JP2013529196A - キナーゼlrrk2の阻害剤としてのピラゾロピリジン - Google Patents
キナーゼlrrk2の阻害剤としてのピラゾロピリジン Download PDFInfo
- Publication number
- JP2013529196A JP2013529196A JP2013509631A JP2013509631A JP2013529196A JP 2013529196 A JP2013529196 A JP 2013529196A JP 2013509631 A JP2013509631 A JP 2013509631A JP 2013509631 A JP2013509631 A JP 2013509631A JP 2013529196 A JP2013529196 A JP 2013529196A
- Authority
- JP
- Japan
- Prior art keywords
- alkyl
- heterocycloalkyl
- aryl
- heteroaryl
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 108010020246 Leucine-Rich Repeat Serine-Threonine Protein Kinase-2 Proteins 0.000 title claims description 65
- 102000009784 Leucine-Rich Repeat Serine-Threonine Protein Kinase-2 Human genes 0.000 title claims description 5
- 239000003112 inhibitor Substances 0.000 title description 7
- 150000005229 pyrazolopyridines Chemical class 0.000 title description 3
- 150000001875 compounds Chemical class 0.000 claims abstract description 216
- 125000003118 aryl group Chemical group 0.000 claims abstract description 105
- 125000001072 heteroaryl group Chemical group 0.000 claims abstract description 103
- 125000001424 substituent group Chemical group 0.000 claims abstract description 95
- 238000000034 method Methods 0.000 claims abstract description 73
- 125000006272 (C3-C7) cycloalkyl group Chemical group 0.000 claims abstract description 63
- -1 substituted Chemical class 0.000 claims abstract description 63
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 40
- 150000002367 halogens Chemical class 0.000 claims abstract description 40
- 125000000592 heterocycloalkyl group Chemical group 0.000 claims abstract description 29
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 23
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 21
- 150000003839 salts Chemical class 0.000 claims abstract description 21
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 16
- 239000001257 hydrogen Substances 0.000 claims abstract description 16
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 14
- 150000002148 esters Chemical class 0.000 claims abstract description 13
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims abstract description 10
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 9
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims abstract description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims abstract description 3
- 150000002431 hydrogen Chemical class 0.000 claims abstract 6
- 108091000080 Phosphotransferase Proteins 0.000 claims description 44
- 102000020233 phosphotransferase Human genes 0.000 claims description 44
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 35
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 30
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 26
- 239000003814 drug Substances 0.000 claims description 25
- 238000003556 assay Methods 0.000 claims description 23
- 239000000460 chlorine Substances 0.000 claims description 23
- 229920006395 saturated elastomer Polymers 0.000 claims description 23
- 208000035475 disorder Diseases 0.000 claims description 21
- 238000002360 preparation method Methods 0.000 claims description 19
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 17
- 230000000694 effects Effects 0.000 claims description 17
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 15
- 229910052760 oxygen Inorganic materials 0.000 claims description 15
- 239000001301 oxygen Substances 0.000 claims description 15
- 125000004076 pyridyl group Chemical group 0.000 claims description 15
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 13
- 229910052717 sulfur Inorganic materials 0.000 claims description 13
- 239000011593 sulfur Substances 0.000 claims description 13
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 12
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 11
- 125000002541 furyl group Chemical group 0.000 claims description 10
- 125000003386 piperidinyl group Chemical group 0.000 claims description 10
- 239000007822 coupling agent Substances 0.000 claims description 9
- 208000015122 neurodegenerative disease Diseases 0.000 claims description 9
- 206010028980 Neoplasm Diseases 0.000 claims description 8
- 239000000654 additive Substances 0.000 claims description 8
- 201000011510 cancer Diseases 0.000 claims description 8
- IKDUDTNKRLTJSI-UHFFFAOYSA-N hydrazine hydrate Chemical compound O.NN IKDUDTNKRLTJSI-UHFFFAOYSA-N 0.000 claims description 8
- 230000002265 prevention Effects 0.000 claims description 8
- 241000124008 Mammalia Species 0.000 claims description 7
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 7
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 7
- 239000003937 drug carrier Substances 0.000 claims description 7
- 230000002401 inhibitory effect Effects 0.000 claims description 7
- 125000002757 morpholinyl group Chemical group 0.000 claims description 7
- 229910052801 chlorine Inorganic materials 0.000 claims description 6
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 6
- 229910052731 fluorine Inorganic materials 0.000 claims description 6
- 238000004519 manufacturing process Methods 0.000 claims description 6
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 6
- 125000004432 carbon atom Chemical group C* 0.000 claims description 5
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 5
- 230000004770 neurodegeneration Effects 0.000 claims description 5
- 125000001715 oxadiazolyl group Chemical group 0.000 claims description 5
- 125000001412 tetrahydropyranyl group Chemical group 0.000 claims description 5
- 230000002159 abnormal effect Effects 0.000 claims description 4
- 150000001412 amines Chemical class 0.000 claims description 4
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 claims description 4
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 4
- 125000005842 heteroatom Chemical group 0.000 claims description 4
- 230000005764 inhibitory process Effects 0.000 claims description 4
- 230000000996 additive effect Effects 0.000 claims description 3
- 229910052794 bromium Inorganic materials 0.000 claims description 3
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 claims description 3
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 3
- 239000007800 oxidant agent Substances 0.000 claims description 3
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 claims description 3
- 229940124597 therapeutic agent Drugs 0.000 claims description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 2
- 108010021119 Trichosanthin Proteins 0.000 claims description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 2
- 230000007170 pathology Effects 0.000 claims description 2
- 125000003107 substituted aryl group Chemical group 0.000 claims description 2
- 230000001225 therapeutic effect Effects 0.000 abstract description 11
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 abstract 1
- XTFIVUDBNACUBN-UHFFFAOYSA-N 1,3,5-trinitro-1,3,5-triazinane Chemical compound [O-][N+](=O)N1CN([N+]([O-])=O)CN([N+]([O-])=O)C1 XTFIVUDBNACUBN-UHFFFAOYSA-N 0.000 abstract 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 112
- 239000000203 mixture Substances 0.000 description 96
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 60
- 102100032693 Leucine-rich repeat serine/threonine-protein kinase 2 Human genes 0.000 description 58
- 239000000243 solution Substances 0.000 description 57
- 239000007787 solid Substances 0.000 description 54
- 239000000543 intermediate Substances 0.000 description 53
- 239000011541 reaction mixture Substances 0.000 description 48
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 47
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 46
- 238000006243 chemical reaction Methods 0.000 description 39
- 235000019439 ethyl acetate Nutrition 0.000 description 37
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 34
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 32
- 208000018737 Parkinson disease Diseases 0.000 description 31
- 238000009472 formulation Methods 0.000 description 31
- 238000003818 flash chromatography Methods 0.000 description 30
- 238000005481 NMR spectroscopy Methods 0.000 description 29
- 102000001253 Protein Kinase Human genes 0.000 description 29
- 108060006633 protein kinase Proteins 0.000 description 29
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 26
- 239000012267 brine Substances 0.000 description 26
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 26
- 235000002639 sodium chloride Nutrition 0.000 description 25
- 239000000741 silica gel Substances 0.000 description 23
- 229910002027 silica gel Inorganic materials 0.000 description 23
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 22
- 239000012074 organic phase Substances 0.000 description 21
- 210000004027 cell Anatomy 0.000 description 20
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 18
- 238000005160 1H NMR spectroscopy Methods 0.000 description 17
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 16
- 239000003446 ligand Substances 0.000 description 16
- 239000007788 liquid Substances 0.000 description 16
- 230000035772 mutation Effects 0.000 description 16
- 239000011734 sodium Substances 0.000 description 16
- 239000000758 substrate Substances 0.000 description 16
- 239000000725 suspension Substances 0.000 description 16
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 15
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 14
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 14
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 14
- 239000012043 crude product Substances 0.000 description 14
- 201000010099 disease Diseases 0.000 description 14
- 239000003921 oil Substances 0.000 description 13
- 230000002829 reductive effect Effects 0.000 description 13
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 12
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 12
- 239000003795 chemical substances by application Substances 0.000 description 12
- 235000019198 oils Nutrition 0.000 description 12
- 239000003208 petroleum Substances 0.000 description 12
- 238000002953 preparative HPLC Methods 0.000 description 12
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 11
- 102000010111 Ezrin/radixin/moesin Human genes 0.000 description 11
- 108050001788 Ezrin/radixin/moesin Proteins 0.000 description 11
- 102100027869 Moesin Human genes 0.000 description 11
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical group [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 11
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 11
- 230000027455 binding Effects 0.000 description 11
- 108010071525 moesin Proteins 0.000 description 11
- 238000000746 purification Methods 0.000 description 11
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 10
- 239000002253 acid Substances 0.000 description 10
- 239000013543 active substance Substances 0.000 description 10
- 239000000706 filtrate Substances 0.000 description 10
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 10
- 108090000623 proteins and genes Proteins 0.000 description 10
- 239000003826 tablet Substances 0.000 description 10
- AQRLNPVMDITEJU-UHFFFAOYSA-N triethylsilane Chemical compound CC[SiH](CC)CC AQRLNPVMDITEJU-UHFFFAOYSA-N 0.000 description 10
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 9
- 239000000969 carrier Substances 0.000 description 9
- 229940079593 drug Drugs 0.000 description 9
- 239000000839 emulsion Substances 0.000 description 9
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 9
- 239000006174 pH buffer Substances 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 8
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 8
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 8
- 235000001014 amino acid Nutrition 0.000 description 8
- 150000001413 amino acids Chemical class 0.000 description 8
- 239000002775 capsule Substances 0.000 description 8
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 description 8
- DWOZNANUEDYIOF-UHFFFAOYSA-L 4-ditert-butylphosphanyl-n,n-dimethylaniline;dichloropalladium Chemical compound Cl[Pd]Cl.CN(C)C1=CC=C(P(C(C)(C)C)C(C)(C)C)C=C1.CN(C)C1=CC=C(P(C(C)(C)C)C(C)(C)C)C=C1 DWOZNANUEDYIOF-UHFFFAOYSA-L 0.000 description 7
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 7
- 238000012512 characterization method Methods 0.000 description 7
- 125000005843 halogen group Chemical group 0.000 description 7
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 7
- 239000000843 powder Substances 0.000 description 7
- 235000018102 proteins Nutrition 0.000 description 7
- 102000004169 proteins and genes Human genes 0.000 description 7
- 239000011780 sodium chloride Substances 0.000 description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 6
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 6
- 101100189356 Mus musculus Papolb gene Proteins 0.000 description 6
- LFTLOKWAGJYHHR-UHFFFAOYSA-N N-methylmorpholine N-oxide Chemical compound CN1(=O)CCOCC1 LFTLOKWAGJYHHR-UHFFFAOYSA-N 0.000 description 6
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 229940024606 amino acid Drugs 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- 238000011161 development Methods 0.000 description 6
- 230000018109 developmental process Effects 0.000 description 6
- 239000000284 extract Substances 0.000 description 6
- 239000004615 ingredient Substances 0.000 description 6
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 6
- 239000000314 lubricant Substances 0.000 description 6
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 6
- 230000004048 modification Effects 0.000 description 6
- 238000012986 modification Methods 0.000 description 6
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 6
- 239000003755 preservative agent Substances 0.000 description 6
- 238000004007 reversed phase HPLC Methods 0.000 description 6
- 239000002904 solvent Substances 0.000 description 6
- 238000003756 stirring Methods 0.000 description 6
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 6
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 5
- 108010010803 Gelatin Proteins 0.000 description 5
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 5
- 241000282412 Homo Species 0.000 description 5
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 5
- 239000011230 binding agent Substances 0.000 description 5
- 230000003197 catalytic effect Effects 0.000 description 5
- 239000002552 dosage form Substances 0.000 description 5
- 239000008273 gelatin Substances 0.000 description 5
- 229940014259 gelatin Drugs 0.000 description 5
- 229920000159 gelatin Polymers 0.000 description 5
- 235000019322 gelatine Nutrition 0.000 description 5
- 235000011852 gelatine desserts Nutrition 0.000 description 5
- 239000008187 granular material Substances 0.000 description 5
- 238000001727 in vivo Methods 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 238000000465 moulding Methods 0.000 description 5
- 239000012071 phase Substances 0.000 description 5
- 230000026731 phosphorylation Effects 0.000 description 5
- 238000006366 phosphorylation reaction Methods 0.000 description 5
- 108020001580 protein domains Proteins 0.000 description 5
- 230000002441 reversible effect Effects 0.000 description 5
- 229910000029 sodium carbonate Inorganic materials 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 5
- RLLPVAHGXHCWKJ-IEBWSBKVSA-N (3-phenoxyphenyl)methyl (1s,3s)-3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropane-1-carboxylate Chemical compound CC1(C)[C@H](C=C(Cl)Cl)[C@@H]1C(=O)OCC1=CC=CC(OC=2C=CC=CC=2)=C1 RLLPVAHGXHCWKJ-IEBWSBKVSA-N 0.000 description 4
- 125000004217 4-methoxybenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1OC([H])([H])[H])C([H])([H])* 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 4
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 4
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 4
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 4
- 102000004190 Enzymes Human genes 0.000 description 4
- 108090000790 Enzymes Proteins 0.000 description 4
- 241000272186 Falco columbarius Species 0.000 description 4
- 241001444201 Falco tinnunculus Species 0.000 description 4
- 102000013446 GTP Phosphohydrolases Human genes 0.000 description 4
- 108091006109 GTPases Proteins 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 4
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 4
- 240000007472 Leucaena leucocephala Species 0.000 description 4
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 4
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 4
- 208000027089 Parkinsonian disease Diseases 0.000 description 4
- 206010034010 Parkinsonism Diseases 0.000 description 4
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 4
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 4
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 4
- 229930006000 Sucrose Natural products 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 229910052786 argon Inorganic materials 0.000 description 4
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 4
- 238000004113 cell culture Methods 0.000 description 4
- 238000007906 compression Methods 0.000 description 4
- 230000006835 compression Effects 0.000 description 4
- 238000013270 controlled release Methods 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 229960001031 glucose Drugs 0.000 description 4
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 4
- 108020001756 ligand binding domains Proteins 0.000 description 4
- CCERQOYLJJULMD-UHFFFAOYSA-M magnesium;carbanide;chloride Chemical compound [CH3-].[Mg+2].[Cl-] CCERQOYLJJULMD-UHFFFAOYSA-M 0.000 description 4
- 239000002808 molecular sieve Substances 0.000 description 4
- 239000012299 nitrogen atmosphere Substances 0.000 description 4
- 230000008506 pathogenesis Effects 0.000 description 4
- 230000037361 pathway Effects 0.000 description 4
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 4
- 125000004193 piperazinyl group Chemical group 0.000 description 4
- 239000002244 precipitate Substances 0.000 description 4
- 239000000651 prodrug Substances 0.000 description 4
- 229940002612 prodrug Drugs 0.000 description 4
- 230000004853 protein function Effects 0.000 description 4
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 4
- JPJALAQPGMAKDF-UHFFFAOYSA-N selenium dioxide Chemical compound O=[Se]=O JPJALAQPGMAKDF-UHFFFAOYSA-N 0.000 description 4
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 4
- 239000005720 sucrose Substances 0.000 description 4
- 229960004793 sucrose Drugs 0.000 description 4
- 239000004094 surface-active agent Substances 0.000 description 4
- 238000004809 thin layer chromatography Methods 0.000 description 4
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 3
- GJXUNKKKYAYFSQ-UHFFFAOYSA-N 4-chloro-3-iodo-6-methyl-2h-pyrazolo[4,3-c]pyridine Chemical compound ClC1=NC(C)=CC2=C1C(I)=NN2 GJXUNKKKYAYFSQ-UHFFFAOYSA-N 0.000 description 3
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 3
- 241000416162 Astragalus gummifer Species 0.000 description 3
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 3
- 241000238631 Hexapoda Species 0.000 description 3
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 3
- 102220577899 Leucine-rich repeat serine/threonine-protein kinase 2_G2019S_mutation Human genes 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 206010037211 Psychomotor hyperactivity Diseases 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- 229920001615 Tragacanth Polymers 0.000 description 3
- 208000025865 Ulcer Diseases 0.000 description 3
- 206010052428 Wound Diseases 0.000 description 3
- 208000027418 Wounds and injury Diseases 0.000 description 3
- 150000001299 aldehydes Chemical class 0.000 description 3
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 125000006297 carbonyl amino group Chemical group [H]N([*:2])C([*:1])=O 0.000 description 3
- 230000004663 cell proliferation Effects 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000012875 competitive assay Methods 0.000 description 3
- 230000000295 complement effect Effects 0.000 description 3
- 239000006071 cream Substances 0.000 description 3
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 3
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 3
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 3
- 239000008121 dextrose Substances 0.000 description 3
- UAOMVDZJSHZZME-UHFFFAOYSA-N diisopropylamine Chemical compound CC(C)NC(C)C UAOMVDZJSHZZME-UHFFFAOYSA-N 0.000 description 3
- 239000000796 flavoring agent Substances 0.000 description 3
- 235000013355 food flavoring agent Nutrition 0.000 description 3
- 239000012634 fragment Substances 0.000 description 3
- 238000013537 high throughput screening Methods 0.000 description 3
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 3
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 238000007918 intramuscular administration Methods 0.000 description 3
- 239000008101 lactose Substances 0.000 description 3
- 229960001375 lactose Drugs 0.000 description 3
- 235000019359 magnesium stearate Nutrition 0.000 description 3
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 3
- 210000002569 neuron Anatomy 0.000 description 3
- 239000002674 ointment Substances 0.000 description 3
- 239000012044 organic layer Substances 0.000 description 3
- LMYJGUNNJIDROI-UHFFFAOYSA-N oxan-4-ol Chemical compound OC1CCOCC1 LMYJGUNNJIDROI-UHFFFAOYSA-N 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- 235000011007 phosphoric acid Nutrition 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 235000011056 potassium acetate Nutrition 0.000 description 3
- 108090000765 processed proteins & peptides Proteins 0.000 description 3
- 239000003380 propellant Substances 0.000 description 3
- 230000000069 prophylactic effect Effects 0.000 description 3
- 230000002685 pulmonary effect Effects 0.000 description 3
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 3
- 238000012207 quantitative assay Methods 0.000 description 3
- 238000012216 screening Methods 0.000 description 3
- 239000012312 sodium hydride Substances 0.000 description 3
- 229910000104 sodium hydride Inorganic materials 0.000 description 3
- RYYKJJJTJZKILX-UHFFFAOYSA-M sodium octadecanoate Chemical compound [Na+].CCCCCCCCCCCCCCCCCC([O-])=O RYYKJJJTJZKILX-UHFFFAOYSA-M 0.000 description 3
- 239000008107 starch Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 229940032147 starch Drugs 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 238000006467 substitution reaction Methods 0.000 description 3
- 239000000829 suppository Substances 0.000 description 3
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N thiocyanic acid Chemical compound SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- 230000000699 topical effect Effects 0.000 description 3
- 235000010487 tragacanth Nutrition 0.000 description 3
- 239000000196 tragacanth Substances 0.000 description 3
- 229940116362 tragacanth Drugs 0.000 description 3
- 231100000397 ulcer Toxicity 0.000 description 3
- CYPYTURSJDMMMP-WVCUSYJESA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].[Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 CYPYTURSJDMMMP-WVCUSYJESA-N 0.000 description 2
- YYRIQTLBJWWQMZ-UHFFFAOYSA-N (2,4-dichloro-6-methylpyridin-3-yl)methanol Chemical compound CC1=CC(Cl)=C(CO)C(Cl)=N1 YYRIQTLBJWWQMZ-UHFFFAOYSA-N 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 2
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 2
- 125000004605 1,2,3,4-tetrahydroisoquinolinyl group Chemical group C1(NCCC2=CC=CC=C12)* 0.000 description 2
- VUDQSRFCCHQIIU-UHFFFAOYSA-N 1-(3,5-dichloro-2,6-dihydroxy-4-methoxyphenyl)hexan-1-one Chemical compound CCCCCC(=O)C1=C(O)C(Cl)=C(OC)C(Cl)=C1O VUDQSRFCCHQIIU-UHFFFAOYSA-N 0.000 description 2
- MOHYOXXOKFQHDC-UHFFFAOYSA-N 1-(chloromethyl)-4-methoxybenzene Chemical compound COC1=CC=C(CCl)C=C1 MOHYOXXOKFQHDC-UHFFFAOYSA-N 0.000 description 2
- FEWLNYSYJNLUOO-UHFFFAOYSA-N 1-Piperidinecarboxaldehyde Chemical compound O=CN1CCCCC1 FEWLNYSYJNLUOO-UHFFFAOYSA-N 0.000 description 2
- HCGRJLPRIPXPQS-UHFFFAOYSA-N 1-[(4-methoxyphenyl)methyl]-6-methyl-n-(oxan-4-yl)-3-[4-(trifluoromethyl)pyridin-2-yl]pyrazolo[4,3-c]pyridin-4-amine Chemical compound C1=CC(OC)=CC=C1CN1C2=CC(C)=NC(NC3CCOCC3)=C2C(C=2N=CC=C(C=2)C(F)(F)F)=N1 HCGRJLPRIPXPQS-UHFFFAOYSA-N 0.000 description 2
- WYWODEVOTBCSQQ-UHFFFAOYSA-N 1-[(4-methoxyphenyl)methyl]-6-methyl-n-(oxan-4-yl)-3-trimethylstannylpyrazolo[4,3-c]pyridin-4-amine Chemical compound C1=CC(OC)=CC=C1CN1C2=CC(C)=NC(NC3CCOCC3)=C2C([Sn](C)(C)C)=N1 WYWODEVOTBCSQQ-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- QUHJCWDZCMZQFW-UHFFFAOYSA-N 2,4-dichloro-6-methylpyridine-3-carbaldehyde Chemical compound CC1=CC(Cl)=C(C=O)C(Cl)=N1 QUHJCWDZCMZQFW-UHFFFAOYSA-N 0.000 description 2
- WZVHLUMAQLUNTJ-UHFFFAOYSA-N 2-bromo-4-(trifluoromethyl)pyridine Chemical compound FC(F)(F)C1=CC=NC(Br)=C1 WZVHLUMAQLUNTJ-UHFFFAOYSA-N 0.000 description 2
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- ZOOGRGPOEVQQDX-UUOKFMHZSA-N 3',5'-cyclic GMP Chemical compound C([C@H]1O2)OP(O)(=O)O[C@H]1[C@@H](O)[C@@H]2N1C(N=C(NC2=O)N)=C2N=C1 ZOOGRGPOEVQQDX-UUOKFMHZSA-N 0.000 description 2
- BEZGMHBTKNSPPY-UHFFFAOYSA-N 3-iodo-1-[(4-methoxyphenyl)methyl]-6-methyl-n-(oxan-4-yl)pyrazolo[4,3-c]pyridin-4-amine Chemical compound C1=CC(OC)=CC=C1CN1C2=CC(C)=NC(NC3CCOCC3)=C2C(I)=N1 BEZGMHBTKNSPPY-UHFFFAOYSA-N 0.000 description 2
- CDOXXVVHKFTYSV-UHFFFAOYSA-N 3-iodo-4-(oxan-4-yloxy)-1-tritylpyrazolo[4,3-c]pyridine-6-carbonitrile Chemical compound C=12C(I)=NN(C(C=3C=CC=CC=3)(C=3C=CC=CC=3)C=3C=CC=CC=3)C2=CC(C#N)=NC=1OC1CCOCC1 CDOXXVVHKFTYSV-UHFFFAOYSA-N 0.000 description 2
- NCWASWYQPJPXFE-UHFFFAOYSA-N 3-iodo-4-methoxy-6-methyl-1-tritylpyrazolo[4,3-c]pyridine Chemical compound N1=C(I)C=2C(OC)=NC(C)=CC=2N1C(C=1C=CC=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 NCWASWYQPJPXFE-UHFFFAOYSA-N 0.000 description 2
- IPBJWBFJRGOWLF-UHFFFAOYSA-N 3-iodo-6-methyl-4-(oxan-4-yloxy)-1-tritylpyrazolo[4,3-c]pyridine Chemical compound N=1C(C)=CC=2N(C(C=3C=CC=CC=3)(C=3C=CC=CC=3)C=3C=CC=CC=3)N=C(I)C=2C=1OC1CCOCC1 IPBJWBFJRGOWLF-UHFFFAOYSA-N 0.000 description 2
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 2
- HZJIGVGLNZISJU-UHFFFAOYSA-N 4-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin-2-yl]morpholine Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC=NC(N2CCOCC2)=C1 HZJIGVGLNZISJU-UHFFFAOYSA-N 0.000 description 2
- SADQHMIOSMBQBN-UHFFFAOYSA-N 4-chloro-3-iodo-1-[(4-methoxyphenyl)methyl]-6-methylpyrazolo[4,3-c]pyridine Chemical compound C1=CC(OC)=CC=C1CN1C2=CC(C)=NC(Cl)=C2C(I)=N1 SADQHMIOSMBQBN-UHFFFAOYSA-N 0.000 description 2
- WBLXENUULTUPRQ-UHFFFAOYSA-N 4-chloro-3-iodo-6-(trifluoromethyl)-1-tritylpyrazolo[4,3-c]pyridine Chemical compound N1=C(I)C=2C(Cl)=NC(C(F)(F)F)=CC=2N1C(C=1C=CC=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 WBLXENUULTUPRQ-UHFFFAOYSA-N 0.000 description 2
- NVSNTEGQPPECRN-UHFFFAOYSA-N 4-chloro-3-iodo-6-(trifluoromethyl)-2h-pyrazolo[4,3-c]pyridine Chemical compound ClC1=NC(C(F)(F)F)=CC2=C1C(I)=NN2 NVSNTEGQPPECRN-UHFFFAOYSA-N 0.000 description 2
- FJKROLUGYXJWQN-UHFFFAOYSA-N 4-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 2
- SSGONHNNSIBLDS-UHFFFAOYSA-N 6-[6-methyl-4-(oxan-4-ylamino)-1h-pyrazolo[4,3-c]pyridin-3-yl]-1h-pyrimidin-4-one Chemical compound C1=2C(NC3CCOCC3)=NC(C)=CC=2NN=C1C1=CC(O)=NC=N1 SSGONHNNSIBLDS-UHFFFAOYSA-N 0.000 description 2
- FUUKOQQORKOMBE-UHFFFAOYSA-N 6-methyl-4-(oxan-4-yloxy)-3-(1H-pyrazol-4-yl)-1-tritylpyrazolo[4,3-c]pyridine Chemical compound C1=2C(OC3CCOCC3)=NC(C)=CC=2N(C(C=2C=CC=CC=2)(C=2C=CC=CC=2)C=2C=CC=CC=2)N=C1C=1C=NNC=1 FUUKOQQORKOMBE-UHFFFAOYSA-N 0.000 description 2
- 108010085238 Actins Proteins 0.000 description 2
- 102000007469 Actins Human genes 0.000 description 2
- 239000005711 Benzoic acid Substances 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- 241000224495 Dictyostelium Species 0.000 description 2
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 description 2
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 2
- 239000007821 HATU Substances 0.000 description 2
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 2
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 2
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 2
- 108010006444 Leucine-Rich Repeat Proteins Proteins 0.000 description 2
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 235000019502 Orange oil Nutrition 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 235000010323 ascorbic acid Nutrition 0.000 description 2
- 229960005070 ascorbic acid Drugs 0.000 description 2
- 239000011668 ascorbic acid Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 235000010233 benzoic acid Nutrition 0.000 description 2
- MUALRAIOVNYAIW-UHFFFAOYSA-N binap Chemical compound C1=CC=CC=C1P(C=1C(=C2C=CC=CC2=CC=1)C=1C2=CC=CC=C2C=CC=1P(C=1C=CC=CC=1)C=1C=CC=CC=1)C1=CC=CC=C1 MUALRAIOVNYAIW-UHFFFAOYSA-N 0.000 description 2
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 2
- 210000004899 c-terminal region Anatomy 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 230000024245 cell differentiation Effects 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 210000000170 cell membrane Anatomy 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- 235000015165 citric acid Nutrition 0.000 description 2
- 239000007891 compressed tablet Substances 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 239000003405 delayed action preparation Substances 0.000 description 2
- 229910052805 deuterium Inorganic materials 0.000 description 2
- WGLUMOCWFMKWIL-UHFFFAOYSA-N dichloromethane;methanol Chemical compound OC.ClCCl WGLUMOCWFMKWIL-UHFFFAOYSA-N 0.000 description 2
- 239000002270 dispersing agent Substances 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- WBJINCZRORDGAQ-UHFFFAOYSA-N formic acid ethyl ester Natural products CCOC=O WBJINCZRORDGAQ-UHFFFAOYSA-N 0.000 description 2
- 239000001530 fumaric acid Substances 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- PZJSZBJLOWMDRG-UHFFFAOYSA-N furan-2-ylboronic acid Chemical compound OB(O)C1=CC=CO1 PZJSZBJLOWMDRG-UHFFFAOYSA-N 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 235000013922 glutamic acid Nutrition 0.000 description 2
- 239000004220 glutamic acid Substances 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 229940093915 gynecological organic acid Drugs 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 150000004679 hydroxides Chemical class 0.000 description 2
- 125000002883 imidazolyl group Chemical group 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- TWBYWOBDOCUKOW-UHFFFAOYSA-N isonicotinic acid Chemical compound OC(=O)C1=CC=NC=C1 TWBYWOBDOCUKOW-UHFFFAOYSA-N 0.000 description 2
- 239000004310 lactic acid Substances 0.000 description 2
- 235000014655 lactic acid Nutrition 0.000 description 2
- 210000004901 leucine-rich repeat Anatomy 0.000 description 2
- 239000011976 maleic acid Substances 0.000 description 2
- 239000001630 malic acid Substances 0.000 description 2
- 235000011090 malic acid Nutrition 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 229960001855 mannitol Drugs 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000002503 metabolic effect Effects 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 229920000609 methyl cellulose Polymers 0.000 description 2
- 239000001923 methylcellulose Substances 0.000 description 2
- 229960002900 methylcellulose Drugs 0.000 description 2
- 235000010981 methylcellulose Nutrition 0.000 description 2
- 239000007932 molded tablet Substances 0.000 description 2
- 210000000214 mouth Anatomy 0.000 description 2
- 239000010502 orange oil Substances 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 235000006408 oxalic acid Nutrition 0.000 description 2
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 2
- 238000007911 parenteral administration Methods 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 230000036470 plasma concentration Effects 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- VVWRJUBEIPHGQF-MDZDMXLPSA-N propan-2-yl (ne)-n-propan-2-yloxycarbonyliminocarbamate Chemical compound CC(C)OC(=O)\N=N\C(=O)OC(C)C VVWRJUBEIPHGQF-MDZDMXLPSA-N 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 238000007493 shaping process Methods 0.000 description 2
- 230000019491 signal transduction Effects 0.000 description 2
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 2
- 239000004299 sodium benzoate Substances 0.000 description 2
- 235000010234 sodium benzoate Nutrition 0.000 description 2
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 2
- HRZFUMHJMZEROT-UHFFFAOYSA-L sodium disulfite Chemical class [Na+].[Na+].[O-]S(=O)S([O-])(=O)=O HRZFUMHJMZEROT-UHFFFAOYSA-L 0.000 description 2
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 2
- 239000012453 solvate Substances 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 229960002920 sorbitol Drugs 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 238000010561 standard procedure Methods 0.000 description 2
- 238000012799 strong cation exchange Methods 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 150000003460 sulfonic acids Chemical class 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 239000011975 tartaric acid Substances 0.000 description 2
- 235000002906 tartaric acid Nutrition 0.000 description 2
- 229940095064 tartrate Drugs 0.000 description 2
- OSBSFAARYOCBHB-UHFFFAOYSA-N tetrapropylammonium Chemical compound CCC[N+](CCC)(CCC)CCC OSBSFAARYOCBHB-UHFFFAOYSA-N 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- 231100000331 toxic Toxicity 0.000 description 2
- 230000002588 toxic effect Effects 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- XFNJVJPLKCPIBV-UHFFFAOYSA-N trimethylenediamine Chemical compound NCCCN XFNJVJPLKCPIBV-UHFFFAOYSA-N 0.000 description 2
- JBWKIWSBJXDJDT-UHFFFAOYSA-N triphenylmethyl chloride Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(Cl)C1=CC=CC=C1 JBWKIWSBJXDJDT-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- CXNIUSPIQKWYAI-UHFFFAOYSA-N xantphos Chemical compound C=12OC3=C(P(C=4C=CC=CC=4)C=4C=CC=CC=4)C=CC=C3C(C)(C)C2=CC=CC=1P(C=1C=CC=CC=1)C1=CC=CC=C1 CXNIUSPIQKWYAI-UHFFFAOYSA-N 0.000 description 2
- LSPHULWDVZXLIL-UHFFFAOYSA-N (+/-)-Camphoric acid Chemical compound CC1(C)C(C(O)=O)CCC1(C)C(O)=O LSPHULWDVZXLIL-UHFFFAOYSA-N 0.000 description 1
- RRYCUINOUZEQSU-UHFFFAOYSA-N (2-morpholin-4-ylpyridin-4-yl)boronic acid Chemical compound OB(O)C1=CC=NC(N2CCOCC2)=C1 RRYCUINOUZEQSU-UHFFFAOYSA-N 0.000 description 1
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 1
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 1
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 1
- YMEBZRNYQBODKB-UHFFFAOYSA-N 1-(2-methylpropyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole Chemical compound C1=NN(CC(C)C)C=C1B1OC(C)(C)C(C)(C)O1 YMEBZRNYQBODKB-UHFFFAOYSA-N 0.000 description 1
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide Substances CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- UCNGGGYMLHAMJG-UHFFFAOYSA-N 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole Chemical compound C1=NN(C)C=C1B1OC(C)(C)C(C)(C)O1 UCNGGGYMLHAMJG-UHFFFAOYSA-N 0.000 description 1
- OGYYMVGDKVJYSU-UHFFFAOYSA-N 1-propan-2-yl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole Chemical compound C1=NN(C(C)C)C=C1B1OC(C)(C)C(C)(C)O1 OGYYMVGDKVJYSU-UHFFFAOYSA-N 0.000 description 1
- YLEWQGYQMZGXSD-UHFFFAOYSA-N 1h-pyrazolo[4,3-c]pyridin-4-amine Chemical compound NC1=NC=CC2=C1C=NN2 YLEWQGYQMZGXSD-UHFFFAOYSA-N 0.000 description 1
- WCXFPLXZZSWROM-UHFFFAOYSA-N 1h-pyrazolo[4,3-c]pyridine Chemical class C1=NC=C2C=NNC2=C1 WCXFPLXZZSWROM-UHFFFAOYSA-N 0.000 description 1
- FJNNGKMAGDPVIU-UHFFFAOYSA-N 2,4,6-trichloropyridine Chemical compound ClC1=CC(Cl)=NC(Cl)=C1 FJNNGKMAGDPVIU-UHFFFAOYSA-N 0.000 description 1
- PAQZWJGSJMLPMG-UHFFFAOYSA-N 2,4,6-tripropyl-1,3,5,2$l^{5},4$l^{5},6$l^{5}-trioxatriphosphinane 2,4,6-trioxide Chemical compound CCCP1(=O)OP(=O)(CCC)OP(=O)(CCC)O1 PAQZWJGSJMLPMG-UHFFFAOYSA-N 0.000 description 1
- BJHUMUZOLTXVSC-UHFFFAOYSA-N 2,4-dichloro-6-(trifluoromethyl)pyridine Chemical compound FC(F)(F)C1=CC(Cl)=CC(Cl)=N1 BJHUMUZOLTXVSC-UHFFFAOYSA-N 0.000 description 1
- CKWYPOOWHICBJN-UHFFFAOYSA-N 2,4-dichloro-6-(trifluoromethyl)pyridine-3-carbaldehyde Chemical compound FC(F)(F)C1=CC(Cl)=C(C=O)C(Cl)=N1 CKWYPOOWHICBJN-UHFFFAOYSA-N 0.000 description 1
- CIVCELMLGDGMKZ-UHFFFAOYSA-N 2,4-dichloro-6-methylpyridine-3-carboxylic acid Chemical compound CC1=CC(Cl)=C(C(O)=O)C(Cl)=N1 CIVCELMLGDGMKZ-UHFFFAOYSA-N 0.000 description 1
- MQJGDXNHVTZVCA-UHFFFAOYSA-N 2-[1-[(4-methoxyphenyl)methyl]-4-(oxan-4-ylamino)pyrazolo[4,3-c]pyridin-3-yl]pyridine-4-carboxylic acid Chemical compound C1=CC(OC)=CC=C1CN1C2=CC=NC(NC3CCOCC3)=C2C(C=2N=CC=C(C=2)C(O)=O)=N1 MQJGDXNHVTZVCA-UHFFFAOYSA-N 0.000 description 1
- VHNGECLFCAOZGE-UHFFFAOYSA-N 2-[1-[(4-methoxyphenyl)methyl]-6-methyl-4-(oxan-4-ylamino)pyrazolo[4,3-c]pyridin-3-yl]-n,n-dimethylpyridine-4-carboxamide Chemical compound C1=CC(OC)=CC=C1CN1C2=CC(C)=NC(NC3CCOCC3)=C2C(C=2N=CC=C(C=2)C(=O)N(C)C)=N1 VHNGECLFCAOZGE-UHFFFAOYSA-N 0.000 description 1
- KISWVXRQTGLFGD-UHFFFAOYSA-N 2-[[2-[[6-amino-2-[[2-[[2-[[5-amino-2-[[2-[[1-[2-[[6-amino-2-[(2,5-diamino-5-oxopentanoyl)amino]hexanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]pyrrolidine-2-carbonyl]amino]-3-hydroxypropanoyl]amino]-5-oxopentanoyl]amino]-5-(diaminomethylideneamino)p Chemical compound C1CCN(C(=O)C(CCCN=C(N)N)NC(=O)C(CCCCN)NC(=O)C(N)CCC(N)=O)C1C(=O)NC(CO)C(=O)NC(CCC(N)=O)C(=O)NC(CCCN=C(N)N)C(=O)NC(CO)C(=O)NC(CCCCN)C(=O)NC(C(=O)NC(CC(C)C)C(O)=O)CC1=CC=C(O)C=C1 KISWVXRQTGLFGD-UHFFFAOYSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- RBFNWOINNIOZKR-UHFFFAOYSA-N 3,5-dichloropyridine-4-carbaldehyde Chemical compound ClC1=CN=CC(Cl)=C1C=O RBFNWOINNIOZKR-UHFFFAOYSA-N 0.000 description 1
- MBCYZFPHWNCLGH-UHFFFAOYSA-N 3-(4-methoxy-6-methyl-1h-pyrazolo[4,3-c]pyridin-3-yl)-n-methylbenzamide Chemical compound CNC(=O)C1=CC=CC(C=2C3=C(OC)N=C(C)C=C3NN=2)=C1 MBCYZFPHWNCLGH-UHFFFAOYSA-N 0.000 description 1
- ATPDWNNAAIJINY-UHFFFAOYSA-N 3-(6-chloropyrimidin-4-yl)-6-methyl-n-(oxan-4-yl)-1h-pyrazolo[4,3-c]pyridin-4-amine Chemical compound C1=2C(NC3CCOCC3)=NC(C)=CC=2NN=C1C1=CC(Cl)=NC=N1 ATPDWNNAAIJINY-UHFFFAOYSA-N 0.000 description 1
- QWRPRRAVTKEBHO-UHFFFAOYSA-N 3-(6-ethoxypyrimidin-4-yl)-6-methyl-n-(oxan-4-yl)-1h-pyrazolo[4,3-c]pyridin-4-amine Chemical compound C1=NC(OCC)=CC(C=2C3=C(NC4CCOCC4)N=C(C)C=C3NN=2)=N1 QWRPRRAVTKEBHO-UHFFFAOYSA-N 0.000 description 1
- FPQQSJJWHUJYPU-UHFFFAOYSA-N 3-(dimethylamino)propyliminomethylidene-ethylazanium;chloride Chemical compound Cl.CCN=C=NCCCN(C)C FPQQSJJWHUJYPU-UHFFFAOYSA-N 0.000 description 1
- ALCBGQOYBWXONQ-UHFFFAOYSA-N 3-[1-(2-methylpropyl)pyrazol-4-yl]-4-(oxan-4-yloxy)-1h-pyrazolo[4,3-c]pyridine-6-carbonitrile Chemical compound C1=NN(CC(C)C)C=C1C1=NNC2=CC(C#N)=NC(OC3CCOCC3)=C12 ALCBGQOYBWXONQ-UHFFFAOYSA-N 0.000 description 1
- NLCAHJGXQGIZPE-UHFFFAOYSA-N 3-iodo-4-(oxan-4-yloxy)-1-tritylpyrazolo[4,3-c]pyridine-6-carbaldehyde Chemical compound C=12C(I)=NN(C(C=3C=CC=CC=3)(C=3C=CC=CC=3)C=3C=CC=CC=3)C2=CC(C=O)=NC=1OC1CCOCC1 NLCAHJGXQGIZPE-UHFFFAOYSA-N 0.000 description 1
- XMIIGOLPHOKFCH-UHFFFAOYSA-M 3-phenylpropionate Chemical compound [O-]C(=O)CCC1=CC=CC=C1 XMIIGOLPHOKFCH-UHFFFAOYSA-M 0.000 description 1
- XJPZKYIHCLDXST-UHFFFAOYSA-N 4,6-dichloropyrimidine Chemical compound ClC1=CC(Cl)=NC=N1 XJPZKYIHCLDXST-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- TVOJIBGZFYMWDT-UHFFFAOYSA-N 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-pyrazole Chemical compound O1C(C)(C)C(C)(C)OB1C1=CNN=C1 TVOJIBGZFYMWDT-UHFFFAOYSA-N 0.000 description 1
- KXVYFOALBMHJLK-UHFFFAOYSA-N 4-(oxan-4-yloxy)-3-(1-propan-2-ylpyrazol-4-yl)-6-(trifluoromethyl)-1h-pyrazolo[4,3-c]pyridine Chemical compound C1=NN(C(C)C)C=C1C1=NNC2=CC(C(F)(F)F)=NC(OC3CCOCC3)=C12 KXVYFOALBMHJLK-UHFFFAOYSA-N 0.000 description 1
- TTWCQUPIRJNGGR-UHFFFAOYSA-N 4-[4-(4-methoxy-6-methyl-1-tritylpyrazolo[4,3-c]pyridin-3-yl)pyridin-2-yl]morpholine Chemical compound C1=2C(OC)=NC(C)=CC=2N(C(C=2C=CC=CC=2)(C=2C=CC=CC=2)C=2C=CC=CC=2)N=C1C(C=1)=CC=NC=1N1CCOCC1 TTWCQUPIRJNGGR-UHFFFAOYSA-N 0.000 description 1
- LUBHCPRJAHZHGB-UHFFFAOYSA-N 4-[4-[6-chloro-4-(oxan-4-yloxy)-1h-pyrazolo[4,3-c]pyridin-3-yl]pyridin-2-yl]morpholine Chemical compound C1=2C(OC3CCOCC3)=NC(Cl)=CC=2NN=C1C(C=1)=CC=NC=1N1CCOCC1 LUBHCPRJAHZHGB-UHFFFAOYSA-N 0.000 description 1
- RBHXAAPOZNMAMC-UHFFFAOYSA-N 4-[4-[6-methyl-4-(oxan-4-yloxy)-1h-pyrazolo[4,3-c]pyridin-3-yl]pyridin-2-yl]morpholine Chemical compound C1=2C(OC3CCOCC3)=NC(C)=CC=2NN=C1C(C=1)=CC=NC=1N1CCOCC1 RBHXAAPOZNMAMC-UHFFFAOYSA-N 0.000 description 1
- NUKYPUAOHBNCPY-UHFFFAOYSA-N 4-aminopyridine Chemical compound NC1=CC=NC=C1 NUKYPUAOHBNCPY-UHFFFAOYSA-N 0.000 description 1
- AGUIEFSLABIYOT-UHFFFAOYSA-N 4-bromo-6-(trifluoromethyl)pyrimidine Chemical compound FC(F)(F)C1=CC(Br)=NC=N1 AGUIEFSLABIYOT-UHFFFAOYSA-N 0.000 description 1
- XCDZWNHCHWCZSW-UHFFFAOYSA-N 4-chloro-3-iodo-6-methyl-1-tritylpyrazolo[4,3-c]pyridine Chemical compound N1=C(I)C=2C(Cl)=NC(C)=CC=2N1C(C=1C=CC=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 XCDZWNHCHWCZSW-UHFFFAOYSA-N 0.000 description 1
- JCYLHBTYZNJFDZ-UHFFFAOYSA-N 4-chloro-6-(trifluoromethyl)-1h-pyrazolo[4,3-c]pyridine Chemical compound ClC1=NC(C(F)(F)F)=CC2=C1C=NN2 JCYLHBTYZNJFDZ-UHFFFAOYSA-N 0.000 description 1
- OHSIWTLJMVJMJD-UHFFFAOYSA-N 4-chloro-6-methyl-1h-pyrazolo[4,3-c]pyridine Chemical compound ClC1=NC(C)=CC2=C1C=NN2 OHSIWTLJMVJMJD-UHFFFAOYSA-N 0.000 description 1
- RJWBTWIBUIGANW-UHFFFAOYSA-N 4-chlorobenzenesulfonic acid Chemical class OS(=O)(=O)C1=CC=C(Cl)C=C1 RJWBTWIBUIGANW-UHFFFAOYSA-N 0.000 description 1
- KZBHZCNVUKINKW-UHFFFAOYSA-N 4-cyclohexyloxy-6-cyclopropyl-3-methyl-2h-pyrazolo[4,3-c]pyridine Chemical compound C=12C(C)=NNC2=CC(C2CC2)=NC=1OC1CCCCC1 KZBHZCNVUKINKW-UHFFFAOYSA-N 0.000 description 1
- 229940090248 4-hydroxybenzoic acid Drugs 0.000 description 1
- OALSKUQPBIJEBT-UHFFFAOYSA-N 4-methoxy-6-methyl-3-[1-(oxan-4-yl)pyrazol-4-yl]-1h-pyrazolo[4,3-c]pyridine Chemical compound C1=2C(OC)=NC(C)=CC=2NN=C1C(=C1)C=NN1C1CCOCC1 OALSKUQPBIJEBT-UHFFFAOYSA-N 0.000 description 1
- TUIDQYRZDZRHPQ-UHFFFAOYSA-N 5-chloropyridin-3-ol Chemical compound OC1=CN=CC(Cl)=C1 TUIDQYRZDZRHPQ-UHFFFAOYSA-N 0.000 description 1
- NILPWMFPNQJXJG-UHFFFAOYSA-N 6-chloro-3-iodo-4-(oxan-4-yloxy)-1-tritylpyrazolo[4,3-c]pyridine Chemical compound N=1C(Cl)=CC=2N(C(C=3C=CC=CC=3)(C=3C=CC=CC=3)C=3C=CC=CC=3)N=C(I)C=2C=1OC1CCOCC1 NILPWMFPNQJXJG-UHFFFAOYSA-N 0.000 description 1
- BMKXWRHRYKXSMH-UHFFFAOYSA-N 6-chloro-4-cyclohexyloxy-3-methyl-2h-pyrazolo[4,3-c]pyridine Chemical compound C=12C(C)=NNC2=CC(Cl)=NC=1OC1CCCCC1 BMKXWRHRYKXSMH-UHFFFAOYSA-N 0.000 description 1
- PXNSLLZBSQFWMF-UHFFFAOYSA-N 6-methyl-4-(oxan-4-yloxy)-3-[1-(oxan-4-yl)pyrazol-4-yl]-1-tritylpyrazolo[4,3-c]pyridine Chemical compound C1=2C(OC3CCOCC3)=NC(C)=CC=2N(C(C=2C=CC=CC=2)(C=2C=CC=CC=2)C=2C=CC=CC=2)N=C1C(=C1)C=NN1C1CCOCC1 PXNSLLZBSQFWMF-UHFFFAOYSA-N 0.000 description 1
- GEAQZZWVYYEZKE-UHFFFAOYSA-N 6-methyl-n-(oxan-4-yl)-3-[4-(trifluoromethyl)pyridin-2-yl]-1h-pyrazolo[4,3-c]pyridin-4-amine Chemical compound C1=2C(NC3CCOCC3)=NC(C)=CC=2NN=C1C1=CC(C(F)(F)F)=CC=N1 GEAQZZWVYYEZKE-UHFFFAOYSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- ATRRKUHOCOJYRX-UHFFFAOYSA-N Ammonium bicarbonate Chemical compound [NH4+].OC([O-])=O ATRRKUHOCOJYRX-UHFFFAOYSA-N 0.000 description 1
- 229910000013 Ammonium bicarbonate Inorganic materials 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-DCSYEGIMSA-N Beta-Lactose Chemical compound OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-DCSYEGIMSA-N 0.000 description 1
- 241000167854 Bourreria succulenta Species 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- 125000001433 C-terminal amino-acid group Chemical group 0.000 description 1
- ANVUICNGPOHXRE-UHFFFAOYSA-N C1(=CC=CC=C1)P(C1=CC=CC=C1)[C-]1C=CC=C1.[CH-]1C=CC=C1.[Fe+2].Cl.Cl Chemical compound C1(=CC=CC=C1)P(C1=CC=CC=C1)[C-]1C=CC=C1.[CH-]1C=CC=C1.[Fe+2].Cl.Cl ANVUICNGPOHXRE-UHFFFAOYSA-N 0.000 description 1
- 0 CC(C)[C@@](CN)[C@@](C)C(C)* Chemical compound CC(C)[C@@](CN)[C@@](C)C(C)* 0.000 description 1
- RUOPESHGFXYMCE-PKPIPKONSA-N CC(N1N=C(C)C[C@@H]1C)I Chemical compound CC(N1N=C(C)C[C@@H]1C)I RUOPESHGFXYMCE-PKPIPKONSA-N 0.000 description 1
- YEBBCAXAPQQICH-UHFFFAOYSA-N CC(c(c(Cl)cc(Cl)n1)c1Cl)=O Chemical compound CC(c(c(Cl)cc(Cl)n1)c1Cl)=O YEBBCAXAPQQICH-UHFFFAOYSA-N 0.000 description 1
- OVRKATYHWPCGPZ-UHFFFAOYSA-N CC1CCOCC1 Chemical compound CC1CCOCC1 OVRKATYHWPCGPZ-UHFFFAOYSA-N 0.000 description 1
- SXVSQYAEJMBGBN-MUMRKEEXSA-N CCCC([C@@H](Cc1c2c(-c3c[n](C(C)C)nc3)n[nH]1)N=C2OC(CCOC)C(C)C)(F)F Chemical compound CCCC([C@@H](Cc1c2c(-c3c[n](C(C)C)nc3)n[nH]1)N=C2OC(CCOC)C(C)C)(F)F SXVSQYAEJMBGBN-MUMRKEEXSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-NJFSPNSNSA-N Carbon-14 Chemical compound [14C] OKTJSMMVPCPJKN-NJFSPNSNSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- UIIOSUKUDFDWQA-UHFFFAOYSA-N Cc1n[nH]c2cc(Cl)nc(NC3CCCCC3)c12 Chemical compound Cc1n[nH]c2cc(Cl)nc(NC3CCCCC3)c12 UIIOSUKUDFDWQA-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 description 1
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- 102100030013 Endoribonuclease Human genes 0.000 description 1
- 101710199605 Endoribonuclease Proteins 0.000 description 1
- 108090000371 Esterases Proteins 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- 102100020903 Ezrin Human genes 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
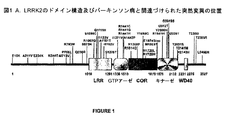
- 101100021877 Homo sapiens LRRK2 gene Proteins 0.000 description 1
- 101000941877 Homo sapiens Leucine-rich repeat serine/threonine-protein kinase 1 Proteins 0.000 description 1
- 101000941879 Homo sapiens Leucine-rich repeat serine/threonine-protein kinase 2 Proteins 0.000 description 1
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 1
- 101150081013 LRRK2 gene Proteins 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 102100032656 Leucine-rich repeat serine/threonine-protein kinase 1 Human genes 0.000 description 1
- 102220578292 Leucine-rich repeat serine/threonine-protein kinase 2_D1994A_mutation Human genes 0.000 description 1
- 238000003820 Medium-pressure liquid chromatography Methods 0.000 description 1
- 108010052285 Membrane Proteins Proteins 0.000 description 1
- 244000246386 Mentha pulegium Species 0.000 description 1
- 235000016257 Mentha pulegium Nutrition 0.000 description 1
- 235000004357 Mentha x piperita Nutrition 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 102000047918 Myelin Basic Human genes 0.000 description 1
- 101710107068 Myelin basic protein Proteins 0.000 description 1
- 108060008487 Myosin Proteins 0.000 description 1
- 102000003505 Myosin Human genes 0.000 description 1
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 1
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 102000012515 Protein kinase domains Human genes 0.000 description 1
- 108050002122 Protein kinase domains Proteins 0.000 description 1
- 102100022127 Radixin Human genes 0.000 description 1
- 108090000829 Ribosome Inactivating Proteins Proteins 0.000 description 1
- 101710113029 Serine/threonine-protein kinase Proteins 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- BCKXLBQYZLBQEK-KVVVOXFISA-M Sodium oleate Chemical compound [Na+].CCCCCCCC\C=C/CCCCCCCC([O-])=O BCKXLBQYZLBQEK-KVVVOXFISA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-M Thiocyanate anion Chemical compound [S-]C#N ZMZDMBWJUHKJPS-UHFFFAOYSA-M 0.000 description 1
- DTQVDTLACAAQTR-UHFFFAOYSA-M Trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-M 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- WNLRTRBMVRJNCN-UHFFFAOYSA-L adipate(2-) Chemical compound [O-]C(=O)CCCCC([O-])=O WNLRTRBMVRJNCN-UHFFFAOYSA-L 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- WQZGKKKJIJFFOK-DVKNGEFBSA-N alpha-D-glucose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-DVKNGEFBSA-N 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 1
- 235000012538 ammonium bicarbonate Nutrition 0.000 description 1
- 239000001099 ammonium carbonate Substances 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 229960004977 anhydrous lactose Drugs 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 239000008365 aqueous carrier Substances 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- 125000005362 aryl sulfone group Chemical group 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 230000035578 autophosphorylation Effects 0.000 description 1
- ZXVOCOLRQJZVBW-UHFFFAOYSA-N azane;ethanol Chemical compound N.CCO ZXVOCOLRQJZVBW-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 150000008107 benzenesulfonic acids Chemical class 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 229940050390 benzoate Drugs 0.000 description 1
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- XMIIGOLPHOKFCH-UHFFFAOYSA-N beta-phenylpropanoic acid Natural products OC(=O)CCC1=CC=CC=C1 XMIIGOLPHOKFCH-UHFFFAOYSA-N 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 230000036765 blood level Effects 0.000 description 1
- SXDBWCPKPHAZSM-UHFFFAOYSA-N bromic acid Chemical compound OBr(=O)=O SXDBWCPKPHAZSM-UHFFFAOYSA-N 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 1
- 229910000024 caesium carbonate Inorganic materials 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- BPKIGYQJPYCAOW-FFJTTWKXSA-I calcium;potassium;disodium;(2s)-2-hydroxypropanoate;dichloride;dihydroxide;hydrate Chemical compound O.[OH-].[OH-].[Na+].[Na+].[Cl-].[Cl-].[K+].[Ca+2].C[C@H](O)C([O-])=O BPKIGYQJPYCAOW-FFJTTWKXSA-I 0.000 description 1
- MIOPJNTWMNEORI-UHFFFAOYSA-N camphorsulfonic acid Chemical compound C1CC2(CS(O)(=O)=O)C(=O)CC1C2(C)C MIOPJNTWMNEORI-UHFFFAOYSA-N 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 229940105329 carboxymethylcellulose Drugs 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 230000035605 chemotaxis Effects 0.000 description 1
- 229940044683 chemotherapy drug Drugs 0.000 description 1
- 235000019693 cherries Nutrition 0.000 description 1
- KYKAJFCTULSVSH-UHFFFAOYSA-N chloro(fluoro)methane Chemical compound F[C]Cl KYKAJFCTULSVSH-UHFFFAOYSA-N 0.000 description 1
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 1
- 229940001468 citrate Drugs 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 229940110456 cocoa butter Drugs 0.000 description 1
- 235000019868 cocoa butter Nutrition 0.000 description 1
- 239000008119 colloidal silica Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- 230000001268 conjugating effect Effects 0.000 description 1
- 230000006552 constitutive activation Effects 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 229940099112 cornstarch Drugs 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- NXQGGXCHGDYOHB-UHFFFAOYSA-L cyclopenta-1,4-dien-1-yl(diphenyl)phosphane;dichloropalladium;iron(2+) Chemical compound [Fe+2].Cl[Pd]Cl.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 NXQGGXCHGDYOHB-UHFFFAOYSA-L 0.000 description 1
- JBDSSBMEKXHSJF-UHFFFAOYSA-N cyclopentanecarboxylic acid Chemical compound OC(=O)C1CCCC1 JBDSSBMEKXHSJF-UHFFFAOYSA-N 0.000 description 1
- JCWIWBWXCVGEAN-UHFFFAOYSA-L cyclopentyl(diphenyl)phosphane;dichloropalladium;iron Chemical compound [Fe].Cl[Pd]Cl.[CH]1[CH][CH][CH][C]1P(C=1C=CC=CC=1)C1=CC=CC=C1.[CH]1[CH][CH][CH][C]1P(C=1C=CC=CC=1)C1=CC=CC=C1 JCWIWBWXCVGEAN-UHFFFAOYSA-L 0.000 description 1
- WLVKDFJTYKELLQ-UHFFFAOYSA-N cyclopropylboronic acid Chemical compound OB(O)C1CC1 WLVKDFJTYKELLQ-UHFFFAOYSA-N 0.000 description 1
- 210000004292 cytoskeleton Anatomy 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 239000007933 dermal patch Substances 0.000 description 1
- 210000004207 dermis Anatomy 0.000 description 1
- 125000004431 deuterium atom Chemical group 0.000 description 1
- VLCINIKIVYNLPT-UHFFFAOYSA-J dicalcium;hydrogen phosphate Chemical compound [Ca+2].[Ca+2].OP(O)([O-])=O.[O-]P([O-])([O-])=O VLCINIKIVYNLPT-UHFFFAOYSA-J 0.000 description 1
- 150000001990 dicarboxylic acid derivatives Chemical class 0.000 description 1
- 150000001991 dicarboxylic acids Chemical class 0.000 description 1
- MQYQOVYIJOLTNX-UHFFFAOYSA-N dichloromethane;n,n-dimethylformamide Chemical group ClCCl.CN(C)C=O MQYQOVYIJOLTNX-UHFFFAOYSA-N 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 229940043279 diisopropylamine Drugs 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- XHFGWHUWQXTGAT-UHFFFAOYSA-N dimethylamine hydrochloride Natural products CNC(C)C XHFGWHUWQXTGAT-UHFFFAOYSA-N 0.000 description 1
- IQDGSYLLQPDQDV-UHFFFAOYSA-N dimethylazanium;chloride Chemical compound Cl.CNC IQDGSYLLQPDQDV-UHFFFAOYSA-N 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 239000000890 drug combination Substances 0.000 description 1
- 238000007878 drug screening assay Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 235000006694 eating habits Nutrition 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- LJQKCYFTNDAAPC-UHFFFAOYSA-N ethanol;ethyl acetate Chemical compound CCO.CCOC(C)=O LJQKCYFTNDAAPC-UHFFFAOYSA-N 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- DEFVIWRASFVYLL-UHFFFAOYSA-N ethylene glycol bis(2-aminoethyl)tetraacetic acid Chemical compound OC(=O)CN(CC(O)=O)CCOCCOCCN(CC(O)=O)CC(O)=O DEFVIWRASFVYLL-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 108010055671 ezrin Proteins 0.000 description 1
- 229960004979 fampridine Drugs 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 238000012252 genetic analysis Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229940050410 gluconate Drugs 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 229960005150 glycerol Drugs 0.000 description 1
- QFWPJPIVLCBXFJ-UHFFFAOYSA-N glymidine Chemical compound N1=CC(OCCOC)=CN=C1NS(=O)(=O)C1=CC=CC=C1 QFWPJPIVLCBXFJ-UHFFFAOYSA-N 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid Chemical compound CCCCCCC(O)=O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 235000001050 hortel pimenta Nutrition 0.000 description 1
- 229910000042 hydrogen bromide Inorganic materials 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 229910000043 hydrogen iodide Inorganic materials 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000003999 initiator Substances 0.000 description 1
- 229910001853 inorganic hydroxide Inorganic materials 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- UEXQBEVWFZKHNB-UHFFFAOYSA-N intermediate 29 Natural products C1=CC(N)=CC=C1NC1=NC=CC=N1 UEXQBEVWFZKHNB-UHFFFAOYSA-N 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- TWBYWOBDOCUKOW-UHFFFAOYSA-M isonicotinate Chemical compound [O-]C(=O)C1=CC=NC=C1 TWBYWOBDOCUKOW-UHFFFAOYSA-M 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 230000000155 isotopic effect Effects 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 229960000829 kaolin Drugs 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 238000000021 kinase assay Methods 0.000 description 1
- 229940001447 lactate Drugs 0.000 description 1
- 229940099584 lactobionate Drugs 0.000 description 1
- JYTUSYBCFIZPBE-AMTLMPIISA-N lactobionic acid Chemical compound OC(=O)[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O JYTUSYBCFIZPBE-AMTLMPIISA-N 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 239000012280 lithium aluminium hydride Substances 0.000 description 1
- DLEDOFVPSDKWEF-UHFFFAOYSA-N lithium butane Chemical compound [Li+].CCC[CH2-] DLEDOFVPSDKWEF-UHFFFAOYSA-N 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- 229940057948 magnesium stearate Drugs 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 229940049920 malate Drugs 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 231100000682 maximum tolerated dose Toxicity 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- OKKJLVBELUTLKV-VMNATFBRSA-N methanol-d1 Chemical compound [2H]OC OKKJLVBELUTLKV-VMNATFBRSA-N 0.000 description 1
- UHNSRFWQBVXBSK-UHFFFAOYSA-N methanol;2,2,2-trifluoroacetic acid Chemical compound OC.OC(=O)C(F)(F)F UHNSRFWQBVXBSK-UHFFFAOYSA-N 0.000 description 1
- GRWIABMEEKERFV-UHFFFAOYSA-N methanol;oxolane Chemical compound OC.C1CCOC1 GRWIABMEEKERFV-UHFFFAOYSA-N 0.000 description 1
- LXQZBPLSOSNWJR-UHFFFAOYSA-N methyl 2,4-dichloro-6-methylpyridine-3-carboxylate Chemical compound COC(=O)C1=C(Cl)C=C(C)N=C1Cl LXQZBPLSOSNWJR-UHFFFAOYSA-N 0.000 description 1
- MULLTHQTADMZDM-UHFFFAOYSA-N methyl 2-bromopyridine-4-carboxylate Chemical compound COC(=O)C1=CC=NC(Br)=C1 MULLTHQTADMZDM-UHFFFAOYSA-N 0.000 description 1
- OSWPMRLSEDHDFF-UHFFFAOYSA-N methyl salicylate Chemical compound COC(=O)C1=CC=CC=C1O OSWPMRLSEDHDFF-UHFFFAOYSA-N 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 210000003632 microfilament Anatomy 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 239000003595 mist Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 239000002324 mouth wash Substances 0.000 description 1
- NATCMBCMJWNMNR-UHFFFAOYSA-N n,n-dimethyl-2-[6-methyl-4-(oxan-4-ylamino)-1h-pyrazolo[4,3-c]pyridin-3-yl]pyridine-4-carboxamide Chemical compound CN(C)C(=O)C1=CC=NC(C=2C3=C(NC4CCOCC4)N=C(C)C=C3NN=2)=C1 NATCMBCMJWNMNR-UHFFFAOYSA-N 0.000 description 1
- SBJUHFSCCCRPAY-UHFFFAOYSA-N n-propan-2-yl-3-pyridin-2-yl-1-tritylpyrazolo[4,3-c]pyridin-4-amine Chemical compound C1=2C(NC(C)C)=NC=CC=2N(C(C=2C=CC=CC=2)(C=2C=CC=CC=2)C=2C=CC=CC=2)N=C1C1=CC=CC=N1 SBJUHFSCCCRPAY-UHFFFAOYSA-N 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 description 1
- 210000003928 nasal cavity Anatomy 0.000 description 1
- 239000007923 nasal drop Substances 0.000 description 1
- 229940100662 nasal drops Drugs 0.000 description 1
- 239000006199 nebulizer Substances 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 235000001968 nicotinic acid Nutrition 0.000 description 1
- 239000011664 nicotinic acid Substances 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 239000012457 nonaqueous media Substances 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 231100000956 nontoxicity Toxicity 0.000 description 1
- 239000000346 nonvolatile oil Substances 0.000 description 1
- UMRZSTCPUPJPOJ-KNVOCYPGSA-N norbornane Chemical compound C1C[C@H]2CC[C@@H]1C2 UMRZSTCPUPJPOJ-KNVOCYPGSA-N 0.000 description 1
- 210000001331 nose Anatomy 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 150000002895 organic esters Chemical class 0.000 description 1
- AHVQYHFYQWKUKB-UHFFFAOYSA-N oxan-4-amine Chemical compound NC1CCOCC1 AHVQYHFYQWKUKB-UHFFFAOYSA-N 0.000 description 1
- GSEZHCLWHDZJAB-UHFFFAOYSA-N oxan-4-yl methanesulfonate Chemical compound CS(=O)(=O)OC1CCOCC1 GSEZHCLWHDZJAB-UHFFFAOYSA-N 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 229940023569 palmate Drugs 0.000 description 1
- 229940014662 pantothenate Drugs 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- JRKICGRDRMAZLK-UHFFFAOYSA-L peroxydisulfate Chemical compound [O-]S(=O)(=O)OOS([O-])(=O)=O JRKICGRDRMAZLK-UHFFFAOYSA-L 0.000 description 1
- 150000004968 peroxymonosulfuric acids Chemical class 0.000 description 1
- 229940124531 pharmaceutical excipient Drugs 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- RLOWWWKZYUNIDI-UHFFFAOYSA-N phosphinic chloride Chemical compound ClP=O RLOWWWKZYUNIDI-UHFFFAOYSA-N 0.000 description 1
- 150000003906 phosphoinositides Chemical class 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 125000004592 phthalazinyl group Chemical group C1(=NN=CC2=CC=CC=C12)* 0.000 description 1
- 229940075930 picrate Drugs 0.000 description 1
- OXNIZHLAWKMVMX-UHFFFAOYSA-M picrate anion Chemical compound [O-]C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O OXNIZHLAWKMVMX-UHFFFAOYSA-M 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229950010765 pivalate Drugs 0.000 description 1
- IUGYQRQAERSCNH-UHFFFAOYSA-N pivalic acid Chemical compound CC(C)(C)C(O)=O IUGYQRQAERSCNH-UHFFFAOYSA-N 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 229940069328 povidone Drugs 0.000 description 1
- 238000012746 preparative thin layer chromatography Methods 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- VVWRJUBEIPHGQF-UHFFFAOYSA-N propan-2-yl n-propan-2-yloxycarbonyliminocarbamate Chemical compound CC(C)OC(=O)N=NC(=O)OC(C)C VVWRJUBEIPHGQF-UHFFFAOYSA-N 0.000 description 1
- 239000001294 propane Substances 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 108010048484 radixin Proteins 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- AWUCVROLDVIAJX-GSVOUGTGSA-N sn-glycerol 3-phosphate Chemical compound OC[C@@H](O)COP(O)(O)=O AWUCVROLDVIAJX-GSVOUGTGSA-N 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 1
- 229940001584 sodium metabisulfite Drugs 0.000 description 1
- 235000010262 sodium metabisulphite Nutrition 0.000 description 1
- 239000012439 solid excipient Substances 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 239000012258 stirred mixture Substances 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 description 1
- DPKBAXPHAYBPRL-UHFFFAOYSA-M tetrabutylazanium;iodide Chemical compound [I-].CCCC[N+](CCCC)(CCCC)CCCC DPKBAXPHAYBPRL-UHFFFAOYSA-M 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- 231100001274 therapeutic index Toxicity 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 230000002110 toxicologic effect Effects 0.000 description 1
- 231100000759 toxicological effect Toxicity 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- CCRMAATUKBYMPA-UHFFFAOYSA-N trimethyltin Chemical compound C[Sn](C)C.C[Sn](C)C CCRMAATUKBYMPA-UHFFFAOYSA-N 0.000 description 1
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 1
- ZDPHROOEEOARMN-UHFFFAOYSA-N undecanoic acid Chemical compound CCCCCCCCCCC(O)=O ZDPHROOEEOARMN-UHFFFAOYSA-N 0.000 description 1
- 238000010200 validation analysis Methods 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 229940045860 white wax Drugs 0.000 description 1
- 239000009637 wintergreen oil Substances 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
- 229930195724 β-lactose Natural products 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/437—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a five-membered ring having nitrogen as a ring hetero atom, e.g. indolizine, beta-carboline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Psychology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB1008134.7 | 2010-05-14 | ||
| GBGB1008134.7A GB201008134D0 (en) | 2010-05-14 | 2010-05-14 | Compounds |
| PCT/GB2011/050937 WO2011141756A1 (en) | 2010-05-14 | 2011-05-16 | Pyrazolopyridines as inhibitors of the kinase lrrk2 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2013529196A true JP2013529196A (ja) | 2013-07-18 |
| JP2013529196A5 JP2013529196A5 (enExample) | 2014-07-03 |
Family
ID=42334821
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013509631A Withdrawn JP2013529196A (ja) | 2010-05-14 | 2011-05-16 | キナーゼlrrk2の阻害剤としてのピラゾロピリジン |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20130267513A1 (enExample) |
| EP (1) | EP2569293A1 (enExample) |
| JP (1) | JP2013529196A (enExample) |
| CN (1) | CN102971306A (enExample) |
| AU (1) | AU2011251733A1 (enExample) |
| CA (1) | CA2798222A1 (enExample) |
| GB (1) | GB201008134D0 (enExample) |
| WO (1) | WO2011141756A1 (enExample) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016516791A (ja) * | 2013-04-18 | 2016-06-09 | ハー・ルンドベック・アクチエゼルスカベット | Lrrk2阻害剤としてのアリールピロロピリジン誘導化合物 |
| JP2016540794A (ja) * | 2013-12-17 | 2016-12-28 | ファイザー・インク | LRRK2阻害薬としての新規の3,4−二置換−1H−ピロロ[2,3−b]ピリジンおよび4,5−二置換−7H−ピロロ[2,3−c]ピリダジン |
| JP2018524390A (ja) * | 2015-07-23 | 2018-08-30 | グラクソスミスクライン、インテレクチュアル、プロパティー、ディベロップメント、リミテッドGlaxosmithkline Intellectual Property Development Limited | 化合物 |
| KR20190067795A (ko) * | 2016-09-09 | 2019-06-17 | 인사이트 코포레이션 | Hpk1 조절제로서의 피라졸로피리딘 유도체 및 암을 치료하기 위한 이의 용도 |
| JP2020519667A (ja) * | 2017-05-15 | 2020-07-02 | ザ・リージェンツ・オブ・ザ・ユニバーシティ・オブ・ミシガンThe Regents Of The University Of Michigan | LSD−1インヒビターとしてのピロロ〔2,3−c〕ピリジン及び関連類似体 |
Families Citing this family (65)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8754114B2 (en) | 2010-12-22 | 2014-06-17 | Incyte Corporation | Substituted imidazopyridazines and benzimidazoles as inhibitors of FGFR3 |
| US8791112B2 (en) | 2011-03-30 | 2014-07-29 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1H-pyrazolo [3, 4-B] pyridine and pyrazolo [3, 4-B] pyridine derivatives as protein kinase inhibitors |
| GB201204985D0 (en) | 2012-03-21 | 2012-05-02 | Genentech Inc | Compounds |
| WO2013166276A1 (en) | 2012-05-02 | 2013-11-07 | Southern Research Institute | Triazolopyridazine compounds, use as inhibitors of the kinase lrrk2, and methods for preparation thereof |
| CN102675310B (zh) * | 2012-05-09 | 2014-12-24 | 林辉 | 制备吡唑并芳杂环化合物的方法 |
| RS58514B1 (sr) | 2012-06-13 | 2019-04-30 | Incyte Holdings Corp | Supstituisana triciklična jedinjenja kao inhibitori fgfr |
| EP2867236B1 (en) * | 2012-06-29 | 2017-06-14 | Pfizer Inc | Novel 4-(substituted-amino)-7h-pyrrolo[2,3-d]pyrimidines as lrrk2 inhibitors |
| US9388185B2 (en) | 2012-08-10 | 2016-07-12 | Incyte Holdings Corporation | Substituted pyrrolo[2,3-b]pyrazines as FGFR inhibitors |
| AU2013343105B2 (en) | 2012-11-08 | 2016-04-14 | Pfizer Inc. | Heteroaromatic compounds as dopamine D1 ligands |
| US9266892B2 (en) | 2012-12-19 | 2016-02-23 | Incyte Holdings Corporation | Fused pyrazoles as FGFR inhibitors |
| WO2014134774A1 (en) * | 2013-03-04 | 2014-09-12 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| WO2014137728A1 (en) | 2013-03-04 | 2014-09-12 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| WO2014134776A1 (en) | 2013-03-04 | 2014-09-12 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| WO2014134772A1 (en) | 2013-03-04 | 2014-09-12 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| CA2904462A1 (en) * | 2013-03-15 | 2014-09-18 | Ipsen Pharma S.A.S | Macrocyclic lrrk2 kinase inhibitors |
| US20140288043A1 (en) * | 2013-03-19 | 2014-09-25 | Genentech, Inc. | Pyrazolopyridine compounds |
| PH12015502383B1 (en) | 2013-04-19 | 2023-02-03 | Incyte Holdings Corp | Bicyclic heterocycles as fgfr inhibitors |
| US9718818B2 (en) | 2013-08-22 | 2017-08-01 | Merck Sharp & Dohme Corp. | Compounds inhibiting leucine-rich repeat kinase enzyme activity |
| CN103819396B (zh) * | 2014-02-26 | 2016-06-15 | 四川大学 | 一种手性的1-(3,5-二氯吡啶-4-基)-乙醇的合成方法 |
| EP3134405B1 (en) | 2014-04-25 | 2019-08-28 | Pfizer Inc | Heteroaromatic compounds and their use as dopamine d1 ligands |
| US10851105B2 (en) | 2014-10-22 | 2020-12-01 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| MA41551A (fr) | 2015-02-20 | 2017-12-26 | Incyte Corp | Hétérocycles bicycliques utilisés en tant qu'inhibiteurs de fgfr4 |
| TWI712601B (zh) | 2015-02-20 | 2020-12-11 | 美商英塞特公司 | 作為fgfr抑制劑之雙環雜環 |
| US9580423B2 (en) | 2015-02-20 | 2017-02-28 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| RU2722149C1 (ru) | 2015-09-14 | 2020-05-27 | Пфайзер Инк. | Новые производные имидазо[4,5-c] хинолинов и имидазо[4,5-c][1,5] нафтиридинов в качестве ингибиторов LRRK2 |
| US11214565B2 (en) | 2015-11-20 | 2022-01-04 | Denali Therapeutics Inc. | Compound, compositions, and methods |
| JP2018537502A (ja) | 2015-12-16 | 2018-12-20 | サウザーン リサーチ インスチチュート | ピロロピリミジン化合物、キナーゼlrrk2阻害剤としての使用、及びその調製方法 |
| US11028080B2 (en) | 2016-03-11 | 2021-06-08 | Denali Therapeutics Inc. | Substituted pyrimidines as LRKK2 inhibitors |
| SMT202100699T1 (it) | 2016-06-16 | 2022-01-10 | Denali Therapeutics Inc | Pirimidin-2-ilammino-1h-pirazoli come inibitori di lrrk2 per l'uso nel trattamento di disturbi neurodegenerativi |
| WO2018049214A1 (en) | 2016-09-09 | 2018-03-15 | Incyte Corporation | Pyrazolopyridine derivatives as hpk1 modulators and uses thereof for the treatment of cancer |
| US20180072741A1 (en) | 2016-09-09 | 2018-03-15 | Incyte Corporation | Pyrazolopyrimidine compounds and uses thereof |
| US10280164B2 (en) | 2016-09-09 | 2019-05-07 | Incyte Corporation | Pyrazolopyridone compounds and uses thereof |
| US10040211B2 (en) | 2016-12-09 | 2018-08-07 | Bettcher Industries, Inc. | Power operated rotary knife |
| WO2018152220A1 (en) | 2017-02-15 | 2018-08-23 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| KR101923852B1 (ko) * | 2017-02-24 | 2018-11-29 | 재단법인 대구경북첨단의료산업진흥재단 | 혈액 뇌관문을 통과할 수 있는 화합물을 유효성분으로 함유하는 뇌암의 예방 또는 치료용 약학적 조성물 |
| AU2018230236B2 (en) | 2017-03-10 | 2022-05-19 | Pfizer Inc. | Novel imidazo[4,5-c]quinoline derivatives as LRRK2 inhibitors |
| WO2018163030A1 (en) | 2017-03-10 | 2018-09-13 | Pfizer Inc. | Cyclic substituted imidazo[4,5-c]quinoline derivatives |
| AR111960A1 (es) | 2017-05-26 | 2019-09-04 | Incyte Corp | Formas cristalinas de un inhibidor de fgfr y procesos para su preparación |
| US10722495B2 (en) | 2017-09-08 | 2020-07-28 | Incyte Corporation | Cyanoindazole compounds and uses thereof |
| US10752635B2 (en) | 2018-02-20 | 2020-08-25 | Incyte Corporation | Indazole compounds and uses thereof |
| EP3755703B1 (en) | 2018-02-20 | 2022-05-04 | Incyte Corporation | N-(phenyl)-2-(phenyl)pyrimidine-4-carboxamide derivatives and related compounds as hpk1 inhibitors for treating cancer |
| US10745388B2 (en) | 2018-02-20 | 2020-08-18 | Incyte Corporation | Indazole compounds and uses thereof |
| US11299473B2 (en) | 2018-04-13 | 2022-04-12 | Incyte Corporation | Benzimidazole and indole compounds and uses thereof |
| AU2019262579B2 (en) | 2018-05-04 | 2024-09-12 | Incyte Corporation | Salts of an FGFR inhibitor |
| HRP20241288T1 (hr) | 2018-05-04 | 2024-12-06 | Incyte Corporation | Čvrsti oblici fgfr inhibitora i postupci za njihovu proizvodnju |
| ES2738911A1 (es) | 2018-07-25 | 2020-01-27 | Consejo Superior Investigacion | Procedimiento para depositar elementos sobre un sustrato de interes y dispositivo |
| US10899755B2 (en) | 2018-08-08 | 2021-01-26 | Incyte Corporation | Benzothiazole compounds and uses thereof |
| ES2744304B2 (es) | 2018-08-24 | 2020-06-22 | Consejo Superior Investigacion | Compuestos inhibidores de LRRK2 y su uso para el tratamiento de enfermedades neurodegenerativas |
| WO2020068729A1 (en) | 2018-09-25 | 2020-04-02 | Incyte Corporation | Pyrazolo[4,3-d]pyrimidine compounds as alk2 abd/or fgfr modulators |
| US11628162B2 (en) | 2019-03-08 | 2023-04-18 | Incyte Corporation | Methods of treating cancer with an FGFR inhibitor |
| US11591329B2 (en) | 2019-07-09 | 2023-02-28 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| BR112022002059A2 (pt) | 2019-08-06 | 2022-06-07 | Incyte Corp | Formas sólidas de um inibidor de hpk1 |
| WO2021067374A1 (en) | 2019-10-01 | 2021-04-08 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| CA3157361A1 (en) | 2019-10-14 | 2021-04-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| WO2021076728A1 (en) | 2019-10-16 | 2021-04-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| JP7832891B2 (ja) | 2019-12-04 | 2026-03-18 | インサイト・コーポレイション | Fgfr阻害剤の誘導体 |
| CA3163875A1 (en) | 2019-12-04 | 2021-06-10 | Incyte Corporation | Tricyclic heterocycles as fgfr inhibitors |
| US12012409B2 (en) | 2020-01-15 | 2024-06-18 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| JP2024512501A (ja) | 2021-03-19 | 2024-03-19 | アルヴィナス・オペレーションズ・インコーポレイテッド | インダゾール系化合物及び関連使用方法 |
| WO2022221170A1 (en) | 2021-04-12 | 2022-10-20 | Incyte Corporation | Combination therapy comprising an fgfr inhibitor and a nectin-4 targeting agent |
| CA3220155A1 (en) | 2021-06-09 | 2022-12-15 | Incyte Corporation | Tricyclic heterocycles as fgfr inhibitors |
| US11939331B2 (en) | 2021-06-09 | 2024-03-26 | Incyte Corporation | Tricyclic heterocycles as FGFR inhibitors |
| WO2023076404A1 (en) | 2021-10-27 | 2023-05-04 | Aria Pharmaceuticals, Inc. | Methods for treating systemic lupus erythematosus |
| TW202515874A (zh) | 2023-09-28 | 2025-04-16 | 美商亞文納營運公司 | 用於靶向降解富含白胺酸之重複激酶2(lrrk2)的基於苯并咪唑及苯并咪唑酮之protac化合物 |
| WO2025248032A1 (en) | 2024-05-31 | 2025-12-04 | Syngenta Crop Protection Ag | Pesticidally active indazole compounds |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NZ207394A (en) | 1983-03-08 | 1987-03-06 | Commw Serum Lab Commission | Detecting or determining sequence of amino acids |
| GB9216783D0 (en) * | 1992-08-07 | 1992-09-23 | Ici Plc | Heterocyclic derivatives |
| US5300478A (en) * | 1993-01-28 | 1994-04-05 | Zeneca Limited | Substituted fused pyrazolo compounds |
| EP1396488A1 (en) * | 2001-05-23 | 2004-03-10 | Mitsubishi Pharma Corporation | Fused heterocyclic compound and medicinal use thereof |
| PE20061119A1 (es) * | 2005-01-19 | 2006-11-27 | Aventis Pharma Sa | PIRAZOLO PIRIDINAS SUSTITUIDAS COMO INHIBIDORES DE CINASAS FAK, KDR Y Tie |
| EP1996588A4 (en) * | 2006-03-03 | 2011-10-05 | Torrent Pharmaceuticals Ltd | NEW DUAL ACTION RECEPTOR (DARA) ANTAGONISTS OF ATI AND ETA RECEPTORS |
| US20100273776A1 (en) * | 2006-03-29 | 2010-10-28 | FOLDRx PHARMACEUTICALS, INC | Inhibition of alpha-synuclein toxicity |
| EP1932845A1 (en) * | 2006-12-15 | 2008-06-18 | Bayer Schering Pharma Aktiengesellschaft | 3-H-pyrazolopyridines and salts thereof, pharmaceutical compositions comprising same, methods of preparing same and uses of same |
| WO2009030270A1 (en) * | 2007-09-03 | 2009-03-12 | Novartis Ag | Dihydroindole derivatives useful in parkinson's disease |
| EP2408772B1 (en) * | 2009-03-19 | 2015-07-01 | Medical Research Council Technology | Compounds |
| NZ598220A (en) * | 2009-08-17 | 2014-02-28 | Intellikine Llc | Heterocyclic compounds and uses thereof |
-
2010
- 2010-05-14 GB GBGB1008134.7A patent/GB201008134D0/en not_active Ceased
-
2011
- 2011-05-16 EP EP11720847A patent/EP2569293A1/en not_active Withdrawn
- 2011-05-16 CN CN201180033483XA patent/CN102971306A/zh active Pending
- 2011-05-16 JP JP2013509631A patent/JP2013529196A/ja not_active Withdrawn
- 2011-05-16 US US13/697,878 patent/US20130267513A1/en not_active Abandoned
- 2011-05-16 AU AU2011251733A patent/AU2011251733A1/en not_active Abandoned
- 2011-05-16 WO PCT/GB2011/050937 patent/WO2011141756A1/en not_active Ceased
- 2011-05-16 CA CA2798222A patent/CA2798222A1/en not_active Abandoned
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016516791A (ja) * | 2013-04-18 | 2016-06-09 | ハー・ルンドベック・アクチエゼルスカベット | Lrrk2阻害剤としてのアリールピロロピリジン誘導化合物 |
| JP2016540794A (ja) * | 2013-12-17 | 2016-12-28 | ファイザー・インク | LRRK2阻害薬としての新規の3,4−二置換−1H−ピロロ[2,3−b]ピリジンおよび4,5−二置換−7H−ピロロ[2,3−c]ピリダジン |
| JP2018524390A (ja) * | 2015-07-23 | 2018-08-30 | グラクソスミスクライン、インテレクチュアル、プロパティー、ディベロップメント、リミテッドGlaxosmithkline Intellectual Property Development Limited | 化合物 |
| KR20190067795A (ko) * | 2016-09-09 | 2019-06-17 | 인사이트 코포레이션 | Hpk1 조절제로서의 피라졸로피리딘 유도체 및 암을 치료하기 위한 이의 용도 |
| JP2019530670A (ja) * | 2016-09-09 | 2019-10-24 | インサイト・コーポレイションIncyte Corporation | Hpk1調節薬としてのピラゾロピリジン誘導体及びがんの治療のためのその用法 |
| JP7076432B2 (ja) | 2016-09-09 | 2022-05-27 | インサイト・コーポレイション | Hpk1調節薬としてのピラゾロピリジン誘導体及びがんの治療のためのその用法 |
| KR102507967B1 (ko) | 2016-09-09 | 2023-03-09 | 인사이트 코포레이션 | Hpk1 조절제로서의 피라졸로피리딘 유도체 및 암을 치료하기 위한 이의 용도 |
| JP2020519667A (ja) * | 2017-05-15 | 2020-07-02 | ザ・リージェンツ・オブ・ザ・ユニバーシティ・オブ・ミシガンThe Regents Of The University Of Michigan | LSD−1インヒビターとしてのピロロ〔2,3−c〕ピリジン及び関連類似体 |
| JP7352284B2 (ja) | 2017-05-15 | 2023-09-28 | ザ・リージェンツ・オブ・ザ・ユニバーシティ・オブ・ミシガン | LSD-1インヒビターとしてのピロロ〔2,3-c〕ピリジン及び関連類似体 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102971306A (zh) | 2013-03-13 |
| CA2798222A1 (en) | 2011-11-17 |
| WO2011141756A1 (en) | 2011-11-17 |
| AU2011251733A1 (en) | 2012-12-20 |
| US20130267513A1 (en) | 2013-10-10 |
| GB201008134D0 (en) | 2010-06-30 |
| EP2569293A1 (en) | 2013-03-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2013529196A (ja) | キナーゼlrrk2の阻害剤としてのピラゾロピリジン | |
| JP5732036B2 (ja) | 化合物 | |
| US8569281B2 (en) | Compounds and their administration for treating a neurodegenerative disease as well as a method for identifying a compound capable of inhibiting a kinase, such as LRRK | |
| RU2745035C1 (ru) | Ингибитор fgfr и его применение | |
| JP6175484B2 (ja) | 新規Syk阻害剤としての置換ピリドピラジン | |
| JP6096879B2 (ja) | 二環式ピラジノン誘導体 | |
| US9550796B2 (en) | Pyrrolopyrrolone derivatives and their use as BET inhibitors | |
| US7723347B2 (en) | Substituted phenylamino-pyrimidines | |
| KR20250004854A (ko) | 피리미딘 화합물, 이의 제조 방법 및 이의 약학적 용도 | |
| WO2013139882A1 (en) | Pyrazolopyridines for treatment of parkinsons disease | |
| JP7247092B2 (ja) | キナーゼ阻害剤としての置換された縮合ヘテロアリール化合物及びその用途 | |
| MX2010014005A (es) | Compuestos de triazolopiridina inhibidores de jak y los metodos. | |
| JP6283688B2 (ja) | カゼインキナーゼ1d/e阻害剤としての新規なピラゾール置換のイミダゾピラジン | |
| RS59921B1 (sr) | (aza-)izohinolinon derivati | |
| JP2016520118A (ja) | Bet阻害剤としてのピラゾロ−ピロリジン−4−オン誘導体および疾患の処置におけるその使用 | |
| JP6885962B2 (ja) | 1,4−ジカルボニル−ピペリジル誘導体 | |
| JP2021518379A (ja) | Jak阻害剤 | |
| AU2016355103B2 (en) | Pyrropyrimidine compounds as MNKs inhibitors | |
| JP6847942B2 (ja) | 二環式複素環式誘導体 | |
| US20140288043A1 (en) | Pyrazolopyridine compounds | |
| CN119306726A (zh) | Pkmyt1抑制剂及其应用 | |
| HK1190143B (en) | Substituted pyridopyrazines as novel syk inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140514 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20140514 |
|
| A761 | Written withdrawal of application |
Free format text: JAPANESE INTERMEDIATE CODE: A761 Effective date: 20140709 |