WO2006057323A1 - アミロイド関連疾患診断用組成物 - Google Patents
アミロイド関連疾患診断用組成物 Download PDFInfo
- Publication number
- WO2006057323A1 WO2006057323A1 PCT/JP2005/021642 JP2005021642W WO2006057323A1 WO 2006057323 A1 WO2006057323 A1 WO 2006057323A1 JP 2005021642 W JP2005021642 W JP 2005021642W WO 2006057323 A1 WO2006057323 A1 WO 2006057323A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- phe
- compound
- amyloid
- composition
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C225/00—Compounds containing amino groups and doubly—bound oxygen atoms bound to the same carbon skeleton, at least one of the doubly—bound oxygen atoms not being part of a —CHO group, e.g. amino ketones
- C07C225/22—Compounds containing amino groups and doubly—bound oxygen atoms bound to the same carbon skeleton, at least one of the doubly—bound oxygen atoms not being part of a —CHO group, e.g. amino ketones having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/0004—Screening or testing of compounds for diagnosis of disorders, assessment of conditions, e.g. renal clearance, gastric emptying, testing for diabetes, allergy, rheuma, pancreas functions
- A61K49/0008—Screening agents using (non-human) animal models or transgenic animal models or chimeric hosts, e.g. Alzheimer disease animal model, transgenic model for heart failure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/041—Heterocyclic compounds
- A61K51/0412—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K51/0421—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/041—Heterocyclic compounds
- A61K51/0412—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K51/0427—Lactones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C205/00—Compounds containing nitro groups bound to a carbon skeleton
- C07C205/45—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by at least one doubly—bound oxygen atom, not being part of a —CHO group
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C49/00—Ketones; Ketenes; Dimeric ketenes; Ketonic chelates
- C07C49/76—Ketones containing a keto group bound to a six-membered aromatic ring
- C07C49/84—Ketones containing a keto group bound to a six-membered aromatic ring containing ether groups, groups, groups, or groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D311/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings
- C07D311/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D311/04—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring
- C07D311/22—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring with oxygen or sulfur atoms directly attached in position 4
- C07D311/26—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring with oxygen or sulfur atoms directly attached in position 4 with aromatic rings attached in position 2 or 3
- C07D311/28—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring with oxygen or sulfur atoms directly attached in position 4 with aromatic rings attached in position 2 or 3 with aromatic rings attached in position 2 only
- C07D311/30—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring with oxygen or sulfur atoms directly attached in position 4 with aromatic rings attached in position 2 or 3 with aromatic rings attached in position 2 only not hydrogenated in the hetero ring, e.g. flavones
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6893—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to diseases not provided for elsewhere
- G01N33/6896—Neurological disorders, e.g. Alzheimer's disease
Definitions
- composition for diagnosing amyloid-related diseases Composition for diagnosing amyloid-related diseases
- the present invention relates to a composition used for diagnosing amyloid-related diseases such as Arno-i-maima disease, a method for screening a therapeutic or prophylactic agent for amyloid-related diseases using the composition, and the composition
- the present invention relates to a method for evaluating a therapeutic or prophylactic agent for amyloid-related diseases using a product.
- AD Arnno-Ima disease
- Hasegawa formula, ADAS, and MMSE clinical diagnostic methods for AD, and all of them generally use a method for quantitatively evaluating the decline of cognitive function in individuals suspected of having AD.
- Other diagnostic imaging methods MRI, CT, etc.
- MRI, CT, etc. are used supplementarily, but these diagnostic methods are not sufficient for definitive diagnosis of AD.
- biopsy of the brain before birth, postmortem brain It is necessary to confirm the appearance of senile plaques and neurofibrils during the pathologic and histological examination.
- Senile plaque is the most characteristic brain lesion of AD, and its main component is amyloid j8 protein with ⁇ -sheet structure. Imaging of senile plaques from outside the body seems to lead to the establishment of an effective diagnostic method for AD Imaging requires a probe compound that specifically binds to amyloid j8 protein.
- Patent Document 1 Patent Document 2, Non-Patent Document 1
- 8 protein There are a number of problems such as low binding specificity to, low permeability of the blood brain barrier, and slow clearance due to nonspecific binding in the brain. Therefore, these reported compounds still accumulate amyloid. The current situation is that it has been put into practical use.
- Patent Document 1 JP 2004-250407 A
- Patent Document 2 JP 2004-250411 A
- Non-Patent Document 1 W.E.Klunk et al, Annals of Neurology Vol55 No.3 March 2004 306-3 19
- the present invention has been made on the basis of the technical background as described above, and has high binding specificity for amyloid j8 protein, high blood-brain barrier permeability, and sites other than senile plaques in the brain.
- the object is to provide a compound having both rapidity and rapid disappearance.
- the present invention provides the following (1) to (20).
- R 2 , R 3 and R 4 are each independently a hydrogen atom, halogen atom, hydroxyl group, carboxyl group, sulfone group, dimethylamino group, methylamino group, amino group, nitro group, C1-4 alkyl group, C1-4 alkoxy group, 2 fluoroethoxy group, 3 fluoropropoxy group, 4 fluorobutoxy group, or 5-fluoropentoxy group
- a composition for diagnosing amyloid-related diseases comprising a compound represented by the formula: or a pharmaceutically acceptable salt thereof.
- composition for diagnosing amyloid-related disease according to (1) wherein any of R 2 , R 3 , and R 4 in the general formula (I) is a halogen atom.
- composition for diagnosing amyloid-related disease wherein any of R 2 , R 3 and R 4 in the general formula (I) is an iodine atom.
- R 5 in the general formula (I) may be substituted with one or more substituents selected from Substituent Group A, and may be a phenyl group (1)
- the composition for diagnosing amyloid-related diseases according to any one of (3) to (3).
- R 5 in the general formula (I) is a 4-dimethylaminophenol group, a 4-methylaminophenol group, a 4-methoxyphenol group, or a 4-hydroxyphenol group.
- the composition for diagnosing amyloid-related disease according to any one of (1) to (3).
- R 5 in the general formula (I) is a group represented by the formula: —CH ⁇ CH—R 6 (wherein R 6 is a substituent group A force is also selected 1 or 2 or more (1) to (3), the composition for diagnosing amyloid-related diseases according to any one of (1) to (3).
- —R 5 in the general formula (I) is a 4 aminostyryl group, a 4 methylaminostyryl group, or The composition for diagnosing amyloid-related disease according to any one of (1) to (3), which is a 4-dimethylaminostyryl group.
- RR 2 , R 3 , and R 4 are each independently a hydrogen atom, halogen atom, hydroxyl group, carboxyl group, sulfone group, dimethylamino group, methylamino group, amino group, nitro group, carbon Represents an alkyl group having 1 to 4 carbon atoms, an alkoxy group having 1 to 4 carbon atoms, 2 fluoroethoxy group, 3 fluoropropoxy group, 4 fluorobutoxy group, or 5-fluoropentoxy group
- R 7 is the following substituent Group A force is also selected from an aryl group optionally substituted by one or more selected substituents, or an aromatic complex optionally substituted by one or more substituents selected from the following substituent group A
- the substituent group A is a halogen atom, hydroxyl group, carboxyl group, sulfone group, dimethylamino group, methylamino group, amino group, nitro group, alkyl group having 1 to 4 carbon atoms,
- a composition for diagnosing amyloid-related diseases comprising a compound represented by the formula: or a pharmaceutically acceptable salt thereof.
- composition for diagnosing amyloid-related diseases according to (8), wherein any of R 2 , R 3 and R 4 in the general formula (II) is a halogen atom.
- R 7 in the general formula (II) is a 4-hydroxy-1-methoxyphenyl group, or a 4-dimethyl group.
- RR 2 , R 3 , and R 4 are each independently a hydrogen atom, halogen atom, hydroxyl group, carboxyl group, sulfone group, dimethylamino group, methylamino group, amino group, nitro group, carbon Represents an alkyl group having 1 to 4 carbon atoms, an alkoxy group having 1 to 4 carbon atoms, 2 fluoroethoxy group, 3 fluoropropoxy group, 4 fluorobutoxy group, or 5-fluoropentoxy group
- R 8 is the following substituent Group A force is also selected from an aryl group optionally substituted by one or more selected substituents, or an aromatic complex optionally substituted by one or more substituents selected from the following substituent group A
- the substituent group A is a halogen atom, hydroxyl group, carboxyl group, sulfone group, dimethylamino group, methylamino group, amino group, nitro group, alkyl group having 1 to 4 carbon atoms,
- a composition for diagnosing amyloid-related diseases comprising a compound represented by the formula: or a pharmaceutically acceptable salt thereof.
- composition for diagnosing amyloid-related disease according to (12), wherein any of R 2 , R 3 , and R 4 in the general formula (III) is an iodine atom.
- Amyloid-related disease power The composition for diagnosing amyloid-related disease according to any one of (1) to (17), which is Alzheimer's disease.
- a step of administering a test substance to a model animal of amyloid-related disease a step of administering to the model animal the composition for diagnosing amyloid-related disease according to any one of (1) to (18), and
- An amyloid-related disease comprising a step of examining the distribution or amount of the compound represented by the general formula (1), the general formula (11), or the general formula (III) contained in the brain of the model animal.
- a screening method for therapeutic or prophylactic agents comprising a step of examining the distribution or amount of the compound represented by the general formula (1), the general formula (11), or the general formula (III) contained in the brain of the model animal.
- An amyloid-related disease comprising the steps of: administering and the method of examining the distribution or amount of the compound represented by the general formula (1), general formula (II), or general formula (III) contained in the brain of the model animal Of evaluation of therapeutic or prophylactic drugs.
- Compounds represented by general formula (I), general formula (II), and general formula (III) are amyloid j8 It has high binding specificity to proteins, has high blood-brain barrier permeability, and rapidly disappears from sites other than senile plaques in the brain. Useful.
- FIG. 1 is a diagram showing a synthesis method (1) of a flavone derivative (numbers in the figure indicate compound numbers).
- FIG. 2 is a diagram showing a synthesis method (2) of a flavone derivative (numbers in the figure indicate compound numbers).
- FIG. 3 is a diagram showing a synthesis method (3) of a flavone derivative (numbers in the figure indicate compound numbers).
- FIG. 4 shows a method for synthesizing chalcone derivatives (numbers in the figure indicate compound numbers).
- FIG. 5 is a diagram showing a synthesis method (1) of a styrylchromone derivative (numbers in the figure indicate compound numbers) Show).
- FIG. 6 shows a synthesis method (2) of a styrylchromone derivative (numbers in the figure indicate compound numbers).
- FIG. 7 shows a method for synthesizing coumarin derivatives (numbers in the figure indicate compound numbers).
- FIG. 8 is a diagram showing a method for labeling flavones, chalcones, styrylchromones, and coumarin derivatives with radioactive iodine.
- FIG. 9 Diagram showing the binding properties of Compound 10 (A), Compound 11 (B), and Compound 19 (C) to A
- FIG. 10 is a graph showing the binding properties of Compound 31 ( ⁇ ) and Compound 36 ( ⁇ ) to ⁇ aggregates ( ⁇ : in the presence of A ⁇ 8 aggregate, country: in the absence of A j8 aggregate).
- FIG. 11 Diagram showing the binding properties of Compound 46 (A), Compound 47 (B), and Compound 48 (C) to A ⁇ aggregates ( ⁇ : ⁇ ⁇ aggregates in the presence of ⁇ ⁇ aggregates In the absence).
- FIG. 12 is a diagram showing a Scatchyard plot obtained by a saturation experiment of Compound 11 with ⁇
- FIG. 13 A photograph showing the binding state of Compound 11 to senile plaque amyloid in Arno and Imah's disease brain tissue (using a 40x objective lens and G filter). Compound 11 (appears white) is bound to amyloid senile plaque (middle part) and cerebrovascular amyloid (lower left).
- FIG. 14 A photograph showing the binding state of Compound 11 to senile plaque amyloid in Arno and Imah's disease brain tissue (X using a 20 ⁇ objective lens, using a G filter). Compound 11 (appears white) binds to cerebrovascular amyloid (four near the center, one in the upper left).
- the “halogen atom” is, for example, a fluorine atom, a chlorine atom, a bromine atom, or an iodine atom.
- the “alkyl group having 1 to 4 carbon atoms” means, for example, methyl group, ethyl group, propyl group, isopropyl group, butyl group, isobutyl group, sec-butyl group, tert-butyl group is there.
- the “C 1-4 alkoxy group” means, for example, a methoxy group, an ethoxy group, a propoxy group, an isopropoxy group, a butoxy group, an isobutoxy group, a sec-butoxy group, a ter t-butoxy It is a group.
- the “aryl group” is, for example, a phenyl group, a 1 naphthyl group, a 2 naphthyl group or the like.
- the “aromatic heterocyclic group” includes, for example, thiophen-2-yl group, thiophen-3-yl group, furan-2-yl group, furan-3-yl group, pyridine
- Examples include 2-yl group, pyridine-3-yl group, pyridine-4-yl group, pyrimidine-2-yl group, pyrimidine-4-yl group, and pyrimidine-5-yl group.
- the “aryl group optionally substituted by a substituent selected from substituent group A” includes, for example, a 2-dimethylaminophenol group, a 2-methylaminophenol group, 2-methoxyphenyl group, 2-hydroxyphenyl group, 3-dimethylaminophenol group, 3-methylaminophenol group, 3-methoxyphenol group, 3-hydroxyphenyl group, 4-dimethyl group Tyraminophenol group, 4-methylaminophenyl group, 4-methoxyphenyl group, 4-hydroxyphenyl group, 3-hydroxy-1-4-dimethylaminophenol group, 3-hydroxy-1-methylaminophenol Group, 3-hydroxy-4-methoxyphenyl group, 3,5-dihydroxy-4-dimethylaminophenol group, 3,5-dihydroxy-4-methylaminophenol group, 3,5-dihydroxy-4 -Methoxyphenyl group etc.
- the “aromatic heterocyclic group optionally substituted by a substituent selected from substituent group A” includes, for example, 5 dimethylaminopyridine-2-yl group, 5 methyl 2-amino group of aminoviridine, 2-yl group of 5-methoxypyridine, 3-yl group of 6-dimethylaminopyridine, 3-yl group of 6-methylaminopyridine, 3-yl group of 6-methoxypyridine And the like.
- R 4 is preferably a nitrogen atom, and more preferably one is an iodine atom.
- R 5 is preferably a halogen atom, a hydroxyl group, a carboxyl group, a sulfone group, a dimethylamino group, a methylamino group, an amino group, a nitro group, or an alkyl having 1 to 4 carbon atoms.
- R 6 is a halogen atom, a hydroxyl group, a carboxyl group, a sulfone group, a dimethylamino group, a methylamino group, an amino group, a nitro group, an alkyl group having 1 to 4 carbon atoms, and an alkoxy group having 1 to 4 carbon atoms.
- Group power as a basic force represents a phenol group which may be substituted with one or more selected substituents.), More preferably 4-dimethylaminophenol group, 4- Methylaminophenol group, 4-methoxyphenyl group, 4 A hydroxyphenyl group, a 4-aminostyryl group, a 4-methylaminostyryl group, or a 4-dimethylaminostyryl group;
- the position of R 5 on the chromone may be either the 2nd or 3rd position.
- R 7 is preferably a halogen atom, a hydroxyl group, a carboxyl group, a sulfone group, a dimethylamino group, a methylamino group, an amino group, a nitro group, or an alkyl having 1 to 4 carbon atoms.
- R 8 is preferably a halogen atom, a hydroxyl group, a carboxyl group, a sulfone group, a dimethylamino group, a methylamino group, an amino group, a nitro group, or a C 1-4 alkyl.
- Alkyl group, and group strength consisting of alkoxy groups having 1 to 4 carbon atoms.
- a phenol group which may be substituted with one or more selected substituents, more preferably 4-methylaminophenol. Or a 4-dimethylaminophenol group.
- ⁇ ⁇ represents a methyl group
- Phe represents a phenol group
- XI represents a 2-fluoroethoxy group
- X2 represents a 3 fluoropropoxy group
- X3 represents a 4 fluorobutoxy group
- X4 represents a 5-fluoropentoxy group.
- H H I H 2-CH CH- (6- NHMe- pyridine- 3- yl>
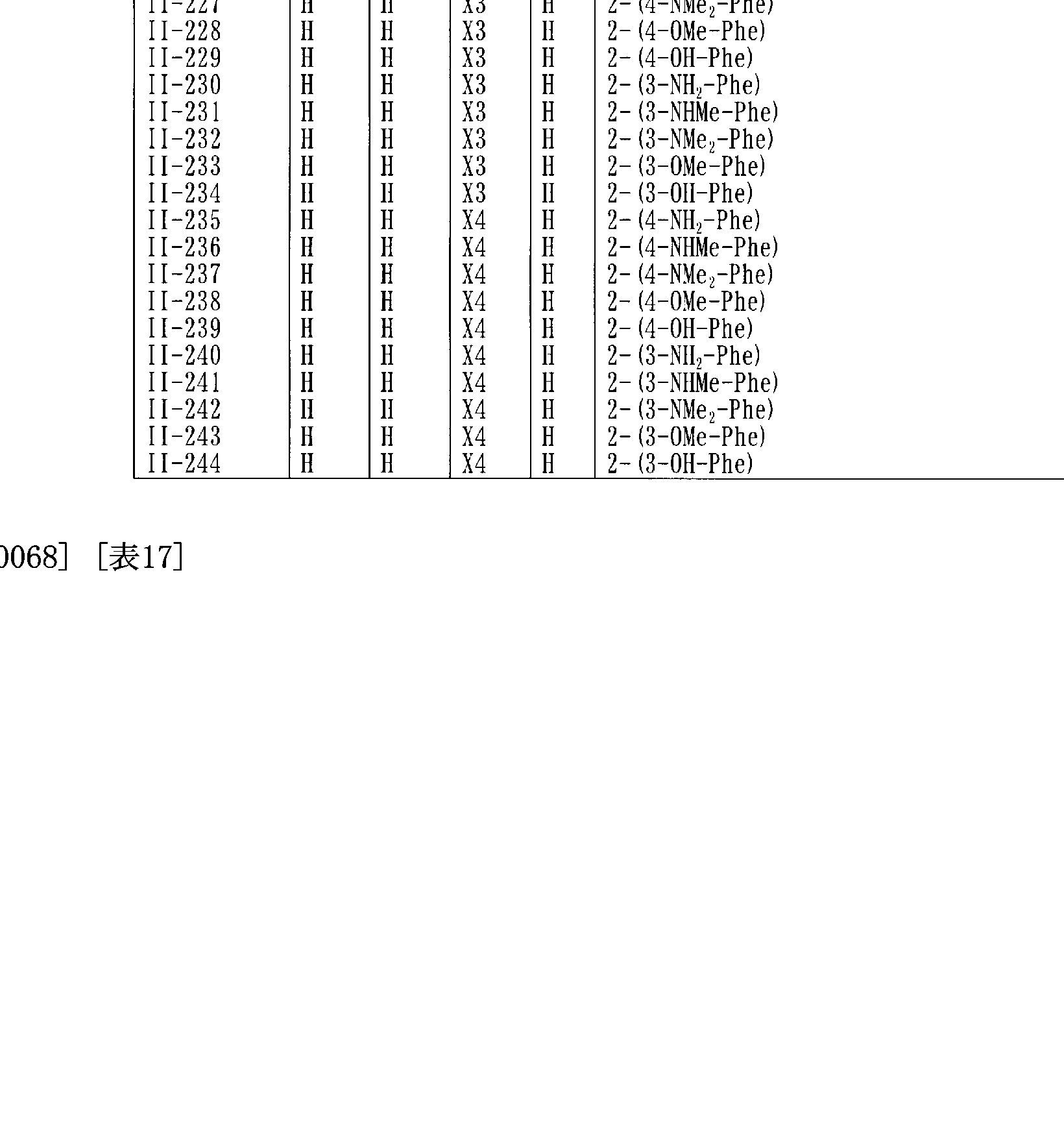
- Tables 10 to 20 show typical compounds represented by the general formula (II).
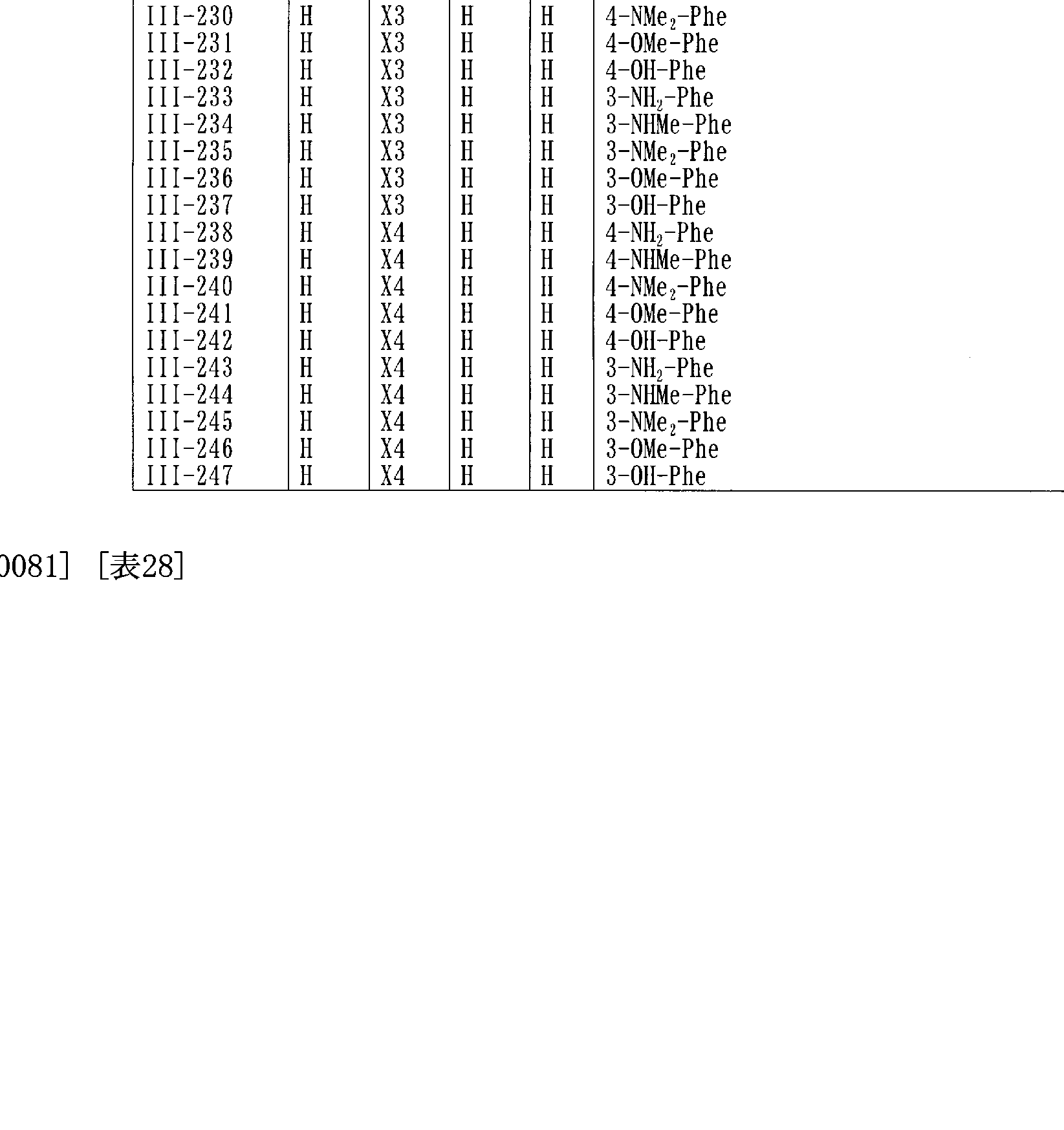
- Table 21 to Table 28 show representative compounds of the general formula (III).
- preferable examples include the compound of III-1 (compound 65), the compound of ⁇ -2 (compound 66), and the compound of ⁇ -3 (compound 67). Can do.
- the compound represented by the general formula (I) or the like is preferably labeled with a labeling substance.
- a labeling substance a fluorescent substance, a facility substance or the like may be used, but it is preferable to use a radionuclide.
- the type of radionuclide used for labeling is not particularly limited, and can be appropriately determined depending on the mode of use.
- SPECT computed tomography
- the radionuclide is 99 m Tc, i In, 67 Ga, 201 T1, 123 ⁇ , 133 Xe (preferably 99m Tc, 12 3 ⁇ 4, etc. can be used a line-emitting nuclides.
- n C ⁇ 13 n, 15 0, 18 F , 62 Cu, 68 Ga ⁇ 76 Br preferably in that, when administered to ⁇ 13 N, 15 0, 18 F
- PET positron emission tomography
- the general formula (I) the animal a compound other than human represented by or the like, A radionuclide having a longer half-life, for example, 125 ⁇ may be used, and the radionuclide may be in a form contained in the molecule of the compound represented by the general formula (I) or the like. It may be in the form of binding to a compound represented by I) or the like.
- a pharmaceutically acceptable salt may be used in place of the compound represented by the general formula (I) and the like.
- pharmaceutically acceptable salts include alkali metal salts (sodium salt, potassium salt, lithium salt), alkaline earth metal salts (calcium salt, magnesium salt), sulfate salt, hydrochloride salt, nitrate salt, phosphate salt, etc. it can.
- amyloid-related disease refers to a disease caused by the accumulation of amyloid j8 protein, which mainly means Alzheimer's disease. With amyloidosis 1 Dutch type: H and HWA—D) are also included.
- precursor symptoms of diseases that are generally not recognized as “diseases” are also included in “amyloid-related diseases” in the present invention. Examples of the prodromal symptoms of such diseases include mild cognitive impairment (MCI), which is observed before the onset of Alzheimer's disease.
- MCI mild cognitive impairment
- Diagnosis of an amyloid-related disease with the composition of the present invention is usually performed by administering the composition of the present invention to a diagnosis subject or a laboratory animal, and then taking a brain image, and the general formula (I ) Etc., based on the state of the compound (quantity, distribution, etc.).
- the administration method of the composition of the present invention is not particularly limited and can be appropriately determined according to the type of compound, the type of labeling substance, etc., but is usually intradermal, intraperitoneal, vein, artery, or spinal cord. Administer by liquid injection or infusion.
- the dosage of the composition of the present invention is not particularly limited and can be determined appropriately according to the type of compound, the type of labeling substance, etc.
- the yarn and composition of the present invention are usually administered by injection or infusion, and therefore may contain components usually contained in injection or infusion.
- components include liquid carriers (eg, potassium phosphate buffer, physiological saline, Ringer's solution, distilled water, polyethylene glycol, vegetable oils, ethanol, glycerin, dimethyl sulfoxide, and propylene glycol).
- Antibacterial agents eg, pro-chlorine hydrochloride, dibu-hydrochloride
- buffers eg, tris-HCl buffer, hepes buffer
- osmotic pressure regulators eg, glucose, Sorbitol, sodium chloride, etc.
- the composition for diagnosing amyloid-related diseases of the present invention can also be used for screening for therapeutic or prophylactic agents for amyloid-related diseases.
- a test substance is administered to a model animal of “disease” such as Alno-i-maima disease
- the model animal is administered with the composition for diagnosing amyloid-related disease of the present invention, and then the brain of the model animal is The distribution or amount of the compound represented by the general formula (I) contained therein is examined, and as a result, a significant difference from the control (model animal not administered with the test substance) (for example, the distribution site).
- the test substance can be a candidate for a therapeutic drug for amyloid-related diseases.
- the composition for amyloid-related disease diagnosis of the present invention is administered to the model animal.
- the distribution or amount of the compound represented by general formula (I), etc. contained in the brain is examined, and as a result, there is a significant difference from the control (for example, the slowness or expansion of the distribution site, the amount of reduction, or the amount). If a decrease or decrease in increase is detected, the test substance can be a candidate for a prophylactic agent for amyloid-related diseases.
- the composition for diagnosing amyloid-related diseases of the present invention can also be used for evaluation of therapeutic and prophylactic agents for amyloid-related diseases whose effects have already been confirmed. That is, after the therapeutic or prophylactic agent for the disease is administered to a model animal for amyloid-related disease, the composition for diagnosing amyloid-related disease of the present invention is administered to the model animal, and then into the brain of the model animal. The distribution or amount of the compound represented by the general formula (I) and the like contained therein is examined, and thereby the therapeutic agent and the preventive agent are evaluated (specifically, effective dose, effective administration method, etc.) .
- Example 1 Example 1
- Radioactive iodine-125 was IODINE-125 (74 MBq) manufactured by Amersham Biosciences. Reversed phase HPLC was performed by Nacalai Testa Cosmosil 5C-AR column (4.6 x 150
- a black mouth form solution (87 mL) of compound 17 (450 mg, 0.831 mmol) was charged with a black mouth form solution (5 mL, 1 M) at room temperature. After reacting for 30 minutes at room temperature, saturated aqueous sodium hydrogensulfite solution (30 mL) was added to complete the reaction. The chloroform layer is separated, dried over sodium sulfate, the solvent is distilled off under reduced pressure, and the residue is subjected to silica gel chromatography using ethyl acetate Z-hexane (1 I 7) as an elution solvent. 6-yodo-4, -methoxyflavone (compound 19) was obtained. Yield 227 mg (Yield: 72.2%) NMR (300 MHz, CDC1) ⁇ 8.55 (s,
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Physics & Mathematics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Epidemiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Urology & Nephrology (AREA)
- Optics & Photonics (AREA)
- Pathology (AREA)
- Immunology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Hematology (AREA)
- Molecular Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Gastroenterology & Hepatology (AREA)
- Toxicology (AREA)
- Rheumatology (AREA)
- Analytical Chemistry (AREA)
- Endocrinology (AREA)
- Diabetes (AREA)
- Microbiology (AREA)
- General Physics & Mathematics (AREA)
- Cell Biology (AREA)
- Biotechnology (AREA)
- Food Science & Technology (AREA)
- Biochemistry (AREA)
- Zoology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/666,507 US8192717B2 (en) | 2004-11-26 | 2005-11-25 | Composition for diagnosing amyloid-related diseases |
| JP2006520584A JP4000353B2 (ja) | 2004-11-26 | 2005-11-25 | アミロイド関連疾患診断用組成物 |
| EP05809739A EP1815872A4 (en) | 2004-11-26 | 2005-11-25 | COMPOSITION FOR THE DIAGNOSIS OF AMYLOID ASSOCIATED DISEASE |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004-341370 | 2004-11-26 | ||
| JP2004341370 | 2004-11-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006057323A1 true WO2006057323A1 (ja) | 2006-06-01 |
Family
ID=36498057
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2005/021642 WO2006057323A1 (ja) | 2004-11-26 | 2005-11-25 | アミロイド関連疾患診断用組成物 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US8192717B2 (ja) |
| EP (1) | EP1815872A4 (ja) |
| JP (1) | JP4000353B2 (ja) |
| WO (1) | WO2006057323A1 (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009072417A1 (ja) * | 2007-12-07 | 2009-06-11 | Nagasaki University | プリオン病診断用組成物 |
| EP2171478A1 (en) * | 2007-03-30 | 2010-04-07 | The Regents Of The University Of California | In vivo imaging of sulfotransferases |
| US8557222B2 (en) | 2008-04-04 | 2013-10-15 | Avid Radiopharmaceuticals, Inc. | Radiopharmaceutical imaging of neurodegenerative diseases |
| CN109293616A (zh) * | 2018-09-29 | 2019-02-01 | 贵州大学 | 一种含香豆素查尔酮类衍生物、其制备方法及应用 |
| WO2019168170A1 (ja) * | 2018-03-02 | 2019-09-06 | 国立大学法人 長崎大学 | クロモン誘導体及びアミロイド関連疾患診断用組成物 |
| JP2020066619A (ja) * | 2018-04-04 | 2020-04-30 | 株式会社Cics | アミロイドβ疾患のホウ素中性子捕捉療法用化合物 |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2706566A1 (en) * | 2007-12-18 | 2009-07-09 | Glenmark Pharmaceuticals, S.A. | Chromane derivatives as trpv3 modulators |
| US8318132B2 (en) | 2008-02-14 | 2012-11-27 | Siemens Medical Solutions Usa, Inc. | Imaging agents for detecting neurological dysfunction |
| US8932557B2 (en) | 2008-02-14 | 2015-01-13 | Eli Lilly And Company | Imaging agents for detecting neurological dysfunction |
| EP2112145A1 (en) * | 2008-04-24 | 2009-10-28 | AxoGlia Therapeutics S.A. | Chromenone derivatives useful for the treatment of neurodegenerative diseases |
| MX2010011762A (es) * | 2008-06-17 | 2011-02-03 | Glenmark Pharmaceuticals Sa | Derivados de cromano como moduladores del trpv3. |
| EP2323697A2 (en) | 2008-07-24 | 2011-05-25 | Siemens Medical Solutions USA, Inc. | Imaging agents useful for identifying ad pathology |
| EP2910945B1 (en) * | 2008-09-30 | 2018-07-18 | Case Western Reserve University | Molecular probes for imaging of myelin |
| ES2343347B2 (es) * | 2009-01-27 | 2011-12-07 | Universidade De Santiago De Compostela | Uso de derivados de 3-fenilcumarinas 6-sustituidas y preparacion de nuevos derivados. |
| CN103739605B (zh) | 2009-03-23 | 2016-08-17 | 伊莱利利公司 | 用于检测神经障碍的显像剂 |
| US8691187B2 (en) | 2009-03-23 | 2014-04-08 | Eli Lilly And Company | Imaging agents for detecting neurological disorders |
| US20140079635A1 (en) * | 2011-03-30 | 2014-03-20 | Case Western Reserve University | Molecular probes for detecting lipid composition |
| WO2016097339A1 (en) * | 2014-12-19 | 2016-06-23 | Ge Healthcare Limited | Labelled coumarin derivatives |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003074519A1 (en) * | 2002-03-06 | 2003-09-12 | Novartis Ag | Coumarines useful as biomarkers |
| WO2004069774A2 (en) * | 2003-02-04 | 2004-08-19 | The University Court Of The University Of St Andrews | Synthesis of 13c-labelled estrogen analogues |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU759945B2 (en) | 1998-08-20 | 2003-05-01 | Regents Of The University Of California, The | Methods for labeling beta-amyloid plaques and neurofibrillary tangles |
-
2005
- 2005-11-25 US US11/666,507 patent/US8192717B2/en not_active Expired - Fee Related
- 2005-11-25 WO PCT/JP2005/021642 patent/WO2006057323A1/ja active Application Filing
- 2005-11-25 JP JP2006520584A patent/JP4000353B2/ja not_active Expired - Fee Related
- 2005-11-25 EP EP05809739A patent/EP1815872A4/en not_active Withdrawn
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003074519A1 (en) * | 2002-03-06 | 2003-09-12 | Novartis Ag | Coumarines useful as biomarkers |
| WO2004069774A2 (en) * | 2003-02-04 | 2004-08-19 | The University Court Of The University Of St Andrews | Synthesis of 13c-labelled estrogen analogues |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1815872A4 * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2171478A1 (en) * | 2007-03-30 | 2010-04-07 | The Regents Of The University Of California | In vivo imaging of sulfotransferases |
| EP2171478A4 (en) * | 2007-03-30 | 2013-01-30 | Univ California | IN VIVO IMAGING METHOD FOR SULPHOTRANSFERASES |
| WO2009072417A1 (ja) * | 2007-12-07 | 2009-06-11 | Nagasaki University | プリオン病診断用組成物 |
| US8557222B2 (en) | 2008-04-04 | 2013-10-15 | Avid Radiopharmaceuticals, Inc. | Radiopharmaceutical imaging of neurodegenerative diseases |
| WO2019168170A1 (ja) * | 2018-03-02 | 2019-09-06 | 国立大学法人 長崎大学 | クロモン誘導体及びアミロイド関連疾患診断用組成物 |
| JP2020066619A (ja) * | 2018-04-04 | 2020-04-30 | 株式会社Cics | アミロイドβ疾患のホウ素中性子捕捉療法用化合物 |
| JP7204568B2 (ja) | 2018-04-04 | 2023-01-16 | 株式会社Cics | アミロイドβ疾患のホウ素中性子捕捉療法用化合物 |
| CN109293616A (zh) * | 2018-09-29 | 2019-02-01 | 贵州大学 | 一种含香豆素查尔酮类衍生物、其制备方法及应用 |
| CN109293616B (zh) * | 2018-09-29 | 2022-06-21 | 贵州大学 | 一种含香豆素查尔酮类衍生物、其制备方法及应用 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20080131367A1 (en) | 2008-06-05 |
| JPWO2006057323A1 (ja) | 2008-06-05 |
| EP1815872A1 (en) | 2007-08-08 |
| US8192717B2 (en) | 2012-06-05 |
| JP4000353B2 (ja) | 2007-10-31 |
| EP1815872A4 (en) | 2011-06-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2006057323A1 (ja) | アミロイド関連疾患診断用組成物 | |
| TWI228120B (en) | Process for making diaryl pyridines useful as COX-2 inhibitors | |
| TWI406856B (zh) | 人類免疫缺乏病毒複製之抑制劑 | |
| CN102046638B (zh) | 检测和治疗阿尔茨海默病和相关疾病的化合物和组合物 | |
| WO2007136059A1 (ja) | アミロイド関連疾患診断のための組成物 | |
| US20120263646A1 (en) | Imaging agents and their use for the diagnostic in vivo of neurodegenerative diseases, notably alzheimer's disease and derivative diseases | |
| Ono et al. | Novel chalcones as probes for in vivo imaging of β-amyloid plaques in Alzheimer’s brains | |
| JP2008525363A (ja) | プロスタグランジン媒介疾患治療のためのピリジン化合物 | |
| KR101530392B1 (ko) | 신규 2-헤테로아릴 치환 벤조티오펜 및 벤조푸란 709 | |
| EP3793992B1 (en) | Matrix metalloproteinase (mmp) inhibitors and methods of use thereof | |
| WO2019089665A1 (en) | Alkene spirocyclic compounds as farnesoid x receptor modulators | |
| WO2019089664A1 (en) | Multicyclic compounds as farnesoid x receptor modulators | |
| US20140296534A1 (en) | Compounds suitable as precursors to compounds that are useful for imaging amyloid deposits | |
| US20200115357A1 (en) | Liver x receptors (lxr) modulators | |
| TWI764977B (zh) | 作爲caspase抑制劑的聯環化合物、含有其的藥物組合物及其應用 | |
| JPH0354945B2 (ja) | ||
| AU583847B2 (en) | Use of 4-(isoxazolyl)-thiazole-2-oxamic acid derivatives | |
| WO2008068974A1 (ja) | オーロン誘導体含有診断用組成物 | |
| KR20210126614A (ko) | 3-브로모-5-(2-에틸이미다조[1,2-a]피리딘-3-카르보닐)-2-하이드록시벤조니트릴의 합성 | |
| JP2007084526A (ja) | アミロイド関連疾患診断用組成物 | |
| JP2884094B2 (ja) | 新規なピリドンカルボン酸またはその塩の可溶化法 | |
| JPH05208959A (ja) | ベンゾ[cフェナンスリジニューム誘導体の製造法及びその新規化合物 | |
| JP2550631B2 (ja) | ピリダジノン誘導体 | |
| JP4000352B2 (ja) | アミロイド関連疾患診断用組成物 | |
| CH634071A5 (en) | Therapeutically active pyranoquinolones |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KN KP KR KZ LC LK LR LS LT LU LV LY MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU LV MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006520584 Country of ref document: JP |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005809739 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005809739 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11666507 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 11666507 Country of ref document: US |