EP0353540B2 - Garniture pour portes coupe-feux comportant un sol de silice - Google Patents
Garniture pour portes coupe-feux comportant un sol de silice Download PDFInfo
- Publication number
- EP0353540B2 EP0353540B2 EP89113178A EP89113178A EP0353540B2 EP 0353540 B2 EP0353540 B2 EP 0353540B2 EP 89113178 A EP89113178 A EP 89113178A EP 89113178 A EP89113178 A EP 89113178A EP 0353540 B2 EP0353540 B2 EP 0353540B2
- Authority
- EP
- European Patent Office
- Prior art keywords
- layer
- wall element
- hydroxide
- element according
- fire
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 title claims abstract description 27
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 claims abstract description 29
- 239000011230 binding agent Substances 0.000 claims abstract description 24
- 239000000203 mixture Substances 0.000 claims abstract description 21
- 239000011490 mineral wool Substances 0.000 claims abstract description 15
- NTHWMYGWWRZVTN-UHFFFAOYSA-N sodium silicate Chemical compound [Na+].[Na+].[O-][Si]([O-])=O NTHWMYGWWRZVTN-UHFFFAOYSA-N 0.000 claims abstract description 14
- 235000019353 potassium silicate Nutrition 0.000 claims abstract description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 32
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical group [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 claims description 23
- 238000000034 method Methods 0.000 claims description 13
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 12
- 239000000463 material Substances 0.000 claims description 11
- 229920001282 polysaccharide Polymers 0.000 claims description 11
- 239000005017 polysaccharide Substances 0.000 claims description 11
- 230000008569 process Effects 0.000 claims description 10
- 150000004676 glycans Chemical class 0.000 claims description 9
- 238000012856 packing Methods 0.000 claims description 8
- 239000000126 substance Substances 0.000 claims description 8
- 238000001035 drying Methods 0.000 claims description 7
- 239000002245 particle Substances 0.000 claims description 7
- 238000004519 manufacturing process Methods 0.000 claims description 6
- 239000000377 silicon dioxide Substances 0.000 claims description 6
- 230000009970 fire resistant effect Effects 0.000 claims description 5
- 239000007787 solid Substances 0.000 claims description 5
- 239000003365 glass fiber Substances 0.000 claims description 4
- 229910010272 inorganic material Inorganic materials 0.000 claims description 4
- 239000011147 inorganic material Substances 0.000 claims description 4
- 239000004033 plastic Substances 0.000 claims description 4
- 239000005995 Aluminium silicate Substances 0.000 claims description 3
- 235000012211 aluminium silicate Nutrition 0.000 claims description 3
- 229920003086 cellulose ether Polymers 0.000 claims description 3
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 claims description 3
- 239000004927 clay Substances 0.000 claims description 2
- 239000002734 clay mineral Substances 0.000 claims description 2
- 239000000470 constituent Substances 0.000 claims description 2
- 229910001679 gibbsite Inorganic materials 0.000 claims description 2
- 229920003002 synthetic resin Polymers 0.000 claims description 2
- 239000000057 synthetic resin Substances 0.000 claims description 2
- -1 polysaccharide ethers Chemical class 0.000 claims 2
- 229910021502 aluminium hydroxide Inorganic materials 0.000 claims 1
- 229910052681 coesite Inorganic materials 0.000 claims 1
- 238000010924 continuous production Methods 0.000 claims 1
- 229910052906 cristobalite Inorganic materials 0.000 claims 1
- 229910052500 inorganic mineral Inorganic materials 0.000 claims 1
- 238000003780 insertion Methods 0.000 claims 1
- 230000037431 insertion Effects 0.000 claims 1
- 239000011707 mineral Substances 0.000 claims 1
- 229920005989 resin Polymers 0.000 claims 1
- 239000011347 resin Substances 0.000 claims 1
- 230000000979 retarding effect Effects 0.000 claims 1
- 229910052682 stishovite Inorganic materials 0.000 claims 1
- 229910052905 tridymite Inorganic materials 0.000 claims 1
- 230000007704 transition Effects 0.000 abstract description 3
- 238000009413 insulation Methods 0.000 description 31
- 230000003578 releasing effect Effects 0.000 description 15
- 238000006243 chemical reaction Methods 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 150000004679 hydroxides Chemical class 0.000 description 6
- 230000008901 benefit Effects 0.000 description 5
- YLGXILFCIXHCMC-JHGZEJCSSA-N methyl cellulose Chemical compound COC1C(OC)C(OC)C(COC)O[C@H]1O[C@H]1C(OC)C(OC)C(OC)OC1COC YLGXILFCIXHCMC-JHGZEJCSSA-N 0.000 description 5
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 4
- 239000010440 gypsum Substances 0.000 description 4
- 229910052602 gypsum Inorganic materials 0.000 description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 4
- 239000000155 melt Substances 0.000 description 4
- 239000004115 Sodium Silicate Substances 0.000 description 3
- 229910000831 Steel Inorganic materials 0.000 description 3
- 239000003513 alkali Substances 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 239000008187 granular material Substances 0.000 description 3
- 239000011777 magnesium Substances 0.000 description 3
