WO2022210045A1 - シーラントシート - Google Patents
シーラントシート Download PDFInfo
- Publication number
- WO2022210045A1 WO2022210045A1 PCT/JP2022/012754 JP2022012754W WO2022210045A1 WO 2022210045 A1 WO2022210045 A1 WO 2022210045A1 JP 2022012754 W JP2022012754 W JP 2022012754W WO 2022210045 A1 WO2022210045 A1 WO 2022210045A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- thiol
- sealant sheet
- less
- sealant
- compound
- Prior art date
Links
- 239000000565 sealant Substances 0.000 title claims abstract description 288
- 229920001021 polysulfide Polymers 0.000 claims abstract description 186
- 125000003396 thiol group Chemical group [H]S* 0.000 claims abstract description 139
- -1 thiol compound Chemical class 0.000 claims abstract description 138
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 claims abstract description 41
- 239000000945 filler Substances 0.000 claims description 48
- 230000001588 bifunctional effect Effects 0.000 claims description 37
- 238000003860 storage Methods 0.000 claims description 33
- 239000003999 initiator Substances 0.000 claims description 20
- 239000004615 ingredient Substances 0.000 claims description 5
- 150000008117 polysulfides Polymers 0.000 abstract description 55
- 239000005077 polysulfide Substances 0.000 abstract description 54
- 239000004593 Epoxy Substances 0.000 description 94
- 150000001875 compounds Chemical class 0.000 description 84
- 125000003700 epoxy group Chemical group 0.000 description 76
- 239000000047 product Substances 0.000 description 57
- 150000003573 thiols Chemical class 0.000 description 40
- 238000006243 chemical reaction Methods 0.000 description 34
- 239000003822 epoxy resin Substances 0.000 description 34
- 229920000647 polyepoxide Polymers 0.000 description 34
- 239000007788 liquid Substances 0.000 description 33
- QTBSBXVTEAMEQO-UHFFFAOYSA-N acetic acid Substances CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 27
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 19
- 238000010438 heat treatment Methods 0.000 description 19
- 238000007789 sealing Methods 0.000 description 19
- 125000002228 disulfide group Chemical group 0.000 description 18
- 239000000203 mixture Substances 0.000 description 18
- 239000003054 catalyst Substances 0.000 description 14
- 239000000463 material Substances 0.000 description 14
- 238000002156 mixing Methods 0.000 description 13
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 12
- 238000000034 method Methods 0.000 description 12
- 239000002245 particle Substances 0.000 description 12
- 239000007795 chemical reaction product Substances 0.000 description 11
- 229920000642 polymer Polymers 0.000 description 11
- 238000007342 radical addition reaction Methods 0.000 description 10
- 230000000694 effects Effects 0.000 description 9
- 229920003986 novolac Polymers 0.000 description 9
- 239000003504 photosensitizing agent Substances 0.000 description 9
- 238000002834 transmittance Methods 0.000 description 9
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 8
- 238000002360 preparation method Methods 0.000 description 8
- 229920002799 BoPET Polymers 0.000 description 7
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- 239000003795 chemical substances by application Substances 0.000 description 7
- 238000005516 engineering process Methods 0.000 description 7
- 238000004519 manufacturing process Methods 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- 150000003254 radicals Chemical class 0.000 description 7
- 125000002947 alkylene group Chemical group 0.000 description 6
- 125000004432 carbon atom Chemical group C* 0.000 description 6
- 230000000052 comparative effect Effects 0.000 description 6
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 6
- 238000000465 moulding Methods 0.000 description 6
- 239000003381 stabilizer Substances 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 239000000454 talc Substances 0.000 description 6
- 229910052623 talc Inorganic materials 0.000 description 6
- 238000007259 addition reaction Methods 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 239000011521 glass Substances 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 239000002184 metal Substances 0.000 description 5
- 229920005989 resin Polymers 0.000 description 5
- 239000011347 resin Substances 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- OXBLVCZKDOZZOJ-UHFFFAOYSA-N 2,3-Dihydrothiophene Chemical compound C1CC=CS1 OXBLVCZKDOZZOJ-UHFFFAOYSA-N 0.000 description 4
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 4
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 4
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 4
- 150000001642 boronic acid derivatives Chemical class 0.000 description 4
- 238000009826 distribution Methods 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 238000010943 off-gassing Methods 0.000 description 4
- 239000003960 organic solvent Substances 0.000 description 4
- DPDQQGBQYIBXDX-UHFFFAOYSA-N 2,4,6-tris(aminomethyl)phenol Chemical compound NCC1=CC(CN)=C(O)C(CN)=C1 DPDQQGBQYIBXDX-UHFFFAOYSA-N 0.000 description 3
- HCZMHWVFVZAHCR-UHFFFAOYSA-N 2-[2-(2-sulfanylethoxy)ethoxy]ethanethiol Chemical compound SCCOCCOCCS HCZMHWVFVZAHCR-UHFFFAOYSA-N 0.000 description 3
- 229910000737 Duralumin Inorganic materials 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 239000004305 biphenyl Substances 0.000 description 3
- 235000010290 biphenyl Nutrition 0.000 description 3
- 239000002131 composite material Substances 0.000 description 3
- 125000000524 functional group Chemical group 0.000 description 3
- 230000014759 maintenance of location Effects 0.000 description 3
- 229920000728 polyester Polymers 0.000 description 3
- 239000002243 precursor Substances 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- 229910052719 titanium Inorganic materials 0.000 description 3
- DXBHBZVCASKNBY-UHFFFAOYSA-N 1,2-Benz(a)anthracene Chemical compound C1=CC=C2C3=CC4=CC=CC=C4C=C3C=CC2=C1 DXBHBZVCASKNBY-UHFFFAOYSA-N 0.000 description 2
- NQMUGNMMFTYOHK-UHFFFAOYSA-N 1-methoxynaphthalene Chemical compound C1=CC=C2C(OC)=CC=CC2=C1 NQMUGNMMFTYOHK-UHFFFAOYSA-N 0.000 description 2
- QPUYECUOLPXSFR-UHFFFAOYSA-N 1-methylnaphthalene Chemical compound C1=CC=C2C(C)=CC=CC2=C1 QPUYECUOLPXSFR-UHFFFAOYSA-N 0.000 description 2
- KJCVRFUGPWSIIH-UHFFFAOYSA-N 1-naphthol Chemical compound C1=CC=C2C(O)=CC=CC2=C1 KJCVRFUGPWSIIH-UHFFFAOYSA-N 0.000 description 2
- IMQFZQVZKBIPCQ-UHFFFAOYSA-N 2,2-bis(3-sulfanylpropanoyloxymethyl)butyl 3-sulfanylpropanoate Chemical compound SCCC(=O)OCC(CC)(COC(=O)CCS)COC(=O)CCS IMQFZQVZKBIPCQ-UHFFFAOYSA-N 0.000 description 2
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 2
- BTJPUDCSZVCXFQ-UHFFFAOYSA-N 2,4-diethylthioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC(CC)=CC(CC)=C3SC2=C1 BTJPUDCSZVCXFQ-UHFFFAOYSA-N 0.000 description 2
- GJKGAPPUXSSCFI-UHFFFAOYSA-N 2-Hydroxy-4'-(2-hydroxyethoxy)-2-methylpropiophenone Chemical compound CC(C)(O)C(=O)C1=CC=C(OCCO)C=C1 GJKGAPPUXSSCFI-UHFFFAOYSA-N 0.000 description 2
- NLGDWWCZQDIASO-UHFFFAOYSA-N 2-hydroxy-1-(7-oxabicyclo[4.1.0]hepta-1,3,5-trien-2-yl)-2-phenylethanone Chemical compound OC(C(=O)c1cccc2Oc12)c1ccccc1 NLGDWWCZQDIASO-UHFFFAOYSA-N 0.000 description 2
- XMLYCEVDHLAQEL-UHFFFAOYSA-N 2-hydroxy-2-methyl-1-phenylpropan-1-one Chemical compound CC(C)(O)C(=O)C1=CC=CC=C1 XMLYCEVDHLAQEL-UHFFFAOYSA-N 0.000 description 2
- GCDBEYOJCZLKMC-UHFFFAOYSA-N 2-hydroxyanthraquinone Chemical compound C1=CC=C2C(=O)C3=CC(O)=CC=C3C(=O)C2=C1 GCDBEYOJCZLKMC-UHFFFAOYSA-N 0.000 description 2
- QIMMUPPBPVKWKM-UHFFFAOYSA-N 2-methylnaphthalene Chemical compound C1=CC=CC2=CC(C)=CC=C21 QIMMUPPBPVKWKM-UHFFFAOYSA-N 0.000 description 2
- JWAZRIHNYRIHIV-UHFFFAOYSA-N 2-naphthol Chemical compound C1=CC=CC2=CC(O)=CC=C21 JWAZRIHNYRIHIV-UHFFFAOYSA-N 0.000 description 2
- VVBLNCFGVYUYGU-UHFFFAOYSA-N 4,4'-Bis(dimethylamino)benzophenone Chemical compound C1=CC(N(C)C)=CC=C1C(=O)C1=CC=C(N(C)C)C=C1 VVBLNCFGVYUYGU-UHFFFAOYSA-N 0.000 description 2
- 229910000838 Al alloy Inorganic materials 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- NQSMEZJWJJVYOI-UHFFFAOYSA-N Methyl 2-benzoylbenzoate Chemical compound COC(=O)C1=CC=CC=C1C(=O)C1=CC=CC=C1 NQSMEZJWJJVYOI-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- LULCPJWUGUVEFU-UHFFFAOYSA-N Phthiocol Natural products C1=CC=C2C(=O)C(C)=C(O)C(=O)C2=C1 LULCPJWUGUVEFU-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 235000000126 Styrax benzoin Nutrition 0.000 description 2
- 244000028419 Styrax benzoin Species 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 235000008411 Sumatra benzointree Nutrition 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- JOBBTVPTPXRUBP-UHFFFAOYSA-N [3-(3-sulfanylpropanoyloxy)-2,2-bis(3-sulfanylpropanoyloxymethyl)propyl] 3-sulfanylpropanoate Chemical compound SCCC(=O)OCC(COC(=O)CCS)(COC(=O)CCS)COC(=O)CCS JOBBTVPTPXRUBP-UHFFFAOYSA-N 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 239000005354 aluminosilicate glass Substances 0.000 description 2
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 2
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 2
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 description 2
- 150000004056 anthraquinones Chemical class 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 229960002130 benzoin Drugs 0.000 description 2
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 2
- 239000012965 benzophenone Substances 0.000 description 2
- 229910000019 calcium carbonate Inorganic materials 0.000 description 2
- 125000002837 carbocyclic group Chemical group 0.000 description 2
- 239000004918 carbon fiber reinforced polymer Substances 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- ISAOCJYIOMOJEB-UHFFFAOYSA-N desyl alcohol Natural products C=1C=CC=CC=1C(O)C(=O)C1=CC=CC=C1 ISAOCJYIOMOJEB-UHFFFAOYSA-N 0.000 description 2
- 239000000975 dye Substances 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 238000005227 gel permeation chromatography Methods 0.000 description 2
- 235000019382 gum benzoic Nutrition 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 230000001965 increasing effect Effects 0.000 description 2
- 230000001678 irradiating effect Effects 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- QWVGKYWNOKOFNN-UHFFFAOYSA-N o-cresol Chemical compound CC1=CC=CC=C1O QWVGKYWNOKOFNN-UHFFFAOYSA-N 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 150000003014 phosphoric acid esters Chemical class 0.000 description 2
- 230000000704 physical effect Effects 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 239000004587 polysulfide sealant Substances 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- IATRAKWUXMZMIY-UHFFFAOYSA-N strontium oxide Chemical compound [O-2].[Sr+2] IATRAKWUXMZMIY-UHFFFAOYSA-N 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- QNODIIQQMGDSEF-UHFFFAOYSA-N (1-hydroxycyclohexyl)-phenylmethanone Chemical compound C=1C=CC=CC=1C(=O)C1(O)CCCCC1 QNODIIQQMGDSEF-UHFFFAOYSA-N 0.000 description 1
- CKGKXGQVRVAKEA-UHFFFAOYSA-N (2-methylphenyl)-phenylmethanone Chemical compound CC1=CC=CC=C1C(=O)C1=CC=CC=C1 CKGKXGQVRVAKEA-UHFFFAOYSA-N 0.000 description 1
- ZNAMMSOYKPMPGC-HTOAHKCRSA-N (2r,3r,4s,5r,6s)-2-(hydroxymethyl)-6-(2-phenylethylsulfanyl)oxane-3,4,5-triol Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1SCCC1=CC=CC=C1 ZNAMMSOYKPMPGC-HTOAHKCRSA-N 0.000 description 1
- URBLVRAVOIVZFJ-UHFFFAOYSA-N (3-methylphenyl)-phenylmethanone Chemical compound CC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1 URBLVRAVOIVZFJ-UHFFFAOYSA-N 0.000 description 1
- WXPWZZHELZEVPO-UHFFFAOYSA-N (4-methylphenyl)-phenylmethanone Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC=CC=C1 WXPWZZHELZEVPO-UHFFFAOYSA-N 0.000 description 1
- UOCLXMDMGBRAIB-UHFFFAOYSA-N 1,1,1-trichloroethane Chemical compound CC(Cl)(Cl)Cl UOCLXMDMGBRAIB-UHFFFAOYSA-N 0.000 description 1
- ROLAGNYPWIVYTG-UHFFFAOYSA-N 1,2-bis(4-methoxyphenyl)ethanamine;hydrochloride Chemical compound Cl.C1=CC(OC)=CC=C1CC(N)C1=CC=C(OC)C=C1 ROLAGNYPWIVYTG-UHFFFAOYSA-N 0.000 description 1
- MSAHTMIQULFMRG-UHFFFAOYSA-N 1,2-diphenyl-2-propan-2-yloxyethanone Chemical compound C=1C=CC=CC=1C(OC(C)C)C(=O)C1=CC=CC=C1 MSAHTMIQULFMRG-UHFFFAOYSA-N 0.000 description 1
- QWQFVUQPHUKAMY-UHFFFAOYSA-N 1,2-diphenyl-2-propoxyethanone Chemical compound C=1C=CC=CC=1C(OCCC)C(=O)C1=CC=CC=C1 QWQFVUQPHUKAMY-UHFFFAOYSA-N 0.000 description 1
- WZRRRFSJFQTGGB-UHFFFAOYSA-N 1,3,5-triazinane-2,4,6-trithione Chemical compound S=C1NC(=S)NC(=S)N1 WZRRRFSJFQTGGB-UHFFFAOYSA-N 0.000 description 1
- KOMNUTZXSVSERR-UHFFFAOYSA-N 1,3,5-tris(prop-2-enyl)-1,3,5-triazinane-2,4,6-trione Chemical compound C=CCN1C(=O)N(CC=C)C(=O)N(CC=C)C1=O KOMNUTZXSVSERR-UHFFFAOYSA-N 0.000 description 1
- TYMYJUHDFROXOO-UHFFFAOYSA-N 1,3-bis(prop-2-enoxy)-2,2-bis(prop-2-enoxymethyl)propane Chemical compound C=CCOCC(COCC=C)(COCC=C)COCC=C TYMYJUHDFROXOO-UHFFFAOYSA-N 0.000 description 1
- DKEGCUDAFWNSSO-UHFFFAOYSA-N 1,8-dibromooctane Chemical compound BrCCCCCCCCBr DKEGCUDAFWNSSO-UHFFFAOYSA-N 0.000 description 1
- HASUCEDGKYJBDC-UHFFFAOYSA-N 1-[3-[[bis(oxiran-2-ylmethyl)amino]methyl]cyclohexyl]-n,n-bis(oxiran-2-ylmethyl)methanamine Chemical compound C1OC1CN(CC1CC(CN(CC2OC2)CC2OC2)CCC1)CC1CO1 HASUCEDGKYJBDC-UHFFFAOYSA-N 0.000 description 1
- DLKQHBOKULLWDQ-UHFFFAOYSA-N 1-bromonaphthalene Chemical compound C1=CC=C2C(Br)=CC=CC2=C1 DLKQHBOKULLWDQ-UHFFFAOYSA-N 0.000 description 1
- VKQJCUYEEABXNK-UHFFFAOYSA-N 1-chloro-4-propoxythioxanthen-9-one Chemical compound S1C2=CC=CC=C2C(=O)C2=C1C(OCCC)=CC=C2Cl VKQJCUYEEABXNK-UHFFFAOYSA-N 0.000 description 1
- JTPNRXUCIXHOKM-UHFFFAOYSA-N 1-chloronaphthalene Chemical compound C1=CC=C2C(Cl)=CC=CC2=C1 JTPNRXUCIXHOKM-UHFFFAOYSA-N 0.000 description 1
- CWLKTJOTWITYSI-UHFFFAOYSA-N 1-fluoronaphthalene Chemical compound C1=CC=C2C(F)=CC=CC2=C1 CWLKTJOTWITYSI-UHFFFAOYSA-N 0.000 description 1
- 239000012956 1-hydroxycyclohexylphenyl-ketone Substances 0.000 description 1
- NHPPIJMARIVBGU-UHFFFAOYSA-N 1-iodonaphthalene Chemical compound C1=CC=C2C(I)=CC=CC2=C1 NHPPIJMARIVBGU-UHFFFAOYSA-N 0.000 description 1
- HECLRDQVFMWTQS-RGOKHQFPSA-N 1755-01-7 Chemical compound C1[C@H]2[C@@H]3CC=C[C@@H]3[C@@H]1C=C2 HECLRDQVFMWTQS-RGOKHQFPSA-N 0.000 description 1
- KAJBSGLXSREIHP-UHFFFAOYSA-N 2,2-bis[(2-sulfanylacetyl)oxymethyl]butyl 2-sulfanylacetate Chemical compound SCC(=O)OCC(CC)(COC(=O)CS)COC(=O)CS KAJBSGLXSREIHP-UHFFFAOYSA-N 0.000 description 1
- KWVGIHKZDCUPEU-UHFFFAOYSA-N 2,2-dimethoxy-2-phenylacetophenone Chemical compound C=1C=CC=CC=1C(OC)(OC)C(=O)C1=CC=CC=C1 KWVGIHKZDCUPEU-UHFFFAOYSA-N 0.000 description 1
- BJELTSYBAHKXRW-UHFFFAOYSA-N 2,4,6-triallyloxy-1,3,5-triazine Chemical compound C=CCOC1=NC(OCC=C)=NC(OCC=C)=N1 BJELTSYBAHKXRW-UHFFFAOYSA-N 0.000 description 1
- PSYGHMBJXWRQFD-UHFFFAOYSA-N 2-(2-sulfanylacetyl)oxyethyl 2-sulfanylacetate Chemical compound SCC(=O)OCCOC(=O)CS PSYGHMBJXWRQFD-UHFFFAOYSA-N 0.000 description 1
- CNDCQWGRLNGNNO-UHFFFAOYSA-N 2-(2-sulfanylethoxy)ethanethiol Chemical compound SCCOCCS CNDCQWGRLNGNNO-UHFFFAOYSA-N 0.000 description 1
- KSJBMDCFYZKAFH-UHFFFAOYSA-N 2-(2-sulfanylethylsulfanyl)ethanethiol Chemical compound SCCSCCS KSJBMDCFYZKAFH-UHFFFAOYSA-N 0.000 description 1
- DJOYTAUERRJRAT-UHFFFAOYSA-N 2-(n-methyl-4-nitroanilino)acetonitrile Chemical compound N#CCN(C)C1=CC=C([N+]([O-])=O)C=C1 DJOYTAUERRJRAT-UHFFFAOYSA-N 0.000 description 1
- XOGPDSATLSAZEK-UHFFFAOYSA-N 2-Aminoanthraquinone Chemical compound C1=CC=C2C(=O)C3=CC(N)=CC=C3C(=O)C2=C1 XOGPDSATLSAZEK-UHFFFAOYSA-N 0.000 description 1
- LUZDYPLAQQGJEA-UHFFFAOYSA-N 2-Methoxynaphthalene Chemical compound C1=CC=CC2=CC(OC)=CC=C21 LUZDYPLAQQGJEA-UHFFFAOYSA-N 0.000 description 1
- AOBIOSPNXBMOAT-UHFFFAOYSA-N 2-[2-(oxiran-2-ylmethoxy)ethoxymethyl]oxirane Chemical compound C1OC1COCCOCC1CO1 AOBIOSPNXBMOAT-UHFFFAOYSA-N 0.000 description 1
- WTYYGFLRBWMFRY-UHFFFAOYSA-N 2-[6-(oxiran-2-ylmethoxy)hexoxymethyl]oxirane Chemical compound C1OC1COCCCCCCOCC1CO1 WTYYGFLRBWMFRY-UHFFFAOYSA-N 0.000 description 1
- TXBCBTDQIULDIA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)CO TXBCBTDQIULDIA-UHFFFAOYSA-N 0.000 description 1
- SJODITPGMMSNRF-UHFFFAOYSA-N 2-aminofluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC(N)=CC=C3C2=C1 SJODITPGMMSNRF-UHFFFAOYSA-N 0.000 description 1
- APSMUYYLXZULMS-UHFFFAOYSA-N 2-bromonaphthalene Chemical compound C1=CC=CC2=CC(Br)=CC=C21 APSMUYYLXZULMS-UHFFFAOYSA-N 0.000 description 1
- CGYGETOMCSJHJU-UHFFFAOYSA-N 2-chloronaphthalene Chemical compound C1=CC=CC2=CC(Cl)=CC=C21 CGYGETOMCSJHJU-UHFFFAOYSA-N 0.000 description 1
- KMNCBSZOIQAUFX-UHFFFAOYSA-N 2-ethoxy-1,2-diphenylethanone Chemical compound C=1C=CC=CC=1C(OCC)C(=O)C1=CC=CC=C1 KMNCBSZOIQAUFX-UHFFFAOYSA-N 0.000 description 1
- RFMXKZGZSGFZES-UHFFFAOYSA-N 2-ethyl-2-(hydroxymethyl)propane-1,3-diol;2-sulfanylacetic acid Chemical compound OC(=O)CS.OC(=O)CS.OC(=O)CS.CCC(CO)(CO)CO RFMXKZGZSGFZES-UHFFFAOYSA-N 0.000 description 1
- SJEBAWHUJDUKQK-UHFFFAOYSA-N 2-ethylanthraquinone Chemical compound C1=CC=C2C(=O)C3=CC(CC)=CC=C3C(=O)C2=C1 SJEBAWHUJDUKQK-UHFFFAOYSA-N 0.000 description 1
- LJKDOMVGKKPJBH-UHFFFAOYSA-N 2-ethylhexyl dihydrogen phosphate Chemical compound CCCCC(CC)COP(O)(O)=O LJKDOMVGKKPJBH-UHFFFAOYSA-N 0.000 description 1
- GXUBPHMYNSICJC-UHFFFAOYSA-N 2-hydroxyfluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC(O)=CC=C3C2=C1 GXUBPHMYNSICJC-UHFFFAOYSA-N 0.000 description 1
- FRNLBIWVMVNNAZ-UHFFFAOYSA-N 2-iodonaphthalene Chemical compound C1=CC=CC2=CC(I)=CC=C21 FRNLBIWVMVNNAZ-UHFFFAOYSA-N 0.000 description 1
- BQZJOQXSCSZQPS-UHFFFAOYSA-N 2-methoxy-1,2-diphenylethanone Chemical compound C=1C=CC=CC=1C(OC)C(=O)C1=CC=CC=C1 BQZJOQXSCSZQPS-UHFFFAOYSA-N 0.000 description 1
- YRNDGUSDBCARGC-UHFFFAOYSA-N 2-methoxyacetophenone Chemical compound COCC(=O)C1=CC=CC=C1 YRNDGUSDBCARGC-UHFFFAOYSA-N 0.000 description 1
- LWRBVKNFOYUCNP-UHFFFAOYSA-N 2-methyl-1-(4-methylsulfanylphenyl)-2-morpholin-4-ylpropan-1-one Chemical compound C1=CC(SC)=CC=C1C(=O)C(C)(C)N1CCOCC1 LWRBVKNFOYUCNP-UHFFFAOYSA-N 0.000 description 1
- SLAMLWHELXOEJZ-UHFFFAOYSA-N 2-nitrobenzoic acid Chemical compound OC(=O)C1=CC=CC=C1[N+]([O-])=O SLAMLWHELXOEJZ-UHFFFAOYSA-N 0.000 description 1
- FAXDZWQIWUSWJH-UHFFFAOYSA-N 3-methoxypropan-1-amine Chemical compound COCCCN FAXDZWQIWUSWJH-UHFFFAOYSA-N 0.000 description 1
- ATVJXMYDOSMEPO-UHFFFAOYSA-N 3-prop-2-enoxyprop-1-ene Chemical compound C=CCOCC=C ATVJXMYDOSMEPO-UHFFFAOYSA-N 0.000 description 1
- IPNDIMIIGZSERC-UHFFFAOYSA-N 4-(2-sulfanylacetyl)oxybutyl 2-sulfanylacetate Chemical compound SCC(=O)OCCCCOC(=O)CS IPNDIMIIGZSERC-UHFFFAOYSA-N 0.000 description 1
- BMVWCPGVLSILMU-UHFFFAOYSA-N 5,6-dihydrodibenzo[2,1-b:2',1'-f][7]annulen-11-one Chemical compound C1CC2=CC=CC=C2C(=O)C2=CC=CC=C21 BMVWCPGVLSILMU-UHFFFAOYSA-N 0.000 description 1
- 102100027123 55 kDa erythrocyte membrane protein Human genes 0.000 description 1
- IXDGHAZCSMVIFX-UHFFFAOYSA-N 6-(dibutylamino)-1h-1,3,5-triazine-2,4-dithione Chemical compound CCCCN(CCCC)C1=NC(=S)NC(=S)N1 IXDGHAZCSMVIFX-UHFFFAOYSA-N 0.000 description 1
- BRUOAURMAFDGLP-UHFFFAOYSA-N 9,10-dibromoanthracene Chemical compound C1=CC=C2C(Br)=C(C=CC=C3)C3=C(Br)C2=C1 BRUOAURMAFDGLP-UHFFFAOYSA-N 0.000 description 1
- FKDIWXZNKAZCBY-UHFFFAOYSA-N 9,10-dichloroanthracene Chemical compound C1=CC=C2C(Cl)=C(C=CC=C3)C3=C(Cl)C2=C1 FKDIWXZNKAZCBY-UHFFFAOYSA-N 0.000 description 1
- FCNCGHJSNVOIKE-UHFFFAOYSA-N 9,10-diphenylanthracene Chemical compound C1=CC=CC=C1C(C1=CC=CC=C11)=C(C=CC=C2)C2=C1C1=CC=CC=C1 FCNCGHJSNVOIKE-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 1
- 229910052582 BN Inorganic materials 0.000 description 1
- JLLMOYPIVVKFHY-UHFFFAOYSA-N Benzenethiol, 4,4'-thiobis- Chemical compound C1=CC(S)=CC=C1SC1=CC=C(S)C=C1 JLLMOYPIVVKFHY-UHFFFAOYSA-N 0.000 description 1
- PZNSFCLAULLKQX-UHFFFAOYSA-N Boron nitride Chemical compound N#B PZNSFCLAULLKQX-UHFFFAOYSA-N 0.000 description 1
- NOEMSRWQFGPZQS-UHFFFAOYSA-N CCC(O)=S.CCC(O)=S.CCC(O)=S.CCC(CO)(CO)CO Chemical compound CCC(O)=S.CCC(O)=S.CCC(O)=S.CCC(CO)(CO)CO NOEMSRWQFGPZQS-UHFFFAOYSA-N 0.000 description 1
- 229910000975 Carbon steel Inorganic materials 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910000881 Cu alloy Inorganic materials 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 239000004641 Diallyl-phthalate Substances 0.000 description 1
- JYFHYPJRHGVZDY-UHFFFAOYSA-N Dibutyl phosphate Chemical compound CCCCOP(O)(=O)OCCCC JYFHYPJRHGVZDY-UHFFFAOYSA-N 0.000 description 1
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 1
- 229910000640 Fe alloy Inorganic materials 0.000 description 1
- 229920002430 Fibre-reinforced plastic Polymers 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 229910002601 GaN Inorganic materials 0.000 description 1
- JMASRVWKEDWRBT-UHFFFAOYSA-N Gallium nitride Chemical compound [Ga]#N JMASRVWKEDWRBT-UHFFFAOYSA-N 0.000 description 1
- 101001057956 Homo sapiens 55 kDa erythrocyte membrane protein Proteins 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- BWPYBAJTDILQPY-UHFFFAOYSA-N Methoxyphenone Chemical compound C1=C(C)C(OC)=CC=C1C(=O)C1=CC=CC(C)=C1 BWPYBAJTDILQPY-UHFFFAOYSA-N 0.000 description 1
- PUZHXYSDMURDFB-UHFFFAOYSA-N O(CCOCCOCCOC)C(CCCOCCOCCOBOCCOCCOCCOCCOC)OCCOCCOCCOC Chemical compound O(CCOCCOCCOC)C(CCCOCCOCCOBOCCOCCOCCOCCOC)OCCOCCOCCOC PUZHXYSDMURDFB-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 206010070834 Sensitisation Diseases 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 229910001069 Ti alloy Inorganic materials 0.000 description 1
- LCKIEQZJEYYRIY-UHFFFAOYSA-N Titanium ion Chemical compound [Ti+4] LCKIEQZJEYYRIY-UHFFFAOYSA-N 0.000 description 1
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 1
- OXCUXICYDJWRNK-UHFFFAOYSA-N [(2,4-dibutoxyphenyl)-(2,4,6-trimethylbenzoyl)phosphoryl]-(2,4,6-trimethylphenyl)methanone Chemical compound CCCCOC1=CC(OCCCC)=CC=C1P(=O)(C(=O)C=1C(=CC(C)=CC=1C)C)C(=O)C1=C(C)C=C(C)C=C1C OXCUXICYDJWRNK-UHFFFAOYSA-N 0.000 description 1
- LFOXEOLGJPJZAA-UHFFFAOYSA-N [(2,6-dimethoxybenzoyl)-(2,4,4-trimethylpentyl)phosphoryl]-(2,6-dimethoxyphenyl)methanone Chemical compound COC1=CC=CC(OC)=C1C(=O)P(=O)(CC(C)CC(C)(C)C)C(=O)C1=C(OC)C=CC=C1OC LFOXEOLGJPJZAA-UHFFFAOYSA-N 0.000 description 1
- MEESPVWIOBCLJW-KTKRTIGZSA-N [(z)-octadec-9-enyl] dihydrogen phosphate Chemical compound CCCCCCCC\C=C/CCCCCCCCOP(O)(O)=O MEESPVWIOBCLJW-KTKRTIGZSA-N 0.000 description 1
- RPRIKYFYCSOOAG-UHFFFAOYSA-N [2,4,6-trimethyl-3,5-bis(sulfanylmethyl)phenyl]methanethiol Chemical compound CC1=C(CS)C(C)=C(CS)C(C)=C1CS RPRIKYFYCSOOAG-UHFFFAOYSA-N 0.000 description 1
- STWRQBYJSPXXQE-UHFFFAOYSA-N [3,5-bis(sulfanylmethyl)phenyl]methanethiol Chemical compound SCC1=CC(CS)=CC(CS)=C1 STWRQBYJSPXXQE-UHFFFAOYSA-N 0.000 description 1
- DBHQYYNDKZDVTN-UHFFFAOYSA-N [4-(4-methylphenyl)sulfanylphenyl]-phenylmethanone Chemical compound C1=CC(C)=CC=C1SC1=CC=C(C(=O)C=2C=CC=CC=2)C=C1 DBHQYYNDKZDVTN-UHFFFAOYSA-N 0.000 description 1
- GUCYFKSBFREPBC-UHFFFAOYSA-N [phenyl-(2,4,6-trimethylbenzoyl)phosphoryl]-(2,4,6-trimethylphenyl)methanone Chemical compound CC1=CC(C)=CC(C)=C1C(=O)P(=O)(C=1C=CC=CC=1)C(=O)C1=C(C)C=C(C)C=C1C GUCYFKSBFREPBC-UHFFFAOYSA-N 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- IHUNBGSDBOWDMA-AQFIFDHZSA-N all-trans-acitretin Chemical compound COC1=CC(C)=C(\C=C\C(\C)=C\C=C\C(\C)=C\C(O)=O)C(C)=C1C IHUNBGSDBOWDMA-AQFIFDHZSA-N 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- OJMOMXZKOWKUTA-UHFFFAOYSA-N aluminum;borate Chemical compound [Al+3].[O-]B([O-])[O-] OJMOMXZKOWKUTA-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 238000010539 anionic addition polymerization reaction Methods 0.000 description 1
- IYYACUGXZLFMGN-UHFFFAOYSA-N anthracene-2,6,9,10-tetracarbonitrile Chemical compound C1=C(C#N)C=CC2=C(C#N)C3=CC(C#N)=CC=C3C(C#N)=C21 IYYACUGXZLFMGN-UHFFFAOYSA-N 0.000 description 1
- BIOPPFDHKHWJIA-UHFFFAOYSA-N anthracene-9,10-dinitrile Chemical compound C1=CC=C2C(C#N)=C(C=CC=C3)C3=C(C#N)C2=C1 BIOPPFDHKHWJIA-UHFFFAOYSA-N 0.000 description 1
- KEQZHLAEKAVZLY-UHFFFAOYSA-N anthracene-9-carbonitrile Chemical compound C1=CC=C2C(C#N)=C(C=CC=C3)C3=CC2=C1 KEQZHLAEKAVZLY-UHFFFAOYSA-N 0.000 description 1
- 150000001454 anthracenes Chemical class 0.000 description 1
- RJGDLRCDCYRQOQ-UHFFFAOYSA-N anthrone Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3CC2=C1 RJGDLRCDCYRQOQ-UHFFFAOYSA-N 0.000 description 1
- 150000008425 anthrones Chemical class 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- HNYOPLTXPVRDBG-UHFFFAOYSA-N barbituric acid Chemical compound O=C1CC(=O)NC(=O)N1 HNYOPLTXPVRDBG-UHFFFAOYSA-N 0.000 description 1
- 229910002113 barium titanate Inorganic materials 0.000 description 1
- JRPBQTZRNDNNOP-UHFFFAOYSA-N barium titanate Chemical compound [Ba+2].[Ba+2].[O-][Ti]([O-])([O-])[O-] JRPBQTZRNDNNOP-UHFFFAOYSA-N 0.000 description 1
- 229910021523 barium zirconate Inorganic materials 0.000 description 1
- DQBAOWPVHRWLJC-UHFFFAOYSA-N barium(2+);dioxido(oxo)zirconium Chemical compound [Ba+2].[O-][Zr]([O-])=O DQBAOWPVHRWLJC-UHFFFAOYSA-N 0.000 description 1
- JTRPLRMCBJSBJV-UHFFFAOYSA-N benzonaphthacene Natural products C1=CC=C2C3=CC4=CC5=CC=CC=C5C=C4C=C3C=CC2=C1 JTRPLRMCBJSBJV-UHFFFAOYSA-N 0.000 description 1
- 150000008366 benzophenones Chemical class 0.000 description 1
- 229950011260 betanaphthol Drugs 0.000 description 1
- HECGKCOICWUUJU-UHFFFAOYSA-N bis(diphenylphosphanylmethyl)-phenylphosphane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CP(C=1C=CC=CC=1)CP(C=1C=CC=CC=1)C1=CC=CC=C1 HECGKCOICWUUJU-UHFFFAOYSA-N 0.000 description 1
- ZPOLOEWJWXZUSP-AATRIKPKSA-N bis(prop-2-enyl) (e)-but-2-enedioate Chemical compound C=CCOC(=O)\C=C\C(=O)OCC=C ZPOLOEWJWXZUSP-AATRIKPKSA-N 0.000 description 1
- QUDWYFHPNIMBFC-UHFFFAOYSA-N bis(prop-2-enyl) benzene-1,2-dicarboxylate Chemical compound C=CCOC(=O)C1=CC=CC=C1C(=O)OCC=C QUDWYFHPNIMBFC-UHFFFAOYSA-N 0.000 description 1
- ZDNFTNPFYCKVTB-UHFFFAOYSA-N bis(prop-2-enyl) benzene-1,4-dicarboxylate Chemical compound C=CCOC(=O)C1=CC=C(C(=O)OCC=C)C=C1 ZDNFTNPFYCKVTB-UHFFFAOYSA-N 0.000 description 1
- HEBKPAWCVUSXMB-UHFFFAOYSA-N bis(prop-2-enyl) cyclohexane-1,2-dicarboxylate Chemical compound C=CCOC(=O)C1CCCCC1C(=O)OCC=C HEBKPAWCVUSXMB-UHFFFAOYSA-N 0.000 description 1
- FPODCVUTIPDRTE-UHFFFAOYSA-N bis(prop-2-enyl) hexanedioate Chemical compound C=CCOC(=O)CCCCC(=O)OCC=C FPODCVUTIPDRTE-UHFFFAOYSA-N 0.000 description 1
- MQDJYUACMFCOFT-UHFFFAOYSA-N bis[2-(1-hydroxycyclohexyl)phenyl]methanone Chemical compound C=1C=CC=C(C(=O)C=2C(=CC=CC=2)C2(O)CCCCC2)C=1C1(O)CCCCC1 MQDJYUACMFCOFT-UHFFFAOYSA-N 0.000 description 1
- VYHBFRJRBHMIQZ-UHFFFAOYSA-N bis[4-(diethylamino)phenyl]methanone Chemical compound C1=CC(N(CC)CC)=CC=C1C(=O)C1=CC=C(N(CC)CC)C=C1 VYHBFRJRBHMIQZ-UHFFFAOYSA-N 0.000 description 1
- 229910002115 bismuth titanate Inorganic materials 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- SMTOKHQOVJRXLK-UHFFFAOYSA-N butane-1,4-dithiol Chemical compound SCCCCS SMTOKHQOVJRXLK-UHFFFAOYSA-N 0.000 description 1
- QHIWVLPBUQWDMQ-UHFFFAOYSA-N butyl prop-2-enoate;methyl 2-methylprop-2-enoate;prop-2-enoic acid Chemical compound OC(=O)C=C.COC(=O)C(C)=C.CCCCOC(=O)C=C QHIWVLPBUQWDMQ-UHFFFAOYSA-N 0.000 description 1
- AOWKSNWVBZGMTJ-UHFFFAOYSA-N calcium titanate Chemical compound [Ca+2].[O-][Ti]([O-])=O AOWKSNWVBZGMTJ-UHFFFAOYSA-N 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 239000010962 carbon steel Substances 0.000 description 1
- 150000001244 carboxylic acid anhydrides Chemical class 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 229910010293 ceramic material Inorganic materials 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 229910052570 clay Inorganic materials 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- KOMDZQSPRDYARS-UHFFFAOYSA-N cyclopenta-1,3-diene titanium Chemical class [Ti].C1C=CC=C1.C1C=CC=C1 KOMDZQSPRDYARS-UHFFFAOYSA-N 0.000 description 1
- 238000004925 denaturation Methods 0.000 description 1
- 230000036425 denaturation Effects 0.000 description 1
- SEGLCEQVOFDUPX-UHFFFAOYSA-N di-(2-ethylhexyl)phosphoric acid Chemical compound CCCCC(CC)COP(O)(=O)OCC(CC)CCCC SEGLCEQVOFDUPX-UHFFFAOYSA-N 0.000 description 1
- CMMUKUYEPRGBFB-UHFFFAOYSA-L dichromic acid Chemical compound O[Cr](=O)(=O)O[Cr](O)(=O)=O CMMUKUYEPRGBFB-UHFFFAOYSA-L 0.000 description 1
- PODOEQVNFJSWIK-UHFFFAOYSA-N diphenylphosphoryl-(2,4,6-trimethoxyphenyl)methanone Chemical compound COC1=CC(OC)=CC(OC)=C1C(=O)P(=O)(C=1C=CC=CC=1)C1=CC=CC=C1 PODOEQVNFJSWIK-UHFFFAOYSA-N 0.000 description 1
- VFHVQBAGLAREND-UHFFFAOYSA-N diphenylphosphoryl-(2,4,6-trimethylphenyl)methanone Chemical compound CC1=CC(C)=CC(C)=C1C(=O)P(=O)(C=1C=CC=CC=1)C1=CC=CC=C1 VFHVQBAGLAREND-UHFFFAOYSA-N 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- ZJXZSIYSNXKHEA-UHFFFAOYSA-N ethyl dihydrogen phosphate Chemical compound CCOP(O)(O)=O ZJXZSIYSNXKHEA-UHFFFAOYSA-N 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 239000011151 fibre-reinforced plastic Substances 0.000 description 1
- 239000011152 fibreglass Substances 0.000 description 1
- 239000003063 flame retardant Substances 0.000 description 1
- 150000002220 fluorenes Chemical class 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000000295 fuel oil Substances 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 239000010440 gypsum Substances 0.000 description 1
- 229910052602 gypsum Inorganic materials 0.000 description 1
- 239000010720 hydraulic oil Substances 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical group I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 238000004898 kneading Methods 0.000 description 1
- 238000007561 laser diffraction method Methods 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 1
- YVVBECLPRBAATK-UHFFFAOYSA-N methyl 3-hydroxynaphthalene-2-carboxylate Chemical compound C1=CC=C2C=C(O)C(C(=O)OC)=CC2=C1 YVVBECLPRBAATK-UHFFFAOYSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- CWQXQMHSOZUFJS-UHFFFAOYSA-N molybdenum disulfide Chemical compound S=[Mo]=S CWQXQMHSOZUFJS-UHFFFAOYSA-N 0.000 description 1
- SJPFBRJHYRBAGV-UHFFFAOYSA-N n-[[3-[[bis(oxiran-2-ylmethyl)amino]methyl]phenyl]methyl]-1-(oxiran-2-yl)-n-(oxiran-2-ylmethyl)methanamine Chemical compound C1OC1CN(CC=1C=C(CN(CC2OC2)CC2OC2)C=CC=1)CC1CO1 SJPFBRJHYRBAGV-UHFFFAOYSA-N 0.000 description 1
- VBEGHXKAFSLLGE-UHFFFAOYSA-N n-phenylnitramide Chemical compound [O-][N+](=O)NC1=CC=CC=C1 VBEGHXKAFSLLGE-UHFFFAOYSA-N 0.000 description 1
- BENSWQOUPJQWMU-UHFFFAOYSA-N naphthalene-1,4-dicarbonitrile Chemical compound C1=CC=C2C(C#N)=CC=C(C#N)C2=C1 BENSWQOUPJQWMU-UHFFFAOYSA-N 0.000 description 1
- 150000002790 naphthalenes Chemical class 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 150000002828 nitro derivatives Chemical class 0.000 description 1
- XVKLLVZBGMGICC-UHFFFAOYSA-N o-[3-propanethioyloxy-2,2-bis(propanethioyloxymethyl)propyl] propanethioate Chemical compound CCC(=S)OCC(COC(=S)CC)(COC(=S)CC)COC(=S)CC XVKLLVZBGMGICC-UHFFFAOYSA-N 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- AFEQENGXSMURHA-UHFFFAOYSA-N oxiran-2-ylmethanamine Chemical compound NCC1CO1 AFEQENGXSMURHA-UHFFFAOYSA-N 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- 229920001568 phenolic resin Polymers 0.000 description 1
- 239000005011 phenolic resin Substances 0.000 description 1
- HPAFOABSQZMTHE-UHFFFAOYSA-N phenyl-(2,4,6-trimethylphenyl)methanone Chemical compound CC1=CC(C)=CC(C)=C1C(=O)C1=CC=CC=C1 HPAFOABSQZMTHE-UHFFFAOYSA-N 0.000 description 1
- KOVXIYLMAMPQLP-UHFFFAOYSA-N phenyl-(3-phenylphenyl)methanone Chemical group C=1C=CC(C=2C=CC=CC=2)=CC=1C(=O)C1=CC=CC=C1 KOVXIYLMAMPQLP-UHFFFAOYSA-N 0.000 description 1
- LYXOWKPVTCPORE-UHFFFAOYSA-N phenyl-(4-phenylphenyl)methanone Chemical compound C=1C=C(C=2C=CC=CC=2)C=CC=1C(=O)C1=CC=CC=C1 LYXOWKPVTCPORE-UHFFFAOYSA-N 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920005668 polycarbonate resin Polymers 0.000 description 1
- 239000004431 polycarbonate resin Substances 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 229920005644 polyethylene terephthalate glycol copolymer Polymers 0.000 description 1
- 229920000223 polyglycerol Polymers 0.000 description 1
- 229920005672 polyolefin resin Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- MHZDONKZSXBOGL-UHFFFAOYSA-N propyl dihydrogen phosphate Chemical compound CCCOP(O)(O)=O MHZDONKZSXBOGL-UHFFFAOYSA-N 0.000 description 1
- 238000004080 punching Methods 0.000 description 1
- 229940079877 pyrogallol Drugs 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 229910052594 sapphire Inorganic materials 0.000 description 1
- 239000010980 sapphire Substances 0.000 description 1
- 238000000790 scattering method Methods 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 230000008313 sensitization Effects 0.000 description 1
- 238000004904 shortening Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 239000005361 soda-lime glass Substances 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- MZLGASXMSKOWSE-UHFFFAOYSA-N tantalum nitride Chemical compound [Ta]#N MZLGASXMSKOWSE-UHFFFAOYSA-N 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- YRHRIQCWCFGUEQ-UHFFFAOYSA-N thioxanthen-9-one Chemical class C1=CC=C2C(=O)C3=CC=CC=C3SC2=C1 YRHRIQCWCFGUEQ-UHFFFAOYSA-N 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- IJJNTMLAAKKCML-UHFFFAOYSA-N tribenzyl borate Chemical compound C=1C=CC=CC=1COB(OCC=1C=CC=CC=1)OCC1=CC=CC=C1 IJJNTMLAAKKCML-UHFFFAOYSA-N 0.000 description 1
- LGQXXHMEBUOXRP-UHFFFAOYSA-N tributyl borate Chemical compound CCCCOB(OCCCC)OCCCC LGQXXHMEBUOXRP-UHFFFAOYSA-N 0.000 description 1
- STCOOQWBFONSKY-UHFFFAOYSA-N tributyl phosphate Chemical compound CCCCOP(=O)(OCCCC)OCCCC STCOOQWBFONSKY-UHFFFAOYSA-N 0.000 description 1
- BOOITXALNJLNMB-UHFFFAOYSA-N tricyclohexyl borate Chemical compound C1CCCCC1OB(OC1CCCCC1)OC1CCCCC1 BOOITXALNJLNMB-UHFFFAOYSA-N 0.000 description 1
- HWJYGSDXNANCJM-UHFFFAOYSA-N tridodecyl borate Chemical compound CCCCCCCCCCCCOB(OCCCCCCCCCCCC)OCCCCCCCCCCCC HWJYGSDXNANCJM-UHFFFAOYSA-N 0.000 description 1
- AJSTXXYNEIHPMD-UHFFFAOYSA-N triethyl borate Chemical compound CCOB(OCC)OCC AJSTXXYNEIHPMD-UHFFFAOYSA-N 0.000 description 1
- DQWPFSLDHJDLRL-UHFFFAOYSA-N triethyl phosphate Chemical compound CCOP(=O)(OCC)OCC DQWPFSLDHJDLRL-UHFFFAOYSA-N 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- WZGVRXXJKGXOBR-UHFFFAOYSA-N trihexadecyl borate Chemical compound CCCCCCCCCCCCCCCCOB(OCCCCCCCCCCCCCCCC)OCCCCCCCCCCCCCCCC WZGVRXXJKGXOBR-UHFFFAOYSA-N 0.000 description 1
- KDQYHGMMZKMQAA-UHFFFAOYSA-N trihexyl borate Chemical compound CCCCCCOB(OCCCCCC)OCCCCCC KDQYHGMMZKMQAA-UHFFFAOYSA-N 0.000 description 1
- WRECIMRULFAWHA-UHFFFAOYSA-N trimethyl borate Chemical compound COB(OC)OC WRECIMRULFAWHA-UHFFFAOYSA-N 0.000 description 1
- MTPVUVINMAGMJL-UHFFFAOYSA-N trimethyl(1,1,2,2,2-pentafluoroethyl)silane Chemical compound C[Si](C)(C)C(F)(F)C(F)(F)F MTPVUVINMAGMJL-UHFFFAOYSA-N 0.000 description 1
- AZLXEMARTGQBEN-UHFFFAOYSA-N trinonyl borate Chemical compound CCCCCCCCCOB(OCCCCCCCCC)OCCCCCCCCC AZLXEMARTGQBEN-UHFFFAOYSA-N 0.000 description 1
- GZKLCETYSGSMRA-UHFFFAOYSA-N trioctadecyl borate Chemical compound CCCCCCCCCCCCCCCCCCOB(OCCCCCCCCCCCCCCCCCC)OCCCCCCCCCCCCCCCCCC GZKLCETYSGSMRA-UHFFFAOYSA-N 0.000 description 1
- DTBRTYHFHGNZFX-UHFFFAOYSA-N trioctyl borate Chemical compound CCCCCCCCOB(OCCCCCCCC)OCCCCCCCC DTBRTYHFHGNZFX-UHFFFAOYSA-N 0.000 description 1
- JLPJTCGUKOBWRJ-UHFFFAOYSA-N tripentyl borate Chemical compound CCCCCOB(OCCCCC)OCCCCC JLPJTCGUKOBWRJ-UHFFFAOYSA-N 0.000 description 1
- MDCWDBMBZLORER-UHFFFAOYSA-N triphenyl borate Chemical compound C=1C=CC=CC=1OB(OC=1C=CC=CC=1)OC1=CC=CC=C1 MDCWDBMBZLORER-UHFFFAOYSA-N 0.000 description 1
- AAAQKTZKLRYKHR-UHFFFAOYSA-N triphenylmethane Chemical compound C1=CC=CC=C1C(C=1C=CC=CC=1)C1=CC=CC=C1 AAAQKTZKLRYKHR-UHFFFAOYSA-N 0.000 description 1
- NHDIQVFFNDKAQU-UHFFFAOYSA-N tripropan-2-yl borate Chemical compound CC(C)OB(OC(C)C)OC(C)C NHDIQVFFNDKAQU-UHFFFAOYSA-N 0.000 description 1
- LTEHWCSSIHAVOQ-UHFFFAOYSA-N tripropyl borate Chemical compound CCCOB(OCCC)OCCC LTEHWCSSIHAVOQ-UHFFFAOYSA-N 0.000 description 1
- DLVYHYUFIXLWKV-UHFFFAOYSA-N tris(2-ethylhexyl) borate Chemical compound CCCCC(CC)COB(OCC(CC)CCCC)OCC(CC)CCCC DLVYHYUFIXLWKV-UHFFFAOYSA-N 0.000 description 1
- RTMBXAOPKJNOGZ-UHFFFAOYSA-N tris(2-methylphenyl) borate Chemical compound CC1=CC=CC=C1OB(OC=1C(=CC=CC=1)C)OC1=CC=CC=C1C RTMBXAOPKJNOGZ-UHFFFAOYSA-N 0.000 description 1
- FYAMVEZOQXNCIE-UHFFFAOYSA-N tris(3-methylphenyl) borate Chemical compound CC1=CC=CC(OB(OC=2C=C(C)C=CC=2)OC=2C=C(C)C=CC=2)=C1 FYAMVEZOQXNCIE-UHFFFAOYSA-N 0.000 description 1
- PLCFYBDYBCOLSP-UHFFFAOYSA-N tris(prop-2-enyl) 2-hydroxypropane-1,2,3-tricarboxylate Chemical compound C=CCOC(=O)CC(O)(CC(=O)OCC=C)C(=O)OCC=C PLCFYBDYBCOLSP-UHFFFAOYSA-N 0.000 description 1
- GRPURDFRFHUDSP-UHFFFAOYSA-N tris(prop-2-enyl) benzene-1,2,4-tricarboxylate Chemical compound C=CCOC(=O)C1=CC=C(C(=O)OCC=C)C(C(=O)OCC=C)=C1 GRPURDFRFHUDSP-UHFFFAOYSA-N 0.000 description 1
- RQNVJDSEWRGEQR-UHFFFAOYSA-N tris(prop-2-enyl) borate Chemical compound C=CCOB(OCC=C)OCC=C RQNVJDSEWRGEQR-UHFFFAOYSA-N 0.000 description 1
- WAXLMVCEFHKADZ-UHFFFAOYSA-N tris-decyl borate Chemical compound CCCCCCCCCCOB(OCCCCCCCCCC)OCCCCCCCCCC WAXLMVCEFHKADZ-UHFFFAOYSA-N 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- 238000002371 ultraviolet--visible spectrum Methods 0.000 description 1
- 239000013585 weight reducing agent Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K3/00—Materials not provided for elsewhere
- C09K3/10—Materials in mouldable or extrudable form for sealing or packing joints or covers
- C09K3/1006—Materials in mouldable or extrudable form for sealing or packing joints or covers characterised by the chemical nature of one of its constituents
- C09K3/1012—Sulfur-containing polymers, e.g. polysulfides
Definitions
- the present invention relates to a sheet-shaped sealant, that is, a sealant sheet.
- liquid polysulfide polymers contains -S-S- bonds in the molecule, by curing this, it forms a rubber-like hardened product with excellent resistance to oils such as jet fuel and hydraulic oil (oil resistance). can. Therefore, liquid polysulfide polymers are used as raw materials for sealants used in aircraft, for example.
- Technical documents relating to liquid polysulfide polymers include Patent Documents 1 to 3.
- a sealant using a liquid polysulfide polymer is generally carried out by mixing a liquid A containing the liquid polysulfide polymer and a liquid B containing a curing agent for the polysulfide polymer immediately before application to prepare a liquid sealant. to an object and then curing the liquid sealant on the object.
- a strong oxidizing agent such as dichromic acid is often used because the curing reaction can easily proceed at room temperature.
- air bubbles may be generated inside the sealant due to outgassing caused by the curing reaction. Such air bubbles grow inside the sealant, and in some cases, the curing reaction proceeds as they are and remain in the cured product. The presence of air bubbles remaining inside the sealant may cause a decrease in the reliability of the cured sealant, and is a matter of concern from the viewpoint of a decrease in sealing quality.
- an object of the present invention is to provide a polysulfide-based sealant that suppresses the generation and growth of air bubbles and allows obtaining a highly reliable molded product (cured product).
- the sealant sheet disclosed herein comprises a polysulfide polymer (A), a thiol compound (B) having two or more thiol groups in one molecule, and an allyl group having two or more allyl groups in one molecule. It contains a compound (C) and a photoradical generator (D).
- the sealant sheet having the above structure can be cured in a state where it is placed at a desired location through a radical addition reaction (thiolene reaction) between a thiol group and an allyl group, thereby improving its strength.
- a radical addition reaction thiolene reaction
- the sealant sheet before curing is in a state of having suitable elasticity to the extent that the sheet shape can be stably maintained, even if there is outgassing due to the radical addition reaction, the sealant sheet The generation and growth of air bubbles inside can be suppressed.
- the cured sealant that is formed tends to contain no air bubbles, or even if it does contain air bubbles, the size of the air bubbles is suppressed.
- the photo-radical generator (D) is considered to be an outgassing source, it is significant to apply the technology of the present invention to a system containing the photo-radical generator (D).
- the formed hardened sealant can exhibit excellent oil resistance due to the contribution of the polysulfide structure. Furthermore, since the thickness of the cured product can be controlled by the thickness of the sealant sheet to be used, there is no need to adjust the coating thickness during construction, unlike when applying a liquid sealant. Therefore, according to the sealant sheet, the polysulfide sealant can be applied easily and accurately. Furthermore, since the sealant sheet is configured to be capable of promoting the addition reaction by generating radicals from the photoradical generator (D) upon irradiation with light, the radicals from the photoradical generator (D) Good preservability can be exhibited by storing in an environment that suppresses generation.
- thiol compound (B) one having a thiol equivalent weight of 45 g/eq or more and 450 g/eq or less can be preferably used in consideration of the balance between preservability before use and curability during use.
- the sealant sheet contains at least one selected from the group consisting of bifunctional allyl compounds and trifunctional allyl compounds as the allyl compound (C). According to the composition containing such an allyl compound (C), the preservability before use and the curability at the time of use are likely to be improved in a well-balanced manner.
- the photoradical generator (D) for example, at least one selected from the group consisting of alkylphenone-based photopolymerization initiators, acylphosphine oxide-based photopolymerization initiators and titanocene compound-based photopolymerization initiators can be preferably employed.
- a sealant sheet that achieves both good preservability before use and good curability (radical photocurability) during use can be suitably realized.
- the sealant sheet disclosed here can contain a filler.
- the use of fillers can improve one or both of the strength and elongation of the cured sealant.
- the sealant sheet preferably has a storage modulus of 0.005 MPa or more and 0.8 MPa or less at 25°C.
- a sealant sheet having a storage elastic modulus within this range tends to favorably balance the adhesion to an object and the ability to maintain the shape of the sheet.
- the sealant sheet disclosed herein is a sheet-shaped sealant sheet comprising a thiol group-containing polysulfide polymer (AB) having two or more thiol groups in one molecule, and one molecule It contains an allyl compound (C) having two or more allyl groups therein and a photoradical generator (D).
- a sealant sheet having such a structure can also be cured in a state where it is placed at a desired location through a radical addition reaction between a thiol group and an allyl group, thereby improving its strength.
- the sealant sheet before curing is in a state of having a suitable elasticity that can stably maintain the sheet shape, even if there is outgassing due to the above radical addition reaction, the inside of the sealant sheet The generation and growth of bubbles in can be suppressed. For this reason, the cured sealant that is formed tends to contain no air bubbles, or even if it does contain air bubbles, the size of the air bubbles is suppressed.
- the sealant sheet before use (that is, before placement at a desired location) is in the form of a sealant sheet with a release liner, which includes the sealant sheet and a release liner having a release surface in contact with at least one surface thereof.
- a sealant sheet having such a form is preferable from the viewpoint of storage stability of the sealant sheet, and handleability during transportation, processing, placement at a desired location, and the like.
- a cured sealant that is a cured polysulfide sealant.
- the cured product contains a disulfide structure, a structure derived from a radical addition reaction between a thiol group and an allyl group, and a compound derived from a photoradical generator.
- a cured sealant is preferable because it can be formed by promoting a radical addition reaction (thiolene reaction) between a thiol group and an allyl group triggered by radicals generated from a photo-radical generator.
- FIG. 4 is a cross-sectional view schematically showing another configuration example of a sealant sheet
- the sealant sheet disclosed herein is preliminarily formed into a sheet shape, and can be placed in the seal target location in such a sheet shape.
- the sealant sheet is prepared by mixing a liquid sealant (for example, a liquid A containing a liquid polysulfide polymer and a liquid B containing a curing agent for the polysulfide polymer) that is applied to a sealing target area in a liquid form immediately before application. liquid sealants prepared together).
- a liquid sealant for example, a liquid A containing a liquid polysulfide polymer and a liquid B containing a curing agent for the polysulfide polymer
- liquid sealants prepared together liquid sealants prepared together.
- the sealant sheet disclosed herein can be cured using an addition reaction (thiolene reaction) between a thiol group and an allyl group.
- the sealant sheet disclosed herein is clearly distinguished from the cured sealant (cured sealant).
- the sealant sheet disclosed herein can be understood as a semi-cured sealant sheet that can be further cured after being placed at the location to be sealed.
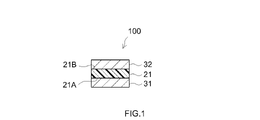
- the sealant sheet 21 shown in FIG. 1 has one surface (first surface) 21A and the other surface (second surface) 21B formed by release liners 31 and 32, at least the sealant sheet 21 side of which is a release surface. each protected.
- the sealant sheet 21 having such a form can be grasped as a component of the sealant sheet 100 with a release liner including the sealant sheet 21 and the release liners 31 and 32 .
- the sealant sheet 21 shown in FIG. 2 has one surface 21A protected by a release liner 31 having release surfaces on both sides. is in contact with the back surface of the release liner 31, so that the surface 21B is also protected by the release liner 31.
- the sealant sheet 21 having such a form can be grasped as a constituent element of the sealant sheet 200 with a release liner including the sealant sheet 21 and the release liner 31 .
- the sealant sheet disclosed herein has shape retention to the extent that it can stably maintain the sheet shape at room temperature (for example, about 25°C).
- the shape retention can also be understood as resistance to plastic deformation such as flow.
- the storage elastic modulus of the sealant sheet at 25° C. (hereinafter also simply referred to as “storage elastic modulus”) may be, for example, 0.005 MPa or more (for example, more than 0.005 MPa), and may be more than 0.01 MPa. preferable.
- storage elastic modulus of the sealant sheet increases, the sealant sheet tends to be improved in handleability and workability (for example, cuttability, antiblocking property, reworkability, etc.).
- the sealant sheet may have a storage modulus of, for example, 0.05 MPa or greater, 0.1 MPa or greater, or 0.2 MPa or greater.
- the upper limit of the storage modulus is not particularly limited.
- the storage modulus of the sealant sheet may be, for example, 2 MPa or less, 1 MPa or less, 0.8 MPa or less, 0.6 MPa or less, 0.5 MPa or less, or 0.5 MPa or less. It may be 4 MPa or less, or 0.3 MPa or less.
- the storage elastic modulus is measured using a viscoelasticity tester under conditions of a frequency of 1 Hz and a strain of 0.5%.
- As the viscoelasticity tester model name "ARES G2" manufactured by TA Instruments Japan Co., Ltd. or its equivalent can be used. More specifically, the storage modulus is measured by the method described in Examples below.
- the storage elastic modulus of the sealant sheet means the storage elastic modulus of the sealant sheet before curing, and is distinguished from the storage elastic modulus of the sealant sheet after curing (cured sealant). be done.
- the storage elastic modulus of the sealant sheet typically means the storage elastic modulus before the sealant sheet is used, that is, before it is placed on a sealing target site by attachment or the like.
- the thickness of the sealant sheet is not particularly limited, and can be selected according to the desired thickness of the cured sealant. From the viewpoint of sealing reliability and the like, in some embodiments, the thickness of the sealant sheet may be, for example, 0.01 mm or more, 0.03 mm or more, 0.05 mm or more, or 0.1 mm or more. , or 0.15 mm or more. Sealant sheets disclosed herein may also suitably be implemented in embodiments where the thickness is, for example, greater than 0.3 mm, greater than 0.5 mm, greater than 1 mm, or greater than 1.5 mm.
- the thickness of the sealant sheet may be, for example, 10 mm or less, 5 mm or less, 3 mm or less, 2 mm or less, 1 mm or less, or 0.5 mm or less, It may be 0.3 mm or less. As the thickness of the sealant sheet becomes smaller, the photocurability tends to improve. Reducing the thickness of the sealant sheet can be advantageous from the viewpoint of conformability to the surface shape of the sealing target location, weight reduction, and the like.
- the sealant sheet disclosed herein comprises a polysulfide polymer (A).
- the polysulfide polymer (A) is a polymer having a repeating unit containing a disulfide structure represented by -SS- and contributes to improving the oil resistance of the cured product formed from the sealant sheet.
- the number of disulfide structures contained in one molecule of polysulfide polymer (A) may be one, or two or more. From the viewpoint of oil resistance of the cured product, a polysulfide polymer (A) containing an average of 3 or more disulfide structures per molecule can be preferably employed.
- the average value of the number of disulfide structures per molecule of the polysulfide polymer (A) (hereinafter also referred to as the average number of disulfide groups) may be, for example, 5 or more, 10 or more, 15 or more, 20 It can be more than that.
- the upper limit of the average number of disulfide groups is not particularly limited, it may be, for example, 100 or less, 70 or less, or 50 or less from the viewpoint of ease of production of the sealant sheet (e.g., easiness of forming into a sheet shape). good.
- the disulfide structure is preferably contained in the main chain of the polysulfide polymer (A). Including a disulfide structure in the main chain tends to form a cured product with good elongation.
- the polysulfide polymer (A) preferably contains repeating units represented by the following general formula (1).
- R 1 , R 2 and R 3 are each independently an alkylene group having 1 to 4 carbon atoms, preferably an alkylene group having 1 to 3 carbon atoms, More preferably, it is an alkylene group having 1 to 2 carbon atoms.
- the repeating unit (1) has a structure in which an ether structure and a disulfide structure are linked. A polysulfide polymer (A) having such a repeating unit (1) tends to form a cured product having excellent oil resistance and flexibility.
- the average number of repeating units (1) contained in one molecule of polysulfide polymer (A) may be, for example, 5 or more, 10 or more, 15 or more, or 20 or more. Further, the average value may be, for example, 100 or less, 70 or less, or 50 or less.
- the polysulfide polymer (A) may have only one region in which the repeating units (1) are continuous, or may have two or more regions in one molecule.
- the polysulfide polymer (A) may contain at least one of a structure represented by general formula (2a) and a structure represented by general formula (2b) below.
- —CH 2 —S—CH 2 CHOH—R′ (2a) —CH 2 —S—CH(CH 2 OH)—R′ (2b)
- R' in general formulas (2a) and (2b) is an organic group having at least one (for example, about 1 to 5) epoxy group.
- the structures of general formulas (2a) and (2b) are formed by an addition reaction of, for example, a thiol having a structural moiety represented by —CH 2 —SH and an epoxy compound having a substituent R′ on the epoxy ring.
- the number of structures represented by general formula (2a) or (2b) (when both the structure represented by general formula (2a) and the structure represented by general formula (2b) are included, the total number of them ) may be, for example, 1.1 or more, 1.3 or more, 1.5 or more, or 1.8 or more, as an average value per molecule of the polysulfide polymer (A). It may be 0 or more, or may be greater than 2.0. Further, the average value may be, for example, 15 or less, 10 or less, 7.0 or less, or 5.0 or less.
- the structures represented by the above general formulas (2a) and (2b) can be formed by an addition reaction between a thiol group and an epoxy group.
- the polysulfide polymer (A) containing the structures represented by the general formulas (2a) and (2b) includes, for example, a thiol group-containing polysulfide having a disulfide structure and a thiol group in one molecule, and two It may be a reaction product or a modified product thereof with an epoxy compound having an epoxy group as described above.
- the weight average molecular weight (Mw) of the thiol group-containing polysulfide as a precursor of the polysulfide polymer (A) is not particularly limited, and may be, for example, 500 or more, 800 or more, 1000 or more, or even more than 1000. Well, it can be over 2000. A thiol group-containing polysulfide with a higher Mw tends to form a sealant sheet that gives a cured product with better elongation. In some embodiments, the Mw of the thiol group-containing polysulfide can be greater than 2500, greater than 3000, greater than 3500, for example.
- the Mw of the thiol group-containing polysulfide may be, for example, 30,000 or less, or may be 10,000 or less. From the viewpoint of handleability and reactivity with the epoxy compound, in some embodiments, the Mw of the thiol group-containing polysulfide may be, for example, less than 9000, less than 8000, less than 7500, or less than 7000. Well, even less than 6500.
- Mw of a polymer such as a thiol group-containing polysulfide, an epoxy group-containing polysulfide described later, or a polysulfide polymer (A) is determined by gel permeation chromatography (GPC) using tetrahydrofuran (THF) as a mobile phase. , can be obtained by converting to polyethylene glycol. Alternatively, nominal values described in catalogs, literature, etc. may be used.
- the thiol group-containing polysulfide preferably contains the disulfide structure in its main chain.
- a sealant sheet containing a polysulfide polymer (A) which is a reaction product of a thiol group-containing polysulfide containing a disulfide structure in the main chain and an epoxy compound having two or more epoxy groups in one molecule or a modified product thereof, It tends to form a cured product with good elongation.
- the number of disulfide structures contained in one molecule of thiol group-containing polysulfide may be, for example, 3 or more, 5 or more, or 10 or more as an average value (average number of disulfide groups) of the entire thiol group-containing polysulfide used. , 15 or more, or 20 or more.
- the upper limit of the average number of disulfide groups is not particularly limited, it may be, for example, 100 or less, 70 or less, or 50 or less from the viewpoint of ease of production of the sealant sheet (e.g., easiness of forming into a sheet shape). good.
- the number of thiol groups contained in the thiol group-containing polysulfide as a precursor of the polysulfide polymer (A) may be 1 or 2 or more per molecule of the thiol group-containing polysulfide. .
- a thiol group-containing polysulfide having an average number of thiol groups contained in one molecule of more than 1 is preferable from the viewpoint of easily realizing a sealant sheet suitable for improving the strength of the cured product and shortening the curing time.
- the average number of thiol groups per molecule of the thiol group-containing polysulfide used may be, for example, 1.1 or more, 1.3 or more, or 1.5 or more.
- a polysulfide having an average number of thiol groups of 2 or more can also be understood as a thiol compound (B) having 2 or more thiol groups in one molecule.
- the thiol group is preferably arranged at the end of the thiol group-containing polysulfide.
- an epoxy group-containing polysulfide polymer having terminal epoxy groups can be suitably formed.
- the thiol group-containing polysulfide used may have a thiol group at one end of the main chain, or may have a thiol group at both ends of the main chain. may further have a thiol group, or may be a mixture of any combination thereof.
- thiol group-containing polysulfides having thiol groups at both ends of the main chain that is, both terminal thiol polysulfides is particularly preferred.
- a sealant sheet containing a polysulfide polymer (A) synthesized using thiol-terminated polysulfide at both ends tends to form a cured product having both strength and elongation in a well-balanced manner.
- the proportion of both terminal thiol polysulfides may be, for example, greater than 50%, greater than 70%, or greater than 90% by weight, It may be greater than 95%, it may be greater than 98%, or it may be substantially 100%.
- Both terminal thiol polysulfides are preferably represented by the following general formula (3).
- R 1 , R 2 and R 3 are each independently an alkylene group having 1 to 4 carbon atoms, preferably an alkylene group having 1 to 3 carbon atoms, more preferably It is an alkylene group having 1 to 2 carbon atoms.
- n in the general formula (3) is, for example, the formula weight of the compound of the general formula (3) is 500 or more and 10000 or less, or 800 or more and less than 9000, or 1000 or more and less than 8000, or more than 1000 and less than 8000, or 2000 may be an integer selected to range from greater than 7500 to less than 7500.
- the compound represented by general formula (3) includes, for example, a thiol group in which R 1 is C 2 H 4 , R 2 is CH 2 and R 3 is C 2 H 4 Containing polysulfide can be preferably employed.
- n in general formula (3) may be, for example, 3-70, 5-60, 7-50, or 10-50.
- the sealant sheet includes an epoxy group-containing polysulfide polymer (AA) having two or more epoxy groups in one molecule as the polysulfide polymer (A), and two or more epoxy groups in one molecule. It contains a thiol compound (B) having a thiol group, an allyl compound (C) having two or more allyl groups in one molecule, and a photoradical generator (D).
- the sealant sheet of this aspect may or may not further contain a polysulfide polymer (A) that does not correspond to the epoxy group-containing polysulfide polymer (AA).
- the average number of epoxy groups per molecule of the epoxy group-containing polysulfide polymer (AA) (hereinafter also referred to as the average number of epoxy groups) can be, for example, about 2 or more and 20 or less. From the viewpoint of flexibility of the cured product, the average number of epoxy groups may be, for example, 15 or less, 10 or less, 7 or less, or 5 or less. In some aspects, the average number of epoxy groups may be 4 or less, or 3 or less. The average number of epoxy groups is typically 2 or more, and may be more than 2 or 2.5 or more from the viewpoint of curability and strength of the cured product. In some aspects, the average number of epoxy groups may be, for example, 3 or more, or 4 or more.
- the epoxy groups contained in the epoxy group-containing polysulfide polymer (AA) are preferably arranged at the ends of the epoxy group-containing polysulfide polymer (AA). Such an epoxy group-containing polysulfide polymer (AA) tends to form a cured product with good elongation.
- the sealant sheet disclosed herein may contain an epoxy group-containing polysulfide polymer (AA) having two or more epoxy groups at one end of the main chain as the polysulfide polymer (A), and Epoxy group-containing polysulfide polymers (AA) each having one or more epoxy groups may be included, or both of these may be included.
- the epoxy group-containing polysulfide polymer (AA) having an epoxy group at one end of the main chain may have a functional group other than the epoxy group at a terminal different from the terminal having the epoxy group.
- Functional groups other than the epoxy group may be, for example, thiol groups, amino groups, hydroxyl groups, and the like.
- the sealant sheet disclosed herein preferably contains at least an epoxy group-containing polysulfide polymer (AA) having epoxy groups at both ends of the main chain.
- the epoxy group-containing polysulfide polymer (AA) having such a structure there is a tendency to form a cured product having both strength and elongation in a well-balanced manner.
- an epoxy group-containing polysulfide polymer (AA) having one epoxy group at each end of the main chain (AA) can be preferably employed.
- Epoxy-group-containing polysulfide polymer (AA) is obtained, for example, by reacting a thiol-group-containing polysulfide as described above with an epoxy compound having two or more epoxy groups in one molecule such that the amount of epoxy groups is excessive.
- the epoxy compound may be a bifunctional epoxy compound having two epoxy groups in one molecule, or a polyfunctional epoxy compound having three or more epoxy groups in one molecule.
- An epoxy compound can be used individually by 1 type or in combination of 2 or more types.
- an epoxy compound that is liquid at room temperature can be preferably used from the viewpoint of operability when reacting with a thiol group-containing polysulfide.
- Bifunctional epoxy compounds include bisphenol A type epoxy resin, bisphenol F type epoxy resin, and hydrogenated bisphenol A type epoxy resin (that is, a structure in which the aromatic ring of bisphenol A type epoxy resin is converted to a cycloalkyl ring by hydrogenation. corresponding epoxy compounds), hydrogenated bisphenol F type epoxy resins, biphenyl type epoxy resins, aliphatic type epoxy resins (e.g., polypropylene glycol type epoxy resins, etc.), 1,6-hexanediol diglycidyl ether, polyethylene glycol diglycidyl ether , etc., but are not limited to these.
- polyfunctional epoxy compounds include novolac type epoxy resins, glycidylamine type epoxy resins, biphenyl type epoxy resins, triphenylmethane type epoxy resins, dicyclopentadiene type epoxy resins, glycerin type epoxy resins, trimethylolpropane type epoxy resins, N , N,N′,N′-tetraglycidyl-m-xylylenediamine, 1,3-bis(N,N-diglycidylaminomethyl)cyclohexane, polyglycerol polyglycidyl ether, and the like, but are not limited thereto. .
- the number of epoxy groups contained in one molecule of the polyfunctional epoxy compound is at least 3 or more, may be 4 or more, or may be 5 or more.
- the number of epoxy groups contained in one molecule of the polyfunctional epoxy compound is usually suitably 10 or less, may be 8 or less, or may be 6 or less.
- a bifunctional epoxy compound can be preferably used as the epoxy compound.
- the use of difunctional epoxy compounds can be advantageous for obtaining sealant sheets that give cured products that exhibit suitable elongation.
- a bifunctional epoxy compound can be used individually by 1 type or in combination of 2 or more types.
- an epoxy compound containing a 5- or more-membered carbon ring structure in the molecule can be preferably employed as the bifunctional epoxy compound.
- a sealant sheet using a bifunctional epoxy compound having such a structure tends to form a cured product having high strength and good elongation.
- the 5- or more-membered carbocyclic structure may be, for example, a benzene ring, a naphthalene ring, a cyclohexyl ring, or the like.
- epoxy compounds containing such a carbocyclic structure include bisphenol A type epoxy resins, bisphenol F type epoxy resins, hydrogenated bisphenol A type epoxy resins, hydrogenated bisphenol F type epoxy resins, biphenyl type epoxy resins, and the like.
- a bisphenol F type epoxy resin can be used as the bifunctional epoxy compound.
- one or more polyfunctional epoxy compounds can be used in combination with a bifunctional epoxy compound or in place of a bifunctional epoxy resin.
- Use of a polyfunctional epoxy compound can improve the strength of the cured product.
- a polyfunctional epoxy compound having a repeating unit containing an epoxy group can be used as the polyfunctional epoxy compound, and for example, a novolak type epoxy resin can be preferably used.
- the novolak type epoxy resins include phenol novolak type epoxy resins and o-cresol novolak type epoxy resins.
- Using a novolak type epoxy resin can be advantageous for obtaining a sealant sheet that gives a cured product having high strength and good elongation. Elongation of the cured product tends to be improved by using a novolak type epoxy resin having a lower molecular weight.
- a phenol novolac type epoxy resin that is liquid at room temperature can be preferably used.
- any suitable catalyst can be used as long as it does not significantly impair the effects obtained by the techniques disclosed herein.
- a known basic catalyst such as 2,4,6-triaminomethylphenol, triethylamine, 1,8-diazabicyclo[5.4.0]undec-7-ene can be appropriately selected and used.
- the amount used is not particularly limited, and is set so that the catalytic function is appropriately exhibited. can do.
- the amount of the basic catalyst used is, for example, 1 part by weight or less with respect to 100 parts by weight of the total amount of the thiol group-containing polysulfide and the epoxy compound having two or more epoxy groups in one molecule. It is usually appropriate to make it 0.5 parts by weight or less, and it may be 0.2 parts by weight or less, 0.1 parts by weight or less, or 0.08 parts by weight or less.
- the amount of the basic catalyst used with respect to the total amount of 100 parts by weight may be, for example, 0.07 parts by weight or less, may be 0.05 parts by weight or less, or may be 0.03 parts by weight or less, It may be 0.02 parts by weight or less.
- the lower limit of the amount of the basic catalyst to be used with respect to the total amount of 100 parts by weight is not particularly limited.
- the above reaction can proceed by mixing a thiol group-containing polysulfide, an epoxy compound having two or more epoxy groups in one molecule, and an optional catalyst in a suitable reaction vessel.
- a thiol group-containing polysulfide, a difunctional epoxy compound, a multifunctional epoxy compound, and a catalyst are mixed in a suitable reaction vessel.
- the method of supplying each material to the reaction vessel and the order of mixing are not particularly limited, and can be selected so as to form an appropriate reaction product.
- the conditions for the above reaction can be appropriately set within a range that does not significantly impair the effects obtained by the techniques disclosed herein.
- the reaction can proceed at a reaction temperature of, for example, 0°C to 120°C, preferably 5°C to 120°C, more preferably 10°C to 120°C.
- the reaction temperature may be, for example, 20°C to 100°C, 30°C to 100°C, or 40°C to 100°C, It may be 60°C to 100°C.
- the reaction time is not particularly limited, and can be selected, for example, from the range of 10 minutes to 720 hours (preferably 1 hour to 240 hours).
- the reaction comprises a first heating step carried out at a temperature of, for example, 60°C to 120°C (preferably 70°C to 110°C) and a and a second heating step performed at a temperature of , in this order.
- a first heating step carried out at a temperature of, for example, 60°C to 120°C (preferably 70°C to 110°C) and a and a second heating step performed at a temperature of , in this order.
- the second heating step is preferably performed at a temperature lower than that of the first heating step.
- the heating time in the first heating step can be, for example, 10 minutes or longer, usually 30 minutes or longer, and may be 1 hour or longer.
- the heating time in the first heating step can be selected, for example, from the range of 10 minutes to 24 hours (preferably 30 minutes to 12 hours, more preferably 1 hour to 6 hours).
- the heating time in the second heating step can be, for example, 3 hours or longer, usually 6 hours or longer, and may be 24 hours or longer.
- the heating time in the second heating step can be selected, for example, from the range of 3 hours to 720 hours (preferably 48 hours to 500 hours, more preferably 72 hours to 300 hours).
- the heating time in the second heating step is preferably longer than the heating time in the first heating step. Note that the heating process may be divided into three or more stages and performed stepwise.
- the ratio of the thiol group-containing polysulfide used and the epoxy compound is included in the epoxy compound with respect to the total number of thiol groups contained in the thiol group-containing polysulfide.
- the ratio of the total number of epoxy groups that is, the equivalent ratio of epoxy groups/thiol groups (hereinafter also referred to as epoxy/thiol ratio) can be set to a value greater than one.
- the epoxy/thiol ratio can be, for example, 1.05 or greater, and can be 1.1 or greater.
- the epoxy/thiol ratio may be, for example, greater than 1.2, greater than 1.4, greater than 1.5, or greater than 1.7. It's okay. Also, the epoxy/thiol ratio can be, for example, less than 7.0, it can be less than 5.0, it can be less than 4.5, it can be less than 4.0. In some embodiments, the epoxy/thiol ratio may be, for example, less than 3.5, may be less than 3.2, may be less than 3.0, may be less than 2.5, from the viewpoint of elongation improvement of the cured product, etc. , less than 2.0, or less than 1.8.
- the amount of the epoxy compound having two or more epoxy groups in one molecule is not particularly limited.
- the amount of the epoxy compound used can be set, for example, so as to achieve any of the epoxy/thiol ratios described above.
- the amount of the epoxy compound used can be, for example, 1 part by weight or more, and usually 3 parts by weight or more, per 100 parts by weight of the thiol group-containing polysulfide. It may be 5 parts by weight or more, or may be 7 parts by weight or more.
- the amount of the epoxy compound used relative to 100 parts by weight of the thiol group-containing polysulfide can be, for example, 50 parts by weight or less, usually 30 parts by weight or less, or even 20 parts by weight or less. It may be 15 parts by weight or less.
- the sealant sheet disclosed herein comprises a thiol group-containing polysulfide polymer (AB) having two or more thiol groups in one molecule as the polysulfide polymer (A), and two or more allyl groups in one molecule. and an allyl compound (C) having and a photoradical generator (D).
- the sealant sheet of this aspect may or may not further contain a polysulfide polymer (A) that does not correspond to the thiol group-containing polysulfide polymer (AB).
- the sealant sheet of the above aspect may or may not further contain a thiol compound (B) that does not correspond to the thiol group-containing polysulfide polymer (AB).
- the sealant sheet disclosed herein includes a thiol group-containing polysulfide polymer (AB) having two or more thiol groups in one molecule as the polysulfide polymer (A), and It contains a thiol compound (B) having two or more thiol groups, an allyl compound (C) having two or more allyl groups in one molecule, and a photoradical generator (D).
- the average number of thiol groups in the thiol group-containing polysulfide polymer (AB) is typically 2 or more, and may be greater than 2.
- the upper limit of the average number of thiol groups is not particularly limited, but from the viewpoint of the flexibility of the cured product, it is usually suitable to be 10 or less, 7 or less, 5 or less, 4 or less, 3 or less, 2 .8 or less or 2.4 or less.
- the thiol group contained in the thiol group-containing polysulfide polymer (AB) is preferably arranged at the end of the thiol group-containing polysulfide polymer (AB).
- Such a thiol group-containing polysulfide polymer (AB) tends to form a cured product with good elongation.
- a thiol group-containing polysulfide polymer (AB) having one or more thiol groups at both ends of the main chain is preferred, and a thiol group-containing polysulfide polymer (AB) having one thiol group at both ends of the main chain is more preferred. preferable.
- the thiol group-containing polysulfide polymer (AB) having such a structure there is a tendency to form a cured product having both strength and elongation in a well-balanced manner.
- the thiol group-containing polysulfide polymer (AB) for example, the above-mentioned epoxy group-containing polysulfide polymer (AA) and a thiol compound having two or more thiol groups in one molecule are mixed together so that the amount of thiol groups is excessive. It is possible to use those obtained by reacting as follows.
- the thiol compound may be a bifunctional thiol compound having two thiol groups in one molecule, or a polyfunctional thiol compound having three or more thiol groups in one molecule.
- a thiol compound can be used individually by 1 type or in combination of 2 or more types.
- a bifunctional thiol compound can be preferably used from the viewpoint of obtaining a sealant sheet that gives a cured product exhibiting suitable elongation.
- 50% by weight or more, 70% by weight or more, or 90% by weight or more of the thiol compounds to be reacted with the epoxy groups of the epoxy group-containing polysulfide polymer (AA) can be bifunctional thiol compounds.
- a bifunctional thiol compound alone may be used as the thiol compound.
- the thiol compound to be reacted with the epoxy group-containing polysulfide polymer (AA) for example, one or more selected from materials that can be used as the thiol compound (B) described later can be used.
- the reaction between the epoxy group-containing polysulfide polymer (AA) and the thiol compound can proceed in the same manner as the reaction between the thiol group-containing polysulfide and the epoxy compound described above.
- the ratio of the epoxy group-containing polysulfide used and the thiol compound used is included in the thiol compound with respect to the total number of epoxy groups contained in the epoxy group-containing polysulfide.
- the ratio of the total number of thiol groups can be set to be less than one.
- the epoxy/thiol ratio can be, for example, 0.95 or less, 0.9 or less, or 0.85 or less.
- the epoxy/thiol ratio may be, for example, 0.1 or more, and usually 0.2 or more is suitable.
- the epoxy/thiol ratio may be, for example, 0.3 or more, 0.5 or more, 0.6 or more, or 0.7 or more from the viewpoint of elongation improvement of the cured product. .
- the amount of the thiol compound having two or more thiol groups in one molecule is not particularly limited.
- the amount of the thiol compound used can be set, for example, so as to achieve any of the epoxy/thiol ratios described above.
- the amount of the thiol compound used can be, for example, 1 part by weight or more, and usually 3 parts by weight or more, per 100 parts by weight of the epoxy group-containing polysulfide. It may be 5 parts by weight or more, or may be 7 parts by weight or more.
- the amount of the thiol compound used relative to 100 parts by weight of the epoxy group-containing polysulfide can be, for example, 50 parts by weight or less, usually 30 parts by weight or less, or even 20 parts by weight or less. It may be 15 parts by weight or less.
- the thiol group-containing polysulfide polymer (AB) for example, the thiol group-containing polysulfide (preferably both terminal thiol polysulfides) described above as a material that can be used as a precursor of the polysulfide polymer (A) may be used.
- thiol compound (B) contained in the sealant sheet disclosed herein a compound having two or more thiol groups in one molecule can be used without particular limitation.
- trimethylolpropane tristhiopropionate also known as trimethylolpropane tris(3-mercaptopropionate)
- pentaerythritol tetrakisthiopropionate ethylene glycol bisthioglycolate, 1,4-butanediol bis Thioglycolate
- trimethylolpropane tristhioglycolate pentaerythrol tetrakisthioglycolate
- di(2-mercaptoethyl) ether 1,4-butanedithiol
- 1,5-dimercapto-3-thiapentane 1,8- dimercapto-3,6-dioxaoctane (alias: 3,6-dioxa
- Examples of commercially available thiol compounds (B) include jER Mate QX11, QX12, jER Cure (registered trademark) QX30, QX40, QX60, QX900 and Capcure CP3-800 manufactured by Mitsubishi Chemical Corporation; Yodo Chemical Co., Ltd. OTG, EGTG, TMTG, PETG, 3-MPA, TMTP, PETP; TEMPIC, TMMP, PEMP, PEMP-II-20P, DPMP manufactured by Sakai Chemical Co., Ltd.; Karenz MT (registered trademark) PE1 manufactured by Showa Denko K.K.
- the average number of thiol groups of the thiol compound (B) contained in the sealant sheet can be, for example, about 2 or more and 10 or less. From the viewpoint of flexibility of the cured product, the average number of thiol groups may be, for example, 7 or less, 5 or less, 4 or less, or less than 4. In some aspects, the average number of thiol groups may be 3 or less, 2.5 or less, or 2.2 or less.
- the thiol compound (B) only one or two or more bifunctional thiol compounds may be used. With such a configuration, it is easy to obtain a sealant sheet that gives a cured product exhibiting suitable elongation.
- Examples of the thiol compound (B) include a compound having a primary thiol group (hereinafter also referred to as a primary thiol compound), a compound having a secondary thiol group (secondary thiol compound), a compound having a tertiary thiol group (3 class thiol compounds) can be used.
- a primary thiol compound can be preferably employed from the viewpoint of curability when the sealant sheet is used.
- a secondary or higher thiol compound that is, a secondary thiol compound and/or a tertiary thiol compound
- a secondary or higher thiol compound that is, a secondary thiol compound and/or a tertiary thiol compound
- a thiol compound having two primary thiol groups in one molecule may be referred to as a primary bifunctional thiol compound, and a thiol compound having two secondary thiol groups in one molecule is a secondary bifunctional They are sometimes called thiol compounds.
- a primary thiol compound and a secondary or higher thiol compound can be used in combination as the thiol compound (B).
- the weight ratio of the primary thiol compound to the total weight of the primary thiol compound and the secondary or higher thiol compound is not particularly limited, and may be, for example, 5% by weight or more, preferably 15% by weight or more, or more. It is preferably 25% by weight or more, and may be 35% by weight or more, and may be, for example, 95% by weight or less, preferably 75% by weight or less, and may be 60% by weight or less. , 45% by weight or less.
- the thiol compound (B) one having a thiol equivalent in the range of 45 g/eq to 450 g/eq is preferably used in consideration of the balance between the preservability of the sealant sheet before use and the curability at the time of use. obtain.
- the thiol equivalent may be, for example, 60 g/eq or more, 70 g/eq or more, or 80 g/eq or more, and may be, for example, 350 g/eq or less, 250 g/eq or less, or 200 g/eq. eq or less, or 150 g/eq or less.
- the storage stability before use tends to improve, but the curability during use tends to decrease.
- the sum of the products of the thiol equivalents and weight fractions of each thiol compound (B) is preferably within the above range.
- the thiol equivalent means the number of grams of a compound containing one equivalent of a thiol group, and can be measured by an iodometric titration method. Alternatively, nominal values described in catalogs, literature, etc. may be used.
- allyl compound (C) contained in the sealant sheet disclosed herein any compound having two or more allyl groups in one molecule can be used without particular limitation.
- allyl compound (C) disclosed herein allyl ether, allyl ester, and the like can be preferably used.
- allyl compounds (C) disclosed herein include diallyl phthalate, diallyl isophthalate, diallyl terephthalate, diallyl fumarate, diallyl adipate, diallyl hexahydrophthalate, 1,3-diallyloxy-2- bifunctional allyl compounds such as propanol; trifunctional allyl compounds such as triallyl isocyanurate, triallyl cyanurate, triallyl citrate, and triallyl trimellitate; and tetrafunctional allyl compounds such as pentaerythritol tetraallyl ether. Not limited.
- the ratio of allyl group equivalents to thiol group equivalents contained in the sealant sheet is not particularly limited.
- the allyl/thiol ratio of the sealant sheet may be, for example, approximately 0.1 or more and 10 or less, 0.2 or more and 5 or less, 0.3 or more and 3 or less, or 0.5 or more and 2 or less. good.
- the allyl/thiol ratio is equal to or more than the lower limit and equal to or less than the upper limit, a cured product having both strength and elongation in a well-balanced manner tends to be formed.
- the allyl/thiol ratio can be, for example, greater than or equal to 0.6, greater than or equal to 0.7, or greater than or equal to 0.8, and less than or equal to 1.7, less than or equal to 1.5, or less than or equal to 1.2. you can
- the amount of the thiol compound (B) contained in the sealant sheet is not particularly limited.
- the amount of the thiol compound (B) contained in the sealant sheet can be set, for example, so as to achieve any of the allyl/thiol ratios described above.
- the amount of the thiol compound (B) that does not correspond to the thiol group-containing polysulfide polymer (AB) relative to 100 parts by weight of the polysulfide polymer (A) can be, for example, 0.05 parts by weight or more.
- the amount of the allyl compound (C) contained in the sealant sheet is not particularly limited.
- the amount of the allyl compound (C) contained in the sealant sheet can be set, for example, so as to achieve any of the allyl/thiol ratios described above.
- the amount of the allyl compound (C) relative to 100 parts by weight of the polysulfide polymer (A) may be, for example, 0.05 parts by weight or more, may be 0.1 parts by weight or more, or may be 0.3 parts by weight or more. It may be 0.5 parts by weight or more, may be 10 parts by weight or less, may be 5 parts by weight or less, may be 3 parts by weight or less, or may be 2 parts by weight or less. .
- Photoradical generator (D) As the photoradical generator (D), one that generates radicals by light irradiation is used. In some preferred embodiments, the photoradical generator (D) is an intramolecularly cleavable photoinitiator. Examples of photoradical generators (D) include alkylphenone-based photopolymerization initiators, acylphosphine oxide-based photopolymerization initiators, titanocene compound-based photopolymerization initiators, acetophenone-based photopolymerization initiators, and benzoin ether-based photopolymerization initiators. agents and the like. A photoradical generator can be used individually by 1 type or in combination of 2 or more types.
- alkylphenone-based photopolymerization initiators include 2-methyl-1-[4-(methylthio)phenyl]-2-morpholinopropan-1-one (eg, trade name "Omnirad 907" manufactured by IGM Resins). ), 2,2-dimethoxy-2-phenylacetophenone (eg, trade name “Omnirad 651” manufactured by IGM Resins), and the like.
- acylphosphine oxide-based photopolymerization initiators include bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide (eg, trade name “Irgacure 819” manufactured by BASF), bis(2,4,6 -trimethylbenzoyl)-2,4-di-n-butoxyphenylphosphine oxide, 2,4,6-trimethylbenzoyldiphenylphosphine oxide (for example, trade name "Lucirin TPO" manufactured by BASF), bis(2,6- dimethoxybenzoyl)-2,4,4-trimethylpentylphosphine oxide and the like.
- bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide eg, trade name “Irgacure 819” manufactured by BASF
- bis(2,4,6 -trimethylbenzoyl)-2,4-di-n-butoxyphenylphosphine oxide 2,4,6-trimethylbenzoyl
- titanocene compound-based photopolymerization initiators include bis(2,4-cyclopentadienyl)bis[2,6-difluoro-3-(1-pyryl)phenyl]titanium (IV) (eg, BASF product name “Irgacure 784”), etc.
- acetophenone-based photopolymerization initiators include 1-hydroxycyclohexyl-phenyl-ketone (eg, trade name “Irgacure 184” manufactured by BASF), 4-phenoxydichloroacetophenone, 4-t-butyl-dichloroacetophenone, 1-[4-(2-hydroxyethoxy)-phenyl]-2-hydroxy-2-methyl-1-propan-1-one (for example, trade name "Irgacure 2959” manufactured by BASF), 2-hydroxy-2 -methyl-1-phenyl-propan-1-one (eg, trade name "Darocure 1173” manufactured by BASF), methoxyacetophenone, and the like.
- 1-hydroxycyclohexyl-phenyl-ketone eg, trade name “Irgacure 184” manufactured by BASF
- 4-phenoxydichloroacetophenone 4-t-butyl-dichloroacetophenone
- benzoin ether photoinitiators include benzoin ethers such as benzoin methyl ether, benzoin ethyl ether, benzoin propyl ether, benzoin isopropyl ether, and benzoin isobutyl ether, and substituted benzoin ethers such as anisole methyl ether.
- alkylphenone-based photopolymerization initiators acylphosphine oxide-based photopolymerization initiators and titanocene compounds can effectively promote radical addition reactions between thiol groups and allyl groups by radicals generated by light irradiation.
- Systemic photoinitiators are preferred.
- the usage amount of the photoradical generator (D) can be set so as to obtain the desired usage effect.
- the amount of the photoradical generator (D) used is, for example, 0.01 with respect to 100 parts by weight of the total amount of the polysulfide polymer (A), the thiol compound (B) and the allyl compound (C). It can be 0.1 part by weight or more from the viewpoint of enhancing the curability of the sealant sheet, and may be 0.5 parts by weight or more, 1 part by weight or more, or 1.5 parts by weight. It can be more than part.
- the amount of the photoradical generator (D) used is usually preferably 10 parts by weight or less, preferably 7 parts by weight or less, with respect to the total amount of 100 parts by weight, from the viewpoint of raw material costs and the like. More preferably, it may be 5 parts by weight or less, 4 parts by weight or less, 3 parts by weight or less, or 2.5 parts by weight or less.
- the sealant sheets disclosed herein may contain a sensitizer.
- a sensitizer By using a sensitizer, it is possible to increase the utilization efficiency of the irradiated light and improve the sensitivity of the photo-radical generator (D).
- the photosensitizer can be appropriately selected from known materials and used.
- Non-limiting examples of photosensitizers include benzophenone, 4-methylbenzophenone, 3-benzoylbiphenyl, 4-(4-methylphenylthio)benzophenone, methyl 2-benzoylbenzoate, 4-phenylbenzophenone, 4,4 '-bis(dimethoxy)benzophenone, 4,4'-bis(dimethylamino)benzophenone, 4,4'-bis(diethylamino)benzophenone, 2-benzoylbenzoic acid methyl ester, 2-methylbenzophenone, 3-methylbenzophenone, 3 ,3'-dimethyl-4-methoxybenzophenone, benzophenone derivatives such as 2,4,6-trimethylbenzophenone; Thioxanthone derivatives such as 4-dimethylthioxanthone, 2,4-diethylthioxanthone, 1-chloro-4-propoxythioxanthone, and 2,4-diethylthioxanthen-9
- the amount used when using a photosensitizer can be set so as to obtain the desired sensitization effect.
- the amount of the photosensitizer used is For example, it may be 0.001 parts by weight or more, 0.005 parts by weight or more, 0.01 parts by weight or more, or 0.05 parts by weight or more.
- the amount of the photosensitizer used is, for example, 0.002 parts by weight with respect to 100 parts by weight of the allyl compound (C).
- the upper limit of the amount of the photosensitizer used is not particularly limited, but from the viewpoint of the storage stability of the sealant sheet, when the sealant sheet contains the thiol compound (B) and the allyl compound (C), the thiol compound (B) and 10 parts by weight or less is usually suitable for 100 parts by weight of the total amount with the allyl compound (C), and it may be 5 parts by weight or less, 1 part by weight or less, or 0.5 parts by weight or less, It may be 0.3 parts by weight or less.
- the amount of the photosensitizer used is usually 20 parts by weight or less with respect to 100 parts by weight of the allyl compound (C). It may be 10 parts by weight or less, 2 parts by weight or less, 1 part by weight or less, or 0.6 parts by weight or less.
- the sealant sheet disclosed herein does not contain a photosensitizer in a preferred embodiment.
- the sealant sheet disclosed herein may further contain any compound that can help suppress radical addition reactions between thiol groups and allyl groups as long as other properties are not significantly impaired. Use of such a compound can enhance the shelf life of the sealant sheet before use.
- the storage stabilizers can be, for example, organic acids, inorganic acids, and oligomers, polymers, borate esters, phosphate esters containing acidic groups in the molecule, which are liquid or solid at room temperature, and which contain functional groups other than acidic groups. You may have a group.
- Examples include, but are not limited to, sulfuric acid, acetic acid, adipic acid, tartaric acid, fumaric acid, barbituric acid, boric acid, pyrogallol, phenolic resins, carboxylic acid anhydrides, and the like.
- the storage stabilizers may be used singly or in appropriate combination of two or more.
- the amount of the storage stabilizer to be used is not particularly limited, and can be set so as to obtain the desired effect.
- Suitable examples of storage stabilizers include borate esters and phosphate esters.
- Borate esters are borate esters that are liquid or solid at room temperature.
- phosphates include but are not limited to: Examples of phosphates include ethyl phosphate, butyl phosphate, propyl phosphate, 2-ethylhexyl phosphate, dibutyl phosphate, di(2-ethylhexyl) phosphate, oleyl phosphate, and ethyl diethyl phosphate. include, but are not limited to.
- the sealant sheet disclosed herein, in one preferred embodiment, does not contain a storage stabilizer.
- the sealant sheet disclosed herein may optionally contain a filler. This can improve one or both of the breaking strength and elongation at break of the cured product. Fillers can also help adjust the storage modulus of the sealant sheet. In addition, appropriate use of fillers can improve the shape retention and workability of the sealant sheet.
- the filler to be used is not particularly limited, and any appropriate filler can be used as long as it does not significantly impair the effects obtained by the technology disclosed herein.
- a filler can be used individually by 1 type or in combination of 2 or more types.
- filler materials include talc, silica, glass, carbon black, alumina, clay, mica, aluminum hydroxide, magnesium hydroxide, calcium carbonate, magnesium carbonate, barium sulfate, titanium dioxide, barium titanate, and titanium.
- filler materials include, but are not limited to, strontium oxide, calcium titanate, magnesium titanate, bismuth titanate, boron nitride, aluminum borate, barium zirconate, calcium zirconate, and the like.
- Preferred examples include talc, silica, glass and calcium carbonate.
- the content of the filler is not particularly limited, and can be selected so as to obtain suitable properties.
- the content of the filler may be, for example, 1% by weight or more, or 5% by weight or more, of the entire sealant sheet, or may be 10% by weight or more, or may be 15% by weight or more from the viewpoint of obtaining a higher effect of use. It may be 20% by weight or more, or 25% by weight or more.
- the content of the filler can be, for example, less than 50% by weight of the entire sealant sheet, and from the viewpoint of improving the moldability into the sheet shape and the elongation of the cured product, it is usually less than 40% by weight. suitable and may be less than 35% by weight.
- the filler content may be less than 30 wt%, or less than 25 wt%.
- the average particle size of the filler is not particularly limited.
- the average particle size is usually suitably 100 ⁇ m or less, preferably 50 ⁇ m or less. A smaller average particle size tends to improve the effect of improving one or both of the breaking strength and elongation at break of the cured product.
- the average particle size of the filler may be, for example, 30 ⁇ m or less, 20 ⁇ m or less, 15 ⁇ m or less, 10 ⁇ m or less, or 5 ⁇ m or less.
- the average particle size of the filler may be, for example, 0.1 ⁇ m or more, 0.2 ⁇ m or more, 0.5 ⁇ m or more, or 1 ⁇ m or more. A not-too-small average particle size can be advantageous from the viewpoint of handling and dispersibility of the filler.
- the average particle size of the filler means the particle size at which the volume-based cumulative particle size is 50% in the particle size distribution obtained by the measurement based on the laser diffraction/scattering method, that is, the 50% volume average particle size. (50% median diameter).
- a filler made of a material having a refractive index in the range of 1.56 or more and less than 1.62 can be preferably used.
- a glass filler having a refractive index within the above range can be used.
- the above refractive index range is a range that is equal to or approximates the refractive index of the polysulfide polymer (A) (typically about 1.60). Therefore, a filler having a refractive index within the above range tends to suppress a decrease in the transmittance of the sealant sheet due to blending the filler, compared to a filler having a refractive index outside the above range.
- the transmittance of the sealant sheets disclosed herein may be, for example, greater than 5%, greater than 10%, greater than 15%, or greater than 20%.
- the upper limit of transmittance is not particularly limited.
- the transmittance of the sealant sheet disclosed herein may be 100%, or may be 80% or less, 60% or less, or 40% or less from a practical standpoint.
- the sealant sheet disclosed herein can also be preferably implemented in a mode in which the transmittance is 30% or less, 20% or less, or 15% or less.
- the transmittance of the sealant sheet can be obtained by measuring the spectrum of the sealant sheet with a thickness of 0.2 mm using a UV-vis spectrum measuring device (Shimadzu Corporation, UV-2550) or its equivalent.
- the transmittance for example, the transmittance at a wavelength of 365 nm can be adopted.
- the sealant sheet disclosed herein is a combination of a filler having a refractive index in the range of 1.56 or more and less than 1.62 (for example, glass filler) and a filler having a refractive index outside the above range (for example, talc).
- the ratio of the filler having a refractive index within the above range to the total amount of filler contained in the sealant sheet may be, for example, 10% by weight or more, may be 25% by weight or more, and preferably 45% by weight or more. It may be 60% by weight or more, 85% by weight or more, or 100% by weight.
- a filler made of a material having a refractive index in the range of 1.56 to 1.61 or in the range of 1.57 to 1.60 can be more preferably employed.
- the refractive index can be measured using generally known techniques such as the minimum deflection angle method, the critical angle method, and the V-block method. The measurement can be performed using, for example, a multi-wavelength Abbe refractometer DR-M4 (manufactured by ATAGO). Alternatively, nominal values described in catalogs, literature, etc. may be used.
- the sealant sheet disclosed here may contain other optional components within the range that does not significantly impair the effects obtained by the technology disclosed here.
- optional components include colorants such as dyes and pigments, dispersants, plasticizers, softeners, flame retardants, antioxidants, ultraviolet absorbers, antioxidants, light stabilizers, and the like. , but not limited to.
- the sealant sheet disclosed herein may further contain a polymer or oligomer (hereinafter also referred to as an arbitrary polymer) other than the above, for the purpose of improving adhesion to the sealing target area, for example.
- a polymer or oligomer hereinafter also referred to as an arbitrary polymer
- the content of the optional polymer is preferably 10 parts by weight or less, more preferably 5 parts by weight or less, relative to 100 parts by weight of the polysulfide polymer (A). It is more preferably 1 part by weight or less.
- the sealant sheet may be substantially free of such optional polymers. In the present specification, “substantially free of a certain component” means that the component is at least intentionally not contained unless otherwise specified.
- the content of the organic solvent based on the weight of the sealant sheet may be, for example, 5% or less, 2% or less, 1% or less, or 0.5% or less. It may be substantially free of organic solvents.
- the content of the organic solvent may be 0%.
- the organic solvent is intended to react with other components in the sealant sheet (in particular, epoxy group-containing polysulfide and a curing agent that can be used as necessary), such as toluene, cyclohexanone, trichloroethane, and the like. It refers to ingredients that are not
- the sealant sheet disclosed herein may contain a thiol compound (B) (hereinafter also referred to as a low-molecular-weight thiol compound) having an Mw of 1000 or less, preferably 600 or less, more preferably 400 or less.
- the content of the low-molecular-weight thiol compound may be 0.1% by weight or more, or 0.3% by weight or more, based on the weight of the total amount of the thiol compound (B) and the polysulfide polymer (A). , or 0.5% by weight or more.
- the low molecular weight thiol compound can serve to increase the tackiness of the sealant sheets disclosed herein.
- the term "temporary fixability" refers to the property of preventing the sealant sheet from being lifted from the sealing target area or being displaced until the sealant sheet placed at the sealing target area is cured.
- the low-molecular-weight thiol compound reacts with light irradiation and is incorporated into the cured product.
- the content of the low molecular weight thiol compound may be less than 0.1% by weight of the total amount of the thiol compound (B) and the polysulfide polymer (A), based on weight. It may be less than 05% by weight, and may be substantially free.
- the sealant sheet disclosed here can have a tack on the surface and can be temporarily fixed to the sealing target part even in such an aspect.
- a release liner can be used in the preparation of the sealant sheet disclosed herein (for example, molding into a sheet shape), storage of the sealant sheet before use, distribution, shape processing, placement at a sealing target site, and the like. .
- the release liner is not particularly limited.
- a release liner or the like made of a low-adhesion material such as polypropylene can be used.
- the release treatment layer may be formed by surface-treating the liner base material with a release treatment agent such as a silicone-based, long-chain alkyl-based, fluorine-based, or molybdenum sulfide release agent.
- the sealant sheet disclosed herein contains a polysulfide polymer (A), a thiol compound (B) (for example, the low-molecular-weight thiol compound described above), and an allyl compound (C) in combination.
- a polysulfide polymer (A) is prepared; a thiol compound (B), an allyl compound (C), a photoradical generator (D) and a filler are added to the polysulfide polymer (A). and mixing the resulting mixture into a sheet shape.
- the polysulfide polymer (A) is a reaction product of a thiol group-containing polysulfide, a bifunctional epoxy compound, and a polyfunctional epoxy compound
- preparing the mixture includes the thiol group-containing polysulfide and the bifunctional epoxy compound. and a polyfunctional epoxy compound; and adding and mixing a thiol compound (B), an allyl compound (C), a photoradical generator (D) and a filler to the reaction product ; in that order.
- fillers may be mixed together during preparation of the reaction product.
- the above description regarding the reaction between the thiol group-containing polysulfide and the epoxy compound having two or more epoxy groups in one molecule can be applied, so redundant description will be omitted.
- Examples of devices that can be used for mixing the reaction product and the additive components include, for example, Banbury Mixers, kneaders, closed kneaders such as two-roll mills and three-roll mills or batch kneaders; and continuous kneaders such as single-screw extruders and twin-screw extruders, but are not limited thereto.
- known sheet forming methods such as press molding, calendar molding, and melt extrusion molding can be used alone or in combination as appropriate.
- the above press molding may be normal pressure press or vacuum press.
- vacuum press molding or calender molding can be preferably applied from the viewpoints of preventing entrainment of air bubbles into the sheet and suppressing thermal denaturation of the mixture.
- the obtained sealant sheet is stored and processed (for example, slitting to a predetermined width, processing from a roll to a sheet, or a predetermined shape in the form of a sealant sheet with a release liner as shown in FIG. 1 or 2). punching, etc.), transportation, etc. can be performed.
- the sealant sheet disclosed here contains a combination of a thiol group-containing polysulfide polymer (AB) and an allyl compound (C).
- a thiol group-containing polysulfide polymer (AB) is prepared; an allyl compound (C), a photoradical generator (D) and a filler are added to the thiol group-containing polysulfide polymer (AB). and mixing the resulting mixture into a sheet shape.
- thiol group-containing polysulfide polymer (AB) is a thiol-modified epoxy group-containing polysulfide polymer
- preparing the mixture is performed by replacing the thiol group of a compound having two or more thiol groups in one molecule with the epoxy group.
- Preparing a thiol group-containing polysulfide polymer (AB) by reacting with the epoxy group of the containing polysulfide polymer; adding and mixing; in that order.
- fillers may be mixed together during preparation of the reaction product.
- the material of the portion to be sealed using the sealant sheet disclosed herein is not particularly limited.
- the material may be, for example, metal, resin, composite material thereof, etc. More specifically, iron, iron alloy (carbon steel, stainless steel, chromium steel, nickel steel, etc.), aluminum, aluminum alloy, nickel , tungsten, copper, copper alloys, titanium, titanium alloys, silicon and other metal or semi-metal materials; polyolefin resins, polycarbonate resins, acrylic resins, acrylonitrile resins (PAN) and other resin materials; alumina, silica, sapphire, silicon nitride, ceramic materials such as tantalum nitride, titanium carbide, silicon carbide, gallium nitride, and gypsum; glass materials such as aluminosilicate glass, soda lime glass, soda aluminosilicate glass, and quartz glass; laminates and composites thereof; .
- Suitable examples of the metal or semimetal material include light metals such as aluminum and titanium, and alloys containing such light metals as main components.
- Examples of aluminum alloys include duralumin (eg, duralumin A2024, duralumin A2017, etc.).
- Examples of the composite include carbon fiber reinforced plastic (CFRP) and glass fiber reinforced plastic (FRP).
- the sealant sheet disclosed herein takes the form of a non-liquid (i.e., solid) sheet in a temperature range of about 25°C. no need to control
- the sealant sheet can be cut into a desired outer shape in advance and placed on the sealing target (typically, it can be attached using the tack of the sealant sheet). is.
- a roll-shaped sealing sheet may be unwound and applied to the target location, and the surplus sealing sheet may be cut off.
- the radical addition reaction between the thiol group and the allyl group contained in the sealant sheet is accelerated by irradiating with light to generate radicals from the photoradical generator (D). hardening proceeds.
- Light irradiation can be performed using a known appropriate light source such as a chemical lamp, a black light (for example, a black light manufactured by Toshiba Lighting & Technology Co., Ltd.), a metal halide lamp, and the like.
- a light source with a spectral distribution in the wavelength region of 250 nm to 450 nm can be preferably used.
- a sensitizer By including a sensitizer in the sealant sheet, the utilization efficiency of the light emitted from the light source can be increased.
- use of a sensitizer is particularly effective when using a light source having a spectral distribution in the wavelength range of 350 nm to 450 nm.
- the sealant sheet disclosed here can be used in a manner in which light irradiation is performed while being placed on a seal target location. Since the radical addition reaction between a thiol group and an allyl group (thiolene addition reaction) progresses faster than anionic polymerization or the like, curing of the sealant sheet disclosed herein tends to progress rapidly after light irradiation. According to the technology disclosed herein, even with a sealant sheet that cures in a shorter time than the time required for air bubbles to fully escape from the sealant after light irradiation, the generation and growth of air bubbles can be suppressed. Therefore, it can be suitably used for applications that require shortened sealing time and high sealing quality.
- the cured product formed from the sealant sheet disclosed herein or the cured sealant suitably has a tensile strength at break of 0.7 MPa or more, preferably 0.9 MPa or more, more preferably 1 0 MPa, and may be 1.1 MPa or more, or 1.15 MPa or more. In some embodiments, the tensile strength at break may be 1.2 MPa or higher, or 1.3 MPa or higher.
- the upper limit of the tensile strength at break is not particularly limited, but from the viewpoint of facilitating compatibility with other physical properties, it may be, for example, 3 MPa or less.
- the cured product formed from the sealant sheet disclosed herein or the cured sealant suitably has an elongation at break of 100% or more, preferably 120% or more, and may be 150% or more. It may be 200% or more, or 250% or more.
- the upper limit of the elongation at break is not particularly limited, but from the viewpoint of facilitating compatibility with other physical properties, it may be, for example, 600% or less, or 400% or less.
- the above tensile strength at break and elongation at break are measured by the following methods.
- Measurement of tensile strength at break and elongation at break One surface of a sealant sheet having a thickness of 0.2 mm is irradiated with light of 2000 mJ/cm 2 using a black light manufactured by Toshiba Lighting & Technology Corporation. After the irradiated sealant sheet is held in an environment of 25° C. for 14 days, the resulting cured product (cured sealant) is cut into a rectangular shape having a width of 10 mm and a length of 50 mm to prepare a sample piece.
- a sealant sheet molded into a sheet shape Ingredients for: polysulfide polymer (A); Thiol compound (B) having two or more thiol groups in one molecule; an allyl compound (C) having two or more allyl groups in one molecule; and a photoradical generator (D); Including sealant sheet.
- the above-mentioned epoxy group-containing polysulfide polymer (AA) is produced by reaction of both terminal thiol polysulfide having Mw 500 to 10000 having a disulfide structure in the main chain and an epoxy compound having two or more epoxy groups in one molecule.
- the sealant sheet according to (2) or (3) above which is a product.
- the bifunctional epoxy compound includes an epoxy compound containing a 5- or more-membered carbon ring structure in the molecule.
- the above-mentioned thiol group-containing polysulfide polymer (AB) is produced by reaction of both terminal epoxy polysulfide having Mw of 500 to 10000 having a disulfide structure in the main chain and a thiol compound having two or more thiol groups in one molecule.
- (12) The sealant sheet according to any one of (1) to (11) above, wherein the thiol compound (B) has a thiol equivalent of 45 g/eq to 450 g/eq.
- the allyl compound (C) is at least one selected from the group consisting of bifunctional allyl compounds and trifunctional allyl compounds.
- the photoradical generator (D) is at least one selected from the group consisting of alkylphenone-based photopolymerization initiators, acylphosphine oxide-based photopolymerization initiators, and titanocene compound-based photopolymerization initiators.
- (16) The sealant sheet according to (15) above, wherein the content of the filler is 1% by weight or more and less than 40% by weight of the entire sealant sheet.
- the sealant sheet according to (15) or (16) above, wherein the filler has an average particle size of 0.1 ⁇ m or more and 30 ⁇ m or less.
- sealant sheet according to any one of (1) to (17) above which has a storage modulus at 25° C. of 0.005 MPa or more and 0.8 MPa or less.
- the sealant sheet according to any one of (1) to (18) above having a thickness of 0.01 mm or more and 10 mm or less.
- a sealant sheet molded into a sheet shape Ingredients for: Thiol group-containing polysulfide polymer (AB) having two or more thiol groups; an allyl compound (C) having two or more allyl groups in one molecule; and a photoradical generator (D); Including sealant sheet.
- a sealant sheet with a release liner comprising: (22) preparing a polysulfide polymer (A); In the polysulfide polymer (A), a thiol compound (B) having two or more thiol groups in one molecule, an allyl compound (C) having two or more allyl groups in one molecule, a photoradical generator (D) and adding and mixing a filler; and forming the resulting mixture into a sheet form; A method of manufacturing a sealant sheet, comprising: (23) preparing a thiol group-containing polysulfide polymer (AB) having two or more thiol groups in one molecule; adding and mixing an allyl compound (C) having two or more allyl groups in one molecule, a photoradical generator (D) and a filler to the thiol group-containing poly
- Example 1 ⁇ Preparation of epoxy polysulfide polymer at both ends> Using a reaction vessel equipped with a stirrer, 90 parts of a liquid polysulfide polymer (both thiol-terminated polysulfide polymer), 7.5 parts of a bifunctional epoxy compound, 2 parts of a polyfunctional epoxy compound, and 0.08 parts of a basic catalyst are stirred. while heating at 90° C. for 3 hours. Thus, a double-ended epoxy polysulfide polymer was synthesized.
- the liquid polysulfide polymer (both thiol-terminated polysulfide polymer) is a product name Thiocol LP-55 (Mw 4000, thiol equivalent 2000 g / eq) manufactured by Toray Fine Chemicals Co., Ltd.
- the bifunctional epoxy compound is bisphenol F type manufactured by Mitsubishi Chemical Corporation.
- Epoxy resin product name jER806, epoxy equivalent 169 g / eq
- Mitsubishi Chemical Corporation phenolborac type epoxy resin product name jER152, epoxy equivalent 177 g / eq
- ⁇ Preparation of sealant sheet> After taking out the contents of the reaction vessel and allowing it to cool to room temperature, 0.5 parts of the secondary bifunctional thiol compound and 0.6 parts of the trifunctional allyl compound shown in Table 1 are added to 50 parts of the epoxy polysulfide polymer at both ends. , 1.0 part of photoradical generator A, and 20 parts of talc as a filler were added and kneaded uniformly using a two-roll mill.
- a sealant sheet according to this example was obtained by forming the resulting mixture into a sheet using a vacuum press. At that time, two types, a sheet with a thickness of 0.2 mm and a sheet with a thickness of 1 mm, were produced.
- the allyl/thiol ratio of the sealant sheet according to this example is 1.0. That is, the sum of the thiol groups (unreacted) contained in the epoxy polysulfide polymer at both ends used to prepare the sealant sheet according to this example and the thiol groups (unreacted) contained in the secondary bifunctional thiol compound added to this polymer The number is almost the same as the number of allyl groups (unreacted) contained in the trifunctional allyl compound added to the polymer.
- the number of thiol groups contained in the epoxy polysulfide polymer at both ends is calculated from the number of thiol groups NT calculated from the thiol equivalent of the liquid polysulfide polymer used in the synthesis of the polymer and the amount used, the bifunctional epoxy compound used and It is obtained by subtracting the number of epoxy groups, NE , calculated from the epoxy equivalent weight and amount used of each polyfunctional epoxy compound.
- Example 2 ⁇ Preparation of thiol-terminated polysulfide polymer> Using a reaction vessel equipped with a stirrer, 90 parts of a liquid polysulfide polymer (both thiol-terminated polysulfide polymer), 10 parts of a bifunctional epoxy compound, 2 parts of a polyfunctional epoxy compound, and 0.08 parts of a basic catalyst are stirred. Heated at 90° C. for 3 hours. Then, 4 parts of a bifunctional thiol compound was added, and the mixture was heated at 90° C. for 1.5 hours while stirring. Thus, a polysulfide polymer with thiol ends at both ends was synthesized.
- a liquid polysulfide polymer both thiol-terminated polysulfide polymer
- 10 parts of a bifunctional epoxy compound 2 parts of a polyfunctional epoxy compound
- 0.08 parts of a basic catalyst 0.08 parts
- liquid polysulfide polymer (both thiol-terminated polysulfide polymer) is a product name Thiocol LP-55 (Mw 4000, thiol equivalent 2000 g / eq) manufactured by Toray Fine Chemicals Co., Ltd., and the bifunctional epoxy compound is bisphenol F type manufactured by Mitsubishi Chemical Corporation.
- Epoxy resin product name jER806, epoxy equivalent 169 g / eq
- Mitsubishi Chemical Corporation phenolborac type epoxy resin product name jER152, epoxy equivalent 177 g / eq
- a polyfunctional epoxy compound Tokyo Kasei as a basic catalyst 2,4,6-triaminomethylphenol manufactured by Tokyo Kasei Co., Ltd.
- ⁇ Preparation of sealant sheet> After taking out the contents of the reaction vessel and allowing it to cool to room temperature, 0.5 parts of secondary bifunctional thiol shown in Table 1, 0.6 parts of trifunctional allyl compound, and 50 parts of epoxy polysulfide polymer at both ends are added. 1.0 part of photo-radical generator A and 20 parts of talc as a filler were added and uniformly kneaded using a two-roll mill. A sealant sheet according to this example was obtained by forming the resulting mixture into a sheet using a vacuum press. At that time, two types, a sheet with a thickness of 0.2 mm and a sheet with a thickness of 1 mm, were produced.
- the sealant sheets of Examples 1 and 2 were measured or evaluated as follows. (Measurement of storage modulus) A sealant sheet with a thickness of 1 mm is punched into a disk shape with a diameter of 8 mm, sandwiched between parallel plates, and a viscoelasticity tester (manufactured by TA Instruments Japan, model name "ARES G2") is used to measure the temperature. Storage modulus G' was measured under conditions of 25°C, frequency of 1 Hz, and strain of 0.5%. As a result, the storage elastic moduli G' of the sealant sheets according to Examples 1 and 2 were both in the range of 0.005 MPa to 0.8 MPa.
- the sealants of Comparative Examples 1 to 4 were evaluated as follows. About 1 g of the sealant was sampled and dropped onto a 38 ⁇ m thick transparent PET film (Mitsubishi Polyester, Diafoil MRF38), and a 38 ⁇ m thick transparent PET film (Mitsubishi Polyester, Diafoil MRF38) was further placed thereon. I covered it and sandwiched the sealant. Next, light irradiation of 2000 mJ/cm 2 was performed from one side of the sealant using a black light manufactured by Toshiba Lighting & Technology Co., Ltd. After that, the tester visually observed the appearance of the sealant from above the transparent PET film to confirm whether or not air bubbles were present inside the sealant.
- a 38 ⁇ m thick transparent PET film Mitsubishi Polyester, Diafoil MRF38
- the sealant sheets according to Examples 1 and 2 were both formed into a flexible sheet shape before light irradiation, and the sheet shape was stably maintained.
- the sealant sheets of Examples 1 and 2 exhibited good curability when irradiated with light, and no air bubbles were observed inside the sheets after curing.
- a sealant sheet was prepared in the same manner as in Examples 1 and 2, except that the photoradical generator A was changed to the photoradical generator B, C or D. It was confirmed that the cured sheet showed good curability and no air bubbles were observed in the cured sheet.
- the sealants according to Comparative Examples 1 to 4 exhibited good curability when irradiated with light, but air bubbles were observed in the sealants after curing.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Sealing Material Composition (AREA)
- Polymers With Sulfur, Phosphorus Or Metals In The Main Chain (AREA)
Abstract
Description
ここに開示されるシーラントシートは、一態様において、ポリサルファイドポリマー(A)と、一分子中に2以上のチオール基を有するチオール化合物(B)と、一分子中に2以上のアリル基を有するアリル化合物(C)と、光ラジカル発生剤(D)と、を含む。
ここに開示されるシーラントシートは、あらかじめシート形状に成形されており、かかるシート形状の形態でシール対象箇所に配置することができる。この点で、上記シーラントシートは、液状の形態でシール対象箇所に塗布される液状シーラント(例えば、液状ポリサルファイドポリマーと含むA液と該ポリサルファイドポリマーの硬化剤を含むB液とを施工の直前に混ぜ合わせて調製される液状のシーラント)とは明確に区別される。また、ここに開示されるシーラントシートは、チオール基とアリル基の付加反応(チオールエン反応)を利用して硬化させることができる。かかる硬化性を有する点において、ここに開示されるシーラントシートは、硬化後のシーラント(シーラント硬化物)とは明確に区別される。ここに開示されるシーラントシートは、シール対象箇所への配置後にさらに硬化させることが可能な、半硬化状態のシーラントシートとして把握され得る。
図1に示すシーラントシート21は、その一方の表面(第一面)21Aおよび他方の表面(第二面)21Bの各々が、少なくともシーラントシート21側が剥離面となっている剥離ライナー31,32によってそれぞれ保護されている。このような形態のシーラントシート21は、シーラントシート21と剥離ライナー31,32とを含む剥離ライナー付きシーラントシート100の構成要素として把握され得る。
図2に示すシーラントシート21は、その一方の表面21Aが、両面が剥離面となっている剥離ライナー31によって保護された構成を有し、これを巻回すると、シーラントシート21の他方の表面21Bが剥離ライナー31の背面に当接することにより、表面21Bもまた剥離ライナー31で保護された構成とできるようになっている。このような形態のシーラントシート21は、シーラントシート21と剥離ライナー31とを含む剥離ライナー付きシーラントシート200の構成要素として把握され得る。
また、この明細書において、シーラントシートの貯蔵弾性率とは、特記しない場合、硬化前のシーラントシートの貯蔵弾性率を意味し、硬化後のシーラントシート(シーラント硬化物)の貯蔵弾性率とは区別される。本明細書において、シーラントシートの貯蔵弾性率とは、典型的には、該シーラントシートの使用前、すなわち貼付け等によってシール対象箇所に配置される前における貯蔵弾性率を意味する。
ここに開示されるシーラントシートは、ポリサルファイドポリマー(A)を含む。ポリサルファイドポリマー(A)は、-S-S-で表されるジサルファイド構造を含む繰返し単位を有するポリマーであって、該シーラントシートから形成される硬化物の耐油性向上に寄与する。
-R1-O-R2-O-R3-S-S- (1)
ここで、一般式(1)中、R1、R2、R3は、それぞれ独立に、炭素原子数1~4のアルキレン基であり、好ましくは炭素原子数1~3のアルキレン基であり、より好ましくは炭素原子数1~2のアルキレン基である。上記繰返し単位(1)は、エーテル構造とジスルフィド構造とが連なった構成を有する。このような繰返し単位(1)を有するポリサルファイドポリマー(A)によると、耐油性および柔軟性に優れた硬化物が形成される傾向にある。一分子のポリサルファイドポリマー(A)に含まれる上記繰返し単位(1)の数の平均値は、例えば5以上であってよく、10以上でもよく、15以上でもよく、20以上でもよい。また、上記平均値は、例えば100以下であってよく、70以下でもよく、50以下でもよい。上記ポリサルファイドポリマー(A)は、一分子中に、上記繰返し単位(1)が連続する領域を、1つのみ有していてもよく、2つ以上有していてもよい。
-CH2-S-CH2CHOH-R’ (2a)
-CH2-S-CH(CH2OH)-R’ (2b)
ここで、一般式(2a),(2b)中のR’は、少なくとも1個(例えば1個~5個程度)のエポキシ基を有する有機基である。一般式(2a)および(2b)の構造は、例えば、-CH2-SHで表される構造部分を有するチオールと、エポキシ環上に置換基R’を有するエポキシ化合物と、の付加反応により形成され得る。一般式(2a)または(2b)で表される構造の数(一般式(2a)で表される構造と一般式(2b)で表される構造との両方を含む場合は、それらの合計数)は、ポリサルファイドポリマー(A)の一分子当たりの平均値として、例えば1.1以上であってよく、1.3以上でもよく、1.5以上でもよく、1.8以上でもよく、2.0以上でもよく、2.0超でもよい。また、上記平均値は、例えば15以下であってよく、10以下でもよく、7.0以下でもよく、5.0以下でもよい。
HS-(R1-O-R2-O-R3-S-S)n-R1-O-R2-O-R3-SH (3)
ここに開示される技術の好ましい一態様において、シーラントシートは、ポリサルファイドポリマー(A)として一分子中に2以上のエポキシ基を有するエポキシ基含有ポリサルファイドポリマー(AA)と、一分子中に2以上のチオール基を有するチオール化合物(B)と、一分子中に2以上のアリル基を有するアリル化合物(C)と、光ラジカル発生剤(D)と、を含む。かかる態様のシーラントシートは、エポキシ基含有ポリサルファイドポリマー(AA)に該当しないポリサルファイドポリマー(A)をさらに含んでいてもよく、含んでいなくてもよい。
ここに開示されるシーラントシートは、好ましい一態様において、ポリサルファイドポリマー(A)として一分子中に2以上のチオール基を有するチオール基含有ポリサルファイドポリマー(AB)と、一分子中に2以上のアリル基を有するアリル化合物(C)と、光ラジカル発生剤(D)と、を含む。かかる態様のシーラントシートは、チオール基含有ポリサルファイドポリマー(AB)に該当しないポリサルファイドポリマー(A)をさらに含んでいてもよく、含んでいなくてもよい。また、上記態様のシーラントシートは、チオール基含有ポリサルファイドポリマー(AB)に該当しないチオール化合物(B)をさらに含んでいてもよく、含んでいなくてもよい。上記チオール化合物(B)を含む場合、ここに開示されるシーラントシートは、ポリサルファイドポリマー(A)として一分子中に2以上のチオール基を有するチオール基含有ポリサルファイドポリマー(AB)と、一分子中に2以上のチオール基を有するチオール化合物(B)と、一分子中に2以上のアリル基を有するアリル化合物(C)と、光ラジカル発生剤(D)と、を含む。
チオール基含有ポリサルファイドポリマー(AB)に含まれるチオール基は、該チオール基含有ポリサルファイドポリマー(AB)の末端に配置されていることが好ましい。このようなチオール基含有ポリサルファイドポリマー(AB)によると、伸びのよい硬化物が形成される傾向にある。主鎖の両末端にそれぞれ1または2以上のチオール基を有するチオール基含有ポリサルファイドポリマー(AB)が好ましく、主鎖の両末端にそれぞれ1つのチオール基を有するチオール基含有ポリサルファイドポリマー(AB)がより好ましい。かかる構造のチオール基含有ポリサルファイドポリマー(AB)を含むことにより、強度と伸びとをバランスよく両立する硬化物が形成される傾向にある。
ここに開示されるシーラントシートに含まれるチオール化合物(B)としては、一分子中に2以上のチオール基を有する化合物を特に限定なく用いることができる。例えば、トリメチロールプロパントリスチオプロピオネート(別名:トリメチロールプロパントリス(3-メルカプトプロピオネート))、ペンタエリストールテトラキスチオプロピオネート、エチレングリコールビスチオグリコレート、1,4-ブタンジオールビスチオグリコレート、トリメチロールプロパントリスチオグリコレート、ペンタエリストールテトラキスチオグリコレート、ジ(2-メルカプトエチル)エーテル、1,4-ブタンジチオール、1,5-ジメルカプト-3-チアペンタン、1,8-ジメルカプト-3,6-ジオキサオクタン(別名:3,6-ジオキサ-1,8-オクタンジチオール)、1,3,5-トリメルカプトメチルベンゼン、4,4’-チオジベンゼンチオール、1,3,5-トリメルカプトメチル-2,4,6-トリメチルベンゼン、2,4,6-トリメルカプト-s-トリアジン、2-ジブチルアミノ-4,6-ジメルカプト-s-トリアジン、ペンタエリスリトールテトラキス(3-メルカプトプロピオナート)、ジペンタエリスリトールヘキサ-3-メルカプトプロピオネート等が挙げられるが、これらに限定されない。
ここに開示されるシーラントシートに含まれるアリル化合物(C)としては、一分子中に2以上のアリル基を有する化合物を特に限定なく用いることができる。特に限定されないが、ここに開示されるアリル化合物(C)としては、アリルエーテル、アリルエステル等が好ましく用いられ得る。ここに開示されるアリル化合物(C)の例としては、フタル酸ジアリル、イソフタル酸ジアリル、テレフタル酸ジアリル、フマル酸ジアリル、アジピン酸ジアリル、ヘキサヒドロフタル酸ジアリル、1,3-ジアリルオキシ-2-プロパノール等の2官能アリル化合物;イソシアヌル酸トリアリル、シアヌル酸トリアリル、クエン酸トリアリル、トリメリット酸トリアリル等の3官能アリル化合物;ペンタエリスリトールテトラアリルエーテル等の4官能アリル化合物等が挙げられるが、これらに限定されない。
光ラジカル発生剤(D)としては、光照射によりラジカルを発生するものが用いられる。好ましいいくつかの態様において、光ラジカル発生剤(D)は、分子内開裂型光開始剤である。光ラジカル発生剤(D)の例としては、アルキルフェノン系光重合開始剤、アシルホスフィンオキサイド系光重合開始剤、チタノセン化合物系光重合開始剤、アセトフェノン系光重合開始剤、ベンゾインエーテル系光重合開始剤等が挙げられる。光ラジカル発生剤は、一種を単独でまたは二種以上を組み合わせて用いることができる。
アシルホスフィンオキサイド系光重合開始剤の具体例には、ビス(2,4,6-トリメチルベンゾイル)フェニルホスフィンオキシド(例えば、BASF社製の商品名「イルガキュア819」)、ビス(2,4,6-トリメチルベンゾイル)-2,4-ジ-n-ブトキシフェニルホスフィンオキシド、2,4,6-トリメチルベンゾイルジフェニルホスフィンオキシド(例えば、BASF社製の商品名「ルシリンTPO」)、ビス(2,6-ジメトキシベンゾイル)-2,4,4-トリメチルペンチルホスフィンオキシド等が含まれる。
チタノセン化合物系光重合開始剤の具体例には、ビス(2,4-シクロペンタジエニル)ビス[2,6-ジフルオロ-3-(1-ピリル)フェニル]チタン(IV)(例えば、BASF社製の商品名「イルガキュア784」)等が含まれる。
アセトフェノン系光重合開始剤の具体例には、1-ヒドロキシシクロヘキシル-フェニル-ケトン(例えば、BASF社製の商品名「イルガキュア184」)、4-フェノキシジクロロアセトフェノン、4-t-ブチル-ジクロロアセトフェノン、1-[4-(2-ヒドロキシエトキシ)-フェニル]-2-ヒドロキシ-2-メチル-1-プロパン-1-オン(例えば、BASF社製の商品名「イルガキュア2959」)、2-ヒドロキシ-2-メチル-1-フェニル-プロパン-1-オン(例えば、BASF社製の商品名「ダロキュア1173」)、メトキシアセトフェノン等が含まれる。
ベンゾインエーテル系光重合開始剤の具体例には、ベンゾインメチルエーテル、ベンゾインエチルエーテル、ベンゾインプロピルエーテル、ベンゾインイソプロピルエーテル、ベンゾインイソブチルエーテル等のベンゾインエーテルおよびアニソールメチルエーテル等の置換ベンゾインエーテルが含まれる。
ここに開示されるシーラントシートは、増感剤を含有してもよい。増感剤の使用により、照射される光の利用効率を高め、光ラジカル発生剤(D)の感度を向上させることができる。光増感剤は、公知の材料から適宜選択して使用することができる。光増感剤の非限定的な例には、ベンゾフェノン、4-メチルベンゾフェノン、3-ベンゾイルビフェニル、4-(4-メチルフェニルチオ)ベンゾフェノン、メチル2-ベンゾイルベンゾエート、4-フェニルベンゾフェノン、4,4’-ビス(ジメトキシ)ベンゾフェノン、4,4’-ビス(ジメチルアミノ)ベンゾフェノン、4,4’-ビス(ジエチルアミノ)ベンゾフェノン、2-ベンゾイル安息香酸メチルエステル、2-メチルベンゾフェノン、3-メチルベンゾフェノン、3,3’-ジメチル-4-メトキシベンゾフェノン、2,4,6-トリメチルベンゾフェノン等のベンゾフェノン誘導体;チオキサントン、キサントン、2-クロロチオキサントン、4-クロロチオキサントン、2-イソプロピルチオキサントン、4-イソプロピルチオキサントン、2,4-ジメチルチオキサントン、2,4-ジエチルチオキサントン、1-クロロ-4-プロポキシチオキサントン、2,4-ジエチルチオキサンテン-9-オン等のチオキサントン誘導体;2-ヒドロキシ-9-フルオレノン等のフルオレン系化合物;アントロン、ジベンゾスベロン、2-アミノ-9-フルオレノン等のアントロン誘導体;アントラキノン、2-エチルアントラキノン、2-ヒドロキシアントラキノン、2-アミノアントラキノン等のアントラキノン誘導体;1-メチルナフタレン、2-メチルナフタレン、1-フルオロナフタレン、1-クロロナフタレン、2-クロロナフタレン、1-ブロモナフタレン、2-ブロモナフタレン、1-ヨードナフタレン、2-ヨードナフタレン、1-ナフトール、2-ナフトール、1-メトキシナフタレン、2-メトキシナフタレン、1,4-ジシアノナフタレン、メチル3-ヒドロキシ-2-ナフトエート等のナフタレン誘導体;アントラセン、1,2-ベンズアントラセン、9,10-ジクロロアントラセン、9,10-ジブロモアントラセン、9,10-ジフェニルアントラセン、9-シアノアントラセン、9,10-ジシアノアントラセン、2,6,9,10-テトラシアノアントラセン等のアントラセン誘導体;ニトロ安息香酸やニトロアニリン等のニトロ化合物;各種の色素;等が含まれるが、これらに限定されない。
ここに開示されるシーラントシートは、他の特性が大きく損なわれない限度で、チオール基とアリル基とのラジカル付加反応の抑制に役立ち得る任意の化合物をさらに含有してもよい。かかる化合物の使用により、使用前のシーラントシートの保存性を高めることができる。貯蔵安定剤は、例えば、室温で液状または固体の有機酸、無機酸、および分子中に酸性基を含むオリゴマー、ポリマー、ホウ酸エステル類、リン酸エステル類であってよく、酸性基以外の官能基を有していても良い。例えば、硫酸、酢酸、アジピン酸、酒石酸、フマル酸、バルビツール酸、ホウ酸、ピロガロール、フェノール樹脂、カルボン酸無水物等が挙げられるが、これらに限定されない。貯蔵安定剤は、一種を単独でまたは二種以上を適宜組み合わせて用いることができる。貯蔵安定剤の使用量は特に限定されず、所望の効果が得られるように設定することができる。
ホウ酸エステル類は、室温で液状または固体のホウ酸エステルである。例えばトリメチルボレート、トリエチルボレート、トリ-n-プロピルボレート、トリイソプロピルボレート、トリ-n-ブチルボレート、トリペンチルボレート、トリアリルボレート、トリヘキシルボレート、トリシクロヘキシルボレート、トリオクチルボレート、トリノニルボレート、トリデシルボレート、トリドデシルボレート、トリヘキサデシルボレート、トリオクタデシルボレート、トリス(2-エチルヘキシロキシ)ボラン、ビス(1,4,7,10-テトラオキサウンデシル)(1,4,7,10,13-ペンタオキサテトラデシル)(1,4,7-トリオキサウンデシル)ボラン、トリベンジルボレート、トリフェニルボレート、トリ-o-トリルボレート、トリ-m-トリルボレート、トリエタノールアミンボレート等が挙げられるが、これらに限定されない。
リン酸エステル類としては、リン酸エチル、リン酸ブチル、リン酸プロピル、リン酸-2-エチルヘキシル、リン酸ジブチル、リン酸-ジ(2-エチルヘキシル)、リン酸オレイル、リン酸エチルジエチル等が挙げられるが、これらに限定されない。ここに開示されるシーラントシートは、好ましい一態様において、貯蔵安定剤を含まない。
ここに開示されるシーラントシートには、必要に応じてフィラーを配合することができる。これにより、硬化物の破断強度および破断時伸びの一方または両方が改善され得る。フィラーは、シーラントシートの貯蔵弾性率の調節にも役立ち得る。また、フィラーの適切な使用により、シーラントシートの保形性や加工性を高めることができる。使用するフィラーは特に制限されず、ここに開示される技術により得られる効果を大きく損なわない範囲で、任意の適切なフィラーを使用し得る。フィラーは、一種を単独でまたは二種以上を組み合わせて用いることができる。
ここに開示されるシーラントシートの作製(例えば、シート形状への成形)、使用前のシーラントシートの保存、流通、形状加工、シール対象箇所への配置等の際に、剥離ライナーを用いることができる。剥離ライナーとしては、特に限定されず、例えば、樹脂フィルムや紙等のライナー基材の表面に剥離処理層を有する剥離ライナーや、フッ素系ポリマー(ポリテトラフルオロエチレン等)やポリオレフィン系樹脂(ポリエチレン、ポリプロピレン等)の低接着性材料からなる剥離ライナー等を用いることができる。上記剥離処理層は、例えば、シリコーン系、長鎖アルキル系、フッ素系、硫化モリブデン等の剥離処理剤により上記ライナー基材を表面処理して形成されたものであり得る。
以下、ここに開示されるシーラントシートの製造方法のいくつかの態様について説明する。ただし、以下の説明は、例示を目的とするものであって、ここに開示されるシーラントシートの製造方法を限定するものではない。例えば、説明の便宜上、以下では主にフィラーを用いる態様について述べるが、ここに開示されるシーラントシートがフィラーを含む態様に限定されることを意味するものではない。
ここに開示されるシーラントシートを用いてシールされる箇所の材質は、特に限定されない。上記材質は、例えば金属、樹脂、これらの複合材料等であってよく、より具体的には、鉄、鉄合金(炭素鋼、ステンレス鋼、クロム鋼、ニッケル鋼等)、アルミニウム、アルミニウム合金、ニッケル、タングステン、銅、銅合金、チタン、チタン合金、シリコン等の金属または半金属材料;ポリオレフィン樹脂、ポリカーボネート樹脂、アクリル樹脂、アクリロニトリル樹脂(PAN)等の樹脂材料;アルミナ、シリカ、サファイア、窒化ケイ素、窒化タンタル、炭化チタン、炭化ケイ素、窒化ガリウム、石膏等のセラミック材料;アルミノシリケートガラス、ソーダライムガラス、ソーダアルミノケイ酸ガラス、石英ガラス等のガラス材料;これらの積層物や複合物;等であり得る。上記金属または半金属材料の好適例として、アルミニウムやチタン等の軽金属または該軽金属を主成分とする合金が挙げられる。アルミニウム合金の例として、ジュラルミン(例えば、ジュラルミンA2024、ジュラルミンA2017等)が挙げられる。また、上記複合物の例としては、炭素繊維強化プラスチック(CFRP)、ガラス繊維強化プラスチック(FRP)等が挙げられる。
また、ここに開示されるシーラントシートから形成される硬化物または上記シーラント硬化物は、破断時伸びが、100%以上であることが適当であり、120%以上が好ましく、150%以上でもよく、200%以上でもよく、250%以上でもよい。破断時伸びの上限は特に制限されないが、他の物性との両立を容易とする観点から、例えば600%以下であってよく、400%以下でもよい。
(引張破断強度および破断時伸びの測定)
厚さ0.2mmのシーラントシートの片面に、東芝ライテック社製のブラックライトを用いて2000mJ/cm2の光照射を行う。照射後のシーラントシートを25℃の環境下に14日間保持した後、得られた硬化物(シーラント硬化物)を幅10mm、長さ50mmの長方形状にカットしてサンプル片を作製する。このサンプル片を、チャック間20mmとなるようにして引張試験機のチャックに挟み、JIS K6767に準じて50mm/分の速度で引っ張り、サンプル片が破断するまでに観測された最大強度を引張破断強度とする。また、サンプルが破断したときのチャック間距離L1および引張り開始時のチャック間距離L0から、以下の式:
破断時伸び(%)=((L1-L0)/L0)×100;
により破断時伸びを算出する。
(1) シート形状に成形されたシーラントシートであって、
以下の成分:
ポリサルファイドポリマー(A);
一分子中に2以上のチオール基を有するチオール化合物(B);
一分子中に2以上のアリル基を有するアリル化合物(C);および
光ラジカル発生剤(D);
を含む、シーラントシート。
(2) 上記シーラントシートは、上記ポリサルファイドポリマー(A)として2以上のエポキシ基を有するエポキシ基含有ポリサルファイドポリマー(AA)を含む、上記(1)に記載のシーラントシート。
(3) 上記エポキシ基含有ポリサルファイドポリマー(AA)は、両末端エポキシポリサルファイドポリマーである、上記(2)に記載のシーラントシート。
(4) 上記エポキシ基含有ポリサルファイドポリマー(AA)は、ジサルファイド構造を主鎖中に有するMw500~10000の両末端チオールポリサルファイドと、一分子中に2以上のエポキシ基を有するエポキシ化合物との反応生成物である、上記(2)または(3)に記載のシーラントシート。
(5) 上記エポキシ化合物は、2官能エポキシ化合物を含む、上記(4)に記載のシーラントシート。
(6) 上記2官能エポキシ化合物として、分子内に5員環以上の炭素環構造を含むエポキシ化合物を含む、上記(5)に記載のシーラントシート。
(7) 上記エポキシ化合物は、3官能以上の多官能エポキシ化合物を含む、上記(4)~(6)のいずれかに記載のシーラントシート。
(8) 上記多官能エポキシ化合物として、ノボラック型エポキシ樹脂を含む、上記(7)に記載のシーラントシート。
(9) 上記シーラントシートは、上記ポリサルファイドポリマー(A)として2以上のチオール基を有するチオール基含有ポリサルファイドポリマー(AB)を含む、上記(1)~(8)のいずれかに記載のシーラントシート。
(10) 上記チオール基含有ポリサルファイドポリマー(AB)は、両末端チオールポリサルファイドポリマーである、上記(9)に記載のシーラントシート。
(11) 上記チオール基含有ポリサルファイドポリマー(AB)は、ジサルファイド構造を主鎖中に有するMw500~10000の両末端エポキシポリサルファイドと、一分子中に2以上のチオール基を有するチオール化合物との反応生成物である、上記(9)または(10)に記載のシーラントシート。
(12) 上記チオール化合物(B)は、チオール当量が45g/eq以上450g/eq以下である、上記(1)~(11)のいずれかに記載のシーラントシート。
(13) 上記アリル化合物(C)は、2官能アリル化合物および3官能アリル化合物からなる群から選ばれる少なくとも一種である、上記(1)~(12)のいずれかに記載のシーラントシート。
(15) さらにフィラーを含む、上記(1)~(14)のいずれかに記載のシーラントシート。
(16) 上記フィラーの含有量は、シーラントシート全体の1重量%以上40重量%未満である、上記(15)に記載のシーラントシート。
(17) 上記フィラーの平均粒子径は0.1μm以上30μm以下である、上記(15)または(16)に記載のシーラントシート。
(18) 25℃における貯蔵弾性率が0.005MPa以上0.8MPa以下である、上記(1)~(17)のいずれかに記載のシーラントシート。
(19) 厚さが0.01mm以上10mm以下である、上記(1)~(18)のいずれかに記載のシーラントシート。
(20) シート形状に成形されたシーラントシートであって、
以下の成分:
2以上のチオール基を有するチオール基含有ポリサルファイドポリマー(AB);
一分子中に2以上のアリル基を有するアリル化合物(C);および
光ラジカル発生剤(D);
を含む、シーラントシート。
上記シーラントシートの少なくとも一方の表面に当接する剥離面を有する剥離ライナーと、
を含む、剥離ライナー付きシーラントシート。
(22) ポリサルファイドポリマー(A)を用意すること;
上記ポリサルファイドポリマー(A)に、一分子中に2以上のチオール基を有するチオール化合物(B)、一分子中に2以上のアリル基を有するアリル化合物(C)、光ラジカル発生剤(D)およびフィラーを添加して混合すること;および、
得られた混合物をシート形状に成形すること;
を含む、シーラントシート製造方法。
(23) 一分子中に2以上のチオール基を有するチオール基含有ポリサルファイドポリマー(AB)を用意すること;
上記チオール基含有ポリサルファイドポリマー(AB)に、一分子中に2以上のアリル基を有するアリル化合物(C)、光ラジカル発生剤(D)およびフィラーを添加して混合すること;および、
得られた混合物をシート形状に成形すること;
を含む、シーラントシート製造方法。
(24) 上記(1)~(20)のいずれかに記載のシーラントシートを用意すること、
上記シーラントシートをシール対象物に貼り付けること、
上記シール対象物上で上記シーラントシートを硬化させてシーラント硬化物を形成すること、
を含み、
ここで、上記シーラントシートをシール対象物に貼り付ける直前および/または上記シーラントシートをシール対象物に貼り付けた後に該シーラントシートに光照射を行う、シーリング施工方法。
<両末端エポキシポリサルファイドポリマーの調製>
攪拌機を備えた反応容器を用いて、液状ポリサルファイドポリマー(両末端チオールポリサルファイドポリマー)90部、2官能エポキシ化合物7.5部、多官能エポキシ化合物2部、および塩基性触媒0.08部を、攪拌しながら90℃で3時間加熱した。これにより、両末端エポキシポリサルファイドポリマーを合成した。
ここで、液状ポリサルファイドポリマー(両末端チオールポリサルファイドポリマー)としては東レファインケミカル社製の製品名チオコールLP-55(Mw4000、チオール当量2000g/eq)、2官能エポキシ化合物としては三菱ケミカル社製のビスフェノールF型エポキシ樹脂(製品名jER806、エポキシ当量169g/eq)、多官能エポキシ化合物としては三菱ケミカル社製のフェノールボラック型エポキシ樹脂(製品名jER152、エポキシ当量177g/eq)、塩基性触媒としては東京化成社製の2,4,6-トリアミノメチルフェノールを用いた。
反応容器の内容物を取り出して室温まで放冷させた後、両末端エポキシポリサルファイドポリマー50部に対して、表1に示す2級2官能チオール化合物0.5部、3官能アリル化合物0.6部、光ラジカル発生剤A1.0部、ならびにフィラーとしてタルク20部を加え、2本ロールミルを用いて均一に練り合わせた。得られた混合物を、真空プレス機を用いてシート状に成形することにより、本例に係るシーラントシートを得た。その際、厚さ0.2mmのシートと厚さ1mmのシートとの2種類を作製した。
<両末端チオールポリサルファイドポリマーの調製>
攪拌機を備えた反応容器を用いて、液状ポリサルファイドポリマー(両末端チオールポリサルファイドポリマー)90部、2官能エポキシ化合物10部、多官能エポキシ化合物2部、および塩基性触媒0.08部を、攪拌しながら90℃で3時間加熱した。次いで、2官能チオール化合物4部を添加し、攪拌しながら90℃で1.5時間加熱した。これにより、両末端チオールポリサルファイドポリマーを合成した。
ここで、液状ポリサルファイドポリマー(両末端チオールポリサルファイドポリマー)としては東レファインケミカル社製の製品名チオコールLP-55(Mw4000、チオール当量2000g/eq)、2官能エポキシ化合物としては三菱ケミカル社製のビスフェノールF型エポキシ樹脂(製品名jER806、エポキシ当量169g/eq)、多官能エポキシ化合物としては三菱ケミカル社製のフェノールボラック型エポキシ樹脂(製品名jER152、エポキシ当量177g/eq)、塩基性触媒としては東京化成社製の2,4,6-トリアミノメチルフェノール、2官能チオール化合物としては東京化成社製の3,6-ジオキサ-1,8-オクタンジチオール(チオール当量91g/eq)を用いた。
反応容器の内容物を取り出して室温まで放冷させた後、両末端エポキシポリサルファイドポリマー50部に対して、表1に示す2級2官能チオール0.5部、3官能アリル化合物0.6部、光ラジカル発生剤A1.0部およびフィラーとしてのタルク20部を加え、2本ロールミルを用いて均一に練り合わせた。得られた混合物を、真空プレス機を用いてシート状に成形することにより、本例に係るシーラントシートを得た。その際、厚さ0.2mmのシートと厚さ1mmのシートとの2種類を作製した。
攪拌機を備えた反応容器を用いて、表1に示す液状ポリサルファイドポリマー(両末端チオールポリサルファイドポリマー)46部、2級2官能チオール8.82部、1級2官能チオール5.46部、3官能アリル化合物9.96部、光ラジカル発生剤A0.4部およびフィラーとしてのタルク20部を加えて混合した。得られた混合物は常温において液体状態であった。上記混合物を本例に係るシーラントとした。
使用する光ラジカル発生剤の種類を表1に示すとおりとした他は比較例1と同様にして、各例に係るシーラントを作製した。比較例2~4に係るシーラントはいずれも常温において液体状態であった。
(貯蔵弾性率の測定)
厚さ1mmのシーラントシートを直径8mmの円盤状に打ち抜き、パラレルプレートで挟み込み、粘弾性試験機(ティー・エイ・インスツルメント・ジャパン社製、機種名「ARES G2」)を用いて、測定温度25℃、周波数1Hz、歪み0.5%の条件において貯蔵弾性率G’を測定した。その結果、実施例1および2に係るシーラントシートの貯蔵弾性率G’は、いずれも0.005MPa~0.8MPaの範囲にあった。
厚さ0.2mmのシーラントシートの両面に、厚さ38μmの透明PETフィルム(三菱ポリエステル製、ダイアホイルMRF38)を貼り付けたのち、その片面側からシーラントシートに向かって東芝ライテック社製のブラックライトを用いて2000mJ/cm2の光照射を行った。その後、透明PETフィルムの上からシーラントシートの外観を試験者が目視で観察し、シーラントシート内部に気泡が存在しているか否か確認した。その結果、気泡が目視で観察された場合は「あり」、気泡が目視で観察されなかった場合は「なし」と評価した。また、上記光照射後に、透明PETフィルムを介してシーラントシートの表面を試験者が手で触れることにより、シーラントシートが硬化しているかどうかを確認した。その結果、シーラントシートが硬化していた場合は硬化性「良」、硬化していなかった場合は硬化性「不良」と評価した。結果は、表1の該当箇所に示す。
21A 一方の表面
21B 他方の表面
31,32 剥離ライナー
100,200 剥離ライナー付きシーラントシート
Claims (8)
- シート形状に成形されたシーラントシートであって、
以下の成分:
ポリサルファイドポリマー(A);
一分子中に2以上のチオール基を有するチオール化合物(B);
一分子中に2以上のアリル基を有するアリル化合物(C);および
光ラジカル発生剤(D);
を含む、シーラントシート。 - 前記チオール化合物(B)は、チオール当量が45g/eq以上450g/eq以下である、請求項1に記載のシーラントシート。
- 前記アリル化合物(C)として2官能アリル化合物および3官能アリル化合物からなる群から選ばれる少なくとも一種を含む、請求項1または2に記載のシーラントシート。
- 前記光ラジカル発生剤(D)は、アルキルフェノン系光重合開始剤、アシルホスフィンオキサイド系光重合開始剤およびチタノセン化合物系光重合開始剤からなる群から選ばれる少なくとも1つである、請求項1から3のいずれか一項に記載のシーラントシート。
- さらにフィラーを含む、請求項1から4のいずれか一項に記載のシーラントシート。
- 25℃における貯蔵弾性率が0.005MPa以上0.8MPa以下である、請求項1から5のいずれか一項に記載のシーラントシート。
- シート形状に成形されたシーラントシートであって、
以下の成分:
一分子中に2以上のチオール基を有するチオール基含有ポリサルファイドポリマー(AB);
一分子中に2以上のアリル基を有するアリル化合物(C);および
光ラジカル発生剤(D);
を含む、シーラントシート。 - 請求項1から7のいずれか一項に記載のシーラントシートと、
前記シーラントシートの少なくとも一方の表面に当接する剥離面を有する剥離ライナーと、
を含む、剥離ライナー付きシーラントシート。
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023510988A JPWO2022210045A1 (ja) | 2021-03-30 | 2022-03-18 | |
| EP22780260.0A EP4317352A1 (en) | 2021-03-30 | 2022-03-18 | Sealant sheet |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-056892 | 2021-03-30 | ||
| JP2021056892 | 2021-03-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022210045A1 true WO2022210045A1 (ja) | 2022-10-06 |
Family
ID=83456124
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/012754 WO2022210045A1 (ja) | 2021-03-30 | 2022-03-18 | シーラントシート |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP4317352A1 (ja) |
| JP (1) | JPWO2022210045A1 (ja) |
| WO (1) | WO2022210045A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024005154A1 (ja) * | 2022-06-30 | 2024-01-04 | 株式会社レゾナック | 組成物、光融解性組成物及び化合物 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1017852A (ja) * | 1996-07-04 | 1998-01-20 | Yokohama Rubber Co Ltd:The | 2液型ポリサルファイド系シーリング材組成物 |
| WO2019107486A1 (ja) * | 2017-11-29 | 2019-06-06 | 日東電工株式会社 | シーラントシート |
| WO2019146421A1 (ja) * | 2018-01-23 | 2019-08-01 | ボスティック・ニッタ株式会社 | 光硬化性シーリング用材料 |
| WO2020004487A1 (ja) * | 2018-06-26 | 2020-01-02 | 日東電工株式会社 | シーラントシート |
| JP2021056892A (ja) | 2019-09-30 | 2021-04-08 | シスメックス株式会社 | 専門家会議をコンピュータを用いて支援する方法、支援装置、専門家会議を支援するためのコンピュータプログラム、支援システム。 |
-
2022
- 2022-03-18 WO PCT/JP2022/012754 patent/WO2022210045A1/ja active Application Filing
- 2022-03-18 EP EP22780260.0A patent/EP4317352A1/en active Pending
- 2022-03-18 JP JP2023510988A patent/JPWO2022210045A1/ja active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1017852A (ja) * | 1996-07-04 | 1998-01-20 | Yokohama Rubber Co Ltd:The | 2液型ポリサルファイド系シーリング材組成物 |
| WO2019107486A1 (ja) * | 2017-11-29 | 2019-06-06 | 日東電工株式会社 | シーラントシート |
| WO2019146421A1 (ja) * | 2018-01-23 | 2019-08-01 | ボスティック・ニッタ株式会社 | 光硬化性シーリング用材料 |
| WO2020004487A1 (ja) * | 2018-06-26 | 2020-01-02 | 日東電工株式会社 | シーラントシート |
| JP2021056892A (ja) | 2019-09-30 | 2021-04-08 | シスメックス株式会社 | 専門家会議をコンピュータを用いて支援する方法、支援装置、専門家会議を支援するためのコンピュータプログラム、支援システム。 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024005154A1 (ja) * | 2022-06-30 | 2024-01-04 | 株式会社レゾナック | 組成物、光融解性組成物及び化合物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4317352A1 (en) | 2024-02-07 |
| JPWO2022210045A1 (ja) | 2022-10-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7262459B2 (ja) | シーラントシート | |
| KR101111372B1 (ko) | 자외선 경화형 조성물 | |
| JP5057016B2 (ja) | 活性エネルギー線の照射により活性化するアミンイミド化合物、それを用いた組成物およびその硬化方法 | |
| JP7273724B2 (ja) | シーラントシート | |
| JP4973868B2 (ja) | 硬化性樹脂組成物および硬化方法 | |
| JP2008001867A (ja) | 硬化性樹脂組成物 | |
| WO2022210045A1 (ja) | シーラントシート | |
| TW201923017A (zh) | 顯示器用封裝劑 | |
| JP2009007404A (ja) | カチオン重合性組成物および該組成物を硬化して得られる硬化物 | |
| JP2005336349A (ja) | カチオン重合型組成物 | |
| JP4639684B2 (ja) | 液晶表示装置のシール剤 | |
| JP7458778B2 (ja) | 剥離ライナー付きシーラントシート | |
| JP7485511B2 (ja) | シーリング方法 | |
| JP6880809B2 (ja) | 活性エネルギー線硬化型封止剤用組成物 | |
| WO2021132486A1 (ja) | 積層体 | |
| JP2023039242A (ja) | 液晶滴下工法用液晶シール剤及びそれを用いた液晶表示パネル | |
| JP2023083652A (ja) | 液晶滴下工法用液晶シール剤及びそれを用いた液晶表示パネル | |
| JP2020187302A (ja) | 液晶表示素子の製造方法 | |
| JP2005336333A (ja) | 活性エネルギー線硬化型組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22780260 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023510988 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18284957 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022780260 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022780260 Country of ref document: EP Effective date: 20231030 |