JP7026876B2 - 一部がコーティングされた放出制御固形製剤 - Google Patents
一部がコーティングされた放出制御固形製剤 Download PDFInfo
- Publication number
- JP7026876B2 JP7026876B2 JP2017504225A JP2017504225A JP7026876B2 JP 7026876 B2 JP7026876 B2 JP 7026876B2 JP 2017504225 A JP2017504225 A JP 2017504225A JP 2017504225 A JP2017504225 A JP 2017504225A JP 7026876 B2 JP7026876 B2 JP 7026876B2
- Authority
- JP
- Japan
- Prior art keywords
- release
- core
- layer
- coating layer
- coating
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000203 mixture Substances 0.000 title claims description 55
- 239000007787 solid Substances 0.000 title claims description 37
- 238000009472 formulation Methods 0.000 title claims description 36
- 239000010410 layer Substances 0.000 claims description 106
- 239000003814 drug Substances 0.000 claims description 63
- 229940079593 drug Drugs 0.000 claims description 61
- 238000000576 coating method Methods 0.000 claims description 60
- 239000011248 coating agent Substances 0.000 claims description 57
- 238000002360 preparation method Methods 0.000 claims description 40
- 238000013268 sustained release Methods 0.000 claims description 38
- 239000012730 sustained-release form Substances 0.000 claims description 38
- 239000000463 material Substances 0.000 claims description 33
- 239000011247 coating layer Substances 0.000 claims description 31
- 238000000034 method Methods 0.000 claims description 23
- 239000004480 active ingredient Substances 0.000 claims description 18
- 239000000654 additive Substances 0.000 claims description 14
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 6
- 239000008101 lactose Substances 0.000 claims description 6
- 238000002156 mixing Methods 0.000 claims description 6
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims description 5
- 239000001267 polyvinylpyrrolidone Substances 0.000 claims description 5
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims description 5
- 229920000178 Acrylic resin Polymers 0.000 claims description 3
- 239000004925 Acrylic resin Substances 0.000 claims description 3
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 claims description 3
- 239000001856 Ethyl cellulose Substances 0.000 claims description 3
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 claims description 3
- 230000000996 additive effect Effects 0.000 claims description 3
- 229920002301 cellulose acetate Polymers 0.000 claims description 3
- 235000019325 ethyl cellulose Nutrition 0.000 claims description 3
- 229920001249 ethyl cellulose Polymers 0.000 claims description 3
- 239000003361 porogen Substances 0.000 claims description 3
- 239000011780 sodium chloride Substances 0.000 claims description 3
- 239000002198 insoluble material Substances 0.000 claims 2
- 101150100677 polo gene Proteins 0.000 claims 1
- 239000002952 polymeric resin Substances 0.000 claims 1
- 238000003825 pressing Methods 0.000 claims 1
- 229920003002 synthetic resin Polymers 0.000 claims 1
- 238000010828 elution Methods 0.000 description 31
- 230000000694 effects Effects 0.000 description 9
- 239000000825 pharmaceutical preparation Substances 0.000 description 8
- 229940127557 pharmaceutical product Drugs 0.000 description 8
- 230000006399 behavior Effects 0.000 description 7
- 239000002356 single layer Substances 0.000 description 7
- 239000004615 ingredient Substances 0.000 description 6
- 238000004090 dissolution Methods 0.000 description 5
- 239000007888 film coating Substances 0.000 description 5
- 238000009501 film coating Methods 0.000 description 5
- 239000002994 raw material Substances 0.000 description 5
- 229940121657 clinical drug Drugs 0.000 description 4
- 238000007906 compression Methods 0.000 description 4
- 230000006835 compression Effects 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 4
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 4
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 4
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 4
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 3
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 239000008107 starch Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 230000001186 cumulative effect Effects 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000012265 solid product Substances 0.000 description 2
- 239000002195 soluble material Substances 0.000 description 2
- 210000002784 stomach Anatomy 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical group CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- 229920002785 Croscarmellose sodium Polymers 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 239000013583 drug formulation Substances 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 230000000541 pulsatile effect Effects 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
- A61K9/2086—Layered tablets, e.g. bilayer tablets; Tablets of the type inert core-active coat
- A61K9/209—Layered tablets, e.g. bilayer tablets; Tablets of the type inert core-active coat containing drug in at least two layers or in the core and in at least one outer layer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/2833—Organic macromolecular compounds
- A61K9/286—Polysaccharides, e.g. gums; Cyclodextrin
- A61K9/2866—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/192—Carboxylic acids, e.g. valproic acid having aromatic groups, e.g. sulindac, 2-aryl-propionic acids, ethacrynic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/38—Heterocyclic compounds having sulfur as a ring hetero atom
- A61K31/381—Heterocyclic compounds having sulfur as a ring hetero atom having five-membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
- A61K31/4045—Indole-alkylamines; Amides thereof, e.g. serotonin, melatonin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/53—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with three nitrogens as the only ring hetero atoms, e.g. chlorazanil, melamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/57—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone
- A61K31/573—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone substituted in position 21, e.g. cortisone, dexamethasone, prednisone or aldosterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2009—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
- A61K9/2018—Sugars, or sugar alcohols, e.g. lactose, mannitol; Derivatives thereof, e.g. polysorbates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
- A61K9/2086—Layered tablets, e.g. bilayer tablets; Tablets of the type inert core-active coat
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2095—Tabletting processes; Dosage units made by direct compression of powders or specially processed granules, by eliminating solvents, by melt-extrusion, by injection molding, by 3D printing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/2813—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/282—Organic compounds, e.g. fats
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/282—Organic compounds, e.g. fats
- A61K9/2826—Sugars or sugar alcohols, e.g. sucrose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2893—Tablet coating processes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
Description
出願番号:201410139563.9
出願日:2014-04-08
発明の名称:新型放出制御錠
1)錠芯を調製するための各組成成分を混合し、I番パンチで一回目のプレスを行って、錠芯を得る工程と、
2)II番パンチに交換し、コーティング層を調製するための各組成成分を混合した後、中金型に充填する工程と、
3)工程1)でプレスした錠芯を中金型の中央に充填し、二回目のプレスを行って、固形製剤を得る工程と
を含み、
前記I番パンチの直径がII番パンチの直径より小さい。
製剤は、1層の徐放層のみを有する単層錠である錠芯と、錠芯外に被覆されたコーティングとを含む。図1に示すように、コアの頂面の一部が、コーティングで被覆されずに固形製剤の表面に露出している。
1、錠芯の原料と添加剤を処方に準じて秤量して、十分に混合した後使用に備え、そして、I番パンチの直径がII番より小さい二種類の規格のパンチを選択する;
製剤は、2層の錠である錠芯と、錠芯外に被覆されたコーティングを含む。図2に示すように、コアの頂面の一部がコーティングで被覆されずに固形製剤の表面に露出している。
1、錠芯の徐放層、錠芯の速放層の原料と添加剤を処方に準じて秤量し、それぞれ十分に混合した後使用に備え、そして、I番パンチの直径がII番より小さい二種類の規格のパンチを選択する;
製剤は、1層の徐放層のみを有する単層錠である錠芯と、錠芯外に被覆されたコーティングとを含む。図1に示すように、コアの頂面の一部がコーティングで被覆されずに固形製剤の表面に露出している。
1、錠芯の原料と添加剤を処方に準じて秤量し、十分に混合した後使用に備え、そして、I番パンチの直径がII番より小さい二種類の規格のパンチを選択する;
製剤は、第1徐放層と第2徐放層からなる2層の錠である錠芯と、錠芯外に被覆されたコーティングとを含む。図3に示すように、コアの頂面の一部がコーティングで被覆されずに固形製剤の表面に露出している。
1、錠芯の第1徐放層、錠芯の第2徐放層の原料と添加剤を処方に準じて秤量して、それぞれ十分に混合した後使用に備え、そして、I番パンチの直径がII番より小さい二種類の規格のパンチを選択する;
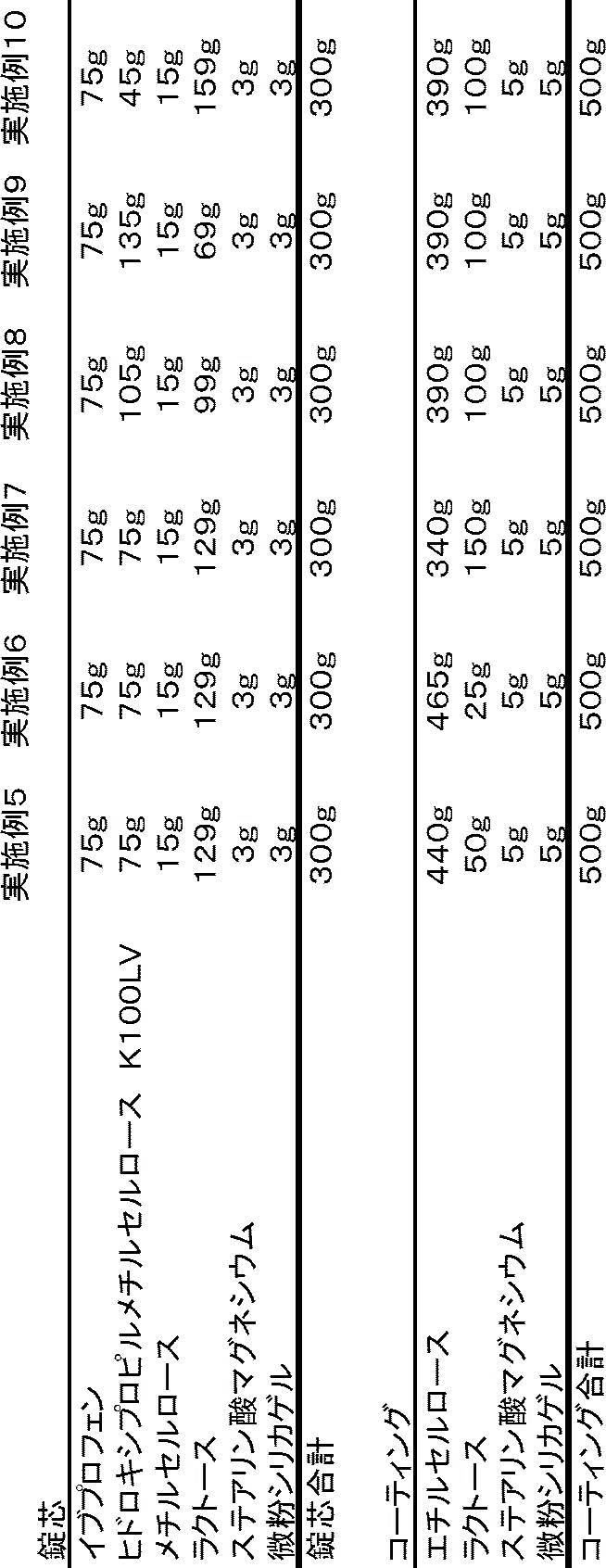
実施例5~10の製剤は、実施例1と同様に、コアが単層である(1層の徐放層のみを有する)放出制御製剤である。
本実施例における放出制御製剤のコアが3層(いずれの層も活性成分を含む)であり、不完全なコーティング層にも活性物質が含まれ、最外層のフィルムコーティングにも活性物質が含まれる。図10に示すように、コアの上層薬物含有層の一部がコーティングで被覆されずに固形製剤の表面に露出している。
1、錠芯の上層、中間層、下層の原料と添加剤を処方に準じて秤量し、それぞれ十分に混合した後使用に備え、そして、I番パンチの直径がII番より小さい二種類の規格のパンチを選択する;
<付記>
<項1>
活性薬物と添加剤を含むコアと、徐放・放出制御素材を含み、活性成分を含むか、または含まないコーティング層とからなる放出制御固形製剤において、前記コアの表面がコーティング層で完全に被覆されず、一部が固形製剤の表面に露出していることを特徴とする、放出制御固形製剤。
<項2>
前記コアが1層または複数層であり、前記層が徐放層であり、あるいは速放層と徐放層を共に含み、コアが2層以上である場合、最外層の成分が最初に放出され、第二外層の成分が次に放出され、コアの最内層の成分が最後に放出されるようにそれぞれの層の成分が順次放出される、<項1>に記載の固形製剤。
<項3>
前記コーティング層が圧縮コーティング法で調製されてなる、<項1>に記載の固形製剤。
<項4>
前記コーティング層の素材が水可溶性または水不溶性であり、前記水可溶性のコーティング素材が、ヒドロキシプロピルメチルセルロース、ヒドロキシプロピルセルロースから選ばれる1種または2種であり、前記水不溶性のコーティング素材が、エチルセルロース、セルロースアセテートポリマー、アクリル樹脂から選ばれる1種または複数種であることを特徴とする、<項1>に記載の固形製剤。
<項5>
前記コーティング層の素材が、ポリビニルピロリドン、ラクトース、塩化ナトリウムから選ばれる1種または複数種である水溶性ポロゲンをさらに含むことを特徴とする、<項1>に記載の固形製剤。
<項6>
前記コーティング層の外面に、活性成分を含むか、または含まない、もう1層のコーティングを有する、<項1>~<項5>のいずれか一項に記載の固形製剤。
<項7>
錠剤である<項1>に記載の固形製剤。
<項8>
コアは、1層以上の錠状物からなる錠芯である、<項7>に記載の固形製剤。
<項9>
<項7>または<項8>に記載の固形製剤の調製方法であって、
打錠機でタブレットのプレスを行い、
1)錠芯を調製するための各組成成分を混合し、I番パンチで一回目のプレスを行って、錠芯を得る工程と、
2)II番パンチに交換し、コーティング層を調製するための各組成成分を混合した後、中金型に充填する工程と、
3)工程1)でプレスした錠芯を中金型の中央に充填し、二回目のプレスを行って、固形製剤を得る工程と
を含み、
前記I番パンチの直径がII番パンチの直径より小さい、固形製剤の調製方法。
<項10>
前記工程2)と3)を、
II番パンチに交換した後、工程1)でプレスした錠芯を中金型の中央に充填し、その後、中金型の中で錠芯の周囲に、均一に混合されたコーティング層の組成成分を充填して、二回目のプレスを行うことへ変更する、<項9>に記載の調製方法。
Claims (7)
- コアと、コーティング層とからなる放出制御固形製剤であって、
前記コアが、2層以上であり、それぞれの層が活性薬物と添加剤とを含み、
前記コーティング層が、水溶性ポロゲンと、徐放・放出制御素材とを含み、
前記コーティング層が、活性成分を含むか、または含まず、
前記コアの表面が、前記コーティング層で完全に被覆されず、前記コアの表面の上層の一部が固形製剤の表面に露出しており、
前記コーティング層の前記徐放・放出制御素材が、水不溶性素材であり、前記コーティング層の総量に対して68~78重量%で含まれ、
前記水不溶性素材が、エチルセルロース、セルロースアセテートポリマー、アクリル樹脂からなる群から選ばれる1種または複数種であり、
前記水溶性ポロゲンが、ポリビニルピロリドン、ラクトース、塩化ナトリウムからなる群から選ばれる1種または複数種であり、前記コーティング層の総量に対して20重量%、又は20~30重量%で含まれ、
前記コアと前記コーティング層の重量比が3:5である、ことを特徴とする、放出制御固形製剤。 - 前記コアが3層であり、前記3層中の成分が、上層の成分が最初に放出され、中間層の成分が次に放出され、下層の成分が最後に放出されるようにそれぞれの層の成分が順次放出される、請求項1に記載の固形製剤。
- 前記コーティング層の外面に、活性成分を含むか、または含まない、もう1層のコーティングを有する、請求項1に記載の固形製剤。
- 錠剤である請求項1に記載の固形製剤。
- 前記コーティング層が1種類の活性薬物を含み、前記コアが別の種類の活性薬物を含む、請求項1に記載の固形製剤。
- 請求項4に記載の固形製剤を、打錠機でプレスすることにより調製する方法であって、
1)錠芯を調製するための各組成成分を混合し、I番パンチで一回目のプレスを行って、錠芯を得る工程と、
2)II番パンチに交換し、コーティング層を調製するための各組成成分を混合した後、中金型に充填する工程と、
3)工程1)でプレスした錠芯を中金型の中央に充填し、二回目のプレスを行って、固形製剤を得る工程と
を含み、
前記I番パンチの直径がII番パンチの直径より小さい、固形製剤の調製方法。 - 前記工程2)と3)を、
II番パンチに交換した後、工程1)でプレスした錠芯を中金型の中央に充填し、その後、中金型の中で錠芯の周囲に、均一に混合されたコーティング層の組成成分を充填して、二回目のプレスを行うことへ変更する、請求項6に記載の調製方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201410139563.9 | 2014-04-08 | ||
| CN201410139563.9A CN103948557A (zh) | 2014-04-08 | 2014-04-08 | 一种新型控释片 |
| PCT/CN2015/076028 WO2015154656A1 (zh) | 2014-04-08 | 2015-04-08 | 部分包衣的控释固体制剂 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2017510654A JP2017510654A (ja) | 2017-04-13 |
| JP7026876B2 true JP7026876B2 (ja) | 2022-03-01 |
Family
ID=51326007
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017504225A Active JP7026876B2 (ja) | 2014-04-08 | 2015-04-08 | 一部がコーティングされた放出制御固形製剤 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US10413514B2 (ja) |
| EP (1) | EP3130334B8 (ja) |
| JP (1) | JP7026876B2 (ja) |
| CN (1) | CN103948557A (ja) |
| DK (1) | DK3130334T3 (ja) |
| ES (1) | ES2939890T3 (ja) |
| WO (1) | WO2015154656A1 (ja) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104490838B (zh) * | 2014-11-20 | 2017-03-01 | 河北医科大学 | 一种骨架型缓控释片剂及其制备方法和应用 |
| CN105078918B (zh) * | 2015-01-09 | 2018-04-17 | 田曼霖 | 一种醋酸地塞米松缓释制剂及其制备方法 |
| CA2987114A1 (en) | 2015-06-03 | 2016-12-08 | Triastek, Inc. | Method of 3d printing oral dosage forms having a drug in a compartment sealed by an erodibleplug |
| US11571391B2 (en) | 2018-01-09 | 2023-02-07 | Triastek, Inc. | Oral drug dosage forms compromising a fixed-dose of an ADHD non-stimulant and an ADHD stimulant |
| CN117427043A (zh) * | 2022-07-21 | 2024-01-23 | 越洋医药开发(广州)有限公司 | 一种双相控释制剂及其制备方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007501773A (ja) | 2003-08-07 | 2007-02-01 | エスビー・ファルムコ・プエルト・リコ・インコーポレイテッド | 長時間にわたって弱塩基を放出するための組成物 |
| WO2008006216A1 (en) | 2006-07-14 | 2008-01-17 | Mistral Pharma, Inc. | Anti-inflammatory and cytoprotectant chronotherapy |
| JP2009543875A (ja) | 2006-07-19 | 2009-12-10 | ワトソン ラボラトリーズ、インコーポレイテッド | 制御性放出製剤および関連する方法 |
| CN102614141A (zh) | 2011-12-02 | 2012-08-01 | 内蒙古元和药业股份有限公司 | 一种盐酸维拉帕米迟释片剂及其制备方法 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5922342A (en) * | 1990-10-01 | 1999-07-13 | Pharmacia & Upjohn Company | Lateral edge coated controlled release pharmaceutical compositions |
| DE69229881T2 (de) * | 1991-10-04 | 1999-12-09 | Yoshitomi Pharmaceutical | Tablette mit verzögerter freisetzung |
| DE4431653C2 (de) * | 1994-09-06 | 2000-01-20 | Lohmann Therapie Syst Lts | Manteltablette zur kontrollierten Freisetzung von Wirkstoffen, ein Verfahren zu ihrer Herstellung und ihre Verwendung |
| US5582838A (en) * | 1994-12-22 | 1996-12-10 | Merck & Co., Inc. | Controlled release drug suspension delivery device |
| IT1282576B1 (it) * | 1996-02-06 | 1998-03-31 | Jagotec Ag | Compressa farmaceutica atta a cedere la sostanza attiva in tempi successivi e predeterminabili |
| US20050013863A1 (en) * | 2003-07-18 | 2005-01-20 | Depomed, Inc., A Corporation Of The State Of California | Dual drug dosage forms with improved separation of drugs |
| DE60307819T2 (de) * | 2003-07-30 | 2007-10-04 | Pharmathen S.A., Pallini | Venlafaxine Hydrochloridformulierungen mit verzögerter Freisetzung |
| CN101132777A (zh) * | 2004-12-20 | 2008-02-27 | 科利吉姆制药公司 | 用于睡眠障碍的药物组合物 |
| US20080317677A1 (en) * | 2007-06-22 | 2008-12-25 | Szymczak Christopher E | Laser Marked Dosage Forms |
| CN101843598B (zh) * | 2010-06-07 | 2015-10-07 | 常州制药厂有限公司 | 非洛地平缓释片的制备方法 |
| AU2015274456B2 (en) | 2014-06-11 | 2020-03-26 | Massachusetts Institute Of Technology | Self-assembled residence devices and related methods |
-
2014
- 2014-04-08 CN CN201410139563.9A patent/CN103948557A/zh active Pending
-
2015
- 2015-04-08 EP EP15776295.6A patent/EP3130334B8/en active Active
- 2015-04-08 DK DK15776295.6T patent/DK3130334T3/da active
- 2015-04-08 JP JP2017504225A patent/JP7026876B2/ja active Active
- 2015-04-08 WO PCT/CN2015/076028 patent/WO2015154656A1/zh active Application Filing
- 2015-04-08 US US15/302,977 patent/US10413514B2/en active Active
- 2015-04-08 ES ES15776295T patent/ES2939890T3/es active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007501773A (ja) | 2003-08-07 | 2007-02-01 | エスビー・ファルムコ・プエルト・リコ・インコーポレイテッド | 長時間にわたって弱塩基を放出するための組成物 |
| WO2008006216A1 (en) | 2006-07-14 | 2008-01-17 | Mistral Pharma, Inc. | Anti-inflammatory and cytoprotectant chronotherapy |
| JP2009543875A (ja) | 2006-07-19 | 2009-12-10 | ワトソン ラボラトリーズ、インコーポレイテッド | 制御性放出製剤および関連する方法 |
| CN102614141A (zh) | 2011-12-02 | 2012-08-01 | 内蒙古元和药业股份有限公司 | 一种盐酸维拉帕米迟释片剂及其制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3130334A4 (en) | 2017-12-06 |
| JP2017510654A (ja) | 2017-04-13 |
| EP3130334A1 (en) | 2017-02-15 |
| WO2015154656A1 (zh) | 2015-10-15 |
| EP3130334B8 (en) | 2023-07-19 |
| US10413514B2 (en) | 2019-09-17 |
| DK3130334T3 (da) | 2023-03-13 |
| US20170027872A1 (en) | 2017-02-02 |
| EP3130334B1 (en) | 2023-02-15 |
| ES2939890T3 (es) | 2023-04-27 |
| CN103948557A (zh) | 2014-07-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7026876B2 (ja) | 一部がコーティングされた放出制御固形製剤 | |
| KR101378973B1 (ko) | 구형에 가까운 형태의 다중 투여 단위 정제를 포함하는 경질 캡슐 복합 제형 및 이의 제조방법 | |
| US20080286344A1 (en) | Solid form | |
| US20230011269A1 (en) | Pharmaceutical composition comprising a potent inhibitor of urat1 | |
| EP2255797A2 (en) | Composite preparation | |
| KR20090091084A (ko) | 방출성이 제어된 약제학적 제제 | |
| HRP20040996A2 (en) | High drug load tablet | |
| US20100092554A1 (en) | Combination with an extended release tablet formulation containing pramipexole or a pharmaceutically acceptable salt thereof | |
| KR20090015890A (ko) | 저 홍조 니아신 제형 | |
| KR20090114332A (ko) | 약제학적 제제 | |
| WO2008140461A1 (en) | Solid form | |
| EA026335B1 (ru) | Снижение флуктуаций опиоидов в крови | |
| HRP20240045T1 (hr) | Tablete s trenutnim oslobađanjem koje sadrže lijek i postupci za proizvodnju tableta | |
| AU2020336093A1 (en) | System and method of multi-drug delivery | |
| CN104755074A (zh) | 美金刚的药物组合物 | |
| CN103655505B (zh) | 一种止痛类双层控释片及其制备方法 | |
| KR101164300B1 (ko) | 약제학적 제제 | |
| CN103285017A (zh) | 复方单硝酸异山梨酯阿司匹林缓释胶囊制剂及制备方法 | |
| WO2018211336A2 (en) | Solid dosage form containing sorafenib tosylate | |
| WO2010075275A1 (en) | Selective release of non-racemic mixtures of two enantiomers from tablets and capsules | |
| JP2018518521A (ja) | モサプリドとラベプラゾールの複合製剤 | |
| WO2015011161A1 (en) | Anti-tuberculosis stable pharmaceutical composition in a form of a coated tablet comprising granules of isoniazid and granules of rifapentine and its process of preparation | |
| KR20110117758A (ko) | 베타 아드레날린 차단제 및 HMG-CoA 환원 효소 억제제를 함유하는 약제학적 제제 | |
| KR20090107960A (ko) | 심혈관계 질환 치료용 약제학적 제제 | |
| WO2015189807A1 (en) | Bi-layer tablet formulations of cyclophosphamide and capecitabine and highly fractionated metronomic administration thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A711 Effective date: 20170307 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20170307 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20180406 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20181227 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20190108 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20190402 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190708 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20190827 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20191227 |
|
| C60 | Trial request (containing other claim documents, opposition documents) |
Free format text: JAPANESE INTERMEDIATE CODE: C60 Effective date: 20191227 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20200114 |
|
| C21 | Notice of transfer of a case for reconsideration by examiners before appeal proceedings |
Free format text: JAPANESE INTERMEDIATE CODE: C21 Effective date: 20200121 |
|
| A912 | Re-examination (zenchi) completed and case transferred to appeal board |
Free format text: JAPANESE INTERMEDIATE CODE: A912 Effective date: 20200214 |
|
| C211 | Notice of termination of reconsideration by examiners before appeal proceedings |
Free format text: JAPANESE INTERMEDIATE CODE: C211 Effective date: 20200218 |
|
| C22 | Notice of designation (change) of administrative judge |
Free format text: JAPANESE INTERMEDIATE CODE: C22 Effective date: 20200721 |
|
| C13 | Notice of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: C13 Effective date: 20201222 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20210319 Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20210317 |
|
| C272 | Notice of ex officio correction |
Free format text: JAPANESE INTERMEDIATE CODE: C272 Effective date: 20210323 |
|
| C22 | Notice of designation (change) of administrative judge |
Free format text: JAPANESE INTERMEDIATE CODE: C22 Effective date: 20210413 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20210521 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210621 |
|
| C13 | Notice of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: C13 Effective date: 20210810 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20211101 |
|
| C23 | Notice of termination of proceedings |
Free format text: JAPANESE INTERMEDIATE CODE: C23 Effective date: 20211130 |
|
| C03 | Trial/appeal decision taken |
Free format text: JAPANESE INTERMEDIATE CODE: C03 Effective date: 20220111 |
|
| C30A | Notification sent |
Free format text: JAPANESE INTERMEDIATE CODE: C3012 Effective date: 20220111 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20220126 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7026876 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313113 |
|
| R360 | Written notification for declining of transfer of rights |
Free format text: JAPANESE INTERMEDIATE CODE: R360 |
|
| R360 | Written notification for declining of transfer of rights |
Free format text: JAPANESE INTERMEDIATE CODE: R360 |
|
| R371 | Transfer withdrawn |
Free format text: JAPANESE INTERMEDIATE CODE: R371 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313113 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |