WO2009100925A1 - Neue materialien für organische elektrolumineszenzvorrichtungen - Google Patents
Neue materialien für organische elektrolumineszenzvorrichtungen Download PDFInfo
- Publication number
- WO2009100925A1 WO2009100925A1 PCT/EP2009/001028 EP2009001028W WO2009100925A1 WO 2009100925 A1 WO2009100925 A1 WO 2009100925A1 EP 2009001028 W EP2009001028 W EP 2009001028W WO 2009100925 A1 WO2009100925 A1 WO 2009100925A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- aromatic
- substituted
- group
- formula
- organic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- UEWHMHUEUVHQQH-UHFFFAOYSA-N OC(c1cc2ccccc2c2c1ccc(Br)c2)=O Chemical compound OC(c1cc2ccccc2c2c1ccc(Br)c2)=O UEWHMHUEUVHQQH-UHFFFAOYSA-N 0.000 description 2
- XMLGWPXWLHFCSN-GXDHUFHOSA-N C=[Br]c(cc1)ccc1/C(/C(O)=O)=C\c1ccccc1[N+]([O-])=O Chemical compound C=[Br]c(cc1)ccc1/C(/C(O)=O)=C\c1ccccc1[N+]([O-])=O XMLGWPXWLHFCSN-GXDHUFHOSA-N 0.000 description 1
- MPYNJELFEMITNE-NTEUORMPSA-N CC(CC=C1)C(/C=C(/C(O)=O)\c(cc2)ccc2Br)=C1N Chemical compound CC(CC=C1)C(/C=C(/C(O)=O)\c(cc2)ccc2Br)=C1N MPYNJELFEMITNE-NTEUORMPSA-N 0.000 description 1
- HKYGDHLRGZLKNA-UHFFFAOYSA-N CC1C(c(c2ccccc22)c(cccc3)c3c2S(O)O)=CC=C2C=CC=CC12 Chemical compound CC1C(c(c2ccccc22)c(cccc3)c3c2S(O)O)=CC=C2C=CC=CC12 HKYGDHLRGZLKNA-UHFFFAOYSA-N 0.000 description 1
- ADQWWONHHPCNFU-UHFFFAOYSA-N OC(c1cc2ccccc2c2c1ccc(-c1c(cccc3)c3c(Cc3cc(cccc4)c4cc3)c3ccccc13)c2)=O Chemical compound OC(c1cc2ccccc2c2c1ccc(-c1c(cccc3)c3c(Cc3cc(cccc4)c4cc3)c3ccccc13)c2)=O ADQWWONHHPCNFU-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/20—Polycyclic condensed hydrocarbons
- C07C15/27—Polycyclic condensed hydrocarbons containing three rings
- C07C15/30—Phenanthrenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C1/00—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon
- C07C1/20—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon starting from organic compounds containing only oxygen atoms as heteroatoms
- C07C1/207—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon starting from organic compounds containing only oxygen atoms as heteroatoms from carbonyl compounds
- C07C1/2078—Preparation of hydrocarbons from one or more compounds, none of them being a hydrocarbon starting from organic compounds containing only oxygen atoms as heteroatoms from carbonyl compounds by a transformation in which at least one -C(=O)-O- moiety is eliminated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/54—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings
- C07C13/547—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings at least one ring not being six-membered, the other rings being at the most six-membered
- C07C13/567—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings at least one ring not being six-membered, the other rings being at the most six-membered with a fluorene or hydrogenated fluorene ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/72—Spiro hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/20—Polycyclic condensed hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/20—Polycyclic condensed hydrocarbons
- C07C15/38—Polycyclic condensed hydrocarbons containing four rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/54—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to two or three six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/57—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton
- C07C211/61—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton with at least one of the condensed ring systems formed by three or more rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D221/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00
- C07D221/02—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00 condensed with carbocyclic rings or ring systems
- C07D221/04—Ortho- or peri-condensed ring systems
- C07D221/06—Ring systems of three rings
- C07D221/10—Aza-phenanthrenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D235/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings
- C07D235/02—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings condensed with carbocyclic rings or ring systems
- C07D235/04—Benzimidazoles; Hydrogenated benzimidazoles
- C07D235/18—Benzimidazoles; Hydrogenated benzimidazoles with aryl radicals directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D251/00—Heterocyclic compounds containing 1,3,5-triazine rings
- C07D251/02—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings
- C07D251/12—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D251/14—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom
- C07D251/24—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom to three ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/08—Hydrogen atoms or radicals containing only hydrogen and carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/72—Benzo[c]thiophenes; Hydrogenated benzo[c]thiophenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
- H10K50/12—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers comprising dopants
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/16—Electron transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/18—Carrier blocking layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/14—The ring being saturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/06—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members
- C07C2603/10—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings
- C07C2603/12—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings only one five-membered ring
- C07C2603/18—Fluorenes; Hydrogenated fluorenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/22—Ortho- or ortho- and peri-condensed systems containing three rings containing only six-membered rings
- C07C2603/24—Anthracenes; Hydrogenated anthracenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/22—Ortho- or ortho- and peri-condensed systems containing three rings containing only six-membered rings
- C07C2603/26—Phenanthrenes; Hydrogenated phenanthrenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/40—Ortho- or ortho- and peri-condensed systems containing four condensed rings
- C07C2603/42—Ortho- or ortho- and peri-condensed systems containing four condensed rings containing only six-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/52—Ortho- or ortho- and peri-condensed systems containing five condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/93—Spiro compounds
- C07C2603/94—Spiro compounds containing "free" spiro atoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1014—Carbocyclic compounds bridged by heteroatoms, e.g. N, P, Si or B
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1059—Heterocyclic compounds characterised by ligands containing three nitrogen atoms as heteroatoms
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/321—Metal complexes comprising a group IIIA element, e.g. Tris (8-hydroxyquinoline) gallium [Gaq3]
- H10K85/324—Metal complexes comprising a group IIIA element, e.g. Tris (8-hydroxyquinoline) gallium [Gaq3] comprising aluminium, e.g. Alq3
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S428/00—Stock material or miscellaneous articles
- Y10S428/917—Electroluminescent
Definitions
- the present invention relates to organic semiconductors and their use in organic electronic devices.
- Organic semiconductors are being developed for a variety of electronic applications.
- OLEDs organic electroluminescent devices
- the structure of organic electroluminescent devices (OLEDs) in which these organic semiconductors are used as functional materials is described, for example, in US Pat. No. 4,539,507, US Pat. No. 5,151,629, EP 0676461 and WO 98/27136.
- further improvements are needed before these devices can be used for high quality and durable displays.
- the lifetime and the efficiency of deep blue-emitting organic electroluminescent devices are currently still a problem in which there is still room for improvement.
- the compounds have a high thermal stability and a high glass transition temperature and can sublime undecomposed.
- a high glass transition temperature is essential for achieving high lifetimes.
- condensed aromatics in particular anthracene derivatives
- host materials for blue-emitting electroluminescent devices, eg. B. 9,10-bis (2-naphthyl) anthracene (US 5935721).
- WO 03/095445 and CN 1362464 disclose 9,10-bis (1-naphthyl) anthracene derivatives for use in OLEDs.
- Further anthracene derivatives are disclosed in WO 01/076323, in WO 01/021729, in WO 04/013073, in WO 04/018588, in WO 03/087023 or in WO 04/018587.
- Host materials based on aryl-substituted pyrenes and chrysenes are disclosed in WO 04/016575. It is necessary for high quality applications to have improved host materials available.
- anthracene derivatives which are substituted in the 9-position with a 3-phenanthrenyl group, wherein both the anthracene and the phenanthrene can be further substituted, here show significant improvements and are very suitable for use in organic electroluminescent devices.
- these compounds it is possible to increase the efficiency and, above all, the lifetime of the organic electronic device in comparison with materials according to the prior art. This is especially true for deep blue fluorescent devices.
- these compounds have a high thermal stability. In general, these materials are very well suited for use in organic electronic devices since they have a high glass transition temperature. These materials and their use in organic electronic devices are therefore the subject of the present invention.
- organic electroluminescent devices which contain phenanthrene derivatives, wherein the phenanthrene in addition to other substituents also by aromatic
- JP 2001/332384 does not teach that just one anthracene, to which in the 9-position one 3-phenanthrenyl group, particularly good results achieved in organic electroluminescent devices.
- WO 07/123256 generally claims fluoranthene-9-anthracene compounds for use in organic electroluminescent devices.
- the anthracene in the 10-position may also be substituted by further aromatic groups.
- two structures are also listed which contain a 3-phenathrenyl group on the anthracene in the 10-position.
- the inventive effect of these compounds is attributed to the combination of the fluoranthene moiety with the 9-anthracene moiety.
- the presence of phenanthrenyl is not attributed importance.
- the invention provides compounds of the formula (1)
- Ar is H 1 D or an aromatic or heteroaromatic ring system having from 5 to 60 aromatic ring atoms which may be substituted with one or more non-aromatic radicals R 1 ;
- R 1 is the same or different at each occurrence H, D, F, Cl 1 Br, I 1
- R2 is the same or different at each occurrence of R1 or two or more adjacent substituents R2 together form a mono- or polycyclic, aliphatic or aromatic ring system;
- the compounds according to formula (1) has a glass transition temperature T 9 of greater than 70 0 C, more preferably greater than 100 0 C, most preferably greater than 120 0 C.
