 2017010321 W JP2017010321 W JP 2017010321W WO 2017175552 A1 WO2017175552 A1 WO 2017175552A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- component
- less
- glass
- content
- preferably less
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C3/00—Glass compositions
- C03C3/04—Glass compositions containing silica
- C03C3/062—Glass compositions containing silica with less than 40% silica by weight
- C03C3/064—Glass compositions containing silica with less than 40% silica by weight containing boron
- C03C3/068—Glass compositions containing silica with less than 40% silica by weight containing boron containing rare earths
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C3/00—Glass compositions
- C03C3/04—Glass compositions containing silica
- C03C3/062—Glass compositions containing silica with less than 40% silica by weight
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C3/00—Glass compositions
- C03C3/04—Glass compositions containing silica
- C03C3/076—Glass compositions containing silica with 40% to 90% silica, by weight
- C03C3/095—Glass compositions containing silica with 40% to 90% silica, by weight containing rare earths
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C3/00—Glass compositions
- C03C3/04—Glass compositions containing silica
- C03C3/076—Glass compositions containing silica with 40% to 90% silica, by weight
- C03C3/097—Glass compositions containing silica with 40% to 90% silica, by weight containing phosphorus, niobium or tantalum
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C4/00—Compositions for glass with special properties
- C03C4/20—Compositions for glass with special properties for chemical resistant glass
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B1/00—Optical elements characterised by the material of which they are made; Optical coatings for optical elements
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C2204/00—Glasses, glazes or enamels with special properties
Definitions
- the present invention relates to an optical glass, a preform material, and an optical element.
- optical systems such as digital cameras and video cameras
- optical systems such as surveillance cameras and in-vehicle cameras
- Lenses used in optical systems used for these applications are required to have durability that can withstand wind and rain, chemicals, and the like.
- n d refractive index
- ⁇ d Abbe number
- the chemical durability of the optical glass in the present invention is 1 to 3 grade, and the wear degree of JOGIS 10-1994 optical glass It is desired that the degree of wear by the measurement method is 200 or less. As a result, there is no spider or interference film on the lens surface caused by water vapor, carbon dioxide gas, rain, etc. in the atmosphere, which is generally called “white discoloration” or “blue discoloration”. It is possible to obtain glass that is not damaged even when rubbed or collided.
- the optical glass used for the optical element is required to be obtained stably when formed as glass.
- stability devitrification resistance
- glass suitable as an optical element can no longer be obtained.
- B 2 O 3 —La 2 O 3 based optical glasses containing a large amount of the B 2 O 3 component exist.
- a large amount of rare earth components can be introduced, contributing to stability and high refractive index during glass formation. It is to do.
- the durability such as chemical durability (acid resistance) and wear level is lowered.
- Patent Documents 1 and 2 are so-called B 2 O 3 —La 2 O 3 based optical glass, which has poor chemical durability (acid resistance) and a high degree of wear. However, it is not preferable when it is assumed to be exposed to the external environment.
- the present invention was made in view of the above problems, and an object has a refractive index (n d) 1.60 to 1.95 of the refractive index (n d), 25
- the object is to obtain a glass having a high chemical durability (acid resistance) and a low degree of wear, while an Abbe number ( ⁇ d ) of 62 or less is within a desired range.
- the present inventors have conducted earnest test research.
- the refractive index (n d ) and the Abbe number ( ⁇ d ) there is in the desired range, in particular, has a refractive index (n d) 1.78 to 1.95 of the refractive index (n d), the glass having a 25 or more 45 or less the Abbe number ( ⁇ d) ( optical glass) of the first aspect, in particular, has a refractive index (n d) 1.60 to 1.85 of the refractive index (n d), the glass having a 33 or 62 following an Abbe number ([nu d) In (the optical glass of the second embodiment), it has been found that devitrification is lowered while reducing the content of the component that lowers chemical durability (acid resistance), particularly the B 2 O 3 component.
- the invention has been completed. Specifically, the present invention provides the following.
- SiO 2 component is 10.0% to 40.0%, La 2 O 3 component 15.0% to 50.0%, TiO 2 component 5.0 to less than 25.0%, Contains,
- the mass ratio B 2 O 3 / SiO 2 is 1.00 or less, Having a refractive index (nd) of 1.78 to 1.95, an Abbe number ( ⁇ d) of 25 to 45;
- Ln 2 O 3 component (wherein Ln is one or more selected from the group consisting of La, Gd, Y, Yb, and Lu) is 15.0% to 65.0%
- RO component (wherein R is one or more selected from the group consisting of Mg, Ca, Sr and Ba) is 25.0% or less
- Rn 2 O component (wherein Rn is Li , One or more selected from the group consisting of Na, K) is 10.0% or less
- the optical glass according to any one of (1) to (7) (9) The optical glass according to any one of (1) to (8), wherein the abrasion degree is 200 or less.
- a glass having a high chemical durability (acid resistance) by a powder method and a low abrasion value while having a refractive index (n d ) and an Abbe number ( ⁇ d ) within desired ranges Obtainable.
- the optical glass according to the first aspect of the present invention contains, in mass%, a SiO 2 component of 10.0% to 40.0% and a La 2 O 3 component of 15.0% to 50.0%.
- the TiO 2 component containing less than 5.0% to 25% weight ratio (B 2 O 3 / SiO 2 ) is 1.0 or less, 1.78 to 1.95 of the refractive index (n d) And has an Abbe number ( ⁇ d ) of 25 to 45.
- the optical glass mainly composed of SiO 2 component and La 2 O 3 component has high acid resistance while having a refractive index (n d ) of 1.78 or more and an Abbe number ( ⁇ d ) of 25 or more and 45 or less. Glass is easily obtained.
- optical glass according to the first aspect of the present invention can be suitably used for applications that transmit visible light due to its high transmittance for visible light.
- the composition range of each component constituting the optical glass of the present invention is described below.
- the contents of the respective components are all expressed in mass% with respect to the total mass number of the oxide conversion composition.
- the “oxide equivalent composition” is based on the assumption that the oxide, composite salt, metal fluoride, etc. used as the raw material of the glass component of the present invention are all decomposed and changed into oxides during melting. It is the composition which described each component contained in glass by making the total mass number of production
- the SiO 2 component is an essential component as a glass-forming oxide in the optical glass of the present invention having high durability.
- the content of the SiO 2 component is preferably 10.0%, more preferably 15.0%, further preferably 20.0%, and further preferably 25.0%.
- the content of the SiO 2 component is 40.0% or less, a larger refractive index can be easily obtained, and deterioration of devitrification can be suppressed.
- the content of the SiO 2 component is preferably 40.0% or less, more preferably less than 37.0%, even more preferably less than 35.0%, and even more preferably less than 33.0%.
- SiO 2 component SiO 2 , K 2 SiF 6 , Na 2 SiF 6 , ZrSiO 4 or the like can be used as a raw material.
- the La 2 O 3 component is an essential component that increases the refractive index and Abbe number of the glass. Therefore, the content of the La 2 O 3 component is preferably 15.0% or more, more preferably more than 16.0%, more preferably more than 18.0%, still more preferably more than 20.0%.
- the content of the La 2 O 3 component is preferably 50.0% or less, more preferably less than 45.0%, and even more preferably less than 40.0%.
- La 2 O 3 component La 2 O 3 , La (NO 3 ) 3 .XH 2 O (X is an arbitrary integer) or the like can be used as a raw material.
- the TiO 2 component is an optional component that can enhance the stability by increasing the refractive index of the glass and lowering the liquidus temperature of the glass.
- the content of the TiO 2 component is preferably 5.0% or more, more preferably more than 6.0%, more preferably more than 7.0%, more preferably more than 8.0%, still more preferably 9. It may be more than 0%.
- the content of the TiO 2 component is set to less than 25.0%, devitrification due to excessive content of the TiO 2 component can be reduced, and the transmittance of the glass with respect to visible light (especially at a wavelength of 500 nm or less) is reduced. It can be suppressed.
- the content of the TiO 2 component is preferably less than 25.0%, more preferably less than 24.0%, even more preferably less than 21.0%, even more preferably less than 19.0%, and most preferably 15. 0% or less.
- TiO 2 component TiO 2 or the like can be used as a raw material.
- the ratio (mass ratio) of the content of the B 2 O 3 component to the content of the SiO 2 component is preferably 1.0 or less.
- the mass ratio B 2 O 3 / SiO 2 is preferably 1.0 or less, more preferably 0.98 or less, still more preferably 0.90 or less, still more preferably 0.80 or less, and even more preferably 0.70. The following.
- the ZnO component is an optional component that enhances the meltability of the raw material, promotes defoaming from the molten glass, and increases the stability of the glass. Moreover, it is also a component which can reduce coloring of glass by shortening melting time. It is also a component that can lower the glass transition point and improve chemical durability (acid resistance). Accordingly, the content of the ZnO component is preferably more than 0%, more preferably more than 1.0%, still more preferably more than 2.5%, still more preferably more than 4.5%, still more preferably more than 6.5%. More preferably, it may be more than 8.5%.
- the content of the ZnO component is preferably 30.0% or less, more preferably less than 28.0%, and even more preferably less than 25.0%.
- ZnO component ZnO, ZnF 2 or the like can be used as a raw material.
- the ZrO 2 component is an optional component that can increase the refractive index and Abbe number of the glass and can improve the devitrification resistance when it is contained in excess of 0%. Therefore, the content of the ZrO 2 component is preferably more than 0%, more preferably more than 1.0%, more preferably more than 3.0%, more preferably more than 5.0%, still more preferably 7.0%. It may be super. On the other hand, by setting the content of the ZrO 2 component to 20.0% or less, devitrification due to excessive inclusion of the ZrO 2 component can be reduced. Therefore, the content of the ZrO 2 component is preferably 20.0% or less, more preferably less than 18.0%, even more preferably less than 16.0%, and even more preferably less than 14.0%. As the ZrO 2 component, ZrO 2 , ZrF 4 or the like can be used as a raw material.
- the Al 2 O 3 component is an optional component that can improve the chemical durability (acid resistance) of the glass and improve the devitrification resistance of the molten glass when it contains more than 0%. Accordingly, the content of the Al 2 O 3 component is preferably more than 0%, more preferably more than 1.0%, still more preferably more than 2.5%, still more preferably more than 5.0%, still more preferably 7. It may be more than 5%. On the other hand, by setting the content of the Al 2 O 3 component to 20.0% or less, the liquidus temperature of the glass can be lowered to increase the devitrification resistance.
- the content of the Al 2 O 3 component is preferably 20.0% or less, more preferably less than 18.0%, still more preferably less than 16.5%, and even more preferably less than 15.0%.
- Al 2 O 3 component Al 2 O 3 , Al (OH) 3 , AlF 3 or the like can be used as a raw material.
- the glass material cost can be suppressed while maintaining a high refractive index and a high Abbe number, and the specific gravity of the glass can be reduced more than other rare earth components. It is an optional component.
- the content of the Y 2 O 3 component 25.0% or less, a decrease in the refractive index of the glass can be suppressed, and the stability of the glass can be enhanced. Moreover, deterioration of the meltability of the glass raw material can be suppressed. Therefore, the content of the Y 2 O 3 component is preferably 25.0% or less, more preferably less than 23.0%, more preferably less than 20.0%, and most preferably 18.0% or less.
- Y 2 O 3 component Y 2 O 3 , YF 3 or the like can be used as a raw material.
- the B 2 O 3 component is an optional component as a glass-forming oxide that lowers the liquidus temperature while increasing devitrification resistance when it is contained in an amount of more than 0%.
- the content of the B 2 O 3 component is preferably 20.0% or less, more preferably less than 16.0%, even more preferably less than 14.0%, still more preferably less than 13.0%, still more preferably It is less than 12.0%, more preferably less than 10.0%.
- H 3 BO 3 , Na 2 B 4 O 7 , Na 2 B 4 O 7 .10H 2 O, BPO 4 or the like can be used as a raw material.
- the Nb 2 O 5 component is an optional component that can increase the refractive index of glass and increase the devitrification resistance by lowering the liquidus temperature of the glass when it contains more than 0%. Accordingly, the content of the Nb 2 O 5 component is preferably more than 0%, more preferably more than 1.0%, still more preferably more than 3.0%, still more preferably more than 4.5%, still more preferably 6. It may be more than 5%. On the other hand, by setting the content of the Nb 2 O 5 component to less than 15.0%, devitrification due to excessive content of the Nb 2 O 5 component can be reduced, and visible light of the glass (particularly, a wavelength of 500 nm or less) Decrease in the transmittance with respect to can be suppressed.
- the content of the Nb 2 O 5 component is preferably less than 15.0%, more preferably less than 13.0%, still more preferably less than 10.0%, and even more preferably less than 7.0%.
- Nb 2 O 5 component Nb 2 O 5 or the like can be used as a raw material.
- WO 3 component is an optional component that can increase the refractive index, lower the glass transition point, and increase the devitrification resistance while reducing the coloring of the glass by other high refractive index components when it contains more than 0%. It is. On the other hand, the material cost of glass can be suppressed by making the content of the WO 3 component less than 10.0%. In addition, the visible light transmittance can be increased by reducing the coloring of the glass by the WO 3 component. Accordingly, the content of the WO 3 component is preferably less than 10.0%, more preferably less than 6.0%, more preferably less than 4.5%, still more preferably less than 3.0%, and even more preferably 1.%. It is less than 0%, more preferably less than 0.5%, more preferably less than 0.1%. As the WO 3 component, WO 3 or the like can be used as a raw material.
- the Gd 2 O 3 component is an optional component that can increase the refractive index of the glass when it exceeds 0%.
- the Gd 2 O 3 component has a high raw material price, and if its content is large, the production cost becomes high. Further, with the Gd 2 O 3 less 25.0% the content of components, it is suppressed an increase in the Abbe number of the glass. Therefore, the content of the Gd 2 O 3 component is preferably 25.0% or less, more preferably less than 23.0%, and even more preferably less than 20.0%.
- Gd 2 O 3 component Gd 2 O 3 , GdF 3 or the like can be used as a raw material.
- the Yb 2 O 3 component is an optional component that can increase the refractive index of the glass when it exceeds 0%.
- the Yb 2 O 3 component has a high raw material price, and if its content is large, the production cost becomes high. Further, by less than 5.0% the content of Yb 2 O 3 component is suppressed an increase in the Abbe number of the glass. Accordingly, the content of the Yb 2 O 3 component is preferably less than 5.0%, more preferably less than 3.0%, even more preferably less than 2.0%, still more preferably less than 0.5%, and still more preferably. Less than 0.1%. As the Yb 2 O 3 component, Yb 2 O 3 or the like can be used as a raw material.
- the Ta 2 O 5 component is an optional component that can increase the refractive index of the glass and increase the devitrification resistance when it exceeds 0%.
- the Ta 2 O 5 component has a high raw material price, and if its content is large, the production cost becomes high. Further, by setting the content of Ta 2 O 5 component to less than 5.0%, melting temperature of the material is lowered, the energy required for the raw material melting is reduced, it is also reduced the production cost of optical glass. Accordingly, the content of the Ta 2 O 5 component is preferably less than 5.0%, more preferably less than 3.0%, even more preferably less than 1.0%, still more preferably less than 0.5%, and still more preferably. Less than 0.1%. Further, in the viewpoint of reducing the material cost, it is most preferably contains no Ta 2 O 5 component. As the Ta 2 O 5 component, Ta 2 O 5 or the like can be used as a raw material.
- the MgO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of glass when it is contained in excess of 0%.
- the content of the MgO component is preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, and still more preferably less than 1.0%.
- MgO component MgCO 3 , MgF 2 or the like can be used as a raw material.
- the CaO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of the glass when it contains more than 0%.
- the content of the CaO component is preferably 35.0% or less, more preferably less than 30.0%, further preferably less than 25.0%, more preferably less than 22.0%, and still more preferably 20.0%. %.
- the CaO component CaCO 3 , CaF 2 or the like can be used as a raw material.
- the SrO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of the glass when it contains more than 0%.
- the content of the SrO component is preferably 35.0% or less, more preferably less than 30.0%, even more preferably less than 25.0%, still more preferably less than 22.0%, still more preferably 20.0%. %.
- Sr (NO 3 ) 2 , SrF 2 or the like can be used as a raw material.
- the BaO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of the glass when it is contained in excess of 0%.
- the content of the BaO component is preferably 35.0% or less, more preferably less than 30.0%, even more preferably less than 29.0%, still more preferably less than 25.0%, still more preferably 22.0%. %, More preferably less than 20.0%.
- BaO component BaCO 3 , Ba (NO 3 ) 2 , BaF 2 or the like can be used as a raw material.
- the Li 2 O component is an optional component that can improve the meltability of the glass and lower the glass transition point when it exceeds 0%.
- the content of the Li 2 O component is preferably 10.0% or less, more preferably less than 5.0%, even more preferably less than 3.0%, still more preferably less than 1.0%, still more preferably 0. Less than 5%, more preferably less than 0.1%.
- Li 2 O component Li 2 CO 3 , LiNO 3 , Li 2 CO 3 or the like can be used as a raw material.
- the Na 2 O component is an optional component that can improve the meltability of the glass and lower the glass transition point when it contains more than 0%.
- the content of the Na 2 O component is 10.0% or less, the refractive index of the glass is hardly lowered and the devitrification of the glass can be reduced.
- the content of the Na 2 O component is preferably 10.0% or less, more preferably less than 6.0%, even more preferably less than 3.0%, still more preferably less than 1.0%, and still more preferably. Less than 0.5%, more preferably less than 0.1%.
- Na 2 O component Na 2 CO 3 , NaNO 3 , NaF, Na 2 SiF 6 or the like can be used as a raw material.
- the K 2 O component is an optional component that can improve the meltability of the glass and lower the glass transition point when it contains more than 0%.