- 235000019795 sodium metasilicate Nutrition 0.000 description 3
- 229910052911 sodium silicate Inorganic materials 0.000 description 3
- 239000010959 steel Substances 0.000 description 3
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 229910004298 SiO 2 Inorganic materials 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 125000000129 anionic group Chemical group 0.000 description 2
- 238000005336 cracking Methods 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 230000008030 elimination Effects 0.000 description 2
- 238000003379 elimination reaction Methods 0.000 description 2
- 239000003063 flame retardant Substances 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 239000012774 insulation material Substances 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- 239000006259 organic additive Substances 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 239000002002 slurry Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 1
- 239000010754 BS 2869 Class F Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 229910021486 amorphous silicon dioxide Inorganic materials 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000010425 asbestos Substances 0.000 description 1
- 229910001593 boehmite Inorganic materials 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- ODINCKMPIJJUCX-UHFFFAOYSA-N calcium oxide Inorganic materials [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 1
- 239000000292 calcium oxide Substances 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000011982 device technology Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000008240 homogeneous mixture Substances 0.000 description 1
- FAHBNUUHRFUEAI-UHFFFAOYSA-M hydroxidooxidoaluminium Chemical compound O[Al]=O FAHBNUUHRFUEAI-UHFFFAOYSA-M 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 235000014413 iron hydroxide Nutrition 0.000 description 1
- NCNCGGDMXMBVIA-UHFFFAOYSA-L iron(ii) hydroxide Chemical compound [OH-].[OH-].[Fe+2] NCNCGGDMXMBVIA-UHFFFAOYSA-L 0.000 description 1
- 238000003475 lamination Methods 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 239000002557 mineral fiber Substances 0.000 description 1
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 229910052895 riebeckite Inorganic materials 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000002341 toxic gas Substances 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000000844 transformation Methods 0.000 description 1
- 229920006186 water-soluble synthetic resin Polymers 0.000 description 1
Images
Classifications
-
- E—FIXED CONSTRUCTIONS
- E06—DOORS, WINDOWS, SHUTTERS, OR ROLLER BLINDS IN GENERAL; LADDERS
- E06B—FIXED OR MOVABLE CLOSURES FOR OPENINGS IN BUILDINGS, VEHICLES, FENCES OR LIKE ENCLOSURES IN GENERAL, e.g. DOORS, WINDOWS, BLINDS, GATES
- E06B5/00—Doors, windows, or like closures for special purposes; Border constructions therefor
- E06B5/10—Doors, windows, or like closures for special purposes; Border constructions therefor for protection against air-raid or other war-like action; for other protective purposes
- E06B5/16—Fireproof doors or similar closures; Adaptations of fixed constructions therefor
-
- E—FIXED CONSTRUCTIONS
- E04—BUILDING
- E04B—GENERAL BUILDING CONSTRUCTIONS; WALLS, e.g. PARTITIONS; ROOFS; FLOORS; CEILINGS; INSULATION OR OTHER PROTECTION OF BUILDINGS
- E04B1/00—Constructions in general; Structures which are not restricted either to walls, e.g. partitions, or floors or ceilings or roofs
- E04B1/62—Insulation or other protection; Elements or use of specified material therefor
- E04B1/74—Heat, sound or noise insulation, absorption, or reflection; Other building methods affording favourable thermal or acoustical conditions, e.g. accumulating of heat within walls
- E04B1/76—Heat, sound or noise insulation, absorption, or reflection; Other building methods affording favourable thermal or acoustical conditions, e.g. accumulating of heat within walls specifically with respect to heat only
- E04B1/78—Heat insulating elements
- E04B1/80—Heat insulating elements slab-shaped
-
- E—FIXED CONSTRUCTIONS
- E06—DOORS, WINDOWS, SHUTTERS, OR ROLLER BLINDS IN GENERAL; LADDERS
- E06B—FIXED OR MOVABLE CLOSURES FOR OPENINGS IN BUILDINGS, VEHICLES, FENCES OR LIKE ENCLOSURES IN GENERAL, e.g. DOORS, WINDOWS, BLINDS, GATES
- E06B3/00—Window sashes, door leaves, or like elements for closing wall or like openings; Layout of fixed or moving closures, e.g. windows in wall or like openings; Features of rigidly-mounted outer frames relating to the mounting of wing frames
- E06B3/70—Door leaves
- E06B3/7015—Door leaves characterised by the filling between two external panels
- E06B2003/7042—Door leaves characterised by the filling between two external panels with a fire retardant layer
Definitions
- the invention relates to a fire-resistant, heat transfer reducing wall element, in particular as an insert for a fire-retardant door, according to the preamble of claim 1 and a method for its production.
- the fire resistance is determined by the duration at which a certain temperature rise on one side of the wall element, the other side of the wall element below a certain limit temperature, for. B. 180 ° C remains.
- the service life of the wall element until this limit temperature is reached on the cold side in minutes gives the fire resistance class, whereby according to DIN 4102, Part 5 z. B. the classification in fire resistance class F 30 means a 30-minute service life, F 90 a 90-minute service life etc.
- thermal insulation measures alone can only achieve a limited delay in the rise in temperature on the cold side, and this requires relatively large wall thicknesses.
- thermal insulation materials with a correspondingly high thermal resistance on the one hand and sufficient temperature resistance on the other hand are not available, with the exception of asbestos, which should not be used due to its health-threatening effects.
- Mineral wool such as rock wool, sinters together from the hot side under the high fire temperatures and thus loses its effectiveness as a thermal insulation material relatively quickly on the hot side, so that relatively large wall thicknesses are required;
- mineral wool has a relatively low heat capacity and can therefore only delay the temperature rise on the cold side by its own heat absorption with small wall thicknesses.
- Gypsum in particular is in practical use as a material for a layer that can delay the rise in temperature on the cold side due to the storage of latent heat as a result of phase change.
- the relatively high enthalpy is used when the crystal water is split off, which occurs from approx. 50 ° C.
- gypsum can only be handled as a layer with the dimensions required in practice in the form of so-called gypsum plasterboard, with a layer of gypsum being laminated on both sides with cardboard.
- the cardboard lining naturally increases the fire load and promotes the formation of flammable smoldering and decomposition gases.
- DE-OS 30 23 632 sodium metasilicate has become known, a substance that melts at around 48 ° C. while absorbing heat in its own crystal water and is stable in the molten state.
- the melt with a relatively low viscosity is formed even at the lowest temperatures and, of course, tends to accumulate in the lower region of the available space.
- the hot upper area is completely exposed from the material of the layered fire protection zone, so that the temperature on the cold side rises practically abruptly there.
- the sodium metasilicate is to be embedded in an open-pore support structure made of a wettable material, for example granulated mineral wool, to avoid draining away too quickly.
- a generic fire resistant, heat transfer reducing wall element according to the preamble of claim 1 is known.
- the two insulation layers made of bound mineral wool are combined with at least one fire protection zone made of a granulate made of alkali or alkaline earth carbonate, the granulate being coated with an inorganic binder, e.g. B. magnesia binder.
- This known wall element offers sufficient resistance to a fire; however, since the material of the fire protection zone consists of alkali or alkaline earth carbonate in the form of granules, the fire protection zone is relatively wide, which leads to a relatively large structural thickness of the wall element. In addition, the known wall element is very heavy and prone to breakage.
- a method for producing the wall element is also known from this document.
- Another fire resistant wall element having the features of the preamble of claim 1 is known from GB-A-1 558 073.
- a layer of kaolin is provided as the inorganic material.
- the invention has for its object to provide a wall element of the type specified in the preamble of claim 1 and a method for its production, which in combination with mineral fiber insulation layers a high fire resistance class such as F 90 according to DIN 4102, part 5 in comparison with the stand the technology of reduced thickness and weight can easily achieve.
- Substances are used as hydroxides or binders to ensure that no flammable and / or toxic gases are generated in the event of a fire.
- As water-releasing hydroxides aluminum hydroxide Al (OH) 3 or z.
- Magnesium hydroxide Mg (OH) 2 , calcium hydroxide Ca (OH) 2 , iron hydroxide Fe (OH) 3 or FeO (OH) can be used, which with elimination of water in the corresponding oxides Al 2 O 3 , MgO, CaO, Fe 2 O 3 etc. are transferred.
- the conversion of the hydroxides into the corresponding oxides is an endothermic reaction, whereby heat is absorbed. Furthermore, water is released in a heat-consuming endothermic reaction, which escapes in the form of water vapor.
- the binder has the function of binding the water-releasing hydroxide on at least one insulation layer in the form of a layer.
- Aluminum hydroxide Al (OH) 3 (hydrargillite) changes to AIO (OH) (boehmite) when heated to approx. 150 ° C, which changes to gamma-aluminum oxide at approx. 400 ° C with elimination of water: 2 Al (OH) 3rd ---- ⁇ 2 AlO (OH) ---- ⁇ Al 2nd O 3rd
- aluminum hydroxide is available in a variety of different preparations, e.g. B. available as a martinal (registered trademark of the Martinswerk, Bergheim) on the market.
- the measure of using the water-releasing hydroxide in different fractions with at least two particle size ranges leads to a higher packing density of the hydroxide.
- increasing the packing density has the advantage that more mass with endothermic and water-releasing properties can be introduced into the layer and, on the other hand, that the proportion of the more expensive binder can be reduced if necessary.
- the increase in the packing density leads to a lower shrinkage of the layer during drying, as a result of which cracking can be largely suppressed.
- the average grain size of the finest fraction is less than 2 microns ensures that this fraction fits well into the packing gaps formed by the spherical packing of the coarsest fraction with an average grain size of less than 100 microns and in particular lower 30 microns and thereby a tightly packed Compound of the dehydrating hydroxide, e.g. B. the aluminum hydroxide is formed.
- Silica sol is a colloidal solution of amorphous silicon dioxide in water, to which small quantities of alkali are added to stabilize it in technical qualities.
- Commercial silica sols which occur as anionic or as cationic silica sols contain, for example, 15 to 45% by weight of solids, calculated as SiO 2 .
- An anionic silica sol with a solids content of 30 to 45% by weight, in particular 40% by weight, is preferably added to the layer between the insulating layers of bound mineral wool of the wall element according to the invention as silica sol.
- silica sol serves as a binder and also splits off water upon transition into the crosslinked gel state and upon aging and during heating, with the formation of silicon dioxide according to the following equation: Si (OH) 4th ---- ⁇ SiO (OH) 2nd ---- ⁇ SiO 2nd .
- Aluminum hydroxide in the form of the commercial article Martinal is inert in the pH range between 3.5 and 10.5 and is insoluble in acids and alkalis. Therefore the mixture of Al (OH) 3 (pH about 9.1) and silica sol (pH about 9 to 10) is stable.
- a water glass for example potassium or sodium water glass in aqueous solution
- a water glass for example potassium or sodium water glass in aqueous solution
- Such solutions are strongly basic so that they slowly attack aluminum hydroxide. If this reaction with water glass has a disruptive effect, Mg (OH) 2 , Ca (OH) 2 , Fe (OH) 3 or FeO (OH) or another hydroxide instead of aluminum hydroxide or a mixture of such hydroxides together with water glass can be used as the water-releasing hydroxide will.
- any water-releasing hydroxide for example Mg (OH) 2 , Ca (OH) 2 , Fe (OH) 3 , FeO (OH) or a mixture of such hydroxides can be used together with silica sol.
- a mass is made from a suitable water-releasing agent Hydroxide, e.g. B. aluminum hydroxide, and a silica sol processed into a plastic mass which is applied to one side of an insulation layer in a uniform layer.
- Hydroxide e.g. B. aluminum hydroxide
- silica sol processed into a plastic mass which is applied to one side of an insulation layer in a uniform layer.
- the silica sol solidifies in the layer over a period of 2 to 8 hours. It can e.g. B. can be significantly shortened by heating or microwave drying.
- the water from the damp mixture of hydroxide and a water glass or silica sol is distributed to a small extent in the underlying insulation layer and can evaporate over time.
- the strength of the layer is not dependent on complete removal of the water, since it is primarily due to the transition of the water glass or silica sol to the crosslinked gel state.
- a second layer of insulation can also be placed on top of the still wet layer and evenly bonded to the layer of plastic mass by applying slight pressure.
- the silica sol By transitioning the silica sol into the cross-linked gel state, the two insulation layers are irreversibly connected or glued to one another in this procedure.
- the binder is an organic substance, such as a polysaccharide, a polysaccharide derivative, in particular a polysaccharide ether, preferably a cellulose ether, such as. B. Tylose (registered trademark) or adding a synthetic resin has the advantage that drastic savings in binder can be achieved by this measure if necessary.
- the organic additive for example tylose, even if it is used in fire-irrelevant concentrations of far below 1% by weight, based on the mixture of hydroxide and binder, still has such a bondability or adhesive strength of the hydroxide / binder Mixture has the effect that the usual binder concentration can be reduced by a factor of approx. 2 to 5. This results in an economical saving of binders, especially the relatively expensive silica brine.
- the wall element produced in this way has sufficient fire resistance for normal applications. If there is an increased requirement for fire resistance, one or more insulation layers can be stacked in the same way with the wall element produced in this way.
- the thickest possible insulation layers made from bound mineral wool can be used.
- the thickness of the insulation layers is limited by the prescribed maximum thickness of fire protection doors and by the high weight of fire protection doors with very thick insulation layers.
- panels with a thickness of 20 to 40 mm, in particular 30 mm and a bulk density of 140 to 280 kg / m 2 , in particular 200 kg / m 2 have proven to be good insulation layers.
- the layer of inorganic material is applied in a thickness of 2 to 5 mm, in particular 3 mm, so that the layer 3 has a basis weight of 5 to 10 kg / m 2 , in particular 7 to 8 kg / m 2 .
- the outer insulation layer made of mineral wool facing the respective fire initially insulates the layer of inorganic material, which also serves as a fire protection zone, and protects it from direct flame access, so that the latter is only later exposed to the full flame effect .
- the second insulation layer made of mineral wool remains, which is arranged on the cold side in order to effect the required insulation on the cold side. This insulation layer is protected by the layer acting as a fire protection zone and can therefore fully develop its insulation effect over a long period of time.
- the layer has a nonwoven, in particular a nonwoven made of glass fibers, the cracking in the layer of water-releasing hydroxide and binder during the drying process and in particular during rapid drying, for. B. reduced or prevented under 4 min at 600 ° C.
- the fleece is integrated into the solidifying mass so that a heterogeneous layer is formed which contains water-releasing hydroxide, e.g. B. aluminum hydroxide, inorganic binder, e.g. B. contains silica sol and the fleece.
- Another advantage of this measure is that the layer has a reinforcement due to the fleece lying on it, which improves handling during production.
- rock wool as mineral wool has the advantage that it is very temperature-resistant on the one hand and, on the other hand, inexpensive in the required packaging, e.g. B. is to be produced as a plate.
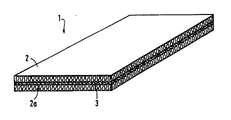
- the figure shows a view of the wall element according to the invention.
- the wall element 1 consists of two insulation layers 2, 2a made of mineral wool, between which a layer 3, made of a mixture of a water-releasing hydroxide and a silica sol, is arranged.
- Layer 3 serves as a fire protection layer since, in the event of a fire, it converts to aluminum oxide or silicon dioxide when heated and not only releases water vapor but also a large one during this endothermic conversion
- the layer 3 forms a tight protective shield for the insulation layer 2 or 2a, which faces away from the fire. While the insulation layer facing the fire sinters together relatively quickly under the action of heat, layer 3 delays the sintering together of the insulation layer 2a or 2 facing away from the fire, as a result of which a rapid rise in temperature on the cold side can be avoided.
- the wall element according to the invention serves in particular as an insert for a fire-retardant door.
- the wall element 1 can be prefabricated on the one hand in the form of a double-layer plate sandwich and inserted between the sheet steel shells of a fire protection door.
- aluminum hydroxide is used as the water-releasing hydroxide in two fractions with different particle size ranges and silica sol as the inorganic binder.
- silica sol for example, dissolved water glass can also be used as the inorganic binder.
- the ingredients are mixed e.g. B. in a screw conveyor and brings the homogeneous mixture obtained onto one of the two insulation layers in a uniform layer. Then layer 3 is dried or, if necessary, the second insulation layer is applied to the still moist layer, pressed on and dried.
- tylose is used as an organic additive
- a further advantageous embodiment uses a glass fiber fleece to reinforce layer 3, which for example has a weight of 50 g / m 2 .
- the fleece is on the layer 3 of aluminum hydroxide and binder z. B. applied by rolling, the fleece is connected to the layer 3 after drying and curing.
- the use of a fleece applied to the hydroxide mixture reduces the formation of cracks, especially when the layer 3 is subjected to rapid drying, e.g. B. 3 minutes at 600 ° C.
- the embodiment of the process according to the invention is particularly economical since the process can be carried out continuously or semi-continuously and the mixture can be produced in accordance with its consumption for the production of the layer.
Landscapes
- Engineering & Computer Science (AREA)
- Architecture (AREA)
- Civil Engineering (AREA)
- Structural Engineering (AREA)
- Physics & Mathematics (AREA)
- Electromagnetism (AREA)
- Acoustics & Sound (AREA)
- Building Environments (AREA)
- Laminated Bodies (AREA)
- Special Wing (AREA)
- Road Signs Or Road Markings (AREA)
- Securing Of Glass Panes Or The Like (AREA)
- Joining Of Glass To Other Materials (AREA)
- Silicon Compounds (AREA)
Claims (19)
- Elément de cloison résistant au feu, réduisant la transmission de chaleur, notamment à incorporer dans une porte coupe-feu, comportant au moins deux couches isolantes (2, 2a) en laine minérale agglomérée, entre lesquelles est disposée une couche (3) constituée d'un mélange de matériaux au moins principalement minéraux,
caractérisé en ce quela couche (3) est formée d'un hydroxyde déshydratant et d'un liant ;l'hydroxyde déshydratant étant utilisé en fractions différentes présentant au moins deux domaines granulométriques. - Elément de cloison selon la revendication 1, caractérisé en ce que l'hydroxyde déshydratant est de l'hydroxyde d'aluminium.
- Elément de cloison selon la revendication 1 ou 2, caractérisé en ce que la taille moyenne des grains de la fraction la plus fine est inférieure à 2 µm.
- Elément de cloison selon la revendication 1 ou 2, caractérisé en ce que la taille moyenne des grains de la fraction la plus grosse est inférieure à 100 µm et est de préférence inférieure à 30 µm.
- Elément de cloison selon l'une des revendications 1 à 4, caractérisé en ce que le liant est du sol siliceux et/ou du verre soluble.
- Elément de cloison selon l'une des revendications 1 à 5, caractérisé en ce que le liant contient un additif organique, sélectionné dans le groupe constitué des polysaccharides, des dérivés de polysaccharides, notamment des éthers de polysaccharides, de préférence des éthers de cellulose et des résines synthétiques, en une concentration insignifiante du point de vue du comportement au feu.
- Elément de cloison selon l'une des revendications 1 à 6, caractérisé en ce que la couche est constituée par un mélange de 70 à 95 % en poids d'hydroxyde d'aluminium, calculé en tant que Al(OH)3, et 30 à 5 % en poids de sol siliceux, calculé en tant que SiO2.
