WO2011115137A1 - 置換ビピリジル基を有する二核ルテニウム錯体色素を有する光電変換素子、及び光化学電池 - Google Patents
置換ビピリジル基を有する二核ルテニウム錯体色素を有する光電変換素子、及び光化学電池 Download PDFInfo
- Publication number
- WO2011115137A1 WO2011115137A1 PCT/JP2011/056110 JP2011056110W WO2011115137A1 WO 2011115137 A1 WO2011115137 A1 WO 2011115137A1 JP 2011056110 W JP2011056110 W JP 2011056110W WO 2011115137 A1 WO2011115137 A1 WO 2011115137A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- group
- ruthenium complex
- group represented
- complex dye
- Prior art date
Links
- 239000012327 Ruthenium complex Substances 0.000 title claims abstract description 135
- 238000006243 chemical reaction Methods 0.000 title claims abstract description 76
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical group N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 title description 13
- 125000004432 carbon atom Chemical group C* 0.000 claims description 47
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 39
- 125000000217 alkyl group Chemical group 0.000 claims description 38
- 239000004065 semiconductor Substances 0.000 claims description 33
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 28
- 239000010419 fine particle Substances 0.000 claims description 25
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 24
- 150000002500 ions Chemical class 0.000 claims description 23
- 125000001424 substituent group Chemical group 0.000 claims description 17
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 15
- 238000000034 method Methods 0.000 claims description 11
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 claims description 8
- 239000003792 electrolyte Substances 0.000 claims description 8
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 7
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 claims description 6
- 229910001887 tin oxide Inorganic materials 0.000 claims description 6
- 229910052799 carbon Inorganic materials 0.000 claims description 4
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 4
- 239000011787 zinc oxide Substances 0.000 claims description 4
- 125000003118 aryl group Chemical group 0.000 claims description 3
- 125000005549 heteroarylene group Chemical group 0.000 claims description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 3
- 239000002243 precursor Substances 0.000 claims description 2
- 230000005611 electricity Effects 0.000 abstract description 5
- 238000010521 absorption reaction Methods 0.000 abstract description 4
- 239000000975 dye Substances 0.000 description 96
- -1 methoxy, ethoxy, propoxy, butoxy, pentoxy, hexyloxy, heptyloxy, octyloxy, nonyloxy, decyloxy, undecyloxy, dodecyloxy Chemical group 0.000 description 95
- 239000000243 solution Substances 0.000 description 55
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 48
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 24
- 239000000203 mixture Substances 0.000 description 24
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 21
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 19
- 230000015572 biosynthetic process Effects 0.000 description 19
- 238000003786 synthesis reaction Methods 0.000 description 19
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 18
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 18
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 16
- 239000000706 filtrate Substances 0.000 description 15
- 238000000862 absorption spectrum Methods 0.000 description 14
- 239000007864 aqueous solution Substances 0.000 description 14
- PNGLEYLFMHGIQO-UHFFFAOYSA-M sodium;3-(n-ethyl-3-methoxyanilino)-2-hydroxypropane-1-sulfonate;dihydrate Chemical compound O.O.[Na+].[O-]S(=O)(=O)CC(O)CN(CC)C1=CC=CC(OC)=C1 PNGLEYLFMHGIQO-UHFFFAOYSA-M 0.000 description 12
- 239000012141 concentrate Substances 0.000 description 11
- 239000000460 chlorine Substances 0.000 description 10
- 239000003446 ligand Substances 0.000 description 10
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 9
- 150000001450 anions Chemical class 0.000 description 9
- 239000012300 argon atmosphere Substances 0.000 description 9
- 239000000434 metal complex dye Substances 0.000 description 9
- 239000002904 solvent Substances 0.000 description 9
- 238000003756 stirring Methods 0.000 description 9
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 8
- 229910052757 nitrogen Inorganic materials 0.000 description 8
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 8
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 7
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 7
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical group C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 6
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 6
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- 230000031700 light absorption Effects 0.000 description 6
- HSZCZNFXUDYRKD-UHFFFAOYSA-M lithium iodide Chemical compound [Li+].[I-] HSZCZNFXUDYRKD-UHFFFAOYSA-M 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 229910052751 metal Inorganic materials 0.000 description 6
- 239000002184 metal Substances 0.000 description 6
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- IUVCFHHAEHNCFT-INIZCTEOSA-N 2-[(1s)-1-[4-amino-3-(3-fluoro-4-propan-2-yloxyphenyl)pyrazolo[3,4-d]pyrimidin-1-yl]ethyl]-6-fluoro-3-(3-fluorophenyl)chromen-4-one Chemical compound C1=C(F)C(OC(C)C)=CC=C1C(C1=C(N)N=CN=C11)=NN1[C@@H](C)C1=C(C=2C=C(F)C=CC=2)C(=O)C2=CC(F)=CC=C2O1 IUVCFHHAEHNCFT-INIZCTEOSA-N 0.000 description 4
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical group C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 description 4
- 230000008033 biological extinction Effects 0.000 description 4
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 4
- 235000019253 formic acid Nutrition 0.000 description 4
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 4
- 125000003707 hexyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 4
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 4
- 229910052697 platinum Inorganic materials 0.000 description 4
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 150000003303 ruthenium Chemical class 0.000 description 4
- 239000010409 thin film Substances 0.000 description 4
- 125000002948 undecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- VYXHVRARDIDEHS-UHFFFAOYSA-N 1,5-cyclooctadiene Chemical compound C1CC=CCCC=C1 VYXHVRARDIDEHS-UHFFFAOYSA-N 0.000 description 3
- 239000004912 1,5-cyclooctadiene Substances 0.000 description 3
- IAFFEFPJCKQBKK-UHFFFAOYSA-N 2,3-dichloro-1-methyl-4-propan-2-ylbenzene;ruthenium Chemical class [Ru].CC(C)C1=CC=C(C)C(Cl)=C1Cl IAFFEFPJCKQBKK-UHFFFAOYSA-N 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 3
- YNPNZTXNASCQKK-UHFFFAOYSA-N Phenanthrene Natural products C1=CC=C2C3=CC=CC=C3C=CC2=C1 YNPNZTXNASCQKK-UHFFFAOYSA-N 0.000 description 3
- 238000002835 absorbance Methods 0.000 description 3
- 150000001298 alcohols Chemical class 0.000 description 3
- 125000003277 amino group Chemical group 0.000 description 3
- 235000013877 carbamide Nutrition 0.000 description 3
- 239000010408 film Substances 0.000 description 3
- 125000005843 halogen group Chemical group 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- GCRLVKBHFZOVLQ-UHFFFAOYSA-H hexachloroosmium Chemical compound Cl[Os](Cl)(Cl)(Cl)(Cl)Cl GCRLVKBHFZOVLQ-UHFFFAOYSA-H 0.000 description 3
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 3
- PJXISJQVUVHSOJ-UHFFFAOYSA-N indium(iii) oxide Chemical class [O-2].[O-2].[O-2].[In+3].[In+3] PJXISJQVUVHSOJ-UHFFFAOYSA-N 0.000 description 3
- 229910052740 iodine Inorganic materials 0.000 description 3
- 238000004811 liquid chromatography Methods 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 150000002825 nitriles Chemical class 0.000 description 3
- NLKNQRATVPKPDG-UHFFFAOYSA-M potassium iodide Chemical compound [K+].[I-] NLKNQRATVPKPDG-UHFFFAOYSA-M 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000011541 reaction mixture Substances 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 125000005504 styryl group Chemical group 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 0 Cc(cc1)ccc1O* Chemical compound Cc(cc1)ccc1O* 0.000 description 2
- ZYSSNSIOLIJYRF-UHFFFAOYSA-H Cl[Ir](Cl)(Cl)(Cl)(Cl)Cl Chemical compound Cl[Ir](Cl)(Cl)(Cl)(Cl)Cl ZYSSNSIOLIJYRF-UHFFFAOYSA-H 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical compound [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 229910002651 NO3 Inorganic materials 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- XZQYTGKSBZGQMO-UHFFFAOYSA-I Rhenium(V) chloride Inorganic materials Cl[Re](Cl)(Cl)(Cl)Cl XZQYTGKSBZGQMO-UHFFFAOYSA-I 0.000 description 2
- 229910021637 Rhenium(VI) chloride Inorganic materials 0.000 description 2
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 229910017052 cobalt Inorganic materials 0.000 description 2
- 239000010941 cobalt Substances 0.000 description 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 2
- 229910001429 cobalt ion Inorganic materials 0.000 description 2
- XLJKHNWPARRRJB-UHFFFAOYSA-N cobalt(2+) Chemical compound [Co+2] XLJKHNWPARRRJB-UHFFFAOYSA-N 0.000 description 2
- 239000006258 conductive agent Substances 0.000 description 2
- ZSWFCLXCOIISFI-UHFFFAOYSA-N cyclopentadiene Chemical compound C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 2
- KXGVEGMKQFWNSR-UHFFFAOYSA-N deoxycholic acid Natural products C1CC2CC(O)CCC2(C)C2C1C1CCC(C(CCC(O)=O)C)C1(C)C(O)C2 KXGVEGMKQFWNSR-UHFFFAOYSA-N 0.000 description 2
- 239000008151 electrolyte solution Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- GSGIQJBJGSKCDZ-UHFFFAOYSA-H hexachlororhenium Chemical compound Cl[Re](Cl)(Cl)(Cl)(Cl)Cl GSGIQJBJGSKCDZ-UHFFFAOYSA-H 0.000 description 2
- QTNLQPHXMVHGBA-UHFFFAOYSA-H hexachlororhodium Chemical compound Cl[Rh](Cl)(Cl)(Cl)(Cl)Cl QTNLQPHXMVHGBA-UHFFFAOYSA-H 0.000 description 2
- 229910003437 indium oxide Inorganic materials 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- SNEMTZZLJPELNU-UHFFFAOYSA-N lithium;methanol;methanolate Chemical compound [Li+].OC.[O-]C SNEMTZZLJPELNU-UHFFFAOYSA-N 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 150000003856 quaternary ammonium compounds Chemical class 0.000 description 2
- 229910052707 ruthenium Inorganic materials 0.000 description 2
- CHGSZSOJDJQCGO-UHFFFAOYSA-N ruthenium(2+);hexacyanide Chemical compound [Ru+2].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-] CHGSZSOJDJQCGO-UHFFFAOYSA-N 0.000 description 2
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000000967 suction filtration Methods 0.000 description 2
- UXMRNSHDSCDMLG-UHFFFAOYSA-J tetrachlororhenium Chemical compound Cl[Re](Cl)(Cl)Cl UXMRNSHDSCDMLG-UHFFFAOYSA-J 0.000 description 2
- 125000000335 thiazolyl group Chemical group 0.000 description 2
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N thiocyanic acid Chemical compound SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 description 2
- 125000003396 thiol group Chemical group [H]S* 0.000 description 2
- 150000003672 ureas Chemical class 0.000 description 2
- BHQCQFFYRZLCQQ-UHFFFAOYSA-N (3alpha,5alpha,7alpha,12alpha)-3,7,12-trihydroxy-cholan-24-oic acid Natural products OC1CC2CC(O)CCC2(C)C2C1C1CCC(C(CCC(O)=O)C)C1(C)C(O)C2 BHQCQFFYRZLCQQ-UHFFFAOYSA-N 0.000 description 1
- RUDATBOHQWOJDD-UHFFFAOYSA-N (3beta,5beta,7alpha)-3,7-Dihydroxycholan-24-oic acid Natural products OC1CC2CC(O)CCC2(C)C2C1C1CCC(C(CCC(O)=O)C)C1(C)CC2 RUDATBOHQWOJDD-UHFFFAOYSA-N 0.000 description 1
- LAXRNWSASWOFOT-UHFFFAOYSA-J (cymene)ruthenium dichloride dimer Chemical compound [Cl-].[Cl-].[Cl-].[Cl-].[Ru+2].[Ru+2].CC(C)C1=CC=C(C)C=C1.CC(C)C1=CC=C(C)C=C1 LAXRNWSASWOFOT-UHFFFAOYSA-J 0.000 description 1
- ISHFYECQSXFODS-UHFFFAOYSA-M 1,2-dimethyl-3-propylimidazol-1-ium;iodide Chemical compound [I-].CCCN1C=C[N+](C)=C1C ISHFYECQSXFODS-UHFFFAOYSA-M 0.000 description 1
- JBOIAZWJIACNJF-UHFFFAOYSA-N 1h-imidazole;hydroiodide Chemical compound [I-].[NH2+]1C=CN=C1 JBOIAZWJIACNJF-UHFFFAOYSA-N 0.000 description 1
- JFJNVIPVOCESGZ-UHFFFAOYSA-N 2,3-dipyridin-2-ylpyridine Chemical compound N1=CC=CC=C1C1=CC=CN=C1C1=CC=CC=N1 JFJNVIPVOCESGZ-UHFFFAOYSA-N 0.000 description 1
- VEZJRJGLFIXQHG-UHFFFAOYSA-N 2-(1h-benzimidazol-2-yl)-1h-benzimidazole Chemical compound C1=CC=C2NC(C=3NC4=CC=CC=C4N=3)=NC2=C1 VEZJRJGLFIXQHG-UHFFFAOYSA-N 0.000 description 1
- FXPLCAKVOYHAJA-UHFFFAOYSA-N 2-(4-carboxypyridin-2-yl)pyridine-4-carboxylic acid Chemical compound OC(=O)C1=CC=NC(C=2N=CC=C(C=2)C(O)=O)=C1 FXPLCAKVOYHAJA-UHFFFAOYSA-N 0.000 description 1
- OOWFYDWAMOKVSF-UHFFFAOYSA-N 3-methoxypropanenitrile Chemical compound COCCC#N OOWFYDWAMOKVSF-UHFFFAOYSA-N 0.000 description 1
- NPYGILQVQLESKX-PHEQNACWSA-N 4-[(e)-2-(4-hexoxyphenyl)ethenyl]-2-[4-[(e)-2-(4-hexoxyphenyl)ethenyl]pyridin-2-yl]pyridine Chemical compound C1=CC(OCCCCCC)=CC=C1\C=C\C1=CC=NC(C=2N=CC=C(\C=C\C=3C=CC(OCCCCCC)=CC=3)C=2)=C1 NPYGILQVQLESKX-PHEQNACWSA-N 0.000 description 1
- VHJFWJXYEWHCGD-UHFFFAOYSA-N 4-nonyl-2-(4-nonylpyridin-2-yl)pyridine Chemical compound CCCCCCCCCC1=CC=NC(C=2N=CC=C(CCCCCCCCC)C=2)=C1 VHJFWJXYEWHCGD-UHFFFAOYSA-N 0.000 description 1
- YSHMQTRICHYLGF-UHFFFAOYSA-N 4-tert-butylpyridine Chemical compound CC(C)(C)C1=CC=NC=C1 YSHMQTRICHYLGF-UHFFFAOYSA-N 0.000 description 1
- MARUHZGHZWCEQU-UHFFFAOYSA-N 5-phenyl-2h-tetrazole Chemical compound C1=CC=CC=C1C1=NNN=N1 MARUHZGHZWCEQU-UHFFFAOYSA-N 0.000 description 1
- JBRZTFJDHDCESZ-UHFFFAOYSA-N AsGa Chemical compound [As]#[Ga] JBRZTFJDHDCESZ-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- XMBQRLRDYNGSAV-UHFFFAOYSA-N CCCCCCC1=CC=C(C2=CC=C(C3(C=CN=CC3)C3=NC=CC(C4=CC=C(C5=CC=C(CCCCCC)S5)S4)=C3)S2)S1 Chemical compound CCCCCCC1=CC=C(C2=CC=C(C3(C=CN=CC3)C3=NC=CC(C4=CC=C(C5=CC=C(CCCCCC)S5)S4)=C3)S2)S1 XMBQRLRDYNGSAV-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 239000004380 Cholic acid Substances 0.000 description 1
- SNLCFABCUVHNSN-UHFFFAOYSA-J Cl[Co](Cl)(Cl)Cl Chemical compound Cl[Co](Cl)(Cl)Cl SNLCFABCUVHNSN-UHFFFAOYSA-J 0.000 description 1
- 229910021581 Cobalt(III) chloride Inorganic materials 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- VMQMZMRVKUZKQL-UHFFFAOYSA-N Cu+ Chemical compound [Cu+] VMQMZMRVKUZKQL-UHFFFAOYSA-N 0.000 description 1
- JPVYNHNXODAKFH-UHFFFAOYSA-N Cu2+ Chemical compound [Cu+2] JPVYNHNXODAKFH-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- VTLYFUHAOXGGBS-UHFFFAOYSA-N Fe3+ Chemical compound [Fe+3] VTLYFUHAOXGGBS-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 229910005540 GaP Inorganic materials 0.000 description 1
- 229910001218 Gallium arsenide Inorganic materials 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- WAEMQWOKJMHJLA-UHFFFAOYSA-N Manganese(2+) Chemical compound [Mn+2] WAEMQWOKJMHJLA-UHFFFAOYSA-N 0.000 description 1
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- XSKPNBNTNNSSKX-UHFFFAOYSA-N [AlH2]/C=C/c1ccnc(-c2cc(/C=C/[AlH2])ccn2)c1 Chemical compound [AlH2]/C=C/c1ccnc(-c2cc(/C=C/[AlH2])ccn2)c1 XSKPNBNTNNSSKX-UHFFFAOYSA-N 0.000 description 1
- XHCLAFWTIXFWPH-UHFFFAOYSA-N [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] XHCLAFWTIXFWPH-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000005277 alkyl imino group Chemical group 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 229910021417 amorphous silicon Inorganic materials 0.000 description 1
- 125000005577 anthracene group Chemical group 0.000 description 1
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 125000003828 azulenyl group Chemical group 0.000 description 1
- JRPBQTZRNDNNOP-UHFFFAOYSA-N barium titanate Chemical compound [Ba+2].[Ba+2].[O-][Ti]([O-])([O-])[O-] JRPBQTZRNDNNOP-UHFFFAOYSA-N 0.000 description 1
- 229910002113 barium titanate Inorganic materials 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000000480 butynyl group Chemical group [*]C#CC([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052793 cadmium Inorganic materials 0.000 description 1
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 description 1
- UHYPYGJEEGLRJD-UHFFFAOYSA-N cadmium(2+);selenium(2-) Chemical compound [Se-2].[Cd+2] UHYPYGJEEGLRJD-UHFFFAOYSA-N 0.000 description 1
- AOWKSNWVBZGMTJ-UHFFFAOYSA-N calcium titanate Chemical compound [Ca+2].[O-][Ti]([O-])=O AOWKSNWVBZGMTJ-UHFFFAOYSA-N 0.000 description 1
- NNLOHLDVJGPUFR-UHFFFAOYSA-L calcium;3,4,5,6-tetrahydroxy-2-oxohexanoate Chemical compound [Ca+2].OCC(O)C(O)C(O)C(=O)C([O-])=O.OCC(O)C(O)C(O)C(=O)C([O-])=O NNLOHLDVJGPUFR-UHFFFAOYSA-L 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- RUDATBOHQWOJDD-BSWAIDMHSA-N chenodeoxycholic acid Chemical compound C([C@H]1C[C@H]2O)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(O)=O)C)[C@@]2(C)CC1 RUDATBOHQWOJDD-BSWAIDMHSA-N 0.000 description 1
- 229960001091 chenodeoxycholic acid Drugs 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- CZKMPDNXOGQMFW-UHFFFAOYSA-N chloro(triethyl)germane Chemical compound CC[Ge](Cl)(CC)CC CZKMPDNXOGQMFW-UHFFFAOYSA-N 0.000 description 1
- BHQCQFFYRZLCQQ-OELDTZBJSA-N cholic acid Chemical compound C([C@H]1C[C@H]2O)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(O)=O)C)[C@@]2(C)[C@@H](O)C1 BHQCQFFYRZLCQQ-OELDTZBJSA-N 0.000 description 1
- 229960002471 cholic acid Drugs 0.000 description 1
- 235000019416 cholic acid Nutrition 0.000 description 1
- JAWGVVJVYSANRY-UHFFFAOYSA-N cobalt(3+) Chemical compound [Co+3] JAWGVVJVYSANRY-UHFFFAOYSA-N 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- UIPVMGDJUWUZEI-UHFFFAOYSA-N copper;selanylideneindium Chemical compound [Cu].[In]=[Se] UIPVMGDJUWUZEI-UHFFFAOYSA-N 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 150000003950 cyclic amides Chemical class 0.000 description 1
- 150000004292 cyclic ethers Chemical class 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000003493 decenyl group Chemical group [H]C([*])=C([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000005070 decynyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C#C* 0.000 description 1
- KXGVEGMKQFWNSR-LLQZFEROSA-N deoxycholic acid Chemical compound C([C@H]1CC2)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(O)=O)C)[C@@]2(C)[C@@H](O)C1 KXGVEGMKQFWNSR-LLQZFEROSA-N 0.000 description 1
- 229960003964 deoxycholic acid Drugs 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 125000005066 dodecenyl group Chemical group C(=CCCCCCCCCCC)* 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- CTXJVXIVHPUFIP-UHFFFAOYSA-N ethyl 2-(4-ethoxycarbonylpyridin-2-yl)pyridine-4-carboxylate Chemical compound CCOC(=O)C1=CC=NC(C=2N=CC=C(C=2)C(=O)OCC)=C1 CTXJVXIVHPUFIP-UHFFFAOYSA-N 0.000 description 1
- 125000004705 ethylthio group Chemical group C(C)S* 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- KTWOOEGAPBSYNW-UHFFFAOYSA-N ferrocene Chemical compound [Fe+2].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 KTWOOEGAPBSYNW-UHFFFAOYSA-N 0.000 description 1
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 238000005194 fractionation Methods 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- HZXMRANICFIONG-UHFFFAOYSA-N gallium phosphide Chemical compound [Ga]#P HZXMRANICFIONG-UHFFFAOYSA-N 0.000 description 1
- 239000011245 gel electrolyte Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 125000005980 hexynyl group Chemical group 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-M iodide Chemical compound [I-] XMBWDFGMSWQBCA-UHFFFAOYSA-M 0.000 description 1
- 229940006461 iodide ion Drugs 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- KBPHJBAIARWVSC-RGZFRNHPSA-N lutein Chemical compound C([C@H](O)CC=1C)C(C)(C)C=1\C=C\C(\C)=C\C=C\C(\C)=C\C=C\C=C(/C)\C=C\C=C(/C)\C=C\[C@H]1C(C)=C[C@H](O)CC1(C)C KBPHJBAIARWVSC-RGZFRNHPSA-N 0.000 description 1
- 239000001656 lutein Substances 0.000 description 1
- 229960005375 lutein Drugs 0.000 description 1
- ORAKUVXRZWMARG-WZLJTJAWSA-N lutein Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1=C(C)CCCC1(C)C)C=CC=C(/C)C=CC2C(=CC(O)CC2(C)C)C ORAKUVXRZWMARG-WZLJTJAWSA-N 0.000 description 1
- 235000012680 lutein Nutrition 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- MMIPFLVOWGHZQD-UHFFFAOYSA-N manganese(3+) Chemical compound [Mn+3] MMIPFLVOWGHZQD-UHFFFAOYSA-N 0.000 description 1
- 125000002960 margaryl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229910001511 metal iodide Inorganic materials 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 125000000250 methylamino group Chemical group [H]N(*)C([H])([H])[H] 0.000 description 1
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 229910021421 monocrystalline silicon Inorganic materials 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229910000484 niobium oxide Inorganic materials 0.000 description 1
- URLJKFSTXLNXLG-UHFFFAOYSA-N niobium(5+);oxygen(2-) Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[Nb+5].[Nb+5] URLJKFSTXLNXLG-UHFFFAOYSA-N 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- QGLKJKCYBOYXKC-UHFFFAOYSA-N nonaoxidotritungsten Chemical compound O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1 QGLKJKCYBOYXKC-UHFFFAOYSA-N 0.000 description 1
- 125000005187 nonenyl group Chemical group C(=CCCCCCCC)* 0.000 description 1
- 125000005071 nonynyl group Chemical group C(#CCCCCCCC)* 0.000 description 1
- 125000004365 octenyl group Chemical group C(=CCCCCCC)* 0.000 description 1
- 125000005069 octynyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C#C* 0.000 description 1
- 235000019645 odor Nutrition 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 229910052762 osmium Inorganic materials 0.000 description 1
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000005582 pentacene group Chemical group 0.000 description 1
- 125000003933 pentacenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC5=CC=CC=C5C=C4C=C3C=C12)* 0.000 description 1
- 125000002958 pentadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 125000005981 pentynyl group Chemical group 0.000 description 1
- VLTRZXGMWDSKGL-UHFFFAOYSA-M perchlorate Chemical compound [O-]Cl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-M 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 description 1
- 125000001388 picenyl group Chemical group C1(=CC=CC2=CC=C3C4=CC=C5C=CC=CC5=C4C=CC3=C21)* 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 229910021420 polycrystalline silicon Inorganic materials 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- UKDIAJWKFXFVFG-UHFFFAOYSA-N potassium;oxido(dioxo)niobium Chemical compound [K+].[O-][Nb](=O)=O UKDIAJWKFXFVFG-UHFFFAOYSA-N 0.000 description 1
- 239000011164 primary particle Substances 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 description 1
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- BJDYCCHRZIFCGN-UHFFFAOYSA-N pyridin-1-ium;iodide Chemical compound I.C1=CC=NC=C1 BJDYCCHRZIFCGN-UHFFFAOYSA-N 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000004064 recycling Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 229910052702 rhenium Inorganic materials 0.000 description 1
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 description 1
- YBCAZPLXEGKKFM-UHFFFAOYSA-K ruthenium(iii) chloride Chemical compound [Cl-].[Cl-].[Cl-].[Ru+3] YBCAZPLXEGKKFM-UHFFFAOYSA-K 0.000 description 1
- FZHCFNGSGGGXEH-UHFFFAOYSA-N ruthenocene Chemical compound [Ru+2].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 FZHCFNGSGGGXEH-UHFFFAOYSA-N 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- SUBJHSREKVAVAR-UHFFFAOYSA-N sodium;methanol;methanolate Chemical compound [Na+].OC.[O-]C SUBJHSREKVAVAR-UHFFFAOYSA-N 0.000 description 1
- 239000007784 solid electrolyte Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000003431 steroids Chemical group 0.000 description 1
- VEALVRVVWBQVSL-UHFFFAOYSA-N strontium titanate Chemical compound [Sr+2].[O-][Ti]([O-])=O VEALVRVVWBQVSL-UHFFFAOYSA-N 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- XSOKHXFFCGXDJZ-UHFFFAOYSA-N telluride(2-) Chemical compound [Te-2] XSOKHXFFCGXDJZ-UHFFFAOYSA-N 0.000 description 1
- DPKBAXPHAYBPRL-UHFFFAOYSA-M tetrabutylazanium;iodide Chemical compound [I-].CCCC[N+](CCCC)(CCCC)CCCC DPKBAXPHAYBPRL-UHFFFAOYSA-M 0.000 description 1
- 125000001935 tetracenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC=CC=C4C=C3C=C12)* 0.000 description 1
- GKXDJYKZFZVASJ-UHFFFAOYSA-M tetrapropylazanium;iodide Chemical compound [I-].CCC[N+](CCC)(CCC)CCC GKXDJYKZFZVASJ-UHFFFAOYSA-M 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- QHGNHLZPVBIIPX-UHFFFAOYSA-N tin(ii) oxide Chemical class [Sn]=O QHGNHLZPVBIIPX-UHFFFAOYSA-N 0.000 description 1
- KBPHJBAIARWVSC-XQIHNALSSA-N trans-lutein Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1=C(C)CC(O)CC1(C)C)C=CC=C(/C)C=CC2C(=CC(O)CC2(C)C)C KBPHJBAIARWVSC-XQIHNALSSA-N 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 229910001428 transition metal ion Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- 125000004665 trialkylsilyl group Chemical group 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 125000002306 tributylsilyl group Chemical group C(CCC)[Si](CCCC)(CCCC)* 0.000 description 1
- IEKWPPTXWFKANS-UHFFFAOYSA-K trichlorocobalt Chemical compound Cl[Co](Cl)Cl IEKWPPTXWFKANS-UHFFFAOYSA-K 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000003960 triphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C3=CC=CC=C3C12)* 0.000 description 1
- 229910001930 tungsten oxide Inorganic materials 0.000 description 1
- 125000005065 undecenyl group Chemical group C(=CCCCCCCCCC)* 0.000 description 1
- 229910001935 vanadium oxide Inorganic materials 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- FJHBOVDFOQMZRV-XQIHNALSSA-N xanthophyll Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1=C(C)CC(O)CC1(C)C)C=CC=C(/C)C=CC2C=C(C)C(O)CC2(C)C FJHBOVDFOQMZRV-XQIHNALSSA-N 0.000 description 1
- RNWHGQJWIACOKP-UHFFFAOYSA-N zinc;oxygen(2-) Chemical class [O-2].[Zn+2] RNWHGQJWIACOKP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M14/00—Electrochemical current or voltage generators not provided for in groups H01M6/00 - H01M12/00; Manufacture thereof
- H01M14/005—Photoelectrochemical storage cells
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B57/00—Other synthetic dyes of known constitution
- C09B57/10—Metal complexes of organic compounds not being dyes in uncomplexed form
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/0006—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table compounds of the platinum group
- C07F15/0046—Ruthenium compounds
- C07F15/0053—Ruthenium compounds without a metal-carbon linkage
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B23/00—Methine or polymethine dyes, e.g. cyanine dyes
- C09B23/10—The polymethine chain containing an even number of >CH- groups
- C09B23/102—The polymethine chain containing an even number of >CH- groups two heterocyclic rings linked carbon-to-carbon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G9/00—Electrolytic capacitors, rectifiers, detectors, switching devices, light-sensitive or temperature-sensitive devices; Processes of their manufacture
- H01G9/20—Light-sensitive devices
- H01G9/2059—Light-sensitive devices comprising an organic dye as the active light absorbing material, e.g. adsorbed on an electrode or dissolved in solution
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L31/00—Semiconductor devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation and specially adapted either for the conversion of the energy of such radiation into electrical energy or for the control of electrical energy by such radiation; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof
- H01L31/04—Semiconductor devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation and specially adapted either for the conversion of the energy of such radiation into electrical energy or for the control of electrical energy by such radiation; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof adapted as photovoltaic [PV] conversion devices
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M14/00—Electrochemical current or voltage generators not provided for in groups H01M6/00 - H01M12/00; Manufacture thereof
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/361—Polynuclear complexes, i.e. complexes comprising two or more metal centers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G9/00—Electrolytic capacitors, rectifiers, detectors, switching devices, light-sensitive or temperature-sensitive devices; Processes of their manufacture
- H01G9/20—Light-sensitive devices
- H01G9/2027—Light-sensitive devices comprising an oxide semiconductor electrode
- H01G9/2031—Light-sensitive devices comprising an oxide semiconductor electrode comprising titanium oxide, e.g. TiO2
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/344—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising ruthenium
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/542—Dye sensitized solar cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the present invention relates to a photoelectric conversion element having a binuclear ruthenium complex dye having a substituted bipyridyl group, and a photochemical battery using the photoelectric conversion element.
- Solar cells are highly expected as a clean renewable energy source.
- all of the batteries must be overcome, such as high manufacturing costs, difficulty in securing raw materials, recycling problems, and difficulty in increasing the area. Have problems.
- solar cells using organic materials have been proposed with the aim of increasing the area and reducing the price, but all have a conversion efficiency of about 1%, which is far from practical use.
- This battery is a wet solar cell using a porous titania thin film sensitized with a ruthenium dye as a working electrode.
- the advantage of this solar cell is that it can be used as an inexpensive photoelectric conversion element because an inexpensive material can be used without being purified to a high purity, and further, the absorption of the dye used is broad, and a wide visible light wavelength range. It is possible to convert sunlight into electricity. However, further improvement in conversion efficiency is necessary for practical use, and development of a dye having a higher extinction coefficient and absorbing light up to a longer wavelength region is desired.
- Patent Document 2 discloses a dipyridyl ligand-containing metal mononuclear complex that is a metal complex dye useful as a photoelectric conversion element, and Non-Patent Document 2 discloses a polynuclear ⁇ -diketonate complex dye. ing.
- Patent Document 3 has a plurality of metals and a plurality of ligands as a novel multinuclear complex having an excellent photoelectric conversion function for extracting electrons upon receiving energy of actinic rays such as light.
- a binuclear complex having a coordination structure in which the bridging ligand (BL) coordinated to has a heteroconjugated ring and a coordination structure not having a heteroconjugated ring is disclosed.
- Patent Document 4 discloses a binuclear metal complex having a coordination structure having a heteroconjugate ring as a metal complex dye from which a photoelectric conversion element having high photoelectric conversion efficiency is obtained.
- An object of the present invention is to provide a photoelectric conversion device having a higher absorption coefficient, a light absorption capability up to a longer wavelength region, and capable of converting sunlight into electricity over a wide light wavelength region. It is to provide a metal complex dye for providing a conversion element and a photochemical battery, or a metal complex dye for providing a highly durable photoelectric conversion element and a photochemical battery.
- the present invention relates to the following matters.
- Z represents a 5-membered heteroarylene group
- n represents the number of Z
- R is a hydrogen atom, a linear or branched carbon atom having 1 to 18 carbon atoms.
- R 1 and R 2 each independently represent a hydrogen atom, a linear or branched alkyl group having 1 to 30 carbon atoms, or R 1 and R present in the same pyridine ring
- R 1 and R 2 are bonded to each other to form a ring, provided that two R 1 and two R 2 may be the same or different.
- Ar represents an aryl group which may have a substituent. However, two Ars may be the same or different.) Any one of the groups represented by X represents a counter ion, and m represents the number of counter ions necessary to neutralize the charge of the complex. However, two
- At least one of the above represents a group represented by the formula (2-1) or a group represented by the formula (2-3), wherein n is an integer of 1 to 4, or
- the protons (H + ) of one or more carboxyl groups (—COOH) may be dissociated.
- a photoelectric conversion element comprising the binuclear ruthenium complex dye described in 1 and a semiconductor fine particle.
- a photochemical battery comprising the photoelectric conversion element as described in 2 above.
- a photochemical battery comprising the photoelectric conversion element as described in 2 above and an opposite electrode as an electrode, and an electrolyte layer therebetween.
- a method for producing a photoelectric conversion element comprising a step of immersing semiconductor fine particles in a solution containing the binuclear ruthenium complex dye described in 1 above.
- the present invention it is possible to provide a metal complex dye having a higher extinction coefficient and a light absorption ability up to a longer wavelength region.
- this metal complex dye it may be possible to provide a photoelectric conversion element and a photochemical battery with high photoelectric conversion efficiency that can convert sunlight into electricity over a wide light wavelength range.
- FIG. 1 is a diagram showing ultraviolet-visible absorption spectra of a dinuclear ruthenium complex (1a) and an existing dinuclear ruthenium complex (3).
- FIG. 2 is a diagram showing ultraviolet-visible absorption spectra of the dinuclear ruthenium complex (2a) and the existing dinuclear ruthenium complex (3).
- FIG. 3 is a diagram showing ultraviolet-visible absorption spectra of the dinuclear ruthenium complex (1b) and the existing dinuclear ruthenium complex (5).
- FIG. 4 is a diagram showing ultraviolet-visible absorption spectra of the dinuclear ruthenium complex (2b) and the existing dinuclear ruthenium complex (6).
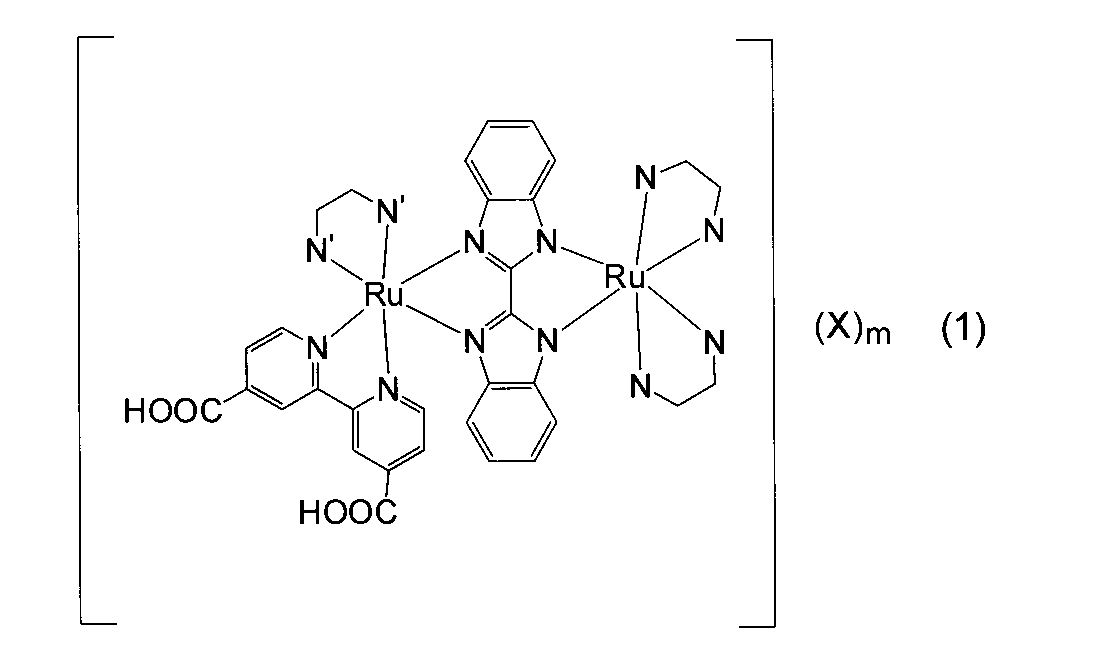
- the binuclear ruthenium complex dye having a substituted bipyridyl group of the present invention is represented by the above general formula (1).
- As the binuclear ruthenium complex dye of the present invention (I) two
- At least one of the above is a group represented by the formula (2-1), wherein n is an integer of 1 to 4, and R is a hydrogen atom or a linear or branched alkyl group having 1 to 18 carbon atoms.
- n is an integer of 1 to 4
- R is a hydrogen atom or a linear or branched alkyl group having 1 to 18 carbon atoms.
- At least one of the groups is a group represented by the formula (2-3) (also referred to as a binuclear ruthenium complex dye (B)).
- (2-3) also referred to as a binuclear ruthenium complex dye (B)
- R 1 and / or R 2 is a group represented by the formula (2-2), which is a linear or branched alkyl group having 1 to 30 carbon atoms (both dinuclear ruthenium complex dye (C)) To tell.) Is preferred.
- n is an integer of 1 to 4 and R is a hydrogen atom or a linear or branched alkyl group having 1 to 18 carbon atoms, or a group represented by the formula (2-1) -2), more preferably R 1 is a linear or branched alkyl group having 1 to 30 carbon atoms, and R 2 is a hydrogen atom.
- R is a hydrogen atom or a linear or branched alkyl group having 1 to 18 carbon atoms, or a group represented by the formula (2-1) -2), more preferably R 1 is a linear or branched alkyl group having 1 to 30 carbon atoms, and R 2 is a hydrogen atom.
- R 1 is a hydrogen atom.
- R 2 is a group represented by the formula (2-2) in which R 2 are bonded to each other to form a benzene ring,
- Ar in the formula (2-3) is preferably a phenyl group which may have a substituent, and the formula (3-1)
- R 3 represents a linear or branched alkyl group having 1 to 18 carbon atoms.
- the group represented by is particularly preferable.
- R 1 is a linear or branched alkyl group having 1 to 30 carbon atoms
- R 2 is a hydrogen atom. 2 or a group represented by the formula (2-2) in which R 1 is a hydrogen atom and R 2 are bonded to each other to form a benzene ring,
- R 1 is a linear or branched alkyl group having 1 to 30 carbon atoms and R 2 is a group represented by the formula (2-2) which is a hydrogen atom.
- Z represents a 5-membered heteroarylene group.
- Z for example, a thiazolyl group is preferable.
- Z may be 1 or 2 to 4, preferably 1 to 3, more preferably 2. That is, in the formula (2-1), n is preferably 1 to 3, and more preferably 2. When n is 2 or more, a plurality of Z may be different. In some cases, Z may be 0, or 0 may be preferable.
- R represents a hydrogen atom, a linear or branched alkyl group having 1 to 18 carbon atoms, or a carboxyl group. Two R may be different.
- R is a hydrogen atom or a linear or branched group such as methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, etc.
- the alkyl group having 1 to 18 carbon atoms is preferable, a hydrogen atom, a linear or branched alkyl group having 1 to 12 carbon atoms is more preferable, and an alkyl group having 1 to 12 carbon atoms is particularly preferable.
- a group represented by the formula (2-1) in which n is 0 and R is a carboxyl group is also particularly preferable.
- Z may have a substituent (that is, any hydrogen atom may be substituted with a substituent).
- substituent include 1 to carbon atoms such as a methyl group, an ethyl group, a propyl group, a butyl group, a pentyl group, a hexyl group, a heptyl group, an octyl group, a nonyl group, a decyl group, an undecyl group, and a dodecyl group.
- alkyl groups alkoxy having 1 to 18 carbon atoms such as methoxy, ethoxy, propoxy, butoxy, pentoxy, hexyloxy, heptyloxy, octyloxy, nonyloxy, decyloxy, undecyloxy, dodecyloxy, etc.
- alkylthio group having 1 to 18 carbon atoms such as a methylthio group, an ethylthio group, a propylthio group, a butylthio group, a pentylthio group, a hexylthio group, a heptylthio group, an octylthio group, a nonylthio group, a decylthio group, an undecylthio group, a dodecylthio group; , N-dimethylamino group, N, N-di Tylamino group, N, N-dipropylamino group, N, N-dibutylamino group, N, N-dipentylamino group, N, N-dihexylamino group, N, N-diheptylamino group, N, N-dioctyl N, N-dialkylamino groups having 1 to 18 carbon atoms such
- Silyl group dimethylsilyl group, diethylsilyl group, dipropylsilyl group, dibutylsilyl group, dipentylsilyl group, dihexylsilyl group, diheptylsilyl group, dioctylsilyl group, dinonylsilyl group, didecylsilyl group, diundecylsilyl group, didodecyl C1-C18 dialkylsilyl groups such as silyl groups; trimethylsilyl groups, triethylsilyl groups, tripropylsilyl groups, tributylsilyl groups, tripentylsilyl groups, trihexylsilyl groups, triheptylsilyl groups, trioctylsilyl groups , Trialkylsilyl groups having 1 to 18 carbon atoms such as trinonylsilyl group, tridecylsilyl group, triundecylsily
- substituents are not particularly limited, and adjacent groups may be bonded to each other to form a ring.
- R 1 and R 2 represent a hydrogen atom, a linear or branched alkyl group having 1 to 30, preferably 1 to 18 carbon atoms, or the same pyridine R 1 and R 2 present in the ring, or R 2 are bonded to each other to form a ring.
- Two R 1 and two R 2 may be different.
- R 1 and R 2 include a hydrogen atom; methyl group, ethyl group, propyl group, butyl group, pentyl group, hexyl group, heptyl group, octyl group, nonyl group, decyl group, undecyl group, dodecyl group, tridecyl Group, tetradecyl group, pentadecyl group, hexadecyl group, heptadecyl group, and octadecyl group.
- R 1 is a linear or branched alkyl group having 1 to 18 carbon atoms, more preferably a linear or branched alkyl group having 1 to 12 carbon atoms
- R 2 is preferably a hydrogen atom.
- R 1 and R 2 may be substituted, and may have a substituent represented by Z.
- substituents are not particularly limited, and adjacent groups may be bonded to each other to form a ring.
- R 1 and R 2 present in the same pyridine ring, or R 2 may be bonded to each other to form a ring.
- the ring formed include a non-conjugated ring such as a cyclohexane ring; a conjugated ring such as a benzene ring, a naphthalene ring, an anthracene ring, and a pentacene ring, preferably a conjugated ring, and more preferably Is a benzene ring.
- R 1 is a hydrogen atom
- R 2 is bonded to each other to form a ring, preferably a benzene ring.
- the formed ring may be substituted or may have a substituent represented by Z.
- these groups include various isomers.
- Ar represents an aryl group which may have a substituent, for example, a phenyl group, a naphthyl group, an anthryl group, a tetracenyl group, a pentacenyl group, an azulenyl group, a fluorenyl group, a phenanthrenyl group, Examples include triphenylenyl group, pyrenyl group, chrycenyl group, picenyl group, perylenyl group, pentaferenyl group, dibenzophenanthrenyl group and the like.
- Ar for example, a substituted or unsubstituted phenyl group is preferable.
- Two Ar may be different.
- Ar may have a substituent (that is, an arbitrary hydrogen atom may be substituted with a substituent), and examples of the substituent include the substituent represented by Z.
- substituents that is, an arbitrary hydrogen atom may be substituted with a substituent
- the number and position of these substituents are not particularly limited, and adjacent groups may be bonded to each other to form a ring.
- R 3 represents a linear or branched alkyl group having 1 to 18 carbon atoms. Is particularly preferred.
- R3 examples include a methyl group, an ethyl group, a propyl group, a butyl group, a pentyl group, a hexyl group, a heptyl group, an octyl group, a nonyl group, a decyl group, an undecyl group, and a dodecyl group.
- X represents a counter ion, for example, hexafluorophosphate ion, perchlorate ion, tetraphenylborate ion, tetrafluoroborate ion, trifluoromethanesulfonate ion, thiocyanic acid.
- m represents the number of counter ions necessary to neutralize the charge of the complex.
- the binuclear ruthenium complex dye (binuclear ruthenium complex dye (A)) having a five-membered heteroaryl group-substituted bipyridyl group of the present invention can be obtained by referring to WO2006 / 038587 and the like, for example, as shown in the following formula: It is obtained by reacting different mononuclear ruthenium complexes.
- X ⁇ represents a monovalent anion which is a counter ion.
- X ⁇ represents a monovalent anion which is a counter ion
- Y represents a halogen atom
- L represents a neutral molecule, for example, an organic compound such as a water molecule or acetone. Solvent molecule etc., where cod is 1,5-cyclooctadiene.
- the counter ion (X) is not limited to a monovalent anion, but other ions can be synthesized in the same manner.
- the binuclear ruthenium complex dye (binuclear ruthenium complex dye (B)) having an unsaturated group-substituted bipyridyl group of the present invention is obtained by reacting different mononuclear ruthenium complexes with each other as shown in the following formula, for example. It is done.
- X ⁇ represents a monovalent anion which is a counter ion
- Y represents a halogen atom
- Z represents a neutral molecule.
- Cod represents 1,5-cyclooctadiene. .
- the counter ion (X) is not limited to a monovalent anion, but other ions can be synthesized in the same manner.
- One mononuclear ruthenium complex is once synthesized via a mononuclear ruthenium complex precursor, but is a synthesis intermediate of the general formula (2)
- the compound of the general formula (3) may have one NH proton as in the general formula (4).
- the dinuclear ruthenium complex dye (binuclear ruthenium complex dye (B)) having another unsaturated group-substituted bipyridyl group of the present invention, for example, reacts different mononuclear ruthenium complexes with each other as shown in the following formula. Obtained by.
- the counter ion (X) is not limited to a monovalent anion, but other ions can be synthesized in the same manner.
- the binuclear ruthenium complex dye (binuclear ruthenium complex dye (C)) having a substituted bipyridyl group according to the present invention is, for example, a ligand of [Ru (p-cymene) Cl 2 ] 2 as shown in the following formula: It is obtained by sequentially reacting mononuclear ruthenium complexes.
- R 1 and R 2 are as defined above.
- X ⁇ represents a monovalent anion which is a counter ion.
- the counter ion (X) is not limited to a monovalent anion, but other ions can be synthesized in the same manner.
- protons (H + ) of one or more carboxyl groups (—COOH) may be dissociated.
- Proton (H + ) dissociation is mainly performed by adjusting the pH of the solution.
- the photoelectric conversion element of the present invention contains the binuclear ruthenium complex dye and semiconductor fine particles.
- the dinuclear ruthenium complex dye is adsorbed on the surface of the semiconductor fine particles, and the semiconductor fine particles are sensitized with the ruthenium complex dye.
- the photoelectric conversion element of the present invention is obtained by fixing semiconductor fine particles sensitized with the ruthenium complex dye on a conductive support (electrode).
- the conductive electrode is preferably a transparent electrode formed on a transparent substrate.
- the conductive agent include metals such as gold, silver, copper, platinum, and palladium, indium oxide compounds represented by tin-doped indium oxide (ITO), and fluorine-doped tin oxide (FTO). Examples thereof include tin oxide compounds and zinc oxide compounds.
- the semiconductor fine particles include titanium oxide, zinc oxide, and tin oxide. Also, complex oxide semiconductors such as indium oxide, niobium oxide, tungsten oxide, vanadium oxide, strontium titanate, calcium titanate, barium titanate, potassium niobate, cadmium or bismuth sulfide, cadmium selenide or telluride And gallium phosphide or arsenide. As the semiconductor fine particles, oxides are preferable, and for example, titanium oxide, zinc oxide, tin oxide, or a mixture containing any one or more of these is particularly preferable.
- the primary particle size of the semiconductor fine particles is not particularly limited, but is usually 1 to 5000 nm, preferably 2 to 500 nm, and particularly preferably 5 to 400 nm.
- a semiconductor layer containing semiconductor fine particles is formed on a conductive support and then immersed in a solution containing the dinuclear ruthenium complex dye.
- the semiconductor layer can be formed by applying a paste of semiconductor fine particles on a conductive support and heating and baking. And after immersing in a pigment
- the solvent for the dye solution examples include water; alcohols such as methanol, ethanol, isopropyl alcohol, t-butyl alcohol, and ethylene glycol; nitriles such as acetonitrile and propionitrile; N, N-dimethylacetamide, N, N Amides such as dimethylformamide; ureas such as N-methylpyrrolidone; sulfoxides such as dimethyl sulfoxide, and the like, preferably water, alcohols, nitriles, more preferably water, ethanol, isopropyl alcohol, t- Butanol and acetonitrile are used.
- these solvents may be used independently and may mix and use 2 or more types.
- the concentration of the dye in the solution is preferably 0.001 mmol / l to the saturation concentration of each complex dye of the present invention, more preferably 0.001 to 100 mmol / l, particularly preferably 0.01 to 10 mmol / l, and more. Preferably, it is 0.05 to 1.0 mmol / l.
- a compound having a steroid skeleton such as cholic acid, deoxycholic acid, chenodeoxycholic acid may be added to the dye solution.
- the temperature for adsorbing the dye is usually 0 to 80 ° C., preferably 20 to 40 ° C.
- the time for adsorbing the dye can be appropriately determined according to conditions such as the type and concentration of the dinuclear ruthenium complex dye.
- the photochemical battery of the present invention uses the photoelectric conversion element of the present invention as described above. More specifically, the photoelectric conversion element of the present invention and a counter electrode are provided as electrodes, and an electrolyte layer is provided therebetween. At least one of the electrode using the photoelectric conversion element of the present invention and the counter electrode is a transparent electrode.
- the counter electrode functions as a positive electrode when combined with a photoelectric conversion element to form a photochemical battery.
- a substrate having a conductive layer can be used as in the case of the conductive electrode.
- the conductive agent used for the counter electrode include metals such as platinum and carbon, and conductive metal oxides such as tin oxide doped with fluorine.
- the electrolyte (redox couple) is not particularly limited, and any known one can be used.
- iodine and iodide for example, metal iodides such as lithium iodide and potassium iodide, or quaternary ammonium compounds such as tetrabutylammonium iodide, tetrapropylammonium iodide, pyridinium iodide, imidazolium iodide) Iodide), bromine and bromide, chlorine and chloride, alkyl viologen and its reduced form, quinone / hydroquinone, iron (II) ion / iron (III) ion, copper (I) ion / Transition metal ion pairs such as copper (II) ion, manganese (II) ion / manganese (III) ion, cobalt ion (II) / cobalt ion (III), ferro
- a preferable electrolyte is an electrolyte in which iodine and lithium iodide or iodide of a quaternary ammonium compound are combined.
- the state of the electrolyte may be a liquid dissolved in an organic solvent, a molten salt, a so-called gel electrolyte immersed in a polymer matrix, or a solid electrolyte.
- the solvent for the electrolyte examples include water, alcohols, nitriles, chain ethers, cyclic ethers, chain esters, cyclic esters, chain amides, cyclic amides, chain sulfones, cyclic Sulfones, chain ureas, cyclic ureas, amines and the like are used.
- the said solvent is not limited to these, It can use individually or in mixture of 2 or more types.
- the photochemical battery of the present invention can be manufactured by a conventionally applied method.
- a semiconductor fine particle paste such as an oxide is applied on a transparent electrode and heated and fired to produce a thin film of semiconductor fine particles.
- the thin film of semiconductor fine particles is titania, it is fired, for example, at a temperature of 450 to 500 ° C. and a heating time of 30 minutes.
- the transparent electrode with the thin film is immersed in a dye solution (a solution containing the dinuclear ruthenium complex dye of the present invention), and the dye is supported to produce a photoelectric conversion element.
- the photochemical cell of the present invention can be manufactured by combining this photoelectric conversion element with a transparent electrode on which platinum or carbon is deposited as a counter electrode, and putting an electrolyte solution therebetween.
- reaction solution was allowed to cool, H 2 dcbpy (0.08 g, 0.328 mmol) was added, the mixture was deaerated again, and reacted at 140 ° C. for 21.5 hours. After allowing the reaction solution to cool, 0.030 g of sodium hydroxide and [Ru (dnbpy) 2 (BiBzIm)] (0.330 g, 0.294 mmol) were added and reacted at 140 ° C. for 6.5 hours.
- reaction solution was filtered, the filtrate was concentrated under reduced pressure, 30 mL of acetone and 90 mL of pH 2.4 hexafluorophosphoric acid aqueous solution were added to the concentrate, stirred for 1 hour at room temperature, filtered, and acetone: pH 2.
- a typical structure of the dinuclear ruthenium complex dye is shown in Formula (1a). Some of the complex dyes have one or more protons of the carboxyl group dissociated.
- Example A2 (Synthesis of dinuclear ruthenium complex dye (2a))
- Example A2-A (Synthesis of mononuclear ruthenium complex (M-1); [Ru (Etcbpy) 2 (H 2 O) 2 ] (OTf) 2 )
- H 2 dcbpy 5.44 g, 22.3 mmol
- concentrated sulfuric acid 10 mL
- ethanol 130 mL were added to a 500 mL three-necked flask, and the mixture was refluxed overnight for reaction.
- the reaction solution was allowed to cool, neutralized, and filtered.
- the residue was washed with hot water and recrystallized with ethanol / water (95: 5).
- the crystals were filtered and dried under vacuum to obtain 4.92 g of Etcbpy.
- Example A2-B Synthesis of mononuclear ruthenium complex (M-2) [(BiBzIm) Ru (BiHeBiTbpy) 2 ]) BiHeBiTbpy (0.375 g, 0.574 mmol), [Ru (cod) Cl 2 ] n (0.096 g, 0.344 mmol) and N, N-dimethylacetamide 36 mL were added to a 200 mL three-necked flask and deaerated. Thereafter, the mixture was refluxed for 24 minutes with stirring under 2.45 GHz microwave irradiation.
- Example A2-C Synthesis of dinuclear ruthenium complex dye (2a)
- M-1 0.038 g, 0.038 mmol
- M-2 0.059 g, 0.036 mmol
- N N— Degassed by adding 12.5 mL of dimethylacetamide. Thereafter, the mixture was refluxed for 24 minutes with stirring under 2.45 GHz microwave irradiation.
- the obtained reaction solution was filtered, the filtrate was concentrated under reduced pressure, and 16.3 mL of 0.2 mol / L sodium hydroxide aqueous solution was added to the concentrate, followed by heating at 100 ° C. for 2 hours. The reaction solution was allowed to cool and then filtered. After adding 7.5 mL of methanol, 7.5 mL of water, and 0.02 mL of 1 mol / L sodium hydroxide aqueous solution to the filtrate and performing ultrasonic agitation for 15 minutes, a solution using 0.72 mol / L hexafluorophosphoric acid aqueous solution was used. Was adjusted to pH 3.8 and allowed to stand overnight.
- This dinuclear ruthenium complex dye is shown in Formula (2a). Some of the complex dyes have one or more protons of the carboxyl group dissociated.
- Example A3-1 (Preparation of porous titania electrode)
- the catalyst chemical titania paste PST-18NR was applied to the transparent layer, and PST-400C was applied to the diffusion layer, and applied onto a transparent conductive glass electrode manufactured by Asahi Glass Co., Ltd. using a screen printer.
- the obtained film was aged for 5 minutes in an atmosphere of 25 ° C. and a relative humidity of 60%, and the aged film was baked at 450 ° C. for 30 minutes. The same operation was repeated on the cooled membrane until a predetermined thickness was obtained, thereby producing a 16 mm 2 porous titania electrode.
- Example A3-2 Preparation of porous titania electrode adsorbed with dye
- Porous titania electrode is immersed in a 0.2 mmol / l dye solution of dinuclear ruthenium complex dye (solvent: 1: 1 mixed solvent of t-butanol / acetonitrile) at 30 ° C. for a predetermined time, dried and dye-adsorbed porous A titania electrode was obtained.
- Example A3-3 (Production of photochemical battery) The dye adsorbing porous titania electrode obtained as described above and a platinum plate (counter electrode) were superposed. Next, an electrolyte solution (3-methoxypropionitrile, lithium iodide, iodine, 4-t-butylpyridine and 1,2-dimethyl-3-propylimidazolium iodide was added at 0.1 mol / l and 0.05 mol, respectively. / L, 0.5 mol / l, and 0.6 mol / l) were soaked into the gaps between the two electrodes using a capillary phenomenon to produce a photochemical battery.
- an electrolyte solution (3-methoxypropionitrile, lithium iodide, iodine, 4-t-butylpyridine and 1,2-dimethyl-3-propylimidazolium iodide was added at 0.1 mol / l and 0.05 mol, respectively.
- Example A4 (measurement of UV-visible absorption spectrum) A 3 ⁇ 10 ⁇ 5 mol / L ethanol solution was prepared for each of the following complexes, and an ultraviolet-visible absorption spectrum was measured.
- Binuclear ruthenium complex of the present invention (1a; synthesized in Example A1)
- Binuclear ruthenium complex of the present invention (2a; synthesized in Example A2)
- Existing dinuclear ruthenium complex (3; synthesized in Reference Example A1)
- the ultraviolet-visible absorption spectrum of the binuclear ruthenium complex (1a) and the binuclear ruthenium complex (3) is shown in FIG. 1, and the ultraviolet-visible absorption spectrum of the binuclear ruthenium complex (2a) and the binuclear ruthenium complex (3) is shown in FIG. .
- the complex of the present invention (binuclear ruthenium complex dye), that is, the complex (1a) in which a [2,2′-bithiophene] -5-yl group is introduced at the 4-position and the 4′-position of the 2,2′-bipyridine ring,
- the absorption wavelength region is increased by about 40 nm, and the absorbance at the peak at the longest wavelength side under the same concentration condition is 1 .3 times increase.
- the complex (2a) of the present invention has a light absorption wavelength region shifted to the long wavelength side of about 58 nm as compared with the known binuclear ruthenium complex (3) in which the 4th and 4 ′ positions are unsubstituted, and 350 The absorbance around ⁇ 450 nm was greatly improved. This revealed that the complex of the present invention can be a dye for producing a high-performance photochemical battery.
- Example B1-A mononuclear ruthenium complex (M-1); synthesis of [Ru (Etcbpy) 2 (H 2 O) 2 ] (OTf) 2 ) [Ru (Etcbpy) 2 (H 2 O) 2 ] (OTf) 2 was synthesized in the same manner as Example A2-A.
- the mononuclear ruthenium complex (M-1) (0.141 g, 0.139 mmol)
- the mononuclear ruthenium complex (M-3) (0.202 g, 0.139 mmol)
- N, N -Degassed 25 mL of dimethylacetamide. Thereafter, the mixture was refluxed for 22 minutes with stirring under microwave irradiation of 2.45 GHz. The resulting reaction solution was allowed to cool and then concentrated under reduced pressure.
- a typical structure of this dinuclear ruthenium complex dye is shown in Formula (1b). Some of the complex dyes have one or more protons of the carboxyl group dissociated.
- Example B2 (Production of photochemical battery) In the same manner as in Examples A3-1 to A3-3, a dye-adsorbing porous titania electrode was produced, and a photochemical battery was produced.
- Example B3 (measurement of UV-visible absorption spectrum) A 3 ⁇ 10 ⁇ 5 mol / L ethanol solution was prepared for each of the following complexes, and an ultraviolet-visible absorption spectrum was measured.
- Binuclear ruthenium complex of the present invention (1b; synthesized in Example B1)
- (2) Existing dinuclear ruthenium complex (5; synthesized in Reference Example B2)
- FIG. 3 shows ultraviolet-visible absorption spectra of the binuclear ruthenium complex (1b) and the existing dinuclear ruthenium complex (5).
- the complex of the present invention (binuclear ruthenium complex dye), that is, the complex (1b) having a styryl group introduced at the 4th and 4 'positions of the 2,2'-bipyridine ring has no known 4th and 4' positions.
- the light absorption wavelength range is shifted to the long wavelength side by about 40 nm, and the absorbance at the same concentration condition at the peak of the longest wavelength side is increased 1.1 times.
- the complex of the present invention can be a dye for producing a high-performance photochemical battery.
- reaction solution was allowed to cool, H 2 dcbpy (0.160 g, 0.655 mmol) was added, degassed again, and reacted at 140 ° C. for 8.5 hours. After allowing the reaction solution to cool, 1.32 mL of a 1 mol / L aqueous sodium hydroxide solution and [Ru (phen) 2 (BiBzIm)] (0.408 g, 0.588 mmol) were added, and the reaction was allowed to reflux for 3 hours.
- the obtained reaction solution was concentrated under reduced pressure, and 30 mL of methanol and 0.1 mL of 2 mol / L hydrochloric acid were added to the concentrate, followed by filtration.
- Fractionation gave a fraction containing the desired product.
- the obtained fraction was concentrated under reduced pressure, and the concentrate was suspended in methanol, acetone and 0.4 mol / L hexafluorophosphoric acid aqueous solution (pH 2), and then allowed to stand overnight.
- the obtained solid was filtered to obtain 0.116 g of a binuclear ruthenium complex dye (2b).
- Example B5 (Production of photochemical battery) In the same manner as in Examples A3-1 to A3-3, a dye-adsorbing porous titania electrode was produced, and a photochemical battery was produced.
- Example B6 (measurement of UV-visible absorption spectrum) A 3 ⁇ 10 ⁇ 5 mol / L ethanol solution was prepared for each of the following complexes, and an ultraviolet-visible absorption spectrum was measured.
- FIG. 4 shows ultraviolet-visible absorption spectra of the binuclear ruthenium complex (2b) and the existing binuclear ruthenium complex (6).
- Example B7 Measurement of photoelectric conversion efficiency
- the photoelectric conversion efficiency of the obtained photochemical battery was simulated using a solar simulator manufactured by Eihiro Seiki Co., Ltd. of 100 mW / cm 2 . Sunlight was irradiated and measured. The results are shown in Table 1.
- the complex of the present invention (binuclear ruthenium complex dye), that is, the complex (2b) in which a styryl group is introduced at the 4th and 4 ′ positions of the 2,2′-bipyridine ring is a known dicarboxylic acid at the 4th and 4 ′ positions.
- the light absorption wavelength range was shifted to the longer wavelength side by about 40 nm, and the extinction coefficient at the peak on the longest wavelength side was increased 1.1 times. Furthermore, a relatively high photoelectric conversion efficiency was exhibited. From these facts, it was found that the complex of the present invention can be a dye for producing a high-performance photochemical battery.
- reaction solution was allowed to cool, H 2 dcbpy (0.080 g, 0.328 mmol) was added, the mixture was degassed again, and reacted at 160 ° C. for 4 hours. After allowing the reaction solution to cool, 0.67 mL of a 1 mol / L aqueous sodium hydroxide solution and [Ru (phen) 2 (BiBzIm)] (0.207 g, 0.298 mmol) were added and reacted while refluxing for 5.5 hours. .
- reaction solution was concentrated under reduced pressure, the concentrate was dissolved in 30 mL of methanol, 8 mL of a hexafluorophosphoric acid aqueous solution having pH 2 was added, and the precipitated solid was filtered.
- a typical structure of this dinuclear ruthenium complex dye is shown in Formula (C2). Some of the complex dyes have one or more protons of the carboxyl group dissociated.
- a 200 mL three-necked flask was degassed by adding dichloro-p-cymene ruthenium dimer (0.200 g, 0.327 mmol), dnbpy (0.277 g, 0.657 mmol) and 100 mL of N, N-dimethylformamide. . Then, it was made to react at 60 degreeC for 4 hours.
- reaction solution was allowed to cool, H 2 dcbpy (0.160 g, 0.656 mmol) was added, the mixture was degassed again, and reacted at 160 ° C. for 4 hours. After allowing the reaction solution to cool, 1.34 mL of a 1 mol / L aqueous sodium hydroxide solution and [Ru (dnbpy) 2 (BiBzIm)] (0.678 g, 0.589 mmol) were added, and the reaction was allowed to reflux for 2 hours.
- the obtained reaction solution is concentrated under reduced pressure, and the concentrate is dissolved in methanol and separated by liquid chromatography (developing solvent: methanol (containing 0.2% by volume of formic acid)) to contain the desired product.
- the fraction to be obtained was obtained.
- the obtained fraction was concentrated under reduced pressure, and the concentrate was dissolved in 2.5 mL of methanol. Thereafter, 2.5 mL of a pH 2 hexafluorophosphoric acid aqueous solution was added. The precipitated solid was filtered to obtain 0.096 g of a binuclear ruthenium complex dye (C3).
- This dinuclear ruthenium complex dye is shown in Formula (C3). Some of the complex dyes have one or more protons of the carboxyl group dissociated.
- Example C3 (Preparation of a photochemical battery) In the same manner as in Examples A3-1 to A3-3, a dye-adsorbing porous titania electrode was produced, and a photochemical battery was produced.
- Example C4 (durability evaluation) The obtained photochemical battery was allowed to stand in a dark place at 60 ° C. for a predetermined time, then returned to room temperature, and the photoelectric conversion efficiency ( ⁇ ) was changed to 100 mW / cm 2 of pseudo-sunlight using a solar simulator manufactured by Eihiro Seiki Co., Ltd. Irradiated and measured. Table 2 shows the maintenance ratio of the photoelectric conversion efficiency after 5 days when the photoelectric conversion efficiency after 100 days in the dark at 60 ° C. of each complex dye is defined as 100%.
- the present invention it is possible to provide a metal complex dye having a higher extinction coefficient and a light absorption ability up to a longer wavelength region.
- the metal complex dye of the present invention it is possible to absorb a wider range of light, convert sunlight into electricity over a wide light wavelength range, and highly efficient photoelectric that can absorb more sunlight.
- a conversion element and a photochemical battery can be provided.
- a photoelectric conversion element and a photochemical battery having high durability can be provided.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Power Engineering (AREA)
- Materials Engineering (AREA)
- General Chemical & Material Sciences (AREA)
- Electrochemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Physics & Mathematics (AREA)
- High Energy & Nuclear Physics (AREA)
- Inorganic Chemistry (AREA)
- Computer Hardware Design (AREA)
- General Physics & Mathematics (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Electromagnetism (AREA)
- Hybrid Cells (AREA)
- Photovoltaic Devices (AREA)
- Pyridine Compounds (AREA)
Abstract
Description
(式中、Zは五員環へテロアリーレン基を示し、nはZの個数を表し、0~4の整数であり、Rは水素原子、直鎖又は分岐状の炭素原子数1~18のアルキル基又はカルボキシル基を示す。ただし、2個のR及び複数個のZは同一でも異なっていてもよい。)
で示される基、又は式(2-2)
で示される基、又は式(2-3)
で示される基のいずれかを示す。Xは対イオンを示し、mは錯体の電荷を中和するのに必要な対イオンの数を示す。但し、2つの
この半導体層を前記1記載の二核ルテニウム錯体色素を含む溶液に浸漬する工程と
を有することを特徴とする光電変換素子の製造方法。
(i)2つの
(ii)2つの
bpy;2,2’-ビピリジン
dnbpy;4,4’-ジノニル-2,2’-ビピリジン
H2dcbpy;2,2’-ビピリジン-4,4’-ジカルボン酸
Etcbpy;2,2’-ビピリジン-4,4’-ジカルボン酸ジエチルエステル
BiBzImH2,BiBzIm;2,2’-ビベンズイミダゾール
BiHeBiTbpy;4,4’-ビス(5’-ヘキシル-[2,2’-ビチオフェン]-5-イル)-2,2’-ビピリジン
BiHexoStbpy;4,4’-ビス(4-(ヘキシロキシ)スチリル)-2,2’-ビピリジン
phen;1,10-フェナントロリン
OTf:トリフルオロメタンスルホン酸イオン
cod;1,5-シクロオクタジエン
実施例A1(二核ルテニウム錯体色素(1a)の合成)
アルゴン雰囲気下、200mLの三口フラスコにジクロロ-p-シメンルテニウムダイマー(0.100g,0.163mmol)、BiHeBiTbpy(0.214g,0.328mmol)及びN,N-ジメチルホルムアミド50mLを加えて脱気した。その後、60℃で4時間反応させた。
実施例A2-A(単核ルテニウム錯体(M-1);[Ru(Etcbpy)2(H2O)2](OTf)2の合成)
窒素雰囲気下、500mLの三口フラスコに市販のH2dcbpy(5.44g,22.3mmol)、濃硫酸(10mL)及びエタノール130mLを加え、一晩還流し、反応させた。反応液を放冷後、中和し、濾過した。濾物を熱水で洗浄し、エタノール/水(95:5)で再結晶を行った。結晶を濾過し、真空乾燥し、Etcbpy4.92gを得た。
200mLの三口フラスコにBiHeBiTbpy(0.375g,0.574mmol)、[Ru(cod)Cl2]n(0.096g,0.344mmol)及びN,N-ジメチルアセトアミド36mLを加えて脱気した。その後、2.45GHzのマイクロ波照射下にて攪拌しながら24分間還流させた。反応液を放冷後、吸引濾過によって濾物を回収し、N,N-ジメチルアセトアミドで洗浄、真空乾燥し、[Ru(BiHeBiTbpy)2Cl2]0.371gを得た。
アルゴン雰囲気下、20mLのシュレンクに単核ルテニウム錯体(M-1)(0.038g,0.038mmol)、単核ルテニウム錯体(M-2)(0.059g,0.036mmol)及びN,N-ジメチルアセトアミド12.5mLを加えて脱気した。その後、2.45GHzのマイクロ波照射下にて攪拌しながら24分間還流させた。得られた反応液を濾過し、その濾液を減圧下で濃縮し、濃縮物に0.2mol/L水酸化ナトリウム水溶液16.3mLを加え、100℃で2時間加熱した。反応液を放冷後、濾過した。濾物にメタノール7.5mL、水7.5mL、1mol/L水酸化ナトリウム水溶液0.02mLを加え、超音波攪拌を15分行った後、0.72mol/Lヘキサフルオロリン酸水溶液を用いて溶液のpHを3.8に調整し、一晩静置した。その後、濾過し、濾物をpH3.8ヘキサフルオロリン酸水溶液、アセトン:ジエチルエーテル=1:8の溶液で洗浄し、真空乾燥後、メタノール12mL、水12mL、1mol/L水酸化ナトリウム水溶液0.02mLを加え、超音波攪拌を15分行った後、0.72mol/Lヘキサフルオロリン酸水溶液を用いて溶液のpHを2.8に調整し、一晩静置した。その後、濾過し、濾物をpH2.8ヘキサフルオロリン酸水溶液で洗浄し、二核ルテニウム錯体色素(2a)0.034gを得た。
既知の方法で二核ルテニウム錯体色素(3)を合成した。
触媒化成製のチタニアペーストPST-18NRを透明層に、PST-400Cを拡散層に用い、旭硝子株式会社製透明導電性ガラス電極上にスクリーン印刷機を用いて塗布した。得られた膜を25℃、相対湿度60%の雰囲気下で5分間エージングし、このエージングした膜を450℃で30分間焼成した。冷却した膜に対し、同じ作業を所定の厚みになるまで繰り返し、16mm2の多孔質チタニア電極を作製した。
二核ルテニウム錯体色素の0.2mmol/l色素溶液(溶媒:t-ブタノール/アセトニトリルの1:1混合溶媒)に多孔質チタニア電極を30℃で所定の時間浸漬し、乾燥して色素吸着多孔質チタニア電極を得た。
以上のようにして得られた色素吸着多孔質チタニア電極と白金板(対極)を重ね合わせた。次に、電解質溶液(3-メトキシプロピオニトリルにヨウ化リチウム、ヨウ素、4-t-ブチルピリジン及び1,2-ジメチル-3-プロピルイミダゾリウムアイオダイドをそれぞれ0.1mol/l、0.05mol/l、0.5mol/l、0.6mol/lとなるように溶解したもの)を両電極の隙間に毛細管現象を利用して染み込ませることにより光化学電池を作製した。
以下の錯体それぞれについて3×10-5mol/Lエタノール溶液を調製し、紫外可視吸収スペクトルを測定した。
(1)本発明の二核ルテニウム錯体(1a;実施例A1で合成)
(2)本発明の二核ルテニウム錯体(2a;実施例A2で合成)
(3)既存の二核ルテニウム錯体(3;参考例A1で合成)
二核ルテニウム錯体(1a)と二核ルテニウム錯体(3)の紫外可視吸収スペクトルを図1に、二核ルテニウム錯体(2a)と二核ルテニウム錯体(3)の紫外可視吸収スペクトルを図2に示す。
参考例B1(BiHexoStbpyの合成)
Journal Of American Chemical Society,2006年,128巻,4146-4154頁を参考に合成した。
実施例B1-A(単核ルテニウム錯体(M-1);[Ru(Etcbpy)2(H2O)2](OTf)2の合成)
実施例A2-Aと同様にして、[Ru(Etcbpy)2(H2O)2](OTf)2を合成した。
200mLの三口フラスコにBiHexoStbpy(1.106g,1.973mmol)、[Ru(cod)Cl2]n(0.268g,0.957mmol)及びN,N-ジメチルホルムアミド100mLを加えて脱気した。その後、2.45GHzのマイクロ波照射下にて攪拌しながら34分間還流させた。反応液を放冷後、吸引濾過によって濾物を回収し、N,N-ジメチルホルムアミドで洗浄、真空乾燥し、[Ru(BiHexoStbpy)2Cl2]0.779gを得た。
アルゴン雰囲気下、100mLの三口フラスコに単核ルテニウム錯体(M-1)(0.141g,0.139mmol)、単核ルテニウム錯体(M-3)(0.202g,0.139mmol)及びN,N-ジメチルアセトアミド25mLを加えて脱気した。その後、2.45GHzのマイクロ波照射下にて攪拌しながら22分間還流させた。得られた反応液を放冷後、減圧下で濃縮し、濃縮物に0.2mol/L水酸化ナトリウム水溶液36mLを加え、100℃で2時間加熱した。反応液を放冷後、濾過した。濾物をアセトン:ジエチルエーテル=1:8の溶液で洗浄した。その後、濾物を水50mLに懸濁させ、0.72mol/Lヘキサフルオロリン酸水溶液を用いて溶液のpHを3.5に調整し、一晩静置した。その後、濾過し、濾物をpH3.5ヘキサフルオロリン酸水溶液、アセトン:ジエチルエーテル=1:8の溶液、及びジエチルエーテルで洗浄し、真空乾燥し、二核ルテニウム錯体色素(1b)0.125gを得た。
既知の方法で二核ルテニウム錯体色素(5)を合成した。
実施例A3-1~A3-3と同様にして、色素吸着多孔質チタニア電極を作製し、光化学電池を作製した。
以下の錯体それぞれについて3×10-5mol/Lエタノール溶液を調製し、紫外可視吸収スペクトルを測定した。
(1)本発明の二核ルテニウム錯体(1b;実施例B1で合成)
(2)既存の二核ルテニウム錯体(5;参考例B2で合成)
二核ルテニウム錯体(1b)と既存の二核ルテニウム錯体(5)の紫外可視吸収スペクトルを図3に示す。
アルゴン雰囲気下、300mLの三口フラスコにジクロロ-p-シメンルテニウムダイマー(0.200g,0.326mmol)、BiHexoStbpy(0.366g,0.653mmol)及びN,N-ジメチルホルムアミド100mLを加えて脱気した。その後、60℃で3.5時間反応させた。
既知の方法で二核ルテニウム錯体色素(6)を合成した。
実施例A3-1~A3-3と同様にして、色素吸着多孔質チタニア電極を作製し、光化学電池を作製した。
以下の錯体それぞれについて3×10-5mol/Lエタノール溶液を調製し、紫外可視吸収スペクトルを測定した。
(1)本発明の二核ルテニウム錯体(2b;実施例B4で合成)
(2)既存の二核ルテニウム錯体(6;参考例B3で合成)
二核ルテニウム錯体(2b)と既存の二核ルテニウム錯体(6)の紫外可視吸収スペクトルを図4に示す。
本発明の二核ルテニウム錯体(2b)と既存の二核ルテニウム錯体(6)について、得られた光化学電池の光電変換効率を、英弘精機株式会社製のソーラーシュミレーターを用い、100mW/cm2の擬似太陽光を照射し測定した。その結果を表1に示す。
実施例C1(二核ルテニウム錯体色素(C2)〔左側のRuの配位子:R1=ノニル基、R2=水素原子;右側のRuの配位子:R2同士が互いに結合してベンゼン環を形成〕の合成)
アルゴン雰囲気下、100mLの三口フラスコにジクロロ-p-シメンルテニウムダイマー(0.100g,0.164mmol)、dnbpy(0.139g,0.329mmol)及びN,N-ジメチルホルムアミド50mLを加えて脱気した。その後、60℃で4時間反応させた。
アルゴン雰囲気下、200mLの三口フラスコにジクロロ-p-シメンルテニウムダイマー(0.200g,0.327mmol)、dnbpy(0.277g,0.657mmol)及びN,N-ジメチルホルムアミド100mLを加えて脱気した。その後、60℃で4時間反応させた。
既知の方法で二核ルテニウム錯体色素(C4)を合成した。
既知の方法で二核ルテニウム錯体色素(C5)を合成した。
実施例A3-1~A3-3と同様にして、色素吸着多孔質チタニア電極を作製し、光化学電池を作製した。
得られた光化学電池を60℃暗所で所定の時間静置した後、室温に戻し、光電変換効率(η)を英弘精機株式会社製のソーラーシュミレーターを用い、100mW/cm2の擬似太陽光を照射し測定した。各々の錯体色素の60℃暗所放置1日後の光電変換効率を100%とした場合の5日後の光電変換効率の維持率を表2に示す。
Claims (25)
- 一般式(1)
で示される基、又は式(2-2)
で示される基、又は式(2-3)
で示される基のいずれかを示す。Xは対イオンを示し、mは錯体の電荷を中和するのに必要な対イオンの数を示す。但し、2つの
で示される二核ルテニウム錯体色素。 - 式(2-3)中のArが、置換基を有していても良いフェニル基を示すことを特徴とする請求項6~10のいずれかに記載の二核ルテニウム錯体色素。
- 請求項1~16のいずれかに記載の二核ルテニウム錯体色素と半導体微粒子を含むことを特徴とする光電変換素子。
- 前記半導体微粒子が、酸化チタン、酸化亜鉛及び酸化スズからなる群より選ばれる少なくとも1種の半導体微粒子であることを特徴とする請求項17記載の光電変換素子。
- 請求項17~18のいずれかに記載の光電変換素子を備えることを特徴とする光化学電池。
- 電極として請求項17~18のいずれかに記載の光電変換素子と対極とを有し、その間に電解質層を有することを特徴とする光化学電池。
- 請求項1~16のいずれかに記載の二核ルテニウム錯体色素を含む溶液に半導体微粒子を浸漬する工程を有することを特徴とする光電変換素子の製造方法。
- 導電性支持体上に、半導体微粒子を含む半導体層を形成する工程と、
この半導体層を請求項1~16のいずれかに記載の二核ルテニウム錯体色素を含む溶液に浸漬する工程と
を有することを特徴とする光電変換素子の製造方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/635,226 US8633380B2 (en) | 2010-03-16 | 2011-03-15 | Photoelectric conversion element comprising binuclear ruthenium complex dye having a substituted bipyridyl group, and photochemical cell |
| JP2012505709A JP5780238B2 (ja) | 2010-03-16 | 2011-03-15 | 置換ビピリジル基を有する二核ルテニウム錯体色素を有する光電変換素子、及び光化学電池 |
| KR1020127026588A KR20130028912A (ko) | 2010-03-16 | 2011-03-15 | 치환 바이피리딜기를 갖는 이핵 루테늄 착물 색소를 포함하는 광전 변환 소자, 및 광화학 전지 |
| AU2011228146A AU2011228146A1 (en) | 2010-03-16 | 2011-03-15 | Photoelectric conversion element that contains dye consisting of binuclear ruthenium complex having substituted bipyridyl groups, and photochemical cell |
| EP11756322A EP2548926A1 (en) | 2010-03-16 | 2011-03-15 | Photoelectric conversion element that contains dye consisting of binuclear ruthenium complex having substituted bipyridyl groups, and photochemical cell |
| CN201180022339.6A CN102884137B (zh) | 2010-03-16 | 2011-03-15 | 包含具有经取代的联吡啶基团的双核钌络合物染料的光电转换元件和光化学电池 |
Applications Claiming Priority (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010059487 | 2010-03-16 | ||
| JP2010-059487 | 2010-03-16 | ||
| JP2010-074553 | 2010-03-29 | ||
| JP2010074552 | 2010-03-29 | ||
| JP2010074553 | 2010-03-29 | ||
| JP2010-074552 | 2010-03-29 | ||
| JP2010-232561 | 2010-10-15 | ||
| JP2010232561 | 2010-10-15 | ||
| JP2010266254 | 2010-11-30 | ||
| JP2010-266254 | 2010-11-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011115137A1 true WO2011115137A1 (ja) | 2011-09-22 |
Family
ID=44649221
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/056110 WO2011115137A1 (ja) | 2010-03-16 | 2011-03-15 | 置換ビピリジル基を有する二核ルテニウム錯体色素を有する光電変換素子、及び光化学電池 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US8633380B2 (ja) |
| EP (1) | EP2548926A1 (ja) |
| JP (1) | JP5780238B2 (ja) |
| KR (1) | KR20130028912A (ja) |
| CN (1) | CN102884137B (ja) |
| AU (1) | AU2011228146A1 (ja) |
| TW (1) | TWI526503B (ja) |
| WO (1) | WO2011115137A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013089194A1 (ja) * | 2011-12-13 | 2013-06-20 | 宇部興産株式会社 | 二核ルテニウム錯体色素、当該色素を有する光電変換素子及び光化学電池 |
| JP2013144721A (ja) * | 2011-12-13 | 2013-07-25 | Ube Industries Ltd | 二核ルテニウム錯体色素、当該色素を有する光電変換素子及び光化学電池 |
| US20130324733A1 (en) * | 2010-12-02 | 2013-12-05 | Ube Industries, Ltd. | Binuclear metal complex, and organic electroluminescence element comprising same |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI520458B (zh) * | 2014-04-03 | 2016-02-01 | 財團法人工業技術研究院 | 靜電放電箝制電路與靜電放電箝制方法 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01220380A (ja) | 1988-02-12 | 1989-09-04 | Gebr Sulzer Ag | 光電気化学電池・その製法及び使用法 |
| JP2003261536A (ja) | 2001-11-27 | 2003-09-19 | Ube Ind Ltd | 金属錯体、金属錯体色素、光電変換素子、及び光化学電池 |
| JP2004359677A (ja) | 2003-05-13 | 2004-12-24 | Asahi Kasei Corp | 複核錯体 |
| WO2006038587A1 (ja) | 2004-10-01 | 2006-04-13 | Ube Industries, Ltd. | 二核金属錯体、金属錯体色素、光電変換素子、及び光化学電池 |
| JP2007302879A (ja) * | 2006-05-11 | 2007-11-22 | National Central Univ | 光増感色素 |
| JP2009080988A (ja) * | 2007-09-25 | 2009-04-16 | Panasonic Electric Works Co Ltd | 光電変換素子 |
| WO2009154275A1 (ja) * | 2008-06-19 | 2009-12-23 | 宇部興産株式会社 | 二核ルテニウム錯体色素、ルテニウム-オスミウム錯体色素、当該錯体色素を有する光電変換素子、及び光化学電池 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11144773A (ja) * | 1997-09-05 | 1999-05-28 | Fuji Photo Film Co Ltd | 光電変換素子および光再生型光電気化学電池 |
| EP0991092B1 (en) * | 1998-09-30 | 2008-07-23 | FUJIFILM Corporation | Semiconductor particle sensitized with methine dye |
| US6291763B1 (en) * | 1999-04-06 | 2001-09-18 | Fuji Photo Film Co., Ltd. | Photoelectric conversion device and photo cell |
| DE60027512T2 (de) * | 1999-08-04 | 2006-10-12 | Fuji Photo Film Co., Ltd., Minami-Ashigara | Elektrolytzusammensetzung und photolektrochemische Zelle |
| JP5054269B2 (ja) * | 2000-07-27 | 2012-10-24 | 日本化薬株式会社 | 色素増感光電変換素子 |
| JP5142307B2 (ja) * | 2000-11-28 | 2013-02-13 | 独立行政法人産業技術総合研究所 | 有機色素を光増感剤とする半導体薄膜電極、光電変換素子 |
| TW541330B (en) * | 2001-03-07 | 2003-07-11 | Nippon Kayaku Kk | Photo-electric conversion device and oxide semiconductor fine particle |
| WO2003005481A1 (fr) * | 2001-07-06 | 2003-01-16 | Nippon Kayaku Kabushiki Kaisha | Element de conversion photoelectrique sensibilise par une matiere colorante |
| US8227690B2 (en) * | 2003-03-14 | 2012-07-24 | Nippon Kayaku Kabushiki Kaisha | Dye-sensitized photoelectric conversion device |
| US7741559B2 (en) | 2003-05-13 | 2010-06-22 | Asahi Kasei Kabushiki Kaisha | Photoelectric conversion element |
| JP5003865B2 (ja) * | 2006-03-17 | 2012-08-15 | 宇部興産株式会社 | 二核金属錯体色素溶液およびこの溶液を用いた光電変換素子、及び光化学電池 |
| WO2009102068A1 (ja) * | 2008-02-14 | 2009-08-20 | Ube Industries, Ltd. | 二核ルテニウム錯体色素溶液、及び当該錯体色素溶液を用いて得られる光増感された半導体微粒子を用いた光電変換素子、並びにそれを用いた光化学電池 |
-
2011
- 2011-03-15 KR KR1020127026588A patent/KR20130028912A/ko not_active Application Discontinuation
- 2011-03-15 EP EP11756322A patent/EP2548926A1/en not_active Withdrawn
- 2011-03-15 WO PCT/JP2011/056110 patent/WO2011115137A1/ja active Application Filing
- 2011-03-15 US US13/635,226 patent/US8633380B2/en not_active Expired - Fee Related
- 2011-03-15 CN CN201180022339.6A patent/CN102884137B/zh not_active Expired - Fee Related
- 2011-03-15 AU AU2011228146A patent/AU2011228146A1/en not_active Abandoned
- 2011-03-15 JP JP2012505709A patent/JP5780238B2/ja not_active Expired - Fee Related
- 2011-03-16 TW TW100108951A patent/TWI526503B/zh not_active IP Right Cessation
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01220380A (ja) | 1988-02-12 | 1989-09-04 | Gebr Sulzer Ag | 光電気化学電池・その製法及び使用法 |
| JP2003261536A (ja) | 2001-11-27 | 2003-09-19 | Ube Ind Ltd | 金属錯体、金属錯体色素、光電変換素子、及び光化学電池 |
| JP2004359677A (ja) | 2003-05-13 | 2004-12-24 | Asahi Kasei Corp | 複核錯体 |
| WO2006038587A1 (ja) | 2004-10-01 | 2006-04-13 | Ube Industries, Ltd. | 二核金属錯体、金属錯体色素、光電変換素子、及び光化学電池 |
| JP2007302879A (ja) * | 2006-05-11 | 2007-11-22 | National Central Univ | 光増感色素 |
| JP2009080988A (ja) * | 2007-09-25 | 2009-04-16 | Panasonic Electric Works Co Ltd | 光電変換素子 |
| WO2009154275A1 (ja) * | 2008-06-19 | 2009-12-23 | 宇部興産株式会社 | 二核ルテニウム錯体色素、ルテニウム-オスミウム錯体色素、当該錯体色素を有する光電変換素子、及び光化学電池 |
Non-Patent Citations (4)
| Title |
|---|
| "Current Technology in Dye-sensitized Solar Battery", 25 May 2001, CMC CO., LTD., pages: 117 |
| JOURNAL OF AMERICAN CHEMICAL SOCIETY, vol. 128, 2006, pages 4146 - 4154 |
| KUANG, DAIBIN ET AL.: "High Molar Extinction Coefficient Heteroleptic Ruthenium Complexes for Thin Film Dye-Sensitized Solar Cells", JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, vol. 128, no. 12, 2006, pages 4146 - 4154, XP008166974 * |
| NATURE, vol. 353, 1991, pages 737 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130324733A1 (en) * | 2010-12-02 | 2013-12-05 | Ube Industries, Ltd. | Binuclear metal complex, and organic electroluminescence element comprising same |
| US9029541B2 (en) * | 2010-12-02 | 2015-05-12 | Ube Industries, Ltd. | Binuclear metal complex, and organic electroluminescence element comprising same |
| WO2013089194A1 (ja) * | 2011-12-13 | 2013-06-20 | 宇部興産株式会社 | 二核ルテニウム錯体色素、当該色素を有する光電変換素子及び光化学電池 |
| JP2013144721A (ja) * | 2011-12-13 | 2013-07-25 | Ube Industries Ltd | 二核ルテニウム錯体色素、当該色素を有する光電変換素子及び光化学電池 |
| JPWO2013089194A1 (ja) * | 2011-12-13 | 2015-04-27 | 宇部興産株式会社 | 二核ルテニウム錯体色素、当該色素を有する光電変換素子及び光化学電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2011115137A1 (ja) | 2013-06-27 |
| CN102884137A (zh) | 2013-01-16 |
| AU2011228146A1 (en) | 2012-10-25 |
| KR20130028912A (ko) | 2013-03-20 |
| TW201200565A (en) | 2012-01-01 |
| JP5780238B2 (ja) | 2015-09-16 |
| EP2548926A1 (en) | 2013-01-23 |
| US20130014824A1 (en) | 2013-01-17 |
| TWI526503B (zh) | 2016-03-21 |
| US8633380B2 (en) | 2014-01-21 |
| CN102884137B (zh) | 2015-07-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5633370B2 (ja) | 二核ルテニウム錯体色素、ルテニウム−オスミウム錯体色素、当該錯体色素を有する光電変換素子、及び光化学電池 | |
| KR20110095306A (ko) | 새로운 리간드 및 감응제를 포함하는 광전 변환 소자 | |
| JPWO2006038587A1 (ja) | 二核金属錯体、金属錯体色素、光電変換素子、及び光化学電池 | |
| Dragonetti et al. | A new thiocyanate-free cyclometallated ruthenium complex for dye-sensitized solar cells: Beneficial effects of substitution on the cyclometallated ligand | |
| JP5761024B2 (ja) | 置換ビピリジル基を有する二核ルテニウム錯体色素を有する光電変換素子、及び光化学電池 | |
| JP5780238B2 (ja) | 置換ビピリジル基を有する二核ルテニウム錯体色素を有する光電変換素子、及び光化学電池 | |
| JP5170357B2 (ja) | 光電変換素子、及び光化学電池 | |
| JP5428312B2 (ja) | 光電変換素子、及び光化学電池 | |
| Konti et al. | A Ru (II) molecular antenna bearing a novel bipyridine–acrylonitrile ligand: Synthesis and application in dye solar cells | |
| JP6086069B2 (ja) | 二核ルテニウム錯体色素、当該色素を有する光電変換素子及び光化学電池 | |
| WO2009102068A1 (ja) | 二核ルテニウム錯体色素溶液、及び当該錯体色素溶液を用いて得られる光増感された半導体微粒子を用いた光電変換素子、並びにそれを用いた光化学電池 | |
| JP5424246B2 (ja) | 新規光増感剤及び光起電力素子 | |
| JP5493857B2 (ja) | 二核ルテニウム錯体色素、二核ルテニウム錯体色素酸性水溶液及びその製造方法 | |
| JP5838820B2 (ja) | 二核ルテニウム錯体色素、当該色素を有する光電変換素子及び光化学電池 | |
| JP5556096B2 (ja) | 電子吸引基を置換基として持つ連結分子を有する二核ルテニウム錯体色素を有する光電変換素子、及び光化学電池 | |
| JP5573056B2 (ja) | 二核ルテニウム錯体色素により増感された半導体微粒子と、アリールアミン化合物を含有する電解質溶液とを備える光化学電池 | |
| JP2009129652A (ja) | 光電変換素子、及び光化学電池 | |
| JP2009067838A (ja) | 二核ルテニウム錯体色素及びその製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180022339.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11756322 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012505709 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13635226 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011756322 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20127026588 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2011228146 Country of ref document: AU Date of ref document: 20110315 Kind code of ref document: A |