- the content of the K 2 O component is preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, still more preferably less than 1.0%, and still more preferably. Less than 0.5%, more preferably less than 0.1%.
- K 2 O component K 2 CO 3 , KNO 3 , KF, KHF 2 , K 2 SiF 6 or the like can be used as a raw material.
- the P 2 O 5 component is an optional component that can reduce the liquidus temperature of the glass and increase the devitrification resistance when it contains more than 0%.
- the content of the P 2 O 5 component is preferably 10.0% or less, more preferably less than 5.0%, and even more preferably less than 3.0%.
- Al (PO 3 ) 3 , Ca (PO 3 ) 2 , Ba (PO 3 ) 2 , BPO 4 , H 3 PO 4 or the like can be used as a raw material.
- the GeO 2 component is an optional component that can increase the refractive index of the glass and improve the devitrification resistance when it contains more than 0%.
- the raw material price of GeO 2 is high, and the production cost increases when the content is large. Therefore, the content of the GeO 2 component is preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, still more preferably less than 1.0%, still more preferably 0.00. Less than 1%. From the viewpoint of reducing the material cost, the GeO 2 component may not be contained.
- the GeO 2 component GeO 2 or the like can be used as a raw material.
- the Ga 2 O 3 component is an optional component that can improve the chemical durability (acid resistance) of the glass and improve the devitrification resistance of the molten glass when it contains more than 0%.
- the content of the Ga 2 O 3 component is preferably 10.0% or less, more preferably less than 8.0%, still more preferably less than 5.0%, and even more preferably less than 3.0%.
- Ga 2 O 3 component Ga 2 O 3 , Ga (OH) 3 or the like can be used as a raw material.
- the Bi 2 O 3 component is an optional component that can increase the refractive index and lower the glass transition point when it exceeds 0%.
- the content of the Bi 2 O 3 component is preferably 10.0% or less, more preferably less than 8.0%, still more preferably less than 5.0%, still more preferably less than 3.0%, and still more preferably. Less than 1.0%, most preferably not contained.
- Bi 2 O 3 component Bi 2 O 3 or the like can be used as a raw material.
- the TeO 2 component is an optional component that can increase the refractive index and lower the glass transition point when it is contained in excess of 0%.
- TeO 2 has a problem that it can be alloyed with platinum when melting a glass raw material in a crucible made of platinum or a melting tank in which a portion in contact with molten glass is formed of platinum. Therefore, the content of the TeO 2 component is preferably 10.0% or less, more preferably less than 8.0%, even more preferably less than 5.0%, still more preferably less than 3.0%, and still more preferably 1.%. Less than 0%. TeO 2 component can use TeO 2 or the like as a raw material.
- the CsO 2 component is an optional component that can improve the meltability of the glass and lower the glass transition point when it exceeds 0%. On the other hand, it is possible to make it difficult to lower the refractive index of the glass and reduce the devitrification of the glass. Therefore, the content of the CsO 2 component is preferably 3.0% or less, more preferably less than 2.0%, further preferably less than 1.0%, more preferably less than 0.1%, most preferably not contained. To do. As the CsO 2 component, Cs 2 CO 3 , CsNO 3 or the like can be used as a raw material.
- the SnO 2 component is an optional component that can be refined by reducing the oxidation of the molten glass and can increase the visible light transmittance of the glass.
- the content of the SnO 2 component is 3.0% or less, the coloring of the glass due to the reduction of the molten glass and the devitrification of the glass can be reduced.
- the alloying of the SnO 2 component and the melting equipment especially a noble metal such as Pt
- the content of the SnO 2 component is preferably 3.0% or less, more preferably less than 1.0%, further preferably less than 0.5%, and further preferably less than 0.1%.
- SnO, SnO 2 , SnF 2 , SnF 4 or the like can be used as a raw material.
- the Sb 2 O 3 component is an optional component that can degas the molten glass when it contains more than 0%.
- the content of the Sb 2 O 3 component is preferably 3.0% or less, more preferably less than 2.0%, still more preferably less than 1.0%, and still more preferably less than 0.5%.
- Sb 2 O 3 component Sb 2 O 3 , Sb 2 O 5 , Na 2 H 2 Sb 2 O 7 .5H 2 O, or the like can be used as a raw material.
- components defoamed fining glass is not limited to the above Sb 2 O 3 component, a known refining agents in the field of glass production, it is possible to use a defoamer or a combination thereof.
- the F component is an optional component that can increase the Abbe number of the glass, lower the glass transition point, and improve the devitrification resistance when it contains more than 0%.
- the content of the F component that is, the total amount of fluoride substituted for one or more of the above-mentioned one or more oxides of each metal element exceeds 15.0%
- F Since the volatilization amount of the component increases, it becomes difficult to obtain a stable optical constant, and it becomes difficult to obtain a homogeneous glass.
- the Abbe number rises more than necessary. Therefore, the content of the component F is preferably 15.0% or less, more preferably less than 10.0%, still more preferably less than 5.0%, and still more preferably less than 3.0%.
- the F component can be contained in the glass by using, for example, ZrF 4 , AlF 3 , NaF, CaF 2 or the like as a raw material.
- the total amount (mass sum) of the B 2 O 3 component and the Nb 2 O 5 component is preferably less than 20.0%.
- acid resistance can be improved while the Abbe number ( ⁇ d ) is within a desired range.
- the mass sum B 2 O 3 + Nb 2 O 5 is preferably less than 20.0%, more preferably less than 18.0%, even more preferably less than 15.0%, even more preferably less than 13.0%, Preferably it is less than 12.0%, more preferably less than 11.0%.
- devitrification resistance can be improved by making the total amount (mass sum) of the B 2 O 3 component and the Nb 2 O 5 component exceed 0%. Therefore, the mass sum B 2 O 3 + Nb 2 O 5 is preferably more than 0%, more preferably more than 3.0%, more preferably more than 5.0%, more preferably more than 6.0%.
- the total amount (mass sum) of the TiO 2 component and the ZrO 2 component is preferably less than 35.0%. Thereby, it can reduce that an Abbe number ((nu) d ) becomes low. Accordingly, the mass sum TiO 2 + ZrO 2 is preferably less than 35.0%, more preferably 33.0% or less, still more preferably less than 30.0%, still more preferably less than 28.0%, and even more preferably 25. The upper limit is 0% or less. On the other hand, when the total amount (mass sum) of the TiO 2 component and the ZrO 2 component exceeds 0%, the refractive index of the glass can be increased.
- the mass sum TiO 2 + ZrO 2 is preferably more than 0%, more preferably more than 5.0%, more preferably more than 8.0%, more preferably more than 10.0%, still more preferably more than 13.0%. More preferably, it may be more than 15.0%.
- the total amount (mass sum) of the ZrO 2 component, the Nb 2 O 5 component, the WO 3 component, and the ZnO component is preferably 5.0% or more.
- the Abbe number ( ⁇ d ) can be adjusted within a desired range.
- the mass sum ZrO 2 + Nb 2 O 5 + WO 3 + ZnO is preferably 5.0% or more, more preferably more than 7.0%, more preferably more than 9.0%, still more preferably more than 11.0%. More preferably, it may be more than 13.0%.
- the mass sum ZrO 2 + Nb 2 O 5 + WO 3 + ZnO is preferably 60.0% or less, more preferably less than 55.0%, more preferably less than 50.0%, more preferably less than 48.0%. good.
- the sum (mass sum) of contents of Ln 2 O 3 components is 15.0% or more and 65.0 % Or less is preferable.
- Ln is one or more selected from the group consisting of La, Gd, Y, Yb, and Lu
- the mass sum of the Ln 2 O 3 component is preferably 15.0% or more, more preferably more than 16.0%, still more preferably more than 18.0%, and even more preferably more than 20.0%.
- the mass sum of the Ln 2 O 3 component is preferably 65.0% or less, more preferably less than 60.0%, further preferably less than 55.0%, and further preferably less than 50.0%.
- the content sum (mass sum) of RO components (wherein R is one or more selected from the group consisting of Mg, Ca, Sr, and Ba) is preferably 35.0% or less. Thereby, the fall of a refractive index can be suppressed and stability of glass can be improved. Therefore, the mass sum of the RO component is preferably 35.0% or less, more preferably less than 33.0%, more preferably less than 30.0%, and more preferably less than 29.0%.
- the sum (mass sum) of the contents of the Rn 2 O component (wherein Rn is one or more selected from the group consisting of Li, Na and K) is preferably 10.0% or less.
- Rn is one or more selected from the group consisting of Li, Na and K
- the mass sum of the Rn 2 O component is preferably 10.0% or less, more preferably less than 8.0%, still more preferably less than 5.0%, and even more preferably less than 3.0%.
- the optical glass of the second aspect of the present invention contains, in mass%, a SiO 2 component of 10.0% to 50.0% and a La 2 O 3 component of 15.0% to 60.0%, A TiO 2 component is contained in an amount of 0.0% to less than 15%, a mass ratio (B 2 O 3 / SiO 2 ) is 1.0 or less, and a refractive index (n d ) of 1.60 or more and 1.85 or less. And an Abbe number ( ⁇ d ) of 33 to 62.
- Optical glass mainly composed of SiO 2 component and La 2 O 3 component has high acid resistance while having a refractive index (n d ) of 1.60 or more and an Abbe number ( ⁇ d ) of 33 or more and 62 or less. Glass is easily obtained.
- optical glass according to the second aspect of the present invention can be suitably used for applications that transmit visible light due to its high transmittance for visible light.
- the SiO 2 component is an essential component as a glass-forming oxide in the optical glass of the present invention having high durability.
- the content of the SiO 2 component is preferably 10.0%, more preferably 15.0%, further preferably 20.0%, and further preferably 25.0%.
- the content of the SiO 2 component 50.0% or less by making the content of the SiO 2 component 50.0% or less, a larger refractive index can be easily obtained, and deterioration of devitrification can be suppressed.
- the content of the SiO 2 component is preferably 50.0% or less, more preferably less than 47.0%, even more preferably less than 45.0%, and even more preferably less than 43.0%.
- SiO 2 component SiO 2 , K 2 SiF 6 , Na 2 SiF 6 , ZrSiO 4 or the like can be used as a raw material.
- the La 2 O 3 component is an essential component that increases the refractive index and Abbe number of the glass. Therefore, the content of the La 2 O 3 component is preferably 15.0% or more, more preferably more than 16.0%, more preferably more than 18.0%, still more preferably more than 20.0%. On the other hand, by setting the content of the La 2 O 3 component to 60.0% or less, devitrification can be reduced by increasing the stability of the glass, and the Abbe number can be prevented from rising more than necessary. Moreover, the meltability of the glass raw material can be enhanced. Therefore, the content of the La 2 O 3 component is preferably 60.0% or less, more preferably less than 58.0%, and even more preferably less than 55.0%. As the La 2 O 3 component, La 2 O 3 , La (NO 3 ) 3 .XH 2 O (X is an arbitrary integer) or the like can be used as a raw material.
- the TiO 2 component When the TiO 2 component is contained in an amount of more than 0%, it is an optional component that can increase the refractive index of the glass and increase the stability by lowering the liquidus temperature of the glass.
- the content of the TiO 2 component is preferably less than 15.0%, more preferably less than 13.0%, even more preferably less than 11.0%, still more preferably less than 10.0%, and even more preferably 9. Less than 0%.
- TiO 2 component TiO 2 or the like can be used as a raw material.
- the ratio (mass ratio) of the content of the B 2 O 3 component to the content of the SiO 2 component is preferably 1.0 or less.
- the mass ratio B 2 O 3 / SiO 2 is preferably 1.0 or less, more preferably 0.98 or less, still more preferably 0.90 or less, still more preferably 0.80 or less, and even more preferably 0.70. The following.
- the ZnO component is an optional component that enhances the meltability of the raw material, promotes defoaming from the molten glass, and increases the stability of the glass. Moreover, it is also a component which can reduce coloring of glass by shortening melting time. It is also a component that can lower the glass transition point and improve chemical durability. Accordingly, the content of the ZnO component is preferably more than 0%, more preferably more than 1.0%, still more preferably more than 2.5%, still more preferably more than 4.5%, still more preferably more than 6.5%. More preferably, it may be more than 8.5%.
- the content of the ZnO component 35.0% or less, a decrease in the refractive index of the glass can be suppressed, and devitrification due to an excessive decrease in viscosity can be reduced. Therefore, the content of the ZnO component is preferably 35.0% or less, more preferably less than 33.0%, still more preferably less than 31.0%, and even more preferably less than 29.0%.
- the ZnO component ZnO, ZnF 2 or the like can be used as a raw material.
- the ZrO 2 component is an optional component that can increase the refractive index and Abbe number of the glass and can improve the devitrification resistance when it is contained in excess of 0%. Therefore, the content of the ZrO 2 component may be preferably more than 0%, more preferably more than 1.0%, and still more preferably more than 2.0%. On the other hand, by setting the content of the ZrO 2 component to 20.0% or less, devitrification due to excessive inclusion of the ZrO 2 component can be reduced. Therefore, the content of the ZrO 2 component is preferably 20.0% or less, more preferably less than 18.0%, even more preferably less than 16.0%, even more preferably less than 14.0%, and most preferably 10. 0% or less. As the ZrO 2 component, ZrO 2 , ZrF 4 or the like can be used as a raw material.
- the Al 2 O 3 component is an optional component that can improve the chemical durability of the glass and improve the devitrification resistance of the molten glass when it contains more than 0%. Accordingly, the content of the Al 2 O 3 component is preferably more than 0%, more preferably more than 1.0%, still more preferably more than 2.5%, still more preferably more than 5.0%, still more preferably 7. It may be more than 5%. On the other hand, by setting the content of the Al 2 O 3 component to 20.0% or less, the liquidus temperature of the glass can be lowered to increase the devitrification resistance.
- the content of the Al 2 O 3 component is preferably 20.0% or less, more preferably less than 18.0%, even more preferably less than 16.5%, even more preferably less than 15.0%, most preferably 13.0% or less.
- Al 2 O 3 component Al 2 O 3 , Al (OH) 3 , AlF 3 or the like can be used as a raw material.
- the Y 2 O 3 component When the Y 2 O 3 component is contained in an amount exceeding 0%, the glass material cost can be suppressed while maintaining a high refractive index and a high Abbe number, and the specific gravity of the glass can be reduced more than other rare earth components. It is an optional component. Therefore, the content of the Y 2 O 3 component is preferably more than 0%, more preferably more than 1.0%, and even more preferably more than 3.0%. On the other hand, by making the content of the Y 2 O 3 component 25.0% or less, a decrease in the refractive index of the glass can be suppressed, and the stability of the glass can be enhanced. Moreover, deterioration of the meltability of the glass raw material can be suppressed.
- the content of the Y 2 O 3 component is preferably 25.0% or less, more preferably less than 23.0%, more preferably less than 20.0%.
- Y 2 O 3 component Y 2 O 3 , YF 3 or the like can be used as a raw material.
- the B 2 O 3 component is an optional component as a glass-forming oxide that lowers the liquidus temperature while increasing devitrification resistance when it is contained in an amount of more than 0%.
- the content of the B 2 O 3 component is preferably 20.0% or less, more preferably less than 16.0%, still more preferably less than 13.0%, and even more preferably less than 10.0%.
- H 3 BO 3 , Na 2 B 4 O 7 , Na 2 B 4 O 7 .10H 2 O, BPO 4 or the like can be used as a raw material.
- the Nb 2 O 5 component is an optional component that can increase the refractive index of glass and increase the devitrification resistance by lowering the liquidus temperature of the glass when it contains more than 0%.
- the content of the Nb 2 O 5 component is set to less than 15.0%, devitrification due to excessive content of the Nb 2 O 5 component can be reduced, and visible light of the glass (particularly, a wavelength of 500 nm or less) Decrease in the transmittance with respect to can be suppressed. Further, this can suppress a decrease in Abbe number.
- the content of the Nb 2 O 5 component is preferably less than 15.0%, more preferably less than 13.0%, more preferably less than 9.0%, still more preferably less than 7.0%, and still more preferably Less than 5.0%.
- Nb 2 O 5 component Nb 2 O 5 or the like can be used as a raw material.
- WO 3 component is an optional component that can increase the refractive index, lower the glass transition point, and increase the devitrification resistance while reducing the coloring of the glass by other high refractive index components when it contains more than 0%. It is. On the other hand, the material cost of glass can be suppressed by making the content of the WO 3 component less than 10.0%. In addition, the visible light transmittance can be increased by reducing the coloring of the glass by the WO 3 component. Accordingly, the content of the WO 3 component is preferably less than 10.0%, more preferably less than 5.0%, even more preferably less than 3.0%, still more preferably less than 1.0%, and still more preferably 0.00. It is less than 5%, more preferably less than 0.1%. As the WO 3 component, WO 3 or the like can be used as a raw material.
- the Gd 2 O 3 component is an optional component that can increase the refractive index of the glass when it exceeds 0%.
- the Gd 2 O 3 component has a high raw material price, and if its content is large, the production cost becomes high. Further, with the Gd 2 O 3 less 25.0% the content of components, it is suppressed an increase in the Abbe number of the glass. Therefore, the content of the Gd 2 O 3 component is preferably 25.0% or less, more preferably less than 23.0%, and even more preferably less than 20.0%.
- Gd 2 O 3 component Gd 2 O 3 , GdF 3 or the like can be used as a raw material.
- the Yb 2 O 3 component is an optional component that can increase the refractive index of the glass when it exceeds 0%.
- the Yb 2 O 3 component has a high raw material price, and if its content is large, the production cost becomes high. Further, by less than 5.0% the content of Yb 2 O 3 component is suppressed an increase in the Abbe number of the glass. Accordingly, the content of the Yb 2 O 3 component is preferably less than 5.0%, more preferably less than 3.0%, even more preferably less than 2.0%, still more preferably less than 0.5%, and still more preferably. Less than 0.1%. As the Yb 2 O 3 component, Yb 2 O 3 or the like can be used as a raw material.
