WO2020129594A1 - 吸水性樹脂、吸収体、吸収性物品、及び吸水性樹脂の製造方法 - Google Patents
吸水性樹脂、吸収体、吸収性物品、及び吸水性樹脂の製造方法 Download PDFInfo
- Publication number
- WO2020129594A1 WO2020129594A1 PCT/JP2019/047068 JP2019047068W WO2020129594A1 WO 2020129594 A1 WO2020129594 A1 WO 2020129594A1 JP 2019047068 W JP2019047068 W JP 2019047068W WO 2020129594 A1 WO2020129594 A1 WO 2020129594A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- water
- absorbent resin
- chain
- absorbent
- monomer
- Prior art date
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/261—Synthetic macromolecular compounds obtained by reactions only involving carbon to carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F6/00—Post-polymerisation treatments
- C08F6/008—Treatment of solid polymer wetted by water or organic solvents, e.g. coagulum, filter cakes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/28—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety
- C08F220/285—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety
- C08F220/286—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety and containing polyethylene oxide in the alcohol moiety, e.g. methoxy polyethylene glycol (meth)acrylate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/24—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives thereof
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/264—Synthetic macromolecular compounds derived from different types of monomers, e.g. linear or branched copolymers, block copolymers, graft copolymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/265—Synthetic macromolecular compounds modified or post-treated polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/265—Synthetic macromolecular compounds modified or post-treated polymers
- B01J20/267—Cross-linked polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28014—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their form
- B01J20/28016—Particle form
- B01J20/28019—Spherical, ellipsoidal or cylindrical
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28014—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their form
- B01J20/28028—Particles immobilised within fibres or filaments
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28014—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their form
- B01J20/28047—Gels
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/3085—Chemical treatments not covered by groups B01J20/3007 - B01J20/3078
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
- C08F2/32—Polymerisation in water-in-oil emulsions
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/04—Acids; Metal salts or ammonium salts thereof
- C08F220/06—Acrylic acid; Methacrylic acid; Metal salts or ammonium salts thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F6/00—Post-polymerisation treatments
- C08F6/24—Treatment of polymer suspensions
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/02—Making solutions, dispersions, lattices or gels by other methods than by solution, emulsion or suspension polymerisation techniques
- C08J3/03—Making solutions, dispersions, lattices or gels by other methods than by solution, emulsion or suspension polymerisation techniques in aqueous media
- C08J3/075—Macromolecular gels
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/12—Powdering or granulating
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/24—Crosslinking, e.g. vulcanising, of macromolecules
- C08J3/245—Differential crosslinking of one polymer with one crosslinking type, e.g. surface crosslinking
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2220/00—Aspects relating to sorbent materials
- B01J2220/50—Aspects relating to the use of sorbent or filter aid materials
- B01J2220/68—Superabsorbents
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2333/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers
- C08J2333/02—Homopolymers or copolymers of acids; Metal or ammonium salts thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2433/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers
- C08J2433/04—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers esters
- C08J2433/06—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers esters of esters containing only carbon, hydrogen, and oxygen, the oxygen atom being present only as part of the carboxyl radical
- C08J2433/08—Homopolymers or copolymers of acrylic acid esters
Definitions
- the present invention relates to a water absorbent resin, an absorbent body, an absorbent article, and a method for producing the water absorbent resin.
- Patent Literature 1 provides a sanitary article in which the water-absorbent resin contained in the absorbent body has improved water-absorbing performance, and thus is unlikely to cause rash even after long-term use.
- the hydrophilic fiber which is bulkier than the water absorbent resin
- it is effective to reduce the content of the hydrophilic fiber, which is bulkier than the water absorbent resin, in order to reduce the thickness of the absorber.
- the content of the hydrophilic fibers having the function of diffusing the liquid in the absorber the liquid absorption may be localized in the absorber, and as a result, the absorbing ability of the absorber may not be sufficiently exhibited.
- the present disclosure has been made in view of the above problems, can configure a thin absorber, and can reduce the amount of reversion of the absorber, water absorption that can impart a sufficient diffusing force to the absorber It is an object of the present invention to provide a resin, an absorber containing the water absorbent resin, and a method for producing the water absorbent resin.
- the present inventors if it is possible to produce a new water-absorbent resin having the functions of both the conventional water-absorbent resin and the hydrophilic fiber, while reducing the thickness of the absorber, its water-absorbing performance compared to the conventional absorber.
- the research was conducted on the assumption that it could be improved or maintained in a necessary and sufficient state.
- the absorber can be made thin, and the amount of reversion of the absorber can be reduced, and the liquid can be sufficiently diffused in the absorber. I found it.
- the water absorbent resin according to an aspect of the present disclosure has a shape in which a plurality of substantially spherical particles are connected in a chain.
- the water-absorbent resin having a shape in which a plurality of substantially spherical particles are connected in a chain form, the particles have water-absorbing performance as a characteristic of the water-absorbent resin, and due to its unique shape, sufficiently diffuse the liquid. Can be made.
- the water-absorbent resin may be a copolymer of two or more kinds of monomers.
- an absorber according to one aspect of the present disclosure includes the above water-absorbent resin.
- the above absorber contains a water absorbent resin in which a plurality of substantially spherical particles are connected in a chain. Therefore, also as the absorber, it is possible to sufficiently diffuse the liquid while having the absorbing performance.
- the present inventors have found that when the water-absorbent resin used in the absorber has a specific shape, it becomes better to the touch by combining the water-absorbent resin with hydrophilic fibers to form the absorber. It was That is, the present inventors have found that a thin absorber can be formed, and not only the amount of reversion of the absorber can be reduced, but also an effect of improving the touch can be obtained.
- an absorber includes a water-absorbent resin having a shape in which a plurality of substantially spherical particles are connected in a chain, and a hydrophilic fiber.
- the water-absorbent resin having a shape in which a plurality of substantially spherical particles are linked in a chain shape is softer than conventional water-absorbent resins, and therefore an absorbent body using this water-absorbent resin has a good feel to the skin.
- “good (good) touch” refers to a state in which the variation (MMD) of the friction coefficient representing the frictional characteristics and the surface roughness (SMD) are both small, and the surface is smooth. It means a state in which a nice touch is obtained.
- the water absorbent resin can be a copolymer of two or more types of monomers.
- an absorbent article according to an aspect of the present disclosure includes the above absorber.
- the above-mentioned absorbent article includes an absorbent body containing a water-absorbent resin in a shape in which a plurality of the above-mentioned substantially spherical particles are connected in a chain. Therefore, the absorbent article also has good touch.
- a method for producing a water absorbent resin includes a polymerization step in which a monomer A and a monomer B are copolymerized by a reverse phase suspension polymerization method, and a hydrogel obtained in the polymerization step. And a drying step for drying the monomer, the monomer A contains at least one of (meth)acrylic acid and a neutralized product thereof, and the monomer B is an alkoxypolyalkylene glycol (meth)acrylate.
- the ratio ( ⁇ / ⁇ ) of the molar amount ( ⁇ ) of the monomer A to be subjected to copolymerization and the molar amount ( ⁇ ) of the monomer B is 1/4 to 4 is there.
- the water-absorbent resin having a high water-absorbing performance and capable of sufficiently diffusing a liquid, a plurality of substantially spherical particles having a shape in which they are linked in a chain shape.
- acrylic and “methacrylic” are collectively referred to as “(meth)acrylic”.
- sucrose fatty acid ester and polyglycerin fatty acid ester may be added to the reaction system as a surfactant.
- a water-absorbent resin having a shape in which a plurality of substantially spherical particles are linked in a chain Can be suitably manufactured.
- the polymerization may be performed in the presence of a polymeric dispersant.
- the particles are appropriately dispersed in the polymerization step, and it is difficult for the particles to be aggregated. Therefore, a plurality of substantially spherical particles are linked in a chain.
- a shaped water absorbent resin can be suitably obtained.
- the hydrogel in the drying step, can be dehydrated by azeotropic distillation of the hydrocarbon dispersion medium until the water content becomes 50% by mass or less.
- the hydrated gel is dehydrated by azeotropic distillation of the hydrocarbon dispersion medium until the water content becomes 50% by mass or less, whereby a plurality of substantially spherical particles are linked in a chain.
- a shaped water absorbent resin can be suitably produced.
- a thin absorbent body can be configured, and the amount of reversion of the absorbent body can be reduced, and a water-absorbent resin capable of sufficiently diffusing a liquid in the absorbent body, the water-absorbent resin.
- a water-absorbent resin capable of sufficiently diffusing a liquid in the absorbent body, the water-absorbent resin.
- an absorber containing a water-soluble resin and a method for producing the water-absorbent resin.
- an absorbent body having a soft touch and an absorbent article including the absorbent body there are provided an absorbent body having a soft touch and an absorbent article including the absorbent body.
- FIG. 1 is a cross-sectional view of an absorbent article including an absorbent body according to an embodiment of the present disclosure.
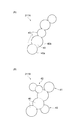
- FIG. 2A and FIG. 2B are diagrams showing examples of the water absorbent resin according to the embodiment of the present invention.
- 3A to 3F are SEM images of one water-absorbent resin according to the example.
- FIG. 4 is a diagram showing SEM images of a plurality of water absorbent resins according to the example.
- FIG. 5 is a figure which shows the measuring apparatus used for the measurement of the water absorption capacity of a water absorbent resin under pressure.
- FIG. 1 is a cross-sectional view showing a configuration of an absorbent article including an absorbent body according to an embodiment of the present disclosure.
- the absorbent article 1 shown in FIG. 1 includes an absorber 2, a liquid permeable sheet 3, and a liquid impermeable sheet 4.
- the absorber 2 is sandwiched between the liquid-permeable sheet 3 and the liquid-impermeable sheet 4 which are arranged to face each other.
- the absorbent article examples include a paper diaper, an incontinence pad, a sanitary napkin, an incontinence liner, a panty liner, a pet sheet, a food drip sheet, and the like, and the water-absorbent resin of the present disclosure realizes a thin absorbent body.
- the absorbent article is a product that comes into contact with the body (paper diaper, incontinence pad, sanitary napkin, incontinence liner, panty liner, etc.). ..
- the water absorbent resin of the present disclosure it is possible to reduce the thickness of the absorbent body and the absorbent article including the absorbent body, so that it is possible to obtain an absorbent article that is inconspicuous even when worn and in which transportation cost can be reduced.
- the absorbent body of the present disclosure the touch of the absorbent article including the absorbent body can be improved.
- the absorber 2 includes at least the absorbent core 20.
- the absorber 2 further includes a core wrap 23 in addition to the absorbent core 20.
- the absorbent core 20 is a core member that influences the absorbent performance of the absorbent body 2, and includes at least a water absorbent resin 21.
- the absorbent core 20 further includes hydrophilic fibers 22 in addition to the water absorbent resin 21, but the hydrophilic fibers 22 may not be included.
- the core wrap 23 is a sheet-shaped member that retains the shape of the absorbent core 20. Examples of the core wrap 23 include tissue and non-woven fabric. When the core wrap 23 is used, the shape retention method of the absorbent core 20 is not particularly limited. For example, as shown in FIG. 1, the absorbent core 20 may be held by wrapping the absorbent core 20 with one core wrap 23, or by sandwiching the absorbent core 20 with two core wraps 23. 20 may be molded (not shown).
- the hydrophilic fiber 22 is a member having a function of diffusing a liquid inside the absorber 2 by a capillary phenomenon.
- the hydrophilic fibers 22 include fibers made of cellulose fibers and synthetic resins.
- the cellulose fibers include natural cellulose fibers such as cotton-like pulp, mechanical pulp, chemical pulp, and semi-chemical pulp obtained from wood; regenerated cellulose fibers such as rayon and fibryl rayon; semisynthetic cellulose fibers such as acetate and triacetate. And so on.
- fibers made of synthetic resin include fibers made of hydrophilized polyamide, polyester, polyolefin and the like. These fibers may be used alone or as a mixed fiber in which two or more kinds are combined.
- the amount of the hydrophilic fibers 22 used in the absorber 2 is, for example, 60% by mass or less based on the mass of the absorbent core 20 (100% by mass) in order to realize the thinning of the absorbent body 2 and the absorbent article 1. , Preferably 55 mass% or less, more preferably 50 mass% or less.
- the hydrophilic fiber 22 may be used in an amount of 0% by mass (that is, the absorbent core 20 may not include the hydrophilic fiber 22).
- the amount of the hydrophilic fibers 22 used is preferably 10% by mass or more, more preferably 20% by mass or more, and further preferably 30% by mass or more, based on the mass of the absorbent core 20.
- the absorbent core 20 may further include synthetic fibers made of synthetic resin such as polyamide, polyester, and polyolefin as a reinforcing agent. Further, an adhesive binder such as a heat-fusible synthetic fiber, a hot-melt adhesive, or an adhesive emulsion may be added to the absorbent core 20 in order to enhance the shape retention of the absorbent body.
- synthetic fibers made of synthetic resin such as polyamide, polyester, and polyolefin as a reinforcing agent.
- an adhesive binder such as a heat-fusible synthetic fiber, a hot-melt adhesive, or an adhesive emulsion may be added to the absorbent core 20 in order to enhance the shape retention of the absorbent body.
- the thickness of the absorber 2 can be, for example, 0.1 to 10 mm.
- the structure of the absorbent core 20 in the absorbent body 2 include a mixing structure in which the water absorbent resin 21 and the hydrophilic fibers 22 are uniformly blended, and the water absorbent resin 21 is held between a plurality of layers of the hydrophilic fibers 22. A sandwich structure etc. are mentioned.
- the absorbent core 2 and the absorbent core 20 of the present embodiment are not limited to such examples.
- the amount of the water absorbent resin 21 used in the absorber 2 is, for example, 5 to 100% by mass, 30 to 90% by mass, or 50 to 80% by mass, based on the mass of the absorbent core 20 (100% by mass).
- the amount of the water absorbent resin 21 used is 5% by mass or more, the absorption capacity of the absorbent body 2 increases, and liquid leakage and backflow tend to be suppressed.
- the water absorbent resin 21 constituting the absorbent body 2 includes a water absorbent resin 211 (hereinafter, referred to as chain water absorbent resin 211) having a shape in which a plurality of substantially spherical particles are connected in a chain shape. There is.
- the chain-shaped water absorbent resin 211 has a long portion (that is, elongated) because the particles are connected in a chain.
- the water absorbent resin 21 may include a water absorbent resin 212 having a shape different from that of the chain water absorbent resin 211 (hereinafter, referred to as a non-chain water absorbent resin 212).
- the water absorbent resin 21 includes a chain water absorbent resin 211 and a non-chain water absorbent resin 212. Details of the chain water absorbent resin 211 will be described later.
- the non-chain water absorbent resin 212 is a resin that does not correspond to the chain water absorbent resin 211 described below, and has, for example, a substantially spherical shape, a crushed shape, a granular shape, or a shape in which a plurality of particles thereof are aggregated. It is a water absorbent resin.
- the shape of the non-chain water absorbent resin 212 is not particularly limited as long as it is different from the chain water absorbent resin 211.
- the ratio of the chain water-absorbent resin 211 in the water-absorbent resin 21 included in the absorbent body 2 is preferably 20% by mass at the lower limit, based on the total amount of the water-absorbent resin 21 (100% by mass).
- the upper limit is, for example, 50% by mass, preferably 60% by mass, more preferably 70% by mass, and particularly preferably 90% by mass.
- the upper limit may be 100% by mass (that is, only the chain water absorbent resin 211 is used as the water absorbent resin 21), but by setting the ratio of the chain water absorbent resin 211 in the above range, The absorber can be made thinner, the amount of reversion of the absorber can be further reduced, and the liquid can be more diffusible to the absorber.
- liquid permeable sheet 3 examples include a nonwoven fabric made of synthetic resin such as polyethylene, polypropylene, polyester, polyamide, and a porous synthetic resin sheet.
- liquid impermeable sheet 4 examples include a sheet made of a synthetic resin such as polyethylene, polypropylene and polyvinyl chloride, a sheet made of a composite material of these synthetic resins and a non-woven fabric, and the like.
- the size and thickness of the liquid-permeable sheet 3 and the liquid-impermeable sheet 4 are appropriately adjusted depending on the application of the absorbent article or the like. Since a thin absorber can be obtained by using the chain water absorbent resin 211 of the present invention, even if a relatively thick liquid permeable sheet 3 and/or liquid impermeable sheet 4 is used, The absorbent article 1 can be obtained.
- the absorber 2 and the absorbent article 1 may further contain additives such as a lubricant, a deodorant, an antibacterial agent, and a fragrance.
- additives such as a lubricant, a deodorant, an antibacterial agent, and a fragrance.
- the addition site of these additives can be appropriately set based on the function of the additives, and for example, inside the absorbent core 20, between the core wrap 23 and the absorbent core 20, the liquid permeable sheet 3 and/or liquid impermeable. It can be added between the sheet 4 and the core wrap 23.
- the above-mentioned absorber 2 preferably has a friction coefficient variation (MMD) described below of more than 0 and 0.03 or less, more preferably more than 0 and 0.01 or less.
- the variation of the friction coefficient (MMD) is an index of roughness, and the larger the value, the more rough the texture is. Therefore, the smaller the measured value of the variation of the friction coefficient (MMD), the less rough it is and the more preferable it is for use in an absorber.
- the method for measuring the variation (MMD) of the friction coefficient is as described in the examples of this specification.
- the surface roughness (SMD) of the absorber 2 is preferably more than 0 and 5.0 or less, and more preferably more than 0 and 3.0 or less.
- the surface roughness (SMD) is an index relating to surface irregularities, and the larger the value, the more remarkable the surface irregularities. Therefore, the smaller the surface roughness (SMD) is, the less the unevenness is, and the more preferable it is for use in the absorber.
- the method of measuring the surface roughness (SMD) is as described in the examples of this specification.
- the absorbent body 2 in which both the variation (MMD) of the friction coefficient and the surface roughness (SMD) satisfy the above-described ranges has a good feel to the skin.
- FIG. 2 shows an example of the chain water absorbent resin 211.
- the chain water absorbent resin 211 has a shape in which a plurality of substantially spherical particles 40 are connected in a chain shape.
- the substantially spherical particle 40 is a particle having a substantially circular cross section.
- the cross-sectional shape of the particles 40 is not limited to a perfect circle, and may be an ellipse or an irregular shape close to a circle. For example, one having an aspect ratio of 1.5 or less can be referred to as “substantially spherical”.
- the particles 40 have a particle size of, for example, 10 ⁇ m to 600 ⁇ m, preferably 50 ⁇ m to 500 ⁇ m.
- the particle size of the particles 40 can be controlled by, for example, the amount of a surfactant to be described later, the stirring speed during polymerization, and the like.
- the plurality of particles 40 are connected in a state in which each particle can be discriminated.
- the particles 40a and the particles 40b included in the plurality of particles 40 in the chain-like water absorbent resin 211A of FIG. 2A are arranged adjacent to each other, and a part of the surface of the particle 40a corresponds to the surface of the particle 40b.
- the particles 40a and the particles 40b are connected so as to overlap with the portion.
- the boundary 45 between the particles 40a and the particles 40b in the connecting portion with the particles 40a and the particle 40b has a width (width when the connecting portion is viewed from the side surface) smaller than the maximum diameter of the particles 40a and 40b.
- a constricted boundary 45 can be specified between the adjacent particles 40 a and 40 b, and the particles 40 a and 40 b can be discriminated with the boundary 45 as a boundary.
- the plurality of particles 40 are connected to adjacent particles in a distinguishable state from each other.
- a plurality of substantially spherical particles 40 are linearly connected to each other in a distinguishable state, so that a chain-shaped water absorbent resin 211 having a “chain-shaped” shape is obtained.
- the chain-shaped water absorbent resin 211 has a shape like a bead (a method in which a plurality of beads having a through hole are continuously connected through a thread so that there is no space between adjacent beads).
- the plurality of particles 40 being connected in a chain shape means a state in which the plurality of particles 40 are connected in a linear or branched chain shape.
- the chain water absorbent resin 211 at least 5 or more particles 40 are connected in a chain shape, and among them, 3 or more particles 40 form a main chain.
- the main chain corresponds to the longest part of the chain water absorbent resin 211.
- the five particles 40 form a straight chain.
- the chain-like water absorbent resin 211 only needs to have the plurality of particles 40 connected in a chain shape, and the structure thereof is not particularly limited.
- the size of each of the branched particles 40 is not particularly limited, and for example, particles 40 having different particle diameters may be connected to each other.
- the number of particles 40 connected in a chain is preferably 5 or more, more preferably 10 or more.
- the upper limit is not particularly limited, and the longer the chain-like water absorbent resin is obtained, the more the upper limit is practically 100 or less.
- the chain-shaped water absorbent resin 211 is most preferably composed of only substantially spherical particles 40 (that is, a structure having only “chain-shaped portions” in which the substantially spherical particles 40 are continuously connected),
- the substantially spherical particles 40 may be connected intermittently via non-spherical particles. That is, non-spherical particles may be interposed between the adjacent substantially spherical particles 40.
- the chain water absorbent resin 211 may have a structure in which non-spherical particles are connected to the ends of the chain portion.
- the ratio of the substantially spherical particles 40 is 70% by mass or more, preferably 80% by mass or more, and more preferably 90% by mass or more.
- the chain-shaped water absorbent resin 211 having a shape in which a plurality of substantially spherical particles 40 are connected in a chain shape has a long shape in which the main chain extends in the longitudinal direction.
- the particle diameter of the chain water absorbent resin 211 is, for example, 150 ⁇ m, preferably 200 ⁇ m, and more preferably 250 ⁇ m.
- the upper limit value thereof is, for example, 5000 ⁇ m, preferably 3000 ⁇ m, more preferably 2500 ⁇ m, and even more preferably 2000 ⁇ m.
- a sieve is used to measure the particle diameter of the chain water absorbent resin 211. Specifically, from the top of the JIS standard sieve, a sieve having an opening of 850 ⁇ m, a sieve having an opening of 600 ⁇ m, a sieve having an opening of 500 ⁇ m, a sieve having an opening of 425 ⁇ m, a sieve having an opening of 300 ⁇ m, a sieve having an opening of 250 ⁇ m, and an opening. A 150 ⁇ m sieve and a saucer are combined in this order. Then, the chain-like water absorbent resin 211 is put in the combined uppermost sieve, and shaken for 20 minutes using a low-tap shaker to perform classification.
- the mass of the chain-like water absorbent resin remaining on each sieve is calculated as a mass percentage with respect to the total amount, and the particle size distribution is obtained.
- this particle size distribution by integrating on the sieve in order from the larger particle size, the relationship between the mesh opening of the sieve and the integrated value of the mass percentage of the chain-like water absorbent resin remaining on the sieve is plotted on a logarithmic probability paper. .. By connecting the plots on the probability paper with straight lines, the particle diameter (medium particle diameter) corresponding to the cumulative mass percentage of 50 mass% is the particle diameter of the chain water-absorbent resin 211.
- the chain water-absorbent resin 211 is a water-absorbent resin itself, it has a higher water retention capacity than hydrophilic fibers and can reduce the amount of reversion of the absorbent body. Further, the chain water-absorbent resin 211 is softer than the conventional water-absorbent resin (non-chain water-absorbent resin), and therefore, it is possible to form an absorbent body and an absorbent article having a good touch. Further, the chain water absorbent resin 211 can sufficiently diffuse the liquid in the absorber. Further, since the chain water absorbent resin 211 is less bulky than the hydrophilic fiber, it is possible to form the thin absorber 2.
- the water-retaining ability of the chain water-absorbent resin 211 with respect to physiological saline is, for example, for the chain water-absorbent resin 1 g, the lower limit value is 15 g, preferably 20 g, and more preferably 25 g.
- the upper limit value is, for example, 60 g, preferably 50 g, and more preferably 45 g.
- the method for measuring the water retention capacity of physiological saline is as described in the examples of the present specification.
- the lower limit of the water absorption rate of the chain water absorbent resin 211 for physiological saline is, for example, 10 seconds, preferably 20 seconds, and more preferably 30 seconds. Further, the upper limit value thereof is, for example, 90 seconds, preferably 70 seconds, and more preferably 60 seconds.
- the method of measuring the water absorption rate is as described in the examples of the present specification.
- the water absorption capacity of the above chain water absorbent resin 211 against physiological saline is, for example, with respect to 1 g of the chain water absorbent resin, the lower limit is 3 ml, preferably 5 ml, and more preferably 6 ml. is there.
- the method of measuring the water absorption capacity under pressure is as described in the examples of the present specification.
- the chain-shaped water absorbent resin 211 has a softer feel than the non-chained water absorbent resin due to its unique shape.
- the chain-like water absorbent resin 211 having a softer touch than the non-chain-like water absorbent resin for the absorbent body 2 and the absorbent article 1 the effect of improving the touch is obtained.
- the feel of the water absorbent resin 21 contained in the absorber 2 greatly affects the feel of the absorber 2. Therefore, by including the chain-like water absorbent resin 211 having a soft touch in the absorbent body 2, the absorbent body 2 has a good texture. Further, since the absorbent body 2 has good touch, the absorbent article 1 also has good touch.
- the chain water-absorbent resin 211 can be produced, for example, by copolymerizing two or more kinds of monomers.
- the method for producing the chain water-absorbent resin 211 includes a polymerization step of copolymerizing a monomer A and a monomer B (different types from the monomer A) by a reverse phase suspension polymerization method, A drying step of drying the hydrogel obtained in the polymerization step.
- the monomer A used in the polymerization step contains at least one of (meth)acrylic acid and a neutralized product thereof, and the monomer B is an alkoxypolyalkylene glycol (meth)acrylate.
- the monomer B is an alkoxypolyalkylene glycol (meth)acrylate.
- the neutralized product of (meth)acrylic acid can be obtained, for example, by neutralizing (meth)acrylic acid with an alkaline neutralizing agent.
- the neutralization degree of the neutralized product is 10 to 100 mol% of the acidic groups in the (meth)acrylic acid. It is desirable that it is 50 to 90 mol %, more preferably 60 to 80 mol %.
- alkaline neutralizing agent examples include alkali metal salts such as sodium hydroxide, sodium carbonate, sodium hydrogen carbonate, potassium hydroxide and potassium carbonate; ammonia and the like. These alkaline neutralizing agents may be used in the form of an aqueous solution in order to simplify the neutralizing operation.
- the above alkaline neutralizing agents may be used alone or in combination of two or more.
- an aqueous solution (neutralizing solution) containing an alkaline neutralizing agent such as sodium hydroxide or potassium hydroxide is added dropwise to the (meth)acrylic acid aqueous solution and mixed. It can be done by The concentration of the neutralized aqueous solution is, for example, 20% by mass to 50% by mass.
- the neutralized product of (meth)acrylic acid is an alkali metal salt of (meth)acrylic acid partially neutralized at the above-mentioned neutralization degree, and more preferably at the above-mentioned neutralization degree. It is a sodium salt of hydrated (meth)acrylic acid.
- the alkoxy polyalkylene glycol (meth)acrylate contained in the monomer B is represented by the following general formula (1).
- n represents the number of repeating units of ethylene glycol and is an integer greater than 0.
- the lower limit value of n is, for example, 5, preferably 7, and more preferably 9.
- the upper limit of n is 18, for example, preferably 15, and more preferably 13.
- R represents hydrogen or a substituted or unsubstituted alkyl group.
- the number of carbon atoms of R is preferably 4 or less, more preferably 3 or less, and more preferably 2 or less because it is easily copolymerized with the above-mentioned (meth)acrylic acid or a neutralized product thereof. More preferably, 1 is most preferable.
- R is preferably an unsubstituted alkyl group having 3 or less carbon atoms (methyl group, ethyl group, propyl group, or isopropyl group), more preferably an unsubstituted alkyl group having 2 or less carbon atoms (methyl group).
- Group or ethyl group and particularly preferably an unsubstituted alkyl group having 1 carbon atom (methyl group).
- the ratio ( ⁇ / ⁇ ) of the molar amount ( ⁇ ) of the monomer A to be subjected to copolymerization and the molar amount ( ⁇ ) of the monomer B is preferably a lower limit value of 1 ⁇ 4, and more preferably It is 3/7, and more preferably 2/3. Further, the upper limit value thereof is preferably 4, more preferably 7/3, and further preferably 3/2.
- the ratio ( ⁇ / ⁇ ) of these monomers is within the above range, the substantially spherical particles 40 are likely to be linked in a chain, so that the chain water absorbent resin can be produced in a high yield. Further, since the chain-like water absorbent resin produced is less likely to aggregate, it can be mixed well with the hydrophilic fiber.
- the chain water-absorbent resin 211 in addition to the above-mentioned two kinds of monomers A and B, another monomer C may be added and copolymerized.
- the monomer C include water-soluble ethylenically unsaturated monomers.
- the water-soluble ethylenically unsaturated monomer include 2-(meth)acrylamide-2-methylpropanesulfonic acid and its alkali salt, (meth)acrylamide, N,N-dimethyl(meth)acrylamide, 2-hydroxy.
- the water-soluble ethylenically unsaturated monomer contains an amino group
- the amino group may be quaternized.
- a water-soluble monomer other than the above-mentioned water-soluble ethylenically unsaturated monomer may be selected as long as the expression of the effect of the present disclosure is not hindered.
- Such a monomer can be used by being mixed with an aqueous solution containing the above water-soluble ethylenically unsaturated monomer.
- the monomer C may be neutralized with an alkali neutralizing agent and used in the same manner as the neutralized product of (meth)acrylic acid.
- the neutralizing agent used for neutralization and the preferred range of the degree of neutralization are the same as those for the neutralized product of (meth)acrylic acid.
- the total amount of the monomer A and the monomer B is preferably 70 mol% or more, and 80 mol% or more with respect to the total amount of the monomers. Is more preferable. Further, in this case, the total amount is necessarily less than 100 mol %.
- the monomers A to C used in the polymerization step are usually preferably used as an aqueous solution.
- the concentration of the monomer in the aqueous solution of the monomer (hereinafter referred to as "monomer aqueous solution") is usually in the range of 25% by mass to the saturated concentration, preferably 35% by mass to 60% by mass. ..
- the water used include tap water, distilled water, ion-exchanged water and the like.
- an aqueous monomer solution is dispersed in a hydrocarbon dispersion medium in the presence of a surfactant, and the above-mentioned monomer is polymerized by using a water-soluble radical polymerization initiator or the like.
- An internal crosslinking agent may be used during the polymerization.
- surfactant polymer dispersant, hydrocarbon dispersion medium, water-soluble radical polymerization initiator, internal cross-linking agent, etc. used in the polymerization reaction, those which can be usually used in the reverse phase suspension polymerization method are used. Good.
- sucrose fatty acid ester among the nonionic surfactants, sucrose fatty acid ester, polyglycerin fatty acid ester, propylene glycol fatty acid ester, sorbitan fatty acid ester and the like can be used. Among these, it is preferable to use the sucrose fatty acid ester and/or the polyglycerin fatty acid ester, whereby the chain water absorbent resin can be suitably obtained.
- An anionic surfactant may be used as the surfactant.
- anionic surfactant examples include fatty acid salt, alkylbenzene sulfonate, alkylmethyl taurate, polyoxyethylene alkylphenyl ether sulfate ester salt, polyoxyethylene alkyl ether sulfonate, polyoxyethylene alkyl ether. And phosphoric acid ester of polyoxyethylene alkyl allyl ether, and the like. Further, these surfactants may be used alone or in combination of two or more kinds.
- the HLB (hydrophilic/lipophilic balance) of the surfactant is preferably 6 or less.
- the lower limit value of HLB of the surfactant is, for example, 2, and preferably 3.
- the amount of the surfactant added to the reaction system is 0.1 parts by mass with respect to 100 parts by mass of the aqueous monomer solution used in the polymerization step, from the viewpoint that the effect on the amount used is sufficiently obtained and the cost is economical.
- the amount is preferably ⁇ 5 parts by mass, more preferably 0.2 parts by mass to 3 parts by mass, further preferably 0.5 parts by mass to 2 parts by mass.
- a polymeric dispersant together with a surfactant in reverse phase suspension polymerization.
- the polymer dispersant particles of the water-absorbent resin are appropriately dispersed in the hydrocarbon dispersion medium, and the chain-like water-absorbent resin can be preferably obtained without forming lumps.
- Polymeric dispersants that can be used include maleic anhydride-modified polyethylene, maleic anhydride-modified polypropylene, maleic anhydride-modified ethylene/propylene copolymer, maleic anhydride-modified EPDM (ethylene/propylene/diene/terpolymer), and anhydrous.
- Maleic acid-modified polybutadiene maleic anhydride/ethylene copolymer, maleic anhydride/propylene copolymer, maleic anhydride/ethylene/propylene copolymer, maleic anhydride/butadiene copolymer, polyethylene, polypropylene, ethylene/propylene Examples thereof include copolymers, oxidized polyethylene, oxidized polypropylene, oxidized ethylene/propylene copolymers, ethylene/acrylic acid copolymers, ethyl cellulose and ethyl hydroxyethyl cellulose.
- maleic anhydride-modified polyethylene maleic anhydride-modified polypropylene
- maleic anhydride-modified ethylene/propylene copolymer maleic anhydride/ethylene copolymer
- maleic anhydride/ethylene copolymer maleic anhydride/ Preferred are propylene copolymer, maleic anhydride/ethylene/propylene copolymer, polyethylene, polypropylene, ethylene/propylene copolymer, oxidized polyethylene, oxidized polypropylene, oxidized ethylene/propylene copolymer and the like.
- These polymeric dispersants may be used alone or in combination of two or more.
- the amount of the polymeric dispersant added to the reaction system is such that the monomer used in the polymerization step is kept in the hydrocarbon dispersion medium in order to keep the dispersed state of the monomer in a good state and to obtain a dispersion effect commensurate with the amount used. It is preferably 0.1 parts by mass to 5 parts by mass, more preferably 0.2 parts by mass to 3 parts by mass, and 0.5 parts by mass to 2 parts by mass with respect to 100 parts by mass of the monomer. More preferably,
- water-soluble radical polymerization initiator examples include persulfates such as potassium persulfate, ammonium persulfate and sodium persulfate; methyl ethyl ketone peroxide, methyl isobutyl ketone peroxide, di-t-butyl peroxide, t-butyl cumylper.
- Peroxides such as oxides, t-butylperoxyacetate, t-butylperoxyisobutyrate, t-butylperoxypivalate, and hydrogen peroxide; 2,2′-azobis(2-methylpropionamidine) 2 Hydrochloride, 2,2'-azobis[2-(N-phenylamidino)propane] dihydrochloride, 2,2'-azobis[2-(N-allylamidino)propane] dihydrochloride, 2,2'- Azobis ⁇ 2-[1-(2-hydroxyethyl)-2-imidazolin-2-yl]propane ⁇ dihydrochloride, 2,2'-azobis ⁇ 2-methyl-N-[1,1-bis(hydroxymethyl) )-2-Hydroxyethyl]propionamide ⁇ , 2,2′-azobis[2-methyl-N-(2-hydroxyethyl)-propionamide], 4,4′-azobis(4-cyanovaleric acid), etc.
- the amount of the water-soluble radical polymerization initiator added to the reaction system is, for example, 0.005 to 1 mol with respect to 100 mol of the monomer used in the polymerization step.
- the amount of the water-soluble radical polymerization initiator used is 0.005 mol or more, the polymerization reaction is efficient because it does not require a long time.
- the amount used is 1 mol or less, a polymerization reaction at an appropriate rate is achieved, which is preferable.
- the water-soluble radical polymerization initiator may be used as a redox polymerization initiator in combination with a reducing agent such as sodium sulfite, sodium hydrogen sulfite, ferrous sulfate, and L-ascorbic acid.
- a reducing agent such as sodium sulfite, sodium hydrogen sulfite, ferrous sulfate, and L-ascorbic acid.
- a chain transfer agent may be included in the aqueous monomer solution used for the polymerization in order to control the liquid absorbing properties of the chain water absorbent resin.
- chain transfer agents include hypophosphites, thiols, thiolic acids, secondary alcohols, amines and the like.
- hydrocarbon dispersion medium examples include chain aliphatic hydrocarbons such as n-hexane, n-heptane, 2-methylhexane, 3-methylhexane, 2,3-dimethylpentane, 3-ethylpentane, and n-octane.
- Alicyclic hydrocarbon such as cyclohexane, methylcyclohexane, cyclopentane, methylcyclopentane, trans-1,2-dimethylcyclopentane, cis-1,3-dimethylcyclopentane, trans-1,3-dimethylcyclopentane;
- aromatic hydrocarbons such as benzene, toluene and xylene.
- hydrocarbon dispersion media may be used alone or in combination of two or more.
- the hydrocarbon dispersion medium may contain at least one compound selected from the group consisting of chain aliphatic hydrocarbons having 6 to 8 carbon atoms and alicyclic hydrocarbons having 6 to 8 carbon atoms. W/O type reversed-phase suspension is in good condition, chain-like water absorbent resin with excellent water absorption rate is easily obtained with suitable particle size, industrially easily available, and stable in quality. From the point of view, the hydrocarbon dispersion medium may contain n-heptane, cyclohexane, or both.
- hydrocarbon dispersion medium for example, commercially available exol heptane (manufactured by Exxon Mobil: n-heptane and 75-85% of isomer hydrocarbons) may be used. Good.
- the amount of the hydrocarbon dispersion medium used is 100 to 1000 parts by mass based on 100 parts by mass of the monomer used in the polymerization step, from the viewpoint of appropriately removing the heat of polymerization and facilitating control of the polymerization temperature.
- the amount is preferably 150 to 800 parts by mass, more preferably 200 to 700 parts by mass.
- the amount of the hydrocarbon dispersion medium used is 100 parts by mass or more, the polymerization temperature tends to be easily controlled.
- the amount of the hydrocarbon dispersion medium used is 1000 parts by mass or less, the productivity of polymerization tends to be improved, which is economical.
- a chain-shaped water-absorbing resin having an internal cross-linking structure inside is obtained by a self-crosslinking reaction that occurs during polymerization, but by using an internal cross-linking agent during polymerization, the internal cross-linking density is adjusted, and the chain water-absorbing resin is adjusted.
- the liquid absorbing property of the resin may be controlled.
- the internal cross-linking agent used include polyols such as ethylene glycol, propylene glycol, trimethylolpropane, glycerin, polyoxyethylene glycol, polyoxypropylene glycol, and polyglycerin; di- or tri(meth) with the above polyols.
- Acrylic esters unsaturated polyesters obtained by reacting the above polyols with unsaturated acids such as maleic acid and fumaric acid; bis(meth)acrylamides such as N,N'-methylenebis(meth)acrylamide; Di- or tri(meth)acrylic acid esters obtained by reacting polyepoxide with (meth)acrylic acid; obtained by reacting polyisocyanates such as tolylene diisocyanate and hexamethylene diisocyanate with hydroxyethyl (meth)acrylate Di(meth)acrylic acid carbamyl esters; two or more polymerizable unsaturated groups such as allylated starch, allylated cellulose, diallyl phthalate, N,N′,N′′-triallyl isocyanurate, and divinylbenzene
- the amount of the internal cross-linking agent added to the reaction system may be 0 mol% (that is, the internal cross-linking agent is not used), but from the viewpoint of ensuring that the resulting chain-shaped water absorbent resin exhibits a sufficient water absorption,
- the amount is preferably more than 0 and 0.03 mol or less, more preferably more than 0 and 0.01 mol or less, and more than 0 and 0.005 mol or less per 1 mol of the monomer used in the step. More preferably,
- the monomer used in the above-mentioned polymerization step, a water-soluble radical polymerization initiator, an internal crosslinking agent, etc., if necessary, a surfactant, a polymeric dispersant, and a hydrocarbon dispersion medium are mixed and heated under stirring. Then, reverse phase suspension polymerization is carried out in a water-in-oil system.
- the order of addition of each component and the like can be appropriately adjusted, for example, a surfactant is premixed with a hydrocarbon dispersion medium, and a water-soluble radical polymerization initiator and an internal cross-linking agent, and a monomer, It is preferable that they are mixed in advance and the mixed liquids obtained in each are mixed to start the polymerization.
- a multi-stage polymerization method in which the aqueous monomer solution is divided and added several times may be used.
- the monomers to be subjected to the first-stage polymerization step include the monomer A and the monomer B.
- the monomer used in the second and subsequent polymerization steps preferably contains the monomer A, more preferably the monomer A alone.
- the amount of the water-soluble radical polymerization initiator and the internal cross-linking agent used in each stage of the polymerization step is based on the monomer used in each stage of the polymerization step.
- the above range is preferable.
- the temperature of the polymerization reaction varies depending on the water-soluble radical polymerization initiator used, but it promotes the polymerization rapidly and shortens the polymerization time to improve the economic efficiency and easily remove the heat of polymerization to allow a smooth reaction. From the viewpoint of carrying out the above, 20 to 110° C. is preferable, and 40 to 90° C. is more preferable.
- the reaction time is usually 0.1 to 4 hours. The completion of the polymerization reaction can be confirmed, for example, by stopping the temperature rise in the reaction system. Thereby, the chain water-absorbent resin is usually obtained in a hydrogel state.
- ⁇ Drying process> In order to remove water from the obtained hydrous gel, it is dried.
- a drying method for example, (a) azeotropic distillation is performed by externally heating the hydrogel in a state of being dispersed in a hydrocarbon dispersion medium, and the hydrocarbon dispersion medium is refluxed to remove water (dehydrate).
- the method (b) the method of taking out the hydrous gel by decantation and drying under reduced pressure, and (c) the method of filtering the hydrous gel by filtration and drying under reduced pressure may be mentioned.
- the method (a) is preferably used to efficiently obtain the chain water-absorbent resin.
- the chain water-absorbing resin can be efficiently obtained by reducing the water content of the hydrogel to about 50 mass% by dehydrating the hydrocarbon dispersant by azeotropic distillation. If the hydrocarbon dispersion medium is first volatilized and dried in a state where the water-containing gel has a high water content, the particles are likely to agglomerate and it becomes difficult to obtain a chain water absorbent resin.
- the particle size of the particles constituting the chain water absorbent resin described in the present embodiment can be controlled by, for example, the amount of the surfactant, the amount of the thickener, the stirring speed during polymerization, and the like.
- the hydrogel surface portion may be crosslinked (post-crosslinked) with a crosslinking agent in the drying step or any of the subsequent steps.
- a crosslinking agent in the drying step or any of the subsequent steps.
- the post-crosslinking is preferably performed at a timing when the hydrogel has a specific water content.
- the time of the post-crosslinking is preferably a time point when the water content of the hydrous gel is 10 to 60% by mass, more preferably 20 to 55% by mass, and further preferably 30 to 50% by mass.
- the water content (mass %) of the water-containing gel is calculated by the following formula.
- Water content [Ww ⁇ (Ww+Ws)] ⁇ 100
- Ww Water used as needed when mixing the post-crosslinking agent, etc. to an amount obtained by subtracting the amount of water extracted outside in the drying process from the amount of water contained in the aqueous liquid before the polymerization in the entire polymerization process
- Water content Ws of the water-containing gel with added amount Solid content calculated from the charged amount of materials such as monomers, cross-linking agents, and initiators constituting the water-containing gel
- a compound having two or more reactive functional groups can be mentioned.
- examples thereof include polyols such as ethylene glycol, propylene glycol, 1,4-butanediol, trimethylolpropane, glycerin, polyoxyethylene glycol, polyoxypropylene glycol and polyglycerin; (poly)ethylene glycol diglycidyl ether, Polyglycidyl compounds such as (poly)glycerin diglycidyl ether, (poly)glycerin triglycidyl ether, trimethylolpropane triglycidyl ether (poly)propylene glycol polyglycidyl ether, (poly)glycerol polyglycidyl ether; epichlorohydrin, epibromhydrin , ⁇ -methylepichlorohydrin and other haloepoxy compounds; 2,4-tolylene diisocyanate, hex
- Carbonate compounds; hydroxyalkylamide compounds such as bis[N,N-di( ⁇ -hydroxyethyl)]adipamide are more preferable.
- polyglycidyl compounds such as (poly)ethylene glycol diglycidyl ether, (poly)glycerin diglycidyl ether, (poly)glycerin triglycidyl ether, (poly)propylene glycol polyglycidyl ether, and polyglycerol polyglycidyl ether are more preferable. preferable.
- These post-crosslinking agents may be used alone or in combination of two or more.
- the amount of the post-crosslinking agent used cannot be unconditionally determined because it depends on the type of the post-crosslinking agent, but it is usually subjected to the polymerization step (when multistage polymerization was performed, it was used for all the polymerization steps. ) It may be 0 mol per 1 mol of the monomer (that is, the post-crosslinking agent is not used), but the crosslinking density on the surface of the chain water absorbent resin is sufficiently increased and the gel strength of the chain water absorbent resin is increased. From the viewpoint, it is more than 0 and not more than 0.2 mol, preferably more than 0 and not more than 0.1 mol, and more preferably more than 0 and not more than 0.05 mol.
- a chain product of the water absorbent resin can be obtained by distilling off the water and the hydrocarbon dispersion medium by a known method.
- the chain water absorbent resin of the present disclosure can be used by adding, for example, a gel stabilizer, a metal chelating agent, a lubricant, and the like.
- the lubricant include aluminum oxide, titanium oxide, kaolin, talc, bentonite, amorphous silica and the like. It is preferable to add amorphous silica because the manufacturing cost can be suppressed and the water absorption performance is less hindered.
- the amount of amorphous silica added to the water-absorbent resin is, for example, the lower limit value is 0.05 part by mass, preferably 0.1 part by mass, based on the mass of the water-absorbent resin (100 parts by mass). Is.
- the upper limit value is, for example, 2 parts by mass, preferably 1 part by mass.
- the chain-like water absorbent resin according to the present embodiment can have a desired particle size distribution at the time when it is obtained by the above-mentioned manufacturing method, but further perform an operation such as particle size adjustment using classification with a sieve. Therefore, the particle size distribution may be predetermined. Further, in the above-mentioned production method, not only the chain water absorbent resin, but also a non-chain water absorbent resin can be obtained. In this case, it is possible to selectively recover the chain water absorbent resin by utilizing classification. it can.
- the chain-like water absorbent resin of the present disclosure produced as described above is not bulkier than the hydrophilic fiber, when used as a substitute for the hydrophilic fiber, the thickness of the absorbent body and the absorbent article is smaller than that of the conventional one. It can be made thinner. Furthermore, since the chain water-absorbent resin of the present disclosure is a water-absorbent resin, it has a high water-retaining capacity by itself (on the other hand, hydrophilic fibers have almost no water-retaining capacity). Therefore, when the chain water absorbent resin of the present disclosure is used as a substitute for the hydrophilic fiber, the amount of reversion of the absorbent body and the absorbent article can be effectively reduced.
- the chain water absorbent resin of the present disclosure can sufficiently diffuse a liquid.
- Conventional non-chain water-absorbing resin is easy to disperse each non-chain water-absorbing resin in an independent state without contact with other non-chain water-absorbing resin when the absorber is manufactured, and this independent dispersion is absorbed. It is considered to be one of the factors that hinder the diffusion of liquid in the body.
- the chain-shaped water absorbent resin of the present disclosure is elongated, it is easy to disperse each chained water absorbent resin in a state of being in contact with other chained water absorbent resin in the absorber, and therefore, the liquid is sufficiently dispersed. It is considered that the liquid can be diffused, and the shape itself (the shape in which adjacent particles are continuously connected in a distinguishable manner) also contributes to the diffusion of the liquid.
- the chain-like water absorbent resin of the present disclosure produced as described above has a softer feel than the conventional water absorbent resin (non-chain water absorbent resin), when used as a substitute for the conventional water absorbent resin, The touch of the body and the absorbent article can be improved as compared with the conventional ones. That is, it is possible to obtain an absorbent body and an absorbent article that have a softer touch than those of conventional absorbent bodies and absorbent articles.
- the chain water absorbent resin of the present disclosure is also a water absorbent resin and has a sufficiently high water retention capacity. Therefore, even when the chain water-absorbent resin of the present disclosure is used as a substitute for the non-chain water-absorbent resin, the absorbent performance of the conventional absorbent body and absorbent article can be sufficiently exhibited.
- Example 1 A round bottom cylindrical separable flask having an inner diameter of 110 mm and a volume of 2 L was prepared, which was equipped with a reflux condenser, a dropping funnel, a nitrogen gas introduction tube, and a stirring blade having two inclined paddle blades with a blade diameter of 50 mm in two stages. 300 g of n-heptane was placed in this flask as a hydrocarbon dispersion medium, and 0.37 g of a maleic anhydride-modified ethylene-propylene copolymer (trade name: Hiwax 1105A, manufactured by Mitsui Chemicals, Inc.) was used as a polymer-based dispersant.
- a maleic anhydride-modified ethylene-propylene copolymer trade name: Hiwax 1105A, manufactured by Mitsui Chemicals, Inc.
- sucrose stearate (trade name: Ryoto Sugar Ester S-370, HLB value 3, manufactured by Mitsubishi Kagaku Foods Co., Ltd.) was added as a surfactant, and dissolved by heating while stirring at 550 rpm. Then, it cooled to 55 degreeC.
- methoxypolyethylene glycol #400 acrylate After dissolution of the thickener, 99 g (0.20 mol) of methoxypolyethylene glycol #400 acrylate, and 0.046 g of 2,2′-azobis(2-amidinopropane) dihydrochloride, which is an azo compound as a polymerization initiator ( 0.17 mmol), 0.005 g (0.029 mmol) of ethylene glycol diglycidyl ether as an internal cross-linking agent and 79.0 g of ion-exchanged water were added and dissolved to prepare an aqueous monomer solution.
- aqueous monomer solution was added to the above separable flask containing the hydrocarbon dispersion medium in which the polymeric dispersant and the surfactant were dissolved. After thoroughly replacing the inside of the system with nitrogen while stirring the separable flask, the flask was immersed in a water bath at 70° C. to raise the temperature to carry out a polymerization reaction to complete the reaction, thereby obtaining a hydrogel.
- the separable flask was immersed in an oil bath at 125° C. to raise the temperature of the reaction solution, and the hydrous gel was dehydrated by azeotropic distillation of n-heptane and water while refluxing n-heptane. Then, 4.42 g (0.51 mmol) of a 2% aqueous solution of ethylene glycol diglycidyl ether was added to the hydrogel as a post-crosslinking agent, and the mixture was kept at 80° C. for 2 hours, then n-heptane was distilled off and dried. ..
- the obtained polymer was passed through a sieve with openings of 850 ⁇ m and 250 ⁇ m, passed through 850 ⁇ m, and 40.5 g of the polymer remaining on the sieve of 250 ⁇ m was recovered. Thereafter, an amount of amorphous silica (trade name: Carplex #80, manufactured by Evonik Degussa Japan Co., Ltd.) corresponding to 0.2% by mass of the recovered polymer was added to the recovered polymer (40.5 g). -Mixed, and the chain water-absorbent resin according to Example 1 was evaluated.
- amorphous silica trade name: Carplex #80, manufactured by Evonik Degussa Japan Co., Ltd.
- Example 2 In Example 1 above, instead of 99 g (0.20 mol) of methoxypolyethylene glycol #400 acrylate, 135 g (0.20 mol) of methoxypolyethylene glycol #550 acrylate, and 2,2′-azobis as an azo compound as a polymerization initiator. The same procedure as in Example 1 was repeated except that 0.046 g (0.17 mmol) of potassium persulfate was used instead of 0.046 g (0.17 mmol) of (2-amidinopropane) dihydrochloride. Then, 28.2 g of a polymer was recovered.
- Example 2 The amount of amorphous silica corresponding to 0.2% by mass of the recovered polymer as in Example 1 was added to and mixed with the recovered polymer (28.2 g) to give the chain-like water absorption property of Example 2. Acquired as a resin. Then, the chain water-absorbent resin of Example 2 was evaluated according to the evaluation method described below.
- Example 3 In the above-mentioned Example 1, in place of 0.37 g of sucrose stearate as a surfactant, polyglyceric acid ester (trade name: polyglycerate B-100D, HLB value 3, manufactured by Mitsubishi Chemical Foods Co., Ltd.) was used. The same operation as in Example 1 was carried out except that 37 g was used for the polymerization, to recover 35.3 g of a polymer. The amount of amorphous silica corresponding to 0.2% by mass of the recovered polymer as in Example 1 was added to and mixed with the recovered polymer (35.3 g) to give the chain-like water absorption property of Example 3. Acquired as a resin. Then, the chain water absorbent resin of Example 3 was evaluated according to the evaluation method described later.
- polyglyceric acid ester trade name: polyglycerate B-100D, HLB value 3, manufactured by Mitsubishi Chemical Foods Co., Ltd.
- Example 4 In a reactor similar to that of Example 1, 300 g of exosol heptane was placed as a hydrocarbon dispersion medium, and a maleic anhydride-modified ethylene-propylene copolymer (trade name: Hiwax 1105A, Mitsui Chemicals, Inc.) was used as a polymeric dispersant. 0.37 g) and 0.37 g of sucrose stearate (trade name: Ryoto Sugar Ester S-370, HLB value 3, manufactured by Mitsubishi Chemical Foods Co., Ltd.) as a surfactant, and stirred at 400 rpm. After heating and dissolving, the mixture was cooled to 55°C.
- a maleic anhydride-modified ethylene-propylene copolymer trade name: Hiwax 1105A, Mitsui Chemicals, Inc.
- sucrose stearate trade name: Ryoto Sugar Ester S-370, HLB value 3, manufactured by Mitsubishi Chemical Foods Co., Ltd.
- aqueous monomer solution was added to the above separable flask containing the hydrocarbon dispersion medium in which the polymeric dispersant and the surfactant were dissolved. After thoroughly replacing the inside of the system with nitrogen while stirring the separable flask, the flask was immersed in a water bath at 70° C. to raise the temperature to carry out a polymerization reaction to complete the reaction, thereby obtaining a hydrogel.
- Example 4 Subsequent operations were the same as in Example 1, and 44.6 g of a polymer was recovered. The amount of amorphous silica corresponding to 0.2% by mass of the recovered polymer as in Example 1 was added to and mixed with the recovered polymer (44.6 g) to give the chain-like water absorption property of Example 4. Acquired as a resin. Then, the chain water absorbent resin of Example 4 was evaluated according to the evaluation method described later.
- Example 5 416 g of n-heptane was placed as a hydrocarbon dispersion medium in the same reactor as in Example 1, and a maleic anhydride-modified ethylene-propylene copolymer (trade name: Hiwax 1105A, Mitsui Chemicals, Inc.) was used as a polymeric dispersant. 0.37 g) and 0.37 g of sucrose stearate (trade name: Ryoto Sugar Ester S-370, HLB value 3, manufactured by Mitsubishi Chemical Foods Co., Ltd.) as a surfactant, and stirred at 550 rpm. After heating and dissolving, the mixture was cooled to 55°C.
- aqueous monomer solution was added to the above separable flask containing the hydrocarbon dispersion medium in which the polymeric dispersant and the surfactant were dissolved. After thoroughly replacing the inside of the system with nitrogen while stirring the separable flask, the flask was immersed in a water bath at 70° C. to raise the temperature to carry out a polymerization reaction to complete the reaction, thereby obtaining a hydrogel.
- Example 5 Subsequent operations were the same as in Example 1, and 36.9 g of a polymer was recovered. The amount of amorphous silica corresponding to 0.2% by mass of the polymer recovered in the same manner as in Example 1 was added to and mixed with the polymer recovered (36.9 g) to give the chain-like water absorption property of Example 5. Acquired as a resin. Then, the chain water-absorbent resin of Example 5 was evaluated according to the evaluation method described below.
- Example 6 A round bottom cylindrical separable flask having an inner diameter of 110 mm and a volume of 2 L was prepared, which was equipped with a reflux condenser, a dropping funnel, a nitrogen gas introduction tube, and a stirring blade having two inclined paddle blades with a blade diameter of 50 mm in two stages. 300 g of n-heptane was placed in this flask as a hydrocarbon dispersion medium, and 0.37 g of a maleic anhydride-modified ethylene-propylene copolymer (trade name: Hiwax 1105A, manufactured by Mitsui Chemicals, Inc.) was used as a polymer-based dispersant.
- a maleic anhydride-modified ethylene-propylene copolymer trade name: Hiwax 1105A, manufactured by Mitsui Chemicals, Inc.
- sucrose stearate (trade name: Ryoto Sugar Ester S-370, HLB value 3, manufactured by Mitsubishi Kagaku Foods Co., Ltd.) was added as a surfactant, and dissolved by heating while stirring at 550 rpm. Then, it cooled to 55 degreeC.
- the above-mentioned first stage monomer aqueous solution was added to the above separable flask charged with the hydrocarbon dispersion medium in which the polymer dispersant and the surfactant were dissolved. After thoroughly replacing the inside of the system with nitrogen while stirring the separable flask, the flask was immersed in a water bath at 70°C to raise the temperature to carry out the polymerization reaction to complete the reaction, and the first-stage hydrogel was obtained. Obtained.
- the entire amount of the second-stage aqueous monomer solution was added to the first-stage hydrogel, and the system was replaced with nitrogen. While maintaining at 25° C. for 30 minutes. After that, the flask was again immersed in a water bath at 70° C. to raise the temperature to carry out the polymerization reaction to complete the reaction, thereby obtaining a hydrogel of the second stage.
- the separable flask was immersed in an oil bath at 125° C. to raise the temperature of the reaction solution, and the hydrous gel was dehydrated by azeotropic distillation of n-heptane and water while refluxing n-heptane. Then, n-heptane was distilled off and dried.
- the obtained polymer was passed through a sieve with openings of 850 ⁇ m and 250 ⁇ m, passed through 850 ⁇ m, and 58.5 g of the polymer remaining on the sieve of 250 ⁇ m was recovered.
- amorphous silica (trade name: Carplex #80, manufactured by Evonik Degussa Japan Co., Ltd.) corresponding to 0.2% by mass of the recovered polymer was added to the recovered polymer (58.5 g). -Mixed and acquired as the chain-like water absorbent resin of Example 6. Then, the chain water-absorbent resin of Example 6 was evaluated according to the evaluation method described below.
- FIGS. 3(A) to 3(F) SEM images of one chain water absorbent resin contained in the water absorbent resins obtained in Examples 1 to 6 are shown in FIGS. 3(A) to 3(F).
- the SEM image of one chain water absorbent resin contained in the water absorbent resin obtained in Example 1 corresponds to FIG.
- Example 2 is FIG. 3B
- Example 3 is FIG. 3C
- Example 4 is FIG. 3D
- Example 5 is FIG. 3E
- Example 6 is FIG. It corresponds to F).
- SEM images of a plurality of water-absorbent resins randomly obtained from the water-absorbent resin obtained in Example 1 are shown in FIG.
- the physiological saline absorption rate was measured in a room controlled at 25°C ⁇ 1°C. 50 ⁇ 0.1 g of physiological saline in a beaker with a volume of 100 mL was adjusted to a temperature of 25 ⁇ 0.2°C in a constant temperature water bath, and then stirred at 600 rpm with a magnetic stirrer bar (8 mm ⁇ ⁇ 30 mm, no ring). Then a vortex was generated.
- FIG. 5 shows a schematic view of a measuring device used for measuring the water absorption capacity of the water absorbent resin under pressure. It measured using the measuring apparatus 50 shown in FIG.
- the measuring device 50 shown in FIG. 5 includes a buret part 51, a conduit 52, a measuring table 53, and a measuring part 54 placed on the measuring table 53.
- a rubber plug 64 is connected to the upper part of the buret 60, and an air introduction pipe 61 and a cock 62 are connected to the lower part.
- the air introduction pipe 61 has a cock 63 at the tip.
- a conduit 52 is attached between the buret portion 51 and the measurement table 53.
- the inner diameter of the conduit 52 is 6 mm.
- the measuring unit 54 has a cylinder 70 (made of Plexiglas), a nylon mesh 71 adhered to the bottom of the cylinder 70, and a weight 72.
- the inner diameter of the cylinder 70 is 20 mm.
- the opening of the nylon mesh 71 is 75 ⁇ m (200 mesh).
- the water absorbing agent 55 (the water absorbing resin to be measured) is evenly spread on the nylon mesh 71.
- the weight 72 has a diameter of 19 mm and a mass of 60 g. The weight 72 is placed on the water absorbing agent 55, and a load of 2.07 kPa can be applied to the water absorbing agent 55.
- Table 1 shows the results relating to the physiological saline water retention capacity, the physiological saline water absorption rate, and the water absorption capacity under pressure of the water absorbent resins according to Examples 1 to 6.
- test liquid artificial urine
- test liquid artificial urine
- 60 g of sodium chloride 1.8 g of calcium chloride dihydrate and 3.6 g of magnesium chloride hexahydrate.
- 0.15 g of polyoxyethylene nonylphenyl ether was added, and distilled water was further added to make the total mass 6000 g.
- a test solution was prepared by coloring with a small amount of Blue No. 1.
- Non-chain water-absorbing resin (commercial name: AquaKeep SA60S, Sumitomo Seika Chemicals Co., Ltd.), which is composed of particles having a single tuft-like shape and having a tufted shape of grape (Made by the company).
- the non-chain water absorbent resin contains 0.1 part by mass of amorphous silica with respect to 100 parts by mass of the water absorbent resin.
- the non-chain water absorbent resin, the chain water absorbent resin according to Example 1 or 5 and/or crushed pulp (Rayflock manufactured by Leonia Co.) was mixed with air so that the total weight thereof was 20 g.
- a sheet-shaped absorbent core having a size of 40 cm ⁇ 12 cm was produced by uniformly mixing by papermaking.
- an absorber was produced by applying a load of 141 kPa to the whole for 30 seconds and pressing. Further, on the upper surface of the absorbent body, an air-through type porous liquid permeable sheet made of polyethylene and having the same size as the absorbent body and having a basis weight of 22 g/m 2 was arranged to obtain an absorbent article.
- Absorbent articles according to Examples A to D and Comparative Example A were manufactured by changing the composition ratio of the water absorbent resin, the pulp and the chain water absorbent resin as shown in Table 2.
- the water-absorbent resins used in Examples A to D are those according to Example 1 or 5.
- the thickness of the absorbent article was obtained by measuring the central portion of the absorbent article with a thickness meter (model number: JB, manufactured by Ozaki Seisakusho).
- the absorbent article was placed on a horizontal table.
- a measuring instrument equipped with a liquid injection cylinder having an inner diameter of 3 cm is placed in the center of the absorbent article, and 50 mL of artificial urine is introduced into the cylinder at one time (first time).
- the cylinder is removed, the absorbent article is allowed to stand as it is, and the same as the first time after 30 minutes (second time) and 60 minutes (third time) from the start of the first test liquid addition.
- the same operation was performed using a measuring instrument at the position.
- Table 2 shows the composition ratios of the absorbent bodies constituting the absorbent articles according to Examples A to D and Comparative Example A, and the evaluation results of the thickness, the amount of reversion, and the diffusion length.
- the absorbent articles according to Examples A to D can be remarkably thinner than the absorbent article according to Comparative Example A, and the diffusion length is smaller than that of Comparative Example A. Although it was inferior, it was confirmed that the amount of reversion was remarkably reduced, and it was confirmed that the absorbent article had sufficient performance.
- the chain-like water absorbent resin of Example 1 or 5 and the chain-like water absorbent resins of Examples 2 to 4 and 6 have water absorption capacity (physiological saline water retention capacity, physiological saline water absorption rate, And the water absorbency under pressure) are the same, the chain water absorbent resin of Example 1 or 5 used in the absorbent articles of Examples A to D is changed to the chain water absorbent resin of Examples 2 to 4 and 6. Even if it substitutes, it is estimated that the result similar to Table 2 will be obtained.
- tissue paper of 40 cm ⁇ 12 cm (basis weight 16 g/m 2 ) to form an absorbent core
- m 2 tissue paper of 40 cm ⁇ 12 cm (basis weight 16 g/m 2 ).
- test absorbers according to Examples E to G and Comparative Example B were produced.
- the thickness of the test absorber was obtained by measuring the center of the test absorber with a thickness meter (model number: JB, manufactured by Ozaki Seisakusho).
- the test absorber was placed on the test stand of the same device and fixed using a rod-shaped "chuck" or tape.
- the surface characteristics were measured by applying a load of 50 g to a 10 mm ⁇ 10 mm friction terminal wound with a wire having a diameter of 0.5 mm and sliding the surface of the test absorber at a speed of 1 mm/sec. From the measurement result, the variation (MMD) of the friction coefficient was obtained.
- the variation of the friction coefficient (MMD) is an index of roughness, and the larger the value, the more rough the texture is.
- the surface characteristics are measured by applying a load of 10 g to the roughness terminal made of a single wire with a width of 5 mm and sliding the surface of the test absorber at a speed of 1 mm/sec. Then, the surface roughness (SMD) was obtained from the results.
- the surface roughness (SMD) is an index relating to surface irregularities, and a larger value indicates more remarkable surface irregularities.
- the present disclosure has been made in view of the above problems, and an object of the present disclosure is to provide an absorbent body having a soft touch and an absorbent article including the absorbent body.
- the present inventors have found that when the water-absorbent resin used for the absorbent body has a specific shape, the absorbent article can be made soft by combining the water-absorbent resin with pulp to form an absorbent body.
- an absorber includes a water-absorbent resin having a shape in which a plurality of substantially spherical particles are connected in a chain, and a hydrophilic fiber.
- the water-absorbent resin having a shape in which a plurality of substantially spherical particles are linked in a chain shape is softer than conventional water-absorbent resins, and therefore an absorbent body using this water-absorbent resin has a good feel to the skin.
- “good (good) touch” refers to a state in which the variation (MMD) of the friction coefficient representing the frictional characteristics and the surface roughness (SMD) are both small, and the surface is smooth. It means a state in which a nice touch is obtained.
- the water absorbent resin can be a copolymer of two or more types of monomers.
- an absorbent article according to an aspect of the present disclosure includes the above absorber.
- the above-mentioned absorbent article includes an absorbent body containing a water-absorbent resin in a shape in which a plurality of the above-mentioned substantially spherical particles are connected in a chain. Therefore, the absorbent article also has good touch.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Analytical Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Dispersion Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Hematology (AREA)
- Materials Engineering (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Absorbent Articles And Supports Therefor (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Polymerisation Methods In General (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
Abstract
Description
図1は、本開示の一実施形態に係る吸収体を含む吸収性物品の構成を示す断面図である。図1に示す吸収性物品1は、吸収体2と、液体透過性シート3と、液体不透過性シート4と、を含む。吸収体2は、対向配置された液体透過性シート3及び液体不透過性シート4の間に、挟まれている。
本実施形態に係る吸水性樹脂21に含まれる鎖状吸水性樹脂211について説明する。図2は、鎖状吸水性樹脂211の一例を示している。図2に示すように、鎖状吸水性樹脂211は、複数の略球状の粒子40が鎖状に連結された形状を有する。
鎖状吸水性樹脂211の製造方法の一例について説明する。鎖状吸水性樹脂211は、例えば、2種類以上の単量体を共重合させることで作製し得る。具体的には、鎖状吸水性樹脂211の製造方法は、単量体A及び単量体B(単量体Aと種類が異なる)を逆相懸濁重合法により共重合させる重合工程と、重合工程において得られた含水ゲルを乾燥する乾燥工程と、を含む。
本実施形態では、重合工程に供される単量体Aは、(メタ)アクリル酸及びその中和物のうち少なくとも一方を含んでおり、単量体Bは、アルコキシポリアルキレングリコール(メタ)アクリレートを含んでいる。なお、本明細書では、「アクリル」及び「メタクリル」を合わせて「(メタ)アクリル」と表記する。
引き続き、得られた含水ゲルより水分を除去するために、乾燥を行なう。乾燥方法としては、例えば(a)上記含水ゲルが炭化水素分散媒に分散した状態で、外部から加熱することにより共沸蒸留を行い、炭化水素分散媒を還流させて水分を除去(脱水)する方法、(b)デカンテーションにより含水ゲルを取り出し、減圧乾燥する方法、(c)フィルタにより含水ゲルをろ別し、減圧乾燥する方法等が挙げられる。このうち、鎖状吸水性樹脂を効率的に得るには、(a)の方法を用いることが好ましい。また、炭化水素分散剤の共沸蒸留による脱水によって、含水ゲルの含水量を50質量%程度まで低下させることで、鎖状吸水性樹脂を効率的に得ることができる。なお、含水ゲルの含水量が高い状態で、炭化水素分散媒を先に揮発させて乾燥させると、粒子が塊状化して鎖状吸水性樹脂が得られ難くなる可能性が高い。
含水量=[Ww÷(Ww+Ws)]×100
Ww:全重合工程の重合前の水性液に含まれる水分量から、乾燥工程により外部に抽出された水分量を差し引いた量に、後架橋剤等を混合する際に必要に応じて用いられる水分量を加えた含水ゲルの水分量
Ws:含水ゲルを構成する単量体、架橋剤、開始剤等の材料の仕込量から算出される固形分量
(実施例1)
還流冷却器、滴下ロート、窒素ガス導入管、及び、翼径50mmの4枚傾斜パドル翼を2段で有する攪拌翼を備えた内径110mmで容積2Lの丸底円筒型セパラブルフラスコを準備した。このフラスコに、炭化水素分散媒としてn-ヘプタン300gを入れ、高分子系分散剤として無水マレイン酸変性エチレン-プロピレン共重合体(商品名:ハイワックス1105A、三井化学株式会社製)0.37g、及び、界面活性剤としてショ糖ステアリン酸エステル(商品名:リョートーシュガーエステルS-370、HLB値3、三菱化学フーズ株式会社製)0.37gを添加し、550rpmで攪拌しつつ加温溶解した後、55℃まで冷却した。
上記実施例1において、メトキシポリエチレングリコール#400アクリレート99g(0.20モル)に代えてメトキシポリエチレングリコール#550アクリレート135g(0.20モル)、重合開始剤としてアゾ系化合物として2,2’-アゾビス(2-アミジノプロパン)二塩酸塩0.046g(0.17ミリモル)に代えて過硫酸カリウム0.046g(0.17ミリモル)を使用して重合した以外は、実施例1と同様の操作を行い、重合物28.2gを回収した。実施例1と同様に回収した重合物の0.2質量%に相当する量の非晶質シリカを、回収した重合物(28.2g)に添加・混合し、実施例2の鎖状吸水性樹脂として取得した。その後、後述する評価方法に従って、実施例2の鎖状吸水性樹脂の評価を行った。
上記実施例1において、界面活性剤としてのショ糖ステアリン酸エステル0.37gの代わりにポリグリセリン酸エステル(商品名:ポリグリエステルB-100D、HLB値3、三菱化学フーズ株式会社製)0.37gを使用して重合した以外は、実施例1と同様の操作を行い、重合物35.3gを回収した。実施例1と同様に回収した重合物の0.2質量%に相当する量の非晶質シリカを、回収した重合物(35.3g)に添加・混合し、実施例3の鎖状吸水性樹脂として取得した。その後、後述する評価方法に従って、実施例3の鎖状吸水性樹脂の評価を行った。
実施例1と同様の反応装置に、炭化水素分散媒としてエクソールヘプタン300gを入れ、高分子系分散剤として無水マレイン酸変性エチレン-プロピレン共重合体(商品名:ハイワックス1105A、三井化学株式会社製)0.37g、及び、界面活性剤としてショ糖ステアリン酸エステル(商品名:リョートーシュガーエステルS-370、HLB値3、三菱化学フーズ株式会社製)0.37gを添加し、400rpmで攪拌しつつ加温溶解した後、55℃まで冷却した。
実施例1と同様の反応装置に、炭化水素分散媒としてn-ヘプタン416gを入れ、高分子系分散剤として無水マレイン酸変性エチレン-プロピレン共重合体(商品名:ハイワックス1105A、三井化学株式会社製)0.37g、及び、界面活性剤としてショ糖ステアリン酸エステル(商品名:リョートーシュガーエステルS-370、HLB値3、三菱化学フーズ株式会社製)0.37gを添加し、550rpmで攪拌しつつ加温溶解した後、55℃まで冷却した。
還流冷却器、滴下ロート、窒素ガス導入管、及び、翼径50mmの4枚傾斜パドル翼を2段で有する攪拌翼を備えた内径110mmで容積2Lの丸底円筒型セパラブルフラスコを準備した。このフラスコに、炭化水素分散媒としてn-ヘプタン300gを入れ、高分子系分散剤として無水マレイン酸変性エチレン-プロピレン共重合体(商品名:ハイワックス1105A、三井化学株式会社製)0.37g、及び、界面活性剤としてショ糖ステアリン酸エステル(商品名:リョートーシュガーエステルS-370、HLB値3、三菱化学フーズ株式会社製)0.37gを添加し、550rpmで攪拌しつつ加温溶解した後、55℃まで冷却した。
(生理食塩水保水能)
容積500mLのビーカーに、0.9質量%塩化ナトリウム水溶液(生理食塩水)500gを量り取り、マグネチックスターラーバー(8mmφ×30mmのリング無し)を用いて600r/minで撹拌させながら、実施例1~5のそれぞれの吸水性樹脂2.0gを、ママコ(ダマ)が発生しないように分散させた。撹拌させた状態で30分間放置し、吸水性樹脂を十分に膨潤させた。その後、綿袋(メンブロード60番、横100mm×縦200mm)中に注ぎ込み、綿袋の上部を輪ゴムで縛り、遠心力が167Gとなるよう設定した脱水機(株式会社コクサン社製、品番:H-122)を用いて綿袋を1分間脱水し、脱水後の膨潤ゲルを含んだ綿袋の質量Wa(g)を測定した。吸水性樹脂を添加せずに同様の操作を行い、綿袋の湿潤時の空質量Wb(g)を測定し、以下の式から各実施例の吸水性樹脂の保水能を算出した。
生理食塩水保水能(g/g)=[Wa-Wb](g)/吸水性樹脂の質量(g)
生理食塩水吸水速度は、25℃±1℃に調節した室内で測定した。容積100mLのビーカー内に入れた生理食塩水50±0.1gを恒温水槽にて25±0.2℃の温度に調整したのち、マグネチックスターラーバー(8mmφ×30mm、リング無し)で600rpmに攪拌して渦を発生させた。吸水性樹脂2.0±0.002gを、上記生理食塩水中に一度に添加し、吸水性樹脂の添加後から、渦が消失し液面が平坦になるまでの時間(秒)を測定し、当該時間を吸水性樹脂の生理食塩水吸水速度とした。
吸水性樹脂の加圧下吸水能測定に使用した測定装置の模式図を図5に示す。図5に示す測定装置50を用いて測定した。図5に示す測定装置50は、ビュレット部51、導管52、測定台53、及び、測定台53上に置かれた測定部54からなる。ビュレット部51は、ビュレット60の上部にゴム栓64が、下部に空気導入管61及びコック62が、それぞれ連結されている。空気導入管61は先端にコック63を有している。ビュレット部51と測定台53の間には、導管52が取り付けられている。導管52の内径は6mmである。測定台53の中央部には、直径2mmの穴があいており、導管52が連結されている。測定部54は、円筒70(プレキシグラス製)と、この円筒70の底部に接着されたナイロンメッシュ71と、重り72とを有している。円筒70の内径は、20mmである。ナイロンメッシュ71の目開きは、75μm(200メッシュ)である。そして、測定時にはナイロンメッシュ71上に吸水剤55(測定対象の吸水性樹脂)が均一に撒布されている。重り72は、直径19mm、質量60gである。この重り72は、吸水剤55上に置かれ、吸水剤55に対して2.07kPaの荷重を加えることができるようになっている。
加圧下吸水能(ml/g)=(VB-VA)/0.1
(1)試験液(人工尿)の調整
容積10Lの容器に適量の蒸留水を入れ、塩化ナトリウム60g、塩化カルシウム・二水和物1.8g及び塩化マグネシウム・六水和物3.6gを添加し、溶解した。次に、ポリオキシエチレンノニルフェニルエーテル0.15gを添加し、さらに蒸留水を追加して、全体の質量を6000gとした。さらに、少量の青色1号で着色して、試験液を調製した。
単粒子状が凝集し、ブドウの房状の形状を有した粒子により構成される非鎖状吸水性樹脂(商品名:アクアキープSA60S、住友精化株式会社製)を用意した。なお、非鎖状吸水性樹脂には、吸水性樹脂100質量部に対し0.1質量部の非晶質シリカが含まれている。
上記の吸収性物品の中心部を厚み測定器(型番:J-B、尾崎製作所社製)で測定することにより吸収性物品の厚みを得た。
水平の台上に吸収性物品を置いた。吸収性物品の中心部に、内径3cmの液投入用シリンダーを具備した測定器具を置き、50mLの人工尿をそのシリンダー内に一度に投入する(1回目)。次に、前記シリンダーをはずし、吸収性物品をそのままの状態で静置し、1回目の試験液投入開始から30分後(2回目)及び60分後(3回目)にも、1回目と同じ位置に測定器具を用いて同様の操作を行った。3回目の投入から60分経過後、吸収性物品上の試験液投入位置付近に、あらかじめ質量(Wc(g))を測定しておいた10cm四方の濾紙を40枚置き、その上に底面が10cm×10cmの質量5kgの重りを載せた。5分間の荷重後、濾紙の質量(Wd(g))を測定し、増加した質量を逆戻り量(g)とした。逆戻り量が小さいほど、吸収性物品として好ましいと言える。
逆戻り量(g)=Wd-Wc
前記逆戻り量の測定後5分以内に試験液が浸透した吸収性物品の長手方向の拡がり寸法(cm)を測定した。拡散長は短手方向の中央と左端と右端の3箇所で測定し、その平均の数値とした。なお、小数点以下の数値は四捨五入した。
(1)吸収体及び吸収性物品の作製
単粒子状が凝集し、ブドウの房状の形状を有した粒子により構成される非鎖状吸水性樹脂(商品名:アクアキープSA60S、住友精化株式会社製)、解砕パルプ(レオニア社製レイフロック)、及び、実施例1の吸水性樹脂(鎖状吸水性樹脂)を用意した。これらを表3に記載の構成割合での合計重量8gとなるように空気抄造によって均一混合した。その後、40cm×12cmの大きさのティッシュッペーパー(坪量16g/m2)にこれを均一散布させて吸収体コアを形成し、さらに40cm×12cmの大きさのティッシュッペーパー(坪量16g/m2)を重ねた。上下2枚のティッシュペーパーがコアラップに相当する。
上記の試験用吸収体の中心部を厚み測定器(型番:J-B、尾崎製作所社製)で測定することにより試験用吸収体の厚みを得た。
肌触りに係る特性の評価を目的として、自動化表面試験機KES-FB4-AUTO-A(カトーテック株式会社製)を用いて試験用吸収体の摩擦係数の変動(MMD)及び表面粗さ(SMD)を測定した。
従来の吸水性樹脂を用いた吸収体、及びこの吸収体を用いた吸収性物品には、肌触りの観点で改善の余地がある。それは、従来の吸水性樹脂自体の表面が硬いため、吸収体のパルプを減らして薄型化していく際にそれら吸水性樹脂の凹凸を感じやすくなることである。
Claims (10)
- 略球状の複数の粒子が鎖状に連結された形状を有する、吸水性樹脂。
- 2種類以上の単量体の共重合物である、請求項1に記載の吸水性樹脂。
- 請求項1または2に記載の吸水性樹脂を含む、吸収体。
- 略球状の複数の粒子が鎖状に連結された形状を有する吸水性樹脂と、親水性繊維と、を含む、吸収体。
- 前記吸水性樹脂は、2種類以上の単量体の共重合物である、請求項4に記載の吸収体。
- 請求項4または5に記載の吸収体を含む、吸収性物品。
- 単量体A及び単量体Bを逆相懸濁重合法により共重合させる重合工程と、
前記重合工程において得られた含水ゲルを乾燥する乾燥工程と、を有し、
前記単量体Aは、(メタ)アクリル酸及びその中和物のうち少なくとも一方を含み、
前記単量体Bは、アルコキシポリアルキレングリコール(メタ)アクリレートを含み、
前記重合工程において、共重合に供する前記単量体Aのモル量(α)と前記単量体Bのモル量(β)の比(α/β)が、1/4~4である、吸水性樹脂の製造方法。 - 前記重合工程において、界面活性剤としてショ糖脂肪酸エステル及びポリグリセリン脂肪酸エステルのうち少なくとも一方を反応系に添加する、請求項7に記載の吸水性樹脂の製造方法。
- 前記重合工程において、高分子系分散剤の存在下に重合を行う、請求項7または8に記載の吸水性樹脂の製造方法。
- 前記乾燥工程において、炭化水素分散媒の共沸蒸留により、含水量が50質量%以下となるまで前記含水ゲルを脱水する、請求項7~9の何れか一項に記載の吸水性樹脂の製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17/413,783 US20220008894A1 (en) | 2018-12-17 | 2019-12-02 | Water absorbent resin, absorbent body, absorbent article, and production method for water absorbent resin |
| CN201980083305.4A CN113195548A (zh) | 2018-12-17 | 2019-12-02 | 吸水性树脂、吸收体、吸收性物品及吸水性树脂的制造方法 |
| KR1020217020719A KR102666774B1 (ko) | 2018-12-17 | 2019-12-02 | 흡수성 수지, 흡수체, 흡수성 물품, 및 흡수성 수지의 제조 방법 |
| JP2020561261A JP7233443B2 (ja) | 2018-12-17 | 2019-12-02 | 吸水性樹脂、吸収体、吸収性物品、及び吸水性樹脂の製造方法 |
| EP19900702.2A EP3901182A4 (en) | 2018-12-17 | 2019-12-02 | WATER-ABSORBENT RESIN, ABSORBENT BODY, ABSORBENT ARTICLE AND METHOD FOR PRODUCTION OF WATER-ABSORBENT RESIN |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018235789 | 2018-12-17 | ||
| JP2018-235789 | 2018-12-17 | ||
| JP2018235785 | 2018-12-17 | ||
| JP2018-235785 | 2018-12-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020129594A1 true WO2020129594A1 (ja) | 2020-06-25 |
Family
ID=71101432
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/047068 WO2020129594A1 (ja) | 2018-12-17 | 2019-12-02 | 吸水性樹脂、吸収体、吸収性物品、及び吸水性樹脂の製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20220008894A1 (ja) |
| EP (1) | EP3901182A4 (ja) |
| JP (1) | JP7233443B2 (ja) |
| KR (1) | KR102666774B1 (ja) |
| CN (1) | CN113195548A (ja) |
| WO (1) | WO2020129594A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023189622A1 (ja) * | 2022-03-30 | 2023-10-05 | 住友精化株式会社 | 吸水性樹脂粒子、吸収体、及び吸収性物品 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07102006A (ja) * | 1993-08-13 | 1995-04-18 | Nippon Chem Ind Co Ltd | 懸濁重合用安定剤の製造方法 |
| JP2005015787A (ja) * | 2003-06-03 | 2005-01-20 | Nippon Shokubai Co Ltd | 吸水材の製造方法 |
| JP2005097593A (ja) * | 2003-09-02 | 2005-04-14 | Nippon Shokubai Co Ltd | 粒子状吸水性樹脂組成物 |
| JP2007197873A (ja) * | 2006-01-27 | 2007-08-09 | Mitsubishi Chemicals Corp | 吸液性複合体及びその製造方法、吸液性物品並びにノズル |
| JP2009084472A (ja) | 2007-10-01 | 2009-04-23 | San-Dia Polymer Ltd | 吸収性樹脂粒子、この製造方法及び吸収性物品 |
| JP2016028117A (ja) * | 2014-07-11 | 2016-02-25 | 住友精化株式会社 | 吸水性樹脂の製造方法、吸水性樹脂、吸水剤、吸収性物品 |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01249808A (ja) * | 1988-03-31 | 1989-10-05 | Nippon Shokubai Kagaku Kogyo Co Ltd | 耐塩性吸水性樹脂微粒子の製造方法 |
| JPH02115201A (ja) * | 1988-10-25 | 1990-04-27 | Nippon Shokubai Kagaku Kogyo Co Ltd | 逆相懸濁重合用分散剤およびそれを用いてなる親水性樹脂の製造方法 |
| JP2802092B2 (ja) * | 1989-03-28 | 1998-09-21 | 株式会社日本触媒 | 優れた耐塩性を有する吸水剤 |
| JP2742447B2 (ja) * | 1989-06-27 | 1998-04-22 | 株式会社日本触媒 | 逆相懸濁重合用分散剤および該分散剤を用いた親水性重合体の製造方法 |
| KR100269980B1 (ko) * | 1994-10-26 | 2000-10-16 | 다나카 쇼소 | 흡수성수지조성물및그제조방법 |
| JP3970818B2 (ja) | 1994-10-26 | 2007-09-05 | 株式会社日本触媒 | 吸水性樹脂の造粒粒子およびこれを含む吸収性物品ならびに吸水性樹脂の造粒粒子の製造方法 |
| FR2732973B1 (fr) * | 1995-04-11 | 1997-06-13 | Atochem Elf Sa | Procede pour l'obtention de polymeres superabsorbants pour l'eau et les fluides aqueux sous forme d'agregats de particules |
| EP1142696A4 (en) * | 1998-11-06 | 2003-04-16 | Mitsubishi Chem Corp | WATER ABSORBING COMPOSITE, PROCESS FOR PREPARING THE SAME, AND WATER ABSORBING ARTICLE |
| JP2000198805A (ja) | 1998-11-06 | 2000-07-18 | Mitsubishi Chemicals Corp | 吸水性複合体およびその製造方法 |
| JP2002284803A (ja) | 2001-03-27 | 2002-10-03 | Mitsubishi Chemicals Corp | 高吸水性樹脂の製造方法及び高吸水性樹脂 |
| US20090036855A1 (en) * | 2004-08-06 | 2009-02-05 | Katsuyuki Wada | Particulate water-absorbing agent with water-absorbing resin as main component, method for production of the same, and absorbing article |
| WO2007116554A1 (ja) * | 2006-03-31 | 2007-10-18 | Asahi Kasei Chemicals Corporation | 吸水性樹脂粒子凝集体及びその製造方法 |
| JP5558096B2 (ja) * | 2007-03-01 | 2014-07-23 | 株式会社日本触媒 | 吸水性樹脂を主成分とする粒子状吸水剤 |
| JP5805639B2 (ja) * | 2010-07-28 | 2015-11-04 | 住友精化株式会社 | 吸水性樹脂の製造方法 |
| JP5557386B2 (ja) | 2010-07-30 | 2014-07-23 | 矢崎エナジーシステム株式会社 | 車両用情報記録システム及び車両用情報記録方法 |
| SG187197A1 (en) * | 2010-09-06 | 2013-02-28 | Sumitomo Seika Chemicals | Water absorbent resin and method for producing same |
| JP6120489B2 (ja) * | 2012-03-23 | 2017-04-26 | 住友精化株式会社 | スプレー噴霧用水性組成物 |
| KR101684649B1 (ko) * | 2014-06-13 | 2016-12-08 | 주식회사 엘지화학 | 고흡수성 수지의 제조 방법 및 이를 통해 제조된 고흡수성 수지 |
| JP5719078B1 (ja) * | 2014-07-11 | 2015-05-13 | 住友精化株式会社 | 吸水性樹脂の製造方法 |
| JP5766344B1 (ja) * | 2014-07-11 | 2015-08-19 | 住友精化株式会社 | 吸水性樹脂及び吸収性物品 |
| WO2018159803A1 (ja) * | 2017-03-02 | 2018-09-07 | 住友精化株式会社 | 吸水性樹脂及び土嚢 |
-
2019
- 2019-12-02 EP EP19900702.2A patent/EP3901182A4/en active Pending
- 2019-12-02 JP JP2020561261A patent/JP7233443B2/ja active Active
- 2019-12-02 KR KR1020217020719A patent/KR102666774B1/ko active IP Right Grant
- 2019-12-02 US US17/413,783 patent/US20220008894A1/en active Pending
- 2019-12-02 WO PCT/JP2019/047068 patent/WO2020129594A1/ja unknown
- 2019-12-02 CN CN201980083305.4A patent/CN113195548A/zh active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07102006A (ja) * | 1993-08-13 | 1995-04-18 | Nippon Chem Ind Co Ltd | 懸濁重合用安定剤の製造方法 |
| JP2005015787A (ja) * | 2003-06-03 | 2005-01-20 | Nippon Shokubai Co Ltd | 吸水材の製造方法 |
| JP2005097593A (ja) * | 2003-09-02 | 2005-04-14 | Nippon Shokubai Co Ltd | 粒子状吸水性樹脂組成物 |
| JP2007197873A (ja) * | 2006-01-27 | 2007-08-09 | Mitsubishi Chemicals Corp | 吸液性複合体及びその製造方法、吸液性物品並びにノズル |
| JP2009084472A (ja) | 2007-10-01 | 2009-04-23 | San-Dia Polymer Ltd | 吸収性樹脂粒子、この製造方法及び吸収性物品 |
| JP2016028117A (ja) * | 2014-07-11 | 2016-02-25 | 住友精化株式会社 | 吸水性樹脂の製造方法、吸水性樹脂、吸水剤、吸収性物品 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3901182A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023189622A1 (ja) * | 2022-03-30 | 2023-10-05 | 住友精化株式会社 | 吸水性樹脂粒子、吸収体、及び吸収性物品 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20220008894A1 (en) | 2022-01-13 |
| JP7233443B2 (ja) | 2023-03-06 |
| EP3901182A1 (en) | 2021-10-27 |
| EP3901182A4 (en) | 2022-10-05 |
| CN113195548A (zh) | 2021-07-30 |
| JPWO2020129594A1 (ja) | 2021-10-28 |
| KR102666774B1 (ko) | 2024-05-16 |
| KR20210100141A (ko) | 2021-08-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10265226B2 (en) | Water-absorbent resin, water-absorbent material, and water-absorbent article | |
| JP5901524B2 (ja) | 吸水性樹脂 | |
| JP5893117B2 (ja) | 吸水性樹脂及び吸収性物品 | |
| WO2012144564A1 (ja) | 吸水性樹脂、吸収体及び吸収性物品 | |
| WO2012132902A1 (ja) | 吸水性樹脂の製造方法 | |
| JP7429121B2 (ja) | 吸水性樹脂 | |
| WO2019189445A1 (ja) | 吸収性物品 | |
| WO2018159802A1 (ja) | 吸水性樹脂及び吸収性物品 | |
| EP3896097A1 (en) | Water-absorptive resin particle, absorption body, and absorptive article | |
| JP7538107B2 (ja) | 吸水性樹脂粒子及びその製造方法、吸収体、並びに吸収性物品 | |
| WO2020184398A1 (ja) | 吸水性樹脂粒子及びその製造方法、吸収体、並びに、吸収性物品 | |
| WO2020129594A1 (ja) | 吸水性樹脂、吸収体、吸収性物品、及び吸水性樹脂の製造方法 | |
| WO2020122219A1 (ja) | 吸水性樹脂粒子、吸収体、吸収性物品、及び液吸引力測定方法 | |
| JP7194197B2 (ja) | 吸収体及び吸収性物品 | |
| WO2020122211A1 (ja) | 吸水性樹脂粒子 | |
| WO2022210678A1 (ja) | 吸水性樹脂、吸収体及び吸収性物品 | |
| WO2023189672A1 (ja) | 吸水性樹脂粒子及び吸収性物品 | |
| JP7143513B2 (ja) | 吸水性樹脂粒子及びその製造方法、吸収体、並びに、吸収性物品 | |
| JP7091556B2 (ja) | 吸水性樹脂粒子、及び吸水シート | |
| WO2020095811A1 (ja) | 吸水性樹脂 | |
| WO2022255300A1 (ja) | 吸水性樹脂の製造方法、吸水性樹脂、吸収体及び吸収性物品 | |
| EP4186930A1 (en) | Water-absorbing resin composition, absorbent, and absorptive article | |
| WO2023176644A1 (ja) | 吸水性樹脂粒子及び吸収性物品 | |
| KR20230008055A (ko) | 입자상 흡수성 수지 조성물 및 그 제조 방법, 흡수체, 및 흡수성 물품 | |
| JP2020093064A (ja) | 吸水性樹脂粒子、吸水性樹脂粒子の液体漏れ性の評価方法、及び吸水性樹脂粒子の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19900702 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020561261 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20217020719 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019900702 Country of ref document: EP Effective date: 20210719 |