WO2013175545A1 - 加熱装置 - Google Patents
加熱装置 Download PDFInfo
- Publication number
- WO2013175545A1 WO2013175545A1 PCT/JP2012/007321 JP2012007321W WO2013175545A1 WO 2013175545 A1 WO2013175545 A1 WO 2013175545A1 JP 2012007321 W JP2012007321 W JP 2012007321W WO 2013175545 A1 WO2013175545 A1 WO 2013175545A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- weight
- silica
- heating element
- fiber
- heating
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B3/00—Ohmic-resistance heating
- H05B3/02—Details
- H05B3/06—Heater elements structurally combined with coupling elements or holders
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C13/00—Fibre or filament compositions
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C25/00—Surface treatment of fibres or filaments made from glass, minerals or slags
- C03C25/002—Thermal treatment
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C25/00—Surface treatment of fibres or filaments made from glass, minerals or slags
- C03C25/10—Coating
- C03C25/42—Coatings containing inorganic materials
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/62227—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products obtaining fibres
- C04B35/62231—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products obtaining fibres based on oxide ceramics
- C04B35/6224—Fibres based on silica
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/628—Coating the powders or the macroscopic reinforcing agents
- C04B35/62844—Coating fibres
- C04B35/62847—Coating fibres with oxide ceramics
- C04B35/62849—Silica or silicates
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/63—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B using additives specially adapted for forming the products, e.g.. binder binders
- C04B35/632—Organic additives
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B3/00—Ohmic-resistance heating
- H05B3/20—Heating elements having extended surface area substantially in a two-dimensional plane, e.g. plate-heater
- H05B3/22—Heating elements having extended surface area substantially in a two-dimensional plane, e.g. plate-heater non-flexible
- H05B3/26—Heating elements having extended surface area substantially in a two-dimensional plane, e.g. plate-heater non-flexible heating conductor mounted on insulating base
- H05B3/265—Heating elements having extended surface area substantially in a two-dimensional plane, e.g. plate-heater non-flexible heating conductor mounted on insulating base the insulating base being an inorganic material, e.g. ceramic
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C2213/00—Glass fibres or filaments
- C03C2213/02—Biodegradable glass fibres
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3205—Alkaline earth oxides or oxide forming salts thereof, e.g. beryllium oxide
- C04B2235/3206—Magnesium oxides or oxide-forming salts thereof
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3205—Alkaline earth oxides or oxide forming salts thereof, e.g. beryllium oxide
- C04B2235/3208—Calcium oxide or oxide-forming salts thereof, e.g. lime
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/72—Products characterised by the absence or the low content of specific components, e.g. alkali metal free alumina ceramics
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/77—Density
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/96—Properties of ceramic products, e.g. mechanical properties such as strength, toughness, wear resistance
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/96—Properties of ceramic products, e.g. mechanical properties such as strength, toughness, wear resistance
- C04B2235/9669—Resistance against chemicals, e.g. against molten glass or molten salts
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B2203/00—Aspects relating to Ohmic resistive heating covered by group H05B3/00
- H05B2203/012—Heaters using non- flexible resistive rods or tubes not provided for in H05B3/42
Definitions
- the present invention relates to a heating device having a heating element such as an electric heater and a heat insulating support.
- an electric heater structure configured by supporting a coiled heating wire (electric heating coil) wound spirally on a heat insulating material, for example, kneading obtained by adding an inorganic binder to ceramic fiber or the like
- a heat insulating material for example, kneading obtained by adding an inorganic binder to ceramic fiber or the like
- An object of the present invention is to provide a heating apparatus including a support that includes biosoluble inorganic fibers and that suppresses the reaction between the fibers and the heating element at high temperatures.
- the present inventors have found that when a biosoluble fiber is used as a heat insulating material for supporting an electrothermal coil, the fiber that contacts the high-temperature electrothermal coil melts, and specifically, by preventing this contact, The present invention was completed by suppressing melting by interposing a component that hardly reacts with both the soluble fiber and the heating element at a high temperature.
- the following heating device is provided. 1. A heating element; A support containing biosoluble inorganic fibers that holds the heating element; With The heating apparatus in which the biosoluble inorganic fiber is not in direct contact with the heating element or the contact between the biosoluble inorganic fiber and the heating element is reduced. 2. 2. The heating apparatus according to 1, wherein the biosoluble inorganic fiber has the following composition.
- the heating apparatus according to 1 or 2 wherein the biosoluble inorganic fiber has the following composition. 50 to 82% by weight of SiO 2 , ZrO 2 , Al 2 O 3 and TiO 2 Total of 18-50% by weight of alkali metal oxide and alkaline earth metal oxide 4). 4.
- the biosoluble inorganic fiber is Mg silicate fiber having the following composition or Ca silicate fiber having the following composition.
- At least a surface of the support in contact with the heating element is coated (2)
- a biosoluble inorganic fiber on the surface of the support at least in contact with the heating element is coated (3) 5.
- the heating element is covered. 6.
- the heating apparatus according to any one of 6 to 8, wherein the silica is one form of silica selected from colloid, powder and metal alkoxide, or a combination of two or more forms of silica. 10.
- the silica-containing treatment agent includes a chelating agent not containing metal ions.
- the silica-containing treatment agent contains an acid. 12
- the silica is acidic type colloidal silica or cationic type colloidal silica. 13.
- the heating apparatus according to any one of 1 to 12, wherein when the temperature of the heating element is 1100 ° C., a reaction between the biosoluble inorganic fiber and the heating element is suppressed. 14 14. The heating apparatus according to any one of 1 to 13, wherein the biosoluble inorganic fiber is heat-treated.
- a heating device including a support that includes biologically soluble inorganic fibers and that suppresses the reaction between the fibers and the heating element at high temperatures.
- the heating device of the present invention includes a heating element and a support for holding the heating element, and the support is made of a heat insulating material containing biosoluble inorganic fibers.
- the heating device of the present invention is configured so that the biosoluble inorganic fiber is preferably not in direct contact so as to reduce the portion in contact with the heating element.
- a silica-containing treatment agent for example, together with a fluid
- a silica-containing treatment agent for example, together with a fluid
- the surface support biosoluble inorganic fibers
- This treatment hardens the surface and prevents powder from falling off the surface.
- the coating of the support may cover only the support (biosoluble inorganic fiber) that contacts the heating element, or may cover the entire surface of the support.
- the silica-containing treatment agent soaks into the majority of the support, whereas in the coating, the majority of the silica-containing treatment agent is on the support to form a film.
- the coating liquid applied to the support can contain an inorganic binder, an organic thickener, a solvent, and the like in order to adjust the viscosity, and these amounts may be adjusted as appropriate.
- the silica-containing treatment agent can contain a component that does not react with the biosoluble fiber or the heating element at a high temperature in addition to silica.
- Silica may be in the form of a colloid, an inorganic powder, a metal alkoxide, or the like, and one or more types may be combined.
- silica-containing treatment agent preferably has less alumina and more silica.
- the content of silica is preferably 20% by weight or more, more preferably 40% by weight or more, more preferably 60% by weight or more, still more preferably 80% by weight or more, and 90% by weight or more. Further, only silica containing inevitable impurities may be used.
- a heating element may be coated. You may coat
- the heating element is covered with a ceramic tube, a quartz tube or the like. The space between the heating element and the coating can be filled with an insulator.
- the heating element may be coated with the same components as the silica-containing treatment agent used for coating the support and the fibers. Also in this case, it is preferable that the amount of alumina is small and the amount of silica is large.
- the biologically soluble inorganic fiber constituting the support is in contact with the heating element, the biologically soluble inorganic fiber and the heating element start to react at high temperatures. This is considered to be caused by the composition of the biosoluble inorganic fiber (particularly alkali metal and / or alkaline earth metal) and the composition of the heating element (particularly the oxide film formed on the outer periphery of the heating element).
- the contact between the biosoluble inorganic fiber and the heating element is reduced as described above, such a reaction can be suppressed and preferably does not react.
- the temperature of the heating element is preferably 400 ° C. or higher, more preferably 800 ° C. or higher, further preferably 1000 ° C. or higher, particularly preferably 1100 ° C. or higher, and the reaction between the biosoluble fiber and the heating element. Is suppressed.
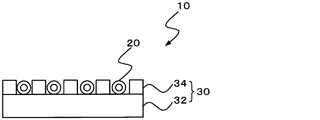
- the heating device 10 includes a heating element 20 and a support 30, and the support 30 includes a base 32 and a side wall 34, which are bonded together.
- the heating element 20 is disposed between the side walls 34 on the base 32.
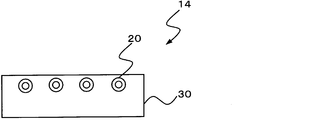
- the heating device 12 of FIG. 1B is the same as the heating device 10 of FIG. 1A except that a base 32 and a side wall 34 are integrally formed.
- the heating element 20 is embedded in the support 30.
- Each of the heating devices 10, 12, and 14 of FIGS. 1A to 1C is configured so that the biosoluble inorganic fiber and the heating element do not contact each other (not shown).
- Fig. 2 shows a side view of a typical heating device.
- a plurality of groove portions 40 extending linearly are formed in parallel at a predetermined interval.
- the support 30 includes a base portion and a side wall, and the base portion and the side wall form a groove 40.
- a heating element (not shown) is disposed in the groove.
- heating device there is a device described in Japanese Patent Application Laid-Open No. 2011-181258.
- a device for closing a groove as shown in FIG. 5 of this publication, a cylindrical or semi-cylindrical device as shown in FIGS.
- the support used in the heating device of the present invention is configured to include inorganic fibers.
- it is composed of 20 to 99% by weight of inorganic fibers (preferably 30 to 95% by weight, more preferably 50 to 95% by weight).
- the inorganic fiber is preferably composed mainly of SiO 2 , CaO, and MgO.
- a main component means that 90 weight% or more or 95 weight% or more is comprised combining these.
- the inorganic fiber may be a biosoluble fiber having the following composition. Total of SiO 2 , ZrO 2 , Al 2 O 3 and TiO 2 50 wt% to 82 wt% Total of 18% to 50% by weight of alkali metal oxide and alkaline earth metal oxide
- the inorganic fiber may be a biosoluble fiber having the following composition. SiO 2 50 to 82% by weight Total 10 to 43% by weight of CaO and MgO
- Biologically soluble fibers can be broadly classified into Mg silicate fibers rich in MgO and Ca silicate fibers rich in CaO.
- the following composition can be illustrated as Mg silicate fiber. SiO 2 66-82% by weight CaO 1-9% by weight MgO 10-30% by weight Al 2 O 3 3 wt% or less Other oxides Less than 2 wt%
- the following composition can be illustrated as Ca silicate fiber.
- SiO 2 66-82 wt% (for example, it can be 68-80 wt%, 70-80 wt%, 71-80 wt% or 71-76 wt%)
- CaO 10-34% by weight (for example, it can be 18-32% by weight, 20-30% by weight, 20-27% by weight or 21-26% by weight)
- MgO 3 wt% or less eg, 1 wt% or less
- Al 2 O 3 5% by weight or less for example, 3.5% by weight or less, 3.4% by weight or less, or 3% by weight or less.
- Other oxides ⁇ 2% by weight
- the total of SiO 2 , CaO, MgO and Al 2 O 3 may be more than 98 wt% or more than 99 wt%.
- the biosoluble inorganic fiber includes alkali metal oxides (K 2 O, Na 2 O, Li 2 O, etc.), Sc, La, Ce, Pr, Nd, Sm, Eu, Gd, Tb as other components. , Dy, Ho, Er, Tm, Yb, Lu, Y, or a mixture thereof, Fe 2 O 3 , ZrO 2 , TiO 2 , P 2 O 5 , B 2 O 3 , MnO, One or more of ZnO, SrO, BaO, Cr 2 O 3 and the like may or may not be included. Other oxides may be 1.0 wt% or less, 0.2 wt% or less, or 0.1 wt% or less, respectively.
- the alkali metal oxide may contain 1.0% by weight or less, 0.2% by weight or less, or 0.1% by weight or less of each oxide.
- the total of the alkali metal oxides may be 1.0% by weight or less, 0.2% by weight or less, or 0.1% by weight or less.
- the biosoluble fiber may further contain other components in addition to SiO 2 and an alkaline earth metal oxide (for example, at least one of MgO and CaO).
- an alkaline earth metal oxide for example, at least one of MgO and CaO.
- alumina Al 2 O 3
- titania TiO 2
- zirconia ZrO 2
- iron oxide Fe 2 O 3
- manganese oxide MnO
- K 2 O potassium oxide
- the biologically soluble inorganic fiber is, for example, an inorganic fiber having a physiological saline dissolution rate at 40 ° C. of 1% or more.
- the physiological saline dissolution rate is measured, for example, as follows. That is, first, 1 g of a sample prepared by pulverizing inorganic fibers to 200 mesh or less and 150 mL of physiological saline are placed in an Erlenmeyer flask (volume: 300 mL) and placed in an incubator at 40 ° C. Next, a horizontal vibration of 120 revolutions per minute is continuously applied to the Erlenmeyer flask for 50 hours.
- the concentration (mg / L) of each element contained in the filtrate obtained by filtration is measured with an ICP emission analyzer. Then, based on the measured concentration of each element and the content (% by mass) of each element in the inorganic fiber before dissolution, the physiological saline dissolution rate (%) is calculated. That is, for example, when the measurement element is silicon (Si), magnesium (Mg), calcium (Ca), and aluminum (Al), the physiological saline dissolution rate C (%) is calculated by the following formula.
- C (%) [filtrate amount (L) ⁇ (a1 + a2 + a3 + a4) ⁇ 100] / [mass of inorganic fiber before dissolution (mg) ⁇ (b1 + b2 + b3 + b4) / 100].
- a1, a2, a3 and a4 are the measured concentrations of silicon, magnesium, calcium and aluminum (mg / L), respectively, and b1, b2, b3 and b4 are respectively in the inorganic fibers before dissolution. It is content (mass%) of silicon, magnesium, calcium, and aluminum.
- the biosoluble inorganic fiber is preferably heated in advance at a constant temperature in the range of 300 to 1300 ° C., because warpage of the resulting board can be suppressed.
- the heat treatment temperature of the Mg silicate fiber is, for example, 600 to 1300 ° C., preferably 800 to 1300 ° C., and more preferably 850 to 1000 ° C.
- the heat treatment temperature of the Ca silicate fiber is, for example, 820 to 1300 ° C., preferably 830 to 1300 ° C., more preferably 840 to 1000 ° C., and most preferably 850 to 1000 ° C.
- the heat treatment temperature may be a temperature equal to or higher than the crystallization temperature of untreated fibers, for example.
- the untreated fiber is subjected to a heat treatment at a temperature equal to or higher than the crystallization temperature to obtain a heat-treated fiber partially crystallized. Since the crystallization temperature varies depending on the chemical composition of the untreated fiber, it cannot be generally determined, but is, for example, 600 to 1300 ° C., 600 to 1100 ° C., or 800 to 1000 ° C.
- the heat-treated fiber When the heat-treated fiber is the above-mentioned Ca silicate fiber, the heat-treated fiber partially crystallized includes, for example, wollastonite crystals. Furthermore, other crystals may be included, for example, one or more crystals selected from the group consisting of wollastonite, cristobalite and tridymite.
- the heat-treated fiber When the heat-treated fiber is the above-described Mg silicate fiber, the heat-treated fiber partially crystallized includes, for example, enstatite crystals. Still other crystals may be included, for example, one or more crystals selected from the group consisting of enstatite, diopsite, cristobalite, and tridymite.
- the heat treatment time is not particularly limited as long as the effect by the above heat treatment can be obtained.
- the heat treatment time is, for example, 1 minute to 48 hours, preferably 3 minutes to 24 hours.
- the heat treatment time is, for example, 3 minutes to 8 hours, preferably 5 minutes to 3 hours.
- the support may contain reinforcing fibers such as silica-alumina fiber, silica fiber, alumina fiber, zirconia fiber, glass fiber, rock wool, etc. in addition to the above-mentioned biosoluble inorganic fiber.
- reinforcing fibers such as silica-alumina fiber, silica fiber, alumina fiber, zirconia fiber, glass fiber, rock wool, etc. in addition to the above-mentioned biosoluble inorganic fiber.
- the support can contain an organic binder, an inorganic binder, and the like in addition to the inorganic fibers. As long as the effects of the present invention are not impaired, those usually used can be used.
- Starch, acrylic resin, polyacrylamide, pulp, acrylic emulsion and the like are used as the organic binder, and anionic colloidal silica is used as the inorganic binder. Examples thereof include colloidal silica such as cationic colloidal silica, fumed silica, zirconia sol, titania sol, alumina sol, bentonite and kaolin.
- the amount is preferably 1 to 50% by weight of the support, more preferably 3 to 30% by weight, and still more preferably 5 to 20% by weight.
- the inorganic powder is, for example, ceramic powder such as zircon, calcium carbonate, cordierite, wollastonite, silica, alumina, titania, zirconia, silicon nitride, and silicon carbide, and carbon powder such as carbon black.
- the amount of the inorganic powder is usually 0% to 80% by weight of the support, more preferably 0% to 50% by weight, and still more preferably 0% to 30% by weight.
- the heating element used in the heating device is not particularly limited, and examples thereof include iron-chromium-aluminum or nickel-chromium metal heating elements. Further, examples of such a metal heating element include a coil shape and a wave shape.
- a heat insulating member (standard shape, board) is manufactured from biosoluble inorganic fibers.
- a slurry containing raw materials such as inorganic fibers and a binder is produced by dehydration molding.
- the member may be cured before forming the support (for example, the base and the side wall are bonded together in FIG. 1A, or processed into a desired shape (cutting etc.) in FIG. 1B). ), A support may be formed and then cured.
- 1C can be manufactured by setting a heating element in a predetermined mold at the time of dehydration molding, and curing treatment after molding.
- dehydration molding may be performed a plurality of times.
- the curing treatment may be performed during the dehydration molding process.
- colloidal silica can be used as a curing agent.
- colloidal silica include alkali type colloidal silica (pH 8 to 10), acidic type colloidal silica (pH 2 to 6), and cationic type colloidal silica.
- the curing process is performed by, for example, immersing a member or a support (hereinafter simply referred to as a member) in a curing process liquid, or applying (hardening, rolling, etc.) or spraying (showering, spraying, etc.) the curing process liquid. ) And dried after the curing treatment. Since the solvent evaporates from the surface during drying, the solvent moves to the surface, and the colloidal silica also moves to the surface. Since the colloidal silica remains even if the solvent evaporates, the colloidal silica is unevenly distributed near the surface, and as a result, the surface is cured.
- the solvent water, monovalent alcohols such as ethanol and propanol, divalent alcohols such as ethylene glycol, and the like can be used.
- the amount of colloidal silica contained in the member by the curing treatment is an amount that increases the density of the member by, for example, 10% to 150%, preferably 10% to 100%, more preferably 10% to 50%.
- the member is immersed in colloidal silica having a solid content ratio of 10 to 30% for 1 second to several minutes, but this is not a limitation. You may process once or twice or more.
- the moisture content of the cured member is usually about 5% by weight or less. Confirm the moisture content by the weight before and after drying.
- the member biologically inorganic fiber
- the hardness of the member is also improved, so that the strength and handling properties (the trace of the hand when grasped, the peeling during processing) , Problems such as falling corners and falling off when cutting).
- alkaline earth metal ions particularly Ca ions and Mg ions are eluted in the curing treatment liquid.
- these ions react with the curing agent on the surface of the member and are heated to 800 ° C. or higher, cracks may be caused.
- the member is treated with a chelating agent not containing metal ions and a curing agent. It is considered that the chelating agent captures the eluted alkaline earth metal, thereby suppressing the reaction between the alkaline earth metal and the curing agent and preventing the occurrence of cracks. Since the chelating agent has two or more electron donating groups, a metal chelate compound can be produced. Examples of the electron donating group include a carboxy group and a hydroxyl group.
- the chelating agent used in the present invention does not contain alkaline earth metals or alkali metals.
- the chelating agent examples include a chelating agent having a composition of EDTA (ethylenediaminetetraacetic acid) ⁇ H ⁇ 3 (NH 4 ), a chelating agent having a composition of EDTA ⁇ 2H ⁇ 2 (NH 4 ), and the like.
- colloidal silica As the curing agent used in combination with the chelating agent, colloidal silica can be used, and alkali type colloidal silica, acidic type colloidal silica, cationic type colloidal silica and the like can be used.
- concentration of the chelating agent can be appropriately set, but when alkali type colloidal silica is used as the curing agent, it is preferably 0.5% by weight or more with respect to the solid content 10% by weight curing treatment liquid.
- the member may be treated with a curing treatment solution having a pH of 6 or less (preferably pH 1 to 6, more preferably 2 to 5, particularly preferably pH 3 to 5).
- a curing treatment solution having a pH of 6 or less (preferably pH 1 to 6, more preferably 2 to 5, particularly preferably pH 3 to 5).
- the member is treated with an acid and a curing agent.
- the acid may be a weak acid or a strong acid.
- the weak acid include citric acid, malic acid, tartaric acid, salicylic acid, glycolic acid, lactic acid, mandelic acid, benzylic acid, coumaric acid, and acetic acid.
- Strong acids include sulfuric acid, hydrochloric acid, and nitric acid. Is mentioned. Since there is no problem of generated gas, a weak acid is preferable.
- colloidal silica of an acidic type may be used.
- acidic type colloidal silica it is possible to use commercially available products such as silica doll 20A (manufactured by Nippon Chemical Industry Co., Ltd., solid content ratio 20%), cataloid SN (manufactured by JGC Catalysts & Chemicals Co., Ltd., solid content ratio 20%). it can.
- Examples of those having both the chelating effect and the property of making pH acidic include hydroxy acids such as citric acid, malic acid, tartaric acid and salicylic acid.
- Cationic colloidal silica is colloidal silica having a positive charge on the surface.
- the surface of the colloidal silica or the inside thereof may contain a compound of a polyvalent metal ion such as an aluminum ion or an organic cationic compound so that the surface of the silica particles is cationically charged.
- commercially available products such as Snowtex AK (manufactured by Nissan Chemical Industries, Ltd., solid content ratio 18%) can be used.
- the curing member is obtained by the above treatment, the curing agent is impregnated from the surface.
- a heating device is completed by attaching a heating element to the support treated as described above.
- the manufacturing method of the apparatus of this invention is not limited above, It can manufacture by a well-known method.
- Production Example 1 ⁇ Manufacture of board (insulation material)> Biologically soluble fiber A (SiO 2 content is 73% by mass, CaO content is 25% by mass, MgO content is 0.3% by mass, Al 2 O 3 content is 2% by mass) (saline dissolution rate 10.4%) 90% by weight, and a board (referred to as an organic board) having a length of 300 mm, a width of 300 mm, and a thickness of 50 mm (density 250 kg / m 3 ) made of starch, polyacrylamide (organic binder) and colloidal silica (inorganic binder). Manufactured.
- a board referred to as an organic board having a length of 300 mm, a width of 300 mm, and a thickness of 50 mm (density 250 kg / m 3 ) made of starch, polyacrylamide (organic binder) and colloidal silica (inorganic binder).
- biosoluble fiber A colloidal silica, alumina sol, bentonite (inorganic binder), inorganic powder (silica, zircon, calcium carbonate, zirconia, cordierite, wollastonite, etc.), acrylamide (organic binder)
- a board having a length of 300 mm, a width of 300 mm, and a thickness of 50 mm (density 250 kg / m 3 ) was manufactured (referred to as an inorganic board).
- Production Example 3 Manufacture of board (insulation material)> Instead of the biosoluble fiber A, inorganic fiber C (SiO 2 content is 47% by mass, Al 2 O 3 content is 52% by mass) (saline dissolution rate 0.1%) is used. In the same manner as in Production Example 1, organic and inorganic boards were produced.
- Processing example 1 ⁇ Curing treatment with silica>
- the organic board and inorganic board (density 250 kg / m 3 ) obtained in Production Examples 1 to 3 were treated (immersion for 1 to 10 seconds) with a curing solution (pH about 9, solvent: water) made of alkali-type colloidal silica. The surface was cured and dried. The density was 300 kg / m 3 . Strength improved after curing.
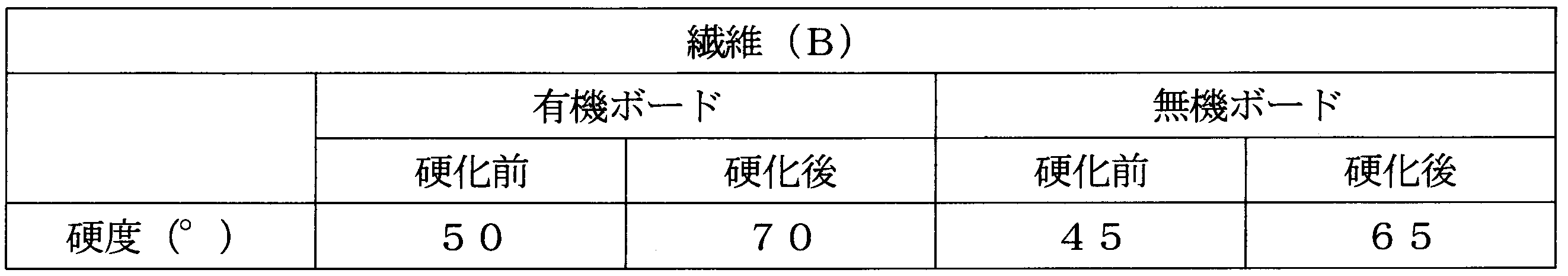
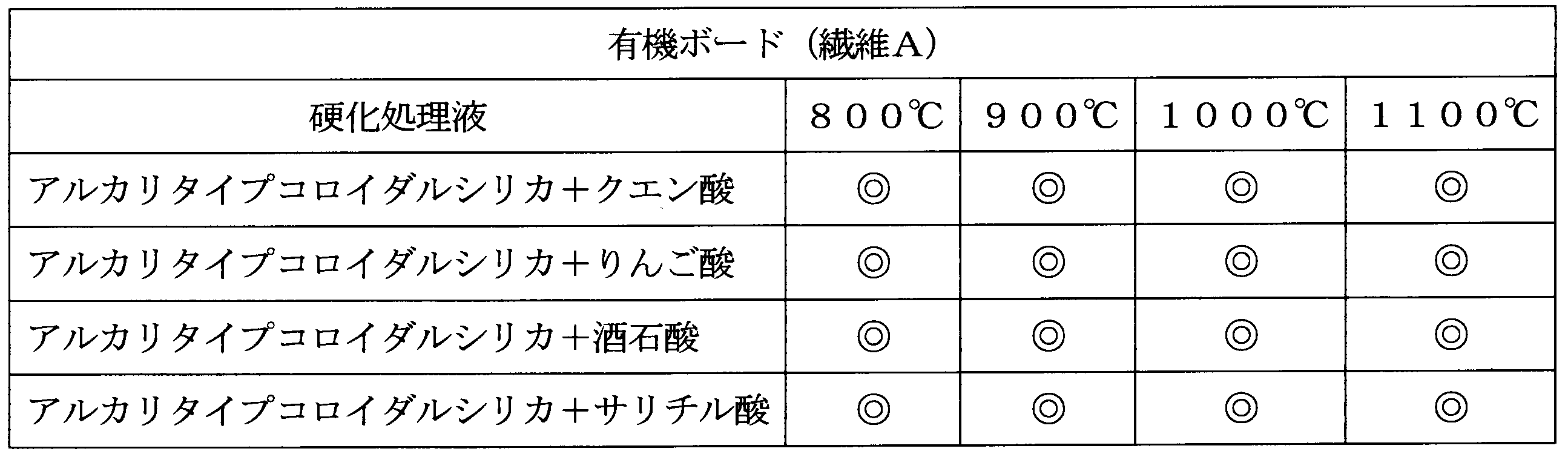
- the hardness of the cured organic and inorganic boards was measured using a hardness meter (manufactured by ASKER: Asker rubber hardness meter C type). The results are shown in Table 1.
- Processing example 2 ⁇ Curing treatment with silica> Except for using the organic board and inorganic board obtained in Production Example 2, the composition was cured and the hardness was measured in the same manner as in Treatment Example 1. The results are shown in Table 2.
- Processing example 3 ⁇ Curing treatment with silica> It was cured in the same manner as in Processing Example 1 except that the organic board and inorganic board obtained in Production Example 3 were used. Strength improved after curing.
- Processing example 4 [Curing treatment with silica and chelating agent] ⁇ Curing treatment>
- the organic and inorganic boards obtained in Production Example 1 were treated with a curing treatment solution (pH about 9, solvent: water) composed of EDTA diammonium salt and alkali type colloidal silica to cure and dry the surface.
- the chelating agent was used at 1% by weight with respect to a solid content 10% by weight curing treatment liquid.
- Processing example 5 [Curing treatment with silica and acid] ⁇ Curing treatment>
- the organic board obtained in Production Example 1 was treated with a curing treatment liquid composed of an acid and alkali type colloidal silica to cure and dry the surface.
- acids citric acid, malic acid, tartaric acid, and salicylic acid were used in an amount of about 1% by weight based on a 10% solid content curing treatment liquid, and the pH of the treatment liquid was adjusted to 3-4.
- Processing example 6 [Curing treatment with acidic type colloidal silica and cationic type colloidal silica] ⁇ Curing treatment>
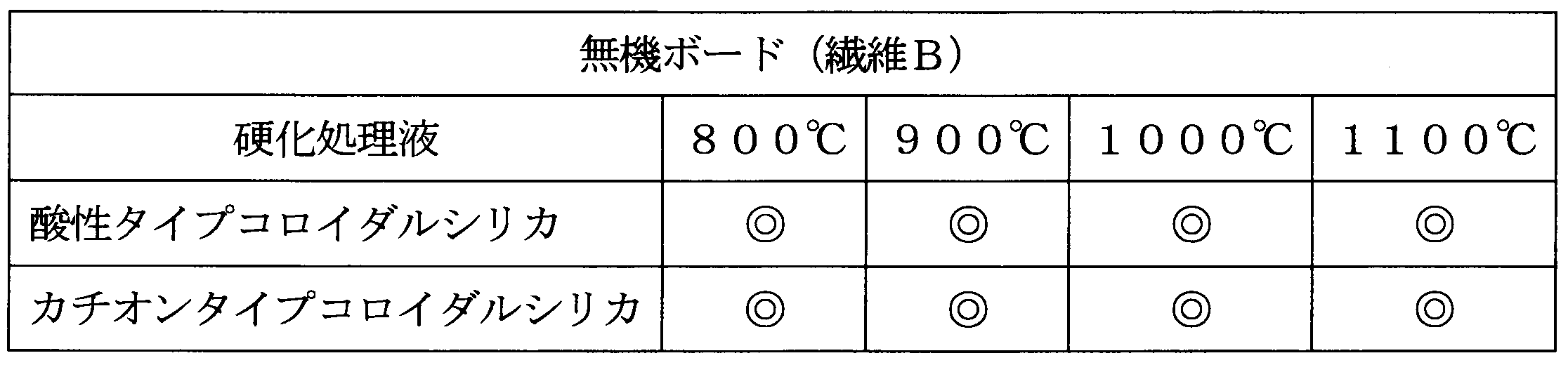
- the inorganic board obtained in Production Example 2 was converted into acidic type colloidal silica (silica doll 20A (manufactured by Nippon Chemical Industry Co., Ltd.), pH 2-4) or cationic type colloidal silica (Snowtex AK, manufactured by Nissan Chemical Industries, Ltd.).
- the surface was cured by treatment with (curing agent) and dried.
- ⁇ Heating test> It carried out like the processing example 4. The results are shown in Table 5.
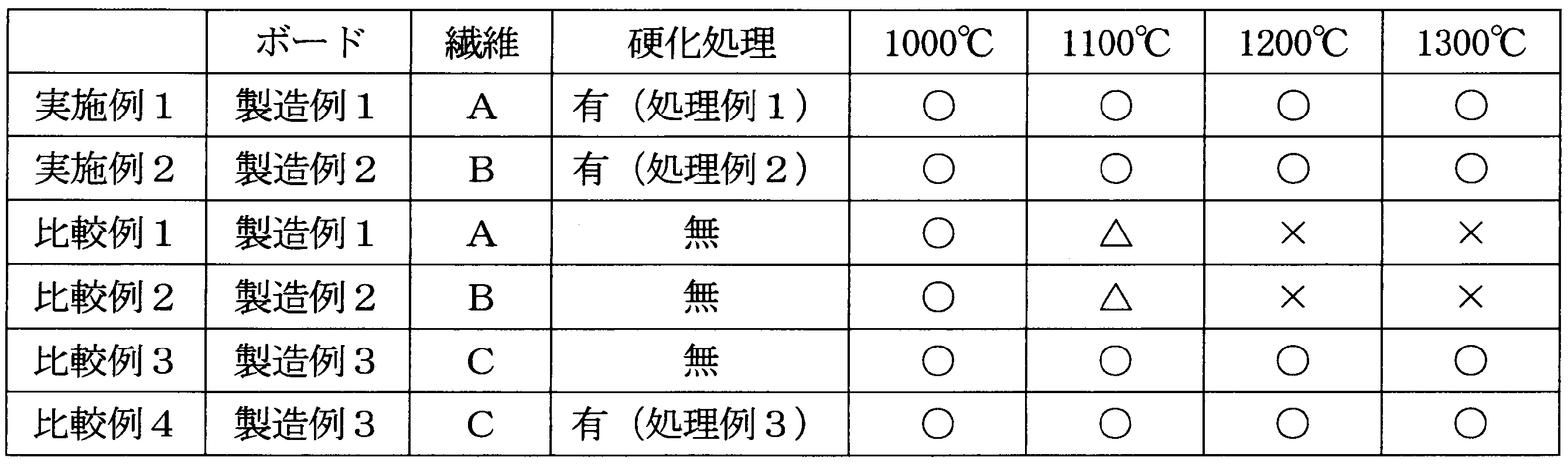
- Example 1 ⁇ Stability test of cured inorganic board at high temperature> A heater wire was placed on the inorganic board (heat insulating material) subjected to the curing treatment obtained in Treatment Example 1, and heat treatment was performed in this state at a temperature of 1000 ° C. to 1300 ° C. for 8 hours. The appearance of the heated board and heater wire was observed to confirm the presence or absence of reaction. The case where there was no reaction between the heater wire and the board was indicated as ⁇ , the case where a small amount of the board fiber was attached to the heater wire was indicated as ⁇ , and the case where the amount of the board fiber attached was large as ⁇ . The results are shown in Table 6.
- Example 2 and Comparative Examples 1 to 4 The stability of the heater wire and the board at high temperature was tested in the same manner as in Example 1 except that the inorganic board shown in Table 6 was used instead of the inorganic board subjected to the curing treatment obtained in Treatment Example 1. . The results are shown in Table 6.
- the heating device of the present invention can be used for various applications such as a firing furnace for electronic parts, melting of glass, an electric furnace for firing, a muffle furnace, a ceramic furnace, and various industrial furnaces.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Ceramic Engineering (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Manufacturing & Machinery (AREA)
- Life Sciences & Earth Sciences (AREA)
- Inorganic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Geochemistry & Mineralogy (AREA)
- Structural Engineering (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Thermal Sciences (AREA)
- Physics & Mathematics (AREA)
- Materials For Medical Uses (AREA)
- Inorganic Fibers (AREA)
- Resistance Heating (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
- Thermotherapy And Cooling Therapy Devices (AREA)
Abstract
Description
本発明によれば、以下の加熱装置が提供される。
1.発熱体と、
前記発熱体を保持する、生体溶解性無機繊維を含む支持体と、
を備え、
前記生体溶解性無機繊維が前記発熱体と直接接触しないか又は前記生体溶解性無機繊維と前記発熱体との接触が低減された加熱装置。
2.前記生体溶解性無機繊維が、以下の組成である1記載の加熱装置。
SiO2 50~82重量%
CaOとMgOとの合計 10~43重量%
3.前記生体溶解性無機繊維が、以下の組成である1又は2記載の加熱装置。
SiO2とZrO2とAl2O3とTiO2との合計 50~82重量%
アルカリ金属酸化物とアルカリ土類金属酸化物との合計 18~50重量%
4.前記生体溶解性無機繊維が、以下の組成を有するMgシリケート繊維、又は以下の組成を有するCaシリケート繊維である1~3のいずれか記載の加熱装置。
[Mgシリケート繊維]
SiO2 66~82重量%
CaO 1~9重量%
MgO 10~30重量%
Al2O3 0~3重量%
他の酸化物 0重量%以上2重量%未満
[Caシリケート繊維]
SiO2 66~82重量%
CaO 10~34重量%
MgO 0~3重量%
Al2O3 0~5重量%
他の酸化物 0重量%以上2重量%未満
5.以下の構成(1)~(3)のいずれかにより、前記生体溶解性無機繊維が前記発熱体と直接接触しないか又は前記生体溶解性無機繊維と前記発熱体との接触が低減された1~4のいずれか記載の加熱装置。
(1)前記支持体の少なくとも前記発熱体と接する表面が被覆されている
(2)前記支持体の、少なくとも前記発熱体と接する表面にある生体溶解性無機繊維が被覆されている
(3)前記発熱体が被覆されている
6.前記構成(1)において、シリカ含有処理剤が、前記支持体の少なくとも前記発熱体と接する表面から浸み込み、前記表面が硬化されている5記載の加熱装置。
7.前記構成(2)において、前記生体溶解性無機繊維がシリカ含有処理剤で被覆されている5又は6記載の加熱装置。
8.前記構成(3)において、前記発熱体がシリカ含有処理剤と同じ成分で被覆されている5~7のいずれか記載の加熱装置。
9.前記シリカが、コロイド、粉末及び金属アルコキシドから選択する1の形態のシリカ、又は2以上の形態のシリカの組み合わせである6~8のいずれか記載の加熱装置。
10.前記シリカ含有処理剤が、金属イオンを含まないキレート剤を含む6~9のいずれか記載の加熱装置。
11.前記シリカ含有処理剤が、酸を含む6~9のいずれか記載の加熱装置。
12.前記シリカが、酸性タイプコロイダルシリカ又はカチオンタイプコロイダルシリカである6~11のいずれか記載の加熱装置。
13.前記発熱体の温度が1100℃であるとき、前記生体溶解性無機繊維と前記発熱体との反応が抑制される1~12のいずれか記載の加熱装置。
14.前記生体溶解性無機繊維が加熱処理されている1~13のいずれか記載の加熱装置。
例えば、シリカ含有処理剤(例えば、流体と共に)を支持体の表面から浸み込ませて、表面の支持体(生体溶解性無機繊維)をシリカで被覆する。この処理は表面を硬化させることになり、表面からの粉落ち等も防げる。支持体(生体溶解性無機繊維)の被覆は、発熱体と接触する支持体(生体溶解性無機繊維)だけを被覆してもよく、支持体の表面全体を被覆してもよい。
図1Bの加熱装置12は、基部32と側壁34が一体形成されている他は図1Aの加熱装置10と同じである。
図1Cの加熱装置14は、発熱体20が支持体30の中に埋め込まれている。
図1A~1Cのいずれの加熱装置10,12,14も、生体溶解性無機繊維と発熱体が接触しないように構成されている(図示せず)。
SiO2とZrO2とAl2O3とTiO2の合計 50重量%~82重量%
アルカリ金属酸化物とアルカリ土類金属酸化物との合計 18重量%~50重量%
SiO2 50~82重量%
CaOとMgOとの合計 10~43重量%
SiO2 66~82重量%
CaO 1~9重量%
MgO 10~30重量%
Al2O3 3重量%以下
他の酸化物 2重量%未満
SiO2 66~82重量%(例えば、68~80重量%、70~80重量%、71~80重量%又は71~76重量%とできる)
CaO 10~34重量%(例えば、18~32重量%、20~30重量%、20~27重量%又は21~26重量%とできる)
MgO 3重量%以下(例えば、1重量%以下とできる)
Al2O3 5重量%以下(例えば3.5重量%以下、3.4重量%以下又は3重量%以下とできる。また、1重量%以上、1.1重量%以上又は2重量%以上とできる)
他の酸化物 2重量%未満
生理食塩水溶解率は、例えば、次のようにして測定される。すなわち、先ず、無機繊維を200メッシュ以下に粉砕して調製された試料1g及び生理食塩水150mLを三角フラスコ(容積300mL)に入れ、40℃のインキュベーターに設置する。次に、三角フラスコに、毎分120回転の水平振動を50時間継続して加える。その後、ろ過により得られた濾液に含有されている各元素の濃度(mg/L)をICP発光分析装置により測定する。そして、測定された各元素の濃度と、溶解前の無機繊維における各元素の含有量(質量%)と、に基づいて、生理食塩水溶解率(%)を算出する。すなわち、例えば、測定元素が、ケイ素(Si)、マグネシウム(Mg)、カルシウム(Ca)及びアルミニウム(Al)である場合には、次の式により、生理食塩水溶解率C(%)を算出する;C(%)=[ろ液量(L)×(a1+a2+a3+a4)×100]/[溶解前の無機繊維の質量(mg)×(b1+b2+b3+b4)/100]。この式において、a1、a2、a3及びa4は、それぞれ測定されたケイ素、マグネシウム、カルシウム及びアルミニウムの濃度(mg/L)であり、b1、b2、b3及びb4は、それぞれ溶解前の無機繊維におけるケイ素、マグネシウム、カルシウム及びアルミニウムの含有量(質量%)である。
結晶化温度は、未処理繊維の化学組成に応じて変化するため、一概に決定できないが、例えば、600~1300℃、600~1100℃、又は800~1000℃である。
まず、生体溶解性無機繊維から断熱部材(定形物、ボード)を製造する。通常、無機繊維、バインダー等の原料を含むスラリーを脱水成形して製造する。この部材を硬化処理してから、支持体を形成してもよいし(例えば、図1Aであれば基部と側壁を張り合わす、図1Bであれば所望の形状に加工(切削加工等)する等)、支持体を形成してから硬化処理してもよい。
図1Cの加熱装置は脱水成形時に発熱体も所定型にセットし、成形後に硬化処理して製造できる。発熱体の埋め込み位置を調整するために脱水成形を複数回行ってもよい。また、埋め込んだ後に硬化処理すると繊維へのシリカ被覆が不十分になる場合は、脱水成形工程の途中に硬化処理を行ってもよい。
上記のように処理した支持体に、発熱体を取り付けて加熱装置が完成する。
尚、本発明の装置の製法は上記に限定されず公知の方法で製造できる。
<ボード(断熱材)の製造>
生体溶解性繊維A(SiO2含有量が73質量%、CaO含有量が25質量%、MgO含有量が0.3質量%、Al2O3含有量が2質量%)(生理食塩水溶解率10.4%)90重量%と、澱粉とポリアクリルアミド(有機バインダー)、コロイダルシリカ(無機バインダー)から、縦300mm、横300mm、厚み50mm(密度250kg/m3)のボード(有機ボードという)を製造した。
さらに、生体溶解性繊維A85重量%と、コロイダルシリカとアルミナゾルとベントナイト(無機バインダー)、無機粉体(シリカ、ジルコン、炭酸カルシウム、ジルコニア、コージライト、ワラストナイト等)、アクリルアミド(有機バインダー)から、縦300mm、横300mm、厚み50mm(密度250kg/m3)のボードを(無機ボードという)製造した。
<ボード(断熱材)の製造>
生体溶解性繊維Aの代わりに、生体溶解性繊維B(SiO2含有量が76質量%、CaO含有量が4質量%、MgO含有量が18質量%、Al2O3含有量が2質量%)(生理食塩水溶解率23.1%)を用いた他は、製造例1と同様にして、有機及び無機ボードを製造した。
<ボード(断熱材)の製造>
生体溶解性繊維Aの代わりに、無機繊維C(SiO2含有量が47質量%、Al2O3含有量が52質量%)(生理食塩水溶解率0.1%)を用いた他は、製造例1と同様にして、有機及び無機ボードを製造した。
<シリカによる硬化処理>
製造例1~3で得た有機ボード及び無機ボード(密度250kg/m3)を、アルカリタイプコロイダルシリカからなる硬化液(pH約9、溶剤:水)で処理(1~10秒浸漬)して、表面を硬化し乾燥した。密度は300kg/m3であった。硬化後は強度が向上した。硬化した有機及び無機ボードの硬度を硬度計(ASKER製:アスカーゴム硬度計C型)を用いて測定した。結果を表1に示す。
<シリカによる硬化処理>
製造例2で得た有機ボード及び無機ボードを用いた他は、処理例1と同様に硬化し硬度を測定した。結果を表2に示す。
<シリカによる硬化処理>
製造例3で得た有機ボード及び無機ボードを用いた他は、処理例1と同様に硬化した。硬化後は強度が向上した。
[シリカとキレート剤による硬化処理]
<硬化処理>
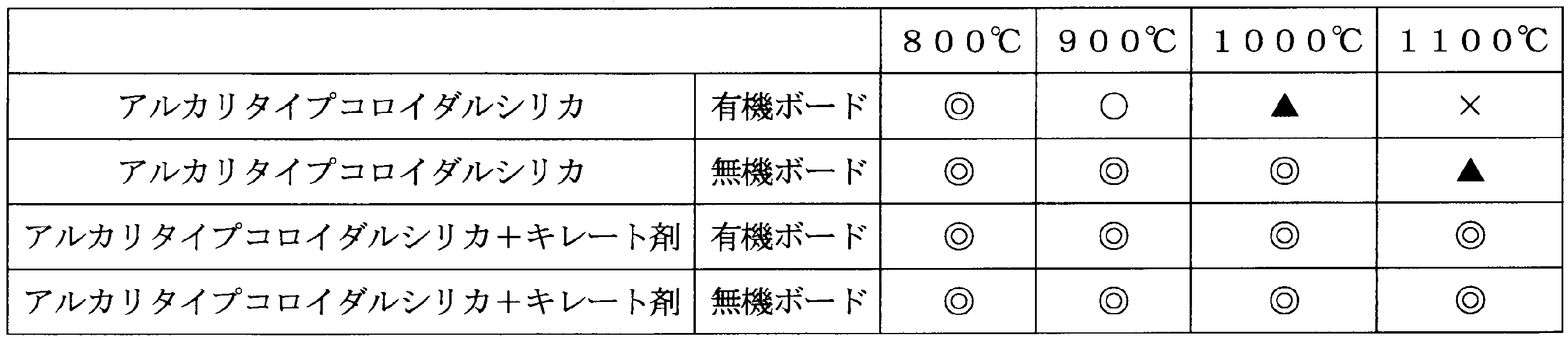
製造例1で得た有機及び無機ボードを、EDTA2アンモニウム塩とアルカリタイプコロイダルシリカからなる硬化処理液(pH約9、溶媒:水)で処理して、表面を硬化し乾燥した。キレート剤は、固形分10重量%硬化処理液に対し1重量%で用いた。
硬化した有機及び無機ボードを、800℃、900℃、1000℃、1100℃で24時間加熱して、外観(クラックの有無)を目視で観察した。比較のため製造例1で得た硬化した有機及び無機ボードも同様に加熱して観察した。正常なものを◎、表面に小さなクラックが発生したものを○、大きなクラックが発生したものを▲、割れが発生したものを×とした。結果を表3に示す。
[シリカと酸による硬化処理]
<硬化処理>
製造例1で得た有機ボードを、酸と、アルカリタイプコロイダルシリカからなる硬化処理液で処理して、表面を硬化し乾燥した。酸として、クエン酸、りんご酸、酒石酸、サリチル酸を、固形分10重量%硬化処理液に対し約1重量%で用い、処理液のpHを3~4とした。
処理例4と同様に実施した。結果を表4に示す。
[酸性タイプコロイダルシリカとカチオンタイプコロイダルシリカによる硬化処理]
<硬化処理>
製造例2で得た無機ボードを、酸性タイプコロイダルシリカ(シリカドール20A(日本化学工業(株)製)、pH2~4)又はカチオンタイプコロイダルシリカ(スノーテックスAK、日産化学工業(株)製)(硬化剤)で処理して、表面を硬化し乾燥した。
<加熱試験>
処理例4と同様に実施した。結果を表5に示す。
<硬化無機ボードの高温での安定性試験>
処理例1で得た硬化処理を施した無機ボード(断熱材)の上にヒーター線を載せ、この状態のまま電気炉内にて1000℃~1300℃の温度で8時間加熱処理を行った。加熱後のボードとヒーター線の外観観察を行い、反応の有無を確認した。ヒーター線とボードとの反応がなかったものを○、ヒーター線に対してボード繊維の付着が少量みられたものを△、ボード繊維の付着量が大きかったものを×とした。結果を表6に示す。
処理例1で得た硬化処理を施した無機ボードの代わりに、表6に示す無機ボードを使用した他は、実施例1と同様にして、ヒーター線とボードの高温での安定性を試験した。結果を表6に示す。
生体溶解性無機繊維を含む断熱材について、硬化処理を施した場合は、1300℃になるとごく微量の繊維の付着がみられたが、断熱材(繊維)とヒーター線との反応は抑制された。
この明細書に記載の文献及び本願のパリ優先の基礎となる日本出願明細書の内容を全てここに援用する。
Claims (14)
- 発熱体と、
前記発熱体を保持する、生体溶解性無機繊維を含む支持体と、
を備え、
前記生体溶解性無機繊維が前記発熱体と直接接触しないか又は前記生体溶解性無機繊維と前記発熱体との接触が低減された加熱装置。 - 前記生体溶解性無機繊維が、以下の組成である請求項1記載の加熱装置。
SiO2 50~82重量%
CaOとMgOとの合計 10~43重量% - 前記生体溶解性無機繊維が、以下の組成である請求項1又は2記載の加熱装置。
SiO2とZrO2とAl2O3とTiO2との合計 50~82重量%
アルカリ金属酸化物とアルカリ土類金属酸化物との合計 18~50重量% - 前記生体溶解性無機繊維が、以下の組成を有するMgシリケート繊維、又は以下の組成を有するCaシリケート繊維である請求項1~3のいずれか記載の加熱装置。
[Mgシリケート繊維]
SiO2 66~82重量%
CaO 1~9重量%
MgO 10~30重量%
Al2O3 3重量%以下
他の酸化物 2重量%未満
[Caシリケート繊維]
SiO2 66~82重量%
CaO 10~34重量%
MgO 3重量%以下
Al2O3 5重量%以下
他の酸化物 2重量%未満 - 以下の構成(1)~(3)のいずれかにより、前記生体溶解性無機繊維が前記発熱体と直接接触しないか又は前記生体溶解性無機繊維と前記発熱体との接触が低減された請求項1~4のいずれか記載の加熱装置。
(1)前記支持体の少なくとも前記発熱体と接する表面が被覆されている
(2)前記支持体の、少なくとも前記発熱体と接する表面にある生体溶解性無機繊維が被覆されている
(3)前記発熱体が被覆されている - 前記構成(1)において、シリカ含有処理剤が、前記支持体の少なくとも前記発熱体と接する表面から浸み込み、前記表面が硬化されている請求項5記載の加熱装置。
- 前記構成(2)において、前記生体溶解性無機繊維がシリカ含有処理剤で被覆されている請求項5又は6記載の加熱装置。
- 前記構成(3)において、前記発熱体がシリカ含有処理剤で被覆されている請求項5~7のいずれか記載の加熱装置。
- 前記シリカが、コロイド、粉末及び金属アルコキシドから選択する1の形態のシリカ、又は2以上の形態のシリカの組み合わせである請求項6~8のいずれか記載の加熱装置。
- 前記シリカ含有処理剤が、金属イオンを含まないキレート剤を含む請求項6~9のいずれか記載の加熱装置。
- 前記シリカ含有処理剤が、酸を含む請求項6~9のいずれか記載の加熱装置。
- 前記シリカが、酸性タイプコロイダルシリカ又はカチオンタイプコロイダルシリカである請求項6~11のいずれか記載の加熱装置。
- 前記発熱体の温度が1100℃であるとき、前記生体溶解性無機繊維と前記発熱体との反応が抑制される請求項1~12のいずれか記載の加熱装置。
- 前記生体溶解性無機繊維が加熱処理されている請求項1~13のいずれか記載の加熱装置。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020147031435A KR20150013513A (ko) | 2012-05-22 | 2012-11-15 | 가열장치 |
| CN201280073417.XA CN104335676B (zh) | 2012-05-22 | 2012-11-15 | 加热装置 |
| KR1020197023826A KR102183051B1 (ko) | 2012-05-22 | 2012-11-15 | 가열장치 |
| AU2012380565A AU2012380565B2 (en) | 2012-05-22 | 2012-11-15 | Heating device |
| EP12877413.0A EP2854476B1 (en) | 2012-05-22 | 2012-11-15 | Heating device and its manufacturing method |
| US14/402,543 US10143042B2 (en) | 2012-05-22 | 2012-11-15 | Heating device |
| IN9708DEN2014 IN2014DN09708A (ja) | 2012-05-22 | 2014-11-18 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-116356 | 2012-05-22 | ||
| JP2012116356A JP5277337B1 (ja) | 2012-05-22 | 2012-05-22 | 加熱装置 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013175545A1 true WO2013175545A1 (ja) | 2013-11-28 |
Family
ID=49179285
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/007321 WO2013175545A1 (ja) | 2012-05-22 | 2012-11-15 | 加熱装置 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US10143042B2 (ja) |
| EP (1) | EP2854476B1 (ja) |
| JP (1) | JP5277337B1 (ja) |
| KR (2) | KR20150013513A (ja) |
| CN (1) | CN104335676B (ja) |
| AU (1) | AU2012380565B2 (ja) |
| IN (1) | IN2014DN09708A (ja) |
| WO (1) | WO2013175545A1 (ja) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2016129723A (ru) * | 2013-12-23 | 2018-01-30 | ЮНИФРАКС АЙ ЭлЭлСи | Неорганическое волокно с улучшенными усадкой и прочностью |
| ES2744914T3 (es) | 2014-07-16 | 2020-02-26 | Unifrax I Llc | Fibra inorgánica con contracción y resistencia mejorados |
| JP5634637B1 (ja) * | 2014-08-08 | 2014-12-03 | ニチアス株式会社 | 生体溶解性無機繊維 |
| WO2019074794A1 (en) | 2017-10-10 | 2019-04-18 | Unifrax 1 Llc | INORGANIC FIBER WITH LOW BIOPERSISTANCE EXEMPT FROM CRYSTALLINE SILICA |
| US10882779B2 (en) | 2018-05-25 | 2021-01-05 | Unifrax I Llc | Inorganic fiber |
| JP7223407B2 (ja) * | 2019-03-11 | 2023-02-16 | 株式会社協同 | 加熱剤 |
| JP7127084B2 (ja) * | 2020-05-18 | 2022-08-29 | イソライト工業株式会社 | 生理食塩水に可溶な耐熱性無機繊維 |
| GB2591039B (en) | 2020-10-23 | 2021-11-24 | Thermal Ceramics Uk Ltd | Thermal insulation |
| DE102022203542A1 (de) | 2022-04-07 | 2023-10-12 | E.G.O. Elektro-Gerätebau GmbH | Wärmedämmformkörper und Verfahren zur Herstellung eines solchen Wärmedämmformkörpers |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000077167A (ja) * | 1998-08-31 | 2000-03-14 | Kyocera Corp | 面状発熱体 |
| JP2001273973A (ja) | 2000-03-27 | 2001-10-05 | Nichias Corp | 電熱ヒータ構造およびその製造方法 |
| JP2005514318A (ja) | 2002-01-04 | 2005-05-19 | ザ・モーガン・クルーシブル・カンパニー・ピーエルシー | 生理食塩水に可溶の無機繊維 |
| JP2010277917A (ja) * | 2009-05-29 | 2010-12-09 | Bridgestone Corp | 熱処理用ヒータおよびその製造方法 |
| JP2011181258A (ja) | 2010-02-26 | 2011-09-15 | Nichias Corp | 加熱装置 |
| WO2012049858A1 (ja) * | 2010-10-14 | 2012-04-19 | ニチアス株式会社 | 無機繊維質成形体及びその製造方法並びに加熱設備 |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2887557A (en) * | 1954-06-29 | 1959-05-19 | Stettner & Co Fabrik Elektro K | Silicones |
| US2880298A (en) * | 1958-06-02 | 1959-03-31 | Gen Electric | Electric heating units |
| DE2363790C3 (de) * | 1973-12-21 | 1981-12-17 | Dynamit Nobel Ag, 5210 Troisdorf | Verfahren zur Herstellung einer wärmeleitenden, hochfeuerfesten, elektrisch isolierenden Einbettungsmasse für elektrische Heizkörper |
| US4087777A (en) * | 1973-12-21 | 1978-05-02 | Dynamit Nobel Aktiengesellschaft | Electrical heating assembly having a thermally conductive refractory electrical insulating embedding composition between an electrically conductive member and a jacket |
| JP2973162B2 (ja) * | 1994-03-30 | 1999-11-08 | ニチアス株式会社 | 溶融金属保持容器用熱板 |
| US6551951B1 (en) * | 1999-03-19 | 2003-04-22 | Johns Manville International, Inc. | Burn through resistant nonwoven mat, barrier, and insulation system |
| JP2006086054A (ja) * | 2004-09-17 | 2006-03-30 | Nichias Corp | 発熱構造体及びその製造方法 |
| CN101115688B (zh) | 2004-12-13 | 2013-03-20 | 3M创新有限公司 | 安装垫和使用该安装垫的污染控制装置 |
| JP5014779B2 (ja) | 2006-12-28 | 2012-08-29 | ニチアス株式会社 | 無機繊維質成形体、無機繊維質焼成体、不定形無機繊維質組成物及び不定形無機繊維質焼成体 |
| JP5015193B2 (ja) * | 2009-04-30 | 2012-08-29 | 強化土株式会社 | 地盤注入材および地盤注入工法 |
| JP5526964B2 (ja) * | 2010-04-15 | 2014-06-18 | 三菱化学株式会社 | 透明面状発熱積層体 |
| WO2011159914A2 (en) * | 2010-06-16 | 2011-12-22 | Unifrax I Llc | Thermally stable inorganic fibers for exhaust gas treatment device insulating mat |
| JP4902797B1 (ja) * | 2011-03-30 | 2012-03-21 | ニチアス株式会社 | 湿潤ブランケット |
-
2012
- 2012-05-22 JP JP2012116356A patent/JP5277337B1/ja active Active
- 2012-11-15 US US14/402,543 patent/US10143042B2/en active Active
- 2012-11-15 KR KR1020147031435A patent/KR20150013513A/ko active Application Filing
- 2012-11-15 KR KR1020197023826A patent/KR102183051B1/ko active IP Right Grant
- 2012-11-15 EP EP12877413.0A patent/EP2854476B1/en active Active
- 2012-11-15 AU AU2012380565A patent/AU2012380565B2/en not_active Ceased
- 2012-11-15 CN CN201280073417.XA patent/CN104335676B/zh active Active
- 2012-11-15 WO PCT/JP2012/007321 patent/WO2013175545A1/ja active Application Filing
-
2014
- 2014-11-18 IN IN9708DEN2014 patent/IN2014DN09708A/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000077167A (ja) * | 1998-08-31 | 2000-03-14 | Kyocera Corp | 面状発熱体 |
| JP2001273973A (ja) | 2000-03-27 | 2001-10-05 | Nichias Corp | 電熱ヒータ構造およびその製造方法 |
| JP2005514318A (ja) | 2002-01-04 | 2005-05-19 | ザ・モーガン・クルーシブル・カンパニー・ピーエルシー | 生理食塩水に可溶の無機繊維 |
| JP2010277917A (ja) * | 2009-05-29 | 2010-12-09 | Bridgestone Corp | 熱処理用ヒータおよびその製造方法 |
| JP2011181258A (ja) | 2010-02-26 | 2011-09-15 | Nichias Corp | 加熱装置 |
| WO2012049858A1 (ja) * | 2010-10-14 | 2012-04-19 | ニチアス株式会社 | 無機繊維質成形体及びその製造方法並びに加熱設備 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2854476A4 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN104335676B (zh) | 2016-05-25 |
| EP2854476B1 (en) | 2021-04-07 |
| AU2012380565B2 (en) | 2015-05-14 |
| CN104335676A (zh) | 2015-02-04 |
| US20150163861A1 (en) | 2015-06-11 |
| AU2012380565A1 (en) | 2014-11-27 |
| KR20190097330A (ko) | 2019-08-20 |
| US10143042B2 (en) | 2018-11-27 |
| EP2854476A4 (en) | 2016-02-10 |
| KR20150013513A (ko) | 2015-02-05 |
| IN2014DN09708A (ja) | 2015-07-31 |
| JP2013243071A (ja) | 2013-12-05 |
| JP5277337B1 (ja) | 2013-08-28 |
| EP2854476A1 (en) | 2015-04-01 |
| KR102183051B1 (ko) | 2020-11-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2013175545A1 (ja) | 加熱装置 | |
| JP4975179B2 (ja) | 無機繊維質成形体及びその製造方法並びに加熱設備 | |
| KR101390509B1 (ko) | 경화 정형물 | |
| US9434656B2 (en) | Wet blanket | |
| KR20130124398A (ko) | 무기 섬유질페이퍼 및 그 제조 방법 및 설비 | |
| JP5634637B1 (ja) | 生体溶解性無機繊維 | |
| WO2013080455A1 (ja) | 不定形組成物 | |
| JP2013243120A (ja) | 加熱装置 | |
| WO2015011930A1 (ja) | 耐熱無機繊維 | |
| JP5138806B1 (ja) | 生体溶解性無機繊維及びその製造方法 | |
| JP6386232B2 (ja) | 表面改質無機繊維及びその製造方法 | |
| WO2018047801A1 (ja) | 無機繊維質成形体 | |
| WO2013183241A1 (ja) | 生理食塩水に可溶なAl-Ca系無機繊維及びその組成物 | |
| WO2014020842A1 (ja) | Si/Al/Ca含有無機繊維 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12877413 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20147031435 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012877413 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14402543 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2012380565 Country of ref document: AU Date of ref document: 20121115 Kind code of ref document: A |