- the Ta 2 O 5 component is an optional component that can increase the refractive index of the glass and increase the devitrification resistance when it exceeds 0%.
- the Ta 2 O 5 component has a high raw material price, and if its content is large, the production cost becomes high. Further, by setting the content of Ta 2 O 5 component to less than 5.0%, melting temperature of the material is lowered, the energy required for the raw material melting is reduced, it is also reduced the production cost of optical glass. Accordingly, the content of the Ta 2 O 5 component is preferably less than 5.0%, more preferably less than 3.0%, even more preferably less than 1.0%, still more preferably less than 0.5%, and still more preferably. Less than 0.1%. Further, in the viewpoint of reducing the material cost, it is most preferably contains no Ta 2 O 5 component. As the Ta 2 O 5 component, Ta 2 O 5 or the like can be used as a raw material.
- the MgO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of glass when it is contained in excess of 0%.
- the content of the MgO component is preferably 15.0% or less, more preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, and still more preferably 1.0. %.
- MgO component MgCO 3 , MgF 2 or the like can be used as a raw material.
- the CaO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of the glass when it contains more than 0%.
- the content of the CaO component is preferably 15.0% or less, more preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, still more preferably 1.0. %.
- the CaO component CaCO 3 , CaF 2 or the like can be used as a raw material.
- the SrO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of the glass when it contains more than 0%.
- the content of the SrO component is preferably 15.0% or less, more preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, still more preferably 1.0%. Less than.
- Sr (NO 3 ) 2 , SrF 2 or the like can be used as a raw material.
- the BaO component is an optional component that can adjust the refractive index, meltability, and devitrification resistance of the glass when it is contained in excess of 0%.
- the content of the BaO component is preferably 15.0% or less, more preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, and still more preferably 1.0. %.
- BaO component BaCO 3 , Ba (NO 3 ) 2 , BaF 2 or the like can be used as a raw material.
- the Li 2 O component is an optional component that can improve the meltability of the glass and lower the glass transition point when it exceeds 0%.
- the content of the Li 2 O component is preferably 10.0% or less, more preferably less than 5.0%, even more preferably less than 3.0%, still more preferably less than 1.0%, still more preferably 0. Less than 5%, more preferably less than 0.1%.
- Li 2 O component Li 2 CO 3 , LiNO 3 , Li 2 CO 3 or the like can be used as a raw material.
- the Na 2 O component is an optional component that can improve the meltability of the glass and lower the glass transition point when it contains more than 0%.
- the content of the Na 2 O component is 10.0% or less, the refractive index of the glass is hardly lowered and the devitrification of the glass can be reduced.
- the content of the Na 2 O component is preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, still more preferably less than 1.0%, and still more preferably. Less than 0.5%, more preferably less than 0.1%.
- Na 2 O component Na 2 CO 3 , NaNO 3 , NaF, Na 2 SiF 6 or the like can be used as a raw material.
- the K 2 O component is an optional component that can improve the meltability of the glass and lower the glass transition point when it contains more than 0%.
- the content of the K 2 O component is preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, still more preferably less than 1.0%, and still more preferably. Less than 0.5%, more preferably less than 0.1%.
- K 2 O component K 2 CO 3 , KNO 3 , KF, KHF 2 , K 2 SiF 6 or the like can be used as a raw material.
- the P 2 O 5 component is an optional component that can reduce the liquidus temperature of the glass and increase the devitrification resistance when it contains more than 0%.
- the content of the P 2 O 5 component is preferably 10.0% or less, more preferably less than 5.0%, and even more preferably less than 3.0%.
- Al (PO 3 ) 3 , Ca (PO 3 ) 2 , Ba (PO 3 ) 2 , BPO 4 , H 3 PO 4 or the like can be used as a raw material.
- the GeO 2 component is an optional component that can increase the refractive index of the glass and improve the devitrification resistance when it contains more than 0%.
- the raw material price of GeO 2 is high, and the production cost increases when the content is large. Therefore, the content of the GeO 2 component is preferably 10.0% or less, more preferably less than 5.0%, still more preferably less than 3.0%, still more preferably less than 1.0%, still more preferably 0.00. Less than 1%. From the viewpoint of reducing the material cost, the GeO 2 component may not be contained.
- the GeO 2 component GeO 2 or the like can be used as a raw material.
- the Ga 2 O 3 component is an optional component that can improve the chemical durability of the glass and improve the devitrification resistance of the molten glass when it contains more than 0%.
- the content of the Ga 2 O 3 component is preferably 10.0% or less, more preferably less than 8.0%, still more preferably less than 5.0%, and even more preferably less than 3.0%.
- Ga 2 O 3 component Ga 2 O 3 , Ga (OH) 3 or the like can be used as a raw material.
- the Bi 2 O 3 component is an optional component that can increase the refractive index and lower the glass transition point when it exceeds 0%.
- the content of the Bi 2 O 3 component is preferably 10.0% or less, more preferably less than 8.0%, still more preferably less than 5.0%, still more preferably less than 3.0%, and still more preferably. Less than 1.0%, most preferably not contained.
- Bi 2 O 3 component Bi 2 O 3 or the like can be used as a raw material.
- the TeO 2 component is an optional component that can increase the refractive index and lower the glass transition point when it is contained in excess of 0%.
- TeO 2 has a problem that it can be alloyed with platinum when melting a glass raw material in a crucible made of platinum or a melting tank in which a portion in contact with molten glass is formed of platinum. Therefore, the content of the TeO 2 component is preferably 10.0% or less, more preferably less than 8.0%, even more preferably less than 5.0%, still more preferably less than 3.0%, and still more preferably 1.%. Less than 0%. TeO 2 component can use TeO 2 or the like as a raw material.
- the CsO 2 component is an optional component that can improve the meltability of the glass and lower the glass transition point when it exceeds 0%. On the other hand, it is possible to make it difficult to lower the refractive index of the glass and reduce the devitrification of the glass. Therefore, the content of the CsO 2 component is preferably 3.0% or less, more preferably less than 2.0%, further preferably less than 1.0%, more preferably less than 0.1%, most preferably not contained. To do. As the CsO 2 component, Cs 2 CO 3 , CsNO 3 or the like can be used as a raw material.
- the SnO 2 component is an optional component that can be refined by reducing the oxidation of the molten glass and can increase the visible light transmittance of the glass.
- the content of the SnO 2 component is 3.0% or less, the coloring of the glass due to the reduction of the molten glass and the devitrification of the glass can be reduced.
- the alloying of the SnO 2 component and the melting equipment especially a noble metal such as Pt
- the content of the SnO 2 component is preferably 3.0% or less, more preferably less than 1.0%, further preferably less than 0.5%, and further preferably less than 0.1%.
- SnO, SnO 2 , SnF 2 , SnF 4 or the like can be used as a raw material.
- the Sb 2 O 3 component is an optional component that can degas the molten glass when it contains more than 0%.
- the content of the Sb 2 O 3 component is preferably 3.0% or less, more preferably less than 2.0%, still more preferably less than 1.0%, and still more preferably less than 0.5%.
- Sb 2 O 3 component Sb 2 O 3 , Sb 2 O 5 , Na 2 H 2 Sb 2 O 7 .5H 2 O, or the like can be used as a raw material.
- components defoamed fining glass is not limited to the above Sb 2 O 3 component, a known refining agents in the field of glass production, it is possible to use a defoamer or a combination thereof.
- the F component is an optional component that can increase the Abbe number of the glass, lower the glass transition point, and improve the devitrification resistance when it contains more than 0%.
- the content of the F component that is, the total amount of fluoride substituted for one or more of the above-mentioned one or more oxides of each metal element exceeds 15.0%
- F Since the volatilization amount of the component increases, it becomes difficult to obtain a stable optical constant, and it becomes difficult to obtain a homogeneous glass.
- the Abbe number rises more than necessary. Therefore, the content of the component F is preferably 15.0% or less, more preferably less than 10.0%, still more preferably less than 5.0%, and still more preferably less than 3.0%.
- the F component can be contained in the glass by using, for example, ZrF 4 , AlF 3 , NaF, CaF 2 or the like as a raw material.
- the total amount (mass sum) of the B 2 O 3 component and the Nb 2 O 5 component is preferably less than 20.0%.
- acid resistance can be improved while the Abbe number ( ⁇ d ) is within a desired range.
- the mass sum B 2 O 3 + Nb 2 O 5 is preferably less than 20.0%, more preferably less than 18.0%, even more preferably less than 15.0%, even more preferably less than 13.0%, Preferably it is less than 12.0%, more preferably less than 11.0%.
- the sum (mass sum) of contents of Ln 2 O 3 components is 15.0% or more and 65.0 % Or less is preferable.
- Ln is one or more selected from the group consisting of La, Gd, Y, Yb, and Lu
- the mass sum of the Ln 2 O 3 component is preferably 15.0% or more, more preferably more than 16.0%, still more preferably more than 18.0%, and even more preferably more than 20.0%.
- the mass sum of the Ln 2 O 3 component is preferably 65.0% or less, more preferably less than 60.0%, further preferably less than 55.0%, and further preferably less than 50.0%.
- the content sum (mass sum) of RO components (wherein R is one or more selected from the group consisting of Mg, Ca, Sr, and Ba) is preferably 25.0% or less. Thereby, the fall of a refractive index can be suppressed and stability of glass can be improved. Therefore, the mass sum of the RO component is preferably 25.0% or less, more preferably less than 20.0%, more preferably less than 15.0%, more preferably less than 10.0%.
- the sum (mass sum) of the contents of the Rn 2 O component (wherein Rn is one or more selected from the group consisting of Li, Na and K) is preferably 10.0% or less.
- Rn is one or more selected from the group consisting of Li, Na and K
- the mass sum of the Rn 2 O component is preferably 10.0% or less, more preferably less than 8.0%, still more preferably less than 5.0%, and even more preferably less than 3.0%.
- each transition metal component such as V, Cr, Mn, Fe, Co, Ni, Cu, Ag and Mo, excluding Ti, Zr, Nb, W, La, Gd, Y, Yb, and Lu, is independent of each other. Or, even when it is contained in a small amount in combination, the glass is colored and has the property of causing absorption at a specific wavelength in the visible range. .
- lead compounds such as PbO and arsenic compounds such as As 2 O 3 are components with high environmental loads, it is desirable that they are not substantially contained, that is, not contained at all except for inevitable mixing.
- each component of Th, Cd, Tl, Os, Be, and Se has tended to be refrained from being used as a harmful chemical material in recent years, and not only in the glass manufacturing process, but also in the processing process and disposal after commercialization. Until then, environmental measures are required. Therefore, when importance is placed on the environmental impact, it is preferable that these are not substantially contained.
- the optical glass of the present invention is produced, for example, as follows. That is, the above raw materials are uniformly mixed so that each component is within a predetermined content range, the prepared mixture is put into a platinum crucible, and 1100 to 1550 ° C. in an electric furnace depending on the difficulty of melting the glass raw material. It is produced by melting in a temperature range of 2 to 5 hours, homogenizing with stirring, lowering to an appropriate temperature, casting into a mold, and slow cooling.
- the optical glass according to the first aspect of the present invention preferably has a high refractive index and a high Abbe number (low dispersion).
- the refractive index (n d ) of the optical glass of the present invention is preferably 1.78, more preferably 1.79, and even more preferably 1.80.
- the refractive index (n d ) is preferably 1.95, more preferably 1.93, and even more preferably 1.90.
- the Abbe number ( ⁇ d ) of the optical glass of the present invention is preferably 25, more preferably 27, and still more preferably 29.
- the Abbe number ( ⁇ d ) is preferably 45, more preferably 43, and still more preferably 41.
- the optical glass according to the second aspect of the present invention preferably has a high refractive index and a high Abbe number (low dispersion).
- the refractive index (n d ) of the optical glass of the present invention is preferably 1.60, more preferably 1.63, and still more preferably 1.68.
- the refractive index (n d ) is preferably 1.85, more preferably 1.84.
- the Abbe number ( ⁇ d ) of the optical glass of the present invention is preferably 33, more preferably 35, and still more preferably 37.
- the Abbe number ( ⁇ d ) is preferably 62, more preferably 57, and still more preferably 55.
- the optical glass of the present invention is useful in optical design. Particularly when an optical system is configured, the optical system can be downsized while achieving high imaging characteristics and the like. The degree of freedom can be expanded.
- the optical glass of the present invention preferably has high acid resistance.
- the chemical durability (acid resistance) of the glass powder method according to JOGIS06-1999 is preferably 1 to 3 grade, more preferably 1 to 2 grade, and most preferably 1 grade.
- “acid resistance” means durability against erosion of glass by acid, and this acid resistance is measured according to the Japan Optical Glass Industry Association Standard “Method for Measuring Chemical Durability of Optical Glass” JOGIS06-1999. Can do.
- the chemical durability (acid resistance) by the powder method is 1 to 3” means that the chemical durability (acid resistance) performed according to JOGIS06-1999 is the mass of the sample before and after the measurement. It means a weight loss rate of less than 0.65% by mass.
- “Class 1” of chemical durability (acid resistance) means that the weight loss rate of the sample before and after measurement is less than 0.20% by mass
- “Class 2” means weight loss of the sample before and after measurement.
- the rate is 0.20% by mass or more and less than 0.35% by mass
- “Class 3” indicates that the mass reduction rate of the sample before and after the measurement is 0.35% by mass or more and less than 0.65% by mass
- “Class” means that the weight loss rate of the sample before and after measurement is 0.65% by mass or more and less than 1.20% by mass
- “Class 5" means that the weight loss rate of the sample before and after measurement is 1.20% by mass.
- the amount is less than 2.20% by mass
- “Class 6” has a mass reduction rate of the sample before and after the measurement of 2.20% by mass or more.
- the optical glass of the present invention preferably has a low degree of wear.
- the upper limit of the degree of wear of the optical glass of the present invention is preferably 200, more preferably 150, more preferably 100, more preferably 80, and still more preferably 60.
- the abrasion degree means a value obtained by measurement according to “JOGIS10-1994 Measuring method of abrasion degree of optical glass”.
- the optical glass of the present invention has high visible light transmittance, in particular, high transmittance of light on the short wavelength side of visible light, and thereby less coloring.
- the wavelength ( ⁇ 70 ) showing a spectral transmittance of 70% in a sample having a thickness of 10 mm is preferably 500 nm, more preferably 480 nm. More preferably, the upper limit is 450 nm, and still more preferably 420 nm.
- the wavelength ( ⁇ 80 ) exhibiting a spectral transmittance of 80% in a sample having a thickness of 10 mm is preferably 500 nm, more preferably 480 nm. More preferably, the upper limit is 450 nm, and still more preferably 420 nm.
- the shortest wavelength ( ⁇ 5 ) having a spectral transmittance of 5% in a sample having a thickness of 10 mm is preferably 400 nm, more preferably 380 nm, still more preferably 370 nm, more preferably 360 nm. To do.
- this optical glass can be preferably used for an optical element that transmits light such as a lens.
- a glass molded body can be produced from the produced optical glass by means of, for example, polishing or molding press molding such as reheat press molding or precision press molding. That is, a glass molded body is produced by performing mechanical processing such as grinding and polishing on the optical glass, or a preform for molding a mold press is produced from the optical glass, and reheat press molding is performed on the preform. Then, polishing is performed to produce a glass molded body, or precision preforming is performed on a preform formed by polishing or a preform formed by known floating molding, etc., to form a glass molded body. Can be produced.
- the means for producing the glass molded body is not limited to these means.
- the optical glass of the present invention is useful for various optical elements and optical designs.
- preforms with large diameters can be formed, so that the optical elements can be made larger, but when they are used in optical equipment such as cameras and projectors, high-definition and high-precision imaging characteristics and projection characteristics. Can be realized.
