WO2004106276A1 - 縮合環化合物 - Google Patents
縮合環化合物 Download PDFInfo
- Publication number
- WO2004106276A1 WO2004106276A1 PCT/JP2004/007770 JP2004007770W WO2004106276A1 WO 2004106276 A1 WO2004106276 A1 WO 2004106276A1 JP 2004007770 W JP2004007770 W JP 2004007770W WO 2004106276 A1 WO2004106276 A1 WO 2004106276A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- ring
- compound
- methyl
- yield
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/73—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids
- C07C69/734—Ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/64—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/64—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings
- C07C59/66—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings the non-carboxylic part of the ether containing six-membered aromatic rings
- C07C59/68—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings the non-carboxylic part of the ether containing six-membered aromatic rings the oxygen atom of the ether group being bound to a non-condensed six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/72—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings and other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/38—Nitrogen atoms
- C07D277/42—Amino or imino radicals substituted by hydrocarbon or substituted hydrocarbon radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/02—Systems containing only non-condensed rings with a three-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/10—One of the condensed rings being a six-membered aromatic ring the other ring being six-membered, e.g. tetraline
Definitions
- the present invention relates to a novel fused ring compound having a GPR40 receptor function regulating action.
- Alkanic acid derivatives are known (JP-A-2002-265457).
- Isoxazole derivatives having an insulin secretion promoting action and a blood glucose lowering action and useful for the prevention and treatment of diabetes and the like are known (Japanese Patent Application Laid-Open No. 2002-212171).
- a nitrogen-containing 5-membered heterocyclic compound having a blood glucose lowering effect and a blood lipid lowering effect and useful for the prevention and treatment of diabetes and the like is known (Japanese Patent Application Laid-Open No. 2001-2263: 50).
- An oximininoal carboxylic acid derivative having a blood glucose lowering effect and a blood lipid lowering effect and useful for prevention and treatment of diabetes and the like is known (Japanese Patent Application Laid-Open No. 2000-198772).
- a 1,3-azole derivative which has a retinoid-related receptor function regulating action and is useful for prevention and treatment of diabetic complications and the like is known (JP-A-2000-80086).
- An iminoalkanoic acid derivative which has a blood glucose lowering effect and a blood lipid lowering effect and is useful for the prevention and treatment of diabetes and the like is known (JP-A-2000-34266).
- An oxazole derivative which has an insulin secretion promoting effect and a blood glucose lowering effect and is useful for the prevention and treatment of diabetes and the like is known (JP-A-09-323983).
- a benzofuran derivative having a blood sugar and blood lipid lowering action is known (Japanese Patent Publication No. 08-311065).
- An object of the present invention is to provide a novel fused ring compound which has a GPR40 receptor function regulating action and is useful as an insulin secretagogue or a prophylactic / therapeutic agent for diabetes mellitus and the like.
- the present inventors have conducted various studies and found that the compound represented by the following formula (I) has unexpectedly excellent GPR40 receptor agonist activity based on its specific chemical structure. It also has excellent properties as pharmaceuticals such as stability, and has been found to be a safe and useful pharmaceutical as a therapeutic agent for the prevention or treatment of mammalian GPR40 receptor-related conditions or diseases.
- the present invention has been completed based on these findings.
- Ar represents an optionally substituted cyclic group
- Ring A is an optionally substituted ring (provided that the ring is not thiol, oxazole, imidazole or pyrazole)
- Xa and Xb are each independently a spacer having 1 to 5 bonds or a main chain.
- Xc is 0, S, SO or S0 2
- Ring B is a 5- or 7-membered ring
- Xd is a bond, CH or CH 2 ′
- R 1 represents an optionally substituted hydroxy group.
- Ar 1 represents an optionally substituted phenyl group or an optionally substituted indanyl group
- Xa 1 represents a bond or a spacer having 1 to 5 atoms in the main chain
- Ring A 2 is a benzene ring which may be further substituted
- Ring B 2 represents a 5- to 7-membered ring.
- Ar 2 represents an optionally substituted thiazolyl group
- Xa 2 is a bond having a bond or a main chain having 1 to 5 atoms
- Ring B 2 represents a 5- to 7-membered ring.
- an insulin secretagogue comprising compound (I) or a prodrug thereof;
- ring A 1 represents an optionally substituted ring
- Xb is a bond having a bond or a main chain having 1 to .5 atoms
- Xc and is 0, S ,. SO or S0 2,
- Ring B 1 is a 5- to 7-membered non-aromatic ring
- Xd is a bond, CH or CH 2 ,
- R 1 represents an optionally substituted hydroxy group. Or a salt thereof (hereinafter sometimes abbreviated as compound (1 ′)) or a GPR40 receptor function modulator comprising a prodrug thereof;
- halogen atom in the present specification includes a fluorine atom, a chlorine atom, a bromine atom and an iodine atom unless otherwise specified.
- optionally substituted hydrocarbon group in the present specification, unless otherwise specified, for example, “optionally substituted alkyl group”, “optionally substituted C 2 one 6 alkenyl group ",” optionally substituted c 2 _ 6 alkynyl group ",
- CI_ 6 alkyl group in the present specification, unless otherwise specified, for example methylation, Echiru, propyl, isopropyl, heptyl, Isopuchiru, sec- heptyl, tert - heptyl, Penchinore, Isopenchinore, neopentyl, to Kishinore Etc.
- C 2 6 alkynyl group for example, 2-leptin one 1 Inore, 4-pentyne one 1 over I / Les, relaxin like one 1 Inore to 5- Can be .
- the - "14 Ariru group C 6" in the present specification for example phenyl, 1 one-naphthyl, 2-naphthyl, 2-Bifue two Lil, 3 Bifue - herein Lil, 4-bifenyl and 2-anthryl can be fisted.
- the C 6 - 1 4 Ariru may be partially saturated, C 6 partially saturated - The 1 4 7 reel, for example, tetrahydronaphthyl and the like.
- the “hydroxy group which may be substituted” in the present specification includes, for example, “hydroxy group”, “optionally substituted C i-i. Alkoxy group”, “ substituted heterocyclic Okishi group “,” optionally substituted c 6 - 1 4 Ariruokishi group ",” optionally substituted c 7 - 1 6 Ararukiruokishi group "of ⁇ the like. '
- the “0 ⁇ 6 alkoxy group” in the present specification includes, for example, methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy, tert-butoxy, pentinoleoxy, hexyloxy and the like, unless otherwise specified.
- alkoxy group examples include heptyloxy, octyloxy, noeroxy, decyloxy and the like in addition to the above-mentioned alkoxy group.
- alkoxy- 16 alkoxy group examples include, for example, methoxymethoxy, methoxyethoxy, ethoxymethoxy, ethoxyethoxy and the like, unless otherwise specified.
- heterocyclic oxy group in the present specification, a hydroxy group substituted by a “heterocyclic group” described below can be mentioned.
- the heterocyclic oxy group include tetrahydrobilanyloxy, thiazolyloxy, pyridyloxy, bilazolyloxy, oxazolyloxy, cheeroki, //, and furyloxy.
- Examples of the “optionally substituted mercapto group” in the present specification include, unless otherwise specified, for example, “mercapto group”, “optionally substituted alkylthio group”, “substituted also heterocyclic Chio group “,” optionally substituted C 6 - 1 4 Ariruchio group ",” optionally substituted C 7 - etc. 1 6 Ararukiruchio group "can be mentioned.
- re- 6 alkylthio group in the present specification, unless otherwise specified, for example, methylthio, ethylthio, propylthio, isopropylthio, butylthio, sec-butylthio, tert-butylthio and the like can be mentioned.
- C. alkylthio group in the present specification includes, in addition to the above-mentioned alkylthio group, heptylthio, octylthio, nonylthio, decylthio and the like.
- heterocyclic thio group in the present specification, a mercapto group substituted by the below-mentioned “heterocyclic group” can be mentioned.
- the heterocyclic thio group include tetrahydropyranylthio, thiazolylthio, pyridylthio, virazolylthio, oxazolylthio, chenylthio, furylthio and the like.
- C 6 1 4 Ariruchio group eg if phenylene thioether, 1 one-naphthylthio, 2-naphthylthio.
- C 7 _ 16 aralkylthio group in the present specification includes, for example, benzylthio, phenethylthio and the like unless otherwise specified. .,
- heterocyclic group means, unless otherwise specified, for example, as a ring-constituting atom, one or two selected from a nitrogen atom, a sulfur atom, and an oxygen atom in addition to a carbon atom; 5 hetero-atoms (monocyclic, bicyclic or tricyclic) heterocyclic groups, preferably (i) 5- to 14-members (preferably 5- to 10-members) that eat four heteroatoms A heterocyclic group; and (ii) a 5- to 10-membered non-aromatic heterocyclic group. Among them, a 5- or 6-membered aromatic heterocyclic group is preferable.
- chenyl eg, 2-chel, 3- chenyl
- full eg, 2-frill, 3-frill
- Pyridyl example: 2-pyridyl, 3-pyridyl, 4-pyridyl
- thiazolyl example: 2-thiazolyl, 4-thiazolyl, 5-thiazolyl
- oxazolyl example: 2-oxazolyl, 4-oxazolyl, 5-oxazolyl
- Biradizyl pyrimidinyl (eg, 2-pyrimidinyl, 4-pyrimidinyl), pyrrolyl (eg, 1-pyrrolyl, 2-pyrrolyl, 3-pyrrolyl), imidazolyl (eg, 1-imidazolyl, 2-imidazolyl, 4 1-imidazolyl), pyrazolyl (eg, 1-pyrazolyl, 3-birazolyl, 4-birazolyl), triazolyl (eg, 1-triazolyl
- 0 ⁇ 6 alkyl monoalkyl group is used unless otherwise specified.
- acetyl, isobutanoyl, isopentanoyl and the like can be used.
- 0 ⁇ 6 alkoxy-carboxy group includes, for example, methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, tert-butoxycarbonyl and the like, unless otherwise specified. .
- nitrogen-containing heterocyclic monocarbonyl group includes, for example, pyrrolidinylcarbonyl, piperidinocarbonyl and the like, unless otherwise specified.
- rC i-e alkylsulfonyl group for example, methylsulfonyl, ethylsulfonyl and the like are mentioned unless otherwise specified.
- ⁇ 6 alkylsulfinyl group specifically mentioned no limit is, for example, methylsulfinyl, E chill sulfide El, and the like.
- the “optionally esterified carbonyl group” in the present specification includes, for example, a carboxyl, 6 alkoxy-carbonyl group (eg, methoxycarbyl, ethoxycarbonyl, propoxycarbonyl, tert-butoxycarbonyl) - le, etc.), C 6 _ 14 Ariruokishi one carbonyl group (e.g., phenoxy Karuboyuru etc.), C 7 - 16 Ararukiruokishi one carbonyl group (e.g. Benjiruoki aryloxycarbonyl, Hue phenethyl Ruo propoxycarbonyl sulfonyl, etc.) and the like.
- 6 alkoxy-carbonyl group eg, methoxycarbyl, ethoxycarbonyl, propoxycarbonyl, tert-butoxycarbonyl
- C 6 _ 14 Ariruokishi one carbonyl group e.g., phenoxy Karuboyur
- alkyl group which may be halogenated herein, unless otherwise specified in particular, but it may also be substituted with 1 to 5 of the above-mentioned "halogen atom” above ". 6 alkyl group ].
- halogen atom 1 to 5 of the above-mentioned "halogen atom” above ". 6 alkyl group ].
- methyl, ethyl, propyl, isopropyl, butyl, tert-butyl, isoptyl, trifluoromethyl and the like can be mentioned.
- the “Ci- 6 alkoxy group which may be halogenated” in the present specification includes the above-mentioned “0 ⁇ 6” which may be substituted with 1 to 5 of the above “halogen atoms”.
- Alkoxy group ".
- methoxy, ethoxy, isopropoxy, tert-butoxy, trifluoromethoxy and the like can be mentioned.
- methylamino, ethylamino, propylamino, dimethylamino, getylamino and the like can be mentioned.
- benzylamino, phenethylamino and the like can be mentioned.
- N-C 6-alkyl one N- C 6 - 1 4 Ariruamino group is a, unless otherwise noted, the re - 6 alkyl group "and the" C 6 - 1 4 ⁇ rie And an amino group substituted with a phenyl group.
- N-methyl-N-phenylamino, N-ethyl-1-N-phenylamino and the like can be mentioned. .
- the ⁇ mono- or di-C 6 alkyl monofunctional rubamoyl group '' in the present specification includes a mono- or di-monosubstituted functional lpamoyl group with the above ⁇ Ci- 16 alkyl group ''.
- methylcarbamoyl, ethylcarbamoyl, dimethylcarbamoyl, getylcarbamoyl, ethylmethylcarbamoyl, and the like can be mentioned.
- the term ⁇ mono- or di-mono- to 5-membered heterocyclic mono-lupamoyl group '' refers to a mono- or di-substituted mono- or di-substituted mono- or di-substituted lubamoyl group unless otherwise specified.
- the substituent include a heterocyclic group containing one or two or one to four hetero atoms selected from a nitrogen atom, a sulfur atom and an oxygen atom in addition to a carbon atom.
- Preferable examples of the “mono- or di- to 5-membered heterocyclic mono-lupamoyl group” include 2-pyridylcarbamoyl, 3-pyridylcarbamoyl, 4-pyridylcarbamoyl, 2-phenyldilpamoyl, and 3-chelidyl. Lucarpamoyl and the like.
- a di-substituted sulfamoinole group is used, and examples thereof include phenylsulfamoyl, diphenylsulfamoyl, 1-naphthinoresnolefamoinole, and 2-naphthylsulfamoyl.
- Ci-6 alkyl group a mono one or di- C - 6 Arukiruamino group, C 6 - 14 Ariru group, mono- one or di- one C 6 - 14 Arirua amino group, C 3 _ 8 cycloalkyl Alkyl group, alkoxy group,.
- a halogen atom arsenic Dorokishi group, an amino group, a nitro group, shea Amino group, Nono halogenated which may be optionally CI- 6 alkyl group, mono- or di-one C i-6 alkyl primary amino groups, C 6 _ 14 Ariru group, mono- one or di- one C 6 - 14 Ariruamino groups, C . 3 _ 8 cycloalkyl group, Ji 6 alkoxy groups, 6 A Kokishi -Ci- 6 alkoxy group, C - 6 alkylthio group, C -.
- Alkoxy -. 6 comprises an alkoxy group) "and includes" 10 alkyl thio group (Ci-e alkylthio group) "can be mentioned.
- “optionally substituted C 3 - 8 cycloalkyl group”',”optionally substituted C 6 - 14 Ariru group”, “substituted C 7 _ 16 optionally 7 aralkyl group “,” optionally substituted heterocyclic group “,” may be substituted heterocycle O key sheet group "," optionally substituted C 6 - 14 Ariruokishi group "," substituted good C 7 - 16 Ararukiruokishi group ",” substituted optionally heterocyclic Chio group optionally ",” optionally substituted C 6 - 14 7 Riruchio group "and” optionally 'substituted C 7 - i
- Examples of the “ 6 aralkylthio group” include (1) a halogen atom; (2) a hydroxy group; ( 3 ) an amino group; (4) a nitro group; (5) a cyano group; and (6) an optionally substituted Ci.
- a halogen atom, hydroxy group, an amino group, a nitro group, Shiano group, an optionally halogenated may also be - 6 alkyl group, mono one or Gee Ji 6 Arukiruamino group, C 6 _ 14 Ariru group, mono- or di-C 6 one 1 4 Ari / Reamino group, C 3 _ 8 cycloalkyl group, - 6 alkoxy groups, - 6 ⁇ alkoxy one d-6 alkoxy group, CI- 6 alkylthio groups, C -.
- optionally substituted amino group includes, unless otherwise specified, (1) an optionally substituted 16 alkyl group; (2) an optionally substituted C 2 - 6 Aruke - Le group; (3) optionally substituted C 2 - 6 alkyl group; (4) ⁇ may be substitution 3 - 8 cycloalkyl group; (5) have been replaced which may be C 6 one 1 4 Ariru group; (6) an optionally substituted alkoxy group; (7) an optionally substituted Ashiru group; (8) optionally substituted ⁇ Hajime Tamaki ( (Preferably furyl, pyridyl, chenyl, pyrazolyl, thiazolyl, oxazolyl); (9) sulfamoyl group; (10) mono- or di-Ci-6 alkyl-sulfamoyl group; (11) mono- or di-C 6 — 14 7 Substituted with one or two substituents selected from And an optionally substituted amino group.
- an optionally substituted 16 alkyl group
- nitrogen-containing heterocyclic ring When the “optionally substituted amino group” is an amino group substituted with two substituents, these substituents form a nitrogen-containing heterocyclic ring together with an adjacent nitrogen atom. Also Good.
- nitrogen-containing heterocycle include, for example, at least one nitrogen atom other than a carbon atom as a ring-constituting atom, and one or two heteroatoms selected from an oxygen atom, a sulfur atom, and a nitrogen atom.
- a 5- or 7-membered nitrogen-containing heterocyclic ring which may be contained.
- nitrogen-containing heterocycle examples include pyrrolidine, imidazolidine, pyrazolidine, piperidine, piperazine, morpholine, thiomorpholin, thiazolidine, oxazolidine and the like.
- substituted optionally may be Ashiru group in the present specification, Ri otherwise stated without limited the formula: - COR 2, one CO - OR 2, - S0 2 R 2, - S OR 2, one PO (OR 2 ) (0 R 3 ), —CO—NR 2a R 3a and —CS—NR 2a R 3a [wherein R 2 and R 3 are the same or different and are each a hydrogen atom or a substituted R 2a and R 3a are the same or different, and each represents a hydrogen atom, an optionally substituted hydrocarbon group or an optionally substituted heterocyclic group. R 2a and R 2 together with an adjacent nitrogen atom may form a substituted or unsubstituted nitrogen-containing heterocyclic ring].
- the “nitrogen-containing heterocycle” in the “optionally substituted nitrogen-containing heterocycle” formed by R 2a and R 3a together with an adjacent nitrogen atom includes, for example, at least one nitrogen atom other than a carbon atom as a ring-constituting atom
- a 5- or 7-membered nitrogen-containing heterocyclic ring which contains an atom and may further contain 1 or 2 heteroatoms selected from an oxygen atom, a sulfur atom and a nitrogen atom.
- the nitrogen-containing heterocyclic ring include pyrrolidine, imidazolidine, pyrazolidine, piperidine, piperazine, monoreforin, thiomorpholine, thiazolidine, oxazolidine and the like.
- the nitrogen-containing heterocyclic ring may have one or two substituents at substitutable positions.
- substituents a hydroxy group, optionally halogenated and C i_ 6 alkyl group, 0 6-1 14 Ariru group, such as Cy-i 6 Ararukiru group.
- Preferable examples of the "optionally substituted Ashiru group" is formyl group; carboxyl group; Karupamoiru group; C Bok 6 alkyl one carbonyl group; - 6 alkoxy Ichiriki Rupoeru group; C 3 - 8 cycloalkyl one carbonyl group; C 6 - 14 Ariru Ichiriki Ruponiru group; Cy-i 6 Ararukiru Ichiriki Ruponiru group; C 6- i 4 Ariruokishi - Cal Poniru group; CT- i 6 Ararukiruokishi one carbonyl group; mono- or di- CI- 6 ⁇ Le kills Scarpa carbamoyl group; mono- or di _c 6 - 14 ⁇ Li one Rukarupamoiru group; mono one or di- c 3 _ 8 cycloalkyl Ichiriki Rubamoiru group; mono- or di- c 7 - 16 Ararukiru Ichiriki Rupamoiru group
- Ar represents a cyclic group which may be substituted.
- the "cyclic group” eg if C 3 - 8 cycloalkyl group, an aromatic hydrocarbon group (eg, C 6 _ 14 7 aryl group), and heterocyclic groups.
- Preferred specific examples of the “cyclic group” include cyclopropyl, cyclohexyl, phenyl, naphthyl, chenyl, furyl, thiazolyl, oxazolyl, imidazolyl, pyrazolyl, triazolyl, pyridyl, pyrazinyl, benzo [b] chel, indolyl, And indanyl.
- Cyclic group represented by Ar is preferably an aromatic hydrocarbon group (e.g., C 6 _ 14 7 aryl group), more preferably from phenyl.
- the cyclic group represented by Ar may have, for example, 1 to 5, preferably 1 to 3, substituents at substitutable positions.
- substituents the "optionally substituted C 3 - 8 cycloalkyl group” those exemplified as the substituents for is found using. If the cyclic group has 2 or more substituents, each substituent may be the same or different ⁇ ⁇ '
- Substituent is preferably a halogen atom; Shiano group; but it may also be halogenated C i_ 6 alkyl group; C 6 - 14 Ariru group; human Dorokishi group; C 3 one 8 Shikuroaruki group, halogenated And may be substituted with 1 to 3 substituents selected from an alkoxy group and the like. .
- aromatic hydrocarbons aromatic ring such as an aromatic heterocyclic ring
- alicyclic hydrocarbon an aromatic ring such as a non-aromatic heterocyclic ring
- aromatic hydrocarbons aromatic ring such as an aromatic heterocyclic ring
- alicyclic hydrocarbon an aromatic ring such as a non-aromatic heterocyclic ring
- aromatic hydrocarbon for example, an aromatic hydrocarbon having 6 to 14 carbon atoms can be used.
- aromatic hydrocarbon include benzene, naphthalene, anthracene, phenanthrene, and acenaphthylene.
- aromatic heterocyclic ring examples include a 5- to 7-membered monocyclic aromatic ring containing 1 to 4 hetero atoms selected from oxygen, sulfur and nitrogen atoms in addition to carbon atoms as ring-constituting atoms. Heterocycle or fused aromatic heterocycle is mentioned.
- condensed aromatic complex ring examples include a 5- to 7-membered monocyclic aromatic heterocycle, a 6-membered ring containing 1 to 2 nitrogen atoms, a benzene ring or a 5-membered ring containing one sulfur atom. And a ring in which a member ring is condensed.
- Preferred examples of the aromatic heterocyclic ring include furan, thiophene, pyridine, pyrimidin, pyridazine, pyrazine, pyrrole, imidazole, pyrazole, isoxazole, isothiazole / le, oxazole, thiazole, oxaziazol, thiaziazole, triazole, and triazole.
- Tetrazole Tetrazole, quinoline, quinazoline, quinoxaline, benzofuran, benzothiophene, benzoxazonole, benzothiazonole, benzimidazole, indole, 1H-indazole, 1H-pyro [2, .3-b] pyrazine, 1 Examples include H-pyromouth pyridine, 1H-imidazopyridine, 1H-imidazopyrazine, triazine, isoquinoline, and benzothiaziazole.
- alicyclic hydrocarbon examples include a saturated or unsaturated alicyclic hydrocarbon having 3 to 12 carbon atoms, such as cycloalkane, cycloalkene, and cycloalkadiene.
- cycloalkane examples include cycloalkane having 3 to 3 carbon atoms, for example, cyclopropane, cyclobutane, cyclopentane, cyclohexane, cyclohexane.
- cycloalgen examples include cycloalkene having 3 to 10 carbon atoms, for example, cycloptene, cyclopentene, cyclohexene and the like.
- cycloalkadiene examples include cycloalkadiene having 4 to 10 carbon atoms, such as 2,4-cyclopentadiene, 2,4-cyclohexadiene, and 2,5-cyclohexadiene.
- the non-aromatic heterocyclic ring includes, for example, a 5- to 7-membered monocyclic non-aromatic containing 1 to 4 heteroatoms selected from oxygen, sulfur and nitrogen atoms in addition to carbon atoms as ring-constituting atoms. Group or a fused non-aromatic heterocycle.
- Examples of the condensed non-aromatic heterocyclic ring include these 5- to 7-membered monocyclic non-aromatic heterocyclic ring, a 6-membered ring containing 1 to 2 nitrogen atoms, a benzene ring or one sulfur atom And a ring fused with a 5-membered ring.
- non-aromatic heterocycles Kisamechire dihydrofuran, tetrahydrofuran, dihydropyran Chio Fen, tetrahydrophthalic Chio Fen, H. 1 ⁇ emissions, pyrroline, pyrazole lysine, piperidine, piperidines Rajin, morpholine, thiomorpholine to, And imine, oxazolidine, thiazolidine, imidazolidine, imidazoline, tetrahydrofuran, azepan, oxepane, tetrahydropyridine and the like. .
- aromatic rings such as benzene, pyrazole, thiazole, oxazole, furan, thiophene, oxaziazole, triazole, tetrazole, pyrimidine, benzimidazole and indole are preferable, and benzene is particularly preferable.
- the fertility indicated by ring A is not thiazole, oxazole, imidazole, or pyrazole.
- the “ring” represented by ring A and ring A 1 has a substituent at a substitutable position, for example, 1 It may have up to 5, preferably 1 to 3.
- substituent the "optionally substituted C 3 - 8 cycloalkyl group” those exemplified as the substituents of the may be used.
- each substituent may be the same or different.
- Substituent is preferably a halogen atom, C 7 - 16 Ararukiru group, C 6 - 14 Ari group,. i. Alkoxy group, and a C 7 _ 16 Ararukiruokishi group.
- Ring A is preferably benzene.
- the “ring” represented by ring A 1 may have a substituent represented by the formula: Ar—Xa— (the symbols have the same meanings as described above).
- Ring A 1 is preferably of the formula: is Ar- Xa- (symbol is as defined above) having a substituent Ru indicated by ring A.
- Xa and Xb each independently represent a bond or a spacer having 1 to 5 atoms in the main chain.
- main chain means a divalent straight chain connecting Ar and ring A, and “a main chain atom” Numbers shall be counted so that the number of atoms in the main chain is minimized.
- the “main. Chain” refers to the divalent straight chain connecting ring A and Xc, and The number of atoms in the chain shall be counted so that the atoms in the main chain are minimized.
- the “main chain” is composed of 1 to 5 atoms selected from a carbon atom and a hetero atom (eg, an oxygen atom, a sulfur atom, and a nitrogen atom), and may be saturated or unsaturated. Further, carbon atoms and sulfur atoms may be oxidized.
- a hetero atom eg, an oxygen atom, a sulfur atom, and a nitrogen atom
- C 3 _ 8 Shikuroa alkyl group As the substituent represented by R 4, those exemplified as the substituent in the "optionally substituted. C 3 _ 8 Shikuroa alkyl group" is used. Among them, an optionally substituted C i-alkyl group (preferably, C ⁇ 6 alkyl group, such as 7 _ 16 ⁇ aralkyl group.) And C 3 - 8 cycloalkyl group is preferable.
- Xa is preferably a bond; one O—; —S—; —CH 2 —; — CO—; one CH 20 —; one CH 2 S—; a Ci- 6 alkyl group on the N atom; 7 - 16 Ararukiru group or al chosen monosubstituted group which may have a CH 2 NH-; -OCH 2 -; .
- Xb is preferably one CH 2 —; — CH 2 CH 2 —; — CO—CH 2 —; — CH 2 CH 2 CH 2- ; one CH 2 CH 2 —; one S— CH 2 CH 2 —; — O— CH 2 CH 2 CH 2 —; — S— CH 2 CH 2 CH 2 —; C 3 - 8 consequent opening alkyl Oyo Pi C 7 - 16
- Ararukiru one substituent group selected from the group may have respective NH - CH 2 CH 2 - or a CH 2 - NH- CH 2 CH 2 - And so on.
- Xb is more preferably one CH 2 —.
- Xc is 0, S, indicates the SO or S0 2, preferably zero.
- ring B for example, among the rings exemplified as ring A, those having a 5- to 7-membered ring can be mentioned.
- cycloalkanes having 5 to 7 carbon atoms preferably cyclopentane, cyclohexane and cycloheptane
- cycloalkenes having 5 to 7 carbon atoms preferably cyclopentene and cyclohexene
- 5 to 7 members and a 5- to 7-membered non-aromatic ring such as a monocyclic non-aromatic heterocycle (preferably tetrahydrofuran, oxepane).
- Ring B is more preferably cyclopentane or tetrahydrofuran, particularly preferably tetrahydrofuran.
- Preferred combinations of ring D and ring B include those in which ring D is benzene and ring B is cyclopentane or tetrahydrofuran (ring B is preferably tetrahydrofuran). That is, Is
- the "5- to 7-membered non-aromatic ring" for ring B 1 include those exemplified as the ring B.
- Ring B 1 represents preferably a cyclopentane or tetrahydrofuran, preferably in further tetrahydrofuran.
- a preferred combination of ring]) and ring B 1 is ring! ) Is benzene, and ring B 1 is cyclopentane or tetrahydrofuran (ring B 1 is preferably tetrahydrofuran). That is,
- Xd represents a bond, CH or CH 2, preferably CH 2.
- R 1 is preferably a hydroxy group or an alkoxy group, more preferably a hydroxy group.
- Ring A is benzene
- the cyclic group represented by Ar is not a quinolinyl group
- Ring B is a 5- to 7-membered aromatic ring
- the ring represented by Ring A is thiophene or Not franc
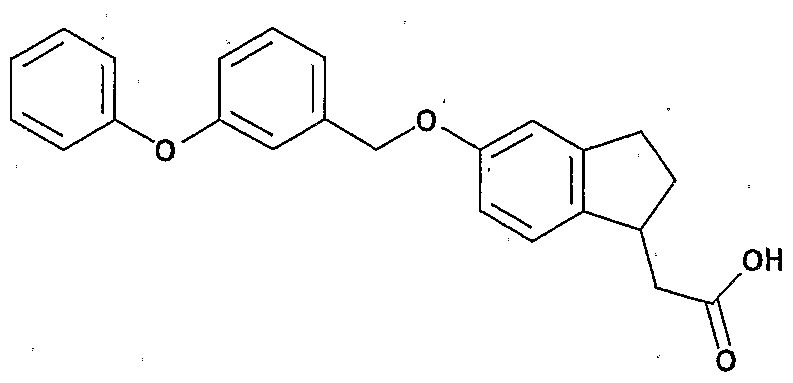
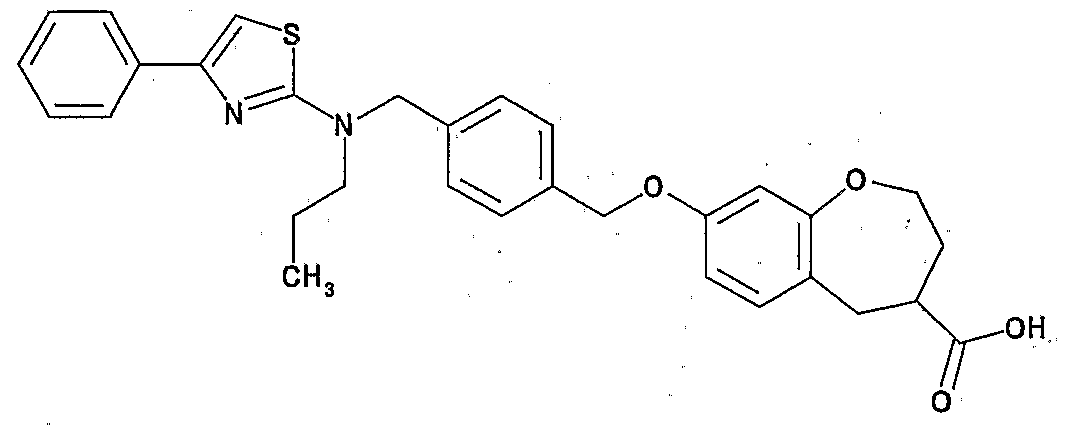
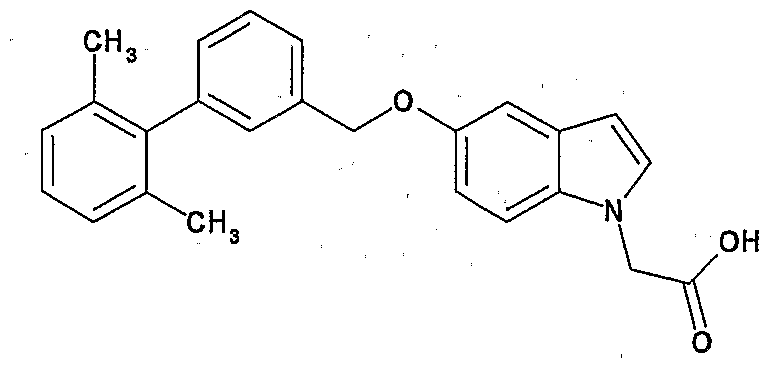
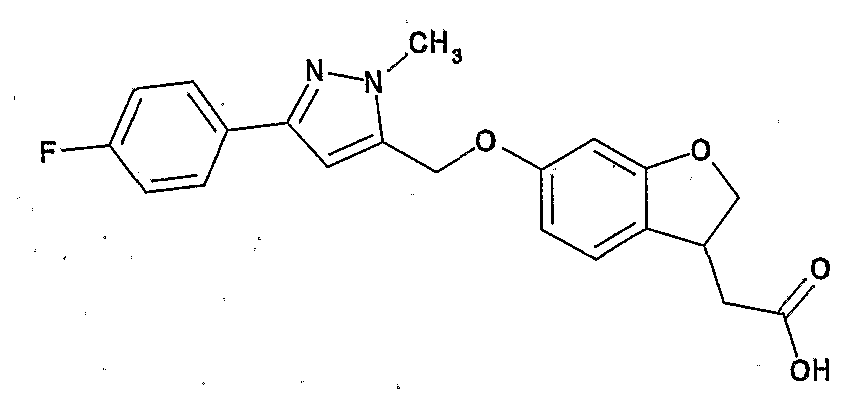
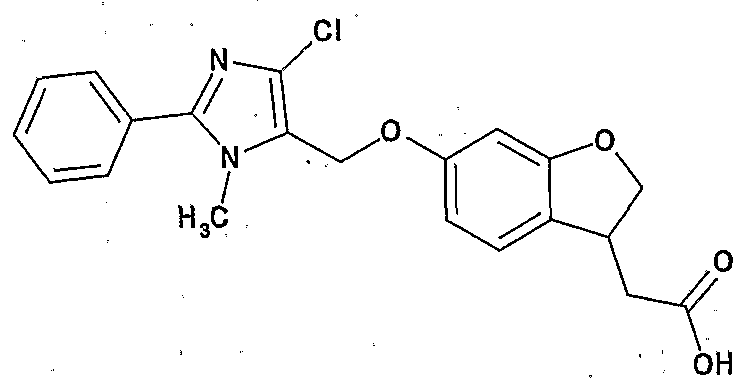
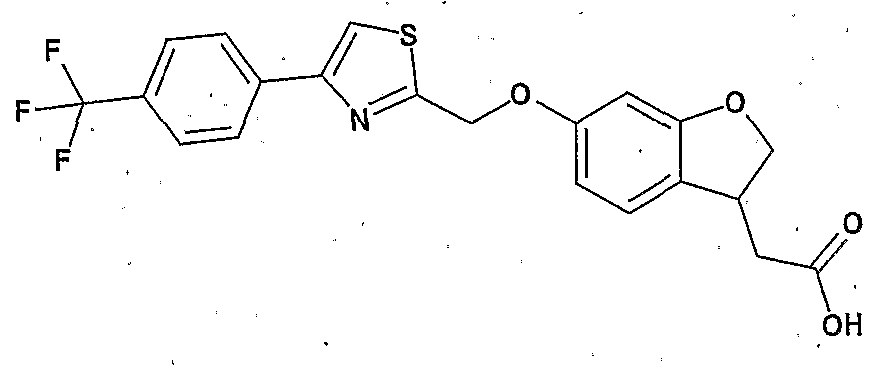
- Preferred examples of the compound (I) include the following compounds.
- Ar force halogen atom .; cyano group; optionally halogenated—6 alkyl group; 6 _ 14 Ariru group; human Dorokishi group; C 3 _ 8 cycloalkyl group, may be halogenated. 1 selected from such Bok 6 alkoxy to Ji may be substituted with 1-3 substituents.
- Ring A halogen atom, C 7 - 16 Ararukiru group, C 6 - 14 Ariru group. 10 alkoxy group, the 1-3 amino aromatic ring which may have a substituent (preferably selected from such C 7 _ 16 7 Rarukiruokishi group, benzene, furan, Chiofen, O key Sajiazoru, triazole, tetrazole, pyrimidine , Benzimidazole, indole; more preferably benzene);
- Preparative 6 alkyl Contact Yopi C 7 - 16 Ararukiru may have a substituent group selected from the group -CH 2 NH-; -OCH 2 -; -SCH 2 -; alkyl Contact Yopi on N atom
- Xb force one CH 2 -; -CH 2 CH 2 -; -CO-CH 2 -;.
- One CH 2 CH 2 CH 2 one; single O- CH 2 CH 2 -; - S -CH 2 CH 2 -; one O- CH 2 CH 2 CH 2 - ; - S -CH 2 CH 2 CH 2 -; or C 3 on the N atom - 8 Shikuroa 7 Rekiru groups and C 7 - i 6 respectively have a substituent selected from Ararukiru group One NH—CH 2 CH 2 — or one CH 2 —NH—CH 2 CH 2 — (preferably one CH 2 —); Xc force S 0;
- R 1 is a hydroxy group or an alkoxy group (preferably a hydroxy group).
- Ar 1 represents an optionally substituted phenyl group or an optionally substituted indaninole group
- Xa 1 is a bond or a spacer having 1 to 5 atoms in the main chain
- Ring A 2 is a benzene ring which may be further substituted; Ring B 2 represents a 5- to 7-membered ring. (Hereinafter, it may be abbreviated as compound (I-12).)
- Ar 1 is preferably a halogen atom, a nitro group, a carboxyl group, may be halogenation - 6 alkyl group, hydroxyalkyl primary alkyl group, a force Rupokishi C - 6 alkylcarbonyl ⁇ plasminogen C 6 alkyl group, it may be halogenated - 6 alkoxy group, C 6 14 Ariru group, C 6 _ 14 Ariruoki shea groups and C 7 - i 6 Ararukiruokishi or a phenyl group which may have each a substituent selected from the group Indanyl group.
- Contact Yopi C 7 _ i 6 Ararukiruokishi may have a location substituent selected from group phenyl groups are preferred, especially a phenyl group which may have a substituent group selected from c port plasminogen atoms and Ha port gain emissions of which may be optionally CI_ 6 alkyl group.
- binding Gote one O- or a CH 2 - O- are preferable p
- Ring A 2 is preferably a further 6 alkyl Nze down ring base may be substituted with a group.
- Ar 2 represents an optionally substituted thiazolyl group
- Xa 2 is a bond having a bond or a main chain having 1 to 5 atoms
- Ring A 3 is a benzene ring which may be further substituted
- Ring B 2 represents a 5- to 7-membered ring. (Hereinafter sometimes abbreviated as compound (1-4).).
- Ar 2 is preferably C 6 - is a 14 Ariru group and Ji which may thiazolyl group optionally having selected Bareru substituent alkyl group (e.g., 2-thiazolyl group).
- Xa 2 is preferably one N (R 5 ) one (CH 2 ) m— or one S— (CH 2 ) m
- R 5 is a hydrogen atom or an alkyl group, m is an integer of 0 to 3), and more preferably 1 N (R 5 ) 1 (CH 2 ) m—.
- Ci- 3 alkyl group such as methyl, ethyl and propyl is preferable, and methyl is particularly preferable.
- Ring A 3 is preferably a benzene ring.
- the present invention further provides a compound represented by the following formula (1-1) and a compound represented by the following formula (I-3).
- ring A 4 represents an optionally substituted benzene ring
- p and q each independently represent a carbon chain having 0 to 4 carbon atoms which may be substituted;
- Xb 1 represents a bond having a bond or a main chain having 1 to 5 atoms;
- Xc 3 is 0, S, SO or ⁇ 0 2,
- Ring B 2 represents a 5- to 7-membered ring. Or a salt thereof (hereinafter may be abbreviated as compound (1-1)).
- ring A 4 is preferably (1) a halogen atom, (2) a 6 alkyl group,
- Ci- 6 alkoxy group (4) halogen atom,. 6 alkyl or - 6 alkoxy substituted by C 6 one optionally 14 Ariru group, (5) C 6 one 14 Ariru Okishi group
- Contact Yopi (6) C 7 - 16 7 have a substituent selected from Rarukiruokishi group It is a benzene ring which may be substituted.
- A4 is preferably
- Xb 1 is preferably a bond.
- Xc 3 is preferably 0.
- ring A 5 represents an optionally substituted benzene ring
- Ring A 6 is an optionally substituted 5-membered heterocyclic ring
- Ring B 2 represents a 5- to 7-membered ring. Or a salt thereof (hereinafter may be abbreviated as compound (1-3)).
- a substituent selected from a halogen atom (eg, a chlorine atom) and an optionally halogenated alkyl group (eg, methyl, trifluoromethyl), It is.
- a halogen atom eg, a chlorine atom
- an optionally halogenated alkyl group eg, methyl, trifluoromethyl
- salts of the compound (compound (I), compound (1 ′), compound (I-11), compound (1-2), compound (1-3), compound (1-4), etc.) used in the present invention examples thereof include metal salts, ammonium salts, salts with organic bases, salts with inorganic acids, salts with organic acids, and salts with basic or acidic amino acids.
- the metal salt include alkali metal salts such as sodium salt and potassium salt; alkaline earth metal salts such as calcium salt, magnesium salt and barium salt; aluminum salt and the like. . .
- salts with an organic base include: Salts with N, N'-dibenzylethylenediamine and the like can be mentioned.
- salts with inorganic acids include salts with hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid, and the like.
- Preferred examples of salts with organic acids include, for example, formic acid, acetic acid, trifluoroacetic acid, phthalic acid, fumaric acid, oxalic acid, tartaric acid, maleic acid, citric acid, succinic acid, malic acid, methanesulfonic acid, benzene Salts with sulfonic acid, p-toluenesulfonic acid and the like can be mentioned. .
- Preferred examples of the salt with a basic amino acid include, for example, salts with arginine, lysine, and orutin.
- Preferred examples of the salt with an acidic amino acid include, for example, salts with asparaginic acid, glutamic acid, and the like. Is mentioned.
- salts pharmaceutically acceptable salts are preferred.
- Etc. (hereinafter sometimes abbreviated as the compound of the present invention) are compounds which are converted into the compound of the present invention by a reaction with an enzyme or gastric acid under physiological conditions in vivo, that is, enzymatically oxidized, A compound which undergoes reduction, hydrolysis or the like to be converted into the compound of the present invention, or a compound which undergoes hydrolysis or the like by stomach acid or the like to be converted into the compound of the present invention.
- a compound in which the amino group of the compound of the present invention is acylated, alkylated, or phosphorylated for example, the amino group of the compound of the present invention is eicosanylated, araerylated, pentylaminocarbonylated
- 5-Methyl-2-oxo-1,3-dioxolene-4-1-f methoxycarbonylation, tetrahydrofuranylation, pyrrolidylmethylation, piperyloxymethylation, tert-butylated compound
- a compound in which the hydroxyl group of the compound of the present invention is acylated, alkylated, phosphorylated, or borated for example, the hydroxyl group of the compound of the present invention is acetylated, palmitoylated, propanoylated, piperoylated, succinyl
- Fumarylation for example, the amino group of the compound of the present invention is acetylated, palmitoylated
- the prodrug of the compound of the present invention can be prepared under physiological conditions as described in Hirokawa Shoten, 1990, “Development of Pharmaceuticals,” Vol. 7, Molecular Design, pp. 163-198. It may change into a compound.
- each symbol in the schematic diagrams in the following reaction formulas has the same meaning as described above unless otherwise specified.
- Each of the compounds in the reaction formulas may be in the form of a salt, as long as the reaction is not inhibited, and examples of the salt include those similar to the salts of the compounds used in the present invention described above.
- the compound obtained in each reaction can be used in the next reaction as a reaction solution or as a crude product, but can also be isolated from the reaction mixture according to a conventional method, using a usual separation method (eg, recrystallization). , Distillation, chromatography, etc.).
- the X c 2 is SO or S 0 2
- the R la is optionally substituted 6 alkoxy group
- L is a hydroxy group or a leaving group
- Examples of the leaving group represented by L include a halogen atom, an optionally halogenated Ci-6 alkylsulfo-loxy group (for example, methanesulfonyloxy, ethanesnorefonoleoxy, and trichloromethanesnolehoninoleo).
- Xy trinoleolomethanesulfo-oxo.
- C e- i which may have a substituent.
- reel sulfonyl O key sheet 3 ⁇ 4 [e.g., an alkyl group which may have 1 to 3 substituents selected from alkoxy groups and nitro groups C 6 - 1 0 ⁇ Li one Le sulfonyl O alkoxy group
- phenylsulfonyloxy phenylsulfonyloxy, naphthylsulfuroxy
- an acyloxy group eg, trichloroacetoxy, trifluoroacetoxy
- Step 1 Compound (la) is obtained by reacting a compound represented by formula (II) with a compound represented by formula (III) (abbreviated as compound (II) and compound (III), respectively). Can be manufactured more. (i) When L is a hydroxy group, compound (la) reacts compound (II) with compound (III) by the Mitsunobu reaction (for example, Synthesis, p. 127, p. Described).
- the compound (II) and the compound (III) are converted into azodicarboxylates such as getyl azodicarboxylate, diisopropyl azodicarboxylate, 1,1,1- (azodicarbonyl) dipiperidine, and phenols such as triphenylphosphine and triptylphosphine.
- azodicarboxylates such as getyl azodicarboxylate, diisopropyl azodicarboxylate, 1,1,1- (azodicarbonyl) dipiperidine, and phenols such as triphenylphosphine and triptylphosphine.
- the azodicarboxylates and phosphines are used in an amount of about 1 to about 5 mol, preferably about 1 to about 2 mol, per 1 mol of compound (II).
- This reaction is advantageously performed using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds.
- examples of such a solvent include dimethyl ether, diisopropylinoleatenole, dipheninoleatenole, tetrahydrofuran, 1,4-dioxane, and 1,2-dimethoxetane.
- Ethers aromatic hydrocarbons such as benzene and toluene; saturated hydrocarbons such as cyclohexane and hexane; N, N-dimethylformamide, N, N-dimethylacetamide, hexamethylphosphoric triamide
- Amides such as dichloromethane, chloroform, carbon tetrachloride, halogenated hydrocarbons such as 1,2-dichloroethane and the like; -tolyls such as acetutrile and pentionitrile; and acetone and ethyl methyl ketone Ketones
- solvents such as sulphoxides such as dimethylsulphoxide or the like
- Etc. are preferable et a mixed-solvent.
- the amount of compound (III) to be used is about 0.5 to about 5 mol, preferably about 1 to about 2 mol, per 1 mol of compound (II).
- the reaction time is generally 5 minutes to 100 hours, preferably 30 minutes to 72 hours.
- the reaction temperature is usually from 120 to 200 ° C, preferably from 0 to 100 ° C.
- compound (la) can be produced by reacting compound (II) with compound (III) in the presence of a base.
- the base examples include an alkali metal hydroxide such as lithium hydroxide, sodium hydroxide, or hydroxide hydroxide; an alkaline earth metal such as parium hydroxide; Alkali metal carbonates such as sodium acid, potassium carbonate and cesium carbonate; alkali metal hydrogencarbonates such as sodium hydrogen carbonate; acetates such as sodium acetate and ammonium acetate; aromatic amines such as pyridine and lutidine; triethylamine, tripropylamine, triptylamine , N-Echillisy Soprot.
- alkali metal hydroxide such as lithium hydroxide, sodium hydroxide, or hydroxide hydroxide
- an alkaline earth metal such as parium hydroxide
- Alkali metal carbonates such as sodium acid, potassium carbonate and cesium carbonate
- alkali metal hydrogencarbonates such as sodium hydrogen carbonate
- acetates such as sodium acetate and ammonium acetate
- aromatic amines such as pyridine and
- Tertiary amines such as luminamine, cyclohexyldimethylamine, 4-dimethylaminopyridine, N, N-dimethylaniline, N-methylbiperidine, N-methylpyrrolidine, N-methylmorpholine; sodium hydride, hydrogen Metal hydrides such as sodium hydride; metal amides such as sodium amide, lithium diisopropylamide, and lithium hexamethyldisilazide; carbons such as sodium methoxide, sodium ethoxide, sodium tert-butoxide, and potassium tert-butoxide Examples include alkali metal alkoxides of the numbers 1 to 6.
- the amount of the base to be used is about 1 to about 10 mol, preferably about 1 to about 3 mol, per 1 mol of compound (II).
- This reaction is advantageously performed using a solvent inert to the reaction.
- a solvent inert those similar to the aforementioned “when L is a hydroxy group” are used.
- the amount of compound (III) to be used is about 0.8 to about 10 mol, preferably about 0.9 to about 2 mol, per 1 mol of compound (II). Also,
- the reaction time is generally 10 minutes to 12 hours, preferably 20 minutes to 6 hours.
- the reaction temperature is usually from 170 to 150 ° C, preferably from 120 to 10'0 ° C.
- Step 2 A> Compound (lb) can be produced by subjecting compound (la) to a hydrolysis reaction.
- the hydrolysis reaction is carried out according to a conventional method using an acid or a base.
- the acid examples include mineral acids such as hydrochloric acid and sulfuric acid; Lewis acids such as boron trichloride and boron tribromide; and organic acids such as trifluoroacetic acid and P -toluenesulfonic acid.
- the Lewis acid can be used in combination with a thiol or a sulfide.
- the base examples include: alkaline metal hydroxides such as lithium hydroxide, sodium hydroxide, and hydroxide hydroxide; alkaline earth metal metals such as parium hydroxide; Alkali metal carbonates such as sodium acid and potassium carbonate; C1-C6 alkali metal alkoxides such as sodium methoxide, sodium ethoxide and potassium tert-butoxide; organic bases such as triethylamine, imidazole and formamidine Including).
- alkaline metal hydroxides such as lithium hydroxide, sodium hydroxide, and hydroxide hydroxide
- alkaline earth metals such as parium hydroxide
- Alkali metal carbonates such as sodium acid and potassium carbonate
- C1-C6 alkali metal alkoxides such as sodium methoxide, sodium ethoxide and potassium tert-butoxide
- organic bases such as triethylamine, imidazole and formamidine Including).
- the amount of the acid or base used is about 0.5 to about 10 mol, preferably about 0.5 to about 6 mol, per 1 mol of compound (la).
- the hydrolysis reaction is performed using a solvent that is inert to the force reaction performed without a solvent.
- a solvent is not particularly limited as long as the reaction proceeds. Examples thereof include alcohols such as methanol, ethanol, and propanol; aromatic hydrocarbons such as benzene and toluene; and saturated hydrocarbons such as cyclohexane and hexane.
- Hydrocarbons organic acids such as formic acid and acetic acid; ethers such as tetrahydrofuran, dioxane, and 1,2-dimethoxyethane; amides such as N, N-dimethylformamide and N, N-dimethylacetamide; dichloromethane; Halogenated hydrocarbons such as chloroform, carbon tetrachloride, and 1,2-dichloroethane; didrills such as acetonitrile and propionitrile; ketones such as acetone and methyl ethyl ketone; sulfoxides such as dimethyl sulfoxide A solvent such as water or a mixture thereof is preferred. Arbitrariness.
- the reaction time is generally 10 minutes to 100 hours, preferably 10 minutes to 24 hours.
- the reaction temperature is usually from 110 to 200 ° C, preferably from 0 to 120 ° C.
- the oxidation reaction is usually performed using an oxidizing agent according to a conventional method.
- oxidizing agent examples include hydrogen peroxide, peracetic acid, sodium metaperiodate, potassium permanganate, sodium perborate, metachloroperoxybenzoic acid (MC PBA), acyl nitrate, nitrous oxide, halogen, N-promosuccinimide (NBS), N-chlorosuccinimide (NCS) and the like.
- the oxidizing agent is used in an amount of about 0.5 to about 10 mol per 1 mol of the compound (la). Preferably about 1 to about 5 moles.
- the oxidation reaction is carried out using a solvent that is free of solvent and a solvent that is inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds. Examples thereof include alcohols such as methanol, ethanol, and propanol; aromatic hydrocarbons such as benzene and toluene; organic acids such as acetic acid and trifluoroacetic acid; Preferred are halogenated hydrocarbons such as chloroform, carbon tetrachloride, and 1,2-dichloroethane; solvents such as water; and mixed solvents thereof.
- the reaction time is generally from 10 minutes to 100 hours, preferably from 10 minutes to 24 hours.
- the reaction temperature is usually from 120 to 150 ° C, preferably from 0 to 100 ° C.
- the compound (lb ') can be produced by subjecting the compound represented by the formula (Ia,) to a hydrolysis reaction.
- This reaction can be carried out in the same manner as in Step 2A or according to a method analogous thereto.
- Compound (Ib,) can be produced by subjecting a compound represented by the formula (lb) (a compound in which X c 1 is S) to an oxidation reaction.
- the oxidation reaction can be carried out in the same manner as in Step 3A or according to a method analogous thereto.
- Compound (III) used in Reaction Scheme 1 can be easily obtained as a commercial product. It can also be produced according to a method known per se.
- X a is — (CH 2 ) kl — Q— (CH 2 ) k2 —
- Xb is Xba—CH 2 (Xba represents a bond or a spacer having 1 to 4 main chain atoms)
- (III ′) (hereinafter sometimes abbreviated as compound (III ′)) can be produced, for example, according to the method shown in Reaction Scheme 2 or a method analogous thereto.
- the number of atoms in the main chain is one to four.
- L 1 represents a leaving group
- R 6 represents a hydrogen atom or an optionally substituted alkoxy group
- M represents a hydrogen atom or a metal (eg, potassium, sodium, lithium, magnesium, copper, mercury, zinc) , Thallium, boron, tin, etc., which may be complexed)
- the leaving group represented by L 1 those exemplified as the aforementioned L can be Chi used.
- Compound (VI) can be prepared by reacting (i) compound (IV-1) with compound (V-1), or (ii) compound (IV-2) with compound (V-2 ).
- the compound (IV-1) and the compound (IV 2) are collectively referred to as the compound (IV), and the compound (V-1) and the compound (V-1), unless otherwise specified.
- (V-2) is generically referred to as compound (V) unless otherwise specified.
- the reaction between compound (IV) and compound (V) is usually performed in the presence of a base.
- the base include alkali metal hydrides such as sodium hydride and hydrogenation power; water such as lithium hydroxide, sodium hydroxide, and hydrogenation power; alkali metal oxides; magnesium hydroxide; Alkaline earth metal hydroxides such as calcium; alkali metal carbonates such as sodium carbonate and carbonated carbonate; alkali metal bicarbonates such as sodium bicarbonate and sodium hydrogen carbonate; sodium methoxide, sodium ethoxide Alkoxides of 1 to 6 carbon atoms, such as sodium tert-butoxide and sodium tert-butoxide; for example, trimethylamine, triethylamine, diisopropylethylamine, pyridine, picoline, N-methylpyrrolidine, N-methylmorpholine, 1,5 —Diazabicyclo [4.3.0] — 5-Nonene, 1,4 Organic bases such as azabicyclo [2.2.2]
- the reaction between compound (IV) and compound (V) is advantageously performed using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds.
- alcohols such as methanol, ethanol, propanol, isopropanol, ptananol, and tert-butanol; dioxane, tetrahydrofuran, diethylenoether, tert- alcohol; Ethers such as p-inolemethinoleatenore, diisopropinoleatenole, and ethylene glycol-dimethyl ether; esters such as ethyl formate, ethyl acetate, and ⁇ -butyl acetate; dichloromethane, chloroform, carbon tetrachloride, and trichloroethylene Halogenated hydrocarbons such as ⁇ -hexane, benzene, toluene, etc .; Amides such as formamide
- the reaction between compound (IV) and compound (V) can be accelerated by using a metal catalyst.
- metal catalyst metal complexes having various ligands are used.
- palladium compounds eg: palladium (II) acetate, tetrakis (triphenylphosphine) palladium (0), bis (triphenyl-) Ruphosphine) palladium (11), dichlorobis (triethylphosphine) palladium '(0), tris (dibenzylideneacetone) dipalladium 1,2,1'bis (diphenyl / lephosphino) 1-1,1, -binaphthyl , Palladium (II) acetate and 1,1,1-bis (diphenylphosphino) phenecene complex etc.]; Nickel compound [Example: tetrax (triphenylphosphine) nickel (0), bis (triethylphosphine) chloride Nickel
- the amount of the metal catalyst to be used is about 0.00001 to about 5 mol, preferably about 0.0001 to about 1 mol, per 1 mol of compound (IV).
- the reaction is preferably performed in an inert gas (eg, argon gas or nitrogen gas) stream.
- the amount of compound (V) to be used is about 0.1 to about 10 mol, preferably about 0.5 to about 2 mol, per 1 mol of compound (IV).
- the amount of the base to be used is about 1-about 20 mol, preferably about 1-about 5 mol, per 1 mol of compound (IV).
- the reaction temperature is from 110 to 250 ° C, preferably from 0 to 150 ° C.
- the reaction time varies depending on the compound (IV), the compound (V), the metal catalyst, the type of the base or the solvent, the reaction temperature, etc., but is usually 1 minute to 200 hours, preferably 5 minutes to 100 hours. is there. Step 5> Compound (II I ′) can be produced from compound (VI).
- Compound (III) wherein L is a hydroxy group can be produced by subjecting compound (VI) to a reduction reaction.
- the reduction reaction is usually performed using a reducing agent according to a conventional method.
- the reducing agent include metal hydrides such as aluminum hydride, diisobutylaluminum hydride and triptyltin hydride; sodium cyanoborohydride, sodium triacetoxyborohydride, sodium borohydride
- Metal hydride complexes such as lithium aluminum hydride; borane tetrahydrofuran complexes, porane complexes such as borane dimethyl sulfide complexes; alkyl boranes such as texyl borane, disiamil porane; diborane; zinc, aluminum, tin, iron, etc.
- alkali metals such as sodium and lithium Z liquid ammonia (perch reduction).
- the amount of the reducing agent used is appropriately determined depending on the type of the reducing agent.
- the amount of the metal hydride, metal hydride complex compound, porane complex, alkylporane or diborane to be used is about 0.25 to about 10 mol, preferably about 0.25 mol, per 1 mol of compound (VI).
- the amount of the metal (including the alkali metal used in the perch reduction) is about 1 to about 20 equivalents, preferably about 1 to about 20 moles per 1 mole of the compound (VI). 5 equivalents.
- This reaction is advantageously performed using a climbing inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds.
- alcohols such as methanol, ethanol, 1-pro / no-no-re, 2-propano-no-re and tert-p-ti-no-re-no-cone Ethers such as getyl ether, diisopropyl ether, diphenyl ether, tetrahydrofuran, 1,4-dioxane, 1,2-dimethoxetane; aromatic hydrocarbons such as benzene and toluene; cyclohexane and hexane; Saturated hydrocarbons; N, N-dimethylformamide, N, N-dimethylacetamide, hexamidemethylphosphoric triamide, etc .; organic acids such as formic acid, acetic acid, propanoic acid, trifluoroacetic acid, methanesulfonic acid, etc
- the reaction time varies depending on the reagents and solvents used, but is usually from 10 minutes to 100 hours, preferably from 30 minutes to 50 hours.
- the reaction temperature is usually from 120 to 100 ° C, preferably. ⁇ 80 ° C.
- Compound (111,) wherein L is a hydroxy group is converted to a halogenating agent
- the compound (III) wherein L is a leaving group can be produced by reacting the compound (III) with a quenching agent.
- halogenating agent for example, thionyl chloride, phosphorus tribromide and the like are used.
- a compound ( ⁇ ) in which L is a halogen atom eg, chlorine, bromine, etc.
- the reaction between the compound (II) and the halogenating agent is usually performed in a solvent that does not adversely influence the reaction.
- Solvents that do not adversely affect the reaction include, for example, halogenated hydrocarbons such as dichloromethane, chloroform, tetrachloromethane, and the like; aromatic hydrocarbons such as benzene, toluene, xylene, and the like; getyl ether, diisopropyl Ethers such as ether, tert-butyl methyl ether, tetrahydrofuran, dioxane, and dimethoxetane; and esters such as methyl acetate, ethyl acetate, n-butyl acetate, and tert-butyl acetate.
- an excess amount of a halogenating agent may be used as a solvent.
- the amount of the halogenating agent to be used is generally about 1 to about 10 mol per 1 mol of compound ( ⁇ ).
- the reaction temperature is usually from 120 to 100 ° C.
- the reaction time is usually 0.5 to 24 hours.
- the sulfonylide for example, methanesulfonyl chloride, chlorobenzenesulfonyl chloride, p-toluenesulfonyl chloride and the like are used.
- the compound (III ′) in which L is, for example, methanesulfonyloxy, benzenesulfonyloxy, P-toluenesulfonyloxy, or the like can be produced by the reaction with the sulfonylating agent.
- the amount of the sulfonylating agent to be used is generally about 1-about 0 mol, per 1 mol of compound (III ′).
- the reaction of compound (III ') with a sulfurating agent is usually performed in a solvent that does not adversely influence the reaction, in the presence of a base.
- Solvents that do not adversely affect the reaction include, for example, halogenated hydrocarbons such as dichloromethane, chloroform, and carbon tetrachloride; aromatic hydrocarbons such as benzene, toluene, and xylene; getyl ether, disopropyl / ether, and the like.
- Ethers such as tert-butyl / lemeth / leate / le, tetrahydrofuran, dioxane, and dimethoxetane; methyl acetate, ethyl acetate, acetic acid Esters such as n-butyl and tert-butyl acetate.
- Examples of the base include amines such as triethylamine and N-methylmorpholine; metal salts such as sodium hydrogencarbonate, potassium hydrogencarbonate and potassium carbonate;
- the amount of the base to be used is generally about 1-about 10 mol per 1 mol of compound (II).
- the reaction temperature is usually from 120 to 100 ° C.
- the reaction time is usually 0.5 to 24 hours.
- Compound (1-2) can be produced, for example, according to the method shown in the following Reaction Scheme 3 or a method analogous thereto.
- Compound (VII) and compound (VIII) can be easily obtained from commercially available ones, and also include those produced according to a method known per se or a method analogous thereto.
- Step 7> Compound (1-2) is prepared from compound (IX) in the same manner as in Step 2 can do.
- Compound (I-14) can be produced, for example, according to the method shown in the following Reaction Scheme 4 or a method analogous thereto.
- Step 8> Compound (XI) can be produced from compound (VII) and compound (X) in the same manner as in step 1.
- Compound (X) can be easily obtained as a commercially available product, and can also be produced according to a method known per se or a method analogous thereto.
- the compound in which X a 2 is -N, (R 5 )-(CH 2 ) in— can be prepared, for example, according to the method represented by the following reaction formula 5 or a method analogous thereto. It can also be manufactured.
- Step 10> Compound (XIII) can be produced from compound (VII) and compound (XII) in the same manner as in step 1.
- Step 1 1> Compound (XV) wherein X a is one N (R 5 ) one (CH 2 ) m—
- X a is one N (R 5 ) one (CH 2 ) m—
- the compound can also be produced from compound (XIII) and compound (XIV) in the same manner as in step 4.
- Compound (1-1) can be produced, for example, according to the method represented by the following reaction formula 6 or a method analogous thereto.
- Compound (XVII) can be produced by reducing the carbonyl group of compound (XVI).
- the amount of the reducing agent used is, for example, in the case of metal hydrides and metal hydride complex compounds, about 1 to about 10 moles, preferably about 1 to about 5 moles, and , Alkylboranes or dipolane, about 1. to about 10 moles, preferably about 1 to about 5 moles, and about 1 to about 20 equivalents for metals, per mole of compound (XVI) Preferably about 1 to about 5 equivalents.
- Lewis acids may be used if desired.
- Lewis acids examples include aluminum chloride, aluminum bromide, titanium chloride (IV), tin chloride (11), zinc chloride, boron trichloride, boron tribromide, and boron trifluoride.
- Can be The amount of Lewis acid to be used is about 1 to about 10 mol, preferably about 1 to about 5 mol, per 1 mol of compound (XVI). It is also reduced by a hydrogenation reaction.
- a catalyst such as palladium carbon, platinum (IV) oxide, Raney-Eckel, and Raney-cobalt is used.
- the amount of the catalyst to be used is about 5 to about 100% by weight, preferably about 10 to about 300% by weight, per 1 mol of compound (XVI).
- hydrogen sources can be used instead of gaseous hydrogen.
- the “hydrogen source” formic acid, ammonium formate, triethylammonium formate, sodium phosbuinate, heptadazine and the like are used.
- the amount of the hydrogen source to be used is about 1 to about 10 mol, preferably about 1 to about 5 mol, per 1 mol of compound (XVI).
- This reaction is advantageously performed using a solvent inert to the reaction.
- a solvent inert there is no particular limitation as long as the reaction proceeds as such a solvent, but examples thereof include methanol, ethanol, 1-propanol, 2-propanol, tert-butyl alcohol, etc.
- the reaction time varies depending on the type and amount of the Liaoning agent used or the activity and amount of the catalyst, but is usually about 0.1 hour to about 100 hours, preferably about 1 hour to about 50 hours.
- the reaction temperature is usually about 120 to about 120 ° C, preferably about 0 to about & 0 ° C.
- the hydrogen pressure is usually about 1 to about 100 atmospheres.
- Compound (XVIII) can be produced by converting the hydroxy group of compound (XVII) into a “leaving group”. .
- halogenating agent used for halogenation examples include halogenated thiols such as thiol chloride and thiol bromide, phosphoryl chloride, and the like.
- Phosphoryl halides such as phosphoryl bromide, phosphorus pentachloride, phosphorus trichloride, phosphorus pentabromide, phosphorus halides such as phosphorus tribromide, etc.
- Oxalyl halides such as luchloride, phosgene and the like.
- the halogenating agent is used in an amount of about 0.1 to about 30 mol, preferably about 0.2 to about 10 mol, per 1 mol.
- the “base” includes triethylamine, tripropylamine, triptylamine, N-ethyldiisopropylamine, cyclohexyldimethylamine, 4-dimethylaminopyridine, N, N-dimethylaniline, N-methylbiperidine And tertiary amines such as N-methylpyrrolidine and N-methylmorpholine.
- the base is used in an amount of about 1 to about 20 mol, preferably about 1 to about 10 mol, per 1 mol of compound (XVII).
- This reaction is carried out without a solvent. It is advantageous to carry out the reaction using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds.
- hydrocarbons such as benzene, toluene, cyclohexane, and hexane, dimethyl ether, diisopropyl ether, diphenyl ether, tetrahydrofuran, 1 Ethers such as 1,4-dioxane and 1,2-dimethoxetane, amides such as N, N-dimethylformamide, ⁇ , ⁇ -dimethylacetamide, hexamethylphosphoric triamide, dichloromethane Preferred are solvents such as chloroforms, carbon tetrachloride, and 1,2-dichloroethane, and the like, or solvents such as mixed solvents thereof.
- the reaction time is generally about 10 minutes to about 12 hours, preferably about 10 minutes to about 5 hours.
- sulfonylating agent for example, halogen I spoon C, such as methanesulfonyl chloride - 6 alkylsulfonyl, benzenesulfonyl chloride, chloride p - halides such as toluenesulfonic Honiru C 6 - 1 0 7 Reelsulfonyl and the like.
- the sulfonylating agent is used in an amount of about 1 to about 20 mol, preferably about 1 to about 10 mol, per 1 mol of compound (XVII).
- This reaction is carried out without a solvent. It is advantageous to carry out the reaction using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds.
- hydrocarbons such as benzene, toluene, cyclohexane, hexane, dimethyl ether, diisopropyl ether, diphenyl ether, tetrahydrofuran, Ethers such as 1,4-dioxane and 1,2-dimethoxetane, halogenated hydrocarbons such as dichloro.methane, chloroform, carbon tetrachloride and 1,2-dichloroethane, methyl acetate, ethyl acetate, acetic acid Solvents such as esters such as butyl and the like or a mixed solvent thereof are preferable.
- This reaction is performed in the presence of a base, if desired.
- bases include triethylamine, tripropylamine, triptylamine, N-ethyldiisopropylamine, cyclohexyldimethylamine, 4-dimethylaminopyridine, N, N-dimethylayuline, and N-methylbiperidine.
- Tertiary amines such as N-methylpyrrolidine, N-methylmorpholine
- inorganic bases such as sodium hydroxide, lithium hydroxide, lithium hydroxide, barium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate
- basic salts such as sodium hydrogencarbonate, sodium acetate, and ammonium acetate.
- the base is used in an amount of about 1 to about 20 mol, preferably about 1 to about 10 mol, per 1 mol of compound (XVII).
- the reaction time is generally about 10 minutes to about 12 hours, preferably about 10 minutes to about 5 hours.
- the reaction temperature is usually about 13.0 to about 150 ° C, preferably about 120 to about 100 ° C.
- compound (XX) can be produced by condensing compound (XVIII) and compound (XIX) in the presence of a base.
- Examples of the base used in this reaction include inorganic bases such as sodium hydroxide, hydroxide hydroxide, lithium hydroxide, barium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate, sodium hydrogen carbonate, sodium acetate, and ammonium acetate.
- inorganic bases such as sodium hydroxide, hydroxide hydroxide, lithium hydroxide, barium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate, sodium hydrogen carbonate, sodium acetate, and ammonium acetate.
- Basic salts such as pyridine, aromatic amines such as pyridine, lutidine, etc., triethylamine, propylamine, triptylamine, N-ethyldiisopropylamine, N-hexyldimethylamine, 4-dimethylaminopyridine, N, N -Dimethylayurin, Tertiary amines such as N-methylpiperidine, N-methylpyrrolidine, N-methylmorpholine, alkali metal hydrides such as sodium hydride, hydrogenation hydride, sodium amide, lithium diisopropylamide, lithium hexamethyldisi Metal amides such as razide; metal alkoxides such as sodium methoxide, sodium ethoxide, sodium tert-butoxide, and potassium tert-butoxide. These bases are used in an amount of about 1-10 mol, preferably about 1-3 mol, per 1 mol of compound (XVIII).
- This reaction is advantageously performed using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds.
- alcohols such as methanol, ethanol, 1-propanol, 2-propanol and tert-butyl alcohol, getyl ether, diisopropyl ether, diphenic acid Ether, tetrahydrofuran, 1,4-dioxane, 1,2-dimethoxetane, etc., ethers such as benzene, toluene, cyclohexane, hexane, etc., ⁇ , ⁇ -dimethylformamide Amides such as ⁇ , ⁇ -dimethylacetoamide and hexamethylphosphoric triamide; halogenated hydrocarbons such as dichloromethane, chloroform, carbon tetrachloride and 1,2-dichloroethane; acetonitrile; -Tolyls such as propionitrile, methyl
- the reaction time is generally about 10 minutes to about 12 hours, preferably ⁇ 20 minutes to about 6 hours.
- the reaction temperature is usually about 150 to about 150 ° C, preferably about 120 to about 100 ° C.
- compound (XX) can also be produced by condensing compound (XVII) and compound (XIX) in the presence of a dehydrating agent if desired. it can.
- Examples of the dehydrating agent used in this reaction include acidic catalysts such as hydrochloric acid, sulfuric acid, phosphoric acid, potassium hydrogen sulfate, oxalic acid, p-toluenesulfonic acid, 10-camphorsulfonic acid, boron trifluoride etherate, and water.
- acidic catalysts such as hydrochloric acid, sulfuric acid, phosphoric acid, potassium hydrogen sulfate, oxalic acid, p-toluenesulfonic acid, 10-camphorsulfonic acid, boron trifluoride etherate, and water.
- carboximides such as ⁇ , ⁇ '-dicyclohexylcarboximide, alumina, sodium dioxide, phosphorus oxychloride, thionyl chloride, methanesulfuryl chloride, etc.
- These acids and bases are used in an amount of about 0.1 to 10 mo
- This reaction is advantageously carried out using a solvent inert to the power of the reaction.
- the solvent is not particularly limited as long as the reaction proceeds. Examples thereof include alcohols such as methanol, ethanol, and propanol; ethers such as dimethyl ether, tetrahydrofuran, dioxane, and 1,2-dimethoxetane; formic acid, and acetic acid.
- Organic acids such as benzene, toluene, cyclohexane, hexane, etc., amides such as ⁇ , ⁇ -dimethylformamide, ⁇ , ⁇ -dimethylacetamide, dimethyl sulfoxide, etc.
- Solvents such as sulfoxides or mixed solvents thereof are preferred.
- the reaction time is usually 30 minutes to 24 hours, preferably 30 minutes to 5 hours.
- the reaction temperature is usually 0 to 200 ° C, preferably 0 to: L50 ° C.
- Compound (XX) can also be produced by subjecting compound (XVII) and compound (XIX) to a Mitsunobu reaction when X -Xc 3 is an oxygen atom.
- the reaction is performed in the same manner as in the above step 1.
- Compound (1-1) can be produced from compound (XX) in the same manner as in Step 2A.
- Compound (1-3) of the present invention can be produced, for example, according to the method represented by the following reaction formula 7 or a method analogous thereto.
- Compound (XXI) can be easily obtained as a commercially available product, and can also be produced according to a method known per se or a method analogous thereto.
- Compound (XXII) can be produced from compound (VII) and compound (XXI) in the same manner as in step 1.
- Compound (1-3) can be produced from compound (XXII) in the same manner as in Step 2A.
- Examples of the protecting group for the amino group include a formyl group; a C i- 6 alkyl-propionyl group which may have a substituent (for example, acetyl, propionyl, etc.), a benzoyl group, and a C ⁇ -group.
- eAlkoxy-carbonyl group for example, methoxycarbonyl, ethoxycarbonyl, tert-butoxycarbinole (Boc), etc.
- aryloxycarbol group A 1 oc
- phenyl / reoxycarbyl group fluorenylmethyloxy group .
- an aralkyloxy-hydroxyl group for example, benzyloxycarbonyl
- a trityl group for example, a phthaloyl group, a dithiasuccinoyl group, an N, N-dimethylaminomethylene group and the like
- substituent for example, a phenyl group, a halogen atom, an alkyl-carboxy group (for example, acetyl, propionyl, parryl, etc.), an optionally halogenated Ce alkoxy group, a nitro group and the like are used.
- the number of substituents is
- Examples of the carboxyl-protecting group include a C 6 alkyl group, an aryl group, a benzyl group, a phenyl group, a trityl group, and a trialkylsilyl group, each of which may have a substituent.
- substituent include a halogen atom, a formyl group, a C 6 alkyl-carbonyl group (for example, acetyl, propionyl, butylcarbonyl, etc.), an optionally halogenated C 6 alkoxy group, a nitro group, i 6 alkyl group, C 6 .
- Aryl groups eg, phenyl, naphthyl, etc. are used, and the number of substituents is about 1 to 3.
- Examples of protecting groups for human Dorokishi groups such as formyl group or Ji each of which may have a substituent ⁇ alkyl,, C 7 1 0 Ararukiru group, C 6 alkyl - Power Ruponiru group (e.g., Asechiru, propionyl, etc. ), A benzoyl group, a phenylcarbonyl group. Ararukiruokishi -.
- Carbo sulfonyl group e.g., benzyl Ruo propoxycarbonyl El
- Ararukiru - carbonyl group e.g., benzyl Rukaruponiru
- Tetorahi Dorobiraniru group Tetorahi Dorofurayuru group
- tetrahydrothienyl group a silyl group Is used.
- substituent include a halogen atom and. - 6 alkyl group,. . Ararukiru group (e.g., benzyl etc.), C 6.
- Aryl groups eg, phenyl, naphthyl, etc.
- alkoxy groups nitro groups, etc., and the number of substituents is about 1 to 4.
- the protection group of mercapto includes, for example, the substituents may be respectively, C i-e alkyl group, C 7 _ 2 0 Ararukiru group (e.g., benzyl, trityl) and the like.
- the substituent for example, halogen atom, C - 6 alkyl group, phenyl group, C 7 1 0 Ararukiru group (e.g., benzyl etc.), C i-6 alkoxy groups and nitro groups, substituted
- the number of groups is one to four.
- a method for removing the protecting group a method known per se or a method analogous thereto is used. Examples thereof include an acid, a base, ultraviolet light, hydrazine, phenylhydrazine, and N-methyl.
- a method of treating with sodium rudithiocarbamate, tetrabutylammonium fluoride, palladium (II) acetate, or a reduction reaction is used.
- a hydrolysis reaction a deprotection reaction, an acylation reaction, an alkylation reaction, a hydrogenation reaction, an oxidation reaction, a reduction reaction, a carbon chain extension reaction, and a substituent exchange reaction are respectively performed.
- the compound of the present invention can be synthesized by performing the reaction alone or by combining two or more thereof. For these reactions, for example, the methods described in New Experimental Chemistry Lectures 14, 15 and 1977 (Maruzen Publishing) are employed.
- the desired product When the desired product is obtained in a free state by the above reaction, it may be converted to a salt according to a conventional method, and when obtained as a salt, it may be converted to a free form or another salt according to a conventional method. You can also.
- the compound of the present invention thus obtained can be isolated and purified from the reaction solution by known means, for example, phase transfer, concentration, solvent extraction, fractionation, crystallization, recrystallization, chromatography and the like. '
- the compound of the present invention exists as a configurational isomer (configuration isomer), a diastereomer, a conformer, or the like, each can be isolated by the above separation and purification means, if desired.
- the compound of the present invention is in a racemic form, it can be separated into S-form and R-form by ordinary optical resolution means.
- the compound of the present invention has a stereoisomer
- the case where the isomer is a single compound and the case where the compound is a mixture thereof are also included in the present invention.
- the compound of the present invention may be a hydrate or a non-hydrate.
- the compound of the present invention may be labeled with an isotope (eg, 3 H, 14 C, 35 S) or the like.
- an isotope eg, 3 H, 14 C, 35 S
- the compound of the present invention or a prodrug thereof has a GPR40 receptor function regulating action, particularly a GPR40 receptor agonist activity, and has low toxicity and few side effects. It is useful as a safe GPR40 receptor function modulator, preferably a GPR40 agonist.
- the compound of the present invention or a prodrug thereof may be a mammal (eg, mouse, rat, Hamsters, egrets, cats, cats, dogs, puppies, sheep, monkeys, humans, etc.). It is useful as a function regulator or a prophylactic / therapeutic agent for pathologies or diseases involving the GPR40 receptor.
- the compound of the present invention or a prodrug thereof is useful as an insulin secretion regulator (preferably an insulin secretagogue), a hypoglycemic agent, and a ⁇ / 3 cell protective agent.
- an insulin secretion regulator preferably an insulin secretagogue
- a hypoglycemic agent preferably an insulin secretagogue
- a ⁇ / 3 cell protective agent preferably an insulin secretagogue
- the compound of the present invention and its prodrug are useful as a blood glucose level-dependent insulin secretagogue based on its GPR40 receptor agonist activity. That is, the compound of the present invention or its prodrug is useful as an insulin secretagogue that does not cause hypoglycemia, unlike the sulfonylprea drug.
- the compound of the present invention or a prodrug thereof may be, for example, diabetic, impaired glucose tolerance, ketosis, acidosis, diabetic neuropathy, diabetic nephropathy, diabetic retinopathy, hyperlipidemia, sexual dysfunction, skin Diseases, arthropathy, osteopenia, arteriosclerosis, thrombotic diseases, dyspepsia, memory and learning disorders, obesity, hypoglycemia, hypertension, edema, insulin resistance, unstable diabetes, lipoatrophy, insulin allergy, insulinoma, Diseases such as lipotoxicity, hyperinsulinemia, and cancer, especially diabetes, impaired glucose tolerance, ketosis, acidosis, diabetic neuropathy, diabetic nephropathy, diabetic retinopathy, hyperlipidemia, sexual dysfunction, Useful as a preventive agent for skin diseases, arthrosis, osteopenia, arteriosclerosis, thrombotic diseases, indigestion, memory and learning disorders A.
- diabetes includes insulin-dependent (type I) diabetes, non-insulin-dependent (type II) diabetes and gestational diabetes.
- Hyperlipidemia also includes hypertriglyceridemia, hypercholesterolemia, hypo-HD Lemia, and postprandial hyperlipidemia.
- criteria for diabetes a new criteria was reported by the Japanese Diabetes Association in 1999.
- diabetes is defined as a fasting blood glucose level (glucose concentration in venous plasma) of more than 126 mg / d1 and a 75 g transglucose tolerance test (75 g OGTT).
- Glucose concentration in plasma is more than 200 mg Z dl
- the blood glucose level is any of 20 Omg / d1 or more.
- glucose concentration in venous plasma glucose in venous plasma
- 75 gOGTT oral glucose tolerance test
- venous plasma A condition in which the glucose concentration is less than 14 Omg / d1 is referred to as a “boundary type.”
- the criteria for diabetes were determined by the ADA (American Diabetes Association) in 1997. In 1998, WHO's reported new criteria.
- diabetes was defined as a fasting blood glucose level (dulcose concentration in venous plasma) of 126 mg Zd 1 or more, and a 2-hour value of 75 g glucose glucose tolerance test (glucose concentration in venous plasma). ) Indicates a state of 20 OmgZd 1 or more.
- impaired glucose tolerance refers to a fasting blood glucose level (glucose concentration in venous plasma) of less than 126 mg Zd1 and a 2-hour value of 75 g transglucose glucose load test (in venous plasma).
- Glucose concentration is 14 Omg / d1 or more and less than 20 OmgZd1.
- IFG Impaired Fasting Glucose
- the compound of the present invention or a prodrug thereof is also used as a prophylactic / therapeutic agent for diabetes, borderline type, impaired glucose tolerance, IFG (Impaired Fasting Glucose) and IFG (Impaired Fasting Glycemia) determined by the above-mentioned new criteria. Used.
- the compound of the present invention can also prevent progression to diabetes from borderline type, impaired glucose tolerance, IFG (Impaired Fasting Glucose) or IFG (Impaired Fasting Glycemia).
- the compound of the present invention or a prodrug thereof has low toxicity and can be used as a method for producing a pharmaceutical preparation.
- the compound of the present invention or its open drug is used as is or in the form of a mixture with a pharmacologically acceptable carrier, according to a generally known means known per se, for example, tablets (including sugar-coated tablets and film-coated tablets). , Powders, granules, capsules, (including soft capsules), liquid preparations, injections, suppositories, sustained-release preparations, etc., and then orally or parenterally (eg, topical, rectal, intravenous administration, etc.) ) Can be administered safely.
- the content of the compound of the present invention in a pharmaceutical preparation is about 0.01 to about 100% by weight of the whole preparation. / 0 .
- the dose varies depending on the administration subject, administration route, disease, symptom and the like. For example, for a diabetic patient (body weight of about 6 O kg), about 13 as an active ingredient [compound of the present invention] per 13 is used. 1 to about 3 O mg / kg body weight, preferably about 0.1 to about 2 O mg / kg body weight, more preferably about 1 to about 2 O mg / kg body weight divided into 1 to 1 or several times It may be administered orally.
- Pharmaceutically acceptable carriers that may be used in the production of the medicament of the present invention include various organic or inorganic carrier substances commonly used as pharmaceutical materials, such as excipients in solid formulations, lubricants, etc. , Binders and disintegrants, or solvents in liquid preparations, solubilizers, suspending agents, isotonic agents, buffers and soothing agents. Further, if necessary, usual additives such as preservatives, antioxidants, coloring agents, sweeteners, adsorbents, and wetting agents can be used in appropriate amounts. :
- Excipients include, for example, lactose, sucrose, D-mantol, starch, corn starch, crystalline cellulose, light caffeic anhydride and the like.
- lubricant examples include magnesium stearate, calcium stearate, talc, colloidal silica and the like.
- Binders include, for example, crystalline cellulose, sucrose, D-mannitol, dextrin, hydroxypropinoreserenolose, hydroxyapine pinoremethy / resenorelose, polyvinylinolevicenidone, starch, sucrose, gelatin, methinoreserose, strength And sodium norboxoxymethylcellulose.
- Disintegrators include, for example, starch, carboxymethylcellulose, carboxymethyl / resenorelose / resin, sodium canolepoxmethinolestearate, L-hide Roxypropyl cellulose and the like.
- solvent examples include water for injection, alcohol, propylene glycol, macrogol, sesame oil, corn oil, and olive oil.
- Solubilizers include, for example, polyethylene daricol and hydroxy. Rendericol, D-mannitol, benzyl benzoate, ethanol, trisaminomethane, cholesterol, triethanolamine, sodium carbonate, sodium citrate and the like.
- suspending agent examples include surfactants such as stearyltriethanolamine, sodium lauryl sulfate, laurylaminopropionic acid, lecithin, benzalkonium chloride, benzethonium chloride, and glycerin monostearate; for example, polyvinylinolea / reconole, Examples include hydrophilic polymers such as polyvinylinolepyrrolidone, sodium casolepoxymethinoresenorelose, methinolace / rerose, hydroxymethylresenololose, hydroxyxethenoresolenolate and hydroxypropylcellulose.
- surfactants such as stearyltriethanolamine, sodium lauryl sulfate, laurylaminopropionic acid, lecithin, benzalkonium chloride, benzethonium chloride, and glycerin monostearate
- polyvinylinolea / reconole examples include hydrophilic poly
- tonicity agent examples include pudose sugar, D-sorbitol, sodium chloride, glycerin, D-mannitol and the like.
- buffers such as phosphate, acetate, carbonate, and citrate.
- Examples of the soothing agent include benzyl alcohol and the like.
- preservative examples include parahydroxybenzoic acid esters, chlorobutanol, benzyl alcohol, phenethyl alcohol, dehydroacetic acid, sorbic acid and the like.
- antioxidant examples include sulfite, ascorbic acid, ⁇ -tocopherol and the like.
- the compound of the present invention or a prodrug thereof can be used in combination with a drug other than the compound of the present invention.
- concomitant drug examples include, for example, other therapeutic agents for diabetes, therapeutic agents for diabetic complications, therapeutic agents for hyperlipidemia, antihypertensive agents, Anti-obesity agents, diuretics, chemotherapeutics, immunotherapy, immunomodulators, anti-inflammatory drugs, anti- Antithrombotics, osteoporosis treatments, antibacterials, antifungals, antiprotozoal drugs, antibiotics, Relaxant, Antiepileptic, Antidepressant, Antiallergic, Cardiotonic, Antiarrhythmic, Vasodilator, Vasoconstrictor, Narcotic antagonist, Vitamin, Vitamin derivative, Antiasthmatic, Antidementia, Frequent urination ⁇ Urine incontinence, dysuria, Atopic dermatitis, allergic rhinitis, vasopressor, endotoxin antagonist or antibody, signal transduction inhibitor, inflammatory mediator inhibitor, inflammation Anti-inflammatory mediator action inhibitory
- insulin preparations eg, animal insulin preparations extracted from the stomach of the stomach and septum; human insulin preparations genetically synthesized using Escherichia coli and yeast; insulin zinc; protamine insulin) Zinc; fragment or derivative of insulin (eg, INS-1 etc.), oral insulin preparation, etc.
- insulin sensitivity enhancer eg, pioglitazone or its salt (preferably hydrochloride), rosiglitazone or its salt (preferably malein) Acid salt
- Reglixane JTT-500
- Nedglitazone MCC—555
- CS-011 Lipoglitazone (Rivoglitazone) (
- GLP-1 receptor agonist [e.g., GLP-1, GLP-1 MR agent, NN-2211, AC-2939 (exendin 4), BIM-51077, Aib (8, 35) hGLP-1 (7, 37) ⁇ 2 CJC-1131 etc.], di peptidyl peptidase IV inhibitors (e.g., NVP-DP P- 278, PT- 1 00, P 32/98, P93 / 01, NVP- DPP- 728, LAF237, TS- 021, etc.), 3 agonists (eg, CL-316243, SR-5861 ⁇ _ ⁇ , UL-TG-307, AJ-9677, AZ 40140, etc.), amiline agonists (eg, pramlintide, etc.), phosphotyrosine phosphatase Inhibitors (eg, panadic acid), gluconeogenesis inhibitors (eg, glycogen phosphorylase inhibitor, glucose 16-phosphatase inhibitor, glu
- Therapeutic agents for diabetic complications include aldose reductase inhibitors (eg, tolrestat, e / norestat, zenarestat, zoponorestat, fidarestat (SNK-86.0), minarestat (A1-509) , CT-112, etc.), neurotrophic factor and its increasing drugs (eg, NGF, NT-3, BDNF, neurotrophin production and secretagogues described in W001 / 14372 (eg, 4- (4- black hole Hue yl) - 2- (2-methyl-1-Imidazoriru) - 5- [3 -. (2-menu Chirufuenokishi) prop le] etc.
- aldose reductase inhibitors eg, tolrestat, e / norestat, zenarestat, zoponorestat, fidarestat (SNK-86.0), minarestat (A1-509) , CT-112, etc.
- neurotrophic factor and its increasing drugs eg, NGF, NT
- PKC protein kinase C
- LY-333351 e.g. , LY-333351, etc.
- AGE inhibitors e.g, ALT-945, pimagedin, vilatoxatin, N-phenacylthiazolidumpromide (ALT-766), EXO-226, ALT-711, pyridrine (Pyridorin), pyridoxamine, etc.
- active acid Element scavengers eg, thioctic acid
- cerebral vasodilators eg, thiopride
- somatostatin receptor agonists eg, thiopride
- ASK-1 apoptosis signal regulatory kinase-1
- a cholesterol synthesis inhibitor As a therapeutic agent for hyperlipidemia, a cholesterol synthesis inhibitor, a scoutin-based compound (eg, prapatin, simpastatin, oral pastatin, atorpastatin, fullpastatin, seripastatin, or a salt thereof (eg, sodium salt, etc.)) Etc.), squalene synthase inhibitors (eg, compounds described in W097 / 10224, for example, N — [[(3R, 5S) -1- (3-acetoxy-2,2-dimethylpropyl) -7-coguchi] -5-(2,3-Dimethoxyphenyl) -2-oxo-1,2,3,5-tetrahydro-4,1-benzoxazepine-3-yl] acetyl] piperidine- 4-acetic acid, etc., fiprate compounds (eg, beza fibrate, clofibrate, simfibrate, clinofib
- Antihypertensive agents include angiotensin-converting enzyme inhibitors (eg, captopril, enalapril, delapril, etc.), angiotensin II antagonists (eg, oral sultan, hordesa / letan cilexetinole, eprosanolethane, panolethanoretane, tenoremisanoretanane , Ilbesartan, tasosartan, 1-[[2,-(2,5-dihydro-5-oxo-4H-4H-1,2,4-oxoxadiazol-3-yl) biphenyl-4-yl] methyl] -2 -Ethoxy-1H-benzimidazole-7-carboxylic acid, etc.), calcium antagonists (eg, mayudipine, difedipin, am.lodipin, efonidipine, dicardipine, etc.), clodin.
- anti-obesity agents include central anti-obesity agents (eg, dexfenfluamine, fenfluramine, phentermine, cyptramine, ampuepramone, dexane fuetamine, mazindore, fuenolepropanolenamine, clovenzolex; MCH Receptor antagonist (eg, SB-568849; SNAP-7941; compounds included in W001 / 82925 and W001 / 87834 fc); Neuropeptide Y antagonist (eg, CP-422935, etc.); Cannabinoid receptor antagonist (eg Ghrelin antagonists; 11 / 3-hydroxyxerodide dehydrogenase inhibitors (eg, BVT-3498 etc.), ⁇ lipase inhibitors (eg, orlistat, ATL-966, etc.) Two ), / 33 agonist (eg, CL-316243, SR-5861 1-A, UL-TG-307, AJ
- Diuretics include, for example, xanthine derivatives (eg, sodium neptimeteop salicylate, calcium theopromine salicylate, etc.), thiazide-based preparations (eg, ethiazide, cyclopentiazide, trichlormethiazide, hydroclothia thiazide, hydrofloxacin ⁇ ⁇ methiazide, Benzi hydrochloride thiazide, penfunorethidide, polythiazide, methyclothiazide, etc., anti-aldosterone preparations (eg, spironolatane, triamterene, etc.), carbonic anhydrase inhibitors (eg, acetazolamide, etc.), chlorbenzene sulphonamide-based preparations (eg, , Mefluside, indapamide, etc.), azosemide, isosorbide, ethacrynic
- Chemotherapeutic agents include, for example, alkylating agents (eg, cyclophosphamide, diphosphamide, etc.), antimetabolites (eg, methotrexate, 5-fluorouracil, etc.), anticancer antibiotics (eg, mitomycin, adriamycin) Etc.), plant-derived anticancer agents (eg, vincristine, vindesine, taxol, etc.), cisplatin, carpoplatin, etopoxide and the like.
- alkylating agents eg, cyclophosphamide, diphosphamide, etc.
- antimetabolites eg, methotrexate, 5-fluorouracil, etc.
- anticancer antibiotics eg, mitomycin, adriamycin Etc.
- plant-derived anticancer agents eg, vincristine, vindesine, taxol, etc.
- cisplatin etopoxide and the like.
- immunotherapeutic agents include microbial or bacterial components (eg, muramyl dipeptide derivatives, picipaniel, etc.), polysaccharides having immunopotentiating activity (eg, lentinan, schizophyllan, krestin, etc.), and genetic engineering techniques.
- Cytokinin eg, interferon, interleukin (IL), etc.
- colony stimulating factor eg, granulocyte colony stimulating factor, erythropoietin, etc.
- IL-1, IL-12, IL— Interleukins such as 12 are preferred.
- anti-inflammatory drug examples include non-steroidal anti-inflammatory drugs such as aspirin, acedaminophen, indomethacin and the like.
- antithrombotic agents include heparin (eg, heparin sodium, heparin calcium, dalteparin sodium, etc.), perfurin (eg, perfurin potassium, etc.), antithrombin drugs (eg, argatroban (aragatroban) )), Thrombolytic drugs (eg, perokinase (urokinase), tisokinase (tisokinase), alteplase (alteplase), nateplase (nateplase), monteplase (monteplase), pamiteplase ⁇ pamiteplase), platelet aggregation inhibitors (eg And ticlopidine hydrochloride, cilostazol, ethyl icosapentate, beraprost sodium, s
- Examples of therapeutic agents for osteoporosis include alfacalcidol (alfacalcidol), canolecitrione (calcitriol), enorecatonin (elcatonin), salmon cane resitto (calcitonin salmon), estriol (estriol) and ipriflavone (ipriflavone). Thorium, pamidronate disodium, 7-lendronate sodium hydrate, incadronate disodium and the like.
- Vitamins drugs such as vitamin B1, vitamin B1 2, and the like.
- anti-dementia agents examples include tacrine, donepezil, rivastigmine, galantamine and the like.
- Examples of the therapeutic agent for pollakiuria and urinary incontinence include flavoxate hydrochloride, oxybutynin hydrochloride, and propiverine hydrochloride. '
- therapeutic agents for dysuria include acetylcholinesterase inhibitors (eg, distigmine) and the like.
- drugs that have been shown to improve cachexia in animal models and clinically, such as cyclooxygenase inhibitors (eg, indomethacin, etc.) [Cancer Research, Vol. 49, Vol. 3 5 ⁇ 5 9 39 pages, 1989 years), professional Gesterone derivatives (eg, Megsterol acetate) [Journal of Clinical Oncology, Vol. 12, pp.
- cyclooxygenase inhibitors eg, indomethacin, etc.
- professional Gesterone derivatives eg, Megsterol acetate
- carbohydrate steroids eg, , Dexamethasone, etc.
- metoc oral bromides e.g., metoc oral bromides, tetrahydrocannabinols (all of which are the same as described above), fat metabolism improvers (eg, eicosapentaenoic acid, etc.) [Pretty Jacinal Cancer (British Journal of Cancer), Vol. 68, 314-3.18, 19993], growth hormone, IGF-1, or TNF- ⁇ , LIF, a factor that induces cachexia.
- Antibodies against IL-16 and oncostatin M can be used in combination with the preparation of the present invention.
- saccharification inhibitors eg, ALT-711 etc.
- nerve regeneration promoters eg, Y-128, VX853, prosaptide, etc.
- antidepressants eg, desipramine, amitriptyline, imipramine
- antiepileptic drugs eg, Eg, lamotrigine
- antiarrhythmic drugs eg, mexiletine
- acetylcholine receptor ligands eg, ABT-594
- endothelin receptor antagonists eg, ABT-627
- monoamine uptake inhibitors eg, tramadol
- narcotics sexual analgesics
- GABA receptor agonists eg, gyapapentin
- ⁇ 2 receptor agonists eg, clonidine
- local analgesics eg, capsaicin
- anxiolytics eg, benzothiaze Pin
- phosphodiesterase inhibitors eg, sil
- the dose can be reduced.
- a drug to be used in combination with the compound of the present invention can be selected according to the patient's symptoms (mild, severe, etc.)
- the treatment period can be set longer
- the timing of administration of the compound of the present invention and the concomitant drug is not limited, and the compound of the present invention and the concomitant drug may be administered simultaneously to a subject to be administered. However, they may be administered at staggered times.
- the dose of the concomitant drug may be in accordance with the dose clinically used, and can be appropriately selected depending on the administration subject, administration route, disease, combination, and the like.
- Examples of the administration form of the compound of the present invention and the concomitant drug include (1) administration of a single preparation obtained by simultaneously formulating the compound of the present invention and the concomitant drug, and (2) separate administration of the compound of the present invention and the concomitant drug. (3) Simultaneous administration of the two preparations obtained in the same administration route by the same administration route, and (3) the time difference in the same administration route of the two preparations obtained by separately formulating the compound of the present invention and the concomitant drug.
- Root temperature in the following Reference Examples and Examples usually indicates about 10 ° C. to about 35 ° C. %, The yield of the mol / mol%, the solvent used in chromatography volume%, others are by weight 0/0. In the proton NMR spectrum, those which cannot be confirmed by prod such as, OH and NH proton are not described in the data.
- Melting point measuring instrument Yanagimoto trace melting point measuring instrument or Buchi melting point measuring instrument, Model B-545 was used.
- NMR measurement equipment Parian Varian Gemini 200 (200MHz) s Varian Gemini 300 (300MHz), Purcar Biospin AVANCE 300.
- Solvent solution A; water containing 0.1% trifluoroacetic acid
- Solution B 0.1% trifluoroacetic acid in acetonitrile
- Ethyl 8- (benzyloxy) -2,3-dihydro-1-benzoxepin-4-carboxylate (2.5 g) was dissolved in trifluoroacetic acid (10 mL), and 42% hydrobromic acid (0.5 mL) was added. ) And heated at 60 ° C for 30 minutes. After concentration, the mixture was extracted with ethyl acetate. The organic layer was washed with water and back-extracted with a 1 M aqueous sodium hydroxide solution. The aqueous layer was acidified with hydrochloric acid and extracted with ethyl acetate. The organic layer was washed with water and brine, dried over magnesium sulfate, and the solvent was distilled off.
- Step 1 5-Methoxy-1-indanone (10.3 g, 63.5 mmol) in toluene (150 mL). The suspension was ice-cooled, and aluminum chloride (16.9 g, 127 mmol) was added little by little. The mixture was refluxed for 4 hours under a nitrogen atmosphere. The reaction solution was allowed to cool to room temperature, poured into ice water, and the organic matter was extracted with a mixed solvent of ethyl acetate and tetrahydrofuran. The extract was dried over anhydrous magnesium sulfate and concentrated under reduced pressure to give 5-hydroxy-1-indanone as yellow crystals.
- Step 2 Suspend the product in acetone (120 mL) and add benzyl bromide (10.9 g, 64.0 mmol) and potassium carbonate (12.3 g, 88.9 mmol) were added, and the mixture was heated to reflux for 1 hour under a nitrogen atmosphere. After allowing the reaction solution to cool to room temperature, ethyl acetate and water were added. The organic layer was washed with saturated saline, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure to give the title compound (14.2 g, 94%) as pale yellow crystals, which was obtained as T (reproduced from ethyl acetate). crystal).
- Reference Example 1 7 2- (benzyloxy) -6,7,8,9-tetrahydro-5H-benzo [7] annulen-5-one
- the title compound was obtained as colorless prism crystals from 2-methoxy-6,7,8,9-tetrahydro-5H-benzo [7] annulen-5-one. Yield 91% (recrystallized from ethyl acetate / hexane).
- Step 1 A mixture of 4- (chloromethyl) -17-hydroxy-2H-chromen-2-one (10.9 g, 51.8 dishes ol) and 1 M aqueous sodium hydroxide solution (500 mL) was prepared. The mixture was heated under reflux for 2 hours. The reaction solution was allowed to cool, acidified with concentrated sulfuric acid, and extracted with ethyl acetate. The extract was washed with brine, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure to give (6-hydroxy-1-benzofuran-3-yl) acetic acid (8.27 g, 83%) as brown crystals. Obtained.
- Step 2 This product (9.85 g, 51.3 mmol) was suspended in methanol (45 mL), concentrated sulfuric acid (5 mL) was added, and the mixture was heated under reflux for 4 hours. The reaction solution was concentrated under reduced pressure, water was added, and the mixture was extracted with getyl ether. The extract was washed with saturated aqueous sodium hydrogen carbonate and saturated brine, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (10% to 50% ethyl ethyl acetate), and the obtained solid was recrystallized from ethyl acetate / hexane to give the title compound (7.38 g, yield 70%). ) Was obtained as pale yellow prism crystals.
- Reference Example 2 4 2,2,4,6, -Tri ⁇ tylbiphenyl-3-butyraldehyde.
- reaction mixture was neutralized with 1 M hydrochloric acid and extracted with ethyl acetate.
- the compound (2.73 g, quantitative) was obtained as a pale yellow oil.
- Step 1 To a solution of ice-cooled tolueneethyl phosphonoacetate (20.2 g, 90.0 mmol) in toluene (100 mL) is added sodium hydride (60% oily, 3.60 g, 90.0 mmol). The temperature was raised to 50 ° C under stirring and the mixture was stirred for 1.5 hours. The reaction solution was cooled on ice, and a solution of 6-methoxy-1-tetralone (10.6 g, 60.0 mmol) in toluene (100 mL) was added dropwise, and the mixture was heated under reflux for 10 hours. After cooling to room temperature, add water Extracted with ethyl acetate.
- Step 2 Dissolve the product in dichloromethane (300 mL), add dropwise a 1 M boron tribromide dichloromethane (135 mL, 135 mmol) solution at -78 ° C in a nitrogen atmosphere, and add the same temperature for 4 hours at room temperature. Then, the mixture was further stirred for 1 hour. The reaction solution was added to an ice-cooled saturated aqueous sodium hydrogen carbonate solution, and the mixture was stirred at room temperature for 12 hours. The organic phase was separated, washed with saturated saline, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure.
- Step 3 This product is dissolved in toluene (25 mL), and 3-phenoxybenzyl alcohol (2.40 g, 12.0 mmol) and tributylphosphine (2.99 mL, 12.0 mmol) are added. After cooling with ice, 1,-(azodicarbonyl) dipiperidine (3.03 g, 12.0 mmol) and tetrahydrofuran (150 mL) were added, and the mixture was stirred at room temperature for 8 hours under a nitrogen atmosphere. The reaction mixture was concentrated under reduced pressure, and the residue was added with a mixed solution of toluene Z hexane (2: 1), and the insolubles produced by the addition were filtered off.
- Example 2 In the same manner as in Example 1 and Example 2, the title compound was obtained as a pale brown viscous oil from 5-methoxy-1-indanone 3-phenyloxybenzyl alcohol. Yield 4%.
- Example 6 In the same manner as in Example 6, the title compound was converted to 8-[(3-phenoxybenzyl) oxy] -2,3,4,5-tetrahydro-1--1-benzoxepin-4-potassium ethyl ester by the same method as in Example 6. Obtained as crystals. 87% yield.
- Example 1 1 ⁇ 6-[(2,6, -dimethylbiphenyl-3-yl) methoxy] -1, 2,3,4-te Trahidronaphthalene-1-yl ⁇ acetic acid
- Example 1 38-[(2,6, -Dimethylbiphenyl-3-yl) methoxy] -2,3,4,5-tetrahydro-1-benzobenzoxepin-4-carboxylic acid
- Example 1 4 ⁇ 5-[(3-phenoxybenzyl) oxy] -2,3-dihydro-1H-indene-1-yl ⁇ ethyl acetate
- Example 11 In the same manner as in Example 11, the title compound was converted from ⁇ 5-[(3-phenoxybenzyl) oxy] -2,3-dihydro-1H-indene-1-yl ⁇ ethyl acetate by colorless prisms. Was obtained. Yield 79% ('recrystallized from ethyl hexane monoacetate).
- Example 2 2 ⁇ 2- [(3 - phenoxyethanol benzyl) Okishi] -6, 7, 8, 9 - Tetorahi mud - 5H - benzo [7] An'nuren - 5-I le ⁇ acetate Echinore Ethyl (2-hydroxy-6,7,8,9-tetrahydro-5H-benzo [7] annulen-5-yl) acetate and 3-phenoxybenzyl alcohol in the same manner as in Example 8. To give the title compound as a colorless oil. Yield '73%.
- Example 2 4 ⁇ 2-[(2,6, -dimethylbiphenyl-3-yl) methoxy] -6,7,8,9-tetrahydro-5H-benzo [7] annulene-5 -Yl ⁇ Acetile acetate
- (2-hydroxy-6,7,8,9-tetrahydro-5H-benzo [7] annulen-5-yl) ethyl acetate and (2 ', 6, -dimethylbiphenyl) The title compound was obtained as a colorless oil from (3-yl) methanol. Yield 72%.
- Example 11 [2-[(2,, 6'-Dimethylbiphenyl-3-yl) methoxy] -6,7,8,9-tetrahydro-5--benzo [ 7]
- the title compound was obtained as colorless crystalline powder from annelen-5-yl ⁇ ethyl acetate. Yield 60% (recrystallized from ethyl hexane monoacetate).
- Example 2 7 ⁇ 6-[(3-Phenoxybenzyl) oxy] -1-benzofuran-3-yl ⁇ vinegar
- Example 3 4 ⁇ 6-[(4- ⁇ [(4-phenyl-1,3-thiazol-2-yl) (propyl) amino] methyl ⁇ benzyl) oxy] -2,3-dihydro-1 -Venzofuran-3-yl ⁇ methyl acetate
- Example 4 1 ⁇ 6-[(6-Methoxy-2,6, -dimethylbiphenyl-3-yl) methoxy] -2,3-dihydro-1-benzobenzofuran-3-acetic acid
- Example 4 5 (6- ⁇ [3- (1-Benzoen-3-yl) benzyl] oxy 2,3-dihydrido / 1-benzofuran-3-yl) acetic acid
- Example 4 7 (6- ⁇ [3- (2-Methyl-1-naphthyl) benzyl] oxy ⁇ -2,3-dihydro-1-benzofuran-3-yl) acetic acid
- Example 4 8 ⁇ 5-[(4- ⁇ [(4-phenyl-1,3-thiazol-2-yl) (propyl) amino] methyl ⁇ benzyl) oxy] -2,3-dihydro-1H -Indene-1-yl ⁇ ethyl acetate
- Example 5 1 ⁇ 6-[(4, -Hydroxy-2, -methylbiphenyl-3-yl) methoxy]-
- Example 5 4.. ⁇ 6-[(4, -Methoxy-2, -methylbiphenyl-3-yl) methoxy] -2,3-dihydro'-1-benzobenzofuran-3-yl ⁇ acetic acid
- Example 5 6 (6- ⁇ [4,-(cyclopropylmethoxy) -2, -methylbiphenyl-3-yl] methoxy ⁇ -2,3-dihydro-1-benzobenzofuran-3-yl) Acetic acid
- Example 6 in the throat Similarly, (6 - ⁇ [4 - (cyclopropyl main butoxy) - 2 '- Mechirubifu Eniru -3 - I le] menu butoxy ⁇ - 2, 3 - dihydric mud - 1 - base Nzofuran - 3-yl)
- the title compound was obtained as colorless prisms from methyl acetate. Yield 63 (recrystallized from ethyl hexane monoacetate).
- Example 5 8 (6- ⁇ [4,-(2-butoxetoxy) -2, -methylbi: 7enyl-3-yl] methoxy ⁇ -2,3-dihydro-1-benzofuran-3-yl) Acetic acid
- Example 5 9 (6- ⁇ [2'-Methyl-4,-(1-propylbutoxy) biphenyl-3-yl] methoxy ⁇ -2,3-dihydric mouth-1-benzofuran-3- Yl) methyl acetate
- Example 6 2 (6- ⁇ [4,-(2-Ethylbutoxy) -2-,-methylbiphenyl-3-yl] methoxy ⁇ -2,3-dihydro-1-benzobenzofuran-3-yl) Acetic acid
- Example 6 (6- ⁇ [4,-(2-ethylpytoxy) -2′-methylbiphenyl-3-yl] methoxy ⁇ -2,3-dihydrobenzobenzofuran-3-y L)
- the title compound was obtained as colorless prism crystals from methyl acetate. Yield 93% (recovered from ethyl hexane monoacetate) crystal).
- Example 6 4 ⁇ 6-[(4- ⁇ [(2-phenylethyl) (4-phenyl-1,3-thiazol-2-yl) amino] methyl ⁇ benzyl) oxy] -2,3-dihydro- 1-Benzofuran-3-yl ⁇ vinegar
- Example 7 1 (6- ⁇ [4,-(2-ethoxyethoxy) -2 ', 6, -dimethylbiphenyl-3-yl] methoxy ⁇ -2,3-dihydro-1-benzobenzofuran-3-y R) Methyl acetate
- Example 7 2 (6- ⁇ [4,-(2-ethoxyxetoxy) -2,6, -dimethylbiphenyl-3-yl] methoxy ⁇ -2,3-dihydro-1-benzobenzofuran-3 -Yl) acetic acid
- Example 73 (6- ⁇ [4,-(2-ethoxyxetoxy) -2,6, -dimethylbiphenyl-3-yl] methoxy ⁇ -2,3-dihydrogen-1-benzofuran-3- Yl) calcium acetate
- Example 7 5 (6- ⁇ [6- (benzyloxy) -2,, 6, -dimethylbiphenyl-3-yl] methoxy ⁇ -2,3-dihydro-1-benzobenzofuran-3-yl ) Acetic acid
- Example 7 6 ⁇ 5-[(2,6, -Dimethylbiphenyl-3-yl) methoxy] -1H-indole-rootinole ⁇ ethyl acetate
- reaction solution was diluted with ethyl acetate, washed sequentially with aqueous citrate, water and brine, dried over magnesium sulfate, and concentrated under reduced pressure.
- Example 7 ⁇ 2-[(3-Phenoxybenzyl) thio] -4,5,6,7-tetrahydro-1,3-beta
- Jetyl malonate (1.21 g, 2.36
- a mixture of (hard ol), 37% hydrochloric acid (10 mL) and acetic acid (10 mL) was heated to 120 ° C and stirred for 12 hours.
- the reaction mixture was concentrated, diluted with ethyl acetate, washed successively with 10% aqueous sodium hydrogen carbonate solution and saturated saline, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure to give the title compound (890 mg, 92%) Was obtained as a pale yellow oil.
- Example 8 1 Methyl ⁇ 6-[(2-phenyl-1,3-thiazol-4-yl) methoxy] -2,3-dihydro-1-benzobenzo-3-yl ⁇ acetate
- Example 8 4. ⁇ 6-[(2-Pyrazine-2-yl-1,3-thiazol-4-yl) methoxy] -2,3-dihydro-1-benzofuran-3-yl ⁇ acetic acid
- Example 8 6 ⁇ 6- [3- (2-Feryl-1H-indole-1-yl) propoxy] -2,3-dihydrofluoride ⁇ 1-benzobenzo-3-yl ⁇ acetic acid
- Example 8 7 (6- ⁇ [4- (2-benzoyl-4-chlorophenyl) -4H-1,2,4-triazol-3-yl] methoxy ⁇ -2,3-dihydroxy Dro-1-benzofuran-3-yl) acetic acid
- Example 8 (6-[cyclohexyl (2-trobenzyl) amino] ethoxy 2,3-dihydro-1-benzofuran-3-yl) acetic acid trifluoroacetate
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP04745580A EP1630152A4 (en) | 2003-05-30 | 2004-05-28 | CONNECTION WITH CONDENSED RING |
| US10/558,846 US7820837B2 (en) | 2003-05-30 | 2004-05-28 | Condensed ring compound |
| CA2527691A CA2527691C (en) | 2003-05-30 | 2004-05-28 | Condensed ring compound |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003153986 | 2003-05-30 | ||
| JP2003-153986 | 2003-05-30 | ||
| JP2004-139144 | 2004-05-07 | ||
| JP2004139144 | 2004-05-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004106276A1 true WO2004106276A1 (ja) | 2004-12-09 |
Family
ID=33492446
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2004/007770 WO2004106276A1 (ja) | 2003-05-30 | 2004-05-28 | 縮合環化合物 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US7820837B2 (ja) |
| EP (1) | EP1630152A4 (ja) |
| CA (1) | CA2527691C (ja) |
| WO (1) | WO2004106276A1 (ja) |
Cited By (70)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1726580A1 (en) * | 2004-03-15 | 2006-11-29 | Takeda Pharmaceutical Company Limited | Aminophenylpropanoic acid derivative |
| WO2007033002A1 (en) * | 2005-09-14 | 2007-03-22 | Amgen Inc. | Conformationally constrained 3- (4-hydroxy-phenyl) - substituted-propanoic acids useful for treating metabolic disorders |
| WO2006127503A3 (en) * | 2005-05-20 | 2007-08-30 | Amgen Inc | Compounds, pharmaceutical compositions and methods for their use in treating metabolic disorders |
| WO2008066097A1 (en) | 2006-12-01 | 2008-06-05 | Astellas Pharma Inc. | Carboxylic acid derivative |
| US7572934B2 (en) | 2007-04-16 | 2009-08-11 | Amgen Inc. | Substituted biphenyl GPR40 modulators |
| US7649110B2 (en) | 2004-02-27 | 2010-01-19 | Amgen Inc. | Compounds, pharmaceutical compositions and methods for use in treating metabolic disorders |
| US7687526B2 (en) | 2006-09-07 | 2010-03-30 | Amgen Inc. | Benzo-fused compounds for use in treating metabolic disorders |
| WO2010035806A1 (ja) | 2008-09-25 | 2010-04-01 | 武田薬品工業株式会社 | 固形医薬組成物 |
| WO2010050445A1 (ja) | 2008-10-27 | 2010-05-06 | 武田薬品工業株式会社 | 二環性化合物 |
| US7714008B2 (en) | 2006-09-07 | 2010-05-11 | Amgen Inc. | Heterocyclic GPR40 modulators |
| US7732626B2 (en) | 2006-06-27 | 2010-06-08 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds |
| WO2010076884A1 (ja) | 2008-12-29 | 2010-07-08 | 武田薬品工業株式会社 | 新規縮合環化合物およびその用途 |
| WO2010080864A1 (en) | 2009-01-12 | 2010-07-15 | Array Biopharma Inc. | Piperidine-containing compounds and use thereof |
| WO2010123016A1 (ja) | 2009-04-22 | 2010-10-28 | アステラス製薬株式会社 | カルボン酸化合物 |
| WO2010143733A1 (en) | 2009-06-09 | 2010-12-16 | Takeda Pharmaceutical Company Limited | Novel fused cyclic compound and use thereof |
| WO2011013639A1 (ja) | 2009-07-28 | 2011-02-03 | 武田薬品工業株式会社 | 錠剤 |
| US7932289B2 (en) | 2004-07-27 | 2011-04-26 | Takeda Pharmaceutical Company Limited | Remedy for diabetes |
| US8030354B2 (en) | 2007-10-10 | 2011-10-04 | Amgen Inc. | Substituted biphenyl GPR40 modulators |
| WO2011136385A1 (en) | 2010-04-27 | 2011-11-03 | Takeda Pharmaceutical Company Limited | Bicyclic compound derivatives and their use as acc inhibitors. |
| WO2011158880A1 (ja) | 2010-06-16 | 2011-12-22 | 武田薬品工業株式会社 | アミド化合物の結晶 |
| JP2012506386A (ja) * | 2008-10-21 | 2012-03-15 | メタボレックス, インコーポレイテッド | アリールgpr120受容体アゴニストおよびその使用 |
| WO2012036293A1 (ja) | 2010-09-17 | 2012-03-22 | 武田薬品工業株式会社 | 糖尿病治療剤 |
| WO2012074126A1 (en) | 2010-11-30 | 2012-06-07 | Takeda Pharmaceutical Company Limited | Bicyclic compound |
| WO2012111849A1 (en) | 2011-02-17 | 2012-08-23 | Takeda Pharmaceutical Company Limited | Production method of optically active dihydrobenzofuran derivative |
| WO2013061962A1 (ja) | 2011-10-24 | 2013-05-02 | 武田薬品工業株式会社 | 二環性化合物 |
| US8450522B2 (en) | 2008-03-06 | 2013-05-28 | Amgen Inc. | Conformationally constrained carboxylic acid derivatives useful for treating metabolic disorders |
| WO2013105676A1 (en) | 2012-01-12 | 2013-07-18 | Takeda Pharmaceutical Company Limited | Benzimidazole derivatives as mch receptor antagonists |
| WO2013104257A1 (zh) | 2012-01-12 | 2013-07-18 | 江苏恒瑞医药股份有限公司 | 多环类衍生物、其制备方法及其在医药上的应用 |
| WO2013122029A1 (ja) | 2012-02-13 | 2013-08-22 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013122260A1 (en) | 2012-02-15 | 2013-08-22 | Takeda Pharmaceutical Company Limited | Tablet |
| WO2013122028A1 (ja) | 2012-02-13 | 2013-08-22 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013125732A1 (en) | 2012-02-24 | 2013-08-29 | Takeda Pharmaceutical Company Limited | Aromatic ring compound |
| WO2013147026A1 (ja) | 2012-03-29 | 2013-10-03 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013168759A1 (ja) | 2012-05-10 | 2013-11-14 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013168760A1 (ja) | 2012-05-10 | 2013-11-14 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013183784A1 (en) | 2012-06-05 | 2013-12-12 | Takeda Pharmaceutical Company Limited | Solid preparation |
| WO2014011926A1 (en) | 2012-07-11 | 2014-01-16 | Elcelyx Therapeutics, Inc. | Compositions comprising statins, biguanides and further agents for reducing cardiometabolic risk |
| WO2014073904A1 (en) | 2012-11-09 | 2014-05-15 | Lg Life Sciences Ltd. | Gpr40 receptor agonist, methods of preparing the same, and pharmaceutical compositions containing the same as an active ingredient |
| US8748462B2 (en) | 2008-10-15 | 2014-06-10 | Amgen Inc. | Spirocyclic GPR40 modulators |
| CN103998451A (zh) * | 2011-10-20 | 2014-08-20 | 葛兰素史密斯克莱有限责任公司 | 用作沉默调节蛋白调节剂的取代的双环氮杂杂环化合物和类似物 |
| WO2014133361A1 (ko) | 2013-02-28 | 2014-09-04 | 에스케이케미칼주식회사 | 삼환식 화합물 및 이의 용도 |
| WO2014142363A1 (en) | 2013-03-14 | 2014-09-18 | Takeda Pharmaceutical Company Limited | Spiro azetidine isoxazole derivatives and their use as sstr5 antagonists |
| WO2015005489A1 (en) | 2013-07-09 | 2015-01-15 | Takeda Pharmaceutical Company Limited | Heterocyclic compound |
| WO2015020184A1 (ja) | 2013-08-09 | 2015-02-12 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2015052910A1 (en) | 2013-10-07 | 2015-04-16 | Takeda Pharmaceutical Company Limited | Antagonists of somatostatin receptor subtype 5 (sstr5) |
| JP2015511612A (ja) * | 2012-03-26 | 2015-04-20 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | 新しいインダニルオキシジヒドロベンゾフラニル酢酸誘導体およびgpr40受容体作動薬としてのそれらの使用 |
| WO2015062486A1 (en) * | 2013-10-31 | 2015-05-07 | Sunshine Lake Pharma Co., Ltd. | Biphenyl compounds and uses thereof |
| WO2015122187A1 (en) | 2014-02-13 | 2015-08-20 | Takeda Pharmaceutical Company Limited | Fused heterocyclic compound |
| WO2015122188A1 (en) | 2014-02-13 | 2015-08-20 | Takeda Pharmaceutical Company Limited | Heterocyclic compound |
| US10005720B2 (en) | 2013-04-05 | 2018-06-26 | North Carolina Central University | Compounds useful for the treatment of metabolic disorders and synthesis of the same |
| WO2018182050A1 (en) | 2017-03-31 | 2018-10-04 | Takeda Pharmaceutical Company Limited | Substituted cyclyl-acetic acid derivatives for the treatment of metabolic disorders |
| WO2018182051A1 (en) | 2017-03-30 | 2018-10-04 | Takeda Pharmaceutical Company Limited | Ip6k inhibitors |
| WO2018181864A1 (en) | 2017-03-31 | 2018-10-04 | Takeda Pharmaceutical Company Limited | Gip receptor activating peptide |
| WO2018181847A1 (ja) | 2017-03-31 | 2018-10-04 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2019134984A1 (en) | 2018-01-08 | 2019-07-11 | Celon Pharma S.A. | 3-phenyl-4-hexynoic acid derivatives as gpr40 agonists |
| PL424227A1 (pl) * | 2018-01-08 | 2019-07-15 | Celon Pharma Spółka Akcyjna | Pochodne kwasu 3-fenylo-4-heksynowego jako agoniści GPR40 |
| WO2020045326A1 (ja) | 2018-08-27 | 2020-03-05 | 株式会社スコヒアファーマ | 安息香酸エステル化合物 |
| WO2020067575A1 (en) | 2018-09-24 | 2020-04-02 | Takeda Pharmaceutical Company Limited | Gip receptor agonist peptide compounds and uses thereof |
| WO2020067557A2 (en) | 2018-09-24 | 2020-04-02 | Takeda Pharmaceutical Company Limited | Gip receptor agonist peptide compounds and uses thereof |
| WO2020166662A1 (ja) | 2019-02-13 | 2020-08-20 | 株式会社ジェクスヴァル | 骨疾患の予防または治療用の医薬組成物 |
| WO2021193983A2 (en) | 2020-03-25 | 2021-09-30 | Takeda Pharmaceutical Company Limited | Qw dosing of gip receptor agonist peptide compounds and uses thereof |
| WO2021193984A2 (en) | 2020-03-25 | 2021-09-30 | Takeda Pharmaceutical Company Limited | Qd dosing of gip receptor agonist peptide compounds and uses thereof |
| WO2022034664A1 (ja) | 2020-08-12 | 2022-02-17 | 株式会社ジェクスヴァル | 骨疾患の予防または治療用の医薬組成物 |
| WO2022053013A1 (zh) | 2020-09-10 | 2022-03-17 | 上海爱博医药科技有限公司 | 苯并含氧杂环类化合物及其医药应用 |
| WO2022241287A2 (en) | 2021-05-13 | 2022-11-17 | Carmot Therapeutics, Inc. | Modulators of g-protein coupled receptors |
| US11535660B1 (en) | 2018-03-23 | 2022-12-27 | Cannot Therapeutics, Inc. | Modulators of G-protein coupled receptors |
| US11583528B2 (en) | 2016-02-17 | 2023-02-21 | Children's Medical Center Corporation | FFA1 (GPR40) as a therapeutic target for neural angiogenesis diseases or disorders |
| WO2023169456A1 (en) | 2022-03-09 | 2023-09-14 | Gasherbrum Bio , Inc. | Heterocyclic glp-1 agonists |
| WO2023179542A1 (en) | 2022-03-21 | 2023-09-28 | Gasherbrum Bio , Inc. | 5,8-dihydro-1,7-naphthyridine derivatives as glp-1 agonists for the treatment of diabetes |
| WO2023198140A1 (en) | 2022-04-14 | 2023-10-19 | Gasherbrum Bio, Inc. | Heterocyclic glp-1 agonists |
Families Citing this family (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007106469A2 (en) * | 2006-03-14 | 2007-09-20 | Amgen Inc. | Bicyclic carboxylic acid derivatives useful for treating metabolic disorders |
| JP2010508268A (ja) * | 2006-10-31 | 2010-03-18 | メルク エンド カムパニー インコーポレーテッド | 抗糖尿病二環式化合物 |
| BRPI0807552A2 (pt) | 2007-02-22 | 2014-07-01 | Irm Llc | Compostos e métodos para modular receptores acoplados à protéina g |
| JP5420400B2 (ja) * | 2007-04-26 | 2014-02-19 | 国立大学法人京都大学 | Gタンパク質共役型レセプター作動剤 |
| TW200932219A (en) * | 2007-10-24 | 2009-08-01 | Astellas Pharma Inc | Oxadiazolidinedione compound |
| AU2008314933B2 (en) * | 2007-10-26 | 2013-05-02 | Japan Tobacco Inc. | Spiro-ring compound and use thereof for medical purposes |
| JP5542058B2 (ja) * | 2007-10-29 | 2014-07-09 | メルク・シャープ・アンド・ドーム・コーポレーション | 抗糖尿病性三環式化合物 |
| US8455500B2 (en) | 2009-10-30 | 2013-06-04 | Mochida Pharmaceutical Co., Ltd. | 3-hydroxy-5-arylisoxazole derivative |
| MX2012004990A (es) | 2009-10-30 | 2012-06-12 | Janssen Pharmaceutica Nv | Deribados de imidazo [1,2-b] pirimideazina y su uso como inhibidores de la enzima fosfodiesterasa 10. |
| MX2012007474A (es) | 2009-12-25 | 2012-08-01 | Mochida Pharm Co Ltd | Derivados de 3-hidroxi-5-arilisotiazol novedosos. |
| AR080754A1 (es) | 2010-03-09 | 2012-05-09 | Janssen Pharmaceutica Nv | Derivados de imidazo (1,2-a) pirazina y su uso como inhibidores de pde10 |
| US9040525B2 (en) | 2010-10-08 | 2015-05-26 | Mochida Pharmaceutical Co., Ltd. | Cyclic amide derivative |
| AU2012248629A1 (en) | 2011-04-27 | 2013-11-28 | Mochida Pharmaceutical Co., Ltd. | Novel 3-hydroxyisothiazole 1-oxide derivative |
| WO2012147516A1 (ja) | 2011-04-28 | 2012-11-01 | 持田製薬株式会社 | 環状アミド誘導体 |
| EP2723744B1 (en) | 2011-06-27 | 2016-03-23 | Janssen Pharmaceutica, N.V. | 1-ARYL-4-METHYL-[1,2,4]TRIAZOLO[4,3-a]QUINOXALINE DERIVATIVES |
| CN103012343B (zh) * | 2011-09-26 | 2015-04-08 | 上海恒瑞医药有限公司 | 稠合环类衍生物、其制备方法及其在医药上的应用 |
| US8809376B2 (en) | 2012-04-30 | 2014-08-19 | Boehringer Ingelheim International Gmbh | Indanyloxydihydrobenzofuranylacetic acids |
| JP6426603B2 (ja) | 2012-06-26 | 2018-11-21 | ヤンセン ファーマシューティカ エヌ.ベー. | 神経障害または代謝障害の治療に使用するための、1−アリール−4−メチル−[1,2,4]トリアゾロ[4,3−a]−キノキサリン化合物などのPDE2阻害剤とPDE10阻害剤との組合せ |
| EP2869822B1 (en) | 2012-07-09 | 2016-09-14 | Janssen Pharmaceutica, N.V. | Inhibitors of phosphodiesterase 10 enzyme |
| JP2015525782A (ja) | 2012-08-02 | 2015-09-07 | メルク・シャープ・アンド・ドーム・コーポレーションMerck Sharp & Dohme Corp. | 抗糖尿病性三環式化合物 |
| KR101942752B1 (ko) * | 2012-11-05 | 2019-01-28 | 주식회사 엘지화학 | Gpr120 효능제로서의 티오아릴 유도체 |
| CN102924406B (zh) * | 2012-11-07 | 2014-11-05 | 南京医科大学 | 取代芳氧乙基哌嗪类衍生物及其制备方法和应用 |
| US20140163025A1 (en) | 2012-12-07 | 2014-06-12 | Boehringer Ingelheim International Gmbh | Indanyloxydihydrobenzofuranylacetic acids |
| CN104994848A (zh) | 2013-02-22 | 2015-10-21 | 默沙东公司 | 抗糖尿病二环化合物 |
| CN103145663B (zh) * | 2013-03-01 | 2016-03-23 | 上海高准医药有限公司 | (s)-2-(2,3-二氢苯并呋喃-3-基)乙酸衍生物,其制备方法及其在医药上的应用 |
| CN104870429B (zh) * | 2013-05-22 | 2017-05-03 | 四川海思科制药有限公司 | 苯并呋喃衍生物、其制备方法及其在医药上的应用 |
| CN104250239B (zh) * | 2013-06-29 | 2016-09-07 | 山东轩竹医药科技有限公司 | 芳香多环羧酸衍生物 |
| WO2015000412A1 (zh) * | 2013-07-02 | 2015-01-08 | 四川海思科制药有限公司 | 苯并环丁烯类衍生物、其制备方法及其在医药上的应用 |
| WO2015010655A1 (zh) * | 2013-07-26 | 2015-01-29 | 四川海思科制药有限公司 | 三元并环羧酸类衍生物、其制备方法及其在医药上的应用 |
| WO2015051496A1 (en) | 2013-10-08 | 2015-04-16 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| CA2928097A1 (en) | 2013-11-14 | 2015-07-02 | Cadila Healthcare Limited | Novel heterocyclic compounds |
| TW201609722A (zh) | 2013-12-13 | 2016-03-16 | 美國禮來大藥廠 | 新穎三唑并吡啶化合物 |
| WO2015089809A1 (en) | 2013-12-19 | 2015-06-25 | Merck Sharp & Dohme Corp. | Antidiabetic substituted heteroaryl compounds |
| CN104788412B (zh) * | 2014-01-22 | 2017-02-15 | 山东轩竹医药科技有限公司 | 芳香多环羧酸衍生物 |
| WO2016022742A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| EP3177282B1 (en) | 2014-08-08 | 2021-10-06 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2016019587A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | [7, 6]-fused bicyclic antidiabetic compounds |
| WO2016022446A1 (en) | 2014-08-08 | 2016-02-11 | Merck Sharp & Dohme Corp. | [5,6]-fused bicyclic antidiabetic compounds |
| CN105017242B (zh) * | 2015-07-22 | 2018-06-08 | 中国药科大学 | 新型联苯杂环类衍生物、其制备方法及其作为药物的用途 |
| CN106478616B (zh) * | 2015-08-27 | 2020-05-12 | 江苏恒瑞医药股份有限公司 | 一种gpr40激动剂的结晶形式及其制备方法 |
| CN105566263A (zh) * | 2016-01-27 | 2016-05-11 | 中国药科大学 | 一类新型含氮杂环衍生物、其制备方法及其作为药物的用途 |
| WO2018077699A1 (en) | 2016-10-25 | 2018-05-03 | Boehringer Ingelheim International Gmbh | Benzylaminopyridylcyclopropanecarboxylic acids, pharmaceutical compositions and uses thereof |
| WO2018138030A1 (en) | 2017-01-26 | 2018-08-02 | Boehringer Ingelheim International Gmbh | Benzyloxypyrazinylcyclopropanecarboxylic acids, pharmaceutical compositions and uses thereof |
| WO2018138027A1 (en) | 2017-01-26 | 2018-08-02 | Boehringer Ingelheim International Gmbh | Benzylaminopyridylcyclopropanecarboxylic acids, pharmaceuticalcompositions and uses thereof |
| US10793530B2 (en) | 2017-01-26 | 2020-10-06 | Boehringer Ingelheim International Gmbh | Benzylaminopyrazinylcyclopropanecarboxylic acids, pharmaceutical compositions and uses thereof |
| US10913720B2 (en) | 2017-01-26 | 2021-02-09 | Boehringer Ingelheim International Gmbh | Benzyloxypyridylcyclopropanecarboxylic acids, pharmaceutical compositions and uses thereof |
| JP7049353B2 (ja) | 2017-02-08 | 2022-04-06 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | 糖尿病の治療のためのインダニルアミノアザジヒドロベンゾフラニル酢酸、医薬組成物 |
| WO2019099315A1 (en) | 2017-11-16 | 2019-05-23 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| TW202033500A (zh) * | 2018-12-06 | 2020-09-16 | 大陸商上海濟煜醫藥科技有限公司 | 作為免疫調節的芳環衍生物及其製備方法和應用 |
| WO2022002225A1 (zh) * | 2020-07-03 | 2022-01-06 | 上海美迪西生物医药股份有限公司 | 一种吲哚衍生物及其应用 |
| CN112480047B (zh) * | 2020-12-01 | 2023-06-02 | 云南省第二人民医院 | 一种具有降糖调脂作用的化合物及其制剂与应用 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001047859A1 (en) * | 1999-12-07 | 2001-07-05 | Karo Bio Ab | Compounds active at the glucocorticoid receptor |
| WO2002057783A2 (en) * | 2000-12-22 | 2002-07-25 | Glaxo Group Limited | Method of screening for gpr40 ligands |
| WO2002083616A1 (fr) * | 2001-04-10 | 2002-10-24 | Sankyo Company, Limited | DERIVE D'ACIDE GRAS φomega;-ARYLE α-SUBSTITUE |
Family Cites Families (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1019378B1 (en) | 1996-07-01 | 2004-09-29 | Eli Lilly And Company | Hypoglycemic and hypolipidemic compounds |
| DK0971878T3 (da) | 1997-02-27 | 2008-06-30 | Takeda Pharmaceutical | Aminforbindelser, fremstillingen og anvendelsen deraf som inhibitorer af amyloid-beta-produktion |
| WO1999011255A1 (fr) | 1997-08-28 | 1999-03-11 | Ono Pharmaceutical Co., Ltd. | Regulateurs du recepteur active par les agents de proliferation des peroxysomes |
