WO2022090757A1 - 電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス - Google Patents
電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス Download PDFInfo
- Publication number
- WO2022090757A1 WO2022090757A1 PCT/IB2020/000884 IB2020000884W WO2022090757A1 WO 2022090757 A1 WO2022090757 A1 WO 2022090757A1 IB 2020000884 W IB2020000884 W IB 2020000884W WO 2022090757 A1 WO2022090757 A1 WO 2022090757A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- positive electrode
- solid electrolyte
- active material
- electrode active
- sulfur
- Prior art date
Links
- 239000007774 positive electrode material Substances 0.000 title claims abstract description 148
- 239000007784 solid electrolyte Substances 0.000 claims abstract description 137
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims abstract description 99
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 97
- 239000011593 sulfur Substances 0.000 claims abstract description 95
- 239000011148 porous material Substances 0.000 claims abstract description 68
- 239000004020 conductor Substances 0.000 claims abstract description 57
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 claims description 39
- 229910001416 lithium ion Inorganic materials 0.000 claims description 39
- 239000002203 sulfidic glass Substances 0.000 claims description 39
- 239000000463 material Substances 0.000 claims description 33
- 229910052744 lithium Inorganic materials 0.000 claims description 27
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 claims description 23
- 239000003575 carbonaceous material Substances 0.000 claims description 16
- 238000000034 method Methods 0.000 claims description 15
- 229910052698 phosphorus Inorganic materials 0.000 claims description 14
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical group [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims description 7
- 229910052796 boron Inorganic materials 0.000 claims description 7
- 150000001340 alkali metals Chemical group 0.000 claims description 6
- 125000004437 phosphorous atom Chemical group 0.000 claims description 5
- 229910052783 alkali metal Inorganic materials 0.000 claims description 4
- 239000010410 layer Substances 0.000 description 85
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 76
- 239000007773 negative electrode material Substances 0.000 description 52
- 229910052799 carbon Inorganic materials 0.000 description 48
- 239000002245 particle Substances 0.000 description 28
- -1 polyethylene Polymers 0.000 description 21
- 229910052782 aluminium Inorganic materials 0.000 description 18
- 239000011149 active material Substances 0.000 description 17
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 17
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 16
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 15
- 229910052751 metal Inorganic materials 0.000 description 15
- 239000002184 metal Substances 0.000 description 15
- 239000012752 auxiliary agent Substances 0.000 description 14
- 229920001973 fluoroelastomer Polymers 0.000 description 14
- 239000010408 film Substances 0.000 description 13
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 12
- 229920002239 polyacrylonitrile Polymers 0.000 description 12
- 230000000052 comparative effect Effects 0.000 description 11
- 239000000843 powder Substances 0.000 description 11
- 239000000956 alloy Substances 0.000 description 10
- 238000010248 power generation Methods 0.000 description 10
- 229910018130 Li 2 S-P 2 S 5 Inorganic materials 0.000 description 9
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 9
- 229910045601 alloy Inorganic materials 0.000 description 9
- 150000001875 compounds Chemical class 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 8
- 229910052802 copper Inorganic materials 0.000 description 8
- 239000010949 copper Substances 0.000 description 8
- 239000008151 electrolyte solution Substances 0.000 description 8
- 239000011574 phosphorus Substances 0.000 description 8
- 229920005989 resin Polymers 0.000 description 8
- 239000011347 resin Substances 0.000 description 8
- 239000010936 titanium Substances 0.000 description 8
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 7
- 239000002131 composite material Substances 0.000 description 7
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 7
- 239000012071 phase Substances 0.000 description 7
- 238000002360 preparation method Methods 0.000 description 7
- 229910052718 tin Inorganic materials 0.000 description 7
- 239000004743 Polypropylene Substances 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 239000012300 argon atmosphere Substances 0.000 description 6
- 229920001940 conductive polymer Polymers 0.000 description 6
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- 229920001155 polypropylene Polymers 0.000 description 6
- 229910001220 stainless steel Inorganic materials 0.000 description 6
- 239000010935 stainless steel Substances 0.000 description 6
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 5
- 229910018091 Li 2 S Inorganic materials 0.000 description 5
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 5
- 239000011230 binding agent Substances 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- 239000011231 conductive filler Substances 0.000 description 5
- 229920001577 copolymer Polymers 0.000 description 5
- 238000010438 heat treatment Methods 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- 239000011244 liquid electrolyte Substances 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 229910052759 nickel Inorganic materials 0.000 description 5
- 239000002861 polymer material Substances 0.000 description 5
- 229910052710 silicon Inorganic materials 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- 229910052719 titanium Inorganic materials 0.000 description 5
- 229920000049 Carbon (fiber) Polymers 0.000 description 4
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 4
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- 239000002033 PVDF binder Substances 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 4
- 229910052794 bromium Inorganic materials 0.000 description 4
- 125000004432 carbon atom Chemical group C* 0.000 description 4
- 239000004917 carbon fiber Substances 0.000 description 4
- 239000002800 charge carrier Substances 0.000 description 4
- 229910052801 chlorine Inorganic materials 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 229910052740 iodine Inorganic materials 0.000 description 4
- 229910044991 metal oxide Inorganic materials 0.000 description 4
- 150000004706 metal oxides Chemical class 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 239000011812 mixed powder Substances 0.000 description 4
- 239000003960 organic solvent Substances 0.000 description 4
- 239000004810 polytetrafluoroethylene Substances 0.000 description 4
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 4
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 3
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 3
- 229910018133 Li 2 S-SiS 2 Inorganic materials 0.000 description 3
- 229910010859 LiI—LiBr—Li3PS4 Inorganic materials 0.000 description 3
- 239000004698 Polyethylene Substances 0.000 description 3
- 239000004642 Polyimide Substances 0.000 description 3
- 239000004793 Polystyrene Substances 0.000 description 3
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 3
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 3
- 239000006230 acetylene black Substances 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 230000005540 biological transmission Effects 0.000 description 3
- 239000002134 carbon nanofiber Substances 0.000 description 3
- 229910021393 carbon nanotube Inorganic materials 0.000 description 3
- 239000002041 carbon nanotube Substances 0.000 description 3
- 238000010277 constant-current charging Methods 0.000 description 3
- 239000000470 constituent Substances 0.000 description 3
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 3
- 238000007599 discharging Methods 0.000 description 3
- 239000006185 dispersion Substances 0.000 description 3
- 239000003792 electrolyte Substances 0.000 description 3
- 125000002573 ethenylidene group Chemical group [*]=C=C([H])[H] 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 229910052733 gallium Inorganic materials 0.000 description 3
- 229910052732 germanium Inorganic materials 0.000 description 3
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 3
- 229910052737 gold Inorganic materials 0.000 description 3
- 239000010931 gold Substances 0.000 description 3
- 229910052736 halogen Inorganic materials 0.000 description 3
- 150000002367 halogens Chemical class 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 229910052738 indium Inorganic materials 0.000 description 3
- 239000003273 ketjen black Substances 0.000 description 3
- 229910003002 lithium salt Inorganic materials 0.000 description 3
- 159000000002 lithium salts Chemical class 0.000 description 3
- VDVLPSWVDYJFRW-UHFFFAOYSA-N lithium;bis(fluorosulfonyl)azanide Chemical compound [Li+].FS(=O)(=O)[N-]S(F)(=O)=O VDVLPSWVDYJFRW-UHFFFAOYSA-N 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 239000005020 polyethylene terephthalate Substances 0.000 description 3
- 229920000139 polyethylene terephthalate Polymers 0.000 description 3
- 229920001721 polyimide Polymers 0.000 description 3
- 238000003825 pressing Methods 0.000 description 3
- 239000010703 silicon Substances 0.000 description 3
- 239000011871 silicon-based negative electrode active material Substances 0.000 description 3
- 229910052709 silver Inorganic materials 0.000 description 3
- 239000004332 silver Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 238000002336 sorption--desorption measurement Methods 0.000 description 3
- 229920003048 styrene butadiene rubber Polymers 0.000 description 3
- 239000010409 thin film Substances 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- ZZXUZKXVROWEIF-UHFFFAOYSA-N 1,2-butylene carbonate Chemical compound CCC1COC(=O)O1 ZZXUZKXVROWEIF-UHFFFAOYSA-N 0.000 description 2
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 2
- GCGGSVAWTYHZBI-CVQRFVFPSA-N 1-hydroxy-2-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-[[(2s,3r,4s,5r)-3,4,5-trihydroxyoxan-2-yl]oxymethyl]oxan-2-yl]oxyanthracene-9,10-dione Chemical compound O[C@@H]1[C@@H](O)[C@H](O)CO[C@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@H](OC=2C(=C3C(=O)C4=CC=CC=C4C(=O)C3=CC=2)O)O1 GCGGSVAWTYHZBI-CVQRFVFPSA-N 0.000 description 2
- JWUJQDFVADABEY-UHFFFAOYSA-N 2-methyltetrahydrofuran Chemical compound CC1CCCO1 JWUJQDFVADABEY-UHFFFAOYSA-N 0.000 description 2
- LBKMJZAKWQTTHC-UHFFFAOYSA-N 4-methyldioxolane Chemical compound CC1COOC1 LBKMJZAKWQTTHC-UHFFFAOYSA-N 0.000 description 2
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 2
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 2
- 239000005977 Ethylene Substances 0.000 description 2
- 229910009324 Li2S-SiS2-Li3PO4 Inorganic materials 0.000 description 2
- 229910009328 Li2S-SiS2—Li3PO4 Inorganic materials 0.000 description 2
- 229910007295 Li2S—SiS2—Li3PO4 Inorganic materials 0.000 description 2
- 229910007306 Li2S—SiS2—P2S5LiI Inorganic materials 0.000 description 2
- 229910010941 LiFSI Inorganic materials 0.000 description 2
- 229910010847 LiI—Li3PO4-P2S5 Inorganic materials 0.000 description 2
- 229910010864 LiI—Li3PO4—P2S5 Inorganic materials 0.000 description 2
- 229910010918 LiLaZrO Inorganic materials 0.000 description 2
- RJUFJBKOKNCXHH-UHFFFAOYSA-N Methyl propionate Chemical compound CCC(=O)OC RJUFJBKOKNCXHH-UHFFFAOYSA-N 0.000 description 2
- 229920002319 Poly(methyl acrylate) Polymers 0.000 description 2
- 239000004952 Polyamide Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 2
- BTFOWJRRWDOUKQ-UHFFFAOYSA-N [Si]=O.[Sn] Chemical class [Si]=O.[Sn] BTFOWJRRWDOUKQ-UHFFFAOYSA-N 0.000 description 2
- 230000001133 acceleration Effects 0.000 description 2
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 2
- 230000000996 additive effect Effects 0.000 description 2
- 229910021383 artificial graphite Inorganic materials 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 2
- 239000006229 carbon black Substances 0.000 description 2
- 229910002092 carbon dioxide Inorganic materials 0.000 description 2
- 239000001569 carbon dioxide Substances 0.000 description 2
- 239000006231 channel black Substances 0.000 description 2
- 238000007600 charging Methods 0.000 description 2
- 239000000571 coke Substances 0.000 description 2
- 230000003750 conditioning effect Effects 0.000 description 2
- 239000006258 conductive agent Substances 0.000 description 2
- VUPKGFBOKBGHFZ-UHFFFAOYSA-N dipropyl carbonate Chemical compound CCCOC(=O)OCCC VUPKGFBOKBGHFZ-UHFFFAOYSA-N 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 239000007772 electrode material Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011888 foil Substances 0.000 description 2
- 229920001903 high density polyethylene Polymers 0.000 description 2
- 239000004700 high-density polyethylene Substances 0.000 description 2
- 238000005470 impregnation Methods 0.000 description 2
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 2
- LHJOPRPDWDXEIY-UHFFFAOYSA-N indium lithium Chemical compound [Li].[In] LHJOPRPDWDXEIY-UHFFFAOYSA-N 0.000 description 2
- 239000010416 ion conductor Substances 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- 239000006233 lamp black Substances 0.000 description 2
- 229920001684 low density polyethylene Polymers 0.000 description 2
- 239000004702 low-density polyethylene Substances 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 238000013507 mapping Methods 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 239000002931 mesocarbon microbead Substances 0.000 description 2
- 239000007769 metal material Substances 0.000 description 2
- TZIHFWKZFHZASV-UHFFFAOYSA-N methyl formate Chemical compound COC=O TZIHFWKZFHZASV-UHFFFAOYSA-N 0.000 description 2
- 229940017219 methyl propionate Drugs 0.000 description 2
- 229910021382 natural graphite Inorganic materials 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 150000002898 organic sulfur compounds Chemical class 0.000 description 2
- 229920002493 poly(chlorotrifluoroethylene) Polymers 0.000 description 2
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 2
- 229920002647 polyamide Polymers 0.000 description 2
- 239000005023 polychlorotrifluoroethylene (PCTFE) polymer Substances 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 239000004926 polymethyl methacrylate Substances 0.000 description 2
- 239000004800 polyvinyl chloride Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011164 primary particle Substances 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- 229910052814 silicon oxide Inorganic materials 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 238000001179 sorption measurement Methods 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 229920006132 styrene block copolymer Polymers 0.000 description 2
- 150000003464 sulfur compounds Chemical class 0.000 description 2
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- 229910001887 tin oxide Inorganic materials 0.000 description 2
- 238000003466 welding Methods 0.000 description 2
- NUTJVZGIRRFKKI-UHFFFAOYSA-N (2-oxo-1,3-dioxolan-4-yl)methyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC1COC(=O)O1 NUTJVZGIRRFKKI-UHFFFAOYSA-N 0.000 description 1
- KCOYHFNCTWXETP-UHFFFAOYSA-N (carbamothioylamino)thiourea Chemical class NC(=S)NNC(N)=S KCOYHFNCTWXETP-UHFFFAOYSA-N 0.000 description 1
- BQCIDUSAKPWEOX-UHFFFAOYSA-N 1,1-Difluoroethene Chemical compound FC(F)=C BQCIDUSAKPWEOX-UHFFFAOYSA-N 0.000 description 1
- OQMIRQSWHKCKNJ-UHFFFAOYSA-N 1,1-difluoroethene;1,1,2,3,3,3-hexafluoroprop-1-ene Chemical group FC(F)=C.FC(F)=C(F)C(F)(F)F OQMIRQSWHKCKNJ-UHFFFAOYSA-N 0.000 description 1
- RXACTIULCNJUNO-UHFFFAOYSA-N 1,1-difluoroethene;1,1,2,3,3-pentafluoroprop-1-ene Chemical group FC(F)=C.FC(F)C(F)=C(F)F RXACTIULCNJUNO-UHFFFAOYSA-N 0.000 description 1
- VAYTZRYEBVHVLE-UHFFFAOYSA-N 1,3-dioxol-2-one Chemical compound O=C1OC=CO1 VAYTZRYEBVHVLE-UHFFFAOYSA-N 0.000 description 1
- COVXBJIKNGVTNV-UHFFFAOYSA-N 1-chloro-1,2,2-trifluoroethene;1,1-difluoroethene Chemical group FC(F)=C.FC(F)=C(F)Cl COVXBJIKNGVTNV-UHFFFAOYSA-N 0.000 description 1
- VSKJLJHPAFKHBX-UHFFFAOYSA-N 2-methylbuta-1,3-diene;styrene Chemical compound CC(=C)C=C.C=CC1=CC=CC=C1.C=CC1=CC=CC=C1 VSKJLJHPAFKHBX-UHFFFAOYSA-N 0.000 description 1
- NZUIYUOUCBYCEV-UHFFFAOYSA-N 2-prop-2-ynoxyethyl hydrogen carbonate Chemical compound OC(=O)OCCOCC#C NZUIYUOUCBYCEV-UHFFFAOYSA-N 0.000 description 1
- WUGCPRNXGBZRMC-UHFFFAOYSA-N 4,4-dimethyl-5-methylidene-1,3-dioxolan-2-one Chemical compound CC1(C)OC(=O)OC1=C WUGCPRNXGBZRMC-UHFFFAOYSA-N 0.000 description 1
- DQHCJQDPISNGEP-UHFFFAOYSA-N 4,5-bis(ethenyl)-1,3-dioxolan-2-one Chemical compound C=CC1OC(=O)OC1C=C DQHCJQDPISNGEP-UHFFFAOYSA-N 0.000 description 1
- SSPIBMWEJNSGAF-UHFFFAOYSA-N 4,5-diethyl-1,3-dioxol-2-one Chemical compound CCC=1OC(=O)OC=1CC SSPIBMWEJNSGAF-UHFFFAOYSA-N 0.000 description 1
- QYIOFABFKUOIBV-UHFFFAOYSA-N 4,5-dimethyl-1,3-dioxol-2-one Chemical compound CC=1OC(=O)OC=1C QYIOFABFKUOIBV-UHFFFAOYSA-N 0.000 description 1
- SROHGOJDCAODGI-UHFFFAOYSA-N 4,5-diphenyl-1,3-dioxol-2-one Chemical compound O1C(=O)OC(C=2C=CC=CC=2)=C1C1=CC=CC=C1 SROHGOJDCAODGI-UHFFFAOYSA-N 0.000 description 1
- UYCGHYYLNBVUGK-UHFFFAOYSA-N 4-(ethenoxymethyl)-1,3-dioxolan-2-one Chemical compound C=COCC1COC(=O)O1 UYCGHYYLNBVUGK-UHFFFAOYSA-N 0.000 description 1
- CANNDADZLNCIIA-UHFFFAOYSA-N 4-ethenyl-1,3-dioxol-2-one Chemical compound C=CC1=COC(=O)O1 CANNDADZLNCIIA-UHFFFAOYSA-N 0.000 description 1
- BJWMSGRKJIOCNR-UHFFFAOYSA-N 4-ethenyl-1,3-dioxolan-2-one Chemical compound C=CC1COC(=O)O1 BJWMSGRKJIOCNR-UHFFFAOYSA-N 0.000 description 1
- IXIDQWJXRMPFRX-UHFFFAOYSA-N 4-ethyl-1,3-dioxol-2-one Chemical compound CCC1=COC(=O)O1 IXIDQWJXRMPFRX-UHFFFAOYSA-N 0.000 description 1
- FOLJHXWWJYUOJV-UHFFFAOYSA-N 4-ethynyl-1,3-dioxolan-2-one Chemical compound O=C1OCC(C#C)O1 FOLJHXWWJYUOJV-UHFFFAOYSA-N 0.000 description 1
- HXXOPVULXOEHTK-UHFFFAOYSA-N 4-methyl-1,3-dioxol-2-one Chemical compound CC1=COC(=O)O1 HXXOPVULXOEHTK-UHFFFAOYSA-N 0.000 description 1
- VMAJRFCXVOIAAS-UHFFFAOYSA-N 4-phenyl-1,3-dioxol-2-one Chemical compound O1C(=O)OC=C1C1=CC=CC=C1 VMAJRFCXVOIAAS-UHFFFAOYSA-N 0.000 description 1
- WHZWLSAAGHOQPZ-UHFFFAOYSA-N 4-prop-2-ynyl-1,3-dioxolan-2-one Chemical compound O=C1OCC(CC#C)O1 WHZWLSAAGHOQPZ-UHFFFAOYSA-N 0.000 description 1
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical compound C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 229910020646 Co-Sn Inorganic materials 0.000 description 1
- 229910020709 Co—Sn Inorganic materials 0.000 description 1
- 229910017755 Cu-Sn Inorganic materials 0.000 description 1
- 229910017927 Cu—Sn Inorganic materials 0.000 description 1
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 229910018068 Li 2 O Inorganic materials 0.000 description 1
- 229910018127 Li 2 S-GeS 2 Inorganic materials 0.000 description 1
- 229910020725 Li0.34La0.51TiO3 Inorganic materials 0.000 description 1
- 229910010500 Li2.9PO3.3N0.46 Inorganic materials 0.000 description 1
- 229910009298 Li2S-P2S5-Li2O Inorganic materials 0.000 description 1
- 229910009320 Li2S-SiS2-LiBr Inorganic materials 0.000 description 1
- 229910009316 Li2S-SiS2-LiCl Inorganic materials 0.000 description 1
- 229910009318 Li2S-SiS2-LiI Inorganic materials 0.000 description 1
- 229910009219 Li2S—P2S5—Li2O Inorganic materials 0.000 description 1
- 229910007281 Li2S—SiS2—B2S3LiI Inorganic materials 0.000 description 1
- 229910007291 Li2S—SiS2—LiBr Inorganic materials 0.000 description 1
- 229910007288 Li2S—SiS2—LiCl Inorganic materials 0.000 description 1
- 229910007289 Li2S—SiS2—LiI Inorganic materials 0.000 description 1
- 229910015015 LiAsF 6 Inorganic materials 0.000 description 1
- 229910013063 LiBF 4 Inorganic materials 0.000 description 1
- 229910013684 LiClO 4 Inorganic materials 0.000 description 1
- 229910012851 LiCoO 2 Inorganic materials 0.000 description 1
- 229910010707 LiFePO 4 Inorganic materials 0.000 description 1
- 229910010833 LiI-Li2S-SiS2 Inorganic materials 0.000 description 1
- 229910010842 LiI—Li2S—P2O5 Inorganic materials 0.000 description 1
- 229910010855 LiI—Li2S—SiS2 Inorganic materials 0.000 description 1
- 229910015643 LiMn 2 O 4 Inorganic materials 0.000 description 1
- 229910014689 LiMnO Inorganic materials 0.000 description 1
- 229910013716 LiNi Inorganic materials 0.000 description 1
- 229910013290 LiNiO 2 Inorganic materials 0.000 description 1
- 229910013870 LiPF 6 Inorganic materials 0.000 description 1
- 239000002228 NASICON Substances 0.000 description 1
- 229910018539 Ni—Mn—Co Inorganic materials 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 229910003870 O—Li Inorganic materials 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- 239000004721 Polyphenylene oxide Substances 0.000 description 1
- 238000001237 Raman spectrum Methods 0.000 description 1
- 229920000297 Rayon Polymers 0.000 description 1
- 229910001128 Sn alloy Inorganic materials 0.000 description 1
- 229910008240 SnB0.4P0.6O3.1 Inorganic materials 0.000 description 1
- 229910005790 SnSiO Inorganic materials 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 239000002174 Styrene-butadiene Substances 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- 229910052977 alkali metal sulfide Inorganic materials 0.000 description 1
- 229910002064 alloy oxide Inorganic materials 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 229910052800 carbon group element Inorganic materials 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 229910010293 ceramic material Inorganic materials 0.000 description 1
- 150000005678 chain carbonates Chemical group 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 238000010280 constant potential charging Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- KUNSUQLRTQLHQQ-UHFFFAOYSA-N copper tin Chemical compound [Cu].[Sn] KUNSUQLRTQLHQQ-UHFFFAOYSA-N 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 239000002178 crystalline material Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- JLQNHALFVCURHW-UHFFFAOYSA-N cyclooctasulfur Chemical compound S1SSSSSSS1 JLQNHALFVCURHW-UHFFFAOYSA-N 0.000 description 1
- 235000013365 dairy product Nutrition 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 150000002019 disulfides Chemical class 0.000 description 1
- OAEGRYMCJYIXQT-UHFFFAOYSA-N dithiooxamide Chemical compound NC(=S)C(N)=S OAEGRYMCJYIXQT-UHFFFAOYSA-N 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 230000005611 electricity Effects 0.000 description 1
- 239000003822 epoxy resin Substances 0.000 description 1
- HQQADJVZYDDRJT-UHFFFAOYSA-N ethene;prop-1-ene Chemical group C=C.CC=C HQQADJVZYDDRJT-UHFFFAOYSA-N 0.000 description 1
- 239000005038 ethylene vinyl acetate Substances 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 229910003472 fullerene Inorganic materials 0.000 description 1
- 239000006232 furnace black Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 1
- 229910021385 hard carbon Inorganic materials 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 238000010884 ion-beam technique Methods 0.000 description 1
- NONOKGVFTBWRLD-UHFFFAOYSA-N isocyanatosulfanylimino(oxo)methane Chemical compound O=C=NSN=C=O NONOKGVFTBWRLD-UHFFFAOYSA-N 0.000 description 1
- 238000010030 laminating Methods 0.000 description 1
- 238000007561 laser diffraction method Methods 0.000 description 1
- 229910000664 lithium aluminum titanium phosphates (LATP) Inorganic materials 0.000 description 1
- HSZCZNFXUDYRKD-UHFFFAOYSA-M lithium iodide Inorganic materials [Li+].[I-] HSZCZNFXUDYRKD-UHFFFAOYSA-M 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 238000003701 mechanical milling Methods 0.000 description 1
- 239000004570 mortar (masonry) Substances 0.000 description 1
- 239000002116 nanohorn Substances 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 150000002829 nitrogen Chemical class 0.000 description 1
- 239000011255 nonaqueous electrolyte Substances 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 239000005486 organic electrolyte Substances 0.000 description 1
- DAFIBNSJXIGBQB-UHFFFAOYSA-N perfluoroisobutene Chemical compound FC(F)=C(C(F)(F)F)C(F)(F)F DAFIBNSJXIGBQB-UHFFFAOYSA-N 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 238000007747 plating Methods 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 1
- 229920002857 polybutadiene Polymers 0.000 description 1
- 229920001083 polybutene Polymers 0.000 description 1
- 229920001707 polybutylene terephthalate Polymers 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920000647 polyepoxide Polymers 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920001195 polyisoprene Polymers 0.000 description 1
- 239000011116 polymethylpentene Substances 0.000 description 1
- 229920000306 polymethylpentene Polymers 0.000 description 1
- 239000005077 polysulfide Substances 0.000 description 1
- 229920001021 polysulfide Polymers 0.000 description 1
- 150000008117 polysulfides Polymers 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 239000002964 rayon Substances 0.000 description 1
- 238000006479 redox reaction Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000006722 reduction reaction Methods 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 238000000790 scattering method Methods 0.000 description 1
- LIVNPJMFVYWSIS-UHFFFAOYSA-N silicon monoxide Chemical compound [Si-]#[O+] LIVNPJMFVYWSIS-UHFFFAOYSA-N 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 235000002639 sodium chloride Nutrition 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 229910021384 soft carbon Inorganic materials 0.000 description 1
- 238000010532 solid phase synthesis reaction Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 239000006234 thermal black Substances 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 125000001391 thioamide group Chemical group 0.000 description 1
- UMGDCJDMYOKAJW-UHFFFAOYSA-N thiourea group Chemical group NC(=S)N UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- QHGNHLZPVBIIPX-UHFFFAOYSA-N tin(ii) oxide Chemical class [Sn]=O QHGNHLZPVBIIPX-UHFFFAOYSA-N 0.000 description 1
- YFHICDDUDORKJB-UHFFFAOYSA-N trimethylene carbonate Chemical compound O=C1OCCCO1 YFHICDDUDORKJB-UHFFFAOYSA-N 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 239000013585 weight reducing agent Substances 0.000 description 1
- 238000004804 winding Methods 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0561—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of inorganic materials only
- H01M10/0562—Solid materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
- H01M4/624—Electric conductive fillers
- H01M4/625—Carbon or graphite
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0065—Solid electrolytes
- H01M2300/0068—Solid electrolytes inorganic
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0065—Solid electrolytes
- H01M2300/0068—Solid electrolytes inorganic
- H01M2300/008—Halides
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- the present invention relates to a positive electrode material for an electric device, and a positive electrode for an electric device and an electric device using the same.
- the lithium secondary battery As a secondary battery for driving a motor, it is required to have extremely high output characteristics and high energy as compared with a consumer lithium secondary battery used for mobile phones, notebook computers, and the like. Therefore, the lithium secondary battery, which has the highest theoretical energy among all realistic batteries, is attracting attention and is currently being rapidly developed.
- the lithium secondary battery currently widely used uses a flammable organic electrolyte as the electrolyte.
- a flammable organic electrolyte as the electrolyte.
- safety measures against liquid leakage, short circuit, overcharge, etc. are required more strictly than other batteries.
- the solid electrolyte is a material composed mainly of an ionic conductor capable of ionic conduction in a solid. Therefore, in the all-solid-state lithium secondary battery, various problems caused by the flammable organic electrolytic solution do not occur in principle unlike the conventional liquid-based lithium secondary battery. Further, in general, when a high potential / large capacity positive electrode material and a large capacity negative electrode material are used, the output density and energy density of the battery can be significantly improved. For example, elemental sulfur ( S8 ) has an extremely large theoretical capacity of about 1670 mAh / g, and has the advantages of low cost and abundant resources.
- metallic lithium which is a negative electrode active material that supplies lithium ions to the positive electrode
- the battery characteristics may deteriorate as a result of the reaction between the metallic lithium and the sulfide solid electrolyte. ..
- Patent Document 1 for the purpose of dealing with such a problem, a composite material containing a conductive agent and an alkali metal sulfide integrated on the surface of the conductive agent is used as a positive electrode material for an all-solid-state battery.
- the technique to be used as is proposed.
- Patent Document 1 by using a positive electrode material having such a configuration, a positive electrode material and a lithium ion battery having a high theoretical capacity and capable of using a negative electrode active material that does not supply lithium ions to the positive electrode are provided.
- an object of the present invention is to provide a means capable of improving the capacity characteristics and the charge / discharge rate characteristics in an electric device using a positive electrode active material containing sulfur.
- the present inventors have made diligent studies to solve the above problems.
- the solid electrolyte and the positive electrode active material containing sulfur are arranged on the inner surface of the pores so as to be in contact with each other. It has been found that the above-mentioned problems can be solved by configuring the above-mentioned structure, and the present invention has been completed.
- One embodiment of the present invention includes a conductive material having pores, a solid electrolyte, and a positive electrode active material containing sulfur, and at least a part of the solid electrolyte and at least a part of the positive electrode active material are in contact with each other. It is a positive electrode material for an electric device, which is arranged on the inner surface of the pores as described above.
- FIG. 1 is a perspective view showing the appearance of a flat laminated all-solid-state lithium-ion secondary battery according to an embodiment of the lithium-ion secondary battery according to the present invention.
- FIG. 2 is a cross-sectional view taken along line 2-2 shown in FIG.
- FIG. 3A is a schematic cross-sectional view of the positive electrode material in the prior art.
- FIG. 3B is a schematic cross-sectional view of a positive electrode material according to an embodiment of the present invention.
- FIG. 4A is an observation image of the powder particles of the sulfur-containing positive electrode material obtained in Example 1 by a scanning electron microscope (SEM).
- SEM scanning electron microscope
- FIG. 4B is an elemental map of the phosphorus (P) element in the observation image of the cross section of the conductive material by TEM-EDX in the sulfur-containing positive electrode material obtained in Example 1.
- FIG. 5A is an observation image of the powder particles of the sulfur-containing positive electrode material obtained in Comparative Example 1 by a scanning electron microscope (SEM).
- FIG. 5B is an elemental map of the phosphorus (P) element in the observation image of the cross section of the conductive material by TEM-EDX in the sulfur-containing positive electrode material obtained in Comparative Example 1.
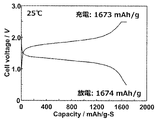
- FIG. 6 is a charge / discharge curve for the test cell (all-solid-state lithium-ion secondary battery) produced in Example 2.
- the present invention will be described by taking as an example a laminated type (internal parallel connection type) all-solid-state lithium secondary battery, which is a form of a secondary battery.
- the solid electrolyte constituting the all-solid-state lithium secondary battery is a material mainly composed of an ionic conductor capable of ionic conduction in a solid.
- the all-solid-state lithium secondary battery has an advantage that various problems caused by the flammable organic electrolytic solution do not occur in principle unlike the conventional liquid-based lithium secondary battery.
- the use of a high-potential, large-capacity positive electrode material and a large-capacity negative electrode material has the advantage that the output density and energy density of the battery can be significantly improved.
- One embodiment of the present invention includes a conductive material having pores, a solid electrolyte, and a positive electrode active material containing sulfur, and at least a part of the solid electrolyte and at least a part of the positive electrode active material are in contact with each other. It is a positive electrode material for an electric device, which is arranged on the inner surface of the pores as described above. According to the positive electrode material for an electric device according to the present embodiment, the capacity characteristics and the charge / discharge rate characteristics of an electric device such as an all-solid-state lithium ion secondary battery are improved despite the use of a positive electrode active material containing sulfur. be able to.
- FIG. 1 is a perspective view showing the appearance of a flat laminated all-solid-state lithium-ion secondary battery according to an embodiment of the lithium-ion secondary battery according to the present invention.
- FIG. 2 is a cross-sectional view taken along line 2-2 shown in FIG. By making it a laminated type, the battery can be made compact and has a high capacity.
- laminated battery hereinafter, also simply referred to as “laminated battery”
- the laminated battery 10a has a rectangular flat shape, and a negative electrode current collector plate 25 and a positive electrode current collector plate 27 for extracting electric power are pulled out from both sides thereof.
- the power generation element 21 is wrapped with a battery exterior material (laminated film 29) of the laminated battery 10a, and the periphery thereof is heat-sealed.
- the power generation element 21 has a negative electrode current collector plate 25 and a positive electrode current collector plate 27 external to the power generation element 21. It is sealed in the state of being pulled out.
- the lithium ion secondary battery according to this embodiment is not limited to a laminated flat battery.
- the wound lithium-ion secondary battery may have a cylindrical shape, or may be formed by deforming such a cylindrical shape into a rectangular flat shape.
- a laminated film may be used for the exterior material, or a conventional cylindrical can (metal can) may be used, and the present invention is not particularly limited.
- the power generation element is housed inside a laminated film containing aluminum. By this form, weight reduction can be achieved.
- the removal of the current collector plates (25, 27) shown in FIG. 1 is not particularly limited.
- the negative electrode current collector plate 25 and the positive electrode current collector plate 27 may be pulled out from the same side, or the negative electrode current collector plate 25 and the positive electrode current collector plate 27 may be divided into a plurality of parts and taken out from each side. It is not limited to what is shown in FIG. 1, such as good.
- the terminal in the winding type lithium ion battery, the terminal may be formed by using, for example, a cylindrical can (metal can) instead of the tab.
- the laminated battery 10a of the present embodiment has a structure in which a flat, substantially rectangular power generation element 21 in which a charge / discharge reaction actually proceeds is sealed inside a laminated film 29 which is a battery exterior material.
- the power generation element 21 has a configuration in which a positive electrode, a solid electrolyte layer 17, and a negative electrode are laminated.
- the positive electrode has a structure in which the positive electrode active material layer 15 containing the positive electrode active material is arranged on both sides of the positive electrode current collector 11 ′′.
- the negative electrode is a negative electrode containing the negative electrode active material on both sides of the negative electrode current collector 11 ′. It has a structure in which the active material layer 13 is arranged.
- one positive electrode active material layer 15 and the negative electrode active material layer 13 adjacent thereto are opposed to each other via the solid electrolyte layer 17.
- the positive electrode, the solid electrolyte layer, and the negative electrode are laminated in this order.
- the adjacent positive electrode, the solid electrolyte layer, and the negative electrode constitute one cell cell layer 19. Therefore, the laminated battery 10a shown in FIG. 1 is It can be said that a plurality of cell cell layers 19 are laminated so as to have a configuration in which they are electrically connected in parallel.
- the negative electrode active material layer 13 is arranged on only one side, but the active material layers are provided on both sides. May be done. That is, instead of using a current collector dedicated to the outermost layer having an active material layer on only one side, a current collector having active material layers on both sides may be used as it is as a current collector for the outermost layer. Further, in some cases, the negative electrode active material layer 13 and the positive electrode active material layer 15 may be used as the negative electrode and the positive electrode, respectively, without using the current collector (11', 11 ").
- the negative electrode current collector 11'and the positive electrode current collector 11' are attached with a negative electrode current collector plate (tab) 25 and a positive electrode current collector plate (tab) 27 that are conductive to each electrode (positive electrode and negative electrode), respectively, and the battery exterior. It has a structure that is led out to the outside of the laminated film 29 so as to be sandwiched between the ends of the laminated film 29, which is a material.
- the positive electrode current collector plate 27 and the negative electrode current collector plate 25 are positive electrodes, if necessary. It may be attached to the positive electrode current collector 11 "and the negative electrode current collector 11'of each electrode by ultrasonic welding, resistance welding, or the like via a lead and a negative electrode lead (not shown).
- the current collector has a function of mediating the movement of electrons from the electrode active material layer.
- the materials that make up the current collector There are no particular restrictions on the materials that make up the current collector.
- a constituent material of the current collector for example, a metal or a resin having conductivity can be adopted.
- examples of the metal include aluminum, nickel, iron, stainless steel, titanium, and copper.
- a clad material of nickel and aluminum, a clad material of copper and aluminum, and the like may be used.
- the foil may be a metal surface coated with aluminum.
- aluminum, stainless steel, copper, and nickel are preferable from the viewpoints of electron conductivity, battery operating potential, adhesion of the negative electrode active material by sputtering to the current collector, and the like.
- examples of the latter resin having conductivity include a resin in which a conductive filler is added to a non-conductive polymer material as needed.
- non-conductive polymer material examples include polyethylene (PE; high density polyethylene (HDPE), low density polyethylene (LDPE), etc.), polypropylene (PP), polyethylene terephthalate (PET), polyether nitrile (PEN), and polyimide.
- PE polyethylene
- HDPE high density polyethylene
- LDPE low density polyethylene
- PP polypropylene
- PET polyethylene terephthalate
- PEN polyether nitrile
- PI Polyimide
- PAI Polypropylene
- PA Polytetrafluoroethylene
- SBR Styrene-butadiene rubber
- PAN Polyacrylonitrile
- PMA Polymethylacrylate
- PMMA Polymethylmethacrylate
- PVC Polyvinyl chloride
- PVdF polyvinylidene fluoride
- PS polystyrene
- Such non-conductive polymer materials may have excellent potential or solvent resistance.
- a conductive filler may be added to the above-mentioned conductive polymer material or non-conductive polymer material as needed.
- a conductive filler is inevitably indispensable in order to impart conductivity to the resin.
- the conductive filler can be used without particular limitation as long as it is a conductive substance.
- materials having excellent conductivity, potential resistance, or lithium ion blocking property include metals and conductive carbon.
- the metal is not particularly limited, and includes at least one metal selected from the group consisting of Ni, Ti, Al, Cu, Pt, Fe, Cr, Sn, Zn, In, and Sb, or at least one of these metals. It preferably contains an alloy or metal oxide.
- the conductive carbon is not particularly limited.
- acetylene black is selected from the group consisting of acetylene black, vulcan (registered trademark), black pearl (registered trademark), carbon nanofiber, Ketjen black (registered trademark), carbon nanotube, carbon nanohorn, carbon nanoballoon, and fullerene. It contains at least one species.
- the amount of the conductive filler added is not particularly limited as long as it can impart sufficient conductivity to the current collector, and is generally 5 to 80% by mass with respect to 100% by mass of the total mass of the current collector. Is.
- the current collector may have a single-layer structure made of a single material, or may have a laminated structure in which layers made of these materials are appropriately combined. From the viewpoint of reducing the weight of the current collector, it is preferable to include a conductive resin layer made of at least a conductive resin. Further, from the viewpoint of blocking the movement of lithium ions between the cells of the cell, a metal layer may be provided on a part of the current collector. Further, if the negative electrode active material layer and the positive electrode active material layer, which will be described later, have conductivity by themselves and can exhibit the current collecting function, a current collector as a member different from these electrode active material layers is used. It doesn't have to be. In such a form, the negative electrode active material layer described later constitutes the negative electrode as it is, and the positive electrode active material layer described later constitutes the positive electrode as it is.
- the negative electrode active material layer 13 contains a negative electrode active material.
- the type of the negative electrode active material is not particularly limited, and examples thereof include a carbon material, a metal oxide, and a metal active material.
- the carbon material include natural graphite, artificial graphite, mesocarbon microbeads (MCMB), highly oriented graphite (HOPG), hard carbon, soft carbon and the like.
- the metal oxide include Nb 2 O 5 and Li 4 Ti 5 O 12 .
- a silicon-based negative electrode active material or a tin-based negative electrode active material may be used.
- silicon and tin belong to Group 14 elements and are known to be negative electrode active materials that can greatly improve the capacity of a non-aqueous electrolyte secondary battery. Since these simple substances can occlude and release a large number of charge carriers (lithium ions, etc.) per unit volume (mass), they are high-capacity negative electrode active materials.
- Si alone as the silicon-based negative electrode active material.
- a silicon oxide such as SiO x (0.3 ⁇ x ⁇ 1.6) disproportionated into two phases, a Si phase and a silicon oxide phase.
- the range of x is more preferably 0.5 ⁇ x ⁇ 1.5, and further preferably 0.7 ⁇ x ⁇ 1.2.
- a silicon-containing alloy silicon-containing alloy-based negative electrode active material
- examples of the negative electrode active material containing a tin element include Sn alone, tin alloys (Cu—Sn alloys, Co—Sn alloys), amorphous tin oxides, tin silicon oxides and the like. Of these, SnB 0.4 P 0.6 O 3.1 is exemplified as the amorphous tin oxide. Further, SnSiO 3 is exemplified as the tin silicon oxide.
- a metal containing lithium may be used as the negative electrode active material.
- a negative electrode active material is not particularly limited as long as it is a lithium-containing active material, and examples thereof include metallic lithium and lithium-containing alloys.
- the lithium-containing alloy include alloys of Li and at least one of In, Al, Si and Sn.
- two or more kinds of negative electrode active materials may be used in combination.
- a negative electrode active material other than the above may be used.
- the negative electrode active material preferably contains metallic lithium, a silicon-based negative electrode active material, or a tin-based negative electrode active material, and particularly preferably contains metallic lithium.
- the shape of the negative electrode active material examples include particulate (spherical and fibrous) and thin film.
- its average particle size (D 50 ) is preferably in the range of, for example, 1 nm to 100 ⁇ m, more preferably in the range of 10 nm to 50 ⁇ m, and further preferably in the range of 100 nm. It is in the range of ⁇ 20 ⁇ m, and particularly preferably in the range of 1 to 20 ⁇ m.
- the value of the average particle size (D 50 ) of the active material can be measured by the laser diffraction / scattering method.
- the content of the negative electrode active material in the negative electrode active material layer is not particularly limited, but is preferably in the range of, for example, 40 to 99% by mass, and preferably in the range of 50 to 90% by mass. More preferred.
- the negative electrode active material layer preferably further contains a solid electrolyte. Since the negative electrode active material layer contains a solid electrolyte, the ionic conductivity of the negative electrode active material layer can be improved.
- the solid electrolyte include a sulfide solid electrolyte and an oxide solid electrolyte, and a sulfide solid electrolyte is preferable.
- Examples of the sulfide solid electrolyte include LiI-Li 2 S-SiS 2 , LiI-Li 2 S-P 2 O 5 , LiI-Li 3 PO 4 -P 2 S 5 , Li 2 S-P 2 S 5 , and Li 2 S-P 2 S 5.
- the sulfide solid electrolyte may have, for example, a Li 3 PS 4 skeleton, a Li 4 P 2 S 7 skeleton, or a Li 4 P 2 S 6 skeleton. ..
- Examples of the sulfide solid electrolyte having a Li 3 PS 4 skeleton include LiI-Li 3 PS 4 , LiI-LiBr-Li 3 PS 4, and Li 3 PS 4 .
- Examples of the sulfide solid electrolyte having a Li 4 P 2 S 7 skeleton include a Li-PS-based solid electrolyte called LPS (for example, Li 7 P 3 S 11 ).
- the sulfide solid electrolyte for example, LGPS represented by Li (4-x) Ge (1-x) P x S 4 (x satisfies 0 ⁇ x ⁇ 1) may be used.
- the sulfide solid electrolyte contained in the active material layer is preferably a sulfide solid electrolyte containing P element, and the sulfide solid electrolyte is a material containing Li 2 SP 2 S 5 as a main component. It is more preferable to have.
- the sulfide solid electrolyte may contain halogen (F, Cl, Br, I).
- the sulfide solid electrolyte comprises Li 6 PS 5 X, where X is Cl, Br or I, preferably Cl.
- the sulfide solid electrolyte may be sulfide glass, crystallized sulfide glass, or a crystalline material obtained by the solid phase method.
- the sulfide glass can be obtained, for example, by performing mechanical milling (ball mill or the like) on the raw material composition.
- the crystallized sulfide glass can be obtained, for example, by heat-treating the sulfide glass at a temperature equal to or higher than the crystallization temperature.
- the ionic conductivity (for example, Li ionic conductivity) of the sulfide solid electrolyte at room temperature (25 ° C.) is preferably 1 ⁇ 10 -5 S / cm or more, for example, 1 ⁇ 10 -4 S / cm. It is more preferably cm or more.
- the value of the ionic conductivity of the solid electrolyte can be measured by the AC impedance method.
- Examples of the oxide solid electrolyte include compounds having a NASICON type structure and the like.
- a compound having a NASION type structure a compound (LAGP) represented by the general formula Li 1 + x Al x Ge 2-x (PO 4 ) 3 (0 ⁇ x ⁇ 2), a general formula Li 1 + x Al x Ti 2
- LAGP a compound represented by the general formula Li 1 + x Al x Ge 2-x (PO 4 ) 3 (0 ⁇ x ⁇ 2)
- a general formula Li 1 + x Al x Ti 2 examples thereof include a compound (LATP) represented by ⁇ x (PO 4 ) 3 (0 ⁇ x ⁇ 2).
- LiLaTIO for example, Li 0.34 La 0.51 TiO 3
- LiPON for example, Li 2.9 PO 3.3 N 0.46
- LiLaZrO for example, LiLaZrO
- the shape of the solid electrolyte examples include a particle shape such as a true spherical shape and an elliptical spherical shape, and a thin film shape.
- its average particle size (D 50 ) is not particularly limited, but is preferably 40 ⁇ m or less, more preferably 20 ⁇ m or less, still more preferably 10 ⁇ m or less.
- the average particle size (D 50 ) is preferably 0.01 ⁇ m or more, and more preferably 0.1 ⁇ m or more.
- the content of the solid electrolyte in the negative electrode active material layer is, for example, preferably in the range of 1 to 60% by mass, and more preferably in the range of 10 to 50% by mass.
- the negative electrode active material layer may further contain at least one of a conductive auxiliary agent and a binder in addition to the negative electrode active material and the solid electrolyte described above.
- the conductive auxiliary agent examples include metals such as aluminum, stainless steel (SUS), silver, gold, copper and titanium, alloys or metal oxides containing these metals; carbon fiber (specifically, vapor-grown carbon fiber). (VGCF), polyacrylonitrile-based carbon fiber, pitch-based carbon fiber, rayon-based carbon fiber, activated carbon fiber, etc.), carbon nanotube (CNT), carbon black (specifically, acetylene black, Ketjen black (registered trademark)) , Furness black, channel black, thermal lamp black, etc.), but is not limited to these. Further, a particulate ceramic material or a resin material coated with the above metal material by plating or the like can also be used as a conductive auxiliary agent.
- metals such as aluminum, stainless steel (SUS), silver, gold, copper and titanium, alloys or metal oxides containing these metals
- carbon fiber specifically, vapor-grown carbon fiber). (VGCF), polyacrylonitrile-based carbon fiber, pitch-based carbon fiber, rayon-based carbon fiber, activate
- these conductive auxiliaries from the viewpoint of electrical stability, it is preferable to contain at least one selected from the group consisting of aluminum, stainless steel, silver, gold, copper, titanium, and carbon, and aluminum, stainless steel. It is more preferable to contain at least one selected from the group consisting of silver, gold, and carbon, and even more preferably to contain at least one carbon. Only one kind of these conductive auxiliary agents may be used alone, or two or more kinds thereof may be used in combination.
- the shape of the conductive auxiliary agent is preferably particulate or fibrous.
- the shape of the particles is not particularly limited, and may be any shape such as powder, sphere, rod, needle, plate, columnar, indefinite, fluffy, and spindle-shaped. It doesn't matter.
- the average particle size (primary particle size) when the conductive auxiliary agent is in the form of particles is not particularly limited, but is preferably 0.01 to 10 ⁇ m from the viewpoint of the electrical characteristics of the battery.
- the "particle diameter of the conductive auxiliary agent” means the maximum distance L among the distances between arbitrary two points on the contour line of the conductive auxiliary agent.
- the particle size of the particles observed in several to several tens of fields using an observation means such as a scanning electron microscope (SEM) or a transmission electron microscope (TEM). The value calculated as the average value of is adopted.
- the content of the conductive auxiliary agent in the negative electrode active material layer is not particularly limited, but is preferably 0 to 10% by mass with respect to the total mass of the negative electrode active material layer. , More preferably 2 to 8% by mass, still more preferably 4 to 7% by mass. Within such a range, it is possible to form a stronger electron conduction path in the negative electrode active material layer, and it is possible to effectively contribute to the improvement of battery characteristics.
- the binder is not particularly limited, and examples thereof include the following materials.
- Fluororesin such as ethylene / tetrafluoroethylene copolymer (ETFE), polychlorotrifluoroethylene (PCTFE), ethylene / chlorotrifluoroethylene copolymer (ECTFE), polyvinylfluorovinyl (PVF), vinylidene fluoride- Hexafluoropropylene-based fluororubber (VDF-HFP-based fluororubber), vinylidene fluoride-hexafluoropropylene-tetrafluoroethylene-based fluororubber (VDF-HFP-TFE-based fluororubber), vinylidene fluoride-pentafluoropropylene-based fluororubber (VDF-PFP-based fluorubber), vinylidene fluoride-pentafluoropropylene-tetrafluoroethylene-based fluororubber (VDF-PFP-TFE-based fluorubber), vinylidene fluoride-p
- the thickness of the negative electrode active material layer varies depending on the configuration of the target secondary battery, but is preferably in the range of 0.1 to 1000 ⁇ m, for example.
- the solid electrolyte layer is a layer interposed between the above-mentioned positive electrode active material layer and the negative electrode active material layer and essentially containing the solid electrolyte.
- the specific form of the solid electrolyte contained in the solid electrolyte layer is not particularly limited, and the solid electrolyte exemplified in the column of the negative electrode active material layer and its preferred form can be similarly adopted. In some cases, a solid electrolyte other than the above-mentioned solid electrolyte may be used in combination.
- the solid electrolyte layer may further contain a binder in addition to the predetermined solid electrolyte described above.
- a binder in addition to the predetermined solid electrolyte described above.
- the binder that can be contained in the solid electrolyte layer the examples and preferred forms described in the column of the negative electrode active material layer can be similarly adopted.
- the thickness of the solid electrolyte layer varies depending on the configuration of the target lithium ion secondary battery, but is preferably 600 ⁇ m or less, more preferably 500 ⁇ m or less, from the viewpoint of improving the volumetric energy density of the battery. , More preferably 400 ⁇ m or less.
- the lower limit of the thickness of the solid electrolyte layer is not particularly limited, but is preferably 1 ⁇ m or more, more preferably 5 ⁇ m or more, and further preferably 10 ⁇ m or more.
- the positive electrode active material layer contains the positive electrode material for an electric device according to one embodiment of the present invention.
- the positive electrode material for an electric device includes a conductive material having pores, a solid electrolyte, and a positive electrode active material containing sulfur.
- the type of the positive electrode active material containing sulfur is not particularly limited, and examples thereof include particles or thin films of an organic sulfur compound or an inorganic sulfur compound in addition to elemental sulfur (S), which can be charged by utilizing the oxidation-reduction reaction of sulfur. Any substance may be used as long as it can release lithium ions at times and can store lithium ions at the time of discharge.
- the organic sulfur compound include a disulfide compound, a sulfur-modified polyacrylonitrile represented by the compound described in International Publication No. 2010/0444437, a sulfur-modified polyisoprene, rubianic acid (dithiooxamide), and polysulfide carbon.
- disulfide compounds sulfur-modified polyacrylonitrile, and rubianic acid are preferable, and sulfur-modified polyacrylonitrile is particularly preferable.
- the disulfide compound a compound having a dithiobiurea derivative, a thiourea group, a thioisocyanate, or a thioamide group is more preferable.
- the sulfur-modified polyacrylonitrile is a modified polyacrylonitrile containing a sulfur atom, which is obtained by mixing sulfur powder and polyacrylonitrile and heating them under an inert gas or under reduced pressure.
- the estimated structure is, for example, Chem. Mater.
- the polyacrylonitrile is ring-closed to form a polycyclic structure, and at least a part of S is bound to C.
- the compounds described in this document have strong peak signals near 1330 cm -1 and 1560 cm -1 in Raman spectra, and peaks near 307 cm -1 , 379 cm -1 , 472 cm -1 , and 929 cm -1 . do.
- inorganic sulfur compounds are preferable because they are excellent in stability.
- S, S-carbon composite, TiS 2 , TiS 3 , TiS 4 , FeS 2 and MoS 2 are preferable, and elemental sulfur (S), TiS 2 and FeS 2 are more preferable, and from the viewpoint of high capacity.
- Elementary sulfur (S) is particularly preferred.
- As the elemental sulfur (S), ⁇ -sulfur, ⁇ -sulfur, or ⁇ -sulfur having an S8 structure can be used.
- the positive electrode material according to this embodiment may further contain a positive electrode active material containing no sulfur in addition to the positive electrode active material containing sulfur.
- the sulfur-free positive electrode active material include layered rock salt type active materials such as LiCoO 2 , LiMnO 2 , LiNiO 2 , LiVO 2 , and Li (Ni-Mn-Co) O 2 , LiMn 2 O 4 , LiNi 0. 5
- spinel-type active materials such as Mn 1.5 O 4
- olivine-type active materials such as LiFePO 4 and LiMnPO 4
- Si-containing active materials such as Li 2 FeSiO 4 and Li 2 MnSiO 4 .
- the oxide active material other than the above include Li 4 Ti 5 O 12 .
- two or more kinds of positive electrode active materials may be used in combination.
- a positive electrode active material other than the above may be used.
- the ratio of the content of the positive electrode active material containing sulfur to 100% by mass of the total amount of the positive electrode active material is preferably 50% by mass or more, more preferably 70% by mass or more, and further preferably 80% by mass.
- the above is more preferably 90% by mass or more, particularly preferably 95% by mass or more, and most preferably 100% by mass.
- the positive electrode material according to this embodiment indispensably contains a solid electrolyte.
- the specific form of the solid electrolyte contained in the positive electrode material according to the present embodiment is not particularly limited, and the solid electrolyte exemplified in the column of the negative electrode active material layer and its preferred form can be similarly adopted. In some cases, a solid electrolyte other than the above-mentioned solid electrolyte may be used in combination.
- the solid electrolyte contained in the positive electrode material according to this embodiment is preferably a sulfide solid electrolyte.
- the solid electrolyte contained in the solid electrolyte layer contains alkali metal atoms.
- the alkali metal that can be contained in the solid electrolyte include Li, Na, and K, and Li is particularly preferable because it has excellent ionic conductivity.
- the solid electrolyte layer contained in the solid electrolyte layer contains alkali metal atoms (eg, Li, Na or K; preferably Li) and phosphorus and / or boron atoms.
- Examples of the sulfide solid electrolyte containing such an alkali metal atom and a phosphorus atom and / or a boron atom include LiI-Li 2 SP 2 O 5 and LiI-Li 3 PO 4 -P 2 S 5 .
- Li 2 SP 2 S 5 LiI-Li 3 PS 4 , LiI-LiBr-Li 3 PS 4, Li 3 PS 4, Li 2 SP 2 S 5 , Li 2 SP 2 S 5 -LiI , Li 2 S-P 2 S 5 -Li 2 O, Li 2 S-P 2 S 5 -Li 2 O-Li I, Li 2 S-SiS 2 -B 2 S 3 -Li I, Li 2 S-SiS 2- P 2 S 5 -LiI, Li 2 SB 2 S 3 , Li 2 S-P 2 S 5 -Z m Sn (where m and n are positive numbers, Z is Ge, Zn, Ga ), Li 2 S-SiS 2 -Li 3 PO 4 , and the like.
- the sulfide solid electrolyte having a Li 4 P 2 S 7 skeleton examples include a Li-PS-based solid electrolyte called LPS (for example, Li 7 P 3 S 11 ). Further, for example, LGPS represented by Li (4-x) Ge (1-x) P x S 4 (x satisfies 0 ⁇ x ⁇ 1) may be used.
- the sulfide solid electrolyte contained in the active material layer is preferably a sulfide solid electrolyte containing a phosphorus atom, and the sulfide solid electrolyte is a material containing Li 2 SP 2 S 5 as a main component. It is more preferable to have.
- the sulfide solid electrolyte may contain halogen (F, Cl, Br, I).
- the sulfide solid electrolyte comprises Li 6 PS 5 X, where X is Cl, Br or I, preferably Cl. Since these solid electrolytes have high ionic conductivity, they can effectively contribute to the manifestation of the effects of the present invention.
- the positive electrode material according to this embodiment indispensably contains a conductive material having pores.
- the specific form of the conductive material contained in the positive electrode material according to this embodiment is not particularly limited as long as it has pores, and conventionally known materials can be appropriately adopted. From the viewpoint of excellent conductivity, easy processing, and easy design of a desired pore distribution, the conductive material having pores is preferably a carbon material.
- Examples of the carbon material having pores include activated carbon, Ketjen black (registered trademark) (highly conductive carbon black), (oil) furnace black, channel black, acetylene black, thermal black, and carbon black such as lamp black. Examples thereof include carbon particles (carbon carriers) made of coke, natural graphite, artificial graphite and the like.
- the main component of the carbon material is preferably carbon.
- the main component is carbon means that carbon atoms are contained as the main component, and is a concept including both carbon atoms and substantially carbon atoms.
- “Substantially composed of carbon atoms” means that impurities of about 2 to 3% by mass or less can be mixed.
- the BET specific surface area of the conductive material having pores is preferably 200 m 2 / g or more, more preferably 500 m 2 / g or more, and more preferably 800 m 2 / g or more. Is more preferable, and 1200 m 2 / g or more is particularly preferable, and 1500 m 2 / g or more is most preferable.
- the pore volume of the conductive material having pores (preferably a carbon material) is preferably 1.0 mL / g or more, more preferably 1.3 mL / g or more, and 1.5 mL / g. It is more preferably g or more.
- the values of the BET specific surface area and the pore volume of the conductive material can be measured by nitrogen adsorption / desorption measurement. This nitrogen adsorption / desorption measurement is carried out using BELSORP mini manufactured by Microtrac Bell Co., Ltd., and is carried out by a multi-point method at a temperature of -196 ° C.
- the BET specific surface area is obtained from the adsorption isotherm in the range of relative pressure of 0.01 ⁇ P / P 0 ⁇ 0.05.
- the pore volume is determined from the volume of adsorption N 2 at a relative pressure of 0.96.
- the average pore diameter of the conductive material is not particularly limited, but is preferably 50 nm or less, and particularly preferably 30 nm or less. If the average pore diameter of the conductive material is within these ranges, sufficient electrons can be transferred to the active material of the positive electrode containing sulfur arranged inside the pores, which is located away from the pore wall. Can be supplied.
- the value of the average pore diameter of the conductive material can be calculated by nitrogen adsorption / desorption measurement in the same manner as in the case of obtaining the values of the BET specific surface area and the pore volume.
- the average particle size (primary particle size) when the conductive material is in the form of particles is not particularly limited, but is preferably 0.05 to 50 ⁇ m, more preferably 0.1 to 20 ⁇ m. , 0.5 to 10 ⁇ m, more preferably.
- the conductive auxiliary agent described above is similarly adopted.
- the positive electrode material according to this embodiment contains a conductive material having pores, a solid electrolyte, and a positive electrode active material containing sulfur, but at least a part of the solid electrolyte and at least a part of the positive electrode activity. It is characterized in that the substances are arranged on the inner surface of the pores of the conductive material so as to be in contact with each other.
- FIG. 3A is a schematic cross-sectional view of the positive electrode material 100'in the prior art.
- FIG. 3B is a schematic cross-sectional view of the positive electrode material 100 according to the embodiment of the present invention.
- the carbon material (for example, activated carbon) 110 which is a conductive material, has a large number of pores 110a.
- Sulfur 120 which is a positive electrode active material, is filled and arranged inside the pores 110a.
- the positive electrode active material (sulfur) 120 is also arranged on the surface of the carbon material (activated carbon) 110.
- the positive electrode material 100' accordinging to the prior art shown in FIG.
- the solid electrolyte 130 is arranged only on the surface of the carbon material (activated carbon) 110. There is. On the other hand, in the positive electrode material 100 according to the embodiment of the present invention shown in FIG. 3B, the solid electrolyte (for example, Li 6 PS 5 Cl, which is a sulfide solid electrolyte) 130 is only on the surface of the carbon material (activated carbon) 110. Instead, it is also arranged on the inner surface of the pores of the carbon material (activated carbon) 110.
- a continuous phase made of the positive electrode active material (sulfur) 120 is filled inside the pores 110a, and the solid electrolyte 130 is arranged as a dispersed phase in the continuous phase.
- the solid electrolyte arranged on the inner surface of the pores and at least a part of the positive electrode active material (sulfur) similarly arranged inside the pores are in contact with each other.
- elemental mapping derived from each material is performed using energy dispersive X-ray spectroscopy (EDX) on an observation image of a cross section of a conductive material using a transmission electron microscope (TEM), and the obtained element map and all elements are used. It is possible to confirm the arrangement form of each material by using the count number of the element derived from each material as an index with respect to the count number (see Examples described later). For example, if the solid electrolyte contains a phosphorus atom and / or a boron atom and the phosphorus atom and / or the boron atom cannot be derived from another material, the above element for phosphorus and / or boron.
- EDX energy dispersive X-ray spectroscopy
- TEM transmission electron microscope
- the value of the ratio is preferably 0.15 or more, more preferably 0.20 or more, still more preferably 0.26 or more, and particularly preferably 0.35 or more.
- the preferable upper limit of the value of the ratio is not particularly limited, but as an example of the preferable upper limit, it is 0.50 or less, and more preferably 0.45 or less.
- the capacity characteristics and the charge / discharge rate characteristics are improved in an electric device such as an all-solid-state lithium ion secondary battery using a positive electrode active material containing sulfur. Is possible. Although the mechanism by which such an excellent effect is obtained by the configuration according to this embodiment has not been completely clarified, the following mechanism has been presumed. That is, in order for the charge / discharge reaction to proceed in the positive electrode active material containing sulfur, it is necessary that charge carriers such as electrons and lithium ions proceed smoothly in and out of the surface of the positive electrode active material.
- the positive electrode active material (sulfur) 120 when the positive electrode active material (sulfur) 120 is held inside the pores 110a of the conductive material such as the carbon material 110 as in the positive electrode material 100'shown in FIG. 3A, it is located deep in the pores 110a. On the surface of the positive electrode active material (sulfur) 120, electrons can flow in and out smoothly to some extent through the conductive material. However, since the solid electrolyte 130 is not arranged inside the pores 110a, charge carriers such as lithium ions smoothly move in and out on the surface of the positive electrode active material (sulfur) 120 located deep inside the pores 110a. do not. As a result, in the positive electrode active material (sulfur) 120 located deep in the pores 110a, the charge / discharge reaction does not proceed sufficiently, and it is considered that problems such as deterioration of capacity characteristics and charge / discharge rate characteristics occur.
- the solid electrolyte 130 is held together with the positive electrode active material (sulfur) 120 on the inner surface of the pores 110a of the conductive material such as the carbon material 110 as in the positive electrode material 100 shown in FIG. 3B, the pores.
- the positive electrode active material (sulfur) 120 located deep in 110a not only the inflow and outflow of electrons through the conductive material but also the inflow and outflow of charge carriers via the solid electrolyte 130 can proceed smoothly.
- the method for producing the positive electrode material according to the present embodiment having the above configuration is not particularly limited, but an example of the production method will be briefly described.
- a solution in which a solid electrolyte is dissolved in an organic solvent is prepared, and a conductive material is dispersed therein to obtain a dispersion liquid.
- heat treatment is performed at a temperature of about 150 to 250 ° C. for about 1 to 5 hours.
- a conductive material in which the pores of the conductive material are impregnated with the solid electrolyte can be obtained.
- the obtained conductive material is mixed with the positive electrode active material in a dry manner, and further heat-treated under the same conditions as described above.
- the positive electrode active material is melted and permeates into the pores of the conductive material, and a composite material in which the positive electrode active material is arranged (filled) together with the solid electrolyte is obtained inside the pores of the conductive material.
- the composite material thus obtained may be used as it is as a positive electrode material, but a solid electrolyte may be further added to the composite material and mixed, and if necessary, treated with an apparatus such as a ball mill to obtain a positive electrode. It is preferable to obtain the material.
- the content of the positive electrode active material in the positive electrode active material layer is not particularly limited, but is preferably in the range of, for example, 35 to 99% by mass, and preferably in the range of 40 to 90% by mass. More preferred. The value of this content shall be calculated based on the mass of only the positive electrode active material excluding the conductive material and the solid electrolyte.
- the positive electrode active material layer may further contain a conductive auxiliary agent (one that does not retain the positive electrode active material or the solid electrolyte inside the pores) and / or a binder, and the specific form and preferable form thereof may be further contained.
- a conductive auxiliary agent one that does not retain the positive electrode active material or the solid electrolyte inside the pores
- the positive electrode active material layer preferably further contains a solid electrolyte, and particularly preferably a sulfide solid electrolyte.
- the specific form and preferable form of the solid electrolyte such as the sulfide solid electrolyte
- those described in the above-mentioned column of the negative electrode active material layer can be similarly adopted.
- the material constituting the current collector plates (25, 27) is not particularly limited, and known highly conductive materials conventionally used as current collector plates for secondary batteries can be used.
- As the constituent material of the current collector plate for example, metal materials such as aluminum, copper, titanium, nickel, stainless steel (SUS), and alloys thereof are preferable. From the viewpoint of light weight, corrosion resistance, and high conductivity, aluminum and copper are more preferable, and aluminum is particularly preferable.
- the same material may be used for the positive electrode current collector plate 27 and the negative electrode current collector plate 25, or different materials may be used.
- the current collector (11', 11 ") and the current collector plate (25, 27) may be electrically connected to each other via a positive electrode lead or a negative electrode lead.
- a material used in a known lithium ion secondary battery can be similarly adopted.
- the portion taken out from the exterior may come into contact with peripheral devices, wiring, etc. and leak electricity. It is preferable to cover the product with a heat-resistant insulating tube or the like so as not to affect the product (for example, automobile parts, particularly electronic devices, etc.).
- Battery exterior material As the battery exterior material, a known metal can case can be used, and a bag-shaped case using a laminated film 29 containing aluminum, which can cover the power generation element as shown in FIGS. 1 and 2, is used. Can be done.
- the laminated film for example, a laminated film having a three-layer structure in which PP, aluminum, and nylon are laminated in this order can be used, but the laminated film is not limited thereto.
- a laminated film is desirable from the viewpoint of high output, excellent cooling performance, and suitable use for batteries for large equipment for EVs and HEVs. Further, since the group pressure applied to the power generation element from the outside can be easily adjusted, a laminated film containing aluminum is more preferable for the exterior body.
- the laminated battery according to this embodiment has a configuration in which a plurality of single battery layers are connected in parallel, so that it has a high capacity and excellent cycle durability. Therefore, the laminated battery according to this embodiment is suitably used as a driving power source for EVs and HEVs.
- the present invention is not limited to the configuration described in the above-described embodiment, and may be appropriately modified based on the description of the claims. It is possible.
- a positive electrode active material layer electrically bonded to one surface of the current collector and an electrical charge to the opposite surface of the current collector are used.
- a bipolar type battery including a bipolar type electrode having a negative electrode active material layer coupled to the above can also be mentioned.
- the secondary battery according to this embodiment does not have to be an all-solid-state type. That is, the solid electrolyte layer may further contain a conventionally known liquid electrolyte (electrolyte solution).
- the amount of the liquid electrolyte (electrolyte solution) that can be contained in the solid electrolyte layer is not particularly limited, but the shape of the solid electrolyte layer formed by the solid electrolyte is maintained and the liquid electrolyte (electrolyte solution) does not leak. Is preferably the amount of.
- the liquid electrolyte (electrolyte solution) that can be used has a form in which a lithium salt is dissolved in an organic solvent.
- organic solvent used include dimethyl carbonate (DMC), diethyl carbonate (DEC), dipropyl carbonate (DPC), ethyl methyl carbonate (EMC), methyl propionate (MP), methyl acetate (MA), and methyl formate.
- the organic solvent is preferably a chain carbonate, more preferably diethyl carbonate (DEC), ethyl methyl carbonate (EMC) and dimethyl carbonate (DMC) from the viewpoint of further improving the quick charge property and the output property. It is at least one selected from the group consisting of, and more preferably selected from ethyl methyl carbonate (EMC) and dimethyl carbonate (DMC).
- Lithium salts include Li (FSO 2 ) 2 N (lithium bis (fluorosulfonyl) imide; LiFSI), Li (C 2 F 5 SO 2 ) 2 N, LiPF 6 , LiBF 4 , LiClO 4 , LiAsF 6 , LiCF 3 SO 3 and the like can be mentioned.
- the lithium salt is preferably Li (FSO 2 ) 2N ( LiFSI) from the viewpoint of battery output and charge / discharge cycle characteristics.
- the liquid electrolyte may further contain additives other than the above-mentioned components.
- additives include, for example, ethylene carbonate, vinylene carbonate, methylvinylene carbonate, dimethylvinylene carbonate, phenylvinylene carbonate, diphenylvinylene carbonate, ethylvinylene carbonate, diethylvinylene carbonate, vinylethylene carbonate, 1,2-.
- An assembled battery is configured by connecting a plurality of batteries. More specifically, it is composed of serialization, parallelization, or both by using at least two or more. By serializing and parallelizing, it becomes possible to freely adjust the capacity and voltage.
- a small assembled battery that can be attached and detached by connecting multiple batteries in series or in parallel. Then, by connecting a plurality of small detachable batteries in series or in parallel, a large capacity and a large capacity suitable for a vehicle drive power source or an auxiliary power source that require a high volume energy density and a high volume output density. It is also possible to form an assembled battery having an output (battery module, battery pack, etc.). How many batteries are connected to make an assembled battery, and how many stages of small assembled batteries are stacked to make a large-capacity assembled battery depends on the battery capacity of the vehicle (electric vehicle) to be mounted. It may be decided according to the output.
- a battery or a combined battery consisting of a plurality of these can be mounted on a vehicle.
- a plug-in hybrid electric vehicle having a long EV mileage and an electric vehicle having a long one-charge mileage can be configured by mounting such a battery.
- hybrid vehicles, fuel cell vehicles, electric vehicles (all four-wheeled vehicles (passenger cars, trucks, commercial vehicles such as buses, light vehicles, etc.)) can be used as batteries or a combination of multiple batteries. This is because it becomes a highly reliable automobile with a long life by using it for two-wheeled vehicles (including motorcycles) and three-wheeled vehicles.
- the application is not limited to automobiles, and can be applied to various power sources of other vehicles, for example, moving objects such as trains, and power supplies for mounting such as uninterruptible power supplies. It is also possible to use it as.
- Example 1 Preparation of solid electrolyte impregnated carbon
- cryoplasma focused ion beam processing device Helios G4 PFIB CXe manufactured by Thermo Scientific Co., Ltd., accelerated voltage: 30 kV
- the powder particles of the contained positive electrode material were exfoliated to a thickness of about 100 nm.
- the stripped observation sample is transported to a TEM device (JEOL, multifunction analysis transmission electron microscope JEM-F200, acceleration voltage: 200 kV) without exposure to the atmosphere to check the microstructure and EDX attached to the TEM.
- JEOL multifunction analysis transmission electron microscope JEM-F200, acceleration voltage: 200 kV
- the element map data of the part corresponding to the inside of the particle was acquired by the device (energy dispersive X-ray spectroscopic analyzer, JEOL Dual SDD, acceleration voltage: 200 kV) (EDX mapping characteristic X-ray measurement energy band: 0 to 5 keV). ). From the obtained element map data, the counts of all the elements contained in the particles and the counts of the elements (phosphorus; P) derived only from the solid electrolyte were obtained. As a result, the ratio of the count number of the element (P) derived only from the solid electrolyte to the count number of all elements was 0.35.
- the solid electrolyte is also arranged inside the pores of the conductive material (carbon) together with the sulfur which is the positive electrode active material.
- the observation image of the powder particles of the sulfur-containing positive electrode material thus obtained by a scanning electron microscope (SEM) is shown in FIG. 4A, and the phosphorus (P) element in the observation image of the cross section of the conductive material by TEM-EDX.
- the elemental map for is shown in FIG. 4B.
- test cell all-solid-state lithium-ion secondary battery
- the battery was manufactured in a glove box having an argon atmosphere with a dew point of ⁇ 68 ° C. or lower.
- a positive electrode active material layer having a diameter of 10 mm and a thickness of about 0.06 mm was formed on one side of the solid electrolyte layer by inserting (also serving as a positive electrode current collector) and pressing at a pressure of 300 MPa for 3 minutes.
- a cylindrical convex punch (which also serves as a negative electrode current collector) on the lower side was extracted, and a lithium foil (manufactured by Niraco, 0.20 mm thick) punched to a diameter of 8 mm and an indium foil punched to a diameter of 9 mm (manufactured by Niraco, 0.20 mm) were punched out as a negative electrode.
- Niraco Co., Ltd., thickness 0.30 mm is layered, and the indium foil is inserted from the underside of the cylindrical tube jig so that it is located on the side of the solid electrolyte layer, and the cylindrical convex punch is inserted again, and the pressure is 75 MPa.
- a lithium-indium negative electrode was formed by pressing for 3 minutes.
- test cell all-solid-state lithium ion battery in which a negative electrode current collector (punch), a lithium-indium negative electrode, a solid electrolyte layer, a positive electrode active material layer, and a positive electrode current collector (punch) are laminated in this order.
- a negative electrode current collector punch
- a lithium-indium negative electrode a lithium-indium negative electrode
- a solid electrolyte layer a positive electrode active material layer
- positive electrode current collector punch
- Example 2 In the preparation of the solid electrolyte-impregnated carbon, an all-solid-state lithium-ion secondary battery was produced by the same method as in Example 1 described above, except that the conditions of the vacuum heat treatment after removing the ethanol under reduced pressure were changed to 230 ° C. for 3 hours. bottom.
- the sulfur-containing positive electrode material prepared in this example TEM-EDX was used, and the ratio of the count number of the element (P) derived only from the solid electrolyte to the count number of all elements was the same as above. Was calculated to be 0.26. From this, it was confirmed that in the sulfur-containing positive electrode material according to the present embodiment, the solid electrolyte is also arranged inside the pores of the conductive material (carbon) together with the sulfur which is the positive electrode active material.
- Example 3 In the thermal impregnation of sulfur, sulfur is added to the solid electrolyte impregnated carbon and mixed sufficiently in a Menou dairy pot, and then the mixed powder is vacuum-sealed in a quartz tube at 1 Pa or less in place of a closed pressure-resistant autoclave container for 3 hours at 170 ° C.
- An all-solid-state lithium-ion secondary battery was produced by the same method as in Example 1 described above, except that the sulfur was melted by heating and the solid electrolyte-impregnated carbon was impregnated with the sulfur.
- the sulfur-containing positive electrode material prepared in this example TEM-EDX was used, and the ratio of the count number of the element (P) derived only from the solid electrolyte to the count number of all elements was the same as above. Was calculated to be 0.20. From this, it was confirmed that in the sulfur-containing positive electrode material according to the present embodiment, the solid electrolyte is also arranged inside the pores of the conductive material (carbon) together with the sulfur which is the positive electrode active material.
- Example 4 An all-solid-state lithium-ion secondary battery was produced by the same method as in Example 1 described above, except that the mixing condition by the planetary ball mill was changed to 3 hours at 540 rpm in the preparation of the sulfur-containing positive electrode material.
- the sulfur-containing positive electrode material prepared in this example TEM-EDX was used, and the ratio of the count number of the element (P) derived only from the solid electrolyte to the count number of all elements was the same as above. Was calculated to be 0.12. From this, it was confirmed that in the sulfur-containing positive electrode material according to the present embodiment, the solid electrolyte is also arranged inside the pores of the conductive material (carbon) together with the sulfur which is the positive electrode active material.
- an all-solid-state lithium-ion secondary battery was produced by the same method as in Example 1 described above.
- test cell The capacity characteristics and charge / discharge rate characteristics of each of the above comparative examples and the test cells prepared in each example were evaluated by the following methods. All of the following measurements were performed using a charge / discharge test device (HJ-SD8 manufactured by Hokuto Denko Co., Ltd.) in a constant temperature and constant temperature bath set at 25 ° C.
- HJ-SD8 manufactured by Hokuto Denko Co., Ltd.
- a test cell is installed in a constant temperature bath, and after the cell temperature becomes constant, discharge is performed at a current density of 0.2 mA / cm 2 to a cell voltage of 0.5 V as cell conditioning, and then the same current density is used.
- the 2.5 V constant current constant voltage charge was set to a cutoff current of 0.01 mA / cm 2 .
- the capacity value (mAh / g) per mass of the positive electrode active material was calculated from the value of the charge / discharge capacity obtained after repeating this conditioning charge / discharge cycle 10 times and the mass of the positive electrode active material contained in the positive electrode. The results are shown in Table 1 below. Further, FIG. 6 shows a charge / discharge curve for the test cell (all-solid-state lithium-ion secondary battery) produced in Example 2.
- the charge rate characteristic is 0 with respect to the charge capacity value obtained by constant current charging at 0.05 C with a cut-off voltage of 2.5 V after full discharge by 0.05 C discharge with a cut-off voltage of 0.5 V.
- the percentage (charge rate maintenance rate) of the charge capacity value obtained by constant current charging at .5C was calculated. The results are shown in Table 1 below.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- General Physics & Mathematics (AREA)
- Inorganic Chemistry (AREA)
- Secondary Cells (AREA)
- Battery Electrode And Active Subsutance (AREA)
Abstract
Description
集電体は、電極活物質層からの電子の移動を媒介する機能を有する。集電体を構成する材料に特に制限はない。集電体の構成材料としては、例えば、金属や、導電性を有する樹脂が採用されうる。
図1および図2に示す実施形態に係る積層型電池において、負極活物質層13は、負極活物質を含む。負極活物質の種類としては、特に制限されないが、炭素材料、金属酸化物および金属活物質が挙げられる。炭素材料としては、例えば、天然黒鉛、人造黒鉛、メソカーボンマイクロビーズ(MCMB)、高配向性グラファイト(HOPG)、ハードカーボン、ソフトカーボン等が挙げられる。また、金属酸化物としては、例えば、Nb2O5、Li4Ti5O12等が挙げられる。さらに、ケイ素系負極活物質やスズ系負極活物質が用いられてもよい。ここで、ケイ素およびスズは第14族元素に属し、非水電解質二次電池の容量を大きく向上させうる負極活物質であることが知られている。これらの単体は単位体積(質量)あたり多数の電荷担体(リチウムイオン等)を吸蔵および放出しうることから、高容量の負極活物質となる。ここで、ケイ素系負極活物質としては、Si単体を用いることが好ましい。また同様に、Si相とケイ素酸化物相との2相に不均化されたSiOx(0.3≦x≦1.6)などのケイ素酸化物を用いることも好ましい。この際、xの範囲は0.5≦x≦1.5であることがより好ましく、0.7≦x≦1.2であることがさらに好ましい。さらには、ケイ素を含有する合金(ケイ素含有合金系負極活物質)が用いられてもよい。一方、スズ元素を含む負極活物質(スズ系負極活物質)としては、Sn単体、スズ合金(Cu−Sn合金、Co−Sn合金)、アモルファススズ酸化物、スズケイ素酸化物等が挙げられる。このうち、アモルファススズ酸化物としてはSnB0.4P0.6O3.1が例示される。また、スズケイ素酸化物としてはSnSiO3が例示される。また、負極活物質として、リチウムを含有する金属を用いてもよい。このような負極活物質は、リチウムを含有する活物質であれば特に限定されず、金属リチウムのほか、リチウム含有合金が挙げられる。リチウム含有合金としては、例えば、Liと、In、Al、SiおよびSnの少なくとも1種との合金が挙げられる。場合によっては、2種以上の負極活物質が併用されてもよい。なお、上記以外の負極活物質が用いられてもよいことは勿論である。負極活物質は、金属リチウム、ケイ素系負極活物質またはスズ系負極活物質を含むことが好ましく、金属リチウムを含むことが特に好ましい。
図1および図2に示す実施形態に係る積層型電池において、固体電解質層は、上述した正極活物質層と負極活物質層との間に介在し、固体電解質を必須に含有する層である。
図1および図2に示す実施形態に係る積層型電池において、正極活物質層は、本発明の一形態に係る電気デバイス用正極材料を含む。当該電気デバイス用正極材料は、細孔を有する導電材料と、固体電解質と、硫黄を含む正極活物質とを含む。
硫黄を含む正極活物質の種類としては、特に制限されないが、硫黄単体(S)のほか、有機硫黄化合物または無機硫黄化合物の粒子または薄膜が挙げられ、硫黄の酸化還元反応を利用して、充電時にリチウムイオンを放出し、放電時にリチウムイオンを吸蔵することができる物質であればよい。有機硫黄化合物としては、ジスルフィド化合物、国際公開第2010/044437号パンフレットに記載の化合物に代表される硫黄変性ポリアクリロニトリル、硫黄変性ポリイソプレン、ルベアン酸(ジチオオキサミド)、ポリ硫化カーボン等が挙げられる。なかでも、ジスルフィド化合物および硫黄変性ポリアクリロニトリル、およびルベアン酸が好ましく、特に好ましくは硫黄変性ポリアクリロニトリルである。ジスルフィド化合物としては、ジチオビウレア誘導体、チオウレア基、チオイソシアネート、またはチオアミド基を有するものがより好ましい。ここで、硫黄変性ポリアクリロニトリルとは、硫黄粉末とポリアクリロニトリルとを混合し、不活性ガス下もしくは減圧下で加熱することによって得られる、硫黄原子を含む変性されたポリアクリロニトリルである。その推定構造は、例えばChem. Mater. 2011,23,5024−5028に示されているように、ポリアクリロニトリルが閉環して多環状になるとともに、Sの少なくとも一部はCと結合している構造である。この文献に記載されている化合物はラマンスペクトルにおいて、1330cm−1と1560cm−1付近に強いピークシグナルがあり、さらに、307cm−1、379cm−1、472cm−1、929cm−1付近にピークが存在する。一方、無機硫黄化合物は安定性に優れることから好ましく、具体的には、硫黄単体(S)、TiS2、TiS3、TiS4、NiS、NiS2、CuS、FeS2、Li2S、MoS2、MoS3、MnS、MnS2、CoS、CoS2等が挙げられる。なかでも、S、S−カーボンコンポジット、TiS2、TiS3、TiS4、FeS2およびMoS2が好ましく、硫黄単体(S)、TiS2およびFeS2がより好ましく、高容量であるという観点からは硫黄単体(S)が特に好ましい。なお、硫黄単体(S)としては、S8構造を有するα硫黄、β硫黄、またはγ硫黄が用いられうる。
本形態に係る正極材料は固体電解質を必須に含む。本形態に係る正極材料に含まれる固体電解質の具体的な形態について特に制限はなく、負極活物質層の欄において例示した固体電解質およびその好ましい形態が同様に採用されうる。場合によっては、上述した固体電解質以外の固体電解質が併用されてもよい。
本形態に係る正極材料は細孔を有する導電材料を必須に含む。本形態に係る正極材料に含まれる導電材料の具体的な形態については、細孔を有するものであれば特に制限はなく、従来公知の材料が適宜採用されうる。導電性に優れ、加工しやすく、所望の細孔分布の設計が容易であるという観点からは、細孔を有する導電材料は炭素材料であることが好ましい。
集電板(25、27)を構成する材料は、特に制限されず、二次電池用の集電板として従来用いられている公知の高導電性材料が用いられうる。集電板の構成材料としては、例えば、アルミニウム、銅、チタン、ニッケル、ステンレス鋼(SUS)、これらの合金等の金属材料が好ましい。軽量、耐食性、高導電性の観点から、より好ましくはアルミニウム、銅であり、特に好ましくはアルミニウムである。なお、正極集電板27と負極集電板25とでは、同一の材料が用いられてもよいし、異なる材料が用いられてもよい。
また、図示は省略するが、集電体(11’、11”)と集電板(25、27)との間を正極リードや負極リードを介して電気的に接続してもよい。正極および負極リードの構成材料としては、公知のリチウムイオン二次電池において用いられる材料が同様に採用されうる。なお、外装から取り出された部分は、周辺機器や配線などに接触して漏電したりして製品(例えば、自動車部品、特に電子機器等)に影響を与えないように、耐熱絶縁性の熱収縮チューブなどにより被覆することが好ましい。
電池外装材としては、公知の金属缶ケースを用いることができるほか、図1および図2に示すように発電要素を覆うことができる、アルミニウムを含むラミネートフィルム29を用いた袋状のケースが用いられうる。該ラミネートフィルムには、例えば、PP、アルミニウム、ナイロンをこの順に積層してなる3層構造のラミネートフィルム等を用いることができるが、これらに何ら制限されるものではない。高出力化や冷却性能に優れ、EV、HEV用の大型機器用電池に好適に利用することができるという観点から、ラミネートフィルムが望ましい。また、外部から掛かる発電要素への群圧を容易に調整することができることから、外装体はアルミニウムを含むラミネートフィルムがより好ましい。
組電池は、電池を複数個接続して構成した物である。詳しくは少なくとも2つ以上用いて、直列化あるいは並列化あるいはその両方で構成されるものである。直列、並列化することで容量および電圧を自由に調節することが可能になる。
電池またはこれらを複数個組み合わせてなる組電池を車両に搭載することができる。本発明では、長期信頼性に優れた高寿命の電池を構成できることから、こうした電池を搭載するとEV走行距離の長いプラグインハイブリッド電気自動車や、一充電走行距離の長い電気自動車を構成できる。電池またはこれらを複数個組み合わせてなる組電池を、例えば、自動車ならばハイブリッド車、燃料電池車、電気自動車(いずれも四輪車(乗用車、トラック、バスなどの商用車、軽自動車など)のほか、二輪車(バイク)や三輪車を含む)に用いることにより高寿命で信頼性の高い自動車となるからである。ただし、用途が自動車に限定されるわけではなく、例えば、他の車両、例えば、電車などの移動体の各種電源であっても適用は可能であるし、無停電電源装置などの載置用電源として利用することも可能である。
[実施例1]
(固体電解質含浸カーボンの調製)
露点−68℃以下のアルゴン雰囲気のグローブボックス内で、硫化物固体電解質(Ampcera社製、Li6PS5Cl)0.500gを100mLの超脱水エタノール(富士フイルム和光純薬株式会社製)に加え、溶液が透明になるまで撹拌して固体電解質をエタノールに溶解させた。得られた固体電解質エタノール溶液にカーボン(関西熱化学株式会社製、活性炭、MSC−30)1.00gを加え、よく撹拌して溶液中にカーボンを十分に分散させた。このカーボン分散液が入った容器を真空装置に接続し、マグネティックスターラーにより容器中のカーボン分散液を撹拌しながら油回転ポンプにより容器中を1Pa以下の減圧状態にした。減圧下では溶媒であるエタノールが揮発するため、時間の経過とともにエタノールが除去され、固体電解質を含浸したカーボンが容器内に残存した。このようにしてエタノールを減圧除去した後に減圧下で180℃に加熱し、3時間熱処理を行うことにより固体電解質含浸カーボンを調製した。
露点−68℃以下のアルゴン雰囲気のグローブボックス内で、上記で調製した固体電解質含浸カーボン0.750gに硫黄(Aldrich社製)2.50gを加えてメノウ乳鉢で十分に混合した後、混合粉末を密閉耐圧オートクレーブ容器に入れて170℃で3時間加熱することにより硫黄を溶融させて、硫黄を固体電解質含浸カーボンに含浸させた。これにより硫黄/固体電解質/カーボン複合材を調製した。
露点−68℃以下のアルゴン雰囲気のグローブボックス内で、5mm径のジルコニアボール40gと、上記で調製した硫黄/固体電解質含浸カーボン0.130gと、固体電解質(Ampcera社製、Li6PS5Cl)0.070gとを容量45mLのジルコニア製容器に入れ、遊星ボールミル(フリッチュ社製、Premium line P−7)により370rpmで6時間処理することにより、硫黄含有正極材料の粉末を得た。硫黄含有正極材料の組成は硫黄:固体電解質:カーボン=50:40:10とした。
硫黄含有正極材料の粉末を構成する導電材料(カーボン)の細孔の内部に含まれる固体電解質の含有量を、以下の手法により定量した。
電池の作製は、露点−68℃以下のアルゴン雰囲気のグローブボックス内で行った。
固体電解質含浸カーボンの調製において、エタノール減圧除去後の減圧熱処理の条件を230℃で3時間に変更したこと以外は、上述した実施例1と同様の手法により、全固体リチウムイオン二次電池を作製した。
硫黄の熱含浸において、固体電解質含浸カーボンに硫黄を加えてメノウ乳鉢で十分に混合した後、混合粉末を密閉耐圧オートクレーブ容器に代えて石英管内に1Pa以下で減圧封管して170℃で3時間加熱することにより硫黄を溶融させて、硫黄を固体電解質含浸カーボンに含浸させたこと以外は、上述した実施例1と同様の手法により、全固体リチウムイオン二次電池を作製した。
硫黄含有正極材料の調製において、遊星ボールミルによる混合条件を540rpmで3時間に変更したこと以外は、上述した実施例1と同様の手法により、全固体リチウムイオン二次電池を作製した。
固体電解質含浸カーボンの調製および硫黄の熱含浸は行わず、露点−68℃以下のアルゴン雰囲気のグローブボックス内で、5mm径のジルコニアボール40gと、硫黄0.100gと、固体電解質0.080gと、カーボン0.020gとを容量45mLのジルコニア製容器に入れ、遊星ボールミルにより370rpmで6時間処理することにより、硫黄含有正極材料の粉末を得た。これらのこと以外は、上述した実施例1と同様の手法により、全固体リチウムイオン二次電池を作製した。
固体電解質含浸カーボンの調製と硫黄の熱含浸は行わず、露点−68℃以下のアルゴン雰囲気のグローブボックス内で、5mm径のジルコニアボール40gと、硫黄0.100gと、固体電解質0.080gと、カーボン0.020gとを容量45mLのジルコニア製容器に入れ、遊星ボールミルにより370rpmで6時間処理した後、硫黄/固体電解質/カーボン混合粉末を密閉耐圧オートクレーブ容器に入れて170℃で3時間加熱することにより、硫黄含有正極材料の粉末を得た。これらのこと以外は、上述した実施例1と同様の手法により、全固体リチウムイオン二次電池を作製した。
固体電解質含浸カーボンの調製は行わず、0.500gのカーボンに2.50gの硫黄を加えてメノウ乳鉢で十分に混合した後、混合粉末を密閉耐圧オートクレーブ容器に入れて170℃で3時間加熱することにより硫黄を溶融させて、硫黄をカーボンに含浸させた。これにより硫黄含浸カーボンを得た。
上記の各比較例および各実施例で作製した試験用セルについて、下記の手法により容量特性および充放電レート特性の評価を行った。なお、以下の測定はすべて、充放電試験装置(北斗電工株式会社製、HJ−SD8)を用い、25℃に設定した定温恒温槽中で行った。
恒温槽内に試験用セルを設置し、セル温度が一定になった後、セルコンディショニングとして、0.2mA/cm2の電流密度でセル電圧0.5Vまで放電を行い、それに続いて同じ電流密度で2.5V定電流定電圧充電をカットオフ電流0.01mA/cm2に設定して行った。このコンディショニング充放電サイクルを10回繰り返した後に得られた充放電容量の値と、正極に含まれる正極活物質の質量とから正極活物質の質量あたりの容量値(mAh/g)を算出した。結果を下記の表1に示す。また、実施例2において作製された試験用セル(全固体リチウムイオン二次電池)についての充放電曲線を図6に示す。
放電レート特性については、0.05C−2.5V定電流定電圧充電でカットオフ電流0.01Cの条件で満充電した後に、カットオフ電圧0.5Vで0.05Cでの放電により得られた放電容量値に対する、0.5Cでの放電により得られた放電容量値の百分率(放電レート維持率)を算出した。結果を下記の表1に示す。
11’ 負極集電体、
11” 正極集電体、
13 負極活物質層、
15 正極活物質層、
17 固体電解質層、
19 単電池層、
21 発電要素、
25 負極集電板、
27 正極集電板、
29 ラミネートフィルム、
100,100’ 正極材料、
110 炭素材料(活性炭)、
110a 細孔、
120 正極活物質(硫黄)、
130 固体電解質。
Claims (12)
- 細孔を有する導電材料と、固体電解質と、硫黄を含む正極活物質と、を含み、
少なくとも一部の前記固体電解質と少なくとも一部の前記正極活物質とが、互いに接するように前記細孔の内部表面に配置されている、電気デバイス用正極材料。 - 前記正極活物質からなる連続相が前記細孔の内部に充填されており、前記固体電解質が前記連続相中に分散相として配置されている、請求項1に記載の電気デバイス用正極材料。
- TEM−EDXによる前記導電材料の断面の観察画像において、全元素のカウント数に対する前記固体電解質のみに由来する元素のカウント数の比の値が0.10以上である、請求項1または2に記載の電気デバイス用正極材料。
- 前記導電材料が炭素材料である、請求項1~3のいずれか1項に記載の電気デバイス用正極材料。
- 前記導電材料の細孔容積が1.0mL/g以上である、請求項1~4のいずれか1項に記載の電気デバイス用正極材料。
- 前記導電材料の平均細孔径が50nm以下である、請求項1~5のいずれか1項に記載の電気デバイス用正極材料。
- 前記固体電解質が硫化物固体電解質である、請求項1~6のいずれか1項に記載の電気デバイス用正極材料。
- 前記固体電解質が、アルカリ金属原子と、リン原子および/またはホウ素原子とを含有する、請求項1~7のいずれか1項に記載の電気デバイス用正極材料。
- 前記アルカリ金属がリチウムである、請求項8に記載の電気デバイス用正極材料。
- 請求項1~9のいずれか1項に記載の電気デバイス用正極材料を含む、電気デバイス用正極。
- 請求項10に記載の電気デバイス用正極を含む、電気デバイス。
- 全固体リチウムイオン二次電池である、請求項11に記載の電気デバイス。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US18/033,541 US20230395788A1 (en) | 2020-10-26 | 2020-10-26 | Positive Electrode Material for Electric Device, Positive Electrode for Electric Device and Electric Device Using Positive Electrode Material for Electric Device |
| CN202080106687.0A CN116472629A (zh) | 2020-10-26 | 2020-10-26 | 电气设备用正极材料以及使用了其的电气设备用正极和电气设备 |
| PCT/IB2020/000884 WO2022090757A1 (ja) | 2020-10-26 | 2020-10-26 | 電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス |
| JP2022558362A JP7493054B2 (ja) | 2020-10-26 | 2020-10-26 | 電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス |
| EP20959660.0A EP4235856A1 (en) | 2020-10-26 | 2020-10-26 | Positive electrode material for electrical devices, positive electrode for electrical devices using same, and electrical device |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/IB2020/000884 WO2022090757A1 (ja) | 2020-10-26 | 2020-10-26 | 電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022090757A1 true WO2022090757A1 (ja) | 2022-05-05 |
Family
ID=81383087
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2020/000884 WO2022090757A1 (ja) | 2020-10-26 | 2020-10-26 | 電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20230395788A1 (ja) |
| EP (1) | EP4235856A1 (ja) |
| JP (1) | JP7493054B2 (ja) |
| CN (1) | CN116472629A (ja) |
| WO (1) | WO2022090757A1 (ja) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010044437A1 (ja) | 2008-10-17 | 2010-04-22 | 独立行政法人産業技術総合研究所 | 硫黄変性ポリアクリロニトリル、その製造方法、及びその用途 |
| JP2012041220A (ja) * | 2010-08-17 | 2012-03-01 | Fukuoka Univ | 硫黄複合化活性炭およびその製造方法 |
| WO2012102037A1 (ja) | 2011-01-27 | 2012-08-02 | 出光興産株式会社 | アルカリ金属硫化物と導電剤の複合材料 |
| JP2015088232A (ja) * | 2013-10-28 | 2015-05-07 | ナガセケムテックス株式会社 | 薄膜硫黄被覆活性炭の製造方法、薄膜硫黄被覆活性炭、正極合材及び全固体型リチウム硫黄電池 |
| JP2016127005A (ja) * | 2014-12-31 | 2016-07-11 | 現代自動車株式会社Hyundai Motor Company | 全固体リチウム電池の陽極及びこれを含む二次電池 |
| JP2019040752A (ja) * | 2017-08-25 | 2019-03-14 | 株式会社サムスン日本研究所 | 全固体型二次電池 |
| WO2019078130A1 (ja) * | 2017-10-19 | 2019-04-25 | 三菱瓦斯化学株式会社 | 全固体電池の製造方法 |
| JP2019179604A (ja) * | 2018-03-30 | 2019-10-17 | 東京電力ホールディングス株式会社 | リチウム硫黄固体電池 |
| JP2019179603A (ja) * | 2018-03-30 | 2019-10-17 | 東京電力ホールディングス株式会社 | リチウム硫黄固体電池、電池用正極シート、電池用負極シート、筒状電池、集電体付き筒状電池、積層電池 |
-
2020
- 2020-10-26 EP EP20959660.0A patent/EP4235856A1/en active Pending
- 2020-10-26 JP JP2022558362A patent/JP7493054B2/ja active Active
- 2020-10-26 WO PCT/IB2020/000884 patent/WO2022090757A1/ja active Application Filing
- 2020-10-26 CN CN202080106687.0A patent/CN116472629A/zh active Pending
- 2020-10-26 US US18/033,541 patent/US20230395788A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010044437A1 (ja) | 2008-10-17 | 2010-04-22 | 独立行政法人産業技術総合研究所 | 硫黄変性ポリアクリロニトリル、その製造方法、及びその用途 |
| JP2012041220A (ja) * | 2010-08-17 | 2012-03-01 | Fukuoka Univ | 硫黄複合化活性炭およびその製造方法 |
| WO2012102037A1 (ja) | 2011-01-27 | 2012-08-02 | 出光興産株式会社 | アルカリ金属硫化物と導電剤の複合材料 |
| JP2015088232A (ja) * | 2013-10-28 | 2015-05-07 | ナガセケムテックス株式会社 | 薄膜硫黄被覆活性炭の製造方法、薄膜硫黄被覆活性炭、正極合材及び全固体型リチウム硫黄電池 |
| JP2016127005A (ja) * | 2014-12-31 | 2016-07-11 | 現代自動車株式会社Hyundai Motor Company | 全固体リチウム電池の陽極及びこれを含む二次電池 |
| JP2019040752A (ja) * | 2017-08-25 | 2019-03-14 | 株式会社サムスン日本研究所 | 全固体型二次電池 |
| WO2019078130A1 (ja) * | 2017-10-19 | 2019-04-25 | 三菱瓦斯化学株式会社 | 全固体電池の製造方法 |
| JP2019179604A (ja) * | 2018-03-30 | 2019-10-17 | 東京電力ホールディングス株式会社 | リチウム硫黄固体電池 |
| JP2019179603A (ja) * | 2018-03-30 | 2019-10-17 | 東京電力ホールディングス株式会社 | リチウム硫黄固体電池、電池用正極シート、電池用負極シート、筒状電池、集電体付き筒状電池、積層電池 |
Non-Patent Citations (1)
| Title |
|---|
| CHEM. MATER., vol. 23, 2011, pages 5024 - 5028 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4235856A1 (en) | 2023-08-30 |
| CN116472629A (zh) | 2023-07-21 |
| JP7493054B2 (ja) | 2024-05-30 |
| JPWO2022090757A1 (ja) | 2022-05-05 |
| US20230395788A1 (en) | 2023-12-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7304014B2 (ja) | 全固体リチウムイオン二次電池用固体電解質層およびこれを用いた全固体リチウムイオン二次電池 | |
| JP7206798B2 (ja) | リチウムイオン伝導性固体電解質、およびこれを用いた電気化学デバイス | |
| JP7406982B2 (ja) | 全固体電池およびその製造方法 | |
| JP2022082060A (ja) | 二次電池 | |
| JP2021136215A (ja) | リチウムイオン二次電池 | |
| JP2021099958A (ja) | 全固体リチウムイオン二次電池 | |
| JP2020140950A (ja) | 硫黄活物質含有電極組成物、並びにこれを用いた電極および電池 | |
| JP2023092911A (ja) | 硫化物固体電解質複合体およびこれを用いた電気デバイス | |
| JP2022115375A (ja) | 電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス | |
| JP2022131400A (ja) | 電気デバイス用正極材料並びにこれを用いた全固体リチウム二次電池 | |
| JP2023082742A (ja) | 正極材料の製造方法 | |
| JP2022090295A (ja) | 二次電池用正極材料およびこれを用いた二次電池 | |
| JP7414520B2 (ja) | 全固体電池用電極 | |
| WO2022090757A1 (ja) | 電気デバイス用正極材料並びにこれを用いた電気デバイス用正極および電気デバイス | |
| JP2022062519A (ja) | 正極活物質層の内部抵抗低減剤、並びにこれを用いた二次電池用電極材料 | |
| JP2021170494A (ja) | 二次電池 | |
| JP7248136B2 (ja) | リチウムイオン二次電池用正極活物質 | |
| WO2023187466A1 (ja) | 正極材料およびこれを用いた二次電池 | |
| JP7523533B2 (ja) | 二次電池用正極 | |
| WO2022069913A1 (ja) | 二次電池 | |
| WO2021250437A1 (ja) | 二次電池 | |
| JP2023064496A (ja) | 正極材料およびこれを用いた二次電池 | |
| JP2023064505A (ja) | 正極材料の製造方法 | |
| JP2024084965A (ja) | 全固体電池 | |
| JP2023086518A (ja) | 全固体リチウム二次電池の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20959660 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2022558362 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18033541 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202080106687.0 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2020959660 Country of ref document: EP Effective date: 20230526 |