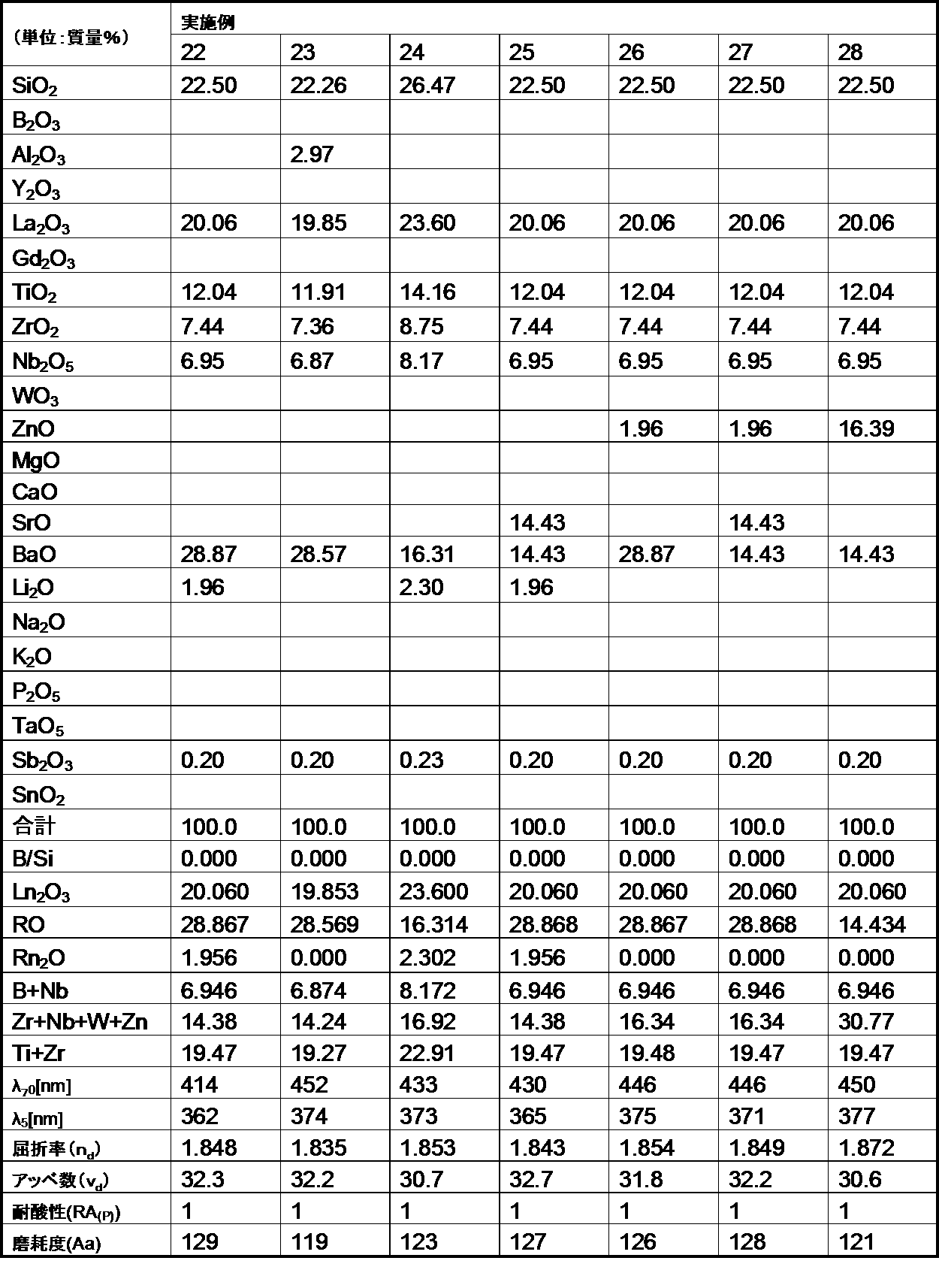
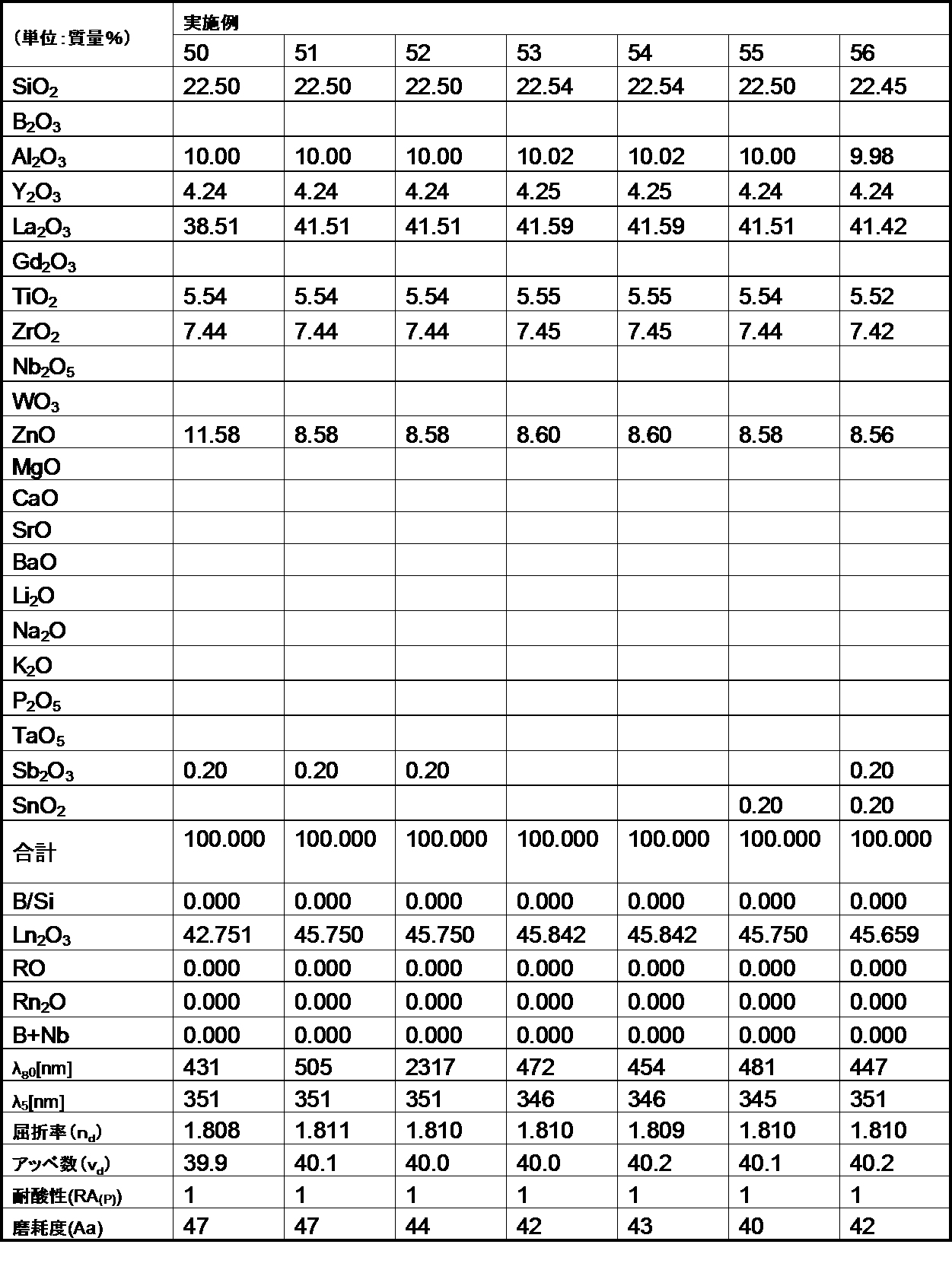
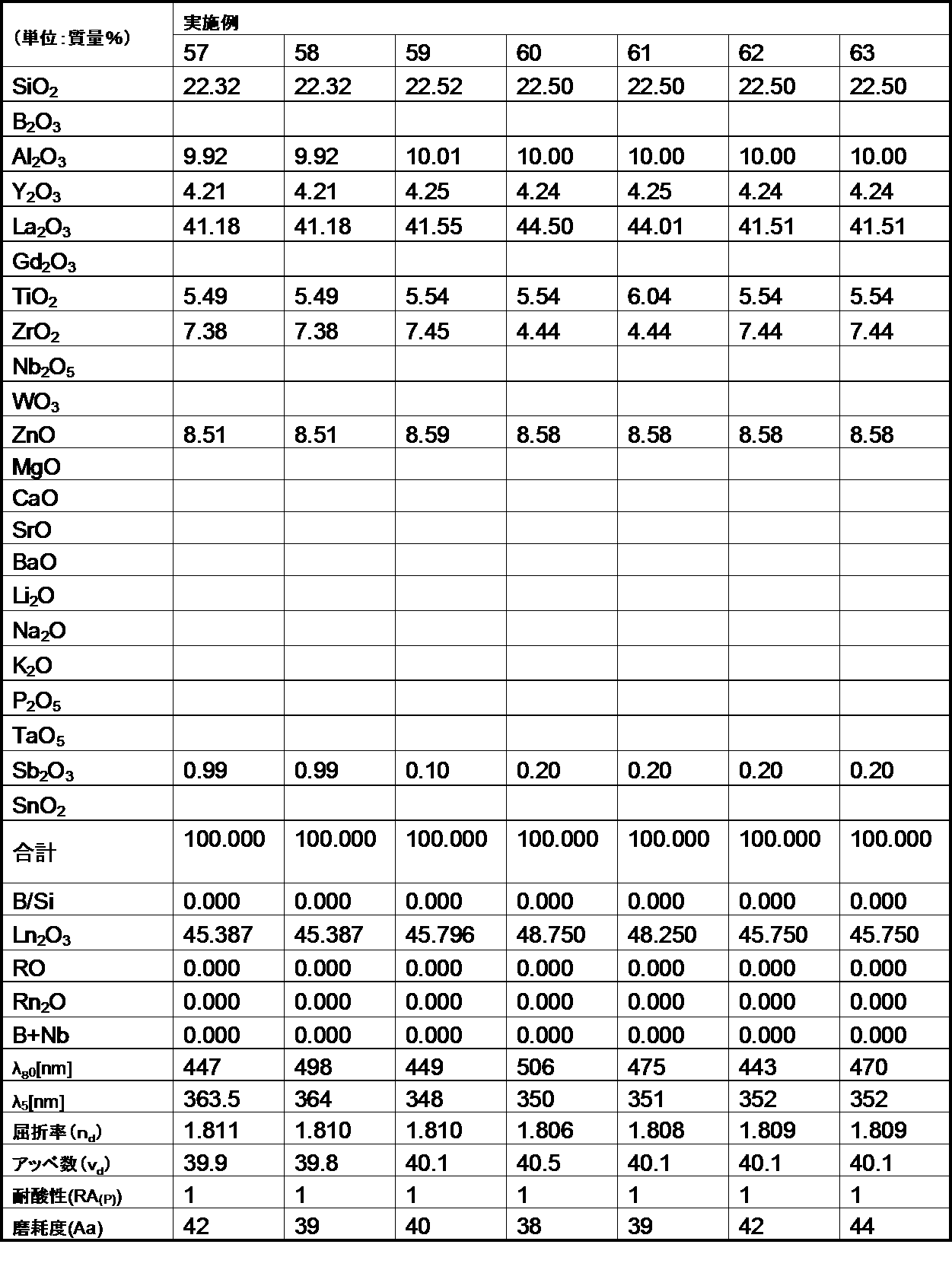
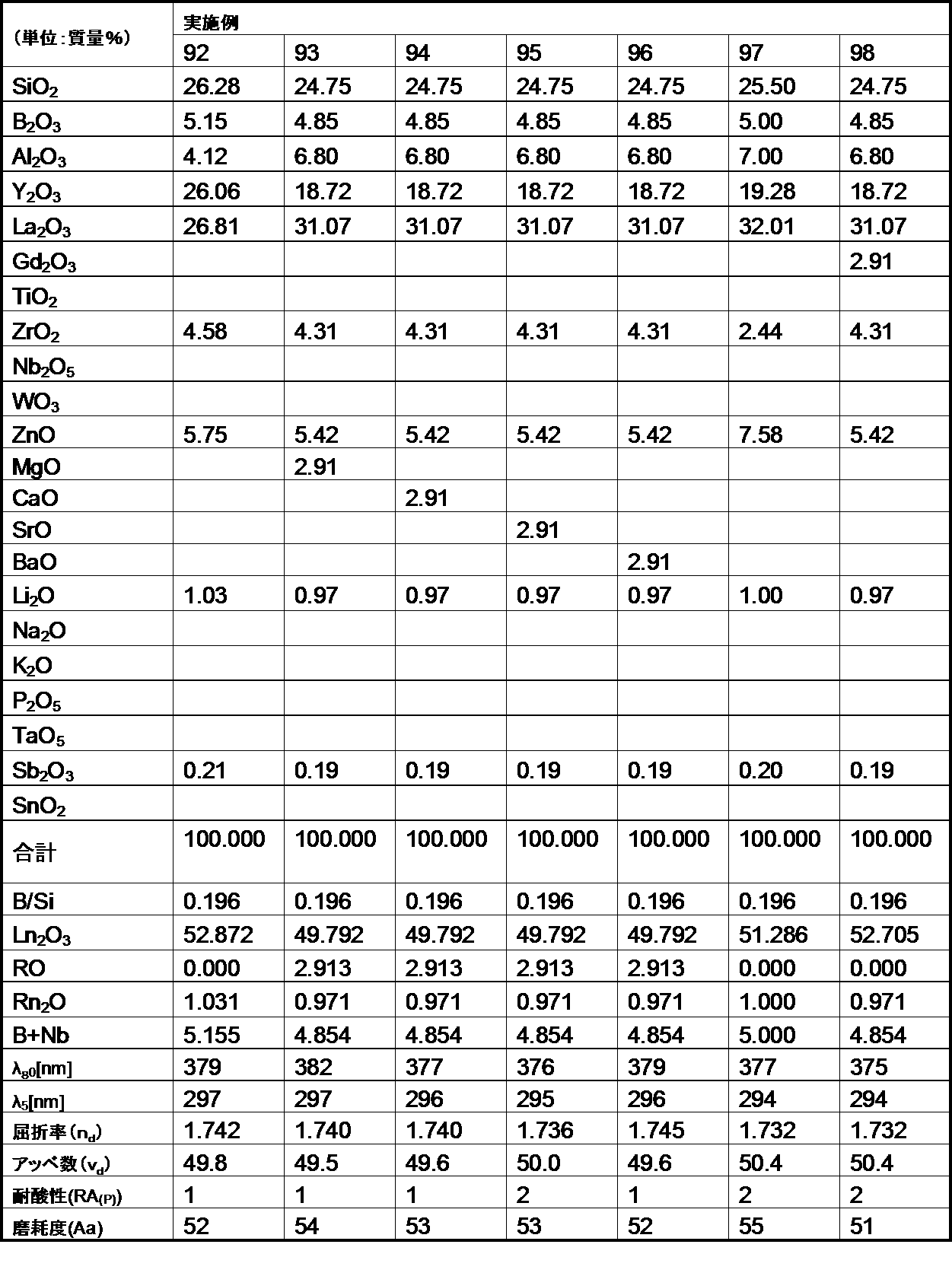
- the following examples are merely for illustrative purposes, and are not limited to these examples.
- the glasses of the examples and comparative examples of the present invention are ordinary optical glasses such as oxides, hydroxides, carbonates, nitrates, fluorides, hydroxides, and metaphosphate compounds corresponding to the raw materials of the respective components.
- Select the high-purity raw materials used in the above weigh them so that they have the composition ratios of the respective examples shown in the table, mix them uniformly, and then put them into a platinum crucible, depending on the difficulty of melting the glass raw materials. After melting in an electric furnace at a temperature range of 1100 to 1550 ° C. for 2 to 5 hours, stirring and homogenizing, the mixture was cast into a mold and slowly cooled.
- the refractive index (n d ) and the Abbe number ( ⁇ d ) of the glass of the example are shown as measured values for the d-line (587.56 nm) of the helium lamp.
- the Abbe number ( ⁇ d ) is the refractive index of the d line, the refractive index (n F ) for the F lamp (486.13 nm) of the hydrogen lamp, and the refractive index (n C ) for the C line (656.27 nm).
- the Abbe number ( ⁇ d ) [(n d ⁇ 1) / (n F ⁇ n C )].
- the transmittance of the glass of Examples and Comparative Examples was measured according to Japan Optical Glass Industry Association Standard JOGIS02-2003 “Measurement Method of Color Degree of Optical Glass”.
- the presence / absence and degree of coloration of the glass were determined by measuring the transmittance of the glass.
- a face parallel polished product having a thickness of 10 ⁇ 0.1 mm was measured for a spectral transmittance of 200 to 800 nm in accordance with JISZ8722, and ⁇ 5 (wavelength at a transmittance of 5%), ⁇ 80 (transmittance). Wavelength at 80%) ⁇ 70 (wavelength at 70% transmittance) was determined.
- the acid resistance of the glasses of Examples and Comparative Examples was measured according to Japan Optical Glass Industry Association Standard JOGIS 06-1999 “Method for Measuring Chemical Durability of Optical Glass”. That is, a glass sample crushed to a particle size of 425 to 600 ⁇ m was placed in a specific gravity bottle and placed in a platinum basket. The platinum basket was placed in a quartz glass round bottom flask containing a 0.01N nitric acid aqueous solution and treated in a boiling water bath for 60 minutes.
- the weight loss rate (mass%) of the glass sample after treatment is calculated, and when the weight loss ratio (mass%) is less than 0.20, the first grade, and when the weight loss rate is less than 0.20 to 0.35, 2 Grade, when the weight loss rate is 0.35 to less than 0.65, Grade 3, when the weight loss rate is 0.65 to less than 1.20, Grade 4, when the weight loss rate is less than 1.20 to 2.20
- the grade 5 and the weight loss rate of 2.20 or more were classified as grade 6. At this time, it means that the acid resistance of glass is excellent, so that the number of grades is small.
- the degree of wear was measured according to JOGIS 10-1994 “Measurement Method of Abrasion Level of Optical Glass”. That is, a sample of a glass square plate having a size of 30 ⁇ 30 ⁇ 10 mm is placed on a fixed position of 80 mm from the center of a flat plate made of cast iron (250 mm ⁇ ) horizontally rotating 60 times per minute, and a load of 9.8 N (1 kgf) is applied. While applying vertically, a polishing solution obtained by adding 10 g of lapping material (alumina A abrasive grains) of # 800 (average particle size 20 ⁇ m) to 20 mL of water is uniformly fed for 5 minutes to cause friction, and the sample mass before and after the lapping is measured.
- lapping material alumina A abrasive grains
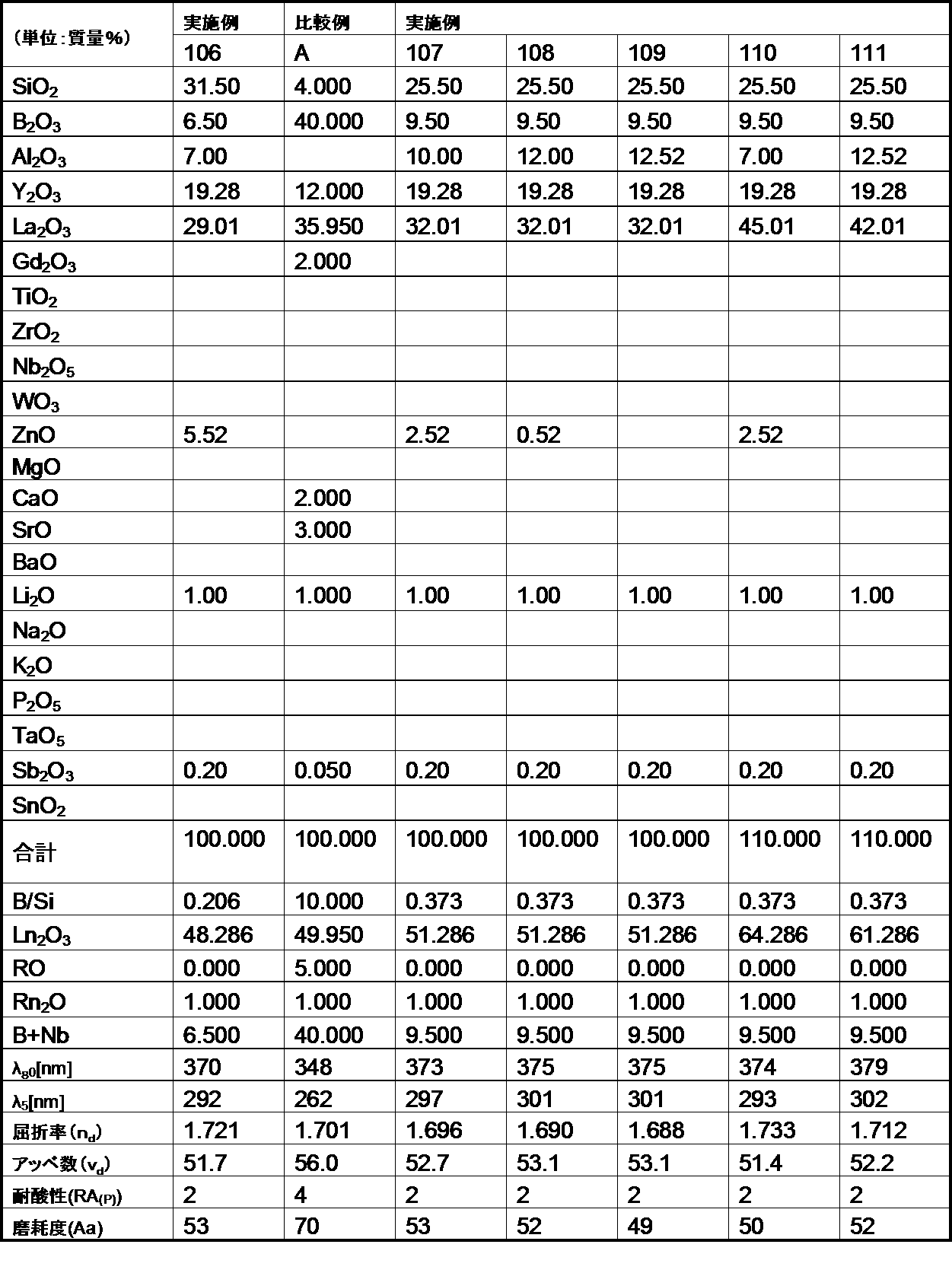
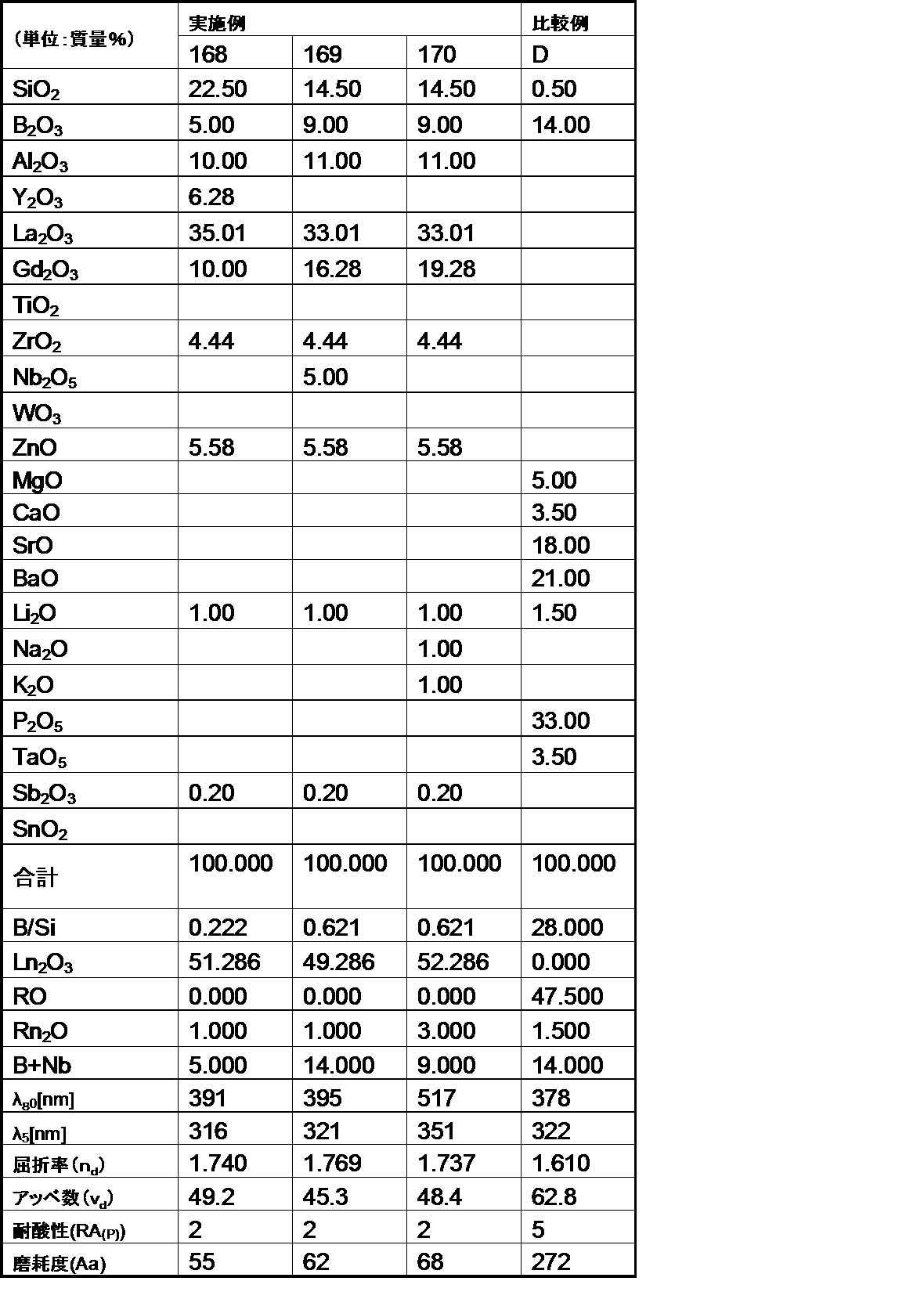
- the optical glass of the example of the first aspect of the present invention has a mass ratio (B 2 O 3 / SiO 2 ) of 1.0 or less. Satisfies.
- the glasses of Comparative Examples A and B have poor acid resistance because the mass ratio (B 2 O 3 / SiO 2 ) exceeds 1.0.
- the optical glasses of the embodiments of the first aspect of the present invention all have a refractive index (n d ) of 1.78 or more, more specifically 1.80 or more, and this refractive index (n d ). Was 1.95 or less, more specifically 1.93 or less, and was within the desired range.
- the optical glasses of the examples of the first aspect of the present invention all have an Abbe number ( ⁇ d ) of 45 or less, more specifically 40 or less, and the Abbe number ( ⁇ d ) of 25 or more. More specifically, it was 28 or more, which was within the desired range.
- each ⁇ 70 (wavelength at 70% transmittance) was 500 nm or less, more specifically 480 nm or less.
- ⁇ 5 (wavelength at 5% transmittance) was all 400 nm or less, more specifically, 380 nm or less, and was in a desired range.
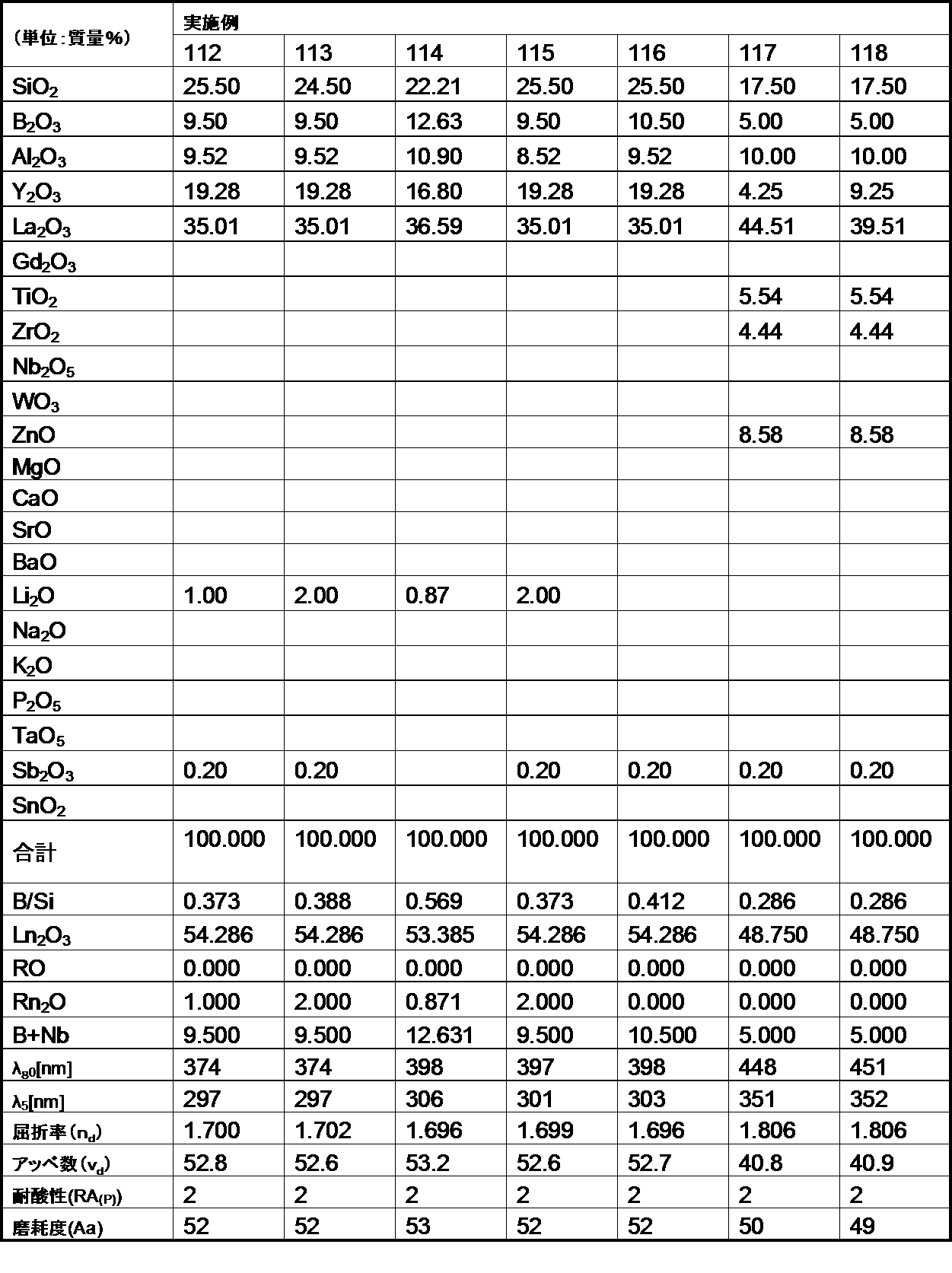
- the optical glass of the example of the second aspect of the present invention has a mass ratio (B 2 O 3 / SiO 2 ) of 1.0 or less. Satisfies.
- the glasses of Comparative Examples A and B have poor acid resistance because the mass ratio (B 2 O 3 / SiO 2 ) exceeds 1.0.
- the optical glasses of the examples of the second aspect of the present invention all have a refractive index (n d ) of 1.60 or more, and this refractive index (n d ) is 1.85 or less, which is within a desired range. It was.
- the optical glasses of the examples of the second aspect of the present invention all have an Abbe number ( ⁇ d ) of 62 or less, more specifically 57 or less, and the Abbe number ( ⁇ d ) of 33 or more. More specifically, it was 35 or more, and was within the desired range.
- the optical glass of the example of the second aspect of the present invention had an abrasion degree of 200 or less. Therefore, it became clear that the optical glass of the example of the present invention is excellent in the degree of wear, and when the optical glass is used as a lens, scratches are hardly generated on the surface.
- each of ⁇ 80 (wavelength at 80% transmittance) was 500 nm or less, more specifically 490 nm or less.
- each of ⁇ 5 (wavelength at 5% transmittance) was 400 nm or less, more specifically 390 nm or less, and was in a desired range.
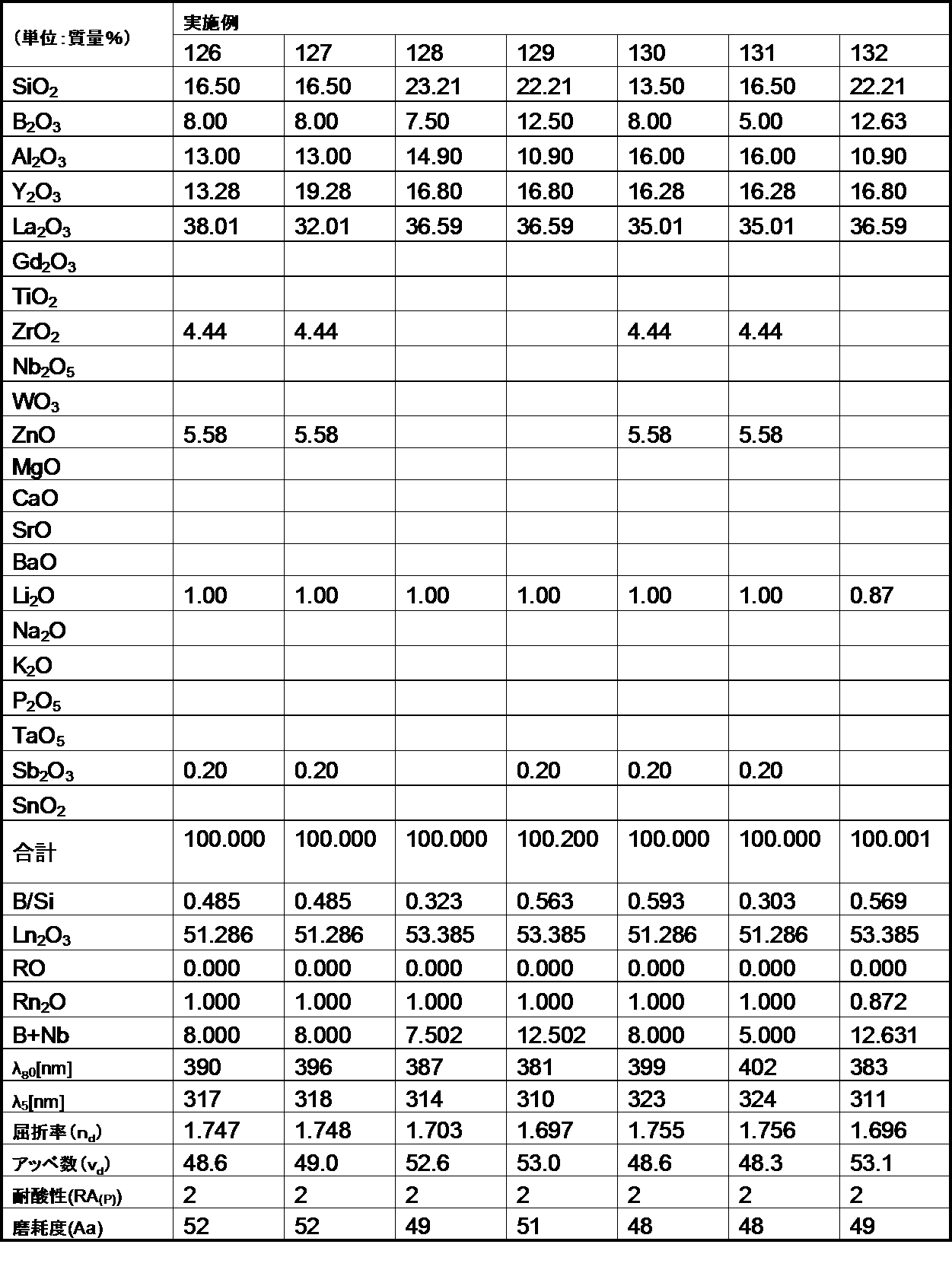
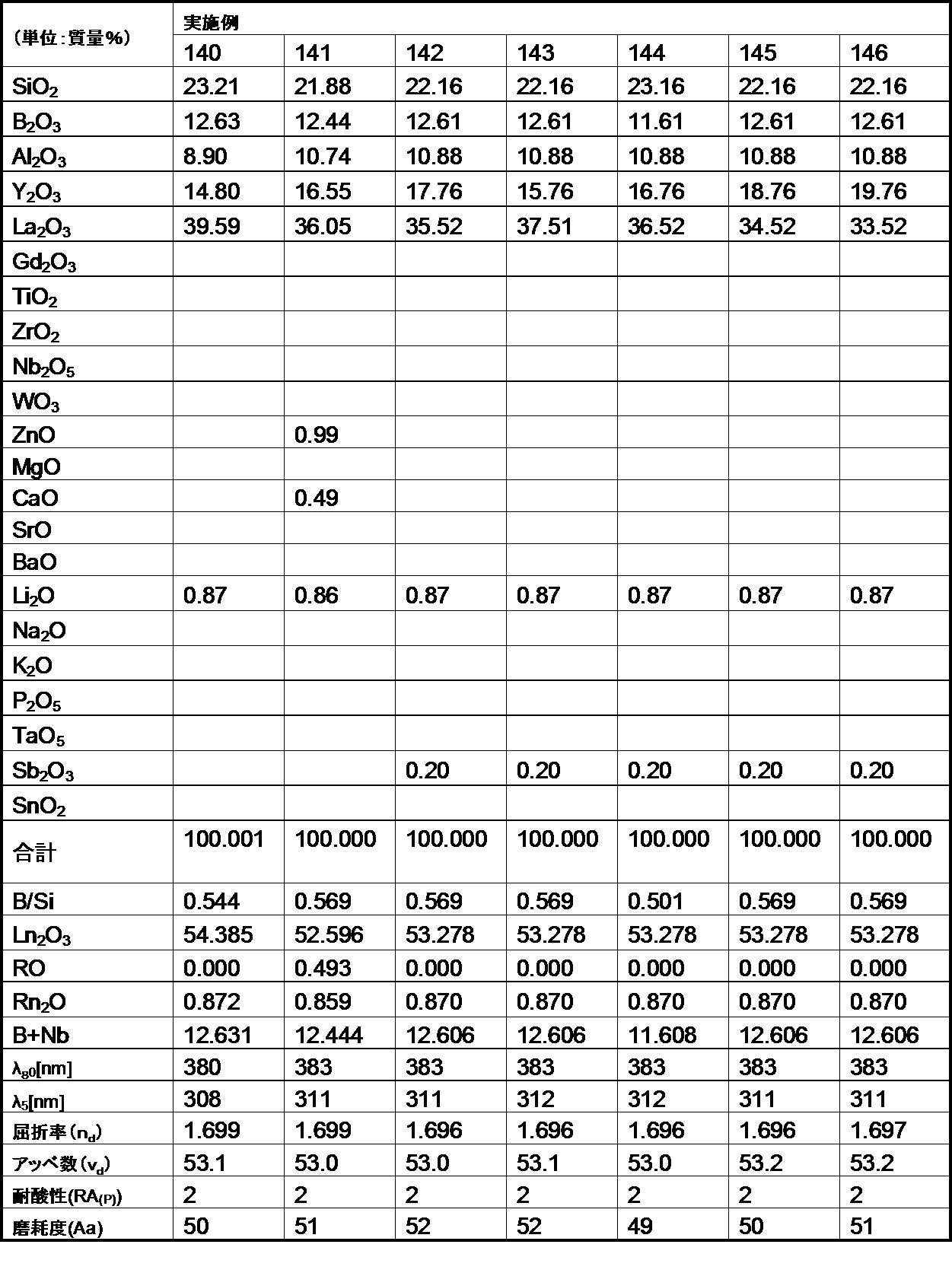
- the optical glass of the Example of this invention was 200 or less abrasion degree. Therefore, it became clear that the optical glass of the example of the present invention is excellent in the degree of wear, and when the optical glass is used as a lens, scratches are hardly generated on the surface.
- the optical glass of the example of the present invention has a chemical durability (acid resistance) of 1 by the powder method, even though the refractive index (n d ) and Abbe number ( ⁇ d ) are within the desired ranges.
- the third grade was within the desired range. For this reason, it became clear that the optical glass of the Example of this invention was excellent in chemical durability (acid resistance).
- a glass block was formed using the optical glass of the example of the present invention, and this glass block was ground and polished to be processed into the shape of a lens and a prism. As a result, it was possible to stably process into various lens and prism shapes.
Abstract
Description
しかしながら、B2O3成分を多量に含有すると、化学的耐久性(耐酸性)や摩耗度のような耐久性が低下するという不利益が生じる。
具体的には、本発明は以下のようなものを提供する。
SiO2成分を10.0%~40.0%、
La2O3成分を15.0 %~50.0%、
TiO2成分 5.0~25.0%未満、
含有し、
質量比B2O3/SiO2が1.00以下であり、
1.78~1.95の屈折率(nd)、25~45のアッベ数(νd)を有し、
粉末法による化学的耐久性(耐酸性)が1級~3級を有する光学ガラス。
ZnO成分を0~30.0%、
ZrO2成分 0~20.0%
Al2O3成分 0~20.0%
Y2O3成分 0~25.0%
B2O3成分 0~20.0%
である(1)記載の光学ガラス。
SiO2成分を10.0%~50.0%、
La2O3成分を15.0 %~60.0%、
TiO2成分 0 ~15.0%未満、
含有し、
質量比B2O3/SiO2が1.00以下であり、
1.60~1.85の屈折率(nd)、33~62のアッベ数(νd)を有し、
粉末法による化学的耐久性(耐酸性)が1級~3級を有する光学ガラス。
ZnO成分を0~35.0%、
ZrO2成分 0~20.0%、
Al2O3成分 0~20.0%、
B2O3成分 0~20.0%、
である(5)記載の光学ガラス。
(9) 摩耗度が200以下である(1)から(8)のいずれか記載の光学ガラス。
本発明の第一の態様に係る光学ガラスは、質量%で、SiO2成分を10.0%以上40.0%以下、La2O3成分を15.0%以上50.0%以下含有し、TiO2成分を5.0%以上25%未満含有し、質量比(B2O3/SiO2)が1.0以下であり、1.78以上1.95以下の屈折率(nd)を有し、25以上45以下のアッベ数(νd)を有する。SiO2成分及びLa2O3成分が主成分の光学ガラスにおいて、1.78以上の屈折率(nd)及び25以上45以下のアッベ数(νd)を有しながらも、耐酸性が高いガラスが得られ易くなる。
本発明の光学ガラスを構成する各成分の組成範囲を以下に述べる。本明細書中において、各成分の含有量は、特に断りがない場合、全て酸化物換算組成の全質量数に対する質量%で表示されるものとする。ここで、「酸化物換算組成」は、本発明のガラス構成成分の原料として使用される酸化物、複合塩、金属弗化物等が熔融時に全て分解され酸化物へ変化すると仮定した場合に、当該生成酸化物の総質量数を100質量%として、ガラス中に含有される各成分を表記した組成である。
SiO2成分は、高い耐久性を有する本発明の光学ガラスでは、ガラス形成酸化物として必須の成分である。特に、SiO2成分の含有量を10.0%以上にすることで、ガラスの耐酸性を高め、磨耗度を低下させ、且つガラスの粘性を高められる。従って、SiO2成分の含有量は、好ましくは10.0%、より好ましくは15.0%、さらに好ましくは20.0%、さらに好ましくは25.0%を下限とする。
一方、SiO2成分の含有量を40.0%以下にすることで、より大きな屈折率を得易くでき、且つ失透性の悪化を抑えられる。従って、SiO2成分の含有量は、好ましくは40.0%以下、より好ましくは37.0%未満、さらに好ましくは35.0%未満、さらに好ましくは33.0%未満とする。
SiO2成分は、原料としてSiO2、K2SiF6、Na2SiF6、ZrSiO4等を用いることができる。
一方、La2O3成分の含有量を50.0%以下にすることで、ガラスの安定性を高めることで失透を低減でき、アッベ数の必要以上の上昇を抑えられる。また、ガラス原料の熔解性を高められる。従って、La2O3成分の含有量は、好ましくは50.0%以下、より好ましくは45.0%未満、さらに好ましくは40.0%未満とする。
La2O3成分は、原料としてLa2O3、La(NO3)3・XH2O(Xは任意の整数)等を用いることができる。
他方で、TiO2成分の含有量を25.0%未満にすることで、TiO2成分の過剰な含有による失透を低減でき、ガラスの可視光(特に波長500nm以下)に対する透過率の低下を抑えられる。また、これによりアッベ数の低下を抑えられる。従って、TiO2成分の含有量は、好ましくは25.0%未満、より好ましくは24.0%未満、さらに好ましくは21.0%未満、さらに好ましくは19.0%未満、最も好ましくは15.0%以下とする。
TiO2成分は、原料としてTiO2等を用いることができる。
特に、この質量比を1.0以下にすることで、耐酸性が向上し、長期間の使用に耐えうるガラスを得易くできる。従って、質量比B2O3/SiO2は、好ましくは1.0以下、より好ましくは0.98以下、さらに好ましくは0.90以下、さらに好ましくは0.80以下、さらに好ましくは0.70以下とする。
他方で、ZnO成分の含有量30.0%以下にすることで、ガラスの屈折率の低下を抑えられ、且つ、過剰な粘性の低下による失透を低減できる。従って、ZnO成分の含有量は、好ましくは30.0%以下、より好ましくは28.0%未満、さらに好ましくは25.0%未満とする。
ZnO成分は、原料としてZnO、ZnF2等を用いることができる。
他方で、ZrO2成分の含有量を20.0%以下にすることで、ZrO2成分の過剰な含有による失透を低減できる。従って、ZrO2成分の含有量は、好ましくは20.0%以下、より好ましくは18.0%未満、さらに好ましくは16.0%未満、さらに好ましくは14.0%未満とする。
ZrO2成分は、原料としてZrO2、ZrF4等を用いることができる。
他方で、Al2O3成分の含有量を20.0%以下にすることで、ガラスの液相温度を下げて耐失透性を高められる。従って、Al2O3成分の含有量は、好ましくは20.0%以下、より好ましくは18.0%未満、さらに好ましくは16.5%未満、さらに好ましくは15.0%未満とする。
Al2O3成分は、原料としてAl2O3、Al(OH)3、AlF3等を用いることができる。
他方で、Y2O3成分の含有量を25.0%以下にすることで、ガラスの屈折率の低下を抑えられ、且つガラスの安定性を高められる。また、ガラス原料の熔解性の悪化を抑えられる。従って、Y2O3成分の含有量は、好ましくは25.0%以下、より好ましくは23.0%未満、より好ましくは20.0%未満、最も好ましくは18.0%以下とする。
Y2O3成分は、原料としてY2O3、YF3等を用いることができる。
他方で、B2O3成分の含有量を20.0%以下にすることで、より大きな屈折率を得易くでき、且つ化学的耐久性(耐酸性)の悪化と磨耗度の上昇を抑えられる。従って、B2O3成分の含有量は、好ましくは20.0%以下、より好ましくは16.0%未満、さらに好ましくは14.0%未満、さらに好ましくは13.0%未満、さらに好ましくは12.0%未満、さらに好ましくは10.0%未満とする。
B2O3成分は、原料としてH3BO3、Na2B4O7、Na2B4O7・10H2O、BPO4等を用いることができる。
他方で、Nb2O5成分の含有量を15.0%未満にすることで、Nb2O5成分の過剰な含有による失透を低減でき、且つ、ガラスの可視光(特に波長500nm以下)に対する透過率の低下を抑えられる。また、これによりアッベ数の低下を抑えられる。従って、Nb2O5成分の含有量は、好ましくは15.0%未満、より好ましくは13.0%未満、さらに好ましくは10.0%未満、さらに好ましくは7.0%未満とする。
Nb2O5成分は、原料としてNb2O5等を用いることができる。
他方で、WO3成分の含有量を10.0%未満にすることで、ガラスの材料コストを抑えられる。また、WO3成分によるガラスの着色を低減して可視光透過率を高められる。従って、WO3成分の含有量は、好ましくは10.0%未満、より好ましくは6.0%未満、より好ましくは4.5%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
WO3成分は、原料としてWO3等を用いることができる。
しかしながら、Gd2O3成分は原料価格が高く、その含有量が多いと生産コストが高くなる。また、Gd2O3成分の含有を25.0%以下とすることにより、ガラスのアッベ数の上昇を抑えられる。従って、Gd2O3成分の含有量は、好ましくは25.0%以下、より好ましくは23.0%未満、さらに好ましくは20.0%未満とする。
Gd2O3成分は、原料としてGd2O3、GdF3等を用いることができる。
しかしながら、Yb2O3成分は原料価格が高く、その含有量が多いと生産コストが高くなる。また、Yb2O3成分の含有を5.0%未満とすることにより、ガラスのアッベ数の上昇を抑えられる。従って、Yb2O3成分の含有量は、好ましくは5.0%未満、より好ましくは3.0%未満、さらに好ましくは2.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
Yb2O3成分は、原料としてYb2O3等を用いることができる。
しかしながら、Ta2O5成分は原料価格が高く、その含有量が多いと生産コストが高くなる。また、Ta2O5成分の含有量を5.0%未満にすることで、原料の熔解温度が低くなり、原料の熔解に要するエネルギーが低減されるため、光学ガラスの製造コストも低減できる。従って、Ta2O5成分の含有量は、好ましくは5.0%未満、より好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。また、材料コストを低減させる観点では、Ta2O5成分を含有しないことが最も好ましい。
Ta2O5成分は、原料としてTa2O5等を用いることができる。
MgO成分の含有量をそれぞれ10.0%以下にすることで、屈折率の低下を抑えることができ、且つこれらの成分の過剰な含有による失透を低減できる。従って、MgO成分の含有量は、それぞれ好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満とする。
MgO成分は、原料としてMgCO3、MgF2等を用いることができる。
CaO成分の含有量を35.0%以下にすることでも、所望の屈折率を得易くでき、且つこれらの成分の過剰な含有による失透を低減できる。従って、CaO成分の含有量は、好ましくは35.0%以下、より好ましくは30.0%未満、さらに好ましくは25.0%未満、さらに好ましくは22.0%未満、さらに好ましくは20.0%未満とする。
CaO成分は、原料としてCaCO3、CaF2等を用いることができる。
SrO成分の含有量を35.0%以下にすることでも、所望の屈折率を得易くでき、且つこれらの成分の過剰な含有による失透を低減できる。従って、SrO成分の含有量は、好ましくは35.0%以下、より好ましくは30.0%未満、さらに好ましくは25.0%未満、さらに好ましくは22.0%未満、さらに好ましくは20.0%未満とする。
SrO成分は、原料としてSr(NO3)2、SrF2等を用いることができる。
BaO成分の含有量を35.0%以下にすることでも、所望の屈折率を得易くでき、且つこれらの成分の過剰な含有による失透を低減できる。従って、BaO成分の含有量は、好ましくは35.0%以下、より好ましくは30.0%未満、さらに好ましくは29.0%未満、さらに好ましくは25.0%未満、さらに好ましくは22.0%未満、さらに好ましくは20.0%未満とする。
BaO成分は、原料としてBaCO3、Ba(NO3)2、BaF2等を用いることができる。
他方で、Li2O成分の含有量を10.0%以下にすることで、化学的耐久性(耐酸性)の悪化を抑制し、ガラスの屈折率を低下し難くし、且つガラスの失透を低減できる。また、Li2O成分の含有量を低減させることで、ガラスの粘性が高められるため、ガラスの脈理を低減できる。従って、Li2O成分の含有量は、好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
Li2O成分は、原料としてLi2CO3、LiNO3、Li2CO3等を用いることができる。
他方で、Na2O成分の含有量は10.0%以下にすることで、ガラスの屈折率を低下し難くし、且つガラスの失透を低減できる。従って、Na2O成分の含有量は、それぞれ好ましくは10.0%以下、より好ましくは6.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
Na2O成分は、原料としてNa2CO3、NaNO3、NaF、Na2SiF6等を用いることができる。
他方で、K2O成分の含有量を10.0%以下にすることで、ガラスの屈折率を低下し難くし、磨耗度の上昇を抑制し、且つガラスの失透を低減できる。従って、K2O成分の含有量は、それぞれ好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
K2O成分は、原料としてK2CO3、KNO3、KF、KHF2、K2SiF6等を用いることができる。
他方で、P2O5成分の含有量を10.0%以下にすることで、ガラスの化学的耐久性(耐酸性)の低下、磨耗度の上昇を抑制できる。従って、P2O5成分の含有量は、好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満とする。
P2O5成分は、原料としてAl(PO3)3、Ca(PO3)2、Ba(PO3)2、BPO4、H3PO4等を用いることができる。
しかしながら、GeO2は原料価格が高く、その含有量が多いと生産コストが高くなる。従って、GeO2成分の含有量は、好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.1%未満とする。材料コストを低減させる観点で、GeO2成分を含有しなくてもよい。
GeO2成分は、原料としてGeO2等を用いることができる。
他方で、Ga2O3成分の含有量を10.0%以下にすることで、ガラスの液相温度を下げて耐失透性を高められる。従って、Ga2O3成分の含有量は、好ましくは10.0%以下、より好ましくは8.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満とする。
Ga2O3成分は、原料としてGa2O3、Ga(OH)3等を用いることができる。
他方で、Bi2O3成分の含有量を10.0%以下にすることで、ガラスの液相温度を下げて耐失透性を高められる。従って、Bi2O3成分の含有量は、好ましくは10.0%以下、より好ましくは8.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、最も好ましくは含有しないこととする。
Bi2O3成分は、原料としてBi2O3等を用いることができる。
他方で、TeO2は白金製の坩堝や、熔融ガラスと接する部分が白金で形成されている熔融槽でガラス原料を熔融する際、白金と合金化しうる問題がある。従って、TeO2成分の含有量は、好ましくは10.0%以下、より好ましくは8.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満とする。
TeO2成分は、原料としてTeO2等を用いることができる。
他方で、ガラスの屈折率を低下し難くし、且つガラスの失透を低減できる。従って、CsO2成分の含有量は、好ましくは3.0%以下、より好ましくは2.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.1%未満、最も好ましくは含有しないことする。
CsO2成分は、原料としてCs2CO3、CsNO3等を用いることができる。
他方で、SnO2成分の含有量を3.0%以下にすることで、熔融ガラスの還元によるガラスの着色や、ガラスの失透を低減できる。また、SnO2成分と熔解設備(特にPt等の貴金属)の合金化が低減されるため、熔解設備の長寿命化を図れる。従って、SnO2成分の含有量は、好ましくは3.0%以下、より好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
SnO2成分は、原料としてSnO、SnO2、SnF2、SnF4等を用いることができる。
他方で、Sb2O3量が多すぎると、可視光領域の短波長領域における透過率が悪くなる。従って、Sb2O3成分の含有量は、好ましくは3.0%以下、より好ましくは2.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満とする。
Sb2O3成分は、原料としてSb2O3、Sb2O5、Na2H2Sb2O7・5H2O等を用いることができる。
しかし、F成分の含有量、すなわち上述した各金属元素の1種又は2種以上の酸化物の一部又は全部と置換した弗化物のFとしての合計量が15.0%を超えると、F成分の揮発量が多くなるため、安定した光学恒数が得られ難くなり、均質なガラスが得られ難くなる。また、アッベ数が必要以上に上昇する。
従って、F成分の含有量は、好ましくは15.0%以下、より好ましくは10.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満とする。
F成分は、原料として例えばZrF4、AlF3、NaF、CaF2等を用いることで、ガラス内に含有することができる。
他方でB2O3成分及びNb2O5成分の合計量(質量和)を0%超とすることで耐失透性を向上できる。従って質量和B2O3+Nb2O5は、好ましくは0%超、より好ましくは3.0%超、より好ましくは5.0%超、より好ましくは6.0%超としても良い。
これにより、アッベ数(νd)が低くなることを低減できる。従って、質量和TiO2+ZrO2は、好ましくは35.0%未満、より好ましくは33.0%以下、さらに好ましくは30.0%未満、さらに好ましくは28.0%未満、さらに好ましくは25.0%以下を上限とする。
他方でTiO2成分及びZrO2成分の合計量(質量和)は0%超とする事でガラスの屈折率を高めることが出来る。従って質量和TiO2+ZrO2は、好ましくは0%超、より好ましくは5.0%以上、より好ましくは8.0%超、より好ましくは10.0%超、さらに好ましくは13.0%超、さらに好ましくは15.0%超としてもよい。
これにより、アッベ数(νd)が所望の範囲内に調整できる。従って、質量和ZrO2+Nb2O5+WO3+ZnOは、好ましくは5.0%以上、より好ましくは7.0%超、より好ましくは9.0%超、さらに好ましくは11.0%超、さらに好ましくは13.0%超としてもよい。
他方でZrO2成分及びNb2O5成分及びWO3成分及びZnO成分の合計量(質量和)を60.0%以下とする事で、ガラスの耐失透性を高められる。従って質量和ZrO2+Nb2O5+WO3+ZnOは、好ましくは60.0%以下、より好ましくは55.0%未満、より好ましくは50.0%未満、より好ましくは48.0%未満としても良い。
特に、この和を15.0%以上にすることで、ガラスの屈折率及びアッベ数が高められるため、所望の屈折率及びアッベ数を有するガラスを得易くすることができる。従って、Ln2O3成分の質量和は、好ましくは15.0%以上、より好ましくは16.0%超、さらに好ましくは18.0%超、さらに好ましくは20.0%超とする。
他方で、この和を65.0%以下にすることで、ガラスの液相温度が低くなるため、ガラスの失透を低減できる。また、アッベ数の必要以上の上昇を抑えられる。従って、Ln2O3成分の質量和は、好ましくは65.0%以下、より好ましくは60.0%未満、さらに好ましくは55.0%未満、さらに好ましくは50.0%未満とする。
本発明の第二の態様の光学ガラスは、質量%で、SiO2成分を10.0%以上50.0%以下、La2O3成分を15.0%以上60.0%以下含有し、TiO2成分を0.0%以上15%未満含有し、質量比(B2O3/SiO2)が1.0以下であり、1.60以上1.85以下の屈折率(nd)を有し、33以上62以下のアッベ数(νd)を有する。SiO2成分及びLa2O3成分が主成分の光学ガラスにおいて、1.60以上の屈折率(nd)及び33以上62以下のアッベ数(νd)を有しながらも、耐酸性が高いガラスが得られ易くなる。
SiO2成分は、高い耐久性を有する本発明の光学ガラスでは、ガラス形成酸化物として必須の成分である。特に、SiO2成分の含有量を10.0%以上にすることで、ガラスの耐酸性を高め、磨耗度を低下させ、且つガラスの粘性を高められる。従って、SiO2成分の含有量は、好ましくは10.0%、より好ましくは15.0%、さらに好ましくは20.0%、さらに好ましくは25.0%を下限とする。
一方、SiO2成分の含有量を50.0%以下にすることで、より大きな屈折率を得易くでき、且つ失透性の悪化を抑えられる。従って、SiO2成分の含有量は、好ましくは50.0%以下、より好ましくは47.0%未満、さらに好ましくは45.0%未満、さらに好ましくは43.0%未満とする。
SiO2成分は、原料としてSiO2、K2SiF6、Na2SiF6、ZrSiO4等を用いることができる。
一方、La2O3成分の含有量を60.0%以下にすることで、ガラスの安定性を高めることで失透を低減でき、アッベ数の必要以上の上昇を抑えられる。また、ガラス原料の熔解性を高められる。従って、La2O3成分の含有量は、好ましくは60.0%以下、より好ましくは58.0%未満、さらに好ましくは55.0%未満とする。
La2O3成分は、原料としてLa2O3、La(NO3)3・XH2O(Xは任意の整数)等を用いることができる。
他方で、TiO2成分の含有量を15.0%未満にすることで、TiO2成分の過剰な含有による失透を低減でき、ガラスの可視光(特に波長500nm以下)に対する透過率の低下を抑えられる。また、これによりアッベ数の低下を抑えられる。従って、TiO2成分の含有量は、好ましくは15.0%未満、より好ましくは13.0%未満、さらに好ましくは11.0%未満、さらに好ましくは10.0%未満、さらに好ましくは9.0%未満とする。
TiO2成分は、原料としてTiO2等を用いることができる。
特に、この質量比を1.0以下にすることで、耐酸性が向上し、長期間の使用に耐えうるガラスを得易くできる。従って、質量比B2O3/SiO2は、好ましくは1.0以下、より好ましくは0.98以下、さらに好ましくは0.90以下、さらに好ましくは0.80以下、さらに好ましくは0.70以下とする。
他方で、ZnO成分の含有量35.0%以下にすることで、ガラスの屈折率の低下を抑えられ、且つ、過剰な粘性の低下による失透を低減できる。従って、ZnO成分の含有量は、好ましくは35.0%以下、より好ましくは33.0%未満、さらに好ましくは31.0%未満、さらに好ましくは29.0%未満とする。
ZnO成分は、原料としてZnO、ZnF2等を用いることができる。
他方で、ZrO2成分の含有量を20.0%以下にすることで、ZrO2成分の過剰な含有による失透を低減できる。従って、ZrO2成分の含有量は、好ましくは20.0%以下、より好ましくは18.0%未満、さらに好ましくは16.0%未満、さらに好ましくは14.0%未満、最も好ましくは10.0%以下とする。
ZrO2成分は、原料としてZrO2、ZrF4等を用いることができる。
他方で、Al2O3成分の含有量を20.0%以下にすることで、ガラスの液相温度を下げて耐失透性を高められる。従って、Al2O3成分の含有量は、好ましくは20.0%以下、より好ましくは18.0%未満、さらに好ましくは16.5%未満、さらに好ましくは15.0%未満、最も好ましくは13.0%以下とする。
Al2O3成分は、原料としてAl2O3、Al(OH)3、AlF3等を用いることができる。
他方で、Y2O3成分の含有量を25.0%以下にすることで、ガラスの屈折率の低下を抑えられ、且つガラスの安定性を高められる。また、ガラス原料の熔解性の悪化を抑えられる。従って、Y2O3成分の含有量は、好ましくは25.0%以下、より好ましくは23.0%未満、より好ましくは20.0%未満とする。
Y2O3成分は、原料としてY2O3、YF3等を用いることができる。
他方で、B2O3成分の含有量を20.0%以下にすることで、より大きな屈折率を得易くでき、且つ化学的耐久性の悪化と磨耗度の上昇を抑えられる。従って、B2O3成分の含有量は、好ましくは20.0%以下、より好ましくは16.0%未満、さらに好ましくは13.0%未満、さらに好ましくは10.0%未満とする。
B2O3成分は、原料としてH3BO3、Na2B4O7、Na2B4O7・10H2O、BPO4等を用いることができる。
他方で、Nb2O5成分の含有量を15.0%未満にすることで、Nb2O5成分の過剰な含有による失透を低減でき、且つ、ガラスの可視光(特に波長500nm以下)に対する透過率の低下を抑えられる。また、これによりアッベ数の低下を抑えられる。従って、Nb2O5成分の含有量は、好ましくは15.0%未満、より好ましくは13.0%未満、より好ましくは9.0%未満、さらに好ましくは7.0%未満、さらに好ましくは5.0%未満とする。
Nb2O5成分は、原料としてNb2O5等を用いることができる。
他方で、WO3成分の含有量を10.0%未満にすることで、ガラスの材料コストを抑えられる。また、WO3成分によるガラスの着色を低減して可視光透過率を高められる。従って、WO3成分の含有量は、好ましくは10.0%未満、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
WO3成分は、原料としてWO3等を用いることができる。
しかしながら、Gd2O3成分は原料価格が高く、その含有量が多いと生産コストが高くなる。また、Gd2O3成分の含有を25.0%以下とすることにより、ガラスのアッベ数の上昇を抑えられる。従って、Gd2O3成分の含有量は、好ましくは25.0%以下、より好ましくは23.0%未満、さらに好ましくは20.0%未満とする。
Gd2O3成分は、原料としてGd2O3、GdF3等を用いることができる。
しかしながら、Yb2O3成分は原料価格が高く、その含有量が多いと生産コストが高くなる。また、Yb2O3成分の含有を5.0%未満とすることにより、ガラスのアッベ数の上昇を抑えられる。従って、Yb2O3成分の含有量は、好ましくは5.0%未満、より好ましくは3.0%未満、さらに好ましくは2.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
Yb2O3成分は、原料としてYb2O3等を用いることができる。
しかしながら、Ta2O5成分は原料価格が高く、その含有量が多いと生産コストが高くなる。また、Ta2O5成分の含有量を5.0%未満にすることで、原料の熔解温度が低くなり、原料の熔解に要するエネルギーが低減されるため、光学ガラスの製造コストも低減できる。従って、Ta2O5成分の含有量は、好ましくは5.0%未満、より好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。また、材料コストを低減させる観点では、Ta2O5成分を含有しないことが最も好ましい。
Ta2O5成分は、原料としてTa2O5等を用いることができる。
MgO成分の含有量をそれぞれ15.0%以下にすることで、屈折率の低下を抑えることができ、且つこれらの成分の過剰な含有による失透を低減できる。従って、MgO成分の含有量は、好ましくは15.0%以下、より好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満とする。
MgO成分は、原料としてMgCO3、MgF2等を用いることができる。
CaO成分の含有量を15.0%以下にすることでも、所望の屈折率を得易くでき、且つこれらの成分の過剰な含有による失透を低減できる。従って、CaO成分の含有量は、好ましくは15.0%以下、より好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満とする。
CaO成分は、原料としてCaCO3、CaF2等を用いることができる。
SrO成分の含有量を15.0%以下にすることでも、所望の屈折率を得易くでき、且つこれらの成分の過剰な含有による失透を低減できる。従ってSrO成分の含有量は、好ましくは15.0%以下、より好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満とする。
SrO成分は、原料としてSr(NO3)2、SrF2等を用いることができる。
BaO成分の含有量を15.0%以下にすることでも、所望の屈折率を得易くでき、且つこれらの成分の過剰な含有による失透を低減できる。従って、BaO成分の含有量は、好ましくは15.0%以下、より好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満とする。
BaO成分は、原料としてBaCO3、Ba(NO3)2、BaF2等を用いることができる。
他方で、Li2O成分の含有量を10.0%以下にすることで、化学的耐久性(耐酸性)の悪化を抑制し、ガラスの屈折率を低下し難くし、且つガラスの失透を低減できる。また、Li2O成分の含有量を低減させることで、ガラスの粘性が高められるため、ガラスの脈理を低減できる。従って、Li2O成分の含有量は、好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
Li2O成分は、原料としてLi2CO3、LiNO3、Li2CO3等を用いることができる。
他方で、Na2O成分の含有量は10.0%以下にすることで、ガラスの屈折率を低下し難くし、且つガラスの失透を低減できる。従って、Na2O成分の含有量は、それぞれ好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
Na2O成分は、原料としてNa2CO3、NaNO3、NaF、Na2SiF6等を用いることができる。
他方で、K2O成分の含有量を10.0%以下にすることで、ガラスの屈折率を低下し難くし、磨耗度の上昇を抑制し、且つガラスの失透を低減できる。従って、K2O成分の含有量は、それぞれ好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
K2O成分は、原料としてK2CO3、KNO3、KF、KHF2、K2SiF6等を用いることができる。
他方で、P2O5成分の含有量を10.0%以下にすることで、ガラスの化学的耐久性(耐酸性)の低下、磨耗度の上昇を抑えられる。従って、P2O5成分の含有量は、好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満とする。
P2O5成分は、原料としてAl(PO3)3、Ca(PO3)2、Ba(PO3)2、BPO4、H3PO4等を用いることができる。
しかしながら、GeO2は原料価格が高く、その含有量が多いと生産コストが高くなる。従って、GeO2成分の含有量は、好ましくは10.0%以下、より好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.1%未満とする。材料コストを低減させる観点で、GeO2成分を含有しなくてもよい。
GeO2成分は、原料としてGeO2等を用いることができる。
他方で、Ga2O3成分の含有量を10.0%以下にすることで、ガラスの液相温度を下げて耐失透性を高められる。従って、Ga2O3成分の含有量は、好ましくは10.0%以下、より好ましくは8.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満とする。
Ga2O3成分は、原料としてGa2O3、Ga(OH)3等を用いることができる。
他方で、Bi2O3成分の含有量を10.0%以下にすることで、ガラスの液相温度を下げて耐失透性を高められる。従って、Bi2O3成分の含有量は、好ましくは10.0%以下、より好ましくは8.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満、最も好ましくは含有しないこととする。
Bi2O3成分は、原料としてBi2O3等を用いることができる。
他方で、TeO2は白金製の坩堝や、熔融ガラスと接する部分が白金で形成されている熔融槽でガラス原料を熔融する際、白金と合金化しうる問題がある。従って、TeO2成分の含有量は、好ましくは10.0%以下、より好ましくは8.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満、さらに好ましくは1.0%未満とする。
TeO2成分は、原料としてTeO2等を用いることができる。
他方で、ガラスの屈折率を低下し難くし、且つガラスの失透を低減できる。従って、CsO2成分の含有量は、好ましくは3.0%以下、より好ましくは2.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.1%未満、最も好ましくは含有しないことする。
CsO2成分は、原料としてCs2CO3、CsNO3等を用いることができる。
他方で、SnO2成分の含有量を3.0%以下にすることで、熔融ガラスの還元によるガラスの着色や、ガラスの失透を低減できる。また、SnO2成分と熔解設備(特にPt等の貴金属)の合金化が低減されるため、熔解設備の長寿命化を図れる。従って、SnO2成分の含有量は、好ましくは3.0%以下、より好ましくは1.0%未満、さらに好ましくは0.5%未満、さらに好ましくは0.1%未満とする。
SnO2成分は、原料としてSnO、SnO2、SnF2、SnF4等を用いることができる。
他方で、Sb2O3量が多すぎると、可視光領域の短波長領域における透過率が悪くなる。従って、Sb2O3成分の含有量は、好ましくは3.0%以下、より好ましくは2.0%未満、さらに好ましくは1.0%未満、さらに好ましくは0.5%未満とする。
Sb2O3成分は、原料としてSb2O3、Sb2O5、Na2H2Sb2O7・5H2O等を用いることができる。
しかし、F成分の含有量、すなわち上述した各金属元素の1種又は2種以上の酸化物の一部又は全部と置換した弗化物のFとしての合計量が15.0%を超えると、F成分の揮発量が多くなるため、安定した光学恒数が得られ難くなり、均質なガラスが得られ難くなる。また、アッベ数が必要以上に上昇する。
従って、F成分の含有量は、好ましくは15.0%以下、より好ましくは10.0%未満、さらに好ましくは5.0%未満、さらに好ましくは3.0%未満とする。
F成分は、原料として例えばZrF4、AlF3、NaF、CaF2等を用いることで、ガラス内に含有することができる。
特に、この和を15.0%以上にすることで、ガラスの屈折率及びアッベ数が高められるため、所望の屈折率及びアッベ数を有するガラスを得易くすることができる。従って、Ln2O3成分の質量和は、好ましくは15.0%以上、より好ましくは16.0%超、さらに好ましくは18.0%超、さらに好ましくは20.0%超とする。
他方で、この和を65.0%以下にすることで、ガラスの液相温度が低くなるため、ガラスの失透を低減できる。また、アッベ数の必要以上の上昇を抑えられる。従って、Ln2O3成分の質量和は、好ましくは65.0%以下、より好ましくは60.0%未満、さらに好ましくは55.0%未満、さらに好ましくは50.0%未満とする。
次に、本発明の光学ガラスに含有すべきでない成分、及び含有することが好ましくない成分について説明する。
本発明の光学ガラスは、例えば以下のように作製される。すなわち、上記原料を各成分が所定の含有量の範囲内になるように均一に混合し、作製した混合物を白金坩堝に投入し、ガラス原料の熔解難易度に応じて電気炉で1100~1550℃の温度範囲で2~5時間熔解させて攪拌均質化した後、適当な温度に下げてから金型に鋳込み、徐冷することにより作製される。
本発明の第一の態様に係る光学ガラスは、高屈折率及び高アッベ数(低分散)を有することが好ましい。特に、本発明の光学ガラスの屈折率(nd)は、好ましくは1.78、より好ましくは1.79、さらに好ましくは1.80を下限とする。この屈折率(nd)は、好ましくは1.95、より好ましくは1.93、さらに好ましくは1.90を上限としてもよい。また、本発明の光学ガラスのアッベ数(νd)は、好ましくは25、より好ましくは27、さらに好ましくは29を下限とする。このアッベ数(νd)は、好ましくは45、より好ましくは43、さらに好ましくは41を上限とする。
本発明の第二の態様に係る光学ガラスは、高屈折率及び高アッベ数(低分散)を有することが好ましい。特に、本発明の光学ガラスの屈折率(nd)は、好ましくは1.60、より好ましくは1.63、さらに好ましくは1.68を下限とする。この屈折率(nd)は、好ましくは1.85、より好ましくは1.84を上限としてもよい。また、本発明の光学ガラスのアッベ数(νd)は、好ましくは33、より好ましくは35、さらに好ましくは37を下限とする。このアッベ数(νd)は、好ましくは62、より好ましくは57、さらに好ましくは55を上限とする。
このような光学恒数を有することで、光学素子の薄型化を図っても大きな光の屈折量を得ることができる。また、このような低分散を有することで、単レンズとして用いたときに光の波長による焦点のずれ(色収差)を小さくできる。そのため、例えば高分散(低いアッベ数)を有する光学素子と組み合わせて光学系を構成した場合に、その光学系の全体として収差を低減させて高い結像特性等を図ることができる。
このように、本発明の光学ガラスは、光学設計上有用であり、特に光学系を構成したときに、高い結像特性等を図りながらも、光学系の小型化を図ることができ、光学設計の自由度を広げることができる。
これにより、光学ガラスを車載用途等で使用する際に、酸性雨等によるガラスの曇りが低減されるため、ガラスからの光学素子の作製をより行い易くできる。
なお、化学的耐久性(耐酸性)の「1級」は、測定前後の試料の質量の減量率が0.20質量%未満であり、「2級」は、測定前後の試料の質量の減量率が0.20質量%以上0.35質量%未満であり、「3級」は、測定前後の試料の質量の減量率が0.35質量%以上0.65質量%未満であり、「4級」は、測定前後の試料の質量の減量率が0.65質量%以上1.20質量%未満であり、「5級」は、測定前後の試料の質量の減量率が1.20質量%以上2.20質量%未満であり、「6級」は、測定前後の試料の質量の減量率が2.20質量%以上である。
なお、摩耗度とは、「JOGIS10-1994光学ガラスの磨耗度の測定方法」に準じて測定して得た値を意味するものとする。
特に、第一の態様に係る本発明の光学ガラスは、ガラスの透過率で表すと、厚み10mmのサンプルで分光透過率70%を示す波長(λ70)は、好ましくは500nm、より好ましくは480nm、より好ましくは450nm、さらに好ましくは420nmを上限とする。
特に、第ニの態様に係る本発明の光学ガラスは、ガラスの透過率で表すと、厚み10mmのサンプルで分光透過率80%を示す波長(λ80)は、好ましくは500nm、より好ましくは480nm、より好ましくは450nm、さらに好ましくは420nmを上限とする。 また、本発明の光学ガラスにおける、厚み10mmのサンプルで分光透過率5%を示す最も短い波長(λ5)は、好ましくは400nm、より好ましくは380nm、さらに好ましくは370nmさらに好ましくは360nmを上限とする。
これらにより、ガラスの吸収端が紫外領域又はその近傍になり、可視光に対するガラスの透明性が高められるため、この光学ガラスを、レンズ等の光を透過させる光学素子に好ましく用いることができる。
作製された光学ガラスから、例えば研磨加工の手段、又は、リヒートプレス成形や精密プレス成形等のモールドプレス成形の手段を用いて、ガラス成形体を作製することができる。すなわち、光学ガラスに対して研削及び研磨等の機械加工を行ってガラス成形体を作製したり、光学ガラスからモールプレス成形用のプリフォームを作製し、このプリフォームに対してリヒートプレス成形を行った後で研磨加工を行ってガラス成形体を作製したり、研磨加工を行って作製したプリフォームや、公知の浮上成形等により成形されたプリフォームに対して精密プレス成形を行ってガラス成形体を作製したりすることができる。なお、ガラス成形体を作製する手段は、これらの手段に限定されない。
また、本発明の第二の態様に係る光学ガラスの実施例(No.44~No.168)及び比較例(C、D)の組成、並びに、これらのガラスの屈折率(nd)、アッベ数(νd)、耐酸性、磨耗度、分光透過率が5%、80%を示す波長(λ5、λ80)、の結果を表8~表25に示す。
なお、以下の実施例はあくまで例示の目的であり、これらの実施例のみ限定されるものではない。
磨耗度={(試料の磨耗質量/比重)/(標準試料の磨耗質量/比重)}×100により計算した。
よって、本発明の実施例の光学ガラスは、摩耗度に優れており、光学ガラスをレンズとして使用する場合に、表面上に傷が生じにくいことが明らかになった。
よって、本発明の実施例の光学ガラスは、摩耗度に優れており、光学ガラスをレンズとして使用する場合に、表面上に傷が生じにくいことが明らかになった。
Claims (12)
- 質量%で、
SiO2成分を10.0%~40.0%、
La2O3成分を15.0 %~50.0%、
TiO2成分 5.0~25.0%未満、
含有し、
質量比B2O3/SiO2が1.00以下であり、
1.78~1.95の屈折率(nd)、25~45のアッベ数(νd)を有し、
粉末法による化学的耐久性(耐酸性)が1級~3級を有する光学ガラス。 - 質量%で、
ZnO成分を0~30.0%、
ZrO2成分 0~20.0%
Al2O3成分 0~20.0%
Y2O3成分 0~25.0%
B2O3成分 0~20.0%
である請求項1に記載の光学ガラス。 - 質量和B2O3+Nb2O5が20.0%未満であり、質量和ZrO2+Nb2O5+WO3+ZnOが25.0%未満である請求項1又は2記載の光学ガラス。
- 質量和TiO2+ZrO2が35.0%未満である請求項1~3のいずれか記載の光学ガラス。
- 質量%で、
SiO2成分を10.0%~50.0%、
La2O3成分を15.0 %~60.0%、
TiO2成分 0 ~15.0%未満、
含有し、
質量比B2O3/SiO2が1.00以下であり、
1.60~1.85の屈折率(nd)、33~62のアッベ数(νd)を有し、
粉末法による化学的耐久性(耐酸性)が1級~3級を有する光学ガラス。 - 質量%で、
ZnO成分を0~35.0%、
ZrO2成分 0~20.0%、
Al2O3成分 0~20.0%、
B2O3成分 0~20.0%、
である請求項5に記載の光学ガラス。 - 質量和B2O3+Nb2O5が20.0%未満である請求項5又は6に記載の光学ガラス。
- Ln2O3成分(式中、LnはLa、Gd、Y、Yb、Luからなる群より選択される1種以上)の質量和が15.0%以上65.0%以下であり、RO成分(式中、RはMg、Ca、Sr、Baからなる群より選択される1種以上)の質量和が25.0%以下であり、Rn2O成分(式中、RnはLi、Na、Kからなる群より選択される1種以上)の質量和が10.0%以下である請求項1~7のいずれか記載の光学ガラス。
- 摩耗度が200以下である請求項1~8のいずれか記載の光学ガラス。
- 請求項1~9のいずれか記載の光学ガラスからなるプリフォーム材。
- 請求項1~10のいずれか記載の光学ガラスからなる光学素子。
- 請求項11に記載の光学素子を備える光学機器。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP20158439.8A EP3677559A1 (en) | 2016-04-04 | 2017-03-15 | Optical glass, preform material and optical element |
| CN201780021626.2A CN108883968B (zh) | 2016-04-04 | 2017-03-15 | 光学玻璃、预成型坯和光学元件 |
| US16/088,698 US20190106352A1 (en) | 2016-04-04 | 2017-03-15 | Optical glass, preform material and optical element |
| JP2018510286A JP7269732B2 (ja) | 2016-04-04 | 2017-03-15 | 光学ガラス、プリフォーム材及び光学素子 |
| EP17778931.0A EP3441372A4 (en) | 2016-04-04 | 2017-03-15 | OPTICAL GLASS, MANUFACTURING MATERIAL AND OPTICAL ELEMENT |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016075377 | 2016-04-04 | ||
| JP2016075378 | 2016-04-04 | ||
| JP2016-075377 | 2016-04-04 | ||
| JP2016-075378 | 2016-04-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017175552A1 true WO2017175552A1 (ja) | 2017-10-12 |
Family
ID=60001132
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/010321 WO2017175552A1 (ja) | 2016-04-04 | 2017-03-15 | 光学ガラス、プリフォーム材及び光学素子 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20190106352A1 (ja) |
| EP (2) | EP3677559A1 (ja) |
| JP (1) | JP7269732B2 (ja) |
| CN (1) | CN108883968B (ja) |
| TW (1) | TWI752949B (ja) |
| WO (1) | WO2017175552A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2020026381A (ja) * | 2018-08-16 | 2020-02-20 | Hoya株式会社 | 光学ガラス、光学素子ブランクおよび光学素子 |
| JP6680424B1 (ja) * | 2019-07-23 | 2020-04-15 | Agc株式会社 | ガラス |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020198176A1 (en) * | 2019-03-25 | 2020-10-01 | Afo Research, Inc. | Alkali free fluorophosphate based glass systems |
| US11787729B2 (en) | 2020-05-18 | 2023-10-17 | Corning Incorporated | Glass compositions with high refractive indexes and low densities |
| CN112745025B (zh) * | 2020-12-31 | 2022-12-06 | 苏州卡利肯新光讯科技有限公司 | 一种用于透镜的钾钡玻璃及其制备方法和应用 |
| CN115304270A (zh) * | 2022-08-26 | 2022-11-08 | 成都光明光电股份有限公司 | 光学玻璃、光学元件和光学仪器 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5950048A (ja) * | 1982-09-16 | 1984-03-22 | Ohara Inc | 光学ガラス |
| JPH06144868A (ja) * | 1992-11-05 | 1994-05-24 | Ohara Inc | 光学ガラス |
| JP2002173334A (ja) | 2000-12-06 | 2002-06-21 | Minolta Co Ltd | 光学ガラス |
| JP2004137145A (ja) * | 2002-09-25 | 2004-05-13 | Nippon Electric Glass Co Ltd | モールドプレス成形用光学ガラス |
| JP2006219365A (ja) * | 2005-01-17 | 2006-08-24 | Ohara Inc | ガラス |
| JP2009269771A (ja) | 2008-04-30 | 2009-11-19 | Ohara Inc | 光学ガラス、光学素子及び光学機器 |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5934140B2 (ja) * | 1980-03-12 | 1984-08-20 | 科学技術庁無機材質研究所長 | 耐アルカリ性アルミノ珪酸塩ガラス |
| BR8605356A (pt) * | 1985-11-29 | 1987-08-04 | Corning Glass Works | Vidro para aplicacoes oticas e/ou oftalmicas |
| FR2682297A1 (fr) * | 1991-10-15 | 1993-04-16 | Tyszblat Michele | Prothese dentaire entierement ceramique a base de spinelle alumine/magnesie et son procede de fabrication. |
| DE4210011C1 (ja) * | 1992-03-27 | 1993-07-15 | Schott Glaswerke, 6500 Mainz, De | |
| JPH1143344A (ja) * | 1997-06-03 | 1999-02-16 | Ohara Inc | 光学ガラス |
| JP4923366B2 (ja) * | 2001-09-13 | 2012-04-25 | 日本電気硝子株式会社 | モールドプレス成形用光学ガラス |
| JP2005350325A (ja) | 2004-06-14 | 2005-12-22 | Ohara Inc | Yb含有ガラス |
| WO2006075785A2 (en) * | 2005-01-17 | 2006-07-20 | Kabushiki Kaisha Ohara | Glass |
| JP4746995B2 (ja) * | 2006-02-02 | 2011-08-10 | 株式会社オハラ | 光学ガラス |
| JP4527193B1 (ja) * | 2009-12-09 | 2010-08-18 | 文化貿易工業株式会社 | 液面計の液位観察窓用ガラス |
| KR101931391B1 (ko) | 2010-10-18 | 2018-12-20 | 오씨브이 인텔렉츄얼 캐피탈 엘엘씨 | 고 굴절율 유리 조성물 |
| JP6553856B2 (ja) * | 2013-12-27 | 2019-07-31 | 株式会社オハラ | ガラス |
| JP2017204317A (ja) * | 2016-05-12 | 2017-11-16 | 日東電工株式会社 | 回路付サスペンション基板およびその製造方法 |
-
2017
- 2017-03-15 WO PCT/JP2017/010321 patent/WO2017175552A1/ja active Application Filing
- 2017-03-15 JP JP2018510286A patent/JP7269732B2/ja active Active
- 2017-03-15 US US16/088,698 patent/US20190106352A1/en not_active Abandoned
- 2017-03-15 CN CN201780021626.2A patent/CN108883968B/zh active Active
- 2017-03-15 EP EP20158439.8A patent/EP3677559A1/en not_active Withdrawn
- 2017-03-15 EP EP17778931.0A patent/EP3441372A4/en not_active Withdrawn
- 2017-03-31 TW TW106111130A patent/TWI752949B/zh active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5950048A (ja) * | 1982-09-16 | 1984-03-22 | Ohara Inc | 光学ガラス |
| JPH06144868A (ja) * | 1992-11-05 | 1994-05-24 | Ohara Inc | 光学ガラス |
| JP2002173334A (ja) | 2000-12-06 | 2002-06-21 | Minolta Co Ltd | 光学ガラス |
| JP2004137145A (ja) * | 2002-09-25 | 2004-05-13 | Nippon Electric Glass Co Ltd | モールドプレス成形用光学ガラス |
| JP2006219365A (ja) * | 2005-01-17 | 2006-08-24 | Ohara Inc | ガラス |
| JP2009269771A (ja) | 2008-04-30 | 2009-11-19 | Ohara Inc | 光学ガラス、光学素子及び光学機器 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3441372A4 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2020026381A (ja) * | 2018-08-16 | 2020-02-20 | Hoya株式会社 | 光学ガラス、光学素子ブランクおよび光学素子 |
| CN110835233A (zh) * | 2018-08-16 | 2020-02-25 | Hoya株式会社 | 光学玻璃、光学元件坯件以及光学元件 |
| JP7305317B2 (ja) | 2018-08-16 | 2023-07-10 | Hoya株式会社 | 光学ガラス、光学素子ブランクおよび光学素子 |
| JP6680424B1 (ja) * | 2019-07-23 | 2020-04-15 | Agc株式会社 | ガラス |
| WO2021014582A1 (ja) * | 2019-07-23 | 2021-01-28 | Agc株式会社 | ガラス |
| US11028005B2 (en) | 2019-07-23 | 2021-06-08 | AGC Inc. | Glass for semiconductor processing |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3441372A4 (en) | 2019-07-31 |
| CN108883968A (zh) | 2018-11-23 |
| TW201806893A (zh) | 2018-03-01 |
| US20190106352A1 (en) | 2019-04-11 |
| TWI752949B (zh) | 2022-01-21 |
| CN108883968B (zh) | 2022-11-29 |
| EP3677559A1 (en) | 2020-07-08 |
| EP3441372A1 (en) | 2019-02-13 |
| JPWO2017175552A1 (ja) | 2019-02-14 |
| JP7269732B2 (ja) | 2023-05-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6409039B2 (ja) | 光学ガラス及び光学素子 | |
| JP6560650B2 (ja) | 光学ガラス及び光学素子 | |
| TWI585056B (zh) | Optical glass and optical components | |
| JP5979723B2 (ja) | 光学ガラス及び光学素子 | |
| JP5767179B2 (ja) | 光学ガラス、プリフォーム及び光学素子 | |
| JP6560651B2 (ja) | 光学ガラス及び光学素子 | |
| WO2013094619A1 (ja) | 光学ガラス及び光学素子 | |
| JP2012229148A (ja) | 光学ガラス及び光学素子 | |
| CN108883968B (zh) | 光学玻璃、预成型坯和光学元件 | |
| JP7075895B2 (ja) | 光学ガラス、プリフォーム及び光学素子 | |
| JP2024003105A (ja) | 光学ガラス、プリフォーム及び光学素子 | |
| JP2017088479A (ja) | 光学ガラス、プリフォーム材及び光学素子 | |
| JP2016216282A (ja) | 光学ガラス及び光学素子 | |
| JP6014573B2 (ja) | 光学ガラス、プリフォーム材及び光学素子 | |
| JPWO2019031095A1 (ja) | 光学ガラス、光学素子及び光学機器 | |
| JP2017171578A (ja) | 光学ガラス及び光学素子 | |
| JP2013209232A (ja) | 光学ガラス及び光学素子 | |
| JP6910702B2 (ja) | 光学ガラス、プリフォーム材及び光学素子 | |
| JP6091251B2 (ja) | 光学ガラス及び光学素子 | |
| JP6033487B2 (ja) | 光学ガラス及び光学素子 | |
| JP6165281B2 (ja) | 光学ガラス及び光学素子 | |
| JP6033488B1 (ja) | 光学ガラス及び光学素子 | |
| JP2017057121A (ja) | 光学ガラス、プリフォーム及び光学素子 | |
| JP6611410B2 (ja) | 光学ガラス、プリフォーム材及び光学素子 | |
| JP2013209233A (ja) | 光学ガラス及び光学素子 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2018510286 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2017778931 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2017778931 Country of ref document: EP Effective date: 20181105 |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17778931 Country of ref document: EP Kind code of ref document: A1 |