- Ar is not a fluoranthene group or does not contain a fluoranthene group which is bonded directly to the anthracene.
- An aryl group in the sense of this invention contains 6 to 60 C atoms;
- a heteroaryl group contains 2 to 60 C atoms and at least 1 heteroatom, with the proviso that the sum of C atoms and heteroatoms gives at least 5.
- the heteroatoms are preferably selected from N, O and / or S.
- aryl group or heteroaryl group either a simple aromatic cycle, ie benzene, or a simple heteroaromatic cycle, for example pyridine, pyrimidine, thiophene, etc., or a fused aryl or heteroaryl group, for example naphthalene, anthracene, pyrene, quinoline, isoquinoline, benzimidazole , Phenanthrene, etc., understood.
- An aromatic ring system in the sense of this invention contains 6 to 60 carbon atoms in the ring system.
- a heteroaromatic ring system in the sense of this invention contains 2 to 60 C atoms and at least one heteroatom in the ring system, with the proviso that the sum of C atoms and heteroatoms gives at least 5.
- the heteroatoms are preferably selected from N, O and / or S.
- An aromatic or heteroaromatic ring system in the sense of this invention is to be understood as meaning a system which does not necessarily contain only aryl or heteroaryl groups but in which also several aryl or heteroaromatic ring systems Heteroaryl groups by a short, non-aromatic unit (preferably less than 10% of the atoms other than H), such as.
- N or O atom may be interrupted.
- systems such as 9,9'-spirobifluorene, 9,9-diarylfluorene, cis- and trans-indenofluorene, benzindene fluorene, dibenzoindenofluorene, triarylamine, diaryl ethers, stilbene, benzophenone, etc. are also to be understood as aromatic ring systems in the context of this invention.
- aromatic or heteroaromatic ring system is understood as meaning systems in which a plurality of aryl or heteroaryl groups are linked together by single bonds, for example biphenyl, terphenyl or bipyridine.
- a C 1 to C 40 alkyl group in which individual H atoms or CH 2 groups can also be substituted by the abovementioned groups, particularly preferably the radicals methyl, ethyl, n-propyl, i-propyl , n-butyl, i-butyl, s-butyl, t-butyl, 2-methylbutyl, n-pentyl, s-pentyl, cyclopentyl, n-hexyl, cyclohexyl, n-heptyl, cycloheptyl, n-octyl, cyclooctyl, 2 Ethylhexyl, trifluoromethyl, pentafluoroethyl, 2,2,2-trifluoroethyl, ethenyl, propenyl, butenyl, pentenyl, cyclopentenyl, hexenyl
- a C 1 to C 40 alkoxy group is more preferably understood as meaning methoxy, trifluoromethoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, s-butoxy, t-butoxy or 2-methylbutoxy.
- aromatic or heteroaromatic ring system having 5-60 aromatic ring atoms, which may each be substituted in each case with the abovementioned radicals R and which may be linked via any desired positions on the aromatic or heteroaromatic compounds, are understood in particular groups which are derived from Benzene, naphthalene, anthracene, phenanthrene, pyrene, chrysene, perylene, fluoranthene, naphthacene, pentacene, benzanthracene, benzpyrene,
- Phenanthroline 1, 2,3-triazole, 1, 2,4-triazole, benzotriazole, 1, 2,3-oxadiazole, 1,2,4-oxadiazole, 1, 2,5-oxadiazole, 1,3,4- Oxadiazole, 1, 2,3-thiadiazole, 1,2,4-thiadiazole, 1, 2,5-thiadiazole, 1, 3,4-thiadiazole, 1, 3,5-triazine, 1, 2,4-triazine, 1, 2,3-triazine. Tetrazole, 1,2,4,5-tetrazine, 1, 2,3,4-tetrazine, 1, 2,3,5-tetrazine, purine, pteridine, indolizine and benzothiadiazole.
- the anthracene unit of the formula (1) at most one group C-R2 is replaced by N. Most preferably, no group C-R2 is replaced by N, that is, it is an anthracene and not a heterocycle.
- no group is in the phenanthrene unit Replaced C-R1 by N, that is, it is a phenanthrene and not a heterocycle.
- Preferred embodiments of the present invention are compounds according to formula (2) to formula (7),
- the group Ar in formula (5) preferably represents H or D.
- the group Ar preferably represents an aromatic or heteroaromatic ring system having 5 to 30 aromatic ring atoms which may be substituted with one or more non-aromatic radicals R1.
- the group Ar is particularly preferably composed of the aromatic or heteroaromatic groups benzene, naphthalene, anthracene, carbazole, phenanthrene, benzanthracene, chrysene, pyrene, phenanthroline, triazine and benzimidazole.
- Very particularly preferred groups Ar are selected from phenyl, ortho, meta- or para-biphenyl, 1- or 2-naphthyl, 2-, 3- or 9-phenanthrenyl, para-phenylene-1-naphthyl, para-phenylene-2 naphthyl, 2-fluorenyl or 2-spirobifluorenyl, each of which may be substituted by one or more nonaromatic radicals R1, or the group Ar is selected from the following formulas (8), (9), (10) or (11),
- R1 has the meaning given above and furthermore:
- Ar 2 is an aryl or heteroaryl group having from 5 to 16 aromatic
- Ring atoms preferably phenyl, 1-naphthyl, 2-naphthyl, 9-anthryl, chrysenyl, 1-pyrenyl, 2-pyrenyl, 2-phenanthrenyl, 3-phenanthrene, 9-phenanthrenyl, 2-benzimidazole or fluoro-phenyl, which are each denoted by one or more R1 may be substituted;
- q is 0, 1, 2 or 3, preferably 1 or 2, more preferably 1.
- the radicals R 2 in compounds of the formula (1) form an aromatic ring system.
- H atoms may be replaced by F, or an aromatic or heteroaromatic ring system having from 5 to 30 aromatic ring atoms, each of which may be substituted by one or more non-aromatic R1 groups, or a combination of these systems.
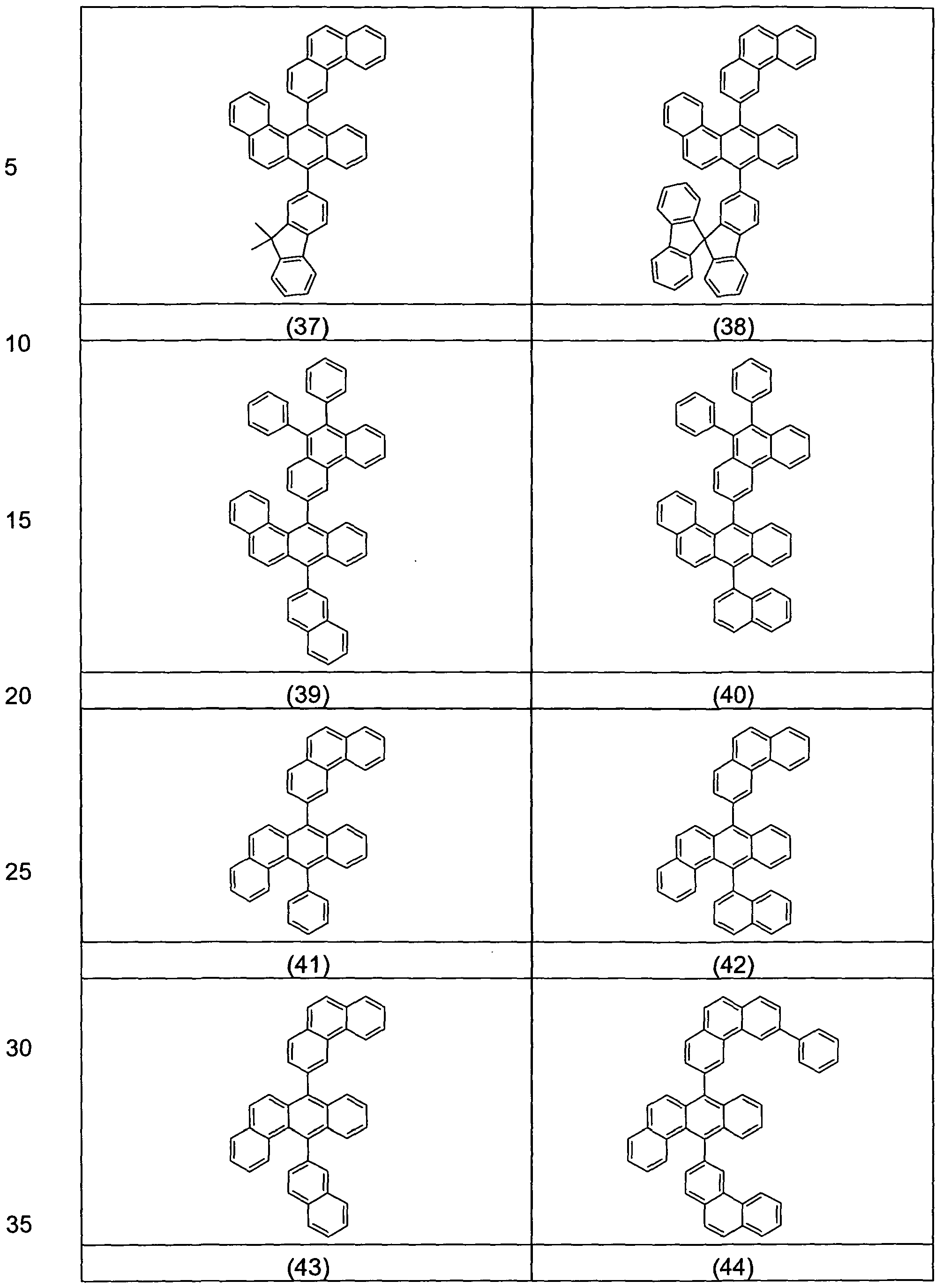
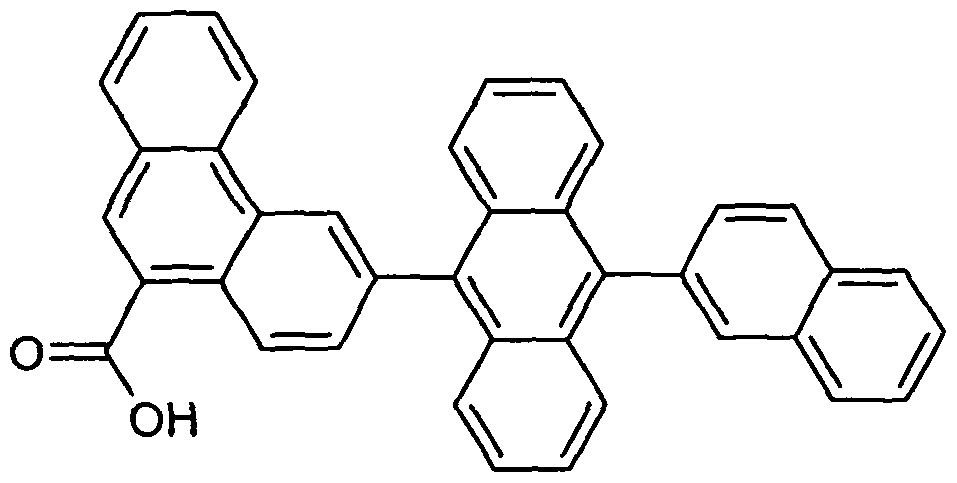
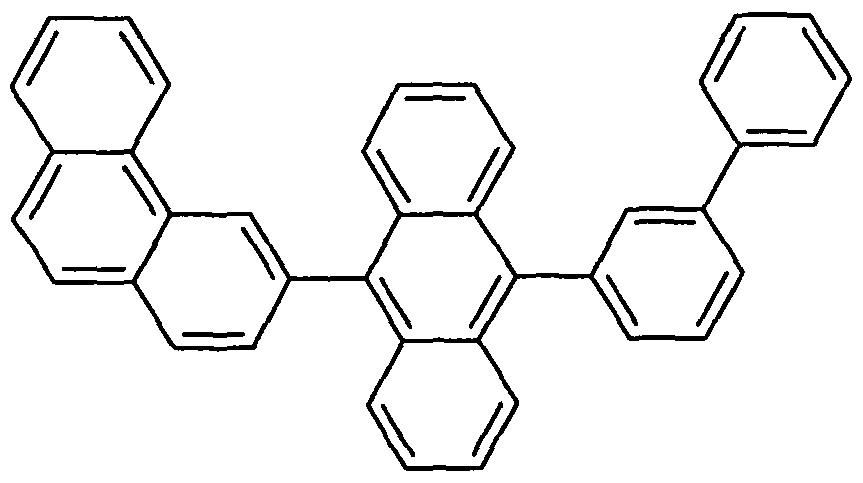
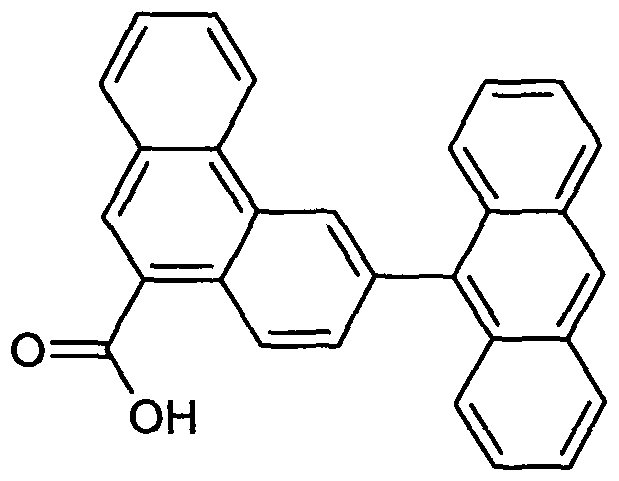
- Examples of preferred compounds according to the formula (1) or according to the formulas (2) to (7) are the structures (1) to (122) depicted below.
- the compounds of the formula (1) according to the invention can be prepared by synthesis steps generally known to the person skilled in the art.
- a starting compound z For example, the corresponding 3-bromophenanthrenes and 9-bromoanthracenes or 9,10-dibromoanthracenes or 7- or 12-bromobenz [a] anthracene or 7,12-dibromobenz [a] anthracene.
- the compounds substituted with appropriate leaving groups such as chlorine, iodine, triflate or tosylate can serve as starting compounds which are then optionally converted into the corresponding boronic acid derivatives. Suzuki coupling of the compounds may take place under standard conditions known to those skilled in the art of organic chemistry.
- 3-Bromo-substituted phenanthrenes can be prepared by a modified Pschorr phenanthrene synthesis, as shown in Scheme 1.
- this consists of a stereospecific Perkin condensation of ortho-nitrobenzaldehyde and para-bromophenylacetic acid (Scheme 1a).
- the nitro group of the nitrocinnamic acid derivative obtained in the first step is reduced to the amino group, for example by iron (II) (Scheme 1 b).
- anthracene component is shown in Scheme 2 using the example of 2-naphthylanthracene.
- 2-naphthyl group another aryl group may be selected and / or the aryl group may be substituted.
- the unsubstituted anthracene a substituted anthracene or a substituted or unsubstituted benzanthracene or tetracene.
- the aromatic group Ar here a 2-naphthyl group, is introduced as a boronic acid derivative in a Suzuki coupling. This may be done under standard conditions as known to those skilled in the art of organic synthesis.
- the 9-arylanthracene derivative thus obtained is functionalized in the 10-position, for example brominated, the bromination being carried out under standard conditions, as known to those skilled in the art, for example with elemental bromine in acetic acid or chloroform or NBS in THF.
- the bromoanthracene can be converted to the boronic acid, for example by lithiation and reaction with B (O 1 Pr) 3 .
- Another object of the present invention is a process for the preparation of compounds of formula (1) by coupling a 9-aryl-substituted anthracene, which in position 10 by a reactive leaving group, in particular chlorine, bromine, iodine, triflate, tosylate, boronic acid or boronic acid ester, or a corresponding benz [a] anthracene or tetracene with a phenanthrene derivative which is functionalized in the 3-position, in particular by chlorine, bromine, iodine, triflate, tosylate, boronic acid or boronic acid ester.
- a reactive leaving group in particular chlorine, bromine, iodine, triflate, tosylate, boronic acid or boronic acid ester
- both the phenanthrene and also the anthracene, benzanthracene or tetracene may be substituted by the abovementioned substituents R 1 or R 2.
- the one component contains bromine as a functional leaving group and the other component contains a boronic acid or a boronic ester as a functional leaving group.
- Suitable coupling reactions are, in particular, transition metal-catalyzed coupling reactions, in particular the Suzuki coupling under palladium catalysis.
- the compounds of the formula (1) are suitable for use in electronic devices, in particular in organic electroluminescent devices (OLEDs, PLEDs). Depending on the substitution, the compounds are used in different functions and layers.
- Another object of the invention is therefore the use of compounds according to formula (1) in electronic devices, in particular in organic electroluminescent devices.
- organic electronic devices containing at least one compound according to formula (1), in particular organic electroluminescent devices containing anode, cathode and at least one emitting layer, characterized in that at least one organic layer containing an emitting layer or another layer, at least one compound according to formula (1) contains.
- the organic electronic device can also have inorganic layers.
- the organic electroluminescent device may contain further layers. These are selected, for example, from one or more hole injection layers, hole transport layers, electron transport layers, electron injection layers and / or charge generation layers
- the organic electroluminescent device contains a plurality of emitting layers, wherein at least one organic layer contains at least one compound according to formula (1).
- these emission layers have a total of several emission maxima between 380 nm and 750 nm, so that a total of white emission results, ie in the emitting layers are different emitting Used compounds that can fluoresce or phosphoresce and emit the blue and yellow, orange or red light.
- the compound according to the formula (1) is preferably used in a blue-emitting layer.
- Particularly preferred are three-layer systems, ie systems with three emitting layers, wherein at least one of these layers contains at least one compound according to formula (1) and wherein the three layers blue, green and orange or red emission show (for the basic structure see, for. WO 05/011013).
- white emission emitters which have broadband emission and thereby show white emission.