| HUP0200997A3 (en) | 1999-04-28 | 2003-03-28 | Sanofi Aventis Deutschland | Tri-aryl acid derivatives as ppar receptor ligands and pharmaceutical compositions containing them |
| JP4053199B2 (ja) | 1999-10-22 | 2008-02-27 | 三井化学株式会社 | ベンジルエーテル誘導体、液晶材料、液晶組成物、および液晶素子 |
| CA2402315A1 (en) | 2000-03-09 | 2001-09-13 | Michael Jaye | Therapeutic uses of ppar mediators |
| IL153461A0 (en) * | 2000-06-28 | 2003-07-06 | Tularik Inc | Quinolinyl and benzothiazolyl derivatives and pharmaceutical compositions containing the same |
| CA2423141A1 (en) * | 2000-09-27 | 2002-04-04 | Merck & Co., Inc. | Benzopyrancarboxylic acid derivatives for the treatment of diabetes and lipid disorders |
| AU2002248221B2 (en) * | 2000-10-31 | 2006-08-17 | Merck & Co., Inc. | Benzopyrancarboxylic acid derivatives for the treatment of diabetes and lipid disorders |
| WO2002053547A1 (fr) | 2000-12-28 | 2002-07-11 | Takeda Chemical Industries, Ltd. | Derives d'acide alcanoique, procede de production et utilisation correspondants |
| CZ20033415A3 (cs) * | 2001-05-29 | 2004-06-16 | Kyoto Pharmaceutical Industries, Ltd. | Nové heterocyklické deriváty a jejich lékařské použití |
| AR036237A1 (es) | 2001-07-27 | 2004-08-25 | Bayer Corp | Derivados del acido indan acetico, intermediarios, y metodo para su preparacion, composicion farmaceutica y el uso de dichos derivados para la manufactura de un medicamento |
| US6867224B2 (en) | 2002-03-07 | 2005-03-15 | Warner-Lambert Company | Compounds that modulate PPAR activity and methods of preparation |
| US6875780B2 (en) * | 2002-04-05 | 2005-04-05 | Warner-Lambert Company | Compounds that modulate PPAR activity and methods for their preparation |
| UA79755C2 (en) | 2002-04-16 | 2007-07-25 | Bayer Pharmaceuticals Corp | Indane acetic acid derivatives and their use as pharmaceutical agents, intermediates, and method of preparation |
| EP1513817A1 (en) | 2002-05-24 | 2005-03-16 | Takeda Pharmaceutical Company Limited | 1, 2-azole derivatives with hypoglycemic and hypolipidemic activity |
| WO2004011446A1 (en) | 2002-07-26 | 2004-02-05 | Bayer Pharmaceuticals Corporation | Indane, dihydrobenzofuran, and tetrahydronaphthalene carboxylic acid derivatives and their use as antidiabetics |
| EP1539136B1 (en) | 2002-07-30 | 2008-07-09 | Merck & Co., Inc. | Ppar alpha selective compounds for the treatment of dyslipidemia and other lipid disorders |
| WO2004019869A2 (en) * | 2002-08-29 | 2004-03-11 | Merck & Co., Inc. | Indoles having anti-diabetic activity |
| AU2003261935A1 (en) | 2002-09-06 | 2004-03-29 | Takeda Pharmaceutical Company Limited | Furan or thiophene derivative and medicinal use thereof |
| EP1559422B1 (en) | 2002-11-08 | 2014-04-30 | Takeda Pharmaceutical Company Limited | Receptor function controlling agent |
| EP1578715B1 (en) * | 2002-12-20 | 2011-03-02 | Bayer Pharmaceuticals Corporation | Indane acetic acid derivatives and their use as pharmaceutical agents, intermediates, and method of preparation |
| WO2004082601A2 (en) * | 2003-03-13 | 2004-09-30 | Bayer Pharmaceuticals Corporation | Method of treating diabetes and diabetes-related disorders |
| US7244763B2 (en) * | 2003-04-17 | 2007-07-17 | Warner Lambert Company Llc | Compounds that modulate PPAR activity and methods of preparation |
| EP1620088A4 (en) * | 2003-04-28 | 2007-08-29 | Bayer Pharmaceuticals Corp | ACETIC INDOLE ACID DERIVATIVES AND THEIR USE AS PHARMACEUTICAL AGENTS |
| JP4875978B2 (ja) | 2004-03-15 | 2012-02-15 | 武田薬品工業株式会社 | アミノフェニルプロパン酸誘導体 |
| EP1749000A4 (en) * | 2004-05-25 | 2009-12-30 | Metabolex Inc | BICYCLIC SUBSTITUTED TRIAZOLE AS PPAR MODULATORS AND METHOD FOR THE PRODUCTION THEREOF |
-
2004
- 2004-05-28 EP EP04745580A patent/EP1630152A4/en not_active Withdrawn
- 2004-05-28 US US10/558,846 patent/US7820837B2/en not_active Expired - Fee Related
- 2004-05-28 WO PCT/JP2004/007770 patent/WO2004106276A1/ja active Application Filing
- 2004-05-28 CA CA2527691A patent/CA2527691C/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001047859A1 (en) * | 1999-12-07 | 2001-07-05 | Karo Bio Ab | Compounds active at the glucocorticoid receptor |
| WO2002057783A2 (en) * | 2000-12-22 | 2002-07-25 | Glaxo Group Limited | Method of screening for gpr40 ligands |
| WO2002083616A1 (fr) * | 2001-04-10 | 2002-10-24 | Sankyo Company, Limited | DERIVE D'ACIDE GRAS φomega;-ARYLE α-SUBSTITUE |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1630152A4 * |
Cited By (89)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7816367B2 (en) | 2004-02-27 | 2010-10-19 | Amgen Inc. | Compounds, pharmaceutical compositions and methods for use in treating metabolic disorders |
| US7649110B2 (en) | 2004-02-27 | 2010-01-19 | Amgen Inc. | Compounds, pharmaceutical compositions and methods for use in treating metabolic disorders |
| EP1726580A4 (en) * | 2004-03-15 | 2008-02-13 | Takeda Pharmaceutical | AMINOPHNYLPROPANO ACID DERIVATIVE |
| US7786165B2 (en) | 2004-03-15 | 2010-08-31 | Takeda Pharmaceutical Company Limited | Aminophenylpropanoic acid derivative |
| EP1726580A1 (en) * | 2004-03-15 | 2006-11-29 | Takeda Pharmaceutical Company Limited | Aminophenylpropanoic acid derivative |
| US7932289B2 (en) | 2004-07-27 | 2011-04-26 | Takeda Pharmaceutical Company Limited | Remedy for diabetes |
| WO2006127503A3 (en) * | 2005-05-20 | 2007-08-30 | Amgen Inc | Compounds, pharmaceutical compositions and methods for their use in treating metabolic disorders |
| US7465804B2 (en) | 2005-05-20 | 2008-12-16 | Amgen Inc. | Compounds, pharmaceutical compositions and methods for their use in treating metabolic disorders |
| WO2007033002A1 (en) * | 2005-09-14 | 2007-03-22 | Amgen Inc. | Conformationally constrained 3- (4-hydroxy-phenyl) - substituted-propanoic acids useful for treating metabolic disorders |
| US7582803B2 (en) | 2005-09-14 | 2009-09-01 | Amgen Inc. | Conformationally constrained 3-(4-hydroxy-phenyl)-substituted-propanoic acids useful for treating metabolic disorders |
| US8598226B2 (en) | 2006-06-27 | 2013-12-03 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds |
| US7732626B2 (en) | 2006-06-27 | 2010-06-08 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds |
| US8492430B2 (en) | 2006-06-27 | 2013-07-23 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds |
| EP2431367A2 (en) | 2006-06-27 | 2012-03-21 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds as GPR40 receptor modulators |
| US8088821B2 (en) | 2006-06-27 | 2012-01-03 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds |
| EP2248812A2 (en) | 2006-06-27 | 2010-11-10 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds as GPR40 receptor modulators |
| EP2743268A2 (en) | 2006-06-27 | 2014-06-18 | Takeda Pharmaceutical Company Limited | Fused cyclic compounds as GPR40 receptor modulators |
| US7687526B2 (en) | 2006-09-07 | 2010-03-30 | Amgen Inc. | Benzo-fused compounds for use in treating metabolic disorders |
| US7714008B2 (en) | 2006-09-07 | 2010-05-11 | Amgen Inc. | Heterocyclic GPR40 modulators |
| US8003648B2 (en) | 2006-09-07 | 2011-08-23 | Amgen Inc. | Heterocyclic GPR40 modulators |
| WO2008066097A1 (en) | 2006-12-01 | 2008-06-05 | Astellas Pharma Inc. | Carboxylic acid derivative |
| US7572934B2 (en) | 2007-04-16 | 2009-08-11 | Amgen Inc. | Substituted biphenyl GPR40 modulators |
| US8030354B2 (en) | 2007-10-10 | 2011-10-04 | Amgen Inc. | Substituted biphenyl GPR40 modulators |
| US8450522B2 (en) | 2008-03-06 | 2013-05-28 | Amgen Inc. | Conformationally constrained carboxylic acid derivatives useful for treating metabolic disorders |
| WO2010035806A1 (ja) | 2008-09-25 | 2010-04-01 | 武田薬品工業株式会社 | 固形医薬組成物 |
| US8748462B2 (en) | 2008-10-15 | 2014-06-10 | Amgen Inc. | Spirocyclic GPR40 modulators |
| JP2012506386A (ja) * | 2008-10-21 | 2012-03-15 | メタボレックス, インコーポレイテッド | アリールgpr120受容体アゴニストおよびその使用 |
| WO2010050445A1 (ja) | 2008-10-27 | 2010-05-06 | 武田薬品工業株式会社 | 二環性化合物 |
| WO2010076884A1 (ja) | 2008-12-29 | 2010-07-08 | 武田薬品工業株式会社 | 新規縮合環化合物およびその用途 |
| WO2010080864A1 (en) | 2009-01-12 | 2010-07-15 | Array Biopharma Inc. | Piperidine-containing compounds and use thereof |
| WO2010123016A1 (ja) | 2009-04-22 | 2010-10-28 | アステラス製薬株式会社 | カルボン酸化合物 |
| WO2010143733A1 (en) | 2009-06-09 | 2010-12-16 | Takeda Pharmaceutical Company Limited | Novel fused cyclic compound and use thereof |
| WO2011013639A1 (ja) | 2009-07-28 | 2011-02-03 | 武田薬品工業株式会社 | 錠剤 |
| WO2011136385A1 (en) | 2010-04-27 | 2011-11-03 | Takeda Pharmaceutical Company Limited | Bicyclic compound derivatives and their use as acc inhibitors. |
| WO2011158880A1 (ja) | 2010-06-16 | 2011-12-22 | 武田薬品工業株式会社 | アミド化合物の結晶 |
| WO2012036293A1 (ja) | 2010-09-17 | 2012-03-22 | 武田薬品工業株式会社 | 糖尿病治療剤 |
| WO2012074126A1 (en) | 2010-11-30 | 2012-06-07 | Takeda Pharmaceutical Company Limited | Bicyclic compound |
| US8729102B2 (en) | 2010-11-30 | 2014-05-20 | Takeda Pharmaceutical Company Limited | Bicyclic compound |
| WO2012111849A1 (en) | 2011-02-17 | 2012-08-23 | Takeda Pharmaceutical Company Limited | Production method of optically active dihydrobenzofuran derivative |
| CN103998451A (zh) * | 2011-10-20 | 2014-08-20 | 葛兰素史密斯克莱有限责任公司 | 用作沉默调节蛋白调节剂的取代的双环氮杂杂环化合物和类似物 |
| WO2013061962A1 (ja) | 2011-10-24 | 2013-05-02 | 武田薬品工業株式会社 | 二環性化合物 |
| JP2015503595A (ja) * | 2012-01-12 | 2015-02-02 | ジエンス ヘンルイ メデイシンカンパニー リミテッドJiangsu Hengrui Medicine Co.,Ltd. | 多環式化合物誘導体、それらの製造方法および医薬用途 |
| WO2013105676A1 (en) | 2012-01-12 | 2013-07-18 | Takeda Pharmaceutical Company Limited | Benzimidazole derivatives as mch receptor antagonists |
| WO2013104257A1 (zh) | 2012-01-12 | 2013-07-18 | 江苏恒瑞医药股份有限公司 | 多环类衍生物、其制备方法及其在医药上的应用 |
| US9139548B2 (en) | 2012-01-12 | 2015-09-22 | Jiangsu Hengrui Medicine Co., Ltd. | Polycyclic derivatives, preparation process and pharmaceutical use thereof |
| WO2013122029A1 (ja) | 2012-02-13 | 2013-08-22 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013122028A1 (ja) | 2012-02-13 | 2013-08-22 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013122260A1 (en) | 2012-02-15 | 2013-08-22 | Takeda Pharmaceutical Company Limited | Tablet |
| WO2013125732A1 (en) | 2012-02-24 | 2013-08-29 | Takeda Pharmaceutical Company Limited | Aromatic ring compound |
| JP2015511612A (ja) * | 2012-03-26 | 2015-04-20 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | 新しいインダニルオキシジヒドロベンゾフラニル酢酸誘導体およびgpr40受容体作動薬としてのそれらの使用 |
| WO2013147026A1 (ja) | 2012-03-29 | 2013-10-03 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013168759A1 (ja) | 2012-05-10 | 2013-11-14 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013168760A1 (ja) | 2012-05-10 | 2013-11-14 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2013183784A1 (en) | 2012-06-05 | 2013-12-12 | Takeda Pharmaceutical Company Limited | Solid preparation |
| WO2014011926A1 (en) | 2012-07-11 | 2014-01-16 | Elcelyx Therapeutics, Inc. | Compositions comprising statins, biguanides and further agents for reducing cardiometabolic risk |
| JP2016503409A (ja) * | 2012-11-09 | 2016-02-04 | エルジー・ライフ・サイエンシーズ・リミテッドLG Life Sciences Ltd. | Gpr40受容体アゴニスト、この製造方法およびこれを活性成分として含有する医薬組成物 |
| WO2014073904A1 (en) | 2012-11-09 | 2014-05-15 | Lg Life Sciences Ltd. | Gpr40 receptor agonist, methods of preparing the same, and pharmaceutical compositions containing the same as an active ingredient |
| JP2018184454A (ja) * | 2012-11-09 | 2018-11-22 | エルジー・ケム・リミテッド | Gpr40受容体アゴニスト、この製造方法およびこれを活性成分として含有する医薬組成物 |
| WO2014133361A1 (ko) | 2013-02-28 | 2014-09-04 | 에스케이케미칼주식회사 | 삼환식 화합물 및 이의 용도 |
| US9643946B2 (en) | 2013-02-28 | 2017-05-09 | Sk Chemicals Co., Ltd. | Tricyclic compound and use thereof |
| JP2016513135A (ja) * | 2013-02-28 | 2016-05-12 | エスケー ケミカルズ カンパニー リミテッド | 三環式化合物およびその使用 |
| WO2014142363A1 (en) | 2013-03-14 | 2014-09-18 | Takeda Pharmaceutical Company Limited | Spiro azetidine isoxazole derivatives and their use as sstr5 antagonists |
| US10005720B2 (en) | 2013-04-05 | 2018-06-26 | North Carolina Central University | Compounds useful for the treatment of metabolic disorders and synthesis of the same |
| WO2015005489A1 (en) | 2013-07-09 | 2015-01-15 | Takeda Pharmaceutical Company Limited | Heterocyclic compound |
| WO2015020184A1 (ja) | 2013-08-09 | 2015-02-12 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2015052910A1 (en) | 2013-10-07 | 2015-04-16 | Takeda Pharmaceutical Company Limited | Antagonists of somatostatin receptor subtype 5 (sstr5) |
| WO2015062486A1 (en) * | 2013-10-31 | 2015-05-07 | Sunshine Lake Pharma Co., Ltd. | Biphenyl compounds and uses thereof |
| WO2015122188A1 (en) | 2014-02-13 | 2015-08-20 | Takeda Pharmaceutical Company Limited | Heterocyclic compound |
| WO2015122187A1 (en) | 2014-02-13 | 2015-08-20 | Takeda Pharmaceutical Company Limited | Fused heterocyclic compound |
| US11583528B2 (en) | 2016-02-17 | 2023-02-21 | Children's Medical Center Corporation | FFA1 (GPR40) as a therapeutic target for neural angiogenesis diseases or disorders |
| WO2018182051A1 (en) | 2017-03-30 | 2018-10-04 | Takeda Pharmaceutical Company Limited | Ip6k inhibitors |
| WO2018181864A1 (en) | 2017-03-31 | 2018-10-04 | Takeda Pharmaceutical Company Limited | Gip receptor activating peptide |
| WO2018181847A1 (ja) | 2017-03-31 | 2018-10-04 | 武田薬品工業株式会社 | 芳香環化合物 |
| WO2018182050A1 (en) | 2017-03-31 | 2018-10-04 | Takeda Pharmaceutical Company Limited | Substituted cyclyl-acetic acid derivatives for the treatment of metabolic disorders |
| WO2019134984A1 (en) | 2018-01-08 | 2019-07-11 | Celon Pharma S.A. | 3-phenyl-4-hexynoic acid derivatives as gpr40 agonists |
| PL424227A1 (pl) * | 2018-01-08 | 2019-07-15 | Celon Pharma Spółka Akcyjna | Pochodne kwasu 3-fenylo-4-heksynowego jako agoniści GPR40 |
| US11535660B1 (en) | 2018-03-23 | 2022-12-27 | Cannot Therapeutics, Inc. | Modulators of G-protein coupled receptors |
| WO2020045326A1 (ja) | 2018-08-27 | 2020-03-05 | 株式会社スコヒアファーマ | 安息香酸エステル化合物 |
| WO2020067575A1 (en) | 2018-09-24 | 2020-04-02 | Takeda Pharmaceutical Company Limited | Gip receptor agonist peptide compounds and uses thereof |
| WO2020067557A2 (en) | 2018-09-24 | 2020-04-02 | Takeda Pharmaceutical Company Limited | Gip receptor agonist peptide compounds and uses thereof |
| WO2020166662A1 (ja) | 2019-02-13 | 2020-08-20 | 株式会社ジェクスヴァル | 骨疾患の予防または治療用の医薬組成物 |
| WO2021193983A2 (en) | 2020-03-25 | 2021-09-30 | Takeda Pharmaceutical Company Limited | Qw dosing of gip receptor agonist peptide compounds and uses thereof |
| WO2021193984A2 (en) | 2020-03-25 | 2021-09-30 | Takeda Pharmaceutical Company Limited | Qd dosing of gip receptor agonist peptide compounds and uses thereof |
| WO2022034664A1 (ja) | 2020-08-12 | 2022-02-17 | 株式会社ジェクスヴァル | 骨疾患の予防または治療用の医薬組成物 |
| WO2022053013A1 (zh) | 2020-09-10 | 2022-03-17 | 上海爱博医药科技有限公司 | 苯并含氧杂环类化合物及其医药应用 |
| WO2022241287A2 (en) | 2021-05-13 | 2022-11-17 | Carmot Therapeutics, Inc. | Modulators of g-protein coupled receptors |
| WO2023169456A1 (en) | 2022-03-09 | 2023-09-14 | Gasherbrum Bio , Inc. | Heterocyclic glp-1 agonists |
| WO2023179542A1 (en) | 2022-03-21 | 2023-09-28 | Gasherbrum Bio , Inc. | 5,8-dihydro-1,7-naphthyridine derivatives as glp-1 agonists for the treatment of diabetes |
| WO2023198140A1 (en) | 2022-04-14 | 2023-10-19 | Gasherbrum Bio, Inc. | Heterocyclic glp-1 agonists |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1630152A1 (en) | 2006-03-01 |
| US7820837B2 (en) | 2010-10-26 |
| CA2527691C (en) | 2013-01-22 |
| US20060258722A1 (en) | 2006-11-16 |
| EP1630152A4 (en) | 2009-09-23 |
| CA2527691A1 (en) | 2004-12-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2004106276A1 (ja) | 縮合環化合物 | |
| JP4855777B2 (ja) | フェニルプロパン酸誘導体 | |
| JP4875978B2 (ja) | アミノフェニルプロパン酸誘導体 | |
| JP4859665B2 (ja) | アルコキシフェニルプロパン酸誘導体 | |
| JP4917634B2 (ja) | 縮合環化合物 | |
| US9382188B2 (en) | Aromatic ring compound | |
| JP5657578B2 (ja) | 新規な縮合環化合物およびその用途 | |
| JP4805552B2 (ja) | 縮合環化合物 | |
| JPWO2008136428A1 (ja) | 含窒素5員複素環化合物 | |
| WO2013122029A1 (ja) | 芳香環化合物 | |
| EP2266983B1 (en) | Nitrogenated 5-membered heterocyclic compound | |
| JP4074616B2 (ja) | 3−(4−ベンジルオキシフェニル)プロパン酸誘導体 | |
| TWI475018B (zh) | 雜環化合物 | |
| KR20070018827A (ko) | 3-(4-벤질옥시페닐)프로판산 유도체 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| REEP | Request for entry into the european phase |
Ref document number: 2004745580 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004745580 Country of ref document: EP Ref document number: 2527691 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006258722 Country of ref document: US Ref document number: 10558846 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004745580 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 10558846 Country of ref document: US |