JP2013515731A - フューリンノックダウン及びgm−csf増強(fang)癌ワクチン - Google Patents
フューリンノックダウン及びgm−csf増強(fang)癌ワクチン Download PDFInfo
- Publication number
- JP2013515731A JP2013515731A JP2012546121A JP2012546121A JP2013515731A JP 2013515731 A JP2013515731 A JP 2013515731A JP 2012546121 A JP2012546121 A JP 2012546121A JP 2012546121 A JP2012546121 A JP 2012546121A JP 2013515731 A JP2013515731 A JP 2013515731A
- Authority
- JP
- Japan
- Prior art keywords
- furin
- cancer
- tgf
- cell
- cells
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 102000004961 Furin Human genes 0.000 title claims abstract description 221
- 108090001126 Furin Proteins 0.000 title claims abstract description 221
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 title claims abstract description 128
- 229940022399 cancer vaccine Drugs 0.000 title claims abstract description 21
- 238000009566 cancer vaccine Methods 0.000 title claims abstract description 21
- 102000004457 Granulocyte-Macrophage Colony-Stimulating Factor Human genes 0.000 title abstract 3
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 178
- 102000004887 Transforming Growth Factor beta Human genes 0.000 claims abstract description 171
- 108090001012 Transforming Growth Factor beta Proteins 0.000 claims abstract description 171
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 claims abstract description 156
- 230000014509 gene expression Effects 0.000 claims abstract description 137
- 108091027967 Small hairpin RNA Proteins 0.000 claims abstract description 121
- 201000011510 cancer Diseases 0.000 claims abstract description 107
- 238000000034 method Methods 0.000 claims abstract description 96
- 229960005486 vaccine Drugs 0.000 claims abstract description 91
- 239000004055 small Interfering RNA Substances 0.000 claims abstract description 86
- 230000001588 bifunctional effect Effects 0.000 claims abstract description 41
- 239000000203 mixture Substances 0.000 claims abstract description 30
- 238000011282 treatment Methods 0.000 claims abstract description 25
- 230000001506 immunosuppresive effect Effects 0.000 claims abstract description 18
- 238000012545 processing Methods 0.000 claims abstract description 14
- 210000004027 cell Anatomy 0.000 claims description 202
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 claims description 166
- 108020004999 messenger RNA Proteins 0.000 claims description 68
- 238000003776 cleavage reaction Methods 0.000 claims description 54
- 230000007017 scission Effects 0.000 claims description 54
- 102000046299 Transforming Growth Factor beta1 Human genes 0.000 claims description 53
- 101800002279 Transforming growth factor beta-1 Proteins 0.000 claims description 53
- 102000039446 nucleic acids Human genes 0.000 claims description 44
- 108020004707 nucleic acids Proteins 0.000 claims description 44
- 150000007523 nucleic acids Chemical class 0.000 claims description 44
- 101000746373 Homo sapiens Granulocyte-macrophage colony-stimulating factor Proteins 0.000 claims description 43
- 108020004459 Small interfering RNA Proteins 0.000 claims description 39
- 102000011117 Transforming Growth Factor beta2 Human genes 0.000 claims description 37
- 101800000304 Transforming growth factor beta-2 Proteins 0.000 claims description 36
- 208000002154 non-small cell lung carcinoma Diseases 0.000 claims description 35
- 230000009368 gene silencing by RNA Effects 0.000 claims description 34
- 239000013612 plasmid Substances 0.000 claims description 33
- 238000001890 transfection Methods 0.000 claims description 33
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 claims description 33
- 238000012228 RNA interference-mediated gene silencing Methods 0.000 claims description 31
- 230000002401 inhibitory effect Effects 0.000 claims description 30
- 239000013604 expression vector Substances 0.000 claims description 26
- 230000001419 dependent effect Effects 0.000 claims description 25
- 230000001404 mediated effect Effects 0.000 claims description 22
- 201000001441 melanoma Diseases 0.000 claims description 22
- 229940030156 cell vaccine Drugs 0.000 claims description 18
- 230000000735 allogeneic effect Effects 0.000 claims description 16
- 238000002560 therapeutic procedure Methods 0.000 claims description 15
- 108020005345 3' Untranslated Regions Proteins 0.000 claims description 14
- 230000000295 complement effect Effects 0.000 claims description 14
- 239000002299 complementary DNA Substances 0.000 claims description 14
- 201000007270 liver cancer Diseases 0.000 claims description 14
- 208000014018 liver neoplasm Diseases 0.000 claims description 14
- 108091070501 miRNA Proteins 0.000 claims description 14
- 239000002679 microRNA Substances 0.000 claims description 14
- 108091032973 (ribonucleotides)n+m Proteins 0.000 claims description 13
- 230000007246 mechanism Effects 0.000 claims description 13
- 208000024891 symptom Diseases 0.000 claims description 12
- 108091026890 Coding region Proteins 0.000 claims description 11
- 206010033128 Ovarian cancer Diseases 0.000 claims description 11
- 238000002648 combination therapy Methods 0.000 claims description 11
- 230000012010 growth Effects 0.000 claims description 11
- 238000002347 injection Methods 0.000 claims description 11
- 239000007924 injection Substances 0.000 claims description 11
- 206010006187 Breast cancer Diseases 0.000 claims description 10
- 208000026310 Breast neoplasm Diseases 0.000 claims description 10
- 238000004520 electroporation Methods 0.000 claims description 10
- 206010009944 Colon cancer Diseases 0.000 claims description 9
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 9
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 9
- 208000001333 Colorectal Neoplasms Diseases 0.000 claims description 8
- 101100508818 Mus musculus Inpp5k gene Proteins 0.000 claims description 8
- 101100366438 Rattus norvegicus Sphkap gene Proteins 0.000 claims description 8
- 230000002265 prevention Effects 0.000 claims description 8
- 239000012646 vaccine adjuvant Substances 0.000 claims description 8
- 229940124931 vaccine adjuvant Drugs 0.000 claims description 8
- 208000006168 Ewing Sarcoma Diseases 0.000 claims description 7
- 208000022072 Gallbladder Neoplasms Diseases 0.000 claims description 7
- 206010027476 Metastases Diseases 0.000 claims description 7
- 239000006285 cell suspension Substances 0.000 claims description 7
- 239000003814 drug Substances 0.000 claims description 7
- 201000010175 gallbladder cancer Diseases 0.000 claims description 7
- 239000003112 inhibitor Substances 0.000 claims description 7
- 230000009401 metastasis Effects 0.000 claims description 7
- 230000009919 sequestration Effects 0.000 claims description 7
- 241000709664 Picornaviridae Species 0.000 claims description 6
- 102000056172 Transforming growth factor beta-3 Human genes 0.000 claims description 6
- 210000005260 human cell Anatomy 0.000 claims description 6
- 230000002062 proliferating effect Effects 0.000 claims description 6
- 229940124597 therapeutic agent Drugs 0.000 claims description 6
- 108090000097 Transforming growth factor beta-3 Proteins 0.000 claims description 5
- 101150084233 ago2 gene Proteins 0.000 claims description 5
- 239000013600 plasmid vector Substances 0.000 claims description 5
- 108700028146 Genetic Enhancer Elements Proteins 0.000 claims description 4
- 230000008014 freezing Effects 0.000 claims description 4
- 238000007710 freezing Methods 0.000 claims description 4
- 238000012544 monitoring process Methods 0.000 claims description 4
- 108020003589 5' Untranslated Regions Proteins 0.000 claims description 3
- 101500025614 Homo sapiens Transforming growth factor beta-1 Proteins 0.000 claims description 3
- 101500025624 Homo sapiens Transforming growth factor beta-2 Proteins 0.000 claims description 3
- 239000002671 adjuvant Substances 0.000 claims description 3
- 230000002708 enhancing effect Effects 0.000 claims description 3
- 229940065638 intron a Drugs 0.000 claims description 3
- 230000009467 reduction Effects 0.000 claims description 3
- 239000012984 antibiotic solution Substances 0.000 claims description 2
- 230000002255 enzymatic effect Effects 0.000 claims description 2
- 230000001678 irradiating effect Effects 0.000 claims description 2
- 239000000243 solution Substances 0.000 claims description 2
- 101500026551 Homo sapiens Transforming growth factor beta-3 Proteins 0.000 claims 1
- 239000002253 acid Substances 0.000 claims 1
- 230000005855 radiation Effects 0.000 claims 1
- 239000000427 antigen Substances 0.000 abstract description 22
- 108091007433 antigens Proteins 0.000 abstract description 22
- 102000036639 antigens Human genes 0.000 abstract description 22
- 108700019146 Transgenes Proteins 0.000 abstract description 8
- 238000003197 gene knockdown Methods 0.000 description 48
- 230000000694 effects Effects 0.000 description 36
- 238000004519 manufacturing process Methods 0.000 description 35
- 210000004881 tumor cell Anatomy 0.000 description 33
- 108090000623 proteins and genes Proteins 0.000 description 31
- 102000000574 RNA-Induced Silencing Complex Human genes 0.000 description 28
- 108010016790 RNA-Induced Silencing Complex Proteins 0.000 description 28
- 210000004443 dendritic cell Anatomy 0.000 description 21
- 102000004169 proteins and genes Human genes 0.000 description 20
- 230000006870 function Effects 0.000 description 19
- 108010029485 Protein Isoforms Proteins 0.000 description 18
- 102000001708 Protein Isoforms Human genes 0.000 description 18
- 210000001744 T-lymphocyte Anatomy 0.000 description 18
- 230000005764 inhibitory process Effects 0.000 description 17
- 102000004127 Cytokines Human genes 0.000 description 15
- 108090000695 Cytokines Proteins 0.000 description 15
- 108020004414 DNA Proteins 0.000 description 15
- 238000001994 activation Methods 0.000 description 14
- 230000000692 anti-sense effect Effects 0.000 description 13
- 230000004083 survival effect Effects 0.000 description 13
- 102100024193 Mitogen-activated protein kinase 1 Human genes 0.000 description 12
- 102000003993 Phosphatidylinositol 3-kinases Human genes 0.000 description 12
- 108090000430 Phosphatidylinositol 3-kinases Proteins 0.000 description 12
- 230000006058 immune tolerance Effects 0.000 description 12
- 239000000523 sample Substances 0.000 description 12
- 239000013598 vector Substances 0.000 description 12
- 230000004913 activation Effects 0.000 description 11
- 210000002540 macrophage Anatomy 0.000 description 11
- ZOOGRGPOEVQQDX-UUOKFMHZSA-N 3',5'-cyclic GMP Chemical compound C([C@H]1O2)OP(O)(=O)O[C@H]1[C@@H](O)[C@@H]2N1C(N=C(NC2=O)N)=C2N=C1 ZOOGRGPOEVQQDX-UUOKFMHZSA-N 0.000 description 10
- 102000004190 Enzymes Human genes 0.000 description 10
- 108090000790 Enzymes Proteins 0.000 description 10
- 108091007960 PI3Ks Proteins 0.000 description 10
- 238000013459 approach Methods 0.000 description 10
- 210000003810 lymphokine-activated killer cell Anatomy 0.000 description 10
- 230000037361 pathway Effects 0.000 description 10
- 230000008569 process Effects 0.000 description 10
- 230000001105 regulatory effect Effects 0.000 description 10
- 208000005017 glioblastoma Diseases 0.000 description 9
- 230000035800 maturation Effects 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 108010007457 Extracellular Signal-Regulated MAP Kinases Proteins 0.000 description 8
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 8
- 238000003556 assay Methods 0.000 description 8
- 230000001965 increasing effect Effects 0.000 description 8
- 201000005202 lung cancer Diseases 0.000 description 8
- 208000020816 lung neoplasm Diseases 0.000 description 8
- 230000002093 peripheral effect Effects 0.000 description 8
- 239000002243 precursor Substances 0.000 description 8
- 230000002829 reductive effect Effects 0.000 description 8
- 206010062016 Immunosuppression Diseases 0.000 description 7
- 108010074328 Interferon-gamma Proteins 0.000 description 7
- 210000000612 antigen-presenting cell Anatomy 0.000 description 7
- 230000007812 deficiency Effects 0.000 description 7
- 230000002950 deficient Effects 0.000 description 7
- 230000003053 immunization Effects 0.000 description 7
- 238000002649 immunization Methods 0.000 description 7
- 230000026731 phosphorylation Effects 0.000 description 7
- 238000006366 phosphorylation reaction Methods 0.000 description 7
- 210000003289 regulatory T cell Anatomy 0.000 description 7
- 230000004044 response Effects 0.000 description 7
- 230000014616 translation Effects 0.000 description 7
- 238000002255 vaccination Methods 0.000 description 7
- 206010018338 Glioma Diseases 0.000 description 6
- 102000008070 Interferon-gamma Human genes 0.000 description 6
- 206010061309 Neoplasm progression Diseases 0.000 description 6
- 230000003915 cell function Effects 0.000 description 6
- 230000000139 costimulatory effect Effects 0.000 description 6
- 230000007423 decrease Effects 0.000 description 6
- 238000011161 development Methods 0.000 description 6
- 230000018109 developmental process Effects 0.000 description 6
- 201000010099 disease Diseases 0.000 description 6
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 6
- 239000002158 endotoxin Substances 0.000 description 6
- 238000000684 flow cytometry Methods 0.000 description 6
- 229920006008 lipopolysaccharide Polymers 0.000 description 6
- 238000012423 maintenance Methods 0.000 description 6
- 108091043187 miR-30a stem-loop Proteins 0.000 description 6
- 230000004048 modification Effects 0.000 description 6
- 238000012986 modification Methods 0.000 description 6
- 208000025113 myeloid leukemia Diseases 0.000 description 6
- 210000000822 natural killer cell Anatomy 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- 238000013518 transcription Methods 0.000 description 6
- 230000035897 transcription Effects 0.000 description 6
- 230000005751 tumor progression Effects 0.000 description 6
- 238000001262 western blot Methods 0.000 description 6
- 238000002965 ELISA Methods 0.000 description 5
- 238000004458 analytical method Methods 0.000 description 5
- 210000001185 bone marrow Anatomy 0.000 description 5
- 230000003828 downregulation Effects 0.000 description 5
- 230000028993 immune response Effects 0.000 description 5
- 238000000338 in vitro Methods 0.000 description 5
- 238000001727 in vivo Methods 0.000 description 5
- 239000003446 ligand Substances 0.000 description 5
- 239000002502 liposome Substances 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 230000028327 secretion Effects 0.000 description 5
- 210000001519 tissue Anatomy 0.000 description 5
- 229940099456 transforming growth factor beta 1 Drugs 0.000 description 5
- 102000015696 Interleukins Human genes 0.000 description 4
- 108010063738 Interleukins Proteins 0.000 description 4
- 102000043131 MHC class II family Human genes 0.000 description 4
- 108091054438 MHC class II family Proteins 0.000 description 4
- 241000699666 Mus <mouse, genus> Species 0.000 description 4
- 102000048850 Neoplasm Genes Human genes 0.000 description 4
- 108700019961 Neoplasm Genes Proteins 0.000 description 4
- 108700026244 Open Reading Frames Proteins 0.000 description 4
- 102000006437 Proprotein Convertases Human genes 0.000 description 4
- 108010044159 Proprotein Convertases Proteins 0.000 description 4
- 108010009583 Transforming Growth Factors Proteins 0.000 description 4
- 230000009471 action Effects 0.000 description 4
- 230000001464 adherent effect Effects 0.000 description 4
- 230000005809 anti-tumor immunity Effects 0.000 description 4
- 230000030741 antigen processing and presentation Effects 0.000 description 4
- 229940037642 autologous vaccine Drugs 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 230000004071 biological effect Effects 0.000 description 4
- 230000000903 blocking effect Effects 0.000 description 4
- 238000012512 characterization method Methods 0.000 description 4
- 239000012228 culture supernatant Substances 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 230000006698 induction Effects 0.000 description 4
- 229960003130 interferon gamma Drugs 0.000 description 4
- 230000036210 malignancy Effects 0.000 description 4
- 230000002018 overexpression Effects 0.000 description 4
- 238000010837 poor prognosis Methods 0.000 description 4
- 230000019491 signal transduction Effects 0.000 description 4
- 238000012385 systemic delivery Methods 0.000 description 4
- 230000008685 targeting Effects 0.000 description 4
- 102000008203 CTLA-4 Antigen Human genes 0.000 description 3
- 108010021064 CTLA-4 Antigen Proteins 0.000 description 3
- 229940045513 CTLA4 antagonist Drugs 0.000 description 3
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 3
- 229940032072 GVAX vaccine Drugs 0.000 description 3
- 208000032612 Glial tumor Diseases 0.000 description 3
- 102000014158 Interleukin-12 Subunit p40 Human genes 0.000 description 3
- 108010011429 Interleukin-12 Subunit p40 Proteins 0.000 description 3
- 108091034117 Oligonucleotide Proteins 0.000 description 3
- 102000035195 Peptidases Human genes 0.000 description 3
- 108091005804 Peptidases Proteins 0.000 description 3
- 101710118538 Protease Proteins 0.000 description 3
- 239000004365 Protease Substances 0.000 description 3
- 102000001253 Protein Kinase Human genes 0.000 description 3
- 238000011529 RT qPCR Methods 0.000 description 3
- 206010070834 Sensitisation Diseases 0.000 description 3
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 3
- 102000043168 TGF-beta family Human genes 0.000 description 3
- 108091085018 TGF-beta family Proteins 0.000 description 3
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 3
- 230000004075 alteration Effects 0.000 description 3
- 230000003042 antagnostic effect Effects 0.000 description 3
- 229940030547 autologous tumor cell vaccine Drugs 0.000 description 3
- 230000002457 bidirectional effect Effects 0.000 description 3
- 230000033228 biological regulation Effects 0.000 description 3
- 229910052791 calcium Inorganic materials 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 230000024245 cell differentiation Effects 0.000 description 3
- 230000010261 cell growth Effects 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 238000005520 cutting process Methods 0.000 description 3
- 230000016396 cytokine production Effects 0.000 description 3
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 3
- NHBJTTGFHCHQHS-VZTVMPNDSA-N decanoyl-L-Arg-L-Val-L-Lys-L-Arg-chloromethylketone Chemical compound CCCCCCCCCC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)CCl NHBJTTGFHCHQHS-VZTVMPNDSA-N 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 230000004041 dendritic cell maturation Effects 0.000 description 3
- 238000013461 design Methods 0.000 description 3
- 230000004069 differentiation Effects 0.000 description 3
- 239000012636 effector Substances 0.000 description 3
- 210000002889 endothelial cell Anatomy 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 238000001415 gene therapy Methods 0.000 description 3
- 239000003102 growth factor Substances 0.000 description 3
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 3
- 230000005934 immune activation Effects 0.000 description 3
- 230000004957 immunoregulator effect Effects 0.000 description 3
- 238000009169 immunotherapy Methods 0.000 description 3
- 230000001771 impaired effect Effects 0.000 description 3
- 210000001616 monocyte Anatomy 0.000 description 3
- 238000011275 oncology therapy Methods 0.000 description 3
- 108010068338 p38 Mitogen-Activated Protein Kinases Proteins 0.000 description 3
- 102000002574 p38 Mitogen-Activated Protein Kinases Human genes 0.000 description 3
- 230000003389 potentiating effect Effects 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 102000004196 processed proteins & peptides Human genes 0.000 description 3
- 108060006633 protein kinase Proteins 0.000 description 3
- 238000003753 real-time PCR Methods 0.000 description 3
- 230000008313 sensitization Effects 0.000 description 3
- 230000011664 signaling Effects 0.000 description 3
- 210000000130 stem cell Anatomy 0.000 description 3
- 230000001629 suppression Effects 0.000 description 3
- 229940072041 transforming growth factor beta 2 Drugs 0.000 description 3
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 2
- 102100026802 72 kDa type IV collagenase Human genes 0.000 description 2
- 101710151806 72 kDa type IV collagenase Proteins 0.000 description 2
- OMLWNBVRVJYMBQ-YUMQZZPRSA-N Arg-Arg Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O OMLWNBVRVJYMBQ-YUMQZZPRSA-N 0.000 description 2
- 201000009030 Carcinoma Diseases 0.000 description 2
- 108020004705 Codon Proteins 0.000 description 2
- 108091035707 Consensus sequence Proteins 0.000 description 2
- 241000766026 Coregonus nasus Species 0.000 description 2
- 102000010911 Enzyme Precursors Human genes 0.000 description 2
- 108010062466 Enzyme Precursors Proteins 0.000 description 2
- 208000001382 Experimental Melanoma Diseases 0.000 description 2
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 2
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 2
- 101710165567 Extracellular signal-regulated kinase 1 Proteins 0.000 description 2
- 108091027874 Group I catalytic intron Proteins 0.000 description 2
- 101000853009 Homo sapiens Interleukin-24 Proteins 0.000 description 2
- 101001098833 Homo sapiens Proprotein convertase subtilisin/kexin type 6 Proteins 0.000 description 2
- 101000831940 Homo sapiens Stathmin Proteins 0.000 description 2
- 102000014150 Interferons Human genes 0.000 description 2
- 108010050904 Interferons Proteins 0.000 description 2
- 102100036671 Interleukin-24 Human genes 0.000 description 2
- 108090000978 Interleukin-4 Proteins 0.000 description 2
- 102000019149 MAP kinase activity proteins Human genes 0.000 description 2
- 108040008097 MAP kinase activity proteins Proteins 0.000 description 2
- 101710166115 Mitogen-activated protein kinase 2 Proteins 0.000 description 2
- 101710166076 Mitogen-activated protein kinase 5 Proteins 0.000 description 2
- 241001529936 Murinae Species 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- 241001144416 Picornavirales Species 0.000 description 2
- 102100038946 Proprotein convertase subtilisin/kexin type 6 Human genes 0.000 description 2
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 2
- 102000005465 Stathmin Human genes 0.000 description 2
- 108050003387 Stathmin Proteins 0.000 description 2
- 102100024237 Stathmin Human genes 0.000 description 2
- 108091036066 Three prime untranslated region Proteins 0.000 description 2
- 102000009618 Transforming Growth Factors Human genes 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 108060008683 Tumor Necrosis Factor Receptor Proteins 0.000 description 2
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 2
- 108010000134 Vascular Cell Adhesion Molecule-1 Proteins 0.000 description 2
- 102100023543 Vascular cell adhesion protein 1 Human genes 0.000 description 2
- 238000009825 accumulation Methods 0.000 description 2
- 230000033289 adaptive immune response Effects 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- VREFGVBLTWBCJP-UHFFFAOYSA-N alprazolam Chemical compound C12=CC(Cl)=CC=C2N2C(C)=NN=C2CN=C1C1=CC=CC=C1 VREFGVBLTWBCJP-UHFFFAOYSA-N 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 230000001093 anti-cancer Effects 0.000 description 2
- 230000006023 anti-tumor response Effects 0.000 description 2
- 108010068380 arginylarginine Proteins 0.000 description 2
- 210000003567 ascitic fluid Anatomy 0.000 description 2
- 230000003305 autocrine Effects 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- 230000000975 bioactive effect Effects 0.000 description 2
- 238000010364 biochemical engineering Methods 0.000 description 2
- 230000008827 biological function Effects 0.000 description 2
- 238000001574 biopsy Methods 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 230000000981 bystander Effects 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 238000004113 cell culture Methods 0.000 description 2
- 230000004709 cell invasion Effects 0.000 description 2
- 239000013592 cell lysate Substances 0.000 description 2
- 230000004663 cell proliferation Effects 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 230000000875 corresponding effect Effects 0.000 description 2
- 230000005574 cross-species transmission Effects 0.000 description 2
- 208000030381 cutaneous melanoma Diseases 0.000 description 2
- 230000001461 cytolytic effect Effects 0.000 description 2
- 231100000433 cytotoxic Toxicity 0.000 description 2
- 230000001472 cytotoxic effect Effects 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 230000009849 deactivation Effects 0.000 description 2
- 238000005034 decoration Methods 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000012217 deletion Methods 0.000 description 2
- 230000037430 deletion Effects 0.000 description 2
- 230000000779 depleting effect Effects 0.000 description 2
- 230000000994 depressogenic effect Effects 0.000 description 2
- 210000003162 effector t lymphocyte Anatomy 0.000 description 2
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 2
- 210000002744 extracellular matrix Anatomy 0.000 description 2
- 230000009123 feedback regulation Effects 0.000 description 2
- 208000029824 high grade glioma Diseases 0.000 description 2
- 238000003018 immunoassay Methods 0.000 description 2
- 230000005847 immunogenicity Effects 0.000 description 2
- 229940121354 immunomodulator Drugs 0.000 description 2
- 230000002452 interceptive effect Effects 0.000 description 2
- 229940079322 interferon Drugs 0.000 description 2
- 108090000237 interleukin-24 Proteins 0.000 description 2
- 102000003898 interleukin-24 Human genes 0.000 description 2
- 230000002601 intratumoral effect Effects 0.000 description 2
- 210000001821 langerhans cell Anatomy 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 238000001638 lipofection Methods 0.000 description 2
- 230000004807 localization Effects 0.000 description 2
- 230000005923 long-lasting effect Effects 0.000 description 2
- 210000004698 lymphocyte Anatomy 0.000 description 2
- 238000002595 magnetic resonance imaging Methods 0.000 description 2
- 201000011614 malignant glioma Diseases 0.000 description 2
- 238000007726 management method Methods 0.000 description 2
- 230000010534 mechanism of action Effects 0.000 description 2
- 230000009437 off-target effect Effects 0.000 description 2
- 230000000174 oncolytic effect Effects 0.000 description 2
- -1 peptidyl chloromethyl ketone Chemical compound 0.000 description 2
- ISWRGOKTTBVCFA-UHFFFAOYSA-N pirfenidone Chemical compound C1=C(C)C=CC(=O)N1C1=CC=CC=C1 ISWRGOKTTBVCFA-UHFFFAOYSA-N 0.000 description 2
- 229960003073 pirfenidone Drugs 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- 230000002797 proteolythic effect Effects 0.000 description 2
- 238000011002 quantification Methods 0.000 description 2
- 238000003762 quantitative reverse transcription PCR Methods 0.000 description 2
- 108020003175 receptors Proteins 0.000 description 2
- 102000005962 receptors Human genes 0.000 description 2
- 230000012121 regulation of immune response Effects 0.000 description 2
- 230000004043 responsiveness Effects 0.000 description 2
- 238000010839 reverse transcription Methods 0.000 description 2
- 238000012552 review Methods 0.000 description 2
- 230000003248 secreting effect Effects 0.000 description 2
- 201000003708 skin melanoma Diseases 0.000 description 2
- 238000000638 solvent extraction Methods 0.000 description 2
- 238000012799 strong cation exchange Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 230000017423 tissue regeneration Effects 0.000 description 2
- 230000001131 transforming effect Effects 0.000 description 2
- 230000001052 transient effect Effects 0.000 description 2
- 238000012384 transportation and delivery Methods 0.000 description 2
- 229940030325 tumor cell vaccine Drugs 0.000 description 2
- 102000003298 tumor necrosis factor receptor Human genes 0.000 description 2
- 241000701161 unidentified adenovirus Species 0.000 description 2
- 238000010200 validation analysis Methods 0.000 description 2
- 238000012795 verification Methods 0.000 description 2
- HNSDLXPSAYFUHK-UHFFFAOYSA-N 1,4-bis(2-ethylhexyl) sulfosuccinate Chemical compound CCCCC(CC)COC(=O)CC(S(O)(=O)=O)C(=O)OCC(CC)CCCC HNSDLXPSAYFUHK-UHFFFAOYSA-N 0.000 description 1
- 102100035248 Alpha-(1,3)-fucosyltransferase 4 Human genes 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- 206010005003 Bladder cancer Diseases 0.000 description 1
- 208000003508 Botulism Diseases 0.000 description 1
- 102000007644 Colony-Stimulating Factors Human genes 0.000 description 1
- 108010071942 Colony-Stimulating Factors Proteins 0.000 description 1
- 201000003883 Cystic fibrosis Diseases 0.000 description 1
- 102000053602 DNA Human genes 0.000 description 1
- 238000001712 DNA sequencing Methods 0.000 description 1
- 101100447432 Danio rerio gapdh-2 gene Proteins 0.000 description 1
- 208000002699 Digestive System Neoplasms Diseases 0.000 description 1
- 238000012286 ELISA Assay Methods 0.000 description 1
- 238000008157 ELISA kit Methods 0.000 description 1
- 201000011001 Ebola Hemorrhagic Fever Diseases 0.000 description 1
- 241001115402 Ebolavirus Species 0.000 description 1
- 102000004533 Endonucleases Human genes 0.000 description 1
