WO2014129104A1 - 二酸化マンガンおよびそれを用いたアルカリ乾電池 - Google Patents
二酸化マンガンおよびそれを用いたアルカリ乾電池 Download PDFInfo
- Publication number
- WO2014129104A1 WO2014129104A1 PCT/JP2014/000119 JP2014000119W WO2014129104A1 WO 2014129104 A1 WO2014129104 A1 WO 2014129104A1 JP 2014000119 W JP2014000119 W JP 2014000119W WO 2014129104 A1 WO2014129104 A1 WO 2014129104A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- manganese dioxide
- negative electrode
- peak intensity
- positive electrode
- particle size
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G45/00—Compounds of manganese
- C01G45/02—Oxides; Hydroxides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M6/00—Primary cells; Manufacture thereof
- H01M6/04—Cells with aqueous electrolyte
- H01M6/06—Dry cells, i.e. cells wherein the electrolyte is rendered non-fluid
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/82—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by IR- or Raman-data
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/61—Micrometer sized, i.e. from 1-100 micrometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/62—Submicrometer sized, i.e. from 0.1-1 micrometer
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25B—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES FOR THE PRODUCTION OF COMPOUNDS OR NON-METALS; APPARATUS THEREFOR
- C25B1/00—Electrolytic production of inorganic compounds or non-metals
- C25B1/01—Products
- C25B1/21—Manganese oxides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/023—Gel electrode
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/06—Electrodes for primary cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2982—Particulate matter [e.g., sphere, flake, etc.]
Definitions
- the present invention relates to manganese dioxide, and more particularly to manganese dioxide used as a positive electrode active material for alkaline dry batteries.
- Alkaline batteries are used in various devices. In recent years, with an increase in the load of equipment in which alkaline dry batteries are used, batteries having excellent high-load discharge characteristics have been demanded.
- the positive electrode of an alkaline battery usually contains a mixture (positive electrode mixture) of powder of electrolytic manganese dioxide (hereinafter may be simply referred to as manganese dioxide), graphite powder, and alkaline electrolyte.
- the positive electrode mixture is formed into a hollow cylindrical pellet (positive electrode pellet) at a high pressure and stored in an alkaline battery.
- Electrolytic manganese dioxide may have structural defects due to micro-tuning (folding in the growth direction) that occurs during crystal growth. This structural defect is said to consist of Mn deficiency and protons included as charge compensation thereof, and is known to affect the heavy load discharge performance of alkaline dry batteries. Since the amount of structural defects due to micro-twining varies depending on the electrolytic conditions such as the electrolyte concentration and the electrolytic current density, efforts have been made to increase the structural defects by variously changing the electrolytic conditions. For example, by reducing the weight loss at 200 to 400 ° C. when heating the electrolytic manganese dioxide to 2.7% by weight or more, the internal defects in the electrolytic manganese dioxide are increased and the diffusion of hydrogen ions is facilitated. A technique for improving the heavy load discharge performance has been proposed (see Patent Document 1).
- Patent Document 1 when the heavy load discharge performance is improved by a method of increasing the internal defects of manganese dioxide, the crystallinity decreases with an increase in the amount of defects in manganese dioxide. Therefore, the shape of the obtained particles becomes non-uniform, and the filling property of the positive electrode mixture decreases when the positive electrode pellet is formed. That is, the density or filling amount of the positive electrode active material is lowered, and it is difficult to obtain an alkaline dry battery having a high energy density. In order to improve the performance of an alkaline battery, it is important how much manganese dioxide contained in the positive electrode can be filled in a battery of a certain volume.
- This invention is made
- the place made into the objective provides the alkaline dry battery which has a high energy density, and shows the outstanding heavy load discharge performance, and manganese dioxide used for it. For the purpose.
- the present invention provides a peak intensity ratio between the peak intensity I ⁇ near 525 cm ⁇ 1 and the peak intensity I ⁇ near 580 cm ⁇ 1 measured by Raman spectroscopy spectrum: I ⁇ / I ⁇ is 0.62 or less. Regarding manganese. Thereby, while having a high energy density, the alkaline dry battery which shows the outstanding heavy load discharge performance can be obtained.
- the peak intensity ratio: I ⁇ / I ⁇ is preferably 0.35 to 0.58. Thereby, the heavy load discharge performance is further improved.
- the proportion of particles having a particle size of 0.5 ⁇ m or less is preferably 5% by volume or less, and more preferably 1.2 to 3.8% by volume. This is because the peak intensity ratio: I ⁇ / I ⁇ can be easily controlled.
- the present invention also includes a hollow cylindrical positive electrode containing manganese dioxide as a positive electrode active material, a gelled negative electrode filled in a hollow portion of the positive electrode and containing a negative electrode active material, the positive electrode and the gelled negative electrode.
- the present invention relates to an alkaline dry battery including a separator disposed therebetween, a negative electrode current collector inserted into the gelled negative electrode, a negative electrode terminal plate electrically connected to the negative electrode current collector, and an electrolytic solution.
- Manganese dioxide is usually a mixed crystal of a phase having a Ramsdellite structure and a phase having a pyrolusite structure (hereinafter sometimes referred to as ⁇ phase).
- the ⁇ phase is known to be inactive because it has a very stable crystal structure.
- efforts have been made to increase the proportion of the phase of the Akhtenskit structure (hereinafter sometimes referred to as the ⁇ phase) that further activates the ramsdellite structure, which is an active crystal structure.
- the method of Patent Document 1 is an example. However, as described above, when the proportion of the ⁇ phase is increased, the crystallinity is lowered, so that the filling amount of the positive electrode mixture is lowered.
- the present inventors have found that the ratio between the ramsdellite structure and the pyrolsite structure ( ⁇ phase) easily changes due to various factors at the production site of electrolytic manganese dioxide.
- the inactive ⁇ phase increases due to unavoidable fluctuations in electrolytic current, local potential increase due to cracks generated on the surface of manganese dioxide deposited on the anode of the electrolytic cell, and the like.
- the content ratio of the ramsdellite structure and pyrolsite structure ( ⁇ phase) differs greatly between manganese dioxide deposited on the anode of the electrolytic cell and fine particles appearing on the surface and inside of the manganese dioxide. I stopped. That is, this fine particle contains a pyrolsite structure ( ⁇ phase) in a larger proportion than usual, and manganese dioxide deposited on the anode contains a phase having a ramsdellite structure in a larger proportion than usual. It is.
- the manganese dioxide of the present invention has a peak intensity ratio I ⁇ / I ⁇ between the peak intensity I ⁇ in the vicinity of 525 cm ⁇ 1 and the peak intensity I ⁇ in the vicinity of 580 cm ⁇ 1 measured by Raman spectroscopy spectrum is 0.62 or less. is there.
- Peak intensity ratio By using manganese dioxide having I ⁇ / I ⁇ of 0.62 or less, an alkaline dry battery having high energy density and excellent heavy load discharge performance can be obtained.
- the peak intensity ratio: I ⁇ / I ⁇ is preferably 0.35 to 0.58, more preferably 0.35 to 0.53 or 0.35 to 0.46. When the peak intensity ratio is within this range, a further improvement effect of the heavy load discharge performance can be obtained.
- the peak intensity ratio: I ⁇ / I ⁇ is calculated from the peak height, not the peak area ratio.
- FIG. 1 shows three types of electrolytic manganese dioxide currently produced and sold industrially (conventional example 1: HH-TF grade manufactured by Tosoh Corporation, conventional example 2: MS10 grade manufactured by Quintal).
- FIG. 2 shows a Raman spectroscopic spectrum of Conventional Example 3: Cegasa general battery grade).
- shaft in FIG. 1 has shown the relative intensity
- the Raman spectrum of manganese dioxide, 525 cm -1, 580 cm -1, near 650 cm -1, peak respectively is observed.
- These peaks include peaks derived from phases having different crystal structures.
- 525 cm -1, the peak of 580 cm -1 and 650 cm -1 vicinity is a peak derived from the phase of the ramsdellite structure, 525 cm -1 and 650 cm -1 near the peak phase of pyrolusite structure (beta phase) It is a peak derived from. That is, the peak in the vicinity of 580 cm ⁇ 1 is a peak derived only from the phase of the ramsdellite structure. Also, among the peaks of 525 cm -1 and 650 cm -1 vicinity from phase (beta phase) of pyrolusite structure, the peak of 525 cm -1 vicinity is relatively sharp.
- the peak intensity in the peak intensity I ⁇ in the vicinity 525 cm -1, near 580 cm -1 derived from the phase of the ramsdellite structure Peak intensity ratio with I ⁇ : I ⁇ / I ⁇ is defined.
- “in the vicinity” of the peak position in the Raman spectrum means that the range of ⁇ 15 cm ⁇ 1 is included.
- the peak intensity ratio I ⁇ / I ⁇ in Conventional Examples 1 to 3 was 0.63 to 0.64.
- the peak intensity ratio: I ⁇ / I ⁇ can be determined as follows, for example. First, about 100 mg of manganese dioxide as a sample is put in a tablet molding machine having an inner diameter of 5 mm, and pressed at about 100 kgf / cm 2 to produce pellets. Using a Raman spectroscopic analyzer (for example, NRS-5100, manufactured by JASCO Corporation), before measuring the sample, metal silicon is measured, and the wave number is calibrated so that the peak is 520 cm ⁇ 1 .
- a Raman spectroscopic analyzer for example, NRS-5100, manufactured by JASCO Corporation
- the amount of structural defects due to micro-twinning is not increased, that is, a phase having a ramsdellite structure. Even if the ⁇ phase is not changed, the high-load discharge performance of the obtained alkaline dry battery can be improved. Therefore, it can suppress that the crystallinity of manganese dioxide falls and the filling amount of a positive electrode mixture becomes small.
- one of the parameters affected by the crystallinity of manganese dioxide is the density of manganese dioxide in the positive electrode pellet.
- the density of manganese dioxide in the positive electrode pellet in the AA type battery can be 2.7 to 3.2 g per cm 3 . This is equivalent to the case where the conventional examples 1 to 3 having the peak intensity ratio: I ⁇ / I ⁇ of 0.63 to 0.64 are used.
- the peak intensity ratio I ⁇ / I ⁇ can be easily controlled by controlling the particle diameter of manganese dioxide.
- a titanium plate as an anode and a carbon plate as a negative electrode are placed facing each other in an electrolytic cell having an internal volume of 20 liters provided with a heating device.
- An electrolytic cell provided with a tube was prepared.
- the anode titanium plate on which manganese dioxide was deposited and fixed was taken out and washed with pure water, and the manganese dioxide on the anode titanium plate was peeled off.
- the obtained massive manganese dioxide was coarsely pulverized to about 10 mm, further finely pulverized with a roller mill pulverizer, and further finely pulverized with a mortar.
- the manganese dioxide powder thus obtained was washed with pure water, and the washed fine particles were dry-classified, and the maximum particle size was 10.0 ⁇ m, 1.0 ⁇ m, 0.7 ⁇ m, 0.5 ⁇ m, 0.3 ⁇ m.
- the manganese dioxide powders of Reference Examples 1 to 5 were obtained. About each obtained manganese dioxide, the Raman spectroscopic analysis was performed by the said method, and peak intensity ratio: I (beta) / I (gamma) was computed.
- the peak intensity ratio I ⁇ / I ⁇ of manganese dioxide having a maximum particle diameter of 0.7 ⁇ m to 10.0 ⁇ m was almost the same.
- the peak intensity ratio: I ⁇ / I ⁇ was larger. From this, it is considered that particles having a maximum particle size of 0.5 ⁇ m or less have an influence on the peak intensity ratio: I ⁇ / I ⁇ . This is probably because manganese dioxide inactivated due to excessive potential concentration during electrolysis cannot continue to precipitate on the electrode surface, so that crystallites do not grow sufficiently and appear as fine particles.
- the proportion of particles having a particle size of 0.5 ⁇ m or less is preferably 5% by volume or less, more preferably 1.2 to 3.8% by volume. I can say.
- the proportion of particles having a particle size of 0.5 ⁇ m or less is 3.2% by volume, 2.3% by volume, and 2.9% by volume, respectively.
- the peak intensity ratio: I ⁇ / I ⁇ by Raman spectroscopic analysis is 0.63 to 0.64. That is, the peak intensity ratio: I ⁇ / I ⁇ is not determined only by the ratio of particles having a particle diameter of 0.5 ⁇ m or less.
- the ratio of the ⁇ phase of the obtained manganese dioxide is larger than that obtained by a normal production method, and the ratio of the phase having a ramsdellite structure and the ⁇ phase has a particle size of It seems to be different.
- the heavy load discharge performance of the alkaline dry batteries using the manganese dioxides of Reference Examples 4 and 5 is significantly lowered as the peak intensity ratio: I ⁇ / I ⁇ is increased. That is, it is considered that the heavy load discharge performance was lowered because a large amount of inactive ⁇ phase was contained.
- the maximum particle diameter of the manganese dioxide of the present invention is preferably 100 to 160 ⁇ m, and the average particle diameter D50 is preferably 25 to 40 ⁇ m. When the maximum particle diameter or average particle diameter D50 of manganese dioxide is within this range, a sufficient amount of manganese dioxide can be filled in the positive electrode pellet.
- the average particle diameter D50 is a median diameter in a volume particle size distribution obtained by a laser diffraction particle size distribution measuring apparatus (hereinafter the same).
- the particle size and particle size distribution can be measured, for example, as follows. Using a laser diffraction / scattering type particle size distribution measuring device (for example, LA-920 manufactured by HORIBA), 120 ml of a 0.05 mass% sodium hexametaphosphate aqueous solution is charged into the measuring device as a dispersion medium for manganese dioxide powder as a sample. After circulating the dispersion medium at the maximum circulation speed of the measurement device, operating the ultrasonic transmitter built in the measurement device, introducing about 20 mg of manganese dioxide as a sample, and continuing the dispersion by ultrasonic waves for 3 minutes, Start measuring particle size and particle size distribution. The measurement is performed three times per sample, the average value is calculated, and the particle size and particle size distribution are obtained.
- a laser diffraction / scattering type particle size distribution measuring device for example, LA-920 manufactured by HORIBA
- 120 ml of a 0.05 mass% sodium hexametaphosphate aqueous solution is charged into the
- manganese dioxide When manganese dioxide is taken out from the alkaline battery and the particle size and particle size distribution are measured, it is performed as follows. About 1 g of the positive electrode pellet taken out by disassembling the alkaline dry battery is pulverized and put into a sodium polytungstate aqueous solution having a specific gravity adjusted to about 3.0 to 4.0 g / cm 3 . Thereafter, manganese dioxide and graphite are separated by centrifugation to obtain manganese dioxide powder. As described above, the obtained manganese dioxide powder is dispersed in 120 ml of a 0.05 mass% sodium hexametaphosphate aqueous solution, and measured with a laser diffraction / scattering particle size distribution analyzer.
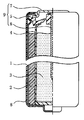
- FIG. 2 is a front view, partly in section, of an AA alkaline battery according to an embodiment of the present invention.
- the alkaline dry battery includes a hollow cylindrical positive electrode 2 disposed in a bottomed cylindrical battery case 1, and a negative electrode such as zinc or zinc alloy powder filled in the hollow portion of the positive electrode 2.
- a gelled negative electrode 3 including an active material, and a separator 4 disposed between the positive electrode 2 and the gelled negative electrode 3 are provided.
- the opening part of the battery case 1 is sealed with the sealing unit 9 comprised by the gasket 5 and the negative electrode terminal plate 7 to which the negative electrode collector 6 was connected.
- the outer surface of the battery case 1 is covered with an exterior label 8.
- the positive electrode 2, the separator 4, and the gelled negative electrode 3 contain an alkaline electrolyte.
- the alkaline electrolyte is, for example, an aqueous potassium hydroxide solution.
- the concentration of potassium hydroxide in the electrolytic solution is preferably 30 to 40% by mass.
- the electrolytic solution may further contain zinc oxide.
- the concentration of zinc oxide in the electrolytic solution is preferably 1 to 3% by mass.
- the positive electrode 2 contains at least manganese dioxide as a positive electrode active material.
- the positive electrode 2 is made of, for example, a mixture of manganese dioxide, a conductive agent, and an alkaline electrolyte. Graphite powder is used as the conductive agent.
- the gelled negative electrode 3 may contain zinc or a zinc alloy as a negative electrode active material.
- the gelled negative electrode 3 is made of, for example, a gelled electrolyte obtained by adding a gelling agent to an alkaline electrolyte and a powdered negative electrode active material dispersed in the gelled electrolyte.
- a gelling agent for example, sodium polyacrylate is used.
- the gasket 5 is obtained, for example, by injection molding nylon or polypropylene into a predetermined size and shape.
- the opening end of the battery case 1 is caulked to the peripheral edge (the flange) of the negative electrode terminal plate 7 via the gasket 5. Thereby, the opening part of the battery case 1 is sealed.
- Procedure (1) Manganese dioxide production In a 20 liter internal electrolytic tank provided with a heating device, a titanium plate as an anode and a carbon plate as a cathode are positioned to face each other, and a manganese sulfate solution (electrolysis) from the upper part of the electrolytic cell An electrolytic cell provided with a pipe for supplying a replenisher was prepared. While maintaining the temperature of the electrolytic cell at 95 ° C.
- the anode titanium plate on which manganese dioxide was deposited and fixed was taken out and washed with pure water, and then the manganese dioxide on the anode titanium plate was peeled off.
- the obtained massive manganese dioxide of about 10 cm square was washed with hot water at 80 to 90 ° C. using a drum washer, and fine particles adhering to the manganese dioxide surface were washed away with hot water.
- the washed lump was dried with a drier maintained at 60 to 80 ° C. Subsequently, the lump was roughly pulverized to about 10 mm, further pulverized with a roller mill pulverizer, washed with water and neutralized, and then filtered with a filter. After preliminarily drying the filtered manganese dioxide, this manganese dioxide was put into an airflow dryer equipped with a cyclone and a bag filter as a recovery device and dried. Next, fine particles collected by the bag filter were mixed with the powder collected by the cyclone at a predetermined ratio, and sufficiently mixed and stirred using a Nauter mixer. Thus, manganese dioxide powders each having a ratio of fine particles having a particle diameter of 0.5 ⁇ m or less and a peak intensity ratio: I ⁇ / I ⁇ having predetermined values shown in Table 2 were prepared.
- manganese dioxide was produced under conditions that caused an electrolysis current fluctuation during electrolysis, and the ratio of particles having a particle diameter of 0.5 ⁇ m or less was changed to obtain a peak intensity ratio: I ⁇ / Although I ⁇ is adjusted, it is not limited to this method.
- Procedure (2) Production of Positive Electrode
- graphite powder (average particle size 8 ⁇ m) as a conductive agent was added to obtain a mixture. .
- the mass ratio of the manganese dioxide powder and the graphite powder was 92.4: 7.6.
- An alkaline electrolyte was added to the mixture, and after sufficiently stirring, the mixture was compression molded into flakes to obtain a positive electrode mixture.
- the mass ratio of the mixture and the alkaline electrolyte was 100: 1.5.
- An alkaline aqueous solution containing 35 mass% potassium hydroxide and 2 mass% zinc oxide was used as the alkaline electrolyte.
- the average particle diameter D50 of the manganese dioxide powder used in Examples 1 to 6 was 26 to 40 ⁇ m.
- the flaky positive electrode mixture was pulverized into granules and classified with a sieve.
- a 10-100 mesh granule is formed into a hollow cylinder with a constant pressure of 3 tons, and a constant volume positive electrode pellet (outer diameter: 13.5 mm, inner diameter: 9.2 mm, height: 22.2 mm) is formed.
- Table 2 shows the filling amounts of two positive electrode pellets (corresponding to one battery) that could be formed with a constant pressure and a constant volume.
- Step (3) Production of Negative Electrode Zinc alloy powder, which is a negative electrode active material, the above alkaline electrolyte, and a gelling agent are mixed at a mass ratio of 200: 100: 2.1 to obtain a gelled negative electrode 3.
- a zinc alloy a zinc alloy containing 0.02 mass% indium, 0.01 mass% bismuth, and 0.005 mass% aluminum was used.
- Step (4) Assembling the alkaline battery
- the bunny height made by Nippon Graphite Co., Ltd. was applied to the inner surface of the bottomed cylindrical battery case made of nickel-plated steel sheet to form a carbon film with a thickness of about 10 ⁇ m. 1 was obtained.
- Two positive electrode pellets produced in the procedure (2) were inserted into the battery case 1 and then pressed to form the positive electrode 2 in close contact with the inner surface of the battery case 1. After placing the bottomed cylindrical separator 4 inside the positive electrode 2, 1.50 g of the same alkaline electrolyte used in the procedure (2) was injected, and the separator 4 was impregnated.
- the peak intensity ratio: I ⁇ / I ⁇ was calculated by the method described above. According to the above procedures (1) to (4), AA alkaline batteries of Examples 1 to 6 and Comparative Examples 1 to 2 were produced and evaluated. The results are shown in Table 2. Moreover, the Raman spectrum of the manganese dioxide used in Example 3 is also shown in FIG.
- the filling property of the positive electrode pellet will be considered.
- the batteries of Examples 1 to 6 using manganese dioxide having a peak intensity ratio: I ⁇ / I ⁇ of 0.62 or less when forming positive electrode pellets of the same size with a constant applied pressure, almost the same as Comparative Examples 1 and 2
- the same amount of positive electrode mixture could be filled. That is, it can be seen that even if the proportion of ⁇ phase in manganese dioxide is reduced, the crystallinity is not greatly changed, and the filling property of the positive electrode pellet is not lowered.
- the alkaline dry battery using manganese dioxide according to the present invention has excellent heavy load discharge characteristics, it is suitably used for devices with high current consumption. While this invention has been described in terms of the presently preferred embodiments, such disclosure should not be construed as limiting. Various changes and modifications will no doubt become apparent to those skilled in the art to which the present invention pertains after reading the above disclosure. Accordingly, the appended claims should be construed to include all variations and modifications without departing from the true spirit and scope of this invention.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Primary Cells (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
- Electrolytic Production Of Non-Metals, Compounds, Apparatuses Therefor (AREA)
Abstract
Description
アルカリ乾電池の正極は、通常、電解二酸化マンガン(以下、単に二酸化マンガンと称する場合がある)の粉末と黒鉛の粉末とアルカリ電解液との混合物(正極合剤)を含む。正極合剤は、中空円筒状のペレット(正極ペレット)に高圧成形され、アルカリ乾電池の中に収納されている。
マイクロツイニングによる構造欠陥量は、電解液濃度、電解電流密度などの電解条件によって変化するため、電解条件を様々に変化させて構造欠陥を増加させる取り組みが従来からなされてきた。例えば、電解二酸化マンガンを加熱した際の200~400℃での重量の減少を2.7重量%以上とすることで、電解二酸化マンガン中の内部欠陥を増加させて、水素イオンの拡散を容易にし、強負荷放電性能を向上させる技術が提案されている(特許文献1参照)。
本発明の新規な特徴を添付の請求の範囲に記述するが、本発明は、構成および内容の両方に関し、本発明の他の目的および特徴と併せ、図面を照合した以下の詳細な説明によりさらによく理解されるであろう。
ピーク強度比:Iβ/Iγは、例えば、以下のようにして求めることができる。
まず、試料である二酸化マンガン約100mgを、内径5mmの錠剤成型器に入れ、約100kgf/cm2で加圧してペレットを作製する。ラマン分光分析装置(例えば、日本分光(株)製、NRS-5100)を用い、試料を測定する前に、金属シリコンを測定し、そのピークが520cm-1となるように波数を校正する。次いで、試料(ペレット)をセットし、レーザー波長532nm、レーザー出力約5mW、対物レンズ20倍、レーザー照射時間120秒、積算2回の測定条件で、1試料につき5回測定を行い、その平均スペクトルを用いてピーク強度を得る。最後に、580cm-1近傍にあるピーク強度Iγと525cm-1近傍にあるピーク強度Iβとのピーク強度比:Iβ/Iγを計算する。
粒子径および粒度分布は、例えば、以下のようにして測定することができる。
レーザー回折/散乱式粒度分布測定装置(例えば、HORIBA製のLA-920)を用い、試料である二酸化マンガン粉末の分散媒として、ヘキサメタリン酸ナトリウム0.05質量%水溶液120mlを測定装置に投入する。測定装置の最大循環速度で分散媒を循環させ、測定装置に内蔵された超音波発信機を作動させて、試料である二酸化マンガン約20mgを投入し、3分間超音波による分散を継続した後、粒子径および粒度分布の測定を開始する。1試料につき3回測定を行い、その平均値を算出し、粒子径および粒度分布とする。
図2は、本発明の一実施形態における単3形のアルカリ乾電池の一部を断面にした正面図である。
図2に示すように、アルカリ乾電池は、有底円筒状の電池ケース1内に配設された中空円筒状の正極2と、正極2の中空部内に充填された亜鉛または亜鉛合金粉末等の負極活物質を含むゲル状負極3と、正極2とゲル状負極3との間に配されたセパレータ4とを備えている。そして、電池ケース1の開口部は、ガスケット5と、負極集電体6が接続された負極端子板7とで構成された封口ユニット9で封口されている。電池ケース1の外表面には外装ラベル8が被覆されている。
以下、実施例により、本発明の一実施形態を詳細に説明する。なお、本発明は、以下の実施形態に限定されるものではない。また、本発明の効果を奏する範囲を逸脱しない範囲で、適宜変更は可能である。さらに、他の実施形態との組み合わせも可能である。
加温装置を設けた内容積20リットルの電解槽に、陽極としてチタン板、陰極としてカーボン板をそれぞれ向かい合うように位置させ、電解槽上部より硫酸マンガン溶液(電解補給液)を補給する為の管を設けた電解槽を準備した。
電解槽の温度を95℃に保ち、硫酸マンガン溶液を電解槽に注入して、電解中の電解液の組成が2価マンガン濃度40g/l、硫酸濃度55g/lとなるように調整しながら、電流密度30A/m2において、55分間電解し、5分間休止させる電解工程を1サイクルとして、繰り返し行った。
手順(1)で得られた各々の二酸化マンガンの粉末(最大粒子径150μm)に、導電剤である黒鉛の粉末(平均粒子径8μm)を加え、混合物を得た。二酸化マンガンの粉末および黒鉛の粉末の質量比は92.4:7.6とした。混合物にアルカリ電解液を加え、充分に攪拌した後、フレーク状に圧縮成形して、正極合剤を得た。混合物およびアルカリ電解液の質量比は100:1.5とした。アルカリ電解液には、35質量%の水酸化カリウムおよび2質量%の酸化亜鉛を含むアルカリ水溶液を用いた。なお、実施例1~6で用いた二酸化マンガンの粉末の平均粒子径D50は、26~40μmであった。
負極活物質である亜鉛合金粉末と、上記のアルカリ電解液と、ゲル化剤とを、200:100:2.1の質量比で混合し、ゲル状の負極3を得た。亜鉛合金には、0.02質量%のインジウムと、0.01質量%のビスマスと、0.005質量%のアルミニウムとを含む亜鉛合金を用いた。
ニッケルめっき鋼板製の有底円筒形の電池ケースの内面に、日本黒鉛(株)製のバニーハイトを塗布して、厚み約10μmの炭素被膜を形成し、電池ケース1を得た。電池ケース1内に、手順(2)で作製した正極ペレットを2個挿入した後、加圧して、電池ケース1の内面に密着した状態の正極2を形成した。有底円筒形のセパレータ4を正極2の内側に配置した後、手順(2)で使用したのと同じアルカリ電解液を1.50g注入し、セパレータ4に含浸させた。この状態で15分間放置し、アルカリ電解液をセパレータ4から正極2へ浸透させた。その後、手順(3)で得られたゲル状の負極3を、セパレータ4の内側に6.00g充填した。そして、ガスケット5、負極集電体6、および負極端子板7を一体化した封口ユニット9で、電池ケース1の開口部を封口し、外装ラベル8で電池ケース1の外表面を被覆して、所定のアルカリ乾電池を得た。
得られたアルカリ乾電池を室温にて7日間静置した後、以下の条件で放電試験を行った。20±1℃の環境下で、電池1個を1500mWで2秒間放電させ、その後650mWで28秒間放電させた。これを1パターンとして10回、合計5分間行い、その後55分間休止した。この1時間の工程を1サイクルとして、電池の閉路電圧が1.05Vになるまでのサイクル回数を計測した。計測は5個の電池で行い、これらの平均値を算出して、強負荷放電特性を評価した。
以上の手順(1)~(4)により、実施例1~6および比較例1~2の単3形のアルカリ乾電池を作製し、各評価を行った。結果を表2に示す。また、実施例3で使用した二酸化マンガンのラマン分光スペクトルを、図1に併せて示す。
また、ピーク強度比:Iβ/Iγが0.62以下の二酸化マンガンを用いた実施例1~6の電池では、ピーク強度比:Iβ/Iγが0.64である比較例1および2の電池と比べて、明らかに強負荷放電性能が向上している。特に、実施例1~5の、ピーク強度比:Iβ/Iγが0.35~0.58の範囲にある二酸化マンガンを用いた電池では、強負荷放電性能がさらに向上している。すなわち、β相を減少させた二酸化マンガンを用いた本発明のアルカリ乾電池によれば、強負荷放電特性の向上効果が得られることが明らかとなった。なお、実施例1(Iβ/Iγ=0.35)および実施例2(Iβ/Iγ=0.38)の結果から、ピーク強度比:Iβ/Iγが0.35の付近で、強負荷放電特性の向上効果は飽和すると考えられる。
本発明を現時点での好ましい実施態様に関して説明したが、そのような開示を限定的に解釈してはならない。種々の変形および改変は、上記開示を読むことによって本発明に属する技術分野における当業者には間違いなく明らかになるであろう。したがって、添付の請求の範囲は、本発明の真の精神および範囲から逸脱することなく、すべての変形および改変を包含する、と解釈されるべきものである。
2 正極
3 負極
4 セパレータ
5 ガスケット
6 負極集電体
7 負極端子板
8 外装ラベル
9 封口ユニット
Claims (5)
- ラマン分光スペクトルで測定される525cm-1近傍のピーク強度Iβと、580cm-1近傍のピーク強度Iγとのピーク強度比:Iβ/Iγが、0.62以下である、二酸化マンガン。
- 前記ピーク強度比:Iβ/Iγが、0.35~0.58である、請求項1に記載の二酸化マンガン。
- 粒子径が0.5μm以下である粒子の割合が、5体積%以下である粒度分布を有する、請求項1に記載の二酸化マンガン。
- 粒子径が0.5μm以下である粒子の割合が、1.2~3.8体積%である粒度分布を有する、請求項1に記載の二酸化マンガン。
- 請求項1~4のいずれか1項に記載の二酸化マンガンを正極活物質として含む中空円筒状の正極と、
前記正極の中空部内に充填され、負極活物質を含むゲル状負極と、
前記正極と前記ゲル状負極との間に配されるセパレータと、
前記ゲル状負極に挿入される負極集電体と、
前記負極集電体と電気的に接続される負極端子板と、
電解液と、を具備するアルカリ乾電池。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014542030A JP5899456B2 (ja) | 2013-02-19 | 2014-01-14 | 二酸化マンガンおよびそれを用いたアルカリ乾電池 |
| US14/383,075 US20140377608A1 (en) | 2013-02-19 | 2014-01-14 | Manganese dioxide and alkaline dry battery |
| EP14754072.8A EP2811556A4 (en) | 2013-02-19 | 2014-01-14 | MANGANO DIOXIDE AND ALKALI DRY BATTERY THEREWITH |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-029607 | 2013-02-19 | ||
| JP2013029607 | 2013-02-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014129104A1 true WO2014129104A1 (ja) | 2014-08-28 |
Family
ID=51390914
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/000119 WO2014129104A1 (ja) | 2013-02-19 | 2014-01-14 | 二酸化マンガンおよびそれを用いたアルカリ乾電池 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20140377608A1 (ja) |
| EP (1) | EP2811556A4 (ja) |
| JP (1) | JP5899456B2 (ja) |
| WO (1) | WO2014129104A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016181589A1 (ja) * | 2015-05-14 | 2016-11-17 | パナソニックIpマネジメント株式会社 | 正極材料およびその製造方法、その正極材料を用いたマンガン乾電池 |
| JP2018076222A (ja) * | 2016-10-31 | 2018-05-17 | 東ソー株式会社 | 電解二酸化マンガン及びその用途 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019044771A1 (ja) * | 2017-08-28 | 2019-03-07 | 株式会社村田製作所 | 非水電解液電池および通信機器 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02145434A (ja) * | 1988-11-25 | 1990-06-04 | Chuo Denki Kogyo Kk | 結晶構造が調整された二酸化マンガンとその製造方法 |
| JP2002289186A (ja) * | 2001-03-23 | 2002-10-04 | Tosoh Corp | 電解二酸化マンガン粉末およびその製造法 |
| JP2002348693A (ja) * | 2000-09-01 | 2002-12-04 | Tosoh Corp | 電解二酸化マンガン粉末及びその製造法 |
| JP2004186127A (ja) | 2002-10-11 | 2004-07-02 | Mitsui Mining & Smelting Co Ltd | 電池用正極活物質及び電解二酸化マンガンの製造方法並びに電池 |
| JP2008066100A (ja) * | 2006-09-07 | 2008-03-21 | Matsushita Electric Ind Co Ltd | アルカリ電池 |
| JP2010251221A (ja) * | 2009-04-20 | 2010-11-04 | Tosoh Corp | 優れたハイレート特性を有する電解二酸化マンガン組成物 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU778788B2 (en) * | 2000-09-01 | 2004-12-23 | Tosoh Corporation | Powder of electrolytic manganese dioxide and process for producing the same |
| CA2453266A1 (en) * | 2001-07-19 | 2003-01-30 | Matsushita Electric Industrial Co., Ltd. | Alkaline dry battery |
| US9103044B2 (en) * | 2009-08-24 | 2015-08-11 | Tosoh Corporation | Electrolytic manganese dioxide, and method for its production and its application |
| WO2012111766A1 (ja) * | 2011-02-18 | 2012-08-23 | 東ソー株式会社 | 電解二酸化マンガン及びその製造方法、並びにリチウムマンガン系複合酸化物の製造方法 |
-
2014
- 2014-01-14 US US14/383,075 patent/US20140377608A1/en not_active Abandoned
- 2014-01-14 JP JP2014542030A patent/JP5899456B2/ja active Active
- 2014-01-14 EP EP14754072.8A patent/EP2811556A4/en not_active Withdrawn
- 2014-01-14 WO PCT/JP2014/000119 patent/WO2014129104A1/ja active Application Filing
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02145434A (ja) * | 1988-11-25 | 1990-06-04 | Chuo Denki Kogyo Kk | 結晶構造が調整された二酸化マンガンとその製造方法 |
| JP2002348693A (ja) * | 2000-09-01 | 2002-12-04 | Tosoh Corp | 電解二酸化マンガン粉末及びその製造法 |
| JP2002289186A (ja) * | 2001-03-23 | 2002-10-04 | Tosoh Corp | 電解二酸化マンガン粉末およびその製造法 |
| JP2004186127A (ja) | 2002-10-11 | 2004-07-02 | Mitsui Mining & Smelting Co Ltd | 電池用正極活物質及び電解二酸化マンガンの製造方法並びに電池 |
| JP2008066100A (ja) * | 2006-09-07 | 2008-03-21 | Matsushita Electric Ind Co Ltd | アルカリ電池 |
| JP2010251221A (ja) * | 2009-04-20 | 2010-11-04 | Tosoh Corp | 優れたハイレート特性を有する電解二酸化マンガン組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2811556A4 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016181589A1 (ja) * | 2015-05-14 | 2016-11-17 | パナソニックIpマネジメント株式会社 | 正極材料およびその製造方法、その正極材料を用いたマンガン乾電池 |
| JP2018076222A (ja) * | 2016-10-31 | 2018-05-17 | 東ソー株式会社 | 電解二酸化マンガン及びその用途 |
| JP7039927B2 (ja) | 2016-10-31 | 2022-03-23 | 東ソー株式会社 | 電解二酸化マンガン及びその用途 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2811556A1 (en) | 2014-12-10 |
| US20140377608A1 (en) | 2014-12-25 |
| JPWO2014129104A1 (ja) | 2017-02-02 |
| JP5899456B2 (ja) | 2016-04-06 |
| EP2811556A4 (en) | 2014-12-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Wei et al. | High reversible pseudocapacity in mesoporous yolk–shell anatase TiO2/TiO2 (B) microspheres used as anodes for Li‐ion batteries | |
| Zhao et al. | Improving the Ni-rich LiNi0. 5Co0. 2Mn0. 3O2 cathode properties at high operating voltage by double coating layer of Al2O3 and AlPO4 | |
| US9440861B2 (en) | Method for modification of lithium ion battery positive electrode material | |
| JP6493853B2 (ja) | リチウムニッケルコバルトアルミニウム酸化物複合正極材料、その製造方法及びリチウムイオン二次電池 | |
| CN111554919A (zh) | 正极活性材料、其制备方法及钠离子电池 | |
| CN104900862B (zh) | 对称钠离子二次电池的 p2 相层状电极材料及制备方法 | |
| Idris et al. | Effects of polypyrrole on the performance of nickel oxide anode materials for rechargeable lithium-ion batteries | |
| CN104993109B (zh) | 一种液相物理法制备石墨烯/纳米硅锂离子电池负极材料的方法 | |
| CN107275606A (zh) | 一种碳包覆尖晶石锰酸锂纳米复合材料及制备方法与应用 | |
| CN104979541B (zh) | 一种钛酸锂复合材料及其制备方法 | |
| CN105845904B (zh) | 一种钠离子电池金属氧化物/聚吡咯空心纳米管负极复合材料及其制备方法 | |
| CN109148875A (zh) | 一种高镍正极材料及其制备方法 | |
| CN106299294A (zh) | 一种二氧化锡纳米晶/二氧化钛纳米管复合材料的制备方法 | |
| CN106450305A (zh) | 一种锂离子电池负极材料CoP/C的制备方法 | |
| JP5899456B2 (ja) | 二酸化マンガンおよびそれを用いたアルカリ乾電池 | |
| CN109037552A (zh) | 一种用于钠硫电池的隔膜材料的制备方法 | |
| CN109767925A (zh) | 用于锂离子超级电容器的T-Nb2O5/蛋清碳复合材料及其制备方法 | |
| TWI651272B (zh) | 一種富鋰-鋰鎳錳氧化物陰極複合材料的製備方法及其用途 | |
| KR102240050B1 (ko) | 리튬 이온 전지의 애노드 | |
| CN105609746B (zh) | 一种同时电化学贮钠和贮锂的复合电极及其制备方法 | |
| CN113184898B (zh) | 一种二氧化锡-碳核壳纳米球复合材料、其制备方法及应用 | |
| Huang et al. | Mitigating first charge overpotential of Li2S-based lithium-sulfur batteries by leveraging PVDF reaction with the LiOH/Li2O layer | |
| CN106450186A (zh) | 一种锂离子电池正极材料硅酸锰锂/碳复合材料的制备方法、正极浆料及应用 | |
| CN112072116A (zh) | 一种四氧化三钴掺杂碳包覆二氧化锡复合材料及其制备方法与应用 | |
| CN105540658B (zh) | 一种二氧化钛负极材料及其制备方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2014542030 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014754072 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14383075 Country of ref document: US |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14754072 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |