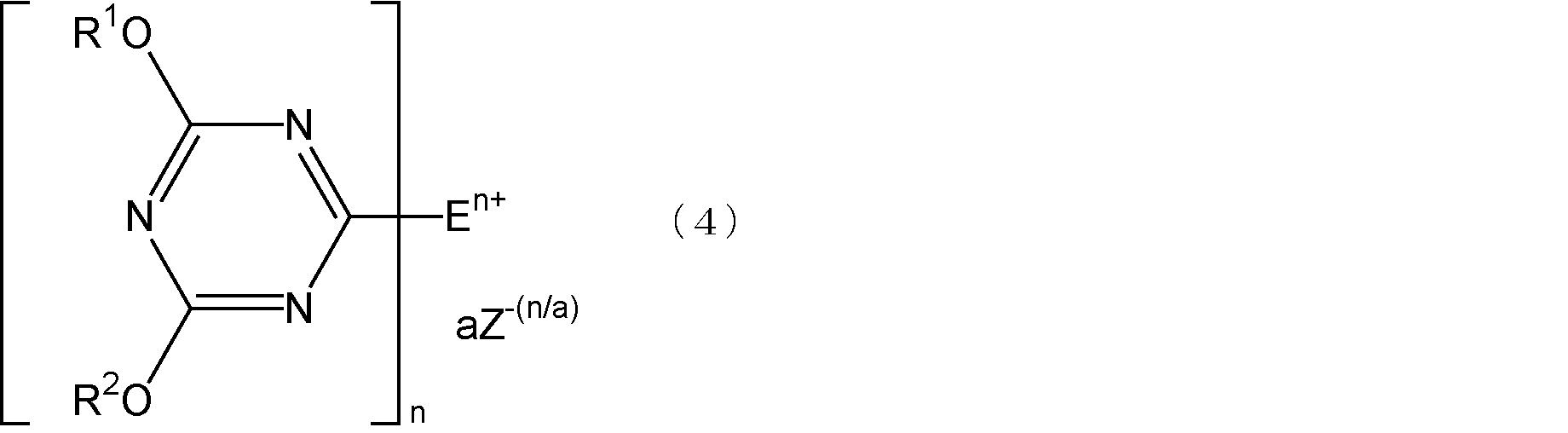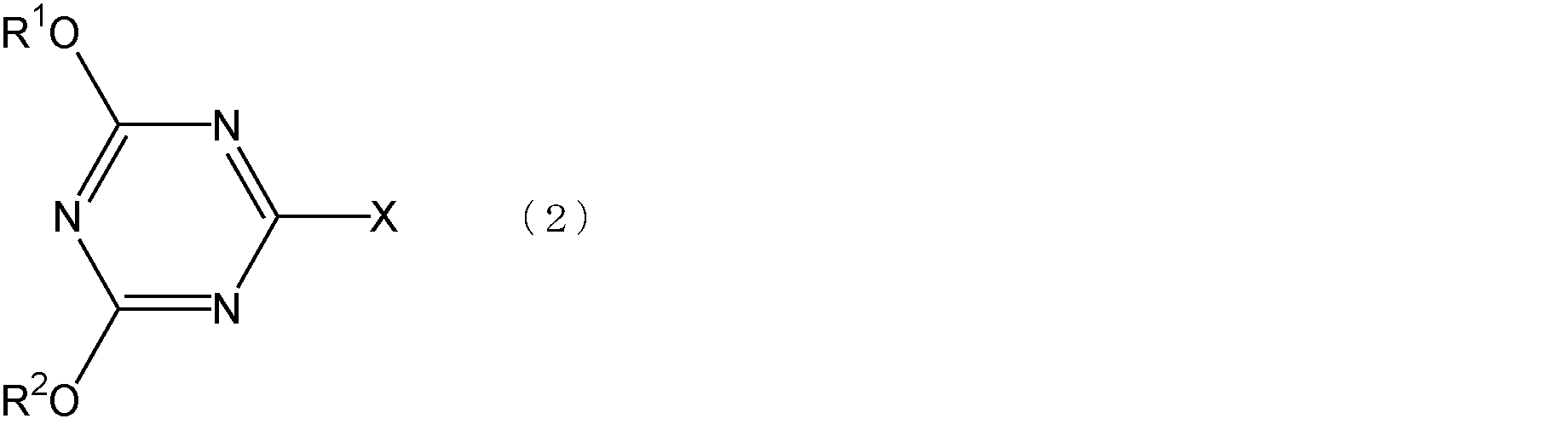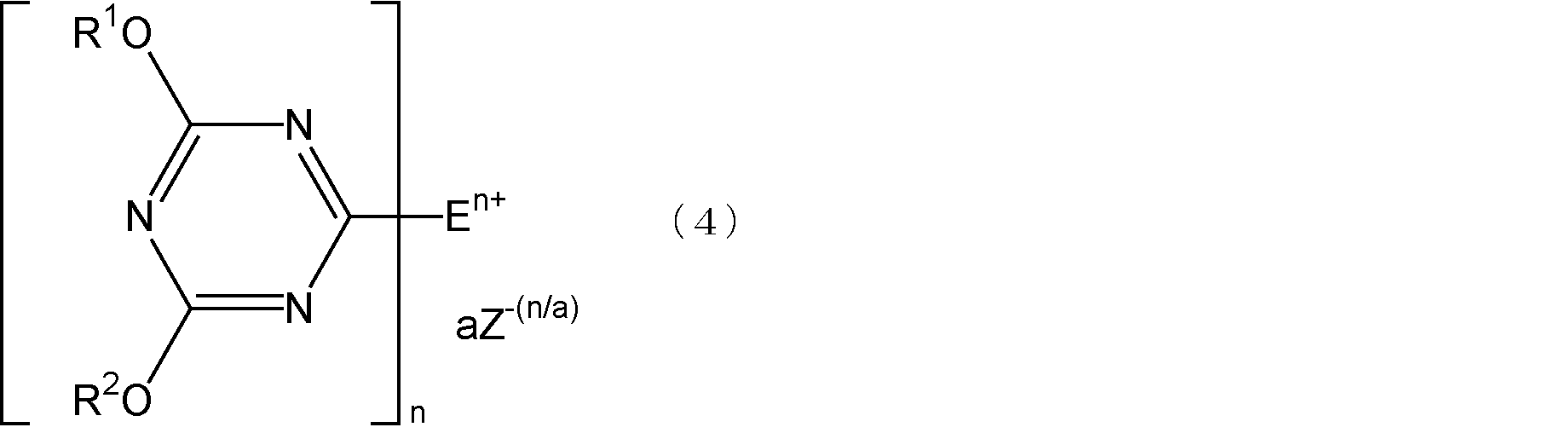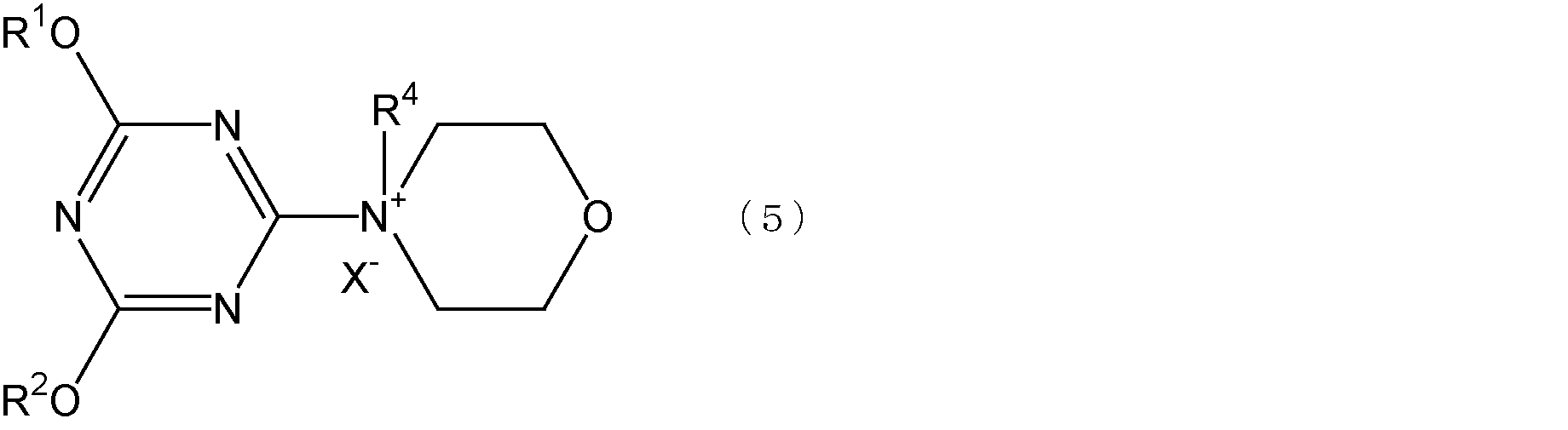WO2014045921A1 - ポリロタキサンの製造方法 - Google Patents
ポリロタキサンの製造方法 Download PDFInfo
- Publication number
- WO2014045921A1 WO2014045921A1 PCT/JP2013/074247 JP2013074247W WO2014045921A1 WO 2014045921 A1 WO2014045921 A1 WO 2014045921A1 JP 2013074247 W JP2013074247 W JP 2013074247W WO 2014045921 A1 WO2014045921 A1 WO 2014045921A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- polyrotaxane
- group
- pseudopolyrotaxane
- Prior art date
Links
- 0 [*+]C(*)(*)c1nc(O)nc(O)n1 Chemical compound [*+]C(*)(*)c1nc(O)nc(O)n1 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G81/00—Macromolecular compounds obtained by interreacting polymers in the absence of monomers, e.g. block polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B37/00—Preparation of polysaccharides not provided for in groups C08B1/00 - C08B35/00; Derivatives thereof
- C08B37/0006—Homoglycans, i.e. polysaccharides having a main chain consisting of one single sugar, e.g. colominic acid
- C08B37/0009—Homoglycans, i.e. polysaccharides having a main chain consisting of one single sugar, e.g. colominic acid alpha-D-Glucans, e.g. polydextrose, alternan, glycogen; (alpha-1,4)(alpha-1,6)-D-Glucans; (alpha-1,3)(alpha-1,4)-D-Glucans, e.g. isolichenan or nigeran; (alpha-1,4)-D-Glucans; (alpha-1,3)-D-Glucans, e.g. pseudonigeran; Derivatives thereof
- C08B37/0012—Cyclodextrin [CD], e.g. cycle with 6 units (alpha), with 7 units (beta) and with 8 units (gamma), large-ring cyclodextrin or cycloamylose with 9 units or more; Derivatives thereof
- C08B37/0015—Inclusion compounds, i.e. host-guest compounds, e.g. polyrotaxanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/32—Polymers modified by chemical after-treatment
- C08G65/329—Polymers modified by chemical after-treatment with organic compounds
- C08G65/331—Polymers modified by chemical after-treatment with organic compounds containing oxygen
- C08G65/332—Polymers modified by chemical after-treatment with organic compounds containing oxygen containing carboxyl groups, or halides, or esters thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/32—Polymers modified by chemical after-treatment
- C08G65/329—Polymers modified by chemical after-treatment with organic compounds
- C08G65/333—Polymers modified by chemical after-treatment with organic compounds containing nitrogen
- C08G65/33303—Polymers modified by chemical after-treatment with organic compounds containing nitrogen containing amino group
- C08G65/33306—Polymers modified by chemical after-treatment with organic compounds containing nitrogen containing amino group acyclic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G83/00—Macromolecular compounds not provided for in groups C08G2/00 - C08G81/00
- C08G83/007—Polyrotaxanes; Polycatenanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L5/00—Compositions of polysaccharides or of their derivatives not provided for in groups C08L1/00 or C08L3/00
- C08L5/16—Cyclodextrin; Derivatives thereof
Definitions
- the present invention relates to a method for producing a polyrotaxane.
- the crosslinked polyrotaxane can be obtained by crosslinking a plurality of polyrotaxanes having blocking groups introduced at both ends of the pseudopolyrotaxane.
- the pseudopolyrotaxane comprises a compound having a polyethylene glycol (hereinafter also referred to as “PEG”) chain (hereinafter also referred to as “PEG compound”) and a cyclodextrin that includes the PEG compound
- the resulting crosslinked polyrotaxane is The cyclodextrin penetrated by the PEG chain of the PEG compound can move along the PEG chain by the so-called pulley effect, so even if tension is applied, the tension is evenly dispersed by the pulley effect Can do. Therefore, the cross-linked polyrotaxane has excellent characteristics not found in conventional cross-linked polymers, such as being less prone to cracks and scratches.
- Patent Document 1 discloses that a pseudopolyrotaxane aqueous dispersion is prepared by dissolving carboxylated polyethylene glycol in which both ends of a PEG chain are carboxylated and ⁇ -cyclodextrin in warm water at 70 ° C., and then mixing and cooling both.
- the aqueous dispersion was freeze-dried to produce a pseudopolyrotaxane, and the obtained pseudopolyrotaxane was used as a condensing agent in dimethylformamide as a BOP reagent (benzotriazol-1-yl-oxy-tris (dimethylamino) phosphonium.
- BOP reagent benzotriazol-1-yl-oxy-tris (dimethylamino) phosphonium.
- Hexafluorophosphate a process for producing a polyrotaxane that is reacted with adamantaneamine in the presence of diisopropylethylamine as
- the pseudo-polyrotaxane used for the production of the polyrotaxane is usually obtained by mixing a PEG compound and cyclodextrin in an aqueous medium, and thus is obtained as an aqueous dispersion.
- a polyrotaxane by introducing a blocking group to both ends of the pseudo-polyrotaxane by an efficient and chemically stable bond, both ends of the PEG chain are made carboxyl groups, and the blocking group is reacted with the carboxyl group. It is effective to react as a group such as an amino group or a hydroxyl group.
- water in the system deactivates the reaction.
- the pseudopolyrotaxane aqueous dispersion is solidified by centrifugation, filtration, or the like. After liquid separation, it is necessary to sufficiently remove water by a method of drying treatment or a method of drying the pseudopolyrotaxane aqueous dispersion as it is.
- water that inhibits the reaction of introducing blocking groups at both ends of the PEG chain needs to be sufficiently removed by drying, and thus requires a very long drying time.
- An object of this invention is to provide the manufacturing method of the polyrotaxane which can manufacture the polyrotaxane which has a high inclusion rate by an industrially advantageous method.
- the present invention includes a cyclodextrin, a compound having a repeating structural unit represented by the following formula (1), which is embedded in a skewered manner in the opening of the cyclodextrin, and the formula (1).
- a compound having a repeating structural unit represented by the formula (1) which is a method for producing a polyrotaxane having a blocking group for preventing elimination of cyclodextrin, disposed at both ends of a compound having a repeating structural unit Pseudopolyrotaxane aqueous solution containing pseudopolyrotaxane particles mixed with cyclodextrin in an aqueous medium, and a compound having a repeating structural unit represented by the above formula (1) included in the opening of the cyclodextrin molecule in a skewered manner Step 1 for obtaining a dispersion, a triazine compound represented by the following formula (2), a compound having at least one tertiary amino group, and the step
- R 1 and R 2 are each independently an alkyl group having 1 to 4 carbon atoms or an aryl group having 6 to 8 carbon atoms, and X represents a halogen atom.
- X represents a halogen atom.
- the inventors of the present invention described above by mixing and reacting the triazine compound represented by the formula (2), the compound having at least one tertiary amino group, the pseudopolyrotaxane aqueous dispersion, and the blocking agent.
- the blocking agent By introducing blocking groups at both ends of the polyethylene glycol chain in the pseudo polyrotaxane, it was found that a polyrotaxane having a high inclusion rate can be produced industrially advantageously without drying the pseudo polyrotaxane aqueous dispersion.
- the present invention has been completed.
- Step 1 of obtaining a pseudopolyrotaxane aqueous dispersion containing pseudopolyrotaxane particles in which a compound having a repeating structural unit is skewered.
- the mass average molecular weight of the compound having a repeating structural unit represented by the formula (1) is preferably 1,000 to 500,000, more preferably 10,000 to 300,000, and 10,000 to 100,000. Is more preferable.
- the compound having the repeating structural unit represented by the formula (1) has a mass average molecular weight of less than 1000, the crosslinked polyrotaxane obtained by crosslinking the obtained polyrotaxane has sufficient characteristics such that cracks and scratches are not easily generated. May not be able to be demonstrated.
- the mass average molecular weight of the compound having a repeating structural unit represented by the formula (1) exceeds 500,000, the fluidity of the pseudopolyrotaxane aqueous dispersion is low, and uniform stirring and mixing in Step 2 may be difficult. is there.
- the mass average molecular weight is a value determined by gel permeation chromatography (GPC) and calculated in terms of polyethylene glycol.
- GPC gel permeation chromatography
- Examples of the column for measuring the mass average molecular weight in terms of polyethylene glycol by GPC include TSKgel SuperAWM-H (manufactured by Tosoh Corporation).
- the compound having a repeating structural unit represented by the formula (1) preferably has a reactive group at both ends.
- the reactive group can be introduced to both ends of the compound having a repeating structural unit represented by the formula (1) by a conventionally known method.
- the reactive group can be appropriately changed depending on the type of blocking group employed, and is not particularly limited, and examples thereof include a hydroxyl group, an amino group, a carboxyl group, a thiol group, and the like, among others, being a carboxyl group. Is preferred.
- both terminal groups are any of a hydroxyl group, a carboxyl group, and an amino group, and it is more preferable that both terminal groups are both carboxyl groups.
- TEMPO 2,2,6,6-tetramethyl-1-piperidinyloxy radical
- sodium hypochlorite are used to oxidize both ends of polyethylene glycol.
- the mass ratio of the compound having a repeating structural unit represented by formula (1) and cyclodextrin is 1: 2. Is preferably ⁇ 1: 5, more preferably 1: 2.5 to 1: 4.5, and even more preferably 1: 3 to 1: 4.
- the mass of the cyclodextrin is less than twice the mass of the compound having the repeating structural unit represented by the formula (1), the number of cyclodextrins that include the compound having the repeating structural unit represented by the formula (1) (Inclusion amount) may decrease. Even if the mass of the cyclodextrin exceeds 5 times the mass of the compound having the repeating structural unit represented by the formula (1), the inclusion amount does not increase so much and it is not economical.
- cyclodextrin examples include ⁇ -cyclodextrin, ⁇ -cyclodextrin, ⁇ -cyclodextrin, and derivatives thereof. Of these, ⁇ -cyclodextrin is preferable from the viewpoint of inclusion. These cyclodextrins may be used individually by 1 type, and may be used in combination of 2 or more type.
- aqueous medium examples include dimethylformamide, dimethylacetamide, dimethyl sulfoxide, acetone, methyl ethyl ketone, aqueous organic solvents such as methanol, ethanol, isopropanol, ethylene glycol, propylene glycol, glycerin, and tetrahydrofuran, and these aqueous organic solvents and water. And a mixed solvent, water and the like. Especially, the mixed solvent or water of an aqueous organic solvent and water is preferable, and water is more preferable from a viewpoint of inclusion.
- a mixing condition of the compound having a repeating structural unit represented by the formula (1) in the step 1 and the cyclodextrin both may be added and mixed in the aqueous medium. It is preferable to dissolve the compound having a repeating structural unit and cyclodextrin in an aqueous medium.
- a substantially transparent mixed solution can be obtained by heating to 50 to 100 ° C, preferably 60 to 90 ° C, more preferably 70 to 80 ° C and dissolving.
- the compound having the repeating structural unit represented by the formula (1) and the cyclodextrin are formed. Pseudopolyrotaxane particles are precipitated, and a substantially white pseudopolyrotaxane aqueous dispersion is obtained.
- the pseudopolyrotaxane aqueous dispersion When cooling the mixed solution, the pseudopolyrotaxane aqueous dispersion has good fluidity and does not decrease fluidity over time by precipitating pseudopolyrotaxane particles by continuously or intermittently cooling the mixed solution while flowing. You can get a body. By using such a pseudo polyrotaxane aqueous dispersion, uniform stirring and mixing can be performed in Step 2, and the blocking reaction can be easily completed.
- the paste-like or cream-like which has a very low fluidity, or a gel-like pseudo-fluid with no fluidity.
- An aqueous polyrotaxane dispersion is obtained. Even the pseudopolyrotaxane aqueous dispersion obtained in the form of a paste or cream loses its fluidity over time, so that it can be given fluidity by stirring and mixing under appropriate conditions before step 2. It is preferable to keep it.
- the temperature reached after cooling is preferably 0 to 30 ° C, more preferably 1 to 20 ° C, and still more preferably 1 to 15 ° C.
- the fluidity of the pseudopolyrotaxane aqueous dispersion obtained by freezing or the like may decrease.
- pseudopolyrotaxane particles may not be sufficiently precipitated.
- the cooling rate for cooling the mixed solution is preferably 0.01 to 30 ° C./min, more preferably 0.05 to 20 ° C./min, and 0.05 to 10 ° C./min. More preferably it is. If the cooling rate when cooling the mixed solution is less than 0.01 ° C./min, the precipitated pseudo polyrotaxane particles become too fine, so that the fluidity of the resulting pseudo polyrotaxane aqueous dispersion decreases, It may be difficult to mix with stirring. When the cooling rate at the time of cooling the mixed solution exceeds 30 ° C./min, the pseudopolyrotaxane particles become large, so that the reaction rate in Step 2 may be reduced, and the yield of polyrotaxane may be reduced.
- pseudopolyrotaxane particles can be precipitated more completely, and can be intermittently cooled.
- by changing the cooling rate and stirring conditions of the mixed solution in the course of cooling, etc. It is also possible to change the flow state.
- the time for maintaining the fluid state of the obtained pseudopolyrotaxane aqueous dispersion is usually several seconds to one week, preferably several hours to 3 days.
- a conventionally known method such as stirring with a stirring blade or ultrasonic irradiation can be used.
- the degree to which the mixed solution is flowed is not particularly limited, and can be arbitrarily selected from the degree that the mixed solution slightly flows by gentle stirring to the degree that the mixed solution is vigorously fluidized by strong stirring with a homogenizer or the like.
- the precipitated pseudopolyrotaxane particles become larger, so the reaction rate in Step 2 may decrease and the yield of polyrotaxane may decrease.
- the deposited pseudopolyrotaxane particles become too fine, Since the fluidity of the resulting pseudopolyrotaxane aqueous dispersion is lowered, stirring and mixing in Step 2 may be difficult.
- the mixed solution is cooled in a state where it does not flow, it becomes a gel-like pseudopolyrotaxane aqueous dispersion having extremely low fluidity or no fluidity.
- the volume average particle size of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion varies depending on the cooling rate, the temperature reached after cooling, the flow state of the mixed solution during cooling, etc., but the fluidity of the pseudopolyrotaxane aqueous dispersion, From the viewpoint of dispersion stability, the thickness is preferably 1 to 200 ⁇ m, more preferably 1 to 100 ⁇ m, and still more preferably 1 to 50 ⁇ m. When the volume average particle diameter of the pseudopolyrotaxane particles is less than 1 ⁇ m, the fluidity of the dispersion is lowered or does not show fluidity, and thus stirring and mixing in Step 2 may be difficult.
- the reaction rate in Step 2 may be lowered, and the yield of polyrotaxane may be lowered.
- the volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion can be analyzed by a laser diffraction particle size distribution analyzer.
- the concentration of the pseudopolyrotaxane in the pseudopolyrotaxane aqueous dispersion obtained in the step 1 (hereinafter, the concentration of the pseudopolyrotaxane in the pseudopolyrotaxane aqueous dispersion is also referred to as “solid concentration of the pseudopolyrotaxane aqueous dispersion”).
- the content is preferably 5 to 25% by mass, more preferably 5 to 20% by mass, and still more preferably 10 to 20% by mass. If the solid content concentration of the pseudopolyrotaxane aqueous dispersion is less than 5% by mass, it is not economical. When the solid content concentration of the pseudopolyrotaxane aqueous dispersion exceeds 25% by mass, the fluidity of the pseudopolyrotaxane aqueous dispersion may be lowered.
- the method for producing a polyrotaxane according to the present invention includes a triazine compound represented by the formula (2), a compound having at least one tertiary amino group, a pseudo polyrotaxane aqueous dispersion obtained in the step 1, and a blocking agent. And capping groups are introduced to both ends of the compound having the repeating structural unit represented by the formula (1) in the pseudopolyrotaxane. Mixing and reacting the triazine compound represented by the formula (2), the compound having at least one tertiary amino group, the pseudopolyrotaxane aqueous dispersion obtained in the step 1, and a blocking agent.
- R 1 and R 2 are each independently an alkyl group having 1 to 4 carbon atoms or an aryl group having 6 to 8 carbon atoms.
- alkyl group having 1 to 4 carbon atoms represented by R 1 and R 2 include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, and the like.
- aryl group include a phenyl group, a tolyl group, and a xylyl group.
- X represents a halogen atom.
- the halogen atom include fluorine, chlorine, bromine, iodine and the like. Of these, chlorine is preferred because it is easy to synthesize.
- Examples of the triazine compound represented by the formula (2) include 2-chloro-4,6-dimethoxy-1,3,5-triazine, 2-chloro-4,6-diethoxy-1,3,5- Triazine, 2-chloro-4,6-di-n-propoxy-1,3,5-triazine, 2-chloro-4,6-diisopropoxy-1,3,5-triazine, 2-chloro-4, Examples include 6-di-n-butoxy-1,3,5-triazine, 2-chloro-4,6-diphenoxy-1,3,5-triazine and the like.
- 2-chloro-4,6-dimethoxy-1,3,5-triazine, 2-chloro-4,6-diethoxy-1,3,5-triazine and 2-chloro-4,6 are easy to synthesize.
- -Diphenoxy-1,3,5-triazine is preferred, and 2-chloro-4,6-dimethoxy-1,3,5-triazine is more preferred.
- these triazine compounds are available as industrial raw materials, they are generally produced by reacting cyanuric chloride with the corresponding alcohol in the presence of potassium carbonate (or sodium bicarbonate, etc.) and a phase transfer catalyst. be able to.
- the amount of the triazine compound represented by the formula (2) can be appropriately selected depending on the type of the end group of the compound having the repeating structural unit represented by the formula (1), the type of the blocking agent, and the like. ) Is preferably from 0.7 to 50 times mol, more preferably from 0.9 to 50 times mol, more preferably from 1.5 to 1.5 times the number of terminal mols of the compound having a repeating structural unit represented by More preferably, it is 40 times mol.
- the usage-amount of the triazine compound represented by said Formula (2) is less than 0.7 times mole, a condensation reaction may not be completed but there exists a possibility that the yield of a polyrotaxane may fall. Even if the triazine compound represented by the formula (2) is used in an amount exceeding 50-fold moles relative to the number of terminal moles of the compound having the repeating structural unit represented by the formula (1), further effects can be obtained. Not economical.
- Examples of the compound having at least one tertiary amino group include morpholine compounds represented by the following formula (3), N-methylpyrrolidine, N-ethylpyrrolidine, N-methylpiperidine, N-ethylpiperidine, N- Methylindoline, N-methylisoindoline, triethylamine, tributylamine, N, N-dimethylisopropylamine, N, N-dimethylcyclohexylamine, N, N-diisopropylmethylamine, N, N-diisopropylethylamine, N, N, N Aliphatic tertiary amines such as', N'-tetramethylethylenediamine, N, N, N ', N'-tetramethylpropanediamine, N, N, N', N'-tetramethylbutanediamine, Aliphatic tertiary dimers such as 4-dimethylpiperazine and 1,4-diethylpiperaz
- R 3 represents an alkyl group having 1 to 4 carbon atoms.
- Examples of the alkyl group having 1 to 4 carbon atoms represented by R 3 include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, and an n-butyl group.
- the morpholine compound represented by the above formula (3), N, N can be industrially easily obtained and a polyrotaxane can be obtained in high yield.
- -Diisopropylethylamine and 1,4-dimethylpiperazine are preferable, and N-methylmorpholine in which R 3 in the formula (3) is a methyl group is more preferable.
- the amount of the compound having at least one tertiary amino group used is preferably 0.1 to 3 moles relative to 1 mole of the triazine compound represented by the formula (2). ⁇ 2 mol is more preferred, and 0.7 to 1.5 mol is still more preferred.
- the amount of the compound having at least one tertiary amino group used is less than 0.1 mol relative to 1 mol of the triazine compound represented by the formula (2), the condensation reaction may not be completed. Even if the compound having at least one tertiary amino group is used in an amount exceeding 3 moles relative to 1 mole of the triazine compound represented by the formula (2), further effects cannot be obtained and it is not economical.
- the method of mixing the triazine compound represented by the formula (2), the compound having at least one tertiary amino group, the pseudopolyrotaxane aqueous dispersion, and the blocking agent is not particularly limited.
- the pseudopolyrotaxane aqueous dispersion obtained in Step 1 may be mixed with a blocking agent, a triazine compound represented by the formula (2), and a compound having at least one tertiary amino group as they are. It may be added after being dispersed or dissolved in water, a solvent or the like.
- the sequestering agent, the triazine compound represented by the formula (2), and the compound having at least one tertiary amino group may be added individually, or mixed in advance and then dispersed in an aqueous pseudopolyrotaxane. It may be added to the body.
- the mixing order of the pseudopolyrotaxane aqueous dispersion, the blocking agent, the triazine compound represented by the formula (2), and the compound having at least one tertiary amino group is not particularly limited, but the formula (2)
- a triazine compound represented by the formula (1) and a compound having at least one tertiary amino group are added to the pseudopolyrotaxane aqueous dispersion, and the terminal of the compound having a repeating structural unit represented by the formula (1) is esterified
- a method of adding a blocking agent to the aqueous dispersion all at once, or intermittently or continuously is preferable because the polyrotaxane is easily obtained in a high yield.
- Step 2 the triazine compound represented by the formula (2) and a compound having at least one tertiary amino group are mixed and reacted to form a quaternary ammonium salt represented by the following formula (4).
- Step 2-2 for mixing the obtained quaternary ammonium salt represented by the formula (4), the pseudopolyrotaxane aqueous dispersion obtained in Step 1, and a blocking agent. You may have.
- E is an organic group having one or two tertiary amino groups, and is a monovalent or divalent organic group bonded to a triazine ring by a nitrogen atom of the tertiary amino group.
- n is 1 when E has one tertiary amino group, and 2 when E has two tertiary amino groups, and R 1 and R 2 each independently have 1 carbon atom
- the counter anion of a valence is shown.
- the quaternary ammonium salt represented by the formula (4) has one or two chemical structures in the molecule in which a triazine ring is bonded to a quaternary nitrogen atom, and the triazine ring has a 4-position and a 6-position. Is a 1,3,5-triazine ring substituted with an alkoxy group or an aryloxy group, and has a chemical structure bonded to the quaternary nitrogen atom at the 2-position.
- the quaternary nitrogen atom has a triazine ring and three other carbon atoms (two carbon atoms when bonded to a carbon atom by a double bond), and these carbon atoms are These may be contained in three organic groups different from each other, or may be contained in one or two organic groups.
- R 1 and R 2 are each independently an alkyl group having 1 to 4 carbon atoms or an aryl group having 6 to 8 carbon atoms.
- alkyl group having 1 to 4 carbon atoms represented by R 1 and R 2 include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, and the like.
- aryl group include a phenyl group, a tolyl group, and a xylyl group.
- quaternary ammonium salts represented by the formula (4) quaternary ammonium salts in which R 1 and R 2 are each a methyl group, an ethyl group, or a phenyl group are preferable.
- n is 1 when E has one tertiary amino group, is 2 when E has two tertiary amino groups, and a is 1 or 2.
- Z 1- (n / a) is an (n / a) valence, that is, a monovalent or divalent counter anion.
- examples of the monovalent or divalent counter anion represented by Z 2- (n / a) include a halide ion, a perchlorate ion, a boron tetrafluoride anion, a sulfate anion, and a carbonic acid. Anion etc. are mentioned.
- halide ions examples include fluoride ions, chloride ions, bromide ions, iodide ions, and the like.
- n 2 and the counter anion is monovalent, a is 2.
- the two counter anions may be of different types.
- the quaternary ammonium salt represented by the formula (4) can be produced by a known method.
- the quaternary ammonium salt represented by the formula (4) in which Z is a chloride ion includes a triazine compound in which X in the formula (2) is a chlorine atom, a compound having at least one tertiary amino group, and Can be reacted in an organic solvent, and the precipitated crystals can be filtered off.
- the quaternary ammonium salt represented by the formula (4) wherein Z is a perchlorate anion is a compound having a triazine compound in which X in the formula (2) is a chlorine atom and at least one tertiary amino group. Can be produced by simultaneously adding sodium perchlorate.
- the quaternary ammonium salt represented by the formula (4) in which Z is a boron tetrafluoride anion has a triazine compound in which X in the formula (2) is a chlorine atom, and at least one tertiary amino group.
- Z is a boron tetrafluoride anion
- X in the formula (2) is a chlorine atom, and at least one tertiary amino group.
- quaternary ammonium salt represented by the formula (4) include 1- (4,6-dimethoxy-1,3,5-triazin-2-yl) -1-methylpiperidinium. Chloride, 1- (4,6-dimethoxy-1,3,5-triazin-2-yl) -1-methylpiperidinium perchlorate, 1- (4,6-dimethoxy-1,3,5-triazine) 2-yl) -1-methylpiperidinium tetrafluoroborate, 1- (4,6-dimethoxy-1,3,5-triazin-2-yl) -1-ethylpiperidinium chloride, 1- (4 6-dimethoxy-1,3,5-triazin-2-yl) -1-ethylpiperidinium perchlorate, 1- (4,6-dimethoxy-1,3,5-triazin-2-yl) -1- Ethyl piperidinium te Lafluoroborate, 1- (4,6-dimethoxy-1,3,3,5-
- R 1 and R 2 are each independently an alkyl group having 1 to 4 carbon atoms or an aryl group having 6 to 8 carbon atoms
- R 4 is an alkyl group having 1 to 4 carbon atoms
- X represents a halogen atom.
- examples of the alkyl group having 1 to 4 carbon atoms represented by R 1 and R 2 include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, and an n-butyl group. It is done.
- examples of the aryl group having 6 to 8 carbon atoms represented by R 1 and R 2 include a phenyl group, a tolyl group, and a xylyl group.
- examples of the alkyl group having 1 to 4 carbon atoms represented by R 4 include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, and an n-butyl group.
- the alkyl group having 1 to 4 carbon atoms represented by R 1 and R 2 is a methyl group or an ethyl group.
- the aryl group having 6 to 8 carbon atoms is preferably a phenyl group
- the alkyl group having 1 to 4 carbon atoms represented by R 4 is preferably a methyl group or an ethyl group.
- quaternary ammonium salt represented by the formula (5) include 4- (4,6-dimethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium chloride. 4- (4,6-dimethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium perchlorate, 4- (4,6-dimethoxy-1,3,5-triazine-2 -Yl) -4-methylmorpholinium tetrafluoroborate, 4- (4,6-diethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium perchlorate, 4- (4 6-diethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium tetrafluoroborate, 4- (4,6-di-n-propoxy-1,3,5-triazine-2- Il) -4-me Lumorpholinium perchlorate, 4- (4,6-di-n-propoxy-1,3,5-triazine-2-yl)
- the amount of the quaternary ammonium salt represented by the formula (4) can be appropriately selected depending on the type of the end group of the compound having the repeating structural unit represented by the formula (1), the type of the blocking agent, etc. It is preferably 0.7 to 50-fold mol, more preferably 0.9 to 50-fold mol based on the number of terminal moles of the compound having a repeating structural unit represented by (1), More preferably, it is ⁇ 40 times mol.
- the usage-amount of the quaternary ammonium salt represented by said Formula (4) is less than 0.7 times mole, a condensation reaction may not be completed but there exists a possibility that the yield of a polyrotaxane may fall. Even when the quaternary ammonium salt represented by the formula (4) is used in a molar amount exceeding 50 times the number of terminal moles of the compound having the repeating structural unit represented by the formula (1), Not effective and not economical.
- the method of mixing the pseudopolyrotaxane aqueous dispersion, the blocking agent, and the quaternary ammonium salt represented by the formula (4) is not particularly limited, and the pseudopolyrotaxane aqueous dispersion obtained in the step 1 is not limited.
- the sequestering agent and the quaternary ammonium salt represented by the formula (4) obtained in the above Step 2-1 may be added and mixed as they are, or dispersed in water, a solvent or the like. Or you may add, after melt
- the sequestering agent and the quaternary ammonium salt represented by the formula (4) may be added individually, or may be added to the pseudopolyrotaxane aqueous dispersion after mixing in advance.
- the order of addition of the blocking agent and the quaternary ammonium salt represented by the formula (4) is not particularly limited, but it is represented by the formula (4) obtained in the step 2-1.
- a quaternary ammonium salt is added to the pseudopolyrotaxane aqueous dispersion, and after the terminal of the compound having the repeating structural unit represented by the formula (1) is active esterified, the blocking agent is collectively or intermittently or continuously. Is preferable because a polyrotaxane is easily obtained in a high yield.
- step 2-2 it is preferable to further add the compound having at least one tertiary amino group as a catalyst in order to allow the reaction to proceed smoothly, and to add the morpholine compound represented by the formula (3). More preferably.
- the amount of the compound having at least one tertiary amino group used is preferably 0.01 to 3 mol with respect to 1 mol of the quaternary ammonium salt represented by the formula (4). 0.05-2 mol is more preferable, and 0.1-1 mol is still more preferable.
- the amount of the compound having at least one tertiary amino group used is less than 0.01 mol with respect to 1 mol of the quaternary ammonium salt represented by the formula (4), the condensation reaction may not be completed. . Even if the compound having at least one tertiary amino group is used in an amount of more than 3 moles per mole of the quaternary ammonium salt represented by the formula (4), no further effect can be obtained. Not.
- the blocking agent examples include a reactive group that reacts with a terminal group of the compound having a repeating structural unit represented by the formula (1) and a cyclodextrin of the resulting polyrotaxane so that the cyclodextrin is not released from the skewered inclusion state.
- the compound is not particularly limited as long as it is a compound having a blocking group for blocking dextrin, and can be appropriately selected depending on the type of terminal group of the compound having a repeating structural unit represented by formula (1).
- the reactive group of the blocking agent is preferably an amino group or a hydroxyl group.
- Examples of the blocking group of the blocking agent include a dinitrophenyl group, an adamantane group, an anthracene group, a trityl group, cyclodextrins, fluoresceins, pyrenes, and derivative groups thereof.
- the blocking agent examples include 2,4-dinitrophenylamine, 1-adamantanamine and salts thereof, 1-hydroxyadamantane, 1-aminoanthracene, etc., and reactivity and chemical bond stability.
- 1-adamantanamine or an adamantaneamine salt such as adamantaneamine hydrochloride is preferable.
- the amount of the blocking agent used depends on the reaction conditions for introducing blocking groups to both ends of the compound having the repeating structural unit represented by the formula (1) in the pseudopolyrotaxane, but the repeating structure represented by the formula (1)
- the amount is preferably 1 to 50 times mol, more preferably 2 to 50 times mol, still more preferably 2 to 40 times mol, based on the number of moles of the terminal compound. If the amount of the blocking agent used is less than 1 mol, the introduction of the blocking group becomes insufficient, and the polyrotaxane yield may be reduced. Even if the sequestering agent is used in an amount exceeding 50-fold moles relative to the number of terminal moles of the compound having a repeating structural unit represented by the formula (1), no further effect can be obtained and it is not economical.
- the temperature in Step 2 is preferably 5 to 80 ° C., more preferably 10 to 80 ° C., and still more preferably 20 to 80 ° C. If the temperature is less than 5 ° C., introduction of a blocking group may be insufficient. When the temperature exceeds 80 ° C., cyclodextrin in the pseudopolyrotaxane is liberated and the inclusion rate may be reduced.
- the introduction of the blocking group can usually be carried out under atmospheric pressure, but it can be carried out in any state of reduced pressure, normal pressure or increased pressure.
- the time in Step 2 depends on conditions, but the introduction of the blocking group can be completed usually in 1 to 20 hours. Moreover, when the time of the process 2 is too short, introduction
- the pseudopolyrotaxane aqueous dispersion obtained in step 1 is a compound having a sequestering agent, a triazine compound represented by formula (2), and at least one tertiary amino group at a solid content concentration as it is.
- a quaternary ammonium salt represented by the formula (4) obtained by mixing a triazine compound represented by the formula (2) and a compound having at least one tertiary amino group.
- the pseudopolyrotaxane aqueous dispersion is diluted by adding an aqueous medium, centrifugation, filtration, etc.
- concentration of the pseudopolyrotaxane aqueous dispersion can be adjusted to a desired solid content concentration.
- the solid content concentration of the pseudopolyrotaxane aqueous dispersion in Step 2 is preferably 5 to 40% by mass, more preferably 5 to 35% by mass, and still more preferably 10 to 30% by mass. If the solid content concentration of the pseudopolyrotaxane aqueous dispersion is less than 5% by mass, it is not economical. If the solid content concentration of the pseudopolyrotaxane aqueous dispersion exceeds 40% by mass, uniform stirring and mixing in Step 2 becomes difficult, and introduction of the blocking group may not be completed.
- cyclodextrin may be liberated and the inclusion rate may fall.
- the step 2 is preferably performed in the presence of an inorganic salt and / or an aqueous organic solvent.
- an inorganic salt and / or an aqueous organic solvent By carrying out in the presence of an inorganic salt and / or an aqueous organic solvent, liberation of cyclodextrin during the reaction process in Step 2 is further suppressed, and a polyrotaxane having a higher inclusion rate can be produced industrially advantageously.
- the inorganic salt and / or the aqueous organic solvent may be mixed at any stage of Step 1 and Step 2.
- Step 2 the pseudo polyrotaxane aqueous dispersion, the blocking agent, and the formula (2) are used.
- the formula (4) obtained by mixing the triazine compound and the compound having at least one tertiary amino group, or the triazine compound represented by the formula (2) and the compound having at least one tertiary amino group What is necessary is just to react in the state with which the quaternary ammonium salt represented, the inorganic salt, and / or the aqueous organic solvent were mixed. From the viewpoint of obtaining a polyrotaxane having a higher inclusion rate more efficiently, it is preferable to mix an inorganic salt and / or an aqueous organic solvent in Step 2.
- the inorganic salt examples include alkali metal salts of inorganic acids such as potassium sulfate, sodium sulfate, potassium nitrate, sodium nitrate, potassium chloride, sodium chloride, dipotassium monohydrogen phosphate, monosodium dihydrogen phosphate, and calcium chloride. And alkaline earth metal salts of inorganic acids such as magnesium sulfate, aluminum sulfate, and aluminum chloride. These inorganic salts may be used alone or in combination of two or more.
- potassium chloride, sodium chloride, potassium sulfate, and sodium sulfate are preferable, and sodium chloride and sodium sulfate are more preferable from the viewpoint of efficiently suppressing liberation of cyclodextrin.
- the amount of the inorganic salt used is not particularly limited, and has a blocking agent to be used, a triazine compound represented by the formula (2) or a quaternary ammonium salt represented by the formula (4), and at least one tertiary amino group. It can select suitably according to the kind etc. of a compound.
- the concentration of the inorganic salt relative to the total mass of the entire aqueous medium and inorganic salt in step 2 is preferably 1% by mass or more, and more preferably 10% by mass or more. When the concentration of the inorganic salt is less than 1% by mass, the liberation of cyclodextrin may not be suppressed.
- concentration of the said inorganic salt is below the saturation density
- the “whole aqueous medium” means an aqueous medium containing the aqueous organic solvent when an aqueous organic solvent is used together with the inorganic salt.
- the aqueous organic solvent that is preferably present in Step 2 is not particularly limited, and examples thereof include the aqueous organic solvent exemplified as the aqueous medium in Step 1 above. These aqueous organic solvents may be used alone or in combination of two or more.
- an aqueous organic solvent having a solubility of cyclodextrin at 25 ° C. of 10 g or less with respect to 100 g of the aqueous organic solvent is preferable. More preferred are acetone, methyl ethyl ketone, and tetrahydrofuran.
- the amount of the aqueous organic solvent used is not particularly limited, and includes a blocking agent to be used, a triazine compound represented by the formula (2) or a quaternary ammonium salt represented by the formula (4), and at least one tertiary amino group. It can select suitably according to the kind etc. of the compound to have.
- the concentration of the aqueous organic solvent relative to the total mass of the aqueous medium in Step 2 is preferably 5 to 95% by mass, more preferably 10 to 90% by mass, and further preferably 20 to 80% by mass.
- concentration of the aqueous organic solvent with respect to the total mass of the aqueous medium is less than 5% by mass, cyclodextrin may be easily liberated, and even if the aqueous organic solvent is used in excess of 95% by mass, further effects can be obtained. It is not obtained and it is not economical.
- the inclusion rate of the polyrotaxane can be 6 to 60%. If the inclusion rate is less than 6%, the pulley effect may not be exhibited. If the inclusion rate exceeds 60%, cyclodextrin, which is a cyclic molecule, may be arranged too densely and the mobility of cyclodextrin may be reduced. In order for cyclodextrin to exhibit moderate mobility, the inclusion rate is preferably 15 to 40%, more preferably 20 to 40%.
- the inclusion rate includes the repeating structural unit represented by the formula (1) with respect to the maximum inclusion amount of cyclodextrin to the compound having the repeating structural unit represented by the formula (1).
- This is the ratio of the amount of cyclodextrin clathrating the compound, and can be changed by changing the mixing ratio of the compound having the repeating structural unit represented by formula (1) and cyclodextrin, the type of aqueous medium, etc. Can be adjusted.
- the maximum inclusion amount refers to the number of cyclodextrins in the closest enclosure state in which one cyclodextrin is included in two repeating units represented by the formula (1).
- the inclusion rate can be measured by 1 H-NMR. Specifically, a polyrotaxane in which blocking groups are introduced at both ends of the pseudopolyrotaxane is dissolved in DMSO-d 6 and measured by an NMR measuring apparatus (for example, “Varian Mercury-400BB” manufactured by Varian Technologies Japan). The inclusion rate can be calculated by comparing the integrated value derived from 4 to 6 ppm of cyclodextrin with the integrated value of 3 to 4 ppm of cyclodextrin and the compound having a repeating structural unit represented by formula (1). it can.
- the manufacturing method of the polyrotaxane which can manufacture the polyrotaxane which has a high inclusion rate by an industrially advantageous method can be provided.
- the methylene chloride was distilled off with an evaporator, dissolved in 2 L of hot ethanol, and then stored in a freezer ( ⁇ 4 ° C.
- the compound having carboxyl groups at both ends of the PEG chain was precipitated, collected, and dried under reduced pressure to obtain 100 g of a compound having carboxyl groups at both ends of the PEG chain.
- the methylene chloride was distilled off with an evaporator, dissolved in 2 L of hot ethanol, and then stored in a freezer ( ⁇ 4 ° C.
- the compound having carboxyl groups at both ends of the PEG chain was precipitated, collected, and dried under reduced pressure to obtain 100 g of a compound having carboxyl groups at both ends of the PEG chain.
- Example 1 (1) Preparation of pseudopolyrotaxane aqueous dispersion In a 1 L flask equipped with a stirrer, 650 mL of water was added, and 20 g of a compound having carboxyl groups at both ends of the PEG chain prepared by the method of Production Example 1 and ⁇ -cyclo 80 g of dextrin was heated to 70 ° C. and dissolved. While stirring at a rotation speed of 700 rpm using a stirring blade, cooling to 5 ° C.
- a pseudopolyrotaxane aqueous dispersion (solid content concentration: 13.3% by mass) was obtained.
- the volume average particle size of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 10 ⁇ m.
- Example 2 In “(2) Pseudopolyrotaxane blockage”, the amount of 2-chloro-4,6-dimethoxy-1,3,5-triazine added is 2.4 g (the end mole of the compound having a carboxyl group at both ends of the PEG chain). 18 g of polyrotaxane was obtained in the same manner as in Example 1 except that the amount was 10 times mol).
- Example 3 In “(2) Pseudopolyrotaxane blockage”, the amount of 2-chloro-4,6-dimethoxy-1,3,5-triazine added was 0.17 g (the end mole of the compound having carboxyl groups at both ends of the PEG chain). The amount of N-methylmorpholine added is 0.25 g (2.55 moles relative to 2-chloro-4,6-dimethoxy-1,3,5-triazine). Except that, 16 g of polyrotaxane was obtained in the same manner as in Example 1.
- Example 4 In “(2) Blocking of pseudopolyrotaxane”, the addition amount of N-methylmorpholine was 5.5 g (2 mol per mol of 2-chloro-4,6-dimethoxy-1,3,5-triazine). Except for the above, 20 g of polyrotaxane was obtained in the same manner as in Example 1.
- Example 5 In “(2) Pseudopolyrotaxane blockage”, the amount of 2-chloro-4,6-dimethoxy-1,3,5-triazine added was 14.4 g (the terminal mole of the compound having a carboxyl group at both ends of the PEG chain). The addition amount of N-methylmorpholine was 12.6 g (1.5-fold mol with respect to 2-chloro-4,6-dimethoxy-1,3,5-triazine). Except for this, 42 g of polyrotaxane was obtained in the same manner as in Example 1.
- Example 6 In “(2) Pseudopolyrotaxane blockade”, the amount of adamantaneamine hydrochloride added was 10.2 g (40-fold moles relative to the number of terminal moles of the compound having a carboxyl group at both ends of the PEG chain), 2-chloro- The addition amount of 4,6-dimethoxy-1,3,5-triazine was 9.6 g (40-fold mol relative to the number of terminal moles of the compound having a carboxyl group at both ends of the PEG chain), and addition of N-methylmorpholine The same as in Example 1 except that the amount was 7.0 g (1.27 mol per mol of 2-chloro-4,6-dimethoxy-1,3,5-triazine) and the reaction temperature was 80 ° C. Thus, 42 g of polyrotaxane was obtained.
- Example 7 In “(2) Pseudopolyrotaxane sequestration”, 245 g of sodium chloride (concentration 24.0 with respect to the total charged amount of “(1) Preparation of pseudopolyrotaxane aqueous dispersion” and “(2) Pseudopolyrotaxane sequestration”). 58 g of polyrotaxane was obtained in the same manner as in Example 6 except that the reaction was performed by adding (mass%).
- Example 8 In “(2) Pseudopolyrotaxane sequestration”, a concentration of 11.6 g was further added relative to the total amount of sodium sulfate 100 g (“(1) Preparation of pseudopolyrotaxane aqueous dispersion” and “(2) Pseudopolyrotaxane sequestration”). 52 g of polyrotaxane was obtained in the same manner as in Example 1 except that the reaction was performed by adding (mass%).
- Example 9 In “(1) Preparation of pseudopolyrotaxane aqueous dispersion”, the obtained pseudopolyrotaxane aqueous dispersion is centrifuged, the supernatant liquid is removed, and the concentration of the polypolyrotaxane in the pseudopolyrotaxane aqueous dispersion (solid content concentration) was added to the obtained liquid-containing cake-like polyrotaxane so as to be 30% by mass, and stirred and mixed to obtain 330 g of a pseudopolyrotaxane aqueous dispersion having a solid content concentration of 30% by mass.
- Example 2 “(2) Pseudopolyrotaxane” The same as Example 1 except that 330 g of the pseudopolyrotaxane aqueous dispersion (solid content concentration of 30% by mass) was used instead of 750 g of the pseudopolyrotaxane aqueous dispersion (solid content concentration of 13.3% by mass). Thus, 41 g of polyrotaxane was obtained. The volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 11 ⁇ m.
- Example 10 In “(1) Preparation of pseudopolyrotaxane aqueous dispersion”, the obtained pseudopolyrotaxane aqueous dispersion is centrifuged, the supernatant liquid is removed, and the concentration of the polypolyrotaxane in the pseudopolyrotaxane aqueous dispersion (solid content concentration) Is added to the obtained liquid cake-like polyrotaxane, stirred and mixed so that the mass ratio of acetone and water is 1: 1.
- the polyrotaxane aqueous dispersion was 400 g, and in “(2) Blocking of the pseudopolyrotaxane”, the pseudopolyrotaxane aqueous dispersion 400 g (solid content concentration 25 mass%) was replaced with the pseudopolyrotaxane aqueous dispersion 750 g (solid content concentration 13.3% by mass). ) was used in the same manner as in Example 1 except that 48 g of polyrotaxane was obtained.
- the volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 11 ⁇ m.
- Example 11 In “(2) Pseudopolyrotaxane blockade”, the amount of adamantaneamine hydrochloride added is 0.31 g (1.2 times moles relative to the number of terminal moles of the compound having carboxyl groups at both ends of the PEG chain), N— 40 g of polyrotaxane was added in the same manner as in Example 10 except that the amount of methylmorpholine added was 2.8 g (equal mol of 2-chloro-4,6-dimethoxy-1,3,5-triazine). Obtained.
- Example 12 In “(1) Preparation of pseudopolyrotaxane aqueous dispersion”, polyrotaxane was obtained in the same manner as in Example 10 except that water and dimethylacetamide were added to the obtained liquid-containing cake-like polyrotaxane instead of water and acetone. 48 g was obtained. The volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 11 ⁇ m.
- the addition amount of 2-chloro-4,6-dimethoxy-1,3,5-triazine is 3.5 g (40 times the number of moles of the compound having carboxyl groups at both ends of the PEG chain) Mol), and the addition amount of N-methylmorpholine was 2.5 g (1.25 mol per mol of 2-chloro-4,6-dimethoxy-1,3,5-triazine),
- a polyrotaxane 42g in the same manner as ⁇ 6.
- the volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 12 ⁇ m.
- Example 14 In “(2) Blocking of pseudopolyrotaxane”, 5.1 g of 1-hydroxyadamantane as a blocking agent instead of adamantaneamine hydrochloride (30 times moles relative to the number of terminal moles of the compound having a carboxyl group at both ends of the PEG chain) ) was added in the same manner as in Example 10 except that 26 g of polyrotaxane was obtained.
- Example 15 (1) Preparation of pseudopolyrotaxane aqueous dispersion
- a milky pseudopolyrotaxane aqueous dispersion having a good fluidity (solid content concentration of 13.3% by mass) was obtained.
- the volume average particle size of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 10 ⁇ m.
- Example 16 In “(2) Pseudopolyrotaxane blockage”, the amount of adamantaneamine hydrochloride added was 10.2 g (40-fold moles relative to the number of moles of the compound having a carboxyl group at both ends of the PEG chain), 4- (4 , 6-Dimethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium chloride was added in an amount of 15.2 g (based on the number of terminal moles of the compound having carboxyl groups at both ends of the PEG chain). 18 g of polyrotaxane was obtained in the same manner as in Example 15 except that the amount was 40 times mol).
- Example 17 In “(2) Blocking of pseudopolyrotaxane”, the amount of 4- (4,6-dimethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium chloride added was 0.27 g (PEG chain). 15 g of polyrotaxane was obtained in the same manner as in Example 16 except that the molar amount of the compound having a carboxyl group at both ends thereof was 0.7 times the molar number of the terminal.
- Example 18 In “(2) Blocking of pseudopolyrotaxane”, 0.7 g (4- (4,6-dimethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium chloride is further added to N-methylmorpholine. 39 g of polyrotaxane was obtained in the same manner as in Example 15 except that 0.25 times mol) was added.
- Example 20 In “(2) Blocking of pseudopolyrotaxane”, the amount of 4- (4,6-dimethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium chloride added was 3.8 g (PEG chain). Of the compound having a carboxyl group at both ends thereof, and the addition amount of N-methylmorpholine to be added is 0.35 g (4- (4,6-dimethoxy-1,3). , 5-triazin-2-yl) -4-methylmorpholinium chloride was obtained in the same manner as in Example 18 except that 20 g of polyrotaxane was obtained.
- Example 21 In “(2) Blocking of pseudopolyrotaxane”, 2.8 g of N-methylmorpholine (4- (4,6-dimethoxy-1,3,5-triazin-2-yl) -4-methylmorpholinium chloride is further added. 42 g of polyrotaxane was obtained in the same manner as in Example 16 except that 0.5 mol per mol) was added.
- Example 22 In “(2) Blocking of pseudopolyrotaxane”, the amount of N-methylmorpholine added is 2.8 g (4- (4,6-dimethoxy-1,3,5-triazin-2-yl) -4- 20 g of polyrotaxane was obtained in the same manner as in Example 18 except that the molar ratio was 1 mol) with respect to methylmorpholinium chloride.
- Example 23 In “(1) Preparation of pseudopolyrotaxane aqueous dispersion”, 245 g of sodium chloride (concentration of 24.6% by mass with respect to the total amount of “(1) Preparation of pseudopolyrotaxane aqueous dispersion”) was added, In (2) Pseudopolyrotaxane blockage, 58 g of polyrotaxane was obtained in the same manner as in Example 19 except that the reaction temperature was 80 ° C. The volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 10 ⁇ m.
- Example 24 In addition to the addition of 100 g of sodium sulfate (concentration of 11.8% by mass with respect to the total charged amount of “(1) Preparation of pseudopolyrotaxane aqueous dispersion”) in “(1) Preparation of pseudopolyrotaxane aqueous dispersion”.
- the volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 10 ⁇ m.
- Example 25 In “(1) Preparation of pseudopolyrotaxane aqueous dispersion”, the obtained pseudopolyrotaxane aqueous dispersion is centrifuged, the supernatant liquid is removed, and the concentration of the polypolyrotaxane in the pseudopolyrotaxane aqueous dispersion (solid content concentration) was added to the obtained liquid-containing cake-like polyrotaxane so as to be 30% by mass, and stirred and mixed to obtain 330 g of a pseudopolyrotaxane aqueous dispersion having a solid content concentration of 30% by mass.
- Example 18 “(2) Pseudopolyrotaxane” The same as Example 18 except that 330 g of the pseudopolyrotaxane aqueous dispersion (solid content concentration of 30% by mass) was used instead of 750 g of the pseudopolyrotaxane aqueous dispersion (solid content concentration of 13.3% by mass). Thus, 41 g of polyrotaxane was obtained. The volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 11 ⁇ m.
- Example 26 In “(1) Preparation of pseudopolyrotaxane aqueous dispersion”, the obtained pseudopolyrotaxane aqueous dispersion is centrifuged, the supernatant liquid is removed, and the concentration of the polypolyrotaxane in the pseudopolyrotaxane aqueous dispersion (solid content concentration) Is added to the obtained liquid cake-like polyrotaxane, stirred and mixed so that the mass ratio of acetone and water is 1: 1.
- the polyrotaxane aqueous dispersion was 400 g, and in “(2) Blocking of the pseudopolyrotaxane”, the pseudopolyrotaxane aqueous dispersion 400 g (solid content concentration 25 mass%) was replaced with the pseudopolyrotaxane aqueous dispersion 750 g (solid content concentration 13.3% by mass). ) was used in the same manner as in Example 18 except that 46 g of polyrotaxane was obtained.
- Example 27 In “(2) Blocking of pseudopolyrotaxane”, the addition amount of adamantaneamine hydrochloride was set to 0.31 g (1.2 moles relative to the number of terminal moles of the compound having a carboxyl group at both ends of the PEG chain). Except for the above, 38 g of polyrotaxane was obtained in the same manner as in Example 18.
- Example 28 In “(1) Preparation of pseudo-polyrotaxane aqueous dispersion”, polyrotaxane was obtained in the same manner as in Example 26 except that water and dimethylacetamide were added to the obtained liquid-containing cake-like polyrotaxane instead of water and acetone. 46 g was obtained. The volume average particle diameter of the pseudopolyrotaxane particles in the pseudopolyrotaxane aqueous dispersion was 11 ⁇ m.
- Example 29 In “(1) Preparation of aqueous dispersion of pseudopolyrotaxane”, instead of the compound having a carboxyl group at both ends of the PEG chain prepared by the method of Production Example 1, both ends of the PEG chain prepared by the method of Production Example 2 were used. Using a compound having a carboxyl group, in “(2) Blocking of pseudopolyrotaxane”, the addition amount of adamantaneamine hydrochloride is 3.5 g (relative to the number of terminal moles of the compound having a carboxyl group at both ends of the PEG chain).
- Example 30 In “(2) Blocking of pseudopolyrotaxane”, 5.1 g of 1-hydroxyadamantane as a blocking agent instead of adamantaneamine hydrochloride (30 times moles relative to the number of terminal moles of the compound having a carboxyl group at both ends of the PEG chain) ) was used in the same manner as in Example 19 except that 26 g of polyrotaxane was obtained.
- Example 31 In “(2) Blocking of pseudopolyrotaxane”, 3.95 g (2-chloro-4,6-dimethoxy-1,3,5-triazine) of 1,4-dimethylpiperazine instead of N-methylmorpholine as a tertiary amine was used. 24 g of polyrotaxane was obtained in the same manner as in Example 1 except that 1.27 mol) was added.
- Example 32 In “(2) Blocking of Pseudopolyrotaxane”, 0.79 g (4- (4,6-dimethoxy-1,3,5-triazine-) is substituted as a tertiary amine in place of N-methylmorpholine. 30 g of polyrotaxane was obtained in the same manner as in Example 18, except that 2-yl) -4-methylmorpholinium chloride was added 0.25 times mol).
- Example 33 In “(2) Blocking of pseudopolyrotaxane”, 0.89 g (4- (4,6-dimethoxy-1,3,5-triazine-) of N, N-diisopropylethylamine instead of N-methylmorpholine as a tertiary amine was used. 31 g of polyrotaxane was obtained in the same manner as in Example 18, except that 2-yl) -4-methylmorpholinium chloride was added (0.25 times mol).
- Example 34 In “(2) Blocking of pseudopolyrotaxane”, the addition amount of N-methylmorpholine was 2.8 g (equal moles relative to 2-chloro-4,6-dimethoxy-1,3,5-triazine), and Except that 0.79 g of 1,4-dimethylpiperazine was added (0.22 moles relative to 2-chloro-4,6-dimethoxy-1,3,5-triazine), the same procedure as in Example 1 was performed. 24 g of polyrotaxane was obtained.
- the manufacturing method of the polyrotaxane which can manufacture the polyrotaxane which has a high inclusion rate by an industrially advantageous method can be provided.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Materials Engineering (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Cosmetics (AREA)
- Plural Heterocyclic Compounds (AREA)
- Polyethers (AREA)
- Processes Of Treating Macromolecular Substances (AREA)
Abstract
Description
しかしながら、このような擬ポリロタキサンの両末端に封鎖基を導入する反応においては、系内の水分が反応を失活させる。そのため、反応を効率よく進行させるには、反応系中に水を存在させないか又は反応系中の水分量を極めて微量に制御する必要があり、擬ポリロタキサン水性分散体を遠心分離、濾過等により固液分離した後、乾燥処理する方法や、擬ポリロタキサン水性分散体をそのまま乾燥処理する方法等により、充分に水を除去する必要がある。
従来のポリロタキサンの製造方法では、PEG鎖の両末端に封鎖基を導入する反応を阻害する水を、乾燥により充分に除去する必要があるため、非常に長い乾燥時間を要する。
また、70℃以下の乾燥温度であっても含水状態で長時間加熱するとシクロデキストリンが遊離し、包接率が低くなってしまうという問題がある。
更に、乾燥することにより得られた擬ポリロタキサンが塊状となるため、ポリエチレングリコールの両末端に封鎖基を導入する反応の前に、粉砕、分級等により粉末状にする必要があり、工程が煩雑になるという問題がある。
本発明は、高い包接率を有するポリロタキサンを工業的に有利な方法で製造することができるポリロタキサンの製造方法を提供することを目的とする。
以下に本発明を詳述する。
なお、本明細書において、前記質量平均分子量は、ゲルパーミエーションクロマトグラフィー(GPC)で測定を行い、ポリエチレングリコール換算により求められる値である。GPCによってポリエチレングリコール換算による質量平均分子量を測定する際のカラムとしては、例えば、TSKgel SuperAWM-H(東ソー社製)等が挙げられる。
前記反応性基は、採用する封鎖基の種類により適宜変更することができ、特に限定されないが、例えば、水酸基、アミノ基、カルボキシル基、チオール基等が挙げられ、なかでも、カルボキシル基であることが好ましい。更に、両末端基がともに水酸基、カルボキシル基、アミノ基のいずれかであることが好ましく、両末端基がともにカルボキシル基であることがより好ましい。
前記式(1)で表される繰り返し構造単位を有する化合物の両末端にカルボキシル基を導入する方法としては、例えば、TEMPO(2,2,6,6-テトラメチル-1-ピペリジニルオキシラジカル)と次亜塩素酸ナトリウムとを用いてポリエチレングリコールの両末端を酸化させる方法等が挙げられる。
なお、前記混合溶液を冷却する際に、静置下で冷却して擬ポリロタキサン粒子を析出させた場合には、極めて流動性の低いペースト状やクリーム状、又は、流動性のないゲル状の擬ポリロタキサン水性分散体が得られる。ペースト状やクリーム状で得られた擬ポリロタキサン水性分散体であっても経時的に流動性を失うため、工程2の前に適当な条件下で撹拌、混合する等により、流動性を付与しておくことが好ましい。
一方、混合溶液を流動させない状態で冷却した場合、極めて流動性が低いか流動性のないゲル状の擬ポリロタキサン水性分散体となる。
なお、本明細書において前記擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は、レーザー回折式粒度分布測定装置により分析することができる。
前記式(2)で表されるトリアジン化合物と、少なくとも1つの三級アミノ基を有する化合物と、前記工程1にて得られた擬ポリロタキサン水性分散体と、封鎖剤とを混合して反応させることにより、従来行われてきた乾燥工程が不要となるだけでなく、乾燥工程でのシクロデキストリンの遊離を回避することができるため、高い包接率を有するポリロタキサンが得られる。また、乾燥工程による擬ポリロタキサンの塊状化を回避することができ、粉砕、分級等が不要となるため、より簡便なプロセスにより包接率の高いポリロタキサンを製造することができる。
R1及びR2で表される炭素数1~4のアルキル基としては、例えば、メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基等が挙げられ、炭素数6~8のアリール基としては、例えば、フェニル基、トリル基、キシリル基等が挙げられる。
前記ハロゲン原子としては、例えば、フッ素、塩素、臭素、ヨウ素等が挙げられる。なかでも、合成が容易であることから、塩素が好ましい。
R3で表される炭素数1~4のアルキル基としては、例えば、メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基等が挙げられる。
前記四級窒素原子には、トリアジン環と他の3個の炭素原子(二重結合により炭素原子と結合している場合は2個の炭素原子)が結合しているが、これらの炭素原子は、互いに別の3個の有機基に含まれていてもよいし、1又は2個の有機基に含まれていてもよい。
R1及びR2で表される炭素数1~4のアルキル基としては、例えば、メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基等が挙げられ、炭素数6~8のアリール基としては、例えば、フェニル基、トリル基、キシリル基等が挙げられる。
前記式(4)において、Z-(n/a)で表される1価又は2価のカウンターアニオンとしては、例えば、ハロゲン化物イオン、過塩素酸イオン、四フッ化ホウ素アニオン、硫酸アニオン、炭酸アニオン等が挙げられる。前記ハロゲン化物イオンとしては、例えば、フッ化物イオン、塩化物イオン、臭化物イオン、ヨウ化物イオン等が挙げられる。前記式(4)において、nが2でありカウンターアニオンが1価である場合、aは2となるが、このとき2個のカウンターアニオンはそれぞれ異なる種類であってもよい。
前記式(5)において、R1及びR2で表される炭素数6~8のアリール基としては、例えば、フェニル基、トリル基、キシリル基等が挙げられる。
前記式(5)において、R4で示される炭素数1~4のアルキル基としては、例えば、メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基等が挙げられる。
封鎖剤の封鎖基としては、例えば、ジニトロフェニル基、アダマンタン基、アントラセン基、トリチル基、シクロデキストリン類、フルオレセイン類、ピレン類、及び、これらの誘導体基等が挙げられる。
封鎖基の導入は通常、大気圧下で実施可能であるが、減圧、常圧、加圧のいずれの状態でも行うことができる。
前記無機塩のなかでも、シクロデキストリンの遊離を効率よく抑制するという観点から、塩化カリウム、塩化ナトリウム、硫酸カリウム、硫酸ナトリウムが好ましく、塩化ナトリウム、硫酸ナトリウムがより好ましい。
また、前記無機塩の濃度は、工程2での温度における無機塩の飽和濃度以下であることが好ましい。工程2での温度における無機塩の飽和濃度を超えて無機塩を使用してもそれ以上の効果が得られず、経済的でない。
なお、「水性媒体全体」とは、無機塩とともに水性有機溶媒を用いる場合は該水性有機溶媒を含む水性媒体を意味する。
なお、本明細書において前記包接率とは、式(1)で表される繰り返し構造単位を有する化合物へのシクロデキストリンの最大包接量に対する式(1)で表される繰り返し構造単位を有する化合物を包接しているシクロデキストリンの包接量の割合であり、式(1)で表される繰り返し構造単位を有する化合物とシクロデキストリンの混合比、水性媒体の種類等を変化させることにより、任意に調整することができる。また、前記最大包接量とは、式(1)で表される繰り返し単位2つに対し、シクロデキストリンが1つ包接された最密包接状態とした場合のシクロデキストリンの個数をいう。
1L容のフラスコ内で、PEG(質量平均分子量35000)100g、TEMPO(2,2,6,6-テトラメチル-1-ピペリジニルオキシラジカル)1g、臭化ナトリウム10gを水1Lに溶解させた。次いで、次亜塩素酸ナトリウム水溶液(有効塩素濃度5%)50mLを添加し、室温で30分間撹拌した。余った次亜塩素酸ナトリウムを分解させるために、エタノールを50mL添加して反応を終了させた。
分液ロートを用い、500mLの塩化メチレンを用いた抽出を3回繰り返して有機層を分取した後、エバポレーターで塩化メチレンを留去し、2Lの温エタノールに溶解させてから冷凍庫(-4℃)中で一晩静置し、PEG鎖の両末端にカルボキシル基を有する化合物を析出させ、回収し、減圧乾燥することにより、PEG鎖の両末端にカルボキシル基を有する化合物100gを得た。
1L容のフラスコ内で、高分子量PEG(質量平均分子量10万)100g、TEMPO(2,2,6,6-テトラメチル-1-ピペリジニルオキシラジカル)1g、臭化ナトリウム10gを水1Lに溶解させた。次いで、次亜塩素酸ナトリウム水溶液(有効塩素濃度5%)50mLを添加し、室温で30分間撹拌した。余った次亜塩素酸ナトリウムを分解させるために、エタノールを50mL添加して反応を終了させた。
分液ロートを用い、500mLの塩化メチレンを用いた抽出を3回繰り返して有機層を分取した後、エバポレーターで塩化メチレンを留去し、2Lの温エタノールに溶解させてから冷凍庫(-4℃)中で一晩静置し、PEG鎖の両末端にカルボキシル基を有する化合物を析出させ、回収し、減圧乾燥することにより、PEG鎖の両末端にカルボキシル基を有する化合物100gを得た。
(1)擬ポリロタキサン水性分散体の調製
撹拌機付きの1L容のフラスコ内に、水650mLを加え、製造例1の方法で調製したPEG鎖の両末端にカルボキシル基を有する化合物20g及びα-シクロデキストリン80gを70℃まで加熱し、溶解させた。
撹拌翼を用い、700rpmの回転速度で撹拌しながら、0.4℃/分の冷却速度にて5℃まで冷却し、更に同温度にて10時間撹拌し続けることにより、流動性のよい乳液状の擬ポリロタキサン水性分散体(固形分濃度13.3質量%)を得た。
レーザー回折式粒径測定装置を用いて測定した結果、擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は10μmであった。
調製した擬ポリロタキサン水性分散体750g(固形分濃度13.3質量%)に、撹拌翼を用い、700rpmの回転速度で撹拌しながら、封鎖剤としてアダマンタンアミン塩酸塩5.1g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して20倍モル)、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンを4.8g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して20倍モル)、N-メチルモルホリン3.5g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して1.27倍モル)を添加し、60℃まで昇温し、同温度にて4時間その温度を保持し、反応させた。得られた混合物を遠心分離し、上澄み液を除去して得られた含液ケーキ状のポリロタキサンに、水300gを加えて撹拌下で70℃まで昇温し、同温度で60分間撹拌して、再度遠心分離し、上澄み液を除去した。この洗浄操作を更に2回繰り返し、得られた含液ケーキ状の精製ポリロタキサンを凍結乾燥(-10~20℃にて48時間乾燥)し、ポリロタキサン36gを得た。
「(2)擬ポリロタキサンの封鎖」において、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンの添加量を2.4g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して10倍モル)としたこと以外は、実施例1と同様にしてポリロタキサン18gを得た。
「(2)擬ポリロタキサンの封鎖」において、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンの添加量を0.17g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して0.7倍モル)、N-メチルモルホリンの添加量を0.25g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して2.55倍モル)としたこと以外は、実施例1と同様にしてポリロタキサン16gを得た。
「(2)擬ポリロタキサンの封鎖」において、N-メチルモルホリンの添加量を5.5g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して2倍モル)としたこと以外は、実施例1と同様にしてポリロタキサン20gを得た。
「(2)擬ポリロタキサンの封鎖」において、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンの添加量を14.4g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して60倍モル)、N-メチルモルホリンの添加量を12.6g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して1.5倍モル)としたこと以外は、実施例1と同様にしてポリロタキサン42gを得た。
「(2)擬ポリロタキサンの封鎖」において、アダマンタンアミン塩酸塩の添加量を10.2g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンの添加量を9.6g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)、N-メチルモルホリンの添加量を7.0g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して1.27倍モル)とし、反応温度を80℃としたこと以外は、実施例1と同様にしてポリロタキサン42gを得た。
「(2)擬ポリロタキサンの封鎖」において、更に塩化ナトリウム245g(「(1)擬ポリロタキサン水性分散体の調整」と「(2)擬ポリロタキサンの封鎖」との総仕込み量に対して濃度24.0質量%)を添加して反応させたこと以外は、実施例6と同様にしてポリロタキサン58gを得た。
「(2)擬ポリロタキサンの封鎖」において、更に硫酸ナトリウム100g(「(1)擬ポリロタキサン水性分散体の調整」と「(2)擬ポリロタキサンの封鎖」との総仕込み量に対して濃度11.6質量%)を添加して反応させたこと以外は、実施例1と同様にしてポリロタキサン52gを得た。
「(1)擬ポリロタキサン水性分散体の調製」において、得られた擬ポリロタキサン水性分散体を遠心分離して、上澄み液を除去し、擬ポリロタキサン水性分散体に占める擬ポリロタキサンの濃度(固形分濃度)が30質量%となるように、得られた含液ケーキ状のポリロタキサンに水を添加し、撹拌、混合して固形分濃度30質量%の擬ポリロタキサン水性分散体330gとし、「(2)擬ポリロタキサンの封鎖」において、擬ポリロタキサン水性分散体750g(固形分濃度13.3質量%)に代えて擬ポリロタキサン水性分散体330g(固形分濃度30質量%)を使用したこと以外は、実施例1と同様にしてポリロタキサン41gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は11μmであった。
「(1)擬ポリロタキサン水性分散体の調製」において、得られた擬ポリロタキサン水性分散体を遠心分離して、上澄み液を除去し、擬ポリロタキサン水性分散体に占める擬ポリロタキサンの濃度(固形分濃度)が25質量%、アセトンと水の質量比が1:1となるように、得られた含液ケーキ状のポリロタキサンに水及びアセトンを添加し、撹拌、混合して固形分濃度25質量%の擬ポリロタキサン水性分散体400gとし、「(2)擬ポリロタキサンの封鎖」において、擬ポリロタキサン水性分散体750g(固形分濃度13.3質量%)に代えて擬ポリロタキサン水性分散体400g(固形分濃度25質量%)を使用したこと以外は、実施例1と同様にしてポリロタキサン48gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は11μmであった。
「(2)擬ポリロタキサンの封鎖」において、アダマンタンアミン塩酸塩の添加量を0.31g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して1.2倍モル)、N-メチルモルホリンの添加量を2.8g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して等倍モル)としたこと以外は、実施例10と同様にしてポリロタキサン40gを得た。
「(1)擬ポリロタキサン水性分散体の調製」において、得られた含液ケーキ状のポリロタキサンに、水及びアセトンに代えて水及びジメチルアセトアミドを添加したこと以外は、実施例10と同様にしてポリロタキサン48gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は11μmであった。
「(1)擬ポリロタキサン水性分散体の調製」において、製造例1の方法で調製したPEG鎖の両末端にカルボキシル基を有する化合物に代えて製造例2の方法で調製したPEG鎖の両末端にカルボキシル基を有する化合物を使用し、「(2)擬ポリロタキサンの封鎖」において、アダマンタンアミン塩酸塩の添加量を3.5g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンの添加量を3.5g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)、N-メチルモルホリンの添加量を2.5g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して1.25倍モル)としたこと以外は、実施例6と同様にしてポリロタキサン42gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は12μmであった。
「(2)擬ポリロタキサンの封鎖」において、封鎖剤としてアダマンタンアミン塩酸塩に代えて1-ヒドロキシアダマンタン5.1g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して30倍モル)を添加したこと以外は、実施例10と同様にしてポリロタキサン26gを得た。
(1)擬ポリロタキサン水性分散体の調製
実施例1と同様にして、流動性のよい乳液状の擬ポリロタキサン水性分散体(固形分濃度13.3質量%)を得た。
レーザー回折式粒径測定装置を用いて測定した結果、擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は10μmであった。
調製した擬ポリロタキサン水性分散体750g(固形分濃度13.3質量%)に、撹拌翼を用い、700rpmの回転速度で撹拌しながら、封鎖剤としてアダマンタンアミン塩酸塩5.1g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して20倍モル)、予め2-クロロ-4,6-ジメトキシ-1,3,5-トリアジン4.8gとN-メチルモルホリン2.8gとを混合し、反応させて得られた4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライド7.6g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して20倍モル)を添加し、60℃まで昇温し、同温度にて4時間その温度を保持し、反応させた。得られた混合物を遠心分離し、上澄み液を除去して得られた含液ケーキ状のポリロタキサンに、水300gを加えて撹拌下で70℃まで昇温し、同温度で60分間撹拌して、再度遠心分離し、上澄み液を除去した。この洗浄操作を更に2回繰り返し、得られた含液ケーキ状の精製ポリロタキサンを凍結乾燥(-10~20℃にて48時間乾燥)し、ポリロタキサン14gを得た。
「(2)擬ポリロタキサンの封鎖」において、アダマンタンアミン塩酸塩の添加量を10.2g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)、4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドの添加量を15.2g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)としたこと以外は、実施例15と同様にしてポリロタキサン18gを得た。
「(2)擬ポリロタキサンの封鎖」において、4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドの添加量を0.27g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して0.7倍モル)としたこと以外は、実施例16と同様にしてポリロタキサン15gを得た。
「(2)擬ポリロタキサンの封鎖」において、更にN-メチルモルホリンを0.7g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して0.25倍モル)添加したこと以外は、実施例15と同様にしてポリロタキサン39gを得た。
「(2)擬ポリロタキサンの封鎖」において、更にN-メチルモルホリンを1.4g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して0.25倍モル)添加したこと以外は、実施例16と同様にしてポリロタキサン50gを得た。
「(2)擬ポリロタキサンの封鎖」において、4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドの添加量を3.8g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して10倍モル)、及び、更に添加するN-メチルモルホリンの添加量を0.35g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して0.25倍モル)としたこと以外は、実施例18と同様にしてポリロタキサン20gを得た。
「(2)擬ポリロタキサンの封鎖」において、更にN-メチルモルホリンを2.8g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して0.5倍モル)添加したこと以外は、実施例16と同様にしてポリロタキサン42gを得た。
「(2)擬ポリロタキサンの封鎖」において、更に添加するN-メチルモルホリンの添加量を2.8g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して等倍モル)としたこと以外は、実施例18と同様にしてポリロタキサン20gを得た。
「(1)擬ポリロタキサン水性分散体の調製」において、更に塩化ナトリウム245g(「(1)擬ポリロタキサン水性分散体の調整」の総仕込み量に対して濃度24.6質量%)を添加し、「(2)擬ポリロタキサンの封鎖」において、反応温度を80℃としたこと以外は、実施例19と同様にしてポリロタキサン58gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は10μmであった。
「(1)擬ポリロタキサン水性分散体の調製」において、更に硫酸ナトリウム100g(「(1)擬ポリロタキサン水性分散体の調整」の総仕込み量に対して濃度11.8質量%)を添加したこと以外は、実施例18と同様にしてポリロタキサン52gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は10μmであった。
「(1)擬ポリロタキサン水性分散体の調製」において、得られた擬ポリロタキサン水性分散体を遠心分離して、上澄み液を除去し、擬ポリロタキサン水性分散体に占める擬ポリロタキサンの濃度(固形分濃度)が30質量%となるように、得られた含液ケーキ状のポリロタキサンに水を添加し、撹拌、混合して固形分濃度30質量%の擬ポリロタキサン水性分散体330gとし、「(2)擬ポリロタキサンの封鎖」において、擬ポリロタキサン水性分散体750g(固形分濃度13.3質量%)に代えて擬ポリロタキサン水性分散体330g(固形分濃度30質量%)を使用したこと以外は、実施例18と同様にしてポリロタキサン41gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は11μmであった。
「(1)擬ポリロタキサン水性分散体の調製」において、得られた擬ポリロタキサン水性分散体を遠心分離して、上澄み液を除去し、擬ポリロタキサン水性分散体に占める擬ポリロタキサンの濃度(固形分濃度)が25質量%、アセトンと水の質量比が1:1となるように、得られた含液ケーキ状のポリロタキサンに水及びアセトンを添加し、撹拌、混合して固形分濃度25質量%の擬ポリロタキサン水性分散体400gとし、「(2)擬ポリロタキサンの封鎖」において、擬ポリロタキサン水性分散体750g(固形分濃度13.3質量%)に代えて擬ポリロタキサン水性分散体400g(固形分濃度25質量%)を使用したこと以外は、実施例18と同様にしてポリロタキサン46gを得た。
「(2)擬ポリロタキサンの封鎖」において、アダマンタンアミン塩酸塩の添加量を0.31g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して1.2倍モル)としたこと以外は、実施例18と同様にしてポリロタキサン38gを得た。
「(1)擬ポリロタキサン水性分散体の調製」において、得られた含液ケーキ状のポリロタキサンに、水及びアセトンに代えて水及びジメチルアセトアミドを添加したこと以外は、実施例26と同様にしてポリロタキサン46gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は11μmであった。
「(1)擬ポリロタキサン水性分散体の調製」において、製造例1の方法で調製したPEG鎖の両末端にカルボキシル基を有する化合物に代えて製造例2の方法で調製したPEG鎖の両末端にカルボキシル基を有する化合物を使用し、「(2)擬ポリロタキサンの封鎖」において、アダマンタンアミン塩酸塩の添加量を3.5g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)、4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドの添加量を5.5g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して40倍モル)、及び、更に添加するN-メチルモルホリンの添加量を0.5g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して0.25倍モル)としたこと以外は、実施例19と同様にしてポリロタキサン42gを得た。
擬ポリロタキサン水性分散体中の擬ポリロタキサン粒子の体積平均粒子径は12μmであった。
「(2)擬ポリロタキサンの封鎖」において、封鎖剤としてアダマンタンアミン塩酸塩に代えて1-ヒドロキシアダマンタン5.1g(PEG鎖の両末端にカルボキシル基を有する化合物の末端モル数に対して30倍モル)を使用したこと以外は、実施例19と同様にしてポリロタキサン26gを得た。
「(2)擬ポリロタキサンの封鎖」において、三級アミンとしてN-メチルモルホリンに代えて1,4-ジメチルピペラジンを3.95g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して1.27倍モル)添加したこと以外は、実施例1と同様にしてポリロタキサン24gを得た。
「(2)擬ポリロタキサンの封鎖」において、三級アミンとしてN-メチルモルホリンに代えて1,4-ジメチルピペラジンを0.79g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して0.25倍モル)添加したこと以外は、実施例18と同様にしてポリロタキサン30gを得た。
「(2)擬ポリロタキサンの封鎖」において、三級アミンとしてN-メチルモルホリンに代えてN,N-ジイソプロピルエチルアミンを0.89g(4-(4,6-ジメトキシ-1,3,5-トリアジン-2-イル)-4-メチルモルホリニウムクロライドに対して0.25倍モル)添加したこと以外は、実施例18と同様にしてポリロタキサン31gを得た。
「(2)擬ポリロタキサンの封鎖」において、N-メチルモルホリンの添加量を2.8g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して等倍モル)とし、更に1,4-ジメチルピペラジンを0.79g(2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンに対して0.22倍モル)添加したこと以外は、実施例1と同様にしてポリロタキサン24gを得た。
「(2)擬ポリロタキサンの封鎖」において、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジン及びN-メチルモルホリンに代えてBOP試薬(ベンゾトリアゾール-1-イル-オキシ-トリス(ジメチルアミノ)ホスホニウム・ヘキサフルオロフォスフェート)4.8g及びジイソプロピルエチルアミン3.5gを使用したこと以外は、実施例1と同様にして擬ポリロタキサンの封鎖を行ったが、封鎖反応が進行しないため、ポリロタキサンを得ることができなかった。
「(1)擬ポリロタキサン水性分散体の調製」において、得られた擬ポリロタキサン水性分散体を凍結乾燥(-10~20℃にて48時間乾燥)し、得られた塊状の擬ポリロタキサンを粉砕して得られた粉末状の擬ポリロタキサン100gにジメチルホルムアミド650mlを加えて撹拌、混合することにより水性分散体とし、「(2)擬ポリロタキサンの封鎖」において、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジン及びN-メチルモルホリンに代えてBOP試薬(ベンゾトリアゾール-1-イル-オキシ-トリス(ジメチルアミノ)ホスホニウム・ヘキサフルオロフォスフェート)4.8g及びジイソプロピルエチルアミン3.5gを使用したこと以外は、実施例1と同様にしてポリロタキサン56gを得た。
「(2)擬ポリロタキサンの封鎖」において、N-メチルモルホリンを使用しなかったこと以外は、実施例1と同様にして擬ポリロタキサンの封鎖を行ったが、封鎖反応が進行しないため、ポリロタキサンを得ることができなかった。
「(2)擬ポリロタキサンの封鎖」において、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンを使用しなかったこと以外は、実施例1と同様にして擬ポリロタキサンの封鎖を行ったが、封鎖反応が進行しないため、ポリロタキサンを得ることができなかった。
実施例及び比較例で得られたポリロタキサンについて、NMR測定装置(バリアン・テクノロジーズ・ジャパン社製、「VARIAN Mercury-400BB」)を用いた1H-NMRにより包接率を測定した。結果を表1に示した。
Claims (16)
- シクロデキストリンと、該シクロデキストリンの開口部に串刺し状に貫通して包接された下記式(1)で表される繰り返し構造単位を有する化合物と、該式(1)で表される繰り返し構造単位を有する化合物の両末端に配置され、シクロデキストリンの脱離を防止する封鎖基とを有するポリロタキサンの製造方法であって、
式(1)で表される繰り返し構造単位を有する化合物とシクロデキストリンとを水性媒体中で混合し、シクロデキストリン分子の開口部に前記式(1)で表される繰り返し構造単位を有する化合物が串刺し状に包接された擬ポリロタキサン粒子を含有する擬ポリロタキサン水性分散体を得る工程1と、
下記式(2)で表されるトリアジン化合物と、少なくとも1つの三級アミノ基を有する化合物と、前記工程1で得られた擬ポリロタキサン水性分散体と、封鎖剤とを混合し、前記擬ポリロタキサンにおける式(1)で表される繰り返し構造単位を有する化合物の両末端に封鎖基を導入する工程2とを有する
ことを特徴とするポリロタキサンの製造方法。
- 式(2)で表されるトリアジン化合物は、2-クロロ-4,6-ジメトキシ-1,3,5-トリアジンであることを特徴とする請求項1記載のポリロタキサンの製造方法。
- 式(2)で表されるトリアジン化合物の使用量が式(1)で表される繰り返し構造単位を有する化合物の末端モル数に対して0.7~50倍モルであることを特徴とする請求項1又は2記載のポリロタキサンの製造方法。
- 式(3)で表されるモルホリン化合物は、N-メチルモルホリンであることを特徴とする請求項4記載のポリロタキサンの製造方法。
- 少なくとも1つの三級アミノ基を有する化合物の使用量が式(2)で表されるトリアジン化合物1モルに対して0.1~3モルであることを特徴とする請求項1、2、3、4又は5記載のポリロタキサンの製造方法。
- 工程2は、式(2)で表されるトリアジン化合物と、少なくとも1つの三級アミノ基を有する化合物とを混合して反応させ、下記式(4)で表される四級アンモニウム塩を得る工程2-1と、
得られた式(4)で表される四級アンモニウム塩と、工程1で得られた擬ポリロタキサン水性分散体と、封鎖剤とを混合する工程2-2とを有する
ことを特徴とする請求項1、2、3、4、5又は6記載のポリロタキサンの製造方法。
- 式(4)で表される四級アンモニウム塩の使用量が式(1)で表される繰り返し構造単位を有する化合物の末端モル数に対して0.7~50倍モルであることを特徴とする請求項7又は8記載のポリロタキサンの製造方法。
- 工程2-2において、更に式(3)で示されるモルホリン化合物を添加することを特徴とする請求項7、8又は9記載のポリロタキサンの製造方法。
- 工程1で用いる式(1)で表される繰り返し構造単位を有する化合物の質量平均分子量が1000~50万であることを特徴とする請求項1、2、3、4、5、6、7、8、9又は10記載のポリロタキサンの製造方法。
- 工程1で用いる式(1)で表される繰り返し構造単位を有する化合物の両末端基は、ともに水酸基、カルボキシル基、アミノ基のいずれかであることを特徴とする請求項1、2、3、4、5、6、7、8、9、10又は11記載のポリロタキサンの製造方法。
- 工程1で用いる式(1)で表される繰り返し構造単位を有する化合物とシクロデキストリンとの質量比が1:2~1:5であることを特徴とする請求項1、2、3、4、5、6、7、8、9、10、11又は12記載のポリロタキサンの製造方法。
- 工程1で用いる水性媒体は、水性有機溶媒と水との混合溶媒又は水であることを特徴とする請求項1、2、3、4、5、6、7、8、9、10、11、12又は13記載のポリロタキサンの製造方法。
- 工程2は、無機塩の存在下で実施されることを特徴とする請求項1、2、3、4、5、6、7、8、9、10、11、12、13又は14記載のポリロタキサンの製造方法。
- 工程2は、水性有機溶媒の存在下で実施されることを特徴とする請求項1、2、3、4、5、6、7、8、9、10、11、12、13、14又は15記載のポリロタキサンの製造方法。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ES13839485.3T ES2642108T3 (es) | 2012-09-19 | 2013-09-09 | Método de fabricación de polirrotaxano |
| EP13839485.3A EP2899221B1 (en) | 2012-09-19 | 2013-09-09 | Method for manufacturing polyrotaxane |
| US14/428,917 US9487630B2 (en) | 2012-09-19 | 2013-09-09 | Method for manufacturing polyrotaxane |
| JP2014509958A JP5604610B2 (ja) | 2012-09-19 | 2013-09-09 | ポリロタキサンの製造方法 |
| KR1020157009130A KR102049137B1 (ko) | 2012-09-19 | 2013-09-09 | 폴리로탁산의 제조 방법 |
| CA2884974A CA2884974C (en) | 2012-09-19 | 2013-09-09 | Method for manufacturing polyrotaxane |
| CN201380048883.7A CN104662066B (zh) | 2012-09-19 | 2013-09-09 | 聚轮烷的制造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-205834 | 2012-09-19 | ||
| JP2012205834 | 2012-09-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014045921A1 true WO2014045921A1 (ja) | 2014-03-27 |
Family
ID=50341235
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/074247 WO2014045921A1 (ja) | 2012-09-19 | 2013-09-09 | ポリロタキサンの製造方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US9487630B2 (ja) |
| EP (1) | EP2899221B1 (ja) |
| JP (1) | JP5604610B2 (ja) |
| KR (1) | KR102049137B1 (ja) |
| CN (1) | CN104662066B (ja) |
| CA (1) | CA2884974C (ja) |
| ES (1) | ES2642108T3 (ja) |
| WO (1) | WO2014045921A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108484925A (zh) * | 2018-04-02 | 2018-09-04 | 王诒凡 | 一种高级轮烷及其合成方法 |
| JP2020062172A (ja) * | 2018-10-16 | 2020-04-23 | 住友ゴム工業株式会社 | ゴルフボール |
| JP2020062171A (ja) * | 2018-10-16 | 2020-04-23 | 住友ゴム工業株式会社 | ゴルフボール |
| WO2024014395A1 (ja) * | 2022-07-11 | 2024-01-18 | 株式会社シクロケムバイオ | ロタキサン化合物 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2781175T3 (es) | 2014-07-31 | 2020-08-31 | Novartis Ag | Subconjunto optimizado de células T que contienen un receptor de antígeno quimérico |
| WO2016168595A1 (en) | 2015-04-17 | 2016-10-20 | Barrett David Maxwell | Methods for improving the efficacy and expansion of chimeric antigen receptor-expressing cells |
| WO2019067786A1 (en) | 2017-09-29 | 2019-04-04 | The Regents Of The University Of California | MULTI-ARM POLYROTAXANE PLATFORM FOR PROTECTED ADMINISTRATION OF NUCLEIC ACIDS |
| IL280432B1 (en) | 2021-01-26 | 2024-01-01 | Israel Aerospace Ind Ltd | Air vehicle configuration |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005052026A1 (ja) | 2003-11-28 | 2005-06-09 | The University Of Tokyo | ポリロタキサン及びその製造方法 |
| WO2005080469A1 (ja) * | 2004-01-08 | 2005-09-01 | The University Of Tokyo | 架橋ポリロタキサンを有する化合物及びその製造方法 |
| JP2005255672A (ja) * | 2004-02-09 | 2005-09-22 | New Industry Research Organization | ハロゲン化シアヌル誘導体、その製造法およびその使用 |
| WO2009041666A1 (ja) * | 2007-09-28 | 2009-04-02 | Nanodex Inc. | 葉酸修飾されたシクロデキストリン化合物、その製造方法、標的指向性薬物送達システム用の薬物送達剤、医薬組成物及び造影剤 |
| WO2012124220A1 (ja) * | 2011-03-14 | 2012-09-20 | 住友精化株式会社 | 親水性修飾ポリロタキサン組成物 |
| WO2013147301A1 (ja) * | 2012-03-30 | 2013-10-03 | 宇部興産株式会社 | ブロック基を有するポリロタキサンの製造方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2678538A1 (en) | 2007-03-06 | 2008-09-12 | Advanced Softmaterials, Inc. | Polyrotaxanes and material having polyrotaxane, crosslinked polyrotaxanes and material having the crosslinked polyrotaxane, and processes for producing these |
-
2013
- 2013-09-09 KR KR1020157009130A patent/KR102049137B1/ko active IP Right Grant
- 2013-09-09 CN CN201380048883.7A patent/CN104662066B/zh active Active
- 2013-09-09 CA CA2884974A patent/CA2884974C/en active Active
- 2013-09-09 EP EP13839485.3A patent/EP2899221B1/en active Active
- 2013-09-09 US US14/428,917 patent/US9487630B2/en active Active
- 2013-09-09 WO PCT/JP2013/074247 patent/WO2014045921A1/ja active Application Filing
- 2013-09-09 JP JP2014509958A patent/JP5604610B2/ja active Active
- 2013-09-09 ES ES13839485.3T patent/ES2642108T3/es active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005052026A1 (ja) | 2003-11-28 | 2005-06-09 | The University Of Tokyo | ポリロタキサン及びその製造方法 |
| JP2005154675A (ja) | 2003-11-28 | 2005-06-16 | Kozo Ito | ポリロタキサン及びその製造方法 |
| WO2005080469A1 (ja) * | 2004-01-08 | 2005-09-01 | The University Of Tokyo | 架橋ポリロタキサンを有する化合物及びその製造方法 |
| JP2005255672A (ja) * | 2004-02-09 | 2005-09-22 | New Industry Research Organization | ハロゲン化シアヌル誘導体、その製造法およびその使用 |
| WO2009041666A1 (ja) * | 2007-09-28 | 2009-04-02 | Nanodex Inc. | 葉酸修飾されたシクロデキストリン化合物、その製造方法、標的指向性薬物送達システム用の薬物送達剤、医薬組成物及び造影剤 |
| WO2012124220A1 (ja) * | 2011-03-14 | 2012-09-20 | 住友精化株式会社 | 親水性修飾ポリロタキサン組成物 |
| WO2013147301A1 (ja) * | 2012-03-30 | 2013-10-03 | 宇部興産株式会社 | ブロック基を有するポリロタキサンの製造方法 |
Non-Patent Citations (2)
| Title |
|---|
| "TSKgel SuperAWM-H", TOSOH CORPORATION |
| See also references of EP2899221A4 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108484925A (zh) * | 2018-04-02 | 2018-09-04 | 王诒凡 | 一种高级轮烷及其合成方法 |
| JP2020062172A (ja) * | 2018-10-16 | 2020-04-23 | 住友ゴム工業株式会社 | ゴルフボール |
| JP2020062171A (ja) * | 2018-10-16 | 2020-04-23 | 住友ゴム工業株式会社 | ゴルフボール |
| JP7218537B2 (ja) | 2018-10-16 | 2023-02-07 | 住友ゴム工業株式会社 | ゴルフボール |
| JP7218536B2 (ja) | 2018-10-16 | 2023-02-07 | 住友ゴム工業株式会社 | ゴルフボール |
| WO2024014395A1 (ja) * | 2022-07-11 | 2024-01-18 | 株式会社シクロケムバイオ | ロタキサン化合物 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20150240036A1 (en) | 2015-08-27 |
| KR20150058290A (ko) | 2015-05-28 |
| ES2642108T3 (es) | 2017-11-15 |
| CN104662066A (zh) | 2015-05-27 |
| CA2884974A1 (en) | 2014-03-27 |
| US9487630B2 (en) | 2016-11-08 |
| JP5604610B2 (ja) | 2014-10-08 |
| JPWO2014045921A1 (ja) | 2016-08-18 |
| EP2899221A1 (en) | 2015-07-29 |
| EP2899221A4 (en) | 2016-04-27 |
| CA2884974C (en) | 2020-03-10 |
| CN104662066B (zh) | 2018-05-18 |
| KR102049137B1 (ko) | 2020-01-08 |
| EP2899221B1 (en) | 2017-08-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5604610B2 (ja) | ポリロタキサンの製造方法 | |
| JP6111072B2 (ja) | 精製ポリロタキサンの製造方法 | |
| EP2687544B1 (en) | Production method for hydrophilic modified polyrotaxane | |
| JP6013191B2 (ja) | 擬ポリロタキサン水性分散体の製造方法 | |
| JP6713702B2 (ja) | 擬ポリロタキサン水性分散体の製造方法 | |
| JP6013318B2 (ja) | 粉末状親水性修飾ポリロタキサンの製造方法 | |
| JP6013319B2 (ja) | ポリロタキサン組成物 | |
| JP6013189B2 (ja) | 擬ポリロタキサンの製造方法 | |
| JP6013190B2 (ja) | 擬ポリロタキサンの製造方法 | |
| JPH0625307A (ja) | ゲスト高分子がエンドキャップされたα−サイクロデキストリンの包接化合物及びゲスト高分子がエンドキャップされたサイクロデキストリンの包接化合物の製造方法 | |
| JP2018016671A (ja) | アルキル化ポリロタキサンの製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2014509958 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13839485 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2884974 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14428917 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2013839485 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013839485 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20157009130 Country of ref document: KR Kind code of ref document: A |