- 108010042407 Endonucleases Proteins 0.000 description 1
- 208000031637 Erythroblastic Acute Leukemia Diseases 0.000 description 1
- 208000036566 Erythroleukaemia Diseases 0.000 description 1
- 229940082999 Furin inhibitor Drugs 0.000 description 1
- 101150112014 Gapdh gene Proteins 0.000 description 1
- CEAZRRDELHUEMR-URQXQFDESA-N Gentamicin Chemical compound O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N CEAZRRDELHUEMR-URQXQFDESA-N 0.000 description 1
- 229930182566 Gentamicin Natural products 0.000 description 1
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 description 1
- 102000004269 Granulocyte Colony-Stimulating Factor Human genes 0.000 description 1
- 102100031573 Hematopoietic progenitor cell antigen CD34 Human genes 0.000 description 1
- 102100038885 Histone acetyltransferase p300 Human genes 0.000 description 1
- 101001022185 Homo sapiens Alpha-(1,3)-fucosyltransferase 4 Proteins 0.000 description 1
- 101000861452 Homo sapiens Forkhead box protein P3 Proteins 0.000 description 1
- 101001022148 Homo sapiens Furin Proteins 0.000 description 1
- 101000777663 Homo sapiens Hematopoietic progenitor cell antigen CD34 Proteins 0.000 description 1
- 101000882390 Homo sapiens Histone acetyltransferase p300 Proteins 0.000 description 1
- 101000914484 Homo sapiens T-lymphocyte activation antigen CD80 Proteins 0.000 description 1
- 101000635958 Homo sapiens Transforming growth factor beta-2 proprotein Proteins 0.000 description 1
- 102000048143 Insulin-Like Growth Factor II Human genes 0.000 description 1
- 108090001117 Insulin-Like Growth Factor II Proteins 0.000 description 1
- 102100037850 Interferon gamma Human genes 0.000 description 1
- 102100020881 Interleukin-1 alpha Human genes 0.000 description 1
- 108010065805 Interleukin-12 Proteins 0.000 description 1
- 102000013462 Interleukin-12 Human genes 0.000 description 1
- 108010082786 Interleukin-1alpha Proteins 0.000 description 1
- 102000000588 Interleukin-2 Human genes 0.000 description 1
- 108010002350 Interleukin-2 Proteins 0.000 description 1
- 102000010789 Interleukin-2 Receptors Human genes 0.000 description 1
- 108010038453 Interleukin-2 Receptors Proteins 0.000 description 1
- 108090001005 Interleukin-6 Proteins 0.000 description 1
- 108091092195 Intron Proteins 0.000 description 1
- 238000000134 MTT assay Methods 0.000 description 1
- 231100000002 MTT assay Toxicity 0.000 description 1
- 102000007651 Macrophage Colony-Stimulating Factor Human genes 0.000 description 1
- 108010046938 Macrophage Colony-Stimulating Factor Proteins 0.000 description 1
- 241001115401 Marburgvirus Species 0.000 description 1
- 108010076557 Matrix Metalloproteinase 14 Proteins 0.000 description 1
- 102100030216 Matrix metalloproteinase-14 Human genes 0.000 description 1
- 102100024129 Matrix metalloproteinase-24 Human genes 0.000 description 1
- 108050005214 Matrix metalloproteinase-24 Proteins 0.000 description 1
- 206010027480 Metastatic malignant melanoma Diseases 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 102000029749 Microtubule Human genes 0.000 description 1
- 108091022875 Microtubule Proteins 0.000 description 1
- 102100030608 Mothers against decapentaplegic homolog 7 Human genes 0.000 description 1
- 102100034256 Mucin-1 Human genes 0.000 description 1
- 108010008707 Mucin-1 Proteins 0.000 description 1
- 101000978776 Mus musculus Neurogenic locus notch homolog protein 1 Proteins 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 108010057466 NF-kappa B Proteins 0.000 description 1
- 102000003945 NF-kappa B Human genes 0.000 description 1
- 108090000028 Neprilysin Proteins 0.000 description 1
- 102000003729 Neprilysin Human genes 0.000 description 1
- 108091028043 Nucleic acid sequence Proteins 0.000 description 1
- 235000008331 Pinus X rigitaeda Nutrition 0.000 description 1
- 235000011613 Pinus brutia Nutrition 0.000 description 1
- 241000018646 Pinus brutia Species 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 241000589516 Pseudomonas Species 0.000 description 1
- 108700033844 Pseudomonas aeruginosa toxA Proteins 0.000 description 1
- 108091030071 RNAI Proteins 0.000 description 1
- 206010070308 Refractory cancer Diseases 0.000 description 1
- 102000006382 Ribonucleases Human genes 0.000 description 1
- 108010083644 Ribonucleases Proteins 0.000 description 1
- 101700026522 SMAD7 Proteins 0.000 description 1
- 102000012479 Serine Proteases Human genes 0.000 description 1
- 108010022999 Serine Proteases Proteins 0.000 description 1
- 206010041067 Small cell lung cancer Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 102100027222 T-lymphocyte activation antigen CD80 Human genes 0.000 description 1
- 108091005735 TGF-beta receptors Proteins 0.000 description 1
- 206010043376 Tetanus Diseases 0.000 description 1
- 102000016715 Transforming Growth Factor beta Receptors Human genes 0.000 description 1
- 102100030737 Transforming growth factor beta-2 proprotein Human genes 0.000 description 1
- 102000003425 Tyrosinase Human genes 0.000 description 1
- 108060008724 Tyrosinase Proteins 0.000 description 1
- 108091023045 Untranslated Region Proteins 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 208000021841 acute erythroid leukemia Diseases 0.000 description 1
- 238000009098 adjuvant therapy Methods 0.000 description 1
- 208000037844 advanced solid tumor Diseases 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 230000000840 anti-viral effect Effects 0.000 description 1
- 210000001130 astrocyte Anatomy 0.000 description 1
- 230000003416 augmentation Effects 0.000 description 1
- 230000003190 augmentative effect Effects 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 239000000090 biomarker Substances 0.000 description 1
- 201000001531 bladder carcinoma Diseases 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 230000020411 cell activation Effects 0.000 description 1
- 230000011712 cell development Effects 0.000 description 1
- 230000022534 cell killing Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000003610 charcoal Substances 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 230000007012 clinical effect Effects 0.000 description 1
- 238000003501 co-culture Methods 0.000 description 1
- 206010009887 colitis Diseases 0.000 description 1
- 210000001072 colon Anatomy 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 229940047120 colony stimulating factors Drugs 0.000 description 1
- 238000007398 colorimetric assay Methods 0.000 description 1
- 238000010835 comparative analysis Methods 0.000 description 1
- 230000001010 compromised effect Effects 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 210000004292 cytoskeleton Anatomy 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 230000009274 differential gene expression Effects 0.000 description 1
- 206010013023 diphtheria Diseases 0.000 description 1
- 238000002224 dissection Methods 0.000 description 1
- 229940000406 drug candidate Drugs 0.000 description 1
- 230000013020 embryo development Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- 230000008029 eradication Effects 0.000 description 1
- 210000003238 esophagus Anatomy 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 101150046339 fur gene Proteins 0.000 description 1
- ZXQYGBMAQZUVMI-GCMPRSNUSA-N gamma-cyhalothrin Chemical compound CC1(C)[C@@H](\C=C(/Cl)C(F)(F)F)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 ZXQYGBMAQZUVMI-GCMPRSNUSA-N 0.000 description 1
- 229940044627 gamma-interferon Drugs 0.000 description 1
- 238000012239 gene modification Methods 0.000 description 1
- 230000005017 genetic modification Effects 0.000 description 1
- 235000013617 genetically modified food Nutrition 0.000 description 1
- 229960002518 gentamicin Drugs 0.000 description 1
- 239000003862 glucocorticoid Substances 0.000 description 1
- 210000003714 granulocyte Anatomy 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 210000002443 helper t lymphocyte Anatomy 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- 102000046157 human CSF2 Human genes 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 230000002519 immonomodulatory effect Effects 0.000 description 1
- 210000002865 immune cell Anatomy 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 239000012642 immune effector Substances 0.000 description 1
- 230000037451 immune surveillance Effects 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 230000002055 immunohistochemical effect Effects 0.000 description 1
- 239000002955 immunomodulating agent Substances 0.000 description 1
- 230000008975 immunomodulatory function Effects 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 230000015788 innate immune response Effects 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 238000007917 intracranial administration Methods 0.000 description 1
- IXHBTMCLRNMKHZ-LBPRGKRZSA-N levobunolol Chemical compound O=C1CCCC2=C1C=CC=C2OC[C@@H](O)CNC(C)(C)C IXHBTMCLRNMKHZ-LBPRGKRZSA-N 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 229940031724 lung cancer vaccine Drugs 0.000 description 1
- 208000037841 lung tumor Diseases 0.000 description 1
- 210000003563 lymphoid tissue Anatomy 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 238000000816 matrix-assisted laser desorption--ionisation Methods 0.000 description 1
- 238000000074 matrix-assisted laser desorption--ionisation tandem time-of-flight detection Methods 0.000 description 1
- 210000003071 memory t lymphocyte Anatomy 0.000 description 1
- 208000021039 metastatic melanoma Diseases 0.000 description 1
- 208000010658 metastatic prostate carcinoma Diseases 0.000 description 1
- 210000000274 microglia Anatomy 0.000 description 1
- 210000004688 microtubule Anatomy 0.000 description 1
- UPSFMJHZUCSEHU-JYGUBCOQSA-N n-[(2s,3r,4r,5s,6r)-2-[(2r,3s,4r,5r,6s)-5-acetamido-4-hydroxy-2-(hydroxymethyl)-6-(4-methyl-2-oxochromen-7-yl)oxyoxan-3-yl]oxy-4,5-dihydroxy-6-(hydroxymethyl)oxan-3-yl]acetamide Chemical compound CC(=O)N[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@H]1[C@H](O)[C@@H](NC(C)=O)[C@H](OC=2C=C3OC(=O)C=C(C)C3=CC=2)O[C@@H]1CO UPSFMJHZUCSEHU-JYGUBCOQSA-N 0.000 description 1
- 210000000581 natural killer T-cell Anatomy 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 210000001672 ovary Anatomy 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 238000011338 personalized therapy Methods 0.000 description 1
- 238000009520 phase I clinical trial Methods 0.000 description 1
- 150000003906 phosphoinositides Chemical class 0.000 description 1
- 108010017843 platelet-derived growth factor A Proteins 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 238000002331 protein detection Methods 0.000 description 1
- 238000000275 quality assurance Methods 0.000 description 1
- 208000016691 refractory malignant neoplasm Diseases 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 238000007634 remodeling Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 230000006807 siRNA silencing Effects 0.000 description 1
- 230000004877 small RNA loading onto RISC Effects 0.000 description 1
- 208000000649 small cell carcinoma Diseases 0.000 description 1
- 208000000587 small cell lung carcinoma Diseases 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 230000003614 tolerogenic effect Effects 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 108700012359 toxins Proteins 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 230000005748 tumor development Effects 0.000 description 1
- 208000010570 urinary bladder carcinoma Diseases 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7105—Natural ribonucleic acids, i.e. containing only riboses attached to adenine, guanine, cytosine or uracil and having 3'-5' phosphodiester links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/13—Tumour cells, irrespective of tissue of origin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/21—Interferons [IFN]
- A61K38/217—IFN-gamma
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001136—Cytokines
- A61K39/001139—Colony stimulating factors [CSF]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
Abstract
Description
1. Murphy, K., Travers, P., Walport, M., ed. Janeway's Immunobiology. 7th ed. 2008, Garland Science: New York. 674-687
2. Fakhrai, H., et al., Phase I clinical trial of a TGF-beta antisense-modified tumor cell vaccine in patients with advanced glioma. Cancer Gene Ther, 2006. 13(12): p. 1052-60
3. Nemunaitis, J., GVAX (GMCSF gene modified tumor vaccine) in advanced stage non small cell lung cancer. J Control Release, 2003. 91(1-2): p. 225-31
4. Nemunaitis, J., et al., Phase 1/2 trial of autologous tumor mixed with an allogeneic GVAX vaccine in advanced-stage non-small-cell lung cancer. Cancer Gene Ther, 2006. 13(6): p. 555-62
5. Nemunaitis, J. and J. Nemunaitis, A review of vaccine clinical trials for non-small cell lung cancer. Expert Opin Biol Ther, 2007. 7(1): p. 89-102
6. Ahmad, M., R.C. Rees, and S.A. Ali, Escape from immunotherapy: possible mechanisms that influence tumor regression/progression. Cancer Immunol Immunother, 2004. 53(10): p. 844-54
7. Hege, K.M., K. Jooss, and D. Pardoll, GM-CSF gene-modifed cancer cell immunotherapies: of mice and men. Int Rev Immunol, 2006. 25(5-6): p. 321-52
8. Dranoff, G., et al., Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci U S A, 1993. 90(8): p. 3539-43
9. Hege, K.M. and D.P. Carbone, Lung cancer vaccines and gene therapy. Lung Cancer, 2003. 41 Suppl 1: p. S103-13
10. Huang, A.Y., et al., Role of bone marrow-derived cells in presenting MHC class I-restricted tumor antigens. Science, 1994. 264(5161): p. 961-5
11. Banchereau, J., et al., Immunobiology of dendritic cells. Annu Rev Immunol, 2000. 18: p. 767-811
12. Hodi, F.S., et al., Immunologic and clinical effects of antibody blockade of cytotoxic T lymphocyte-associated antigen 4 in previously vaccinated cancer patients. Proc Natl Acad Sci U S A, 2008. 105(8): p. 3005-10
13. Wick, W., U. Naumann, and M. Weller, Transforming growth factor-beta: a molecular target for the future therapy of glioblastoma. Curr Pharm Des, 2006. 12(3): p. 341-9
14. Bierie, B. and H.L. Moses, Tumour microenvironment: TGFbeta: the molecular Jekyll and Hyde of cancer. Nat Rev Cancer, 2006. 6(7): p. 506-20
15. Levy, L. and C.S. Hill, Alterations in components of the TGF-beta superfamily signaling pathways in human cancer. Cytokine Growth Factor Rev, 2006. 17(1-2): p. 41-58
16. Sporn, M.B., et al., Transforming growth factor-beta: biological function and chemical structure. Science, 1986. 233(4763): p. 532-4
17. Massague, J., The TGF-beta family of growth and differentiation factors. Cell, 1987. 49(4): p. 437-8
18. Bodmer, S., et al., Immunosuppression and transforming growth factor-beta in glioblastoma. Preferential production of transforming growth factor-beta 2. J Immunol, 1989. 143(10): p. 3222-9
19. Border, W.A. and E. Ruoslahti, Transforming growth factor-beta in disease: the dark side of tissue repair. J Clin Invest, 1992. 90(1): p. 1-7
20. Chen, T.C., et al., TGF-B2 and soluble p55 TNFR modulate VCAM-1 expression in glioma cells and brain derived endothelial cells. J Neuroimmunol, 1997. 73(1-2): p. 155-61
21. Li, M.O., et al., Transforming growth factor-beta regulation of immune responses. Annu Rev Immunol, 2006. 24: p. 99-146
22. Yamaguchi, Y., et al., Contrasting effects of TGF-beta 1 and TNF-alpha on the development of dendritic cells from progenitors in mouse bone marrow. Stem Cells, 1997. 15(2): p. 144-53
23. Geissmann, F., et al., TGF-beta 1 prevents the noncognate maturation of human dendritic Langerhans cells. J Immunol, 1999. 162(8): p. 4567-75
24. Ardeshna, K.M., et al., The PI3 kinase, p38 SAP kinase, and NF-kappaB signal transduction pathways are involved in the survival and maturation of lipopolysaccharide-stimulated human monocyte-derived dendritic cells. Blood, 2000. 96(3): p. 1039-46
25. Montenegro, D.E., et al., TGFbeta inhibits GM-CSF-induced phosphorylation of ERK and MEK in human myeloid leukaemia cell lines via inhibition of phosphatidylinositol 3-kinase (PI3-k). Cell Prolif, 2009. 42(1): p. 1-9
26. Steinman, R.M., et al., Dendritic cell function in vivo during the steady state: a role in peripheral tolerance. Ann N Y Acad Sci, 2003. 987: p. 15-25
27. Ashcroft, G.S., Bidirectional regulation of macrophage function by TGF-beta. Microbes Infect, 1999. 1(15): p. 1275-82
28. Du, C. and S. Sriram, Mechanism of inhibition of LPS-induced IL-12p40 production by IL-10 and TGF-beta in ANA-1 cells. J Leukoc Biol, 1998. 64(1): p. 92-7
29. Takeuchi, M., P. Alard, and J.W. Streilein, TGF-beta promotes immune deviation by altering accessory signals of antigen-presenting cells. J Immunol, 1998. 160(4): p. 1589-97
30. Ruffini, P.A., et al., Factors, including transforming growth factor beta, released in the glioblastoma residual cavity, impair activity of adherent lymphokine-activated killer cells. Cancer Immunol Immunother, 1993. 36(6): p. 409-16
31. Fakhrai, H., et al., Eradication of established intracranial rat gliomas by transforming growth factor beta antisense gene therapy. Proc Natl Acad Sci U S A, 1996. 93(7): p. 2909-14
32. Fantini, M.C., et al., Cutting edge: TGF-beta induces a regulatory phenotype in CD4+CD25- T cells through Foxp3 induction and down-regulation of Smad7. J Immunol, 2004. 172(9): p. 5149-53
33. Thomas, D.A. and J. Massague, TGF-beta directly targets cytotoxic T cell functions during tumor evasion of immune surveillance. Cancer Cell, 2005. 8(5): p. 369-80
34. Polak, M.E., et al., Mechanisms of local immunosuppression in cutaneous melanoma. Br J Cancer, 2007. 96(12): p. 1879-87
35. Rook, A.H., et al., Effects of transforming growth factor beta on the functions of natural killer cells: depressed cytolytic activity and blunting of interferon responsiveness. J Immunol, 1986. 136(10): p. 3916-20
36. Kasid, A., G.I. Bell, and E.P. Director, Effects of transforming growth factor-beta on human lymphokine-activated killer cell precursors. Autocrine inhibition of cellular proliferation and differentiation to immune killer cells. J Immunol, 1988. 141(2): p. 690-8
37. Tsunawaki, S., et al., Deactivation of macrophages by transforming growth factor-beta. Nature, 1988. 334(6179): p. 260-2
38. Hirte, H. and D.A. Clark, Generation of lymphokine-activated killer cells in human ovarian carcinoma ascitic fluid: identification of transforming growth factor-beta as a suppressive factor. Cancer Immunol Immunother, 1991. 32(5): p. 296-302
39. Naganuma, H., et al., Transforming growth factor-beta inhibits interferon-gamma secretion by lymphokine-activated killer cells stimulated with tumor cells. Neurol Med Chir (Tokyo), 1996. 36(11): p. 789-95
40. Penafuerte, C. and J. Galipeau, TGF beta secreted by B16 melanoma antagonizes cancer gene immunotherapy bystander effect. Cancer Immunol Immunother, 2008. 57(8): p. 1197-206
41. Nemunaitis, J., et al., Phase II trial of Belagenpumatucel-L, a TGF-beta2 antisense gene modified allogeneic tumor vaccine in advanced non small cell lung cancer (NSCLC) patients. Cancer Gene Ther, 2009. 16(8): p. 620-4
42. Maples PB, K.P., Oxendine I, Jay C, Yu Y, Kuhn J, Nemunaitis J, TAG Vaccine: Autologous Tumor Vaccine Genetically Modified to Express GM-CSF and Block Production of TGFB2. BioProcessing Journal, 2009. 8(2)
43. Nemunaitis, J., Kumar, P., Senzer, N., Yu, Y., Oxendine, I., Tong, A.W., Maples, P.B., A phase I trial of GMCSF gene-TGFbeta antisense gene autologous tumor cell (TAG) vaccine in advanced cancer. Mol Therapy, 2009. 17 (Suppl 1): p. S206
44. Maples, P.B., et al. Autologous Tumor Cell Vaccine Genetically Modified To Express GM-CSF and Block Expression of TGFb2 (Abstract # 553). in The Twelfth Annual Meeting of the American Society of Gene Therapy. 2009. San Diego, California
45. Page, R.E., et al., Increased expression of the pro-protein convertase furin predicts decreased survival in ovarian cancer. Cell Oncol, 2007. 29(4): p. 289-99
46. Schalken, J.A., et al., fur gene expression as a discriminating marker for small cell and nonsmall cell lung carcinomas. J Clin Invest, 1987. 80(6): p. 1545-9
47. Mbikay, M., et al., Comparative analysis of expression of the proprotein convertases furin, PACE4, PC1 and PC2 in human lung tumours. Br J Cancer, 1997. 75(10): p. 1509-14
48. Cheng, M., et al., Pro-protein convertase gene expression in human breast cancer. Int J Cancer, 1997. 71(6): p. 966-71
49. Bassi, D.E., H. Mahloogi, and A.J. Klein-Szanto, The proprotein convertases furin and PACE4 play a significant role in tumor progression. Mol Carcinog, 2000. 28(2): p. 63-9
50. Bassi, D.E., et al., Elevated furin expression in aggressive human head and neck tumors and tumor cell lines. Mol Carcinog, 2001. 31(4): p. 224-32
51. Lopez de Cicco, R., et al., Human carcinoma cell growth and invasiveness is impaired by the propeptide of the ubiquitous proprotein convertase furin. Cancer Res, 2005. 65(10): p. 4162-71
52. Khatib, A.M., et al., Proprotein convertases in tumor progression and malignancy: novel targets in cancer therapy. Am J Pathol, 2002. 160(6): p. 1921-35
53. Thomas, G., Furin at the cutting edge: from protein traffic to embryogenesis and disease. Nat Rev Mol Cell Biol, 2002. 3(10): p. 753-66
54. Pesu, M., et al., T-cell-expressed proprotein convertase furin is essential for maintenance of peripheral immune tolerance. Nature, 2008. 455(7210): p. 246-50
55. Pesu, M., et al., Proprotein convertase furin is preferentially expressed in T helper 1 cells and regulates interferon gamma. Blood, 2006. 108(3): p. 983-5
56. Lu, J., et al., TAP-independent presentation of CTL epitopes by Trojan antigens. J Immunol, 2001. 166(12): p. 7063-71
57. Fogel-Petrovic, M., et al., Physiological concentrations of transforming growth factor beta1 selectively inhibit human dendritic cell function. Int Immunopharmacol, 2007. 7(14): p. 1924-33
58. Bommireddy, R. and T. Doetschman, TGFbeta1 and Treg cells: alliance for tolerance. Trends Mol Med, 2007. 13(11): p. 492-501
59. Henrich, S., et al., The crystal structure of the proprotein processing proteinase furin explains its stringent specificity. Nat Struct Biol, 2003. 10(7): p. 520-6
60. Pearton, D.J., et al., Proprotein convertase expression and localization in epidermis: evidence for multiple roles and substrates. Exp Dermatol, 2001. 10(3): p. 193-203
61. Rao, D., Maples, P.B., Senzer, N., Kumar, P., Wang, Z., papper, B.O., Yu, Y., Haddock, C., Tong, A., Nemunaitis, J., Bi-functional shRNA: A novel approach of RNA interference. (submitted), 2009
62. Matranga, C., et al., Passenger-strand cleavage facilitates assembly of siRNA into Ago2-containing RNAi enzyme complexes. Cell, 2005. 123(4): p. 607-20
63. Leuschner, P.J., et al., Cleavage of the siRNA passenger strand during RISC assembly in human cells. EMBO Rep, 2006. 7(3): p. 314-20
64. Rana, S., et al., Stathmin 1: a novel therapeutic target for anticancer activity. Expert Rev Anticancer Ther, 2008. 8(9): p. 1461-70
65. Azuma-Mukai, A., et al., Characterization of endogenous human Argonautes and their miRNA partners in RNA silencing. Proc Natl Acad Sci U S A, 2008. 105(23): p. 7964-9
66. Jackson, S.A., S. Koduvayur, and S.A. Woodson, Self-splicing of a group I intron reveals partitioning of native and misfolded RNA populations in yeast. RNA, 2006. 12(12): p. 2149-59
67. Funston, G.M., et al., Expression of heterologous genes in oncolytic adenoviruses using picornaviral 2A sequences that trigger ribosome skipping. J Gen Virol, 2008. 89(Pt 2): p. 389-96
68. Tong, A.W., et al., Intratumoral injection of INGN 241, a nonreplicating adenovector expressing the melanoma-differentiation associated gene-7 (mda-7/IL24): biologic outcome in advanced cancer patients. Mol Ther, 2005. 11(1): p. 160-72
69. Hu, X., et al., Characterization of a unique factor-independent variant derived from human factor-dependent TF-1 cells: a transformed event. Leuk Res, 1998. 22(9): p. 817-26
70. Santoli, D., et al., Synergistic and antagonistic effects of recombinant human interleukin (IL) 3, IL-1 alpha, granulocyte and macrophage colony-stimulating factors (G-CSF and M-CSF) on the growth of GM-CSF-dependent leukemic cell lines. J Immunol, 1987. 139(10): p. 3348-54
71. Romero, P., Current state of vaccine therapies in non-small-cell lung cancer. Clin Lung Cancer, 2008. 9 Suppl 1: p. S28-36
72. Robinson, J., et al., The European searchable tumour line database. Cancer Immunol Immunother, 2009
73. http:jura.wi. mit.edu/bioc/ siRNAext
74. Kumar, P.J., C. Oxendine, I Nemunaitis, J. Maples, P., TAG Xenograft Vaccine: Xenograft-Expanded Autologous Tumor Vaccine Genetically Modified to Express GM-CSF and Block Production of TGFβ2. BioProcessing Journal, 2009(Spring 2009): p. 30-36
75. Burghardt, I., et al., Pirfenidone inhibits TGF-beta expression in malignant glioma cells. Biochem Biophys Res Commun, 2007. 354(2): p. 542-7
76. McMahon, S., M.H. Laprise, and C.M. Dubois, Alternative pathway for the role of furin in tumor cell invasion process. Enhanced MMP-2 levels through bioactive TGFbeta. Exp Cell Res, 2003. 291(2): p. 326-39
77. Arteaga, C.L., Inhibition of TGFbeta signaling in cancer therapy. Curr Opin Genet Dev, 2006. 16(1): p. 30-7
78. Constam, D.B., et al., Differential expression of transforming growth factor-beta 1, -beta 2, and -beta 3 by glioblastoma cells, astrocytes, and microglia. J Immunol, 1992. 148(5): p. 1404-10
79. Eastham, J.A., et al., Transforming growth factor-beta 1: comparative immunohistochemical localization in human primary and metastatic prostate cancer. Lab Invest, 1995. 73(5): p. 628-35
80. Friedman, E., et al., High levels of transforming growth factor beta 1 correlate with disease progression in human colon cancer. Cancer Epidemiol Biomarkers Prev, 1995. 4(5): p. 549-54
81. Jakowlew, S.B., et al., Expression of transforming growth factor beta ligand and receptor messenger RNAs in lung cancer cell lines. Cell Growth Differ, 1995. 6(4): p. 465-76
82. Kong, F.M., et al., Elevated plasma transforming growth factor-beta 1 levels in breast cancer patients decrease after surgical removal of the tumor. Ann Surg, 1995. 222(2): p. 155-62
83. Yamada, N., et al., Enhanced expression of transforming growth factor-beta and its type-I and type-II receptors in human glioblastoma. Int J Cancer, 1995. 62(4): p. 386-92
84. Eder, I.E., et al., Transforming growth factors-beta 1 and beta 2 in serum and urine from patients with bladder carcinoma. J Urol, 1996. 156(3): p. 953-7
Claims (76)
- 顆粒球マクロファージコロニー刺激因子(GM−CSF)cDNAをコードする、プロモーターに作動可能に連結した第一の核酸インサート及び
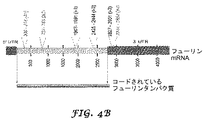
フューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサート
を含むbishRNAフューリン/GMCSF発現ベクタープラスミドと、
1又は2以上の必要に応じたワクチンアジュバントと
を含むワクチン組成物。 - GM−CSFがヒトのものである、請求項1に記載の組成物。
- shRNAが、siRNA(切断依存的)及びmiRNA(切断非依存的)モチーフを組み入れている、請求項1に記載の組成物。
- shRNAが、切断依存的及び切断非依存的の両方であるフューリン発現阻害因子である、請求項1に記載の組成物。
- shRNAが、二機能性shRNAとしてさらに定義される、請求項1に記載の組成物。
- ピコルナウイルス2Aリボソームスキップペプチドが、第一及び第二の核酸インサート間にインターカレートしている、請求項1に記載の組成物。
- プロモーターがCMV哺乳類プロモーターである、請求項1に記載の組成物。
- CMV哺乳類プロモーターが、CMV IE 5’UTRエンハンサー配列及びCMV IEイントロンAを含有する、請求項7に記載の組成物。
- shRNAの標的となる領域が、フューリンmRNA転写産物の3’UTR領域配列である、請求項1に記載の組成物。
- shRNAの標的となる領域が、フューリンmRNA転写産物のコード領域である、請求項1に記載の組成物。
- 癌の症状の予防、治療及び/又は寛解を必要とする患者を同定するステップと、
顆粒球マクロファージコロニー刺激因子(GM−CSF)cDNAをコードする、プロモーターに作動可能に連結した第一の核酸インサート及びフューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサートを含むbishRNAフューリン/GMCSF発現ベクタープラスミド、並びに1又は2以上の必要に応じたワクチンアジュバントを含む自家細胞ワクチンを投与するステップと
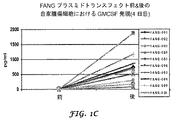
を含むことにより、患者における癌の症状を予防、治療及び/又は寛解させる方法。 - 1又は2以上の癌細胞におけるトランスフォーミング増殖因子ベータ(TGF−ベータ又はTGF−β)及びGM−CSFのレベルを測定することにより療法の進行をモニターするステップであって、TGF−βレベルの低下及びGM−CSFレベルの上昇が療法の成功を示すステップと、
前記TGF−β及びGM−CSFのレベルに基づき自家細胞ワクチンの投与を変更するステップと
をさらに含む、請求項11に記載の方法。 - TGF−βが、TGF−β1、TGF−β2及びTGF−β3のうち少なくとも1種類から選択される、請求項12に記載の方法。
- 癌が、メラノーマ、非小細胞肺癌、胆嚢癌、結腸直腸癌、乳癌、卵巣癌、肝癌、肝癌転移及びユーイング肉腫からなる群から選択される、請求項11に記載の方法。
- shRNAが、siRNA(切断依存的)及びmiRNA(切断非依存的)モチーフを組み入れている、請求項11に記載の方法。
- shRNAが、切断依存的及び切断非依存的両方のフューリン発現阻害因子である、請求項11に記載の方法。
- shRNAが、二機能性shRNAとしてさらに定義される、請求項11に記載の方法。
- GM−CSF cDNAをコードする、プロモーターに作動可能に連結した第一の核酸インサート及び
フューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサート
を含むbishRNAフューリン/GMCSF発現ベクタープラスミドと、
1又は2以上の必要に応じたワクチンアジュバントと
を含む、自家フューリンノックダウン及び顆粒球マクロファージコロニー刺激因子(GM−CSF)増強(FANG)癌ワクチン組成物。 - 組成物が、癌の症状の予防、治療及び/又は寛解に用いられ、前記癌が、メラノーマ、非小細胞肺癌、胆嚢癌、結腸直腸癌、乳癌、卵巣癌、肝癌、肝癌転移及びユーイング肉腫からなる群から選択される、請求項18に記載の組成物。
- NSCLCの症状の予防、治療及び/又は寛解を必要とする患者を同定するステップと、
GM−CSF cDNAをコードする、プロモーターに作動可能に連結した第一の核酸インサート、及びフューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサートを含むbishRNAフューリン/GMCSF発現ベクタープラスミド、並びに1又は2以上の必要に応じたワクチンアジュバントを含むFANGワクチンを投与するステップと
を含む、フューリンノックダウン及び顆粒球マクロファージコロニー刺激因子(GM−CSF)増強(FANG)癌ワクチンの投与により、患者における非小細胞肺癌(NSCLC)の症状を治療、予防及び/又は寛解させる方法。 - 1又は2以上のNSCLC細胞におけるトランスフォーミング増殖因子ベータ(TGF−ベータ又はTGF−β)及びGM−CSFのレベルを測定することにより療法の進行をモニターするステップであって、TGF−βレベルの低下及びGM−CSFレベルの上昇が療法の成功を示すステップと、
前記TGF−β及びGM−CSFのレベルに基づき自家細胞ワクチンの投与を変更するステップと
をさらに含む、請求項20に記載の方法。 - TGF−βが、ヒトTGF−β1、TGF−β2及びTGF−β3のうち少なくとも1種類から選択される、請求項21に記載の方法。
- 1又は2以上の癌細胞を患者から無菌的に収集するステップと、
前記収集した細胞を無菌容器内の抗生物質溶液中に置くステップと、
前記収集した溶液から細胞懸濁液を生成するステップであって、前記細胞懸濁液の生成が酵素による解体、機械的脱凝集又は両者によって達成されるステップと、
前記細胞懸濁液を電気穿孔することによって前記細胞を遺伝的に改変し、GM−CSF cDNAをコードする、プロモーターに作動可能に連結した第一の核酸インサート、及びフューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサートを含むbishRNAフューリン/GMCSF発現ベクタープラスミドを有するワクチンを作製するステップと、
前記ワクチンを収集するステップと、
前記ワクチンに放射線照射するステップと、
前記ワクチンを凍結するステップと
を含む、フューリンノックダウン及び顆粒球マクロファージコロニー刺激因子(GM−CSF)増強(FANG)癌ワクチンを作製する方法。 - 1又は2以上の癌細胞が、メラノーマ、非小細胞肺癌、胆嚢癌、結腸直腸癌、乳癌、卵巣癌、肝癌、肝癌転移及びユーイング肉腫からなる群から選択される癌を患う患者から収集される、請求項23に記載の方法。
- 遺伝的に改変された細胞が、放射線照射によって増殖能力のない状態にされている、請求項23に記載の方法。
- 遺伝的に改変された細胞が自家細胞である、請求項23に記載の方法。
- 遺伝的に改変された細胞が異種移植片増幅細胞である、請求項23に記載の方法。
- 遺伝的に改変された細胞が同種異系細胞である、請求項23に記載の方法。
- 同種異系細胞が樹立細胞系である、請求項28に記載の方法。
- 遺伝的に改変された細胞が、月に1回を最大12用量まで対象に投与される、請求項23に記載の方法。
- 対象に投与される遺伝的に改変された細胞の用量が、1×107細胞/注射〜5×107細胞/注射である、請求項23に記載の方法。
- 遺伝的に改変された細胞の投与が、追加的な治療薬との併用療法の一部である、請求項31に記載の方法。
- 併用療法に用いられる追加的な治療薬がγIFNである、請求項32に記載の方法。
- 併用療法において対象に投与されるγIFNの用量が、50又は100μg/m2である、請求項33に記載の方法。
- トランスフェクション後に、遺伝的に改変された細胞をγIFNとインキュベートするさらなるステップを含む、請求項23に記載の方法。
- トランスフェクション後に、遺伝的に改変された細胞に適用されるγIFNの用量が、約250U/mlである、請求項35に記載の方法。
- 標的細胞を選択するステップと、
前記標的細胞に、プロモーター及び前記プロモーターに作動可能に連結した核酸インサートを含む発現ベクターをトランスフェクトするステップであって、前記インサートが、フューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、ステップと
を含む、フューリンノックダウンを介してトランスフォーミング増殖因子ベータ(TGF−ベータ又はTGF−β)発現を阻害するためのsiRNAを介在した方法。 - shRNAが、siRNA(切断依存的)及びmiRNA(切断非依存的)モチーフを組み入れている、請求項37に記載の方法。
- shRNAが、切断依存的及び切断非依存的の両方であるフューリン発現阻害因子である、請求項37に記載の方法。
- shRNAが、二機能性shRNAとしてさらに定義される、請求項37に記載の方法。
- shRNAの標的となる領域が、フューリンmRNA転写産物の3’UTR領域配列である、請求項37に記載の方法。
- shRNAの標的となる領域が、フューリンmRNA転写産物のコード領域である、請求項37に記載の方法。
- 標的細胞を選択するステップと、
前記標的細胞に、GM−CSFをコードする、プロモーターに作動可能に連結した第一の核酸インサート、及びフューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサートを含む発現ベクターをトランスフェクトするステップと
を含む、標的細胞における抗原の発現、提示及びプロセシングを増強するため並びにトランスフォーミング増殖因子ベータ(TGF−ベータ又はTGF−β)の分泌性免疫抑制活性を減衰するための方法。 - 標的細胞が自家細胞である、請求項43に記載の方法。
- 標的細胞が同種異系細胞である、請求項43に記載の方法。
- 同種異系細胞が樹立ヒト細胞系である、請求項45に記載の方法。
- 標的細胞が、プラスミドベクター電気穿孔によりトランスフェクトされる、請求項43に記載の方法。
- TGF−βが、TGF−β1、TGF−β2及びTGF−β3のうち少なくとも1種類から選択される、請求項43に記載の方法。
- 治療を必要とする対象を同定するステップと、
前記対象から癌組織試料を収集するステップと、
前記収集された癌試料における癌細胞を、GM−CSFをコードする、プロモーターに作動可能に連結した第一の核酸インサート、及びフューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる1又は2以上の低分子ヘアピン型RNA(shRNA)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサートを含む発現ベクターを前記細胞にトランスフェクトすることによって、遺伝的に改変するステップと、
癌の症状の治療又は寛解に十分な量で、前記対象に治療上有効用量の前記遺伝的に改変された細胞を投与するステップと
を含む、癌を治療する方法。 - 癌が、メラノーマ、非小細胞肺癌、胆嚢癌、結腸直腸癌、乳癌、卵巣癌、肝癌、肝癌転移及びユーイング肉腫からなる癌の群から選ばれる、請求項49に記載の方法。
- 遺伝的に改変された細胞が、放射線照射により増殖能力のない状態にされている、請求項49に記載の方法。
- 遺伝的に改変された細胞が自家細胞である、請求項49に記載の方法。
- 遺伝的に改変された細胞が同種異系細胞である、請求項49に記載の方法。
- 同種異系細胞が樹立細胞系である、請求項53に記載の方法。
- 遺伝的に改変された細胞が異種移植片増幅細胞である、請求項49に記載の方法。
- 遺伝的に改変された細胞が、月に1回を最大12用量まで対象に投与される、請求項49に記載の方法。
- 対象に投与される遺伝的に改変された細胞の用量が、1×107細胞/注射〜5×107細胞/注射である、請求項49に記載の方法。
- 遺伝的に改変された細胞の投与が、追加的な治療薬との併用療法の一部である、請求項49に記載の方法。
- 併用療法に用いられる追加的な治療薬がγIFNである、請求項58に記載の方法。
- 併用療法において対象に投与されるγIFNの用量が、50又は100μg/m2である、請求項59に記載の方法。
- トランスフェクション後に、遺伝的に改変された細胞をγIFNとインキュベートするさらなるステップを含む、請求項49に記載の方法。
- トランスフェクション後に、遺伝的に改変された細胞に適用されるγIFNの用量が、約250U/mlである、請求項61に記載の方法。
- 顆粒球マクロファージコロニー刺激因子(GM−CSF)cDNAをコードする、プロモーターに作動可能に連結した第一の核酸インサート、並びに
RNA干渉の切断及び隔離機構両方のための単一の標的部位を提供する1又は2以上の二機能性低分子ヘアピン型RNA(shRNAフューリン)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサートであって、二機能性shRNAフューリンが、フューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる、完全に相補的なガイド鎖及びパッセンジャー鎖を含む第一のステム−ループ構造並びに前記パッセンジャー鎖の1又は2以上の塩基対ミスマッチを含む第二のステム−ループ構造を含む第二の核酸インサート
を含むbishRNAフューリン/GMCSF発現ベクタープラスミドと、
1又は2以上の必要に応じたワクチンアジュバントと
を含む、RNA干渉を介したフューリン発現の阻害による癌治療のための自家細胞ワクチン組成物。 - 第二のステム−ループ構造が、3塩基対ミスマッチを含む、請求項63に記載の組成物。
- 塩基対ミスマッチが、パッセンジャー鎖のポジション9〜11に位置する、請求項63に記載の組成物。
- 顆粒球マクロファージコロニー刺激因子(GM−CSF)cDNAをコードする、プロモーターに作動可能に連結した第一の核酸インサート、並びに
RNA干渉の切断及び隔離機構両方のための単一の標的部位を提供する1又は2以上の二機能性低分子ヘアピン型RNA(shRNAフューリン)をコードする、前記プロモーターに作動可能に連結した第二の核酸インサートであって、二機能性shRNAフューリンが、フューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害できる、完全に相補的なガイド鎖及びパッセンジャー鎖を含む第一のステム−ループ構造並びに前記ガイド鎖の1又は2以上の塩基対ミスマッチを含む第二のステム−ループ構造を含む第二の核酸インサート
を含むbishRNAフューリン/GMCSF発現ベクタープラスミドと、
1又は2以上の必要に応じたワクチンアジュバントと
を含む、RNA干渉を介したフューリン発現の阻害による癌治療のための自家細胞ワクチン組成物。 - 第二のステム−ループ構造が、3塩基対ミスマッチを含む、請求項66に記載の組成物。
- 塩基対ミスマッチが、ガイド鎖のポジション9〜11に位置する、請求項66に記載の組成物。
- 標的細胞を選択するステップと、
前記標的細胞に、1又は2以上のGM−CSFと、完全に相補的なガイド鎖及びパッセンジャー鎖を含む第一のステム−ループ構造並びに前記パッセンジャー鎖の1又は2以上の塩基対ミスマッチを含む第二のステム−ループ構造を含み、フューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害する二機能性低分子ヘアピン(shRNA)フューリンとを含むコンストラクトをトランスフェクトするステップと
を含む、標的細胞においてRNA干渉を介してフューリン発現を阻害する方法。 - 第二のステム−ループ構造が、3塩基対ミスマッチを含む、請求項69に記載の方法。
- 塩基対ミスマッチが、パッセンジャー鎖のポジション9〜11に位置する、請求項69に記載の方法。
- 塩基対ミスマッチが、Ago2介在性の切断を妨げることによってパッセンジャー鎖離脱を促す、請求項69に記載の方法。
- 塩基対ミスマッチがガイド鎖のポジションを占める、請求項69に記載の方法。
- 標的細胞を選択するステップと、
前記標的細胞に、1又は2以上のGM−CSFと、完全に相補的なガイド鎖及びパッセンジャー鎖を含む第一のステム−ループ構造並びに前記ガイド鎖の1又は2以上の塩基対ミスマッチを含む第二のステム−ループ構造を含み、フューリンをコードするmRNA転写産物の領域とハイブリダイズして、これによりRNA干渉を介してフューリン発現を阻害する二機能性低分子ヘアピン(shRNA)フューリンとを含むコンストラクトをトランスフェクトするステップと
を含む、標的細胞におけるRNA干渉を介してフューリン発現を阻害する方法。 - 第二のステム−ループ構造が、3塩基対ミスマッチを含む、請求項74に記載の方法。
- 塩基対ミスマッチが、ガイド鎖のポジション9〜11に位置する、請求項74に記載の方法。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US28968109P | 2009-12-23 | 2009-12-23 | |
| US61/289,681 | 2009-12-23 | ||
| US30977710P | 2010-03-02 | 2010-03-02 | |
| US61/309,777 | 2010-03-02 | ||
| PCT/US2010/061344 WO2011079077A1 (en) | 2009-12-23 | 2010-12-20 | Furin-knockdown and gm-csf-augmented (fang) cancer vaccine |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2014111779A Division JP5820506B2 (ja) | 2009-12-23 | 2014-05-29 | フューリンノックダウン及びgm−csf増強(fang)癌ワクチン |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2013515731A true JP2013515731A (ja) | 2013-05-09 |
| JP5553906B2 JP5553906B2 (ja) | 2014-07-23 |
Family
ID=44196113
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012546121A Active JP5553906B2 (ja) | 2009-12-23 | 2010-12-20 | フューリンノックダウン及びgm−csf増強(fang)癌ワクチン |
| JP2014111779A Active JP5820506B2 (ja) | 2009-12-23 | 2014-05-29 | フューリンノックダウン及びgm−csf増強(fang)癌ワクチン |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2014111779A Active JP5820506B2 (ja) | 2009-12-23 | 2014-05-29 | フューリンノックダウン及びgm−csf増強(fang)癌ワクチン |
Country Status (13)
| Country | Link |
|---|---|
| US (4) | US9132146B2 (ja) |
| EP (1) | EP2515916B1 (ja) |
| JP (2) | JP5553906B2 (ja) |
| KR (2) | KR101548567B1 (ja) |
| CN (2) | CN102917709B (ja) |
| AR (1) | AR081057A1 (ja) |
| AU (1) | AU2010333753B2 (ja) |
| CA (1) | CA2785258A1 (ja) |
| HK (1) | HK1258331A1 (ja) |
| IL (1) | IL220589A0 (ja) |
| SG (3) | SG10201606680QA (ja) |
| TW (2) | TW201130975A (ja) |
| WO (1) | WO2011079077A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017511128A (ja) * | 2014-03-26 | 2017-04-20 | トカジェン インコーポレーテッド | 免疫刺激活性を有するレトロウイルスベクター |
| JP2021509253A (ja) * | 2017-10-10 | 2021-03-25 | ナントバイオ,インコーポレイテッド | ウイルス生成ペイロードに対して低い毒性を有する改変ec7細胞 |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8603991B2 (en) | 2005-11-18 | 2013-12-10 | Gradalis, Inc. | Individualized cancer therapy |
| SG181881A1 (en) | 2009-12-23 | 2012-07-30 | Gradalis Inc | Furin-knockdown bi-functional rna |
| CN102917709B (zh) | 2009-12-23 | 2018-04-24 | 格兰达利斯有限公司 | 弗林蛋白酶敲减和gm-csf增强的(fang)癌症疫苗 |
| US20140141015A1 (en) | 2010-09-20 | 2014-05-22 | Douglas Lake | QSOX1 as an Anti-Neoplastic Drug Target |
| US8946186B2 (en) | 2010-09-20 | 2015-02-03 | Arizona Board Of Regents, A Body Corporate Of The State Of Arizona Acting For And On Behalf Of Arizona State University | QSOX1 as an anti-neoplastic drug target |
| WO2013115579A1 (ko) * | 2012-01-31 | 2013-08-08 | 연세대학교 산학협력단 | TGF-β2 발현을 억제하는 shRNA |
| US9890215B2 (en) * | 2012-06-22 | 2018-02-13 | King's College London | Vista modulators for diagnosis and treatment of cancer |
| US20160024600A1 (en) * | 2013-03-15 | 2016-01-28 | The United States of America, as represented by the Secretary, Department of Health and Human Ser | Coincidence reporter gene system |
| US10640542B2 (en) * | 2013-07-18 | 2020-05-05 | Vib Vzw | Fusokines involving cytokines with strongly reduced receptor binding affinities |
| WO2015152609A1 (ko) * | 2014-03-31 | 2015-10-08 | 연세대학교 산학협력단 | GM-CSF 유전자; 데코린 유전자; TGF-β2 발현을 억제하는 shRNA; 및 FoxP3 발현을 억제하는 shRNA를 포함하는 항종양 조성물 |
| KR101713407B1 (ko) * | 2014-03-31 | 2017-03-07 | 연세대학교 산학협력단 | 아데노바이러스 감염 및 복제가 가능한 흑색종 세포주 |
| EP3134528A4 (en) * | 2014-04-25 | 2017-12-06 | Strike Bio, Inc. | Multiple targeted rnai for the treatment of cancers |
| ES2852002T3 (es) | 2015-07-30 | 2021-09-10 | Endor Tech S L | Factor estimulante de colonias para su uso en el tratamiento del cáncer de páncreas o de colon |
| JP6898325B2 (ja) * | 2015-12-08 | 2021-07-07 | ユニバーシティ−インダストリー・ファンデーション・ヨンセイ・ユニバーシティ | GM−CSF遺伝子;Flt3L−TRAIL融合遺伝子;TGF−βの発現を抑制するshRNA;およびHSP発現を抑制するshRNAを含む抗腫瘍組成物 |
| CN105734080A (zh) * | 2016-01-30 | 2016-07-06 | 山西大学 | 一种靶向抗癌基因-质粒及其构建方法和应用 |
| WO2020048942A1 (en) * | 2018-09-04 | 2020-03-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for enhancing cytotoxic t lymphocyte-dependent immune responses |
| CN109160888B (zh) * | 2018-10-09 | 2022-03-01 | 四川医立特生物医药有限公司 | 一种含脒基的对称化合物及其应用 |
| JP7211139B2 (ja) * | 2019-02-14 | 2023-01-24 | 日本電信電話株式会社 | 校閲方法、情報処理装置および校閲プログラム |
| CN110760477A (zh) * | 2019-10-10 | 2020-02-07 | 杭州广科安德生物科技有限公司 | 一种小鼠肝脏高转移肠癌细胞株的建立方法,细胞株和应用 |
| CN115551537A (zh) | 2019-12-03 | 2022-12-30 | 纽沃进公司 | 肿瘤细胞疫苗 |
| EP4090380A1 (en) * | 2020-01-13 | 2022-11-23 | Gradalis, Inc. | Methods for treating cancers using gm-csf encoding polynucleotide and additional agents |
| WO2021146213A1 (en) * | 2020-01-13 | 2021-07-22 | Gradalis, Inc. | Methods for treating cancers using gm-csf encoding polynucleotide and additional agents |
| EP4158025A1 (en) * | 2020-05-26 | 2023-04-05 | Gradalis, Inc. | Methods for the treatment of viral respiratory infections |
| WO2022055817A1 (en) | 2020-09-09 | 2022-03-17 | Gradalis, Inc. | Composition formulated into inhalable dosage forms for the treatment of lung|tumors |
| CN117136237A (zh) * | 2021-03-10 | 2023-11-28 | 格兰达利斯有限公司 | 用于治疗癌症的方法 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62164695A (ja) * | 1985-12-21 | 1987-07-21 | ヘキスト、アクチエンゲゼルシヤフト | Gm−csfタンパク質、その誘導体、このタイプのタンパク質の調製及びそれらの用途 |
| US20070224194A1 (en) * | 2004-05-20 | 2007-09-27 | Ludwig Institute For Cancer Research | Methods for Inhibiting Angiogenesis and/or Lymphangiogenesis |
