WO2014030477A1 - 芳香族炭化水素処理用活性白土 - Google Patents
芳香族炭化水素処理用活性白土 Download PDFInfo
- Publication number
- WO2014030477A1 WO2014030477A1 PCT/JP2013/069791 JP2013069791W WO2014030477A1 WO 2014030477 A1 WO2014030477 A1 WO 2014030477A1 JP 2013069791 W JP2013069791 W JP 2013069791W WO 2014030477 A1 WO2014030477 A1 WO 2014030477A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- clay
- acid
- activated clay
- range
- treatment
- Prior art date
Links
- 150000004945 aromatic hydrocarbons Chemical class 0.000 title claims abstract description 25
- 239000004927 clay Substances 0.000 claims abstract description 92
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 claims abstract description 56
- 229910021529 ammonia Inorganic materials 0.000 claims abstract description 28
- GUJOJGAPFQRJSV-UHFFFAOYSA-N dialuminum;dioxosilane;oxygen(2-);hydrate Chemical compound O.[O-2].[O-2].[O-2].[Al+3].[Al+3].O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O GUJOJGAPFQRJSV-UHFFFAOYSA-N 0.000 claims abstract description 17
- 229910052901 montmorillonite Inorganic materials 0.000 claims abstract description 17
- 238000000034 method Methods 0.000 claims description 14
- 229910018072 Al 2 O 3 Inorganic materials 0.000 claims description 13
- 238000003795 desorption Methods 0.000 claims description 13
- 229910004298 SiO 2 Inorganic materials 0.000 claims description 10
- 238000001179 sorption measurement Methods 0.000 claims description 10
- 239000002253 acid Substances 0.000 abstract description 25
- 230000003197 catalytic effect Effects 0.000 abstract description 8
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 abstract description 4
- 239000002994 raw material Substances 0.000 abstract description 3
- 229910021647 smectite Inorganic materials 0.000 abstract description 3
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 abstract description 2
- 238000010521 absorption reaction Methods 0.000 abstract 1
- 229910052681 coesite Inorganic materials 0.000 abstract 1
- 229910052593 corundum Inorganic materials 0.000 abstract 1
- 229910052906 cristobalite Inorganic materials 0.000 abstract 1
- 230000005923 long-lasting effect Effects 0.000 abstract 1
- 239000000377 silicon dioxide Substances 0.000 abstract 1
- 229910052682 stishovite Inorganic materials 0.000 abstract 1
- 229910052905 tridymite Inorganic materials 0.000 abstract 1
- 229910001845 yogo sapphire Inorganic materials 0.000 abstract 1
- 238000010306 acid treatment Methods 0.000 description 32
- 239000003054 catalyst Substances 0.000 description 21
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 18
- 239000011973 solid acid Substances 0.000 description 17
- 238000005259 measurement Methods 0.000 description 15
- 238000000746 purification Methods 0.000 description 15
- 239000000523 sample Substances 0.000 description 14
- 238000004519 manufacturing process Methods 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 239000002245 particle Substances 0.000 description 10
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 9
- 150000001336 alkenes Chemical class 0.000 description 8
- 230000000052 comparative effect Effects 0.000 description 8
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 238000005804 alkylation reaction Methods 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 6
- -1 BTX Chemical class 0.000 description 5
- 230000029936 alkylation Effects 0.000 description 5
- 150000002430 hydrocarbons Chemical class 0.000 description 5
- 238000007873 sieving Methods 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 238000002441 X-ray diffraction Methods 0.000 description 4
- 239000000440 bentonite Substances 0.000 description 4
- 229910000278 bentonite Inorganic materials 0.000 description 4
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- 238000006116 polymerization reaction Methods 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 238000009835 boiling Methods 0.000 description 3
- 150000001993 dienes Chemical class 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 230000001737 promoting effect Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- 229910004283 SiO 4 Inorganic materials 0.000 description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 2
- 229910052794 bromium Inorganic materials 0.000 description 2
- 150000001768 cations Chemical class 0.000 description 2
- 238000004042 decolorization Methods 0.000 description 2
- 238000007872 degassing Methods 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 235000010446 mineral oil Nutrition 0.000 description 2
- 239000010453 quartz Substances 0.000 description 2
- 239000004576 sand Substances 0.000 description 2
- 238000007086 side reaction Methods 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000011260 aqueous acid Substances 0.000 description 1
- 238000009933 burial Methods 0.000 description 1
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 description 1
- VNSBYDPZHCQWNB-UHFFFAOYSA-N calcium;aluminum;dioxido(oxo)silane;sodium;hydrate Chemical compound O.[Na].[Al].[Ca+2].[O-][Si]([O-])=O VNSBYDPZHCQWNB-UHFFFAOYSA-N 0.000 description 1
- 238000011088 calibration curve Methods 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 239000002734 clay mineral Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000005443 coulometric titration Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 238000007323 disproportionation reaction Methods 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000010433 feldspar Substances 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 238000011010 flushing procedure Methods 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 229910003439 heavy metal oxide Inorganic materials 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000010813 internal standard method Methods 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 238000002386 leaching Methods 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000004570 mortar (masonry) Substances 0.000 description 1
- 229910000273 nontronite Inorganic materials 0.000 description 1
- 239000011022 opal Substances 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000000607 poisoning effect Effects 0.000 description 1
- 239000002685 polymerization catalyst Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 238000010298 pulverizing process Methods 0.000 description 1
- 238000004445 quantitative analysis Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000013535 sea water Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 235000011121 sodium hydroxide Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 239000013638 trimer Substances 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J21/00—Catalysts comprising the elements, oxides, or hydroxides of magnesium, boron, aluminium, carbon, silicon, titanium, zirconium, or hafnium
- B01J21/16—Clays or other mineral silicates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/58—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with silicon, germanium, tin, lead, antimony, bismuth or compounds thereof
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/30—Catalysts, in general, characterised by their form or physical properties characterised by their physical properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/06—Washing
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G45/00—Refining of hydrocarbon oils using hydrogen or hydrogen-generating compounds
- C10G45/58—Refining of hydrocarbon oils using hydrogen or hydrogen-generating compounds to change the structural skeleton of some of the hydrocarbon content without cracking the other hydrocarbons present, e.g. lowering pour point; Selective hydrocracking of normal paraffins
- C10G45/68—Aromatisation of hydrocarbon oil fractions
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/02—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material
- B01J20/10—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising inorganic material comprising silica or silicate
- B01J20/12—Naturally occurring clays or bleaching earth
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/60—Catalysts, in general, characterised by their form or physical properties characterised by their surface properties or porosity
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G45/00—Refining of hydrocarbon oils using hydrogen or hydrogen-generating compounds
- C10G45/02—Refining of hydrocarbon oils using hydrogen or hydrogen-generating compounds to eliminate hetero atoms without changing the skeleton of the hydrocarbon involved and without cracking into lower boiling hydrocarbons; Hydrofinishing
- C10G45/04—Refining of hydrocarbon oils using hydrogen or hydrogen-generating compounds to eliminate hetero atoms without changing the skeleton of the hydrocarbon involved and without cracking into lower boiling hydrocarbons; Hydrofinishing characterised by the catalyst used
Definitions
- the present invention relates to an activated clay for treating aromatic hydrocarbons, and more specifically, activated clay used for purification of aromatic hydrocarbons such as BTX (benzene, toluene, xylene) and having a particularly excellent life. About.
- aromatic hydrocarbons such as BTX (benzene, toluene, xylene)
- purification treatment For the purpose of removing unsaturated hydrocarbon compounds such as olefins and diolefins from aromatic hydrocarbons such as BTX, purification treatment has been conventionally performed.
- an unsaturated compound contained in an aromatic hydrocarbon such as BTX is converted into a dimer or trimer by polymerization, or converted to a polycyclic aromatic compound by alkylation to an aromatic hydrocarbon.
- the molecular weight is increased and removed as a high boiling fraction.
- activated clay has been used for the above purification treatment from the viewpoint of acting as an unsaturated hydrocarbon polymerization catalyst (for example, Patent Documents 1 and 2).
- Activated clay is used for decolorization of mineral oil, and it is known that such activated clay for decolorization can also be used for the purification treatment of aromatic hydrocarbons (Patent Documents 3 and 4). ).
- JP-A-6-263431 JP 11-179202 A JP 2000-344513 A JP 2010-95436 A
- the activated clay used in Patent Document 1 has an extremely poor catalyst life and deteriorates the catalyst performance in a short period of time, so that it cannot be used satisfactorily as a purification process for aromatic hydrocarbons. .
- the activated clay shown in Patent Document 2 has improved catalyst life to some extent, it contains a high concentration of Fe 2 O 3 component, so that the place where the raw clay is produced contains a lot of iron (for example, India).
- activated clay is obtained by acid-treating clay (acid clay) containing montmorillonite as a main component and has a large specific surface area, but the Fe 2 O 3 component is eluted by this acid treatment. Therefore, in order to obtain an activated clay containing a certain amount or more of Fe 2 O 3 component, naturally, as the raw clay (acid clay) for acid treatment, one having a high content of Fe 2 O 3 component must be selected. Therefore, the production area is limited to India.
- the activated clay of Patent Document 3 or 4 is extremely excellent in the decoloring performance of mineral oil, but its catalyst life is not sufficient for use in the purification treatment of aromatic hydrocarbons. Further, this activated clay has a solid acid amount with an acid strength of Ho ⁇ ⁇ 3.0 within a certain range.
- the regulation of the amount of solid acid in such a wide range of acid strength distribution is not able to properly grasp the specific acid strength distribution that contributes to the catalytic function and the amount of solid acid in that range during the purification process of aromatic hydrocarbons. For this reason, there is a drawback that the performance as a catalyst is likely to vary.
- an object of the present invention is to provide an activated clay for treating aromatic hydrocarbons which is composed of an acid-treated product of smectite clay, the production area of the raw clay is not limited, and has a long catalyst life and no variation.
- the present inventors have determined that the degree of acid treatment is in accordance with the properties of the raw clay.
- the amount of solid acid in acid strength is selectively increased, the catalytic ability is not lowered, and the catalyst life is greatly improved, and the present invention has been completed.
- the montmorillonite content is 34% by mass or more
- the SiO 2 / Al 2 O 3 molar ratio is in the range of 3.8 to 8.0
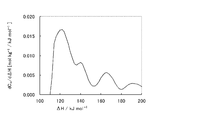
- An activated clay for treating aromatic hydrocarbons is provided, wherein the amount of ammonia desorption with an adsorption heat in the range of mol is in the range of 0.11 to 0.20 mmol / g.
- the activated clay of the present invention does not need to contain a high concentration of components such as heavy metal oxides removed by acid treatment, and the montmorillonite content may be 34% by mass or more. Therefore, the production area and face (burial place) of the raw clay are not limited, and the activated clay of the present invention can be obtained by an appropriate acid treatment even if it is montmorillonite produced from any production area.
- the activated clay of the present invention is not obtained by acid treatment using a high concentration of acid. It is obtained by using a rather weak acid treatment. This is because when the treatment using high-concentration acid wp is performed, the elution amount of Al 2 O 3 is large, and this molar ratio becomes a considerably large value.
- the activated clays of Patent Documents 1 to 4 described above have a large SiO 2 / Al 2 O 3 molar ratio as compared with the present invention.
- the activated clay of the present invention has a desorption amount of ammonia (with an adsorption heat in the range of 128 to 148 kJ / mol) measured by the ammonia TPD method within a certain range (0.11 to 0.20 mmol / g). is there.
- the heat of adsorption is related to the strength of the solid acid
- the amount of ammonia desorbed is related to the amount of solid acid. That is, in the ammonia TPD method, as described in the examples described later, the amount and temperature of ammonia desorbed by adsorbing ammonia, which is a base probe molecule, to a solid sample and continuously increasing the temperature. Are measured simultaneously.
- Ammonia adsorbed on weak acid sites desorbs at low temperatures (equivalent to desorption in a low adsorption heat range), and ammonia adsorbed on strong acid sites desorbs at high temperatures (in a high adsorption heat range). Is equivalent to detachment).
- the acid strength is indicated by temperature and heat of adsorption, and no color reaction is used. Therefore, compared with the conventional n-butylamine titration method, the solid acid strength and the solid acid amount are higher. Since it is an accurate value, the activated clay of the present invention can appropriately evaluate the characteristics.
- the ammonia desorption amount (related to the solid acid amount) specified by the ammonia TPD method is in the above range, so that the catalytic ability in the purification treatment of aromatic hydrocarbons is high. It exhibits a very long catalyst life, which is the greatest advantage of the present invention.
- aromatic hydrocarbons such as BTX
- reactions such as polymerization and alkylation of unsaturated hydrocarbon compounds such as olefins and diolefins contained therein are caused by the solid acid of the activated clay, and the acid strength is increased. It has been believed that the higher the amount of high solid acid, the more these reactions are promoted.
- solid acids that contribute to such reactions are limited to those in a specific acid strength range, and solid acids in other acid strength ranges promote side reactions and reduce catalyst life. End up.
- the amount of solid acid in the strength range that contributes to the alkylation reaction is selectively increased by an appropriate acid treatment, so that the catalytic ability (accelerating alkylation of olefins) is not impaired. An extremely long catalyst life can be realized.
- Montmorillonite which is the main component of clay used in the production of the activated clay of the present invention, is a clay mineral belonging to dioctahedral smectite, an AlO 6 octahedral layer sandwiched between two SiO 4 tetrahedral layers, and AlO 6 octa
- a three-layer structure in which a part of Al in the plane layer is isomorphously substituted with Fe or Mg and a part of Si in the SiO 4 tetrahedral layer is isomorphously substituted with Al is defined as a basic layer unit, and this basic layer unit is in the c-axis direction.
- Cations are present between the basic layers between the basic layers to compensate for the lack of charge due to isomorphous substitution. That is, when a clay mainly composed of montmorillonite having such a laminated structure is acid-treated, cations existing between the layers of the laminated structure are eluted, and the laminated structure of the basic three-layer structure is partially cut and broken apart. At the same time, the AlO 6 octahedral layer part elutes from the edge of the basic trilayer structure. A structure exhibiting characteristics as a solid acid is also generated at this end. In addition, with the acid treatment, the specific surface area also increases, and the SiO 2 / Al 2 O 3 molar ratio also increases due to the elution of the Al component.
- montmorillonite is thought to have been generated by the transformation of volcanic ash, lava, etc. under the influence of seawater.
- the chemical composition of the typical raw clay used for the production of the activated clay of the present invention is roughly as follows, and no special chemical composition is required except that it contains montmorillonite.
- the face is not limited. SiO 2 : 45 to 65% by mass Al 2 O 3 : 13 to 25% by weight MgO: 2-7% by weight CaO: 0.1 to 3.0% by weight Fe 2 O 3 : 2 to 25% by weight K 2 O: 0.1 to 3.0% by weight Na 2 O: 0.1 to 3.0% by weight Burning loss: 5-12% by weight
- the above-mentioned raw clay can be refined such as stone sand separation, buoyancy beneficiation, magnetic beneficiation, water tank, wind dredging, etc. to remove impurities such as quartz and feldspar present in the clay, and to make the particle size range uniform. is important. This is because it is easy to obtain physical properties suitable for the activated clay for aromatic hydrocarbon treatment of the present invention by performing the acid treatment described later uniformly.
- the acid treatment of the raw clay has an ammonia desorption amount of 0.11 to 0.20 mmol / g, particularly 0, as measured by the ammonia TPD method with an adsorption heat in the range of 128 to 148 kJ / mol. .11 to 0.17 mmol / g. That is, even if the acid treatment is excessively performed or insufficient, the ammonia desorption amount is reduced, and the catalyst life of the obtained activated clay is greatly reduced. Therefore, the degree of this acid treatment is extremely important.
- the acid treatment operation is performed by a known means, for example, by filling a treatment tank with a clay suspension, adding an aqueous acid solution, and stirring.
- the degree of acid treatment for obtaining the above ammonia desorption amount is compared with the acid treatment for obtaining activated clay for conventionally known aromatic hydrocarbon treatment. Then it is done at a mild level.
- mineral acids sulfuric acid, hydrochloric acid, etc.
- sulfuric acid are used as the acid, but the amount of treatment is conventionally about 75 parts by weight with respect to 100 parts by weight of dry clay. In the invention, the amount is as small as about 50 to 68 parts by mass.
- the processing temperature may be selected from the range of 60 to 100 ° C.
- the processing time may be selected from the range of 4 to 48 hours so that the above-described ammonia desorption amount can be obtained.
- the acid-treated product thus obtained has a montmorillonite content of at least 34% by mass.
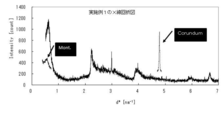
- the montmorillonite content can be calculated from the peak intensity of the (001) plane of montmorillonite by X-ray diffraction, as shown in the examples described later.
- the acid-treated product obtained as described above is washed with water and then subjected to drying, pulverization and classification, and adjusted to a particle size according to the use form.
- the obtained activated clay granule is appropriately heat-treated, whereby the particle strength can be increased.
- the heat treatment temperature is generally about 80 to 500 ° C., particularly about 100 to 300 ° C. and about 0.5 to 10 hours, particularly about 0.7 to 5 hours.
- the activated clay thus obtained is used as a catalyst for promoting the polymerization and alkylation of unsaturated hydrocarbon compounds in the purification process of aromatic hydrocarbons.
- the particle size is generally adjusted to 20 to 40 ⁇ m, particularly 25 to 35 ⁇ m median diameter powder, and when used on a fixed bed, the particle size is generally , And adjusted to a granular form in the range of 0.25 to 1.0 mm.
- the particle shape may be any shape such as a spherical shape, a granular shape, a cubic shape, a tablet shape, a cylindrical shape, and an indefinite shape.
- Such activated clay contains 34% by mass or more of montmorillonite, and as already described, the ammonia desorption amount with an adsorption heat in the range of 128 to 148 kJ / mol measured by the ammonia TPD method is 0.11. To 0.20 mmol / g, especially in the range of 0.11 to 0.17 mmol / g. Further, in the composition, the SiO 2 / Al 2 O 3 molar ratio is in the range of 3.8 to 8.0, particularly 3.8 to 7.0.
- the SiO 2 / Al 2 O 3 molar ratio is higher or lower than the above range, the ammonia desorption amount is less than the above range, and as a result, a sufficient solid acid amount cannot be obtained, and the catalytic ability ( The alkylation promoting function of unsaturated hydrocarbon compounds) becomes unsatisfactory.
- Such an activated clay of the present invention has an excellent function as a purification catalyst for unsaturated hydrocarbon compounds such as olefins and diolefins, as shown in the examples described later, and has a remarkable catalyst life. It is long and used effectively for the purification process of aromatic hydrocarbons. It should be noted that the activity and catalyst life of the activated clay for the treatment of aromatic hydrocarbons are the index of the olefin content for those obtained by removing the high boiling fraction by distillation after passing through the granular clay packed bed. It can evaluate by calculating
- the sample is filled in the holder by the NBS method [“Standard X-ray diffraction powder patterns”, NBS Monograph, 25 (1971)], and quantitative measurement is performed.
- the measurement conditions at that time are as follows: 2 ⁇ is 3 to 7, 20 to 27.5, and 42 to 44.5 [deg], the voltage is 40 [V] and the current is 40 [mA], and D Slit & S Slit: 2 / 3, V Slit: 10 [mm], R Slit 0.3 [mm], Step: 0.02 [deg].
- montmorillonite ethylene glycol-treated Kunipia F
- matrix flushing method which is a kind of internal standard method
- the peak area of the X-ray diffractogram was 220%
- the relative area intensity ratio (%) for each sample was The montmorillonite content was calculated.
- Ammonia desorption amount (ammonia TPD method) About 0.1 g of a sample is set in a quartz cell (inner diameter: 10 mm) of a TPD-AT-1 type thermal desorption apparatus manufactured by Bell Japan, and 10 Kmin ⁇ 1 up to 383 K under the flow of O 2 (60 cm 3 min ⁇ 1 , 1 atm). The temperature was raised at a temperature of 1 hour and the temperature reached 1 hour. Thereafter, the mixture was allowed to cool to 373 K while O 2 was circulated, then vacuum degassed, 100 Torr NH 3 was introduced and adsorbed for 30 min, and then degassed for 30 min, followed by steam treatment.
- the sample was sized with a sieve of 24 to 60 mesh, dried at 150 ° C. for 3 hours, and then used for the test.
- the BI of the sample tube outlet oil collected every 12 hours was measured, and the resulting break through curve was calculated as A.I. Wheeler & A. J. et al. Robert, J.M. Catal. , 13, 299 (1969).
- Ts was obtained by analysis using the following formula described in (1).
- BI 0 Entrance BI [mg / 100 g]
- BI Exit BI after time t [mg / 100 g]
- k 0 Initial catalyst primary reaction rate constant [1 / hr] and a value of Ka or less k
- A Olefin adsorption rate constant [1 / hr]
- W catalyst mass [g]
- F Oil flow rate [g / hr]
- Ws High boiling point olefin adsorbed per catalyst weight after ts time Weight [mg / 100 g]
- WHSV space velocity [1 / hr]
- Example 1 Water is added to the raw clay of the production area A to a concentration of 2 wt%, and after stirring and dispersing, it is passed through a sieve having an opening of 45 ⁇ m to remove sand having a size of 45 ⁇ m or more (hereinafter, the treatment so far is referred to as sieving treatment. Is the same even if the raw clay changes.) Thereafter, 2-45 ⁇ m silt particles were collected with a water tank and then centrifuged to purify the raw clay. The purified raw clay was added to water to obtain 351 g of a suspension having a water content of 88.6%. To this suspension was added 75% strength sulfuric acid.
- H 2 SO 4 / Clay when this ratio is shown
- H 2 SO 4 / Clay when this ratio is shown
- a concentration of 75% by mass sulfuric acid was added so as to be a mass% (hereinafter, when this ratio is shown, it is expressed as H 2 SO 4 / Liquid).
- the mixture was stirred with heating at 90 ° C. for 24 hours.
- the hydrolysis prevention treatment with the same concentration of 0.5% by mass sulfuric acid as the slurry, washed with water, dried and coarsely crushed to obtain activated clay.
- the acid treatment conditions are shown in Table 2, and various measurement results for the obtained activated clay are shown in Table 3.
- An X-ray diffraction diagram is shown in FIG.
- Example 2 The raw clay of the production area B was used. After sieving, the raw material clay was purified by centrifugation as it was. Except that the subsequent acid treatment conditions are listed in Table 2, acid treatment was performed in the same manner as in Example 1 to obtain activated clay. Table 3 shows various measurement results for the obtained activated clay.
- Example 3 The raw clay of the production area B was used after leaching opal contaminated in advance with caustic soda. After sieving, 2-45 ⁇ m silt particles were collected with a water tank, centrifuged, and then freeze-dried to purify the raw clay. Except that the subsequent acid treatment conditions are listed in Table 2, acid treatment was performed in the same manner as in Example 1 to obtain activated clay. Table 3 shows various measurement results for the obtained activated clay.
- Example 2 In Comparative Example 3, after sieving, particles having a size of 2 ⁇ m or more were removed with a water tank, and then centrifuged to purify the raw clay. Except that the subsequent acid treatment conditions are listed in Table 2, acid treatment was performed in the same manner as in Example 1 to obtain activated clay. Table 3 shows various measurement results for the obtained activated clay.
- Example 4 The raw clay of the production area C was used. After sieving, the recovered liquid was removed by removing particles of 2 ⁇ m or more with a water tank, and 0.3 to 2 ⁇ m clay was obtained. Except that the subsequent acid treatment conditions are listed in Table 2, acid treatment was performed in the same manner as in Example 1 to obtain activated clay. Table 3 shows various measurement results for the obtained activated clay.
- Example 3 In Comparative Example 4, an activated clay was obtained in the same manner except that the acid treatment conditions were listed in Table 2. Table 3 shows various measurement results for the obtained activated clay.
- Example 5 Except having used the raw material clay of the production center D, it carried out like Example 3 and obtained activated clay. Table 3 shows various measurement results for the obtained activated clay.
- Example 6 The raw clay of the production area E was used after being activated bentonite with NaHCO 3 in advance. Other than that was carried out similarly to Example 3, and activated clay was obtained. Table 3 shows various measurement results for the obtained activated clay. The acid strength distribution is shown in FIG.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Materials Engineering (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Crystallography & Structural Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Dispersion Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Production Of Liquid Hydrocarbon Mixture For Refining Petroleum (AREA)
- Silicates, Zeolites, And Molecular Sieves (AREA)
- Catalysts (AREA)
Abstract
Description
このようなアンモニアTPD法では、酸強度が温度や吸着熱量により示され、呈色反応を利用していないため、従来のn-ブチルアミン滴定法に比して、固体酸強度および固体酸量がより正確な値となっており、このため、本発明の活性白土は、特性の評価を適切に行うことができる。
従来、BTX等の芳香族炭化水素の精製処理では、これに含まれるオレフィンやジオレフィンなどの不飽和炭化水素化合物の重合やアルキル化といった反応が、活性白土の固体酸により引き起こされ、酸強度の高い固体酸の量が多くなるほど、これらの反応が促進されるものと考えられてきた。しかしながら、このような反応に寄与する固体酸は、特定の酸強度の範囲にあるものに限られ、これ以外の酸強度の範囲にある固体酸は、副反応を促進し、触媒寿命を低下させてしまう。しかるに、本発明では、適度な酸処理により、このアルキル化反応に寄与する強度範囲の固体酸量を選択的に多くしているため、触媒能(オレフィン類のアルキル化促進)を損なわずに、極めて長い触媒寿命を実現することが可能となるのである。
本発明の活性白土の製造に使用する粘土の主成分であるモンモリロナイトは、ジオクタヘドラル型スメクタイトに属する粘土鉱物であり、AlO6八面体層が2つのSiO4四面体層でサンドイッチされ、且つAlO6八面体層のAlの一部がFeやMgで同形置換され且つSiO4四面体層のSiの一部がAlで同形置換された三層構造を基本層単位とし、この基本層単位がc軸方向に積層された積層構造から成り、この基本層層間には同形置換による電荷の不足を補う形でカチオンが存在している。
即ち、このような積層構造を有するモンモリロナイトを主成分とする粘土を酸処理すると、積層構造の層間に存在するカチオンが溶出して、基本三層構造の積層構造が部分的に切断され、バラバラになると共に、AlO6八面体層の部分が基本三層構造の端から溶出する。この端にも固体酸としての特性を示す構造が生成することとなる。また、酸処理に伴って、比表面積も増大し、さらに、Al成分の溶出により、SiO2/Al2O3モル比の値も上昇していく。
SiO2:45~65質量%
Al2O3:13~25重量%
MgO:2~7重量%
CaO:0.1~3.0重量%
Fe2O3:2~25重量%
K2O:0.1~3.0重量%
Na2O:0.1~3.0重量%
灼熱減量:5~12重量%
原料粘土の酸処理は図1に示されるように、アンモニアTPD法で測定される128乃至148kJ/molの範囲の吸着熱でのアンモニア脱離量が0.11乃至0.20mmol/g、特に0.11乃至0.17mmol/gの範囲となる程度に行われる。即ち、酸処理を過度に行っても、或いは不十分であっても、このアンモニア脱離量が小さくなり、得られる活性白土の触媒寿命は大きく低下してしまう。従って、この酸処理の程度は極めて重要である。
また、このようにして得られた酸処理物のモンモリロナイト含量は少なくとも34質量%以上である。なお、このモンモリロナイト含量は、後述する実施例にも示されているように、X線回折により、モンモリロナイトの(001)面のピーク強度から算出することができる。
なお、得られた活性白土の粒状物は、適宜、熱処理され、これにより粒子強度を高めることができる。熱処理温度は、一般に、80乃至500℃、特に100乃至300℃の温度で0.5乃至10時間、特に0.7乃至5時間程度である。
かくして得られる活性白土は、芳香族炭化水素の精製処理に不飽和炭化水素化合物の重合およびアルキル化を促進する触媒として使用される。
例えば、芳香族炭化水素の精製処理をバッチで行う場合には、その粒径は一般に20乃至40μm、特に25乃至35μmのメジアン径の粉末に調整され、固定床で用いる場合は、一般に粒径が、0.25乃至1.0mmの範囲の粒状物の形態に調整される。粒子形状は、球状、顆粒状、立方体状、タブレット状、円柱状、不定形状等の何れの形状であってもよい。
また、その組成では、SiO2/Al2O3モル比が3.8乃至8.0、特に3.8乃至7.0の範囲となっている。即ち、SiO2/Al2O3モル比が上記範囲よりも高くても低くても、アンモニア脱離量が上記範囲よりも少なく、この結果、十分な固体酸量が得られず、触媒能(不飽和炭化水素化合物のアルキル化促進機能)が不満足となってしまう。
なお、芳香族炭化水素処理用活性白土の精製触媒としての活性や触媒寿命は、粒状白土充填層を通過させた後蒸留により高沸点留分を除去したものについて、オレフィン分の含有量の指標となる臭素指数を求めることにより評価することができる。
試料1gに10vol%エチレングリコール/エタノール溶液を加え、50℃で一晩乾燥させる。
乾燥した試料を乳鉢で粉砕してエチレングリコール処理した試料を得る。試料の測定対象回折ピーク2θ=5[deg]とα-Al2O3の測定対象回折ピーク2θ=43.3[deg]の面積強度が同程度になるように配合割合を加減して、試料に添加するα-Al2O3の量を決定する。
この混合物をアルミナボールと共に、振動ミルを用い50Hzで20分間振とうさせて均一化する。
測定角度範囲に応じて、NBS法[“Standard X-ray diffraction powder patterns”, NBS Monograph, 25(1971)]で試料をホルダーに充填し、定量測定を行う。
その際の測定条件は、2θが3~7、20~27.5および42~44.5[deg]について、電圧40[V]及び電流40[mA]で、D Slit & S Slit:2/3、V Slit:10[mm]、R Slit 0.3[mm]、Step:0.02[deg]であった。
標準物質として、montmorillonite(エチレングリコール処理したクニピアF)を用い、内部標準法の一種であるmatrixflushing法にしたがい、X線回折図のピーク面積を220%として各試料について相対面積強度比(%)を算出し、モンモリロナイト含有率とした。
粉末ろ紙法XRFで測定した結果から、SiとAlの各元素を酸化物としてSiO2/Al2O3モル比を求めた。
試料約0.1gを日本ベル製TPD-AT-1型昇温脱離装置の石英セル(内径10mm)にセットし、O2(60cm3 min-1、1atm)流通下、383Kまで10Kmin-1で昇温し、到達温度で1hr保った。その後O2を流通させたまま373Kまで放冷した後に真空脱気し、100TorrのNH3を導入して30min間吸着させ、その後30min間脱気した後に水蒸気処理を行った。
水蒸気処理としては、100℃で約25Torrの蒸気圧の水蒸気を導入、そのまま30min保ち、30min脱気、再び30min水蒸気導入、再び30min脱気の順に繰り返した。その後He0.041mmol s-1を減圧(100Torr、13.3kPa)に保ちながら流通させ、100℃で30min保った後に試料床を10Kmin-1で1073Kまで昇温し、出口気体を質量分析計(ANELVA M-QA 100F)で分析した。W/Fは13kgs m-3である。
測定に際しては質量数(m/e)16のマススペクトルを記録した。終了後に1mol%-NH3/He標準ガスをさらにヘリウムで希釈してNH3濃度0,0.1,0.2,0.3,0.4mol%,合計流量が0.041mmol s-1となるようにして検出器に流通させ,スペクトルを記録し、アンモニアの検量線を作成して検出器強度を補正した。
得られたTPDスペクトルから酸強度分布(Cw/ΔH)への変換は、鳥取大学大学院工学研究科/工学部研究報告,40,23(2009)に従って行った。
芳香族炭化水素成分の測定は、JIS K 2536-3に準拠し、島津製作所(株)製ガスクロマトグラフGC-2010を用いて測定した。また、臭素指数(Br-Index、以下、BIと略記)は、平沼産業(株)製電量滴定式BR-7で測定した。
供試油の成分を表1に示す。なお、供試油のBI(BI0)は646であった。
BI0:入り口BI[mg/100g]
BI:t時間後における出口BI[mg/100g]
k0:初期触媒一次反応速度定数[1/hr]でKa以下の値
kA:オレフィン吸着速度定数[1/hr]
W:触媒質量[g]
F:通油量[g/hr]
Ws:ts時間後における触媒重量あたり吸着した高沸点オレフィン
重量[mg/100g]
WHSV:空間速度[1/hr]
市販活性白土(BASF製F-124)を用いた。各種測定を行い、結果を表3に示す。
産地Aの原料粘土に水を加え濃度2wt%とし、撹拌分散後、目開き45μmの篩を通過させ、45μm以上の砂等を除去した(以後、ここまでの処理を篩分処理とする。これは、原料粘土が変わっても同様である。)。その後、水簸により2~45μmのシルト粒子を回収してから遠心分離して原料粘土を精製した。精製した原料粘土を水に加えて水分88.6%の懸濁液351gを得た。この懸濁液に濃度75%硫酸を加えた。このとき、乾燥粘土100質量部に対して硫酸(H2SO4)68質量部(以下、この割合を示す場合は、H2SO4/Clayと表す)、溶液全体に対する硫酸濃度を4.0質量%(以下、この割合を示す場合は、H2SO4/Liquidと表す)の割合となるように濃度75質量%硫酸を加えた。添加後、90℃で24時間加熱撹拌した。その後、スラリーと同量の濃度0.5質量%硫酸で加水分解防止処理後、水洗、乾燥・粗砕して活性白土を得た。酸処理条件は表2に示し、得られた活性白土についての各種測定結果は表3に示す。また、X線回折図を図2に示す。
産地Bの原料粘土を用いた。篩分処理後、そのまま遠心分離して原料粘土を精製した。その後の酸処理条件を表2に記載した以外は、実施例1と同様にして酸処理を行い、活性白土を得た。得られた活性白土についての各種測定結果を表3に示す。
産地Bの原料粘土を、あらかじめ夾雑するオパールを苛性ソーダで溶脱してから用いた。篩分処理後、水簸により2~45μmのシルト粒子を回収してから遠心分離後、凍結乾燥し原料粘土を精製した。その後の酸処理条件を表2に記載した以外は、実施例1と同様にして酸処理を行い、活性白土を得た。得られた活性白土についての各種測定結果を表3に示す。
比較例3において、篩分処理後、水簸により2μm以上の粒子を除いてから遠心分離し原料粘土を精製した。その後の酸処理条件を表2に記載した以外は、実施例1と同様にして酸処理を行い、活性白土を得た。得られた活性白土についての各種測定結果を表3に示す。
産地Cの原料粘土を用いた。篩分処理後、水簸により2μm以上の粒子を除いて回収した液を遠心分離し、0.3~2μmの粘土を得た。その後の酸処理条件を表2に記載した以外は、実施例1と同様にして酸処理を行い、活性白土を得た。得られた活性白土についての各種測定結果を表3に示す。
比較例4において、酸処理条件を表2に記載した以外は、同様にして行い活性白土を得た。得られた活性白土についての各種測定結果を表3に示す。
比較例4において、原料粘土を精製するところまでは同様にして行った。その後酸処理を表2に記載した条件で行った。但し、酸処理は初めにH2SO4/Liquid=4.0質量%になるように硫酸を仕込み、6時間後に残りの硫酸を追加して処理を行った(合計のH2SO4/Liquid=4.6質量%)。酸処理の温度は90℃で、処理時間は合計で24時間加熱撹拌した。その後の処理は、比較例4と同様にして行い、活性白土を得た。得られた活性白土についての各種測定結果を表3に示す。
産地Dの原料粘土を用いた以外は、実施例3と同様にして行い、活性白土を得た。得られた活性白土について各種測定結果を表3に示す。
産地Eの原料粘土を、あらかじめ、NaHCO3で活性ベントナイト化してから用いた。それ以外は、実施例3と同様にして行い、活性白土を得た。得られた活性白土についての各種測定結果を表3に示す。また、その酸強度分布を図1に示した。
Claims (1)
- モンモリロナイト含有率が34質量%以上、SiO2/Al2O3モル比が3.8乃至8.0の範囲にあり、アンモニアTPD法で測定される128乃至148kJ/molの範囲の吸着熱でのアンモニア脱離量が0.11乃至0.20mmol/gの範囲にあることを特徴とする芳香族炭化水素処理用活性白土。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MYPI2015700281A MY194724A (en) | 2012-08-23 | 2013-07-22 | Activated clay for treating aromatic hydrocarbons |
| IN1232DEN2015 IN2015DN01232A (ja) | 2012-08-23 | 2013-07-22 | |
| US14/416,179 US9605090B2 (en) | 2012-08-23 | 2013-07-22 | Activated clay for treating aromatic hydrocarbons |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-184021 | 2012-08-23 | ||
| JP2012184021A JP5837467B2 (ja) | 2012-08-23 | 2012-08-23 | 芳香族炭化水素処理用活性白土 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014030477A1 true WO2014030477A1 (ja) | 2014-02-27 |
Family
ID=50149789
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/069791 WO2014030477A1 (ja) | 2012-08-23 | 2013-07-22 | 芳香族炭化水素処理用活性白土 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US9605090B2 (ja) |
| JP (1) | JP5837467B2 (ja) |
| IN (1) | IN2015DN01232A (ja) |
| MY (1) | MY194724A (ja) |
| WO (1) | WO2014030477A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN117427481A (zh) * | 2023-08-24 | 2024-01-23 | 葫芦岛康达环保工贸有限公司 | 一种基于纳米二氧化硅改性的除臭剂及其制备方法 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6332860B2 (ja) * | 2014-10-08 | 2018-05-30 | 公益財団法人北九州産業学術推進機構 | 炭化水素油の精製方法 |
| JP6618769B2 (ja) * | 2015-01-09 | 2019-12-11 | 水澤化学工業株式会社 | 活性白土粒子 |
| JP6684112B2 (ja) * | 2016-03-02 | 2020-04-22 | 黒崎播磨株式会社 | 遮熱タイルの製造方法 |
| CN114917865B (zh) * | 2022-06-06 | 2023-08-22 | 中海油天津化工研究设计院有限公司 | 一种芳烃吸附剂及其在增产乙烯裂解原料中的应用 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6461310A (en) * | 1987-09-01 | 1989-03-08 | Shinkyowa Petrochem | Modified saponite |
| JPH11179202A (ja) * | 1997-12-25 | 1999-07-06 | Mizusawa Ind Chem Ltd | 芳香族炭化水素処理用活性白土 |
| JP2010095436A (ja) * | 2008-09-18 | 2010-04-30 | Mizusawa Ind Chem Ltd | 新規な活性白土及び動植物の油脂類もしくは鉱物油の脱色剤 |
| WO2010116603A1 (ja) * | 2009-03-30 | 2010-10-14 | 財団法人石油産業活性化センター | アルキルベンゼン類の製造方法及びそれに用いる触媒 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2466047A (en) * | 1946-01-30 | 1949-04-05 | Houdry Process Corp | Preparation of clay catalysts |
| US3787330A (en) * | 1968-10-01 | 1974-01-22 | Mizusawa Industrial Chem | Refining agent for oily substances |
| US5330946A (en) | 1993-01-29 | 1994-07-19 | American Colloid Company | Process of acid binding fine smectite clay particles into granules |
| JP4404991B2 (ja) | 1999-06-01 | 2010-01-27 | 水澤化学工業株式会社 | 活性白土定形粒子、その製造方法及びその用途 |
-
2012
- 2012-08-23 JP JP2012184021A patent/JP5837467B2/ja active Active
-
2013
- 2013-07-22 WO PCT/JP2013/069791 patent/WO2014030477A1/ja active Application Filing
- 2013-07-22 MY MYPI2015700281A patent/MY194724A/en unknown
- 2013-07-22 IN IN1232DEN2015 patent/IN2015DN01232A/en unknown
- 2013-07-22 US US14/416,179 patent/US9605090B2/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6461310A (en) * | 1987-09-01 | 1989-03-08 | Shinkyowa Petrochem | Modified saponite |
| JPH11179202A (ja) * | 1997-12-25 | 1999-07-06 | Mizusawa Ind Chem Ltd | 芳香族炭化水素処理用活性白土 |
| JP2010095436A (ja) * | 2008-09-18 | 2010-04-30 | Mizusawa Ind Chem Ltd | 新規な活性白土及び動植物の油脂類もしくは鉱物油の脱色剤 |
| WO2010116603A1 (ja) * | 2009-03-30 | 2010-10-14 | 財団法人石油産業活性化センター | アルキルベンゼン類の製造方法及びそれに用いる触媒 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN117427481A (zh) * | 2023-08-24 | 2024-01-23 | 葫芦岛康达环保工贸有限公司 | 一种基于纳米二氧化硅改性的除臭剂及其制备方法 |
| CN117427481B (zh) * | 2023-08-24 | 2024-04-09 | 葫芦岛康达环保工贸有限公司 | 一种基于纳米二氧化硅改性的除臭剂及其制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5837467B2 (ja) | 2015-12-24 |
| IN2015DN01232A (ja) | 2015-06-26 |
| MY194724A (en) | 2022-12-15 |
| JP2014040351A (ja) | 2014-03-06 |
| US9605090B2 (en) | 2017-03-28 |
| US20150203603A1 (en) | 2015-07-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2014030477A1 (ja) | 芳香族炭化水素処理用活性白土 | |
| Schutz et al. | Interlamellar chemistry of hydrotalcites: I. Polymerization of silicate anions | |
| US8609911B1 (en) | Catalytic pyrolysis using UZM-44 aluminosilicate zeolite | |
| US20050013773A1 (en) | Catalyst containing microporous zeolite in mesoporous support and method for making same | |
| Belviso et al. | Synthesis of composite zeolite-layered double hydroxides using ultrasonic neutralized red mud | |
| EP0165647A2 (en) | Process for preparation of synthetic crystalline zinc silicate mineral | |
| KR102408127B1 (ko) | 시티나카이트 구조를 가진 실리코티타네이트를 포함하는 조성물 및 그의 제조 방법 | |
| JP2013209284A (ja) | Zsm−5の製造方法 | |
| CN108910911A (zh) | 一种利用程序升温法合成全硅Beta分子筛母液的回收利用方法 | |
| WO2007101643A1 (en) | Mineral composition | |
| JP2011207687A (ja) | ベントナイト粒子 | |
| Mamudu et al. | The production of zeolite Y catalyst from palm kernel shell for fluid catalytic cracking unit | |
| EP0491520B1 (en) | Kandite clay compositions | |
| JP5646807B2 (ja) | 人工ゼオライトの製造方法 | |
| JP6045890B2 (ja) | 新規な結晶構造を有するmcm−22型ゼオライト及び該ゼオライトからなる芳香族炭化水素精製触媒 | |
| Takahashi et al. | Study of synthetic clay minerals. III. Synthesis and characterization of two dimensional talc | |
| Mbaye et al. | Mineralogical and physico-chemical characterizations of clay from Keur Saër (Senegal) | |
| WO2016086781A1 (zh) | 一种催化剂及其制备方法和应用其制备异丁烯的方法 | |
| US3001853A (en) | Synthetic magnesium aluminum silicate product | |
| JPH11156197A (ja) | 炭化水素油の分解触媒 | |
| CN115536036B (zh) | 改性高岭土及其制备方法和原位晶化合成分子筛的方法以及NaY分子筛 | |
| JP2636183B2 (ja) | 水熱安定性の向上した層間架橋粘土の合成法 | |
| JP2938103B2 (ja) | トリポリリン酸二水素アルミニウム型層間化合物 | |
| Acton | Aluminum Silicates—Advances in Research and Application: 2013 Edition: ScholarlyBrief | |
| JP6236728B2 (ja) | ゼオライトzsm−18、その合成およびその使用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13831446 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14416179 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IDP00201500993 Country of ref document: ID |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13831446 Country of ref document: EP Kind code of ref document: A1 |