- Elément de cloison selon la revendication 7, caractérisé en ce que le mélange est constitué d'environ 85 parties en poids d'hydroxyde d'aluminium et d'environ 15 parties en poids de sol siliceux.
- Elément de cloison selon l'une des revendications 1 à 8, caractérisé en ce que le mélange contient, au lieu d'une partie de l'hydroxyde déshydratant ou en plus de l'oxyde déshydratant, une argile ou un minéral à base d'argile réfractaire.
- Elément de cloison selon l'une des revendications 1 à 9, caractérisé en ce que la couche est d'une épaisseur inférieure à celle de chaque couche isolante.
- Elément de cloison selon la revendication 10, caractérisé en ce que la couche est d'une épaisseur de 1 à 5 mm, notamment de 3 mm.
- Elément de cloison selon la revendication 10 ou 11, caractérisé en ce que la couche est un non tissé, notamment un non tissé de fibres de verre.
- Elément de cloison selon l'une des revendications 1 à 12, caractérisé en ce que la laine minérale est de la laine de roche.
- Procédé de fabrication d'un élément de cloison résistant au feu, réduisant la transmission de chaleur selon la revendication 1,
caractérisé en ce queselon un processus continu, on ajoute un liant à un hydroxyde déshydratant, sous forme solide ou en suspension aqueuse, auquel cas on utilise l'hydroxyde en fractions différentes avec au moins deux domaines granulométriques pour obtenir une densité de tassement supérieure ;on mélange les deux composants ;on applique le mélange obtenu sous la forme d'une couche uniforme (3) sur l'une des deux couches isolantes (2, 2a) ; eton laisse le liant durcir par séchage et on applique la seconde couche isolante (2a) sur la couche durcie (3) ou on applique la seconde couche isolante (2a) sur la couche (3) encore humide et on laisse durcir le liant. - Procédé selon la revendication 14, caractérisé en ce qu'on utilise comme liant du sol siliceux ou du verre soluble dissous.
- Procédé selon la revendication 14 ou 15, caractérisé en ce qu'on ajoute au liant une substance organique du groupe constitué par des polysaccharides, des dérivés de polysaccharides, notamment des éthers de polysaccharides, de préférence des éthers de cellulose et des résines synthétiques, en une concentration insignifiante du point de vue du comportement au feu.
- Procédé selon la revendication 14, caractérisé en ce qu'une fraction la plus fine ayant une taille de grains inférieure à 100 µm et de préférence inférieure à 30 µm.
- Procédé selon la revendication 14, caractérisé en ce qu'on utilise une fraction la plus grosse avec une taille de grains moyenne inférieure à 100 µm et de préférence inférieure à 30 µm.
- Procédé selon l'une des revendications 14 à 18, caractérisé en ce que la couche est armée d'un non tissé, notamment d'un non tissé de fibres de verre.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE3824598 | 1988-07-19 | ||
| DE3824598A DE3824598A1 (de) | 1988-07-19 | 1988-07-19 | Einlage fuer feuerschutztueren mit kieselsol |
Publications (4)
| Publication Number | Publication Date |
|---|---|
| EP0353540A2 EP0353540A2 (fr) | 1990-02-07 |
| EP0353540A3 EP0353540A3 (fr) | 1991-12-11 |
| EP0353540B1 EP0353540B1 (fr) | 1994-10-05 |
| EP0353540B2 true EP0353540B2 (fr) | 1997-12-29 |
Family
ID=6359119
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP89113178A Expired - Lifetime EP0353540B2 (fr) | 1988-07-19 | 1989-07-18 | Garniture pour portes coupe-feux comportant un sol de silice |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP0353540B2 (fr) |
| AT (1) | ATE112601T1 (fr) |
| DE (2) | DE3824598A1 (fr) |
| DK (1) | DK354289A (fr) |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE4036088A1 (de) * | 1990-11-13 | 1992-05-14 | Gruenzweig & Hartmann | Brandschutzmittel aus metallhydroxid und magnesiabinder, sowie dessen verwendung |
| DE4313820C2 (de) * | 1993-04-27 | 2003-04-10 | Woellner Silikat Gmbh | Beschichtungsmasse und deren Verwendung |
| DE4338619C5 (de) * | 1993-11-11 | 2007-12-27 | Saint-Gobain Isover G+H Ag | Beschichtetes Mineralwolleprodukt und Verfahren zu dessen Herstellung |
| NL9400839A (nl) * | 1994-05-24 | 1996-01-02 | Isobouw Systems Bv | Paneel. |
| DE29507498U1 (de) * | 1995-05-05 | 1995-07-13 | SAINT-GOBAIN ISOVER G+H AG, 67059 Ludwigshafen | Brandschutzelement mit Schichtstruktur, insbesondere als Einlage für Feuerschutztüren, sowie Halbzeug hierfür |
| ES2102969B1 (es) * | 1995-06-27 | 1998-04-01 | Andreu Barbera Jose Vicente | Aislamiento perfeccionado para puertas cortafuegos |
| DE19525961A1 (de) * | 1995-07-17 | 1997-01-23 | Webu Spezialtueren Gmbh | Brandschutztür in Schichtaufbau |
| DE19546980C2 (de) * | 1995-12-15 | 1999-08-19 | Gruenzweig & Hartmann | Brandschutzelement |
| DE29915463U1 (de) * | 1999-09-03 | 2001-01-18 | Deutsche Rockwool Mineralwoll-Gmbh, 45966 Gladbeck | Dämmelement zur Wärme- und/oder Schalldämmung von Gebäudewänden |
| DE19952931A1 (de) * | 1999-11-03 | 2001-05-10 | Saint Gobain Isover G & H Ag | Gebundenes Mineralwolleprodukt mit Feuerschutzfunktion sowie Brandschutzelement mit dem gebundenen Mineralwolleprodukt |
| DE20119043U1 (de) | 2001-02-21 | 2002-08-08 | Deutsche Rockwool Mineralwoll GmbH & Co. oHG, 45966 Gladbeck | Brandschutzelement, insbesondere für Feuerschutztüren |
| EP1239093A3 (fr) * | 2001-02-21 | 2003-08-06 | Deutsche Rockwool Mineralwoll GmbH & Co. OHG | Elément stratifié de protection contre le feu, en particulier pour portes coupe-feu |
| DE10212332B4 (de) * | 2002-03-20 | 2004-02-12 | Deutsche Rockwool Mineralwoll Gmbh & Co. Ohg | Brandschutzelement, insbesondere für Feuerschutztüren |
| DE10212331A1 (de) * | 2002-03-20 | 2003-10-16 | Rockwool Mineralwolle | Brandschutzelement, insbesondere für Feuerschutztüren |
| BE1014759A6 (nl) * | 2002-04-15 | 2004-03-02 | Wienerberger Bricks N V | Werkwijze voor het prefabriceren van een wandelement. |
| ITVI20060160A1 (it) * | 2006-05-25 | 2007-11-26 | Giampaolo Nelzi | Portone tagliafuoco ad apertura verticale e/o orizzontale |
| ATE404749T1 (de) * | 2006-07-19 | 2008-08-15 | Pavatex Sa | Holzweichfaserplatte für die raumseitige dämmung |
| CH706060B1 (de) * | 2012-01-16 | 2015-09-15 | Rwd Schlatter Ag | Türschichtplatte, Tür, Verwendung der Tür und Bausatz. |
| ITMO20120297A1 (it) * | 2012-11-28 | 2014-05-29 | Basic & Co Srl | Porta antincendio |
| NL2016698B1 (nl) * | 2016-04-29 | 2017-11-20 | M J Vroegop Holding B V | Brandwerend paneel. |
| US20230282924A1 (en) * | 2022-03-01 | 2023-09-07 | GM Global Technology Operations LLC | Intumescent inorganic composites for mitigating a thermal runaway event in a battery |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE7321823U (de) * | 1973-09-27 | Bolliger W | Brandschutz-Bauelement | |
| DK364375A (da) * | 1975-08-12 | 1977-02-13 | Rockwool As | Branddroj plade |
| DE3220821C2 (de) * | 1982-06-03 | 1985-02-07 | Karl 6902 Sandhausen Serwane | Brandgeschützte Dichtungsbahn |
| DE3510935A1 (de) * | 1985-03-26 | 1986-10-09 | Grünzweig + Hartmann und Glasfaser AG, 6700 Ludwigshafen | Feuerwiderstandsfaehiges, den waermedurchgang reduzierendes wandelement, insbesondere als einlage fuer eine feuerhemmende tuer, sowie verfahren zu seiner herstellung und hiermit ausgestattete feuerhemmende tuer |
| DE3540524A1 (de) * | 1985-11-15 | 1987-05-27 | Bayer Ag | Fuellstoffhaltige intumeszenzmassen auf epoxidharzbasis |
| DE3607839A1 (de) * | 1986-03-10 | 1987-09-17 | Paul Couwenbergs | Herstellung und verfahren einer doppelseitigen brand-, isolier-, abschottungs-, schallabsorbierungs-platte fuer waende und decken, sowie fuer kabelabschottung - mit einsatz von feuerverzinkten allzweck-halterungen |
-
1988
- 1988-07-19 DE DE3824598A patent/DE3824598A1/de not_active Withdrawn
-
1989
- 1989-07-18 DK DK354289A patent/DK354289A/da not_active Application Discontinuation
- 1989-07-18 EP EP89113178A patent/EP0353540B2/fr not_active Expired - Lifetime
- 1989-07-18 AT AT89113178T patent/ATE112601T1/de not_active IP Right Cessation
- 1989-07-18 DE DE58908472T patent/DE58908472D1/de not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| DE3824598A1 (de) | 1990-01-25 |
| DK354289A (da) | 1990-01-20 |
| EP0353540A2 (fr) | 1990-02-07 |
| EP0353540B1 (fr) | 1994-10-05 |
| ATE112601T1 (de) | 1994-10-15 |
| EP0353540A3 (fr) | 1991-12-11 |
| DK354289D0 (da) | 1989-07-18 |
| DE58908472D1 (de) | 1994-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0353540B2 (fr) | Garniture pour portes coupe-feux comportant un sol de silice | |
| EP0114965B1 (fr) | Plaques d'isolation revêtues pour façades et toitures à base de fibres minérales et procédé pour leur fabrication | |
| EP0485867B1 (fr) | Composition retardratice de feu à base d'hydroxyde métallique et de liant magnésien et utilisation | |
| DE60009853T2 (de) | Binderzusammensetzungen zum Binden von teilchenförmigem Material | |
| DE2725779A1 (de) | Selbsthaertende verbindung fuer eine feuerhemmende schicht | |
| EP0956277B1 (fr) | Materiau leger contenant de la perlite soufflee et procedes permettant de le produire | |
| EP1097807B1 (fr) | Produit pour la protection contre le feu à base de laine minérale lié et élément pour la protection contre le feu comprenant ledit produit | |
| EP0741003B1 (fr) | Elément stratifié de protection contre le feu, en particulier comme pièce intercalaire pour portes coupe-feu ainsi que le demi-produit utilisé à cet effet | |
| DE3325467A1 (de) | Mit einem waermeisolierenden material beschichtetes zellulaeres glas | |
| DE2842858A1 (de) | Verbundplatte mit zwei deckschichten und einem dazwischenliegenden kern | |
| AT391107B (de) | Verbundbauteil, bestehend aus mindestens zwei teilen aus unterschiedlichen fasermaterialien | |
| DE69627897T2 (de) | Wärmedammende bauteile | |
| DE4401983C2 (de) | Dämmstoffmaterial aus Stroh, dessen Herstellung und Verwendung | |
| DE2831505C2 (de) | Hitzebeständiger, exothermer, wärmeisolierender Gegenstand, seine Verwendung und Verfahren zu seiner Herstellung | |
| DE3624736A1 (de) | Brandschutzdaemmung auf mineralfaserbasis sowie verfahren zur herstellung der brandschutzdaemmung | |
| DD213915A5 (de) | Verfahren zur herstellung von plastischen leichtmassen zur weiterverarbeitung zu feuerbestaendigen und feuerfesten materialien | |
| DE3202488C2 (fr) | ||
| EP0310138A1 (fr) | Elément de construction et procédé de production | |
| DE2217315B2 (de) | Verspritzbare feuerfeste masse und verfahren zu ihrer herstellung | |
| EP0829459A1 (fr) | Plaques calorifuges incombustibles à base de granules de perlite expansée | |
| DE3248664A1 (de) | Beschichtete fassaden- oder dachdaemmplatte aus mineralfasern sowie verfahren zu ihrer herstellung | |
| CH691541A5 (de) | Feuerbeständiges Stoffgemenge. | |
| DE4313435A1 (de) | Wasserfest beschichtete Dämmplatte | |
| DE4313820C2 (de) | Beschichtungsmasse und deren Verwendung | |
| DE3346120C2 (de) | Geblähter Perlit |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE CH DE ES FR GB IT LI NL SE |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| RHK1 | Main classification (correction) |
Ipc: E04B 1/94 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH DE ES FR GB IT LI NL SE |
|
| 17P | Request for examination filed |
Effective date: 19920214 |
|
| 17Q | First examination report despatched |
Effective date: 19930319 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE ES FR GB IT LI NL SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRE;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED.SCRIBED TIME-LIMIT Effective date: 19941005 Ref country code: ES Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19941005 Ref country code: BE Effective date: 19941005 Ref country code: NL Effective date: 19941005 Ref country code: GB Effective date: 19941005 Ref country code: FR Effective date: 19941005 |
|
| REF | Corresponds to: |
Ref document number: 112601 Country of ref document: AT Date of ref document: 19941015 Kind code of ref document: T |
|
| REF | Corresponds to: |
Ref document number: 58908472 Country of ref document: DE Date of ref document: 19941110 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Effective date: 19950105 |
|
| EN | Fr: translation not filed | ||
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| GBV | Gb: ep patent (uk) treated as always having been void in accordance with gb section 77(7)/1977 [no translation filed] |
Effective date: 19941005 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Effective date: 19950718 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Effective date: 19950731 Ref country code: LI Effective date: 19950731 |
|
| 26 | Opposition filed |
Opponent name: ROCKWOOL INTERNATIONAL A/S Effective date: 19950630 |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PUAH | Patent maintained in amended form |
Free format text: ORIGINAL CODE: 0009272 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT MAINTAINED AS AMENDED |
|
| 27A | Patent maintained in amended form |
Effective date: 19971229 |
|
| AK | Designated contracting states |
Kind code of ref document: B2 Designated state(s): AT BE CH DE ES FR GB IT LI NL SE |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: AEN Free format text: AUFRECHTERHALTUNG DES PATENTES IN GEAENDERTER FORM |
|
| EN | Fr: translation not filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20020806 Year of fee payment: 14 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040203 |