Family Cites Families (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6506559B1 (en) | 1997-12-23 | 2003-01-14 | Carnegie Institute Of Washington | Genetic inhibition by double-stranded RNA |
| BR9908267A (pt) * | 1998-02-27 | 2000-10-24 | Univ Pennsylvania | Plasmìdeo, composição farmacêutica, processos para induzir uma resposta imunológica em um indivìduo contra um imunógeno, para imunizar um indivìduo contra uma infecção por vìrus de herpes simples e para tratar um indivìduo que tem uma doença autoimune, vacina recombinante, e, patógeno atenuado vivo |
| US8791081B2 (en) | 1999-08-13 | 2014-07-29 | Case Western Reserve University | MGMT inhibitor combination for the treatment of neoplastic disorders |
| JP4095895B2 (ja) | 2000-12-01 | 2008-06-04 | マックス−プランク−ゲゼルシャフト ツール フォーデルング デル ヴィッセンシャフテン エー.ヴェー. | Rna干渉を媒介する短鎖rna分子 |
| JP2004519247A (ja) | 2001-03-20 | 2004-07-02 | オーソ・クリニカル・ダイアグノスティックス・インコーポレーテッド | 発現プロファイルおよび使用法 |
| US20050143333A1 (en) | 2001-05-18 | 2005-06-30 | Sirna Therapeutics, Inc. | RNA interference mediated inhibition of interleukin and interleukin receptor gene expression using short interfering nucleic acid (SINA) |
| US20050080031A1 (en) | 2001-05-18 | 2005-04-14 | Sirna Therapeutics, Inc. | Nucleic acid treatment of diseases or conditions related to levels of Ras, HER2 and HIV |
| BR0211111A (pt) | 2001-07-12 | 2004-06-22 | Univ Massachusetts | Molécula de ácido nucleico isolada, vetor, célula hospedeira, transgene, precursor de rna engenheirado, animal transgênico não humano, e, método de induzir a interferência de ácido ribonucleico de um gene alvo em uma célula |
| US20030144823A1 (en) | 2001-11-01 | 2003-07-31 | Fox Jeffrey J. | Scale-free network inference methods |
| JP2005508396A (ja) | 2001-11-02 | 2005-03-31 | イントラディグム、コーポレイション | 核酸送達ビヒクルのための治療方法 |
| EP1474433A4 (en) | 2002-02-20 | 2005-02-23 | Sirna Therapeutics Inc | TARGET LOCALIZATION TARGETED BY RNA INTERFERENCE AND TARGET VALIDATION WITH SHORT INTERFERING NUCLEIC ACID (siNA) |
| US20040241854A1 (en) | 2002-08-05 | 2004-12-02 | Davidson Beverly L. | siRNA-mediated gene silencing |
| US20040023390A1 (en) | 2002-08-05 | 2004-02-05 | Davidson Beverly L. | SiRNA-mediated gene silencing with viral vectors |
| DE60321689D1 (de) | 2002-08-29 | 2008-07-31 | Gene Network Sciences Inc | Systeme und verfahren zum schliessen auf biologische netzwerke |
| CN1756568A (zh) | 2002-10-09 | 2006-04-05 | 衣阿华中央卫生系统 | 使用表达α(1,3)-半乳糖基转移酶的同种异型肿瘤细胞的抗肿瘤免疫 |
| US20040088116A1 (en) | 2002-11-04 | 2004-05-06 | Gene Network Sciences, Inc. | Methods and systems for creating and using comprehensive and data-driven simulations of biological systems for pharmacological and industrial applications |
| US7763722B2 (en) | 2003-01-17 | 2010-07-27 | University Of Florida Research Foundation, Inc. | Small interference RNA gene therapy |
| WO2004087930A1 (en) | 2003-03-28 | 2004-10-14 | Saint Louis University | Adenovirus replication-competent vectors expressing trail |
| CA2530125A1 (en) | 2003-06-27 | 2005-01-06 | Atugen Ag | Use of double-stranded ribonucleic acid for inducing cell lysis |
| MXPA06008157A (es) | 2003-12-24 | 2007-09-07 | Johnson & Johnson | Tratamiento de gliomas malignos con inhibidores de factor de crecimiento transformante-beta. |
| US20050146081A1 (en) | 2004-01-07 | 2005-07-07 | Maclean David | Needle protection device with gauge specific color coding and method for manufacturing thereof |
| EP1765375A1 (en) | 2004-05-20 | 2007-03-28 | The Ludwig Institute for Cancer Research | Method for inhibiting angiogenesis and/or lymphangiogenesis |
| US7615618B2 (en) | 2004-06-30 | 2009-11-10 | Alnylam Pharmaceuticals, Inc. | Oligonucleotides comprising a non-phosphate backbone linkage |
| EP1797183B1 (en) | 2004-09-02 | 2012-08-01 | Yale University | Regulation of oncogenes by micrornas |
| US20080152633A1 (en) | 2005-02-16 | 2008-06-26 | The University Of Queensland | Flavivirus Replicon Constructs for Tumor Therapy |
| US20070179113A1 (en) * | 2005-05-19 | 2007-08-02 | Schering Aktiengesellachaft | GM-CSF gene therapy for Crohn's disease using an improved regulated expression system |
| US8053183B2 (en) | 2005-07-27 | 2011-11-08 | Oncotherapy Science, Inc. | Method of diagnosing esophageal cancer |
| US8603991B2 (en) | 2005-11-18 | 2013-12-10 | Gradalis, Inc. | Individualized cancer therapy |
| US8916530B2 (en) | 2005-11-18 | 2014-12-23 | Gradalis, Inc. | Individualized cancer therapy |
| US8906874B2 (en) | 2006-11-09 | 2014-12-09 | Gradalis, Inc. | Bi-functional shRNA targeting Stathmin 1 and uses thereof |
| US8252526B2 (en) | 2006-11-09 | 2012-08-28 | Gradalis, Inc. | ShRNA molecules and methods of use thereof |
| US8758998B2 (en) | 2006-11-09 | 2014-06-24 | Gradalis, Inc. | Construction of bifunctional short hairpin RNA |
| WO2008101087A1 (en) | 2007-02-14 | 2008-08-21 | Ontherex Llc | Compositions and methods for modulation of pdx-1 |
| WO2008144900A1 (en) * | 2007-05-25 | 2008-12-04 | Peel Sean A | A method of enhancing recombinant protein production |
| US20090004668A1 (en) | 2007-06-22 | 2009-01-01 | The Board Of Trustees Of The Leland Stanford Junior University | Pre-miRNA loop-modulated target regulation |
| WO2010008726A1 (en) | 2008-06-16 | 2010-01-21 | Immunogen Inc. | Novel synergistic effects |
| US20110045534A1 (en) * | 2009-08-20 | 2011-02-24 | Cell Signaling Technology, Inc. | Nucleic Acid Cassette For Producing Recombinant Antibodies |
| WO2011053660A2 (en) | 2009-10-30 | 2011-05-05 | Gradalis, Inc. | Novel therapeutic rna interference technology targeted to the pdx-1 oncogene in pdx-1 expressing neuroendocrine tumors |
| CN102917709B (zh) | 2009-12-23 | 2018-04-24 | 格兰达利斯有限公司 | 弗林蛋白酶敲减和gm-csf增强的(fang)癌症疫苗 |
| SG181881A1 (en) * | 2009-12-23 | 2012-07-30 | Gradalis Inc | Furin-knockdown bi-functional rna |
| US20110286979A1 (en) | 2010-05-20 | 2011-11-24 | Gradalis, Inc. | CHEMOSENSITIZATION BY BI-FUNCTIONAL SMALL HAIRPIN RNA (bi-shRNA) |
| SG11201407239SA (en) | 2012-05-09 | 2014-12-30 | Gradalis Inc | BI-FUNCTIONAL SHORT-HAIRPIN RNA (BI-SHRNA) SPECIFIC FOR SINGLE-NUCLEOTIDE <i>KRAS</i> MUTATIONS |
-
2010
- 2010-12-20 CN CN201080064642.8A patent/CN102917709B/zh active Active
- 2010-12-20 EP EP10840035.9A patent/EP2515916B1/en active Active
- 2010-12-20 AU AU2010333753A patent/AU2010333753B2/en active Active
- 2010-12-20 CA CA2785258A patent/CA2785258A1/en not_active Abandoned
- 2010-12-20 SG SG10201606680QA patent/SG10201606680QA/en unknown
- 2010-12-20 CN CN201810243419.8A patent/CN108295087B/zh active Active
- 2010-12-20 JP JP2012546121A patent/JP5553906B2/ja active Active
- 2010-12-20 US US12/973,823 patent/US9132146B2/en active Active
- 2010-12-20 KR KR1020127019530A patent/KR101548567B1/ko not_active IP Right Cessation
- 2010-12-20 SG SG2013011713A patent/SG188160A1/en unknown
- 2010-12-20 SG SG2012046330A patent/SG181880A1/en unknown
- 2010-12-20 WO PCT/US2010/061344 patent/WO2011079077A1/en active Application Filing
- 2010-12-20 KR KR1020147022999A patent/KR101589951B1/ko active IP Right Grant
- 2010-12-23 TW TW99145635A patent/TW201130975A/zh unknown
- 2010-12-23 AR ARP100104932 patent/AR081057A1/es unknown
- 2010-12-23 TW TW103127810A patent/TW201522632A/zh unknown
-
2012
- 2012-06-21 IL IL220589A patent/IL220589A0/en unknown
- 2012-09-07 US US13/606,476 patent/US20130078279A1/en not_active Abandoned
-
2014
- 2014-05-29 JP JP2014111779A patent/JP5820506B2/ja active Active
-
2015
- 2015-07-31 US US14/815,721 patent/US9790518B2/en active Active
-
2017
- 2017-08-29 US US15/690,092 patent/US10253331B2/en active Active
-
2019
- 2019-01-16 HK HK19100699.3A patent/HK1258331A1/zh unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62164695A (ja) * | 1985-12-21 | 1987-07-21 | ヘキスト、アクチエンゲゼルシヤフト | Gm−csfタンパク質、その誘導体、このタイプのタンパク質の調製及びそれらの用途 |
| US20070224194A1 (en) * | 2004-05-20 | 2007-09-27 | Ludwig Institute For Cancer Research | Methods for Inhibiting Angiogenesis and/or Lymphangiogenesis |
Non-Patent Citations (4)
| Title |
|---|
| JPN6013063989; Advanced Drug Delivery Reviews Vol.61, 200904, Vol.746-759 * |
| JPN6013063991; BioProcessing Journal Vol.8, No.1, 200902, p.30-36 * |
| JPN6013063992; Nature Vol.455, 2008, p.246-251 * |
| JPN6013063993; BioProcessing Journal Vol.8, No.4, 20100330, p.4-14 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017511128A (ja) * | 2014-03-26 | 2017-04-20 | トカジェン インコーポレーテッド | 免疫刺激活性を有するレトロウイルスベクター |
| JP2021509253A (ja) * | 2017-10-10 | 2021-03-25 | ナントバイオ,インコーポレイテッド | ウイルス生成ペイロードに対して低い毒性を有する改変ec7細胞 |
| US11485957B2 (en) | 2017-10-10 | 2022-11-01 | Nantbio, Inc. | Modified EC7 cells having low toxicity to viral production payloads |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5820506B2 (ja) | フューリンノックダウン及びgm−csf増強(fang)癌ワクチン | |
| TWI500764B (zh) | 弗林蛋白酶(furin)基因抑制之雙功能性rna | |
| CN111849905B (zh) | 间充质干细胞靶向运输趋化因子和细胞因子的免疫疗法 | |
| AU2015252009B2 (en) | Furin-knockdown and gm-csf-augmented (fang) cancer vaccine | |
| AU2013201841B2 (en) | Furin-knockdown and gm-csf-augmented (fang) cancer vaccine | |
| AU2016208280A1 (en) | Furin-knockdown and gm-csf-augmented (fang) cancer vaccine | |
| US20220175816A1 (en) | Aim2 inhibitors and uses thereof | |
| TWI432731B (zh) | 篩選治療因肺癌所引起樹突細胞功能低下之候選劑之方法 | |
| Harden | IFN-gamma is Central to Both Immunogenic and Tolerogenic Properties of Dendritic Cells After IL-12 and GM-CSF Microsphere Treatment |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140107 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140402 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20140507 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20140527 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5553906 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |