WO2014125797A1 - 硬質シート及び硬質シートの製造方法 - Google Patents
硬質シート及び硬質シートの製造方法 Download PDFInfo
- Publication number
- WO2014125797A1 WO2014125797A1 PCT/JP2014/000616 JP2014000616W WO2014125797A1 WO 2014125797 A1 WO2014125797 A1 WO 2014125797A1 JP 2014000616 W JP2014000616 W JP 2014000616W WO 2014125797 A1 WO2014125797 A1 WO 2014125797A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- hardness
- fiber
- elastic body
- hard sheet
- polymer elastic
- Prior art date
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B24—GRINDING; POLISHING
- B24B—MACHINES, DEVICES, OR PROCESSES FOR GRINDING OR POLISHING; DRESSING OR CONDITIONING OF ABRADING SURFACES; FEEDING OF GRINDING, POLISHING, OR LAPPING AGENTS
- B24B37/00—Lapping machines or devices; Accessories
- B24B37/11—Lapping tools
- B24B37/20—Lapping pads for working plane surfaces
- B24B37/22—Lapping pads for working plane surfaces characterised by a multi-layered structure
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B24—GRINDING; POLISHING
- B24B—MACHINES, DEVICES, OR PROCESSES FOR GRINDING OR POLISHING; DRESSING OR CONDITIONING OF ABRADING SURFACES; FEEDING OF GRINDING, POLISHING, OR LAPPING AGENTS
- B24B37/00—Lapping machines or devices; Accessories
- B24B37/11—Lapping tools
- B24B37/20—Lapping pads for working plane surfaces
- B24B37/24—Lapping pads for working plane surfaces characterised by the composition or properties of the pad materials
-
- D—TEXTILES; PAPER
- D04—BRAIDING; LACE-MAKING; KNITTING; TRIMMINGS; NON-WOVEN FABRICS
- D04H—MAKING TEXTILE FABRICS, e.g. FROM FIBRES OR FILAMENTARY MATERIAL; FABRICS MADE BY SUCH PROCESSES OR APPARATUS, e.g. FELTS, NON-WOVEN FABRICS; COTTON-WOOL; WADDING ; NON-WOVEN FABRICS FROM STAPLE FIBRES, FILAMENTS OR YARNS, BONDED WITH AT LEAST ONE WEB-LIKE MATERIAL DURING THEIR CONSOLIDATION
- D04H1/00—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres
- D04H1/40—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties
- D04H1/42—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties characterised by the use of certain kinds of fibres insofar as this use has no preponderant influence on the consolidation of the fleece
- D04H1/4382—Stretched reticular film fibres; Composite fibres; Mixed fibres; Ultrafine fibres; Fibres for artificial leather
- D04H1/43825—Composite fibres
- D04H1/4383—Composite fibres sea-island
-
- D—TEXTILES; PAPER
- D04—BRAIDING; LACE-MAKING; KNITTING; TRIMMINGS; NON-WOVEN FABRICS
- D04H—MAKING TEXTILE FABRICS, e.g. FROM FIBRES OR FILAMENTARY MATERIAL; FABRICS MADE BY SUCH PROCESSES OR APPARATUS, e.g. FELTS, NON-WOVEN FABRICS; COTTON-WOOL; WADDING ; NON-WOVEN FABRICS FROM STAPLE FIBRES, FILAMENTS OR YARNS, BONDED WITH AT LEAST ONE WEB-LIKE MATERIAL DURING THEIR CONSOLIDATION
- D04H1/00—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres
- D04H1/40—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties
- D04H1/42—Non-woven fabrics formed wholly or mainly of staple fibres or like relatively short fibres from fleeces or layers composed of fibres without existing or potential cohesive properties characterised by the use of certain kinds of fibres insofar as this use has no preponderant influence on the consolidation of the fleece
- D04H1/4382—Stretched reticular film fibres; Composite fibres; Mixed fibres; Ultrafine fibres; Fibres for artificial leather
- D04H1/43838—Ultrafine fibres, e.g. microfibres
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/564—Polyureas, polyurethanes or other polymers having ureide or urethane links; Precondensation products forming them
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06N—WALL, FLOOR, OR LIKE COVERING MATERIALS, e.g. LINOLEUM, OILCLOTH, ARTIFICIAL LEATHER, ROOFING FELT, CONSISTING OF A FIBROUS WEB COATED WITH A LAYER OF MACROMOLECULAR MATERIAL; FLEXIBLE SHEET MATERIAL NOT OTHERWISE PROVIDED FOR
- D06N3/00—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof
- D06N3/0002—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof characterised by the substrate
- D06N3/0004—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof characterised by the substrate using ultra-fine two-component fibres, e.g. island/sea, or ultra-fine one component fibres (< 1 denier)
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06N—WALL, FLOOR, OR LIKE COVERING MATERIALS, e.g. LINOLEUM, OILCLOTH, ARTIFICIAL LEATHER, ROOFING FELT, CONSISTING OF A FIBROUS WEB COATED WITH A LAYER OF MACROMOLECULAR MATERIAL; FLEXIBLE SHEET MATERIAL NOT OTHERWISE PROVIDED FOR
- D06N3/00—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof
- D06N3/0002—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof characterised by the substrate
- D06N3/0011—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof characterised by the substrate using non-woven fabrics
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06N—WALL, FLOOR, OR LIKE COVERING MATERIALS, e.g. LINOLEUM, OILCLOTH, ARTIFICIAL LEATHER, ROOFING FELT, CONSISTING OF A FIBROUS WEB COATED WITH A LAYER OF MACROMOLECULAR MATERIAL; FLEXIBLE SHEET MATERIAL NOT OTHERWISE PROVIDED FOR
- D06N3/00—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof
- D06N3/12—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof with macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. gelatine proteins
- D06N3/14—Artificial leather, oilcloth or other material obtained by covering fibrous webs with macromolecular material, e.g. resins, rubber or derivatives thereof with macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. gelatine proteins with polyurethanes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/35—Abrasion, pilling or fibrillation resistance
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06N—WALL, FLOOR, OR LIKE COVERING MATERIALS, e.g. LINOLEUM, OILCLOTH, ARTIFICIAL LEATHER, ROOFING FELT, CONSISTING OF A FIBROUS WEB COATED WITH A LAYER OF MACROMOLECULAR MATERIAL; FLEXIBLE SHEET MATERIAL NOT OTHERWISE PROVIDED FOR
- D06N2211/00—Specially adapted uses
- D06N2211/08—Cleaning articles
Definitions
- Patent Documents 5 to 14 disclose a nonwoven fabric type polishing pad obtained by impregnating a nonwoven fabric with wet-coagulated porous polyurethane. Since the nonwoven fabric type polishing pad is excellent in flexibility, the polishing pad is easily deformed. For this reason, since it is difficult for the load to be selectively applied to the abrasive grains agglomerated on the polished surface, scratches are hardly generated. However, since the non-woven polishing pad is flexible, the polishing rate is low. Moreover, since the nonwoven fabric type polishing pad deforms following the surface shape of the substrate to be polished, the flattening performance, which is a property of flattening the substrate to be polished, was low.
- Patent Documents 15 to 18 below disclose polishing pads having a high leveling performance including a nonwoven fabric of ultrafine fibers.
- Patent Document 15 discloses a polishing pad made of a sheet-like material obtained by impregnating a non-woven fabric obtained by entanglement of polyester microfiber bundles having an average fineness of 0.0001 to 0.01 dtex with a polymer elastic body mainly composed of polyurethane. Is disclosed. This document discloses that such a polishing pad achieves a polishing process with higher accuracy than before.
- a polishing pad having a high polishing rate and having a polishing rate that hardly changes over time is provided.
- thermoplastic resin that forms the island component a thermoplastic resin that is not dissolved or removed by water, an alkaline aqueous solution, an acidic aqueous solution, or the like and that can be melt-spun is used.
- the water-insoluble thermoplastic resin include the above-described various resins that form ultrafine fibers, preferably the thermoplastic resin having a Tg of 50 ° C. or more and a water absorption of 4% by mass or less.
- Non-water-soluble thermoplastic resins include, for example, catalysts, anti-coloring agents, heat-resistant agents, flame retardants, lubricants, antifouling agents, fluorescent whitening agents, matting agents, coloring agents, gloss improvers, antistatic agents, You may contain additives, such as a fragrance
- a silicone-based oil agent or a mineral oil-based oil agent such as a needle breakage preventing oil agent, an antistatic oil agent, or an entanglement improving oil agent is applied to the web.
- a web is entangled with a needle punch.
- the basis weight of the entangled web is preferably in the range of 100 to 1500 g / m 2 from the viewpoint of excellent handleability.
- a non-porous polymer elastic body can be formed.
- the polymer elastic body is preferably a hydrogen-bonding polymer elastic body from the viewpoint of high adhesion to fibers.
- the hydrogen-bonding polymer elastic body is an elastic body made of a polymer that crystallizes or aggregates by hydrogen bonding, such as polyurethane, polyamide-based elastic body, and polyvinyl alcohol-based elastic body.
- polyurethane examples include various polyurethanes obtained by reacting a polymer polyol having an average molecular weight of 200 to 6000, an organic polyisocyanate, and a chain extender in a predetermined molar ratio.
- organic polyisocyanate examples include, for example, non-yellowing diisocyanates such as aliphatic or alicyclic diisocyanates such as hexamethylene diisocyanate, isophorone diisocyanate, norbornene diisocyanate, 4,4′-dicyclohexylmethane diisocyanate; 2,4-tri Examples thereof include aromatic diisocyanates such as diisocyanate, 2,6-tolylene diisocyanate, 4,4′-diphenylmethane diisocyanate, and xylylene diisocyanate polyurethane.
- non-yellowing diisocyanates such as aliphatic or alicyclic diisocyanates such as hexamethylene diisocyanate, isophorone diisocyanate, norbornene diisocyanate, 4,4′-dicyclohexylmethane diisocyanate
- 2,4-tri Examples thereof include aromatic diisocyanates such as
- chain extender examples include, for example, diamines such as hydrazine, ethylenediamine, propylenediamine, hexamethylenediamine, nonamethylenediamine, xylylenediamine, isophoronediamine, piperazine and derivatives thereof, adipic acid dihydrazide, and isophthalic acid dihydrazide; Triamines such as diethylenetriamine; tetramines such as triethylenetetramine; ethylene glycol, propylene glycol, 1,4-butanediol, 1,6-hexanediol, 1,4-bis ( ⁇ -hydroxyethoxy) benzene, 1,4 -Diols such as cyclohexanediol; Triols such as trimethylolpropane; Pentaols such as pentaerythritol; Aminoethyl alcohol, Aminopropyl alcohol It includes amino alcohols such as and the like
- a carbodiimide group and / or an oxazoline group are particularly preferable because they are excellent in crosslinking performance and pot life of the emulsion, and have no problem in safety.
- the crosslinking agent having a carbodiimide group include water-dispersed carbodiimide compounds such as “Carbodilite E-01”, “Carbodilite E-02”, and “Carbodilite V-02” manufactured by Nisshinbo Industries, Ltd.
- the content of the polymer polyol component in the polyurethane is preferably 65% by mass or less, and more preferably 60% by mass or less. Moreover, it is preferable from the point which can suppress generation
- the method for preparing the polyurethane emulsion is not particularly limited, and a known method can be used. Specifically, for example, by using a monomer having a hydrophilic group such as a carboxyl group, a sulfonic acid group, and a hydroxyl group as a copolymerization component, a method of imparting self-emulsifying property to water to a polyurethane, or a polyurethane A method of emulsifying by adding a surfactant is mentioned. Since the polymer elastic body containing a monomer unit having a hydrophilic group as a copolymerization component is excellent in water wettability, it can hold a large amount of slurry.
- a monomer having a hydrophilic group such as a carboxyl group, a sulfonic acid group, and a hydroxyl group
- the surfactant used for emulsification include, for example, sodium lauryl sulfate, ammonium lauryl sulfate, polyoxyethylene tridecyl ether sodium acetate, sodium dodecylbenzene sulfonate, alkyl diphenyl ether sodium disulfonate, sodium dioctyl sulfosuccinate and the like.
- Anionic surfactants; nonionic surfactants such as polyoxyethylene nonylphenyl ether, polyoxyethylene octylphenyl ether, polyoxyethylene lauryl ether, polyoxyethylene stearyl ether, polyoxyethylene-polyoxypropylene block copolymer Etc.
- heat-sensitive gelation property can also be provided to an emulsion by selecting suitably the cloud point of surfactant.
- the solid content concentration of the emulsion is preferably 15 to 40% by mass, and more preferably 25 to 35% by mass, from the viewpoint that the polymer entangled sheet can be uniformly filled with a polymer elastic body in the thickness direction.
- the particle size of the emulsion is preferably 0.01 to 1 ⁇ m, more preferably 0.03 to 0.5 ⁇ m.
- the first emulsion contains a gelling agent containing ions that cause a pH change of water.
- the gelling agent is used to gel emulsion particles by heating by changing the pH of the emulsion.
- the moisture in the emulsion impregnated in the nonwoven fabric dries from the surface. Therefore, it is easy to cause migration in which the emulsion in the nonwoven fabric moves to the surface layer as the evaporation of moisture proceeds.
- the polymer elastic body migrates, the polymer elastic body is unevenly distributed in the vicinity of the surface layer of the nonwoven fabric, the polymer elastic body in the vicinity of the middle layer is reduced, and voids are likely to remain in the vicinity of the middle layer.
- the gelling agent is not particularly limited as long as it is a water-soluble salt that changes the pH of the emulsion to such an extent that the emulsion particles are gelled by heating.
- Specific examples thereof are monovalent or divalent inorganic salts such as sodium sulfate, ammonium sulfate, sodium carbonate, calcium chloride, calcium sulfate, calcium nitrate, zinc oxide, zinc chloride, magnesium chloride, potassium chloride, potassium carbonate.
- sodium nitrate, lead nitrate and the like are monovalent or divalent inorganic salts such as sodium sulfate, ammonium sulfate, sodium carbonate, calcium chloride, calcium sulfate, calcium nitrate, zinc oxide, zinc chloride, magnesium chloride, potassium chloride, potassium carbonate. Sodium nitrate, lead nitrate and the like.
- the content ratio of the gelling agent in the first emulsion is 0.5 to 5 parts by mass, more preferably 0.6 to 4 parts by mass with respect to 100 parts by mass of the elastic polymer. It is preferable from the viewpoint that it can be imparted moderately.
- the first emulsion is a penetrant, an antifoaming agent, a lubricant, a water repellent, an oil repellent, a thickener, a bulking agent, a curing accelerator, an antioxidant, an ultraviolet absorber, a fluorescent agent, an antifungal agent, and a foaming agent.
- Water-soluble polymer compounds such as polyvinyl alcohol and carboxymethyl cellulose, dyes, pigments, inorganic fine particles and the like may be further contained.
- the method of impregnating the fiber emulsion sheet with the first emulsion is not particularly limited, and for example, a method such as dip nip, knife coating, bar coating, or roll coating may be used.
- the first emulsion gels in the fiber entangled sheet by heating.
- a heating condition for gelation for example, a condition of holding at 40 to 90 ° C., further 50 to 80 ° C. for about 0.5 to 5 minutes, is preferably used.
- the polymer elastic body is solidified by heating and drying.
- Examples of heat drying include a method of heat drying in a drying apparatus such as a hot air dryer, a method of heat drying in a dryer after infrared heating, and the like.
- Examples of the heating and drying conditions include conditions in which heating is performed for 2 to 10 minutes so that the maximum temperature is 130 to 160 ° C., further 135 to 150 ° C.
- the sea-island type composite fiber contained in the fiber entangled sheet impregnated with the polymer elastic body is subjected to ultrafine fiber treatment to form a first composite containing a nonwoven fabric of ultrafine fibers and a polymer elastic body.
- ultrafine fibers are formed from a sea-island composite fiber containing a water-soluble thermoplastic resin that is an island component and a water-insoluble thermoplastic resin that is a sea component, by ultrafine fiber removal treatment that removes the water-soluble thermoplastic resin. It is a process.
- the ultra-fine fiber treatment is performed by dissolving and removing the water-soluble thermoplastic resin forming the sea component by subjecting the fiber entangled sheet containing the sea-island type composite fiber to hydrothermal heating treatment with water, an alkaline aqueous solution, an acidic aqueous solution, or the like. This is a process of disassembling and removing.
- the fiber entangled sheet is immersed in hot water of 65 to 90 ° C. for 5 to 300 seconds as the first step, and then further 85 to 100 ° C. as the second step.
- a method of treating in hot water for 100 to 600 seconds is preferably used.
- the water-soluble thermoplastic resin is dissolved from the sea-island composite fiber to form ultrafine fibers.
- the ultrafine fibers are greatly crimped.
- the fiber density of the ultrafine fibers becomes dense.
- a void is formed in the portion where the water-soluble thermoplastic resin was present. This void is filled with a polymer elastic body in a later step.
- the gelling agent contained in the fiber entangled sheet is dissolved and removed in the hot water by subjecting the fiber entangled sheet to hot water heating treatment. In this way, the first complex is formed.
- the second emulsion After impregnating the first composite with the second emulsion containing the gelling agent and the polymer elastic body, the second emulsion is gelled and further heated and dried to obtain the polymer elastic body. Solidifying to form a second composite
- the voids in the first composite are filled with a polymer elastic body to restrain the ultrafine fibers.
- the fine fibers can be focused and the porosity of the hard sheet can be reduced.
- the ultrafine fibers form a fiber bundle, the emulsion is easily impregnated by capillary action.
- each layer in order from any one of the surface sides, becomes the first surface layer, the middle layer, and the second surface layer, It is preferable that the second emulsion is applied for gelation so that the difference in porosity between the surface layer and the middle layer is 5% or less, further 3% or less.
- the porosity of each layer is obtained as follows. A cross section in the thickness direction of the second composite is photographed with a scanning electron microscope at a magnification of 30 times. Then, using the image analysis software Popimaging (manufactured by Digital being kids. Co), the obtained photograph is binarized by a dynamic threshold method to identify the void. Then, an inscribed circle is drawn in each void portion, and the total area of the inscribed circle is defined as the total layer void amount.
- the total area of the inscribed circle was determined for each layer, and the amount of voids in each layer was obtained.
- the porosity of each layer the amount of voids of each layer / the amount of voids of all layers ⁇ 100 (%).
- the method of impregnating the first emulsion into the first composite, the method of gelation, and the method of heat drying are the same as the method of impregnation of the first emulsion, the method of gelation, and the method of heat drying.
- a method may be used. In this way, a second complex is formed.
- the hard sheet of the present embodiment uses an emulsion containing a gelling agent in order to suppress migration of the emulsion to the surface layer when a polymer elastic body is applied to the nonwoven fabric.
- the present inventors have found that when a large amount of ions contained in the gelling agent remains in the obtained hard sheet, the polishing rate is reduced during polishing. And it discovered that the fall of a grinding
- the step of washing with water is carried out so that the total content of ions causing the pH change of the water contained in the hard sheet is 400 ⁇ g / cm 3 or less, preferably 350 ⁇ g / cm 3 , more preferably 100 ⁇ g / cm 3 or less. It is a process to do.
- a heated water washing treatment is preferable from the viewpoint of high water washing efficiency.
- the second composite is immersed in hot water of 80 ° C. or higher. Specifically, for example, as a first step, after being immersed in hot water at 65 to 90 ° C. for 5 to 300 seconds, and further as a second step, treatment is performed in hot water at 85 to 100 ° C. for 100 to 600 seconds. Conditions.
- the void existing inside the hard sheet reduces the hardness and hardness uniformity.
- the voids are reduced by hot pressing the first composite, the second composite, and / or the hard sheet.
- the hot press treatment conditions are preferably such that the ultrafine fibers and the polymer elastic body are pressed at a linear pressure of 30 to 100 kg / cm with a metal roll heated to 160 to 180 ° C., for example.
- the hard sheet of this embodiment is preferably used as a polishing layer of a polishing pad.
- the polishing layer can be formed by performing desired processing on the hard sheet as necessary. For example, brushing with sandpaper or needle cloth, diamond, reverse brushing, hot pressing or embossing is performed.
- grooves, holes such as lattices, concentric circles, and spirals may be formed on the surface of the hard sheet.
- an elastic body layer such as a knitted fabric, a woven fabric, a nonwoven fabric, an elastic resin film, or an elastic sponge body may be laminated with the hard sheet as a polishing layer.
- elastic films and elastic sponge bodies non-woven fabrics impregnated with polyurethane currently used for general purposes (for example, “Suba400” (manufactured by Nitta Haas Co., Ltd.)), natural rubber, nitrile rubber, polybutadiene rubber, Examples thereof include rubbers such as silicone rubbers; thermoplastic elastomers such as polyester-based thermoplastic elastomers, polyamide-based thermoplastic elastomers, and fluorine-based thermoplastic elastomers; foamed plastics; polyurethanes and the like.
- the polishing pad is one in which the polishing layer and the elastic body layer are directly bonded by fusion bonding or the like, in which both layers are bonded by an adhesive or a double-sided adhesive tape, or between the two layers. In addition, those having another layer are also included.
- the polishing pad using the hard sheet of the present embodiment is a chemical mechanical polishing (CMP) in which a known CMP apparatus is used and the surface to be polished and the polishing pad are brought into contact with each other at a constant speed under a pressure through a slurry.
- CMP chemical mechanical polishing
- the slurry contains components such as a liquid medium such as water and oil; an abrasive such as silica, aluminum oxide, cerium oxide, zirconium oxide, and silicon carbide; a base, an acid, and a surfactant.
- the article to be polished is not particularly limited, and examples thereof include crystal, silicon, glass, an optical substrate, an electronic circuit substrate, a multilayer wiring substrate, and a hard disk.
- the object to be polished is preferably a silicon wafer or a semiconductor wafer.
- semiconductor wafers include, for example, insulating films such as silicon oxide, silicon oxyfluoride, and organic polymers, wiring metal films such as copper, aluminum, and tungsten, and barrier metals such as tantalum, titanium, tantalum nitride, and titanium nitride. Examples thereof include those having a film or the like on the surface.
- the apparent density (g / cm 3 ) was obtained by dividing the mass (g / cm 2 ) per unit area of the hard sheet by the thickness (cm). Then, the apparent density was measured at any ten locations on the hard sheet, and the value obtained by arithmetic averaging was defined as the apparent density. The thickness was measured at a load of 240 gf / cm 2 according to JISL1096.
- the D hardness of the surface of the hard sheet, the first surface layer and the middle layer was measured according to JIS K 7311. Specifically, the D hardness of the surface of the hard sheet is obtained by stacking eight hard sheets having a thickness of about 1.25 mm, measuring the hardness at three points evenly in the width direction, and calculating the average of the D hardness of the surface of the hard sheet. Hardness. The D hardness of the first surface layer was obtained by grinding a hard sheet having a thickness of about 1.25 mm from the second surface layer side to obtain a sheet of the first surface layer having a thickness of 0.40 mm.
- the hardness at three points was measured uniformly in the width direction, and the average was defined as the JIS-D hardness of the first surface layer.
- the D hardness of the middle layer was obtained by uniformly grinding the hard sheet from the first surface layer side and the second surface layer side to obtain a middle layer sheet having a thickness of 0.40 mm.
- 25 sheets of the obtained middle layer were piled up, the hardness of 3 points
- R (%) (D hardness maximum value ⁇ D hardness minimum value) / D hardness average value ⁇ 100.
- Total ion content causing water pH change A piece of a hard sheet cut into a strip shape and 10 mL of water were cut and placed in a screw test tube. And the water-soluble substance in a hard sheet was hot-water-extracted by heating a screw opening test tube with a block heater at 90 degreeC for 2 hours. Then, ion components in the extract were detected using an ion chromatograph (ICS-1600). In this, the total amount of sulfate ions and ammonium ions, which are ions that cause the pH change of water, was measured and converted to the amount of ions contained in the hard sheet per unit volume.
- ICS-1600 ion chromatograph
- the hard sheet was cut into a circular shape having a diameter of 51 cm, and grooves having a width of 1.0 mm and a depth of 0.5 mm were formed on the surface in a lattice pattern at intervals of 15.0 mm to prepare a polishing pad. Then, after sticking an adhesive tape on the back surface of the polishing pad, it was mounted on a CMP polishing apparatus (“PP0-60S” manufactured by Nomura Seisakusho Co., Ltd.).
- slurry (SHOROXA-31 manufactured by Showa Denko KK) was supplied at a rate of 100 ml / min under the conditions of a platen rotation rate of 70 rpm, a head rotation rate of 69 rpm, and a polishing pressure of 40 g / cm 2.
- synthetic quartz having a diameter of 4 inches was polished for 3 hours.
- polishing was measured, and the grinding
- Example 1 Water-soluble PVA was used as the sea component, and isophthalic acid-modified PET having a modification degree of 6 mol% was used as the island component.
- Water-soluble PVA and isophthalic acid-modified PET were discharged from a melt compound spinning die (number of islands: 25 islands / fiber) at 260 ° C. so as to be 25/75 (mass ratio). Then, the ejector pressure was adjusted so that the spinning speed was 3700 m / min, and long fibers having a fineness of 3 dtex were collected on a net to obtain a web having a basis weight of 35 g / m 2 .
- the entangled web was steamed for 70 seconds at 110 ° C. and 23.5% RH.
- the area shrinkage rate at this time was 44%.
- a fiber entangled sheet having a basis weight of 1312 g / m 2 , an apparent density of 0.544 g / cm 3 , and a thickness of 2.41 mm Got.
- Polyurethane is a mixture of polycarbonate polyol and polyalkylene glycol having 2 to 3 carbon atoms in a ratio of 99.8: 0.2 (molar ratio) as a polyol component, and 1.5% by mass of a carboxyl group-containing monomer. It is a non-yellowing polyurethane contained. Polyurethane is a non-porous polyurethane that forms a crosslinked structure by heat treatment.
- the first emulsion contains 4.6 parts by mass of a carbodiimide-based crosslinking agent and 1.8 parts by mass of ammonium sulfate as a gelling agent with respect to 100 parts by mass of polyurethane, and is adjusted so that the solid content of polyurethane is 20%. It is.
- the fiber entangled sheet impregnated with the first emulsion was heated at 90 ° C. in a 30% RH atmosphere to gel the first emulsion, and further dried at 150 ° C. Further, by hot pressing at 140 ° C., the basis weight was adjusted to 1403 g / m 2 , the apparent density 0.716 g / cm 3 , and the thickness 1.96 mm.
- the fiber-entangled sheet provided with polyurethane is immersed in hot water at 95 ° C. for 10 minutes to dissolve and remove the water-soluble PVA, and have a fineness of 0.09 dtex. It was converted to ultrafine fibers and further dried. In this way, a first composite having a basis weight of 1009 g / m 2 , an apparent density of 0.538 g / cm 3 , and a thickness of 1.87 mm was obtained.
- the first composite was impregnated with a polyurethane emulsion (solid content 30% by mass) as the second emulsion.
- the polyurethane is the same as the previously impregnated polyurethane.
- the second emulsion contains 4.6 parts by mass of a carbodiimide-based crosslinking agent and 1.0 part by mass of ammonium sulfate with respect to 100 parts by mass of polyurethane, and is adjusted so that the solid content of polyurethane is 30%.
- the first composite impregnated with the second emulsion was heated at 90 ° C. in a 60% RH atmosphere to gel the second emulsion, and further dried at 150 ° C. In this way, a second composite having a basis weight of 1245 g / m 2 , an apparent density of 0.748 g / cm 3 and a thickness of 1.66 mm was obtained. The difference in porosity between the first surface layer and the middle layer of the second composite was 1.8%.
- the second composite was washed with water by immersing it in hot water at 95 ° C. for 10 minutes using nip treatment and high-pressure water flow treatment. And it dried at 180 degreeC.
- seat which is 1212 g / m ⁇ 2 > of fabric weights, the apparent density of 0.795 g / cm ⁇ 3 >, and the thickness of 1.53 mm was obtained by carrying out the hot press process on linear pressure of 100 kg / cm and 160 degreeC conditions.
- the surface layers on both sides of the intermediate of the hard sheet are ground by 0.15 mm each using # 100 paper to obtain a hard sheet having a basis weight of 994 g / m 2 , an apparent density of 0.788 g / cm 3 and a thickness of 1.26 mm. Finished.
- the JIS-D hardness of the hard sheet was 52, and R% of the JIS-D hardness was 11.3%.
- the total amount of sulfate ions and ammonium ions, which are ions that cause pH change, contained in the hard sheet was 26.9 ⁇ g / cm 3 .
- Example 2 A hard sheet was produced and evaluated in the same manner as in Example 1 except that the first composite before applying the second emulsion was subjected to hot press treatment under the conditions of a linear pressure of 100 kg / cm and a temperature of 160 ° C.
- the obtained hard sheet had a basis weight of 996 g / m 2 , an apparent density of 0.808 g / cm 3 , and a thickness of 1.23 mm. The results are shown in Table 1.
- Example 3 In Example 1, a hard sheet was produced and evaluated in the same manner as in Example 1 except that the water washing degree of the second composite was lowered. The total amount of sulfate ions and ammonium ions, which are ions that cause pH change, contained in the hard sheet was 300 ⁇ g / cm 3 . The results are shown in Table 1.
- Example 1 In Example 1, instead of washing the second composite by immersing it in 95 ° C. hot water for 10 minutes, it was the same as in Example 1 except that the second composite was not washed with water. Were manufactured and evaluated. The results are shown in Table 1.
- Example 2 In Example 1, the first composite was further hot-pressed under the conditions of a linear pressure of 100 kg / cm and a temperature of 160 ° C. Then, instead of impregnating the second emulsion containing the gelling agent, a hard sheet was produced and evaluated in the same manner as in Example 1 except that the emulsion having the same composition not containing the gelling agent was impregnated.
- the obtained hard sheet had a basis weight of 969 g / m 2 , an apparent density of 0.817 g / cm 3 , and a thickness of 1.19 mm. The results are shown in Table 1.
- Example 4 In Example 1, a hard sheet was produced and evaluated in the same manner as in Example 1 except that the water washing degree of the second composite was lowered. The total amount of sulfate ions and ammonium ions, which are ions that cause pH change, contained in the hard sheet was 504 ⁇ g / cm 3 . The results are shown in Table 1.
- the hard sheet of Comparative Example 2 is obtained by homogenizing the hard sheet by hot pressing instead of blending the second emulsion with a gelling agent and uniformly filling the polymer elastic body.
- the polishing pad of Comparative Example 2 had a small total content of ions, but was inhomogeneous with an R% of 30.2%. As a result, only 89% of the initial polishing rate was maintained on average after 5 hours. Further, the total content of ions Comparative Example 4 Comparative Example 3, and 504 ⁇ g / cm 3 of 404 ⁇ g / cm 3 were not only able to maintain about 84% of the initial polishing rate either an average of up to 5 hours .
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Mechanical Engineering (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dispersion Chemistry (AREA)
- Finish Polishing, Edge Sharpening, And Grinding By Specific Grinding Devices (AREA)
- Nonwoven Fabrics (AREA)
- Computer Hardware Design (AREA)
- Manufacturing & Machinery (AREA)
- General Physics & Mathematics (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Power Engineering (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Physics & Mathematics (AREA)
- Mechanical Treatment Of Semiconductor (AREA)
- Synthetic Leather, Interior Materials Or Flexible Sheet Materials (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
- Reinforced Plastic Materials (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Laminated Bodies (AREA)
Abstract
Description
R(%)=(D硬度最大値―D硬度最小値)/D硬度平均値×100、から算出されるR%が0~20%であり、かつ、水のpH変化を生じさせるイオンの総含有量が400μg/cm3以下である硬質シートである。
R(%)=(6点中のD硬度最大値-6点中のD硬度最小値)/6点のD硬度平均値×100、から算出されるR%が0~20%である。また、好ましくは、第2表層5及び中層4のJIS-D硬度を任意の点でそれぞれ3点ずつ計6点測定し、計6点のD硬度を用いて上記式から算出されるR%も0~20%である。そして、水のpH変化を生じさせるイオンの総含有量が400μg/cm3以下である硬質シートである。
R(%)=(D硬度最大値―D硬度最小値)/D硬度平均値×100、から算出されるR%が0~20%になるように、厚み方向に均質になるように調整されている。また、好ましくは、第2表層5及び中層2のJIS-D硬度をそれぞれ3点ずつ計6点測定し、計6点のD硬度を用いて、算出されたR%も0~20%になるように、厚み方向に均質になるように調整されていることが好ましい。このように、硬度が均質になるように調整されていることにより、均質な研磨が可能になる。
また、複数の繊維束同士は、繊維束の外側に存在する高分子弾性体により結着され、さらには繊維束の半数以上の束が高分子弾性体で結着されて、塊(バルク)状に存在していることが好ましい。繊維束同士が結着されることにより、硬質シートの形態安定性が向上して研磨安定性が向上する。高分子弾性体で極細繊維を集束したり、繊維束同士を結着したりすることにより、硬度が均質に高い硬質シートが得られる。
次に、硬質シートの製造方法の一例について詳しく説明する。硬質シートは、例えば、次のような工程を経て製造することができる。
本工程においては、極細繊維発生型繊維の長繊維の繊維絡合シートを準備する。極細繊維発生型繊維の長繊維の繊維絡合シートは、例えば、次のように製造できる。
(収縮処理前の絡合されたウェブの面積-収縮処理後の絡合されたウェブの面積)/収縮処理前の絡合されたウェブの面積×100
第1表層と中層との空隙率の差(%)=絶対値(中層の空隙率(%)-第1表層の空隙率(%))
各層の空隙率=各層空隙量/全層空隙量×100(%)、の式により求める。
硬質シートの単位面積あたりの質量(g/cm2)を厚さ(cm)で除した値を見掛け密度(g/cm3)とした。そして、硬質シートの任意の10箇所について見掛け密度を測定して算術平均した値を見掛け密度とした。なお、厚さは、JISL1096に準じて荷重240gf/cm2で測定した。
JIS K 7311に準じて硬質シートの表面、第1表層及び中層のD硬度を測定した。具体的には、硬質シートの表面のD硬度は、厚さ約1.25mmの硬質シートを8枚重ね、幅方向に均等に3点の硬度を測定し、その平均を硬質シートの表面のD硬度とした。
また、第1表層のD硬度は、厚さ約1.25mmの硬質シートを第2表層側から研削することにより、厚さ0.40mmの第1表層のシートを得た。そして、得られた第1表層のシートを25枚重ね、幅方向に均等に3点の硬度を測定し、その平均を第1表層のJIS-D硬度とした。さらに、中層のD硬度は、硬質シートを第1表層側及び第2表層側から均等に研削することにより厚さ0.40mmの中層のシートを得た。そして、得られた中層のシートを25枚重ね、幅方向に均等に3点の硬度を測定し、その平均を中層の硬度とした。そして、得られた第1表層の3点のD硬度と中層の3点のD硬度の計6点のJIS-D硬度の値を用いて以下の式:
R(%)=(D硬度最大値-D硬度最小値)/D硬度平均値×100、から、R(%)を求めた。
短冊状に切断した硬質シートの断片及び水10mLを切りネジ口試験管に入れた。そして、ネジ口試験管をブロックヒーターで90℃、2時間加熱することにより、硬質シート中の水溶性物質を熱水抽出した。そして、その抽出液中のイオン成分をイオンクロマトグラフ(ICS-1600)を用いて検出した。この中で水のpH変化を生じるイオンである硫酸イオン及びアンモニウムイオンの合計量を測定し、単位体積あたりの硬質シートに含まれるイオン量に換算した。
硬質シートを直径51cmの円形状に切断し、表面に幅1.0mm、深さ0.5mmの溝を格子状に15.0mm間隔で形成することにより研磨パッドを作成した。そして研磨パッドの裏面に粘着テープを貼り付けた後、CMP研磨装置((株)野村製作所製「PP0-60S」)に装着した。次に、プラテン回転数70回転/分、ヘッド回転数69回転/分、研磨圧力40g/cm2の条件で、スラリー(昭和電工(株)製のSHOROXA-31)を100ml/分の速度で供給しながら、直径4インチの合成石英を3時間研磨した。そして、研磨後の合成石英の面内の任意の25点の厚みを測定し、各点における研磨された厚さの平均を研磨時間で除することにより研磨レート(nm/分)を求めた。
なお、厚さ約1.25mmの硬質シートの第1表層、及び、中層を露出させた厚さ0.70mmの硬質シートの研磨レートをそれぞれ測定した。
水溶性PVAを海成分、変性度6モル%のイソフタル酸変性PETを島成分として用いた。水溶性PVA及びイソフタル酸変性PETを25/75(質量比)になるように、260℃で溶融複合紡糸用口金(島数:25島/繊維)から吐出した。そして、紡糸速度が3700m/minとなるようにエジェクター圧力を調整し、繊度3dtexの長繊維をネット上に捕集し、目付35g/m2のウェブを得た。
第2のエマルジョンを付与する前の第1の複合体を、線圧100kg/cm、160℃条件で熱プレス処理した以外は実施例1と同様して硬質シートを製造し、評価した。なお、得られた硬質シートは、目付996g/m2、見掛け密度0.808g/cm3、厚み1.23mmであった。結果を表1に示す。
実施例1において、第2の複合体の水洗度合いを低くした以外は実施例1と同様して硬質シートを製造し、評価した。硬質シート中に含まれていた、pH変化を生じるイオンである硫酸イオン及びアンモニウムイオンを合計量は、300μg/cm3であった。結果を表1に示す。
実施例1において、第2の複合体を95℃の熱水中に10分間浸漬することにより水洗する代わりに、第2の複合体を水洗しなかった以外は実施例1と同様して硬質シートを製造し、評価した。結果を表1に示す。
実施例1において、第1の複合体をさらに線圧100kg/cm、160℃条件で熱プレス処理した。そして、ゲル化剤を含む第2のエマルジョンを含浸させる代わりに、ゲル化剤を含まない同様の組成のエマルジョンを含浸させた以外は実施例1と同様して硬質シートを製造し、評価した。なお、得られた硬質シートは、目付969g/m2、見掛け密度0.817g/cm3、厚み1.19mmであった。結果を表1に示す。
実施例1において、第2の複合体の水洗度合いを低くした以外は実施例1と同様して硬質シートを製造し、評価した。硬質シート中に含まれていた、pH変化を生じるイオンである硫酸イオン及びアンモニウムイオンを合計量は、404μg/cm3であった。結果を表1に示す。
実施例1において、第2の複合体の水洗度合いを低くした以外は実施例1と同様して硬質シートを製造し、評価した。硬質シート中に含まれていた、pH変化を生じるイオンである硫酸イオン及びアンモニウムイオンを合計量は、504μg/cm3であった。結果を表1に示す。
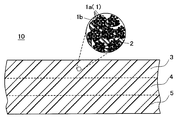
1a 極細繊維
1b 繊維束
2 高分子弾性体
3 第1表層
4 中層
5 第2表層
Claims (16)
- 0.0001~0.5dtexの繊度を有する極細繊維の不織布と、前記不織布に付与された高分子弾性体とを含む硬質シートであって、
JIS-D硬度が45以上であり、
厚み方向の断面において、均等に3分割したときの各層を、何れか一方の表面側から順に、第1表層、中層及び第2表層とした場合、
前記第1表層及び前記中層のJIS-D硬度を任意の点でそれぞれ3点ずつで計6点測定し、計6点のD硬度を用いて、下記式:
R(%)=(D硬度最大値-D硬度最小値)/D硬度の平均値×100
から算出されるR%が0~20%であり、
かつ、水のpH変化を生じさせるイオンの総含有量が400μg/cm3以下であることを特徴とする硬質シート。 - 前記イオンの総含有量が1~100μg/cm3である請求項1に記載の硬質シート。
- 前記極細繊維は長繊維であり、且つ、繊維束を形成している請求項1に記載の硬質シート。
- 前記不織布の見掛け密度が、0.35~0.90g/cm3である請求項3に記載の硬質シート。
- 厚み方向の断面において、前記繊維束を形成する前記極細繊維の少なくとも一部分が前記高分子弾性体で集束されている請求項3に記載の硬質シート。
- 厚み方向の断面において、前記繊維束の少なくとも一部分が、互いに前記高分子弾性体で結着されている請求項5に記載の硬質シート。
- 厚み方向の断面において、前記繊維束を形成する前記極細繊維の半数以上の本数が前記高分子弾性体で集束されている請求項3に記載の硬質シート。
- 厚み方向の断面において、前記繊維束の半数以上の束が互いに前記高分子弾性体で結着されている請求項7に記載の硬質シート。
- 前記高分子弾性体が、非多孔質の高分子弾性体である請求項1に記載の硬質シート。
- 前記不織布と前記高分子弾性体との質量比率(不織布/高分子弾性体)が、90/10~55/45である請求項1に記載の硬質シート。
- 見掛け密度が0.50~1.2g/cm3である請求項10に記載の硬質シート。
- 前記第2表層のJIS-D硬度が45以上であり、
前記第2表層及び前記中層のJIS-D硬度を任意の点でそれぞれ3点ずつで計6点測定し、計6点のD硬度を用いて、下記式:
R(%)=(D硬度最大値―D硬度最小値)/D硬度平均値×100から
算出されるR%が0~20%である請求項1に記載の硬質シート。 - 請求項1~12のいずれか1項に記載の硬質シートを研磨層として備えることを特徴とする研磨パッド。
- (1) 0.5dtex以下の繊度を有する極細繊維の見掛け密度0.35g/cm3以上の不織布を極細繊維化処理により形成しうる、極細繊維発生型繊維の長繊維の繊維絡合シートを準備する工程と、
(2)前記繊維絡合シートに、水のpH変化を生じさせるイオンを含むゲル化剤及び高分子弾性体を含む第1のエマルジョンを含浸させた後、前記第1のエマルジョンをゲル化させ、さらに、加熱乾燥することにより前記高分子弾性体を凝固させる工程と、
(3)前記極細繊維発生型繊維を極細繊維化処理することにより、前記不織布と前記高分子弾性体とを含有する第1の複合体を形成する工程と、
(4)前記第1の複合体に前記ゲル化剤及び前記高分子弾性体を含む第2のエマルジョンを含浸させ、さらに、加熱乾燥することにより前記高分子弾性体を凝固させて、厚み方向に均等に3分割したときの各層を、何れか一方の表面側から順に、第1表層、中層及び第2表層とした場合、前記第1表層と前記中層との空隙率の差が5%以下である第2の複合体を形成する工程と、
(5)前記第2の複合体を前記イオンの総含有量が400μg/cm3以下になるように水洗することにより硬質シートを得る工程と、
(6)前記硬質シートの表面硬度をJIS-D硬度45以上にするために、前記第1の複合体、前記第2の複合体、及び前記硬質シートから選ばれる少なくとも一つを熱プレスする工程と、を備えることを特徴とする硬質シートの製造方法。 - 前記イオンの総含有量が1~100μg/cm3である請求項14に記載の硬質シートの製造方法。
- 前記極細繊維発生型繊維が、海成分として水溶性熱可塑性ポリビニルアルコール系樹脂、島成分として非水溶性熱可塑性樹脂を含む海島型複合繊維であり、
前記工程(3)の極細繊維化処理が、前記水溶性熱可塑性ポリビニルアルコール系樹脂を温水に溶解させて選択的に除去する工程である請求項14に記載の硬質シートの製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/767,115 US20160002835A1 (en) | 2013-02-12 | 2014-02-05 | Hard sheet and method for producing the same |
| KR1020157023576A KR102136441B1 (ko) | 2013-02-12 | 2014-02-05 | 경질 시트 및 경질 시트의 제조 방법 |
| EP14752242.9A EP2957672B1 (en) | 2013-02-12 | 2014-02-05 | Hard sheet and method for producing the same |
| CN201480007963.2A CN105008614B (zh) | 2013-02-12 | 2014-02-05 | 硬质片材及硬质片材的制造方法 |
| JP2015500137A JP6220378B2 (ja) | 2013-02-12 | 2014-02-05 | 硬質シート及び硬質シートの製造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013024525 | 2013-02-12 | ||
| JP2013-024525 | 2013-02-12 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014125797A1 true WO2014125797A1 (ja) | 2014-08-21 |
Family
ID=51353810
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/000616 WO2014125797A1 (ja) | 2013-02-12 | 2014-02-05 | 硬質シート及び硬質シートの製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20160002835A1 (ja) |
| EP (1) | EP2957672B1 (ja) |
| JP (1) | JP6220378B2 (ja) |
| KR (1) | KR102136441B1 (ja) |
| CN (1) | CN105008614B (ja) |
| TW (1) | TWI607832B (ja) |
| WO (1) | WO2014125797A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019021897A1 (ja) * | 2017-07-25 | 2019-01-31 | ニッタ・ハース株式会社 | 研磨布 |
| JP2020104197A (ja) * | 2018-12-27 | 2020-07-09 | 株式会社クラレ | 繊維複合研磨パッドおよびそれを用いたガラス系基材の研磨方法 |
| WO2021131590A1 (ja) * | 2019-12-26 | 2021-07-01 | 株式会社クラレ | 銀付皮革様シート |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107431467B (zh) * | 2015-04-17 | 2021-02-19 | 株式会社村田制作所 | 谐振电路、带阻滤波器以及带通滤波器 |
| DE102016222063A1 (de) * | 2016-11-10 | 2018-05-17 | Siltronic Ag | Verfahren zum beidseitigen Polieren einer Halbleiterscheibe |
| US20180134918A1 (en) * | 2016-11-11 | 2018-05-17 | Jh Rhodes Company, Inc. | Soft polymer-based material polishing media |
| WO2020044911A1 (ja) * | 2018-08-27 | 2020-03-05 | 株式会社クラレ | 人工皮革基材、その製造方法及び立毛人工皮革 |
| US20220380976A1 (en) * | 2019-12-20 | 2022-12-01 | Toray Industries, Inc. | Sheet material and method for producing same |
| WO2021125029A1 (ja) * | 2019-12-20 | 2021-06-24 | 東レ株式会社 | シート状物およびその製造方法 |
Citations (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03234475A (ja) | 1990-02-08 | 1991-10-18 | Kanebo Ltd | 研磨布 |
| JPH10128674A (ja) | 1996-10-28 | 1998-05-19 | Rooder Nitta Kk | 研磨用パッド |
| JPH10225864A (ja) | 1997-02-17 | 1998-08-25 | Sony Corp | 研磨パッドとその製造方法並びにその研磨パッドを用いたウエハの研磨方法 |
| JPH1199479A (ja) | 1997-09-30 | 1999-04-13 | Teijin Ltd | 研磨パッド |
| JPH11322878A (ja) | 1998-05-13 | 1999-11-26 | Dainippon Ink & Chem Inc | 泡含有ポリウレタン成形物の製造方法、泡含有成形物用ウレタン樹脂組成物及びそれを用いた研磨パッド |
| JP2000178374A (ja) | 1998-12-15 | 2000-06-27 | Toyo Tire & Rubber Co Ltd | ポリウレタン発泡体の製造方法及び研磨シート |
| JP2000248034A (ja) | 1999-03-02 | 2000-09-12 | Mitsubishi Chemicals Corp | 研磨材用ポリウレタン系樹脂組成物及びその発泡体 |
| US6130163A (en) * | 1999-06-03 | 2000-10-10 | Promos Technologies, Inc. | Stabilization of slurry used in chemical mechanical polishing of semiconductor wafers by adjustment of PH of deionized water |
| JP2001089548A (ja) | 1999-09-22 | 2001-04-03 | Toyo Tire & Rubber Co Ltd | ポリウレタン発泡体の製造方法及び研磨シート |
| JP2002009026A (ja) | 2000-06-21 | 2002-01-11 | Toray Ind Inc | 研磨用パッドおよびそれを用いた研磨装置及び研磨方法 |
| JP2002172555A (ja) | 2000-12-05 | 2002-06-18 | Teijin Ltd | 研磨用基布および研磨方法 |
| JP2003170347A (ja) | 2001-12-06 | 2003-06-17 | Teijin Ltd | 研磨基布および研磨方法 |
| JP2003201676A (ja) | 2001-12-28 | 2003-07-18 | Dainichiseika Color & Chem Mfg Co Ltd | 繊維シート及びその製造方法 |
| JP2004130395A (ja) | 2002-10-08 | 2004-04-30 | Toray Ind Inc | ガラステクスチャー加工用研磨布及びそれを用いた磁気記録媒体の製造方法 |
| JP2004311731A (ja) | 2003-04-08 | 2004-11-04 | Hitachi Chem Co Ltd | 研磨用パッド及びそれを用いた被研磨物の研磨方法 |
| JP2005518286A (ja) | 2001-10-29 | 2005-06-23 | トーマス・ウエスト,インコーポレイテッド | Cmp及び基板研磨用パッド |
| JP2005212055A (ja) | 2004-01-30 | 2005-08-11 | Kanebo Ltd | 不織布ベースの研磨布及びその製造方法 |
| JP2005334997A (ja) | 2004-05-25 | 2005-12-08 | Asahi Kasei Fibers Corp | 研磨パッド用不織布および研磨パッド |
| JP2006036909A (ja) * | 2004-07-27 | 2006-02-09 | Nicca Chemical Co Ltd | 研磨用シートの製造方法及び研磨用シート |
| JP2007054910A (ja) | 2005-08-24 | 2007-03-08 | Toray Ind Inc | 研磨布 |
| JP2008207323A (ja) | 2007-02-01 | 2008-09-11 | Kuraray Co Ltd | 研磨パッド及び研磨パッドの製造方法 |
| JP2010058170A (ja) * | 2008-08-08 | 2010-03-18 | Kuraray Co Ltd | 研磨パッド |
| JP2011200984A (ja) | 2010-03-26 | 2011-10-13 | Toray Ind Inc | 研磨パッド |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4356056B2 (ja) * | 2002-05-15 | 2009-11-04 | 東レ株式会社 | 樹脂含浸体および研磨パッドおよびその研磨パッドを用いた研磨装置と研磨方法 |
| US7829486B2 (en) * | 2003-02-06 | 2010-11-09 | Kuraray Co., Ltd. | Stretchable leather-like sheet substrate and process for producing same |
| WO2008093850A1 (ja) * | 2007-02-01 | 2008-08-07 | Kuraray Co., Ltd. | 研磨パッド及び研磨パッドの製造方法 |
| KR100943244B1 (ko) * | 2007-12-28 | 2010-02-18 | 대원화성 주식회사 | 표면 연마용 폴리우레탄 패드 및 그 제조방법 |
| JP2009241184A (ja) * | 2008-03-31 | 2009-10-22 | Kuraray Co Ltd | 研磨パッドおよびそれを用いた研磨方法 |
-
2014
- 2014-02-05 JP JP2015500137A patent/JP6220378B2/ja not_active Expired - Fee Related
- 2014-02-05 WO PCT/JP2014/000616 patent/WO2014125797A1/ja active Application Filing
- 2014-02-05 EP EP14752242.9A patent/EP2957672B1/en not_active Not-in-force
- 2014-02-05 US US14/767,115 patent/US20160002835A1/en not_active Abandoned
- 2014-02-05 CN CN201480007963.2A patent/CN105008614B/zh active Active
- 2014-02-05 KR KR1020157023576A patent/KR102136441B1/ko active IP Right Grant
- 2014-02-10 TW TW103104189A patent/TWI607832B/zh active
Patent Citations (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03234475A (ja) | 1990-02-08 | 1991-10-18 | Kanebo Ltd | 研磨布 |
| JPH10128674A (ja) | 1996-10-28 | 1998-05-19 | Rooder Nitta Kk | 研磨用パッド |
| JPH10225864A (ja) | 1997-02-17 | 1998-08-25 | Sony Corp | 研磨パッドとその製造方法並びにその研磨パッドを用いたウエハの研磨方法 |
| JPH1199479A (ja) | 1997-09-30 | 1999-04-13 | Teijin Ltd | 研磨パッド |
| JPH11322878A (ja) | 1998-05-13 | 1999-11-26 | Dainippon Ink & Chem Inc | 泡含有ポリウレタン成形物の製造方法、泡含有成形物用ウレタン樹脂組成物及びそれを用いた研磨パッド |
| JP2000178374A (ja) | 1998-12-15 | 2000-06-27 | Toyo Tire & Rubber Co Ltd | ポリウレタン発泡体の製造方法及び研磨シート |
| JP2000248034A (ja) | 1999-03-02 | 2000-09-12 | Mitsubishi Chemicals Corp | 研磨材用ポリウレタン系樹脂組成物及びその発泡体 |
| US6130163A (en) * | 1999-06-03 | 2000-10-10 | Promos Technologies, Inc. | Stabilization of slurry used in chemical mechanical polishing of semiconductor wafers by adjustment of PH of deionized water |
| JP2001089548A (ja) | 1999-09-22 | 2001-04-03 | Toyo Tire & Rubber Co Ltd | ポリウレタン発泡体の製造方法及び研磨シート |
| JP2002009026A (ja) | 2000-06-21 | 2002-01-11 | Toray Ind Inc | 研磨用パッドおよびそれを用いた研磨装置及び研磨方法 |
| JP2002172555A (ja) | 2000-12-05 | 2002-06-18 | Teijin Ltd | 研磨用基布および研磨方法 |
| JP2005518286A (ja) | 2001-10-29 | 2005-06-23 | トーマス・ウエスト,インコーポレイテッド | Cmp及び基板研磨用パッド |
| JP2003170347A (ja) | 2001-12-06 | 2003-06-17 | Teijin Ltd | 研磨基布および研磨方法 |
| JP2003201676A (ja) | 2001-12-28 | 2003-07-18 | Dainichiseika Color & Chem Mfg Co Ltd | 繊維シート及びその製造方法 |
| JP2004130395A (ja) | 2002-10-08 | 2004-04-30 | Toray Ind Inc | ガラステクスチャー加工用研磨布及びそれを用いた磁気記録媒体の製造方法 |
| JP2004311731A (ja) | 2003-04-08 | 2004-11-04 | Hitachi Chem Co Ltd | 研磨用パッド及びそれを用いた被研磨物の研磨方法 |
| JP2005212055A (ja) | 2004-01-30 | 2005-08-11 | Kanebo Ltd | 不織布ベースの研磨布及びその製造方法 |
| JP2005334997A (ja) | 2004-05-25 | 2005-12-08 | Asahi Kasei Fibers Corp | 研磨パッド用不織布および研磨パッド |
| JP2006036909A (ja) * | 2004-07-27 | 2006-02-09 | Nicca Chemical Co Ltd | 研磨用シートの製造方法及び研磨用シート |
| JP2007054910A (ja) | 2005-08-24 | 2007-03-08 | Toray Ind Inc | 研磨布 |
| JP2008207323A (ja) | 2007-02-01 | 2008-09-11 | Kuraray Co Ltd | 研磨パッド及び研磨パッドの製造方法 |
| JP2010058170A (ja) * | 2008-08-08 | 2010-03-18 | Kuraray Co Ltd | 研磨パッド |
| JP2011200984A (ja) | 2010-03-26 | 2011-10-13 | Toray Ind Inc | 研磨パッド |
Non-Patent Citations (1)
| Title |
|---|
| "CMP No Saiensu (The Science of CMP", 20 August 1997, SCIENCE FORUM INC., pages: 113 - 119 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019021897A1 (ja) * | 2017-07-25 | 2019-01-31 | ニッタ・ハース株式会社 | 研磨布 |
| JP2019025549A (ja) * | 2017-07-25 | 2019-02-21 | ニッタ・ハース株式会社 | 研磨布 |
| JP2020104197A (ja) * | 2018-12-27 | 2020-07-09 | 株式会社クラレ | 繊維複合研磨パッドおよびそれを用いたガラス系基材の研磨方法 |
| JP7111609B2 (ja) | 2018-12-27 | 2022-08-02 | 株式会社クラレ | 繊維複合研磨パッドおよびそれを用いたガラス系基材の研磨方法 |
| WO2021131590A1 (ja) * | 2019-12-26 | 2021-07-01 | 株式会社クラレ | 銀付皮革様シート |
| JP7550172B2 (ja) | 2019-12-26 | 2024-09-12 | 株式会社クラレ | 銀付皮革様シート |
Also Published As
| Publication number | Publication date |
|---|---|
| US20160002835A1 (en) | 2016-01-07 |
| CN105008614B (zh) | 2017-06-13 |
| KR20150116876A (ko) | 2015-10-16 |
| CN105008614A (zh) | 2015-10-28 |
| JPWO2014125797A1 (ja) | 2017-02-02 |
| TWI607832B (zh) | 2017-12-11 |
| EP2957672A1 (en) | 2015-12-23 |
| JP6220378B2 (ja) | 2017-10-25 |
| TW201440956A (zh) | 2014-11-01 |
| KR102136441B1 (ko) | 2020-07-21 |
| EP2957672A4 (en) | 2016-07-27 |
| EP2957672B1 (en) | 2018-05-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6220378B2 (ja) | 硬質シート及び硬質シートの製造方法 | |
| JP5411862B2 (ja) | 研磨パッド及び研磨パッドの製造方法 | |
| JP5204502B2 (ja) | 研磨パッド及び研磨パッドの製造方法 | |
| JP5289787B2 (ja) | 研磨パッド及び研磨パッドの製造方法 | |
| KR101146966B1 (ko) | 연마 패드 및 연마 패드의 제조 방법 | |
| JP5629266B2 (ja) | 研磨パッド及びケミカルメカニカル研磨方法 | |
| JP5143518B2 (ja) | 繊維複合研磨パッド | |
| JP6180873B2 (ja) | 繊維複合シート、研磨パッド及びその製造方法 | |
| JP7111609B2 (ja) | 繊維複合研磨パッドおよびそれを用いたガラス系基材の研磨方法 | |
| JP2010064153A (ja) | 研磨パッド | |
| JP5356149B2 (ja) | 研磨パッドの表面加工方法およびそれによって得られる研磨パッド | |
| JP5522929B2 (ja) | 研磨パッド及び研磨方法 | |
| JP5551022B2 (ja) | 被研磨物のラッピング方法 | |
| JP5789557B2 (ja) | ガラス系基材の研磨方法 | |
| JP5415700B2 (ja) | 研磨パッド及び研磨パッドの製造方法 | |
| JP5502661B2 (ja) | 化合物半導体ウエハの研磨方法 | |
| JP2010058170A (ja) | 研磨パッド |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14752242 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2015500137 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14767115 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014752242 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20157023576 Country of ref document: KR Kind code of ref document: A |