- the compounds of the formula (1) or (2) to (7) are used as host material for fluorescent dopants, in particular for blue-fluorescent dopants.
- the group Ar is preferably selected from simple or condensed aryl groups, in particular phenyl, ortho, meta- or para-biphenyl, 1-naphthyl, 2-naphthyl, 2-, 3- or 9-phenanthrenyl or 4- or 5-Benz [a] anthracene, which may each be substituted by one or more radicals R1.
- a host material in a system of host and dopant is understood to mean the component which is present in the system in the higher proportion.
- the host is understood to be that component whose proportion is the highest in the mixture.
- the proportion of the host material according to formula (1) or (2) to (7) in the emitting layer is between 50.0 and 99.9 wt .-%, preferably between 80.0 and 99.5 wt .-%, particularly preferably between 90.0 and 99.0 wt. -%. Accordingly, the proportion of the dopant is between 0.01 and 50.0 wt .-%, preferably between 0.1 and 20.0 wt .-%, more preferably between 0.5 and 15 wt .-%, most preferably between 1.0 and 10.0 wt .-%.
- Preferred dopants are selected from the class of monostyrylamines, the distyrylamines, the tristyrylamines, the tetrastyrylamines, the Styrylphosphi ⁇ e, the styryl ether and the arylamine.
- a monostyrylamine is understood as meaning a compound which contains a substituted or unsubstituted styryl group and at least one, preferably aromatic, amine.
- a distyrylamine is understood as meaning a compound which contains two substituted or unsubstituted styryl groups and at least one, preferably aromatic, amine.
- a tristyrylamine is understood as meaning a compound which contains three substituted or unsubstituted styryl groups and at least one, preferably aromatic, amine.
- a tetrastyrylamine is meant a compound containing four substituted or unsubstituted styryl groups and at least one, preferably aromatic, amine.
- the styryl groups are particularly preferred stilbenes, which may also be further substituted.
- Corresponding phosphines and ethers are defined in analogy to the amines.
- An arylamine or an aromatic amine in the context of this invention is understood as meaning a compound which contains three substituted or unsubstituted aromatic or heteroaromatic ring systems bonded directly to the nitrogen.
- At least one of these aromatic or heteroaromatic ring systems is preferably a fused ring system, preferably having at least 14 aromatic ring atoms.
- Preferred examples thereof are aromatic anthraceneamines, aromatic anthracenediamines, aromatic pyrenamines, aromatic pyrenediamines, aromatic chrysenamines or aromatic chrysenediamines.
- aromatic anthracene amine is meant a compound in which a diarylamino group is bonded directly to an anthracene group, preferably in the 9-position.
- An aromatic anthracenediamine is understood as meaning a compound in which two diarylamino groups are bonded directly to an anthracene group, preferably in the 9,10-position.
- Aromatic pyrenamines, pyrenediamines, chrysenamines and chrysenediamines are defined analogously thereto, the diarylamino groups being preferably attached to the pyrene in the 1-position or in the 1,6-position.
- Further preferred dopants are selected from indenofluorenamines or diamines, for example according to WO 06/122630, benzoindenofluorenamines or diamines, for example according to WO 08/006449, and dibenzoindenofluorenamines or diamines, for example according to WO 07/140847.
- Examples of dopants from the class of styrylamines are substituted or unsubstituted tristylbenzenes. amines or the dopants described in WO 06/000388, WO 06/058737, WO 06/000389, WO 07/065549 and WO 07/115610.
- the compounds of the formula (1) or (2) to (7) are used as electron transport material and / or as hole blocking material.
- the group Ar is an electron-poor heterocycle, for example imidazole, pyrazole, thiazole, benzimidazole, benzothiazole, triazole, oxadiazole , Benzothiadiazole, phenanthroline, etc., or when the group Ar contains at least one of these heterocycles.
- Particularly preferred Ar groups when the compound of the formulas (1) to (7) is to be used as the electron transporting compound, are the groups of the formulas (9), (10) and (11). Furthermore, it may be preferred if the compound is doped with electron donor compounds.
- an electron transport layer is understood as meaning a layer which adjoins an emission layer or a hole blocking layer on one side and an electron injection layer or the cathode on the other side.
- An electron injection layer is understood as meaning a layer which adjoins the cathode on one side and to an emission layer or a hole blocking layer or an electron transport layer on the other side.
- a hole-blocking compound When the compound according to the formulas (1) or (2) to (7) is used as a hole-blocking compound, it is preferably used in a perforated compound.
- blocking layer used in a fluorescent or phosphorescent electroluminescent device.
- a hole blocking layer is understood as meaning a layer which adjoins an emission layer on one side and an electron transport layer on the other side.
- the compounds of the formula (1) or (2) to (7) are used as the emitting material, in particular in combination with a host material.
- one or more substituents R 1 and or R 2, in particular one or more substituents R 2 represent a group of the formula N (ArI) z.
- an organic electroluminescent device characterized in that one or more layers are coated with a sublimation process.
- 10 * 6 "7 mbar the materials in vacuum sublimation at a pressure below 10 -5 mbar, preferably below mbar, particularly preferably less than 10 by vapor deposition.
- an organic electroluminescent device characterized in that one or more layers are coated with the OVPD (Organic Vapor Phase Deposition) method or with the aid of a carrier gas sublimation.
- the materials are applied at a pressure between 10 "5 mbar and 1 bar.
- an organic electroluminescent device characterized in that one or more layers of solution, such. B. by spin coating, or with any printing process, such.
- any printing process such as screen printing, flexographic printing or offset printing, but more preferably LITI (Light Induced Thermal Imaging, thermal transfer printing) or inkjet printing (ink jet printing), are produced.
- LITI Light Induced Thermal Imaging, thermal transfer printing
- inkjet printing ink jet printing
- soluble compounds are needed. High solubility can be achieved by suitable substitution of the compounds.
- processing and application of the substances in the form of a microemulsion are generally known to the person skilled in the art and the person skilled in the art can apply them to the materials according to the invention without inventive step.
- the compounds according to the invention When used in organic electroluminescent devices, the compounds according to the invention have a higher efficiency and a significantly longer service life, as a result of which the organic electroluminescent devices according to the invention are particularly well suited for use in high-quality and long-lived displays. This is especially true when using the materials as host material for blue dopants and / or as electron transport material. Furthermore, the compounds according to the invention have a high thermal stability and a high glass transition temperature and can be sublimated without decomposition.
- organic field-effect transistors O-FETs
- organic thin-film transistors O-TFTs
- organic light-emitting transistors O-LETs
- organic integrated circuits O-ICs
- organic solar cells O-SCs
- organic FeId quench devices O-FQDs
- LECs organic laser diodes
- O-lasers organic photoreceptors.
- Example 1 6- (10-Naphthalen-2-yl-anthracen-9-yl) -phenanthrene a) (E) -2- (4-Bromophenyl) -3- (2-nitrophenyl) -acrylic acid
- reaction mixture is filtered hot over Celite, washed with warm 10% NH 4 OH solution, neutralized with acetic acid, the resulting solid is filtered off with suction, washed with water and MeOH and dried. Yield of yellow, felted crystals: 52 g (72.3%).
- Solution 2 A solution of 93.6 g (551 mmol) of calcium hypophosphite in 770 mL of 80 0 C hot water is added to a solution of sodium carbonate in 550 mL of 80 0 C hot water, the precipitate is filtered off, the filtrate in a second apparatus, on brought an internal temperature of 50 0 C and 440 mg (7 mmol) of copper powder was added. Solution 1 is added dropwise to this suspension and stirred vigorously for 3 h at 50 ° C. The precipitate is filtered off, washed with water and MeOH, dissolved in toluene and filtered through silica gel. Yield: 21.8 g (90.4%) of yellow powder.
- OLEDs takes place according to a method which is generally described in WO 04/058911 and which in individual cases is adapted to the respective circumstances (eg layer thickness variation in order to achieve optimum efficiency or color).
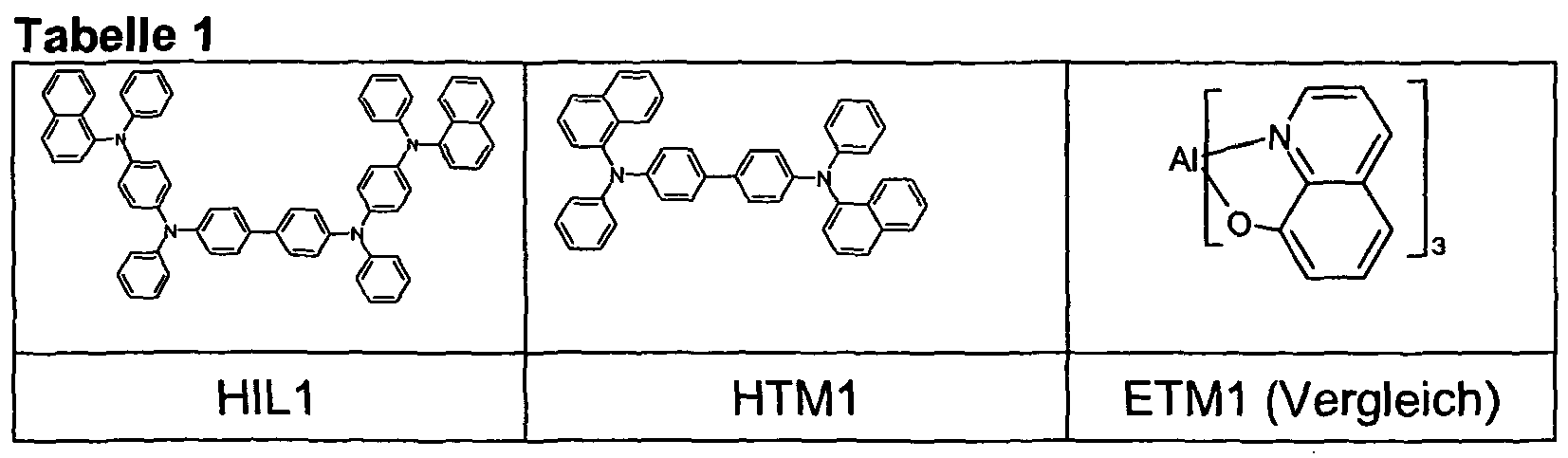
- the OLEDs consist of the following layer sequence: substrate / hole injection layer (HIL1) 60 nm / hole transport layer (HTM1) 60 nm / emission layer (EML) 30 nm / electron transport layer (ETM) 20 nrn and finally a cathode.
- HIL1 substrate / hole injection layer
- HTM1 hole transport layer
- EML emission layer
- ETM electron transport layer
- cathode The materials are thermally evaporated in a vacuum chamber.
- the emission layer always consists of a matrix material (host) and a dopant (dopant), which is mixed by cover evaporation to the host.
- the cathode is formed by a 1 nm thin LiF layer and a 150 nm Al layer deposited thereon.
- Table 1 shows the chemical structures of the materials used to construct the OLEDs.
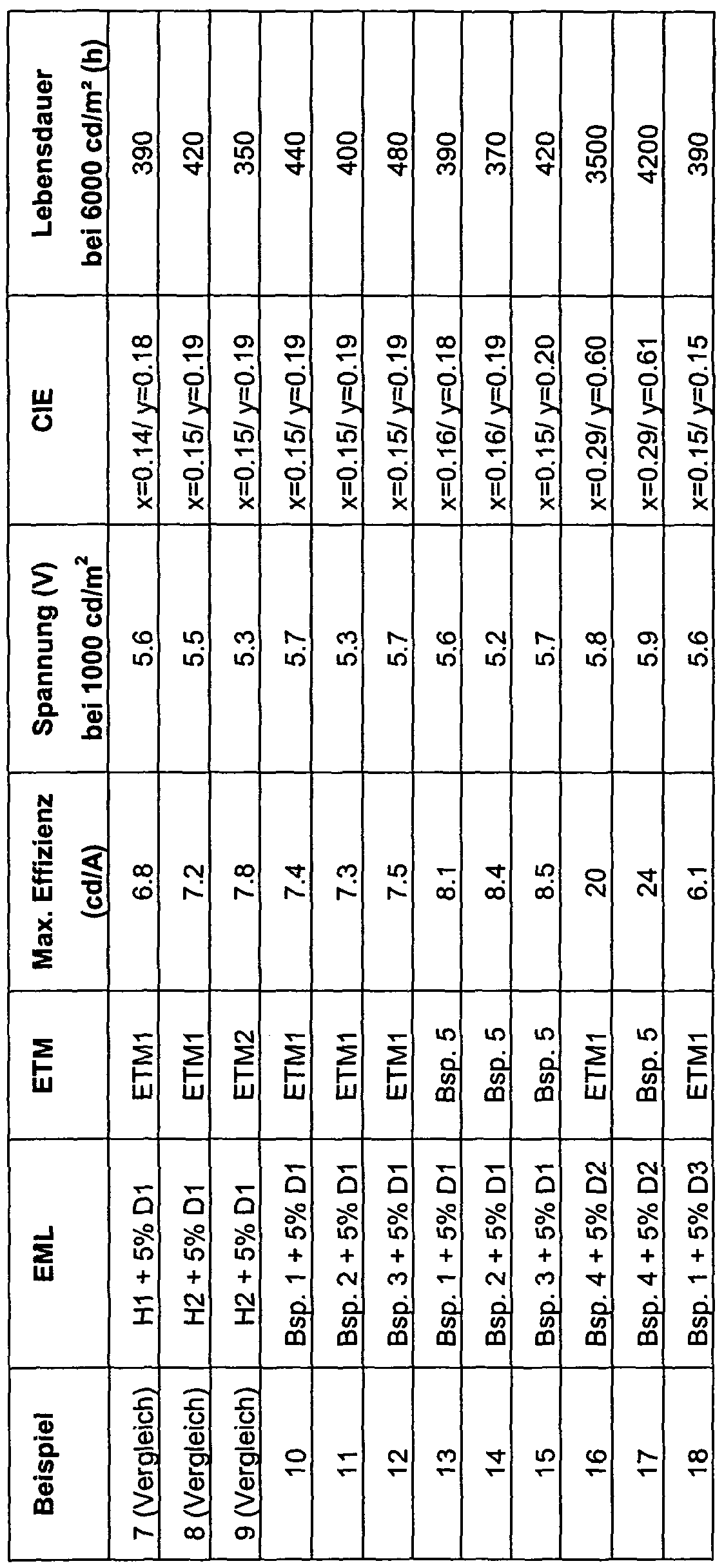
- OLEDs are characterized by default; For this purpose, the electroluminescence spectra, the efficiency (measured in cd / A), the power efficiency (measured in Im / W) as a function of the brightness, calculated from current-voltage-brightness characteristics (IUL characteristics), and the lifetime are determined.
- the lifetime is defined as the time after which the initial brightness of 6000 cd / m 2 has fallen to half.
- Table 2 summarizes the results of some OLEDs (Examples 7 to 17).
- the compounds of Examples 1 to 4 or 5 are used.
- the host H1 and the electron transport materials ETM1 and ETM2 are used according to the prior art.
- organic electroluminescent devices containing the compounds according to the invention have a longer lifetime and a better efficiency than organic electroluminescent devices according to the prior art.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Photovoltaic Devices (AREA)
- Indole Compounds (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/867,648 US8993123B2 (en) | 2008-02-13 | 2009-02-13 | Materials for organic electroluminescent devices |
| CN200980104579.3A CN101939279B (zh) | 2008-02-13 | 2009-02-13 | 用于有机电致发光器件的新材料 |
| JP2010546263A JP5638960B2 (ja) | 2008-02-13 | 2009-02-13 | 有機エレクトロルミネセントデバイスのための新規材料 |
| EP09709699A EP2240425A1 (de) | 2008-02-13 | 2009-02-13 | Neue materialien für organische elektrolumineszenzvorrichtungen |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102008008953.2 | 2008-02-13 | ||
| DE102008008953.2A DE102008008953B4 (de) | 2008-02-13 | 2008-02-13 | Neue Materialien für organische Elektrolumineszenzvorrichtungen |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009100925A1 true WO2009100925A1 (de) | 2009-08-20 |
Family
ID=40718755
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/001028 Ceased WO2009100925A1 (de) | 2008-02-13 | 2009-02-13 | Neue materialien für organische elektrolumineszenzvorrichtungen |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US8993123B2 (de) |
| EP (1) | EP2240425A1 (de) |
| JP (1) | JP5638960B2 (de) |
| KR (1) | KR101573096B1 (de) |
| CN (1) | CN101939279B (de) |
| DE (1) | DE102008008953B4 (de) |
| TW (1) | TWI473778B (de) |
| WO (1) | WO2009100925A1 (de) |
Cited By (54)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012060307A1 (en) * | 2010-11-05 | 2012-05-10 | Canon Kabushiki Kaisha | Phenanthrene compound and organic light emitting device using the same |
| WO2012091225A1 (ko) * | 2010-12-31 | 2012-07-05 | 제일모직 주식회사 | 유기광전자소자용 화합물, 이를 포함하는 유기발광소자 및 상기 유기발광소자를 포함하는 표시장치 |
| WO2014067614A1 (de) | 2012-10-31 | 2014-05-08 | Merck Patent Gmbh | Elektronische vorrichtung |
| WO2014106523A1 (de) | 2013-01-03 | 2014-07-10 | Merck Patent Gmbh | Elektronische vorrichtung |
| WO2014106524A2 (de) | 2013-01-03 | 2014-07-10 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2016119992A1 (en) | 2015-01-30 | 2016-08-04 | Merck Patent Gmbh | Materials for electronic devices |
| WO2017036573A1 (en) | 2015-08-28 | 2017-03-09 | Merck Patent Gmbh | Compounds for electronic devices |
| WO2017133829A1 (de) | 2016-02-05 | 2017-08-10 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018069167A1 (de) | 2016-10-10 | 2018-04-19 | Merck Patent Gmbh | Elektronische vorrichtung |
| DE102017008794A1 (de) | 2016-10-17 | 2018-04-19 | Merck Patent Gmbh | Materialien zur Verwendung in elektronischen Vorrichtungen |
| WO2018083053A1 (de) | 2016-11-02 | 2018-05-11 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018087020A1 (en) | 2016-11-08 | 2018-05-17 | Merck Patent Gmbh | Compounds for electronic devices |
| WO2018095839A1 (de) | 2016-11-22 | 2018-05-31 | Merck Patent Gmbh | Verbrückte triarylamine für elektronische vorrichtungen |
| WO2018114882A1 (de) | 2016-12-22 | 2018-06-28 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018141706A1 (de) | 2017-02-02 | 2018-08-09 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018157981A1 (de) | 2017-03-02 | 2018-09-07 | Merck Patent Gmbh | Materialien für organische elektronische vorrichtungen |
| EP3378857A1 (de) | 2012-11-12 | 2018-09-26 | Merck Patent GmbH | Materialien für elektronische vorrichtungen |
| WO2018197447A1 (de) | 2017-04-25 | 2018-11-01 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen |
| WO2018234220A1 (de) | 2017-06-21 | 2018-12-27 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018234346A1 (en) | 2017-06-23 | 2018-12-27 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2019002198A1 (en) | 2017-06-26 | 2019-01-03 | Merck Patent Gmbh | HOMOGENEOUS MIXTURES |
| WO2019002190A1 (en) | 2017-06-28 | 2019-01-03 | Merck Patent Gmbh | MATERIALS FOR ELECTRONIC DEVICES |
| WO2019020654A1 (en) | 2017-07-28 | 2019-01-31 | Merck Patent Gmbh | SPIROBIFLUORENE DERIVATIVES FOR USE IN ELECTRONIC DEVICES |
| WO2019048443A1 (de) | 2017-09-08 | 2019-03-14 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2019101719A1 (de) | 2017-11-23 | 2019-05-31 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2019115577A1 (en) | 2017-12-15 | 2019-06-20 | Merck Patent Gmbh | Substituted aromatic amines for use in organic electroluminescent devices |
| WO2019121483A1 (en) | 2017-12-20 | 2019-06-27 | Merck Patent Gmbh | Heteroaromatic compounds |
| WO2019175149A1 (en) | 2018-03-16 | 2019-09-19 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2020178230A1 (en) | 2019-03-04 | 2020-09-10 | Merck Patent Gmbh | Ligands for nano-sized materials |
| WO2021180950A1 (en) | 2020-03-13 | 2021-09-16 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022017997A1 (en) | 2020-07-22 | 2022-01-27 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022017998A1 (en) | 2020-07-22 | 2022-01-27 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022112541A1 (en) | 2020-11-30 | 2022-06-02 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022214506A1 (en) | 2021-04-09 | 2022-10-13 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022214507A1 (en) | 2021-04-09 | 2022-10-13 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2023052313A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023052272A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023052314A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023052275A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023094412A1 (de) | 2021-11-25 | 2023-06-01 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023117837A1 (de) | 2021-12-21 | 2023-06-29 | Merck Patent Gmbh | Verfahren zur herstellung von deuterierten organischen verbindungen |
| WO2023152346A1 (de) | 2022-02-14 | 2023-08-17 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023222559A1 (de) | 2022-05-18 | 2023-11-23 | Merck Patent Gmbh | Verfahren zur herstellung von deuterierten organischen verbindungen |
| WO2024013004A1 (de) | 2022-07-11 | 2024-01-18 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2024133048A1 (en) | 2022-12-20 | 2024-06-27 | Merck Patent Gmbh | Method for preparing deuterated aromatic compounds |
| WO2024170605A1 (en) | 2023-02-17 | 2024-08-22 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2024218109A1 (de) | 2023-04-20 | 2024-10-24 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2024240725A1 (de) | 2023-05-25 | 2024-11-28 | Merck Patent Gmbh | Tris[1,2,4]triazolo[1,5-a:1',5'-c:1'',5''-e][1,3,5]triazin-derivate zur verwendung in organischen elektrolumineszenzvorrichtungen |
| WO2025012253A1 (en) | 2023-07-12 | 2025-01-16 | Merck Patent Gmbh | Materials for electronic devices |
| WO2025021855A1 (de) | 2023-07-27 | 2025-01-30 | Merck Patent Gmbh | Materialien für organische lichtemittierende vorrichtungen und organische sensoren |
| WO2025132547A1 (en) | 2023-12-21 | 2025-06-26 | Merck Patent Gmbh | Mechanochemical method for deuterating organic compounds |
| WO2025132551A1 (de) | 2023-12-22 | 2025-06-26 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2025196145A1 (en) | 2024-03-22 | 2025-09-25 | Merck Patent Gmbh | Materials for organic light emitting devices |
| WO2025210013A1 (de) | 2024-04-04 | 2025-10-09 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen, insbesondere verbindungen für oleds |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102010048608A1 (de) * | 2010-10-15 | 2012-04-19 | Merck Patent Gmbh | Materialien für organische Elektrolumineszenzvorrichtungen |
| KR101996648B1 (ko) * | 2011-04-15 | 2019-07-04 | 에스에프씨 주식회사 | 신규한 화합물 및 이를 포함하는 유기전계발광소자 |
| KR102168730B1 (ko) | 2012-09-12 | 2020-10-22 | 이데미쓰 고산 가부시키가이샤 | 신규 화합물, 유기 일렉트로 루미네선스 소자용 재료, 유기 일렉트로 루미네선스 소자 및 전자 기기 |
| KR102013879B1 (ko) * | 2012-11-30 | 2019-08-29 | 엘지디스플레이 주식회사 | 유기발광장치 및 그 제조방법 |
| KR20150026114A (ko) | 2013-08-30 | 2015-03-11 | 삼성디스플레이 주식회사 | 인데노피리딘계 화합물 및 이를 포함한 유기 발광 소자 |
| US10559756B2 (en) | 2013-10-14 | 2020-02-11 | Merck Patent Gmbh | Materials for electronic devices |
| US10727413B2 (en) * | 2014-03-07 | 2020-07-28 | Merck Patent Gmbh | Materials for electronic devices |
| JP6305798B2 (ja) * | 2014-03-17 | 2018-04-04 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子および電子機器 |
| US10103334B2 (en) | 2014-12-29 | 2018-10-16 | Dow Global Technologies Llc | Compositions with triarylamine derivatives and OLED device containing the same |
| KR102424977B1 (ko) | 2015-04-14 | 2022-07-26 | 삼성디스플레이 주식회사 | 축합환 화합물 및 이를 포함한 유기 발광 소자 |
| KR102471110B1 (ko) | 2015-08-11 | 2022-11-28 | 삼성디스플레이 주식회사 | 유기 발광 소자 |
| KR102606275B1 (ko) | 2015-10-27 | 2023-12-05 | 삼성디스플레이 주식회사 | 유기 발광 소자 |
| KR102630644B1 (ko) | 2015-12-17 | 2024-01-30 | 삼성디스플레이 주식회사 | 유기 발광 소자 |
| KR102603865B1 (ko) | 2016-04-21 | 2023-11-21 | 삼성디스플레이 주식회사 | 유기 발광 소자 |
| KR102780240B1 (ko) | 2017-04-26 | 2025-03-14 | 오티아이 루미오닉스 인크. | 표면의 코팅을 패턴화하는 방법 및 패턴화된 코팅을 포함하는 장치 |
| US11751415B2 (en) | 2018-02-02 | 2023-09-05 | Oti Lumionics Inc. | Materials for forming a nucleation-inhibiting coating and devices incorporating same |
| CN108191767A (zh) * | 2018-02-07 | 2018-06-22 | 瑞声科技(南京)有限公司 | Tadf材料及在有机发光二极管器件中的应用 |
| KR102230988B1 (ko) * | 2018-03-14 | 2021-03-23 | 주식회사 엘지화학 | 화합물 및 이를 포함하는 유기 발광 소자 |
| EP3598515B1 (de) | 2018-07-18 | 2024-09-04 | Novaled GmbH | Verbindung und organische halbleitende schicht, organische elektronische vorrichtung, anzeigevorrichtung und beleuchtungsvorrichtung damit |
| CN111349055B (zh) * | 2018-12-24 | 2023-04-21 | 烟台显华光电材料研究院有限公司 | 一类用作电致发光材料的多环化合物及应用 |
| WO2020178804A1 (en) | 2019-03-07 | 2020-09-10 | Oti Lumionics Inc. | Materials for forming a nucleation-inhibiting coating and devices incorporating same |
| US12101987B2 (en) | 2019-04-18 | 2024-09-24 | Oti Lumionics Inc. | Materials for forming a nucleation-inhibiting coating and devices incorporating same |
| CN110041159A (zh) * | 2019-04-24 | 2019-07-23 | 北京诚志永华显示科技有限公司 | 新型化合物、有机电致发光材料、有机电致发光元件、电子装置 |
| CN114072705A (zh) | 2019-05-08 | 2022-02-18 | Oti照明公司 | 用于形成成核抑制涂层的材料和结合所述成核抑制涂层的装置 |
| US11985841B2 (en) | 2020-12-07 | 2024-05-14 | Oti Lumionics Inc. | Patterning a conductive deposited layer using a nucleation inhibiting coating and an underlying metallic coating |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5935721A (en) * | 1998-03-20 | 1999-08-10 | Eastman Kodak Company | Organic electroluminescent elements for stable electroluminescent |
| US20070037983A1 (en) * | 2005-08-11 | 2007-02-15 | Semiconductor Energy Laboratory Co., Ltd. | Phenanthroline derivative compound |
| WO2007123256A1 (en) * | 2006-04-20 | 2007-11-01 | Canon Kabushiki Kaisha | Compound and organic light emitting device |
| US20080004445A1 (en) * | 2006-04-18 | 2008-01-03 | Idemitsu Kosan Co., Ltd. | Aromatic amine derivative and organic electroluminescence device using the same |
| US20080079356A1 (en) * | 2006-09-29 | 2008-04-03 | Sang-Hoon Park | Organoelectroluminescent compound and organoelectroluminescent device employing the same |
| WO2008094399A1 (en) * | 2007-01-30 | 2008-08-07 | Eastman Kodak Company | Oleds having high efficiency and excellent lifetime |
Family Cites Families (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4539507A (en) | 1983-03-25 | 1985-09-03 | Eastman Kodak Company | Organic electroluminescent devices having improved power conversion efficiencies |
| US5151629A (en) | 1991-08-01 | 1992-09-29 | Eastman Kodak Company | Blue emitting internal junction organic electroluminescent device (I) |
| DE59510315D1 (de) | 1994-04-07 | 2002-09-19 | Covion Organic Semiconductors | Spiroverbindungen und ihre Verwendung als Elektrolumineszenzmaterialien |
| DE19652261A1 (de) | 1996-12-16 | 1998-06-18 | Hoechst Ag | Arylsubstituierte Poly(p-arylenvinylene), Verfahren zur Herstellung und deren Verwendung in Elektroluminszenzbauelementen |
| KR100809132B1 (ko) | 1999-09-21 | 2008-02-29 | 이데미쓰 고산 가부시키가이샤 | 유기 전자발광 소자 및 유기 발광 매체 |
| JP4094203B2 (ja) | 2000-03-30 | 2008-06-04 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び有機発光媒体 |
| JP2001332384A (ja) | 2000-05-19 | 2001-11-30 | Toray Ind Inc | 発光素子 |
| CN1239447C (zh) | 2002-01-15 | 2006-02-01 | 清华大学 | 一种有机电致发光材料 |
| JP4170655B2 (ja) | 2002-04-17 | 2008-10-22 | 出光興産株式会社 | 新規芳香族化合物及びそれを利用した有機エレクトロルミネッセンス素子 |
| JP2005531552A (ja) | 2002-05-07 | 2005-10-20 | エルジー・ケム・リミテッド | 新たな有機発光化合物及びこれを利用した有機発光素子 |
| EP1541657B2 (de) | 2002-07-19 | 2014-03-05 | Idemitsu Kosan Co., Ltd. | Organische elektrolumineszenzvorrichtungen und organisches lumineszenzmedium |
| JP4025137B2 (ja) | 2002-08-02 | 2007-12-19 | 出光興産株式会社 | アントラセン誘導体及びそれを利用した有機エレクトロルミネッセンス素子 |
| JP2004075567A (ja) | 2002-08-12 | 2004-03-11 | Idemitsu Kosan Co Ltd | オリゴアリーレン誘導体及びそれを利用した有機エレクトロルミネッセンス素子 |
| ATE452954T1 (de) | 2002-08-23 | 2010-01-15 | Idemitsu Kosan Co | Organische elektrolumineszenzvorrichtung und anthracenderivat |
| CN100489056C (zh) | 2002-12-23 | 2009-05-20 | 默克专利有限公司 | 有机电致发光元件 |
| DE10333232A1 (de) | 2003-07-21 | 2007-10-11 | Merck Patent Gmbh | Organisches Elektrolumineszenzelement |
| JP4070676B2 (ja) | 2003-07-25 | 2008-04-02 | 三井化学株式会社 | 非対称置換アントラセン化合物、および該非対称置換アントラセン化合物を含有する有機電界発光素子 |
| US7887931B2 (en) * | 2003-10-24 | 2011-02-15 | Global Oled Technology Llc | Electroluminescent device with anthracene derivative host |
| TWI373506B (en) | 2004-05-21 | 2012-10-01 | Toray Industries | Light-emitting element material and light-emitting material |
| TW200613515A (en) | 2004-06-26 | 2006-05-01 | Merck Patent Gmbh | Compounds for organic electronic devices |
| DE102004031000A1 (de) | 2004-06-26 | 2006-01-12 | Covion Organic Semiconductors Gmbh | Organische Elektrolumineszenzvorrichtungen |
| ITRM20040352A1 (it) | 2004-07-15 | 2004-10-15 | Univ Roma La Sapienza | Derivati oligomerici dello spirobifluorene, loro preparazione e loro uso. |
| JP4677221B2 (ja) | 2004-11-26 | 2011-04-27 | キヤノン株式会社 | 有機発光素子 |
| TW200639140A (en) | 2004-12-01 | 2006-11-16 | Merck Patent Gmbh | Compounds for organic electronic devices |
| DE102005023437A1 (de) | 2005-05-20 | 2006-11-30 | Merck Patent Gmbh | Verbindungen für organische elektronische Vorrichtungen |
| US7300511B2 (en) | 2005-07-01 | 2007-11-27 | Sun Chemical Corporation | Low viscosity, highly pigmented oil based dispersions exhibiting a low relative interfacial tension drop |
| DE102005058543A1 (de) | 2005-12-08 | 2007-06-14 | Merck Patent Gmbh | Organische Elektrolumineszenzvorrichtungen |
| JP2007230951A (ja) | 2006-03-02 | 2007-09-13 | Canon Inc | シリル化合物、発光材料およびそれを用いた有機発光素子 |
| DE102006015183A1 (de) | 2006-04-01 | 2007-10-04 | Merck Patent Gmbh | Materialien für organische Elektrolumineszenzvorrichtungen |
| JP5317414B2 (ja) * | 2006-04-20 | 2013-10-16 | キヤノン株式会社 | ジベンゾアントラセン化合物およびそれを有する有機発光素子 |
| DE102006025846A1 (de) | 2006-06-02 | 2007-12-06 | Merck Patent Gmbh | Neue Materialien für organische Elektrolumineszenzvorrichtungen |
| DE102006031990A1 (de) | 2006-07-11 | 2008-01-17 | Merck Patent Gmbh | Neue Materialien für organische Elektrolumineszenzvorrichtungen |
| KR101408504B1 (ko) | 2007-11-29 | 2014-06-17 | 이데미쓰 고산 가부시키가이샤 | 벤조페난트렌 유도체 및 이것을 사용한 유기 전계 발광 소자 |
-
2008
- 2008-02-13 DE DE102008008953.2A patent/DE102008008953B4/de active Active
-
2009
- 2009-02-10 TW TW98104217A patent/TWI473778B/zh active
- 2009-02-13 US US12/867,648 patent/US8993123B2/en active Active
- 2009-02-13 WO PCT/EP2009/001028 patent/WO2009100925A1/de not_active Ceased
- 2009-02-13 KR KR1020107019981A patent/KR101573096B1/ko active Active
- 2009-02-13 CN CN200980104579.3A patent/CN101939279B/zh active Active
- 2009-02-13 JP JP2010546263A patent/JP5638960B2/ja active Active
- 2009-02-13 EP EP09709699A patent/EP2240425A1/de not_active Withdrawn
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5935721A (en) * | 1998-03-20 | 1999-08-10 | Eastman Kodak Company | Organic electroluminescent elements for stable electroluminescent |
| US20070037983A1 (en) * | 2005-08-11 | 2007-02-15 | Semiconductor Energy Laboratory Co., Ltd. | Phenanthroline derivative compound |
| US20080004445A1 (en) * | 2006-04-18 | 2008-01-03 | Idemitsu Kosan Co., Ltd. | Aromatic amine derivative and organic electroluminescence device using the same |
| WO2007123256A1 (en) * | 2006-04-20 | 2007-11-01 | Canon Kabushiki Kaisha | Compound and organic light emitting device |
| US20080079356A1 (en) * | 2006-09-29 | 2008-04-03 | Sang-Hoon Park | Organoelectroluminescent compound and organoelectroluminescent device employing the same |
| WO2008094399A1 (en) * | 2007-01-30 | 2008-08-07 | Eastman Kodak Company | Oleds having high efficiency and excellent lifetime |
Cited By (65)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012097056A (ja) * | 2010-11-05 | 2012-05-24 | Canon Inc | フェナンスレン化合物及びこれを用いた有機発光素子 |
| US8729541B2 (en) | 2010-11-05 | 2014-05-20 | Canon Kabushiki Kaisha | Phenanthrene compound and organic light emitting device using the same |
| KR101523153B1 (ko) * | 2010-11-05 | 2015-05-26 | 캐논 가부시끼가이샤 | 페난트렌 화합물 및 이를 사용한 유기 발광 디바이스 |
| WO2012060307A1 (en) * | 2010-11-05 | 2012-05-10 | Canon Kabushiki Kaisha | Phenanthrene compound and organic light emitting device using the same |
| WO2012091225A1 (ko) * | 2010-12-31 | 2012-07-05 | 제일모직 주식회사 | 유기광전자소자용 화합물, 이를 포함하는 유기발광소자 및 상기 유기발광소자를 포함하는 표시장치 |
| US8796917B2 (en) | 2010-12-31 | 2014-08-05 | Cheil Industries, Inc | Compound for an organic optoelectronic device, organic light emitting diode including the same, and display including the organic light emitting diode |
| KR101432600B1 (ko) | 2010-12-31 | 2014-08-21 | 제일모직주식회사 | 유기광전소자용 화합물 및 이를 포함하는 유기광전소자 |
| EP3806176A1 (de) | 2012-10-31 | 2021-04-14 | Merck Patent GmbH | Elektronische vorrichtung |
| WO2014067614A1 (de) | 2012-10-31 | 2014-05-08 | Merck Patent Gmbh | Elektronische vorrichtung |
| EP3378857A1 (de) | 2012-11-12 | 2018-09-26 | Merck Patent GmbH | Materialien für elektronische vorrichtungen |
| WO2014106523A1 (de) | 2013-01-03 | 2014-07-10 | Merck Patent Gmbh | Elektronische vorrichtung |
| WO2014106524A2 (de) | 2013-01-03 | 2014-07-10 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2016119992A1 (en) | 2015-01-30 | 2016-08-04 | Merck Patent Gmbh | Materials for electronic devices |
| WO2017036573A1 (en) | 2015-08-28 | 2017-03-09 | Merck Patent Gmbh | Compounds for electronic devices |
| WO2017133829A1 (de) | 2016-02-05 | 2017-08-10 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018069167A1 (de) | 2016-10-10 | 2018-04-19 | Merck Patent Gmbh | Elektronische vorrichtung |
| EP4113643A1 (de) | 2016-10-10 | 2023-01-04 | Merck Patent GmbH | Elektronische vorrichtung |
| EP4255151A2 (de) | 2016-10-10 | 2023-10-04 | Merck Patent GmbH | Spiro[fluoren-9,9'-(thio)xanthen] verbindungen |
| DE102017008794A1 (de) | 2016-10-17 | 2018-04-19 | Merck Patent Gmbh | Materialien zur Verwendung in elektronischen Vorrichtungen |
| WO2018083053A1 (de) | 2016-11-02 | 2018-05-11 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018087020A1 (en) | 2016-11-08 | 2018-05-17 | Merck Patent Gmbh | Compounds for electronic devices |
| WO2018095839A1 (de) | 2016-11-22 | 2018-05-31 | Merck Patent Gmbh | Verbrückte triarylamine für elektronische vorrichtungen |
| WO2018114882A1 (de) | 2016-12-22 | 2018-06-28 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018141706A1 (de) | 2017-02-02 | 2018-08-09 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018157981A1 (de) | 2017-03-02 | 2018-09-07 | Merck Patent Gmbh | Materialien für organische elektronische vorrichtungen |
| WO2018197447A1 (de) | 2017-04-25 | 2018-11-01 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen |
| WO2018234220A1 (de) | 2017-06-21 | 2018-12-27 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2018234346A1 (en) | 2017-06-23 | 2018-12-27 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2019002198A1 (en) | 2017-06-26 | 2019-01-03 | Merck Patent Gmbh | HOMOGENEOUS MIXTURES |
| WO2019002190A1 (en) | 2017-06-28 | 2019-01-03 | Merck Patent Gmbh | MATERIALS FOR ELECTRONIC DEVICES |
| EP4603562A2 (de) | 2017-07-28 | 2025-08-20 | Merck Patent GmbH | Spirobifluorenderivate zur verwendung in elektronischen vorrichtungen |
| WO2019020654A1 (en) | 2017-07-28 | 2019-01-31 | Merck Patent Gmbh | SPIROBIFLUORENE DERIVATIVES FOR USE IN ELECTRONIC DEVICES |
| WO2019048443A1 (de) | 2017-09-08 | 2019-03-14 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2019101719A1 (de) | 2017-11-23 | 2019-05-31 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| EP4242286A2 (de) | 2017-11-23 | 2023-09-13 | Merck Patent GmbH | Materialien für elektronische vorrichtungen |
| WO2019115577A1 (en) | 2017-12-15 | 2019-06-20 | Merck Patent Gmbh | Substituted aromatic amines for use in organic electroluminescent devices |
| WO2019121483A1 (en) | 2017-12-20 | 2019-06-27 | Merck Patent Gmbh | Heteroaromatic compounds |
| EP4451832A2 (de) | 2017-12-20 | 2024-10-23 | Merck Patent GmbH | Heteroaromatische verbindungen |
| WO2019175149A1 (en) | 2018-03-16 | 2019-09-19 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2020178230A1 (en) | 2019-03-04 | 2020-09-10 | Merck Patent Gmbh | Ligands for nano-sized materials |
| WO2021180950A1 (en) | 2020-03-13 | 2021-09-16 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022017998A1 (en) | 2020-07-22 | 2022-01-27 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022017997A1 (en) | 2020-07-22 | 2022-01-27 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022112541A1 (en) | 2020-11-30 | 2022-06-02 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022214507A1 (en) | 2021-04-09 | 2022-10-13 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2022214506A1 (en) | 2021-04-09 | 2022-10-13 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2023052275A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023052314A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023052272A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023052313A1 (de) | 2021-09-28 | 2023-04-06 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023094412A1 (de) | 2021-11-25 | 2023-06-01 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023117837A1 (de) | 2021-12-21 | 2023-06-29 | Merck Patent Gmbh | Verfahren zur herstellung von deuterierten organischen verbindungen |
| WO2023152346A1 (de) | 2022-02-14 | 2023-08-17 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2023222559A1 (de) | 2022-05-18 | 2023-11-23 | Merck Patent Gmbh | Verfahren zur herstellung von deuterierten organischen verbindungen |
| WO2024013004A1 (de) | 2022-07-11 | 2024-01-18 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2024133048A1 (en) | 2022-12-20 | 2024-06-27 | Merck Patent Gmbh | Method for preparing deuterated aromatic compounds |
| WO2024170605A1 (en) | 2023-02-17 | 2024-08-22 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2024218109A1 (de) | 2023-04-20 | 2024-10-24 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2024240725A1 (de) | 2023-05-25 | 2024-11-28 | Merck Patent Gmbh | Tris[1,2,4]triazolo[1,5-a:1',5'-c:1'',5''-e][1,3,5]triazin-derivate zur verwendung in organischen elektrolumineszenzvorrichtungen |
| WO2025012253A1 (en) | 2023-07-12 | 2025-01-16 | Merck Patent Gmbh | Materials for electronic devices |
| WO2025021855A1 (de) | 2023-07-27 | 2025-01-30 | Merck Patent Gmbh | Materialien für organische lichtemittierende vorrichtungen und organische sensoren |
| WO2025132547A1 (en) | 2023-12-21 | 2025-06-26 | Merck Patent Gmbh | Mechanochemical method for deuterating organic compounds |
| WO2025132551A1 (de) | 2023-12-22 | 2025-06-26 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2025196145A1 (en) | 2024-03-22 | 2025-09-25 | Merck Patent Gmbh | Materials for organic light emitting devices |
| WO2025210013A1 (de) | 2024-04-04 | 2025-10-09 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen, insbesondere verbindungen für oleds |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101939279A (zh) | 2011-01-05 |
| TWI473778B (zh) | 2015-02-21 |
| CN101939279B (zh) | 2015-03-25 |
| US20100327270A1 (en) | 2010-12-30 |
| DE102008008953B4 (de) | 2019-05-09 |
| KR20100110895A (ko) | 2010-10-13 |
| KR101573096B1 (ko) | 2015-11-30 |
| JP5638960B2 (ja) | 2014-12-10 |
| DE102008008953A1 (de) | 2009-08-20 |
| TW200951100A (en) | 2009-12-16 |
| JP2011514885A (ja) | 2011-05-12 |
| EP2240425A1 (de) | 2010-10-20 |
| US8993123B2 (en) | 2015-03-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE102008008953B4 (de) | Neue Materialien für organische Elektrolumineszenzvorrichtungen | |
| DE112009003904B4 (de) | Materialien für organische Elektrolumineszenzvorrichtungen | |
| EP2148909B1 (de) | Benzanthracen-derivate für organische elektrolumineszenzvorrichtungen | |
| DE112010003082B4 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP2265689B1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP2260023B1 (de) | Fluorenderivate für organische elektrolumineszenzvorrichtungen | |
| EP3211058B1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP1999226B1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP2038370B1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP1859005B1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP1957606B1 (de) | Neue materialien für organische elektroluminieszenzvorrichtungen | |
| WO2012048781A1 (de) | Materialien auf basis von triphenylen für organische elektrolumineszenzvorrichtungen | |
| WO2010006680A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| WO2006131192A1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| DE102006035035A1 (de) | Neue Materialien für organische Elektrolumineszenzvorrichtungen | |
| DE102006025846A1 (de) | Neue Materialien für organische Elektrolumineszenzvorrichtungen | |
| EP2340290A1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP1871856A1 (de) | Organische elektrolumineszenzvorrichtungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980104579.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09709699 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009709699 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010546263 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12867648 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20107019981 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3369/KOLNP/2010 Country of ref document: IN |